WO2011121593A1 - Stable pharmaceutical composition of imatinib - Google Patents
Stable pharmaceutical composition of imatinib Download PDFInfo
- Publication number
- WO2011121593A1 WO2011121593A1 PCT/IN2010/000202 IN2010000202W WO2011121593A1 WO 2011121593 A1 WO2011121593 A1 WO 2011121593A1 IN 2010000202 W IN2010000202 W IN 2010000202W WO 2011121593 A1 WO2011121593 A1 WO 2011121593A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- film coated
- coated tablet
- imatinib
- tablet according
- pharmaceutically acceptable
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4965—Non-condensed pyrazines
- A61K31/497—Non-condensed pyrazines containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
- A61K9/2018—Sugars, or sugar alcohols, e.g. lactose, mannitol; Derivatives thereof, e.g. polysorbates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2833—Organic macromolecular compounds
- A61K9/286—Polysaccharides, e.g. gums; Cyclodextrin
- A61K9/2866—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
Abstract
The present invention relates to a compressed film-coated tablet comprising imatinib its pharmaceutically acceptable salts there of in an amount of more than 80 % based on the total weight of the finished dosage form.
Description
STABLE PHARMACEUTICAL COMPOSITION OF IMATINIB
Field of the Invention
The present invention relates to a compressed film-coated tablet comprising imatinib its pharmaceutically acceptable salts there of in an amount of more than 80% based on the total weight of the finished dosage form and a film coating on said tablet core is applied in an amount of 1 to 2 % w/w of the tablet.
Background of the Invention
Imatinib mesylate is chemically 4-[(4-Methyl l-piperazinyl)methyl]-N-[4- methyl-3-[[4-(3-pyridinyl)-2-pyrimidinyl]amino]-phenyl]benzamide methanesulfonate and is used to treat chronic myelogenous leukemia (CML), gastrointestinal stromal tumors (GISTs) and other cancers. In this document the term "imatinib" will refer to the mono methane sulfonate salt of imatinib unless otherwise specified.
Imatinib mesylate is a protein-tyrosine kinase inhibitor that inhibits the bcr-abl tyrosine kinase, the constitutive abnormal tyrosine kinase created by the Philadelphia chromosome abnormality in CML. Imatinib inhibits proliferation and induces apoptosis in bcr-abl positive cell lines as well as fresh leukemic cells from Philadelphia chromosome positive chronic myeloid leukemia. Imatinib inhibits colony formation in assays using ex vivo peripheral blood and bone marrow samples from CML patients.
US patent 6894051 discloses the novel crystalline form of the mono methane sulfonic acid addition salt of imatinib and its preparation process and designated the form as β-crystalline form.
US patent 6958335 describes the use of imatinib or a pharmaceutically acceptable salt thereof for the manufacture of pharmaceutical compositions for use in the treatment of gastrointestinal stromal tumours.
US patent 7544799 discloses a particular form of methane sulfonic acid addition salt of imatinib.
PCT application WO 03/090720 describes a tablet comprising a pharmacologically effective amount of imatinib or a pharmaceutically acceptable salt
thereof in an amount from about 30% to 80% in weight of the active moiety based on the total weight of tablet and pharmaceutically acceptable excipient.
PCT application WO 01/47507 describes a pharmaceutical composition comprising imatinib mesylate in an amount of 22% W/W.
US patent application US 2009/0087489 describes a pharmaceutical composition, preferably tablet, containing about 23-29% W/W of imatinib mesylate which were prepared using dry granulation or direct compaction.
As it is a high drug loaded composition, the present inventors have come across several problems in preparing the tablets of imatinib with such a low quantity of excipients. Thus, still there is a need for commercially acceptable high drug load compositions of imatinib.
Surprisingly, the present inventors have obtained a film coated tablet with high drug load of imatinib or pharmaceutical acceptable salts thereof and the tablet is film coated with opadry brown/yellow having poly vinyl alcohol as main ingredient.
