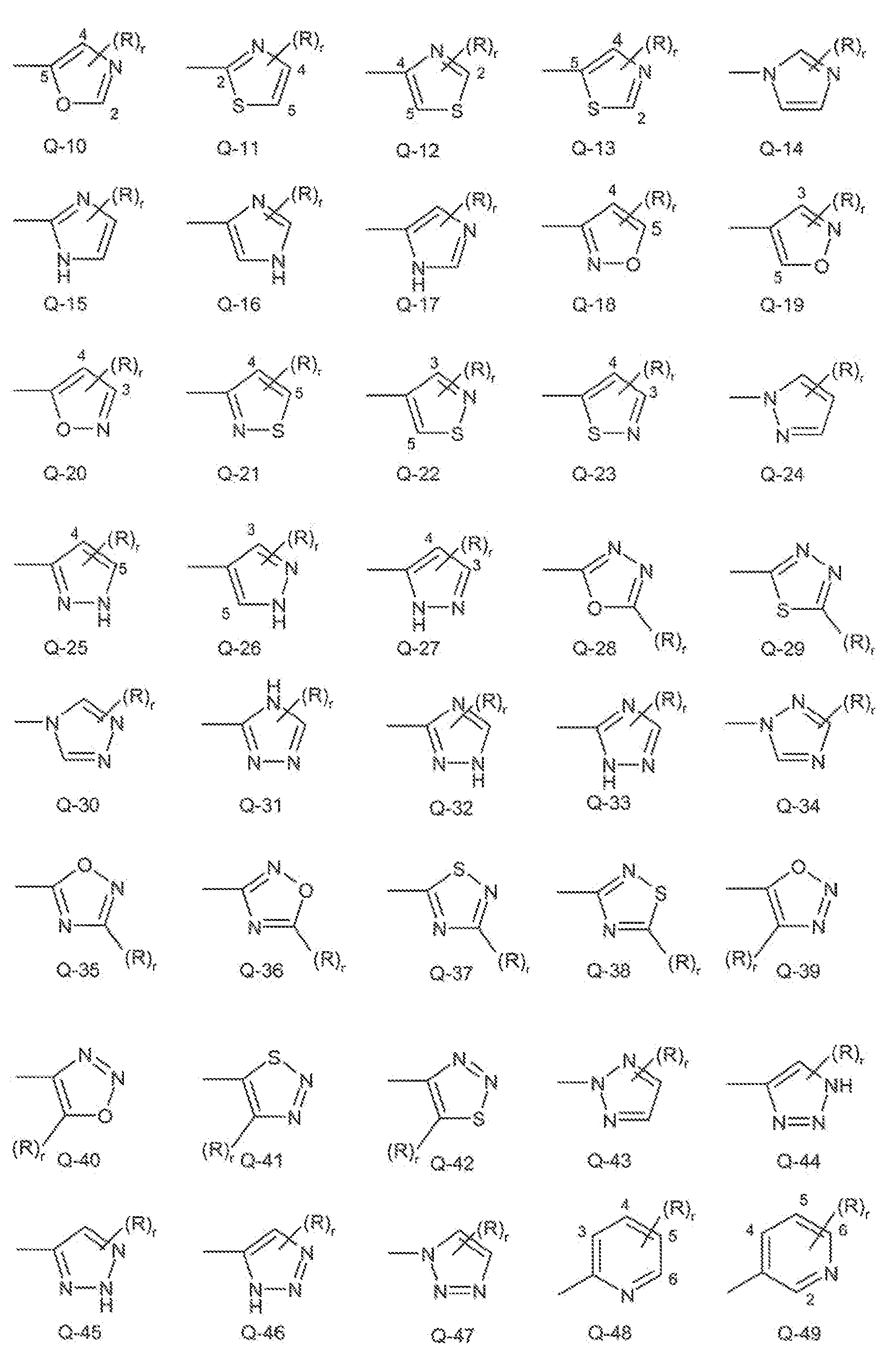WO2010079077A1 - Isoxazolines derivatives and their use as pesticide - Google Patents
Isoxazolines derivatives and their use as pesticide Download PDFInfo
- Publication number
- WO2010079077A1 WO2010079077A1 PCT/EP2009/067437 EP2009067437W WO2010079077A1 WO 2010079077 A1 WO2010079077 A1 WO 2010079077A1 EP 2009067437 W EP2009067437 W EP 2009067437W WO 2010079077 A1 WO2010079077 A1 WO 2010079077A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- crc
- halogen
- group
- formula
- compound
- Prior art date
Links
- 239000000575 pesticide Substances 0.000 title description 5
- 150000002547 isoxazolines Chemical class 0.000 title description 2
- 150000001875 compounds Chemical class 0.000 claims abstract description 82
- 241001465754 Metazoa Species 0.000 claims abstract description 34
- 244000045947 parasite Species 0.000 claims abstract description 17
- 239000000203 mixture Substances 0.000 claims description 52
- 229910052736 halogen Inorganic materials 0.000 claims description 44
- 239000004480 active ingredient Substances 0.000 claims description 43
- -1 2-substituted phenyl Chemical group 0.000 claims description 40
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 claims description 38
- 150000002367 halogens Chemical class 0.000 claims description 36
- JCXJVPUVTGWSNB-UHFFFAOYSA-N Nitrogen dioxide Chemical compound O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 claims description 34
- 125000000623 heterocyclic group Chemical group 0.000 claims description 27
- 150000003254 radicals Chemical group 0.000 claims description 27
- 238000000034 method Methods 0.000 claims description 25
- 125000001424 substituent group Chemical group 0.000 claims description 20
- 229910052757 nitrogen Inorganic materials 0.000 claims description 19
- 238000002360 preparation method Methods 0.000 claims description 18
- 125000001997 phenyl group Chemical class [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 16
- 125000005843 halogen group Chemical group 0.000 claims description 15
- 125000004122 cyclic group Chemical group 0.000 claims description 13
- 125000001797 benzyl group Chemical class [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 10
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 8
- 229910052760 oxygen Inorganic materials 0.000 claims description 8
- 230000008569 process Effects 0.000 claims description 8
- 229910052717 sulfur Inorganic materials 0.000 claims description 8
- 150000001204 N-oxides Chemical class 0.000 claims description 7
- 125000000951 phenoxy group Chemical class [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 7
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 7
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 claims description 6
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 6
- 125000005842 heteroatom Chemical group 0.000 claims description 5
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 5
- 229940124530 sulfonamide Drugs 0.000 claims description 5
- 150000003456 sulfonamides Chemical class 0.000 claims description 5
- 239000002270 dispersing agent Substances 0.000 claims description 3
- 125000000094 2-phenylethyl group Chemical class [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 claims description 2
- 125000004737 (C1-C6) haloalkoxy group Chemical group 0.000 claims 1
- 239000008194 pharmaceutical composition Substances 0.000 claims 1
- 125000006513 pyridinyl methyl group Chemical group 0.000 claims 1
- 125000005344 pyridylmethyl group Chemical group [H]C1=C([H])C([H])=C([H])C(=N1)C([H])([H])* 0.000 claims 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 claims 1
- 150000003839 salts Chemical group 0.000 abstract description 14
- 244000078703 ectoparasite Species 0.000 abstract description 9
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 abstract description 2
- 150000003230 pyrimidines Chemical class 0.000 abstract 1
- 239000000243 solution Substances 0.000 description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 24
- 238000006243 chemical reaction Methods 0.000 description 19
- 239000012074 organic phase Substances 0.000 description 18
- 238000009472 formulation Methods 0.000 description 17
- 241000607479 Yersinia pestis Species 0.000 description 16
- 230000000694 effects Effects 0.000 description 16
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 15
- 239000012043 crude product Substances 0.000 description 15
- 229920006395 saturated elastomer Polymers 0.000 description 15
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 14
- 241000238876 Acari Species 0.000 description 13
- 239000002904 solvent Substances 0.000 description 13
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 12
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 12
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 12
- 239000002253 acid Substances 0.000 description 12
- 241000238631 Hexapoda Species 0.000 description 11
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 11
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 10
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 10
- 238000011894 semi-preparative HPLC Methods 0.000 description 10
- 239000004544 spot-on Substances 0.000 description 10
- 239000007864 aqueous solution Substances 0.000 description 9
- 150000002148 esters Chemical class 0.000 description 9
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 9
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 9
- 239000007787 solid Substances 0.000 description 9
- 235000014113 dietary fatty acids Nutrition 0.000 description 8
- 239000000194 fatty acid Substances 0.000 description 8
- 229930195729 fatty acid Natural products 0.000 description 8
- 239000004540 pour-on Substances 0.000 description 8
- 239000011541 reaction mixture Substances 0.000 description 8
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 7
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 7
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 7
- 239000003085 diluting agent Substances 0.000 description 7
- 239000007788 liquid Substances 0.000 description 7
- 230000014759 maintenance of location Effects 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- 229910052739 hydrogen Inorganic materials 0.000 description 6
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 6
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 6
- 238000000746 purification Methods 0.000 description 6
- 239000003826 tablet Substances 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 5
- 239000007832 Na2SO4 Substances 0.000 description 5
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 241000258242 Siphonaptera Species 0.000 description 5
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 5
- 239000012141 concentrate Substances 0.000 description 5
- 239000013078 crystal Substances 0.000 description 5
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 5
- 235000013772 propylene glycol Nutrition 0.000 description 5
- 229960004063 propylene glycol Drugs 0.000 description 5
- 229910052938 sodium sulfate Inorganic materials 0.000 description 5
- 239000004094 surface-active agent Substances 0.000 description 5
- 241000283690 Bos taurus Species 0.000 description 4
- 241000255925 Diptera Species 0.000 description 4
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 4
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 4
- 241001494479 Pecora Species 0.000 description 4
- 241001674048 Phthiraptera Species 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 4
- 229920002472 Starch Polymers 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 125000003118 aryl group Chemical group 0.000 description 4
- 239000002585 base Substances 0.000 description 4
- 235000019445 benzyl alcohol Nutrition 0.000 description 4
- 239000002775 capsule Substances 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 239000001257 hydrogen Substances 0.000 description 4
- 239000002917 insecticide Substances 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- 230000003071 parasitic effect Effects 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 230000007480 spreading Effects 0.000 description 4
- 238000003892 spreading Methods 0.000 description 4
- 235000019698 starch Nutrition 0.000 description 4
- 239000008107 starch Substances 0.000 description 4
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 241000283086 Equidae Species 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 241000282326 Felis catus Species 0.000 description 3
- 241000257303 Hymenoptera Species 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 241000282887 Suidae Species 0.000 description 3
- 239000013543 active substance Substances 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 239000003054 catalyst Substances 0.000 description 3
- 235000013601 eggs Nutrition 0.000 description 3
- 150000002170 ethers Chemical class 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 244000000013 helminth Species 0.000 description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 3
- 239000012528 membrane Substances 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 235000019198 oils Nutrition 0.000 description 3
- 150000002923 oximes Chemical class 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 239000012071 phase Substances 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 239000011148 porous material Substances 0.000 description 3
- 229910000027 potassium carbonate Inorganic materials 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 239000005871 repellent Substances 0.000 description 3
- 230000002940 repellent Effects 0.000 description 3
- 229920002545 silicone oil Polymers 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- 239000007921 spray Substances 0.000 description 3
- 150000003512 tertiary amines Chemical class 0.000 description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 description 3
- 239000008158 vegetable oil Substances 0.000 description 3
- WYRSGXAIHNMKOL-UHFFFAOYSA-N $l^{1}-sulfanylethane Chemical compound CC[S] WYRSGXAIHNMKOL-UHFFFAOYSA-N 0.000 description 2
- QSLPNSWXUQHVLP-UHFFFAOYSA-N $l^{1}-sulfanylmethane Chemical compound [S]C QSLPNSWXUQHVLP-UHFFFAOYSA-N 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 2
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 2
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 2
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 2
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 2
- 241000251468 Actinopterygii Species 0.000 description 2
- 239000005995 Aluminium silicate Substances 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- 241000271566 Aves Species 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 241000700198 Cavia Species 0.000 description 2
- 241000134426 Ceratopogonidae Species 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 2
- 241001124179 Chrysops Species 0.000 description 2
- 241000254173 Coleoptera Species 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- 241000699800 Cricetinae Species 0.000 description 2
- 101100114828 Drosophila melanogaster Orai gene Proteins 0.000 description 2
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 2
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical group C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N Formic acid Chemical compound OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 241000287828 Gallus gallus Species 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 241000255777 Lepidoptera Species 0.000 description 2
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 241000244206 Nematoda Species 0.000 description 2
- 239000005642 Oleic acid Substances 0.000 description 2
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- 241000283898 Ovis Species 0.000 description 2
- 235000019483 Peanut oil Nutrition 0.000 description 2
- 241000286209 Phasianidae Species 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical class O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 241000255628 Tabanidae Species 0.000 description 2
- 241001454295 Tetranychidae Species 0.000 description 2
- 241000256856 Vespidae Species 0.000 description 2
- 230000000895 acaricidal effect Effects 0.000 description 2
- 239000000642 acaricide Substances 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 2
- 235000012211 aluminium silicate Nutrition 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 239000008346 aqueous phase Substances 0.000 description 2
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 2
- 125000002619 bicyclic group Chemical group 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 239000004359 castor oil Substances 0.000 description 2
- 235000019438 castor oil Nutrition 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 235000013330 chicken meat Nutrition 0.000 description 2
- 235000020971 citrus fruits Nutrition 0.000 description 2
- 239000003240 coconut oil Substances 0.000 description 2
- 235000019864 coconut oil Nutrition 0.000 description 2
- 230000002508 compound effect Effects 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 2
- 229940093471 ethyl oleate Drugs 0.000 description 2
- 150000002191 fatty alcohols Chemical class 0.000 description 2
- YYJNOYZRYGDPNH-MFKUBSTISA-N fenpyroximate Chemical compound C=1C=C(C(=O)OC(C)(C)C)C=CC=1CO/N=C/C=1C(C)=NN(C)C=1OC1=CC=CC=C1 YYJNOYZRYGDPNH-MFKUBSTISA-N 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 150000002334 glycols Chemical class 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 125000004438 haloalkoxy group Chemical group 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 150000007529 inorganic bases Chemical class 0.000 description 2
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 2
- HJOVHMDZYOCNQW-UHFFFAOYSA-N isophorone Chemical compound CC1=CC(=O)CC(C)(C)C1 HJOVHMDZYOCNQW-UHFFFAOYSA-N 0.000 description 2
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 2
- 150000002576 ketones Chemical class 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 244000144972 livestock Species 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 229920000609 methyl cellulose Polymers 0.000 description 2
- 239000001923 methylcellulose Substances 0.000 description 2
- 235000010981 methylcellulose Nutrition 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 239000000312 peanut oil Substances 0.000 description 2
- 229940068917 polyethylene glycols Drugs 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 239000008159 sesame oil Substances 0.000 description 2
- 235000011803 sesame oil Nutrition 0.000 description 2
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- 239000002689 soil Substances 0.000 description 2
- 239000003549 soybean oil Substances 0.000 description 2
- 235000012424 soybean oil Nutrition 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 235000001508 sulfur Nutrition 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 150000003626 triacylglycerols Chemical class 0.000 description 2
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 235000013311 vegetables Nutrition 0.000 description 2
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N (R)-alpha-Tocopherol Natural products OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- PZNPLUBHRSSFHT-RRHRGVEJSA-N 1-hexadecanoyl-2-octadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[C@@H](COP([O-])(=O)OCC[N+](C)(C)C)COC(=O)CCCCCCCCCCCCCCC PZNPLUBHRSSFHT-RRHRGVEJSA-N 0.000 description 1
- UUFQTNFCRMXOAE-UHFFFAOYSA-N 1-methylmethylene Chemical compound C[CH] UUFQTNFCRMXOAE-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- KAESVJOAVNADME-UHFFFAOYSA-N 1H-pyrrole Natural products C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 1
- FJRPOHLDJUJARI-UHFFFAOYSA-N 2,3-dihydro-1,2-oxazole Chemical compound C1NOC=C1 FJRPOHLDJUJARI-UHFFFAOYSA-N 0.000 description 1
- ZFFMLCVRJBZUDZ-UHFFFAOYSA-N 2,3-dimethylbutane Chemical group CC(C)C(C)C ZFFMLCVRJBZUDZ-UHFFFAOYSA-N 0.000 description 1
- IQHSSYROJYPFDV-UHFFFAOYSA-N 2-bromo-1,3-dichloro-5-(trifluoromethyl)benzene Chemical group FC(F)(F)C1=CC(Cl)=C(Br)C(Cl)=C1 IQHSSYROJYPFDV-UHFFFAOYSA-N 0.000 description 1
- GTPNXFKONRIHRW-UHFFFAOYSA-N 2-iodo-1,3,5-trimethylbenzene Chemical compound CC1=CC(C)=C(I)C(C)=C1 GTPNXFKONRIHRW-UHFFFAOYSA-N 0.000 description 1
- 125000004105 2-pyridyl group Chemical class N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- FDMFUZHCIRHGRG-UHFFFAOYSA-N 3,3,3-trifluoroprop-1-ene Chemical compound FC(F)(F)C=C FDMFUZHCIRHGRG-UHFFFAOYSA-N 0.000 description 1
