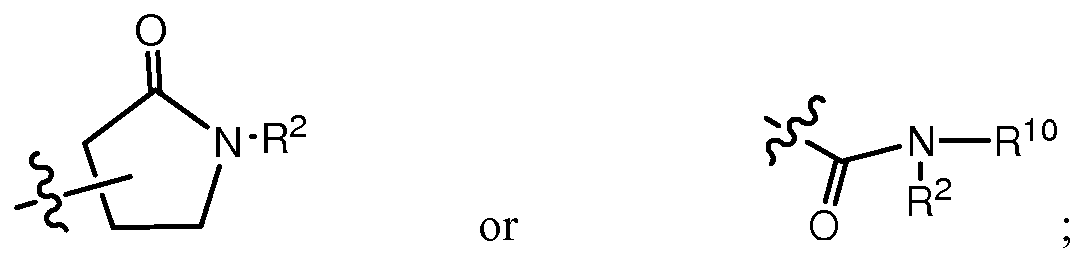WO2012155352A1 - Dihydroisoxazole compounds, parasiticidal uses and formulations thereof - Google Patents
Dihydroisoxazole compounds, parasiticidal uses and formulations thereof Download PDFInfo
- Publication number
- WO2012155352A1 WO2012155352A1 PCT/CN2011/074294 CN2011074294W WO2012155352A1 WO 2012155352 A1 WO2012155352 A1 WO 2012155352A1 CN 2011074294 W CN2011074294 W CN 2011074294W WO 2012155352 A1 WO2012155352 A1 WO 2012155352A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- thiophene
- trifluoromethyl
- dihydroisoxazol
- carboxamide
- tetrahydrobenzo
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C1(C(F)(F)F)ON=C(*)C1 Chemical compound *C1(C(F)(F)F)ON=C(*)C1 0.000 description 2
- JPIGXMNVGKVJMC-UHFFFAOYSA-N CC(NC(CCN1)C1=O)=O Chemical compound CC(NC(CCN1)C1=O)=O JPIGXMNVGKVJMC-UHFFFAOYSA-N 0.000 description 1
- QVAVYHPUBLJLBG-UHFFFAOYSA-N CC(NCC(NCC(F)(F)F)=O)=O Chemical compound CC(NCC(NCC(F)(F)F)=O)=O QVAVYHPUBLJLBG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/42—Oxazoles
- A61K31/422—Oxazoles not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/427—Thiazoles not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/428—Thiazoles condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/4365—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system having sulfur as a ring hetero atom, e.g. ticlopidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/14—Ectoparasiticides, e.g. scabicides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- Ectoparasites such as fleas, lice, flies, mosquitoes, ticks and mites, as well as endoparasites such as gastrointestinal tract nematodes, flukes, and filarids, are problematic for man and animal alike. Such parasites seriously impact productivity in the domesticated animal industry by reducing weight gain, causing poor quality hide, wool, and meat, and in some cases resulting in death. Ecto- and endoparasites are also responsible, in part, for the spread of disease and discomfort in food and companion animals. Ectoparasites in particular are known to harbor and transmit a variety of microbial pathogens, including bacteria, viruses and protozoan parasites, many of which are pathogenic to humans, other warm-blooded mammals and birds.
- ectoparasites in which ectoparasites have been implicated include, but are not limited to, malaria, lymphatic- and blood-born filariasis, trachoma, trypanosomiasis, Leishmaniasis, Rocky Mountain Spotted Fever, Lyme Disease, babesiosis, and food-borne illnesses due to Salmonella, E. coli and Campylobacter, for example.
- ticks parasitize wild as well as domesticated animals and humans, and are known or suspected to be responsible for the transmission of pathogens including bacteria, viruses and protozoan parasites.
- pathogens including bacteria, viruses and protozoan parasites.
- ticks are considered to be second in the world to mosquitoes as vectors of human diseases, but they are considered to be the most important vector of pathogens in North America. Effective elimination of tick infestations is difficult and often impractical, due to the need for concomitant treatment of the immediate host as well as the environmental reservoir.
- tick control is effected by integrated pest management in which different control methods are adapted to one area or against one tick species with due consideration to their environmental effects. While the use of insecticides and pesticides have been beneficial, alternative or improved compounds, formulations, and methods are needed.
- Desirable compounds, formulations, and methods would not only provide alternative therapies, but would also overcome at least some limitations of current approaches. Such limitations include toxicity and safety of both the animal and the user/owner, limited efficacy (potency and duration), and resistance issues. Also impacting the beneficial use of insecticides and pesticides are administration obstacles, which include mode and recurrence of administration. For example, reducing the frequency of administration while maintaining efficacy is desirable, as excessive and repeated treatment of animals is often inconvenient and/or difficult.
- the present invention encompasses parasiticidal compounds, methods, and formulations for use in and on animals and plants, and which provide alternative options for combating parasiticidal infestations, particularly ectoparasiticidal infestations. Further, certain aspects of the invention overcome at least some limitations in the use of current insecticides and pesticides, particularly in providing effective long term, safe control of parasites.
- n 0 or 1 ;
- R 1 is thienyl or phenyl, said thienyl or phenyl substituted with 2 or 3 of the same or different halo atoms;
- R 2 is at each occurrence independently hydrogen, C 1 -C5 alkyl, C3-C6 cycloalkyl, or C 1 -C5 haloalkyl;
- R 3 is -1 ⁇ 2-( CH 2)P- R4 ; p is at each occurrence independently 0 or 1 ;
- R 4 is Ci-C 5 alkyl, Ci-C 5 haloalkyl, Ci-C 5 cyanoalkyl, Ci-C 5 alkylthio, C 3 -C 6 cycloalkyl optionally substituted with hydroxy, halo, or C 1 -C5 alkyl: C3-C5 cyclohetero alkyl optionally substituted with C 1 -C5 alkyl, C 3 -C 6 cycloalkyl, or C 1 -C5 haloalkyl: phenyl, thienyl,
- pyridinyl or O , wherein one of the carbons in said cycloalkyls, independently, or cycloheteroalkyl may form a carbonyl group, and wherein said phenyl, thienyl, or pyridinyl is optionally substituted with halo or a carbamoyl group;
- R 5 is hydroxy, -0-(Ci-C 5 alkyl), or
- R 6 is hydrogen, C 1 -C5 alkyl, C 1 -C5 haloalkyl, C 1 -C5 cyanoalkyl, C 1 -C5 alkylthio, or C 2 -C5 alkynyl; or R 2 and R 3 combine to form, with the nitrogen to which they are attached,
- Yi, Y 2 , and Y 3 are carbon or nitrogen with at most only one of Yi, Y 2 , and Y 3 being nitrogen, and when Yi, Y 2 , or Y 3 is a carbon, each may be substituted by C 1 -C5 alkyl; R 7 is hydrogen, halo, C 1 -C5 alkyl, or
- R is hydroxy, -0-(Ci-C 5 alkyl), or
- R 9 is C1-C5 alkyl
- R 10 is hydrogen, C1-C5 alkyl, C1-C5 haloalkyl, C1-C5 cyanoalkyl, C1-C5 alkylthio, or C2-C5 alkynyl.
- the invention provides a formulation, including a pharmaceutical formulation, comprising a compound or salt of formula I and one or more acceptable carriers.
- the formulation may further comprise at least one additional active ingredient.
- a pharmaceutical formulation of the invention may be a human pharmaceutical formulation or a veterinary pharmaceutical formulation.
- the invention provides a method of controlling ecto- and endoparasite infestations of an animal in need thereof comprising administering an effective amount of a compound or salt of formula I to said animal.
- the method may further provide administering at least one other active ingredient to said animal.
- the animal may be a mammal, and may be a human or a companion animal, for example, a dog or cat.
- the present invention provides a method for preventing and treating diseases transmitted through parasites comprising administering at least one compound of the invention to an animal in need thereof.
- the invention provides a method for controlling parasites, characterized in that a compound of formula I is allowed to act on the pests and/or their habitat.
- the invention provides the use of compounds or salts thereof of formula I for controlling pests.
- the invention provides a compound or salt of formula I for use in therapy.
- the invention further provides a compound or salt of formula I for use in controlling ecto- and endoparasite infestations.
- the invention also provides use of a compound or salt of formula I for the manufacture of a formulation or medicament for controlling ecto- and endoparasite infestations.
- the host animal may be a mammal or non-mammal, such as a bird (turkeys, chickens) or fish. Where the host animal is a mammal, it may be a human or non-human mammal.
- Non-human mammals include domestic animals, such as livestock animals and companion animals. Livestock animals include cattle, camellids, pigs, sheep, goats, and horses. Companion animals include dogs, rabbits, cats, and other pets owned and maintained in close association with humans as part of the human-animal bond.
- Parasites sometimes also referred to as pests, include both ectoparasites and endoparasites.
- Ectoparasites include insect and acarine pests which commonly infest or infect animals, and include the egg, larval, pupal, nymphal, and adult stages thereof. Such pests include fleas, lice, mosquitoes, mites, ticks, beetles, and blood-sucking, biting, or nuisance fly species.
- Endoparasites include nematode pests which commonly infect animals, and include the egg, larval, and adult stages thereof. Such pests include helminths (hookworms, tapeworms, heartworms), and are commercially important because they cause serious diseases in animals, e.g.
- Typical nematodes are Haemonchus, Trichostrcngyius, Qstertagia, Nematotiirus, Cooperia, Ascaris, Bunostonum, Gesophagostonum, Charbertia, Trichuris, Strongyius, Trichonema, Dictyocaulus, Capsliarsa, Heterakis, Toxocara, Ascaridia, Oxyuris, Ancyiostoma, Uncinaria, Toxascaris and Par 'ascaris.
- the trematodes include, in particular, the family of Fasciolideae, especially Fasciola hepatica.
- Controlling refers to either ameliorating or eliminating a current infestation, or preventing an infestation, in or on an animal host or a plant.
- Effective amount refers to the amount of a compound of formula I, or a salt thereof, sufficient to control an ecto- or endoparasite infestation, and includes causing a measurable reduction in the ecto- or endoparasite infestation population, and as such will depend upon several factors.
- ranges for a compound of formula I, or a salt thereof, in the methods include from 0.01 to 1000 mg/kg and more desirably, 0.1 to 100 mg/kg of the animal's body weight.
- the frequency of the administration will also be dependent upon several factors, and can be a single dose administered once a day, once a week, or once a month, for a duration determined by the attending doctor or veterinarian. Additional active ingredients may be administered with a compound of formula I.
- compositions and formulation components such as carriers includes “veterinarily acceptable”, and thus includes both human and animal applications independently.
- Salts of the compounds of the invention including pharmaceutically acceptable salts, and common methodology for preparing them, are known in the art. See, e.g., P. Stahl, et ah, HANDBOOK OF PHARMACEUTICAL SALTS: PROPERTIES, SELECTION AND USE, (VCHA Wiley-VCH, 2002); S.M. Berge, et ah, "Pharmaceutical Salts," Journal of Pharmaceutical Sciences, Vol. 66, No. 1 , January 1977.
- the compounds of the invention and their salts may be formulated as pharmaceutical compositions for administration.
- Such pharmaceutical compositions and processes for making the same are known in the art for both humans and non-human mammals. See, e.g., REMINGTON: THE SCIENCE AND PRACTICE OF PHARMACY, (A. Gennaro, et al, eds., 19 th ed., Mack Publishing Co., 1995).
- Formulations can be administered through various means, including oral administration, parenteral administration such as injection (intramuscular, subcutaneous, intravenous, intraperitoneal) or the like; topical application with or without transdermal penetration such as dipping, spray, bathing, washing, pouring-on and spotting-on, and dusting, or the like.
- Additional active ingredients may be included in the formulation containing a compound of the invention or a salt thereof.
- Carrier is used herein to describe any ingredient other than the active component(s) in a formulation.
- the choice of carrier will to a large extent depend on factors such as the particular mode of administration or application, the effect of the carrier on solubility and stability, and the nature of the dosage form.
- C 1 -C5 alkyl refers to straight chain and branched alkyls having one to five carbon atoms, and includes methyl, ethyl, propyl, n-butyl, iso-butyl, pentyl, isopentyl, and neopentyl.
- C2-C5 alkynyl refers to straight chain and branched alkynyls having two to five carbon atoms, and includes ethynyl, 1-propynyl, 2-propynyl, 1-butynyl, 3-methyl-l-butynyl, pentynyl, isopentynyl, and neopentynyl.
- Halogen or halo refers to fluorine, bromine, chlorine, and iodine.
- Haloalkyl refers to an alkyl (as noted above) substituted with one or more halo atoms. Such groups include trifluoromethyl, methylchloride, dichloromethyl, pentylchloride, butyl chloride, and isopropyl chloride.
- Cyanoalkyl refers to an alkyl (as noted above) substituted with a cyano group.
- Alkylthio refers to an alkyl (as noted above) having a sulfur in the group.
- C3-C6 cycloalkyl refers to cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
- C3-C5 cycloheteroalkyl refers to a saturated ring which has 3 to 5 carbons and a hetero atom.
- the hetero atom may be sulfur, oxygen, nitrogen, or a sulfonyl group.
- Diseases transmitted through parasites are, for example bacterial, viral, rickettsial and protozoal vector-borne diseases.
- viral diseases transmitted through arboviruses i.e. arthropod borne viruses
- Febrile illness Papataci fever, Encephalitis, Meningitis, which are caused by Bunyaviridae such as Bunyavirus, Nairovirus or Phlebovirus
- Febrile illness, rash, encephalitis, polyarthritis, lymphadenitis which are caused by Togaviridae, such as Sindbisvirus, Chikungunya Virus; tick-borne meningoencephalitis, Dengue hemorhagic fever, encephalitis, Febrile illness, Yellow fever
- bacterial diseases transmitted through parasites are Rickettsiosis, such as Rocky Mountain spotted fever, tick typhus caused by infection through Rickettsia ssp; Tularemia caused by infection through Francisella tularensis; Borreliosis or Spirochaetosis, such as Lyme disease, or relapsing fever, caused, by infection through Borrelia ssp.; Ehrllichiosis caused by infection through Ehrlichia ssp.; Plague, caused by infection through Yersinia ssp.
- Rickettsiosis such as Rocky Mountain spotted fever, tick typhus caused by infection through Rickettsia ssp
- Tularemia caused by infection through Francisella tularensis
- Borreliosis or Spirochaetosis such as Lyme disease, or relapsing fever, caused, by infection through Borrelia ssp.
- Ehrllichiosis caused by
- Examples of protozoal or rickettsial borne diseases are Babesiosis, such as Texas fever, red water disease, Q-fever caused by infection through Babesia ssp.; Theileriosis, such as east coast fever, Mediterranean coast fever, caused by infection through Theileria ssp.; Nagana disease, Sleeping sickness caused by infection through Trypanosoma ssp., Anaplasmosis caused by infection through Anaplasma ssp.; Malaria caused by infection through Plasmodium ssp.; Leishmaniasis caused by infection through Leishmania ssp.
- Babesiosis such as Texas fever, red water disease, Q-fever caused by infection through Babesia ssp.
- Theileriosis such as east coast fever, Mediterranean coast fever, caused by infection through Theileria ssp.
- Nagana disease Sleeping sickness caused by infection through Trypanosoma ssp.
- Anaplasmosis caused by infection through Ana
- certain of the compounds of the invention are suitable as soil insecticides against pests in the soil, as well as insecticides for plants, such as cereals, cotton, rice, maize, soya, potatoes, vegetables, fruit, tobacco, hops, citrus, and avocados.
- compounds according to the invention are suitable for protecting plants and plant organs, for increasing the harvest yields, and for improving the quality of the harvested material which are encountered in agriculture, in horticulture, in forests, in gardens, and leisure facilities, and in the protection of stored products and of materials. They may be employed as plant protection agents.
- Plants are to be understood as meaning in the present context all plants and plant populations such as desired and undesired wild plants or crop plants (including naturally occurring crop plants).
- Crop plants can be plants which can be obtained by conventional plant breeding and optimization methods or by biotechnological and genetic engineering methods or by combinations of these methods, including the transgenic plants and including the plant cultivars protectable or not protectable by plant breeders' rights.
- Plant parts are to be understood as meaning all parts and organs of plants above and below the ground, such as shoot, leaf, flower and root, examples which may be mentioned being leaves, needles, stalks, stems, flowers, fruit bodies, fruits, seeds, roots, tubers and rhizomes.
- the plant parts also include harvested material, and vegetative and generative propagation material, for example cuttings, tubers, rhizomes, offshoots and seeds.
- Treatment according to the invention of the plants and plant parts with the active compounds is carried out by conventional and known means, including directly acting on, or by allowing the compounds to act on, the surroundings, habitat or storage space by the customary treatment methods, for example by immersion, spraying, evaporation, fogging, scattering, painting on, injection and, in the case of propagation material, in particular in the case of seeds, also by applying one or more coats.
