WO2014004697A2 - Method of removing inorganic scales - Google Patents
Method of removing inorganic scales Download PDFInfo
- Publication number
- WO2014004697A2 WO2014004697A2 PCT/US2013/047938 US2013047938W WO2014004697A2 WO 2014004697 A2 WO2014004697 A2 WO 2014004697A2 US 2013047938 W US2013047938 W US 2013047938W WO 2014004697 A2 WO2014004697 A2 WO 2014004697A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- carbon atoms
- treatment composition
- acid
- well treatment
- Prior art date
Links
- 238000000034 method Methods 0.000 title claims description 40
- 239000000203 mixture Substances 0.000 claims abstract description 78
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 claims abstract description 67
- 230000015572 biosynthetic process Effects 0.000 claims abstract description 63
- 238000011282 treatment Methods 0.000 claims abstract description 62
- 239000002253 acid Substances 0.000 claims abstract description 60
- 150000001875 compounds Chemical class 0.000 claims abstract description 32
- 150000003242 quaternary ammonium salts Chemical class 0.000 claims abstract description 28
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 claims abstract description 25
- 150000003839 salts Chemical class 0.000 claims abstract description 22
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims abstract description 19
- 229910052796 boron Inorganic materials 0.000 claims abstract description 19
- 150000001282 organosilanes Chemical class 0.000 claims abstract description 19
- 150000002148 esters Chemical class 0.000 claims abstract description 13
- 229930195733 hydrocarbon Natural products 0.000 claims abstract description 7
- 239000004215 Carbon black (E152) Substances 0.000 claims abstract description 4
- 125000004432 carbon atom Chemical group C* 0.000 claims description 40
- 229910052739 hydrogen Inorganic materials 0.000 claims description 25
- 239000001257 hydrogen Substances 0.000 claims description 25
- -1 aminoalkyl siloxane Chemical class 0.000 claims description 24
- 125000000217 alkyl group Chemical group 0.000 claims description 21
- 239000004927 clay Substances 0.000 claims description 16
- 150000002431 hydrogen Chemical group 0.000 claims description 14
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 13
- 229910052736 halogen Inorganic materials 0.000 claims description 13
- 150000002367 halogens Chemical class 0.000 claims description 13
- 229920006395 saturated elastomer Polymers 0.000 claims description 12
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 10
- 150000001450 anions Chemical group 0.000 claims description 10
- 239000011734 sodium Chemical group 0.000 claims description 10
- 229910052708 sodium Inorganic materials 0.000 claims description 10
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 claims description 9
- 229910000077 silane Inorganic materials 0.000 claims description 9
- 230000008961 swelling Effects 0.000 claims description 9
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical group [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 claims description 8
- 125000002947 alkylene group Chemical group 0.000 claims description 8
- 239000011591 potassium Chemical group 0.000 claims description 8
- 229910052700 potassium Inorganic materials 0.000 claims description 8
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical group C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims description 7
- 150000001412 amines Chemical class 0.000 claims description 7
- 125000003118 aryl group Chemical group 0.000 claims description 7
- 229910001634 calcium fluoride Inorganic materials 0.000 claims description 7
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 7
- 229910019142 PO4 Inorganic materials 0.000 claims description 6
- ORUIBWPALBXDOA-UHFFFAOYSA-L magnesium fluoride Chemical compound [F-].[F-].[Mg+2] ORUIBWPALBXDOA-UHFFFAOYSA-L 0.000 claims description 6
- 229910001635 magnesium fluoride Inorganic materials 0.000 claims description 6
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 claims description 6
- 239000010452 phosphate Substances 0.000 claims description 6
- PTMHPRAIXMAOOB-UHFFFAOYSA-L phosphoramidate Chemical compound NP([O-])([O-])=O PTMHPRAIXMAOOB-UHFFFAOYSA-L 0.000 claims description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical group [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 claims description 5
- 150000003863 ammonium salts Chemical class 0.000 claims description 5
- 150000001639 boron compounds Chemical class 0.000 claims description 5
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 claims description 5
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 claims description 5
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 5
- 230000002401 inhibitory effect Effects 0.000 claims description 5
- RILZRCJGXSFXNE-UHFFFAOYSA-N 2-[4-(trifluoromethoxy)phenyl]ethanol Chemical compound OCCC1=CC=C(OC(F)(F)F)C=C1 RILZRCJGXSFXNE-UHFFFAOYSA-N 0.000 claims description 4
- 229910002651 NO3 Inorganic materials 0.000 claims description 4
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 claims description 4
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 4
- 125000002252 acyl group Chemical group 0.000 claims description 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 4
- 150000001768 cations Chemical class 0.000 claims description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 4
- JZMJDSHXVKJFKW-UHFFFAOYSA-M methyl sulfate(1-) Chemical compound COS([O-])(=O)=O JZMJDSHXVKJFKW-UHFFFAOYSA-M 0.000 claims description 4
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 claims description 3
- ZMZDMBWJUHKJPS-UHFFFAOYSA-M Thiocyanate anion Chemical compound [S-]C#N ZMZDMBWJUHKJPS-UHFFFAOYSA-M 0.000 claims description 3
- 150000008051 alkyl sulfates Chemical class 0.000 claims description 3
- 150000004820 halides Chemical class 0.000 claims description 3
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N hydrogen thiocyanate Natural products SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 claims description 3
- 125000004356 hydroxy functional group Chemical group O* 0.000 claims description 3
- 239000011159 matrix material Substances 0.000 claims description 3
- 125000005156 substituted alkylene group Chemical group 0.000 claims description 3
- 150000007942 carboxylates Chemical class 0.000 claims description 2
- 230000002708 enhancing effect Effects 0.000 claims description 2
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 2
- 229910001506 inorganic fluoride Inorganic materials 0.000 claims description 2
- 230000000246 remedial effect Effects 0.000 claims description 2
- 125000000547 substituted alkyl group Chemical group 0.000 claims description 2
- PTMHPRAIXMAOOB-UHFFFAOYSA-N phosphoramidic acid Chemical compound NP(O)(O)=O PTMHPRAIXMAOOB-UHFFFAOYSA-N 0.000 claims 3
- 238000005086 pumping Methods 0.000 claims 3
- XLJMAIOERFSOGZ-UHFFFAOYSA-M cyanate Chemical compound [O-]C#N XLJMAIOERFSOGZ-UHFFFAOYSA-M 0.000 claims 2
- VLTRZXGMWDSKGL-UHFFFAOYSA-M perchlorate Inorganic materials [O-]Cl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-M 0.000 claims 2
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 claims 2
- 229910017048 AsF6 Inorganic materials 0.000 claims 1
