WO2011153493A2 - Biodegradable lipids for the delivery of active agents - Google Patents
Biodegradable lipids for the delivery of active agents Download PDFInfo
- Publication number
- WO2011153493A2 WO2011153493A2 PCT/US2011/039164 US2011039164W WO2011153493A2 WO 2011153493 A2 WO2011153493 A2 WO 2011153493A2 US 2011039164 W US2011039164 W US 2011039164W WO 2011153493 A2 WO2011153493 A2 WO 2011153493A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- independently
- gene
- lipid
- compound
- alkyl
- Prior art date
Links
- 0 C*(CN(C)C)C1OC(C)(C)OC1 Chemical compound C*(CN(C)C)C1OC(C)(C)OC1 0.000 description 3
- KIKBRFJSKMZEQJ-MCOFMCJXSA-N CCOC(CCCC/C=C\CCCCCCCCC(CCCCCCCC/C=C\CCCCC(OC)=O)OC(CCCN(C)C)=O)=O Chemical compound CCOC(CCCC/C=C\CCCCCCCCC(CCCCCCCC/C=C\CCCCC(OC)=O)OC(CCCN(C)C)=O)=O KIKBRFJSKMZEQJ-MCOFMCJXSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/06—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton
- C07C229/10—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings
- C07C229/12—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings to carbon atoms of acyclic carbon skeletons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/44—Oils, fats or waxes according to two or more groups of A61K47/02-A61K47/42; Natural or modified natural oils, fats or waxes, e.g. castor oil, polyethoxylated castor oil, montan wax, lignite, shellac, rosin, beeswax or lanolin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/04—Immunostimulants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/62—Quaternary ammonium compounds
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/87—Introduction of foreign genetic material using processes not otherwise provided for, e.g. co-transformation
- C12N15/88—Introduction of foreign genetic material using processes not otherwise provided for, e.g. co-transformation using microencapsulation, e.g. using amphiphile liposome vesicle
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering N.A.
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2320/00—Applications; Uses
- C12N2320/30—Special therapeutic applications
Definitions
- the present invention relates to biodegradable lipids and to their use for the delivery of active agents such as nucleic acids.
- Therapeutic nucleic acids include, e.g. , small interfering RNA (siRNA), micro RNA (miRNA), antisense oligonucleotides, ribozymes, plasmids, immune stimulating nucleic acids, antisense, antagomir, antimir, microRNA mimic, supermir, Ul adaptor, and aptamer. These nucleic acids act via a variety of mechanisms. In the case of siRNA or miRNA, these nucleic acids can down-regulate intracellular levels of specific proteins through a process termed RNA interference (RNAi). Following introduction of siRNA or miRNA into the cell cytoplasm, these double- stranded RNA constructs can bind to a protein termed RISC.
- siRNA small interfering RNA
- miRNA micro RNA
- antisense oligonucleotides e.g., antisense oligonucleotides
- ribozymes e.g., antisense oligonucleo
- the sense strand of the siRNA or miRNA is displaced from the RISC complex providing a template within RISC that can recognize and bind mRNA with a complementary sequence to that of the bound siRNA or miRNA. Having bound the complementary mRNA the RISC complex cleaves the mRNA and releases the cleaved strands.
- RNAi can provide down-regulation of specific proteins by targeting specific destruction of the corresponding mRNA that encodes for protein synthesis.
- RNAi The therapeutic applications of RNAi are extremely broad, since siRNA and miRNA constructs can be synthesized with any nucleotide sequence directed against a target protein. To date, siRNA constructs have shown the ability to specifically down-regulate target proteins in both in vitro and in vivo models. In addition, siRNA constructs are currently being evaluated in clinical studies.
- siRNA or miRNA constructs Two problems currently faced by siRNA or miRNA constructs are, first, their susceptibility to nuclease digestion in plasma and, second, their limited ability to gain access to the intracellular compartment where they can bind RISC when administered systemically as the free siRNA or miRNA.
- These double- stranded constructs can be stabilized by incorporation of chemically modified nucleotide linkers within the molecule, for example, phosphothioate groups
- these chemical modifications provide only limited protection from nuclease digestion and may decrease the activity of the construct.
- Intracellular delivery of siRNA or miRNA can be facilitated by use of carrier systems such as polymers, cationic liposomes or by chemical modification of the construct, for example by the covalent attachment of cholesterol molecules.
- carrier systems such as polymers, cationic liposomes or by chemical modification of the construct, for example by the covalent attachment of cholesterol molecules.
- improved delivery systems are required to increase the potency of siRNA and miRNA molecules and reduce or eliminate the requirement for chemical
- Antisense oligonucleotides and ribozymes can also inhibit mRNA translation into protein.
- these single stranded deoxynucleic acids have a complementary sequence to that of the target protein mRNA and can bind to the mRNA by Watson-Crick base pairing. This binding either prevents translation of the target mRNA and/or triggers RNase H degradation of the mRNA transcripts. Consequently, antisense
- oligonucleotides have tremendous potential for specificity of action (i.e., down-regulation of a specific disease-related protein). To date, these compounds have shown promise in several in vitro and in vivo models, including models of inflammatory disease, cancer, and HIV (reviewed in Agrawal, Trends in Biotech. 14:376-387 (1996)). Antisense can also affect cellular activity by hybridizing specifically with chromosomal DNA. Advanced human clinical assessments of several antisense drugs are currently underway. Targets for these drugs include the bcl2 and apolipoprotein B genes and mRNA products.
- Immune- stimulating nucleic acids include deoxyribonucleic acids and ribonucleic acids.
- deoxyribonucleic acids certain sequences or motifs have been shown to illicit immune stimulation in mammals. These sequences or motifs include the CpG motif, pyrimidine-rich sequences and palindromic sequences. It is believed that the CpG motif in deoxyribonucleic acids is specifically recognized by an endosomal receptor, toll-like receptor 9 (TLR-9), which then triggers both the innate and acquired immune stimulation pathway.
- TLR-9 endosomal receptor
- Certain immune stimulating ribonucleic acid sequences have also been reported. It is believed that these RNA sequences trigger immune activation by binding to toll-like receptors 6 and 7 (TLR-6 and TLR-7). In addition, double- stranded RNA is also reported to be immune stimulating and is believe to activate via binding to TLR-3.
- siRNA has been administered systemically in cationic liposomes, and these nucleic acid-lipid particles have been reported to provide improved down-regulation of target proteins in mammals including non-human primates (Zimmermann et al, Nature 441: 111-114 (2006)).
- compositions that are suitable for general therapeutic use.
- these compositions would encapsulate nucleic acids with high-efficiency, have high drug:lipid ratios, protect the encapsulated nucleic acid from degradation and clearance in serum, be suitable for systemic delivery, and provide intracellular delivery of the encapsulated nucleic acid.
- these lipid-nucleic acid particles should be well-tolerated and provide an adequate therapeutic index, such that patient treatment at an effective dose of the nucleic acid is not associated with significant toxicity and/or risk to the patient.
- Compositions, methods of making the compositions, and methods of using the compositions to introduce nucleic acids into cells, including for the treatment of diseases are provided.
- the present invention relates to a cationic lipid having one or more biodegradable groups located in the mid- or distal section of a lipidic moiety (e.g., a hydrophobic chain) of the cationic lipid.
- a lipidic moiety e.g., a hydrophobic chain
- These cationic lipids may be incorporated into a lipid particle for delivering an active agent, such as a nucleic acid (e.g., an siRNA).
- an active agent such as a nucleic acid (e.g., an siRNA).
- the incorporation of the biodegradable group(s) into the cationic lipid results in faster metabolism and removal of the cationic lipid from the body following delivery of the active agent to a target area.
- these cationic lipids have substantially lower toxicity than similar cationic lipids without the biodegradable groups.
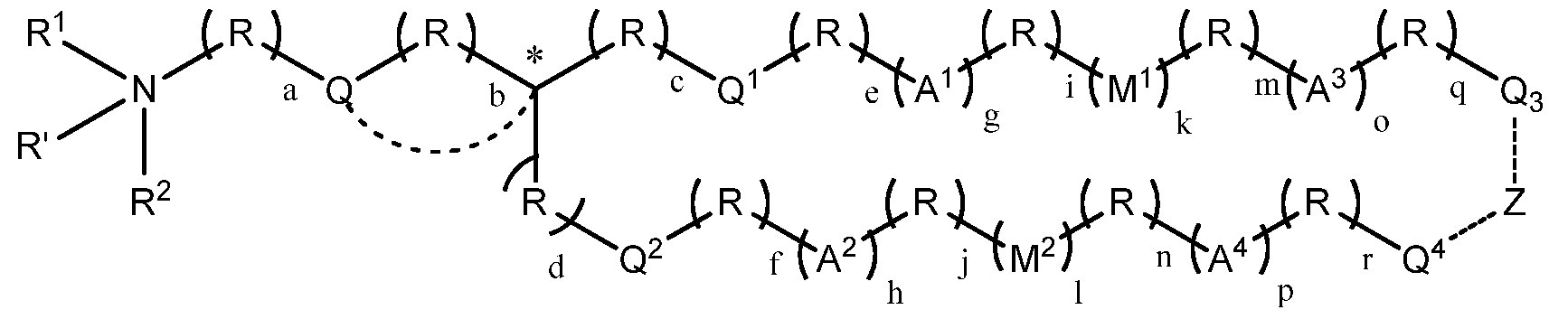
- the cationic lipid is a compound of the formula:
- R' is absent, hydrogen, or alkyl (e.g., Ci-C4 alkyl); with respect to 1 2
- R and R are each, independently, optionally substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, or heterocycle;
- R and R together with the nitrogen atom to which they are attached, form an optionally substituted heterocylic ring;
- R and R is optionally substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, or heterocycle, and the other forms a 4-10 member heterocyclic ring or heteroaryl (e.g., a 6-member ring) with (a) the adjacent nitrogen atom and (b) the (R) a group adjacent to the nitrogen atom; each occurrence of R is, independently, -(CR 3 R 4 )-; each occurrence of R 3 and R 4 are, independently H, OH, alkyl, alkoxy, -NH 2 , alkylamino, or dialkylamino (in one preferred embodiment, each occurrence of R 3 and R 4 are, independently H or C1-C4 alkyl); or R 3 and R 4 , together with the carbon atom to which they are directly attached, form a cycloalkyl group, wherein no more than three R groups in each chain attached to the carbon C* are cycloalkyl (e.g
- Q 1 and Q 2 are each, independently, absent, -0-, -S-, -OC(O)-, -C(0)0-, -SC(O)-, -C(0)S- , -OC(S)-, -C(S)0-, -S-S-, -C(0)(NR 5 )-, -N(R 5 )C(0)-, -C(S)(NR 5 )-, -N(R 5 )C(0)-, - N(R 5 )C(0)N(R 5 )-, or -OC(0)0-;
- a biodegradable group e.g.
- Z is absent, alkylene or -0-P(0)(OH)-0-; each attached to Z is an optional bond, such that when Z is absent, Q 3 and Q 4 are not directly covalently bound together;
- a is 1, 2, 3, 4, 5 or 6;
- b is 0, 1, 2, or 3;
- c, d, e, f, i, j, m, n, q and r are each, independently, 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10;
- g and h are each, independently, 0, 1 or 2;
- k and 1 are each, independently, 0 or 1, where at least one of k and 1 is 1; and
- o and p are each, independently, 0, 1 or 2,
- Q 3 and Q 4 are each, independently, separated from the tertiary carbon atom marked with an asterisk (*) by a chain of 8 or more atoms (e.g., 12 or 14 or more atoms).
- R 1 and R 2 are each, independently, optionally substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, or heterocycle; or (ii) R 1 and R 2 , together with the nitrogen atom to which they are attached, form an optionally substituted heterocylic ring.
- R 1 and R 2 are each, independently, optionally substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, or heterocycle; or (ii) R 1 and R 2 , together with the nitrogen atom to which they are attached, form an optionally substituted heterocylic ring.
- Q 3 and Q 4 are each, independently, separated from the tertiary carbon atom marked with an asterisk (*) by a chain of 10 or more atoms (e.g., 12 or 14 or more atoms).
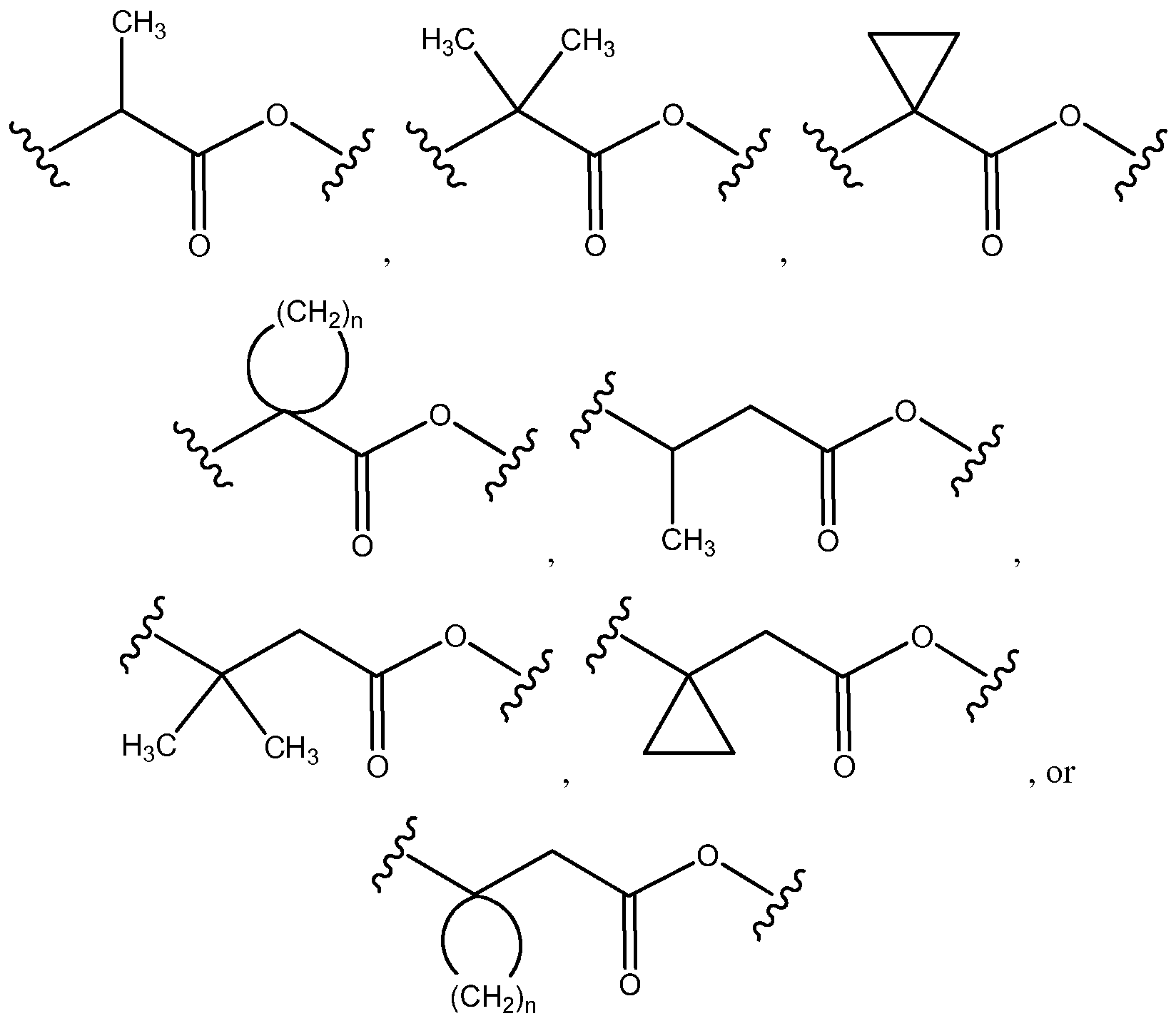
- a carbon atom alpha or beta to a biodegradable group in formula (I) may be substituted with one or two alkyl groups (e.g., one CrC 4 alkyl group, such as a -CH 3 substituent, or two Q-G* alkyl groups, such as two -CH 3 substituents) or have a spirocyclic group (e.g., a C3-C5 cycloalkyl such as a C 3 cycloalkyl).
- a carbon atom alpha or beta to a biodegradable group can be independently selected from
- the M 1 or M 2 group and neighboring variable(s) form the group:
- R 1 , R 2 , R, a, and b are as defined with respect to formula (I);
- R' is absent, hydrogen, or alkyl (e.g., C C 4 alkyl);
- each of R 9 and R 10 are independently C 12 -C24 alkyl (e.g., C 12 -C 2 o alkyl), C 12 -C 2 4 alkenyl (e.g., C 12 -C 2 o alkenyl), or C 12 -C 2 4 alkoxy (e.g., C 12 -C 2 o alkoxy) having one or more
- each biodegradable group independently interrupts the C 12 -C 2 4 alkyl, alkenyl, or alkoxy group or is substituted at the terminus of the C 12 -C 2 4 alkyl, alkenyl, or alkoxy group, wherein
- the cationic lipid is a compound of the formula:
- Formula (IA-2) or a salt thereof e.g., a pharmaceutically acceptable salt thereof, wherein R' is absent, hydrogen, or alkyl (e.g., C C 4 alkyl);
- R 1 and R 2" are each, independently, optionally substituted C C 4 alkyl, C2-C4 alkenyl, C 2 - C 4 alkynyl, C 3 -C 6 cycloalkyl, (C 3 -C 6 cycloalkyl)C 1 -C 4 alkyl, or a monocyclic heterocycle; or
- R 1 and R 2 together with the nitrogen atom to which they are attached, form an optionally substituted 5- or 6-membered heterocylic ring (e.g., a C5 or C 6 heterocyclic ring); each occurrence of R is, independently, -(CR 3 R 4 )-; each occurrence of R 3 and R 4 are, independently H, OH, alkyl, alkoxy, -NH 2 , alkylamino, or dialkylamino (in one preferred embodiment, each occurrence of R 3 and R 4 are, independently H or C1-C4 alkyl); or R 3 and R 4 , together with the carbon atom to which they are directly attached, form a C 3 -C 6 cycloalkyl group, wherein no more than three R groups in each chain attached to the carbon C* are cycloalkyl (e.g., cyclopropyl); the dashed line to Q is absent or a bond; when the dashed line to Q is absent, Q is absent or is -
- Z is absent, alkylene or -0-P(0)(OH)-0-; each attached to Z is an optional bond, such that when Z is absent, Q 3 and Q 4 are not directly covalently bound together;
- a is 1, 2, 3, 4, 5 or 6;
- b is 0, 1, 2, or 3;
- d, e, i, j, m, n, q and r are each, independently, 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10;
- g and h are each, independently, 0, 1 or 2; the sum of d + 3h is at least 4, and the sum of e + 3g is at least 4;
- k and 1 are each, independently, 0 or 1, where at least one of k and 1 is 1 ; and
- o and p are each, independently, 0, 1 or 2, wherein Q 3 and Q 4 are each, independently, separated from the tertiary carbon atom marked with an asterisk (*) by a chain of 8 or more atoms (e.g., 12
- R' in formula (IA-2) is absent or hydrogen. In one embodiment, R' in formula (IA-2) is absent or alkyl (e.g., methyl).
- R and R in formula (IA-2) are each, independently, C C 4 alkyl (e.g., methyl or ethyl).
- each occurrence of R in formula (IA-2) is, independently, -CH 2 - or - CH(CH 3 )-.
- Q 3 and Q 4 in formula (IA-2) are each, independently, H, aryl, or a cholesterol moiety.
- M and M in formula (IA-2) are each -C(0)-0-.
- Z is absent and each is absent (i.e., Q 3 and Q 4 are not directly covalently bound together).
- the sum of e+3g+i+m+3o+q in formula (IA-2) is from about 8 to about 20. In another embodiment, the sum of e+3g+i+m+3o+q in formula (IA-2) is from about 12 to about 20.
- the sum of d+3h+j+n+3p+r in formula (IA-2) is from about 8 to about 20. In another embodiment, the sum of d+3h+j+n+3p+r in formula (IA-2) is from about 12 to about 20.
- the cationic lipid is a compound of the formula
- R , R , R, a, b, M and are as defined with respect to formula (I);
- R' is absent, hydrogen, or alkyl (e.g., Ci-C4 alkyl); each of R 9 and R 10 are independently alkylene, or alkenylene; and each of R 11 and R 12 are independently alkyl or alkenyl, optionally terminated by COOR where each R 13 is independently alkyl (e.g., C C 4 alkyl such as methyl or ethyl);
- R 9 , M 1 , and R 11 are together at least 8 carbons atoms in length (e.g., 12 or 14 carbon atoms or longer);
- R 10 , M 2 , and R 12 are together at least 8 carbons atoms in length (e.g., 12 or 14 carbon atoms or longer).
- R 9 and R 10 are each
- R 9 , M 1 , and R 11 are C 4 -C 12 alkylene or C 4 -C 12 alkenylene.
- R 9 , M 1 , and R 11 are together at 12 to 24 carbons atoms in length.
- R 9 , M 1 , and R 11 are together at 14 to 18 carbons atoms in length.
- R 10 , M 2 , and R 12 are together at 12 to 24 carbons atoms in length.
- R 10 , M 2 , and R 12 are together at 14 to 18 carbons atoms in length.
- R'R R N-(R) a -Q-(R)t > - group can be any of the head groups described herein, including those shown in Table 1 below, and salts thereof.
- R 1 , R 2 , R, a, and b are as defined with respect to formula (I);
- R' is absent, hydrogen, or alkyl (e.g., C1-C4 alkyl); each of R 9 and R 10 are independently C 12 -C24 alkyl or alkenyl substituted at its termin
- R 9 and R 10 are each independently C 14 -C 18 alkylene or C 14 -C 18 alkenylene.
- the biodegradable group is -COOR 13 where R 13 is C C 4 alkyl (such as methyl or ethyl).
- the R'R R N-(R) a -Q-(R)t > - group can be any of the head groups described herein,
- R' is absent, hydrogen, or alkyl (e.g., CrC 4 alkyl);
- R 1 and 2 are each, independently, optionally substituted alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkylalkyl, or heterocycle; or R 1 and R 2 , together with the nitrogen atom to which they are attached, form an optionally substituted heterocylic ring; each occurrence of R is, independently, -(CR 3 R 4 )-; each occurrence of R 3 and R 4 are, independently H, OH, alkyl, alkoxy, -NH 2 , alkylamino, or dialkylamino (in one preferred embodiment, each occurrence of R 3 and R 4 are, independently H or alkyl); or R 3 and R 4 , together with the carbon atom to which they are directly attached, form a cycloalkyl group, wherein no more than three R groups in each chain attached to the carbon C* are cycloalkyl (e.g., cyclopropyl); the dashed line to Q is absent or a bond; when the
- Q 1 and Q 2 are each, independently, absent, -0-, -S-, -OC(O)-, -C(0)0-, -SC(O)-, -C(0)S- , -OC(S)-, -C(S)0-, -S-S-, -C(0)(NR 5 )-, -N(R 5 )C(0)-, -C(S)(NR 5 )-, -N(R 5 )C(0)-, - N(R 5 )C(0)N(R 5 )-, or -OC(0)0-;
- a biodegradable group e.g., -
- Z is absent, alkylene or -0-P(0)(OH)-0-; each attached to Z is an optional bond, such that when Z is absent, Q 3 and Q 4 are not directly covalently bound together;
- a is 1, 2, 3, 4, 5 or 6;
- b is 0, 1, 2, or 3;
- c, d, e, f, i, j, m, n, q and r are each, independently, 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10;
- g and h are each, independently, 0, 1 or 2;
- k and 1 are each, independently, 0 or 1 ;
- o and p are each, independently, 0, 1 or 2,
- Q 3 and Q 4 are each, independently, separated from the tertiary carbon atom marked with an asterisk (*) by a chain of 8 or more atoms (e.g., 12 or 14 or more atoms).
