WO2013038643A1 - Chemical-conversion-treatment-plated steel sheet and method for manufacturing same - Google Patents
Chemical-conversion-treatment-plated steel sheet and method for manufacturing same Download PDFInfo
- Publication number
- WO2013038643A1 WO2013038643A1 PCT/JP2012/005722 JP2012005722W WO2013038643A1 WO 2013038643 A1 WO2013038643 A1 WO 2013038643A1 JP 2012005722 W JP2012005722 W JP 2012005722W WO 2013038643 A1 WO2013038643 A1 WO 2013038643A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- chemical conversion
- plated steel
- conversion treatment
- film
- steel sheet
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/05—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions
- C23C22/06—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6
- C23C22/34—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 containing fluorides or complex fluorides
- C23C22/36—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 containing fluorides or complex fluorides containing also phosphates
- C23C22/361—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals using aqueous solutions using aqueous acidic solutions with pH less than 6 containing fluorides or complex fluorides containing also phosphates containing titanium, zirconium or hafnium compounds
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C28/00—Coating for obtaining at least two superposed coatings either by methods not provided for in a single one of groups C23C2/00 - C23C26/00 or by combinations of methods provided for in subclasses C23C and C25C or C25D
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2222/00—Aspects relating to chemical surface treatment of metallic material by reaction of the surface with a reactive medium
- C23C2222/20—Use of solutions containing silanes
Definitions
- the present invention relates to a chemically treated Zn-based plated steel sheet having excellent weather resistance, water resistance, blackening resistance, film adhesion and workability, and a method for producing the same.
- the plated steel sheet is sometimes formed with a chemical conversion treatment film containing an organic resin on its surface in order to prevent galling during forming (for example, see Patent Documents 1 and 2).
- Patent Documents 1 and 2 describe that a chemical conversion treatment film containing an organic resin such as a urethane resin is formed on the surface of a zinc-based plated steel sheet.
- a chemical conversion treatment film containing an organic resin such as a urethane resin is formed on the surface of a zinc-based plated steel sheet.
- a fluororesin having excellent weather resistance may be used as an organic resin constituting the chemical conversion film.
- an organic solvent type fluororesin composition is used in many cases.
- organic solvent-based fluororesin compositions have problems such as fire hazard and harmfulness and air pollution.
- Patent Document 4 a water-based fluororesin composition capable of forming a film even at a low temperature by introducing a curable site (organic functional group) has been proposed (for example, Patent Document 4). reference).
- a film cured by reacting with an organic functional group the weather resistance deteriorates preferentially from the cured portion, so that the film becomes porous and water resistance decreases.
- the base treatment with an epoxy resin or a urethane resin is performed for improving the adhesion, the organic resin is preferentially deteriorated in weather resistance, and the film adhesion is rapidly lowered. .
- galling resistance, corrosion resistance, discoloration resistance, and the like can be improved by forming a chemical conversion film containing an organic resin on the surface of a plated steel sheet.
- the conventional chemical conversion treatment plated steel sheet formed with the chemical conversion treatment film containing the organic resin may have insufficient weather resistance when used as an exterior building material.
- many organic resins such as urethane resins are deteriorated by ultraviolet rays
- the chemical conversion film covering the surface of the plated steel sheet is lost over time. There is a risk that. If the chemical conversion treatment film is lost in this manner, discoloration, rust, etc. may occur and the appearance may be impaired, which is not preferable as an exterior building material.
- a conventional chemical conversion-treated steel sheet on which a chemical conversion treatment film containing an organic resin is formed may have insufficient weather resistance.
- the use of water-based fluororesin as the organic resin can improve the weather resistance (ultraviolet light resistance) of the chemically treated steel sheet, but on the other hand, the film forming property, water resistance and film adhesion are reduced. Therefore, it is impossible to achieve both weather resistance, water resistance, blackening resistance and film adhesion.
- An object of the present invention is a chemical conversion treatment Zn-plated steel sheet having a chemical conversion treatment film containing an organic resin, which is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion and workability. It is to provide a steel plate.
- the present inventor uses a high molecular weight fluorine-containing resin into which a hydrophilic functional group is introduced as an organic resin, and crosslinks these fluorine-containing resins with a group 4A metal compound, thereby enabling the weather resistance and water resistance of the chemical conversion coating.
- the present invention has been completed through further investigations, which have been found to improve the property, blackening resistance and film adhesion.
- the first of the present invention relates to the following chemical conversion treatment Zn-plated steel sheet.
- An Al-containing Zn-based alloy plated steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al, and a film thickness of 0.5 to 0.5 formed on the surface of the Al-containing Zn-based alloy plated steel sheet A chemical conversion treatment Zn-plated steel sheet having a chemical treatment film of 10 ⁇ m; the chemical conversion treatment film having a hydrophilic functional group of 0.05 to 5 selected from the group consisting of a carboxyl group, a sulfonic acid group and salts thereof
- a fluorine-containing resin containing 5% by mass and 7 to 20% by mass of F atoms, 0.1 to 5% by mass of a 4A group metal compound in terms of metal with respect to the fluorine-containing resin A chemical conversion-treated Zn-based plated steel sheet, comprising 10 ⁇ m resin particles; and an area occupancy ratio of the resin particles on the surface of the chemical conversion coating is
- the chemical conversion coating further contains a polyethylene resin; the polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating in part of the surface of the chemical conversion coating;
- the chemical conversion film further contains a phosphate; the amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P.
- the chemical conversion treatment film further contains a silane coupling agent; the amount of the silane coupling agent with respect to the fluorine-containing resin is in the range of 0.5 to 5% by mass.
- An underlayer chemical conversion coating formed between the Al-containing Zn-based alloy-plated steel sheet and the chemical conversion coating and containing a valve metal oxide or hydroxide and a valve metal fluoride.
- 2nd of this invention is related with the manufacturing method of the following chemical conversion treatment Zn-plated steel plate.
- a fluorine-containing resin containing 0.05 to 5% by mass of a group and 7 to 20% by mass of F atoms and having a number average molecular weight in the range of 1,000 to 2,000,000, a group 4A metal oxyacid salt, fluoride, Any one of hydroxide, organic acid salt, carbonate or peroxide salt, and resin particles having an average particle size of 0.1 to 10 ⁇ m; an oxyacid salt of the group 4A metal with respect to the fluorine-containing resin; Fluoride, hydroxide, existence The amount of the acid salt, carbonate or peroxide is in the range of 0.1 to 5% by mass in terms of metal; the amount of the resin particles relative to the solid content in the chemical conversion solution is 0.5 to The manufacturing method of the chemical conversion treatment Zn-plated steel plate which exists in the range of 20 mass%.
- the chemical conversion treatment liquid further contains a phosphate; the amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P, [9] to The manufacturing method of the chemical conversion treatment Zn-plated steel plate as described in any one of [12].
- the chemical conversion treatment liquid further contains a silane coupling agent; the amount of the silane coupling agent with respect to the fluorine-containing resin is in the range of 0.5 to 5% by mass. 13] The manufacturing method of the chemical conversion treatment Zn-plated steel plate as described in any one of [13].
- the method further includes the step of applying a base chemical conversion treatment solution to the surface of the Al-containing Zn-based alloy plated steel sheet and drying to form a base chemical conversion treatment film;
- the present invention it is possible to provide a chemically treated Zn-based plated steel sheet that is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion, and workability. Since the chemical conversion treatment Zn-plated steel sheet of the present invention is excellent in weather resistance, water resistance, corrosion resistance, discoloration resistance and workability, it is useful as a plated steel sheet for exterior building materials, for example.
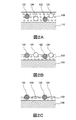
- 1A to 1D are SEM images of a chemical conversion coating containing polyethylene resin particles or polyethylene-fluorine resin particles heated at a predetermined temperature.
- 2A to 2C are schematic cross-sectional views showing the formation process of the chemical conversion film.
- 3A to 3C are SEM images showing the formation process of the chemical conversion film.
- 2 is a fluorescent X-ray spectrum of polyethylene-fluorine resin particles in a chemical conversion coating. It is a graph which shows the relationship between the quantity of the 4A group metal in a fluororesin membrane
- Chemical conversion treatment Zn-plated steel sheet The chemical conversion treatment Zn-plated steel sheet of the present invention has an Al-containing Zn-based alloy-plated steel sheet (chemical conversion-treated original plate) and a chemical conversion-treated film formed on the surface of the Al-containing Zn-based alloy-plated steel sheet. .
- the chemical conversion treatment Zn-based plated steel sheet of the present invention comprises a high molecular weight fluorine-containing resin, a group 4A metal compound and a resin particle (lubricant) in which a chemical conversion treatment film has introduced a hydrophilic functional group (such as a carboxyl group or a sulfonic acid group). Inclusion is a feature.
- Al-containing Zn-based alloy plated steel plate having excellent corrosion resistance and design properties is used.
- Al-containing Zn-based alloy-plated steel sheet means a steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al.
- Al-containing Zn alloy-plated steel sheets examples include hot-dip Al-Zn-plated steel sheets (hot Zn-0.1% Al plating, hot Zn-55% Al plating, hot Zn-6% Al-3% Mg plating, hot Zn-11% Al-3% Mg-0.2% Si, hot-dip Zn-5% Al-0.75% Mg), alloyed Zn-plated steel sheet (alloyed after hot-dip 0.1% Al-Zn plating) Alloyed molten Al—Zn plating) and the like.
- hot-dip Al-Zn-plated steel sheets hot Zn-0.1% Al plating, hot Zn-55% Al plating, hot Zn-6% Al-3% Mg plating, hot Zn-11% Al-3% Mg-0.2% Si, hot-dip Zn-5% Al-0.75% Mg
- alloyed Zn-plated steel sheet alloyed Zn-plated steel sheet (alloyed after hot-dip 0.1% Al-Zn plating) Alloyed molten Al—Zn plating) and the like
- the base steel of the Al-containing Zn-based alloy plated steel sheet low carbon steel, medium carbon steel, high carbon steel, alloy steel, or the like is used.
- steel sheets for deep drawing such as low carbon Ti-added steel and low carbon Nb-added steel are preferred as the base steel.
- an Al-containing Zn-based alloy-plated steel sheet used as a chemical conversion treatment original plate has a base chemical conversion treatment film containing a valve metal oxide or hydroxide and a valve metal fluoride. is there.
- the base chemical conversion film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet
- the chemical conversion film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet via the base chemical conversion film.
- substrate chemical conversion treatment film containing the oxide or hydroxide of valve metal acts as a resistor with respect to the movement of an electron. Therefore, the reduction reaction of dissolved oxygen contained in the moisture in the atmosphere is suppressed, and the oxidation reaction of the paired Al-containing Zn-based alloy plated steel sheet is also suppressed. As a result, elution (corrosion) of metal components from the Al-containing Zn-based alloy-plated steel sheet serving as the substrate is suppressed.
- the tetravalent compound of IV group A element, such as Ti, Zr, and Hf is a stable compound, and forms the outstanding highly insulating film.
- a film defect is inevitably generated in a chemical conversion treatment film at the time of chemical conversion treatment or molding. Since the base material is exposed at the film defect portion, the corrosion inhibiting action cannot be expected even if chemical conversion treatment is performed.
- the above-mentioned base chemical conversion treatment film also contains a fluoride of valve metal, it has a self-repairing action. That is, the fluoride of the valve metal dissolves in moisture in the atmosphere and then re-deposits as a hardly soluble oxide or hydroxide on the surface of the base steel exposed from the film defect portion. As a result, since the film defect portion is filled, a self-repairing action is exhibited.
- the chemical conversion-treated Zn-based alloy-plated steel sheet of the present invention is manufactured by subjecting an Al-containing Zn-based alloy-plated steel sheet to chemical conversion treatment, but the Zn-based alloy-plated steel sheet before chemical conversion treatment has insufficient corrosion resistance. Therefore, corrosion may occur during storage or transportation of the Zn-based alloy-plated steel sheet before chemical conversion treatment or during forming. Therefore, by forming a base chemical conversion coating on the Zn-based alloy plated steel sheet before the chemical conversion treatment, it is possible to reliably prevent the occurrence of corrosion in the Zn-based alloy plated steel sheet before the chemical conversion treatment.
- the Al-containing Zn-based alloy-plated steel sheet on which the base chemical conversion treatment film is formed is stored, transported or formed (including welding), a part of the base chemical conversion treatment film may be peeled off, missing, or missing. Thereby, the surface of the Al-containing Zn-based alloy-plated steel sheet is exposed, and the chemical conversion treatment film containing the fluorine-containing resin and the 4A group metal compound is in direct contact with the exposed surface.
- the base chemical conversion treatment film exhibits an effect of improving the adhesion between the Al-containing Zn-based alloy-plated steel sheet and the chemical conversion treatment film. For this reason, the chemical conversion treatment film formed at the place where the base chemical conversion treatment film is peeled is generally considered to have a reduced film adhesion.
- Al is eluted from the plating layer in direct contact with the chemical conversion coating.
- the corrosion resistance and film adhesion of the chemical conversion film are improved by Al eluted in the chemical conversion film.
- the adhesiveness of a chemical conversion treatment film and an Al containing Zn type alloy plating steel plate is high, and the corrosion resistance of a chemical conversion treatment film increases. The mechanism by which the presence of Al in the chemical conversion film improves the corrosion resistance and film adhesion of the chemical conversion film will be described later.
- the base chemical conversion treatment film can be formed by drying a coating film of the base chemical conversion treatment liquid formed on the surface of the Al-containing Zn-based alloy-plated steel sheet as the base material.
- the base chemical conversion treatment solution contains valve metal salt, fluoride ions, and water as a solvent. By drying the coating film of the base chemical conversion treatment solution, the valve metal salt becomes an oxide or hydroxide or a fluoride of valve metal contained in the base chemical conversion coating film.
- valve metals examples include Ti, Zr, Hf, V, Nb, Ta, Mo, and W.
- the valve metal salt added to the base chemical conversion treatment liquid may be a valve metal halide or oxyacid salt. If the added valve metal salt is fluoride, it also acts as a fluoride ion source.
- titanium salts examples include K n TiF 6 (K: alkali metal or alkaline earth metal, n: 1 or 2), K 2 [TiO (COO) 2 ], (NH 4 ) 2 TiF 6 , TiCl 4 , TiOSO 4 , Ti (SO 4 ) 2 , Ti (OH) 4 and the like are included.
- the fluoride ion source contained in the base chemical conversion treatment liquid may be a valve metal salt containing a fluorine atom or a soluble fluoride (for example, (NH 4 ) F).
- an organic acid having a chelating action is added to the base chemical conversion treatment solution in order to stabilize the valve metal salt.
- the organic acid can chelate metal ions and stabilize the chemical conversion solution. Therefore, the addition amount of the organic acid is set so that the molar ratio of organic acid / metal ion is 0.02 or more.
- the organic acid include tartaric acid, tannic acid, citric acid, succinic acid, malonic acid, lactic acid, acetic acid, ascorbic acid and the like.
- oxycarboxylic acids such as tartaric acid and polyhydric phenols such as tannic acid stabilize the base chemical conversion solution and also complement the fluoride's self-healing action, improving adhesion. It is valid.
- Various metal orthophosphates or polyphosphates may be added to the base chemical conversion treatment solution. This is because the base chemical conversion treatment film to be formed contains a soluble or hardly soluble metal phosphate or composite phosphate.
- Soluble metal phosphate or composite phosphate elutes from the base chemical conversion coating to the film defects and reacts with the plating components (Zn, Al, etc.) of the Al-containing Zn-based alloy-plated steel sheet as the substrate. Insoluble phosphate is precipitated. In this way, the self-healing action of titanium fluoride is complemented. Further, since the atmosphere is slightly acidified when the soluble phosphate is dissociated, the hydrolysis of titanium fluoride, and hence the generation of hardly soluble titanium oxide or hydroxide, is promoted.
- the metal of the soluble phosphate or the composite phosphate can be an alkali metal, an alkaline earth metal, Mn, or the like.
- the soluble phosphate or the composite phosphate may be added to the base chemical conversion treatment liquid in the form of various metal phosphates, or may be formed by combining various metal salts with phosphoric acid, polyphosphoric acid or phosphate. It may be added to the treatment liquid.
- poorly soluble metal phosphates or composite phosphates are dispersed in the base chemical conversion treatment film to eliminate film defects and improve the film strength.
- the metal of the poorly soluble phosphate or the composite phosphate may be Al, Ti, Zr, Hf, Zn or the like.
- the hardly soluble phosphate or the composite phosphate may be added to the chemical conversion treatment liquid in the form of various metal phosphates, or a chemical conversion treatment by combining various metal salts with phosphoric acid, polyphosphoric acid or phosphate. It may be added to the liquid.
- organic waxes such as fluorine, polyethylene, and styrene
- inorganic lubricants such as silica, molybdenum disulfide, and talc can be added.
- the lubricity of the base chemical conversion coating can be improved. It is considered that the low melting point organic wax bleeds on the film surface and develops lubricity when the coating film of the base chemical conversion treatment liquid is dried.
- high-melting organic waxes and inorganic lubricants are present in a dispersed state in the film, but it is considered that the outermost surface layer of the film is exposed to the surface of the film in an island-like distribution to exhibit lubricity.
- the concentrations of O and F contained in the base chemical conversion film are measured.
- the concentration ratio F / O (atomic ratio) of these elements is preferably 1/100 or more. It is for suppressing the corrosion of the obtained chemical conversion treatment steel plate.
- the element concentration ratio F / O (atomic ratio) is 1/100 or more, the occurrence of corrosion starting from a film defect portion is significantly reduced. This is presumably because a sufficient amount of titanium fluoride is contained in the base chemical conversion treatment film and exhibits a self-repairing action.
- the chemical conversion treatment film is formed on the surface of the above-mentioned Al-containing Zn-based alloy plated steel sheet (chemical conversion treatment original plate).
- the surface of the chemical conversion treatment original plate may be subjected to a base chemical conversion treatment such as forming a coating as a base, but the base chemical conversion treatment may not be performed.
- a chemical conversion treatment film is directly formed on the surface of the chemical conversion treatment original plate. This chemical conversion treatment film improves the weather resistance, blackening resistance, workability (lubricity), and the like of the Al-containing Zn-based alloy plated steel sheet.
- the object of the present invention is to improve all of the weather resistance, water resistance, blackening resistance, film adhesion and workability of the chemically treated Zn-based plated steel sheet.
- a fluorine-containing resin may be used as the organic resin. Fluorine-containing resins are roughly classified into solvent-based fluorine-containing resins and water-based fluorine-containing resins. When a chemical conversion film is formed using a solvent-based fluorine-containing resin, recovery of the volatilized solvent becomes a problem, but when a water-based fluorine-containing resin is used, such a problem does not occur.
- the present inventor uses a water-based fluorine-containing resin that is easy to handle to form a chemically treated Zn-based plated steel sheet that is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion, and workability. Tried.
- the present inventor examined not only suppressing the decrease in water resistance of the chemical conversion coating but also improving the water resistance. As a result of studying from various viewpoints, it was found that the water resistance of the chemical conversion coating can be remarkably improved by increasing the molecular weight of the aqueous fluorine-containing resin and crosslinking the aqueous fluorine resin with a group 4A metal compound. It was.
- the present inventor has studied to improve the workability (lubricity) of the chemical conversion treatment Zn-plated steel sheet. As a result of studying various means, the present inventor disperses resin particles having an average particle diameter of 0.1 to 10 ⁇ m in the chemical conversion film, thereby improving weather resistance, water resistance, blackening resistance, and film adhesion. In addition to the properties, it has been found that a chemical conversion film excellent in workability (lubricity) can be formed.
- 1) weather resistance is improved by blending a fluorine-containing resin (preferably a fluorine-containing olefin resin).
- a fluorine-containing resin preferably a fluorine-containing olefin resin
- 2) the use of a fluorine-containing resin into which a hydrophilic functional group has been introduced reduces the use of an emulsifier during emulsion production
- 3) the molecular weight of the fluorine-containing resin is increased
- 4) the fluorine-containing resin is 4A.
- By cross-linking with a group metal compound weather resistance (ultraviolet light resistance) and water resistance are improved.
- Workability lubricity is improved by dispersing resin particles having an average particle diameter of 0.1 to 10 ⁇ m.
- the chemical conversion film contains a fluorine-containing resin, more specifically, a fluorine-containing olefin resin as a main component.
- the amount of the fluorine-containing resin contained as a main component in the chemical conversion film is preferably in the range of 70 to 99% by mass.
- the weather resistance (ultraviolet resistance) of the chemical conversion coating can be improved.
- the fluorine-containing resin is preferably a water-based fluorine-containing resin that is easier to handle than the organic solvent-based fluorine resin.
- “Aqueous fluorine-containing resin” refers to a fluorine-containing resin having a hydrophilic functional group. Preferred examples of the hydrophilic functional group include a carboxyl group, a sulfonic acid group, and salts thereof. Examples of the salt of the carboxyl group or sulfonic acid group include ammonium salt, amine salt, alkali metal salt and the like.
- a preferred aqueous fluorine-containing resin (preferably a fluorine-containing olefin resin) has a hydrophilic functional group of 0.05 to 5% by mass.
- a fluorine-containing resin having 0.05 to 5% by mass of a hydrophilic functional group can be made into an aqueous emulsion with little use of an emulsifier.
- the chemical conversion film containing almost no emulsifier can be a chemical conversion film excellent in water resistance.
- the content of the hydrophilic functional group in the aqueous fluorine-containing resin may be obtained by dividing the total molar mass of the hydrophilic functional group contained in the aqueous fluorine-containing resin by the number average molecular weight of the aqueous fluorine-containing resin. Since the molar mass of the carboxyl group is 45 and the molar mass of the sulfonic acid group is 81, the number of each of the carboxyl group and sulfonic acid group contained in the aqueous fluorine-containing resin is determined, and each is multiplied by the molar mass. The total molar mass of the hydrophilic functional group contained in the aqueous fluorine-containing resin is obtained. The number average molecular weight of the water-based fluorine-containing resin is measured by GPC.
- the carboxyl group in the water-based fluorine-containing resin forms a hydrogen bond with the plating layer surface and contributes to improving the adhesion between the chemical conversion film and the plating layer surface.
- H + is difficult to dissociate, The cross-linking reaction is unlikely to occur.
- the sulfonic acid group in the water-based fluorine-containing resin tends to dissociate H +, but if it remains unreacted in the film without cross-linking reaction with the group 4A metal compound, the water molecule adsorbing action is strong, so There is a risk that the water resistance will be significantly reduced.
- the water-based fluorine-containing resin includes both a carboxyl group and a sulfonic acid group in order to make use of each feature.
- the ratio of carboxyl group to sulfonic acid group is preferably in the range of 5 to 60 in terms of the molar ratio of carboxyl group / sulfonic acid group.
- the number average molecular weight of the water-based fluorine-containing resin (preferably fluorine-containing olefin resin) contained in the chemical conversion film is preferably 1000 or more, more preferably 10,000 or more, and particularly preferably 200,000 or more.
- the molecular weight of the water-based fluorine-containing resin contained in the chemical conversion coating is too small, the water permeability and water resistance of the chemical conversion coating cannot be sufficiently improved. In such a case, moisture or corrosive gas easily penetrates the chemical conversion coating and reaches the plated steel sheet, so that the plated steel sheet may be easily corroded.
- an aqueous fluorine-containing resin with a low molecular weight is used, radicals generated by the action of light energy or the like are likely to act on the end of the polymer chain, so the aqueous fluorine-containing resin is easily hydrolyzed by a synergistic action such as water. There is a risk of being.
- the molecular weight of the aqueous fluorine-containing resin contained in the chemical conversion coating may be increased to some extent, or a crosslinked structure may be formed between the aqueous fluorine-containing resins.
- the intermolecular force increases and the cohesive strength of the chemical conversion coating increases, so that the water resistance is improved.
- hydrolysis is less likely to occur.
- the number average molecular weight of the water-based fluorine-containing resin contained in the chemical conversion coating is preferably 2 million or less.
- the number average molecular weight exceeds 2 million, there is a possibility that a problem may occur in the stability of the treatment liquid such as gelation.
- the content of F atoms in the aqueous fluorine-containing resin contained in the chemical conversion coating is preferably within the range of 7 to 20% by mass.
- content of F atom is less than 7 mass%, the weather resistance of a chemical conversion treatment film cannot fully be improved.
- the content of F atoms is more than 20% by mass, it is difficult to form a paint and the adhesion and drying properties may be reduced.
- the content of F atoms in the aqueous fluorine-containing resin can be measured by using a fluorescent X-ray analyzer.
- water-based fluorine-containing resin examples include a copolymer of a fluoroolefin and a hydrophilic functional group-containing monomer.
- the hydrophilic functional group-containing monomer is a carboxyl group-containing monomer or a sulfonic acid group-containing monomer.
- fluoroolefins examples include tetrafluoroethylene, trifluoroethylene, chlorotrifluoroethylene, hexafluoropropylene, vinyl fluoride, vinylidene fluoride, pentafluoropropylene, 2,2,3,3-tetrafluoropropylene, 3, 3,3-trifluoropropylene, bromotrifluoroethylene, 1-chloro-1,2-difluoroethylene, 1,1-dichloro-2,2-difluoroethylene and the like are included. These fluoroolefins may be used alone or in combination of two or more.
- perfluoroolefins such as tetrafluoroethylene and hexafluoropropylene, and vinylidene fluoride are preferable.
- Fluoroolefins containing chlorine such as chlorotrifluoroethylene are not preferred because corrosion due to chlorine ions may occur.
- carboxyl group-containing monomer examples include unsaturated carboxylic acids represented by the following formula (1) and unsaturated carboxylic acids such as esters or acid anhydrides thereof.
- R 1 , R 2 and R 3 are the same or different, and each is a hydrogen atom, an alkyl group, a carboxyl group or an ester group.
- N is in the range of 0-20.
- Examples of the unsaturated carboxylic acid represented by the above formula (1) include acrylic acid, methacrylic acid, vinyl acetic acid, crotonic acid, cinnamic acid, itaconic acid, itaconic acid monoester, maleic acid, maleic acid monoester, fumaric acid , Fumaric acid monoester, 5-hexenoic acid, 5-heptenoic acid, 6-heptenoic acid, 7-octenoic acid, 8-nonenoic acid, 9-decenoic acid, 10-undecylene acid, 11-dodecylene acid, 17-octadecylenic acid Oleic acid and the like.
- carboxyl group-containing monomer includes a carboxyl group-containing vinyl ether monomer represented by the following formula (2). (Wherein R 4 and R 5 are the same or different, and each is a saturated or unsaturated linear or cyclic alkyl group. N is 0 or 1. m is 0 or 1.)
- Examples of the carboxyl group-containing vinyl ether monomer represented by the above formula (2) include 3- (2-allyloxyethoxycarbonyl) propionic acid, 3- (2-allyloxybutoxycarbonyl) propionic acid, 3- (2-vinylidene). Roxyethoxycarbonyl) propionic acid, 3- (2-vinyloxybutoxycarbonyl) propionic acid and the like.
- sulfonic acid group-containing monomers examples include vinyl sulfonic acid, allyl sulfonic acid, methallyl sulfonic acid, styrene sulfonic acid, 2-acrylamido-2-methylpropane sulfonic acid, 2-methacryloyloxyethane sulfonic acid, and 3-methacryloyloxy.
- Propanesulfonic acid 4-methacryloyloxybutanesulfonic acid, 3-methacryloyloxy-2-hydroxypropanesulfonic acid, 3-acryloyloxypropanesulfonic acid, allyloxybenzenesulfonic acid, methallyloxybenzenesulfonic acid, isoprenesulfonic acid, 3-allyloxy-2-hydroxypropane sulfonic acid and the like are included.
- the copolymer of the fluoroolefin and the hydrophilic functional group-containing monomer may be copolymerized with another copolymerizable monomer as necessary.
- examples of other copolymerizable monomers include carboxylic acid vinyl esters, alkyl vinyl ethers, non-fluorinated olefins, and the like.
- Carboxylic acid vinyl esters can improve compatibility and gloss, and increase the glass transition temperature.
- vinyl carboxylates include vinyl acetate, vinyl propionate, vinyl butyrate, vinyl isobutyrate, vinyl pivalate, vinyl caproate, vinyl versatate, vinyl laurate, vinyl stearate, vinyl cyclohexylcarboxylate, benzoate Vinyl acid, para-t-butyl vinyl benzoate and the like are included.
- Alkyl vinyl ethers can improve gloss and flexibility.
- alkyl vinyl ethers include methyl vinyl ether, ethyl vinyl ether, butyl vinyl ether and the like.
- Non-fluorinated olefins can improve flexibility.
- Examples of non-fluorinated olefins include ethylene, propylene, n-butene, isobutene and the like.
- a fluoroolefin copolymer having a hydrophilic functional group can be obtained by copolymerizing the above monomers by a known polymerization method. At this time, by adjusting the amount of the fluoroolefin in the raw material monomer composition so that the fluoroolefin copolymer has a hydrophilic functional group of 0.05 to 5% by mass, the fluoroolefin copolymer can be used with almost no emulsifier. An aqueous emulsion of the polymer can be produced. The chemical conversion film formed using an emulsion of a fluoroolefin copolymer containing almost no emulsifier (1% by mass or less) contains almost no emulsifier.
- a chemical conversion treatment film containing almost no emulsifier can be easily formed.
- the chemical conversion film thus formed exhibits excellent water resistance with little deterioration in water resistance due to the residual emulsifier.
- the chemical conversion film contains a group 4A metal compound.
- the 4A group metal compound easily reacts with a functional group such as a carboxyl group or a sulfonic acid group in the aqueous fluorine-containing resin, and accelerates the curing or crosslinking reaction of the aqueous fluorine-containing resin. Therefore, the water resistance of the chemical conversion film can be improved even by low-temperature drying.
- a melamine resin or an isocyanate resin is used for crosslinking of the fluorine-containing resin, there is a problem that the weather resistance is easily deteriorated.
- weather resistance deterioration is immediately caused by oxidation and hydrolysis of ester bonds and form ether bonds.
- weathering deterioration also proceeds when the crosslinked structure is cut by an acidic substance such as sulfate ion or nitrate ion contained in acid rain.
- the urethane bond formed at the cross-linked portion is weaker than the F bond, so that the cross-linked structure is preferentially cut, and the weather resistance deteriorates.
- the group 4A metal compound also improves film adhesion, water resistance and blackening resistance. That is, the strong Al oxide present on the surface of the Al-containing Zn-based alloy-plated steel sheet decreases the adhesion of the chemical conversion coating, but this Al oxide can be obtained by including a group 4A metal compound in the chemical conversion coating. It is possible to suppress a decrease in film adhesion due to.
- the group 4A metal compound also serves as a supply source of group 4A metal ions that react with Al ions eluted by the etching reaction. The reaction product is concentrated at the interface between the plating layer and the chemical conversion film to improve the initial corrosion resistance and blackening resistance. Examples of Group 4A metals include Ti, Zr, Hf, and the like.
- the content of the group 4A metal compound in the chemical conversion film is preferably in the range of 0.1 to 5% by mass in terms of metal with respect to the fluorine-containing resin.
- the content is less than 0.1% by mass in terms of metal, the adverse effect due to the concentration of Al oxide cannot be sufficiently suppressed, and the aqueous fluorine-containing resin fat cannot be sufficiently crosslinked. As a result, the water resistance of the chemical conversion coating cannot be sufficiently improved.
- the content is more than 5% by mass in terms of metal, the chemical conversion film becomes porous, and the workability and weather resistance may be reduced.
- the metal equivalent amount of the group 4A metal compound in the chemical conversion coating can be measured by using a fluorescent X-ray analyzer.
- Al eluted from the plating layer is present in the chemical conversion film.