- 125000004103 aminoalkyl group Chemical group 0.000 claims 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims 1
- 125000001183 hydrocarbyl group Chemical group 0.000 claims 1
- 230000000149 penetrating effect Effects 0.000 claims 1
- 238000005755 formation reaction Methods 0.000 abstract description 55
- 150000002430 hydrocarbons Chemical class 0.000 abstract description 6
- 230000035699 permeability Effects 0.000 description 31
- 239000000243 solution Substances 0.000 description 21
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 15
- 239000012530 fluid Substances 0.000 description 14
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 12
- 101000618110 Homo sapiens Sperm-associated antigen 7 Proteins 0.000 description 11
- 102100021912 Sperm-associated antigen 7 Human genes 0.000 description 11
- 239000004327 boric acid Substances 0.000 description 11
- 238000002347 injection Methods 0.000 description 11
- 239000007924 injection Substances 0.000 description 11
- DDFHBQSCUXNBSA-UHFFFAOYSA-N 5-(5-carboxythiophen-2-yl)thiophene-2-carboxylic acid Chemical compound S1C(C(=O)O)=CC=C1C1=CC=C(C(O)=O)S1 DDFHBQSCUXNBSA-UHFFFAOYSA-N 0.000 description 10
- MIMUSZHMZBJBPO-UHFFFAOYSA-N 6-methoxy-8-nitroquinoline Chemical compound N1=CC=CC2=CC(OC)=CC([N+]([O-])=O)=C21 MIMUSZHMZBJBPO-UHFFFAOYSA-N 0.000 description 10
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 9
- 238000004519 manufacturing process Methods 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 7
- 230000005012 migration Effects 0.000 description 7
- 238000013508 migration Methods 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- 238000002474 experimental method Methods 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 5
- 239000010459 dolomite Substances 0.000 description 5
- 229910000514 dolomite Inorganic materials 0.000 description 5
- 229910052500 inorganic mineral Inorganic materials 0.000 description 5
- 239000011707 mineral Substances 0.000 description 5
- 125000003342 alkenyl group Chemical group 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 230000005764 inhibitory process Effects 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 4
- 239000011148 porous material Substances 0.000 description 4
- 239000010453 quartz Substances 0.000 description 4
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical group OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 239000011575 calcium Substances 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000005553 drilling Methods 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 238000005067 remediation Methods 0.000 description 3
- 239000004576 sand Substances 0.000 description 3
- 150000004760 silicates Chemical class 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- RPAJSBKBKSSMLJ-DFWYDOINSA-N (2s)-2-aminopentanedioic acid;hydrochloride Chemical class Cl.OC(=O)[C@@H](N)CCC(O)=O RPAJSBKBKSSMLJ-DFWYDOINSA-N 0.000 description 2
- WYTZZXDRDKSJID-UHFFFAOYSA-N (3-aminopropyl)triethoxysilane Chemical compound CCO[Si](OCC)(OCC)CCCN WYTZZXDRDKSJID-UHFFFAOYSA-N 0.000 description 2
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 2
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 2
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 2
- 229910011255 B2O3 Inorganic materials 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- DBVJJBKOTRCVKF-UHFFFAOYSA-N Etidronic acid Chemical compound OP(=O)(O)C(O)(C)P(O)(O)=O DBVJJBKOTRCVKF-UHFFFAOYSA-N 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 2
- 206010016825 Flushing Diseases 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N Lactic Acid Natural products CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- 238000002441 X-ray diffraction Methods 0.000 description 2
- 238000010306 acid treatment Methods 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 150000004703 alkoxides Chemical class 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 229910001919 chlorite Inorganic materials 0.000 description 2
- 229910052619 chlorite group Inorganic materials 0.000 description 2
- QBWCMBCROVPCKQ-UHFFFAOYSA-N chlorous acid Chemical compound OCl=O QBWCMBCROVPCKQ-UHFFFAOYSA-N 0.000 description 2
- 230000007797 corrosion Effects 0.000 description 2
- 238000005260 corrosion Methods 0.000 description 2
- 230000001186 cumulative effect Effects 0.000 description 2
- 125000003700 epoxy group Chemical group 0.000 description 2
- 239000010433 feldspar Substances 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 2
- 229910052622 kaolinite Inorganic materials 0.000 description 2
- CYPPCCJJKNISFK-UHFFFAOYSA-J kaolinite Chemical compound [OH-].[OH-].[OH-].[OH-].[Al+3].[Al+3].[O-][Si](=O)O[Si]([O-])=O CYPPCCJJKNISFK-UHFFFAOYSA-J 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 150000003009 phosphonic acids Chemical class 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000002244 precipitate Substances 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- VZSRBBMJRBPUNF-UHFFFAOYSA-N 2-(2,3-dihydro-1H-inden-2-ylamino)-N-[3-oxo-3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)propyl]pyrimidine-5-carboxamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C(=O)NCCC(N1CC2=C(CC1)NN=N2)=O VZSRBBMJRBPUNF-UHFFFAOYSA-N 0.000 description 1
- 239000001763 2-hydroxyethyl(trimethyl)azanium Substances 0.000 description 1
- BJLAHYUZLVYPAA-UHFFFAOYSA-N 4-(3-aminophenyl)benzenesulfonamide Chemical compound NC=1C=C(C=CC=1)C1=CC=C(C=C1)S(=O)(=O)N BJLAHYUZLVYPAA-UHFFFAOYSA-N 0.000 description 1
- QISOBCMNUJQOJU-UHFFFAOYSA-N 4-bromo-1h-pyrazole-5-carboxylic acid Chemical compound OC(=O)C=1NN=CC=1Br QISOBCMNUJQOJU-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 229910015444 B(OH)3 Inorganic materials 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 0 CC(C1)(C1O*)N(C)N Chemical compound CC(C1)(C1O*)N(C)N 0.000 description 1
- 235000019743 Choline chloride Nutrition 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Natural products OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 101100409194 Rattus norvegicus Ppargc1b gene Proteins 0.000 description 1
- 229910018540 Si C Inorganic materials 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 1
- YDONNITUKPKTIG-UHFFFAOYSA-N [Nitrilotris(methylene)]trisphosphonic acid Chemical compound OP(O)(=O)CN(CP(O)(O)=O)CP(O)(O)=O YDONNITUKPKTIG-UHFFFAOYSA-N 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- DLHONNLASJQAHX-UHFFFAOYSA-N aluminum;potassium;oxygen(2-);silicon(4+) Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[Al+3].[Si+4].[Si+4].[Si+4].[K+] DLHONNLASJQAHX-UHFFFAOYSA-N 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- ZPECUSGQPIKHLT-UHFFFAOYSA-N bis(ethenyl)-dimethoxysilane Chemical compound CO[Si](OC)(C=C)C=C ZPECUSGQPIKHLT-UHFFFAOYSA-N 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- FQEKAFQSVPLXON-UHFFFAOYSA-N butyl(trichloro)silane Chemical compound CCCC[Si](Cl)(Cl)Cl FQEKAFQSVPLXON-UHFFFAOYSA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- PDNUHAXBKKDGAM-UHFFFAOYSA-N chloro-diethyl-methylsilane Chemical compound CC[Si](C)(Cl)CC PDNUHAXBKKDGAM-UHFFFAOYSA-N 0.000 description 1
- LFJMRJHKLLWKAN-UHFFFAOYSA-N chloro-dimethoxy-propylsilane Chemical compound CCC[Si](Cl)(OC)OC LFJMRJHKLLWKAN-UHFFFAOYSA-N 0.000 description 1