Objective of the Invention
Accordingly, the main objective of the invention is to provide a film coated tablet comprising imatinib or pharmaceutically acceptable salts thereof in an amount of more than 80% based on the total weight of the coated tablet and a film coating comprising poly vinyl alcohol applied to the tablet core in an amount of 1 to 2 % w/w of the tablet.
Yet another objective of the present invention is to provide a process for the preparation of stable film coated tablet of imatinib or its pharmaceutically acceptable salts. Summary of the Invention
Accordingly, the main embodiment of the present invention is to provide a film coated tablet comprising imatinib or pharmaceutically acceptable salts thereof in an amount of more than 80% based on the total weight of the coated tablet and a film coating comprising poly vinyl alcohol applied to the tablet core in an amount of 1 to 2 % w/w of the tablet.
Detailed Description of the Invention
According to the present invention there is provided a film coated tablet comprising imatinib or its pharmaceutically acceptable salts in an amount of more than 80% based on the total weight of the coated tablet and a film coating comprising poly vinyl alcohol applied to the tablet core in an amount of 1 to 2 % w/w of the tablet.
Preferably, the present invention containing imatinib or pharmaceutically acceptable salts thereof in an amount of about 85 to 98% based on the total weight of the composition.
More preferably, the present invention containing imatinib or pharmaceutically acceptable salts thereof in an amount of about 90 to 97% based on the total weight of the composition.
The film coating, in addition to polyvinyl alcohol may consists of other ingredients such as titanium dioxide, talc, lecithin (soy), black iron oxide, polyethylene glycol and red iron oxide/yellow iron oxide or mixtures thereof.
Preferably, imatinib is in the mono mesylate form.
In another aspect, the present invention provides a film coated tablet wherein the mono mesylate salt of imatinib is in crystalline form or amorphous form.
Preferably, the mono mesylate salt of imatinib is in amorphous, alpha or beta crystalline form.
More preferably, the mono mesylate salt of imatinib is in alpha or beta crystalline form.
Still more preferably, the mono mesylate salt of imatinib is in alpha crystalline form.
Preferably, the film coating applied to the tablet core is in an amount of 1.3 to 1.6 % w/w of the tablet.
The film coated tablet composition of the invention may contain one or more additional excipients. These excipients may be selected from binders and lubricants.
Preferably, the lubricant is selected from sodium stearyl fumarate, magnesium stearate, zinc stearate, calcium stearate, stearic acid, talc, Glyceryl behenate and collidal silicon dioxide.
More preferably, the lubricant is selected from magnesium stearate and colloidal silicon dioxide.
In a preferred embodiment of the present invention, the lubricant present in an amount of 0.8-2.5%w/w based on the total weight of the tablet.
The preferable binder is selected from L-Hydroxy propyl cellulose, polyvinyl pyrrolidine, hydroxyl propyl methyl cellulose, hydroxyl ethyl cellulose and pre- gelatinized starch.
More preferable binder is selected from L-Hydroxy propyl cellulose and pre- gelatinized starch.
The film coated tablet composition of imatinib or its pharmaceutically acceptable salts may be prepared by direct compression, wet granulation or roll compaction.
Preferably, the film coated tablet of imatinib or pharmaceutically acceptable salts thereof may be prepared by wet granulation.
The wet granulation process may be carried out using the solvent selected from isopropyl alcohol, ethanol, mixture of isopropyl alcohol and water and mixture of ethanol and water.
Preferably, the wet granulation process may be carried out using isopropyl alcohol.
The present invention also provides a process for the preparation of film coated tablet composition of imatinib or pharmaceutically acceptable salts thereof comprising: i) Granulating imatinib or its pharmaceutically acceptable salts with binder solution.
ii) Drying the granules and then lubricating the dried granules
iii) Compressing the blended granules of step (ii) to obtained tablets.
iv) Coating the tablets of step (iii) with the coating suspension comprising polyvinyl alcohol.
