ANTI-PHYTOPATHOGENIC COMPOSITION
FIELD OF THE INVENTION
[0001] This invention relates to anti-phytopathogenic bacteria, compositions comprising or prepared using said anti-phytopathogenic bacteria, and the use of such bacteria and compositions as biological control agents. Methods for the biological control of phytopathogens, including phytopathogenic bacteria, and phytopathogenic fungi, including botrytis, fire blight (Envininia amylomra), Pythiu spp., Fusariiim spp. Colktotrichum spp., Penicillium spp., Xanthamonas spp., Pseudomonas pathovars (including Pseudomonas syringae pathovars such as Pseudomonas syringae pv actinidiae), and the like using the anti- phytopathogenic bacteria of the invention and compositions comprising or prepared using the anti-phytopathogenic bacteria of the invention are also provided.
BACKGROUND OF THE INVENTION
[0002] Plant diseases caused by pathogens such as bacteria and fungi are a significant economic cost to plant based agriculture and industries. Losses may arise through spoilage of produce both pre and post harvest, loss of the plants themselves, or through the reduction in growth and production abilities of tlie plant or the locus where a plant is or would be planted, through for example, soil borne pathogens.
[0003] Traditionally, control of plant pathogens has been pursued through the application of chemical pesticides. The use of chemicals is subject to a number of disadvantages. The pathogens can and have developed tolerance to chemicals over time, producing resistant populations. Indeed, resistance to pesticides is tlie greatest challenge to the viability of the horticultural industry.
[0004] The problem is particularly illustrated with reference to a number of economically important phytopathogenic fungi. Populations of botrytis worldwide are reported to be resistant to many pesticides including, for example the commonly used fungicide iprodione (Rovra!"). Other examples of plant pathogens that have developed resistance to chemical pesticides include Monilinia friicticok and Didymelta bryoniae, which have become resistant to benzimidazole. Similarly, phytopathogenic bacteria such as Pseudomonas syringae (various species) and Unvinia amyhvora have developed resistance to either copper (applied as either copper sulphate, copper hydroxide or cuprous oxide) or the antibiotic streptomycin after many years of use.
[0005] Chemical residues may also pose environmental hazards, and raise health concerns. The revival of interest in biological control such as microbial fungicides over the last 20 years has come directly from public pressure in response to concerns about chemical toxicities. Biological control presents an alternative means of controlling plant pathogens which is potentially more effective and specific than current methods, as well as reducing dependence on chemicals. Such biological control methods are perceived as a "natural" alternative to chemical pesticides with the advantage of greater public acceptance, reduced environmental contamination, and increased sustainability.
[0006] Mechanisms of biological control are diverse. One mechanism which has been demonstrated to be effective is the use of antagonistic microorganisms such as bacteria to control phytopatliogenic insects. For example, the large scale production of Bacillus thumigiensis enabled the use of this bacterio-insecticide to control painted apple moth in Auckland, New Zealand.
[0007] There is, however, little information on the successful application of biological control agents that are effective against phytopatliogenic bacteria or phytopatliogenic fungi, particularly those that are effective against both phytopatliogenic bacteria and
phytopatliogenic fungi.
[0008] There is therefore a need for pesticides, particularly fungicides and bactericides, that act faster, have increased efficacy in controlling phytopatliogenic microbial populations, in particular bacterial or fungal phytopathogens, require less frequent or less intensive application, have lower cost, or lower resulting toxicity than the currently- available pesticides.
[0009] It is therefore an object of the present invention to provide anti- phytopathogenic bacteria useful in the biological control of phytopatliogenic fungi or phytopatliogenic bacteria, or at least to provide the public with a useful choice.
SUMMARY OF THE INVENTION
[0010] Accordingly, in one aspect the present invention provides an isolated or biologically pure culture of ' Psendomonas putida strain Psl on deposit at National
Measurement Institute of Australia (NMIA) under Accession No. V13/001974 deposited 20 March 2013 or a culture having the identifying characteristics thereof.
[0011] In another aspect, the present invention provides a culture extract obtained from Pseudomonas putida strain Psl (V13/001974), or a culture having the identifying characteristics thereof.
[0012] In another aspect, the present invention provides the use of Pseudomonas putida strain Psl (V13/001974), a culture extract obtained from Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics of Pseudomonas putida strain Psl (V13/001974), or any combination thereof, together with at least one carrier in the preparation of a composition.
[0013] In a further aspect the invention provides a composition comprising
Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics of Pseudomonas putida strain Psl (V13/001974), a culture extract obtained from Pseudomonas putida strain Psl (V 13/ 001974) or a strain having the identifying characteristics of
Pseudomonas putida strain Psl (V13/001974), or any combination thereof, together with at least one carrier.
[0014] In one embodiment, the composition comprises Pseudomonas putida strain Psl (V13/ 001974) or a strain having the identifying characteristics of Pseudomonas putida strain Psl (V13/001974) together with at least one carrier.
[0015] In one embodiment, the Pseudomonas putida strain Psl (VI 3/001974) is in a reproductively viable form and amount.
[0016] The invention further relates to a method of controlling one or more phytopathogenic microbes, the method comprising contacting the one or more microbes or a locus with an effective amount of Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition
[0017] In various embodiments the one or more phytopathogenic microbes is or comprises a phytopathogenic microbial population. Accordingly in one embodiment the invention relates to a method of controlling one or more phytopathogenic microbial population, the method comprising contacting the population or a locus with an effective amount of Pseudomonas putid strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition
[0018] Accordingly, the invention relates to a method of controlling a
phytopathogenic bacterial population, the method comprising contacting the population or a locus with a bacteriocidally-effective amount of Pseudomonas putida strain Psl
(V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described.
[0019] The invention further relates to a method of controlling a phytopathogenic fungal population, the method comprising contacting the population or a locus with a fungicidally-effective amount of Pseudomonas putida strain Psl (V13/001974) or a strain
having the identifying characteristics thereof or a composition of the invention as herein described.
[0020] Such methods may be used to kill or reduce the numbers of target bacteria or fungi in a given area, or may be prophylactically applied to a locus, such as an
environmental area, to prevent infestation by one or more phytopathogenic bacteria or phytopathogenic fungi or one or more phytopathogenic bacterial or phytopathogenic fungal populations.
[0021] In another aspect, the present invention provides a mediod of reversing, wholly or in part, the resistance of a phytopathogenic bacterial population to one or more bacteriocidal agents, the method comprising contacting the phytopathogenic bacterial population or a locus with an effective amount of Pseudomonas putida strain Psl
(V'13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described.
[0022] Optionally, the method comprises contacting the phytopathogenic bacterial population with an effective amount of Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described together with one or more bacteriocidal agents.
[0023] In various embodiments, the one or more bacteriocidal agents administered is the same as that to which the bacterial population is or is predicted to be or become resistant.
[0024] In a further aspect, the invention provides a method of controlling a phytopathogenic bacterial population which has been contacted with Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described with an amount of one or more bacteriocidal agents effective to control said population.
[0025] The one or more bacteriocidal agents may be administered prior to, concurrently with, or after administration of the Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described. Accordingly, administration of the Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described and the one or more bacteriocidal agents may be simultaneous, sequential, or separate.
[0026] In another aspect, the present invention provides a method of reversing, wholly or in part, the resistance of a phytop thogenic fungal population to one or more
fungicidal agents, the method comprising contacting the phytopathogenic fungal population or a locus with an effective amount of Psendomonas putida strain Psl
(V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described.
[0027] Optionally, the method comprises contacting the phytopathogenic fungal population with an effective amount of Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described together with one or more fungicidal agents.
[0028] In various embodiments, the one or more fungicidal agents administered is the same as that to which the phytopathogenic fungal population is or is predicted to be or become resistant.
[0029] In a further aspect, the invention provides a method of controlling a phytopathogenic fungal population which has been contacted with Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described with an amount of one or more fungicidal agents effective to control said population.
[0030] The one or more fungicidal agents may be administered prior to, concurrently with, or after administration of the Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described. Accordingly, administration of the Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described and the one or more fungicidal agents may be simultaneous, sequential, or separate.
[0031] In a further aspect the present invention provides a method for controlling one or more phytopathogens, the method comprising applying to a plant or its surroundings a reproductively viable form and amount of Pseudomonas putida strain Psl (V13/001974), or a strain having the identifying characteristics of Pseudomonas putida strain Psl (V13/001974), optionally together with at least one other anti-fungal bacteria as described herein.
[0032] In one embodiment, the one or more phytopathogens are one or more phytopathogenic fungi, including one or more phytopathogenic fungi selected from the group comprising Botrytis spp. (e.g. Botrytis cinered), Venturia spp. (e.g. Venturia inequalis), Sckrotinia spp. (e.g. Sderotinia sclerotioruni), Fusanum spp. (e.g. Fusanum oxyspomm andFusarium culmoruni), Phytophthera spp., Pythomyces spp. (e.g. Pythomyces chartaru ), Assochyta spp. (e.g. Ascochyta pisi), Anthracnose causing fungi spp. (e.g. Apiognomonia spp., Colletotrichum spp.
(e.g. Colktotrichum acutatum), Disciila spp., Gheosporium spp., Ghmerelh spp., Gnomonia spp., Microdochium spp., Monographell spp., Pe^icuk spp., Pblycte a spp., Pseudope^i^- a spp.), Rhi^ ctouia spp. (e.g. Rhi^pctoui so/am), Septoria spp. (e.g. Septoria tritici), Uncinula necator (Erysiphe necator), Armillaria spp. and Phomopsis spp., and Penicillium spp..
[0033] In one embodiment, the one or more phytopathogens are one or more phytopathogenic bacteria, including one or more phytopathogenic bacteria selected from ;- a. Erivinia species; causing fire blight of pear and apple (E. amylowrd), Stewart's wilt in corn, and soft rot of fleshy vegetables.
b. Pseudomonas species; causing numerous leaf spots, blights, vascular wilts, soft rots, cankers, and galls. For example Pseudomonas syringae pv actinidiae causing bacterial canker in kiwifruit (^Actinidiae species)
c. Xanthomoiias species; causing numerous leaf spots, fruit spots, blights of annual and perennial plants, vascular wilts and citrus canker including X. campestris.
d. Pantoea species; causing wilt of corn.
e. Serratia species; S. marcescens causing yellow vine disease of cucurbits.
f. Sphingomonas species; causing brown spot of yellow Spanish melon fruit. g. Acidowrax; causing leaf spots in corn, orchids and watermelon. h. Ralftonia species; causing wilts of solanaceous crops.
i. Rhi^pbacter species; causing the bacterial gall of carrots.
j.
species; causing the corky root rot of lettuce.
k. Xylophilus species; causing the bacterial necrosis and canker of grapevines.
1. Agrobactemim species; the cause of crown gall disease.
m. Xyklla species; xylem-inhabiting, causing leaf scorch and dieback disease on trees and vines.
n. Candidatus liberobacter, Phloem inhabiting bacteria causing Citrus Greening
Disease (Huanglongbing, HLB).
o. Bacillus species; causing rot of tubers, seeds, and seedlings and white stripe of wheat.
p. Clostridium species; causing rot of stored tubers and leaves and wetwood of elm and poplar.
q. Arthrobacter species; causing bacterial blight of holly, thought to be the cause of Douglas-fir bacterial gall,
r. Ckvibacter species; causing bacterial wilts in alfalfa, potato, and tomato.
s. Curtobactenum species; causing wilt in beans and other plants.
t. Lxifsonia species; causing ratoon stunting of sugarcane.
u. Jjodococcus species; causing fasciation of sweet pea.
v. Streptomjces species; causing common potato scab.
[0034] In a further aspect the present invention provides a method for controlling one or more phytopathogenic microbes, the method comprising applying to a plant or its surroundings a composition of the present invention.
[0035] In one embodiment, the one or more phytopathogenic microbes is one or more phytopathogenic fungi.
[0036] In one embodiment, the one or more phytopathogenic microbes is one or more phytopathogenic bacteria.
[0037] In one embodiment, the one or more phytopathogenic microbes is one or more phytopathogenic fungi and one or more phytopathogenic bacteria.
[0038] In still a further aspect, the invention provides a method of producing a composition comprising Pseudomona putida strain Psl (V13/001974), optionally together with one or more other anti-phytopathogenic microorganism as described herein, said method comprising admixing a reproductively viable form of Pseudomonas putida strain Psl (V13/001974) with at least one agriculturally acceptable carrier.
[0039] In still a further aspect, the invention provides a method for producing a biological control composition, the method comprising
providing a culture of Pseudomonas putida strain Psl (V13/001974),
maintaining the culture in media under conditions suitable for growth of
Pseudomonas putida strain Psl (V13/001974); and
admixing the Pseudomonas putida strain Psl (V13/001974) with a carrier.
[0040] In one embodiment, the carrier is water.
[0041] In still a further aspect, the invention provides a method for producing a biological control composition, the method comprising:
providing a culture of Pseudomonas putida strain Psl (V13/001974),
maintaining die culture in media under conditions suitable for growth of
Pseudomonas putida strain Psl (V13/001974); and
i) admixing the media with a carrier, or
ii) admixing the media with one or more additional microorganisms described herein, or
iii) at least partially separating the media from the Pseudomonas putida strain Psl (V13/001974), or
iv) any combination of two or more of (i) to (iii).
[0042] In various embodiments the separation is by centrifugation or by filtration.
[0043] In various embodiments, the separation is effective to remove greater than about 50% of the Pseudomonas putida strain Psl (V13/001974), greater than about 55%, greater than about 60%, greater than about 65%, greater than about 70%, greater than about 75%, greater than about 80%, greater than about 85%, greater than about 90%, greater than about 95%, greater than about 99%, or about 100% of the Pseudomonas putida strain Psl (V13/001974).
[0044] The following embodiments may relate to any of the aspects herein.
