WO2011050476A1 - Methods for reprogramming cells and uses thereof - Google Patents
Methods for reprogramming cells and uses thereof Download PDFInfo
- Publication number
- WO2011050476A1 WO2011050476A1 PCT/CA2010/001727 CA2010001727W WO2011050476A1 WO 2011050476 A1 WO2011050476 A1 WO 2011050476A1 CA 2010001727 W CA2010001727 W CA 2010001727W WO 2011050476 A1 WO2011050476 A1 WO 2011050476A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cell
- cells
- expression
- nslc
- stem
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0662—Stem cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/30—Nerves; Brain; Eyes; Corneal cells; Cerebrospinal fluid; Neuronal stem cells; Neuronal precursor cells; Glial cells; Oligodendrocytes; Schwann cells; Astroglia; Astrocytes; Choroid plexus; Spinal cord tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/16—Otologicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0618—Cells of the nervous system
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0618—Cells of the nervous system
- C12N5/0619—Neurons
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0618—Cells of the nervous system
- C12N5/0623—Stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0647—Haematopoietic stem cells; Uncommitted or multipotent progenitors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0656—Adult fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0657—Cardiomyocytes; Heart cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0662—Stem cells
- C12N5/0667—Adipose-derived stem cells [ADSC]; Adipose stromal stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0662—Stem cells
- C12N5/0668—Mesenchymal stem cells from other natural sources
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0696—Artificially induced pluripotent stem cells, e.g. iPS
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/48—Reproductive organs
- A61K35/54—Ovaries; Ova; Ovules; Embryos; Foetal cells; Germ cells
- A61K35/545—Embryonic stem cells; Pluripotent stem cells; Induced pluripotent stem cells; Uncharacterised stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
- C12N2500/10—Metals; Metal chelators
- C12N2500/20—Transition metals
- C12N2500/24—Iron; Fe chelators; Transferrin
- C12N2500/25—Insulin-transferrin; Insulin-transferrin-selenium
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/06—Anti-neoplasic drugs, anti-retroviral drugs, e.g. azacytidine, cyclophosphamide
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/065—Modulators of histone acetylation
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/105—Insulin-like growth factors [IGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/11—Epidermal growth factor [EGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/115—Basic fibroblast growth factor (bFGF, FGF-2)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/13—Nerve growth factor [NGF]; Brain-derived neurotrophic factor [BDNF]; Cilliary neurotrophic factor [CNTF]; Glial-derived neurotrophic factor [GDNF]; Neurotrophins [NT]; Neuregulins
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/155—Bone morphogenic proteins [BMP]; Osteogenins; Osteogenic factor; Bone inducing factor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/16—Activin; Inhibin; Mullerian inhibiting substance
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/30—Hormones
- C12N2501/38—Hormones with nuclear receptors
- C12N2501/395—Thyroid hormones
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/60—Transcription factors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/60—Transcription factors
- C12N2501/602—Sox-2
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/60—Transcription factors
- C12N2501/604—Klf-4
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/70—Enzymes
- C12N2501/72—Transferases [EC 2.]
- C12N2501/727—Kinases (EC 2.7.)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/998—Proteins not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/09—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from epidermal cells, from skin cells, from oral mucosa cells
- C12N2506/094—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from epidermal cells, from skin cells, from oral mucosa cells from keratinocytes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/11—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from blood or immune system cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/13—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells
- C12N2506/1307—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells from adult fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/13—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells
- C12N2506/1346—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells from mesenchymal stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/13—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells
- C12N2506/1346—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells from mesenchymal stem cells
- C12N2506/1384—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells from mesenchymal stem cells from adipose-derived stem cells [ADSC], from adipose stromal stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2510/00—Genetically modified cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2513/00—3D culture
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0676—Pancreatic cells
Definitions
- the present invention relates to the field of eukaryotic cell reprogramming, and particularly to cell dedifferentiation.
- the invention is also concerned with methods of generating stable Neural Stem-Like Cells (NSLCs) from human somatic cells (and other cells) and the use of the cells so generated in human therapy.
- NSLCs Neural Stem-Like Cells
- a stem cell can naturally divide or differentiate into another stem cell, progenitor, precursor, or somatic cell.
- somatic cell can sometimes transiently change its phenotype or express certain markers when placed in certain conditions, and then revert back when placed back into the original conditions.
- the phenotype of many cells can be changed through forced expression of certain genes (for example, stably transfecting the c-myc gene into fibroblasts turns them into immortal cells having neuroprogenitor characteristics), however once this forced gene expression is removed, the cells slowly revert back to their original state.
- the first is considered natural differentiation which is part of a cell program that is already in place (going from a more undifferentiated to a more differentiated state)
- the second is a transient phenotypical change
- the third is a constantly forced cell type.
- a true stem cell (i) self-renews almost 'indefinitely' (for significantly longer than a somatic cell), (ii) is not a cancerous cell, (iii) is not artificially maintained by forced gene expression or similar means (must also be able to be maintained in standard stem cell media), (iv) can differentiate to progenitor, precursor, somatic or other more differentiated cell type (of the same lineage), and (v) has all the characteristics of a stem cell and not just certain markers or gene expression or morphological appearance.
- Bhasin (WO2010/088735), Cifarelli et al. (US2010/0003223), Kremer et al. (US2004/0009595), and Winnier et al. (US2010/0047908) all refer to reprogramming, dedifferentiation, and/or obtained stem cells (or progenitors) as phenotypical cell changes based only on a change in cell surface markers after culture in different media with supplements, with no evidence of true reprogramming or an actual stem cell (non-cancerous self-renewal with stem cells markers and no differentiation markers). The same is true for Benneti (WO2009/079007) who used increased expression of Oct4 and Sox2. Others, such as Akamatsu et al.
- iPS cells induced pluripotent stem cells
- Yamanaka's group Yamanaka et al., 2007
- Thomson's group Yamanaka et al., 2007
- These cells can be induced by true reprogramming since it was later shown that they can also be induced by non-gene integrating transient transfection (Soldner et al., 2009; Woltjen et al., 2009; Yu et al., 2009) as well as by RNA (Warren et al., 2010) or protein (Kim et al., 2009; Zhou et al., 2009) alone or by small molecules (Lyssiotis et al., 2009), and by similar methods.
- these cells are essentially identical to embryonic stem cells and have the same problems of uncontrolled growth, teratoma formation, and potential tumor formation.
- a more desirable option is to have multipotent stem cells or pluripotent-like cells whose lineage and differentiation potential is more restricted so that they do not readily form teratomas and uncontrolled growth.
- NSC Neural stem-like cells
- Neural stem cells have promise for tissue regeneration from disease or injury; however, such therapies will require precise control over cell function to create the necessary cell types. There is not yet a complete understanding of the mechanisms that regulate cell proliferation and differentiation, and it is thus difficult to fully explore the plasticity of neural stem cell population derived from any given region of the brain or developing fetus.
- the CNS traditionally believed to have limited regenerative capabilities, retains a limited number of neural stem cells in adulthood, particularly in the dentate gyrus of the hippocampus and the subventricular zone that replenishes olfactory bulb neurons (Singec I ef a/., 2007; Zielton R, 2008).
- the availability of precursor cells is a key prerequisite for a transplant-based repair of defects in the mature nervous system.
- donor cells for neural transplants are largely derived from the fetal brain. This creates enormous ethical problems, in addition to immuno-rejection, and it is questionable whether such an approach can be used for the treatment of a large number of patients since neural stem cells can lose some of their potency with each cell division.
- Neural stem cells provide promising therapeutic potential for cell-replacement therapies in neurodegenerative disease (Mimeault ef a/., 2007). To date, numerous therapeutic transplantations have been performed exploiting various types of human fetal tissue as the source of donor material. However, ethical and practical considerations and their inaccessibility limit the availability as a cell source for transplantation therapies (Ninomiy M ef a/., 2006).
- Mammalian epithelial cells can be induced to acquire muscle-like shape and function (Paterson and Rudland, 1985), pancreatic exocrine duct cells can acquire an insulin-secreting endocrine phenotype (Bouwens, 1998a, b), and bone marrow stem cells can be differentiated into liver cells (Theise ef al., 2000) and into neuronal cells (Woodbury et al., 2000). Other such as Page et al.
- MASH1 , NeuroD, NeuroD2, MATH1 -3, and Neurogenin 1 -3 are bHLH transcription factors expressed during mammalian neuronal determination and differentiation (Johnson et al., 1990; Takebyashi et al., 1997; McCormick et al., 1996; Akazawa ef al. , 1995).
- Targeted disruptions of MASH1 , Ngn1 , Ngn2 or NeuroD in mice lead to the loss of specific subsets of neurons (Guillemot et al., 1993; Fode et al., 1998; Miyata et al. , 1999).
- U.S. patent No. 6,087, 168 (Levesque et al. ,) describes a method for converting or transdifferentiating epidermal basal cells into viable neurons.
- this method comprises the transfection of the epidermal cells with one or more expression vector(s) containing at least one cDNA encoding for a neurogenic transcription factor responsible for neural differentiation.
- Suitable cDNAs include: basic-helix-loop-helix activators, such as NeuroDI , NeuroD2, ASH1 , and zinc-finger type activators, such as Zic3, and MyT1 .
- the transfection step was followed by adding at least one antisense oligonucleotide known to suppress neuronal differentiation to the growth medium, such as the human MSX1 gene and/or the human HES1 gene (or non-human, homologous counterparts).
- the transfected cells were grown in the presence of a retinoid and a least one neurotrophin or cytokine, such as brain derived neurotrophic factor (BDNF), nerve growth factor (NGF), neurotrophin 3 (NT-3), or neurotrophin 4 (NT-4).
- BDNF brain derived neurotrophic factor
- NGF nerve growth factor
- NT-3 neurotrophin 3
- NT-4 neurotrophin 4
- a later process mentions the conversion of the epidermal basal cell into a neural progenitor, neuronal, or glial cell by exposing the epidermal basal cell to an antagonist of bone morphogenetic protein (BMP) and growing the cell in the presence of at least one antisense oligonucleotide comprising a segment of a MSX 1 gene and/or HES1 gene.
- BMP bone morphogenetic protein
- the present invention addresses these needs and provides various types of stem-like and progenitor-like cells and cells derived or differentiated from these stem-like or progenitor-like cells, as well as methods that can result in true cell dedifferentiation and cell reprogramming.
- the present invention relates to stem-like and progenitor-like cells and cells derived or differentiated from these stem-like or progenitor-like cells.
- the invention further relates to methods for cell dedifferentiation and cell reprogramming.
- the invention further features compositions and methods that are useful for reprogramming cells and related therapeutic compositions and methods.
- One particular aspect relates to the development of a technology to reprogram a somatic cell or non-neuronal cell to a cell having one or more morphological physiological, and/or immunological features of a neural stem cell and which possess the capacity to differentiate along neuronal and glial lineages.
- the invention is more particularly concerned with methods of generating stable Neural Stem-Like Cells (NSLCs) from human somatic cells, human progenitor cells and/or of human stem cells, as well as cells, cell lines and tissues obtained by using such methods.
- the invention further relates to compositions and methods to induce de-differentiation of human somatic cells into Neural Stem-Like Cells that express neural stem cell specific markers.
- the present invention it is possible to effect the conversion of cells to various types of differentiated neuronal cells that can be created from a single cell type taken from an individual donor and then reprogrammed and transplanted into the same individual.
- Upon induction cells according to the invention express neural stem-cell specific markers and become Neural Stem-Like cells.
- the invention relates to a method of transforming a cell of a first type to a desired cell of a different type.
- The comprises i) obtaining a cell of a first type; ii) transiently increasing in the cell of a first type intracellular levels of at least one reprogramming agent, whereby the transient increase induces direct or indirect endogenous expression of at least one gene regulator; iii) placing the cell in conditions for supporting the growth and/or the transformation of the desired cell and maintaining intracellular levels of the at least one reprogramming agent for a sufficient period of time to allow stable expression of the at least one gene regulator in absence of the reprogramming agent; and iv) maintaining the cell in culture conditions supporting the growth and/or the transformation of the desired cell.
- the expression of one or more of the secondary genes is characteristic of phenotypicai and functional properties of the desired cell while being not characteristic of phenotypicai and functional properties of an embryonic stem cell. Therefore, at the end of the period of time, the desired cell of a different type is obtained.
- the invention relates to a method of transforming a cell of a first type to a cell of a second different type.
- the method comprises contacting the cell of a first type with one or more agents capable of increasing within said cell levels of at least one reprogramming agent and directly or indirectly remodeling the chromatin and/or DNA of the cell.
- the at least one reprogramming agent is selected for inducing directly or indirectly the expression of morphological and functional characteristics of a desired cell of a different type or different cell lineage.
- the invention relates to a method of transforming a cell of a first type to a cell of a second different type.
- the method comprises contacting the chromatin and/or DNA of a cell of a first type with an agent capable of remodeling chromatin and/or DNA of said cell; and increasing intracellular levels of at least one reprogramming agent.
- the at least one reprogramming agent is selected for inducing directly or indirectly the expression of morphological and functional characteristics of a desired cell of a different type or cell lineage.
- a further aspect of the invention relates to a method of transforming a cell of a first type to a cell of a desired cell of a different type, comprising increasing intracellular levels of at least one reprogramming agent, wherein the at least one reprogramming agent is selected for inducing directly or indirectly the expression of morphological and functional characteristics of a desired second cell type; and maintaining the cell of a first type in culture conditions for supporting the transformation of the desired cell for a sufficient period of time to allow stable expression of a plurality of secondary genes whose expression is characteristic of phenotypical and functional properties of the desired cell, wherein at least one of the secondary genes is not characteristic of phenotypical and functional properties of an embryonic stem cell.
- the desired cell of a different type is obtained and the obtained cell is further characterized by a stable repression of a plurality of genes expressed in the first cell type.
- a further aspect of the invention concerns a process wherein a cell of a first type is reprogrammed to a desired cell of a different type, the process comprising:
- transient increase of intracellular levels of at least one reprogramming agent wherein the at least one reprogramming agent induces a direct or indirect endogenous expression of at least one gene regulator, and wherein the endogenous expression of the said at least one gene regulator is necessary for the existence of the desired cell of a different type;
- stable expression of the secondary genes is the result of the stable expression of the at least one gene regulator, and wherein: (i) stable expression of the plurality of secondary genes is characteristic of phenotypical and/or functional properties of the desired cell, (ii) stable expression of at least one of said secondary genes is not characteristic of phenotypical and functional properties of an embryonic stem cell, and wherein (i) and (ii) are indicative of successful reprogramming of the cell of the first type to the desired cell of the different type.
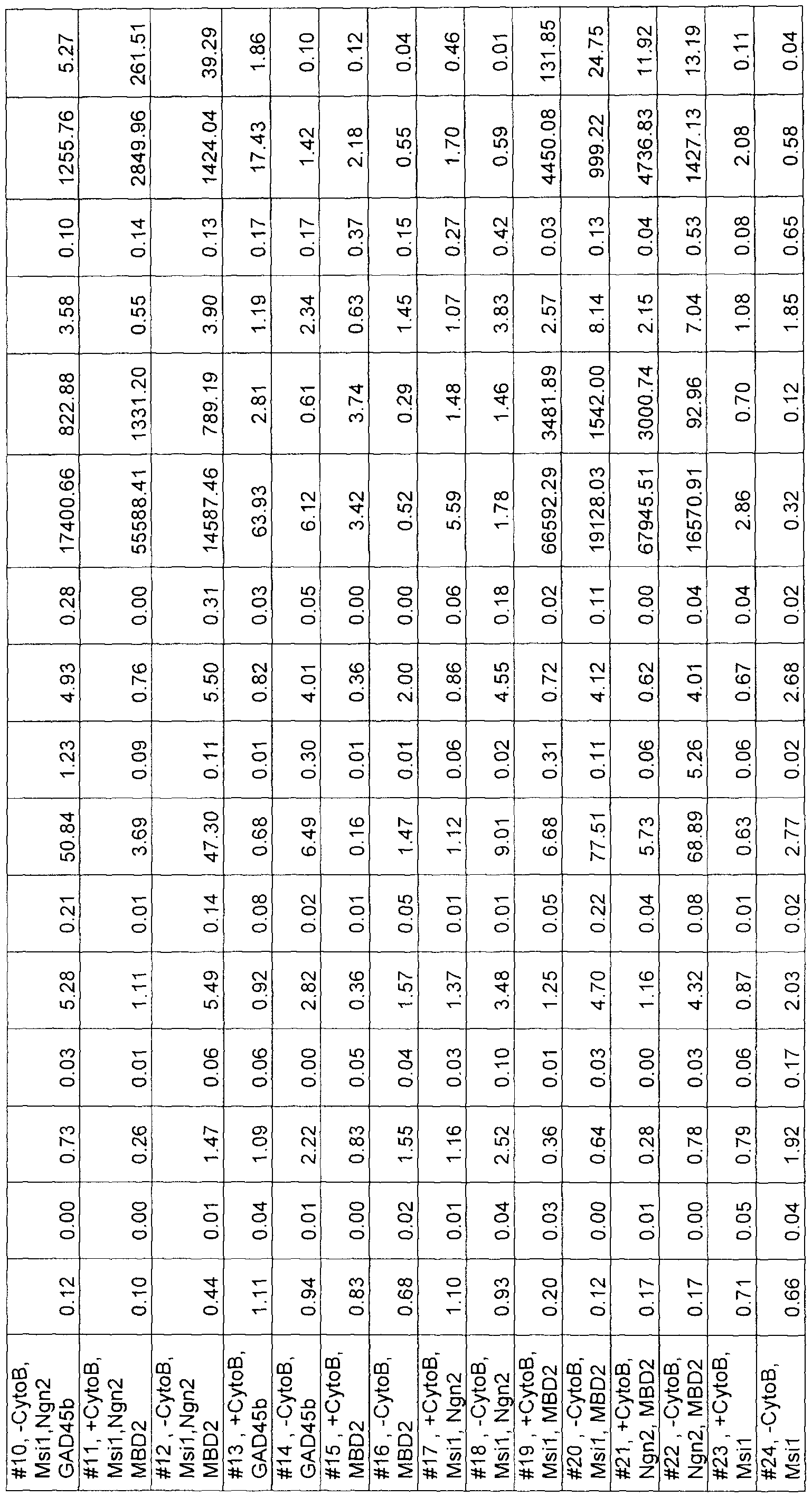
- the at least one reprogramming agent in the process is a Msi1 polypeptide, or a Ngn2 polypeptide together with a MDB2 polypeptide.
- the at least one gene regulator is Sox2 Msi1 , or both.
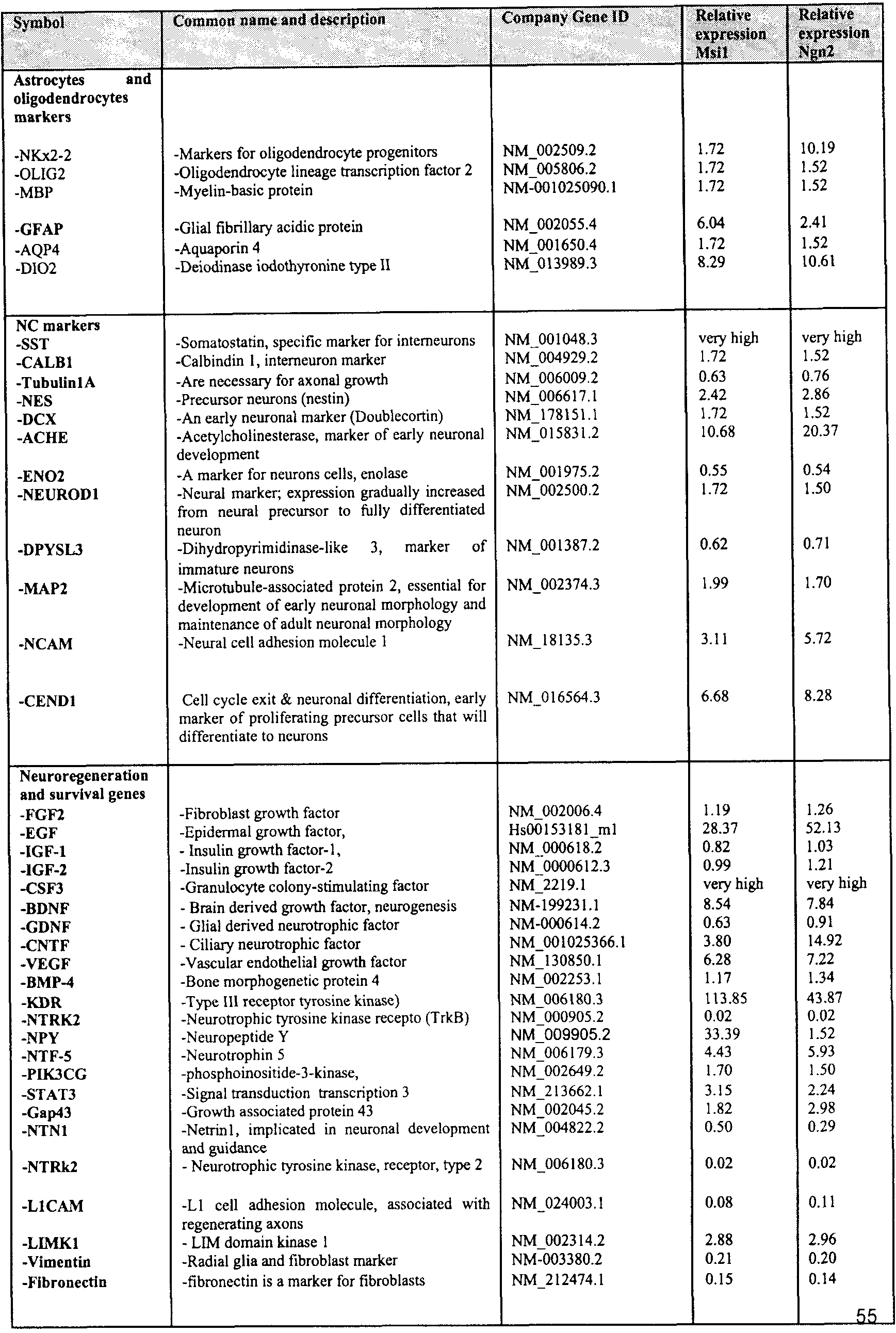
- the at least one gene regulator may is one or more of the genes listed in Table A for Neural Stem-Like Cells.
- the invention relates to a method of obtaining a Stem-Like Cell (SLC), comprising:
- the invention relates to a method of obtaining a Stem-Like Cell.
- the method comprises increasing intracellular levels of at least one polypeptide specific to the desired stem cell type that is able to drive directly or indirectly transformation of the cell of the first type into the Stem-Like Cell.
- the method may further comprises contacting chromatin and/or DNA of a cell of a first type with a histone acetylator, an inhibitor of histone deacetylation, a DNA demethylator, and/or an inhibitor of DNA methylation; and/or increasing intracellular levels of at least one other polypeptide specific to the desired stem cell type that is able to drive directly or indirectly transformation of the cell of the first type into a Stem-Like Cell.
- the invention relates to a method of obtaining a Neural Stem-Like Cell (NSLC).
- the method comprises increasing intracellular levels of at least one neural stem cell specific polypeptide that is able to drive directly or indirectly transformation of the cell of the first type into a NSLC.
- the method further comprises.contacting chromatin and/or DNA of a cell of a first type with a histone acetylator, an inhibitor of histone deacetylation, a DNA demethylator, and/or an inhibitor of DNA methylation; and/or increasing intracellular levels of at least one other neural stem cell specific polypeptide that is able to drive directly or indirectly transformation of the cell of the first type into a NSLC.
- Another aspect of the invention concerns a method of obtaining a Neural Stem-Like Cell (NSLC).
- the method comprises transfecting a skin cell with a polynucleotide encoding Musashil , Musashil and Neurogenin 2, Musashil and Methyl-CpG Binding Domain Protein 2 (MBD2), or Neurogenin 2 and Methyl-CpG Binding Domain Protein 2, thereby reprogramming the skin cell into a NSLC.
- MBD2 Methyl-CpG Binding Domain Protein 2
- the method comprises exposing a skin cell to: (i) an inhibitor of histone deacetylation, (ii) an inhibitor of DNA methylation, (iii) a histone acetylator, and/or (iv) a DNA demethylator such as a MBD2 polypeptide and/or transfecting with a polynucleotide encoding a MBD2 polypeptide; and further transfecting the cell (either simultaneously, before, or afterwards) with a polynucleotide encoding MUSASHI1 and/or with a polynucleotide encoding NGN2, thereby reprogramming the skin cell into a NSLC.
- Some other cells such as keratinocytes and CD34 + cells, can also be used and reprogrammed.
- the method of obtaining a Neural Stem-Like Cell comprises:
- NSLC is obtained and the obtained NSLC is further characterized by a stable repression of a plurality of genes expressed in the first cell type.
- the method of obtaining a Neural Stem-Like Cell comprises:
- the method of obtaining a Neural Stem-Like Cell comprises:
- Certain aspects of the invention concerns isolated cells, cell lines, compositions, 3D assembly of cells, and tissues comprising cells obtained using the methods described herein. Additional aspects concerns the use of such isolated cells, cell lines, compositions, 3D assembly of cells, and tissues of medical treatment and methods of regenerating a mammalian tissue or organ.
- a further aspect concerns a method for repairing or regenerating a tissue in a subject.
- the method comprises the administration of a reprogrammed cell as defined herein to a subject in need thereof, wherein the administration provides a dose of reprogrammed cells sufficient to increase or support a biological function of a given tissue or organ, thereby ameliorating the subject's condition.
- the benefits of the present invention are significant and include lower cost of cell therapy by eliminating the need of immuno-suppressive agents, no need for embryos or fetal tissue, thus eliminating ethical and time constraints, lower cost of production, and no health risks due to possible transmission of viruses or other disease.
- the cells since the cells are created fresh, they tend to be more potent than cells that have been passaged multiple times.
- Figure 1 is a panel of light micrograph (10X) presenting cell morphology changes of untransfected and transfected cells with Msi1 and MBD2 at various time points.
- Figure 2 is a panel of photomicrographs obtained using CellomicsTM (10x) and revealing NCAM positive cells in transfected cells with Msi1 or Ngn2 in the presence of MBD2.
- HFFs were pre- treated with cytochalasin B (10 g/ml) and transfected with pCMV6-XL5-Msi1 and pCMV6-XL5- MBD2 or pCMV6-XL4-Ngn2 and pCMV6-XL5-MBD2.
- proliferation medium NPBM, Lonza
- NbActive Basal growth factor (BitsTM) supplemented with NGF (20ng/ml), bFGF (20 ng/ml), ATRA (5 ⁇ ) and Forskolin (10 ⁇ ). Cells were incubated at 37°C, 5%C0 2 , 5%0 2 for 20 days.
- Figure 3 is a panel of photomicrographs obtained using CellomicsTM (10x) and revealing MAP2b positive cells in transfected cells with Msi1 or Ngn2 in the presence of MBD2.
- MAP2b positive cells were undetectable in untransfected cells and cells transfected with Pax6/MBD2.
- HFFs were pre-treated with cytochalasin B ( ⁇ 0 ⁇ g/r ⁇ ) and transfected with pCMV6-XL5-Msi1 , pCMV6-XL4-Ngn2 or pCMV6-XL5-Pax6, and pCMV6-XL5-MBD2.
- NPBM proliferation medium
- Peprotech EGF (20ng/ml.
- bFGF bFGF (20ng/ml, Peprotech)
- Differentiation was induced by changing the medium to NbActive (BrainBitsTM) supplemented with NT-3 (20ng/ml), bFGF (20 ng/ml), ATRA (5 ⁇ ) and Forskolin (10 ⁇ ).
- Cells were incubated at 37°C, 5%C0 2 , 5%0 2 for 2 weeks.
- Figure 4A is a panel of photographs showing that neurospheres formed by NSLCs from Example V were completely dissociated into single cell suspensions using Accutase and one single cell was monitored over time to reveal neurosphere formation capacity (A, Light microscope observation). Neurospheres stained positive for Sox2.
- Figure 4B is a panel of photographs from immunohistochemistry results obtained using CellomicsTM. Immunohistochemistry was performed, on day 20, to detect makers for neurospheres and compared to expression levels in neurospheres formed by normal human neuroprogenitor cells (hNPC, Lonza). In addition to Sox2, cells stained positive for the neural stem cells markers Musashi, CD133, Nestin, and GFAP.