The present invention also provides a process for the preparation of stable pharmaceutical composition of imatinib or its pharmaceutically acceptable salts comprising:
i) Compacting Imatinib or its pharmaceutically acceptable salts, optionally along with one pharmaceutically acceptable excipient.
ii) Lubricating the compact of step (i) with atleast one lubricant.
iii) Compressing the blend of step (ii) to obtained tablets.
iv) Coating the tablets of step (iii) coating suspension comprising polyvinyl alcohol.
In one of the embodiment, the film coating may be carried out with 12% W/W suspension of opadry brown/yellow in water.
The film coating, in addition to polyvinyl alcohol may comprises other ingredients such as titanium dioxide, talc, lecithin (soy), black iron oxide, polyethylene glycol and red iron oxide/yellow iron oxide or mixtures thereof.
The coating bed temperature is in the range of 40-45°C.
The film coated tablet composition of the present invention comprising about 100 to 400 mg of imatinib or its pharmaceutically acceptable salts.
The following examples further exemplify the invention and are not intended to limit the scope of the invention.
Example 1:
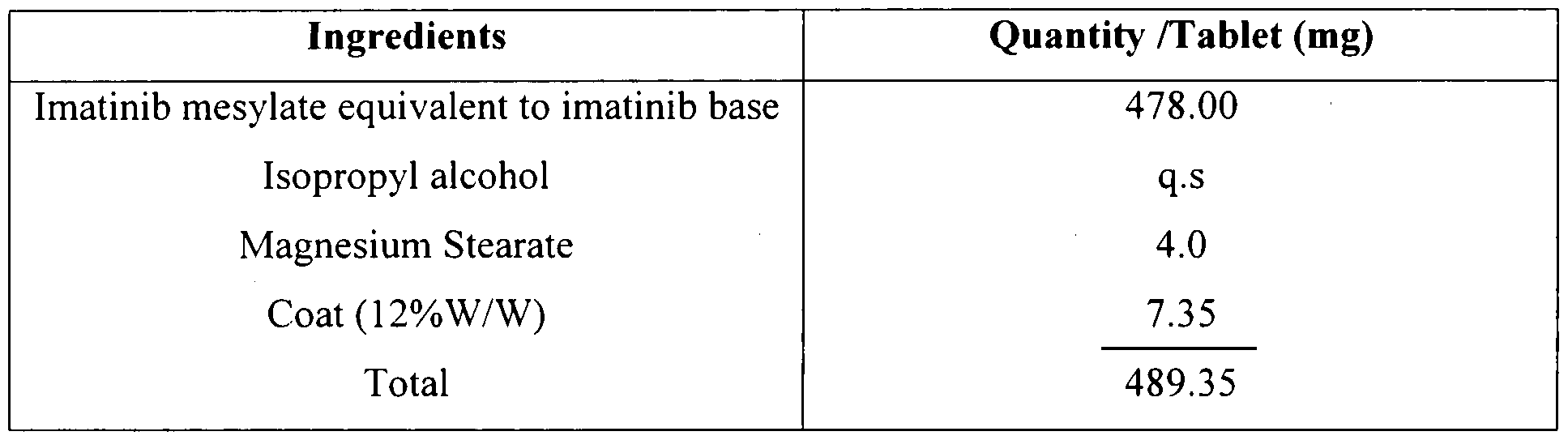
The process steps involved in the preparation of Imatinib tablets are given below: i) Granulated imatinib with binder solution i.e., isopropyl alcohol and dried the granulated mass
ii) Lubricated the dried granules with magnesium stearate
iii) Compressed the lubricated blend of step (ii) into tablets.
iv) Coated the tablets of step (iii) with 12% solution of Opadry brown/yell water.
Example 2;
The process steps involved in the preparation of Imatinib tablets are given below: i) Granulated imatinib with binder solution i.e., isopropyl alcohol and dried the granulated mass
ii) Lubricated the dried granules with magnesium stearate
iii) Compressed the lubricated blend of step (ii) into tablets.
iv) Coated the tablets of step (iii) with 12% solution of Opadry brown/yellow in water.
