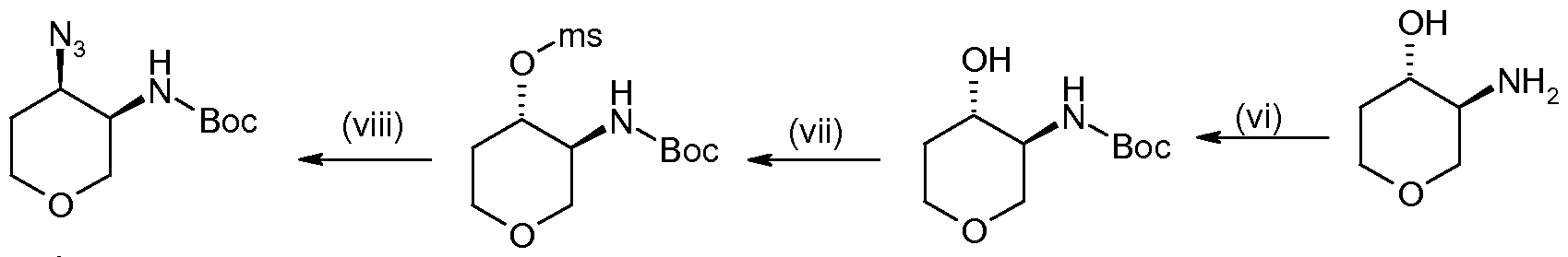WO2010097248A1 - Pyrimidinecarboxamide derivatives as inhibitors of syk kinase - Google Patents
Pyrimidinecarboxamide derivatives as inhibitors of syk kinase Download PDFInfo
- Publication number
- WO2010097248A1 WO2010097248A1 PCT/EP2010/050228 EP2010050228W WO2010097248A1 WO 2010097248 A1 WO2010097248 A1 WO 2010097248A1 EP 2010050228 W EP2010050228 W EP 2010050228W WO 2010097248 A1 WO2010097248 A1 WO 2010097248A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- formula
- salt
- pharmaceutically acceptable
- amino
- Prior art date
Links
- 0 *[C@@](COCC1)[C@@]1N Chemical compound *[C@@](COCC1)[C@@]1N 0.000 description 1
- FNYKKNODQVBHHI-WXHSDQCUSA-N C[C@H](c1ccccc1)N[C@@H](COCC1)[C@H]1O Chemical compound C[C@H](c1ccccc1)N[C@@H](COCC1)[C@H]1O FNYKKNODQVBHHI-WXHSDQCUSA-N 0.000 description 1
- DTWVVLYRSMTMDC-UHFFFAOYSA-N Cc(cc1)ccc1Nc1nc(Cl)ncc1C(N)=O Chemical compound Cc(cc1)ccc1Nc1nc(Cl)ncc1C(N)=O DTWVVLYRSMTMDC-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/02—Nasal agents, e.g. decongestants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/04—Drugs for disorders of the respiratory system for throat disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/16—Central respiratory analeptics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/04—Antipruritics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/14—Decongestants or antiallergics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present invention relates to novel chemical compounds which have activity against the spleen tyrosine kinase (Syk kinase), processes for their preparation, pharmaceutically acceptable formulations containing them and their use in therapy.
- Syk kinase spleen tyrosine kinase
- Syk kinase is a non-receptor tyrosine kinase that is involved in coupling activated immunoreceptors to signal downstream events that mediate diverse cellular responses, including proliferation, differentiation, and phagocytosis. Syk kinase is widely expressed in hematopoietic cells. Syk kinase inhibitors have potential antiinflammatory and immunomodulating activities. They inhibit Syk kinase-mediated IgG Fc epsilon and gamma receptor and BCR receptor signaling, resulting in inhibition of the activation of mast cells, macrophages, and B-cells and related inflammatory responses and tissue damage. Accordingly, Syk kinase inhibitors have attracted interest in a number of therapeutic areas, including the treatment of rheumatoid arthritis, B-cell lymphoma and asthma / rhinitis.
- RA Rheumatoid Arthritis
- Syk inhibitors may also be useful in cancer therapy, specifically heme malignancies, particularly Non-Hodgkin's Lymphomas including follicular (FL), mantle cell, Burkitt and diffuse large B cell (DLBCL) lymphomas.
- FL follicular
- DLBCL diffuse large B cell
- Syk is dysregulated by overexpression and / or constitutively activation in a variety of primary B-lymphoma tumors and also in B- lymphoma cell lines.
- Syk through the PI3K / AKT pathway, the PLD pathway and AKT independent signalling, activates mTOR (mammalian target of rapamycin) which in turn increases B-cell survival and proliferation.
- mTOR mimmalian target of rapamycin
- Inhibition of Syk results in decreased mTOR activation and a reduction of clonicity in FL cells.
- Inhibition of Syk kinase with curcumin in a murine model of B lymphoma (BKS-2) gave a significant reduction of tumour burden as measured by the total splenocyte number.
- Syk inhibitors may also be useful in the treatment of asthma and rhinitis as they are important in transducing the downstream cellular signals associated with cross- linking Fc ⁇ R1 and or Fc ⁇ R1 receptors, and is positioned early in the signalling cascade.
- the early sequence of Fc ⁇ R1 signalling following allergen cross-linking of receptor-lgE complexes involves first Lyn (a Src family tyrosine kinase) and then Syk kinase.
- Allergic rhinitis and asthma are diseases associated with hypersensitivity reactions and inflammatory events involving a multitude of cell types including mast cells, eosinophils, T cells and dendritic cells.
- high affinity immunoglobulin receptors for IgE (Fc ⁇ RI) and IgG (Fc ⁇ RI) become cross-linked and activate downstream processes in mast cells and other cell types leading to the release of pro-inflammatory mediators and airway spasmogens.
- IgE receptor cross-linking by allergen leads to release of mediators including histamine from pre-formed granules, as well as the synthesis and release of newly synthesised lipid mediators including prostaglandins and leukotrienes.
- the Syk kinase inhibitor R112 (Rigel), dosed intranasally in a phase l/ll study for the treatment of allergic rhinitis, was shown to give a statistically significant decrease in PGD 2 , a key immune mediator that is highly correlated with improvements in allergic rhinorrhea, as well as being safe across a range of indicators, thus providing the first evidence for the clinical safety and efficacy of a topical Syk kinase inhibitor (see Meltzer, EIi O.; Berkowitz, Robert B.; Grossbard, Elliott B. An intranasal Syk kinase inhibitor (R1 12) improves the symptoms of seasonal allergic rhinitis in a park environment.
- EP1 184376B1 / WO200007513 and EP1054004 / WO9903101073 describe novel heterocyclic carboxamide derivatives that have Syk inhibitory activity. These are further described in "Synthetic studies on novel Syk Inhibitors. Part 1 : Synthesis and structure-activity relationships of 5- pyrimidine-5-carboaxamidr derivatives (H. Hisamichi et al, Bioorg Med Chem 13 (2005) 4936 - 4951 ). In particular, it would appear from this paper that the preferred compound is the compound of formula (A):
- WO9903101073 describes a wider range of analogues, including a set in which the ethylene diamine moiety is replaced by c/s-1 ,2-diaminocyclohexyl.
- WO 04/035604 discloses the structural co-ordinates of the human Syk protein.
- the present invention provides a compound of formula (I):
- the compound of formula (I) has the chemical name:
- Compounds of the present invention are useful as inhibitors of Syk.
- Compounds of the present invention also exhibit selectivity for the Syk kinase against other key kinases, for instance at least 1Ox (based on either pKi or plC 5 o values for the enzymes), in particular the kinases VEGFR2 and Aurora B.
- Compounds of the present invention also exhibit low activity in the hERG assay, a key measure of potential cardiac toxicity.
- Compounds of the present invention are thus potentially of use in treating some cancer therapies, in particular heme malignancies, as well as inflammatory conditions which involve B cells and/or activated macrophages, and also diseases resulting from inappropriate mast cell activation, for instance allergic and inflammatory diseases.
- the term “pharmaceutically acceptable” refers to those compounds, materials, compositions, and dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- pharmaceutically acceptable salts of the compound of the present invention may be prepared.
- pharmaceutically acceptable salts refers to salts that retain the desired biological activity of the subject compound and exhibit minimal undesired toxicological effects.
- compositions may be prepared in situ during the final isolation and purification of the compound, or by separately reacting the purified compound in its free acid or free base form with a suitable base or acid, respectively. Indeed, in certain embodiments of the invention, pharmaceutically acceptable salts may be preferred over the respective free base or free acid because such salts impart greater stability or solubility to the molecule thereby facilitating formulation into a dosage form.
- the compound of formula (I) is basic and accordingly generally capable of forming pharmaceutically acceptable acid addition salts by treatment with a suitable acid.
- suitable acids include pharmaceutically acceptable inorganic acids and pharmaceutically acceptable organic acids.
- Representative pharmaceutically acceptable acid addition salts include hydrochloride, hydrobromide, nitrate, methylnitrate, sulfate, bisulfate, sulfamate, phosphate A acetate, hydroxyacetate, phenylacetate, propionate, butyrate, isobutyrate, valerate, maleate, hydroxymaleate, acrylate, fumarate, malate, tartrate, citrate, salicylate, p-aminosalicyclate, glycollate, lactate, heptanoate, phthalate, oxalate, succinate, benzoate, o-acetoxybenzoate, chlorobenzoate, methylbenzoate, dinitrobenzoate, hydroxybenzoate, methoxybenzoate, mandelate, tanna
- a compound of the present invention may exist in solid or liquid form.
- the compound of the present invention may exist in crystalline or noncrystalline (amorphous) form, or as a mixture thereof.
- a compound of the present invention that is in crystalline form the skilled artisan will appreciate that pharmaceutically acceptable solvates may be formed wherein solvent molecules are incorporated into the crystalline lattice during crystallization.
- Solvates may involve non-aqueous solvents such as, but not limited to, ethanol, isopropanol, n-butanol, i- butanol, acetone, tetrahydrofuran, dioxane, DMSO, acetic acid, ethanolamine, and ethyl acetate, or they may involve water as the solvent that is incorporated into the crystalline lattice. Solvates wherein water is the solvent incorporated into the crystalline lattice are typically referred to as "hydrates.” Hydrates include stoichiometric hydrates as well as compositions containing variable amounts of water. The invention includes all such solvates.
- a compound of the present invention that exists in crystalline form may exhibit polymorphism (i.e. the capacity to occur in different crystalline structures). These different crystalline forms are typically known as “polymorphs.”
- the invention includes all such polymorphs. Polymorphs have the same chemical composition but differ in packing, geometrical arrangement, and other descriptive properties of the crystalline solid state. Polymorphs, therefore, may have different physical properties such as shape, density, hardness, deformability, stability, and dissolution properties. Polymorphs typically exhibit different melting points, IR spectra, and X-ray powder diffraction patterns, which may be used for identification.
- polymorphs may be produced, for example, by changing or adjusting the reaction conditions or reagents, used in making the compound. For example, changes in temperature, pressure, or solvent may result in polymorphs. In addition, one polymorph may spontaneously convert to another polymorph under certain conditions.
- the compound of formula (I), thereof, may be prepared by the general synthetic scheme described hereinafter.
- the present invention provides a process for preparing a compound of formula (I) which process comprises treating a compound of formula (II):
- P is a protecting group eg t-butoxycarbonyl (Boc), and thereafter, removing the protecting group.
- X is N 3 or NH 2 and Y is a protecting group, for instance t-butoxycarbonyl (Boc), and which has the (3R,4R) stereochemistry; are novel and of use in the preparation of compounds of formula (I) and therefore provide a further aspect of the invention.
- Y is a protecting group, for instance t-butoxycarbonyl (Boc), and which has the (3R,4R) stereochemistry; are novel and of use in the preparation of compounds of formula (I) and therefore provide a further aspect of the invention.