- Cells also stained positive for ⁇ - tubulin (a marker for neurons), 04 (a marker for oligodendrocytes), and GFAP (a marker for astrocytes), indicating the tri-potent differentiation potential of both sets of cells (NSLC and hNPC), and negative for NGFrec and NeuN (markers for differentiated neurons) indicating that the cells were not terminally differentiated.
- ⁇ - tubulin a marker for neurons
- 04 a marker for oligodendrocytes
- GFAP a marker for astrocytes
- Figure 5 is a panel photomicrographs from immunohistochemistry results obtained using CellomicsTM. Immunohistochemistry was performed on HFFs, NSLCs, and hNPCs to detect expression of markers for fibroblasts as well as neural stem cells (Sox2, Nestin, GFAP) in adherent cultures (that prevented cells from floating and forming neurospheres). Nuclei were stained with Hoechst (upper level pictures). HFFs expressed fibroblasts markers while NSLCs created from these HFFs did not. In comparison, the NSLCs expressed neural stem cell markers similarly to hNPCs while the HFFs did not express any of these markers.
- Figure 6 is a panel photomicrographs showing Human NSLCs.
- Human NSLCs were induced to differentiate into neuronal lineages in the presence of NS-A differentiation medium (StemCell Technologies) in the presence of BDNF (20ng/ml, Peprotech) and bFGF (40ng/ml, Peprotech) for three weeks.
- BDNF 20ng/ml, Peprotech
- bFGF 40ng/ml, Peprotech
- immunostaining using CellomicsTM (10x) revealed differentiation of the cells as shown by the decrease of Sox2 positive cells and increase in the number and intensity of staining of p75, ⁇ -tubulin and GABA positive cells, as well as differentiated morphology, while the total number of cells increased as shown by Hoechst staining.
- Figure 7 is another panel of photomicrographs.
- HFF, Keratinocytes, and CD34+ were transfected with pCMV6-Msi1-Ngn2 and pCMV6-XL5-MBD2.
- the medium was changed to proliferation medium (StemCell Technologies) supplemented with EGF (20ng/ml. Peprotech) and bFGF (20ng/ml, Peprotech) for two week and then analyzed.
- Photomicrographs using CellomicsTM (10x) show that NSLCs created from all three types of cells are positive for Nestin, Sox2 and GFAP (markers for neural stem cells), while the original HFFs are not.
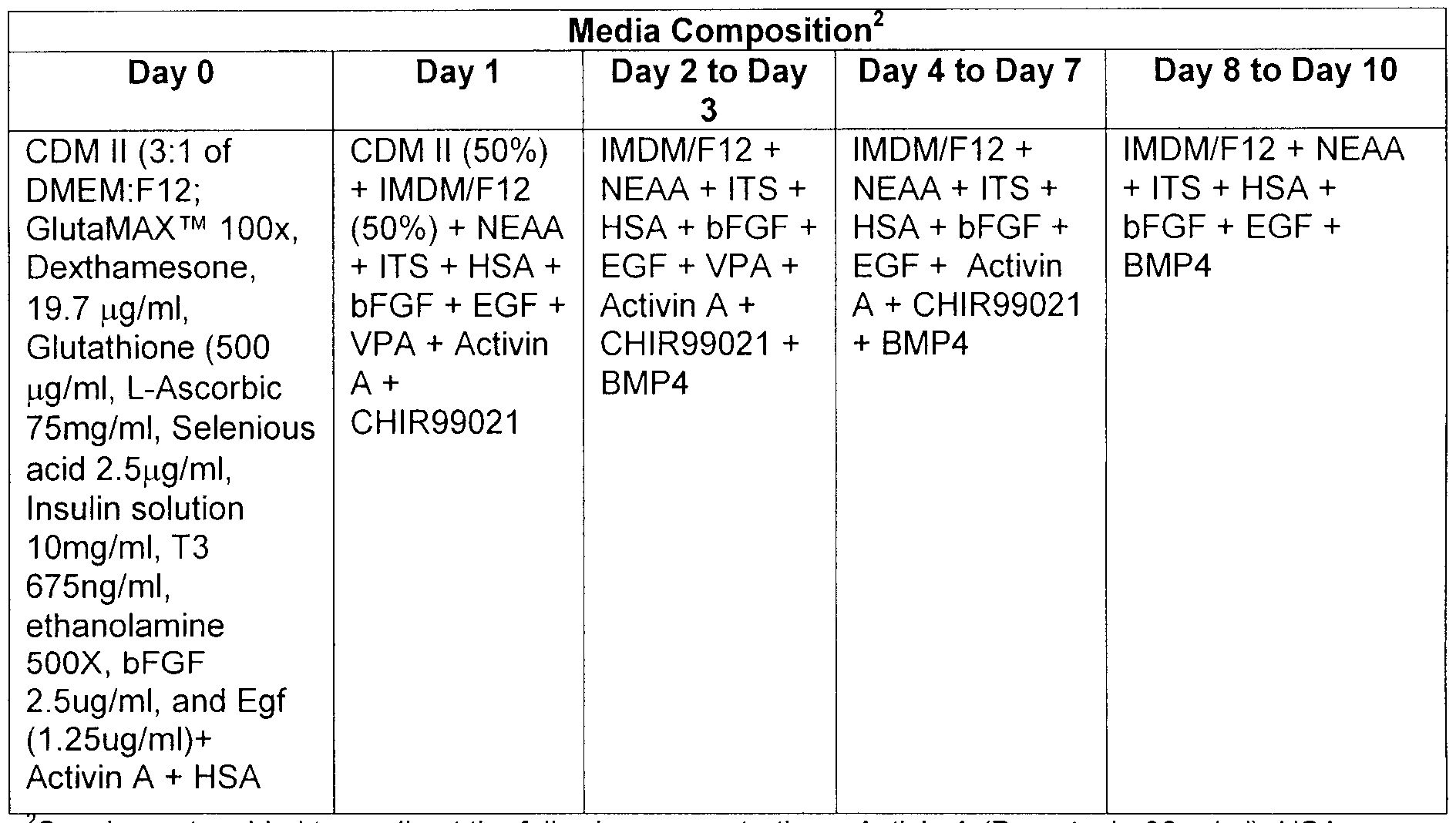
- FIG. 8 is panel of photomicrographs showing the effect of CDM medium on the trans- differentiation of HFF towards neurons.
- HFF were pre-treated with cytochalasin B (1 C ⁇ g/ml) and histone deacetylation inhibitor (VPA, 4 mM) and DNA methylation inhibitor (5-Aza, 5 ⁇ and cultured in CDM medium containing 3:1 ratio of Dulbecco's modified Eagle medium (DMEM, high glucose (4.5g/L) with L-glutamine and sodium pyruvate) and Ham's F-12 medium supplemented with the following components: EGF (4.2x10 "10 M), bFGF (2.8x10 "10 M), ITS (8.6x10 ⁇ 5 M), dexamethasone (1.0x10 "7 M), L-ascorbic acid phosphate magnesium salt n-hydrate (3.2x10 "4 M), L-3,3',5-triiodothyronine (2.0x10 "10 M), ethanolamine (10 " " ⁇ ), Gluta
- Figure 9 is panel of photomicrographs showing characterization of reprogrammed cells within CDM at different time points following the transfection with Msi1 and Ngn2.
- the transfected cells were treated with Cytochalasin B (10 ⁇ g/ml), VPA (4mM) and 5-AZA (5 ⁇ ) resulting in a disruption of the microfilaments and rounding up of the cells and loosening of the chromatin.
- Immunohistochemistry on the 3-Dimensional CDM was performed after one and two weeks using CellomicsTM (10X). The cells were positive for neuronal mature marker, such as MAP2b, but were absent in the untransfected control CDM.
- Figure 10 is another panel of photomicrographs.
- Cells within Day 4 CDM were lipotransfected with the two vectors pCMV6-XL5-Msi1 and pCMV6-XL4-Ngn2 individually or together in combination with pCMV-XL5-MBD2 for a period of 6 hours.
- transfection was performed on fresh HFFs after the 6 hours using Nucleofection, and these fresh HFFs were placed on top of the CDM at the same time as the lipofectamine media was changed to fresh CDM medium after 6 hours.
- Neural proliferation medium NPBM, Lonza
- Noggin 50ng/ml, Peprotech
- recombinant hFGF 20ng/ml, Peprotech
- recombinant hEGF 20ng/ml, Peprotech
- Differentiation was induced at day 7, by adding NS-A differentiation medium (StemCell Technologies) for 24 days.
- Immunohistochemistry was performed at various time points using CellomicsTM (10X).
- the CDM was stained with a specific antibody against Nestin (a marker for neural stem cells), and cells within the CDM expressed Nestin at all timepoints tested (Day 8, 15, and 21 ) following transfection.
- Figure 11 is a panel showing a picture of a polyacrylamide gel electrophoresis.
- NSLCs grown as adherent cultures or suspension cultures (as neurospheres) both express telomerase (which is expresses in all stem cells, but not in normal differentiated somatic cells). Both early (p5) and late (p27) passage NSLCs express telomerase. (The original HFFs from which the NSLCs were created did not express telomerase.) The samples (NSLCs) were spun down and protein concentration of the supernatant was determined using the BCA Assay.
- telomere extension primer 900ng of protein from each cell extract was added directly to the TRAP reaction mixture containing TRAP reaction buffer, dNTPs, template substrate (TS) primer, TRAP primer mix and Taq polymerase.
- the reaction mixtures were incubated at 30°C for 30 minutes for template synthesis, followed by a PCR procedure (95°C/15 min for initial denaturation, 94°C/30 sec, 59°C/30 sec, 72°C/1 min for 32 cycles) for amplification of the extended telomerase products.
- PCR polyacrylamide gel electrophoresis
- Figure 12 is a panel showing a picture showing Southern blot analysis of two different NSLC samples analyzed for Msi1 and Ngn2 gene integration two weeks after transient transfection.
- the Dig-labeled PCR probe revealed distinct signals in the positive control samples where the Msi1/Ngn2 plasmid DNA was spiked into HFF genomic DNA for the equivalence of 1 , 10 or 100 integrations per genome. There were a few weak and identical bands that appeared in the restriction enzyme digested genomic DNA from untransfected HFF and NSLC samples #1 and #2, suggesting that there was no plasmid DNA integration into the genomic DNA of NSLCs.
- Figure 13 is a panel whith a line graph and a bar graph showing improvement and significantly better clinical scores in EAE mice treated with NSLCs.
- Female 8 weeks old C57BL/6 mice were immunized with MOG 35 _55 (Sheldon Biotechnology Centre McGill University) in CFA containing 5 mg/ml of desiccated (killed and dried) Mycobacterium tuberculosis H37Ra (Difco, inc) at two sites on the back, and injected with 200 ng of pertussis toxin (List Biological Laboratories, Inc) in PBS intraperitoneally on days 0 and 2.
- mice Once the mice started showing symptoms of EAE (on Day 13 post-immunization), they were intravenously injected with 200 ⁇ of NSLC (1 million cells), hNPC (1 million cells), saline, or saline with cyclosporine. All mice except the saline control group received daily injections of cyclosporine. Mice were scored daily for clinical disease; data represent average daily scores. Mice that received a single injection of NSLCs had a significantly lower disease severity than mice that received hNPCs or cyclosporine alone.
- Figure 14 is a line graph showing the results of rotarod assessments according to Example XVII part 2. Rats were trained on the rotarod prior to the start of the experiment. Rats were placed on a stationary and rotating rotarod (rotating at 20 rpm) and the amount of time spent by the rats walking on the rotarod before falling off was monitored. Measurements were taken before (pre- surgery) and after (post-surgery) surgical left brain hemisphere ablation and treatment. The data points represent the mean number of falls by each animal during each 60 second testing session carried out at a constant speed of 20 rpm. Each group consisted of eight rats.
- Figure 15 is a line graph showing the results of the walking beam assessments according to Example XVII part 2. Rats were measured on their ability to cross a 100 cm long beam after surgical left brain hemisphere ablation and treatment. Two days after surgery, all groups fail to pass the test, and the animals are not able to stay in balance on the beam. One week after the surgery, all the animals show an improvement on their walking capacity, but no significant difference was noticeable between the different treated groups. From week 4 until week 26, the animals treated with NSLCs show significant improvement in their walking capacity compared to the other groups.
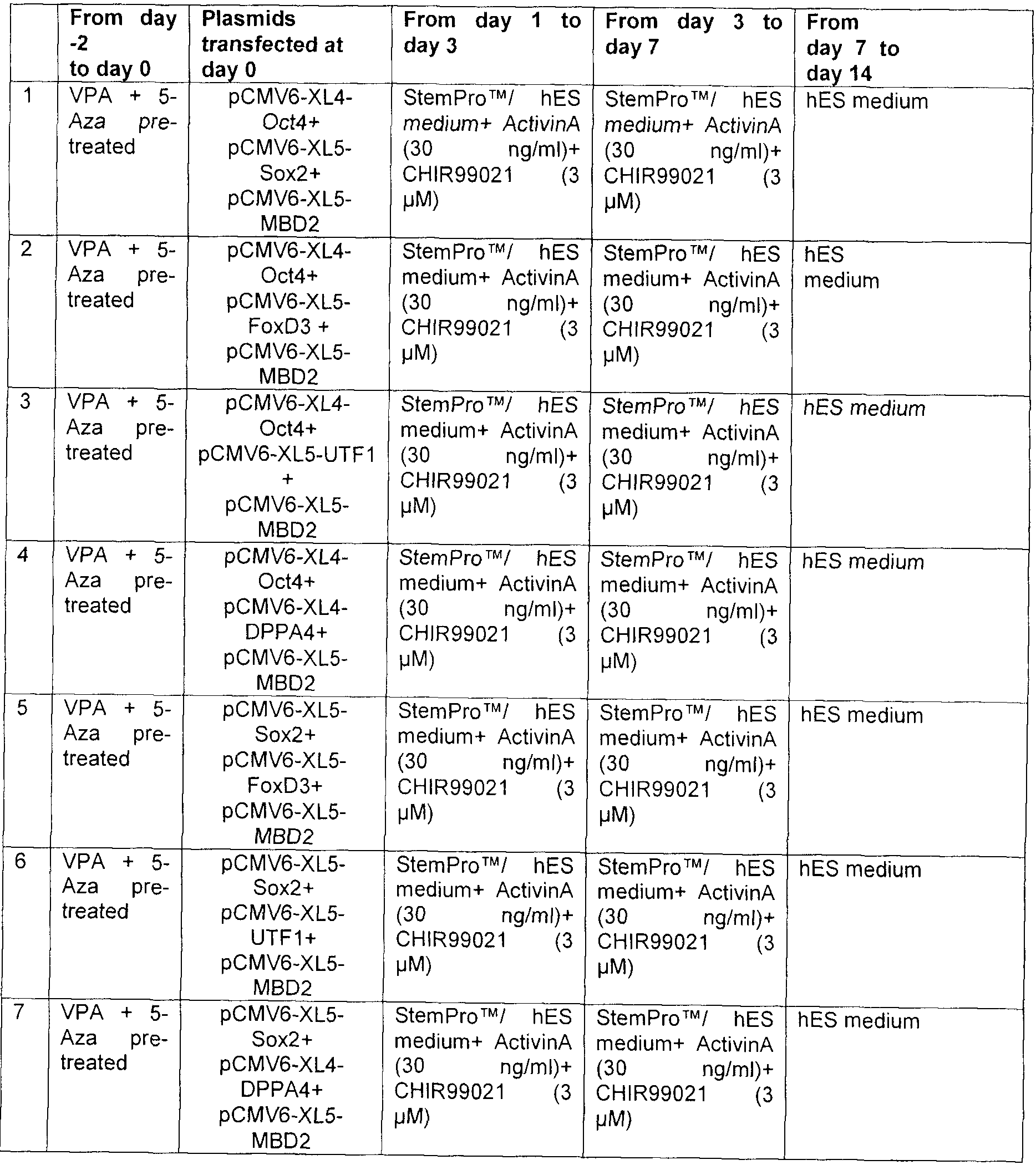
- Figure 16 is a panel showing photographs of ADSCs transiently transfected with various pluripotent vectors using nucieofector as described in Example XIX. Following the transfection cells were cultured in 6-well plates in suspension with a 50:50 mixture of ADSC complete medium (StemProTM-43) and embryonic stem cells medium (mTeSRITM, StemCell Technologies).
- Figure 17 is a panel showing photographs of ADSCs transiently transfected with pCMV6-XL5- Rex1/pCMV6-XL5-Klf4 and pCMV6-XL5-Rex1/pCMV6-XL4-Oct4.
- ADSCs were cultured in 96-well plates coated with MatrigelTM for 24 days in the presence of mTeSRITM medium supplemented with SB341542 and PD0325901 at 37°C, 5%C0 2 , 5%0 2 .
- live staining immunohistochemistry and AP staining were used. 1 -5% of total cells transfected with Rex1/Oct4 or Rex1/Klf4 showed a SSEA-4 + and TRA-1 -81 + phenotype (early pluripotency markers).
- FIG. 18 is a panel showing photographs of ADSCs transiently transfected with various pluripotent vectors. Following transfection the cells were plated in StemProTM MSC SFM medium on MatrigelTM (BD Biosciences) coated 24 well plates and incubated at 37°C, 5% C0 2, 5%0 2 .
- hES cell medium consisted in Dulbecco's Modified Eagle's Medium (DMEM, Invitrogen) supplemented with 20% KnockoutTM Serum Replacement (KSR, Invitrogen), 1 mM GlutaMAXTM, 100 ⁇ Non-essential Amino acids, 100 ⁇ ⁇ -mercaptoethanol and 10 ng/ml Fgf- 2.
- DMEM Dulbecco's Modified Eagle's Medium
- KSR KnockoutTM Serum Replacement
- Transfected cells transfected with Oct4/UTF1/MBD2, Oct4/Dppa4/MBD2, FoxD3/Dppa4/MBD2, Oct4/FoxD3/Dppa4, and Sox2/FoxD3/UTF1 were positive for SSEA-4 + , TRA1-60, and TRA-1-81 + phenotype (early pluripotency markers) at day 14.
- FIG 19 is a panel showing photographs of transiently transfected HFFs.
- HFFs were transiently transfected using the Nucleofector ® II Device (Lonza) following the procedure described in Example II with the exception that 1 pg of each of the following 3 DNA plasmids was used: pCMV-Oct4nuc-IRES2-Sox2nuc, pCMV-Klf4nuc-IRES2-Cmycnuc and pCMV- Nanognuc-IRES2-Lin28. The cells were pre-treated with or without VPA and 5-Aza.
- the cells were plated in the fibroblast medium, supplemented with or without VPA (2mM) and 5-AZA (2.5 ⁇ ) on MatrigelTM (BD Biosciences) coated 6-well plates and incubated at 37°C, 5% C0 2 .
- media was changed to 100% mTeSRITM medium (StemCell Technologies) supplemented with or without VPA and 5-AZA.
- cells were re- transfected as above and plated on MatrigelTM coated plates in mTeSRITM medium supplemented with or without VPA and 5-AZA. Media was changed daily as above.
- Y27632 Stemgent, 10 ⁇
- AP Alkaline Phosphatase Detection Kit
- TRA-1-81 similar to Mel2 human embryonic stem cell line (positive control)
- Figure 20 is a panel showing photographs of transfected NSLCs and BG-01.
- NSLCs and BG-01 NS were transfected as previously described in Example II by two episomal vectors, pEF- Oct4nuc-IRES2-MBD2 (NC1 ) or pCMV-FoxD3-2A-Oct4-2A-Klf4 (F72).
- N1 pEF- Oct4nuc-IRES2-MBD2
- F72 pCMV-FoxD3-2A-Oct4-2A-Klf4
- Figure 21 is a panel showing bright field pictures at day 17 of fibroblasts transfected with Msi1/Ngn2 and pCMV6-XL5-MBD2 placed in different media conditions and showing different morphologies and degree of differentiation, (a) Cells in neural proliferation medium from day 1 to day 12, and then in neural differentiation medium with cytokines from day 12 to 17. (b) Cells in neural proliferation medium from day 1 to day 12, and then in NbActive4 medium with cytokines from day 12 to 17. (c) Cells in neural differentiation medium with cytokines plus Fgf-2 from day 1 to day 12, and then in the same medium but without Fgf-2 from day 12 to 17.
- FIG. 22 is a panel showing pictures of immunochemistry results at day 17 of fibroblasts transfected with Msi1/Ngn2 and pCMV6-XL5-MBD2 in Figure 21. Fig.
- FIGS. 22A and 22B Cells were in NS-A Proliferation Medium from day 1 to day 12, and then in NS-A Differentiation Medium (A) or NBActive4 medium (B) with cytokines from day 12 to 7. There were more cells in B, but Differentiation from day 12-17 was too short to induce expression of ⁇ -tubulin in both cases.
- Figs. 22C-E Cells were in NS-A Differentiation Medium (C) or NbActive4 medium (D) from day 1-17 (with FGF-2 supplementation from day 1-12), or CDM II medium from day 1-12 and then NS-A Differentiation Medium from day 12-17 (E). There were a large number of cells in C and a much smaller number of cells in D and E.
- Cells were immunopositive for both GFAP and ⁇ - tubulin in all cases and placing the cells in differentiation or non-proliferation media from day 1 onwards appears to have induced a more direct transformation into neurons and glia, with more intense ⁇ -tubulin than GFAP positive cells in E.
- Figure 23 is a panel showing two heat maps providing a global overview of the gene expression comparison between either NSLC vs. HFF (Set 1), or NSLC vs. hNPC (Set 2).
- NSLC has a distinct gene expression profile when compared to either HFF or hNPC. Based on the intensity (the higher the intensity, the higher the relative change in expression), NSLC is much more similar to hNPC than to HFF.
- Figure 24 is a panel showing pictures of NSLCs.
- NSLCs were tested to determine if they are a population of Skin-Derived Precursors Cells (SKPs).
- SKPs capable of proliferating in response to EGF and bFGF, express nestin and fibronectin, and can differentiate into both neuronal and mesodermal progeny including into adipocytes.
- adipocyte-derived stem cells ADSCs
- NSLCs StemProTM proliferation medium
- differentiation towards adipocytes were induced by culturing these cells in differentiation medium consisting in DMEM/F12 (50:50), ITS (1 : 100), HEPES (1 :100), GlutaMAXTM (1 :100), T3 (0.2 nM), Rosiglitasone (0.5 ⁇ ), IBMX (100 ⁇ ) and Dexamethasone (1 ⁇ ).
- IBMX and Dexamethasone were withdrawn from the medium.
- NSLCs are not a population of Skin- Derived Precursors Cells (SKPs).
- the present invention relates to methods for cell dedifferentiation and cell reprogramming.
- a significant aspect of the present invention is that it permits the use of a patient's own cells to develop different types of cells that can be transplanted after steps of in vitro dedifferentiation and in vitro reprogramming.
- this technology eliminates the problems associated with transplantation of non-host cells, such as, immunological rejection and the risk of transmitting disease.
- the cells since the cells are "newly created", they have the potential to be more potent than alternative sources of natural cells that have already divided multiple times. Definitions
- polynucleotide refers to any DNA or RNA sequence or molecule, comprising encoding nucleotide sequences.
- the term is intended to encompass all polynucleotides whether occurring naturally or non-naturally in a particular cell, tissue or organism. This includes DNA and fragments thereof, RNA and fragments thereof, cDNAs and fragments thereof, expressed sequence tags, artificial sequences including randomized artificial sequences.
- polypeptide refers to any amino acid sequence having a desired functional biological activity (e.g. DNA demethylation).
- the term is intended to encompass complete proteins, fragments thereof, fusion proteins and the like, including carbohydrate or lipid chains or compositions.
- Trans-differentiation refers to a direct switch of an already differentiated cell to another type of differentiated cell.
- De-differentiation refers to the loss of phenotypic characteristics of a differentiated cell by activating or deactivating genes or metabolic pathways.
- Marker refers to a gene, polypeptide, or biological function that is characteristic of a particular cell type or cellular phenotype.
- Genetically-engineered DNA sequence is meant a DNA sequence wherein the component sequence elements of DNA sequence are organized within the DNA sequence in a manner not found in nature.
- Signal sequence refers to a nucleic acid sequence which, when incorporated into a nucleic acid sequence encoding a polypeptide, directs secretion of the translated polypeptide from cells which express said polypeptide, or allows the polypeptide to readily cross the cell membrane into a cell.
- the signal sequence is preferably located at the 5' end of the nucleic acid sequence encoding the polypeptide, such that the polypeptide sequence encoded by the signal sequence is located at the N-terminus of the translated polypeptide.
- signal peptide is meant the peptide sequence resulting from translation of a signal sequence.
- Ubiquitous promoter refers to a promoter that drives expression of a polypeptide or peptides encoded by nucleic acid sequences to which promoter is operably linked.
- Preferred ubiquitous promoters include human cytomegalovirus immediate early (CMV); simian virus 40 early promter (SV40); Rous sarcoma virus (RSV); or adenovirus major late promoter.
- Gene expression profiling means an assay that measures the activity of multiple genes at once, creating a global picture of cellular function. For example, these profiles can distinguish between human neural stem cells and somatic cells that are actively dividing or differentiating.
- Transfection refers to a method of gene delivery that introduces a foreign nucleotide sequences (e.g. DNA molecules) into a cell preferably by a non-viral method.
- foreign DNA is introduced to a cell by transient transfection of an expression vector encoding a polypeptide of interest, whereby the foreign DNA is introduced but eliminated over time by the cell and during mitosis.
- transient transfection is meant a method where the introduced expression vectors and the polypeptide encoded by the vector, are not permanently integrated into the genome of the host cell, or anywhere in the cell, and therefore may be eliminated from the host cell or its progeny over time. Proteins, polypeptides, or other compounds can also be delivered into a cell using transfection methods.
- Neuron-like Cell refers to an immature cell of the nervous system, which can differentiate into neurons and glia (oligodendrocytes and astrocytes).
- Neuro Stem Cell is an ectoderm germ layer derived multipotent stem cell having, as a physiological feature, a capacity to form neuroprogenitor cells and under physiological conditions that favor differentiation to form neurons and glia.
- Neuro Stem-Like Cell or “NSLC” refers to any cell-derived multipotent stem cell having, as a physiological feature, a capacity to form other neural stem-like cells and neuroprogenitor-like cells and under physiological conditions that favor differentiation to form neuron-like cells and glial-like cells.
- Neurosphere refers to a cellular aggregate of neural stem cells and neuroprogenitor cells that form a floating sphere formed as a result of proliferation of the neural stem cells and neuroprogenitor cells in appropriate proliferation conditions.
- NSLCs also form neurospheres consisting of aggregates of NSLCs and neuroprogenitor-like cells.
- Reprogrammed cell refers to a cell that has undergone stable trans-differentiation, de- differentiation, or transformation. Some reprogrammed cells can be subsequently induced to re- differentiate. The reprogrammed cell stably expresses a cell-specific marker or set of markers, morphology, and/or biological function that was not characteristic of the original cell.
- Reprogrammed somatic cell refers to a process that alters or reverses the differentiation status of a somatic cell, which can be either complete or partial conversion of the differentiated state to an either less differentiated state or a new differentiated state.
- Regeneration refers to the capability of contributing to the repair or de novo construction of a cell, tissue or organ.
- Differentiation refers to the developmental process of lineage commitment of a cell. Differentiation can be assayed by measuring an increase in one or more cell-differentiation specific markers relative to the expression of the undifferentiated cell markers.
- Lineage refers to a pathway of cellular development, in which a more undifferentiated cell undergoes progressive physiological changes to become a more differentiated cell type having a characteristic function (e.g., neurons and glia are of a neuroprogenitor linage, which is of an ectoderm lineage which formed from blastocysts and embryonic stem (ES) cells).
- a characteristic function e.g., neurons and glia are of a neuroprogenitor linage, which is of an ectoderm lineage which formed from blastocysts and embryonic stem (ES) cells.
- tissue refers to an ensemble of cells (identical or not) and an extracellular matrix (ECM) that together carry out a specific function or set of functions.
- ECM extracellular matrix
- CDM is meant a living tissue equivalent or matrix, a living scaffold, or cell-derived matrix.
- Some aspects of the invention concerns methods and cells to transform or reprogram a given somatic cell into a pluripotent, multipotent and/or unipotent cell.
- Some aspects of the invention relates to methods for conditioning a somatic cell to reprogramming into a pluripotent, multipotent or unipotent cell.
- the terms "transform” or “reprogram” are used interchangeably to refer to the phenomenon in which a cell is dedifferentiated or transdifferentiated to become pluripotent, multipotent and/or unipotent. The dedifferentiated cell could subsequently be redifferentiated into a different type of cell. Cells can be reprogrammed or converted to varying degrees.
