WO2010088668A2 - Methods of detecting sepsis - Google Patents
Methods of detecting sepsis Download PDFInfo
- Publication number
- WO2010088668A2 WO2010088668A2 PCT/US2010/022885 US2010022885W WO2010088668A2 WO 2010088668 A2 WO2010088668 A2 WO 2010088668A2 US 2010022885 W US2010022885 W US 2010022885W WO 2010088668 A2 WO2010088668 A2 WO 2010088668A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- target rna
- seq
- nos
- sequence
- complementary
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/178—Oligonucleotides characterized by their use miRNA, siRNA or ncRNA
Definitions
- Sepsis is the presence in the blood or other tissues of pathogenic microorganisms or their toxins combined with the host's inflammatory response, known as systemic inflammatory response syndrome (“SIRS”) caused by the infection.
- SIRS systemic inflammatory response syndrome
- TLR toll-like receptors
- TLRs recognize them and stimulate an immune response.
- sepsis is also characterized by symptoms of acute inflammation brought on by the host's immune response. These latter symptoms may include fever and elevated white blood cell count, or low white blood cell count and low body temperature.
- SIRS is characterized by hemodynamic compromise and resultant metabolic dysregulation, and may be accompanied by symptoms such as high heart rate, high respiratory rate and elevated body temperature.
- the immunological response also causes widespread activation of acute phase proteins, affecting the complement system and the coagulation pathways, which then cause damage to the vasculature and organs.
- Various neuroendocrine counter-regulatory systems are then activated as well, often compounding the problem.
- Sepsis is often treated in the intensive care unit with intravenous fluids and antibiotics and/or antiviral compounds.
- sepsis progresses quickly, and so even with immediate and aggressive treatment, severe sepsis can lead to organ failure and death.
- Severe sepsis is estimated to cause 215,000 deaths per year in the United States, more than acute myocardial infarction, stroke or pneumonia, which is likely due to late diagnosis or misdiagnosis of sepsis.
- a method comprises detecting a level of at least one target RNA in a sample from the subject.
- the at least one target RNA (i) is capable of specifically hybridizing to a nucleic acid having a sequence selected from SEQ ID NOs: 1 to 86; or (ii) comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86; or (iii) comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- a method comprises comparing the level of the at least one target RNA in the sample to a normal level of the at least one target RNA. In some embodiments, a level of at least one target RNA in the sample that is greater than a normal level of the at least one target RNA indicates the presence of sepsis in the subject.
- the method comprises detecting a level of at least one target RNA in a sample from the subject.
- the at least one target RNA (i) is capable of specifically hybridizing to a nucleic acid having a sequence selected from SEQ ID NOs: 1 to 86; or (ii) comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86; or (iii) comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- a method comprises communicating the results of the detection to a medical practitioner for the purpose of determining whether the subject has sepsis.
- detecting a level of at least one target RNA in a sample comprises hybridizing nucleic acids of the sample with at least one polynucleotide that is complementary to a target RNA in the sample or to a complement thereof.
- a method further comprises detecting at least one complex comprising a polynucleotide hybridized to at least one nucleic acid selected from the target RNA, a DNA amplicon of the target RNA, and a complement of the target RNA.
- a method for detecting the presence of sepsis in a subject comprises obtaining a sample from the subject and providing the sample to a laboratory for detection of the level of at least one target RNA in the sample.
- the at least one target RNA (i) is capable of specifically hybridizing to a nucleic acid having a sequence selected from SEQ ID NOs: 1 to 86; or (ii) comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86; or (iii) comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- the method comprises receiving from the laboratory a communication indicating the level of at least one target RNA in the sample. In some embodiments, a level of at least one target RNA that is greater than a normal level of the at least one target RNA indicates the presence of sepsis.
- a method comprises detecting levels of at least two, at least three, at least five, or at least ten target RNAs. In some embodiments, detection of a level of at least one target RNA that is greater than a normal level of the at least one target RNA indicates the presence of sepsis. In some embodiments, detection of levels of at least two target RNAs that are greater than normal levels of the at least two target RNAs indicates the presence of sepsis. In some embodiments, detection of levels of at least three target RNAs that are greater than normal levels of the at least two target RNAs indicates the presence of sepsis. In some embodiments, detection of levels of at least five target RNAs that are greater than normal levels of the at least two target RNAs indicates the presence of sepsis.
- a method comprises detecting a level of at least one target RNA that (i) does not specifically hybridize to a nucleic acid having a sequence selected from SEQ ID NOs: 1 to 86; and (ii) does not comprise a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86; and (iii) does not comprise at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- a synthetic polynucleotide comprises a first region, wherein the first region comprises a sequence of at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, or at least 18 contiguous nucleotides that is identical or complementary to a sequence of at least 8 contiguous nucleotides of one of SEQ ID NOs: 1 to 67 and 215 to 399.
- the first region is identical or complementary to a region of a target RNA.
- a synthetic polynucleotide comprises a second region that is not identical or complementary to a region of the target RNA. In some embodiments, a synthetic polynucleotide comprises a detectable label. In some embodiments, a synthetic polynucleotide comprises a FRET label.
- a composition is provided.
- a composition comprises a plurality of synthetic polynucleotides.
- a kit is provided.
- a kit comprises a synthetic polynucleotide.
- a kit comprises a composition.
- a kit comprises at least one polymerase and/or dNTPs.
- FIG. 1 shows an electropherogram obtained on an Agilent Bioanalyser 2100 to assess the quality of total RNA purified as described in Example 1 from human monocyte cell line THP-I after stimulation for 8h with an agonist, Pam3CSK4.
- Methods detecting sepsis by measuring levels of microRNA species are provided.
- elevated levels of microRNA species are indicative of sepsis.
- reduced levels of microRNA species are indicative of sepsis.
- the method comprises detecting an above-normal level of at least one target RNA that is capable of specifically hybridizing to a sequence selected from SEQ ID NOs: 1 to 86.
- the method comprises detecting an above-normal level of at least one target RNA that comprises at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NO.: 196 to 399, 565 to 707, and 863 to 897.
- the method comprises detecting an above-normal level of at least one target RNA that comprises a sequence that is complementary to at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NO.:1 to 86.
- the target RNA in its mature form, comprises fewer than 30 nucleotides.
- the target RNA in some embodiments, is a microRNA.
- sequence selected from encompasses both “one sequence selected from” and “one or more sequences selected from.” Thus, when “a sequence selected from” is used, it is to be understood that one, or more than one, of the listed sequences may be chosen.
- Detection of a level of target RNA that is greater than a normal level of target RNA indicates the presence of sepsis in the patient from whom the sample is taken sample. In some embodiments, the detecting is done quantitatively. In other embodiments, the detecting is done qualitatively. In some embodiments, detecting a target RNA comprises forming a complex comprising a polynucleotide and a nucleic acid selected from a target RNA, a DNA amplicon of a target RNA, and a complement of a target RNA. In some embodiments, the level of the complex is then detected and compared to a normal level of the same complex. The level of the complex, in some embodiments, correlates with the level of the target RNA in the sample.
- Sepsis is an infection accompanied by an acute inflammatory reaction (systemic inflammatory response syndrome) with systemic manifestations associated with release of endogenous mediators of inflammation into the bloodstream. If left untreated, sepsis can become severe sepsis, which is often accompanied by the failure of at least one organ or septic shock, which is severe sepsis accompanied by organ hypoperfusion and hypotension that are poorly responsive to initial fluid resuscitation.
- the systemic inflammatory response is mediated by toll-like receptors ("TLRs").
- TLRs Toll-like receptors
- TLRs are a class of proteins in vertebrates and invertebrates that recognize particular structurally conserved molecules on microorganisms that are distinguishable from host molecules, and which mediate immune cell responses. TLRs are located either on the surface of cells or in cellular compartments and are classified by the types of molecules they recognize and that stimulate them, as shown in Table 1.
- Stimulation of various TLRs results in over-expression of one or more target RNAs, as shown in Table 2.
- one or more target RNAs is over-expressed as a result of stimulation of a subset of TLRs that recognize bacteria (e.g., TLRl, TLR2, TLR4 or TLR5).
- one or more target RNAs is over- expressed as a result of stimulation of a subset of TLRs that recognize viruses (e.g. TLR3 or TLR7).
- one or more target RNAs is over-expressed as a result of stimulation of a subset of TLRs that recognize molecules common to both bacteria and viruses (TLR9).
- one or more target RNAs is over-expressed as a result of stimulation of a subset of TLRs that recognize gram-negative bacteria (e.g., TLR4a and TLR4b). In some embodiments, one or more target RNAs is over-expressed as a result of stimulation of a subset of TLRs that recognize both gram-negative and gram-positive bacteria (e.g., TLR2a, TLR2b and TLR5). In some embodiments, one or more target RNAs is over-expressed as a result of stimulation of a subset of TLRs that recognize gram-positive bacteria, gram-negative bacteria and mycobacteria (e.g., TLR2a).
- Table 2 lists 86 hybridization probes that have been found to be complimentary to, and to hybridize with, target RNAs in human monocytes stimulated with various toll-like receptor agonists (ligands). These target RNAs can be detected at elevated levels in stimulated THP-I cells as demonstrated in Example 1. Sixty-seven of the probes are complementary to, and hybridize with, novel target RNA species that are expressed in human cells. The other nineteen probes are complementary to, and hybridize with, publicly known microRNAs that have been submitted by others to miRBase (http://microrna.sanger.ac.uk/; see Griffiths- Jones S. et al. (2007) Nucl. Acids Res.
- microRNAs have not been disclosed to have utility for detection of sepsis.
- the sequences of those microRNAs are shown in Table 4.
- Certain candidate microRNAs that may hybridize to certain probes listed in Table 2 are shown in Table 11.
- Table 12 lists microRNAs that are present at elevated levels in a sepsis patient sample. Some pairs of microRNAs listed in Table 12 have the same sequences. In such instances, the precursor gene for that microRNA sequence is located at multiple locations in the genome, so the sequence may be from any of those genes. When a precursor gene for a particular microRNA sequence is present at multiple locations in the genome, multiple candidate names are shown (based on each of the precursor genes), with the same ranking and same sequence. One or more of those candidates may be upregulated in the sepsis patient sample. Some of the microRNAs listed in Table 12 are isomirs of one another. When multiple isomirs are listed in Table 12, one or more than one of the isomirs may be present at elevated levels in a sample from a patient with sepsis.
- Table 14 lists microRNAs from miRBase that are present at elevated levels in a sepsis patient sample.
- Table 16 lists microRNA star forms that are present at elevated levels in a sepsis patient sample. While the mature microRNAs for the listed star forms have been identified and are submitted into miRBase, none of the star forms in Table 16 have, to the inventors' knowledge, been previously identified or submitted to miRBase.
- a method comprises detecting multiple isomirs with a single probe. Detection of an elevated level of one or multiple isomirs is considered to be indicative of sepsis.
- Detection of an elevated level of one or multiple isomirs is considered to be indicative of sepsis.
- one or more of the genes may be upregulated in a sepsis patient. Detection of a microRNA expressed from any one of the genes is considered to be indicative of sepsis.
- target RNA species are denominated “microRNAs” in the tables set forth herein and Example 1.
- the target RNA is a single mature microRNA capable of specifically hybridizing to a hybridization probe set forth in Table 2.
- a target RNA is a single mature microRNA that comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NO.:1 to 86.
- a target RNA is a single mature microRNA that comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- target RNA may include a plurality of target RNAs, all of which are capable of specifically hybridizing to a single complementary probe sequence (for example, when two or more target microRNAs are isomirs).
- the so-denominated "microRNA" is one or more RNA species capable of specifically hybridizing to the respective hybridization probe, such that one or more target RNAs do not meet canonical definitions for mature microRNAs.
- a target RNA is an mRNA.
- the "target RNA” is a piwi-interacting RNA (piRNA), i.e., a small RNA expressed in animal cells that is distinct in size (26-31 nt) from microRNA and that forms distinct complexes with Piwi proteins that are involved in transcriptional gene silencing.
- piRNA piwi-interacting RNA
- Mature human microRNAs are typically composed of 17-27 contiguous ribonucleotides, and often are 21 or 22 nucleotides in length.
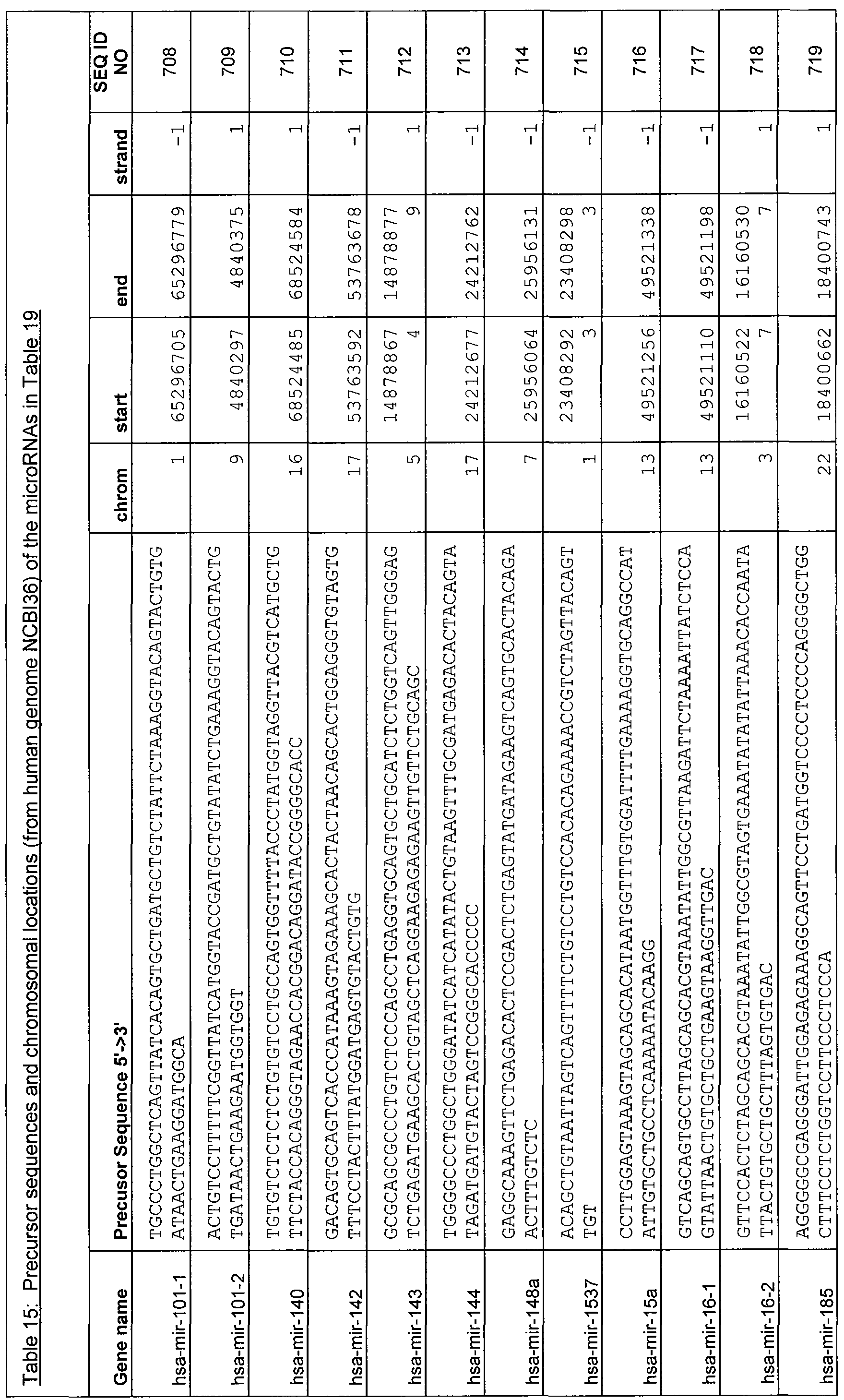
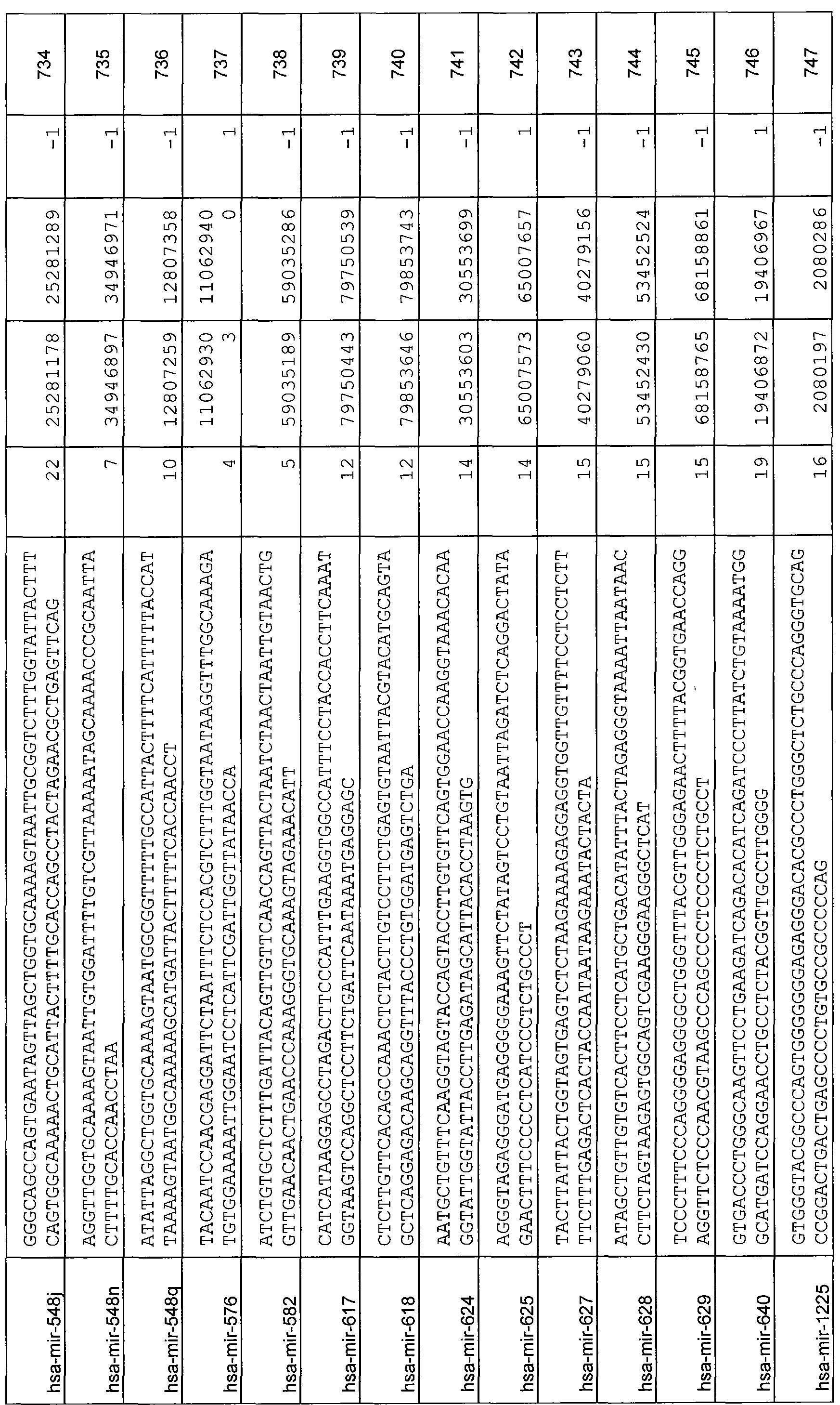
- the sequences of some target microRNAs that can be detected in accordance with the present disclosure can be found within the pre-microRNA sequences shown in Tables 3, 13, 15, and 17 (SEQ ID NOs: 87 to 177, 400 to 564, 708 to 862, and 898 to 932).
