WO2013166121A1 - Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides - Google Patents
Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides Download PDFInfo
- Publication number
- WO2013166121A1 WO2013166121A1 PCT/US2013/039025 US2013039025W WO2013166121A1 WO 2013166121 A1 WO2013166121 A1 WO 2013166121A1 US 2013039025 W US2013039025 W US 2013039025W WO 2013166121 A1 WO2013166121 A1 WO 2013166121A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sirna
- different
- tetragalnac
- ligands
- attached
- Prior art date
Links
- XXJDLFDYEZLYGG-UHFFFAOYSA-N [N-]=[N+]=NCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCNC(CCSSc1ccccn1)=O Chemical compound [N-]=[N+]=NCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCNC(CCSSc1ccccn1)=O XXJDLFDYEZLYGG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/549—Sugars, nucleosides, nucleotides or nucleic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H15/00—Compounds containing hydrocarbon or substituted hydrocarbon radicals directly attached to hetero atoms of saccharide radicals
- C07H15/26—Acyclic or carbocyclic radicals, substituted by hetero rings
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering N.A.
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
Definitions
- lipid nanoparticle (LNP) encapsulation typically employs a targeting ligand or a lipid or a solubilizing group or an endosomolytic peptide or a cell penetrating peptide and/or a combination of two or all four attached to an oligonucleotide.
- Linkers may be present in the conjugate as well as other functionalities.
- Single chemical conjugates comprising tetraGalNAc ligands disclosed herein have surprising properties of improved delivery efficiency, cell uptake and/or endosomal escape.
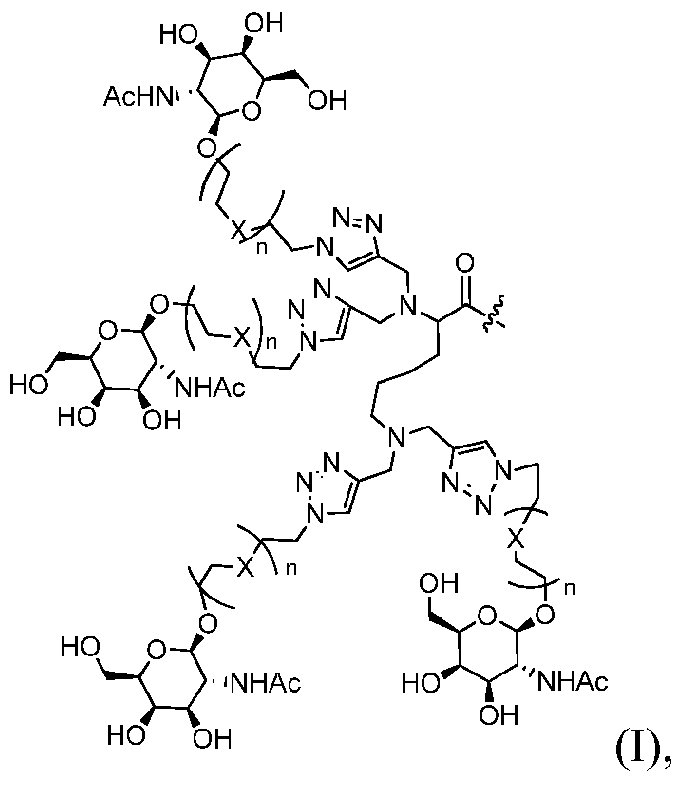
- a modular composition disclosed herein comprises: 1) a single stranded or double stranded oligonucleotide; 2) one or more tetraGalNAc ligands of Formula (I), which may be the same or different:
- X is -0-, -S-, -CR R 2 - or -NR 1 -, wherein R 1 and R 2 are each independently selected from the group consisting of hydrogen and Cl-C6alkyl; n is 1 , 2, 3, or 4; and the bond with " ⁇ vw- " indicates point of attachment; optionally, 3) one or more linkers, which may be the same or different; and optionally, 4) one or more targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- R 1 and R 2 are each independently selected from the group consisting of hydrogen, methyl and ethyl. In another embodiment, R 1 and R 2 are each hydrogen.
- the tetraGalNAc ligand has Formula (II) wherein X, R 1 , R 2 and n are as defined above. In another embodiment, the tetraGalNAc ligand has Formula (III) wherein X, R 1 , R 2 and n are as defined above:
- a modular composition comprises: 1) a single stranded or double stranded oligonucleotide; 2) 1-8 tetraGalNAc ligands of Formula (I), (II) or (III), which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; and n is 1 , 2, 3, or 4; 3) 1-24 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- Formula (I), (II) or (III) which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; and n is 1 , 2, 3, or 4; 3) 1-24 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents,
- a modular composition comprises: 1) a single stranded or double stranded siRNA; 2) 1-8 tetraGalNAc ligands of Formula (I), (II) or (III), which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; and n is 1, 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- Formula (I), (II) or (III) which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; and n is 1, 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipid
- the linkers are attached to the oligonucleotide or siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the oligonucleotide or siRNA.
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA optionally via linkers.
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the oligonucleotide or siRNA; and the tetraGalNAc ligands are attached to the oligonucleotide or siRNA optionally via linkers.
- X of Formula (I), (II) or (III), is - 0-, -S-, or -CH 2 -; and n is 1, 2 or 3.
- X of Formula (I), (II) or (III), is - O- or -CH 2 - and n is 1 or 2.
- X of Formula (I), (II) or (III), is - O- and n is 1 or 2.
- X of Formula (I), (II) or (III), is - CH 2 - and n is 1 or 2.
- the composition comprises 1-6 tetraGalNAc ligands, or more specifically, 1-4 tetraGalNAc ligands, which may be the same or different.
- the oligonucleotide or siRNA is double stranded; and the tetraGalNAc ligands are attached to the guide strand or the passenger strand of the oligonucleotide or siRNA at different 2'-positions of the ribose rings.
- the oligonucleotide or siRNA is double stranded; and the tetraGalNAc ligands are attached to the guide strand or the passenger strand of the oligonucleotide or siRNA at different terminal 3' and/or 5'-positions.
- the oligonucleotide or siRNA is double stranded; and two or more tetraGalNAc ligands are attached to both the guide strand and the passenger strand of the oligonucleotide or siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions.
- each linker is independently selected from Table 1.
- each linker is independently selected from Table 2.
- the oligonucleotide or siRNA is double stranded; and the optional targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents are attached to the same or different strands of the oligonucleotide or siRNA.
- a modular composition comprises 1) a double stranded siRNA; 2) 1-8 tetraGalNAc ligands of Formula (IV), (V) or (VI):
- 1-16 linkers independently selected from Table 1, which may be the same or different; and, optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- a modular composition comprises 1) a double stranded siRNA; 2) 1-4 tetraGalNAc ligands of Formula (IV), (V) or (VI); 3) 1-8 linkers independently selected from Table 1 , which may be the same or different; and, optionally, 4) 1-4 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents; wherein the tetraGalNAc ligands are attached to the siRNA at different 2'- positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the siRNA; and wherein the tetraGalNAc ligands are attached to the siRNA optionally via linkers.
- the tetraGalNAc ligands are attached to the same strand of the siRNA via linkers.
- a modular composition comprises 1) a double stranded siRNA; 2) 1-4 tetraGalNAc ligands of Formula (V); 3) 1-8 linkers independently selected from Table 2, which may be the same or different; and, optionally, 4) 1-4 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents; wherein the tetraGalNAc ligands are attached to the siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the siRNA; and wherein the tetraGalNAc ligands are attached to the siRNA via linkers.
- the tetraGalNAc ligands are attached to the same strand of the siRNA via linkers.
- single chemical conjugates comprising a single stranded or double stranded oligonucleotide; and one or more tetraGalNAc ligands of Formula (I), which may be the same or different:
- X is -0-, -S-, -CR R 2 - or -NR 1 -, wherein R 1 and R 2 are each independently selected from the group consisting of hydrogen and Cl-C6alkyl; n is 1, 2, 3, or 4; and the bond with " indicates the point of attachment.
- R 1 and R 2 are each independently selected from the group consisting of hydrogen, methyl and ethyl. In another embodiment, R 1 and R 2 are each hydrogen.
- the tetraGalNAc ligand has Formula (II) wherein X, R 1 , R 2 and n are as defined above. In another embodiment, the tetraGalNAc ligand has Formula (III) wherein X, R 1 , R 2 and n are as defined above:
- the oligonucleotide is a short interfering RNA (siRNA).