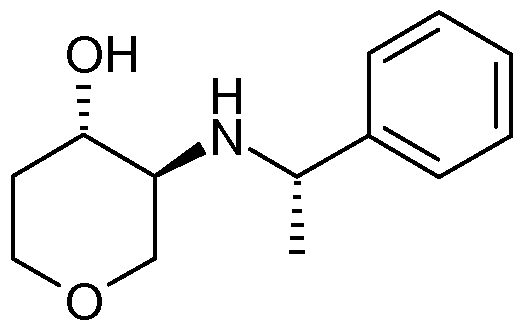
- (V) at C-3 by reaction with a chiral amine precursor, such as [(1S)-1-phenylethyl]amine, in a C 2 - 4 alcohol, preferably a secondary alcohol, such as 2-propanol or 2-butanol, at an elevated temperature, preferably under reflux conditions.
- the reaction may also be carried out in the presence of trimethylaluminium, in a solvent such as dichloromethane, followed by work-up with sodium fluoride, to decompose the aluminate.
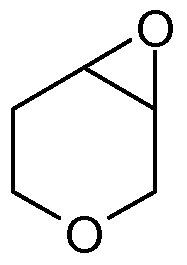
- the initial reaction product is potentially a mixture of two C-3 diastereoisomers and two C-4 diastereoisomers, the C-3 : C-4 ratio depending on the regiospecificity of the epoxide ring opening.
- the C-3 regioisomer mixture may then be separated out and the chiral moiety removed, to give the desired 3-amino, 4- hydroxy tetrahydropyran intermediate of formula (Vl):
- the present invention provides for the preparation of a compound of formula (IV) or (IV), which processes comprises the step of reacting the compound of formula (V) with with a chiral amine precursor, such as [(1 S)-I- phenylethyl]amine, in a C 2-4 alcohol, preferably a secondary alcohol, such as 2- propanol or 2-butanol, at an elevated temperature, preferably under reflux conditions.
- a chiral amine precursor such as [(1 S)-I- phenylethyl]amine
- Suitable amine protecting groups include, but are not restricted to, sulphonyl (such as tosyl), acyl (such as benzyloxycarbonyl or t-butoxycarbonyl) and arylalkyl (such as benzyl), which may be removed by hydrolysis or hydrogenolysis as appropriate.
- Suitable amine protecting groups include trifluoroacetyl (-C(O)CF 3 ), which may be removed by base catalysed hydrolysis, or a solid phase resin bound benzyl group, such as a Merrifield resin bound 2,6-dimethoxybenzyl group (Ellman linker) which may be removed by acid catalysed hydrolysis (using, for example, trifluoroacetic acid).
- a solid phase resin bound benzyl group such as a Merrifield resin bound 2,6-dimethoxybenzyl group (Ellman linker) which may be removed by acid catalysed hydrolysis (using, for example, trifluoroacetic acid).
- Compounds of the present invention are useful as inhibitors of Syk and thus potentially of use in treating some cancer therapies, in particular heme malignancies, as well as inflammatory conditions which involve B cells, and also diseases resulting from inappropriate mast cell activation, for instance allergic and inflammatory diseases.
- the present invention provides for a compound of formula (I), or a pharmaceutically acceptable salt thereof, for use in therapy.
- the present invention provides a method comprising administering to a patient in need thereof an effective amount of a compound of formula (I), or a pharmaceutically acceptable salt thereof, to inhibit a Syk kinase.
- Syk inhibitors may be useful in cancer therapy, specifically heme malignancies, particularly Non-Hodgkin's Lymphomas including follicular (FL), mantle cell, small lymphocytic lymphoma/chronic lymphocytic lymphoma (SLL/CLL), Burkitt and diffuse large B cell (DLBCL) lymphomas.
- FL follicular
- SLL/CLL small lymphocytic lymphoma/chronic lymphocytic lymphoma
- DLBCL diffuse large B cell lymphomas
- the present invention provides for a method of treating cancer, specifically heme malignancies, particularly Non-Hodgkin's Lymphomas including follicular (FL), mantle cell, Burkitt and diffuse large B cell (DLBCL) lymphomas, which method comprises administering to a patient in need thereof an effective amount of a compound of formula (I), or a pharmaceutically acceptable salt thereof.
- heme malignancies particularly Non-Hodgkin's Lymphomas including follicular (FL), mantle cell, Burkitt and diffuse large B cell (DLBCL) lymphomas
- Compounds of the present invention may also be used in cancer chemotherapy in combination with other classes of cancer chemotherapy agents which are known in the art.
- Representative classes of agents for use in such combinations for Non- Hodgkin's Lymphomas include ritaximab, BEXXAR (tositumomab and Iodine I 131 tositumomab), pixantrone and chemotherapy.
- Combination of compounds of the present invention may also be used in combination with the CHOP drug regime (Cyclophosphamide, Adriamycin, Vincristine, Prednisone) or CHOP plus ritaximab (CHOP+R).
- Compounds of the present invention are potentially of use in treating auto immune conditions which involve B cells and/or macrophage activation, for instance systemic lupus erythematosus, Sjorgens Syndrome, Wegners granulomatosis, Bullous Pemphigoid, Idiopathic Thrombocytopenic Purpura (ITP), Giant Cell Arteriosis, Chronic Idiopathic Urticaria with and without auto-antibody status (Chronic Autoimmune Urticaria) (New concepts in chronic urticaria Current Opinions in Immunology 2008 20:709-716), Glomerulonephritis, Chronic Transplant Rejection, and rheumatoid arthritis.
- the present invention provides a method of treating an inflammatory disease which involves B cells which method comprises administering to a patient in need thereof an effective amount of a compound of formula (I), or a pharmaceutically acceptable salt thereof.
- Compounds of the present invention are potentially of use in treating diseases resulting from inappropriate mast cell activation, for instance allergic and inflammatory diseases.
- the present invention provides for a method of treating inappropriate mast cell activation which method comprises administering to a patient in need thereof an effective amount of a compound of formula I, or a pharmaceutically acceptable salt thereof.
- the present invention provides a method of treating an inflammatory disease which method comprises administering to a patient in need thereof an effective amount of a compound of formula (I), or a pharmaceutically acceptable salt thereof.
- the present invention provides a method of treating an allergic disorder which comprises administering to a patient in need thereof an effective amount of a compound of formula (I), or a pharmaceutically acceptable salt thereof.
- Syk kinase diseases and pathological conditions thought to be mediated by Syk kinase include inflammatory and allergic disorders involving mast cell activation, such as chronic obstructive pulmonary disease (COPD), adult respiratory distress syndrome (ARDS), asthma, ulcerative colitis, Crohn's Disease, bronchitis, conjunctivitis, psoriasis, sclerodoma, urticaria, dermatitis, and allergic rhinitis.
- COPD chronic obstructive pulmonary disease
- ARDS adult respiratory distress syndrome
- asthma ulcerative colitis
- Crohn's Disease bronchitis
- conjunctivitis conjunctivitis
- psoriasis psoriasis
- sclerodoma urticaria
- dermatitis dermatitis
- allergic rhinitis allergic rhinitis
- Compounds of the present invention may also be used in combination with other classes of therapeutic agents which are known in the art.
- Representative classes of agents for use in such combinations include, for treating asthma, anti-inflammatory steroids (in particular corticosteroids), PDE4 inhibitors, IKK2 inhibitors, A2a agonists, ⁇ 2 -adrenoreceptor agonists (including both short acting and long acting ⁇ 2 -adrenoreceptor agonists), alpha 4 integrin inhibitors, and anti-muscarinics, and, for treating allergies, the foregoing agents, as well as histamine receptor antagonists, including H1 and H1/H3 antagonists.
- Representative agents for use in combination therapy for treating severe asthma include topically acting p38 inhibitors, and IKK2 inhibitors.
- Anti-inflammatory corticosteroids are well known in the art. Representative examples include fluticasone propionate (e.g. see US patent 4,335,121 ), beclomethasone 17- propionate ester, beclomethasone 17,21-dipropionate ester, dexamethasone or an ester thereof, mometasone or an ester thereof (e.g. mometasone furoate), ciclesonide, budesonide, and flunisolide.
- fluticasone propionate e.g. see US patent 4,335,121
- beclomethasone 17- propionate ester beclomethasone 17,21-dipropionate ester
- dexamethasone or an ester thereof dexamethasone or an ester thereof
- mometasone or an ester thereof e.g. mometasone furoate
- ciclesonide e.g. mometasone furoate
- ciclesonide eson
- anti-inflammatory corticosteroids are described in WO 02/12266 A1 (Glaxo Group Ltd), in particular, the compounds of Example 1 ( 6 ⁇ ,9 ⁇ -difluoro-17 ⁇ -[(2-furanylcarbonyl)oxy]-11 ⁇ -hydroxy- 16 ⁇ -methyl-3-oxo-androsta-1 ,4-diene-17 ⁇ -carbothioic acid S-fluoromethyl ester) and Example 41 (6 ⁇ ,9 ⁇ -difluoro-11 ⁇ -hydroxy-16 ⁇ -methyl-17 ⁇ -[(4-methyl-1 ,3-thiazole-5- carbonyl)oxy]-3-oxo-androsta-1 ,4-diene-17 ⁇ -carbothioic acid S-fluoromethyl ester), or a pharmaceutically acceptable salt thereof.
- ⁇ 2 -adrenoreceptor agonists examples include salmeterol (e.g. as racemate or a single enantiomer such as the R-enantiomer), salbutamol, formoterol, salmefamol, fenoterol or terbutaline and salts thereof, for example the xinafoate salt of salmeterol, the sulphate salt or free base of salbutamol or the fumarate salt of formoterol.
- Long- acting ⁇ 2 -adrenoreceptor agonists are preferred, especially those having a therapeutic effect over a 24 hour period such as salmeterol or formoterol.
- anti-histamines examples include methapyrilene, or loratadine, cetirizine, desloratadine or fexofenadine.
- anticholinergic compounds include muscarinic (M) receptor antagonists, in particular M-
- muscarinic M3 antagonists include ipratropium bromide, oxitropium bromide or tiotropium bromide.
- Representative PDE4 or mixed PDE3/4 inhibitors that may be used in combination with compounds of the invention include AWD-12-281 (Elbion), PD-168787 (Pfizer), roflumilast, and cilomilast (GlaxoSmithKline).
- PDE4 inhibitors are described in WO 2004/103998, WO2005/030212, WO2005/030725, WO2005/058892, WO2005/090348, WO2005/090352, WO2005/090353, WO2005/090354, WO2006/053784, WO2006/097340, WO2006/133942, WO2007/036733, WO2007/036734 and WO2007/045861 (Glaxo Group Ltd).
- the present invention also provides for so-called "triple combination” therapy, comprising a compound of formula (I) or a pharmaceutically acceptable salt thereof together with ⁇ 2 -adrenoreceptor agonist and an anti-inflammatory corticosteroid.
- this combination is for treatment and/or prophylaxis of asthma, COPD or allergic rhinitis.
- the ⁇ 2 -adrenoreceptor agonist and/or the anti-inflammatory corticosteroid can be as described above and/or as described in WO 03/030939 A1.
- a representative example of such a "triple" combination comprises a compound of formula (I) or a pharmaceutically acceptable salt thereof, salmeterol or a pharmaceutically acceptable salt thereof (e.g. salmeterol xinafoate) and fluticasone propionate.
- the compound of the present invention will normally, but not necessarily, be formulated into pharmaceutical compositions prior to administration to a patient. Accordingly, in another aspect the invention is directed to pharmaceutical compositions comprising a compound of the invention and one or more pharmaceutically acceptable excipient.
- compositions of the invention may be prepared and packaged in bulk form wherein a safe and effective amount of a compound of the invention can be extracted and then given to the patient, such as with powders or syrups.
- the pharmaceutical compositions of the invention may be prepared and packaged in unit dosage form wherein each physically discrete unit contains a safe and effective amount of a compound of the invention.
- the pharmaceutical compositions of the invention may also be prepared and packaged in a sub-unit dosage form wherein two or more sub-unit dosage forms provide the unit dosage form.
