WO2012168289A2 - Topical composition - Google Patents
Topical composition Download PDFInfo
- Publication number
- WO2012168289A2 WO2012168289A2 PCT/EP2012/060676 EP2012060676W WO2012168289A2 WO 2012168289 A2 WO2012168289 A2 WO 2012168289A2 EP 2012060676 W EP2012060676 W EP 2012060676W WO 2012168289 A2 WO2012168289 A2 WO 2012168289A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- topical composition
- hydrogen
- composition according
- alkyl
- derivative
- Prior art date
Links
- 0 *C(*)c1cc(*)cc(-[n]2nc(cc(*)cc3)c3n2)c1O Chemical compound *C(*)c1cc(*)cc(-[n]2nc(cc(*)cc3)c3n2)c1O 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/41—Amines
- A61K8/415—Aminophenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/35—Ketones, e.g. benzophenone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
- A61K8/496—Triazoles or their condensed derivatives, e.g. benzotriazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
- A61K8/4966—Triazines or their condensed derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/10—General cosmetic use
Definitions
- the present invention relates to topical compositions comprising at least one benzotriazol derivative and at least one UV filter substance selected from the group consisting of a triazine derivative and an amino substituted hydroxybenzophenone as well as mixtures thereof.
- UV-A radiation is equally or even more important in the development of solar damage and skin diseases, such as lupus erythematosus and melanoma and non-melanoma skin cancer.
- SPF's Sun Protection Factor
- Sun care products often exhibit a significant stickiness which results in the adherence of sand onto the skin which is highly undesirable.
- compositions comprising specific benzotriazol derivatives in combination with a UV filter substance selected from the group consisting of triazine derivatives and/ or an amino substituted hydroxybenzophenone exhibit a significantly reduced stickiness.
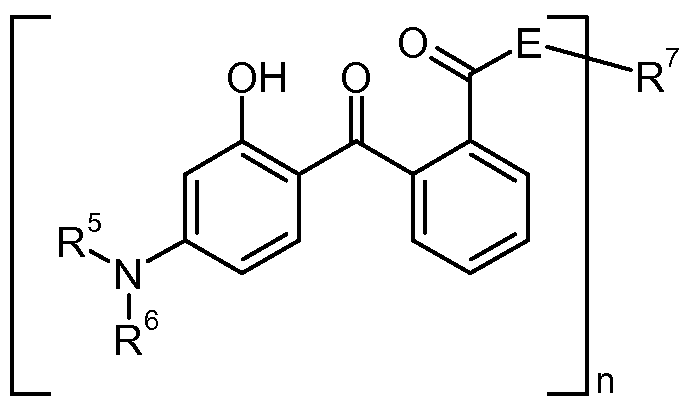
- the invention relates in one aspect to topical compositions comprising at least one benzotriazol derivative of formula (I) and at least one UV filter substance selected from the group consisting of triazine derivatives and/ or an amino substituted hydroxybenzophenone
- R 1 is hydrogen; Ci -5 alkyl; Ci -5 alkoxy or halogen; preferably hydrogen or chloride; most preferably hydrogen;
- R 2 is hydrogen; Ci -2 oalkyl; Ci -5 alkoxy; Ci -5 alkoxycarbonyl; C 5 -iocycloalkyl; C 6 -ioaryl or aralkyl; preferably hydrogen or Ci -5 alkyl; most preferably methyl;
- R 3 is Ci-2oalkyl, C 5 -iocycloalkyl, Ci -2 oalkoxy or C 5 -iocycloalkoxy, preferably C 5- i5alkyl or C 5- i 5 alkoxy;
- R 4 is hydrogen or Ci -5 alkyl; preferably hydrogen.
- compositions in addition contain a phosphate ester emulsifier.
- compositions do not contain a phosphate ester emulsifier.
- compositions furthermore comprise a phosphate ester surfactant.
- compositions do not contain a phosphate ester emulsifier.
- the invention relates to the use of a benzotriazol derivative of formula (I) as sand repellent.
- compositions according to the present invention furthermore comprise a dibenzoylmethane derivative, such as particularly butyl methoxy dibenzoylmethane.
- a dibenzoylmethane derivative such as particularly butyl methoxy dibenzoylmethane.
- C x -C y alkyl refers to straight-chain or branched alkyl radicals having x to y carbon atoms such as e.g.
- C 5 -iocycloalkyl denotes to unsubstituted or Ci-i 0 alkyl (mono- or poly-)substituted, in particular Ci -5 alkyl (mono- or poly-)substituted cyclic, bicyclic or tricyclic hydrocarbon residues such as in particular cyclopentyl, cyclohexyl, cycoheptyl or decahydronaphtyl.
- C 5-10 cycloalkyl denotes to unsubstituted or C 1-2 alkyl (mono- or poly-)substituted cyclopentyl, cyclohexyl or cycoheptyl such as in particular to unsubstituted or (mono- or poly-)methyl substituted cyclohexyl such as most in particular cyclohexyl or 3,3,5-trimethyl- cyclohexyl.
- C x -C y alkoxy denotes to linear or branched alkoxy-, respectively unsubstituted or (mono- or poly-)substituted cycloalkoxy radicals having from x to y carbon atoms such as e.g. methoxy, ethoxy, propoxy, butyloxy or pentyloxy, 2,5,5-trimethylhexyloxy, 3,5,5-trimethylhexyloxy, isoamyloxy, 2-ethylhexyloxy or 3,3,5- trimethyl-cyclohexyloxy.