- siRNA is a single stranded siRNA.
- siRNA is a double stranded siRNA.
- the use of the tetraGalNAc disclosed herein may provide effective delivery of the oligonuleotide or siRNA by directing the modular composition to a particular cell.
- the targeting ligand may specifically or non-specifically bind with a molecule on the surface of a target cell and facilitate uptake of the ligand-siRNA conjugate.
- a linker may be present between each tetraGalNAc and the oligonucleotide.
- the linkers are attached to the oligonucleotide at different 2'-positions of the ribose rings and/or the terminal 3' and/or 5'-positions of the oligonucleotide.
- a modular composition comprises 1) a single stranded or double stranded oligonucleotide; 2) one or more tetraGalNAc ligands of Formula (I), which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; n is 1, 2, 3, or 4; and the bond with indicates the point of attachment; optionally, 3) one or more linkers, which may be the same or different; and optionally, 4) one or more targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- Formula (I) which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; n is 1, 2, 3, or 4; and the bond with indicates the point of attachment; optionally, 3) one or more linkers, which may be the same or different; and optionally, 4) one or more targeting ligands
- a modular composition comprises 1) a single stranded or double stranded oligonucleotide; 2) 1-8 tetraGalNAc ligands of Formula (I), (II) or (III), which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; n is 1, 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- Formula (I), (II) or (III) which may be the same or different, wherein X is -0-, -S-, -CH 2 - or -NH-; n is 1, 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents
- a modular composition comprises 1) a single stranded or double stranded siRNA; 2) 1-8 tetraGalNAc ligands of Formula (I), (II) or (III), which may be the same or different, wherein X is -0-, -S-, -CH2- or -NH-; n is 1, 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents.
- Formula (I), (II) or (III) which may be the same or different, wherein X is -0-, -S-, -CH2- or -NH-; n is 1, 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the oligonucleotide or siRNA.
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA optionally via linkers.
- the linkers are present.
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the oligonucleotide or siRNA; and the tetraGalNAc ligands are attached to the oligonucleotide or siRNA via linkers.
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA via linkers and the linkers are attached to the oligonucleotide or siRNA at different 2'-positions of the ribose rings.
- the tetraGalNAc ligands are attached to the oligonucleotide or siRNA via linkers and the linkers are attached to the oligonucleotide or siRNA at different terminal 3' and/or 5'-positions of the oligonucleotide.
- X is -0-, -S-, or -CH 2 -. In another embodiment, X is -O- or -CH 2 -. In another embodiment, n is 1, 2 or 3. In another embodiment, X is -O- and n is 1 or 2. In another embodiment, X is -CH 2 - and n is 1 or 2. In another embodiment, X is -O- and n is 1. In yet another embodiment, X is -CH 2 - and n is 1.
- the oligonucleotide or siRNA is single stranded. In another embodiment, the oligonucleotide or siRNA is double stranded.
- the composition comprises 1-6 tetraGalNAc ligands. In another embodiment, the composition comprises 1-4 tetraGalNAc ligands. In another embodiment, the composition comprises 1-2 tetraGalNAc ligands. In yet another embodiment, the composition comprises 1 tetraGalNAc ligand.
- the oligonucleotide or siRNA is double stranded and the tetraGalNAc ligands are attached to the guide strand at different 2'- positions of the ribose rings.
- the oligonucleotide or siRNA is double stranded the tetraGalNAc ligands are attached to the guide strand at different terminal 3' and/or 5'-positions.
- the oligonucleotide or siRNA is double stranded and the tetraGalNAc ligands are attached to the passenger strand at different 2'-positions of the ribose rings.
- the oligonucleotide or siRNA is double stranded and the tetraGalNAc ligands are attached to the passenger strand at different terminal 3' and/or 5'-positions.
- the oligonucleotide or siRNA is double stranded and the tetraGalNAc ligands are attached to both the guide strand and the passenger strand at different 2'-positions of the ribose rings and/or different terminal 3' and/or 5'-positions.
- the oligonucleotide or siRNA is double stranded and the tetraGalNAc ligands are attached to the same strand.
- the oligonucleotide or siRNA is double stranded and the tetraGalNAc ligands are attached to different strands.
- the oligonucleotide or siRNA is double stranded and the optional targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents are attached to the same or different strands.
- the oligonucleotide or siRNA is double stranded and the optional targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents are attached to the same or different strands via linkers.
- each linker is independently selected Table 1. In another embodiment, each linker is independently selected Table 2.
- a modular composition comprises 1) a single stranded or double stranded siRNA; 2) 1-8 tetraGalNAc ligands of Formula (I), (II) or (III), which may be the same or different; wherein X is -0-, -S-, -CH 2 - or -NH-; and n is 1 , 2, 3, or 4; 3) 1-16 linkers, which may be the same or different; and optionally, 4) 1-8 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents; wherein the tetraGalNAc ligands are attached to the siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the siRNA; and wherein the tetraGalNAc ligands are attached to the siRNA optionally via linkers.
- the linkers are present.
- X is -0-, -S-, or -CH 2 -, and n is 1, 2 or 3.
- X is -O- or -CH 2 -, and n is 1 or 2.
- a modular composition comprises 1) a double stranded siRNA; 2) 1 -6 tetraGalNAc ligands of Formula (I), which may be the same or different; wherein X is -0-, -S-, or -CH 2 -; and n is 1, 2 or 3; 3) 1-18 linkers, which may be the same or different; and optionally, 4) 1-6 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents; wherein the tetraGalNAc ligands are attached to the siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the siRNA; and wherein the tetraGalNAc ligands are attached to the siRNA optionally via linkers.
- the linkers are present.
- X is -0-, -S-, or -CH 2 - and n is 1 or 2.
- the linkers are independently selected from Table 1.
- the linkers are independently selected from Table 2.
- a modular composition comprises 1) a double stranded siRNA; 2) 1 -4 tetraGalNAc ligands of Formula (I), which may be the same or different; wherein X is -0-, -S-, or -CH 2 -; and n is 1 or 2; 3) 1-8 linkers, which may be the same or different; and optionally, 4) 1-4 targeting ligands, solubilizing agents,
- tetraGalNAc ligands are attached to the siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or impositions of the siRNA; and wherein the tetraGalNAc ligands are attached to the siRNA via linkers.
- X is -O- or -CH 2 - and n is 1 or 2.
- the linkers are independently selected from Table 1.
- the linkers are independently selected from Table 2.
- a modular composition comprises 1) a double stranded siRNA; 2) 1-4 tetraGalNAc ligands of Formula (IV), (V) or (VI):
- a modular composition comprises 1) a double stranded siRNA; 2) 1-4 tetraGalNAc ligands of Formula (V); 3) 1-8 linkers independently selected from Table 2, which may be the same or different; and optionally, 4) 1-4 targeting ligands, solubilizing agents, pharmacokinetics enhancing agents, lipids, and/or masking agents; wherein the tetraGalNAc ligands are attached to the siRNA at different 2'-positions of the ribose rings and/or at different terminal 3' and/or 5'-positions of the siRNA; and wherein the tetraGalNAc ligands are attached to the siRNA via linkers.
- the tetraGalNAc ligands are attached to the siRNA via linkers; and wherein the tetraGalNAc ligands are attached to the same strand.
- the tetraGalNAc ligands are attached to the siRNA via linkers; and wherein the tetraGalNAc ligands are attached to different strands.
- the invention features a modular composition, comprising an oligonucleotide ([Oi][0 2 ][0 3 ] [O n ]), one or more linkers (L), one or more tetraGalNAc ligands (G), and one or more optional lipid(s) (X), targeting ligand(s) (X), and/or solubilizing group(s) (X).
- the modular composition may have the formula:
- the modular composition may have the formula:
- Non-limiting examples of modular compositions comprising double stranded oligonucleotides with terminal conjugations are:
- Non-limiting examples of modular compositions comprising double stranded oligonucleotides with internal conjugations are:
- linkers Any number of linkers, and therefore any number of tetraGalNAc ligands, can be attached to the oligonucleotide.
- a preferred range of numbers of linkers is from 1-16.
- a more preferred range of numbers of linkers is from 1-8, or more specfically, 1-4.
- a preferred range of numbers of tetraGalNAc ligands is from 1-8.
- a more preferred range of numbers of peptides is from 1-8, or more specifically, 1-4.
- the two strands contain n and n' nucleotides respectively.
- the numbers n and n' can be equal or different.
- the numbers are integers ranging from 8 to 50.