Example 3:
The process steps involved in the preparation of Imatinib tablets are given below: i) Granulated imatinib with binder solution i.e., isopropyl alcohol and dried the granulated mass
ii) Lubricated the dried granules with magnesium stearate
iii) Compressed the lubricated blend of step (ii) into tablets.
iv) Coated the tablets of step (iii) with 12% solution of Opadry brown/yellow in water.
Example 4:
The process steps involved in the preparation of Imatinib tablets are given below:
i) Imatinib, L-Hydroxy propyl cellulose and 50% of the magnesium stearate were subjected to roller compaction.
ii) Compacted blend was lubricated with remaining 50% of magnesium stearate iii) Compressed the lubricated blend of step (ii) into tablets.
iv) Coated the tablets of step (iii) with 12% solution of Opadry brown/yellow in water.
Example 5:
The process steps involved in the preparation of Imatinib tablets are given below:
i) Imatinib was subjected to roller compaction.
ii) Compacted imatinib was blended with colloidal silicon dioxide and mag stearate.
iii) Compressed the blend of step (ii) into tablets.
iv) Coated the tablets of step (iii) with 12% solution of Opadry brown/yellow in water.
Claims
1 . A film coated tablet comprising imatinib or pharmaceutically acceptable salts thereof in an amount of more than 80% based on the total weight of the coated tablet and a film coating comprising poly vinyl alcohol applied to the tablet core in an amount of 1 to 2 % w/w of the tablet.
2. A film coated tablet according to claim 1 wherein imatinib or pharmaceutically acceptable salt there of is present in an amount of 85-98% based on the total weight of the dosage form.
3. A film coated tablet according to claim 1 wherein imatinib or pharmaceutically acceptable salt there of is present in an amount of 90-97%) based on the total weight of the dosage form.
4. A film coated tablet according to claim 1 wherein the film coating, in addition to polyvinyl alcohol may consists of other ingredients such as titanium dioxide, talc, lecithin (soy), black iron oxide, polyethylene glycol and red iron oxide/yellow iron oxide or mixtures thereof.
5. A film coated tablet according to claim 1 wherein the imatinib is in the mono mesylate form.
6. A film coated tablet according to claim 1 wherein the mono mesylate salt of imatinib is in amorphous or crystalline form.
7. A film coated tablet according to claim 1 wherein the mono mesylate salt of imatinib is in amorphous, alpha or beta crystalline form.
8. A film coated tablet according to claim 1 wherein the mono mesylate salt of imatinib is in alpha or beta crystalline form.
9. A film coated tablet according to claim 1 wherein the mono mesylate salt of imatinib is in alpha crystalline form.
10. A film coated tablet according to claim 1 wherein the film coating applied to the tablet core is in an amount of from 1.3 to 1.6 % w/w of the tablet.
1 1. A film coated tablet according to claim 1 further comprises one or more pharmaceutically acceptable excipients.
12. A film coated tablet according to claim 1 1 wherein the pharmaceutically acceptable excipients are selected from binders and lubricants.
13. A film coated tablet to claim 1 1 wherein the lubricant is selected from sodium stearyl fumarate, magnesium stearate, zinc stearate, calcium stearate, stearic acid, talc, Glyceryl behenate and colloidal silicon dioxide.
14. A film coated tablet according to claim 1 1 wherein the lubricant is magnesium stearate and colloidal silicon dioxide.
15. A film coated tablet according to claim 1 1 wherein the lubricant is in an amount of 0.8 to 2.5% w/w based on the total weight of the tablet.
16. A film coated tablet according to claim 1 1 wherein the binder is selected from L- Hydroxy propyl cellulose, polyvinyl pyrrolidine, hydroxyl propyl methyl cellulose, hydroxyl ethyl cellulose and pre-gelatinized starch.
17. A film coated tablet according to claim 1 1 wherein the binder is selected from L- Hydroxy propyl cellulose and pre-gelatinized starch.