- the cationic lipid is a compound of formula IE:
- R 1 is a Cio to C 30 group having the formula -L la -(CR la R lb ) compassion-[L lb -(CR la R lb ) p ] y -L lc -R lc , where L la is a bond, -CR la R lb -, -0-, -CO-, -NR ld -, -S-, or a combination thereof; each R la and each R lb , independently, is H; halo; hydroxy; cyano; CrC 6 alkyl optionally substituted by halo, hydroxy, or alkoxy; C 3 -C8 cycloalkyl optionally substituted by halo, hydroxy, or alkoxy; -0R lc ; -NR lc R ld ; aryl; heteroaryl; or heterocyclyl;
- each L lb is a bond, -(CR la R lb )i_ 2 -, -0-, -CO-, -NR ld -, -S-, ,
- R lf and R lg are each independently R lb , or adjacent R lf and R lg , taken together, are optionally a bond;
- R lf and R lg are each independently R lb , or adjacent R lf and R lg , taken together, are optionally a bond;
- Het- is a 3 to 14 membered heterocyclylene or heteroarylene group optionally substituted by zero to six independent R la groups;
- L lc is -(CR la R lb ) 1-2 -, -0-, -CO-, -NR ld -, -S-, , , I I , or a combination thereof;
- R lc is H; halo; hydroxy; cyano; CrC 6 alkyl optionally substituted by halo, hydroxy, alkoxy, or aryl; C 3 -C 8 cycloalkyl optionally substituted by halo, hydroxy, alkoxy, or aryl; aryl;
- R is H; halo; hydroxy; cyano; CrC 6 alkyl optionally substituted by halo, hydroxy, or alkoxy; C 3 -C 8 cycloalkyl optionally substituted by halo, hydroxy, or alkoxy; aryl; heteroaryl; or heterocyclyl; a is 0-6, inclusive; each ⁇ , independently, is 0-6, inclusive; ⁇ is 0-6, inclusive;
- R 2 is a Cio to C 30 group having the formula -L 2a -(CR 2a R 2b ) 8 -[L 2b -(CR 2a R 2b ) E ] c -L 2c -R 2c , where L 2a is a bond, -CR 2a R 2b -, -0-, -CO-, -NR 2d -, -S-, or a combination thereof; each R 2a and each R 2b , independently, can be H; halo; hydroxy; cyano; C]_-C alkyl optionally substituted by halo, hydroxy, or alkoxy; C 3 -C8 cycloalkyl optionally substituted by halo, hydroxy, or alkoxy; -OR 2c ; -NR 2c R 2d ; aryl; heteroaryl; or heterocyclyl; each L 2b , independently, can be a bond, -(CR 2a R 2b )i_ 2
- R 2f and R 2g are each independently R 2b , or adjacent R 2f and R 2g , taken together, are optionally a bond;
- j and k are each independently 0, 1, 2, 3, or 4 provided that the sum of j and k is at least 1; and R 2f and R 2g are each independently R 2b , or adjacent R 2f and R 2g , taken together, are optionally a bond; -Aiy
- Het- is a 3 to 14 membered heterocyclylene or heteroarylene group optionally substituted by zero to six independent R 2a groups;
- R 2c is H; halo; hydroxy; cyano; Q-Ce alkyl optionally substituted by halo, hydroxy, alkoxy or aryl; C 3 -C 8 cycloalkyl optionally substituted by halo, hydroxy, alkoxy or aryl; aryl;
- R c has the formula:
- R 2d is H; halo; hydroxy; cyano; C]_-C alkyl optionally substituted by halo, hydroxy, or alkoxy; C 3 -C 8 cycloalkyl optionally substituted by halo, hydroxy, or alkoxy; aryl; heteroaryl; or heterocyclyl; ⁇ is 0-6, inclusive; each ⁇ , independently, is 0-6, inclusive; ⁇ is 0-6, inclusive;
- U is C(R a ), -(CR 5 R 6 ) x C(R a )-, or P(Q 2 );
- R a is H, alkyl, alkoxy, -OH, -N(Q)Q, or -SQ;
- Yi is alkyl, cycloalkyl, aryl, aralkyl, or alkynyl, wherein Yi is optionally substituted by 0 to 6 independent R n ;
- Y 2 is alkyl, cycloalkyl, aryl, aralkyl, or alkynyl, wherein Y 2 is optionally substituted by 0 to 6 independent R n ;
- Y 3 is absent, or if present, is alkyl, cycloalkyl, aryl, aralkyl, or alkynyl, wherein Y 3 is optionally substituted by 0 to 6 independent R n ;
- Y 4 is absent, or if present, is alkyl, cycloalkyl, aryl, aralkyl, or alkynyl, wherein Y 4 is optionally substituted by 0 to 6 independent R n ; or any two of Y 1 ; Y 2 , and Y are taken together with the N atom to which they are attached to form a 3- to 8- member heterocycle optionally substituted by 0 to 6 independent R n ; or Y 1 ; Y 2 , and Y 3 are all be taken together with the N atom to which they are attached to form a bicyclic 5- to 12- member heterocycle optionally substituted by 0 to 6 independent R n ; each R n , independently, can be H, halo, cyano, hydroxy, amino, alkyl, alkoxy, cycloalkyl, aryl, heteroaryl, or heterocyclyl;
- L 3 is a bond, -N(Q)-, -0-, -S-, -(CR 7 Rg) a -, -C(O)-, or a combination of any two of these;
- L 4 c is a bond, -N(Q)-, -0-, -S-, -(CR 7 Rg) a -, -C(0)-, or a combination of any two of these;
- L5 is a bond, -N(Q)-, -0-, -S-, -(CR 7 Rg) a -, -C(0)-, or a combination of any two of these; each occurrence of R 7 and R 8 is, independently, H, halo, cyano, hydroxy, amino, alkyl, alkoxy, cycloalkyl, aryl, heteroaryl, or heterocyclyl; or two R 7 groups on adjacent carbon atoms can be taken together to form a double bond between their respective carbon atoms; or two R 7 groups on adjacent carbon atoms and two R 8 groups on the same adjacent carbon atoms can be taken together to form a triple bond between their respective carbon atoms; or, an R 7 or Rg substituent from any of L 3 , L 4 , or L5 can be optionally taken with an R 7 or Rg substituent from any of L 3 , L 4 , or L5 to form a 3- to 8-member cycloalkyl, heterocyclyl,
- Each Q 2 is O, S, N(Q)Q, alkyl or alkoxy.
- Li can be -C(RsR 6 ) x C(R a )-; or Li can be -CH 2 -C(R a )-.
- at least one L la , L lb , and L lc present in the compound is a biodegradable group, such as ester -C(0)0-, -OC(O)-, disulfide (-S-S-), -C(R 5 )
- a in R 1 is at least 4
- ⁇ in R 2 is at least 4
- at least one L la , L lb , and L lc present in the compound is a biodegradable group and at one L 2a , L 2b , or L 2c present in the compound is a biodegradable group
- the carbon chain in R 1 and/or R 2 is saturated. In yet another embodiment, the carbon chain in R 1 and/or R 2 contains one or two double bonds.
- the cationic lipid is a compound selected from compounds of formulas II-XXIII:
- the present invention relates to a cationic lipid or a salt thereof having:
- each hydrophobic tail comprising a Cg or greater aliphatic group (preferably a C 14 or greater aliphatic group) attached to the central carbon atom, where one or both of the aliphatic group(s) (a) is interrupted by a biodegradable group such that there is a chain of at least four carbon atoms between the biodegradable group and the central carbon atom, or (b) includes a biodegradable group at the terminal end of the hydrophobic tail.
- the biodegradable group is selected from -OC(O)-, -C(0)0-, -SC(O)-, -C(0)S-, -OC(S)-, -C(S)0-, -S-S-, -C(0)(NR 5 )-, -N(R 5 )C(0)-, - C(S)(NR 5 )-, -N(R 5 )C(0)-, -N(R 5 )C(0)N(R 5 )-, and -OC(0)0-.
- lipid particle that includes a cationic lipid of the present invention.
- the lipid particle includes a compound of any of formulas II- XXIII as described herein.
- the lipid particle includes a compound of formula I as described herein.
- the lipid particle includes a compound of formula IA-1, IA-2, IB, IC, ID or IE as described herein.
- the lipid particle includes a neutral lipid, a lipid capable of reducing aggregation, a cationic lipid, and optionally, a sterol (e.g., cholesterol).
- Suitable neutral lipids include, but are not limited to, distearoylphosphatidylcholine (DSPC),
- Suitable lipids capable of reducing aggregation include, but are not limited to, a PEG lipid, such as PEG-DMA, PEG- DMG, or a combination thereof.
- the lipid particle may further include an active agent (e.g., a therapeutic agent).
- the active agent can be a nucleic acid such as a plasmid, an immuno stimulatory oligonucleotide, an siRNA, an antisense oligonucleotide, a microRNA, an antagomir, an aptamer, or a ribozyme.
- the lipid particle includes a cationic lipid of the present invention, a neutral lipid and a sterol.
- the lipid particle may further include an active agent, such as a nucleic acid.
- a pharmaceutical composition which includes a lipid particle of the present invention and a pharmaceutically acceptable carrier.
- Yet another embodiment is a method of delivering a nucleic acid molecule in a subject comprising administering to the subject a lipid particle comprising the nucleic acid molecule and a cationic lipid (or a salt thereof), the cationic lipid having
- each hydrophobic tail comprising a Cg or greater aliphatic group (preferably a C 14 or greater aliphatic group) attached to the central carbon atom, where one or both of the aliphatic group(s) (a) is interrupted by a biodegradable group such that there is a chain of at least four carbon atoms between the biodegradable group and the central carbon atom, or (b) includes a biodegradable group at the terminal end of the hydrophobic tail.
- the cationic lipid remains intact until delivery of the nucleic acid molecule after which cleavage of the hydrophobic tail occurs in vivo.
- Yet another aspect is a method of modulating the expression of a target gene in a cell by providing to the cell a lipid particle of the present invention.
- the active agent can be a nucleic acid selected from a plasmid, an immuno stimulatory oligonucleotide, an siRNA, an antisense oligonucleotide, a microRNA, an antagomir, an aptamer, and a ribozyme.
- Yet another aspect is a method of treating a disease or disorder characterized by the overexpression of a polypeptide in a subject by providing to the subject a pharmaceutical composition of the present invention, wherein the active agent is a nucleic acid selected from an siRNA, a microRNA, and an antisense oligonucleotide, and wherein the siRNA, microRNA, or antisense oligonucleotide includes a polynucleotide that specifically binds to a polynucleotide that encodes the polypeptide, or a complement thereof.
- the active agent is a nucleic acid selected from an siRNA, a microRNA, and an antisense oligonucleotide
- the siRNA, microRNA, or antisense oligonucleotide includes a polynucleotide that specifically binds to a polynucleotide that encodes the polypeptide, or a complement thereof.
- Yet another aspect is a method of treating a disease or disorder characterized by underexpression of a polypeptide in a subject by providing to the subject a pharmaceutical composition of the present invention, wherein the active agent is a plasmid that encodes the polypeptide or a functional variant or fragment thereof.
- Yet another aspect is a method of inducing an immune response in a subject by providing to the subject a pharmaceutical composition wherein the active agent is an immuno stimulatory oligonucleotide.
- composition or lipid particles described above include a nucleic acid.
- the agent when contacted with cells, can efficiently deliver nucleic acids to the cells.
- a method of delivering a nucleic acid to the interior of a cell by obtaining or forming a composition or lipid particles described above, and contacting the composition or lipid particles with a cell.
- Figure 1 is a graph of the concentration of a cationic lipid (Compounds 1-3 and reference lipid) in the liver of mice over time, after administration of the cationic lipid in a lipid particle as described in Example 14.
- FIG. 1 shows the anticipated metabolic pathway for compounds 1 and 3 of Example 14.
- Figure 3 is a graph of the concentration of a cationic lipid (Compounds 1-3 and reference lipid) in the spleen of mice over time, after administration of the cationic lipid in a lipid particle as described in Example 14.
- Figure 4 is a graph of the concentration of a cationic lipid (Compounds 1-3 and reference lipid) in the plasma of mice over time, after administration of the cationic lipid in a lipid particle as described in Example 14. Detailed Description
- the present invention relates to a lipid particle that includes a neutral lipid, a lipid capable of reducing aggregation, a cationic lipid, and optionally a sterol.
- the lipid particle further includes an active agent (e.g., a therapeutic agent).
- the cationic lipid is a compound of formula I-XXIII. In another embodiment, the cationic lipid is a compound of one of formulas II-XXIII. In one embodiment, the cationic lipid is a compound of formula I. In another embodiment, the cationic lipid is a compound of formula IA-1, IA-2, IB, IC or ID. The following disclosure represents various embodiments of a compound of Formula I.
- M 1 and M 2 are each, independently:
- M 1 and M 2 are each, independently:
- M 1 and M 2 are each, independently:
- M 1 and M 2 are each -C(0)0-.
- R 1 and R 2 are each, individually, optionally substituted alkyl, cycloalkyl, cycloalkylalkyl, or heterocycle.
- R 1 is alkyl and R 2 is alkyl, cycloalkyl or cycloalkylalkyl.
- R 1 and R 2 are each, individually, alkyl (e.g.,
- R 1 and R 2 are both methyl.
- R 1 and R 2 together with the nitrogen atom to which they are attached, form an optionally substituted heterocylic ring (e.g., -methylpiperazinyl).
- an optionally substituted heterocylic ring e.g., -methylpiperazinyl
- one of R 1 and R 2 is or (e.g., R 1 is one of the two aforementioned groups and R is hydrogen).
- R' is hydrogen or alkyl. In another embodiment, R' is hydrogen or methyl. In one embodiment, R' is absent. In one embodiment, R' is absent or methyl.
- the nitrogen atom to which R' is attached carries a positive charge, and the compound also contains a negatively charged counter ion.
- the counterion can be any anion, such as an organic or inorganic anion. Suitable examples of anions include, but are not limited to, tosylate, methanesulfonate, acetate, citrate, malonate, tartarate, succinate, benzoate, ascorbate, a-ketoglutarate, a- glycerophosphate, halide (e.g., chloride), sulfate, nitrate, bicarbonate, and carbonate.
- the counterion is a halide (e.g., CI).
- each R is, independently, -(CR 3 R 4 )-, wherein R 3 and R 4 are each, independently, H or alkyl (e.g., C C 4 alkyl).
- each R is, independently, -(CHR 4 )-, wherein each R 4 is, independently H or alkyl (e.g., CrC 4 alkyl).
- each R is, independently, -CH 2 -, -C(CH 3 ) 2 - or -CH(iPr)- (where iPr is isopropyl).
- each R is -CH 2 -.
- R 5 is, in each case, hydrogen or methyl.
- R 5 can be, in each case, hydrogen.
- Q is -C(0)0-.
- Q 1 and Q 2 are each, independently, absent or -0-.
- Q 1 and Q2 are each absent.
- Q 1 and Q2 are each -0-.
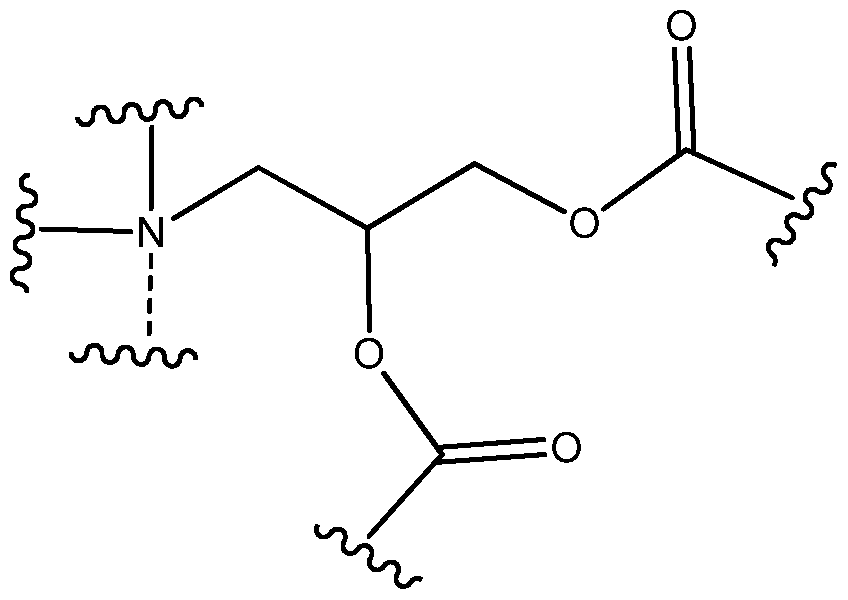
- the dashed line to Q is absent, b is 0 and R'R 1 R 2 N-(R) a -Q- and the tertiary carbon adjacent to it (C*) form the following group:
- n 1 to 4 (e.g., n is 2).
- the dashed line to Q is absent, b is 0 and R'R 1 R 2 N-(R) a -Q- and the tertiary carbon adjacent to it form the following group:
- n 1 to 4 (e.g., n is 2), and R 1 , R 2 , R, a, and b are as defined with respect to formula (I). In one embodiment, a is 3.
- the dashed line to Q is absent, b is 0 and R'R 1 R 2 N-(R) a -Q- and the tertiary carbon adjacent to it form the following group:
- n 1 to 4 (e.g., n is 2)
- R 1 , R 2 , R, a, and b are as defined with respect to formula (I).
- a is 0.
- the group can be:
- b is 0. In another embodiment, a is 2, 3, or 4 and b is 0. For example embodiment, a is 3 and b is 0. In another embodiment, a is 3, b is 0, and Q is -C(0)0-.
- the compound of formula (I) is of subformula:
- R, R', R 1 , R 2 , A 1 , A 2 , A 3 , A 4 , Q 1 , Q 2 , Q 3 , Q 4 , c, d, e, f, g, h, i, j, k, 1, m, n, o, p, q and r are as defined in any of the embodiments disclosed herein.
- the aliphatic group in one or both of the hydrophobic tails of the cationic lipid includes at least one carbon-carbon double bond.
- a suitable cholesterol moiety for the cationic lipids of the present invention (including compounds of formulas (I), IA-2, ID, IE and IF) has the formula:
- Additional embodiments include a cationic lipid having a head group, one or more hydrophobic tails, and a linker between the head group and the one or more tails.
- the head group can include an amine; for example an amine having a desired pK a .
- the pK a can be influenced by the structure of the lipid, particularly the nature of head group; e.g., the presence, absence, and location of functional groups such as anionic functional groups, hydrogen bond donor functional groups, hydrogen bond acceptor groups, hydrophobic groups (e.g., aliphatic groups), hydrophilic groups (e.g., hydroxyl or methoxy), or aryl groups.
- the head group amine can be a cationic amine; a primary, secondary, or tertiary amine; the head group can include one amine group (monoamine), two amine groups (diamine), three amine groups (triamine), or a larger number of amine groups, as in an oligoamine or polyamine.
- the head group can include a functional group that is less strongly basic than an amine, such as, for example, an imidazole, a pyridine, or a guanidinium group.
- the head group can be zwitterionic. Other head groups are suitable as well.
- the one or more hydrophobic tails can include two hydrophobic chains, which may be the same or different.
- the tails can be aliphatic, for example, they can be composed of carbon and hydrogen, either saturated or unsaturated but without aromatic rings.
- the tails can be fatty acid tails. Some such groups include octanyl, nonanyl, decyl, lauryl, myristyl, palmityl, stearyl, a-linoleyl, stearidonyl, linoleyl, ⁇ -linolenyl, arachadonyl, and oleyl. Other hydrophobic tails are suitable as well.
- the linker can include, for example, a glyceride linker, an acyclic glyceride analog linker, or a cyclic linker (including a spiro linker, a bicyclic linker, and a polycyclic linker).
- the linker can include functional groups such as an ether, an ester, a phosphate, a phosphonate, a phosphorothioate, a sulfonate, a disulfide, an acetal, a ketal, an imine, a hydrazone, or an oxime.
- Other linkers and functional groups are suitable as well.
- the cationic lipid is a racemic mixture.
- the cationic lipid is enriched in one diastereomer, e.g. the cationic lipid has at least 95%, at least 90%, at least 80% or at least 70% diastereomeric excess. In yet another embodiment, the cationic lipid is enriched in one enantiomer, e.g. the lipid has at least 95%, at least 90%, at least 80% or at least 70% enantiomer excess. In yet another embodiment, the cationic lipid is chirally pure, e.g. is a single optical isomer. In yet another embodiment, the cationic lipid is enriched for one optical isomer.
- a double bond e.g., a carbon-carbon double bond or carbon-nitrogen double bond
- isomerism in the configuration about the double bond (i.e. cis/trans or E/Z isomerism).
- the configuration of a double bond is illustrated in a chemical structure, it is understood that the corresponding isomer can also be present.
- the amount of isomer present can vary, depending on the relative stabilities of the isomers and the energy required to convert between the isomers. Accordingly, some double bonds are, for practical purposes, present in only a single configuration, whereas others (e.g., where the relative stabilities are similar and the energy of conversion low) may be present as inseparable equilibrium mixture of configurations.
- a double-bonded unsaturation can be replaced by a cyclic unsaturation.
- the cyclic unsaturation can be a cycloaliphatic unsaturation, e.g., a cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, or cyclooctyl group.
- the cyclic group can be a polycyclic group, e.g., a bicyclic group or tricyclic group. A bicyclic group can be bridged, fused, or have a spiro structure.
- a double y can be replaced by a cyclopropyl moiety, e.g., can be replaced by
- the moiety shown below has two carbon-carbon double bonds, each of which can independently be replaced by a cyclic moiety, e.g., a cyclopropyl moiety.
- substitutes for: can include:
- the cationic lipid includes one or more biodegradable groups.
- the biodegradable group(s) include one or more bonds that may undergo bond breaking reactions in a biological environment, e.g., in an organism, organ, tissue, cell, or organelle.
- Functional groups that contain a biodegradable bond include, for example, esters, dithiols, and oximes.
- Biodegradation can be a factor that influences the clearance of the compound from the body when administered to a subject. Biodegredation can be measured in a cell based assay, where a formulation including a cationic lipid is exposed to cells, and samples are taken at various time points.
- the lipid fractions can be extracted from the cells and separated and analyzed by LC-MS. From the LC-MS data, rates of biodegradation (e.g., as t 1/2 values) can be measured.
- Compound 1 includes an ester linkage in each aliphatic chain, which can undergo hydrolysis in a biological environment, for example, when exposed to, e.g., a lipase or an esterase.
- the structure of the compound influences the rate at which the compound undergoes biodegradation.
- a related compound such as
- Compound 2 would be expected to exhibit a different rate of biodegradation. Greater effects on that rate would be expected from changes in the structure of the compound at the site of hydrolysis.
- a cationic lipid of any of the embodiments described herein has an in vivo half life (t ⁇ ) (e.g., in the liver, spleen or plasma) of less than about 3 hours, such as less than about 2.5 hours, less than about 2 hours, less than about 1.5 hours, less than about 1 hour, less than about 0.5 hour or less than about 0.25 hours.
- t ⁇ in vivo half life
- a cationic lipid of any of the embodiments described herein containing a biodegradable group or groups has an in vivo half life (t ⁇ ) (e.g., in the liver, spleen or plasma) of less than about 10% (e.g., less than about 7.5%, less than about 5%, less than about 2.5%) of that for the same cationic lipid without the biodegrable group or groups.
- t ⁇ in vivo half life
- Some cationic lipids can be conveniently represented as a hydrophobic group combined with a headgroup.
- head groups include those depicted in Table 1:
- X halogen (e.g., CI)
- X halogen (e.g., CI)
- hydrophobic tail groups include those depicted in Table 2:
- the present invention relates to a method of preparing a compound of any of formulas I-XXIII.
- Suitable exemplary synthetic methods are illustrated in Schemes A-G below.
- the variables in the schemes below are the same as those variables at the same position in formulas I-XXIII above.
- the lipid chain length and linker length in Scheme A can be varied. Additionally, the R group in the ester functionality and substituents on the nitrogen atom can be derivatized.
- Rx is alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, aryl, or substituted aryl, and HEAD Group is defined in Table 1.
- the HEAD Group in Scheme C can be any head group disclosed herein (see, e.g., Table
- cationic lipids of the present invention include those shown in Tables 3-13 below, and salts thereof (including pharmaceutically acceptable salts thereof).
- the following compounds may be used as intermediates in the synthesis of cationic lipids according to the present invention.
- the cationic lipid of the present invention is selected from the following compounds, and salts thereof (including pharmaceutically acceptable salts thereof):
- Compound 3 Cationic lipids include those having alternative fatty acid groups and other dialkylamino groups, including those in which the alkyl substituents are different (e.g.,
- R 1 and R 2 are both long chain alkyl, alkenyl, alkynyl, or cycloalkylalkyl groups, they can be the same or different.
- lipids e.g., a cationic lipid
- Cationic lipids containing unsaturated fatty acids with carbon chain lengths in the range of C 10 to C 2 o are typical.
- Other scaffolds can also be used to separate the amino group (e.g., the amino group of the cationic lipid) and the fatty acid or fatty alkyl portion of the cationic lipid. Suitable scaffolds are known to those of skill in the art.
- cationic lipids have at least one protonatable or deprotonatable group, such that the lipid is positively charged at a pH at or below physiological pH (e.g. pH 7.4), and neutral at a second pH, preferably at or above physiological pH.
- a pH at or below physiological pH e.g. pH 7.4
- a second pH preferably at or above physiological pH.
- Such lipids are also referred to as cationic lipids.
- the lipids can have more than one protonatable or deprotonatable group, or can be zwiterrionic.
- protonatable lipids i.e., cationic lipids
- lipids will have a pK a of the protonatable group in the range of about 4 to about 11.
- lipids will have a pK a of about 4 to about 7, e.g., between about 5 and 7, such as between about 5.5 and 6.8, when incorporated into lipid particles.
- Such lipids will be cationic at a lower pH formulation stage, while particles will be largely (though not completely) surface neutralized at physiological pH around pH 7.4.
- pK a measurements of lipids within lipid particles can be performed, for example, by using the fluorescent probe 2-(p-toluidino)-6-napthalene sulfonic acid (TNS), using methods described in Cullis et al., (1986) Chem Phys Lipids 40, 127-144, which is incorporated by reference in its entirety.
- the lipids are charged lipids.
- charged lipid is meant to include those lipids having one or two fatty acyl or fatty alkyl chains and a quaternary amino head group.
- the quaternary amine carries a permanent positive charge.
- the head group can optionally include a ionizable group, such as a primary, secondary, or tertiary amine that may be protonated at physiological pH.
- a charged lipid is referred to as an "amino lipid.” See, for example, provisional U.S. patent application 61/267,419, filed December 7, 2009, which is incorporated by reference in its entirety.
- cationic lipids which carry a net positive charge at about physiological pH, in addition to those specifically described above, may also be included in lipid particles and compositions described herein.
- cationic lipids include, but are not limited to, N,N-dioleyl-N,N-dimethylammonium chloride ("DODAC");
- DOTMA N-(2,3-dioleyloxy)propyl-N,N-N-triethylammonium chloride
- DDAB N,N-distearyl-N,N-dimethylammonium bromide
- DOTAP N-(2,3-dioleoyloxy)propyl)-N,N,N-trimethylammonium chloride
- DC-Chol 3P-(N-(N',N'-dimethylaminoethane)-carbamoyl)cholesterol
- DOPE l,2-dileoyl-sn-3-phosphoethanolamine
- DODAP l,2-dioleoyl-3-dimethylammonium propane
- DODMA N, N-dimethyl-2,3-dioleyloxy)propylamine
- DMRIE N-(l,2-dimyristyloxyprop-3-yl)-N,N-dimethyl-N-hydroxyethyl ammonium bromide
- LIPOFECTIN including DOTMA and DOPE, available from GIBCO/BRL
- DOPE GIBCO/BRL
- LIPOFECT AMINE comprising DOSPA and DOPE, available from GIBCO/BRL.
- a cationic lipid is an amino lipid.
- the lipid particles and compositions described herein may also include one or more neutral lipids.
- Neutral lipids when present, can be any of a number of lipid species which exist either in an uncharged or neutral zwitterionic form at physiological pH.
- Such lipids include, for example, diacylphosphatidylcholine, diacylphosphatidylethanolamine, ceramide, sphingomyelin, dihydrosphingomyelin, cephalin, and cerebrosides.
- the selection of neutral lipids for use in the particles described herein is generally guided by consideration of, e.g. , liposome size and stability of the liposomes in the bloodstream.
- the neutral lipid component is a lipid having two acyl groups, (i.e., diacylphosphatidylcholine and diacylphosphatidylethanolamine).
- Lipids having a variety of acyl chain groups of varying chain length and degree of saturation are available or may be isolated or synthesized by well-known techniques.
- lipids containing saturated fatty acids with carbon chain lengths in the range of Cio to C 2 o are preferred.
- lipids with mono or diunsaturated fatty acids with carbon chain lengths in the range of C 10 to C 2 o are used.
- lipids having mixtures of saturated and unsaturated fatty acid chains can be used.
- the neutral lipids used are DOPE, DSPC, POPC, DPPC or any related phosphatidylcholine.
- the neutral lipids may also be composed of sphingomyelin, dihydrosphingomyeline, or
- phospholipids with other head groups such as serine and inositol.
- the lipid particles and compositosn described herein may also include one or more lipids capable of reducing aggregation.
- lipids that reduce aggregation of particles during formation include polyethylene glycol (PEG)-modified lipids, mono sialoganglio side Gml, and polyamide oligomers ("PAO") such as (described in U.S. Patent No. 6,320,017, which is incorporated by reference in its entirety).
- PEG polyethylene glycol
- PAO polyamide oligomers
- ATTA-lipids are described, e.g. , in U.S. Patent No. 6,320,017
- PEG-lipid conjugates are described, e.g. , in U.S. Patent Nos. 5,820,873, 5,534,499 and
- the concentration of the lipid component selected to reduce aggregation is about 1 to 15% (by mole percent of lipids).
