WO2012071261A1 - Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same - Google Patents
Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same Download PDFInfo
- Publication number
- WO2012071261A1 WO2012071261A1 PCT/US2011/061349 US2011061349W WO2012071261A1 WO 2012071261 A1 WO2012071261 A1 WO 2012071261A1 US 2011061349 W US2011061349 W US 2011061349W WO 2012071261 A1 WO2012071261 A1 WO 2012071261A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- polyamine
- mixture
- active liquid
- liquid
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 221
- 239000007788 liquid Substances 0.000 title claims abstract description 148
- 238000000034 method Methods 0.000 title claims abstract description 46
- 239000011159 matrix material Substances 0.000 title claims abstract description 40
- 229920000768 polyamine Polymers 0.000 claims description 116
- 239000003205 fragrance Substances 0.000 claims description 107
- 150000001412 amines Chemical class 0.000 claims description 63
- 150000001875 compounds Chemical class 0.000 claims description 54
- -1 aromatic isocyanate compound Chemical class 0.000 claims description 39
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 39
- 229910001868 water Inorganic materials 0.000 claims description 39
- 239000004952 Polyamide Substances 0.000 claims description 38
- 229920002647 polyamide Polymers 0.000 claims description 38
- 239000002253 acid Substances 0.000 claims description 33
- 239000000463 material Substances 0.000 claims description 25
- 239000012948 isocyanate Substances 0.000 claims description 24
- 125000000524 functional group Chemical group 0.000 claims description 23
- 239000002245 particle Substances 0.000 claims description 22
- 239000006185 dispersion Substances 0.000 claims description 21
- 150000003335 secondary amines Chemical class 0.000 claims description 21
- 150000008064 anhydrides Chemical group 0.000 claims description 13
- 150000004982 aromatic amines Chemical class 0.000 claims description 13
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 claims description 12
- 125000003277 amino group Chemical group 0.000 claims description 10
- 239000007795 chemical reaction product Substances 0.000 claims description 9
- 125000003700 epoxy group Chemical group 0.000 claims description 9
- NIXOWILDQLNWCW-UHFFFAOYSA-M acrylate group Chemical group C(C=C)(=O)[O-] NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims description 8
- RWZYAGGXGHYGMB-UHFFFAOYSA-N anthranilic acid Chemical compound NC1=CC=CC=C1C(O)=O RWZYAGGXGHYGMB-UHFFFAOYSA-N 0.000 claims description 8
- 239000002417 nutraceutical Substances 0.000 claims description 4
- 235000021436 nutraceutical agent Nutrition 0.000 claims description 4
- 239000002537 cosmetic Substances 0.000 claims description 3
- 230000000361 pesticidal effect Effects 0.000 claims description 3
- 230000001225 therapeutic effect Effects 0.000 claims description 3
- 239000003921 oil Substances 0.000 description 69
- 235000019198 oils Nutrition 0.000 description 69
- 238000002156 mixing Methods 0.000 description 62
- 229940060184 oil ingredients Drugs 0.000 description 52
- 239000000243 solution Substances 0.000 description 49
- 239000002386 air freshener Substances 0.000 description 41
- 238000006243 chemical reaction Methods 0.000 description 38
- 239000011521 glass Substances 0.000 description 37
- 239000000126 substance Substances 0.000 description 37
- 150000001299 aldehydes Chemical class 0.000 description 30
- 239000000047 product Substances 0.000 description 29
- 239000007787 solid Substances 0.000 description 23
- 239000000499 gel Substances 0.000 description 21
- 150000002513 isocyanates Chemical class 0.000 description 21
- 238000002360 preparation method Methods 0.000 description 21
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 20
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 20
- 239000000575 pesticide Substances 0.000 description 20
- 238000003756 stirring Methods 0.000 description 20
- 239000004094 surface-active agent Substances 0.000 description 17
- 239000008240 homogeneous mixture Substances 0.000 description 16
- 229920000642 polymer Polymers 0.000 description 15
- 239000004593 Epoxy Substances 0.000 description 14
- 239000003085 diluting agent Substances 0.000 description 13
- 229920001296 polysiloxane Polymers 0.000 description 13
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 12
- WTFAGPBUAGFMQX-UHFFFAOYSA-N 1-[2-[2-(2-aminopropoxy)propoxy]propoxy]propan-2-amine Chemical compound CC(N)COCC(C)OCC(C)OCC(C)N WTFAGPBUAGFMQX-UHFFFAOYSA-N 0.000 description 11
- 241000196324 Embryophyta Species 0.000 description 11
- 241000238631 Hexapoda Species 0.000 description 11
- 239000000539 dimer Substances 0.000 description 11
- ALYNCZNDIQEVRV-UHFFFAOYSA-N 4-aminobenzoic acid Chemical compound NC1=CC=C(C(O)=O)C=C1 ALYNCZNDIQEVRV-UHFFFAOYSA-N 0.000 description 10
- 239000009261 D 400 Substances 0.000 description 10
- 125000003118 aryl group Chemical group 0.000 description 10
- 229910052757 nitrogen Inorganic materials 0.000 description 10
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 10
- 229920003023 plastic Polymers 0.000 description 10
- 239000004033 plastic Substances 0.000 description 10
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 9
- 239000004480 active ingredient Substances 0.000 description 9
- 238000009826 distribution Methods 0.000 description 9
- 238000010438 heat treatment Methods 0.000 description 9
- 125000001931 aliphatic group Chemical group 0.000 description 8
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 7
- 241000167854 Bourreria succulenta Species 0.000 description 7
- 241001465754 Metazoa Species 0.000 description 7
- 241001442654 Percnon planissimum Species 0.000 description 7
- 239000004698 Polyethylene Substances 0.000 description 7
- 239000004721 Polyphenylene oxide Substances 0.000 description 7
- 241000607479 Yersinia pestis Species 0.000 description 7
- 239000001045 blue dye Substances 0.000 description 7
- 229920002678 cellulose Polymers 0.000 description 7
- 239000001913 cellulose Substances 0.000 description 7
- 239000000919 ceramic Substances 0.000 description 7
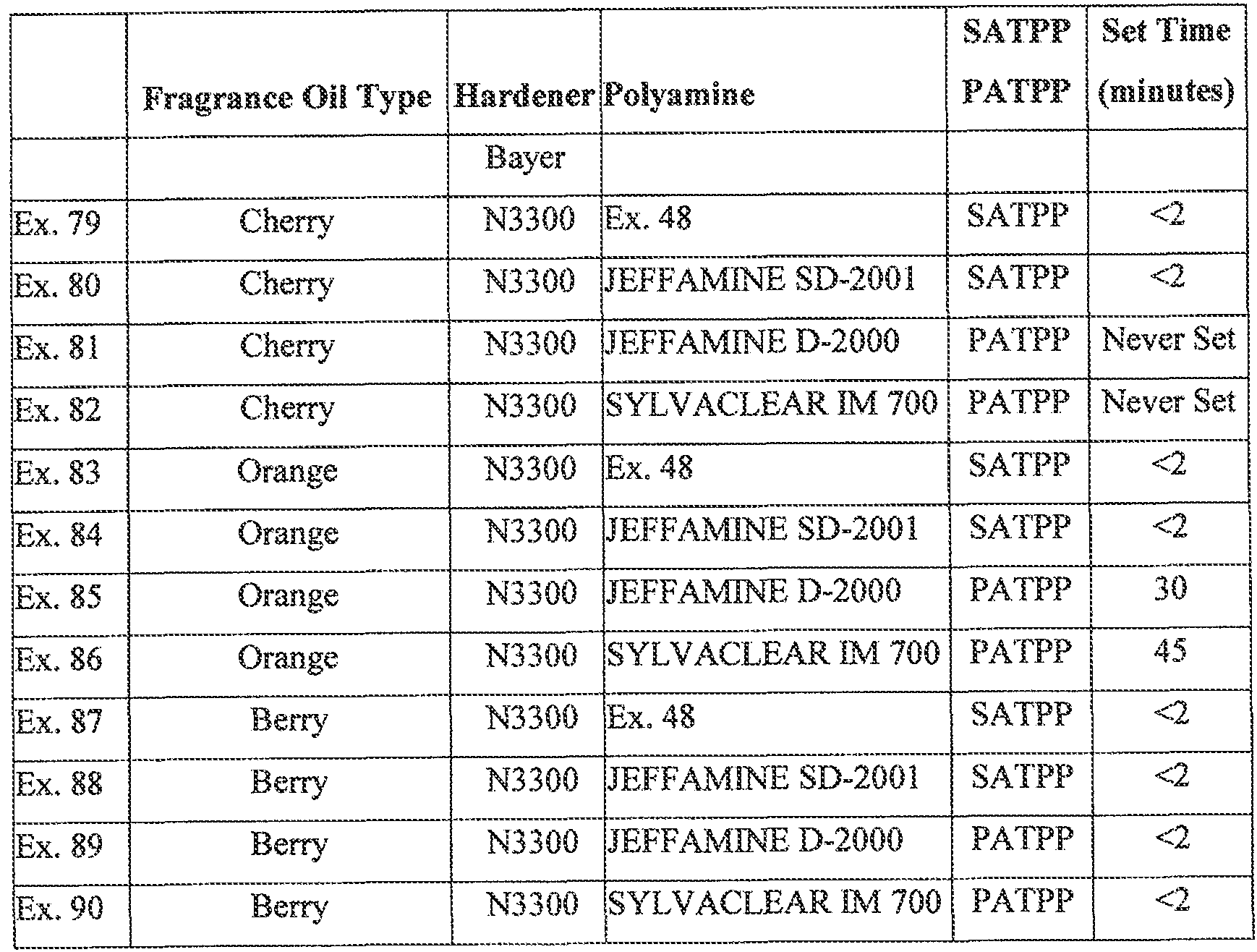
- 235000019693 cherries Nutrition 0.000 description 7
- 239000003431 cross linking reagent Substances 0.000 description 7
- 150000004985 diamines Chemical class 0.000 description 7
- 238000009472 formulation Methods 0.000 description 7
- 239000003607 modifier Substances 0.000 description 7
- 239000003016 pheromone Substances 0.000 description 7
- 229920000570 polyether Polymers 0.000 description 7
- 229920000573 polyethylene Polymers 0.000 description 7
- 239000004848 polyfunctional curative Substances 0.000 description 7
- 229920001228 polyisocyanate Polymers 0.000 description 7
- 239000005056 polyisocyanate Substances 0.000 description 7
- 208000028681 postaxial polydactyly type A Diseases 0.000 description 7
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 6
- 239000003054 catalyst Substances 0.000 description 6
- 239000003086 colorant Substances 0.000 description 6
- 235000014113 dietary fatty acids Nutrition 0.000 description 6
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical class C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 6
- 239000000194 fatty acid Substances 0.000 description 6
- 229930195729 fatty acid Natural products 0.000 description 6
- 150000004665 fatty acids Chemical class 0.000 description 6
- 239000011888 foil Substances 0.000 description 6
- 230000003100 immobilizing effect Effects 0.000 description 6
- FWWQKRXKHIRPJY-UHFFFAOYSA-N octadecanal Chemical compound CCCCCCCCCCCCCCCCCC=O FWWQKRXKHIRPJY-UHFFFAOYSA-N 0.000 description 6
- 239000000123 paper Substances 0.000 description 6
- 239000004014 plasticizer Substances 0.000 description 6
- 229920000647 polyepoxide Polymers 0.000 description 6
- 238000003860 storage Methods 0.000 description 6
- 241000894006 Bacteria Species 0.000 description 5
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 5
- 229920003091 Methocel™ Polymers 0.000 description 5
- 150000007513 acids Chemical class 0.000 description 5
- 150000001298 alcohols Chemical class 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 239000003822 epoxy resin Substances 0.000 description 5
- 239000012530 fluid Substances 0.000 description 5
- 239000006260 foam Substances 0.000 description 5
- 239000001046 green dye Substances 0.000 description 5
- 239000004615 ingredient Substances 0.000 description 5
- 244000005700 microbiome Species 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 239000011541 reaction mixture Substances 0.000 description 5
- 239000001044 red dye Substances 0.000 description 5
- 238000010792 warming Methods 0.000 description 5
- MOQGCGNUWBPGTQ-UHFFFAOYSA-N 2,6,6-trimethyl-1-cyclohexene-1-carboxaldehyde Chemical compound CC1=C(C=O)C(C)(C)CCC1 MOQGCGNUWBPGTQ-UHFFFAOYSA-N 0.000 description 4
- VZWGRQBCURJOMT-UHFFFAOYSA-N Dodecyl acetate Chemical compound CCCCCCCCCCCCOC(C)=O VZWGRQBCURJOMT-UHFFFAOYSA-N 0.000 description 4
- WEEGYLXZBRQIMU-UHFFFAOYSA-N Eucalyptol Chemical compound C1CC2CCC1(C)OC2(C)C WEEGYLXZBRQIMU-UHFFFAOYSA-N 0.000 description 4
- 241000233866 Fungi Species 0.000 description 4
- MMOXZBCLCQITDF-UHFFFAOYSA-N N,N-diethyl-m-toluamide Chemical compound CCN(CC)C(=O)C1=CC=CC(C)=C1 MMOXZBCLCQITDF-UHFFFAOYSA-N 0.000 description 4
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 4
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 4
- QLBRROYTTDFLDX-UHFFFAOYSA-N [3-(aminomethyl)cyclohexyl]methanamine Chemical compound NCC1CCCC(CN)C1 QLBRROYTTDFLDX-UHFFFAOYSA-N 0.000 description 4
- 150000003934 aromatic aldehydes Chemical class 0.000 description 4
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 239000011111 cardboard Substances 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- NEHNMFOYXAPHSD-UHFFFAOYSA-N citronellal Chemical compound O=CCC(C)CCC=C(C)C NEHNMFOYXAPHSD-UHFFFAOYSA-N 0.000 description 4
- DLAHAXOYRFRPFQ-UHFFFAOYSA-N dodecyl benzoate Chemical compound CCCCCCCCCCCCOC(=O)C1=CC=CC=C1 DLAHAXOYRFRPFQ-UHFFFAOYSA-N 0.000 description 4
- 239000000975 dye Substances 0.000 description 4
- 229920001971 elastomer Polymers 0.000 description 4
- 239000000839 emulsion Substances 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 239000004744 fabric Substances 0.000 description 4
- 239000002979 fabric softener Substances 0.000 description 4
- 239000000945 filler Substances 0.000 description 4
- 239000000796 flavoring agent Substances 0.000 description 4
- 235000019634 flavors Nutrition 0.000 description 4
- NAQMVNRVTILPCV-UHFFFAOYSA-N hexane-1,6-diamine Chemical compound NCCCCCCN NAQMVNRVTILPCV-UHFFFAOYSA-N 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 230000000813 microbial effect Effects 0.000 description 4
- ZRSNZINYAWTAHE-UHFFFAOYSA-N p-methoxybenzaldehyde Chemical class COC1=CC=C(C=O)C=C1 ZRSNZINYAWTAHE-UHFFFAOYSA-N 0.000 description 4
- 239000002985 plastic film Substances 0.000 description 4
- 229920006255 plastic film Polymers 0.000 description 4
- 150000003141 primary amines Chemical class 0.000 description 4
- 239000005060 rubber Substances 0.000 description 4
- 238000007789 sealing Methods 0.000 description 4
- 239000000377 silicon dioxide Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- KJPRLNWUNMBNBZ-QPJJXVBHSA-N (E)-cinnamaldehyde Chemical compound O=C\C=C\C1=CC=CC=C1 KJPRLNWUNMBNBZ-QPJJXVBHSA-N 0.000 description 3
- RNLHGQLZWXBQNY-UHFFFAOYSA-N 3-(aminomethyl)-3,5,5-trimethylcyclohexan-1-amine Chemical compound CC1(C)CC(N)CC(C)(CN)C1 RNLHGQLZWXBQNY-UHFFFAOYSA-N 0.000 description 3
- WTEVQBCEXWBHNA-UHFFFAOYSA-N Citral Natural products CC(C)=CCCC(C)=CC=O WTEVQBCEXWBHNA-UHFFFAOYSA-N 0.000 description 3
- 229920000742 Cotton Polymers 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 240000000249 Morus alba Species 0.000 description 3
- 235000008708 Morus alba Nutrition 0.000 description 3
- 241000244206 Nematoda Species 0.000 description 3
- 229920002732 Polyanhydride Polymers 0.000 description 3
- 241000283984 Rodentia Species 0.000 description 3
- VBIIFPGSPJYLRR-UHFFFAOYSA-M Stearyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)C VBIIFPGSPJYLRR-UHFFFAOYSA-M 0.000 description 3
- 241000700605 Viruses Species 0.000 description 3
- 239000013543 active substance Substances 0.000 description 3
- 239000001361 adipic acid Substances 0.000 description 3
- 235000011037 adipic acid Nutrition 0.000 description 3
- XTKDAFGWCDAMPY-UHFFFAOYSA-N azaperone Chemical compound C1=CC(F)=CC=C1C(=O)CCCN1CCN(C=2N=CC=CC=2)CC1 XTKDAFGWCDAMPY-UHFFFAOYSA-N 0.000 description 3
- VEMKTZHHVJILDY-UXHICEINSA-N bioresmethrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OCC1=COC(CC=2C=CC=CC=2)=C1 VEMKTZHHVJILDY-UXHICEINSA-N 0.000 description 3
- WOWHHFRSBJGXCM-UHFFFAOYSA-M cetyltrimethylammonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCC[N+](C)(C)C WOWHHFRSBJGXCM-UHFFFAOYSA-M 0.000 description 3
- 239000003610 charcoal Substances 0.000 description 3
- 229940117916 cinnamic aldehyde Drugs 0.000 description 3
- KJPRLNWUNMBNBZ-UHFFFAOYSA-N cinnamic aldehyde Natural products O=CC=CC1=CC=CC=C1 KJPRLNWUNMBNBZ-UHFFFAOYSA-N 0.000 description 3
- 229940043350 citral Drugs 0.000 description 3
- 239000004927 clay Substances 0.000 description 3
- 238000004132 cross linking Methods 0.000 description 3
- 229960001673 diethyltoluamide Drugs 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 235000013305 food Nutrition 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- WTEVQBCEXWBHNA-JXMROGBWSA-N geranial Chemical compound CC(C)=CCC\C(C)=C\C=O WTEVQBCEXWBHNA-JXMROGBWSA-N 0.000 description 3
- 150000002466 imines Chemical class 0.000 description 3
- 239000010985 leather Substances 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- OSWPMRLSEDHDFF-UHFFFAOYSA-N methyl salicylate Chemical compound COC(=O)C1=CC=CC=C1O OSWPMRLSEDHDFF-UHFFFAOYSA-N 0.000 description 3
- 239000003595 mist Substances 0.000 description 3
- 239000002304 perfume Substances 0.000 description 3
- 230000003389 potentiating effect Effects 0.000 description 3
- 238000003918 potentiometric titration Methods 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- HYJYGLGUBUDSLJ-UHFFFAOYSA-N pyrethrin Natural products CCC(=O)OC1CC(=C)C2CC3OC3(C)C2C2OC(=O)C(=C)C12 HYJYGLGUBUDSLJ-UHFFFAOYSA-N 0.000 description 3
- 239000002689 soil Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- MWOOGOJBHIARFG-UHFFFAOYSA-N vanillin Chemical class COC1=CC(C=O)=CC=C1O MWOOGOJBHIARFG-UHFFFAOYSA-N 0.000 description 3
- FGQOOHJZONJGDT-UHFFFAOYSA-N vanillin Chemical class COC1=CC(O)=CC(C=O)=C1 FGQOOHJZONJGDT-UHFFFAOYSA-N 0.000 description 3
- 235000012141 vanillin Nutrition 0.000 description 3
- 239000002023 wood Substances 0.000 description 3
- VLUMOWNVWOXZAU-VQHVLOKHSA-N (e)-2-methyl-3-phenylprop-2-enal Chemical compound O=CC(/C)=C/C1=CC=CC=C1 VLUMOWNVWOXZAU-VQHVLOKHSA-N 0.000 description 2
- PXGZQGDTEZPERC-UHFFFAOYSA-N 1,4-cyclohexanedicarboxylic acid Chemical compound OC(=O)C1CCC(C(O)=O)CC1 PXGZQGDTEZPERC-UHFFFAOYSA-N 0.000 description 2
- OJMSFYDWZDKGJI-UHFFFAOYSA-N 2,2-dimethylthietane Chemical compound CC1(C)CCS1 OJMSFYDWZDKGJI-UHFFFAOYSA-N 0.000 description 2
- COQGQTARORRRMG-UHFFFAOYSA-N 2,5-dimethylheptadecane Chemical compound CCCCCCCCCCCCC(C)CCC(C)C COQGQTARORRRMG-UHFFFAOYSA-N 0.000 description 2
- HIGURUTWFKYJCH-UHFFFAOYSA-N 2-[[1-(oxiran-2-ylmethoxymethyl)cyclohexyl]methoxymethyl]oxirane Chemical compound C1OC1COCC1(COCC2OC2)CCCCC1 HIGURUTWFKYJCH-UHFFFAOYSA-N 0.000 description 2
- IQVAERDLDAZARL-UHFFFAOYSA-N 2-phenylpropanal Chemical compound O=CC(C)C1=CC=CC=C1 IQVAERDLDAZARL-UHFFFAOYSA-N 0.000 description 2
- GQBLJZKBAFSDSX-UHFFFAOYSA-N 2-propylthietane Chemical compound CCCC1CCS1 GQBLJZKBAFSDSX-UHFFFAOYSA-N 0.000 description 2
- UWKAYLJWKGQEPM-UHFFFAOYSA-N 3,7-dimethylocta-1,6-dien-3-yl acetate Chemical compound CC(C)=CCCC(C)(C=C)OC(C)=O UWKAYLJWKGQEPM-UHFFFAOYSA-N 0.000 description 2
- PYSRRFNXTXNWCD-UHFFFAOYSA-N 3-(2-phenylethenyl)furan-2,5-dione Chemical compound O=C1OC(=O)C(C=CC=2C=CC=CC=2)=C1 PYSRRFNXTXNWCD-UHFFFAOYSA-N 0.000 description 2
- IITQJMYAYSNIMI-UHFFFAOYSA-N 3-Methyl-2-cyclohexen-1-one Chemical compound CC1=CC(=O)CCC1 IITQJMYAYSNIMI-UHFFFAOYSA-N 0.000 description 2
- UJJKWQRTTYLTQL-UHFFFAOYSA-N 3-methyl-6-isopropenyl-9-decenyl acetate Natural products CC(=O)OCCC(C)CCC(CCC=C)C(C)=C UJJKWQRTTYLTQL-UHFFFAOYSA-N 0.000 description 2
- XAUQKOJHYTYNRM-UHFFFAOYSA-N 4,8-dimethyldecanal Chemical compound CCC(C)CCCC(C)CCC=O XAUQKOJHYTYNRM-UHFFFAOYSA-N 0.000 description 2
- ORMHZBNNECIKOH-UHFFFAOYSA-N 4-(4-hydroxy-4-methylpentyl)cyclohex-3-ene-1-carbaldehyde Chemical compound CC(C)(O)CCCC1=CCC(C=O)CC1 ORMHZBNNECIKOH-UHFFFAOYSA-N 0.000 description 2
- YOQFOABVDRBYCG-UHFFFAOYSA-N 6,6-dimethyl-4-methylidenebicyclo[3.1.1]hept-2-ene Chemical compound C1C2C(C)(C)C1C=CC2=C YOQFOABVDRBYCG-UHFFFAOYSA-N 0.000 description 2
- NJADMLYOHSJLKV-UHFFFAOYSA-N 6-Ethyl-2,3-dihydro-2-methyl-4H-pyran-4-one Natural products CCC1=CC(=O)CC(C)O1 NJADMLYOHSJLKV-UHFFFAOYSA-N 0.000 description 2
- 241000238876 Acari Species 0.000 description 2
- 241000193388 Bacillus thuringiensis Species 0.000 description 2
- OTKQNSSMCDLVQV-UHFFFAOYSA-N Butyl 2-methylbutanoate Chemical compound CCCCOC(=O)C(C)CC OTKQNSSMCDLVQV-UHFFFAOYSA-N 0.000 description 2
- 241000207199 Citrus Species 0.000 description 2
- 241000288921 Desmodus Species 0.000 description 2
- 239000005698 Dodecyl acetate Substances 0.000 description 2
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 2
- 241000237858 Gastropoda Species 0.000 description 2
- GLZPCOQZEFWAFX-UHFFFAOYSA-N Geraniol Chemical compound CC(C)=CCCC(C)=CCO GLZPCOQZEFWAFX-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- AMIMRNSIRUDHCM-UHFFFAOYSA-N Isopropylaldehyde Chemical compound CC(C)C=O AMIMRNSIRUDHCM-UHFFFAOYSA-N 0.000 description 2
- IGFHQQFPSIBGKE-UHFFFAOYSA-N Nonylphenol Natural products CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 2
- ZYEMGPIYFIJGTP-UHFFFAOYSA-N O-methyleugenol Chemical compound COC1=CC=C(CC=C)C=C1OC ZYEMGPIYFIJGTP-UHFFFAOYSA-N 0.000 description 2
- VQXSOUPNOZTNAI-UHFFFAOYSA-N Pyrethrin I Natural products CC(=CC1CC1C(=O)OC2CC(=O)C(=C2C)CC=C/C=C)C VQXSOUPNOZTNAI-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- 229920000147 Styrene maleic anhydride Polymers 0.000 description 2
- MOYAFQVGZZPNRA-UHFFFAOYSA-N Terpinolene Chemical compound CC(C)=C1CCC(C)=CC1 MOYAFQVGZZPNRA-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- AXMVYSVVTMKQSL-UHFFFAOYSA-N UNPD142122 Natural products OC1=CC=C(C=CC=O)C=C1O AXMVYSVVTMKQSL-UHFFFAOYSA-N 0.000 description 2
- 244000290333 Vanilla fragrans Species 0.000 description 2
- 235000009499 Vanilla fragrans Nutrition 0.000 description 2
- 235000012036 Vanilla tahitensis Nutrition 0.000 description 2
- WONIGEXYPVIKFS-UHFFFAOYSA-N Verbenol Chemical compound CC1=CC(O)C2C(C)(C)C1C2 WONIGEXYPVIKFS-UHFFFAOYSA-N 0.000 description 2
- MAAMIILYYYQTQW-OUKQBFOZSA-N [(e)-dodec-1-enyl] acetate Chemical compound CCCCCCCCCC\C=C\OC(C)=O MAAMIILYYYQTQW-OUKQBFOZSA-N 0.000 description 2
- CEMVQLPAAAYDAK-WUKNDPDISA-N [(e)-hexadec-1-enyl] acetate Chemical compound CCCCCCCCCCCCCC\C=C\OC(C)=O CEMVQLPAAAYDAK-WUKNDPDISA-N 0.000 description 2
- 239000000642 acaricide Substances 0.000 description 2
- YASYVMFAVPKPKE-UHFFFAOYSA-N acephate Chemical compound COP(=O)(SC)NC(C)=O YASYVMFAVPKPKE-UHFFFAOYSA-N 0.000 description 2
- 230000001476 alcoholic effect Effects 0.000 description 2
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 2
- XCPQUQHBVVXMRQ-UHFFFAOYSA-N alpha-Fenchene Natural products C1CC2C(=C)CC1C2(C)C XCPQUQHBVVXMRQ-UHFFFAOYSA-N 0.000 description 2
- XUEHVOLRMXNRKQ-KHMAMNHCSA-N alpha-cubebene Chemical compound CC(C)[C@@H]([C@H]12)CC[C@@H](C)[C@]32[C@@H]1C(C)=CC3 XUEHVOLRMXNRKQ-KHMAMNHCSA-N 0.000 description 2