- This Al contributes to improvement of corrosion resistance. It is presumed that the corrosion resistance is improved by the presence of Al due to the following mechanism. That is, 1) Since the chemical conversion treatment solution is weakly alkaline, when the chemical conversion treatment solution is applied, Al oxide and metal Al contained in the plating layer are selectively eluted into the chemical conversion treatment solution (Zn is almost eluted). do not do). 2) In the pH range of the chemical conversion treatment liquid, Al is Al (OH) 4 - is dissolved in the chemical conversion treatment liquid state. 3) When forming the chemical conversion film by drying the chemical conversion liquid, Al in the chemical conversion liquid is taken into the chemical conversion film by dehydration condensation or the like. 4) As a result, the insulating properties and density of the chemical conversion coating are improved, and the corrosion resistance is improved.
- the chemical conversion film contains resin particles.
- the resin particles are dispersed in the chemical conversion treatment film, and at least a part thereof is exposed (protruded) on the surface of the chemical conversion treatment film (see FIG. 3C).
- the resin particles protruding on the surface of the chemical conversion treatment film function like a “roller” during the molding process, thereby improving the lubricity of the chemical conversion treatment film surface. As a result, the workability of the chemical conversion treated Zn-plated steel sheet can be improved.
- the average particle diameter of the resin particles is preferably within the range of 0.1 to 10 ⁇ m.
- the average particle diameter of the resin particles is less than 0.1 ⁇ m, most of the resin particles are buried in the chemical conversion coating, so that the lubricity of the chemical conversion coating surface cannot be improved efficiently.
- the average particle diameter of the resin particles exceeds 10 ⁇ m, the resin particles may be easily dropped during the molding process. If the resin particles fall off in this way, defects will occur in the chemical conversion film, and the corrosion resistance will be reduced.
- the average particle diameter of the resin particles means a particle diameter (median diameter) at an integrated value of 50% in a particle diameter distribution measured by a laser diffraction scattering method.
- the content of the resin particles in the chemical conversion coating is adjusted so that the area occupancy ratio of the resin particles on the surface of the chemical conversion coating is 0.1 area% or more. If the area occupancy of the resin particles is less than 0.1 area%, the lubricity of the chemical conversion coating cannot be sufficiently improved.
- the area occupancy ratio of the resin particles on the surface of the chemical conversion coating can be determined by observing the surface of the chemical conversion coating with a scanning electron microscope (SEM).
- the type of resin particles is not particularly limited, but polyethylene-fluorine resin particles are preferable from the viewpoint of heat resistance and dispersibility.
- Polyethylene resin particles are excellent in dispersibility in the chemical conversion liquid because of low specific gravity (eg, 0.95), but poor in heat resistance because of low melting point (eg, 123 ° C.).
- the shape of the particles may not be maintained when the chemical conversion solution is heated and dried to form a chemical conversion coating. Specifically, when the chemical conversion treatment liquid is heated and dried at about 140 to 180 ° C., the polyethylene resin particles are melted. When the resin particles are thus melted, the resin particles cannot function as “rollers” and cannot contribute to improvement of lubricity.
- the fluororesin particles have a high melting point (eg, 330 ° C.), they are excellent in heat resistance, but because of their large specific gravity (eg, 2.2), they are inferior in dispersibility in the chemical conversion treatment liquid. Thus, when resin particles with low dispersibility in the chemical conversion treatment liquid are used, it becomes difficult to uniformly disperse the resin particles in the chemical conversion treatment film.
- the polyethylene-fluororesin particles are obtained by bonding (adsorbing) fluororesin fine particles to the surface of the polyethylene resin particles by bringing the fluororesin fine particles into contact with the polyethylene resin particles softened by heating.
- the heat resistance is such that the shape of the particles can be maintained even when heated at about 140 to 180 ° C., and the dispersibility in the chemical conversion liquid (specific gravity: For example, 1.1) can be made compatible.
- a weather resistance can also be improved by couple
- FIG. 1 is an SEM image (plan view) of a chemical conversion coating containing polyethylene resin particles or polyethylene-fluororesin particles heated to 50 ° C. or 150 ° C.
- FIG. 1A is an SEM image of a chemical conversion coating formed by applying a chemical conversion treatment liquid containing polyethylene resin particles to the surface of a plated steel sheet and drying at 50 ° C.
- FIG. 1B is an SEM image of a chemical conversion film formed by applying a chemical conversion treatment liquid containing polyethylene resin particles to the surface of a plated steel sheet and drying at 150 ° C.
- FIG. 1 is an SEM image (plan view) of a chemical conversion coating containing polyethylene resin particles or polyethylene-fluororesin particles heated to 50 ° C. or 150 ° C.
- FIG. 1A is an SEM image of a chemical conversion coating formed by applying a chemical conversion treatment liquid containing polyethylene resin particles to the surface of a plated steel sheet and drying at 50 ° C.
- FIG. 1B is an SEM
- FIG. 1C is an SEM image of a chemical conversion film formed by applying a chemical conversion liquid containing polyethylene-fluorine resin particles to the surface of a plated steel sheet and drying at 50 ° C.
- FIG. 1D is an SEM image of a chemical conversion coating formed by applying a chemical conversion treatment liquid containing polyethylene-fluorine resin particles to the surface of a plated steel sheet and drying at 150 ° C.
- the average particle diameter of the polyethylene resin particles constituting the polyethylene-fluorine resin particles is not particularly limited as long as the average particle diameter of the polyethylene-fluorine resin particles is within the range of 0.1 to 10 ⁇ m.
- Examples of commercially available polyethylene resin particles that can be used include HYTEC E-9016, HYTEC E-1000 (all are Toho Chemical Co., Ltd.), CJ-172B, CJ-137 (all are Koyo Chemical Co., Ltd.), and Permarin KUE. -4, Permarin KUE-5 (both are Sanyo Chemical Industries).
- the average particle diameter of the fluororesin fine particles constituting the polyethylene-fluororesin particles may be appropriately set according to the average particle diameter of the polyethylene resin particles.
- the average particle diameter of the fluororesin fine particles is preferably 0.3 ⁇ m or less.
- the ratio of the fluororesin in the polyethylene-fluororesin particles is preferably in the range of 5 to 40% by mass.
- the ratio of the fluororesin in the polyethylene-fluororesin particles can be measured by using a fluorescent X-ray analyzer.
- the chemical conversion film preferably further contains a polyethylene resin.
- the polyethylene resin covers all or a part of the surface of the chemical conversion film where the polyethylene-fluororesin particles do not protrude (see FIG. 2C).
- the polyethylene resin that coats the surface of the chemical conversion coating is a polyethylene resin that bleeds on the surface of the chemical conversion coating in the manufacturing process.
- the polyethylene resin particles, together with the polyethylene-fluorine resin particles improve the lubricity of the surface of the chemical conversion treatment film, thereby further improving the workability of the chemical conversion treatment Zn-plated steel sheet.
- the content of the polyethylene resin in the chemical conversion coating is preferably in the range of 0.1 to 16% by mass with respect to the chemical conversion coating.
- the content of the polyethylene resin is less than 0.1% by mass, the effect of the polyethylene resin cannot be exhibited sufficiently.
- the content of the polyethylene resin is more than 16% by mass, the weather resistance may be lowered.
- the chemical conversion treatment film preferably further contains a phosphate. Phosphate reacts with the plating layer surface of the Al-containing Zn-based alloy plated steel sheet to improve the adhesion of the chemical conversion coating to the Al-containing Zn-based alloy plated steel sheet.
- the type of phosphate is not particularly limited as long as it is a compound having a phosphate anion and is water-soluble.
- phosphates include sodium phosphate, ammonium phosphate, magnesium phosphate, potassium phosphate, manganese phosphate, zinc phosphate, orthophosphoric acid, metaphosphoric acid, pyrophosphoric acid (diphosphoric acid), triphosphoric acid, Tetraphosphate etc. are included. These phosphates may be used alone or in combination of two or more.
- the content of phosphate in the chemical conversion coating is preferably in the range of 0.05 to 3% by mass in terms of P with respect to the fluorine-containing resin.
- P conversion amount is less than 0.05% by mass, the reaction with the plating layer surface is insufficient, and the adhesion of the chemical conversion film cannot be sufficiently improved.
- P conversion amount exceeds 3 mass%, reaction with 4A group metal compound will advance excessively, and the crosslinking effect by 4A group metal compound will be impaired.
- the amount of phosphate converted to P in the chemical conversion coating can be measured by using a fluorescent X-ray analyzer.
- Silane coupling agent It is preferable that a chemical conversion treatment film contains a silane coupling agent further. By mix
- Silane coupling agents include silanes containing one or more functional groups such as amino, epoxy, mercapto, acryloxy, methacryloxy, alkoxy, vinyl, styryl, isocyanate, and chloropropyl groups. A compound is used.
- the content of the silane coupling agent in the chemical conversion film is preferably in the range of 0.5 to 5% by mass with respect to the fluorine-containing resin.
- content of a silane coupling agent is less than 0.5 mass%, the adhesiveness of a chemical conversion treatment film cannot fully be improved.
- content of the silane coupling agent exceeds 5% by mass, the film adhesion is saturated and no further improvement is observed.
- the content of the silane coupling agent in the chemical conversion film can be measured by using a fluorescent X-ray analyzer.
- the film thickness of the chemical conversion coating is preferably in the range of 0.5 to 10 ⁇ m.
- the film thickness is less than 0.5 ⁇ m, sufficient corrosion resistance and discoloration resistance cannot be imparted.
- the film thickness exceeds 10 ⁇ m, it cannot be expected to improve the performance with the increase in film thickness.
- the manufacturing method of the chemical conversion treatment Zn-plated steel plate Although the manufacturing method of the chemical conversion treatment Zn-plated steel plate of this invention is not specifically limited, For example, it can manufacture by the following method.
- the manufacturing method of the chemical conversion treatment Zn-plated steel sheet of the present invention includes 1) a first step of preparing an Al-containing Zn-based alloy plated steel sheet (chemical conversion treatment original plate), and 2) a second step of preparing a chemical conversion treatment liquid. And 3) a third step of forming a chemical conversion film on the surface of the Al-containing Zn-based alloy plated steel sheet.
- a chemical conversion treatment film on the surface of an Al content Zn system alloy plating steel plate via a foundation chemical conversion treatment film before the 3rd step of forming a chemical conversion treatment film, an Al content Zn system alloy plating steel plate
- the method further includes the step of forming a base chemical conversion coating on the surface of the substrate.
- the aforementioned Al-containing Zn-based alloy plated steel sheet is prepared as a chemical conversion treatment original sheet.
- a chemical conversion liquid containing a fluorine-containing resin having a hydrophilic functional group preferably a fluorine-containing olefin resin
- a group 4A metal compound preferably a group 4A metal compound, and resin particles
- a group 4A metal compound and resin particles are added to the aqueous emulsion of the fluorine-containing resin having a hydrophilic functional group (preferably fluorine-containing olefin resin).
- a group 4A metal compound to be added to the chemical conversion treatment liquid a group 4A metal oxyacid salt, fluoride, hydroxide, organic acid salt, carbonate, peroxide salt, or the like is used.
- oxyacid salts include hydrates, ammonium salts, alkali metal salts, alkaline earth metal salts, and the like. You may add a polyethylene resin particle, a phosphate, a silane coupling agent, etc. to a chemical conversion liquid as needed.
- the number average molecular weight of the fluorine-containing resin contained in the aqueous emulsion is preferably 1000 or more, more preferably 10,000 or more, and particularly preferably 200,000 or more. As described above, this is to impart water resistance to the chemical conversion coating. On the other hand, from the viewpoint of the stability of the treatment liquid, the number average molecular weight of the fluorine-containing resin is preferably 2 million or less.
- the fluorine-containing resin preferably has a hydrophilic functional group of 0.05 to 5% by mass from the viewpoint of preparing an aqueous emulsion without using an emulsifier.
- the content of the emulsifier in the aqueous emulsion of the fluorine-containing resin is preferably 1% by mass or less.
- the emulsifier exceeds 1% by mass, the emulsifier may remain in the chemical conversion film depending on the drying temperature when the chemical conversion film is formed in the third step. If the emulsifier remains in the chemical conversion coating as described above, the water resistance of the chemical conversion coating is remarkably lowered, which is not preferable.
- an aqueous emulsion can be prepared even if the amount of the emulsifier is 1% by mass or less as long as it is a fluorine-containing resin having a hydrophilic functional group.
- the emulsifier that may be contained in the aqueous emulsion of the fluorine-containing resin is preferably a fluorine-based emulsifier such as an ammonium salt of perfluorooctanoic acid or an ammonium salt of perfluorononanoic acid from the viewpoint of weather resistance and water resistance.
- a known fluorosurfactant can also be used as an emulsifier.
- the content of the fluorine-containing resin in the chemical conversion treatment liquid is preferably in the range of 10 to 70 parts by mass with respect to 100 parts by mass of water.
- the content of the fluorine-containing resin is less than 10 parts by mass, the amount of water evaporation increases in the drying process, and the film formability and denseness of the chemical conversion film may be reduced.
- content of fluorine-containing resin is more than 70 mass parts, there exists a possibility that the storage stability of a chemical conversion liquid may fall.
- the content of group 4A metal oxyacid salt, fluoride, hydroxide, organic acid salt, carbonate or peroxide in the chemical conversion solution is 0. A range of 1 to 5 parts by mass is preferable.
- the content of these salts is less than 0.1 part by mass, the crosslinking reaction and the reaction with the plating layer surface are insufficient, and the water resistance and film adhesion of the chemical conversion film cannot be sufficiently improved.
- the content of these salts exceeds 5 parts by mass, the cross-linking reaction proceeds and the storage stability of the chemical conversion solution may be reduced.
- the content of the resin particles (for example, polyethylene-fluorine resin particles) in the chemical conversion liquid is in the range of 0.5 to 20 parts by mass with respect to 100 parts by mass of the solid content (fluorine-containing resin, 4A group metal compound, etc.)
- the inside is preferable.
- the resin particle content is less than 0.5% by mass, the lubricity of the chemical conversion coating cannot be sufficiently improved.
- the content of the resin particles is more than 20% by mass, the weather resistance of the chemical conversion film may be deteriorated.
- the content of the polyethylene resin particles in the chemical conversion treatment liquid is 0.1 to 16 masses with respect to 100 mass parts of the solid content. Within the range of parts is preferred. As described above, when the content of the polyethylene resin particles is less than 0.1% by mass, the effect of the polyethylene resin cannot be exhibited sufficiently. On the other hand, when the addition amount exceeds 16% by mass, the weather resistance of the chemical conversion coating may be lowered.
- the average particle diameter of the polyethylene resin particles is preferably in the range of 0.1 to 10 ⁇ m.
- the average particle diameter is less than 0.1 ⁇ m, most of the polyethylene resin particles are buried in the chemical conversion film, and the polyethylene resin cannot be bleed on the surface of the chemical conversion film. On the other hand, when the average particle size is more than 10 ⁇ m, the polyethylene resin particles may fall off while the chemical conversion solution is being dried.
- the phosphate content in the chemical conversion liquid is preferably in the range of 0.05 to 3 parts by mass in terms of P with respect to 100 parts by mass of the fluorine-containing resin. .
- the phosphate content is less than 0.05 parts by mass, the adhesion of the chemical conversion film cannot be sufficiently improved.
- the content of the phosphate is more than 3 parts by mass, the reaction with the 4A group metal compound may proceed excessively and the crosslinking effect by the 4A group metal compound may be impaired.
- the content of the silane coupling agent in the chemical conversion solution is preferably in the range of 0.5 to 5 parts by mass with respect to 100 parts by mass of the fluorine-containing resin.
- content of a silane coupling agent is less than 0.5 mass part, the adhesiveness of a chemical conversion treatment film cannot fully be improved.
- content of the silane coupling agent exceeds 5 parts by mass, the film adhesion is saturated and no further improvement is observed.
- the stability of the chemical conversion liquid may be reduced.
- Etching agents, inorganic compounds, inorganic lubricants, color pigments, dyes, and the like may be added to the chemical conversion treatment liquid as necessary as other components.
- Fluoride etc. are used as an etching agent.
- An etching agent improves the adhesiveness of a chemical conversion treatment film more by activating the plating layer surface.
- Inorganic compounds (oxides, phosphates, etc.) such as Mg, Ca, Sr, V, W, Mn, B, Si, and Sn improve the water resistance by densifying the chemical conversion film.
- Inorganic lubricants such as molybdenum disulfide and talc further improve the lubricity of the chemical conversion coating and, further, the workability of the chemical conversion Zn-plated steel sheet.
- prescribed color tone can be provided to a chemical conversion treatment film by mix
- a chemical conversion treatment film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step.
- the chemical conversion solution prepared in the second step may be applied to the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step and dried.
- the method for applying the chemical conversion liquid is not particularly limited, and may be appropriately selected from known methods.
- Examples of such a coating method include a roll coating method, a curtain flow method, a spin coating method, a spray method, and a dip pulling method.
- the chemical conversion solution may be dried at room temperature, but considering continuous operation, it is preferable to keep the temperature at 50 ° C. or higher to shorten the drying time. However, when the temperature is kept above 300 ° C., the organic component may be thermally decomposed to deteriorate the performance of the chemical conversion coating. In the production method of the present invention, since the content of the emulsifier contained in the chemical conversion treatment liquid is small, even if the drying temperature is about 50 ° C., the emulsifier is hardly contained, and a chemical conversion treatment film having excellent water resistance can be formed. .
- FIG. 2 is a schematic cross-sectional view showing the formation process of the chemical conversion coating.
- FIG. 2 shows a state in which a chemical conversion treatment liquid to which polyethylene-fluorine resin particles and polyethylene resin particles are added is applied.
- a chemical conversion treatment liquid is applied to the surface of the Al-containing Zn-based alloy plated steel sheet 110 to form a coating film 120 of the chemical conversion treatment liquid (see FIG. 2A).
- Polyethylene-fluorine resin particles 122 and polyethylene resin particles 124 are dispersed in the coating film 120 of the chemical conversion treatment liquid.
- FIG. 3A is an SEM image (plan view) of the chemical conversion coating after the chemical conversion coating is dried at 50 ° C.
- FIG. Although it is not possible to distinguish between polyethylene-fluorine resin particles and polyethylene resin particles, it can be seen that these particles protrude from the surface of the chemical conversion coating (see “PE-F or PE” in the figure).
- FIG. 3B is an SEM image (plan view) of the chemical conversion coating after the coating of the chemical conversion solution is dried at 150 ° C. It can be seen that only the polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating (see “PE-F” in the figure).
- FIG. 3B is an SEM image (plan view) of the chemical conversion coating after the coating of the chemical conversion solution is dried at 150 ° C. It can be seen that only the polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating (see “PE-F” in the figure).
- 3C is an SEM image (cross-sectional view) of polyethylene-fluororesin particles protruding from the surface of the chemical conversion coating. It can be confirmed by fluorescent X-ray analysis that the particles protruding from the surface of the chemical conversion coating are polyethylene-fluororesin particles.
- FIG. 4 is a fluorescent X-ray spectrum of polyethylene-fluororesin particles protruding from the surface of the chemical conversion coating.
- the chemical conversion-treated Zn-based plated steel sheet of the present invention which is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion and workability can be produced.
- the base chemical conversion treatment liquid is applied to the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step to form a coating film.
- the base chemical conversion treatment liquid can be applied by, for example, a roll coating method, a spin coating method, a spray method, or the like.
- the coating amount of the base chemical conversion treatment liquid is preferably adjusted so that the valve metal adhesion amount is 1 mg / m 2 or more. It is for providing sufficient corrosion resistance to the chemical conversion treatment steel plate obtained.
- substrate chemical conversion liquid so that the thickness of the foundation
- An undercoat chemical conversion coating can be formed by drying the coating film formed on the surface of the Al-containing Zn-based alloy-plated steel sheet without washing with water. Although it can dry at normal temperature, when continuous operation is considered, it is preferable to hold at 50 degreeC or more and to shorten drying time. However, when the drying temperature exceeds 200 ° C., the organic component contained in the chemical conversion film is thermally decomposed, and the properties imparted with the organic component may be impaired.
- Example 1 Preparation of chemical conversion treated Zn-based plated steel sheet The following three types of molten Al-containing Zn-based alloy plated steel sheets were prepared using SPCC having a thickness of 0.8 mm as a base material. In this example, these three types of molten Al-containing Zn-based alloy-plated steel sheets were used as chemical conversion treatment original sheets.
- a chemical conversion treatment liquid having the composition shown in Table 1 was applied to the surface of each Al-containing Zn-based alloy-plated steel sheet, and dried by heating at a final plate temperature of 140 ° C. to form a chemical conversion treatment film having a thickness of 2.0 ⁇ m.
- the chemical conversion treatment liquids of treatment liquid Nos. 1 to 12 shown in Table 1 are aqueous emulsions containing a predetermined amount of a fluorine-containing resin containing a carboxyl group and a sulfonic acid group and an emulsifier (nonvolatile content: 25% by mass; see Table 2) 4A group metal compound, polyethylene-fluorine resin particles (resin particles) and the like.
- the chemical conversion treatment liquid of treatment liquid No. 13 is a 4A group metal compound, polyethylene-fluorine resin particles (resin particles), etc. added to an aqueous emulsion containing a urethane resin and an emulsifier (nonvolatile content: 25% by mass; see Table 2). Prepared.
- An aqueous emulsion containing a fluorine-containing resin was obtained by adding a predetermined amount of a fluoroolefin, a carboxyl group-containing monomer, a sulfonic acid group-containing monomer and an emulsifier to an aqueous solvent and copolymerizing them.
- PR135 Sudika essence Urethane Co., Ltd.
- A-1891 Momentive Performance Materials Japan GK
- the amounts of Group 4A metal, phosphate, and silane coupling agent relative to the organic resin in the chemical conversion coating of each chemical conversion treatment Zn-plated steel sheet were measured using a fluorescent X-ray analyzer. The contents of phosphate and silane coupling agent were calculated from the measured values of P and Si. Further, the area occupancy ratio of the polyethylene-fluororesin particles on the surface of the chemical conversion coating of each chemical conversion treatment Zn-plated steel sheet was measured using a scanning electron microscope. Table 3 shows the amounts of Group 4A metal, phosphate, and silane coupling agent relative to the organic resin in the chemical conversion coating formed, and the area occupancy ratio of the polyethylene-fluororesin particles on the surface of the chemical conversion coating for each chemical conversion coating. Shown in
- the weather resistance was evaluated by the coating film remaining rate of the chemical conversion coating after the accelerated weather resistance test.
- the chemical conversion treatment Zn-plated steel sheets of Comparative Example 6 Comparative Example 12 and Comparative Example 18 in which a chemical conversion treatment film containing a urethane resin was formed, the chemical conversion treatment film disappeared after 500 cycles (equivalent to outdoor exposure for 5 years).
- the conversion-treated Zn-based plated steel sheets of Comparative Example 4 Comparative Example 10 and Comparative Example 16 in which the chemical conversion treatment film containing excessively large polyethylene-fluororesin particles was formed, the polyethylene-fluororesin particles dropped off from the chemical conversion treatment film. As a result, the weather resistance was poor.
- Blackening resistance was evaluated by the difference in brightness ( ⁇ L * value) before and after the accelerated weather resistance test.
- the chemical conversion treatment Zn-plated steel sheets of Comparative Example 6 Comparative Example 12 and Comparative Example 18 in which a chemical conversion treatment film containing a urethane resin was formed, as the number of cycles increased, the blackening of the plating layer progressed and the brightness decreased. I have.
- the chemical conversion-treated Zn-based plated steel sheets of Examples 1 to 21 in which a chemical conversion film containing a fluorine-containing resin having a predetermined amount of a hydrophilic functional group and a group 4A metal compound was formed 1000 cycles (equivalent to 10 years of outdoor exposure) Even after repeating, the brightness was hardly lowered.
- Corrosion resistance was evaluated based on the white rust generation area ratio after the salt spray test.
- Comparative Example 6 Comparative Example 12 and Comparative Example 18 in which a chemical conversion treatment film containing a urethane resin was formed, although the corrosion resistance was good before the accelerated weather resistance test, the corrosion resistance was accompanied by the disappearance of the film. Has fallen significantly.
- the chemical conversion of Comparative Example 1, Comparative Example 2, Comparative Example 7, Comparative Example 8, Comparative Example 13, and Comparative Example 14 in which a chemical conversion treatment film containing a fluorine-containing resin having an excessive amount or a small amount of a hydrophilic functional group was formed.
- Lubricity was evaluated by the pulling force required for pulling out a test piece to which a load was applied.
- the chemical conversion-treated Zn-plated steel sheets of Comparative Example 1, Comparative Example 7 and Comparative Example 13 were inferior in lubricity because the amount of polyethylene-fluororesin particles in the chemical conversion coating was small.
- the chemically treated Zn-based plated steel sheets of Comparative Example 5, Comparative Example 11 and Comparative Example 17 that did not contain polyethylene-fluorine resin particles also had poor lubricity.
- the chemical conversion-treated Zn-based plated steel sheets of Comparative Example 6, Comparative Example 12 and Comparative Example 18 have poor lubricity because the average particle diameter of the polyethylene-fluorine resin particles is small and buried in the chemical conversion film. It was.
- the chemical conversion treatment Zn-plated steel sheets of Examples 1 to 21 in which the chemical conversion treatment film containing a predetermined amount of polyethylene-fluororesin particles having an average particle diameter within a predetermined range was excellent in lubricity.
- the chemically treated Zn-based plated steel sheet of the present invention is excellent in weather resistance, blackening resistance and workability (lubricity).
- a predetermined amount of group 4A was added to an aqueous emulsion of a fluorine-containing resin having a hydrophilic functional group prepared by adding a hydrophilic functional group-containing monomer to 1% by mass and adding an emulsifier to 1% by mass.
- the chemical conversion treatment solution prepared by adding the metal compound was applied to the surface of the plated steel plate with a bar coater and dried by heating at a final plate temperature of 140 ° C. to form a fluororesin film having a thickness of 30 ⁇ m. This fluororesin film was peeled off from the plated steel sheet and cut into a predetermined size to obtain a test piece. About each test piece (free fluororesin film
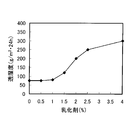
- FIG. 5 is a graph showing the relationship between the amount of group 4A metal in the fluororesin film and the water vapor transmission rate. From this graph, it is understood that the moisture permeability of the fluororesin film can be remarkably lowered by setting the amount of the group 4A metal in the fluororesin film to 0.1 mass% or more.
- FIG. 6 is a graph showing the relationship between the concentration of the emulsifier in the aqueous emulsion of the fluorine-containing resin and the moisture permeability of the fluorine resin film. From this graph, it is understood that the moisture permeability of the fluororesin film can be remarkably reduced by setting the concentration of the emulsifier in the emulsion to 1% by mass or less.
- the fluororesin film having a large amount of the group 4A metal compound and a small amount of the remaining emulsifier has excellent water resistance.
- the chemical conversion-treated Zn-based plated steel sheet of the present invention is useful in various applications such as exterior building materials because it is excellent in weather resistance, water resistance, blackening resistance, film adhesion and workability.
- the chemical conversion-treated Zn-based plated steel sheet of the present invention includes: 1) steel pipe, shape steel, support, beam, conveying member for a greenhouse or agricultural house, 2) sound insulation wall, sound insulation wall, sound absorption wall, snow insulation wall, guardrail, It can be suitably used for applications such as railings, protective fences, columns, 3) railway vehicle members, overhead wire members, electrical equipment members, safety environment members, structural members, and solar mounts.
- the Al-containing Zn-based alloy-plated steel sheet is sufficiently in close contact with the coating film under high temperature and high humidity and has excellent corrosion resistance. Therefore, the chemical conversion treatment Zn-plated steel sheet of the present invention is particularly suitable as an exterior material used in a high temperature and high humidity environment.
Abstract
The surface of an Al-containing Zn-alloy-plated steel sheet is coated with a chemical conversion treatment liquid, which is dried to form a chemical conversion treatment film having a thickness of 0.5-10 μm. The chemical conversion treatment liquid contains: a fluororesin which includes 0.05-5 wt% of a hydrophilic functional group and 7-20 wt% of F atoms, the fluororesin having a number-average molecular weight within the range of 1,000-2,000,000; an oxygen acid salt, fluoride, hydroxide, organic acid salt, carbonate, or peroxide salt of a group 4A metal; and resin particles having an average particle size of 0.1-10 μm. The amount of the oxygen acid salt, fluoride, hydroxide, organic acid salt, carbonate, or peroxide salt of a group 4A metal relative to the fluororesin in the chemical conversion treatment liquid is within the range of 0.1-5 wt% expressed as metal.
Description
本発明は、耐候性、耐水性、耐黒変性、皮膜密着性および加工性に優れる化成処理Zn系めっき鋼板およびその製造方法に関する。
The present invention relates to a chemically treated Zn-based plated steel sheet having excellent weather resistance, water resistance, blackening resistance, film adhesion and workability, and a method for producing the same.
めっき鋼板は、成形加工時のカジリを防止するために、その表面に有機樹脂を含む化成処理皮膜を形成されることがある(例えば、特許文献1、2参照)。特許文献1、2には、亜鉛系めっき鋼板の表面に、ウレタン樹脂などの有機樹脂を含む化成処理皮膜を形成することが記載されている。このようにめっき鋼板の表面を、有機樹脂を含む化成処理皮膜で被覆することで、耐カジリ性だけでなく、耐食性や耐変色性なども向上させることができる。
The plated steel sheet is sometimes formed with a chemical conversion treatment film containing an organic resin on its surface in order to prevent galling during forming (for example, see Patent Documents 1 and 2). Patent Documents 1 and 2 describe that a chemical conversion treatment film containing an organic resin such as a urethane resin is formed on the surface of a zinc-based plated steel sheet. Thus, by covering the surface of the plated steel sheet with a chemical conversion treatment film containing an organic resin, not only galling resistance but also corrosion resistance and discoloration resistance can be improved.
一方、化成処理めっき鋼板の耐候性を向上させるために、化成処理皮膜を構成する有機樹脂として耐候性に優れるフッ素樹脂を使用することがある。このように耐候性の向上を目的としてフッ素樹脂を使用する場合、有機溶剤系フッ素樹脂組成物が使用されることが多い。しかし、このような有機溶剤系フッ素樹脂組成物には、火災の危険性や有害性、大気汚染などの問題がある。
On the other hand, in order to improve the weather resistance of the chemically treated steel sheet, a fluororesin having excellent weather resistance may be used as an organic resin constituting the chemical conversion film. Thus, when using a fluororesin for the purpose of an improvement in a weather resistance, an organic solvent type fluororesin composition is used in many cases. However, such organic solvent-based fluororesin compositions have problems such as fire hazard and harmfulness and air pollution.
また、水系フッ素樹脂組成物も様々なものが提案されている(例えば、特許文献3参照)。しかし、このような水系フッ素樹脂組成物は、いずれも高温での焼付けを必要とする(例えば180~230℃、特許文献3参照)。このような高温での焼き付けは、現場での塗装においては現実的に不可能であり(通常は常乾樹脂を使用する)、加熱乾燥が主流である工場ラインにおいても不利である。
Various water-based fluororesin compositions have been proposed (see, for example, Patent Document 3). However, all such water-based fluororesin compositions require baking at a high temperature (for example, 180 to 230 ° C., see Patent Document 3). Such high-temperature baking is practically impossible in field painting (usually using a normally dry resin), and is disadvantageous even in factory lines where heat drying is the mainstream.