- the sequences of some publicly known microRNAs are shown in Tables 4 and 14.
- a microRNA comprises at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, at least 25, or at least 26 contiguous nucleotides of a sequence in Table 11, 12, or 16 (SEQ ID NOs: 215 to 399 and 863 to 897).
- mammalian microRNAs mature as described herein.
- a gene coding for a microRNA is transcribed, leading to production of a microRNA precursor known as the "pri -microRNA" or "pri-miRNA.”
- the pri-miRNA can be part of a polycistronic RNA comprising multiple pri-miRNAs.
- the pri-miRNA forms a hairpin with a stem and loop, which may comprise mismatched bases.
- the hairpin structure of the pri-miRNA is recognized by Drosha, which is an RNase III endonuclease protein.
- Drosha can recognize terminal loops in the pri-miRNA and cleave approximately two helical turns into the stem to produce a 60-70 nucleotide precursor known as the "pre-microRNA” or "pre-miRNA.”
- Drosha can cleave the pri-miRNA with a staggered cut typical of RNase III endonucleases yielding a pre-miRNA stem loop with a 5' phosphate and an approximately 2-nucleotide 3' overhang.
- Approximately one helical turn of the stem (about 10 nucleotides) extending beyond the Drosha cleavage site can be essential for efficient processing.
- the pre-miRNA is subsequently actively transported from the nucleus to the cytoplasm by Ran-GTP and the export receptor Exportin-5.
- the pre-miRNA can be recognized by Dicer, another RNase III endonuclease.
- Dicer recognizes the double-stranded stem of the pre-miRNA.
- Dicer may also recognize the 5' phosphate and 3' overhang at the base of the stem loop.
- Dicer may cleave off the terminal loop two helical turns away from the base of the stem loop leaving an additional 5' phosphate and an approximately 2-nucleotide 3' overhang.
- the resulting siRNA-like duplex which may comprise mismatches, comprises the mature microRNA and a similar-sized fragment known as the microRNA*.
- the microRNA and microRNA* may be derived from opposing arms of the pri-miRNA and pre-miRNA.
- the mature microRNA is then loaded into the RNA-induced silencing complex ("RISC"), a ribonucleoprotein complex.
- RISC RNA-induced silencing complex
- the microRNA* also has gene silencing or other activity
- the expression levels of target RNAs measured for stimulation of each of the identified TLRs are expressed as fold-changes in expression relative to expression levels measured in total RNA from human monocytes of healthy donors (see Example 1).
- target RNAs can be measured in samples collected at one or more times from a patient to monitor the status or progress of sepsis in the patient.
- the clinical sample to be tested is obtained from individuals who exhibit one or more symptoms of a systemic inflammatory response, including a body temperature greater than 38° C or less than 36° C, a heart rate greater than 90 beats/minute, a respiratory rate greater than 20 breaths/min (or Paco2 less than 32 mm Hg), and a white blood cell count greater than 12,000 cells/ ⁇ L or less than 4000 cells/ ⁇ L, or with a content of greater than 10% immature forms.
- the clinical sample to be tested is obtained from individuals who exhibit two or more of the above-described symptoms.
- the clinical sample to be tested is obtained from asymptomatic individuals who are at risk for contracting sepsis, such as individuals who are elderly, immuno-compromised, critically ill, or are currently patients in, or have recently been discharged from, a hospital.
- the methods described herein are used for early detection of sepsis in a sample of human cells, such as those obtained by routine blood test.
- the sample of human cells is a sample of human leukocytes.
- the sample of human cells is a sample of human monocytes.
- methods of the present disclosure can be used for routine screening of individuals at risk for sepsis.
- methods herein are used to (1) screen individuals who are elderly, (2) screen individuals who are immuno-compromised, (3) screen individuals who are critically ill or (4) screen individuals who are patients in, or have recently been discharged from, a hospital.
- methods herein are used to screen neonates (less than 90 days old) with fever.
- the methods described herein can be used to determine the source of the underlying infection in a septic individual for targeted treatment of the underlying infection.
- an increase in expression levels of one or more target RNAs associated with the stimulation of TLR2a, TLR2b, TLR4a, TLR4b or TLR5 indicates the presence of a bacterial infection in a septic individual.
- an increase in expression levels of one or more target RNAs associated with the stimulation of TLR4a or TLR4b indicates the presence of an infection of gram-negative bacterial infection in the septic individual.
- an increase in expression levels of one or more target RNAs associated with stimulation of TLR2a, TLR2b or TLR5 without concomitant stimulation of either TLR4a or TLR4b indicates the presence of a gram-positive bacterial infection in the septic individual.
- an increase in expression levels of one or more target RNAs associated with stimulation of TLR2a without concomitant stimulation of TLR4a or TLR4b indicates the presence of either a gram-positive bacterial infection or a mycobacterial infection.
- an increase in expression levels of one or more target RNAs associated with the stimulation of TLR3 or TLR7 indicates the presence of a viral infection.
- an increase in expression levels of one or more target RNAs associated with the stimulation of TLR9 indicates the presence of a viral infection and/or a bacterial infection.
- an increase in expression levels of: (i) one or more target RNAs associated with stimulation of TLR2a, TLR2b, TLR4a, TLR4b or TLR5; and (ii) one or more target RNAs associated with stimulation of TLR3 or TLR7 indicates the presence of both viral and bacterial infection.
- the methods described herein can be used to assess the effectiveness of a treatment for sepsis in a patient.
- the target RNA expression levels are determined at various times during the treatment, and are compared to target RNA expression levels from an archival sample taken from the patient, e.g., by blood test, before the manifestation of any signs of sepsis or before beginning treatment.
- target RNA expression levels in the normal blood sample evidence no aberrant changes in target RNA expression levels.
- the progress of treatment of an individual with sepsis can be assessed by comparison to a sample from the same individual when he was healthy or prior to beginning treatment.
- the sample to be tested is a bodily fluid, such as blood, sputum, mucus, saliva, urine, semen, etc.
- a sample to be tested is a blood sample.
- the blood sample is whole blood, plasma, serum, or blood cells.
- the blood sample is separated monocytes and/or lymphocytes. Monocytes and/or lymphocytes can be separated from whole blood by any method.
- monocytes can be separated from whole blood or a fractionated or separated portion of whole blood using antibodies, e.g., to a cell surface receptor on the monocytes (such as CD 14).
- the antibodies are coupled to beads, such as magnetic beads.
- the clinical sample to be tested is, in some embodiments, freshly obtained. In other embodiments, the sample is a fresh frozen specimen.
- the expression levels of the plurality of target RNAs may be detected concurrently or simultaneously in the same assay reaction. In some embodiments, expression levels are detected concurrently or simultaneously in separate assay reactions. In some embodiments, expression levels are detected at different times, e.g., in serial assay reactions.
- a method comprises detecting the level of at least one target RNA in a sample from a subject, wherein detection of a level of at least one target RNA that is greater than a normal level of the at least one target RNA indicates the presence of sepsis in the subject.
- a method comprises detecting the level of at least one target RNA in a sample from a subject and comparing the level of the at least one target RNA in the sample to a normal level of the at least one target RNA, wherein a level of at least one target RNA in the sample that is greater than a normal level of the at least one target RNA indicates the presence of sepsis in the subject.
- a method of facilitating diagnosis of sepsis in a subject comprises detecting the level of at least one target RNA in a sample from the subject.
- information concerning the level of at least one target RNA in the sample from the subject is communicated to a medical practitioner.
- a "medical practitioner,” as used herein, refers to an individual or entity that diagnoses and/or treats patients, such as a hospital, a clinic, a physician's office, a physician, a nurse, or an agent of any of the aforementioned entities and individuals.
- detecting the level of at least one target RNA is carried out at a laboratory that has received the subject's sample from the medical practitioner or agent of the medical practitioner.
- the laboratory carries out the detection by any method, including those described herein, and then communicates the results to the medical practitioner.
- a result is "communicated," as used herein, when it is provided by any means to the medical practitioner.
- such communication may be oral or written, may be by telephone, in person, by e-mail, by mail or other courier, or may be made by directly depositing the information into, e.g., a database accessible by the medical practitioner, including databases not controlled by the medical practitioner.
- the information is maintained in electronic form.
- the information can be stored in a memory or other computer readable medium, such as RAM, ROM, EEPROM, flash memory, computer chips, digital video discs (DVD), compact discs (CDs), hard disk drives (HDD), magnetic tape, etc.
- a memory or other computer readable medium such as RAM, ROM, EEPROM, flash memory, computer chips, digital video discs (DVD), compact discs (CDs), hard disk drives (HDD), magnetic tape, etc.
- methods of detecting the presence sepsis are provided.
- methods of diagnosing sepsis are provided.
- the method comprises obtaining a sample from a subject and providing the sample to a laboratory for detection of at least one target RNA level in the sample.
- the method further comprises receiving a communication from the laboratory that indicates the at least one target RNA level in the sample.
- sepsis is present if the level of at least one target RNA in the sample is greater than a normal level of the at least one target RNA.
- a "laboratory,” as used herein, is any facility that detects the level of at least one target RNA in a sample by any method, including the methods described herein, and communicates the level to a medical practitioner.
- a laboratory is under the control of a medical practitioner. In some embodiments, a laboratory is not under the control of the medical practitioner.
- a laboratory communicates the level of at least one target RNA to a medical practitioner
- the laboratory communicates a numerical value representing the level of at least one target RNA in the sample, with or without providing a numerical value for a normal level.
- the laboratory communicates the level of at least one target RNA by providing a qualitative value, such as "high,” “elevated,” etc.
- a method when a method relates to detecting sepsis, determining the presence of sepsis, and/or diagnosing sepsis, the method includes activities in which the steps of the method are carried out, but the result is negative for the presence of sepsis. That is, detecting, determining, and diagnosing sepsis include instances of carrying out the methods that result in either positive or negative results (e.g., whether target RNA levels are normal or greater than normal).
- the term "subject" means a human. In some embodiments, the methods described herein may be used on samples from non-human animals.
- Table 3 identifies the chromosomal location of each of the 86 target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86 in Table 2.
- Table 13 identifies the chromosomal location of the target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 226 to 399 in Table 12.
- Table 15 identifies the chromosomal location of the target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 565 to 707 in Table 14.
- Table 17 identifies the chromosomal location of the target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 863 to 897 in Table 16.
- the level of expression of one or more target RNAs located within about 1 kilobase (kb), within about 2 kb, within about 5 kb, within about 10 kb, within about 20 kb, within about 30 kb, within about 40 kb, and even within about 50 kb of the chromosomal locations in Table 2 and Table 14 is detected in lieu of, or in addition to, measurement of expression of the respective tabulated target RNA in the methods described herein. See Baskerville, S. and Bartel D.P. (2005) RNA 11:241-247.
- methods herein in combination with detecting one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 67 and/or detecting one or more target RNAs comprising at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897 and/or detecting one or more target RNAs that comprise a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 67, methods herein further comprise detecting the level(s) of expression of at least one microRNA from the human miRNome.
- At least one target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86.
- at least one target RNA comprises at least 15 contiguous nucleotides that are complementary to at least a portion of a sequence selected from SEQ ID NOs: 1 to 86.
- at least one target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- a target RNA, in its mature form comprises fewer than 30 nucleotides.
- a target RNA is a microRNA.
- more than one target RNA is detected simultaneously in a single reaction. In some embodiments, at least 2, at least 3, at least 5, or at least 10 target RNAs are detected simultaneously in a single reaction. In some embodiments, all target RNAs are detected simultaneously in a single reaction.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 1 to 86 in Table 2 in a sample is indicative of the presence of sepsis in an individual from whom the sample of blood or tissue has been taken.
- an increase in expression of one or more target RNAs that comprise at least 15 contiguous nucleotides that are complementary to at least a portion of a sequence selected from SEQ ID NO: 1 to 86 in Table 2 in a sample is indicative of the presence of sepsis in an individual from whom the sample of blood or tissue has been taken.
- an increase in expression of one or more target RNAs that comprise at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897 in a sample is indicative of the presence of sepsis in an individual from whom the sample of blood or tissue has been taken.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 35, 36, 37, 41, 42, 43, 44, 53, 54, 55, 60, 61, 63, 65, 66, 68, 71, 77, 80, 81, 82, 83 or 85 in Table 2 is indicative of the presence of sepsis caused by viral infection.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 6, 11, 13, 15, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 56, 58, 69, 71, 73, 76, 84 or 86 in Table 2 in a sample of human monocytes is indicative of the presence of sepsis caused by an infection of gram-negative bacteria.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 23, 30, 39, 52, 57, 60 65, 67 or 79 in Table 2 in a sample of human monocytes is indicative of the presence of sepsis caused by an infection of gram positive bacteria.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 8, 14, 59, 62, 63, 64, 74 or 78 in Table 2 in a sample of human monocytes is indicative of the presence of sepsis caused by an infection of gram positive bacteria or mycobacteria.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: SEQ ID NO: 9, 50, 51, 70, 72 or 75 in Table 2 in a sample of human monocytes is indicative of the presence of unmethylated CpG nucleic acids caused by a bacterial and/or a viral infection.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 35, 36, 37, 41, 42, 43, 44, 53, 54, 55, 60, 61, 63, 65, 66, 68, 71, 77, 80, 81, 82, 83 or 85 in Table 2 in a sample of human monocytes is indicative of stimulation of a toll-like receptor that recognizes virally-derived molecules.
- these toll-like receptors are selected from TLR3 and TLR7.
- these toll-like receptors are selected from TLR2a, TLR2b, TLR4a, TLR4b and TLR5.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 6, 11, 13, 15, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 56, 58, 69, 71, 73, 76, 84 or 86 in Table 2 in a sample of human monocytes is indicative of stimulation of a toll-like receptor that recognizes molecules derived from gram-negative bacteria.
- these toll-like receptors are selected from TLR2a, TLR2b, TLR4a, TLR4b and TLR5.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 23, 30, 39, 52, 57, 60 65, 67 or 79 in Table 2 in a sample of human monocytes is indicative of stimulation of a toll-like receptor that recognizes molecules derived from gram-positive bacteria.
- these toll-like receptors are selected from TLR2a, TLR2b and TLR5.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 8, 14, 59, 62, 63, 64, 74 or 78 in Table 2 in a sample of human monocytes is indicative of stimulation of a toll-like receptor that recognizes molecules derived from gram-positive bacteria or mycobacteria, such as TLR2a.
- an increase in expression of one or more target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NO: 9, 50, 51, 70, 72 or 75 in Table 2 in a sample of human monocytes is indicative of stimulation of TLR9, which recognizes unmethylated CpG nucleic acids caused by a bacterial and/or a viral infection.
- an increase in expression of one or more target RNAs comprising at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NOs: 226 to 289, 565 to 604, and 863 to 868 in a sample of human monocytes is indicative of sepsis.
- an increase in expression of one or more target RNAs comprising at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, and 648 in a sample of human monocytes is indicative of sepsis.
- an increase in expression of one or more target RNAs comprising at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, and 632 in a sample of human monocytes is indicative of sepsis.
- an increase in expression of one or more target RNAs comprising at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, and 352 in a sample of human monocytes is indicative of sepsis.
- an increase in expression of one or more target RNAs comprising at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 242, 260, 261, 266, and 287 in a sample of human monocytes is indicative of sepsis.
- a normal level (a "control") for each target RNA can be determined as an average level or range that is characteristic of normal human monocytes or other reference material, against which the level measured in the sample can be compared.
- the determined average or range of target RNA in normal subjects can be used as a benchmark for detecting above-normal or below-normal levels of target RNA indicative of sepsis.
- normal levels of target RNA can be determined using individual or pooled RNA-containing samples from one or more individuals, such as from healthy individuals or from intensive care patients with similar clinical severity of disease (e.g., having matched ICU clinical (APACHE II) scores) to those diagnosed with sepsis syndrome, but without diagnosis of sepsis syndrome.
- determining a normal level of expression of a target RNA comprises detecting a complex comprising a probe hybridized to a nucleic acid selected from a target RNA, a DNA amplicon of the target RNA, and a complement of the target RNA. That is, in some embodiments, a normal level of expression can be determined by detecting a DNA amplicon of the target RNA, or a complement of the target RNA rather than the target RNA itself. In some embodiments, a normal level of such a complex is determined and used as a control. The normal level of the complex, in some embodiments, correlates to the normal level of the target RNA. Thus, when a normal level of a target is discussed herein, that level can, in some embodiments, be determined by detecting such a complex.
- a control comprises RNA from cells of a single individual, e.g., a healthy individual or an intensive care patient with similar clinical severity of disease (e.g., having matched ICU clinical (APACHE II) scores) to a patient being tested for sepsis, but without diagnosis of sepsis syndrome.
- a control comprises RNA from a pool of cells from multiple individuals.
- a control comprises commercially-available human RNA, such as, for example, total RNA from CD 14+ cells.
- a normal level or normal range has already been predetermined prior to testing a sample for an elevated level.
- the normal level of target RNA can be determined from one or more continuous cell lines, typically cell lines previously shown to have expression levels of the at least one target RNA that approximate the level of expression in normal human monocytes.
- a method comprises detecting the level of expression of at least one target RNA. In some embodiments, a method further comprises comparing the level of expression of at least one target RNA to a normal level of expression of the at least one target RNA. In some embodiments, a method further comprises comparing the level of expression of at least one target RNA to a control level of expression of the at least one target RNA.
- a control level of expression of the at least one target RNA is, in some embodiments, the level of expression of the at least one target RNA in a normal cell. In some such embodiments, a control level may be referred to as a normal level.
- a greater level of expression of the at least one target RNA relative to the level of expression of the at least one target RNA in a normal cell indicates sepsis.
- a reduced level of expression of the at least one target RNA relative to the level of expression of the at least one target RNA in a normal cell indicates sepsis.
- the level of expression of the at least one target RNA is compared to a reference level of expression, e.g., from a patient with a confirmed case of sepsis syndrome. In some such embodiments, a similar level of expression of the at least one target RNA relative to the reference sample indicates sepsis.
- a level of expression of at least one target RNA that is at least about two-fold greater than a normal level of expression of the respective at least one target RNA indicates the presence of sepsis. In some embodiments, a level of expression of at least one target RNA that is at least about two-fold greater than the level of the respective at least one target RNA in a control sample comprised of normal cells indicates the presence of a sepsis.
- a level of expression of at least one target RNA that is at least about 3-fold, at least about 4-fold, at least about 5- fold, at least about 6-fold, at least about 7-fold, at least about 8-fold, at least about 9-fold, or at least about 10-fold greater than the level of expression of the respective at least one target RNA in a control sample comprised of normal cells indicates the presence of sepsis.
- a level of expression of at least one target RNA that is at least about 3 -fold, at least about 4-fold, at least about 5 -fold, at least about 6-fold, at least about 7-fold, at least about 8-fold, at least about 9-fold, or at least about 10-fold greater than a normal level of expression of the at least one target RNA indicates the presence of sepsis.