- C 6- ioaryl refers e.g. to naphthyl or phenyl radicals, preferably phenyl.
- R 5 and R 6 independently of each other are hydrogen; d-C 2 oalkyl; C 2 -C 2 oalkenyl; C 5 - Ciocycloalkyl or C 5 -Ci 0 cycloalkenyl; or R 5 and R 6 , together with the nitrogen atom they are bound to, form a 5 to 6 membered ring;
- n is an integer from 1 or 2;
- E is -O- or -N(R 8 )-and
- R 8 is hydrogen; CrC 5 alkyl; or CrC 5 hydroxyalkyl; with the proviso that
- R 7 is Ci-C 2 oalkyl; C 2 -C 20 alkenyl; CrC 5 hydroxyalkyl; C 5 -Ci 0 cycloalkyl; C 5 -Ci 0 cycloalkeny; C 6- ioaryl; or aralkyl optionally substituted by O, N or S; or a Ci-C 5 aminocarbonyl or alkylcarbonyl radical;
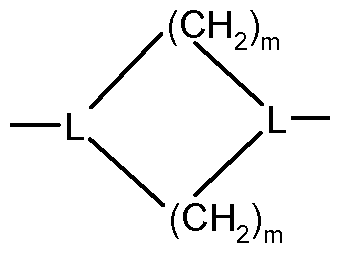
- R 7 is an CrC 20 alkyl; C 5 -Ci 0 cycloalkyl-; C 2 -C 20 alkenyl- or aryl-diradical or R 7 with E forms a diradical of formula (III) resulting in a compound of formula (IV)
- n 1 and 3.
- a particular suitable amino substituted hydroxybenzophenone derivative according to the present invention is diethylamino hydroxybenzoyi hexyl benzoate [lUPAC Name: Hexyl 2-[4- (diethylamino)-2-hydroxybenzoyl]benzoate] sold under the tradename Uvinul ® A plus by BASF SE Ludwigshafen.
- Another suitable amino substituted hydroxybenzophenone derivative according to the present invention is 1 , 1 '-(1 ,4-piperazinediyl)bis[1-[2-[4-(diethylamino)-2- hydroxybenzoyl]phenyl]-methanone [CAS No 919803-06-8] which is e.g. disclosed in WO 2007071584.
- triazine derivatives are ethylhexyl triazone (Uvinul ® T-150), diethylhexyl butamido triazone (Uvasorb ® HEB) and bis-ethylhexyloxyphenol methoxyphenyl triazine (Tinosorb ® S) such as most preferably bis-ethylhexyloxyphenol methoxyphenyl triazine.
- the amount of the at least one benzotriazol derivative of formula (I) in the compositions according to the present invention is preferable selected in the range of 1 to 20 wt.-%, such as preferably 2 to 20 wt.-%, such as more preferably in the range of 2 to 15 wt.-% and in particular in the range of 4 to 10 wt.-%, and most in particular in the range of 4 to 8 wt.-% based on the total weight of the composition.
- the amount of the amino substituted hydroxybenzophenone derivative such as in particular of diethylamino hydroxybenzoyl hexyl benzoate in the compositions according to the invention is preferable selected in the range of 0.5 to 8 wt.-%, in particular in the range of 1 to 5 wt.-%, most particular in the range of 3 to 5 wt.-% based on the total weight of the composition.
- the amount of the triazine derivative such as in particular of bis-ethylhexyloxyphenol methoxyphenyl triazine in the compositions according to the invention is preferable selected in the range of 0.5 to 8 wt.-%, in particular in the range of 1 to 5 wt.-%, most particular in the range of 2 to 4 wt.-% based on the total weight of the composition.
- the benzotriazol derivative is selected from compounds of formula (I) wherein R 1 and R 4 are hydrogen, R 2 is methyl and R 3 is C 5- ioalkoxy such as preferably C 6 -ioalkoxy, or C 6 cycloalkoxy such as in particular 2,5,5-trimethylhexyloxy, 3,5,5-trimethylhexyloxy, isoamyloxy, 2-ethylhexyloxy or 3,3,5- trimethyl-cyclohexyloxy.
- R 1 and R 4 are hydrogen
- R 2 is methyl
- R 3 is C 5- ioalkoxy such as preferably C 6 -ioalkoxy, or C 6 cycloalkoxy such as in particular 2,5,5-trimethylhexyloxy, 3,5,5-trimethylhexyloxy, isoamyloxy, 2-ethylhexyloxy or 3,3,5- trimethyl-cyclohexyloxy.
- the compound of formula (I) is a compound wherein R 1 and R 4 are hydrogen, R 2 is methyl and R 3 is undecyl (C11 H2 3 ) which is commercially available as Tinogard TL [INCI Name: benzotriazolyl dodecyl p-cresol; lUPAC Name: 2-(2H-benzotriazol-2-yl)-6-dodecyl-4-methyl-phenol] at BASF SE Ludwigshafen.
- the benzotriazol derivative of formula (I) is benzotriazolyl dodecyl p-cresol
- the triazine derivative is bis-ethylhexyloxyphenol methoxyphenyl triazine
- the amino substituted hydroxybenzophenone derivative is diethylamino hydroxybenzoyl hexyl benzoate.
