WO2012132558A1 - Oxime ester compound and photoinitiator containing said compound - Google Patents
Oxime ester compound and photoinitiator containing said compound Download PDFInfo
- Publication number
- WO2012132558A1 WO2012132558A1 PCT/JP2012/052775 JP2012052775W WO2012132558A1 WO 2012132558 A1 WO2012132558 A1 WO 2012132558A1 JP 2012052775 W JP2012052775 W JP 2012052775W WO 2012132558 A1 WO2012132558 A1 WO 2012132558A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- compound
- carbon atoms
- acid
- atom
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C391/00—Compounds containing selenium
- C07C391/02—Compounds containing selenium having selenium atoms bound to carbon atoms of six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C251/00—Compounds containing nitrogen atoms doubly-bound to a carbon skeleton
- C07C251/32—Oximes
- C07C251/62—Oximes having oxygen atoms of oxyimino groups esterified
- C07C251/64—Oximes having oxygen atoms of oxyimino groups esterified by carboxylic acids
- C07C251/66—Oximes having oxygen atoms of oxyimino groups esterified by carboxylic acids with the esterifying carboxyl groups bound to hydrogen atoms, to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/23—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and nitrogen atoms, not being part of nitro or nitroso groups, bound to the same carbon skeleton
- C07C323/46—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and nitrogen atoms, not being part of nitro or nitroso groups, bound to the same carbon skeleton having at least one of the nitrogen atoms, not being part of nitro or nitroso groups, further bound to other hetero atoms
- C07C323/47—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and nitrogen atoms, not being part of nitro or nitroso groups, bound to the same carbon skeleton having at least one of the nitrogen atoms, not being part of nitro or nitroso groups, further bound to other hetero atoms to oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/62—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/64—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and sulfur atoms, not being part of thio groups, bound to the same carbon skeleton
- C07C323/65—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and sulfur atoms, not being part of thio groups, bound to the same carbon skeleton containing sulfur atoms of sulfone or sulfoxide groups bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C327/00—Thiocarboxylic acids
- C07C327/20—Esters of monothiocarboxylic acids
- C07C327/28—Esters of monothiocarboxylic acids having sulfur atoms of esterified thiocarboxyl groups bound to carbon atoms of hydrocarbon radicals substituted by singly-bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D303/00—Compounds containing three-membered rings having one oxygen atom as the only ring hetero atom
- C07D303/02—Compounds containing oxirane rings
- C07D303/12—Compounds containing oxirane rings with hydrocarbon radicals, substituted by singly or doubly bound oxygen atoms
- C07D303/18—Compounds containing oxirane rings with hydrocarbon radicals, substituted by singly or doubly bound oxygen atoms by etherified hydroxyl radicals
- C07D303/20—Ethers with hydroxy compounds containing no oxirane rings
- C07D303/22—Ethers with hydroxy compounds containing no oxirane rings with monohydroxy compounds
- C07D303/23—Oxiranylmethyl ethers of compounds having one hydroxy group bound to a six-membered aromatic ring, the oxiranylmethyl radical not being further substituted, i.e.
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/34—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D307/38—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D307/40—Radicals substituted by oxygen atoms
- C07D307/42—Singly bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/50—Organo-phosphines
- C07F9/5022—Aromatic phosphines (P-C aromatic linkage)
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/20—Filters
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/0005—Production of optical devices or components in so far as characterised by the lithographic processes or materials used therefor
- G03F7/0007—Filters, e.g. additive colour filters; Components for display devices
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
- G03F7/028—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds with photosensitivity-increasing substances, e.g. photoinitiators
- G03F7/031—Organic compounds not covered by group G03F7/029
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
- G03F7/032—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds with binders
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/038—Macromolecular compounds which are rendered insoluble or differentially wettable
- G03F7/0388—Macromolecular compounds which are rendered insoluble or differentially wettable with ethylenic or acetylenic bands in the side chains of the photopolymer
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
Definitions
- the present invention relates to a novel oxime ester compound useful as a photopolymerization initiator for use in a photosensitive composition, a photopolymerization initiator containing the compound, a polymerizable compound having an ethylenically unsaturated bond, and the photopolymerization initiator. And a cured product of the photosensitive composition.
- the photosensitive composition is obtained by adding a photopolymerization initiator to a polymerizable compound having an ethylenically unsaturated bond, and can be polymerized and cured by irradiating energy rays (light). It is used for photosensitive printing plates and various photoresists.
- Patent Documents 1 to 3 propose oxime ester compounds.
- the oxime ester compound having satisfactory sensitivity has a problem of low transmittance in the visible light region and a desired color cannot be obtained with a color filter (particularly, protection).
- a color filter particularly, protection
- photosensitive compositions used for applications where transparency is required, such as films, and resists for color filters using blue pigments or dyes brightness and color purity decrease when compounds having absorption at 380 to 450 nm are mixed.
- the oxime ester compound having a high visible light transmittance has a problem that the sensitivity is not sufficiently satisfactory, and a photopolymerization initiator having both characteristics has been demanded.
- the colored alkali-developable photosensitive resin composition containing a color material used for a color filter or the like is required to have high sensitivity, and the photopolymerization initiator in the resist needs to have a high concentration.
- a high concentration photopolymerization initiator has caused residues due to deterioration of developability, contamination of the photomask and heating furnace due to sublimation, and the like.
- the problem to be solved is that there has never been a photopolymerization initiator having satisfactory sensitivity, high transmittance in the visible light region, and low sublimation.
- the object of the present invention is high sensitivity that is excellent in stability, low sublimation, excellent developability, high transmittance in the visible light region, and efficiently absorbs near UV light such as 365 nm and is activated. It is in providing the photoinitiator of this.
- the present inventors have made the compound used as a photopolymerization initiator react with a resin or the like when it is made into a resin composition, or react with a resin or the like when irradiated with energy rays (light). It has been found that the sublimation property is lower than that in the case where the compound exists as it is without reacting with.
- the present invention has been made on the basis of the above findings, and provides an oxime ester compound represented by the following general formula (1) and a photopolymerization initiator containing the compound.
- R 1 , R 2 , R 3 and R 4 each independently represents R 11 , OR 11 , COOR 11 , SR 11 , SO 2 R 11 , CONR 12 R 13 or CN;
- R 11 , R 12 and R 13 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms or a carbon atom.
- the hydrogen atom of the substituent represented by R 11 , R 12 and R 13 is further OR 21 , COR 21 , SR 21 , NR 22 R 23 , CONR 22 R 23 , —NR 22 —OR 23 , —NR 22 CO—.
- R 21 , R 22 and R 23 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms or a carbon atom.
- the hydrogen atom of the substituent represented by R 21 , R 22 and R 23 may be further substituted with CN, a halogen atom, a hydroxyl group or a carboxyl group,
- the alkylene part of the substituent represented by R 11 , R 12 , R 13 , R 21 , R 22 and R 23 is —O—, —S—, —COO—, —OCO—, —NR 24 —, — May be interrupted 1 to 5 times by NR 24 COO—, —OCONR 24 —, —SCO—, —COS—, —OCS— or —CSO—
- R 24 represents a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms, or a heterocyclic group having 2 to 20 carbon
- X represents an oxygen atom, a sulfur atom, a selenium atom, CR 31 R 32 , CO, NR 33 or PR 34
- R 31 , R 32 , R 33 and R 34 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms or an arylalkyl having 7 to 30 carbon atoms.
- R 31 , R 32 , R 33 and R 34 may have a branched side chain or may be a cyclic alkyl.
- R 31 , R 32 , R 33 and R 34 The alkyl terminal of the substituent represented by the above formula may be an unsaturated bond, and R 31 , R 32 , R 33 and R 34 each independently form a ring together with one of the adjacent benzene rings.
- a represents an integer of 0 to 4
- b represents an integer of 1 to 5
- At least one of R 4 is —CR 21 ⁇ CR 22 R 23 , —CO—CR 21 ⁇ CR 22 R 23 , —O—CO—CR 21 ⁇ CR 22 R 23 , —N ⁇ C ⁇ O or an epoxy group It is group which has. )
- the present invention also provides a photosensitive composition comprising the photopolymerization initiator and a polymerizable compound having an ethylenically unsaturated bond. Moreover, this invention provides the alkali developable photosensitive resin composition formed by making the said photosensitive composition contain the compound which provides alkali developability. The present invention also provides a colored alkali-developable photosensitive resin composition obtained by further adding a coloring material to the alkali-developable photosensitive resin composition.
- the present invention also provides a cured product obtained by irradiating the photosensitive composition, the alkali-developable photosensitive resin composition, or the colored alkali-developable photosensitive resin composition with energy rays.
- the oxime ester compound of the present invention has a high transmittance in the visible light region and efficiently generates radicals with respect to bright lines such as 365 nm (i-line) and is useful as a photopolymerization initiator.
- the oxime ester compound of the present invention when used as a photopolymerization initiator in a resin composition, it reacts with a resin having a polymerizable group, so that there are few sublimates and further improves the heat resistance of the cured product. be able to.
- the oxime ester compound of the present invention is a novel compound represented by the general formula (1).
- the oxime ester compound has two geometric isomers due to an oxime double bond, but the present invention does not distinguish them. That is, in this specification, the general formula (1), the general formula (2) representing a preferred form of the compound represented by the general formula (1) described later, and the exemplified compound Nos. 1-No.
- the chemical structural formula of 39 shows only one of the two geometric isomers, the oxime ester compound of the present invention is not limited to the geometric isomer represented by these formulas, and the other geometric isomer. Or a mixture of two geometric isomers.
- alkyl group for example, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, s-butyl, t-butyl, amyl, isoamyl, t-amyl, hexyl, heptyl, octyl, isooctyl, 2-ethylhexyl, t-butyl
- alkyl group for example, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, s-butyl, t-butyl, amyl, isoamyl, t-amyl, hexyl, heptyl, octyl, isooctyl, 2-ethylhexyl, t-butyl
- Examples include octyl, nonyl, isononyl, decyl, isodecyl
- aryl groups include phenyl, tolyl, xylyl, ethylphenyl, naphthyl, anthryl, phenanthrenyl, and phenyl, biphenylyl, naphthyl, anthryl substituted with one or more of the above alkyl groups.
- the optionally substituted heterocyclic group having 2 to 20 carbon atoms represented by R 11 , R 12 , R 13 , R 21 , R 22 , R 23 , R 24 in the general formula (1) Is, for example, pyridyl, pyrimidyl, furyl, thienyl, tetrahydrofuryl, dioxolanyl, benzoxazol-2-yl, tetrahydropyranyl, pyrrolidyl, imidazolidyl, pyrazolidyl, thiazolidyl, isothiazolidyl, oxazolidyl, isoxazolidyl, piperidyl, piperazyl, morpholinyl Preferred examples thereof include 5- to 7-membered heterocycles such as
- R 12 and R 13 , R 22 and R 23 can be formed together, and R 31 , R 32 , R 33 and R 34 are combined with the adjacent benzene ring.
- Preferred examples of the ring that can be formed include 5- to 7-membered rings such as cyclopentane ring, cyclohexane ring, cyclopentene ring, benzene ring, piperidine ring, morpholine ring, lactone ring, and lactam ring.
- examples of the halogen atom that may substitute R 11 , R 12 , R 13 , R 21 , R 22 , and R 23 include fluorine, chlorine, bromine, and iodine.
- the alkylene moiety of the substituent represented by R 11 , R 12 , R 13 , R 21 , R 22 , R 23 is —O—, —S—, —COO—, —OCO—, —NR 24 —. , —NR 24 COO—, —OCONR 24 —, —SCO—, —COS—, —OCS—, or —CSO— may be interrupted 1 to 5 times. Two or more groups may be used, and in the case of a group that can be interrupted continuously, two or more groups may be interrupted continuously.
- the alkyl part of the substituent may have a branched side chain or may be a cyclic alkyl.
- X is a sulfur atom or NR 33
- R 33 may have a branched side chain, and may be a cyclic alkyl.
- a compound having an alkyl group of ⁇ 20; a compound represented by the following general formula (2) is particularly preferred because of its high sensitivity and easy production.
- R 5 is the same as R 4 in the above general formula (1), and at least R 5 represents at least a structure selected from —CR 21 ⁇ CR 22 R 23 , —CO—CR 21 ⁇ CR 22 R 23 , —O—CO—CR 21 ⁇ CR 22 R 23 , —N ⁇ C ⁇ O and an epoxy group.
- c represents an integer of 0 to 4.
- R 5 is —CR 21 ⁇ CR 22 R 23 , —CO—CR 21 ⁇ CR 22 R 23, or —O—CO—CR 21 ⁇ CR 22 R 23. Is more preferable because it has high reactivity and exhibits high heat resistance when combined with a resin composition having an unsaturated double bond, and R 5 has —N ⁇ C ⁇ O or an epoxy group. The compound is also more preferable because it exhibits high heat resistance when combined with a resin having active hydrogen such as alcohol, amine, amide and the like.
- R 1 and R 2 in the above general formulas (1) and (2) may have 1 to 20 carbon atoms (especially 1 to 10 carbon atoms, especially carbon atoms). It is preferably an alkyl group having 1 to 4 atoms, a in the general formulas (1) and (2) is preferably 0 to 1, and b in the general formula (1) is 1 to 2 is preferable, and c in the general formula (2) is preferably 0 to 1.
- R 3 is preferably a hydrogen atom.
- X is preferably a sulfur atom.
- —OCO—CR 21 ⁇ CR 22 R 23 is bonded to the carbon atom farthest from the oxygen atom of OR 11 when the hydrogen atom of R 11 of OR 11 is substituted.
- the hydrogen atom of R 11 of COOR 11 is substituted, it is preferably bonded to the carbon atom located farthest from the COO group of COOR 11 .
- b is 1, and R 4 is preferably bonded to the para position with respect to the carbon atom to which X is bonded.
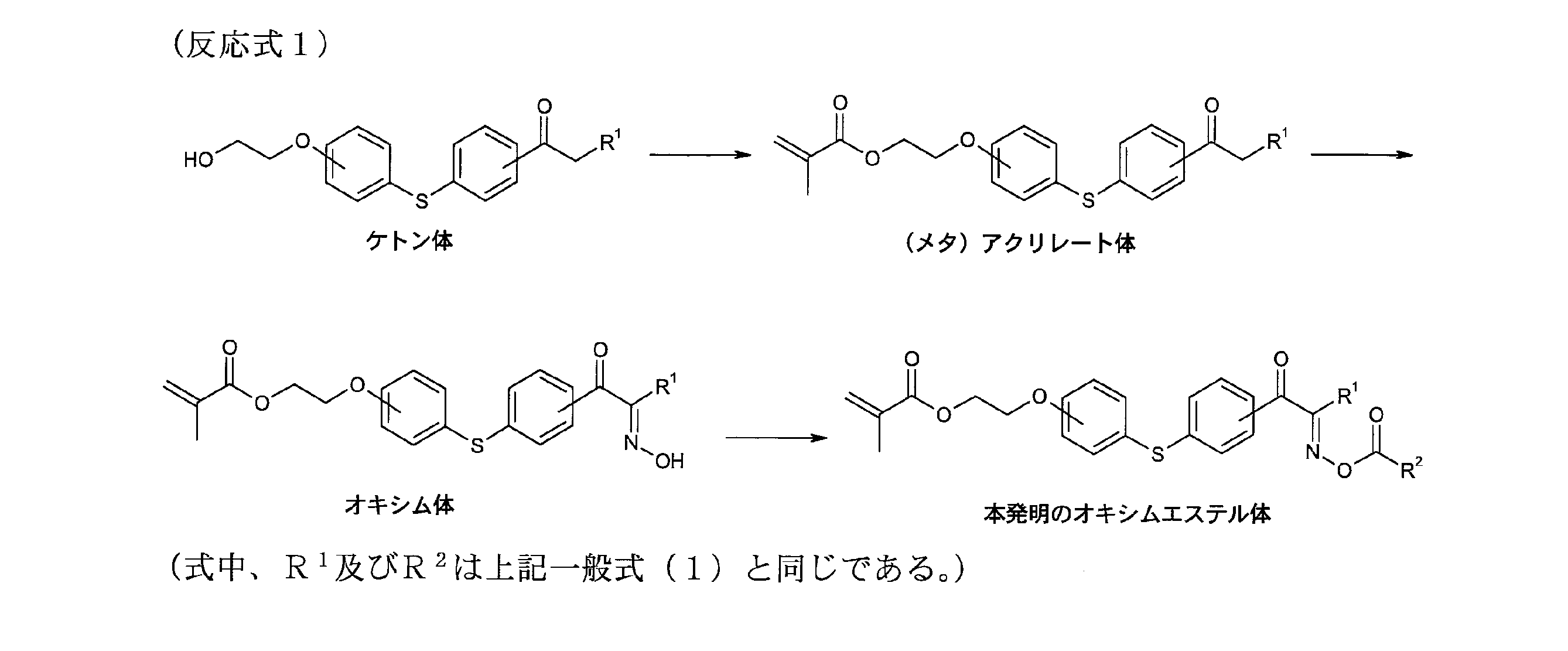
- the oxime ester compound of the present invention represented by the general formula (1) is not particularly limited, but can be produced, for example, according to the method described in JP-A No. 2000-80068. As one of the methods, it can be produced by the following method according to the following reaction formula 1. First, a (meth) acrylate body is obtained by reacting a ketone body with (meth) acrylic acid in the presence of a Lewis acid. Next, the oxime ester compound of the present invention represented by the above general formula (1) is obtained by reacting the (meth) acrylate body with a nitrite in the presence of hydrochloric acid, and subsequently reacting with an acid anhydride or acid chloride. Get.
- reaction formula 1 describes the case where X is a sulfur atom, those in which X is an oxygen atom, a selenium atom, CR 31 R 32 , NR 33 or PR 34 are also produced according to the above method. be able to.
- novel oxime ester compound of the present invention described above is useful as a photopolymerization initiator.
- the photopolymerization initiator of the present invention contains at least one oxime ester compound of the present invention, and is particularly useful as a photopolymerization initiator for a polymerizable compound having an ethylenically unsaturated bond.
- the content of the oxime ester compound of the present invention in the photopolymerization initiator of the present invention is preferably 30 to 100% by mass, more preferably 50 to 100% by mass.
- the photopolymerization initiator of the present invention may contain other photopolymerization initiator in addition to the oxime ester compound of the present invention. As other photopolymerization initiators, conventionally known compounds can be used.
- R 1 , R 2 and R 33 are the same as those in the general formula (1), Y 3 represents a halogen atom or an alkyl group, and n is 0 to 5.