- SGMZJAMFUVOLNK-UHFFFAOYSA-M choline chloride Chemical compound [Cl-].C[N+](C)(C)CCO SGMZJAMFUVOLNK-UHFFFAOYSA-M 0.000 description 1
- 229960003178 choline chloride Drugs 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- JKWMSGQKBLHBQQ-UHFFFAOYSA-N diboron trioxide Chemical compound O=BOB=O JKWMSGQKBLHBQQ-UHFFFAOYSA-N 0.000 description 1
- LIQOCGKQCFXKLF-UHFFFAOYSA-N dibromo(dimethyl)silane Chemical compound C[Si](C)(Br)Br LIQOCGKQCFXKLF-UHFFFAOYSA-N 0.000 description 1
- OSXYHAQZDCICNX-UHFFFAOYSA-N dichloro(diphenyl)silane Chemical compound C=1C=CC=CC=1[Si](Cl)(Cl)C1=CC=CC=C1 OSXYHAQZDCICNX-UHFFFAOYSA-N 0.000 description 1
- UOZZKLIPYZQXEP-UHFFFAOYSA-N dichloro(dipropyl)silane Chemical compound CCC[Si](Cl)(Cl)CCC UOZZKLIPYZQXEP-UHFFFAOYSA-N 0.000 description 1
- JWCYDYZLEAQGJJ-UHFFFAOYSA-N dicyclopentyl(dimethoxy)silane Chemical compound C1CCCC1[Si](OC)(OC)C1CCCC1 JWCYDYZLEAQGJJ-UHFFFAOYSA-N 0.000 description 1
- GPTNEHSWPUSHFX-UHFFFAOYSA-N diethyl(diiodo)silane Chemical compound CC[Si](I)(I)CC GPTNEHSWPUSHFX-UHFFFAOYSA-N 0.000 description 1
- YQGOWXYZDLJBFL-UHFFFAOYSA-N dimethoxysilane Chemical compound CO[SiH2]OC YQGOWXYZDLJBFL-UHFFFAOYSA-N 0.000 description 1
- UQGFMSUEHSUPRD-UHFFFAOYSA-N disodium;3,7-dioxido-2,4,6,8,9-pentaoxa-1,3,5,7-tetraborabicyclo[3.3.1]nonane Chemical compound [Na+].[Na+].O1B([O-])OB2OB([O-])OB1O2 UQGFMSUEHSUPRD-UHFFFAOYSA-N 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- DUYCTCQXNHFCSJ-UHFFFAOYSA-N dtpmp Chemical compound OP(=O)(O)CN(CP(O)(O)=O)CCN(CP(O)(=O)O)CCN(CP(O)(O)=O)CP(O)(O)=O DUYCTCQXNHFCSJ-UHFFFAOYSA-N 0.000 description 1
- FWDBOZPQNFPOLF-UHFFFAOYSA-N ethenyl(triethoxy)silane Chemical compound CCO[Si](OCC)(OCC)C=C FWDBOZPQNFPOLF-UHFFFAOYSA-N 0.000 description 1
- 239000004088 foaming agent Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 239000003349 gelling agent Substances 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 229910000040 hydrogen fluoride Inorganic materials 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229910052900 illite Inorganic materials 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 230000033001 locomotion Effects 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 150000001455 metallic ions Chemical class 0.000 description 1
- JSWYYQXCMPNWMB-UHFFFAOYSA-N methoxy-di(propan-2-yl)silane Chemical compound CO[SiH](C(C)C)C(C)C JSWYYQXCMPNWMB-UHFFFAOYSA-N 0.000 description 1
- 239000005055 methyl trichlorosilane Substances 0.000 description 1
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 1
- JLUFWMXJHAVVNN-UHFFFAOYSA-N methyltrichlorosilane Chemical compound C[Si](Cl)(Cl)Cl JLUFWMXJHAVVNN-UHFFFAOYSA-N 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- LNOPIUAQISRISI-UHFFFAOYSA-N n'-hydroxy-2-propan-2-ylsulfonylethanimidamide Chemical compound CC(C)S(=O)(=O)CC(N)=NO LNOPIUAQISRISI-UHFFFAOYSA-N 0.000 description 1
- VGIBGUSAECPPNB-UHFFFAOYSA-L nonaaluminum;magnesium;tripotassium;1,3-dioxido-2,4,5-trioxa-1,3-disilabicyclo[1.1.1]pentane;iron(2+);oxygen(2-);fluoride;hydroxide Chemical compound [OH-].[O-2].[O-2].[O-2].[O-2].[O-2].[F-].[Mg+2].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[K+].[K+].[K+].[Fe+2].O1[Si]2([O-])O[Si]1([O-])O2.O1[Si]2([O-])O[Si]1([O-])O2.O1[Si]2([O-])O[Si]1([O-])O2.O1[Si]2([O-])O[Si]1([O-])O2.O1[Si]2([O-])O[Si]1([O-])O2.O1[Si]2([O-])O[Si]1([O-])O2.O1[Si]2([O-])O[Si]1([O-])O2 VGIBGUSAECPPNB-UHFFFAOYSA-L 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- VGTPKLINSHNZRD-UHFFFAOYSA-N oxoborinic acid Chemical compound OB=O VGTPKLINSHNZRD-UHFFFAOYSA-N 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 229920002851 polycationic polymer Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000009877 rendering Methods 0.000 description 1
- 239000011435 rock Substances 0.000 description 1
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 1
- FZHAPNGMFPVSLP-UHFFFAOYSA-N silanamine Chemical class [SiH3]N FZHAPNGMFPVSLP-UHFFFAOYSA-N 0.000 description 1
- 150000004756 silanes Chemical class 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- FDNAPBUWERUEDA-UHFFFAOYSA-N silicon tetrachloride Chemical compound Cl[Si](Cl)(Cl)Cl FDNAPBUWERUEDA-UHFFFAOYSA-N 0.000 description 1
- 239000010802 sludge Substances 0.000 description 1
- 229910021647 smectite Inorganic materials 0.000 description 1
- 239000004328 sodium tetraborate Substances 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- LGQXXHMEBUOXRP-UHFFFAOYSA-N tributyl borate Chemical compound CCCCOB(OCCCC)OCCCC LGQXXHMEBUOXRP-UHFFFAOYSA-N 0.000 description 1
- FRGPKMWIYVTFIQ-UHFFFAOYSA-N triethoxy(3-isocyanatopropyl)silane Chemical compound CCO[Si](OCC)(OCC)CCCN=C=O FRGPKMWIYVTFIQ-UHFFFAOYSA-N 0.000 description 1
- CPUDPFPXCZDNGI-UHFFFAOYSA-N triethoxy(methyl)silane Chemical compound CCO[Si](C)(OCC)OCC CPUDPFPXCZDNGI-UHFFFAOYSA-N 0.000 description 1
- 239000003180 well treatment fluid Substances 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/52—Compositions for preventing, limiting or eliminating depositions, e.g. for cleaning
- C09K8/528—Compositions for preventing, limiting or eliminating depositions, e.g. for cleaning inorganic depositions, e.g. sulfates or carbonates
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2208/00—Aspects relating to compositions of drilling or well treatment fluids
- C09K2208/12—Swell inhibition, i.e. using additives to drilling or well treatment fluids for inhibiting clay or shale swelling or disintegrating
Definitions
- TITLE METHOD OF REMOVING INORGANIC SCALES
- the invention relates to a method of enhancing the productivity of a hydrocarbon bearing siliceous or calcareous formation by use of a well treatment composition which contains a phosphonate acid, ester or salt, a hydrofluoric acid source, a quaternary ammonium salt and an orga.nosila.ne.
- the swelling and migration of formation clay particles is often increased when formation days are disturbed by foreign substances, such as aqueous well treatment fluids.
- the swelling and migration of formation clay reduces the permeability of the formation by obstructing the formation capillaries, resulting in a loss of formation permeability and significant reduction in the flow rate of hydrocarbons.
- the use of clay stabilizers is required during treatment operations in order to control any change or movement of the clay.
- such additives are useful in the control of fines generation which further contribute to a reduction in permeability, It is known that permeability impairment may also be improved by injecting acid, formulations containing HF into the formation.
- sandstone formation are composed of over 70% sand quartz, i.e. silica, bonded together by various amount of cementing material including carbonate, dolomite and silicates. Suitable silicates include clays and feldspars.
- a common method of treating sandstone formations involves introducing hydrofluoric acid into the wellbore and allowing the hydrofluoric acid to react with the surrounding formation. Hydrofluoric acid exhibits high reactivity towards siliceous minerals, such as clays and quartz fines.
- hydrofluoric acid reacts very quickly with authi genie clays, such as smectite, kaolinite, illite and chlorite, especially at temperatures above 150°F.
- authi genie clays such as smectite, kaolinite, illite and chlorite, especially at temperatures above 150°F.