18. A film coated tablet according to any one of the preceding claims prepared by wet granulation or roller compaction.
19. A film coated tablet according to claims 18 wherein the wet granulation is carried out using solvents selected from isopropyl alcohol, ethanol, mixture of isopropyl alcohol and water and mixture of ethanol and water.
20. A film coated tablet according to claims 18 wherein the wet granulation is carried out using isopropyl alcohol.
21. A process for the preparation of a stable solid oral composition according to any one of the preceeding claims, comprises
i) . Granulating imatinib or its pharmaceutically acceptable salts with binder solution.
ii) . Drying the granules and then lubricating the dried granules iii). Compressing the blended granules of step (ii) to obtained tablets.
22. A process for the preparation of a stable solid oral composition according to any one of the preceeding claims, comprises
i). Compacting Imatinib or its pharmaceutically acceptable salts, optionally along with atleast one pharmaceutically acceptable excipient. ii). Lubricating the compact of step (i) with atleast one lubricant iii) Compressing the blend of step (ii) to obtained tablets.
23. A film coated tablet according to any one of the preceding claims wherein the tablet is film coated with 12% coating suspension in water.
24. A film coated tablet according to claim 1 wherein the composition comprises about 100 to 400 mg of imatinib or its pharmaceutically acceptable salts.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/637,165 US20130011477A1 (en) | 2010-03-29 | 2010-03-29 | Stable Pharmaceutical Composition of Imatinib |
| EP10848804.0A EP2552447A4 (en) | 2010-03-29 | 2010-03-29 | Stable pharmaceutical composition of imatinib |
| PCT/IN2010/000202 WO2011121593A1 (en) | 2010-03-29 | 2010-03-29 | Stable pharmaceutical composition of imatinib |
| CA2789307A CA2789307A1 (en) | 2010-03-29 | 2010-03-29 | Stable pharmaceutical composition of imatinib |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/IN2010/000202 WO2011121593A1 (en) | 2010-03-29 | 2010-03-29 | Stable pharmaceutical composition of imatinib |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011121593A1 true WO2011121593A1 (en) | 2011-10-06 |
Family
ID=44711420
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2010/000202 WO2011121593A1 (en) | 2010-03-29 | 2010-03-29 | Stable pharmaceutical composition of imatinib |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20130011477A1 (en) |
| EP (1) | EP2552447A4 (en) |
| CA (1) | CA2789307A1 (en) |
| WO (1) | WO2011121593A1 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102349875A (en) * | 2011-10-11 | 2012-02-15 | 浙江华海药业股份有限公司 | Preparation method of methylsulfonic acid imatinib tablet |
| WO2012019633A1 (en) * | 2010-08-11 | 2012-02-16 | Synthon B.V. | Pharmaceutical granulate comprising imatinib mesylate |
| EP2497464A3 (en) * | 2011-03-09 | 2012-09-19 | Adamed SP. Z O.O. | Pharmaceutical composition of imatinibe methanesulphonate and a process for its manufacture |
| WO2012176014A1 (en) * | 2011-06-22 | 2012-12-27 | Natco Pharma Limited | Imatinib mesylate oral pharmaceutical composition and process for preparation thereof |
| WO2013124774A1 (en) | 2012-02-21 | 2013-08-29 | Ranbaxy Laboratories Limited | Stable dosage forms of imatinib mesylate |
| JP2013245202A (en) * | 2012-05-28 | 2013-12-09 | Nipro Corp | Pharmaceutical composition |
| EP2803353A1 (en) | 2013-05-14 | 2014-11-19 | Hetero Research Foundation | Compositions of Imatinib |