- 125000003349 3-pyridyl group Chemical class N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000339 4-pyridyl group Chemical class N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000002373 5 membered heterocyclic group Chemical group 0.000 description 1
- 125000004070 6 membered heterocyclic group Chemical group 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 241000256111 Aedes <genus> Species 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 241000256186 Anopheles <genus> Species 0.000 description 1
- 241001427556 Anoplura Species 0.000 description 1
- 241000272517 Anseriformes Species 0.000 description 1
- 241001480748 Argas Species 0.000 description 1
- 241000204727 Ascaridia Species 0.000 description 1
- 241000244186 Ascaris Species 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- 241001674044 Blattodea Species 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 238000006443 Buchwald-Hartwig cross coupling reaction Methods 0.000 description 1
- 241000282832 Camelidae Species 0.000 description 1
- 241000282421 Canidae Species 0.000 description 1
- 241000282465 Canis Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 241000700114 Chinchillidae Species 0.000 description 1
- 241000255930 Chironomidae Species 0.000 description 1
- 229920002101 Chitin Polymers 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 241001307956 Chorioptes bovis Species 0.000 description 1
- 241000207199 Citrus Species 0.000 description 1
- 244000060011 Cocos nucifera Species 0.000 description 1
- 241000694440 Colpidium aqueous Species 0.000 description 1
- 241001126268 Cooperia Species 0.000 description 1
- 241000239250 Copepoda Species 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- 241000238424 Crustacea Species 0.000 description 1
- 241000258922 Ctenocephalides Species 0.000 description 1
- 241000490513 Ctenocephalides canis Species 0.000 description 1
- 241000258924 Ctenocephalides felis Species 0.000 description 1
- 241000256054 Culex <genus> Species 0.000 description 1
- 241000268912 Damalinia Species 0.000 description 1
- 241001128004 Demodex Species 0.000 description 1
- 241000202828 Dermatobia hominis Species 0.000 description 1
- 241001147667 Dictyocaulus Species 0.000 description 1
- 241000709823 Dictyoptera <beetle genus> Species 0.000 description 1
- 102100025027 E3 ubiquitin-protein ligase TRIM69 Human genes 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 241000283074 Equus asinus Species 0.000 description 1
- 241000371383 Fannia Species 0.000 description 1
- 241000242711 Fasciola hepatica Species 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 239000001828 Gelatine Substances 0.000 description 1
- 241000257324 Glossina <genus> Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 241000219146 Gossypium Species 0.000 description 1
- 241001480796 Haemaphysalis Species 0.000 description 1
- 241000257224 Haematobia Species 0.000 description 1
- 241000790933 Haematopinus Species 0.000 description 1
- 241000562576 Haematopota Species 0.000 description 1
- 241000243976 Haemonchus Species 0.000 description 1
- 241000258937 Hemiptera Species 0.000 description 1
- 241000920462 Heterakis Species 0.000 description 1
- 241001480224 Heterodera Species 0.000 description 1
- 241001466007 Heteroptera Species 0.000 description 1
- 101000830203 Homo sapiens E3 ubiquitin-protein ligase TRIM69 Proteins 0.000 description 1
- 235000008694 Humulus lupulus Nutrition 0.000 description 1
- 244000025221 Humulus lupulus Species 0.000 description 1
- 241000257174 Hypoderma lineatum Species 0.000 description 1
- 206010061217 Infestation Diseases 0.000 description 1
- 241000575946 Ione Species 0.000 description 1
- 241001495069 Ischnocera Species 0.000 description 1
- 241000256602 Isoptera Species 0.000 description 1
- 241000238681 Ixodes Species 0.000 description 1
- 241000545319 Ixodes canisuga Species 0.000 description 1
- 239000004166 Lanolin Substances 0.000 description 1
- LTXREWYXXSTFRX-QGZVFWFLSA-N Linagliptin Chemical compound N=1C=2N(C)C(=O)N(CC=3N=C4C=CC=CC4=C(C)N=3)C(=O)C=2N(CC#CC)C=1N1CCC[C@@H](N)C1 LTXREWYXXSTFRX-QGZVFWFLSA-N 0.000 description 1
- 241001113970 Linognathus Species 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241000771994 Melophagus ovinus Species 0.000 description 1
- 101100059320 Mus musculus Ccdc85b gene Proteins 0.000 description 1
- 241000257229 Musca <genus> Species 0.000 description 1
- 241000257159 Musca domestica Species 0.000 description 1
- 241001124166 Musca vetustissima Species 0.000 description 1
- 241000257226 Muscidae Species 0.000 description 1
- 241000282339 Mustela Species 0.000 description 1
- 241000772415 Neovison vison Species 0.000 description 1
- 235000002637 Nicotiana tabacum Nutrition 0.000 description 1
- 244000061176 Nicotiana tabacum Species 0.000 description 1
- 241000272458 Numididae Species 0.000 description 1
- 241000543819 Oestrus ovis Species 0.000 description 1
- 241000238814 Orthoptera Species 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 241001480755 Otobius Species 0.000 description 1
- 241000904715 Oxyuris Species 0.000 description 1
- 241000244187 Parascaris Species 0.000 description 1
- 235000008673 Persea americana Nutrition 0.000 description 1
- 244000025272 Persea americana Species 0.000 description 1
- 241000255129 Phlebotominae Species 0.000 description 1
- 229920002556 Polyethylene Glycol 300 Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920001710 Polyorthoester Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 241001415024 Psocoptera Species 0.000 description 1
- 241000255131 Psychodidae Species 0.000 description 1
- 235000019484 Rapeseed oil Nutrition 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 241001480809 Rhipicentor Species 0.000 description 1
- 241001481703 Rhipicephalus <genus> Species 0.000 description 1
- 235000004443 Ricinus communis Nutrition 0.000 description 1
- 241000304160 Sarcophaga carnaria Species 0.000 description 1
- 241000257185 Sarcophagidae Species 0.000 description 1
- 241000894243 Sericata Species 0.000 description 1
- 229910008433 SnCU Inorganic materials 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- 239000005708 Sodium hypochlorite Substances 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 241000044136 Solenopotes Species 0.000 description 1
- 229930182558 Sterol Natural products 0.000 description 1
- 241001494115 Stomoxys calcitrans Species 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 238000006069 Suzuki reaction reaction Methods 0.000 description 1
- 241000255626 Tabanus <genus> Species 0.000 description 1
- 241001669733 Tabanus nigrovittatus Species 0.000 description 1
- 241000607216 Toxascaris Species 0.000 description 1
- 241000244031 Toxocara Species 0.000 description 1
- 241000869417 Trematodes Species 0.000 description 1
- 241001489151 Trichuris Species 0.000 description 1
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 241000571986 Uncinaria Species 0.000 description 1
- LEHOTFFKMJEONL-UHFFFAOYSA-N Uric Acid Chemical compound N1C(=O)NC(=O)C2=C1NC(=O)N2 LEHOTFFKMJEONL-UHFFFAOYSA-N 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000016383 Zea mays subsp huehuetenangensis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 229920002494 Zein Polymers 0.000 description 1
- 108010055615 Zein Proteins 0.000 description 1
- 241001414985 Zygentoma Species 0.000 description 1
- GCCZDWOUVDUKRF-UHFFFAOYSA-N [NH2+2]C1C(CC2)CC2C1 Chemical compound [NH2+2]C1C(CC2)CC2C1 GCCZDWOUVDUKRF-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 125000005396 acrylic acid ester group Chemical group 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- WNLRTRBMVRJNCN-UHFFFAOYSA-L adipate(2-) Chemical compound [O-]C(=O)CCCCC([O-])=O WNLRTRBMVRJNCN-UHFFFAOYSA-L 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 230000009418 agronomic effect Effects 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000003674 animal food additive Substances 0.000 description 1
- 230000000507 anthelmentic effect Effects 0.000 description 1
- 239000000921 anthelmintic agent Substances 0.000 description 1
- 229940124339 anthelmintic agent Drugs 0.000 description 1
- 230000002141 anti-parasite Effects 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 239000005667 attractant Substances 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- UHOVQNZJYSORNB-UHFFFAOYSA-N benzene Substances C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 239000003139 biocide Substances 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 150000001649 bromium compounds Chemical class 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 125000005997 bromomethyl group Chemical group 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 229920003086 cellulose ether Polymers 0.000 description 1
- 235000013339 cereals Nutrition 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 230000031902 chemoattractant activity Effects 0.000 description 1
- 239000012320 chlorinating reagent Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 239000007931 coated granule Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 229940075614 colloidal silicon dioxide Drugs 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- GBRBMTNGQBKBQE-UHFFFAOYSA-L copper;diiodide Chemical compound I[Cu]I GBRBMTNGQBKBQE-UHFFFAOYSA-L 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 238000006880 cross-coupling reaction Methods 0.000 description 1
- 150000001954 decanoic acid esters Chemical class 0.000 description 1
- 235000013681 dietary sucrose Nutrition 0.000 description 1
- FFHWGQQFANVOHV-UHFFFAOYSA-N dimethyldioxirane Chemical compound CC1(C)OO1 FFHWGQQFANVOHV-UHFFFAOYSA-N 0.000 description 1
- 150000004844 dioxiranes Chemical class 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- FLVVJZDJSIUVQK-UHFFFAOYSA-N dodecanoic acid;oxalic acid Chemical compound OC(=O)C(O)=O.CCCCCCCCCCCC(O)=O FLVVJZDJSIUVQK-UHFFFAOYSA-N 0.000 description 1
- 235000013399 edible fruits Nutrition 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 235000019197 fats Nutrition 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 229940074076 glycerol formal Drugs 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 125000003976 glyceryl group Chemical group [H]C([*])([H])C(O[H])([H])C(O[H])([H])[H] 0.000 description 1
- 239000003630 growth substance Substances 0.000 description 1
- 125000004995 haloalkylthio group Chemical group 0.000 description 1
- 125000001475 halogen functional group Chemical group 0.000 description 1
- 230000009931 harmful effect Effects 0.000 description 1
- 230000012447 hatching Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 244000144980 herd Species 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 150000004678 hydrides Chemical class 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 239000008172 hydrogenated vegetable oil Substances 0.000 description 1
- 150000002432 hydroperoxides Chemical class 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 230000000749 insecticidal effect Effects 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- CTAPFRYPJLPFDF-UHFFFAOYSA-N isoxazole Chemical compound C=1C=NOC=1 CTAPFRYPJLPFDF-UHFFFAOYSA-N 0.000 description 1
- 229930014550 juvenile hormone Natural products 0.000 description 1
- 239000002949 juvenile hormone Substances 0.000 description 1
- 150000003633 juvenile hormone derivatives Chemical class 0.000 description 1
- 229930191400 juvenile hormones Natural products 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- 229940039717 lanolin Drugs 0.000 description 1
- 235000019388 lanolin Nutrition 0.000 description 1
- 229940099367 lanolin alcohols Drugs 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 231100001231 less toxic Toxicity 0.000 description 1
- 239000000944 linseed oil Substances 0.000 description 1
- 235000021388 linseed oil Nutrition 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 235000009973 maize Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229940057917 medium chain triglycerides Drugs 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229940105132 myristate Drugs 0.000 description 1
- BJXFCYHVICCYIU-UHFFFAOYSA-N naphthalene-1-carbaldehyde Chemical compound C1=CC=C[C]2C(C=O)=CC=C=C21 BJXFCYHVICCYIU-UHFFFAOYSA-N 0.000 description 1
- 239000005445 natural material Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000003883 ointment base Substances 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 230000000590 parasiticidal effect Effects 0.000 description 1
- 239000002297 parasiticide Substances 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N pentanoic acid group Chemical class C(CCCC)(=O)O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 150000004965 peroxy acids Chemical class 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- XNGIFLGASWRNHJ-UHFFFAOYSA-L phthalate(2-) Chemical compound [O-]C(=O)C1=CC=CC=C1C([O-])=O XNGIFLGASWRNHJ-UHFFFAOYSA-L 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 239000010665 pine oil Substances 0.000 description 1
- 229940068196 placebo Drugs 0.000 description 1
- 239000000902 placebo Substances 0.000 description 1
- 239000003880 polar aprotic solvent Substances 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 239000002745 poly(ortho ester) Substances 0.000 description 1
- 150000004291 polyenes Chemical class 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 235000012015 potatoes Nutrition 0.000 description 1
- 244000144977 poultry Species 0.000 description 1
- 235000013594 poultry meat Nutrition 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000012521 purified sample Substances 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 239000010499 rapseed oil Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 125000006413 ring segment Chemical group 0.000 description 1
- 235000021003 saturated fats Nutrition 0.000 description 1
- 239000012047 saturated solution Substances 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L sodium carbonate Substances [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- SUKJFIGYRHOWBL-UHFFFAOYSA-N sodium hypochlorite Chemical compound [Na+].Cl[O-] SUKJFIGYRHOWBL-UHFFFAOYSA-N 0.000 description 1
- 229960001922 sodium perborate Drugs 0.000 description 1
- YKLJGMBLPUQQOI-UHFFFAOYSA-M sodium;oxidooxy(oxo)borane Chemical compound [Na+].[O-]OB=O YKLJGMBLPUQQOI-UHFFFAOYSA-M 0.000 description 1
- QGLITUFXHVRMGV-UHFFFAOYSA-M sodium;tetratriacontyl sulfate Chemical compound [Na+].CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCOS([O-])(=O)=O QGLITUFXHVRMGV-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 239000001590 sorbitan monolaureate Substances 0.000 description 1
- 235000011067 sorbitan monolaureate Nutrition 0.000 description 1
- 239000008347 soybean phospholipid Substances 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 238000011146 sterile filtration Methods 0.000 description 1
- 235000003702 sterols Nutrition 0.000 description 1
- 150000003432 sterols Chemical class 0.000 description 1
- 239000003351 stiffener Substances 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000004548 suspo-emulsion Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 description 1
- TUNFSRHWOTWDNC-UHFFFAOYSA-N tetradecanoic acid Chemical compound CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 230000036962 time dependent Effects 0.000 description 1
- AOBORMOPSGHCAX-DGHZZKTQSA-N tocofersolan Chemical compound OCCOC(=O)CCC(=O)OC1=C(C)C(C)=C2O[C@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C AOBORMOPSGHCAX-DGHZZKTQSA-N 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 229940099259 vaseline Drugs 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 210000002268 wool Anatomy 0.000 description 1
- 239000005019 zein Substances 0.000 description 1
- 229940093612 zein Drugs 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing aromatic rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/10—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof
- A01N47/16—Carbamic acid derivatives, i.e. containing the group —O—CO—N<; Thio analogues thereof the nitrogen atom being part of a heterocyclic ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/14—Ectoparasiticides, e.g. scabicides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
Definitions
- This invention relates to novei isoxazoltnes, their N-oxsdes and salts, processes for their manufacture, their use in the controi of ectoparasites, especially insects and acari. on non- hur ⁇ an animais, especiaily productive livestock and domestic animals, and furthermore pestic ⁇ dai compositions which contain one or more of these compounds.