- hydrofluoric acid is capable of attacking and dissolving siliceous minerals.
- Subterranean sandstone or siliceous formations and calcareous formations penetrated by oil, gas or geothermal wells may be treated, with an aqueous well treatment composition containing a hydrofluoric acid source in combination with an organos lane, a quaternary ammonium salt and a phosphonate acid, ester or salt.
- the well treatment composition may further contain a boron containing compound for forming a BF 4 complex.
- the aqueous well treatment composition aids in the inhibition of inorganic scales and in most instances the prevention of formation of the undesirable scales. Additionally, it will minimize corrosion potential on downhole metal tubulars.
- Such compositions have been shown to increase the permeability of the formation being treated by inhibiting or preventing the formation of undesirable inorganic scales, such as calcium fluoride, magnesium fluoride, potassium fluorosilicate, sodium fluorosilicate, fluoroa laminate, etc. As a result, production from the formation is increased or improved.
- hydrofluoric acid source may be hydrofluoric acid
- an excess of ammonium bifluoride or ammonium fluoride is used such that all of the hydrochloric acid is consumed in the production of hydrofluoric acid, leaving a small amount of unconverted ammonium bifluoride or ammonium fluoride.
- the boron containing compound is preferably fluoroboric acid or a boron compound which is capable of being hydrolyzed to form a BF 4 " complex when exposed to F or a hydrofluoric acid source.
- the phosphonate of the well treatment composition is preferably a phosphonate acid, ester or salt thereof, such as those of the formula:
- R3 O— R5 w r herein Rl , R2 and R3 are independently selected from hydrogen, alkyl, aryl. phosphonie, phosphonate, phosphate, aminophosphonic, aminophosphonate, acyl, amine, hydroxy and. carboxyl groups and salts thereof and R4 and R.5 are independently selected from hydrogen, sodium, potassium, ammonium or an organic radical.
- the presence of the combination of the organosilane and the quaternary ammonium salt in the well treatment composition further provides for greater control of clay migration and inhibits or prevents swelling. Further, the presence of the combination of the organosilane and the quaternary ammonium salt inhibits or prevents formation fines from becoming dispersed, in the well treatment composition. In addition, this combination aids in the inhibition and thus the control of scale formation.
- the pH of the well treatment composition is typically maintained at a range of from about 0 to about 3.0.
- the permeability of sandstone or siliceous formations and calcareous formations is increased during acid treatment of the well by including in an aqueous well treatment composition a synergistic amount of an organosilane and a quaternary ammonium salt.
- the aqueous well treatment composition further contains a hydrofluoric acid source and a phosphonate compound.
- the combination of the organosilane and the quaternary ammonium salt acts synergistically to control the swelling and migration of clay within the formation and to minimize the generation of fines.
- the effect imparted by the combination of the quaternary ammonium salt and the organosilane is substantially greater than the effect imparted by either component when used individually in an otherwise identical well treatment composition.
- the combination of the quaternary ammonium salt and organosilane acts to retain the naturally occurring clay platelets in the formation in position by controlling the charge and electrolytic characteristics of the treatment fluid and thus substantially reduces or eliminates clay and formation fines from becoming dispersed and plugging the formation matrix.
- the aqueous well treatment composition may further contain a boron containing compound.
- the boron containing compound principally functions to inhibit or prevent the formation of fluoride scales or to remove such scales from wellbores, screens or other equipment and/or pipelines.
- the boron containing compound is fluoroboric acid or tetrafluoro boric acid of the formula BF 4 ⁇ I- .
- the boron containing compound may further be an acid soluble boric acid and/or an organic boron containing compound, including those which are capable of forming a BF ⁇ complex when hydrolyzed and exposed to F " or HF containing solution.
- the reaction, where the boron containing compound is boric acid, may be represented by the equation:
- BF 4 ⁇ controls the concentration of active HF at any given time.
- Borate esters further acid hydrolyze to boric acid which tender the BF 4 complex, as set forth by equation (I) above. Hydrolysis may not occur, however, until higher than ambient temperatures are reached. For instance, hydrolysis may not occur until formation temperature is reached or sufficient heat is generated from the acid reaction.
- Suitable boron containing compounds include boric acid, H 3 B0 3 as well as esters of boric acid. Preferred as the boron containing compounds are those of the formula R6R 7 R 8 B0 3 wherein each of Re, R?
- Rg are independently hydrogen or a unsubstituted or substituted alkyl or alkylene group, and is preferably independently selected from hydrogen or C 1 -C4 alkyl group, optionally substituted with one or more -OH groups.
- Preferred boron compounds include tributyl borate which is very moisture sensitive.
- tetraborates such as sodium tetraborate.
- Boric oxide, B 2 0 3 , metaboric acid and HB0 2 are further preferred since they easily hydrolyze to boric acid, B(OH) 3 .
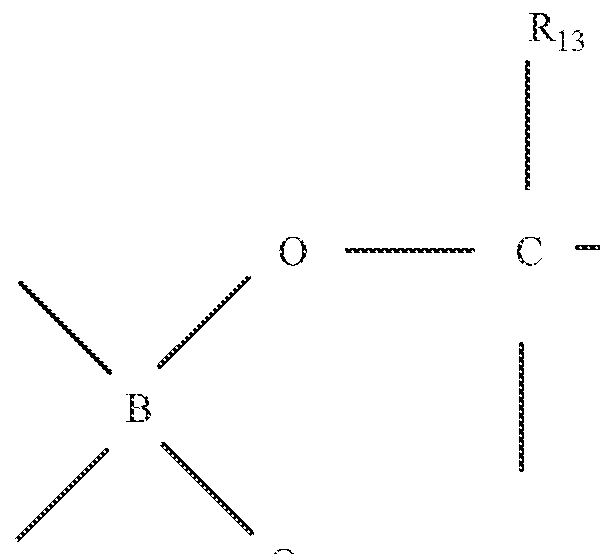
- boron containing compounds are cyclic borate esters, such as those of the formula:
- each of R9, R ] 0 , R; ;, R12, Ri 3 , R 14 , R15 and R !6 is independently selected from hydrogen or a substituted or unsubstituted alkyl or alkenvl group, and is preferably independently selected from hydrogen or a C1-C4 alkyl group, optionally substituted with one or more -OH groups or ORo (which can readily cleave to form the desired BF4 " complex), wherein R 13 is a C1-C alkyl or aryl group.
- Suitable esters include those formed with salicyclic acid or acetic acid.
- Other cyclic borates include CH 3 B 3 O 3 which hydrolyze rapidly in water.
- the amount of boron containing compound in the well treating composition is that sufficient to impart to the composition between from about 0,5 to about 10 g of BF 4 ⁇ complex per 100 cc of phosphonate, hydrofluoric acid source and water.
- the hydrofluoric acid source useful in the formation of the BF 4 " complex may ⁇ be hydrofluoric acid. More typically, however, the hydrofluoric acid source is the combination of a mineral acid and ammonium bifluoride or ammonium fluoride. Reaction of the acid with the ammonium bifluoride or ammonium fluoride renders HF. The use of the combination of acid and ammonium bifluoride or am.moni.um fluoride and boric acid, to control hydrogen fluoride significantly slows the hydrofluoric acid reaction rate.
- Preferred as the acid is hydrochloric acid, though other acids such as citric, c oroacetic, methanesuffonic, sulfuric, sulfamic, nitric, acetic, lactic, fumaric and formic acid may also be used.
- Preferred organic acids include citric acid, acetic acid and formic acid.
- a relarder may also be used, such as an aluminum salt.
- ammonium bifluoride or ammonium fluoride ydrolyzes and is converted to hydrofluoric acid.