- the compounds can be converted to the customary formulations, such as solutions, emulsions, wettable powders, water- and oil-based suspensions, powders, dusts, pastes, soluble powders, soluble granules, granules for broadcasting, suspension-emulsion concentrates, natural materials impregnated with active compound, synthetic materials impregnated with active compound, fertilizers and microencapsulations in polymeric substances.
- customary formulations such as solutions, emulsions, wettable powders, water- and oil-based suspensions, powders, dusts, pastes, soluble powders, soluble granules, granules for broadcasting, suspension-emulsion concentrates, natural materials impregnated with active compound, synthetic materials impregnated with active compound, fertilizers and microencapsulations in polymeric substances.
- formulations are produced in a known manner, for example by mixing the active compounds with extenders, that is liquid solvents and/or solid carriers, optionally with the use of surfactants, that is emulsifiers and/or dispersants and/or foam-formers.
- extenders that is liquid solvents and/or solid carriers
- surfactants that is emulsifiers and/or dispersants and/or foam-formers.
- the formulations are prepared either in suitable plants or else before or during the application.
- auxiliaries are substances which are suitable for imparting to the composition itself and/or to preparations derived therefrom (for example spray liquors, seed dressings) particular properties such as certain technical properties and/or also particular biological properties.
- suitable auxiliaries are extenders, solvents, and carriers.
- Example 90 The following compound is prepared essentially by the method of Example 90.
- Example 240 The following compound is prepared essentially by the method of Example 240.
- Compounds may be evaluated against one or more life stages of helminth to measure anthelmintic activity. Compounds may be evaluated at a single concentration followed by serial dilution in order to determine minimal effective concentration. Typically, worms are exposed to compounds in a liquid solution for a predetermined period of time. Activity is measured through one or more variables, which may include an effect on worm motility ⁇ e.g., moving versus non-moving) or viability (e.g., live versus dead).
- Compounds may be evaluated against one or more life stages of helminth infestation in an animal to measure in vivo anthelmintic activity. Compounds may be evaluated a single dose, administered on a milligram per kilogram body weight basis, followed by dose titration in order to determine minimal effective point dose.
- a rodent anthelmintic model for example, adult Mongolian gerbils (Meriones unguiculates) infected with one or more species of Strongylid nematode (e.g., Haemonchus contortus and/or Trichostrongylus colubriformis) are dosed with compounds, administered via oral gavage.
- Strongylid nematode e.g., Haemonchus contortus and/or Trichostrongylus colubriformis
- Gerbils are necropsied and gastrointestinal tract worm burden is measured and compared to untreated, infected control gerbils to determine the degree of anthelmintic activity. Similar testing may be conducted in higher species (e.g., dogs, cats, sheep, cattle) whereby nematode burden in treated animals is compared to burden in untreated, infected animals to measure the potency and duration of anthelmintic activity.
- higher species e.g., dogs, cats, sheep, cattle
- the larval immersion microassay may be conducted as described in White, et ah, J. Med. Entomol. 41 : 1034-1042 (2004). Briefly, experimental test articles are formulated in dimethylsulfoxide (DMSO) to prepare a stock solution at a concentration of
- test articles may be conducted using a modified version of the assay as described in Gutierrez et ah, J. Med. Entomol. 43(3): 526-532(2006).
- the assay may be modified by using a different tick species (the reference describes Ambfyomma americanum ticks) such as Dermacentor variabilis ticks. Further, the reference describes using topical administration, but oral administration may be used.
- experimental test articles are formulated in a solution of polyethylene glycol-300, propylene glycol and water to the desired concentration, typically 1 - 25 mg/ml, depending on solubility and desired point dose.
- Tick containment units (comprised of a baby nipple, ventilated screw cap top and reinforcing rubber washer) are attached to the dorsum of adult Sprague-Dawley rats. After attachment of containment units, approximately 10 unfed nymphal stage American dog ticks (Dermacentor variabilis) are placed inside of each containment unit. Approximately 24 hours after infestation, test article formulations are administered to rats via oral gavage. Negative control rats receive polyethylene glycol-300, propylene glycol and water alone. Depending on compound availability, a minimum of three (3) and a maximum of five (5) rats are utilized per treatment group. Forty-eight (48) hours post-treatment, containment units are removed and live and dead ticks were counted. Live tick counts are transformed using the natural logarithm
- Geometric mean (GM) group tick counts are obtained via back-transformation of group mean transformed counts and subtracting one.
- the non-treated control group is used for comparison to the groups receiving experimental test articles for the calculation of percent efficacy (% reduction in live tick counts).
- the efficacy of treatments is calculated by comparing the geometric mean (GM) number of live ticks observed on treated rats with the GM number of live ticks counted on the negative control rats, using the following formula:
- % Efficacy (GM # live ticks control - GM # live ticks treated) x 100
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Environmental Sciences (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Tropical Medicine & Parasitology (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Provided are dihydroisoxazole compounds useful for controlling parasites both in animals and agriculture. Further provided are methods for controlling parasite infestations of an animal by administering an effective amount of a compound as described above, or a pharmaceutically acceptable salt thereof, to an animal, as well as formulations for controlling parasite infestations using the compounds described above or an acceptable salt thereof, and an acceptable carrier.
Description
DIHYDROISOXAZOLE COMPOUNDS, PARASITICIDAL USES
AND FORMULATIONS THEREOF
Ectoparasites such as fleas, lice, flies, mosquitoes, ticks and mites, as well as endoparasites such as gastrointestinal tract nematodes, flukes, and filarids, are problematic for man and animal alike. Such parasites seriously impact productivity in the domesticated animal industry by reducing weight gain, causing poor quality hide, wool, and meat, and in some cases resulting in death. Ecto- and endoparasites are also responsible, in part, for the spread of disease and discomfort in food and companion animals. Ectoparasites in particular are known to harbor and transmit a variety of microbial pathogens, including bacteria, viruses and protozoan parasites, many of which are pathogenic to humans, other warm-blooded mammals and birds. Diseases in which ectoparasites have been implicated include, but are not limited to, malaria, lymphatic- and blood-born filariasis, trachoma, trypanosomiasis, Leishmaniasis, Rocky Mountain Spotted Fever, Lyme Disease, babesiosis, and food-borne illnesses due to Salmonella, E. coli and Campylobacter, for example.
The medical importance of parasiticide infestations has prompted the development of reagents capable of controlling such. Commonly encountered methods to control parasiticide infestation, for example, have generally focused on use of insecticides, which are often unsuccessful or unsatisfactory for one or more of the following reasons: (1) failure of owner or applicator compliance (frequent administration is required); (2) behavioral or physiological intolerance of the animal to the pesticide product or means of administration; (3) the emergence of ectoparasites resistant to the reagent; and (4) negative impact on the environment and/or toxicity.
Specifically, ticks parasitize wild as well as domesticated animals and humans, and are known or suspected to be responsible for the transmission of pathogens including bacteria, viruses and protozoan parasites. Currently, ticks are considered to be second in the world to mosquitoes as vectors of human diseases, but they are considered to be the most important vector of pathogens in North America. Effective elimination of tick infestations is difficult and often impractical, due to the need for concomitant treatment of the immediate host as well as the environmental reservoir. Presently, tick control is effected by integrated pest management in which different control methods are adapted to one area or against one tick species with due consideration to their environmental effects.
While the use of insecticides and pesticides have been beneficial, alternative or improved compounds, formulations, and methods are needed. Desirable compounds, formulations, and methods would not only provide alternative therapies, but would also overcome at least some limitations of current approaches. Such limitations include toxicity and safety of both the animal and the user/owner, limited efficacy (potency and duration), and resistance issues. Also impacting the beneficial use of insecticides and pesticides are administration obstacles, which include mode and recurrence of administration. For example, reducing the frequency of administration while maintaining efficacy is desirable, as excessive and repeated treatment of animals is often inconvenient and/or difficult.
The present invention encompasses parasiticidal compounds, methods, and formulations for use in and on animals and plants, and which provide alternative options for combating parasiticidal infestations, particularly ectoparasiticidal infestations. Further, certain aspects of the invention overcome at least some limitations in the use of current insecticides and pesticides, particularly in providing effective long term, safe control of parasites.
Provided are compounds, and salts thereof, of formula I:
I wherein A is
n is 0 or 1 ;
R1 is thienyl or phenyl, said thienyl or phenyl substituted with 2 or 3 of the same or different halo atoms;
R2 is at each occurrence independently hydrogen, C1-C5 alkyl, C3-C6 cycloalkyl, or C1-C5 haloalkyl;
R3 is -½-(CH2)P-R4 ; p is at each occurrence independently 0 or 1 ;
R4 is Ci-C5 alkyl, Ci-C5 haloalkyl, Ci-C5 cyanoalkyl, Ci-C5 alkylthio, C3-C6 cycloalkyl optionally substituted with hydroxy, halo, or C1-C5 alkyl: C3-C5 cyclohetero alkyl optionally substituted with C1-C5 alkyl, C3-C6 cycloalkyl, or C1-C5 haloalkyl: phenyl, thienyl,
¾(CH2)p^ R5
pyridinyl, or O , wherein one of the carbons in said cycloalkyls, independently, or cycloheteroalkyl may form a carbonyl group, and wherein said phenyl, thienyl, or pyridinyl is optionally substituted with halo or a carbamoyl group;
R6 is hydrogen, C1-C5 alkyl, C1-C5 haloalkyl, C1-C5 cyanoalkyl, C1-C5 alkylthio, or C2-C5 alkynyl; or R2 and R3 combine to form, with the nitrogen to which they are attached,
Yi, Y2, and Y3 are carbon or nitrogen with at most only one of Yi, Y2, and Y3 being nitrogen, and when Yi, Y2, or Y3 is a carbon, each may be substituted by C1-C5 alkyl; R7 is hydrogen, halo, C1-C5 alkyl, or
R is hydroxy, -0-(Ci-C5 alkyl), or
(CH2) -R9
R2
R9 is C1-C5 alkyl,
R10 is hydrogen, C1-C5 alkyl, C1-C5 haloalkyl, C1-C5 cyanoalkyl, C1-C5 alkylthio, or C2-C5 alkynyl.
The invention provides a formulation, including a pharmaceutical formulation, comprising a compound or salt of formula I and one or more acceptable carriers. The formulation may further comprise at least one additional active ingredient. A pharmaceutical formulation of the invention may be a human pharmaceutical formulation or a veterinary pharmaceutical formulation.
The invention provides a method of controlling ecto- and endoparasite infestations of an animal in need thereof comprising administering an effective amount of a compound or salt of formula I to said animal. The method may further provide administering at least one other active ingredient to said animal. The animal may be a mammal, and may be a human or a companion animal, for example, a dog or cat.
The present invention provides a method for preventing and treating diseases transmitted through parasites comprising administering at least one compound of the invention to an animal in need thereof.
The invention provides a method for controlling parasites, characterized in that a compound of formula I is allowed to act on the pests and/or their habitat. The invention provides the use of compounds or salts thereof of formula I for controlling pests.
The invention provides a compound or salt of formula I for use in therapy. The invention further provides a compound or salt of formula I for use in controlling ecto- and endoparasite infestations. The invention also provides use of a compound or salt of formula I for the manufacture of a formulation or medicament for controlling ecto- and endoparasite infestations.
The host animal may be a mammal or non-mammal, such as a bird (turkeys, chickens) or fish. Where the host animal is a mammal, it may be a human or non-human mammal. Non-human mammals include domestic animals, such as livestock animals and companion animals. Livestock animals include cattle, camellids, pigs, sheep, goats, and horses. Companion animals include dogs, rabbits, cats, and other pets owned and maintained in close association with humans as part of the human-animal bond.
Parasites, sometimes also referred to as pests, include both ectoparasites and endoparasites. Ectoparasites include insect and acarine pests which commonly infest or infect animals, and include the egg, larval, pupal, nymphal, and adult stages thereof. Such pests include fleas, lice, mosquitoes, mites, ticks, beetles, and blood-sucking, biting, or nuisance fly species. Endoparasites include nematode pests which commonly infect animals, and include the egg, larval, and adult stages thereof. Such pests include helminths (hookworms, tapeworms, heartworms), and are commercially important because they cause serious diseases in animals, e.g. in sheep, pigs, goats, cattle, horses, donkeys, camels, dogs, cats, rabbits, guinea-pigs, hamsters, chicken, turkeys, guinea fowls and other farmed birds, as well as exotic birds. Typical nematodes are Haemonchus, Trichostrcngyius, Qstertagia, Nematotiirus, Cooperia, Ascaris, Bunostonum, Gesophagostonum, Charbertia, Trichuris, Strongyius, Trichonema, Dictyocaulus, Capsliarsa, Heterakis, Toxocara, Ascaridia, Oxyuris, Ancyiostoma, Uncinaria, Toxascaris and Par 'ascaris. The trematodes include, in particular, the family of Fasciolideae, especially Fasciola hepatica.
Controlling refers to either ameliorating or eliminating a current infestation, or preventing an infestation, in or on an animal host or a plant.
Effective amount refers to the amount of a compound of formula I, or a salt thereof, sufficient to control an ecto- or endoparasite infestation, and includes causing a measurable reduction in the ecto- or endoparasite infestation population, and as such will depend upon several factors. For use on or in animals, ranges for a compound of formula I, or a salt thereof, in the
methods include from 0.01 to 1000 mg/kg and more desirably, 0.1 to 100 mg/kg of the animal's body weight. The frequency of the administration will also be dependent upon several factors, and can be a single dose administered once a day, once a week, or once a month, for a duration determined by the attending doctor or veterinarian. Additional active ingredients may be administered with a compound of formula I.
Pharmaceutically acceptable as used in this application, for example with reference to salts and formulation components such as carriers, includes "veterinarily acceptable", and thus includes both human and animal applications independently.
Salts of the compounds of the invention, including pharmaceutically acceptable salts, and common methodology for preparing them, are known in the art. See, e.g., P. Stahl, et ah, HANDBOOK OF PHARMACEUTICAL SALTS: PROPERTIES, SELECTION AND USE, (VCHA Wiley-VCH, 2002); S.M. Berge, et ah, "Pharmaceutical Salts," Journal of Pharmaceutical Sciences, Vol. 66, No. 1 , January 1977.
The compounds of the invention and their salts may be formulated as pharmaceutical compositions for administration. Such pharmaceutical compositions and processes for making the same are known in the art for both humans and non-human mammals. See, e.g., REMINGTON: THE SCIENCE AND PRACTICE OF PHARMACY, (A. Gennaro, et al, eds., 19th ed., Mack Publishing Co., 1995). Formulations can be administered through various means, including oral administration, parenteral administration such as injection (intramuscular, subcutaneous, intravenous, intraperitoneal) or the like; topical application with or without transdermal penetration such as dipping, spray, bathing, washing, pouring-on and spotting-on, and dusting, or the like. Additional active ingredients may be included in the formulation containing a compound of the invention or a salt thereof.
Carrier is used herein to describe any ingredient other than the active component(s) in a formulation. The choice of carrier will to a large extent depend on factors such as the particular mode of administration or application, the effect of the carrier on solubility and stability, and the nature of the dosage form.
C1-C5 alkyl refers to straight chain and branched alkyls having one to five carbon atoms, and includes methyl, ethyl, propyl, n-butyl, iso-butyl, pentyl, isopentyl, and neopentyl.
C2-C5 alkynyl refers to straight chain and branched alkynyls having two to five carbon atoms, and includes ethynyl, 1-propynyl, 2-propynyl, 1-butynyl, 3-methyl-l-butynyl, pentynyl, isopentynyl, and neopentynyl.
Halogen or halo refers to fluorine, bromine, chlorine, and iodine.
Haloalkyl as used herein refers to an alkyl (as noted above) substituted with one or more halo atoms. Such groups include trifluoromethyl, methylchloride, dichloromethyl, pentylchloride, butyl chloride, and isopropyl chloride.
Cyanoalkyl as used herein refers to an alkyl (as noted above) substituted with a cyano group.
Alkylthio as used herein refers to an alkyl (as noted above) having a sulfur in the group.
C3-C6 cycloalkyl refers to cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl. C3-C5 cycloheteroalkyl refers to a saturated ring which has 3 to 5 carbons and a hetero atom. The hetero atom may be sulfur, oxygen, nitrogen, or a sulfonyl group.