- each of A 1 -A 8 and 8,-B 3 are C(R 3 ), Ri is haioaikyl and W is O or S.
- the compounds are mainly used in the control of invertebrate pests in agronomic environments. Many products are commercially available for these purposes, but the need continues for new compounds that are more effective, less costly, less toxic, environmentally safer or have different modes of action. It now has been surprisingly found that novel naphthyl derivatives with a modified heterocyclic side chain have superior properties in the control of pests.
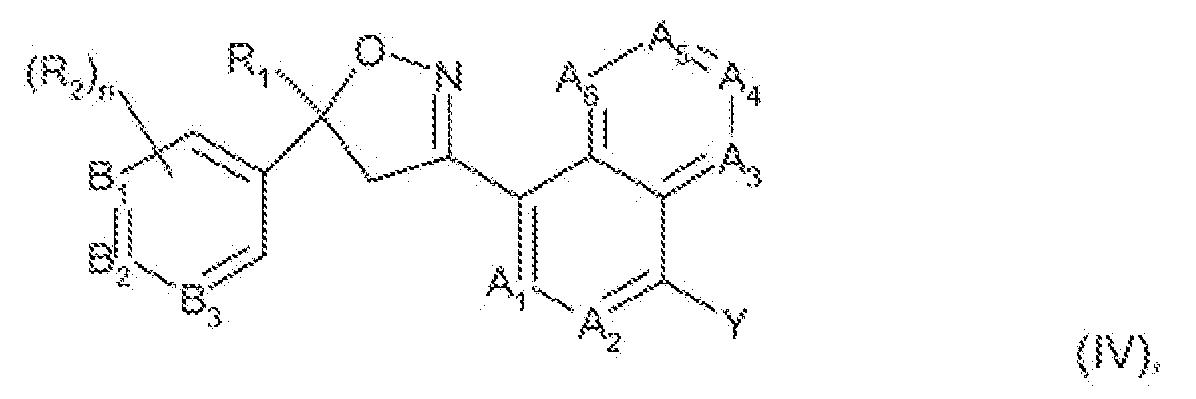
- This present invention is directed to a compound of formula
- Ai, A 2 , A 3 , A ⁇ 5 A 5 and A 6 are each independently selected from the group consisting of CR 3 ' and N; n is an integer from 0 to 4;
- Bi, 8 2 sn ⁇ 8 3 are each independently selected from the group consisting of CfV and N; each R 2 ' is independently of the other H or R 2 ; each R 3 ' is independently of the other H or R 3 ;
- Ri is C-rCVaikyi, C 2 -C 6 -alkenyi, C 2 -C 6 -aikynyJ, CVCs-cycloaikyi, C 4 -Craikylcycioa!ky1 or C 4 -
- each R 2 is independently halogen, CrC e ⁇ aiky!, CrC ⁇ ⁇ hafoaiky!, d-Cs-aikoxy, C 1 -C 6 - haioaikoxy, C r C 8 ⁇ aiky!thio, C r C 8 ⁇ haioalkyithio, C r C 6 -aikyisu!firtyS, C r C 6 -haioaiky1suifiny!,
- R 4 is haiog ⁇ fi, C r C t raikyi, C r C 6 -aikoxy, d-C ⁇ -aikyithio, CrC 6 -a!ky!s ⁇ lf ⁇ nyi, CrC 6 ⁇ aikyi- suSfonyi, -CN or -NO 2 ;
- Q is a 5- or 6 ⁇ membered heterocyclic ring, or a QrCio ⁇ carbocyciic ring system or a 8», 9- or
- Ai, A 2 , A 3 , Aj, A 5 and A 8 are each independently selected from the group consisting of CR 3 ' and N; n is an integer from 0 to 4;
- B 1 , S 2 and B 3 are each independently selected from the group consisting of CR 2 ' and N; each R 2 ' is independently of the other H or R 2 ; each R 3 ' is independently of the other H or R 3 ;
- R t is CrC ⁇ -aikyi, C 2 ⁇ C 6 ⁇ alkenyi, C 2 -CVa!kyny!, QrQrCyct ⁇ aiky!, C ⁇ CrSikylcycioalkyl or C 4 -
- each R 2 is independently halogen, Ci-C «-alkyl, CrC ⁇ -haioalkyl, Ci-C 6 -alkoxy, CrC 6 - haloaikoxy, C r C 6 -aiky!thio, Ci-C 6 -ha!oaikyithio, CrCg-aikylsuifinyl, CrCVhaioalkyisuifinyi,
- each R 3 Is independently halogen, CyCe-alkyi, CrC f i-haloaikyl, Ca-Cg-cycioalkyl, CrC fi ⁇ haiccycloalkyi, CrC ⁇ -aikoxy, CrCe-haloalkoxy, C r C 6 -a!kylthio, C r Cs-haloaikySthio, CrC 6 aikyl-suSfinyl, Ci-C 6 -haioaikyisu!finy! ( CrC 6 -a!kylsuifony
- R 4 is halogen, Ci-C ⁇ -alkyi « CrC ⁇ -aikoxy, Ci-C 6 ⁇ a!ky!thio, Ci-C ⁇ -aikylsulfi ⁇ yi, Ci-C 6 -a!kyi- suifonyl, -CN or -NO 2 ; and
- Q is a 5- or ⁇ -membered heterocyclic ring, or a Cg-Cio-carbocyclic ring system or a 8-, 9- or
- Ce-alkoxycarbonyl suifonamido, N-mono- or N 1 N, di-CrC ⁇ -alkyfsulfonamido, N-mono- or
- TNs invention aiso provides a composition comprising a compound of formula (S) 1 an N-oxide or a sait thereof, and at least one additional component selected from the group consisting of a surfactant, a solid diluent &n ⁇ a liquid diluent.
- this invention also provides a composition for controiling parasites, in particular ectoparasites, comprising a biologlcaHy effective amount of a compound of formula (!), an N-oxsde or a sait thereof, and at ieast one additional component selected from the group consisting of a surfactant, a soiid diluent and a liquid diluent, said composition optionally further comprising a biologically effective amount of at ieast one additional biologically active compound or agent.
- This invention further provides the composition described above in the form of a bait composition wherein the solid diiuent and/or the liquid diluent comprises one or more food materials, said composition optionally comprising an attractant and/or a bumectant
- This invention further provides a trap device for controlling parasites, in particular ectoparasites, comprising said bait composition and a housing adapted to receive said bait composition, wherein the housing has at ieast one opening sized to permit the parasites to pass through the opening, so the invertebrate pest can gain access to said bait composition from a location outside the housing, and wherein the housing is further adapted to be placed in or near a iocus of potential or known activity for the parasites pest.
- This invention also provides a method for controiling parasites comprising contacting the parasites or their environment with a biologically effective amount of a compound of formula (1), an N-oxkJe or a sait thereof, (e.g., as a composition described herein).
- This invention also relates to such method wherein the parasites or their environment are contacted with a composition comprising a biologically effective amount of a compound of formula 0). an N- oxide or a sait thereof, and at least one additional component selected from the group consisting of a surfactant, a solid diluent and a liquid diluent, said composition optionally further comprising a biologically effective amount of at ieast one additional biologically active compound or agent.
- This invention also provides a composition for protecting an animal from an parasitic pest comprising a parasiticidaiiy effective amount of a compound of formula (I) Bn N-oxide or a salt thereof, and at least one carrier.
- the present invention further provides the composition described above in a form for orai administration.
- This invention also provides a method for protecting an animal from a parasitic pest comprising administering to the animsf a parasiticus Hy effective amount of a compound of formula (i), an N ⁇ oxide or a sait thereof.
- alkyS used either alone or in compound words such as "aikyithio" or “haSoafkyP includes straight-chain or branched aiky.1, such as, methyl, ethyl, n ⁇ propyl, i-pro ⁇ yi, or the different butyl, penty! or hexyi isomers.
- Alke ⁇ yi includes straight-chain or branched aikenes such as ethenyl, 1 ⁇ propenyl, 2- propenyi, and the different butenyi, pentenyl and hexenyl isomers.
- ASkenyi also includes polyenes such as 1 ,2- ⁇ ropadienyi and 2,4-hexadienyl.
- Alkynyf includes straight-chain or branched alkynes such as ethynyS, 1-propynyl, 2-propynyi and the different b ⁇ tynyS, pentynyi and hexynyi isomers.
- Alkynyi can also include moieties comprised of multiple triple bonds such as 2,5-hexadiynyi.
- Alkoxy includes, for example, methoxy, ethoxy, n-propytoxy, isopropyioxy and the different butoxy, perttoxy and hexyloxy isomers.
- Alkyithio includes branched or straight-chain aikyithio moieties such as methylthio, ethylthio, and the different propyithio, butyithio, pentyithio and hexyithio isomers.
- alkyisulfinyf Includes both enantiomers of an aikyls ⁇ ifinyi group.
- alkylsuifinyl include CH 3 S(O)-, CH 3 CH 2 S(O)-, CH 3 CH 2 CH 2 S(O)-, ⁇ CH 3 ) 2 CHS(O ⁇ - Bn ⁇ the different butylsuifinyi pentylsuifinyi and hexylsulfiny! isomers.
- alkylsulfonyl examples include CH 3 S(O) 2 -, CH 3 CH 2 S ⁇ O) r , CH 3 CH 2 CH 2 S(O) 2 -, (CH 3 ) J CHS(O) 2 -, and the different butyls ⁇ ifonyl, pertiylsuffony! and hexylsulfonyi isomers.
- N-alkyiamino “N.N-di-aikyamino”, and the like, are defined analogously to the above examples.
- Cyeioalkyi incS ⁇ des, for example, cyciopropyl, eyclobutyi, cyciopentyi and cyclohexyi
- aifcyicydoalkyi denotes alkyl substitution on a eyci ⁇ aikyi moiety and includes, for example, ethyteyciopropyl, i-propylcydobutyi, 3-methyicy ⁇ iopentyl and 4 ⁇ m ⁇ thy1cycl ⁇ hexyi
- cycloaikylaikyi denotes cycioaikyi substitution on an alkyl moiety.
- cycl ⁇ aikytaikyf examples include cyciopropyimethyi, cyetopentyiethyl, and other cycioaikyi moieties bonded to straight-chain or branched alkyi groups.
- halogen either atone or in compound words such as “haioalkyi” includes fluorine, chlorine, bromine or iodine. Further, when used in compound words such as “baioalkyf, said aikyi may be partially or fully substituted with halogen atoms which may be the same or different. Examples of “haioaikyP inciude F 3 C-, ClCH 2 -, CF 3 CH 2 - and CF 3 CCI 2 -. The terms “hafocyeloaikyf, “haloafkoxy”, “hal ⁇ aikyithio", and the like, &t® defined analogously to the term “haioaikyi”.
- haioaikoxy examples include CFjO-, CCI 3 CH 2 O-, HCFsCH 2 CH 2 O- and CF 3 CH 2 O-.
- haioaikoxy examples include CFjO-, CCI 3 CH 2 O-, HCFsCH 2 CH 2 O- and CF 3 CH 2 O-.
- haioaikoxy examples include CFjO-, CCI 3 CH 2 O-, HCFsCH 2 CH 2 O- and CF 3 CH 2 O-.
- Exampies of "haloaikyisulfinyl” include CF 3 S(O)-, CCi 3 S(O)-, CF 3 CH 2 S(O)- and CF 3 CF 2 S(O)-.
- hatoalkyteuHtonyT examples include CF 3 S(O) 2 -, CCi 3 S(O) 2 -, CF 3 CH 2 S(O) 2 - and CF 3 CF 2 S(O) 2 -.
- Alkyicarbonyl denotes a straight-chain or branched alky! moteties bonded to a C(-0) moiety.
- alkoxycarbonyl inciude CH 3 OC( ⁇ O)-, CH 3 CH 2 0C ⁇ 0), CH 3 CH 2 CH 2 OC( ⁇ O) ⁇ , (CH 3 ⁇ CH0C( ⁇ 0)- and the different butoxy- or pentoxycarbonyi isomers, for example tert.-butoxycarbo ⁇ yi (Boc).
- C 1 -Cf prefix where i and j are integers.
- CrC 4 alkylsulfonyi designates methyisuifonyl through butylsuifo ⁇ yi
- C 2 -alkoxyalkyl designates CH 3 OCH 2
- C 3 ⁇ alkoxyaikyl designates, for example, CH 3 CH(OCH 3 ), CH 3 OCH 2 CH 2 or CH 3 CH 2 OCH 2
- C 4 -alkoxyaikyi designates the various isomers of an aikyl group substituted with o,n aikoxy group containing a total of four carbon atoms, examples including CH 3 CH 2 CH 2 OCH 2 and CH 3 CH 2 OCH 2 CHr-
- heterocyclic ring or “heterocycfe” denote a ring in which at ieast one atom forming the ring backbone is not carbon, e.g., nitrogen, oxygen or sulfur. Typically a heterocyclic ring contains no more than 4 nitrogens, no more than 2 oxygens and no more than 2 sulfurs. Unless otherwise indicated, a heterocyclic ring can be a saturated, partially unsaturated, or fully unsaturated ring. When a fully unsaturated heterocyclic ring satisfies H ⁇ ckei's rule, then said ring is also called a “heteroaror ⁇ atic ring", “aromatic heterocyclic ring”. Unless otherwise indicated, heterocyclic rings and ring systems can be attached through any available carbon or nitrogen by replacement of a hydrogen on said carbon or nitrogen.
- Q is a 5- or 6 ⁇ membered nitrogen-containing heterocyclic ring, it may be attached to the remainder of formula (I) through any available carbon or nitrogen ring atom, unless otherwise described.
- Each R 2 is independently of the other preferably halogen, CrC ⁇ ⁇ haioalkyl, Ci-Ce haloalkoxy or -CN, more preferably halogen, CF 3 , GCF 3 or -CN, and in particular halogen.
- B 1 , S 2 and B 3 are each independently of the other preferred the group CR 2 ', wherein R 2 ' is H or R 2 , and for R 2 the above-given meanings and preferences apply. R 2 ' is most preferably H or halogen.
- n is meant to summarize all radicals R 2 in the ⁇ -mernbered ring, n is preferably am integer from 0 to 4, more preferably from 1 to 3, and in particular 2 or 3.