- the pharmaceutical compositions of the invention typically contain from about 0.1 to 99.9 wt.%, of the compound of the invention, depending on the nature of the formulation.
- compositions of the invention may optionally further comprise one or more additional pharmaceutically active compounds.
- pharmaceutically acceptable excipient means a pharmaceutically acceptable material, composition or vehicle involved in giving form or consistency to the pharmaceutical composition.
- Each excipient must be compatible with the other ingredients of the pharmaceutical composition when commingled, such that interactions which would substantially reduce the efficacy of the compound of the invention when administered to a patient and would result in pharmaceutically unacceptable compositions are avoided.
- each excipient must of course be of sufficiently high purity to render it pharmaceutically acceptable.
- compositions of the present invention comprising a compound of the invention and the pharmaceutically acceptable excipient or excipients will typically be provided as a dosage form adapted for administration to the patient by the desired route of administration.
- dosage forms include those adapted for (1 ) inhalation, such as aerosols and solutions; (2) intranasal administration, such as solutions or sprays; (3) oral administration, such as tablets, capsules, caplets, pills, troches, powders, syrups, elixers, suspensions, solutions, emulsions, sachets, and cachets; and (4) parenteral administration, such as sterile solutions, suspensions, and powders for reconstitution.
- dosage forms adapted for inhalation or oral administration are commonly used for treating COPD; dosage forms adapted for intranasal administration are commonly used for treating allergic rhinitis; and dosage forms adapted for oral administration are commonly used for treating rheumatoid arthritis and heme malignancies.
- Suitable pharmaceutically acceptable excipients will vary depending upon the particular dosage form chosen.
- suitable pharmaceutically acceptable excipients may be chosen for a particular function that they may serve in the composition.
- certain pharmaceutically acceptable excipients may be chosen for their ability to facilitate the production of uniform dosage forms.
- Certain pharmaceutically acceptable excipients may be chosen for their ability to facilitate the production of stable dosage forms.
- Certain pharmaceutically acceptable excipients may be chosen for their ability to facilitate the carrying or transporting the compound of the present invention once administered to the patient from one organ, or portion of the body, to another organ, or portion of the body.
- Certain pharmaceutically acceptable excipients may be chosen for their ability to enhance patient compliance.
- Suitable pharmaceutically acceptable excipients include the following types of excipients: Diluents, fillers, binders, disintegrants, lubricants, glidants, granulating agents, coating agents, wetting agents, solvents, co-solvents, suspending agents, emulsifiers, sweetners, flavoring agents, flavor masking agents, coloring agents, anticaking agents, humectants, chelating agents, plasticizers, viscosity increasing agents, antioxidants, preservatives, stabilizers, surfactants, and buffering agents.
- excipients may serve more than one function and may serve alternative functions depending on how much of the excipient is present in the formulation and what other ingredients are present in the formulation.
- compositions of the invention are prepared using techniques and methods known to those skilled in the art. Some of the methods commonly used in the art are described in Remington's Pharmaceutical Sciences (Mack Publishing Company).
- Oral solid dosage forms such as tablets will typically comprise one or more pharmaceutically acceptable excipients, which may for example help impart satisfactory processing and compression characteristics, or provide additional desirable physical characteristics to the tablet.
- pharmaceutically acceptable excipients may be selected from diluents, binders, glidants, lubricants, disintegrants, colorants, flavorants, sweetening agents, polymers, waxes or other solubility- modulating materials.
- Dosage forms for parenteral administration will generally comprise fluids, particularly intravenous fluids, i.e., sterile solutions of simple chemicals such as sugars, amino acids or electrolytes, which can be easily carried by the circulatory system and assimilated.
- fluids are typically prepared with water for injection USP.
- Fluids used commonly for intravenous (IV) use are disclosed in Remington, The Science and Practice of Pharmacy [full citation previously provided].
- the pH of such IV fluids may vary, and will typically be from 3.5 to 8 as known in the art.
- Dosage forms for nasal or inhaled administration may conveniently be formulated as aerosols, solutions, drops, gels or dry powders.
- Dosage forms for topical administration to the nasal cavity include pressurised aerosol formulations and aqueous formulations administered to the nose by pressurised pump.
- Formulations which are non-pressurised and adapted for nasal administration are of particular interest. Suitable formulations contain water as the diluent or carrier for this purpose.
- Aqueous formulations for administration to the nose may be provided with conventional excipients such as buffering agents, tonicity modifying agents and the like. Aqueous formulations may also be administered to the nose by nebulisation.
- dosage forms for nasal administration are provided in a metered dose device.
- the dosage form may be provided as a fluid formulation for delivery from a fluid dispenser having a dispensing nozzle or dispensing orifice through which a metered dose of the fluid formulation is dispensed upon the application of a user-applied force to a pump mechanism of the fluid dispenser.
- Such fluid dispensers are generally provided with a reservoir of multiple metered doses of the fluid formulation, the doses being dispensable upon sequential pump actuations.
- the dispensing nozzle or orifice may be configured for insertion into the nostrils of the user for spray dispensing of the fluid formulation into the nasal cavity.
- the fluid dispenser is of the general type described and illustrated in WO-A-2005/044354.
- the dispenser has a housing which houses a fluid discharge device having a compression pump mounted on a container for containing a fluid formulation.
- the housing has at least one finger-operable side lever which is movable inwardly with respect to the housing to cam the container upwardly in the housing to cause the pump to compress and pump a metered dose of the formulation out of a pump stem through a nasal nozzle of the housing.
- a particularly preferred fluid dispenser is of the general type illustrated in Figures 30-40 of WO-A- 2005/044354.
- the compound or salt of formula (I) is in a particle-size-reduced form, and more preferably the size-reduced form is obtained or obtainable by micronisation.
- the preferable particle size of the size-reduced (e.g. micronised) compound or salt or solvate is defined by a D50 value of about 0.5 to about 10 microns (for example as measured using laser diffraction).
- Aerosol compositions can comprise a solution or fine suspension of the active substance in a pharmaceutically acceptable aqueous or non-aqueous solvent. Aerosol formulations can be presented in single or multidose quantities in sterile form in a sealed container, which can take the form of a cartridge or refill for use with an atomising device or inhaler. Alternatively the sealed container may be a unitary dispensing device such as a single dose nasal inhaler or an aerosol dispenser fitted with a metering valve (metered dose inhaler) which is intended for disposal once the contents of the container have been exhausted.
- a metering valve metered dose inhaler
- the dosage form comprises an aerosol dispenser
- it preferably contains a suitable propellant under pressure such as compressed air, carbon dioxide or an organic propellant such as a hydrofluorocarbon (HFC).
- suitable HFC propellants include 1 ,1 ,1 ,2,3,3,3-heptafluoropropane and 1 ,1 ,1 ,2-tetrafluoroethane.
- the aerosol dosage forms can also take the form of a pump-atomiser.
- the pressurised aerosol may contain a solution or a suspension of the active compound. This may require the incorporation of additional excipients e.g. co-solvents and/or surfactants to improve the dispersion characteristics and homogeneity of suspension formulations. Solution formulations may also require the addition of co-solvents such as ethanol.
- Other excipient modifiers may also be incorporated to improve, for example, the stability and/or taste and/or fine particle mass characteristics (amount and/or profile) of the formulation.
- the pharmaceutical composition is a dry powder inhalable composition.
- a dry powder inhalable composition can comprise a powder base such as lactose, glucose, trehalose, mannitol or starch, the compound of formula (I) or salt or solvate thereof (preferably in particle-size-reduced form, e.g. in micronised form), and optionally a performance modifier such as L-leucine or another amino acid, cellobiose octaacetate and/or metals salts of stearic acid such as magnesium or calcium stearate.
- the dry powder inhalable composition comprises a dry powder blend of lactose and the compound of formula (I) or salt thereof.
- the lactose is preferably lactose hydrate e.g. lactose monohydrate and/or is preferably inhalation- grade and/or fine-grade lactose.
- the particle size of the lactose is defined by 90% or more (by weight or by volume) of the lactose particles being less than 1000 microns (micrometres) (e.g. 10-1000 microns e.g. 30-1000 microns) in diameter, and/or 50% or more of the lactose particles being less than 500 microns (e.g. 10-500 microns) in diameter. More preferably, the particle size of the lactose is defined by 90% or more of the lactose particles being less than 300 microns (e.g.
- the particle size of the lactose is defined by 90% or more of the lactose particles being less than 100-200 microns in diameter, and/or 50% or more of the lactose particles being less than 40- 70 microns in diameter.
- a suitable inhalation-grade lactose is E9334 lactose (10% fines) (Borculo Domo Ingredients, Hanzeplein 25, 8017 JD Zwolle, Netherlands).
- a pharmaceutical composition for inhaled administration can be incorporated into a plurality of sealed dose containers (e.g. containing the dry powder composition) mounted longitudinally in a strip or ribbon inside a suitable inhalation device.
- the container is rupturable or peel-openable on demand and the dose of e.g. the dry powder composition can be administered by inhalation via the device such as the DISKUS TM device, marketed by GlaxoSmithKline.
- the DISKUS TM inhalation device is for example described in GB 2242134 A, and in such a device at least one container for the pharmaceutical composition in powder form (the container or containers preferably being a plurality of sealed dose containers mounted longitudinally in a strip or ribbon) is defined between two members peelably secured to one another; the device comprises: a means of defining an opening station for the said container or containers; a means for peeling the members apart at the opening station to open the container; and an outlet, communicating with the opened container, through which a user can inhale the pharmaceutical composition in powder form from the opened container.
- a composition of the present invention, for intranasal administration may also be adapted for dosing by insufflation, as a dry powder formulation.
- the compound of the present invention when administered in combination with other therapeutic agents normally administered by the inhaled, intravenous, oral or intranasal route, that the resultant pharmaceutical composition may be administered by the same routes.
- the compound of the present invention may conveniently be administered in amounts of, for example, 1 ⁇ g to 2g.
- the precise dose will of course depend on the age and condition of the patient and the particular route of administration chosen. Biological test methods
- Recombinant human Syk was expressed as a His-tagged protein * .
- the activity of Syk was assessed using a time-resolved fluorescence resonance energy transfer (TR-FRET) assay.
- TR-FRET time-resolved fluorescence resonance energy transfer
- Syk was pre-activated at room temperature for 30mins in the presence of 16.6mM MgCI 2 , 8.3mM ATP and then diluted to 4nM in 4OmM Hepes pH 7.4, 0.01% BSA.
- 3 ⁇ l of substrate reagent containing biotinylated peptide, Biotin-AAAEEIYGEI (0.5 ⁇ M final), ATP (30 ⁇ M final) and MgCI 2 (1OmM final) in 4OmM HEPES pH 7.4, 0.01% BSA were added to wells containing 0.1 ⁇ l of various concentrations of compound or DMSO vehicle (1.7% final) in Greiner low volume 384 well black plate. The reaction was initiated by the addition of 3 ⁇ l of diluted Syk (2nM final).
- the reaction was incubated for 60min at room temperature, then terminated by the addition of 3 ⁇ l of read reagent containing 60 mM EDTA, 15OmM NaCI, 5OnM Streptavidin APC (Prozyme, San Leandro, California, USA), 0.5nM antiphosphotyrosine antibody labelled with W-1024 europium chelate (Wallac OY, Turku, Finland) in 4OmM HEPES pH 7.4, 0.03% BSA. The reaction was further incubated for 45min at room temperature.
- the degree of phosphorylation of Biotin-AAAEEIYGEI was measured using a BMG Rubystar plate reader (BMG LabTechnologies Ltd, Aylesbury, UK) as a ratio of specific 665 nm energy transfer signal to reference europium 620 nm signal.
- the compound of formula (I) has an IC 50 value in this assay of 40 nM.
- the 30OmM Imidazole fractions were pooled buffer exchanged using G25M (Amersham Biosciences, Buckinghamshire, UK) into 2OmM MES pH 6.0, 2OmM NaCI, 1OmM ⁇ McEtOH,10% glycerol.