- PEG-modified lipids or lipid-polyoxyethylene conjugates
- anchoring lipid portions to secure the PEG portion to the surface of the lipid vesicle
- PEG-modified phosphatidylethanolamine and phosphatidic acid PEG-ceramide conjugates (e.g., PEG-CerC14 or PEG-CerC20) which are described in U.S. Patent No.
- PEG-modified dialkylamines and PEG-modified l,2-diacyloxypropan-3-amines. Particularly preferred are PEG-modified diacylglycerols and dialkylglycerols.
- lipid anchor In embodiments where a sterically-large moiety such as PEG or ATTA are conjugated to a lipid anchor, the selection of the lipid anchor depends on what type of association the conjugate is to have with the lipid particle. It is well known that mPEG
- the PEG-modified lipid may be rapidly lost from the nucleic acid-lipid particle in vivo and hence the PEG-modified lipid will possess relatively short lipid anchors. In other therapeutic applications it may be preferable for the nucleic acid-lipid particle to exhibit a longer plasma circulation lifetime and hence the PEG-modified lipid will possess relatively longer lipid anchors.
- aggregation preventing compounds do not necessarily require lipid conjugation to function properly. Free PEG or free ATTA in solution may be sufficient to prevent aggregation. If the particles are stable after formulation, the PEG or ATTA can be dialyzed away before administration to a subject. Lipid Particles
- the present invent relates to lipid particles that include one or more of the cationic lipids described herein.
- the lipid particle includes one or more compound of formula I- XXIII.
- the lipid particle includes one or more compound of formula II-XXIII.
- the lipid particle includes one or more compound of formula I.
- the lipid particle includes a compound of formula IA-1, IA-2, IB, IC,ID or IE.
- Lipid particles include, but are not limited to, liposomes.
- a liposome is a structure having lipid-containing membranes enclosing an aqueous interior. Liposomes may have one or more lipid membranes. Liposomes can be single-layered, referred to as unilamellar, or multi-layered, referred to as multilamellar. When complexed with nucleic acids, lipid particles may also be lipoplexes, which are composed of cationic lipid bilayers sandwiched between DNA layers.
- the lipid particles may further comprise one or more additional lipids and/or other components such as cholesterol.
- Other lipids may be included in the liposome compositions for a variety of purposes, such as to prevent lipid oxidation or to attach ligands onto the liposome surface. Any of a number of lipids may be present in liposomes, including amphipathic, neutral, cationic, and anionic lipids. Such lipids can be used alone or in combination.
- bilayer stabilizing components such as polyamide oligomers (see, e.g. , U.S. Patent No. 6,320,017, which is incorporated by reference in its entirety), peptides, proteins, detergents, lipid-derivatives, such as PEG coupled to phosphatidylethanolamine and PEG conjugated to ceramides (see, U.S. Patent No. 5,885,613, which is incorporated by reference in its entirety).
- the lipid particles include one or more of a second amino lipid or cationic lipid, a neutral lipid, a sterol, and a lipid selected to reduce aggregation of lipid particles during formation, which may result from steric stabilization of particles which prevents charge-induced aggregation during formation.
- Lipid particles can include two or more cationic lipids.
- the lipids can be selected to contribute different advantageous properties.
- cationic lipids that differ in properties such as amine pK a , chemical stability, half-life in circulation, half-life in tissue, net accumulation in tissue, or toxicity can be used in a lipid particle.
- the cationic lipids can be chosen so that the properties of the mixed- lipid particle are more desireable than the properties of a single-lipid particle of individual lipids.
- Net tissue accumulation and long term toxicity (if any) from the cationic lipids can be modulated in a favorable way by choosing mixtures of cationic lipids instead of selecting a single cationic lipid in a given formulation. Such mixtures can also provide better encapsulation and/or release of the drug. A combination of cationic lipids also can affect the systemic stability when compared to single entity in a formulation.
- a series of structurally similar compounds can have varying pK a values that span a range, e.g. of less than 1 pK a unit, from 1 to 2 pK a units, or a range of more than 2 pK a units.
- a pK a in the middle of the range is associated with an enhancement of advantageous properties (greater effectiveness) or a decrease in disadvantageous properties (e.g., reduced toxicity), compared to compounds having pK a values toward the ends of the range.
- two (or more) different compounds having pK a values toward opposing ends of the range can be selected for use together in a lipid particle.
- the net properties of the lipid particle (for instance, charge as a function of local pH) can be closer to that of a particle including a single lipid from the middle of the range.
- Cationic lipids that are structurally dissimilar (for example, not part of the series of structurally similar compounds mentioned above) can also be used in a mixed-lipid particle.
- two or more different cationic lipids may have widely differing pK a values, e.g., differing by 3 or more pK a units.
- the net behavior of a mixed lipid particle will not necessarily mimic that of a single-lipid particle having an intermediate pK a . Rather, the net behavior may be that of a particle having two distinct protonatable (or deprotonatable, as the case may be) site with different pK a values.
- the fraction of protonatable sites that are in fact protonated varies sharply as the pH moves from below the pK a to above the pK a (when the pH is equal to the pK a value, 50% of the sites are protonated).
- the lipid particle can show a more gradual transition from non-protonated to protonated as the pH is varied.
- two or more lipids may be selected based on other considerations. For example, if one lipid by itself is highly effective but moderately toxic, it might be combined with a lipid that is less effective but non-toxic. In some cases, the combination can remain highly effective but have a greatly reduced toxicity, even where it might be predicted that the combination would be only moderately effective and only slightly less toxic.
- the selection may be guided by a measured value of an experimentally determinable characteristic, e.g., a characteristic tha can be assigned a numerical value from the results of an experiment.
- Experimentally determinable characteristics can include a measure of safety, a measure of efficacy, a measure of interaction with a predetermined biomolecule, or pK a .
- a measure of safety might include a survival rate, an LD 50 , or a level of a biomarker (such as a serum biomarker) associated with tissue damage (e.g., liver enzymes for liver; CPK for muscle; ionic balance for kidney).
- a measure of efficacy can be any measurement that indicates whether a therapeutic agent is producing an effect; particularly, whether and/or to what degree it is producing a desired effect, such as treating, preventing, ameliorating, or otherwise improving a disease, disorder, or other clinical condition.
- the measure of efficacy can be an indirect measure; for example, if a therapeutic agent is intended to produce a particular effect at a cellular level, measurements of that effect on cell cultures can be a measure of efficacy.
- a measure of interaction with predetermined biomolecules can include a K ⁇ j for binding to a particular protein or a measure of the character, degree or extent of interaction with other lipids, including cellular substructures such as cell membranes, endosomal membranes, nuclear membranes, and the like.
- the cationic lipids can be selected on the basis of mechanism of action, e.g., whether, under what conditions, or to what extent the lipids interact with predetermined biomolecules.
- a first cationic lipid can be chosen, in part, because it is associated with an ApoE- dependent mechanism; a second cationic lipid can be chosen, in part, because it is associated with an ApoE-independent mechanism.
- a lipid particle can also include a mixture of the cationic lipids described in, e.g., WO 2009/086558, and provisional U.S. Application No. 61/104,219, filed October 9, 2008 (each of which is incorporated by reference in its entirety), and ester analogs thereof.
- a lipid particle can include a mixture of a lipid, for example, Lipid A, described in PCT/US 10/22614, filed January 29, 2010 and a lipid, for example, the lipid of formula V or formula VI, described in US Provisional Application 61/175,770, filed May 5, 2009.
- targeting moieties that are specific to a cell type or tissue.
- Targeting of lipid particles using a variety of targeting moieties such as ligands, cell surface receptors, glycoproteins, vitamins (e.g., riboflavin) and monoclonal antibodies, has been previously described (see, e.g., U.S. Patent Nos. 4,957,773 and 4,603,044, each of which is incorporated by reference in its entirety).
- the targeting moieties can comprise the entire protein or fragments thereof.
- Targeting mechanisms generally require that the targeting agents be positioned on the surface of the lipid particle in such a manner that the target moiety is available for interaction with the target, for example, a cell surface receptor.
- lipid particles i.e., liposomes
- hydrophilic polymer chains such as polyethylene glycol (PEG) chains
- a ligand such as an antibody, for targeting the lipid particle is linked to the polar head group of lipids forming the lipid particle.
- the targeting ligand is attached to the distal ends of the PEG chains forming the hydrophilic polymer coating (Klibanov, et al., Journal of Liposome Research 2: 321-334 (1992); Kirpotin et al, FEBS Letters 388: 115-118 (1996)).
- Standard methods for coupling the target agents can be used.
- phosphatidylethanolamine which can be activated for attachment of target agents, or derivatized lipophilic compounds, such as lipid-derivatized bleomycin, can be used.
- Antibody-targeted liposomes can be constructed using, for instance, liposomes that incorporate protein A (see, Renneisen, et al., J. Bio.
- targeting moieties can also include other proteins, specific to cellular components, including antigens associated with neoplasms or tumors. Proteins used as targeting moieties can be attached to the liposomes via covalent bonds (see, Heath, Covalent Attachment of Proteins to Liposomes, 149 Methods in Enzymology 111-119 (Academic Press, Inc. 1987)). Other targeting methods include the biotin-avidin system.
- the lipid particle includes a mixture of a cationic lipid and a fusion-promoting lipid.
- the lipid particle can further include a neutral lipid, a sterol, a PEG- modified lipid, or a combination of these.
- the lipid particle can include a cationic lipid, a fusion-promoting lipid (e.g., DPPC), and a neutral lipid, but no sterol or PEG-modified lipid.
- the lipid particle can include a cationic lipid, a fusion-promoting lipid, and a neutral lipid, but no sterol or PEG-modified lipid.
- the lipid particle can include a cationic lipid, a fusion- promoting lipid, and a PEG-modified lipid, but no sterol or neutral lipid.
- the lipid particle can include a cationic lipid, a fusion-promoting lipid, a sterol, and a neutral lipid, but no PEG- modified lipid.
- the lipid particle can include a cationic lipid, a fusion-promoting lipid, a sterol, and a PEG-modified lipid, but no neutral lipid.
- the lipid particle can include a cationic lipid, a fusion-promoting lipid, a neutral lipid, and a PEG-modified lipid, but no sterol.
- the lipid particle can include a cationic lipid, a fusion-promoting lipid, a sterol, neutral lipid, and a PEG-modified lipid.
- the lipid particle can include a cationic lipid, a fusion
- the lipid particle comprises a mixture of a cationic lipid, a fusion-promoting lipid, neutral lipids (other than a cationic lipid), a sterol (e.g., cholesterol) and a PEG-modified lipid (e.g., a PEG-DMG or PEG-DMA).
- the lipid mixture consists of or consists essentially of a cationic lipid, a fusion-promoting lipid, a neutral lipid, cholesterol, and a PEG-modified lipid.
- the lipid particle includes the above lipid mixture in molar ratios of about 20-70% cationic lipid: 0.1-50% fusion promoting lipid: 5-45% neutral lipid: 20-55% cholesterol: 0.5-15% PEG-modified lipid.
- the fusion-promoting lipid can be present in a molar ratio of 0.1-50%, 0.5-50%, 1- 50%, 5%-45%, 10%-40%, or 15%-35%.
- the fusion-promoting lipid can be present in a molar ratio of 0.1-50%, 0.5-50%, 1-50%, 5%-45%, 10%-40%, or 15%-35%.
- the fusion-promoting lipid can be present in a molar ratio of 0.1-50%, 10- 50%, 20-50%, or 30-50%. In some embodiments, the fusion-promoting lipid can be present in a molar ratio of 0.1-50%, 0.5-45%, 1-40%, l%-35%, l%-30%, or l%-20%.
- the lipid particle consists of or consists essentially of the above lipid mixture in molar ratios of about 20-70% cationic lipid: 0.1-50% fusion promoting lipid: 5-45% neutral lipid: 20-55% cholesterol: 0.5-15% PEG-modified lipid.
- the molar lipid ratio is approximately 40/10/40/10, 35/15/40/10 or 52/13/30/5; this mixture is further combined with a fusion-promoting lipid in a molar ratio of 0.1-50%, 0.1-50%, 0.5-50%, 1-50%, 5%-45%, 10%-40%, or 15%-35%; in other words, when a 40/10/40/10 mixture of lipid/DSPC/Chol/PEG-DMG or PEG-DMA is combined with a fusion- promoting peptide in a molar ratio of 50%, the resulting lipid particles can have a total molar ratio of (mol% cationic lipid/DSPC/Chol/PEG-DMG or PEG-DMA/fusion-promoting peptide) 20/5/20/5/50.
- the neutral lipid, DSPC in these compositions is replaced with
- compositions that include a lipid particle and an active agent, where the active agent is associated with the lipid particle, are provided.
- the active agent is a therapeutic agent.
- the active agent is encapsulated within an aqueous interior of the lipid particle.
- the active agent is present within one or more lipid layers of the lipid particle.
- the active agent is bound to the exterior or interior lipid surface of a lipid particle.
- “Fully encapsulated” as used herein indicates that the nucleic acid in the particles is not significantly degraded after exposure to serum or a nuclease assay that would significantly degrade free nucleic acids. In a fully encapsulated system, preferably less than 25% of particle nucleic acid is degraded in a treatment that would normally degrade 100% of free nucleic acid, more preferably less than 10% and most preferably less than 5% of the particle nucleic acid is degraded. Alternatively, full encapsulation may be determined by an Oligreen ® assay.
- Oligreen ® is an ultra-sensitive fluorescent nucleic acid stain for quantitating oligonucleotides and single- stranded DNA in solution (available from Invitrogen Corporation, Carlsbad, CA). Fully encapsulated also suggests that the particles are serum stable, that is, that they do not rapidly decompose into their component parts upon in vivo administration.
- the lipid particles comprise a cationic lipid of the present invention, a neutral lipid, a sterol and a PEG-modified lipid.
- the lipid particles include from about 25% to about 75% on a molar basis of cationic lipid, e.g., from about 35 to about 65%, from about 45 to about 65%, about 60%, about 57.5%, about 57.1%, about 50% or about 40% on a molar basis.
- the lipid particles include from about 0% to about 15% on a molar basis of the neutral lipid, e.g., from about 3 to about 12%, from about 5 to about 10%, about 15%, about 10%, about 7.5%, about 7.1% or about 0% on a molar basis.
- the neutral lipid is DPPC. In one embodiment, the neutral lipid is DSPC.
- the formulation includes from about 5% to about 50% on a molar basis of the sterol, e.g., about 15 to about 45%, about 20 to about 40%, about 48%, about 40%, about 38.5%, about 35%, about 34.4%, about 31.5% or about 31% on a molar basis.
- the sterol e.g., about 15 to about 45%, about 20 to about 40%, about 48%, about 40%, about 38.5%, about 35%, about 34.4%, about 31.5% or about 31% on a molar basis.
- the sterol is cholesterol
- the lipid particles include from about 0.1% to about 20% on a molar basis of the PEG-modified lipid, e.g., about 0.5 to about 10%, about 0.5 to about 5%, about 10%, about 5%, about 3.5%, about 1.5%, about 0.5%, or about 0.3% on a molar basis.
- the PEG-modified lipid is PEG- DMG.
- the PEG-modified lipid is PEG-c-DMA.
- the lipid particles include 25-75% of cationic lipid, 0.5-15% of the neutral lipid, 5-50% of the sterol, and 0.5- 20% of the PEG-modified lipid on a molar basis.
- the lipid particles include 35-65% of cationic lipid, 3-12% of the neutral lipid, 15-45% of the sterol, and 0.5- 10% of the PEG-modified lipid on a molar basis. In one embodiment, the lipid particles include 45-65% of cationic lipid, 5-10% of the neutral lipid, 25-40% of the sterol, and 0.5- 5% of the PEG-modified lipid on a molar basis. In one embodiment, the PEG modified lipid comprises a PEG molecule of an average molecular weight of 2,000 Da. In one embodiment, the PEG modified lipid is PEG-distyryl glycerol (PEG-DSG).
- PEG-DSG PEG-distyryl glycerol
- the ratio of lipid: siRNA is at least about 0.5: 1, at least about 1: 1, at least about 2: 1, at least about 3: 1, at least about 4: 1, at least about 5: 1, at least about 6: 1, at least about 7: 1, at least about 11: 1 or at least about 33: 1. In one embodiment, the ratio of lipid: siRNA ratio is between about 1: 1 to about 35: 1, about 3: 1 to about 15: 1, about 4: 1 to about 15: 1, or about 5: 1 to about 13: 1. In one embodiment, the ratio of lipid:siRNA ratio is between about 0.5: 1 to about 12: 1.
- the lipid particles are nanoparticles.
- the lipid particles have a mean diameter size of from about 50 nm to about 300 nm, such as from about 50 nm to about 250 nm, for example, from about 50 nm to about 200 nm.
- a lipid particle containing a cationic lipid of any of the embodiments described herein has an in vivo half life (t ⁇ ) (e.g., in the liver, spleen or plasma) of less than about 3 hours, such as less than about 2.5 hours, less than about 2 hours, less than about 1.5 hours, less than about 1 hour, less than about 0.5 hour or less than about 0.25 hours.
- t ⁇ in vivo half life
- a lipid particle containing a cationic lipid of any of the embodiments described herein has an in vivo half life (t ⁇ ) (e.g., in the liver, spleen or plasma) of less than about 10 % (e.g., less than about 7.5%, less than about 5%, less than about 2.5%) of that for the same cationic lipid without the biodegrable group or groups.
- t ⁇ in vivo half life
- lipid particles and compositions described herein can further include an
- apolipoprotein or “lipoprotein” refers to
- apolipoproteins known to those of skill in the art and variants and fragments thereof and to apolipoprotein agonists, analogues or fragments thereof described below.
- Suitable apolipoproteins include, but are not limited to, ApoA-I, ApoA-II, ApoA-IV, ApoA-V and ApoE, and active polymorphic forms, isoforms, variants and mutants as well as fragments or truncated forms thereof.
- the apolipoprotein is a thiol containing apolipoprotein.
- Thiol containing apolipoprotein refers to an apolipoprotein, variant, fragment or isoform that contains at least one cysteine residue.
- thiol containing apolipoproteins are ApoA-I Milano (ApoA-I M ) and ApoA-I Paris (ApoA-I P ) which contain one cysteine residue (Jia et al., 2002, Biochem. Biophys. Res. Comm. 297: 206-13; Bielicki and Oda, 2002, Biochemistry 41: 2089-96).
- ApoA-II, ApoE2 and ApoE3 are also thiol containing apolipoproteins. Isolated ApoE and/or active fragments and polypeptide analogues thereof, including recombinantly produced forms thereof, are described in U.S. Pat. Nos.
- the apolipoprotein can be in its mature form, in its
- the apolipoprotein can be a fragment, variant or isoform of the apolipoprotein.
- fragment refers to any apolipoprotein having an amino acid sequence shorter than that of a native apolipoprotein and which fragment retains the activity of native apolipoprotein, including lipid binding properties.
- variant is meant substitutions or alterations in the amino acid sequences of the apolipoprotein, which substitutions or alterations, e.g., additions and deletions of amino acid residues, do not abolish the activity of native apolipoprotein, including lipid binding properties.
- a variant can comprise a protein or peptide having a substantially identical amino acid sequence to a native apolipoprotein provided herein in which one or more amino acid residues have been conservatively substituted with chemically similar amino acids.
- conservative substitutions include the substitution of at least one hydrophobic residue such as isoleucine, valine, leucine or methionine for another.
- the substitution of at least one hydrophilic residue such as, for example, between arginine and lysine, between glutamine and asparagine, and between glycine and serine (see U.S. Pat. Nos. 6,004,925, 6,037,323 and 6,046,166) are conservative substitutions.
- isoform refers to a protein having the same, greater or partial function and similar, identical or partial sequence, and may or may not be the product of the same gene and usually tissue specific (see Weisgraber 1990, J. Lipid Res. 31(8): 1503-11 ; Hixson and Powers 1991, J. Lipid Res. 32(9): 1529-35; Lackner et al., 1985, J. Biol. Chem. 260(2):703-6; Hoeg et al., 1986, J. Biol. Chem. 261(9):3911-4; Gordon et al., 1984, J. Biol. Chem. 259(l):468-74; Powell et al., 1987, Cell 50(6):831-40; Aviram et al., 1998, Arterioscler. Thromb. Vase. Biol. 18(10): 1617-24;
- the lipid particles and compositions described herein include a chimeric construction of an apolipoprotein.
- a chimeric construction of an apolipoprotein can be comprised of an apolipoprotein domain with high lipid binding capacity associated with an apolipoprotein domain containing ischemia reperfusion protective properties.
- a chimeric construction of an apolipoprotein can be a construction that includes separate regions within an apolipoprotein (i.e., homologous construction) or a chimeric construction can be a construction that includes separate regions between different apolipoproteins (i.e., heterologous constructions).
- Compositions comprising a chimeric construction can also include segments that are apolipoprotein variants or segments designed to have a specific character (e.g., lipid binding, receptor binding, enzymatic, enzyme activating, antioxidant or reduction-oxidation property) (see Weisgraber 1990, J. Lipid Res. 31(8): 1503-11 ; Hixson and Powers 1991, J. Lipid Res. 32(9): 1529-35; Lackner et al., 1985, J. Biol. Chem. 260(2):703-6; Hoeg et al, 1986, J. Biol. Chem. 261(9):3911-4; Gordon et al., 1984, J. Biol. Chem. 259(l):468-74; Powell et al., 1987, Cell 50(6):831-40; Aviram et al., 1998, Arterioscler. Thromb. Vase. Biol. 18(10): 1617-24;
- a specific character e.g., lipid
- Apolipoproteins utilized also include recombinant, synthetic, semi-synthetic or purified apolipoproteins. Methods for obtaining apolipoproteins or equivalents thereof are well-known in the art. For example, apolipoproteins can be separated from plasma or natural products by, for example, density gradient centrifugation or immunoaffinity chromatography, or produced synthetically, semi- synthetically or using recombinant DNA techniques known to those of the art (see, e.g., Mulugeta et al., 1998, J. Chromatogr. 798(1-2): 83-90; Chung et al., 1980, J. Lipid Res.
- Apolipoproteins further include apolipoprotein agonists such as peptides and peptide analogues that mimic the activity of ApoA-I, ApoA-I Milano (APOA-I M ), ApoA-I Paris
- apolipoprotein can be any of those described in U.S. Pat. Nos. 6,004,925, 6,037,323, 6,046,166, and 5,840,688, the contents of which are incorporated herein by reference in their entireties.
- Apolipoprotein agonist peptides or peptide analogues can be synthesized or manufactured using any technique for peptide synthesis known in the art including, e.g., the techniques described in U.S. Pat. Nos. 6,004,925, 6,037,323 and 6,046,166.
- the peptides may be prepared using the solid-phase synthetic technique initially described by Merrifield (1963, J. Am. Chem. Soc. 85:2149-2154).
- Other peptide synthesis techniques may be found in Bodanszky et al., Peptide Synthesis, John Wiley & Sons, 2d Ed., (1976) and other references readily available to those skilled in the art.
- peptides for use in different peptide syntheses are described in the above-mentioned texts as well as in McOmie, Protective Groups in Organic Chemistry, Plenum Press, New York, N.Y. (1973).
- the peptides might also be prepared by chemical or enzymatic cleavage from larger portions of, for example, apolipoprotein A-I.
- the apolipoprotein can be a mixture of apolipoproteins.
- the apolipoprotein can be a homogeneous mixture, that is, a single type of apolipoprotein.
- the apolipoprotein can be a heterogeneous mixture of apolipoproteins, that is, a mixture of two or more different apolipoproteins.
- Embodiments of heterogenous mixtures of apolipoproteins can comprise, for example, a mixture of an
- a heterogenous mixture can comprise, for example, a mixture of ApoA-I and ApoA-I Milano.
- a heterogeneous mixture can comprise, for example, a mixture of ApoA-I Milano and ApoA-I Paris. Suitable mixtures for use in the methods and compositions descreibed herein will be apparent to one of skill in the art.
- the apolipoprotein is obtained from natural sources, it can be obtained from a plant or animal source. If the apolipoprotein is obtained from an animal source, the apolipoprotein can be from any species. In certain embodiments, the apolipoprotien can be obtained from an animal source. In certain embodiments, the apolipoprotein can be obtained from a human source. In preferred embodiments, the apolipoprotein is derived from the same species as the individual to which the apolipoprotein is administered.
- the lipid particles and compositions described herein may further contain a sterol component of the lipid mixture. When present, the sterol can be any of those sterols
- the sterol is cholesterol.
- lipid particles and compositions described herein may further include an anionic lipid.
- Anionic lipids suitable for use in lipid particles include, but are not limited to,
- N-dodecanoyl phosphatidylethanoloamine N-succinyl phosphatidylethanolamine, N-glutaryl phosphatidylethanolamine, lysylphosphatidylglycerol, and other anionic modifying groups joined to neutral lipids.
- amphipathic lipids are also included in the lipid particles and compositions described herein.
- “Amphipathic lipids” refer to any suitable material, wherein the hydrophobic portion of the lipid material orients into a hydrophobic phase, while the hydrophilic portion orients toward the aqueous phase.
- Such compounds include, but are not limited to, phospholipids, aminolipids, and sphingolipids.
- Representative phospholipids include sphingomyelin, phosphatidylcholine, phosphatidylethanolamine, phosphatidylserine, phosphatidylinositol, phosphatidic acid, palmitoyloleoyl phosphatdylcholine,
- lysophosphatidylcholine lysophosphatidylethanolamine, dipalmitoylphosphatidylcholine, dioleoylphosphatidylcholine, distearoylphosphatidylcholine, or dilinoleoylphosphatidylcholine.
- Other phosphorus-lacking compounds such as sphingolipids, glycosphingolipid families, diacylglycerols, and ⁇ -acyloxyacids, can also be used. Additionally, such amphipathic lipids can be readily mixed with other lipids, such as triglycerides and sterols.
- lipid particles and compositions described herein are programmable fusion lipids or fusion-promoting lipids.
- Such lipid particles have little tendency to fuse with cell membranes and deliver their payload until a given signal event occurs. This allows the lipid particle to distribute more evenly after injection into an organism or disease site before it starts fusing with cells.
- the signal event can be, for example, a change in pH, temperature, ionic environment, or time.
- the fusion promoting-lipids can be, for example, compounds of formula (I) as described above.
- the signal event can be a change in pH, for example, such as the difference in pH between an extracelluar environment and an intracellular environment, or between an intracellular environment and an endosomal environment.
- a fusion delaying or "cloaking" component such as an ATTA-lipid conjugate or a PEG-lipid conjugate, can simply exchange out of the lipid particle membrane over time.
- a fusion delaying or "cloaking” component such as an ATTA-lipid conjugate or a PEG-lipid conjugate
- cloaking agent such as an ATTA-lipid conjugate or a PEG-lipid conjugate
- the lipid particles and compositions described herein may further include one or more active agents (e.g., therapeutic agents).
- Active agents include any molecule or compound capable of exerting a desired effect on a cell, tissue, organ, or subject. Such effects may be biological, physiological, or cosmetic, for example.