[0045] In various embodiments, compositions of the invention comprise or may be culture extract obtained from Pseudomonas putida strain Psl (V13/001974), including a cell extract, cell suspension, cell homogenate, cell lysate, cell supernatant, cell filtrate, or cell pellet of or from Pseudomonas putida strain Psl (V13/001974).
[0046] In one embodiment, said composition is a biological control composition, for example said biological control composition is an anti-bacterial composition, an anti-fungal composition, or both an antibacterial and an antifungal composition.
[0047] For example, said biological control composition is a stable composition capable of supporting reproductive viability of the bacterial strain for a period greater than about two weeks, for example greater than about one month, about two months, about three months, about four months, about five months, for example greater than about six months.
[0048] In one embodiment, said biological control composition comprises at least one agriculturally acceptable carrier.
[0049] In one embodiment, said at least one carrier is an agriculturally acceptable carrier, for example selected from the group consisting of a filler stimulant, an anti-caking agent, a wetting agent, an emulsifier, and an antioxidant, for example said composition comprises at least one of each of a filler stimulant, an anti-caking agent, a wetting agent, an emulsifier, and an antioxidant.
[0050] In one embodiment, said filler stimulant is a carbohydrate source, such as a disaccharide including, for example, sucrose, an oligosaccharide including for example starch, fructose, glucose, mannitol or dextrose, said anti-caking agent is selected from talc, silicon dioxide, calcium silicate, or kaolin clay, said wetting agent is skimmed milk powder,
or any commercially available product such as Duwett™, Latron™, said emulsifier is a soy- based emulsifier such as lecithin or a vegetable-based emulsifier such as monodiglyceride, and said antioxidant is sodium glutamate or citric acid or potassium sorbate or an alcohol.
[0051] In certain embodiments, the composition comprises a single strain of bacteria, wherein the bacteria is Pseudomonas piitida strain Psl (V13/001974).
[0052] Alternatively, the composition comprises Pseudomonas putida strain Psl
(V13/001974) together with at least one additional strain of microorganism. In one embodiment, the at least one additional strain of microorganism, for example an anti- phytopathogenic microorganism, such as an anti-phytopathogenic strain of bacteria.
[0053] In various embodiments, the method comprises applying to a locus, such as a plant or its surroundings, Pseudomonas putida strain Psl (VI 3/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described.
[0054] In various embodiments, Pseudomona putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described is applied prophylactically, for example before die locus, such as soil, container, surroundings, or a plant, is infected by or exposed to the phytopathogenic bacterial or phytopathogenic fungal population. In other embodiments, the composition is applied when infection is established or the phytopathogenic bacterial or phytopathogenic fungal pathogen is present, for example when a locus such as a plant or its surroundings is infected by or exposed to a phytopathogenic bacterial or phytopathogenic fungal population, or when a phytopathogenic bacterial or phytopathogenic fungal population is present on or in the locus.
[0055] In one embodiment, Pseudomonas putida strain Psl (V13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described are applied directly to the locus, for example are applied directly to a plant or its surroundings. For example, a composition of the invention is admixed with a solvent or emulsified (for example with water) and applied as described herein. In other
embodiments, Pseudomonas putida strain Psl ( 13/001974) or a strain having the identifying characteristics thereof or a composition of the invention as herein described are applied indirectly to the locus, such as for example by application to a substrate that is subsequently applied to the locus.
[0056] In one embodiment, the composition is admixed with water to a final concentration of about O.lml/L to about 500ml/L prior to application, for example to a final concentration of about lml/L.
[0057] An exemplary concentration range is from about 1 x 102 to about 1 x 1012 colony forming units (CFU) per ml, from about 1 x 102 to about 1 x 1011 CFU per ml, from about 1 x 102 to about 1 x 1010 CFU per ml, from about 1 x 102 to about 1 x 109 CFU per ml, from about 1 x 103 to about 1 x 109 CFU per ml, from about 1 x 104 to about 1 x 109 CFU per ml, for example from about 1 x 106 to about 2 x 109.
[0058] In one embodiment, said composition comprises at least 107 to 109 CFU per ml at application.
[0059] In one embodiment, said application is by spraying.
[0060] In one embodiment, said application is by drenching.
[0061] In one embodiment, said application is direct application to the soil or surroundings.
[0062] In one embodiment, said application is by admixture to a growth medium, for example by adding to plant hydroponic growth media.
[0063] In one embodiment, a composition comprising Psendomonas putida strain Psl (V13/001974) or a culture having the identifying characteristics thereof is applied at a rate of from about 1 x 108 to about 1 x 1015 CFU per hectare, from about 1 x 109 to about 1 x 1015 CFU per hectare, from about 1 x 1010 to about 1 x 101S CFU per hectare, from about 1 x 10" to about 1 x 1015 CFU per hectare, for example from about 1 x 1010 to about 1 x 1014 CFU per hectare, for example from about 5 x 1010 to about 1 x 1014 CFU per hectare, for example about 1 x 1012 CFU per hectare.
[0064] Conveniently, such a rate of application can be achieved by formulating said composition at about 109 CFU per millilitre or more, and applying said composition at a rate of between about 10ml to 2 litre per hectare, for example between 50 mL to 500 mL per hectare. As discussed herein, such an application rate can be conveniently achieved by dissolution of the composition in a larger volume of agriculturally acceptable solvent, for example, water.
[0065] Embodiments of the invention are applicable to any plant or its surroundings. Exemplary plants are in certain embodiments monocotyledonous or dicotyledonous plants such as alfalfa/lucerne, apricot, apple, avocado, barley, carrot, canola, cherry, citrus, corn/maize, cotton, flax, grape, kapok, kiwifruit, lettuce, olives, peanut, pear,
pepper/ capsicum, potato, oat, rice, rye, sorghum, soybean, strawberries, sugarbeet, sugarcane, sunflower, tobacco, tomato, wheat, turf grass, pasture grass, pasture legume, berry, fruit, legume, vegetable, cane fruit, pip fruit, stone fruit, ornamental plants, nut trees, forestry trees, shrubs, cactuses, succulents, and trees.
[0066] In further illustrative embodiments, the plant may be any plant, including but not restricted to plants selected from the genus Actinidia spp., Prunus spp., Pyn/s spp., Mains spp., and Vitis spp..
[0067] To those skilled in the art to which the invention relates, many changes in construction and differing embodiments and applications of the invention will suggest themselves without departing from the scope of the invention as defined in the appended claims. The disclosures and the descriptions herein are purely illustrative and are not intended to be in any sense limiting.
[0068] In this specification where reference has been made to patent specifications, other external documents, or other sources of information, this is generally for the purpose of providing a context for discussing the features of the invention. Unless specifically stated otherwise, reference to such external documents is not to be construed as an admission that such documents, or such sources of information, in any jurisdiction, are prior art, or form part of the common general knowledge in the art.
[0069] It is intended that reference to a range of numbers disclosed herein (for example, 1 to 10) also incorporates reference to all rational numbers within that range (for example, 1, 1.1, 2, 3, 3.9, 4, 5, 6, 6.5, 7, 8, 9 and 10) and also any range of rational numbers within that range (for example, 2 to 8, 1.5 to 5.5 and 3.1 to 4.7) and, therefore, all subranges of all ranges expressly disclosed herein are hereby expressly disclosed. These are only examples of what is specifically intended and all possible combinations of numerical values between the lowest value and the highest value enumerated are to be considered to be expressly stated in this application in a similar manner.
DETAILED DESCRIPTION OF THE INVENTION
[0070] The present invention is in part directed to an anti-phytopathogenic strain of the bacteria Pseudomonas pntida strain Psl (V 13/001974) having efficacy against phytopathogens including phyto athogenic bacteria and phyto athogenic fungi, and the use of such bacteria in controlling phytopathogens.
1. Definitions
[0071] The phrases "anti-phytopathogenic activity" and "anti-phytopathogenic efficacy" are used interchangeably herein and refer to the ability of certain agents, such as certain microorganisms, to antagonise one or more phytopathogens.
[0072] In one embodiment, said anti-phytopathogenic efficacy is the ability to parasitise and incapacitate, render infertile, impede the growth of, or kill one or more phytopathogens, such as a phytopathogenic fungi, for example within 14 days of contact with the phytopathogen, for example within 7 days, for example the ability to kill one or more phytopathogens within 1-2 days.
[0073] The term "anti-bacterial" means an ability to antagonise one or more bacteria, particularly one or more phytopathogenic bacteria. Accordingly an anti-bacterial agent, such as an anti-bacterial bacterial strain, is an agent that is an antagonist of one or more bacteria, for example of one or more phytopathogenic bacterial. Such an agent is herein considered to have anti-bacterial efficacy, and encompass agents that are referred to herein as bacteriocidal agents.
[0074] The term "anti-fungal" means an ability to antagonise one or more fungi, particularly one or more phytopathogenic fungi. Accordingly an anti- fungal agent, such as an anti-fungal bacterial strain, is an agent that is an antagonist of one or more fungi, for example of one or more phytopathogenic fungi. Such an agent is herein considered to have anti-fungal efficacy, and encompass agents that are referred to herein as fungicidal agents.
[0075] The term "biological control agent" (BCA) as used herein refers to a biological agent which acts as an antagonist of one or more target organisms, for example one or more bacteria or fungi, including one or more phytopathogens, such as a phytopathogenic fungi, a phytopathogenic bacteria, or is able to control one or more populations of such organisms, including one or more populations of phytopathogens. Antagonism may take a number of forms. In one form, the biological control agent may simply act as a repellent In another form, the biological control agent may render the environment unfavourable for the phytopathogen. In a further expressly considered form, the biological control agent may parasitise, incapacitate, render infertile, impeded the growth of, and/or kill the phytopathogen. Accordingly, the antagonistic mechanisms include but are not limited to antibiosis, parasitism, infertility, and toxicity. Therefore, agents which act as antagonists of one or more phytopathogens can be said to have anti-phytopathogenic efficacy. For example, an agent that is an antagonist of a phytopathogenic fungi can be said to have anti- fungal efficacy, and in particular embodiments encompasses fungicidal agents. Likewise, an agent that is an antagonist of a phytopathogenic bacteria can be said to have anti-bacterial efficacy, and in particular embodiments encompasses bacteriocidal agents.
[0076] As used herein, a "biological control composition" is a composition comprising or including at least one biological control agent, for example includes at least
one biological control agent that is an antagonist of one or more phytopathogens. Such control agents include, but are not limited to, agents that act as repellents, agents d at render the environment unfavourable for the organism or pathogen, and agents that incapacitate, render infertile, and/or kill the organism or pathogen. Accordingly, such a composition is herein considered to encompass biological control compositions having anti-phytopathogenic efficacy.
[0077] Accordingly, as used herein an "anti-bacterial composition" is a composition which comprises or includes at least one agent that is an antagonist of one or more phytopathogenic bacteria. Such a composition is herein considered to have anti-bacterial efficacy, and in certain embodiments encompass bacteriocidal compositions. The term "anti-phytopathogenic bacterial composition" will be interpreted accordingly.
[0078] Accordingly, as used herein an "anti-fungal composition" is a composition which comprises or includes at least one agent that is an antagonist of one or more phytopathogenic fungi. Such a composition is herein considered to have anti-fungal efficacy, and in certain embodiments encompass fungicidal compositions. The term "anti- phytopathogenic fungal composition" will be interpreted accordingly.
[0079] The term "comprising" as used in this specification means "consisting at least in part of. When interpreting each statement in this specification that includes the term "comprising", features other than that or those prefaced by the term may also be present. Related terms such as "comprise" and "comprises" are to be interpreted in the same manner.
[0080] The term "control" or "controlling" as used herein generally comprehends preventing, reducing, or eradicating a population of one or more organisms, such as a phytopathogen infection or inhibiting the rate and extent of such infection, or reducing the population of said organism, such as reducing a phytopathogen population in or on a plant or its surroundings, wherein such prevention or reduction in the population(s) is statistically significant with respect to untreated population(s). Curative treatment is also contemplated. In one embodiment, such control is achieved by increased mortality amongst the population, for example increased mortality in the phytopathogen population.
[0081] As used herein, die term "culture extract" and grammatical equivalents thereof when used with reference to bacteria (including use with reference to a specific strain of bacteria such as Psendo on s species, Bacillus species, Envinia species and Xcmthomonas species) contemplates killed or attenuated bacteria such as but not limited to heat-killed, lysed, fractionated, pressure-killed, irradiated, and UV- or light-treated bacteria, material
derived from the bacteria including but not limited to bacterial cell wall compositions, bacterial cell lysates, lyophilised bacteria, and the like, as well as bacterial fermentates and fractions thereof (whether still comprising the bacteria or material derived there from or not). Culture extracts obtained from anti-phytopathogenic bacteria will desirably retain anti-phytopathogenic activity. Culture extracts obtained from one or more additional microorganisms, such as Pseudomonas species, Bacillus species, Etwi/iia species and
Xanthomonas species, will desirably retain the activity of the strain from which they were obtained, or may provide additional functionality, such as potentiating or supporting the growth or anti-phytopathogenic efficacy of the anti-phytopathogenic composition in which they may be incorporated. Methods to produce such culture extracts, such as but not limited to one or more culture extracts of Pseudomonas species, Bacillus species, Envinia species and Xanthomonas species, and particularly culture extracts suitable for use in the control of phytopathogens (for example, in a composition) are well-known in the art.
[0082] Accordingly, a "culture extract" obtained from Pseudomonas species, Bacillus species, Envinia species and Xanthomonas species as contemplated herein may comprise the media or other substrate in which Pseudomonas species, Bacillus species, Envinia species and Xanthomonas species has been grown or maintained, whether or not Pseudomonas species, Bacillus species, Envinia species and Xanthomonas species has subsequently been removed from the media or otherwise attenuated or killed. Such culture extracts may also comprise partially purified media in which Pseudomonas species, Bacillus species, Envinia species and Xanthomonas species has been grown or maintained, such as a fraction of the media.