- R 1 is preferably CrC 6 -a!kyl optionally substituted with one or more s ⁇ bstituents independently selected from R 4 , more preferably CrC 3 -alky! optionally substituted with halogen, even more preferably CrC 3 -HaIOaIKyI, especially preferably CrCa-aiky! substituted with F, and in particular CF 3 .
- Each R 3 is independently of the other preferably halogen, CrC 4 - ⁇ lkyl, CrCt-hal ⁇ aikyi, C 3 -CV cycioaikyi, CrC ⁇ -alkoxy, CrC 4 -haioaikoxy, N-mono- or N,N-di-C r C 2 -a!kyiamino, -CN or -NO 2 , more preferably halogen, d-Qralkyj, CrC 2 -hafoa!kyi, cyclopropyS, Ci-C 2 -aikoxy, -CN or -NO 2 , and even more preferably halogen, C 1 -C 2 a!ky! ( CrC 2 afkoxy.-CN or -NO 2 .
- a 1 , A 2 , A 3 , A 4 , A 5 and Ag are each independently of the other preferably a group CR 3 '.
- R 3 ' is preferably H or R 3 , wherein for R 3 the above-given meanings and preferences apply.
- three of the radicals A L A 2 , A 3 , A 4 , A 5 an ⁇ A 6 are each CH and the other three radicals are either CH or CR 3 .
- AL A 5 , A 3 , A 4 , A 5 and A 5 are each CH.
- R 4 is preferably halogen, CrC 2 -aiky1, C,-C 2 ⁇ atkoxy, -CN or -NO 2 , more preferably halogen, - CN or -NO 2 , an ⁇ in particular halogen.
- Q is a C 6 ⁇ Ci 0 -carbocyclic ring system, for example phenyl, ⁇ aphthyi, i ⁇ trahydronaphthyi, i ⁇ danyf, indenyl, hydrindanyi or octahydro-pentaien, in particular phenyl, which is each ⁇ nsubstituted or substituted by one or more same or different substsiuents selected from the group of substituents as defined before for Q.
- Q is preferably phenyl which is substituted by 1 to 4, preferably 1 to 3 and in particular 1 or 2 same or different substituents selected from the group consisting of halogen, CrC 4 -alkyS, Cr&rhaioaikyl, CrC ⁇ alkoxy, CrC 4 -haioalkoxy, CrC.
- Q is more preferably phenyl, which is substituted by 1 to 3, in particular 1 or 2, same or dtfferent substituents selected from the group consisting of halogen, CrQraikyt, CrCrhaloalkyf, CrC 2 -alkoxy, Ci-C 2 -haioalkoxy, Cr C 2 «haloalkylthio, -CN, -NO 2 , and unsubstituted or halogen-, Ci-C 2 -alkyl-, d-Ca-haloalkyl-, Ci- Cr-altoxy-, d-C ⁇ -haioaikoxy', nitro- or cyano-subsiU ⁇ ted pheny! or phenoxy.
- substituents selected from the group consisting of halogen, CrQraikyt, CrCrhaloalkyf, CrC 2 -alkoxy, Ci-C 2 -haioalkoxy, Cr C 2 «haloalkylthio, -CN
- Q is a 5- or 6 ⁇ membered heterocyclic ring, which may be saturated or preferably unsaturated, &n ⁇ which is unsubstituted or substituted with one or more substituents selected from the group of substituents as defined before for Q.
- Pref ⁇ rr ⁇ d substituents of the heterocyclic ring Q are, for example, CrC ⁇ alkyt, CrCXrhaio* aikyi, C r C 4 -aikoxy, Ci-C ⁇ haioaikoxy, CrC ⁇ alkyiihio, CrC-haloalkylthio, d-Co-alkylsuifinyl, CrC 4 ⁇ hak>a!ky1sulfinyi t GrC 4 -aiky!suJfonyi GrCrhai ⁇ alkylsuifonyl, -CN, -NO 2 , C r C 4 ⁇ aikoxy- carbony!, N-mo ⁇ o- or N,N-di» CrCValkylcarbonyiamino, sulfonamide, CrC 3 ⁇ aikanoy!
- the heterocyclic ring Q are selected from the group consisting of halogen, C, ⁇ C 2 -alkyf, Ci-C 2 -ha!oaikyl, CrC 2 ⁇ alkoxy, CrC 2 -haloalkoxy, C 1 -C 2 - naloalkylthio, -CN, -NO 2 , and CrCs-alkoxycarbonyi, in particular CrC r aikyi, C r C 2 -haioalkyi and d-C ⁇ alkoxycarbo ⁇ yl,
- a suitable heterocyeiic ring is, for example, a 5 ⁇ or 6-membered heteroaromatic ring having from 1 to 4, preferably from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, which is further unsubstituted or substituted by one or more substituents as defined before for Q including the preferences given therefore.
- the heterocyclic radical Q is preferably substituted by 0 to 3, in particular 0, 1 or 2 substiluents from the group as defined before for O.
- Examples of a 5- or 6-membered unsaturated aromatic heterocyclic ring optionally substituted with from one or more substituents include the rings Q-1 through Q-80 illustrated in Exhibit 1 wherein R is any su ⁇ stituent as defined before for Q including the preferences given, and r is an integer from 0 to 4, limited by the number of available positions on each Q group.
- R is any su ⁇ stituent as defined before for Q including the preferences given
- r is an integer from 0 to 4, limited by the number of available positions on each Q group.
- Q «28, ⁇ Q-29, Q-35, Q-36, Q-37, Q-3B, Q ⁇ 3 ⁇ , Q-40, Q-41 and Q-42 have only one available position, for these Q groups r is limited to the integers 0 or 1 and r being 0 means that the Q group is unsubstituted and a hydrogen is present at the position indicated by (R),
- a further group of suitable heterocyclic radicals comprises, for example, a 5- or 8-membered heter ⁇ aiiphat ⁇ c or partly unsaturated ring having from 1 to 4, preferably from 1 to 3 same or different h ⁇ t ⁇ roatoms selected from the group consisting of N 1 O and S. which is further u ⁇ substitut ⁇ d or substituted by one or mom substituents as defined before for Q including the preferences given therefore.
- heteroaiiphatio or partly unsaturated rings include the radicals illustrated in Exhibit 2 below, wherein R and r are as defined above including the preferences given.
- preferred heterocyclic raclicaS Q is of formula wherei ⁇ r is an integer from 0 to 3 and R is independently selected from the group given for Q including the preferences.
- Q is particularly preferred the unsubstituted radical G-34, G-43 or Q-47, wherein r is 0 in each case.
- a further particularly preferred radical Q is a radical G-44, wherein r is an integer from 0 to 3, in particular 1 , and R is independency selected from the group given for Q including the preferences.
- Another preferred radical Q is a radical G-75, wherein r is an integer from 0 to 3, in particular 1 , and R is independently selected from the group given for Q including the
- a further preferred radical G is a radical G-5, Q-14, Q-24, G-25, Q-26, G-3G, Q-32, Q-34, Q- 43, Q-44, Q-47, Q-49, Q-50 or Q ⁇ 75, in particular a radical Q-24, Q-26, Q-30, Q-32, Q-34, Q-44 or Q-47, which is each unsubslituted or substituted by one or more substituents selected from the group consisting of CrC 2 ⁇ aikyi, halo- CrG 2 -SlKyS, halo- C 1 -C 2 - alkyicarbonyl, cyano, CrC t -aikoxycarbonyl ⁇ CrC ⁇ -alkoxycarbonylmethyi, carboxy-C t -Cr alkyi, aminocarbonyl, N-mono- or N ⁇ -di-Ci-Ca-afkytarfiinocarbonyi, halo- Ci-C 2 -
- a further particularly preferred radical G is a radicai G-5, Q-14, Q-24, Q-25, Q-26, Q-30, Q- 32, Q-34, Q-43, Q-44, Q-47, Q-49 Q-50 or Q-75, in particular a radical G-24, G ⁇ 2 ⁇ , Q-30, Q- 32, Q-34, Q-44 or Q-47, which is each unsubstituted or substituted by one substitu ⁇ nt selected from the group consisting of methyl, trifluoromethyi 2 ⁇ trifiuoroethyf, trifiuormethyicarbonyi, cyano, methoxycarbonyl, tert.-buloxycarbonyi, carboxymelhyl, ethoxy- carbonyimethyl, aminocarbonyl, N-metliytaminocarbony!, 2 ⁇ trif1uorethylaminocarbonyl- methyl, 24rifiuorethylaminocarbonyimethyiamino
- a suitable fused heierobicyciic ring system comprises, for example a 5- or ⁇ -memh ⁇ red heterocyclic ring having from 1 to 4, preferably from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, to which is attached an a ⁇ iated ring; in addition said fused bicycHc system is further unsubstituted or substituted by one or more subsiituente as defsned before for Q deluding the preferences given. Those rings can be saturated sing or unsaturated rings.
- Q is even more preferred the unsubstituted radical G-105, G106, Q-107, Q-108, Q-109, Q- 110 or G-111 , wherein r is 0 in each case.
- Particuiariy preferred fused bicyclic st ⁇ scfur ⁇ s Q are of formula
- a further preferred fused bicyclic structure Q is of formula
- n is an integer from 1 to 3
- Rt is CrCa-haioalkyi
- each Rg and R 3 is independently selected from the group consisting of halogen, C r C 2 -aikyl, CrCz-hsioaikyl, Ct-C 2 - alkoxy, C r -C 2 ⁇ haloaikoxy, C-rCa-haioalkyithio, -CU and -NO 2
- m is an integer from 0 to 2 2
- Q is either (i) phenyl, which is unsubstituied or substituted by 1 to 3, Sn particular 1 or 2, same or different substituents selected from the group consisting of halogen, CrCraikyl, CrC 2 - haloalkyi, C,-C 2 -aikoxy, C r C 2 -ha!oaikoxy, CrQrhaloalkyiihio, -CN, -NO 2 , and
- a 5- or ⁇ -membered heterocyclic ring having from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, which is further unsubstituted or substituted by halogen, CrCa-aikyi, Ci-C 2 -haioa!kyi, d-Cj-aikoxy, d-Cz-haloalkoxy, C 1 -C 2 - haioaikylthio, -CN, -NO 2 or Ci-C ⁇ aikoxycarbonyi, or is
- a fused bicyelic ring system comprising a 5- or 6-membered heterocyclic ring having from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, to which is attached an annulat ⁇ d ring, and which is further unsubsiituted or substituted by haiog ⁇ n, d-Craikyf, Ci-C 2 -haioaikyi, Ci ⁇ C 2 ⁇ alkoxy, Ci-C 2 -ha!oaikoxy, Ci-Ca-haioaikyithio, -CN, -NO 2 or C r C 4 ⁇ alkoxycarbonyi.
- a particuiariy prefe ⁇ e ⁇ embodiment of the invention relates to a compound of formula (Sa) above, wherein n is an integer from 1 to 3, Ri is CF 3 , each R 2 is independently selected from the group consisting of halogen, CrC a -haioaikyl, Ci-C 2 -haloalkoxy and -CH, m is 0 and Q is either
- a 5- or ⁇ -m ⁇ mb ⁇ red hetsroaromatic ring selected from the group consisting of the above- given radicals Q-S 1 Q-6, Q ⁇ 7, Q-14, CM5, Q » 16, Q-17, Q-24, Q-2 ⁇ , Q-30, Q-31 , 0-32, Q-33, Q-34, Q-43, Q-47, Q-49, Q-50, Q-52 and Q-54, wherein r is an integer from 0 to 3 and R is independently selected from the group consisting of halogen, CrQralkyi, C r C 2 ⁇ ha!oa!kyi, d-C' 2 -aikoxy, CrC 2 -haioaikoxy, Ci-Crhaloalkyithio t -CN, -NO 2 ⁇ n ⁇ CrC ⁇ alkoxycarbonyl, or is (iii) a fused bicycfic ring system of formula Q-107a, Q-107b or Q-
- n is an integer from 1 to 3
- Ri is CF 3
- each R 2 is independently selected from the group consisting of haiog ⁇ n, CrQrhaloalkyS, CrC 2 -ria!oaikoxy and -CN
- m is O
- Q is a radical Q ⁇ 5, Q-14, Q-24, G-25, Q-26, Q-30, Q-32, Q-34, Q-43, Q-44, Q-47, Q-49, Q-50 or Q-75, which is each unsubsfituted or substituted by one or more substifuents selected from the group consisting of CrC 2 ⁇ alkyi, halo- C r C 2 ⁇ aikyi, halo- C t -C 2 - alkylcarbonyl, cya ⁇ o, CrCralkoxycarbonyi, CrC 4 -alkoxycarbony1methyl,
- n is an integer from 1 to 3
- R 1 is CF 3
- each R 2 is independently halogen
- rn is 0,
- R is hydrogen or d-Cralkyi, haio- Ci-C 2 » alky1, haio- CrC ⁇ -alkyicarbonyl, cyano, CrCV alkoxycarbonyl, CrC « ⁇ alkoxycarbony!methyi, carboxy-CrCz-alkyi, aminocarbony!, M-roono- or N.N-di-CrCa-alkylsrninocarbonyl, halo- CrCa-alkylaminocarbo ⁇ ylmethyl, ha!o ⁇ C r C 2 -alkyi
- Compounds of this invention can exist as one or more stereoisomers.
- the various stereoisomers include enantlomers, diastereorners, atropisomers and geomei ⁇ c isomers.
- One skilled in the art wiii appreciate that one stereoisomer may h® more active and/or may exhibit beneficial effects when enriched relative to the other stereoisomers) or when separated from the other stereoisomer(s). Additionally, the skilled artisan knows how to separate, enrich, and/or to selectively prepare said stereoisomers.
- the compounds of the invention may be present as a mixture of stereoisomers, individual stereoisomers, or as an optically active forot.
- N-oxides can form N-oxides since the nitrogen requires an available ione pair for oxidation to the oxide; one skilled in the art wiii recognize those nitrogen containing heterocyclic rings which can form N-oxides.
- tertiary amines can form N- oxides. Synthetic methods for the preparation of N-oxides of heterocyclic rings and tertiary amines are very well known by one skilled m the & ⁇ including the oxidation of heterocyclic rings ⁇ m ⁇ tertiary amines with peroxy acids such as peracetic and m-chloroperberszoic acid (MCPBA), hydrogen peroxide, alky! hydroperoxides such as t-butyl hydroperoxide, sodium perborate, and dioxiranes such as dimethyl dioxirane
- MCPBA peroxy acids
- alky! hydroperoxides such as t-butyl hydroperoxide, sodium perborate
- dioxiranes such as dimethyl di
- salts of chemical compounds are in equilibrium with their corresponding nonsalt forms, salts share the biological utility of the nonsalt forms,
- salts of the compounds of formula (i) are useful for control of invertebrate pests (i.e. are veterinariiy or agriculturally suitable).
- the salts of the compounds of formula (I) include acid-addition salts with inorganic or organic acids such as hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric, fumarie, lactic, mai ⁇ ic, maionic, oxalic, propionic, salicylic, tartaric, 4 ⁇ toluenesuifonic or valeric acids.
- inorganic or organic acids such as hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric, fumarie, lactic, mai ⁇ ic, maionic, oxalic, propionic, salicylic, tartaric, 4 ⁇ toluenesuifonic or valeric acids.
- a compound of formula (i) contains an acidic moiety such as a carb ⁇ xyisc acid or phenol
- salts also inciude those formed with organic or inorganic bases such as pyridine, t ⁇ ethylamine or ammonia, or amides, hydrides, hydroxides or carbonates of sodium, potassium, lithium, caicium, magnesium or barium.