- a level of expression of at least one target RNA that is reduced by at least about two-fold relative to a normal level of expression of the respective at least one target RNA indicates the presence of sepsis.
- a level of expression of at least one target RNA that is reduced by at least about two-fold as compared to the level of the respective at least one target RNA in a control sample comprised of normal cells indicates the presence of a sepsis.
- a level of expression of at least one target RNA that is reduced by at least about 3-fold, at least about 4-fold, at least about 5-fold, at least about 6-fold, at least about 7-fold, at least about 8-fold, at least about 9-fold, or at least about 10-fold as compared to the level of expression of the respective at least one target RNA in a control sample comprised of normal cells indicates the presence of sepsis.
- a level of expression of at least one target RNA that is reduced by at least about 3-fold, at least about 4-fold, at least about 5-fold, at least about 6-fold, at least about 7-fold, at least about 8-fold, at least about 9-fold, or at least about 10-fold as compared to a normal level of expression of the at least one target RNA indicates the presence of sepsis.
- a control level of expression of a target RNA is determined contemporaneously, such as in the same assay or batch of assays, as the level of expression of the target RNA in a sample. In some embodiments, a control level of expression of a target RNA is not determined contemporaneously as the level of expression of the target RNA in a sample. In some such embodiments, the control level of expression has been determined previously.
- the level of expression of a target RNA is not compared to a control level of expression, for example, when it is known that the target RNA is expressed at very low levels, or not at all, in normal cells. In such embodiments, detection of a high level of the target RNA in a sample is indicative of sepsis.
- Target RNA can be prepared by any appropriate method.
- Total RNA can be isolated by any method, including, but not limited to, the protocols set forth in Wilkinson, M. (1988) Nucl. Acids Res. 16(22): 10,933; and Wilkinson, M. (1988) Nucl. Acids Res. 16(22): 10934, or by using commercially-available kits or reagents, such as the TRJzol® reagent (InvitrogenTM), Total RNA Extraction Kit (iNtRON Biotechnology), Total RNA Purification Kit (Norgen Biotek Corp.), RNAqueousTM (Ambion), MagMAXTM (Ambion), RecoverAllTM (Ambion), RNeasy (Qiagen), etc.
- small RNAs are isolated or enriched.
- small RNA refers to RNA molecules smaller than about 200 nucleotides (nt) in length.
- small RNA refers to RNA molecules smaller than about 100 nt, smaller than about 90 nt, smaller than about 80 nt, smaller than about 70 nt, smaller than about 60 nt, smaller than about 50 nt, or smaller than about 40 nt.
- Enrichment of small RNAs can be accomplished by method. Such methods include, but are not limited to, methods involving organic extraction followed by adsorption of nucleic acid molecules on a glass fiber filter using specialized binding and wash solutions, and methods using spin column purification.
- Enrichment of small RNAs may be accomplished using commercially-available kits, such as mirVanaTM Isolation Kit (Applied Biosystems), mirPremierTM microRNA Isolation Kit (Sigma-Aldrich), PureLinkTM miRNA Isolation Kit (Invitrogen), miRCURYTM RNA isolation kit (Exiqon), microRNA Purification Kit (Norgen Biotek Corp.), miRNeasy kit (Qiagen), etc.
- purification can be accomplished by the TRIzol® (Invitrogen) method, which employs a phenol/isothiocyanate solution to which chloroform is added to separate the RNA-containing aqueous phase.
- Small RNAs are subsequently recovered from the aqueous by precipitation with isopropyl alcohol.
- small RNAs can be purified using chromatographic methods, such as gel electrophoresis using the flashPAGETM Fractionator available from Applied Biosystems.
- small RNA is isolated from other RNA molecules to enrich for target RNAs, such that the small RNA fraction (e.g., containing RNA molecules that are 200 nucleotides or less in length, such as less than 100 nucleotides in length, such as less than 50 nucleotides in length, such as from about 10 to about 40 nucleotides in length) is substantially pure, meaning it is at least about 80%, 85%, 90%, 95% pure or more, but less than 100% pure, with respect to larger RNA molecules.
- enrichment of small RNA can be expressed in terms of fold-enrichment.
- small RNA is enriched by about, at least about, or at most about 5X, 10X, 2OX, 3OX, 4OX, 50X, 6OX, 7OX, 80X, 9OX, 10OX, HOX, 120X, 130X, 140X, 150X, 160X, 170X, 180X, 190X, 200X, 210X, 220X, 230X, 240X, 250X, 260X, 270X, 280X, 290X, 300X, 310X, 320X, 330X, 340X, 350X, 360X, 370X, 380X, 390X, 400X, 410X, 420X, 430X, 440X, 450X, 460X, 470X, 480X, 490X, 500X, 600X, 700X, 800X, 900X, 100OX, 1100X, 1200X, 1300X, 1400X, 1500X, 1600X, 1700X, 1800X,
- RNA is modified before target RNAs are detected.
- the modified RNA is total RNA.
- the modified RNA is small RNA that has been purified from total RNA or from cell lysates, such as RNA less than 200 nucleotides in length, such as less than 100 nucleotides in length, such as less than 50 nucleotides in length, such as from about 10 to about 40 nucleotides in length.
- RNA modifications that can be utilized in the methods described herein include, but are not limited to, the addition of a poly-dA or a poly-dT tail, which can be accomplished chemically or enzymatically, and/or the addition of a small molecule, such as biotin.
- one or more target RNAs are reverse transcribed.
- RNA is modified when it is reverse transcribed, such as when a poly-dA or a poly-dT tail is added to the cDNA during reverse transcription.
- RNA is modified before it is reverse transcribed.
- total RNA is reverse transcribed.
- small RNAs are isolated or enriched before the RNA is reverse transcribed.
- a complement of the target RNA is formed.
- the complement of the target RNA is detected rather than the target RNA itself (or a DNA copy thereof).
- detection or determination may be carried out on a complement of the target RNA instead of, or in addition to, the target RNA itself.
- a probe is used that is complementary to the complement of the target RNA.
- the probe comprises at least a portion that is identical in sequence to the target RNA, although it may contain thymidine in place of uridine, and/or comprise other modified nucleotides.
- the method of detecting one or more target RNAs comprises amplifying cDNA complementary to said target RNA.
- amplification can be accomplished by any method. Exemplary methods include, but are not limited to, real time PCR, endpoint PCR, and amplification using T7 polymerase from a T7 promoter annealed to a cDNA, such as provided by the SenseAmp PlusTM Kit available at Implen, Germany.
- a DNA amplicon of a target RNA is formed.
- a DNA amplicon may be single stranded or double-stranded.
- the sequence of the DNA amplicon is related to the target RNA in either the sense or antisense orientation.
- the DNA amplicon of the target RNA is detected rather than the target RNA itself.
- a target RNA when the methods discussed herein indicate that a target RNA is detected, or the level of a target RNA is determined, such detection or determination may be carried out on a DNA amplicon of the target RNA instead of, or in addition to, the target RNA itself.
- a probe when the DNA amplicon of the target RNA is detected rather than the target RNA, a probe is used that is complementary to the complement of the target RNA.
- a probe is used that is complementary to the target RNA.
- multiple probes may be used, and some probes may be complementary to the target RNA and some probes may be complementary to the complement of the target RNA.
- the method of detecting one or more target RNAs comprises RT-PCR, as described below.
- detecting one or more target RNAs comprises real-time monitoring of an RT-PCR reaction, which can be accomplished by any method.
- methods include, but are not limited to, the use of TaqMan®, Molecular beacon, or Scorpion probes (i.e., FRET probes) and the use of intercalating dyes, such as SYBR green, EvaGreen, thiazole orange, YO-PRO, TO-PRO, etc.
- the method comprises detecting a level of expression of at least one target RNA capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86 set forth in Table 2 that is greater in the sample than a normal level of expression of the at least one target RNA in a control sample, such as a sample from a patient that has not been diagnosed with sepsis syndrome, or a sample of normal human monocytes.
- a method comprises detecting a level of one or more target RNAs that comprise a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86 that is greater in the sample than a normal level of expression of the at least one target RNA in a control sample. In some embodiments, a method comprises detecting a level of one or more target RNAs that comprise at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897 that is greater in the sample than a normal level of expression of the at least one target RNA in a control sample. In some embodiments, a target RNA, in its mature form, comprises fewer than 30 nucleotides. In some embodiments, a target RNA is a microRNA.
- the method further comprises detecting a level of expression of at least one target RNA of the human miRNome that does not specifically hybridize to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86 and does not comprise at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897, that is greater in the sample than a normal level of expression of the at least one target RNA in a control sample.
- the term "human miRNome” refers to all microRNA genes in a human cell and the mature microRNAs produced therefrom.
- Any analytical procedure capable of permitting specific and quantifiable (or semi-quantifiable) detection of the desired at least one target RNA may be used in the methods herein presented.
- Such analytical procedures include, but are not limited to, the microarray methods set forth in Example 1 , the microbead methods set forth in Example 2, and methods known to those skilled in the art.
- detection of a target RNA comprises forming a complex comprising a polynucleotide that is complementary to a target RNA or to a complement thereof, and a nucleic acid selected from the target RNA, a DNA amplicon of the target RNA, and a complement of the target RNA.
- the polynucleotide forms a complex with a target RNA.
- the polynucleotide forms a complex with a complement of the target RNA, such as a cDNA that has been reverse transcribed from the target RNA.
- the polynucleotide forms a complex with a DNA amplicon of the target RNA.
- the complex may comprise one or both strands of the DNA amplicon.
- a complex comprises only one strand of the DNA amplicon.
- a complex is a triplex and comprises the polynucleotide and both strands of the DNA amplicon.
- the complex is formed by hybridization between the polynucleotide and the target RNA, complement of the target RNA, or DNA amplicon of the target RNA.
- the polynucleotide in some embodiments, is a primer or probe.
- a method comprises detecting the complex.
- the complex does not have to be associated at the time of detection. That is, in some embodiments, a complex is formed, the complex is then dissociated or destroyed in some manner, and components from the complex are detected.
- An example of such a system is a TaqMan® assay.
- detection of the complex may comprise amplification of the target RNA, a complement of the target RNA, or a DNA amplicon of a target RNA.
- the analytical method used for detecting at least one target RNA in the methods set forth herein includes real-time quantitative RT-PCR. See Chen, C. et al. (2005) Nucl. Acids Res. 33 :e 179 and PCT Publication No. WO 2007/117256, which are incorporated herein by reference in its entirety.
- the analytical method used for detecting at least one target RNA includes the method described in U.S. Publication No. US2009/0123912 Al, which is incorporated herein by reference in its entirety.
- an extension primer comprising a first portion and second portion, wherein the first portion selectively hybridizes to the 3' end of a particular microRNA and the second portion comprises a sequence for universal primer, is used to reverse transcribe the microRNA to make a cDNA.
- a reverse primer that selectively hybridizes to the 5' end of the microRNA and a universal primer are then used to amplify the cDNA in a quantitative PCR reaction.
- the analytical method used for detecting at least one target RNA includes the use of a TaqMan® probe.
- the analytical method used for detecting at least one target RNA includes a TaqMan® assay, such as the TaqMan® MicroRNA Assays sold by Applied Biosystems, Inc.
- a TaqMan® assay such as the TaqMan® MicroRNA Assays sold by Applied Biosystems, Inc.
- total RNA is isolated from the sample.
- the assay can be used to analyze about 10 ng of total RNA input sample, such as about 9 ng of input sample, such as about 8 ng of input sample, such as about 7 ng of input sample, such as about 6 ng of input sample, such as about 5 ng of input sample, such as about 4 ng of input sample, such as about 3 ng of input sample, such as about 2 ng of input sample, and even as little as about 1 ng of input sample containing microRNAs.
- the TaqMan® assay utilizes a stem-loop primer that is specifically complementary to the 3 '-end of a target RNA.
- hybridizing the stem-loop primer to the target RNA is followed by reverse transcription of the target RNA template, resulting in extension of the 3' end of the primer.
- the result of the reverse transcription is a chimeric (DNA) amplicon with the step-loop primer sequence at the 5' end of the amplicon and the cDNA of the target RNA at the 3' end.
- Quantitation of the target RNA is achieved by real time RT-PCR using a universal reverse primer having a sequence that is complementary to a sequence at the 5' end of all stem- loop target RNA primers, a target RNA-specific forward primer, and a target RNA sequence-specific TaqMan® probe.
- the assay uses fluorescence resonance energy transfer ("FRET") to detect and quantitate the synthesized PCR product.
- the TaqMan® probe comprises a fluorescent dye molecule coupled to the 5 '-end and a quencher molecule coupled to the 3 '-end, such that the dye and the quencher are in close proximity, allowing the quencher to suppress the fluorescence signal of the dye via FRET.
- FRET fluorescence resonance energy transfer
- the TaqMan® probe comprises a fluorescent dye molecule coupled to the 5 '-end and a quencher molecule coupled to the 3 '-end, such that the dye and the quencher are in close proximity, allowing the quencher to suppress the fluorescence signal of the dye via FRET.
- the polymerase replicates the chimeric amplicon template to which the TaqMan® probe is bound
- the 5 '-nuclease of the polymerase cleaves the probe, decoupling the dye and the quencher so that FRET is abolished and
- RNA detection and/or quantification are described, e.g., in U.S. Publication No. US 2007/0077570 (Lao et al.), PCT Publication No. WO 2007/025281 (Tan et al.), U.S. Publication No. US2007/0054287 (Bloch), PCT Publication No. WO2006/0130761 (Bloch), and PCT Publication No. WO 2007/011903 (Lao et al.), which are incorporated by reference herein in their entireties for any purpose.
- quantitation of the results of real-time RT-PCR assays is done by constructing a standard curve from a nucleic acid of known concentration and then extrapolating quantitative information for target RNAs of unknown concentration.
- the nucleic acid used for generating a standard curve is an RNA (e.g., microRNA) of known concentration.
- the nucleic acid used for generating a standard curve is a purified double- stranded plasmid DNA or a single-stranded DNA generated in vitro.
- Ct values are inversely proportional to the amount of nucleic acid target in a sample.
- Ct values of the target RNA of interest can be compared with a control or calibrator, such as RNA (e.g., microRNA) from normal tissue.
- the Ct values of the calibrator and the target RNA samples of interest are normalized to an appropriate endogenous housekeeping gene.
- RT-PCR chemistries useful for detecting and quantitating PCR products in the methods presented herein include, but are not limited to, Molecular Beacons, Scorpion probes and intercalating dyes, such as SYBR Green, EvaGreen, thiazole orange, YO-PRO, TO-PRO, etc., which are discussed below.
- real-time RT-PCR detection is performed specifically to detect and quantify the expression of a single target RNA.
- the target RNA in some embodiments, is selected from a target RNA capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86.
- the target RNA specifically hybridizes to a nucleic acid comprising a sequence selected from SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 and 86.
- the target RNA specifically hybridizes to a nucleic acid comprising a sequence selected from SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 and 79. In some embodiments, the target RNA specifically hybridizes to a nucleic acid comprising a sequence selected from SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 and 84. In some embodiments, the target RNA specifically hybridizes to a nucleic acid comprising a sequence selected from SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, and 78.
- the target RNA specifically hybridizes to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 and 85.
- the target RNA specifically hybridizes to a nucleic acid comprising a sequence selected from SEQ ID NOs: 9, 50, 51, 70, 72 and 75.
- the target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- the target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, and 648.
- the target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868. In some embodiments, the target RNA comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86. In some embodiments, a target RNA, in its mature form, comprises fewer than 30 nucleotides. In some embodiments, a target RNA is a microRNA.
- real-time RT-PCR detection is utilized to detect, in a single multiplex reaction, at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, or at least 8 target RNAs.
- At least one target RNA in some embodiments, is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86.
- at least one target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- At least one target RNA comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86.
- a target RNA in its mature form, comprises fewer than 30 nucleotides.
- a target RNA is a microRNA.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least 2, at least 3, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, or at least 40 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 and 86.
- the method comprises detecting greater than normal expression, using a single multiplex RT-PCR reaction, of at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, or at least 15 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 and 79.
- the method comprises detecting greater than normal expression, using a single multiplex RT-PCR reaction, of at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, or at least 15 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 and 84.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, or at least 8 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, and 78.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least 2, at least 3, at least 5, at least 10, at least 15, at least 20, at least 25, or at least 30 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 and 85.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least 2, at least 3, at least 4, at least 5, or at least 6 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 9, 50, 51, 70, 72 and 75.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least two, at least five, at least 10, at least 15, at least 20, at least 25, or at least 30 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, and 648.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least two, at least five, at least 10, at least 15, or at least 20 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, and 632.
- the method comprises detecting expression in a multiplex RT-PCR reaction of at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 70 target RNAs, wherein each target RNA is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 226 to 289, 565 to 604, and 863 to 868.
- a plurality of probes such as TaqMan® probes, each specific for a different RNA target, is used.
- each target RNA-specific probe is spectrally distinguishable from the other probes used in the same multiplex reaction.
- quantitation of real-time RT PCR products is accomplished using a dye that binds to double-stranded DNA products, such as SYBR Green, EvaGreen, thiazole orange, YO-PRO, TO-PRO, etc.
- the assay is the QuantiTect SYBR Green PCR assay from Qiagen. In this assay, total RNA is first isolated from a sample. Total RNA is subsequently poly-adenylated at the 3 '-end and reverse transcribed using- a universal primer with poly-dT at the 5 '-end. In some embodiments, a single reverse transcription reaction is sufficient to assay multiple target RNAs.
- Real-time RT-PCR is then accomplished using target RNA-specific primers and an miScript Universal Primer, which comprises a poly-dT sequence at the 5 '-end.
- SYBR Green dye binds non-specifically to double-stranded DNA and upon excitation, emits light.
- buffer conditions that promote highly-specific annealing of primers to the PCR template e.g., available in the QuantiTect SYBR Green PCR Kit from Qiagen
- the signal from SYBR Green increases, allowing quantitation of specific products.
- Real-time RT-PCR is performed using any RT-PCR instrumentation available in the art.
- instrumentation used in real-time RT-PCR data collection and analysis comprises a thermal cycler, optics for fluorescence excitation and emission collection, and optionally a computer and data acquisition and analysis software.
- the analytical method used in the methods described herein is a DASL® (cDNA-mediated Annealing, Selection, Extension, and Ligation) Assay, such as the MicroRNA Expression Profiling Assay available from Illumina, Inc. (See http://www.illumina.com/downloads/MicroRNAAssayWorkflow.pdf).
- total RNA is isolated from a sample to be analyzed by any method.
- small RNAs are isolated from a sample to be analyzed by any method. Total RNA or isolated small RNAs may then be polyadenylated (> 18 A residues are added to the 3 '-ends of the RNAs in the reaction mixture).
- the RNA is reverse transcribed using a biotin-labeled DNA primer that comprises from the 5' to the 3' end, a sequence that includes a PCR primer site and a poly-dT region that binds to the poly-dA tail of the sample RNA.
- the resulting biotinylated cDNA transcripts are then hybridized to a solid support via a biotin- streptavidin interaction and contacted with one or more target RNA-specific polynucleotides.
- the target RNA-specific polynucleotides comprise, from the 5 '-end to the 3 '-end, a region comprising a PCR primer site, region comprising an address sequence, and a target RNA-specific sequence.