- ammonium bifluoride or ammonium fluoride is used as a source of hydrofluoric acid, typically less acid is present than is necessary to hydrolyze all of the ammonium bifluoride or ammonium fluoride. Thus, there remains some unconverted ammonium bifluoride or ammonium fluoride in the composition.
- the hydrofluoric acid source of the aqueous well treatment composition generally provides between from about 0.25 to about .10, typically between from about 1.0 to about 6.0, weight percent of hydrofluoric acid to the well treatment composition (based on the total weight of the well treatment composition).
- the well treatment composition may further contain between from about 1 to about 50 weight percent of organic acid, preferably about 10 weight percent based on the total weight of the well treatment composition.
- the phosphonate compound principally functions as a stabilizer.
- the phosphonate compound may be a polyphosphonic acid and their salts and esters and is preferably a phosphonate acid, salt or ester thereof.
- Rl , R2 and R3 are independently selected from hydrogen, alkyl, aryl, phosphonic, phosphonate, phosphate, aminophosphonic, aminophosphonate, acyf, amine, hydroxy and carboxyl groups and salts thereof and R4 and R5 are independently selected from hydrogen, sodium, potassium, ammonium or an organic radical.
- Preferred organic radicals are dFfe n+ i wherein n is between from i to about 5.
- Rl , R2 and R3 are aminophosphonate and aminophosphonic groups which may optionally be substituted with alkyl, phosphonic, aminophosphonic, phosphate and phosphonate groups.
- Examples of preferred phosphonate acids, esters or salts include aminotri (methylene phosphonic acid) and its pentasodium salt, 1 -hydroxy ethyli dene- 1, 1- diphosphonic acid and its tetrasodiura salt, hexamethylenediammeietra (methylene phosphonic acid) and its hexapotassium salt, and diethylenetriaminepenta (methylene phosphonic acid) and its hexasodium salt.
- 1 -hydroxy ethyl idene- 1 ,1 -diphosphonic acid available as BEQUEST 2010 and diethylenediamine penta (methylene phosphonic) acid, commercially as DEQUES ' ! 2060S, both available from Solutia, Inc. in 60% strength.
- both R4 and R5 are more desirably -H versus the stated salt derivatives.
- those phosphonic acid salts which generate the corresponding phosphonic acid in-situ in the presence of a slight amount of strong- acid, such as HQ.
- the amount of phosphonate in the well treatment composition is generally between from about 0.1 to about 10, preferably from about 0.25 to about 6, more preferably from about 0.5 to about 3, percent by volume based on the total volume of water, phosphonate and hydrofluoric acid source.
- the organosiianes for use herein contain a Si-C bond and includes polysiloxanes.
- organosiianes especially suitable for use in this invention are those organosilane halides of the formula:
- X is a halogen selected from the group consisting of chlorine, bromine and iodine with chlorine being preferred
- Rj is an alkyl, alkenyl, alkoxide or aryl group having from 1 to 18 carbon atoms and R 2 and R3 ⁇ 4 are the same or different halogens, or alkyl, alkenyl, alkoxide or aryl group having from 1 to 18 carbon atoms.
- Suitable specific organosilane halides include methyldiethylchlorosilane, dimethyldichforosilane, memyliriehlorosilane, dimethyldibromosilane, diethyldiiodosilane, dipropyldichlorosilane, dipropyidibromosilane, butyltrichlorosilane, phenyltribromosiiane, diphenyldichlorosilane, toly1tribromosi1ane, methylphenyldichlorosifane, propyldimethoxychlorosilane and the like as well as organosilane alkoxides and amino silanes.
- organosilane alkoxides suitable for use in this invention are those having the formula:
- R4, R5, and R5 are independently selected from hydrogen and organic radicals having from 1 to 50 carbon atoms, provided not all of R 4 , R5, and g are hydrogen, and R 7 is an organic radical having from 1 to 50 carbon atoms.
- R4, R 5 , and Re are independently selected, from hydrogen, amine, alkyl, alkenyl. aiyi, and carbhydryloxy groups having from 1 to 18 carbon atoms, with at least one of the R4, R5, and Rg groups not being hydrogen, and R 7 is selected from amine, alkyl, alkenyl, and aryf groups having from 1 to 18 carbon atoms.
- R 4 , R5 , and 3 ⁇ 4 are carbhydiyloxy groups, alkoxy groups are preferred.
- the organosilane may be of the formula:
- R is branched, or linear aliphatic carbon chain that may be saturated or unsaturated (e.g., containing one or more double and/or triple bonds), and which may have from about 1 to about 10 carbon atoms, alternatively from about 1 to about 5 carbon atoms, and further alternatively about 3 carbon atoms; and. wherein each R' is independently branched or linear carbon chain that may be saturated, or unsaturated (e.g., containing one or more double and/or triple bonds), and which may have from about 1 to about 4 carbon atoms, alternatively from about 1 to about 2 carbon atoms, and further alternatively about 2 carbon atoms; it being understood that each R' group may be the same or different structure than one or both of the other R' groups.
- R may be further characterized as alkanyl or alkenyl carbon chain having the above-properties.
- R may be characterized, as an aromatic carbon chain or alicyclic carbon chain.
- one or more of the carbon chains R and/or R' may be optionally and independently derivatized, e.g., the R carbon chain and/or one or more of the R' carbon chains may each contain one or more amino functional groups, one or more halogen groups (e.g., tetrachlorosilane, methyltrichlorosilane, etc.), two or more isocyanate functional groups, two or more epoxy groups, etc.
- halogen groups e.g., tetrachlorosilane, methyltrichlorosilane, etc.
- a silane may include an amrao-funetional silane-based compound such as aminoalkyl siloxanes like gamma- aminopropyltriethoxy silane, a isocyanate-functional silane-based compound such as gamma isocyanatopropyltriethoxy silane and mixtures thereof.
- an amrao-funetional silane-based compound such as aminoalkyl siloxanes like gamma- aminopropyltriethoxy silane
- a isocyanate-functional silane-based compound such as gamma isocyanatopropyltriethoxy silane and mixtures thereof.
- silane-based products available from liquid isobuytlisopropyldimethoxysilane, liquid diisopropylmethoxysilane, liquid diisobutyldimethoxysiiane, liquid dicyclopentyldimethoxysilane, gamma- aminopropyltriethoxy silane epoxy functional silanes.
- the silane may include one or more siioxane-based compounds having the following chemical formula:
- R is a branched, or linear aliphatic carbon chain that may be saturated or unsaturated (e.g.. containing one or more double and/or triple bonds), and which may have from about 1 to about 10 carbon atoms, aiteraatively from about 1 to about 5 carbon atoms, and further aiteraatively about 3 carbon atoms; wherein each R and each R" is independently a branched or linear carbon chain that may be saturated or unsaturated (e.g., containing one or more double and/or triple bonds), and which may have from about 1 to about 4 carbon atoms, alternatively from about 1 to about 2 carbon atoms, and further alternatively about 2 carbon atoms; it being understood that one R' may be the same or different than the other R' group, that one R" may be the same or different than the other R" group, and that one or both R' groups may be the same or different than one or both R" groups.
- R may be farther characterized as alkanyl or alkenyl carbon chain having the above-properties.
- R may be characterized as an aromatic carbon chain or alicyclie carbon chain.
- one or more of the carbon chains R and/or R' may be optionally and independently derivatized, e.g., the R carbon chain and/or one or more of the R and/or R" carbon chains may each contain one or more amino functional groups, two or more isocyanate functional groups, two or more epoxy groups, etc.
- siloxane -based compounds include, but are not limited to, an aqueous solution of aminoalkyl siloxane available from Baker Hughes Incorporated as FSA-1.
