WO2012161116A1 - 新規アミロイド親和性化合物 - Google Patents
新規アミロイド親和性化合物 Download PDFInfo
- Publication number
- WO2012161116A1 WO2012161116A1 PCT/JP2012/062778 JP2012062778W WO2012161116A1 WO 2012161116 A1 WO2012161116 A1 WO 2012161116A1 JP 2012062778 W JP2012062778 W JP 2012062778W WO 2012161116 A1 WO2012161116 A1 WO 2012161116A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- amyloid
- brain
- phenyl
- pyridine
- Prior art date
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 59
- 230000002285 radioactive effect Effects 0.000 claims abstract description 17
- 239000000032 diagnostic agent Substances 0.000 claims abstract description 13
- 229940039227 diagnostic agent Drugs 0.000 claims abstract description 13
- 208000024827 Alzheimer disease Diseases 0.000 claims abstract description 11
- 150000003839 salts Chemical class 0.000 claims abstract description 9
- 125000005843 halogen group Chemical group 0.000 claims abstract 4
- -1 triphenylstannyl groups Chemical group 0.000 claims description 8
- 125000001424 substituent group Chemical group 0.000 claims description 7
- 125000000217 alkyl group Chemical group 0.000 claims description 4
- 125000004432 carbon atom Chemical group C* 0.000 claims description 4
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 3
- 125000005208 trialkylammonium group Chemical group 0.000 claims description 2
- 210000004556 brain Anatomy 0.000 abstract description 32
- 239000000523 sample Substances 0.000 abstract description 4
- 238000002059 diagnostic imaging Methods 0.000 abstract description 2
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 30
- 239000000243 solution Substances 0.000 description 29
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 27
- 238000006243 chemical reaction Methods 0.000 description 22
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 19
- 238000000034 method Methods 0.000 description 18
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 16
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- 229940125904 compound 1 Drugs 0.000 description 15
- 230000015572 biosynthetic process Effects 0.000 description 14
- UTCSSFWDNNEEBH-UHFFFAOYSA-N imidazo[1,2-a]pyridine Chemical compound C1=CC=CC2=NC=CN21 UTCSSFWDNNEEBH-UHFFFAOYSA-N 0.000 description 13
- 238000003786 synthesis reaction Methods 0.000 description 13
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 12
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 12
- 229940125782 compound 2 Drugs 0.000 description 11
- 239000002904 solvent Substances 0.000 description 11
- VNHBYKHXBCYPBJ-UHFFFAOYSA-N 5-ethynylimidazo[1,2-a]pyridine Chemical compound C#CC1=CC=CC2=NC=CN12 VNHBYKHXBCYPBJ-UHFFFAOYSA-N 0.000 description 10
- 150000002367 halogens Chemical class 0.000 description 10
- 239000000203 mixture Substances 0.000 description 10
- 239000002243 precursor Substances 0.000 description 10
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 9
- 239000002953 phosphate buffered saline Substances 0.000 description 9
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 8
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 8
- 238000003745 diagnosis Methods 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- 229910052736 halogen Inorganic materials 0.000 description 7
- 125000001462 1-pyrrolyl group Chemical group [*]N1C([H])=C([H])C([H])=C1[H] 0.000 description 6
- 238000005160 1H NMR spectroscopy Methods 0.000 description 6
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- 238000005481 NMR spectroscopy Methods 0.000 description 6
- 239000011630 iodine Substances 0.000 description 6
- 229910052740 iodine Inorganic materials 0.000 description 6
- 239000002244 precipitate Substances 0.000 description 6
- FVAUCKIRQBBSSJ-UHFFFAOYSA-M sodium iodide Chemical compound [Na+].[I-] FVAUCKIRQBBSSJ-UHFFFAOYSA-M 0.000 description 6
- 230000003941 amyloidogenesis Effects 0.000 description 5
- 206010002022 amyloidosis Diseases 0.000 description 5
- 238000004458 analytical method Methods 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 238000004128 high performance liquid chromatography Methods 0.000 description 5
- 239000011259 mixed solution Substances 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- FVAUCKIRQBBSSJ-VVUPZWBASA-M sodium;iodine-123(1-) Chemical compound [Na+].[123I-] FVAUCKIRQBBSSJ-VVUPZWBASA-M 0.000 description 5
- 230000002194 synthesizing effect Effects 0.000 description 5
- POQYEHYKVSZQIG-UHFFFAOYSA-N 2-(4-azidophenyl)-6-iodoimidazo[1,2-a]pyridine Chemical compound C=1N2C=C(I)C=CC2=NC=1C1=CC=C(N=[N+]=[N-])C=C1 POQYEHYKVSZQIG-UHFFFAOYSA-N 0.000 description 4
- IVILGUFRMDBUEQ-UHFFFAOYSA-N 5-iodopyridin-2-amine Chemical compound NC1=CC=C(I)C=N1 IVILGUFRMDBUEQ-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- 238000009825 accumulation Methods 0.000 description 4
- 230000027455 binding Effects 0.000 description 4
- 230000008499 blood brain barrier function Effects 0.000 description 4
- 239000013078 crystal Substances 0.000 description 4
- 238000003384 imaging method Methods 0.000 description 4
- 238000012744 immunostaining Methods 0.000 description 4
- 238000002372 labelling Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 4
- LZJPDRANSVSGOR-UHFFFAOYSA-N 1-(4-azidophenyl)-2-bromoethanone Chemical compound BrCC(=O)C1=CC=C(N=[N+]=[N-])C=C1 LZJPDRANSVSGOR-UHFFFAOYSA-N 0.000 description 3
- 150000000215 1-octanols Chemical class 0.000 description 3
- 208000037259 Amyloid Plaque Diseases 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 3
- 238000000211 autoradiogram Methods 0.000 description 3
- 238000000376 autoradiography Methods 0.000 description 3
- 210000005013 brain tissue Anatomy 0.000 description 3
- 230000003920 cognitive function Effects 0.000 description 3
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 3
- 239000003480 eluent Substances 0.000 description 3
- 239000000945 filler Substances 0.000 description 3
- 210000004884 grey matter Anatomy 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 3
- 210000000056 organ Anatomy 0.000 description 3
- 239000007800 oxidant agent Substances 0.000 description 3
- 230000035699 permeability Effects 0.000 description 3
- 239000002504 physiological saline solution Substances 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 238000010898 silica gel chromatography Methods 0.000 description 3
- RAIRZFJPGKTTJS-UHFFFAOYSA-N tributyl-[2-(4-pyrrol-1-ylphenyl)imidazo[1,2-a]pyridin-6-yl]stannane Chemical compound C=1N2C=C([Sn](CCCC)(CCCC)CCCC)C=CC2=NC=1C(C=C1)=CC=C1N1C=CC=C1 RAIRZFJPGKTTJS-UHFFFAOYSA-N 0.000 description 3
- 239000003643 water by type Substances 0.000 description 3
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- IAVCEBMLYVGBLA-UHFFFAOYSA-N 2-[1-[6-[2-fluoroethyl(methyl)amino]naphthalen-2-yl]ethylidene]propanedinitrile Chemical compound C1=C(C(C)=C(C#N)C#N)C=CC2=CC(N(CCF)C)=CC=C21 IAVCEBMLYVGBLA-UHFFFAOYSA-N 0.000 description 2
- ZQAQXZBSGZUUNL-UHFFFAOYSA-N 2-[4-(methylamino)phenyl]-1,3-benzothiazol-6-ol Chemical compound C1=CC(NC)=CC=C1C1=NC2=CC=C(O)C=C2S1 ZQAQXZBSGZUUNL-UHFFFAOYSA-N 0.000 description 2
- ZWIARAQZLULYPG-NSCUHMNNSA-N 4-[(e)-2-[4-(methylamino)phenyl]ethenyl]phenol Chemical compound C1=CC(NC)=CC=C1\C=C\C1=CC=C(O)C=C1 ZWIARAQZLULYPG-NSCUHMNNSA-N 0.000 description 2
- 102000013455 Amyloid beta-Peptides Human genes 0.000 description 2
- 108010090849 Amyloid beta-Peptides Proteins 0.000 description 2