Diseases transmitted through parasites, particularly ectoparasites, are, for example bacterial, viral, rickettsial and protozoal vector-borne diseases. Examples of viral diseases transmitted through arboviruses, i.e. arthropod borne viruses, are Crimean-Congo Hemorhagic Fever (CCHF), Febrile illness, Papataci fever, Encephalitis, Meningitis, which are caused by Bunyaviridae such as Bunyavirus, Nairovirus or Phlebovirus; Bluetongue, meningoencephalits, Febrile illness, hemorhagic fever, which are caused by Reoviridae, such as Orbivirus, Colitivirus; Febrile illness, rash, encephalitis, polyarthritis, lymphadenitis, which are caused by Togaviridae, such as Sindbisvirus, Chikungunya Virus; tick-borne meningoencephalitis, Dengue hemorhagic fever, encephalitis, Febrile illness, Yellow fever, which are caused by Flaviviridae, such as Flavivirus (including diverse sub-groups). Examples of bacterial diseases transmitted through parasites are Rickettsiosis, such as Rocky Mountain spotted fever, tick typhus caused by infection through Rickettsia ssp; Tularemia caused by infection through Francisella tularensis; Borreliosis or Spirochaetosis, such as Lyme disease, or relapsing fever, caused, by infection through Borrelia ssp.; Ehrllichiosis caused by infection through Ehrlichia ssp.; Plague, caused by infection through Yersinia ssp. Examples of protozoal or rickettsial borne diseases are Babesiosis, such as Texas fever, red water disease, Q-fever caused by infection through Babesia ssp.; Theileriosis, such as
east coast fever, Mediterranean coast fever, caused by infection through Theileria ssp.; Nagana disease, Sleeping sickness caused by infection through Trypanosoma ssp., Anaplasmosis caused by infection through Anaplasma ssp.; Malaria caused by infection through Plasmodium ssp.; Leishmaniasis caused by infection through Leishmania ssp.
Given their activity, certain of the compounds of the invention are suitable as soil insecticides against pests in the soil, as well as insecticides for plants, such as cereals, cotton, rice, maize, soya, potatoes, vegetables, fruit, tobacco, hops, citrus, and avocados. Certain
compounds according to the invention are suitable for protecting plants and plant organs, for increasing the harvest yields, and for improving the quality of the harvested material which are encountered in agriculture, in horticulture, in forests, in gardens, and leisure facilities, and in the protection of stored products and of materials. They may be employed as plant protection agents.
All plants and plant parts can be treated in accordance with the invention. Plants are to be understood as meaning in the present context all plants and plant populations such as desired and undesired wild plants or crop plants (including naturally occurring crop plants). Crop plants can be plants which can be obtained by conventional plant breeding and optimization methods or by biotechnological and genetic engineering methods or by combinations of these methods, including the transgenic plants and including the plant cultivars protectable or not protectable by plant breeders' rights. Plant parts are to be understood as meaning all parts and organs of plants above and below the ground, such as shoot, leaf, flower and root, examples which may be mentioned being leaves, needles, stalks, stems, flowers, fruit bodies, fruits, seeds, roots, tubers and rhizomes. The plant parts also include harvested material, and vegetative and generative propagation material, for example cuttings, tubers, rhizomes, offshoots and seeds.
Treatment according to the invention of the plants and plant parts with the active compounds is carried out by conventional and known means, including directly acting on, or by allowing the compounds to act on, the surroundings, habitat or storage space by the customary treatment methods, for example by immersion, spraying, evaporation, fogging, scattering, painting on, injection and, in the case of propagation material, in particular in the case of seeds, also by applying one or more coats.
The compounds can be converted to the customary formulations, such as solutions, emulsions, wettable powders, water- and oil-based suspensions, powders, dusts, pastes, soluble
powders, soluble granules, granules for broadcasting, suspension-emulsion concentrates, natural materials impregnated with active compound, synthetic materials impregnated with active compound, fertilizers and microencapsulations in polymeric substances.
These formulations are produced in a known manner, for example by mixing the active compounds with extenders, that is liquid solvents and/or solid carriers, optionally with the use of surfactants, that is emulsifiers and/or dispersants and/or foam-formers. The formulations are prepared either in suitable plants or else before or during the application.
Suitable for use as auxiliaries are substances which are suitable for imparting to the composition itself and/or to preparations derived therefrom (for example spray liquors, seed dressings) particular properties such as certain technical properties and/or also particular biological properties. Typical suitable auxiliaries are extenders, solvents, and carriers.
Following are Schemes A-E and examples for preparing the compounds of the invention.
Scheme A
X = CI, Br, or I
Preparation 1
Methyl 2,2,2-trifluoro-l-(3,4,5-trichlorothiophen-2-yl)ethanone
Add a solution of n-BuLi (21.6 mL, 2.5 M in hexane, 54.0 mmol) to a solution of
2,3,4,5-tetrachloro-thiophene (10 g, 45.0 mmol) in dry THF (160 mL) at -78 °C and stir the mixture for 2 hours. Add a solution of trifluoro-acetic acid ethyl ester (9.59 g, 67.6 mmol) in THF (15 mL) and stir at -78 °C for additional 2.5 hours. Quench the reaction with saturated NH4CI solution (100 mL). Extract the aqueous mixture with EtOAc (100 mL X 3). The combined organic layers are washed with brine, dried over anhydrous Na2SC>4, and evaporate under vacuum. Purify the residue by a flash column on silica gel eluting with PE:EtOAc (10: 1 to 5: 1) to afford 2,2,2-trifluoro-l-(3,4,5-trichloro- thiophen-2-yl)-ethanone as a brown oil (10.4 g, 81.9%). 13F NMR (400 MHz, CDC13) δ -73.38 (s, 3F).
Preparation 2
Methyl 3-acetyl-4,5,6,7-tetrah drobenzo[c]thiophene- 1 -carboxylate
Add methyl 2-(2-oxopropylthio)acetate (35.4 g, 183.2 mmol) to a freshly prepared solution of solid sodium (8.78 g, 381.5 mmol) in dry MeOH (300 mL) at 0 °C, followed by addition of a solution of cyclohexane-l,2-dione (20 g, 152.6 mmol) in MeOH (30 mL). Stir the mixture at 0° C for 30 min and then at 50-60 °C for additional 1.5 hour. Remove the solvent under vacuum and dilute the residue with water (100 mL). Extract the aqueous mixture with ethyl acetate (100 mL X 3). The combined organic layers are washed with brine, dried over anhydrous Na2SC>4, and evaporated under vacuum. Purify the residue by a flash column on silica gel eluting with
PE:EtOAc (50: 1 to 30: 1) to afford methyl
3-acetyl-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxylate as a white solid (10 g, 27.6%). MS (m z): 239 (M+l).
Preparation 3
Methyl 3-acetyl-4,5,6,7-tetrah drobenzo[c]thiophene- 1 -carboxylate
Stir a mixture of Cyclopentane-l,2-dione (2.00 g, 20.4 mmol), methyl
2- (2-oxopropylthio)acetate ( 3.31 g, 20.4 mmol) and potassium carbonate (5.63 g, 40.8 mmol) in DMF (40 mL) at 80 °C for 4 hours. Filter off the mixture and remove the solvent under vacuum. Purify the residue by a flash column on silica gel eluting with PE:EtOAc (8: 1 to 6: 1) to afford
3- Acetyl-5,6-dihydro-4H-cyclopenta[c]thiophene-l- carboxylic acid methyl ester as a pale yellow solid (206mg, 4.5 %). MS (m/z): 225 (M+l).
Preparation 4
Methyl
3-(3-(3,5-dichlorophenyl)-4,4,4-trifluoro-3-hydroxybutanoyl)-4,5,6,7-tetrahydrobenzo[c]thiophe ne- 1 -carboxylate
Add a solution of LDA (2M in THF, 19.5 mL, 3.89 mmol) to a suspension of
3-acetyl-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxylate (7.4 g, 3.11 mmol) in dry THF (80
mL) at -78°C under N2. After stirring for 1.5 h, add l-(3,5-Dichloro-phenyl)- 2,2,2-trifiuoro-ethanone (9.9 g, 3.73 mmol) to the reaction mixture and stir the resultant mixture at the same temperature for additional 2 hours. Quench the reaction with saturated NH4CI aqueous solution. Extract the aqueous mixture with EtOAc (100 mL X 3). The combined organic layers are washed with brine, dried over anhydrous Na2S04 and concentrated under vacuum. Purify the residue by silica gel chromatograph (PE:EtOAc 50: 1) to afford methyl
3-(3-(3,5-dichlorophenyl)- 4,4,4-trifluoro-3-hydroxybutanoyl)
-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxylate as an orange solid (9.1 g, 60.4 %). MS (m/z) 481 (M+l).
The following compounds are prepared essentially by the method of Preparation 4.
Prep.
Chemical name Structure Physical data No.
3-[3-(3,5-Dichloro-ph
enyl)-4,4,4-trifluoro-
3 -hy droxy-butyry 1] -5 ,
6-dihydro-4H-cyclop
5 enta[c]thiophene- 1 -ca MS (m/z): 465 (M-1). rboxylic acid methyl
ester
3-[4,4,4-Trifiuoro-3-h
ydroxy-3-(3,4,5-trichl
oro-thiophen-2-yl)-bu
e- 1 -carboxylic acid
Chemical name Structure Physical data No. hene- 1 -carboxylic
acid methyl ester
Preparation 11
3-[3-(3,5-Dichloro-phenyl)-4,4,4-trifluoro-but-2-enoyl]-4,5,6,7-tetrahydro-benzo[c]thiophene-l- carboxylic acid methyl ester
Stir a mixture of methyl 3-(3-(3,5-dichlorophenyl)-4,4,4-trifluoro -3- hydroxyl
butanoyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxylate (9.1 g, 18.9 mmol), SOCl2 (9.0 g, 5.5 mL, 75.6 mmol) and pyridine (2.99 g, 3.1 mL, 37.8 mmol) in anhydrous DCM (100 mL) at ambient temperature overnight. Dilute the resultant mixture with saturated NH4CI aqueous solution. Extract the aqueous mixture with DCM (100 mLX 3). The combined organic layers are washed with brine, dried over anhydrous a2S04 and concentrated under vacuum. Purify the residue by silica gel chromatograph (PE:EtOAc 50: 1) to afford
3-[3-(3,5-Dichloro-phenyl)-4,4,4-trifluoro-but-2-enoyl]-4,5,6,7- tetrahydro-benzo[c]thiophene-l -carboxylic acid methyl ester as an orange solid (8.75 g, 100 %). (m/z): 463 (M+l).
The following compounds are prepared essentially by the method of Preparation
11.
Prep.
Chemical name Structure Physical data No.
3-[3-(3,5-Dichloro-ph
enyl)-4,4,4-trifluoro- but-2-enoyl]-5,6-dihy
12 dro-4H-cyclopenta[c] MS (m/z): 447 (M-1). thiophene- 1 -carboxyl
ic acid methyl ester
3-[4,4,4-Trifiuoro-3-(
3 ,4,5 -trichloro-thioph
en-2-yl)-but-2-enoyl]
-4,5,6,7-tetrahydro-be F3C 0
13 nzo [c]thiophene- 1 -ca MS (m/z): 501 (M-1). rboxylic acid methyl
ester]thiophene- 1 -car
boxylic acid methyl
ester
3-[4,4,4-Trifiuoro-3-(
3,4,5-trichloro-phenyl
)-but-2-enoyl]-4,5,6,7
14 MS (m/z): 497 (M+1).
-tetrahydro-benzo[c]t
hiophene- 1 -carboxyli
c acid methyl ester
Prep.
Chemical name Structure Physical data No.
3-[3-(3,5-Dichloro-4- fluoro-phenyl)-4,4,4-t
r ifluoro -but-2 -enoy 1] -
15 4,5,6,7-tetrahydro-be MS (m/z): 481 (M+l). nzo [c]thiophene- 1 -ca
rboxylic acid methyl
ester
3-[4,4,4-Trifluoro-3-(
3,4,5-trichloro-phenyl
)-but-2-enoyl]-5,6-di
16 hydro-4H-cyclopenta MS (m z): 483 (M+l).
xylic acid methyl
ester
3-[3-(3,5-Dichloro-4- fluoro-phenyl)-4,4,4-t
r ifluoro -but-2 -enoy 1] -
17 5,6-dihydro-4H-cyclo 0 yJi MS (m/z): 467 (M+l). penta[c]thiophene- 1 -c
arboxylic acid methyl
ester
Preparation 18
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl] -4,5,6,7-tetrahydro-benzo[c]thiophene-l-carboxylic acid methyl ester
Stir a mixture of 3-[3-(3,5-Dichloro-phenyl)-4,4,4-trifluoro-but-2-enoyl]- 4,5,6,7- tetrahydro-benzo[c]thiophene-l-carboxylic acid methyl ester (8.75 g, 18.9 mmol), NaOH (2.65 g, 66.1 mmol) and NH20H-HC1 (2.6 g, 37.8 mmol) in MeOH (60 mL) and water (15 mL) at room temperature for 2.5 hour. After removal of solvent under vacuum, dilute the residue with ice water (50 mL). Acidify the aqueous mixture with cone. HC1 to pH = 1 and extract the resultant mixture with EtOAc (50 mL X 3). The combined organic layers are washed with brine, dried over anhydrous Na2SC>4 and concentrated under vacuum. Purify the residue by silica gel
chromatograph (PE:EtOAc 50: 1) to afford
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7-tetrahydro-benz o[c]thiophene-l-carboxylic acid methyl ester as an orange solid (8.1 g, 93.1%). MS (m/z): 478 (M+l).
The following compounds are prepared essentially by the method of Preparation
18.
Prep.
Chemical name Structure Physical data No.
3-[5-(3,5-Dichloro-phen
yl)-5-trifiuoromethyl-4,5
-dihydro-isoxazol-3-yi]-
19 MS (m/z): 464 (M+1).
5 ,6-dihydro-4H-cyclope
CI
nta[c]thiophene- 1 -carbo
xylic acid methyl ester
3-[5-(3,4,5-Trichloro-thi
ophen-2-yl)-5-trifiuorom
ethyl-4,5-dihydro-isoxaz
arboxylic acid methyl
ester
3-[5-(3,4,5-Trichloro-thi
ophen-2-yl)-5-trifiuorom
ethyl-4,5-dihydro-isoxaz 0
21 ol-3-yl]-4,5,6,7-tetrahydr MS (m/z): 512 (M+1). o-benzo [c]thiophene- 1 -c
arboxylic acid methyl
ester
3-[5-(3,5-Dichloro-4-fiu
22 MS (m/z): 496 (M+1). oro-phenyl)-5-trifluorom
ethyl-4,5-dihydro-isoxaz
Prep.
Chemical name Structure Physical data No. ol-3-yl]-4,5,6,7-tetrahydr
o-benzo [c]thiophene- 1 -c
arboxylic acid methyl
ester
3-[5-(3,4,5-Trichloro-ph
enyl)-5-trifluoromethyl- 4,5-dihydro-isoxazol-3-y
23 MS (m/z): 498 (M+1).
1] -5 ,6-dihydro-4H-cyclo
penta[c]thiophene- 1 -carb
oxylic acid methyl ester
3-[5-(3,5-Dichloro-4-flu
oro-phenyl)-5-trifluorom
ethyl-4,5-dihydro-isoxaz
24 ol-3-yl]-5,6-dihydro-4H- MS (m/z): 482 (M+1). cyclopenta[c]thiophene- 1 -carboxylic acid methyl
ester
Preparation 25
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol- -4,5, 6,7-tetrahydro-benzo[c]thiophene-l -carboxylic acid
Stir a mixture of 3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro- isoxazol-3-yl]-4,5,6,7-tetrahydro-benzo[c]thiophene-l-carboxylic acid methyl ester (8.1 g, 17.0 mmol) and LiOH-H20 (3.57 g, 84.9 mmol) in MeOH (64 mL) and water (16 mL) at room temperature overnight. After removal of organic solvent under vacuum, dilute the residue with ice water (80 mL). Acidify the aqueous mixture with cone. HC1 to pH = 1, and extract the resultant mixture with EtOAc (100 mL X 3). The combined organic layers are washed brine, dried over anhydrous Na2SC>4 and concentrated under vacuum. Purify the residue by silica gel chromatograph (PE:EtOAc 1 : 1) to afford 3-[5-(3,5-Dichloro- phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7-tetrahydro-benzo[c]thiophene-l-car boxylic acid as pale yellow solid (6.8 g, 86.4 %). MS (m/z): 464 (M+l).
The following compounds may be prepared essentially by the method of
Preparation 25.
Chemical name Structure Physical data No.