- the numbers are integers ranging from 12-28. More preferably, the numbers are integers ranging from 19-21.
- each nucleotide [O n ] or [ ⁇ ⁇ ' ], that contains a linker (L-G and/or L-X) has generic structures shown in the following cartoon:
- D oxygen (O).
- the two nucleotides [O n _i] and [O n ] or [O n ⁇ _i] and [ ⁇ ⁇ ' ] are connected via phosphodiester or thio-phosphodiester bonds.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group may be located on the same strand or on different strands.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group are on the same strand.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group are on the passenger strand.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group are on the guide strand.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group are located on different strands.
- the "G-L" is on the passenger strand while the lipid, targeting ligand, and/or solubilizing group is on the guide strand.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group are on different strands but on the same terminal end of the double- stranded oligonucleotide.
- the "G-L" and the lipid, targeting ligand, and/or solubilizing group are on different strands and on the opposite terminal ends of the double- stranded oligonucleotide.
- an additional "G-L" of identical or different nature can be used in place of the lipid, targeting ligand, and/or solubilizing group noted in the above embodiments.
- the "G-L” can be located on multiple terminal ends of either the passenger or guide strand and the the lipid, targeting ligand, and/or solubilizing group can be located on the remaining terminal ends of the passenger and guide strands.
- one "G-L” and two or more lipids, targeting ligands, and/or solubilizing groups are present in the oligonucleotide.
- two or more "G-L" and two or more lipids, targeting ligands and/or solubilizing groups are present in the oligonucleotide.
- oligonucleotide when the oligonucleotide is a double-stranded oligonucleotide and multiple "G-L" components and/or lipids, targeting ligands, and/or solubilizing groups are present, such multiple "G-L” components and/or lipids, targeting ligands, and/or solubilizing groups may all be present in one strand or both strands of the double stranded oligonucleotide.
- G-L When multiple "G-L” components and/or lipids, targeting ligands, and/or solubilizing groups are present, they may all be the same or different.
- the "G-L" are on internal nucleotides only (i.e.
- the invention includes a method of delivering an oligonucleotide or siRNA to a cell.
- the method includes (a) providing or obtaining a modular composition disclosed herein; (b) contacting a cell with the modular composition; and (c) allowing the cell to internalize the modular composition.
- the method can be performed in vitro, ex vivo or in vivo, e.g., to treat a subject identified as being in need of an oligonucleotide or siRNA.
- a subject in need of said oligonucleotide is a subject, e.g., a human, in need of having the expression of a gene or genes, e.g., a gene related to a disorder, downregulated or silenced.
- the invention provides a method for inhibiting the expression of one or more genes.
- the method comprising contacting one or more cells with an effective amount of an oligonucleotide of the invention, wherein the effective amount is an amount that suppresses the expression of the one or more genes.
- the method can be performed in vitro, ex vivo or in vivo.
- the methods and compositions of the invention can be used with any oligonucleotides or siRNAs known in the art.
- the methods and compositions of the invention can be used for the treatment of any disease or disorder known in the art, and for the treatment of any subject, e.g., any animal, any mammal, such as any human.
- any subject e.g., any animal, any mammal, such as any human.
- the methods and compositions of the invention may be used for the treatment of any disease that would benefit from downregulating or silencing a gene or genes.
- compositions of the invention may be used with any dosage and/or formulation described herein, or any dosage or formulation known in the art.
- routes of administration described herein a person skilled in the art will also appreciate that other routes of administration may be used to administer the modular composition of the invention.
- oligonucleotide is a double stranded or single stranded, unmodified or modified RNA or DNA
- modified RNAs include those which have greater resistance to nuclease degradation than do unmodified RNAs. Further examples include those which have a 2' sugar modification, a base modification, a modification in a single strand overhang, for example a 3' single strand overhang, or, particularly if single stranded, a 5' modification which includes one or more phosphate groups or one or more analogs of a phosphate group. Examples and a further discription of oligonucleotides can be found in WO2009/126933, which is hereby incorporated by reference.
- an oligonucleotide is an antisense, miRNA, peptide nucleic acid (PNA), poly-morpholino (PMO) or siRNA.
- the preferred oligonucleotide is an siRNA.
- Another preferred oligonuleotide is the passenger strand of an siRNA.
- Another preferred oligonucleotide is the guide strand of an siRNA.
- siRNA directs the sequence-specific silencing of mRNA through a process known as RNA interference (RNAi).
- RNAi RNA interference
- the process occurs in a wide variety of organisms, including mammals and other vertebrates.
- Methods for preparing and administering siRNA and their use for specifically inactivating gene function are known.
- siRNA includes modified and unmodified siRNA. Examples and a further discription of siRNA can be found in WO2009/126933, which is hereby incorporated by reference.
- siRNA can be formulated according to any exemplary method known in the art. Examples and a further discription of siRNA
- short interfering nucleic acid refers to any nucleic acid molecule capable of inhibiting or down regulating gene expression or viral replication by mediating RNA interference ("RNAi") or gene silencing in a sequence- specific manner.
- RNAi RNA interference
- gene silencing in a sequence- specific manner.
- the siNA can be a double-stranded nucleic acid molecule comprising self-complementary sense and antisense strands, wherein the antisense strand comprises a nucleotide sequence that is complementary to a nucleotide sequence in a target nucleic acid molecule or a portion thereof and the sense strand comprises a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof.
- the siNA can be a polynucleotide with a duplex, asymmetric duplex, hairpin or asymmetric hairpin secondary structure, having self-complementary sense and antisense regions, wherein the antisense region comprises a nucleotide sequence that is complementary to a nucleotide sequence in a separate target nucleic acid molecule or a portion thereof and the sense region comprises a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof.
- the siNA can be a circular single-stranded polynucleotide having two or more loop structures and a stem comprising self-complementary sense and antisense regions, wherein the antisense region comprises nucleotide sequence that is complementary to a nucleotide sequence in a target nucleic acid molecule or a portion thereof and the sense region comprises a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof, and wherein the circular polynucleotide can be processed either in vivo or in vitro to generate an active siNA molecule capable of mediating RNAi.
- the siNA can also comprise a single-stranded polynucleotide having a nucleotide sequence complementary to nucleotide sequence in a target nucleic acid molecule or a portion thereof (for example, where such siNA molecule does not require the presence within the siNA molecule of a nucleotide sequence corresponding to the target nucleic acid sequence or a portion thereof), wherein the single-stranded polynucleotide can further comprise a terminal phosphate group, such as a '-phosphate (see for example, Martinez et al, 2002, Cell, 110, 563-574 and Schwarz et al, 2002, Molecular Cell, 10, 537-568), or 5 ',3 '-diphosphate.
- Linkers such as a '-phosphate (see for example, Martinez et al, 2002, Cell, 110, 563-574 and Schwarz et al, 2002, Molecular Cell, 10, 537-568), or 5 ',3 '-diphosphate.
- linker may be cleavable or non-cleavable, depending on the application.
- a cleavable linker may be used to release the oligonucleotide after transport from the endosome to the cytoplasm.
- Linker groups may be combined or branched to provide more complex architectures. Suitable linkers include those as described in WO2009/126933, which is hereby incorporated by reference.
- linkers of the instant invention are shown in Table 1 :
- R H, Boc, Cbz, Ac, PEG, lipid, targeting ligand, linker(s) and/or peptide(s).
- n 0 to 750.
- nucleotide can be substituted with non-nucleotide moiety such as abasic or linkers as are generally known in the art.
- R H, Boc, Cbz, Ac, PEG, lipid,
- n 0 to 750.
- nucleotide can be substituted with
- non-nucleotide moiety such as
- linkers are available from various suppliers such as Pierce or Quanta Biodesign including combinations of said linkers.
- commercial linkers attached via phosphate bonds can be used independently as linkers or in combination with said linker.
- the linkers may also be combined to produce more complex branched architectures accomodating from 1 to 8 tetraGalNAc ligands as illustrated in one such example below:
- the modular compositions of the present invention may comprise a targeting ligand.
- this targeting ligand may direct the modular composition to a particular cell.
- the targeting ligand may specifically or non-specifically bind with a molecule on the surface of a target cell.
- the targeting moiety can be a molecule with a specific affinity for a target cell.
- Targeting moieties can include antibodies directed against a protein found on the surface of a target cell, or the ligand or a receptor-binding portion of a ligand for a molecule found on the surface of a target cell. Examples and a further discription of targeting ligands can be found in WO2009/126933, which is hereby incorporated by reference.