[0083] The term "plant" as used herein encompasses not only whole plants, but extends to plant parts, cuttings as well as plant products including roots, leaves, flowers, seeds, stems, callus tissue, nuts and fruit, bulbs, tubers, corms, grains, cuttings, root stock, or scions, and includes any plant material whether pre-planting, during growth, and at or post harvest. Plants that may benefit from the application of the present invention cover a broad range of agricultural and horticultural crops. The compositions of the present invention are also especially suitable for application in organic production systems.
[0084] When used in respect of an anti-phytopathogenic agent, such as an anti- phytopathogenic bacterial strain, the phrase "retaining anti-phytopathogenic efficacy" and grammatical equivalents and derivatives thereof is intended to mean that the agent still has useful anti-phytopathogenic activity. In one embodiment, the retained activity is at least about 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 99 or 100% of the original activity, and useful ranges may be selected between any of these values (for example, from about 35
to about 100%, from about 50 to about 100%, from about 60 to about 100%, from about 70 to about 100%, from about 80 to about 100%, and from about 90 to about 100%). For example, to be useful in the present invention a strain having the identifying characteristics of a specified strain should retain anti-phytopathogenic activity, that is, retain at least about 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 99 or 100% of the anti-phytopathogenic activity of the specified strain. Accordingly, a strain having the identifying characteristics of Pseudomonas species, Envinia species and Xanthomonas species, such as a homologue or mutant of Pseudomonas species, Envinia species and Xanthomonas species, should retain at least about 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 99 or 100% of the anti- phytopathogenic activity of Pseudomonas species, Envinia species and Xanthomonas species. Similarly, exemplar}' compositions of the invention are capable of supporting the maintenance of useful anti-phytopathogenic activity of the anti-pathogenic agent (s) they comprise, and can be said to retain anti-phytopathogenic activity, ideally until applied using the methods contemplated herein.
[0085] As used herein, the term "stable" when used in relation to a composition of the invention means a composition capable of supporting reproductive viability of the anti- phytopathogenic bacteria for several weeks, for example about one, about two, about three, about four, for example about five, for example about six months, or longer.
[0086] A "strain having the identifying characteristics of Pseudomonas species, Bacillus species, Envinia species and Xanthomonas species, including a homologue or mutant of the specified strain, is closely related to (i.e., shares a common ancestor with) or derived from the specified strain, but will usually differ from the specified strain in one or more genotypic or phenotypic characteristics. Mutants are generally identifiable through assessment of genetic differences. Homologues are identifiable through assessment of the degree of genetic, biochemical and morphological difference and use of taxonomic methods, including for example analyses such as cladistics. However, a strain having the identifying characteristics of [a specified strain], including a homologue or mutant of the specified strain will retain anti-phytopathogenic efficacy, will be distinguishable from other bacterial strains, and will be identifiable as a homologue or mutant of the parent strain using the techniques described above.
[0087] The term "surroundings" when used in reference to a plant subject to the bacteria, methods and compositions of the present invention includes soil, water, leaf litter, and/or growth media adjacent to or around the plant or the roots, tubers or the like thereof, adjacent plants, cuttings of said plant, supports, water to be administered to the
plant, and coatings including seed coatings. It further includes storage, packaging or processing materials such as protective coatings, boxes and wrappers, and planting, maintenance or harvesting equipment.
2. Control of phytopathogens
[0088] The present invention recognises that the horticultural sectors of many countries are faced with the problem of increasing pesticide resistance amongst phytopathogens. This is compounded under some regulatory regimes by a reduction in the availability of new chemical pesticides due to regulatory barriers including increasing withholding periods such that the residue from such chemical pesticides renders the pesticide unusable.
[0089] The use of anti-phytopathogenic bacteria as biological control agents presents a solution to this problem. Effective biological control agents can be selected according to their ability to incapacitate or kill one or more target phytopathogens or phytopathogen populations. Under conducive conditions, phytopathogens such as phytopathogenic fungi including botrytis, apple black spot, stem rot, and the like may infect plants and their surroundings including soil, leaf litter, adjacent plants, and supports. Anti-phytopathogenic bacteria may be applied so as to incapacitate and/or kill the phytopathogens, thereby preventing or limiting the disease-causing capability of the pathogen. The effectiveness of these anti-phytopathogenic bacteria in the field is in turn dependent on their ability to survive varying climatic conditions, such as interrupted wet periods and desiccation.
[0090] The present invention further recognises there are distinct advantages to identifying and cultivating strains that are able to flourish under a wide variety of environmental conditions.
[0091] Methods to determine growth of anti-phytopathogenic bacteria under different conditions, including on or around different plant species, at different temperatures, altitudes, humidities, and on different soils, media or other substrates, are well known in the art.
[0092] Similarly, methods to establish whether an isolate is able to grow on a given artificial medium are exemplified herein. The use of such methods recognises that an isolate must be capable of being grown in sufficient quantity for it to be suitable for use as a biological control agent. Methods of growing sufficient amounts of bacteria of the invention are discussed further herein.
[0093] Likewise, methods to establish successful treatment regimens, including for example rates, routes and times of application, are presented herein. The Examples presented herein show that the bacteria, compositions and methods of the invention are effective to control phytopathogenic microbes when applied in the field.
[0094] An exemplary method for establishing efficacy of a composition, a particular application rate, or treatment regimen, of the invention, for example against a soil borne disease, is outlined as follows;
• Sow seeds into pots;
• Divide the pots into two groups (a) disease pots treated with a phytopathogenic microbe, in this example Pythium spp. mycelium, and (b) disease-free controls;
• Treat a subset of each group (i.e., half the disease pots, and half of the disease-free pots) with one or more compositions of the invention (e.g., P. p iida strain Psl with or without other anti-phytopathogenic agent(s));
• Observe disease status of plants at various time points;
where results showing less disease, less impact on growth, or better crop yield, in groups treated with compositions of the invention establish the efficacy of the particular treatment.
[0095] A strain of anti-phytopathogenic bacteria, for example a strain of anti-fungal bacteria effective against phytopathogenic fungi, and therefore suitable for use in accordance with the invention, is identified as one which is effective at reducing the population of the target phytopathogen species by a statistically significant amount with respect to the control treatment against which the strains are compared. Such strains can be considered as having anti-phytopathogenic efficacy. As described herein, the reduction in the population of the target phytopathogen may be by various antagonistic mechanisms. For example, the anti-phytopathogenic bacteria may parasitise, incapacitate, render infertile, and/or kill the phytopathogen. The anti-phytop thogenic bacteria may also reduce the population of the target phytopathogen by rendering the environment, for example the plant to which the anti-phytopathogenic bacteria is applied or its surroundings, unfavourable for the phytopathogen. In this embodiment, die anti-phytopathogenic bacteria may be considered to be acting as a repellent, and reducing the effective population of the target phytopad ogen in the vicinity of the plant or its surroundings.
[0096] In one embodiment, suitable strains exhibit at least about 5% anti- phytopathogenic efficacy, at least about 10%, at least about 15%, at least about 20%, at least about 25%, at least about 30%, at least about 35%, at least about 40%, at least about 45%, for example at least about 50% anti-phytopathogenic efficacy expressed as a
percentage reduction of the population of the relevant phytopathogen species compared to the control treatment. By way of illustration, the methodology described herein was employed to identify an anti-fungal bacteria isolate effective against a variety of target phytopathogenic fungi, whereas procedures analogous to those described herein can be employed in relation to other phytopathogens and other anti-phytopathogenic bacterial strains.
[0097] Although anti-phytopathogenic efficacy is a principal requisite for an isolate to be considered suitable for use as a biological control agent, the bacterial isolate must have additional characteristics to be suitable for use as a biological control agent.
[0098] For example, the bacterial strain must be able to be stored in a viable form for a reasonable period, ultimately so as to allow it to be applied to the target plant or its surroundings in a form and concentration that is effective as a biological control agent.
[0099] The bacterial strain should also be able to achieve infection threshold when applied to a plant or its surroundings for it to be suitable for use as a biological control agent. As used herein, infection threshold refers to the concentration of bacteria required for the bacteria to become established on the target plant or its surroundings so as to then have anti-phytopathogenic efficacy. As will be appreciated, in order to achieve infection threshold, some isolates of bacteria may require application at such a high rate as to be impractical or unviable. Furthermore, some bacterial isolates may not be able to achieve infection threshold irrespective of the concentration or rate at which they are applied. Suitable anti-phytopathogenic bacteria are typically able to achieve infection threshold when applied at a rate of not less than 1012 CFU per hectare, or applied at a concentration not less than 107 CFU per millilitre of composition when said composition is applied at a rate of about llitre/500L/hectare.
[00100] Methods to determine infection threshold are well known in the art, and examples of such methods are presented herein. In certain embodiments, infection threshold can be determined directly, for example by analysing one or more samples obtained from a target plant, its surroundings, and/or a pathogen of said plant, and determining the presence or amount of anti-phytopathogenic bacteria on or in said sample. In other embodiments, infection threshold can be determined indirecdy, for example by observing a reduction in the population of one or more phytopathogens. Combinations of such methods are also envisaged.
3. Bacterial strains of the invention
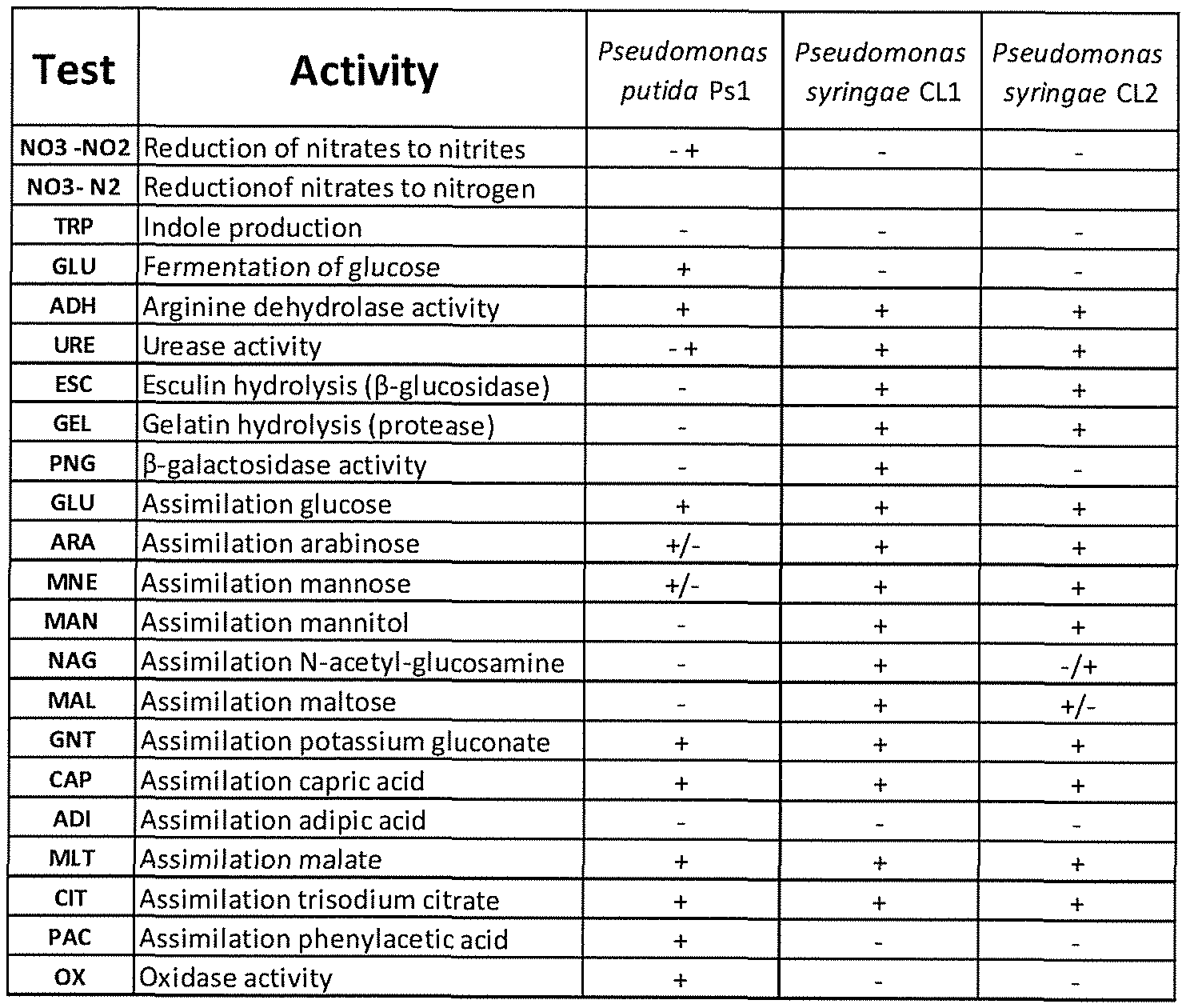
[00101] Pseudomonas putida strain Psl (V13/001974) is a gram-negative bacteria commonly found in soils. The anti-phytopathogenic Pseudomonas putida strain Psl
(V13/001974) of the invention was isolated and identified as described herein in the Examples.
[00102] Pseudomonas putida strain Psl was deposited with the National Measurement Institute of Australia (NMIA, formerly the Australian Government Analytical Laboratories (AGAL)), 1/153 Bertie Street, Port Melbourne VIC 3207, Australia on 20 March 2013 according to the Budapest Treaty on the International Recognition of the Deposit of Microorganisms for the Purpose of Patent Procedures and was designated as accession number VI 3/001974.