- the target RNA-specific sequence comprises at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides having a sequence identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- the target RNA-specific sequence comprises a probe sequence that is complementary to at least a portion of a microRNA of the human miRNome.
- the target RNA-specific polynucleotide is extended, and the extended products are then eluted from the immobilized cDNA array.
- a second PCR reaction using a fluorescently-labeled universal primer generates a fiuorescently-labeled DNA comprising the target RNA-specific sequence.
- the labeled PCR products are then hybridized to a microbead array for detection and quantitation.
- the analytical method used for detecting and quantifying the expression of the at least one target RNA in the methods described herein is a bead-based flow cytometric assay. See Lu J. et al. (2005) Nature 435:834-838, which is incorporated herein by reference in its entirety.
- An example of a bead-based flow cytometric assay is the xMAP® technology of Luminex, Inc. (See http://www.luminexcorp.com/ technology/index.html).
- total RNA is isolated from a sample and is then labeled with biotin.
- RNA-specific capture probes e.g., FlexmiRTM products sold by Luminex, Inc. at httpV/www.luminexcorp.com/products/assays/index.html
- a streptavidin-bound reporter molecule e.g., streptavidin- phycoerythrin, also known as "SAPE"
- SAPE streptavidin- phycoerythrin
- the RNA sample (total RNA or enriched small RNAs) is first polyadenylated, and is subsequently labeled with a biotinylated 3DNATM dendrimer (i.e., a multiple-arm DNA with numerous biotin molecules bound thereto), such as those sold by Marligen Biosciences as the VantageTM microRNA Labeling Kit, using a bridging polynucleotide that is complementary to the 3 '-end of the poly-dA tail of the sample RNA and to the 5 '-end of the polynucleotide attached to the biotinylated dendrimer.
- a biotinylated 3DNATM dendrimer i.e., a multiple-arm DNA with numerous biotin molecules bound thereto
- a bridging polynucleotide that is complementary to the 3 '-end of the poly-dA tail of the sample RNA and to the 5 '-end of the polynucleotide attached to the bio
- biotin-labeled RNA is first exposed to SAPE, and the RNA/SAPE complex is subsequently exposed to an anti-phycoerythrin antibody attached to a DNA dendrimer, which can be bound to as many as 900 biotin molecules. This allows multiple SAPE molecules to bind to the biotinylated dendrimer through the biotin-streptavidin interaction, thus increasing the signal from the assay.
- the analytical method used for detecting and quantifying the expression of the at least one target RNA in the methods described herein is by gel electrophoresis and detection with labeled probes (e.g., probes labeled with a radioactive or chemiluminescent label), such as by Northern blotting.
- labeled probes e.g., probes labeled with a radioactive or chemiluminescent label
- Northern blotting e.g., total RNA is isolated from the sample, and then is size-separated by SDS polyacrylamide gel electrophoresis. The separated RNA is then blotted onto a membrane and hybridized to radiolabeled complementary probes.
- exemplary probes contain one or more affinity-enhancing nucleotide analogs as discussed below, such as locked nucleic acid (“LNA”) analogs, which contain a bicyclic sugar moiety instead of deoxyribose or ribose sugars.
- LNA locked nucleic acid
- the total RNA sample can be further purified to enrich for small RNAs.
- target RNAs can be amplified by, e.g., rolling circle amplification using a long probe that is complementary to both ends of a target RNA ("padlocked probes"), ligation to circularize the probe followed by rolling circle replication using the target RNA hybridized to the circularized probe as a primer.
- rolling circle amplification using a long probe that is complementary to both ends of a target RNA ("padlocked probes")
- ligation to circularize the probe followed by rolling circle replication using the target RNA hybridized to the circularized probe as a primer.
- the amplified product can then be detected and quantified using, e.g., gel electrophoresis and Northern blotting.
- labeled probes are hybridized to isolated total RNA in solution, after which the RNA is subjected to rapid ribonuclease digestion of single-stranded RNA, e.g., unhybridized portions of the probes or unhybridized target RNAs.
- the ribonuclease treated sample is then analyzed by SDS-PAGE and detection of the radiolabeled probes by, e.g., Northern blotting. See mirVanaTM miRNA Detection Kit sold by Applied Biosystems, Inc. product literature at http://www.ambion.com/catalog/CatNum.php71552.
- the analytical method used for detecting and quantifying the at least one target RNA in the methods described herein is by hybridization to a microarray. See, e.g., Liu, CG. et al. (2004) Proc. Nat'l Acad. Sci. USA 101 :9740-9744; Lim, L.P. et al. (2005) Nature 433:769-773, each of which is incorporated herein by reference in its entirety, and Example 1.
- RNA using a microarray is accomplished by surface plasmon resonance. See, e.g., Nanotech News (2006), available at http://nano.cancer.gov/news_center/nanotech_news_2006- 10-30b.asp.
- total RNA is isolated from a sample being tested.
- the RNA sample is further purified to enrich the population of small RNAs.
- the RNA sample is bound to an addressable microarray containing probes at defined locations on the microarray.
- Nonlimiting exemplary probes include probes comprising sequences set forth in SEQ ID NOs: 1 to 86.
- Exemplary probes also include, but are not limited to, probes comprising a region that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 868 to 897.
- Exemplary probes also include, but are not limited to, probes comprising at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86.
- the probes contain one or more affinity-enhancing nucleotide analogs as discussed below, such as locked nucleic acid (“LNA”) nucleotide analogs.
- LNA locked nucleic acid
- the RNA that is hybridized to the array is first polyadenylated, and the array is then exposed to gold particles having poly-dT bound to them. The amount of bound target RNA is quantitated using surface plasmon resonance.
- microarrays are utilized in a RNA-primed
- RNA 12(2): l-5 each of which is incorporated herein by reference in its entirety.
- total RNA is isolated from a sample.
- small RNAs are isolated from a sample. The RNA sample is then hybridized to DNA probes immobilized at the 5'-end on an addressable array.
- the DNA probes comprise, in some embodiments, from the 5 '-end to the 3 '-end, a first region comprising a "spacer" sequence which is the same for all probes, a second region comprising three thymidine-containing nucleosides, and a third region comprising a sequence that is complementary to a target RNA of interest.
- target RNAs of interest include, but are not limited to, target RNAs capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86, target RNAs comprising a region that is identical to at least 15 contiguous nucleotides of a sequence selected from 196 to 399, 565 to 707, and 863 to 897, and target RNAs comprising a region that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86.
- Target RNAs also include target RNAs in the miRNome that do not specifically hybridize to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86.
- a target RNA in its mature form, comprises fewer than 30 nucleotides.
- a target RNA is a microRNA.
- the sample is hybridized to the array, it is exposed to exonuclease I to digest any unhybridized probes.
- the Klenow fragment of DNA polymerase I is then applied along with biotinylated dATP, allowing the hybridized target RNAs to act as primers for the enzyme with the DNA probe as template.
- the slide is then washed and a streptavidin-conjugated fluorophore is applied to detect and quantitate the spots on the array containing hybridized and Klenow-extended target RNAs from the sample.
- the RNA sample is reverse transcribed.
- the RNA sample is reverse transcribed using a biotin/poly-dA random octamer primer.
- primer When than primer is used, the RNA template is digested and the biotin-containing cDNA is hybridized to an addressable microarray with bound probes that permit specific detection of target RNAs.
- the microarray includes at least one probe comprising at least 8, at least 9, at least 10, at least 11 , at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides identically present in, or complementary to a region of, a sequence selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- the microarray is exposed to a streptavidin-bound detectable marker, such as a fluorescent dye, and the bound cDNA is detected. See Liu CG. et al. (2008) Methods 44:22-30, which is incorporated herein by reference in its entirety.
- target RNAs are detected and quantified in an ELISA-like assay using probes bound in the wells of microtiter plates. See Mora J.R. and Getts R.C. (2006) BioTechniques 41 :420-424 and supplementary material in BioTechniques 41(4):l-5; U.S. Patent Publication No. 2006/0094025 to Getts et al., each of which is incorporated by reference herein in its entirety.
- a sample of RNA that is enriched in small RNAs is either polyadenylated, or is reverse transcribed and the cDNA is polyadenylated.
- RNA or cDNA is hybridized to probes immobilized in the wells of a microtiter plates, wherein each of the probes comprises a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897, or a sequence such as one or more sequences of target RNAs (or the reverse complement thereof) of the human miRNome, depending on whether RNA or cDNA is hybridized to the array.
- the hybridized RNAs are labeled using a capture sequence, such as a DNA dendrimer (such as those available from Genisphere, Inc., http://www.genisphere.com/about 3dna.html) that is labeled with a plurality of biotin molecules or with a plurality of horseradish peroxidase molecules, and a bridging polynucleotide that contains a poly-dT sequence at the 5 '-end that binds to the poly-dA tail of the captured nucleic acid, and a sequence at the 3 '-end that is complementary to a region of the capture sequence.
- a capture sequence such as a DNA dendrimer (such as those available from Genisphere, Inc., http://www.genisphere.com/about 3dna.html) that is labeled with a plurality of biotin molecules or with a plurality of horseradish peroxidase molecules, and a bridging polynucleotide that contains a
- the microarray is then exposed to streptavidin-bound horseradish peroxidase. Hybridization of target RNAs is detected by the addition of a horseradish peroxidase substrate such as tetramethylbenzidine (TMB) and measurement of the absorbance of the solution at 45OnM.
- a horseradish peroxidase substrate such as tetramethylbenzidine (TMB)
- total RNA is isolated from a sample.
- small RNAs are isolated from the sample. The 3 '-ends of the target RNAs are biotinylated using biotin-X- hydrazide.
- the biotinylated target RNAs are captured on a microarray comprising immobilized probes comprising sequences that are identically present in, or complementary to a region of, one or more of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897 and/or probes comprising sequences other than those that are complementary to one or more microRNAs of the human miRNome.
- the hybridized target RNAs are then labeled with quantum dots via a biotin-streptavidin binding.
- a confocal laser causes the quantum dots to fluoresce and the signal can be quantified.
- small RNAs can be detected using a colorimetric assay.
- small RNAs are labeled with streptavidin-conjugated gold followed by silver enhancement.
- the gold nanoparticles bound to the hybridized target RNAs catalyze the reduction of silver ions to metallic silver, which can then be detected colorimetrically with a CCD camera
- target RNAs in a sample of isolated total RNA are hybridized to two probes, one which is complementary to nucleic acids at the 5 '-end of the target RNA and the second which is complementary to the 3 '-end of the target RNA.
- Each probe comprises, in some embodiments, one or more affinity-enhancing nucleotide analogs, such as LNA nucleotide analogs and each is labeled with a different fluorescent dye having different fluorescence emission spectra.
- the sample is then flowed through a microfluidic capillary in which multiple lasers excite the fluorescent probes, such that a unique coincident burst of photons identifies a particular target RNA, and the number of particular unique coincident bursts of photons can be counted to quantify the amount of the target RNA in the sample.
- a microfluidic capillary in which multiple lasers excite the fluorescent probes, such that a unique coincident burst of photons identifies a particular target RNA, and the number of particular unique coincident bursts of photons can be counted to quantify the amount of the target RNA in the sample.
- a target RNA-specific probe can be labeled with 3 or more distinct labels selected from, e.g., fluorophores, electron spin labels, etc., and then hybridized to an RNA sample, such as total RNA, or a sample that is enriched in small RNAs.
- Nonlimiting exemplary target RNA-specific probes include probes comprising sequences selected from of SEQ ID NOs: 1 to 86.
- Nonlimiting exemplary target RNA-specific probes include probes comprising sequences that are complementary to sequences selected from of SEQ ID NOs: 1 to 86.
- Nonlimiting exemplary target RNA-specific probes also include probes comprising at least 15 contiguous nucleotides of, or the complement of at least 15 contiguous nucleotides of, a sequence selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- the sample RNA is modified before hybridization.
- the target RNA/probe duplex is then passed through channels in a microfluidic device and that comprise detectors that record the unique signal of the 3 labels. In this way, individual molecules are detected by their unique signal and counted. See U.S. Patent Nos. 7,402,422 and 7,351,538 to Fuchs et al., U.S. Genomics, Inc., each of which is incorporated herein by reference in its entirety.
- the detection and quantification of one or more target RNAs is accomplished by a solution-based assay, such as a modified Invader assay.
- a solution-based assay such as a modified Invader assay. See Allawi H.T. et al. (2004) RNA 10:1153-1161, which is incorporated herein by reference in its entirety.
- the modified invader assay can be performed on unfractionated detergent lysates of cells.
- the modified invader assay can be performed on total RNA isolated from cells or on a sample enriched in small RNAs. The target RNAs in a sample are annealed to two probes which form hairpin structures.
- a first probe has a hairpin structure at the 5' end and a region at the 3 '-end that has a sequence that is complementary to the sequence of a region at the 5'- end of a target RNA.
- the 3 '-end of the first probe is the "invasive polynucleotide”.
- a second probe has, from the 5' end to the 3 '-end a first "flap" region that is not complementary to the target RNA, a second region that has a sequence that is complementary to the 3 '-end of the target RNA, and a third region that forms a hairpin structure.
- the two probes When the two probes are bound to a target RNA target, they create an overlapping configuration of the probes on the target RNA template, which is recognized by the Cleavase enzyme, which releases the flap of the second probe into solution.
- the flap region then binds to a complementary region at the 3 '-end of a secondary reaction template ("SRT").
- SRT secondary reaction template
- a FRET polynucleotide (having a fluorescent dye bound to the 5 '-end and a quencher that quenches the dye bound closer to the 3' end) binds to a complementary region at the 5 '-end of the SRT, with the result that an overlapping configuration of the 3 '-end of the flap and the 5 '-end of the FRET polynucleotide is created.
- Cleavase recognizes the overlapping configuration and cleaves the 5 '-end of the FRET polynucleotide, generates a fluorescent signal when the dye is released into solution.
- polynucleotides are provided.
- synthetic polynucleotides are provided.
- Synthetic polynucleotides refer to polynucleotides that have been synthesized in vitro either chemically or enzymatically.
- Chemical synthesis of polynucleotides includes, but is not limited to, synthesis using polynucleotide synthesizers, such as OligoPilot (GE Healthcare), ABI 3900 DNA Synthesizer (Applied Biosystems), and the like.
- Enzymatic synthesis includes, but is not limited, to producing polynucleotides by enzymatic amplification, e.g., PCR.
- a polynucleotide comprises at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897 and sequences complementary to SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- the polynucleotide further comprises a region having a sequence that is not found in, or complementary to, any of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- a polynucleotide comprises at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 67, 215 to 399, and 863 to 897, and sequences complementary to SEQ ID NOs: 1 to 67, 215 to 399, and 863 to 897.
- the polynucleotide further comprises a region having a sequence that is not found in, or complementary to, any of SEQ ID NOs: 1 to 67, 215 to 399, or 863 to 897.
- a "region" can comprise the full-length sequence, or the complement of the full-length sequence, of a particular sequence, such as any of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897, or it can comprise a subsequence, or the complement of a subsequence, of a particular sequence, such as any of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- Such subsequences may comprise, in some embodiments, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or more contiguous nucleotides from a particular SEQ ID NO or its complement.
- a polynucleotide comprises fewer than
- a polynucleotide is between 8 and 200, between 8 and 150, between 8 and 100, between 8 and 75, between 8 and 50, between 8 and 40, or between 8 and 30 nucleotides long.
- the polynucleotide is a primer.
- the primer is labeled with a detectable moiety.
- a primer is not labeled.
- a primer is a polynucleotide that is capable of specifically hybridizing to a target RNA or to a cDNA reverse transcribed from the target RNA or to an amplicon that has been amplified from a target RNA or a cDNA (collectively referred to as "template"), and, in the presence of the template, a polymerase and suitable buffers and reagents, can be extended to form a primer extension product.
- the polynucleotide is a probe.
- the probe is labeled with a detectable moiety.
- a detectable moiety includes both directly detectable moieties, such as fluorescent dyes, and indirectly detectable moieties, such as members of binding pairs. When the detectable moiety is a member of a binding pair, in some embodiments, the probe can be detectable by incubating the probe with a detectable label bound to the second member of the binding pair.
- a probe is not labeled, such as when a probe is a capture probe, e.g., on a microarray or bead.
- a probe is not extendable, e.g., by a polymerase. In other embodiments, a probe is extendable.
- the polynucleotide is a FRET probe that in some embodiments is labeled at the 5 '-end with a fluorescent dye (donor) and at the 3'- end with a quencher (acceptor), a chemical group that absorbs (i.e., suppresses) fluorescence emission from the dye when the groups are in close proximity (i.e., attached to the same probe).
- the donor and acceptor are not at the ends of the FRET probe.
- the emission spectrum of the donor moiety should overlap considerably with the absorption spectrum of the acceptor moiety.
- RNA described herein employ one or more polynucleotides that have been modified, such as polynucleotides comprising one or more affinity-enhancing nucleotide analogs.
- Modified polynucleotides useful in the methods described herein include primers for reverse transcription, PCR amplification primers, and probes.
- the incorporation of affinity-enhancing nucleotides increases the binding affinity and specificity of a polynucleotide for its target nucleic acid as compared to polynucleotides that contain only deoxyribonucleotides, and allows for the use of shorter polynucleotides or for shorter regions of complementarity between the polynucleotide and the target nucleic acid.
- affinity-enhancing nucleotide analogs include nucleotides comprising one or more base modifications, sugar modifications and/or backbone modifications.
- modified bases for use in affinity-enhancing nucleotide analogs include 5 -methyl cytosine, isocytosine, pseudoisocytosine, 5- bromouracil, 5-propynyluracil, 6-aminopurine, 2-aminopurine, inosine, diaminopurine, 2- chloro-6-aminopurine, xanthine and hypoxanthine.
- affinity-enhancing nucleotide analogs include nucleotides having modified sugars such as 2 '-substituted sugars, such as 2'-O- alkyl-ribose sugars, 2'-amino-deoxyribose sugars, 2'-fluoro- deoxyribose sugars, T- fluoro-arabinose sugars, and 2'-O-methoxyethyl-ribose (2'MOE) sugars.
- modified sugars are arabinose sugars, or d-arabino-hexitol sugars.
- affinity-enhancing nucleotide analogs include backbone modifications such as the use of peptide nucleic acids (PNA; e.g., an oligomer including nucleobases linked together by an amino acid backbone).
- PNA peptide nucleic acids
- backbone modifications include phosphorothioate linkages, phosphodiester modified nucleic acids, combinations of phosphodiester and phosphorothioate nucleic acid, methylphosphonate, alkylphosphonates, phosphate esters, alkylphosphonothioates, phosphoramidates, carbamates, carbonates, phosphate triesters, acetamidates, carboxymethyl esters, methylphosphorothioate, phosphorodithioate, p-ethoxy, and combinations thereof.
- a polynucleotide includes at least one affinity-enhancing nucleotide analog that has a modified base, at least nucleotide (which may be the same nucleotide) that has a modified sugar, and/or at least one internucleotide linkage that is non-naturally occurring.
- an affinity-enhancing nucleotide analog contains a locked nucleic acid ("LNA") sugar, which is a bicyclic sugar.
- a polynucleotide for use in the methods described herein comprises one or more nucleotides having an LNA sugar.
- a polynucleotide contains one or more regions consisting of nucleotides with LNA sugars.