3-[5-(3,4,5-Trichloro-ph
enyl)-5-trifluoromethyl- 0
4,5-dihydro-isoxazol-3-y
28 MS (m/z): 496 (M-1). l]-4,5,6,7-tetrahydro-ben
zo [c]thiophene- 1 -carbox
ylic acid
3-[5-(3,5-Dichloro-4-flu
oro-phenyl)-5-trifluorom 0
ethyl-4,5-dihydro-isoxaz
29 MS (m/z): 480 (M-1). ol-3-yl]-4,5,6,7-tetrahydr
o-benzo [c]thiophene- 1 -c
arboxylic acid
3-[5-(3,5-Dichloro-4-flu
oro-phenyl)-5-trifluorom
ethyl-4,5-dihydro-isoxaz
30 MS (m/z): 466 (M-1). ol-3-yl]-5,6-dihydro-4H- cyclopenta[c]thiophene- 1 -carboxylic acid
3-[5-(3,4,5-Trichloro-ph
enyl)-5-trifluoromethyl- 4,5-dihydro-isoxazol-3-y
31 MS (m/z): 482 (M-1).
1] -5 ,6-dihydro-4H-cyclo
penta[c]thiophene- 1 -carb
oxylic acid
Example 32
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2-oxopyrrolidm
3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide
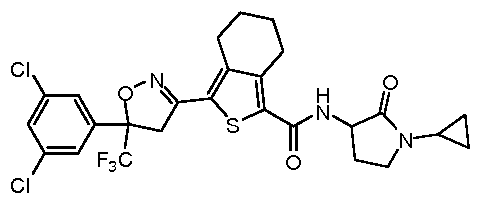
-4,5,6,7-tetrahydro-benzo[c]thiophene-l-carboxylic acid (850 mg, 1.84 mmol), HATU (837 mg, 2.20 mmol) and DIEA (539 mg, 0.8 mL, 4.6 mmol) in DCM (8 mL) at room temperature for 15 min, followed by addition of (R)-3-aminopyrrolidin-2-one hydrochloride (315 mg, 2.76 mmol). Stir the reaction mixture at room temperature for additional 1.5 hour. Dilute the reaction mixture with water (20 mL) and extract with DCM (20 mL X 3). The combined organic layers are washed brine, dried over anhydrous Na2SC>4 and concentrated under vacuum. Purify the residue by preparative HPLC to afford 3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5- dihydroisoxazol-3-yl)-N-((R)-2-oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carb oxamide as a white solid (730 mg, 73.0 %). MS (m/z): 546 (M+l); 1H NMR (400 MHz, CDC13) δ 7.52-7.46 (m, 3H), 6.49 (s, 1H), 5.83 (s, 1H), 4.51-4.48 (m, 1H), 4.22 (d, J=17.2, 1H), 3.85 (d, J=17.2, 1H), 3.52-3.47 (m, 2H), 3.09-2.87 (m, 5H), 2.12-2.03 (m, 1H), 1.89-1.73 (m, 4H).
The following compounds may be prepared essentially by the method of Example
Ex.
Chemical name Structure Physical data o.
N,N-Dimethyl-2-(4-{3-[ MS (m/z): 659 (M+l); 1H 5 - (3 ,4 ,5 -trichloro -thioph NMR (400 MHz, CDC13) δ en-2-yl)-5-trifluorometh 4.07-4.02 (m, 2H), 3.68-3.80 yl-4,5-dihydro-isoxazol- (m, 4H), 3.49-3.41 (m, 2H),
33
3 -yl] -4,5 ,6 ,7-tetrahydro- 3.09-3.01 (s, 3H), 3.01-2.95 (s, benzo[c]thiophene- 1 -car 3H), 2.91-2.85 (m, 2H), bonyl}-piperazin-l-yl)-a 2.84-2.78 (m, 4H), 2.69-2.63 cetamide (m, 2H), 1.87-1.72 (m, 4H).
({3-[5-(3,5-Dichloro-phe
nyl)-5-trifluoromethyl-4,
5-dihydro-isoxazol-3-yl]
34 -4,5,6,7-tetrahydro-benz MS (m/z): 535 (M+l). o [c]thiophene- 1 -carbony
l}-amino)-acetic acid
methyl ester
({3-[5-(3,4,5-Trichloro-t
hiophen-2-yl)-5-trifluoro
methyl-4,5-dihydro-isox
35 azol-3-yl]-4,5,6,7-tetrah MS (m/z): 575 (M+l). ydro-benzo[c]thiophene- 1 -carbonyl} -amino)-acet
ic acid methyl ester
Chemical name Stracture Physical data o.
2.58-2.48 (m, 2H), 2.29-2.26 (s,
1H).
N-(2-oxo-2-(2,2,2-trifluo MS (m/z): 636(M+1); 'H NMR roethylamino)ethyl)-3-(5 (400 MHz, CDC13) δ 7.62 (s, -(3,4,5-trichlorophenyl)- 2H), 7.32 (br, 1H), 6.95 (br,
44 5-(trifluoromethyl)-4,5-d 1H), 4.26-4.25 (m, 2H), ihydroisoxazol-3-yl)-4,5, 4.06-3.83 (m, 3H), 3.66 (d, J = 6,7-tetrahydrobenzo[c]th 16.8 Hz, 1H), 2.98-2.89 (m, iophene- 1 -carboxamide 4H), 1.79 (br, 4H).
MS (m/z): 580(M+1); 'H NMR
N-((R)-2-oxopyrrolidin- (400 MHz, CDCI3) δ 7.62 (s, 3-yl)-3-(5-(3,4,5-trichlor
2H), 6.62 (br, lH), 6.17 (br, ophenyl)-5-(trifluoromet
1H), 4.47-4.46 (m, 1H),
45 hyl)-4,5-dihydroisoxazol
4.07-4.02 (m, 1H), 3.75-3.64 -3-yl)-4,5,6,7-tetrahydro
(m, 1H), 3.49-3.43 (m, 2H), benzo[c]thiophene- 1 -car
3.02-2.88 (m, 2H), 2.09-2.01 boxamide
(m, 1H), 1.77 (br, 4H).
Ex.
Chemical name Structure Physical data o.
N-(2-(cyanomethylamin MS (m/z): 593(M+1); 'H NMR o)-2-oxoethyl)-3-(5-(3,4, (CDC13, 400 MHz) δ 7.63 (s, 2 5-trichlorophenyl)-5-(trif H), 7.47 (m, 1 H), 6.81 (m, 1
oisoxazol-3-yl)-4,5,6,7-t J=17.2, 1 H), 3.70 (d, J=17.2, 1 etrahydrobenzo[c]thioph H), 2.90 (m, 4 H), 1.79 (m, 4 ene- 1 -carboxamide H).
MS (m/z): 592(M+1). 'H NMR
N-(2-oxo-2-(prop-2-ynyl
(CDC13, 400 MHz) δ 7. 63 (s, 2 amino)ethyl)-3-(5-(3,4,5
H), 6.78 (m, 1 H), 6.50 (m, 1 -trichlorophenyl)-5-(trifl
H), 4.15 (m, 2 H), 4.10 (m, 2
47 uoromethyl)-4,5-dihydro
H), 4.05 (d, J=16.8, 1 H), 3.67 isoxazol-3-yl)-4,5,6,7-tet
(d, J=16.8, 1 H), 2.95 (m, 2 H), rahydrobenzo [c]thiophen
2.90 (m, 2 H), 2.26 (m, 1 H), e-1 -carboxamide
1.79 (m, 4H).
3-(5-(3,5-dichloro-4-fluo
MS (m/z): 620(M+1); 'H NMR rophenyl)-5-(trifluorome
(CDOD3, 400 MHz) δ 7.76 (d, thyl)-4,5-dihydroisoxazo
J=6.4, 2H), 4.25 (d, J=17.6, l-3-yl)-N-(2-oxo-2-(2,2,2
48 1H), 4.07 (s, 2H), 4.04-3.92 (m, -trifluoroethylamino)eth
3H), 3.01-2.98 (m, 2H), yl)-4,5,6,7-tetrahydroben
2.90-2.89 (m, 2H), 1.79 (m, zo [c]thiophene- 1 -carbox
4H).
amide
Ex.
Chemical name Structure Physical data o.
MS (m/z): 564(M+1); 'H NMR
3-(5-(3,5-dichloro-4-fluo (CDOD3, 400 MHz) δ 7.76 (d, rophenyl)-5-(trifluorome J=6.4, 2H), 4.63 (t, J=9.6, 1H), thyl)-4,5-dihydroisoxazo 4.24 (d, J=17.6, 1H), 4.02 (d,
49 l-3-yl)-N-((R)-2-oxopyrr J=17.6, 1H), 3.44-3.40 (m, 2H), olidin-3-yl)-4,5,6,7-tetra 2.99-2.98 (m, 2H), 2.89 (m, hydrobenzo [c]thiophene- 2H), 2.59-2.52 (m, 1H), 1 -carboxamide 2.23-2.18 (m, 1H), 1.79-1.77
(m, 4H).
N-(2-(cyanomethylamin
o)-2-oxoethyl)-3-(5-(3,5- MS (m/z): 577(M+1); 'H NMR dichloro-4-fluorophenyl) (CDOD3, 400 MHz) δ 7.77 (d, -5-(trifluoromethyl)-4,5- J=6.4, 2H), 4.28-4.21 (m, 3H),
50
dihydroisoxazol-3-yl)-4, 4.05-4.00 (m, 3H), 3.00(t, 5,6,7-tetrahydrobenzo[c] J=6.0, 2H), 2.93-2.90 (m, 2H), thiophene- 1 -carboxamid 1.81-1.80 (m, 4H).
e
3-(5-(3,5-dichloro-4-fluo MS (m/z): 576(M+1); 'H NMR rophenyl)-5-(trifluorome (CDOD3, 400 MHz) δ 7.76 (d, thyl)-4,5-dihydroisoxazo J=6.4, 2H), 4.24 (d, J=17.6,
51 l-3-yl)-N-(2-oxo-2-(prop 1H), 4.03-3.99 (m, 5H), 2.98 (t, -2-ynylamino)ethyl)-4,5 , J=6.0, 2H), 2.88-2.86 (m, 2H), 6,7-tetrahydrobenzo[c]th 2.62 (t, J=2.4, 1H), 1.79-1.77 iophene- 1 -carboxamide (m, 4H).
Chemical name Structure Physical data o.
MS (m/z): 547 (M+l); IH NMR (CDC13, 400 MHz) 57.48
3-(5-(3,5-dichlorophenyl (s, 2H), 7.42 (s, IH), 6.23 (s, )-5-(trifluoromethyl)-4,5 IH), 4.08-4.02 (m, 2H), -dihydroisoxazol-3-yl)-N 3.91-3.86 (m, IH), 3.81-3.76
64 -((tetrahydrofuran-2-yl) (m, 2H), 3.69-3.65 (m, J=16 methyl)-4,5,6,7-tetrahyd
Hz, IH), 3.34-3.28 (m, IH), robenzo [c]thiophene- 1 -c 3.00-2.85 (m, 4H), 2.06-1.98 arboxamide (m, IH), 1.96-1.89 (m, 2H),
1.79-1.77 (m, 4H), 1.64-1.59
(m, IH).
MS (m/z): 537 (M+l); IH
3-(5-(3,5-dichlorophenyl
NMR (CDC13, 400 MHz) 5 )-5-(trifluoromethyl)-4,5
7.49 (s, 2H), 7.43 (s, IH), 6.33 -dihydroisoxazol-3-yl)-N
(s, IH), 4.07-4.03 (d, J=16 Hz,
65 -(2-(methylthio)ethyl)-4,
Y IH), 3.69-3.62 (m, 3H), 2.98 (s, 5,6,7-tetrahydrobenzo[c] o
2H), 2.92-2.90 (m, 2H), thiophene- 1 -carboxamid
2.76-2.74 (m, 2H), 2.14 (s, 3H), e
1.80-1.79 (m, 4H).
3-(5-(3,5-dichlorophenyl
MS (m/z): 559 (M+l); IH )-5-(trifluoromethyl)-4,5
NMR (CDC13, 400 MHz) 5 -dihydroisoxazol-3-yl)-N
7.49 (s, 2H), 7.43 (s, IH),
66 -(3 ,3 ,3 -trifluoropropyl)-4
6.08-6.05 (m, IH), 4.06-4.02 ,5,6,7-tetrahydrobenzo[c o
(d, J=16 Hz, IH), 3.72-3.64 (m, ]thiophene- 1 -carboxami 3H), 2.94-2.85 (m, 4H), de
2.51-2.40 (m, 2H), 1.80-1.78
Ex.
Chemical name Structure Physical data o.
(m, 4H).
MS (m/z): 561 (M+l); 1H
3-(5-(3,5-dichlorophenyl
NMR (CDC13, 400 MHz) δ )-5-(trifluoromethyl)-4,5
7.49 (s, 2H), 7.43 (s, 1H), 6.85 -dihydroisoxazol-3-yl)-N
(s, 1H), 4.21 (s, 1H), 4.09-4.02
67 -(3-hydroxycyclohexyl)- (m, 2H), 3.69-3.64 (d, J=20 Hz, 4,5,6,7-tetrahydrobenzo[
1H), 2.96-2.89 (m, 5H), c]thiophene- 1 -carboxami
2.05-1.78 (m, 9H), 1.49-1.38 de
(m, 2H).
MS (m/z): 558 (M+l); 1H
3-(5-(3,5-dichlorophenyl NMR (CDC13, 400 MHz) δ )-5-(trifluoromethyl)-4,5 8.36 (s, 1H), 8.32-8.29 (m, 1H), -dihydroisoxazol-3-yl)-N 8.16 (d, J=2.4 Hz, 1H),
68 -(5-fluoropyridin-2-yl)-4 7.51-7.44 (m, 3H), 7.43 (s, 1H), ,5,6,7-tetrahydrobenzo[c 4.09-4.05 (d, J=16 Hz, 1H), ]thiophene- 1 -carboxami 3.71-3.67 (d, J=16 Hz, 1H), de 3.09 (s, 2H), 2.94-2.93 (m, 2H),
1.83 (s, 4H)
Ex.
Chemical name Structure Physical data o.
MS (m/z): 547 (M+l); IH
3-(5-(3,5-dichlorophenyl
NMR (CDC13, 400 MHz) δ )-5-(trifluoromethyl)-4,5
7.49 (s, 2H), 7.40 (s, IH), 6.23 -dihydroisoxazol-3-yl)-N
(d, .7=9.6 Hz, IH), 4.18-4.16 (s,
69 -(tetrahydro-2H-pyran-3-
CF3 o IH), 4.07-4.03 (d, J=16 Hz, yl)-4,5,6,7-tetrahydroben CI
IH), 3.83-3.77 (m, 2H), zo [c]thiophene- 1 -carbox
3.69-3.57 (m, 3H), 2.98-2.90 amide
(m, 4H), 1.90-1.75 (m, 8H).
MS (m/z): 535 (M+l); IH
3-(5-(3,5-dichlorophenyl NMR (CDC13, 400 MHz) δ )-5-(trifluoromethyl)-4,5 7.49 (s, 2H), 7.44 (s, 1H), 6.18 -dihydroisoxazol-3-yl)-N (d, J= 7.6 Hz, IH), 5.40-5.34
70
-(thietan-3-yl)-4,5,6,7-tet ) F3C 0 S (m, 1H), 4.06 (d, J = 17.2 Hz,
CI
rahydrobenzo [c]thiophen IH), 3.66 (d, J= 17.2 Hz, IH), e-l-carboxamide 3.46-3.36 (m, 4H), 2.97-2.90
(m, 4H), 1.81-1.78 (m, 4H).
MS (m/z): 547 (M+l); IH
3-(5-(3,5-dichlorophenyl NMR (CDC13, 400 MHz) δ )-5-(trifluoromethyl)-4,5 7.47 (s, 2H), 7.41 (s, IH), -dihydroisoxazol-3-yl)-N 5.74-5.72 (d, J=7.6 Hz, IH),
71 -(tetrahydro-2H-pyran-4- 4.19-4.1 1 (m, IH), 4.10-4.06 yl)-4,5,6,7-tetrahydroben
(d, J=16 Hz, IH), 4.04-3.96 (m, zo [c]thiophene- 1 -carbox 2H), 3.69-3.65 (d, J=16 Hz, amide IH), 3.53-3.43 (m, 2H),
2.94-2.88 (m, 4H), 2.00-1.97 (m, 2H), 1.77 (s, 4H), 1.60-1.50
Ex.
Chemical name Stracture Physical data o.
(m, 2H).
MS (m/z): 586 (M+l); 1H
N-( 1 -cyclopropyl-2-oxo NMR (CDC13, 400 MHz) δ pyrrolidin-3-yl)-3-(5-(3, 7.49 (s, 2 H), 7.43 (s, 1H), 6.50 5-dichlorophenyl)-5-(trif (brs, 1 H), 4.43-4.40 (m, 1 H),
72 luoromethyl)-4,5-dihydr 4.04 (d, J=16.8, 1 H), 3.65 (d, oisoxazol-3-yl)-4,5,6,7-t
J = 16.8, 1 H), 3.42-3.30 (m, 2 etrahydrobenzo[c]thioph H), 2.99-2.82 (m, 5 H), ene- 1 -carboxamide 1.91-1.80 (m, 5 H), 0.83-0.763
(m, 4 H)
N-(2-oxo-2-(prop-2-ynyl
MS (m/z): 600 (M+l); 1H amino)ethyl)-3-(5-(3,4,5
NMR (CDC13, 400 MHz) δ -trichlorothiophen-2-yl)- 6.92-6.77 (m, 2H), 4.21-4.09
74 5-(trifluoromethyl)-4,5-d
cr 0 (m, 4H), 4.08-4.04 (m, 2H), ihydroisoxazol-3-yl)-4,5,
3.09-2.88 (m, 4H), 1.87-1.71 6,7-tetrahydrobenzo[c]th
(m, 4H).