- IMUDHTPIFIBORV-UHFFFAOYSA-N aminoethylpiperazine Chemical compound NCCN1CCNCC1 IMUDHTPIFIBORV-UHFFFAOYSA-N 0.000 description 2
- 125000000129 anionic group Chemical group 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 229940097012 bacillus thuringiensis Drugs 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 2
- 235000021028 berry Nutrition 0.000 description 2
- UAHWPYUMFXYFJY-UHFFFAOYSA-N beta-myrcene Chemical compound CC(C)=CCCC(=C)C=C UAHWPYUMFXYFJY-UHFFFAOYSA-N 0.000 description 2
- 230000000853 biopesticidal effect Effects 0.000 description 2
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 2
- XUPYJHCZDLZNFP-UHFFFAOYSA-N butyl butanoate Chemical compound CCCCOC(=O)CCC XUPYJHCZDLZNFP-UHFFFAOYSA-N 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- CRPUJAZIXJMDBK-UHFFFAOYSA-N camphene Chemical compound C1CC2C(=C)C(C)(C)C1C2 CRPUJAZIXJMDBK-UHFFFAOYSA-N 0.000 description 2
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 2
- BQOFWKZOCNGFEC-UHFFFAOYSA-N carene Chemical compound C1C(C)=CCC2C(C)(C)C12 BQOFWKZOCNGFEC-UHFFFAOYSA-N 0.000 description 2
- 239000003093 cationic surfactant Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- BIWJNBZANLAXMG-YQELWRJZSA-N chloordaan Chemical compound ClC1=C(Cl)[C@@]2(Cl)C3CC(Cl)C(Cl)C3[C@]1(Cl)C2(Cl)Cl BIWJNBZANLAXMG-YQELWRJZSA-N 0.000 description 2
- 229930003633 citronellal Natural products 0.000 description 2
- 235000000983 citronellal Nutrition 0.000 description 2
- 235000020971 citrus fruits Nutrition 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 238000013270 controlled release Methods 0.000 description 2
- MZZBPDKVEFVLFF-UHFFFAOYSA-N cyanazine Chemical compound CCNC1=NC(Cl)=NC(NC(C)(C)C#N)=N1 MZZBPDKVEFVLFF-UHFFFAOYSA-N 0.000 description 2
- SSJXIUAHEKJCMH-UHFFFAOYSA-N cyclohexane-1,2-diamine Chemical compound NC1CCCCC1N SSJXIUAHEKJCMH-UHFFFAOYSA-N 0.000 description 2
- VEIOBOXBGYWJIT-UHFFFAOYSA-N cyclohexane;methanol Chemical compound OC.OC.C1CCCCC1 VEIOBOXBGYWJIT-UHFFFAOYSA-N 0.000 description 2
- MWKFXSUHUHTGQN-UHFFFAOYSA-N decan-1-ol Chemical compound CCCCCCCCCCO MWKFXSUHUHTGQN-UHFFFAOYSA-N 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 239000000645 desinfectant Substances 0.000 description 2
- 238000007599 discharging Methods 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- JBFHTYHTHYHCDJ-UHFFFAOYSA-N gamma-caprolactone Chemical compound CCC1CCC(=O)O1 JBFHTYHTHYHCDJ-UHFFFAOYSA-N 0.000 description 2
- 150000002334 glycols Chemical class 0.000 description 2
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 2
- FXHGMKSSBGDXIY-UHFFFAOYSA-N heptanal Chemical compound CCCCCCC=O FXHGMKSSBGDXIY-UHFFFAOYSA-N 0.000 description 2
- NIOYUNMRJMEDGI-UHFFFAOYSA-N hexadecanal Chemical compound CCCCCCCCCCCCCCCC=O NIOYUNMRJMEDGI-UHFFFAOYSA-N 0.000 description 2
- JARKCYVAAOWBJS-UHFFFAOYSA-N hexanal Chemical compound CCCCCC=O JARKCYVAAOWBJS-UHFFFAOYSA-N 0.000 description 2
- AOGQPLXWSUTHQB-UHFFFAOYSA-N hexyl acetate Chemical compound CCCCCCOC(C)=O AOGQPLXWSUTHQB-UHFFFAOYSA-N 0.000 description 2
- XAPCMTMQBXLDBB-UHFFFAOYSA-N hexyl butyrate Chemical compound CCCCCCOC(=O)CCC XAPCMTMQBXLDBB-UHFFFAOYSA-N 0.000 description 2
- 150000002576 ketones Chemical class 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- XMGQYMWWDOXHJM-UHFFFAOYSA-N limonene Chemical compound CC(=C)C1CCC(C)=CC1 XMGQYMWWDOXHJM-UHFFFAOYSA-N 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- IVSZLXZYQVIEFR-UHFFFAOYSA-N m-xylene Chemical group CC1=CC=CC(C)=C1 IVSZLXZYQVIEFR-UHFFFAOYSA-N 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- YMBRJMLOGNZRFY-UHFFFAOYSA-N multistriatin Chemical compound O1C2COC1(CC)C(C)CC2C YMBRJMLOGNZRFY-UHFFFAOYSA-N 0.000 description 2
- GYHFUZHODSMOHU-UHFFFAOYSA-N nonanal Chemical compound CCCCCCCCC=O GYHFUZHODSMOHU-UHFFFAOYSA-N 0.000 description 2
- SNQQPOLDUKLAAF-UHFFFAOYSA-N nonylphenol Chemical compound CCCCCCCCCC1=CC=CC=C1O SNQQPOLDUKLAAF-UHFFFAOYSA-N 0.000 description 2
- HFPZCAJZSCWRBC-UHFFFAOYSA-N p-cymene Chemical compound CC(C)C1=CC=C(C)C=C1 HFPZCAJZSCWRBC-UHFFFAOYSA-N 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- IZUPBVBPLAPZRR-UHFFFAOYSA-N pentachlorophenol Chemical compound OC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl IZUPBVBPLAPZRR-UHFFFAOYSA-N 0.000 description 2
- 150000002989 phenols Chemical class 0.000 description 2
- 229920005862 polyol Polymers 0.000 description 2
- 150000003077 polyols Chemical class 0.000 description 2
- 229920001184 polypeptide Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- LFULEKSKNZEWOE-UHFFFAOYSA-N propanil Chemical compound CCC(=O)NC1=CC=C(Cl)C(Cl)=C1 LFULEKSKNZEWOE-UHFFFAOYSA-N 0.000 description 2
- VJFUPGQZSXIULQ-XIGJTORUSA-N pyrethrin II Chemical compound CC1(C)[C@H](/C=C(\C)C(=O)OC)[C@H]1C(=O)O[C@@H]1C(C)=C(C\C=C/C=C)C(=O)C1 VJFUPGQZSXIULQ-XIGJTORUSA-N 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- SMQUZDBALVYZAC-UHFFFAOYSA-N salicylaldehyde Chemical compound OC1=CC=CC=C1C=O SMQUZDBALVYZAC-UHFFFAOYSA-N 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 239000004576 sand Substances 0.000 description 2
- CXMXRPHRNRROMY-UHFFFAOYSA-N sebacic acid Chemical compound OC(=O)CCCCCCCCC(O)=O CXMXRPHRNRROMY-UHFFFAOYSA-N 0.000 description 2
- 230000035945 sensitivity Effects 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- XLNZEKHULJKQBA-UHFFFAOYSA-N terbufos Chemical compound CCOP(=S)(OCC)SCSC(C)(C)C XLNZEKHULJKQBA-UHFFFAOYSA-N 0.000 description 2
- UHUFTBALEZWWIH-UHFFFAOYSA-N tetradecanal Chemical compound CCCCCCCCCCCCCC=O UHUFTBALEZWWIH-UHFFFAOYSA-N 0.000 description 2
- 229920001169 thermoplastic Polymers 0.000 description 2
- 229920001187 thermosetting polymer Polymers 0.000 description 2
- 239000004416 thermosoftening plastic Substances 0.000 description 2
- KMPQYAYAQWNLME-UHFFFAOYSA-N undecanal Chemical compound CCCCCCCCCCC=O KMPQYAYAQWNLME-UHFFFAOYSA-N 0.000 description 2
- 238000009827 uniform distribution Methods 0.000 description 2
- 239000000341 volatile oil Substances 0.000 description 2
- NFLGAXVYCFJBMK-RKDXNWHRSA-N (+)-isomenthone Natural products CC(C)[C@H]1CC[C@@H](C)CC1=O NFLGAXVYCFJBMK-RKDXNWHRSA-N 0.000 description 1
- ZCVAOQKBXKSDMS-AQYZNVCMSA-N (+)-trans-allethrin Chemical compound CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)OC1C(C)=C(CC=C)C(=O)C1 ZCVAOQKBXKSDMS-AQYZNVCMSA-N 0.000 description 1
- WTARULDDTDQWMU-RKDXNWHRSA-N (+)-β-pinene Chemical compound C1[C@H]2C(C)(C)[C@@H]1CCC2=C WTARULDDTDQWMU-RKDXNWHRSA-N 0.000 description 1
- WTARULDDTDQWMU-IUCAKERBSA-N (-)-Nopinene Natural products C1[C@@H]2C(C)(C)[C@H]1CCC2=C WTARULDDTDQWMU-IUCAKERBSA-N 0.000 description 1
- GEWDNTWNSAZUDX-WQMVXFAESA-N (-)-methyl jasmonate Chemical compound CC\C=C/C[C@@H]1[C@@H](CC(=O)OC)CCC1=O GEWDNTWNSAZUDX-WQMVXFAESA-N 0.000 description 1
- ZXQYGBMAQZUVMI-RDDWSQKMSA-N (1S)-cis-(alphaR)-cyhalothrin Chemical compound CC1(C)[C@H](\C=C(/Cl)C(F)(F)F)[C@@H]1C(=O)O[C@@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 ZXQYGBMAQZUVMI-RDDWSQKMSA-N 0.000 description 1
- GXEKYRXVRROBEV-FBXFSONDSA-N (1r,2s,3r,4s)-7-oxabicyclo[2.2.1]heptane-2,3-dicarboxylic acid Chemical compound C1C[C@@H]2[C@@H](C(O)=O)[C@@H](C(=O)O)[C@H]1O2 GXEKYRXVRROBEV-FBXFSONDSA-N 0.000 description 1
- YONXEBYXWVCXIV-HLTSFMKQSA-N (1r,5s,7r)-7-ethyl-5-methyl-6,8-dioxabicyclo[3.2.1]octane Chemical compound C1CC[C@@H]2[C@@H](CC)O[C@@]1(C)O2 YONXEBYXWVCXIV-HLTSFMKQSA-N 0.000 description 1
- YONXEBYXWVCXIV-YIZRAAEISA-N (1r,5s,7s)-7-ethyl-5-methyl-6,8-dioxabicyclo[3.2.1]octane Chemical compound C1CC[C@@H]2[C@H](CC)O[C@@]1(C)O2 YONXEBYXWVCXIV-YIZRAAEISA-N 0.000 description 1
- AZWKCIZRVUVZPX-JGVFFNPUSA-N (1s,5r)-1,5-dimethyl-6,8-dioxabicyclo[3.2.1]octane Chemical compound C1CC[C@]2(C)OC[C@@]1(C)O2 AZWKCIZRVUVZPX-JGVFFNPUSA-N 0.000 description 1
- UDPGUMQDCGORJQ-UHFFFAOYSA-N (2-chloroethyl)phosphonic acid Chemical compound OP(O)(=O)CCCl UDPGUMQDCGORJQ-UHFFFAOYSA-N 0.000 description 1
- MBDOYVRWFFCFHM-SNAWJCMRSA-N (2E)-hexenal Chemical compound CCC\C=C\C=O MBDOYVRWFFCFHM-SNAWJCMRSA-N 0.000 description 1
- LDVVMCZRFWMZSG-OLQVQODUSA-N (3ar,7as)-2-(trichloromethylsulfanyl)-3a,4,7,7a-tetrahydroisoindole-1,3-dione Chemical compound C1C=CC[C@H]2C(=O)N(SC(Cl)(Cl)Cl)C(=O)[C@H]21 LDVVMCZRFWMZSG-OLQVQODUSA-N 0.000 description 1
- XUNYDVLIZWUPAW-UHFFFAOYSA-N (4-chlorophenyl) n-(4-methylphenyl)sulfonylcarbamate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)NC(=O)OC1=CC=C(Cl)C=C1 XUNYDVLIZWUPAW-UHFFFAOYSA-N 0.000 description 1
- UNYFVMZKRMXDRF-XQRVVYSFSA-N (6z)-14-methyl-1-oxacyclotetradec-6-en-2-one Chemical compound CC1CCCCCC\C=C/CCCC(=O)O1 UNYFVMZKRMXDRF-XQRVVYSFSA-N 0.000 description 1
- HZTKMMXHERVWFC-UHFFFAOYSA-N (7-methyl-3-methylideneoctyl) propanoate Chemical compound CCC(=O)OCCC(=C)CCCC(C)C HZTKMMXHERVWFC-UHFFFAOYSA-N 0.000 description 1
- RTMMLEZUIUUHHW-HWKANZROSA-N (E)-2-Butenyl butyrate Chemical compound CCCC(=O)OC\C=C\C RTMMLEZUIUUHHW-HWKANZROSA-N 0.000 description 1
- IQVNEKKDSLOHHK-FNCQTZNRSA-N (E,E)-hydramethylnon Chemical compound N1CC(C)(C)CNC1=NN=C(/C=C/C=1C=CC(=CC=1)C(F)(F)F)\C=C\C1=CC=C(C(F)(F)F)C=C1 IQVNEKKDSLOHHK-FNCQTZNRSA-N 0.000 description 1
- DCSCXTJOXBUFGB-JGVFFNPUSA-N (R)-(+)-Verbenone Natural products CC1=CC(=O)[C@@H]2C(C)(C)[C@H]1C2 DCSCXTJOXBUFGB-JGVFFNPUSA-N 0.000 description 1
- DCSCXTJOXBUFGB-SFYZADRCSA-N (R)-(+)-verbenone Chemical compound CC1=CC(=O)[C@H]2C(C)(C)[C@@H]1C2 DCSCXTJOXBUFGB-SFYZADRCSA-N 0.000 description 1
- HSGUJTMCFWXGAP-UHFFFAOYSA-N (R)-(E)-14-Methyl-8-hexadecenal Natural products CCC(C)CCCCC=CCCCCCCC=O HSGUJTMCFWXGAP-UHFFFAOYSA-N 0.000 description 1
- DSSYKIVIOFKYAU-XCBNKYQSSA-N (R)-camphor Chemical compound C1C[C@@]2(C)C(=O)C[C@@H]1C2(C)C DSSYKIVIOFKYAU-XCBNKYQSSA-N 0.000 description 1
- QFPVVMKZTVQDTL-UHFFFAOYSA-N (Z)-9-hexadecenal Natural products CCCCCCC=CCCCCCCCC=O QFPVVMKZTVQDTL-UHFFFAOYSA-N 0.000 description 1
- XDBKLFODBADBED-BUHFOSPRSA-N (e)-2-methyloctadec-7-ene Chemical compound CCCCCCCCCC\C=C\CCCCC(C)C XDBKLFODBADBED-BUHFOSPRSA-N 0.000 description 1
- GXANMBISFKBPEX-ONEGZZNKSA-N (e)-hex-3-enal Chemical compound CC\C=C\CC=O GXANMBISFKBPEX-ONEGZZNKSA-N 0.000 description 1
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 1
- HSGUJTMCFWXGAP-ALCCZGGFSA-N (z)-14-methylhexadec-8-enal Chemical compound CCC(C)CCCC\C=C/CCCCCCC=O HSGUJTMCFWXGAP-ALCCZGGFSA-N 0.000 description 1
- IGOWHGRNPLFNDJ-ZPHPHTNESA-N (z)-9-tricosene Chemical compound CCCCCCCCCCCCC\C=C/CCCCCCCC IGOWHGRNPLFNDJ-ZPHPHTNESA-N 0.000 description 1
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Polymers FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 1
- PAAZPARNPHGIKF-UHFFFAOYSA-N 1,2-dibromoethane Chemical compound BrCCBr PAAZPARNPHGIKF-UHFFFAOYSA-N 0.000 description 1
- RRFBTKHQZRCRSS-UHFFFAOYSA-N 1,3-bis(methoxymethyl)-5,5-diphenyl-1,3-diazinane-2,4,6-trione Chemical compound O=C1N(COC)C(=O)N(COC)C(=O)C1(C=1C=CC=CC=1)C1=CC=CC=C1 RRFBTKHQZRCRSS-UHFFFAOYSA-N 0.000 description 1
- WURBVZBTWMNKQT-UHFFFAOYSA-N 1-(4-chlorophenoxy)-3,3-dimethyl-1-(1,2,4-triazol-1-yl)butan-2-one Chemical compound C1=NC=NN1C(C(=O)C(C)(C)C)OC1=CC=C(Cl)C=C1 WURBVZBTWMNKQT-UHFFFAOYSA-N 0.000 description 1
- 239000005968 1-Decanol Substances 0.000 description 1
- PZBPKYOVPCNPJY-UHFFFAOYSA-N 1-[2-(allyloxy)-2-(2,4-dichlorophenyl)ethyl]imidazole Chemical compound ClC1=CC(Cl)=CC=C1C(OCC=C)CN1C=NC=C1 PZBPKYOVPCNPJY-UHFFFAOYSA-N 0.000 description 1
- YIKWKLYQRFRGPM-UHFFFAOYSA-N 1-dodecylguanidine acetate Chemical compound CC(O)=O.CCCCCCCCCCCCN=C(N)N YIKWKLYQRFRGPM-UHFFFAOYSA-N 0.000 description 1
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 1
- FMTFEIJHMMQUJI-NJAFHUGGSA-N 102130-98-3 Natural products CC=CCC1=C(C)[C@H](CC1=O)OC(=O)[C@@H]1[C@@H](C=C(C)C)C1(C)C FMTFEIJHMMQUJI-NJAFHUGGSA-N 0.000 description 1
- ANBOMSJGDBBKMR-UHFFFAOYSA-N 11-Dodecenyl acetate Chemical compound CC(=O)OCCCCCCCCCCC=C ANBOMSJGDBBKMR-UHFFFAOYSA-N 0.000 description 1
- PPNRRQMJESAXGJ-SREVYHEPSA-N 13Z-Octadecenyl acetate Chemical compound CCCC\C=C/CCCCCCCCCCCCOC(C)=O PPNRRQMJESAXGJ-SREVYHEPSA-N 0.000 description 1
- NAEZQVWAWSVOSD-UHFFFAOYSA-N 14-methyloctadec-1-ene Chemical compound CCCCC(C)CCCCCCCCCCCC=C NAEZQVWAWSVOSD-UHFFFAOYSA-N 0.000 description 1
- HDXDEODQBXLZPZ-UHFFFAOYSA-N 2,10-dimethyldodecanoic acid Chemical compound CCC(C)CCCCCCCC(C)C(O)=O HDXDEODQBXLZPZ-UHFFFAOYSA-N 0.000 description 1
- VILCJCGEZXAXTO-UHFFFAOYSA-N 2,2,2-tetramine Chemical compound NCCNCCNCCN VILCJCGEZXAXTO-UHFFFAOYSA-N 0.000 description 1
- AHDSRXYHVZECER-UHFFFAOYSA-N 2,4,6-tris[(dimethylamino)methyl]phenol Chemical compound CN(C)CC1=CC(CN(C)C)=C(O)C(CN(C)C)=C1 AHDSRXYHVZECER-UHFFFAOYSA-N 0.000 description 1
- 239000002794 2,4-DB Substances 0.000 description 1
- YIVXMZJTEQBPQO-UHFFFAOYSA-N 2,4-DB Chemical compound OC(=O)CCCOC1=CC=C(Cl)C=C1Cl YIVXMZJTEQBPQO-UHFFFAOYSA-N 0.000 description 1
- 239000005631 2,4-Dichlorophenoxyacetic acid Substances 0.000 description 1
- YSFBEAASFUWWHU-UHFFFAOYSA-N 2,4-dichlorobenzaldehyde Chemical compound ClC1=CC=C(C=O)C(Cl)=C1 YSFBEAASFUWWHU-UHFFFAOYSA-N 0.000 description 1
- KPUBVYARPYJPPM-UHFFFAOYSA-N 2,5,5-trimethyl-4h-1,3-thiazole Chemical compound CC1=NCC(C)(C)S1 KPUBVYARPYJPPM-UHFFFAOYSA-N 0.000 description 1
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 1
- WNTGYJSOUMFZEP-UHFFFAOYSA-N 2-(4-chloro-2-methylphenoxy)propanoic acid Chemical compound OC(=O)C(C)OC1=CC=C(Cl)C=C1C WNTGYJSOUMFZEP-UHFFFAOYSA-N 0.000 description 1
- GOCUAJYOYBLQRH-UHFFFAOYSA-N 2-(4-{[3-chloro-5-(trifluoromethyl)pyridin-2-yl]oxy}phenoxy)propanoic acid Chemical compound C1=CC(OC(C)C(O)=O)=CC=C1OC1=NC=C(C(F)(F)F)C=C1Cl GOCUAJYOYBLQRH-UHFFFAOYSA-N 0.000 description 1
- OWZPCEFYPSAJFR-UHFFFAOYSA-N 2-(butan-2-yl)-4,6-dinitrophenol Chemical compound CCC(C)C1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1O OWZPCEFYPSAJFR-UHFFFAOYSA-N 0.000 description 1
- KUBDPQJOLOUJRM-UHFFFAOYSA-N 2-(chloromethyl)oxirane;4-[2-(4-hydroxyphenyl)propan-2-yl]phenol Chemical compound ClCC1CO1.C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 KUBDPQJOLOUJRM-UHFFFAOYSA-N 0.000 description 1
- HVGZQCSMLUDISR-UHFFFAOYSA-N 2-Phenylethyl propanoate Chemical compound CCC(=O)OCCC1=CC=CC=C1 HVGZQCSMLUDISR-UHFFFAOYSA-N 0.000 description 1
- CABMTIJINOIHOD-UHFFFAOYSA-N 2-[4-methyl-5-oxo-4-(propan-2-yl)-4,5-dihydro-1H-imidazol-2-yl]quinoline-3-carboxylic acid Chemical compound N1C(=O)C(C(C)C)(C)N=C1C1=NC2=CC=CC=C2C=C1C(O)=O CABMTIJINOIHOD-UHFFFAOYSA-N 0.000 description 1
- WVQBLGZPHOPPFO-UHFFFAOYSA-N 2-chloro-N-(2-ethyl-6-methylphenyl)-N-(1-methoxypropan-2-yl)acetamide Chemical compound CCC1=CC=CC(C)=C1N(C(C)COC)C(=O)CCl WVQBLGZPHOPPFO-UHFFFAOYSA-N 0.000 description 1
- ABMJUFOPOPEJRG-UHFFFAOYSA-N 2-decyl-3-hexyloxirane Chemical compound CCCCCCCCCCC1OC1CCCCCC ABMJUFOPOPEJRG-UHFFFAOYSA-N 0.000 description 1
- UTEDUYINKYCEKZ-UHFFFAOYSA-N 2-methyl-4-methylsulfanylbut-1-ene Chemical compound CSCCC(C)=C UTEDUYINKYCEKZ-UHFFFAOYSA-N 0.000 description 1
- JZUHIOJYCPIVLQ-UHFFFAOYSA-N 2-methylpentane-1,5-diamine Chemical compound NCC(C)CCCN JZUHIOJYCPIVLQ-UHFFFAOYSA-N 0.000 description 1
- GQJLFAGGBJJGDA-UHFFFAOYSA-N 3,11-dimethylnonacosan-2-one Chemical compound CCCCCCCCCCCCCCCCCCC(C)CCCCCCCC(C)C(C)=O GQJLFAGGBJJGDA-UHFFFAOYSA-N 0.000 description 1
- UPMXNNIRAGDFEH-UHFFFAOYSA-N 3,5-dibromo-4-hydroxybenzonitrile Chemical compound OC1=C(Br)C=C(C#N)C=C1Br UPMXNNIRAGDFEH-UHFFFAOYSA-N 0.000 description 1
- XMTQQYYKAHVGBJ-UHFFFAOYSA-N 3-(3,4-DICHLOROPHENYL)-1,1-DIMETHYLUREA Chemical compound CN(C)C(=O)NC1=CC=C(Cl)C(Cl)=C1 XMTQQYYKAHVGBJ-UHFFFAOYSA-N 0.000 description 1
- FSCWZHGZWWDELK-UHFFFAOYSA-N 3-(3,5-dichlorophenyl)-5-ethenyl-5-methyl-2,4-oxazolidinedione Chemical compound O=C1C(C)(C=C)OC(=O)N1C1=CC(Cl)=CC(Cl)=C1 FSCWZHGZWWDELK-UHFFFAOYSA-N 0.000 description 1
- VVJPJXKHBZNADP-QQIRETTESA-N 3E,13E-Octadecadienyl acetate Chemical compound CCCC\C=C\CCCCCCCC\C=C\CCOC(C)=O VVJPJXKHBZNADP-QQIRETTESA-N 0.000 description 1
- HYSSAOQHKOCKIC-HJWRWDBZSA-N 3Z-Decenyl acetate Chemical compound CCCCCC\C=C/CCOC(C)=O HYSSAOQHKOCKIC-HJWRWDBZSA-N 0.000 description 1
- 229940084874 4-(p-hydroxyphenyl)-2-butanone acetate Drugs 0.000 description 1
- AVPYQKSLYISFPO-UHFFFAOYSA-N 4-chlorobenzaldehyde Chemical compound ClC1=CC=C(C=O)C=C1 AVPYQKSLYISFPO-UHFFFAOYSA-N 0.000 description 1
- SHRABZPQLACWEN-UHFFFAOYSA-N 5,11-dimethylheptadecane Chemical compound CCCCCCC(C)CCCCCC(C)CCCC SHRABZPQLACWEN-UHFFFAOYSA-N 0.000 description 1
- QTGIYXFCSKXKMO-KTKRTIGZSA-N 5-[(z)-dec-1-enyl]oxolan-2-one Chemical compound CCCCCCCC\C=C/C1CCC(=O)O1 QTGIYXFCSKXKMO-KTKRTIGZSA-N 0.000 description 1
- CTSLUCNDVMMDHG-UHFFFAOYSA-N 5-bromo-3-(butan-2-yl)-6-methylpyrimidine-2,4(1H,3H)-dione Chemical compound CCC(C)N1C(=O)NC(C)=C(Br)C1=O CTSLUCNDVMMDHG-UHFFFAOYSA-N 0.000 description 1
- VTUFOIHYMMMNOM-VOTSOKGWSA-N 5E-Decenyl acetate Chemical compound CCCC\C=C\CCCCOC(C)=O VTUFOIHYMMMNOM-VOTSOKGWSA-N 0.000 description 1
- FOEWNRTZASNZJY-CMDGGOBGSA-N 5E-Dodecenyl acetate Chemical compound CCCCCC\C=C\CCCCOC(C)=O FOEWNRTZASNZJY-CMDGGOBGSA-N 0.000 description 1
- VTUFOIHYMMMNOM-SREVYHEPSA-N 5Z-Decenyl acetate Chemical compound CCCC\C=C/CCCCOC(C)=O VTUFOIHYMMMNOM-SREVYHEPSA-N 0.000 description 1
- FOEWNRTZASNZJY-HJWRWDBZSA-N 5Z-Dodecenyl acetate Chemical compound CCCCCC\C=C/CCCCOC(C)=O FOEWNRTZASNZJY-HJWRWDBZSA-N 0.000 description 1
- IBSREHMXUMOFBB-JFUDTMANSA-N 5u8924t11h Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](OC)C[C@H](O[C@@H]2C(=C/C[C@@H]3C[C@@H](C[C@@]4(O3)C=C[C@H](C)[C@@H](C(C)C)O4)OC(=O)[C@@H]3C=C(C)[C@@H](O)[C@H]4OC\C([C@@]34O)=C/C=C/[C@@H]2C)/C)O[C@H]1C.C1=C[C@H](C)[C@@H]([C@@H](C)CC)O[C@]11O[C@H](C\C=C(C)\[C@@H](O[C@@H]2O[C@@H](C)[C@H](O[C@@H]3O[C@@H](C)[C@H](O)[C@@H](OC)C3)[C@@H](OC)C2)[C@@H](C)\C=C\C=C/2[C@]3([C@H](C(=O)O4)C=C(C)[C@@H](O)[C@H]3OC\2)O)C[C@H]4C1 IBSREHMXUMOFBB-JFUDTMANSA-N 0.000 description 1
- UEZQOSGCHCNWOE-HJWRWDBZSA-N 7Z-Tetradecenyl acetate Chemical compound CCCCCC\C=C/CCCCCCOC(C)=O UEZQOSGCHCNWOE-HJWRWDBZSA-N 0.000 description 1
- CYJRNFFLTBEQSQ-UHFFFAOYSA-N 8-(3-methyl-1-benzothiophen-5-yl)-N-(4-methylsulfonylpyridin-3-yl)quinoxalin-6-amine Chemical compound CS(=O)(=O)C1=C(C=NC=C1)NC=1C=C2N=CC=NC2=C(C=1)C=1C=CC2=C(C(=CS2)C)C=1 CYJRNFFLTBEQSQ-UHFFFAOYSA-N 0.000 description 1
- SUCYDSJQVVGOIW-WAYWQWQTSA-N 8Z-Dodecenyl acetate Chemical compound CCC\C=C/CCCCCCCOC(C)=O SUCYDSJQVVGOIW-WAYWQWQTSA-N 0.000 description 1
- QFPVVMKZTVQDTL-BQYQJAHWSA-N 9-hexadecenal Chemical compound CCCCCC\C=C\CCCCCCCC=O QFPVVMKZTVQDTL-BQYQJAHWSA-N 0.000 description 1
- MFFQOUCMBNXSBK-SNAWJCMRSA-N 9E-Dodecenyl acetate Chemical compound CC\C=C\CCCCCCCCOC(C)=O MFFQOUCMBNXSBK-SNAWJCMRSA-N 0.000 description 1
- XXPBOEBNDHAAQH-VOTSOKGWSA-N 9E-Tetradecenyl acetate Chemical compound CCCC\C=C\CCCCCCCCOC(C)=O XXPBOEBNDHAAQH-VOTSOKGWSA-N 0.000 description 1
- MFFQOUCMBNXSBK-PLNGDYQASA-N 9Z-Dodecenyl acetate Chemical compound CC\C=C/CCCCCCCCOC(C)=O MFFQOUCMBNXSBK-PLNGDYQASA-N 0.000 description 1
- QFPVVMKZTVQDTL-FPLPWBNLSA-N 9Z-Hexadecenal Chemical compound CCCCCC\C=C/CCCCCCCC=O QFPVVMKZTVQDTL-FPLPWBNLSA-N 0.000 description 1
- XXPBOEBNDHAAQH-SREVYHEPSA-N 9Z-Tetradecenyl acetate Chemical compound CCCC\C=C/CCCCCCCCOC(C)=O XXPBOEBNDHAAQH-SREVYHEPSA-N 0.000 description 1
- 239000005660 Abamectin Substances 0.000 description 1
- RSWGJHLUYNHPMX-UHFFFAOYSA-N Abietic-Saeure Natural products C12CCC(C(C)C)=CC2=CCC2C1(C)CCCC2(C)C(O)=O RSWGJHLUYNHPMX-UHFFFAOYSA-N 0.000 description 1
- VTNQPKFIQCLBDU-UHFFFAOYSA-N Acetochlor Chemical compound CCOCN(C(=O)CCl)C1=C(C)C=CC=C1CC VTNQPKFIQCLBDU-UHFFFAOYSA-N 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- XKJMBINCVNINCA-UHFFFAOYSA-N Alfalone Chemical compound CON(C)C(=O)NC1=CC=C(Cl)C(Cl)=C1 XKJMBINCVNINCA-UHFFFAOYSA-N 0.000 description 1
- 241000238421 Arthropoda Species 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 239000005878 Azadirachtin Substances 0.000 description 1
- RRNIZKPFKNDSRS-UHFFFAOYSA-N Bensulide Chemical compound CC(C)OP(=S)(OC(C)C)SCCNS(=O)(=O)C1=CC=CC=C1 RRNIZKPFKNDSRS-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 239000005489 Bromoxynil Substances 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- BMTAFVWTTFSTOG-UHFFFAOYSA-N Butylate Chemical compound CCSC(=O)N(CC(C)C)CC(C)C BMTAFVWTTFSTOG-UHFFFAOYSA-N 0.000 description 1