| CN105311635A (en) * | 2014-06-16 | 2016-02-10 | 万菱药品科技股份有限公司 | High drug-loading pharmaceutical composition with adjustable release rate and preparation method thereof |
| EP3019159A4 (en) * | 2013-07-09 | 2017-01-18 | Shilpa Medicare Limited | Oral pharmaceutical compositions comprising imatinib mesylate |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB201304699D0 (en) * | 2013-03-15 | 2013-05-01 | Remedica Ltd | Pharmaceutical compositions |
| CA2937365C (en) | 2016-03-29 | 2018-09-18 | F. Hoffmann-La Roche Ag | Granulate formulation of 5-methyl-1-phenyl-2-(1h)-pyridone and method of making the same |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5785994A (en) * | 1990-08-31 | 1998-07-28 | Alza Corporation | Method for administering drug to gastrointestinal tract |
| US20070265288A1 (en) * | 2004-09-02 | 2007-11-15 | Pathi Srinivas L | Stable Crystal Form of Imatinib Mesylate and Process for the Preparation Thereof |
| US20080226731A1 (en) * | 2005-05-10 | 2008-09-18 | Madhav Vasanthavada | Pharmaceutical Compositions Comprising I Matinib and a Release Retardant |
| US20090238884A1 (en) * | 2008-03-21 | 2009-09-24 | Elan Pharma International Limited | Compositions for site-specific delivery of imatinib and methods of use |
| US20090264438A1 (en) * | 2006-04-27 | 2009-10-22 | Alexandr Jegorov | Polymorphic forms of imatinib mesylate and processes for preparation of novel crystalline forms as well as amorphous and form alpha |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060084628A1 (en) * | 2004-10-19 | 2006-04-20 | Achillion Pharmaceuticals | Combination therapy for treating viral infections |
| MX2007004294A (en) * | 2005-07-26 | 2008-03-11 | Ucb Pharma Sa | Pharmaceutical compositions comprising levetiracetam and process for their preparation. |
| ATE445392T1 (en) * | 2005-08-15 | 2009-10-15 | Siegfried Generics Int Ag | FILM TABLET OR GRANULES CONTAINING A PYRIDYLPYRIMIDINE |
-
2010
- 2010-03-29 CA CA2789307A patent/CA2789307A1/en not_active Abandoned
- 2010-03-29 EP EP10848804.0A patent/EP2552447A4/en not_active Withdrawn
- 2010-03-29 WO PCT/IN2010/000202 patent/WO2011121593A1/en active Application Filing
- 2010-03-29 US US13/637,165 patent/US20130011477A1/en not_active Abandoned
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5785994A (en) * | 1990-08-31 | 1998-07-28 | Alza Corporation | Method for administering drug to gastrointestinal tract |
| US20070265288A1 (en) * | 2004-09-02 | 2007-11-15 | Pathi Srinivas L | Stable Crystal Form of Imatinib Mesylate and Process for the Preparation Thereof |
| US20080226731A1 (en) * | 2005-05-10 | 2008-09-18 | Madhav Vasanthavada | Pharmaceutical Compositions Comprising I Matinib and a Release Retardant |
| US20090264438A1 (en) * | 2006-04-27 | 2009-10-22 | Alexandr Jegorov | Polymorphic forms of imatinib mesylate and processes for preparation of novel crystalline forms as well as amorphous and form alpha |
| US20090238884A1 (en) * | 2008-03-21 | 2009-09-24 | Elan Pharma International Limited | Compositions for site-specific delivery of imatinib and methods of use |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2552447A4 * |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012019633A1 (en) * | 2010-08-11 | 2012-02-16 | Synthon B.V. | Pharmaceutical granulate comprising imatinib mesylate |
| EP2497464A3 (en) * | 2011-03-09 | 2012-09-19 | Adamed SP. Z O.O. | Pharmaceutical composition of imatinibe methanesulphonate and a process for its manufacture |
| WO2012176014A1 (en) * | 2011-06-22 | 2012-12-27 | Natco Pharma Limited | Imatinib mesylate oral pharmaceutical composition and process for preparation thereof |