- R 1 , R 2 and R 33 are the same as those in the general formula (1), Y 3 and n are the same as those in the general formula (4), and R ′ 1 and R ′ 2 are R 1 and R 2 are the same, R ′ 33 is the same as R 33 , Y ′ 3 is the same as Y 3 , R 8 is a diol residue or a dithiol residue, Z 5 is an oxygen atom Or represents a sulfur atom.
- R 1 , R 2 and R 33 are the same as in the general formula (1), Y 3 and n are the same as in the general formula (4), and Z 6 is an oxygen atom or a sulfur atom. Or represents a selenium atom, A represents a heterocyclic group, t is an integer of 0 to 5, and u is 0 or 1.
- the photosensitive composition of the present invention contains (A) a photopolymerization initiator of the present invention and (B) a polymerizable compound having an ethylenically unsaturated bond as essential components, and (C) an inorganic compound as an optional component. (D) It contains a combination of components such as a coloring material and a solvent.
- the content of the photopolymerization initiator of the present invention is not particularly limited, but the content of the oxime ester compound of the present invention in the photosensitive composition of the present invention.
- the amount is preferably 1 to 70 parts by weight, more preferably 1 to 50 parts by weight, and most preferably 5 to 30 parts by weight with respect to 100 parts by weight of the polymerizable compound (B) having an ethylenically unsaturated bond.
- the polymerizable compound having an ethylenically unsaturated bond (B) is not particularly limited, and those conventionally used in photosensitive compositions can be used.
- those conventionally used in photosensitive compositions can be used.
- ethylene, propylene, butylene, isobutylene can be used.
- Unsaturated polybasic acids such as polyfunctional (meth) acrylates; (meth) (-2-hydroxyethyl acrylate, 2-hydroxypropyl (meth) acrylate, glycidyl (meth) acrylate; A1-No.
- A4 methyl (meth) acrylate, butyl (meth) acrylate, isobutyl (meth) acrylate, (t-butyl) (meth) acrylate, cyclohexyl (meth) acrylate, n-octyl (meth) acrylate, ( Isooctyl (meth) acrylate, isononyl (meth) acrylate, stearyl (meth) acrylate, lauryl (meth) acrylate, methoxyethyl (meth) acrylate, dimethylaminomethyl (meth) acrylate, dimethyl (meth) acrylate Aminoethyl, aminopropyl (meth) acrylate, dimethylaminopropyl (meth) acrylate, ethoxyethyl (meth) acrylate, poly (ethoxy) ethyl (meth) acrylate, butoxyethoxyethyl (
- Examples of the polymerizable compound (B) having an ethylenically unsaturated bond include acrylic acid ester copolymers, phenol and / or cresol novolac epoxy resins, polyphenylmethane type epoxy resins having polyfunctional epoxy groups, and epoxy.
- Resins obtained by allowing an unsaturated monobasic acid to act on an acrylate resin or an epoxy compound such as an epoxy compound represented by the following general formula (3), and further causing a polybasic acid anhydride to act on the epoxy compound can also be used.
- a resin obtained by allowing an unsaturated monobasic acid to act on an epoxy compound such as an epoxy compound represented by the following general formula (3) and further causing a polybasic acid anhydride to act is preferable.
- these compounds preferably contain 0.2 to 1.0 equivalent of unsaturated groups.
- X 1 is a direct bond, methylene group, alkylidene group having 1 to 4 carbon atoms, alicyclic hydrocarbon group having 3 to 20 carbon atoms, O, S, SO 2 , SS, SO, CO, OCO or a substituent represented by the following formula (I), (B) or (C), wherein the alkylidene group may be substituted with a halogen atom, and R 51 , R 52 , R 53 and R 54 are Each independently represents a hydrogen atom, an alkyl group having 1 to 5 carbon atoms, an alkoxy group having 1 to 8 carbon atoms, an alkenyl group having 2 to 5 carbon atoms, or a halogen atom, and the above alkyl group, alkoxy group and The alkenyl group may be substituted with a halogen atom, m is an integer of 0 to 10, and the optical isomer present when m is not 0 may be any isomer.)
- Z 3 is a hydrogen atom, an alkyl group having 1 to 10 carbon atoms, a phenyl group optionally substituted by an alkoxy group having 1 to 10 carbon atoms, or an alkyl group having 1 to 10 carbon atoms, or A cycloalkyl group having 3 to 10 carbon atoms which may be substituted by an alkoxy group having 1 to 10 carbon atoms
- Y 1 is an alkyl group having 1 to 10 carbon atoms, alkoxy having 1 to 10 carbon atoms Group, an alkenyl group having 2 to 10 carbon atoms or a halogen atom, wherein the alkyl group, alkoxy group and alkenyl group may be substituted with a halogen atom, and d is an integer of 0 to 5.
- Y 2 and Z 4 are each independently an alkyl group having 1 to 10 carbon atoms which may be substituted with a halogen atom, or 6 to 20 carbon atoms which may be substituted with a halogen atom.
- Examples of the unsaturated monobasic acid that acts on the epoxy compound include acrylic acid, methacrylic acid, crotonic acid, cinnamic acid, sorbic acid, hydroxyethyl methacrylate / malate, hydroxyethyl acrylate / malate, hydroxypropyl methacrylate / malate, hydroxypropyl Acrylate / malate, dicyclopentadiene / malate and the like can be mentioned.
- the polybasic acid anhydride to be acted after the unsaturated monobasic acid is allowed to act is biphenyltetracarboxylic dianhydride, tetrahydrophthalic anhydride, succinic anhydride, biphthalic anhydride, maleic anhydride, Trimellitic anhydride, pyromellitic anhydride, 2,2'-3,3'-benzophenone tetracarboxylic anhydride, ethylene glycol bisanhydro trimellitate, glycerol tris anhydro trimellitate, hexahydrophthalic anhydride , Methyltetrahydrophthalic anhydride, nadic anhydride, methylnadic anhydride, trialkyltetrahydrophthalic anhydride, hexahydrophthalic anhydride, 5- (2,5-dioxotetrahydrofuryl) -3-methyl-3-cyclohexene -1,2-dicarboxylic an
- the reaction molar ratio of the epoxy compound, the unsaturated monobasic acid and the polybasic acid anhydride is preferably as follows. That is, in an epoxy adduct having a structure in which 0.1 to 1.0 carboxyl groups of the unsaturated monobasic acid are added to one epoxy group of the epoxy compound, the hydroxyl group 1 of the epoxy adduct is It is preferable that the ratio of the acid anhydride structure of the polybasic acid anhydride is 0.1 to 1.0. Reaction of the said epoxy compound, the said unsaturated monobasic acid, and the said polybasic acid anhydride can be performed in accordance with a conventional method.
- the amount used is preferably 50 to 99% by mass in the component (B).
- the compound having an acid value can be used after adjusting the acid value by further reacting with a monofunctional or polyfunctional epoxy compound.
- the alkali developability of the photosensitive resin can be improved by adjusting the acid value of the compound having an acid value.
- the compound having an acid value (that is, a polymerizable compound having an ethylenically unsaturated bond imparting alkali developability) preferably has a solid acid value in the range of 5 to 120 mgKOH / g, and is monofunctional or polyfunctional.
- the amount of the functional epoxy compound used is preferably selected so as to satisfy the acid value.
- Examples of the monofunctional epoxy compound include glycidyl methacrylate, methyl glycidyl ether, ethyl glycidyl ether, propyl glycidyl ether, isopropyl glycidyl ether, butyl glycidyl ether, isobutyl glycidyl ether, t-butyl glycidyl ether, pentyl glycidyl ether, hexyl glycidyl ether, heptyl Glycidyl ether, octyl glycidyl ether, nonyl glycidyl ether, decyl glycidyl ether, undecyl glycidyl ether, dodecyl glycidyl ether, tridecyl glycidyl ether, tetradecyl glycidyl ether, pentadecy
- the polyfunctional epoxy compound when one or more compounds selected from the group consisting of bisphenol-type epoxy compounds and glycidyl ethers are used, a (colored) alkali-developable photosensitive resin composition having better characteristics can be obtained. Is preferable.
- the bisphenol-type epoxy compound the epoxy compound represented by the general formula (3) can be used, and for example, a bisphenol-type epoxy compound such as a hydrogenated bisphenol-type epoxy compound can also be used.
- glycidyl ethers examples include ethylene glycol diglycidyl ether, propylene glycol diglycidyl ether, 1,4-butanediol diglycidyl ether, 1,6-hexanediol diglycidyl ether, 1,8-octanediol diglycidyl ether, 1,10-decanediol diglycidyl ether, 2,2-dimethyl-1,3-propanediol diglycidyl ether, diethylene glycol diglycidyl ether, triethylene glycol diglycidyl ether, tetraethylene glycol diglycidyl ether, hexaethylene glycol diglycidyl Ether, 1,4-cyclohexanedimethanol diglycidyl ether, 1,1,1-tri (glycidyloxymethyl) propane, 1,1,1-to (Glycidyloxymethyl) ethane, 1,1,1-tri (glycidy
- novolac epoxy compounds such as phenol novolac epoxy compounds, biphenyl novolac epoxy compounds, cresol novolac epoxy compounds, bisphenol A novolac epoxy compounds, dicyclopentadiene novolac epoxy compounds; 3,4-epoxy-6-methyl Cycloaliphatic epoxy such as cyclohexylmethyl-3,4-epoxy-6-methylcyclohexanecarboxylate, 3,4-epoxycyclohexylmethyl-3,4-epoxycyclohexanecarboxylate, 1-epoxyethyl-3,4-epoxycyclohexane Compound: Glycidyl esters such as diglycidyl phthalate, diglycidyl tetrahydrophthalate, glycidyl dimer, tetraglycidyl diamino Glycidylamines such as phenylmethane, triglycidyl P-aminophenol and N, N-diglycidylaniline; heterocycl
- Examples of the inorganic compound (C) that can be contained in the photosensitive composition of the present invention include nickel oxide, iron oxide, iridium oxide, titanium oxide, zinc oxide, magnesium oxide, calcium oxide, potassium oxide, silica, Metal oxides such as alumina; layered clay minerals, miloli blue, calcium carbonate, magnesium carbonate, cobalt, manganese, glass powder (especially glass frit), mica, talc, kaolin, ferrocyanide, various metal sulfates, sulfides , Selenide, aluminum silicate, calcium silicate, aluminum hydroxide, platinum, gold, silver, copper and the like.
- glass frit, titanium oxide, silica, layered clay mineral, silver and the like are preferable.
- the content of the inorganic compound (C) is preferably 0.1 to 1000 parts by mass with respect to 100 parts by mass of the polymerizable compound (B) having an ethylenically unsaturated bond.
- the amount is preferably 10 to 800 parts by mass.
- inorganic compounds are used, for example, as fillers, antireflection agents, conductive agents, stabilizers, flame retardants, mechanical strength improvers, special wavelength absorbers, ink repellents, and the like.
- examples of the color material (D) that can be contained in the photosensitive composition of the present invention include pigments, dyes, and natural pigments. These color materials can be used alone or in admixture of two or more.
- the pigment examples include nitroso compounds; nitro compounds; azo compounds; diazo compounds; xanthene compounds; quinoline compounds; anthraquinone compounds; coumarin compounds; phthalocyanine compounds; isoindolinone compounds; Compounds; perylene compounds; diketopyrrolopyrrole compounds; thioindigo compounds; dioxazine compounds; triphenylmethane compounds; quinophthalone compounds; naphthalene tetracarboxylic acids; azo dyes, metal complex compounds of cyanine dyes; Carbon black obtained by the method, or carbon black such as acetylene black, ketjen black or lamp black; Prepared by coating or coating with an epoxy resin, carbon black previously dispersed with a resin in a solvent and adsorbed with 20 to 200 mg / g of resin, carbon black treated with an acidic or alkaline surface, average Carbon black having a particle size of 8 nm or more and a DBP oil absorption of 90 m
- Graphitized carbon black activated carbon, carbon fiber, carbon nanotube, carbon microcoil, carbon nanohorn, carbon aerogel, fullerene
- aniline black pigment black 7, titanium black
- chromium oxide green, miloli blue, cobalt green, cobalt blue, manganese series Ferrus Organics such as fluoride, phosphate blue, bitumen, ultramarine, cerulean blue, pyridian, emerald green, lead sulfate, yellow lead, zinc yellow, red bean (red iron (III) oxide), cadmium red, synthetic iron black, amber, etc.
- an inorganic pigment can be used. These pigments can be used alone or in combination.
- pigments can also be used as the pigment, for example, Pigment Red 1, 2, 3, 9, 10, 14, 17, 22, 23, 31, 38, 41, 48, 49, 88, 90, 97, 112, 119, 122, 123, 144, 149, 166, 168, 169, 170, 171, 177, 179, 180, 184, 185, 192, 200, 202, 209, 215, 216, 217, 220, 223, 224, 226, 227, 228, 240, 254; Pigment Orange 13, 31, 34, 36, 38, 43, 46, 48, 49, 51, 52, 55, 59, 60, 61, 62, 64, 65, 71; Pigment Yellow 1, 3, 12, 13, 14, 16, 17, 20, 24, 55, 60, 73, 81, 83, 86, 93, 95, 9 98, 100, 109, 110, 113, 114, 117, 120, 125, 126, 127, 129, 137, 138, 139, 147, 148,
- dyes As the above dyes, azo dyes, anthraquinone dyes, indigoid dyes, triarylmethane dyes, xanthene dyes, alizarin dyes, acridine dyes stilbene dyes, thiazole dyes, naphthol dyes, quinoline dyes, nitro dyes, indamine dyes, oxazine dyes, phthalocyanine dyes And dyes such as cyanine dyes, and a plurality of these may be used in combination.
- the content thereof is preferably 50 to 350 parts by mass with respect to 100 parts by mass of the polymerizable compound (B) having an ethylenically unsaturated bond. More preferably, it is 100 to 250 parts by mass.
- the photosensitive composition of the present invention may contain a compound that does not have an ethylenically unsaturated bond and imparts alkali developability.
- an alkaline aqueous solution can be obtained by having an acid value.
- a typical example is an alkali-soluble novolak resin (hereinafter simply referred to as “novolak resin”).
- the novolak resin is obtained by polycondensation of phenols and aldehydes in the presence of an acid catalyst.
- phenols examples include phenol, o-cresol, m-cresol, p -cresol, o-ethylphenol, m-ethylphenol, p-ethylphenol, o-butylphenol, m-butylphenol, p-butylphenol, 2, 3-xylenol, 2,4-xylenol, 2,5-xylenol, 3,4-xylenol, 3,5-xylenol, 2,3,5-trimethylphenol, p-phenylphenol, hydroquinone, catechol, resorcinol, 2- Methyl resorcinol, pyrogallol, ⁇ -naphthol, bisphenol A, dihydroxybenzoic acid ester, gallic acid ester, etc.
- phenols are used.
- phenol, o-cresol, m-cresol, p-cresol, 2,5-kis Silenol, 3,5-xylenol, 2,3,5-trimethylphenol, resorcinol, 2-methylresorcinol and bisphenol A are preferred.
- These phenols are used alone or in combination.
- aldehydes examples include formaldehyde, paraformaldehyde, acetaldehyde, propylaldehyde, benzaldehyde, phenylacetaldehyde, ⁇ -phenylpropylaldehyde, ⁇ -phenylpropylaldehyde, o-hydroxybenzaldehyde, m-hydroxybenzaldehyde, p-hydroxybenzaldehyde, o -Chlorobenzaldehyde, m-chlorobenzaldehyde, p-chlorobenzaldehyde, o-nitrobenzaldehyde, m-nitrobenzaldehyde, p-nitrobenzaldehyde, o-methylbenzaldehyde, m-methylbenzaldehyde, p-methylbenzaldehyde, p-ethylbenzaldehyde, p N-butylbenzaldehyde is
- the acid catalyst examples include inorganic acids such as hydrochloric acid, nitric acid, and sulfuric acid, or organic acids such as formic acid, oxalic acid, and acetic acid.
- the amount of these acid catalysts used is preferably 1 ⁇ 10 ⁇ 4 to 5 ⁇ 10 ⁇ 1 mol per mol of phenols.
- water is usually used as a reaction medium.
- the reaction medium is hydrophilic.
- a solvent can also be used.
- hydrophilic solvents examples include alcohols such as methanol, ethanol, propanol and butanol, and cyclic ethers such as tetrahydrofuran and dioxane.
- the amount of these reaction media used is usually 20 to 1000 parts by mass per 100 parts by mass of the reaction raw material.
- the reaction temperature of the condensation reaction can be appropriately adjusted according to the reactivity of the reaction raw materials, but is usually 10 to 200 ° C., preferably 70 to 150 ° C. After completion of the condensation reaction, in order to remove unreacted raw materials, acid catalyst and reaction medium present in the system, the internal temperature is generally increased to 130 to 230 ° C., and the volatile component is distilled off under reduced pressure.
- the melted novolac resin is collected on a steel belt or the like.
- the reaction mixture is dissolved in the hydrophilic solvent and added to a precipitating agent such as water, n-hexane or n-heptane to precipitate a novolak resin, and the precipitate is separated and dried by heating. It can also be recovered by doing so.
- Examples other than the novolak resin include polyhydroxystyrene or a derivative thereof, a styrene-maleic anhydride copolymer, polyvinylhydroxybenzoate, and the like.
- a solvent can be further added to the photosensitive composition of the present invention.
- a solvent capable of dissolving or dispersing each of the above components ((A) the photopolymerization initiator of the present invention and (B) a polymerizable compound having an ethylenically unsaturated bond), for example, methyl ethyl ketone, Ketones such as methyl amyl ketone, diethyl ketone, acetone, methyl isopropyl ketone, methyl isobutyl ketone, cyclohexanone, 2-heptanone; ethyl ether, dioxane, tetrahydrofuran, 1,2-dimethoxyethane, 1,2-diethoxyethane, di Ether solvents such as propylene glycol dimethyl ether; ester solvents such as methyl acetate, ethyl acetate, n-propyl acetate, isopropyl
- ketones, ether ester solvents, etc. particularly propylene glycol-1-monomethyl ether-2-acetate, cyclohexanone, etc. are preferable because the compatibility between the resist and the photopolymerization initiator is improved in the photosensitive composition.
- the content of the solvent is not particularly limited, each component is uniformly dispersed or dissolved, and the photosensitive composition of the present invention exhibits a liquid or paste shape suitable for each application.
- the solvent be contained so that the solid content (all components other than the solvent) in the photosensitive composition of the present invention is 10 to 50% by mass.
- a dispersant for dispersing (C) an inorganic compound and / or (D) a coloring material can be added.