- 206010002091 Anaesthesia Diseases 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 2
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 description 2
- 241000700157 Rattus norvegicus Species 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- VLSOAXRVHARBEQ-UHFFFAOYSA-N [4-fluoro-2-(hydroxymethyl)phenyl]methanol Chemical compound OCC1=CC=C(F)C=C1CO VLSOAXRVHARBEQ-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000037005 anaesthesia Effects 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 2
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 102000034240 fibrous proteins Human genes 0.000 description 2
- 108091005899 fibrous proteins Proteins 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 125000004857 imidazopyridinyl group Chemical group N1C(=NC2=C1C=CC=N2)* 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- XMBWDFGMSWQBCA-RNFDNDRNSA-M iodine-131(1-) Chemical compound [131I-] XMBWDFGMSWQBCA-RNFDNDRNSA-M 0.000 description 2
- 230000004770 neurodegeneration Effects 0.000 description 2
- 210000002682 neurofibrillary tangle Anatomy 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 238000012856 packing Methods 0.000 description 2
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 2
- 230000001575 pathological effect Effects 0.000 description 2
- 238000012636 positron electron tomography Methods 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 239000012488 sample solution Substances 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 238000002603 single-photon emission computed tomography Methods 0.000 description 2
- 235000009518 sodium iodide Nutrition 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- CWMFRHBXRUITQE-UHFFFAOYSA-N trimethylsilylacetylene Chemical group C[Si](C)(C)C#C CWMFRHBXRUITQE-UHFFFAOYSA-N 0.000 description 2
- 0 *c(cc1)c[n]2c1nc(-c(cc1)ccc1-[n]1cccc1)c2 Chemical compound *c(cc1)c[n]2c1nc(-c(cc1)ccc1-[n]1cccc1)c2 0.000 description 1
- 125000001607 1,2,3-triazol-1-yl group Chemical group [*]N1N=NC([H])=C1[H] 0.000 description 1
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical class C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- HMUNWXXNJPVALC-UHFFFAOYSA-N 1-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C(CN1CC2=C(CC1)NN=N2)=O HMUNWXXNJPVALC-UHFFFAOYSA-N 0.000 description 1
- KAQIGYQIHKNJHL-UHFFFAOYSA-N 2-bromo-1-(4-pyrrol-1-ylphenyl)ethanone Chemical compound C1=CC(C(=O)CBr)=CC=C1N1C=CC=C1 KAQIGYQIHKNJHL-UHFFFAOYSA-N 0.000 description 1
- KJDSORYAHBAGPP-UHFFFAOYSA-N 4-(3,4-diaminophenyl)benzene-1,2-diamine;hydron;tetrachloride Chemical compound Cl.Cl.Cl.Cl.C1=C(N)C(N)=CC=C1C1=CC=C(N)C(N)=C1 KJDSORYAHBAGPP-UHFFFAOYSA-N 0.000 description 1
- FGQRNGPGBIJMET-UHFFFAOYSA-N 4-(6-iodo-1,3-benzothiazol-2-yl)-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=NC2=CC=C(I)C=C2S1 FGQRNGPGBIJMET-UHFFFAOYSA-N 0.000 description 1
- ZDGRIJNRIGFCGI-UHFFFAOYSA-N 4-(6-iodo-1,3-benzoxazol-2-yl)-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=NC2=CC=C(I)C=C2O1 ZDGRIJNRIGFCGI-UHFFFAOYSA-N 0.000 description 1
- RCVUDYJMEJWTNN-ONEGZZNKSA-N 4-[(e)-2-(4-iodophenyl)ethenyl]-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1\C=C\C1=CC=C(I)C=C1 RCVUDYJMEJWTNN-ONEGZZNKSA-N 0.000 description 1
- GSZMUPHKOPBPPS-UHFFFAOYSA-N 5-[2-[6-(2-fluoroethoxy)-1,3-benzoxazol-2-yl]ethenyl]-n,n-dimethyl-1,3-thiazol-2-amine Chemical compound S1C(N(C)C)=NC=C1C=CC1=NC2=CC=C(OCCF)C=C2O1 GSZMUPHKOPBPPS-UHFFFAOYSA-N 0.000 description 1
- UGSBCCAHDVCHGI-UHFFFAOYSA-N 5-nitropyridin-2-amine Chemical compound NC1=CC=C([N+]([O-])=O)C=N1 UGSBCCAHDVCHGI-UHFFFAOYSA-N 0.000 description 1
- YTOLNVVJAQBNRF-UHFFFAOYSA-N 6-iodo-2-(4-pyrrol-1-ylphenyl)imidazo[1,2-a]pyridine Chemical compound C=1N2C=C(I)C=CC2=NC=1C(C=C1)=CC=C1N1C=CC=C1 YTOLNVVJAQBNRF-UHFFFAOYSA-N 0.000 description 1
- KCMOCNVLWSZRNX-UHFFFAOYSA-N 6-iodo-2-[4-(triazol-1-yl)phenyl]imidazo[1,2-a]pyridine Chemical compound C=1N2C=C(I)C=CC2=NC=1C(C=C1)=CC=C1N1C=CN=N1 KCMOCNVLWSZRNX-UHFFFAOYSA-N 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 206010012289 Dementia Diseases 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 235000010724 Wisteria floribunda Nutrition 0.000 description 1
- YMXHOFJMHQMLNB-UHFFFAOYSA-N [1-[4-(6-iodoimidazo[1,2-a]pyridin-2-yl)phenyl]triazol-4-yl]-trimethylsilane Chemical compound N1=NC([Si](C)(C)C)=CN1C1=CC=C(C=2N=C3C=CC(I)=CN3C=2)C=C1 YMXHOFJMHQMLNB-UHFFFAOYSA-N 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 238000001574 biopsy Methods 0.000 description 1
- 210000001218 blood-brain barrier Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- IYYIVELXUANFED-UHFFFAOYSA-N bromo(trimethyl)silane Chemical compound C[Si](C)(C)Br IYYIVELXUANFED-UHFFFAOYSA-N 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- KPMVHELZNRNSMN-UHFFFAOYSA-N chembl1985849 Chemical compound N1=CC=C2NCCN21 KPMVHELZNRNSMN-UHFFFAOYSA-N 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- JZCCFEFSEZPSOG-UHFFFAOYSA-L copper(II) sulfate pentahydrate Chemical compound O.O.O.O.O.[Cu+2].[O-]S([O-])(=O)=O JZCCFEFSEZPSOG-UHFFFAOYSA-L 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000002224 dissection Methods 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 238000011067 equilibration Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- KRHYYFGTRYWZRS-BJUDXGSMSA-M fluorine-18(1-) Chemical compound [18F-] KRHYYFGTRYWZRS-BJUDXGSMSA-M 0.000 description 1
- 210000001652 frontal lobe Anatomy 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000003365 glass fiber Substances 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 238000010191 image analysis Methods 0.000 description 1
- 125000002962 imidazol-1-yl group Chemical group [*]N1C([H])=NC([H])=C1[H] 0.000 description 1
- 150000005232 imidazopyridines Chemical class 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- WXTYQJQQGUYCGM-UHFFFAOYSA-N n,n-dimethyl-4-(6-tributylstannylimidazo[1,2-a]pyridin-2-yl)aniline Chemical compound C=1N2C=C([Sn](CCCC)(CCCC)CCCC)C=CC2=NC=1C1=CC=C(N(C)C)C=C1 WXTYQJQQGUYCGM-UHFFFAOYSA-N 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 238000010534 nucleophilic substitution reaction Methods 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 239000003444 phase transfer catalyst Substances 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 238000002601 radiography Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- JADVWWSKYZXRGX-UHFFFAOYSA-M thioflavine T Chemical class [Cl-].C1=CC(N(C)C)=CC=C1C1=[N+](C)C2=CC=C(C)C=C2S1 JADVWWSKYZXRGX-UHFFFAOYSA-M 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/041—Heterocyclic compounds
- A61K51/044—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine, rifamycins
- A61K51/0455—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine, rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/041—Heterocyclic compounds
- A61K51/044—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine, rifamycins
- A61K51/0453—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine, rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/22—Tin compounds
- C07F7/2208—Compounds having tin linked only to carbon, hydrogen and/or halogen
Definitions
- the present invention relates to a compound used for diagnosis of head degenerative diseases. More specifically, the present invention relates to a compound useful for detecting amyloid at a lesion site in the diagnosis of a disease in which amyloid accumulates such as Alzheimer's disease.