- the lipid particles and compositions can be used to deliver any of a variety of active agents.
- the active agent can be a nucleic acid, peptide, polypeptide (e.g., an antibody), cytokines, growth factors, apoptotic factors,
- Suitable therapeutic agents also include anti-inflammatory compounds,
- lipid particles of the present invention can also deliver aptamers.
- the therapeutic agent is an oncology drug, which may also be referred to as an anti-tumor drug, an anti-cancer drug, a tumor drug, an antineoplastic agent, or the like.
- oncology drugs include, but are not limited to, adriamycin, alkeran, allopurinol, altretamine, amifostine, anastrozole, araC, arsenic trioxide, azathioprine, bexarotene, biCNU, bleomycin, busulfan intravenous, busulfan oral, capecitabine
- the active agent is a nucleic acid, such as a siRNA.
- the active agent can be a nucleic acid encoded with a product of interest, including but not limited to, RNA, antisense oligonucleotide, an antagomir, a DNA, a plasmid, a ribosomal RNA (rRNA), a micro RNA (miRNA) (e.g., a miRNA which is single stranded and 17-25 nucleotides in length), transfer RNA (tRNA), a small interfering RNA (siRNA), small nuclear RNA (snRNA), antigens, fragments thereof, proteins, peptides, vaccines and small molecules or mixtures thereof.
- RNA antisense oligonucleotide
- an antagomir e.g., a DNA, a plasmid, a ribosomal RNA (rRNA), a micro RNA (miRNA) (e.g., a mi
- the nucleic acid is an oligonucleotide (e.g., 15-50 nucleotides in length (or 15-30 or 20-30 nucleotides in length)).
- An siRNA can have, for instance, a duplex region that is 16-30 nucleotides long.
- the nucleic acid is an immuno stimulatory oligonucleotide, decoy oligonucleotide, supermir, miRNA mimic, or miRNA inhibitor.
- a supermir refers to a single stranded, double stranded or partially double stranded oligomer or polymer of ribonucleic acid (RNA) or deoxyribonucleic acid (DNA) or both or modifications thereof, which has a nucleotide sequence that is substantially identical to an miRNA and that is antisense with respect to its target.
- miRNA mimics represent a class of molecules that can be used to imitate the gene silencing ability of one or more miRNAs.
- miRNA mimic refers to synthetic non-coding RNAs (i.e. the miRNA is not obtained by purification from a source of the endogenous miRNA) that are capable of entering the RNAi pathway and regulating gene expression.
- the nucleic acid that is present in a lipid-nucleic acid particle can be in any form.
- the nucleic acid can, for example, be single-stranded DNA or RNA, or double- stranded DNA or RNA, or DNA-RNA hybrids.
- Non-limiting examples of double- stranded RNA include siRNA.
- Single- stranded nucleic acids include, e.g., antisense oligonucleotides, ribozymes, microRNA, and triplex-forming oligonucleotides.
- the lipid particles of the present invention can also deliver nucleic acids which are conjugated to one or more ligands.
- the lipid particles may be formulated as a pharmaceutical composition, e.g., which further comprises a pharmaceutically acceptable diluent, excipient, or carrier, such as physiological saline or phosphate buffer, selected in accordance with the route of administration and standard pharmaceutical practice.
- a pharmaceutically acceptable diluent, excipient, or carrier such as physiological saline or phosphate buffer, selected in accordance with the route of administration and standard pharmaceutical practice.
- compositions for the delivery of siRNA molecules are described. These compositions are effective in down-regulating the protein levels and/or mRNA levels of target proteins.
- the activity of these compositions can be influenced by the presence of cationic lipids and the molar ratio of cationic lipid in the formulation.
- compositions comprising the lipid-nucleic acid particles are prepared according to standard techniques and further comprise a
- compositions comprising saline or other salt containing carriers the carrier is preferably added following lipid particle formation.
- the compositions can be diluted into
- compositions may be sterilized by conventional, well known sterilization techniques.
- the aqueous solutions can then be packaged for use or filtered under aseptic conditions and lyophilized, the lyophilized preparation being combined with a sterile aqueous solution prior to administration.
- the compositions may contain pharmaceutically acceptable auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, tonicity adjusting agents and the like, for example, sodium acetate, sodium lactate, sodium chloride, potassium chloride, calcium chloride, etc.
- the lipidic suspension may include lipid-protective agents which protect lipids against free-radical and lipid-peroxidative damages on storage.
- Lipophilic free -radical quenchers such as a-tocopherol and water-soluble iron-specific chelators, such as ferrioxamine, are suitable.
- the concentration of lipid particle or lipid-nucleic acid particle in the pharmaceutical formulations can vary widely, i.e., from less than about 0.01%, usually at or at least about 0.05-5% to as much as 10 to 30% by weight and will be selected primarily by fluid volumes, viscosities, etc., in accordance with the particular mode of administration selected.
- the concentration may be increased to lower the fluid load associated with treatment. This may be particularly desirable in patients having atherosclerosis-associated congestive heart failure or severe hypertension.
- complexes composed of irritating lipids may be diluted to low concentrations to lessen inflammation at the site of administration.
- the nucleic acid will have an attached label and will be used for diagnosis (by indicating the presence of complementary nucleic acid).
- the amount of complexes administered will depend upon the particular label used, the disease state being diagnosed and the judgement of the clinician but will generally be between about 0.01 and about 50 mg per kilogram of body weight, preferably between about 0.1 and about 5 mg/kg of body weight.
- the lipid-therapeutic agent ⁇ e.g., nucleic acid particles may include polyethylene glycol (PEG) -modified phospholipids, PEG-ceramide, or ganglioside GMi-modified lipids or other lipids effective to prevent or limit aggregation. Addition of such components does not merely prevent complex aggregation. Rather, it may also provide a means for increasing circulation lifetime and increasing the delivery of the lipid-nucleic acid composition to the target tissues.
- PEG polyethylene glycol
- PEG-ceramide polyethylene glycol
- ganglioside GMi-modified lipids or other lipids effective to prevent or limit aggregation additive of such components does not merely prevent complex aggregation. Rather, it may also provide a means for increasing circulation lifetime and increasing the delivery of the lipid-nucleic acid composition to the target tissues.
- Lipid-therapeutic agent compositions can also be provided in kit form.
- the kit will typically be comprised of a container that is compartmentalized for holding the various elements of the kit.
- the kit will contain the particles or pharmaceutical compositions, preferably in dehydrated or concentrated form, with instructions for their rehydration or dilution and administration.
- the particles comprise the active agent, while in other embodiments, they do not.
- WO 2010/054406, WO 2010/054401, WO 2010/054405, and WO 2010/054384, WO 2010/042877, WO 2010/129709, WO 2009/086558, and WO 2008/042973 are described in, for example, International Publication Nos. WO 2010/054406, WO 2010/054401, WO 2010/054405, and WO 2010/054384, WO 2010/042877, WO 2010/129709, WO 2009/086558, and WO 2008/042973, each of which is incorporated by reference in its entirety.
- lipid particles and pharmaceutical compositions containing the lipid particles are also described in, for example, US Publication Nos. 2004/0142025, 2006/0051405 and 2007/0042031, each of which is incorporated by reference in its entirety.
- methods of preparing lipid particles, including those associated with a therapeutic agent, e.g. , a nucleic acid are described.
- a mixture of lipids is combined with a buffered aqueous solution of nucleic acid to produce an intermediate mixture containing nucleic acid encapsulated in lipid particles.
- the encapsulated nucleic acids are present in a nucleic acid/lipid ratio of about 3 wt to about 25 wt , preferably 5 to 15 wt .
- the intermediate mixture may optionally be sized to obtain lipid-encapsulated nucleic acid particles wherein the lipid portions are unilamellar vesicles, preferably having a diameter of 30 to 150 nm, more preferably about 40 to 90 nm.
- the pH is then raised to neutralize at least a portion of the surface charges on the lipid-nucleic acid particles, thus providing an at least partially surface-neutralized lipid-encapsulated nucleic acid composition.
- a solution of one or more lipids (including a cationic lipid of any of the embodiments described herein) in an organic solution (e.g., ethanol) is prepared.
- an organic solution e.g., ethanol
- active (therapeutic) agents such as, for example an siRNA molecule or a 1 : 1 molar mixture of two siRNA molecules
- siRNA lipid particles have an average particle size of about 80-90 nm.
- the dispersion may be filtered through 0.45/2 micron filters, concentrated and diafiltered by tangential flow filtration.
- concentration of the resulting product is adjusted to about 2 mg/mL.
- the product is sterile filtered, aseptically filtered and packaged.
- these cationic lipids are amino lipids that are charged at a pH below the pK a of the amino group and substantially neutral at a pH above the pK a .
- These cationic lipids are termed titratable cationic lipids and can be used in the formulations using a two-step process. First, lipid vesicles can be formed at the lower pH with titratable cationic lipids and other vesicle components in the presence of nucleic acids. In this manner, the vesicles will encapsulate and entrap the nucleic acids.
- the surface charge of the newly formed vesicles can be neutralized by increasing the pH of the medium to a level above the pK a of the titratable cationic lipids present, i.e., to physiological pH or higher.
- Particularly advantageous aspects of this process include both the facile removal of any surface adsorbed nucleic acid and a resultant nucleic acid delivery vehicle which has a neutral surface. Liposomes or lipid particles having a neutral surface are expected to avoid rapid clearance from circulation and to avoid certain toxicities which are associated with cationic liposome preparations. Additional details concerning these uses of such titratable cationic lipids in the formulation of nucleic acid-lipid particles are provided in U.S. Patent 6,287,591 and U.S. Patent 6,858,225, incorporated herein by reference.
- the vesicles formed in this manner provide formulations of uniform vesicle size with high content of nucleic acids. Additionally, the vesicles have may have a size range of from about 30 to about 150 nm, more preferably about 30 to about 90 nm.
- nucleic acid encapsulation is a result of electrostatic interaction at low pH.
- acidic pH e.g. pH 4.0
- the vesicle surface is charged and binds a portion of the nucleic acids through electrostatic interactions.
- a more neutral buffer e.g. pH 7.5
- the surface of the lipid particle or liposome is neutralized, allowing any external nucleic acid to be removed.
- a mixture of lipids is combined with a buffered aqueous solution of nucleic acid to produce an intermediate mixture containing nucleic acid encapsulated in lipid particles, e.g. , wherein the encapsulated nucleic acids are present in a nucleic acid/lipid ratio of about 10 wt% to about 20 wt%.
- the intermediate mixture may optionally be sized to obtain lipid-encapsulated nucleic acid particles wherein the lipid portions are unilamellar vesicles, preferably having a diameter of 30 to 150 nm, more preferably about 40 to 90 nm.
- the pH is then raised to neutralize at least a portion of the surface charges on the lipid-nucleic acid particles, thus providing an at least partially surface-neutralized lipid-encapsulated nucleic acid composition.
- the mixture of lipids includes at least two lipid components: a first lipid component that is selected from among lipids which have a pK a such that the lipid is cationic at pH below the pK a and neutral at pH above the pK a , and a second lipid component that is selected from among lipids that prevent particle aggregation during lipid-nucleic acid particle formation.
- the amino lipid is a cationic lipid.
- the mixture of lipids is typically a solution of lipids in an organic solvent. This mixture of lipids can then be dried to form a thin film or lyophilized to form a powder before being hydrated with an aqueous buffer to form liposomes.
- the lipid mixture can be solubilized in a water miscible alcohol, such as ethanol, and this ethanolic solution added to an aqueous buffer resulting in spontaneous liposome formation.
- the alcohol is used in the form in which it is commercially available.
- ethanol can be used as absolute ethanol (100%), or as 95% ethanol, the remainder being water. This method is described in more detail in U.S. Patent 5,976,567).
- the mixture of lipids is a mixture of cationic lipids, neutral lipids (other than a cationic lipid), a sterol (e.g., cholesterol) and a PEG-modified lipid (e.g., a PEG-DMG or PEG-DMA) in an alcohol solvent.
- the lipid mixture consists essentially of a cationic lipid, a neutral lipid, cholesterol and a PEG-modified lipid in alcohol, more preferably ethanol.
- the first solution consists of the above lipid mixture in molar ratios of about 20-70% cationic lipid: 5-45% neutral lipid:20-55% cholesterol:0.5-15% PEG-modified lipid.
- the first solution consists essentially of a mixture of cationic lipids chosen from lipids described in Tables 1-5, DSPC, Choi and PEG-DMG or PEG-DMA, more preferably in a molar ratio of about 20-60% cationic lipid: 5-25% DSPC:25-55% Chol:0.5- 15% PEG-DMG or PEG-DMA.
- the molar lipid ratio is approximately 40/10/40/10 (mol% cationic lipid/DSPC/Chol/PEG-DMG or PEG-DMA), 35/15/40/10 (mol% cationic
- the neutral lipid in these compositions is replaced with POPC, DPPC, DOPE or SM.
- the lipid mixture is combined with a buffered aqueous solution that may contain the nucleic acids.
- the buffered aqueous solution of is typically a solution in which the buffer has a pH of less than the pK a of the protonatable lipid in the lipid mixture.
- suitable buffers include citrate, phosphate, acetate, and MES.
- a particularly preferred buffer is citrate buffer.
- Preferred buffers will be in the range of 1- 1000 mM of the anion, depending on the chemistry of the nucleic acid being encapsulated, and optimization of buffer concentration may be significant to achieving high loading levels (see, e.g., U.S. Patent 6,287,591 and U.S.
- Patent 6,858,225 each of which is incorporated by reference in its entirety).
- pure water acidified to pH 5-6 with chloride, sulfate or the like may be useful.
- the amount of nucleic acid in buffer can vary, but will typically be from about 0.01 mg/mL to about 200 mg/mL, more preferably from about 0.5 mg/mL to about 50 mg/mL.
- the mixture of lipids and the buffered aqueous solution of therapeutic nucleic acids is combined to provide an intermediate mixture.
- the intermediate mixture is typically a mixture of lipid particles having encapsulated nucleic acids. Additionally, the intermediate mixture may also contain some portion of nucleic acids which are attached to the surface of the lipid particles (liposomes or lipid vesicles) due to the ionic attraction of the negatively-charged nucleic acids and positively-charged lipids on the lipid particle surface (the amino lipids or other lipid making up the protonatable first lipid component are positively charged in a buffer having a pH of less than the pK a of the protonatable group on the lipid).
- the mixture of lipids is an alcohol solution of lipids and the volumes of each of the solutions is adjusted so that upon combination, the resulting alcohol content is from about 20% by volume to about 45% by volume.
- the method of combining the mixtures can include any of a variety of processes, often depending upon the scale of formulation produced. For example, when the total volume is about 10-20 mL or less, the solutions can be combined in a test tube and stirred together using a vortex mixer. Large-scale processes can be carried out in suitable production scale glassware.
- the lipid-encapsulated therapeutic agent e.g. , nucleic acid
- the compositions provided herein will be sized to a mean diameter of from about 70 to about 200 nm, more preferably about 90 to about 130 nm.
- Several techniques are available for sizing liposomes to a desired size. One sizing method is described in U.S. Pat. No. 4,737,323, incorporated herein by reference.
- Sonicating a liposome suspension either by bath or probe sonication produces a progressive size reduction down to small unilamellar vesicles (SUVs) less than about 0.05 microns in size.
- Homogenization is another method which relies on shearing energy to fragment large liposomes into smaller ones.
- multilamellar vesicles are recirculated through a standard emulsion homogenizer until selected liposome sizes, typically between about 0.1 and 0.5 microns, are observed.
- the particle size distribution can be monitored by conventional laser-beam particle size determination.
- extrusion is used to obtain a uniform vesicle size.
- Extrusion of liposome compositions through a small-pore polycarbonate membrane or an asymmetric ceramic membrane results in a relatively well-defined size distribution.
- the suspension is cycled through the membrane one or more times until the desired liposome complex size distribution is achieved.
- the liposomes may be extruded through successively smaller-pore membranes, to achieve a gradual reduction in liposome size.
- the lipid-nucleic acid compositions which are formed can be used without any sizing.
- methods further comprise a step of neutralizing at least some of the surface charges on the lipid portions of the lipid-nucleic acid compositions.
- a step of neutralizing at least some of the surface charges on the lipid portions of the lipid-nucleic acid compositions By at least partially neutralizing the surface charges, unencapsulated nucleic acid is freed from the lipid particle surface and can be removed from the composition using conventional techniques.
- unencapsulated and surface adsorbed nucleic acids are removed from the resulting compositions through exchange of buffer solutions.
- buffer solutions For example, replacement of a citrate buffer (pH about 4.0, used for forming the compositions) with a HEPES -buffered saline (HBS pH about 7.5) solution, results in the neutralization of liposome surface and nucleic acid release from the surface.
- HBS pH about 7.5 HEPES -buffered saline
- the released nucleic acid can then be removed via chromatography using standard methods, and then switched into a buffer with a pH above the pK a of the lipid used.
- the lipid vesicles can be formed by hydration in an aqueous buffer and sized using any of the methods described above prior to addition of the nucleic acid.
- the aqueous buffer should be of a pH below the pK a of the amino lipid.
- a solution of the nucleic acids can then be added to these sized, preformed vesicles.
- the mixture should contain an alcohol, such as ethanol. In the case of ethanol, it should be present at a concentration of about 20% (w/w) to about 45% (w/w).
- nucleic acid encapsulation process it may be necessary to warm the mixture of pre-formed vesicles and nucleic acid in the aqueous buffer-ethanol mixture to a temperature of about 25° C to about 50° C depending on the composition of the lipid vesicles and the nature of the nucleic acid. It will be apparent to one of ordinary skill in the art that optimization of the encapsulation process to achieve a desired level of nucleic acid in the lipid vesicles will require manipulation of variable such as ethanol concentration and temperature. Examples of suitable conditions for nucleic acid encapsulation are provided in the Examples. Once the nucleic acids are encapsulated within the prefromed vesicles, the external pH can be increased to at least partially neutralize the surface charge. Unencapsulated and surface adsorbed nucleic acids can then be removed as described above.
- lipid particles and compositions described herein may be used for a variety of purposes, including the delivery of associated or encapsulated therapeutic agents to cells, both in vitro and in vivo. Accordingly, methods of treating diseases or disorders in a subject in need thereof can include contacting the subject with a lipid particle associated with a suitable therapeutic agent.
- the lipid particles are particularly useful for the delivery of nucleic acids, including, e.g., siRNA molecules and plasmids. Therefore, the lipid particles and compositions may be used to modulate the expression of target genes and proteins both in vitro and in vivo by contacting cells with a lipid particle associated with a nucleic acid that reduces target gene expression (e.g., an siRNA) or a nucleic acid that may be used to increase expression of a desired protein ⁇ e.g., a plasmid encoding the desired protein).
- a nucleic acid that reduces target gene expression e.g., an siRNA
- a nucleic acid that may be used to increase expression of a desired protein ⁇ e.g., a plasmid encoding the desired protein.
- the lipid particles may be used to deliver a therapeutic agent to a cell, in vitro or in vivo.
- the therapeutic agent is a nucleic acid, which is delivered to a cell using nucleic acid-lipid particles. While the following description of various methods of using the lipid particles and related pharmaceutical compositions are exemplified by description related to nucleic acid-lipid particles, it is understood that these methods and compositions may be readily adapted for the delivery of any therapeutic agent for the treatment of any disease or disorder that would benefit from such treatment.
- nucleic acids for introduction into cells are siRNA, immune- stimulating
- oligonucleotides oligonucleotides, plasmids, antisense and ribozymes. These methods may be carried out by contacting the particles or compositions with the cells for a period of time sufficient for intracellular delivery to occur.
- compositions can be adsorbed to almost any cell type.
- the nucleic acid-lipid particles can either be endocytosed by a portion of the cells, exchange lipids with cell membranes, or fuse with the cells. Transfer or incorporation of the nucleic acid portion of the complex can take place via any one of these pathways. Without intending to be limited, it is believed that in the case of particles taken up into the cell by endocytosis the particles then interact with the endosomal membrane, resulting in destabilization of the endosomal membrane, possibly by the formation of non-bilayer phases, resulting in introduction of the encapsulated nucleic acid into the cell cytoplasm.
- the liposome membrane is integrated into the cell membrane and the contents of the liposome combine with the intracellular fluid.
- Contact between the cells and the lipid-nucleic acid compositions when carried out in vitro, will take place in a biologically compatible medium.
- concentration of compositions can vary widely depending on the particular application, but is generally between about 1 ⁇ and about 10 mmol.
- treatment of the cells with the lipid-nucleic acid compositions will generally be carried out at physiological temperatures (about 37 °C) for periods of time from about 1 to 24 hours, preferably from about 2 to 8 hours.
- the delivery of nucleic acids can be to any cell grown in culture, whether of plant or animal origin, vertebrate or invertebrate, and of any tissue or type.
- the cells will be animal cells, more preferably mammalian cells, and most preferably human cells.
- a lipid-nucleic acid particle suspension is added to 60-80% confluent plated cells having a cell density of from about 10 3 to about 105 cells/mL, more preferably about 2 x 10 4 cells/mL.
- the concentration of the suspension added to the cells is preferably of from about 0.01 to 20 ⁇ g/mL, more preferably about 1 ⁇ g/mL.
- the lipid particles can be may be used to deliver a nucleic acid to a cell or cell line (for example, a tumor cell line).
- a cell or cell line for example, a tumor cell line.
- cell lines include: HELA (ATCC Cat N: CCL-2), KB (ATCC Cat N: CCL-17), HEP3B (ATCC Cat N: HB-8064), SKOV-3 (ATCC Cat N: HTB-77), HCT-116 (ATCC Cat N: CCL-247), HT-29 (ATCC Cat N: HTB-38), PC-3 (ATCC Cat N: CRL-1435), A549 (ATCC Cat N: CCL-185), MDA-MB-231 (ATCC Cat N: HTB-26).
- Typical applications include using well known procedures to provide intracellular delivery of siRNA to knock down or silence specific cellular targets.
- Alternatively applications include delivery of DNA or mRNA sequences that code for therapeutically useful polypeptides.
- therapy is provided for genetic diseases by supplying deficient or absent gene products (i.e., for Duchenne's dystrophy, see Kunkel, et ah, Brit. Med. Bull. 45(3):630-643 (1989), and for cystic fibrosis, see Goodfellow, Nature 341: 102-103 (1989)).
- Other uses for the compositions include introduction of antisense oligonucleotides in cells (see, Bennett, et ah, Mol. Pharm. 41: 1023-1033 (1992)).
- compositions can also be used for deliver of nucleic acids to cells in vivo, using methods which are known to those of skill in the art.
- delivery of DNA or mRNA sequences Zhu, et al., Science 261:209-211 (1993), incorporated herein by reference, describes the intravenous delivery of cytomegalovirus (CM V) -chloramphenicol acetyltransferase (CAT) expression plasmid using DOTMA-DOPE complexes.
- CM V cytomegalovirus
- CAT chloramphenicol acetyltransferase
- Hyde et al., Nature 362:250-256 (1993), incorporated herein by reference, describes the delivery of the cystic fibrosis transmembrane conductance regulator (CFTR) gene to epithelia of the airway and to alveoli in the lung of mice, using liposomes.
- CTR cystic fibrosis transmembrane conductance regulator
- compositions can be used in the treatment of infectious diseases.
- compositions are preferably administered parenterally, i.e., intraarticularly, intravenously, intraperitoneally, subcutaneously, or
- the pharmaceutical compositions are administered intravenously or intraperitoneally by a bolus injection.
- a bolus injection see Stadler, et ah, U.S. Patent No. 5,286,634, which is incorporated herein by reference.
- Intracellular nucleic acid delivery has also been discussed in Straubringer, et ah, Methods in Enzymology, Academic Press, New York. 101:512-527 (1983); Mannino, et al, Biotechniques 6:682-690 (1988);
- the pharmaceutical preparations may be contacted with the target tissue by direct application of the preparation to the tissue.
- the application may be made by topical, "open” or “closed” procedures.
- topical it is meant the direct application of the
- Open procedures are those procedures which include incising the skin of a patient and directly visualizing the underlying tissue to which the pharmaceutical preparations are applied. This is generally accomplished by a surgical procedure, such as a thoracotomy to access the lungs, abdominal laparotomy to access abdominal viscera, or other direct surgical approach to the target tissue.
- “Closed” procedures are invasive procedures in which the internal target tissues are not directly visualized, but accessed via inserting instruments through small wounds in the skin.
- the preparations may be
- the preparations may be administered to the peritoneum by needle lavage.
- the pharmaceutical preparations may be administered to the meninges or spinal cord by infusion during a lumbar puncture followed by appropriate positioning of the patient as commonly practiced for spinal anesthesia or metrazamide imaging of the spinal cord.
- the preparations may be administered through endoscopic devices.
- lipid-nucleic acid compositions can also be administered in an aerosol inhaled into the lungs (see, Brigham, et ah, Am. J. Sci. 298(4):278-281 (1989)) or by direct injection at the site of disease (Culver, Human Gene Therapy, Mary Ann Liebert, Inc., Publishers, New York. pp.70-71 (1994)).
- Dosages for the lipid-therapeutic agent particles will depend on the ratio of therapeutic agent to lipid and the administrating physician's opinion based on age, weight, and condition of the patient.
- a method of modulating the expression of a target polynucleotide or polypeptide generally comprise contacting a cell with a lipid particle that is associated with a nucleic acid capable of modulating the expression of a target polynucleotide or polypeptide.
- modulating refers to altering the expression of a target polynucleotide or polypeptide. In different embodiments, modulating can mean increasing or enhancing, or it can mean decreasing or reducing.
- Methods of measuring the level of expression of a target polynucleotide or polypeptide include, e.g., methods employing reverse transcription-polymerase chain reaction (RT-PCR) and immunohistochemical techniques.
- RT-PCR reverse transcription-polymerase chain reaction
- the level of expression of a target polynucleotide or polypeptide is increased or reduced by at least 10%, 20%, 30%, 40%, 50%, or greater than 50% as compared to an appropriate control value.
- the nucleic acid may be an expression vector that includes a polynucleotide that encodes the desired polypeptide.
- the nucleic acid may be, e.g. , an antisense oligonucleotide, siRNA, or microRNA that comprises a polynucleotide sequence that specifically hybridizes to a polnucleotide that encodes the target polypeptide, thereby disrupting expression of the target polynucleotide or polypeptide.
- the nucleic acid may be a plasmid that expresses such an antisense
- oligonucletoide siRNA, or microRNA.
- the therapeutic agent is selected from an siRNA, a microRNA, an antisense oligonucleotide, and a plasmid capable of expressing an siRNA, a microRNA, or an antisense oligonucleotide, and wherein the siRNA, microRNA, or antisense RNA comprises a polynucleotide that specifically binds to a polynucleotide that encodes the polypeptide, or a complement thereof, such that the expression of the polypeptide is reduced.
- the nucleic acid is a plasmid that encodes the polypeptide or a functional variant or fragment thereof, such that expression of the polypeptide or the functional variant or fragment thereof is increased.