- a polynucleotide contains nucleotides with LNA sugars interspersed with deoxyribonucleotides. See, e.g., Frieden, M. et al. (2008) Curr. Pharm. Des. 14(11): 1138- 1142.
- a primer is provided.
- a primer is identical or complementary to at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of a target RNA.
- a primer may also comprise portions or regions that are not identical or complementary to the target RNA.
- a region of a primer that is identical or complementary to a target RNA is contiguous, such that any region of a primer that is not identical or complementary to the target RNA does not disrupt the identical or complementary region.
- a primer comprises a portion that is identically present in a target RNA.
- a primer that comprises a region that is identically present in the target RNA is capable of selectively hybridizing to a cDNA that has been reverse transcribed from the RNA, or to an amplicon that has been produced by amplification of the target RNA or cDNA.
- the prime zxr is complementary to a sufficient portion of the cDNA or amplicon such that it selectively hybridizes to the cDNA or amplicon under the conditions of the particular assay being used.
- “selectively hybridize” means that a polynucleotide, such as a primer or probe, will hybridize to a particular nucleic acid in a sample with at least 5-fold greater affinity than it will hybridize to another nucleic acid present in the same sample that has a different nucleotide sequence in the hybridizing region. Exemplary hybridization conditions are discussed in Example 1. In some embodiments, a polynucleotide will hybridize to a particular nucleic acid in a sample with at least 10-fold greater affinity than it will hybridize to another nucleic acid present in the same sample that has a different nucleotide sequence in the hybridizing region.
- Nonlimiting exemplary primers include primers comprising sequences that are identically present in, or complementary to a region of, sequences selected from SEQ ID NOs: 1 to 86.
- Exemplary primers also include, but are not limited to, primers comprising regions that are identical or complementary to at least 15 contiguous nucleotides of sequences selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- a primer is used to reverse transcribe a target RNA, for example, as discussed herein.
- a primer is used to amplify a target RNA or a cDNA reverse transcribed therefrom. Such amplification, in some embodiments, is quantitative PCR, for example, as discussed herein.
- a primer comprises a detectable moiety.
- methods of detecting the presence of a sepsis comprise hybridizing nucleic acids of a human sample with a probe.
- the probe comprises a portion that is complementary to a target RNA.
- the probe comprises a portion that is identically present in the target RNA.
- a probe that is complementary to a target RNA is complementary to a sufficient portion of the target RNA such that it selectively hybridizes to the target RNA under the conditions of the particular assay being used.
- a probe that is complementary to a target RNA is complementary to at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of the target RNA.
- a probe that is complementary to a target RNA comprises a region that is complementary to at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of the target RNA.
- a probe that is complementary to a target RNA may also comprise portions or regions that are not complementary to the target RNA.
- a region of a probe that is complementary to a target RNA is contiguous, such that any region of a probe that is not complementary to the target RNA does not disrupt the complementary region.
- the probe comprises a portion that is identically present in the target RNA.
- a probe that comprises a region that is identically present in the target RNA is capable of selectively hybridizing to a cDNA that has been reverse transcribed from the RNA, or to an amplicon that has been produced by amplification of the target RNA or cDNA.
- the probe is complementary to a sufficient portion of the cDNA or amplicon such that it selectively hybridizes to the cDNA or amplicon under the conditions of the particular assay being used.
- a probe that is complementary to a cDNA or amplicon is complementary to at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of the cDNA or amplicon.
- a probe that is complementary to a target RNA comprises a region that is complementary to at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides of the cDNA or amplicon.
- a probe that is complementary to a cDNA or amplicon may also comprise portions or regions that are not complementary to the cDNA or amplicon.
- a region of a probe that is complementary to a cDNA or amplicon is contiguous, such that any region of a probe that is not complementary to the cDNA or amplicon does not disrupt the complementary region.
- Nonlimiting exemplary probes include probes comprising sequences set forth in SEQ ID NOs: 1 to 86.
- Nonlimiting exemplary probes include probes comprising sequences that are identically present in, or complementary to a region of, sequences selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- Exemplary probes also include, but are not limited to, probes comprising regions that are identical or complementary to at least 15 contiguous nucleotides of sequences selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- the method of detectably quantifying one or more target RNAs comprises: (a) isolating total RNA; (b) reverse transcribing a target RNA to produce a cDNA that is complementary to the target RNA; (c) amplifying the cDNA from (b); and (d) detecting the amount of a target RNA using real time RT-PCR and a detection probe.
- the real time RT-PCR detection is performed using a FRET probe, which includes, but is not limited to, a TaqMan® probe, a Molecular beacon probe and a Scorpion probe.
- a FRET probe which includes, but is not limited to, a TaqMan® probe, a Molecular beacon probe and a Scorpion probe.
- the real time RT-PCR detection and quantification is performed with a TaqMan® probe, i.e., a linear probe that typically has a fluorescent dye covalently bound at one end of the DNA and a quencher molecule covalently bound at the other end of the DNA.
- the FRET probe comprises a sequence that is complementary to a region of the cDNA such that, when the FRET probe is hybridized to the cDNA, the dye fluorescence is quenched, and when the probe is digested during amplification of the cDNA, the dye is released from the probe and produces a fluorescence signal.
- the amount of target RNA in the sample is proportional to the amount of fluorescence measured during cDNA amplification.
- the TaqMan® probe typically comprises a region of contiguous nucleotides having a sequence that is complementary to a region of a target RNA or its complementary cDNA that is reverse transcribed from the target RNA template (i.e., the sequence of the probe region is complementary to or identically present in the target RNA to be detected) such that the probe is specifically hybridizable to the resulting PCR amplicon.
- the probe comprises a region of at least 6 contiguous nucleotides having a sequence that is fully complementary to or identically present in a region of a cDNA that has been reverse transcribed from a target RNA template, such as comprising a region of at least 8 contiguous nucleotides, at least 10 contiguous nucleotides, at least 12 contiguous nucleotides, at least 14 contiguous nucleotides, or at least 16 contiguous nucleotides having a sequence that is complementary to or identically present in a region of a cDNA reverse transcribed from a target RNA to be detected.
- the region of the cDNA that has a sequence that is complementary to the TaqMan® probe sequence is at or near the center of the cDNA molecule.
- Molecular Beacons can be used to detect and quantitate PCR products. Like TaqMan® probes, Molecular Beacons use FRET to detect and quantitate a PCR product via a probe having a fluorescent dye and a quencher attached at the ends of the probe. Unlike TaqMan® probes, Molecular Beacons remain intact during the PCR cycles. Molecular Beacon probes form a stem-loop structure when free in solution, thereby allowing the dye and quencher to be in close enough proximity to cause fluorescence quenching. When the Molecular Beacon hybridizes to a target, the stem-loop structure is abolished so that the dye and the quencher become separated in space and the dye fluoresces. Molecular Beacons are available, e.g., from Gene LinkTM (see http://www.genelink.com/newsite/products/mbintro.asp).
- Scorpion probes can be used as both sequence-specific primers and for PCR product detection and quantitation. Like Molecular Beacons, Scorpion probes form a stem-loop structure when not hybridized to a target nucleic acid. However, unlike Molecular Beacons, a Scorpion probe achieves both sequence-specific priming and PCR product detection. A fluorescent dye molecule is attached to the 5 '-end of the Scorpion probe, and a quencher is attached to the 3 '-end. The 3' portion of the probe is complementary to the extension product of the PCR primer, and this complementary portion is linked to the 5 '-end of the probe by a non-amplifiable moiety.
- Scorpion probes are available from, e.g, Premier Biosoft International (see http://www.premierbiosofit.com/tech_notes/Sco ⁇ ion.html).
- labels that can be used on the FRET probes include colorimetric and fluorescent labels such as Alexa Fluor dyes, BODIPY dyes, such as BODIPY FL; Cascade Blue; Cascade Yellow; coumarin and its derivatives, such as 7- amino-4-methylcoumarin, aminocoumarin and hydroxycoumarin; cyanine dyes, such as Cy3 and Cy5; eosins and erythrosins; fluorescein and its derivatives, such as fluorescein isothiocyanate; macrocyclic chelates of lanthanide ions, such as Quantum DyeTM; Marina Blue; Oregon Green; rhodamine dyes, such as rhodamine red, tetramethylrhodamine and rhodamine 6G; Texas Red; fluorescent energy transfer dyes, such as thiazole orange- ethidium heterodimer; and, TOTAB.
- Alexa Fluor dyes such as Alexa Fluor dyes, BODIPY dyes, such
- dyes include, but are not limited to, those identified above and the following: Alexa Fluor 350, Alexa Fluor 405, Alexa Fluor 430, Alexa Fluor 488, Alexa Fluor 500. Alexa Fluor 514, Alexa Fluor 532, Alexa Fluor 546, Alexa Fluor 555, Alexa Fluor 568, Alexa Fluor 594, Alexa Fluor 610, Alexa Fluor 633, Alexa Fluor 647, Alexa Fluor 660, Alexa Fluor 680, Alexa Fluor 700, and, Alexa Fluor 750; amine-reactive BODIPY dyes, such as BODIPY 493/503, BODIPY 530/550, BODIPY 558/568, BODIPY 564/570, BODIPY 576/589, BODIPY 581/591, BODIPY 630/650, BODIPY 650/655, BODIPY FL, BODIPY R6G, BODIPY TMR, and
- fluorescently labeled ribonucleotides useful in the preparation of RT-PCR probes for use in some embodiments of the methods described herein are available from Molecular Probes (Invitrogen), and these include, Alexa Fluor 488-5-UTP, Fluorescein- 12-UTP, BODIPY FL-14-UTP, BODIPY TMR-14- UTP, Tetramethylrhodamine-6-UTP, Alexa Fluor 546-14-UTP, Texas Red-5-UTP, and BODIPY TR-14-UTP.
- Other fluorescent ribonucleotides are available from Amersham Biosciences (GE Healthcare), such as Cy3-UTP and Cy5-UTP.
- Examples of fluorescently labeled deoxyribonucleotides useful in the preparation of RT-PCR probes for use in the methods described herein include Dinitrophenyl (DNP)-l'-dUTP, Cascade Blue-7-dUTP, Alexa Fluor 488-5-dUTP, Fluorescein- 12-dUTP, Oregon Green 488-5-dUTP, BODIPY FL-14-dUTP, Rhodamine Green-5-dUTP, Alexa Fluor 532-5-dUTP, BODIPY TMR-14-dUTP, Tetramethylrhodamine-6-dUTP, Alexa Fluor 546-14-dUTP, Alexa Fluor 568-5-dUTP, Texas Red-12-dUTP, Texas Red-5-dUTP, BODIPY TR-14-dUTP, Alexa Fluor 594-5- dUTP, BODIPY 630/650- 14-dUTP, BODIPY 650/665-14-dUTP; Alexa
- dyes and other moieties are introduced into polynucleotide used in the methods described herein, such as FRET probes, via modified nucleotides.
- a "modified nucleotide” refers to a nucleotide that has been chemically modified, but still functions as a nucleotide.
- the modified nucleotide has a chemical moiety, such as a dye or quencher, covalently attached, and can be introduced into a polynucleotide, for example, by way of solid phase synthesis of the polynucleotide.
- the modified nucleotide includes one or more reactive groups that can react with a dye or quencher before, during, or after incorporation of the modified nucleotide into the nucleic acid.
- the modified nucleotide is an amine-modified nucleotide, i.e., a nucleotide that has been modified to have a reactive amine group.
- the modified nucleotide comprises a modified base moiety, such as uridine, adenosine, guanosine, and/or cytosine.
- the amine-modified nucleotide is selected from 5-(3- aminoallyl)-UTP; 8-[(4-amino)butyl]-amino-ATP and 8-[(6-amino)butyl]-amino-ATP; N6-(4-amino)butyl-ATP, N6-(6-amino)butyl-ATP, N4-[2,2-oxy-bis-(ethylamine)]-CTP; N6-(6-Amino)hexyl-ATP; 8-[(6-Amino)hexyl]-amino-ATP; 5-propargylamino-CTP, 5- propargylamino-UTP.
- nucleotides with different nucleobase moieties are similarly modified, for example, 5-(3-aminoallyl)-GTP instead of 5-(3- aminoallyl)-UTP.
- Many amine modified nucleotides are commercially available from, e.g., Applied Biosystems, Sigma, Jena Bioscience and TriLink.
- Exemplary detectable moieties also include, but are not limited to, members of binding pairs. In some such embodiments, a first member of a binding pair is linked to a polynucleotide. The second member of the binding pair is linked to a detectable label, such as a fluorescent label.
- binding pairs include, but are not limited to, biotin and streptavidin, antibodies and antigens, etc.
- each probe that is targeted to a unique cDNA is spectrally distinguishable when released from the probe.
- each target RNA is detected by a unique fluorescence signal.
- One skilled in the art can select a suitable detection method for a selected assay, e.g., a real-time RT-PCR assay.
- the selected detection method need not be a method described above, and may be any method.
- compositions are provided.
- compositions are provided for use in the methods described herein.
- a composition comprises at least one polynucleotide. In some embodiments, a composition comprises at least one primer. In some embodiments, a composition comprises at least one probe. In some embodiments, a composition comprises at least one primer and at least one probe.
- compositions that comprise at least one target RNA-specific primer.
- target RNA-specific primer encompasses primers that have a region of contiguous nucleotides having a sequence that is (i) identically present in one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897, or (ii) complementary to the sequence of a region of contiguous nucleotides found in one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- compositions that comprise at least one target RNA-specific probe.
- target RNA-specific probe encompasses probes that have a region of contiguous nucleotides having a sequence that is (i) identically present in one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897, or (ii) complementary to the sequence of a region of contiguous nucleotides found in one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- target RNA-specific primers and probes comprise deoxyribonucleotides.
- target RNA-specific primers and probes comprise at least one nucleotide analog.
- Nonlimiting exemplary nucleotide analogs include, but are not limited to, analogs described herein, including LNA analogs and peptide nucleic acid (PNA) analogs.
- target RNA-specific primers and probes comprise at least one nucleotide analog which increases the hybridization binding energy (e.g., an affinity-enhancing nucleotide analog, discussed above).
- a target RNA-specific primer or probe in the compositions described herein binds to one target RNA in the sample.
- a single primer or probe binds to multiple target RNAs, such as multiple isomirs.
- more than one primer or probe specific for a single target RNA is present in the compositions, the primers or probes capable of binding to overlapping or spatially separated regions of the target RNA.
- the composition comprises at least one target RNA-specific primer or probe (or region thereof) having a sequence that is identically present in a target RNA (or region thereof).
- a target RNA is capable of specifically hybridizing to at least one probe comprising a sequence selected from SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 and 86.
- a target RNA is capable of specifically hybridizing to at least one nucleic acid probe comprising a sequence selected from SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 and 79. In some embodiments, a target RNA is capable of specifically hybridizing to at least one nucleic acid probe comprising a sequence selected from SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 and 84.
- a target RNA is capable of specifically hybridizing to at least one nucleic acid probe comprising a sequence selected from SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, and 78. In some embodiments, a target RNA is capable of specifically hybridizing to at least one nucleic acid probe comprising a sequence selected from SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 and 85.
- a target RNA is capable of specifically hybridizing to at least one nucleic acid probe comprising a sequence selected from SEQ ID NOs: 9, 50, 51, 70, 72 and 75. In some embodiments, a target RNA is capable of specifically hybridizing to at least one probe comprising a sequence selected from SEQ ID NOs: 1 to 86. In some embodiments, a target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- a target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, and 648.
- a target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, and 632.
- a target RNA comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 226 to 289, 565 to 604, and 863 to 868.
- a target RNA comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86. In some embodiments, a target RNA, in its mature form, comprises fewer than 30 nucleotides. In some embodiments, a target RNA is a microRNA.
- the composition comprises a plurality of target RNA-specific primers and/or probes for each of at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, or at least 8 target RNAs, the target RNAs comprising a region of contiguous nucleotides having a sequence that is identically present in one of SEQ ID NOs: 87 to 177, 400 to 564, 708 to 862, and 898 to 932.
- the plurality includes a target RNA-specific primer and/or probe specific for each of at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 target RNAs, the target RNAs comprising a region of contiguous nucleotides having a sequence that is identically present in one of SEQ ID NOs: 87 to 177, 400 to 564, 708 to 862, and 898 to 932.
- the plurality includes a target RNA-specific primer and/or probe specific for each of at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 75, or at least 100 target RNAs comprising a region of contiguous nucleotides having a sequence that is identically present in one of SEQ ID NOs: 87 to 177, 400 to 564, 708 to 862, and 898 to 932.
- target RNAs described herein comprise a sequence identically present in a sequence set forth in Table 3, 13, 15, or 17, except that thymine (T) bases in the sequences shown in Table 3, 13, 15, or 17 are replaced by uracil (U) bases in the target RNAs.
- a composition is an aqueous composition.
- the aqueous composition comprises a buffering component, such as phosphate, tris, HEPES, etc., and/or additional components, as discussed below.
- a composition is dry, for example, lyophilized, and suitable for reconstitution by addition of fluid.
- a dry composition may include a buffering component and/or additional components.
- a composition comprises one or more additional components.
- Additional components include, but are not limited to, salts, such as NaCl , KCl, and MgCl 2 ; polymerases, including thermostable polymerases; dNTPs; RNase inhibitors; bovine serum albumin (BSA) and the like; reducing agents, such as ⁇ - mercaptoethanol; EDTA and the like; etc.
- salts such as NaCl , KCl, and MgCl 2
- polymerases including thermostable polymerases
- dNTPs including RNase inhibitors
- BSA bovine serum albumin
- reducing agents such as ⁇ - mercaptoethanol
- EDTA and the like
- an addressable microarray component comprises target RNA-specific probes attached to a substrate.
- Microarrays for use in the methods described herein comprise a solid substrate onto which the probes are covalently or non-covalently attached.
- probes capable of hybridizing to one or more target RNAs or cDNAs are attached to the substrate at a defined location ("addressable array").
- Probes can be attached to the substrate in a wide variety of ways, as will be appreciated by those in the art.
- the probes are synthesized first and subsequently attached to the substrate.
- the probes are synthesized on the substrate.
- probes are synthesized on the substrate surface using techniques such as photopolymerization and photolithography.
- the solid substrate is a material that is modified to contain discrete individual sites appropriate for the attachment or association of the probes and is amenable to at least one detection method.
- substrates include glass and modified or functionalized glass, plastics (including acrylics, polystyrene and copolymers of styrene and other materials, polypropylene, polyethylene, polybutylene, polyurethanes, TeflonJ, etc.), polysaccharides, nylon or nitrocellulose, resins, silica or silica-based materials including silicon and modified silicon, carbon, metals, inorganic glasses and plastics.
- the substrates allow optical detection without appreciably fluorescing.
- the substrate is planar.
- probes are placed on the inside surface of a tube, such as for flow-through sample analysis to minimize sample volume.
- probes can be in the wells of multi-well plates.
- probes can be attached to an addressable microbead array.
- the probes can be attached to a flexible substrate, such as a flexible foam, including closed cell foams made of particular plastics.
- the substrate and the probe can each be derivatized with functional groups for subsequent attachment of the two.
- the substrate is derivatized with one or more chemical functional groups including, but not limited to, amino groups, carboxyl groups, oxo groups and thiol groups.
- probes are attached directly to the substrate through one or more functional groups.
- probes are attached to the substrate indirectly through a linker (i.e., a region of contiguous nucleotides that space the probe regions involved in hybridization and detection away from the substrate surface).