- the present invention comprises compounds selected from formula (I), N-oxides and veterinary acceptable and agriculturally suitable salts thereof.
- the compounds of the present invention made be prepared, for exar ⁇ pie, in analogy to the processes as outined in WO 2007/75459 on pages 29-31. Accordingly, the compounds of formula (! or (Sa) may be prepared, for example, by cycioaddition of a compound of formula
- reaction typicaily proceeds through the int ⁇ trnediacy of an in situ generated hydroxamyi chloride, in a typicai procedure a chlorinating reagent such as sodium hypochlorite, N- chiorosuccinimide, or chioraroine-T is combined with the oxime in the presence of the styrene.
- a chlorinating reagent such as sodium hypochlorite, N- chiorosuccinimide, or chioraroine-T is combined with the oxime in the presence of the styrene.
- amine bases such as pyridine or triethylami ⁇ e may be necessary.
- the reaction can be run in a wide variety of solvents including tetrahydrofuran, diethyl ether, methylene chloride, dioxane, and toluene with optimum temperatures ranging from room temperature to the reflux temperature of the solvent.
- Typical azole heterocyclic rings of formuia include optionally substituted pyrazoi ⁇ s, imidazoles, triazoles and teirazoles. Bromides can be displaced with the use of copper iodide and a palladium catalyst, see for example Kanemasa et a!., European Journal of Organic Chemistry, 2004, 685-709.
- reaction For direct fluorine displacement the reaction is typically run m a polar aprotic solvent such as N.N-dirnethyiformamide or N,N-dimethyiacetamide and in the presence of an inorganic base such as sodium or potassium carbonate.
- a polar aprotic solvent such as N.N-dirnethyiformamide or N,N-dimethyiacetamide
- an inorganic base such as sodium or potassium carbonate.
- Another process for the preparation of compounds of the formula (I) or (ia), wherein Q is a phenyl, naphthyl or heteroaramatic ring includes the Suzuki reaction via Pd ⁇ catalyzed cross coupling of an aromatic iodide or bromide of formuia (IV) above, wherein Y is Br or J 1 with an aryi or heteroary! bor ⁇ nic acid of formula
- a typical catalyst is tetrakis- (triphenylphosphine)paiiadium(O).
- Solvents such as teirahydrofuran, acetonitriie, diethyl ether and dioxa ⁇ e are suitable.
- the boro ⁇ ic acids of Formula 6 &r& either commercially avaiiabie or can be prepared by known methods. Other methods including the Heck, StiHe, K ⁇ roada and Buchwald-Hartwig coupling procedures offer many alternatives for introduction of Q heterocyclic groups. For leading references see for example Zificsak, Craig A and Hiasta, Dennis J., Tetrahedron, 2004, 60, 8991-9016.
- the compounds of of formulae (Ii) and (SV) are known, for example, from WO 2008/49459 or may be prepared in analogy to the methods disclosed therein.
- the compounds of formula (HI) may be prepared, for example, in analogy to the processes disclosed in WO 2007/75459 for the preparation of compounds of formuis (3),
- Th ⁇ compounds of the formula (i) according to the Invention are notable for their broad activity spectrum and are valuable active ingredients for use in pest control They are particularly suitable in the control of ectoparasites and to a certain extent also for controlling endoparas ⁇ tes on and in animals and in the hygiene field, whilst being weli tolerated by warm-blooded animals, in the context of the present invention, ectoparasites are understood to be in particular insects, acari (mites and ticks), and crustaceans (sea lice).
- insects of the following orders Lepidoptera, Coleoptera, Homoptera, Herniptera, Heteroptera, Diptera, Oictyoptera, Thysan ⁇ ptera, Orthopiera, Anoplura, Siphonaptera, Mallophaga, Thysanura, isoptera, Psocoptera and Hymenoptera.
- the ectoparasites which may be mentioned in particular are those which trouble humans or animais and carry pathogens, for example Hies such as Musca domestica, Musca vetustissima, Musca autumnai ⁇ s, Fannia oanicuiaris, Sarcophaga carnaria, l ⁇ cilia cuprina, LuciHa sericata, Hypoderrna bov ⁇ s, Hypoderma lineatum, Chrysomyia chloropyga, Dermatobia hominis, Cochiiomyia horninivorax, Gasterophiius intestinalis, Oestrus ovis, biting flies such as Haematobia irrilans irritans, Ha ⁇ matobia irritans exigua, Stomoxys calcitrans, horse-flies (Tabanids) with the subfamilies of Tabanidae such as Haematop ⁇ ta spp.
- Hies such as Musca domestica, Mus
- Chrysopsi ⁇ a ⁇ such as Chrysops spp.
- Chrysops ca ⁇ cutiens Hippoboscids such as Melophagus ovinus (sheep Red); tsetse flies, such as Glossinia spp,; other biting insects like midges, such as Ceratopogonidae (biting midges), Simuisidae (Biackfiies), Psychodidae (Sandflies); but also biood-sucksng insects, for example mosquitoes, such as Anopheles spp, Aedes spp and Culex spp, fleas, such as Ctenocephalides fells and Cienocephaiides canis (cat and dog fleas), Xenopsyila cheopis, P ⁇ iex irrita ⁇ s, CeratopftyOus gafSlnae, Dermatophiius penetrans, blood-sucking lice (A ⁇ opl ⁇ ra) such as Linognathus spp, Haemat
- Ectoparasites also include members of the order Acarina, such as mites (e.g. Chorioptes bovis, Cheyietieiia spp., Dermanyss ⁇ s gafifnae, Ortnithonyssus spp., Demodex cams, Sarcopt ⁇ s scabiei, Psor ⁇ ptes ovis and Psor ⁇ rgat ⁇ s spp. and ticks.
- mites e.g. Chorioptes bovis, Cheyietieiia spp., Dermanyss ⁇ s gafifnae, Ortnithonyssus spp., Demodex cams, Sarcopt ⁇ s scabiei, Psor ⁇ ptes ovis and Psor ⁇ rgat ⁇ s spp. and ticks.
- ticks are, for example, Boopfciius, Arnblyomma, Anocenior, Derroaeentor, Haemaphysalis, Hyaiomma, Ixodes, Rhipicentor, Margaropus, Rhipicephalus, Argas, Otobius and Qrnithodoros and the like, which preferably infest warmblooded animals inciuding farm animais.
- cattle such as cattle, horses, pigs, sheep and goats
- poultry such as chickens, turkeys, guineafowls and geese, fur-bearing animais such as mink, foxes, chinchillas, rabbits and the like
- companion animals such as ferrets, guinea pigs, rats, hamster, cats and dogs, but aiso humans.
- the compounds of the formula (!) according to the invention are also active against all or individual development stages of animal pests showing norma! sensitivity, as well as those showing resistance to widely used parasiticides. This is especiaily true for resistant insects and members of the order Acarina.
- the insecticldai, ovicidai and/or acaricidai effect of the active substances of the invention can manifest itself directly, i.e. killing the pests either immediately or after some time has elapsed, for example when moulting occurs, or by destroying their eggs, or indirectly, e.g. reducing the number of eggs laid ⁇ n ⁇ /ot the hatching rate, good efficacy corresponding to a pestiddaf rate (mortality) of at least 50 to 60%.
- Compounds of the formula (I) can afso be used against hygiene pests, especially of the order Diptera of the families Muscidae, Sarcophagidae, Anophiiidae and Cuticidae; the orders Orthoptera, Dictyoptera (e.g. the family Bt ⁇ ttidae (cockroaches), such as Blateiia gerrrsansca, Biaita o ⁇ entaiis, Peripianeta americana) and Hymenoptera (e.g. the families Formicida ⁇ (ants) and Vespidae (wasps).
- Dictyoptera e.g. the family Bt ⁇ ttidae (cockroaches), such as Blateiia gerrrsansca, Biaita o ⁇ entaiis, Peripianeta americana
- Hymenoptera e.g. the families Formicida ⁇ (ants) and Vespidae
- the compounds of formula (I) are also effective against ectoparasites of fishes, especially the sub-class of Copepoda (e.g. order of Siphonostomatoidae (sea lice), whiist being we!i tolerated by fish.
- Compounds of the formula (I) also have sustainable efficacy on parasitic mites ®n ⁇ insects of plants. In the case of spider mites of the order Acarina, they are effective against eggs, nymphs and adults of Tetranychidae (Tetmnychus spp. and Pmonychus spp,).
- sucking insects of the order Homopters especially against pests of the families Aphidida ⁇ , D ⁇ lpha ⁇ da ⁇ , Cicad&Uidae, Psyilidae, Loccidae, Diaspidi ⁇ ae and Eriophydidae (e.g. r ⁇ st mite on citrus fruits); the orders Hemipt&ra, Het ⁇ roptera and Thysanoptem, and on the plant-eating insects of the orders Lepidoptera, Coleoptera, Diptem and Qriboptera
- the compounds of forrouia (1) are therefore effective against aii stages of development of sucking insects and eating insects on crops such as cereals, cotton, rice, maize, soya, potatoes, vegetables, fruit, tobacco, hops, citrus, avocados and other crops.
- the compounds of formula i are also effective against piant nematodes of the species Meioidogyne, Heterodera, Pratyienchus, Dityi&nchus, Radophoius, Rizoglyphus etc.
- Helminths are commercially important because they cause serious diseases in mammals and pouitry, e.g. in sheep, pigs, goats, cattle, horses, donkeys, camels, dogs, cats, rabbits, guinea-pigs, hamsters, chicken, turkeys, guinea fowis and other farmed birds, as we ⁇ i as exotic birds.
- Typical nematodes are; Haemonchus, Trichostrcngyius, Qstertagia, Nematotiirus, Cooperia, Ascaris, Bunostonum, Gesophagostonum, Charbertia, Trichuris, Strongyius, Tr ⁇ chonema, Dictyocaulus, Capsliarsa, Heterakis, Toxocara, Ascaridia, Oxyuris, Ancyiostoma, Uncinaria, Toxascaris and Parascaris.
- the trematodes include, in particular, the family of Fasci ⁇ ikJeae, especiaify Fasciola hepatica.
- the good pesticida! activity of the compounds of formula (! according to the invention corresponds to a mortality rate of at least 50-60% of the pests mentioned, more preferably to a mortality rate over 90%, most preferably to 95-100%.
- the compounds of formula (I) are preferably employed internally and externally in unmodified form or preferably together with the adjuvants conventionally used in the art of formulation and may therefore be processed in a known manner to give, for example, liquid formulations (e.g. spot-on, pour-on, spray-on, emulsions, suspensions, solutions, emulssfiable concentrates, solution concentrates), semisolid formulations (e.g.
- preparations containing the active ingredient of formula ⁇ ! ⁇ , or combinations of these active ingredients with other active ingredients, and optionally a soiid, semi-solid or liquid adjuvant, are produced in a manner known per se, for example by intimately mixing, kneading or dispersing the active ingredients with compositions of excipients, whereby the physiological compatibility of the formulation excipients must be taken into consideration.
- the solvents in question may be: alcohols (aliphatic and aromatic), such as benzylaicohol, ethanof, propanol, isopropanol or butanol, fatty aieohois, such as oieyl alcohol and glycols and their ethers an ⁇ esters, such as glycerin, propylene glycol, dipropylene glycol ether, ethylene glycol, ethylene glycol monomethyi or -ethyl ether and butyl dloxytoi, carbonates, such as propylene carbonate, ketones, such as cydohexanone, isophorone or diacetanoS aicohoi and polyethylene glycols, such as PEG 300.
- alcohols aliphatic and aromatic
- benzylaicohol such as benzylaicohol, ethanof, propanol, isopropanol or butanol
- compositions may comprise strong polar solvents, such as N-rnethyi-2-pyrroiidone, dimethyl sulfoxide or dimefhyiforroamide, or water, fatty acid esters, such as ethyl oleate or isopropyf pa imitate, vegetable oils, such as rape, castor, coconut, or soybean oil, synthetic mono-, di-, triglycerides like e.g. glyceryl monostearafe and medium chain triglycerides and also, if appropriate, silicone oils.
- the mentioned ingredients may also serve as carrier for particulate application froms.
- ointment base r ⁇ sp. structure building ingredients the following excipients may be used: Petroleum based substances, such as Vaseline or paraffines, bases made from wool fat, like e.g. lanolin or lanolin alcohols, polyethylene glycols like e.g. macrogols and Upi ⁇ bases like e.g. phospholipids or triglyceride, such as hydrogenated vegetable oils.
- Petroleum based substances such as Vaseline or paraffines
- bases made from wool fat like e.g. lanolin or lanolin alcohols
- polyethylene glycols like e.g. macrogols
- Upi ⁇ bases like e.g. phospholipids or triglyceride, such as hydrogenated vegetable oils.
- emuisifiers, wetting agents and spreading agents may also be required, in general, lecithins like soy lecithin, salts of fatty acids with alkaline earth and alkali metals, aikyl sulfates like sodium cetylstearyl sulphate, chelates, fatty alcohols like cetyi alcohol, sterols like cholestesterol, pofyoxyeihylene sorbitan fatty acid esters like poiysorbate 20, sorbitan fatty acid esters like sorbitan mono laureate, fatty acid esters and fatty aicohoi ethers of polyoxyethyiene like poloxyl ol ⁇ yl eih&r, polyoxypropyiene pdyoxy ⁇ thytene block copolymers as e.g.
- PiuronicTM saccharose esters like saccharose distearate, poiygiyceryl fatty acid esters like poiygiyc ⁇ r ⁇ i ⁇ leate and fatty acid esters like e.g. ethyl oleate or isopropyimyristate.
- the formulations may aiso include gelifying and stiffening agents, iik ⁇ e.g. polyacryi ⁇ c acid derivatives, cellulose ethers, polyvinyl alcohols, polyvinylpyrroiidons and fine disperse siliciurn dioxide.
- gelifying and stiffening agents iik ⁇ e.g. polyacryi ⁇ c acid derivatives, cellulose ethers, polyvinyl alcohols, polyvinylpyrroiidons and fine disperse siliciurn dioxide.
- polymeric agents with controlled release properties may be applied derivatives made by e.g. pofyiactte acid, poiylactic cogiycoiic acid, poly orthoester, poiyethylene carbonate, poly anhydrids and starch and PVC based matrices,
- penetration enhancers like ketones, sulfoxides, amides, fatty acid esters and fatty alcohols may be necessary.
- Aiso preservatives like sorbic acid, benzyl alcohol and parabenes, and antioxidants as e.g. alpha tocopherol may be added.
- the active ingredient or combinations of the active ingredient may aiso applied in capsules, like hard gelatine capsules or soft capsules.
- the binders for tablets and boli may be chemically modified polymeric natural substances that are soluble in water or in alcohol, such as starch, carrotluiose or protein derivatives (e.g. methyl cellulose, carboxymethy! cellulose, ethyihydroxyethyl celiuiose, proteins such as zein, gelatin and the like), as well as synthetic polymers, such as polyvinyl alcohol, polyvinyl pyrroHdone etc.
- the tablets also contain fillers (e.g. starch, microcrystalline cellulose, sugar, lactose etc.), lubricants (e.g. magnesium stearate), giidants (e.g. colloidal silicon dioxide) and disintegrants ( ⁇ ,g. cellulose derivatives) and acid resistant coatings, like e.g. acrylic acid esters.
- fillers e.g. starch, microcrystalline cellulose, sugar, lactose etc.