- a method of treating a disease or disorder characterized by overexpression of a polypeptide in a subject includes providing to the subject a pharmaceutical composition, wherein the therapeutic agent is selected from an siRNA, a microRNA, an antisense oligonucleotide, and a plasmid capable of expressing an siRNA, a microRNA, or an antisense oligonucleotide, and wherein the siRNA, microRNA, or antisense RNA comprises a polynucleotide that specifically binds to a polynucleotide that encodes the polypeptide, or a complement thereof.
- the therapeutic agent is selected from an siRNA, a microRNA, an antisense oligonucleotide, and a plasmid capable of expressing an siRNA, a microRNA, or an antisense oligonucleotide
- the siRNA, microRNA, or antisense RNA comprises a polynucleotide that specifically binds to a polynucle
- the pharmaceutical composition comprises a lipid particle that consists of or consists essentially of a mixture of cationic lipids chosen from lipids described in
- Tables 1-5, DSPC, Choi and PEG-DMG or PEG-DMA e.g. , in a molar ratio of about 20-60% cationic lipid: 5-25% DSPC:25-55% Chol:0.5- 15% PEG-DMG or PEG-DMA, wherein the lipid particle is assocated with the therapeutic nucleic acid.
- the molar lipid ratio is approximately 40/10/40/10 (mol% cationic lipid/DSPC/Chol/PEG-DMG or PEG-DMA),
- the neutral lipid in these compositions is replaced with POPC, DPPC, DOPE or SM.
- a method of treating a disease or disorder characterized by underexpression of a polypeptide in a subject includes providing to the subject a
- composition wherein the therapeutic agent is a plasmid that encodes the polypeptide or a functional variant or fragment thereof.
- a method of inducing an immune response in a subject can include providing to the subject the pharmaceutical composition, wherein the therapeutic agent is an immuno stimulatory oligonucleotide.
- the immune response is a humoral or mucosal immune response.
- the pharmaceutical composition is provided to the subject in combination with a vaccine or antigen.
- vaccines can include a lipid particle, which comprises an immuno stimulatory oligonucleotide, and is also associated with an antigen to which an immune response is desired.
- the antigen is a tumor antigen or is associated with an infective agent, such as, e.g. , a virus, bacteria, or parasiste.
- antigens include, but are not limited to, polypeptide antigens and DNA antigens.
- suitable antigens include, but are not limited to, polypeptide antigens and DNA antigens.
- Specific examples of antigens are Hepatitis A, Hepatitis B, small pox, polio, anthrax, influenza, typhus, tetanus, measles, rotavirus, diphtheria, pertussis, tuberculosis, and rubella antigens.
- the antigen is a Hepatitis B recombinant antigen.
- the antigen is a Hepatitis A recombinant antigen.
- the antigen is a tumor antigen. Examples of such tumor-associated antigens are MUC-1, EBV antigen and antigens associated with Burkitt's lymphoma.
- the antigen is a
- Tumor-associated antigens suitable for use include both mutated and non-mutated molecules that may be indicative of single tumor type, shared among several types of tumors, and/or exclusively expressed or overexpressed in tumor cells in comparison with normal cells.
- tumor-specific patterns of expression of carbohydrates, gangliosides, glycolipids and mucins have also been documented.
- Exemplary tumor-associated antigens for use in the subject cancer vaccines include protein products of oncogenes, tumor suppressor genes and other genes with mutations or rearrangements unique to tumor cells, reactivated embryonic gene products, oncofetal antigens, tissue-specific (but not tumor-specific) differentiation antigens, growth factor receptors, cell surface carbohydrate residues, foreign viral proteins and a number of other self proteins.
- tumor-associated antigens include, e.g. , mutated antigens such as the protein products of the Ras p21 protooncogenes, tumor suppressor p53 and BCR-abl oncogenes, as well as CDK4, MUM1, Caspase 8, and Beta catenin; overexpressed antigens such as galectin 4, galectin 9, carbonic anhydrase, Aldolase A, PRAME, Her2/neu, ErbB-2 and KSA, oncofetal antigens such as alpha fetoprotein (AFP), human chorionic gonadotropin (hCG); self antigens such as carcinoembryonic antigen (CEA) and melanocyte differentiation antigens such as Mart 1/Melan A, gplOO, gp75, Tyrosinase, TRPl and TRP2; prostate associated antigens such as PSA, PAP, PSMA, PSM-P1 and PSM-P2; reactivated embryonic gene products
- Pathogens include, but are not limited to, infectious agents, e.g. , viruses, that infect mammals, and more particularly humans.
- infectious virus include, but are not limited to: Retroviridae (e.g. , human immunodeficiency viruses, such as HIV-1 (also referred to as HTLV-III, LAV or HTLV-III/LAV, or HIV-III; and other isolates, such as HIV-LP;
- Picornaviridae e.g., polio viruses, hepatitis A virus; enteroviruses, human Coxsackie viruses, rhinoviruses, echoviruses); Calciviridae (e.g., strains that cause gastroenteritis); Togaviridae (e.g., equine encephalitis viruses, rubella viruses); Flaviridae (e.g., dengue viruses, encephalitis viruses, yellow fever viruses); Coronoviridae (e.g., coronaviruses); Rhabdoviradae (e.g., vesicular stomatitis viruses, rabies viruses); Coronaviridae (e.g. , coronaviruses); Rhabdoviridae (e.g., vesicular stomatitis viruses, rabies viruses); Filoviridae (e.g., ebola viruses);
- Paramyxoviridae e.g., parainfluenza viruses, mumps virus, measles virus, respiratory syncytial virus
- Orthomyxoviridae e.g., influenza viruses
- Bungaviridae e.g., Hantaan viruses, bunga viruses, phleboviruses and Nairo viruses
- Arena viridae hemorrhagic fever viruses
- Reoviridae e.g., reoviruses, orbiviurses and rotaviruses
- Birnaviridae Hepadnaviridae (Hepatitis B virus); Parvovirida (parvoviruses)
- Papovaviridae papilloma viruses, polyoma viruses
- Adenoviridae most adenoviruses
- Herpesviridae herpes simplex virus (HSV) 1 and 2, varicella zoster virus, cytomegalovirus (CMV
- gram negative and gram positive bacteria serve as antigens in vertebrate animals.
- Such gram positive bacteria include, but are not limited to Pasteurella species, Staphylococci species, and Streptococcus species.
- Gram negative bacteria include, but are not limited to, Escherichia coli, Pseudomonas species, and Salmonella species.
- infectious bacteria include but are not limited to: Helicobacterpyloris, Borelia burgdorferi, Legionella pneumophilia, Mycobacteria sps (e.g., M. tuberculosis, M. avium, M. intracellulare, M. kansaii, M.
- Streptococcus pyogenes Group A Streptococcus
- Streptococcus agalactiae Group B Streptococcus
- Streptococcus viridans group
- Streptococcusfaecalis Streptococcus bovis
- Streptococcus anaerobic sps.
- Streptococcus pneumoniae pathogenic Campylobacter sp., Enterococcus sp., Haemophilus infuenzae, Bacillus antracis, corynebacterium diphtheriae, corynebacterium sp., Erysipelothrix rhusiopathiae, Clostridium perfringers, Clostridium tet
- infectious parasites include Plasmodium such as Plasmodium falciparum, Plasmodium malariae,
- Plasmodium ovale, and Plasmodium vivax Other infectious organisms (i.e., protists) include Toxoplasma gondii.
- the formulations can be used to silence or modulate a target gene such as but not limited to FVII, Eg5, PCSK9, TPX2, apoB, SAA, TTR, RSV, PDGF beta gene, Erb-B gene, Src gene, CRK gene, GRB2 gene, RAS gene, MEKK gene, JNK gene, RAF gene, Erkl/2 gene, PCNA(p21) gene, MYB gene, JUN gene, FOS gene, BCL-2 gene, Cyclin D gene, VEGF gene, EGFR gene, Cyclin A gene, Cyclin E gene, WNT- 1 gene, beta-catenin gene, c-MET gene, PKC gene, NFKB gene, STAT3 gene, survivin gene, Her2/Neu gene, SORTl gene, XBP1 gene, topoisomerase I gene, topoisomerase II alpha gene, p73 gene, p21(WAFl/CIPl) gene, p27 (KIP 1) gene, PPM
- encephalitis virus replication Murray Valley encephalitis virus gene, a gene that is required for Murray Valley encephalitis virus replication, dengue virus gene, a gene that is required for dengue virus gene replication, Simian Virus 40 gene, a gene that is required for Simian Virus 40 replication, Human T Cell Lymphotropic Virus gene, a gene that is required for Human T Cell Lymphotropic Virus replication, Moloney-Murine Leukemia Virus gene, a gene that is required for Moloney-Murine Leukemia Virus replication, encephalomyocarditis virus gene, a gene that is required for encephalomyocarditis virus replication, measles virus gene, a gene that is required for measles virus replication, Vericella zoster virus gene, a gene that is required for Vericella zoster virus replication, adenovirus gene, a gene that is required for adenovirus replication, yellow fever virus gene, a gene that is required for yellow fever virus replication,
- the present invention relates to a method of delivering a nucleic acid molecule comprising administering a nucleic lipid particle comprising the nucleic acid molecule and a cationic lipid, the cationic lipid having
- each hydrophobic tail comprising a C 14 or greater aliphatic group attached to the central atom, where the aliphatic group is (a) interrupted by a biodegradable group such that there is a chain of at least four carbon atoms between the biodegradable group and the central carbon atom, or (b) includes a biodegradable group at the terminal end of the hydrophobic tail, such that the cationic lipid remains intact until delivery of the nucleic acid molecule after which cleavage of the
- hydrophobic tail occurs in vivo.
- cationic lipid inlcudes those lipids having one or two fatty acid or fatty aliphatic chains and an amino head group (including an alkylamino or dialkylamino group) that may be protonated to form a cationic lipid at physiological pH.
- amino head group including an alkylamino or dialkylamino group
- a cationic lipid is referred to as an "amino lipid.”
- a subject or patient in whom administration of the complex is an effective therapeutic regimen for a disease or disorder is preferably a human, but can be any animal, including a laboratory animal in the context of a clinical trial or screening or activity experiment.
- the methods, compounds and compositions of the present invention are particularly suited to administration to any animal, particularly a mammal, and including, but by no means limited to, humans, domestic animals, such as feline or canine subjects, farm animals, such as but not limited to bovine, equine, caprine, ovine, and porcine subjects, wild animals (whether in the wild or in a zoological garden), research animals, such as mice, rats, rabbits, goats, sheep, pigs, dogs, and cats, avian species, such as chickens, turkeys, and songbirds, i.e., for veterinary medical use.
- M 1 is defined as - C(0)NH- (i.e., -(R)i-C(0)-NH-(R) m -)
- the compound where M 1 is -NHC(O)- i.e., -(R) ; - NHC(0)-(R) m -
- M 1 is -NHC(O)- (i.e., -(R) ; - NHC(0)-(R) m -) is also contemplated unless otherwise specified.
- biodegradable group referes to a group that include one or more bonds that may undergo bond breaking reactions in a biological environment, e.g., in an organism, organ, tissue, cell, or organelle.
- the biodegradable group may be metabolizable by the body of a mammal, such as a human (e.g., by hydrolysis).
- Some groups that contain a biodegradable bond include, for example, but are not limited to esters, dithiols, and oximes.
- an "aliphatic” group is a non-aromatic group in which carbon atoms are linked into chains, and is either saturated or unsaturated.
- alkyl and alkylene refer to a straight or branched chain saturated hydrocarbon moiety.
- the alkyl group is a straight chain saturated hydrocarbon.
- the "alkyl” or “alkylene” group contains from 1 to 24 carbon atoms.
- Representative saturated straight chain alkyl groups include methyl, ethyl, n-propyl, n-butyl, n-pentyl, and n-hexyl.
- Representative saturated branched alkyl groups include isopropyl, sec-butyl, isobutyl, ie/t-butyl, and isopentyl.
- alkenyl refers to a straight or branched chain hydrocarbon moiety having one or more carbon-carbon double bonds. In one embodiment, the alkenyl group contains 1, 2, or 3 double bonds and is otherwise saturated. Unless otherwise specified, the "alkenyl” group contains from 2 to 24 carbon atoms. Alkenyl groups include both cis and trans isomers.
- Representative straight chain and branched alkenyl groups include ethylenyl, propylenyl,
- alkynyl refers to a straight or branched chain hydrocarbon moiety having one or more carbon-carbon triple bonds. Unless otherwise specified, the "alkynyl” group contains from 2 to 24 carbon atoms. Representative straight chain and branched alkynyl groups include acetylenyl, propynyl, 1-butynyl, 2-butynyl, 1-pentynyl, 2-pentynyl, and 3-methyl-l-butynyl.
- acyl refers to a carbonyl group substituted with hydrogen, alkyl, partially saturated or fully saturated cycloalkyl, partially saturated or fully saturated heterocycle, aryl, or heteroaryl.
- acyl groups include groups such as (C 1 -C2o)alkanoyl (e.g., formyl, acetyl, propionyl, butyryl, valeryl, caproyl, and t-butylacetyl), (C3-C2o)cycloalkylcarbonyl (e.g., cyclopropylcarbonyl, cyclobutylcarbonyl, cyclopentylcarbonyl, and cyclohexylcarbonyl), heterocyclic carbonyl (e.g., pyrrolidinylcarbonyl, pyrrolid-2-one-5-carbonyl,
- C 1 -C2o)alkanoyl e.g., formyl, acetyl, propionyl
- aryl refers to an aromatic monocyclic, bicyclic, or tricyclic hydrocarbon ring system. Unless otherwise specified, the “aryl” group contains from 6 to 14 carbon atoms.
- aryl moieties include, but are not limited to, phenyl, naphthyl, anthracenyl, and pyrenyl.
- cycloalkyl and cycloalkylene refer to a saturated monocyclic or bicyclic hydrocarbon moiety such as cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl. Unless otherwise specified, the "cycloalkyl” or “cycloalkylene” group contains from 3 to 10 carbon atoms.
- cycloalkylalkyl refers to a cycloalkyl group bound to an alkyl group, where the alkyl group is bound to the rest of the molecule.
- heterocycle refers to a non-aromatic 5- to 8-membered monocyclic, or 7- to 12-membered bicyclic, or 11- to 14-membered tricyclic ring system which is either saturated or unsaturated, and which contains from 1 to 3 heteroatoms if monocyclic, 1-6 heteroatoms if bicyclic, or 1-9 heteroatoms if tricyclic, independently selected from nitrogen, oxygen and sulfur, and wherein the nitrogen and sulfur heteroatoms may be optionally oxidized, and the nitrogen heteroatom may be optionally quaternized.
- the heterocycle may be a cycloalkoxy group.
- heterocycle may be attached to the rest of the molecule via any heteroatom or carbon atom in the heterocycle.
- Heterocycles include, but are not limited to, morpholinyl, pyrrolidinonyl, pyrrolidinyl, piperidinyl, piperizynyl, hydantoinyl, valerolactamyl, oxiranyl, oxetanyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydropyridinyl,
- heteroaryl refers to an aromatic 5-8 membered monocyclic, 7-12 membered bicyclic, or 11-14 membered tricyclic ring system having 1-3 heteroatoms if monocyclic, 1-6 heteroatoms if bicyclic, or 1-9 heteroatoms if tricyclic, where the heteroatoms are selected from O, N, or S (e.g., carbon atoms and 1-3, 1-6, or 1-9 heteroatoms of N, O, or S if monocyclic, bicyclic, or tricyclic, respectively).
- the heteroaryl groups herein described may also contain fused rings that share a common carbon-carbon bond.
- substituted refers to the replacement of one or more hydrogen radicals in a given structure with the radical of a specified substituent including, but not limited to: halo, alkyl, alkenyl, alkynyl, aryl, heterocyclyl, thiol, alkylthio, oxo, thioxy, arylthio, alkylthioalkyl, arylthioalkyl, alkylsulfonyl, alkylsulfonylalkyl, arylsulfonylalkyl, alkoxy, aryloxy, aralkoxy, aminocarbonyl, alkylaminocarbonyl, arylaminocarbonyl,
- halogen refers to fluoro, chloro, bromo and iodo.
- alkylamine and “dialkylamine” refer to -NH(alkyl) and -N(alkyl) 2 radicals respectively.
- alkylphosphate refers to -0-P(Q')(Q")-0-R, wherein Q' and Q" are each independently O, S, N(R) 2 , optionally substituted alkyl or alkoxy; and R is optionally substituted alkyl, ⁇ -aminoalkyl or ro-(substituted)aminoalkyl.
- alkylphosphorothioate refers to an alkylphosphate wherein at least one of Q' or Q" is S.
- alkylphosphonate refers to an alkylphosphate wherein at least one of Q' or Q" is alkyl.
- hydroxyalkyl refers to -O-alkyl radical.
- alkylheterocycle refers to an alkyl where at least one methylene has been replaced by a heterocycle.
- ⁇ -aminoalkyl refers to -alkyl-NH 2 radical.
- ro-(substituted)aminoalkyl refers to an ⁇ -aminoalkyl wherein at least one of the H on N has been replaced with alkyl.
- ⁇ -phosphoalkyl refers to -alkyl-0-P(Q')(Q")-0-R, wherein Q' and Q" are each independently O or S and R optionally substituted alkyl.
- co-thiophosphoalkyl refers to ⁇ -phosphoalkyl wherein at least one of Q' or Q" is S.
- DSPC distearoylphosphatidylcholine
- DPPC l,2-Dipalmitoyl-sn-glycero-3-phosphocholine
- POPC 1- palmitoyl-2-oleoyl-sn-phosphatidylcholine
- DOPE l,2-dileoyl-sn-3- phosphoethanolamine
- PEG-DMG generally refers to 1,2-dimyristoyl-sn-glycerol-methoxy polyethylene glycol (e.g., PEG 2000)
- TBDPSC1 tert-Butylchlorodiphenylsilane
- DMAP DMAP:
- HMPA hexamethylphosphoramide
- EDC l-ethyl-3-(3- dimethylaminopropyl) carbodiimide
- DIPEA diisopropylethylamine
- DCM dichloromethane
- TEA triethylamine
- TBAF tetrabutylammonium fluoride
- the methods may require the use of protecting groups.
- protecting group methodology is well known to those skilled in the art (see, for example, Protective Groups in Organic Synthesis, Green, T.W. et. al., Wiley-Interscience, New York City, 1999). Briefly, protecting groups are any group that reduces or eliminates unwanted reactivity of a functional group. A protecting group can be added to a functional group to mask its reactivity during certain reactions and then removed to reveal the original functional group. In some embodiments an "alcohol protecting group" is used. An “alcohol protecting group” is any group which decreases or eliminates unwanted reactivity of an alcohol functional group. Protecting groups can be added and removed using techniques well known in the art.
- the compounds may be prepared by at least one of the techniques described herein or known organic synthesis techniques.
- the di-aldehyde 4 can be synthesized as shown in Scheme 3, using l-bromo-9-decene.
- Di-aldehyde containing a head group 27 can be useful for the synthesis of terminal ester- substituted lipids using, e.g., a Wittig reaction. Ozonolysis can afford di-aldehyde 4 and 27.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- Epidemiology (AREA)
- Biochemistry (AREA)
- Immunology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Microbiology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Biophysics (AREA)
- Virology (AREA)
- Mycology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Inorganic Chemistry (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Pain & Pain Management (AREA)
- AIDS & HIV (AREA)
- Tropical Medicine & Parasitology (AREA)
- Rheumatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SG2012087441A SG186085A1 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| NZ605079A NZ605079A (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| ES11726579.3T ES2634087T3 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the administration of active agents |
| MX2012014079A MX341332B (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents. |
| KR1020197009607A KR20190039347A (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| JP2013513394A JP5893611B2 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for delivery of active agents |
| KR1020137000092A KR101967411B1 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| IL300109A IL300109A (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| CN201180037927.7A CN103096875B (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipid for delivery of active agent |
| DK11726579.3T DK2575764T3 (en) | 2010-06-03 | 2011-06-03 | BIODEGRADABLE LIPIDS FOR THE ACTIVATION OF ACTIVE AGENTS |
| CA2800401A CA2800401C (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| EA201291100A EA039892B1 (en) | 2011-05-23 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| EP11726579.3A EP2575764B1 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| AU2011261247A AU2011261247B2 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
| IL223207A IL223207B (en) | 2010-06-03 | 2012-11-22 | Biodegradable lipids for the delivery of active agents |
| IL259334A IL259334B2 (en) | 2010-06-03 | 2018-05-14 | Biodegradable lipids for the delivery of active agents |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US35114610P | 2010-06-03 | 2010-06-03 | |
| US61/351,146 | 2010-06-03 | ||
| US201161489197P | 2011-05-23 | 2011-05-23 | |
| US61/489,197 | 2011-05-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2011153493A2 true WO2011153493A2 (en) | 2011-12-08 |
| WO2011153493A3 WO2011153493A3 (en) | 2012-01-19 |
Family
ID=45067325
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2011/039164 WO2011153493A2 (en) | 2010-06-03 | 2011-06-03 | Biodegradable lipids for the delivery of active agents |
Country Status (14)
| Country | Link |
|---|---|
| US (3) | US9012498B2 (en) |
| EP (2) | EP2575764B1 (en) |
| JP (3) | JP5893611B2 (en) |
| KR (2) | KR101967411B1 (en) |
| CN (1) | CN103096875B (en) |
| AU (6) | AU2011261247B2 (en) |
| CA (1) | CA2800401C (en) |
| DK (1) | DK2575764T3 (en) |
| ES (1) | ES2634087T3 (en) |
| IL (3) | IL300109A (en) |
| MX (1) | MX341332B (en) |
| NZ (1) | NZ605079A (en) |
| SG (1) | SG186085A1 (en) |
| WO (1) | WO2011153493A2 (en) |
Cited By (101)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013086354A1 (en) | 2011-12-07 | 2013-06-13 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2013086322A1 (en) * | 2011-12-07 | 2013-06-13 | Alnylam Pharmaceuticals, Inc. | Branched alkyl and cycloalkyl terminated biodegradable lipids for the delivery of active agents |
| WO2013116126A1 (en) * | 2012-02-01 | 2013-08-08 | Merck Sharp & Dohme Corp. | Novel low molecular weight, biodegradable cationic lipids for oligonucleotide delivery |
| EP2629802A2 (en) * | 2010-10-21 | 2013-08-28 | Merck Sharp & Dohme Corp. | Novel low molecular weight cationic lipids for oligonucleotide delivery |
| WO2014008334A1 (en) | 2012-07-06 | 2014-01-09 | Alnylam Pharmaceuticals, Inc. | Stable non-aggregating nucleic acid lipid particle formulations |
| WO2014089239A1 (en) | 2012-12-07 | 2014-06-12 | Alnylam Pharmaceuticals, Inc. | Improved nucleic acid lipid particle formulations |
| WO2014089486A1 (en) | 2012-12-07 | 2014-06-12 | Shire Human Genetic Therapies, Inc. | Lipidic nanoparticles for mrna delivering |
| WO2015095351A1 (en) | 2013-12-19 | 2015-06-25 | Novartis Ag | LEPTIN mRNA COMPOSITIONS AND FORMULATIONS |
| US9365610B2 (en) | 2013-11-18 | 2016-06-14 | Arcturus Therapeutics, Inc. | Asymmetric ionizable cationic lipid for RNA delivery |
| WO2016100716A1 (en) | 2014-12-18 | 2016-06-23 | Vasant Jadhav | Reversirtm compounds |
| WO2017019636A1 (en) * | 2015-07-24 | 2017-02-02 | Insmed Incorporated | Compositions and methods for the treatment of sarcoidosis |