- the dispersant is not limited as long as it can disperse and stabilize the (C) inorganic compound or (D) coloring material, and a commercially available dispersant, for example, BYK series manufactured by BYK Chemie, Inc. can be used.
- a compound having an amine value of 1 to 100 m g KOH / g is preferably used.
- cured material can also be improved by using another organic polymer with (B) the polymeric compound which has an ethylenically unsaturated bond.
- the organic polymer include polystyrene, polymethyl methacrylate, methyl methacrylate-ethyl acrylate copolymer, poly (meth) acrylic acid, styrene- (meth) acrylic acid copolymer, (meth) acrylic acid-methyl methacrylate.
- Copolymer ethylene-vinyl chloride copolymer, ethylene-vinyl copolymer, polyvinyl chloride resin, ABS resin, nylon 6, nylon 66, nylon 12, urethane resin, polycarbonate polyvinyl butyral, cellulose ester, polyacrylamide, saturated
- polyester phenol resin, phenoxy resin, polyamideimide resin, polyamic acid resin, epoxy resin, etc.
- polystyrene, (meth) acrylic acid-methyl methacrylate copolymer, and epoxy resin are preferred. Arbitrariness.
- the amount used is preferably 10 to 500 parts by mass with respect to 100 parts by mass of the polymerizable compound (B) having an ethylenically unsaturated bond.
- a chain transfer agent a sensitizer, a surfactant, a silane coupling agent, a melamine compound, and the like can be further used in combination.
- a sulfur atom-containing compound is generally used.
- Alkyl compounds trimethylolpropane tris (3-mercaptoisobutyrate), butanediol bis (3-mercaptoisobutyrate), hexanedithiol, decanedithiol, 1,4- Methyl mercaptobenzene, butanediol bisthiopropionate, butanediol bisthioglycolate, ethylene glycol bisthioglycolate, trimethylolpropane tristhioglycolate, butanediol bisthiopropionate, trimethylolpropane tristhiopropionate , Trimethylolpropane tristhioglycolate, pentaerythritol tetrakisthiopropionate, pentaerythritol tetrakisthioglycolate, trishydroxyethyltristhiopropionate, diethylthioxanthone, diisopropyl
- the surfactant examples include fluorine surfactants such as perfluoroalkyl phosphates and perfluoroalkyl carboxylates; anionic surfactants such as higher fatty acid alkali salts, alkyl sulfonates, and alkyl sulfates; higher amines Cationic surfactants such as halogenates and quaternary ammonium salts; Nonionic surfactants such as polyethylene glycol alkyl ethers, polyethylene glycol fatty acid esters, sorbitan fatty acid esters, and fatty acid monoglycerides; amphoteric surfactants; silicone surfactants Surfactants such as agents can be used, and these may be used in combination.
- fluorine surfactants such as perfluoroalkyl phosphates and perfluoroalkyl carboxylates
- anionic surfactants such as higher fatty acid alkali salts, alkyl sulfonates, and alkyl sulfates
- silane coupling agent for example, a silane coupling agent manufactured by Shin-Etsu Chemical Co., Ltd. can be used. Among them, an isocyanate group, a methacryloyl group, or an epoxy group, such as KBE-9007, KBM-502, and KBE-403, can be used. A silane coupling agent is preferably used.
- Examples of the melamine compound include all or part of active methylol groups (CH 2 OH groups) in nitrogen compounds such as (poly) methylol melamine, (poly) methylol glycoluril, (poly) methylol benzoguanamine, and (poly) methylol urea.
- nitrogen compounds such as (poly) methylol melamine, (poly) methylol glycoluril, (poly) methylol benzoguanamine, and (poly) methylol urea.
- Examples include compounds in which (at least two) are alkyl etherified.
- examples of the alkyl group constituting the alkyl ether include a methyl group, an ethyl group, and a butyl group, which may be the same as or different from each other.
- the methylol group which is not alkyletherified may be self-condensed within one molecule, or may be condensed between two molecules, and as a result, an oligomer component may be formed.
- an oligomer component may be formed.
- hexamethoxymethyl melamine, hexabutoxymethyl melamine, tetramethoxymethyl glycoluril, tetrabutoxymethyl glycoluril and the like can be used.
- alkyl etherified melamines such as hexamethoxymethyl melamine and hexabutoxymethyl melamine are preferable.
- the photosensitive composition of the present invention includes, if necessary, a thermal polymerization inhibitor such as p-anisole, hydroquinone, pyrocatechol, t-butylcatechol, phenothiazine; a plasticizer; an adhesion promoter; a filler; Conventional additives such as a foaming agent, a leveling agent, a surface conditioner, an antioxidant, an ultraviolet absorber, a dispersion aid, a coagulation inhibitor, a catalyst, an effect accelerator, a cross-linking agent, and a thickener can be added.
- the photopolymerization initiator of the present invention and (B) any component other than the polymerizable compound having an ethylenically unsaturated bond (however, (C) inorganic compound, (D) color
- the amount of use is appropriately selected according to the purpose of use and is not particularly limited, but is preferably (B) in total based on 100 parts by mass of the polymerizable compound having an ethylenically unsaturated bond. 50 parts by mass or less.
- the photosensitive composition of the present invention can be cured by irradiating energy rays.
- cured material is formed as a suitable shape according to a use.
- the photosensitive composition of the present invention is prepared by using a known means such as a spin coater, a roll coater, a bar coater, a die coater, a curtain coater, various printing, dipping, etc.
- the present invention can be applied to a supporting substrate such as glass, quartz glass, semiconductor substrate, metal, paper, and plastic. Moreover, after once applying on support bases, such as a film, it can also transfer on another support base
- the light source of the energy ray used for curing the photosensitive composition of the present invention includes an ultrahigh pressure mercury lamp, a high pressure mercury lamp, a medium pressure mercury lamp, a low pressure mercury lamp, a mercury vapor arc lamp, a xenon arc lamp, a carbon arc.
- High energy rays such as electromagnetic energy, electron beam, X-ray, radiation, etc. having a wavelength of 2000 angstrom to 7000 angstrom obtained from lamp, metal halide lamp, fluorescent lamp, tungsten lamp, excimer lamp, germicidal lamp, light emitting diode, CRT light source, etc.
- the laser direct drawing method that directly forms an image from digital information such as a computer without using a mask improves not only productivity but also resolution and positional accuracy.
- the laser light light having a wavelength of 340 to 430 nm is preferably used, but excimer laser, nitrogen laser, argon ion laser, helium cadmium laser, helium neon laser, krypton ion laser.
- Various semiconductor lasers and YAG lasers that emit light in the visible to infrared region can also be used. When these laser beams are used, a sensitizing dye that absorbs the visible to infrared region is preferably added.
- the photosensitive composition of the present invention comprises a photocurable paint or varnish; a photocurable adhesive; a printed circuit board; a color filter in a color display liquid crystal display element such as a color television, a PC monitor, a personal digital assistant, a digital camera; Color filter of image sensor; Electrode material for plasma display panel; Powder coating; Printing ink; Printing plate; Adhesive; Dental composition; Gel coat; Photoresist for electronics; Electroplating resist; Solder resists; resists for manufacturing color filters for various display applications or forming their structures in the manufacturing process of plasma display panels, electroluminescent display devices, and LCDs; for encapsulating electrical and electronic components Composition; solder resist; magnetic recording material; micromachine Article; waveguide; optical switch; plating mask; etching mask; color test system; glass fiber cable coating; stencil for screen printing; material for manufacturing three-dimensional objects by stereolithography; holographic recording material; Materials; fine electronic circuits; bleaching materials; bleaching materials for image recording materials; bleaching materials for image recording materials
- the photosensitive composition of the present invention can also be used for the purpose of forming a spacer for a liquid crystal display panel and for forming a protrusion for a vertical alignment type liquid crystal display element.
- it is useful as a photosensitive composition for simultaneously forming protrusions and spacers for a vertical alignment type liquid crystal display element.
- the spacer for a liquid crystal display panel includes (1) a step of forming a coating film of the photosensitive composition of the present invention on a substrate, and (2) an energy beam (through a mask having a predetermined pattern shape on the coating film). (3) baking step after exposure, (4) step of developing the film after exposure, and (5) step of heating the film after development.
- the photosensitive composition of the present invention to which a coloring material is added is suitably used as a resist constituting each pixel such as RGB in a color filter and a black matrix resist forming a partition of each pixel. Furthermore, in the case of a black matrix resist to which an ink repellent agent is added, it is preferably used for a partition for an ink jet color filter having a profile angle of 50 ° or more.
- the ink repellent agent a fluorosurfactant and a composition containing a fluorosurfactant are preferably used.
- the partition formed from the photosensitive composition of the present invention partitions the transferred object, and droplets are applied to the recessed portions on the partitioned transferred object by the inkjet method.
- an optical element is manufactured by a method of forming an image region.
- the droplets contain a colorant and the image area is colored.
- the optical element produced by the manufacturing method described above is formed from a plurality of colored areas on the substrate. And at least a partition that separates each colored region of the pixel group.
- the photosensitive composition of the present invention can also be used as a protective film or insulating film composition.
- an ultraviolet absorber, an alkylated modified melamine and / or an acrylic modified melamine, a mono- or bifunctional (meth) acrylate monomer containing an alcoholic hydroxyl group in the molecule, and / or a silica sol can be contained.
- the photosensitive composition for the protective film and insulating film As the photosensitive composition for the protective film and insulating film, (A) the photopolymerization initiator of the present invention, (B) a polymerizable compound having an ethylenically unsaturated bond (preferably containing a carboxyl group-containing polymerizable compound having a weight average molecular weight of 2,000 to 40,000 and an acid value of 50 to 200 mgKOH / g), And (X) a resin composition mainly comprising an epoxy compound, With respect to 100 parts by weight of the component (B), the component (A) is 0.01 to 2.0 parts by weight in terms of the amount of the oxime ester compound of the present invention, and the component (X) is 10 to 40 parts by weight. Some are preferred.
- the insulating film is used for the insulating resin layer in a laminate in which an insulating resin layer is provided on a peelable support substrate, and the laminate can be developed with an alkaline aqueous solution.
- the film thickness is preferably 10 to 100 ⁇ m.
- the photosensitive composition of the present invention can be used as a photosensitive paste composition by containing an inorganic compound (C).
- the photosensitive paste composition can be used for forming a fired product pattern such as a partition pattern, a dielectric pattern, an electrode pattern, and a black matrix pattern of a plasma display panel.
- Example 1 Compound No. 1 ⁇ Step 1> Methacrylateation 1- (4- (4- (2-hydroxyethoxy) phenylthio) phenyl) propan-1-one (hereinafter also referred to as ketone body a) 19.8 g (65 mmol), methacrylic acid A solution containing 56.2 g (653 mmol), p-toluenesulfonic acid monohydrate 5.0 g (26 mmol), BHT 1.2 g (5.6 mmol) and dichloroethane 121 g was stirred and refluxed for 10.5 hours. After cooling, ethyl acetate and water were added to separate the oil and water.
- the organic layer was washed with water, an aqueous sodium bicarbonate solution, and water in this order.
- the organic layer was dried over anhydrous magnesium sulfate and then the solvent was removed to obtain 26.3 g of the following methacrylate body a.
- Step 2 Oximation A solution containing 26.0 g (70 mmol) of the methacrylate body a obtained in Step 1, 7.3 g (70 mmol) of concentrated hydrochloric acid and 70 g of dimethylformamide was cooled to 5 ° C., and isobutyl nitrite 10. 8 g (105 mmol) was added and stirred at room temperature for 3 hours. Ethyl acetate and water were added to separate the oil and water, and the organic layer was washed with water, an aqueous sodium hydrogen carbonate solution and water in this order. The organic layer was dried over anhydrous magnesium sulfate and then the solvent was removed to obtain 23.8 g of the following oxime body a.
- Step 3 Oxime esterification A solution containing 23.8 g (60 mmol) of the oxime body a obtained in Step 2, 9.1 g (90 mmol) of acetic anhydride and 79 g of ethyl acetate was stirred at 70 ° C. for 4 hours. After cooling, water was added and the oil was separated. The organic layer was washed with water, an aqueous sodium bicarbonate solution, and water in this order. The organic layer was dried over anhydrous magnesium sulfate and then desolvated. 27.2 g of the obtained crude product was purified by a column chromatogram, and the compound no. 2.3 g of 1 was obtained. It was confirmed by analysis that the obtained compound was the target product. The analysis results are shown in [Table 1] to [Table 3].
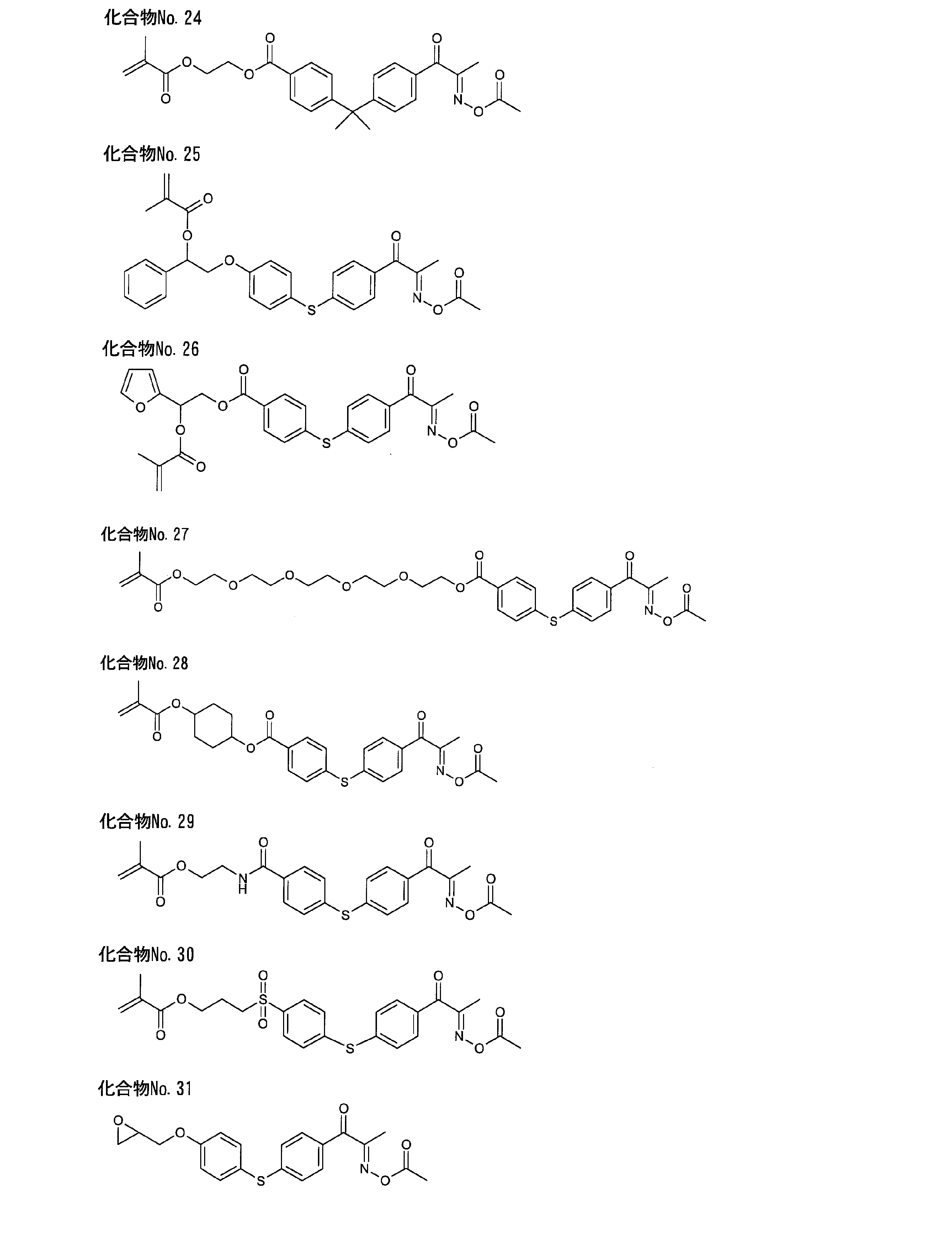
- Example 2 to 4 Compound No. 2, Compound No. 3, Compound No. 4 According to the method described in Example 1, except that the corresponding ketone body was used instead of ketone body a (and acrylic acid was used instead of methacrylic acid for compound No. 3), compound No. 3 was used. 2, Compound No. 3 and compound no. 4 were produced respectively.
- the analysis results are shown in [Table 1] to [Table 3].
- ⁇ Step 2> Alkali-developable photosensitive resin composition No. Preparation of Alkali Developable Resin No. 1 obtained in ⁇ Step 1> as component (B) imparting alkali developability 14.7 g of dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) as a component (B), surfactant FZ-2122 (manufactured by Nippon Unicar Co., Ltd.) 1.8 g of a cyclohexanone 1% solution, 10.0 g of propylene glycol-1-monomethyl ether-2-acetate and 20.2 g of cyclohexanone were mixed, and the compound No. 1 obtained in Example 1 as component (A) was mixed. 0.3 g of No. 1 was added and stirred well, and the alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention. 1 was obtained.
- component (B)
- Example 6 Alkali-developable photosensitive resin composition No. Preparation of Compound No. 2 obtained in Example 1 1 to compound No. 1 obtained in Example 2. Except for changing to No. 2, in the same manner as in ⁇ Step 2> of Example 5, the alkali-developable photosensitive resin composition No. 1 as the photosensitive composition of the present invention was used. 2 was obtained.
- Example 7 Alkali-developable photosensitive resin composition No. Preparation of Compound No. 3 obtained in Example 1 1 to compound No. 1 obtained in Example 3. 3 except that the alkaline developing photosensitive resin composition No. 1 which is the photosensitive composition of the present invention was prepared in the same manner as in ⁇ Step 2> of Example 5. 3 was obtained.
- the permeability test of No. 5 was conducted as follows. That is, the alkali-developable photosensitive resin composition was spin-coated on a glass substrate (500 rpm for 2 seconds, 800 rpm for 6 seconds), and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, after exposure to 150 mJ / cm 2 using a high-pressure mercury lamp as a light source, post-baking was performed at 230 ° C. for 30 minutes using an oven to obtain a cured film. About the obtained cured film, the transmittance
- the photosensitive composition of the present invention using the oxime ester compound of the present invention as a photopolymerization initiator is more transparent than the photosensitive compositions of Comparative Examples 1 and 2 using a comparative compound. The rate is high.