- lubricants e.g. magnesium stearate
- the compounds of formula (I) according to the invention may be used alone or in combination with other biocides. They may be combined with pesticides having the same sphere of activity e.g. to increase activity, or with substances having another sphere of activity e.g. to broaden the range of activity. It can also be sensibi ⁇ to add so-called repellents.
- pesticides having the same sphere of activity e.g. to increase activity, or with substances having another sphere of activity e.g. to broaden the range of activity. It can also be sensibi ⁇ to add so-called repellents.
- repellents for example, an case of a compound of formula (!) having a particular efficacy as aduiticlde, e.g. since the compounds of formula (!) are effective in particular against the adult stage of the target parasites, the addition of pesticides which instead attack the juveniie stages of the parasites may be very advantageous. In this way, the greatest part of those parasites that produce great economic damage will be covered.
- Suitable partners in the mixture may be biocid ⁇ s, e.g. the insecticides an ⁇ acarieides with a varying mechanism of activity, which are named in the foiiowi ⁇ g and have been known to the person skilled in the art for a long feme, e.g.
- chitin synthesis inhibitors growth regulators; active ingredients which act as juvenile hormones; active ingredients which act as aduitiddes; broad-band insecticides, broad-band acaricides and nemologiedes; and also the well known antheSminthies and insect- and/or aearrci-deterring substances, said repellents or detachers.
- suitable insecticides and acaricides are mentioned in WO 2009/071500, compounds Nos. 1-284 on pages 18-21.
- suitable anthelmintics are mentioned in WO 2009/071500, compounds (A1 ) - (A31 ) on page 21.
- Non-iimitative examples of suitable repellents and detachers are mentioned in WO 2009/071500, compounds ⁇ R1 ⁇ ⁇ R3) on page 21 and 22.
- Non-limitative examples of suiiabie synergists are mentioned in WO 2009/071500, compounds (S1 ) -(S3) on page 22.
- the said partners in the mixture are best known to specialists in this field. Most ar& described in various editions of the Pesticide Manual, The British Crop Protection Council, London, and others in the various editions of The Merck Index, Merck & Co., Inc., Rahway, New Jersey, USA or in patent literature.
- a iist of suitable partners including a reference is disclosed in WO 2005/058802 on pages 16-21, No.
- a further aspect of the present invention relates to a combination preparation for the control of parasites on warm-biooded animals, characterised in that It contains, in addition to a compound of formula (!), at least one further active ingredient having the same or different sphere of activity m ⁇ at ieast one physiologically acceptable carrier.
- the present invention is not restricted to two-fold combinations.
- the insecticidai and acaricidai compositions according to the invention contain 0.1 to 99 % by weight, especially 0.1 to 95 % by weight of one or more active ingredients of formuia (i), 99.9 to 1 % by weight, especially 99.8 to 5 % by weight of a solid or liquid admixture, including 0 to 25 % by weight, especially 0.1 to 25 % by weight of a surfactant
- Application of the compositions according to the invention to ⁇ h& animals to be treated may take piace topically, peroraily, parenteral ⁇ or subcutaneo ⁇ siy, the composition being present, for example, in the form of solutions, emuisions, suspensions, (drenches), powders, tablets, boJi, capsules, collars, eartags and po ⁇ r-on formulations.
- Preferred topicaS formulations are understood to refer to a ready-to-use solution in form of a spot-on, pour-on or spray-on forrnuiation often consisting of a dispersion or suspoemulsion or a combination of active ingredient and spreading auxiliaries.
- spot-on or pour-on method is understood to refer to a ready-to-use concentrate intended to be applied topically &n ⁇ locally on the animal
- This sort of formulation is intended to be applied directly to a relatively smaii area of lhe animal, preferably on the animal's back and breech or at one or several points along the line of the back and breech.
- !t is applied as a iow volume of about 0.05 to 1 mi per kg, preferably about 0.1 mi per kg, with a total volume from 0.1 to 100 ml per animal, preferably limited to a maximum of about 50 mi.
- the total volume has to be adapted to the animal that is in ne& ⁇ of the treatment and will clearly be different, for example, in young cats and in cattle.
- These pour-on and spot-on formulations are designed to spread ai! around the animal giving protection or treatment to almost any part of the animal.
- Pour ⁇ on or spot-on formulations suitably contain carriers, which promote rapid dispersement over the skin surface or in the coat of the host animal, and are generally regarded as spreading oils.
- Suitable carriers are e.g. oily solutions; alcoholic m ⁇ isopropanoiic solutions such as solutions of 2-octyidodecanoi or oleyi aicohol; solutions in esters of monocarboxylic adds, such as isopropyi myhstate, isopropyi paSmitate, lauric acid oxalate, oleic acid oley!
- esters of dicarboxyiie acids such as dibutyi phthalate, diisopropyl isophthaiate, adipic acid diisopropyi ester, di-n-butyf adipate or aiso soiutions of esters of aliphatic acids, e.g. glycols.
- a dispersing agent may be additionally pms ⁇ nt, such as one known from the pharmaceutical or cosmetic industry.
- examples are 2-pyrroiidone, 2-(N-a!ky!)pyrro ⁇ done, acetone, polyethylene glycol and the ethers and esters thereof, propylene glycol or synthetic triglycerides.
- the oily soiutions include e.g. vegetable oils such as olive oil, groundnut oil, sesame oil, pine oil, linseed oil or castor oii, The vegetable oils may aiso be present in epoxidised form. Paraffins and silicone oils may also be used.
- a pour-on or spot-on formulation generally contains 1 to 98.9 % by weight of a compound of formula (I), 0.1 to 80 % by weight of dispersing agent an ⁇ 1 to 98.9 % by weight of solvent.
- the pour-on or spot-on method is especially advantageous for use on herd animals such as cattle, horses, sheep or pigs, in which it is difficult or time-consuming to treat all the animals orally or by injection. Because of its simplicity, this method can of course also be used for ail other animals, including individual domestic animals or pets, and is greatly favoured by the keepers of the animals, as it can often be carried out witliout the specialist presence of the veterinarian.
- compositions may also contain further additives, such as stabilisers, anti-foaming agents, viscosity regulators, binding agents or tackifiers, as weii as other active ingredients, in order to achieve special effects.
- further additives such as stabilisers, anti-foaming agents, viscosity regulators, binding agents or tackifiers, as weii as other active ingredients, in order to achieve special effects.
- the active ingredients of formula (!) can be used in ail of their sterie configurations or in mixtures thereof.
- the invention also includes a method of prophyfacticaiiy protecting animals, especially productive livestock, domestic animals s>n ⁇ pets, against parasitic helminths, which is characterised in that the active ingredients of formula (I) or the active ingredient formulations prepared therefrom are administered to the animals as an additive to the feed, or to the drinks or also in solid or liquid form, orally or by injection or parenteraliy.
- the invention also includes the compounds of formula (I) according to the invention for usage in one of the said processes
- the active ingredient is dissolved in methylene chloride, sprayed onto the carrier and the solvent subsequently concentrated by evaporation under vacuum.
- Granulates of this kind can be mixed with the animai feed, (ii) active ingredient 3 % poiyethyiene glycol (mw 200) 3 % kaolin 94 %
- the fineiy ground active Ingredient is evenly applied in a mixer to the kaolin which has been moistened with poiyethyiene glycol. In this way, dust-free coated granules Site obtained.
- Ii lactose, cryst. 22.50 % corn starch 17.00 % microcryst. cellulose 16.50 % magnesium stearate 1.00 % i Methyl cellulose is stirred into water. After the material has swollen, silicic acid is stirred in an ⁇ the mixture homogeneously suspended. The active ingredient and the corn starch are mixed. The aqueous suspension Is worked into this mixture and kneaded to a dough. The resulting mass is granulated through a 12 M sieve and dried. M Ail 4 exdpients are mixed thoroughly.
- Aqueous solubilisate (rapid release) (i) active ingredient 0,1-1 ,0 g p ⁇ iyethoxylated castor oil (40 ethylene oxide units) 10 g
- Preparation The active ingredient is dissolved in the solvents an ⁇ the surfactant, and made up with water to the desired volume. Sterile filtration through an appropriate membrane filter of 0,22 ⁇ m pore size. 4. Pour on
- active ingredient 2 g hexyi Ia urate 5 g medium-chained triglyceride 15 g ethanof ad 100 ml
- active ingredient 10-15 g octyl palrnitate 10 g isopropanol ad 100 ml
- the aqueous systems may also preferably be used for orai and/or intraluminal application.
- the compositions may also contain further additives, such as stabilisers, e.g. where appropriate epoxidised vegetable oiis ⁇ epoxidised coconut oil, rapeseed oil, or soybean oil); antifoams, e.g. silicone oil, preservatives, viscosity regulators, binders, tackifiers, as weif as fertilisers or other active ingredients to achieve special effects.
- stabilisers e.g. where appropriate epoxidised vegetable oiis ⁇ epoxidised coconut oil, rapeseed oil, or soybean oil
- antifoams e.g. silicone oil, preservatives, viscosity regulators, binders, tackifiers, as weif as fertilisers or other active ingredients to achieve special effects.
- the following examples serve to illustrate the invention. They do not limit the invention.
- the letter 'h' stands for hour.
- the starting materials am known and partially commercially available or may be produced in analogy to methods known per se.
- Analysis of the purified samples is in each case done using a Waters Autopurificatio ⁇ (HPLC/MS) system with a reversed phase column (Daisogel SP-120-ODS-AP 5 ⁇ m, 150X3mm) from Bischoff, L ⁇ onb ⁇ rg, Germany.
- the samples are characterized by m/s and retention time.
- Steps ⁇ i SnCU 13 mi, 1M solution In dichiorcmethane
- a solution of 1-fiuoro-naphtatene (1.46 g) in dtohioromethane (4 ml) is added over 10 minutes.
- the reaction is stirred overnight at room temperature and then quenched with c ⁇ ict water (30 ml).
- the organic phase is separated an ⁇ the aqueous phase is extracted 3 times with dichior ⁇ m ⁇ thane.
- StejLCj, 1,2,4-triazoie (248 rog) is a ⁇ to a solution of 4-fluoro ⁇ naphthalene-1- carbaldehyde (522 mg) and potassium carbonate (49? mg ⁇ in DMF (4 ml), After 3 hours at room temperature, the reaction is quenched with water ⁇ 40 ml) and ethyi acetate (50 mi). The organic phase is separated and the aqueous phase is extracted two times with ethyi acetate. The organic phases are combined, extracted with water and with a saturated aqueous solution of NaCI, dried over Na 2 SO 4 and concentrated in vacuo.
- Step D Sodium acetate ⁇ 282 mg) is added to a solution of hydr ⁇ xyiamtne hydrochloride (193 mg) an ⁇ 4 ⁇ (1 > 2,4]tdazoi ⁇ 1-yi ⁇ naphtha!ene-1-carbaidehyd ⁇ (4S5 mg) in THF (18 mi), water (2 ml) an ⁇ DiVISO (2 ml). After 5 hours at room temperature, the reaction is quenched with water and ethyi acetate. The organic phase is separated and the water phase is extracted with ethyi acetate.
- StepjE t ⁇ -chi ⁇ r ⁇ succinirred (220 mg ⁇ is added to a solution of 4 ⁇ [1 ,2,43tria2 ⁇ l ⁇ 1 ⁇ yi ⁇ naphtha!ene-1-carbaldehyde ⁇ xfrne (390 mg) in DMF (5 mi). After 45 minutes at 40 0 C, the reaction is cold down to 0 0 C an ⁇ 1 ,3 ⁇ dichioro ⁇ 5 ⁇ (14rifluoromethyl-vinyi)-benzene (394 mg, Example 1, step A) and t ⁇ etriylarnine (0.25 ml) are added. The reaction is then further reacted at room temperature for 3 hours.
- Step A A solution of benzoyl peroxide ⁇ 87 mg) in trifiuorotoluene (40 mi) is refiuxed for 30 min under nitrogen.
- the reaction mixture is cooled to 78 0 C and 1-bromo-4-methyl- naphthaiene (1.6 mi) an ⁇ W-bromosuccinimide (2.0 g) are added.
- the reaction is stirred for 1.5 hours at 78 0 C and filtered.
- the filtrate is concentrated in vacuo and the residue crystallized in heptane to yield 1 ⁇ brom ⁇ -4-bromometnyi-naphfchaien8.
- the crude product is used in the next step without further purification.
- Step B Sodium bicarbonate ⁇ 1.4 g) is added to a solution of crude 1-bromo-4 ⁇ bromomethyl ⁇ naphthalene (2.5 g) in DMSO (25 rn!). The reaction is stirred for 3h at 95 "C and cooled down to room temperature. Water is added and the mixture is extracted with ethyi acetate. The organic phase is dried over MgSO 4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield 4 ⁇ bromo ⁇ naphtha!ene-1-carbafdehyd ⁇ as colorless crystals.
- Step C Hydroxyiamin ⁇ (50% aqueous, 0.37 ml) is added to a solution of 4-bromo- naphthalene-1-earbaidehyde (0,7 g) in methanol (8 ml). After 1 ⁇ hours at room temperature the reaction is concentrated in vacuo to yield crude 4-bromo- ⁇ aphthalen ⁇ -i-carbaidehyde oxime, which Is used in the next step without further purification.
- Step D W-chiorosucclnirnid (134 mg) is added to a solution of crude 4 ⁇ bromo ⁇ naphthalene-1- carbaidehyde oxime (250 rog) in DMF (3 mi). After 1 hour at 40 0 C, the reaction is cooled down to 0 "C and 1,3-dichlor ⁇ -5- ⁇ 1-trifiuoromethy!-vinyl ⁇ -b ⁇ nzene (240 mg) and trieihyiamine (0.15 m! are added. The reaction is further reacted at room temperature for 16 hours. Water is added and the reaction mixture is extracted with ethyl acetate.
- the organic phase is washed with a saturated aqueous solution of NaCt, dried over MgSO 4 and concentrated in vacuo.
- the catde product is purified on a semi-preparative HPLC to yield 3 ⁇ (4-br ⁇ m ⁇ " naphthalen-i-yi ⁇ -CS.S-dichloro-phenyO-S-trifl ⁇ oromethyl ⁇ . ⁇ -dihydro-isoxazole as coiorless crystals: m.p. 127-130 0 C.
- Step F Trifiuoroacetic acid anhydride (0.18 mi) is added to a soiution of_4- ⁇ 4-[5-(3,5- dfChioro-phenyO-S-trifluoromethyi ⁇ .S-dshydro-isoxazol-S-y ⁇ naphthalen'i-yl ⁇ pyrazoie-i- carboxyiie acid t&rt ⁇ buty ⁇ ester (135 mg) in didiloromethane (1 ml). After 1 hour at room temperature an aqueous soiution of sodium hydroxide (30%) is added.
- the aqueous phase is extracted with d ⁇ chiorornethane and the combined organic phases are dried over MgSQ* and concentrated in vacuo.
- the crude product is purified on a semi-preparative HPLC to yield 5-(3,5»dichioro ⁇ heny1)-3-f4-(1H-pyra2 ⁇ i-4-yi)-naphthalen-1-yi]-5-trifl ⁇ oromethyi-4,5- dihydro-ssoxazoie as a coloriess solid: m.p. 117-120 0 C (compound 1.17, table 1 ).