[00103] The deposits herein were made under the provisions of the Budapest Treaty on the International Recognition of the Deposit of Microorganisms for the Purpose of Patent Procedure and the Regulations thereunder (Budapest Treaty). This assures maintenance of a viable culture of the deposits for 30 years from the date of deposit. The deposits will be made available by the NMIA under the terms of the Budapest Treaty, and subject to an agreement between the Applicant and the NMIZ, which assures permanent and unrestricted availability of the progeny of the culture of the deposits to the public upon issuance of the pertinent U.S. or foreign patent or upon laying open to the public of any U.S. or foreign patent application, whichever comes first, and assures availability of the progeny to one determined to be entitled thereto according to the relevant jurisdictional law, for example by the U.S. Commissioner of Patents and Trademarks to be entitled thereto according to 35 U.S.C. 122 and the Commissioner's rules pursuant to thereto (including 37 C.F.R. 1.14 with particular reference to 886 OG 638).
[00104] The applicant of the present application has agreed that if a culture of the materials on deposits should die or be lost or destroyed when cultivated under suitable conditions, the materials will be promptly replaced on notification with another of the same. Availability of the deposited material is not to be construed as a license to practice the invention in contravention of the rights granted under the authority of any government in accordance with its patent laws.
[00105] Comparison of the amplified 16S rRNA variable gene region with sequence databases established the organism to be a Pseudomonas putida strain.
[00106] Accordingly, in one aspect the present invention provides a biologically pure culture of Pseudomonas putida ' strain Psl (V13/001974).
[00107] Pseudomonas putida strain Psl (V13/001974) may be further characterised by the functional attributes described herein, including its particular anti-phytopathogenic activity against the specified phytopathogens described herein, and other phenotypic characteristics such as the morphological, biochemical and growth characteristics described herein. It will be appreciated that there are a wide variety of methods known and available to the skilled artisan that can be used to confirm the identity of Pseudomonas putida strain Psl
(V13/001974), wherein exemplary methods include molecular biological methods including biochemical profile testing, DNA fingerprinting, genomic analysis, sequencing, and related genomic and proteomic techniques. In particular, methods for the identification of bacterial strains using one or more analyses of ribosomal RNA (rRNA) are well established and are amenable to application in identifying Pseudomonas putida strain Psl (V13/001974) and strains having the identifying characteristics thereof.
[00108] It will be appreciated that methods suitable for identifying Pseudomonas putida strain Psl (V13/001974), such as those described above, are similarly suitable for identifying strains having the identifying characteristics of Pseudomonas putid strain Psl
(V13/001974), including for example mutants or homologues of Pseudomonas putida strain Psl (V13/001974).
[00109] It is apparent that many phytopathogenic bacteria and phytopathogenic fungi have developed resistance to a number of chemical pesticides; in these and other instances, Pseudomonas putida strain Psl (V13/001974) provides an effective alternative for phytopathogenic bacteria and phytopathogenic fungi control. This potent activity in the control of plant disease coupled with the absence of any observations of plant
pathogenicity induced by the Pseudomonas putida strain Psl (V13/001974) strains of the invention demonstrate that these isolates have desirable attributes for use as biological control agents.
[00110] The isolates of the invention may be used singly, or in combination with other anti-phytopathogenic agents, including other anti-phytopathogenic bacteria, as described herein. Examples of other anti-phytopatinogenic bacteria are described in more detail below. 4. Compositions and methods of the invention
[00111] In a further aspect the present invention provides a composition which comprises Pseudomonas putida strain Psl (V13/001974), optionally with one or more other anti-phytopathogenic agents or bacteria, together with at least one carrier.
[00112] The composition may include multiple strains of anti-phytopathogenic bacteria, and in certain embodiments, multiple strains may be utilised to target a number of ph top thogenic species, or a number of different developmental stages of a single phytopathogen, or indeed a combination of same. For example, the conidial form of a phytopathogenic fungus may be targeted with one bacterial strain, while the adult form of the phytopathogenic fungus may be targeted with another bacterial strain, wherein both strains are included in a composition of the invention. In some embodiments, three strains or less are present, and frequently a single strain will be present.
[00113] Examples of compositions comprising bacteria are well known in the art. To be suitable for application to a plant or its surroundings, a composition comprising an anti- phytopathogenic bacterial strain of the invention will usefully comprise at least one carrier. Typically, said at least one carrier is an agriculturally acceptable carrier, for example is selected from the group consisting of a filler stimulant, an anti-caking agent, a wetting agent, an emulsifier, and an antioxidant, for example said composition comprises at least one of each of a filler stimulant, an anti-caking agent, a wetting agent, an emulsifier, and an antioxidant. In one embodiment, said filler stimulant is a carbohydrate source, such as a disaccharide including, for example, sucrose, or an oligosaccharide for example starch, fructose, glucose, mannitol or dextrose, said anti-caking agent is selected from talc, silicon dioxide, calcium silicate, or kaolin clay, said wetting agent is skimmed milk powder, said emulsifier is a soy-based emulsifier such as lecithin or a vegetable-based emulsifier such as monodiglyceride, and said antioxidant is sodium glutamate or citric acid. However, other examples well known in the art may be substituted, provided the ability of the composition to support bacterial viability is maintained.
[00114] In one embodiment, said composition is a biological control composition. The concentration of the anti-phytopathogenic bacteria of the invention present in the composition that is required to be effective as biological control agents may vary depending on the end use, physiological condition of the plant; type (including bacterial species), concentration and degree of pathogen infection; temperature, season, humidity, stage in the growing season and the age of plant; number and type of conventional fungicides or other treatments (including fungicides) being applied; and plant treatments (such as deleafing and pruning) may all be taken into account in formulating the composition.
[00115] For use as a biological control agent, when present in the composition Pseudomonas putida strain Psl (VT3/001974) will typically be in a viable form and amount. The term viable as used herein includes motile forms of the anti-phytopathogenic bacteria.
The concentration of the bacteria, for example the concentration of bacterial CFU's, in the composition will depend on the utility to which the composition is to be put. An exemplary concentration range is from about 1 x 102 to 1 x 1015 CFU's per ml, for example from about 1 x 107 to 2 x 1012, for example 1 x 107 to 1 x 109 CFU per ml.
[00116] In theory one infective unit should be sufficient to infect a host, but in actual situations a minimum number of infective units are required to initiate an infection. The concepts of lethal dose (LD) regularly used with chemical pesticides are inappropriate for phytopathogenic microbial pesticides. Concepts of infective dose (ID) or infective concentration (IC) are more precise or applicable. ID or IC refer to the actual number of infective units needed to initiate infection or the number of infective units exposed to the pathogen to cause death. Therefore, the number of infective units applied in the field or greenhouse against a pathogen will affect the degree of control. It is important to apply the desired concentration of the anti-phytopathogenic bacteria, property placed and at the right time, to obtain good control of the pest: this is known as the "infection threshold".
[00117] It will be apparent that the concentration of bacteria in a composition formulated for application may be less than that in a composition formulated for, for example, storage.
[00118] Accordingly, in one exemplary embodiment, a composition formulated for application will have a concentration of at least about 106 units (such as CFU, or such as spores) per ml. In another example, a composition formulated for storage (for example, a composition such as a wettable powder capable of formulation into a composition suitable for application) will have a concentration of about 1010 units per litre. It will be apparent that the concentration of a composition formulated for storage and subsequent formulation into a composition suitable for application must be adequate to allow said composition for application to also be sufficiently concentrated so as to be able to be applied to reach infection threshold.
[00119] In other embodiments of the present invention, Pseudomonas putida strain Psl (V13/001974) is used to prepare a composition comprising a culture extract obtained from Pseudomonas putida strain Psl (V13/001974), wherein the culture extract comprises an anti- phytopathogenic composition.
[00120] One exemplary composition comprises a culture extract obtained from
Pseudomonas putida strain Psl (VT3/001974). A culture extract as contemplated herein may be the media in which Pseudomonas putida strain Psl (V13/001974) has been grown or maintained, whether or not Pseudomonas putida strain Psl (V13/ 001974) has subsequently
been removed from the media or otherwise attenuated or killed. Culture extracts as contemplated herein may also comprise partially purified media in which Pseudomonas putida strain Psl (V13/001974) has been grown or maintained, such as a fraction of the media.
[00121] Accordingly, the invention provides methods for producing a composition comprising a culture extract obtained from Pseudomonas putida strain Psl (V13/001974) as described herein.
[00122] In one embodiment, the composition is a stable composition capable of supporting reproductive viability of said anti-phytopathogenic bacteria or anti- phytopathogenic efficacy of the anti-phytopathogenic agent for a period greater than about two weeks, for example greater than about one month, about two months, about three months, about four months, about five months, for example greater than about six months. To be suitable for use as a biological control composition, the composition is able to support reproductive viability of the bacteria or anti-phytopathogenic efficacy, and in certain exemplary embodiments is able to do so for a period greater than about six months.
[00123] Using conventional solid substrate and liquid fermentation technologies well known in the art, the anti-phytopathogenic bacteria of the invention can be grown in sufficient amounts to allow use as biological control agents. For example, culture extracts from selected strains can be produced in bulk for field application using agar plate culture, liquid culture,_nutrient film, submerged culture, and rice substrate growing techniques. Growth is generally effected under aerobic conditions at any temperature satisfactory for growth of the organism. For example, for Pseudomonas putida strain Psl (V13/001974), a temperature range of from 10 to 37°C, for example 20 to 30°C, such as 25°C, is preferred.
[00124] The culture extracts may be harvested by methods well known in the art, for example, by conventional filtering or sedimentation methodologies (e.g. centrifugation) or harvested dry using a cyclone system. Culture extracts can be used immediately or stored for some periods of time by freezing below -4°C, for up to 18 months for Pseudomonas putida strain Psl (V 13/001974). For Pseudomonas putida strain Psl (V13/001974) culture extracts can be stored for up to a few months at 4 °C.
[00125] The composition of the invention may also include one or more carriers, for example one or more agriculturally acceptable carrier. In one embodiment the carrier, such as an agriculturally acceptable carrier, can be solid or liquid. Carriers useful herein include any substance typically used to formulate agricultural composition.
[00126] In one embodiment the agriculturally acceptable carrier maybe selected from the group comprising fillers, solvents, excipients, surfactants, suspending agents,
speaders/ stickers (adhesives), antifoaming agents, dispersants, wetting agents, drift reducing agents, auxiliaries, adjuvants or a mixture thereof.
[00127] Compositions of the invention may be formulated as, for example, concentrates, solutions, sprays, aerosols, immersion baths, dips, emulsions, wettable powders, soluble powders, suspension concentrates, dusts, granules, water dispersible granules, microcapsules, pastes, gels and other formulation types by well-established procedures.
[00128] These procedures include mixing and/or milling of the active ingredients with agriculturally acceptable carrier substances, such as fillers, solvents, excipients, surfactants, suspending agents, speaders/ stickers (adhesives), antifoaming agents, dispersants, wetting agents, drift reducing agents, auxiliaries and adjuvants.
[00129] In one embodiment solid carriers include but are not limited to mineral earths such as silicic acids, silica gels, silicates, talc, kaolin, attapulgus clay, limestone, lime, chalk, bole, loess, clay, bentonite, dolomite, diatomaceous earth, aluminas calcium sulfate, magnesium sulfate, magnesium oxide, peat, humates, ground plastics, fertilizers such as ammonium sulfate, ammonium phosphate, ammonium nitrate, and ureas, and vegetable products such as grain meals, bark meal, wood meal, and nutshell meal, cellulosic powders, seaweed powders, peat, talc, carbohydrates such as mono-saccharides and di-saccharides, starch extracted from corn or potato or tapioca, chemically or physically altered corn starch and the like. As solid carriers for granules the following are suitable: crushed or fractionated natural rocks such as calcite, marble, pumice, sepiolite and dolomite; synthetic granules of inorganic or organic meals; granules of organic material such as sawdust, coconut shells, corn cobs, corn husks or tobacco stalks; kieselguhr, tricalcium phosphate, powdered cork, or absorbent carbon black; water soluble polymers, resins, waxes; or solid fertilizers. Such solid compositions may, if desired, contain one or more compatible wetting, dispersing, emulsifying or colouring agents which, when solid, may also serve as a diluent.
[00130] In one embodiment the carrier may also be liquid, for example, water; alcohols, particularly butanol or glycol, as well as their ethers or esters, particularly methylglycol acetate; ketones, particularly acetone, cyclohexanone, methylethyl ketone,
methylisobutylketone, or isophorone; petroleum fractions such as paraffinic or aromatic hydrocarbons, particularly xylenes or alkyl naphthalenes; mineral or vegetable oils; aliphatic chlorinated hydrocarbons, particularly trichloroethane or methylene chloride; aromatic chlorinated hydrocarbons, particularly chlorobenzenes; water-soluble or strongly polar
solvents such as dimethylformamide, dimethyl sulfoxide, or N-mefhylpyrrolidone; liquefied gases; or the like or a mixture thereof.
[00131] In one embodiment surfactants include nonionic surfactants, anionic surfactants, cationic surfactants and/ or amphoteric surfactants and promote the ability of aggregates to remain in solution during spraying.
[00132] Spreaders/ stickers promote the ability of the compositions of the invention to adhere to plant surfaces. Examples of surfactants, spreaders/ stickers include but are not limited to Tween and Triton (Rhom and Hass Company), Fortune®, Pulse, C. Daxoil®, Codacide oil®, D-C. Tate®, Supamet Oil, Bond®, Penetrant, Glowelt®, and Freeway, Citowett®, Fortune Plus™, Fortune Plus Lite, Fruimec, Fruimec lite, alkali metal, alkaline earth metal and ammonium salts of aromatic sulfonic acids, e.g., ligninsulfonic acid, phenolsulfonic acid, naphthalenesulfonic acid and dibutylnaphthalenesulfonic acid, and of fatty acids, alkyl and alkylaryl sulfonates, and alkyl, lauryl ether and fatty alcohol sulfates, and salts of sulfated hexadecanols, heptadecanols, and octadecanols, salts of fatty alcohol glycol ethers, condensation products of sulfonated naphthalene and naphthalene derivatives with formaldehyde, condensation products of naphthalene or
naphthalenesulfonic acids with phenol and formaldehyde, polyoxyethylene octylphenol ethers, efhoxylated isooctylphenol, ethoxylated octylphenol and ethoxylated nonylphenol, alkylphenol polyglycol ethers, tributylphenyl polyglycol ethers, alkylaryl polyether alcohols, isotridecyl alcohol, fatty alcohol ethylene oxide condensates, ethoxylated castor oil, polyoxyethylene alkyl ethers, ethoxylated polyoxypropylene, lauryl alcohol polyglycol ether acetal, sorbitol esters, lignin-sulfite waste liquors and methyl cellulose. Where selected for inclusion, one or more agricultural surfactants, such as Tween are desirably included in the composition according to known protocols.