さらに、水系フッ素樹脂組成物の問題点を解消すべく、硬化性部位(有機官能基)を導入して、低温でも造膜可能な水系フッ素樹脂組成物も提案されている(例えば、特許文献4参照)。しかし、有機官能基を反応させて硬化させた皮膜では、当該硬化部から優先的に耐候劣化してしまうため、皮膜が多孔質状になり、耐水性が低下してしまう。また、密着性改善のためにエポキシ系樹脂やウレタン樹脂などによる下地処理が施された場合も、有機樹脂が優先的に耐候劣化してしまい、皮膜密着性が急激に低下してしまうことになる。
Furthermore, in order to solve the problems of the water-based fluororesin composition, a water-based fluororesin composition capable of forming a film even at a low temperature by introducing a curable site (organic functional group) has been proposed (for example, Patent Document 4). reference). However, in a film cured by reacting with an organic functional group, the weather resistance deteriorates preferentially from the cured portion, so that the film becomes porous and water resistance decreases. In addition, when the base treatment with an epoxy resin or a urethane resin is performed for improving the adhesion, the organic resin is preferentially deteriorated in weather resistance, and the film adhesion is rapidly lowered. .
前述の通り、めっき鋼板の表面に有機樹脂を含む化成処理皮膜を形成することで、耐カジリ性や耐食性、耐変色性などを向上させることができる。しかしながら、有機樹脂を含む化成処理皮膜を形成された従来の化成処理めっき鋼板は、外装建材として使用した場合に耐候性が不十分である場合がある。すなわち、ウレタン樹脂などの多くの有機樹脂は紫外線により劣化してしまうため、従来の化成処理めっき鋼板を外装建材として使用した場合、めっき鋼板の表面を被覆する化成処理皮膜が時間の経過とともに失われてしまうおそれがある。このように化成処理皮膜が失われてしまうと、変色や錆などが発生して美観が損なわれるおそれがあり、外装建材として好ましくない。
As described above, galling resistance, corrosion resistance, discoloration resistance, and the like can be improved by forming a chemical conversion film containing an organic resin on the surface of a plated steel sheet. However, the conventional chemical conversion treatment plated steel sheet formed with the chemical conversion treatment film containing the organic resin may have insufficient weather resistance when used as an exterior building material. In other words, since many organic resins such as urethane resins are deteriorated by ultraviolet rays, when a conventional chemical conversion-treated plated steel sheet is used as an exterior building material, the chemical conversion film covering the surface of the plated steel sheet is lost over time. There is a risk that. If the chemical conversion treatment film is lost in this manner, discoloration, rust, etc. may occur and the appearance may be impaired, which is not preferable as an exterior building material.
このような化成処理めっき鋼板の耐候性を向上させる手段としては、化成処理皮膜を構成する有機樹脂として耐候性に優れるフッ素樹脂を使用することが考えられる。そこで、本発明者は取り扱いが容易な水系フッ素樹脂のエマルションを用いてめっき鋼板の表面に化成処理皮膜を形成する予備実験を行った。その結果、水系フッ素樹脂のエマルションを使用することで、耐紫外線性を向上させることはできたが、その一方で造膜性、耐水性および皮膜密着性が低下してしまった。本発明者によるさらなる検討の結果、これらの品質の低下は、水系フッ素樹脂のエマルションを製造する際に使用される乳化剤(例えば、ペルフルオロオクタン酸アンモニウム塩)が化成処理皮膜中に残存するためであることが推察された(後述の参考実験参照)。
As a means for improving the weather resistance of such a chemical conversion-treated plated steel sheet, it is conceivable to use a fluororesin having excellent weather resistance as the organic resin constituting the chemical conversion film. Then, this inventor performed the preliminary experiment which forms a chemical conversion treatment film on the surface of a plated steel plate using the emulsion of the water-based fluororesin which is easy to handle. As a result, it was possible to improve the ultraviolet resistance by using an emulsion of a water-based fluororesin, but on the other hand, the film forming property, water resistance and film adhesion were lowered. As a result of further studies by the present inventors, these deteriorations in quality are because an emulsifier (for example, perfluorooctanoic acid ammonium salt) used in producing an aqueous fluororesin emulsion remains in the chemical conversion film. (See the reference experiment below).
以上のように、有機樹脂を含む化成処理皮膜を形成された従来の化成処理めっき鋼板は、耐候性が不十分である場合がある。また、有機樹脂として水系フッ素樹脂を使用することで、化成処理めっき鋼板の耐候性(耐紫外線性)を向上させることはできるが、その一方で造膜性、耐水性および皮膜密着性が低下してしまうため、耐候性と、耐水性、耐黒変性および皮膜密着性とを両立させることはできない。
As described above, a conventional chemical conversion-treated steel sheet on which a chemical conversion treatment film containing an organic resin is formed may have insufficient weather resistance. In addition, the use of water-based fluororesin as the organic resin can improve the weather resistance (ultraviolet light resistance) of the chemically treated steel sheet, but on the other hand, the film forming property, water resistance and film adhesion are reduced. Therefore, it is impossible to achieve both weather resistance, water resistance, blackening resistance and film adhesion.
本発明の目的は、有機樹脂を含む化成処理皮膜を有する化成処理Zn系めっき鋼板であって、耐候性、耐水性、耐黒変性、皮膜密着性および加工性のすべてに優れる化成処理Zn系めっき鋼板を提供することである。
An object of the present invention is a chemical conversion treatment Zn-plated steel sheet having a chemical conversion treatment film containing an organic resin, which is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion and workability. It is to provide a steel plate.
本発明者は、有機樹脂として親水性官能基を導入した高分子量のフッ素含有樹脂を使用し、かつこれらのフッ素含有樹脂を4A族金属化合物で架橋することで、化成処理皮膜の耐候性、耐水性、耐黒変性および皮膜密着性を向上させうることを見出し、さらに検討を加えて本発明を完成させた。
The present inventor uses a high molecular weight fluorine-containing resin into which a hydrophilic functional group is introduced as an organic resin, and crosslinks these fluorine-containing resins with a group 4A metal compound, thereby enabling the weather resistance and water resistance of the chemical conversion coating. The present invention has been completed through further investigations, which have been found to improve the property, blackening resistance and film adhesion.
すなわち、本発明の第一は、以下の化成処理Zn系めっき鋼板に関する。
[1]Alを0.05~60質量%含むZn系合金めっき層を有するAl含有Zn系合金めっき鋼板と、前記Al含有Zn系合金めっき鋼板の表面に形成された、膜厚0.5~10μmの化成処理皮膜と、を有する化成処理Zn系めっき鋼板であって;前記化成処理皮膜は、カルボキシル基、スルホン酸基およびこれらの塩からなる群から選ばれる親水性官能基0.05~5質量%とF原子7~20質量%とを含有するフッ素含有樹脂と、前記フッ素含有樹脂に対して金属換算で0.1~5質量%の4A族金属化合物と、平均粒子径0.1~10μmの樹脂粒子とを含有し;前記化成処理皮膜の表面における前記樹脂粒子の面積占有率は、0.1面積%以上である、化成処理Zn系めっき鋼板。
[2]前記フッ素含有樹脂が有するカルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内である、[1]に記載の化成処理Zn系めっき鋼板。
[3]前記樹脂粒子は、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子が結合されているポリエチレン-フッ素樹脂粒子である、[1]または[2]に記載の化成処理Zn系めっき鋼板。
[4]前記化成処理皮膜は、さらにポリエチレン樹脂を含有し;前記ポリエチレン-フッ素樹脂粒子は、前記化成処理皮膜の表面の一部において、前記化成処理皮膜の表面から突出しており;前記ポリエチレン樹脂は、前記化成処理皮膜の表面の残部の全部または一部を被覆している、[3]に記載の化成処理Zn系めっき鋼板。
[5]前記化成処理皮膜は、さらにリン酸塩を含有し;前記フッ素含有樹脂に対する前記リン酸塩の量は、P換算で0.05~3質量%の範囲内である、[1]~[4]のいずれか一項に記載の化成処理Zn系めっき鋼板。
[6]前記化成処理皮膜は、さらにシランカップリング剤を含有し;前記フッ素含有樹脂に対する前記シランカップリング剤の量は、0.5~5質量%の範囲内である、[1]~[5]のいずれか一項に記載の化成処理Zn系めっき鋼板。
[7]前記4A族金属は、Ti、Zr、Hfおよびこれらの組み合わせからなる群から選ばれる、[1]~[6]のいずれか一項に記載の化成処理Zn系めっき鋼板。
[8]前記Al含有Zn系合金めっき鋼板と、前記化成処理皮膜との間に形成された、バルブメタルの酸化物または水酸化物、およびバルブメタルのフッ化物を含有する下地化成処理皮膜をさらに有する、[1]~[7]のいずれか一項に記載の化成処理Zn系めっき鋼板。 That is, the first of the present invention relates to the following chemical conversion treatment Zn-plated steel sheet.
[1] An Al-containing Zn-based alloy plated steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al, and a film thickness of 0.5 to 0.5 formed on the surface of the Al-containing Zn-based alloy plated steel sheet A chemical conversion treatment Zn-plated steel sheet having a chemical treatment film of 10 μm; the chemical conversion treatment film having a hydrophilic functional group of 0.05 to 5 selected from the group consisting of a carboxyl group, a sulfonic acid group and salts thereof A fluorine-containing resin containing 5% by mass and 7 to 20% by mass of F atoms, 0.1 to 5% by mass of a 4A group metal compound in terms of metal with respect to the fluorine-containing resin, A chemical conversion-treated Zn-based plated steel sheet, comprising 10 μm resin particles; and an area occupancy ratio of the resin particles on the surface of the chemical conversion coating is 0.1 area% or more.
[2] The chemical conversion treatment Zn-based plating according to [1], wherein the ratio of the carboxyl group and the sulfonic acid group of the fluorine-containing resin is within a range of 5 to 60 in terms of a molar ratio of carboxyl group / sulfonic acid group. steel sheet.
[3] The chemical conversion-treated Zn-based plated steel sheet according to [1] or [2], wherein the resin particles are polyethylene-fluororesin particles in which fluororesin fine particles are bonded to the surface of polyethylene resin particles.
[4] The chemical conversion coating further contains a polyethylene resin; the polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating in part of the surface of the chemical conversion coating; The chemical conversion treatment Zn-plated steel sheet according to [3], which covers all or part of the remaining part of the surface of the chemical conversion treatment film.
[5] The chemical conversion film further contains a phosphate; the amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P. [1] to [4] The chemical conversion treatment Zn-plated steel sheet according to any one of [4].
[6] The chemical conversion treatment film further contains a silane coupling agent; the amount of the silane coupling agent with respect to the fluorine-containing resin is in the range of 0.5 to 5% by mass. 5] The chemical conversion treatment Zn-plated steel sheet according to any one of [5].
[7] The chemical conversion-treated Zn-plated steel sheet according to any one of [1] to [6], wherein the group 4A metal is selected from the group consisting of Ti, Zr, Hf, and combinations thereof.
[8] An underlayer chemical conversion coating formed between the Al-containing Zn-based alloy-plated steel sheet and the chemical conversion coating and containing a valve metal oxide or hydroxide and a valve metal fluoride. The chemical conversion-treated Zn-based plated steel sheet according to any one of [1] to [7].
[1]Alを0.05~60質量%含むZn系合金めっき層を有するAl含有Zn系合金めっき鋼板と、前記Al含有Zn系合金めっき鋼板の表面に形成された、膜厚0.5~10μmの化成処理皮膜と、を有する化成処理Zn系めっき鋼板であって;前記化成処理皮膜は、カルボキシル基、スルホン酸基およびこれらの塩からなる群から選ばれる親水性官能基0.05~5質量%とF原子7~20質量%とを含有するフッ素含有樹脂と、前記フッ素含有樹脂に対して金属換算で0.1~5質量%の4A族金属化合物と、平均粒子径0.1~10μmの樹脂粒子とを含有し;前記化成処理皮膜の表面における前記樹脂粒子の面積占有率は、0.1面積%以上である、化成処理Zn系めっき鋼板。
[2]前記フッ素含有樹脂が有するカルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内である、[1]に記載の化成処理Zn系めっき鋼板。
[3]前記樹脂粒子は、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子が結合されているポリエチレン-フッ素樹脂粒子である、[1]または[2]に記載の化成処理Zn系めっき鋼板。
[4]前記化成処理皮膜は、さらにポリエチレン樹脂を含有し;前記ポリエチレン-フッ素樹脂粒子は、前記化成処理皮膜の表面の一部において、前記化成処理皮膜の表面から突出しており;前記ポリエチレン樹脂は、前記化成処理皮膜の表面の残部の全部または一部を被覆している、[3]に記載の化成処理Zn系めっき鋼板。
[5]前記化成処理皮膜は、さらにリン酸塩を含有し;前記フッ素含有樹脂に対する前記リン酸塩の量は、P換算で0.05~3質量%の範囲内である、[1]~[4]のいずれか一項に記載の化成処理Zn系めっき鋼板。
[6]前記化成処理皮膜は、さらにシランカップリング剤を含有し;前記フッ素含有樹脂に対する前記シランカップリング剤の量は、0.5~5質量%の範囲内である、[1]~[5]のいずれか一項に記載の化成処理Zn系めっき鋼板。
[7]前記4A族金属は、Ti、Zr、Hfおよびこれらの組み合わせからなる群から選ばれる、[1]~[6]のいずれか一項に記載の化成処理Zn系めっき鋼板。
[8]前記Al含有Zn系合金めっき鋼板と、前記化成処理皮膜との間に形成された、バルブメタルの酸化物または水酸化物、およびバルブメタルのフッ化物を含有する下地化成処理皮膜をさらに有する、[1]~[7]のいずれか一項に記載の化成処理Zn系めっき鋼板。 That is, the first of the present invention relates to the following chemical conversion treatment Zn-plated steel sheet.
[1] An Al-containing Zn-based alloy plated steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al, and a film thickness of 0.5 to 0.5 formed on the surface of the Al-containing Zn-based alloy plated steel sheet A chemical conversion treatment Zn-plated steel sheet having a chemical treatment film of 10 μm; the chemical conversion treatment film having a hydrophilic functional group of 0.05 to 5 selected from the group consisting of a carboxyl group, a sulfonic acid group and salts thereof A fluorine-containing resin containing 5% by mass and 7 to 20% by mass of F atoms, 0.1 to 5% by mass of a 4A group metal compound in terms of metal with respect to the fluorine-containing resin, A chemical conversion-treated Zn-based plated steel sheet, comprising 10 μm resin particles; and an area occupancy ratio of the resin particles on the surface of the chemical conversion coating is 0.1 area% or more.
[2] The chemical conversion treatment Zn-based plating according to [1], wherein the ratio of the carboxyl group and the sulfonic acid group of the fluorine-containing resin is within a range of 5 to 60 in terms of a molar ratio of carboxyl group / sulfonic acid group. steel sheet.
[3] The chemical conversion-treated Zn-based plated steel sheet according to [1] or [2], wherein the resin particles are polyethylene-fluororesin particles in which fluororesin fine particles are bonded to the surface of polyethylene resin particles.
[4] The chemical conversion coating further contains a polyethylene resin; the polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating in part of the surface of the chemical conversion coating; The chemical conversion treatment Zn-plated steel sheet according to [3], which covers all or part of the remaining part of the surface of the chemical conversion treatment film.
[5] The chemical conversion film further contains a phosphate; the amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P. [1] to [4] The chemical conversion treatment Zn-plated steel sheet according to any one of [4].
[6] The chemical conversion treatment film further contains a silane coupling agent; the amount of the silane coupling agent with respect to the fluorine-containing resin is in the range of 0.5 to 5% by mass. 5] The chemical conversion treatment Zn-plated steel sheet according to any one of [5].
[7] The chemical conversion-treated Zn-plated steel sheet according to any one of [1] to [6], wherein the group 4A metal is selected from the group consisting of Ti, Zr, Hf, and combinations thereof.
[8] An underlayer chemical conversion coating formed between the Al-containing Zn-based alloy-plated steel sheet and the chemical conversion coating and containing a valve metal oxide or hydroxide and a valve metal fluoride. The chemical conversion-treated Zn-based plated steel sheet according to any one of [1] to [7].
本発明の第二は、以下の化成処理Zn系めっき鋼板の製造方法に関する。
[9]Alを0.05~60質量%含むZn系合金めっき層を有するAl含有Zn系合金めっき鋼板を準備するステップと、前記Al含有Zn系合金めっき鋼板の表面に化成処理液を塗布し、乾燥させて、膜厚が0.5~10μmの化成処理皮膜を形成するステップとを含み;前記化成処理液は、カルボキシル基、スルホン酸基およびこれらの塩からなる群から選ばれる親水性官能基0.05~5質量%とF原子7~20質量%とを含有し、数平均分子量が1000~200万の範囲内であるフッ素含有樹脂と、4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩のいずれかと、平均粒子径が0.1~10μmの樹脂粒子とを含有し;前記フッ素含有樹脂に対する、前記4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩の量は、金属換算で0.1~5質量%の範囲内であり;前記化成処理液中の固形分に対する、前記樹脂粒子の量は、0.5~20質量%の範囲内である、化成処理Zn系めっき鋼板の製造方法。
[10]前記フッ素含有樹脂が有するカルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内である、[9]に記載の化成処理Zn系めっき鋼板の製造方法。
[11]前記樹脂粒子は、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子が結合されているポリエチレン-フッ素樹脂粒子である、[9]または[10]に記載の化成処理Zn系めっき鋼板の製造方法。
[12]前記化成処理液は、さらにポリエチレン樹脂粒子を含有する、[11]に記載の化成処理Zn系めっき鋼板の製造方法。
[13]前記化成処理液は、さらにリン酸塩を含有し;前記フッ素含有樹脂に対する前記リン酸塩の量は、P換算で0.05~3質量%の範囲内である、[9]~[12]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。
[14]前記化成処理液は、さらにシランカップリング剤を含有し;前記フッ素含有樹脂に対する前記シランカップリング剤の量は、0.5~5質量%の範囲内である、[9]~[13]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。
[15]前記4A族金属は、Ti、Zr、Hfおよびこれらの組み合わせからなる群から選ばれる、[9]~[14]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。
[16]前記化成処理皮膜を形成するステップの前に、前記Al含有Zn系合金めっき鋼板の表面に下地化成処理液を塗布し、乾燥させて、下地化成処理皮膜を形成するステップをさらに含み;前記下地化成処理液は、バルブメタル塩とフッ化物イオンとを含有する、[9]~[15]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。 2nd of this invention is related with the manufacturing method of the following chemical conversion treatment Zn-plated steel plate.
[9] A step of preparing an Al-containing Zn-based alloy plated steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al, and applying a chemical conversion treatment solution to the surface of the Al-containing Zn-based alloy plated steel sheet And drying to form a chemical conversion film having a film thickness of 0.5 to 10 μm; the chemical conversion solution is a hydrophilic functional group selected from the group consisting of carboxyl groups, sulfonic acid groups, and salts thereof. A fluorine-containing resin containing 0.05 to 5% by mass of a group and 7 to 20% by mass of F atoms and having a number average molecular weight in the range of 1,000 to 2,000,000, a group 4A metal oxyacid salt, fluoride, Any one of hydroxide, organic acid salt, carbonate or peroxide salt, and resin particles having an average particle size of 0.1 to 10 μm; an oxyacid salt of the group 4A metal with respect to the fluorine-containing resin; Fluoride, hydroxide, existence The amount of the acid salt, carbonate or peroxide is in the range of 0.1 to 5% by mass in terms of metal; the amount of the resin particles relative to the solid content in the chemical conversion solution is 0.5 to The manufacturing method of the chemical conversion treatment Zn-plated steel plate which exists in the range of 20 mass%.
[10] The chemical conversion treatment Zn-based plating according to [9], wherein the ratio of the carboxyl group and the sulfonic acid group of the fluorine-containing resin is within a range of 5 to 60 in terms of a molar ratio of carboxyl group / sulfonic acid group. A method of manufacturing a steel sheet.
[11] The method for producing a chemically treated Zn-based plated steel sheet according to [9] or [10], wherein the resin particles are polyethylene-fluorine resin particles in which fluororesin fine particles are bonded to the surface of polyethylene resin particles.
[12] The method for producing a chemical conversion-treated Zn-based plated steel sheet according to [11], wherein the chemical conversion treatment liquid further contains polyethylene resin particles.
[13] The chemical conversion treatment liquid further contains a phosphate; the amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P, [9] to The manufacturing method of the chemical conversion treatment Zn-plated steel plate as described in any one of [12].
[14] The chemical conversion treatment liquid further contains a silane coupling agent; the amount of the silane coupling agent with respect to the fluorine-containing resin is in the range of 0.5 to 5% by mass. 13] The manufacturing method of the chemical conversion treatment Zn-plated steel plate as described in any one of [13].
[15] The method for producing a chemically treated Zn-based plated steel sheet according to any one of [9] to [14], wherein the group 4A metal is selected from the group consisting of Ti, Zr, Hf, and combinations thereof.
[16] Before the step of forming the chemical conversion treatment film, the method further includes the step of applying a base chemical conversion treatment solution to the surface of the Al-containing Zn-based alloy plated steel sheet and drying to form a base chemical conversion treatment film; The method for producing a chemical conversion-treated Zn-based plated steel sheet according to any one of [9] to [15], wherein the base chemical conversion treatment solution contains a valve metal salt and fluoride ions.
[9]Alを0.05~60質量%含むZn系合金めっき層を有するAl含有Zn系合金めっき鋼板を準備するステップと、前記Al含有Zn系合金めっき鋼板の表面に化成処理液を塗布し、乾燥させて、膜厚が0.5~10μmの化成処理皮膜を形成するステップとを含み;前記化成処理液は、カルボキシル基、スルホン酸基およびこれらの塩からなる群から選ばれる親水性官能基0.05~5質量%とF原子7~20質量%とを含有し、数平均分子量が1000~200万の範囲内であるフッ素含有樹脂と、4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩のいずれかと、平均粒子径が0.1~10μmの樹脂粒子とを含有し;前記フッ素含有樹脂に対する、前記4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩の量は、金属換算で0.1~5質量%の範囲内であり;前記化成処理液中の固形分に対する、前記樹脂粒子の量は、0.5~20質量%の範囲内である、化成処理Zn系めっき鋼板の製造方法。
[10]前記フッ素含有樹脂が有するカルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内である、[9]に記載の化成処理Zn系めっき鋼板の製造方法。
[11]前記樹脂粒子は、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子が結合されているポリエチレン-フッ素樹脂粒子である、[9]または[10]に記載の化成処理Zn系めっき鋼板の製造方法。
[12]前記化成処理液は、さらにポリエチレン樹脂粒子を含有する、[11]に記載の化成処理Zn系めっき鋼板の製造方法。
[13]前記化成処理液は、さらにリン酸塩を含有し;前記フッ素含有樹脂に対する前記リン酸塩の量は、P換算で0.05~3質量%の範囲内である、[9]~[12]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。
[14]前記化成処理液は、さらにシランカップリング剤を含有し;前記フッ素含有樹脂に対する前記シランカップリング剤の量は、0.5~5質量%の範囲内である、[9]~[13]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。
[15]前記4A族金属は、Ti、Zr、Hfおよびこれらの組み合わせからなる群から選ばれる、[9]~[14]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。
[16]前記化成処理皮膜を形成するステップの前に、前記Al含有Zn系合金めっき鋼板の表面に下地化成処理液を塗布し、乾燥させて、下地化成処理皮膜を形成するステップをさらに含み;前記下地化成処理液は、バルブメタル塩とフッ化物イオンとを含有する、[9]~[15]のいずれか一項に記載の化成処理Zn系めっき鋼板の製造方法。 2nd of this invention is related with the manufacturing method of the following chemical conversion treatment Zn-plated steel plate.
[9] A step of preparing an Al-containing Zn-based alloy plated steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al, and applying a chemical conversion treatment solution to the surface of the Al-containing Zn-based alloy plated steel sheet And drying to form a chemical conversion film having a film thickness of 0.5 to 10 μm; the chemical conversion solution is a hydrophilic functional group selected from the group consisting of carboxyl groups, sulfonic acid groups, and salts thereof. A fluorine-containing resin containing 0.05 to 5% by mass of a group and 7 to 20% by mass of F atoms and having a number average molecular weight in the range of 1,000 to 2,000,000, a group 4A metal oxyacid salt, fluoride, Any one of hydroxide, organic acid salt, carbonate or peroxide salt, and resin particles having an average particle size of 0.1 to 10 μm; an oxyacid salt of the group 4A metal with respect to the fluorine-containing resin; Fluoride, hydroxide, existence The amount of the acid salt, carbonate or peroxide is in the range of 0.1 to 5% by mass in terms of metal; the amount of the resin particles relative to the solid content in the chemical conversion solution is 0.5 to The manufacturing method of the chemical conversion treatment Zn-plated steel plate which exists in the range of 20 mass%.
[10] The chemical conversion treatment Zn-based plating according to [9], wherein the ratio of the carboxyl group and the sulfonic acid group of the fluorine-containing resin is within a range of 5 to 60 in terms of a molar ratio of carboxyl group / sulfonic acid group. A method of manufacturing a steel sheet.
[11] The method for producing a chemically treated Zn-based plated steel sheet according to [9] or [10], wherein the resin particles are polyethylene-fluorine resin particles in which fluororesin fine particles are bonded to the surface of polyethylene resin particles.
[12] The method for producing a chemical conversion-treated Zn-based plated steel sheet according to [11], wherein the chemical conversion treatment liquid further contains polyethylene resin particles.
[13] The chemical conversion treatment liquid further contains a phosphate; the amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P, [9] to The manufacturing method of the chemical conversion treatment Zn-plated steel plate as described in any one of [12].
[14] The chemical conversion treatment liquid further contains a silane coupling agent; the amount of the silane coupling agent with respect to the fluorine-containing resin is in the range of 0.5 to 5% by mass. 13] The manufacturing method of the chemical conversion treatment Zn-plated steel plate as described in any one of [13].
[15] The method for producing a chemically treated Zn-based plated steel sheet according to any one of [9] to [14], wherein the group 4A metal is selected from the group consisting of Ti, Zr, Hf, and combinations thereof.
[16] Before the step of forming the chemical conversion treatment film, the method further includes the step of applying a base chemical conversion treatment solution to the surface of the Al-containing Zn-based alloy plated steel sheet and drying to form a base chemical conversion treatment film; The method for producing a chemical conversion-treated Zn-based plated steel sheet according to any one of [9] to [15], wherein the base chemical conversion treatment solution contains a valve metal salt and fluoride ions.
本発明によれば、耐候性、耐水性、耐黒変性、皮膜密着性および加工性のすべてに優れる化成処理Zn系めっき鋼板を提供することができる。本発明の化成処理Zn系めっき鋼板は、耐候性、耐水性、耐食性、耐変色性および加工性に優れているため、例えば外装建材用のめっき鋼板として有用である。
According to the present invention, it is possible to provide a chemically treated Zn-based plated steel sheet that is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion, and workability. Since the chemical conversion treatment Zn-plated steel sheet of the present invention is excellent in weather resistance, water resistance, corrosion resistance, discoloration resistance and workability, it is useful as a plated steel sheet for exterior building materials, for example.
1.化成処理Zn系めっき鋼板
本発明の化成処理Zn系めっき鋼板は、Al含有Zn系合金めっき鋼板(化成処理原板)と、Al含有Zn系合金めっき鋼板の表面に形成された化成処理皮膜とを有する。本発明の化成処理Zn系めっき鋼板は、化成処理皮膜が親水性官能基(カルボキシル基やスルホン酸基など)を導入した高分子量のフッ素含有樹脂、4A族金属化合物および樹脂粒子(潤滑剤)を含むことを一つの特徴とする。 1. Chemical conversion treatment Zn-plated steel sheet The chemical conversion treatment Zn-plated steel sheet of the present invention has an Al-containing Zn-based alloy-plated steel sheet (chemical conversion-treated original plate) and a chemical conversion-treated film formed on the surface of the Al-containing Zn-based alloy-plated steel sheet. . The chemical conversion treatment Zn-based plated steel sheet of the present invention comprises a high molecular weight fluorine-containing resin, a group 4A metal compound and a resin particle (lubricant) in which a chemical conversion treatment film has introduced a hydrophilic functional group (such as a carboxyl group or a sulfonic acid group). Inclusion is a feature.
本発明の化成処理Zn系めっき鋼板は、Al含有Zn系合金めっき鋼板(化成処理原板)と、Al含有Zn系合金めっき鋼板の表面に形成された化成処理皮膜とを有する。本発明の化成処理Zn系めっき鋼板は、化成処理皮膜が親水性官能基(カルボキシル基やスルホン酸基など)を導入した高分子量のフッ素含有樹脂、4A族金属化合物および樹脂粒子(潤滑剤)を含むことを一つの特徴とする。 1. Chemical conversion treatment Zn-plated steel sheet The chemical conversion treatment Zn-plated steel sheet of the present invention has an Al-containing Zn-based alloy-plated steel sheet (chemical conversion-treated original plate) and a chemical conversion-treated film formed on the surface of the Al-containing Zn-based alloy-plated steel sheet. . The chemical conversion treatment Zn-based plated steel sheet of the present invention comprises a high molecular weight fluorine-containing resin, a group 4A metal compound and a resin particle (lubricant) in which a chemical conversion treatment film has introduced a hydrophilic functional group (such as a carboxyl group or a sulfonic acid group). Inclusion is a feature.
以下、本発明の化成処理Zn系めっき鋼板の各構成要素について説明する。
Hereafter, each component of the chemical conversion treatment Zn-plated steel plate of this invention is demonstrated.
[化成処理原板]
化成処理原板としては、耐食性および意匠性に優れる、Al含有Zn系合金めっき鋼板が使用される。ここで「Al含有Zn系合金めっき鋼板」とは、Alを0.05~60質量%含むZn系合金めっき層を有する鋼板を意味する。Al含有Zn系合金めっき鋼板の例には、溶融Al-Znめっき鋼板(溶融Zn-0.1%Alめっき、溶融Zn-55%Alめっき、溶融Zn-6%Al-3%Mgめっき、溶融Zn-11%Al-3%Mg-0.2%Si、溶融Zn-5%Al-0.75%Mg)、合金化Znめっき鋼板(溶融0.1%Al-Znめっき後に合金化処理した合金化溶融Al-Znめっき)などが含まれる。 [Chemical conversion treatment master]
As the chemical conversion treatment original plate, an Al-containing Zn-based alloy plated steel plate having excellent corrosion resistance and design properties is used. Here, “Al-containing Zn-based alloy-plated steel sheet” means a steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al. Examples of Al-containing Zn alloy-plated steel sheets include hot-dip Al-Zn-plated steel sheets (hot Zn-0.1% Al plating, hot Zn-55% Al plating, hot Zn-6% Al-3% Mg plating, hot Zn-11% Al-3% Mg-0.2% Si, hot-dip Zn-5% Al-0.75% Mg), alloyed Zn-plated steel sheet (alloyed after hot-dip 0.1% Al-Zn plating) Alloyed molten Al—Zn plating) and the like.
化成処理原板としては、耐食性および意匠性に優れる、Al含有Zn系合金めっき鋼板が使用される。ここで「Al含有Zn系合金めっき鋼板」とは、Alを0.05~60質量%含むZn系合金めっき層を有する鋼板を意味する。Al含有Zn系合金めっき鋼板の例には、溶融Al-Znめっき鋼板(溶融Zn-0.1%Alめっき、溶融Zn-55%Alめっき、溶融Zn-6%Al-3%Mgめっき、溶融Zn-11%Al-3%Mg-0.2%Si、溶融Zn-5%Al-0.75%Mg)、合金化Znめっき鋼板(溶融0.1%Al-Znめっき後に合金化処理した合金化溶融Al-Znめっき)などが含まれる。 [Chemical conversion treatment master]
As the chemical conversion treatment original plate, an Al-containing Zn-based alloy plated steel plate having excellent corrosion resistance and design properties is used. Here, “Al-containing Zn-based alloy-plated steel sheet” means a steel sheet having a Zn-based alloy plated layer containing 0.05 to 60% by mass of Al. Examples of Al-containing Zn alloy-plated steel sheets include hot-dip Al-Zn-plated steel sheets (hot Zn-0.1% Al plating, hot Zn-55% Al plating, hot Zn-6% Al-3% Mg plating, hot Zn-11% Al-3% Mg-0.2% Si, hot-dip Zn-5% Al-0.75% Mg), alloyed Zn-plated steel sheet (alloyed after hot-dip 0.1% Al-Zn plating) Alloyed molten Al—Zn plating) and the like.