- Step 8 A soiution of t ⁇ trabutyiamr ⁇ onium chloride (1 M in THF, 2.25 ml) is siowly added to a solution of crude 5"(3,5-dichloro-phenyi>5'triffuoromethy!-3-(4-t ⁇ methyisiiany!8thy ⁇ yi- naphtbaien-1-yi) ⁇ 4.5 ⁇ dihydro-isoxazole (step A) in THF (25ml) at ⁇ °C. After 3 hours at 5'C a saturated soiution of ammonium ehioride and ethyl acetate are added.
- Step A Sodium acetate (4.8 g) is added to a soiution of hydroxyiamine hydrochloride (2.9 g) and 4-ftuoro ⁇ naphthaiene ⁇ 1 ⁇ carbaicfehyde (6,1 g, example 1 step B) In THF (40 ml) and water (10 mi). After 3 hours at room temperature, the reaction is quenched with water and ethy! acetate. The organic phase Is separated and the water phase is extracted with ethy! acetate.
- Step C Sodium azide (3.3 g) is a ⁇ de ⁇ to a soiution ot ⁇ -(3,5 ⁇ dichioro ⁇ phenyi)-3-(4-fiuoro ⁇ ⁇ aphihaie ⁇ -1"yi ⁇ -5-trifiuorornethyi » 4,5-d3hydro-isoxa2 ⁇ le (10.7 g) in OUF (50 ml). After 20 hours at 80 ⁇ C another portion of sodium azide is added (3.3 g) and the reaction mixture is stirred for 20 hours at 8O 0 C. The soiution is cooi ⁇ d to room temperature and the volume concentrated to 10 mi in vacuo.
- the crude product is purified on a semi-preparative HPLC to yield A/-(1 ⁇ 4 ⁇ [5-(3,5-dsch!oro «phenyi) ⁇ 5 ⁇ trifluoromethyi-4,5-dfhydro-isoxazo!-3-yl]-naphthalen-1 ⁇ y! ⁇ -1 W-[1 ,2,3
- Tabie 1 provides further compounds of formula wherein the meaning of Q is given Tabie 1.
- the substances are prepared analogously to the above-described methods.
- the following physical data are obtained according to the above- described HPLC/MS characterization process.
- the values of the meiting point are indicated in "C. Table 1:
- a mixed adult population of fleas is placed in a suitably formatted 96-well plate allowing fleas to access and feed on treated biood via an artificial feeding system.
- Fleas are fed on treated blood for 24 hours, after which the compound's effect is recorded, insecticidal acliviiy is determined on the basis of the number of dead ffeas recovered from the feeding system- Compound 1 ,1, 1.2, 1.5.
- a clean aduit teck population is used to seed a suitably formatted 9 ⁇ weii plate containing the test substances to be evaluated for antiparasitic activity.
- Each compound is tested by serial ⁇ llutlon in order to determine its minimal affective dose (MED).
- MED minimal affective dose
- Ticks ate left in contact with the test compound for 10 minutes and are then incubated at 28"C and 80% relative humidity for 7 days, during which the test compound's effect is monitored.
- Acaricidai activity is confirmed if adult ticks are dead.
- gerbiSs are treated with the test compound at a given dose by spray application.
- the animals & ⁇ B infested with nymphs of R sanguineus. Ticks are left on the animate until f ⁇ ii repletion. Seven days after infestation nymphs dropped off fully engorged are collected and counted. Efficacy in killing is expressed as a tick number reduction in comparison with a placebo treated group, using the Abbot's formula.
Abstract
The invention relates to new pyrimidine compounds of formula (I) wherein the variables have the meaning as indicated in the claims; in free form and in salt form; and optionally the enantiomers and geometrical isomers thereof. The compounds of formula (I) are useful in the control of parasites, in particular ectoparasites, in and on warmblooded animals.
Description
ISOXAZOLINES DERIVATIVES AND THEIR USE AS PESTICIDE
FIELD OF THE INVENTION
This invention relates to novei isoxazoltnes, their N-oxsdes and salts, processes for their manufacture, their use in the controi of ectoparasites, especially insects and acari. on non- hurπan animais, especiaily productive livestock and domestic animals, and furthermore pesticϊdai compositions which contain one or more of these compounds.
BACKGROUND OF THE INVENTION
PCT Patent Publication WO 2007/079162 discloses isoxazoiine derivatives of Formula (A)
wherein, inter alia, each of A1-A8 and 8,-B3 are C(R3), Ri is haioaikyl and W is O or S. The compounds are mainly used in the control of invertebrate pests in agronomic environments. Many products are commercially available for these purposes, but the need continues for new compounds that are more effective, less costly, less toxic, environmentally safer or have different modes of action. It now has been surprisingly found that novel naphthyl derivatives with a modified heterocyclic side chain have superior properties in the control of pests.
SUMMARY OF THE INVENTION
This present invention is directed to a compound of formula
(0
including ail geometric and stereoisomers, N~oxides, and salts thereof, anύ compositions containing them and their use for controlling parasites, wherein
Ai, A2, A3, A^5 A5 and A6 are each independently selected from the group consisting of CR3' and N; n is an integer from 0 to 4;
Bi, 82 snύ 83 are each independently selected from the group consisting of CfV and N; each R2' is independently of the other H or R2; each R3' is independently of the other H or R3;
Ri is C-rCVaikyi, C2-C6-alkenyi, C2-C6-aikynyJ, CVCs-cycloaikyi, C4-Craikylcycioa!ky1 or C4-
C7-cyc!oaikylalky!, each unsubstituted or substituted with one or more sυbstituents independently seiected from R4; each R2 is independently halogen, CrCe~aiky!, CrCδ~hafoaiky!, d-Cs-aikoxy, C1-C6- haioaikoxy, CrC8~aiky!thio, CrC8~haioalkyithio, CrC6-aikyisu!firtyS, CrC6-haioaiky1suifiny!,
Ci-Cβ-alkylsυifonyi, Ci«C8-haioaikyisuifonyi. N-mono- or N.N-di-d-Ce-aikyiamino, C3-C8 aikoxycarbonyl, cyano (-CN) or nitro (-NO2); each R3 is independently halogen, CrC6-aikyi, CrC6-haloaSkyi, C,rCβ-cydoa!kyl, CrQ- halocycioaikyl, CrCg-aikoxy, CrCs-haioaikoxy, Ci-C6-alkytth!θ, CrCδ-haloalky!thio, CrCe alkyl-sυifinyi, CrCβ~rfaioalkyisu!finyi, CrCe-a!ky[su!fonyi( CrCβ-haloaikyisυlfonyl, amino.
N-rπono- or N.N-di-Ci-Cβ-alkyiarπino, Ci-Cβ-alkoxycarbonyi, -CN or -NO2;
R4 is haiogβfi, CrCtraikyi, CrC6-aikoxy, d-Cβ-aikyithio, CrC6-a!ky!sυlfιnyi, CrC6~aikyi- suSfonyi, -CN or -NO2;
Q is a 5- or 6~membered heterocyclic ring, or a QrCio~carbocyciic ring system or a 8», 9- or
1G~membered fuse<i hetero-bicydic ring system, each of them being unsubstituted or substituted with one or more substitυents independently selected from halogen, CrCδ-alkyi,
CrQrhafoalkyi, C3-C6-CyClOaSkVl1 C3-C8-halocycloalkyl, CrQ-aikoxy, C3-C6-cycioaikoxy( C1-
Cδ-haioalkoxy, CrCδ-alkenyl, Ca-Cs-haioalkenyi, CrC6-alkyithio, CrCa-liaioaikyithio, CrCe- aikyisυlflnyi, CrCe-haloaikyisυlfinyl, d-Craikyisulfonyl, CrCβ-hatoaSkylsuifonyi, -CN, -NO2. amino, N-mono- or N.N-di-CrCg-alkylarnino, CO2H, CrC6~a!koxycarbonyi, suifonamido, N- mono* or N1N, di-CrCe-alkylsulfonamido, CONH2, N-mono- or H,H, di-Ci-C6-a!kyfamino- carbonyl, N-mono- or N,N, di-CrCs-haloalkylaminocarbonyl, N*CrC2-ha!oaikyiamino- carbonyl-Ci-C2-alkyfamiπocarbonyi, CrCe-aikyicafbonyiamino, Cz-Ce-aikanoy!, C2"Cβ-halo« alkanoyi, CH2R5 , a group C(O)N=CHN(CH3}2, a group C(O)N^CHNHOCH3, pyrroltdonyi- carbonyi, and unsubstituted or habgen-, CrCg-aikyt-, CrCδ-haloaikyi-, Ci-Cδ-alkoxy~, d-Cδ- haloalkoxy-, nitro- or cyano-substituied phenyl, benzyl, phenethyl, pyridyi, pyridinyimethyl, pyrimidinyl, pyrimidiπyimethyS, benzoyi or phenoxy; and
R8 is CO2H, CrCe-aikoxycarbonyl, CONH2, N-mαnø- or H,H, di-CrCg-alkylaminocarbonyl, N-monO" or N,N, di-C-rCβ-haioalkyiaminocarbonyi, CrCs-alkylearbonylamino, pyridinyfaminocarfoonyi or tetrahydrofuraπyi.
According to a preferred embodiment there is provided a compound of formula (I) above, wherein Ai, A2, A3, Aj, A5 and A8 are each independently selected from the group consisting of CR3' and N; n is an integer from 0 to 4;
B1, S2 and B3 are each independently selected from the group consisting of CR2' and N; each R2' is independently of the other H or R2; each R3' is independently of the other H or R3;
Rt is CrCβ-aikyi, C2~C6~alkenyi, C2-CVa!kyny!, QrQrCyctøaiky!, C^CrSikylcycioalkyl or C4-
Cr-cvcioalkyialkyt, each υnsubstituted or substituted with one or more substituents independently selected from RA; each R2 is independently halogen, Ci-C«-alkyl, CrCδ-haioalkyl, Ci-C6-alkoxy, CrC6- haloaikoxy, CrC6-aiky!thio, Ci-C6-ha!oaikyithio, CrCg-aikylsuifinyl, CrCVhaioalkyisuifinyi,
CrCs-alkylsuifony!, CrCδ-haioaikylsu!fonyl, N-mono- or NtN-di-CrCe-a!kyiam!no, Ca-C6 aikoxycarbonyi, cyano (-CN) or nitro (-NO2); each R3 Is independently halogen, CyCe-alkyi, CrCfi-haloaikyl, Ca-Cg-cycioalkyl, CrCfi~ haiccycloalkyi, CrCβ-aikoxy, CrCe-haloalkoxy, CrC6-a!kylthio, CrCs-haloaikySthio, CrC6 aikyl-suSfinyl, Ci-C6-haioaikyisu!finy!( CrC6-a!kylsuifony1, CrCδ»hatoa!kylsuifony), amino,
N-mono- or N.N-di-CrCe-aikyiarnino, Ca-Cβ-aSkoxycarbonyi, -CN or -NO2;
R4 is halogen, Ci-Cβ-alkyi« CrCδ-aikoxy, Ci-C6~a!ky!thio, Ci-Cδ-aikylsulfiπyi, Ci-C6-a!kyi- suifonyl, -CN or -NO2; and
Q is a 5- or δ-membered heterocyclic ring, or a Cg-Cio-carbocyclic ring system or a 8-, 9- or
10-membered fused heterø-bicyctic ring system, each of them being unsubstitυted or substituted with one or more substituents iπdependentiy selected from halogen, Ci-C6-a!kyi,
C-Ce-haioalkyl, C3-Cδ-cycioa)kyl, C3-C6-halocycSoatkyi, d-Cβ-alkoxy, CrC6~haloalkoxy, C1-
Cβ-aikyithio, d-Cβ-fraioalkyithio, Ct-Cβ-alkyisuifinyl, CrCe-haioalkylsulfinyl, Ci-C6-a!kyl- sυlfonyi, CrCc-haloaikylsulfonyl, -CN1 -NO2, amino, N-mono- or N.N-di-CrCe-alkylamino, C2*
Ce-alkoxycarbonyl, suifonamido, N-mono- or N1N, di-CrCδ-alkyfsulfonamido, N-mono- or
N.N-di-CrCfe-aikyiearbonylamirjo, C2-Ce-alksnoyi and unsubstituted or halogen-, CrC6-alkyl-,
CrCs-haloaikyi-, d-Ce-aikoxy-, Cr€6-ftaloaikoxy-, nitro- or cyano-substituted phenyl, benzyl, benzoyl or phenoxy.
TNs invention aiso provides a composition comprising a compound of formula (S)1 an N-oxide or a sait thereof, and at least one additional component selected from the group consisting of a surfactant, a solid diluent &nό a liquid diluent.
In one embodiment, this invention also provides a composition for controiling parasites, in particular ectoparasites, comprising a biologlcaHy effective amount of a compound of formula (!), an N-oxsde or a sait thereof, and at ieast one additional component selected from the group consisting of a surfactant, a soiid diluent and a liquid diluent, said composition optionally further comprising a biologically effective amount of at ieast one additional biologically active compound or agent.
This invention further provides the composition described above in the form of a bait composition wherein the solid diiuent and/or the liquid diluent comprises one or more food materials, said composition optionally comprising an attractant and/or a bumectant
This invention further provides a trap device for controlling parasites, in particular ectoparasites, comprising said bait composition and a housing adapted to receive said bait composition, wherein the housing has at ieast one opening sized to permit the parasites to pass through the opening, so the invertebrate pest can gain access to said bait composition from a location outside the housing, and wherein the housing is further adapted to be placed in or near a iocus of potential or known activity for the parasites pest.
This invention also provides a method for controiling parasites comprising contacting the parasites or their environment with a biologically effective amount of a compound of formula (1), an N-oxkJe or a sait thereof, (e.g., as a composition described herein). This invention also relates to such method wherein the parasites or their environment are contacted with a composition comprising a biologically effective amount of a compound of formula 0). an N- oxide or a sait thereof, and at least one additional component selected from the group consisting of a surfactant, a solid diluent and a liquid diluent, said composition optionally further comprising a biologically effective amount of at ieast one additional biologically active compound or agent.
This invention also provides a composition for protecting an animal from an parasitic pest comprising a parasiticidaiiy effective amount of a compound of formula (I) Bn N-oxide or a
salt thereof, and at least one carrier. The present invention further provides the composition described above in a form for orai administration. This invention also provides a method for protecting an animal from a parasitic pest comprising administering to the animsf a parasiticus Hy effective amount of a compound of formula (i), an N~oxide or a sait thereof.
DETAILS OF THE INVENTION in the above recitations, the term "aikyS", used either alone or in compound words such as "aikyithio" or "haSoafkyP includes straight-chain or branched aiky.1, such as, methyl, ethyl, n~ propyl, i-proρyi, or the different butyl, penty! or hexyi isomers.
"Alkeπyi" includes straight-chain or branched aikenes such as ethenyl, 1~propenyl, 2- propenyi, and the different butenyi, pentenyl and hexenyl isomers. "ASkenyi" also includes polyenes such as 1 ,2-ρropadienyi and 2,4-hexadienyl.
"Aikynyf includes straight-chain or branched alkynes such as ethynyS, 1-propynyl, 2-propynyi and the different bυtynyS, pentynyi and hexynyi isomers. "Alkynyi" can also include moieties comprised of multiple triple bonds such as 2,5-hexadiynyi.
"Alkoxy" includes, for example, methoxy, ethoxy, n-propytoxy, isopropyioxy and the different butoxy, perttoxy and hexyloxy isomers. "Aikyithio" includes branched or straight-chain aikyithio moieties such as methylthio, ethylthio, and the different propyithio, butyithio, pentyithio and hexyithio isomers..