- suitable specific organosilane aikoxides and amino siianes include methyltriethoxysilane, dimethyidiethoxysilane, meihyltrimethoxysilane, divinyldimethoxysilane, divmyldi-2-methoxyethoxy silane, di.(3-glycidoxypropyl) dimethoxysilane, vinyltriethoxysilane, vinyitris-2-methoxyethoxysilane, 3- glyeidoxypropyltrimethoxysilane, 3-methacr loxypropyltrimethoxysilane, 2-(3,4- epoxycyclohexyl) ethyUrimethoxysilane, N-2-aminoethyl-3- propy
- the amount of organosilane in the aqueous well treatment composition is between from about 0.01 to about 10 percent, preferably from about 0.1 to about 5 percent, by volume based on the total volume amount of the well treatment composition.
- the amount of the ammonium salt in the aqueous well treatment composition is between from about 0.01 to about 10 percent, preferably from about 0.1 to about 5 percent by volume based on the total volume amount of the well treatment composition.
- ammonium salt is preferably a quaternary ammonium salt represented by the formula:
- R°, R 7 , R 8 and R 9 are independently hydrogen, a C] -C 2 o aikyl group (preferably a Ci-Ce alkyl group), a hydroxyaikyl group wherein the alkyl group is preferably a Ci-Cio alkyl and more preferably a Ci-Q alkyl or a radical of the structure:
- each R 6 , R' and R 8 is a hydroxyaikyl, such as 2 -hydroxy ethyl, and R y is an alkyl group, such as methyl.
- each of R 6 , R' and R 8 are alkyl and R 9 is a hydroxyalkyl, such as 2-hydroxyethyl.
- each of R 6 , R ', R 8 and R 9 are an alky] group.
- the anion, Y " is a salt, preferably a halide, X, perc orate, thiocyanate, eyanate, a Ci- , carboxylate, an alkyl sulfate, methanesulfonate, BX 4 " , PF 6 ⁇ , AsFY, SbFY, NO? " , N0 3 " or SO 4 " .
- X is a halide, such as chloride.
- Such salts are set forth in U.S. Patent No. 5,342,530, herein incorporated by reference.
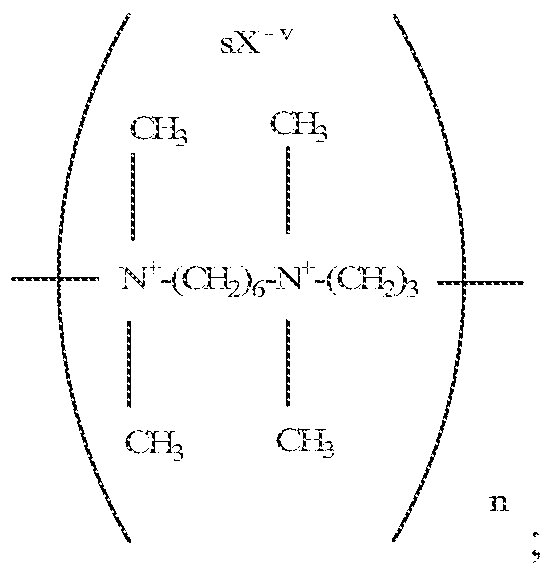
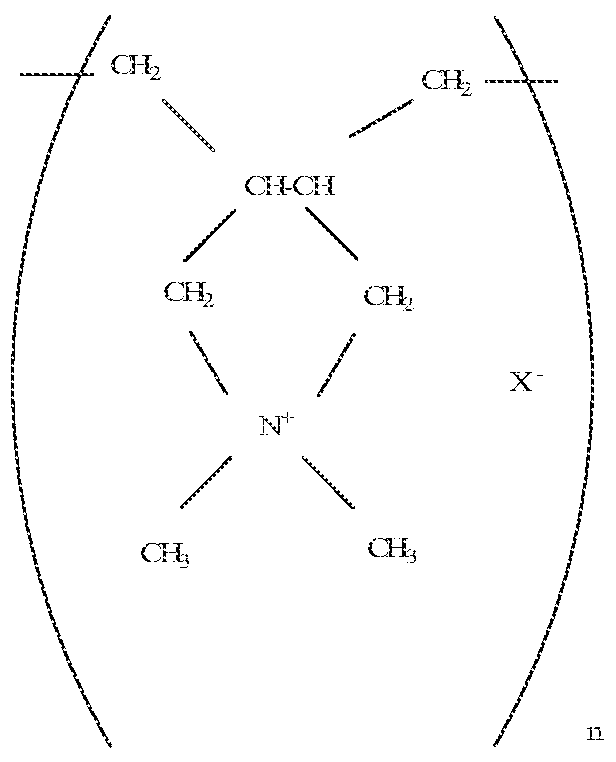
- acceptable salts include polycationic polymers having a molecular weight up to about 300,000, for instance from 50,000 to 300,000 and having the repeating units represented by the formulae:
- R and R J are independently selected from the group consisting of an alkylene group having from about 2 to about 4 carbon atoms;
- R 9 are independently selected from the group consisting of methyl and ethyl;
- R° is selected from the group consisting of an unsubstituted alkylene group having from about 2 to 4 carbon atoms and a substituted alkylene group having from about 2 to about 4 carbon atoms and containing a hydroxy group;
- X is an anion selected from the group consisting of a halogen, methyl sulfate, sulfate, and nitrate;
- v represents the valency of the anion represented by X; and, s is an integer equal to the number of said anions required to maintain electronic neutrality.
- R and R 3 are preferably independently selected from the group consisting of ethylene, trirnethylene, tetramethyiene, and 2-methyltrirnethylene;
- R° is preferably selected from the group consisting of 2-hydroxytriniethylene, 2-hydroxy tetramethyiene, and 3- hydroxy tetraniethlene;
- R 1 and R are preferably methyl;
- R 4 and R J are preferably ethyl;
- R 7 , R 8 and R 9 are preferably methyl;
- X is preferably selected from the group consisting of a halogen such as chloride, bromide, and iodide, methyl sulfate and sulfate, most preferably halogen.
- Preferred are those compounds of the formula;
- the ammonium salt is a quaternary ammonium salt like choline chloride and preferably contains the ⁇ , , -trimethyiethanolammonium cation.
- quaternary ammonium salts include Claytreai-3C clay stabilizer (CT- 3C) or Claymaster-5C both by Baker Hughes Inc.
- the ⁇ of the well treatment composition is typically maintained at a range of 0 to about 3.0. Enough acid should be used to maintain the pH of the aqueous HF solution and to hydrolyze ammonium fluoride or biffuoride, if it is used. Maintenance of the desired pH range aids in the inhibition of inorganic scales and in most instances the prevention of formation of such scales. In addition, maintenance of the pH range maximizes the effect of the organosilane and the quaternary ammonium salt on the inhibition and control of fines and swellable clay.
- the composition may include or have added thereto corrosion inhibitors, surfactants, iron control agents, non-emuisifiers, foaming agents, water-wetting surfactants, anti- sludge agents, mutual solvents or alcohols (such as methanol or isopropanol ⁇ , gelling agents, bactericides, or fluid loss control agents.
- corrosion inhibitors such as methanol or isopropanol ⁇
- non-emuisifiers such as methanol or isopropanol ⁇
- gelling agents such as bactericides, or fluid loss control agents.
- mutual solvents or alcohols such as methanol or isopropanol ⁇
- gelling agents such as bactericides, or fluid loss control agents.
- the well treatment composition is introduced into the formation at the location where treatment is desired.
- the well treatment composition may be applied after treatment of the formation with a pre-flush.
- the well treatment composition of the invention enhances the production of hydrocarbons from hydrocarbon bearing calcareous or siliceous formations.
- the treatment method is especially effective if applied prior to gravel packing or fracturing.