- the topical composition furthermore comprises butyl methoxydibenzoylmethane.
- benzotriazol derivative of formula (I) is selected from compounds of formula (I) wherein R 1 and R 4 are hydrogen, R 2 is methyl and R 3 is C 5-10 alkoxy such as in particular C 6- ioalkoxy, or C 6 cycloalkoxy such as most in particular 2,5,5-trimethylhexyloxy, 3,5,5-trimethylhexyloxy, isoamyloxy, 2-ethylhexyloxy or 3,3,5-trimethyl-cyclohexyloxy and the triazine derivative is bis-ethylhexyloxyphenol methoxyphenyl triazine and the amino substituted hydroxybenzophenone derivative is diethylamino hydroxybenzoyl hexyl benzoate.
- topical composition furthermore comprises butyl methoxydibenzoylmethane.
- compositions according to the present invention are substantially free of a polyglycerol based UV-filter such as e.g.
- EP09178503.0, EP09178501 .4, EP09178502.2 EP09178495.9, EP09178506.3, EP09178505.5 or EP10150832.3 which are obtainable by a process comprising the steps of ring-opening polymerization of x mol equivalents of glycidol using 1 mol equivalent of a polyol starter unit with y mol equivalents hydroxyl-groups, followed by block copolymerization with z X (x+y) mole equivalents of propylene oxide to form a hyperbranched polyether-polyol backbone carrying (x+y) mol equivalents hydroxyl-groups followed by partial or total esterification, respectively partial or total etherification of the hydroxyl groups with a UV-light absorbing chromophore such as particularly with p-dimethylamino benzoic acid, 3-[1 -(4-Hydroxymethyl-phenyl)-meth-(E)-
- Particular suitable dibenzoylmethane derivatives to be used in the topical compositions according to the present invention encompass 2-methyldibenzoylmethane,
- the amount of the dibenzoylmethane derivative such as in particular of butyl methoxydibenzoylmethane in the compositions according to the invention is preferable selected in the range of 2 to 8 wt.-%, in particular in the range of 3 to 6 wt.-%, most particular in the range of 3 to 5 wt.-% based on the total weight of the composition.
- phosphate ester emulsifier refers to phosphate esters emulsifier of formula (II) formula (II) wherein R 5 , R 6 and R 7 may be hydrogen, an alkyl of from 1 to 22 carbons, preferably from 12 to 18 carbons; or an alkoxylated alkyl having 1 to 22 carbons, preferably from 12 to 18 carbons, and having 1 or more, preferably from 2 to 25, most preferably 2 to 12, moles ethylene oxide, with the provision that at least one of R 5 , R 6 and R 7 is an alkyl or alkoxylated alkyl as previously defined but having at least 6 alkyl carbons in said alkyl or alkoxylated alkyl group.
- Monoesters in which R 5 and R 6 are hydrogen and R 7 is selected from alkyl groups of 10 to 18 carbons and alkoxylated fatty alcohols of 10 to 18 carbons and 2 to 12 moles ethylene oxide are preferred.
- the preferred phosphate ester emulsifier are C 8- io Alkyl Ethyl Phosphate, C 9- i 5 Alkyl Phosphate, Ceteareth-2 Phosphate, Ceteareth-5 Phosphate, Ceteth- 8 Phosphate, Ceteth-10 Phosphate, Cetyl Phosphate, C6-10 Pareth-4 Phosphate, C12-15 Pareth-2 Phosphate, C12-15 Pareth-3 Phosphate, DEA-Ceteareth-2 Phosphate, DEA-Cetyl Phosphate, DEA-Oleth-3 Phosphate, Potassium cetyl phosphate, Deceth-4 P
- a particular phosphate ester emulsifier according to the invention is potassium cetyl phosphate e.g. commercially available as Amphisol ® K at DSM Nutritional Products Ltd Kaiseraugst.
- the term "topical” is understood here to mean external application to keratinous substances, which are in particular the skin, scalp, eyelashes, eyebrows, nails, mucous membranes and hair.
- compositions according to the invention are intended for topical application, they comprise a physiologically acceptable medium, that is to say a medium compatible with keratinous substances, such as the skin, mucous membranes, and keratinous fibres.
- physiologically acceptable medium is a cosmetically acceptable carrier.
- cosmetically acceptable carrier refers to all carriers and/or excipients and/ or diluents conventionally used in cosmetic compositions.
- Preferred topical compositions according to the invention are skin care preparations, decorative preparations, and functional preparations.
- skin care preparations are, in particular, light protective preparations, anti- ageing preparations, preparations for the treatment of photo-ageing, body oils, body lotions, body gels, treatment creams, skin protection ointments, skin powders, moisturizing gels, moisturizing sprays, face and/or body moisturizers, skin-tanning preparations (i.e. compositions for the artificial/sunless tanning and/or browning of human skin), for example self-tanning creams as well as skin lightening preparations.
- Examples of decorative preparations are, in particular, lipsticks, eye shadows, mascaras, dry and moist make-up formulations, rouges and/or powders.
- Examples of functional preparations are cosmetic or pharmaceutical compositions containing active ingredients such as hormone preparations, vitamin preparations, vegetable extract preparations, anti-ageing preparations, and/or antimicrobial (antibacterial or antifungal) preparations without being limited thereto.