[00133] Wetting agents reduce surface tension of water in the composition and thus increase the surface area over which a given amount of the composition may be applied. Examples of wetting agents include but are not limited to salts of polyacrylic acids, salts of lignosulfonic acids, salts of phenolsulfonic or naphthalenesulfonic acids, polycondensates of ethylene oxide with fatty alcohols or fatty acids or fatty esters or fatty amines, substituted phenols (particularly alkylphenols or arylphenols), salts of sulfosuccinic acid esters, taurine derivatives (particularly alkyltaurates), phosphoric esters of alcohols or of polycondensates of ethylene oxide with phenols, esters of fatty acids with polyols, or sulfate, sulfonate or phosphate functional derivatives of the above compounds.
[00134] In one embodiment the exemplary method of applying the compound or composition of the invention is to spray a dilute or concentrated solution by handgun or commercial airblast.
[00135] As described above, the compositions of the present invention may be used alone or in combination with one or more other agricultural agents, including pesticides, insecticides, acaracides, additional fungicides, bactericides, herbicides, antibiotics, antiphytopathogenic microbials, nematicides, rodenticides, entomopathogens, pheromones, attractants, plant growth regulators, plant hormones, insect growth regulators,
chemosterilants, phytopathogenic microbial pest control agents, repellents, viruses, phagostimulents, plant nutrients, plant fertilisers and biological controls. When used in combination with other agricultural agents the administration of the two agents may be separate, simultaneous or sequential. Specific examples of these agricultural agents are known to those skilled in the art, and many are readily commercially available.
[00136] Examples of plant nutrients include but are not limited to nitrogen, magnesium, calcium, boron, potassium, copper, iron, phosphorus, sulphate, manganese, molybdenum, cobalt, boron, copper, silicon, selenium, nickel, aluminium, chromium and zinc.
[00137] Examples of antibiotics include but are not limited to oxytetracyline and streptomycin.
[00138] Examples of fungicides include but are not limited to the following classes of fungicides: carboxamides, benzimidazoles, triazoles, hydroxypyridines, dicarboxamides, phenylamides, thiadiazoles, carbamates, cyano-oximes, cinnamic acid derivatives, morpholines, imidazoles, beta-methoxy acrylates and pyridines/pyrimidines.
[00139] Further examples of fungicides include but are not limited to natural fungicides, organic fungicides, sulphur-based fungicides, copper/calcium fungicides and elicitors of plant host defences.
[00140] Examples of natural fungicides include but are not limited to whole milk, whey, fatty acids or esterified fatty acids.
[00141] Examples of organic fungicides include but are not limited to any fungicide which passes an organic certification standard such as biocontrol agents, natural products, elicitors (some of may also be classed as natural products), and sulphur and copper fungicides (limited to restricted use).
[00142] An example of a sulphur-based fungicide is Kumulus™ DF (BASF, Germany).
[00143] An example of a copper fungicide is ocide® 2000 DF (Griffin Corporation, USA).
[00144] Examples of elicitors include but are not limited to chitosan, Bion™, BABA (DL-3-amino-n-butanoic acid, β-aminobutyric acid), salicylic acid or its derivatives (e.g. Actigard, Syngenta) and Milsana™ (Western Farm Service, Inc., USA).
[00145] In some embodiments non-organic fungicides may be employed. Examples of non-organic fungicides include but are not limited to Bravo™ (for control of powdery mildew on cucurbits); Supershield™ (Yates, NZ) (for control of Botrytis and powdery mildew on roses); Topas® 200EW (for control of PM on grapes and cucurbits); Flint™ (for control of powdery mildew on apples and cucurbits); Amistar® WG (for control of rust and powdery mildew on cereals); and Captan™, Dithane™, Euparen™, Rovral™, Scala™, Shirlan™, Switch™ and Teldor™ (for control of Botrytis on grapes).
[00146] Examples of pesticides include but are not limited to azoxystrobin, bitertanol, carboxin, Cu20, copper hydroxide, copper sulphate, cymoxanil, cyproconazole, cyprodinil, dichlofluamid, difenoconazole, diniconazole, epoxiconazole, fenpiclonil, fludioxonil, fluquiconazole, flusilazole, flutriafol, furalaxyl, guazatin, hexaconazole, hymexazol, imazalil, imibenconazole, ipconazole, kresoxim-methyl, lime sulphur, mancozeb, metalaxyl, R- metalaxyl, metconazole, oxadixyl, pefurazoate, penconazole, pencycuron, prochloraz, propiconazole, pyroquilone, SSF-109, spiroxamin, tebuconazole, thiabendazole, tolifluamid, triazoxide, triadimefon, triadimenol, triflumizole, triticonazole and uniconazole.
[00147] An example of a biological control agent other than a bacterial strain of the present invention is the BotryZen™ biological control agent comprising Olockdium oudemansii.
[00148] The compositions may also comprise a broad range of additives such as stabilisers and penetrants used to enhance the active ingredients, and so-called 'stressing' agents to improve vigour, germination and survivability such as potassium chloride, glycerol, sodium chloride and glucose. Additives may also include compositions which assist in maintaining microorganism viability in long term storage, for example unrefined corn oil and so called invert emulsions such as emulsions containing a mixture of oils and waxes on the outside and water, sodium alginate and microorganism on the inside.
[00149] It is important that any additives used are present in amounts that do not interfere with the effectiveness of the biological control agents.
[00150] Examples of suitable compositions including carriers, preservations, surfactants and wetting agents, spreaders, and nutrients are provided in US 5780023, incorporated herein in its entirety by reference.
[00151] Preferred compositions may comprise trace elements, such as but not limited to manganese, magnesium, zinc, potassium, sodium, cobalt, sulphur (which may conveniently be provided as a sulphate), molybdate, sorbate, and iron (which may conveniently be provided as a chelate); carbohydrates, such as but not limited to molasses; one or more gums, such as but not limited to guar gum, xanthan gum, locust bean gum, cassia gum, konjac flour, beta-glucan, tara gum, gum arabic, gellan gum,
carboxymethylcellulose, methylcellulose, hydroxypropyl methylcellulose, tragacanth gum, karaya gum, gum acacia, chitosan, arabinoglactins, alginate, pectin, carrageenan, or psyllium; acids, particularly weak acids such as citric acid, and other ingredients, such as one or more algae, seaweed, or extracts thereof.
[00152] An exemplary composition of the invention comprises Pseudomonas putida strain Psl (V13/001974) cultures and is made up in water.
[00153] The Applicants have also determined that many commonly used pesticides or herbicides do not adversely affect the anti-phytopathogenic bacteria of the invention. Examples are presented herein— see in particular Examples 17 - 21 herein. The compositions of the invention may therefore also include one or more additional pesticides, for example one or more additional anti-phytopathogens, including one or more additional anti-phytopathogenic bacteria or fungi. Alternatively, the compositions may be used separately but in conjunction with such pesticides in control programmes.
[00154] The invention also provides a method of producing a composition comprising one or more anti-phytopathogenic bacteria of the invention, said method comprising obtaining a reproductively viable form of said anti-phytopathogenic bacteria, and admixing said reproductively viable form of said anti-phytopathogenic bacteria with at least one agriculturally acceptable diluent, carrier or excipient.
[00155] The compositions may be prepared in a number of forms. One preparation comprises inoculating a bacterial culture media, such as King's Medium B, with a single colony of either Pseudomonas putida strain Psl (V13/001974) and then incubating
(fermenting) the media until the bacteria reach a "stationery" growth stage. This bacterial culture can then be used directly as a biocontrol agent or processed or formulated in a number of other ways.
[00156] Compositions formulated for other methods of application such as root drenching, injection, rubbing or brushing, may also be used, as are known in the art.
Indirect applications of the composition to the plant surroundings or environment such as soil, water, or as seed coatings are particularly contemplated.
[00157] As discussed above, the concentration at which the compositions comprising anti-phytopathogenic bacteria of the invention are to be applied so as to be effective biological control agents may vary depending on the end use, physiological condition of the plant; type (including bacterial species), concentration and degree of pathogen infection; temperature, season, humidity, stage in the growing season and the age of plant; number and type of conventional pesticides or other treatments (including fungicides) being- applied; and plant treatments (such as leaf plucking and pruning).
[00158] For example, in certain applications, a composition comprising Pse domonas pittid strain Psl (V13/001974) may be applied, at a rate of from about 1 x 1010 to about 1 x lO13 CFU per hectare, for example from about 1 x 1012 to about 1 x 1014 CFU per hectare, for example from about 5 x 1012 to about 1 x 1014 CFU per hectare, for example about 1-3 x 1012 CFU per hectare.
[00159] In a further aspect the present invention provides a method for controlling one or more phytopathogens, the method comprising applying to a plant or its surroundings a reproductively viable form and amount of Pseudomonas pittida strain Psl (VI 3/001 74).
[00160] In one embodiment, the phytopathogen is a phytopathogenic fungi, and in another embodiment the phytopathogen is a bacteria.
[00161] In one embodiment, the application is of Pseudomonas putida strain Psl
(V13/001974) together with one or more other anti-phytopathogenic bacteria or fungi as described herein.
[00162] In a further aspect the present invention provides a method for controlling one or more phytopathogens, the method comprising applying to a plant or its surroundings a composition as herein described.
[00163] Repeated applications at the same or different times in a crop cycle are also contemplated. The anti-phytopathogenic bacteria of the invention may be applied either earlier or later in the season. This may be over flowering or during fruiting. The anti- phytopathogenic bacteria of the invention may also be applied immediately prior to harvest, or after harvest to rapidly colonise necrotic or senescing leaves, fruit, stems, machine harvested stalks and the like to prevent phytopathogenic fungi colonisation. The
anti-phytopathogeriic bacteria of the invention may also be applied to dormant plants in winter to slow phytopathogen growth on dormant tissues.
[00164] In particular embodiments, application is at a time before or after bud burst and before and after harvest. In certain embodiments, treatment occurs between flowering and harvest. To increase efficacy, multiple applications (for example, 2 to 6 applications over the stages of flowering through fruiting) of the anti-phytopathogenic bacteria of the invention or a composition of the invention are contemplated.
[00165] Reapplication of the anti-phytopathogenic bacteria of the invention or composition should also be considered after rain. Using pathogen infectivity prediction models or infection analysis data, application of the BCA can also be timed to account for infection risk periods.
[00166] In specifically contemplated embodiments, the anti-phytopathogenic bacteria of the invention or a composition comprising same is applied in a solution, for example as described above, using a pressurised sprayer. The plant parts should be lightly sprayed until just before run off. Applications may be made to any part of the plant and/or its surroundings, for example to the whole plant canopy, to the area in the canopy where the flowers and developing fruit are concentrated, or to the plant stem and/or soil, water or growth media adjacent to or surrounding the roots, tubers or the like.
[00167] In a further embodiment, the anti-phytopathogenic bacteria can be applied directly to the soil either before or whilst the plant is growing in order to control phytopathogenic bacteria or phytopathogenic fungi, such as for example phytopathogenic bacteria or phytopathogenic fungi affecting the plant roots in order to control soil borne plant diseases. Compositions for direct application to a locus, such as the soil or surroundings of a plant or area wherein a plant is to be planted, are known in the art, and include drenches and solid dosage forms such as prills and pellets, and methods for direct application to a locus include drenching, such as drenching while seeding, undersowing, and the like.
[00168] In various exemplary embodiments, the invention relates to bacteria, compositions and methods to control soil borne fungal or bacterial diseases by, for example, soil drenching, seed coating, seed spraying, and drenching plants prior to planting out. It will be appreciated that such embodiments will frequently be prophylactic treatments, for example where a phytopathogenic microbial population is not present or is not well established, but treatments including remedial treatments where a phytopathogenic microbial population is present are also contemplated.
[00169] Specifically contemplated examples include soil drenches and soil drenching where bacteria and compositions of the invention are applied to soils to alter the microbial population in the treated area, spraying bacteria and compositions of the invention on to seeds prior to planting, for example as a seed coating and/or by spraying seed as they are sown during the seed drilling process, drenching plants in pots prior to planting out, and remediating soils from which a diseased plant has been removed.
[00170] Those skilled in the art will recognise that in certain situations; for example in orchards or other densely planted horticultures, loss of a continuous canopy is highly detrimental to the health of surrounding plants, such that the rapid recovery of a continuous canopy is highly desirable. Where loss of a continuous canopy is the result of a phytopathogenic microbe, particularly a soil borne phytopathogenic microbe, the presence of the phytopathogen at the locus where replanting would ideally occur will in most circumstances preclude replanting, and thus, preclude recovery of a continuous canopy. The bacteria, compositions and methods of the invention find particular application in such situations.
[00171] In one embodiment the anti-phytopathogenic composition is stable, including a composition capable of supporting reproductive viability of the anti-phytopathogenic bacteria or anti-phytopathogenic efficacy of the composition for several weeks, for example about one, about two, about three, about four, for example about five, for example about six months, or longer. In one embodiment, the composition is stable without a requirement for storage under special conditions, such as, for example, refrigeration or freezing.