- the buffer exchanged 6His-Syk was loaded onto a Source15S column (Amersham Biosciences, Buckinghamshire, UK) and the column eluted using a NaCI gradient 0-50OmM over 50 column volumes.
- the 6His-Syk containing fractions were pooled and concentrated by ultra-filtration. The identity of 6His-Syk was confirmed by peptide mass finger printing and intact LC-MS.
- Recombinant human Aurora B (2-344) was expressed as a Flag-6His-Thr-tagged protein * .
- the activity of Aurora B was assessed using a Fluorescence Polarisation IMAP assay (Molecular Devices, Sunnyvale, US).
- Aurora B (2 ⁇ M) was preactivated by equivalent concentration of GST-INCENP ⁇ in 3OmM Tris-HCI pH 8.0, 0.4mM ATP, 2mM MgCI 2 , 0.1 mM EGTA, 0.1% BME (beta mercaptoethanol), 0.1 mM sodium vanadate, 1 OmM DTT for 3 hours at 30 0 C. This solution was then dialysed for 5 hours against 5OmM Tris-HCI, pH 7.5, 27OmM sucrose, 15OmM NaCI, 0.1 mM EDTA, 0.1 % BME, 1 mM benzamidine and 0.2mM PMSF at 4°C. Aurora B/INCENP complex was aliquoted and frozen at -80 0 C.
- a final concentration of 2nM of Aurora B/INCENP complex was added to the assay buffer (25mM HEPES, 25mM NaCI 0.0025% Tween-20, pH 7.2 0.015% BSA, 1 ⁇ M DTT). 3 ⁇ l of this solution was added to wells containing 0.1 ⁇ l of various concentrations of compound or DMSO vehicle in Greiner low volume 384 well black plate at room temperature for 30mins.
- reaction was initiated by the presence of 3 ⁇ l of substrate reagent containing 10OnM 5FAM-PKA-tide (GRTGRRNSI-NH 2 ), 2 ⁇ M ATP and 2mM MgCI 2 in assay buffer (25mM HEPES, 25mM NaCI 0.0025% Tween- 20, pH 7.2 0.015% BSA, 1 ⁇ M DTT) with a final DMSO level of 1.7%.
- the reaction was incubated for a further 120mins at room temperature, and then terminated by the addition of 6 ⁇ l of a 1 :500 dilution Progressive Binding Reagent solution (Part: R7287) in the manufacturers buffer A (Part: R7285) and manufacturers buffer B (Part R7286) and left to incubate for 120mins at room temperature.
- the degree of phosphorylation of the 5FAM-PKA-tide (GRTGRRNSI-NH 2 ) was measured using an Acquest plate reader (Molecular Devices, Sunnyvale, US) with excitation 485nM, emission at 53OnM and using a 505nmM dichroic lens. Data was captured in parallel and perpendicular directions and converted to mp by the instrument.
- the compound of formula (I) has an activity in this assay of 20 ⁇ M.
- VEGFR2 (KDR) Enzyme Assay - Time-resolved fluorescence resonance energy transfer kinase assay
- VEGFR2 Recombinant human VEGFR2 (KDR) intracellular domain (including the entire kinase domain) was expressed as a GST-6His-tagged protein * .
- the activity of VEGFR2 was assessed using a time-resolved fluorescence resonance energy transfer (TR- FRET) assay.
- Test compounds at the desired concentrations in 100% DMSO or 100% DMSO vehicle were added in 0.1 ⁇ L to a Greiner low-volume, 384-well, black plate (#784076). The plate was centrifuged minimally at 1000 RPM for 1 min. to force all of the liquid to the bottom of the wells prior to addition of any assay reagents.
- VEGFR2 (10OnM typically) was activated at room temperature for 20min. in the presence of 10OmM HEPES, pH 7.5, 1OmM MgCI 2 , 100 ⁇ M ATP, 300 ⁇ M DTT, and 0.1 mg/ml_ BSA.
- a substrate solution containing 2OmM MgCI 2 , 100 ⁇ M ATP, 0.72 ⁇ M biotinylated peptide (Biotin-aminohexyl-EEEEYFELVAKKKK-NH 2 ) was added in 5 ⁇ L to the assay plate.
- the solution of activated VEGFR2 was diluted 100-fold in 20OmM HEPES, pH 7.5, 0.2mg/ml_ BSA, and 0.6mM DTT.
- the VEGFR2 catalyzed reaction was initiated by the addition of 5 ⁇ l_ of the diluted, activated VEGFR2.
- Final assay concentrations were 10OmM HEPES, pH 7.5, 1OmM MgCI 2 , 50 ⁇ M ATP, 0.1 mg/ml_ BSA, 300 ⁇ M DTT, 0.36 ⁇ M biotinylated peptide substrate, and 0.5nM VEGFR2 (the final assay concentration of VEGFR2 may vary depending on the specific activity of different batches of enzyme).
- the reaction was run for 90min. at room temperature and then terminated by the addition of 5 ⁇ l_ of 15OmM EDTA, pH 8.
- the background signal of the assay was established in wells where the addition of the 15OmM EDTA was instead made prior to adding substrate and enzyme solutions.
- HTRF detection solution containing 20OmM HEPES, pH 7.5, 0.1 mg/ml_ BSA, 3OnM Streptavidin Surel_ight®-APC (PerkinElmer, Boston, MA, USA), and 4nM LANCE® europium- labelled antiphosphotyrosine antibody (PerkinElmer, Boston, MA, USA) was added in 5 ⁇ L. After incubation for 10 min.
- phosphorylation of the biotinylated peptide substrate was measured as a ratio of specific 665nm energy transfer signal to reference europium 615nm signal using a Viewlux 1430 ultraHTS Microplate Imager (PerkinElmer, Turku, Finland).
- the compound of formula (I) has an IC 50 value in this assay of >7.9 ⁇ M.
- GST-6His-VEGFR2 was overexpressed with N-terminal GST and 6His tags using the baculovirus expression system in Sf9 insect cells.
- Cells (100-120 grams) were suspended in 5OmM HEPES pH 8.0, 10OmM NaCI, and 2OmM imidazole (5 ml/g cells) at room temperature. All other purification procedures were at 4C.
- Cells were lysed with a Branson 450 sonifier (70% power, 50% cycle for one min), and the cell lysate was centrifuged at 30,000 x g for 30 min.
- the column was washed with 5OmM HEPES pH 8.0, 10OmM NaCI, and 2OmM imidazole until the absorbance at 280 nm was less than 0.1 , then eluted with a 5 column volume gradient from 5OmM HEPES pH 8.0, 10OmM NaCI, 2OmM imidazole to 5OmM HEPES pH 8.0, 10OmM NaCI, 25OmM imidazole. Fractions (10-30 ml) were collected.
- Desired protein fractions were pooled and loaded (5ml/min) onto a 25ml glutathione Sepharose (GE Healthcare, Piscataway, NJ, USA) column equilibrated with 5OmM HEPES pH 7.5, 15OmM NaCI, and 2mM EDTA.
- the column was washed with 5OmM HEPES pH 7.5, 15OmM NaCI, and 2mM EDTA, and protein was eluted with a 3 column volume gradient to 5OmM HEPES pH 7.5, 15OmM NaCI, 1 mM EDTA, and 2OmM glutathione.
- Fractions are collected, and the desired protein fractions were pooled and concentrated to approximately 20ml with a Pall JumboSep concentrator with 10,000 MWCO membrane (Pall Corporation, Portsmouth, England).
- An 1800ml Superdex S200 or 23ml G25 (GE Healthcare, Piscataway, NJ, USA) column is equilibrated with 2OmM HEPES pH 7.5, 5OmM NaCI, 0.1 mM EDTA, and 1 mM DTT.
- the concentrate was loaded onto the column at 8ml/min., and the column was eluted with 2OmM HEPES pH 7.5, 5OmM NaCI, 0.1 mM EDTA, and 1 mM DTT.
- Protein fractions (approximately 20ml) were collected, and the desired fractions are pooled and concentrated with a Pall JumboSep concentrator with 10,000 MWCO membrane. Concentrated protein was stored at -80C in aliquots of desired volume for later use in the VEGFR2 enzyme activity assay. The identity of GST-6His-VEGFR2 was confirmed by intact liquid chromatography and mass spectrometry (LC/MS) and by proteolytic digestion followed by analysis of the resulting peptides by liquid chromatography and tandem mass spectrometry (LC/MS/MS).
- LC/MS liquid chromatography and mass spectrometry
- Ramos B cells human B cells of Burkitt's Lymphoma
- Ramos B cells are stimulated using anti-lgM. This results in the recruitment of SYK to the B cell receptor.
- the subsequent autophosphorylation of Syk leads to initiation of a signalling cascade resulting in B cell activation via the Erk MAP Kinase pathway.
- Erk is phosphorylated and following cell lysis is detected by an immune capture assay.
- Cells were plated at a density of 5x10 5 /well in a volume of 25 ⁇ l assay medium (RPMI containing 10% heat inactivated foetal calf serum, 1% L-glutamine and 1% Penicillin/Streptomycin) in 96 v-well polypropylene plates. 25 ⁇ l appropriately diluted compound solution was added and the plate incubated for 30min at 37°C with 5% CO 2 . Cells were stimulated with 5 ⁇ l Fab' 2 fragments of goat anti-human IgM (5 ⁇ g/ml final) for 7min at 37°C. Cells are lysed by the addition of 55 ⁇ l_ 2x RIPA lysis buffer for 2h at 4°C.
- RPMI containing 10% heat inactivated foetal calf serum, 1% L-glutamine and 1% Penicillin/Streptomycin
- 50 ⁇ l cell lysate was transferred to a 96 well MSD plate coated with anti-pErk1/2 (Thr/Thy: 202/204; 185/187) capture antibody and incubated for 16 hours at 4°C.
- the plate was washed and an anti-pErk detection antibody added (25 ⁇ l/well) for 2h at room temperature. This was removed, 150 ⁇ l_ MSD read buffer added and the resultant electrochemiluminescence signal measured.
- the compound of formula (I) has an IC 50 value in this assay of 50 nM.
- Compound was prepared as a 1OmM stock in DMSO and a dilution series prepared in DMSO using 9 successive 5-fold dilutions. This dilution series was diluted a further 1 :100 with assay medium to give the concentration range to be tested of 5x10 "5 to 2.56x10 "11 M. Compound dilutions were prepared using the Biomek 2000 and Biomek Nx automated robotic pipetting systems.
- Peripheral blood B cells are stimulated ex-vivo using anti-lgM. This results in the recruitment of Syk to the B cell receptor. The subsequent autophosphorylation of Syk leads to initiation of a signalling cascade resulting in B cell activation as indicated by expression of the activation marker CD69 on the cell surface.
- CD20/CD69+ve whole blood B cells are detected by flow cytometry.
- Peripheral blood B cells were prepared from heparinised human blood by density gradient centrifugation. Cells were plated at a density of 1x10 5 /well in a volume of 25 ⁇ l assay medium (RPMI containing 10% heat inactivated foetal calf serum, 1% L- glutamine and 1% Penicillin/Streptomycin) in 96 v-well polypropylene plates. 25 ⁇ l appropriately diluted compound solution was added and the plate incubated for 30min at 37°C with 5% CO 2 . Cells were stimulated with 5 ⁇ l Fab' 2 fragments of goat anti-human IgM (5 ⁇ g/ml final) for a further 3.5h under the conditions previously described. Any red blood cells present were lysed, and all other cells fixed, by the addition of 200 ⁇ l Lyse/Fix buffer for 10min at room temperature.
- RPMI containing 10% heat inactivated foetal calf serum, 1% L- glutamine and 1% Penicillin/Streptomycin
- CD69 assay The cells were stained using a cocktail of mouse anti-human CD20 FITC and mouse anti-human CD69 APC conjugated antibodies. CD20/CD69+ve B cells present in the sample were detected by flow cytometry.