- the targeting ligands are selected from the group consisting of an antibody, a ligand-binding portion of a receptor, a ligand for a receptor, an aptamer, D-galactose, N- acetyl-D-galactose (GalNAc), multivalent N-acetyl-D-galactose, D-mannose, cholesterol, a fatty acid, a lipoprotein, folate, thyrotropin, melanotropin, surfactant protein A, mucin, carbohydrate, multivalent lactose, multivalent galactose, N-acetyl-galactosamine, N-acetyl- glucosamine, multivalent mannose, multivalent fructose, glycosylated polyaminoacids, transferin, bisphosphonate, polyglutamate, polyaspartate, a lipophilic moiety that enhances plasma protein binding, a steroid, bile acid, vitamin B12
- the preferred targeting ligands are selected from the group consisting of D- galactose, N-acetyl-D-galactose (GalNAc), GalNAc2, and GalNAc3, cholesterol, folate, and analogs and derivatives thereof.
- Lipophilic moieties such as cholesterol or fatty acids, when attached to highly hydrophilic molecules such as nucleic acids can substantially enhance plasma protein binding and consequently circulation half life.
- lipophilic groups can increase cellular uptake.
- lipids can bind to certain plasma proteins, such as lipoproteins, which have consequently been shown to increase uptake in specific tissues expressing the corresponding lipoprotein receptors (e.g., LDL-receptor or the scavenger receptor SR-B1).
- Lipophilic conjugates can also be considered as a targeted delivery approach and their intracellular trafficking could potentially be further improved by the combination with endosomolytic agents.
- Exemplary lipophilic moieties that enhance plasma protein binding include, but are not limited to, sterols, cholesterol, fatty acids, cholic acid, lithocholic acid, dialkylglycerides, diacylglyceride, phospholipids, sphingolipids, adamantane acetic acid, 1- pyrene butyric acid, dihydrotestosterone, l,3-Bis-0(hexadecyl)glycerol, geranyloxyhexyl group, hexadecylglycerol, borneol, menthol, 1,3 -propanediol, heptadecyl group, palmitic acid, myristic acid, 03-(oleoyl)lithocholic acid, 03-(oleoyl)cholenic acid, dimethoxytrityl, phenoxazine, aspirin, naproxen, ibuprofen, vitamin E and biotin etc
- the preferred lipid is cholesterol.
- the modular composition may comprise one or more other moieties/ligands that may enhance aqueous solubility, circulation half life and/or cellular uptake.
- moieties/ligands that may enhance aqueous solubility, circulation half life and/or cellular uptake.
- These can include naturally occurring substances, such as a protein (e.g., human serum albumin (HSA), low-density lipoprotein (LDL), high-density lipoprotein (HDL), or globulin); or a carbohydrate (e.g perhaps a dextran, pullulan, chitin, chitosan, inulin, cyclodextrin or hyaluronic acid).
- HSA human serum albumin
- LDL low-density lipoprotein
- HDL high-density lipoprotein
- globulin e.g., a carbohydrate
- moieties may also be a recombinant or synthetic molecule, such as a synthetic poly
- Examples include polylysine (PLL), poly L-aspartic acid, poly L-glutamic acid, styrene-maleic acid anhydride copolymer, poly(L-lactide-co- glycolied) copolymer, divinyl ether-maleic anhydride copolymer, N-(2- hydroxypropyl)methacrylamide copolymer (HMPA), polyethylene glycol (PEG, e.g., PEG- 0.5K, PEG-2K, PEG-5K, PEG-10K, PEG-12K, PEG-15K, PEG-20 , PEG-40 ), methyl- PEG (mPEG), [mPEG]2, polyvinyl alcohol (PVA), polyurethane, poly(2 ethylacryllic acid), N-isopropylacrylamide polymers, or polyphosphazine.
- PLL polylysine
- PEG poly L-aspartic acid
- poly L-glutamic acid styrene
- the preferred solubilizing group is PEG 0.5K to 30K.
- the invention features, a method of treating a subject at risk for or afflicted with a disease that may benefit from the administration of the modular composition of the invention.
- the method comprises administering the modular composition of the invention to a subject in need thereof, thereby treating the subject.
- the oligonucleotide that is administered will depend on the disease being treated. See WO2009/126933 for additional details regarding methods of treatments for specific indications.
- Linker groups may be connected to the oligonucleotide or siRNA strand(s) at a linkage attachment point (LAP) and may include any carbon-containing moiety, in some embodiments having at least one oxygen atom, at least one phosphorous atom, and/or at least one nitrogen atom.
- the phosphorous atom forms part of a terminal phosphate, or phosphorothioate, group on the linker group, which may serve as a connection point for the oligonucleotide strand.
- the nitrogen atom forms part of a terminal ether, ester, amino or amido (NHC(O)-) group on the linker group, which may serve as a connection point for the linkers of interest, endosomo lytic unit, cell penetrating peptide, solubilizing group, lipid, targeting group, or additional linkers of interest.
- These terminal linker groups include, but are not limited to, a Ce hexyl, C5 secondary-hydroxy, C3 thiol or Ce thiol moiety.
- An example from the RNA sequences described below is C6 hexyl: [(CH 2 ) 6 NH 2 ] .
- ome 2' methoxy
- flu 2' fluoro
- click 2' propagyl
- iB inverted abasic
- siRNA depictions below are for illustrative purposes. Specific sequence information can be found in Table 3.
- Lys-alkyne Compound 1 130 mg, 0.436 mmol
- GalNAc Azide 6 999 mg, 2.178 mmol
- Copper (I) bromide-dimethyl sulfide complex 17.91 mg, 0.087 mmol
- the reaction color changed to blue/green, indicating Cu 2+
- fresh sodium ascorbate 37 mg in 0.2 mL of water was added to reaction mixture and allowed to react overnight.
- the reaction was concentrated and purified by RP HPLC 5-60 MeCN(0.5% TFA)/Water(0.5% TFA) over 20 min.
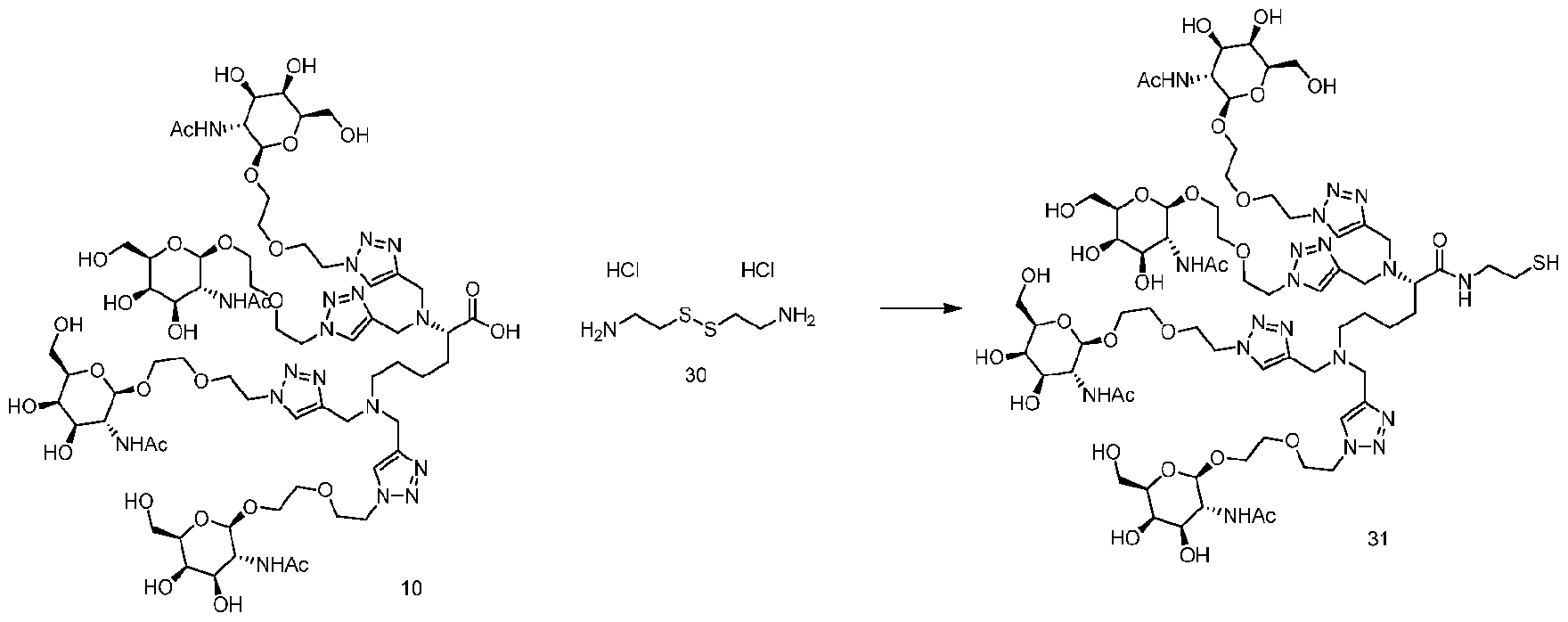
- Scheme 3 shows a general scheme that can be used to prepare tetraGalNAc-siRNA conjugates.