- BXJHOKLLMOYSRQ-UHFFFAOYSA-N C(C)(=O)OCCCCCCC=CCCC=CCCCC Chemical compound C(C)(=O)OCCCCCCC=CCCC=CCCCC BXJHOKLLMOYSRQ-UHFFFAOYSA-N 0.000 description 1
- RPRPDTXKGSIXMD-UHFFFAOYSA-N Caproic acid n-butyl ester Natural products CCCCCC(=O)OCCCC RPRPDTXKGSIXMD-UHFFFAOYSA-N 0.000 description 1
- 239000005745 Captan Substances 0.000 description 1
- VEDTXTNSFWUXGQ-UHFFFAOYSA-N Carbophenothion Chemical compound CCOP(=S)(OCC)SCSC1=CC=C(Cl)C=C1 VEDTXTNSFWUXGQ-UHFFFAOYSA-N 0.000 description 1
- 239000005746 Carboxin Substances 0.000 description 1
- DCWKALWZHORJAO-DTWKUNHWSA-N Chalcogran Chemical compound O1[C@@H](CC)CC[C@]11OCCC1 DCWKALWZHORJAO-DTWKUNHWSA-N 0.000 description 1
- DCWKALWZHORJAO-RKDXNWHRSA-N Chalcogran Natural products C(C)[C@H]1O[C@@]2(OCCC2)CC1 DCWKALWZHORJAO-RKDXNWHRSA-N 0.000 description 1
- HSSBORCLYSCBJR-UHFFFAOYSA-N Chloramben Chemical compound NC1=CC(Cl)=CC(C(O)=O)=C1Cl HSSBORCLYSCBJR-UHFFFAOYSA-N 0.000 description 1
- 239000005747 Chlorothalonil Substances 0.000 description 1
- KZCBXHSWMMIEQU-UHFFFAOYSA-N Chlorthal Chemical compound OC(=O)C1=C(Cl)C(Cl)=C(C(O)=O)C(Cl)=C1Cl KZCBXHSWMMIEQU-UHFFFAOYSA-N 0.000 description 1
- 235000007516 Chrysanthemum Nutrition 0.000 description 1
- 240000005250 Chrysanthemum indicum Species 0.000 description 1
- IRZWAJHUWGZMMT-UHFFFAOYSA-N Chrysanthenol Natural products CC1=CCC2C(C)(C)C1C2O IRZWAJHUWGZMMT-UHFFFAOYSA-N 0.000 description 1
- 241000723346 Cinnamomum camphora Species 0.000 description 1
- 235000005979 Citrus limon Nutrition 0.000 description 1
- 244000131522 Citrus pyriformis Species 0.000 description 1
- 241001672694 Citrus reticulata Species 0.000 description 1
- 240000000560 Citrus x paradisi Species 0.000 description 1
- 239000005497 Clethodim Substances 0.000 description 1
- 239000005499 Clomazone Substances 0.000 description 1
- 244000068485 Convallaria majalis Species 0.000 description 1
- 235000009046 Convallaria majalis Nutrition 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- 239000005946 Cypermethrin Substances 0.000 description 1
- YVGGHNCTFXOJCH-UHFFFAOYSA-N DDT Chemical compound C1=CC(Cl)=CC=C1C(C(Cl)(Cl)Cl)C1=CC=C(Cl)C=C1 YVGGHNCTFXOJCH-UHFFFAOYSA-N 0.000 description 1
- NDUPDOJHUQKPAG-UHFFFAOYSA-N Dalapon Chemical compound CC(Cl)(Cl)C(O)=O NDUPDOJHUQKPAG-UHFFFAOYSA-N 0.000 description 1
- NOQGZXFMHARMLW-UHFFFAOYSA-N Daminozide Chemical compound CN(C)NC(=O)CCC(O)=O NOQGZXFMHARMLW-UHFFFAOYSA-N 0.000 description 1
- 239000005975 Daminozide Substances 0.000 description 1
- 239000005892 Deltamethrin Substances 0.000 description 1
- 239000005504 Dicamba Substances 0.000 description 1
- 239000005893 Diflubenzuron Substances 0.000 description 1
- 239000005947 Dimethoate Substances 0.000 description 1
- UDSFAEKRVUSQDD-UHFFFAOYSA-N Dimethyl adipate Chemical compound COC(=O)CCCCC(=O)OC UDSFAEKRVUSQDD-UHFFFAOYSA-N 0.000 description 1
- JYGLAHSAISAEAL-UHFFFAOYSA-N Diphenadione Chemical compound O=C1C2=CC=CC=C2C(=O)C1C(=O)C(C=1C=CC=CC=1)C1=CC=CC=C1 JYGLAHSAISAEAL-UHFFFAOYSA-N 0.000 description 1
- 241000255925 Diptera Species 0.000 description 1
- 239000005630 Diquat Substances 0.000 description 1
- 239000005510 Diuron Substances 0.000 description 1
- 239000005766 Dodine Substances 0.000 description 1
- GUVLYNGULCJVDO-UHFFFAOYSA-N EPTC Chemical compound CCCN(CCC)C(=O)SCC GUVLYNGULCJVDO-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 239000005895 Esfenvalerate Substances 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- 239000005976 Ethephon Substances 0.000 description 1
- ZFDIRQKJPRINOQ-HWKANZROSA-N Ethyl crotonate Chemical compound CCOC(=O)\C=C\C ZFDIRQKJPRINOQ-HWKANZROSA-N 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 239000005958 Fenamiphos (aka phenamiphos) Substances 0.000 description 1
- 239000005898 Fenoxycarb Substances 0.000 description 1
- PNVJTZOFSHSLTO-UHFFFAOYSA-N Fenthion Chemical compound COP(=S)(OC)OC1=CC=C(SC)C(C)=C1 PNVJTZOFSHSLTO-UHFFFAOYSA-N 0.000 description 1
- 239000005533 Fluometuron Substances 0.000 description 1
- 239000005789 Folpet Substances 0.000 description 1
- AIKKULXCBHRFOS-UHFFFAOYSA-N Formothion Chemical compound COP(=S)(OC)SCC(=O)N(C)C=O AIKKULXCBHRFOS-UHFFFAOYSA-N 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 239000005792 Geraniol Substances 0.000 description 1
- GLZPCOQZEFWAFX-YFHOEESVSA-N Geraniol Natural products CC(C)=CCC\C(C)=C/CO GLZPCOQZEFWAFX-YFHOEESVSA-N 0.000 description 1
- CAWXEEYDBZRFPE-UHFFFAOYSA-N Hexazinone Chemical compound O=C1N(C)C(N(C)C)=NC(=O)N1C1CCCCC1 CAWXEEYDBZRFPE-UHFFFAOYSA-N 0.000 description 1
- 241000257303 Hymenoptera Species 0.000 description 1
- 239000005795 Imazalil Substances 0.000 description 1
- 239000005981 Imazaquin Substances 0.000 description 1
- XVOKUMIPKHGGTN-UHFFFAOYSA-N Imazethapyr Chemical compound OC(=O)C1=CC(CC)=CN=C1C1=NC(C)(C(C)C)C(=O)N1 XVOKUMIPKHGGTN-UHFFFAOYSA-N 0.000 description 1
- 239000005906 Imidacloprid Substances 0.000 description 1
- 239000005867 Iprodione Substances 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- 239000005573 Linuron Substances 0.000 description 1
- 239000005574 MCPA Substances 0.000 description 1
- 239000005949 Malathion Substances 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 239000005802 Mancozeb Substances 0.000 description 1
- IMYZQPCYWPFTAG-UHFFFAOYSA-N Mecamylamine Chemical compound C1CC2C(C)(C)C(NC)(C)C1C2 IMYZQPCYWPFTAG-UHFFFAOYSA-N 0.000 description 1
- NFLGAXVYCFJBMK-UHFFFAOYSA-N Menthone Chemical compound CC(C)C1CCC(C)CC1=O NFLGAXVYCFJBMK-UHFFFAOYSA-N 0.000 description 1
- 239000005807 Metalaxyl Substances 0.000 description 1
- 239000005956 Metaldehyde Substances 0.000 description 1
- 239000005916 Methomyl Substances 0.000 description 1
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 1
- 239000005809 Metiram Substances 0.000 description 1
- 239000005583 Metribuzin Substances 0.000 description 1
- 239000005584 Metsulfuron-methyl Substances 0.000 description 1
- 241000237852 Mollusca Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 240000008790 Musa x paradisiaca Species 0.000 description 1
- 235000018290 Musa x paradisiaca Nutrition 0.000 description 1
- WXZVAROIGSFCFJ-UHFFFAOYSA-N N,N-diethyl-2-(naphthalen-1-yloxy)propanamide Chemical compound C1=CC=C2C(OC(C)C(=O)N(CC)CC)=CC=CC2=C1 WXZVAROIGSFCFJ-UHFFFAOYSA-N 0.000 description 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 1
- 239000005585 Napropamide Substances 0.000 description 1
- 239000005586 Nicosulfuron Substances 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- 239000005587 Oryzalin Substances 0.000 description 1
- 241000118205 Ovicides Species 0.000 description 1
- 239000005950 Oxamyl Substances 0.000 description 1
- 239000005590 Oxyfluorfen Substances 0.000 description 1
- OQMBBFQZGJFLBU-UHFFFAOYSA-N Oxyfluorfen Chemical compound C1=C([N+]([O-])=O)C(OCC)=CC(OC=2C(=CC(=CC=2)C(F)(F)F)Cl)=C1 OQMBBFQZGJFLBU-UHFFFAOYSA-N 0.000 description 1
- 241000237509 Patinopecten sp. Species 0.000 description 1
- 239000005591 Pendimethalin Substances 0.000 description 1
- 239000005921 Phosmet Substances 0.000 description 1
- 239000005595 Picloram Substances 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920002367 Polyisobutene Polymers 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 229920002396 Polyurea Polymers 0.000 description 1
- PXRCIOIWVGAZEP-UHFFFAOYSA-N Primaeres Camphenhydrat Natural products C1CC2C(O)(C)C(C)(C)C1C2 PXRCIOIWVGAZEP-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- WTARULDDTDQWMU-UHFFFAOYSA-N Pseudopinene Natural products C1C2C(C)(C)C1CCC2=C WTARULDDTDQWMU-UHFFFAOYSA-N 0.000 description 1
- 241000221535 Pucciniales Species 0.000 description 1
- 239000005614 Quizalofop-P-ethyl Substances 0.000 description 1
- ISRUGXGCCGIOQO-UHFFFAOYSA-N Rhoden Chemical compound CNC(=O)OC1=CC=CC=C1OC(C)C ISRUGXGCCGIOQO-UHFFFAOYSA-N 0.000 description 1
- 241000220317 Rosa Species 0.000 description 1
- KHPCPRHQVVSZAH-HUOMCSJISA-N Rosin Natural products O(C/C=C/c1ccccc1)[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 KHPCPRHQVVSZAH-HUOMCSJISA-N 0.000 description 1
- 241001253920 Ryania Species 0.000 description 1
- OKUGPJPKMAEJOE-UHFFFAOYSA-N S-propyl dipropylcarbamothioate Chemical compound CCCSC(=O)N(CCC)CCC OKUGPJPKMAEJOE-UHFFFAOYSA-N 0.000 description 1
- LSMIOFMZNVEEBR-KIZPAXIPSA-N Scilliroside Natural products O=C(O[C@@H]1C=2[C@@](C)([C@@H]3[C@](O)([C@]4(O)[C@@](C)([C@H](C5=COC(=O)C=C5)CC4)CC3)C1)CC[C@H](O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1)C=2)C LSMIOFMZNVEEBR-KIZPAXIPSA-N 0.000 description 1
- CSPPKDPQLUUTND-NBVRZTHBSA-N Sethoxydim Chemical compound CCO\N=C(/CCC)C1=C(O)CC(CC(C)SCC)CC1=O CSPPKDPQLUUTND-NBVRZTHBSA-N 0.000 description 1
- 206010040880 Skin irritation Diseases 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- HBPDKDSFLXWOAE-UHFFFAOYSA-N Tebuthiuron Chemical compound CNC(=O)N(C)C1=NN=C(C(C)(C)C)S1 HBPDKDSFLXWOAE-UHFFFAOYSA-N 0.000 description 1
- NBQCNZYJJMBDKY-UHFFFAOYSA-N Terbacil Chemical compound CC=1NC(=O)N(C(C)(C)C)C(=O)C=1Cl NBQCNZYJJMBDKY-UHFFFAOYSA-N 0.000 description 1
- IOUUIFSIQMVYKP-UHFFFAOYSA-N Tetradecanoyl acetate Natural products CCCCCCCCCCCCCCOC(C)=O IOUUIFSIQMVYKP-UHFFFAOYSA-N 0.000 description 1
- 239000005843 Thiram Substances 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- WHKUVVPPKQRRBV-UHFFFAOYSA-N Trasan Chemical compound CC1=CC(Cl)=CC=C1OCC(O)=O WHKUVVPPKQRRBV-UHFFFAOYSA-N 0.000 description 1
- 239000005627 Triclopyr Substances 0.000 description 1
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 1
- 229930195482 Validamycin Natural products 0.000 description 1
- FOEWNRTZASNZJY-UHFFFAOYSA-N Z-5-dodecenyl acetate Natural products CCCCCCC=CCCCCOC(C)=O FOEWNRTZASNZJY-UHFFFAOYSA-N 0.000 description 1
- UEZQOSGCHCNWOE-UHFFFAOYSA-N Z-7-tetradecenyl acetate Natural products CCCCCCC=CCCCCCCOC(C)=O UEZQOSGCHCNWOE-UHFFFAOYSA-N 0.000 description 1
- 239000005870 Ziram Substances 0.000 description 1
- VXSIXFKKSNGRRO-MXOVTSAMSA-N [(1s)-2-methyl-4-oxo-3-[(2z)-penta-2,4-dienyl]cyclopent-2-en-1-yl] (1r,3r)-2,2-dimethyl-3-(2-methylprop-1-enyl)cyclopropane-1-carboxylate;[(1s)-2-methyl-4-oxo-3-[(2z)-penta-2,4-dienyl]cyclopent-2-en-1-yl] (1r,3r)-3-[(e)-3-methoxy-2-methyl-3-oxoprop-1-enyl Chemical class CC1(C)[C@H](C=C(C)C)[C@H]1C(=O)O[C@@H]1C(C)=C(C\C=C/C=C)C(=O)C1.CC1(C)[C@H](/C=C(\C)C(=O)OC)[C@H]1C(=O)O[C@@H]1C(C)=C(C\C=C/C=C)C(=O)C1 VXSIXFKKSNGRRO-MXOVTSAMSA-N 0.000 description 1
- QYXUDXTZJFIJFY-ZROIWOOFSA-N [(3z)-3-methyl-6-prop-1-en-2-yldeca-3,9-dienyl] acetate Chemical compound CC(=O)OCC\C(C)=C/CC(C(C)=C)CCC=C QYXUDXTZJFIJFY-ZROIWOOFSA-N 0.000 description 1
- NTKDSWPSEFZZOZ-VNKDHWASSA-N [(8e,10e)-dodeca-8,10-dienyl] acetate Chemical compound C\C=C\C=C\CCCCCCCOC(C)=O NTKDSWPSEFZZOZ-VNKDHWASSA-N 0.000 description 1
- RYTIKHKWIBYADW-VHEBQXMUSA-N [(e)-octadec-1-enyl] acetate Chemical compound CCCCCCCCCCCCCCCC\C=C\OC(C)=O RYTIKHKWIBYADW-VHEBQXMUSA-N 0.000 description 1
- UECPLNNAVLEZGO-CCEZHUSRSA-N [(e)-tetradec-1-enyl] acetate Chemical compound CCCCCCCCCCCC\C=C\OC(C)=O UECPLNNAVLEZGO-CCEZHUSRSA-N 0.000 description 1
- HQVIAWLVNPAJHL-BUHFOSPRSA-N [(e)-tridec-1-enyl] acetate Chemical compound CCCCCCCCCCC\C=C\OC(C)=O HQVIAWLVNPAJHL-BUHFOSPRSA-N 0.000 description 1
- VAKBQCYSUVICLV-HJWRWDBZSA-N [(z)-hexadec-9-enyl] acetate Chemical compound CCCCCC\C=C/CCCCCCCCOC(C)=O VAKBQCYSUVICLV-HJWRWDBZSA-N 0.000 description 1
- DUCVQKMNGSZPAV-KHPPLWFESA-N [(z)-tridec-4-enyl] acetate Chemical compound CCCCCCCC\C=C/CCCOC(C)=O DUCVQKMNGSZPAV-KHPPLWFESA-N 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- INISTDXBRIBGOC-CGAIIQECSA-N [cyano-(3-phenoxyphenyl)methyl] (2s)-2-[2-chloro-4-(trifluoromethyl)anilino]-3-methylbutanoate Chemical compound N([C@@H](C(C)C)C(=O)OC(C#N)C=1C=C(OC=2C=CC=CC=2)C=CC=1)C1=CC=C(C(F)(F)F)C=C1Cl INISTDXBRIBGOC-CGAIIQECSA-N 0.000 description 1
- 229950008167 abamectin Drugs 0.000 description 1
- 230000000895 acaricidal effect Effects 0.000 description 1
- VQFYMJNHSLSNRE-UHFFFAOYSA-N acetic acid;4-(4-hydroxyphenyl)butan-2-one Chemical compound CC(O)=O.CC(=O)CCC1=CC=C(O)C=C1 VQFYMJNHSLSNRE-UHFFFAOYSA-N 0.000 description 1
- OIPILFWXSMYKGL-UHFFFAOYSA-N acetylcholine Chemical compound CC(=O)OCC[N+](C)(C)C OIPILFWXSMYKGL-UHFFFAOYSA-N 0.000 description 1
- 229960004373 acetylcholine Drugs 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- XCSGPAVHZFQHGE-UHFFFAOYSA-N alachlor Chemical compound CCC1=CC=CC(CC)=C1N(COC)C(=O)CCl XCSGPAVHZFQHGE-UHFFFAOYSA-N 0.000 description 1
- QGLZXHRNAYXIBU-WEVVVXLNSA-N aldicarb Chemical compound CNC(=O)O\N=C\C(C)(C)SC QGLZXHRNAYXIBU-WEVVVXLNSA-N 0.000 description 1
- 239000003619 algicide Substances 0.000 description 1
- 150000005215 alkyl ethers Chemical class 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 239000011717 all-trans-retinol Substances 0.000 description 1
- 235000019169 all-trans-retinol Nutrition 0.000 description 1
- 229940024113 allethrin Drugs 0.000 description 1
- UZFLPKAIBPNNCA-BQYQJAHWSA-N alpha-ionone Chemical compound CC(=O)\C=C\C1C(C)=CCCC1(C)C UZFLPKAIBPNNCA-BQYQJAHWSA-N 0.000 description 1
- UZFLPKAIBPNNCA-UHFFFAOYSA-N alpha-ionone Natural products CC(=O)C=CC1C(C)=CCCC1(C)C UZFLPKAIBPNNCA-UHFFFAOYSA-N 0.000 description 1
- VYBREYKSZAROCT-UHFFFAOYSA-N alpha-myrcene Natural products CC(=C)CCCC(=C)C=C VYBREYKSZAROCT-UHFFFAOYSA-N 0.000 description 1
- GUUHFMWKWLOQMM-UHFFFAOYSA-N alpha-n-hexylcinnamic aldehyde Natural products CCCCCCC(C=O)=CC1=CC=CC=C1 GUUHFMWKWLOQMM-UHFFFAOYSA-N 0.000 description 1
- 229920005603 alternating copolymer Polymers 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- RQVYBGPQFYCBGX-UHFFFAOYSA-N ametryn Chemical compound CCNC1=NC(NC(C)C)=NC(SC)=N1 RQVYBGPQFYCBGX-UHFFFAOYSA-N 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 229960002587 amitraz Drugs 0.000 description 1
- QXAITBQSYVNQDR-ZIOPAAQOSA-N amitraz Chemical compound C=1C=C(C)C=C(C)C=1/N=C/N(C)\C=N\C1=CC=C(C)C=C1C QXAITBQSYVNQDR-ZIOPAAQOSA-N 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 239000002519 antifouling agent Substances 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 238000000149 argon plasma sintering Methods 0.000 description 1
- 150000008378 aryl ethers Chemical class 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- MXWJVTOOROXGIU-UHFFFAOYSA-N atrazine Chemical compound CCNC1=NC(Cl)=NC(NC(C)C)=N1 MXWJVTOOROXGIU-UHFFFAOYSA-N 0.000 description 1
- 239000005667 attractant Substances 0.000 description 1
- VEHPJKVTJQSSKL-UHFFFAOYSA-N azadirachtin Natural products O1C2(C)C(C3(C=COC3O3)O)CC3C21C1(C)C(O)C(OCC2(OC(C)=O)C(CC3OC(=O)C(C)=CC)OC(C)=O)C2C32COC(C(=O)OC)(O)C12 VEHPJKVTJQSSKL-UHFFFAOYSA-N 0.000 description 1
- FTNJWQUOZFUQQJ-NDAWSKJSSA-N azadirachtin A Chemical compound C([C@@H]([C@]1(C=CO[C@H]1O1)O)[C@]2(C)O3)[C@H]1[C@]23[C@]1(C)[C@H](O)[C@H](OC[C@@]2([C@@H](C[C@@H]3OC(=O)C(\C)=C\C)OC(C)=O)C(=O)OC)[C@@H]2[C@]32CO[C@@](C(=O)OC)(O)[C@@H]12 FTNJWQUOZFUQQJ-NDAWSKJSSA-N 0.000 description 1
- FTNJWQUOZFUQQJ-IRYYUVNJSA-N azadirachtin A Natural products C([C@@H]([C@]1(C=CO[C@H]1O1)O)[C@]2(C)O3)[C@H]1[C@]23[C@]1(C)[C@H](O)[C@H](OC[C@@]2([C@@H](C[C@@H]3OC(=O)C(\C)=C/C)OC(C)=O)C(=O)OC)[C@@H]2[C@]32CO[C@@](C(=O)OC)(O)[C@@H]12 FTNJWQUOZFUQQJ-IRYYUVNJSA-N 0.000 description 1
- 239000003899 bactericide agent Substances 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- XEGGRYVFLWGFHI-UHFFFAOYSA-N bendiocarb Chemical compound CNC(=O)OC1=CC=CC2=C1OC(C)(C)O2 XEGGRYVFLWGFHI-UHFFFAOYSA-N 0.000 description 1
- RIOXQFHNBCKOKP-UHFFFAOYSA-N benomyl Chemical compound C1=CC=C2N(C(=O)NCCCC)C(NC(=O)OC)=NC2=C1 RIOXQFHNBCKOKP-UHFFFAOYSA-N 0.000 description 1
- ZOMSMJKLGFBRBS-UHFFFAOYSA-N bentazone Chemical compound C1=CC=C2NS(=O)(=O)N(C(C)C)C(=O)C2=C1 ZOMSMJKLGFBRBS-UHFFFAOYSA-N 0.000 description 1
- 150000003935 benzaldehydes Chemical class 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- VVKREWPWSWPBGC-UHFFFAOYSA-N benzoic acid;2-(2-hydroxypropoxy)propan-1-ol Chemical compound CC(O)COC(C)CO.OC(=O)C1=CC=CC=C1 VVKREWPWSWPBGC-UHFFFAOYSA-N 0.000 description 1
- MITFXPHMIHQXPI-UHFFFAOYSA-N benzoxaprofen Natural products N=1C2=CC(C(C(O)=O)C)=CC=C2OC=1C1=CC=C(Cl)C=C1 MITFXPHMIHQXPI-UHFFFAOYSA-N 0.000 description 1
- 229930006722 beta-pinene Natural products 0.000 description 1
- 239000003139 biocide Substances 0.000 description 1
- 239000003124 biologic agent Substances 0.000 description 1
- 229920001222 biopolymer Polymers 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- ZXFZFLUNBACXQF-UHFFFAOYSA-N bromo-chloro-methoxymethane Chemical compound COC(Cl)Br ZXFZFLUNBACXQF-UHFFFAOYSA-N 0.000 description 1
- VASBMTKQLWDLDL-UHFFFAOYSA-N but-3-enoxybenzene Chemical compound C=CCCOC1=CC=CC=C1 VASBMTKQLWDLDL-UHFFFAOYSA-N 0.000 description 1
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 description 1
- 239000001343 butyl (2R)-2-methylbutanoate Substances 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 229930006739 camphene Natural products 0.000 description 1
- ZYPYEBYNXWUCEA-UHFFFAOYSA-N camphenilone Natural products C1CC2C(=O)C(C)(C)C1C2 ZYPYEBYNXWUCEA-UHFFFAOYSA-N 0.000 description 1
- 229930008380 camphor Natural products 0.000 description 1
- 229960000846 camphor Drugs 0.000 description 1
- JHRWWRDRBPCWTF-OLQVQODUSA-N captafol Chemical compound C1C=CC[C@H]2C(=O)N(SC(Cl)(Cl)C(Cl)Cl)C(=O)[C@H]21 JHRWWRDRBPCWTF-OLQVQODUSA-N 0.000 description 1
- 229940117949 captan Drugs 0.000 description 1
- 229930006737 car-3-ene Natural products 0.000 description 1
- 229960005286 carbaryl Drugs 0.000 description 1
- CVXBEEMKQHEXEN-UHFFFAOYSA-N carbaryl Chemical compound C1=CC=C2C(OC(=O)NC)=CC=CC2=C1 CVXBEEMKQHEXEN-UHFFFAOYSA-N 0.000 description 1
- DUEPRVBVGDRKAG-UHFFFAOYSA-N carbofuran Chemical compound CNC(=O)OC1=CC=CC2=C1OC(C)(C)C2 DUEPRVBVGDRKAG-UHFFFAOYSA-N 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- GYSSRZJIHXQEHQ-UHFFFAOYSA-N carboxin Chemical compound S1CCOC(C)=C1C(=O)NC1=CC=CC=C1 GYSSRZJIHXQEHQ-UHFFFAOYSA-N 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- MXZACTZQSGYANA-UHFFFAOYSA-N chembl545463 Chemical compound Cl.C1=CC(OC)=CC=C1C(N=C1)=CN2C1=NC(C)=C2O MXZACTZQSGYANA-UHFFFAOYSA-N 0.000 description 1
- LFHISGNCFUNFFM-UHFFFAOYSA-N chloropicrin Chemical compound [O-][N+](=O)C(Cl)(Cl)Cl LFHISGNCFUNFFM-UHFFFAOYSA-N 0.000 description 1
- CRQQGFGUEAVUIL-UHFFFAOYSA-N chlorothalonil Chemical compound ClC1=C(Cl)C(C#N)=C(Cl)C(C#N)=C1Cl CRQQGFGUEAVUIL-UHFFFAOYSA-N 0.000 description 1
- 125000002871 cinnamic aldehydes group Chemical class 0.000 description 1
- 239000010630 cinnamon oil Substances 0.000 description 1
- ZDKZHVNKFOXMND-UHFFFAOYSA-N cis-Nepetalactone Natural products O=C1OC=C(C)C2C1C(C)CC2 ZDKZHVNKFOXMND-UHFFFAOYSA-N 0.000 description 1
- ZDKZHVNKFOXMND-NBEYISGCSA-N cis-trans-nepetalactone Chemical compound O=C1OC=C(C)[C@@H]2[C@H]1[C@@H](C)CC2 ZDKZHVNKFOXMND-NBEYISGCSA-N 0.000 description 1
- SILSDTWXNBZOGF-JWGBMQLESA-N clethodim Chemical compound CCSC(C)CC1CC(O)=C(C(CC)=NOC\C=C\Cl)C(=O)C1 SILSDTWXNBZOGF-JWGBMQLESA-N 0.000 description 1
- KIEDNEWSYUYDSN-UHFFFAOYSA-N clomazone Chemical compound O=C1C(C)(C)CON1CC1=CC=CC=C1Cl KIEDNEWSYUYDSN-UHFFFAOYSA-N 0.000 description 1
- MRUAUOIMASANKQ-UHFFFAOYSA-N cocamidopropyl betaine Chemical compound CCCCCCCCCCCC(=O)NCCC[N+](C)(C)CC([O-])=O MRUAUOIMASANKQ-UHFFFAOYSA-N 0.000 description 1
- 229940073507 cocamidopropyl betaine Drugs 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- BXNANOICGRISHX-UHFFFAOYSA-N coumaphos Chemical compound CC1=C(Cl)C(=O)OC2=CC(OP(=S)(OCC)OCC)=CC=C21 BXNANOICGRISHX-UHFFFAOYSA-N 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- VNLZLLDMKRKVEX-UHFFFAOYSA-N cyclohex-3-enone Chemical compound O=C1CCC=CC1 VNLZLLDMKRKVEX-UHFFFAOYSA-N 0.000 description 1
- WXJXAGZUCSFYCV-UHFFFAOYSA-N cyclohexanamine;propan-1-amine Chemical compound CCCN.NC1CCCCC1 WXJXAGZUCSFYCV-UHFFFAOYSA-N 0.000 description 1