| US9567296B2 (en) | 2013-11-18 | 2017-02-14 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| KR20170039722A (en) | 2014-08-07 | 2017-04-11 | 다케다 야쿠힝 고교 가부시키가이샤 | Cationic lipid |
| JP2017121240A (en) * | 2012-04-02 | 2017-07-13 | モデルナティエックス インコーポレイテッドModernaTX,Inc. | Modified polynucleotides for production of membrane proteins |
| WO2017222016A1 (en) | 2016-06-24 | 2017-12-28 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Cationic lipid |
| WO2018033254A2 (en) | 2016-08-19 | 2018-02-22 | Curevac Ag | Rna for cancer therapy |
| WO2018104538A1 (en) | 2016-12-08 | 2018-06-14 | Curevac Ag | Rna for treatment or prophylaxis of a liver disease |
| WO2018104540A1 (en) | 2016-12-08 | 2018-06-14 | Curevac Ag | Rnas for wound healing |
| WO2018115527A2 (en) | 2016-12-23 | 2018-06-28 | Curevac Ag | Mers coronavirus vaccine |
| WO2018115507A2 (en) | 2016-12-23 | 2018-06-28 | Curevac Ag | Henipavirus vaccine |
| WO2018115525A1 (en) | 2016-12-23 | 2018-06-28 | Curevac Ag | Lassa virus vaccine |
| WO2018167320A1 (en) | 2017-03-17 | 2018-09-20 | Curevac Ag | Rna vaccine and immune checkpoint inhibitors for combined anticancer therapy |
| US10081598B2 (en) | 2014-12-26 | 2018-09-25 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| WO2018172556A1 (en) | 2017-03-24 | 2018-09-27 | Curevac Ag | Nucleic acids encoding crispr-associated proteins and uses thereof |
| WO2018222926A1 (en) | 2017-05-31 | 2018-12-06 | Ultragenyx Pharmaceutical Inc. | Therapeutics for glycogen storage disease type iii |
| WO2018222890A1 (en) | 2017-05-31 | 2018-12-06 | Arcturus Therapeutics, Inc. | Synthesis and structure of high potency rna therapeutics |
| US10166298B2 (en) | 2015-10-28 | 2019-01-01 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| EP3424524A2 (en) | 2017-07-04 | 2019-01-09 | CureVac AG | Cancer rna-vaccine |
| EP3434667A1 (en) | 2012-04-19 | 2019-01-30 | Sirna Therapeutics, Inc. | Novel diester and triester based low molecular weight, biodegradable cationic lipids for oligonucleotide delivery |
| WO2019027055A1 (en) | 2017-08-04 | 2019-02-07 | 協和発酵キリン株式会社 | Nucleic-acid-containing lipid nanoparticles |
| WO2019038332A1 (en) | 2017-08-22 | 2019-02-28 | Curevac Ag | Bunyavirales vaccine |
| US10221127B2 (en) | 2015-06-29 | 2019-03-05 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| WO2019077001A1 (en) | 2017-10-19 | 2019-04-25 | Curevac Ag | Novel artificial nucleic acid molecules |
| WO2019092153A1 (en) | 2017-11-08 | 2019-05-16 | Curevac Ag | Rna sequence adaptation |
| WO2019115635A1 (en) | 2017-12-13 | 2019-06-20 | Curevac Ag | Flavivirus vaccine |
| WO2019131770A1 (en) | 2017-12-27 | 2019-07-04 | 武田薬品工業株式会社 | Nucleic acid-containing lipid nano-particle and use thereof |
| WO2019131580A1 (en) | 2017-12-27 | 2019-07-04 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Cationic lipid |
| US10383952B2 (en) | 2016-12-21 | 2019-08-20 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| WO2019193183A2 (en) | 2018-04-05 | 2019-10-10 | Curevac Ag | Novel yellow fever nucleic acid molecules for vaccination |
| US10501513B2 (en) | 2012-04-02 | 2019-12-10 | Modernatx, Inc. | Modified polynucleotides for the production of oncology-related proteins and peptides |
| US10501512B2 (en) | 2012-04-02 | 2019-12-10 | Modernatx, Inc. | Modified polynucleotides |
| WO2020002525A1 (en) | 2018-06-27 | 2020-01-02 | Curevac Ag | Novel lassa virus rna molecules and compositions for vaccination |
| US10526284B2 (en) | 2016-12-21 | 2020-01-07 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| WO2020072324A1 (en) | 2018-10-01 | 2020-04-09 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2020080475A1 (en) | 2018-10-18 | 2020-04-23 | 武田薬品工業株式会社 | Method for activation/proliferation of t cells |
| WO2020118115A1 (en) | 2018-12-06 | 2020-06-11 | Arcturus Therapeutics, Inc. | Compositions and methods for treating ornithine transcarbamylase deficiency |
| WO2020123300A2 (en) | 2018-12-14 | 2020-06-18 | Eli Lilly And Company | Kras variant mrna molecules |
| WO2020128031A2 (en) | 2018-12-21 | 2020-06-25 | Curevac Ag | Rna for malaria vaccines |
| US10703789B2 (en) | 2012-04-02 | 2020-07-07 | Modernatx, Inc. | Modified polynucleotides for the production of secreted proteins |
| US10723692B2 (en) | 2014-06-25 | 2020-07-28 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| WO2020161342A1 (en) | 2019-02-08 | 2020-08-13 | Curevac Ag | Coding rna administered into the suprachoroidal space in the treatment of ophtalmic diseases |
| WO2020254535A1 (en) | 2019-06-18 | 2020-12-24 | Curevac Ag | Rotavirus mrna vaccine |
| WO2021028439A1 (en) | 2019-08-14 | 2021-02-18 | Curevac Ag | Rna combinations and compositions with decreased immunostimulatory properties |
| WO2021123332A1 (en) | 2019-12-20 | 2021-06-24 | Curevac Ag | Lipid nanoparticles for delivery of nucleic acids |
| WO2021156267A1 (en) | 2020-02-04 | 2021-08-12 | Curevac Ag | Coronavirus vaccine |
| WO2021239880A1 (en) | 2020-05-29 | 2021-12-02 | Curevac Ag | Nucleic acid based combination vaccines |
| WO2022008613A1 (en) | 2020-07-08 | 2022-01-13 | Janssen Sciences Ireland Unlimited Company | Rna replicon vaccines against hbv |
| WO2022023559A1 (en) | 2020-07-31 | 2022-02-03 | Curevac Ag | Nucleic acid encoded antibody mixtures |
| WO2022043551A2 (en) | 2020-08-31 | 2022-03-03 | Curevac Ag | Multivalent nucleic acid based coronavirus vaccines |
| US11357856B2 (en) | 2017-04-13 | 2022-06-14 | Acuitas Therapeutics, Inc. | Lipids for delivery of active agents |
| WO2022137133A1 (en) | 2020-12-22 | 2022-06-30 | Curevac Ag | Rna vaccine against sars-cov-2 variants |
| WO2022135993A2 (en) | 2020-12-22 | 2022-06-30 | Curevac Ag | Pharmaceutical composition comprising lipid-based carriers encapsulating rna for multidose administration |
| WO2022146654A1 (en) | 2020-12-28 | 2022-07-07 | Janssen Pharmaceuticals, Inc. | Transcription activator-like effector nucleases (talens) targeting hbv |
| WO2022162027A2 (en) | 2021-01-27 | 2022-08-04 | Curevac Ag | Method of reducing the immunostimulatory properties of in vitro transcribed rna |
| WO2022168884A1 (en) | 2021-02-04 | 2022-08-11 | 塩野義製薬株式会社 | Cationic lipid |
| US11453639B2 (en) | 2019-01-11 | 2022-09-27 | Acuitas Therapeutics, Inc. | Lipids for lipid nanoparticle delivery of active agents |
| WO2022200575A1 (en) | 2021-03-26 | 2022-09-29 | Glaxosmithkline Biologicals Sa | Immunogenic compositions |
| WO2022207862A2 (en) | 2021-03-31 | 2022-10-06 | Curevac Ag | Syringes containing pharmaceutical compositions comprising rna |
| WO2022215758A1 (en) | 2021-04-09 | 2022-10-13 | 相互薬工株式会社 | Lipid and composition |
| WO2022233880A1 (en) | 2021-05-03 | 2022-11-10 | Curevac Ag | Improved nucleic acid sequence for cell type specific expression |
| US11524932B2 (en) | 2017-08-17 | 2022-12-13 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| US11542225B2 (en) | 2017-08-17 | 2023-01-03 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| WO2023006999A2 (en) | 2021-07-30 | 2023-02-02 | CureVac SE | Mrnas for treatment or prophylaxis of liver diseases |
| WO2023031392A2 (en) | 2021-09-03 | 2023-03-09 | CureVac SE | Novel lipid nanoparticles for delivery of nucleic acids comprising phosphatidylserine |
| WO2023031394A1 (en) | 2021-09-03 | 2023-03-09 | CureVac SE | Novel lipid nanoparticles for delivery of nucleic acids |
| WO2023073228A1 (en) | 2021-10-29 | 2023-05-04 | CureVac SE | Improved circular rna for expressing therapeutic proteins |
| WO2023121975A1 (en) * | 2021-12-20 | 2023-06-29 | Beam Therapeutics Inc. | Ionizable amine lipids and lipid nanoparticles |
| WO2023144193A1 (en) | 2022-01-25 | 2023-08-03 | CureVac SE | Mrnas for treatment of hereditary tyrosinemia type i |
| WO2023144330A1 (en) | 2022-01-28 | 2023-08-03 | CureVac SE | Nucleic acid encoded transcription factor inhibitors |
| EP4227319A1 (en) | 2018-04-17 | 2023-08-16 | CureVac SE | Novel rsv rna molecules and compositions for vaccination |
| US11744887B2 (en) | 2020-03-09 | 2023-09-05 | Arcturus Therapeutics, Inc. | Coronavirus vaccine compositions and methods |
| WO2023218420A1 (en) | 2022-05-13 | 2023-11-16 | Janssen Pharmaceuticals, Inc. | Mrna compositions for inducing latent hiv-1 reversal |
| US11820728B2 (en) | 2017-04-28 | 2023-11-21 | Acuitas Therapeutics, Inc. | Carbonyl lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| WO2023227608A1 (en) | 2022-05-25 | 2023-11-30 | Glaxosmithkline Biologicals Sa | Nucleic acid based vaccine encoding an escherichia coli fimh antigenic polypeptide |
| WO2024026308A2 (en) | 2022-07-29 | 2024-02-01 | Massachusetts Institute Of Technology | COMPOSITIONS AND METHODS FOR INHIBITING EXPRESSION OF THE SIGNAL REGULATORY PROTEIN ALPHA (SIRPα) GENE |
| DE202023106198U1 (en) | 2022-10-28 | 2024-03-21 | CureVac SE | Nucleic acid-based vaccine |
| US11939600B2 (en) | 2017-05-31 | 2024-03-26 | Arcturus Therapeutics, Inc. | Compositions and methods for treating phenylketonuria |
| WO2024068545A1 (en) | 2022-09-26 | 2024-04-04 | Glaxosmithkline Biologicals Sa | Influenza virus vaccines |
| US11976019B2 (en) | 2020-07-16 | 2024-05-07 | Acuitas Therapeutics, Inc. | Cationic lipids for use in lipid nanoparticles |
| GB202404607D0 (en) | 2024-03-29 | 2024-05-15 | Glaxosmithkline Biologicals Sa | RNA formulation |
| WO2024121814A1 (en) | 2022-12-09 | 2024-06-13 | Takeda Pharmaceutical Company Limited | Modified immunomodulators |
| EP4385523A1 (en) | 2022-12-14 | 2024-06-19 | Beijing Jitai Pharmaceutical Technology Co., Ltd. | Lipid-based topical injection formulations |
| WO2024160936A1 (en) | 2023-02-03 | 2024-08-08 | Glaxosmithkline Biologicals Sa | Rna formulation |
| WO2024168010A2 (en) | 2023-02-09 | 2024-08-15 | Alnylam Pharmaceuticals, Inc. | Reversir molecules and methods of use thereof |
| US12065396B2 (en) | 2017-08-17 | 2024-08-20 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| WO2024171052A1 (en) | 2023-02-14 | 2024-08-22 | Glaxosmithkline Biologicals Sa | Analytical method |
| US12070509B2 (en) | 2020-05-01 | 2024-08-27 | Arcturus Therapeutics, Inc. | Nucleic acids and methods of treatment for cystic fibrosis |
| WO2024184500A1 (en) | 2023-03-08 | 2024-09-12 | CureVac SE | Novel lipid nanoparticle formulations for delivery of nucleic acids |
| WO2024200823A1 (en) | 2023-03-30 | 2024-10-03 | Ose Immunotherapeutics | Lipid-based nanoparticle targeted at activated immune cells for the expression of immune cell enhancing molecule and use thereof |
| WO2024200826A1 (en) | 2023-03-30 | 2024-10-03 | Ose Immunotherapeutics | Lipid-based nanoparticle targeted at activated immune cells for the expression of immune cell inhibiting molecule and use thereof |
| WO2024210160A1 (en) | 2023-04-07 | 2024-10-10 | Takeda Pharmaceutical Company Limited | Conjugation complex |
Families Citing this family (103)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009088892A1 (en) * | 2008-01-02 | 2009-07-16 | Alnylam Pharmaceuticals, Inc. | Liver screening method |
| PT3338765T (en) | 2009-12-01 | 2019-03-18 | Translate Bio Inc | Steroid derivative for the delivery of mrna in human genetic diseases |
| CA2800401C (en) * | 2010-06-03 | 2020-09-15 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| AU2012267531B2 (en) | 2011-06-08 | 2017-06-22 | Translate Bio, Inc. | Lipid nanoparticle compositions and methods for mRNA delivery |
| US9255154B2 (en) | 2012-05-08 | 2016-02-09 | Alderbio Holdings, Llc | Anti-PCSK9 antibodies and use thereof |
| EP3536787A1 (en) | 2012-06-08 | 2019-09-11 | Translate Bio, Inc. | Nuclease resistant polynucleotides and uses thereof |
| EA201492055A1 (en) | 2012-06-08 | 2015-11-30 | Шир Хьюман Дженетик Терапис, Инк. | INHALATIVE DELIVERY OF mRNA IN TIGHTNESS CELL TARGETS |
| US10942184B2 (en) | 2012-10-23 | 2021-03-09 | Caris Science, Inc. | Aptamers and uses thereof |
| CA2928520C (en) | 2012-10-23 | 2023-03-14 | Caris Life Sciences Switzerland Holdings, S.A.R.L. | Aptamers and uses thereof |
| EP2935628B1 (en) | 2012-12-19 | 2018-03-21 | Caris Life Sciences Switzerland Holdings GmbH | Compositions and methods for aptamer screening |
| HUE042640T2 (en) | 2013-03-14 | 2019-07-29 | Translate Bio Inc | Cftr mrna compositions and related methods and uses |
| JP6586075B2 (en) | 2013-03-14 | 2019-10-02 | トランスレイト バイオ, インコーポレイテッド | Method for purifying messenger RNA |
| EP3060258A1 (en) | 2013-10-22 | 2016-08-31 | Shire Human Genetic Therapies, Inc. | Mrna therapy for phenylketonuria |
| ES2707966T3 (en) | 2013-10-22 | 2019-04-08 | Translate Bio Inc | MRNA therapy for the deficiency in argininosuccinate synthesis |
| KR20220158867A (en) | 2014-04-25 | 2022-12-01 | 샤이어 휴먼 지네틱 테라피즈 인크. | Methods for purification of messenger rna |
| EP3184506B1 (en) * | 2014-08-18 | 2019-07-24 | NOF Corporation | Cationic lipid for nucleic acid delivery |
| EP3252043B1 (en) * | 2015-01-30 | 2020-09-30 | NOF Corporation | Cationic lipid |
| WO2018081480A1 (en) | 2016-10-26 | 2018-05-03 | Acuitas Therapeutics, Inc. | Lipid nanoparticle formulations |
| WO2017186711A1 (en) | 2016-04-25 | 2017-11-02 | Invivogen | Novel complexes of immunostimulatory compounds, and uses thereof |
| EP3585417B1 (en) | 2017-02-27 | 2023-02-22 | Translate Bio, Inc. | Method of making a codon-optimized cftr mrna |
| WO2018191719A1 (en) | 2017-04-13 | 2018-10-18 | Acuitas Therapeutics, Inc. | Lipid delivery of therapeutic agents to adipose tissue |
| WO2018213476A1 (en) | 2017-05-16 | 2018-11-22 | Translate Bio, Inc. | Treatment of cystic fibrosis by delivery of codon-optimized mrna encoding cftr |
| WO2019036008A1 (en) | 2017-08-16 | 2019-02-21 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| JP6826014B2 (en) | 2017-09-13 | 2021-02-03 | 株式会社東芝 | Compositions and kits containing biodegradable compounds, lipid particles, lipid particles |
| WO2019089828A1 (en) | 2017-10-31 | 2019-05-09 | Acuitas Therapeutics, Inc. | Lamellar lipid nanoparticles |
| CN111372576A (en) | 2017-11-17 | 2020-07-03 | 塞尔利克斯生物私人有限公司 | Compositions and methods for treating ocular disorders |
| CN112533909A (en) * | 2018-05-30 | 2021-03-19 | 川斯勒佰尔公司 | Vitamin cationic lipid |
| EP4331681A3 (en) | 2018-08-21 | 2024-05-22 | Kabushiki Kaisha Toshiba | Method for delivering activators to cells |
| CA3108544A1 (en) | 2018-08-24 | 2020-02-27 | Translate Bio, Inc. | Methods for purification of messenger rna |
| EP3853234A1 (en) | 2018-09-18 | 2021-07-28 | Nikang Therapeutics, Inc. | Fused tricyclic ring derivatives as src homology-2 phosphatase inhibitors |
| WO2020061426A2 (en) | 2018-09-21 | 2020-03-26 | Acuitas Therapeutics, Inc. | Systems and methods for manufacturing lipid nanoparticles and liposomes |
| HRP20240586T1 (en) | 2018-10-09 | 2024-07-19 | The University Of British Columbia | Compositions and systems comprising transfection-competent vesicles free of organic-solvents and detergents and methods related thereto |
| BR112021010853A2 (en) * | 2018-12-05 | 2021-08-31 | Intellia Therapeutics, Inc. | MODIFIED AMINE LIPIDS |
| MX2022001720A (en) | 2019-08-14 | 2022-03-11 | Acuitas Therapeutics Inc | Improved lipid nanoparticles for delivery of nucleic acids. |
| BR112022019769A2 (en) | 2020-03-30 | 2022-12-13 | BioNTech SE | RNA COMPOSITIONS THAT TARGET CLAUDIN-18.2 |
| EP4153223A1 (en) | 2020-05-20 | 2023-03-29 | Flagship Pioneering Innovations VI, LLC | Immunogenic compositions and uses thereof |
| CA3179420A1 (en) | 2020-05-20 | 2021-11-25 | Avak Kahvejian | Coronavirus antigen compositions and their uses |
| CA3182026A1 (en) | 2020-05-29 | 2021-12-02 | Flagship Pioneering Innovations Vi, Llc. | Trem compositions and methods relating thereto |
| MX2022015042A (en) | 2020-05-29 | 2023-03-09 | Flagship Pioneering Innovations Vi Llc | Trem compositions and methods relating thereto. |
| AU2021293197A1 (en) | 2020-06-15 | 2023-02-09 | Research Institute At Nationwide Children's Hospital | Adeno-associated virus vector delivery for muscular dystrophies |
| AU2021336976A1 (en) | 2020-09-03 | 2023-03-23 | Flagship Pioneering Innovations Vi, Llc | Immunogenic compositions and uses thereof |
| US20240175020A1 (en) | 2020-12-23 | 2024-05-30 | Flagship Pioneering Innovations Vi, Llc | Compositions of modified trems and uses thereof |
| CA3206128A1 (en) * | 2021-02-09 | 2022-08-18 | J. Milton Harris | Polyoxazoline-lipid conjugates and lipid nanoparticles and pharmaceutical compositions including same |
| KR20230165276A (en) | 2021-03-31 | 2023-12-05 | 플래그쉽 파이어니어링 이노베이션스 브이, 인크. | Thanotransmission polypeptides and their use in the treatment of cancer |
| CN117510830A (en) * | 2021-04-08 | 2024-02-06 | 厦门赛诺邦格生物科技股份有限公司 | Pegylated lipid and modified liposome thereof, pharmaceutical composition containing liposome and preparation and application of pharmaceutical composition |
| CN115197080A (en) * | 2021-04-08 | 2022-10-18 | 厦门赛诺邦格生物科技股份有限公司 | Polyethylene glycol lipid and liposome modified by same, pharmaceutical composition containing liposome, preparation and application of pharmaceutical composition |
| EP4319803A1 (en) | 2021-04-08 | 2024-02-14 | Vaxthera SAS | Coronavirus vaccine comprising a mosaic protein |
| CN115385820A (en) * | 2021-05-09 | 2022-11-25 | 英维沃生物科技(苏州)有限公司 | Cationic lipids and uses thereof |
| US20240336945A1 (en) | 2021-07-26 | 2024-10-10 | Flagship Pioneering Innovations Vi, Llc | Trem compositions and uses thereof |
| WO2023023055A1 (en) | 2021-08-16 | 2023-02-23 | Renagade Therapeutics Management Inc. | Compositions and methods for optimizing tropism of delivery systems for rna |
| TW202328067A (en) | 2021-09-14 | 2023-07-16 | 美商雷納嘉德醫療管理公司 | Cyclic lipids and methods of use thereof |
| CA3231523A1 (en) | 2021-09-14 | 2023-03-23 | Renagade Therapeutics Management Inc. | Acyclic lipids and methods of use thereof |
| KR20240099185A (en) | 2021-09-17 | 2024-06-28 | 플래그쉽 파이어니어링 이노베이션스 브이아이, 엘엘씨 | Compositions and methods for producing circular polyribonucleotides |
| CA3235625A1 (en) | 2021-10-18 | 2023-04-27 | Flagship Pioneering Innovations Vi, Llc | Compositions and methods for purifying polyribonucleotides |
| CA3237482A1 (en) | 2021-11-03 | 2023-05-11 | The J. David Gladstone Institutes, A Testamentary Trust Established Under The Will Of J. David Gladstone | Precise genome editing using retrons |
| AU2022394985A1 (en) | 2021-11-16 | 2024-05-30 | Sail Biomedicines, Inc. | Novel ionizable lipids and lipid nanoparticles and methods of using the same |
| EP4436549A1 (en) | 2021-11-22 | 2024-10-02 | Sail Biomedicines, Inc. | Novel ionizable lipids and lipid nanoparticles and methods of using the same |
| CA3239266A1 (en) | 2021-11-24 | 2023-06-01 | Flagship Pioneering Innovations Vi, Llc | Coronavirus immunogen compositions and their uses |
| CN118401544A (en) | 2021-11-24 | 2024-07-26 | 旗舰创业创新六公司 | Varicella-zoster virus immunogenic compositions and uses thereof |
| WO2023097003A2 (en) | 2021-11-24 | 2023-06-01 | Flagship Pioneering Innovations Vi, Llc | Immunogenic compositions and their uses |
| TW202340460A (en) | 2021-12-17 | 2023-10-16 | 美商旗艦先鋒創新有限責任公司 | Methods for enrichment of circular rna under denaturing conditions |
| KR20240117149A (en) | 2021-12-22 | 2024-07-31 | 플래그쉽 파이어니어링 이노베이션스 브이아이, 엘엘씨 | Compositions and methods for purifying polyribonucleotides |
| WO2023122752A1 (en) | 2021-12-23 | 2023-06-29 | Renagade Therapeutics Management Inc. | Constrained lipids and methods of use thereof |
| KR20240118881A (en) | 2021-12-23 | 2024-08-05 | 플래그쉽 파이어니어링 이노베이션스 브이아이, 엘엘씨 | Circular polyribonucleotide encoding an antifusogenic polypeptide |
| WO2023141602A2 (en) | 2022-01-21 | 2023-07-27 | Renagade Therapeutics Management Inc. | Engineered retrons and methods of use |
| CN114409554A (en) * | 2022-01-27 | 2022-04-29 | 英维沃生物科技(苏州)有限公司 | Novel cationic lipid compound, composition and application thereof |
| WO2023147090A1 (en) | 2022-01-27 | 2023-08-03 | BioNTech SE | Pharmaceutical compositions for delivery of herpes simplex virus antigens and related methods |
| AU2023239151A1 (en) | 2022-03-25 | 2024-10-03 | Sail Biomedicines, Inc. | Novel ionizable lipids and lipid nanoparticles and methods of using the same |
| CN116354836A (en) * | 2022-03-25 | 2023-06-30 | 深圳市新合生物医疗科技有限公司 | Cationic lipid compound, preparation method and application thereof and mRNA delivery system |
| WO2023196931A1 (en) | 2022-04-07 | 2023-10-12 | Renagade Therapeutics Management Inc. | Cyclic lipids and lipid nanoparticles (lnp) for the delivery of nucleic acids or peptides for use in vaccinating against infectious agents |
| WO2023196634A2 (en) | 2022-04-08 | 2023-10-12 | Flagship Pioneering Innovations Vii, Llc | Vaccines and related methods |
| WO2023220083A1 (en) | 2022-05-09 | 2023-11-16 | Flagship Pioneering Innovations Vi, Llc | Trem compositions and methods of use for treating proliferative disorders |
| WO2023220729A2 (en) | 2022-05-13 | 2023-11-16 | Flagship Pioneering Innovations Vii, Llc | Double stranded dna compositions and related methods |
| WO2023218431A1 (en) | 2022-05-13 | 2023-11-16 | BioNTech SE | Rna compositions targeting hiv |
| WO2023230295A1 (en) | 2022-05-25 | 2023-11-30 | BioNTech SE | Rna compositions for delivery of monkeypox antigens and related methods |
| WO2023232747A1 (en) | 2022-05-30 | 2023-12-07 | BioNTech SE | Complexes for delivery of nucleic acids |
| CN117263818A (en) * | 2022-06-14 | 2023-12-22 | 杭州高田生物医药有限公司 | Cationic lipid compound, and preparation method and application thereof |
| WO2023250112A1 (en) | 2022-06-22 | 2023-12-28 | Flagship Pioneering Innovations Vi, Llc | Compositions of modified trems and uses thereof |
| TW202413424A (en) | 2022-08-01 | 2024-04-01 | 美商旗艦先鋒創新有限責任(Vii)公司 | Immunomodulatory proteins and related methods |
| WO2024035952A1 (en) | 2022-08-12 | 2024-02-15 | Remix Therapeutics Inc. | Methods and compositions for modulating splicing at alternative splice sites |
| WO2024049979A2 (en) | 2022-08-31 | 2024-03-07 | Senda Biosciences, Inc. | Novel ionizable lipids and lipid nanoparticles and methods of using the same |
| CN115197431B (en) * | 2022-09-15 | 2022-11-29 | 清华大学 | Synthesis and application of lipid-coupled completely degradable water-soluble polymer |
| WO2024064931A1 (en) | 2022-09-23 | 2024-03-28 | BioNTech SE | Compositions for delivery of liver stage antigens and related methods |
| WO2024063789A1 (en) | 2022-09-23 | 2024-03-28 | BioNTech SE | Compositions for delivery of malaria antigens and related methods |
| WO2024064934A1 (en) | 2022-09-23 | 2024-03-28 | BioNTech SE | Compositions for delivery of plasmodium csp antigens and related methods |
| WO2024063788A1 (en) | 2022-09-23 | 2024-03-28 | BioNTech SE | Compositions for delivery of malaria antigens and related methods |
| WO2024077191A1 (en) | 2022-10-05 | 2024-04-11 | Flagship Pioneering Innovations V, Inc. | Nucleic acid molecules encoding trif and additionalpolypeptides and their use in treating cancer |
| WO2024074634A1 (en) | 2022-10-06 | 2024-04-11 | BioNTech SE | Rna compositions targeting claudin-18.2 |
| WO2024074211A1 (en) | 2022-10-06 | 2024-04-11 | BioNTech SE | Rna compositions targeting claudin-18.2 |
| WO2024097664A1 (en) | 2022-10-31 | 2024-05-10 | Flagship Pioneering Innovations Vi, Llc | Compositions and methods for purifying polyribonucleotides |
| WO2024102799A1 (en) | 2022-11-08 | 2024-05-16 | Flagship Pioneering Innovations Vi, Llc | Compositions and methods for producing circular polyribonucleotides |