- Compound Preparation Compound was prepared as a 1OmM stock in DMSO and a dilution series prepared in DMSO using 9 successive 5-fold dilutions. This dilution series was diluted a further 1 :100 with assay medium to give the concentration range to be tested of 5x10 "5 to 2.56x10 "11 M. Compound dilutions were prepared using the Biomek 2000 and Biomek Nx automated robotic pipetting systems.
- heparinised human blood 100 ⁇ l heparinised human blood was added to a 5ml polypropylene tube containing 1 ⁇ l appropriately diluted compound solution and incubated for 30min at 37°C with 5% CO 2 .
- B cells were stimulated with 10 ⁇ l Fab' 2 fragments of goat anti-human IgM (67.5 ⁇ g/ml final) for a further 3.5h under the conditions previously described.
- the red blood cells were lysed and all other cells fixed by the addition of 2ml Lyse/Fix buffer for 10min at room temperature.
- the cells were stained using a cocktail of mouse anti-human CD20 FITC and mouse anti-human CD69 APC conjugated antibodies. CD20/CD69+ve B cells present in the sample were detected by flow cytometry.
- Compound Preparation Compound was prepared as a 1OmM stock in DMSO and a dilution series prepared in DMSO using 7 successive 3-fold dilutions to give the concentration range to be tested of 1x10 "5 to 4.5x10 "10 M. Compound dilutions were prepared using the Biomek 2000 automated robotic pipetting system.
- LAD2 is a stem cell factor (SCF)-dependent human mast cell line that was established by the NIH from bone marrow aspirates from a patient with mast cell sarcoma/leukaemia.
- SCF stem cell factor
- LAD2 cells resemble CD34+-derived human mast cells and express functional Fc ⁇ RI.
- the Fc ⁇ RI is up-regulated in the presence of IL-4, SCF and IgE, subsequent cross linking of cell-bound IgE results in degranulation which can be measured as hexosaminidase release.
- LAD2 cells are re-suspended at 1x10 5 /ml in complete stem pro-34SFM (Gibco Cat 10640-019 media containing Stem Pro-34 nutrient supplement (1 :40), glutamine (2mM), penicillin (100 ⁇ g/ml), streptomycin (100 ⁇ g/ml)) with additional supplements of human recombinant SCF (100ng/ml; R&D systems), human recombinant Interleukin- 4 (6ng/ml; R&D Systems) and IgE (100 ⁇ g/ml; Calbiochem). Cells are then maintained for 5 days at 37°C, 5% CO2 in a humidified atmosphere.
- LAD2 cells Activation of LAD2 cells with anti-lqE Primed LAD2 cells are centrifuged (40Og, 5min), the supernatant discarded and the cell pellet re-suspended at 1x10 4 cells/ml in RPMI supplemented with glutamine (2mM). Following a further centrifugation (40Og, 5min) the cells are re-suspended in fresh RPMI with glutamine (2mM), adjusted to a density of 5.7 x10 5 /ml, and pipetted into sterile V-well plates (70 ⁇ l/well; Greiner) containing 20 ⁇ l diluted compound (prepared as detailed above).
- Cells are then incubated for 1 h (37°C, 5% CO 2 in a humidified atmosphere) before activating with a sub-maximal concentration of anti- IgE (1 O ⁇ l volume to give a final assay dilution of 1 :2700; Sigma).
- plates are centrifuged (120Og, 10min, 4°C) and the supernatant removed for hexosaminidase assay.
- the cell pellet is lysed in 100 ⁇ l/well triton-X (0.5% in RPMI 2mM glutamine) at 37°C for 30min.
- Beta-hexosaminidase assay Beta-hexosaminidase activity is measured by the conversion of 4-methylumbelliferyl N-acetyl- ⁇ -D glucosaminide (Sigma) to a fluorescent product.
- Compound potencies were determined by a fluoro-ligand (Cy3b-Dofetilide) fluorescence polarisation assay.
- hERG-expressing CHO-K1 membranes * (60 ⁇ g/ml) were incubated with 1.OnM fluoro- ligand ⁇ , in assay buffer (25mM HEPES, 1.2mM MgCI 2 , 10OmM KCI and 0.1 % pluronic, pH adjusted to 7.4 using 5M KOH). The final potassium concentration in the assay was 10OmM. After 70min mixing at room temperature, in the dark, 10 ⁇ l was dispensed into each well of a black LV Greiner 384-well plate containing 0.1 ⁇ l of test compound in DMSO. The plates were left to equilibrate for 2h before reading on an AcquestTM / AnalystTM imager.
- plC 5 o data were generated using from an 11 -point inhibition curve (top assay concentration of 50 ⁇ M and a 1 :3 step-dilution), a six parameter curve-fit being applied using ABase and XC50 to analyse data and generate curve fits.
- the compound of formula (I) has an IC 50 value in this assay of 25 ⁇ M.
- Chinese Hamster Ovary (CHO) cells stably expressing the human hERG receptor were grown to 80% confluency before being harvested by trypsinisation and subsequent centrifugation at 50Og for 10min.
- Cell pellets were frozen at -80C before membrane production. The frozen pellet was thawed on ice, re-suspended and homogenised in 10 volumes of membrane buffer (5OmM HEPES, pH 7.4, 1 mM EDTA, 1 mM PMSF, 2x10-6M Pepstatin A). The membrane suspension was centrifuged for 20min at 50Og, the pellet discarded and the supernatent spun again at 48,00Og for 30min.
- membrane buffer 5OmM HEPES, pH 7.4, 1 mM EDTA, 1 mM PMSF, 2x10-6M Pepstatin A
- the major component eluted between 46% and 48%B and collected in one fraction which was evaporated to dryness and the purple solid transferred to a vial using methanol as solvent. The methanol was removed under reduced pressure and the purple solid triturated with dry ether. The solid was dried overnight at 1 mbar in a drying pistol to give the title compound (1.2mg).
- Reference example 1 is the compound:
- DBU refers to 1 ,8-diazabicyclo[5.4.0]undec-7-ene
- DCM refers to dichloromethane
- DMSO dimethylsulfoxide
- DMF refers to ⁇ /, ⁇ /-dimethylformamide
- dppf refers to 1 ,1 '-bis(diphenylphosphino)ferrocene
- Ether refers to diethyl ether
- HPLC refers to high performance liquid chromatography
- IPA refers to propan-2-ol mCPBA refers to m-chloroperbenzoic acid r.t. refers to room temperature
- TBME refers to t-butylmethylether
- THF refers to tetrahydrofuran
- LC/MS (Method A) was conducted on an Acquity UPLC BEH C18 column (50mm x 2.1 mm i.d. 1.7 ⁇ m packing diameter) at 40 degrees centigrade, eluting with 10 mM Ammonium Bicarbonate in water adjusted to pH 10 with Ammonia solution (Solvent A) and Acetonitrile (Solvent B) using the following elution gradient 0-1.5min 1 - 97% B, 1.5-1.9min 97% B, 1.9 - 2.0min 100% B at a flow rate of 1 ml/min.
- the UV detection was a summed signal from wavelength of 210nm to 350nm.
- the mass spectra were recorded on a Waters ZQ Mass Spectrometer using Alternate-scan Positive and Negative Electrospray. lonisation data was rounded to the nearest integer.
- LC/MS (Method B) was conducted on an Acquity UPLC BEH C18 column (50mm x 2.1 mm i.d. 1.7 ⁇ m packing diameter) at 40 degrees centigrade, eluting with 0.1% v/v solution of Formic Acid in Water (Solvent A) and 0.1% v/v solution of Formic Acid in Acetonitrile (Solvent B) using the following elution gradient 0-1.5min 3 - 100% B, 1.5- 1.9min 100% B, 1.9 - 2.0min 3% B at a flow rate of 1 ml/min.
- the UV detection was a summed signal from wavelength of 210nm to 350nm.
- the mass spectra were recorded on a Waters ZQ Mass Spectrometer using Alternate-scan Positive and
- Negative Electrospray. lonisation data was rounded to the nearest integer.
- Silica chromatography techniques include either automated (Flashmaster) techniques or manual chromatography on pre-packed cartridges (SPE) or manually- packed flash columns.
- SPE pre-packed cartridges
- Silica chromatography techniques include either automated (Flashmaster) techniques or manual chromatography on pre-packed cartridges (SPE) or manually- packed flash columns.
- Compounds of the present invention have the (3R,4R) absolute stereochemistry.
- Tetrahydro-4-pyranol 1005.2g
- DCM 5530ml
- triethylamine 1640ml
- Mesyl chloride 1243.8g was added to the cooled and stirred mixture in a controlled manner over ⁇ 2.5h maintaining the temperature below 15°C.
- the mesyl chloride was washed in with DCM (500ml) and the reaction allowed to warm to ambient temperature overnight.
- the mixture was treated with aqueous ammonium chloride ( ⁇ 2I, 9.8% w/w), stirred for 5min and the phases separated.
- the organic phase was washed with aqueous ammonium chloride (-2I, 9.8% w/w), water ( ⁇ 2I) and dried (sodium sulphate).
- the organic phase was concentrated in vacuo (39°C, ⁇ 15mbar) to an oil which rapidly solidified on standing (1733.9g).
- This material was treated slowly with DBU (-30OmI) at 52°C, over 30min a solution formed and this was treated with DBU (1.71) and the mixture warmed to ⁇ 100°C (external temperature) over 1 h and maintained at this temperature for 2h. The temperature was raised slowly to 148°C (external) and the distilling 3,6-dihydro-2H-pyran collected (527.5g).
- the mixture was filtered, the residue washed with TBME (660ml), TBME/heptane (1 :1 , 660ml), and heptane (2x 1320ml) and dried at 4O 0 C in vacuo overnight to give the title compound (297.5g).
- the TBME and TBME/heptane washings were combined and reduced to dryness under vacuum.
- the residue was dissolved in TBME (990ml) with warming, the solution cooled to 32 0 C and rotated overnight.
- the solid was isolated by filtration, washed with TBME (130ml), TBME/heptane (1 :1 , 130ml), and heptane (2x 260ml). The solid was dried at 4O 0 C in vacuo overnight to give a second crop of the title compound (58.69g).
- the vessel was purged with nitrogen (x5), then hydrogen (x1 ) and hydrogenation under 15psi of hydrogen continued for ⁇ 15h.
- the reaction was filtered through Celite and then through a 1 micron Dominick Hunter before evaporation of the solvent in vacuo.
- the residue was dissolved in methanol with warming, filtered through Celite, then through a 0.2 micron Dominick Hunter before evaporation of the solvent in vacuo to leave the title compound. This material was used without further purification.
- Methanesulfonyl chloride (30ml) in DCM (100ml) was added dropwise to a solution of 1 ,5-anhydro-2,4-dideoxy-2-( ⁇ [(1 ,1-dimethylethyl)oxy]carbonyl ⁇ amino)-L-threo-pentitol (75g) and triethylamine (58ml) in DCM (900ml) at 0 0 C, maintaining the temperature below 3°C during the addition. The mixture was stirred for 30min, warmed to 25°C and stirred for 2h.
- Sodium acetate (129g), sodium azide (102g) and 1 ,5-anhydro-2,4-dideoxy-2-( ⁇ [(1 ,1- dimethylethyl)oxy]carbonyl ⁇ amino)-3-O-(methylsulfonyl)-L-threo-pentitol (232g) were mixed in DMF (11) and stirred and heated at 95°C for 6h. Water (21) was added and the mixture thoroughly mixed, ethyl acetate 1.51) was added and the mixture stirred for 5min.
- a mixture of platinum oxide and 1 ,1-dimethylethyl [(3R,4R)-4-azidotetrahydro-2H- pyran-3-yl]carbamate (42g) was purged with nitrogen (x3) and ethanol (11) was added.
- the vessel was purged (x3), charged with hydrogen and stirred at 400rpm while cooling at 20 0 C and stirred for 3h.
- the vessel was purged with nitrogen (x3), refilled with hydrogen and stirred for a further 3.5h.