- the terms "transforming” or “reprogramming” methods can refer to methods wherein it is possible to reprogram a cell such that the "new" cell shows morphological and functional characteristics of a new or different specific cell lineage (e.g. the transformation of fibroblast cells into neuronal cells).
- the term "somatic cell” refers to any differentiated cell forming the body of an organism, apart from stem cells, progenitor cells, and germline cells (i.e. ovogonies and spermatogonies) and the cells derived therefrom (e.g.
- Somatic cells can be differentiated cells isolated from adult or can be fetal somatic cells. Somatic cells are obtained from animals, preferably human subjects, and cultured according to standard cell culture protocols available to those of ordinary skill in the art.
- “Stem cell” refers to those cells which retain the ability to renew themselves through mitotic cell division and which can differentiate into a diverse range of specialized cell types. It includes both embryonic stem cells that are found in blastocysts, and adult stem cells that are found in adult tissues. “Totipotent cells” refers to cells that have the ability to develop into cells derived from all three embryonic germ layers (mesoderm, endoderm and ectoderm) and an entire organism (e.g., human being if placed in a woman's uterus in the case of humans). Totipotent cells may give rise to an embryo, the extra embryonic membranes and all post-embryonic tissues and organs.
- pluripotent refers to cells that can produce only cells of a closely related family of cells (e.g. hematopoietic stem cells differentiate into red blood cells, white blood cells, platelets, etc.).
- unipotent cells refers to cells that have the capacity to develop/differentiate into only one type of tissue/cell type (e.g. skin cells).
- the present invention allows the reprogramming of any cell to a different type of cell.
- the present application focuses primarily on the preparation of Stem-Like cells, especially, Neural Stem-Like Cells (NSLCs)
- the invention is not so restricted because many different types of cells can be generated according to the principles described herein.
- NSLCs Neural Stem-Like Cells
- the Examples section describes embodiments where fibroblasts, keratinocytes, CD34 + cells, adipose-derived stem cells (ADSCs), neural stem cells (including NSLCs), and cells within a Cell-Derived Matrix (CDM) are reprogrammed
- the invention is not limited such cells.
- the invention may be employed for the reprogramming of virtually any cell of interest.
- a general aspect of the invention relates to a method of transforming a cell of a first type to a cell of a second different type.
- examples of cells of a first type include, but are not limited to germ cells, embryonic stem cells and derivations thereof, adult stem cells and derivations thereof, progenitor cells and derivations thereof, cells derived from mesoderm, endoderm or ectoderm, and a cell of mesoderm, endoderm or ectoderm lineage such as an adipose-derived stem cell (ADSC), mesenchymal stem cell, hematopoietic stem cell (CD34 + cell), skin derived precursor cell, hair follicle cell, fibroblast, keratinocyte, epidermal cell, endothelial cell, epithelial cell, granulosa epithelial cell, melanocyte, adipocyte, chondrocyte, hepatocyte, lymphocyte (B and T lymphocyte),
- ADSC
- examples of cells of a second type include, but are not limited to germ cells, embryonic stem cells and derivations thereof, adult stem cells and derivations thereof, progenitor cells and derivations thereof, cells derived from mesoderm, endoderm or ectoderm, and a cell of mesoderm, endoderm or ectoderm lineage such as an adipose-derived stem cell, mesenchymal stem cell, hematopoietic stem cell, skin derived precursor cell, hair follicle cell, fibroblast, keratinocyte, epidermal cell, endothelial cell, epithelial cell, granulosa epithelial cell, melanocyte, adipocyte, chondrocyte, hepatocyte, lymphocyte (B and T lymphocyte), granulocyte, macrophage, monocyte, mononuclear cell, pancreatic islet cell, Sertoli cell, neuron, glial cell, cardiac muscle cell, and other muscle
- each of the above "-like" cell (a cell that has similar but not completely identical characteristics of the known natural type of the cell) is also included in the examples of cells of a second type.
- the method of transforming a cell of a first type into a cell of a second different type comprises the steps of:
- the cell of a different type obtained after the transformation is further characterized by a stable repression of a plurality of genes expressed in the first cell type.
- step iii) may be carried out consecutively to step ii), simultaneously with step ii), or before step ii).
- the invention relates to a process wherein a cell of a first type is reprogrammed to a desired cell of a different type, the process comprising: - a transient increase of intracellular levels of at least one reprogramming agent, wherein the at least one reprogramming agent induces a direct or indirect endogenous expression of at least one gene regulator, wherein the endogenous expression of the at least one gene regulator is necessary for the existence of the desired cell of a different type;
- stable expression of the plurality of secondary genes is the result of the stable expression of the at least one gene regulator, and wherein: (i) stable expression of the plurality of secondary genes is characteristic of phenotypical and/or functional properties of the desired cell, (ii) stable expression of at least one of the secondary genes is not characteristic of phenotypical and functional properties of an embryonic stem cell, and wherein (i) and (ii) are indicative of successful reprogramming of the cell of the first type to the desired cell of the different type.
- transiently increasing refers to an increase that is not necessarily permanent and therefore, which may decrease or disappear over time.
- transiently increasing intracellular levels of at least one reprogramming agent in a cell it means that the increase in present for a sufficient period of time for causing particular cellular events to occur (e.g. inducing stable endogenous expression of a gene regulator).
- a transient increase is not permanent and is not associated for instance to genome integration of an expression vector.
- the term "reprogramming agent” refers to a compound that is capable of inducing directly or indirectly the expression of morphological and/or functional characteristics of the desired cell of a different type. Preferred compounds include those capable of driving directly or indirectly transformation of the cell of the first type into the desired cell of a different type.
- the reprogramming agent is selected for inducing a direct or indirect endogenous expression of at least one gene regulator as defined herein. There are many compounds that may be helpful in reprogramming a cell according to the invention and these compounds can be used alone or in combinations.
- the reprogramming agent is a polynucleotide or polypeptide selected according to TABLE A: TABLE A:
- Pax3 isoform NMJ 81457.3 P23760 Hs.42146 Pax3 Pax3, isoform NM_000438.5 P23760 Hs.42146 Pax3a
- Pax3 isoform NM_181458.3 Q494Z3, Q494Z4 Hs.42146 Pax3d (TrE BL)
- Pax3 isoform N _181459.3 Q494Z3, Q494Z4 Hs.42146 Pax3e (TrEMBL)
- Pax3 isoform NM_001127366.2 Q494Z4 Hs.42146 Pax3i (TrEMBL)
- the reprogramming agent is a polypeptide which shares at least 75%, 80%, 85%, 90%, 95%, 97%, 99% or more of the functionality or sequence identity of any one of the reprogramming agents in the table hereinbefore. Identifying the "sufficient period of time" to allow stable expression of the at least one gene regulator in absence of the reprogramming agent and the "sufficient period of time" in which the cell is to be maintained in culture conditions supporting the transformation of the desired cell is within the skill of those in the art. The sufficient or proper time period will vary according to various factors, including but not limited to, the particular type and epigenetic status of cells (e.g. the cell of the first type and the desired cell), the amount of starting material (e.g.
- the sufficient period of time to allow a stable expression of the at least one gene regulator in absence of the reprogramming agent is about 1 day, about 2-4 days, about 4-7 days, about 1 -2 weeks, about 2-3 weeks or about 3-4 weeks.
- the sufficient period of time in which the cells are to be maintained in culture conditions supporting the transformation of the desired cell and allow a stable expression of a plurality of secondary genes is about 1 day, about 2-4 days, about 4-7 days, or about 1 -2 weeks, about 2-3 weeks, about 3-4 weeks, about 4-6 weeks or about.6-8 weeks.
- the number of transformed desired cells is substantially equivalent or even higher than an amount of cells a first type provided at the beginning.
- the present invention encompasses various types of compounds that are suitable for increasing in a cell of a first type the intracellular levels of at least one reprogramming agent.
- the compound should also be able to directly or indirectly remodel the chromatin and/or DNA of the cell, thus resulting directly or indirectly in the expression of morphological and functional characteristics of the desired cell of a different type.
- Preferred compounds are reprogramming agents as defined herein or any other compound having a similar activity and having the ability to activate or enhance the expression of the endogenous version of genes listed in the table of reprogramming agents hereinbefore and which are capable of driving directly or indirectly transformation of the cell of the first type into the desired cell of a different type.
- the increase in intracellular levels of the at least one reprogramming agent can be achieved by different means.
- the reprogramming agent is a polypeptide and increasing intracellular levels of such polypeptide include transfection (or co-transferction) of an expression vector having a polynucleotide (ex.
- DNA or RNA encoding the polypeptide(s), or by an intracellular delivery of polypeptide(s).
- transient expression is generally preferable.
- Additional suitable compounds may include compounds capable of increasing the expression of the endogenous version of genes listed in the table of reprogramming agents and gene regulators including, but not limited to, reprogramming factors listed in Table B.
- BMP4 Epidermal growth factor-Cripto/FRL-1 /Cryptic
- Vg1/GDF1 growth and differentiation factor-1
- IGF-1 Insulin-like growth factor 1
- HGF hepatocyte growth factor
- Valproic Acid BIX01294, R(+)BayK8644, RG108,
- Valproic Acid BIX01294, R(+)BayK8644, RG108,
- gene regulator refers to a polynucleotide or polypeptide whose expression is associated with a series of intracellular events leading to the transformation of a given cell of a first type into a pluripotent, multipotent and/or unipotent cell.
- expression of a gene regulator direcly or indirectly activates genes necessary for the phenotypical and functional characteristic of pluripotent, multipotent and/or unipotent cells, while repressing genes of the cell of a first type.
- the gene regulator may be the same or be different than the reprogramming agent. Examples of gene regulators according to the invention include, but are not limited to, the polynucleotides and polypeptides listed herein before in TABLE A.
- the gene regulator is a polypeptide which shares at least 75%, 80%, 85%, 90%, 95%, 97%, 99% or more of the functionality or sequence identity of any one of the gene regulators provided in the Table A hereinbefore.
- “conditions supporting growth” or “conditions supporting the transformation” when referring to a desired cell refers to various suitable culture conditions (temperature, pH, 0 2 tension, cell media, factors, compounds, growth substrate (ex. laminin, collagen, fibronectin, MatrigelTM, low-bind surface, nanostructured or charged surface, etc.), 3D environment, etc.)
- suitable culture conditions temperature, pH, 0 2 tension, cell media, factors, compounds, growth substrate (ex. laminin, collagen, fibronectin, MatrigelTM, low-bind surface, nanostructured or charged surface, etc.), 3D environment, etc.)
- phenotypical and functional properties when referring to a desired cell or to an embryonic stem cell, means the biological, biochemical, physiological and visual characteristics of a cell, including expression of certain genes and cell surface markers, which can be measured or assessed for confirming its identity or function(s).
- MUSASHI1 An example of a suitable reprogramming agent according to preferered embodiments of the invention is MUSASHI1.
- this polypeptide is preferred for driving a first cell, such as a fibroblast, into a Neural Stem-Like Cell (NSLC).
- the at least one reprogramming agent which said intracellular levels is increased is(are) either Musashil (Msi1 ) alone; Musashil (Msi1 ) and Neurogenin 2 (Ngn2); Musashil (Msi1 ) and methyl-CpG binding domain protein 2 (MBD2); or Neurogenin 2 (Ngn2) and methyl-CpG binding domain protein 2 (MBD2).
- Adequate intracellular levels of these polypeptides are preferred since they tend to be expressed throughout an entire cell lineage, from as early as embryonic stem cells (or even earlier) to pre-somatic cells (or even later).
- MBD2 is a member of a family of methyl-CpG-binding proteins that has been reported to be both a transcriptional repressor and a DNA demethylase (dMTase).
- MBD2 generally refers to the human methyl-CpG binding domain protein 2.
- NCBI GeneBankTM accession number of human MBD2 is N _003927.3/AF072242, the UniProtTM accession number is NP-003918/Q9UBB5 and the UniGeneTM accession number is Hs.25674.
- siV generally refers to the human musashi homolog 1 .
- the GeneBankTM (NCBI) accession number of human Msi1 is NM_002442.2/AB012851 , the UniProtTM accession number is NP-002433/O43347 and the UniGeneTM accession number is Hs.15831 1 .
- Ngn2 generally refers to the human neurogenin 2.
- the GeneBankTM (NCBI) accession number of human Ngn2 is NM_024019.2/BC036847, the UniProtTM accession number is NP-076924/Q9H2A3 and the UniGeneTM accession number is Hs.567563.
- the method of transforming a cell of a first type to a desired cell of a different different type comprises the steps of either:
- step 2) may be carried out consecutively to step 1 ), simultaneously with step 1 ), or before step 1 ).
- the invention relates to a method for obtaining a Neural Stem- Like Cell (NSLC), comprising:
- the term "remodelling the chromatin and/or DNA” refers to dynamic structural changes to the chromatin. These changes can range from local changes necessary for transcriptional regulation, to global changes necessary for opening up the chromatin structure or chromosome segregation to allow transcription of the new set of genes characteristic of the desired cell of a different type, to closing up of the chromatin structure or chromosome segregation to prevent transcription of certain genes that are not characteristic of the desired cell of a different type.
- opening up of the chromatin structure refers more specifically to acetylation of histones, and demethylation of DNA
- closing up of the chromatin structure refers more specifically to deacetylation of histones, and methylation of DNA.
- compound refers to a compound capable of effecting a desired biological function. The term includes, but is not limited to, DNA, RNA, protein, polypeptides, and other compounds including growth factors, cytokines, hormones or small molecules.
- compounds capable of remodeling chromatin and/or DNA include, but are not limited to, histone acetylators, inhibitors of histone deacetylation, DNA demethylators, inhibitors of DNA methylation and combination thereof.
- “Inhibitor of DNA methylation” refers to an agent that can inhibit DNA methylation.
- DNA methylation inhibitors have demonstrated the ability to restore suppressed gene expression.
- Suitable agents for inhibiting DNA methylation include, but are not limited to 5-azacytidine, 5- aza-2-deoxycytidine, 1- -D-arabinofuranosil-5-azacytosine, and dihydro- 5-azacytidine, and zebularine (ZEB), BIX (histone lysine methytransferase inhibitor), and RG108.
- “Inhibitor of histone deacetylation” refers to an agent that prevents the removal of the acetyl groups from the lysine residues of histones that would otherwise lead to the formation of a condensed and transcriptionally silenced chromatin.
- Histone deacetylase inhibitors fall into several groups, incuding: (1 ) hydroxamic acids such as trichostatin (A), (2) cyclic tetrapeptides, (3) benzamides, (4) electrophilic ketones, and (5) aliphatic acid group of compounds such as phenylbutyrate and valporic acid.
- Suitable agents to inhibit histone deacetylation include, but are not limited to, valporic acid (VPA), phenylbutyrate Trichostatin A (TSA), Na-butyrate, and benzamides.
- VPA valporic acid
- TSA phenylbutyrate Trichostatin A
- Na-butyrate benzamides.
- VPA promotes neuronal fate and inhibits glial fate simultaneously through the induction of neurogenic transcription factors including NeuroD.
- Histone Acetylator refers to an agent that inserts acetyl groups to the lysine residues of histones that opens up the chromatin and turns it into a transcriptionally active state.
- Suitable Histone Acetylator agents include, but are not limited to, Polyamine, CREB (cAMP element binding protein), and BniP3.
- DNA demethylator refers to an agent that removes the methyl groups from DNA and possesses the ability to inhibit hypermethylation and restore suppressed gene expression. A demethylase is expected to activate genes by removing the repressive methyl residues. Suitable DNA demethylators include, but are not limited to, MBD2 and Gadd45b.
- the reprogramming agent has one or more of the following functions: it decrease the expression of one or more markers of cells of the first type (ex. see Table C), and/or increase the expression of one or more markers of the desired cell of the different type (ex. see Table A). Cells that exhibit a selectable marker for the desired cell of a different type are then selected and assessed for characteristics of the desired cell of a different type.
- transformation into the desired cell results in stable expression of a plurality of secondary genes whose expression is characteristic of phenotypical and/or functional properties of the desired cell.
- Genes whose expression is characteristic of phenotypical and/or functional properties of the desired cell include, but is not limited to, those listed in Table A.
- expression of secondary genes whose expression is characteristic of phenotypical and functional properties of the desired cell results in the expression of markers defined according to the following table:
- transformation of a cell of a first type into the desired cell results in a stable repression of a plurality of genes typically expressed in the cell of the first type.
- suppressed genes include, but are not limited to, those defined in Table C:
- PROM1 PROM1 , NM_006017.2 043490 Hs.614734 Leptin isoform 1
- PROM1 PROM1 , NM_001145847.1 043490 Hs.614734
- PROM1 PROM1 , NM_001145852.1 043490 Hs.614734
- PROM1 PROM1 , NM_001145851.1 043490 Hs.614734
- PROM1 PROM1 , NM_001 145850.1 043490 Hs.614734
- PROM1 PROM1 , NM_001145849.1 043490 Hs.614734
- stable repression of any one or more of the genes listed in Table C being expressed in the first cell type is also characterized by a disappearance of the corresponding markers (see Table C).
- Those include destabilizing the cell's cytoskeletal structure (for example, by exposing the cell to cytochalasin B), loosening the chromatin structure of the cell (for example, by using agents such as 5 azacytidine (5-Aza) and Valproic acid (VPA) or DNA demethylator agents such as MBD2), transfecting the cell with one or more expression vector(s) containing at least one cDNA encoding a neurogenic transcription factor (for example, Msi1 or Ngn2), using an appropriate medium for the desired cell of a different type and an appropriate differentiation medium to induce differentiation commitment of the desired cell of a different type, inhibiting repressive pathways that negatively affects induction into commitment the desired cell of a different type, growing the cells on an appropriate substrate for the desired cell of a different type (for example, laminin for NSLCs or a low-bind surface for culturing floating neurospheres), and growing the cells in an environment that the desired cell of a different type (or "-like" cell)
- the method of transforming a cell of a first type into a cell of a second different type may comprise additional facultative steps.
- the method of transforming a cell further comprises the step of pretreating the cell of a first type with a cytoskeleton disruptor.
- cytoskeleton refers to the filamentous network of F-actin, Myosin light and heavy chain, microtubules, and intermediate filaments (IFs) composed of one of three chemically distinct subunits, actin, tubulin, or one of several classes of IF protein.
- cytoskeleton disruptor refers to any molecules that can inhibit the cell cytoskeleton to destabilize the cell and consequently remove the feedback mechanisms between the cell's shape and cellular and nuclear function.
- Suitable cytoskeleton disruptor according to the invention include, but are not limited to, the cytochalasin family of actin cytoskeleton inhibitors, such as Cytochalasin B or D, and myosin inhibitors such as 2,3-butanedione monoxime. Such pretreatment may boost reprogramming.
- the cell is cultured in the presence of at least one cytoskeleton inhibitor one day before, during, or after introducing a neurogenic transcription factor(s).
- Placing the cell in conditions in conditions for supporting the transformation of the desired cell, and/or maintaining the cell in culture conditions supporting the transformation of the desired cell may comprises culturing the cell in a media comprising one or more factors appropriate for inducing the expression of the morphological and functional characteristics of the desired desired cell of a different type.
- the one or more factors are reprogramming factors helpful in reprogramming a cell and these reprogramming factors can be used alone or in combinations.
- the step of culturing the cell in a media comprising one or more factors appropriate for inducing the expression of the morphological and functional characteristics of the desired desired cell of a different type is carried out subsequently or simultaneously to steps iii) or iv), or subsequently or simultaneously to steps 1 ) or 2), as defined hereinbefore.
- a media comprising one or more factors appropriate for inducing the expression of the morphological and functional characteristics of the desired desired cell of a different type is carried out subsequently or simultaneously to steps iii) or iv), or subsequently or simultaneously to steps 1 ) or 2), as defined hereinbefore.
- the reprogramming factor is selected according to TABLE B.
- reprogramming factors have one or more of the following functions: decrease the expression of one or more markers of the first type of cell and/or increase the expression of one or more markers of the desired cell.
- Cells that exhibit a selectable marker for the desired cell are then selected and assessed for unipotency, multipotency, pluripotency, or similar characteristics (as appropriate).
- the cells are cultured in serum-free medium before, during or after any one of steps i) to iv) as defined hereinbefore, or during or after steps 1 ) or 2), as defined hereinbefore.
- NSCs Neural Stem-Like Cells
- the methods of the invention are carried out such that cells are treated with selected agents, compounds and factors to promote the reprogramming and/or dedifferentiation towards Stem-Like Cells (SLCs). Such reprogrammed somatic cells can then be further treated with agents and/or cultured under conditions suitable for promoting reprogramming towards Neural Stem-Like Cells (NSLCs), and expansion of the NSLCs for the long-term.
- NSLCs according to the invention have the potential to differentiate to neuronal-like and/or glial-like cells, as well as neuronal and/or glial cells, for potential treatment of neurological diseases and injuries such as Parkinson's disease and spinal cord injury. The methods described herein are also useful for producing histocompatible cells for cell therapy.
- some aspects of the present invention relates to generating neurons from an individual patient, thus making autologous transplantations possible as a treatment modality for many neurological conditions including neurotrauma, stroke, neurodegenerative diseases such as Multiple Sclerosis, Parkinson's disease, Huntington disease, Alzheimer's diseases.
- the invention provides for neurological therapies to treat the disease or trauma of interest.
- NSC Neural Stem-Like Cell
- the step 1 ) comprises increasing intracellular levels of a MUSASHI1 polypeptide.
- this can be achieved by different means including, but not limited to, transient expression of the MUSASHI1 polypeptide, preferably by transfecting an expression vector encoding the polypeptide.
- the step 2) comprises increasing intracellular levels of a MBD2 polypeptide or treating the cells with VPA and 5-AZA.
- this can be achieved by different means including, but not limited to, transient expression of the MBD2 polypeptide, preferably by transfecting an expression vector encoding the polypeptide(s), and/or pre-treating and/or treating the cells with VPA and 5-AZA.
- reprogramming a cell of a first type to another type of cell that exhibits at least two selectable markers for neural stem cells requires transfecting the cell of a first type with one vector containing a cDNA encoding for a neurogenic transcription factor and one DNA demethylator.
- the cells are exposed or pre-exposed to an agent(s) that inhibits DNA methylation, inhibits histone deacetylation, and/or disrupts the cell cytoskeleton.
- the dedifferentiation can be enhanced by pre-treating the cells with an agent that disrupts the cell cytoskeleton followed by transfecting the cells with one or more vector(s) containing two neurogenic transcription factors in the presence of a DNA demethylator and/or inhibitor of DNA methylation and histone deacetylation.
- the histone deacetylator, inhibitor of histone deacetylation, DNA demethylator, and/or an inhibitor of DNA methylation are as defined previously.
- the method may further comprise a preliminary step of pre-treating the cell of a first type with a cytoskeleton disruptor, as defined previously, and/or culturing the cell in a media comprising one or more reprogramming factors appropriate for appearance and maintenance of the morphological and functional characteristics of NSLCs as defined previously (e.g. a retinoid compound, a neurotrophic factor, bFGF, EGF, SHH, Wnt 3a, , neuropeptide Y, Estrogen).
- the method further comprises inhibiting cellular BMP signaling pathways (e.g. by NOGGIN, fetuin, or follistatin).
- generation of a NSLC from a first cell comprises the use of one or more reprogramming agents.
- Suitable agents include, but are not limited to, Musashi-1 (Msi1 ) and Neurogenin 2 (Ngn2). Other potential agents are listed in Table A and B.
- the present invention is also directed to the use of DNA expression vectors encoding a protein or transcript which upregulates the expression of neurogenesis.
- the genetically-engineered DNA sequence, encoding a defined reprogramming agent such as Msi1 and Ngn2 can be introduced into cells by using a mono-, bi-, or poly-cistronic vectors.
- an endogenous multipotency gene indicates that the cDNA encodes a protein whose expression in the cell result directly or indirectly in the de-differentiation of the cell.
- the newly de-differentiated mammalian cells are capable of re-differentiating to neuronal lineages to regenerate said mammalian cells, tissues, and organs.
- the present invention is further directed to a method for generating NSLCs by introducing a genetically-engineered DNA sequence into human somatic cells via transient transfection. Since the DNA introduced in the transfection process is not inserted into the nuclear genome, the foreign DNA decreases over time and when the cells undergo mitosis. Nonviral vectors remain in a non-replicative form, have low immunogenicity, and are easy and safe to prepare and to use. Furthermore, plasmids may accommodate large fragments of DNA.
- the method starts with obtaining cells from the individual, and reprogramming the cells in vitro to generate NSLCs.
- the significant aspect of the present invention is the stable reprogramming of a somatic cell or non-neuronal cell into a NSLC that can give rise to different types of, neuronal or glial cells (including neuronal-like or glial-like cells). These can then be implanted back into the same patient from which the cells were obtained, thus making an autologous treatment modality for many neurological conditions including neurotrauma, stroke, and neurodegenerative disease possible. These can also be implanted into a different individual from which the cells were obtained. Accordingly, the cells and methods of the present invention may be helpful to treat, prevent, or to stabilize a neurological disease such as Alzheimer's disease, Parkinson's disease, multiple sclerosis, or spinal cord injury.
- a neurological disease such as Alzheimer's disease, Parkinson's disease, multiple sclerosis, or spinal cord injury.
- This technology provides an ample source of neural stem cells, neuro- progenitor cells, neurons and glia for clinical treatment, which can be performed by implantation of NSLCs in vivo or inducing the differentiation in vitro and implantation of neuro-progenitor cells or specific neurons or glia in vivo.
- the method comprises isolating somatic or non-neuronal cells and exposing the cells to one or more agents that alter cell morphology and chromatin structure, and transfecting the cells with one or more genes containing at least one cDNA encoding for a neurogenic transcription factor.
- the gene transfection step may be replaced with alternative agents that induce the expression of the neurogenic transcription factor(s) in the cell.
- Inducing epigenetic modifications to DNA and histones (especially DNA demethylation and an open chromatic structure) facilitate true reprogramming of the cells.
- the cells are incubated in a low oxygen environment, for example 5% 0 2 , thereby helping in reprogramming the cells.
- This methodology allows the reprogramming of a cell into a NSLC.
- the further course of development and the expansion of the reprogrammed cell depend on the in situ environment cues to which it is exposed.
- the embodiments of the invention further include growing the reprogrammed cell in an appropriate proliferation medium to expand the generated NSLC, for example Neural Progenitor proliferation Medium (StemCell Technologies) with the presence of epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF), to promote the neural stem cell to proliferate.
- an appropriate proliferation medium for example Neural Progenitor proliferation Medium (StemCell Technologies) with the presence of epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF), to promote the neural stem cell to proliferate.
- EGF epidermal growth factor
- bFGF basic fibroblast growth factor
- the NSLCs obtained according to the invention can be differentiated into neuronal, astrocyte, and/or oligodendrocyte lineages in appropriate differentiation medium, for example NS-A differentiation medium (StemCell, Technologies) or NbActive medium (BrainBitsTM) including a retinoid compound, such as all-trans-retinoic acid or vitamin A, and BDNF, to induce the differentiation of NSLCs towards neuronal and/or glial cells.
- NS-A differentiation medium Stein Cell, Technologies
- NbActive medium Neuronal cells include cells that display one or more neural-specific morphological, physiological, functional and/or immunological features associated with a neuronal cell type.
- Useful criteria features includes: morphological features (e.g., long processes or neurites), physiological and/or immunological features such as expression of a set of neuronal-specific markers or antigens, synthesis of neurotransmitter(s) such as dopamine or gamma aminobutyric acid (GABA), and functional features such as ion channels or action potentials characteristic of neurons.
- morphological features e.g., long processes or neurites
- physiological and/or immunological features such as expression of a set of neuronal-specific markers or antigens
- synthesis of neurotransmitter(s) such as dopamine or gamma aminobutyric acid (GABA)
- functional features such as ion channels or action potentials characteristic of neurons.
- reprogrammed cells can be selected based on differential adherence properties as compared to untransfected cells; for example, reprogrammed cells can form floating neurospheres or grow well on laminin while untransfected fibroblasts attach and grow well on regular cell culture treated plates.
- Reprogrammed cells include cells that exhibit one or more neural stem specific markers and morphology and the loss of some or all of the specific markers related to the original cells.
- some of the functionality of the neural-like cells (NLCs) can be assessed at different time points by, for example, patch- clamping, immunostaining for synaptophysin and MAP2b, and by immunochemical means such as by enzyme-linked immunosorbent assay (ELISA).
- ELISA enzyme-linked immunosorbent assay
- the present invention provides NSLCs that are able to initiate and direct central nervous system regeneration at a site of tissue damage and can be customized for individual patients using their own cells as the donor or starting cell.