| WO2024129988A1 (en) | 2022-12-14 | 2024-06-20 | Flagship Pioneering Innovations Vii, Llc | Compositions and methods for delivery of therapeutic agents to bone |
| WO2024141955A1 (en) | 2022-12-28 | 2024-07-04 | BioNTech SE | Rna compositions targeting hiv |
| WO2024151685A1 (en) | 2023-01-09 | 2024-07-18 | Beth Israel Deaconess Medical Center, Inc. | Recombinant nucleic acid molecules and their use in wound healing |
| US20240269251A1 (en) | 2023-01-09 | 2024-08-15 | Flagship Pioneering Innovations V, Inc. | Genetic switches and their use in treating cancer |
| WO2024151583A2 (en) | 2023-01-09 | 2024-07-18 | Flagship Pioneering Innovations Vii, Llc | Vaccines and related methods |
| US20240238473A1 (en) | 2023-01-09 | 2024-07-18 | Beth Israel Deaconess Medical Center, Inc. | Recombinant nucleic acid molecules and their use in wound healing |
| US20240269263A1 (en) | 2023-02-06 | 2024-08-15 | Flagship Pioneering Innovations Vii, Llc | Immunomodulatory compositions and related methods |
| WO2024173307A2 (en) | 2023-02-13 | 2024-08-22 | Flagship Pioneering Innovation Vii, Llc | Cleavable linker-containing ionizable lipids and lipid carriers for therapeutic compositions |
| US20240285805A1 (en) | 2023-02-17 | 2024-08-29 | Flagship Pioneering Innovations Vii, Llc | Dna compositions comprising modified uracil |
| US20240293582A1 (en) | 2023-02-17 | 2024-09-05 | Flagship Pioneering Innovations Vii, Llc | Dna compositions comprising modified cytosine |
| WO2024192420A1 (en) | 2023-03-15 | 2024-09-19 | Flagship Pioneering Innovations Vi, Llc | Compositions comprising polyribonucleotides and uses thereof |
| WO2024192422A1 (en) | 2023-03-15 | 2024-09-19 | Flagship Pioneering Innovations Vi, Llc | Immunogenic compositions and uses thereof |
Citations (48)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3993754A (en) | 1974-10-09 | 1976-11-23 | The United States Of America As Represented By The United States Energy Research And Development Administration | Liposome-encapsulated actinomycin for cancer chemotherapy |
| US4145410A (en) | 1976-10-12 | 1979-03-20 | Sears Barry D | Method of preparing a controlled-release pharmaceutical preparation, and resulting composition |
| US4224179A (en) | 1977-08-05 | 1980-09-23 | Battelle Memorial Institute | Process for the preparation of liposomes in aqueous solution |
| US4235871A (en) | 1978-02-24 | 1980-11-25 | Papahadjopoulos Demetrios P | Method of encapsulating biologically active materials in lipid vesicles |
| US4522803A (en) | 1983-02-04 | 1985-06-11 | The Liposome Company, Inc. | Stable plurilamellar vesicles, their preparation and use |
| US4588578A (en) | 1983-08-08 | 1986-05-13 | The Liposome Company, Inc. | Lipid vesicles prepared in a monophase |
| US4603044A (en) | 1983-01-06 | 1986-07-29 | Technology Unlimited, Inc. | Hepatocyte Directed Vesicle delivery system |
| WO1986004920A1 (en) | 1985-02-13 | 1986-08-28 | Biotechnology Research Partners, Limited | Human metallothionein-ii promoter in mammalian expression system |
| WO1987002062A1 (en) | 1985-10-04 | 1987-04-09 | Biotechnology Research Partners, Ltd. | Recombinant apolipoproteins and methods |
| US4737323A (en) | 1986-02-13 | 1988-04-12 | Liposome Technology, Inc. | Liposome extrusion method |
| US4957773A (en) | 1989-02-13 | 1990-09-18 | Syracuse University | Deposition of boron-containing films from decaborane |
| US5013556A (en) | 1989-10-20 | 1991-05-07 | Liposome Technology, Inc. | Liposomes with enhanced circulation time |
| US5059528A (en) | 1987-05-28 | 1991-10-22 | Ucb, S.A. | Expression of human proapolipoprotein a-i |
| US5116739A (en) | 1984-10-16 | 1992-05-26 | Mitsubishi Chemical Industries Limited | Process for the production of human apolipoprotein e, and transformed hosts and products thereof |
| US5168045A (en) | 1989-08-18 | 1992-12-01 | The Scripps Research Institute | Diagnostic systems and methods using polypeptide analogs of apolipoprotein e |
| US5177189A (en) | 1989-08-18 | 1993-01-05 | The Scripps Research Institute | Polypeptide analogs of Apolipoprotein E |
| US5182364A (en) | 1990-02-26 | 1993-01-26 | The Scripps Research Institute | Polypeptide analogs of apolipoprotein E |
| US5286634A (en) | 1989-09-28 | 1994-02-15 | Stadler Joan K | Synergistic method for host cell transformation |
| US5473039A (en) | 1989-08-18 | 1995-12-05 | The Scripps Research Institute | Polypeptide analogs of apolipoprotein E, diagnostic systems and methods using the analogs |
| US5525472A (en) | 1991-06-26 | 1996-06-11 | Bio-Technology General Corp. | Method for production and purification or recombinant Apolipoprotein E from bacteria |
| US5532130A (en) | 1993-07-20 | 1996-07-02 | Dyad Pharmaceutical Corporation | Methods and compositions for sequence-specific hybridization of RNA by 2'-5' oligonucleotides |
| US5534499A (en) | 1994-05-19 | 1996-07-09 | The University Of British Columbia | Lipophilic drug derivatives for use in liposomes |
| US5672685A (en) | 1995-10-04 | 1997-09-30 | Duke University | Source of apolipoprotein E and method of isolating apolipoprotein E |
| US5721114A (en) | 1992-12-11 | 1998-02-24 | Pharmacia & Upjohn Aktiebolag | Expression system for producing apolipoprotein AI-M |
| US5820873A (en) | 1994-09-30 | 1998-10-13 | The University Of British Columbia | Polyethylene glycol modified ceramide lipids and liposome uses thereof |
| US5834596A (en) | 1995-03-03 | 1998-11-10 | Pharmacia & Upjohn Ab | Process for purifying ApoA or ApoE |
| US5840688A (en) | 1994-03-22 | 1998-11-24 | Research Corporation Technologies, Inc. | Eating suppressant peptides |
| US5876968A (en) | 1991-12-13 | 1999-03-02 | Pharmacia & Upjohn Aktiebolag | Dimer of molecular variant of apolipoprotein and processes for the production thereof |
| US5885613A (en) | 1994-09-30 | 1999-03-23 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| US5976567A (en) | 1995-06-07 | 1999-11-02 | Inex Pharmaceuticals Corp. | Lipid-nucleic acid particles prepared via a hydrophobic lipid-nucleic acid complex intermediate and use for gene transfer |
| US6004925A (en) | 1997-09-29 | 1999-12-21 | J. L. Dasseux | Apolipoprotein A-I agonists and their use to treat dyslipidemic disorders |
| US6027726A (en) | 1994-09-30 | 2000-02-22 | Inex Phamaceuticals Corp. | Glycosylated protein-liposome conjugates and methods for their preparation |
| US6037323A (en) | 1997-09-29 | 2000-03-14 | Jean-Louis Dasseux | Apolipoprotein A-I agonists and their use to treat dyslipidemic disorders |
| US6046166A (en) | 1997-09-29 | 2000-04-04 | Jean-Louis Dasseux | Apolipoprotein A-I agonists and their use to treat dyslipidemic disorders |
| US6287591B1 (en) | 1997-05-14 | 2001-09-11 | Inex Pharmaceuticals Corp. | Charged therapeutic agents encapsulated in lipid particles containing four lipid components |
| US6320017B1 (en) | 1997-12-23 | 2001-11-20 | Inex Pharmaceuticals Corp. | Polyamide oligomers |
| US6372886B1 (en) | 1992-06-23 | 2002-04-16 | Arch Development Corp. | Expression and purification of kringle domains of human apolipoprotein (a) in E. coli |
| US20040142025A1 (en) | 2002-06-28 | 2004-07-22 | Protiva Biotherapeutics Ltd. | Liposomal apparatus and manufacturing methods |
| US20060051405A1 (en) | 2004-07-19 | 2006-03-09 | Protiva Biotherapeutics, Inc. | Compositions for the delivery of therapeutic agents and uses thereof |
| US20070042031A1 (en) | 2005-07-27 | 2007-02-22 | Protiva Biotherapeutics, Inc. | Systems and methods for manufacturing liposomes |
| WO2008042973A2 (en) | 2006-10-03 | 2008-04-10 | Alnylam Pharmaceuticals, Inc. | Lipid containing formulations |
| WO2009086558A1 (en) | 2008-01-02 | 2009-07-09 | Tekmira Pharmaceuticals Corporation | Improved compositions and methods for the delivery of nucleic acids |
| WO2010042877A1 (en) | 2008-10-09 | 2010-04-15 | Tekmira Pharmaceuticals Corporation | Improved amino lipids and methods for the delivery of nucleic acids |
| WO2010054405A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| WO2010054384A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Lipids and compositions for the delivery of therapeutics |
| WO2010088537A2 (en) | 2009-01-29 | 2010-08-05 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| WO2010129709A1 (en) | 2009-05-05 | 2010-11-11 | Alnylam Pharmaceuticals, Inc. | Lipid compositions |
| US10421908B2 (en) | 2016-03-08 | 2019-09-24 | Dic Corporation | Liquid crystal composition and liquid crystal display device including the same |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4013632A1 (en) * | 1990-04-27 | 1991-10-31 | Max Planck Gesellschaft | LIPOSOMES WITH POSITIVE EXCESS CHARGE |
| EP0685234B1 (en) * | 1993-02-19 | 2000-05-10 | Nippon Shinyaku Company, Limited | Drug composition containing nucleic acid copolymer |
| JP3563505B2 (en) * | 1995-10-16 | 2004-09-08 | 花王株式会社 | Novel quaternary ammonium salt, method for producing the same, and hair cosmetic containing the same |
| JPH09278726A (en) * | 1996-04-11 | 1997-10-28 | Takeshi Imanishi | New liposome |
| JPH09301936A (en) * | 1996-05-09 | 1997-11-25 | Kao Corp | New quaternary ammonium salt, its production, softening and finishing agent containing the same and hair cosmetic |
| US20030229037A1 (en) * | 2000-02-07 | 2003-12-11 | Ulrich Massing | Novel cationic amphiphiles |
| AU2006259415B2 (en) * | 2005-06-15 | 2012-08-30 | Massachusetts Institute Of Technology | Amine-containing lipids and uses thereof |
| BRPI0713331A2 (en) * | 2006-06-30 | 2012-03-13 | Hokkaido System Science Co., Ltd. | composition for nucleic acid transfection |
| FR2909378B1 (en) * | 2006-12-05 | 2012-01-13 | Biochimie Appliquee Soc | CERAMIDES OR PESUDO-CERAMIDES FOURERNISES AND COSMETIC COMPOSITIONS CONTAINING SAME |
| TW201002126A (en) * | 2007-12-21 | 2010-01-01 | Du Pont | Encapsulation assembly for electronic devices |
| CA2742846A1 (en) * | 2008-11-17 | 2010-05-20 | Enzon Pharmaceuticals, Inc. | Releasable fusogenic lipids for nucleic acids delivery systems |
| CA2800401C (en) * | 2010-06-03 | 2020-09-15 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| CA3165769A1 (en) * | 2011-12-07 | 2013-06-13 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
-
2011
- 2011-06-03 CA CA2800401A patent/CA2800401C/en active Active
- 2011-06-03 ES ES11726579.3T patent/ES2634087T3/en active Active
- 2011-06-03 JP JP2013513394A patent/JP5893611B2/en active Active
- 2011-06-03 KR KR1020137000092A patent/KR101967411B1/en active IP Right Grant
- 2011-06-03 NZ NZ605079A patent/NZ605079A/en unknown
- 2011-06-03 KR KR1020197009607A patent/KR20190039347A/en not_active Application Discontinuation
- 2011-06-03 MX MX2012014079A patent/MX341332B/en active IP Right Grant
- 2011-06-03 SG SG2012087441A patent/SG186085A1/en unknown
- 2011-06-03 CN CN201180037927.7A patent/CN103096875B/en active Active
- 2011-06-03 IL IL300109A patent/IL300109A/en unknown
- 2011-06-03 AU AU2011261247A patent/AU2011261247B2/en active Active
- 2011-06-03 EP EP11726579.3A patent/EP2575764B1/en active Active
- 2011-06-03 EP EP17000656.3A patent/EP3254672A1/en active Pending
- 2011-06-03 DK DK11726579.3T patent/DK2575764T3/en active
- 2011-06-03 WO PCT/US2011/039164 patent/WO2011153493A2/en active Application Filing
- 2011-06-03 US US13/153,200 patent/US9012498B2/en active Active
-
2012
- 2012-11-22 IL IL223207A patent/IL223207B/en active IP Right Grant
-
2015
- 2015-03-18 US US14/661,945 patent/US9682922B2/en active Active
-
2016
- 2016-02-23 JP JP2016031867A patent/JP6409011B2/en active Active
- 2016-08-05 AU AU2016210739A patent/AU2016210739A1/en not_active Abandoned
-
2017
- 2017-05-15 US US15/595,646 patent/US10266484B2/en active Active
-
2018
- 2018-05-14 IL IL259334A patent/IL259334B2/en unknown
- 2018-05-15 AU AU2018203382A patent/AU2018203382A1/en not_active Abandoned
- 2018-09-21 JP JP2018177190A patent/JP2019011345A/en active Pending
-
2020
- 2020-01-15 AU AU2020200286A patent/AU2020200286B2/en active Active
-
2022
- 2022-06-14 AU AU2022204146A patent/AU2022204146B2/en active Active
-
2024
- 2024-08-02 AU AU2024205466A patent/AU2024205466A1/en active Pending
Patent Citations (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3993754A (en) | 1974-10-09 | 1976-11-23 | The United States Of America As Represented By The United States Energy Research And Development Administration | Liposome-encapsulated actinomycin for cancer chemotherapy |
| US4145410A (en) | 1976-10-12 | 1979-03-20 | Sears Barry D | Method of preparing a controlled-release pharmaceutical preparation, and resulting composition |
| US4224179A (en) | 1977-08-05 | 1980-09-23 | Battelle Memorial Institute | Process for the preparation of liposomes in aqueous solution |
| US4235871A (en) | 1978-02-24 | 1980-11-25 | Papahadjopoulos Demetrios P | Method of encapsulating biologically active materials in lipid vesicles |
| US4603044A (en) | 1983-01-06 | 1986-07-29 | Technology Unlimited, Inc. | Hepatocyte Directed Vesicle delivery system |
| US4522803A (en) | 1983-02-04 | 1985-06-11 | The Liposome Company, Inc. | Stable plurilamellar vesicles, their preparation and use |
| US4588578A (en) | 1983-08-08 | 1986-05-13 | The Liposome Company, Inc. | Lipid vesicles prepared in a monophase |
| US5116739A (en) | 1984-10-16 | 1992-05-26 | Mitsubishi Chemical Industries Limited | Process for the production of human apolipoprotein e, and transformed hosts and products thereof |
| WO1986004920A1 (en) | 1985-02-13 | 1986-08-28 | Biotechnology Research Partners, Limited | Human metallothionein-ii promoter in mammalian expression system |
| WO1987002062A1 (en) | 1985-10-04 | 1987-04-09 | Biotechnology Research Partners, Ltd. | Recombinant apolipoproteins and methods |
| US4737323A (en) | 1986-02-13 | 1988-04-12 | Liposome Technology, Inc. | Liposome extrusion method |
| US5059528A (en) | 1987-05-28 | 1991-10-22 | Ucb, S.A. | Expression of human proapolipoprotein a-i |
| US4957773A (en) | 1989-02-13 | 1990-09-18 | Syracuse University | Deposition of boron-containing films from decaborane |
| US5473039A (en) | 1989-08-18 | 1995-12-05 | The Scripps Research Institute | Polypeptide analogs of apolipoprotein E, diagnostic systems and methods using the analogs |
| US5168045A (en) | 1989-08-18 | 1992-12-01 | The Scripps Research Institute | Diagnostic systems and methods using polypeptide analogs of apolipoprotein e |
| US5177189A (en) | 1989-08-18 | 1993-01-05 | The Scripps Research Institute | Polypeptide analogs of Apolipoprotein E |
| US5286634A (en) | 1989-09-28 | 1994-02-15 | Stadler Joan K | Synergistic method for host cell transformation |
| US5013556A (en) | 1989-10-20 | 1991-05-07 | Liposome Technology, Inc. | Liposomes with enhanced circulation time |
| US5182364A (en) | 1990-02-26 | 1993-01-26 | The Scripps Research Institute | Polypeptide analogs of apolipoprotein E |
| US5525472A (en) | 1991-06-26 | 1996-06-11 | Bio-Technology General Corp. | Method for production and purification or recombinant Apolipoprotein E from bacteria |
| US5876968A (en) | 1991-12-13 | 1999-03-02 | Pharmacia & Upjohn Aktiebolag | Dimer of molecular variant of apolipoprotein and processes for the production thereof |
| US6372886B1 (en) | 1992-06-23 | 2002-04-16 | Arch Development Corp. | Expression and purification of kringle domains of human apolipoprotein (a) in E. coli |
| US5721114A (en) | 1992-12-11 | 1998-02-24 | Pharmacia & Upjohn Aktiebolag | Expression system for producing apolipoprotein AI-M |
| US5532130A (en) | 1993-07-20 | 1996-07-02 | Dyad Pharmaceutical Corporation | Methods and compositions for sequence-specific hybridization of RNA by 2'-5' oligonucleotides |
| US5840688A (en) | 1994-03-22 | 1998-11-24 | Research Corporation Technologies, Inc. | Eating suppressant peptides |
| US5534499A (en) | 1994-05-19 | 1996-07-09 | The University Of British Columbia | Lipophilic drug derivatives for use in liposomes |
| US5820873A (en) | 1994-09-30 | 1998-10-13 | The University Of British Columbia | Polyethylene glycol modified ceramide lipids and liposome uses thereof |
| US5885613A (en) | 1994-09-30 | 1999-03-23 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| US6027726A (en) | 1994-09-30 | 2000-02-22 | Inex Phamaceuticals Corp. | Glycosylated protein-liposome conjugates and methods for their preparation |
| US5834596A (en) | 1995-03-03 | 1998-11-10 | Pharmacia & Upjohn Ab | Process for purifying ApoA or ApoE |
| US5976567A (en) | 1995-06-07 | 1999-11-02 | Inex Pharmaceuticals Corp. | Lipid-nucleic acid particles prepared via a hydrophobic lipid-nucleic acid complex intermediate and use for gene transfer |
| US5672685A (en) | 1995-10-04 | 1997-09-30 | Duke University | Source of apolipoprotein E and method of isolating apolipoprotein E |
| US6287591B1 (en) | 1997-05-14 | 2001-09-11 | Inex Pharmaceuticals Corp. | Charged therapeutic agents encapsulated in lipid particles containing four lipid components |
| US6858225B2 (en) | 1997-05-14 | 2005-02-22 | Inex Pharmaceuticals Corporation | Lipid-encapsulated polyanionic nucleic acid |
| US6004925A (en) | 1997-09-29 | 1999-12-21 | J. L. Dasseux | Apolipoprotein A-I agonists and their use to treat dyslipidemic disorders |
| US6037323A (en) | 1997-09-29 | 2000-03-14 | Jean-Louis Dasseux | Apolipoprotein A-I agonists and their use to treat dyslipidemic disorders |
| US6046166A (en) | 1997-09-29 | 2000-04-04 | Jean-Louis Dasseux | Apolipoprotein A-I agonists and their use to treat dyslipidemic disorders |
| US6320017B1 (en) | 1997-12-23 | 2001-11-20 | Inex Pharmaceuticals Corp. | Polyamide oligomers |
| US20040142025A1 (en) | 2002-06-28 | 2004-07-22 | Protiva Biotherapeutics Ltd. | Liposomal apparatus and manufacturing methods |
| US20060051405A1 (en) | 2004-07-19 | 2006-03-09 | Protiva Biotherapeutics, Inc. | Compositions for the delivery of therapeutic agents and uses thereof |
| US20070042031A1 (en) | 2005-07-27 | 2007-02-22 | Protiva Biotherapeutics, Inc. | Systems and methods for manufacturing liposomes |
| WO2008042973A2 (en) | 2006-10-03 | 2008-04-10 | Alnylam Pharmaceuticals, Inc. | Lipid containing formulations |
| WO2009086558A1 (en) | 2008-01-02 | 2009-07-09 | Tekmira Pharmaceuticals Corporation | Improved compositions and methods for the delivery of nucleic acids |
| WO2010042877A1 (en) | 2008-10-09 | 2010-04-15 | Tekmira Pharmaceuticals Corporation | Improved amino lipids and methods for the delivery of nucleic acids |
| WO2010054405A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| WO2010054401A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| WO2010054406A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Novel lipids and compositions for the delivery of therapeutics |
| WO2010054384A1 (en) | 2008-11-10 | 2010-05-14 | Alnylam Pharmaceuticals, Inc. | Lipids and compositions for the delivery of therapeutics |
| WO2010088537A2 (en) | 2009-01-29 | 2010-08-05 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| WO2010129709A1 (en) | 2009-05-05 | 2010-11-11 | Alnylam Pharmaceuticals, Inc. | Lipid compositions |
| US10421908B2 (en) | 2016-03-08 | 2019-09-24 | Dic Corporation | Liquid crystal composition and liquid crystal display device including the same |
Non-Patent Citations (83)
| Title |
|---|
| "The Proteins", vol. II, 1976, ACADEMIC PRESS, pages: 105 - 237 |
| ABRA, RM ET AL., J. LIPOSOME RES., vol. 12, 2002, pages 1 - 3 |
| AGRAWAL, TRENDS IN BIOTECH, vol. 14, 1996, pages 376 - 387 |
| ALLEN, BIOCHIMICA ET BIOPHYSICA ACTA, vol. 1237, 1995, pages 99 - 108 |
| AVIRAM ET AL., ARTERIOSCLER. THROMB. VASC. BIOL., vol. 18, no. 10, 1998, pages 1617 - 24 |
| AVIRAM ET AL., ARTERIOSCLER. THROMB. VASE. BIOL., vol. 18, no. 10, 1998, pages 1617 - 24 |
| AVIRAM ET AL., J. CLIN. INVEST., vol. 101, no. 8, 1998, pages 1581 - 90 |
| BEHR, ACC. CHEM. RES., vol. 26, 1993, pages 274 - 278 |
| BENNETT ET AL., MOL. PHARM., vol. 41, 1992, pages 1023 - 1033 |
| BIELICKI, ODA, BIOCHEMISTRY, vol. 41, 2002, pages 2089 - 96 |
| BILLECKE ET AL., DRUG METAB. DISPOS., vol. 28, no. 11, 2000, pages 1335 - 42 |
| BLUME, BIOCHIMICA ET BIOPHYSICA ACTA, vol. 1149, 1993, pages 180 - 184 |
| BODANSZKY ET AL.: "Peptide Synthesis", 1976, JOHN WILEY & SONS |
| BRIGHAM ET AL., AM. J. MED. SCI., vol. 298, 1989, pages 278 - 281 |
| BRIGHAM ET AL., AM. J. SCI., vol. 298, no. 4, 1989, pages 278 - 281 |
| CHEUNG ET AL., J. LIPID RES., vol. 28, no. 8, 1987, pages 913 - 29 |
| CHUNG ET AL., J. LIPID RES., vol. 21, no. 3, 1980, pages 284 - 91 |
| CULLIS ET AL., CHEM PHYS LIPIDS, vol. 40, 1986, pages 127 - 144 |
| CULVER: "Human Gene Therapy", 1994, MARYANN LIEBERT, INC., pages: 70 - 71 |
| DAUM ET AL., J. MOL. MED., vol. 77, 1999, pages 614 - 22 |
| DEFREES ET AL., JOURNAL OF THE AMERICAN CHEMISTRY SOCIETY, vol. 118, 1996, pages 6101 - 6104 |
| DRAGANOV ET AL., J. BIOL. CHEM., vol. 275, no. 43, 2000, pages 33435 - 42 |
| DUVERGER ET AL., ARTERIOSCLER. THROMB. VASC. BIOL., vol. 16, no. 12, 1996, pages 1424 - 29 |
| DUVERGER ET AL., EURO. J. BIOCHEM., vol. 201, no. 2, 1991, pages 373 - 83 |
| DYER ET AL., J. LIPID RES., vol. 36, no. 1, 1995, pages 80 - 8 |
| DYER, J. BIOL. CHEM., vol. 266, no. 23, 1991, pages 150009 - 15 |
| FRANCESCHINI ET AL., J. BIOL. CHEM., vol. 260, 1985, pages 1632 - 35 |
| GALBRAITH ET AL., ANTISENSE NUCL. ACID DRUG DES., vol. 4, 1994, pages 201 - 206 |
| GONG ET AL., J. BIOL. CHEM., vol. 277, no. 33, 2002, pages 29919 - 26 |
| GOODFELLOW, NATURE, vol. 341, 1989, pages 102 - 103 |
| GORDON ET AL., J. BIOL. CHEM., vol. 259, no. 1, 1984, pages 468 - 74 |
| GREEN, T.W.: "Protective Groups in Organic Synthesis", 1999, WILEY-INTERSCIENCE |
| HEATH: "Methods in Enzymology", vol. 149, 1987, ACADEMIC PRESS, INC., article "Covalent Attachment of Proteins to Liposomes", pages: 111 - 119 |
| HILL, J. BIOL. CHEM., vol. 273, no. 47, 1998, pages 30979 - 84 |
| HIXSON, POWERS, J. LIPID RES., vol. 32, no. 9, 1991, pages 1529 - 35 |
| HOEG ET AL., J. BIOL. CHEM., vol. 261, no. 9, 1986, pages 3911 - 4 |
| HYDE ET AL., NATURE, vol. 362, 1993, pages 250 - 256 |
| J. MED. CHEM., vol. 38, 1995, pages 636 - 46 |
| JIA ET AL., BIOCHEM. BIOPHYS. RES. COMM., vol. 297, 2002, pages 206 - 13 |
| KIRPOTIN ET AL., FEBS LETTERS, vol. 388, 1996, pages 115 - 118 |
| KLIBANOV ET AL., JOURNAL OF LIPOSOME RESEARCH, vol. 2, 1992, pages 321 - 334 |
| KLIBANOV, JOURNAL OF LIPOSOME RESEARCH, vol. 2, 1992, pages 321 - 334 |
| KLON ET AL., BIOPHYS. J., vol. 79, no. 3, 2000, pages 1679 - 87 |
| KUNKEL, BRIT. MED. BULL., vol. 45, no. 3, 1989, pages 630 - 643 |
| LACKNER ET AL., J. BIOL. CHEM., vol. 260, no. 2, 1985, pages 703 - 6 |
| LEONETTI ET AL., PROC. NATL. ACAD. SCI. (USA, vol. 87, 1990, pages 2448 - 2451 |
| MANNINO ET AL., BIOTECHNIQUES, vol. 6, 1988, pages 682 - 690 |
| MCLEAN ET AL., J. BIOL. CHEM., vol. 258, no. 14, 1983, pages 8993 - 9000 |
| MCOMIE: "Protective Groups in Organic Chemistry", 1973, PLENUM PRESS |
| MERRIFIELD, J. AM. CHEM. SOC., vol. 85, 1963, pages 2149 - 2154 |
| MULUGETA ET AL., J. CHROMATOGR., vol. 798, no. 1-2, 1998, pages 83 - 90 |
| NICOLAU ET AL., CRIT. REV. THER. DRUG CARRIER SYST., vol. 6, 1989, pages 239 - 271 |
| OHTA ET AL., J. BIOL. CHEM., vol. 259, no. 23, 1984, pages 14888 - 93 |
| PALGUNACHARI, ARTERIOSCLER. THROB. VASC. BIOL., vol. 16, no. 2, 1996, pages 328 - 38 |
| PERSSON ET AL., J. CHROMATOGR, vol. 711, 1998, pages 97 - 109 |
| POWELL ET AL., CELL, vol. 50, no. 6, 1987, pages 831 - 40 |
| RALL ET AL.: "Structural basis for receptor binding heterogeneity of apolipoprotein E from type III hyperlipoproteinemic subjects", PROC. NAT. ACAD. SCI., vol. 79, 1982, pages 4696 - 4700 |