- 229960001591 cyfluthrin Drugs 0.000 description 1
- QQODLKZGRKWIFG-QSFXBCCZSA-N cyfluthrin Chemical compound CC1(C)[C@@H](C=C(Cl)Cl)[C@H]1C(=O)O[C@@H](C#N)C1=CC=C(F)C(OC=2C=CC=CC=2)=C1 QQODLKZGRKWIFG-QSFXBCCZSA-N 0.000 description 1
- 229960005424 cypermethrin Drugs 0.000 description 1
- KAATUXNTWXVJKI-UHFFFAOYSA-N cypermethrin Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)OC(C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 KAATUXNTWXVJKI-UHFFFAOYSA-N 0.000 description 1
- DEOHUYGDZACDBU-UHFFFAOYSA-N dec-7-enyl acetate Chemical compound CCC=CCCCCCCOC(C)=O DEOHUYGDZACDBU-UHFFFAOYSA-N 0.000 description 1
- 229960002483 decamethrin Drugs 0.000 description 1
- 229910021641 deionized water Inorganic materials 0.000 description 1
- OWZREIFADZCYQD-NSHGMRRFSA-N deltamethrin Chemical compound CC1(C)[C@@H](C=C(Br)Br)[C@H]1C(=O)O[C@H](C#N)C1=CC=CC(OC=2C=CC=CC=2)=C1 OWZREIFADZCYQD-NSHGMRRFSA-N 0.000 description 1
- WEBQKRLKWNIYKK-UHFFFAOYSA-N demeton-S-methyl Chemical group CCSCCSP(=O)(OC)OC WEBQKRLKWNIYKK-UHFFFAOYSA-N 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- FHIVAFMUCKRCQO-UHFFFAOYSA-N diazinon Chemical compound CCOP(=S)(OCC)OC1=CC(C)=NC(C(C)C)=N1 FHIVAFMUCKRCQO-UHFFFAOYSA-N 0.000 description 1
- LCXAARCEIWRIMU-UHFFFAOYSA-M dibenzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C=1C=CC=CC=1C[N+](C)(C)CC1=CC=CC=C1 LCXAARCEIWRIMU-UHFFFAOYSA-M 0.000 description 1
- IWEDIXLBFLAXBO-UHFFFAOYSA-N dicamba Chemical compound COC1=C(Cl)C=CC(Cl)=C1C(O)=O IWEDIXLBFLAXBO-UHFFFAOYSA-N 0.000 description 1
- OEBRKCOSUFCWJD-UHFFFAOYSA-N dichlorvos Chemical compound COP(=O)(OC)OC=C(Cl)Cl OEBRKCOSUFCWJD-UHFFFAOYSA-N 0.000 description 1
- 229950001327 dichlorvos Drugs 0.000 description 1
- UOAMTSKGCBMZTC-UHFFFAOYSA-N dicofol Chemical compound C=1C=C(Cl)C=CC=1C(C(Cl)(Cl)Cl)(O)C1=CC=C(Cl)C=C1 UOAMTSKGCBMZTC-UHFFFAOYSA-N 0.000 description 1
- KORSJDCBLAPZEQ-UHFFFAOYSA-N dicyclohexylmethane-4,4'-diisocyanate Chemical compound C1CC(N=C=O)CCC1CC1CCC(N=C=O)CC1 KORSJDCBLAPZEQ-UHFFFAOYSA-N 0.000 description 1
- JXSJBGJIGXNWCI-UHFFFAOYSA-N diethyl 2-[(dimethoxyphosphorothioyl)thio]succinate Chemical compound CCOC(=O)CC(SP(=S)(OC)OC)C(=O)OCC JXSJBGJIGXNWCI-UHFFFAOYSA-N 0.000 description 1
- 229940019503 diflubenzuron Drugs 0.000 description 1
- 125000005442 diisocyanate group Chemical group 0.000 description 1
- MCWXGJITAZMZEV-UHFFFAOYSA-N dimethoate Chemical compound CNC(=O)CSP(=S)(OC)OC MCWXGJITAZMZEV-UHFFFAOYSA-N 0.000 description 1
- IQDGSYLLQPDQDV-UHFFFAOYSA-N dimethylazanium;chloride Chemical compound Cl.CNC IQDGSYLLQPDQDV-UHFFFAOYSA-N 0.000 description 1
- 229960000267 diphenadione Drugs 0.000 description 1
- 235000013601 eggs Nutrition 0.000 description 1
- 125000006575 electron-withdrawing group Chemical group 0.000 description 1
- YONXEBYXWVCXIV-UHFFFAOYSA-N endo-Brevicomin Natural products C1CCC2C(CC)OC1(C)O2 YONXEBYXWVCXIV-UHFFFAOYSA-N 0.000 description 1
- RDYMFSUJUZBWLH-SVWSLYAFSA-N endosulfan Chemical compound C([C@@H]12)OS(=O)OC[C@@H]1[C@]1(Cl)C(Cl)=C(Cl)[C@@]2(Cl)C1(Cl)Cl RDYMFSUJUZBWLH-SVWSLYAFSA-N 0.000 description 1
- 229960002125 enilconazole Drugs 0.000 description 1
- NYPJDWWKZLNGGM-RPWUZVMVSA-N esfenvalerate Chemical compound C=1C([C@@H](C#N)OC(=O)[C@@H](C(C)C)C=2C=CC(Cl)=CC=2)=CC=CC=1OC1=CC=CC=C1 NYPJDWWKZLNGGM-RPWUZVMVSA-N 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- LYCAIKOWRPUZTN-UHFFFAOYSA-N ethylene glycol Natural products OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 1
- 239000010642 eucalyptus oil Substances 0.000 description 1
- 229940044949 eucalyptus oil Drugs 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- ZCJPOPBZHLUFHF-UHFFFAOYSA-N fenamiphos Chemical compound CCOP(=O)(NC(C)C)OC1=CC=C(SC)C(C)=C1 ZCJPOPBZHLUFHF-UHFFFAOYSA-N 0.000 description 1
- ZNOLGFHPUIJIMJ-UHFFFAOYSA-N fenitrothion Chemical compound COP(=S)(OC)OC1=CC=C([N+]([O-])=O)C(C)=C1 ZNOLGFHPUIJIMJ-UHFFFAOYSA-N 0.000 description 1
- HJUFTIJOISQSKQ-UHFFFAOYSA-N fenoxycarb Chemical compound C1=CC(OCCNC(=O)OCC)=CC=C1OC1=CC=CC=C1 HJUFTIJOISQSKQ-UHFFFAOYSA-N 0.000 description 1
- NYPJDWWKZLNGGM-UHFFFAOYSA-N fenvalerate Aalpha Natural products C=1C=C(Cl)C=CC=1C(C(C)C)C(=O)OC(C#N)C(C=1)=CC=CC=1OC1=CC=CC=C1 NYPJDWWKZLNGGM-UHFFFAOYSA-N 0.000 description 1
- 239000003337 fertilizer Substances 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000002657 fibrous material Substances 0.000 description 1
- 239000013020 final formulation Substances 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- VAIZTNZGPYBOGF-CYBMUJFWSA-N fluazifop-P-butyl Chemical group C1=CC(O[C@H](C)C(=O)OCCCC)=CC=C1OC1=CC=C(C(F)(F)F)C=N1 VAIZTNZGPYBOGF-CYBMUJFWSA-N 0.000 description 1
- GBIHOLCMZGAKNG-CGAIIQECSA-N flucythrinate Chemical compound O=C([C@@H](C(C)C)C=1C=CC(OC(F)F)=CC=1)OC(C#N)C(C=1)=CC=CC=1OC1=CC=CC=C1 GBIHOLCMZGAKNG-CGAIIQECSA-N 0.000 description 1
- RZILCCPWPBTYDO-UHFFFAOYSA-N fluometuron Chemical compound CN(C)C(=O)NC1=CC=CC(C(F)(F)F)=C1 RZILCCPWPBTYDO-UHFFFAOYSA-N 0.000 description 1
- HKIOYBQGHSTUDB-UHFFFAOYSA-N folpet Chemical compound C1=CC=C2C(=O)N(SC(Cl)(Cl)Cl)C(=O)C2=C1 HKIOYBQGHSTUDB-UHFFFAOYSA-N 0.000 description 1
- KVGLBTYUCJYMND-UHFFFAOYSA-N fonofos Chemical compound CCOP(=S)(CC)SC1=CC=CC=C1 KVGLBTYUCJYMND-UHFFFAOYSA-N 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 229920001002 functional polymer Polymers 0.000 description 1
- 239000000417 fungicide Substances 0.000 description 1
- WOLATMHLPFJRGC-UHFFFAOYSA-N furan-2,5-dione;styrene Chemical compound O=C1OC(=O)C=C1.C=CC1=CC=CC=C1 WOLATMHLPFJRGC-UHFFFAOYSA-N 0.000 description 1
- LCWMKIHBLJLORW-UHFFFAOYSA-N gamma-carene Natural products C1CC(=C)CC2C(C)(C)C21 LCWMKIHBLJLORW-UHFFFAOYSA-N 0.000 description 1
- JLYXXMFPNIAWKQ-GNIYUCBRSA-N gamma-hexachlorocyclohexane Chemical compound Cl[C@H]1[C@H](Cl)[C@@H](Cl)[C@@H](Cl)[C@H](Cl)[C@H]1Cl JLYXXMFPNIAWKQ-GNIYUCBRSA-N 0.000 description 1
- JLYXXMFPNIAWKQ-UHFFFAOYSA-N gamma-hexachlorocyclohexane Natural products ClC1C(Cl)C(Cl)C(Cl)C(Cl)C1Cl JLYXXMFPNIAWKQ-UHFFFAOYSA-N 0.000 description 1
- 239000007863 gel particle Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 239000010437 gem Substances 0.000 description 1
- 229940113087 geraniol Drugs 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000008821 health effect Effects 0.000 description 1
- FRCCEHPWNOQAEU-UHFFFAOYSA-N heptachlor Chemical compound ClC1=C(Cl)C2(Cl)C3C=CC(Cl)C3C1(Cl)C2(Cl)Cl FRCCEHPWNOQAEU-UHFFFAOYSA-N 0.000 description 1
- 239000004009 herbicide Substances 0.000 description 1
- CKAPSXZOOQJIBF-UHFFFAOYSA-N hexachlorobenzene Chemical compound ClC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl CKAPSXZOOQJIBF-UHFFFAOYSA-N 0.000 description 1
- GAPJZAHIPBLCCF-UHFFFAOYSA-N hexadec-1-en-1-one Chemical compound CCCCCCCCCCCCCCC=C=O GAPJZAHIPBLCCF-UHFFFAOYSA-N 0.000 description 1
- CTTMBAVSCSLGMR-UHFFFAOYSA-N hexadeca-1,3,5-trienyl acetate Chemical compound CCCCCCCCCCC=CC=CC=COC(C)=O CTTMBAVSCSLGMR-UHFFFAOYSA-N 0.000 description 1
- FRZKUYZMXXQHSN-UHFFFAOYSA-N hexadeca-1,3-dienyl acetate Chemical compound CCCCCCCCCCCCC=CC=COC(C)=O FRZKUYZMXXQHSN-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- GOKKOFHHJFGZHW-UHFFFAOYSA-N hexyl propanoate Chemical compound CCCCCCOC(=O)CC GOKKOFHHJFGZHW-UHFFFAOYSA-N 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 229940056881 imidacloprid Drugs 0.000 description 1
- YWTYJOPNNQFBPC-UHFFFAOYSA-N imidacloprid Chemical compound [O-][N+](=O)\N=C1/NCCN1CC1=CC=C(Cl)N=C1 YWTYJOPNNQFBPC-UHFFFAOYSA-N 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- 239000003317 industrial substance Substances 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 239000002917 insecticide Substances 0.000 description 1
- ONUFESLQCSAYKA-UHFFFAOYSA-N iprodione Chemical compound O=C1N(C(=O)NC(C)C)CC(=O)N1C1=CC(Cl)=CC(Cl)=C1 ONUFESLQCSAYKA-UHFFFAOYSA-N 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- HOQADATXFBOEGG-UHFFFAOYSA-N isofenphos Chemical compound CCOP(=S)(NC(C)C)OC1=CC=CC=C1C(=O)OC(C)C HOQADATXFBOEGG-UHFFFAOYSA-N 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- 239000002949 juvenile hormone Substances 0.000 description 1
- 150000003893 lactate salts Chemical class 0.000 description 1
- CONWAEURSVPLRM-UHFFFAOYSA-N lactofen Chemical compound C1=C([N+]([O-])=O)C(C(=O)OC(C)C(=O)OCC)=CC(OC=2C(=CC(=CC=2)C(F)(F)F)Cl)=C1 CONWAEURSVPLRM-UHFFFAOYSA-N 0.000 description 1
- 239000005910 lambda-Cyhalothrin Substances 0.000 description 1
- 235000001510 limonene Nutrition 0.000 description 1
- 229940087305 limonene Drugs 0.000 description 1
- 229960002809 lindane Drugs 0.000 description 1
- SHTFZHTWSLHVEB-BDNRQGISSA-N lineatin Chemical compound O1C(C)(C)[C@H]2[C@@]3(C)C[C@@H]1O[C@@H]2C3 SHTFZHTWSLHVEB-BDNRQGISSA-N 0.000 description 1
- 239000004850 liquid epoxy resins (LERs) Substances 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 229960000453 malathion Drugs 0.000 description 1
- 230000007758 mating behavior Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000001683 mentha spicata herb oil Substances 0.000 description 1
- 229930007503 menthone Natural products 0.000 description 1
- GKKDCARASOJPNG-UHFFFAOYSA-N metaldehyde Chemical compound CC1OC(C)OC(C)OC(C)O1 GKKDCARASOJPNG-UHFFFAOYSA-N 0.000 description 1
- NNKVPIKMPCQWCG-UHFFFAOYSA-N methamidophos Chemical compound COP(N)(=O)SC NNKVPIKMPCQWCG-UHFFFAOYSA-N 0.000 description 1
- MEBQXILRKZHVCX-UHFFFAOYSA-N methidathion Chemical compound COC1=NN(CSP(=S)(OC)OC)C(=O)S1 MEBQXILRKZHVCX-UHFFFAOYSA-N 0.000 description 1
- UHXUZOCRWCRNSJ-QPJJXVBHSA-N methomyl Chemical compound CNC(=O)O\N=C(/C)SC UHXUZOCRWCRNSJ-QPJJXVBHSA-N 0.000 description 1
- 229950003442 methoprene Drugs 0.000 description 1
- 229930002897 methoprene Natural products 0.000 description 1
- GEPDYQSQVLXLEU-AATRIKPKSA-N methyl (e)-3-dimethoxyphosphoryloxybut-2-enoate Chemical compound COC(=O)\C=C(/C)OP(=O)(OC)OC GEPDYQSQVLXLEU-AATRIKPKSA-N 0.000 description 1
- BACHBFVBHLGWSL-UHFFFAOYSA-N methyl 2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate Chemical group C1=CC(OC(C)C(=O)OC)=CC=C1OC1=CC=C(Cl)C=C1Cl BACHBFVBHLGWSL-UHFFFAOYSA-N 0.000 description 1
- GEWDNTWNSAZUDX-UHFFFAOYSA-N methyl 7-epi-jasmonate Natural products CCC=CCC1C(CC(=O)OC)CCC1=O GEWDNTWNSAZUDX-UHFFFAOYSA-N 0.000 description 1
- ZQEIXNIJLIKNTD-UHFFFAOYSA-N methyl N-(2,6-dimethylphenyl)-N-(methoxyacetyl)alaninate Chemical compound COCC(=O)N(C(C)C(=O)OC)C1=C(C)C=CC=C1C ZQEIXNIJLIKNTD-UHFFFAOYSA-N 0.000 description 1
- 229960001047 methyl salicylate Drugs 0.000 description 1
- 229940116837 methyleugenol Drugs 0.000 description 1
- PRHTXAOWJQTLBO-UHFFFAOYSA-N methyleugenol Natural products COC1=CC=C(C(C)=C)C=C1OC PRHTXAOWJQTLBO-UHFFFAOYSA-N 0.000 description 1
- 229920000257 metiram Polymers 0.000 description 1
- FOXFZRUHNHCZPX-UHFFFAOYSA-N metribuzin Chemical compound CSC1=NN=C(C(C)(C)C)C(=O)N1N FOXFZRUHNHCZPX-UHFFFAOYSA-N 0.000 description 1
- 229960001952 metrifonate Drugs 0.000 description 1
- RSMUVYRMZCOLBH-UHFFFAOYSA-N metsulfuron methyl Chemical group COC(=O)C1=CC=CC=C1S(=O)(=O)NC(=O)NC1=NC(C)=NC(OC)=N1 RSMUVYRMZCOLBH-UHFFFAOYSA-N 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 230000000116 mitigating effect Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- DEDOPGXGGQYYMW-UHFFFAOYSA-N molinate Chemical compound CCSC(=O)N1CCCCCC1 DEDOPGXGGQYYMW-UHFFFAOYSA-N 0.000 description 1
- KRTSDMXIXPKRQR-AATRIKPKSA-N monocrotophos Chemical compound CNC(=O)\C=C(/C)OP(=O)(OC)OC KRTSDMXIXPKRQR-AATRIKPKSA-N 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- BUYMVQAILCEWRR-UHFFFAOYSA-N naled Chemical compound COP(=O)(OC)OC(Br)C(Cl)(Cl)Br BUYMVQAILCEWRR-UHFFFAOYSA-N 0.000 description 1
- 239000005645 nematicide Substances 0.000 description 1
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- RTCOGUMHFFWOJV-UHFFFAOYSA-N nicosulfuron Chemical compound COC1=CC(OC)=NC(NC(=O)NS(=O)(=O)C=2C(=CC=CN=2)C(=O)N(C)C)=N1 RTCOGUMHFFWOJV-UHFFFAOYSA-N 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen(.) Chemical compound [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 239000007764 o/w emulsion Substances 0.000 description 1
- NWRSOYAGXTXEMK-UHFFFAOYSA-N octadeca-2,13-dienyl acetate Chemical compound CCCCC=CCCCCCCCCCC=CCOC(C)=O NWRSOYAGXTXEMK-UHFFFAOYSA-N 0.000 description 1
- 239000001048 orange dye Substances 0.000 description 1
- UNAHYJYOSSSJHH-UHFFFAOYSA-N oryzalin Chemical compound CCCN(CCC)C1=C([N+]([O-])=O)C=C(S(N)(=O)=O)C=C1[N+]([O-])=O UNAHYJYOSSSJHH-UHFFFAOYSA-N 0.000 description 1
- KZAUOCCYDRDERY-UHFFFAOYSA-N oxamyl Chemical compound CNC(=O)ON=C(SC)C(=O)N(C)C KZAUOCCYDRDERY-UHFFFAOYSA-N 0.000 description 1
- FIKAKWIAUPDISJ-UHFFFAOYSA-L paraquat dichloride Chemical compound [Cl-].[Cl-].C1=C[N+](C)=CC=C1C1=CC=[N+](C)C=C1 FIKAKWIAUPDISJ-UHFFFAOYSA-L 0.000 description 1
- LCCNCVORNKJIRZ-UHFFFAOYSA-N parathion Chemical compound CCOP(=S)(OCC)OC1=CC=C([N+]([O-])=O)C=C1 LCCNCVORNKJIRZ-UHFFFAOYSA-N 0.000 description 1
- RLBIQVVOMOPOHC-UHFFFAOYSA-N parathion-methyl Chemical compound COP(=S)(OC)OC1=CC=C([N+]([O-])=O)C=C1 RLBIQVVOMOPOHC-UHFFFAOYSA-N 0.000 description 1
- CHIFOSRWCNZCFN-UHFFFAOYSA-N pendimethalin Chemical compound CCC(CC)NC1=C([N+]([O-])=O)C=C(C)C(C)=C1[N+]([O-])=O CHIFOSRWCNZCFN-UHFFFAOYSA-N 0.000 description 1
- LKPLKUMXSAEKID-UHFFFAOYSA-N pentachloronitrobenzene Chemical compound [O-][N+](=O)C1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl LKPLKUMXSAEKID-UHFFFAOYSA-N 0.000 description 1
- 229960000490 permethrin Drugs 0.000 description 1
- RLLPVAHGXHCWKJ-UHFFFAOYSA-N permethrin Chemical compound CC1(C)C(C=C(Cl)Cl)C1C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 RLLPVAHGXHCWKJ-UHFFFAOYSA-N 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- 230000002085 persistent effect Effects 0.000 description 1
- SBNFWQZLDJGRLK-UHFFFAOYSA-N phenothrin Chemical compound CC1(C)C(C=C(C)C)C1C(=O)OCC1=CC=CC(OC=2C=CC=CC=2)=C1 SBNFWQZLDJGRLK-UHFFFAOYSA-N 0.000 description 1
- 229960003536 phenothrin Drugs 0.000 description 1
- BULVZWIRKLYCBC-UHFFFAOYSA-N phorate Chemical compound CCOP(=S)(OCC)SCSCC BULVZWIRKLYCBC-UHFFFAOYSA-N 0.000 description 1
- IOUNQDKNJZEDEP-UHFFFAOYSA-N phosalone Chemical compound C1=C(Cl)C=C2OC(=O)N(CSP(=S)(OCC)OCC)C2=C1 IOUNQDKNJZEDEP-UHFFFAOYSA-N 0.000 description 1
- LMNZTLDVJIUSHT-UHFFFAOYSA-N phosmet Chemical compound C1=CC=C2C(=O)N(CSP(=S)(OC)OC)C(=O)C2=C1 LMNZTLDVJIUSHT-UHFFFAOYSA-N 0.000 description 1
- 125000005498 phthalate group Chemical class 0.000 description 1
- NQQVFXUMIDALNH-UHFFFAOYSA-N picloram Chemical compound NC1=C(Cl)C(Cl)=NC(C(O)=O)=C1Cl NQQVFXUMIDALNH-UHFFFAOYSA-N 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000005648 plant growth regulator Substances 0.000 description 1
- 244000000003 plant pathogen Species 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 230000007096 poisonous effect Effects 0.000 description 1
- 229920006122 polyamide resin Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920006124 polyolefin elastomer Polymers 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 150000003097 polyterpenes Chemical class 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 238000004313 potentiometry Methods 0.000 description 1
- 239000012254 powdered material Substances 0.000 description 1
- 230000002028 premature Effects 0.000 description 1
- 150000003139 primary aliphatic amines Chemical class 0.000 description 1
- AAEVYOVXGOFMJO-UHFFFAOYSA-N prometryn Chemical compound CSC1=NC(NC(C)C)=NC(NC(C)C)=N1 AAEVYOVXGOFMJO-UHFFFAOYSA-N 0.000 description 1
- WJNRPILHGGKWCK-UHFFFAOYSA-N propazine Chemical compound CC(C)NC1=NC(Cl)=NC(NC(C)C)=N1 WJNRPILHGGKWCK-UHFFFAOYSA-N 0.000 description 1
- 239000003380 propellant Substances 0.000 description 1
- BZNDWPRGXNILMS-VQHVLOKHSA-N propetamphos Chemical compound CCNP(=S)(OC)O\C(C)=C\C(=O)OC(C)C BZNDWPRGXNILMS-VQHVLOKHSA-N 0.000 description 1
- HTUIWRWYYVBCFT-UHFFFAOYSA-N propyl hexanoate Chemical compound CCCCCC(=O)OCCC HTUIWRWYYVBCFT-UHFFFAOYSA-N 0.000 description 1
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 229940070846 pyrethrins Drugs 0.000 description 1
- 239000002728 pyrethroid Substances 0.000 description 1
- OSUHJPCHFDQAIT-GFCCVEGCSA-N quizalofop-P-ethyl Chemical group C1=CC(O[C@H](C)C(=O)OCC)=CC=C1OC1=CN=C(C=C(Cl)C=C2)C2=N1 OSUHJPCHFDQAIT-GFCCVEGCSA-N 0.000 description 1
- BACHBFVBHLGWSL-JTQLQIEISA-N rac-diclofop methyl Natural products C1=CC(O[C@@H](C)C(=O)OC)=CC=C1OC1=CC=C(Cl)C=C1Cl BACHBFVBHLGWSL-JTQLQIEISA-N 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 239000005871 repellent Substances 0.000 description 1
- 230000002940 repellent Effects 0.000 description 1
- 230000001846 repelling effect Effects 0.000 description 1
- 230000033458 reproduction Effects 0.000 description 1
- 229940108410 resmethrin Drugs 0.000 description 1
- VEMKTZHHVJILDY-FIWHBWSRSA-N resmethrin Chemical compound CC1(C)[C@H](C=C(C)C)C1C(=O)OCC1=COC(CC=2C=CC=CC=2)=C1 VEMKTZHHVJILDY-FIWHBWSRSA-N 0.000 description 1
- 239000011369 resultant mixture Substances 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 239000003128 rodenticide Substances 0.000 description 1
- 239000010666 rose oil Substances 0.000 description 1
- 235000019719 rose oil Nutrition 0.000 description 1
- 229940080817 rotenone Drugs 0.000 description 1
- JUVIOZPCNVVQFO-UHFFFAOYSA-N rotenone Natural products O1C2=C3CC(C(C)=C)OC3=CC=C2C(=O)C2C1COC1=C2C=C(OC)C(OC)=C1 JUVIOZPCNVVQFO-UHFFFAOYSA-N 0.000 description 1
- 150000003902 salicylic acid esters Chemical class 0.000 description 1
- 235000020637 scallop Nutrition 0.000 description 1
- LSMIOFMZNVEEBR-ICLSSMQGSA-N scilliroside Chemical compound C=1([C@@H]2[C@@]3(C)CC[C@H]4[C@@]([C@]3(CC2)O)(O)C[C@H](C2=C[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O3)O)CC[C@@]24C)OC(=O)C)C=CC(=O)OC=1 LSMIOFMZNVEEBR-ICLSSMQGSA-N 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 239000004945 silicone rubber Substances 0.000 description 1
- ODCWYMIRDDJXKW-UHFFFAOYSA-N simazine Chemical compound CCNC1=NC(Cl)=NC(NCC)=N1 ODCWYMIRDDJXKW-UHFFFAOYSA-N 0.000 description 1
- XGVXKJKTISMIOW-ZDUSSCGKSA-N simurosertib Chemical compound N1N=CC(C=2SC=3C(=O)NC(=NC=3C=2)[C@H]2N3CCC(CC3)C2)=C1C XGVXKJKTISMIOW-ZDUSSCGKSA-N 0.000 description 1
- 230000036556 skin irritation Effects 0.000 description 1
- 231100000475 skin irritation Toxicity 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M sodium bicarbonate Substances [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 235000019721 spearmint oil Nutrition 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 235000020354 squash Nutrition 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid group Chemical class S(O)(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 230000035900 sweating Effects 0.000 description 1
- 230000009182 swimming Effects 0.000 description 1
- 239000006273 synthetic pesticide Substances 0.000 description 1
- 239000003760 tallow Substances 0.000 description 1
- WWJZWCUNLNYYAU-UHFFFAOYSA-N temephos Chemical compound C1=CC(OP(=S)(OC)OC)=CC=C1SC1=CC=C(OP(=S)(OC)OC)C=C1 WWJZWCUNLNYYAU-UHFFFAOYSA-N 0.000 description 1
- IROINLKCQGIITA-UHFFFAOYSA-N terbutryn Chemical compound CCNC1=NC(NC(C)(C)C)=NC(SC)=N1 IROINLKCQGIITA-UHFFFAOYSA-N 0.000 description 1
- OIAAITUGKVSGEF-UHFFFAOYSA-N tetradec-1-en-1-one Chemical compound CCCCCCCCCCCCC=C=O OIAAITUGKVSGEF-UHFFFAOYSA-N 0.000 description 1
- UECPLNNAVLEZGO-UHFFFAOYSA-N tetradec-1-enyl acetate Chemical compound CCCCCCCCCCCCC=COC(C)=O UECPLNNAVLEZGO-UHFFFAOYSA-N 0.000 description 1
- ZZGJZGSVLNSDPG-UHFFFAOYSA-N tetradeca-9,12-dienyl acetate Chemical compound CC=CCC=CCCCCCCCCOC(C)=O ZZGJZGSVLNSDPG-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- 238000005979 thermal decomposition reaction Methods 0.000 description 1
- 229920006345 thermoplastic polyamide Polymers 0.000 description 1
- 239000004308 thiabendazole Substances 0.000 description 1
- WJCNZQLZVWNLKY-UHFFFAOYSA-N thiabendazole Chemical compound S1C=NC(C=2NC3=CC=CC=C3N=2)=C1 WJCNZQLZVWNLKY-UHFFFAOYSA-N 0.000 description 1