| US9750700B2 (en) | 2011-06-22 | 2017-09-05 | Natco Pharma Limited | Imatinib mesylate oral pharmaceutical composition and process for preparation thereof |
| CN102349875A (en) * | 2011-10-11 | 2012-02-15 | 浙江华海药业股份有限公司 | Preparation method of methylsulfonic acid imatinib tablet |
| WO2013124774A1 (en) | 2012-02-21 | 2013-08-29 | Ranbaxy Laboratories Limited | Stable dosage forms of imatinib mesylate |
| JP2013245202A (en) * | 2012-05-28 | 2013-12-09 | Nipro Corp | Pharmaceutical composition |
| EP2803353A1 (en) | 2013-05-14 | 2014-11-19 | Hetero Research Foundation | Compositions of Imatinib |
| EP3019159A4 (en) * | 2013-07-09 | 2017-01-18 | Shilpa Medicare Limited | Oral pharmaceutical compositions comprising imatinib mesylate |
| CN105311635A (en) * | 2014-06-16 | 2016-02-10 | 万菱药品科技股份有限公司 | High drug-loading pharmaceutical composition with adjustable release rate and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| US20130011477A1 (en) | 2013-01-10 |
| EP2552447A4 (en) | 2013-09-11 |
| CA2789307A1 (en) | 2011-10-06 |
| EP2552447A1 (en) | 2013-02-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20130011477A1 (en) | Stable Pharmaceutical Composition of Imatinib | |
| JP6682522B2 (en) | Crystal form of 2-((4S) -6- (4-chlorophenyl) -1-methyl-4H-benzo [C] isoxazolo [4,5-E] azepin-4-yl) acetamide | |
| US20130122093A1 (en) | Formulations of a src/abl inhibitor | |
| EP2582362A1 (en) | Pharmaceutical compositions comprising imatinib or pharmaceutically acceptable salt thereof and processes for the manufacture thereof | |
| EP3860605A1 (en) | Crystalline forms and processes of lenvatinib besylate | |
| EP3606511A1 (en) | Pharmaceutical composition comprising lenvatinib mesylate | |
| EP2497464A2 (en) | Pharmaceutical composition of imatinibe methanesulphonate and a process for its manufacture | |
| EP3860606B1 (en) | Pharmaceutical composition comprising lenvatinib esylate or tosylate | |
| TWI720115B (en) | Preparation method of a pharmaceutical composition comprising quinoline derivatives or salt thereof | |
| WO2011161689A1 (en) | Imatinib mesilate pharmaceutical tablet | |
| KR101739731B1 (en) | Pharmaceutical composition containing gefitinib | |
| AU2014288866B2 (en) | Oral pharmaceutical compositions comprising Imatinib mesylate | |
| KR20180103089A (en) | A pharmaceutical composition comprising a quinoline derivative or a salt thereof | |
| JP2020147542A (en) | Multilayer tablet comprising dabigatran etexilate or pharmaceutically acceptable salt thereof | |
| EP2803353B1 (en) | Compositions of Imatinib | |
| US20150297603A1 (en) | HIGH DRUG LOAD TABLET FORMULATION OF [(1R), 2S]-2-AMINOPROPIONIC ACID 2-[4-(4-FLUORO-2-METHYL-1H-INDOL-5-YLOXY)-5-METHYLPYRROLO[2,1-f][1,2,4]TRIAZIN-6-YLOXY]-1-METHYLETHYL ESTER | |
| JP6739275B2 (en) | Pharmaceutical composition containing gefitinib as an active ingredient | |
| TW202342040A (en) | Pharmaceutical formulation | |
| WO2017078647A1 (en) | Pharmaceutical compositions of imatinib | |
| EP2968175A1 (en) | Pharmaceutical compositions comprising imatinib | |
| EA017781B1 (en) | Film-coated tablet containing imatinib mesylate and method for preparing same | |
| JP2019189554A (en) | Pharmaceutical composition with improved dissolution | |
| WO2013139826A1 (en) | Pharmaceutical compositions comprising imatinib |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10848804 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2789307 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13637165 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010848804 Country of ref document: EP |