- the well treatment composition may easily be applied in the stimulation of sandstone formations containing calcareous materials and calcareous formations such as carbonate or dolomite. In addition to its use in matrix acidizing, it may be used, in acid fracturing as well as pre- fracturing treatment on sandstone, carbonate and dolomite formations. They ma also be used for remedial workovers of wells to keep silicates in suspension and to remove clay, fine and sand deposits as well as inorganic scales from downhole screens and. from drilling fluid damage.
- the well treatment composition is capable of dissolving carbonates, as well as siliceous minerals, while minimizing the formation of calcium fluoride and magnesium fluoride or sodium or potassium fluorosilicate or fluoroalummate.
- Such well treatments may be simplified by use of the well treatment composition defined herein since the need to pump multiple fluids in a carefully choreographed sequence is eliminated. Further, acid placement and distribution is improved and equipment requirements are reduced, e.g., in terms of tankage, etc. Use of the well treatment composition improves logistics, reduces costs, along with improved results, while simultaneously rendering treatments which are easier to implement and control at the field level.
- the well treatment composition may further be employed in the remediation of oil and gas and geothermal wells by preventing and/or inhibiting the formation of unwanted deposits on the surfaces of the wellbore, downhole assembly, sand control screens, production equipment and pipelines.
- unwanted deposits form and/or accumulate in the wellbore, production equipment, recovery equipment and well casing.
- Remediation treatment fluids are further typically used to remove such undesired deposits prior to the introduction of stimulation fluids or to restore well productivity from the undesired deposits.
- the invention is used to remove siliceous or calcareous deposits inside well tubulars.
- the well treatment composition may also be used to treat pipelines from undesired deposits.
- the well treatment composition is preferably injected directly into the wellbore through the production tubing or through the use of coiled tubing or similar delivery mechanisms.
- the composition remedies damage caused during well treating such as, for instance, by stimulation fluids and drilling fluid muds, by dispersing and removing siliceous materials from the formation and wellbore.
- compositions of Examples 1-6 were illustrated on a formation containing calcareous minerals as follows.
- a composition consisting of 75 wt. % quartz, 5 wt. % kaoiinite, 10 wt. % potassium-feldspar and 10 wt. % calcium carbonate (powder) was prepared.
- the composition was tested for its solubility in a HF acid at 150°F over 4 and 24 hrs. After solubility testing, the un-dissolved solid or precipitate was analyzed.
- Tables 2-5 represents the 4 hour solubility testing of the formation composition at 150° F.
- Tables 3-5 represent the 4 and 24 hour solubility testing of the formation composition at 150° F,
- Tables 2-5 demonstrate that the well treatment compositions defined herein can control or minimize the formation of inorganic fluoride scales, such as calcium fluoride, in the hydrofluoric acid.
- Table 7 shows the composition of acids systems used in the eoreflood. experiment wherein HV represents an organophosphonate.
- Test A The pressure drop across the core is a function of cumulative injected pore volumes during the five fluid stage injections. Initially a FLsCi solution was injected at a rate of 5 ml/min for 1 PV then reduced to 2 ml/ ' min to calculate an accurate average value of initial permeability. Ail acid stages were injected at a rate of 2 ml/min and did not contain the combination of Claymaster-5C and FSA-1. An increase in the pressure drop across the core during the injection of the pre-flushed stage (which mainly 10 wt% HC1) was observed. Also, during the injection of main acid the pressure drop across the core initially decreased then increased again as evidence of damage occurring during the main acid injection. At the end of the experiment H 4 CI solution was injected at a rate of 2 ml/min and then increased to 5 ml/min to calculate an accurate average value of final permeability. A -10.9 % permeability enhancement was observed
- Test B liiitially a NFLjCl solution was injected at a rate of 5 ml/min then reduced to 2 ml/min to calculate an accurate average value of initial permeability.
- Ail acid stages were injected at a rate of 2 ml/min and contained the combination of 3 gpt of Ciaymasier ⁇ 5C and 5 gpt of FSA-1, An increase in the pressure drop across the core during the injection of the pre-flushed stage (mainly 10 wt% HC1) was observed. However, no increase in pressure drop was observed across the core during the injection of main acid. This illustrates that the combination of Claymaster-5C and FSA- 1 enhanced the permeability of the core during main acid injection. Finally, a NH 4 CI solution was injected at a rate of 2 ml/min and then increased to 5 ml/min to calculate an accurate average value of final permeability. A 42% permeability enhancement was observed.
- Example 9 A coreflood study was conducted using a Bandera Sandstone core at 180°F. wherein the Bandera Sandstone had the composition illustrated in Table 6 above. Table 9 sets forth the composition of the acid system:
- the main acid used had less amount of phosphonate acid, no boric acid and no acetic acid, as shown in Table 10.
- the pressure drop across the core was a function of cumulative injected pore volume during the five fluid stage injections. Initially a NH 4 CI solution was injected at a rate of 5 ml/min then reduced to 2 ml/min to calculate an accurate average value of initial permeability. All acid stages, set forth in Table 9, were injected at a rate of 2 ml/min and contained the combination of 3 gpt of Claymaster-5C and 5 gpt of FSA-1. An increase in the pressure drop across the core during the injection of the pre-flush stage, principally 15 wt% HC1, was observed.
- the main acid used contained the combination of Claymaster-5C (3 gpt) and FSA-1 (5 gpt). A 86% increase in core permeability was observed as illustrated in Table 12.