- 229960004546 thiabendazole Drugs 0.000 description 1
- 235000010296 thiabendazole Nutrition 0.000 description 1
- 229960002447 thiram Drugs 0.000 description 1
- KUAZQDVKQLNFPE-UHFFFAOYSA-N thiram Chemical compound CN(C)C(=S)SSC(=S)N(C)C KUAZQDVKQLNFPE-UHFFFAOYSA-N 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- KHPCPRHQVVSZAH-UHFFFAOYSA-N trans-cinnamyl beta-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OCC=CC1=CC=CC=C1 KHPCPRHQVVSZAH-UHFFFAOYSA-N 0.000 description 1
- ZFDIRQKJPRINOQ-UHFFFAOYSA-N transbutenic acid ethyl ester Natural products CCOC(=O)C=CC ZFDIRQKJPRINOQ-UHFFFAOYSA-N 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- REEQLXCGVXDJSQ-UHFFFAOYSA-N trichlopyr Chemical compound OC(=O)COC1=NC(Cl)=C(Cl)C=C1Cl REEQLXCGVXDJSQ-UHFFFAOYSA-N 0.000 description 1
- NFACJZMKEDPNKN-UHFFFAOYSA-N trichlorfon Chemical compound COP(=O)(OC)C(O)C(Cl)(Cl)Cl NFACJZMKEDPNKN-UHFFFAOYSA-N 0.000 description 1
- ZSDSQXJSNMTJDA-UHFFFAOYSA-N trifluralin Chemical compound CCCN(CCC)C1=C([N+]([O-])=O)C=C(C(F)(F)F)C=C1[N+]([O-])=O ZSDSQXJSNMTJDA-UHFFFAOYSA-N 0.000 description 1
- RROQIUMZODEXOR-UHFFFAOYSA-N triforine Chemical compound O=CNC(C(Cl)(Cl)Cl)N1CCN(C(NC=O)C(Cl)(Cl)Cl)CC1 RROQIUMZODEXOR-UHFFFAOYSA-N 0.000 description 1
- 239000013638 trimer Substances 0.000 description 1
- JARYYMUOCXVXNK-IMTORBKUSA-N validamycin Chemical compound N([C@H]1C[C@@H]([C@H]([C@H](O)[C@H]1O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)CO)[C@H]1C=C(CO)[C@H](O)[C@H](O)[C@H]1O JARYYMUOCXVXNK-IMTORBKUSA-N 0.000 description 1
- 230000017260 vegetative to reproductive phase transition of meristem Effects 0.000 description 1
- DCSCXTJOXBUFGB-UHFFFAOYSA-N verbenone Natural products CC1=CC(=O)C2C(C)(C)C1C2 DCSCXTJOXBUFGB-UHFFFAOYSA-N 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 229960005080 warfarin Drugs 0.000 description 1
- PJVWKTKQMONHTI-UHFFFAOYSA-N warfarin Chemical compound OC=1C2=CC=CC=C2OC(=O)C=1C(CC(=O)C)C1=CC=CC=C1 PJVWKTKQMONHTI-UHFFFAOYSA-N 0.000 description 1
- 239000009637 wintergreen oil Substances 0.000 description 1
- DUBNHZYBDBBJHD-UHFFFAOYSA-L ziram Chemical compound [Zn+2].CN(C)C([S-])=S.CN(C)C([S-])=S DUBNHZYBDBBJHD-UHFFFAOYSA-L 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/02—Polyamines
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/60—Polyamides or polyester-amides
- C08G18/603—Polyamides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L9/00—Disinfection, sterilisation or deodorisation of air
- A61L9/01—Deodorant compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L9/00—Disinfection, sterilisation or deodorisation of air
- A61L9/01—Deodorant compositions
- A61L9/012—Deodorant compositions characterised by being in a special form, e.g. gels, emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L9/00—Disinfection, sterilisation or deodorisation of air
- A61L9/015—Disinfection, sterilisation or deodorisation of air using gaseous or vaporous substances, e.g. ozone
- A61L9/04—Disinfection, sterilisation or deodorisation of air using gaseous or vaporous substances, e.g. ozone using substances evaporated in the air without heating
- A61L9/042—Disinfection, sterilisation or deodorisation of air using gaseous or vaporous substances, e.g. ozone using substances evaporated in the air without heating with the help of a macromolecular compound as a carrier or diluent
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B3/00—Layered products comprising a layer with external or internal discontinuities or unevennesses, or a layer of non-planar shape; Layered products comprising a layer having particular features of form
- B32B3/26—Layered products comprising a layer with external or internal discontinuities or unevennesses, or a layer of non-planar shape; Layered products comprising a layer having particular features of form characterised by a particular shape of the outline of the cross-section of a continuous layer; characterised by a layer with cavities or internal voids ; characterised by an apertured layer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/48—Polyethers
- C08G18/50—Polyethers having heteroatoms other than oxygen
- C08G18/5021—Polyethers having heteroatoms other than oxygen having nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/77—Polyisocyanates or polyisothiocyanates having heteroatoms in addition to the isocyanate or isothiocyanate nitrogen and oxygen or sulfur
- C08G18/78—Nitrogen
- C08G18/79—Nitrogen characterised by the polyisocyanates used, these having groups formed by oligomerisation of isocyanates or isothiocyanates
- C08G18/791—Nitrogen characterised by the polyisocyanates used, these having groups formed by oligomerisation of isocyanates or isothiocyanates containing isocyanurate groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/50—Amines
- C08G59/54—Amino amides>
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
- C08L101/02—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
Definitions
- compositions and articles containing an active liquid intermixed with a polymer matrix are described herein.
- JP Q32558899A requires the use of a solid powder system
- JP07145299 requires the use of a pre-formed urethane-containing epoxy resin cross-linked in the absence of a polyamlne and/or an active liquid containing a perfume. Further, the above- mentioned JP references refer specifically and only to fragranced articles, such as air fresheners. Because of this narrow goal to make such articles, the reaction and reaction products described therein fail to have a dynamic range of performance capabilities, Moreover, they fail to provide a product that is durable in the absence of a support. Therefore, a need arises for controllable reaction conditions that yield dynamic reaction products containing durable matrices capable of immobilizing any and/or all types of active liquids therein.
- compositions such as fragrance objects, even more specifically air fresheners, are well known devices that release a fragrance into the air of a room of a house, area of a public building (e.g., a lavatory), or the interior of a car to render the air in that area more pleasing to the occupant.
- a fragrance for example, the thermoplastic polyamide-based products described in US Patent Nos. 6,111,655 and 6,503,577 and the thermo-set poly(armde ⁇ acid)s of US Patent No. 5,780,527 and US Patent No.
- 6,846,491 are homogeneous, transparent solids that can be easily charged, when in liquid form, to a mold and thus made into a visually attractive solid shape without the use of a means of support.
- the components must be heated to a temperature above the gelation temperature of the mixture, a process detrimental to the volatile and sometimes temperature sensitive active liquid such as fragrance, pesticide, or surfactant.
- these gels must not be exposed to low temperatures because they can turn unattractively cloudy.
- thermoplastic gels are soft solids that are easily deformed if scraped, dropped, poked, or wiped.
- these conventional gels do not provide compositions and/or articles that are readily durable and capable of operating at a wide range of operating parameters.
- Air care articles can contain a variety of fragrances.
- Aldehydes are common fragrance oil ingredients and can react with primary amines and interfere with polymer matrix setting times, especially for polymer matrices based on isocyanate-polyamine curing systems. It is not unusual that compositions fail to form the desired articles because aldehydes consume free primary amine groups.
- compositions and articles containing an active liquid intermixed with a polymeric matrix include a polymeric matrix comprising the reaction product of a polyamine and a compound having at least two functional groups and an active liquid intermixed with at least a portion of the polymeric matrix.
- the functional groups are selected from the group consisting of epoxy groups, isocyanate groups, anhydride groups, and acrylate groups.
- the polyamine and the compound are reacted in the presence of the active liquid.
- the polyamine is a polyamide polyamine and/or a secondary amine terminated polyamine.
- the reactive amine groups of the polyamine can include amino groups derived from at least one of ortho-aminobenzoic acid or para-aminobenzoic acid.
- the polyamine is a non-water soluble polyamide polyamine with a molecular weight in the range of 4,000 to 30,000 Daltons.
- the active liquid is present in an amount from 10 weight % to 85 weight % based on the weight of the composition (e.g., from 50 weight % to 85 weight % based on the weight of the composition).
- the active liquid can include, for example, a therapeutic active liquid, a nutraceutical active liquid, a cosmeceutical active liquid, a pesticidal active liquid, a laundry care active liquid, a fragrance, or a mixture thereof.
- the compound includes at least one non-aromatic isocyanate compound.
- composition described herein can be in the form of a gel.
- the composition can be in the form of a particle and can be present in an aqueous dispersion.
- the particle size can be, for example, from 1 micron to 100 microns (e.g., from 2 microns to 15 microns).
- articles comprising a porous support material and the composition described herein.
- the methods can include reacting a polyamine with a compound having at least two functional groups, the functional groups selected from the group consisting of epoxy groups, isocyanate groups, anhydride groups, and acrylate groups in the presence of an active liquid,
- the polyamine is liquid at room temperature.
- the polyamine has an amine number of from 10 meq KOH g to 100 meq KOH/g.
- the polyamine can have a viscosity of 500cP or less at 150°C, In some examples, the reacting step occurs at room temperature.
- compositions and/or articles containing an active liquid-intermixed polymeric matrix and methods for their preparation and use are described herein.
- the polymeric matrix can be a thermoset (i.e., a cross-linked) polymeric matrix that includes an active liquid intermixed within the matrix.
- the active liquid is uniformly (i.e., homogenousiy) intermixed within the matrix.
- the polymeric matrices described herein are durable and stable over a wide range of conditions,
- the polymeric matrix is the reactio product of a polyamine and a compound having at least two functional groups selected from the group consisting of epoxy groups, isocyanate groups, anhydride groups, and acrylate groups.
- the polyamine can include a polyamide polyamine (PAPA) and/or a secondary amine terminated polyamine (SATPA),
- PAPA polyamide polyamine
- SATPA secondary amine terminated polyamine
- the reaction is carried out in the presence of the active liquid. A small amount of water can be intermixed as a part of the active liquid.
- the composition can then be dispersed in an aqueous phase in the form of a particle dispersion.
- the compound having at least two functional groups selected from the group consisting of epoxy groups, isocyanate groups, anhydride groups, and acrylate groups can be, for example, crosslinking agents.
- the crosslinking agent is an epoxy crosslinking agent (i.e., a compound having at least two functional groups that include an epoxy group).
- the epoxy crosslinking agent can be any epoxy.
- the epoxy crosslinking agent is in the form of a liquid. Examples of liquid epoxy resins that can be used in the compositions described herein include diglycidyl ethers of bisphenol A and F, commercially available as EPON 828 and EPON 8620 from Resolution Performance Products (Houston, TX);
- the compound having at least two functional groups described herein can be a compound including at least two anhydride functional groups (i.e., a polyanhydride).
- the polyanhydride is in the form of a liquid.
- the anhydride can be a solid polymer dissolved in a suitable carrier liquid.
- the polyanhydride is not a maleated polyolefin rubber. Examples of polymers for the anhydrides include, for example, maleated olefin polymers other than a maleated rubber (e.g., a polybutadiene or a
- poly(isobutylene)), olefin-maleic anhydride co-polymers, and aipha-olefin-maleic anhydride alternating co-polymers include styrene-maleic anhydride copolymers such as DYLARK 232 and DYLARK 332, available from NOVA Chemicals (Moon Township, PA), and poly(l-octadecene-alt-maleic anhydride), commercially available from Chevron Corporation (San Ramon, CA). These anhydride- containing polymers are representative and many additional anhydride-containing polymers are applicable.
- the compound having at least two functional groups described herein can be a compound including at least two isocyanate functional groups (i.e., a polyisocyanate).
- the polyisocyanate is in the form of a liquid.
- the compound includes at least one non-aromatic isocyanate compound.
- Specific examples of the isocyanate-containing compounds include aliphatic difunctional isocyanate materials such as liquid diisocyanates (e.g., isophorone diisocyanate and bis(4-isocyanato cyclohexyl) methane).
- the polyfiinctional isocyanates can have low volatility and reduced toxicity.
- isocyanates examples include the DESMODUR N-series aliphatic isocyanurates (e.g.. DESMODUR N- 3300, DESMODUR N-3600, and DESMODUR N-380Q), and the DESMODUR. Z-series (e.g., DESMODUR Z4470), all commercially available from Bayer Corporation, industrial Chemicals Division (Pittsburgh, PA). These isocyanate-containing compounds are representative and additional isocyanate-containing compounds are applicable. In some embodiments, the equivalent weight for the isocyanate-containing compounds is in the range of 180 to 500.
- a polyamine compound for reaction with epoxy-functional compounds can be a liquid at room temperature (e.g., 25°C); can dissolve in, and can be compatible with, many active liquids; can have a viscosity, measured at 100°C, of no greater than about 100 cP; and can have an amine number of from 100 to 1200 meq KOH/g.
- the amine number can be 100, 200, 500, 750, 1000 and 1200 meq KOH/g, including any and all ranges and subranges there between.
- Suitable polyamines include, for example, 1,2- diaminocyclohexane, isophorone diamine, meta-xylene diamine, and 1,3- bis(aminomethyl)cyclohexane (1,3-BAC).
- the polyamines can be
- poly(alkyleneoxy) polyamines i.e., polyether amines
- polyether amines that are liquid at 25°C and include polyether segments such that greater than 50% by weight of the amine is derived from a polyether.
- the polyether can be an oligomerized ethylene oxide, propylene oxide, butylenes oxide, tetrahydrofuran, or combinations of these supplied by, for example, Huntsman Corporation (The Woodlands, TX) and BASF Corporation (Florham Park, NJ).
- suitable polyamines include, for example. JEFF AMINE D-230, D-400, D-2000, T-5000, T-403. and XT J511 XTJ-51 1 , all polyether diamines commercially available from Huntsman
- Liquid polyamines can also be chosen from the polyamido- amine family, examples of which are the UNIREZ series of amidoamide-amine curing agents commercially available from Arizona Chemical (Jacksonville, FL). These materials are known to impart adhesion and have lowered skin sensitivity.
- the amines can be mixtures of two or more amines blended to optimize viscosity, reaction rate and product performance.
- the polyamine compound suitable for reaction with isocyanate- functional compounds can be a material having a polymeric backbone comprising repeating monomer units terminated by amine groups that are different from the repeating amine groups.
- This polymeric polyamine can be a liquid at a temperature below 50 °C 3 e.g., a liquid or low melt point amine.
- the polyamine can be a liquid at normal room temperature.
- the amine has a melting or softening point at or below 50°C, (e.g., 45 °C, 40 °C, 30 °C, 20 °C, and 10 °C 3 including any and all ranges and subranges there between).
- the polyamine is a liquid and/or tacky and/or a semisolid at a temperature below 10 °C.
- the polymeric polyamine dissolves in, and is compatible with, many active liquids; has a number-average molecular weight of greater than 1,000; has an amine number of from 10 to 100 meq KOH/g; and has a viscosity, measured at 150 °C, of no greater than about 500 cP.
- the amine number can be, for example, 10, 25, 50, 75, or 100 meq KOH/g, including any and all ranges and subranges there between.
- the viscosity, measured at 150 °C, of the polyamine can be 500 cP or less.
- the viscosity, measured at 150 °C, of the polyamine can be about 450 cP, 350 cP, 250 cP, 150 cP, and 100 cP, including any and all ranges and subranges there between.
- the polymeric polyamine for reacting with isocyanate-functional compounds can be a polyamide polyamine (or "PAPA").
- the polyamide polyamines can be polyamide polyether block copolymers resulting from the reaction of one or more
- polyalkyleneoxy polyamines with one or more aliphatic polyacids as further described below.
- ether-based polyamide polyamines can be made by reacting a polyacid or mixture of polyacids with a stoichiometric excess of polyether polyamine admixed with optional lower diamines including piperazine, ethylene diamine, isophorone diamine, hexamethylene diamine, 2-methyl-l ,5-pentane diamine, and the like.
- Suitable polyacids for the preparation of PAPAs are adipic acid, azeleic acid, sebacic acid, dodecandioic acid or other aliphatic diacid or its ester equivalent.
- the polyamide polyamine is not soluble in water.
- the amine number of the PAPA can be less than 100, as measured by titration with dilute alcoholic hydrochloric acid and expressed as mg KOH g sample. In some examples, the amine number of the PAPA is less than 80 mg KOH/g or less than 70 mg KOH/g.
- PAPAs include the reaction products of polymerized fatty acids, also known as dimer acids (e.g., material produced by Arizona Chemical Company under the trade name "UNIDYME”; Unichema Corporation (Wilmington, DE) under the name “PRIPOL”; and Cognis Corporation (Cincinnati, OH) under the trad name “EMPOL”) and a stoichiometric excess of one or more poly(alkyleneoxy) polyamines chosen from the group of Huntsman JEFFAMINE polyamines, including, for example, D-400, D-2000, T-403, and XTJ-500.
- dimer acids e.g., material produced by Arizona Chemical Company under the trade name "UNIDYME”; Unichema Corporation (Wilmington, DE) under the name “PRIPOL”; and Cognis Corporation (Cincinnati, OH) under the trad name "EMPOL”
- a stoichiometric excess of one or more poly(alkyleneoxy) polyamines
- the resulting polymeric polyamines can be liquid at room temperature, have an acid value of less than about 5 and an amine value of from about 10 to about 70; and have a viscosity of less than 500 cP measured at 150 °C.
- the PAPA is liquid at room temperature, has an acid value of less than 2 and an amine value of 20-60, and has a viscosity of less than 300 cP at 150 °C.
- a polymeric polyamine can be obtained by reacting 29.5 weight % of PRiPOL 1009 hydrogenated dimer acid, 44.5 weight % of JEFFAMINE D- 2000, 22.5 weight % of JEFFAMINE® D-400, and 3.5 weight % of JEFFAMINE T-403 at 215°C under a sweep of dry nitrogen until the acid number drops to about 1.0 and the amine value is adjusted to be about 30-40.
- the resulting material can be, for example, a viscous liquid at room temperature with a viscosity of about 100 cP at 130 °C and a weight average molecular weight of about 25,000 Daltons.
- Reaction rates for forming the matrix vary with the type of terminal amine present in the polymeric polyamine component.
- the shortest cure times result from the use of a compound whose polymer chain terminates in an aliphatic primary or secondary amine. Amines hindered by substitution with a bulky group such as a tertiary butyl moiety react more slowly.