- the topical compositions according to the invention are light- protective preparations (sun care products), such as sun protection milks, sun protection lotions, sun protection creams, sun protection oils, sun blocks or tropical's or day care creams with a SPF (sun protection factor).
- light- protective preparations such as sun protection milks, sun protection lotions, sun protection creams, sun protection oils, sun blocks or tropical's or day care creams with a SPF (sun protection factor).
- SPF sun protection factor
- compositions according to the present invention may be in the form of a suspension or dispersion in solvents or fatty substances, or alternatively in the form of an emulsion or micro emulsion (in particular of oil-in-water (0/W-) or water-in-oil (VWO-)type, silicone-in-water (Si/W-) or water-in-silicone (W/Si-)type, PIT-emulsion, multiple emulsion (e.g.
- compositions according to the present invention are advantageously in the form of an oil-in-water (O/W) emulsion comprising an oily phase dispersed in an aqueous phase in the presence of an O/W emulsifier.
- O/W emulsifier is no phosphate ester emulsifier.
- the topical composition according to the invention is an O/W emulsion, then it contains advantageously at least one O/W- or Si/W-emulsifier selected from the list of PEG-30 Dipolyhydroxystearate, PEG-4 Dilaurate, PEG-8 Dioleate, PEG-40 Sorbitan Peroleate, PEG-7 Glyceryl Cocoate, PEG-20 Almond Glycerides, PEG-25 Hydrogenated Castor Oil, Glyceryl Stearate (and) PEG-100 Stearate , PEG-7 Olivate, PEG-8 Oleate, PEG-8 Laurate, PEG-60 Almond Glycerides, PEG-20 Methyl Glucose Sesquistearate, PEG-40 Stearate, PEG-100 Stearate, PEG-80 Sorbitan Laurate, Steareth-2, Steareth-12, Oleth-2, Ceteth-2, Laureth-4, Oleth-10, Oleth-10/Polyoxyl 10 Oleyl Ether
- emulsifiers are sorbitan oleate, sorbitan sesquioleate, sorbitan isostearate, sorbitan trioleate, Lauryl Glucoside, Decyl Glucoside, Sodium Stearoyl Glutamate, Sucrose Polystearate and Hydrated Polyisobuten.
- one or more synthetic polymers may be used as an emulsifier.
- PVP eicosene copolymer acrylates/Cio-3o alkyl acrylate crosspolymer, acrylates/steareth-20 methacrylate copolymer, PEG-22/dodecyl glycol copolymer, PEG-45/dodecyl glycol copolymer, and mixtures thereof.
- the at least one O/W respectively Si/W emulsifier is preferably used in an amount of 0.5 to 10 wt.-% such as in particular in the range of 0.5 to 5 wt.-% such as most in particular in the range of 1 to 4 wt.-% based on the total weight of the composition.
- Suitable O/W emulsifiers are polyethyleneglycol (PEG) esters or diesters such as e.g. [I NCI Names] PEG-100 Stearate, PEG-30 Dipolyhydroxystearate, PEG-4 Dilaurate, PEG-8 Dioleate, PEG-40 Sorbitan Peroleate, PEG-7 Glyceryl Cocoate, PEG-20 Almond Glycerides, PEG-25 Hydrogenated Castor Oil, PEG-7 Olivate, PEG-8 Oleate, PEG-8 Laurate, PEG-60 Almond Glycerides, PEG-20 Methyl Glucose Sesquistearate, PEG-40 Stearate, PEG-100 Stearate, PEG-80 Sorbitan Laurate.
- PEG polyethyleneglycol
- PEG-100 Stearate sold under the tradename ArlacelTM 165 (INCI Glyceryl Stearate (and) PEG-100 Stearate) by Croda.
- O/W emulsifiers are non ionic self-emulsifying system derived from olive oil e.g. known as (INCI Name) cetearyl olivate and sorbitan olivate (Chemical Composition: sorbitan ester and cetearyl ester of olive oil fatty acids) sold under the tradename OLIVEM 1000.
- polymeric emulsifiers such as hydrophobically modified polyacrylic acid such as Acrylates/CI O-30 Alkyl Acrylate Crosspolymers which are commercially available under the tradename Pemulen ® TR-1 and TR-2 by Noveon.
- polyglycerol esters or diesters of fatty acids also called polyglyceryl ester/ diester (i.e. a polymer in which fatty acid(s) is/ are bound by esterification with polyglycerine), such as e.g. commercially available at Evonik as Isolan GPS [INCI Name Polyglyceryl-4 Diisostearate/Polyhydroxystearate/Sebacate (i.e. diester of a mixture of isostearic, polyhydroxystearic and sebacic acids with Polyglycerin-4)] or Dehymuls PGPH available at Cognis (I NCI Polyglyceryl-2 Dipolyhydroxystearate).
- polyalkylenglycolether such as Brij 72 (Polyoxyethylen(2)stearylether) or Brij 721 (Polyoxyethylene (21 ) Stearyl Ether e.g. available at Croda.
- compositions according to the present invention are advantageously in the form of an oil-in-water (O/W) emulsion comprising an oily phase dispersed in an aqueous phase in the presence of an O/W emulsifier selected from the group consisting of polyalkylenglycolether, polyethyleneglycol (PEG) ester or diester, non ionic self-emulsifying system derived from olive oil, as well as hydrophobically modified polyacrylic acid such as preferably from polyalkylenglycolether, polyethyleneglycol (PEG) ester or diester, non ionic self-emulsifying system derived from olive oil as well as mixtures thereof.