Chemical name Stracture Physical data o.
1.79 (s, 4H), 1.64-1.50 (m, 2H).
MS (m/z): 622 (M+l); 1H
N-( 1 -cyclopropyl-2-oxo
NMR (CDC13, 400 MHz) δ pyrrolidin-3-yl)-3-(5-(3,
8.00 (s, 2 H), 6.53 (brs, 1 H), 4,5-trichlorophenyl)-5-(t
4.43 (m, 1 H), 4.04 (d, J=17.2,
78 rifluoromethyl)-4,5-dihy
1 H), 3.65 (d, J=17.2, 1 H), droisoxazol-3-yl)-4,5,6,7
3.36 (m, 2 H), 2.83 (m, 5 H), -tetrahydrobenzo[c]thiop
2.73 (m, 1 H), 1.84 (m, 5 H), hene- 1 -carboxamide
0.79 (m, 4 H).
3-[5-(3,5-Dichloro-4-flu MS (m/z): 585 (M+l); 1H oro-phenyl)-5-trifluorom NMR (CDC13, 400 MHz) δ ethyl-4,5-dihydro-isoxaz 7.76 (d, J=6.0, 2H), 4.65-4.56 ol-3-yl]-4,5,6,7-tetrahydr (m, 1H), 4.53 (t, J=4.8, 2H),
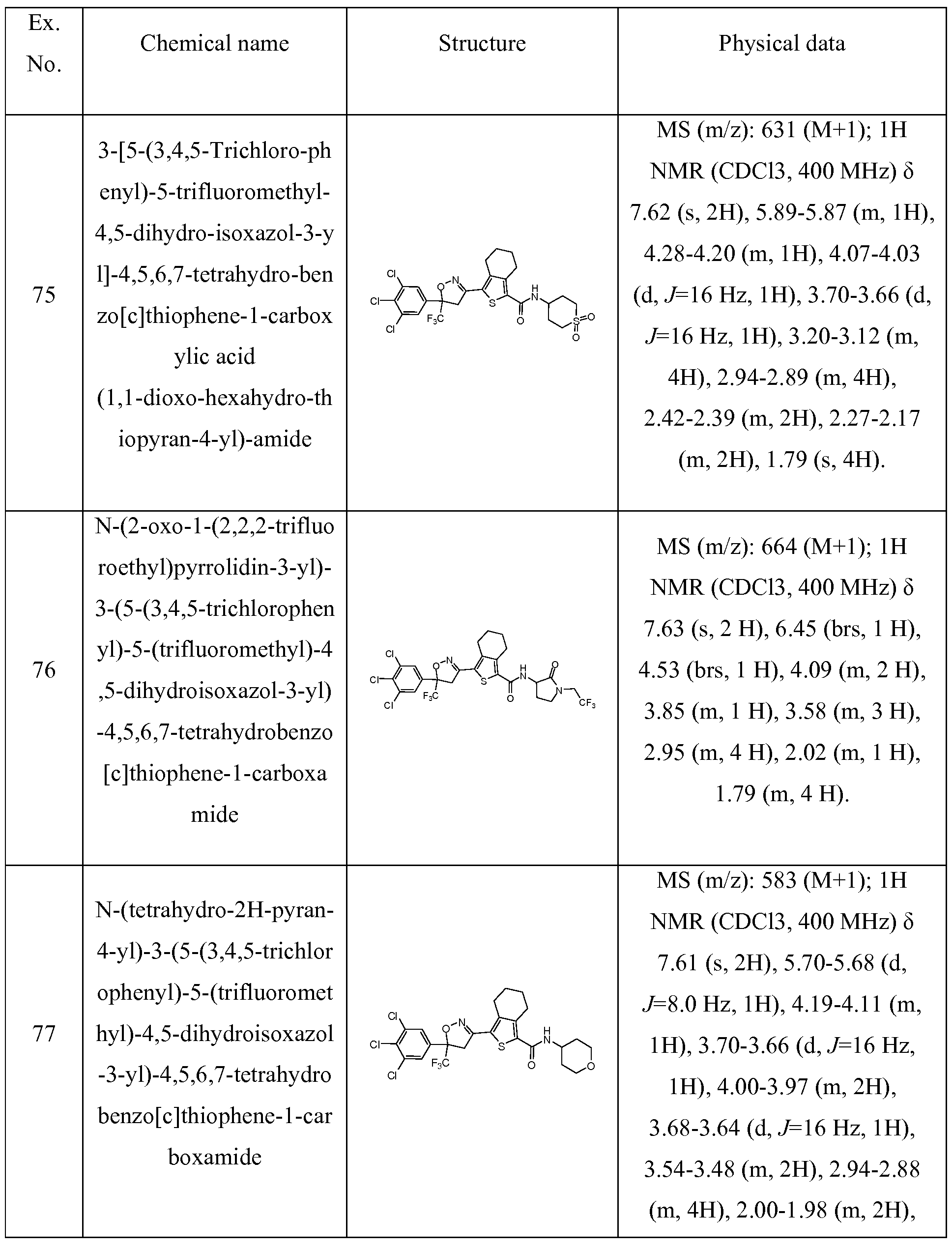
79
o-benzo [c]thiophene- 1 -c 4.28-4.23 (m, 3H), 4.02 (d, arboxylic acid J=17.6, lH), 2.97 (t, J=6.0, (1,1 -dioxo-thietan-3-yl)- 2H), 2.90 (t, J=6.0, 2H), amide 1.80-1.76 (m, 4H).
Ex.
Chemical name Structure Physical data o.
3-[5-(3,5-Dichloro-4-flu MS (m/z): 613 (M+l); 1H oro-phenyl)-5-trifluorom NMR (CDC13, 400 MHz) δ ethyl-4,5-dihydro-isoxaz 7.56(d,J=6.0,2H), 5.75 (d, ol-3-yl]-4,5,6,7-tetrahydr J=7.2, 1 H),4.06 (d,J=16.8, 1
80
o-benzo [c]thiophene- 1 -c H),3.65 (d,J=16.8, 1 H),3.13 arboxylic acid (m,4H),2.91 (m,4H),2.41 (1,1 -dioxo-hexahydro-th (m,2H),2.23 (m, 2 H), 1.84 iopyran-4-yl)-amide (m, 4 H).
3-(5-(3,5-dichloro-4-fluo MS (m/z): 577 (M+l); 1H rophenyl)-5-(trifluorome NMR (CDC13, 400 MHz) δ thyl)-4,5-dihydroisoxazo 7.48(d, J=6.0,2H), 5.67 (d,
81 1-3 -yl)-N-(4-oxocyclohe J=6.8, 1H),4.33 (m, 1H), xyl)-4,5,6,7-tetrahydrobe
3.98(d,J=16.8, lH),3.58(d, nzo[c]thiophene- 1 -carbo J=16.8, 1H), 2.82 (m, 4H), 2.40 xamide (m, 6H), 1.65 (m, 6H)
3-(5-(3,5-dichloro-4-fluo MS (m/z): 646 (M+l); 1H rophenyl)-5-(trifluorome NMR (CDC13 , 400 MHz) δ 7. thyl)-4,5-dihydroisoxazo 50
l-3-yl)-N-(2-oxo-l -(2,2,2 6.44 (dd, J,=4.8, J2=12.0, 1 H),
82
-trifluoroethyl)pyrrolidin 4.46 (m, 1 H),3.98 (m, 2 H), -3-yl)-4,5,6,7-tetrahydro 3.78 (m, 1 H), 3.55 (m, 3 H), benzo[c]thiophene- 1 -car 2.83 (m, 5H), 1.963 (m, 1 H), boxamide 1.71 (m,4H).
Ex.
Chemical name Structure Physical data o.
MS (m/z): 565 (M+l); 1H
3-(5-(3,5-dichloro-4-fluo NMR (CDC13, 400 MHz) δ rophenyl)-5-(trifluorome 7.49 (d, J=5.6, 2 H), 5.59 (d, thyl)-4,5-dihydroisoxazo J=7.6, 1 H), 4.10 (m, 1 H), 3.98
83 l-3-yl)-N-(tetrahydro-2H (d, J=16.8, 1 H), 3.91 (m, 2 H), -pyran-4-yl)-4,5,6,7-tetra 3.58 (d, J=16.8, 1 H), 3.45 (m, hydrobenzo [c]thiophene- 2 H), 2.84 (m, 4 H), 1.95 (m, 2 1 -carboxamide H), 1.72 (m, 4 H), 1.49 (m, 3
H).
MS (m/z): 604 (M+l); 1H NMR (CDC13, 400 MHz) δ
N-( 1 -cyclopropyl-2-oxo
7.77 (d, J=6.0, 2H), 4.62 (t, pyrrolidin-3-yl)-3-(5-(3,
J=9.6, 1H), 4.24 (d, J=17.6, 5-dichloro-4-fluorophen
1H), 4.02 (d, J=17.6, 1H), yl)-5-(trifluoromethyl)-4
84 3.43-3.39 (m, 2H), 2.99-2.96 ,5-dihydroisoxazol-3-yl)
(m, 2H), 2.90-2.88 (m, 2H), -4,5,6,7-tetrahydrobenzo
2.75-2.69 (m, 1H), 2.50-2.42 [c]thiophene- 1 -carboxa
(m, 1H), 2.10-1.99 (m, 1H), mide
1.81-1.78 (m, 4H), 0.85-0.82
(m, 4H).
3-(5-(3,5-dichlorophenyl MS (m/z): 545 (M+l); 1H )-5-(trifluoromethyl)-4,5 NMR (CDC13, 400 MHz) δ
85 -dihydroisoxazol-3-yl)-N 7.52-7.44 (m, 3H), 5.72-5.68 -(4-oxocyclohexyl)-5,6- o (m, 1H), 4.48-4.37 (m, 1H), dihydro-4H-cyclopenta[c 4.04-3.97 (d, J=17.2, 1H), ]thiophene- 1 -carboxami 3.65-3.59 (d, J=17.2, 1H),
Ex.
Chemical name Structure Physical data o. de 2.92-2.84 (m, 4H), 2.58-2.48
(m, 6H), 2.47-2.31 (m, 2H), 1.84-1.71 (m, 2H).
MS (m/z): 614 (M+l); 1H
3-(5-(3,5-dichlorophenyl
NMR (CDC13, 400 MHz) )-5-(trifluoromethyl)-4,5
57.52-7.44 (m, 3H), 6.43-6.38 -dihydroisoxazol-3-yl)-N
(s, 1H), 4.52-4.48 (m, 1H), -(2-oxo- 1 -(2,2,2-trifluor
86 N 4.17-4.05 (m, 2H), 4.04-3.97 oethyl)pyrrolidin-3-yl)-5 Q YNY
(d, .7=17.2, 1H), 3.89-3.77 (m, ,6 -dihydro -4H- cy clopent
1H), 3.68-3.51 (m, 3H), a[c]thiophene- 1 -carboxa
2.93-2.82 (m, 5H), 2.58-2.46 mide
(m, 2H), 2.08-1.97 (m, 1H).
Example 87
(A) 3-((R)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2- oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide and
(B) 3-((S)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2- oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide
Separate 3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)
-N-((R)-2-oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide (6.2 g 11.29 mmol) by SFC (Column: Chiralcel OD 250 X 30mm I.D., 5um. Mobile phase: Supercritical C02/ MeOH=60/40, Flow rate:200 ml/min) to afford two diastereoisomers
3-((R)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2-oxo pyrrolidin-3-yl)- 4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide (2.7 g, 4.92 mmol) and 3-((S)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2-oxopyrroli din-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide (2.6 g, 4.74 mmol) as a white solid.
(A) MS (m/z): 619.1 (M+73). !H NMR (CDC13, 400 MHz) δ 7.49 (s, 2H), 7.42 (s, 1H ), 6.56 (d, J=4.4 Hz 1H), 6.03 (s, 1H), 4.50-4.44 (m, 1H), 4.06-4.02 (d, J=16 Hz, 1H), 3.69-3.65 (d, J=16 Hz, 1H), 3.48-3.43 (m, 2H), 3.03-2.98 (m, 5H), 2.11-2.00 (m, 1H), 1.78 (s, 4H).
(B) MS (m/z): 619.1 (M+73). 1H MR (CDC13, 400 MHz) δ 7.49 (m, 2H), 7.41 (m, 1H ), 6.77-6.73 (m, 1H), 6.38-6.31 (m, 1H), 4.51-4.45 (m, 1H), 4.05-4.01 (d, J=16 Hz, 1H), 3.71-3.67 (d, J=16 Hz, 1H), 3.49-3.39 (m, 2H), 3.00-2.85 (m, 5H), 2.04-2.03 (m, 1H), 1.74 (s, 4H).
Scheme B
NH R1R2, Pyridine
Ar
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- 4,5,6,7-tetrahydrobenzo[c]thiophene-l-carbonyl chloride
Stir a mixture of
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7-tetrahydro-benz o[c]thiophene-l-carboxylic acid (600 mg, 1.2 mmol), 2 drops DMF in oxalyl dichloride (5 mL) at ambient temperature for 3 hours. Remove the solvent under vacuum to afford
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- 4,5,6,7-tetrahydrobenzo[c]thiophene-l-carbonyl chloride as a yellow solid (615 mg, 98 %).
N-(4-carbamoylphenyl)-3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5- dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo [c]thiophene- 1 -carbonyl chloride (48 mg, 0.1 mmol) and 4-aminobenzamide (27 mg, 0.2 mmo) in pyridine (3 mL) at ambient temperature overnight. After removal solvent under vacuum, purify the residue by preparative HPLC to afford
N-(4-carbamoylphenyl)-3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl) -4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide as a white solid (36 mg, 62.0%). MS (m/z): 582.1 (M+l). 1H NMR (CDC13, 400 MHz) δ 7.84 (d, J=8.4 Hz, 2H), 7.65-7.63 (m, 3H), 7.49 (s, 2H), 7.44 (s, 1H), 6.03 (s, 1H), 5.61 (s, 1H), 4.09-4.05 (d, J=16 Hz, 1H), 3.72-3.68 (d, J=16 Hz, 1H), 3.07 (s, 2H), 2.94 (s, 2H), 1.76 (s, 4H).
The following compound is prepared essentially by the method of Example 90.
Chemical name Structure Physical data No.
-2-yl)-amide
Scheme C
Example 92
2-(3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- 4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamido)acetic acid
Stir a mixture of methyl 2-(3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5- dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamido)acetate (534 mg, 1.0 mmol) and LiOH-H20 (168 mg, 4.0 mmol) in MeOH (20 mL) and water (5 mL) at room
temperature for overnight. After removal of organic solvent under vacuum, dilute the residue with ice water (10 mL). Acidify the aqueous mixture with cone. HC1 to pH = 1 , and extract the resultant mixture with EtOAc (15 mL X 3). The combined organic layers are washed brine, dried over anhydrous Na2SC>4 and concentrated under vacuum. Purify the residue by silica gel chromatograph (PE:EtOAc 1 : 1) to afford
2-(3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobe nzo[c]thiophene-l-carboxamido)acetic acid as a pale yellow solid (427 mg, 82.0 %). MS (m/z): 521(M+1).
The following compound is prepared essentially by the method of Example 92.
Example 94
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,5-dichlorophenyl)-5- (trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l- carboxamide
Stir a mixture of 2-(3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5- dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamido)acetic acid (230 mg, 0.44 mmol), HATU (251 mg, 0.66 mmol) and Et3 (133 mg, 1.32 mmol) in DCM (5 mL) at room temperature for 15 min, followed by addition of 2-aminoacetonitrile hydrochloride (61 mg, 0.65 mmol). Stir the reaction mixture at room temperature for additional 1.5 hour. Dilute the reaction mixture with water (20 mL) and extract with DCM (20 mLX 3). The combined organic layers are washed with brine, dried over anhydrous Na2SC>4 and concentrated under vacuum. Purify the residue by preparative HPLC to afford
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,5- dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophe ne-l-carboxamide as a white solid (110 mg, 44.7 %). MS (m/z): 559.1 (M+l). 1H NMR
(DMSC /e, 400 MHz) δ 8.64 (t, J=5.6 Hz, 1H), 8.22 (t, J=5.6 Hz, 1H ), 7.81-7.80 (m 1H), 7.68 (s, 2H), 4.35-4.22 (m, 2H), 4.16-4.15 (m, 2H), 3.88-3.86 (m, 2H), 2.94-2.86 (m, 4H), 1.70-1.69 (m, 4H).
The following compounds are prepared essentially by the method of Example 94.
Ex.
Chemical name Structure Physical data o.