Al含有Zn系合金めっき鋼板の下地鋼としては、低炭素鋼や中炭素鋼、高炭素鋼、合金鋼などが使用される。加工性が必要とされる場合は、低炭素Ti添加鋼、低炭素Nb添加鋼などの深絞り用鋼板が下地鋼として好ましい。
As the base steel of the Al-containing Zn-based alloy plated steel sheet, low carbon steel, medium carbon steel, high carbon steel, alloy steel, or the like is used. When workability is required, steel sheets for deep drawing such as low carbon Ti-added steel and low carbon Nb-added steel are preferred as the base steel.
[下地化成処理皮膜]
化成処理原板として使用されるAl含有Zn系合金めっき鋼板の表面には、バルブメタルの酸化物または水酸化物と、バルブメタルのフッ化物とを含有する下地化成処理皮膜が形成されている場合もある。このように、Al含有Zn系合金めっき鋼板の表面に下地化成処理皮膜が形成されている場合、化成処理皮膜は、下地化成処理皮膜を介してAl含有Zn系合金めっき鋼板の表面に形成される。 [Base chemical conversion coating]
In some cases, an Al-containing Zn-based alloy-plated steel sheet used as a chemical conversion treatment original plate has a base chemical conversion treatment film containing a valve metal oxide or hydroxide and a valve metal fluoride. is there. Thus, when the base chemical conversion film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet, the chemical conversion film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet via the base chemical conversion film. .
化成処理原板として使用されるAl含有Zn系合金めっき鋼板の表面には、バルブメタルの酸化物または水酸化物と、バルブメタルのフッ化物とを含有する下地化成処理皮膜が形成されている場合もある。このように、Al含有Zn系合金めっき鋼板の表面に下地化成処理皮膜が形成されている場合、化成処理皮膜は、下地化成処理皮膜を介してAl含有Zn系合金めっき鋼板の表面に形成される。 [Base chemical conversion coating]
In some cases, an Al-containing Zn-based alloy-plated steel sheet used as a chemical conversion treatment original plate has a base chemical conversion treatment film containing a valve metal oxide or hydroxide and a valve metal fluoride. is there. Thus, when the base chemical conversion film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet, the chemical conversion film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet via the base chemical conversion film. .
バルブメタルの酸化物および水酸化物は、高い絶縁抵抗を示す。このため、バルブメタルの酸化物または水酸化物を含む下地化成処理皮膜は、電子の移動に対する抵抗体として作用する。したがって、雰囲気中の水分に含まれている溶存酸素の還元反応が抑えられ、対となるAl含有Zn系合金めっき鋼板の酸化反応も抑えられる。その結果、基材となるAl含有Zn系合金めっき鋼板からの金属成分の溶出(腐食)が抑制される。なかでも、Ti、Zr、HfなどのIV族A元素の4価化合物は安定な化合物であり、優れた高絶縁性皮膜を形成する。
¡Valve metal oxides and hydroxides exhibit high insulation resistance. For this reason, the foundation | substrate chemical conversion treatment film containing the oxide or hydroxide of valve metal acts as a resistor with respect to the movement of an electron. Therefore, the reduction reaction of dissolved oxygen contained in the moisture in the atmosphere is suppressed, and the oxidation reaction of the paired Al-containing Zn-based alloy plated steel sheet is also suppressed. As a result, elution (corrosion) of metal components from the Al-containing Zn-based alloy-plated steel sheet serving as the substrate is suppressed. Especially, the tetravalent compound of IV group A element, such as Ti, Zr, and Hf, is a stable compound, and forms the outstanding highly insulating film.
また、化成処理皮膜には、通常、化成処理時や成形加工時に、皮膜欠陥が不可避的に発生する。皮膜欠陥部では基材が露出するため、化成処理されていても、腐食抑制作用を期待できない。これに対して、前述の下地化成処理皮膜は、バルブメタルのフッ化物をも含むので、自己修復作用を有する。つまり、バルブメタルのフッ化物は、雰囲気中の水分に溶け出した後、皮膜欠陥部から露出している下地鋼の表面に難溶性酸化物または水酸化物となって再析出する。その結果、皮膜欠陥部が埋められるので、自己修復作用が発揮される。
In addition, a film defect is inevitably generated in a chemical conversion treatment film at the time of chemical conversion treatment or molding. Since the base material is exposed at the film defect portion, the corrosion inhibiting action cannot be expected even if chemical conversion treatment is performed. On the other hand, since the above-mentioned base chemical conversion treatment film also contains a fluoride of valve metal, it has a self-repairing action. That is, the fluoride of the valve metal dissolves in moisture in the atmosphere and then re-deposits as a hardly soluble oxide or hydroxide on the surface of the base steel exposed from the film defect portion. As a result, since the film defect portion is filled, a self-repairing action is exhibited.
本発明の化成処理Zn系合金めっき鋼板は、Al含有Zn系合金めっき鋼板に化成処理をすることで製造されるが、化成処理前のZn系合金めっき鋼板は、耐食性が十分でない。そのため、化成処理前のZn系合金めっき鋼板を保存または運搬したり、成形加工をしたりする間に、腐食が発生するおそれがある。そこで、化成処理前のZn系合金めっき鋼板に下地化成処理皮膜を形成しておくことで、化成処理前のZn系合金めっき鋼板における腐食の発生を確実に防止することができる。
The chemical conversion-treated Zn-based alloy-plated steel sheet of the present invention is manufactured by subjecting an Al-containing Zn-based alloy-plated steel sheet to chemical conversion treatment, but the Zn-based alloy-plated steel sheet before chemical conversion treatment has insufficient corrosion resistance. Therefore, corrosion may occur during storage or transportation of the Zn-based alloy-plated steel sheet before chemical conversion treatment or during forming. Therefore, by forming a base chemical conversion coating on the Zn-based alloy plated steel sheet before the chemical conversion treatment, it is possible to reliably prevent the occurrence of corrosion in the Zn-based alloy plated steel sheet before the chemical conversion treatment.
下地化成処理皮膜を形成したAl含有Zn系合金めっき鋼板を、保存、運搬または成形加工(溶接などを含む)したときに、一部の下地化成処理皮膜が剥離、欠損または欠落することがある。それにより、Al含有Zn系合金めっき鋼板の表面が露出するが、この露出面には、フッ素含有樹脂および4A族金属化合物を含む化成処理皮膜が直接接触する。
When the Al-containing Zn-based alloy-plated steel sheet on which the base chemical conversion treatment film is formed is stored, transported or formed (including welding), a part of the base chemical conversion treatment film may be peeled off, missing, or missing. Thereby, the surface of the Al-containing Zn-based alloy-plated steel sheet is exposed, and the chemical conversion treatment film containing the fluorine-containing resin and the 4A group metal compound is in direct contact with the exposed surface.
下地化成処理皮膜は、Al含有Zn系合金めっき鋼板と、化成処理皮膜との密着性を向上させる作用を示す。このため、下地化成処理皮膜が剥離した箇所に形成された化成処理皮膜は、一般的にはその皮膜密着性が低下するとも思われる。しかしながら、化成処理皮膜には、直接接触するめっき層からAlが溶出している。この化成処理皮膜に溶出したAlによって、化成処理皮膜の耐食性および皮膜密着性が向上する。このように、下地化成処理皮膜が剥離した箇所においても、化成処理皮膜とAl含有Zn系合金めっき鋼板との密着性が高く、化成処理皮膜の耐食性が高まる。化成処理皮膜中にAlが存在することによって、化成処理皮膜の耐食性および皮膜密着性が向上するメカニズムについては後述する。
The base chemical conversion treatment film exhibits an effect of improving the adhesion between the Al-containing Zn-based alloy-plated steel sheet and the chemical conversion treatment film. For this reason, the chemical conversion treatment film formed at the place where the base chemical conversion treatment film is peeled is generally considered to have a reduced film adhesion. However, Al is eluted from the plating layer in direct contact with the chemical conversion coating. The corrosion resistance and film adhesion of the chemical conversion film are improved by Al eluted in the chemical conversion film. Thus, also in the location where the foundation | substrate chemical conversion treatment film peeled, the adhesiveness of a chemical conversion treatment film and an Al containing Zn type alloy plating steel plate is high, and the corrosion resistance of a chemical conversion treatment film increases. The mechanism by which the presence of Al in the chemical conversion film improves the corrosion resistance and film adhesion of the chemical conversion film will be described later.
下地化成処理皮膜は、基材となるAl含有Zn系合金めっき鋼板の表面に形成された下地化成処理液の塗布膜を、乾燥させることにより形成されうる。下地化成処理液には、バルブメタル塩と、フッ化物イオンと、溶媒である水とが含まれる。下地化成処理液の塗膜を乾燥させることで、バルブメタル塩が、下地化成処理皮膜に含まれるバルブメタルの酸化物もしくは水酸化物、またはフッ化物となる。
The base chemical conversion treatment film can be formed by drying a coating film of the base chemical conversion treatment liquid formed on the surface of the Al-containing Zn-based alloy-plated steel sheet as the base material. The base chemical conversion treatment solution contains valve metal salt, fluoride ions, and water as a solvent. By drying the coating film of the base chemical conversion treatment solution, the valve metal salt becomes an oxide or hydroxide or a fluoride of valve metal contained in the base chemical conversion coating film.
バルブメタルの例には、Ti、Zr、Hf、V、Nb、Ta、Mo、およびWが含まれる。下地化成処理液に添加されるバルブメタル塩は、バルブメタルのハロゲン化物や酸素酸塩などでありうる。添加されるバルブメタル塩がフッ化物であれば、フッ化物イオン源としても作用する。
Examples of valve metals include Ti, Zr, Hf, V, Nb, Ta, Mo, and W. The valve metal salt added to the base chemical conversion treatment liquid may be a valve metal halide or oxyacid salt. If the added valve metal salt is fluoride, it also acts as a fluoride ion source.
チタン塩の例には、KnTiF6(K:アルカリ金属またはアルカリ土類金属、n:1または2)、K2[TiO(COO)2]、(NH4)2TiF6、TiCl4、TiOSO4、Ti(SO4)2、およびTi(OH)4などが含まれる。一方、下地化成処理液に含まれるフッ化物イオン源は、フッ素原子を含むバルブメタル塩であってもよいし、可溶性フッ化物(例えば、(NH4)Fなど)であってもよい。
Examples of titanium salts include K n TiF 6 (K: alkali metal or alkaline earth metal, n: 1 or 2), K 2 [TiO (COO) 2 ], (NH 4 ) 2 TiF 6 , TiCl 4 , TiOSO 4 , Ti (SO 4 ) 2 , Ti (OH) 4 and the like are included. On the other hand, the fluoride ion source contained in the base chemical conversion treatment liquid may be a valve metal salt containing a fluorine atom or a soluble fluoride (for example, (NH 4 ) F).
下地化成処理液には、バルブメタル塩を安定化するために、キレート作用のある有機酸が添加されていることが好ましい。有機酸は、金属イオンをキレート化して化成処理液を安定させることができる。そのため、有機酸の添加量は、有機酸/金属イオンのモル比が、0.02以上となるように設定される。有機酸の例には、酒石酸、タンニン酸、クエン酸、蓚酸、マロン酸、乳酸、酢酸、およびアスコルビン酸などが含まれる。なかでも、酒石酸などのオキシカルボン酸や、タンニン酸などの多価フェノール類は、下地化成処理液を安定化させるとともに、フッ化物の自己修復作用を補完する作用も示し、密着性の向上にも有効である。
It is preferable that an organic acid having a chelating action is added to the base chemical conversion treatment solution in order to stabilize the valve metal salt. The organic acid can chelate metal ions and stabilize the chemical conversion solution. Therefore, the addition amount of the organic acid is set so that the molar ratio of organic acid / metal ion is 0.02 or more. Examples of the organic acid include tartaric acid, tannic acid, citric acid, succinic acid, malonic acid, lactic acid, acetic acid, ascorbic acid and the like. Among them, oxycarboxylic acids such as tartaric acid and polyhydric phenols such as tannic acid stabilize the base chemical conversion solution and also complement the fluoride's self-healing action, improving adhesion. It is valid.
下地化成処理液には、各種金属のオルソリン酸塩またはポリリン酸塩が添加されていてもよい。形成される下地化成処理皮膜に、可溶性または難溶性の、金属リン酸塩または複合リン酸塩を含ませるためである。
Various metal orthophosphates or polyphosphates may be added to the base chemical conversion treatment solution. This is because the base chemical conversion treatment film to be formed contains a soluble or hardly soluble metal phosphate or composite phosphate.
可溶性の金属リン酸塩または複合リン酸塩は、下地化成処理皮膜から皮膜欠陥部に溶出して、基材であるAl含有Zn系合金めっき鋼板のめっき成分(ZnやAlなど)と反応して、不溶性リン酸塩を析出させる。このようにして、チタンフッ化物の自己修復作用を補完する。また、可溶性リン酸塩が解離する際に、雰囲気が若干酸性化するため、チタンフッ化物の加水分解、ひいては難溶性チタン酸化物または水酸化物の生成が促進される。
Soluble metal phosphate or composite phosphate elutes from the base chemical conversion coating to the film defects and reacts with the plating components (Zn, Al, etc.) of the Al-containing Zn-based alloy-plated steel sheet as the substrate. Insoluble phosphate is precipitated. In this way, the self-healing action of titanium fluoride is complemented. Further, since the atmosphere is slightly acidified when the soluble phosphate is dissociated, the hydrolysis of titanium fluoride, and hence the generation of hardly soluble titanium oxide or hydroxide, is promoted.
可溶性リン酸塩または複合リン酸塩の金属は、アルカリ金属やアルカリ土類金属、Mnなどでありうる。可溶性リン酸塩または複合リン酸塩は、各種金属リン酸塩の形態で下地化成処理液に添加されてもよいし、各種金属塩と、リン酸、ポリリン酸またはリン酸塩とを組み合わせて化成処理液に添加されてもよい。
The metal of the soluble phosphate or the composite phosphate can be an alkali metal, an alkaline earth metal, Mn, or the like. The soluble phosphate or the composite phosphate may be added to the base chemical conversion treatment liquid in the form of various metal phosphates, or may be formed by combining various metal salts with phosphoric acid, polyphosphoric acid or phosphate. It may be added to the treatment liquid.
一方、難溶性の金属リン酸塩または複合リン酸塩は、下地化成処理皮膜に分散して、皮膜欠陥を解消するとともに皮膜強度を向上させる。難溶性リン酸塩または複合リン酸塩の金属は、Al、Ti、Zr、Hf、Znなどでありうる。難溶性リン酸塩または複合リン酸塩は、各種金属リン酸塩の形態で化成処理液に添加されてもよいし、各種金属塩とリン酸、ポリリン酸またはリン酸塩とを組み合わせて化成処理液に添加されてもよい。
On the other hand, poorly soluble metal phosphates or composite phosphates are dispersed in the base chemical conversion treatment film to eliminate film defects and improve the film strength. The metal of the poorly soluble phosphate or the composite phosphate may be Al, Ti, Zr, Hf, Zn or the like. The hardly soluble phosphate or the composite phosphate may be added to the chemical conversion treatment liquid in the form of various metal phosphates, or a chemical conversion treatment by combining various metal salts with phosphoric acid, polyphosphoric acid or phosphate. It may be added to the liquid.
下地化成処理液には、フッ素系、ポリエチレン系、スチレン系などの有機ワックスや、シリカ、二硫化モリブデン、タルクなどの無機質潤滑剤などを添加することもできる。これらを添加することで、下地化成処理皮膜の潤滑性を向上させることができる。低融点の有機ワックスは、下地化成処理液の塗布膜を乾燥させるときに、膜表面にブリードし、潤滑性を発現すると考えられる。一方、高融点の有機ワックスや無機系潤滑剤は、皮膜中に分散して存在するが、皮膜の最表層では島状分布で皮膜表面に露出することによって潤滑性を発現させるものと考えられる。
In the base chemical conversion treatment liquid, organic waxes such as fluorine, polyethylene, and styrene, inorganic lubricants such as silica, molybdenum disulfide, and talc can be added. By adding these, the lubricity of the base chemical conversion coating can be improved. It is considered that the low melting point organic wax bleeds on the film surface and develops lubricity when the coating film of the base chemical conversion treatment liquid is dried. On the other hand, high-melting organic waxes and inorganic lubricants are present in a dispersed state in the film, but it is considered that the outermost surface layer of the film is exposed to the surface of the film in an island-like distribution to exhibit lubricity.
下地化成処理液の塗布膜を乾燥させて得た下地化成処理皮膜を、蛍光X線、ESCAなどで元素分析すると、下地化成処理皮膜に含まれているOおよびFの濃度が測定される。これらの元素の濃度比F/O(原子比率)は、1/100以上であることが好ましい。得られた化成処理鋼板の腐食を抑制するためである。特に、元素濃度比F/O(原子比率)が1/100以上であると、皮膜欠陥部を起点とする腐食の発生が大幅に減少する。これは、十分な量のチタンフッ化物が下地化成処理皮膜中に含まれており、自己修復作用を発揮しているためと推察される。
When the base chemical conversion film obtained by drying the coating film of the base chemical conversion liquid is subjected to elemental analysis using fluorescent X-ray, ESCA or the like, the concentrations of O and F contained in the base chemical conversion film are measured. The concentration ratio F / O (atomic ratio) of these elements is preferably 1/100 or more. It is for suppressing the corrosion of the obtained chemical conversion treatment steel plate. In particular, when the element concentration ratio F / O (atomic ratio) is 1/100 or more, the occurrence of corrosion starting from a film defect portion is significantly reduced. This is presumably because a sufficient amount of titanium fluoride is contained in the base chemical conversion treatment film and exhibits a self-repairing action.
[化成処理皮膜]
化成処理皮膜は、上述のAl含有Zn系合金めっき鋼板(化成処理原板)の表面に形成されている。化成処理原板の表面には、下地となる皮膜を形成するなどの下地化成処理をしてもよいが、下地化成処理をしなくてもよい。なお、化成処理原板の表面に下地化成処理をしない場合には、化成処理原板の表面には化成処理皮膜が直接形成される。この化成処理皮膜は、Al含有Zn系合金めっき鋼板の耐候性や耐黒変性、加工性(潤滑性)などを向上させる。 [Chemical conversion coating]
The chemical conversion treatment film is formed on the surface of the above-mentioned Al-containing Zn-based alloy plated steel sheet (chemical conversion treatment original plate). The surface of the chemical conversion treatment original plate may be subjected to a base chemical conversion treatment such as forming a coating as a base, but the base chemical conversion treatment may not be performed. In addition, when the surface chemical conversion treatment is not performed on the surface of the chemical conversion treatment original plate, a chemical conversion treatment film is directly formed on the surface of the chemical conversion treatment original plate. This chemical conversion treatment film improves the weather resistance, blackening resistance, workability (lubricity), and the like of the Al-containing Zn-based alloy plated steel sheet.
化成処理皮膜は、上述のAl含有Zn系合金めっき鋼板(化成処理原板)の表面に形成されている。化成処理原板の表面には、下地となる皮膜を形成するなどの下地化成処理をしてもよいが、下地化成処理をしなくてもよい。なお、化成処理原板の表面に下地化成処理をしない場合には、化成処理原板の表面には化成処理皮膜が直接形成される。この化成処理皮膜は、Al含有Zn系合金めっき鋼板の耐候性や耐黒変性、加工性(潤滑性)などを向上させる。 [Chemical conversion coating]
The chemical conversion treatment film is formed on the surface of the above-mentioned Al-containing Zn-based alloy plated steel sheet (chemical conversion treatment original plate). The surface of the chemical conversion treatment original plate may be subjected to a base chemical conversion treatment such as forming a coating as a base, but the base chemical conversion treatment may not be performed. In addition, when the surface chemical conversion treatment is not performed on the surface of the chemical conversion treatment original plate, a chemical conversion treatment film is directly formed on the surface of the chemical conversion treatment original plate. This chemical conversion treatment film improves the weather resistance, blackening resistance, workability (lubricity), and the like of the Al-containing Zn-based alloy plated steel sheet.
本発明は、化成処理Zn系めっき鋼板の耐候性、耐水性、耐黒変性、皮膜密着性および加工性のすべてを向上させることを目的としている。前述の通り、化成処理皮膜の耐候性(耐紫外線性)を向上させるためには、有機樹脂としてフッ素含有樹脂を使用すればよい。フッ素含有樹脂は、溶剤系フッ素含有樹脂と水系フッ素含有樹脂に大別される。溶剤系フッ素含有樹脂を用いて化成処理皮膜を形成する場合は、揮発した溶剤の回収が問題となるが、水系フッ素含有樹脂を用いた場合は、このような問題は生じない。そこで、本発明者は、取り扱いが容易な水系フッ素含有樹脂を使用して耐候性、耐水性、耐黒変性、皮膜密着性および加工性のすべてに優れた化成処理Zn系めっき鋼板を形成することを試みた。
The object of the present invention is to improve all of the weather resistance, water resistance, blackening resistance, film adhesion and workability of the chemically treated Zn-based plated steel sheet. As described above, in order to improve the weather resistance (ultraviolet light resistance) of the chemical conversion coating, a fluorine-containing resin may be used as the organic resin. Fluorine-containing resins are roughly classified into solvent-based fluorine-containing resins and water-based fluorine-containing resins. When a chemical conversion film is formed using a solvent-based fluorine-containing resin, recovery of the volatilized solvent becomes a problem, but when a water-based fluorine-containing resin is used, such a problem does not occur. Therefore, the present inventor uses a water-based fluorine-containing resin that is easy to handle to form a chemically treated Zn-based plated steel sheet that is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion, and workability. Tried.
前述の通り、本発明者の予備実験によれば、水系フッ素含有樹脂のエマルションを用いて化成処理皮膜を形成した場合に耐水性が低下するのは、水系フッ素含有樹脂のエマルションを製造する際に使用される乳化剤が化成処理皮膜中に残存するためと考えられた(後述の参考実験参照)。そこで、本発明者は、乳化剤をほとんど使用せずに水系フッ素含有樹脂のエマルションを製造することができれば、化成処理皮膜の耐水性の低下を抑制できると考えた。そして、本発明者は、様々な水系フッ素含有樹脂について検討した結果、一定量の親水性官能基を導入したフッ素含有樹脂は、乳化剤をほとんど使用せずに水系エマルションを製造することができ、乳化剤をほとんど含まない化成処理皮膜を容易に形成できることを見出した。
As described above, according to the preliminary experiment of the present inventor, when a chemical conversion film is formed using an emulsion of a water-based fluorine-containing resin, the water resistance decreases when an emulsion of a water-based fluorine-containing resin is produced. It was considered that the emulsifier used remained in the chemical conversion film (see the reference experiment described later). Then, this inventor thought that the fall of the water resistance of a chemical conversion treatment film could be suppressed if the emulsion of water-type fluorine-containing resin could be manufactured, using few emulsifiers. And as a result of studying various water-based fluorine-containing resins, the present inventor is able to produce water-based emulsions with little use of emulsifiers. It has been found that a chemical conversion treatment film containing almost no can be easily formed.
また、本発明者は、化成処理皮膜の耐水性の低下を抑制するだけでなく、耐水性を向上させることについても検討した。そして、様々な観点から検討した結果、水系フッ素含有樹脂の分子量を大きくし、かつ水系フッ素樹脂を4A族金属化合物で架橋することで、化成処理皮膜の耐水性を顕著に向上させうることを見出した。
In addition, the present inventor examined not only suppressing the decrease in water resistance of the chemical conversion coating but also improving the water resistance. As a result of studying from various viewpoints, it was found that the water resistance of the chemical conversion coating can be remarkably improved by increasing the molecular weight of the aqueous fluorine-containing resin and crosslinking the aqueous fluorine resin with a group 4A metal compound. It was.
そして、本発明者は、親水性官能基を導入した高分子量のフッ素含有樹脂をベースとする化成処理液に、さらに4A族金属化合物を配合することで、耐候性と、耐水性、耐黒変性および皮膜密着性とを両立した化成処理皮膜を形成できることを見出したのである。
And this inventor is adding a 4A group metal compound to the chemical conversion treatment liquid based on the high molecular weight fluorine-containing resin which introduce | transduced the hydrophilic functional group, and is weather resistance, water resistance, and blackening resistance. It was also found that a chemical conversion treatment film compatible with film adhesion can be formed.
さらに、本発明者は、化成処理Zn系めっき鋼板の加工性(潤滑性)を向上させることについて検討した。そして、本発明者は、様々な手段について検討した結果、平均粒子径が0.1~10μmの樹脂粒子を化成処理皮膜中に分散させることで、耐候性、耐水性、耐黒変性および皮膜密着性に加えて、さらに加工性(潤滑性)にも優れた化成処理皮膜を形成できることを見出したのである。
Furthermore, the present inventor has studied to improve the workability (lubricity) of the chemical conversion treatment Zn-plated steel sheet. As a result of studying various means, the present inventor disperses resin particles having an average particle diameter of 0.1 to 10 μm in the chemical conversion film, thereby improving weather resistance, water resistance, blackening resistance, and film adhesion. In addition to the properties, it has been found that a chemical conversion film excellent in workability (lubricity) can be formed.
本発明の化成処理Zn系めっき鋼板の化成処理皮膜では、1)フッ素含有樹脂(好ましくはフッ素含有オレフィン樹脂)を配合することで、耐候性(耐紫外線性)を向上させている。また、2)親水性官能基を導入したフッ素含有樹脂を使用することでエマルション製造時の乳化剤の使用を極力減らし、かつ3)フッ素含有樹脂の分子量を大きくし、かつ4)フッ素含有樹脂を4A族金属化合物で架橋させることで、耐候性(耐紫外線性)および耐水性を向上させている。また、5)平均粒子径が0.1~10μmの樹脂粒子を分散させることで加工性(潤滑性)を向上させている。
In the chemical conversion treatment film of the chemical conversion treatment Zn-plated steel sheet of the present invention, 1) weather resistance (ultraviolet resistance) is improved by blending a fluorine-containing resin (preferably a fluorine-containing olefin resin). In addition, 2) the use of a fluorine-containing resin into which a hydrophilic functional group has been introduced reduces the use of an emulsifier during emulsion production, 3) the molecular weight of the fluorine-containing resin is increased, and 4) the fluorine-containing resin is 4A. By cross-linking with a group metal compound, weather resistance (ultraviolet light resistance) and water resistance are improved. 5) Workability (lubricity) is improved by dispersing resin particles having an average particle diameter of 0.1 to 10 μm.
以下、化成処理皮膜に含まれる各成分について説明する。
Hereinafter, each component contained in the chemical conversion coating will be described.
1)水系フッ素含有樹脂
化成処理皮膜は、フッ素含有樹脂、より具体的にはフッ素含有オレフィン樹脂を主成分として含む。化成処理皮膜に主成分として含まれるフッ素含有樹脂の量は、70~99質量%の範囲内が好ましい。前述の通り、化成処理皮膜を構成する有機樹脂としてフッ素含有樹脂を用いることで、化成処理皮膜の耐候性(耐紫外線性)を向上させることができる。 1) Water-based fluorine-containing resin The chemical conversion film contains a fluorine-containing resin, more specifically, a fluorine-containing olefin resin as a main component. The amount of the fluorine-containing resin contained as a main component in the chemical conversion film is preferably in the range of 70 to 99% by mass. As described above, by using a fluorine-containing resin as the organic resin constituting the chemical conversion coating, the weather resistance (ultraviolet resistance) of the chemical conversion coating can be improved.
化成処理皮膜は、フッ素含有樹脂、より具体的にはフッ素含有オレフィン樹脂を主成分として含む。化成処理皮膜に主成分として含まれるフッ素含有樹脂の量は、70~99質量%の範囲内が好ましい。前述の通り、化成処理皮膜を構成する有機樹脂としてフッ素含有樹脂を用いることで、化成処理皮膜の耐候性(耐紫外線性)を向上させることができる。 1) Water-based fluorine-containing resin The chemical conversion film contains a fluorine-containing resin, more specifically, a fluorine-containing olefin resin as a main component. The amount of the fluorine-containing resin contained as a main component in the chemical conversion film is preferably in the range of 70 to 99% by mass. As described above, by using a fluorine-containing resin as the organic resin constituting the chemical conversion coating, the weather resistance (ultraviolet resistance) of the chemical conversion coating can be improved.
フッ素含有樹脂は、有機溶剤系フッ素樹脂よりも、取り扱いが容易な水系フッ素含有樹脂であることが好ましい。「水系フッ素含有樹脂」とは、親水性官能基を有するフッ素含有樹脂をいう。親水性官能基の好ましい例には、カルボキシル基、スルホン酸基およびこれらの塩が含まれる。カルボキシル基またはスルホン酸基の塩の例としては、アンモニウム塩、アミン塩、アルカリ金属塩などが含まれる。
The fluorine-containing resin is preferably a water-based fluorine-containing resin that is easier to handle than the organic solvent-based fluorine resin. “Aqueous fluorine-containing resin” refers to a fluorine-containing resin having a hydrophilic functional group. Preferred examples of the hydrophilic functional group include a carboxyl group, a sulfonic acid group, and salts thereof. Examples of the salt of the carboxyl group or sulfonic acid group include ammonium salt, amine salt, alkali metal salt and the like.
好ましい水系フッ素含有樹脂(好ましくはフッ素含有オレフィン樹脂)は、親水性官能基を0.05~5質量%有する。親水性官能基を0.05~5質量%有するフッ素含有樹脂は、乳化剤をほとんど使用せずとも、水系エマルションとすることができる。乳化剤をほとんど含まない化成処理皮膜は、耐水性に優れた化成処理皮膜とすることができる。
A preferred aqueous fluorine-containing resin (preferably a fluorine-containing olefin resin) has a hydrophilic functional group of 0.05 to 5% by mass. A fluorine-containing resin having 0.05 to 5% by mass of a hydrophilic functional group can be made into an aqueous emulsion with little use of an emulsifier. The chemical conversion film containing almost no emulsifier can be a chemical conversion film excellent in water resistance.
水系フッ素含有樹脂中の親水性官能基の含有量は、水系フッ素含有樹脂に含まれる親水性官能基の総モル質量を、水系フッ素含有樹脂の数平均分子量で除して求めればよい。カルボキシル基のモル質量は45であり、スルホン酸基のモル質量は81であるので、水系フッ素含有樹脂に含まれるカルボキシル基およびスルホン酸基それぞれの数を求め、それぞれにモル質量を乗じることで、水系フッ素含有樹脂に含まれる親水性官能基の総モル質量が求まる。水系フッ素含有樹脂の数平均分子量はGPCで測定される。
The content of the hydrophilic functional group in the aqueous fluorine-containing resin may be obtained by dividing the total molar mass of the hydrophilic functional group contained in the aqueous fluorine-containing resin by the number average molecular weight of the aqueous fluorine-containing resin. Since the molar mass of the carboxyl group is 45 and the molar mass of the sulfonic acid group is 81, the number of each of the carboxyl group and sulfonic acid group contained in the aqueous fluorine-containing resin is determined, and each is multiplied by the molar mass. The total molar mass of the hydrophilic functional group contained in the aqueous fluorine-containing resin is obtained. The number average molecular weight of the water-based fluorine-containing resin is measured by GPC.