- the present invention can be used to generate cells from an individual patient, thus making autologous transplantations possible as a treatment modality for many neurological conditions.
- this technology eliminates the problems associated with transplantations of non-host cells, such as, immunological rejection and the risk of transmitted disease.
- the great advantage of the present invention is that it provides an essentially limitless supply for autologous grafts suitable for transplantation. Therefore, it will obviate some significant problems associated with current source of materials and methods of transplantation.
- the invention concerns the use of polynucleotides, e.g. a polynucleotide encoding a MBD2 polypeptide, a MUSASHI1 polypeptide and/or a Ngn2 polypeptide.
- a polynucleotide encoding a MBD2 polypeptide e.g. a MBD2 polypeptide, a MUSASHI1 polypeptide and/or a Ngn2 polypeptide.
- Means for introducing polynucleotides into a cell are well known in the art. Transfection methods of a cell such as nucleofection and/or lipofection, or other types of transfection methods may be used.
- a polynucleotide encoding a desired polypeptide can be cloned into intermediate vectors for transfection in eukaryotic cells for replication and/or expression.
- Intermediate vectors for storage or manipulation of the nucleic acid or production of protein can be prokaryotic vectors, (e.g., plasmids), shuttle vectors, insect vectors, or viral vectors for example.
- a desired polypeptide can also be encoded by a fusion nucleic acid.
- a cloned nucleic acid is typically subcloned into an expression vector that contains a promoter to direct transcription.
- Suitable bacterial and eukaryotic promoters are well known in the art and described, e.g., in Sambrook and Russell (Molecular Cloning: a laboratory manual, Cold Spring Harbor Laboratory Press).
- the promoter used to direct expression of a nucleic acid of choice depends on the particular application. For example, a strong constitutive promoter is typically used for expression and purification. In contrast, when a dedifferentiation protein or compound is to be used in vivo, either a constitutive or an inducible promoter or compound is used, depending on the particular use of the protein.
- a weak promoter can be used, such as HSV TK or a promoter having similar activity.
- the promoter typically can also include elements that are responsive to transactivation, e.g., hypoxia response elements, Ga14 response elements, lac repressor response element, and small molecule control systems such as tet-regulated systems and the RU-486 system.
- an expression vector typically contains a transcription unit or expression cassette that contains additional elements required for the expression of the nucleic acid in host cells, either prokaryotic or eukaryotic.
- a typical expression cassette thus contains a promoter operably linked, e.g., to the nucleic acid sequence, and signals required, e.g., for efficient polyadenylation of the transcript, transcriptional termination, ribosome binding, and/or translation termination. Additional elements of the cassette may include, e.g., enhancers, and heterologous spliced intronic signals.
- Expression vectors containing regulatory elements from eukaryotic viruses are often used in eukaryotic expression vectors, e.g., SV40 vectors, papilloma virus vectors, and vectors derived from Epstein-Barr virus.
- eukaryotic vectors include pMSG, pAV009/A+, pMTO10/A+, pMAMneo-5, baculovirus pDSVE, and any other vector allowing expression of proteins under the direction of the SV40 early promoter, SV40 late promoter, metallothionein promoter, murine mammary tumor virus promoter, Rous sarcoma virus promoter, polyhedrin promoter, or other promoters shown effective for expression in eukaryotic cells.
- Standard transfection methods can be used to produce bacterial, mammalian, yeast, insect, or other cell lines that express large quantities of dedifferentiation proteins, which can be purified, if desired, using standard techniques. Transformation of eukaryotic and prokaryotic cells is performed according to standard techniques.
- Any procedure for introducing foreign nucleotide sequences into host cells can be used. These include, but are not limited to, the use of calcium phosphate transfection, DEAE-dextran- mediated transfection, polybrene, protoplast fusion, electroporation, lipid-mediated delivery (e.g., liposomes), microinjection, particle bombardment, introduction of naked DNA, plasmid vectors, viral vectors (both episomal and integrative) and any of the other well known methods for introducing cloned genomic DNA, cDNA, synthetic DNA or other foreign genetic material into a host cell (see, e.g., Sambrook et al., supra). It is only necessary that the particular genetic engineering procedure used be capable of successfully introducing at least one gene into the host cell capable of expressing the protein of choice.
- Non-viral vector delivery systems include DNA plasmids, naked nucleic acid, and nucleic acid complexed with a delivery vehicle such as a liposome
- virai vector delivery systems include DNA and RNA viruses, which have either episomal or integrated genomes after delivery to the cell.
- nucleic acids include lipofection, microinjection, ballistics, virosomes, liposomes, immunoliposomes, polycation or lipid-nucleic acid conjugates, naked DNA, artificial virions, and agent-enhanced uptake of DNA.
- Lipofection reagents are sold commercially (e.g., TransfectamTM and LipofectinTM). Cationic and neutral lipids suitable for efficient receptor-recognition lipofection of polynucleotides are known.
- Nucleic acid can be delivered to cells (ex vivo administration) or to target tissues (in vivo administration). The preparation of lipid:nucleic acid complexes, including targeted liposomes such as immunolipid complexes, is well known to those of skill in the art.
- RNA or DNA virus-based systems for the delivery of nucleic acids take advantage of highly evolved processes for targeting a virus to specific cells in the body and trafficking the viral payload to the nucleus.
- Viral vectors can be administered directly to patients (in vivo) or they can be used to treat cells in vitro, wherein the modified cells are administered to patients (ex vivo).
- Conventional viral based systems for the delivery include retroviral, lentiviral, poxviral, adenoviral, adeno-associated viral, vesicular stomatitis viral and herpesviral vectors, althoughntegration in the host genome is possible with certain viral vectors, including the retrovirus, lentivirus, and adeno-associated virus gene transfer methods, often resulting in long term expression of the inserted transgene. Additionally, high transduction efficiencies have been observed in many different cell types and target tissues. pLASN and MFG-S are examples of retroviral vectors that have been used in clinical trials. In applications for which transient expression is preferred, adenoviral-based systems are useful.
- Adenoviral based vectors are capable of very high transduction efficiency in many cell types and are capable of infecting, and hence delivering nucleic acid to, both dividing and non-dividing cells. With such vectors, high titers and levels of expression have been obtained. Adenovirus vectors can be produced in large quantities in a relatively simple system.
- Gene therapy vectors can be delivered in vivo by administration to an individual patient, typically by systemic administration (e.g., intravenous, intraperitoneal, intramuscular, subdermal, or intracranial infusion) or topical application.
- vectors can be delivered to cells ex vivo, such as cells explanted from an individual patient (e.g. , lymphocytes, bone marrow aspirates, tissue biopsy) or universal donor hematopoietic stem cells, followed by reimplantation of the cells into a patient, usually after selection for cells which have been reprogrammed.
- Ex vivo cell transfection for diagnostics, research, or for gene therapy is well known to those of skill in the art.
- cells are isolated from the subject organism, transfected with a nucleic acid (gene or cDNA), and re-infused back into the subject organism (e.g., patient).
- a nucleic acid gene or cDNA
- Various cell types suitable for ex vivo transfection are well known to those of skill in the art.
- Vectors e.g., retroviruses, adenoviruses, liposomes, etc.
- therapeutic nucleic acids can be also administered directly to the organism for transfection of cells in vivo.
- naked DNA can be administered.
- Administration is by any of the routes normally used for introducing a molecule into ultimate contact with blood or tissue cells. Suitable methods of administering such nucleic acids are available and well known to those of skill in the art, and, although more than one route can be used to administer a particular composition, a particular route can often provide a more immediate and more effective reaction than another route.
- compositions of the present invention are determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions of the present invention. Delivery of Polypeptides
- an alternative possibility consists of bypassing the use of a polynucleotide and contacting a cell of a first type cell directly with a compound (e.g. a polypeptide) for which an increased intracellular level is desired.
- a compound e.g. a polypeptide
- the cells are cultured in a medium containing one or more functional polypeptides.
- polypeptides have the ability to traverse the plasma membrane of a cell, or the membrane of an intra-cellular compartment such as the nucleus.
- Cellular membranes are composed of lipid-protein bilayers that are freely permeable to small, nonionic lipophilic compounds and are inherently impermeable to polar compounds, macromolecules, and therapeutic or diagnostic agents.
- proteins, lipids and other compounds, which have the ability to translocate polypeptides across a cell membrane have been described.
- membrane translocation polypeptides have amphiphilic or hydrophobic amino acid subsequences that have the ability to act as membrane-translocating carriers.
- Polypeptides for which an increased intracellular level is desired according to the invention can be linked to suitable peptide sequences for facilitating their uptake into cells.
- suitable chemical moieties that provide enhanced cellular uptake can also be linked, either covalently or non-covalently, to the polypeptides.
- suitable carriers having the ability to transport polypeptides across cell membranes may also be used.
- a desired polypeptide can also be introduced into an animal cell, preferably a mammalian cell, via liposomes and liposome derivatives such as immunoliposomes.
- liposome refers to vesicles comprised of one or more concentrically ordered lipid bilayers, which encapsulate an aqueous phase.
- the aqueous phase typically contains the compound to be delivered to the cell.
- the invention encompasses the cells, cell lines, stem cells and purified cell preparations derived from any of the methods described herein.
- the cells, cells lines, stem cells and purified cells preparations of the invention are of mammalian origins, including but not limited to human, primates, rodent, dog, cat, horse, cow, or sheep. In preferred embodiments, they originate from a human.
- another aspect of the invention relates to modified cells, cell lines, pluripotent, multipotent or unipotent cells and purified cell preparations, wherein any of these cells comprise an exogenous polynucleotide encoding Musashh (Msi1 ); Msi1 and Ngn2; Msi1 and MBD2; and Ngn2 and MBD2; Msi1 , Ngn2 and MBD2; Msi1 , Ngn2, Nestin and MBD2; and other potential combinations from Table A preferably including Msi1 and Ngn2 and MBD2.
- the cell according to the invention is a stem-like cell, more preferably a Neural Stem-Like Cell (NSLC), the cell possessing one or more of the following characteristics:
- NSLC Neural Stem-Like Cell
- telomerase decreased expression of telomerase and one or more neural stem cell markers after differentiation
- - having one or more morphological neurite-like processes (axons and/or dendrites) greater than one cell diameter in length after differentiation into a neuronal-like cell;
- neural-specific antigen selected from the group consisting of neural-specific tubulin, microtubule associated protein 2, NCAM, and marker for a neurotransmitter after differentiation into a neuronal-like cell;
- one or more functional neural markers e.g. synapsin
- BDNF neurotrophic factor
- a NSLC according to the inventions possesses all of the following characteristics:
- - is not a cancerous cell
- - is stable and not artificially maintained by forced gene expression or by similar means and may be maintained in standard neural stem cell media;
- - can differentiate to a progenitor, precursor, somatic cell or to another more differentiated cell type of the same lineage;
- - has the characteristics of a stem cell and not just certain markers or gene expression or morphological appearance
- the reprogrammed cells (NSLCs) according to the invention are capable of proliferating for several months without losing their neural stem cell markers and their ability to differentiate towards neuron-like, astrocyte-like, and oligodendrocyte-like cells.
- the generation of the neural lineages is characterized based on morphology, phenotypic changes and functionality.
- the cells of the invention may have one or more of the following characteristics and properties: self-renewal, multilineage differentiation in vitro and in vivo, clonogenicity, a normal karyotype, extensive proliferation in vitro under well defined culture conditions, and the ability to be frozen and thawed, as well as any of the commonly known and/or desired properties or characteristics typical of stem cells.
- the cells of the invention may further express molecular markers of multipotent or pluripotent cells (i.e. gene and surface markers as defined previously).
- tissue specific autologous (self) stem and/or progenitor cells may be used in cell therapy applications to treat diseases of cellular degeneration.
- Diseases of cellular degeneration include, for example, neurodegenerative diseases such as stroke, Alzheimer's disease, Parkinson's disease, multiple sclerosis, Amyotrophic lateral sclerosis, macular degeneration, osteolytic diseases such as osteoporosis, osteoarthritis, bone fractures, bone breaks, diabetes, liver injury, degenerative diseases, myocardial infarct, burns and cancer. It is envisioned that cells according to the invention may be implanted or transplanted into a host.
- An advantage of the invention is that large numbers of autologous stem cells can be produced for implantation without the risk of immune system mediated rejection. Those cells can lead to production of tissue suitable for transplant into the individual. Since the tissue is derived from the transplant recipient, it should not stimulate an immune response, as would tissue from an unrelated donor.
- Such transplants can constitute tissues (e.g. vein, artery, skin, muscle), solid organ transplants (e.g., heart, liver, kidney), neuronal cell transplants, or bone marrow transplants such as are used in the treatment of various malignancies such as, for example, leukemias and lymphomas.
- Neural stem cell, neuroprogenitor, or neuronal cell (as well as NSLCs and derivations thereof) transplants can also be used in the treatment of, for example, neurological disorders, stroke, spinal cord injury, Parkinson's disease, and the like, as well as potentially some non- neurological disorders such as a cardiac infarct.
- Another aspect of the invention relates to a method to produce ex vivo engineered tissues for subsequent implantation or transplantation into a host, wherein the cellular components of those engineered tissues comprise cells according to the invention, or cells derived therefrom.
- expanded cultures of the cells of the invention may be differentiated by in vitro treatment with growth factors and/or morphogens. Populations of differentiated cells are then implanted into the recipient host near the site of injury or damage, or cultured in vitro to generate engineered tissues, as described.
- the methods and cells of the invention described herein can be used to immortalize cells, for example to generate a cell line.
- a somatic cell can be transformed into one possessing a dedifferentiated phenotype, thereby facilitating the generation of cell lines from a variety of tissues. Therefore, the invention encompasses such immortalized cells.
- the methods of deriving the cells according to the invention may be helpful in scientific and therapeutic applications including, but not limited to, (a) scientific discovery and research involving cellular development and genetic research (e.g. uses in lieu of human stem cells as a model cell line to study the differentiation, dedifferentiation, or reprogramming of human cells), (b) drug development and discovery (e.g.
- Additional aspects concern therapeutic methods, methods of treatment and methods of regenerating a tissue or organ in a mammal (e.g. a human subject).
- One particular method concerns a method of regenerating a mammalian tissue or organ which comprises contacting the tissue or organ to be regenerated with a SLC, NSLC, or other desired cell or artificial tissue construct as defined herein.
- the SLC, NSLC, desired cell or artificial tissue construct may be placed in proximity to the tissue or organ to be regenerated by administering to the subject using any suitable route (e.g. injecting the cell intrathecally, directy into the tissue or organ, or into the blood stream).
- Another method for repairing or regenerating a tissue or organ in a subject in need thereof comprises administering to the subject a compound inducing a direct or indirect endogenous expression of at least one gene regulator in cells of the tissue or organ and/or a compound inducing a direct or indirect endogenous expression of at least one gene regulator in cells capable of transformation or dedifferentiation in vivo in the subject.
- the expression of the at least one gene regulator reprograms the cells into desired cells of a different type (e.g. neural stem-like cells), and these cells of a different type are effective in repairing or regenerating said tissue or organ.
- Another method comprises obtaining cells or tissue from a patient (e.g. hematopoietic stem cells, fibroblasts, or keratinocytes), reprogramming a plurality of such cells or the tissue, and reintroducing the reprogrammed cells or tissue into the patient.
- a related aspect concerns pharmaceutical compositions comprising a plurality of a desired cell, SLC and/or Neural Stem- Like Cell (NSLC) or reprogrammed tissue as defined herein.
- the therapeutic methods of the invention may be applicable to the regeneration or repair of various tissues and organs including, but not limited to, the brain, the spine cord, the heart, the eye, the retina, the cochlea, the skin, muscles, intestines, pancreas (including beta cells), kidney, liver, lungs, bone, bone marrow, cartilage, cartilage discs, hair follicles, teeth, blood vessels, glands (including endocrine and exocrine glands), ovaries, reproductive organs, mammary and breast tissue.
- various tissues and organs including, but not limited to, the brain, the spine cord, the heart, the eye, the retina, the cochlea, the skin, muscles, intestines, pancreas (including beta cells), kidney, liver, lungs, bone, bone marrow, cartilage, cartilage discs, hair follicles, teeth, blood vessels, glands (including endocrine and exocrine glands), ovaries, reproductive organs, mammary and breast tissue.
- a related aspect concerns pharmaceutical compositions comprising a plurality of desired cell, SLC and/or Neural Stem-Like Cell (NSLC) as defined herein.
- tissue containing reprogrammed cells as defined herein that can be implanted into a subject in need thereof.
- the present invention provides for the reprogramming of cells within a tissue, for example an in vitro produced 3D tissue construct comprising cells and extracellular matrix produced by these cells.
- transfected cells can be seeded on top of these 3D tissue constructs that can be made completely autologously, thus preventing host rejection, making it completely immunocompatible and as carrier for reprogrammed cells to be transplanted in vivo.
- these newly created cells can be used in their undifferentiated and/or differentiated state within these tissues for in vitro diagnostic purposes or transplanted into a patient in need of such a construct in cell therapy / tissue replacement approaches.
- the invention further encompasses 3D Neuronal-Like multilayer tissue.
- Cells within CDM reprogrammed to Neural Stem-Like Cells according to the invention readily differentiate into neuronal-like cells, astrocyte-like cells, and oligodendrocyte-like cells within the CDM. It is thus possible to use CDM and reprogramming methods of the invention to reprogram the cells within the CDM to form 3D Neuronal-Like multilayer tissue (up to >30 cell layers).
- Such 3D tissue comprises neurons (or specifically, neuron-like cells), astrocytes (or specifically, astrocyte-like cells), and oligodendrocytes (or specifically, oligodendrocyte-like cells) and it can be made completely autologously, can be manually handled and implanted with relative ease, or can used as an in vitro CNS tissue model.
- One particular aspect concerns an artificial tissue construct which comprises a 3D assembly of in vitro cultured cells and extracellular matrix produced by these cells.
- the cells may be desired cells, SLC and/or a plurality of Neural Stem-Like Cell (NSLC) obtained using any one of the methods described herein.
- NSLC Neural Stem-Like Cell
- Another aspect of the invention relates to methods for identifying new compounds (e.g. small molecules, drugs, etc) capable of transforming a cell of a first type to a desired cell of a different type.
- new compounds e.g. small molecules, drugs, etc
- These new compounds may be usefull for research purposes or as medicaments for use in reparing or regenerating tissues in a subject.
- a candidate compound to be tested e.g. a library of small molecules or compounds
- Individual or mixture of active compounds would be selected if they have the same activity and/or if they can provide the same or similar effects as these polypeptides (e.g. cell transformation and/or appearance of any desirable markers or desirable characteristics as defined hereinbefore).
- a compound or mixture of compounds capable of transforming a fibroblast into a NSLC could be identified by:
- stem-like cells including pluripotent-like cells, mesendoderm-like cells, pancreatic progenitor-like cells, etc.
- Tables A and B, and the Examples section provides, for each of these types of cells, a list of potential genes and/or compounds to be considered in such screening methods.
- the present invention encompasses these and any equivalent screening methods where candidate compounds are tested for their efficacy in transforming a cell of a first type to a desired cell of a different type when compared to the efficacy of the reprogramming factor and/or gene regulator as defined herein. Delivery of neurotrophic factors
- a related aspect of the invention relates to overcoming these problems by using NSLC cells and cell lines according to the invention which can stably express and secrete growth factors of potential interest after transplantation.
- the present invention provides a plentiful source of Neural Stem-Like Cells, Neuron-Like Cells, Astrocyte-Like Cells or Oligodendrocyte-Like Cells for potential clinical treatments which require transplantation of neural stem cells, neurons, astrocytes or oligodendrocytes 1 ) to compensate for a loss of host cells (ex. neurons) or 2) as vehicles to deliver genetically-based drugs. Further, the invention provides a novel neurological tool for use in basic research and drug screening.
- NSCs Neural Stem-Like Cells
- exemplary protocols for obtaining Reprogrammed and Dedifferentiated cells, including Neural Stem-Like Cells (NSLCs). Also provided are exemplary protocols, molecular tools, probes, primers and techniques.
- HFF Human Foreskin fibroblast
- DMEM Dulbecco's Modified Eagle's Medium
- FCS heat-inactivated fetal calf serum
- FCS Hyclone Laboratories
- Cells were trypsinized using Trypsin 0.25% for 4 minutes at 37°C, followed by adding trypsin inhibitor solution, pelleting the cells by centrifugation, washing the cells once with PBS, and plating the cells at a ratio of 1 :2 onto tissue culture flasks until a suitable number of cells was reached.
- CDM I Medium consisting of a 3:1 ratio of Dulbecco's modified Eagle medium (DMEM, high glucose (4.5g/L) with L-glutamine and sodium pyruvate) and Ham's F-12 medium supplemented with the following components: EGF (4.2x10 " 0 M), bFGF (2.8x10- 10 M), ITS (8.6x10 "5 M), dexamethasone (1.0x10 "7 M), L-3,3',5-triiodothyronine (2.0x10 '10 M), ethanoiamine ( ⁇ ⁇ ), GlutaMAXTM (4x10 " 3 M), and glutathione (3.3x10 "6 M), but without the presence of L-ascorbic acid.
- DMEM Dulbecco's modified Eagle medium
- ITS 8.6x10 "5 M
- dexamethasone 1.0x10 "7 M
- L-3,3',5-triiodothyronine 2.0x10 '10
- CDM II Medium consisting of a 3:1 ratio of Dulbecco's modified Eagle medium (DMEM, high glucose (4.5g/L) with L-glutamine and sodium pyruvate) and Ham's F-12 medium supplemented with the following components: EGF (2.5 ng/ml), bFGF (10ng/ml), ethanoiamine (2.03mg/ml), insulin (10 mg/ml), Selenious acid (2.5 ⁇ g/ml), dexamethasone (19.7 ⁇ g ml), L- 3,3',5-triiodothyronine (675 ng/ml), GlutaMAXTM (4x10 "3 M), and glutathione (3.3x10 "6 M).
- DMEM Dulbecco's modified Eagle medium
- bFGF 10ng/ml
- ethanoiamine 2.03mg/ml
- insulin 10 mg/ml
- Selenious acid 2.5 ⁇ g/ml
- NPBM Neural Progenitor Basal Medium
- Noggin (20 ng/ml, Peprotech
- EGF (20ng/ml, Peprotech)
- bFGF 20ng/ml, Peprotech
- Proliferation Medium (NbActive, BrainBitsTM) supplemented with Forskolin (10 ⁇ , Tocris), a//-frans-Retinoic Acid (ATRA, 5 ⁇ , Spectrum), bFGF (20ng/ml, Peprotech), NGF (20ng/ml, Peprotech), and BDNF (20ng/ml, Peprotech); medium was changed every day by increasing the percentage of Differentiation Medium over Proliferation Medium, and the cells were cultured for 20 days.
- Differentiation Medium NabActive, BrainBitsTM
- ATRA a//-frans-Retinoic Acid
- bFGF (20ng/ml, Peprotech
- NGF (20ng/ml, Peprotech
- BDNF 20ng/ml, Peprotech
- Characterization of the newly engineering cells after transfection was performed using a neuronal gene-array containing 48 partial cDNAs coding for these genes and controls.
- TLDA TaqmanTM low-density array
- TLDA TaqmanTM low-density array
- Each 2 ⁇ well contains specific, user-defined primers and probes, capable of detecting a single gene.
- a customized Neuronal Markers 2 TLDA was configured into eight identical 48-gene sets, i.e. 1 loading port for each 48-gene set. Genes were chosen based on literature.
- Each set of 48 genes also contains three housekeeping genes: ACTIN, GAPDH, and PPIA.
- a sample-specific master mix was made for each sample by mixing cDNA (160 ng for each loading port), 2X TaqmanTM Gene Expression Master Mix (Applied Biosystems) and nuclease-free water (USB) for a total of 100 ⁇ per loading port. After gentle mixing and centrifugation, the mixture was then transferred into a loading port on a TLDA card. The array was centrifuged twice for 1 minute each at 1200 rpm to distribute the samples from the loading port into each well. The card was then sealed and PCR amplification was performed using Applied Biosystems 7900HTTM Fast Real-time PCR system. Thermal cycler conditions were as follows: 2 minutes at 50°C, 10 minutes at 94.5°C, and 30 seconds at 97°C, 1 minute at 59.7°C for 40 cycles. 1 TLDA's was prepared for 8 samples.
- Relative Expression values were calculated using the Comparative C T method. Briefly, this technique uses the formula 2 "AACT to calculate the expression of target genes normalized to a calibrator.
- the threshold cycle (C T ) indicates the cycle number at which the amount of amplified target reaches a fixed threshold. C T values range from 0 to 40 (the latter representing the default upper limit PCR cycle number that defines failure to detect a signal).
- oligodendrocyte gene expression allowed identification of the majority of the genes that are differentially expressed in reprogrammed cells. Data in Table 1 were analyzed by using a significance analysis algorithm to identify genes that were reproducibly found to be enriched in reprogrammed cells compared to untransfected cells. After the transfection with Msi1 or Ngn2 in the presence of MBD2, the expression of oligodendrocytes progenitors such as NKx2.2, olig2, and MAG and two markers for astrocytes (GFAP and AQP4) were highly increased. Also, several markers of early neuronal cells were enhanced after the transfection of HFF.
- TDLA data revealed a remarkable increase in specific markers for interneurons, such as somatostatin and calbindinl
- DCX Doublecortin
- ACHE acetylcholine
- Transfection increased the expression of dihydropyrimidinase-like 3 (DPYSL3), an early marker of newborn neurons, to fivefold with Msi1 and seven fold with Ngn2.
- MAP2 Microtubule-Associated Protein 2
- GAP-43 growth associated protein
- -fibronectin is a marker for fibroblasts
- Ceiis were fixed with a 4% formaidehyde /PBS solution for l Omin ai room temperature and subsequently permeabilized for 5min with 0.1 % Triton X-100TM in 4% formaldehyde/PBS. After two brief washes with PBS, unspecific antibody binding was blocked by a 30min incubation with 5% normal goat serum in PBS. Then primary antibodies were added in 5% normal goat serum / PBS as follows: Mouse anti-Nestin (1 :100, BD) as an intermediate microfilament present in neural stem cells and mouse anti-NCAM (1 :100, Neuromics) as neuronal adhesion molecule.
- NCAM was present in cells during the 6 days post-transfection and increase at day 12 and 20 following differentiation, while the inverse pattern was observed for the nestin staining.
- This study showed the ability to reprogram HFF cells using one neurogenic transcription factor with the presence of a DNA demethylator towards cells that expressed neuronal genes and proteins specific to neural stem cells and neuronal cells. These reprogrammed cells were stable in culture for at least 2 weeks.
- HFF cells were cultured as described in Example I and plated in CDM I medium. Cells were transfected using the Amaxa NucleofectorTMTM Device (Lonza). The HFFs were harvested with TrypLETM (Gibco), resuspended in CDM Medium and centrifuged for 10 min at 90xg (1x10 s cells / tube). The supernatant was discarded and gently resuspended in 100 ⁇ of Basic NucleofectorTM Solution (basic NucleofectorTM kit for primary mammalian fibroblasts, Lonza).
- Basic NucleofectorTM Solution basic NucleofectorTM kit for primary mammalian fibroblasts, Lonza.
- Each 100 ⁇ of cell suspension was combined with a different mix of plasmid DNA (for example, sample 1 was mixed with 2 ⁇ of pCMV6-XL5-Pax6 and 2 g pCMV6-XL5-MBD2).
- Cell suspension was transferred into an Amaxa certified cuvette and transfected with the appropriate program (U023).
- the sample was transferred without any further resuspension into a coated culture plate with LAS-Lysine/Alanine (BrainBitsTM, ⁇ / ⁇ ) and the cells were incubated at 37°C, 5% C0 2 . These steps were repeated for each sample that was transfected. After 24 hours, the medium was changed to Proliferation Medium.
- RNA isolation and quantification was performed as previously described in Example I.
- cDNA was prepared using the High Capacity cDNA RT kit (Applied Biosystems) as per the manufacturer's instructions with a final cDNA concentration of 2ng ⁇ l.
- Real-time PCR was then performed for each gene of interest using the FAST PCR master mix (Applied Biosystems) and the TaqmanTM® Gene Expression Assays (Applied Biosystems) listed below:
- GAPDH (housekeeping gene) Hs99999905 ml
- the FAST 96-well reaction was performed with 8ng cDNA per well in a 10 ⁇ reaction with 40 cycles.
- Thermal cycler conditions were as follows: 20 seconds at 95°C, and 1 second at 95°C, 20 seconds at 60°C for 40 cycles.