- amyloidosis Diseases that develop when fibrous proteins called amyloid are deposited in various organs or tissues in the body are collectively called amyloidosis.
- a common feature of amyloidosis is that a fibrous protein called amyloid rich in ⁇ -sheet structure is deposited in various organs or regions throughout the body, causing functional abnormalities in the organs and tissues.
- AD Alzheimer's disease
- AD Alzheimer's disease
- AD Alzheimer's disease
- AD is a typical disease of amyloidosis
- AD causing dementia Since this disease is a disease in which amyloid gradually deposits in the brain and causes death, it can be said that it is a disease with high social interest compared to other amyloidosis.
- AD patients In recent years, in advanced countries, the number of AD patients has rapidly increased with the aging of society, which has become a social problem.
- AD is characterized by three brain pathological findings: the appearance of senile plaques, neurofibrillary tangles and extensive neuronal loss.
- Senile plaques are structures containing amyloid as a major component, and are considered to be pathological findings in the brain that appear in the first stage of AD onset, that is, more than 10 years before clinical symptoms appear.
- Diagnosis of AD is performed by performing various cognitive function evaluations (for example, Hasegawa scale, ADAS-JCog, MMSE, etc.) after supplementarily combining image diagnosis such as CT and MRI.
- various cognitive function evaluations for example, Hasegawa scale, ADAS-JCog, MMSE, etc.
- image diagnosis such as CT and MRI.
- Such a method based on the evaluation of cognitive function has a drawback that the diagnostic sensitivity is low in the early stage of onset, and further, the diagnosis result is easily affected by the cognitive function inherent in each individual.
- a biopsy of a diseased part is indispensable for a definitive diagnosis, it is virtually impossible to make a definitive diagnosis of AD during the lifetime of a patient (Non-patent Document 1).
- amyloid constituting senile plaques is an aggregate of amyloid ⁇ protein (hereinafter referred to as A ⁇ ), and the aggregate of A ⁇ may exhibit neurocytotoxicity by taking a ⁇ sheet structure. It has been reported by many studies. Based on these findings, the so-called “amyloid cascade hypothesis” has been proposed in which the deposition of A ⁇ in the brain is a trigger and the formation of neurofibrillary tangles and neuronal loss occur as downstream phenomena (non-patented). Reference 2).
- thioflavine derivatives such as benzothiazole (hereinafter referred to as 6-OH-BTA-1) (Patent Document 1, Non-Patent Document 3), (E) -4-methylamino-4′-hydroxystilbene (hereinafter referred to as SB-13) and (E) -4-dimethylamino-4′-iodostilbene (hereinafter referred to as m-I-SB) (Patent Document 2, Non-Patent Document 4, Non-Patent Document) 5), 6-iodo-2- [4 ′-(N, N-dimethylamino) phenyl] benzoxazole (hereinafter referred to as IBOX), 6- [2- (fluoro) ethoxy] -2- [2- ( -Dimethylaminothiazol-5-yl) ethenyl] benzoxazole and other benzoxazole derivatives (Non-patent document 6, Non-patent document 7), 2- (1-
- Non-patent Document 10 Non-patent document 11, Non-patent document 12, Non-patent document 13).
- the present invention has been made in view of the above circumstances, and an object thereof is to obtain a novel compound effective as an imaging diagnostic probe targeting amyloid and an Alzheimer's disease diagnostic agent containing the compound.
- the inventors use a compound in which a 5-membered nitrogen-containing heterocyclic ring is bonded to the 4′-position carbon of the phenyl group in the imidazopyridine phenyl skeleton via the nitrogen atom of the nitrogen-containing heterocyclic ring.
- a diagnostic agent for amyloidosis having sufficient diagnostic performance was obtained, and the present invention was completed.
- a salt thereof, and a diagnostic agent for Alzheimer's disease comprising the compound represented by formula (1) or a salt thereof.
- R 1 is a radioactive halogen substituent.
- various radioactive halogens can be used, preferably a radioactive halogen selected from the group consisting of 18 F, 76 Br, 123 I, 124 I, 125 I and 131 I can be used, and more preferably. 18 F or 123 I can be used.
- a 1 and A 2 are each independently CH or N. Therefore, according to a preferred embodiment of the present invention, the following formula (3), (4) or (5):
- a salt thereof, and a diagnostic agent for Alzheimer's disease comprising the compound represented by formula (3), (4) or (5) or a salt thereof.
- R 2 represents a non-radioactive halogen substituent, a nitro group, a trialkylammonium group having 1 to 4 carbon atoms in the alkyl chain, and a trialkylstannyl substitution in which the alkyl chain has 1 to 4 carbon atoms.
- a group selected from the group consisting of a group or a triphenylstannyl group, A 3 and A 4 are each independently CH or N.
- the compound represented by formula (2) can be suitably used as a labeling precursor for the compound of formula (1) described above.
- halogen that can be a target in a nucleophilic substitution reaction using radioactive fluorine or halogen that can be a target of an isotope exchange reaction with radioactive iodine can be used, preferably chlorine, iodine or Bromine can be used.
- Various substituents can be used as the trialkylstannyl substituent, and a trimethylstannyl substituent and a tributylstannyl substituent can be preferably used. Therefore, according to a preferred embodiment of the present invention, a compound represented by the following formula (6), (7) or (8) is provided.
- FIG. 1 A synthetic scheme of 2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] -6-tributylstannylimidazo [1,2-a] pyridine is shown in FIG.
- 2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] -6-tributylstannylimidazo [1,2-a] pyridine first, 4-azidophenacyl The bromide is reacted with 2-amino-5-iodopyridine to synthesize 2- (4-azidophenyl) -6-iodoimidazo [1,2-a] pyridine (FIG. 1, step 1).
- the reaction at this time can be performed according to a conventional method, for example, a method described in literature (Zhi-Ping Zhuang et al.,. J. Med. Chem, 2003, 46, p.237-243).
- 6-iodo-2- (4-azidophenyl) imidazo [1,2-a] pyridine synthesized above is converted into a known method (for example, James James T. Fletcher Ket et al., Tetrahedron Lett, 2008, 49, p.7030-7032) and 6-iodo-2- [4- (4-trimethylsilyl-1H-1,2,3-triazol-1-yl) phenyl] by reacting with trimethylsilylacetylene
- the trimethylsilyl group was removed (FIG. 1, step 3), and 6-iodo-2- [4- (1H-1,2,3, -Triazol-1-yl) phenyl] imidazo [1,2-a] pyridine is obtained.
- 6-iodo-2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] imidazo [1,2-a] pyridine is then purified by known methods (eg, literature Zhi-Ping Zhuang et al., J. Med. Chem, 2003, 46, p.237-243)) (reaction with bistributyltin) (Fig. 1, step 4) 2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] -6-tributylstannylimidazo [1,2-a] pyridine is obtained.
- the substituent at the 6-position in the imidazopyridine ring is a trialkylstannyl substituent other than the tributylstannyl substituent
- the bistrialkyltin may be used.
- a reaction similar to the above may be performed using bistrimethyltin in step 4 of FIG.
- the compound in which A 3 is CH and A 4 is N in the above formula (2) can be obtained by using 4- (1H-imidazol-1-yl) phena instead of 4-azidophenacyl bromide in step 1 of FIG.
- 4-azidophenacyl bromide in step 1 of FIG. 1
- sylbromide and omitting step 3 in FIG. 1 it can be synthesized according to the process in FIG.