- the longest cure times result from the use of a polymeric polyam ine terminated with a certain type of aromatic amine whose aromatic ring bears a carbonyl, particularly an ester or amide group, or other strong electron-withdrawing group. While it is believed that the carbonyl-substituted aromatic amines can be utilized for reaction with any of the functional groups described herein, they are particularly useful when the functional group is the highly-reactive isocyanate group.
- polyamines are those derived from para-aminobenzoic acid and ortho-aminobenzoic acid. These compounds are readily incorporated onto the termini of polyamides described herein by reaction with the specified polyamines along with the specified diacids.
- a PAPA can include, for example, a polymer produced by reacting any of the above-described diacids and ether diamines in the presence of para-amino benzoic acid and/or ortho-amino benzoic acid.
- a PAPA can be obtained by reacting 24.0 weight% PRIPOL 1009 hydrogenated dimer acid, 5.0 weight % para-aminobenzoic acid, 54.0 weight % JEFFAMINE D-2000, 11.5 weight % JEFFAMINE D-400, and 5.5 weight % JEFFAMINE® T-403 at 215 °C under a sweep of dry nitrogen until the acid number drops to about 1.0 and the amine value is adjusted to 15 by non- potentiometric titration and 30-35 by potentiometric titration.
- This material is a viscous liquid at room temperature with a viscosity of about 250 cP at 130°C and a weight average molecular weight of about 13,000 Daltons,
- the weight-average molecular weight (Mw) and/or number-average molecular weight (Mn) of the PAPA can be as high as desired but can be limited by the desired amine value and viscosity.
- the Mw can be in the range of 3000-40,000 Daltons and can be greater than 3000, 4000, 5000, 6000, 7000, 8000, 9000, or 10,000 and/or less than 40,000, 38,000, 36,000, 34,000, 32,000 or 30,000 Daltons.
- the polydispersity can be any value but is desirably greater than 1.5 and less than 6, or in the range 2,0-4.0, including any and all ranges and subranges there between.
- Co-diacids and co-diamines can be used to prepare PAPAs described herein in an amount of less than 50% on an equivalents basis.
- Co-diacids can be, for example, adipic acid and similar linear aliphatic diaeids.
- Co-diamines can include, for example, ethylene diamine, piperazine, 1,2-diaminocyclohexane, isophorone diamine, l ,3-bis(aminomethyl)cyclohexane, dimer diamine (e.g., VERSAMI E 551, commercially available from Cognis Corporation (Cincinnati, OH)), hexamethylene diamine, 2 -methyl -1,5 -pentane diamine, and similar linear, branched and cyclic aliphatic diamines.
- the polyamidification reaction can be carried out in the presence of catalysts known to increase the reaction rate such as acids, particularly para-toluene sulfonic, phosphoric and sulfuric acids, and with removal of water of reaction via application of a vacuum.
- Suitable PAPAs further include those that are not liquid at room temperature.
- the non-liquid PAPAs can be solid at room temperature (e.g., low melting
- polyamines can result from the reaction of a major diacid portion of 1,4- cyclohexane dicarboxylic acid and a stoichiometric excess of polyamine, the majority of which is a poly(alkyleneoxy) polyamine chosen from the group of Huntsman JEFF AMINE® polyamines, including, for example, D-400, D-2000, T-403, and XTJ-500 such that, after the reaction is complete, the PAPA is a solid at 25 °C, has an acid value of less than 5, has an amine value of from about 10 to about 70, and has a Ring & Ball softening point less than 50 °C.
- the dimer acid can be used as a co-diacid along with other co-diacids such as those mentioned above.
- Co-diamines can also be used to prepare the PAPAs described herein.
- polymeric polyamines for use in the compositions and articles described herein include those described in U.S. Patent Nos. 6,399,713, 6,870,01 1 ; and 6,956,099, which are incorporated, in their entireties, herein by reference.
- the polyamine is a secondary amine terminated polyamine (SATPA).
- SATPA secondary amine terminated polyamine
- the amine number of the SATPA can be 100 meq KOH/g or less.
- the SATPA can have an amine number from 10 to 100 meq KOH/g.
- the composition can be, for example, a reaction product of a secondary amine terminated polyamine (SATPA) and an isocyanate cross- linking agent in the presence of an active liquid to be intermixed.
- the gel composition can be prepared by blending the SATPA, the liquid actives, and the cross-linking agent.
- the form of the composition can depend on the reactants used to form the polymeric matrix.
- the polymeric matrix can include the reaction product of a secondary amine terminated polyamine and a compound having at least two functional groups selected from the group consisting of epoxy groups, isocyanate groups, anhydride groups, and acrylate groups.
- the polymeric matrix can include the reaction product of a secondary amine terminated polyamine and a compound having at least two isocyanate functional groups.
- the compositions can be in the form of a gel (e.g., a clear, crosslinked polymeric gel).
- the polymeric matrix can include the reaction product of a polyamide polyamine and the compound having the at least two functional groups as described above.
- the resulting compositions can be in the form of a particle (e.g., a particle in an aqueous dispersion).
- the particle size of the particles can be from 1 micron to 100 microns, for example, from 2 microns to 15 microns.
- the particle size of the particles can be 3 microns, 4 microns, 5 microns, 6 microns. 7 microns, 8 microns, 9 microns, 10 microns, 11 microns, 12 microns, 33 microns, or 14 microns.
- the reaction to produce the polymeric matrix is carried out in the presence of an active liquid.
- the resulting polymeric matrix includes the active liquid intermixed with at least a portion of the matrix and, in some embodiments, throughout the matrix.
- the active liquid can be any liquid that imparts a function upon the resultant composition and/or article a function.
- the active liquid can be a volatile or nonvolatile organic liquid.
- the active liquid can be a semi-solid or a solid dissolved in a carrier liquid (e.g., a diluent).
- suitable active liquids include therapeutic active liquids, nutraceutical active liquids, cosmeceutical active liquids, pesticidal active liquids, laundry care active liquids, fragrance oils, surface treating chemicals, radiotracers, surfactants, or a mixture of these.
- the active liquid can be a fragrance oil (i.e., a scent or perfume).
- a fragrance oil can be any blend of the large number of synthetic aroma chemicals and aromatic natural oils known to one of skill in the art. Examples of useful classes of chemicals include esters such as linalool acetate and butyl acetate (present in banana oil), phenols such as methyl salicylate (present in oil of wintergreen), ethers such as 1,8-cineole (present in eucalyptus oil), alcohols such as geraniol (present in rose oil), ketones such as menthone (present in spearmint oil), and aldehydes such as cinnamaldehyde (present in cinnamon oil).
- aldehydes include citral, benzaldehyde, p-alkyl-substituted benzaldehydes, anisaldehyde, vanillin, he!iotropin, and alkyl-substituted cinnamic aldehydes.
- aldehydes may react with primary amines and interfere with polymer matrix setting times, especially for polymer matrices based on isocyanate-polyamine curing systems. While not wishing to be limited to theory, it is believed that secondary amines do not react with aldehydes because they do not have a proton available. Thus, the aldehydes in fragrances do not interfere with secondary amine cross! inking agent in preparation of the compositions and articles described herein. The interference from aldehydes in fragrances can be eliminated, which also leads to more consistent products and efficient manufacturing.
- fragrance types can be used to prepare the intermixed fragrance oils with high fragrance loading (e.g., greater than 50%, greater than 55%, greater than 60%, greater than 65%, greater than 70%, greater than 75%, greater than 80%, or greater than 85%).
- fragrance oils useful for the compositions described herein are Ocean, Country Wildflower, Spring Meadow, and Morning Rain, supplied by Continental Aromatics (Hawthorne, NJ); Macintosh supplied by Or!andi. Inc. (Farmingdale, NY); Evergreen, Green Apple, and Yankee Home supplied by Belle Aire Fragrances (Mundelein, IL); Cherry, Vanilla, Downey, and Mulberry supplied by Aromatic Fragrances and Flavors International (Marietta, GA); Gamet supplied by International Fragrances Technology, Inc.
- the active liquid can be used at a level so as to impart efficacy to the composition for the intended application.
- the active ingredient can be extremely potent and need be present only in a very low level, e.g., less than 0.1%. In such a case, the active liquid is said to be the solution of potent agent in carrier.
- the active liquid (or potent agent dissolved in carrier) can be used in the compositions and/or articles at levels from 1% for lightly-loaded objects to 90% or more.
- the loading can depend on the function of the particular active liquid, polymer matrix, and any other compounds present, it can also depend upon the final configuration of the formed product, that is, whether it is free-standing, contained, or supported.
- the active liquid can be present in an amount of from 10 weight % to 85 weight % or from 50 weight % to 85 weight %.
- the amount of active liquid can be 1%, 2%, 5%, 10%, 15%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, or 90% inclusive of all ranges and subranges there between.
- the fragrance oil level for air fresheners can be from 15-75% (e.g., from 30-70%) by weight of any embedded objects.
- the amount of fragrance oil can be 15%, 20%, 25%, 30%, 4G%, 50%, 60% or 75% by weight of the composition (not counting the weight of any supports or embedded objects) inclusive of all ranges and subranges there between.
- Inactive diluent or plasticizer can be present in an additional amount such that the total liquid level can be from 20% to 90% by weight of the composition, for example, from 40% to 80% by weight of the composition.
- the mixture of reactive components, active liquid and optional liquids, while still uncured, can be dispersed in water or other aqueous medium and the resulting oil-in-water emulsion stabilized by means of a surfactant.
- a surfactant can be anionic, cationic, or non-ionic in nature.
- Examples include the anionic salt sodium lauryl sulfate, the cationic quaternary ammonium salts di(hydrogenated tallow) dimethyl ammonium chloride, cocamido propyl betaine, and dibenzyl dimethyl ammonium chloride, and the non-ionic polyethoxylated sorbitan mono-oleate.
- Such an emulsion is a milky liquid and can, as such, be impregnated into a porous medium such as paper, cardboard, cellulose pad, cellulose pulp, felt, fabric, a porous synthetic foam, a porous ceramic, activated carbon, soil, diatomaceous earth, kieselguhr, charcoal, silica, clay, and the like or coated onto a non-porous substrate included but not limited to plastic films, metallic foils, rubber, ceramics, wood, glass, and leather.
- a porous medium such as paper, cardboard, cellulose pad, cellulose pulp, felt, fabric, a porous synthetic foam, a porous ceramic, activated carbon, soil, diatomaceous earth, kieselguhr, charcoal, silica, clay, and the like or coated onto a non-porous substrate included but not limited to plastic films, metallic foils, rubber, ceramics, wood, glass, and leather.
- Surfactant compounds can themselves be active compounds when used in excess of the amount needed to stabilize the gel dispersion.
- the surfactants can be used with or without water. Surfactants thus intermixed within the polymeric matrix are released slowly into their use environment along with the fragrance and other active components, and can thus serve as, for example, a toilet air freshener/cleaner, a pesticide/disinfectant, or a fabric softener in a laundry dryer either in the form of a liquid or, if impregnated into a porous medium, a sheet.
- the active liquid can be a liquid pesticide or a solid pesticide dissolved in a carrier liquid.
- pesticide refers to any substance or mixture of substances intended for preventing, destroying, repelling, or mitigating any organism that causes or is able to cause harm or annoyance to humans, valuable animals (e.g., livestock), or valuable plants
- Pesticides include chemical substances or biological agents (such as viruses or bacteria) used to control insects, plant pathogens, weeds, mollusks, birds, mammals, fish, nematodes (roundworms) and microbes that compete with humans for food, destroy property, spread disease, or are a nuisance. Because many pesticides are poisonous to humans, it is useful to control their application and release by, for example, dissolving them in a harmless carrier liquid and then intermixing and immobilizing them within the polymeric matrix.
- the pesticides can be naturally derived or synthetic. Examples of synthetic pesticides include organophosphates, carbamates, organochlorines, and pyrethroids,
- Organophosphates and carbamates can affect the nervous system by disrupting the enzyme that regulates acetylcholine, a neurotransmitter. They usually are not persistent in the environment. Immobilization by intermixing, then, can help the organophosphates to be effective for a longer period of time without harming the environment, Organochlorines (e.g., DDT and chlordane) were commonly used in the past, but many have been removed from the market due to their health and environmental effects and their persistence. Pyrethroids were developed as synthetic versions of the naturally occurring pyrethrin to increase stability in the environment and lower their cost.
- Organochlorines e.g., DDT and chlordane
- Some pesticides are derived from such natural materials as animals, plants, bacteria, an example being the naturally-occurring material, pyrethrin, extracted from chrysanthemums.
- Biopesticides include microbial pesticides that consist of a microorganism (e.g., a bacterium, fungus, virus or protozoan) as the active ingredient.
- Microbial pesticides can control many different kinds of pests, although each separate active ingredient is relatively specific for its target pest. For example, there are fungi that control certain weeds, and other fungi that kill specific insects.
- the most widely used microbial pesticides are subspecies and strains of Bacillus thuringiensis, or Bt.
- Pesticides can be classified according to the type of pest that they combat.
- useful pesticides include algicides that control algae in lakes, canals, swimming pools, water tanks, and other sites; antifouling agents that kill or repel organisms that attach to underwater surfaces, such as boat bottoms; antimicrobials that kill microorganisms (such as bacteria and viruses); attractants that attract pests (for example, to lure an insect or rodent to a trap) including foods such as sugar; biopesticides that are active agents derived from natural materials such as animals, plants, bacteria, and certain minerals; biocides that kill microorganisms, disinfectants and sanitizers that kill or inactivate disease-producing microorganisms on inanimate objects, fungicides that kill fungi (including blights, mildews, molds, and rusts); herbicides that kill weeds and other plants that grow where they are not wanted; insecticides that kill insects and other arthropods, miticides (also called acaricides) that
- microorganisms moliuscicides that kill snails and slugs; nematicides that kill nematodes (microscopic, worm-like organisms that feed on plant roots); ovicides that kill eggs of insects and mites; pheromones that disrapt the mating behavior of insects; repellents that are chemicals that repel pests, including insects (such as mosquitoes) and birds from a surface such as skin or seeds; rodenticides that sicken, repel, or kill mice and other rodents; insect growth regulators that disrupt the molting, maturity from pupal stage to adult, or other life processes of insects; and plant growth regulators that are substances (excluding fertilizers or other plant nutrients) that alter the expected growth, flowering, or reproduction rate of plants.
- Liquid pheromones or solid pheromones dissolved in a carrier liquid can also be intermixed with the polymeric matrixes described herein to produce, for example, articles that can serve as baits or lures in insect traps, fishing lures, rodent traps, and the like, Pheromones are typically six-to-twenty carbon atom esters, aldehydes, alcohols and ketones and for that reason resemble fragrance compounds and can be immobilized as described earlier for fragrance compounds. There are many hundreds of such compounds identified for many animal and insect species, many of which are not considered pests.
- Representative examples that can be used in the articles described herein include, for example, E or Z-13-octadecenyl acetate; E or Z-l 1- hexadecenal; E or Z-9-hexadecenal; hexadecanal: E or Z-l 1 hexadecenyl acetate; E or Z-9- hexadecenyl acetate; E or Z-l 1-tetradecenal; E or Z-9-tetradecenai; tetradecanal; E or Z-l 1- tetradecenyl acetate; E or Z-9-tetradecenyl acetate; E or Z-7-tetradecenyl acetate; E or Z-5- tetradecenyi acetate; E or Z-4-tridecenyl acetate; E or Z-9-dodecenyl acetate; E or Z-8 dodecenvl acetate; E
- Specific pheromones include the following: 8-methyl-2-decyl- propionate; 14-methyl-l-octadecene; 9-tricosense; tridecenyl acetate; dodecyl acetate; dodecenyl acetate; tetradecenyl acetate; tetradecadienyl acetate; hexadecenyl acetate; hexadecadienyl acetate; hexadecatrienyl acetate; octadecenyl acetate; dodecadienyl acetate; octadecadienyl acetate; and Z,E-9, 12-tetradecadiene- 1 -oi.
- the active liquid can be a liquid form of the active ingredient, or can be a solid, liquid or gaseous form of the active ingredient that is dissolved (contained) and diluted by a carrier liquid (diluent).
- the active liquid can include or consist of water and an active agent dissolved in the water.
- the active liquid can include or consist of an organic liquid and an active agent dissolved in the liquid.
- active ingredients contained in the active liquid can be therapeutically active ingredients (for humans or animals) such as medicines, drugs, pharmaceuticals, bioceuticals which are optionally combined with a biologically-acceptable carrier.
- active ingredient contained in the active liquid can be biological compound such as amino acids, vitamins, carbohydrates, and/or steroids.
- biological compounds include biopolymers, biocopolymers, or chimera comprising DNA, RNA,
- oligonucleotides modified DNA, modified RNA, proteins, polypeptides, and modified polypeptides.
- Additional components for use in the polymeric matrixes include, for example, plasticizers, diluents, accelerators, retardants, tackiflers, fillers, and colorants. Phthalates, benzoates, salicylates, and lactate esters, alcohols, polyols, poly(alkylene glycol)s and alkyl and aryl ethers of alcohols, polyols and poly(alkyiene glycols) are examples of useful
- plasticizers/diluents These increase product flexibility, improve active release, and lower product cost.
- Reactive diluents and inert diluents can also be used to lower the initial blend viscosity.
- Possible diluents include, but are not limited to, various mono- and diglycidyl ethers, glycols, and N-methyl pyrolidinone.
- Phenols such as nonyl phenol and 2,4,6- tris(dimethylaminomethyl) phenol, are examples of known accelerators of the epoxy-amine curing reaction that can shorten the time needed to cure the air fresheners described herein.
- Reaction accelerators include, for example, any alcohol-containing compound and/or water and/or mixtures thereof.
- resins such as rosin esters and polyterpenes can be dissolved in the epoxy or the diluent plasticizer to add tack to the final product.
- suitable resins include SYLVATAC, SYLVARES, and SYLVALITE, commercially available from Arizona Chemical (Jacksonville, FL).
- the compositions and/or articles described herein can be made by reacting (for example, by contacting, mixing, or blending) a compound having at least two functional groups selected from epoxy, isocyanate, anhydride, and acrylate with a polyamine in the presence of an active liquid.
- the resultant mixture prior to and after curing, can be homogeneous.
- Such contacting, mixing, and blending of the reactive components and active liquid can occur at a temperature from 10-50 °C.
- the reacting step occurs at room temperature.
- the reacting step can occur, for example, at 10 °C, 15 °C, 20 °C, 25 °C, 30 °C, 35 °C, 40 °C, 45 °C, or 50 °C, inclusive of all ranges and subranges there between.
- the components and optional ingredients can be added in any order.
- the active liquid is added before the matrix-forming reaction proceeds to a point where its high viscosity and increasing elasticity precludes a blending operation.
- the amine is a solid, it can first be dissolved in diluent liquid, in the active liquid, or in a mixture of both.
- Temperature and blending conditions can be controlled so as to preclude premature curing, that is extensive curing during the contacting, mixing, or blending step.
- the mixture can become a homogeneous thermoset solid thereafter.
- Curing temperatures can differ from blending operation temperatures and can be in the range of from 10-100 °C, for example, 10 °C, 20 °C, 30 °C, 40 °C, 50 °C, 60 °C, 70 °C, 80 °C, 90 °C, and 100°C, inclusive of all ranges and subranges there between.
- Curing rate is a function of at least six factors: curing temperature, functional group and amine group concentrations, ratio of these, structure of the amine, accelerator/retardant concentration, and composition of the fragrance oil/diluent. Accordingly, cure times can vary widely.
- a low temperature procedure can include blending at room temperature, pouring the blend into a moid, sealing it, and allowing the blend to stand at room temperature.
- a procedure can take from a few minutes to a few days depending on the functional groups chosen and the reaction conditions.
- the isocyanate-amine matrix reacts significantly faster than the epoxy-amine matrix.
- a pre-curing procedure useful more for the epoxy-amine matrix can include blending at room temperature, sealing tightly, heating to 70 °C for 30-90 minutes to obtain a partial cure but not gelling the composition, then pouring the resultant partial cure into a mold, letting it cool and stand at room temperature.
- Such a procedure can take from an hour to two days.
- Another example is a high temperature procedure which can include blending at room temperature, pouring into a pouch or mold, sealing it tightly, and heating it to a temperature ranging from 60 to 100 °C. Such a procedure can take from a few minutes to a few hours.
- the curing time can range from 0.01 hour to 60 hours (e.g., from 5 minutes to 20 hours or from 10 minutes to 100 minutes). In some examples, the curing time can be 10 hours, 20 hours, 30 hours, 40 hours, 50 hours, 60 hours, 70 hours, 80 hours, 90 hours, or 100 hours, inclusive of all ranges and subranges there between.
- the method includes blending an active liquid, a liquid polyepoxy, and a liquid poiyamine to form a mixture. Blending the components can occur at 10-40 °C.
- the blending can be performed so as not to cause a loss of any temperature-sensitive active component.
- the temperature of blending can be 10 °C, 15 °C, 20 °C, 25 °C, 30 °C, 35 °C, or 40 °C, inclusive of all ranges and subranges there between.
- the temperature of curing can be room temperature, i.e. 25 °C, but can be higher, depending on the temperature sensitivity of the active liquid component and its volatility. if the active liquid does not degrade readily and the curing is performed in a sealed mold, the curing temperature, for example, can be about 60 °C. At this temperature, curing for a typical formulation takes place in about 3-6 hours, or less if an accelerator is used.
- the methods described herein include blending an active liquid, a liquid diluent, a liquid polyisocyanate, and a liquid poiyamine to form a mixture that cures to a liquid-immobilized polyurea composition.
- Blending the components can occur, for example, at 10-40 °C. However, the blending can be performed so as not to cause a loss of any temperature- sensitive active component.
- the temperature of blending can be 10 °C, 15 °C, 20 °C, 25 °C, 30 °C, 35 °C, or 40°C, inclusive of all ranges and subranges there between.
- a catalyst is not present in the reaction between a poiyamine and an isocyanate.
- a rate modifier i.e., a reiardant
- Useful rate modifiers include, for example, aldehydes such as those normally present in common essential oils and fragrance oils.
- Other rate modifiers include those that are either bland in odor or those that can enhance the odor of the active liquid.
- useful retardants include aromatic aldehydes such as benzaldehyde, vanillin, and salicylaldehyde; , ⁇ unsaturated aromatic aldehydes such as cinnamic aldehyde and methyl cinnamic aldehyde; terpenic aldehydes such as citral, cyclocitral, and citronellal; and C 4 -Cig aliphatic and cycloaliphatic aldehydes such as isobutyraldehyde, lyral, 2-phenyl propionaldehyde, and the like. While a retardant described above can be used when an isocyanate-containing compound is used, such a retardant can be optionally utilized in any of the methods described herein.
- An aldehyde rate modifier retards the rate of the reaction by reacting with the polyamine to form a "blocked" amine in the form of an imine.
- this reaction can be performed at room temperature.
- the rate of this reaction depends on several factors, such as the concentration of the aldehyde; whether the aldehyde is aliphatic or aromatic, or linear or branched; the functionality of the side chain(s); the acidity/alkalinity of the side chain(s); the electron donating or accepting capacity of the side chain(s); steric factors; and other factors.
- the aldehyde and polyamine reaetants are in equilibrium with the imine and water is the by-product.
- the reaction that generates the imine can proceed until all of the available aldehyde has interacted with the amine.
- unreacted amine can be present due to the reversible reaction and the value of the equilibrium constant.
- the amine can react with the isocyanate, thus driving the equilibrium towards generating more amine.
- an effective level of amine can be less than a stoichiometric amount.
- the reaction between a polyamine and an isocyanate can be rapid at room temperature even in the absence of a catalyst, in some examples, a catalyst is not present, in these examples, a rate modifier (i.e., retardant) can be used to slow the reaction, allowing ample time for the ingredients to be blended and poured into a mold.
- a rate modifier i.e., retardant
- Useful rate modifiers include, for example, aldehydes such as those normally present in common essential oils and fragrance oils. Others include those that are either bland in odor or enhance the odor of the active liquid.
- Examples of useful retard ants are aromatic aldehydes such as benzaldehyde, vanillin, and salicy!aidehyde; ⁇ , ⁇ unsaturated aromatic aldehydes such as cinnamic aldehyde and methyl cinnamic aldehyde; terpenic aldehydes such as citral, cyclocitral, and citronellal; and C4-C38 aliphatic and cycloaliphatic aldehydes such as isobutyra!dehyde, lyraL 2-phenyl propionaldehyde and the like. While a retardant described above can be used when an isocyanate-eontaming compound is used, such a reiardant can be optionally utilized in any of the methods described herein.
- Another method for increasing cure times includes employing PAPA terminated with a carbonyl-substituted aromatic amine prepared according to the methods described herein. Shown below are the set times (i.e., the time from mixing to lack of flow) for four commercial fragrances immobilized at 50% concentration with matrix derived from the reaction of PAPA and DESMODUR N330Q, the PAPA being terminated either by a non- aromatic primary amine or by a carbonyl-substituted aromatic amine, i.e., the PAPA terminated by reaction with para-am inobenzoic acid.
- the curing temperature can be room temperature, i.e. 25 °C, but can be higher or lower, depending on the cure time desired.
- the curing can be carried out in a sealed mold, and at a curing temperature of about 50 °C.
- curing for a typical formulation based on PAPA terminated by a primary aliphatic amine and carried out in the presence of little or no retardant, typical setting times are from less than 1 second to about 30 minutes.
- the time can be 0.1 minute, 0.5 minute, 1 minute, 5 minutes, 10 minutes, 20 minutes, or 30 minutes, including any and all ranges and subranges there between.
- Curing at room temperature for a typical formulation based on the carbonyl-substituted aromatic amine terminated polyamine can take place in from about 10 minutes to over 2 days when carried out in the presence of retardant but can be in the range 20-600 minutes in the absence of retardant.
- the time can be 20 minutes, 50 minutes, 100 minutes, 200 minutes, 300 minutes, or 600 minutes, including any and all ranges and subranges there between,
- the articles further include a support material that can optionally be a porous support material.
- the articles described herein can include the gelled compositions.
- Such articles include, but are not limited to, medicinal devices having an active liquid that is medicinally active, pesticide devices having an active liquid that is a pesticide, laundry care devices having an active liquid for laundry care (i.e., softener, fragrance, conditioner, cleaner, anti-stain, surface treating, and the like), or air freshener having an active liquid that is a fragrance.
- the articles described herein can include the compositions in the fonn of aqueous dispersions.
- Examples of these articles include sun care products, skin care products, air fresheners, laundry fragrance sheets, laundry fabric softener sheets, laundry anti-static sheets, storage fragrance articles, pharmaceutical distribution articles, nutraceutical distribution articles, bioceutical distribution articles, moldicide distribution articles, bactericide distribution articles, pesticide distributions, decorative articles, biomedical sensors, and/or analytical devices.
- the articles described herein can be processed into any desired shape that is appealing to a potential consumer.
- Such shapes can be 3-D shapes formed in a mold or a flat shape stamp-cut from pre-formed thin sheets.
- Shapes can include those geometrical in nature, e.g., triangular, square, circular, spherical, oval, regular geometric figure, irregular geometric figure, etc.