水系フッ素含有樹脂におけるカルボキシル基は、めっき層表面と水素結合などを形成して化成処理皮膜とめっき層表面との密着性の向上に寄与するが、H+が解離しにくいため4A族金属化合物との架橋反応が生じにくい。一方、水系フッ素含有樹脂におけるスルホン酸基は、H+が解離しやすいものの、4A族金属化合物と架橋反応せずに未反応のまま皮膜中に残存すると、水分子の吸着作用が強いため皮膜の耐水性を著しく低下させてしまうおそれがある。したがって、それぞれの特徴を活かすべく、水系フッ素含有樹脂には、カルボキシル基およびスルホン酸基の両方を含むことが好ましい。この場合、カルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内が好ましい。
The carboxyl group in the water-based fluorine-containing resin forms a hydrogen bond with the plating layer surface and contributes to improving the adhesion between the chemical conversion film and the plating layer surface. However, since H + is difficult to dissociate, The cross-linking reaction is unlikely to occur. On the other hand, the sulfonic acid group in the water-based fluorine-containing resin tends to dissociate H +, but if it remains unreacted in the film without cross-linking reaction with the group 4A metal compound, the water molecule adsorbing action is strong, so There is a risk that the water resistance will be significantly reduced. Therefore, it is preferable that the water-based fluorine-containing resin includes both a carboxyl group and a sulfonic acid group in order to make use of each feature. In this case, the ratio of carboxyl group to sulfonic acid group is preferably in the range of 5 to 60 in terms of the molar ratio of carboxyl group / sulfonic acid group.
化成処理皮膜に含まれる水系フッ素含有樹脂(好ましくはフッ素含有オレフィン樹脂)の数平均分子量は、1000以上が好ましく、1万以上がより好ましく、20万以上が特に好ましい。
The number average molecular weight of the water-based fluorine-containing resin (preferably fluorine-containing olefin resin) contained in the chemical conversion film is preferably 1000 or more, more preferably 10,000 or more, and particularly preferably 200,000 or more.
化成処理皮膜に含まれる水系フッ素含有樹脂の分子量が小さすぎると、化成処理皮膜の透水性および耐水性を十分に向上させることができない。このような場合、湿気や腐食性ガスなどが化成処理皮膜を容易に貫通してめっき鋼板に達するため、めっき鋼板は容易に腐食してしまうおそれがある。また、分子量が小さい水系フッ素含有樹脂を使用した場合は、光エネルギーなどの作用により発生したラジカルがポリマー鎖の末端に作用しやすいため、水などの相乗作用により水系フッ素含有樹脂が容易に加水分解されてしまうおそれがある。これらの問題を防ぐためには、化成処理皮膜に含まれる水系フッ素含有樹脂の分子量をある程度大きくしたり、水系フッ素含有樹脂間に架橋構造を形成したりすればよい。水系フッ素含有樹脂の分子量を大きくすることにより、分子間力が強くなり、化成処理皮膜の凝集力が高まるため、耐水性が向上する。また、水系フッ素含有樹脂の主鎖における原子間の結合が安定化されるため、加水分解も生じにくくなる。
If the molecular weight of the water-based fluorine-containing resin contained in the chemical conversion coating is too small, the water permeability and water resistance of the chemical conversion coating cannot be sufficiently improved. In such a case, moisture or corrosive gas easily penetrates the chemical conversion coating and reaches the plated steel sheet, so that the plated steel sheet may be easily corroded. In addition, when an aqueous fluorine-containing resin with a low molecular weight is used, radicals generated by the action of light energy or the like are likely to act on the end of the polymer chain, so the aqueous fluorine-containing resin is easily hydrolyzed by a synergistic action such as water. There is a risk of being. In order to prevent these problems, the molecular weight of the aqueous fluorine-containing resin contained in the chemical conversion coating may be increased to some extent, or a crosslinked structure may be formed between the aqueous fluorine-containing resins. By increasing the molecular weight of the water-based fluorine-containing resin, the intermolecular force increases and the cohesive strength of the chemical conversion coating increases, so that the water resistance is improved. Moreover, since the bond between atoms in the main chain of the water-based fluorine-containing resin is stabilized, hydrolysis is less likely to occur.
一方で、化成処理皮膜に含まれる水系フッ素含有樹脂の数平均分子量は、200万以下が好ましい。数平均分子量が200万超の場合、ゲル化などの処理液安定性に問題が生じるおそれがある。
On the other hand, the number average molecular weight of the water-based fluorine-containing resin contained in the chemical conversion coating is preferably 2 million or less. When the number average molecular weight exceeds 2 million, there is a possibility that a problem may occur in the stability of the treatment liquid such as gelation.
化成処理皮膜に含まれる水系フッ素含有樹脂中のF原子の含有量は、7~20質量%の範囲内が好ましい。F原子の含有量が7質量%未満の場合、化成処理皮膜の耐候性を十分に向上させることができない。一方、F原子の含有量が20質量%超の場合、塗料化が困難であり、かつ密着性および乾燥性が低下するおそれがある。水系フッ素含有樹脂中のF原子の含有量は、蛍光X線分析装置を用いることで測定することができる。
The content of F atoms in the aqueous fluorine-containing resin contained in the chemical conversion coating is preferably within the range of 7 to 20% by mass. When content of F atom is less than 7 mass%, the weather resistance of a chemical conversion treatment film cannot fully be improved. On the other hand, when the content of F atoms is more than 20% by mass, it is difficult to form a paint and the adhesion and drying properties may be reduced. The content of F atoms in the aqueous fluorine-containing resin can be measured by using a fluorescent X-ray analyzer.
水系フッ素含有樹脂の例には、フルオロオレフィンと親水性官能基含有モノマーとの共重合体が含まれる。親水性官能基含有モノマーとは、カルボキシル基含有モノマーやスルホン酸基含有モノマーである。
Examples of the water-based fluorine-containing resin include a copolymer of a fluoroolefin and a hydrophilic functional group-containing monomer. The hydrophilic functional group-containing monomer is a carboxyl group-containing monomer or a sulfonic acid group-containing monomer.
フルオロオレフィンの例には、テトラフルオロエチレン、トリフルオロエチレン、クロロトリフルオロエチレン、ヘキサフルオロプロピレン、フッ化ビニル、フッ化ビニリデン、ペンタフルオロプロピレン、2,2,3,3-テトラフルオロプロピレン、3,3,3-トリフルオロプロピレン、ブロモトリフルオロエチレン、1-クロロ-1,2-ジフルオロエチレン、1,1-ジクロロ-2,2-ジフルオロエチレンなどが含まれる。これらのフルオロオレフィンは、単独で使用されてもよいし、2種類以上を組み合わせて使用されてもよい。耐候性(耐紫外線性)の観点からは、これらのフルオロオレフィンの中でも、テトラフルオロエチレン、ヘキサフルオロプロピレンなどのパーフルオロオレフィンや、フッ化ビニリデンなどが好ましい。クロロトリフルオロエチレンなどの塩素を含むフルオロオレフィンは、塩素イオンによる腐食が生じるおそれがあるため好ましくない。
Examples of fluoroolefins include tetrafluoroethylene, trifluoroethylene, chlorotrifluoroethylene, hexafluoropropylene, vinyl fluoride, vinylidene fluoride, pentafluoropropylene, 2,2,3,3-tetrafluoropropylene, 3, 3,3-trifluoropropylene, bromotrifluoroethylene, 1-chloro-1,2-difluoroethylene, 1,1-dichloro-2,2-difluoroethylene and the like are included. These fluoroolefins may be used alone or in combination of two or more. From the viewpoint of weather resistance (ultraviolet light resistance), among these fluoroolefins, perfluoroolefins such as tetrafluoroethylene and hexafluoropropylene, and vinylidene fluoride are preferable. Fluoroolefins containing chlorine such as chlorotrifluoroethylene are not preferred because corrosion due to chlorine ions may occur.
カルボキシル基含有モノマーの一例には、以下の式(1)に示される不飽和カルボン酸や、これらのエステルまたは酸無水物などの不飽和カルボン酸類が含まれる。
(式中、R1、R2およびR3は同じかまたは異なり、いずれも水素原子、アルキル基、カルボキシル基またはエステル基である。nは0~20の範囲内である。)
Examples of the carboxyl group-containing monomer include unsaturated carboxylic acids represented by the following formula (1) and unsaturated carboxylic acids such as esters or acid anhydrides thereof.
(In the formula, R 1 , R 2 and R 3 are the same or different, and each is a hydrogen atom, an alkyl group, a carboxyl group or an ester group. N is in the range of 0-20.)
上記式(1)に示される不飽和カルボン酸の例には、アクリル酸、メタクリル酸、ビニル酢酸、クロトン酸、桂皮酸、イタコン酸、イタコン酸モノエステル、マレイン酸、マレイン酸モノエステル、フマル酸、フマル酸モノエステル、5-ヘキセン酸、5-ヘプテン酸、6-ヘプテン酸、7-オクテン酸、8-ノネン酸、9-デセン酸、10-ウンデシレン酸、11-ドデシレン酸、17-オクタデシレン酸、オレイン酸などが含まれる。
Examples of the unsaturated carboxylic acid represented by the above formula (1) include acrylic acid, methacrylic acid, vinyl acetic acid, crotonic acid, cinnamic acid, itaconic acid, itaconic acid monoester, maleic acid, maleic acid monoester, fumaric acid , Fumaric acid monoester, 5-hexenoic acid, 5-heptenoic acid, 6-heptenoic acid, 7-octenoic acid, 8-nonenoic acid, 9-decenoic acid, 10-undecylene acid, 11-dodecylene acid, 17-octadecylenic acid Oleic acid and the like.
カルボキシル基含有モノマーの別の例には、以下の式(2)に示されるカルボキシル基含有ビニルエーテルモノマーが含まれる。
(式中、R4およびR5は同じかまたは異なり、いずれも飽和または不飽和の直鎖または環状アルキル基である。nは0または1である。mは0または1である。)
Another example of the carboxyl group-containing monomer includes a carboxyl group-containing vinyl ether monomer represented by the following formula (2).
(Wherein R 4 and R 5 are the same or different, and each is a saturated or unsaturated linear or cyclic alkyl group. N is 0 or 1. m is 0 or 1.)
上記式(2)に示されるカルボキシル基含有ビニルエーテルモノマーの例には、3-(2-アリロキシエトキシカルボニル)プロピオン酸、3-(2-アリロキシブトキシカルボニル)プロピオン酸、3-(2-ビニロキシエトキシカルボニル)プロピオン酸、3-(2-ビニロキシブトキシカルボニル)プロピオン酸などが含まれる。
Examples of the carboxyl group-containing vinyl ether monomer represented by the above formula (2) include 3- (2-allyloxyethoxycarbonyl) propionic acid, 3- (2-allyloxybutoxycarbonyl) propionic acid, 3- (2-vinylidene). Roxyethoxycarbonyl) propionic acid, 3- (2-vinyloxybutoxycarbonyl) propionic acid and the like.
スルホン酸基含有モノマーの例には、ビニルスルホン酸、アリルスルホン酸、メタリルスルホン酸、スチレンスルホン酸、2-アクリルアミド-2-メチルプロパンスルホン酸、2-メタクリロイルオキシエタンスルホン酸、3-メタクリロイルオキシプロパンスルホン酸、4-メタクリロイルオキシブタンスルホン酸、3-メタクリロイルオキシ-2-ヒドロキシプロパンスルホン酸、3-アクリロイルオキシプロパンスルホン酸、アリルオキシベンゼンスルホイン酸、メタリルオキシベンゼンスルホン酸、イソプレンスルホン酸、3-アリロキシ-2-ヒドロキシプロパンスルホン酸などが含まれる。
Examples of sulfonic acid group-containing monomers include vinyl sulfonic acid, allyl sulfonic acid, methallyl sulfonic acid, styrene sulfonic acid, 2-acrylamido-2-methylpropane sulfonic acid, 2-methacryloyloxyethane sulfonic acid, and 3-methacryloyloxy. Propanesulfonic acid, 4-methacryloyloxybutanesulfonic acid, 3-methacryloyloxy-2-hydroxypropanesulfonic acid, 3-acryloyloxypropanesulfonic acid, allyloxybenzenesulfonic acid, methallyloxybenzenesulfonic acid, isoprenesulfonic acid, 3-allyloxy-2-hydroxypropane sulfonic acid and the like are included.
フルオロオレフィンと親水性官能基含有モノマーとの共重合体には、必要に応じてさらに共重合可能な他のモノマーを共重合されていてもよい。共重合可能な他のモノマーの例には、カルボン酸ビニルエステル類、アルキルビニルエーテル類、非フッ素系オレフィン類などが含まれる。
The copolymer of the fluoroolefin and the hydrophilic functional group-containing monomer may be copolymerized with another copolymerizable monomer as necessary. Examples of other copolymerizable monomers include carboxylic acid vinyl esters, alkyl vinyl ethers, non-fluorinated olefins, and the like.
カルボン酸ビニルエステル類は、相溶性および光沢を向上させたり、ガラス転移温度を上昇させたりすることができる。カルボン酸ビニルエステル類の例には、酢酸ビニル、プロピオン酸ビニル、酪酸ビニル、イソ酪酸ビニル、ピバリン酸ビニル、カプロン酸ビニル、バーサチック酸ビニル、ラウリン酸ビニル、ステアリン酸ビニル、シクロヘキシルカルボン酸ビニル、安息香酸ビニル、パラ-t-ブチル安息香酸ビニルなどが含まれる。
Carboxylic acid vinyl esters can improve compatibility and gloss, and increase the glass transition temperature. Examples of vinyl carboxylates include vinyl acetate, vinyl propionate, vinyl butyrate, vinyl isobutyrate, vinyl pivalate, vinyl caproate, vinyl versatate, vinyl laurate, vinyl stearate, vinyl cyclohexylcarboxylate, benzoate Vinyl acid, para-t-butyl vinyl benzoate and the like are included.
アルキルビニルエーテル類は、光沢および柔軟性を向上させることができる。アルキルビニルエーテル類の例には、メチルビニルエーテル、エチルビニルエーテル、ブチルビニルエーテルなどが含まれる。
Alkyl vinyl ethers can improve gloss and flexibility. Examples of the alkyl vinyl ethers include methyl vinyl ether, ethyl vinyl ether, butyl vinyl ether and the like.
非フッ素系オレフィン類は、可とう性を向上させることができる。非フッ素系オレフィン類の例には、エチレン、プロピレン、n-ブテン、イソブテンなどが含まれる。
Non-fluorinated olefins can improve flexibility. Examples of non-fluorinated olefins include ethylene, propylene, n-butene, isobutene and the like.
上記モノマーを公知の重合法で共重合させることで、親水性官能基を有するフルオロオレフィン共重合体を得ることができる。このとき、フルオロオレフィン共重合体が親水性官能基を0.05~5質量%有するように、原料モノマー組成物におけるフルオロオレフィンの量を調整することで、乳化剤をほとんど使用せずにフルオロオレフィン共重合体の水系エマルションを製造することができる。乳化剤をほとんど含有しない(1質量%以下)フルオロオレフィン共重合体のエマルションを用いて形成された化成処理皮膜には、乳化剤がほとんど含まれない。
A fluoroolefin copolymer having a hydrophilic functional group can be obtained by copolymerizing the above monomers by a known polymerization method. At this time, by adjusting the amount of the fluoroolefin in the raw material monomer composition so that the fluoroolefin copolymer has a hydrophilic functional group of 0.05 to 5% by mass, the fluoroolefin copolymer can be used with almost no emulsifier. An aqueous emulsion of the polymer can be produced. The chemical conversion film formed using an emulsion of a fluoroolefin copolymer containing almost no emulsifier (1% by mass or less) contains almost no emulsifier.
このように、化成処理皮膜を構成する水系フッ素含有樹脂として、親水性官能基を有するフッ素含有樹脂を用いることで、乳化剤をほとんど含まない化成処理皮膜を容易に形成することができる。このようにして形成された化成処理皮膜は、乳化剤の残留による耐水性の劣化がほとんど見られず、優れた耐水性を発揮する。
Thus, by using a fluorine-containing resin having a hydrophilic functional group as the water-based fluorine-containing resin constituting the chemical conversion treatment film, a chemical conversion treatment film containing almost no emulsifier can be easily formed. The chemical conversion film thus formed exhibits excellent water resistance with little deterioration in water resistance due to the residual emulsifier.
2)4A族金属化合物
化成処理皮膜は、4A族金属化合物を含む。4A族金属化合物は、水系フッ素含有樹脂中のカルボキシル基やスルホン酸基などの官能基と反応しやすく、水系フッ素含有樹脂の硬化または架橋反応を促進する。そのため、低温乾燥でも化成処理皮膜の耐水性を向上させることができる。 2) Group 4A metal compound The chemical conversion film contains a group 4A metal compound. The 4A group metal compound easily reacts with a functional group such as a carboxyl group or a sulfonic acid group in the aqueous fluorine-containing resin, and accelerates the curing or crosslinking reaction of the aqueous fluorine-containing resin. Therefore, the water resistance of the chemical conversion film can be improved even by low-temperature drying.
化成処理皮膜は、4A族金属化合物を含む。4A族金属化合物は、水系フッ素含有樹脂中のカルボキシル基やスルホン酸基などの官能基と反応しやすく、水系フッ素含有樹脂の硬化または架橋反応を促進する。そのため、低温乾燥でも化成処理皮膜の耐水性を向上させることができる。 2) Group 4A metal compound The chemical conversion film contains a group 4A metal compound. The 4A group metal compound easily reacts with a functional group such as a carboxyl group or a sulfonic acid group in the aqueous fluorine-containing resin, and accelerates the curing or crosslinking reaction of the aqueous fluorine-containing resin. Therefore, the water resistance of the chemical conversion film can be improved even by low-temperature drying.
フッ素含有樹脂の架橋に、メラミン樹脂やイソシアネート樹脂などを使用した場合、耐候劣化しやすいという問題がある。たとえば、メラミン樹脂を使用して硬化させた化成処理皮膜では、エステル結合やホルムエーテル結合などが酸化、加水分解することによりすぐに耐候劣化してしまう。また、酸性雨に含まれる硫酸イオンや硝酸イオンなどの酸性物質によって架橋構造が切断されることによっても、耐候劣化が進行する。イソシアネート樹脂を使用して硬化させた化成処理皮膜では、架橋部分に形成されるウレタン結合はF結合よりも弱いため、架橋構造が優先的に切断されてしまい、耐候劣化が進行する。
When a melamine resin or an isocyanate resin is used for crosslinking of the fluorine-containing resin, there is a problem that the weather resistance is easily deteriorated. For example, in a chemical conversion treatment film cured using a melamine resin, weather resistance deterioration is immediately caused by oxidation and hydrolysis of ester bonds and form ether bonds. In addition, weathering deterioration also proceeds when the crosslinked structure is cut by an acidic substance such as sulfate ion or nitrate ion contained in acid rain. In a chemical conversion film cured using an isocyanate resin, the urethane bond formed at the cross-linked portion is weaker than the F bond, so that the cross-linked structure is preferentially cut, and the weather resistance deteriorates.
これに対し、フッ素含有樹脂の架橋に4A族金属化合物を使用することで、このような問題を回避することが可能であり、耐候性を向上させることもできる。
On the other hand, by using a group 4A metal compound for cross-linking of the fluorine-containing resin, such a problem can be avoided and weather resistance can be improved.
また、4A族金属化合物は、皮膜密着性、耐水性および耐黒変性も向上させる。すなわち、Al含有Zn系合金めっき鋼板の表面に存在する強固なAl酸化物は、化成処理皮膜の密着性を低下させるが、化成処理皮膜に4A族金属化合物を含ませることにより、このAl酸化物による皮膜密着性の低下を抑制することができる。また、4A族金属化合物は、エッチング反応により溶出したAlイオンと反応する4A族金属イオンの供給源ともなる。反応生成物は、めっき層と化成処理皮膜の界面に濃化して、初期の耐食性および耐黒変性を向上させる。4A族金属の例には、TiやZr、Hfなどが含まれる。
The group 4A metal compound also improves film adhesion, water resistance and blackening resistance. That is, the strong Al oxide present on the surface of the Al-containing Zn-based alloy-plated steel sheet decreases the adhesion of the chemical conversion coating, but this Al oxide can be obtained by including a group 4A metal compound in the chemical conversion coating. It is possible to suppress a decrease in film adhesion due to. The group 4A metal compound also serves as a supply source of group 4A metal ions that react with Al ions eluted by the etching reaction. The reaction product is concentrated at the interface between the plating layer and the chemical conversion film to improve the initial corrosion resistance and blackening resistance. Examples of Group 4A metals include Ti, Zr, Hf, and the like.
化成処理皮膜中の4A族金属化合物の含有量は、フッ素含有樹脂に対して金属換算で0.1~5質量%の範囲内が好ましい。含有量が金属換算で0.1質量%未満の場合、Al酸化物の濃化に起因する悪影響を十分に抑制することができず、また水系フッ素含有樹脂脂を十分に架橋させることができず、結果として化成処理皮膜の耐水性を十分に向上させることができない。一方、含有量が金属換算で5質量%超の場合、化成処理皮膜が多孔質状となり、加工性、耐候性が低下するおそれがある。
The content of the group 4A metal compound in the chemical conversion film is preferably in the range of 0.1 to 5% by mass in terms of metal with respect to the fluorine-containing resin. When the content is less than 0.1% by mass in terms of metal, the adverse effect due to the concentration of Al oxide cannot be sufficiently suppressed, and the aqueous fluorine-containing resin fat cannot be sufficiently crosslinked. As a result, the water resistance of the chemical conversion coating cannot be sufficiently improved. On the other hand, when the content is more than 5% by mass in terms of metal, the chemical conversion film becomes porous, and the workability and weather resistance may be reduced.
化成処理皮膜中の4A族金属化合物の金属換算量は、蛍光X線分析装置を用いることで測定することができる。
The metal equivalent amount of the group 4A metal compound in the chemical conversion coating can be measured by using a fluorescent X-ray analyzer.
上述のように化成処理皮膜中にはめっき層から溶出したAlが存在する。このAlは、耐食性の向上に寄与する。Alの存在により耐食性が向上するのは、以下のメカニズムによるものと推察される。すなわち、1)化成処理液が弱アルカリ性であるため、化成処理液を塗布した際に、めっき層に含まれるAlの酸化物および金属Alが選択的に化成処理液に溶出する(Znはほとんど溶出しない)。2)化成処理液のpH域では、AlはAl(OH)4
-の状態で化成処理液に溶解する。3)化成処理液を乾燥させて化成処理皮膜を形成する際に、化成処理液中のAlは脱水縮合などにより化成処理皮膜中に取り込まれる。4)その結果として、化成処理皮膜の絶縁性や緻密度などが向上し、耐食性が向上する。
As described above, Al eluted from the plating layer is present in the chemical conversion film. This Al contributes to improvement of corrosion resistance. It is presumed that the corrosion resistance is improved by the presence of Al due to the following mechanism. That is, 1) Since the chemical conversion treatment solution is weakly alkaline, when the chemical conversion treatment solution is applied, Al oxide and metal Al contained in the plating layer are selectively eluted into the chemical conversion treatment solution (Zn is almost eluted). do not do). 2) In the pH range of the chemical conversion treatment liquid, Al is Al (OH) 4 - is dissolved in the chemical conversion treatment liquid state. 3) When forming the chemical conversion film by drying the chemical conversion liquid, Al in the chemical conversion liquid is taken into the chemical conversion film by dehydration condensation or the like. 4) As a result, the insulating properties and density of the chemical conversion coating are improved, and the corrosion resistance is improved.
3)樹脂粒子
化成処理皮膜は、樹脂粒子を含む。樹脂粒子は、化成処理皮膜中に分散しており、少なくともその一部は化成処理皮膜の表面において露出(突出)している(図3C参照)。化成処理皮膜の表面において突出している樹脂粒子は、成形加工時に「コロ」のように機能することで、化成処理皮膜表面の潤滑性を向上させる。その結果、化成処理Zn系めっき鋼板の加工性を向上させることができる。 3) Resin particle The chemical conversion film contains resin particles. The resin particles are dispersed in the chemical conversion treatment film, and at least a part thereof is exposed (protruded) on the surface of the chemical conversion treatment film (see FIG. 3C). The resin particles protruding on the surface of the chemical conversion treatment film function like a “roller” during the molding process, thereby improving the lubricity of the chemical conversion treatment film surface. As a result, the workability of the chemical conversion treated Zn-plated steel sheet can be improved.
化成処理皮膜は、樹脂粒子を含む。樹脂粒子は、化成処理皮膜中に分散しており、少なくともその一部は化成処理皮膜の表面において露出(突出)している(図3C参照)。化成処理皮膜の表面において突出している樹脂粒子は、成形加工時に「コロ」のように機能することで、化成処理皮膜表面の潤滑性を向上させる。その結果、化成処理Zn系めっき鋼板の加工性を向上させることができる。 3) Resin particle The chemical conversion film contains resin particles. The resin particles are dispersed in the chemical conversion treatment film, and at least a part thereof is exposed (protruded) on the surface of the chemical conversion treatment film (see FIG. 3C). The resin particles protruding on the surface of the chemical conversion treatment film function like a “roller” during the molding process, thereby improving the lubricity of the chemical conversion treatment film surface. As a result, the workability of the chemical conversion treated Zn-plated steel sheet can be improved.
樹脂粒子の平均粒子径は、0.1~10μmの範囲内が好ましい。樹脂粒子の平均粒子径が0.1μm未満の場合、大部分の樹脂粒子が化成処理皮膜中に埋没してしまうため、化成処理皮膜表面の潤滑性を効率よく向上させることができない。一方、樹脂粒子の平均粒子径が10μm超の場合、成形加工時に樹脂粒子が脱落しやすくなるおそれがある。このように樹脂粒子が脱落してしまうと、化成処理皮膜に欠陥が生じてしまい、耐食性が低下してしまうことになる。なお、本明細書において、樹脂粒子の平均粒子径とは、レーザ回折散乱法によって測定した粒子径分布における積算値50%での粒子径(メジアン径)を意味する。
The average particle diameter of the resin particles is preferably within the range of 0.1 to 10 μm. When the average particle diameter of the resin particles is less than 0.1 μm, most of the resin particles are buried in the chemical conversion coating, so that the lubricity of the chemical conversion coating surface cannot be improved efficiently. On the other hand, when the average particle diameter of the resin particles exceeds 10 μm, the resin particles may be easily dropped during the molding process. If the resin particles fall off in this way, defects will occur in the chemical conversion film, and the corrosion resistance will be reduced. In the present specification, the average particle diameter of the resin particles means a particle diameter (median diameter) at an integrated value of 50% in a particle diameter distribution measured by a laser diffraction scattering method.
化成処理皮膜中の樹脂粒子の含有量は、化成処理皮膜の表面における樹脂粒子の面積占有率が0.1面積%以上となるように調整される。樹脂粒子の面積占有率が0.1面積%未満の場合、化成処理皮膜の潤滑性を十分に向上させることができない。化成処理皮膜の表面における樹脂粒子の面積占有率は、化成処理皮膜の表面を走査型電子顕微鏡(SEM)で観察することで求めることができる。
The content of the resin particles in the chemical conversion coating is adjusted so that the area occupancy ratio of the resin particles on the surface of the chemical conversion coating is 0.1 area% or more. If the area occupancy of the resin particles is less than 0.1 area%, the lubricity of the chemical conversion coating cannot be sufficiently improved. The area occupancy ratio of the resin particles on the surface of the chemical conversion coating can be determined by observing the surface of the chemical conversion coating with a scanning electron microscope (SEM).
樹脂粒子の種類は、特に限定されないが、耐熱性および分散性の観点からはポリエチレン-フッ素樹脂粒子が好ましい。
The type of resin particles is not particularly limited, but polyethylene-fluorine resin particles are preferable from the viewpoint of heat resistance and dispersibility.
ポリエチレン樹脂粒子は、比重が小さいため(例えば、0.95)化成処理液中の分散性に優れるが、融点が低いため(例えば、123℃)耐熱性に劣る。このように耐熱性が低い樹脂粒子を使用した場合、化成処理皮膜を形成するために化成処理液を加熱乾燥する際に粒子の形状を維持できないおそれがある。具体的には、化成処理液を140~180℃程度で加熱乾燥すると、ポリエチレン樹脂粒子は溶融してしまう。このように樹脂粒子が溶融してしまうと、樹脂粒子は「コロ」として機能することができず、潤滑性の向上に寄与することができない。
Polyethylene resin particles are excellent in dispersibility in the chemical conversion liquid because of low specific gravity (eg, 0.95), but poor in heat resistance because of low melting point (eg, 123 ° C.). When resin particles having low heat resistance are used as described above, the shape of the particles may not be maintained when the chemical conversion solution is heated and dried to form a chemical conversion coating. Specifically, when the chemical conversion treatment liquid is heated and dried at about 140 to 180 ° C., the polyethylene resin particles are melted. When the resin particles are thus melted, the resin particles cannot function as “rollers” and cannot contribute to improvement of lubricity.
一方、フッ素樹脂粒子は、融点が高いため(例えば、330℃)耐熱性に優れるが、比重が大きいため(例えば、2.2)化成処理液中の分散性に劣る。このように化成処理液中の分散性が低い樹脂粒子を使用した場合、化成処理皮膜中に均一に樹脂粒子を分散させることが困難となる。
On the other hand, since the fluororesin particles have a high melting point (eg, 330 ° C.), they are excellent in heat resistance, but because of their large specific gravity (eg, 2.2), they are inferior in dispersibility in the chemical conversion treatment liquid. Thus, when resin particles with low dispersibility in the chemical conversion treatment liquid are used, it becomes difficult to uniformly disperse the resin particles in the chemical conversion treatment film.
ポリエチレン-フッ素樹脂粒子は、加熱して軟化させたポリエチレン樹脂粒子にフッ素樹脂微粒子を接触させることにより、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子を結合(吸着)させたものである。ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子を結合させることで、140~180℃程度で加熱しても粒子の形状を維持することができる程度の耐熱性と、化成処理液中の分散性(比重:例えば1.1)とを両立させることができる。また、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子を結合させることで、耐候性も向上させることができる。
The polyethylene-fluororesin particles are obtained by bonding (adsorbing) fluororesin fine particles to the surface of the polyethylene resin particles by bringing the fluororesin fine particles into contact with the polyethylene resin particles softened by heating. By bonding fluororesin fine particles to the surface of the polyethylene resin particles, the heat resistance is such that the shape of the particles can be maintained even when heated at about 140 to 180 ° C., and the dispersibility in the chemical conversion liquid (specific gravity: For example, 1.1) can be made compatible. Moreover, a weather resistance can also be improved by couple | bonding a fluororesin fine particle with the surface of a polyethylene resin particle.
図1は、50℃または150℃まで加熱された、ポリエチレン樹脂粒子またはポリエチレン-フッ素樹脂粒子を含む化成処理皮膜のSEM像(平面視)である。図1Aは、ポリエチレン樹脂粒子を含む化成処理液をめっき鋼板の表面に塗布し、50℃で乾燥させることにより形成した化成処理皮膜のSEM像である。図1Bは、ポリエチレン樹脂粒子を含む化成処理液をめっき鋼板の表面に塗布し、150℃で乾燥させることにより形成した化成処理皮膜のSEM像である。図1Cは、ポリエチレン-フッ素樹脂粒子を含む化成処理液をめっき鋼板の表面に塗布し、50℃で乾燥させることにより形成した化成処理皮膜のSEM像である。図1Dは、ポリエチレン-フッ素樹脂粒子を含む化成処理液をめっき鋼板の表面に塗布し、150℃で乾燥させることにより形成した化成処理皮膜のSEM像である。
FIG. 1 is an SEM image (plan view) of a chemical conversion coating containing polyethylene resin particles or polyethylene-fluororesin particles heated to 50 ° C. or 150 ° C. FIG. 1A is an SEM image of a chemical conversion coating formed by applying a chemical conversion treatment liquid containing polyethylene resin particles to the surface of a plated steel sheet and drying at 50 ° C. FIG. FIG. 1B is an SEM image of a chemical conversion film formed by applying a chemical conversion treatment liquid containing polyethylene resin particles to the surface of a plated steel sheet and drying at 150 ° C. FIG. 1C is an SEM image of a chemical conversion film formed by applying a chemical conversion liquid containing polyethylene-fluorine resin particles to the surface of a plated steel sheet and drying at 50 ° C. FIG. 1D is an SEM image of a chemical conversion coating formed by applying a chemical conversion treatment liquid containing polyethylene-fluorine resin particles to the surface of a plated steel sheet and drying at 150 ° C.