- the radioiodine labeled compound was synthesized by dissolving the labeled precursor compound synthesized as described above in an inert organic solvent, and adding a [ 123I ] sodium iodide solution obtained by a known method to this.
- the reaction can be performed by adding an acid and an oxidizing agent.
- the inert organic solvent for dissolving the labeled precursor compound various solvents having no reactivity between the labeled precursor and [ 123 I] sodium iodide can be used, and preferably acetonitrile is used. Can do.
- the oxidizing agent is not particularly limited as long as it can oxidize iodine in the reaction solution, and preferably hydrogen peroxide or peracetic acid can be used.
- the addition amount of the oxidizing agent may be an amount sufficient to oxidize iodine in the reaction solution.
- Radiolabeled substances other than iodine can be synthesized by labeling a labeling precursor according to the purpose of synthesis with a radiohalogen according to the purpose.
- a labeling precursor for example, when synthesizing [ 19 F] -6-fluoro-2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] imidazo [1,2-a] pyridine, Precursor 6-nitro-2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] imidazo [1,2-a] pyridine is transformed into a phase transfer catalyst and the presence of potassium carbonate. The reaction may be performed with [ 18 F] fluoride ion below.
- the diagnostic agent according to the present invention is blended with water or physiological saline adjusted to an appropriate pH or Ringer's solution, if necessary, in the same manner as other generally known radioactive diagnostic agents. It can be prepared as a liquid.
- the concentration of the present compound needs to be not more than the concentration at which the stability of the blended present compound is obtained.
- the dose of the compound is not particularly limited as long as it is a concentration sufficient to image the distribution of the administered drug. For example, in the case of 123 I-labeled compound and 18 F-labeled compound, about 50 to 600 MBq per adult with a body weight of 60 kg can be used by intravenous administration or local administration.
- the distribution of the administered drug can be imaged by a known method.
- a known method for example, in the case of 123 I-labeled compound, it can be imaged using a SPECT apparatus, and in the case of 18 F-labeled compound, it can be imaged using a PET apparatus.
- 6-iodo-2- [4- (1H-1,2,3-triazol-1-yl) phenyl] imidazo [1,2-a] pyridine 50 mg, equivalent to 0.129 mmol was dissolved in dioxane (2.0 mL). After adding 0.5 mL of triethylamine, 0.129 mL (corresponding to 0.258 mmol) of bistributyltin and 14.9 mg (catalytic amount) of tetrakistriphenylphosphine palladium were added. After stirring the reaction mixture at 100 ° C.
- a solution obtained by adding 10 mL of water to the fraction was passed through a Sep-Pak C18 column (trade name: Sep-Pak (registered trademark) Light C18 Cartridges, manufactured by Waters, packing amount of filler 130 mg), and [ 123 I ] -6-iodo-2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] imidazo [1,2-a] pyridine was adsorbed and collected on the column.
- Sep-Pak C18 column trade name: Sep-Pak (registered trademark) Light C18 Cartridges, manufactured by Waters, packing amount of filler 130 mg
- the column was washed with 1 mL of water, and then 1 mL of diethyl ether was passed through and [ 123 I] -6-iodo-2- [4- (1H-1,2,3, -triazol-1-yl) phenyl] Imidazo [1,2-a] pyridine was eluted.
- the amount of radioactivity obtained was 134.5 MBq immediately after synthesis.
- the radiochemical purity was 99.5%.
- [ 123 I] -IMPY was synthesized according to the following steps for use in a comparative example in the study of logP octanol measurement and brain accumulation.
- a solution obtained by adding 10 mL of water to the fraction was added to a Sep-Pak C18 column (trade name: Sep-Pak (registered trademark)).
- the solution was passed through Light C18 Cartridges, manufactured by Waters, and the packing amount of the filler was 130 mg), and [ 123 I] -IMPY was adsorbed and collected on the column.
- the amount of radioactivity obtained was 170 MBq immediately after synthesis.
- the radiochemical purity was 98.5%.
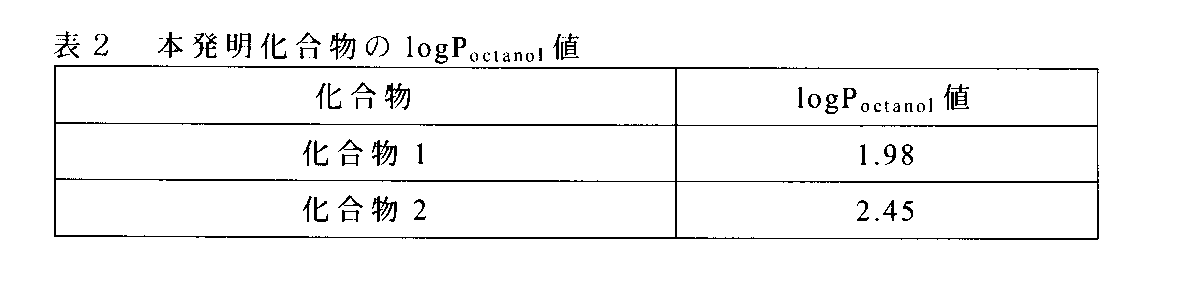
- a distribution coefficient (hereinafter referred to as logP octanol ) using an octanol extraction method generally known as an indicator of the blood-brain barrier (hereinafter referred to as BBB) permeability of the compound was measured.
- Each mixed solution is centrifuged (23 ° C., 3000 g ⁇ 20 minutes) with a centrifuge (model: T2-MC, manufactured by BECKMAN), and 50 ⁇ L of water-saturated 1-octanol and 1-octanol-saturated water is collected.
- the radioactivity was measured with an autowell gamma system (model: ARC-7001, manufactured by Aroka). Using the obtained count, logP octanol was calculated from the following formula (1).
- Example 6 Calculation of dissociation constant (hereinafter referred to as K d ) and maximum number of bonds (hereinafter referred to as B max ) by binding experiments using Alzheimer's disease (hereinafter referred to as AD) patient brain tissue
- AD patient gray matter brain homogenate prepared from AD patient brain tissue (Frontal lobe) commercially available from Analytical Biological Services (USA).
- the filtered filter was washed with 5 mmol / L phosphate buffered saline containing 0.1% BSA (200 ⁇ L x 3 times), and the remaining radioactivity of the filter was determined by the Autowell Gamma System (Type: ARC-7001, Aloka) Manufactured).
- Non-specific binding is described in 6-OH-BTA-1 (literature (CA Mathis et al., J. Med. Cem., (2003), 46, p. 2740) so that the concentration is 1 ⁇ mol / L in the reaction solution. It was set as the count when the same operation was carried out with the addition of synthesis according to the method. The obtained count was analyzed with GraphPad Prism Ver.5 (manufactured by GraphPad Software), and the binding parameters (K d , B max ) were calculated.
- the mass of the brain was measured, and the radioactivity of the brain was further measured using a single channel analyzer (detector model number: SP-20, manufactured by Applied Koken Kogyo Co., Ltd.) (hereinafter referred to as A in this example). . Further, the remaining whole body radioactivity including blood was measured in the same manner (hereinafter referred to as B in this example). Using these measurement results, the amount of radioactivity accumulated in the brain per unit weight (% ID / g) at each dissection time point was calculated from the following mathematical formula (2).
- AD patient brain sections with a thickness of 5 ⁇ m were prepared using AD patient brain tissue commercially available from Analytical Biological Services (USA).
- FIG. 3 After the brain section after washing was sufficiently dried, it was exposed on an imaging plate for 16 hours, and autoradiogram image analysis was performed using a bioimaging analyzer (form: BAS-2500, manufactured by Fuji Photo Film Co., Ltd.).
- FIG. 3, FIG. 4, FIG. (3) Using the adjacent section, immunostaining of the amyloid deposition site with anti-amyloid antibody was performed.
- Anti-Human Amyloid ⁇ (N) (82E1) Mouse IgG MoAb (Immuno-Biological Laboratories) is used as the anti-amyloid antibody, and Anti-Mouse IgG (H + L) Goat IgG Fab'-HRP (stock) as the secondary antibody Immune Biological Laboratories).