- O/W emulsions is well known to a person skilled in the art and illustrated in the examples.
- the invention relates to topical compositions in the form of O/W emulsions comprising an oily phase dispersed in an aqueous phase in the presence of an O/W emulsifier wherein the benzotriazol derivative of formula (I) is benzotriazolyl dodecyl p- cresol, the triazine derivative is bis-ethylhexyloxyphenol methoxyphenyl triazine and the amino substituted hydroxybenzophenone derivative is diethylamino hydroxybenzoyl hexyl benzoate and the O/W emulsifier is selected from the group consisting of polyalkylenglycolether, polyethyleneglycol (PEG) esters or diesters, as well as non ionic self-emulsifying systems derived from olive oil such as particularly Arlacel 165, Olivem 1000, Brij72, and Brij 721 as well as mixtures thereof. It is particularly preferred if the topical composition furthermore comprises butyl
- the invention relates to topical compositions in the form of O/W emulsions comprising an oily phase dispersed in an aqueous phase in the presence of an O/W emulsifier wherein the benzotriazol derivative of formula (I) is a compound of formula (I) wherein R 1 and R 4 are hydrogen, R 2 is methyl and R 3 is 2,5,5-trimethylhexyloxy, 3,5,5- trimethylhexyloxy, isoamyloxy, 2-ethylhexyloxy or 3,3,5-trimethyl-cyclohexyloxy, the triazine derivative is bis-ethylhexyloxyphenol methoxyphenyl triazine and the amino substituted hydroxybenzophenone derivative is diethylamino hydroxybenzoyl hexyl benzoate and the O/W emulsifier is selected from the group consisting of polyalkylenglycolether, polyethyleneglycol (PEG) esters
- compositions according to the present invention are advantageously in the form of a water-in-oil (W/O) emulsion comprising a water phase dispersed in an oily phase in the presence of an W/O emulsifier selected from the group consisting of polyglyceryl esters/ diesters.
- W/O water-in-oil
- the preparation of such W/O emulsions is well known to a person skilled in the art and illustrated in the examples.
- the invention relates to topical compositions in the form of water- in-oil (W/O) emulsion comprising a water phase dispersed in an oily phase in the presence of an W/O emulsifier wherein the benzotriazol derivative of formula (I) is benzotriazolyl dodecyl p-cresol, the triazine derivative is bis-ethylhexyloxyphenol methoxyphenyl triazine and the amino substituted hydroxybenzophenone derivative is diethylamino hydroxybenzoyl hexyl benzoate and the W/O emulsifier is selected from the group consisting of polyglyceryl esters or diesters such as particularly Dehymuls PGPH. It is particularly preferred if the topical composition furthermore comprises butyl methoxydibenzoylmethane.
- W/O emulsifier is selected from the group consisting of polyglyceryl esters or diesters such as particularly Dehy
- the invention relates to topical compositions in the form of water- in-oil (W/O) emulsion comprising a water phase dispersed in an oily phase in the presence of an W/O emulsifier wherein the benzotriazol derivative of formula (I) is a compound of formula (I) wherein R 1 and R 4 are hydrogen, R 2 is methyl and R 3 is 2,5,5-trimethylhexyloxy, 3,5,5-trimethylhexyloxy, isoamyloxy, 2-ethylhexyloxy or 3,3,5-trimethyl-cyclohexyloxy, the triazine derivative is bis-ethylhexyloxyphenol methoxyphenyl triazine and the amino substituted hydroxybenzophenone derivative is diethylamino hydroxybenzoyl hexyl benzoate and the W/O emulsifier is selected from the group consisting of polyglyceryl esters or diesters such as particularly De
- the topical compositions according to the present invention furthermore advantageously contain at least one co-surfactant such as e.g. selected from the group of mono- and diglycerides and/ or fatty alcohols.
- the co-surfactant is generally used in an amount selected in the range of 0.1 to 10 wt.-%, such as in particular in the range of 0.5 to 5 wt.-%, such as most in particular in the range of 1 to 3 wt.-%, based on the total weight of the composition.
- Particular suitable co-surfactants are selected from the list of alkyl alcohols such as cetyl alcohol (Lorol C16, Lanette 16), cetearyl alcohol (Lanette O), stearyl alcohol (Lanette 18), behenyl alcohol (Lanette 22), glyceryl stearate, glyceryl myristate (Estol 3650), hydrogenated coco-glycerides (Lipocire Na10) as well as mixtures thereof
- compositions in form of O/W emulsions according to the invention can be provided, for example, in all the formulation forms for O/W emulsions, for example in the form of serum, milk or cream, and they are prepared according to the usual methods.
- the compositions which are subject-matters of the invention are intended for topical application and can in particular constitute a dermatological or cosmetic composition, for example intended for protecting human skin against the adverse effects of UV radiation (antiwrinkle, anti-ageing, moisturizing, anti-sun protection and the like).
- the compositions constitute cosmetic composition and are intended for topical application to the skin.
- a subject-matter of the invention is a method for the cosmetic treatment of keratinous substances such as in particular the skin, characterized in that a composition as defined above is applied to the said keratinous substances such as in particular to the skin.