- Relative Expression values were calculated as previously described in Example I, except the Average of 2 Housekeeping genes (GAPDH & PPIA) was used for normalization instead of the Average of 3 Housekeeping genes. Identification of neuronal lineage genes was investigated following the transfection with three independent vectors containing Msi1 , Ngn2, and Pax6.
- mRNAs level of the tripotent-associated genes ⁇ - tubulin, MAP2b, acetycholine, and GFAP were undetectable in Pax6 transfected cells, indicating that Pax6 alone was not Implicated In the reprog ramming process toward neuronal lineage.
- Table 2 Relative expression of gene expression of different neuronal lineage performed by RT-PCR following the transfection of HFF by Msi1 , Ngn2, or Pax6 in the presence of MBD2 and cultured for 14 days.
- Cells were co- transfected with either of two DNA demethylators, MBD2 or GAdd45B, (e.g. 2x10 6 cells were transfected with pCMV6-XL5Msi1 ⁇ g) and pCMV6-XL5-MBD2 ⁇ g)).
- Neural Proliferation Medium NeuroCultTM proliferation Kit, StemCell Technologies
- DMEM/F12 1 :1
- glucose 0.8%
- sodium bicarbonate 0.1 %
- glutamine 20 mM
- HEPES 5mM
- insulin 230 ⁇ g/ml
- transferrin 100 ⁇ g/ml
- progesterone 200nM
- putrescine 90 ⁇ g/ml
- sodium selenite 300nM
- RT-PCR analysis showed that the relative expression of Sox2, nestin and GFAP was enhanced after transfecting the cells with the neurogenic transcription factors.
- Table 3 transfecting the cells with one transcription factor Msi1 in the presence of Gadd45b was associated with up-regulation of relative expression of Sox2 (22.3+5.26) and GFAP (10.14+0.15) and the expression of the these genes was highly increase when transfecting the cells with Ngn2 by 20 fold and 10 fold respectively.
- Combining the two neurogenic fators (Msi1 and Ngn2) with Gadd45b enhanced further the expression of Sox2 and GFAP.
- Transfecting the cells with one transcription factor (Msi1 or Ngn2) in the presence of MBD2 was associated with up-regulation of relative expression of Sox2, Nestin, and GFAP and down-regulation of Col5A2, while co-tranfection with Gadd45b did not increased the expression of nestin and the expression of Col5A2 was not regulated.
- the enhancement of neural stem cells relative expression was observed when transfecting the cells with two neurogenic genes in combination with MBD2; a small increase in the expression was noticed in the presence of cytochalasin B under certain conditions.
- Table 3 RT-PCR analysis of relative expression of neuronal precursor cell markers such as nestin, Sox2, and GFAP after transfection of fibroblast cells with different combinations with or without the co-treatment with cytochalasin B.
- Relative expression of Sox2, nestin, and GFAP in NSLCs was increased after transfection with both transcription factors (Ngn2 and Msi1 ) with MBD2 as the DNA demethyaltor.
- this upregulation of neural stem cell gene expression was associated with a decrease of CoL5A2, a specific gene for fibroblast cells.
- Cells were co-transfected pCMV-Msi1-Ngn2 with different DNA demethylators (MBD1 , MBD2, MBD3, MBD4, MeCP2, AICDA). Another assay was performed to assess the effect of nestin on the reprogramming efficiency; therefore cells were transfected with nestin individually or in combination with one vector containing one neurogenic factor (Msi1 or Ngn2) or both neurogenic factors in the presence of MBD2. Cells were cultured following transfection in the presence of proliferation medium supplemented with EGF (20ng/ml), FGF (20ng/ml), and Noggin (20ng/ml) with and without VPA (1 mM) treatment for 12 days at 37°C, 5% C0 2 and 5%0 2 .
- Table 5 RT-PCR analysis of relative expression of neuronal precursor cell markers such as nestin, Sox2, ⁇ -tubulin, and GFAP after transfection of fibroblast cells with various combinations of pCMV-Msi1 -Ngn2 (MSI 1/NGN2), pCMV-XL5-Msi1 , pCMV-XL4-Ngn2, pCMV- XL-Nestin with different combinations of DNA demethylators, with and without the co-treatment with VPA.
- Immunohsitochemtstry analysis revealed a small increase of immunopositive Sox2 cells (89.49 ⁇ 3.18) after co-transfecting the cells with nestin and Msi1/Ngn2 in the presence of MBD2 (Table 6).
- Table 6 Percentage of positive cells for Sox2 after transfection of fibroblast cells with different expression vectors with or without the presence of various DNA demethylators. After transfection the cells were cultured in proliferation medium (StemCell Technologies) supplemented by EGF (20ng/ml, Peprotech) and FGF (20ng/ml, Peprotech) for two weeks at 37°C/5%C0 2 /5%0 2 . The percentage of immunopositive cells was determined by CellomicsTM
- HFF cells were cultured as described in Example I, and transfected using the NucleofectorTM 1® 96-well Shuttle ® Device (Lonza) following procedure described in Example IV, except for the untreated HFF control and the untransfected HFF control (for determining the effect of the complete media & compound treatments on the cells).
- the cells that had been pre-treated with VPA and 5-Aza and the untreated cells were transfected with the mixes of DNA as described in Table 7.
- the cells were plated on Laminin-coated plates and incubated at 37°C, 5% C0 2 . Media was changed daily according to Table 7. Cells were analysed at day 3, 7, 12 by immunohistochemistry analysis and at Day 9 by gene array for multipotent and pluripotent gene expression.
- Table 7 Plasmids and media composition from day 1 to day 12.
- each sample was normalized to the average Ct of the 6 housekeeping genes (ACTB, 18S, CTNNB1 , EEF1A1 , GAPD, RAF1 ), and calibrated to the Untreated HFF (Passage 8) control.
- Relative Expression values with asterisk (*) indicate values with significant up or down-regulation (>2-fold or ⁇ 0.5-fold). For these samples, for Ct values ⁇ 35 is considered that the expression of the gene is adequate for quantification.
- the values could have significant error due to the low expression of the gene (Ct>35), and thus the up or down-regulation could be merely a result of the high standard deviation of the high Ct values of the genes, or fluctuations in the housekeeping genes.
- the Relative Expression values that are between 0.5-fold and 2-fold it indicates no significant change in the expression of the gene for these samples.
- each sample was normalized to the average Ct of the 6 housekeeping genes (ACTB, 18S, CTNNB1 , EEF1 A1 , GAPD, RAF1 ), and calibrated to the Untreated HFF (Passage 8) control.
- Relative Expression values with asterisk (*) indicate values with significant up or down-regulation (>2-fold or ⁇ 0.5-fold). For these samples, for Ct values ⁇ 35 is considered that the expression of the gene is adequate for quantification.
- the values could have significant error due to the low expression of the gene (Ct>35), and thus the up or down-regulation could be merely a result of the high standard deviation of the high Ct values of the genes, or fluctuations in the housekeeping genes.
- the Relative Expression values that are between 0.5-fold and 2-fold it indicates no significant change in the expression of the gene for these samples.
- Table 8c Results for relative expression of Embryonic Stem Cell, Ectoderm, Endoderm/mesoderm, and neuronal markers in untransfected and transfected HFF with Msi1/Ngn2/MBD2 calibrated to untreated HFF (passage 8).
- Genes with asterisk ( * ) indicate that the Ct values of the test samples are within the quantifiable range (Ct ⁇ 35), suggesting the expression of the gene in the test sample is adequate for quantification.
- NS-A Differentiation Medium SteinCell Technologies
- NbActive4 BrainBitsTM
- BDNF 20 ng/ml
- NT-3 20 ng/ml
- NGF 20 ng/ml
- Retinoic acid 5 ⁇
- Noggin 20 ng/ml
- Forskolin 10 ⁇
- NS-A Differentiation Medium StemCell Technologies
- NbActive4 BrainBits
- CDM I I medium CDM I I medium
- these first two were supplemented with the same cytokines as previously described but with the addition of Fgf-2 (20 ng/ml).
- Fgf-2 was removed from the first two differentiation media while cells in the CDM II medium were switched to the NS-A Differentiation Medium (StemCell Technologies) supplemented with cytokines without Fgf-2.
- NS-A Differentiation Medium StemCell Technologies
- TUBB3 neuronal genes ⁇ -tubulin (TUBB3) and Map2b were slightly elevated already from Day 2 onwards, but significantly increased on Day 5 onwards.
- ACHE acetylcholine esterase
- Sox2 was highly and early expressed which could then be directly or indirectly interact with the exogenous Msi/Ngn2 and/or other genes in the activation of Nestin, GFAP, and endogenous Msi1 and Ngn2 and other genes that promote the reprogramming and cell fate change, as well as the activation of neuronal genes like ⁇ ⁇ -tubulin (TUBB3), Map2b, and ACHE.
- Table 9 RT-PCR analysis of exogenous and endogenous relative expression of Msi1 , Ngn2 and MBD2 from Day 2-10 after transfection of fibroblast cells with pCMV-Msi1-Ngn2(Msi1/Ngn2) and MBD2 and cultured for 10 days in proliferation medium. Cells were collected at different time point to analyse endogenous gene expression.
- Table 10 RT-PCR analysis of relative expression of Nestin, Map2b, TUBB3, ACHE, GFAP, and Sox2 from Day 2-10 after transfection of fibroblast cells with pCMV-Msi1-Ngn2 (Msi1/Ngn2) and MBD2 and cultured for 10 days in proliferation medium. Cells were collected at different time point to analyse endogenous gene expression.
- HFF cells were cultured as described in Example I, and transfected using the NucleofectorTM ® II Device (Lonza) as previously described in Example II or the NucleofectorTM ® 96-well Shuttle ® Device (Lonza).
- the HFFs were harvested with TrypLETM (Gibco), and 1 x10 6 cells / transfection with the NucleofectorTM ® II Device for 10 min at 90g and 6x10 5 cells / transfection with the NucleofectorTM ® 96-well Shuttle ® Device for 5 min at 80xg.
- each 100 ⁇ of cell suspension was combined with 2 different mixes of plasmid DNA (sample 1 was mixed with 2 pg of pCMV6-XL5-Msi1 and 2pg pCMV6-XL5-MBD2, and sample 2 with 2 pg of Msi1/Ngn2 and 2pg pCMV6-XL5-MBD2).
- Each cell suspension was transferred into an Amaxa certified cuvette and transfected with the appropriate program (U-023). Right after transfection, 900 ⁇ of warm CDM1 medium was added to each cuvette and the sample was transferred into a culture plate coated with Laminin (Stemgent, 10 pg/ml) at a cell density of 1 x10 5 to 1 .5x10 5 cells per cm 2 or into non-cell culture treated Petri dishes for neurosphere formation. The cells were incubated at 37°C, 5% C0 2 overnight.
- Laminin Stegent, 10 pg/ml
- the steps described before were similar with the following exceptions: the cell suspension was mixed with 0.6 ⁇ g of each DNA of the same 2 DNA mixes, the cell suspension was transferred to a well of a 96-well NucleoplateTM (Lonza) and transfected with the program FF-130TM. After transfection, 80 ⁇ of warm CDM1 medium was added to each well and the samples were left for 10 min in the incubator prior to being transferred into a laminin coated plate or non-cell culture treated Petri dishes at the same cell density as previously mentioned. For both devices, these steps were repeated for each sample that was transfected. Prior to transfection cells were cultured in CDM1 as described in Example I.
- the medium was switched to a mix of 75% CDM medium and 25% Proliferation Medium which was supplemented with EGF (20 ng/ml), FGF-2 (20 ng/ml), Noggin (20 ng/ml) and Cytochalasin B (10 pg/ml) and the cells were incubated at 37°C, 5% C0 2 and 5% 0 2 .
- the medium was changed daily with an increased proportion of Neural proliferation medium up to 100% by Day 4 and a decreased proportion of Cytochalasin B that was completely omitted by Day 5.
- Forskolin (10 ⁇ ) was added to the medium from Day 4 onwards.
- the cells in floating conditions were pelleted by centrifugation and their medium changed daily as described for the adherent condition. Cells were collected at Day 3, 7, and 12 for imunohistochemistry analysis.
- the highest percentage at Day 12 was obtained with Msi1/Ngn2 and pCMV6-XL5-MBD2 transfected with the NucleofectorTM ® 96-well Shuttle ® Device (-80%).
- the same combination transfected with the NucleofectorTM ® II yielded only -35% positive cells.
- the pCMV6-XL5-Musashi and pCMV6-XL5-MBD2 with the Shuttle ® produced -20% positive cells, while generally none were observed with the NucleofectorTM ® II.
- the percentage of positive cells varied strongly between wells. The staining indicated that the cell population was not homogenous, since fields of densely arranged Sox2 positive cells and complete fields with only negative cells could be found in all cases.
- the Shuttle ® was initially more toxic to cells than the NucleofectorTM ® II, however at least in the case of Msi1/Ngn2 and pC V6-XL5-MBD2 shuttle, the Sox2 positive population rapidly expanded from Day 7 to Day 12 to have twice as many Sox2 positive cells as compared to the NucleofectorTM ® II.
- the cells in floating conditions did not form spheres during the 12 day experiment in any of the conditions, suggesting that the formation of neurospheres requires either the generation of neural stem-like cells in adherent conditions first or more time.
- Table 11 shows the percentage of Sox2 positive cells with a typical neural stem cell morphology using both the NucleofectorTM ® II Device and the NucleofectorTM ® 96-well Shuttle ® Device. The latter had the advantages of requiring a smaller starting material (less cells and less DNA required) and in addition gave rise to a higher number of Sox2 positive cells. Moreover a very small population of Sox2 positive cells was observed with the Shuttle ® Device only upon transfection with only one neurogenic transcription factor (Msi) in the presence of the DNA demethylator MBD2. Table 11 : Percentage of positive cells for Sox2 after transfection of fibroblast cells with different expression vectors.
- HFFs were cultured and treated with cytochalasin B as described in Example III, and treated simultaneously with VPA (1 mM) and 5-Azacytidine (0.5 ⁇ ). After two days of treatment, cells were transfected by Nucleofection as described in Example II with the constructed vector Msi1/Ngn2. After preparing the cells, they were mixed with 2 ⁇ g of total DNA (Msi1/Ngn2) and cells that had not been treated with chemical inhibitors (VPA and 5-Aza) were co-transfect with MBD2 (2ng), using the appropriate program (U023). The samples were transferred into a coated culture plate with Laminin (10 ⁇ g/ml, Sigma) and incubated in a humidified 37°C/5%0 2 /5%C0 2 incubator.
- Laminin 10 ⁇ g/ml, Sigma
- the medium was changed to the proliferation basal media, Neural Proliferation Medium (NeuroCultTM proliferation Kit, StemCell Technologies), with the presence of Noggin (20 ng/ml, Peprotech), recombinant hFGF (20ng/ml, Peprotech), and recombinant hEGF (20ng/ml, Peprotech).
- Neural Proliferation Medium NeuroCultTM proliferation Kit, StemCell Technologies
- Noggin 20 ng/ml, Peprotech
- hFGF recombinant hFGF
- hEGF recombinant hEGF
- cells were sedimented by centrifugation at 150 x g for 3 min at room temperature (RT). The pellet was then resuspended in fresh medium and plated into new uncoated, low-bind cell culture dishes. Cultures were incubated at 37°C, 5%C0 2 , 5%0 2 and were fed daily for at least two months.
- neurospheres were dissociated into single cells and these single cells were isolated and cultured in proliferation medium in suspension, and neurosphere formation was monitored by taking bright field images using light microscope (Nikon, 10X) and by CellomicsTM. These cells started to proliferate and grew as spheres starting day 6 to day 10 ( Figure 4A). Immunohistochemistry analysis of these spheres (Table 12 and Figure 4) on Day 20, revealed immunopositive staining for the neural stem cells markers Sox2, Musashi, CD133, Nestin, and GFAP.
- Cells also stained positive for ⁇ -tubulin (a marker for neurons), 04 (a marker for oligodendrocytes), and GFAP (a marker for astrocytes), indicating the tri-potent differentiation potential of both sets of cells (NSLC and hNPC), and negative for NGFrec and NeuN (markers for differentiated neurons) indicating that the cells were not terminally differentiated.
- ⁇ -tubulin a marker for neurons
- 04 a marker for oligodendrocytes
- GFAP a marker for astrocytes
- Table 12 Percentage of positive cells for neural stem cells, and neuronal, astrocyte and oligodendrocyte lineage markers in neurospheres formed from single NSLCs and hNPCs cultured in proliferation medium (StemCell Technologies) supplemented by EGF (20ng/ml, Peprotech) and FGF (20ng/ml, Peprotech) for 20 days at 37°C / 5% C0 2 / 5% 0 2 .
- HFF cells were cultured as described in Example I, and transfected using the NucleofectorTM II device (Lonza) as described in Example II. Cells were co-transfected with pCMV6-XL5-Msi/pCMV6-XL4-Ngn2, pCMV-Msi1-Ngn2 with MBD2 or pre-treated with VPA/5aza. Cells were cultured in proliferation medium as suspension or adherent cultures.
- fibroblast-specific genes (Col3A1 , Lox, S100A4) were down- regulated in reprogrammed cells, indicating the loss of fibroblast-specific genes following transfection (note that not all cells got transfected and reprogrammed, so the presence of fibroblast-specific gene expression in the cultures is mostly from the un-programmed fibroblasts left in the culture).
- the expression of these genes is observed to increase when HFFs were transfected in the absence of DNA demethylator or the DNA methylation inhibitor, indicating that down-regulation of differentiated markers of fibroblast cells requires DNA demethylation.
- ectoderm genes such as Msi1 , Sox2, and Nestin was remarkably increased following transfection in conjunction with DNA demethylation.
- the expression of neuronal markers, such as synaptogaminl (a synaptic vesicle protein) and NeuroDI was up-regulated in transfected cells with Msi1/Ngn2/MBD2, and slightly increased in transfected cells with Msi1/Ngn2 VPA and 5-AZA.
- the selected three markers of oligodendrocytes were detected in the transfected cells with a strong increase of Olig2.
- Two markers for astrocytes, GFAP and ALDH1 L1 were enhanced following transfection. The results support the idea that neurospheres are composed of heterogeneous progenitor subtypes.
- GAP-43 neuropeptide Y
- NPY neuropeptide Y
- GAP-43 has long been acknowledged to play a pivotal role in axonal plasticity and is used as a marker of regenerating neurite outgrowth and synaptogenesis, both in embryonic development and in neuronal regeneration in injured brain and spinal cord.
- Expression of receptors for growth and neurotrophic factors was increased, such as neurotrophic receptor tyrosine kinase expression.
- Table 14 Gene array analysis was performed after one month of transfection of human fibroblast cells with Msi1/Ngn2, in the presence MBD2 or VPA and 5-Aza.
- Cells were cultured on coated culture plates as adherent cells or on untreated culture plates as neurospheres in proliferation medium (StemCell Technologies) supplemented with EGF (20ng/ml) and FGF (20ng/ml). Untransfected cells were considered as negative control and ReNcell (Millipore) as positive control.
- HFF markers such as fibronectin and fibroblast protein marker
- Figure 5 such as fibronectin and fibroblast protein marker
- Table 15 The percentage of cells stained positive for neural stem cell markers and fibroblast markers in untransfected cells and transfected cells with pMsi1/Ngn2/MBD2.
- Transfected cells possess a high percentage of neural stem markers but a very low percentage of fibroblast markers as compared to untransfected cells.
- NSLCs have the capacity to proliferate in culture and exhibit stable morphology, gene and protein expression that were maintained for the entire study period, which was for over five month in culture (Table 16).
- Table 16 Doubling time of NSLCs over serial passages.
- NSLCs were maintained in proliferation conditions for 35 passages in a 37°C, 5%C0 2 and 5%0 2 incubator.
- the average generation time was 25.4h over 35 passages.
- Microarray expression analysis was performed to get a global overview to compare the gene expression profile of passage 7 NSLC to both HFF (the cells that the NSLC were created from) and hNPCs.
- Genotypics extracted RNA from the samples and performed Quality Control using an Agilent BioanalyzerTM. Labelling was done using Agilent's Quick AmpTM kit (cDNA synthesis and in vitro transcription), followed by Labelling QC. Hybridization was then performed using the 8 x 60K array, and scanning was done using high throughput Agilent scanner with SureScanTM technology. The Agilent Feature Extraction software was used for automated feature extraction, followed by Raw Data QC and Image QC. Advanced Data Analysis was then performed, including Pathway and Gene Ontology analyisis using Agilent's GeneSpring GXTM v10.0 and Genotypic's Biointerpreter Software.
- the NSLC samples were compared to the HFF samples (Set 1 ) and hNPC samples (Set 2)
- the NSLC samples had a global gene expression pattern that was much closer to the hNPCs than the HFFs from which the NSLCs were created (Figure 23). Pearson correlation analysis revealed that NSLCs are closely related to hNPCs, including in terms of neuronal lineage markers, regenerative genes and migration genes. These data confirm that NSLCs are similar, but not identical, to hNPCs.
- Microarray analysis revealed an up-regulation of neural precursor genes in the NSLC samples as compared to the HFF samples.
- ACTL6A and PHF10 which both belong to the neural progenitors-specific chromatin remodelling complex (npbaf complex) and are required for the proliferation of neural progenitors, were up-regulated by 2.9-fold and 2.3 fold respectively.
- MSI2 which plays a role in the proliferation and maintenance of stem cells in the central nervous system, was up-regulated by 6-fold (Table X1 ).
- Glia genes were up-regulated in the NSLC samples as compared to the HFF samples.
- GFAP is a neural stem cell- and astrocyte-specific marker that, during the development of the central nervous system, distinguishes astrocytes from other glial cells, is highly up-regulated in the NSLC sample as compared to HFF (690-fold).
- OLIG1 which promotes formation and maturation of oligodendrocytes, especially within the brain, is also highly up-regulated in NSLC sample as compared to HFF (370-fold) (Table X2)
- Table X3 lists a subset of regenerative genes that are up-regulated in the NSLC samples as compared to the HFF samples.
- SOX2 a gene critical for early embryogenesis and for embryonic stem cell pluripotency as well as neural stem cells, is highly up-regulated in the NSLC samples as compared to the HFF samples (5000-fold).
- CCND2 which is essential for the control of the cell cycle at the G1/S (start) transition, is also up-regulated in NSLC samples (70- fold as compared to HFF samples).
- numerous fibroblast genes were down-regulated in the NSLC samples as compared to the HFF samples. This shows that the NSLC lose the expression of numerous fibroblast genes as it gets reprogrammed from HFF to NSLC.
- Table X5 show that neural precursor genes were also up-regulated in the NSLC samples as compared to the hNPC samples.
- BDNF which promotes the survival and differentiation of selected neuronal populations of the peripheral and central nervous systems during development, is even more highly expressed in NSLC samples than in hNPC samples (34-fold up-regulation).
- Table X6 shows that a subset of Glia genes are also up-regulated in the NSLC samples as compared to the hNPC samples.
- GFAP a neural stem cell- and astrocyte- specific marker that, during the development of the central nervous system, distinguishes astrocytes from other glial cells, is more highly expressed in NSLC samples than hNPC samples (13-fold).
- PLP1 the major myelin protein of the central nervous system which plays an important role in the formation or maintenance of the multilamellar structure of myelin, is also more highly expressed in NSLC samples than in hNPC samples (20-fold).
- Regenerative genes were also up-regulated in the NSLC samples as compared to the hNPC samples (Table X7).
- BMP2 a neural crest marker, but which induces growth especially of cartilage and bone formation
- BMP4 which in turn induces cartilage and bone formation and acts in mesoderm induction, tooth development, limb formation and fracture repair, but also in neural stem cells, were both more highly expressed in NSLC samples than in hNPC samples (18-fold and 20-fold respectively).
- GAP43 which is a major component of the motile growth cones that form the tips of elongating axons was more highly expressed in NSLC samples than hNPC samples (4-fold). This suggests the regenerative potential of NSLC.
- HOXB4 a transcription factor that is involved in development and also in the expansion of neural stem cells as well as hematopoietic stem and progenitor cells in vivo and in vitro making it a potential candidate for therapeutic stem cell expansion, was also more highly expressed in NSLCs than in hNPCs. This data indicates that NSLCs are more 'stem-like' or have more 'sternness' than hNPCs.
- ble X7 Up-regulated Regenerative genes (NSLC vs. hNPC)
- neurospheres were dissociated and plated in laminin/poly-D-Lysine (1 C ⁇ g/ml; Sigma) in differentiation medium for two weeks.
- NbActive medium Brain Derived Neurotrophin Factor (BDNF, 20ng/ml, Peprotech)
- BDNF Brain Derived Neurotrophin Factor
- ATRA a//-frans-retinoic acid
- bFGF 40ng/ml, Peprotech
- NeuroCultTM differentiation medium NeuroCultTM Differentiation kit, StemCell Technologies
- the cells were stained with the neuronal marker ⁇ -tubulin, astrocyte markers GFAP and S100p, andoligodendrocyte marker CNPase.
- the cells were fixed with 4% formaldehyde and the primary antibodies were added in 5% normal goat serum / PBS as follows: Mouse antibody ⁇ -tubulin (1 :200, Abeam), rabbit antibody ⁇ ⁇ (1 :100, Abeam), and Chicken antibody CNPase (1 :50, Abeam).
- NbActive medium promoted the differentiation equally to neuronal (48.66 ⁇ 14.07%, ⁇ -tubulin) and potential early oligodendrocyte lineages (50.01 +4.04%, CNPase) and to a lower percentage of astrocyte cells (2.68 ⁇ 1 .13%, 5100 ⁇ ), while NS-A differentiation medium induced the differentiation mainly to neurons (64.89 ⁇ 4.1 1 %, ⁇ -tubulin) and astrocytes (35.94 ⁇ 4.04%, S100beta), and a low percentage of potential early oligodendrocytes cells (8.68 ⁇ 2.71 %, CNPase) .
- the NSC-A medium was selected over NbActive for further differentiation studies. Differentiation of cells in NS-A differentiation medium promote the differentiation of hNPC and NSLC similarly as shown in Table 17 by the decrease of the percentage of sox2, musashi and nestin positive cells. NSLCs were differentiated to neuronal (74.3 ⁇ 0.1 , GABA), astrocyte lineage (65.6 ⁇ 0.0, S100beta) and to a lower percentage of oligodendrocyte cells (5.2 ⁇ 0.6, CNPase). The same pattern of tripotent lineage differentiation was observed with hNPCs (Table 17).
- Table 17 The percentage of cells stained positive for neural stem cell and neuronal lineage markers in transfected and untransfected cells.
- NSLCs and hNPCs were cultured in NS-A- differentiation medium supplemented with BDNF (20ng/ml) and FGF (40 ng/ml), cultures were incubated at 37°C, 5% C0 2 , 5%0 2 for three weeks. The percentage of immunopositive cells was
- MAP2b microtubule-associated protein
- NCAM neuropeptide-binding protein
- synaptophysin an antibody used to identify functional neuronal cells, was increased following 2 and 3 weeks of differentiation, indicating maturity of the neuronal cells.
- GABA and acetycholine markers were increased following 2 weeks of differentiation and decreased at week 3.
- the morphological changes and expression of a number of neuronal antigens and genes show that the above method results in normal and viable neuronal cells. Additionally, the newly formed neuronal cells have the morphological criteria of neurons. In addition to the above markers, the differentiated cells were evaluated by characterizing morphological markers of neurite differentiation. Neuron type cells (cells strongly expressing ⁇ -tubulin) showed neurite formation after differentiation, , including an increase in the average number of neurites per neuron (from e.g. 1.38 +0.1) The same pattern was observed in ⁇ -tubulin positive cells. Accordingly, the average neurite length (118.3+3.5 ⁇ ) and the number of branch points (3.28 ⁇ 0.3) per neuron also increased. The differentiated neuron-like cells developed long neurites that were greater than three cell diameters in length with a growth cone at the end, expressed neuron-specific genes, and stopped proliferating after the induction of differentiation.
- NSLCs and hNPCs were cultured in NS-A differentiation medium as described previously supplemented with FGF-2 (10ng/ml, Peprotech) and sonic hedgehog (SHH, 100ng/ml, Peprotech) for 4 days. After 4 days medium was changed to NS-A differentiation medium supplemented by T3 (60 ng/ml, Peprotech), IGF1 (10ng/ml, Peprotech), NT-3 (10ng/ml, Peprotech), and PDGF (10ng/ml, Peprotech). Cells were cultured for 20 days at 37°C, 5% C0 2 .
- Table 18 The percentage of cells stained positive for neural stem cell and neuronal lineage markers in transfected and untransfected cells.