[00172] The applied compositions control phytopathogens, including phytopathogenic fungi. Phytopathogenic fungi are responsible for many of the pre- and post-harvest diseases which attack plant parts and reduce growth rate, flowering, fruiting, and production and may cause death of afflicted plants. As used herein, phytopathogens include organisms which are diemselves plant pathogens, and organisms which may act as a vector for other plant pathogens, for example, phytopathogenic fungi or bacteria. It will be appreciated that by controlling host organisms which act as vectors for other
phytopathogens, the incidence and/ or severity of plant disease can be minimised.
[00173] The anti-phytopathogenic bacteria of the invention may also be used in the control of pathogenic organisms that exhibit their primary pathogenicity against non-plant species. Such pathogens include those which have a plant species as a vector or host (including a secondary host) but which exhibit their pathogenic effect, or their main pathogenic effect, against a non-plant species. For example, the bacteria of the invention
may be used to control fungi, such as Pythomyces spp. which are resident on plant species and produce toxins (such as sporedesmin), that cause their primary detrimental effect on subsequent consumers of the plant or plant product, such as for example a human consuming fruit or vegetables, or grazing stock (e.g. facial eczema). Those skilled in the art will recognise that the present invention has utility in the control of such pathogens, despite their primary pathogenicity being directed at a non-plant species.
[00174] Examples of the major phytopathogenic microbes afflicting a number of important horticultural crops are presented in Table 1 below.
Table 1. Major Phytopathogenic Fungi and Bacteria
1 - produces a toxin called sporedesmin that causes facial eczema in grazing livestock
[00175] Control of Pseudomonas species, Erwini species and Xanthomo/zas species in the crops outlined above using the compositions and method of the present invention is particularly contemplated in one embodiment of the invention.
[00176] In another embodiment, control of seed rot and damping off diseases are particularly contemplated. Exemplary seed rots and damping off diseases are caused by Pythium spp., Fusarium spp., Penicilium spp., Phytophthora spp.,
spp. as well as other fungi in a wide range of plant species. Specific examples include
Pythium stalk rot in corn Pythium aphanidermatum, Pythium spp.)
Seedling or damping off diseases in alfalfa {Pythium ultimum, P. irregular, P. violae,
Phytophthora megasperma, Rhi^pctonia sohni)
Damping off in tomato is typically caused by Pythium spp. (P. myriotyluni) and/or Phytophthora spp. and/or Rhi^pctonia spp.
Seedling and root rots in cucurbits is typically caused by Pythium spp., Fusarium sohni, F. equiseti, Phytophthora spp., Rhi^octonia sohni or Acremoniumi spp. In potatoes "leak" or water rot disease is typically caused by Pythium spp. Cavity spot in carrots is typically caused by Pythium sulcatum and P. viola.
In turfgrass pythium blight (grease spot) and pythium root rot is caused by Pythium spp.
Damping off or pythium root rot in floriculture or ornamental plant species is typically caused by Pythium spp. and/ or Rhi^octouia sohni and/ or other fungi.
[00177] Pythium species include; P. acanthicum, P. aphanidermatum, P. aristosporum, P. arrhenomanes, P. carolinianum, P. catenulatum, P. debaryanum, P. dissotocum, P. irregular, P. mastophorum, P. myriotylum, P. paroecandrum, P. periplocum, P.
polymastum, P. splendens, P. tracheiphilum, P. uncinulatum, and P. ultimum. The control of these and other Pythium species is expressly contemplated.
[00178] In one embodiment the invention has particular application to plants and plant products, either pre- or post-harvest. For example, the composition of the invention may be applied to stored products of the type listed above including fruits, vegetables, cut flowers and seeds. Suitable application techniques encompass those identified above, particularly spraying.
[00179] The compositions of the invention can be used to treat or pre-treat soils or seeds, as opposed to direct application to a plant. The compositions of the invention also find use in plant processing materials such as protective coatings, boxes and wrappers.
[00180] Also encompassed by the present invention are plants, plant products, soils and seeds treated directly with the bacteria of the invention or a composition of the invention.
[00181] In a further aspect, the present invention extends to the use of anti- phytopathogenic bacteria of the invention in a composition of the invention.
[00182] The invention consists in the foregoing and also envisages constructions of which the following gives examples only and in no way limit the scope thereof.
EXAMPLE 1 - Identification and isolation of Pseudomonas putida strain Psl (V13/001974)
[00183] The Pseudomonas putida strain Psl (V13/001974) was isolated from a test sample growing on Kings Medium B (Schaad, N. W. 1980. Laboratory Guide for the Identification of Plant Pathogenic Bacteria. The American Phytopathological Society, St. Paul, MN. 72 pp. (p. 3)) agar plates. Colonies of the Pseudomonas putida strain Psl (V13/001974) fluoresced under long wave UV light (365 nm) when grown on Kings Medium B or
Mannitol Glutamate Yeast Extract (MGY; Bender and Cooksey (1986) Journal of Bacteriology 165:534-541) agar plates.
[00184] The 16S rRNA gene was amplified by the polymerase chain reaction (PCR) using DNA prepared from Pseudomonas putida r strain Psl (V13/001974) as a template in the reaction. The DNA primers used were 16S-forward (5'-
AGAGTTTGATCMTGGCTCAG -3' [SEQ ID NO.l]) and 16S-Reverse (5'- GGTTACCTTGTTACGACTT -3' [SEQ ID NO.2]). The reaction consisted of 35 amplification cycles. The resulting amplified DNA fragment was characterised by DNA sequencing and the DNA sequence, presented herein as SEQ ID NO. 3, was used in BLASTN (Basic Local Alignment Search Tool) searches of the Genbank DNA databases. These DNA alignment searches established that Pseudomonas putid strain Psl (V13/001974) was a Pseudomonas putida strain.
EXAMPLE 2 - Identification and isolation of Pseudomonas putida strain Psl (V13/001974), Cherry Pseudomonas syringae strains CLl and CL2 using the API 20NE kit
[00185] The API 20NE Kit produced by Biomeriuex is suitable for characterising non- fastidious, Gram-negative rods which do not belong to the Enterobacteriaceae. Liquid cultures of Pseudomonas putid strain Psl (V13/001974), Cherry Pseudomonas syringae strains
CL1 and CL2 were prepared in Kings Media B. These bacterial cultures were used in the API 20NE kit following the manufacturer's instructions.
[00186] Results are displayed in Table 2 below. The profile for P. putida strain Psl (V13/001974) confirmed this was indeed a P.putida..
Table 2— Metabolic characterisation of bacterial strains
EXAMPLE 3— Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the bacteria Pseudomonas syringae pv actinidiae
[00187] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against a variety of pathogens. Methods
[00188] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) and the negative control Pseudomonas graminis strain BCG6 by inoculating a Kings Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00189] Test Pseudomonas Pathogens— the Pseudomonas syringae pv actinidiae (Ps ) used in these experiments was isolated from infected kiwifruit leaf spots. The identity of the
Psetidomonas synngae pv actinidiae pathogen was confirmed using a diagnostic PCR test utilising the PsaFl/R2 PCR primer pair and the procedure outlined by Rees-George et al. {Plant Pathology (2010) 59, 453-464). The DNA sequence of the resulting PCR product was determined and confirmed the identity of the Psa isolate.
[00190] Zone of Inhibition Testing— Tests were carried out on Kings Medium B or MGY agar plates. Each plate was inoculated with 0.1 ml of a cell suspension per plate of Psa (at a suitable inoculum concentrations for each respective pathogen ~107 cfu/plate), which was spread across the agar surface using a sterile rod. In each plate 4 independent holes were punched into the agar. Into each of these holes 5 μΐ of either Pseudomonas putida strain Psl (V13/001974) culture or Pseudomonas graminis strain BCG6 culture or Kings
Medium B or water was pipetted. The plate was then incubated at 25 °C for 24 hours. Tn another study, colonies for Pseudomonas putida strain Psl (V13/001974) was scraped from an MGY agar plate and resuspended in water. These bacterial suspensions were used in zone of inhibition experiments in the same manner as the cultures.
Results
[00191] There were large zones of inhibition around the holes containing either the Pseudomonas putida strain Psl (V13/001974) whereas there was no zone of inhibition for the control Pseudomonas graminis strain BCG6 or Kings Medium B or the water control. This ZOI activity was observed for both Pseudomonas putida strain Psl (V 13/001974) Kings Medium B cultures and bacterial water suspension. This indicates that the Pseudomonas putida strain Psl (V13/001974) produced compounds active against Psa isolated from a kiwi fruit leaf spot.
Discussion
[00192] Pseudomonas putida strain Psl (V13/001974) provides excellent control against Psa a plant disease-causing bacteria. The control was maintained for up to 14 days when the plates were stored at 4 °C. The results above also show that Pseudomonas putida strain Psl (V13/001974) are active when formulated in water.
EXAMPLE 4— Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against two different Psuedomonas syringae bacteria isolated from Cherry leaves
[00193] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against two Psuedomonas synngae species, strains CL1 and CL2, isolated from leaf spots on Cherry leaves.
Methods
[00194] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) and a control strain Pseudomonas graminis strain BCG6 by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00195] Test Pseudomonas Pathogens - Two Pseudomonas syringae species were isolated from the cherry leaves by extracting microbes from cherry leaf spot and growing the resultant microbes on MGY agar plates. These two cherry Pseudomonas syringae isolates, CLl and CL2, fluoresced under longwave UV light when grown on MGY agar plates. These two Pseudomonas syringae, were the major bacteria species isolated and both species fluoresced when grown on Kings Medium B agar plates and wThen subjected to long wave UV light. Both the CLl and CL2 strains was able to grow on Kings Medium B agar plates that were supplemented with either 500 ppm copper sulphate or 50 ppm streptomycin sulphate indicating that these two isolates were tolerant to copper and streptomycin.
[00196] Zone of Inhibition Testing— Tests were carried out on Kings Medium B or MGY agar plates. Each plate was inoculated with 0.1 ml of a cell suspension per plate of the test pathogen (at a suitable inoculum concentrations for each respective pathogen ~107 cfu/plate), which was spread across the agar surface using a sterile rod. In each plate 4 independent holes were punched into the agar. Into each of these holes 5 μΐ of either Pseudomonas putida strain Psl (V13/001974) culture or Pseudomonas graminis strain BCG6 culture or water was pipetted. The plate was then incubated at 25 °C for 24 hours. In another study colonies for Pseudomonas putida strain Psl (V13/001974) and Pseudomonas syringae strain BCG4 were scraped from an MGY agar plates and resuspended in water. These bacterial suspensions were used in zone of inhibition experiments in the same manner as the cultures.
Results
[00197] There were large zones of inhibition around the holes containing the
Pseudomonas putida strain Psl (V13/001974), whereas there was no zone of inhibition for the control Pseudomonas graminis strain BCG6 or the water control. This indicated that
Pseudomonas putida strain Psl (V13/001974) produced compounds active against Pseudomonas syringae strains CLl and CL2 isolated from cherry leaves.
Discussion
[00198] The Pseudomonas putida strain Psl (V13/001974) provided excellent control against the Pseudomonas syringae strains CLl and CL2. The control was maintained for up to 14 days when the plates were stored at 4 °C. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Pseudomonas syringae plant disease-causing bacteria that are resistant to copper and streptomycin treatment.
EXAMPLE 5 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against two different Psuedomonas syringae bacteria isolated from Apricot leaves
[00199] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (VT3/001974) against two Pseudomonas syringae, strains Al and A6, isolated from Apricot leaves.
Methods
[00200] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00201] Test Psuedomonas Pathogens— Two Pseudomonas syringae species were isolated from the apricot leaves collected from an orchard infected with bacterial blast disease and growing the resultant microbes on MGY agar plates. These two apricot Pseudomonas syringae isolates, Al and A6, fluoresced under long wave UV light when grown on MGY agar plates. Both the Al and A6 strains was able to grow on Kings Medium B agar plates that were supplemented with either 500 ppm copper sulphate or 50 ppm streptomycin sulphate indicating that these two isolates were tolerant to copper and streptomycin.
[00202] Zone of Inhibition Testing— Tests were carried out on Kings Medium B or MGY agar plates. Each plate was inoculated with 0.1 ml of a cell suspension per plate of the test pathogen (at a suitable inoculum concentrations for each respective pathogen—10' cfu/plate), which was spread across the agar surface using a sterile rod. In each plate 4 independent holes were punched into the agar. Into each of these holes 5 ul of either Pseudomonas putida strain Psl (VT3/ 001974) culture or Kings Medium B or water was pipetted. The plate was then incubated at 25 °C for 24 hours.
Results
[00203] There were large zones of inhibition around the holes containing either the Pseudomonas putida strain Psl (V13/001974) cultures whereas there was no zone of inhibition for the Kings Medium B or the wrater control. This indicates that the
Pseudomonas putida strain Psl (V13/001974) produced compounds active against Pseudomonas syringae strains Al and A6 isolated from apricot leaves.
Discussion
[00204] The Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Pseudomonas syringae strains Al and A6. The control was maintained for up to 14 days when the plates were stored at 4 °C. This demonstrates that Pseudomonas putida strain Psl (V 13/001974) is a control agent for Pseudomonas syringae plant disease-causing bacteria that are resistant to copper and streptomycin treatment.
EXAMPLE 6 - Identification and isolation of Pseudomonas syringae strains CL1 and CL2
[00205] The Pseudomonas syringae strains CL1 and CL2 were isolated from cherry leaves by extracting microbes from cherry leaf spots and growing the resultant microbes on MGY agar plates.
[00206] The 16S rRNA gene was amplified by the polymerase chain reaction (PCR) using DNA prepared from Pseudomonas syringae strains CL1 and CL2 as a template in the reaction. The DNA primers used were 16S-forward (AGAGTTTGATCMTGGCTCAG [SEQ ID NO. l]) and 16S-Reverse (GGTTACCTTGTTACGACTT [SEQ ID N0.2J). The reaction consisted of 35 amplification cycles.