図1Aに示されるように、ポリエチレン樹脂粒子を含む化成処理液を50℃で乾燥させた場合、ポリエチレン樹脂粒子の形状は維持され、化成処理皮膜の表面からポリエチレン樹脂粒子が突出していた(図中「PE」参照)。一方、図1Bに示されるように、ポリエチレン樹脂粒子を含む化成処理液を150℃で乾燥させた場合は、ポリエチレン樹脂粒子は溶融してしまい、ポリエチレン樹脂は化成処理皮膜の表面に均一に分布していた(図中「PE」参照)。
As shown in FIG. 1A, when the chemical conversion treatment liquid containing polyethylene resin particles was dried at 50 ° C., the shape of the polyethylene resin particles was maintained, and the polyethylene resin particles protruded from the surface of the chemical conversion treatment film (in the figure). (See “PE”). On the other hand, as shown in FIG. 1B, when the chemical conversion treatment liquid containing polyethylene resin particles is dried at 150 ° C., the polyethylene resin particles are melted, and the polyethylene resin is uniformly distributed on the surface of the chemical conversion treatment film. (See “PE” in the figure).
図1Cに示されるように、ポリエチレン-フッ素樹脂粒子を含む化成処理液を50℃で乾燥させた場合、ポリエチレン-フッ素樹脂粒子の形状は維持され、化成処理皮膜の表面からポリエチレン-フッ素樹脂粒子が突出していた(図中「PE-F」参照)。また、図1Dに示されるように、ポリエチレン-フッ素樹脂粒子を含む化成処理液を150℃で乾燥させた場合、一部のポリエチレン-フッ素樹脂粒子(フッ素樹脂微粒子の付着量が少ないもの)は溶融したが(図中「PE」参照)、残部のポリエチレン-フッ素樹脂(フッ素樹脂微粒子の付着量が多いもの)は溶融しなかった(図中「PE-F」参照)。
As shown in FIG. 1C, when the chemical conversion treatment liquid containing polyethylene-fluororesin particles is dried at 50 ° C., the shape of the polyethylene-fluororesin particles is maintained, and the polyethylene-fluororesin particles are removed from the surface of the chemical conversion coating. It protruded (see “PE-F” in the figure). In addition, as shown in FIG. 1D, when the chemical conversion treatment liquid containing polyethylene-fluororesin particles is dried at 150 ° C., some polyethylene-fluororesin particles (those with a small amount of fluororesin fine particles attached) are melted. However, the remaining polyethylene-fluororesin (with a large amount of fluororesin fine particles attached) did not melt (see “PE-F” in the figure).
このように、乾燥温度が低い場合は、ポリエチレン樹脂粒子でも問題ないが、乾燥温度が高い場合は、ポリエチレン-フッ素樹脂粒子を使用することが好ましい。また、耐候性の観点からも、ポリエチレン-フッ素樹脂粒子を使用することが好ましい。
Thus, when the drying temperature is low, there is no problem with the polyethylene resin particles, but when the drying temperature is high, it is preferable to use polyethylene-fluororesin particles. From the viewpoint of weather resistance, it is preferable to use polyethylene-fluororesin particles.
ポリエチレン-フッ素樹脂粒子を構成するポリエチレン樹脂粒子の平均粒子径は、ポリエチレン-フッ素樹脂粒子の平均粒子径が0.1~10μmの範囲内となれば特に限定されない。使用できるポリエチレン樹脂粒子の市販品の例には、HYTEC E-9016、HYTEC E-1000(いずれも東邦化学株式会社)、CJ-172B、CJ-137(いずれも興洋化学株式会社)、パーマリンKUE-4、パーマリンKUE-5(いずれも三洋化成工業株式会社)などが含まれる。一方、ポリエチレン-フッ素樹脂粒子を構成するフッ素樹脂微粒子の平均粒子径は、ポリエチレン樹脂粒子の平均粒子径に応じて適宜設定すればよい。たとえば、フッ素樹脂微粒子の平均粒子径は、0.3μm以下が好ましい。
The average particle diameter of the polyethylene resin particles constituting the polyethylene-fluorine resin particles is not particularly limited as long as the average particle diameter of the polyethylene-fluorine resin particles is within the range of 0.1 to 10 μm. Examples of commercially available polyethylene resin particles that can be used include HYTEC E-9016, HYTEC E-1000 (all are Toho Chemical Co., Ltd.), CJ-172B, CJ-137 (all are Koyo Chemical Co., Ltd.), and Permarin KUE. -4, Permarin KUE-5 (both are Sanyo Chemical Industries). On the other hand, the average particle diameter of the fluororesin fine particles constituting the polyethylene-fluororesin particles may be appropriately set according to the average particle diameter of the polyethylene resin particles. For example, the average particle diameter of the fluororesin fine particles is preferably 0.3 μm or less.
ポリエチレン-フッ素樹脂粒子中のフッ素樹脂の割合は、5~40質量%の範囲内が好ましい。フッ素樹脂の割合が5質量%未満の場合、フッ素樹脂を付着させることによる効果を十分に発揮させることができないおそれがある。一方、フッ素樹脂の割合が40質量%超の場合、ポリエチレン-フッ素樹脂粒子が化成処理皮膜から脱落しやすくなるおそれがある。ポリエチレン-フッ素樹脂粒子中におけるフッ素樹脂の割合は、蛍光X線分析装置を用いることで測定することができる。
The ratio of the fluororesin in the polyethylene-fluororesin particles is preferably in the range of 5 to 40% by mass. When the ratio of a fluororesin is less than 5 mass%, there exists a possibility that the effect by making a fluororesin adhere cannot fully be exhibited. On the other hand, when the ratio of the fluororesin is more than 40% by mass, the polyethylene-fluororesin particles may be easily removed from the chemical conversion film. The ratio of the fluororesin in the polyethylene-fluororesin particles can be measured by using a fluorescent X-ray analyzer.
4)ポリエチレン樹脂
化成処理皮膜がポリエチレン-フッ素樹脂粒子を含む場合、化成処理皮膜は、さらにポリエチレン樹脂を含むことが好ましい。通常、ポリエチレン樹脂は、化成処理皮膜の表面のうち、ポリエチレン-フッ素樹脂粒子が突出していない領域の全部または一部を被覆している(図2C参照)。この化成処理皮膜の表面を被覆しているポリエチレン樹脂は、製造工程において化成処理皮膜の表面にブリードしたポリエチレン樹脂である。ポリエチレン樹脂粒子は、ポリエチレン-フッ素樹脂粒子と共に化成処理皮膜表面の潤滑性を向上させることで、化成処理Zn系めっき鋼板の加工性をさらに向上させる。 4) Polyethylene resin When the chemical conversion film contains polyethylene-fluororesin particles, the chemical conversion film preferably further contains a polyethylene resin. Usually, the polyethylene resin covers all or a part of the surface of the chemical conversion film where the polyethylene-fluororesin particles do not protrude (see FIG. 2C). The polyethylene resin that coats the surface of the chemical conversion coating is a polyethylene resin that bleeds on the surface of the chemical conversion coating in the manufacturing process. The polyethylene resin particles, together with the polyethylene-fluorine resin particles, improve the lubricity of the surface of the chemical conversion treatment film, thereby further improving the workability of the chemical conversion treatment Zn-plated steel sheet.
化成処理皮膜がポリエチレン-フッ素樹脂粒子を含む場合、化成処理皮膜は、さらにポリエチレン樹脂を含むことが好ましい。通常、ポリエチレン樹脂は、化成処理皮膜の表面のうち、ポリエチレン-フッ素樹脂粒子が突出していない領域の全部または一部を被覆している(図2C参照)。この化成処理皮膜の表面を被覆しているポリエチレン樹脂は、製造工程において化成処理皮膜の表面にブリードしたポリエチレン樹脂である。ポリエチレン樹脂粒子は、ポリエチレン-フッ素樹脂粒子と共に化成処理皮膜表面の潤滑性を向上させることで、化成処理Zn系めっき鋼板の加工性をさらに向上させる。 4) Polyethylene resin When the chemical conversion film contains polyethylene-fluororesin particles, the chemical conversion film preferably further contains a polyethylene resin. Usually, the polyethylene resin covers all or a part of the surface of the chemical conversion film where the polyethylene-fluororesin particles do not protrude (see FIG. 2C). The polyethylene resin that coats the surface of the chemical conversion coating is a polyethylene resin that bleeds on the surface of the chemical conversion coating in the manufacturing process. The polyethylene resin particles, together with the polyethylene-fluorine resin particles, improve the lubricity of the surface of the chemical conversion treatment film, thereby further improving the workability of the chemical conversion treatment Zn-plated steel sheet.
化成処理皮膜中のポリエチレン樹脂の含有量は、化成処理皮膜に対して0.1~16質量%の範囲内が好ましい。ポリエチレン樹脂の含有量が0.1質量%未満の場合、ポリエチレン樹脂の効果を十分に発揮させることができない。一方、ポリエチレン樹脂の含有量が16質量%超の場合、耐候性が低下してしまうおそれがある。
The content of the polyethylene resin in the chemical conversion coating is preferably in the range of 0.1 to 16% by mass with respect to the chemical conversion coating. When the content of the polyethylene resin is less than 0.1% by mass, the effect of the polyethylene resin cannot be exhibited sufficiently. On the other hand, when the content of the polyethylene resin is more than 16% by mass, the weather resistance may be lowered.
5)リン酸塩
化成処理皮膜は、さらにリン酸塩を含むことが好ましい。リン酸塩は、Al含有Zn系合金めっき鋼板のめっき層表面と反応して、化成処理皮膜のAl含有Zn系合金めっき鋼板への密着性を向上させる。 5) Phosphate The chemical conversion treatment film preferably further contains a phosphate. Phosphate reacts with the plating layer surface of the Al-containing Zn-based alloy plated steel sheet to improve the adhesion of the chemical conversion coating to the Al-containing Zn-based alloy plated steel sheet.
化成処理皮膜は、さらにリン酸塩を含むことが好ましい。リン酸塩は、Al含有Zn系合金めっき鋼板のめっき層表面と反応して、化成処理皮膜のAl含有Zn系合金めっき鋼板への密着性を向上させる。 5) Phosphate The chemical conversion treatment film preferably further contains a phosphate. Phosphate reacts with the plating layer surface of the Al-containing Zn-based alloy plated steel sheet to improve the adhesion of the chemical conversion coating to the Al-containing Zn-based alloy plated steel sheet.
リン酸塩の種類は、リン酸アニオンを有する化合物であって、水溶性のものであれば特に限定されない。リン酸塩の例には、リン酸ナトリウム、リン酸アンモニウム、リン酸マグネシウム、リン酸カリウム、リン酸マンガン、リン酸亜鉛、オルトリン酸、メタリン酸、ピロリン酸(二リン酸)、三リン酸、四リン酸などが含まれる。これらのリン酸塩は、単独で使用されてもよいし、2種類以上を組み合わせて使用されてもよい。
The type of phosphate is not particularly limited as long as it is a compound having a phosphate anion and is water-soluble. Examples of phosphates include sodium phosphate, ammonium phosphate, magnesium phosphate, potassium phosphate, manganese phosphate, zinc phosphate, orthophosphoric acid, metaphosphoric acid, pyrophosphoric acid (diphosphoric acid), triphosphoric acid, Tetraphosphate etc. are included. These phosphates may be used alone or in combination of two or more.
化成処理皮膜中のリン酸塩の含有量は、フッ素含有樹脂に対してP換算量として0.05~3質量%の範囲内が好ましい。P換算量が0.05質量%未満の場合、めっき層表面との反応が不足して、化成処理皮膜の密着性を十分に向上させることができない。一方、P換算量が3質量%超の場合、4A族金属化合物との反応が過剰に進行して、4A族金属化合物による架橋効果が損なわれてしまう。
The content of phosphate in the chemical conversion coating is preferably in the range of 0.05 to 3% by mass in terms of P with respect to the fluorine-containing resin. When the P conversion amount is less than 0.05% by mass, the reaction with the plating layer surface is insufficient, and the adhesion of the chemical conversion film cannot be sufficiently improved. On the other hand, when P conversion amount exceeds 3 mass%, reaction with 4A group metal compound will advance excessively, and the crosslinking effect by 4A group metal compound will be impaired.
化成処理皮膜中のリン酸塩のP換算量は、蛍光X線分析装置を用いることで測定することができる。
The amount of phosphate converted to P in the chemical conversion coating can be measured by using a fluorescent X-ray analyzer.
6)シランカップリング剤
化成処理皮膜は、さらにシランカップリング剤を含むことが好ましい。シランカップリング剤を配合することで、化成処理皮膜の密着性をより向上させることができる。シランカップリング剤としては、アミノ基、エポキシ基、メルカプト基、アクリロキシ基、メタクリロキシ基、アルコキシ基、ビニル基、スチリル基、イソシアネート基、クロロプロピル基などの官能基を1種類または2種類以上含むシラン化合物が使用される。 6) Silane coupling agent It is preferable that a chemical conversion treatment film contains a silane coupling agent further. By mix | blending a silane coupling agent, the adhesiveness of a chemical conversion treatment film can be improved more. Silane coupling agents include silanes containing one or more functional groups such as amino, epoxy, mercapto, acryloxy, methacryloxy, alkoxy, vinyl, styryl, isocyanate, and chloropropyl groups. A compound is used.
化成処理皮膜は、さらにシランカップリング剤を含むことが好ましい。シランカップリング剤を配合することで、化成処理皮膜の密着性をより向上させることができる。シランカップリング剤としては、アミノ基、エポキシ基、メルカプト基、アクリロキシ基、メタクリロキシ基、アルコキシ基、ビニル基、スチリル基、イソシアネート基、クロロプロピル基などの官能基を1種類または2種類以上含むシラン化合物が使用される。 6) Silane coupling agent It is preferable that a chemical conversion treatment film contains a silane coupling agent further. By mix | blending a silane coupling agent, the adhesiveness of a chemical conversion treatment film can be improved more. Silane coupling agents include silanes containing one or more functional groups such as amino, epoxy, mercapto, acryloxy, methacryloxy, alkoxy, vinyl, styryl, isocyanate, and chloropropyl groups. A compound is used.
化成処理皮膜中のシランカップリング剤の含有量は、フッ素含有樹脂に対して0.5~5質量%の範囲内が好ましい。シランカップリング剤の含有量が0.5質量%未満の場合、化成処理皮膜の密着性を十分に向上させることができない。一方、シランカップリング剤の含有量が5質量%超の場合、皮膜密着性は飽和し、それ以上の向上は認められない。
The content of the silane coupling agent in the chemical conversion film is preferably in the range of 0.5 to 5% by mass with respect to the fluorine-containing resin. When content of a silane coupling agent is less than 0.5 mass%, the adhesiveness of a chemical conversion treatment film cannot fully be improved. On the other hand, when the content of the silane coupling agent exceeds 5% by mass, the film adhesion is saturated and no further improvement is observed.
化成処理皮膜中のシランカップリング剤の含有量は、蛍光X線分析装置を用いることで測定することができる。
The content of the silane coupling agent in the chemical conversion film can be measured by using a fluorescent X-ray analyzer.
化成処理皮膜の膜厚は、0.5~10μmの範囲内が好ましい。膜厚が0.5μm未満の場合、耐食性や耐変色性などを十分に付与することができない。一方、膜厚を10μm超としても、膜厚の増加に伴う性能向上を期待することはできない。
The film thickness of the chemical conversion coating is preferably in the range of 0.5 to 10 μm. When the film thickness is less than 0.5 μm, sufficient corrosion resistance and discoloration resistance cannot be imparted. On the other hand, even if the film thickness exceeds 10 μm, it cannot be expected to improve the performance with the increase in film thickness.
2.化成処理Zn系めっき鋼板の製造方法
本発明の化成処理Zn系めっき鋼板の製造方法は、特に限定されないが、例えば以下の方法により製造されうる。 2. The manufacturing method of the chemical conversion treatment Zn-plated steel plate Although the manufacturing method of the chemical conversion treatment Zn-plated steel plate of this invention is not specifically limited, For example, it can manufacture by the following method.
本発明の化成処理Zn系めっき鋼板の製造方法は、特に限定されないが、例えば以下の方法により製造されうる。 2. The manufacturing method of the chemical conversion treatment Zn-plated steel plate Although the manufacturing method of the chemical conversion treatment Zn-plated steel plate of this invention is not specifically limited, For example, it can manufacture by the following method.
本発明の化成処理Zn系めっき鋼板の製造方法は、1)Al含有Zn系合金めっき鋼板(化成処理原板)を準備する第1のステップと、2)化成処理液を準備する第2のステップと、3)Al含有Zn系合金めっき鋼板の表面に化成処理皮膜を形成する第3のステップとを含む。なお、下地化成処理皮膜を介してAl含有Zn系合金めっき鋼板の表面に化成処理皮膜を形成する場合には、化成処理皮膜を形成する第3のステップの前に、Al含有Zn系合金めっき鋼板の表面に下地化成処理皮膜を形成するステップをさらに含む。
The manufacturing method of the chemical conversion treatment Zn-plated steel sheet of the present invention includes 1) a first step of preparing an Al-containing Zn-based alloy plated steel sheet (chemical conversion treatment original plate), and 2) a second step of preparing a chemical conversion treatment liquid. And 3) a third step of forming a chemical conversion film on the surface of the Al-containing Zn-based alloy plated steel sheet. In addition, when forming a chemical conversion treatment film on the surface of an Al content Zn system alloy plating steel plate via a foundation chemical conversion treatment film, before the 3rd step of forming a chemical conversion treatment film, an Al content Zn system alloy plating steel plate The method further includes the step of forming a base chemical conversion coating on the surface of the substrate.
[化成処理原板の準備]
第1のステップでは、化成処理原板として前述のAl含有Zn系合金めっき鋼板を準備する。 [Preparation of raw material for chemical conversion treatment]
In the first step, the aforementioned Al-containing Zn-based alloy plated steel sheet is prepared as a chemical conversion treatment original sheet.
第1のステップでは、化成処理原板として前述のAl含有Zn系合金めっき鋼板を準備する。 [Preparation of raw material for chemical conversion treatment]
In the first step, the aforementioned Al-containing Zn-based alloy plated steel sheet is prepared as a chemical conversion treatment original sheet.
[化成処理液の準備]
第2のステップでは、前述の親水性官能基を有するフッ素含有樹脂(好ましくは、フッ素含有オレフィン樹脂)、4A族金属化合物および樹脂粒子を含む化成処理液を準備する。 [Preparation of chemical conversion solution]
In the second step, a chemical conversion liquid containing a fluorine-containing resin having a hydrophilic functional group (preferably a fluorine-containing olefin resin), a group 4A metal compound, and resin particles is prepared.
第2のステップでは、前述の親水性官能基を有するフッ素含有樹脂(好ましくは、フッ素含有オレフィン樹脂)、4A族金属化合物および樹脂粒子を含む化成処理液を準備する。 [Preparation of chemical conversion solution]
In the second step, a chemical conversion liquid containing a fluorine-containing resin having a hydrophilic functional group (preferably a fluorine-containing olefin resin), a group 4A metal compound, and resin particles is prepared.
化成処理液は、前述の親水性官能基を有するフッ素含有樹脂(好ましくは、フッ素含有オレフィン樹脂)の水系エマルションに、4A族金属化合物および樹脂粒子(例えば、ポリエチレン-フッ素樹脂粒子)を添加することで調製されうる。化成処理液に添加する4A族金属化合物としては、4A族金属の酸素酸塩やフッ化物、水酸化物、有機酸塩、炭酸塩、過酸化塩などが用いられる。酸素酸塩の例には、水素酸塩、アンモニウム塩、アルカリ金属塩、アルカリ土類金属塩などが含まれる。化成処理液には、さらに必要に応じてポリエチレン樹脂粒子やリン酸塩、シランカップリング剤などを添加してもよい。
In the chemical conversion treatment liquid, a group 4A metal compound and resin particles (for example, polyethylene-fluorine resin particles) are added to the aqueous emulsion of the fluorine-containing resin having a hydrophilic functional group (preferably fluorine-containing olefin resin). Can be prepared. As the group 4A metal compound to be added to the chemical conversion treatment liquid, a group 4A metal oxyacid salt, fluoride, hydroxide, organic acid salt, carbonate, peroxide salt, or the like is used. Examples of oxyacid salts include hydrates, ammonium salts, alkali metal salts, alkaline earth metal salts, and the like. You may add a polyethylene resin particle, a phosphate, a silane coupling agent, etc. to a chemical conversion liquid as needed.
水系エマルションに含まれるフッ素含有樹脂の数平均分子量は、1000以上が好ましく、1万以上がより好ましく、20万以上が特に好ましい。前述の通り、化成処理皮膜に耐水性を付与するためである。一方、処理液の安定性の観点から、フッ素含有樹脂の数平均分子量は、200万以下が好ましい。
The number average molecular weight of the fluorine-containing resin contained in the aqueous emulsion is preferably 1000 or more, more preferably 10,000 or more, and particularly preferably 200,000 or more. As described above, this is to impart water resistance to the chemical conversion coating. On the other hand, from the viewpoint of the stability of the treatment liquid, the number average molecular weight of the fluorine-containing resin is preferably 2 million or less.
フッ素含有樹脂は、乳化剤をほとんど使用せずに水系エマルションを調製する観点から、親水性官能基を0.05~5質量%有することが好ましい。
The fluorine-containing resin preferably has a hydrophilic functional group of 0.05 to 5% by mass from the viewpoint of preparing an aqueous emulsion without using an emulsifier.
フッ素含有樹脂の水系エマルション中の乳化剤の含有量は、1質量%以下が好ましい。乳化剤が1質量%超の場合、第3のステップにおいて化成処理皮膜を形成した場合に、乾燥温度によっては化成処理皮膜中に乳化剤が残存してしまうおそれがある。このように化成処理皮膜中に乳化剤が残存してしまうと、化成処理皮膜の耐水性が顕著に低下してしまうため、好ましくない。前述の通り、親水性官能基を有するフッ素含有樹脂であれば、乳化剤の量を1質量%以下としても、水系エマルションを調製することができる。
The content of the emulsifier in the aqueous emulsion of the fluorine-containing resin is preferably 1% by mass or less. When the emulsifier exceeds 1% by mass, the emulsifier may remain in the chemical conversion film depending on the drying temperature when the chemical conversion film is formed in the third step. If the emulsifier remains in the chemical conversion coating as described above, the water resistance of the chemical conversion coating is remarkably lowered, which is not preferable. As described above, an aqueous emulsion can be prepared even if the amount of the emulsifier is 1% by mass or less as long as it is a fluorine-containing resin having a hydrophilic functional group.
フッ素含有樹脂の水系エマルション中に含まれてもよい乳化剤としては、耐候性および耐水性の観点から、パーフルオロオクタン酸のアンモニウム塩、パーフルオロノナン酸のアンモニウム塩などのフッ素系乳化剤が好ましい。その他、公知のフッ素系界面活性剤も乳化剤として使用することができる。
The emulsifier that may be contained in the aqueous emulsion of the fluorine-containing resin is preferably a fluorine-based emulsifier such as an ammonium salt of perfluorooctanoic acid or an ammonium salt of perfluorononanoic acid from the viewpoint of weather resistance and water resistance. In addition, a known fluorosurfactant can also be used as an emulsifier.
化成処理液中のフッ素含有樹脂の含有量は、水100質量部に対して、10~70質量部の範囲内が好ましい。フッ素含有樹脂の含有量が10質量部未満の場合、乾燥過程において水の蒸発量が多くなり、化成処理皮膜の成膜性および緻密性が低下するおそれがある。一方、フッ素含有樹脂の含有量が70質量部超の場合、化成処理液の保存安定性が低下するおそれがある。
The content of the fluorine-containing resin in the chemical conversion treatment liquid is preferably in the range of 10 to 70 parts by mass with respect to 100 parts by mass of water. When the content of the fluorine-containing resin is less than 10 parts by mass, the amount of water evaporation increases in the drying process, and the film formability and denseness of the chemical conversion film may be reduced. On the other hand, when content of fluorine-containing resin is more than 70 mass parts, there exists a possibility that the storage stability of a chemical conversion liquid may fall.
化成処理液中の4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩の含有量は、フッ素含有樹脂100質量部に対して、金属換算で0.1~5質量部の範囲内が好ましい。これらの塩の含有量が0.1質量部未満の場合、架橋反応およびめっき層表面との反応が不足して、化成処理皮膜の耐水性および皮膜密着性を十分に向上させることができない。一方、これらの塩の含有量が5質量部超の場合、架橋反応が進行して、化成処理液の保存安定性が低下するおそれがある。
The content of group 4A metal oxyacid salt, fluoride, hydroxide, organic acid salt, carbonate or peroxide in the chemical conversion solution is 0. A range of 1 to 5 parts by mass is preferable. When the content of these salts is less than 0.1 part by mass, the crosslinking reaction and the reaction with the plating layer surface are insufficient, and the water resistance and film adhesion of the chemical conversion film cannot be sufficiently improved. On the other hand, when the content of these salts exceeds 5 parts by mass, the cross-linking reaction proceeds and the storage stability of the chemical conversion solution may be reduced.
化成処理液中の樹脂粒子(例えば、ポリエチレン-フッ素樹脂粒子)の含有量は、固形分(フッ素含有樹脂や4A族金属化合物など)100質量部に対して、0.5~20質量部の範囲内が好ましい。前述の通り、樹脂粒子の含有量が0.5質量%未満の場合、化成処理皮膜の潤滑性を十分に向上させることができない。一方、樹脂粒子の含有量が20質量%超の場合、化成処理皮膜の耐候性が低下してしまうおそれがある。
The content of the resin particles (for example, polyethylene-fluorine resin particles) in the chemical conversion liquid is in the range of 0.5 to 20 parts by mass with respect to 100 parts by mass of the solid content (fluorine-containing resin, 4A group metal compound, etc.) The inside is preferable. As described above, when the resin particle content is less than 0.5% by mass, the lubricity of the chemical conversion coating cannot be sufficiently improved. On the other hand, when the content of the resin particles is more than 20% by mass, the weather resistance of the chemical conversion film may be deteriorated.
ポリエチレン-フッ素樹脂粒子に加えて、さらにポリエチレン樹脂粒子を化成処理液に添加する場合、化成処理液中のポリエチレン樹脂粒子の含有量は、固形分100質量部に対して、0.1~16質量部の範囲内が好ましい。前述の通り、ポリエチレン樹脂粒子の含有量が0.1質量%未満の場合、ポリエチレン樹脂の効果を十分に発揮させることができない。一方、添加量が16質量%超の場合、化成処理皮膜の耐候性が低下してしまうおそれがある。また、ポリエチレン樹脂粒子の平均粒子径は、0.1~10μmの範囲内が好ましい。平均粒子径が0.1μm未満の場合、大部分のポリエチレン樹脂粒子が化成処理皮膜中に埋没してしまい、ポリエチレン樹脂を化成処理皮膜の表面にブリードさせることができない。一方、平均粒子径が10μm超の場合、化成処理液を乾燥させている間にポリエチレン樹脂粒子が脱落してしまうおそれがある。
When polyethylene resin particles are further added to the chemical conversion treatment liquid in addition to the polyethylene-fluorine resin particles, the content of the polyethylene resin particles in the chemical conversion treatment liquid is 0.1 to 16 masses with respect to 100 mass parts of the solid content. Within the range of parts is preferred. As described above, when the content of the polyethylene resin particles is less than 0.1% by mass, the effect of the polyethylene resin cannot be exhibited sufficiently. On the other hand, when the addition amount exceeds 16% by mass, the weather resistance of the chemical conversion coating may be lowered. The average particle diameter of the polyethylene resin particles is preferably in the range of 0.1 to 10 μm. When the average particle diameter is less than 0.1 μm, most of the polyethylene resin particles are buried in the chemical conversion film, and the polyethylene resin cannot be bleed on the surface of the chemical conversion film. On the other hand, when the average particle size is more than 10 μm, the polyethylene resin particles may fall off while the chemical conversion solution is being dried.
化成処理液にリン酸塩を添加する場合、化成処理液中のリン酸塩の含有量は、フッ素含有樹脂100質量部に対して、P換算で0.05~3質量部の範囲内が好ましい。リン酸塩の含有量が0.05質量部未満の場合、化成処理皮膜の密着性を十分に向上させることができない。一方、リン酸塩の含有量が3質量部超の場合、4A族金属化合物との反応が過剰に進行して、4A族金属化合物による架橋効果が損なわれてしまうおそれがある。
When adding a phosphate to the chemical conversion liquid, the phosphate content in the chemical conversion liquid is preferably in the range of 0.05 to 3 parts by mass in terms of P with respect to 100 parts by mass of the fluorine-containing resin. . When the phosphate content is less than 0.05 parts by mass, the adhesion of the chemical conversion film cannot be sufficiently improved. On the other hand, when the content of the phosphate is more than 3 parts by mass, the reaction with the 4A group metal compound may proceed excessively and the crosslinking effect by the 4A group metal compound may be impaired.
化成処理液にシランカップリング剤を添加する場合、化成処理液中のシランカップリング剤の含有量は、フッ素含有樹脂100質量部に対して、0.5~5質量部の範囲内が好ましい。シランカップリング剤の含有量が0.5質量部未満の場合、化成処理皮膜の密着性を十分に向上させることができない。一方、シランカップリング剤の含有量が5質量部超の場合、皮膜密着性は飽和し、それ以上の向上は認められない。また、化成処理液の安定性が低下してしまうおそれもある。
When a silane coupling agent is added to the chemical conversion solution, the content of the silane coupling agent in the chemical conversion solution is preferably in the range of 0.5 to 5 parts by mass with respect to 100 parts by mass of the fluorine-containing resin. When content of a silane coupling agent is less than 0.5 mass part, the adhesiveness of a chemical conversion treatment film cannot fully be improved. On the other hand, when the content of the silane coupling agent exceeds 5 parts by mass, the film adhesion is saturated and no further improvement is observed. In addition, the stability of the chemical conversion liquid may be reduced.
化成処理液には、その他の成分として、エッチング剤や無機化合物、無機潤滑剤、着色顔料、染料などを必要に応じて添加してもよい。エッチング剤としては、フッ化物などが使用される。エッチング剤は、めっき層表面を活性化することにより化成処理皮膜の密着性をより高める。MgやCa、Sr、V、W、Mn、B、Si、Snなどの無機化合物(酸化物、リン酸塩など)は、化成処理皮膜を緻密化して耐水性を向上させる。二硫化モリブデンやタルクなどの無機潤滑剤は、化成処理皮膜の潤滑性、さらには化成処理Zn系めっき鋼板の加工性をさらに向上させる。また、無機顔料や有機顔料、有機染料などを配合することで、化成処理皮膜に所定の色調を付与することができる。
Etching agents, inorganic compounds, inorganic lubricants, color pigments, dyes, and the like may be added to the chemical conversion treatment liquid as necessary as other components. Fluoride etc. are used as an etching agent. An etching agent improves the adhesiveness of a chemical conversion treatment film more by activating the plating layer surface. Inorganic compounds (oxides, phosphates, etc.) such as Mg, Ca, Sr, V, W, Mn, B, Si, and Sn improve the water resistance by densifying the chemical conversion film. Inorganic lubricants such as molybdenum disulfide and talc further improve the lubricity of the chemical conversion coating and, further, the workability of the chemical conversion Zn-plated steel sheet. Moreover, predetermined | prescribed color tone can be provided to a chemical conversion treatment film by mix | blending an inorganic pigment, an organic pigment, organic dye, etc.