- the vessel was purged and refilled with hydrogen at 15psi and stirred overnight.
- the vessel was purged and refilled and stirred for 1.5h.
- XRPD data were acquired on a PANalytical X'Pert Pro powder diffractometer, equipped with an X'Celerator detector.
- the acquisition conditions were: radiation: Cu Ka, generator tension: 40 kV, generator current: 45 mA, start angle: 2.0 ° 2 ⁇ , end angle: 40.0 ° 2 ⁇ , step size: 0.0167 ° 2 ⁇ .
- the time per step was 31.750s.
- the sample was prepared by mounting a few milligrams of sample on a Si wafer (zero background) plate, resulting in a thin layer of powder. The spectrum thus obtained is shown as Figure 1.
Abstract
Description
Claims
Priority Applications (13)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP10700169.5A EP2376481B1 (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase |
| ES10700169T ES2433109T3 (en) | 2009-01-13 | 2010-01-11 | Pyrimidine-carboxamide derivatives as inhibitors of Syk kinase |
| SG2011050390A SG172943A1 (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase |
| CN2010800117299A CN102348707A (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of SYK kinase |
| EA201190062A EA201190062A1 (en) | 2009-01-13 | 2010-01-11 | DERIVATIVES OF PYRIMIDINCARBOXAMIDE AS SYK-KINASE INHIBITORS |
| MX2011007499A MX2011007499A (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase. |
| AU2010219097A AU2010219097A1 (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of SYK kinase |
| US13/144,136 US8470835B2 (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of Syk kinase |
| CA2749403A CA2749403A1 (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase |
| JP2011544882A JP2012515148A (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as SYK kinase inhibitors |
| BRPI1006162A BRPI1006162A2 (en) | 2009-01-13 | 2010-01-11 | "compound, process for preparing a compound, pharmaceutical formulation, and use of a compound". |
| ZA2011/04949A ZA201104949B (en) | 2009-01-13 | 2011-05-05 | Pyrimidinecarboxamide derivatives as inhitors of syk kinase |
| IL213906A IL213906A0 (en) | 2009-01-13 | 2011-07-03 | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14421009P | 2009-01-13 | 2009-01-13 | |
| US61/144,210 | 2009-01-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010097248A1 true WO2010097248A1 (en) | 2010-09-02 |
Family
ID=41694078
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2010/050228 WO2010097248A1 (en) | 2009-01-13 | 2010-01-11 | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase |
Country Status (15)
| Country | Link |
|---|---|
| US (1) | US8470835B2 (en) |
| EP (1) | EP2376481B1 (en) |
| JP (1) | JP2012515148A (en) |
| KR (1) | KR20110100679A (en) |
| CN (1) | CN102348707A (en) |
| AU (1) | AU2010219097A1 (en) |
| BR (1) | BRPI1006162A2 (en) |
| CA (1) | CA2749403A1 (en) |
| EA (1) | EA201190062A1 (en) |
| ES (1) | ES2433109T3 (en) |
| IL (1) | IL213906A0 (en) |
| MX (1) | MX2011007499A (en) |
| SG (1) | SG172943A1 (en) |
| WO (1) | WO2010097248A1 (en) |
| ZA (1) | ZA201104949B (en) |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011134971A1 (en) | 2010-04-29 | 2011-11-03 | Glaxo Group Limited | 7-(1h-pyrazol-4-yl)-1,6-naphthyridine compounds as syk inhibitors |
| WO2012002577A1 (en) | 2010-06-30 | 2012-01-05 | 富士フイルム株式会社 | Novel nicotinamide derivatives or salts thereof |
| EP2489663A1 (en) | 2011-02-16 | 2012-08-22 | Almirall, S.A. | Compounds as syk kinase inhibitors |
| WO2013052393A1 (en) | 2011-10-05 | 2013-04-11 | Merck Sharp & Dohme Corp. | 3-PYRIDYL CARBOXAMIDE-CONTAINING SPLEEN TYROSINE KINASE (Syk) INHIBITORS |
| WO2013078468A1 (en) * | 2011-11-23 | 2013-05-30 | Portola Pharmaceuticals, Inc. | Selective kinase inhibitors |
| WO2013099041A1 (en) | 2011-12-28 | 2013-07-04 | 富士フイルム株式会社 | Novel nicotinamide derivative or salt thereof |
| CN103282352A (en) * | 2010-11-01 | 2013-09-04 | 波托拉医药品公司 | Benzamides and nicotinamides as syk modulators |
| WO2013155381A1 (en) | 2012-04-12 | 2013-10-17 | Alcon Research, Ltd. | Treatment for microbe-induced inflammatory responses in the eye |
| JP2014505020A (en) * | 2010-11-24 | 2014-02-27 | ガバメント オブ ザ ユナイテッド ステイツ オブ アメリカ, アズ リプレゼンテッド バイ ザ セクレタリー, デパートメント オブ ヘルス アンド ヒューマン サービシーズ | Compositions and methods for treating or preventing lupus |
| US8877760B2 (en) | 2011-11-23 | 2014-11-04 | Portola Pharmaceuticals, Inc. | Substituted pyrazine-2-carboxamide kinase inhibitors |
| US8952027B2 (en) | 2008-04-16 | 2015-02-10 | Portola Pharmaceuticals, Inc. | Inhibitors of syk and JAK protein kinases |
| EP2763974A4 (en) * | 2011-10-05 | 2015-06-03 | Merck Sharp & Dohme | PHENYL CARBOXAMIDE-CONTAINING SPLEEN TYROSINE KINASE (Syk) INHIBITORS |
| US9139534B2 (en) | 2011-04-22 | 2015-09-22 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| US9365524B2 (en) | 2014-01-30 | 2016-06-14 | Signal Pharmaceuticals, Llc | Solid forms of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide, compositions thereof and methods of their use |
| US9513297B2 (en) | 2014-12-16 | 2016-12-06 | Signal Pharmaceuticals, Llc | Methods for measurement of inhibition of c-Jun N-terminal kinase in skin |
| WO2017017571A1 (en) | 2015-07-24 | 2017-02-02 | Glaxo Group Limited | Treatment for vitiligo |
| US9796685B2 (en) | 2014-12-16 | 2017-10-24 | Signal Pharmaceuticals, Llc | Formulations of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-Methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US9868729B2 (en) | 2008-04-16 | 2018-01-16 | Portola Pharmaceuticals, Inc. | Inhibitors of protein kinases |
| US10017762B2 (en) | 2010-11-24 | 2018-07-10 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Compositions and methods for treating or preventing lupus |
| US10252981B2 (en) | 2015-07-24 | 2019-04-09 | Celgene Corporation | Methods of synthesis of (1R,2R,5R)-5-amino-2-methylcyclohexanol hydrochloride and intermediates useful therein |
| US10689351B2 (en) | 2015-01-29 | 2020-06-23 | Signal Pharmaceuticals, Llc | Isotopologues of 2-(tert butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide |
| WO2020188015A1 (en) | 2019-03-21 | 2020-09-24 | Onxeo | A dbait molecule in combination with kinase inhibitor for the treatment of cancer |
| WO2021089791A1 (en) | 2019-11-08 | 2021-05-14 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for the treatment of cancers that have acquired resistance to kinase inhibitors |
| WO2021148581A1 (en) | 2020-01-22 | 2021-07-29 | Onxeo | Novel dbait molecule and its use |
| WO2022059779A1 (en) | 2020-09-18 | 2022-03-24 | 大日本住友製薬株式会社 | Amine derivative |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014051654A2 (en) * | 2012-09-27 | 2014-04-03 | Portola Pharmaceuticals, Inc. | Bicyclic oxa-lactam kinase inhibitors |
| ES2691742T5 (en) | 2012-11-01 | 2022-03-18 | Infinity Pharmaceuticals Inc | Treatment of Cancers Using Modulators of PI3 Kinase Isoforms |
| NZ629037A (en) | 2013-03-15 | 2017-04-28 | Infinity Pharmaceuticals Inc | Salts and solid forms of isoquinolinones and composition comprising and methods of using the same |
| US20160113932A1 (en) | 2013-05-30 | 2016-04-28 | Infinity Pharmaceuticals, Inc. | Treatment of cancers using pi3 kinase isoform modulators |
| WO2015051241A1 (en) | 2013-10-04 | 2015-04-09 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| CA2925944C (en) | 2013-10-04 | 2023-01-10 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| WO2015061204A1 (en) | 2013-10-21 | 2015-04-30 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| EP4066834A1 (en) | 2014-03-19 | 2022-10-05 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds for use in the treatment of pi3k-gamma mediated disorders |
| US20150320754A1 (en) | 2014-04-16 | 2015-11-12 | Infinity Pharmaceuticals, Inc. | Combination therapies |
| WO2016054491A1 (en) | 2014-10-03 | 2016-04-07 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| US10919914B2 (en) | 2016-06-08 | 2021-02-16 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| WO2017223422A1 (en) | 2016-06-24 | 2017-12-28 | Infinity Pharmaceuticals, Inc. | Combination therapies |
| EP3512519A1 (en) | 2016-09-14 | 2019-07-24 | Gilead Sciences, Inc. | Syk inhibitors |
| TW201822764A (en) | 2016-09-14 | 2018-07-01 | 美商基利科學股份有限公司 | Syk inhibitors |
| WO2018195471A1 (en) | 2017-04-21 | 2018-10-25 | Gilead Sciences, Inc. | Syk inhibitors in combination with hypomethylating agents |
Citations (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4335121A (en) | 1980-02-15 | 1982-06-15 | Glaxo Group Limited | Androstane carbothioates |
| EP0123456A2 (en) | 1983-03-28 | 1984-10-31 | Compression Labs, Inc. | A combined intraframe and interframe transform coding method |
| GB2242134A (en) | 1990-03-02 | 1991-09-25 | Glaxo Group Ltd | Inhalation device |
| WO1999031073A1 (en) * | 1997-12-15 | 1999-06-24 | Yamanouchi Pharmaceutical Co., Ltd. | Novel pyrimidine-5-carboxamide derivatives |
| WO1999031181A1 (en) | 1997-12-17 | 1999-06-24 | Carnegie Mellon University | Rigidized trimethine cyanine dyes |
| WO2000007513A1 (en) | 1998-08-06 | 2000-02-17 | Smith & Nephew, Inc. | Orthopaedic bone screw apparatus |
| WO2002012266A1 (en) | 2000-08-05 | 2002-02-14 | Glaxo Group Limited | 17.beta.-carbothioate 17.alpha.-arylcarbonyloxyloxy androstane derivative as anti-inflammatory agents |
| EP1184376A1 (en) * | 1999-06-09 | 2002-03-06 | Yamanouchi Pharmaceutical Co. Ltd. | Novel heterocyclic carboxamide derivatives |
| US20020052312A1 (en) | 2000-05-30 | 2002-05-02 | Reiss Theodore F. | Combination therapy of chronic obstructive pulmonary disease using muscarinic receptor antagonists |
| WO2002069945A2 (en) | 2001-03-07 | 2002-09-12 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel medicament compositions on the basis of anticholinergics and pde iv inhibitors |
| US20020193393A1 (en) | 2001-03-07 | 2002-12-19 | Michel Pairet | Pharmaceutical compositions based on anticholinergics and PDE-IV inhibitors |
| WO2003011274A2 (en) | 2001-07-27 | 2003-02-13 | Glaxo Group Limited | Use of a pde4 inhibitor in combination with an anticholinergic agent for the treatment of pulmonary disease such as asthma |