[00207] The resulting amplified DNA fragment was characterised by DNA sequencing and the DNA sequence used in BLASTN (Basic Local Alignment Search Tool) searches of the Genbank DNA databases. These DNA alignment searches established that Pseudomonas syringae strains CL1 and CL2 was a Pseudomonas syringae strain.
[00208] The DNA sequences for Pseudomonas syringae strains CL1 and CL2 have been deposited into Genbank, and have been accorded the accession numbers KC776126 and KC776127, respectively.
EXAMPLE 7 - Identification and isolation of Pseudomonas syringae strains Al and A6
[00209] The Pseudomonas syringae strains Al and A6 were isolated from apricot leaves by extracting microbes from apricot leaf spots and growing the resultant microbes on MGY agar plates.
[00210] The 16S rRNA gene was amplified by the polymerase chain reaction (PCR) using DNA prepared from Pseudo on s syringae strains Al and A6 as a template in the reaction. The DNA primers used were 16S-forward (AGAGTTTGATCMTGGCTCAG [SEQ ID NO.l]) and 16S-Reverse (GGTTACCTTGTTACGACTT [SEQ ID NO.2]). The reaction consisted of 35 amplification cycles.
[00211] The resulting amplified DNA fragment was characterised by DNA sequencing and the DNA sequence used in BLASTN (Basic Local Alignment Search Tool) searches of the Genbank DNA databases. These DNA alignment searches established that Pseudomonas syringae strains Al and A6 was a Pseudomonas syringae strain.
[00212] The DNA sequences for Pseudomonas syringae strains Al and A6 have been deposited into Genbank, and have been accorded the accession numbers KC776128 and C776129, respectively.
EXAMPLE 8 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl V13/001974) against the bacteria Erwinia amylovora
[00213] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V 13/ 001974) against Envinia amyhvora species (ICMP 15973) .
Methods
[00214] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00215] Test Envinia Pathogen— The Erwinia amyh ra species was purchased from Landcare Research (NZ) and grown on Plate Count Agar (PCA) plates (0.5% peptone, 0.25% yeast extract, 0.1% glucose, 1.5% agar).
[00216] Zone of Inhibition Testing - Tests were carried out on PCA plates. Each plate was inoculated with 0.1 ml of a cell suspension per plate of the test pathogen (at a suitable inoculum concentrations for each respective pathogen ~107 cfu/plate), which was spread across the agar surface using a sterile rod. In each plate 3 independent holes were punched into the agar. Into each of these holes 300 μΐ of either Pseudomonas putida strain Psl
(V13/001974) culture or control media was pipetted. The plate was then incubated at
25 °C for 1-5 days.
Results
[00217] There were zones of inhibition around the holes containing the Pseudomonas putida strain Psl (V13/001974) whereas there was no zone of inhibition for the control media. This indicated that Pseudomonas putida strain Psl (V13/001974) produced compounds active against Envinia amylovora strain ICMP 15973.
Discussion
[00218] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Envinia amyhvora strain ICMP 15973. The control was maintained for up to 14 days when the plates were stored at 4 °C. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Envinia amylovora plant disease-causing bacteria.
EXAMPLE 9 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the bacteria Xanthomonas campestris
[00219] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against Xanthomonas campestris species (ICMP No. 11163) isolated from pepper {Capsicum annuum) leaf spots.
Methods
[00220] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00221] Test Xanthomonas Pathogen— The Xanthomonas campestris species was purchased from Landcare Research (NZ) and grown on Plate Count Agar (PCA) plates.
[00222] Zone of Inhibition Testing - Tests were carried out on PCA plates. Each plate was inoculated with 0.1 ml of a cell suspension per plate of the test pathogen (at a suitable inoculum concentrations for each respective pathogen ~107 cfu/plate), which was spread across the agar surface using a sterile rod. In each plate 3 independent holes were punched into the agar. Into each of these holes 300 μΐ of Pseudomonas putid strain Psl (V13/001974) culture or a control mediawas pipetted. The plate was then incubated at 25 °C for 1 to 5 days.
Results
[00223] There were zones of inhibition around the holes containing the Pseudomonas putida strain Psl (V13/001974) whereas there was no zone of inhibition for the control media. This indicated that Pseudomonas putida- strain Psl (V 13/001974) produced compounds active against Xanthomonas campestris strain ICMP 11163 isolated from pepper leaf spots.
Discussion
[00224] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Xanthomonas campestris strain ICMP 11136. The control was maintained for up to 14 days when the plates were stored at 4 °C. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Xanthomonas campestris plant disease-causing b cteria.
EXAMPLE 10 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the fungi Penicillium species
[00225] This example describes the in-vilro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against Penicillium species.
Methods
[00226] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00227] Test Penicilhum Pathogen— The Penicillium species was isolated from mouldy hay and grown on PDA agar plates.
[00228] Zone of Inhibition Testing - Tests were carried out on three PDA agar plates which had been inoculated with 0.1 ml of a PenicilRum spore suspension per plate (test pathogen) which was spread across the agar surface using a sterile rod. A central hole was punched into each plate and 300 μΐ of a Pseudomonas putida strain Psl (V13/001974) culture was pipetted into each central hole. The plates were then incubated at 25 °C for 1 to 7 days and were assessed daily for zones of inhibition.
Results
[00229] There were large zones of inhibition around the holes containing the
Pseudomonas putida strain Psl (V13/001974). This indicated that Pseudomonas putida strain Psl (V13/001974) produced compounds active against this Penicillium species.
Discussion
[00230] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Penicillium species. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Penicillium species plant disease-causing fungi. EXAMPLE 11 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the fungi Fusarium species
[00231] This example describes the in-vittv analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (VI 3/001974) against Fusatium spp. 1 (F.
oxyspornum a hay spoilage organism) and Fusatium spp. 2 (F. culmorum causes head blight of wheat).
Methods
[00232] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ΖΟΓ) studies.
[00233] Test Fusarium Pathogens - The Fusatium spp. 1 was isolated from mouldy hay and grown on PDA agar plates. The Fusatium spp. 2 was isolated from wheat infected with head blight and grown on PDA agar plates.
[00234] Zone of Inhibition Testing— Tests were carried out on PDA agar plates which had been inoculated with 0.1 ml of either Fusarium spp. 1 (3 plates) or a Fusarium spp. 2 (3 plates) spore suspension (test pathogens) which was spread across the agar surface using a sterile rod. A central hole was punched into each plate and 300 μΐ of a Pseudomonas putida strain Psl (V13/001974) culture was pipetted into each central hole. The plates were then incubated at 25 °C for 1 to 7 days and were assessed daily for zones of inhibition.
Results
[00235] There were large zones of inhibition around the holes containing the
Pseudomonas putid strain Psl (V13/001974). This indicated that Pseudomonas putida strain Psl (V13/001974) produced compounds active against these Fusatium spp. 1 and Fusarium spp. 2 species.
Discussion
[00236] Pseudomonas putid strain Psl (V13/001974) provide excellent control against the Fusarium species. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Fusarium spp. 1 and Fusarium spp. 2 plant, disease-causing fungi.
EXAMPLE 12 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the fungi Pithomyces chartarum
[00237] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against Pithomyces chartarum— the organism responsible for producing the toxin sporedesmin that causes facial eczema in grazing ruminant livestock.
Methods
[00238] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00239] Test Pithomyces chartarum Pathogen— The Pithomyces chartarum was isolated from pasture grasses and grown on PDA agar plates.
[00240] Zone of Inhibition Testing— Tests were carried out on three PDA agar plates which had been inoculated with 0.1 ml of a Pithomyces chartarum spore suspension per plate (test pathogen) which was spread across the agar surface using a sterile rod. A central hole was punched into each plate and 300 μΐ of a Pseudomonas putida strain Psl (V13/001974) culture was pipetted into each central hole. The plates were then incubated at 25 °C for 1 to 7 days and were assessed daily for zones of inhibition.
Results
[00241] There were large zones of inhibition around the holes containing the
Pseudomonas putida strain Psl VT3/001974). This indicated that Pseudomonas putida strain Psl (VT3/001974) produced compounds active against this Pithomyces chartarum.
Discussion
[00242] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Pithomyces chartarum. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Pithomyces chartaruma. fungi that produces toxins that cause diseases in grazing livestock. EXAMPLE 13 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the fungi Botrytis cinerea
[00243] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V 13/001974) against Botrytis cinerea an organism causing bunch rot and grey mould of grapes and other fruit and vegetables.
Methods
[00244] Inoculum was prepared for Psettdomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ΖΟΓ) studies.
[00245] Test Botrytis cinerea Pathogen - The Botrytis cinerea was isolated from a Botrytis- infected bunch of grapes and grown on PDA agar plates.
[00246] Zone of Inhibition Testing— Tests were carried out on three PDA agar plates which had been inoculated with 0.1 ml of a Botrytis cinere spore suspension per plate (test pathogen) which was spread across the agar surface using a sterile rod. A central hole was punched into each plate and 300 μΐ of a Pseudomonas putida strain Psl (V13/001974) culture was pipetted into each central hole. The plates were then incubated at 25 °C for 1 to 7 days and were assessed daily for zones of inhibition.
Results
[00247] There were large zones of inhibition around the holes containing the
Pseudomonas putida strain Psl (V13/001974). This indicated that Pseudomonas putida strain Psl (V13/001974) produced compounds active against this Botrytis nerea.
Discussion
[00248] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Botrytis dnerea. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Botrytis cinerea plant disease-causing fungi.
EXAMPLE 14 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the fungi Sclerotina sclerotiorum
[00249] This example describes the in-vitrv analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against Sclerotina sclerotiorum isolated from kiwifruit isolate.
Methods
[00250] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was d en used in the subsequent zone of inhibition (ZOI) studies.
[00251] Test Sclerotina sclerotiorum Pathogen - The Sclerotina sclerotiorum was isolated from an infected kiwifruit and grown on PDA agar plates.
[00252] Agar Plug Testing— The Sckrotina sclerotionm species was grown on PDA agar plates. Three agar plugs (containing fungal mycelium) made from these plates were suspended in a 1:10 dilution of the Pseudomonas putida strain Psl (V13/001974) culture for 14 hours. After the incubation the agar plugs were placed on fresh PDA agar plates.
These plates were then incubated at 25 °C for 1 to 7 days and were assessed daily for suppression of growth of fungal mycelium.
Results
[00253] There was complete suppression and inhibition of fungal mycelia growth around the holes containing the Pseudomonas putida strain Psl (V13/001974). This indicated that Pseudomonas putida strain Psl (V13/001974) produced compounds active against this Sckrotina sckrotiomm.
Discussion
[00254] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Sckrotina sckrotiomm. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Sckrotina sckrotiomm plant disease-causing fungi.
EXAMPLE 15 - Anti-phytopathogenic efficacy of Pseudomonas putida strain Psl (V13/001974) against the fungi Pythium species
[00255] This example describes the in-vitro analysis of the anti-phytopathogenic efficacy of the bacteria Pseudomonas putida strain Psl (V13/001974) against Pythium species isolated from soil.
Methods
[00256] Inoculum was prepared for Pseudomonas putida strain Psl (VI 3/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent zone of inhibition (ZOI) studies.
[00257] Test Pythium species Pathogen - The Pythiu species was isolated from soil and grown on PDA agar plates.
[00258] Agar Plug Testing— The Pythium species was grown on PDA agar plates. Three agar plugs (containing fungal mycelium) from these plates were suspended in a 1:10 dilution of the Pseudomonas putida strain Psl (V13/001974) culture for 14 hours. After the incubation the agar plugs were placed on fresh PDA agar plates. These plates were then incubated at 25 °C for 1 to 7 days and were assessed daily for suppression of growth of fungal mycelium.
Results
[00259] There was complete suppression and inhibition of fungal mycelia growth around the holes containing the Pseudomonas putida strain Psl (V13/001974). Tliis indicated that Pseudomonas putida strain Psl (V13/001974) produced compounds active against this Pythium species.
Discussion
[00260] Pseudomonas putida strain Psl (V13/001974) provide excellent control against the Pythium species. This demonstrates that Pseudomonas putida strain Psl (V13/001974) is a control agent for Pythium species plant disease-causing fungi.
EXAMPLE 16 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Bacillus amyloliquefaciens strain Bslb
[00261] This example describes the in-vitro analysis of the compatibility of the bacteria Pseudomonas putida strain Psl (VI 3/ 001974) with Bacillus amyloliquefaciens strain Bslb the active component of Triplex™ (Biostart NZ); a Botrytis and other plant fungal biocontrol product.
Methods
[00262] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00263] Compatibility Testing— Tests were carried out in 50 mL tubes into which had been added 49.5 mL water, 0.5 mL Triplex™ and 5 μL of Pseudomonas putida strain Psl (V13/001974) culture. Control tubes had either the Triplex™ and no Pseudomonas putida strain Psl ( 13/001974) culture or Pseudomonas putida strain Psl (V 13/001974) culture and no Triplex™. This mixture was incubated at room temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain Psl (V13/001974) and Bacillus amyloliquefaciens strain Bslb was determined by making serial dilutions and plating microbes on to Kings Medium B and PDA agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Results
[00264] There was the same number of Pseudomonas putida strain Psl (V13/001974) colonies in the presence or absence of Triplex™. Similarly, there was the same number of colonies of the Bacillus amykliquefaciens strain Bslb in the presence or absence of Pseudomonas
putida strain Psl (V13/001974) culture. This indicates that the Pseudomonas putid strain Psl (V13/001974) can be co-applied with Triplex™ and/or Bacillus microbes.
Discussion
[00265] Pseudomonas putida strain Psl (V13/001974) is compatible with Triplex™ and Bacillus amyloliquefaciens strain Bslb.