- the method is in particular suitable to protect the skin against the adverse effects of UV- radiation such as in particular sun-burn and/ or photoageing.
- compositions according to the invention may comprise further ingredients such as ingredients for skin lightening; tanning prevention; treatment of hyperpigmentation; preventing or reducing acne, wrinkles, lines, atrophy and/or inflammation; chelators and/or sequestrants; anti-cellulites and slimming (e.g. phytanic acid), firming, moisturizing and energizing, self tanning, soothing, as well as agents to improve elasticity and skin barrier and/or further UV-filter substances and carriers and/or excipients or diluents conventionally used in topical compositions. If nothing else is stated, the excipients, additives, diluents, etc.
- compositions according to the present invention are suitable for topical compositions according to the present invention.
- the necessary amounts of the cosmetic and dermatological adjuvants and additives can, based on the desired product, easily be determined by the skilled person.
- the additional ingredients can either be added to the oily phase, the aqueous phase or separately as deemed appropriate.
- the mode of addition can easily be adapted by a person skilled in the art.
- the cosmetically active ingredients useful herein can in some instances provide more than one benefit or operate via more than one mode of action.
- the topical cosmetic compositions of the invention can also contain usual cosmetic adjuvants and additives, such as preservatives/ antioxidants, fatty substances/ oils, water, organic solvents, silicones, thickeners, softeners, emulsifiers, sunscreens, antifoaming agents, moisturizers, aesthetic components such as fragrances, surfactants, fillers, sequestering agents, anionic, cationic, nonionic or amphoteric polymers or mixtures thereof, propellants, acidifying or basifying agents, dyes, colorings/colorants, abrasives, absorbents, essential oils, skin sensates, astringents, antifoaming agents, pigments or nanopigments, e.g.
- cosmetic adjuvants and additives such as preservatives/ antioxidants, fatty substances/ oils, water, organic solvents, silicones, thickeners, softeners, emulsifiers, sunscreens, antifoaming agents, moisturizers, aesthetic components such as fragrances, surfactants
- cosmetic ingredients those suited for providing a photoprotective effect by physically blocking out ultraviolet radiation, or any other ingredients usually formulated into cosmetic compositions.
- Such cosmetic ingredients commonly used in the skin care industry, which are suitable for use in the compositions of the present invention are e.g. described in the CTFA Cosmetic Ingredient Handbook, Second Edition (1992), The Cosmetic, Toiletry and Fragrance Association, Inc. without being limited thereto.
- the necessary amounts of the cosmetic and dermatological adjuvants and additives can - based on the desired product - easily be chosen by a skilled person in this field and will be illustrated in the examples, without being limited hereto. Of course, one skilled in this art will take care to select the above mentioned optional additional compound or compounds and/or their amounts such that the advantageous properties intrinsically associated with the combination in accordance with the invention are not, or not substantially, detrimentally affected by the envisaged addition or additions.
- the topical compositions according to the invention in general have a pH in the range of 3 to 10, preferably a pH in the range of 4 to 8 and most preferably a pH in the range of 4 to 7.
- suitable acids such as e.g. citric acid or bases such as NaOH according to standard methods in the art.
- the following examples are provided to further illustrate the compositions and effects of the present invention. These examples are illustrative only and are not intended to limit the scope of the invention in any way.
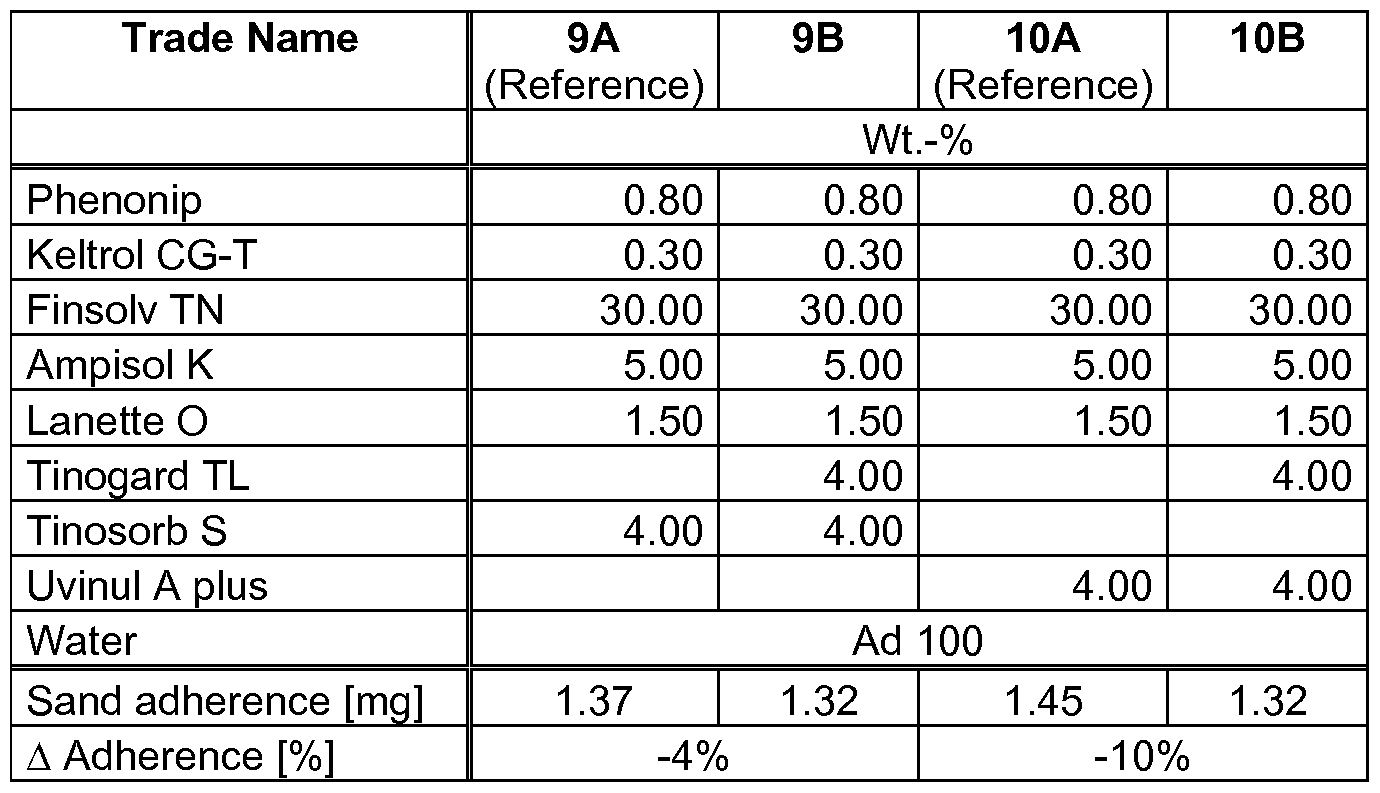