| WO2003030939A1 (en) | 2001-10-05 | 2003-04-17 | Glaxo Group Limited | Therapies for treating respiratory diseases |
| WO2004035604A2 (en) | 2002-10-16 | 2004-04-29 | Millennium Pharmaceuticals, Inc. | Spleen tyrosine kinase catalytic domain:crystal structure and binding pockets thereof |
| WO2004103998A1 (en) | 2003-05-21 | 2004-12-02 | Glaxo Group Limited | Quinoline derivatives as phosphodiesterase inhibitors |
| WO2005030725A1 (en) | 2003-09-27 | 2005-04-07 | Glaxo Group Limited | Derivatives of 3-aminocarbonylquinoline, pharmaceutical compositions containing them and processes and intermediates for their preparation |
| WO2005030212A1 (en) | 2003-09-27 | 2005-04-07 | Glaxo Group Limited | 4-aminoquinoline-3-carboxamide derivatives as pde4 inhibitors |
| WO2005044354A1 (en) | 2003-11-03 | 2005-05-19 | Glaxo Group Limited | A fluid dispensing device |
| WO2005058892A1 (en) | 2003-12-19 | 2005-06-30 | Glaxo Group Limited | Pyrazolo [3,4-b] pyridine compounds, and their use as phosphodiesterase inhibitors |
| WO2005090353A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | PYRAZOLO[3,4-b]PYRIDINE COMPOUNDS, AND THEIR USE AS PDE4 INHIBITORS |
| WO2005090354A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | PYRAZOLO[3,4-b] PYRIDINE COMPOUNDS, AND THEIR USE AS PDE4 INHIBITORS |
| WO2005090348A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | Pyrazolo ’3,4-b! pyridine compounds, and their use as phosphodiesterase type 4 (pde4) inhibitors |
| WO2006053784A2 (en) | 2004-11-19 | 2006-05-26 | Glaxo Group Limited | 1,7-naphthyridines as pde4 inhibitors |
| WO2006097340A1 (en) | 2005-03-18 | 2006-09-21 | Glaxo Group Limited | 1,7-naphthyridines as pde4 inhibitors |
| WO2006133942A1 (en) | 2005-06-15 | 2006-12-21 | Glaxo Group Limited | NOVEL SALT FORM OF A β2 -ADRENERGIC AGONIST QUINOLIN-2-ONE DERIVATIVE |
| WO2007036734A1 (en) | 2005-09-29 | 2007-04-05 | Glaxo Group Limited | Pyrazolo[3,4-b]pyridine compound, and its use as a pde4 inhibitor |
| WO2007045861A1 (en) | 2005-10-21 | 2007-04-26 | Glaxo Group Limited | Cinnoline compounds as inhibitors of phosphodiesterase type iv (pde4) |
| WO2009136995A2 (en) * | 2008-04-16 | 2009-11-12 | Portola Pharmaceuticals, Inc. | Inhibitors of syk protein kinase |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102008007700A1 (en) * | 2007-10-31 | 2009-05-07 | Siemens Aktiengesellschaft | Method for the computer-aided exploration of states of a technical system |
-
2010
- 2010-01-11 BR BRPI1006162A patent/BRPI1006162A2/en not_active IP Right Cessation
- 2010-01-11 EA EA201190062A patent/EA201190062A1/en unknown
- 2010-01-11 ES ES10700169T patent/ES2433109T3/en active Active
- 2010-01-11 WO PCT/EP2010/050228 patent/WO2010097248A1/en active Application Filing
- 2010-01-11 AU AU2010219097A patent/AU2010219097A1/en not_active Abandoned
- 2010-01-11 KR KR1020117018877A patent/KR20110100679A/en not_active Application Discontinuation
- 2010-01-11 EP EP10700169.5A patent/EP2376481B1/en active Active
- 2010-01-11 US US13/144,136 patent/US8470835B2/en not_active Expired - Fee Related
- 2010-01-11 MX MX2011007499A patent/MX2011007499A/en not_active Application Discontinuation
- 2010-01-11 SG SG2011050390A patent/SG172943A1/en unknown
- 2010-01-11 CA CA2749403A patent/CA2749403A1/en not_active Abandoned
- 2010-01-11 CN CN2010800117299A patent/CN102348707A/en active Pending
- 2010-01-11 JP JP2011544882A patent/JP2012515148A/en active Pending
-
2011
- 2011-05-05 ZA ZA2011/04949A patent/ZA201104949B/en unknown
- 2011-07-03 IL IL213906A patent/IL213906A0/en unknown
Patent Citations (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4335121A (en) | 1980-02-15 | 1982-06-15 | Glaxo Group Limited | Androstane carbothioates |
| EP0123456A2 (en) | 1983-03-28 | 1984-10-31 | Compression Labs, Inc. | A combined intraframe and interframe transform coding method |
| GB2242134A (en) | 1990-03-02 | 1991-09-25 | Glaxo Group Ltd | Inhalation device |
| WO1999031073A1 (en) * | 1997-12-15 | 1999-06-24 | Yamanouchi Pharmaceutical Co., Ltd. | Novel pyrimidine-5-carboxamide derivatives |
| EP1054004A1 (en) | 1997-12-15 | 2000-11-22 | Yamanouchi Pharmaceutical Co. Ltd. | Novel pyrimidine-5-carboxamide derivatives |
| WO1999031181A1 (en) | 1997-12-17 | 1999-06-24 | Carnegie Mellon University | Rigidized trimethine cyanine dyes |
| WO2000007513A1 (en) | 1998-08-06 | 2000-02-17 | Smith & Nephew, Inc. | Orthopaedic bone screw apparatus |
| EP1184376B1 (en) | 1999-06-09 | 2005-02-02 | Yamanouchi Pharmaceutical Co. Ltd. | Novel heterocyclic carboxamide derivatives |
| EP1184376A1 (en) * | 1999-06-09 | 2002-03-06 | Yamanouchi Pharmaceutical Co. Ltd. | Novel heterocyclic carboxamide derivatives |
| US20020052312A1 (en) | 2000-05-30 | 2002-05-02 | Reiss Theodore F. | Combination therapy of chronic obstructive pulmonary disease using muscarinic receptor antagonists |
| WO2002012266A1 (en) | 2000-08-05 | 2002-02-14 | Glaxo Group Limited | 17.beta.-carbothioate 17.alpha.-arylcarbonyloxyloxy androstane derivative as anti-inflammatory agents |
| WO2002069945A2 (en) | 2001-03-07 | 2002-09-12 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Novel medicament compositions on the basis of anticholinergics and pde iv inhibitors |
| US20020193393A1 (en) | 2001-03-07 | 2002-12-19 | Michel Pairet | Pharmaceutical compositions based on anticholinergics and PDE-IV inhibitors |
| WO2003011274A2 (en) | 2001-07-27 | 2003-02-13 | Glaxo Group Limited | Use of a pde4 inhibitor in combination with an anticholinergic agent for the treatment of pulmonary disease such as asthma |
| WO2003030939A1 (en) | 2001-10-05 | 2003-04-17 | Glaxo Group Limited | Therapies for treating respiratory diseases |
| WO2004035604A2 (en) | 2002-10-16 | 2004-04-29 | Millennium Pharmaceuticals, Inc. | Spleen tyrosine kinase catalytic domain:crystal structure and binding pockets thereof |
| WO2004103998A1 (en) | 2003-05-21 | 2004-12-02 | Glaxo Group Limited | Quinoline derivatives as phosphodiesterase inhibitors |
| WO2005030725A1 (en) | 2003-09-27 | 2005-04-07 | Glaxo Group Limited | Derivatives of 3-aminocarbonylquinoline, pharmaceutical compositions containing them and processes and intermediates for their preparation |
| WO2005030212A1 (en) | 2003-09-27 | 2005-04-07 | Glaxo Group Limited | 4-aminoquinoline-3-carboxamide derivatives as pde4 inhibitors |
| WO2005044354A1 (en) | 2003-11-03 | 2005-05-19 | Glaxo Group Limited | A fluid dispensing device |
| WO2005058892A1 (en) | 2003-12-19 | 2005-06-30 | Glaxo Group Limited | Pyrazolo [3,4-b] pyridine compounds, and their use as phosphodiesterase inhibitors |
| WO2005090353A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | PYRAZOLO[3,4-b]PYRIDINE COMPOUNDS, AND THEIR USE AS PDE4 INHIBITORS |
| WO2005090354A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | PYRAZOLO[3,4-b] PYRIDINE COMPOUNDS, AND THEIR USE AS PDE4 INHIBITORS |
| WO2005090352A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | Pyrazolo[3,4-b]pyridine compound, and its use as a pde4 inhibitor |
| WO2005090348A1 (en) | 2004-03-16 | 2005-09-29 | Glaxo Group Limited | Pyrazolo ’3,4-b! pyridine compounds, and their use as phosphodiesterase type 4 (pde4) inhibitors |
| WO2006053784A2 (en) | 2004-11-19 | 2006-05-26 | Glaxo Group Limited | 1,7-naphthyridines as pde4 inhibitors |
| WO2006097340A1 (en) | 2005-03-18 | 2006-09-21 | Glaxo Group Limited | 1,7-naphthyridines as pde4 inhibitors |
| WO2006133942A1 (en) | 2005-06-15 | 2006-12-21 | Glaxo Group Limited | NOVEL SALT FORM OF A β2 -ADRENERGIC AGONIST QUINOLIN-2-ONE DERIVATIVE |
| WO2007036734A1 (en) | 2005-09-29 | 2007-04-05 | Glaxo Group Limited | Pyrazolo[3,4-b]pyridine compound, and its use as a pde4 inhibitor |
| WO2007036733A1 (en) | 2005-09-29 | 2007-04-05 | Glaxo Group Limited | Pyrazolo[3,4-b]pyridine compounds, and their use as pde4 inhibitors |
| WO2007045861A1 (en) | 2005-10-21 | 2007-04-26 | Glaxo Group Limited | Cinnoline compounds as inhibitors of phosphodiesterase type iv (pde4) |
| WO2009136995A2 (en) * | 2008-04-16 | 2009-11-12 | Portola Pharmaceuticals, Inc. | Inhibitors of syk protein kinase |
Non-Patent Citations (12)
| Title |
|---|
| ARTHRITIS, RHEUMATIS, vol. 58, no. 11, 2008, pages 3309 - 3318 |
| CHENG ET AL., NATURE, vol. 378, 1995, pages 303 - 306 |
| CURRENT OPINIONS IN IMMUNOLOGY, vol. 20, 2008, pages 709 - 716 |
| GURURAJAN M. ET AL., JOURNAL OF IMMUNOLOGY, vol. 178, 2007, pages 111 - 121 |
| H. HISAMICHI ET AL., BIOORG MED CHEM, vol. 13, 2005, pages 4936 - 4951 |
| J.C.W. EDWARDS ET AL., NEW ENG. J. MED., vol. 350, 2004, pages 2572 - 2581 |
| J.M.C., vol. 50, no. 13, 2007, pages 2931 - 2941 |
| KURASAKI ET AL., IMMUNOL. REV., vol. 176, 2000, pages 19 - 29 |
| LESEUX L. ET AL., BLOOD, vol. 108, no. 13, 15 December 2006 (2006-12-15), pages 4156 - 4162 |
| M. TURNER ET AL., NATURE, vol. 379, 1995, pages 298 - 302 |
| MELTZER, ELI 0.; BERKOWITZ, ROBERT B.; GROSSBARD, ELLIOTT B.: "An intranasal Syk kinase inhibitor (R112) improves the symptoms of seasonal allergic rhinitis in a park environment", JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY, vol. 115, no. 4, 2005, pages 791 - 796 |
| T. W. GREENE: "Protective Groups in Organic Synthesis", 1991, J. WILEY AND SONS |
Cited By (50)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10533001B2 (en) | 2008-04-16 | 2020-01-14 | Portola Pharmaceuticals, Inc. | Inhibitors of protein kinases |
| US9579320B2 (en) | 2008-04-16 | 2017-02-28 | Portola Pharmaceuticals, Inc. | Inhibitors of syk and JAK protein kinases |
| US11414410B2 (en) | 2008-04-16 | 2022-08-16 | Alexion Pharmaceuticals, Inc. | Inhibitors of protein kinases |
| US9868729B2 (en) | 2008-04-16 | 2018-01-16 | Portola Pharmaceuticals, Inc. | Inhibitors of protein kinases |
| US8952027B2 (en) | 2008-04-16 | 2015-02-10 | Portola Pharmaceuticals, Inc. | Inhibitors of syk and JAK protein kinases |
| WO2011134971A1 (en) | 2010-04-29 | 2011-11-03 | Glaxo Group Limited | 7-(1h-pyrazol-4-yl)-1,6-naphthyridine compounds as syk inhibitors |
| WO2012002577A1 (en) | 2010-06-30 | 2012-01-05 | 富士フイルム株式会社 | Novel nicotinamide derivatives or salts thereof |