- NSLCs and hNPCs were cultured in differentiation medium supplemented with SHH (100ng/ml, Peprotech ), T3 (60 ng/ml, Peprotech), IGF1 (10ng/ml, Peprotech), NT-3 (10ng/ml, Peprotech), and PDGF (10ng/ml, Peprotech) to induce differentiation towards oligodendrocytes.
- SHH 100ng/ml, Peprotech
- T3 60 ng/ml, Peprotech
- IGF1 10ng/ml, Peprotech
- NT-3 10ng/ml, Peprotech
- PDGF 10ng/ml, Peprotech
- NSLCs stable reprogrammed cells
- epigenetic modification inhibition of acetylation and methylation
- These cells possess and retain neural stem cell properties as determined by: (1) the expression of neural stem cell genes and proteins, (2) the capacity to generate and grow as neurospheres starting from a single cell, and (3) to differentiate to neuronal lineages in differentiation conditions.
- neural stem cell properties as determined by: (1) the expression of neural stem cell genes and proteins, (2) the capacity to generate and grow as neurospheres starting from a single cell, and (3) to differentiate to neuronal lineages in differentiation conditions.
- neurons display one or more neural- specific morphological, physiological and/or immunological features associated with a neuronal cell type.
- Useful criteria include morphological features (long processes or neurites), physiological, and/or immunological features such as expression of a set of neuronal-specific markers or antigens.
- NSLCs readily turn into a tripotent-like precursor cell with differentiation potential to a high percentage of neuronal, astrocytes and lower percentage of oligodendrocyte populations.
- HFFs were cultured and treated with cytochalasin B as described in Example III. After two days of treatment, cells were transfected by Nucleofection as described in Example II with the constructed vector Msi1/Ngn2. Briefly, after preparing the cells, they were mixed with 2 pg of total DNA (Msi1/Ngn2) and were co-transfect with MBD2 (2 ⁇ 9), by the Amaxa's NucleofectorTM according to the manufacturer's protocol.
- the samples were then transferred into a Laminin (10 pg/ml, Sigma) coated culture plate and cultured in the presence of Neural Proliferation Medium (NeuroCultTM proliferation Kit, StemCell Technologies)with recombinant hFGF (20ng/ml, Peprotech), recombinant hEGF (20ng/ml, Peprotech), and with or without the presence of Noggin (20 ng/ml, Peprotech).
- Neural Proliferation Medium NeuroCultTM proliferation Kit, StemCell Technologies
- Samples were collected at different time points (1 , 3, 4, 6, and 8 days) to analyze neuronal gene expression by RT-PCR and protein expression levels by immunohistochemistry.
- Table 19 RT-PCR analysis of relative expression of neuronal precursor cell markers such as nestin and Sox2 after transfection of HFF with pCMV-Msi1-2A-Ngn2 and pCMV6-XL5-MBD2 with or without Noggin (20ng/ml). Relative expression of Sox2, and nestin was increased after transfection with and without No in.
- NSLCs created from HFF cells are not Skin-Derived Precursors (SKPs)
- SSPs skin-derived precursors
- ADSC Adipose derived stem cells
- CellStartTM was diluted 1 : 100 in dPBS/Ca 2+ /Mg 2+ and the flask incubated for 2 hours at 37°C. Cells are passaged every 3 to 4 days using AccutaseTM and medium was changed every 2 days. Three to four days before initiating differentiation, ADSCs and NSLCs were seeded in 6- wellplates in CellStartTM (1 : 100 in dPBS/Ca 2+ /Mg 2+ / 2 hours at 37°C) coated tissue culture plates.
- proliferation media were replaced by differentiation medium consisting in DMEM/F12 (50:50), ITS (1 : 100), HEPES (1 :100), GlutaMAXTM (1 :100), T3 (0.2 nM), Rosiglitasone (0.5 ⁇ / ⁇ ), IBMX (100 ⁇ ) and Dexamethasone (1 ⁇ ).
- cells were fixed with a 4% formaldehyde solution for 10 min and stained with Oil Red O (Invitrogen) staining solution for 5 min. Staining was removed and cells washed twice with PBS.
- Adipose cells appeared red with lipid droplets specifically stained with Oil Red O, however NSLCs were stained negative, with no presence of lipid droplets in the cells, and the cells adopted neuronal cell morphology.
- Neural Stem-Like Cells differentiated into neuronal and glial cells were kept in culture for 55 days, and BDNF released in the conditioned medium was measured by antigen- capture ELISA at different time points and compared to the release in mature neurons (ScienCell), undifferentiated Neural Human Normal Precursor cells (NHNP, Lonza) as well as to undifferentiated NSLCs and untransfected cells (HFF).
- ScienCell mature neurons
- NHNP undifferentiated Neural Human Normal Precursor cells
- HFF untransfected cells
- Conditioned medium from each group was collected, centrifuged, and then stored at -80°C until assaying.
- BDNF concentrations were measured by ELISA kits (BDNF E max Immunoassay System, Promega Corporation, USA), according to the manufacturer's instructions.
- 96-well ELISA immunoplates were coated with Anti-BDNF (CatNb#G700B) diluted 1/1000 in carbonate buffer (pH 9.7) and incubated at 4°C overnight. The following day, all wells were washed with TBS-TweenTM 0.5% before incubation with Block/Sample buffer 1X at room temperature for one hour without shaking. After blocking, standards and samples were added to the plates and incubated and shaken (450 ⁇ 100rpm) for 2 h at room temperature. Subsequently, after washing with TBS-TweenTM wash buffer, plates were incubated for 2h with Anti-Human BDNF pAb (1 :500 dilution in Block & Sample 1X Buffer) at 4°C.
- BDNF was released at the same concentration from differentiated Neuron-Like Cells (NLCs differentiated from NSLCs) and normal Human neuron cells starting at day 11 and remained until day 55 (Table 20), while no BDNF (except for tiny amounts in the untransfected HFF group) was released in the other groups.
- NLCs Neural-Like Cells
- NSLCs Neural Stem-Like Cells
- the NLCs were functional and possessed the capacity to release neurotrophic factor (BDNF).
- BDNF neurotrophic factor
- Generating reprogrammed neuronal-like cell lines that can locally deliver these neurotrophic factors could be used as a method to treat several neurological conditions and may offer crucial benefits in regeneration and functional recovery from brain and other injuries.
- Human mobilized peripheral blood CD34 + cells were purchased from StemCell Technologies and expanded as a floating culture in Petri Dishes in complete StemProTM®-34 Serum-free Medium (Invitrogen) supplemented with Stem Cell Factor (SCF, 150 g/ml, Peprotech), Granulocyte Colony-Stimulating Factor (GM-CSF, 37.5 ng/ml, Peprotech) and IL-3 (75 ng/ml, Peprotech).
- SCF Stem Cell Factor
- GM-CSF Granulocyte Colony-Stimulating Factor
- IL-3 75 ng/ml, Peprotech
- ADSC Human Adipose-Derived Stem Cells
- the cells were incubated at 37°C, 5% C0 2 .
- Media was replaced with fresh, complete growth media every two to three days until subculture.
- RT-PCR Analysis revealed an increase of relative expression of neural stem cell markers such as Sox2, nestin, GFAP, and ⁇ -tubulin after transfecting Keratinocyte and CD34 + by Msi1 and Ngn2. Relative expression of nestin and GFAP was enhanced in NSLCs created from keratinocytes and CD34 + cells as compared to NSLCs from HFFs; however, the reverse was true for Sox2 and ACHE expression.
- ⁇ -tubulin (TUBB3) and Map2b expression was highest in NSLCs created from CD34 + cells, followed by NSLCs created from HFF (Table 21).
- This data shows that different types of NSLCs with different gene expression profiles (and characteristics) can be created from different types of starting / source cells (and the same has been observed for creating some other types of stemlike cells discussed in this application).
- the data is also interesting since it was not expected that keratinocytes (which are derived from the ectoderm just as endogenous neural stem cells) would have a lower expression than HFFs for all the genes analyzed except for Nestin (it was expected that keratinocytes would be the easiest to reprogram into NSLCs since they are derived from the ectoderm).
- Ill Table 21 RT-PCR analysis was performed after one month of transfection of human fibroblasts (HFF), Keratinocytes, and CD34 + cells with Msi1/Ngn2 (MSI 1 /NGN2), in the presence MBD2 with VPA treatment.
- HFF human fibroblasts
- MSI 1 /NGN2 Msi1/Ngn2
- VPA VPA treatment
- NSLCs developed from HFF yield a higher percentage of positive staining for Sox2 and GFAP (55.8 ⁇ 3.8 and 78.1 +2.4) as compared to CD34 + cells (42.8+2.7 and 24.2 ⁇ 4.4), and keratinocytes (47.1 ⁇ 2.1 and 43.4 ⁇ 8.9).
- the percentage of nestin positive cells was high in Keratinocytes (77.6 ⁇ 10.7) and HFF (68.45 ⁇ 12.9) and lower in CD34 + cells (15.5 ⁇ 2.7) (Table 22). Sox2 and Nestin positive staining was undetectable in ADSCs.
- Table 22 The percentage of Sox2 and nestin positive cells for neural stem cell markers after transfecting fibroblast, keratinocyte, and CD34 + cells with pCMV-Msi1 -Ngn2 in the presence of MBD2 and VPA.
- Cells were cultured on coated culture plates in proliferation medium (StemCell Technologies) supplemented with EGF (20ng/ml) and FGF (20ng/ml) for 18 days. Untransfected cells were considered as negative control. The percentage of immunopositive cells was
- NSLCs generated from keratinocytes and CD34 + cells were tested for tripotent capacity. Further differentiation studies were performed to induce differentiation of these NSLCs towards neuronal lineage, using NeuroCultTM differentiation medium (NeuroCultTM differentiation Kit, StemCell Technologies) supplemented with BDNF (20ng/ml, Peprotech) and bFGF (40ng/ml, Peprotech) as described in Example V.
- NeuroCultTM differentiation medium NeuroCultTM differentiation Kit, StemCell Technologies
- BDNF 20ng/ml, Peprotech
- bFGF 40ng/ml, Peprotech
- RT-PCR analysis revealed decrease of undifferentiated genes (Nestin and Sox2) and increased of differentiated genes (Map2, ⁇ -tubulin, CNPase, and GFAP) as shown in Tables 23A, 23B, 23C and 23D.
- Table: 23A RT-PCR analysis was performed on NSLCs generated from human fibroblasts (HFF), keratinocytes, and CD34 + cells that were cultured on Poly-D-Lysin/Laminin coated culture plates in differentiation medium for 28 days (StemCell Technologies) supplemented with BDNF (20ng/ml) and FGF (40ng/ml).
- hNPCs (Lonza) were considered as a positive control.
- hNPCs had a much lower increase in ACHE, GFAP, and MAP2b (which actually decreased in hNPCs), but an increase in Nestin, compared to NSLCs under differentiation conditions.
- Table 23B RT-PCR analysis was performed on undifferentiated NSLCs generated from human fibroblasts (HFF), keratinocytes, and CD34 + cells that were cultured on Laminin coated culture plates in Proliferation medium for 4 days (StemCell Technologies) supplemented with EGF (20ng/ml) and FGF (20ng/ml). Relative expression calibrated to undifferentiated hNPCs.
- Table 23C RT-PCR analysis was performed on differentiated NSLCs generated from human fibroblasts (HFF), keratinocytes, and CD34 + cells that were cultured on Poly-D-Lysin/Laminin coated culture plates in differentiation medium for 14 days (StemCell Technologies) supplemented BDNF (20ng/ml) and FGF (40ng/ml). Relative expression calibrated to Day 14 differentiated hNPCs.
- HFF human fibroblasts
- keratinocytes keratinocytes
- CD34 + cells were cultured on Poly-D-Lysin/Laminin coated culture plates in differentiation medium for 14 days (StemCell Technologies) supplemented BDNF (20ng/ml) and FGF (40ng/ml). Relative expression calibrated to Day 14 differentiated hNPCs.
- Table 23D RT-PCR analysis was performed on differentiated NSLCs generated from human fibroblasts (HFF), keratinocytes, and CD34 + cells that were cultured on Poly D-Lysin/Laminin coated culture plates in differentiation medium for 28 days (StemCell Technologies) supplemented with BDNF (20ng/ml) and FGF (40ng/ml). Relative expression calibrated to Day 28 differentiated hNPCs.
- HFF human fibroblasts
- keratinocytes keratinocytes
- CD34 + cells were cultured on Poly D-Lysin/Laminin coated culture plates in differentiation medium for 28 days (StemCell Technologies) supplemented with BDNF (20ng/ml) and FGF (40ng/ml). Relative expression calibrated to Day 28 differentiated hNPCs.
- ⁇ -tubulin positive cells were generated from differentiated NSLCs generated from HFF (57.83 ⁇ 4.49) as compared to ⁇ -tubulin positive cells generated from Keratinocytes (23.27+2.91 ) and CD34 + cells (39.15+7.99) (Table 24)
- Table 24 The percentage of cells stained positive for neural stem cell markers and neuronal lineage markers in hNPCs (Lonza) and transfected keratinocytes, HFF, and CD34 + cells with pMsi1/Ngn2/MBD2.
- Transfected cells were cultured in Proliferation medium or differentiation medium for 28 days at 37°C, 5%C0 2 , 5%0 2 .
- CD34 + -NC neuronal cells generated after differentiation of NSLCs generated from CD34 + cells. Each data point represents the analysis of at least 1000 cells from at least 8 images.
- the % of Sox2 positive cells decreased faster, the % of Nestin positive cells generally decreased slower, and the % of cells expressing one of the differentiation markers (5100 ⁇ , ⁇ - tubulin, GFAP) generally increased slower in hNPCs than in the NSLCs during differentiation.
- the % of cells expressing one of the differentiation markers (5100 ⁇ , ⁇ -tubulin, GFAP) generally increased slowest in NSLCs created from keratinocytes and fastest in NSLCs created from HFFs. This study indicates that NSLCs can be created from keratinocytes and CD34 + blood cells, and these cells share morphology and markers similarly to NSLCs generated from HFF.
- NSLCs created from from keratinocytes, CD34 + cells, and HFFs had a tendency to differentiate more towards an astrocyte lineage than a neuronal lineage (except NSLCs created from HFFs had an almost similar number of ⁇ -tubulin positive and GFAP positive cells) as shown by the high percentage of GFAP positive cells during differentiation, which was confirmed by Sl OObeta staining.
- the proportion of astrocyte and neuronal cells generated from hNPCs was lower in same culture conditions, indicating that NSLCs generated from HFF, Keratinocytes, and CD34 + cells can give rise to a higher number of neuronal and astrocyte cells as compared to hNPCs.
- NSLCs are tripotent cells and possess the capacity to differentiate to neurons, astrocytes, and oligodendrocytes similarly to hNPCs.
- RT-PCR and immunohistochemistry analysis of transfected ADSCs did not reveal any significant expression of neural stem cell genes, indicating a need to optimize conditions for turning ADSCs to NSLCs or to investigate the effect of others neurogenic factors that could turn these into NSLCs.
- Fibroblast cells were cultured in DMEM medium in the presence of 10% FCS as described in Example I, followed by seeding onto 12-well plates pre-coated with laminin ( ⁇ 0 ⁇ g/m ⁇ ) at a concentration of 2x10 6 cells/ml in defined CDM Medium consisting of a 3:1 ratio of Dulbecco's modified Eagle medium (DMEM, high glucose (4.5g/L) with L-glutamine and sodium pyruvate) and Ham's F-12 medium supplemented with the following components: EGF (4.2x10 10 M), bFGF (2.8x10 "10 M), ITS (8.6x10 "5 M), dexamethasone (1 .0x10 "7 M), L-ascorbic acid phosphate magnesium salt n-hydrate (3.2x10 ⁇ 4 M), L-3,3',5-triiodothyronine (2.0x10 ⁇ 10 M), ethanolamine (10 "4 M),GlutaMAXTM (4x 0 "3 M), glutathione
- fibroblast cells By culturing the fibroblast cells at hyperconfluent density in this completely chemically defined medium causes them to enter a high synthetic phase with a slow-down in proliferation, leading to the production of a living tissue equivalent (LTE) consisting of multiple layers of fibroblasts within de novo 3D extracellular matrix (CDM) that is completely synthesized by the fibroblasts themselves.
- LTE living tissue equivalent
- CDM de novo 3D extracellular matrix
- CDM samples were treated with cytochalsin B (10 ⁇ g/ml, Calbiochem), with the concentration of cytochalsin B reduced from ⁇ 0 ⁇ g/m ⁇ to 0 ⁇ g/ml (none) over 5 days while at the same time switching the medium from CDM Medium to NbActive medium. Samples were cultured for another 12 days at 37°C, 5% C0 2 , and the medium was changed every day. Samples were fixed to perform immunohistochemistry as described previously to detect Neuronal markers. The following antibodies were used: mouse anti-nestin 647 (1 :100, BD) and anti-plll-tubulin (1 :200, Neuromics).
- Figure 18 shows immunostained cells with ⁇ -tubulin at day 7, indicating the de-differentiation of fibroblast cells to neurons. However, one week later, these trans-differentiated cells reverted back to fibroblast cells and ⁇ -tubulin expression was lost ( Figure 8). The loss of morphology and ⁇ I [-tubulin expression after withdrawal of the priming agents indicate that complete conversion to functional and stable reprogrammed cells did not occur.
- CDM was treated with VPA (4mM), 5-Aza (5 ⁇ ) and cytochalasin B ( ⁇ g/ml) as above.
- fibroblast cells within the CDM were transfected with DNA using Lipofectamine reagent (Invitrogen) as per the manufacturer's protocol.
- 15 ⁇ g of the eukaryotic DNA expression vectors pCMV6-XL5-Pax6, pCMV6-XL5-Msi1 and pCMV6-XL4- Ngn2 (Origene) were used to transfect the cells.
- Neural Progenitor Basal Medium (Lonza) supplemented with Noggin (50 ng/ml), EGF (20ng/ml), and bFGF (20ng/ml), and the cells were cultured at 37°C, 5% C0 2 and 5% 0 2 , and the medium was changed every day.
- differentiation was initiated by adding gradually NBActive medium (BrainBitsTM) supplemented with NT-3 (20ng/ml, Peprotech), a//-frans-retinoic acid (ATRA, 5 ⁇ , Spectrum), BDNF (20ng/ml, Peprotech), and bFGF (40ng/ml, Peprotech).
- Table 25 RT-PCR analysis of relative expression of neuronal precursor cell markers such as nestin, ⁇ -tubulin, MAP2b, ACHE, and GFAP after transfection of fibroblast cells with pCMV6-XL5-Msi1 , pCMV6-XL4-Ngn2, pCMV6-XL5-Pax6, and pCMV6-XL5-MBD2.
- CDM I Medium was changed and cells were cultured in proliferation medium (NPBM, Lonza) supplemented withEGF (20ng/ml. Peprotech) and bFGF (20ng/ml, Peprotech) for one week.
- NbActive Basal growth factor
- NT-3 (20ng/ml)
- bFGF 20 ng/ml
- ATRA 5 ⁇
- Forskolin 10 ⁇
- Cells were incubated at 37°C, 5%C0 2 , 5%0 2 for 12 days.
- Relative expression of Msi1 , Ngn2, Pax6, nestin, ⁇ -tubulin, ACHE, MAP2b and GFAP in NSLCs and NLCs was increased after transfection with both transcription factors Ngn2 and Msi1 with MBD2 as the DNA demethylator.
- CDM:Neural Progenitor Maintenance Medium (1 :1 Supplemented with Noggin (50 ng/ml), EGF (20ng/ml), and bFGF (20ng/ml).
- Medium was changed every day by increasing the percentage of NPBM and decreasing CDM medium.
- Cells were cultured for 6 days at 37°C, 5% C0 2 and 5% 0 2 .
- differentiation was initiated by gradually supplementing the NPBM Medium with NT-3 (20ng/ml, Peprotech), all- frans-retinoic acid (ATRA, 5 ⁇ , Spectrum), BDNF (20ng/ml, Peprotech), and bFGF (40ng/ml, Peprotech). Samples were collected at the end of the study (day 14) and data were analyzed by gene array to identify genes that were reproducibly found to be specific for neuronal lineages.
- oligodendrocyte genes such as NKx2.2, olig2, and MAG was increased by Msi1 and Ngn2; however, the increased was more pronounced by Msi1 as compared to Ngn2 (Table 26).
- Two markers for astrocytes (GFAP and AQP4) were highly expressed after transfection with Msi1 and Ngn2 in the presence of the DNA demethylator BD2.
- TDLA data revealed increases in specific markers for interneurons, such as somatostatin and calbindinl Doublecortin (DCX), which is expressed by migrating immature cells during development, and acetylcholine (ACHE), an early marker of neuronal cells, were highly expressed in reprogrammed cells (Table 26).
- Transfection with Msi1 or Ngn2 increased the expression of dihydropyrimidinase-like 3 (DPYSL3), an early marker of newborn neurons to five-fold with Msi1 and seven-fold with Ngn2.
- DPYSL3 dihydropyrimidinase-like 3
- MAP2 microtubule-associated protein 2
- NCAM neuronal cell adhesion molecule
- VEGF and GDNF genes were increased to almost five-fold and seven-fold by Msi1 and Ngn2, respectively.
- BDNF, EGF, and bFGF were not activated and even down-regulated as compared to untransfected cells.
- GAP-43 growth associated protein
- receptors for growth and neurotrophic factors was increased, such as type III receptor tyrosine kinase, Neurotrophic tyrosine kinase receptor, and neurotrophic tyrosine kinase.
- the fibrobiast-specific markers vimentin and fibronectin were down-regulated in the reprogrmmed cells.
- Table 26 Gene array of CDM transfected with pMsil and pNgn2 following the pre-treatment with cytochalasin B (10 g/ml), VPA (4mM) and 5-Azacytidine (5 ⁇ ).
- Transfected cells were cultured in differentiation medium (NbActive, BrainBitsTM) supplemented by ATRA (5 ⁇ ), bFGF (40ng/ml) and BDNF (20ng/ml).
- This study was designed to improve transfection of CDM by combining lipofectamine and nucleofection and using two vectors pCMV6-XL5-Msi1 and pCMV6-XL4-Ngn2 individually or in combination together with pCMV-XL5-MBD2.
- Cells within Day 4 CDM were lipotransfected for 6 hours with Msi1 /MBD2, Ngn2/MBD2 or Msi/Ngn2/MBD2 after 2 days of pre-treatment with or without cytochalasin B.
- transfection was performed on fresh HFFs after the 6 hours using Nucleofection as described in Example II , and transferred on top of the CDM when the lipofectamine media was changed to fresh CDM medium.
- NPBM Neural Progenitor Basal Medium
- Noggin 50 ng/ml, Peprotech
- recombinant hFGF 20ng/ml, Peprotech
- recombinant hEGF 20ng/ml, Peprotech
- Table 27 RT-PCR analysis of relative expression of neuronal stem cell markers such as nestin, Sox2, and GFAP after transfection of fibroblast cells within the CDM with different combinations with or without the co-treatment with cytochalasin B. Relative expression of Sox2, nestin, and GFAP in NSLCs was increased after transfection with both transcription factors Ngn2 and Msi1 with MBD2 as the DNA demethylator.
- Telomerase is active in neural precursor cells and suggest that its regulation is an important parameter for cellular proliferation to occur in the mammalian brain (Caporaso GL et, 2003). This study was performed to evaluate telomerase activity in cell extracts of adherent NSLCs (NSLCs cultured on laminin-coated plates) as well as NSLCs in floating neurospheres (NSLCs cultured in plates with a low-bind surface) at early (P7) and late passage (P27). The telomerase activity of the 4 samples was measured by the PCR-based telomere repeat amplification protocol (TRAP) using the TRAPeze ® Telomerase Detection Kit (Chemicon).
- TRAP PCR-based telomere repeat amplification protocol
- the cells were grown in 24-well plates, washed in PBS, and homogenized for 30 min on ice in buffer containing 10mM Tris-HCI, pH 7.5, 1 mM MgCI 2 , 1 mM EGTA, 0.1 mM Benzamidine, 5mM ⁇ -mercaptoethanol, 0.5% CHAPS and 10% Glycerol (1X CHAPS Lysis Buffer, provided in kit) and RNase Inhibitor.
- the samples were spun down and the protein concentration of the supernatant was determined using the BCA Assay.
- Malignantly transformed cells show reduced requirements for extracellular growth promoting factors, are not restricted by cell-cell contact, and are often immortal.
- Anchorage- independent growth and proliferation is one of the hallmarks of malignant transformation, which is considered the most accurate and stringent in vitro assay for detecting malignant transformation of cells.
- HFFs were used as a negative control and cervical carcinoma HeLa cells were used as a positive control.
- Cells were sedimented by centrifugation at 150 x g for 3 min at room temperature (RT). The assay was performed using the CytoSelectTM 96-well cell transformation assay (CellBiolabs). The base agar layer (1 .2%) was dissolved in 2X DMEM / 20% PBS solution and 50 ⁇ of the agar solution was added to the plate and incubated for 30 min at 4°C to solidify.
- the plate Prior to adding the cell agar layer, the plate was allowed to warm up for 15 minutes at 37°C.
- the cells were resuspended at different density (20.000 and 5000 cells/well), except the hNPCs were resuspended only at 5000 cells/well due to a lack of enough cells.
- the cells were mixed with the 1.2% agar solution, 2X DMEM/ 20%PBS, and cell suspension (1 :1 : 1 ), and 75 ⁇ of the mixture was transferred to wells already containing the solidified base agar layer, and was then placed in 4°C for 15 minutes to allow the cell agar layer to solidify.
- Table 28 Fluorescence measurement (Relative Fluorescence Unit, RFU) indicate that under the same conditions only carcinoma HeLa cells grow as an anchorage-independent colony, while both hNPCs and NSLCs (adherent and floating neurospheres) were negative for tumor growth in the standard agar plate tumor formation assay (CytoSelectTM cell transformation kit, Cell Biolabs Inc.).
- the DNA plasmid Msi1/Ngn2 (designed and constructed in house) was used in transient transfection for generation of NSLCs along with MBD2 (for sample 1 ), or 5-Aza and VPA (for sample 2). Two weeks after transfection, Southern blot was performed to test for possible genomic integration of the plasmid DNA. 3 pg of genomic DNA extracted from the NSLC samples, as well as from HFF (a human fibroblast cell line) used as a negative control, was digested with several restriction enzymes including Bg II I , Pstl and Stul , subjected to electrophoresis on a 1 % agarose gel and transferred to a positively charged nylon membrane (Roche).
- the membrane was hybridized in the DIG Easy HybTM buffer (Roche) at 42°C overnight with a 1.2 kb Dig-labeled PCR probe amplified from the plasmid DNA using a set of primers.
- the membrane was washed twice at room temperature with 2 ⁇ SSC, 0.1 % SDS for 5 min per wash, twice with 0.5 ⁇ SSC, 0.1 % SDS at 65°C for 15 min per wash.
- Hybridization signals of the membrane were detected using the CDP-StarTM substrate (Roche).
- the membrane was exposed to an X-ray film for analysis. The signals were stripped from the membrane using stripping buffer (0.2 M NaOH, 0.1 % SDS).
- the membrane was re-hybridized with a 0.9 kb Dig-labeled PCR probe amplified from the plasmid DNA using a set of primers.
- MS Multiple Sclerosis
- CNS central nervous system
- EAE Experimental Allergic Encephalomyelitis
- EAE can be induced in a variety of species and strains of animals [mice, Rat, marmoset monkey, rhesus macaques] using various CNS antigens [Myelin Oligodendrocyte Glycoprotein (MOG), proteolipid protein (PLP) and myelin basic protein (MBP)].
- MOG Myelin Oligodendrocyte Glycoprotein
- PGP proteolipid protein
- MBP myelin basic protein
- mice Female 7 to 8 weeks old C57BL/6 mice were purchased from Charles Rivers, and housed at MISPRO animal facility for one week before experimentation for adaption to the new environment. C57BL/6 mice were injected s.c. with 100 MOG 35-55 in CFA (Sheldon Biotechnology, McGill University) containing 5 mg/ml Mycobacterium tuberculosis H37Ra (Difco, inc), at 2 sites on the back. All mice received 200 ng pertussis toxin (List Biological Laboratories, Inc) i.p.