3-(5-(3,5-dichlorophenyl MS (m/z): 558 (M+l); JH )-5-(trifluoromethyl)-4,5 NMR (CDCI3, 400 MHz) δ -dihydroisoxazol-3-yl)-N 7.49-7.42 (m, 3H), 6.85 (br,
95 -(2-oxo-2-(prop-2-ynyla lH), 6.72 (br, 1H), 4.17-4.03 mino)ethyl)-4,5,6,7-tetra (m, 5H), 3.68 (d, J = 17.2 Hz, hydrobenzo [c]thiophene- 1H), 3.00-2.90 (m, 4H), 2.25 (s, 1 -carboxamide 1H), 1.79 (br, 4H).
N-(2-(cyanomethylamin
o)-2-oxoethyl)-3-(5-(3,4,
MS (m/z): 599 (M+l); !H 5 -trichlorothiophen-2 -yl)
NMR (CDCI3, 400 MHz) δ -5-(trifluoromethyl)-4,5-
96 4.28-4.18 (m, 4H), 4.10-4.07 dihydroisoxazol-3-yl)-4, cr s
(m, 2H), 3.04-2.87 (m, 4H), 5,6,7-tetrahydrobenzo[c]
1.88-1.73 (m, 4H).
thiophene- 1 -carboxamid
e
Scheme D
Preparation 201 4-chlorobenzo[b]thiophene-2-carboxylic
Stir a mixture of methyl 4-chlorobenzo[b]thiophene-2-carboxylate (1.0 g, 4.44 mmol) and LiOH-H20 (0.56 g, 13.3 mmol) in MeOH (30 mL) and water (10 mL) at room temperature overnight. Concentrate the reaction mixture under vacuum and then dilute the residue with ice water (20 mL). Acidify the aqueous mixture with cone. HC1 solution to pH = 1. Extract the resultant mixture with EtOAc (15 mLx3). The combined organic layers are washed with brine, dried over anhydrous Na2SC>4 and evaporate under vacuum to afford
4-chlorobenzo[b]thiophene-2-carboxylic acid as a white solid (0.94 g, 100 %). !H NMR (400 MHz, CDC13) δ 8.10-8.02 (m, 2H), 7.61-7.51 (m, 2H)
The following compounds are prepared essentially by the method of Preparation
201.
Stir a mixture of 4-chlorobenzo[b]thiophene-2-carboxylic acid (0.94 g, 4.44 mmol),
Ν,Ο-dimethylhydroxylamine hydrochloride (0.86 g, 8.87 mmol), DCC (1.1 g, 5.32 mmol) and DIEA (1.43 g, 1.9 mL, 11.08 mmol) in DCM (8 mL) at ambient temperature for 2 hours. Filter the reaction mixture and the filtrate was washed with brine, dried over anhydrous Na2SC>4 and concentrated under vacuum. Purify the residue by column chromatography on silica gel eluting with PE: EtOAc (5: 1 to 3: 1) to afford
4-chloro-N-methoxy-N-methylbenzo[b]thiophene-2-carboxamide as white solid (0.85 g, 75.2 %). 'H NMR (400 MHz, CDC13) δ 8.10-8.02 (m, 2H), 7.61-7.51 (m, 2H), 3.84-3.79 (s, 3H), 3.37-3.35 (s, 3H).
boxamide / \
Add a solution of C¾MgBr (3 M in THF, 1.7 ml, 4.99 mmol) to a suspension of
4-chloro-N-methoxy-N-methylbenzo[b]thiophene-2-carboxamide (0.85 g, 3.33 mmol) in dry THF (10 mL) at 0 °C. Then stir the mixture for overnight at ambient temperature. Quench the reaction with saturated NH4CI aqueous solution (15 mL) and extract the aqueous mixture with EtOAc (10 mLx3). The combined organic layers are washed with brine, dried over anhydrous Na2S04 and concentrated under vacuum. Purify the residue by silica gel chromatograph (PE:EtOAc = 6: 1) to afford l-(4-chlorobenzo[b]thiophen-2-yl)ethanone as white solid (0.6 g, 86.9 %). !H NMR (400 MHz, CDCI3) δ 8.10-8.07 (s, 1H), 7.75 (d, J=5.2, 1H), 7.47-7.36 (m, 2H), 2.72-2.68 (s, 3H).
The following compounds are prepared essentially by the method of Preparation
207.
Chemical name Structure
No. data
1 -(5-Bromo-thieno [2 ,3 -b]p MS (m/z):
209
yridin-2-yl)-ethanone 258 (M+l).
l-Thieno[2,3-b]pyridin-2- MS (m/z):
210
yl-ethanone OH 178 (M+l)
Preparation 211 l-(4-chlorobenzo[b]thiophen-2- -3-(3,5-dichlorophenyl)-4,4,4-trifluoro-3-hydroxybutan-l-one
Add a solution of LiHMDS (1 M in THF, 4.3 ml, 4.31 mmol) to a mixture of
l-(4-chlorobenzo[b]thiophen-2-yl)ethanone (0.6 g, 2.87 mmol) in dry THF (10 mL) at -78 °C under N2. After stirring 1.5 hour at -78 °C, add l-(3,5-dichlorophenyl)-2,2,2-trifluoroethanone (836 mg, 3.44 mmol) to the reaction mixture and stir the resultant mixture for additional 2 hours. Quench the reaction with saturated NH4CI solution and extract the aqueous mixture with EtOAc (10 mLx3). The combined organic layers are washed with brine, dried over anhydrous Na2S04 and concentrated under vacuum afford crude
l-(4-chlorobenzo[b]thiophen-2-yl)-3-(3,5-dichlorophenyl)-4,4,4-trifluoro-3-hydroxybutan-l-one as brown solid (1.1 g, 84.6%). 'H NMR (400 MHz, CDCI3) δ 8.10-8.07 (s, 1H), 7.75 (d, J=5.2, 1H), 7.47-7.36 (m, 2H), 2.72-2.68 (s, 3H).
The following compounds are prepared essentially by the method of Preparation
Preparation 220 l-(4-chlorobenzo[b]thiophen-2-yl)-3-(3,5-dichlorophenyl)-4,4,4-trifluorobut-2-en-l-one
Stir a mixture of
l-(4-chlorobenzo[b]thiophen-2-yl)-3-(3,5-dichlorophenyl)-4,4,4-trifluoro-3-hydroxybutan-l-one (1.1 g, crude, 2.43 mmol), SOCl2 (1.16 g, 0.7 mL, 9.43 mmol) and pyridine (384 mg, 0.4 mL, 4.86 mmol) in anhydrous DCM (10 mL) at ambient temperature for overnight. Dilute the mixture with saturated NH4CI solution and extract the aqueous mixture with DCM (10 mLx3). The combined organic layers are washed with brine, dried over anhydrous a2S04 and concentrated under vacuum to afford crude
l-(4-chlorobenzo[b]thiophen-2-yl)-3-(3,5-dichlorophenyl)-4,4,4-trifluorobut-2-en-l-one as brown solid (1.05 g, 100%). 1H NMR (400 MHz, CDC13) 58.12-8.10 (s, 1H), 7.69-7.64 (m, 1H), 7.47-7.44 (m, 2H), 7.38-7.35 (m, 2H), 7.27-7.24 (m, 1H), 6.96 -6.92 (s, 1H).
The following compounds are prepared essentially by the method of Preparation
Chemical name Structure Physical data No.
1 -(5 -chlorobenzo [b]thio CF3 O
phen-2-yl)-3-(3,5-dichlo MS (m/z): 433
228
rophenyl)-4,4,4-trifluoro
CI
but-2-en-l-one
Example 229
3-(4-chlorobenzo[b]thiophen-2-yl)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxaz ole
Stir a mixture of
l-(4-chlorobenzo[b]thiophen-2-yl)-3-(3,5-dichlorophenyl)-4,4,4-trifluorobut-2-en-l-one (1.05 g, crude, 2.43 mmol), NaOH (389 mg, 9.72 mmol) andNH2OH-HCl(335 mg, 4.8 mmol) in MeOH (8 mL) and water (8 mL) at ambient temperature for 4 hours. After removal of solvent under vacuum, dilute the residue with ice water (20 mL). Extract the aqueous mixture with EtOAc (15 mLx3). The combined organic layers are washed with brine, dried over anhydrous a2S04 and concentrated under vacuum. Purify the residue by preparative HPLC to afford
3-(4-chlorobenzo[b]thiophen-2-yl)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxaz ole as white solid (305 mg, 28.1 %). MS (m z): 450 (M+l). 'HNMR (400 MHz, CDC13) δδ 7.74 (d, J=8.0, 1H), 7.64-7.60 (s, 1H), 7.57-7.53 (m, 2H), 7.47-7.43 (m, 1H), 7.40-7.32.
Chemical name Structure Physical data
No.
3-(benzo[d]thiazol- MS (m/z): 417 (M+1). IH NMR 2-yl)-5-(3,5-dichlor (400 MHz, CDC13) δ 8.07 (d,
234 ophenyl)-5 -(trifluo 7.2, IH), 7.93 (d, J=7.2, IH), romethyl)-4,5-dihy CF3 7.58-7.47 (m, 5H), 4.37 (d,
CI ά
droisoxazole J=18.0, IH), 4.00 (d, J=18.0, IH)
5 -(3 , 5 -dichlorophe MS (m/z): 430 (M+1). IH NMR nyl)-3-(3-methylbe (400 MHz, CDCI3) δ 7.83-7.78 nzo [b] thiophen-2 -y (m, 2H), 7.53 (s, 2H), 7.53-7.43
235
1) - 5 - (tri fluor ometh (m, 3H), 4.17 (d, 16.8, IH),
CF3
yl)-4,5-dihydroisox CI 3 3.79 (d, 16.8, IH), 2.67 (s, azole 3H).
MS (m/z): 430 (M+1). IH NMR
5 -(3 , 5 -dichlorophe
(400 MHz, CDCI3) δ 7.71 (d, nyl)-3-(5-methylbe
8.0, IH), 7.57 (s, IH), 7.53 (s, nzo [b] thiophen-2 -y
236 2H), 7.43 (t, 3.2, IH), 7.36 (d, 1) - 5 - (tri fluor ometh
4.17 (d, 16.8, IH), 3.78 (d, azole
16.8, IH), 2.47 (s, 3H).
3-(5-chlorobenzo[b MS (m/z): 450 (M+1). IH NMR ]thiophen-2-yl)-5-( (400 MHz, CDCI3) δ 7.76-7.75 3 ,5-dichlorophenyl (m, 2H), 7.51 (s, 2H), 7.45-7.44
237
) - 5 - (tri fluor omethy (m, IH), 7.40-7.38 (m, 2H), 4.18
CF3
CI 6
l)-4,5-dihydroisoxa (d, J= 16.8, lH), 3.78 (d, J= 17.6, zole IH).
Scheme E
Example 238 methyl
2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)thieno[2,3-b]pyridine-5- carboxylate
Stir a mixture of
5-bromo-2-[5-(3,5-dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3- '/]-thieno[2,3-b]p yridine (494 mg, 1 mmol), Pd(dppf)Ci2 (100 mg) and triethyl amine (1 mL) in anhydrous THF (10 mL) and methanol (5 mL) under carbon monoxide (50 psi) at 70 °C for lOh. After removal of solvent under vacuum, purify the residue with silica gel chromatography (eluting with 10% ethyl acetate in petroleum ether) to afford methyl
2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)thieno[2,3-b]pyridine-5- carboxylate as a white solid (210 mg, 44.18%). MS (m/z): 475 (M+l).
Example 239
2-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-j/]-thieno[2,3-b]pyri
-carboxylic acid
Add a solution of LiOH-H20 (76 mg, 2 mmol) in water (0.5 mL) to a solution of methyl 2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)thieno[2,3-b]pyridine-5- carboxylate (237 mg, 0.5 mmol) in THF(3 mL). Stir the mixture at temperature for 12 hours. After addition of 10 mL of water, acidify the mixture with concentrated HC1 to PH = 6~7. Extract the resultant mixture with ethyl acetate (3 X 10 mL). The combined organic layers are washed brine, dried over anhydrous Na2SC>4 and concentrated under vacuum to afford
2-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3- '/]-thieno[2,3-b]pyridine-5 -carboxylic acid as pale yellow solid (170 mg, 73.9%), which is used in next step without further purification. MS (m/z): 461 (M+l).
Example 240
2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo-2-(2,2,2-triflu oroethylamino)ethyl)thieno[2,3-b]pyridine-5-carboxamide
Stir a mixture of 2-Amino-N-(2,2,2-trifluoro-ethyl)-acetamide (156 mg, 1 mmol),
2-[5-(3,5-dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-thieno[2,3-b]pyridine-5- carboxylic acid (160 mg, 0.24 mmol), HATU (150 mg, 0.39 mmol) and DIPEA (0.2 mL) at room temperature for for 10 hours. After removal of solvent, purify the mixture by preparetive-HPLC to
afford
2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5^
oroethylamino)ethyl)thieno[2,3-b]pyridine-5-carboxamide as a white solid (50 mg, 34.78%). MS (m/z): 599 (M+l). 1H NMR (400 MHz, CDC13) δ 9.02 (s, 1H), 8.51 (s, 1H), 7.52 (s, 2H), 7.44-7.41 (m, 2H), 7.22 (s, 1H), 6.64 (s, 1H), 4.27 (d, J= 5.2, 2H), 4.17 (d, J = 16.8, 1H), 4.04-3.95 (m, 2H), 3.80 (d, J = 16.08, 1H).
The following compound is prepared essentially by the method of Example 240.
Ex.
Chemical name Structure Physical data
No.
MS (m/z): 543 (M+l). LH NMR
2-(5-(3,5-dichlorophe
(400 MHz, CDCI3) δ 9.01 (s, 1H), nyl)-5 -(trifluorometh
8.49 (s, 1H), 7.54 (s, 2H), yl)-4,5 -dihydroisoxaz VNH
7.44-7.40 (m, 2H), 7.15 (S, 1H),
241 ol_3_yl)_N-((R)-2-ox
5.89 (s, 1H), 4.60 (s, lH), 4.18 (d, opyrrolidin-3 -yl)thie
J = 16.8, 1H), 3.80 (d, J = 16.8, no[2,3-b]pyridine-5-c
1H), 3.51-3.48 (m, 2H), 3.00-2.93 arboxamide
(m, 1H), 2.14-2.08 (m, 1H)
In Vitro Helminth Assay
Compounds may be evaluated against one or more life stages of helminth to measure anthelmintic activity. Compounds may be evaluated at a single concentration followed by serial dilution in order to determine minimal effective concentration. Typically, worms are exposed to compounds in a liquid solution for a predetermined period of time. Activity is measured through one or more variables, which may include an effect on worm motility {e.g., moving versus non-moving) or viability (e.g., live versus dead).
In Vivo Activity Against Nematodes Compounds may be evaluated against one or more life stages of helminth infestation in an animal to measure in vivo anthelmintic activity. Compounds may be evaluated a single dose, administered on a milligram per kilogram body weight basis, followed by dose titration in order to determine minimal effective point dose. In a rodent anthelmintic model, for example, adult Mongolian gerbils (Meriones unguiculates) infected with one or more species of Strongylid nematode (e.g., Haemonchus contortus and/or Trichostrongylus colubriformis) are dosed with compounds, administered via oral gavage. Gerbils are necropsied and gastrointestinal tract worm burden is measured and compared to untreated, infected control gerbils to determine the degree of anthelmintic activity. Similar testing may be conducted in higher species (e.g., dogs, cats, sheep, cattle) whereby nematode burden in treated animals is compared to burden in untreated, infected animals to measure the potency and duration of anthelmintic activity.
In Vitro Larval Immersion Microassay (LIM)
The larval immersion microassay may be conducted as described in White, et ah, J. Med. Entomol. 41 : 1034-1042 (2004). Briefly, experimental test articles are formulated in dimethylsulfoxide (DMSO) to prepare a stock solution at a concentration of lOmM. Using
96-well micro titer plates, an aliquot of the lOmM sample is subsequently diluted in a water-based solution containing 1% ethanol and 0.2% Triton X-100, to obtain the desired concentration (typically 0.3mM or lower) of experimental test article in a volume of 0.1ml (minimum n=3
replicates per compound or concentration). Approximately 30-50 Lone star tick larvae
(Ambfyomma americanum) are submerged into each well containing experimental test articles. After a 30 minute immersion period, larvae are removed with a wide -bore pipette tip in 0.05ml of fluid, dispensed into a commercial paper tissue biopsy bag which is sealed at the top with a plastic dialysis clip, inverted and allowed to air dry for 60 minutes. Bags containing larvae are then incubated at approximately 27 degrees Celsius and >90% relative humidity. After 24 hours, bags are opened, live and dead larvae are counted and percent larval mortality is calculated.