- probes are attached to the solid support through the 5' terminus. In other embodiments, probes are attached through the 3' terminus.
- probes are attached to the substrate through an internal nucleotide.
- the probe is attached to the solid support non-covalently, e.g., via a biotin-streptavidin interaction, wherein the probe biotinylated and the substrate surface is covalently coated with streptavidin.
- the compositions comprise a microarray having probes attached to a substrate, wherein at least one of the probes (or a region thereof) comprises a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- at least 2, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, or at least 100 of the probes comprise a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the microarray comprises at least one target RNA-specific probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897 and at least one target RNA-specific probe comprising a sequence that is identically present in, or complementary to a region of, a target RNA of the human miRNome.
- the microarray comprises each target RNA-specific probe at only one location on the microarray.
- the microarray comprises at least one target RNA-specific probe at multiple locations on the microarray.
- the terms “complementary” or “partially complementary” to a target RNA (or target region thereof), and the percentage of “complementarity” of the probe sequence to that of the target RNA sequence is the percentage “identity” to the reverse complement of the sequence of the target RNA.
- the degree of “complementarity” is expressed as the percentage identity between the sequence of the probe (or region thereof) and the reverse complement of the sequence of the target RNA that best aligns therewith.
- the microarray comprises at least one probe having a region with a sequence that is fully complementary to a target region of a target RNA. In other embodiments, the microarray comprises at least one probe having a region with a sequence that comprises one or more base mismatches when compared to the sequence of the best-aligned target region of a target RNA.
- a "region" of a probe or target RNA may comprise or consist of 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29 or more contiguous nucleotides from a particular SEQ ID NO or the complement thereof.
- the region is of the same length as the probe or the target RNA. In other embodiments, the region is shorter than the length of the probe or the target RNA.
- the microarray comprises at least one probe having a region of at least 10, at least 11, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, or at least 25 contiguous nucleotides with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 68, 196 to 399, 565 to 707, or 863 to 897.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86.
- the microarray comprises at least one, at least two, at least three, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, or at lest 40 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 and 86.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 or 79.
- the microarray comprises at least one, at least two, at least three, at least five, at least eight, at least 10, at least 12, or at least 15 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 and 79.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 and 79.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- the microarray comprises at least one, at least two, at least three, at least five, at least eight, at least 10, at least 12, or at least 15 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- the microarray comprises at least one, at least two, at least three, at least four, at least five, at least six, at least seven, or at least eight probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the microarray comprises at least one, at least two, at least three, at least four, at least five, at least ten, at least 15, at least 20, at least 25, or at least 30 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 9, 50, 51, 70, 72 or 75. In some embodiments, the microarray comprises at least one, at least two, at least three, at least four, at least five, or at least six probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 9, 50, 51, 70, 72 or 75. In some embodiments, the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 9, 50, 51, 70, 72 or 75.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- the microarray comprises at least two, at least five, at least 10, at least 15, at least 20, at least 25, or at least 30 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- the microarray comprises at least two, at least five, at least 10, at least 15, or at least 20 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- the microarray comprises at least one probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- the microarray comprises at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 70 probes that each comprise a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- the microarray further comprises additional probes that do not have a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- the microarrays comprise probes having a region with a sequence that is complementary to target RNAs that comprise a substantial portion of the human miRNome (i.e., the publicly known microRNAs that have been accessioned by others into miRBase (http://microrna.sanger.ac.uk/ at the time the microarray is fabricated), such as at least about 60%, at least about 70%, at least about 80%, at least about 90%, or at least about 95% of the human miRNome.
- the human miRNome i.e., the publicly known microRNAs that have been accessioned by others into miRBase (http://microrna.sanger.ac.uk/ at the time the microarray is fabricated
- the microarrays comprise probes that have a region with a sequence that is identically present in target RNAs that comprise a substantial portion of the human miRNome, such as at least about 60%, at least about 70%, at least about 80%, at least about 90%, or at least about 95% of the human miRNome.
- components are provided that comprise probes attached to microbeads, such as those sold by Luminex, each of which is internally dyed with red and infrared fluorophores at different intensities to create a unique signal for each bead.
- the compositions useful for carrying out the methods described herein include a plurality of microbeads, each with a unique spectral signature. Each uniquely labeled microbead is attached to a unique target RNA-specific probe such that the unique spectral signature from the dyes in the bead is associated with a particular probe sequence.
- Nonlimiting exemplary probe sequences include SEQ ID NOs: 1 to 86.
- Nonlimiting exemplary probe sequences also include probes comprising a region that is identically present in, or complementary to, a sequence selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- a probe sequence comprises at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, or at least 24 contiguous nucleotides that are identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- a uniquely labeled microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the uniquely labeled microbead has attached thereto a probe having a region with a sequence that comprises one or more base mismatches when compared to the most similar sequence selected from SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897, and sequences complementary to SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, and 863 to 897.
- a composition comprises a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region of at least 10, at least 11, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, or at least 25 contiguous nucleotides with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the compositions comprise a plurality of uniquely labeled microbeads, at least one of which has attached thereto a target RNA- specific probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86.
- the compositions comprise at least two, at least three, at least five, at least 8, at least ten, at least 15, at least 20, at least 25, at least 30, at least 35, or at least 40 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86
- compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 or 79.
- compositions comprise at least two, at least three, at least five, at least eight, at least 10, at least 12, at least 15, or at least 18 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 or 79.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 or 79.
- compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- compositions comprise at least two, at least three, at least five, at least eight, at least 10, at least 12, at least 15, or at least 18 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- compositions comprise at least two, at least three, at least four, at least five, at least six, at least seven, or at least eight uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- the compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- compositions comprise at least two, at least three, at least five, at least 8, at least ten, at least 15, at least 20, at least 25, at least 30, or at least 35 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 9, 50, 51, 70, 72 or 75.
- the compositions comprise at least two, at least three, at least four, at least five, or at least six uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 9, 50, 51 , 70, 72 or 75.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 9, 50, 51, 70, 72 or 75.
- the compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- compositions comprise at least two, at least five, at least 10, at least 15, at least 20, at least 25, or at least 30 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566,
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- the compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- compositions comprise at least two, at least five, at least 10, at least 15, or at least 20 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567,
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 231 , 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- compositions comprise a plurality of uniquely labeled microbeads, wherein at least one microbead has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- compositions comprise at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 70 uniquely labeled microbeads that each have attached thereto a unique target RNA-specific probe having a region with a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- the composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- compositions comprise a plurality of uniquely labeled microbeads, wherein the plurality comprises at least one microbead having attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the plurality comprises at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, at least 75, or at least 100 microbeads each of which having attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- a composition comprises at least one uniquely labeled microbead having attached thereto a target RNA-specific probe having a region with a sequence that is not present in, or complementary to a region of, any of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the compositions comprise a plurality of uniquely labeled microbeads, at least one of which has attached thereto a probe having a region with a sequence that identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897 and at least a second bead that has attached thereto a probe having a region with a sequence that is identically present in, or complementary to a region of, a target RNA from the human miRNome.
- compositions comprise a plurality of uniquely labeled microbeads, each of which has attached thereto a unique probe having a region that is complementary to target RNAs that comprise a substantial portion of the human miRNome, such as at least about 60%, at least about 70%, at least about 80%, at least about 90%, or at least about 95% of the human miRNome.
- compositions comprise a plurality of uniquely labeled microbeads having attached thereto a unique probe having a region with a sequence that is identically present in target RNAs that comprise a substantial portion of the human miRNome, such as at least about 60%, at least about 70%, at least about 80%, at least about 90%, or at least about 95% of the human miRNome.
- compositions are provided that comprise at least one polynucleotide for detecting at least one target RNA.
- the polynucleotide is used as a primer for a reverse transcriptase reaction.
- the polynucleotide is used as a primer for amplification.
- the polynucleotide is used as a primer for RT-PCR.
- the polynucleotide is used as a probe for detecting at least one target RNA.
- the polynucleotide is detectably labeled.
- the polynucleotide is a FRET probe.
- the polynucleotide is a TaqMan® probe, a Molecular Beacon, or a Scorpion probe.
- a composition comprises at least one FRET probe having a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- a composition comprises at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, at least 75, or at least 100 FRET probes, each of which has a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- a FRET probe is labeled with a donor/acceptor pair such that when the probe is digested during the PCR reaction, it produces a unique fluorescence emission that is associated with a specific target RNA.
- each probe is labeled with a different donor/acceptor pair such that when the probe is digested during the PCR reaction, each one produces a unique fluorescence emission that is associated with a specific probe sequence and/or target RNA.
- the sequence of the FRET probe is complementary to a target region of a target RNA.
- the FRET probe has a sequence that comprises one or more base mismatches when compared to the sequence of the best-aligned target region of a target RNA.
- a composition comprises a FRET probe consisting of at least 8, at least 9, at least 10, at least 11, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, or at least 25 nucleotides, wherein at least a portion of the sequence is identically present in, or complementary to a region of, one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the FRET probe has a sequence with one, two or three base mismatches when compared to the sequence or complement of one of SEQ ID NOs: 1 to 86, 196 to 399, 565 to 707, or 863 to 897.
- the compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86.
- the compositions comprise at least two, at least three, at least five, at least 8, at least ten, at least 15, at least
- target RNA-specific FRET probes each comprising a sequence that is identically present in, or complementary to a region of, a different one of 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 or 86.
- compositions comprise at least one target RNA-specif ⁇ c FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 or 79.
- compositions comprise at least two, at least three, at least five, at least eight, at least 10, at least 12, at least 15, or at least 18 uniquely labeled target RNA-specif ⁇ c FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 or 79.
- compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- compositions comprise at least two, at least three, at least five, at least eight, at least 10, at least 12, at least 15, or at least 18 uniquely labeled target RNA-specific FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 or 84.
- the compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- the compositions comprise at least two, at least three, at least four, at least five, at least six, at least seven, or at least eight uniquely labeled target RNA-specific FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, or 78.
- compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the compositions comprise at least two, at least three, at least five, at least 8, at least ten, at least 15, at least 20, at least 25, at least 30, or at least 35 uniquely labeled target RNA-specific FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 or 85.
- the compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 9, 50, 51, 70, 72 or 75.
- the compositions comprise at least two, at least three, at least four, at least five, or at least six uniquely labeled target RNA-specific FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 9, 50, 51, 70, 72 or 75.
- compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- the compositions comprise at least two, at least five, at least 10, at least 15, at least 20, at least 25, or at least 30 uniquely labeled target RNA-specific FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 231 , 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, or 648.
- compositions comprise at least one target RNA-specific FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- compositions comprise at least two, at least five, at least 10, at least 15, or at least 20 uniquely labeled target RNA-specific FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, or 632.
- the compositions comprise at least one target RNA-specif ⁇ c FRET probe comprising a sequence that is identically present in, or complementary to a region of, one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- the compositions comprise at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 70 uniquely labeled target RNA-specif ⁇ c FRET probes, each of which comprises a sequence that is identically present in, or complementary to a region of, a different one of SEQ ID NOs: 226 to 289, 565 to 604, or 863 to 868.
- kits comprises a polynucleotide discussed above. In some embodiments, a kit comprises at least one primer and/or probe discussed above. In some embodiments, a kit comprises at least one polymerase, such as a thermostable polymerase. In some embodiments, a kit comprises dNTPs. In some embodiments, kits for use in the real time RT-PCR methods described herein comprise one or more target RNA-specif ⁇ c FRET probes and/or one or more primers for reverse transcription of target RNAs and/or one or more primers for amplification of target RNAs or cDNAs reverse transcribed therefrom.
- one or more of the primers and/or probes is
- a “linear" primer refers to a polynucleotide that is a single stranded molecule, and typically does not comprise a short region of, for example, at least 3, 4 or 5 contiguous nucleotides, which are complementary to another region within the same polynucleotide such that the primer forms an internal duplex.
- the primers for use in reverse transcription comprise a region of at least 4, at least 5, at least 6, at least 7 or more contiguous nucleotides at the 3 '-end that has a sequence that is complementary to region of at least 4, at least 5, at least 6, at least 7 or more contiguous nucleotides at the 5'-end of a target RNA.
- a kit comprises one or more pairs of linear primers (a "forward primer” and a “reverse primer”) for amplification of a cDNA reverse transcribed from a target RNA.
- a first primer comprises a region of at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at least 10 contiguous nucleotides having a sequence that is identical to the sequence of a region of at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at least 10 contiguous nucleotides at the 5 '-end of a target RNA.
- a second primer comprises a region of at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at least 10 contiguous nucleotides having a sequence that is complementary to the sequence of a region of at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at least 10 contiguous nucleotides at the 3 '-end of a target RNA.
- the kit comprises at least a first set of primers for amplification of a cDNA that is reverse transcribed from a target RNA capable of specifically hybridizing to a nucleic acid comprising a sequence identically present in one of SEQ ID NOs: 1 to 86 and/or a cDNA that is reverse transcribed from a target RNA that comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- the kit comprises at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, at least 75, or at least 100 sets of primers, each of which is for amplification of a cDNA that is reverse transcribed from a different target RNA capable of specifically hybridizing to a sequence selected from SEQ ID NOs: 1 to 86 and/or a cDNA that is reverse transcribed from a target RNA that comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707 and 863 to 897.
- the kit comprises at least one set of primers that is capable of amplifying more than one cDNA reverse transcribed from a target RNA in a sample.
- probes and/or primers for use in the compositions described herein comprise deoxyribonucleotides.
- probes and/or primers for use in the compositions described herein comprise deoxyribonucleotides and one or more nucleotide analogs, such as LNA analogs or other duplex-stabilizing nucleotide analogs described above.
- probes and/or primers for use in the compositions described herein comprise all nucleotide analogs.
- the probes and/or primers comprise one or more duplex- stabilizing nucleotide analogs, such as LNA analogs, in the region of complementarity.
- compositions described herein also comprise probes, and in the case of RT-PCR, primers, that are specific to one or more housekeeping genes for use in normalizing the quantities of target RNAs.
- probes and primers
- Such probes (and primers) include those that are specific for one or more products of housekeeping genes selected from U6 snRNA, RNU44, RNU48, U47, 7SL scRNA, Ul snRNA, 5.8S rRNA, and U87 scaRNA.
- kits for use in real time RT-PCR methods described herein further comprise reagents for use in the reverse transcription and amplification reactions.
- the kits comprise enzymes such as reverse transcriptase, and a heat stable DNA polymerase, such as Taq polymerase.
- the kits further comprise deoxyribonucleotide triphosphates (dNTPs) for use in reverse transcription and amplification.
- the kits comprise buffers optimized for specific hybridization of the probes and primers.
- quantitation of target RNA expression levels requires assumptions to be made about the total RNA per cell and the extent of sample loss during sample preparation. In order to correct for differences between different samples or between samples that are prepared under different conditions, the quantities of target RNAs in some embodiments are normalized to the expression of at least one endogenous housekeeping gene.
- Appropriate genes for use as reference genes in the methods described herein include those as to which the quantity of the product does not vary between normal samples and samples from sepsis patients, or between different cell lines or under different growth and sample preparation conditions.
- endogenous housekeeping genes useful as normalization controls in the methods described herein include, but are not limited to, U6 snRNA, RNU44, RNU48, U47, 7SL scRNA, Ul snRNA, 5.8S rRNA, and U87 scaRNA.
- the at least one endogenous housekeeping gene for use in normalizing the measured quantity of microRNAs is selected from U6 snRNA, RNU44, RNU48, U47, 7SL scRNA, Ul snRNA, 5.8S rRNA, and U87 scaRNA.
- one housekeeping gene is used for normalization. In some embodiments, more than one housekeeping gene is used for normalization.
- methods comprise detecting a qualitative change in a target RNA profile generated from a human sample as compared to a normal target RNA profile (in some exemplary embodiments, a target RNA profile of a control sample). Some qualitative changes in the expression profile are indicative of the presence of sepsis in a sample from a subject.
- target RNA profile refers to a set of data regarding the concurrent expression of a plurality of target RNAs in the same sample.
- At least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least 10, at least 12, at least 15, at least 20, at least 25, at least 30, at least 35, or at least 40 of the target RNAs of the plurality of target RNAs are capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 6, 8, 11, 13, 14, 15, 17, 19, 20, 21, 23, 27, 29, 30, 33, 34, 35, 38, 39, 45, 46, 47, 48, 49, 52, 56, 57, 58, 59, 60, 62, 63, 64, 65, 67, 69, 71, 73, 74, 76, 78, 79, 84 and 86.
- At least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least nine, at least 10, at least 11, at least 12, at least 15, or at least 18 of the target RNAs of the plurality of target RNAs is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 8, 14, 23, 30, 39, 52, 57, 59, 60, 62, 63, 64, 65, 67, 74, 76, 78 and 79.
- At least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least nine, at least 10, at least 11, at least 12, at least 15, or at least 18 of the target RNAs of the plurality of target RNAs is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 6, 17, 19, 20, 21, 27, 29, 33, 34, 35, 38, 45, 46, 47, 48, 49, 69 and 84.
- At least one, at least two, at least three, at least four, at least five, at least six, at least seven, or at least eight of the target RNAs of the plurality of target RNAs is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 8, 14, 59, 62, 63, 64, 74, and 78.
- At least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least 10, at least 12, at least 15, at least 20, at least 25, at least 30, or at least 35 of the target RNAs of the plurality of target RNAs is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1, 2, 3, 4, 5, 7, 10, 12, 16, 18, 22, 24, 25, 26, 28, 30, 31, 32, 37, 41, 42, 43, 44, 53, 54, 55, 61, 66, 68, 77, 80, 81, 82, 83 and 85.
- At least one, at least two, at least three, at least four, at least five, or at least six of the target RNAs of the plurality of target RNAs is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 9, 50, 51, 70, 72 and 75.
- At least one, at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 75 of the plurality of target RNAs is capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86.
- at least one, at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 75, or at least 100 of the plurality of target RNAs comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897.
- At least one, at least two, at least five, at least 10, at least 15, at least 20, or at least 25 of the plurality of target RNAs comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 237, 242, 245, 253, 260, 261, 262, 263, 266, 269, 275, 287, 303, 342, 352, 566, 567, 568, 571, 570, 573, 574, 575, 577, 579, 580, 581, 588, 591, 598, 601, 608, 612, 613, 624, 626, 629, 632, 635, 637, 641, 642, 644, and 648.
- At least one, at least two, at least five, at least 10, at least 15, at least 20, or at least 25 of the plurality of target RNAs comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 231, 236, 242, 260, 261, 266, 287, 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, and 632.
- At least one, at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 70 of the plurality of target RNAs comprises at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 226 to 289, 565 to 604, and 863 to 868.
- at least one, at least two, at least five, at least 10, at least 15, at least 20, at least 25, at least 30, at least 40, at least 50, at least 60, or at least 75 of the plurality of target RNAs comprises a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86.
- a target RNA, in its mature form comprises fewer than 30 nucleotides.
- a target RNA is a microRNA.
- concurrent expression data are obtained using, e.g., a microarray, as described above.
- a microarray comprising probes having sequences that are complementary to a substantial portion of the miRNome may be employed to carry out target RNA gene expression profiling, for analysis of target RNA expression patterns.
- distinct target RNA signatures are associated with established markers for sepsis.