- Example 8 Conductive composition No. Production of Alkali Developable Resin No. 1 obtained in ⁇ Step 1> of Example 5 as component (B) for imparting alkali developability 14.1 g of 1, dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) as component (B), surfactant BYK-323 (manufactured by Big Chemie Japan Co., Ltd.) 0.05 g, 11.3 g of texanol (2,2,4-trimethyl-1,3-pentanediol monoisobutyrate), compound No.
- Example 1 obtained in Example 1 as component (A) 0.7 g of 1 and 3.1 g of glass frit and 63.7 g of silver powder as component (C) were stirred and kneaded with a three-roll mill, and the conductive composition which is the photosensitive composition of the present invention. No. 1 was obtained.
- Example 9 Colored alkali-developable photosensitive resin composition No. ⁇ Step 1> Production of Blue Dispersion 57.6 g of Blue Pigment Pigment Blue 15: 6, Dispersant Azisper PB821 (manufactured by Ajinomoto Fine Techno Co., Ltd.) 20.0 g, Dispersing Agent Solsperse 5000 (Avicia Co., Ltd.) )) 2.4 g and propylene glycol-1-monomethyl ether-2-acetate 320.0 g are put into a 500 ml polyethylene container, and 350 g of zirconia beads having a particle diameter of 0.5 mm are used for 10 hours in a paint conditioner. After shaking, the zirconia beads were filtered off to obtain a blue dispersion.
- ⁇ Step 2> Colored alkali-developable photosensitive resin composition No. Production of 1 (D) 10.63 g of the blue dispersion obtained in ⁇ Step 1> as component (B) Alkali developability obtained in ⁇ Step 1> of Example 5 as component (B) that imparts alkali developability Resin No.
- the permeability test of No. 3 was conducted as follows. A colored alkali-developable photosensitive resin composition was spin-coated (500 rpm, 7 seconds) on a glass substrate, and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, after exposure to 150 mJ / cm 2 using a high-pressure mercury lamp as a light source, post-baking was performed at 230 ° C. for 30 minutes using an oven to obtain a cured film. As a heat resistance test, the obtained cured film was further heated at 260 ° C. for 1 hour. The cured film after the heat resistance test was measured for transmittance at 420 nm using an absorptiometer. The results are shown in [Table 5].
- the colored alkali-developable photosensitive resin composition of the present invention using the oxime ester compound of the present invention as a photopolymerization initiator is a colored alkali-developable feeling of Comparative Examples 3 and 4 using a comparative compound.
- the transmittance is higher than that of the light-sensitive resin composition.
- the weight reduction rate of the cured product was measured as follows. That is, the colored alkali-developable photosensitive resin composition was spin coated (500 rpm, 7 seconds) on a glass substrate, and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, the film was exposed to 100 mJ / cm 2 using a high-pressure mercury lamp as a light source to obtain a cured film. About 3 mg of this cured film was scraped off, and the weight reduction rate when held at 230 ° C. for 30 minutes was measured with TG-DTA (manufactured by Seiko Instruments Inc.) using the scraped cured film as a sample. The results are shown in [Table 6].
- the colored alkali-developable photosensitive resin composition which is the photosensitive composition of the present invention using the oxime ester compound of the present invention as a photopolymerization initiator, is colored in Comparative Examples 4 to 6 using the comparative compound. It is clear that it has excellent heat resistance and less outgas compared to the alkali-developable photosensitive resin composition. In addition, from this result, it is guessed that the oxime ester compound of this invention as a photoinitiator reacted with the resin component, and, thereby, the sublimation property of the oxime ester compound and / or resin component of this invention became low.
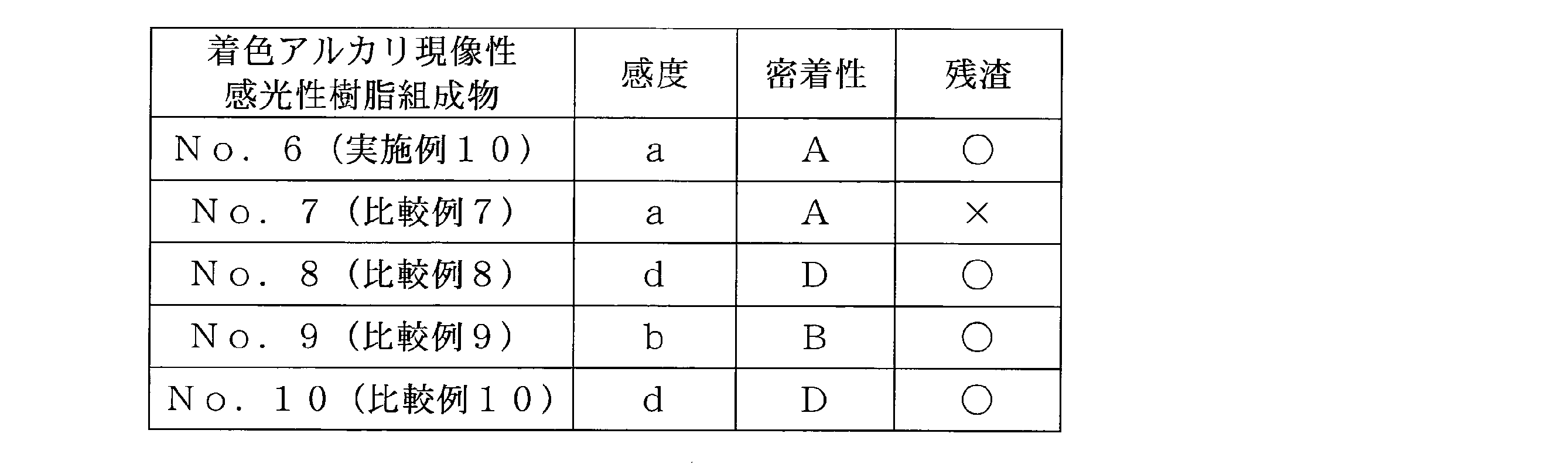
- Example 10 Colored alkali-developable photosensitive resin composition No. 6.
- Preparation (6) As component (D) 10.63 g of the blue dispersion obtained in ⁇ Step 1> of Example 9, and as component (B) imparting alkali developability, obtained in ⁇ Step 1> of Example 5 Alkali developable resin No.
- sensitivity The sensitivity of the colored alkali-developable photosensitive resin composition was evaluated in the following four stages. That is, the exposure amount when the line width of the formed pattern exceeds the mask opening line width is 50 mJ / cm 2 , a is 100 mJ / cm 2 , b is 150 mJ / cm 2 The case where there was c and the case where a pattern was not formed even at 150 mJ / cm 2 was taken as d. (Adhesion) The adhesion of the colored alkali-developable photosensitive resin composition was evaluated in the following four stages.
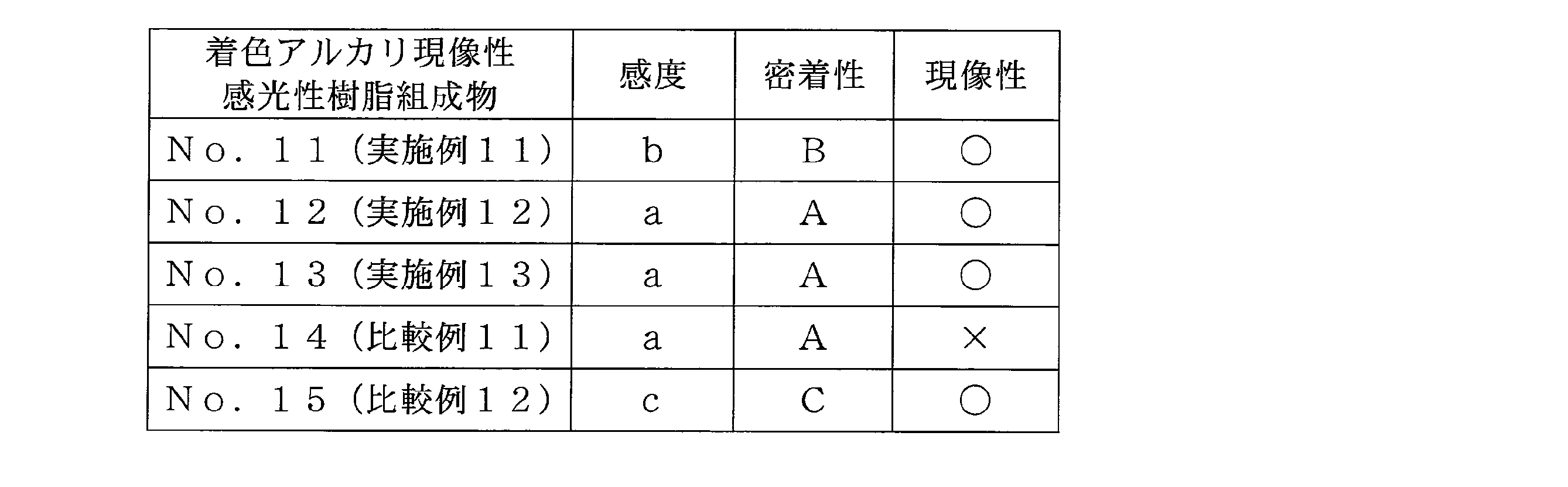
- Example 11 Colored alkali-developable photosensitive resin composition No. 11: (D) 7.06 g of a carbon black dispersion prepared by changing Pigment Blue 15: 6 in ⁇ Step 1> of Example 9 to carbon black as component (D), component (B) imparting alkali developability As the alkali developable resin No. obtained in ⁇ Step 1> of Example 5.
- Example 12 Colored alkali-developable photosensitive resin composition No. No. 12 Compound No. obtained in Example 1 1 to compound No. 1 obtained in Example 2. A colored alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention was prepared in the same manner as in Example 11 except that it was changed to 2. 12 was obtained.
- Example 13 Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 13 obtained in Example 1 1 to compound No. 1 obtained in Example 3. Except for the change to 3 in the same manner as in Example 11, the colored alkali-developable photosensitive resin composition No. 1 as the photosensitive composition of the present invention was used. 13 was obtained.
- sensitivity The sensitivity of the colored alkali-developable photosensitive resin composition was evaluated in the following three stages. That is, when the line width of the formed pattern exceeds the mask opening line width, the exposure amount is 100 mJ / cm 2 , a is 120 mJ / cm 2 , b is 120 mJ / cm 2 The case where a pattern was not formed was defined as c.
- Adhesion The adhesion of the colored alkali-developable photosensitive resin composition was evaluated in the following four stages.
- the transparency and developability in the visible light region are excellent as compared with the compounds used in Comparative Examples 2, 3, 7, and 11.
- the oxime ester compound of the present invention it is excellent in transparency, heat resistance, sensitivity, and adhesion in the visible light region as compared with the compounds used in Comparative Examples 4 and 8.
- the oxime ester compound of this invention it is excellent in heat resistance, a sensitivity, and adhesiveness compared with the compound used in Comparative Examples 5, 9, and 12.
- the oxime ester compound of the present invention it is excellent in transparency, heat resistance, sensitivity and adhesion in the visible light region as compared with the compounds used in Comparative Examples 1, 6 and 10.
- the visible light region has high transparency, excellent heat resistance, and excellent photolithography properties.
- the oxime ester compound of the present invention is useful as a photopolymerization initiator. is there.
Abstract
Description
しかし、特許文献1~3に記載のオキシムエステル化合物のうち、感度が満足できるオキシムエステル化合物は、可視光領域の透過率が低く、カラーフィルタで所望する色が得られないという問題(特に、保護膜のような透明性が求められる用途に使用する感光性組成物や、青色の顔料又は色素を用いたカラーフィルタ向けレジストでは、380~450nmに吸収を持つ化合物が混在すると明度、色純度が低下する)があり、また、可視光領域の透過率が高いオキシムエステル化合物は、感度が十分満足できるものではないという問題があり、両特性を兼ね備える光重合開始剤が求められていた。
また、カラーフィルタ等に用いられる色材を含有する着色アルカリ現像性感光性樹脂組成物は高感度であることが求められ、レジスト中における光重合開始剤を高濃度にする必要があった。しかし、高濃度の光重合開始剤は、現像性の悪化による残渣の発生や、昇華物によるフォトマスクや加熱炉の汚染等の原因となっていた。 As photopolymerization initiators used in the photosensitive composition, Patent Documents 1 to 3 propose oxime ester compounds.
However, among the oxime ester compounds described in Patent Documents 1 to 3, the oxime ester compound having satisfactory sensitivity has a problem of low transmittance in the visible light region and a desired color cannot be obtained with a color filter (particularly, protection). In photosensitive compositions used for applications where transparency is required, such as films, and resists for color filters using blue pigments or dyes, brightness and color purity decrease when compounds having absorption at 380 to 450 nm are mixed. The oxime ester compound having a high visible light transmittance has a problem that the sensitivity is not sufficiently satisfactory, and a photopolymerization initiator having both characteristics has been demanded.
In addition, the colored alkali-developable photosensitive resin composition containing a color material used for a color filter or the like is required to have high sensitivity, and the photopolymerization initiator in the resist needs to have a high concentration. However, a high concentration photopolymerization initiator has caused residues due to deterioration of developability, contamination of the photomask and heating furnace due to sublimation, and the like.
R11、R12及びR13は、それぞれ独立に、水素原子、又は炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基、炭素原子数7~30のアリールアルキル基若しくは炭素原子数2~20の複素環基である置換基を表し、
R11、R12及びR13で表わされる置換基の水素原子は、更にOR21、COR21、SR21、NR22R23、CONR22R23、-NR22-OR23、-NR22CO-OCOR23、-C(=N-OR21)-R22、-C(=N-OCOR21)-R22、CN、ハロゲン原子、-COOR21、-CR21=CR22R23、-CO-CR21=CR22R23、-O-CO-CR21=CR22R23、-N=C=O又はエポキシ基で置換されていてもよく、
R21、R22及びR23は、それぞれ独立に、水素原子、又は炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基、炭素原子数7~30のアリールアルキル基若しくは炭素原子数2~20の複素環基である置換基を表し、
R21、R22及びR23で表される置換基の水素原子は、更にCN、ハロゲン原子、水酸基又はカルボキシル基で置換されていてもよく、
R11、R12、R13、R21、R22及びR23で表される置換基のアルキレン部分は、-O-、-S-、-COO-、-OCO-、-NR24-、-NR24COO-、-OCONR24-、-SCO-、-COS-、-OCS-又は-CSO-により1~5回中断されていてもよく、
R24は、水素原子、炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基、炭素原子数7~30のアリールアルキル基又は炭素原子数2~20の複素環基を表し、
R11、R12、R13、R21、R22及びR23で表される置換基のアルキル部分は、分岐側鎖があってもよく、環状アルキルであってもよく、また、R12とR13及びR22とR23はそれぞれ一緒になって環を形成していてもよく、
Xは、酸素原子、硫黄原子、セレン原子、CR31R32、CO、NR33又はPR34を表し、
R31、R32、R33及びR34は、それぞれ独立に、水素原子、又は炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基若しくは炭素原子数7~30のアリールアルキル基である置換基を表し、
R31、R32、R33及びR34で表される置換基のアルキル部分は、分岐側鎖があってもよく、環状アルキルであってもよく、R31、R32、R33及びR34で表される置換基のアルキル末端は不飽和結合であってもよく、R31、R32、R33及びR34は、それぞれ独立に、隣接するどちらかのベンゼン環と一緒になって環を形成していてもよく、
aは、0~4の整数を表し、
bは、1~5の整数を表し、
R4の少なくとも一つは、-CR21=CR22R23、-CO-CR21=CR22R23、-O-CO-CR21=CR22R23、-N=C=O又はエポキシ基を有する基である。)
R 11 , R 12 and R 13 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms or a carbon atom. Represents a substituent which is a heterocyclic group having 2 to 20 atoms;
The hydrogen atom of the substituent represented by R 11 , R 12 and R 13 is further OR 21 , COR 21 , SR 21 , NR 22 R 23 , CONR 22 R 23 , —NR 22 —OR 23 , —NR 22 CO—. OCOR 23 , —C (═N—OR 21 ) —R 22 , —C (═N—OCOR 21 ) —R 22 , CN, halogen atom, —COOR 21 , —CR 21 ═CR 22 R 23 , —CO— CR 21 ═CR 22 R 23 , —O—CO—CR 21 ═CR 22 R 23 , —N═C═O or an epoxy group may be substituted,
R 21 , R 22 and R 23 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms or a carbon atom. Represents a substituent which is a heterocyclic group having 2 to 20 atoms;
The hydrogen atom of the substituent represented by R 21 , R 22 and R 23 may be further substituted with CN, a halogen atom, a hydroxyl group or a carboxyl group,
The alkylene part of the substituent represented by R 11 , R 12 , R 13 , R 21 , R 22 and R 23 is —O—, —S—, —COO—, —OCO—, —NR 24 —, — May be interrupted 1 to 5 times by NR 24 COO—, —OCONR 24 —, —SCO—, —COS—, —OCS— or —CSO—,
R 24 represents a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms, or a heterocyclic group having 2 to 20 carbon atoms. ,
Alkyl moiety of the substituents represented by R 11, R 12, R 13 , R 21, R 22 and R 23, there may be branched side chain, may be a cyclic alkyl, and the R 12 R 13 and R 22 and R 23 may be combined to form a ring,
X represents an oxygen atom, a sulfur atom, a selenium atom, CR 31 R 32 , CO, NR 33 or PR 34 ;
R 31 , R 32 , R 33 and R 34 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms or an arylalkyl having 7 to 30 carbon atoms. Represents a substituent that is a group,
The alkyl part of the substituent represented by R 31 , R 32 , R 33 and R 34 may have a branched side chain or may be a cyclic alkyl. R 31 , R 32 , R 33 and R 34 The alkyl terminal of the substituent represented by the above formula may be an unsaturated bond, and R 31 , R 32 , R 33 and R 34 each independently form a ring together with one of the adjacent benzene rings. May be formed,
a represents an integer of 0 to 4,
b represents an integer of 1 to 5,
At least one of R 4 is —CR 21 ═CR 22 R 23 , —CO—CR 21 ═CR 22 R 23 , —O—CO—CR 21 ═CR 22 R 23 , —N═C═O or an epoxy group It is group which has. )
また、本発明は、上記感光性組成物に、アルカリ現像性を付与する化合物を含有させてなるアルカリ現像性感光性樹脂組成物を提供するものである。
また、本発明は、上記アルカリ現像性感光性樹脂組成物に、更に色材を含有させてなる着色アルカリ現像性感光性樹脂組成物を提供するものである。 The present invention also provides a photosensitive composition comprising the photopolymerization initiator and a polymerizable compound having an ethylenically unsaturated bond.
Moreover, this invention provides the alkali developable photosensitive resin composition formed by making the said photosensitive composition contain the compound which provides alkali developability.