- Conjugates 10-1, 10-2, 10-3, lOa-l , 17a-l , 17b-l , 17c-l can be obtained.
- the coupling procedure can be performed on a preformed siRNA duplex or on a single strand followed by annealing.
- the mixture was diluted with water (1.5 mL) and purified on a XBridge Prep Phenyl column (5 uM, 19 x 250 mm) using a gradient of 0 - 30% CH 3 CN/ water containing 100 mM TEAA. The fractions were concentrated via dialysis and lyophilized to yield Compound 18.
- the concentrated mixture was purified on a XBridge Prep Phenyl column (5 uM, 19 x 250 mm) using a gradient of 0 - 50% CH 3 CN/ water containing 100 mM TEAA. The fractions were concentrated via dialysis and lyophilized to yield Compound 26.
- Step 1 Passenger-RNA and Linker, Example with Proline to Illustrate Protocol
- the resulting solution was purified by Gilson PLC 2020 using XBRIDGE PHENYL, 10 - 27% CH 3 CN with 200 ⁇ TEAA for 35 minutes. Collection solution was concentrated via centrifugal dialysis using 3kDa cut-off membrane. The resulting concentrated solution was treated with 1.0N NaCl and centrifugal dialysis. The process was repeated five times with water (14 mL each time). The resulting concentrated solution ( ⁇ 1.5 mL) was frozen and lyophilized overnight to yield product as a white fluffy solid. LC/MS confirms product [9002.5].
- SPDP Acid (2.2 mg, 10.3 ⁇ ) was dissolved DMSO 100 ⁇ . and N,N- diisopropylethylamine (14.0 ⁇ , 0.08 mmol), HATU (19.6 mg, 0.051 mmol) were added sequentially.
- RNA 15 mg, 2.06 ⁇ ) in 200 ⁇ , of DMSO: Water (9:1) was added and the resulting reaction mixture was stirred for 1 h, reaction was quenched by addition of 3 mL water and dialyzed down to 500 ⁇ ⁇ , diluted by formamide to 3 mL and purified by SAX (Buffer A: 60% TFE in water, 20 mM TEA, Buffer B: 60% TFE in water, 20 mM TEA, 1 M CsCl, gradient A/B from 100/0 to 35/65 over 15 min). The collected fractions were combined and dialyzed against water and lyophilized to afford Compound 46 as a white solid. Calculated mass: [M-H] " : C234H300F8N72O150P23S3, 7480.1; observed: 7483.0.
- the reaction mixture was purified by SAX (Buffer A: 60% TFE in water, 20 mM TEA, Buffer B: 60% TFE in water, 20 mM TEA, 1 M CsCl, gradient A/B from 100/0 to 35/65 over 15 min).
- the collected fractions were combined and dialyzed against water and lyophilized to afford Conjuate 47 as a white solid. Calculated mass: [M-H] " :
- Conjugate 47 (10.9 mg, 1.20 ⁇ ) and guide strand (7.81 mg, 1.14 ⁇ ) were mixed in RNAse free water lmL for 2 h. The reaction mixture was lyophilized to afford duplex Conjugate 48 in quantitative yield.
- RNA Compound 49 33.3mg of siRNA passenger strand was weighed into a 4mL vial then lmL lOOmM NaHC03 was added to dissolve. Added 0.86uL of propionic anhydride and let stir at RT. After aging ⁇ 2h, spin dialyzed 3x against water. Filtered through frit and the solution was dried via lypophilization to afford 30.8mg RNA Compound 49.
- Step 2 Charge Compound 10 and 1ml DMSO. Charge 6uL of DIPEA and agitate for 2min. Charge 6mg HBTU and agitate for 2min. Charge siRNA mixture from Step 1. The reaction was not complete so repeated with half of previous reagent charge.
- Conjugate 49 10.20mg in 1.17ml water. Added 8.7mg of Conjugate 49 to all of Conjugate 50 to form a 1 : 1 duplex. Heat to 90°C for lmin, cool to RT over 15min. The solution was filtered and dried via lyophilizaiton to afford Conjugate 51 as a white solid.
- HE 293 cells stably transfected with luciferase vector that contains target sites for siRNA in 3'UTR of renilla luciferase were generated. These cells were seeded on 96-well tissue culture plates (Corning: # 3903) at a density of 7.5e3 cells per well in
- DMEM 10% serum media fetal calf serum
- Cellular plates were then incubated at 37°C / 5% C02 for 24hr. After incubation, plates were treated with test compounds co-transfected with transfection reagent Lipofectamine 2000 (invitrogen: # 11668-019) in Opti-MEM (Gibco: #31985) in accordance to manufacturers protocol. The treatment concentrations ranged from ⁇ to 0.03pM. Treated plates were then incubated for 24hr at 37°C / 5% C02. Following treatment incubation, cells were lysed and processed in accordance to Dual- GloTM Luciferase Assay (Promega: E2920) and read on a TECAN safire2 plate reader.
- Dual- GloTM Luciferase Assay Promega: E2920
- HepG2 cells (ATCC: HB-8065) were seeded on collagen coated plates (BioCoat: 356649) at a density of 7.5e3 cells per well in DMEM 10% serum media.
- Resulting cell lysate was reverse transcribed to cDNA using High Capacity cDNA Kit (AB: 4368813) and run through qPCR using Life Technology 7900.
- CD1 female mice were dosed by subcutaneous injection in 200 ul volume. Animals were observed for behavioral or physiological changes. Animals were sacrificed 72hrs post dose by C02 asphyxiation followed by ex-sanguination via cardiac puncture. The liver samples were as 3mm punches from the medial lobe and put into RNAlater tubes for isolation of total RNA. The mRNA knockdown analysis was conducted by Taqman analysis using standard procedures.