- DAB + (3,3′-diaminobenzidine tetrahydrochloride) / substrate kit (Dako)
- the compound according to the present invention can be used in the field of diagnostic agents.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Physics & Mathematics (AREA)
- Medicinal Chemistry (AREA)
- Optics & Photonics (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Urology & Nephrology (AREA)
- Biomedical Technology (AREA)
- Hematology (AREA)
- Immunology (AREA)
- Food Science & Technology (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Analytical Chemistry (AREA)
- Biochemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
このような脳内アミロイド画像診断用プローブの多くは、アミロイドに対する親和性が高く、かつ脳移行性の高い疎水性の低分子化合物を、種々の放射性核種、例えば11C、18F及び123I等で標識した化合物である。具体例として、6-ヨード-2-[4’-(N,N-ジメチルアミノ)フェニル]ベンゾチアゾール(以下、TZDMという)や6-ヒドロキシ-2-[4’-(N-メチルアミノ)フェニル]ベンゾチアゾール(以下、6-OH-BTA-1という)を始めとする種々のチオフラビン誘導体(特許文献1、非特許文献3)、(E)-4-メチルアミノ-4’―ヒドロキシスチルベン(以下、SB-13という)や(E)-4-ジメチルアミノ-4’―ヨードスチルベン(以下、m-I-SBという)を初めとするスチルベン化合物(特許文献2、非特許文献4,非特許文献5)、6-ヨード-2-[4’-(N,N-ジメチルアミノ)フェニル]ベンゾオキサゾール(以下、IBOXという),6-[2-(フルオロ)エトキシ]-2-[2-(2-ジメチルアミノチアゾール-5-イル)エテニル]ベンゾオキサゾールを初めとするベンゾオキサゾール誘導体(非特許文献6,非特許文献7)、2-(1-{6-[(2-フルオロエチル)(メチル)アミノ]-2-ナフチル}エチリデン)マロノニトリル(以下、FDDNPという)を初めとするDDNP誘導体(特許文献4、非特許文献8)及び6-ヨード-2-[4’-(N,N-ジメチルアミノ)フェニル]イミダゾ[1,2-a]ピリジン(以下、IMPYという)を初めとするイミダゾピリジン誘導体(特許文献3、非特許文献9)等を11Cや放射性ハロゲンで標識した化合物、イミダゾピリジン-フェニルに炭素を介して窒素含有5員芳香族複素環式基を結合した化合物を放射性ハロゲン等で標識した化合物(特許文献5、特許文献6)が報告されている。さらに、これらの画像診断用プローブの一部については、ヒトイメージング研究が実施され、AD患者において健常例とは明らかに異なる脳への放射能集積を示すことが報告されている(非特許文献10、非特許文献11、非特許文献12、非特許文献13)。
A1及びA2は、それぞれ独立に、CH又はNである。
従って、本発明の好ましい実施形態によれば、下記式(3)、(4)若しくは(5):
非放射性ハロゲン置換基としては、放射性フッ素を用いた求核置換反応における標的となりうるハロゲン又は放射性ヨウ素との間の同位体交換反応の標的となりうるハロゲンを用いることができ、好ましくは塩素、ヨウ素又は臭素を用いることができる。トリアルキルスタニル置換基としては種々の置換基を用いることができ、トリメチルスタニル置換基及びトリブチルスタニル置換基を好ましく用いることができる。
従って、本発明の好ましい実施形態によれば、下記式(6)、(7)又は(8)で表される化合物が提供される。
以下、2-[4-(1H-1,2,3,-トリアゾール-1-イル)フェニル]-6-トリブチルスタニルイミダゾ[1,2-a]ピリジンを例にとり、本発明の一つの実施形態に係る、放射性ハロゲン標識化合物の前駆体化合物の合成方法を説明する。本化合物は、本発明に係る放射性ヨウ素標識化合物の前駆体化合物として、好適に用いられる化合物である。
次に、放射性ヨード標識体化合物を例にとり、本発明の別の一側面に係る、放射性ハロゲン標識化合物の製造方法について説明する。
酸化剤は、反応液中のヨウ素を酸化させることができるものであれば特に限定する必要はなく、好ましくは過酸化水素又は過酢酸を用いることができる。酸化剤の添加量は、反応溶液中のヨウ素を酸化させるのに十分な量であれば良い。
本発明に係る診断剤は、他の一般に知られている放射性診断剤と同様、本発明に係る放射性ハロゲン標識化合物を所望により適当なpHに調整された水又は生理食塩水、あるいはリンゲル液等に配合させた液として調製することができる。この場合における本化合物の濃度は、配合された本化合物の安定性が得られる濃度以下とする必要がある。本化合物の投与量は、投与された薬剤の分布を画像化するために十分な濃度であれば特に限定する必要はない。例えば、123I標識化合物及び18F標識化合物の場合は、体重60kgの成人一人当り50~600MBq程度、静脈投与又は局所投与して使用することができる。投与された薬剤の分布は、公知の方法にて画像化することができ、例えば123I標識化合物の場合はSPECT装置、18F標識化合物の場合はPET装置を用いて画像化することができる。
1H-NMR(溶媒:重クロロホルム、共鳴周波数:500MHz):δ8.89 ( s, 1H ), 8.31 ( s, 1H), 7.99 ( d, J = 8.7 Hz, 2H), 7.42 ( s, 1H), 7.42 ( s, 1H), 7.19 ( d, J = 8.7 Hz, 2H)。
1H-NMR(溶媒:重クロロホルム、共鳴周波数:500MHz):δ8.92 ( s, 1H ), 8.84 ( d, J = 0.9 Hz, 1H), 8.41 (s, 1H), 8.15 ( d, J = 8.7 Hz, 2H), 7.98 ( d, J = 8.7 Hz, 2H), 7.96( d, J = 0.9 Hz, 1H), 7.45 ( s, 1H)。
1H-NMR(溶媒:重クロロホルム、共鳴周波数:500MHz):δ8.12 ( d, J = 8.7 Hz, 2H), 8.04 ( d, J = 1.2 Hz, 1H), 8.01 ( s, 1H), 7.90 ( s, 1H), 7.87 ( d, J = 1.2 Hz, 1H), 7.82 ( d, J = 8.7 Hz, 2H), 7.61( d, J = 8.7 Hz, 1H), 7.20 ( d, J = 8.7 Hz, 1H), 1.64-1.49 ( m, 6H), 1.36 ( tt, J = 7.3, 7.3 Hz, 6H), 1.20-1.06 ( m, 6H), 0.91( t, J = 7.3 Hz, 9H )。
カラム:YMC PackPro C8(商品名、YMC社製、サイズ:4.6×150mm)
移動相:0.1%トリフルオロ酢酸/アセトニトリル=20/80→0/100(20分)
流速:1.0 mL/分
検出器:紫外可視吸光光度計(検出波長:260nm)及び放射線検出器 (raytest社 STEFFI型)
TLCプレート:TLCプレート:Silica Gel 60 F254(製品名、メルク社製)
展開相:クロロホルム/メタノール/ジエチルアミン=100/1/2
検出器:Rita Star(製品名、raytest社製)
1H-NMR(溶媒:重クロロホルム、共鳴周波数:500MHz):δ8.07 ( d, J = 8.5 Hz, 2H ), 7.70 ( d, J = 8.5 Hz, 2H), 7.19-7.18 ( m, 2H), 6.41-6.40 ( m, 1H), 4.44 ( s, 2H)。
1H-NMR(溶媒:重クロロホルム、共鳴周波数:500MHz):δ8.90 ( s, 1H ), 8.34 ( s, 1H), 8.02 ( d, J = 8.7 Hz, 2H), 7.65 ( d, J = 8.7 Hz, 2H), 7.45-7.41 ( m, 3H), 6.19 ( brs, 1H)。
1H-NMR(溶媒:重クロロホルム、共鳴周波数:500MHz):δ8.02-8.00 ( m, 3H), 7.83 ( s, 1H), 7.60 ( d, J = 8.7 Hz, 1H), 7.46 ( d, J = 8.7 Hz, 2H), 7.18-7.14( m, 3H), 6.37-6.36 ( m, 2H), 1.62-1.50 ( m, 6H), 1.36 ( tt, J = 7.3, 7.3 Hz, 6H), 1.19-1.06 ( m, 6H), 0.91( t, J = 7.3 Hz, 9H )。
カラム:YMC PackPro C8(商品名、YMC社製、サイズ:4.6×150mm)
移動相:0.1%トリフルオロ酢酸/アセトニトリル=20/80→0/100(20分)
流速:1.0 mL/分
検出器:紫外可視吸光光度計(検出波長:260nm)及び放射線検出器 (raytest社 STEFFI型)
TLCプレート:TLCプレート:Silica Gel 60 F254(製品名、メルク社製)
展開相:クロロホルム/メタノール/ジエチルアミン=100/1/2
検出器:Rita Star(製品名、raytest社製)
水飽和1-オクタノール溶液を用いて化合物1及び化合物2を約1MBq/mLに調製し、30μLを平衡容器に添加した。水飽和1-オクタノール及び1-オクタノール飽和水をそれぞれ200μL、400μL又は800μLになるよう平衡容器に添加した。平衡容器を攪拌した後、5分間振とうした(20~25±2℃,回転数20回/分)。それぞれの混合液を遠心機(形式:T2-MC、BECKMAN社製)で遠心分離(23℃、3000g×20分)した後、水飽和1-オクタノール及び1-オクタノール飽和水をそれぞれ50μL分取し、放射能をオートウェル・ガンマシステム(形式:ARC-7001,アロカ社製)にて計測した。得られたカウントを用い、下記数式(1)よりlogPoctanolを算出した。
結果を表2に示す。化合物1及び化合物2のlogPoctanolの値は、それぞれ1.98及び2.45であった。BBB透過性における化合物の最適なlogPoctanol値の範囲は1~3であることが知られている(Douglas D. Dischino et al., J.Nucl.Med., (1983), 24, p.1030-1038)。以上の結果より、化合物1及び化合物2は、BBB透過性を有することが示唆された。