The present invention also provides a colored alkali-developable photosensitive resin composition obtained by further adding a coloring material to the alkali-developable photosensitive resin composition.
即ち、本明細書において、上記一般式(1)、並びに後述する上記一般式(1)で表される化合物の好ましい形態を表す一般式(2)、及び例示化合物No.1~No.39の化学構造式は、2つの幾何異性体のうちの一方のみを示しているが、本発明のオキシムエステル化合物は、これらの式で示される幾何異性体に限定されず、もう一方の幾何異性体でもよいし、2つの幾何異性体の混合物であってもよい。 The oxime ester compound of the present invention is a novel compound represented by the general formula (1). The oxime ester compound has two geometric isomers due to an oxime double bond, but the present invention does not distinguish them.
That is, in this specification, the general formula (1), the general formula (2) representing a preferred form of the compound represented by the general formula (1) described later, and the exemplified compound Nos. 1-No. Although the chemical structural formula of 39 shows only one of the two geometric isomers, the oxime ester compound of the present invention is not limited to the geometric isomer represented by these formulas, and the other geometric isomer. Or a mixture of two geometric isomers.
本発明の感光性組成物は、必須成分として、(A)本発明の光重合開始剤及び(B)エチレン性不飽和結合を有する重合性化合物を含有し、任意成分として、(C)無機化合物、(D)色材、溶媒等の成分を組み合わせて含有するものである。 Next, the photosensitive composition of the present invention will be described in detail.
The photosensitive composition of the present invention contains (A) a photopolymerization initiator of the present invention and (B) a polymerizable compound having an ethylenically unsaturated bond as essential components, and (C) an inorganic compound as an optional component. (D) It contains a combination of components such as a coloring material and a solvent.
即ち、上記エポキシ化合物のエポキシ基1個に対し、上記不飽和一塩基酸のカルボキシル基が0.1~1.0個で付加させた構造を有するエポキシ付加物において、該エポキシ付加物の水酸基1個に対し、上記多塩基酸無水物の酸無水物構造が0.1~1.0個となる比率となるようにするのが好ましい。
上記エポキシ化合物、上記不飽和一塩基酸および上記多塩基酸無水物の反応は、常法に従って行なうことができる。 The reaction molar ratio of the epoxy compound, the unsaturated monobasic acid and the polybasic acid anhydride is preferably as follows.
That is, in an epoxy adduct having a structure in which 0.1 to 1.0 carboxyl groups of the unsaturated monobasic acid are added to one epoxy group of the epoxy compound, the hydroxyl group 1 of the epoxy adduct is It is preferable that the ratio of the acid anhydride structure of the polybasic acid anhydride is 0.1 to 1.0.
Reaction of the said epoxy compound, the said unsaturated monobasic acid, and the said polybasic acid anhydride can be performed in accordance with a conventional method.
また、上記酸価を有する化合物は、更に単官能又は多官能エポキシ化合物を反応させることにより酸価調整してから用いることもできる。上記酸価を有する化合物の酸価を調整することにより、感光性樹脂のアルカリ現像性を改良することができる。上記酸価を有する化合物(即ちアルカリ現像性を付与するエチレン性不飽和結合を有する重合性化合物)は、固形分の酸価が5~120mgKOH/gの範囲であることが好ましく、単官能又は多官能エポキシ化合物の使用量は、上記酸価を満たすように選択するのが好ましい。 Among the polymerizable compounds having an ethylenically unsaturated bond, when a compound having an acid value is used, alkali developability can be imparted to the photosensitive composition of the present invention. When the compound having the acid value is used, the amount used is preferably 50 to 99% by mass in the component (B).
The compound having an acid value can be used after adjusting the acid value by further reacting with a monofunctional or polyfunctional epoxy compound. The alkali developability of the photosensitive resin can be improved by adjusting the acid value of the compound having an acid value. The compound having an acid value (that is, a polymerizable compound having an ethylenically unsaturated bond imparting alkali developability) preferably has a solid acid value in the range of 5 to 120 mgKOH / g, and is monofunctional or polyfunctional. The amount of the functional epoxy compound used is preferably selected so as to satisfy the acid value.
上記ビスフェノール型エポキシ化合物としては、上記一般式(3)で表されるエポキシ化合物を用いることができる他、例えば、水添ビスフェノール型エポキシ化合物等のビスフェノール型エポキシ化合物も用いることができる。
また上記グリシジルエーテル類としては、エチレングリコールジグリシジルエーテル、プロピレングリコールジグリシジルエーテル、1,4-ブタンジオールジグリシジルエーテル、1,6-ヘキサンジオールジグリシジルエーテル、1,8-オクタンジオールジグリシジルエーテル、1,10-デカンジオールジグリシジルエーテル、2,2-ジメチル-1,3-プロパンジオールジグリシジルエーテル、ジエチレングリコールジグリシジルエーテル、トリエチレングリコールジグリシジルエーテル、テトラエチレングリコールジグリシジルエーテル、ヘキサエチレングリコールジグリシジルエーテル、1,4-シクロヘキサンジメタノールジグリシジルエーテル、1,1,1-トリ(グリシジルオキシメチル)プロパン、1,1,1-トリ(グリシジルオキシメチル)エタン、1,1,1-トリ(グリシジルオキシメチル)メタン、1,1,1,1-テトラ(グリシジルオキシメチル)メタン等を用いることができる。
その他、フェノールノボラック型エポキシ化合物、ビフェニルノボラック型エポキシ化合物、クレゾールノボラック型エポキシ化合物、ビスフェノールAノボラック型エポキシ化合物、ジシクロペンタジエンノボラック型エポキシ化合物等のノボラック型エポキシ化合物;3,4-エポキシ-6-メチルシクロヘキシルメチル-3,4-エポキシ-6-メチルシクロヘキサンカルボキシレート、3,4-エポキシシクロヘキシルメチル-3,4-エポキシシクロヘキサンカルボキシレート、1-エポキシエチル-3,4-エポキシシクロヘキサン等の脂環式エポキシ化合物;フタル酸ジグリシジルエステル、テトラヒドロフタル酸ジグリシジルエステル、ダイマー酸グリシジルエステル等のグリシジルエステル類;テトラグリシジルジアミノジフェニルメタン、トリグリシジルP-アミノフェノール、N,N-ジグリシジルアニリン等のグリシジルアミン類;1,3-ジグリシジル-5,5-ジメチルヒダントイン、トリグリシジルイソシアヌレート等の複素環式エポキシ化合物;ジシクロペンタジエンジオキシド等のジオキシド化合物;ナフタレン型エポキシ化合物;トリフェニルメタン型エポキシ化合物;ジシクロペンタジエン型エポキシ化合物等を用いることもできる。 As the polyfunctional epoxy compound, when one or more compounds selected from the group consisting of bisphenol-type epoxy compounds and glycidyl ethers are used, a (colored) alkali-developable photosensitive resin composition having better characteristics can be obtained. Is preferable.
As the bisphenol-type epoxy compound, the epoxy compound represented by the general formula (3) can be used, and for example, a bisphenol-type epoxy compound such as a hydrogenated bisphenol-type epoxy compound can also be used.
Examples of the glycidyl ethers include ethylene glycol diglycidyl ether, propylene glycol diglycidyl ether, 1,4-butanediol diglycidyl ether, 1,6-hexanediol diglycidyl ether, 1,8-octanediol diglycidyl ether, 1,10-decanediol diglycidyl ether, 2,2-dimethyl-1,3-propanediol diglycidyl ether, diethylene glycol diglycidyl ether, triethylene glycol diglycidyl ether, tetraethylene glycol diglycidyl ether, hexaethylene glycol diglycidyl Ether, 1,4-cyclohexanedimethanol diglycidyl ether, 1,1,1-tri (glycidyloxymethyl) propane, 1,1,1-to (Glycidyloxymethyl) ethane, 1,1,1-tri (glycidyloxymethyl) methane, 1,1,1,1- tetra (glycidyloxymethyl) can be used such as methane.
Other novolac epoxy compounds such as phenol novolac epoxy compounds, biphenyl novolac epoxy compounds, cresol novolac epoxy compounds, bisphenol A novolac epoxy compounds, dicyclopentadiene novolac epoxy compounds; 3,4-epoxy-6-methyl Cycloaliphatic epoxy such as cyclohexylmethyl-3,4-epoxy-6-methylcyclohexanecarboxylate, 3,4-epoxycyclohexylmethyl-3,4-epoxycyclohexanecarboxylate, 1-epoxyethyl-3,4-epoxycyclohexane Compound: Glycidyl esters such as diglycidyl phthalate, diglycidyl tetrahydrophthalate, glycidyl dimer, tetraglycidyl diamino Glycidylamines such as phenylmethane, triglycidyl P-aminophenol and N, N-diglycidylaniline; heterocyclic epoxy compounds such as 1,3-diglycidyl-5,5-dimethylhydantoin and triglycidyl isocyanurate; Dioxide compounds such as pentadiene dioxide; naphthalene type epoxy compounds; triphenylmethane type epoxy compounds; dicyclopentadiene type epoxy compounds can also be used.
これらの中でも、ガラスフリット、酸化チタン、シリカ、層状粘土鉱物、銀等が好ましい。本発明の感光性組成物において、(C)無機化合物の含有量は、(B)エチレン性不飽和結合を有する重合性化合物100質量部に対して、好ましくは0.1~1000質量部、より好ましくは10~800質量部である。尚、これらの無機化合物は1種又は2種以上を使用することができる。 Examples of the inorganic compound (C) that can be contained in the photosensitive composition of the present invention include nickel oxide, iron oxide, iridium oxide, titanium oxide, zinc oxide, magnesium oxide, calcium oxide, potassium oxide, silica, Metal oxides such as alumina; layered clay minerals, miloli blue, calcium carbonate, magnesium carbonate, cobalt, manganese, glass powder (especially glass frit), mica, talc, kaolin, ferrocyanide, various metal sulfates, sulfides , Selenide, aluminum silicate, calcium silicate, aluminum hydroxide, platinum, gold, silver, copper and the like.
Among these, glass frit, titanium oxide, silica, layered clay mineral, silver and the like are preferable. In the photosensitive composition of the present invention, the content of the inorganic compound (C) is preferably 0.1 to 1000 parts by mass with respect to 100 parts by mass of the polymerizable compound (B) having an ethylenically unsaturated bond. The amount is preferably 10 to 800 parts by mass. These inorganic compounds can be used alone or in combination of two or more.
また縮合反応終了後に、前記親水性溶媒に反応混合物を溶解し、水、n-ヘキサン、n-ヘプタン等の沈殿剤に添加することにより、ノボラック樹脂を析出させ、析出物を分離し、加熱乾燥することにより回収することもできる。 Examples of the acid catalyst include inorganic acids such as hydrochloric acid, nitric acid, and sulfuric acid, or organic acids such as formic acid, oxalic acid, and acetic acid. The amount of these acid catalysts used is preferably 1 × 10 −4 to 5 × 10 −1 mol per mol of phenols. In the condensation reaction, water is usually used as a reaction medium. However, if the phenols used in the condensation reaction do not dissolve in an aqueous solution of aldehydes and become heterogeneous from the beginning of the reaction, the reaction medium is hydrophilic. A solvent can also be used. Examples of these hydrophilic solvents include alcohols such as methanol, ethanol, propanol and butanol, and cyclic ethers such as tetrahydrofuran and dioxane. The amount of these reaction media used is usually 20 to 1000 parts by mass per 100 parts by mass of the reaction raw material. The reaction temperature of the condensation reaction can be appropriately adjusted according to the reactivity of the reaction raw materials, but is usually 10 to 200 ° C., preferably 70 to 150 ° C. After completion of the condensation reaction, in order to remove unreacted raw materials, acid catalyst and reaction medium present in the system, the internal temperature is generally increased to 130 to 230 ° C., and the volatile component is distilled off under reduced pressure. Next, the melted novolac resin is collected on a steel belt or the like.
After completion of the condensation reaction, the reaction mixture is dissolved in the hydrophilic solvent and added to a precipitating agent such as water, n-hexane or n-heptane to precipitate a novolak resin, and the precipitate is separated and dried by heating. It can also be recovered by doing so.
これらの中でも、ケトン類、エーテルエステル系溶媒等、特にプロピレングリコール-1-モノメチルエーテル-2-アセテート、シクロヘキサノン等が、感光性組成物において、レジストと光重合開始剤との相溶性がよくなるので好ましい。
本発明の感光性組成物において、溶媒の含有量は、特に制限されず、各成分が均一に分散又は溶解され、また本発明の感光性組成物が各用途に適した液状ないしペースト状を呈する量であればよいが、通常、本発明の感光性組成物中の固形分(溶媒以外の全成分)の量が10~50質量%となる範囲で溶媒を含有させることが好ましい。 If necessary, a solvent can be further added to the photosensitive composition of the present invention. As the solvent, usually, a solvent capable of dissolving or dispersing each of the above components ((A) the photopolymerization initiator of the present invention and (B) a polymerizable compound having an ethylenically unsaturated bond), for example, methyl ethyl ketone, Ketones such as methyl amyl ketone, diethyl ketone, acetone, methyl isopropyl ketone, methyl isobutyl ketone, cyclohexanone, 2-heptanone; ethyl ether, dioxane, tetrahydrofuran, 1,2-dimethoxyethane, 1,2-diethoxyethane, di Ether solvents such as propylene glycol dimethyl ether; ester solvents such as methyl acetate, ethyl acetate, n-propyl acetate, isopropyl acetate, n-butyl acetate, cyclohexyl acetate, ethyl lactate, dimethyl succinate, and texanol; ethylene glycol monomethyl ether Cellosolv solvents such as ethylene glycol monoethyl ether; alcohol solvents such as methanol, ethanol, iso- or n-propanol, iso- or n-butanol, amyl alcohol; ethylene glycol monomethyl acetate, ethylene glycol monoethyl acetate, propylene Ether ester solvents such as glycol-1-monomethyl ether-2-acetate, dipropylene glycol monomethyl ether acetate, 3-methoxybutyl acetate, ethoxyethyl propionate; BTX solvents such as benzene, toluene, xylene; hexane, heptane , Octane, cyclohexane, and other aliphatic hydrocarbon solvents; terpene hydrocarbon oils, such as turpentine oil, D-limonene, and pinene; mineral spirits, swazol # 31 (Cosmo Matsuyama Oil Co., Ltd.), Solvesso # 100 (Exxon Chemical Co., Ltd.) and other paraffinic solvents; carbon tetrachloride, chloroform, trichloroethylene, methylene chloride, 1,2-dichloroethane and other halogenated aliphatic hydrocarbons Solvent; Halogenated aromatic hydrocarbon solvent such as chlorobenzene; Carbitol solvent; Aniline; Triethylamine; Pyridine; Acetic acid; Acetonitrile; Carbon disulfide; N, N-dimethylformamide; Pyrrolidone; dimethyl sulfoxide; water and the like can be used, and these solvents can be used alone or as a mixed solvent of two or more.
Among these, ketones, ether ester solvents, etc., particularly propylene glycol-1-monomethyl ether-2-acetate, cyclohexanone, etc. are preferable because the compatibility between the resist and the photopolymerization initiator is improved in the photosensitive composition. .
In the photosensitive composition of the present invention, the content of the solvent is not particularly limited, each component is uniformly dispersed or dissolved, and the photosensitive composition of the present invention exhibits a liquid or paste shape suitable for each application. However, it is usually preferred that the solvent be contained so that the solid content (all components other than the solvent) in the photosensitive composition of the present invention is 10 to 50% by mass.
他の有機重合体を使用する場合、その使用量は、(B)エチレン性不飽和結合を有する上記重合性化合物100質量部に対して、好ましくは10~500質量部である。 Moreover, in the photosensitive composition of this invention, the characteristic of hardened | cured material can also be improved by using another organic polymer with (B) the polymeric compound which has an ethylenically unsaturated bond. Examples of the organic polymer include polystyrene, polymethyl methacrylate, methyl methacrylate-ethyl acrylate copolymer, poly (meth) acrylic acid, styrene- (meth) acrylic acid copolymer, (meth) acrylic acid-methyl methacrylate. Copolymer, ethylene-vinyl chloride copolymer, ethylene-vinyl copolymer, polyvinyl chloride resin, ABS resin, nylon 6, nylon 66, nylon 12, urethane resin, polycarbonate polyvinyl butyral, cellulose ester, polyacrylamide, saturated Examples thereof include polyester, phenol resin, phenoxy resin, polyamideimide resin, polyamic acid resin, epoxy resin, etc. Among these, polystyrene, (meth) acrylic acid-methyl methacrylate copolymer, and epoxy resin are preferred. Arbitrariness.
When another organic polymer is used, the amount used is preferably 10 to 500 parts by mass with respect to 100 parts by mass of the polymerizable compound (B) having an ethylenically unsaturated bond.
ここで、アルキルエーテルを構成するアルキル基としては、メチル基、エチル基又はブチル基が挙げられ、互いに同一であってもよいし、異なっていてもよい。また、アルキルエーテル化されていないメチロール基は、一分子内で自己縮合していてもよく、二分子間で縮合して、その結果オリゴマー成分が形成されていてもよい。
具体的には、ヘキサメトキシメチルメラミン、ヘキサブトキシメチルメラミン、テトラメトキシメチルグリコールウリル、テトラブトキシメチルグリコールウリル等を用いることができる。
これらの中でも、ヘキサメトキシメチルメラミン、ヘキサブトキシメチルメラミン等のアルキルエーテル化されたメラミンが好ましい。 Examples of the melamine compound include all or part of active methylol groups (CH 2 OH groups) in nitrogen compounds such as (poly) methylol melamine, (poly) methylol glycoluril, (poly) methylol benzoguanamine, and (poly) methylol urea. Examples include compounds in which (at least two) are alkyl etherified.
Here, examples of the alkyl group constituting the alkyl ether include a methyl group, an ethyl group, and a butyl group, which may be the same as or different from each other. Moreover, the methylol group which is not alkyletherified may be self-condensed within one molecule, or may be condensed between two molecules, and as a result, an oligomer component may be formed.
Specifically, hexamethoxymethyl melamine, hexabutoxymethyl melamine, tetramethoxymethyl glycoluril, tetrabutoxymethyl glycoluril and the like can be used.
Among these, alkyl etherified melamines such as hexamethoxymethyl melamine and hexabutoxymethyl melamine are preferable.