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015510418A JP6239592B2 (en) | 2012-05-02 | 2013-05-01 | Novel conjugates comprising TETRAGALNAC and methods for delivering oligonucleotides |
| AU2013256400A AU2013256400B2 (en) | 2012-05-02 | 2013-05-01 | Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| EP13784779.4A EP2844663B1 (en) | 2012-05-02 | 2013-05-01 | Tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| US14/398,317 US9540639B2 (en) | 2012-05-02 | 2013-05-01 | Tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| CA2872100A CA2872100A1 (en) | 2012-05-02 | 2013-05-01 | Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| HK15108687.4A HK1208227A1 (en) | 2012-05-02 | 2015-09-07 | Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides galnac |
| US15/363,490 US10214742B2 (en) | 2012-05-02 | 2016-11-29 | Tetragalnac containing conjugates and methods for delivery of oligonucleotides |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261641761P | 2012-05-02 | 2012-05-02 | |
| US61/641,761 | 2012-05-02 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/398,317 A-371-Of-International US9540639B2 (en) | 2012-05-02 | 2013-05-01 | Tetragalnac containing conjugates and methods for delivery of oligonucleotides |
| US15/363,490 Continuation US10214742B2 (en) | 2012-05-02 | 2016-11-29 | Tetragalnac containing conjugates and methods for delivery of oligonucleotides |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013166121A1 true WO2013166121A1 (en) | 2013-11-07 |
Family
ID=49514844
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2013/039025 WO2013166121A1 (en) | 2012-05-02 | 2013-05-01 | Novel tetragalnac containing conjugates and methods for delivery of oligonucleotides |
Country Status (9)
| Country | Link |
|---|---|
| US (2) | US9540639B2 (en) |
| EP (1) | EP2844663B1 (en) |
| JP (2) | JP6239592B2 (en) |
| AR (1) | AR090906A1 (en) |
| AU (1) | AU2013256400B2 (en) |
| CA (1) | CA2872100A1 (en) |
| HK (1) | HK1208227A1 (en) |
| TW (2) | TW201808342A (en) |
| WO (1) | WO2013166121A1 (en) |
Cited By (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014179625A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | COMPOSITIONS AND METHODS FOR MODULATING APOLIPOPROTEIN (a) EXPRESSION |
| EP2844662A1 (en) * | 2012-05-02 | 2015-03-11 | Sirna Therapeutics, Inc. | Novel tetragalnac and peptide containing conjugates and methods for delivery of oligonucleotides |
| WO2015006740A3 (en) * | 2013-07-11 | 2015-05-07 | Alnylam Pharmaceuticals, Inc. | Oligonucleotide-ligand conjugates and process for their preparation |
| JP2015105230A (en) * | 2013-11-28 | 2015-06-08 | ソマール株式会社 | Sugar chain-containing polymer and sugar chain-containing polymer composite |
| WO2015168589A2 (en) | 2014-05-01 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating angiopoietin-like 3 expression |
| WO2015168172A1 (en) | 2014-04-28 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Linkage modified oligomeric compounds |
| WO2015168618A2 (en) | 2014-05-01 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| KR20160147892A (en) * | 2014-05-01 | 2016-12-23 | 아이오니스 파마수티컬즈, 인코포레이티드 | Compositions and methods for modulating complement factor b expression |
| WO2017053722A1 (en) | 2015-09-24 | 2017-03-30 | Ionis Pharmaceuticals, Inc. | Modulators of kras expression |
| WO2017079745A1 (en) | 2015-11-06 | 2017-05-11 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds for use in therapy |
| WO2018014041A2 (en) | 2016-07-15 | 2018-01-18 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulation of smn2 |
| US10023861B2 (en) | 2011-08-29 | 2018-07-17 | Ionis Pharmaceuticals, Inc. | Oligomer-conjugate complexes and their use |
| US10246709B2 (en) | 2016-03-07 | 2019-04-02 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands for therapeutic compounds |
| US10294474B2 (en) | 2016-09-02 | 2019-05-21 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands |
| WO2019140452A1 (en) | 2018-01-15 | 2019-07-18 | Ionis Pharmaceuticals, Inc. | Modulators of dnm2 expression |
| WO2019193189A1 (en) * | 2018-04-05 | 2019-10-10 | Silence Therapeutics Gmbh | siRNAs WITH AT LEAST TWO LIGANDS AT DIFFERENT ENDS |
| WO2019217527A1 (en) | 2018-05-09 | 2019-11-14 | Ionis Pharmaceuticals, Inc. | Compounds and methods for reducing fxi expression |
| US10557137B2 (en) | 2015-11-06 | 2020-02-11 | Ionis Pharmaceuticals, Inc. | Modulating apolipoprotein (a) expression |
| WO2020033748A1 (en) | 2018-08-08 | 2020-02-13 | Arcturus Therapeutics, Inc. | Compositions and agents against nonalcoholic steatohepatitis |
| US10570169B2 (en) | 2014-05-22 | 2020-02-25 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2020061200A1 (en) | 2018-09-19 | 2020-03-26 | Ionis Pharmaceuticals, Inc. | Modulators of pnpla3 expression |
| US10745700B2 (en) | 2016-01-29 | 2020-08-18 | Kyowa Kirin Co., Ltd. | Nucleic acid conjugate |
| WO2021074772A1 (en) | 2019-10-14 | 2021-04-22 | Astrazeneca Ab | Modulators of pnpla3 expression |
| US11053502B1 (en) | 2014-10-10 | 2021-07-06 | Dicerna Pharmaceuticals, Inc. | Therapeutic inhibition of lactate dehydrogenase and agents therefor |
| EP3862362A2 (en) | 2014-05-01 | 2021-08-11 | Ionis Pharmaceuticals, Inc. | Conjugates of modified antisense oligonucleotides and their use for modulating pkk expression |
| US11149264B2 (en) | 2018-02-12 | 2021-10-19 | Ionis Pharmaceuticals, Inc. | Modified compounds and uses thereof |
| US11253601B2 (en) | 2016-07-11 | 2022-02-22 | Translate Bio Ma, Inc. | Nucleic acid conjugates and uses thereof |
| US11299737B1 (en) | 2020-02-28 | 2022-04-12 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulating SMN2 |
| WO2022140702A1 (en) | 2020-12-23 | 2022-06-30 | Flagship Pioneering, Inc. | Compositions of modified trems and uses thereof |
| US11400161B2 (en) | 2016-10-06 | 2022-08-02 | Ionis Pharmaceuticals, Inc. | Method of conjugating oligomeric compounds |
| US11447521B2 (en) | 2020-11-18 | 2022-09-20 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulating angiotensinogen expression |
| EP4092119A2 (en) | 2015-07-10 | 2022-11-23 | Ionis Pharmaceuticals, Inc. | Modulators of diacyglycerol acyltransferase 2 (dgat2) |
| WO2022266032A1 (en) | 2021-06-14 | 2022-12-22 | Generation Bio Co. | Cationic lipids and compositions thereof |
| US11661604B2 (en) | 2017-10-13 | 2023-05-30 | Dicerna Pharmaceuticals, Inc. | Methods and compositions for inhibiting expression of LDHA |
| WO2023250112A1 (en) | 2022-06-22 | 2023-12-28 | Flagship Pioneering Innovations Vi, Llc | Compositions of modified trems and uses thereof |
| EP4309732A2 (en) | 2017-03-24 | 2024-01-24 | Ionis Pharmaceuticals, Inc. | Modulators of pcsk9 expression |
| WO2024040222A1 (en) | 2022-08-19 | 2024-02-22 | Generation Bio Co. | Cleavable closed-ended dna (cedna) and methods of use thereof |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3065783A4 (en) * | 2013-11-06 | 2017-06-21 | Merck Sharp & Dohme Corp. | Dual molecular delivery of oligonucleotides and peptide containing conjugates |
| EP3610884B1 (en) * | 2013-11-06 | 2021-08-18 | Merck Sharp & Dohme Corp. | Peptide containing conjugates for dual molecular delivery of oligonucleotides |
| US11479575B2 (en) | 2019-01-22 | 2022-10-25 | Korro Bio, Inc. | RNA-editing oligonucleotides and uses thereof |
| KR20210129646A (en) | 2019-01-22 | 2021-10-28 | 코로 바이오, 인크. | RNA-editing oligonucleotides and uses thereof |
| CN111748005B (en) * | 2020-06-24 | 2022-02-11 | 河北大学 | GalNAc modified methylene blue derivative, preparation method and application thereof, liver-targeting fluorescent probe and HClO detection method |
| WO2023171587A1 (en) * | 2022-03-08 | 2023-09-14 | 大原薬品工業株式会社 | MODIFIED siRNA FOR SELECTIVELY INHIBITING EXPRESSION OF MUTANT FUS |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005041859A2 (en) | 2003-04-30 | 2005-05-12 | Sirna Therapeutics, Inc. | Conjugates and compositions for cellular delivery. |
| WO2005100584A2 (en) * | 2004-04-15 | 2005-10-27 | Glycofi, Inc. | Production of galactosylated glycoproteins in lower eukaryotes |
| WO2008036825A2 (en) | 2006-09-22 | 2008-03-27 | Dharmacon, Inc. | Duplex oligonucleotide complexes and methods for gene silencing by rna interference |