The following Examples exhibited > 80% activity when tested in this assay at 300 micromolar: 32, 36, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51 , 52, 53, 54, 55, 56, 57, 58, 59, 60, 61 , 62, 63, 64, 65, 66, 67, 68, 69, 70, 71 , 72, 73, 74, 75, 76, 77, 78, 79, 80, 81 , 82, 83, 84, 85, 86, 87b, 94, 95, 96, 230, 231, 232, 233, 236, 237, 240, and 241.
In Vivo Rodent Acaricide Test (RAT)
Evaluation of experimental test articles may conducted using a modified version of the assay as described in Gutierrez et ah, J. Med. Entomol. 43(3): 526-532(2006). The assay may be modified by using a different tick species (the reference describes Ambfyomma americanum ticks) such as Dermacentor variabilis ticks. Further, the reference describes using topical administration, but oral administration may be used. Briefly, experimental test articles are formulated in a solution of polyethylene glycol-300, propylene glycol and water to the desired concentration, typically 1 - 25 mg/ml, depending on solubility and desired point dose. Tick containment units (comprised of a baby nipple, ventilated screw cap top and reinforcing rubber washer) are attached to the dorsum of adult Sprague-Dawley rats. After attachment of containment units, approximately 10 unfed nymphal stage American dog ticks (Dermacentor variabilis) are placed inside of each containment unit. Approximately 24 hours after infestation, test article formulations are administered to rats via oral gavage. Negative control rats receive polyethylene glycol-300, propylene glycol and water alone. Depending on compound availability, a minimum of three (3) and a maximum of five (5) rats are utilized per treatment group. Forty-eight (48) hours post-treatment, containment units are removed and live and dead ticks were counted. Live tick counts are transformed using the natural logarithm
transformation plus one (Ln count + 1); addition of one to each count serve to adjust for counts
that were zero. Geometric mean (GM) group tick counts are obtained via back-transformation of group mean transformed counts and subtracting one. The non-treated control group is used for comparison to the groups receiving experimental test articles for the calculation of percent efficacy (% reduction in live tick counts). The efficacy of treatments is calculated by comparing the geometric mean (GM) number of live ticks observed on treated rats with the GM number of live ticks counted on the negative control rats, using the following formula:
% Efficacy = (GM # live ticks control - GM # live ticks treated) x 100
GM # live ticks control
The following Examples exhibited > 50% efficacy when tested in this assay at a dose of not more than 25 mpk: 32, 36, 38, 39, 41, 42, 43, 45, 46, 47, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 61, 73, 87b, 95, 96, 234, 235, 240, and 241.
Activity in the above assays demonstrates the compounds of the invention are useful for controlling ecto- or endoparasite infestations.
Claims
A compound, or a salt thereof, of formula I
wherein A is
R1 is thienyl or phenyl, said thienyl or phenyl substituted with 2 or 3 of the same or different halo atoms;
R2 is at each occurrence independently hydrogen, C1-C5 alkyl, C3-C6 cycloalkyl, or C1-C5 haloalkyl;
3 · - -(CH2)p-R4
RJ is p is at each occurrence independently 0 or 1 ;
R4 is C1-C5 alkyl, C1-C5 haloalkyl, C1-C5 cyanoalkyl, C1-C5 alkylthio, C3-C6 cycloalkyl optionally substituted with hydroxy, halo, or C1-C5 alkyl: C3-C5 cyclohetero alkyl optionally substituted with C1-C5 alkyl, C3-C6 cycloalkyl, or C1-C5 haloalkyl: phenyl, thienyl, R5
,(CH2)"
pyridinyl, or O , wherein one of the carbons in said cycloalkyls, independently, or cycloheteroalkyl may form a carbonyl group, and wherein said phenyl, thienyl, or pyridinyl is optionally substituted with halo or a carbamoyl group;
R6 is hydrogen, Ci-C5 alkyl, Ci-C5 haloalkyl, Ci-C5 cyanoalkyl, Ci-C5 alkylthio, or C2-C5 alkynyl;
2 3 ·
or R and R combine to form, with the nitrogen to which they are attached,
„ /
o
Yi, Y2, and Y3 are carbon or nitrogen with at most only one of Yi, Y2, and Y3 being nitrogen, and when Yi, Y2, or Y3 is a carbon, each may be substituted by C1-C5 alkyl;
R7 is hydrogen, halo, C1-C5 alkyl, or
¾(CH2)^ R8
O ;
R9 is C1-C5 alkyl,
2. A compound of claim 1 wherein A is
3. A compound according to any of claims 1-2 wherein 2
R is hydrogen and n is 1.
4. A compound of according to any of claims 1-3 wherein R3 is
5. The compound of claim 1 wherein it is
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2-o xopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N,N-Dimethyl-2-(4-{3-[5-(3,4,5-trichloro-thiophen-2-yl)-5-trifluoromethyl-4,5-dih ydro-isoxazol-3-yl]-4,5,6,7-tetrahydro-benzo[c]thiophene-l-carbonyl}-piperazin-l-yl)-acetamide;
({3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7 -tetrahydro-benzo[c]thiophene-l-carbonyl}-amino)-acetic acid methyl ester;
({3-[5-(3,4,5-Trichloro-thiophen-2-yl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-y l]-4,5,6,7-tetrahydro-benzo[c]thiophene-l-carbonyl}-amino)-acetic acid methyl ester;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo -2-(2,2,2-trifluoroethylamino)ethyl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7-t etrahydro-benzo[c]thiophene-l -carboxylic acid (l,l-dioxo-thietan-3-yl)-amide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(3-oxo cyclohexyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7-t etrahydro-benzo [c]thiophene- 1 -carboxylic acid (1,1 -dioxo-hexahydro- 116-thiopyran-4-yl)-amide;
3-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6,7-t etrahydro-benzo [c]thiophene- 1 -carboxylic acid [2-oxo-l-(2,2,2-trifluoro-ethyl)-pyrrolidin- 3-yl]-amide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2 -oxopyrrolidin-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,5-dichlorophenyl)-5-(trifluoromet hyl)-4,5-dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo -2-(prop-2-ynylamino)ethyl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-(2-oxo-2-(2,2,2-trifluoroethylamino)ethyl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifl uoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-((R)-2-oxopyrrolidin-3-yl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluoromethyl)-4,5 -dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide; N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluorom ethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(2-oxo-2-(prop-2-ynylamino)ethyl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluoromet hyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(2-oxo-2-(2,2,2-trifluoroethylamino)ethyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamid e;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-((R)-2-oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifl uoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(2-oxo-2-(prop-2-ynylamino)ethyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(2-oxo-2-(2,2,2-trifluoroethylamino)ethyl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifl uoromethyl)-4,5-dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-((R)-2-oxopyrrolidin-3-yl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluoromethyl)-4,5 -dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluorom ethyl)-4,5-dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-(2-oxo-2-(prop-2-ynylamino)ethyl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluoromet hyl)-4,5-dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-(2-oxo-l-(2,2,2-trifluoroethyl)pyrrolidin-3-yl)-3-(5-(3,4,5-trichlorophenyl)-5-(t rifluoromethyl)-4,5-dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamid e;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(2-oxo-2-(2,2,2-trifluoroethylamino)ethyl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxa mide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-((R)-2-oxopyrrolidin-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifl uoromethyl)-4,5-dihydroisoxazol-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide; 3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(2-oxo-l-(2,2,2-trifluoroethyl)pyrrolidin-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carb oxamide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(2-oxo-2-(prop-2-ynylamino)ethyl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo -2-(2,2,2-trifluoroethylamino)ethyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-(eth ylamino)-2-oxoethyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((tetra hydrofuran-2-yl)methyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-(me thylthio)ethyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(3,3,3- trifluoropropyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(3-hyd roxycyclohexyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(5-flu oropyridin-2-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(tetrah ydro-2H-pyran-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(thieta n-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(tetrah ydro-2H-pyran-4-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(l-cyclopropyl-2-oxopyrrolidin-3-yl)-3-(5-(3,5-dichlorophenyl)-5-(trifluoromet hyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(2-oxo-2-(prop-2-ynylamino)ethyl)-3-(5-(3,4,5-trichlorothiophen-2-yl)-5-(triflu oromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-[5-(3,4,5-Trichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-4,5,6, 7-tetrahydro-benzo[c]thiophene- 1 -carboxylic acid (1,1 -dioxo-hexahydro-thiopyran-4-yl)-amide; N-(2-oxo-l-(2,2,2-trifluoroethyl)pyrrolidin-3-yl)-3-(5-(3,4,5-trichlorophenyl)-5-(t rifluoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(tetrahydro-2H-pyran-4-yl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluoromethyl)-4,5 -dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(l-cyclopropyl-2-oxopyrrolidin-3-yl)-3-(5-(3,4,5-trichlorophenyl)-5-(trifluoro methyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-[5-(3,5-Dichloro-4-fluoro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl] -4,5,6,7-tetrahydro-benzo[c]thiophene-l-carboxylic acid (l,l-dioxo-thietan-3-yl)-amide;
3-[5-(3,5-Dichloro-4-fluoro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl] -4,5,6,7-tetrahydro-benzo[c]thiophene-l-carboxylic acid
( 1 , 1 -dioxo-hexahydro-thiopyran-4-yl)-amide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(4-oxocyclohexyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(2-oxo-l-(2,2,2-trifluoroethyl)pyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxa mide;
3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- N-(tetrahydro-2H-pyran-4-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(l-cyclopropyl-2-oxopyrrolidin-3-yl)-3-(5-(3,5-dichloro-4-fluorophenyl)-5-(trif luoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(4-oxo cyclohexyl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo -l-(2,2,2 rifluoroethyl)pyrrolidin-3-yl)-5,6-dihydro-4H-cyclopenta[c]thiophene-l-carboxamide;
3-((R)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(( R)-2-oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3-((S)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(( R)-2-oxopyrrolidin-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
N-(4-carbamoylphenyl)-3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5- dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide; 3-[5-(3,4,5-Trichloro-thiophen-2-yl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl] -4,5,6,7-tetrahydro-benzo[c]thiophene-l-carboxylic acid (3-carbamoyl-thiophen-2-yl)-amide;
2- (3-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)- 4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamido)acetic acid;
({3-[5-(3,4,5-Trichloro-thiophen-2-yl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-y l]-4,5,6,7-tetrahydro-benzo[c]thiophene-l-carbonyl}-amino)-acetic acid;
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,5-dichlorophenyl)-5- (trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide;
3- (5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo -2-(prop-2-ynylamino)ethyl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide; or
N-(2-(cyanomethylamino)-2-oxoethyl)-3-(5-(3,4,5-trichlorothiophen-2-yl)-5-(trifl uoromethyl)-4,5-dihydroisoxazol-3-yl)-4,5,6,7-tetrahydrobenzo[c]thiophene-l-carboxamide; or a salt thereof.
6. A compound of claim 1 wherein A is
7. A compound of claim 6 wherein Yi is nitrogen.
8. A compound of claim 6 wherein R7 is
9. The compound of claim 1 wherein it is
3-(4-chlorobenzo[b]thiophen-2-yl)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5 -dihydroisoxazole;
2-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-thieno[2
,3-c]pyridine;
5-Bromo-2-[5-(3,5-dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl] -thieno[2,3-b]pyridine;
2- [5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-thieno[2
,3-b]pyridine;
3- (benzo[b]thiophen-2-yl)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydro isoxazole;
3-(benzo[d]thiazol-2-yl)-5-(3,5-dichlorophenyl)-5-(trifiuoromethyl)-4,5-dihydrois oxazole;
5-(3,5-dichlorophenyl)-3-(3-methylbenzo[b]thiophen-2-yl)-5-(trifluoromethyl)-4, 5-dihydroisoxazole;
5-(3,5-dichlorophenyl)-3-(5-methylbenzo[b]thiophen-2-yl)-5-(trifluoromethyl)-4, 5-dihydroisoxazole; 3-(5-chlorobenzo[b]thiophen-2-yl)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5 -dihydroisoxazole;
methyl
2- (5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)thieno[2,3-b]pyridine-5- carboxylate;
2-[5-(3,5-Dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3- '/]-thieno[2,
3- b]pyridine-5-carboxylic acid;
2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-(2-oxo -2-(2,2,2-trifluoroethylamino)ethyl)thieno[2,3-b]pyridine-5-carboxamide; or
2-(5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-yl)-N-((R)-2 -oxopyrrolidin-3-yl)thieno[2,3-b]pyridine-5-carboxamide; or a salt thereof.
10. A formulation comprising a compound or salt of any of claims 1-9 and one or more acceptable carriers.
11. The formulation of claim 10 wherein it further comprises at least one additional active ingredient.
12. The formulation of claim 10 or claim 11 wherein it is a human pharmaceutical formulation.
13. The formulation of claim 10 or claim 1 1 wherein it is a veterinary pharmaceutical formulation.
14. A method of controlling a parasite infestation in or on an animal in need thereof comprising administering an effective amount of a compound or salt of any of claims 1-9 to said animal.
15. The method of claim 14 wherein at least one other active ingredient is administered to said animal.
16. The method of claim 14 or claim 15 wherein said animal is a human.
The method of claim 14 or claim 15 wherein said animal is a companion animal.
18. The method of claim 16 or claim 17 wherein said companion animal is a dog or cat.
19. The method of claim 18 wherein said parasite is a tick.
20. The method of claim 14 wherein said animal is a livestock animal.
21. A method for preventing and treating diseases transmitted through parasites comprising administering an effective amount of a compound of any of claims 1-9 to an animal in need thereof.
22. The method of claim 21 or claim 22 wherein at least one other active ingredient is administered to said animal.
23. The method of claim 21 or claim 22 wherein said animal is a human.
24. The method of claim 21 wherein said animal is a companion animal.
25. The method of claim 24 wherein said companion animal is a dog or cat.
26. The method according to any of claims 21-25 wherein said parasite is a tick.
27. The method of claim 21 or claim 22 wherein said animal is a livestock animal.
28. A method for controlling parasites, characterized in that a compound of any of claims 1 -9 is allowed to act on the pests or their habitat, or both.