(A)本発明の光重合開始剤、
(B)エチレン性不飽和結合を有する重合性化合物(重量平均分子量が2,000~40,000、酸価が50~200mgKOH/gであるカルボキシル基含有の重合性化合物を含むことが好ましい)、及び
(X)エポキシ化合物
を主成分とする樹脂組成物であって、
(B)成分100重量部に対して、(A)成分が本発明のオキシムエステル化合物量に換算して0.01~2.0重量部であり、(X)成分が10~40重量部であるものが好ましい。 As the photosensitive composition for the protective film and insulating film,
(A) the photopolymerization initiator of the present invention,
(B) a polymerizable compound having an ethylenically unsaturated bond (preferably containing a carboxyl group-containing polymerizable compound having a weight average molecular weight of 2,000 to 40,000 and an acid value of 50 to 200 mgKOH / g), And (X) a resin composition mainly comprising an epoxy compound,
With respect to 100 parts by weight of the component (B), the component (A) is 0.01 to 2.0 parts by weight in terms of the amount of the oxime ester compound of the present invention, and the component (X) is 10 to 40 parts by weight. Some are preferred.
<ステップ1>メタクリレート化
1-(4-(4-(2-ヒドロキシエトキシ)フェニルチオ)フェニル)プロパン-1-オン(以下、ケトン体aともいう)19.8g(65mmol)、メタクリル酸56.2g(653mmol)、パラトルエンスルホン酸一水和物5.0g(26mmol)、BHT1.2g(5.6mmol)及びジクロロエタン121gを含む溶液を10.5時間攪拌還流した。冷却後、酢酸エチルと水を加え油水分離した。有機層を水、炭酸水素ナトリウム水溶液、水の順序で洗浄した。有機層を無水硫酸マグネシウムで乾燥後、脱溶媒し下記メタクリレート体aを26.3g得た。 Example 1 Compound No. 1 <Step 1> Methacrylateation 1- (4- (4- (2-hydroxyethoxy) phenylthio) phenyl) propan-1-one (hereinafter also referred to as ketone body a) 19.8 g (65 mmol), methacrylic acid A solution containing 56.2 g (653 mmol), p-toluenesulfonic acid monohydrate 5.0 g (26 mmol), BHT 1.2 g (5.6 mmol) and dichloroethane 121 g was stirred and refluxed for 10.5 hours. After cooling, ethyl acetate and water were added to separate the oil and water. The organic layer was washed with water, an aqueous sodium bicarbonate solution, and water in this order. The organic layer was dried over anhydrous magnesium sulfate and then the solvent was removed to obtain 26.3 g of the following methacrylate body a.
ステップ1で得られたメタクリレート体aの26.0g(70mmol)、濃塩酸7.3g(70mmol)及びジメチルホルムアミド70gを含む溶液を5℃に冷却し、亜硝酸イソブチル10.8g(105mmol)を加え室温で3時間攪拌した。酢酸エチルと水を加え油水分離し、有機層を水、炭酸水素ナトリウム水溶液、水の順序で洗浄した。有機層を無水硫酸マグネシウムで乾燥後、脱溶媒し、下記オキシム体aを23.8g得た。 <Step 2> Oximation A solution containing 26.0 g (70 mmol) of the methacrylate body a obtained in Step 1, 7.3 g (70 mmol) of concentrated hydrochloric acid and 70 g of dimethylformamide was cooled to 5 ° C., and isobutyl nitrite 10. 8 g (105 mmol) was added and stirred at room temperature for 3 hours. Ethyl acetate and water were added to separate the oil and water, and the organic layer was washed with water, an aqueous sodium hydrogen carbonate solution and water in this order. The organic layer was dried over anhydrous magnesium sulfate and then the solvent was removed to obtain 23.8 g of the following oxime body a.
ステップ2で得られたオキシム体aの23.8g(60mmol)、無水酢酸9.1g(90mmol)及び酢酸エチル79gを含む溶液を70℃で4時間攪拌した。冷却後、水を加え油水分離した。有機層を水、炭酸水素ナトリウム水溶液、水の順序で洗浄した。有機層を無水硫酸マグネシウムで乾燥後、脱溶媒した。得られた粗生成物27.2gをカラムクロマトグラムにより精製し化合物No.1を2.3g得た。得られた化合物が目的物であることは、分析により確認した。分析結果を〔表1〕~〔表3〕に示す。 <Step 3> Oxime esterification A solution containing 23.8 g (60 mmol) of the oxime body a obtained in Step 2, 9.1 g (90 mmol) of acetic anhydride and 79 g of ethyl acetate was stirred at 70 ° C. for 4 hours. After cooling, water was added and the oil was separated. The organic layer was washed with water, an aqueous sodium bicarbonate solution, and water in this order. The organic layer was dried over anhydrous magnesium sulfate and then desolvated. 27.2 g of the obtained crude product was purified by a column chromatogram, and the compound no. 2.3 g of 1 was obtained. It was confirmed by analysis that the obtained compound was the target product. The analysis results are shown in [Table 1] to [Table 3].
ケトン体aに代えて対応するケトン体を用い(且つ化合物No.3については、メタクリル酸に代えてアクリル酸を用い)た以外は、実施例1に記載した方法に従って、化合物No.2、化合物No.3及び化合物No.4をそれぞれ製造した。分析結果を〔表1〕~〔表3〕に示す。 [Examples 2 to 4] Compound No. 2, Compound No. 3, Compound No. 4 According to the method described in Example 1, except that the corresponding ketone body was used instead of ketone body a (and acrylic acid was used instead of methacrylic acid for compound No. 3), compound No. 3 was used. 2, Compound No. 3 and compound no. 4 were produced respectively. The analysis results are shown in [Table 1] to [Table 3].
<ステップ1>アルカリ現像性樹脂〔アルカリ現像性を付与する(B)成分〕の調製
反応容器に、ビスフェノールフルオレン型エポキシ樹脂(エポキシ当量231、前記一般式(3)で表されるエポキシ化合物)184g、アクリル酸58g、2,6-ジ-tert-ブチル-p-クレゾール0.26g、テトラ-n-ブチルアンモニウムブロミド0.11g及びプロピレングリコール-1-モノメチルエーテル-2-アセテート23gを仕込み、120℃で16時間攪拌した。反応液を室温まで冷却し、プロピレングリコール-1-モノメチルエーテル-2-アセテート35g、ビフタル酸無水物59g及びテトラ-n-ブチルアンモニウムブロミド0.24gを加えて、120℃で4時間攪拌した。更に、テトラヒドロ無水フタル酸20gを加え、120℃で4時間、100℃で3時間、80℃で4時間、60℃で6時間、40℃で11時間攪拌した後、プロピレングリコール-1-モノメチルエーテル-2-アセテート90gを加えて、プロピレングリコール-1-モノメチルエーテル-2-アセテート溶液として目的物のアルカリ現像性樹脂No.1を得た(Mw=5000、Mn=2100、酸価(固形分)92.7mgKOH/g)。 [Example 5] Alkali-developable photosensitive resin composition No. 1. Preparation of Step 1 <Preparation of Alkali Developable Resin [Component (B) that Gives Alkali Developability] In a reaction vessel, a bisphenolfluorene type epoxy resin (epoxy equivalent 231; an epoxy represented by the above general formula (3) Compound) 184 g, acrylic acid 58 g, 2,6-di-tert-butyl-p-cresol 0.26 g, tetra-n-butylammonium bromide 0.11 g and propylene glycol-1-monomethyl ether-2-acetate 23 g , And stirred at 120 ° C. for 16 hours. The reaction solution was cooled to room temperature, 35 g of propylene glycol-1-monomethyl ether-2-acetate, 59 g of biphthalic anhydride and 0.24 g of tetra-n-butylammonium bromide were added, and the mixture was stirred at 120 ° C. for 4 hours. Further, 20 g of tetrahydrophthalic anhydride was added and stirred at 120 ° C. for 4 hours, at 100 ° C. for 3 hours, at 80 ° C. for 4 hours, at 60 ° C. for 6 hours, and at 40 ° C. for 11 hours, and then propylene glycol-1-monomethyl ether Add 90 g of -2-acetate to obtain the target alkali developing resin No. 1 as a propylene glycol-1-monomethyl ether-2-acetate solution. 1 (Mw = 5000, Mn = 2100, acid value (solid content) 92.7 mgKOH / g) was obtained.
アルカリ現像性を付与する(B)成分として<ステップ1>で得られたアルカリ現像性樹脂No.1の14.7g、(B)成分としてジペンタエリスリトールペンタ及びヘキサアクリレート(アロニックスM-402;東亜合成(株)製)3.0g、界面活性剤FZ-2122(日本ユニカー(株)製)のシクロヘキサノン1%溶液1.8g、プロピレングリコール-1-モノメチルエーテル-2-アセテート10.0g、並びにシクロヘキサノン20.2gを混合し、(A)成分として実施例1で得られた化合物No.1の0.3gを添加してよく攪拌し、本発明の感光性組成物であるアルカリ現像性感光性樹脂組成物No.1を得た。 <Step 2> Alkali-developable photosensitive resin composition No. Preparation of Alkali Developable Resin No. 1 obtained in <Step 1> as component (B) imparting alkali developability 14.7 g of dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) as a component (B), surfactant FZ-2122 (manufactured by Nippon Unicar Co., Ltd.) 1.8 g of a cyclohexanone 1% solution, 10.0 g of propylene glycol-1-monomethyl ether-2-acetate and 20.2 g of cyclohexanone were mixed, and the compound No. 1 obtained in Example 1 as component (A) was mixed. 0.3 g of No. 1 was added and stirred well, and the alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention. 1 was obtained.
実施例1で得られた化合物No.1を実施例2で得られた化合物No.2に替えた以外は、実施例5の<ステップ2>と同様の手法で、本発明の感光性組成物であるアルカリ現像性感光性樹脂組成物No.2を得た。 [Example 6] Alkali-developable photosensitive resin composition No. Preparation of Compound No. 2 obtained in Example 1 1 to compound No. 1 obtained in Example 2. Except for changing to No. 2, in the same manner as in <Step 2> of Example 5, the alkali-developable photosensitive resin composition No. 1 as the photosensitive composition of the present invention was used. 2 was obtained.
実施例1で得られた化合物No.1を実施例3で得られた化合物No.3に替えた以外は、実施例5の<ステップ2>と同様の手法で、本発明の感光性組成物であるアルカリ現像性感光性樹脂組成物No.3を得た。 [Example 7] Alkali-developable photosensitive resin composition No. Preparation of Compound No. 3 obtained in Example 1 1 to compound No. 1 obtained in Example 3. 3 except that the alkaline developing photosensitive resin composition No. 1 which is the photosensitive composition of the present invention was prepared in the same manner as in <Step 2> of Example 5. 3 was obtained.
実施例1で得られた化合物No.1を2-ベンジル-2-ジメチルアミノ-1-(4-モルフォリノフェニル)-1-ブタノンに替えた以外は、実施例5の<ステップ2>と同様の手法で、比較品であるアルカリ現像性感光性樹脂組成物No.4を得た。 [Comparative Example 1] Alkali-developable photosensitive resin composition No. Preparation of Compound No. 4 obtained in Example 1 Alkaline development as a comparative product was performed in the same manner as in <Step 2> of Example 5 except that 1 was replaced with 2-benzyl-2-dimethylamino-1- (4-morpholinophenyl) -1-butanone. Photosensitive resin composition No. 4 was obtained.
実施例1で得られた化合物No.1をエタノン,1-[9-エチル-6-(2-メチルベンゾイル)-9H-カルバゾール-3-イル]-,1-(O-アセチルオキシム)に替えた以外は、実施例5の<ステップ2>と同様の手法で、比較品であるアルカリ現像性感光性樹脂組成物No.5を得た。 [Comparative Example 2] Alkali-developable photosensitive resin composition No. Production of Compound No. 5 obtained in Example 1 <Step of Example 5 except that 1 is replaced with ethanone, 1- [9-ethyl-6- (2-methylbenzoyl) -9H-carbazol-3-yl]-, 1- (O-acetyloxime) 2>, the comparative alkali-developable photosensitive resin composition No. 5 was obtained.
得られたアルカリ現像性感光性樹脂組成物No.1~No.5の透過性試験を以下のようにして行った。
即ち、ガラス基板上にアルカリ現像性感光性樹脂組成物をスピンコート(500rpmで2秒間、800rpmで6秒間)し、ホットプレートを用いて90℃で90秒間プリベークを行った。次いで、光源として高圧水銀ランプを用いて150mJ/cm2露光した後、オーブンを用いて230℃で30分間ポストベークを行って硬化膜を得た。得られた硬化膜について、吸光光度計を用いて380nmにおける透過率を測定した。結果を〔表4〕に示す。 <Transparency>
The obtained alkali-developable photosensitive resin composition No. 1-No. The permeability test of No. 5 was conducted as follows.
That is, the alkali-developable photosensitive resin composition was spin-coated on a glass substrate (500 rpm for 2 seconds, 800 rpm for 6 seconds), and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, after exposure to 150 mJ / cm 2 using a high-pressure mercury lamp as a light source, post-baking was performed at 230 ° C. for 30 minutes using an oven to obtain a cured film. About the obtained cured film, the transmittance | permeability in 380 nm was measured using the absorptiometer. The results are shown in [Table 4].
アルカリ現像性を付与する(B)成分として実施例5の<ステップ1>で得られたアルカリ現像性樹脂No.1の14.1g、(B)成分としてジペンタエリスリトールペンタ及びヘキサアクリレート(アロニックスM-402;東亜合成(株)製)7.1g、界面活性剤BYK-323(ビックケミー・ジャパン(株)製)0.05g、テキサノール(2,2,4-トリメチル-1,3-ペンタンジオールモノイソブチレート)11.3g、(A)成分として実施例1で得られた化合物No.1の0.7g、並びに(C)成分としてガラスフリット3.1g及び銀粉63.7gを混合して攪拌後、3本ロールミルにて混練し、本発明の感光性組成物である導電性組成物No.1を得た。 [Example 8] Conductive composition No. Production of Alkali Developable Resin No. 1 obtained in <Step 1> of Example 5 as component (B) for imparting alkali developability 14.1 g of 1, dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) as component (B), surfactant BYK-323 (manufactured by Big Chemie Japan Co., Ltd.) 0.05 g, 11.3 g of texanol (2,2,4-trimethyl-1,3-pentanediol monoisobutyrate), compound No. obtained in Example 1 as component (A) 0.7 g of 1 and 3.1 g of glass frit and 63.7 g of silver powder as component (C) were stirred and kneaded with a three-roll mill, and the conductive composition which is the photosensitive composition of the present invention. No. 1 was obtained.
<ステップ1>青色分散液の製造
青色顔料ピグメントブルー15:6の57.6g、分散剤アジスパーPB821(味の素ファインテクノ(株)製)20.0g、分散助剤ソルスパース5000(アビシア(株)製)2.4g、及びプロピレングリコール-1-モノメチルエーテル-2-アセテート320.0gを、500mlポリエチレン製容器に入れ、粒子径0.5mmのジルコニアビーズ350gを使用して、ペイントコンディショナーで10時間振とう後、ジルコニアビーズをろ別して青色分散液を得た。 [Example 9] Colored alkali-developable photosensitive resin composition No. <Step 1> Production of Blue Dispersion 57.6 g of Blue Pigment Pigment Blue 15: 6, Dispersant Azisper PB821 (manufactured by Ajinomoto Fine Techno Co., Ltd.) 20.0 g, Dispersing Agent Solsperse 5000 (Avicia Co., Ltd.) )) 2.4 g and propylene glycol-1-monomethyl ether-2-acetate 320.0 g are put into a 500 ml polyethylene container, and 350 g of zirconia beads having a particle diameter of 0.5 mm are used for 10 hours in a paint conditioner. After shaking, the zirconia beads were filtered off to obtain a blue dispersion.
(D)成分として<ステップ1>で得られた青色分散液の10.63g、アルカリ現像性を付与する(B)成分として実施例5の<ステップ1>で得られたアルカリ現像性樹脂No.1の2.98g、(B)成分としてジペンタエリスリトールペンタ及びヘキサアクリレート(アロニックスM-402;東亜合成(株)製)0.31g、(B)成分としてウレタンアクリレートUN3320HS(根上工業(株)製)0.31g、界面活性剤BYK-323(ビックケミー・ジャパン(株)製)のシクロヘキサノン1%溶液0.30g、プロピレングリコール-1-モノメチルエーテル-2-アセテート4.70g、並びにシクロヘキサノン10.36gを混合し、(A)成分として実施例1で得られた化合物No.1の0.41gを添加して攪拌し、本発明の感光性組成物である着色アルカリ現像性感光性樹脂組成物No.1を得た。 <Step 2> Colored alkali-developable photosensitive resin composition No. Production of 1 (D) 10.63 g of the blue dispersion obtained in <Step 1> as component (B) Alkali developability obtained in <Step 1> of Example 5 as component (B) that imparts alkali developability Resin No. 2.98 g of 1, dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) as component (B), urethane acrylate UN3320HS (produced by Negami Kogyo Co., Ltd.) as component (B) 0.31 g, 0.30 g of a cyclohexanone 1% solution of the surfactant BYK-323 (manufactured by BYK Japan KK), 4.70 g of propylene glycol-1-monomethyl ether-2-acetate, and 10.36 g of cyclohexanone The compound No. obtained in Example 1 as component (A) was mixed. No. 1 of 0.41 g was added and stirred, and the colored alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention. 1 was obtained.
実施例1で得られた化合物No.1をエタノン,1-[9-エチル-6-(2-メチル ベンゾイル)-9H-カルバゾール-3-イル]-,1-(O-アセチルオキシム)に替えた以外は実施例9の<ステップ2>と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.2を得た。 [Comparative Example 3] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 2 obtained in Example 1 <Step 2 of Example 9 except that 1 is replaced with ethanone, 1- [9-ethyl-6- (2-methylbenzoyl) -9H-carbazol-3-yl]-, 1- (O-acetyloxime) > In the same manner as above, the colored alkali-developable photosensitive resin composition No. 2 was obtained.
実施例1で得られた化合物No.1を2-メチル-1-(4-メチルチオフェニル)-2-モルフォリノプロパン-1-オンに替えた以外は実施例9の<ステップ2>と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.3を得た。 [Comparative Example 4] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 3 obtained in Example 1 Colored alkali development as a comparative product in the same manner as in <Step 2> of Example 9 except that 1 was replaced with 2-methyl-1- (4-methylthiophenyl) -2-morpholinopropan-1-one Photosensitive resin composition No. 3 was obtained.
実施例1で得られた化合物No.1を1,2-オクタンジオン,1-[4-(フェニルチオ)フェニル]-,2-(O-ベンゾイルオキシム)に替えた以外は実施例9の<ステップ2>と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.4を得た。 [Comparative Example 5] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 4 obtained in Example 1 A comparative product was prepared in the same manner as in <Step 2> of Example 9 except that 1 was replaced with 1,2-octanedione, 1- [4- (phenylthio) phenyl]-, 2- (O-benzoyloxime). Colored alkali-developable photosensitive resin composition No. 4 was obtained.