- compositions as outlined in table 1 and 2 were prepared according to standard methods in the art.
- Adherence [%] ⁇ (Adherence Sample [mg]) - (Adherence Reference [mg]) ⁇ * 100%.
- Keltrol CG-T Xanthan Gum 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30 0.30
- Tinosorb* 5 S Bis-Ethylhexyl- 4.00 4.00 4.00 4.00 4.00
- TinogarcT TL Benzotriazolyl 4.00 4.00 4.00 4.00 dodecyl
- Pemulen TR2 Acrylates/C 10-30 AlkyI Acrylate 0.40 0.40 0.40 0.40
- Tinogarcf TL Benzotriazolyl dodecyl 4.00 4.00 4.00 p-Cresol
- compositions according to the present invention exhibit a reduced sand-adherence.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Dermatology (AREA)
- Emergency Medicine (AREA)
- Cosmetics (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112013031220A BR112013031220A2 (en) | 2011-06-08 | 2012-06-06 | topical composition |
| EP12727818.2A EP2717835A2 (en) | 2011-06-08 | 2012-06-06 | Topical composition |
| US14/124,762 US20140227205A1 (en) | 2011-06-08 | 2012-06-06 | Topical composition |
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP11004658 | 2011-06-08 | ||
| EP11004658.8 | 2011-06-08 | ||
| EP11169927 | 2011-06-15 | ||
| EP11169934.3 | 2011-06-15 | ||
| EP11169927.8 | 2011-06-15 | ||
| EP11169934 | 2011-06-15 | ||
| EP11173617.9 | 2011-07-12 | ||
| EP11173617 | 2011-07-12 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012168289A2 true WO2012168289A2 (en) | 2012-12-13 |
| WO2012168289A3 WO2012168289A3 (en) | 2014-05-15 |
Family
ID=46317352
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2012/060676 WO2012168289A2 (en) | 2011-06-08 | 2012-06-06 | Topical composition |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20140227205A1 (en) |
| EP (1) | EP2717835A2 (en) |
| BR (1) | BR112013031220A2 (en) |
| WO (1) | WO2012168289A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3093007A1 (en) * | 2015-05-13 | 2016-11-16 | Beiersdorf AG | Octocrylene-free sunscreen comprising diethylamino hydroxybenzoyl hexyl benzoate |
| EP3093009B1 (en) | 2015-05-13 | 2019-04-24 | Beiersdorf AG | Octocrylene-free sunscreen agent with diethylaminohydroxybenzoylhexylbenzoate |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11918672B2 (en) | 2017-09-01 | 2024-03-05 | Basf Se | Water based concentrated product forms of oil-soluble organic UV absorbers |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0917850A1 (en) | 1997-11-20 | 1999-05-26 | Vorwerk & Co. Interholding GmbH | Air leading pipe, in particular suction pipe |
| EP0917849A1 (en) | 1997-11-21 | 1999-05-26 | Innoversions International, Inc. | Toothbrush storage device |
| EP1015083A1 (en) | 1997-09-18 | 2000-07-05 | Lego A/S | A toy building set comprising a tubular, elongated, flexible toy building element, and such toy building element |
| EP1046391A2 (en) | 1999-04-20 | 2000-10-25 | Basf Aktiengesellschaft | Use of aminosubstituted hydroxybenzophenones as photostable UV-filters in cosmetic and pharmaceutical preparations |
| EP1133980A2 (en) | 2000-03-15 | 2001-09-19 | Basf Aktiengesellschaft | Use of combinations of photoprotectors comprising as essential components aminosubstituted hydroxybenzophenones as photostable UV-filters in cosmetic and pharmaceutical preparations |
| WO2007071584A2 (en) | 2005-12-21 | 2007-06-28 | Ciba Holding Inc. | Use of transmission dyes for protecting human skin from browning and ageing |
| WO2011086124A1 (en) | 2010-01-15 | 2011-07-21 | Dsm Ip Assets B.V. | 2 - phenyl - 1, 2, 3 - benzotriazoles for uv radiation absorbance |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE10140536A1 (en) * | 2001-08-17 | 2003-02-27 | Beiersdorf Ag | Cosmetic and dermatological light protection formulations containing benzotriazoles and iminodisuccinic acid and / or their salts |
| CN104800098A (en) * | 2007-07-09 | 2015-07-29 | 巴斯夫欧洲公司 | Water based concentrated product forms of oil-soluble organic uv absorbers |
| KR20100063084A (en) * | 2007-08-24 | 2010-06-10 | 바스프 에스이 | Mixtures comprising benzotriazoles and merocyanines |