29. The method of claim 28 wherein the compound is placed on a plant or an animal.
30. Use of compounds or salts thereof of any of claims 1-9 for controlling parasites.
31. A compound or salt according to any of claims 1 -9 for use in therapy.
32. A compound or salt according to any of claims 1-9 for use in controlling an ectoparasite infestation.
Priority Applications (37)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2011/074294 WO2012155352A1 (en) | 2011-05-19 | 2011-05-19 | Dihydroisoxazole compounds, parasiticidal uses and formulations thereof |
| PCT/CN2012/072878 WO2012155676A1 (en) | 2011-05-19 | 2012-03-23 | Parasiticidal compounds, methods, and formulations |
| ARP120101602A AR086296A1 (en) | 2011-05-19 | 2012-05-07 | USED DIHYDROISOXASOL DERIVATIVES AS PARASITICIDES BOTH IN ANIMALS AND IN PLANTS |
| CN201280024353.4A CN103547576B (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydro isoxazole compound |
| PE2013002523A PE20141041A1 (en) | 2011-05-19 | 2012-05-08 | COMPOUNDS, METHODS AND PARASITICIDE FORMULATIONS |
| AU2012256220A AU2012256220B2 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| MEP-2015-73A ME02113B (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| JP2014511394A JP5989099B2 (en) | 2011-05-19 | 2012-05-08 | Dihydroisoxazole compounds for parasite control |
| EP15160519.3A EP2918583A1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| HRP20150588TT HRP20150588T1 (en) | 2011-05-19 | 2012-05-08 | PARASITOCIDE COMPOUNDS OF DIHYDROISOXAZOLE |
| RS20150379A RS54048B1 (en) | 2011-05-19 | 2012-05-08 | PARASITICIDE COMPOUNDS OF DIHYDROISOXAZOLE |
| KR1020137030230A KR101586553B1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| EA201391542A EA023711B1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal compounds, methods and formulations |
| SG2013082615A SG194836A1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| PT127211464T PT2710000E (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| TW101116393A TWI554508B (en) | 2011-05-19 | 2012-05-08 | Parasitic compounds, methods and formulations |
| PCT/US2012/036883 WO2012158396A1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| US13/883,025 US8648081B2 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| HK14104609.9A HK1191337B (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| EP20120721146 EP2710000B1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| ES12721146.4T ES2543392T3 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| BR112013028132-4A BR112013028132B1 (en) | 2011-05-19 | 2012-05-08 | dihydroxysoxazole compounds, its intermediate and its preparation process, and formulation |
| CA2835436A CA2835436C (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| PL12721146T PL2710000T3 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| PH1/2013/502374A PH12013502374A1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| SI201230196T SI2710000T1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| NZ616409A NZ616409B2 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
| MX2013013446A MX341739B (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds. |
| DK12721146.4T DK2710000T3 (en) | 2011-05-19 | 2012-05-08 | PARASITICIDE DIHYDROISOXAZOLE COMPOUNDS |
| UAA201313451A UA110519C2 (en) | 2011-05-19 | 2012-05-08 | PARASITOCIDE DIGIDROISOXAZOLE COMPOUNDS |
| CR20130486A CR20130486A (en) | 2011-05-19 | 2013-09-26 | PARASITICIDE COMPOUNDS OF DIHYDROISOXAZOL |
| IL229007A IL229007A (en) | 2011-05-19 | 2013-10-21 | Dihydroisoxazole compounds are parasitic |
| CL2013003287A CL2013003287A1 (en) | 2011-05-19 | 2013-11-15 | Compounds derived from 5-trifluoromethyl-4,5-dihydro-isoxazole; intermediate compounds; preparation procedure; pharmaceutical compositions comprising them; and use in the treatment of a parasite infestation in animals. |
| CO13271916A CO6811873A2 (en) | 2011-05-19 | 2013-11-19 | Parasiticidal Dihydroisoxazole Compound |
| US14/137,279 US8889710B2 (en) | 2011-05-19 | 2013-12-20 | Parasiticidal dihydroisoxazole compounds |
| US14/546,590 US9255111B2 (en) | 2011-05-19 | 2014-11-18 | Parasiticidal dihydroisoxazole compounds |
| CY20151100494T CY1116450T1 (en) | 2011-05-19 | 2015-06-04 | DIAHRODOXAZOLI COMPOUNDS |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2011/074294 WO2012155352A1 (en) | 2011-05-19 | 2011-05-19 | Dihydroisoxazole compounds, parasiticidal uses and formulations thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012155352A1 true WO2012155352A1 (en) | 2012-11-22 |
Family
ID=47176150
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2011/074294 Ceased WO2012155352A1 (en) | 2011-05-19 | 2011-05-19 | Dihydroisoxazole compounds, parasiticidal uses and formulations thereof |
| PCT/CN2012/072878 Ceased WO2012155676A1 (en) | 2011-05-19 | 2012-03-23 | Parasiticidal compounds, methods, and formulations |
| PCT/US2012/036883 Ceased WO2012158396A1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2012/072878 Ceased WO2012155676A1 (en) | 2011-05-19 | 2012-03-23 | Parasiticidal compounds, methods, and formulations |
| PCT/US2012/036883 Ceased WO2012158396A1 (en) | 2011-05-19 | 2012-05-08 | Parasiticidal dihydroisoxazole compounds |
Country Status (30)
| Country | Link |
|---|---|
| US (3) | US8648081B2 (en) |
| EP (2) | EP2918583A1 (en) |
| JP (1) | JP5989099B2 (en) |
| KR (1) | KR101586553B1 (en) |
| CN (1) | CN103547576B (en) |
| AR (1) | AR086296A1 (en) |
| AU (1) | AU2012256220B2 (en) |
| BR (1) | BR112013028132B1 (en) |
| CA (1) | CA2835436C (en) |
| CL (1) | CL2013003287A1 (en) |
| CO (1) | CO6811873A2 (en) |
| CR (1) | CR20130486A (en) |
| CY (1) | CY1116450T1 (en) |
| DK (1) | DK2710000T3 (en) |
| EA (1) | EA023711B1 (en) |
| ES (1) | ES2543392T3 (en) |
| HR (1) | HRP20150588T1 (en) |
| IL (1) | IL229007A (en) |
| ME (1) | ME02113B (en) |
| MX (1) | MX341739B (en) |
| PE (1) | PE20141041A1 (en) |
| PH (1) | PH12013502374A1 (en) |
| PL (1) | PL2710000T3 (en) |
| PT (1) | PT2710000E (en) |
| RS (1) | RS54048B1 (en) |
| SG (1) | SG194836A1 (en) |
| SI (1) | SI2710000T1 (en) |
| TW (1) | TWI554508B (en) |
| UA (1) | UA110519C2 (en) |
| WO (3) | WO2012155352A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018166855A1 (en) * | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2019122324A1 (en) | 2017-12-21 | 2019-06-27 | Intervet International B.V. | Antiparasitic pour-on compositions |
| CN110167540A (en) * | 2016-10-31 | 2019-08-23 | 斯克利普斯研究所 | For preventing the method and composition of the propagation of vector-borne disease |
| WO2020088949A1 (en) * | 2018-10-29 | 2020-05-07 | Basf Se | Process for preparation of optically enriched aldol compounds |
| US11179372B2 (en) | 2015-06-23 | 2021-11-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| WO2022256284A1 (en) * | 2021-06-02 | 2022-12-08 | Fmc Corporation | Fused pyridines for controlling invertebrate pests |
Families Citing this family (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| UY33403A (en) * | 2010-06-17 | 2011-12-30 | Novartis Ag | ORGANIC COMPOUNDS WITH NEW ISOXAZOLINES, THEIR N-OXIDES, S-OXIDES AND SALTS |
| WO2012155352A1 (en) | 2011-05-19 | 2012-11-22 | Eli Lilly And Company | Dihydroisoxazole compounds, parasiticidal uses and formulations thereof |
| HUE040143T2 (en) * | 2012-12-11 | 2019-02-28 | Bracco Imaging Spa | Continuous process for the preparation of (s)-2-acetyloxypropionic acid |
| ES2656012T3 (en) | 2013-02-06 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted pyrazole derivatives as pesticides |
| AU2014375265B2 (en) | 2014-01-03 | 2018-11-01 | Elanco Animal Health Gmbh | Novel pyrazolyl-heteroarylamides as pesticides |
| TW201623265A (en) | 2014-05-09 | 2016-07-01 | 奇尼塔公司 | Anti-viral compounds, pharmaceutical compositions, and methods of use thereof |
| EP3154928B1 (en) * | 2014-06-10 | 2020-03-04 | Bracco Imaging S.p.A | Method for the preparation of (s)-2-acetyloxypropionic acid and derivatives thereof |
| MX2017000673A (en) | 2014-07-15 | 2017-04-27 | Bayer Animal Health Gmbh | Aryl-triazolyl pyridines as pest control agents. |
| CN119033777A (en) * | 2016-05-10 | 2024-11-29 | 瑞士伊兰科动物保健有限公司 | Dihydroisoxazole compounds for controlling sea lice |
| CN110234645A (en) | 2017-01-25 | 2019-09-13 | 巴斯夫欧洲公司 | The method for preparing Bian type amide |
| WO2021126281A1 (en) | 2019-12-20 | 2021-06-24 | Nammi Therapeutics, Inc. | Formulated and/or co-formulated liposome compositions containing toll-like receptor ("tlr") agonist prodrugs useful in the treatment of cancer and methods thereof |
| JP2024513690A (en) * | 2021-03-11 | 2024-03-27 | イン ザ ボウル アニマル ヘルス,インコーポレーテッド | Methods for controlling flea and tick infestations in mammals - Patents.com |
| JP2024511956A (en) * | 2021-03-11 | 2024-03-18 | イン ザ ボウル アニマル ヘルス,インコーポレーテッド | Oral canine feed and methods of controlling tick infestations in canids |
| WO2022192623A1 (en) * | 2021-03-11 | 2022-09-15 | In The Bowl Animal Health, Inc. | Oral canine feed and methods for controlling flea infestations in a canine |
| CN117769356A (en) * | 2021-06-25 | 2024-03-26 | 碗中动物健康公司 | Oral feed for felines and method for controlling tick infestations in felines |
| CN117915782A (en) * | 2021-06-25 | 2024-04-19 | 碗中动物健康公司 | Oral feed for felines and method for controlling flea infestations in felines |
| CN114085159B (en) * | 2021-11-25 | 2023-04-07 | 浙江大学 | Method for preparing 3-aminocyclohexanone by reductive amination of 1, 3-cyclohexanedione |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
| WO2026037853A1 (en) | 2024-08-14 | 2026-02-19 | Basf Se | Benzoxazole derivatives as pesticidal compounds |
| WO2026041702A1 (en) | 2024-08-21 | 2026-02-26 | Basf Se | Benzoxazole derivatives as pesticidal compounds |
| CN120192309A (en) * | 2025-04-18 | 2025-06-24 | 南开大学 | Isoxazoline derivatives containing bisamides and preparation methods and applications thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008019760A1 (en) * | 2006-08-15 | 2008-02-21 | Bayer Cropscience Ag | Insecticidal isoxazolines |
| WO2008154528A2 (en) * | 2007-06-13 | 2008-12-18 | E. I. Du Pont De Nemours And Company | Isoxazoline insecticides |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101768129B (en) * | 2004-03-05 | 2012-05-23 | 日产化学工业株式会社 | Preparation intermediate of isoxazoline substituted benzamide compound |
| HUP0501170A2 (en) * | 2005-12-20 | 2007-09-28 | Richter Gedeon Nyrt | 2-heteroaryl-3-phenyl-thieno[2,3-b]pyridines, process for their preparation, their use and pharmaceutical compositions containing them |
| TWI412322B (en) * | 2005-12-30 | 2013-10-21 | Du Pont | Isoxazolines for controlling invertebrate pests |
| MY146638A (en) | 2007-04-10 | 2012-09-14 | Bayer Cropscience Ag | Insecticidal aryl isoxazoline derivatives |
| TWI430995B (en) | 2007-06-26 | 2014-03-21 | Du Pont | Naphthalene isoxazoline invertebrate pest control agents |
| TWI556741B (en) | 2007-08-17 | 2016-11-11 | 英特威特國際股份有限公司 | Isoxazoline compositions and their use as antiparasitics |
| CN102088856B (en) | 2008-07-09 | 2015-11-25 | 巴斯夫欧洲公司 | Comprise the insecticidal activity mixture I of different * isoxazoline compound |
| JP2012501989A (en) * | 2008-09-04 | 2012-01-26 | シンジェンタ パーティシペーションズ アクチェンゲゼルシャフト | Insecticidal compound |
| US8377942B2 (en) * | 2008-12-18 | 2013-02-19 | Novartis Ag | Isoxazolines derivatives and their use as pesticide |
| HUE015568T4 (en) | 2008-12-19 | 2017-10-30 | Elanco Tiergesundheit Ag | Isoxazoline derivatives and their use as pesticide |
| JP2011098956A (en) * | 2009-10-09 | 2011-05-19 | Sumitomo Chemical Co Ltd | Iso-oxazoline compound and application thereof |
| WO2011073098A1 (en) * | 2009-12-15 | 2011-06-23 | Bayer Cropscience Ag | 1-(heteroaryl)-pyrazol-4-yl-acetic acids, method for the production thereof, and the use thereof as herbicides and plant growth regulators |
| CN103270036B (en) | 2010-08-05 | 2015-11-25 | 佐蒂斯有限责任公司 | As antiparasitic isoxazoline derivative |
| BR112013006686A2 (en) * | 2010-09-24 | 2017-11-14 | Zoetis Llc | isoxazoline oximes as antiparasitic agents |
| JP2014505089A (en) * | 2011-02-10 | 2014-02-27 | ノバルティス アーゲー | Isoxazoline derivatives for controlling invertebrate pests |
| WO2012155352A1 (en) | 2011-05-19 | 2012-11-22 | Eli Lilly And Company | Dihydroisoxazole compounds, parasiticidal uses and formulations thereof |
-
2011
- 2011-05-19 WO PCT/CN2011/074294 patent/WO2012155352A1/en not_active Ceased
-
2012
- 2012-03-23 WO PCT/CN2012/072878 patent/WO2012155676A1/en not_active Ceased
- 2012-05-07 AR ARP120101602A patent/AR086296A1/en active IP Right Grant
- 2012-05-08 WO PCT/US2012/036883 patent/WO2012158396A1/en not_active Ceased
- 2012-05-08 PH PH1/2013/502374A patent/PH12013502374A1/en unknown
- 2012-05-08 CN CN201280024353.4A patent/CN103547576B/en active Active
- 2012-05-08 PT PT127211464T patent/PT2710000E/en unknown
- 2012-05-08 EP EP15160519.3A patent/EP2918583A1/en not_active Withdrawn
- 2012-05-08 CA CA2835436A patent/CA2835436C/en active Active
- 2012-05-08 ME MEP-2015-73A patent/ME02113B/en unknown
- 2012-05-08 TW TW101116393A patent/TWI554508B/en active
- 2012-05-08 UA UAA201313451A patent/UA110519C2/en unknown
- 2012-05-08 AU AU2012256220A patent/AU2012256220B2/en active Active
- 2012-05-08 DK DK12721146.4T patent/DK2710000T3/en active
- 2012-05-08 EP EP20120721146 patent/EP2710000B1/en active Active
- 2012-05-08 HR HRP20150588TT patent/HRP20150588T1/en unknown
- 2012-05-08 RS RS20150379A patent/RS54048B1/en unknown
- 2012-05-08 PL PL12721146T patent/PL2710000T3/en unknown
- 2012-05-08 EA EA201391542A patent/EA023711B1/en not_active IP Right Cessation
- 2012-05-08 BR BR112013028132-4A patent/BR112013028132B1/en active IP Right Grant
- 2012-05-08 PE PE2013002523A patent/PE20141041A1/en not_active Application Discontinuation
- 2012-05-08 SG SG2013082615A patent/SG194836A1/en unknown
- 2012-05-08 US US13/883,025 patent/US8648081B2/en active Active
- 2012-05-08 MX MX2013013446A patent/MX341739B/en active IP Right Grant
- 2012-05-08 SI SI201230196T patent/SI2710000T1/en unknown
- 2012-05-08 JP JP2014511394A patent/JP5989099B2/en active Active
- 2012-05-08 KR KR1020137030230A patent/KR101586553B1/en active Active
- 2012-05-08 ES ES12721146.4T patent/ES2543392T3/en active Active
-
2013
- 2013-09-26 CR CR20130486A patent/CR20130486A/en unknown
- 2013-10-21 IL IL229007A patent/IL229007A/en not_active IP Right Cessation
- 2013-11-15 CL CL2013003287A patent/CL2013003287A1/en unknown
- 2013-11-19 CO CO13271916A patent/CO6811873A2/en active IP Right Grant
- 2013-12-20 US US14/137,279 patent/US8889710B2/en active Active
-
2014
- 2014-11-18 US US14/546,590 patent/US9255111B2/en active Active
-
2015
- 2015-06-04 CY CY20151100494T patent/CY1116450T1/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008019760A1 (en) * | 2006-08-15 | 2008-02-21 | Bayer Cropscience Ag | Insecticidal isoxazolines |
| WO2008154528A2 (en) * | 2007-06-13 | 2008-12-18 | E. I. Du Pont De Nemours And Company | Isoxazoline insecticides |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11179372B2 (en) | 2015-06-23 | 2021-11-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| US11878006B2 (en) | 2015-06-23 | 2024-01-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| CN110167540A (en) * | 2016-10-31 | 2019-08-23 | 斯克利普斯研究所 | For preventing the method and composition of the propagation of vector-borne disease |
| EP3532041A4 (en) * | 2016-10-31 | 2020-06-24 | The Scripps Research Institute | METHOD AND COMPOSITIONS FOR PREVENTING VECTOR-INDUCED DISEASE TRANSMISSION |
| WO2018166855A1 (en) * | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2019122324A1 (en) | 2017-12-21 | 2019-06-27 | Intervet International B.V. | Antiparasitic pour-on compositions |
| EP4559482A2 (en) | 2017-12-21 | 2025-05-28 | Intervet International B.V. | Antiparasitic pour-on compositions |
| US12390450B2 (en) | 2017-12-21 | 2025-08-19 | Intervet Inc. | Antiparasitic pour-on compositions |
| WO2020088949A1 (en) * | 2018-10-29 | 2020-05-07 | Basf Se | Process for preparation of optically enriched aldol compounds |
| WO2022256284A1 (en) * | 2021-06-02 | 2022-12-08 | Fmc Corporation | Fused pyridines for controlling invertebrate pests |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2012155352A1 (en) | Dihydroisoxazole compounds, parasiticidal uses and formulations thereof | |
| TWI832838B (en) | Novel anthranilamides, their use as insecticide and processes for preparing the same | |
| TWI454472B (en) | Organic compounds | |
| JP2014224128A (en) | Novel heteroaromatic amides and thioamides as pesticides | |
| EP2956453A1 (en) | 3,5-disubstituted-4,5-dihydro-1,2,4-oxadiazoles for controlling nematode pests | |
| TWI822717B (en) | Pyrazolopyridine-diamides, their use as insecticide and processes for preparing the same | |
| TW202216700A (en) | Isoxazoline compounds and their use as pest control agents | |
| WO2001007413A1 (en) | 1-aryl-3-thioalkyl pyrazoles, the synthesis thereof and the use thereof as insecticides | |
| KR20240044416A (en) | Isoxazoline derivatives | |
| HK1191337B (en) | Parasiticidal dihydroisoxazole compounds | |
| NZ616409B2 (en) | Parasiticidal dihydroisoxazole compounds | |
| TW201927776A (en) | Anthranilamides, their use as insecticide and processes for preparing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11865721 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 11865721 Country of ref document: EP Kind code of ref document: A1 |