- air care articles can have an immense variety of geometric and artistic shapes such as, but not limited to, disks, rings, cylinders, squares, rectangles, pentagons, hexagons, stars, hearts, hemispheres, spheres, cubes, flowers, animals, letters, numbers, logos, trademarks, and faces. Such shapes are limited only by methods known to make appropriate-shaped molds.
- These articles can be colored with soluble dyes or with pigments. These colorants can be dissolved or dispersed prior to final mixing of the reactive components. These colorants can be conventional, fluorescent, pearlescent, temperature-sensitive, light-sensitive, pH- sensitive, or moisture sensitive. The latter four colorants allow for the preparation of novelty products that change color as environmental conditions change or that signal the depletion of the active component in the article.
- composition prior to curing is fluid, it can be poured easily into such molds and thus take on exacting shapes such as dimples, curves, logos, etchings, and any other embossed or engraved image. This is especially advantageous if the article is designed to fit directly into a holder, to adhere to a sur face of complex shape, for example, a body part, a curved surface such as a heated potpourri dish, light bulb, or the inside of a package.
- Suspended matter can be decorative items such as icons, beads, glitter, gems, shards and the like; botanicals such as leaves, seeds, stems, needles, nuts, and the like; insoluble powdered materials such as wax, sugar, coffee grounds, bait particles, insoluble plain, colored or flavored salts, water, glycerin, silicone fluids, and aqueous solutions of dyes, active materials, acids, bases and the like with or without the aid of a surfactant to stabilize the dispersion thus formed; or with air or other gas by a whipping action or other deliberate mixing with the gas to form bubbles in the matrix- forming fluid.
- decorative items such as icons, beads, glitter, gems, shards and the like
- botanicals such as leaves, seeds, stems, needles, nuts, and the like
- insoluble powdered materials such as wax, sugar, coffee grounds, bait particles, insoluble plain, colored or flavored salts, water, glycerin, silicone fluids, and aqueous solutions of dyes
- gas can be generated inside the matrix-forming composition by chemical means, such as, for example, thermal decomposition of a nitrogen-, oxygen-, or carbon dioxide-generating substance.
- a nitrogen-, oxygen-, or carbon dioxide-generating substance examples include carboxylic acids, azobis(isobutyronitrile), hydrogen peroxide, and sodium carbonate or bicarbonate.
- a carboxylic acid that can be used in this way is polymerized fatty acid.
- the article described herein can include the fragrance oil or other active liquid and components selected from those listed above as immobilized by the cross- linked matrix.
- the article can consist of the immobilized liquid and a support, be it a container, bracket, or holder into which the mixture of the reactive components, actives and other liquids and optional components are poured before curing takes place or fitted after curing takes place.
- the article after curing can be coated, printed, or otherwise decorated, wrapped or supported by a stand, plate, bowl, dish, bracket, holder, or other supporting device.
- the container can be made of glass, ceramic, metal, paper, plastic, or any other oil-impermeable material and be in any convenient shape such as a cylinder, tube, bowl, dish, etc .
- the container can itself be shaped to fit into a holder, chamber, or receptacle designed to fit into a fragrance dispensing device that can be fitted with a heater, fan, blower, or other mechanical aid.
- the heater can be external to the cross-linked matrix-immobilized active liquid or it can be internal, that is, surrounded by or embedded in the cross-linked article.
- An example of such a device is a reactive composition poured into a container threaded with resistive heating wires that, after the matrix cures, can be electrified, thus heating the cross-linked composition from within.
- composition while still fluid can be impregnated into a porous material such as paper, cardboard, cellulose pad, cellulose pulp, felt, fabric, a porous synthetic foam, a porous ceramic, activated carbon, soil, diatomaeeous earth, kieselguhr, sand, charcoal, silica, clay, and the like or coated onto a non-porous substrate included but not limited to plastic films, metallic foils, rubber, ceramics, wood, glass, and leather.
- a porous material such as paper, cardboard, cellulose pad, cellulose pulp, felt, fabric, a porous synthetic foam, a porous ceramic, activated carbon, soil, diatomaeeous earth, kieselguhr, sand, charcoal, silica, clay, and the like
- a non-porous substrate included but not limited to plastic films, metallic foils, rubber, ceramics, wood, glass, and leather.
- the mixture of reactive components, active liquid and optional liquids, while still uncured, can be dispersed in water or other aqueous medium and the resulting emulsion optionally stabilized by means of a surfactant. Droplets of the composition described herein, thus emulsified, can then cure, resulting in a dispersion of solid gel particles. This can be considered a process for preparing encapsulated active oils in dispersed form.
- Such a material is a milky liquid and can, as such, be impregnated into a porous medium such as paper, cardboard, cellulose pad, cellulose pulp, felt, fabric, a porous synthetic foam, a porous ceramic, activated carbon, soil, diatomaeeous earth, kieselguhr, sand, charcoal, silica, clay, and the like or coated onto a non-porous substrate included but not limited to plastic films, metallic foils, rubber, ceramics, wood, glass, and leather.
- a porous medium such as paper, cardboard, cellulose pad, cellulose pulp, felt, fabric, a porous synthetic foam, a porous ceramic, activated carbon, soil, diatomaeeous earth, kieselguhr, sand, charcoal, silica, clay, and the like
- a non-porous substrate included but not limited to plastic films, metallic foils, rubber, ceramics, wood, glass, and leather.
- a container can be nearly filled with a volatile active liquid and can then be filled up with and sealed by the composition described herein, thus trapping the volatile material behind a barrier or membrane of cross-linked matrix.
- a barrier or membrane of cross-linked matrix Such an arrangement allows the reservoir of volatile liquid to be released very slowly and continuously as it diffuses through the barrier of liquid-impregnated matrix.
- the article components can be insoluble in water without losing any of the desired final properties (e.g., fragrance release, stability) so that the water can optionally serve some useful purpose if incorporated in the cross-linked composition such as causing shrinkage to indicate end-of-use-Hfe or introduction of a water-soluble active ingredient such as a dye or a salt.
- desired final properties e.g., fragrance release, stability
- the articles can be prepared by (1) blending the polyamine, the active liquid and any desired optional components including diluents, plasticizers, fillers, stabilizers, and colorants: (2) blending this mixture with the polyepoxy or polyisocyanate component optionally diluted with further amounts of plasticizers, fillers, stabilizers, and colorants; (3) pouring out the final blend as a sheet or slab or into a support, form, container, or mold; (4) optionally covering or sealing the poured blend to protect it from contaminants and prevent volatile components from evaporating; (5) optionally storing it until the blend cures; and (6) optionally removing the cured immobilized liquid article from the sheet, slab, form, container, or mold and cutting it to another shape or using it as made in the container.
- Active air fresheners encompass relatively complex devices having moving parts such as heaters and fans to dispense concentrated or diluted aroma compounds or spray cans charged with aroma chemical, carrier liquid, and propellant. Active air fresheners require the occupant to dispense the materia! into the area to be treated. Passive air fresheners are available in many forms, but are in essence "fixed” liquid chemicals: a multi-component article including fragrance oil immobilized in and/or a solid support.
- the support material can be simple, e.g., a piece of cardboard, blotter paper, cotton, or other fibrous materials.
- the support material can be complex, e.g., an aqueous dispersion (gelatin) or a non-aqueous gel (gelled, e.g., by polyamide resin).
- the air fresheners can be transparent, but, in some embodiments, can be opaque.
- the article is a visually attractive solid air freshener, in particular a room, closet, drawer, bag, area, container, or car interior freshener, that is both transparent or nearly transparent (e.g. "frosted") and robust.
- the active liquid is an aromatic composition (i.e. fragrance oil, scent, or perfume).
- the term "robust" means that the article can be packaged inexpensively and handled without being deformed.
- the composition containing the aromatic material can be supported (i.e., in a container or holder) or free-standing. In particular, no special care is needed when the air freshener is taken out of its package or wrapper.
- the air freshener can resist changes in temperature, humidity, and exposure to light over the lifetime of its use or, with reasonable protection in a suitable package, over the lifetime of its storage and handling.
- the air care compositio can also be free of syneresis (also known as "sweating").
- the matrix material of the product is to be effectively non-toxic and not cause skin irritation if handled out of its storage wrapper.
- the air care composition Sends itself readily to, but does not require the use of, porous powders, fabrics or fibers as a support for the fragrance oil
- Air freshener components including a small amount of green dye, which were weighed into a glass vial and stirred together at ambient temperature by hand with a wooden stir stick. A portion of the mixture (8.0g) was then poured into a flat, rectangular, 2.50 inch x 3.25 inch uncoated polystyrene mold;
- Epoxy Resin EPALLOY® 5001, lO.OOg; 55.1%
- a poiyamide polyamine was prepared by charging adipic acid (20.0g, 274 meq acid), JEFF AMINE® T-403 polyamine (20g, 132 meq amine) and Huntsman XTJ-500 (80g, 254 meq. amine) to a 250 mL glass flask equipped with a stirrer and heating this charge to 210- 220°C under a stream of dry nitrogen. After holding this mixture under these conditions for 5 hours, the reaction mixture was discharged to a container. The product was a clear, viscous, nearly water-white liquid having an acid number of 1.4, an amine number of 42.2, and a Brookfield viscosity at 150°C of 340 cP.
- air freshener components totaling 100 parts by weight were blended at room temperature: cyclohexane dimethanol diglycidyl ether (25,3 parts), EPON® 828 (17,2 parts), Arizona proprietary polyamido-amine hardener #X54-327-004 (34,5 parts), Continental Aromatics "Ocean” fragrance oil (23.0 parts), and a trace of green dye. This blend was held for about 45 minutes at about 67°C, at which time it was allowed to cool to room temperature.
- Example 2 polypropylene glycol) diglycidyl ether (13.0 parts), EPON® 828 (22.0 parts), Arizona UNI-REZ® 2801 amido-amine (14.0 parts), "Vanilla” fragrance oil from Aromatic Flavors and Fragrances, dipropyleneglycol benzoate (19.5 parts) and commercial ground coffee (29.5 parts).
- the resulting article after curing was firm, slightly flexible, non-tacky. The coffee grounds were uniformly distributed and gave the article a rich brown, opaque appearance, smooth at the bottom where the mold was smooth and rough on top where the grounds were allowed to settle freely.
- ⁇ CHDA is 1 , 4 cyclohexane dicarboxylic acid from Eastman Chemical
- Empol is EMPOL® 1008 dimer acid supplied by Cognis Corporation;
- T-403 is JAFFAMI E® T-403 poly(alkyleneoxy) diamine supplied by Huntsman Corporation;
- ⁇ D-2000 is JEFFAMINE®T-2000 poiy(a!kyleneoxy) diamine also from Huntsman; 8 V-551 is VERSAMINE® 551 dimer diamine supplied by Cognis Corporation;
- N-3300 is DESMODUR® N-3300 or N-3300A, Bayer Corporation, Industrial
- N-3800 is DESMODUR® N-3800, also from Bayer; * Z-4470 is DESMODUR® Z4470, also from Bayer.
- a polyamide polyamine was prepared by charging EMPOL® 1008 polymerized fatty acid (63,0g, 219 meq acid), JEFFAMiNE®T-403 polyamine (18g, 118 meq amine) and JEFFAMINE®D-400 (45g, 205 meq. amine) to a 250 mL glass flask equipped with a stirrer and heating this charge to 210-220°C under a stream of dry nitrogen. After holding this mixture under these conditions for 5 hours, the reaction mixture was discharged to a container. The product was a clear, viscous, nearly water-white liquid having an acid number of 0.3, an amine number of 41.8, a weight average molecular weight of 2,270, and a
- a solution was prepared by warming lO.Og of this polyamide polyamine with 5,0g FINSOLV® TN benzoate ester and lO.Og fragrance oil ("Linen Fresh", Wessel Fragrances), cooled to room temperature and blended thoroughly with a mixture of DESMODUR® Z4470 and 5,lg additional fragrance oil. To the composition was then added a small amount of red dye and red glitter. A few minutes later about 25g of this final formulation was poured into a flat, circular rose-shaped silicone rubber mold and the remainder retained in ajar. A total of 33 minutes after the component were blended, the retained material was set to an immobile gel. After standing at room temperature for 16 hours, the immobilized fragrance oil article was removed from the mold.
- Polyamide po!yamines were prepared according to the procedure of Example 9 by charging acids and amines of the types listed in the TABLE A (below) in the weight percentages indicated to a reactor and heating the charge to 200-220°C under a stream of dry nitrogen for about 4-5 hours and discharging the product. Products properties were then measured and are also recorded in TABLE 1.
- Immobilized fragrance oils were prepared by warming a mixture of 2.0 grams PAPA of the example and 2.0 grams fragrance oil to about 55°C and then blending the warm mixture by hand with a stir stick.
- Test fragrances were: “Ocean” (Continental Aromaiics), "Linen Fresh” (Wessel Fragrances), and “Cherry” (Aromatic Flavors and Fragrances). After blending, one equivalent of isocyanate hardener dissolved in an equal weight of oil was added with manual stirring, a stopwatch was started, and the mixture monitored for its consistency. When the mixture no longer could flow under its own weight, the time (in minutes) was noted as the "gel time”.
- Polyamide polyamines were prepared according to the procedure of Example 9 by charging acids and amines of the types listed in the TABLE C in the weight percentages indicated to a reactor and heating the charge to 200 ⁇ 220°C ursder a stream of dry nitrogen for about 5 hours and discharging the product. Products properties were then measured and are also recorded in TABLE 3.
- immobilized fragrance oils were prepared by warming a mixture of 2.0 grams polyamide po!yamine of the example and 2,0 grams fragrance oil to about 55°C and then blending the warm mixture by hand with a stir stick.
- Test fragrances were: Oceanside Mist, Tropical (Atlas Products), Spring Meadow, Country Wildflower, Ocean (Continental Aromatics), Linen Fresh (Wessel Fragrances), Yankee Home (Belle Aire), Mulberry and Cherry (Aromatic Flavors and Fragrances).
- isocyanate hardener dissolved in an equal weight of oil was added with manual stirring, a stopwatch was started, and the mixture monitored for its consistency. When the mixture no longer could flow under its own weight, the time (in minutes) was noted as the "gel time".
- TABLE 4 shows that all of these polyamide polyamines were effective in immobilizing the target oils when cross-linked with poiyisocyanates. Gel times were short but not so short as to preclude the preparation of useful articles and followed the consistent pattern:
- a number of batches of a PAPA were prepared by the method of Example 9 using a charge (weight percentages in brackets) of either EMPOL® 1008 or UMDYME® 12 (a low trimer content, hydrogenated dimer acid obtained from Arizona Chemical) [29.5%], T-403 [3.7%], D-400 [22.6%], and D-2000 [44.2%].
- This polymer, used in Examples #22-35 typically had an amine number of 30-35 (equivalent wt. of 1 ,800-1,600), a weight-average molecular weight of 10,700-12.100, a number-average molecular weight of 4,300-4,900, and a viscosity at 150°C of 40-70 cP.
- This example illustrates the preparation of an air freshener in a simple geometric shape.
- To a glass mixing jar was charged 13. Ig of the Example 21 PAPA and 15g of "Cotton Fresh" fragrance oil (Symrlse Corp.) and the mixture was stirred gently for 15 minutes at ambient temperature. Blue dye (2 drops) was added to the mixture, turning the solution light blue.
- To this homogeneous mixture was then added 1.5g ofDESMODUR® N3300A. This mixture was then stirred until homogeneous, allowed to stand a few minutes to allow any air bubbles to dissipate, and 13g total was poured into a rectangular-shaped flexible silicone mold of uniform length of 1.87 inches, height of 0.3 inches, and width of 1.0 inches.
- the set time was recorded at 28 minutes. The mixture was covered with polyethylene film and allowed to cure undisturbed for 24 hours. After this time the mold was stripped away from the cross-!inked air freshener object that was now firm, flexible, transparent and non-tacky to the touch.
- This example illustrates the preparation of an air freshener in a complex shape.
- To a glass mixing jar was charged 13 , 1 g of the Example 21 po!yamine and 15g of "Snuggle Type” fragrance oil (Alpha Aromatics) and the mixture was stirred gently for 15 minutes at ambient temperature. Red dye (3 drops) was added to the mixture, turning the solution light pink/red. To this homogeneous mixture was then added 1 ,5g of DESMODUR® N3300A.
- This example illustrates the preparation of an air freshener in a complex shape.
- a glass mixing jar was charged 19g of the Example 21 polyamme and 20g of "Tropical Splash” fragrance oil (obtained from Symrise Corp.) and the mixture was stirred gently for 15 minutes at ambient temperature. Blue dye (3 drops) was added to the mixture, turning the solution light green.
- To this homogeneous mixture was then added 2.0g of DESMODUR® N3300A, This mixture was then stirred briefly (until homogeneous), allowed to stand a few minutes to allow any air bubbles to dissipate, 20g total was poured into a scallop-shaped flexible silicone mold of uniform top-width of 2.375 inches, height of 0.125 inches, and bottom-width of 2.25 inches. The set time was recorded at 24 minutes. The mixture was covered and allowed to cure undisturbed for 24 hours. After this time the mold was stripped away from the cross- linked air freshener article that was now firm, flexible, transparent, and non-tacky to
- This example illustrates the preparation of an air freshener containing suspended insoluble particles.
- Example 21 polyamine and 20g of "Clean Citrus” fragrance oil (from Symrise Corp.) and the mixture was stirred gently for 15 minutes at ambient temperature. Yellow aluminum flake “glitter” (0.04g) was added to the mixture.
- To this homogeneous mixture was then added 2.0g of DESMODUR® N3300A, This mixture was then stirred briefly (until homogeneous), allowed to stand a few minutes to allow any air bubbles to dissipate, 18.0g total was poured into a disk-shaped flexible silicone mold of uniform circumference of 9.75 inches, height of 0,75 inches, and width of 3.0 inches.
- the set time was recorded at 30 minutes. The mixture was covered and allowed to cure undisturbed for 24 hours. After this time the mold was stripped away from the cross-linked air freshener article that was now firm, flexible, transparent, and non-tacky to the touch and displayed a uniform distribution of glitter.
- This example illustrates the preparation of an immobilized phase-transfer liquid.
- To a glass mixing jar was charged 10.4g of the Example 21 polyamine and 18g of 1-decanol (freezing point, 5-7oC) as the active oil, 0.6g benzaldehyde as odorant and cross-linking reaction retardant and the mixture was stirred gently for 15 minutes at ambient temperature. To this homogeneous mixture was then added 1.5g of DESMODUR® N3300A.
- This example illustrates the preparation of a small air freshener for use in a purse or other small enclosed space
- a small air freshener for use in a purse or other small enclosed space
- This mixture was then stirred briefly (until homogeneous), allowed to stand a few minutes to allow any air bubbles to dissipate, 5.0g total was poured into a lozenge-shape polyethylene bulb mold of uniform middle-circumference of 1.5 inches, height of 1.625 inches, and top and bottom-width of 0.5 inches. The set time was 7 minutes. The mixture was sealed and allowed to cure undisturbed for 24 hours. After this time the mold was stripped away from the cross-linked air freshener object that was now firm, transparent, and non-tacky to the touch.
- Example 21 polyamine and 40g of "Cherry Berry" fragrance oil (Belle-Aire) and the mixture was stirred gently for 15 minutes at ambient temperature. Red dye (3 drops) was added to the mixture, turning the solution red. To this homogeneous mixture was then added 4.0g of DESMODUR® N330GA. This mixture was then stirred until homogeneous, allowed to stand a few minutes to allow any air bubbles to dissipate, 60.0g total was poured into a rose flower-shaped flexible silicone mold of uniform top and bottom- width of 3.75 inches, height of 0,75 inches, and circumference of 12.25 inches. The set time was recorded at 155 minutes. The mixture was covered and allowed to cure undisturbed for 24 hours. After this time the mold was stripped away from the cross-linked air freshener object that was now firm, flexible, transparent, and non-tacky to the touch.
- Red dye 3 drops
- DESMODUR® N330GA DESMODUR® N330GA
- This mixture was then stirred briefly (until homogeneous), allowed to stand a few minutes to allow any air bubbles to dissipate, 40g total was poured into a baking cup paper mold of uniform top and bottom-width of 2.0 inches, height of 1.25 inches, and circumference of 7.5 inches. The set time was 8 minutes. The mixture was allowed to cure undisturbed for an additional 24 hours. During this time the object became filled with trapped bubbles (foam) and doubled in size, forming a rounded crown. This foam air freshener was now firm and non-tacky to the touch. When compressed (squeezed), it returned to its rounded shape.
- a number of batches of a polyamide polyamine terminated with a carbonyl-substituted aromatic amine were prepared by charging (weight percentages in brackets) of PRIPOL® 1009 hydrogenated dimer acid [24.0], para-aminobenzoic acid [5.0], JEFF AMINE® D-2000 [54.0], JEFF AMINE® D-400 [11.5], and JEFF AMINE® T-403[5.5] to a 3L glass round- bottomed reactor equipped with an overhead mechanical stirrer and heating this charge to 215°C under a stream of dry nitrogen. After holding this mixture under these conditions for about 25 hours, the reaction mixture was discharged to a container. The product was a clear, viscous, slightly yellow liquid.
- This polymer had a titrated amine number in the range 13-15 (non-potentiometric method, or 30-35 by potentiometric titration, amine reactive equivalent wt. of 1,800-1,600), a weight-average molecular weight of 13,000-14,000, a number-average molecular weight of 4,500-5,500, and a viscosity at 130°C of 250 cP.
- This material was used in a series of tests of immobilizing, at the 30 weight% use level, liquid test media (70% by weight), free of active, catalyst, or retardant.
- the results demonstrate that set times can vary up to about 1 day for such a modified PAPA even in the absence of retardant aldehyde.
- the data also demonstrate the accelerating effect of the use of an alcoholic diluent, such as a polypropylene glycol or its alkyl ether, on the cure rate.
- Example 36 illustrates the preparation of another type of polyamide polyamine terminated with a earbonyl-substituted aromatic amine.
- the procedure of Example 36 was followed using a charge (weight percentages in brackets) of T-5000 [92.9] and para- aminobenzoic acd [7.1].
- This example illustrates the preparation of an article containing liquid fragrance trapped behind a membrane of matrix.
- Example 37 polyamine To a glass mixing jar was charged 5.0g of the Example 37 polyamine and 5,0g of FINSOLV® TN and the mixture was stirred gently for 15 minutes at ambient temperature.
- To this homogeneous mixture was then added 0.6g of DESMODUR® N3300A. This mixture was then stirred briefly (until homogeneous), allowed to stand a few mi utes to allow air bubbles to dissipate, and then a 1.0g portion was poured gently, without stirring, into a loz. glass vial containing lOg of "Lily of the Valley" green fragrance oil (Wellington Fragrances).
- This example illustrates the preparation of a article containing an aromatic filler.
- a glass mixing jar was charged 15g of the Example 37 polyamine, 6g of castor oil, and 9g of commercial ground coffee and the mixture was stirred gently for 30 minutes at ambient temperature.
- To this viscous paste was then added 2.0g of DEMODUR® N3300A.
- This mixture was then stirred briefly, allowed to stand a few minutes to allow any air bubbles to dissipate, poured (18.0g used) into a disk-shaped flexible mold of uniform circumference of 8,25 inches, height of 0.25 inches, and top and bottom-width of 2.5 inches.
- the set time was recorded at 165 minutes.
- the mixture was covered and allowed to cure undisturbed for 24 hours. After this time the mold was stripped away from the object that was now fragrant (coffee odor), firm, flexible, and non-tacky to the touch.
- This example illustrates the preparation of an article containing water.
- 20g of the Example 37 polyamine 20g of "Snuggle Type” fragrance oil (from Alpha Aromatics), and 8g of de-ionized water, and the mixture was stirred gently for 15 minutes at ambient temperature, resulting in a milky suspension of water in the matrix- fragrance solution. Blue dye (2 drops) was added to the mixture. To this light blue, milky mixture was then added 2,5g of DESMODU ® N3300A. This mixture was then stirred briefly and allowed to stand a few minutes to allow any air bubbles to dissipate.
- Solution A to a glass mixing jar was charged 8g of the Example 37 polyamine and 8g of FINSOLV® TN and the mixture was stirred gently for 15 minutes at ambient temperature. To this homogeneous mixture was then added 0.8g of DESMODU ® N3300A. This mixture was then stirred briefly (until homogeneous), allowed to stand a few minutes to allow any air bubbles to dissipate.
- Solution B to another glass mixing jar was charged 32g deionized water and 0.8g of surfactant (T- DET A-136). This mixture was stirred (10 minutes). Solution A was then poured into
- DEET controlled-release diethyl toluamide
- This example illustrates the use of a styrene-maleic anhydride copolymer as the reactive partner with a polyamide polyamine for preparation of a lightly-scented disk-shaped air freshener.
- a glass mixing vial was charged 6.0g of a 25 wt% solution FINSOLV® TN solution of the Example 21 polyamide polyamine, 7.5g of a 20 wt% solution of DYLARK® 232 poIy(styrene-co-ma!eic anhydride, NOVA Chemicals), and ca. 2g of "Ocean” fragrance oil (provided by Wellington, Inc.).
- the mixture was stirred gently for a few minutes at ambient temperature and blue dye (4 drops) added.
- the mixture was initially slightly turbid but cleared after a few more minutes and remained clear and apparently homogeneous.
- the mixture was then poured (about 1 Ig was used) into a disk-shaped polyethylene mold and allowed to stand undisturbed.
- the mixture set to a sticky, elastic mass inside about 2 hours and after 24 hours could be stripped from the mold. After this time the mold was stripped away from the cross-linked air freshener object that was now firm, transparent, and flexible with a light tack to the touch.
- This example illustrates the use of a cationic surfactant to prepare an immobilized fragrance emulsion useful as a fabric softener.
- a blend of PAPA of Example 13 (4,Gg), "Cinnamon Chai” fragrance oil (3.0g), and VARIQUAT® B1216 alky! dimethyl benzyl ammonium chloride (80% active, Degussa Corporation, l.Og) was first prepared by warming and stirring the ingredients. To the blend was added water (9,0g) and then, with stirring, DESMODUR® N3300A (0.65g). The mixture soon became viscous and uniformly cloudy, it was storage stable and was dilutable with water, indicating it was an oil-in- water dispersion.
- Light scattering particle size measurement on the material determined the particle size distribution to be bi-modal, with about 50% of the weight of particles having a size grouping around 0.4 microns and the other 50% grouping around 3.0 microns.
- This example illustrates the preparation of an immobilized cationic surfactant useful as a fabric softener.
- a blend of PAPA of Example 21 (3.0g), DOWANOL® DPM (l .Og) and VARIQUAT® B1216 alky! dimethyl benzyl ammonium chloride (80% active, Degussa Corporation, 6.0g) was first prepared by warming and stirring the ingredients and then cooling them to room temperature.
- a second blend was prepared of DOWANOL® DPM (4.2g) and DESMODUR® N3300A (G.Sg). The two clear mixtures were then mixed together and immediately poured into a mold. The blended components set almost immediately and were firm enough to pick up out of the mold in less than 30 minutes.
- the final article contained 32% by weight active quaternary compound.