実施例1で得られた化合物No.1を2-ベンジル-2-ジメチルアミノ-1-(4-モルフォリノフェニル)-1-ブタノンに替えた以外は実施例9の<ステップ2>と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.5を得た。 [Comparative Example 6] Colored alkali-developable photosensitive resin composition No. Production of Compound No. 5 obtained in Example 1 Colored alkali development as a comparative product in the same manner as in <Step 2> of Example 9 except that 1 is replaced with 2-benzyl-2-dimethylamino-1- (4-morpholinophenyl) -1-butanone Photosensitive resin composition No. 5 was obtained.
得られた着色アルカリ現像性感光性樹脂組成物No.1~No.3の透過性試験を以下のようにして行った。
ガラス基板上に着色アルカリ現像性感光性樹脂組成物をスピンコート(500rpm、7秒間)し、ホットプレートを用いて90℃で90秒間プリベークを行った。次いで、光源として高圧水銀ランプを用いて150mJ/cm2露光した後、オーブンを用いて230℃で30分間ポストベークを行って硬化膜を得た。耐熱性試験として、得られた硬化膜を更に260℃で1時間加熱した。耐熱性試験後の硬化膜について、吸光光度計を用いて 420nmにおける透過率を測定した。結果を〔表5〕に示す。 <Transparency>
The obtained colored alkali-developable photosensitive resin composition No. 1-No. The permeability test of No. 3 was conducted as follows.
A colored alkali-developable photosensitive resin composition was spin-coated (500 rpm, 7 seconds) on a glass substrate, and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, after exposure to 150 mJ / cm 2 using a high-pressure mercury lamp as a light source, post-baking was performed at 230 ° C. for 30 minutes using an oven to obtain a cured film. As a heat resistance test, the obtained cured film was further heated at 260 ° C. for 1 hour. The cured film after the heat resistance test was measured for transmittance at 420 nm using an absorptiometer. The results are shown in [Table 5].
得られた着色アルカリ現像性感光性樹脂組成物No.1、No.3~No.5のアウトガス性試験として、以下のようにして硬化物の重量減少率の測定を行った。
即ち、ガラス基板上に着色アルカリ現像性感光性樹脂組成物をスピンコート(500rpm、7秒間)し、ホットプレートを用いて90℃で90秒間プリベークを行った。次いで、光源として高圧水銀ランプを用いて100mJ/cm2露光して硬化膜を得た。この硬化膜を約3mg削り取り、削り取った硬化膜を試料として用い、TG-DTA(セイコーインスツル(株)製)により、230℃で30分間保持した時の重量減少率を測定した。結果を〔表6〕に示す。 <Outgassing>
The obtained colored alkali-developable photosensitive resin composition No. 1, no. 3 to No. As an outgas test of No. 5, the weight reduction rate of the cured product was measured as follows.
That is, the colored alkali-developable photosensitive resin composition was spin coated (500 rpm, 7 seconds) on a glass substrate, and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, the film was exposed to 100 mJ / cm 2 using a high-pressure mercury lamp as a light source to obtain a cured film. About 3 mg of this cured film was scraped off, and the weight reduction rate when held at 230 ° C. for 30 minutes was measured with TG-DTA (manufactured by Seiko Instruments Inc.) using the scraped cured film as a sample. The results are shown in [Table 6].
(D)成分として実施例9の<ステップ1>で得られた青色分散液の10.63g、アルカリ現像性を付与する(B)成分として実施例5の<ステップ1>で得られたアルカリ現像性樹脂No.1の2.93g、(B)成分としてジペンタエリスリトールペンタ及びヘキサアクリレート(アロニックスM-402;東亞合成(株)製)0.30g、(B)成分としてウレタンアクリレートUN3320HS(根上工業(株)製)0.30g、増感剤DETX-S(日本化薬(株)製)0.05g、界面活性剤BYK-323(ビック ケミー・ジャパン(株)製)のシクロヘキサノン1%溶液0.30g、プロピレングリコール-1-モノメチルエーテル-2-アセテート4.72g、並びにシクロヘキサノン10.36gを混合し、(A)成分として実施例1で得られた化合物No.1の0.41gを添加してよく攪拌し、本発明の感光性組成物である着色アルカリ現像性感光性樹脂組成物No.6を得た。 [Example 10] Colored alkali-developable photosensitive resin composition No. 6. Preparation (6) As component (D) 10.63 g of the blue dispersion obtained in <Step 1> of Example 9, and as component (B) imparting alkali developability, obtained in <Step 1> of Example 5 Alkali developable resin No. 1.93 g of 1, dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) as component (B), urethane acrylate UN3320HS (produced by Negami Kogyo Co., Ltd.) as component (B) 0.30 g, Sensitizer DETX-S (Nippon Kayaku Co., Ltd.) 0.05 g, Surfactant BYK-323 (Bic Chemie Japan Co., Ltd.) cyclohexanone 1% solution 0.30 g, Propylene Glycol-1-monomethyl ether-2-acetate (4.72 g) and cyclohexanone (10.36 g) were mixed, and the compound No. 1 obtained in Example 1 as component (A) was mixed. 0.41 g of No. 1 was added and stirred well, and the colored alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention. 6 was obtained.
実施例1で得られた化合物No.1をエタノン,1-[9-エチル-6-(2-メチルベンゾイル)-9H-カルバゾール-3-イル]-,1-(O-アセチルオキシム)に替えた以外は実施例10と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.7を得た。 [Comparative Example 7] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 7 obtained in Example 1 The same procedure as in Example 10 except that 1 was replaced with ethanone, 1- [9-ethyl-6- (2-methylbenzoyl) -9H-carbazol-3-yl]-, 1- (O-acetyloxime) In comparison, the colored alkali-developable photosensitive resin composition No. 7 was obtained.
実施例1で得られた化合物No.1を2-メチル-1-(4-メチルチオフェニル)-2-モルフォリノプロパン-1-オンに替えた以外は実施例10と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.8を得た。 [Comparative Example 8] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 8 obtained in Example 1 Colored alkali-developable photosensitive resin composition as a comparative product in the same manner as in Example 10 except that 1 was replaced with 2-methyl-1- (4-methylthiophenyl) -2-morpholinopropan-1-one Item No. 8 was obtained.
実施例1で得られた化合物No.1を1,2-オクタンジオン,1-[4-(フェニルチオ)フェニル]-,2-(O-ベンゾイルオキシム)に替えた以外は実施例10と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.9を得た。 [Comparative Example 9] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 9 obtained in Example 1 Colored alkali development as a comparative product in the same manner as in Example 10 except that 1 is replaced with 1,2-octanedione, 1- [4- (phenylthio) phenyl]-, 2- (O-benzoyloxime) Photosensitive resin composition No. 9 was obtained.
実施例1で得られた化合物No.1を2-ベンジル-2-ジメチルアミノ-1-(4-モルフォリノフェニル)-1-ブタノンに替えた以外は実施例10と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.10を得た。 [Comparative Example 10] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 10 obtained in Example 1 Colored alkali-developable photosensitive resin composition as a comparative product in the same manner as in Example 10 except that 1 was replaced with 2-benzyl-2-dimethylamino-1- (4-morpholinophenyl) -1-butanone Item No. 10 was obtained.
得られた着色アルカリ現像性感光性樹脂組成物No.6~10のフォトリソグラフィー性の評価として、以下の手順における感度、密着性及び残渣について、それぞれ以下の方法により評価を行った。結果を〔表7〕に示す。
ガラス基板上に着色アルカリ現像性感光性樹脂組成物をスピンコート(500rpm、7秒間)し、ホットプレートを用いて90℃で90秒間プリベークを行った。次いで、光源として高圧水銀ランプを用いてマスクを介して所定量(50、100又は150mJ/cm2)露光した。現像液として2.5質量%炭酸ナトリウム水溶液を用い、スピン現像機で45秒間現像後、よく水洗し、次いでオーブンを用いて230℃で30分ポストベークを行い、パターンを定着させた。 <Photolithography>
The obtained colored alkali-developable photosensitive resin composition No. As photolithography evaluations of 6 to 10, sensitivity, adhesion and residue in the following procedures were evaluated by the following methods, respectively. The results are shown in [Table 7].
A colored alkali-developable photosensitive resin composition was spin-coated (500 rpm, 7 seconds) on a glass substrate, and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, a predetermined amount (50, 100 or 150 mJ / cm 2 ) of light was exposed through a mask using a high pressure mercury lamp as a light source. A 2.5 mass% sodium carbonate aqueous solution was used as a developing solution, developed with a spin developing machine for 45 seconds, washed thoroughly with water, and then post-baked at 230 ° C. for 30 minutes using an oven to fix the pattern.
着色アルカリ現像性感光性樹脂組成物の感度を、次の4段階で評価した。
即ち、形成されたパターンの線幅が、マスク開口線幅を超えた時の露光量が50mJ/cm2であった場合をa、100mJ/cm2であった場合をb、150mJ/cm2であった場合をc、150mJ/cm2でもパターンが形成されなかった場合をdとした。
(密着性)
着色アルカリ現像性感光性樹脂組成物の密着性を、次の4段階で評価した。
即ち、100mJ/cm2で露光、現像した後に残ったパターンの最も細かいマスク線 幅が3μm以下であった場合をA、3μm超10μm以下であった場合をB、10μm超15μm以下であった場合をC、15μm超であった場合をDとした。
(残渣)
現像後、非露光部のガラス面に残渣が全く観察されなかった場合を○、一面に残渣が観察された場合を×とした。 (sensitivity)
The sensitivity of the colored alkali-developable photosensitive resin composition was evaluated in the following four stages.
That is, the exposure amount when the line width of the formed pattern exceeds the mask opening line width is 50 mJ / cm 2 , a is 100 mJ / cm 2 , b is 150 mJ / cm 2 The case where there was c and the case where a pattern was not formed even at 150 mJ / cm 2 was taken as d.
(Adhesion)
The adhesion of the colored alkali-developable photosensitive resin composition was evaluated in the following four stages.
That is, when the finest mask line width of the pattern remaining after exposure and development at 100 mJ / cm 2 is 3 μm or less, A is more than 3 μm and 10 μm or less, and B is more than 10 μm and 15 μm or less. Is C, and D is the case when it exceeds 15 μm.
(Residue)
After development, the case where no residue was observed on the glass surface of the non-exposed part was marked with ◯, and the case where the residue was observed on one side was marked with x.
(D)成分として実施例9の<ステップ1>におけるピグメントブルー15:6をカーボンブラックに変更して作製したカーボンブラック分散液の7.06g、アルカリ現像性を付与する(B)成分として実施例5の<ステップ1>で得られたアルカリ現像性樹脂No.1の3.07g、(B)成分としてジペンタエリスリトールペンタ及びヘキサアクリレート(アロニックスM-402;東亜合成(株)製)0.53g、界面活性剤BYK-323(ビックケミー・ジャパン(株)製)のシクロヘキサノン1%溶液0.30g、プロピレングリコール-1-モノメチルエーテル-2-アセテート8.16g、並びにシクロヘキサノン10.69gを混合し、(A)成分として実施例1で得られた化合物No.1の0.19gを添加してよく攪拌し、本発明の感光性組成物である着色アルカリ現像性感光性樹脂組成物No.11を得た。 Example 11 Colored alkali-developable photosensitive resin composition No. 11: (D) 7.06 g of a carbon black dispersion prepared by changing Pigment Blue 15: 6 in <Step 1> of Example 9 to carbon black as component (D), component (B) imparting alkali developability As the alkali developable resin No. obtained in <Step 1> of Example 5. 3.07 g of component 1, dipentaerythritol penta and hexaacrylate (Aronix M-402; manufactured by Toagosei Co., Ltd.) 0.53 g, surfactant BYK-323 (produced by Big Chemie Japan Co., Ltd.) A mixture of 0.30 g of a 1% solution of cyclohexanone, 8.16 g of propylene glycol-1-monomethyl ether-2-acetate, and 10.69 g of cyclohexanone was mixed and compound No. 1 obtained in Example 1 as component (A) was mixed. 0.19 g of No. 1 was added and stirred well, and the colored alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention. 11 was obtained.
実施例1で得られた化合物No.1を実施例2で得られた化合物No.2に替えた以外は実施例11と同様の手法で、本発明の感光性組成物である着色アルカリ現像性感光性樹脂組成物No.12を得た。 [Example 12] Colored alkali-developable photosensitive resin composition No. No. 12 Compound No. obtained in Example 1 1 to compound No. 1 obtained in Example 2. A colored alkali-developable photosensitive resin composition No. 1 which is the photosensitive composition of the present invention was prepared in the same manner as in Example 11 except that it was changed to 2. 12 was obtained.
実施例1で得られた化合物No.1を実施例3で得られた化合物No.3に替えた以外は実施例11と同様の手法で、本発明の感光性組成物である着色アルカリ現像性感光性樹脂組成物No.13を得た。 [Example 13] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 13 obtained in Example 1 1 to compound No. 1 obtained in Example 3. Except for the change to 3 in the same manner as in Example 11, the colored alkali-developable photosensitive resin composition No. 1 as the photosensitive composition of the present invention was used. 13 was obtained.
実施例1で得られた化合物No.1をエタノン,1-[9-エチル-6-(2-メチル ベンゾイル)-9H-カルバゾール-3-イル]-,1-(O-アセチルオキシム)に替えた以外は実施例11と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.14を得た。 [Comparative Example 11] Colored alkali-developable photosensitive resin composition No. Preparation of Compound No. 14 obtained in Example 1 The same procedure as in Example 11 except that 1 is replaced with ethanone, 1- [9-ethyl-6- (2-methylbenzoyl) -9H-carbazol-3-yl]-, 1- (O-acetyloxime) In comparison, the colored alkali-developable photosensitive resin composition No. 14 was obtained.
実施例1で得られた化合物No.1を1,2-オクタンジオン,1-[4-(フェニルチオ)フェニル]-,2-(O-ベンゾイルオキシム)に替えた以外は実施例11と同様の手法で、比較品である着色アルカリ現像性感光性樹脂組成物No.15を得た。 [Comparative Example 12] Colored alkali-developable photosensitive resin composition No. No. 15 Compound No. obtained in Example 1 Colored alkali development as a comparative product in the same manner as in Example 11 except that 1 was replaced with 1,2-octanedione, 1- [4- (phenylthio) phenyl]-, 2- (O-benzoyloxime) Photosensitive resin composition No. 15 was obtained.
得られた着色アルカリ現像性感光性樹脂組成物No.11~15のフォトリソグラフィー性の評価として、以下の手順における感度、密着性及び現像性について、それぞれ以下の方法により評価を行った。結果を〔表8〕に示す。
ガラス基板上に着色アルカリ現像性感光性樹脂組成物をスピンコート(500rpm、7秒間)し、ホットプレートを用いて90℃で90秒間プリベークを行った。次いで、光源として高圧水銀ランプを用いてマスクを介して所定量(100又は120mJ/cm2)露光した。現像液として2.5質量%炭酸ナトリウム水溶液を用い、スピン現像機で45秒間現像後、よく水洗し、次いでオーブンを用いて230℃で30分ポストベークを行い、パターンを定着させた。 <Photolithography>
The obtained colored alkali-developable photosensitive resin composition No. As evaluations of photolithographic properties 11 to 15, the sensitivity, adhesion and developability in the following procedures were evaluated by the following methods, respectively. The results are shown in [Table 8].
A colored alkali-developable photosensitive resin composition was spin-coated (500 rpm, 7 seconds) on a glass substrate, and prebaked at 90 ° C. for 90 seconds using a hot plate. Next, a predetermined amount (100 or 120 mJ / cm 2 ) of light was exposed through a mask using a high-pressure mercury lamp as a light source. A 2.5 mass% sodium carbonate aqueous solution was used as a developing solution, developed with a spin developing machine for 45 seconds, washed thoroughly with water, and then post-baked at 230 ° C. for 30 minutes using an oven to fix the pattern.
着色アルカリ現像性感光性樹脂組成物の感度を、次の3段階で評価した。
即ち、形成されたパターンの線幅が、マスク開口線幅を超えた時の露光量が100mJ/cm2であった場合をa、120mJ/cm2であった場合をb、120mJ/cm2でもパターンが形成されなかった場合をcとした。
(密着性)
着色アルカリ現像性感光性樹脂組成物の密着性を、次の4段階で評価した。
即ち、100mJ/cm2で露光、現像した後に残ったパターンの最も細かいマスク線幅が3μm以下であった場合をA、3μm超5μm以下であった場合をB、5μm超であった場合をCとした。
(現像性)
現像後、非露光部のガラス面に残渣が全く観察されなかった場合を○、一面に残渣が観察された場合を×とした。 (sensitivity)
The sensitivity of the colored alkali-developable photosensitive resin composition was evaluated in the following three stages.
That is, when the line width of the formed pattern exceeds the mask opening line width, the exposure amount is 100 mJ / cm 2 , a is 120 mJ / cm 2 , b is 120 mJ / cm 2 The case where a pattern was not formed was defined as c.
(Adhesion)
The adhesion of the colored alkali-developable photosensitive resin composition was evaluated in the following four stages.
That is, when the finest mask line width of the pattern remaining after exposure and development at 100 mJ / cm 2 is 3 μm or less, A is more than 3 μm and 5 μm or less, B is more than 5 μm, and C is more than 5 μm. It was.
(Developability)
After development, the case where no residue was observed on the glass surface of the non-exposed part was marked with ◯, and the case where the residue was observed on one side was marked with x.
本発明のオキシムエステル化合物を用いると、比較例2、3、7及び11で用いた化合物と比較して、可視光領域の透過性及び現像性に優れる。
また、本発明のオキシムエステル化合物を用いると、比較例4及び8で用いた化合物と比較して、可視光領域の透過性、耐熱性、感度及び密着性に優れる。
また、本発明のオキシムエステル化合物を用いると、比較例5、9及び12で用いた化合物と比較して、耐熱性、感度及び密着性に優れる。
また、本発明のオキシムエステル化合物を用いると、比較例1、6及び10で用いた化合物と比較して、可視光領域の透過性、耐熱性、感度及び密着性に優れる。 From the above results, the following can be understood.
When the oxime ester compound of the present invention is used, the transparency and developability in the visible light region are excellent as compared with the compounds used in Comparative Examples 2, 3, 7, and 11.