化合物1(約35kBq/100μL)と化合物3(62.5nmol/L)の混合溶液を0.1% ウシ血清アルブミン(以下、BSAとする)含有5mmol/Lリン酸緩衝生理食塩液を用いて希釈していき、反応溶液中で0.2nmol/Lから25nmol/Lになるよう調製した。96穴マイクロプレートの各ウェルに0.1%BSA含有5mmol/Lリン酸緩衝生理食塩液を100μL、調製した化合物1と化合物3の混合溶液を100μL加えた後、0.5μg/μL のAD患者脳灰白質ホモジネートを50μL添加し反応を開始した。反応溶液を3時間振とうした後(22℃、400rpm)、グラスファイバーフィルター(マルチスクリーンHTS FB、ミリポア社製)を用いて反応溶液をろ過した。ろ過後のフィルターを0.1% BSA含有5mmol/Lリン酸緩衝生理食塩液で洗浄した後(200μL×3回)、フィルター残存放射能をオートウェル・ガンマシステム(形式:ARC-7001、アロカ社製)にて測定した。非特異的結合は反応溶液中1μmol/Lになるように6-OH-BTA-1(文献(C. A. Mathis et al., J. Med. Cem., (2003), 46, p.2740)記載の方法に従って合成)を添加して同様に操作したときのカウントとした。得られたカウントをGraphPad Prism Ver.5(GraphPad Software社製)で解析し、結合パラメータ(Kd、Bmax)を算出した。
化合物1はKd:4.94nmol/L、Bmax:2242fmol/mg proteinを示した。この結果より、化合物1がAD患者脳内のアミロイド凝集体に対し高い結合活性を持つことが示された。
化合物1及び化合物2を50mmol/LのL-システイン塩酸塩を含む生理食塩液に溶解した液をそれぞれ調製し、試料溶液とした(放射能濃度共に37MBq/mL)。この試料溶液を、無麻酔下で尾静脈より雄性のWistar系ラット(8週齢)に投与した(投与量:0.2mL、投与した放射能:7.4MBq相当)。投与後2分、5分、15分、30分、60分に無麻酔下で断頭し、血液及び脳を採取した。脳の質量を測定し、さらに脳の放射能をシングルチャネルアナライザー(検出器型番:SP-20、応用光研工業株式会社製)を用いて計測した(以下、本実施例にてAとする)。また、血液を含む残り全身の放射能量を同様に測定した(以下、本実施例にてBとする)。これらの測定結果を用い、下記数式(2)より、各解剖時間点における、脳への単位重量当たりの放射能集積量(%ID/g)を算出した。
なお、本実施例においては、各時間点において、それぞれ3匹の動物を用いて実験を行った。
結果を表3に示す。表3に示すように、化合物1及び化合物2は、投与後2分点において、123I-IMPY同様、高い放射能集積が認められ、その後60分にかけて速やかに消失する傾向を示していた。この結果より、化合物1及び化合物2は、123I-IMPYと同様、高い脳移行性及び速やかな脳からのクリアランスを有することが示唆された。
(1)Analytical Biological Services社(米国)より市販されているAD患者脳組織を用いて、厚さ5μmのAD患者脳切片を作製した。
(2)脳切片をPBSに15分間、5分間、5分間ずつ浸漬し、次に1%BSA含有PBSに30分間浸漬した後、化合物1、化合物2および[123I]-IMPYを含む1%BSA含有PBS(放射能濃度10kBq/mL)をそれぞれ調製し、脳切片を室温下で30分間浸漬した。その後、1%BSA含有PBS、PBS、PBSの各溶液に5分間ずつ浸漬し、脳切片の洗浄を行った。洗浄後の脳切片を十分に乾燥した後、イメージングプレート上で16時間露光させ、バイオイメージングアナライザー(形式:BAS-2500、富士写真フィルム株式会社製)を用いてオートラジオグラム画像解析を行った(図3、図4、図5)。
(3)隣接切片を用いて抗アミロイド抗体によるアミロイド沈着部位の免疫染色を行った。抗アミロイド抗体にAnti-Human Amyloidβ(N)(82E1)Mouse IgG MoAb(株式会社免疫生物研究所)を用い、二次抗体にはAnti-Mouse IgG(H+L)Goat IgG Fab’-HRP(株式会社免疫生物研究所)を用いた。二次抗体に結合するHRPに対してDAB+(3、3'-ジアミノベンジジンテトラヒドロクロライド)・基質キット(Dako)を適用することで、アミロイド沈着部位を検出した(図6)。
化合物1、化合物2及び[123I]-IMPYを使用した溶液に浸漬させた切片におけるオートラジオグラムをそれぞれ図3、図4及び図5に示す。本実験に使用したAD患者脳凍結切片の灰白質部分には、免疫染色によってアミロイドの沈着が確認でき(図6)、いずれのオートラジオグラム上でも、免疫染色によって確認したアミロイド沈着部位への化合物の結合が確認できた。以上の結果より、本発明に係る化合物1及び化合物2は、[123I]-IMPYと同様に脳内におけるアミロイド沈着部位を画像化し得ることが示された。
Claims (5)
- R1が、18F、76Br、123I、124I、125I及び131Iからなる群より選択される、請求項1記載の化合物並びにその塩。
- R1が、18F、76Br、123I、124I、125I及び131Iからなる群より選択される、請求項4記載のアルツハイマー病診断剤。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201280023320.8A CN103534253B (zh) | 2011-05-20 | 2012-05-18 | 对淀粉状蛋白具有亲和性的化合物 |
| EP12789191.9A EP2716641B1 (en) | 2011-05-20 | 2012-05-18 | Radioactive iodine labeled compound having affinity for amyloid |
| AU2012259929A AU2012259929B2 (en) | 2011-05-20 | 2012-05-18 | Novel compound having affinity for amyloid |
| JP2012540218A JP5272110B2 (ja) | 2011-05-20 | 2012-05-18 | 新規アミロイド親和性化合物 |
| ES12789191.9T ES2628221T3 (es) | 2011-05-20 | 2012-05-18 | Compuesto marcado con yodo radioactivo con afinidad por amiloide |
| KR1020137031024A KR20140040722A (ko) | 2011-05-20 | 2012-05-18 | 신규 아밀로이드 친화성 화합물 |
| CA2836872A CA2836872A1 (en) | 2011-05-20 | 2012-05-18 | Novel compound having affinity for amyloid |
| US14/118,317 US9149546B2 (en) | 2011-05-20 | 2012-05-18 | Compound having affinity for amyloid |
| HK14107164.9A HK1193815A1 (zh) | 2011-05-20 | 2014-07-14 | 對澱粉狀蛋白具有親和性的化合物 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011-114198 | 2011-05-20 | ||
| JP2011114198 | 2011-05-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012161116A1 true WO2012161116A1 (ja) | 2012-11-29 |
Family
ID=47217193
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/062778 WO2012161116A1 (ja) | 2011-05-20 | 2012-05-18 | 新規アミロイド親和性化合物 |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US9149546B2 (ja) |
| EP (1) | EP2716641B1 (ja) |
| JP (1) | JP5272110B2 (ja) |
| KR (1) | KR20140040722A (ja) |
| CN (1) | CN103534253B (ja) |
| AU (1) | AU2012259929B2 (ja) |
| CA (1) | CA2836872A1 (ja) |
| ES (1) | ES2628221T3 (ja) |
| HK (1) | HK1193815A1 (ja) |
| TW (1) | TWI528977B (ja) |
| WO (1) | WO2012161116A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014218454A (ja) * | 2013-05-07 | 2014-11-20 | 日本メジフィジックス株式会社 | スチリルピリジン誘導体化合物 |
| JP2014218455A (ja) * | 2013-05-07 | 2014-11-20 | 日本メジフィジックス株式会社 | スチリルピリジン誘導体化合物 |
| JP2017529340A (ja) * | 2014-08-29 | 2017-10-05 | シーエイチディーアイ ファウンデーション,インコーポレーテッド | ハンチントンタンパク質のイメージング用プローブ |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9267232B2 (en) | 2005-03-24 | 2016-02-23 | Tarkett Inc. | Synthetic turf system having an infill trapping structure |
| EP2120737B1 (en) | 2007-02-05 | 2020-04-01 | Boston Scientific Limited | Thrombectomy apparatus |
| US9510854B2 (en) | 2008-10-13 | 2016-12-06 | Boston Scientific Scimed, Inc. | Thrombectomy catheter with control box having pressure/vacuum valve for synchronous aspiration and fluid irrigation |
| WO2012176587A1 (ja) * | 2011-06-24 | 2012-12-27 | 日本メジフィジックス株式会社 | 新規アミロイド親和性化合物 |