- hNSLC and hNPCs were given by single injection i.v. via the tail vein when the animals started to show symptoms of EAE (day 13 i.v). Both animals groups received cyclosporine (10mg/kg/day) one day before the injection of cells and daily from the day of transplantation to avoid any rejection of the human cells. Sham- treated age-, sex-, and strain-matched mice, injected i.p. with PBS alone, were used as controls. All groups of animals were observed for 43 days. Animals were sacrificed at 43 days p.t., brains and spinal cord were harvested in 30% sucrose in PBS.
- the treatments (Alginate , Alginate + hNPC, Alginate + NSLCs, RM X + NSLCs, RM X Only, Fibrin Gel, or Saline) were applied directly on the brain after ablation.
- the opening in the skull was then filled with Bone Wax.
- small pieces of sterile homeostatic tissue were inserted into the lesion in order to stop the bleeding.
- the sutures were performed using EthiconTM monofilament suture 1/2 circle needle shape.
- Surgeries were performed in sterile clean rooms, and topical antibiotics (Cicatrin®, GlaxoSmithKline) were applied to the exposed skull and scalp to limit local infection. Rats were immunosuppressed by daily injection i.p.
- cyclosporine A (10 mg/kg/day) starting the day before the surgery until the end of the study period.
- the purpose of the cyclosporine A injection was to reduce the rat's immune reaction to the treatment.
- the immune-suppression was sustained until the end of the study to ensure that any potential failure of regeneration (if taken place) was not due to the immune reaction against the treatment.
- Functional scores were performed weekly, in all groups, sensorimotor impairment was evaluated based on the behavioural tests as described below.
- Rotarod Test The rotarod speed was manually calibrated for the 10 and 20 RPM speed before all procedures. Animals were required to perch on the stationary rod for 30 sec to acclimate themselves to the environment. During this time, if any animal fell, it was placed back on the rod until it had achieved stationary capabilities for a period of 30 seconds. The animals were allowed 3 trials. The animals that were comfortable staying on the stationary rod for 30 sec were allowed to run with a constant speed of 10 and 20 RPM for 60 sec, and the number of falls were electronically recorded.
- Beam walking measures hindlimb coordination by means of distance travelled across 100 cm beam (2.3 cm in diameter, 48 cm off the floor). Rats were systematically trained to walk along the elevated beam from start to finish with the aim of completing the task. A safe location, i.e, a flat box, is placed at the end of the beam so that the rat is motivated to complete the task. Scale used for evaluation of beam-walking performance
- HFF cells were cultured as described in CDM II medium as described in Example I with only modifying EGF (5ng/ml) and FGF (10ng/ml), and transfecting using the NucleofectorTM ® 96- well Shuttle ® Device (Lonza) following the procedure described in Example IV.
- the cells were transfected with various combinations of cDNA clones as described in Table 29. After transfection, the cells were plated on 0.1 % Gelatin-coated plates and incubated at 37°C, 5% C0 2 , 5% 0 2 . Medium was changed every other day according to Table 30. Cells were analyzed at Day 4 by Quantitative Real-time PCR. Table 29: Various combinations of plasmids with potential to transfect the cells towards mesendoderm lineage.
- Table 30 Medium composition from Day -2 to Day 10
- Activin A Proliferative Assay for media at the following concentrations: Activin A (Peprotech, 30ng/ml), HSA (Baxter, 0.5%), NEAA (Gibco, 1X), ITS (Gibco, 1X), EGF (Peprotech, 5ng/ml), bFGF (Peprotech, 10ng/ml), CHIR99021 (Stemgent, 2uM), VPA (Stemgent, 1 mM), 5-Aza (Sigma, 0.5uM), BMP4
- RNA isolation and quantification was performed as previously described in Example I.
- cDNA was prepared and quantitative real-time PCR was performed as previously described in Example II, except the following TaqmanTM® Gene Expression Assays (Applied Biosystems) were used:
- Table 31 Relative Expression FoxA2, Sox17, and Cxcr4 after transfecting HFFs once with various gene combinations with potential to reprogram cells into mesoendoderm-like cells .
- the exact values are not significantly accurate due to low RNA yield, however a trend of increasing
- Table 32 Expression of GATA4, CDH1 (E-cadherin), p63, and SOX2 relative to untreated HFF control 4 days after transfecting HFF cells with various gene combinations with potential to re ro ram cells into mesoendoderm-like cells.
- SOX17 is still highly expressed in the SOX17-transfected samples (50,000 to 400,000-fold as compared to the untreated HFF sample).
- the SOX17 gene expression represents leftover plasmid DNA (exogenous SOX17) that still remains 4 days post-transfection, and any endogenous SOX17 expression that may have been induced.
- Ectoderm markers CDH1 , p63 and Sox2 were also up-regulated in some samples (e.g. Oct4/FoxD3/MBD2, Oct4/Sox17/MBD2).
- HFF cells were cultured as described in Example I, and transfected using the NucleofectorTM ® 96-well Shuttle ® Device (Lonza) following the procedure described in Example IV. The cells were transfected with various combinations of cDNA clones as described in Table 27. After transfection, the cells were plated on Fibronectin-coated collagen gels and incubated at 37°C, 5% C0 2 , 5% 0 2 . Fibronectin-coated Collagen gel plates were prepared prior to transfection.
- Rat Tail Collagen I (Gibco) was diluted to 1 .125mg/ml using 10X PBS and distilled water, where 125 ⁇ was added to each well of a 24-well plate and incubated in 37°C for 40 minutes. After rinsing with 1X PBS, Fibronectin (BD Biosciences) was added on top of the gel at a concentration of 1.9ug/well. Media was changed every other day according to Table 33. Cells were analyzed at Day 7 by Quantitative Real-time PCR. Table 33: Plasmids and media composition from Day 0 to Day 14
- Activin A (Peprotech, 30ng/ml), HSA (Baxter, 0.5%), NEAA (Gibco, 1X), ITS (Gibco, 1X), B27 (Gibco, 1 %), EGF (Peprotech, 5ng/ml), bFGF (Peprotech, 10ng/ml), CHIR99021 (Stemgent, 2uM), Na Butyrate (Stemgent, 1 mM), VPA (Stemgent, 1 mM), 5-Aza (Sigma, 0.5u ), Retinoic Acid (Sigma, 2uM), FGF10 (Peprotech, 50ng/ml), Cyclopamine (Stemgent, 2.5uM), Noggin (Peprotech, 50ng/ml)
- RNA isolation and quantification was performed as previously described in Example I.
- cDNA was prepared and quantitative real-time PCR was performed as previously described in Example II, except the following TaqmanTM® Gene
- Pancreatic Progenitor-like cells were investigated by transfection with combinations of FoxD3, Sox17, Pdx1 , Ngn3, Mixl1 , and MBD2.
- FoxA2 a marker for Endoderm and Mesoderm, was slightly up- regulated for the FoxD3/Sox17/Ngn3/MBD2-transfected sample as compared to the GFP mock- transfected control sample.
- CXCR4 also a marker for both endoderm and mesoderm, was slightly up-regulated (3-fold compared to GFP-ctrl) for the FoxD3/Mixl1/Ngn3/MBD2-transfected sample.
- SOX17 can still be detected for the samples transfected with SOX17 at varying levels (4 to 570-fold up-regulation as compared to the GFP-ctrl).
- the highest SOX17 expression up-regulation is detected for the sample transfected with Sox17/Mixl1/Pdx1/Ngn3 (570-fold as compared to GFP-ctrl), which may suggest that this gene combination may increase the amount of SOX17 RNA in cells.
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Cell Biology (AREA)
- Developmental Biology & Embryology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Rheumatology (AREA)
- Immunology (AREA)
- Reproductive Health (AREA)
- Virology (AREA)
- Epidemiology (AREA)
- Hematology (AREA)
- Ophthalmology & Optometry (AREA)
- Transplantation (AREA)
- Cardiology (AREA)
- Physical Education & Sports Medicine (AREA)
- Gynecology & Obstetrics (AREA)
- Urology & Nephrology (AREA)
- Dermatology (AREA)
Abstract
Description
Claims
Priority Applications (36)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP19170686.0A EP3567103B1 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| DK10825907.8T DK2494039T3 (en) | 2009-10-31 | 2010-11-01 | PROCEDURES FOR REPROGRAMMING CELLS AND APPLICATIONS THEREOF |
| CA2779310A CA2779310C (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| BR112012009921-3A BR112012009921B1 (en) | 2009-10-31 | 2010-11-01 | METHOD TO OBTAIN A NEURAL STEM CELL |
| MX2012004881A MX353245B (en) | 2009-10-31 | 2010-11-01 | METHODS FOR REPROGRAMMING CELLS and USES THEREOF. |
| HRP20191213TT HRP20191213T1 (en) | 2009-10-31 | 2010-11-01 | PROCEDURES FOR REPROGRAMMING STATIONS AND THEIR USES |
| EP10825907.8A EP2494039B1 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| MX2018000102A MX390753B (en) | 2009-10-31 | 2010-11-01 | METHODS FOR REPROGRAMMING CELLS AND THEIR USES. |
| ES10825907T ES2739672T3 (en) | 2009-10-31 | 2010-11-01 | Procedures for reprogramming cells and their uses |
| KR1020187008318A KR102014977B1 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| JP2012535565A JP5982286B2 (en) | 2009-10-31 | 2010-11-01 | Methods and uses for cell reprogramming |
| US13/504,988 US9528087B2 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| LTEP10825907.8T LT2494039T (en) | 2009-10-31 | 2010-11-01 | METHODS OF CELL REPLACEMENT AND THEIR USE |
| KR1020197024507A KR102134240B1 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| AU2010312240A AU2010312240B2 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| SI201031910T SI2494039T1 (en) | 2009-10-31 | 2010-11-01 | The process of reprogramming cells and their use |
| KR1020127014081A KR101874463B1 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| IL219453A IL219453A (en) | 2009-10-31 | 2012-04-29 | Methods for reprogramming cells and uses thereof |
| US13/464,987 US9453205B2 (en) | 2009-10-31 | 2012-05-05 | Methods for reprogramming cells and uses thereof |
| ZA2012/03902A ZA201203902B (en) | 2009-10-31 | 2012-05-29 | Methods for reprogramming cells and uses thereof |
| US13/843,713 US20140038291A1 (en) | 2009-10-31 | 2013-03-15 | Methods for reprogramming cells and uses thereof |
| US14/958,791 US20160160184A1 (en) | 2009-10-31 | 2015-12-03 | Methods for reprogramming cells and uses thereof |
| US15/275,921 US10563176B2 (en) | 2009-10-31 | 2016-09-26 | Methods for reprogramming cells and uses thereof |
| US15/297,972 US10017737B2 (en) | 2009-10-31 | 2016-10-19 | Methods for reprogramming cells and uses thereof |
| US15/298,034 US10260046B2 (en) | 2009-10-31 | 2016-10-19 | Methods for reprogramming cells and uses thereof |
| US15/298,006 US10131879B2 (en) | 2009-10-31 | 2016-10-19 | Methods for reprogramming cells and uses thereof |
| AU2017201774A AU2017201774B2 (en) | 2009-10-31 | 2017-03-15 | Methods for reprogramming cells and uses thereof |
| IL254144A IL254144A0 (en) | 2009-10-31 | 2017-08-24 | Methods for reprogramming cells and their use |
| US16/125,528 US12024722B2 (en) | 2009-10-31 | 2018-09-07 | Methods for reprogramming cells and uses thereof |
| US16/195,005 US10557123B2 (en) | 2009-10-31 | 2018-11-19 | Methods for reprogramming cells and uses thereof |
| AU2019202451A AU2019202451B2 (en) | 2009-10-31 | 2019-04-09 | Methods for reprogramming cells and uses thereof |
| US16/752,535 US11795439B2 (en) | 2009-10-31 | 2020-01-24 | Methods for reprogramming cells and uses thereof |
| US16/752,462 US20200165570A1 (en) | 2009-10-31 | 2020-01-24 | Methods for reprogramming cells and uses thereof |
| IL272848A IL272848B (en) | 2009-10-31 | 2020-02-23 | Methods for reprogramming cells and uses thereof |
| US18/188,421 US20230227785A1 (en) | 2009-10-31 | 2023-03-22 | Methods for reprogramming cells and uses thereof |
| US18/761,109 US20240344037A1 (en) | 2009-10-31 | 2024-07-01 | Methods for reprogramming cells and uses thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US25696709P | 2009-10-31 | 2009-10-31 | |
| US61/256,967 | 2009-10-31 |
Related Child Applications (6)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/504,988 A-371-Of-International US9528087B2 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
| US13/464,987 Continuation-In-Part US9453205B2 (en) | 2009-10-31 | 2012-05-05 | Methods for reprogramming cells and uses thereof |
| US13/464,987 Continuation US9453205B2 (en) | 2009-10-31 | 2012-05-05 | Methods for reprogramming cells and uses thereof |
| US15/298,034 Continuation US10260046B2 (en) | 2009-10-31 | 2016-10-19 | Methods for reprogramming cells and uses thereof |
| US15/297,972 Continuation US10017737B2 (en) | 2009-10-31 | 2016-10-19 | Methods for reprogramming cells and uses thereof |
| US15/298,006 Division US10131879B2 (en) | 2009-10-31 | 2016-10-19 | Methods for reprogramming cells and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011050476A1 true WO2011050476A1 (en) | 2011-05-05 |
Family
ID=43921216
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CA2010/001727 Ceased WO2011050476A1 (en) | 2009-10-31 | 2010-11-01 | Methods for reprogramming cells and uses thereof |
Country Status (17)
| Country | Link |
|---|---|
| US (7) | US9528087B2 (en) |
| EP (2) | EP3567103B1 (en) |
| JP (5) | JP5982286B2 (en) |
| KR (3) | KR101874463B1 (en) |
| AU (3) | AU2010312240B2 (en) |
| BR (1) | BR112012009921B1 (en) |
| CA (1) | CA2779310C (en) |
| DK (1) | DK2494039T3 (en) |
| ES (1) | ES2739672T3 (en) |
| HR (1) | HRP20191213T1 (en) |
| IL (3) | IL219453A (en) |
| LT (1) | LT2494039T (en) |
| MX (2) | MX390753B (en) |
| SG (1) | SG10201702120VA (en) |
| SI (1) | SI2494039T1 (en) |
| WO (1) | WO2011050476A1 (en) |
| ZA (1) | ZA201203902B (en) |
Cited By (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012068170A3 (en) * | 2010-11-15 | 2012-07-26 | Jau-Nan Lee | Generation of neural stem cells from human trophoblast stem cells |
| JP2013518584A (en) * | 2010-02-05 | 2013-05-23 | オークランド ユニサービシズ リミテッド | Cell programming |
| WO2013086570A1 (en) * | 2011-12-13 | 2013-06-20 | Flinders University Of South Australia | Method of producing multipotent stem cells |
| WO2013013105A3 (en) * | 2011-07-19 | 2013-07-11 | Vivoscript,Inc. | Compositions and methods for re-programming cells without genetic modification for repairing cartilage damage |
| US8497120B2 (en) | 2005-02-24 | 2013-07-30 | Jau-Nan Lee | Human trophoblast stem cells and use thereof |
| WO2013130196A1 (en) * | 2012-02-29 | 2013-09-06 | Mjn U.S. Holdings Llc | Neurogenesis screening method and system using adipose tissue derived stem cells |
| WO2013138368A1 (en) * | 2012-03-12 | 2013-09-19 | Loma Linda University Medical Center | Substances and methods for the treatment of cerebral amyloid angiopathy related conditions or diseases |
| US20140024119A1 (en) * | 2011-01-14 | 2014-01-23 | Korea Research Institute Of Bioscience And Biotechnology | Cell reprogramming composition comprising rex1 and an induced pluripotent stem cell production method using the same |
| WO2014015261A1 (en) * | 2012-07-19 | 2014-01-23 | The Penn State Research Foundation | Regenerating functional neurons for treatment of disease and injury in the nervous system |
| US20140134139A1 (en) * | 2012-11-07 | 2014-05-15 | Northwestern University | Cd34+ cells and methods of use |
| EP2773380A2 (en) * | 2011-11-03 | 2014-09-10 | UCL Business Plc. | Method for de-differentiating a cell |
| WO2014170488A1 (en) * | 2013-04-19 | 2014-10-23 | Universita' Degli Studi Di Milano | Methods for the conversion of somatic cells into pancreatic-hormone secreting cells |
| US20150030570A1 (en) * | 2012-02-29 | 2015-01-29 | Guangzhou Institutes of Biomedicine and Health Chinese Academy of Sciences | Culture medium for preparing neural stem cells and use thereof |
| WO2015084908A1 (en) * | 2013-12-03 | 2015-06-11 | The Johns Hopkins University | Method for highly efficient conversion of human stem cells to lineage-specific neurons |
| WO2015104424A1 (en) * | 2014-01-13 | 2015-07-16 | Centre Leon Berard | Method for producing induced pluripotent cells |
| CN105062953A (en) * | 2015-08-06 | 2015-11-18 | 深圳爱生再生医学科技有限公司 | Method for three-dimensional induction of transformation of mesenchymal stem cells into islet cells |
| US9414967B2 (en) | 2014-04-16 | 2016-08-16 | Loma Linda University | Composition, preparation, and use of chitosan shards for biomedical applications |
| US9428730B2 (en) | 2012-02-29 | 2016-08-30 | Mead Johnson Nutrition Company | Coatings and culture media for promoting neurogenesis in adipose tissue derived stem cells |
| US9457053B2 (en) | 2012-11-30 | 2016-10-04 | Accelerated Biosciences Corp. | Methods of differentiating stem cells by modulating MIR-124 |
| US9512406B2 (en) | 2013-12-20 | 2016-12-06 | The J. David Gladstone Institute, a testamentary trust established under the Will of J. David Gladstone | Generating hepatocytes |
| US9752127B2 (en) | 2013-07-27 | 2017-09-05 | Korea University Research And Business Foundation | Composition for maintaining chromosomal stability of pluripotent stem cells, containing small-molecule compounds |
| US9808490B2 (en) | 2014-11-26 | 2017-11-07 | Accelerated Biosciences Corp. | Induced hepatocytes and uses thereof |
| WO2017221155A1 (en) | 2016-06-20 | 2017-12-28 | Genesis Technologies Limited | Automated cell processing systems and methods |
| WO2018157769A1 (en) * | 2017-03-01 | 2018-09-07 | 中国科学院动物研究所 | Method for transdifferentiating non-neuronal cells into neuronal cells |
| EP3401391A1 (en) * | 2012-11-29 | 2018-11-14 | Takara Bio Europe AB | Maturation of hepatocyte-like cells derived from human pluripotent stem cells |
| CN110093309A (en) * | 2018-01-29 | 2019-08-06 | 中国科学院动物研究所 | A kind of induced fibroblast transdifferentiation is the method for fat cell |
| CN110093310A (en) * | 2018-01-29 | 2019-08-06 | 中国科学院动物研究所 | A kind of method and its application converting fibroblast to immortalized cells |
| CN110093305A (en) * | 2018-01-29 | 2019-08-06 | 中国科学院动物研究所 | A kind of method of inducing hepatocyte amplification in vitro |
| WO2019241846A1 (en) * | 2018-06-22 | 2019-12-26 | The University Of Sydney | Expansion and differentiation of neuronal precursor cells |
| US20200087626A1 (en) * | 2018-09-19 | 2020-03-19 | Regeneration Biomedical, Inc. | Wnt-activated adipose-derived stem cell apparatuses, methods and systems |
| CN111344392A (en) * | 2018-01-29 | 2020-06-26 | 中国科学院动物研究所 | A method of cell induction |
| EP3702443A1 (en) * | 2013-03-14 | 2020-09-02 | The Brigham and Women's Hospital, Inc. | Compositions and methods for epithelial stem cell expansion and culture |
| US10973930B2 (en) | 2016-02-18 | 2021-04-13 | The Penn State Research Foundation | Generating GABAergic neurons in brains |
| US11021687B2 (en) | 2016-01-08 | 2021-06-01 | The Brigham And Women's Hospital, Inc. | Production of differentiated enteroendocrine cells and insulin producing cells |
| US11033546B2 (en) | 2016-03-02 | 2021-06-15 | Frequency Therapeutics, Inc. | Solubilized compositions for controlled proliferation of stem cells / generating inner ear hair cells using a GSK3 inhibitor: I |
| US11066419B2 (en) | 2016-12-30 | 2021-07-20 | Frequency Therapeutics, Inc. | 1H-pyrrole-2,5-dione compounds and methods of using same |
| US11162071B2 (en) | 2018-08-17 | 2021-11-02 | Frequency Therapeutics, Inc. | Compositions and methods for generating hair cells by upregulating JAG-1 |
| US11160868B2 (en) | 2016-03-02 | 2021-11-02 | Frequency Therapeutics, Inc. | Thermoreversible compositions for administration of therapeutic agents |
| US11260130B2 (en) | 2016-03-02 | 2022-03-01 | Frequency Therapeutics, Inc. | Solubilized compositions for controlled proliferation of stem cells / generating inner ear hair cells using a GSK3 inhibitor: IV |
| US11369607B2 (en) | 2014-09-03 | 2022-06-28 | The Brigham And Women's Hospital, Inc. | Compositions, systems, and methods for generating inner ear hair cells for treatment of hearing loss |
| US11617745B2 (en) | 2018-08-17 | 2023-04-04 | Frequency Therapeutics, Inc. | Compositions and methods for generating hair cells by downregulating FOXO |
| US12077777B2 (en) | 2019-05-06 | 2024-09-03 | Accelerated Biosciences Corp. | Precursory regulatory cytotrophoblast cells and uses thereof |
Families Citing this family (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20140038291A1 (en) | 2009-10-31 | 2014-02-06 | New World Laboratories Inc. | Methods for reprogramming cells and uses thereof |
| US9453205B2 (en) | 2009-10-31 | 2016-09-27 | Genesis Technologies Limited | Methods for reprogramming cells and uses thereof |
| CA2779310C (en) * | 2009-10-31 | 2021-11-30 | New World Laboratories Inc. | Methods for reprogramming cells and uses thereof |
| WO2012087965A2 (en) | 2010-12-22 | 2012-06-28 | Fate Therapauetics, Inc. | Cell culture platform for single cell sorting and enhanced reprogramming of ipscs |
| AU2013359076A1 (en) * | 2012-12-14 | 2015-07-02 | Anthrogenesis Corporation | Anoikis resistant placental stem cells and uses thereof |
| US9982232B2 (en) * | 2012-12-17 | 2018-05-29 | John PIMANDA | Methods of generating cells with multilineage potential |
| EP2970881A4 (en) * | 2013-03-14 | 2017-01-25 | Children's Medical Center Corporation | Compositions and methods for reprogramming hematopoietic stem cell lineages |
| US9828582B2 (en) | 2013-03-19 | 2017-11-28 | Duke University | Compositions and methods for the induction and tuning of gene expression |
| US11221327B2 (en) | 2013-10-10 | 2022-01-11 | Mcmaster University | Method for diagnosing and monitoring inflammatory disease progression |
| WO2015051451A1 (en) * | 2013-10-10 | 2015-04-16 | Mcmaster University | Diagnosis and monitoring of endometriosis through bdnf and full-length ntrk2 levels |
| EP3099786A1 (en) * | 2014-01-29 | 2016-12-07 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Trans-differentiation of differentiated cells |
| SG11201606934SA (en) | 2014-03-04 | 2016-09-29 | Fate Therapeutics Inc | Improved reprogramming methods and cell culture platforms |
| US20150353623A1 (en) * | 2014-04-03 | 2015-12-10 | Loma Linda University | Substances and methods for the treatment of cerebral amyloid angiopathy related conditions or diseases |
| KR101731624B1 (en) * | 2014-07-01 | 2017-05-04 | 광주과학기술원 | Compositions for inducing a cell re-programming |
| US9730975B2 (en) | 2014-11-25 | 2017-08-15 | The Penn State Research Foundation | Chemical reprogramming of human glial cells into neurons for brain and spinal cord repair |
| MA41296A (en) * | 2014-12-30 | 2017-11-07 | Orgenesis Ltd | TRANSDIFFERENTIATION PROCESSES AND METHODS FOR USING THE SAME |
| US10676726B2 (en) | 2015-02-09 | 2020-06-09 | Duke University | Compositions and methods for epigenome editing |
| JP6841609B2 (en) | 2015-07-10 | 2021-03-10 | 3スキャン インコーポレイテッド | Spatial multiplexing of histological staining |
| SG10202111851YA (en) | 2015-10-16 | 2021-12-30 | Fate Therapeutics Inc | Platform for the Induction & Maintenance of Ground State Pluripotency |
| EA201891317A3 (en) | 2015-11-30 | 2019-04-30 | Дьюк Юниверсити | THERAPEUTIC TARGETS FOR CORRECTION OF HUMAN DISTROPHIN GENE BY EDITING GENES AND METHODS OF THEIR APPLICATION |
| US20190127713A1 (en) | 2016-04-13 | 2019-05-02 | Duke University | Crispr/cas9-based repressors for silencing gene targets in vivo and methods of use |
| CN107142240B (en) * | 2016-06-03 | 2021-01-29 | 中国人民解放军军事医学科学院野战输血研究所 | Method for reprogramming epithelial cells derived from digestive tract into endoderm stem/progenitor cells and application |
| CN105861447B (en) * | 2016-06-13 | 2017-12-19 | 广州市搏克生物技术有限公司 | A kind of non-viral iPSCs inducing compositions and its kit |
| WO2018017754A1 (en) | 2016-07-19 | 2018-01-25 | Duke University | Therapeutic applications of cpf1-based genome editing |
| CA3054120A1 (en) * | 2017-02-24 | 2018-08-30 | Koji Tanabe | Nerve cell production method by introduction of inducing factor rna |
| JP6916318B2 (en) * | 2017-06-30 | 2021-08-11 | ステムラボ・インコーポレイテッド | A method for directly redifferentiating urinary cells to keratinocyte stem cells and a method for producing a composition for promoting skin regeneration using the reversely differentiated keratinocyte stem cells. |
| WO2019144061A1 (en) | 2018-01-19 | 2019-07-25 | Duke University | Genome engineering with crispr-cas systems in eukaryotes |
| EP3746109A4 (en) * | 2018-02-02 | 2021-11-03 | The Penn State Research Foundation | Methods and materials for treating brain injuries |
| CN110305846A (en) * | 2018-03-20 | 2019-10-08 | 中山大学中山眼科中心 | The preparation method of pattern of retinal ganglion cells |
| US11518971B2 (en) | 2018-11-27 | 2022-12-06 | Research Triangle Institute | Method and apparatus for spatial control of cellular growth |
| KR102450790B1 (en) * | 2019-01-02 | 2022-10-06 | 주식회사 셀라퓨틱스바이오 | Novel glial-like cells differentiated from somatic cells, a method for preparing the same, a cocktail composition for the preparation thereof, a cell therapeutic agent for preventing or treating a neurological disease comprising the same, and a method for preventing and treating a neurological disease by administering the same |
| AU2020207410A1 (en) | 2019-01-11 | 2021-08-19 | Figene, Llc | Fibroblast regenerative cells |
| KR102183208B1 (en) * | 2019-07-25 | 2020-11-25 | 한국과학기술연구원 | Methods for reprogramming astrocytes into neurons in spinal cord injury(SCI) animal model using Ngn2 |
| AU2020331968A1 (en) * | 2019-08-19 | 2022-03-10 | Duke University | Compositions and methods for identifying regulators of cell type fate specification |
| IL293685A (en) * | 2019-12-09 | 2022-08-01 | Brigham & Womens Hospital Inc | Methods for generating hematopoietic stem cells |
| CN111471651A (en) * | 2020-04-24 | 2020-07-31 | 中国人民解放军第四军医大学 | A kind of human adipose stem cell serum-free medium and preparation method thereof |