[化成処理皮膜の形成]
第3のステップでは、第1のステップで準備したAl含有Zn系合金めっき鋼板の表面に、化成処理皮膜を形成する。化成処理皮膜を形成するには、第2のステップで準備した化成処理液を第1のステップで準備したAl含有Zn系合金めっき鋼板の表面に塗布し、乾燥させればよい。 [Formation of chemical conversion coating]
In the third step, a chemical conversion treatment film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step. In order to form the chemical conversion film, the chemical conversion solution prepared in the second step may be applied to the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step and dried.
第3のステップでは、第1のステップで準備したAl含有Zn系合金めっき鋼板の表面に、化成処理皮膜を形成する。化成処理皮膜を形成するには、第2のステップで準備した化成処理液を第1のステップで準備したAl含有Zn系合金めっき鋼板の表面に塗布し、乾燥させればよい。 [Formation of chemical conversion coating]
In the third step, a chemical conversion treatment film is formed on the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step. In order to form the chemical conversion film, the chemical conversion solution prepared in the second step may be applied to the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step and dried.
化成処理液の塗布方法は、特に限定されず、公知の方法から適宜選択すればよい。そのような塗布方法の例には、ロールコート法やカーテンフロー法、スピンコート法、スプレー法、浸漬引き上げ法などが含まれる。
The method for applying the chemical conversion liquid is not particularly limited, and may be appropriately selected from known methods. Examples of such a coating method include a roll coating method, a curtain flow method, a spin coating method, a spray method, and a dip pulling method.
化成処理液の乾燥は、常温乾燥であってもよいが、連続操業を考慮すると50℃以上に保持して乾燥時間を短縮することが好ましい。しかし、300℃超に保持した場合、有機成分が熱分解して化成処理皮膜の性能が低下するおそれがある。本発明の製造方法では、化成処理液中に含まれる乳化剤の含有量が少ないため、乾燥温度を50℃程度としても乳化剤がほとんど含まれず、耐水性に優れた化成処理皮膜を形成することができる。
The chemical conversion solution may be dried at room temperature, but considering continuous operation, it is preferable to keep the temperature at 50 ° C. or higher to shorten the drying time. However, when the temperature is kept above 300 ° C., the organic component may be thermally decomposed to deteriorate the performance of the chemical conversion coating. In the production method of the present invention, since the content of the emulsifier contained in the chemical conversion treatment liquid is small, even if the drying temperature is about 50 ° C., the emulsifier is hardly contained, and a chemical conversion treatment film having excellent water resistance can be formed. .
図2は、化成処理皮膜の形成過程を示す断面模式図である。図2では、ポリエチレン-フッ素樹脂粒子およびポリエチレン樹脂粒子を添加した化成処理液を塗布した様子を示している。
FIG. 2 is a schematic cross-sectional view showing the formation process of the chemical conversion coating. FIG. 2 shows a state in which a chemical conversion treatment liquid to which polyethylene-fluorine resin particles and polyethylene resin particles are added is applied.
まず、Al含有Zn系合金めっき鋼板110の表面に化成処理液を塗布して、化成処理液の塗膜120を形成する(図2A参照)。化成処理液の塗膜120には、ポリエチレン-フッ素樹脂粒子122およびポリエチレン樹脂粒子124が分散している。
First, a chemical conversion treatment liquid is applied to the surface of the Al-containing Zn-based alloy plated steel sheet 110 to form a coating film 120 of the chemical conversion treatment liquid (see FIG. 2A). Polyethylene-fluorine resin particles 122 and polyethylene resin particles 124 are dispersed in the coating film 120 of the chemical conversion treatment liquid.
次いで、50℃程度まで温度を上げると、水分が蒸発して、化成処理皮膜120’が形成される(図2B参照)。このとき、ポリエチレン-フッ素樹脂粒子122およびポリエチレン樹脂粒子124の大部分は、化成処理皮膜120’の表面から突出する。図3Aは、化成処理液の塗膜を50℃で乾燥させた後の、化成処理皮膜のSEM像(平面視)である。ポリエチレン-フッ素樹脂粒子とポリエチレン樹脂粒子とを区別することはできないが、これらの粒子が化成処理皮膜の表面から突出していることがわかる(図中「PE-FまたはPE」参照)。
Then, when the temperature is raised to about 50 ° C., the water evaporates and a chemical conversion treatment film 120 ′ is formed (see FIG. 2B). At this time, most of the polyethylene-fluorine resin particles 122 and the polyethylene resin particles 124 protrude from the surface of the chemical conversion coating 120 '. FIG. 3A is an SEM image (plan view) of the chemical conversion coating after the chemical conversion coating is dried at 50 ° C. FIG. Although it is not possible to distinguish between polyethylene-fluorine resin particles and polyethylene resin particles, it can be seen that these particles protrude from the surface of the chemical conversion coating (see “PE-F or PE” in the figure).
さらに、150℃程度まで温度を上げると、ポリエチレン樹脂粒子124が溶融して、ポリエチレン樹脂は表面にブリードする(図2C参照)。ブリードしたポリエチレン樹脂124’は、化成処理皮膜120’の表面の全部または一部を被覆する。一方、ポリエチレン-フッ素樹脂粒子122は、溶融することなくそのままの状態で維持される。図3Bは、化成処理液の塗膜を150℃で乾燥させた後の、化成処理皮膜のSEM像(平面視)である。ポリエチレン-フッ素樹脂粒子のみが化成処理皮膜の表面から突出していることがわかる(図中「PE-F」参照)。図3Cは、化成処理皮膜の表面から突出しているポリエチレン-フッ素樹脂粒子のSEM像(断面視)である。この化成処理皮膜の表面から突出している粒子が、ポリエチレン-フッ素樹脂粒子であることは、蛍光X線分析により確認することができる。図4は、化成処理皮膜の表面から突出しているポリエチレン-フッ素樹脂粒子の蛍光X線スペクトルである。
Furthermore, when the temperature is raised to about 150 ° C., the polyethylene resin particles 124 melt and the polyethylene resin bleeds to the surface (see FIG. 2C). The bleed polyethylene resin 124 ′ covers the whole or part of the surface of the chemical conversion coating 120 ′. On the other hand, the polyethylene-fluorine resin particles 122 are maintained as they are without melting. FIG. 3B is an SEM image (plan view) of the chemical conversion coating after the coating of the chemical conversion solution is dried at 150 ° C. It can be seen that only the polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating (see “PE-F” in the figure). FIG. 3C is an SEM image (cross-sectional view) of polyethylene-fluororesin particles protruding from the surface of the chemical conversion coating. It can be confirmed by fluorescent X-ray analysis that the particles protruding from the surface of the chemical conversion coating are polyethylene-fluororesin particles. FIG. 4 is a fluorescent X-ray spectrum of polyethylene-fluororesin particles protruding from the surface of the chemical conversion coating.
以上の手順により、耐候性、耐水性、耐黒変性、皮膜密着性および加工性のすべてに優れる本発明の化成処理Zn系めっき鋼板を製造することができる。
By the above procedure, the chemical conversion-treated Zn-based plated steel sheet of the present invention which is excellent in all of weather resistance, water resistance, blackening resistance, film adhesion and workability can be produced.
[下地化成処理皮膜の形成]
下地化成処理皮膜を形成するステップでは、化成処理皮膜を形成する前に、第1のステップで準備したAl含有Zn系合金めっき鋼板の表面に下地化成処理液を塗布して塗布膜を形成する。下地化成処理液は、例えば、ロールコート法、スピンコート法、スプレー法等などで塗布することができる。下地化成処理液の塗布量は、バルブメタル付着量が1mg/m2以上となるように調整することが好ましい。得られる化成処理鋼板に十分な耐食性を付与するためである。また、下地化成処理液の塗布量は、形成される下地化成処理皮膜の厚さが3nm以上1000nm以下となるように調整することが好ましい。3nm以上で十分な耐食性が発現し、1000nmを超えると鋼板を成形加工しようとするときに、応力によってクラックが発生するおそれがある。 [Formation of base chemical conversion coating]
In the step of forming the base chemical conversion coating, before the chemical conversion coating is formed, the base chemical conversion treatment liquid is applied to the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step to form a coating film. The base chemical conversion treatment liquid can be applied by, for example, a roll coating method, a spin coating method, a spray method, or the like. The coating amount of the base chemical conversion treatment liquid is preferably adjusted so that the valve metal adhesion amount is 1 mg / m 2 or more. It is for providing sufficient corrosion resistance to the chemical conversion treatment steel plate obtained. Moreover, it is preferable to adjust the application quantity of a foundation | substrate chemical conversion liquid so that the thickness of the foundation | substrate chemical conversion treatment film formed may be 3 nm or more and 1000 nm or less. Sufficient corrosion resistance is exhibited at 3 nm or more, and when it exceeds 1000 nm, cracks may occur due to stress when forming a steel sheet.
下地化成処理皮膜を形成するステップでは、化成処理皮膜を形成する前に、第1のステップで準備したAl含有Zn系合金めっき鋼板の表面に下地化成処理液を塗布して塗布膜を形成する。下地化成処理液は、例えば、ロールコート法、スピンコート法、スプレー法等などで塗布することができる。下地化成処理液の塗布量は、バルブメタル付着量が1mg/m2以上となるように調整することが好ましい。得られる化成処理鋼板に十分な耐食性を付与するためである。また、下地化成処理液の塗布量は、形成される下地化成処理皮膜の厚さが3nm以上1000nm以下となるように調整することが好ましい。3nm以上で十分な耐食性が発現し、1000nmを超えると鋼板を成形加工しようとするときに、応力によってクラックが発生するおそれがある。 [Formation of base chemical conversion coating]
In the step of forming the base chemical conversion coating, before the chemical conversion coating is formed, the base chemical conversion treatment liquid is applied to the surface of the Al-containing Zn-based alloy plated steel sheet prepared in the first step to form a coating film. The base chemical conversion treatment liquid can be applied by, for example, a roll coating method, a spin coating method, a spray method, or the like. The coating amount of the base chemical conversion treatment liquid is preferably adjusted so that the valve metal adhesion amount is 1 mg / m 2 or more. It is for providing sufficient corrosion resistance to the chemical conversion treatment steel plate obtained. Moreover, it is preferable to adjust the application quantity of a foundation | substrate chemical conversion liquid so that the thickness of the foundation | substrate chemical conversion treatment film formed may be 3 nm or more and 1000 nm or less. Sufficient corrosion resistance is exhibited at 3 nm or more, and when it exceeds 1000 nm, cracks may occur due to stress when forming a steel sheet.
Al含有Zn系合金めっき鋼板の表面に形成された塗布膜を、水洗することなく乾燥することにより、下地化成処理皮膜を形成することができる。常温で乾燥することもできるが、連続操業を考慮すると50℃以上に保持して乾燥時間を短縮することが好ましい。ただし、200℃を超える乾燥温度では、化成処理皮膜に含まれている有機成分が熱分解し、有機成分で付与された特性が損なわれることがある。
An undercoat chemical conversion coating can be formed by drying the coating film formed on the surface of the Al-containing Zn-based alloy-plated steel sheet without washing with water. Although it can dry at normal temperature, when continuous operation is considered, it is preferable to hold at 50 degreeC or more and to shorten drying time. However, when the drying temperature exceeds 200 ° C., the organic component contained in the chemical conversion film is thermally decomposed, and the properties imparted with the organic component may be impaired.
以下、本発明を実施例を参照して詳細に説明するが、本発明はこれらの実施例により限定されない。
Hereinafter, the present invention will be described in detail with reference to examples, but the present invention is not limited to these examples.
[実施例]
1.化成処理Zn系めっき鋼板の作製
板厚0.8mmのSPCCを基材として、以下の3種類の溶融Al含有Zn系合金めっき鋼板を作製した。本実施例では、これら3種類の溶融Al含有Zn系合金めっき鋼板を化成処理原板として使用した。
[Al含有Zn系合金めっき鋼板A]
・溶融Zn-0.18質量%Al合金めっき鋼板
・めっき付着量45g/m2
[Al含有Zn系合金めっき鋼板B]
・溶融Zn-6質量%Al-3質量%Mg合金めっき鋼板
・めっき付着量45g/m2
[Al含有Zn系合金めっき鋼板C]
・溶融Zn-55質量%Al合金めっき鋼板
・めっき付着量45g/m2 [Example]
1. Preparation of chemical conversion treated Zn-based plated steel sheet The following three types of molten Al-containing Zn-based alloy plated steel sheets were prepared using SPCC having a thickness of 0.8 mm as a base material. In this example, these three types of molten Al-containing Zn-based alloy-plated steel sheets were used as chemical conversion treatment original sheets.
[Al-containing Zn-based alloy-plated steel sheet A]
・ Fused Zn-0.18 mass% Al alloy-plated steel sheet ・ Plating adhesion 45 g / m 2
[Al-containing Zn-based alloy-plated steel sheet B]
・ Fused Zn-6 mass% Al-3 mass% Mg alloy plated steel sheet ・ Plating adhesion 45 g / m 2
[Al-containing Zn-based alloy plated steel sheet C]
・ Fused Zn-55 mass% Al alloy-plated steel sheet ・ Plating adhesion 45 g / m 2
1.化成処理Zn系めっき鋼板の作製
板厚0.8mmのSPCCを基材として、以下の3種類の溶融Al含有Zn系合金めっき鋼板を作製した。本実施例では、これら3種類の溶融Al含有Zn系合金めっき鋼板を化成処理原板として使用した。
[Al含有Zn系合金めっき鋼板A]
・溶融Zn-0.18質量%Al合金めっき鋼板
・めっき付着量45g/m2
[Al含有Zn系合金めっき鋼板B]
・溶融Zn-6質量%Al-3質量%Mg合金めっき鋼板
・めっき付着量45g/m2
[Al含有Zn系合金めっき鋼板C]
・溶融Zn-55質量%Al合金めっき鋼板
・めっき付着量45g/m2 [Example]
1. Preparation of chemical conversion treated Zn-based plated steel sheet The following three types of molten Al-containing Zn-based alloy plated steel sheets were prepared using SPCC having a thickness of 0.8 mm as a base material. In this example, these three types of molten Al-containing Zn-based alloy-plated steel sheets were used as chemical conversion treatment original sheets.
[Al-containing Zn-based alloy-plated steel sheet A]
・ Fused Zn-0.18 mass% Al alloy-plated steel sheet ・ Plating adhesion 45 g / m 2
[Al-containing Zn-based alloy-plated steel sheet B]
・ Fused Zn-6 mass% Al-3 mass% Mg alloy plated steel sheet ・ Plating adhesion 45 g / m 2
[Al-containing Zn-based alloy plated steel sheet C]
・ Fused Zn-55 mass% Al alloy-plated steel sheet ・ Plating adhesion 45 g / m 2
各Al含有Zn系合金めっき鋼板の表面に表1に示す組成の化成処理液を塗布し、到達板温140℃で加熱乾燥して、膜厚2.0μmの化成処理皮膜を形成した。
A chemical conversion treatment liquid having the composition shown in Table 1 was applied to the surface of each Al-containing Zn-based alloy-plated steel sheet, and dried by heating at a final plate temperature of 140 ° C. to form a chemical conversion treatment film having a thickness of 2.0 μm.
表1に示される処理液No.1~12の化成処理液は、所定量のカルボキシル基およびスルホン酸基を含むフッ素含有樹脂と、乳化剤とを含む水系エマルション(不揮発分25質量%;表2参照)に、4A族金属化合物、ポリエチレン-フッ素樹脂粒子(樹脂粒子)などを添加して調製した。処理液No.13の化成処理液は、ウレタン樹脂と乳化剤とを含む水系エマルション(不揮発分25質量%;表2参照)に、4A族金属化合物、ポリエチレン-フッ素樹脂粒子(樹脂粒子)などを添加して調製した。
The chemical conversion treatment liquids of treatment liquid Nos. 1 to 12 shown in Table 1 are aqueous emulsions containing a predetermined amount of a fluorine-containing resin containing a carboxyl group and a sulfonic acid group and an emulsifier (nonvolatile content: 25% by mass; see Table 2) 4A group metal compound, polyethylene-fluorine resin particles (resin particles) and the like. The chemical conversion treatment liquid of treatment liquid No. 13 is a 4A group metal compound, polyethylene-fluorine resin particles (resin particles), etc. added to an aqueous emulsion containing a urethane resin and an emulsifier (nonvolatile content: 25% by mass; see Table 2). Prepared.
フッ素含有樹脂を含む水系エマルションは、水溶媒に、所定量のフルオロオレフィン、カルボキシル基含有モノマー、スルホン酸基含有モノマーおよび乳化剤を添加して、それらを共重合反応させることで得た。ウレタン樹脂を含む水系エマルションは、PR135(住化バイエルンウレタン株式会社)を使用した。シランカップリング剤は、A-1891(モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社)を使用した。
An aqueous emulsion containing a fluorine-containing resin was obtained by adding a predetermined amount of a fluoroolefin, a carboxyl group-containing monomer, a sulfonic acid group-containing monomer and an emulsifier to an aqueous solvent and copolymerizing them. PR135 (Sumika Bayern Urethane Co., Ltd.) was used as an aqueous emulsion containing a urethane resin. A-1891 (Momentive Performance Materials Japan GK) was used as the silane coupling agent.
各化成処理Zn系めっき鋼板の化成処理皮膜における有機樹脂に対する4A族金属、リン酸塩およびシランカップリング剤の量を蛍光X線分析装置を用いて測定した。リン酸塩およびシランカップリング剤の含有量は、PおよびSiの測定値から算出した。また、各化成処理Zn系めっき鋼板の化成処理皮膜の表面におけるポリエチレン-フッ素樹脂粒子の面積占有率を走査型電子顕微鏡を用いて測定した。各化成処理液について、形成される化成処理皮膜における有機樹脂に対する4A族金属、リン酸塩およびシランカップリング剤の量、ならびに化成処理皮膜の表面におけるポリエチレン-フッ素樹脂粒子の面積占有率を表3に示す。
The amounts of Group 4A metal, phosphate, and silane coupling agent relative to the organic resin in the chemical conversion coating of each chemical conversion treatment Zn-plated steel sheet were measured using a fluorescent X-ray analyzer. The contents of phosphate and silane coupling agent were calculated from the measured values of P and Si. Further, the area occupancy ratio of the polyethylene-fluororesin particles on the surface of the chemical conversion coating of each chemical conversion treatment Zn-plated steel sheet was measured using a scanning electron microscope. Table 3 shows the amounts of Group 4A metal, phosphate, and silane coupling agent relative to the organic resin in the chemical conversion coating formed, and the area occupancy ratio of the polyethylene-fluororesin particles on the surface of the chemical conversion coating for each chemical conversion coating. Shown in
2.化成処理Zn系めっき鋼板の評価
(1)促進耐候性試験
各化成処理Zn系めっき鋼板から試験片を切り出し、JIS K5600-7-7:2008に準拠して促進耐候性試験(キセノンランプ法)を実施した。本試験では、キセノンアーク灯の光を120分間照射する間に18分間水を噴射する工程を1サイクル(2時間)とし、この工程を0~1000サイクル(0、500、1000サイクル)繰り返した。 2. Evaluation of chemical conversion treatment Zn-plated steel sheet (1) Accelerated weather resistance test A test piece was cut out from each chemical conversion treatment Zn-plated steel sheet and subjected to an accelerated weather resistance test (xenon lamp method) in accordance with JIS K5600-7-7: 2008. Carried out. In this test, the process of spraying water for 18 minutes while irradiating the light of a xenon arc lamp for 120 minutes was defined as 1 cycle (2 hours), and this process was repeated 0 to 1000 cycles (0, 500, 1000 cycles).
(1)促進耐候性試験
各化成処理Zn系めっき鋼板から試験片を切り出し、JIS K5600-7-7:2008に準拠して促進耐候性試験(キセノンランプ法)を実施した。本試験では、キセノンアーク灯の光を120分間照射する間に18分間水を噴射する工程を1サイクル(2時間)とし、この工程を0~1000サイクル(0、500、1000サイクル)繰り返した。 2. Evaluation of chemical conversion treatment Zn-plated steel sheet (1) Accelerated weather resistance test A test piece was cut out from each chemical conversion treatment Zn-plated steel sheet and subjected to an accelerated weather resistance test (xenon lamp method) in accordance with JIS K5600-7-7: 2008. Carried out. In this test, the process of spraying water for 18 minutes while irradiating the light of a xenon arc lamp for 120 minutes was defined as 1 cycle (2 hours), and this process was repeated 0 to 1000 cycles (0, 500, 1000 cycles).
(2)耐候性の評価
各化成処理Zn系めっき鋼板について、促進耐候試験前後の化成処理皮膜の厚さを断面検鏡により測定し、塗膜残存率を求めた。各化成処理Zn系めっき鋼板について、塗膜残存率が95%以上の場合は「◎」、80%以上95%未満の場合は「○」、60%以上80%未満の場合は「△」、30%以上60%未満の場合は「▲」、30%未満の場合は「×」と評価した。 (2) Evaluation of weather resistance About each chemical conversion treatment Zn-plated steel sheet, the thickness of the chemical conversion treatment film before and after the accelerated weathering test was measured with a cross-sectional microscope, and the coating film residual rate was determined. About each chemical conversion treatment Zn-plated steel sheet, when the coating film residual ratio is 95% or more, “◎”, when it is 80% or more and less than 95%, “◯”, when it is 60% or more and less than 80%, “△”, When it was 30% or more and less than 60%, “▲” was evaluated, and when it was less than 30%, “×” was evaluated.
各化成処理Zn系めっき鋼板について、促進耐候試験前後の化成処理皮膜の厚さを断面検鏡により測定し、塗膜残存率を求めた。各化成処理Zn系めっき鋼板について、塗膜残存率が95%以上の場合は「◎」、80%以上95%未満の場合は「○」、60%以上80%未満の場合は「△」、30%以上60%未満の場合は「▲」、30%未満の場合は「×」と評価した。 (2) Evaluation of weather resistance About each chemical conversion treatment Zn-plated steel sheet, the thickness of the chemical conversion treatment film before and after the accelerated weathering test was measured with a cross-sectional microscope, and the coating film residual rate was determined. About each chemical conversion treatment Zn-plated steel sheet, when the coating film residual ratio is 95% or more, “◎”, when it is 80% or more and less than 95%, “◯”, when it is 60% or more and less than 80%, “△”, When it was 30% or more and less than 60%, “▲” was evaluated, and when it was less than 30%, “×” was evaluated.
(3)耐黒変性の評価
各化成処理Zn系めっき鋼板について、促進耐候試験前後の化成処理皮膜の表面の明度差(ΔL*値)を測定し、耐黒変性を評価した。各化成処理Zn系めっき鋼板について、明度差(ΔL*値)が1以下の場合は「◎」、1を超え2以下の場合は「○」、2を超え5以下の場合は「△」、5を超え10以下の場合は「▲」、10を超える場合は「×」と評価した。 (3) Evaluation of blackening resistance About each chemical conversion treatment Zn-plated steel sheet, the lightness difference ((DELTA) L * value) of the surface of a chemical conversion treatment film before and behind an accelerated weathering test was measured, and blackening resistance was evaluated. About each chemical conversion treatment Zn-plated steel sheet, when the lightness difference (ΔL * value) is 1 or less, “◎”, when it exceeds 1 and 2 or less, “◯”, when it exceeds 2 and 5 or less, “△”, When it exceeded 5 and was 10 or less, it was evaluated as “▲” and when it exceeded 10, it was evaluated as “x”.
各化成処理Zn系めっき鋼板について、促進耐候試験前後の化成処理皮膜の表面の明度差(ΔL*値)を測定し、耐黒変性を評価した。各化成処理Zn系めっき鋼板について、明度差(ΔL*値)が1以下の場合は「◎」、1を超え2以下の場合は「○」、2を超え5以下の場合は「△」、5を超え10以下の場合は「▲」、10を超える場合は「×」と評価した。 (3) Evaluation of blackening resistance About each chemical conversion treatment Zn-plated steel sheet, the lightness difference ((DELTA) L * value) of the surface of a chemical conversion treatment film before and behind an accelerated weathering test was measured, and blackening resistance was evaluated. About each chemical conversion treatment Zn-plated steel sheet, when the lightness difference (ΔL * value) is 1 or less, “◎”, when it exceeds 1 and 2 or less, “◯”, when it exceeds 2 and 5 or less, “△”, When it exceeded 5 and was 10 or less, it was evaluated as “▲” and when it exceeded 10, it was evaluated as “x”.
(4)平坦部の耐食性の評価
各化成処理Zn系めっき鋼板について、促進耐候試験後の試験片を用いて塩水噴霧試験(JIS Z2371に準拠;120時間)を行い、平坦部の白錆発生面積率を評価した。各化成処理Zn系めっき鋼板について、白錆発生面積率が5%以下の場合は「◎」、5%を超え10%以下の場合は「○」、10%を超え30%以下の場合は「△」、30%を超え50%以下の場合は「▲」、50%を超える場合は「×」と評価した。 (4) Evaluation of corrosion resistance of flat part About each chemical conversion treatment Zn-plated steel sheet, a salt spray test (according to JIS Z2371; 120 hours) is performed using the test piece after the accelerated weathering test, and the white rust generation area of the flat part Rate was evaluated. About each chemical conversion treatment Zn-plated steel sheet, when the white rust generation area ratio is 5% or less, “◎”, when it exceeds 5% and 10% or less, “◯”, and when it exceeds 10% and 30% or less, “ “△” was evaluated as “▲” when 30% was exceeded and 50% or less, and “X” was evaluated when 50% was exceeded.
各化成処理Zn系めっき鋼板について、促進耐候試験後の試験片を用いて塩水噴霧試験(JIS Z2371に準拠;120時間)を行い、平坦部の白錆発生面積率を評価した。各化成処理Zn系めっき鋼板について、白錆発生面積率が5%以下の場合は「◎」、5%を超え10%以下の場合は「○」、10%を超え30%以下の場合は「△」、30%を超え50%以下の場合は「▲」、50%を超える場合は「×」と評価した。 (4) Evaluation of corrosion resistance of flat part About each chemical conversion treatment Zn-plated steel sheet, a salt spray test (according to JIS Z2371; 120 hours) is performed using the test piece after the accelerated weathering test, and the white rust generation area of the flat part Rate was evaluated. About each chemical conversion treatment Zn-plated steel sheet, when the white rust generation area ratio is 5% or less, “◎”, when it exceeds 5% and 10% or less, “◯”, and when it exceeds 10% and 30% or less, “ “△” was evaluated as “▲” when 30% was exceeded and 50% or less, and “X” was evaluated when 50% was exceeded.
(5)潤滑性の評価
各化成処理Zn系めっき鋼板から幅30mm、長さ300mmの試験片を切り出した。各試験片の両面にSKD11製の金型を接触させ、金型を介して600kgfの荷重を加えながら試験片を100mm/分の速度で引き抜いた。このときの引き抜き力を測定して、潤滑性を評価した。各化成処理Zn系めっき鋼板について、引き抜き力が200kgf以下の場合は「◎」、200kgfを超え250kgf以下の場合は「○」、250kgfを超え300kgf以下の場合は「△」、300kgfを超え400kgf以下の場合は「▲」、400kgfを超える場合は「×」と評価した。 (5) Evaluation of lubricity A test piece having a width of 30 mm and a length of 300 mm was cut out from each chemically treated Zn-based plated steel sheet. A mold made of SKD11 was brought into contact with both surfaces of each test piece, and the test piece was pulled out at a speed of 100 mm / min while applying a load of 600 kgf through the mold. The pulling force at this time was measured to evaluate the lubricity. About each chemical conversion treatment Zn-plated steel sheet, it is “は” when the pulling force is 200 kgf or less, “◯” when it exceeds 200 kgf and 250 kgf or less, “△” when it exceeds 250 kgf and 300 kgf or less, “△”, and exceeds 300 kgf and 400 kgf or less. In the case of, “「 ”was evaluated, and in the case of exceeding 400 kgf,“ × ”was evaluated.
各化成処理Zn系めっき鋼板から幅30mm、長さ300mmの試験片を切り出した。各試験片の両面にSKD11製の金型を接触させ、金型を介して600kgfの荷重を加えながら試験片を100mm/分の速度で引き抜いた。このときの引き抜き力を測定して、潤滑性を評価した。各化成処理Zn系めっき鋼板について、引き抜き力が200kgf以下の場合は「◎」、200kgfを超え250kgf以下の場合は「○」、250kgfを超え300kgf以下の場合は「△」、300kgfを超え400kgf以下の場合は「▲」、400kgfを超える場合は「×」と評価した。 (5) Evaluation of lubricity A test piece having a width of 30 mm and a length of 300 mm was cut out from each chemically treated Zn-based plated steel sheet. A mold made of SKD11 was brought into contact with both surfaces of each test piece, and the test piece was pulled out at a speed of 100 mm / min while applying a load of 600 kgf through the mold. The pulling force at this time was measured to evaluate the lubricity. About each chemical conversion treatment Zn-plated steel sheet, it is “は” when the pulling force is 200 kgf or less, “◯” when it exceeds 200 kgf and 250 kgf or less, “△” when it exceeds 250 kgf and 300 kgf or less, “△”, and exceeds 300 kgf and 400 kgf or less. In the case of, “「 ”was evaluated, and in the case of exceeding 400 kgf,“ × ”was evaluated.
(6)評価結果
各化成処理Zn系めっき鋼板(実施例1~21、比較例1~18)についての、化成処理原板の種類、使用した処理液の種類、ならびに耐候性試験、耐黒変性試験、平坦部耐食性試験および潤滑性試験の評価結果を表4および表5に示す。 (6) Evaluation Results For each chemical conversion treated Zn-based plated steel sheet (Examples 1 to 21 and Comparative Examples 1 to 18), the type of chemical conversion treatment original plate, the type of treatment liquid used, the weather resistance test, and the blackening resistance test Tables 4 and 5 show the evaluation results of the flat portion corrosion resistance test and the lubricity test.
各化成処理Zn系めっき鋼板(実施例1~21、比較例1~18)についての、化成処理原板の種類、使用した処理液の種類、ならびに耐候性試験、耐黒変性試験、平坦部耐食性試験および潤滑性試験の評価結果を表4および表5に示す。 (6) Evaluation Results For each chemical conversion treated Zn-based plated steel sheet (Examples 1 to 21 and Comparative Examples 1 to 18), the type of chemical conversion treatment original plate, the type of treatment liquid used, the weather resistance test, and the blackening resistance test Tables 4 and 5 show the evaluation results of the flat portion corrosion resistance test and the lubricity test.
耐候性は、促進耐候性試験後の化成処理皮膜の塗膜残存率により評価した。ウレタン樹脂を含む化成処理皮膜を形成した比較例6、比較例12および比較例18の化成処理Zn系めっき鋼板では、500サイクル(屋外暴露5年相当)で化成処理皮膜が消失してしまった。また、過剰に大きいポリエチレン-フッ素樹脂粒子を含む化成処理皮膜を形成した比較例4、比較例10および比較例16の化成処理Zn系めっき鋼板でも、ポリエチレン-フッ素樹脂粒子が化成処理皮膜から脱落してしまったため、耐候性が劣っていた。一方、所定量の親水性官能基を有するフッ素含有樹脂および4A族金属化合物を含む化成処理皮膜を形成した実施例1~21の化成処理Zn系めっき鋼板では、1000サイクル(屋外暴露10年相当)繰り返した後でも化成処理皮膜の膜厚はほとんど変化しなかった。
The weather resistance was evaluated by the coating film remaining rate of the chemical conversion coating after the accelerated weather resistance test. In the chemical conversion treatment Zn-plated steel sheets of Comparative Example 6, Comparative Example 12 and Comparative Example 18 in which a chemical conversion treatment film containing a urethane resin was formed, the chemical conversion treatment film disappeared after 500 cycles (equivalent to outdoor exposure for 5 years). Further, in the conversion-treated Zn-based plated steel sheets of Comparative Example 4, Comparative Example 10 and Comparative Example 16 in which the chemical conversion treatment film containing excessively large polyethylene-fluororesin particles was formed, the polyethylene-fluororesin particles dropped off from the chemical conversion treatment film. As a result, the weather resistance was poor. On the other hand, in the chemical conversion-treated Zn-based plated steel sheets of Examples 1 to 21 in which a chemical conversion film containing a fluorine-containing resin having a predetermined amount of a hydrophilic functional group and a group 4A metal compound was formed, 1000 cycles (equivalent to 10 years of outdoor exposure) Even after repeating, the film thickness of the chemical conversion film hardly changed.