| US9883877B2 (en) | 2014-05-19 | 2018-02-06 | Walk Vascular, Llc | Systems and methods for removal of blood and thrombotic material |
| US10561440B2 (en) | 2015-09-03 | 2020-02-18 | Vesatek, Llc | Systems and methods for manipulating medical devices |
| US10492805B2 (en) | 2016-04-06 | 2019-12-03 | Walk Vascular, Llc | Systems and methods for thrombolysis and delivery of an agent |
| JP6831802B2 (ja) * | 2018-01-12 | 2021-02-17 | 上海富吉医療器械有限公司Shanghai Chartwell Medical Instrument Co., Ltd. | 放射性核種標識化合物及びこれを含有するイメージング剤 |
| US11678905B2 (en) | 2018-07-19 | 2023-06-20 | Walk Vascular, Llc | Systems and methods for removal of blood and thrombotic material |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002523383A (ja) | 1998-08-20 | 2002-07-30 | リージェンツ オブ ザ ユニバーシティー オブ カリフォルニア | βアミロイド斑及び神経原線維変化の標識方法 |
| JP2004506723A (ja) | 2000-08-24 | 2004-03-04 | ユニバーシティー オブ ピッツバーグ | アルツハイマー病の生前診断ならびにアミロイド沈着物のインビボ画像化および予防に用いるためのチオフラビン誘導体 |
| JP2005504055A (ja) | 2001-08-27 | 2005-02-10 | ザ トラスティーズ オブ ザ ユニバーシティ オブ ペンシルバニア | スチルベン誘導体、およびアミロイド斑の結合および画像化のためのその使用 |
| JP2005512945A (ja) | 2001-04-23 | 2005-05-12 | ザ・トラスティーズ・オブ・ザ・ユニバーシティ・オブ・ペンシルベニア | アミロイド斑凝集阻害剤および診断用造影剤 |
| WO2007063946A1 (ja) | 2005-11-30 | 2007-06-07 | Fujifilm Ri Pharma Co., Ltd. | アミロイドの凝集及び/又は沈着に起因する疾患の診断薬及び治療薬 |
| WO2007135890A1 (ja) * | 2006-05-19 | 2007-11-29 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物 |
| WO2009054497A1 (ja) * | 2007-10-26 | 2009-04-30 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物 |
| WO2009054496A1 (ja) * | 2007-10-24 | 2009-04-30 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物 |
| WO2009057575A1 (ja) * | 2007-10-30 | 2009-05-07 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物の使用及び製造方法 |
| WO2009057578A1 (ja) * | 2007-10-30 | 2009-05-07 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物の使用及び製造方法 |
| WO2009057576A1 (ja) * | 2007-10-30 | 2009-05-07 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物の使用及び製造方法 |
| WO2010128595A1 (ja) | 2009-05-07 | 2010-11-11 | 富士フイルムRiファーマ株式会社 | 放射性ヨウ素標識イミダゾピリジン誘導体の製造法 |
-
2012
- 2012-05-18 EP EP12789191.9A patent/EP2716641B1/en not_active Not-in-force
- 2012-05-18 WO PCT/JP2012/062778 patent/WO2012161116A1/ja active Application Filing
- 2012-05-18 US US14/118,317 patent/US9149546B2/en not_active Expired - Fee Related
- 2012-05-18 TW TW101117846A patent/TWI528977B/zh not_active IP Right Cessation
- 2012-05-18 CA CA2836872A patent/CA2836872A1/en not_active Abandoned
- 2012-05-18 JP JP2012540218A patent/JP5272110B2/ja not_active Expired - Fee Related
- 2012-05-18 ES ES12789191.9T patent/ES2628221T3/es active Active
- 2012-05-18 CN CN201280023320.8A patent/CN103534253B/zh not_active Expired - Fee Related
- 2012-05-18 AU AU2012259929A patent/AU2012259929B2/en not_active Ceased
- 2012-05-18 KR KR1020137031024A patent/KR20140040722A/ko not_active Application Discontinuation
-
2014
- 2014-07-14 HK HK14107164.9A patent/HK1193815A1/zh not_active IP Right Cessation
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002523383A (ja) | 1998-08-20 | 2002-07-30 | リージェンツ オブ ザ ユニバーシティー オブ カリフォルニア | βアミロイド斑及び神経原線維変化の標識方法 |
| JP2004506723A (ja) | 2000-08-24 | 2004-03-04 | ユニバーシティー オブ ピッツバーグ | アルツハイマー病の生前診断ならびにアミロイド沈着物のインビボ画像化および予防に用いるためのチオフラビン誘導体 |
| JP2005512945A (ja) | 2001-04-23 | 2005-05-12 | ザ・トラスティーズ・オブ・ザ・ユニバーシティ・オブ・ペンシルベニア | アミロイド斑凝集阻害剤および診断用造影剤 |
| JP2005504055A (ja) | 2001-08-27 | 2005-02-10 | ザ トラスティーズ オブ ザ ユニバーシティ オブ ペンシルバニア | スチルベン誘導体、およびアミロイド斑の結合および画像化のためのその使用 |
| WO2007063946A1 (ja) | 2005-11-30 | 2007-06-07 | Fujifilm Ri Pharma Co., Ltd. | アミロイドの凝集及び/又は沈着に起因する疾患の診断薬及び治療薬 |
| WO2007135890A1 (ja) * | 2006-05-19 | 2007-11-29 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物 |
| WO2009054496A1 (ja) * | 2007-10-24 | 2009-04-30 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物 |
| WO2009054497A1 (ja) * | 2007-10-26 | 2009-04-30 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物 |
| WO2009057575A1 (ja) * | 2007-10-30 | 2009-05-07 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物の使用及び製造方法 |
| WO2009057578A1 (ja) * | 2007-10-30 | 2009-05-07 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物の使用及び製造方法 |
| WO2009057576A1 (ja) * | 2007-10-30 | 2009-05-07 | Nihon Medi-Physics Co., Ltd. | 新規アミロイド親和性化合物の使用及び製造方法 |
| WO2010128595A1 (ja) | 2009-05-07 | 2010-11-11 | 富士フイルムRiファーマ株式会社 | 放射性ヨウ素標識イミダゾピリジン誘導体の製造法 |
Non-Patent Citations (18)
| Title |
|---|
| C. A. MATHIS ET AL., J. MED. CHEM., vol. 46, 2003, pages 2740 |
| CHRISTOPHER M. CLARK ET AL.: "Imaging Amyloid with I123 IMPY SPECT", ALZHEIMER'S & DEMENTIA: THE JOURNAL OF THE ALZHEIMER'S ASSOCIATION, vol. 2, no. 1, 2006, pages 342 |
| DOUGLAS D. DISCHINO ET AL., J. NUCL. MED., vol. 24, 1983, pages 1030 - 1038 |
| ERIC D. AGDEPPA ET AL.: "2-Dialkylamino-6-Acylmalononitrile Substituted Naphthalenes (DDNP Analogs): Novel Diagnostic and Therapeutic Tools in Alzheimer's Disease", MOLECULAR IMAGING AND BIOLOGY, vol. 5, 2003, pages 404 - 417 |