- a secondary amine terminated polyamide polyamme (SATPP) was prepared by charging PRIPOL 1006 polymerized fatty acid (48.8 g, 219 meq acid) (Croda, Inc.; Edison, NJ), JEFFAMINE D-2000 (54.9 g, 1000 meq amine) (Huntsman Corporation; The
- a secondary amine terminated polyamide polyamine was prepared by charging PRiPOL 1006 polymerized fatty acid (48.8 g. 219 meq acid) (Croda, Inc.; Edison, NJ), JEFF AMINE D-2000 (54.9 g, 1000 meq amine) (Huntsman Corporation; The
- AEP aminoethylpiperazine
- a secondary am ine terminated polyamide polyamine (SATPP) (1.25 g) and fragrance oils (3.5g) were manually mixed in a glass vessel to obtain a homogenous solution.
- the fragrance oils were obtained from Belcan Inc.
- Examples 79-90 illustrate the gel setting time differences between secondary amine terminated polyamide pol amine (SATPP) and primary amine terminated polyamide polyaniine (PATPP), as shown in Table 7.
- SATPP secondary amine terminated polyamide pol amine
- PATPP primary amine terminated polyamide polyaniine
- a secondary amine terminated polyamide polyamine (SATPP), SYLVACLEAR ⁇ 800, a polyamide polyamine terminated with a carbonyl- substituted aromatic amine commercially available from Arizona Chemical Company (Jacksonville, FL), and fragrance oils were manually mixed in a glass vessel to obtain a homogenous solution. The mixtwe was allowed to stand for 10 minutes. One equivalent of isocyanate hardener DESMODUR N 3300 (Bayer Corporation; Pittsburgh, PA) was then added with manual stirring. The gel time was measured by observing the amount of time lapsed to provide a mixture no longer able to flow under its own weight. Table 8 illustrates the gel setting time for the formulations with SATPP can be adjusted by blending with po!yamide polyamines terminated with carbonyl-substituted aromatic amine.
- immobilized fragrance oil dispersions were prepared according to the following generic process. Details on the individual components for each of the dispersions are described in Tables 9 and 10. To form Part A, the indicated amounts of water. 1% METHOCEL 311 cellulose water solution (Dow Chemical; Midland, Mi),
- FINSOLV-TN an alkyl benzoate commercially available from Innospec Active Chemicals (Edison, NJ), and a surfactant ARQUAD 18-50 (Akzo Nobel Surface Chemistry LLC;
- Part B was prepared by pre-mixing the indicated amounts of fragrance Natalie (Givaudan; Vernier, Switzerland) and polyamide polyamine SYLVACLEAR IM 700 (Arizona Chemical Company; Jacksonville, FL) to form a homogenous solution in 2 minutes.
- Part B was dispersed dropwise into Part A aqueous phase immediately at the indicated rpm.
- Part B addition was completed in 3 minutes.
- the mixture was agitated at the indicated rpm for 60 minutes and discharged to a container.
- the dispersions were milky solutions.
- the indicated amount of additional surfactant ARQUAD 18-50 was added and mixed for 5 minutes. Table 9
- the fragrance oil dispersions were prepared according to the following generic process, with details regarding the individual components shown in Table 1 1.
- the indicated amounts of SYLVACLEAR IM700 (Arizona Chemical Company; Jacksonville, FL) and the fragrance Berry (Belmay Fragrances Ltd.; Yonkers, NY) were prefixed to form a homogenous solution.
- the solution was added dropwise to an aqueous solution having the indicated amounts of water, 1% METHOCEL 311 (Dow Chemical; Midland, Ml) water solution, and surfactant ARQUAD 18-50 (Akzo Nobel Surface
- SYLVACLEAR 1M800 (4,50 g) (Arizona Chemical Company; Jacksonville, IL) and 10.50 g of the fragrance Natalie (Givaudan; Vernier, Switzerland) were mixed to obtain a homogenous solution. Then 0.58 g DESMODUR N3300 (Bayer Corporation; Pittsburgh, PA) was added to the solution and mixed well to form a homogenous solution in 5 minutes. The solution was added dropwise to an aqueous solution having 12.50 g water, 5.0 g 1% METHOCEL 311 water solution (Dow Chemical; Midland, MI), and 2.68 g surfactant ARQUAD 16-50 (Akzo Nobel Surface Chemistry LLC; Chicago, IL) with mixing at 1200 rpm in a 100 mL plastic cup. The addition was completed within 3 minutes. The mixing was continued for 130 minutes and the resulting mixture was discharged to a container. The dispersions were milky solutions. The mean particle size was 10.5 microns.
- FINSOLV-TN a solvent commercially available from Innospec Active Chemicals (Edison, NJ)
- DEET N,N-diethyl-jMeta-toluaniide
- the solution was added dropwise to an aqueous solution having 12,50 g water, 5.0 g 1% METHOCEL 311 water solution (Dow Chemical; Midland, Ml), and 2,68 g ARQUAD 16-50 (Akzo Nobel Surface Chemistry LLC; Chicago, IL) with mixing at 1200 rpm in a 100 mL plastic cup. The addition was completed within 3 minutes. The mixing was continued for 120 minutes and the resulting mixture was discharged to a container. The dispersions were milky solutions.
- SYLVACLEAR LM800 (4.50 g) (Arizona Chemical Company; Jacksonville, FL), 5.5 g of FINSOLV-TN (a solvent commercially available from Innospec Active Chemicals (Edison, NJ)) S and 5.0 g sumithrin were mixed to obtain a homogenous solution. Then 0.58 g DESMODUR N3300 (Bayer Corporation; Pittsburgh, PA) was added to the solution and mixed well to form a homogenous solution.
- the solution was added dropwise to an aqueous solution having 12.50 g water, 5,0 g 1% METHOCEL 311 water solution (Dow Chemical; Midland, Ml), and 2.68 g surfactant ARQUAD 16-50 (Akzo Nobel Surface Chemistry LLC; Chicago, IL) with mixing at 1200 rpm in a 100 mL plastic cup. The addition was completed within 3 minutes. The mixing was continued for 120 minutes and discharged to a container. The dispersions were milky solutions.
- compositions, methods, and apparatuses of the appended claims are not limited in scope by the specific compositions, methods, and articles described herein, which are intended as illustrations of a few aspects of the claims and any compositions, methods, and articles that are functionally equivalent are intended to fall within the scope of the claims.
- Various modifications of the compositions, methods, and articles in addition to those shown and described herein are intended to fall within the scope of the appended claims.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Epidemiology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Dispersion Chemistry (AREA)
- Polyurethanes Or Polyureas (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Polyamides (AREA)
- Epoxy Resins (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2817152A CA2817152A1 (en) | 2011-11-18 | 2011-11-18 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same |
| MX2013005683A MX2013005683A (en) | 2010-11-22 | 2011-11-18 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same. |
| EP11791152.9A EP2643155A1 (en) | 2010-11-22 | 2011-11-18 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same |
| KR1020137015461A KR20130117814A (en) | 2010-11-22 | 2011-11-18 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same |
| JP2013540043A JP2013543049A (en) | 2010-11-22 | 2011-11-18 | Compositions and articles containing an active liquid in a polymer matrix and methods for making and using the same |
| BR112013012481A BR112013012481A2 (en) | 2010-11-22 | 2011-11-18 | compositions and articles containing an active liquid in a polymeric matrix and methods of preparing and using them |
| CN2011800561551A CN103282193A (en) | 2010-11-22 | 2011-11-18 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/951,421 | 2010-11-22 | ||
| US12/951,421 US20110117156A1 (en) | 2004-05-27 | 2010-11-22 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012071261A1 true WO2012071261A1 (en) | 2012-05-31 |
Family
ID=45094275
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2011/061349 WO2012071261A1 (en) | 2010-11-22 | 2011-11-18 | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20110117156A1 (en) |
| EP (1) | EP2643155A1 (en) |
| JP (1) | JP2013543049A (en) |
| KR (1) | KR20130117814A (en) |
| CN (1) | CN103282193A (en) |
| BR (1) | BR112013012481A2 (en) |
| MX (1) | MX2013005683A (en) |
| TW (1) | TW201237099A (en) |
| WO (1) | WO2012071261A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3192566A1 (en) | 2016-01-15 | 2017-07-19 | International Flavors & Fragrances Inc. | Polyalkoxy-polyimine adducts for use in delayed release of fragrance ingredients |
| US10526447B2 (en) | 2015-04-15 | 2020-01-07 | Houghton Technical Corp. | Materials that provide bioresistance and/or defoaming and slower cooling properties for aqueous quenchants |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8664292B2 (en) * | 2004-05-27 | 2014-03-04 | Croda International Plc | Compositions and articles containing a cross-linked polymer matrix and an immobilized active liquid, as well as methods of making and using the same |
| CN201712857U (en) | 2010-05-10 | 2011-01-19 | S.C.约翰逊父子公司 | Diffusing device used for volatile material and casing and diffusing piece thereof |
| BR112014001050B1 (en) * | 2011-07-21 | 2017-12-05 | Croda International Plc | POLYESTER POLYESTER BLOCK POLYMER, METHOD FOR PREPARATION OF THE SAME, COMPOSITION, CONTROLLED RELEASE AND PERSONAL CARE PRODUCTS, AND METHOD FOR PREPARING A GEL COMPOSITION |
| ITPR20110085A1 (en) * | 2011-10-03 | 2013-04-04 | San Pio S R L | DEODORANT FOR VEHICLES. |
| US9498554B2 (en) | 2012-07-24 | 2016-11-22 | S.C. Johnson & Son, Inc. | Dispensing device |
| US10694747B2 (en) | 2012-11-21 | 2020-06-30 | S. C. Johnson & Son, Inc. | Dispenser comprising only one single hinge |
| US9205163B2 (en) | 2012-11-27 | 2015-12-08 | S.C. Johnson & Son, Inc. | Volatile material dispenser |
| US9278151B2 (en) | 2012-11-27 | 2016-03-08 | S.C. Johnson & Son, Inc. | Volatile material dispenser |
| US20150098922A1 (en) * | 2013-10-04 | 2015-04-09 | The Procter & Gamble Company | Compositions comprising polyamine polymer compatible perfume materials |
| WO2015106070A1 (en) * | 2014-01-10 | 2015-07-16 | Agilex Flavors & Fragrances, Inc. | Air freshener composition and methods thereof |
| WO2016112262A1 (en) * | 2015-01-09 | 2016-07-14 | Agilex Flavors & Fragrances, Inc. | Polymeric gel composition for fragrancing and fabric treatment in a clothes dryer |
| FR3037257B1 (en) * | 2015-06-15 | 2020-09-25 | Eurotab | HUMIDITY ABSORBING TABLET RELEASING A VOLATILE ACTIVE SUBSTANCE |
| US10258707B2 (en) | 2016-02-10 | 2019-04-16 | Henkel IP & Holding GmbH | Diminishing gel compositions and associated methods |
| JP6853524B2 (en) * | 2016-07-29 | 2021-03-31 | 山本香料株式会社 | Odor modulation method |
| US10645915B2 (en) * | 2016-08-11 | 2020-05-12 | Kory McGavin | Insect trap |
| WO2020179413A1 (en) * | 2019-03-06 | 2020-09-10 | 三菱瓦斯化学株式会社 | Epoxy resin composition and cured product thereof, and fiber-reinforced composite |
| JP7221889B2 (en) * | 2020-01-24 | 2023-02-14 | 信越化学工業株式会社 | Sustained-release pheromone preparation and method for simultaneous control of mulberry mealybug and matsumoto mealybug using the same |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03258899A (en) | 1990-03-07 | 1991-11-19 | Koukandou:Kk | Fragrance emitting sheet having living thing activity |
| FR2695033A1 (en) * | 1992-08-25 | 1994-03-04 | Oreal | Prepn. of water-soluble melanic polymer by oxidn. of indole or indoline cpd. - in presence of water-soluble polymer, used in compsn. for colouring hair or skin or make=up compsn. giving natural brown colour. |
| JPH07145299A (en) | 1993-11-25 | 1995-06-06 | Yokohama Rubber Co Ltd:The | Perfume material composition |
| US5780527A (en) | 1994-08-19 | 1998-07-14 | Firmenich Sa | Perfuming device for perfuming and sanitizing ambient air |
| US6111655A (en) | 1993-11-11 | 2000-08-29 | Dainippon Screen Mfg Co., Ltd. | Method and system for managing component data of the design process and prepress process of a printing operation |
| US6399713B1 (en) | 2001-01-24 | 2002-06-04 | Arizona Chemical Company | Hydrocarbon-terminated polyether-polyamide block copolymers and uses thereof |
| US6503577B2 (en) | 2000-03-20 | 2003-01-07 | Sulzer Metco Ag | Method of thermally coating a cylinder barrel of a cylinder block of a combustion engine |
| WO2003020412A1 (en) * | 2001-08-28 | 2003-03-13 | International Fragrance & Technology, Inc. | Clear, polymeric gel composition and method for producing the same |
| US6870011B2 (en) | 2001-01-24 | 2005-03-22 | Arizona Chemical Company | Hydrocarbon-terminated polyether-polyamide block copolymers and uses thereof |
| US6956099B2 (en) | 2003-03-20 | 2005-10-18 | Arizona Chemical Company | Polyamide-polyether block copolymer |
Family Cites Families (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4081384A (en) * | 1975-07-21 | 1978-03-28 | The Proctor & Gamble Company | Solvent-free capsules and fabric conditioning compositions containing same |
| US4328322A (en) * | 1979-12-03 | 1982-05-04 | Polaroid Corporation | Synthetic polymers by polyisocyanate polyaddition process |
| JPS6056724B2 (en) * | 1980-10-22 | 1985-12-11 | 株式会社クラレ | Manufacturing method of water absorbent resin |
| US4590111A (en) * | 1982-10-21 | 1986-05-20 | Hakugen, Ltd. | Fragrance releasing sheet article and method of making same |
| US4594380A (en) * | 1985-05-01 | 1986-06-10 | At&T Bell Laboratories | Elastomeric controlled release formulation and article comprising same |
| IT1187748B (en) * | 1985-10-02 | 1987-12-23 | Resem Spa | SINGLE-COMPONENT POLYURETHANE ADHESIVE |
| US4847416A (en) * | 1986-10-27 | 1989-07-11 | The Dow Chemical Company | Capping of polyols with aromatic amines |
| FR2610537A1 (en) * | 1987-02-11 | 1988-08-12 | Rhone Poulenc Chimie | IMPROVED MICROENCAPSULATION METHOD BY INTERFACIAL POLYADDITION |
| US5008115A (en) * | 1988-04-22 | 1991-04-16 | Dow Corning Corporation | Matrix for release of active ingredients |
| US5051305A (en) * | 1988-12-30 | 1991-09-24 | Arcade, Inc. | Stabilized perfume-containing microcapsules and method of preparing the same |
| CA2009047C (en) * | 1989-02-27 | 1999-06-08 | Daniel Wayne Michael | Microcapsules containing hydrophobic liquid core |
| US5104930A (en) * | 1990-02-27 | 1992-04-14 | Raychem Corporation | Polyurea gel compositions and articles therefrom |
| US5162481A (en) * | 1990-03-30 | 1992-11-10 | Minnesota Mining And Manufacturing Company | Polyurethaneurea composition |
| US5900245A (en) * | 1996-03-22 | 1999-05-04 | Focal, Inc. | Compliant tissue sealants |
| GB9518749D0 (en) * | 1995-09-13 | 1995-11-15 | Smith & Nephew | Curable compositions |
| DK0876205T3 (en) * | 1995-12-29 | 2002-08-12 | Ciba Spec Chem Water Treat Ltd | Polymer sheath particles and their preparation |
| US5645632A (en) * | 1996-02-14 | 1997-07-08 | Union Camp Corporation | Diesters of polymerized fatty acids useful in formulating hot-melt inks |
| US5783657A (en) * | 1996-10-18 | 1998-07-21 | Union Camp Corporation | Ester-terminated polyamides of polymerized fatty acids useful in formulating transparent gels in low polarity liquids |
| US5777023A (en) * | 1996-10-01 | 1998-07-07 | Union Camp Corporation | Diamidediurethanes and hot-melt printing therewith |
| CN1238678A (en) * | 1996-10-18 | 1999-12-15 | 友联坎普公司 | Ester-terminated polyamide gels |
| US6020392A (en) * | 1998-09-04 | 2000-02-01 | Air Products And Chemicals, Inc. | Polyurea elastomeric microcellular foam |
| US6592857B2 (en) * | 1999-01-04 | 2003-07-15 | Arizona Chemical Company | Tertiary amide terminated polyamides in cosmetics |
| US6503077B2 (en) * | 1999-01-04 | 2003-01-07 | Arizona Chemical Company | Gelled articles containing tertiary amide-terminated polyamide |
| US6268466B1 (en) * | 1999-01-04 | 2001-07-31 | Arizona Chemical Company | Tertiary amide terminated polyamides and uses thereof |
| US6350889B1 (en) * | 1999-06-24 | 2002-02-26 | Arizona Chemical Company | Ink jet printing compositions containing ester-terminated dimer acid-based oligo (ester/amide) |
| US6407272B1 (en) * | 1999-07-14 | 2002-06-18 | Arizona Chemical Company | Secondary alcohol esters of hydroxyacids and uses thereof |
| US6958379B2 (en) * | 1999-12-03 | 2005-10-25 | Acushnet Company | Polyurea and polyurethane compositions for golf equipment |
| DE10009996B4 (en) * | 2000-03-02 | 2005-10-13 | Cognis Ip Management Gmbh | Solid granules with monodisperse particle size distribution, a process for their preparation and their use |
| DK1272267T3 (en) * | 2000-03-17 | 2004-06-07 | Bayer Cropscience Ag | microcapsule |
| US6492458B1 (en) * | 2000-05-16 | 2002-12-10 | Arizona Chemical Company | Polyalkyleneoxydiamine polyamides useful for formulating inks for phase-change jet printing |
| US6375966B1 (en) * | 2000-05-26 | 2002-04-23 | Scented Technologies, Llc | Polyurethane/polyurea matrices for the delivery of active agents |
| US7803058B2 (en) * | 2001-04-09 | 2010-09-28 | Chrisman Iii John W | Scented bowling balls and methods |
| US6552160B2 (en) * | 2001-05-14 | 2003-04-22 | Arizona Chemical Company | Ester-terminated poly(ester-amides) useful for formulating transparent gels in low polarity fluids |
| WO2002094900A1 (en) * | 2001-05-21 | 2002-11-28 | Dow Global Technologies Inc. | Transparent polyurethane-hydrogel composition, method of making transparent polyurethane-hydrogel composition, and air-freshener application |
| DK1453885T3 (en) * | 2001-12-13 | 2009-12-07 | Arizona Chem | Aqueous suspensions containing polymerized fatty acid-based polyamides |
| US7160337B2 (en) * | 2001-12-17 | 2007-01-09 | International Flavors & Fragrances Inc. | Transparent, vegetable-based, substantially hydrocarbon-free candle article |
| WO2003061817A1 (en) * | 2002-01-24 | 2003-07-31 | Bayer Aktiengesellschaft | Coagulates containing microcapsules |
| US6897281B2 (en) * | 2002-04-05 | 2005-05-24 | Noveon Ip Holdings Corp. | Breathable polyurethanes, blends, and articles |
| JP2005528200A (en) * | 2002-05-31 | 2005-09-22 | マクマスター・ユニバーシテイ | Method for encapsulating hydrophobic organic molecules in polyurea capsules |
| US7138475B2 (en) * | 2002-08-27 | 2006-11-21 | Acushnet Company | Compositions for golf equipment |
| US7335631B2 (en) * | 2002-09-09 | 2008-02-26 | Symrise, Inc. | Encapsulated perfume compositions in hair and skin products which release a burst of fragrance after initial topical application |
| US7125835B2 (en) * | 2002-10-10 | 2006-10-24 | International Flavors & Fragrances Inc | Encapsulated fragrance chemicals |
| DE10253257A1 (en) * | 2002-11-15 | 2004-06-03 | Air & D - Sarl | Process for the deodorization of large-scale plants |
| BR0317278A (en) * | 2002-12-13 | 2005-11-08 | Monsato Technology Llc | Microcapsules with adjusted amine release rates |
| US7253249B2 (en) * | 2003-04-22 | 2007-08-07 | Arizona Chemical Company | Ester-terminated poly(ester-amide) in personal care products |
| DE10335938A1 (en) * | 2003-08-04 | 2005-03-24 | Bruno Guillaume | Gel-like mass in a flat form for the treatment of ambient air |
| US7256162B2 (en) * | 2003-09-26 | 2007-08-14 | Arizona Chemical Company | Fatty acid esters and uses thereof |
| US7207669B2 (en) * | 2003-12-19 | 2007-04-24 | Arizona Chemical Company | Jet printing inks containing polymerized fatty acid-based polyamides |
| US8664292B2 (en) * | 2004-05-27 | 2014-03-04 | Croda International Plc | Compositions and articles containing a cross-linked polymer matrix and an immobilized active liquid, as well as methods of making and using the same |
| KR20070041690A (en) * | 2004-05-27 | 2007-04-19 | 아리조나 케미칼 캄파니 | Compositions and articles containing a crosslinked polymer matrix and an immobilized active liquid, as well as methods of making and using the same |
| US7906476B2 (en) * | 2005-02-11 | 2011-03-15 | Invista North America S.A.R.L. | Fabric care compositions |
| US20060248665A1 (en) * | 2005-05-06 | 2006-11-09 | Pluyter Johan G L | Encapsulated fragrance materials and methods for making same |
| JP5279176B2 (en) * | 2006-06-29 | 2013-09-04 | エア プロダクツ アンド ケミカルズ インコーポレイテッド | Curing agent for epoxy resin and coating composition |
| US20080223519A1 (en) * | 2006-12-06 | 2008-09-18 | Locko George A | Polyamide polyols and polyurethanes, methods for making and using, and products made therefrom |
| MX2010002966A (en) * | 2007-09-17 | 2010-04-01 | Arizona Chem | Polyamide and emollient compositions, products made therefrom, and methods of making and using such compositions and products. |
| US8058386B2 (en) * | 2008-09-16 | 2011-11-15 | Arizona Chemical Company, Llc | Polyalkylene glycol-based poly(ester-amide) polymers, methods of making and methods of using same, compositions and products comprising same |
-
2010
- 2010-11-22 US US12/951,421 patent/US20110117156A1/en not_active Abandoned
-
2011
- 2011-11-18 JP JP2013540043A patent/JP2013543049A/en active Pending
- 2011-11-18 BR BR112013012481A patent/BR112013012481A2/en not_active Application Discontinuation
- 2011-11-18 MX MX2013005683A patent/MX2013005683A/en unknown
- 2011-11-18 CN CN2011800561551A patent/CN103282193A/en active Pending
- 2011-11-18 EP EP11791152.9A patent/EP2643155A1/en not_active Withdrawn
- 2011-11-18 WO PCT/US2011/061349 patent/WO2012071261A1/en active Application Filing
- 2011-11-18 KR KR1020137015461A patent/KR20130117814A/en not_active Application Discontinuation
- 2011-11-21 TW TW100142506A patent/TW201237099A/en unknown
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH03258899A (en) | 1990-03-07 | 1991-11-19 | Koukandou:Kk | Fragrance emitting sheet having living thing activity |
| FR2695033A1 (en) * | 1992-08-25 | 1994-03-04 | Oreal | Prepn. of water-soluble melanic polymer by oxidn. of indole or indoline cpd. - in presence of water-soluble polymer, used in compsn. for colouring hair or skin or make=up compsn. giving natural brown colour. |
| US6111655A (en) | 1993-11-11 | 2000-08-29 | Dainippon Screen Mfg Co., Ltd. | Method and system for managing component data of the design process and prepress process of a printing operation |
| JPH07145299A (en) | 1993-11-25 | 1995-06-06 | Yokohama Rubber Co Ltd:The | Perfume material composition |
| US5780527A (en) | 1994-08-19 | 1998-07-14 | Firmenich Sa | Perfuming device for perfuming and sanitizing ambient air |
| US6503577B2 (en) | 2000-03-20 | 2003-01-07 | Sulzer Metco Ag | Method of thermally coating a cylinder barrel of a cylinder block of a combustion engine |
| US6399713B1 (en) | 2001-01-24 | 2002-06-04 | Arizona Chemical Company | Hydrocarbon-terminated polyether-polyamide block copolymers and uses thereof |
| US6870011B2 (en) | 2001-01-24 | 2005-03-22 | Arizona Chemical Company | Hydrocarbon-terminated polyether-polyamide block copolymers and uses thereof |
| WO2003020412A1 (en) * | 2001-08-28 | 2003-03-13 | International Fragrance & Technology, Inc. | Clear, polymeric gel composition and method for producing the same |
| US6846491B1 (en) | 2001-08-28 | 2005-01-25 | International Fragrance & Techology | Clear, polymeric gel composition and method for producing the same |
| US6956099B2 (en) | 2003-03-20 | 2005-10-18 | Arizona Chemical Company | Polyamide-polyether block copolymer |
Non-Patent Citations (4)
| Title |
|---|
| "The Epoxy Formulators Manual", 1984, SOCIETY OF PLASTICS INDUSTRY, INC. |
| HENRY LEE; KRIS NEVILLE: "Handbook of Epoxy Resins", 1967, MCGRAW HILL |
| KIRK-OTHMER: "Encyclopedia of Science and Technology", 1994, JOHN WILEY & SONS |
| See also references of EP2643155A1 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10526447B2 (en) | 2015-04-15 | 2020-01-07 | Houghton Technical Corp. | Materials that provide bioresistance and/or defoaming and slower cooling properties for aqueous quenchants |
| EP3192566A1 (en) | 2016-01-15 | 2017-07-19 | International Flavors & Fragrances Inc. | Polyalkoxy-polyimine adducts for use in delayed release of fragrance ingredients |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013543049A (en) | 2013-11-28 |
| US20110117156A1 (en) | 2011-05-19 |
| KR20130117814A (en) | 2013-10-28 |
| EP2643155A1 (en) | 2013-10-02 |
| CN103282193A (en) | 2013-09-04 |
| TW201237099A (en) | 2012-09-16 |
| MX2013005683A (en) | 2013-10-03 |
| BR112013012481A2 (en) | 2016-09-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8664292B2 (en) | Compositions and articles containing a cross-linked polymer matrix and an immobilized active liquid, as well as methods of making and using the same | |
| US20110117156A1 (en) | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same | |
| CA2567344C (en) | Compositions and articles containing a crosslinked polymer matrix and an immobilized active liquid, as well as methods of making and using the same | |
| US6730311B2 (en) | Air fresheners comprising polyurethane/polyurea matrices for the delivery of fragrance agents | |
| CA1207486A (en) | Polyurethane gels serviceable as controlled release agents | |
| AU611282B2 (en) | Matrix for release of active ingredients | |
| JPH0634811B2 (en) | Vessels, methods and compositions for controlling volatile liquid emissions from aqueous mixtures | |
| WO2008021631A1 (en) | Polymeric gel composition and method for making | |
| JPH0332579B2 (en) | ||
| US20060067963A1 (en) | Gel compositions and methods of producing same | |
| CA2817152A1 (en) | Compositions and articles containing an active liquid in a polymeric matrix and methods of making and using the same | |
| EP1142959A1 (en) | Gel-form molded resin composition, packaged volatile gel-form chemical, and process for producing the same | |
| EP3205331A1 (en) | Diminishing gel compositions and associated methods |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11791152 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2817152 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011791152 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2013540043 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2013/005683 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20137015461 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013012481 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013012481 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130520 |