EXAMPLE 17 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Insecticides
[00266] This example describes the in-vitro analysis of the compatibility of the bacteria Pseudomonas putida strain Psl (VT3/001974) with several commercial formulations of insecticides.
Methods
[00267] Inoculum was prepared for Pseudomonas putida strain Psl (V13/001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00268] Compatibility Testing - Tests were carried out in 50 mL tubes into which had been added 49.5 mL water and 5 uL of ' Pseudomona putida strain Psl (V13/001974) culture and an appropriate amount of the insecticide (following commercial recommendations of the manufacturer; Table 3). Control tubes had Pseudomonas putida strain Psl (YT3/001974) culture and no insecticide. This mixture was incubated at room temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain Psl (V13/001974) was determined by making serial dilutions and plating microbes on to Kings Medium B agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Table 3 - Insecticides used in compatibility testing of Psl
Insecticide Tested Active Ingredient Formulation Use Rate
Talstar 100 EC bifenthrin EC 40 mL/100 L
Calypso thiacloprid SC 20 mL/100 L
Movento 240 SC spirotetramat SC 20 mL/100 L
Comic/Mimic 200 tebufenozide WP 8.6 g/100 L
Prodigy methoxyfenozide SC 25 mL/100 L
Proclaim emamectin benzoate WG 2 g/100 L
Key Pyrethrum pyrethrins EC 500 n L/100 L
Pyganic pyrethrins EC 500 niL/100 L
BioBit/Dipel bacillus thuringiensis Bt WP 50 g/100 L
Results
[00269] There was the same number of Pseudomonas putida strain Psl (V 13/001974) colonies in the presence of Talstar 100 EC, Calypso, Movento 240 SC, Comic/Mimic 200, Prodigy, Proclaim, Key Pyrethrum, Pyganic, and BioBit/Dipel. This indicates that the
Pseudomonas putida strain Psl (V13/001974) can be co-applied with Talstar 100 EC, Calypso, Movento 240 SC, Comic /Mimic 200, Prodigy, Proclaim, Key Pyrethrum, Pyganic, and BioBit/Dipel or similar products.
Discussion
[00270] Pseudomonas putida strain Psl (V13/001974) is compatible with Talstar 100 EC, Calypso, Movento 240 SC, Comic/Mimic 200, Prodigy, Proclaim, Key Pyrethrum, Pyganic, and BioBit/Dipel or products containing bifenthrin, thiacloprid, spirotetramat, tebufenozide, methoxyfenozide, emamectin benzoate, pyrethrins, and Bacillus thuringiensis Bt.
EXAMPLE 18 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Vegetable Oils, Mineral Oils and Spreader and Sticking Agents
[00271] This example describes the in-vitro analysis of the compatibility of the bacteria Pseudomonas putida strain Psl (V13/001974) with several commercial formulations of V egetable Oils, Mineral Oils and Spreader and Sticking Agents.
Methods
[00272] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00273] Compatibility Testing— Tests were carried out in 50 mL tubes into which had been added 49.5 mL water and 5 μϊ^ of Pseudomonas putida strain Psl (V13/001974) culture and an appropriate amount of the Vegetable Oils, Mineral Oils and Spreader and Sticking Agents (following commercial recommendations of the manufacturer; Table 4). Control tubes had Pseudomonas putida strain Psl (V 13/ 001974) culture and no Vegetable Oils, Mineral Oils and Spreader and Sticking Agents. This mixture was incubated at room
temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain Psl (V13/001974) was determined by making serial dilutions and plating microbes on to Kings Medium B agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Table 4— Carriers used in compatibility testing of Psl
Results
[00274] There was the same number of Pseudomonas putida strain Psl (VI 3/ 001974) colonies in the presence of EcoOil, Excel Oil, Excel Organic Oil, Duwett, Filmstar, Bond Xtra. This indicates that the Pseudomonas putida strain Psl (V13/001974) can be co-applied with canola oil sprays, or mineral oil (parrafinic oil) sprays and stickers or similar such products.
Discussion
[00275] Pseudomonas putida strain Psl (V13/001974) is compatible with EcoOil, Excel Oil, Excel Organic Oil, Duwett, Filmstar, Bond Xtra or products containing vegetable or mineral oil sprays and stickers or similar products.
EXAMPLE 19 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Glyphosate
[00276] This example describes the in-vitn analysis of the compatibility of the bacteria Pseudomonas putida strain Psl (V13/001974) with a commercial formulation of the herbicide glyphosate.
Methods
[00277] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile
techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00278] Compatibility Testing - Tests were carried out in 50 mL tubes into which had been added 49.5 mL water and 5 of Pseudomonas putida strain Psl (V13/001974) culture and an appropriate amount of glyphosate. Control tubes had Pseudomonas putida strain Psl (V13/001974) culture and no glyphosate. This mixture was incubated at room temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain Psl (V13/001974) was determined by making serial dilutions and plating microbes on to Kings Medium B agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Results
[00279] There was the same number of Pseudomonas putida strain Psl (V13/001974) colonies in the presence of glyphosate. This indicates that the Pseudomonas putida strain Psl (V13/001974) can be co-applied with glyphosate or similar such products.
Discussion
[00280] Pseudomonas putid strain Psl (V13/001974) is compatible with glyphosate or similar products.
EXAMPLE 20 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Foliar Fertilisers
[00281] This example describes the in-vittv analysis of the compatibility of the bacteria Pseudomonas putid strain Psl (V13/001974) with commercial formulations of foliar fertilisers such as blood and bone extracts and seaweed extracts.
Methods
[00282] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00283] Compatibility Testing - Tests were carried out in 50 mL tubes into which had been added 49.5 mL water and 5 μΐ^ of Pseudomonas putida strain Psl (V13/001974) culture and an appropriate amount of Nitrosol (500 mL/lOOL) or the seaweed Algal 600 or Acadian Seaweed products (2 kg/ 100L). Control tubes had Pseudomonas putida strain Psl (V13/001974) culture and no foliar fertiliser. This mixture was incubated at room temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain
Psl (V13/001974) was determined by making serial dilutions and plating microbes on to Kings Medium B agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Results
[00284] There was the same number of Pseudomonas putida strain Psl (V13/001974) colonies in the presence of Nitrosol or the Algal 600 or Acadian Seaweed. This indicates that the Pseudomonas putida strain Psl (V 13/001974) can be co- applied with foliar fertilisers or similar such products.
Discussion
[00285] Pseudomonas putida strain Psl (V13/001974) is compatible with foliar fertiliser, seaweed or similar products.
EXAMPLE 21 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Bird Repellant
[00286] This example describes the in-vitro analysis of the compatibility of the bacteria Pseudomonas putida strain Psl (V13/001974) with the Bird Repellant Mesurol 500 SC.
Methods
[00287] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a King Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00288] Compatibility Testing— Tests were carried out in 50 mL tubes into which had been added 49.5 mL water and 5 uL of Pseudomonas putida strain Psl (V13/001974) culture and an appropriate amount of Mesurol 500 SC (150 mL/lOOL). Control tubes had Pseudomonas putid strain Psl (V13/001974) culture and no foliar fertiliser. This mixture was incubated at room temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain Psl (V13/001974) was determined by making serial dilutions and plating microbes on to Kings Medium B agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Results
[00289] There was the same number of Pseudomonas putida strain Psl (V13/001974) colonies in the presence of Mesurol 500 SC. This indicates that the Pseudomonas putida strain Psl (VT3/001974) can be co-applied with methiocarb or similar such products. Discussion
[00290] Pseudomonas putida strain Psl (V13/ 001974) is compatible with the bird repellent Mesurol 500 SC or methiocarb or similar products.
EXAMPLE 22 - Compatibility of Pseudomonas putida strain Psl (V13/001974) with Biostart Products
[00291] This example describes the in-vitro analysis of the compatibility of the bacteria Pseudomonas putida strain Psl (V13/001974) with commercial Biostart (Auckland, New Zealand) product formulations including, Digester™ (a product mat activates breakdown of crop stubble and debris), Mycorrcin™ (a soil mycorrhizal fungi stimulant), Elicyon™ (a plant elicitor), and Foliacin™ (a plant elicitor).
Methods
[00292] Inoculum was prepared for Pseudomonas putida strain Psl (V13/ 001974) by inoculating a I'Cing Medium B liquid culture with a single isolated colony using sterile techniques. This culture was grown for 24 hours at 25 °C with shaking. This culture was then used in the subsequent compatibility studies.
[00293] Compatibility Testing— Tests were carried out in 50 mL tubes into which had been added 49.5 mL water and 5
of Pseudomonas putid strain Psl (V13/001974) culture and an appropriate amount of Digester™ (0.5 mL), Digester™ + glyphosate (0.5 mL and 0.5 mL), Mycorrcin™ (0.5 mL), Elicyon™ (0.5 mL) and Foliacin™ (0.5 mL). Control tubes had Pseudomonas putida strain Psl (V13/001974) culture and no foliar fertiliser. This mixture was incubated at room temperature with shaking for 6 h, after which the bacterial titre for Pseudomonas putida strain Psl (V13/001974) was determined by making serial dilutions and plating microbes on to Kings Medium B agar plates. The plates were incubated at 30 °C for 1-2 days and bacterial titres determined for each microbe.
Results
[00294] There was the same number of Pseudomonas putida strain Psl (V 13/001974) colonies in the presence of Digester™, Digester™ + glyphosate, Mycorrcin™, Elicyon™ and Foliacin™. This indicates that the Pseudomonas putida strain Psl (V13/001974) can be co-applied with Digester™, Digester™ + glyphosate, Mycorrcin™, Elicyon™ and
Foliacin™ or similar such products.
Discussion
[00295] Pseudomonas putida strain Psl (V13/001974) is compatible with established agricultural products such as Digester™, Digester™ + glyphosate, Mycorrcin™, Elicyon™, Foliacin™ or similar products.
EXAMPLE 23 - Use of Pseudomonas putida strain Psl (V13/001974) as a biocontrol agent of Psa in a green kiwifmit orchard
[00296] This example describes a field trial using the bacteria Pseudomonas putida strain Psl (V 13/001974) as a biocontrol agent of the kiwifruit disease Pseudomonas syringae pv actinidiae (Psa) in a commercial green kiwifruit orchard.
Methods
[00297] A fermentation culture of Pseudomonas putida strain Psl (VI 3/001974) was prepared in a suitable media and then used in the field trial.
[00298] Field trial - A commercial kiwifruit orchard of green kiwifruit (Actinidia delicios ) in a highly Psa-infectious area of Te Puke was the trial site. Three co-applications of Pseudomonas putida ? strain Psl (V13/001974) culture (50 mL/ha) and Foliacin™ (0.5 L/ha) were made during the flowering period in response to rain events; 5 October 2012, 11 October 2012 and 20 October 2012. On 28 October vines were scored for disease severity. This period coincided with a highly infectious period of Psa infection in kiwifruit orchards.
Results
[00299] The use of Pseudomonas putida strain Psl (V 13/001974) on kiwifruit vines reduced disease severity to 20 % compared to 30 % in untreated vines.
Discussion
[00300] The Pseudomonas putida strain Psl (VT3/001974) can be used to reduce disease severity of Psa infections in kiwifruit orchards.
[00301] Accordingly, the control agents, compositions, and methods of the present invention enable the effective control of a serious plant pathogen, and provide an alternative to chemical control regimens.
EXAMPLE 24 - Use of Pseudomonas putida strain Psl (V13/001974) as a biocontrol agent of Psa in a gold kiwifruit orchard
[00302] This example describes a field trial using the bacteria Pseudomonas putida strain Psl (V13/001974) as a biocontrol agent of the kiwifruit disease Pseudomonas syringae pv actinidiae (Psa) in a commercial gold kiwifruit orchard.
Methods
[00303] A fermentation culture of Pseudomonas putida strain Psl (V13/001974) was prepared in a suitable media and then used in the field trial.
[00304] Field trial— A commercial kiwifruit orchard of gold kiwifruit (Actinidia chinensis), in a highly Psa-infectious area of Te Puke, was the trial site. The plants used in the trial had been previously infected with Psa and in November and December 2011 all the vines were removed. In July 2012 the rootstocks were re-grafted to a new gold kiwifruit variety. The trial consisted of two blocks and was started on 24 September 2012. Block 1 (999 vines) received 10 applications of Pseudomonas putida strain Psl (νΤ3/001974) culture (50 mL/ha), whereas Block 2 (1361 vines) received a "current commercial best practice" treatment consisting of 10 applications total of either Kocide Opti (copper hydroxide; 5 applications), Keystrepto (streptomycin; 2 applications), Nordox (copper oxide; 1 application) or Actiguard (salicylic acid analogue; 1 application). On 15 January 2013 vines in Block and Block 2 were scored for disease severity.
Results
[00305] In Block 1 (Psl Programme) 4.6 % of the vines showed some sign of Psa infection compared to 4.8 % of the vines in Block 2 (Commercial Programme). The use of Pseudomonas putida strain Psl (V'13/001974) on kiwifruit vines had a similar effect in controlling Psa as the commercial programme consisting of copper products.
Discussion
[00306] The Pseudomonas putida strain Psl (V13/001974) can be used to control Psa infections in kiwifruit orchards with a similar efficiency to a spray programme consisting of copper products, elicitors and antibiotic treatments.
[00307] Accordingly, the bacteria, compositions, and methods of the present invention enable the effective control of a serious plant pathogen, and provide an alternative to chemical control regimens.
INDUSTRIAL APPLICATION
[00308] As will be evident from the above description, the present invention provides a strain of anti-phytop thogenic bacteria, together with the compositions comprising said bacteria, which are useful for the control of microbes and phytopathogens. The use of such bacteria and compositions comprising or derived therefrom in the control of microbes and phytopathogens, and methods to control microbes and phytopathogens, including phytopathogenic microbial phytopathogens, are also provided.