耐黒変性は、促進耐候性試験前後の明度差(ΔL*値)により評価した。ウレタン樹脂を含む化成処理皮膜を形成した比較例6、比較例12および比較例18の化成処理Zn系めっき鋼板では、サイクル数の増加に伴いめっき層の黒変が進行して、明度が低下してしまった。一方、所定量の親水性官能基を有するフッ素含有樹脂および4A族金属化合物を含む化成処理皮膜を形成した実施例1~21の化成処理Zn系めっき鋼板では、1000サイクル(屋外暴露10年相当)繰り返した後でも明度はほとんど低下しなかった。
Blackening resistance was evaluated by the difference in brightness (ΔL * value) before and after the accelerated weather resistance test. In the chemical conversion treatment Zn-plated steel sheets of Comparative Example 6, Comparative Example 12 and Comparative Example 18 in which a chemical conversion treatment film containing a urethane resin was formed, as the number of cycles increased, the blackening of the plating layer progressed and the brightness decreased. I have. On the other hand, in the chemical conversion-treated Zn-based plated steel sheets of Examples 1 to 21 in which a chemical conversion film containing a fluorine-containing resin having a predetermined amount of a hydrophilic functional group and a group 4A metal compound was formed, 1000 cycles (equivalent to 10 years of outdoor exposure) Even after repeating, the brightness was hardly lowered.
耐食性は、塩水噴霧試験後の白錆発生面積率により評価した。ウレタン樹脂を含む化成処理皮膜を形成した比較例6、比較例12および比較例18の化成処理Zn系めっき鋼板では、促進耐候性試験前は耐食性が良好であったものの、皮膜の消失に伴い耐食性が著しく低下してしまった。また、過剰量または過少量の親水性官能基を有するフッ素含有樹脂を含む化成処理皮膜を形成した比較例1、比較例2、比較例7、比較例8、比較例13および比較例14の化成処理Zn系めっき鋼板、ならびに4A族金属化合物を含まない化成処理皮膜を形成した比較例3、比較例5、比較例9、比較例11、比較例15および比較例17の化成処理Zn系めっき鋼板では、促進耐候性試験前から耐食性が劣っていた。一方、所定量の親水性官能基を有するフッ素含有樹脂および4A族金属化合物を含む化成処理皮膜を形成した実施例1~21の化成処理Zn系めっき鋼板では、1000サイクル(屋外暴露10年相当)繰り返した後でも耐食性が良好であった。
Corrosion resistance was evaluated based on the white rust generation area ratio after the salt spray test. In the chemical conversion treatment Zn-plated steel sheets of Comparative Example 6, Comparative Example 12 and Comparative Example 18 in which a chemical conversion treatment film containing a urethane resin was formed, although the corrosion resistance was good before the accelerated weather resistance test, the corrosion resistance was accompanied by the disappearance of the film. Has fallen significantly. Further, the chemical conversion of Comparative Example 1, Comparative Example 2, Comparative Example 7, Comparative Example 8, Comparative Example 13, and Comparative Example 14 in which a chemical conversion treatment film containing a fluorine-containing resin having an excessive amount or a small amount of a hydrophilic functional group was formed. Chemically treated Zn-plated steel sheets of Comparative Example 3, Comparative Example 5, Comparative Example 9, Comparative Example 11, Comparative Example 15, Comparative Example 15 and Comparative Example 17 in which a treated Zn-based plated steel sheet and a chemical conversion film not containing a 4A group metal compound were formed Then, corrosion resistance was inferior before the accelerated weather resistance test. On the other hand, in the chemical conversion-treated Zn-based plated steel sheets of Examples 1 to 21 in which a chemical conversion film containing a fluorine-containing resin having a predetermined amount of a hydrophilic functional group and a group 4A metal compound was formed, 1000 cycles (equivalent to 10 years of outdoor exposure) Even after repetition, the corrosion resistance was good.
潤滑性は、荷重を加えてられている試験片を引き抜く際に要する引き抜き力により評価した。比較例1、比較例7および比較例13の化成処理Zn系めっき鋼板は、化成処理皮膜中のポリエチレン-フッ素樹脂粒子の量が少ないため、潤滑性が劣っていた。同様に、ポリエチレン-フッ素樹脂粒子を含まない比較例5、比較例11および比較例17の化成処理Zn系めっき鋼板も、潤滑性が劣っていた。また、比較例6、比較例12および比較例18の化成処理Zn系めっき鋼板は、ポリエチレン-フッ素樹脂粒子の平均粒子径が小さく、化成処理皮膜中に埋没してしまったため、潤滑性が劣っていた。一方、平均粒子径が所定の範囲内のポリエチレン-フッ素樹脂粒子を所定量含む化成処理皮膜を形成した実施例1~21の化成処理Zn系めっき鋼板では、潤滑性が良好であった。
Lubricity was evaluated by the pulling force required for pulling out a test piece to which a load was applied. The chemical conversion-treated Zn-plated steel sheets of Comparative Example 1, Comparative Example 7 and Comparative Example 13 were inferior in lubricity because the amount of polyethylene-fluororesin particles in the chemical conversion coating was small. Similarly, the chemically treated Zn-based plated steel sheets of Comparative Example 5, Comparative Example 11 and Comparative Example 17 that did not contain polyethylene-fluorine resin particles also had poor lubricity. Further, the chemical conversion-treated Zn-based plated steel sheets of Comparative Example 6, Comparative Example 12 and Comparative Example 18 have poor lubricity because the average particle diameter of the polyethylene-fluorine resin particles is small and buried in the chemical conversion film. It was. On the other hand, the chemical conversion treatment Zn-plated steel sheets of Examples 1 to 21 in which the chemical conversion treatment film containing a predetermined amount of polyethylene-fluororesin particles having an average particle diameter within a predetermined range was excellent in lubricity.
以上の結果から、本発明の化成処理Zn系めっき鋼板は、耐候性、耐黒変性および加工性(潤滑性)に優れていることがわかる。
From the above results, it can be seen that the chemically treated Zn-based plated steel sheet of the present invention is excellent in weather resistance, blackening resistance and workability (lubricity).
[参考実験]
参考実験として、フッ素樹脂皮膜中の4A族金属化合物の量および乳化剤の量と、透湿度との関係を調べた結果を示す。 [Reference experiment]
As a reference experiment, the results of examining the relationship between the moisture permeability and the amount of the group 4A metal compound and the amount of the emulsifier in the fluororesin film are shown.
参考実験として、フッ素樹脂皮膜中の4A族金属化合物の量および乳化剤の量と、透湿度との関係を調べた結果を示す。 [Reference experiment]
As a reference experiment, the results of examining the relationship between the moisture permeability and the amount of the group 4A metal compound and the amount of the emulsifier in the fluororesin film are shown.
親水性官能基含有モノマーを1質量%となるように添加し、乳化剤を1質量%となるように添加して作製した親水性官能基を有するフッ素含有樹脂の水系エマルションに、所定量の4A族金属化合物を添加して調製した化成処理液を、めっき鋼板の表面にバーコーターで塗布し、到達板温140℃で加熱乾燥して、膜厚30μmのフッ素樹脂皮膜を形成した。このフッ素樹脂皮膜をめっき鋼板から剥がし、所定の大きさに切り出して試験片とした。各試験片(遊離フッ素樹脂皮膜)について、JIS Z0208に準拠して透湿度を測定した(測定条件、温度40±0.5℃、相対湿度90±2%、24時間)。
A predetermined amount of group 4A was added to an aqueous emulsion of a fluorine-containing resin having a hydrophilic functional group prepared by adding a hydrophilic functional group-containing monomer to 1% by mass and adding an emulsifier to 1% by mass. The chemical conversion treatment solution prepared by adding the metal compound was applied to the surface of the plated steel plate with a bar coater and dried by heating at a final plate temperature of 140 ° C. to form a fluororesin film having a thickness of 30 μm. This fluororesin film was peeled off from the plated steel sheet and cut into a predetermined size to obtain a test piece. About each test piece (free fluororesin film | membrane), the water vapor transmission rate was measured based on JIS Z0208 (measuring conditions, temperature 40 +/- 0.5 degreeC, relative humidity 90 +/- 2%, 24 hours).
図5は、フッ素樹脂皮膜における4A族金属の量と透湿度との関係を示すグラフである。このグラフから、フッ素樹脂皮膜中の4A族金属の量を0.1質量%以上とすることで、フッ素樹脂皮膜の透湿度を顕著に低下させうることがわかる。
FIG. 5 is a graph showing the relationship between the amount of group 4A metal in the fluororesin film and the water vapor transmission rate. From this graph, it is understood that the moisture permeability of the fluororesin film can be remarkably lowered by setting the amount of the group 4A metal in the fluororesin film to 0.1 mass% or more.
親水性官能基含有モノマーを1質量%となるように添加し、所定量の乳化剤を添加して作製した親水性官能基を有するフッ素含有樹脂の水系エマルションに、4A族金属化合物を終濃度が金属換算で1質量%となるように添加して調製した化成処理液を、めっき鋼板の表面にバーコーターで塗布し、到達板温140℃で加熱乾燥して、膜厚30μmのフッ素樹脂皮膜を形成した。このフッ素樹脂皮膜をめっき鋼板から剥がし、所定の大きさに切り出して試験片とした。各試験片(遊離フッ素樹脂皮膜)について、JIS Z0208に準拠して透湿度を測定した(測定条件、温度40±0.5℃、相対湿度90±2%、24時間)。
Add a hydrophilic functional group-containing monomer to 1% by mass and add a predetermined amount of emulsifier to an aqueous emulsion of a fluorine-containing resin having a hydrophilic functional group. A chemical conversion treatment solution prepared by adding 1% by mass in terms of conversion is applied to the surface of the plated steel plate with a bar coater and dried by heating at a final plate temperature of 140 ° C. to form a fluororesin film having a thickness of 30 μm. did. This fluororesin film was peeled off from the plated steel sheet and cut into a predetermined size to obtain a test piece. About each test piece (free fluororesin film | membrane), the water vapor transmission rate was measured based on JIS Z0208 (measuring conditions, temperature 40 +/- 0.5 degreeC, relative humidity 90 +/- 2%, 24 hours).
図6は、フッ素含有樹脂の水系エマルション中の乳化剤の濃度とフッ素樹脂皮膜の透湿度との関係を示すグラフである。このグラフから、エマルション中の乳化剤の濃度を1質量%以下とすることで、フッ素樹脂皮膜の透湿度を顕著に低下させうることがわかる。
FIG. 6 is a graph showing the relationship between the concentration of the emulsifier in the aqueous emulsion of the fluorine-containing resin and the moisture permeability of the fluorine resin film. From this graph, it is understood that the moisture permeability of the fluororesin film can be remarkably reduced by setting the concentration of the emulsifier in the emulsion to 1% by mass or less.
以上の結果から、4A族金属化合物の量が多く、乳化剤の残存量が少ないフッ素樹脂皮膜は、耐水性に優れていることがわかる。
From the above results, it can be seen that the fluororesin film having a large amount of the group 4A metal compound and a small amount of the remaining emulsifier has excellent water resistance.
本出願は、2011年9月13日出願の特願2011-199465に基づく優先権を主張する。当該出願明細書および図面に記載された内容は、すべて本願明細書に援用される。
This application claims priority based on Japanese Patent Application No. 2011-199465 filed on September 13, 2011. The contents described in the application specification and the drawings are all incorporated herein by reference.
本発明の化成処理Zn系めっき鋼板は、耐候性、耐水性、耐黒変性、皮膜密着性および加工性に優れているため、外装建材などの様々な用途において有用である。たとえば、本発明の化成処理Zn系めっき鋼板は、1)ビニールハウスまたは農業ハウス用の鋼管、形鋼、支柱、梁、搬送用部材、2)遮音壁、防音壁、吸音壁、防雪壁、ガードレール、高欄、防護柵、支柱、3)鉄道車両用部材、架線用部材、電気設備用部材、安全環境用部材、構造用部材、太陽光架台などの用途に好適に使用されうる。
The chemical conversion-treated Zn-based plated steel sheet of the present invention is useful in various applications such as exterior building materials because it is excellent in weather resistance, water resistance, blackening resistance, film adhesion and workability. For example, the chemical conversion-treated Zn-based plated steel sheet of the present invention includes: 1) steel pipe, shape steel, support, beam, conveying member for a greenhouse or agricultural house, 2) sound insulation wall, sound insulation wall, sound absorption wall, snow insulation wall, guardrail, It can be suitably used for applications such as railings, protective fences, columns, 3) railway vehicle members, overhead wire members, electrical equipment members, safety environment members, structural members, and solar mounts.
Al含有Zn系合金めっき鋼板は、高温高湿下において塗膜と十分に密着するとともに、耐食性に優れる。よって、本発明の化成処理Zn系めっき鋼板は、高温高湿環境下で使用される外装用材料として特に好適である。
The Al-containing Zn-based alloy-plated steel sheet is sufficiently in close contact with the coating film under high temperature and high humidity and has excellent corrosion resistance. Therefore, the chemical conversion treatment Zn-plated steel sheet of the present invention is particularly suitable as an exterior material used in a high temperature and high humidity environment.
110 Al含有Zn系合金めっき鋼板
120 化成処理液の塗膜
120’ 化成処理皮膜
122 ポリエチレン-フッ素樹脂粒子
124 ポリエチレン樹脂粒子
124’ ブリードしたポリエチレン樹脂 110 Al-containing Zn-based alloy platedsteel sheet 120 Chemical conversion coating film 120 'Chemical conversion coating film 122 Polyethylene-fluorine resin particles 124 Polyethylene resin particles 124' Bleed polyethylene resin
120 化成処理液の塗膜
120’ 化成処理皮膜
122 ポリエチレン-フッ素樹脂粒子
124 ポリエチレン樹脂粒子
124’ ブリードしたポリエチレン樹脂 110 Al-containing Zn-based alloy plated
Claims (16)
- Alを0.05~60質量%含むZn系合金めっき層を有するAl含有Zn系合金めっき鋼板と、
前記Al含有Zn系合金めっき鋼板の表面に形成された、膜厚0.5~10μmの化成処理皮膜と、を有する化成処理Zn系めっき鋼板であって、
前記化成処理皮膜は、カルボキシル基、スルホン酸基およびこれらの塩からなる群から選ばれる親水性官能基0.05~5質量%とF原子7~20質量%とを含有するフッ素含有樹脂と、前記フッ素含有樹脂に対して金属換算で0.1~5質量%の4A族金属化合物と、平均粒子径0.1~10μmの樹脂粒子とを含有し、
前記化成処理皮膜の表面における前記樹脂粒子の面積占有率は、0.1面積%以上である、
化成処理Zn系めっき鋼板。 An Al-containing Zn-based alloy plated steel sheet having a Zn-based alloy plating layer containing 0.05 to 60% by mass of Al;
A chemical conversion-treated Zn-based plated steel sheet having a film thickness of 0.5 to 10 μm formed on the surface of the Al-containing Zn-based alloy-plated steel sheet,
The chemical conversion film comprises a fluorine-containing resin containing 0.05 to 5% by mass of a hydrophilic functional group selected from the group consisting of a carboxyl group, a sulfonic acid group and salts thereof, and 7 to 20% by mass of F atoms, Containing 0.1 to 5% by mass of a 4A group metal compound in terms of metal with respect to the fluorine-containing resin, and resin particles having an average particle size of 0.1 to 10 μm;
The area occupation ratio of the resin particles on the surface of the chemical conversion film is 0.1 area% or more.
Chemical conversion treatment Zn-plated steel sheet. - 前記フッ素含有樹脂が有するカルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内である、請求項1に記載の化成処理Zn系めっき鋼板。 The chemical conversion-treated Zn-based plated steel sheet according to claim 1, wherein the ratio of carboxyl group to sulfonic acid group of the fluorine-containing resin is in the range of 5 to 60 in terms of a molar ratio of carboxyl group / sulfonic acid group.
- 前記樹脂粒子は、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子が結合されているポリエチレン-フッ素樹脂粒子である、請求項1に記載の化成処理Zn系めっき鋼板。 The chemical conversion-treated Zn-based plated steel sheet according to claim 1, wherein the resin particles are polyethylene-fluorine resin particles in which fluorine resin fine particles are bonded to the surface of polyethylene resin particles.
- 前記化成処理皮膜は、さらにポリエチレン樹脂を含有し、
前記ポリエチレン-フッ素樹脂粒子は、前記化成処理皮膜の表面の一部において、前記化成処理皮膜の表面から突出しており、
前記ポリエチレン樹脂は、前記化成処理皮膜の表面の残部の全部または一部を被覆している、
請求項3に記載の化成処理Zn系めっき鋼板。 The chemical conversion film further contains a polyethylene resin,
The polyethylene-fluororesin particles protrude from the surface of the chemical conversion coating on a part of the surface of the chemical conversion coating,
The polyethylene resin covers all or part of the remaining part of the surface of the chemical conversion film,
The chemical conversion treatment Zn-plated steel sheet according to claim 3. - 前記化成処理皮膜は、さらにリン酸塩を含有し、
前記フッ素含有樹脂に対する前記リン酸塩の量は、P換算で0.05~3質量%の範囲内である、
請求項1に記載の化成処理Zn系めっき鋼板。 The chemical conversion film further contains a phosphate,
The amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P.
The chemical conversion treatment Zn-plated steel sheet according to claim 1. - 前記化成処理皮膜は、さらにシランカップリング剤を含有し、
前記フッ素含有樹脂に対する前記シランカップリング剤の量は、0.5~5質量%の範囲内である、
請求項1に記載の化成処理Zn系めっき鋼板。 The chemical conversion film further contains a silane coupling agent,
The amount of the silane coupling agent relative to the fluorine-containing resin is in the range of 0.5 to 5% by mass.
The chemical conversion treatment Zn-plated steel sheet according to claim 1. - 前記4A族金属は、Ti、Zr、Hfおよびこれらの組み合わせからなる群から選ばれる、請求項1に記載の化成処理Zn系めっき鋼板。 The said 4A group metal is a chemical conversion treatment Zn-plated steel plate of Claim 1 selected from the group which consists of Ti, Zr, Hf, and these combination.
- 前記Al含有Zn系合金めっき鋼板と、前記化成処理皮膜との間に形成された、
バルブメタルの酸化物または水酸化物、およびバルブメタルのフッ化物を含有する下地化成処理皮膜をさらに有する、請求項1に記載の化成処理Zn系めっき鋼板。 Formed between the Al-containing Zn-based alloy-plated steel sheet and the chemical conversion film,
The chemical conversion treatment Zn-plated steel plate of Claim 1 which further has the foundation | substrate chemical conversion treatment film containing the oxide or hydroxide of valve metal, and the fluoride of valve metal. - Alを0.05~60質量%含むZn系合金めっき層を有するAl含有Zn系合金めっき鋼板を準備するステップと、
前記Al含有Zn系合金めっき鋼板の表面に化成処理液を塗布し、乾燥させて、膜厚が0.5~10μmの化成処理皮膜を形成するステップとを含み、
前記化成処理液は、カルボキシル基、スルホン酸基およびこれらの塩からなる群から選ばれる親水性官能基0.05~5質量%とF原子7~20質量%とを含有し、数平均分子量が1000~200万の範囲内であるフッ素含有樹脂と、4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩のいずれかと、平均粒子径が0.1~10μmの樹脂粒子とを含有し、
前記フッ素含有樹脂に対する、前記4A族金属の酸素酸塩、フッ化物、水酸化物、有機酸塩、炭酸塩または過酸化塩の量は、金属換算で0.1~5質量%の範囲内であり、
前記化成処理液中の固形分に対する、前記樹脂粒子の量は、0.5~20質量%の範囲内である、
化成処理Zn系めっき鋼板の製造方法。 Preparing an Al-containing Zn-based alloy-plated steel sheet having a Zn-based alloy plating layer containing 0.05 to 60% by mass of Al;
Applying a chemical conversion treatment liquid to the surface of the Al-containing Zn-based alloy plated steel sheet and drying to form a chemical conversion treatment film having a thickness of 0.5 to 10 μm.
The chemical conversion treatment solution contains 0.05 to 5% by mass of a hydrophilic functional group selected from the group consisting of a carboxyl group, a sulfonic acid group and a salt thereof and 7 to 20% by mass of F atoms, and has a number average molecular weight. Fluorine-containing resin in the range of 1,000 to 2,000,000, and group 4A metal oxyacid salt, fluoride, hydroxide, organic acid salt, carbonate or peroxide salt, and an average particle size of 0.1 Containing ~ 10 μm resin particles,
The amount of the oxyacid salt, fluoride, hydroxide, organic acid salt, carbonate or peroxide of the group 4A metal in the fluorine-containing resin is within the range of 0.1 to 5% by mass in terms of metal. Yes,
The amount of the resin particles relative to the solid content in the chemical conversion treatment liquid is in the range of 0.5 to 20% by mass.
The manufacturing method of a chemical conversion treatment Zn-plated steel plate. - 前記フッ素含有樹脂が有するカルボキシル基とスルホン酸基との比率は、カルボキシル基/スルホン酸基のモル比で5~60の範囲内である、請求項9に記載の化成処理Zn系めっき鋼板の製造方法。 The process for producing a chemically treated Zn-based plated steel sheet according to claim 9, wherein a ratio of carboxyl groups to sulfonic acid groups of the fluorine-containing resin is within a range of 5 to 60 in terms of a molar ratio of carboxyl groups / sulfonic acid groups. Method.
- 前記樹脂粒子は、ポリエチレン樹脂粒子の表面にフッ素樹脂微粒子が結合されているポリエチレン-フッ素樹脂粒子である、請求項9に記載の化成処理Zn系めっき鋼板の製造方法。 The method for producing a chemically treated Zn-based plated steel sheet according to claim 9, wherein the resin particles are polyethylene-fluorine resin particles in which fluorine resin fine particles are bonded to the surface of polyethylene resin particles.
- 前記化成処理液は、さらにポリエチレン樹脂粒子を含有する、請求項11に記載の化成処理Zn系めっき鋼板の製造方法。 The method for producing a chemical conversion-treated Zn-based plated steel sheet according to claim 11, wherein the chemical conversion treatment liquid further contains polyethylene resin particles.
- 前記化成処理液は、さらにリン酸塩を含有し、
前記フッ素含有樹脂に対する前記リン酸塩の量は、P換算で0.05~3質量%の範囲内である、
請求項9に記載の化成処理Zn系めっき鋼板の製造方法。 The chemical conversion treatment liquid further contains a phosphate,
The amount of the phosphate with respect to the fluorine-containing resin is in the range of 0.05 to 3% by mass in terms of P.
The manufacturing method of the chemical conversion treatment Zn-plated steel plate of Claim 9. - 前記化成処理液は、さらにシランカップリング剤を含有し、
前記フッ素含有樹脂に対する前記シランカップリング剤の量は、0.5~5質量%の範囲内である、
請求項9に記載の化成処理Zn系めっき鋼板の製造方法。 The chemical conversion treatment liquid further contains a silane coupling agent,
The amount of the silane coupling agent relative to the fluorine-containing resin is in the range of 0.5 to 5% by mass.
The manufacturing method of the chemical conversion treatment Zn-plated steel plate of Claim 9. - 前記4A族金属は、Ti、Zr、Hfおよびこれらの組み合わせからなる群から選ばれる、請求項9に記載の化成処理Zn系めっき鋼板の製造方法。 The said 4A group metal is a manufacturing method of the chemical conversion treatment Zn-plated steel plate of Claim 9 chosen from the group which consists of Ti, Zr, Hf, and these combination.
- 前記化成処理皮膜を形成するステップの前に、
前記Al含有Zn系合金めっき鋼板の表面に下地化成処理液を塗布し、乾燥させて、下地化成処理皮膜を形成するステップをさらに含み、
前記下地化成処理液は、バルブメタル塩とフッ化物イオンとを含有する、請求項9に記載の化成処理Zn系めっき鋼板の製造方法。 Before the step of forming the chemical conversion film,
Applying a base chemical conversion treatment liquid on the surface of the Al-containing Zn-based alloy-plated steel sheet and drying to further form a base chemical conversion treatment film;
The said base chemical conversion liquid is a manufacturing method of the chemical conversion treatment Zn-plated steel plate of Claim 9 containing a valve metal salt and fluoride ion.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-199465 | 2011-09-13 | ||
| JP2011199465A JP5674605B2 (en) | 2011-09-13 | 2011-09-13 | Chemical conversion treated steel sheet and method for producing the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013038643A1 true WO2013038643A1 (en) | 2013-03-21 |
Family
ID=47882894
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/005722 WO2013038643A1 (en) | 2011-09-13 | 2012-09-10 | Chemical-conversion-treatment-plated steel sheet and method for manufacturing same |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JP5674605B2 (en) |
| TW (1) | TWI531677B (en) |
| WO (1) | WO2013038643A1 (en) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5808626B2 (en) * | 2011-09-14 | 2015-11-10 | 日新製鋼株式会社 | Chemical conversion treated steel sheet and method for producing the same |
| EP3604615B1 (en) * | 2017-03-30 | 2021-07-14 | JFE Steel Corporation | Zinc-plated steel sheet and production method therefor |
| JP6962216B2 (en) * | 2018-01-24 | 2021-11-05 | 日本製鉄株式会社 | Anti-rust treatment liquid for welded steel pipes, chemical conversion treatment methods for welded steel pipes, molded products of welded steel pipes and welded steel pipes |
| JP6962215B2 (en) * | 2018-01-24 | 2021-11-05 | 日本製鉄株式会社 | End face rust preventive treatment liquid for plated steel sheet, chemical conversion treatment method for end face of plated steel sheet, chemical conversion treated steel sheet and molded products |
| KR102428620B1 (en) * | 2018-05-25 | 2022-08-03 | 닛폰세이테츠 가부시키가이샤 | surface treatment steel plate |
| JP2020037659A (en) * | 2018-09-05 | 2020-03-12 | Dicグラフィックス株式会社 | Aqueous coating agent, and printed matter coated therewith |
| JP7332948B2 (en) * | 2019-11-21 | 2023-08-24 | 日本製鉄株式会社 | Non-oriented electrical steel sheet and manufacturing method thereof |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0328380A (en) * | 1989-06-26 | 1991-02-06 | Kawasaki Steel Corp | Surface treated steel sheet having excellent lubricity and corrosion resistance |
| JP2001072819A (en) * | 1999-09-06 | 2001-03-21 | Daikin Ind Ltd | Aqueous dispersion composition of cross-linking fluororesin |
| JP2003105555A (en) * | 2001-07-23 | 2003-04-09 | Nkk Corp | Surface treated steel sheet having excellent white rust resistance, and production method therefor |
| JP2003293169A (en) * | 2002-04-04 | 2003-10-15 | Furukawa Electric Co Ltd:The | FLUORORESIN-COATED Al MATERIAL |
| JP2004501252A (en) * | 2000-06-22 | 2004-01-15 | ポーハング アイアン アンド スティール シーオー.,エルティディ. | Method for producing aqueous urethane resin composition |
| JP2006175817A (en) * | 2004-12-24 | 2006-07-06 | Nippon Steel Corp | Coated metal sheet good in reflectivity and molding processability, and its manufacturing method |
| JP2007098864A (en) * | 2005-10-06 | 2007-04-19 | Kobe Steel Ltd | Precoated metal sheet and its manufacturing method |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06248227A (en) * | 1993-02-24 | 1994-09-06 | Asahi Glass Co Ltd | Fluororesin coating composition |
| JP2004250560A (en) * | 2003-02-19 | 2004-09-09 | Nippon Yushi Basf Coatings Kk | Water-based coating material composition |
| JP2005138294A (en) * | 2003-11-04 | 2005-06-02 | Nisshin Steel Co Ltd | Coated metal sheet excellent in lubricity and processability |
| JP2005162993A (en) * | 2003-12-05 | 2005-06-23 | Daikin Ind Ltd | Aqueous fluorine-containing curable composition |
| CN103069047A (en) * | 2010-06-18 | 2013-04-24 | 日新制钢株式会社 | Chemical conversion coated plated steel sheet and method for producing same |
-
2011
- 2011-09-13 JP JP2011199465A patent/JP5674605B2/en active Active
-
2012
- 2012-09-10 WO PCT/JP2012/005722 patent/WO2013038643A1/en active Application Filing
- 2012-09-12 TW TW101133355A patent/TWI531677B/en not_active IP Right Cessation
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0328380A (en) * | 1989-06-26 | 1991-02-06 | Kawasaki Steel Corp | Surface treated steel sheet having excellent lubricity and corrosion resistance |
| JP2001072819A (en) * | 1999-09-06 | 2001-03-21 | Daikin Ind Ltd | Aqueous dispersion composition of cross-linking fluororesin |
| JP2004501252A (en) * | 2000-06-22 | 2004-01-15 | ポーハング アイアン アンド スティール シーオー.,エルティディ. | Method for producing aqueous urethane resin composition |
| JP2003105555A (en) * | 2001-07-23 | 2003-04-09 | Nkk Corp | Surface treated steel sheet having excellent white rust resistance, and production method therefor |
| JP2003293169A (en) * | 2002-04-04 | 2003-10-15 | Furukawa Electric Co Ltd:The | FLUORORESIN-COATED Al MATERIAL |
| JP2006175817A (en) * | 2004-12-24 | 2006-07-06 | Nippon Steel Corp | Coated metal sheet good in reflectivity and molding processability, and its manufacturing method |
| JP2007098864A (en) * | 2005-10-06 | 2007-04-19 | Kobe Steel Ltd | Precoated metal sheet and its manufacturing method |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013060624A (en) | 2013-04-04 |
| TWI531677B (en) | 2016-05-01 |
| JP5674605B2 (en) | 2015-02-25 |
| TW201323657A (en) | 2013-06-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5328980B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| JP5328981B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| JP5674605B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| JP5595305B2 (en) | Welded steel pipe | |
| JP5837245B1 (en) | Chemical conversion treated steel sheet, method for producing the same, and chemical conversion treatment liquid | |
| JP5575009B2 (en) | Formed product of plated steel sheet, method for producing the same, and chemical conversion treatment liquid | |
| JP5469556B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| JP6271062B1 (en) | Water-based treatment liquid, chemical conversion treatment method, and chemical conversion treatment steel plate | |
| JP5808626B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| TWI791746B (en) | Antirust treatment liquid for welded steel pipes, chemical conversion treatment method for welded steel pipes, welded steel pipes and formed products of welded steel pipes | |
| JP5469577B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| JP5631231B2 (en) | Chemical conversion Zn-plated steel sheet and method for producing the same | |
| JP5674606B2 (en) | Chemical conversion treated steel sheet and method for producing the same | |
| WO2015145514A1 (en) | Chemical conversion coated steel sheet, and manufacturing method and chemical conversion solution therefor | |
| JP5631239B2 (en) | Chemical conversion Al-plated steel sheet and method for producing the same | |
| JP2019127617A (en) | End face rustproofing liquid for plated sheet steel, chemical conversion method for end face of plated sheet steel, chemical conversion steel sheet and molded product |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12831504 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12831504 Country of ref document: EP Kind code of ref document: A1 |