| US20090124571A1 (en) * | 2006-04-28 | 2009-05-14 | Francois Morvan | Method for the synthesis of oligonucleotide derivatives |
| WO2009126933A2 (en) | 2008-04-11 | 2009-10-15 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| WO2010039546A1 (en) | 2008-09-30 | 2010-04-08 | Bridgelux, Inc. | Led phosphor deposition |
| WO2011126974A1 (en) * | 2010-04-09 | 2011-10-13 | Merck Sharp & Dohme Corp. | Novel single chemical entities and methods for delivery of oligonucleotides |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4074344A1 (en) * | 2007-12-04 | 2022-10-19 | Arbutus Biopharma Corporation | Targeting lipids |
| US8962580B2 (en) * | 2008-09-23 | 2015-02-24 | Alnylam Pharmaceuticals, Inc. | Chemical modifications of monomers and oligonucleotides with cycloaddition |
| SG183374A1 (en) * | 2010-02-24 | 2012-09-27 | Arrowhead Res Corp | Compositions for targeted delivery of sirna |
| AR090905A1 (en) * | 2012-05-02 | 2014-12-17 | Merck Sharp & Dohme | CONJUGATES CONTAINING TETRAGALNAC AND PEPTIDES AND PROCEDURES FOR THE ADMINISTRATION OF OLIGONUCLEOTIDES, PHARMACEUTICAL COMPOSITION |
-
2013
- 2013-04-30 AR ARP130101481A patent/AR090906A1/en unknown
- 2013-04-30 TW TW106117248A patent/TW201808342A/en unknown
- 2013-04-30 TW TW102115516A patent/TWI595885B/en active
- 2013-05-01 US US14/398,317 patent/US9540639B2/en active Active
- 2013-05-01 CA CA2872100A patent/CA2872100A1/en not_active Abandoned
- 2013-05-01 WO PCT/US2013/039025 patent/WO2013166121A1/en active Application Filing
- 2013-05-01 EP EP13784779.4A patent/EP2844663B1/en active Active
- 2013-05-01 AU AU2013256400A patent/AU2013256400B2/en active Active
- 2013-05-01 JP JP2015510418A patent/JP6239592B2/en active Active
-
2015
- 2015-09-07 HK HK15108687.4A patent/HK1208227A1/en unknown
-
2016
- 2016-11-29 US US15/363,490 patent/US10214742B2/en active Active
-
2017
- 2017-11-01 JP JP2017211436A patent/JP2018048186A/en active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005041859A2 (en) | 2003-04-30 | 2005-05-12 | Sirna Therapeutics, Inc. | Conjugates and compositions for cellular delivery. |
| WO2005100584A2 (en) * | 2004-04-15 | 2005-10-27 | Glycofi, Inc. | Production of galactosylated glycoproteins in lower eukaryotes |
| US20090124571A1 (en) * | 2006-04-28 | 2009-05-14 | Francois Morvan | Method for the synthesis of oligonucleotide derivatives |
| WO2008036825A2 (en) | 2006-09-22 | 2008-03-27 | Dharmacon, Inc. | Duplex oligonucleotide complexes and methods for gene silencing by rna interference |
| WO2009126933A2 (en) | 2008-04-11 | 2009-10-15 | Alnylam Pharmaceuticals, Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| WO2010039546A1 (en) | 2008-09-30 | 2010-04-08 | Bridgelux, Inc. | Led phosphor deposition |
| WO2011126974A1 (en) * | 2010-04-09 | 2011-10-13 | Merck Sharp & Dohme Corp. | Novel single chemical entities and methods for delivery of oligonucleotides |
Non-Patent Citations (6)
| Title |
|---|
| BIOCON;UF! CHEM., vol. 22, 2011, pages 1723 - 8 |
| DENIAUD ET AL.: "Insights in the rational design of synthetic multivalent glycoconjugates as lectin ligands.", ORG. BIOMOL. CHEM., vol. 9, 2011, pages 966 - 979, XP055040478 * |
| HERMANSON, G. T.: "Bioconjugate Techniques", 1996, ACADEMIC PRESS |
| MARTINEZ, CELL, vol. 110, 2002, pages 563 - 574 |
| SCHWARZ ET AL., MOLECULAR CELL, vol. 10, 2002, pages 537 - 568 |
| See also references of EP2844663A4 |
Cited By (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10023861B2 (en) | 2011-08-29 | 2018-07-17 | Ionis Pharmaceuticals, Inc. | Oligomer-conjugate complexes and their use |
| EP2844662A1 (en) * | 2012-05-02 | 2015-03-11 | Sirna Therapeutics, Inc. | Novel tetragalnac and peptide containing conjugates and methods for delivery of oligonucleotides |
| EP2844662A4 (en) * | 2012-05-02 | 2015-12-09 | Sirna Therapeutics Inc | Novel tetragalnac and peptide containing conjugates and methods for delivery of oligonucleotides |
| US9714421B2 (en) | 2013-05-01 | 2017-07-25 | Ionis Pharmaceuticals, Inc. | Compositions and methods |
| WO2014179620A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2014179627A2 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating hbv and ttr expression |
| EP3633039A1 (en) | 2013-05-01 | 2020-04-08 | Ionis Pharmaceuticals, Inc. | Compositions and methods |
| WO2014179626A2 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein c-iii expression |
| US9127276B2 (en) | 2013-05-01 | 2015-09-08 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US9145558B2 (en) | 2013-05-01 | 2015-09-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating HBV expression |
| US9163239B2 (en) | 2013-05-01 | 2015-10-20 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein C-III expression |
| US11851655B2 (en) | 2013-05-01 | 2023-12-26 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| WO2014179625A1 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | COMPOSITIONS AND METHODS FOR MODULATING APOLIPOPROTEIN (a) EXPRESSION |
| US11299736B1 (en) | 2013-05-01 | 2022-04-12 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US9181550B2 (en) | 2013-05-01 | 2015-11-10 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| US9181549B2 (en) | 2013-05-01 | 2015-11-10 | Isis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| WO2014179629A2 (en) | 2013-05-01 | 2014-11-06 | Isis Pharmaceuticals, Inc. | Compositions and methods |
| EP3828275A1 (en) | 2013-05-01 | 2021-06-02 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating ttr expression |
| US10883104B2 (en) | 2013-05-01 | 2021-01-05 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| US9957504B2 (en) | 2013-05-01 | 2018-05-01 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein (a) expression |
| EP3690049A1 (en) | 2013-05-01 | 2020-08-05 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein c-iii expression |
| EP3524680A1 (en) | 2013-05-01 | 2019-08-14 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating ttr expression |
| US10683499B2 (en) | 2013-05-01 | 2020-06-16 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating TTR expression |
| US9932581B2 (en) | 2013-05-01 | 2018-04-03 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating apolipoprotein C-III expression |
| US9932580B2 (en) | 2013-05-01 | 2018-04-03 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating HBV expression |
| US10808246B2 (en) | 2013-07-11 | 2020-10-20 | Alnylam Pharmaceuticals, Inc. | Oligonucleotide-ligand conjugates and process for their preparation |
| WO2015006740A3 (en) * | 2013-07-11 | 2015-05-07 | Alnylam Pharmaceuticals, Inc. | Oligonucleotide-ligand conjugates and process for their preparation |
| JP2015105230A (en) * | 2013-11-28 | 2015-06-08 | ソマール株式会社 | Sugar chain-containing polymer and sugar chain-containing polymer composite |
| US9926556B2 (en) | 2014-04-28 | 2018-03-27 | Ionis Pharmaceuticals, Inc. | Linkage modified oligomeric compounds |
| EP3647318A1 (en) | 2014-04-28 | 2020-05-06 | Ionis Pharmaceuticals, Inc. | Linkage modified oligomeric compounds |
| WO2015168172A1 (en) | 2014-04-28 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Linkage modified oligomeric compounds |
| JP2020058368A (en) * | 2014-05-01 | 2020-04-16 | アイオーニス ファーマシューティカルズ, インコーポレーテッドIonis Pharmaceuticals,Inc. | Compositions and methods for modulating complement factor B expression |
| WO2015168618A2 (en) | 2014-05-01 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| EP4219718A3 (en) * | 2014-05-01 | 2024-01-10 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor b expression |
| US10280423B2 (en) | 2014-05-01 | 2019-05-07 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor B expression |
| WO2015168589A2 (en) | 2014-05-01 | 2015-11-05 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating angiopoietin-like 3 expression |
| US11732265B2 (en) | 2014-05-01 | 2023-08-22 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor B expression |
| EP4219718A2 (en) | 2014-05-01 | 2023-08-02 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor b expression |
| EP3608406A1 (en) | 2014-05-01 | 2020-02-12 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor b expression |
| EP3862362A2 (en) | 2014-05-01 | 2021-08-11 | Ionis Pharmaceuticals, Inc. | Conjugates of modified antisense oligonucleotides and their use for modulating pkk expression |