- distinct target RNA signatures are associated with established markers for sepsis caused by bacterial infection, such as for sepsis caused by gram-positive bacterial infection, sepsis caused by gram-negative bacterial infection or sepsis caused by mycobacterial infection.
- distinct target RNA signatures are associated with established markers for sepsis caused by viral infection.
- distinct target RNA signatures are associated with established markers for sepsis caused by multiple infection, such as by co-infection with bacteria and viruses, or by co-infection with more than one viral or more than one bacterial strain.
- distinct target RNA signatures are associated directly with the level of severity of the sepsis.
- total RNA from a sample from a subject suspected of having sepsis is quantitatively reverse transcribed to provide a set of labeled oligonucleotides complementary to the RNA in the sample.
- the oligonucleotides are then hybridized to a microarray comprising target RNA-specific probes to provide a hybridization profile for the sample.
- the result is a hybridization profile for the sample representing the expression pattern of target RNAs in the sample.
- the hybridization profile comprises the signal from the binding of the oligonucleotides reverse transcribed from the sample to the target RNA-specific probes in the microarray.
- the profile is recorded as the presence or absence of binding (signal vs. zero signal). In some embodiments, the profile recorded includes the intensity of the signal from each hybridization. The profile is compared to the hybridization profile generated from a normal, i.e., nonseptic sample, or in some embodiments, a control sample. An alteration in the signal is indicative of the presence of sepsis in the subject.
- methods herein in combination with detecting one or more target RNAs that are capable of specifically hybridizing to a nucleic acid comprising a sequence selected from SEQ ID NOs: 1 to 86 and/or detecting one or more target RNAs comprising at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 196 to 399, 565 to 707, and 863 to 897 and/or detecting one or more target RNAs that comprise a sequence that is complementary to at least 15 contiguous nucleotides of a sequence selected from SEQ ID NOs: 1 to 86, methods herein further comprise detecting the level(s) of expression of at least one other marker associated with sepsis.
- the methods described herein further comprise detecting altered expression of sepsis-associated small RNAs with non- canonical hairpins.
- the methods described herein further comprise detecting chromosomal codependents, i.e., target RNAs clustered near each other in the human genome which tend to be regulated together. Accordingly, in further embodiments, the methods comprise detecting the expression of one or more target microRNAs, each situated within the chromosome no more than 50,000 bp from the chromosomal location of the pre-microRNA sequences in Table 2.
- THP-I cells and pooled human monocytes from healthy donors were stimulated with different Toll-Like Receptors (TLR) agonists set forth in Table 5 below.
- TLR Toll-Like Receptors
- THP-I cells were obtained from American Type Culture Collection
- THP-I cells were grown to a concentration of 2.5 million per milliliter in RPMI medium 1640 supplemented with 10% FBS, Ix nonessential amino acids, 100 units/ml penicillin, 100 units/ml streptomycin, and 2 mM glutamine in a humidified incubator containing 5% CO 2 at 37°C.
- Human monocytes were isolated from whole blood of healthy donors (St Guy's hospital, UK), using CD14 positive magnetic microbead positive selection (according to manufacturer's protocol, Miltenyi Biotec). Isolated monocytes were then cultured with RPMI 1640 medium to a concentration of 2.5 million per milliliter in 6-well plates and cultured in a humidified incubator containing 5% CO 2 at 37 0 C.
- RNA samples were diluted in RNase-free water and stored in -80°C (-112 0 F).
- RNA quality was assessed by calculating OD 260/280 ratios. The quality of all RNA samples was high as assessed using an Agilent Bioanalyser 2100, as exemplified by the electropherogram shown in FIG. 1 obtained for total RNA from human monocyte cell line THP-I after stimulation for 8h with agonist Pam3CSK4. Similar electropherograms were obtained for total RNA from the other cell samples as well.
- RNA fractionator (Ambion). The Ambion gel purification protocol enriches for small RNAs less than 40 nucleotides (nt) long, including microRNAs. Briefly, a total RNA sample was loaded onto a pre-cast gel using the Flash PAGE Fractionator. The total RNA fraction smaller than 40 nt (the "microRNA fraction") was recovered after gel migration and resuspended into nuclease free water. Microarray Analysis
- the oligonucleotide probes used for microarray preparation had the configuration 5'-NH 2 -(C) 6 -(spacer)-(oligomer probe sequence)-3'.
- the 5 '-amino group allowed chemical bonding onto the array support.
- the probes were spotted in duplicate using the Nexterion spotting buffer provided with the array glass support by Schott with 1 % SDS (sodium dodecyl sulfate) added to allow larger spot sizes (e.g., 100-150 microns compared to 70-100 microns without SDS).
- the spotter used was the QArray mini (Genetix) equipped with Stealth SMP3 pins (Telechem). After deposition of one series of spots, the spotting needle was washed 5 times with 6OmM NaOH before spotting the next series of probes. Each slide is designed with 32 blocks of spotted probes, with each block being a 20x20 square of spotted probes. Each probe was spotted in duplicate. Spotted glass slides were stored at 4°C until use.
- microRNA fraction was adapted from a published protocol developed at EMBL (Heidelburg, Germany) by the European Molecular Biology Group (Castoldi et al, "A sensitive array for microRNA expression profiling (miChip) based on locked nucleic acids (LNA),” RNA 2006 May;12(5):913-20. Epub 2006 Mar 15, incorporated herein by reference in its entirety).
- the microRNA fraction was incubated for 6 hours at 4°C with a mixture containing 10 ⁇ M of dye-labelled tetra-nucleotide (5'-rUrUrUrU -Cy5- 3') (or alternatively, 5'-rUrUrUrU - Cy3-3') (Biospring, Germany) in Ambion buffer diluted to IX with RNase free water, 8% polyethylene glycol (PEG), 2 mM adenosine triphosphate (ATP), and T4 RNA ligase (0.7U/ ⁇ l).
- PEG polyethylene glycol
- ATP 2 mM adenosine triphosphate
- T4 RNA ligase 0.7U/ ⁇ l
- the labelled microRNA fraction was hybridized to the spotted arrays using a Discovery hybridization station (Ventana, Arlington, AZ). Briefly, 2 mL of a mixture of 1% BSA, 2X SSC, and 0.2 % SDS was incubated with the chips for 30 min at 42°C. Then the chips were washed once using EZ Prep buffer (Ventana) and then three more times with Ribowash (Ventana). Next, 20 ⁇ l of the labelled microRNA mixture and 180 ⁇ l of ChipHybe Reagent (Ventana) were added to the array. The arrays were heated for 6 minutes at 37 0 C, then were incubated at 42°C for 8 hours, after which the heating was stopped.
- EZ Prep buffer Ventana
- ChipHybe Reagent ChipHybe Reagent
- the chips were washed once with Ribowash (Ventana) and then heated for 2 minutes at 37°C.
- the chips were washed again with Ribowash (Ventana) with one drop of CheapClean (Ventana) added, and incubated for 2 minutes at 37°C.
- the chips were washed two more times using Ribowash (Ventana).
- the chips were stored dry overnight. On the following day, the final washes were done according to Ventana' s instructions for the Discovery hybridization station.
- the slides were washed twice with 2X SSC + 0.2X SDS buffer and then one more time with 0. IX SSC. All the slides were dried using a speed centrifuge from Arrayit (TeleChem International, Sunnyvale, CA) at room temperature and kept in the dark before scanning.
- the following solution may be used for array hybridization (solution 2) to form probe:target RNA hybrids by mixing 2 parts of the 1.5X TMAC Hybridization Solution to 1 part (v:v) sample, so that the final component concentrations are 3M TMAC, 0.10% Sarkosyl, 50 mM Tris, and 4 mM EDTA, and incubating on the array at 42 0 C for 8h: Table 6: 1.5X TMAC Hybridization Solution
- the image was formatted in tif format, defined by an image color depth of 16 bits/pixel (1600*1600). At such setting, pixels can assume intensity values ranging from 0 to 65,535. Pixels exhibiting the maximum intensity value are "saturated” and were assigned the value of 65,535. The resolution of the array scan was set at 10 ⁇ m/pixel. For hybridization experiments using different fluorescent dyes (e.g., Cy5 and Cy3) the photomultiplier tube (PMT) was adjusted to the higher intensity spot (Cy3 is scanned at lower PMT settings than Cy5).
- fluorescent dyes e.g., Cy5 and Cy3
- the PMT of the laser scanner digitized the captured fluorescence intensity for each given "point" of a slide and stored the numerical value as a pixel corresponding to that point. A picture composed of such pixels was then analyzed.
- the first task for image analysis was to detect the spot position, using a process called segmentation. Spots were segmented by circles of adaptable or fixed radius. To be reliably segmented and quantified, the spot diameter was required to be more than 5 - 6 pixels. Before segmentation an indexing grid was provided giving the approximate positions of the spots. The segmentation itself detected the limits of spots near the grid circles. Briefly, the Genepix software assigns a circle to each spot on the array (segmentation). The segmentation had to be conducted in a somewhat flexible way due to spotting imperfections and/or support deformation, as the spots were almost never on a perfectly rectangular grid.
- the second task of image analysis was to quantify spots and export the data into a result file. This was a relatively easy and well-defined task once the spots were located on the image.
- the statistical approach used most frequently to quantify spot intensity was the mean or median of pixels belonging to a spot.
- the median approach was more robust than the mean value in the presence of outlier pixels. In practice, however, there was little difference in the results obtained using mean or median.
- Array data were first tested for quality by comparing the spot intensities for the internal controls.
- Tables 7 and 8 One internal control (SEQ ID NO: 178) was used as a labelling control (this synthetic RNA is added to the purified microRNA fraction before labelling), and 7 other internal controls (SEQ ID NOs: 179- 185) were used for the normalization of the data (these synthetic RNA controls are added to the total RNA fraction before hybridization at 520 fmol each/array).
- Log2ratio where the Log2ratio equals average intensity signal of the duplicated spots/median intensity of all positives controls for the block.
- the normalization was done per block to avoid non-homogenous labelling of all blocks of the array. This block-by- block normalization has been shown to be more efficient then using overall normalization of the slide.
- the obtained values are Log2 values.
- the expression fold-change corresponds to 2 (Log2ratlo) .
- Log2ratio is the ratio between the two conditions compared, or log2(Xcell-line/Xnormal), which is the same as (log2Xcell-line - log2Xnormal), where X is the measured intensity value. In cases where there was no signal from the "normal" condition, the lowest measured intensity value in the experiment was used as the baseline from which a fold- change expression value was calculated. A fold-change value of less than zero corresponds to a down-regulation of (1 /fold-change) times.
- Data are tabulated in Table 2, and include all microRNAs over- expressed in monocytes in response to stimulation with one or more TLR agonists.
- Luminex technology (Luminex Corp., Austin, TX) is based on liquid phase hybridization to probe-labelled beads, followed by flow cytometry detection of beads with differing ratios of fluorescent dyes. Beads with up to 100 different dye ratios are available, making it possible to interrogate a single sample for up to 100 analytes simultaneously. Monocyte samples
- Human monocytes are isolated form whole blood of healthy donors using CD 14 positive megnetic microbead positive selection (according to manufacturer's protocol, Miltenyi Biotec). Isolated monocytes are then cultured with RPMI 1640 medium to a concentration of 2.5 million per milliliter in 6-well plates, in a humidified incubator containing 5% CO 2 at 37°C.
- RNA samples are prepared using TRIzol ® Reagent, Invitrogen (Carlsbad, CA) according to the manufacturer's protocol. All RNA samples are diluted in RNase-free water and stored in -80 0 C (-112 0 F).
- 5'AminoC6-probe sequence i.e., similar to the structure in Example 1, but without the linker sequence
- the probes are coupled to the beads using carbodiimide chemistry according to the manufacturer's protocol (Luminex bead coupling protocol).
- the probe- coupled beads are stored at 4°C.
- RNAs used for the array controls are added to the total RNA fraction isolated from the patient samples. For each sample, three replicates are assayed in parallel. For each replicate, 250 ng of total RNA is used.
- the total RNA preparation Prior to hybridization with Luminex beads, the total RNA preparation is treated with calf intestinal phosphatase (CIP; Invitrogen) to prevent the formation of dendrimers, which result from the circularization of a single RNA molecule, or concatenation to another RNA molecule. Pre-treatment with CIP is according to the manufacturer's protocol, and removes 5'-phosphate groups.
- CIP calf intestinal phosphatase
- the total RNA fraction is labelled with biotin using the Vantage microRNA Labelling Kit (Marligen).
- the labelled fraction is hybridized to the Luminex beads using the Marligen protocol. Briefly, the polynucleotide beads are mixed with the Marligen hybridization solution (1.5 X TMAC) and the labelled total RNA. The hybridization is performed at 60°C for an hour in the dark. After hybridization, the beads are washed using the Luminex standard 6X SSPET wash buffer (sodium phosphate, sodium chloride, EDTA, Triton X-100, pH 7.4).
- Luminex beads The detection of the Luminex beads is done using streptavidin phycoerythrin (SAPE) (Europa Bioproducts, Cambridge, UK). The SAPE is added to the washed beads according to the Luminex protocol. The beads are then read using the Luminex IS-200 instrument on the high gain setting for better resolution.
- SAPE streptavidin phycoerythrin
- the Luminex IS-200 reads at least 25 beads of each dye-ratio in the reaction mix. Each dye-ratio bead corresponds to a particular probe sequence, and the intensity value is returned as an average value of all read beads.
- the mean fluorescence intensity (MFI) data is normalized using synthetic RNA controls or alternatively using the mean of expressed oligonucleotides, and fold changes between normal and stimulated or diseased samples are computed using the Bioplex software (Bio-Rad, Hercules, CA) and the R bioconductor package (Bioconductor: open software development for computational biology and bioinformatics, Genome Biol. 2004;5(10):R80. Epub 2004 Sep 15).
- Table 9 lists exemplary internal controls RNAs that can be added to total RNA prior to hybridization to the beads.
- Table 10 shows the corresponding probe sequences that are coupled to control beads.
- Log2ratio where the Log2ratio equals average intensity signal of 50 replicates of the same bead (each coupled to the same oligo sequence) divided by the median intensity of all of the positive controls in one well.
- the normalization is done per well to avoid non- homogenous labelling of all wells of the plate. This well-by- well normalization has been shown to be more efficient then using overall normalization of the plate.
- the obtained values are Log2 values.
- the intensities of the beads for each coupled bead are compared in the sample from monocytes from healthy donors grown in PMI medium, which does not activate the cells, versus monocytes ultimatelyted in vitro (stimulated with a TLR agonist or grown in PMA medium, which activates the cells), resulting in an evaluation of the relative expression for each microRNA.
- the expression fold-change corresponds to 2 (Log2ratio) .
- Log2ratio is the ratio between the two conditions compared, or log2(Xcell-line/Xnormal), which is the same as (log2Xcell-line - log2Xnormal), where X is the measured intensity value. In cases where there is no signal from the "normal" condition, the lowest measured intensity value in the experiment is used as the baseline from which a fold- change expression value was calculated. A fold-change value of less than zero corresponds to a down-regulation of (1 /fold-change) times. A two-fold change, either upregulated or downregulated, is considered significant.
- RNA from a sepsis patient was used for preparing a smRNASeq dataset. Briefly, 5 ⁇ g of total RNA was used for small RNA sequencing on a Solexa GA II (Illumina) using a standard library and sequencing protocol provided by the manufacturer.
- the sum of all of the counts for all of the isomirs was used for the comparison. Furthermore, when a precursor gene for a particular microRNA sequence is present at multiple locations in the genome, both candidate names are shown, with the same ranking and same sequence. Either or both of those candidates may be present at increased levels in the sepsis patient sample.
- Table 13 Precursor sequences and chromosomal locations (from human genome NCBI36) of the microRNAs in Table 17
- microRNAs in miRBase human version 14.0
- miRBase human version 14.0
- the microRNAs were ranked from 1 to 5, depending on whether the sepsis patient smRNASeq dataset contained the highest number of counts of the particular microRNA, the second highest, etc. The results are shown in Table 14.
- Table 14 are shown in Table 15.
- microRNAs in miRBase human version 14.0
- miRBase human version 14.0
- the novel star forms identified in this analysis are not.
- the microRNAs were ranked from 1 to 5, depending on whether the sepsis patient smRNASeq dataset contained the highest number of counts of the particular microRNA, the second highest, etc. The results are shown in Table 16.
- Table 16 Novel star- form microRNAs u re ulated in a se sis smRNASe dataset
- microRNA precursor sequences for the microRNAs listed in Table 16 are shown in Table 17.
- Table 17 Precursor sequences and chromosomal locations (from human genome NCBI36) of the microRNAs in Table 16
- Table 18 shows the ratio of the number of counts in the sepsis smRNASeq dataset versus the average number of counts from all of the other smRNASeq datasets for each novel microRNA for which the ratio was 2 or higher.
- microRNAs 13446-R, 13642-R, 13677-L, 14086-L, 14093-L, 14177- L, and 6415-R had counts in the sepsis smRNASeq dataset of at least 10-fold greater than the average counts in the other datasets.
- Table 19 shows the ratio of the number of counts in the sepsis smRNASeq dataset versus the average number of counts from all of the other smRNASeq datasets for each microRNA from miRBase for which the ratio was 2 or higher.