| CN113862218B (en) * | 2021-10-12 | 2024-06-25 | 广州医科大学附属第一医院(广州呼吸中心) | A tumor-related vascular pericyte subpopulation and its preparation method and application |
| US12209394B2 (en) * | 2022-02-16 | 2025-01-28 | Caterpillar Inc. | Adapter retention plug |
| CN120098922A (en) * | 2025-03-10 | 2025-06-06 | 中山大学 | Composition and method for reprogramming astrocytes into functional neurons |
| CN121221644B (en) * | 2025-12-01 | 2026-03-03 | 北京国卫生物科技有限公司 | Application of umbilical cord mesenchymal stem cells in the treatment of early-onset ovarian insufficiency |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6087168A (en) * | 1999-01-20 | 2000-07-11 | Cedars Sinai Medical Center | Conversion of non-neuronal cells into neurons: transdifferentiation of epidermal cells |
| CA2698091A1 (en) * | 2007-08-31 | 2009-03-12 | Brett Chevalier | Wnt pathway stimulation in reprogramming somatic cells |
| CA2660123A1 (en) * | 2007-10-31 | 2009-04-30 | Kyoto University | Nuclear reprogramming method |
Family Cites Families (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020136709A1 (en) | 2000-12-12 | 2002-09-26 | Nucleus Remodeling, Inc. | In vitro-derived adult pluripotent stem cells and uses therefor |
| AU2002313817A1 (en) | 2001-08-27 | 2003-03-10 | Advanced Cell Technology, Inc. | Trans-differentiation and re-differentiation of somatic cells and production of cells for cell therapies |
| US20030217378A1 (en) | 2002-04-05 | 2003-11-20 | The University Of Georgia Research Foundation, Inc | Cloning using rapidly matured oocytes |
| KR100519227B1 (en) * | 2002-08-17 | 2005-10-07 | 서해영 | A method for transdifferentiating mesenchymal stem cells into neuronal cells |
| EP1856248A4 (en) | 2005-02-09 | 2010-01-20 | Burnham Inst Medical Research | HOMOGENEOUS NEURAL PRECURSOR CELLS |
| US8440460B2 (en) | 2005-07-27 | 2013-05-14 | The Board Of Regents Of The University Of Texas System | Methods for transdifferentiating cells |
| CN101389761A (en) | 2006-02-27 | 2009-03-18 | 银怎株式会社 | Dedifferentiation of astrocytes into neural stem cells using BMI-1 |
| WO2008073331A2 (en) * | 2006-12-07 | 2008-06-19 | Teva Pharmaceutical Industries Ltd. | Method of generation and expansion of tissue-progenitor cells and mature tissue cells from intact bone marrow or intact umbilical cord tissue |
| WO2009018832A1 (en) * | 2007-08-09 | 2009-02-12 | Rigshospitalet | Method for increasing the plasticity level of a cell |
| US20090136461A1 (en) * | 2007-11-28 | 2009-05-28 | Moon Suk Kim | Neuronal differentiation method of adult stem cells using small molecules |
| AU2008333972A1 (en) * | 2007-11-30 | 2009-06-11 | New York Medical College | Compositions comprising HDAC inhibitors and methods of their use in restoring stem cell function and preventing heart failure |
| US20100330063A1 (en) | 2007-12-17 | 2010-12-30 | Gliamed, Inc. | Stem-like cells, method for de-differentiating mammalian somatic cells into stem-like cells, and method for differentiating stem-like cells |
| SG188904A1 (en) * | 2008-03-17 | 2013-04-30 | Scripps Research Inst | Combined chemical and genetic approaches for generation of induced pluripotent stem cells |
| AU2009234424A1 (en) * | 2008-04-07 | 2009-10-15 | Nupotential, Inc. | Reprogramming a cell by inducing a pluripotent gene through use of an HDAC modulator |
| CN102272293B (en) | 2008-11-05 | 2014-06-04 | 学校法人庆应义塾 | Method for producing neural stem cells |
| WO2010088735A1 (en) | 2009-02-05 | 2010-08-12 | Regenertech Pty Ltd | Method of producing progenitor cells from differentiated cells |
| US20120115225A1 (en) * | 2009-04-23 | 2012-05-10 | Xu C W | Reprogramming of somatic cells with purified proteins |
| CN102498399A (en) * | 2009-05-20 | 2012-06-13 | 卡迪欧参生物科技有限公司 | Method for determining the cardiac growth potential of mammalian cells |
| CA2779310C (en) * | 2009-10-31 | 2021-11-30 | New World Laboratories Inc. | Methods for reprogramming cells and uses thereof |
| US9453205B2 (en) * | 2009-10-31 | 2016-09-27 | Genesis Technologies Limited | Methods for reprogramming cells and uses thereof |
| US20130097717A1 (en) * | 2010-06-23 | 2013-04-18 | Vivoscript, Inc. | Compositions and methods for re-programming cells without genetic modification for treatment of cardiovascular diseases |
| WO2013033213A1 (en) * | 2011-08-30 | 2013-03-07 | The J. David Gladstone Institutes | Methods for generating cardiomyocytes |
| WO2013116307A1 (en) | 2012-01-30 | 2013-08-08 | Mount Sinai School Of Medicine | Method for programming differentiated cells into hematopoietic stem cells |
-
2010
- 2010-11-01 CA CA2779310A patent/CA2779310C/en active Active
- 2010-11-01 LT LTEP10825907.8T patent/LT2494039T/en unknown
- 2010-11-01 KR KR1020127014081A patent/KR101874463B1/en active Active
- 2010-11-01 WO PCT/CA2010/001727 patent/WO2011050476A1/en not_active Ceased
- 2010-11-01 ES ES10825907T patent/ES2739672T3/en active Active
- 2010-11-01 BR BR112012009921-3A patent/BR112012009921B1/en not_active IP Right Cessation
- 2010-11-01 KR KR1020187008318A patent/KR102014977B1/en active Active
- 2010-11-01 SG SG10201702120VA patent/SG10201702120VA/en unknown
- 2010-11-01 MX MX2018000102A patent/MX390753B/en unknown
- 2010-11-01 US US13/504,988 patent/US9528087B2/en active Active
- 2010-11-01 EP EP19170686.0A patent/EP3567103B1/en active Active
- 2010-11-01 JP JP2012535565A patent/JP5982286B2/en active Active
- 2010-11-01 KR KR1020197024507A patent/KR102134240B1/en active Active
- 2010-11-01 SI SI201031910T patent/SI2494039T1/en unknown
- 2010-11-01 EP EP10825907.8A patent/EP2494039B1/en active Active
- 2010-11-01 MX MX2012004881A patent/MX353245B/en active IP Right Grant
- 2010-11-01 DK DK10825907.8T patent/DK2494039T3/en active
- 2010-11-01 AU AU2010312240A patent/AU2010312240B2/en active Active
- 2010-11-01 HR HRP20191213TT patent/HRP20191213T1/en unknown
-
2012
- 2012-04-29 IL IL219453A patent/IL219453A/en active IP Right Grant
- 2012-05-29 ZA ZA2012/03902A patent/ZA201203902B/en unknown
-
2016
- 2016-03-24 JP JP2016059620A patent/JP2016146841A/en active Pending
- 2016-10-19 US US15/298,006 patent/US10131879B2/en active Active
- 2016-10-19 US US15/298,034 patent/US10260046B2/en active Active
- 2016-10-19 US US15/297,972 patent/US10017737B2/en active Active
-
2017
- 2017-03-15 AU AU2017201774A patent/AU2017201774B2/en active Active
- 2017-08-24 IL IL254144A patent/IL254144A0/en unknown
-
2018
- 2018-07-04 JP JP2018127585A patent/JP2018183152A/en active Pending
- 2018-11-19 US US16/195,005 patent/US10557123B2/en active Active
-
2019
- 2019-04-09 AU AU2019202451A patent/AU2019202451B2/en active Active
-
2020
- 2020-01-24 US US16/752,462 patent/US20200165570A1/en not_active Abandoned
- 2020-02-23 IL IL272848A patent/IL272848B/en active IP Right Grant
- 2020-05-07 JP JP2020082207A patent/JP2020124219A/en active Pending
-
2022
- 2022-06-16 JP JP2022097154A patent/JP7407865B2/en active Active
-
2023
- 2023-03-22 US US18/188,421 patent/US20230227785A1/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6087168A (en) * | 1999-01-20 | 2000-07-11 | Cedars Sinai Medical Center | Conversion of non-neuronal cells into neurons: transdifferentiation of epidermal cells |
| US6949380B1 (en) | 1999-01-20 | 2005-09-27 | Cedars-Sinai Medical Center | Transdifferentiation of epidermal basal cells into neural progenitor cells, neuronal cells and/or glial cells |
| CA2698091A1 (en) * | 2007-08-31 | 2009-03-12 | Brett Chevalier | Wnt pathway stimulation in reprogramming somatic cells |
| CA2660123A1 (en) * | 2007-10-31 | 2009-04-30 | Kyoto University | Nuclear reprogramming method |
Non-Patent Citations (17)
| Title |
|---|
| ABELIOVICH A ET AL.: "Reprogramming Therapeutics: iPS Cell Prospects for Neurodegenerative Disease", NEURON, vol. 61, no. 3, 12 February 2009 (2009-02-12), pages 337 - 339, XP008158166 * |
| CHAMBERS SM; FASANO CA; PAPAPETROU EP; TOMISHIMA M; SADELAIN M; STUDER L: "Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling", NAT BIOTECHNOL., vol. 27, no. 3, 2009, pages 275 - 80, XP055007827, DOI: doi:10.1038/nbt.1529 |
| FENG B ET AL.: "Reprogramming of fibroblasts into induced pluripotent stem cells with orphan nuclear receptor Esrrb", NAT. CELL BIOL., vol. 11, no. 2, February 2009 (2009-02-01), pages 197 - 203, XP002606352 * |
| FERNANDES KJL; MCKENZIE IA; MILL P; SMITH KM; AKHAVAN M; BARNABE-HEIDER F; BIERNASKIE J; JUNEK A ET AL.: "A dermal niche for multipotent adult skin-derived precursor cells", NATURE CELL BIOLOGY, vol. 6, 2004, pages 1082 - 1093, XP003011063, DOI: doi:10.1038/ncb1181 |
| FODE C; GRADWOHL G; MORIN X; DIERICH A; LEMEUR M; GORIDIS C; GUILLEMOT F: "The bHLH protein NEUROGENIN 2 is a determination factor for epibranchial placode-derived sensory neurons", NEURON, vol. 20, no. 3, 1998, pages 483 - 94 |
| GASH DM; ZHANG Z; OVADIA A; CASS WA; YI A; SIMMERMAN L; RUSSEL D; MARTIN D; LAPCHAK PA; COLLINS F: "Functional recovery in parkinsonian monkeys treated with GDNF", NATURE, vol. 380, no. 6571, 1996, pages 252 - 5 |
| GUILLEMOT F; LO LC; JOHNSON JE; AUERBACH A; ANDERSON DJ; JOYNER AL: "Mammalian achaete-scute homolog 1 is required for the early development of olfactory and autonomic neurons", CELL, vol. 75, no. 3, 1993, pages 463 - 76, XP024245943, DOI: doi:10.1016/0092-8674(93)90381-Y |
| JACOBSEN F; HIRSCH T; MITTLER D; SCHULTE M; LEHNHARDT M; DRUECKE D; HOMANN HH; STEINAU HU; STEINSTRAESSER L: "Polybrene improves transfection efficacy of recombinant replication-deficient adenovirus in cutaneous cells and burned skin", J GENE MED, vol. 8, no. 2, 2006, pages 138 - 46 |
| KANEKO Y ET AL.: "Musashi 1: An Evolutionally Conserved Marker for CNS Progenitor Cells Including Neural Stem Cells", DEV NEUROSCI.., vol. 22, no. 1-2, February 2000 (2000-02-01), pages 139 - 153, XP001033925 * |
| KEARNS CM; GASH DM: "GDNF protects nigral dopamine neurons against 6-hydroxydopamine in vivo", BRAIN RES, vol. 672, no. 1-2, 1995, pages 104 - 11, XP002293325, DOI: doi:10.1016/0006-8993(94)01366-P |
| KIM D ET AL.: "Generation of Human Induced Pluripotent Stem Cells by Direct Delivery of Reprogramming Proteins", CELL STEM CELL, vol. 4, no. 6, 5 June 2009 (2009-06-05), pages 472 - 476, XP002564507 * |
| KORDOWER JH; EMBORG ME; BLOCH J; MA SY; CHU Y; LEVENTHAL L; MCBRIDE J; CHEN EY; PALFI S; ROITBERG BZ: "Neurodegeneration prevented by lentiviral vector delivery of GDNF in primate models of Parkinson's disease", SCIENCE, vol. 290, no. 5492, 2000, pages 767 - 73, XP002207711, DOI: doi:10.1126/science.290.5492.767 |
| LINDNER MD; WINN SR; BAETGE EE; HAMMANG JP; GENTILE FT; DOHERTY E; MCDERMOTT PE; FRYDEL B; ULLMAN MD; SCHALLERT T ET AL.: "Implantation of encapsulated catecholamine and GDNF-producing cells in rats with unilateral dopamine depletions and parkinsonian symptoms", EXP NEUROL., vol. 132, no. 1, 1995, pages 62 - 76, XP004537486, DOI: doi:10.1016/0014-4886(95)90059-4 |
| MARTINEZ-SERRANO A; BJORKLUND A: "Immortalized neural progenitor cells for CNS gene transfer and repair", TRENDS NEUROSCI., vol. 20, no. 11, 1997, pages 530 - 8, XP002926796, DOI: doi:10.1016/S0166-2236(97)01119-3 |
| PEI, D.: "Regulation of Pluripotency and Reprogramming by Transcription Factors", J. BIOL. CHEM., vol. 284, no. 6, 6 February 2009 (2009-02-06), pages 3365 - 3369, XP002560044 * |
| SILVA J ET AL.: "Nanog Is the Gateway to the Pluripotent Ground State", CELL, vol. 138, no. 4, 21 August 2009 (2009-08-21), pages 722 - 737, XP008158163 * |
| TAKAHASHI K ET AL.: "Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors", CELL., vol. 126, no. 4, 25 August 2006 (2006-08-25), pages 663 - 676 * |
Cited By (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9335322B2 (en) | 2005-02-24 | 2016-05-10 | Accelerated Biosciences Corporation | Human trophoblast stem cells and methods of therapeutic screening |
| US8497120B2 (en) | 2005-02-24 | 2013-07-30 | Jau-Nan Lee | Human trophoblast stem cells and use thereof |
| US9927426B2 (en) | 2005-02-24 | 2018-03-27 | Accelerated Biosciences Corp. | Methods of treating diseases with human trophoblast stem cells |
| US12066431B2 (en) | 2005-02-24 | 2024-08-20 | Accelerated Biosciences Corp. | Human trophoblast stem cells and uses thereof |
| US10746728B2 (en) | 2005-02-24 | 2020-08-18 | Accelerated Biosciences Corp. | Human trophoblast stem cells and uses thereof |
| US10983111B2 (en) | 2005-02-24 | 2021-04-20 | Accelerated Biosciences Corp. | Human trophoblast stem cells and uses thereof |
| JP2013518584A (en) * | 2010-02-05 | 2013-05-23 | オークランド ユニサービシズ リミテッド | Cell programming |
| US11891623B2 (en) | 2010-11-15 | 2024-02-06 | Accelerated Biosciences Corp. | Generation of neural stem cells from human trophoblast stem cells |
| US11254911B2 (en) | 2010-11-15 | 2022-02-22 | Accelerated Biosciences Corp. | Generation of neural stem cells from human trophoblast stem cells |
| US9574173B2 (en) | 2010-11-15 | 2017-02-21 | Accelerated Biosciences Corp. | Generation of neural stem cells from human trophoblast stem cells |
| GB2502704A (en) * | 2010-11-15 | 2013-12-04 | Jau-Nan Lee | Generation of neural stem cells from human trophoblast stem cells |
| WO2012068170A3 (en) * | 2010-11-15 | 2012-07-26 | Jau-Nan Lee | Generation of neural stem cells from human trophoblast stem cells |
| GB2502704B (en) * | 2010-11-15 | 2019-12-04 | Accelerated Biosciences Corp | Generation of neural stem cells from human trophoblast stem cells |
| US20140024119A1 (en) * | 2011-01-14 | 2014-01-23 | Korea Research Institute Of Bioscience And Biotechnology | Cell reprogramming composition comprising rex1 and an induced pluripotent stem cell production method using the same |
| CN103857797A (en) * | 2011-07-19 | 2014-06-11 | 帷幄生物技术公司 | Compositions and methods for re-programming cells without genetic modification for repairing cartilage damage |
| WO2013013105A3 (en) * | 2011-07-19 | 2013-07-11 | Vivoscript,Inc. | Compositions and methods for re-programming cells without genetic modification for repairing cartilage damage |
| EP2773380A2 (en) * | 2011-11-03 | 2014-09-10 | UCL Business Plc. | Method for de-differentiating a cell |
| KR102029391B1 (en) | 2011-12-13 | 2019-11-08 | 유니사 벤쳐스 피티와이 엘티디 | Method of producing multipotent stem cells |
| US10196606B2 (en) | 2011-12-13 | 2019-02-05 | Unisa Ventures Pty Ltd | Method of producing multipotent stem cells |
| KR20140103132A (en) * | 2011-12-13 | 2014-08-25 | 플린더스 유니버시티 오브 사우스 오스트레일리아 | Method of producing multipotent stem cells |
| AU2012350353B2 (en) * | 2011-12-13 | 2018-01-04 | Unisa Ventures Pty Ltd | Method of producing multipotent stem cells |
| WO2013086570A1 (en) * | 2011-12-13 | 2013-06-20 | Flinders University Of South Australia | Method of producing multipotent stem cells |
| CN104285147A (en) * | 2012-02-29 | 2015-01-14 | Mjn美国控股有限责任公司 | Neurogenesis screening method and system using adipose tissue derived stem cells |
| CN104285147B (en) * | 2012-02-29 | 2017-10-27 | Mjn 美国控股有限责任公司 | Use the nerve to occur screening technique and system of adipose tissue-derived stem cell |
| US20150030570A1 (en) * | 2012-02-29 | 2015-01-29 | Guangzhou Institutes of Biomedicine and Health Chinese Academy of Sciences | Culture medium for preparing neural stem cells and use thereof |
| US9428730B2 (en) | 2012-02-29 | 2016-08-30 | Mead Johnson Nutrition Company | Coatings and culture media for promoting neurogenesis in adipose tissue derived stem cells |
| WO2013130196A1 (en) * | 2012-02-29 | 2013-09-06 | Mjn U.S. Holdings Llc | Neurogenesis screening method and system using adipose tissue derived stem cells |
| US10011819B2 (en) * | 2012-02-29 | 2018-07-03 | Guangzhou Institutes Of Biomedicine And Health, Chinese Academy Of Sciences | Culture medium for preparing neural stem cells and use thereof |
| WO2013138368A1 (en) * | 2012-03-12 | 2013-09-19 | Loma Linda University Medical Center | Substances and methods for the treatment of cerebral amyloid angiopathy related conditions or diseases |
| US11167044B2 (en) | 2012-07-19 | 2021-11-09 | The Penn State Research Foundation | Regenerating functional neurons for treatment of disease in the nervous system |
| US9717804B2 (en) | 2012-07-19 | 2017-08-01 | The Penn State Research Foundation | Regenerating functional neurons for treatment of disease and injury in the nervous system |
| US10561742B2 (en) | 2012-07-19 | 2020-02-18 | The Penn State Research Foundation | Methods and compositions for treatment of disease or injury of the nervous system |
| US10201619B2 (en) | 2012-07-19 | 2019-02-12 | The Penn State Research Foundation | Regenerating functional neurons for treatment of disease and injury in the nervous system |
| WO2014015261A1 (en) * | 2012-07-19 | 2014-01-23 | The Penn State Research Foundation | Regenerating functional neurons for treatment of disease and injury in the nervous system |
| US20140134139A1 (en) * | 2012-11-07 | 2014-05-15 | Northwestern University | Cd34+ cells and methods of use |
| EP3401391A1 (en) * | 2012-11-29 | 2018-11-14 | Takara Bio Europe AB | Maturation of hepatocyte-like cells derived from human pluripotent stem cells |
| US10294457B2 (en) | 2012-11-29 | 2019-05-21 | Takara Bio Europe Ab | Maturation of hepatocyte-like cells derived from human pluripotent stem cells |
| US9457053B2 (en) | 2012-11-30 | 2016-10-04 | Accelerated Biosciences Corp. | Methods of differentiating stem cells by modulating MIR-124 |
| US10294458B2 (en) | 2012-11-30 | 2019-05-21 | Accelerated Biosciences Corp. | Methods of differentiating stem cells by modulating miR-124 |
| US11028366B2 (en) | 2012-11-30 | 2021-06-08 | Accelerated Biosciences Corp. | Methods of differentiating stem cells by modulating MIR-124 |
| US10954490B2 (en) | 2013-03-14 | 2021-03-23 | The Brigham And Women's Hospital, Inc. | Compositions and methods for epithelial stem cell expansion and culture |
| EP3702443A1 (en) * | 2013-03-14 | 2020-09-02 | The Brigham and Women's Hospital, Inc. | Compositions and methods for epithelial stem cell expansion and culture |
| WO2014170488A1 (en) * | 2013-04-19 | 2014-10-23 | Universita' Degli Studi Di Milano | Methods for the conversion of somatic cells into pancreatic-hormone secreting cells |
| US9752127B2 (en) | 2013-07-27 | 2017-09-05 | Korea University Research And Business Foundation | Composition for maintaining chromosomal stability of pluripotent stem cells, containing small-molecule compounds |
| WO2015084908A1 (en) * | 2013-12-03 | 2015-06-11 | The Johns Hopkins University | Method for highly efficient conversion of human stem cells to lineage-specific neurons |
| US11104881B2 (en) | 2013-12-03 | 2021-08-31 | The Johns Hopkins University | Method for highly efficient conversion of human stem cells to lineage-specific neurons |
| US10227565B2 (en) | 2013-12-20 | 2019-03-12 | The J. David Gladstone Institutes | Generating hepatocytes |
| US9512406B2 (en) | 2013-12-20 | 2016-12-06 | The J. David Gladstone Institute, a testamentary trust established under the Will of J. David Gladstone | Generating hepatocytes |
| WO2015104424A1 (en) * | 2014-01-13 | 2015-07-16 | Centre Leon Berard | Method for producing induced pluripotent cells |
| FR3016372A1 (en) * | 2014-01-13 | 2015-07-17 | Ct Leon Berard | PROCESS FOR THE PREPARATION OF INDUCED PLURIPOTENT CELLS |
| CN106459909A (en) * | 2014-01-13 | 2017-02-22 | 利昂贝拉尔中心 | Method for producing induced pluripotent cells |
| US9414967B2 (en) | 2014-04-16 | 2016-08-16 | Loma Linda University | Composition, preparation, and use of chitosan shards for biomedical applications |
| US11369607B2 (en) | 2014-09-03 | 2022-06-28 | The Brigham And Women's Hospital, Inc. | Compositions, systems, and methods for generating inner ear hair cells for treatment of hearing loss |
| US10765704B2 (en) | 2014-11-26 | 2020-09-08 | Accelerated Biosciences Corp. | Induced hepatocytes and uses thereof |
| US9808490B2 (en) | 2014-11-26 | 2017-11-07 | Accelerated Biosciences Corp. | Induced hepatocytes and uses thereof |
| US11026979B2 (en) | 2014-11-26 | 2021-06-08 | Accelerated Biosciences Corp. | Human hepatocytes and uses thereof |
| CN105062953A (en) * | 2015-08-06 | 2015-11-18 | 深圳爱生再生医学科技有限公司 | Method for three-dimensional induction of transformation of mesenchymal stem cells into islet cells |
| US11021687B2 (en) | 2016-01-08 | 2021-06-01 | The Brigham And Women's Hospital, Inc. | Production of differentiated enteroendocrine cells and insulin producing cells |
| US10973930B2 (en) | 2016-02-18 | 2021-04-13 | The Penn State Research Foundation | Generating GABAergic neurons in brains |
| US11033546B2 (en) | 2016-03-02 | 2021-06-15 | Frequency Therapeutics, Inc. | Solubilized compositions for controlled proliferation of stem cells / generating inner ear hair cells using a GSK3 inhibitor: I |
| US11160868B2 (en) | 2016-03-02 | 2021-11-02 | Frequency Therapeutics, Inc. | Thermoreversible compositions for administration of therapeutic agents |
| US11260130B2 (en) | 2016-03-02 | 2022-03-01 | Frequency Therapeutics, Inc. | Solubilized compositions for controlled proliferation of stem cells / generating inner ear hair cells using a GSK3 inhibitor: IV |
| US11796552B2 (en) | 2016-06-20 | 2023-10-24 | Genesis Technologies Limited | Automated cell processing systems and methods |
| WO2017221155A1 (en) | 2016-06-20 | 2017-12-28 | Genesis Technologies Limited | Automated cell processing systems and methods |
| US11125767B2 (en) | 2016-06-20 | 2021-09-21 | Genesis Technologies Limited | Automated cell processing systems and methods |
| US11156627B2 (en) | 2016-06-20 | 2021-10-26 | Genesis Technologies Limited | Automated cell processing systems and methods |
| US11066419B2 (en) | 2016-12-30 | 2021-07-20 | Frequency Therapeutics, Inc. | 1H-pyrrole-2,5-dione compounds and methods of using same |
| WO2018157769A1 (en) * | 2017-03-01 | 2018-09-07 | 中国科学院动物研究所 | Method for transdifferentiating non-neuronal cells into neuronal cells |
| CN110093310A (en) * | 2018-01-29 | 2019-08-06 | 中国科学院动物研究所 | A kind of method and its application converting fibroblast to immortalized cells |
| CN110093305A (en) * | 2018-01-29 | 2019-08-06 | 中国科学院动物研究所 | A kind of method of inducing hepatocyte amplification in vitro |
| CN110093309A (en) * | 2018-01-29 | 2019-08-06 | 中国科学院动物研究所 | A kind of induced fibroblast transdifferentiation is the method for fat cell |
| CN111344392B (en) * | 2018-01-29 | 2021-07-30 | 中国科学院动物研究所 | Cell induction method |
| US12163156B2 (en) | 2018-01-29 | 2024-12-10 | Institute Of Zoology, Chinese Academy Of Sciences | Cell induction method |
| CN110093305B (en) * | 2018-01-29 | 2021-07-02 | 中国科学院动物研究所 | A method for inducing hepatocyte expansion in vitro |
| CN110093309B (en) * | 2018-01-29 | 2021-07-02 | 中国科学院动物研究所 | A method for inducing transdifferentiation of fibroblasts into adipocytes |
| CN110093310B (en) * | 2018-01-29 | 2021-07-02 | 中国科学院动物研究所 | A method for transforming fibroblasts into immortalized cells and application thereof |
| CN111344392A (en) * | 2018-01-29 | 2020-06-26 | 中国科学院动物研究所 | A method of cell induction |
| US20210269770A1 (en) * | 2018-06-22 | 2021-09-02 | Skin2Neuron Pty Ltd | Expansion and differentiation of neuronal precursor cells |
| AU2019290037B2 (en) * | 2018-06-22 | 2022-11-03 | Skin2Neuron Pty Ltd | Expansion and differentiation of neuronal precursor cells |
| WO2019241846A1 (en) * | 2018-06-22 | 2019-12-26 | The University Of Sydney | Expansion and differentiation of neuronal precursor cells |
| CN112384612A (en) * | 2018-06-22 | 2021-02-19 | 皮肤2神经元私人有限公司 | Expansion and differentiation of neuronal precursor cells |
| AU2019290037C1 (en) * | 2018-06-22 | 2025-08-28 | Skin2Neuron Pty Ltd | Expansion and differentiation of neuronal precursor cells |
| US11617745B2 (en) | 2018-08-17 | 2023-04-04 | Frequency Therapeutics, Inc. | Compositions and methods for generating hair cells by downregulating FOXO |
| US11162071B2 (en) | 2018-08-17 | 2021-11-02 | Frequency Therapeutics, Inc. | Compositions and methods for generating hair cells by upregulating JAG-1 |
| US20200087626A1 (en) * | 2018-09-19 | 2020-03-19 | Regeneration Biomedical, Inc. | Wnt-activated adipose-derived stem cell apparatuses, methods and systems |
| US12529034B2 (en) * | 2018-09-19 | 2026-01-20 | Regeneration Biomedical, Inc. | Wnt-activated adipose-derived stem cell apparatuses, methods and systems |
| US12077777B2 (en) | 2019-05-06 | 2024-09-03 | Accelerated Biosciences Corp. | Precursory regulatory cytotrophoblast cells and uses thereof |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230227785A1 (en) | Methods for reprogramming cells and uses thereof | |
| US11795439B2 (en) | Methods for reprogramming cells and uses thereof | |
| US12024722B2 (en) | Methods for reprogramming cells and uses thereof | |
| HK1174941B (en) | Methods for reprogramming cells and uses thereof | |
| HK1174941A (en) | Methods for reprogramming cells and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10825907 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010825907 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012535565 Country of ref document: JP Ref document number: MX/A/2012/004881 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010312240 Country of ref document: AU |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13504988 Country of ref document: US Ref document number: 2779310 Country of ref document: CA Ref document number: 1092/MUMNP/2012 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2010312240 Country of ref document: AU Date of ref document: 20101101 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20127014081 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112012009921 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112012009921 Country of ref document: BR Kind code of ref document: A2 Effective date: 20120427 |