| EP3845547A1 (en) | 2014-05-01 | 2021-07-07 | Ionis Pharmaceuticals, Inc. | Galnac3 conjugated modified oligonucleotide for modulating angiopoietin-like 3 expression |
| US9382540B2 (en) | 2014-05-01 | 2016-07-05 | Isis Pharmaceuticals, Inc | Compositions and methods for modulating angiopoietin-like 3 expression |
| US11312964B2 (en) | 2014-05-01 | 2022-04-26 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| US9994855B2 (en) | 2014-05-01 | 2018-06-12 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| AU2015252858C1 (en) * | 2014-05-01 | 2021-09-16 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating Complement Factor B expression |
| EP3137596A4 (en) * | 2014-05-01 | 2017-09-20 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating complement factor b expression |
| EP3974534A1 (en) | 2014-05-01 | 2022-03-30 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| KR102369736B1 (en) | 2014-05-01 | 2022-03-02 | 아이오니스 파마수티컬즈, 인코포레이티드 | Compositions and methods for modulating complement factor b expression |
| US10793862B2 (en) | 2014-05-01 | 2020-10-06 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| JP7001663B2 (en) | 2014-05-01 | 2022-02-04 | アイオーニス ファーマシューティカルズ, インコーポレーテッド | Compositions and Methods for Regulating Complement B Factor Expression |
| AU2015252858B2 (en) * | 2014-05-01 | 2020-12-10 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating Complement Factor B expression |
| US10875884B2 (en) | 2014-05-01 | 2020-12-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulating angiopoietin-like 3 expression |
| EP3757215A2 (en) | 2014-05-01 | 2020-12-30 | Ionis Pharmaceuticals, Inc. | Compositions and methods for modulating growth hormone receptor expression |
| KR20160147892A (en) * | 2014-05-01 | 2016-12-23 | 아이오니스 파마수티컬즈, 인코포레이티드 | Compositions and methods for modulating complement factor b expression |
| US10570169B2 (en) | 2014-05-22 | 2020-02-25 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds and their use |
| US11359203B2 (en) | 2014-10-10 | 2022-06-14 | Dicerna Pharmaceuticals, Inc. | Therapeutic inhibition of lactate dehydrogenase and agents therefor |
| US11053502B1 (en) | 2014-10-10 | 2021-07-06 | Dicerna Pharmaceuticals, Inc. | Therapeutic inhibition of lactate dehydrogenase and agents therefor |
| EP4092119A2 (en) | 2015-07-10 | 2022-11-23 | Ionis Pharmaceuticals, Inc. | Modulators of diacyglycerol acyltransferase 2 (dgat2) |
| WO2017053722A1 (en) | 2015-09-24 | 2017-03-30 | Ionis Pharmaceuticals, Inc. | Modulators of kras expression |
| WO2017079745A1 (en) | 2015-11-06 | 2017-05-11 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds for use in therapy |
| US10557137B2 (en) | 2015-11-06 | 2020-02-11 | Ionis Pharmaceuticals, Inc. | Modulating apolipoprotein (a) expression |
| US11319536B2 (en) | 2015-11-06 | 2022-05-03 | Ionis Pharmacueticals, Inc. | Modulating apolipoprotein (a) expression |
| EP4119569A1 (en) | 2015-11-06 | 2023-01-18 | Ionis Pharmaceuticals, Inc. | Conjugated antisense compounds for use in therapy |
| US10745700B2 (en) | 2016-01-29 | 2020-08-18 | Kyowa Kirin Co., Ltd. | Nucleic acid conjugate |
| US10246709B2 (en) | 2016-03-07 | 2019-04-02 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands for therapeutic compounds |
| US11253601B2 (en) | 2016-07-11 | 2022-02-22 | Translate Bio Ma, Inc. | Nucleic acid conjugates and uses thereof |
| WO2018014041A2 (en) | 2016-07-15 | 2018-01-18 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulation of smn2 |
| EP4206213A1 (en) | 2016-07-15 | 2023-07-05 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulation of smn2 |
| US11174481B2 (en) | 2016-09-02 | 2021-11-16 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands |
| US10294474B2 (en) | 2016-09-02 | 2019-05-21 | Arrowhead Pharmaceuticals, Inc. | Targeting ligands |
| US11400161B2 (en) | 2016-10-06 | 2022-08-02 | Ionis Pharmaceuticals, Inc. | Method of conjugating oligomeric compounds |
| EP4309732A2 (en) | 2017-03-24 | 2024-01-24 | Ionis Pharmaceuticals, Inc. | Modulators of pcsk9 expression |
| US11661604B2 (en) | 2017-10-13 | 2023-05-30 | Dicerna Pharmaceuticals, Inc. | Methods and compositions for inhibiting expression of LDHA |
| WO2019140452A1 (en) | 2018-01-15 | 2019-07-18 | Ionis Pharmaceuticals, Inc. | Modulators of dnm2 expression |
| US11149264B2 (en) | 2018-02-12 | 2021-10-19 | Ionis Pharmaceuticals, Inc. | Modified compounds and uses thereof |
| US11332733B2 (en) | 2018-02-12 | 2022-05-17 | lonis Pharmaceuticals, Inc. | Modified compounds and uses thereof |
| WO2019193189A1 (en) * | 2018-04-05 | 2019-10-10 | Silence Therapeutics Gmbh | siRNAs WITH AT LEAST TWO LIGANDS AT DIFFERENT ENDS |
| WO2019217527A1 (en) | 2018-05-09 | 2019-11-14 | Ionis Pharmaceuticals, Inc. | Compounds and methods for reducing fxi expression |
| WO2020033748A1 (en) | 2018-08-08 | 2020-02-13 | Arcturus Therapeutics, Inc. | Compositions and agents against nonalcoholic steatohepatitis |
| WO2020061200A1 (en) | 2018-09-19 | 2020-03-26 | Ionis Pharmaceuticals, Inc. | Modulators of pnpla3 expression |
| WO2021074772A1 (en) | 2019-10-14 | 2021-04-22 | Astrazeneca Ab | Modulators of pnpla3 expression |
| US11299737B1 (en) | 2020-02-28 | 2022-04-12 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulating SMN2 |
| US11447521B2 (en) | 2020-11-18 | 2022-09-20 | Ionis Pharmaceuticals, Inc. | Compounds and methods for modulating angiotensinogen expression |
| WO2022140702A1 (en) | 2020-12-23 | 2022-06-30 | Flagship Pioneering, Inc. | Compositions of modified trems and uses thereof |
| WO2022266032A1 (en) | 2021-06-14 | 2022-12-22 | Generation Bio Co. | Cationic lipids and compositions thereof |
| WO2023250112A1 (en) | 2022-06-22 | 2023-12-28 | Flagship Pioneering Innovations Vi, Llc | Compositions of modified trems and uses thereof |
| WO2024040222A1 (en) | 2022-08-19 | 2024-02-22 | Generation Bio Co. | Cleavable closed-ended dna (cedna) and methods of use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201808342A (en) | 2018-03-16 |
| JP6239592B2 (en) | 2017-11-29 |
| TWI595885B (en) | 2017-08-21 |
| JP2015520742A (en) | 2015-07-23 |
| EP2844663A1 (en) | 2015-03-11 |
| AU2013256400A1 (en) | 2014-11-06 |
| HK1208227A1 (en) | 2016-02-26 |
| AU2013256400B2 (en) | 2017-09-21 |
| US20150203843A1 (en) | 2015-07-23 |
| US10214742B2 (en) | 2019-02-26 |
| AR090906A1 (en) | 2014-12-17 |
| CA2872100A1 (en) | 2013-11-07 |
| US9540639B2 (en) | 2017-01-10 |
| TW201350131A (en) | 2013-12-16 |
| EP2844663A4 (en) | 2015-09-30 |
| US20170233731A1 (en) | 2017-08-17 |
| EP2844663B1 (en) | 2020-03-25 |
| JP2018048186A (en) | 2018-03-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10214742B2 (en) | Tetragalnac containing conjugates and methods for delivery of oligonucleotides | |
| US11117917B2 (en) | Tetragalnac and peptide containing conjugates and methods for delivery of oligonucleotides | |
| AU2019204784A1 (en) | Compositions and methods for modulating hbv and ttr expression | |
| EP3718572A1 (en) | Nucleic acid, composition and conjugate containing nucleic acid, preparation method and use | |
| US11918600B2 (en) | Nucleic acid, pharmaceutical composition and conjugate containing nucleic acid, and use thereof | |
| US20220031847A1 (en) | Therapeutic methods | |
| AU2011296268A1 (en) | Novel single chemical entities and methods for delivery of oligonucleotides | |
| JP2013523149A (en) | Novel single chemical and oligonucleotide delivery methods | |
| KR20220115579A (en) | Peptide Docking Vehicles for Targeted Nucleic Acid Delivery | |
| RU2816898C2 (en) | Nucleic acid, pharmaceutical composition and conjugate, method of production and use | |
| CN116474107A (en) | Conjugate, intermediate compound thereof and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13784779 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013784779 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2872100 Country of ref document: CA Ref document number: 2015510418 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14398317 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2013256400 Country of ref document: AU Date of ref document: 20130501 Kind code of ref document: A |