- the acid system is set forth in Table 1 1. Three fluid stages were injected in the following sequence:
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Inorganic Chemistry (AREA)
- Production Of Liquid Hydrocarbon Mixture For Refining Petroleum (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Detergent Compositions (AREA)
- Silicon Compounds (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Sealing Material Composition (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2877138A CA2877138C (en) | 2012-06-26 | 2013-06-26 | Method of removing inorganic scales |
| PL13810249T PL2864585T3 (en) | 2012-06-26 | 2013-06-26 | Method of removing inorganic scales |
| EP13810249.6A EP2864585B1 (en) | 2012-06-26 | 2013-06-26 | Method of removing inorganic scales |
| IN10957DEN2014 IN2014DN10957A (en) | 2012-06-26 | 2013-06-26 | |
| BR112014032580-4A BR112014032580B1 (en) | 2012-06-26 | 2013-06-26 | METHODS TO INCREASE THE PRODUCTIVITY OF A SILICIOUS OR LIME-BASED HYDROCARBIDE FORMATION, TO PREVENT AND/OR INHIBIT CLAY BLOWING WITHIN AN UNDERGROUND FORMATION PENETRATED BY A WELL HOLE, AND FOR ORGANIC SCALES REMOVAL |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/533,031 US20120325485A1 (en) | 2007-09-18 | 2012-06-26 | Method of removing inorganic scales |
| US13/533,031 | 2012-06-26 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2014004697A2 true WO2014004697A2 (en) | 2014-01-03 |
| WO2014004697A3 WO2014004697A3 (en) | 2015-04-02 |
Family
ID=49784010
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2013/047938 WO2014004697A2 (en) | 2012-06-26 | 2013-06-26 | Method of removing inorganic scales |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP2864585B1 (en) |
| AR (1) | AR091560A1 (en) |
| BR (1) | BR112014032580B1 (en) |
| CA (1) | CA2877138C (en) |
| HU (1) | HUE036346T2 (en) |
| IN (1) | IN2014DN10957A (en) |
| PL (1) | PL2864585T3 (en) |
| WO (1) | WO2014004697A2 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9404067B2 (en) | 2014-08-26 | 2016-08-02 | Ecolab Usa Inc. | Fluoro-inorganics for inhibiting or removing silica or metal silicate deposits |
| US9932255B2 (en) | 2015-06-30 | 2018-04-03 | Ecolab Usa Inc. | Metal silicate and organic deposit inhibitor/dispersant for thermal recovery operations of hydrocarbon fuels |
| US10035949B2 (en) | 2015-08-18 | 2018-07-31 | Ecolab Usa Inc. | Fluoro-inorganics for well cleaning and rejuvenation |
| WO2020257076A1 (en) * | 2019-06-21 | 2020-12-24 | Baker Hughes, A Ge Company, Llc | Aqueous delayed acid system for well stimulation |
| WO2021102545A1 (en) * | 2019-11-29 | 2021-06-03 | Petróleo Brasileiro S.A. - Petrobras | Combined treatment process for removing and inhibiting scale |
| US11585196B2 (en) | 2019-10-23 | 2023-02-21 | Baker Hughes Oilfield Operations Llc | Methods of using energized well treating fluids |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4447342A (en) | 1982-04-19 | 1984-05-08 | Halliburton Co. | Method of clay stabilization in enhanced oil recovery |
| US4536305A (en) | 1984-09-21 | 1985-08-20 | Halliburton Company | Methods for stabilizing swelling clays or migrating fines in subterranean formations |
| US5342530A (en) | 1991-02-25 | 1994-08-30 | Nalco Chemical Company | Clay stabilizer |
| US5529125A (en) | 1994-12-30 | 1996-06-25 | B. J. Services Company | Acid treatment method for siliceous formations |
| US6443230B1 (en) | 1999-06-22 | 2002-09-03 | Bj Services Company | Organic hydrofluoric acid spearhead system |
| US20090075844A1 (en) | 2007-09-18 | 2009-03-19 | Bj Services Company | Method and composition for controlling inorganic fluoride scales |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IT1207517B (en) * | 1985-12-19 | 1989-05-25 | Enichem Sintesi | STAINLESS STEEL CORROSION INHIBITORS. |
| ES2725499T3 (en) * | 2007-04-20 | 2019-09-24 | Evonik Degussa Gmbh | Mixture containing an organosilicon compound and its use |
-
2013
- 2013-06-25 AR ARP130102247 patent/AR091560A1/en active IP Right Grant
- 2013-06-26 WO PCT/US2013/047938 patent/WO2014004697A2/en active Application Filing
- 2013-06-26 EP EP13810249.6A patent/EP2864585B1/en active Active
- 2013-06-26 CA CA2877138A patent/CA2877138C/en active Active
- 2013-06-26 IN IN10957DEN2014 patent/IN2014DN10957A/en unknown
- 2013-06-26 BR BR112014032580-4A patent/BR112014032580B1/en active IP Right Grant
- 2013-06-26 HU HUE13810249A patent/HUE036346T2/en unknown
- 2013-06-26 PL PL13810249T patent/PL2864585T3/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4447342A (en) | 1982-04-19 | 1984-05-08 | Halliburton Co. | Method of clay stabilization in enhanced oil recovery |
| US4536305A (en) | 1984-09-21 | 1985-08-20 | Halliburton Company | Methods for stabilizing swelling clays or migrating fines in subterranean formations |
| US5342530A (en) | 1991-02-25 | 1994-08-30 | Nalco Chemical Company | Clay stabilizer |
| US5529125A (en) | 1994-12-30 | 1996-06-25 | B. J. Services Company | Acid treatment method for siliceous formations |
| US6443230B1 (en) | 1999-06-22 | 2002-09-03 | Bj Services Company | Organic hydrofluoric acid spearhead system |
| US20090075844A1 (en) | 2007-09-18 | 2009-03-19 | Bj Services Company | Method and composition for controlling inorganic fluoride scales |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2864585A4 |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9404067B2 (en) | 2014-08-26 | 2016-08-02 | Ecolab Usa Inc. | Fluoro-inorganics for inhibiting or removing silica or metal silicate deposits |
| US9932255B2 (en) | 2015-06-30 | 2018-04-03 | Ecolab Usa Inc. | Metal silicate and organic deposit inhibitor/dispersant for thermal recovery operations of hydrocarbon fuels |
| US10035949B2 (en) | 2015-08-18 | 2018-07-31 | Ecolab Usa Inc. | Fluoro-inorganics for well cleaning and rejuvenation |
| WO2020257076A1 (en) * | 2019-06-21 | 2020-12-24 | Baker Hughes, A Ge Company, Llc | Aqueous delayed acid system for well stimulation |
| US11390799B2 (en) | 2019-06-21 | 2022-07-19 | Baker Hughes Holdings Llc | Aqueous delayed acid system for well stimulation |
| US11585196B2 (en) | 2019-10-23 | 2023-02-21 | Baker Hughes Oilfield Operations Llc | Methods of using energized well treating fluids |
| WO2021102545A1 (en) * | 2019-11-29 | 2021-06-03 | Petróleo Brasileiro S.A. - Petrobras | Combined treatment process for removing and inhibiting scale |
| CN115038773A (en) * | 2019-11-29 | 2022-09-09 | 巴西石油公司 | Combined treatment process for removing and inhibiting scale |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2864585A4 (en) | 2016-04-20 |
| EP2864585B1 (en) | 2017-10-18 |
| CA2877138C (en) | 2017-06-20 |
| BR112014032580A8 (en) | 2021-04-06 |
| AR091560A1 (en) | 2015-02-11 |
| IN2014DN10957A (en) | 2015-09-18 |
| HUE036346T2 (en) | 2018-07-30 |
| BR112014032580A2 (en) | 2017-06-27 |
| PL2864585T3 (en) | 2018-06-29 |
| BR112014032580B1 (en) | 2021-06-01 |
| CA2877138A1 (en) | 2014-01-03 |
| WO2014004697A3 (en) | 2015-04-02 |
| EP2864585A2 (en) | 2015-04-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8211836B2 (en) | Method of removing calcareous or siliceous deposits or scales | |
| US20120325485A1 (en) | Method of removing inorganic scales | |
| US4646835A (en) | Acidizing method | |
| EP0800613B1 (en) | Acid treatment method for siliceous formations | |
| US7431089B1 (en) | Methods and compositions for selectively dissolving sandstone formations | |
| EP2864585B1 (en) | Method of removing inorganic scales | |
| US8802601B2 (en) | Method of treating sandstone formations with reduced precipitation of silica | |
| US6806236B2 (en) | Composition and method for treating a subterranean formation | |
| US9334716B2 (en) | Treatment fluids comprising a hydroxypyridinecarboxylic acid and methods for use thereof | |
| US6924255B2 (en) | Composition and method for treating a subterranean formation | |
| US9738823B2 (en) | Treatment fluids comprising a stabilizing compound having quaternized amine groups and methods for use thereof | |
| CN110785471A (en) | Compositions and methods for controlling strong acid systems | |
| US9725642B2 (en) | Complexation of calcium ions in the presence of quaternized amine compounds while acidizing a subterranean formation | |
| US11198812B2 (en) | Use of sequestering agent in GLDA-based treatments for siliceous formations | |
| WO2000070186A1 (en) | Method for acidizing a subterranean formation | |
| US5039434A (en) | Acidizing composition comprising organosilicon compound | |
| EP2825614B1 (en) | Treatment fluids comprising a hydroxypyridinecarboxylic acid and methods for use thereof | |
| EP0265563B1 (en) | Acidizing method | |
| US20190309218A1 (en) | Inhibition of precipitation during sandstone acidizing | |
| GB2177145A (en) | Acidizing method | |
| NO174821B (en) | Method for acid treatment of an underground formation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13810249 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2877138 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2013810249 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013810249 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112014032580 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112014032580 Country of ref document: BR Kind code of ref document: A2 Effective date: 20141224 |