| FURUMOTO Y ET AL.: "11C]BF-227: A New 11C-Labeled 2-Ethenylbenzoxazole Derivative for Amyloid-P Plaques Imaging", EUROPEAN JOURNAL OF NUCLEAR MEDICINE AND MOLECULAR IMAGING, vol. 32, no. 1, 2005, pages 759 |
| G. MCKHANN ET AL.: "Clinical diagnosis of Alzheimer's disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease", NEUROLOGY, vol. 34, 1984, pages 939 - 944, XP000671871 |
| H. F. KUNG ET AL.: "Novel Stilbenes as Probes for amyloid plaques", J. AMERICAN CHEMICAL SOCIETY, vol. 123, 2001, pages 12740 - 12741 |
| HIROYUKI ARAI ET AL.: "11C]-BF-227 AND PET to Visualize Amyloid in Alzheimer's Disease Patients", ALZHEIMER'S & DEMENTIA: THE JOURNAL OF THE ALZHEIMER'S ASSOCIATION, vol. 2, no. 1, 2006, pages 312 |
| J. A. HARDY; G. A. HIGGINS: "Alzheimer's Disease: The Amyloid Cascade Hypothesis", SCIENCE, vol. 256, 1992, pages 184 - 185 |
| JAMES T. FLETCHER ET AL., TETRAHEDRON LETT, vol. 49, 2008, pages 7030 - 7032 |
| MASAHIRO ONO ET AL.: "11C-labeled stilbene derivatives as Afi-aggregate-specific PET imaging agents for Alzheimer's disease", NUCLEAR MEDICINE AND BIOLOGY, vol. 30, 2003, pages 565 - 571, XP004444174, DOI: doi:10.1016/S0969-8051(03)00049-0 |
| NICOLAAS P. L. G. VERHOEFF ET AL.: "In-Vivo Imaging of Alzheimer Disease p-Amyloid With [11C] SB-13 PET", AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY, vol. 12, 2004, pages 584 - 595, XP009121906 |
| See also references of EP2716641A4 * |
| W. E. KLUNK ET AL.: "Imaging brain amyloid in Alzheimer's disease with Pittsburgh Compound-B", ANN. NEUROL., vol. 55, 2004, pages 306 - 319, XP008031485, DOI: doi:10.1002/ana.20009 |
| Z.-P. ZHUANG ET AL.: "Radioiodinated Styrylbenzenes and Thioflavins as Probes for Amyloid Aggregates", J. MED. CHEM., vol. 44, 2001, pages 1905 - 1914, XP002410969, DOI: doi:10.1021/jm010045q |
| ZHI-PING ZHUANG ET AL., J. MED. CHEM, vol. 46, 2003, pages 237 - 243 |
| ZHI-PING ZHUANG ET AL.: "IBOX(2- (4'-dimethylaminophenyl)-6-iodobensoxazole): a ligand for imaging amyloid plaques in the brain", NUCLEAR MEDICINE AND BIOLOGY, vol. 28, 2001, pages 887 - 894 |
| ZHI-PING ZHUANG ET AL.: "Structure-Activity Relationship of Imidazo[1,2-a]pyridines as Ligands for Detecting ?-Amyloid Plaques in the Brain", J. MED. CHEM, vol. 46, 2003, pages 237 - 243, XP002446716, DOI: doi:10.1021/jm020351j |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014218454A (ja) * | 2013-05-07 | 2014-11-20 | 日本メジフィジックス株式会社 | スチリルピリジン誘導体化合物 |
| JP2014218455A (ja) * | 2013-05-07 | 2014-11-20 | 日本メジフィジックス株式会社 | スチリルピリジン誘導体化合物 |
| JP2017529340A (ja) * | 2014-08-29 | 2017-10-05 | シーエイチディーアイ ファウンデーション,インコーポレーテッド | ハンチントンタンパク質のイメージング用プローブ |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2012161116A1 (ja) | 2014-07-31 |
| CN103534253B (zh) | 2016-02-24 |
| CN103534253A (zh) | 2014-01-22 |
| US9149546B2 (en) | 2015-10-06 |
| EP2716641A4 (en) | 2014-12-10 |
| CA2836872A1 (en) | 2012-11-29 |
| AU2012259929A1 (en) | 2014-01-16 |
| EP2716641B1 (en) | 2017-04-05 |
| HK1193815A1 (zh) | 2014-10-03 |
| TW201247226A (en) | 2012-12-01 |
| ES2628221T3 (es) | 2017-08-02 |
| JP5272110B2 (ja) | 2013-08-28 |
| AU2012259929B2 (en) | 2016-08-04 |
| EP2716641A1 (en) | 2014-04-09 |
| US20140228569A1 (en) | 2014-08-14 |
| TWI528977B (zh) | 2016-04-11 |
| KR20140040722A (ko) | 2014-04-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5272110B2 (ja) | 新規アミロイド親和性化合物 | |
| JP5313158B2 (ja) | 新規アミロイド親和性化合物 | |
| JPWO2009054496A1 (ja) | 新規アミロイド親和性化合物 | |
| JPWO2009057578A1 (ja) | 新規アミロイド親和性化合物の使用及び製造方法 | |
| JP5180838B2 (ja) | 新規アミロイド親和性化合物 | |
| JP5048655B2 (ja) | 新規アミロイド親和性化合物 | |
| JP5167436B2 (ja) | 新規アミロイド親和性化合物 | |
| US20100267952A1 (en) | Use of novel compound having affinity for amyloid, and process for production of the same | |
| JPWO2008059714A1 (ja) | 新規アミロイド親和性化合物 | |
| US20100216994A1 (en) | Use of novel compound having affinity for amyloid, and process for production of the same | |
| US20100249408A1 (en) | Use of novel compound having affinity for amyloid, and process for production of the same | |
| JP2009120591A (ja) | 新規アミロイド親和性化合物 | |
| JP2009120590A (ja) | 新規アミロイド親和性化合物 | |
| JPWO2007148755A1 (ja) | 新規アミロイド親和性化合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201280023320.8 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 2012540218 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12789191 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2836872 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20137031024 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2012789191 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012789191 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2012259929 Country of ref document: AU Date of ref document: 20120518 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14118317 Country of ref document: US |