- microRNAs miR-140-5p, miR-142-3p, miR-142-5p, miR-144*, miR-144, miR-15a, miR-223, miR-223*, miR-451, miR-618, miR-627, miR-148a*, miR-140-3p, miR-146b-5p, and miR-17* (SEQ ID NOs: 566, 567, 568, 571, 570, 574, 580, 581, 588, 598, 601, 608, 624, 626, 629, and 632, respectively) had counts in the sepsis smRNASeq dataset of at least 10-fold greater than the average counts in the other datasets.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Analytical Chemistry (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Pathology (AREA)
- Immunology (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Biotechnology (AREA)
- Biophysics (AREA)
- Physics & Mathematics (AREA)
- Biochemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2010800143880A CN102388149A (en) | 2009-02-02 | 2010-02-02 | Methods of detecting sepsis |
| EP10736571.0A EP2391738B1 (en) | 2009-02-02 | 2010-02-02 | Methods of detecting sepsis |
| CA2751213A CA2751213A1 (en) | 2009-02-02 | 2010-02-02 | Methods of detecting sepsis |
| JP2011548400A JP5890686B2 (en) | 2009-02-02 | 2010-02-02 | How to detect sepsis |
| AU2010207975A AU2010207975B2 (en) | 2009-02-02 | 2010-02-02 | Methods of detecting sepsis |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14927709P | 2009-02-02 | 2009-02-02 | |
| US61/149,277 | 2009-02-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010088668A2 true WO2010088668A2 (en) | 2010-08-05 |
| WO2010088668A3 WO2010088668A3 (en) | 2010-09-23 |
Family
ID=42396408
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/022885 WO2010088668A2 (en) | 2009-02-02 | 2010-02-02 | Methods of detecting sepsis |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9493832B2 (en) |
| EP (1) | EP2391738B1 (en) |
| JP (1) | JP5890686B2 (en) |
| CN (1) | CN102388149A (en) |
| AU (1) | AU2010207975B2 (en) |
| CA (1) | CA2751213A1 (en) |
| WO (1) | WO2010088668A2 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102191327A (en) * | 2011-04-18 | 2011-09-21 | 中国人民解放军总医院 | Kit for forecasting death rate of patients with sepsis and application thereof |
| WO2012072685A1 (en) * | 2010-12-02 | 2012-06-07 | Katholieke Universiteit Leuven, K.U.Leuven R&D | Irak-related interventions and diagnosis |
| CN102628078A (en) * | 2011-12-27 | 2012-08-08 | 芮屈生物技术(上海)有限公司 | MicroRNA-155 level in various types of cancerous lesions prophase in situ hybridization detection kit, detection method and application thereof |
| WO2013040379A1 (en) * | 2011-09-16 | 2013-03-21 | Cepheid | Methods of detecting sepsis |
| WO2014164480A1 (en) * | 2013-03-12 | 2014-10-09 | Cepheid | Methods of detecting cancer |
| US20160024505A1 (en) * | 2009-05-06 | 2016-01-28 | Curna, Inc. | Treatment of lipid transport and metabolism gene related diseases by inhibition of natural antisense transcript to a lipid transport and metabolism gene |
| US10000808B2 (en) | 2009-01-16 | 2018-06-19 | Cepheid | Methods of detecting cervical cancer |
| WO2018219998A1 (en) * | 2017-05-30 | 2018-12-06 | Siemens Aktiengesellschaft | Mirnas as biomarkers for a systemic inflammatory response syndrome |
| US20220204973A1 (en) * | 2020-12-18 | 2022-06-30 | Hoffmann-La Roche Inc. | Antisense Oligonucleotide for Targeting Progranulin |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009155455A1 (en) * | 2008-06-19 | 2009-12-23 | John Wayne Cancer Institute | Use of runx3 and mir-532-5p as cancer markers and therapeutic targets |
| WO2012103355A2 (en) | 2011-01-26 | 2012-08-02 | Cepheid | Methods of detecting lung cancer |
| ITRM20110685A1 (en) | 2011-12-23 | 2013-06-24 | Internat Ct For Genetic En Gineering And | MICRORNA FOR CARDIAC REGENERATION THROUGH THE INDUCTION OF THE PROLIFERATION OF CARDIAC MYCYCLES |
| JP5933010B2 (en) * | 2012-08-20 | 2016-06-08 | 国立研究開発法人国立がん研究センター | Cancer treatment |
| CN102839175B (en) * | 2012-08-24 | 2013-05-29 | 中国医科大学附属第一医院 | Molecular marker miR-526a for progress of human immunodeficiency virus (HIV) infectious disease |
| EP2733220B1 (en) * | 2012-11-16 | 2017-10-18 | Siemens Aktiengesellschaft | Novel miRNA as a diagnostic marker for Alzheimer's Disease |
| EP2733219B1 (en) | 2012-11-16 | 2017-09-20 | Siemens Aktiengesellschaft | Diagnostic miRNA markers for Alzheimer |
| US10011873B2 (en) | 2012-11-16 | 2018-07-03 | Universitat Des Saarlandes | Diagnostic miRNA markers for Parkinson disease |
| ES2734803T3 (en) * | 2015-06-16 | 2019-12-12 | Centro De Investig Biomedica En Red Ciber | Method for diagnosis / prognosis of adolescent idiopathic scoliosis |
| CN105543227A (en) * | 2016-01-11 | 2016-05-04 | 山东农业大学 | Chicken enteritis salmonella infection resistance character molecular marker gga-miR-1662 and detection method thereof |
| CN105462982A (en) * | 2016-01-11 | 2016-04-06 | 山东农业大学 | Molecular marker gga-miR-1416-5p of chicken salmonella enteritidis infection resistance characters and detection method of molecular marker gga-miR-1416-5p |
| WO2018118691A1 (en) | 2016-12-19 | 2018-06-28 | Indiana University Research & Technology Corporation | Dna methylation in inflammatory disease |
| CN110273012B (en) * | 2018-03-13 | 2022-11-04 | 厦门大学 | Method for detecting septicemia pathogen |
| CN109706239A (en) * | 2018-09-26 | 2019-05-03 | 南京市妇幼保健院 | A kind of application of miR-1976 in prevention and treatment bacterial vaginosis BV |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087949A2 (en) | 2003-04-02 | 2004-10-14 | Sirs-Lab Gmbh | Method for recognising acute generalised inflammatory conditions (sirs), sepsis, sepsis-like conditions and systemic infections |
| WO2006013076A1 (en) | 2004-08-02 | 2006-02-09 | Societe De Technologie Michelin | Tyre/wheel assembly for motor vehicle |
| US20060094025A1 (en) | 2004-11-02 | 2006-05-04 | Getts Robert C | Methods for detection of microrna molecules |
| US20060292616A1 (en) | 2005-06-23 | 2006-12-28 | U.S. Genomics, Inc. | Single molecule miRNA-based disease diagnostic methods |
| WO2007011903A2 (en) | 2005-07-15 | 2007-01-25 | Applera Corporation | Analyzing messenger rna and micro rna in the same reaction mixture |
| WO2007025281A2 (en) | 2005-08-24 | 2007-03-01 | Applera Corporation | A method to quantify sirnas, mirnas and polymorphic mirnas |
| US20070054287A1 (en) | 2005-05-31 | 2007-03-08 | Applera Corporation | Method for identifying medically important cell populations using micro rna as tissue specific biomarkers |
| US20070077570A1 (en) | 2005-05-31 | 2007-04-05 | Applera Corporation | Multiplexed amplification of short nucleic acids |
| US7351538B2 (en) | 2004-08-23 | 2008-04-01 | U.S. Genomics | Systems and methods for detecting and analyzing polymers |
| US20090123912A1 (en) | 2005-01-25 | 2009-05-14 | Rosetta Inpharmatics Llc | Methods for quantitating small RNA molecules |
Family Cites Families (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9805918D0 (en) * | 1998-03-19 | 1998-05-13 | Nycomed Amersham Plc | Sequencing by hybridisation |
| US6900016B1 (en) | 2000-09-08 | 2005-05-31 | Applera Corporation | Polymorphisms in known genes associated with inflammatory autoimmune disease, methods of detection and uses thereof |
| US6706867B1 (en) | 2000-12-19 | 2004-03-16 | The United States Of America As Represented By The Department Of Health And Human Services | DNA array sequence selection |
| DE102004009952B4 (en) * | 2004-03-01 | 2011-06-01 | Sirs-Lab Gmbh | Method of detecting sepsis |
| DK1735459T3 (en) | 2004-04-07 | 2012-05-29 | Exiqon As | Methods for Quantification of MicroRNAs and Small Interfering RNAs |
| EP2471923B1 (en) | 2004-05-28 | 2014-08-20 | Asuragen, Inc. | Methods and compositions involving microRNA |
| US7629444B1 (en) * | 2004-06-15 | 2009-12-08 | Monsanto Technology Llc | Nucleotide and amino acid sequences from Xenorhabdus bovienii strain Xs85831 and uses thereof |
| US7361468B2 (en) * | 2004-07-02 | 2008-04-22 | Affymetrix, Inc. | Methods for genotyping polymorphisms in humans |
| US7312035B2 (en) * | 2004-09-03 | 2007-12-25 | Affymetrix, Inc. | Methods of genetic analysis of yeast |
| AU2005300688B2 (en) * | 2004-11-03 | 2012-02-02 | Almac Diagnostics Limited | Transcriptome microarray technology and methods of using the same |
| ES2503765T3 (en) * | 2004-11-12 | 2014-10-07 | Asuragen, Inc. | Procedures and compositions involving miRNA and miRNA inhibitor molecules |
| EP1959012A3 (en) | 2004-12-29 | 2009-12-30 | Exiqon A/S | Novel oligonucleotide compositions and probe sequences useful for detection and analysis of microRNAs and their target mRNAs |
| NO322618B1 (en) * | 2005-04-20 | 2006-11-06 | 2K Tech As | Condition control device and method. |
| DE102005050933A1 (en) | 2005-10-21 | 2007-04-26 | Justus-Liebig-Universität Giessen | Invention relating to expression profiles for the prediction of septic states |
| EP1783645A1 (en) | 2005-11-08 | 2007-05-09 | Actigenics | Methods for the identification of microRNA and their applications in research and human health |
| EP1966390A1 (en) | 2005-12-29 | 2008-09-10 | Exiqon A/S | Detection of tissue origin of cancer |
| WO2007109350A2 (en) | 2006-03-23 | 2007-09-27 | California Institute Of Technology | Modulation of innate immunity receptors' signaling by micrornas mir-146a and mir-146b |
| US20080193943A1 (en) | 2006-09-05 | 2008-08-14 | Abbott Laboratories | Companion diagnostic assays for cancer therapy |
| WO2008040355A2 (en) | 2006-10-06 | 2008-04-10 | Exiqon A/S | Novel methods for quantification of micrornas and small interfering rnas |
| EP2090665A2 (en) | 2006-10-20 | 2009-08-19 | Exiqon A/S | Novel human microRNAs associated with cancer |
| US20090137504A1 (en) | 2006-12-21 | 2009-05-28 | Soren Morgenthaler Echwald | Microrna target site blocking oligos and uses thereof |
| US8415096B2 (en) * | 2007-05-23 | 2013-04-09 | University Of South Florida | Micro-RNAs modulating immunity and inflammation |
| WO2009079592A2 (en) | 2007-12-17 | 2009-06-25 | California Institute Of Technology | Modulating immune system development and function through microrna mir-146 |
| US20100240049A1 (en) | 2009-01-16 | 2010-09-23 | Cepheid | Methods of Detecting Cervical Cancer |
| EP2628803A3 (en) | 2009-02-25 | 2014-01-01 | Cepheid | Methods of detecting lung cancer |
| WO2012103355A2 (en) | 2011-01-26 | 2012-08-02 | Cepheid | Methods of detecting lung cancer |
| AU2012308365A1 (en) | 2011-09-16 | 2014-04-24 | Cepheid | Methods of detecting sepsis |
-
2010
- 2010-02-02 JP JP2011548400A patent/JP5890686B2/en active Active
- 2010-02-02 US US12/698,715 patent/US9493832B2/en active Active
- 2010-02-02 EP EP10736571.0A patent/EP2391738B1/en active Active
- 2010-02-02 CN CN2010800143880A patent/CN102388149A/en active Pending
- 2010-02-02 CA CA2751213A patent/CA2751213A1/en active Pending
- 2010-02-02 AU AU2010207975A patent/AU2010207975B2/en active Active
- 2010-02-02 WO PCT/US2010/022885 patent/WO2010088668A2/en active Application Filing
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004087949A2 (en) | 2003-04-02 | 2004-10-14 | Sirs-Lab Gmbh | Method for recognising acute generalised inflammatory conditions (sirs), sepsis, sepsis-like conditions and systemic infections |
| WO2006013076A1 (en) | 2004-08-02 | 2006-02-09 | Societe De Technologie Michelin | Tyre/wheel assembly for motor vehicle |
| US7351538B2 (en) | 2004-08-23 | 2008-04-01 | U.S. Genomics | Systems and methods for detecting and analyzing polymers |
| US7402422B2 (en) | 2004-08-23 | 2008-07-22 | U.S. Genomics, Inc. | Systems and methods for detecting and analyzing polymers |
| US20060094025A1 (en) | 2004-11-02 | 2006-05-04 | Getts Robert C | Methods for detection of microrna molecules |
| US20090123912A1 (en) | 2005-01-25 | 2009-05-14 | Rosetta Inpharmatics Llc | Methods for quantitating small RNA molecules |
| US20070054287A1 (en) | 2005-05-31 | 2007-03-08 | Applera Corporation | Method for identifying medically important cell populations using micro rna as tissue specific biomarkers |
| US20070077570A1 (en) | 2005-05-31 | 2007-04-05 | Applera Corporation | Multiplexed amplification of short nucleic acids |
| WO2007117256A1 (en) | 2005-05-31 | 2007-10-18 | Applera Corporation | Multiplexed amplification of short nucleic acids |
| US20060292616A1 (en) | 2005-06-23 | 2006-12-28 | U.S. Genomics, Inc. | Single molecule miRNA-based disease diagnostic methods |
| WO2007011903A2 (en) | 2005-07-15 | 2007-01-25 | Applera Corporation | Analyzing messenger rna and micro rna in the same reaction mixture |
| WO2007025281A2 (en) | 2005-08-24 | 2007-03-01 | Applera Corporation | A method to quantify sirnas, mirnas and polymorphic mirnas |
Non-Patent Citations (23)
| Title |
|---|
| "Bioconductor: open software development for computational biology and bioinformatics", GENOME BIOL., vol. 5, no. 10, 15 September 2004 (2004-09-15), pages R80 |
| ALLAWI H.T. ET AL., RNA, vol. 10, 2004, pages 1153 - 1161 |
| BASKERVILLE, S.; BARTEL D.P., RNA, vol. 11, 2005, pages 241 - 247 |
| BIOTECHNIQUES, vol. 41, no. 4, pages 1 - 5 |
| CASTOLDI ET AL.: "A sensitive array for microRNA expression profiling (miChip) based on locked nucleic acids (LNA", RNA, vol. 12, no. 5, 15 March 2006 (2006-03-15), pages 913 - 20 |
| CHEN, C. ET AL., NUCL. ACIDS RES., vol. 33, 2005, pages E179 |
| FRIEDEN, M. ET AL., CURR. PHARM. DES., vol. 14, no. 11, 2008, pages 1138 - 1142 |
| GRIFFITHS-JONES S. ET AL., NUCL. ACIDS RES., vol. 36, 2007, pages 154 - 158 |
| JONSTRUP, S.P. ET AL., RNA, vol. 12, 2006, pages 1 - 6 |
| LIANG, R.Q. ET AL., NUCL. ACIDS RES., vol. 33, no. 2, 2005, pages E17, Retrieved from the Internet <URL:http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid= 548377> |
| LIM, L.P. ET AL., NATURE, vol. 433, 2005, pages 769 - 773 |
| LIU C.G. ET AL., METHODS, vol. 44, 2008, pages 22 - 30 |
| LIU, C.G. ET AL., PROC. NAT'1 ACAD. SCI. USA, vol. 101, 2004, pages 9740 - 9744 |
| LU J. ET AL., NATURE, vol. 435, 2005, pages 834 - 838 |
| MORA J.R.; GETTS R.C., BIOTECHNIQUES, vol. 41, 2006, pages 420 - 424 |
| NANOTECH NEWS, 2006, Retrieved from the Internet <URL:http://nano.cancer.gov/news_center/nanotech_news_2006-10-30b.asp> |
| NELSON, P.T. ET AL., NATURE METHODS, vol. 1, no. 2, 2004, pages 1 - 7 |
| NELSON, P.T. ET AL., RNA, vol. 12, no. 2, 2006, pages 1 - 5 |
| See also references of EP2391738A4 |
| TAGANOV, K. ET AL., PNAS, vol. 103, no. 33, pages 12481 - 6 |
| VDRALLYAY, E. ET AL., NATURE PROTOCOLS, vol. 3, no. 2, 2008, pages 190 - 196 |
| WILKINSON, M., NUCL. ACIDS RES., vol. 16, no. 22, 1988, pages 10,933 |
| WILKINSON, M., NUCL. ACIDS RES., vol. 16, no. 22, 1988, pages 10934 |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10000808B2 (en) | 2009-01-16 | 2018-06-19 | Cepheid | Methods of detecting cervical cancer |
| US20160024505A1 (en) * | 2009-05-06 | 2016-01-28 | Curna, Inc. | Treatment of lipid transport and metabolism gene related diseases by inhibition of natural antisense transcript to a lipid transport and metabolism gene |
| US10604755B2 (en) * | 2009-05-06 | 2020-03-31 | Curna, Inc. | Treatment of lipid transport and metabolism gene related diseases by inhibition of natural antisense transcript to a lipid transport and metabolism gene |
| WO2012072685A1 (en) * | 2010-12-02 | 2012-06-07 | Katholieke Universiteit Leuven, K.U.Leuven R&D | Irak-related interventions and diagnosis |
| CN102191327A (en) * | 2011-04-18 | 2011-09-21 | 中国人民解放军总医院 | Kit for forecasting death rate of patients with sepsis and application thereof |
| WO2013040379A1 (en) * | 2011-09-16 | 2013-03-21 | Cepheid | Methods of detecting sepsis |
| EP2756118A4 (en) * | 2011-09-16 | 2015-09-16 | Cepheid | Methods of detecting sepsis |
| CN102628078A (en) * | 2011-12-27 | 2012-08-08 | 芮屈生物技术(上海)有限公司 | MicroRNA-155 level in various types of cancerous lesions prophase in situ hybridization detection kit, detection method and application thereof |
| US20160024586A1 (en) * | 2013-03-12 | 2016-01-28 | Cepheid | Methods of detecting cancer |
| WO2014164480A1 (en) * | 2013-03-12 | 2014-10-09 | Cepheid | Methods of detecting cancer |
| WO2018219998A1 (en) * | 2017-05-30 | 2018-12-06 | Siemens Aktiengesellschaft | Mirnas as biomarkers for a systemic inflammatory response syndrome |
| US20220204973A1 (en) * | 2020-12-18 | 2022-06-30 | Hoffmann-La Roche Inc. | Antisense Oligonucleotide for Targeting Progranulin |
| US12104153B2 (en) * | 2020-12-18 | 2024-10-01 | Hoffmann-La Roche Inc. | Antisense oligonucleotide for targeting progranulin |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2010207975A2 (en) | 2011-09-01 |
| US20100227325A1 (en) | 2010-09-09 |
| CN102388149A (en) | 2012-03-21 |
| AU2010207975B2 (en) | 2016-04-28 |
| CA2751213A1 (en) | 2010-08-05 |
| JP2012516688A (en) | 2012-07-26 |
| EP2391738B1 (en) | 2016-11-02 |
| JP5890686B2 (en) | 2016-03-22 |
| AU2010207975A1 (en) | 2011-08-18 |
| EP2391738A2 (en) | 2011-12-07 |
| WO2010088668A3 (en) | 2010-09-23 |
| US9493832B2 (en) | 2016-11-15 |
| EP2391738A4 (en) | 2012-07-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2391738B1 (en) | Methods of detecting sepsis | |
| JP5755569B2 (en) | How to detect lung cancer | |
| US10000808B2 (en) | Methods of detecting cervical cancer | |
| US9365903B2 (en) | Compositions comprising polynucleotides for detecting lung cancer | |
| US20160024586A1 (en) | Methods of detecting cancer | |
| US20150376699A1 (en) | Methods of detecting sepsis | |
| US20160257998A1 (en) | Methods of detecting chlamydia and gonorrhea and of screening for infection/inflammation based on genomic copy number | |
| EP2852686A1 (en) | Methods of detecting chlamydia and gonorrhea and of screening for infection/inflammation based on genomic copy number | |
| EP2839028B1 (en) | Methods of detecting bladder cancer | |
| EP2641976A2 (en) | Methods of detecting cervical cancer | |
| US20160108478A1 (en) | Methods of detecting lung cancer | |
| US20160122834A1 (en) | Methods of detecting ebola |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080014388.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10736571 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2751213 Country of ref document: CA Ref document number: 2011548400 Country of ref document: JP Ref document number: 3217/KOLNP/2011 Country of ref document: IN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2010207975 Country of ref document: AU Date of ref document: 20100202 Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010736571 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010736571 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI1008066 Country of ref document: BR |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: PI1008066 Country of ref document: BR |
|
| ENPW | Started to enter national phase and was withdrawn or failed for other reasons |
Ref document number: PI1008066 Country of ref document: BR |