| RENNEISEN ET AL., J. BIO. CHEM., vol. 265, 1990, pages 16337 - 16342 |
| SACRE ET AL., FEBS LETT., vol. 540, no. 1-3, 2003, pages 181 - 7 |
| SAPRA, P., ALLEN, TM, PROG. LIPID RES., vol. 42, no. 5, 2003, pages 439 - 62 |
| SHELNESS ET AL., J. BIOL. CHEM., vol. 259, no. 15, 1984, pages 9929 - 35 |
| SHELNESS ET AL., J. BIOL. CHEM., vol. 260, no. 14, 1985, pages 8637 - 46 |
| SORENSON ET AL., ARTERIOSCLER. THROMB. VASC. BIOL., vol. 19, no. 9, 1999, pages 2214 - 25 |
| STEINMETZ, UTERMANN, J. BIOL. CHEM., vol. 260, no. 4, 1985, pages 2258 - 64 |
| STRAUBRINGER ET AL.: "Methods in Enzymology", vol. 101, 1983, ACADEMIC PRESS, pages: 512 - 527 |
| STUART, YOUNG: "Solid Phase Peptide. Synthesis", 1984, PIERCE CHEMICAL COMPANY |
| TETRAHEDRON, vol. 63, 2006, pages 1140 - 1145 |
| THIERRY, A.R. ET AL.: "Gene Regulation: Biology of Antisense RNA and DNA", 1992, RAVEN PRESS, pages: 147 - 161 |
| THURBERG ET AL., J. BIOL. CHEM., vol. 271, no. 11, pages 6062 - 70 |
| UHLMANN E. ET AL.: "Antisense: Chemical Modifications. Encyclopedia of Cancer", vol. X., 1997, ACADEMIC PRESS INC., pages: 64 - 81 |
| VLASSOV ET AL., BIOCHIM. BIOPHYS. ACTA, vol. 1197, 1994, pages 95 - 1082 |
| WEERS ET AL., BIOPHYS. CHEM., vol. 100, no. 1-3, 2003, pages 481 - 92 |
| WEISGRABER ET AL.: "Human E apoprotein heterogeneity: cysteine-arginine interchanges in the amino acid sequence of the apo-E isoforms", J. BIOL. CHEM., vol. 256, 1981, pages 9077 - 9083, XP055343671 |
| WEISGRABER, J. LIPID RES, vol. 31, no. 8, 1990, pages 1503 - 11 |
| WEISGRABER, J. LIPID RES., vol. 31, no. 8, 1990, pages 1503 - 11 |
| WIDLER ET AL., J. BIOL. CHEM., vol. 255, no. 21, 1980, pages 10464 - 71 |
| ZALIPSKY, BIOCONJUGATE CHEMISTRY, vol. 4, 1993, pages 296 - 299 |
| ZALIPSKY, FEBS LETTERS, vol. 353, 1994, pages 71 - 74 |
| ZALIPSKY: "Stealth Liposomes", 1995, CRC PRESS |
| ZELPHATI, 0, SZOKA, F.C., J. CONTR. REL., vol. 41, 1996, pages 99 - 119 |
| ZELPHATI, O. ET AL., ANTISENSE. RES. DEV., vol. 3, 1993, pages 323 - 338 |
| ZHU ET AL., SCIENCE, vol. 261, 1993, pages 209 - 211 |
| ZIMMERMANN ET AL., NATURE, vol. 441, 2006, pages 111 - 114 |
Cited By (164)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2629802A2 (en) * | 2010-10-21 | 2013-08-28 | Merck Sharp & Dohme Corp. | Novel low molecular weight cationic lipids for oligonucleotide delivery |
| EP2629802A4 (en) * | 2010-10-21 | 2014-06-25 | Sirna Therapeutics Inc | Novel low molecular weight cationic lipids for oligonucleotide delivery |
| US11679158B2 (en) | 2011-12-07 | 2023-06-20 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US11071784B2 (en) | 2011-12-07 | 2021-07-27 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US10369226B2 (en) | 2011-12-07 | 2019-08-06 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| JP2018111707A (en) * | 2011-12-07 | 2018-07-19 | アルニラム・ファーマシューティカルズ・インコーポレーテッド | Biodegradable lipids for delivery of active agents |
| US11633480B2 (en) | 2011-12-07 | 2023-04-25 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2013086322A1 (en) * | 2011-12-07 | 2013-06-13 | Alnylam Pharmaceuticals, Inc. | Branched alkyl and cycloalkyl terminated biodegradable lipids for the delivery of active agents |
| JP2015500835A (en) * | 2011-12-07 | 2015-01-08 | アルニラム・ファーマシューティカルズ・インコーポレーテッド | Biodegradable lipids for delivery of active agents |
| US11590229B2 (en) | 2011-12-07 | 2023-02-28 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US11246933B1 (en) | 2011-12-07 | 2022-02-15 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US11612657B2 (en) | 2011-12-07 | 2023-03-28 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| EP3988537A1 (en) | 2011-12-07 | 2022-04-27 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US11382979B2 (en) | 2011-12-07 | 2022-07-12 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US9463247B2 (en) | 2011-12-07 | 2016-10-11 | Alnylam Pharmaceuticals, Inc. | Branched alkyl and cycloalkyl terminated biodegradable lipids for the delivery of active agents |
| AU2012347637B2 (en) * | 2011-12-07 | 2017-09-14 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US11400158B2 (en) | 2011-12-07 | 2022-08-02 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2013086354A1 (en) | 2011-12-07 | 2013-06-13 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US11633479B2 (en) | 2011-12-07 | 2023-04-25 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| US9061063B2 (en) | 2011-12-07 | 2015-06-23 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2013116126A1 (en) * | 2012-02-01 | 2013-08-08 | Merck Sharp & Dohme Corp. | Novel low molecular weight, biodegradable cationic lipids for oligonucleotide delivery |
| US10703789B2 (en) | 2012-04-02 | 2020-07-07 | Modernatx, Inc. | Modified polynucleotides for the production of secreted proteins |
| US10493167B2 (en) | 2012-04-02 | 2019-12-03 | Modernatx, Inc. | In vivo production of proteins |
| US10463751B2 (en) | 2012-04-02 | 2019-11-05 | Modernatx, Inc. | Modified polynucleotides for the production of cytoplasmic and cytoskeletal proteins |
| JP2017121240A (en) * | 2012-04-02 | 2017-07-13 | モデルナティエックス インコーポレイテッドModernaTX,Inc. | Modified polynucleotides for production of membrane proteins |
| US10501513B2 (en) | 2012-04-02 | 2019-12-10 | Modernatx, Inc. | Modified polynucleotides for the production of oncology-related proteins and peptides |
| US11564998B2 (en) | 2012-04-02 | 2023-01-31 | Modernatx, Inc. | Modified polynucleotides for the production of cytoplasmic and cytoskeletal proteins |
| US10501512B2 (en) | 2012-04-02 | 2019-12-10 | Modernatx, Inc. | Modified polynucleotides |
| US10583203B2 (en) | 2012-04-02 | 2020-03-10 | Modernatx, Inc. | In vivo production of proteins |
| EP3434667A1 (en) | 2012-04-19 | 2019-01-30 | Sirna Therapeutics, Inc. | Novel diester and triester based low molecular weight, biodegradable cationic lipids for oligonucleotide delivery |
| US10307378B2 (en) | 2012-04-19 | 2019-06-04 | Sirna Therapeutics, Inc. | Diester and triester based low molecular weight, biodegradeable cationic lipids for oligonucleotide delivery |
| WO2014008334A1 (en) | 2012-07-06 | 2014-01-09 | Alnylam Pharmaceuticals, Inc. | Stable non-aggregating nucleic acid lipid particle formulations |
| WO2014089486A1 (en) | 2012-12-07 | 2014-06-12 | Shire Human Genetic Therapies, Inc. | Lipidic nanoparticles for mrna delivering |
| EP2929035A1 (en) * | 2012-12-07 | 2015-10-14 | Shire Human Genetic Therapies, Inc. | Lipidic nanoparticles for mrna delivering |
| US9687448B2 (en) | 2012-12-07 | 2017-06-27 | Alnylam Pharmaceuticals, Inc. | Nucleic acid lipid particle formulations |
| WO2014089239A1 (en) | 2012-12-07 | 2014-06-12 | Alnylam Pharmaceuticals, Inc. | Improved nucleic acid lipid particle formulations |
| US9850202B2 (en) | 2013-11-18 | 2017-12-26 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US9365610B2 (en) | 2013-11-18 | 2016-06-14 | Arcturus Therapeutics, Inc. | Asymmetric ionizable cationic lipid for RNA delivery |
| US9567296B2 (en) | 2013-11-18 | 2017-02-14 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US9580711B2 (en) | 2013-11-18 | 2017-02-28 | Arcturus Therapeutics, Inc. | Lipid particles with asymmetric cationic lipids for RNA delivery |
| USRE49233E1 (en) | 2013-11-18 | 2022-10-04 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US9593077B2 (en) | 2013-11-18 | 2017-03-14 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US10781169B2 (en) | 2013-11-18 | 2020-09-22 | Arcturus Therapeutics, Inc. | Method of synthesis of an ionizable cationic lipid |
| US10556861B2 (en) | 2013-11-18 | 2020-02-11 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US9896413B2 (en) | 2013-11-18 | 2018-02-20 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US10399937B2 (en) | 2013-11-18 | 2019-09-03 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US9670152B2 (en) | 2013-11-18 | 2017-06-06 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| WO2015095351A1 (en) | 2013-12-19 | 2015-06-25 | Novartis Ag | LEPTIN mRNA COMPOSITIONS AND FORMULATIONS |
| US11634379B2 (en) | 2014-06-25 | 2023-04-25 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| US10723692B2 (en) | 2014-06-25 | 2020-07-28 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| US10252974B2 (en) | 2014-08-07 | 2019-04-09 | Takeda Pharmaceutical Company Limited | Cationic lipid |
| KR20170039722A (en) | 2014-08-07 | 2017-04-11 | 다케다 야쿠힝 고교 가부시키가이샤 | Cationic lipid |
| WO2016100716A1 (en) | 2014-12-18 | 2016-06-23 | Vasant Jadhav | Reversirtm compounds |
| US10081598B2 (en) | 2014-12-26 | 2018-09-25 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| US11168051B2 (en) | 2015-06-29 | 2021-11-09 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| US10221127B2 (en) | 2015-06-29 | 2019-03-05 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| WO2017019636A1 (en) * | 2015-07-24 | 2017-02-02 | Insmed Incorporated | Compositions and methods for the treatment of sarcoidosis |
| US11712481B2 (en) | 2015-10-28 | 2023-08-01 | Acuitas Therapeutics, Inc. | Lipid nanoparticle formulations |
| US10166298B2 (en) | 2015-10-28 | 2019-01-01 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| US11040112B2 (en) | 2015-10-28 | 2021-06-22 | Acuitas Therapeutics, Inc. | Lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| WO2017222016A1 (en) | 2016-06-24 | 2017-12-28 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Cationic lipid |
| AU2017282459B2 (en) * | 2016-06-24 | 2021-05-06 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| KR20190021218A (en) | 2016-06-24 | 2019-03-05 | 에자이 알앤드디 매니지먼트 가부시키가이샤 | Cationic lipid |
| KR102358341B1 (en) | 2016-06-24 | 2022-02-07 | 에자이 알앤드디 매니지먼트 가부시키가이샤 | cationic lipids |
| RU2740921C2 (en) * | 2016-06-24 | 2021-01-21 | Эйсай Ар Энд Ди Менеджмент Ко., Лтд. | Cationic lipid |
| JPWO2017222016A1 (en) * | 2016-06-24 | 2019-04-18 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Cationic lipid |
| US10501416B2 (en) | 2016-06-24 | 2019-12-10 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| TWI745386B (en) * | 2016-06-24 | 2021-11-11 | 日商衛材R&D企管股份有限公司 | Cationic lipid |
| WO2018033254A2 (en) | 2016-08-19 | 2018-02-22 | Curevac Ag | Rna for cancer therapy |
| WO2018104538A1 (en) | 2016-12-08 | 2018-06-14 | Curevac Ag | Rna for treatment or prophylaxis of a liver disease |
| EP3808380A1 (en) | 2016-12-08 | 2021-04-21 | CureVac AG | Rna for treatment or prophylaxis of a liver disease |
| WO2018104540A1 (en) | 2016-12-08 | 2018-06-14 | Curevac Ag | Rnas for wound healing |
| US10526284B2 (en) | 2016-12-21 | 2020-01-07 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US10980895B2 (en) | 2016-12-21 | 2021-04-20 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| US10383952B2 (en) | 2016-12-21 | 2019-08-20 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| WO2018115527A2 (en) | 2016-12-23 | 2018-06-28 | Curevac Ag | Mers coronavirus vaccine |
| WO2018115525A1 (en) | 2016-12-23 | 2018-06-28 | Curevac Ag | Lassa virus vaccine |
| WO2018115507A2 (en) | 2016-12-23 | 2018-06-28 | Curevac Ag | Henipavirus vaccine |
| WO2018167320A1 (en) | 2017-03-17 | 2018-09-20 | Curevac Ag | Rna vaccine and immune checkpoint inhibitors for combined anticancer therapy |
| WO2018172556A1 (en) | 2017-03-24 | 2018-09-27 | Curevac Ag | Nucleic acids encoding crispr-associated proteins and uses thereof |
| US11357856B2 (en) | 2017-04-13 | 2022-06-14 | Acuitas Therapeutics, Inc. | Lipids for delivery of active agents |
| US11820728B2 (en) | 2017-04-28 | 2023-11-21 | Acuitas Therapeutics, Inc. | Carbonyl lipids and lipid nanoparticle formulations for delivery of nucleic acids |
| US11377643B2 (en) | 2017-05-31 | 2022-07-05 | Ultragenyx Pharmaceutical Inc. | Therapeutics for glycogen storage disease type III |
| US11939600B2 (en) | 2017-05-31 | 2024-03-26 | Arcturus Therapeutics, Inc. | Compositions and methods for treating phenylketonuria |
| WO2018222926A1 (en) | 2017-05-31 | 2018-12-06 | Ultragenyx Pharmaceutical Inc. | Therapeutics for glycogen storage disease type iii |
| WO2018222890A1 (en) | 2017-05-31 | 2018-12-06 | Arcturus Therapeutics, Inc. | Synthesis and structure of high potency rna therapeutics |
| EP3424524A2 (en) | 2017-07-04 | 2019-01-09 | CureVac AG | Cancer rna-vaccine |
| WO2019008001A1 (en) | 2017-07-04 | 2019-01-10 | Curevac Ag | Novel nucleic acid molecules |
| WO2019027055A1 (en) | 2017-08-04 | 2019-02-07 | 協和発酵キリン株式会社 | Nucleic-acid-containing lipid nanoparticles |
| US11542225B2 (en) | 2017-08-17 | 2023-01-03 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| US11524932B2 (en) | 2017-08-17 | 2022-12-13 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| US12065396B2 (en) | 2017-08-17 | 2024-08-20 | Acuitas Therapeutics, Inc. | Lipids for use in lipid nanoparticle formulations |
| WO2019038332A1 (en) | 2017-08-22 | 2019-02-28 | Curevac Ag | Bunyavirales vaccine |
| WO2019077001A1 (en) | 2017-10-19 | 2019-04-25 | Curevac Ag | Novel artificial nucleic acid molecules |
| WO2019092153A1 (en) | 2017-11-08 | 2019-05-16 | Curevac Ag | Rna sequence adaptation |
| WO2019115635A1 (en) | 2017-12-13 | 2019-06-20 | Curevac Ag | Flavivirus vaccine |
| US10961188B2 (en) | 2017-12-20 | 2021-03-30 | Arcturus Therapeutics, Inc. | Ionizable cationic lipid for RNA delivery |
| WO2019131580A1 (en) | 2017-12-27 | 2019-07-04 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Cationic lipid |
| JP7164547B2 (en) | 2017-12-27 | 2022-11-01 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | cationic lipid |
| US10947193B2 (en) | 2017-12-27 | 2021-03-16 | Eisai R&D Management Co., Ltd. | Cationic lipid |
| WO2019131770A1 (en) | 2017-12-27 | 2019-07-04 | 武田薬品工業株式会社 | Nucleic acid-containing lipid nano-particle and use thereof |
| JPWO2019131580A1 (en) * | 2017-12-27 | 2020-12-24 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | Cationic lipid |
| WO2019193183A2 (en) | 2018-04-05 | 2019-10-10 | Curevac Ag | Novel yellow fever nucleic acid molecules for vaccination |
| EP4227319A1 (en) | 2018-04-17 | 2023-08-16 | CureVac SE | Novel rsv rna molecules and compositions for vaccination |
| WO2020002525A1 (en) | 2018-06-27 | 2020-01-02 | Curevac Ag | Novel lassa virus rna molecules and compositions for vaccination |
| EP4218722A2 (en) | 2018-10-01 | 2023-08-02 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2020072324A1 (en) | 2018-10-01 | 2020-04-09 | Alnylam Pharmaceuticals, Inc. | Biodegradable lipids for the delivery of active agents |
| WO2020080475A1 (en) | 2018-10-18 | 2020-04-23 | 武田薬品工業株式会社 | Method for activation/proliferation of t cells |
| EP4299750A2 (en) | 2018-12-06 | 2024-01-03 | Arcturus Therapeutics, Inc. | Compositions and methods for treating ornithine transcarbamylase deficiency |
| WO2020118115A1 (en) | 2018-12-06 | 2020-06-11 | Arcturus Therapeutics, Inc. | Compositions and methods for treating ornithine transcarbamylase deficiency |
| US11685906B2 (en) | 2018-12-06 | 2023-06-27 | Arcturus Therapeutics, Inc. | Compositions and methods for treating ornithine transcarbamylase deficiency |
| WO2020123300A2 (en) | 2018-12-14 | 2020-06-18 | Eli Lilly And Company | Kras variant mrna molecules |
| WO2020128031A2 (en) | 2018-12-21 | 2020-06-25 | Curevac Ag | Rna for malaria vaccines |
| US11453639B2 (en) | 2019-01-11 | 2022-09-27 | Acuitas Therapeutics, Inc. | Lipids for lipid nanoparticle delivery of active agents |
| WO2020161342A1 (en) | 2019-02-08 | 2020-08-13 | Curevac Ag | Coding rna administered into the suprachoroidal space in the treatment of ophtalmic diseases |
| WO2020254535A1 (en) | 2019-06-18 | 2020-12-24 | Curevac Ag | Rotavirus mrna vaccine |
| WO2021028439A1 (en) | 2019-08-14 | 2021-02-18 | Curevac Ag | Rna combinations and compositions with decreased immunostimulatory properties |
| WO2021123332A1 (en) | 2019-12-20 | 2021-06-24 | Curevac Ag | Lipid nanoparticles for delivery of nucleic acids |
| DE112021000012T5 (en) | 2020-02-04 | 2021-11-18 | Curevac Ag | Coronavirus vaccine |
| DE202021004123U1 (en) | 2020-02-04 | 2022-10-26 | Curevac Ag | Coronavirus Vaccine |
| DE202021004130U1 (en) | 2020-02-04 | 2022-10-26 | Curevac Ag | Coronavirus Vaccine |
| DE202021003575U1 (en) | 2020-02-04 | 2022-01-17 | Curevac Ag | Coronavirus Vaccine |
| EP4147717A1 (en) | 2020-02-04 | 2023-03-15 | CureVac SE | Coronavirus vaccine |
| WO2021156267A1 (en) | 2020-02-04 | 2021-08-12 | Curevac Ag | Coronavirus vaccine |
| US11744887B2 (en) | 2020-03-09 | 2023-09-05 | Arcturus Therapeutics, Inc. | Coronavirus vaccine compositions and methods |
| US11759515B2 (en) | 2020-03-09 | 2023-09-19 | Arcturus Therapeutics, Inc. | Compositions and methods for inducing immune responses |
| US12070509B2 (en) | 2020-05-01 | 2024-08-27 | Arcturus Therapeutics, Inc. | Nucleic acids and methods of treatment for cystic fibrosis |
| WO2021239880A1 (en) | 2020-05-29 | 2021-12-02 | Curevac Ag | Nucleic acid based combination vaccines |
| WO2022008613A1 (en) | 2020-07-08 | 2022-01-13 | Janssen Sciences Ireland Unlimited Company | Rna replicon vaccines against hbv |
| US11976019B2 (en) | 2020-07-16 | 2024-05-07 | Acuitas Therapeutics, Inc. | Cationic lipids for use in lipid nanoparticles |
| WO2022023559A1 (en) | 2020-07-31 | 2022-02-03 | Curevac Ag | Nucleic acid encoded antibody mixtures |
| WO2022043551A2 (en) | 2020-08-31 | 2022-03-03 | Curevac Ag | Multivalent nucleic acid based coronavirus vaccines |
| WO2022135993A2 (en) | 2020-12-22 | 2022-06-30 | Curevac Ag | Pharmaceutical composition comprising lipid-based carriers encapsulating rna for multidose administration |
| WO2022137133A1 (en) | 2020-12-22 | 2022-06-30 | Curevac Ag | Rna vaccine against sars-cov-2 variants |
| WO2022146654A1 (en) | 2020-12-28 | 2022-07-07 | Janssen Pharmaceuticals, Inc. | Transcription activator-like effector nucleases (talens) targeting hbv |
| WO2022162027A2 (en) | 2021-01-27 | 2022-08-04 | Curevac Ag | Method of reducing the immunostimulatory properties of in vitro transcribed rna |
| WO2022168884A1 (en) | 2021-02-04 | 2022-08-11 | 塩野義製薬株式会社 | Cationic lipid |
| WO2022200575A1 (en) | 2021-03-26 | 2022-09-29 | Glaxosmithkline Biologicals Sa | Immunogenic compositions |
| WO2022207862A2 (en) | 2021-03-31 | 2022-10-06 | Curevac Ag | Syringes containing pharmaceutical compositions comprising rna |
| WO2022215758A1 (en) | 2021-04-09 | 2022-10-13 | 相互薬工株式会社 | Lipid and composition |
| WO2022233880A1 (en) | 2021-05-03 | 2022-11-10 | Curevac Ag | Improved nucleic acid sequence for cell type specific expression |
| WO2023006999A2 (en) | 2021-07-30 | 2023-02-02 | CureVac SE | Mrnas for treatment or prophylaxis of liver diseases |
| WO2023031394A1 (en) | 2021-09-03 | 2023-03-09 | CureVac SE | Novel lipid nanoparticles for delivery of nucleic acids |
| WO2023031392A2 (en) | 2021-09-03 | 2023-03-09 | CureVac SE | Novel lipid nanoparticles for delivery of nucleic acids comprising phosphatidylserine |
| WO2023073228A1 (en) | 2021-10-29 | 2023-05-04 | CureVac SE | Improved circular rna for expressing therapeutic proteins |
| WO2023121975A1 (en) * | 2021-12-20 | 2023-06-29 | Beam Therapeutics Inc. | Ionizable amine lipids and lipid nanoparticles |
| WO2023144193A1 (en) | 2022-01-25 | 2023-08-03 | CureVac SE | Mrnas for treatment of hereditary tyrosinemia type i |
| WO2023144330A1 (en) | 2022-01-28 | 2023-08-03 | CureVac SE | Nucleic acid encoded transcription factor inhibitors |
| WO2023218420A1 (en) | 2022-05-13 | 2023-11-16 | Janssen Pharmaceuticals, Inc. | Mrna compositions for inducing latent hiv-1 reversal |
| WO2023227608A1 (en) | 2022-05-25 | 2023-11-30 | Glaxosmithkline Biologicals Sa | Nucleic acid based vaccine encoding an escherichia coli fimh antigenic polypeptide |
| WO2024026308A2 (en) | 2022-07-29 | 2024-02-01 | Massachusetts Institute Of Technology | COMPOSITIONS AND METHODS FOR INHIBITING EXPRESSION OF THE SIGNAL REGULATORY PROTEIN ALPHA (SIRPα) GENE |
| WO2024068545A1 (en) | 2022-09-26 | 2024-04-04 | Glaxosmithkline Biologicals Sa | Influenza virus vaccines |
| DE202023106198U1 (en) | 2022-10-28 | 2024-03-21 | CureVac SE | Nucleic acid-based vaccine |
| WO2024121814A1 (en) | 2022-12-09 | 2024-06-13 | Takeda Pharmaceutical Company Limited | Modified immunomodulators |
| EP4385523A1 (en) | 2022-12-14 | 2024-06-19 | Beijing Jitai Pharmaceutical Technology Co., Ltd. | Lipid-based topical injection formulations |
| WO2024160936A1 (en) | 2023-02-03 | 2024-08-08 | Glaxosmithkline Biologicals Sa | Rna formulation |
| WO2024168010A2 (en) | 2023-02-09 | 2024-08-15 | Alnylam Pharmaceuticals, Inc. | Reversir molecules and methods of use thereof |
| WO2024171052A1 (en) | 2023-02-14 | 2024-08-22 | Glaxosmithkline Biologicals Sa | Analytical method |
| WO2024184500A1 (en) | 2023-03-08 | 2024-09-12 | CureVac SE | Novel lipid nanoparticle formulations for delivery of nucleic acids |
| WO2024200823A1 (en) | 2023-03-30 | 2024-10-03 | Ose Immunotherapeutics | Lipid-based nanoparticle targeted at activated immune cells for the expression of immune cell enhancing molecule and use thereof |
| WO2024200826A1 (en) | 2023-03-30 | 2024-10-03 | Ose Immunotherapeutics | Lipid-based nanoparticle targeted at activated immune cells for the expression of immune cell inhibiting molecule and use thereof |
| WO2024200820A1 (en) | 2023-03-30 | 2024-10-03 | Ose Immunotherapeutics | Method of synthesis of targeted lipid nanoparticle and uses thereof |
| WO2024210160A1 (en) | 2023-04-07 | 2024-10-10 | Takeda Pharmaceutical Company Limited | Conjugation complex |
| GB202404607D0 (en) | 2024-03-29 | 2024-05-15 | Glaxosmithkline Biologicals Sa | RNA formulation |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2022204146B2 (en) | Biodegradable lipids for the delivery of active agents | |
| US10653780B2 (en) | Amino lipids and methods for the delivery of nucleic acids | |
| AU2015210364B2 (en) | Improved amino lipids and methods for the delivery of nucleic acids | |
| CA2532228C (en) | Lipid encapsulated interfering rna | |
| WO2010048536A2 (en) | Processes for preparing lipids | |
| AU2010208035A1 (en) | Improved lipid formulation for the delivery of nucleic acids | |
| WO2011036557A1 (en) | Compositions and methods for enhancing cellular uptake and intracellular delivery of lipid particles | |
| EA039892B1 (en) | Biodegradable lipids for the delivery of active agents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180037927.7 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11726579 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2800401 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2586/MUMNP/2012 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 223207 Country of ref document: IL Ref document number: 201291100 Country of ref document: EA |
|
| REEP | Request for entry into the european phase |
Ref document number: 2011726579 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011726579 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2013513394 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2012/014079 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 2011261247 Country of ref document: AU Date of ref document: 20110603 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20137000092 Country of ref document: KR Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11726579 Country of ref document: EP Kind code of ref document: A2 |