| CN103282352B (en) * | 2010-11-01 | 2016-08-10 | 波托拉医药品公司 | Benzamides and nicotinamide as SYK regulator |
| CN103282352A (en) * | 2010-11-01 | 2013-09-04 | 波托拉医药品公司 | Benzamides and nicotinamides as syk modulators |
| JP2014505020A (en) * | 2010-11-24 | 2014-02-27 | ガバメント オブ ザ ユナイテッド ステイツ オブ アメリカ, アズ リプレゼンテッド バイ ザ セクレタリー, デパートメント オブ ヘルス アンド ヒューマン サービシーズ | Compositions and methods for treating or preventing lupus |
| US10907151B2 (en) | 2010-11-24 | 2021-02-02 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Compositions and methods for treating or preventing lupus |
| US10017762B2 (en) | 2010-11-24 | 2018-07-10 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Compositions and methods for treating or preventing lupus |
| US9657292B2 (en) | 2010-11-24 | 2017-05-23 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Compositions and methods for treating or preventing lupus |
| EP2489663A1 (en) | 2011-02-16 | 2012-08-22 | Almirall, S.A. | Compounds as syk kinase inhibitors |
| US10919865B2 (en) | 2011-04-22 | 2021-02-16 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| US9139534B2 (en) | 2011-04-22 | 2015-09-22 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| US10266500B2 (en) | 2011-04-22 | 2019-04-23 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| US10040770B2 (en) | 2011-04-22 | 2018-08-07 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| US9701643B2 (en) | 2011-04-22 | 2017-07-11 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| US11325890B2 (en) | 2011-04-22 | 2022-05-10 | Signal Pharmaceuticals, Llc | Substituted diaminocarboxamide and diaminocarbonitrile pyrimidines, compositions thereof, and methods of treatment therewith |
| EP2763974A4 (en) * | 2011-10-05 | 2015-06-03 | Merck Sharp & Dohme | PHENYL CARBOXAMIDE-CONTAINING SPLEEN TYROSINE KINASE (Syk) INHIBITORS |
| WO2013052393A1 (en) | 2011-10-05 | 2013-04-11 | Merck Sharp & Dohme Corp. | 3-PYRIDYL CARBOXAMIDE-CONTAINING SPLEEN TYROSINE KINASE (Syk) INHIBITORS |
| WO2013078468A1 (en) * | 2011-11-23 | 2013-05-30 | Portola Pharmaceuticals, Inc. | Selective kinase inhibitors |
| US8877760B2 (en) | 2011-11-23 | 2014-11-04 | Portola Pharmaceuticals, Inc. | Substituted pyrazine-2-carboxamide kinase inhibitors |
| US9359308B2 (en) | 2011-11-23 | 2016-06-07 | Portola Pharmaceuticals, Inc. | Pyrazine kinase inhibitors |
| WO2013099041A1 (en) | 2011-12-28 | 2013-07-04 | 富士フイルム株式会社 | Novel nicotinamide derivative or salt thereof |
| WO2013155381A1 (en) | 2012-04-12 | 2013-10-17 | Alcon Research, Ltd. | Treatment for microbe-induced inflammatory responses in the eye |
| US10517873B2 (en) | 2014-01-30 | 2019-12-31 | Signal Pharmaceuticals, Llc | Solid forms of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide, compositions thereof and methods of their use |
| EP3738954A1 (en) * | 2014-01-30 | 2020-11-18 | Signal Pharmaceuticals, LLC | Method of preparation of substituted 2,4-diamino-pyrimidine-5-carboxamides |
| US9814713B2 (en) | 2014-01-30 | 2017-11-14 | Signal Pharmaceuticals, Llc | Solid forms of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide, compositions thereof and methods of their use |
| US10226461B2 (en) | 2014-01-30 | 2019-03-12 | Signal Pharmaceuticals, Llc | Solid forms of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide, compositions thereof and methods of their use |
| US11241430B2 (en) | 2014-01-30 | 2022-02-08 | Signal Pharmaceuticals, Llc | Solid forms of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide, compositions thereof and methods of their use |
| US9365524B2 (en) | 2014-01-30 | 2016-06-14 | Signal Pharmaceuticals, Llc | Solid forms of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide, compositions thereof and methods of their use |
| US10590089B2 (en) | 2014-12-16 | 2020-03-17 | Signal Pharmaceuticals, Llc | Formulations of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US9796685B2 (en) | 2014-12-16 | 2017-10-24 | Signal Pharmaceuticals, Llc | Formulations of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-Methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US9513297B2 (en) | 2014-12-16 | 2016-12-06 | Signal Pharmaceuticals, Llc | Methods for measurement of inhibition of c-Jun N-terminal kinase in skin |
| US10131639B2 (en) | 2014-12-16 | 2018-11-20 | Signal Pharmaceuticals, Llc | Formulations of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US10197579B2 (en) | 2014-12-16 | 2019-02-05 | Signal Pharmaceuticals, Llc | Methods for measurement of inhibition of c-Jun N-terminal kinase in skin |
| US10689351B2 (en) | 2015-01-29 | 2020-06-23 | Signal Pharmaceuticals, Llc | Isotopologues of 2-(tert butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US11492332B2 (en) | 2015-01-29 | 2022-11-08 | Signal Pharmaceuticals, Llc | Isotopologues of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US10975039B2 (en) | 2015-01-29 | 2021-04-13 | Signal Pharmaceuticals, Llc | Isotopologues of 2-(tert-butylamino)-4-((1R,3R,4R)-3-hydroxy-4-methylcyclohexylamino)-pyrimidine-5-carboxamide |
| US10774033B2 (en) | 2015-07-24 | 2020-09-15 | Celgene Corporation | Methods of synthesis of (1R, 2R, 5R)-5-amino-2-methylcyclohexanol hydrochloride and intermediates useful therein |
| US11192847B2 (en) | 2015-07-24 | 2021-12-07 | Celgene Corporation | Methods of synthesis of (1R,2R,5R)-5-amino-2-methylcyclohexanol hydrochloride and intermediates useful therein |
| US10252981B2 (en) | 2015-07-24 | 2019-04-09 | Celgene Corporation | Methods of synthesis of (1R,2R,5R)-5-amino-2-methylcyclohexanol hydrochloride and intermediates useful therein |
| WO2017017571A1 (en) | 2015-07-24 | 2017-02-02 | Glaxo Group Limited | Treatment for vitiligo |
| US11780801B2 (en) | 2015-07-24 | 2023-10-10 | Celgene Corporation | Methods of synthesis of (1R,2R,5R)-5-amino-2-methyl-cyclohexanol hydrochloride and intermediates useful therein |
| WO2020188015A1 (en) | 2019-03-21 | 2020-09-24 | Onxeo | A dbait molecule in combination with kinase inhibitor for the treatment of cancer |
| WO2021089791A1 (en) | 2019-11-08 | 2021-05-14 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for the treatment of cancers that have acquired resistance to kinase inhibitors |
| WO2021148581A1 (en) | 2020-01-22 | 2021-07-29 | Onxeo | Novel dbait molecule and its use |
| WO2022059779A1 (en) | 2020-09-18 | 2022-03-24 | 大日本住友製薬株式会社 | Amine derivative |
Also Published As
| Publication number | Publication date |
|---|---|
| MX2011007499A (en) | 2011-08-04 |
| US8470835B2 (en) | 2013-06-25 |
| ES2433109T3 (en) | 2013-12-09 |
| JP2012515148A (en) | 2012-07-05 |
| AU2010219097A1 (en) | 2011-08-04 |
| EP2376481A1 (en) | 2011-10-19 |
| BRPI1006162A2 (en) | 2019-09-24 |
| KR20110100679A (en) | 2011-09-14 |
| EA201190062A1 (en) | 2012-02-28 |
| ZA201104949B (en) | 2012-12-27 |
| EP2376481B1 (en) | 2013-08-07 |
| CA2749403A1 (en) | 2010-09-02 |
| SG172943A1 (en) | 2011-08-29 |
| CN102348707A (en) | 2012-02-08 |
| IL213906A0 (en) | 2011-07-31 |
| US20110275655A1 (en) | 2011-11-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2376481B1 (en) | Pyrimidinecarboxamide derivatives as inhibitors of syk kinase | |
| WO2006129100A1 (en) | Novel compounds | |
| WO2007009681A1 (en) | 1 , 1-DIOXID0-2 , 3-DIHYDRO-l , 2-BENZISOTHIAZ0L-6-YL-1H-INDAZOL-4-YL-2 , 4-PYRIMIDINEDI AMINE DERIVATIVES | |
| EP3119766B1 (en) | Heteroaryl syk inhibitors | |
| JP5520969B2 (en) | Pyrazole derivatives for use as CCR4 receptor antagonists | |
| WO2007028445A1 (en) | 6-indolyl-4-yl-amino-5-halogeno-2-pyrimidinyl-amino derivatives | |
| EP2613781B1 (en) | Indazole derivatives for use in the treatment of influenza virus infection | |
| JP6663857B2 (en) | Pyrazolopyridine and pyrazolopyrimidine | |
| JP5959537B2 (en) | Substituted pyridinyl-pyrimidines and their use as pharmaceuticals | |
| JP6182592B2 (en) | Amino-indolyl-substituted imidazolyl-pyrimidines and their use as pharmaceuticals | |
| WO2007085540A1 (en) | 1h-indaz0l-4-yl-2 , 4-pyrimidinediamine derivatives | |
| CA2987179C (en) | Heterocyclic compounds as inhibitors of vanin-1 enzyme | |
| US20080004295A1 (en) | Novel compounds | |
| KR20150068953A (en) | Pyrrolotriazinone derivatives as pi3k inhibitors | |
| JP6463680B2 (en) | 2- (2-Aminocyclohexyl) aminopyrimidine-5-carboxamides as spleen tyrosine kinase I (SYK) inhibitors | |
| AU2011290727B2 (en) | Disubstituted tetrahydofuranyl compounds as antagonists of the bradykinin B1 receptor | |
| WO2011143105A1 (en) | Bifunctional quinoline derivatives | |
| WO2011143106A1 (en) | Bi - functional pyrazolopyridine compounds | |
| WO2008015416A1 (en) | Pyrazolo[3,4-b]pyridine compounds, and their use as pde4 inhibitors | |
| US20160368919A1 (en) | Novel pyridine pyrazinones as bet-family bromodomain inhibitors | |
| TW201139426A (en) | Salts and hydrates of 4-[(3-chlor-4-fluor-phenyl)amino]-6-(cis-4-{n-[(morph-1-olin-4-yl)carbonyl]-n-methyl-amino}-cyclohexan-1-yloxy)-7-methoxy-quinazoline, their use as a medicament and the preparation thereof | |
| MX2011008505A (en) | 2-morpholino-pyrido[3,2-d]pyrimidines. | |
| NZ614199B2 (en) | Pyridinyl- and pyrazinyl -methyloxy - aryl derivatives useful as inhibitors of spleen tyrosine kinase (syk) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080011729.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10700169 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201190062 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 2011544882 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2749403 Country of ref document: CA Ref document number: 13144136 Country of ref document: US Ref document number: 2010700169 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010219097 Country of ref document: AU Ref document number: MX/A/2011/007499 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3115/KOLNP/2011 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2010219097 Country of ref document: AU Date of ref document: 20100111 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20117018877 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI1006162 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: PI1006162 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110712 |