| EP2078521A1 (en) * | 2008-01-08 | 2009-07-15 | Stada Arzneimittel Ag | Cosmetic composition containing a derivative of benzotriazol and an AHR antagonist |
| FR2947174B1 (en) * | 2009-06-24 | 2011-07-15 | Oreal | COSMETIC COMPOSITION COMPRISING A SUPERABSORBENT POLYMER AND AN ORGANIC UV FILTER |
-
2012
- 2012-06-06 BR BR112013031220A patent/BR112013031220A2/en not_active IP Right Cessation
- 2012-06-06 EP EP12727818.2A patent/EP2717835A2/en not_active Withdrawn
- 2012-06-06 WO PCT/EP2012/060676 patent/WO2012168289A2/en active Application Filing
- 2012-06-06 US US14/124,762 patent/US20140227205A1/en not_active Abandoned
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1015083A1 (en) | 1997-09-18 | 2000-07-05 | Lego A/S | A toy building set comprising a tubular, elongated, flexible toy building element, and such toy building element |
| EP0917850A1 (en) | 1997-11-20 | 1999-05-26 | Vorwerk & Co. Interholding GmbH | Air leading pipe, in particular suction pipe |
| EP0917849A1 (en) | 1997-11-21 | 1999-05-26 | Innoversions International, Inc. | Toothbrush storage device |
| EP1046391A2 (en) | 1999-04-20 | 2000-10-25 | Basf Aktiengesellschaft | Use of aminosubstituted hydroxybenzophenones as photostable UV-filters in cosmetic and pharmaceutical preparations |
| EP1133980A2 (en) | 2000-03-15 | 2001-09-19 | Basf Aktiengesellschaft | Use of combinations of photoprotectors comprising as essential components aminosubstituted hydroxybenzophenones as photostable UV-filters in cosmetic and pharmaceutical preparations |
| DE10012408A1 (en) | 2000-03-15 | 2001-09-20 | Basf Ag | Use of sunscreen combinations which contain as essential constituent amino-substituted hydroxybenzophenones as photostable UV filters in cosmetic and pharmaceutical preparations |
| WO2007071584A2 (en) | 2005-12-21 | 2007-06-28 | Ciba Holding Inc. | Use of transmission dyes for protecting human skin from browning and ageing |
| WO2011086124A1 (en) | 2010-01-15 | 2011-07-21 | Dsm Ip Assets B.V. | 2 - phenyl - 1, 2, 3 - benzotriazoles for uv radiation absorbance |
Non-Patent Citations (1)
| Title |
|---|
| "CTFA Cosmetic Ingredient Handbook", 1992, THE COSMETIC, TOILETRY AND FRAGRANCE ASSOCIATION, INC. |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3093007A1 (en) * | 2015-05-13 | 2016-11-16 | Beiersdorf AG | Octocrylene-free sunscreen comprising diethylamino hydroxybenzoyl hexyl benzoate |
| EP3093009B1 (en) | 2015-05-13 | 2019-04-24 | Beiersdorf AG | Octocrylene-free sunscreen agent with diethylaminohydroxybenzoylhexylbenzoate |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2012168289A3 (en) | 2014-05-15 |
| EP2717835A2 (en) | 2014-04-16 |
| BR112013031220A2 (en) | 2016-09-06 |
| US20140227205A1 (en) | 2014-08-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2717834A2 (en) | Topical composition | |
| US20140328777A1 (en) | Sunscreens | |
| EP2717833A2 (en) | Cosmetic compositions | |
| WO2012168289A2 (en) | Topical composition | |
| EP2720672A2 (en) | Sunscreens comprising uv absorbers of benzotriazol structure | |
| EP2717828A2 (en) | Cosmetic compositions | |
| US20210161793A1 (en) | Topical composition | |
| EP3793694B1 (en) | Topical composition | |
| US11547646B2 (en) | Topical composition | |
| US11534374B2 (en) | Topical composition | |
| US20240173234A1 (en) | Sunscreen composition | |
| WO2012168276A2 (en) | Sunscreen | |
| EP4340804A1 (en) | Sunscreen composition comprising bemotrizinol | |
| WO2024033221A1 (en) | Sunscreen composition | |
| KR20200101383A (en) | Topical composition | |
| WO2012168280A2 (en) | Cosmetic compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12727818 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012727818 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14124762 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013031220 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013031220 Country of ref document: BR Kind code of ref document: A2 Effective date: 20131204 |