In addition, when the oxime ester compound of the present invention is used, it is excellent in transparency, heat resistance, sensitivity, and adhesion in the visible light region as compared with the compounds used in Comparative Examples 4 and 8.
Moreover, when the oxime ester compound of this invention is used, it is excellent in heat resistance, a sensitivity, and adhesiveness compared with the compound used in Comparative Examples 5, 9, and 12.
In addition, when the oxime ester compound of the present invention is used, it is excellent in transparency, heat resistance, sensitivity and adhesion in the visible light region as compared with the compounds used in Comparative Examples 1, 6 and 10.
Claims (8)
- 下記一般式(1)で表されるオキシムエステル化合物。
R11、R12及びR13は、それぞれ独立に、水素原子、又は炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基、炭素原子数7~30のアリールアルキル基若しくは炭素原子数2~20の複素環基である置換基を表し、
R11、R12及びR13で表わされる置換基の水素原子は、更にOR21、COR21、SR21、NR22R23、CONR22R23、-NR22-OR23、-NR22CO-OCOR23、-C(=N-OR21)-R22、-C(=N-OCOR21)-R22、CN、ハロゲン原子、-COOR21、-CR21=CR22R23、-CO-CR21=CR22R23、-O-CO-CR21=CR22R23、-N=C=O又はエポキシ基で置換されていてもよく、
R21、R22及びR23は、それぞれ独立に、水素原子、又は炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基、炭素原子数7~30のアリールアルキル基若しくは炭素原子数2~20の複素環基である置換基を表し、
R21、R22及びR23で表される置換基の水素原子は、更にCN、ハロゲン原子、水酸基又はカルボキシル基で置換されていてもよく、
R11、R12、R13、R21、R22及びR23で表される置換基のアルキレン部分は、-O-、-S-、-COO-、-OCO-、-NR24-、-NR24COO-、-OCONR24-、-SCO-、-COS-、-OCS-又は-CSO-により1~5回中断されていてもよく、
R24は、水素原子、炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基、炭素原子数7~30のアリールアルキル基又は炭素原子数2~20の複素環基を表し、
R11、R12、R13、R21、R22及びR23で表される置換基のアルキル部分は、分岐側鎖があってもよく、環状アルキルであってもよく、また、R12とR13及びR22とR23はそれぞれ一緒になって環を形成していてもよく、
Xは、酸素原子、硫黄原子、セレン原子、CR31R32、CO、NR33又はPR34を表し、
R31、R32、R33及びR34は、それぞれ独立に、水素原子、又は炭素原子数1~20のアルキル基、炭素原子数6~30のアリール基若しくは炭素原子数7~30のアリールアルキル基である置換基を表し、
R31、R32、R33及びR34で表される置換基のアルキル部分は、分岐側鎖があってもよく、環状アルキルであってもよく、R31、R32、R33及びR34で表される置換基のアルキル末端は不飽和結合であってもよく、R31、R32、R33及びR34は、それぞれ独立に、隣接するどちらかのベンゼン環と一緒になって環を形成していてもよく、
aは、0~4の整数を表し、
bは、1~5の整数を表し、
R4の少なくとも一つは、-CR21=CR22R23、-CO-CR21=CR22R23、-O-CO-CR21=CR22R23、-N=C=O又はエポキシ基を有する基である。) An oxime ester compound represented by the following general formula (1).
R 11 , R 12 and R 13 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms or a carbon atom. Represents a substituent which is a heterocyclic group having 2 to 20 atoms;
The hydrogen atom of the substituent represented by R 11 , R 12 and R 13 is further OR 21 , COR 21 , SR 21 , NR 22 R 23 , CONR 22 R 23 , —NR 22 —OR 23 , —NR 22 CO—. OCOR 23 , —C (═N—OR 21 ) —R 22 , —C (═N—OCOR 21 ) —R 22 , CN, halogen atom, —COOR 21 , —CR 21 ═CR 22 R 23 , —CO— CR 21 ═CR 22 R 23 , —O—CO—CR 21 ═CR 22 R 23 , —N═C═O or an epoxy group may be substituted,
R 21 , R 22 and R 23 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms or a carbon atom. Represents a substituent which is a heterocyclic group having 2 to 20 atoms;
The hydrogen atom of the substituent represented by R 21 , R 22 and R 23 may be further substituted with CN, a halogen atom, a hydroxyl group or a carboxyl group,
The alkylene part of the substituent represented by R 11 , R 12 , R 13 , R 21 , R 22 and R 23 is —O—, —S—, —COO—, —OCO—, —NR 24 —, — May be interrupted 1 to 5 times by NR 24 COO—, —OCONR 24 —, —SCO—, —COS—, —OCS— or —CSO—,
R 24 represents a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms, an arylalkyl group having 7 to 30 carbon atoms, or a heterocyclic group having 2 to 20 carbon atoms. ,
Alkyl moiety of the substituents represented by R 11, R 12, R 13 , R 21, R 22 and R 23, there may be branched side chain, may be a cyclic alkyl, and the R 12 R 13 and R 22 and R 23 may be combined to form a ring,
X represents an oxygen atom, a sulfur atom, a selenium atom, CR 31 R 32 , CO, NR 33 or PR 34 ;
R 31 , R 32 , R 33 and R 34 are each independently a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 30 carbon atoms or an arylalkyl having 7 to 30 carbon atoms. Represents a substituent that is a group,
The alkyl part of the substituent represented by R 31 , R 32 , R 33 and R 34 may have a branched side chain or may be a cyclic alkyl. R 31 , R 32 , R 33 and R 34 The alkyl terminal of the substituent represented by the above formula may be an unsaturated bond, and R 31 , R 32 , R 33 and R 34 each independently form a ring together with one of the adjacent benzene rings. May be formed,
a represents an integer of 0 to 4,
b represents an integer of 1 to 5,
At least one of R 4 is —CR 21 ═CR 22 R 23 , —CO—CR 21 ═CR 22 R 23 , —O—CO—CR 21 ═CR 22 R 23 , —N═C═O or an epoxy group It is group which has. ) - 請求項1に記載のオキシムエステル化合物を含有してなる光重合開始剤。 A photopolymerization initiator comprising the oxime ester compound according to claim 1.
- (A)請求項2に記載の光重合開始剤、及び(B)エチレン性不飽和結合を有する重合性化合物を含有してなる感光性組成物。 (A) A photosensitive composition comprising the photopolymerization initiator according to claim 2 and (B) a polymerizable compound having an ethylenically unsaturated bond.
- 上記(B)エチレン性不飽和結合を有する重合性化合物の少なくとも一つが、アルカリ現像性を付与する化合物である請求項3に記載の感光性組成物。 The photosensitive composition according to claim 3, wherein at least one of the polymerizable compound (B) having an ethylenically unsaturated bond is a compound imparting alkali developability.
- 更に(C)無機化合物を含有してなる請求項3又は4に記載の感光性組成物。 The photosensitive composition according to claim 3 or 4, further comprising (C) an inorganic compound.
- 更に(D)色材を含有してなる請求項3~5の何れか一項に記載の感光性組成物。 The photosensitive composition according to any one of claims 3 to 5, further comprising (D) a coloring material.
- 請求項3~6の何れか一項に記載の感光性組成物にエネルギー線を照射してなる硬化物。 A cured product obtained by irradiating the photosensitive composition according to any one of claims 3 to 6 with an energy ray.
- 請求項7に記載の硬化物を用いたカラーフィルタ。 A color filter using the cured product according to claim 7.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020137016528A KR101930099B1 (en) | 2011-03-25 | 2012-02-07 | Oxime ester compound and photoinitiator containing said compound |
| JP2013507233A JP6000942B2 (en) | 2011-03-25 | 2012-02-07 | Oxime ester compound and photopolymerization initiator containing the compound |
| CN201280004665.9A CN103298782B (en) | 2011-03-25 | 2012-02-07 | Oxime ester compound and the Photoepolymerizationinitiater initiater containing this compound |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011067406 | 2011-03-25 | ||
| JP2011-067406 | 2011-03-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012132558A1 true WO2012132558A1 (en) | 2012-10-04 |
Family
ID=46930324
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2012/052775 WO2012132558A1 (en) | 2011-03-25 | 2012-02-07 | Oxime ester compound and photoinitiator containing said compound |
Country Status (5)
| Country | Link |
|---|---|
| JP (1) | JP6000942B2 (en) |
| KR (1) | KR101930099B1 (en) |
| CN (1) | CN103298782B (en) |
| TW (1) | TWI541221B (en) |
| WO (1) | WO2012132558A1 (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5337293B1 (en) * | 2012-11-13 | 2013-11-06 | 積水化学工業株式会社 | Sealant for liquid crystal display element, vertical conduction material, and liquid crystal display element |
| CN104395824A (en) * | 2012-11-01 | 2015-03-04 | 株式会社艾迪科 | Alkali developable photosensitive composition |
| WO2015029846A1 (en) * | 2013-08-29 | 2015-03-05 | 積水化学工業株式会社 | Sealant for display element, and oxime ester initiator |
| JP2017078049A (en) * | 2015-10-22 | 2017-04-27 | 株式会社Adeka | Oxime ester compound and polymerization initiator comprising the compound |
| JP2017132863A (en) * | 2016-01-26 | 2017-08-03 | 積水化学工業株式会社 | Oxime ester initiator, curable resin composition, sealant for liquid crystal display elements, vertical conduction material, and liquid crystal display element |
| JP2019519518A (en) * | 2016-05-19 | 2019-07-11 | サムヤン コーポレイション | Oxime ester derivative compound, photoinitiator containing the same, and photosensitive composition |
| WO2019168113A1 (en) * | 2018-03-01 | 2019-09-06 | 日本化薬株式会社 | Novel compound, photopolymerization initiator containing said compound, and photosensitive resin composition containing said photopolymerization initiator |
| US10921708B2 (en) | 2015-12-08 | 2021-02-16 | Fujifilm Corporation | Radiation-sensitive resin composition, cured film, pattern forming method, solid-state imaging device, and image display device |
| WO2023085058A1 (en) | 2021-11-09 | 2023-05-19 | 住友化学株式会社 | Resin composition, resin film, and display device |
| WO2023085061A1 (en) | 2021-11-09 | 2023-05-19 | 住友化学株式会社 | Resin film and display device |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6788971B2 (en) * | 2016-01-14 | 2020-11-25 | 東京応化工業株式会社 | Photosensitive composition |
| WO2017169819A1 (en) * | 2016-03-29 | 2017-10-05 | 株式会社Adeka | Black photosensitive resin composition |
| WO2017200354A1 (en) * | 2016-05-19 | 2017-11-23 | 주식회사 삼양사 | Oxime ester derivative compound, photopolymerization initiator comprising same, and photosensitive composition |
| TWI774931B (en) * | 2018-03-02 | 2022-08-21 | 日商日本化藥股份有限公司 | Novel compound, photopolymerization initiator comprising the compound, and photosensitive resin composition containing the photopolymerization initiator |
| CN114127637A (en) * | 2019-07-12 | 2022-03-01 | 株式会社三养社 | Photosensitive resin composition |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007310317A (en) * | 2006-05-22 | 2007-11-29 | Fujifilm Corp | Photopolymerization composition, color filter, and manufacturing method therefor |
| JP2009047871A (en) * | 2007-08-20 | 2009-03-05 | Fujifilm Corp | Composition for holographic recording, holographic recording medium, information recording method and new compound |
| JP2009242469A (en) * | 2008-03-28 | 2009-10-22 | Fujifilm Corp | Polymerizable composition, color filter, method for manufacturing color filter, and solid imaging element |
| JP2011099974A (en) * | 2009-11-05 | 2011-05-19 | Fujifilm Corp | Colored photosensitive composition, color filter, and liquid crystal display device |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SG77689A1 (en) * | 1998-06-26 | 2001-01-16 | Ciba Sc Holding Ag | New o-acyloxime photoinitiators |
| JP5179379B2 (en) * | 2006-12-27 | 2013-04-10 | 株式会社Adeka | Oxime ester compound and photopolymerization initiator containing the compound |
| JP4818458B2 (en) * | 2009-11-27 | 2011-11-16 | 株式会社Adeka | Oxime ester compound and photopolymerization initiator containing the compound |
-
2012
- 2012-02-07 JP JP2013507233A patent/JP6000942B2/en active Active
- 2012-02-07 CN CN201280004665.9A patent/CN103298782B/en active Active
- 2012-02-07 WO PCT/JP2012/052775 patent/WO2012132558A1/en active Application Filing
- 2012-02-07 KR KR1020137016528A patent/KR101930099B1/en active IP Right Grant
- 2012-02-15 TW TW101104967A patent/TWI541221B/en active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007310317A (en) * | 2006-05-22 | 2007-11-29 | Fujifilm Corp | Photopolymerization composition, color filter, and manufacturing method therefor |
| JP2009047871A (en) * | 2007-08-20 | 2009-03-05 | Fujifilm Corp | Composition for holographic recording, holographic recording medium, information recording method and new compound |
| JP2009242469A (en) * | 2008-03-28 | 2009-10-22 | Fujifilm Corp | Polymerizable composition, color filter, method for manufacturing color filter, and solid imaging element |
| JP2011099974A (en) * | 2009-11-05 | 2011-05-19 | Fujifilm Corp | Colored photosensitive composition, color filter, and liquid crystal display device |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104395824A (en) * | 2012-11-01 | 2015-03-04 | 株式会社艾迪科 | Alkali developable photosensitive composition |
| TWI619760B (en) * | 2012-11-01 | 2018-04-01 | Adeka Corp | Alkali developed photosensitive composition |
| JP5337293B1 (en) * | 2012-11-13 | 2013-11-06 | 積水化学工業株式会社 | Sealant for liquid crystal display element, vertical conduction material, and liquid crystal display element |
| WO2015029846A1 (en) * | 2013-08-29 | 2015-03-05 | 積水化学工業株式会社 | Sealant for display element, and oxime ester initiator |
| JP2017078049A (en) * | 2015-10-22 | 2017-04-27 | 株式会社Adeka | Oxime ester compound and polymerization initiator comprising the compound |
| TWI731895B (en) * | 2015-12-08 | 2021-07-01 | 日商富士軟片股份有限公司 | Radiation-sensitive resin composition, cured film, pattern forming method, solid-state imaging device, and image display device |
| US10921708B2 (en) | 2015-12-08 | 2021-02-16 | Fujifilm Corporation | Radiation-sensitive resin composition, cured film, pattern forming method, solid-state imaging device, and image display device |
| JP2017132863A (en) * | 2016-01-26 | 2017-08-03 | 積水化学工業株式会社 | Oxime ester initiator, curable resin composition, sealant for liquid crystal display elements, vertical conduction material, and liquid crystal display element |
| JP2021011486A (en) * | 2016-05-19 | 2021-02-04 | サムヤン コーポレイション | Oxime ester derivative compounds, and photopolymerization initiator and photosensitive composition containing the same |
| JP2019519518A (en) * | 2016-05-19 | 2019-07-11 | サムヤン コーポレイション | Oxime ester derivative compound, photoinitiator containing the same, and photosensitive composition |
| JP7002617B2 (en) | 2016-05-19 | 2022-01-20 | サムヤン コーポレイション | Oxime ester derivative compound, photopolymerization initiator and photosensitive composition containing the same. |
| WO2019168113A1 (en) * | 2018-03-01 | 2019-09-06 | 日本化薬株式会社 | Novel compound, photopolymerization initiator containing said compound, and photosensitive resin composition containing said photopolymerization initiator |
| JPWO2019168113A1 (en) * | 2018-03-01 | 2021-02-25 | 日本化薬株式会社 | A novel compound, a photopolymerization initiator containing the compound, and a photosensitive resin composition containing the photopolymerization initiator. |
| JP7232241B2 (en) | 2018-03-01 | 2023-03-02 | 日本化薬株式会社 | Novel compound, photopolymerization initiator comprising said compound, and photosensitive resin composition containing said photopolymerization initiator |
| US11608316B2 (en) | 2018-03-01 | 2023-03-21 | Nippon Kayaku Kabushiki Kaisha | Compound, photopolymerization initiator containing said compound, and photosensitive resin composition containing said photopolymerization initiator |
| WO2023085058A1 (en) | 2021-11-09 | 2023-05-19 | 住友化学株式会社 | Resin composition, resin film, and display device |
| WO2023085061A1 (en) | 2021-11-09 | 2023-05-19 | 住友化学株式会社 | Resin film and display device |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6000942B2 (en) | 2016-10-05 |
| CN103298782B (en) | 2016-04-06 |
| CN103298782A (en) | 2013-09-11 |
| KR101930099B1 (en) | 2018-12-17 |
| TWI541221B (en) | 2016-07-11 |
| KR20130140096A (en) | 2013-12-23 |
| JPWO2012132558A1 (en) | 2014-07-24 |
| TW201242931A (en) | 2012-11-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6000942B2 (en) | Oxime ester compound and photopolymerization initiator containing the compound | |
| JP4818458B2 (en) | Oxime ester compound and photopolymerization initiator containing the compound | |
| JP6595983B2 (en) | Oxime ester compound and photopolymerization initiator containing the compound | |
| JP6301249B2 (en) | Photosensitive composition | |
| KR102160115B1 (en) | Photo-curable composition | |
| JP2015007729A (en) | Photocurable black composition | |
| JP6572213B2 (en) | Novel polymerization initiator and radically polymerizable composition containing the polymerization initiator | |
| JP6457719B2 (en) | Photocurable composition | |
| JP2019168654A (en) | Photosensitive composition | |
| JP2015093842A (en) | Oxime ester compound and photopolymerization initiator containing the compound | |
| WO2020241529A1 (en) | Carbamoyl oxime compound, and polymerization initiator and polymerizable composition containing said compound | |
| JP6530902B2 (en) | Novel polymerization initiator and radically polymerizable composition containing the polymerization initiator | |
| KR102545326B1 (en) | Oxime ester compound and photopolymerization initiator containing the compound | |
| JP6539476B2 (en) | Oxime ester compound and photopolymerization initiator containing the compound | |
| JP2017179211A (en) | Curable composition, alkali development curable composition, cured article and method for manufacturing cured article | |
| JP2016079157A (en) | New polymerization initiator and radical polymerizable composition containing polymerization initiator | |
| JP6778108B2 (en) | Photosensitive composition | |
| JP2017124998A (en) | Compound, composition and photopolymerization initiator |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201280004665.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12763061 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2013507233 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20137016528 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12763061 Country of ref document: EP Kind code of ref document: A1 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013018908 Country of ref document: BR |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: 112013018908 Country of ref document: BR |
|
| ENPW | Started to enter national phase and was withdrawn or failed for other reasons |
Ref document number: 112013018908 Country of ref document: BR |