"Aikyisulfinyf Includes both enantiomers of an aikylsυifinyi group. Examples of "alkylsuifinyl" include CH3S(O)-, CH3CH2S(O)-, CH3CH2CH2S(O)-, {CH3)2CHS(O}- Bnύ the different butylsuifinyi pentylsuifinyi and hexylsulfiny! isomers.
Examples of "alkylsulfonyl" include CH3S(O)2-, CH3CH2S{O)r, CH3CH2CH2S(O)2-, (CH3)JCHS(O)2-, and the different butylsυifonyl, pertiylsuffony! and hexylsulfonyi isomers.
"N-alkyiamino", "N.N-di-aikyamino", and the like, are defined analogously to the above examples.
"Cyeioalkyi" incSυdes, for example, cyciopropyl, eyclobutyi, cyciopentyi and cyclohexyi The term "aifcyicydoalkyi" denotes alkyl substitution on a eyciøaikyi moiety and includes, for example, ethyteyciopropyl, i-propylcydobutyi, 3-methyicyεiopentyl and 4~mβthy1cyclαhexyi, The term "cycloaikylaikyi" denotes cycioaikyi substitution on an alkyl moiety. Examples of "cycløaikytaikyf include cyciopropyimethyi, cyetopentyiethyl, and other cycioaikyi moieties bonded to straight-chain or branched alkyi groups.
The term "halogen", either atone or in compound words such as "haioalkyi", includes fluorine, chlorine, bromine or iodine. Further, when used in compound words such as "baioalkyf, said aikyi may be partially or fully substituted with halogen atoms which may be the same or different. Examples of "haioaikyP inciude F3C-, ClCH2-, CF3CH2- and CF3CCI2-. The terms "hafocyeloaikyf, "haloafkoxy", "haløaikyithio", and the like, &t® defined analogously to the term "haioaikyi". Examples of "haioaikoxy" include CFjO-, CCI3CH2O-, HCFsCH2CH2O- and CF3CH2O-. Examples of "hatoalkytthio* inciude CCl3S-, CF3S-, CCl3CH2S- and CICH2CH2CH2S-. Exampies of "haloaikyisulfinyl" include CF3S(O)-, CCi3S(O)-, CF3CH2S(O)- and CF3CF2S(O)-. Examples of "hatoalkyteuHtonyT include CF3S(O)2-, CCi3S(O)2-, CF3CH2S(O)2- and CF3CF2S(O)2-.
"Aikyicarbonyl" denotes a straight-chain or branched alky! moteties bonded to a C(-0) moiety. Examples of "alkyicarbonyi" include CH3C(=0)-, CH3CH2CH2CC=O)- and (CH3)2CHC(=:O)~. Examples of "alkoxycarbonyl" inciude CH3OC(^O)-, CH3CH20C{~0), CH3CH2CH2OC(~O)~, (CH3^CH0C(~0)- and the different butoxy- or pentoxycarbonyi isomers, for example tert.-butoxycarboπyi (Boc).
The total number of carbon atoms in a substituent group is indicated by the "C1-Cf prefix where i and j are integers. For example, CrC4 alkylsulfonyi designates methyisuifonyl through butylsuifoπyi; C2-alkoxyalkyl designates CH3OCH2; C3~alkoxyaikyl designates, for example, CH3CH(OCH3), CH3OCH2CH2 or CH3CH2OCH2; and C4-alkoxyaikyi designates the various isomers of an aikyl group substituted with o,n aikoxy group containing a total of four carbon atoms, examples including CH3CH2CH2OCH2 and CH3CH2OCH2CHr-
When a compound is substituted with a substituent bearing a subscript thai indicates the number of said substituβnts can exceed 1 , said substituenfs (when they exceed 1 ) are independently selected from the group of defined substituents, e.g., (R2)m n is 1 or 2.
" Aromatic" indicates that each of the ring atoms is essentially in the same plane and has a p~ orbital perpendicular to the ring plane, and in which (4n + 2) n electrons, where n is a positive integer, are associated with the ring Io comply with Muckers rule.
The terms "heterocyclic ring" or "heterocycfe" denote a ring in which at ieast one atom forming the ring backbone is not carbon, e.g., nitrogen, oxygen or sulfur. Typically a heterocyclic ring contains no more than 4 nitrogens, no more than 2 oxygens and no more than 2 sulfurs. Unless otherwise indicated, a heterocyclic ring can be a saturated, partially unsaturated, or fully unsaturated ring. When a fully unsaturated heterocyclic ring satisfies Hϋckei's rule, then said ring is also called a "heteroarorπatic ring", "aromatic heterocyclic ring". Unless otherwise indicated, heterocyclic rings and ring systems can be attached through any available carbon or nitrogen by replacement of a hydrogen on said carbon or nitrogen.
When Q is a 5- or 6~membered nitrogen-containing heterocyclic ring, it may be attached to the remainder of formula (I) through any available carbon or nitrogen ring atom, unless otherwise described.
Each R2 is independently of the other preferably halogen, CrCδ~haioalkyl, Ci-Ce haloalkoxy or -CN, more preferably halogen, CF3, GCF3 or -CN, and in particular halogen.
B1, S2 and B3 are each independently of the other preferred the group CR2', wherein R2' is H or R2, and for R2 the above-given meanings and preferences apply. R2' is most preferably H or halogen.
The variable n is meant to summarize all radicals R2 in the δ-mernbered ring, n is preferably am integer from 0 to 4, more preferably from 1 to 3, and in particular 2 or 3.
R1 is preferably CrC6-a!kyl optionally substituted with one or more sυbstituents independently selected from R4, more preferably CrC3-alky! optionally substituted with halogen, even more preferably CrC3-HaIOaIKyI, especially preferably CrCa-aiky! substituted with F, and in particular CF3.
Each R3 is independently of the other preferably halogen, CrC4-ølkyl, CrCt-halαaikyi, C3-CV cycioaikyi, CrC^-alkoxy, CrC4-haioaikoxy, N-mono- or N,N-di-CrC2-a!kyiamino, -CN or -NO2, more preferably halogen, d-Qralkyj, CrC2-hafoa!kyi, cyclopropyS, Ci-C2-aikoxy, -CN or -NO2, and even more preferably halogen, C1-C2 a!ky!( CrC2 afkoxy.-CN or -NO2.
A1, A2, A3, A4, A5 and Ag are each independently of the other preferably a group CR3'. R3' is preferably H or R3, wherein for R3 the above-given meanings and preferences apply. Preferably, three of the radicals AL A2, A3, A4, A5 anά A6 are each CH and the other three radicals are either CH or CR3. Most preferably, AL A5, A3, A4, A5 and A5 are each CH.
R4 is preferably halogen, CrC2-aiky1, C,-C2~atkoxy, -CN or -NO2, more preferably halogen, - CN or -NO2, anύ in particular halogen.
According to one preferred embodiment of the invention, Q is a C6~Ci0-carbocyclic ring system, for example phenyl, πaphthyi, iβtrahydronaphthyi, iπdanyf, indenyl, hydrindanyi or octahydro-pentaien, in particular phenyl, which is each υnsubstituted or substituted by one or more same or different substsiuents selected from the group of substituents as defined before for Q. Q is preferably phenyl which is substituted by 1 to 4, preferably 1 to 3 and in particular 1 or 2 same or different substituents selected from the group consisting of halogen, CrC4-alkyS, Cr&rhaioaikyl, CrC^alkoxy, CrC4-haioalkoxy, CrC.raikyithio, C1-C4- haloaikylthio, CrC4-alkylsuHwyi, CrCrhaløafkyisulfinyi, Ci-C4-alkyisuifonyl, Ci-C4-ha!oa!kyi~ sulfoπyl, -CN, -NO2, CrCά-alkoxycarbonyi, sulfonamide, CrC3-aikanoyl and unsubstitufed or halogen-, CrCd-a!kyi.., CrC4-haloa!kyk CrC4-a!koxy-, C1-C4-HaIOaIkOXy-, nitro- or cyano- substituted phenyl, benzyi, benzoyl and phenoxy. Q is more preferably phenyl, which is substituted by 1 to 3, in particular 1 or 2, same or dtfferent substituents selected from the group consisting of halogen, CrQraikyt, CrCrhaloalkyf, CrC2-alkoxy, Ci-C2-haioalkoxy, Cr C2«haloalkylthio, -CN, -NO2, and unsubstituted or halogen-, Ci-C2-alkyl-, d-Ca-haloalkyl-, Ci- Cr-altoxy-, d-C^-haioaikoxy', nitro- or cyano-subsiUυted pheny! or phenoxy.
According to another preferred embodiment of the invention, Q is a 5- or 6~membered heterocyclic ring, which may be saturated or preferably unsaturated, &nά which is unsubstituted or substituted with one or more substituents selected from the group of substituents as defined before for Q.
Preførrβd substituents of the heterocyclic ring Q are, for example, CrC^alkyt, CrCXrhaio* aikyi, CrC4-aikoxy, Ci-C^haioaikoxy, CrC^alkyiihio, CrC-haloalkylthio, d-Co-alkylsuifinyl, CrC4~hak>a!ky1sulfinyit GrC4-aiky!suJfonyi GrCrhaiøalkylsuifonyl, -CN, -NO2, CrC4~aikoxy- carbony!, N-moπo- or N,N-di» CrCValkylcarbonyiamino, sulfonamide, CrC3~aikanoy! and unsubstituted or halogen- or Ci-C4~alkyl'Substitυt8d phenyl, benzyl, benzoyl and pnenoxy. Evert more preferred substituents of the heterocyclic ring Q are selected from the group consisting of halogen, C,~C2-alkyf, Ci-C2-ha!oaikyl, CrC2~alkoxy, CrC2-haloalkoxy, C1-C2- naloalkylthio, -CN, -NO2, and CrCs-alkoxycarbonyi, in particular CrCraikyi, CrC2-haioalkyi and d-C^alkoxycarboπyl,
A suitable heterocyeiic ring is, for example, a 5~ or 6-membered heteroaromatic ring having from 1 to 4, preferably from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, which is further unsubstituted or substituted by one or more substituents as defined before for Q including the preferences given therefore. The heterocyclic radical Q is preferably substituted by 0 to 3, in particular 0, 1 or 2 substiluents from the group as defined before for O.
Examples of a 5- or 6-membered unsaturated aromatic heterocyclic ring optionally substituted with from one or more substituents include the rings Q-1 through Q-80 illustrated in Exhibit 1 wherein R is any suøstituent as defined before for Q including the preferences given, and r is an integer from 0 to 4, limited by the number of available positions on each Q group. As Q«28,~ Q-29, Q-35, Q-36, Q-37, Q-3B, Q~3δ, Q-40, Q-41 and Q-42 have only one available position, for these Q groups r is limited to the integers 0 or 1 and r being 0 means that the Q group is unsubstituted and a hydrogen is present at the position indicated by (R),,
Exhibit 1
A further group of suitable heterocyclic radicals comprises, for example, a 5- or 8-membered heterαaiiphatϊc or partly unsaturated ring having from 1 to 4, preferably from 1 to 3 same or different hβtβroatoms selected from the group consisting of N1 O and S. which is further uπsubstitutβd or substituted by one or mom substituents as defined before for Q including the preferences given therefore.
Examples of heteroaiiphatio or partly unsaturated rings include the radicals illustrated in Exhibit 2 below, wherein R and r are as defined above including the preferences given.
Exhibit 2
preferred heterocyclic raclicaS Q is of formula
whereiπ r is an integer from 0 to 3 and R is independently selected from the group given for Q including the preferences. Q is particularly preferred the unsubstituted radical G-34, G-43 or Q-47, wherein r is 0 in each case.
A further particularly preferred radical Q is a radical G-44, wherein r is an integer from 0 to 3, in particular 1 , and R is independency selected from the group given for Q including the preferences. Another preferred radical Q is a radical G-75, wherein r is an integer from 0 to 3, in particular 1 , and R is independently selected from the group given for Q including the
A further preferred radical G is a radical G-5, Q-14, Q-24, G-25, Q-26, G-3G, Q-32, Q-34, Q- 43, Q-44, Q-47, Q-49, Q-50 or Q~75, in particular a radical Q-24, Q-26, Q-30, Q-32, Q-34, Q-44 or Q-47, which is each unsubslituted or substituted by one or more substituents selected from the group consisting of CrC2~aikyi, halo- CrG2-SlKyS, halo- C1-C2- alkyicarbonyl, cyano, CrCt-aikoxycarbonyl^ CrC^-alkoxycarbonylmethyi, carboxy-Ct-Cr alkyi, aminocarbonyl, N-mono- or N^-di-Ci-Ca-afkytarfiinocarbonyi, halo- Ci-C2- alkyiaminocarbonyimethyl, halo-C^Ca-alkylaminocarbonylmethyiaminocarbonyl, haiovinyl, - C(G}N~CHN(CH3}2, -C(O)N=CHNHOCH3, unsubstituted or halogen or CrC2-aikoxy- substitutβd phenyl, benzyl and phenyiβthyl, unsubstiiuted and haiogen-substituted 2~, 3- mά 4-pyridyi and pyridylmethyi, pyridylcarbonylaminomethyi, pyrroiidinøcarbonyl, and 2-
A further particularly preferred radical G is a radicai G-5, Q-14, Q-24, Q-25, Q-26, Q-30, Q- 32, Q-34, Q-43, Q-44, Q-47, Q-49 Q-50 or Q-75, in particular a radical G-24, G~2δ, Q-30, Q- 32, Q-34, Q-44 or Q-47, which is each unsubstituted or substituted by one substituβnt selected from the group consisting of methyl, trifluoromethyi 2~trifiuoroethyf, trifiuormethyicarbonyi, cyano, methoxycarbonyl, tert.-buloxycarbonyi, carboxymelhyl, ethoxy- carbonyimethyl, aminocarbonyl, N-metliytaminocarbony!, 2~trif1uorethylaminocarbonyl- methyl, 24rifiuorethylaminocarbonyimethyiaminocarvbonyi,1-fiuoro-2-iodo-1-vinyf( - C{O}N~CHN(CH3)2, C(O}N~CHNHOCH3, 4~chlorphenyi, 4-ffuorophenyi, 4-methoxyphβnyl, benzyi, 4-chiorophenylethyi, 2-, 3~ and 4-ρyridyi, chloropyridyi, 2~, 3- and 4-pyridyimethyi, pyrroiidinecarbonyl, tetrahydrofuranyimethyi and pyridyicarbonyiaminomethyl.
Another preferred radical Q ts a radical of formula
wherein R is hydrogen or CrC2-aikyi, hab- CrC2-aSkyl, halo- CrC2-alk;y1carboπy!, cyan©, CrCa-aikoxycarboπy!, CrCralHoxycarboπylmethyi, carboxy-C, -Qj-aikyi, aminocarbonyl, hS-moπo- or N,N-di-CrC2-aikylamiπocarb©nyl, halo- Ci-Craikyiaminocarbonylmethyi, halo- CrC2-aSKyiaminocarboπyimetr\ylaminoGarbonyl} haioviπyi, -G(O)N-CHN(CH^)2, -C(O)N=CHNHOCHa, unsubstituted or halogen or CrCa-alkoxy-substituted phenyl, benzyl or phenyiethyl, unsubstituted or hsiogeπ-substitυtβd 2-, 3- and 4-pyridyi and pyridyimethyi, pyridyicarbonyiaminomethy! , pyrrosidiπecarbony!, or 2-tetrahydrof υranyimethyS.
A suitable fused heierobicyciic ring system comprises, for example a 5- or β-memhβred heterocyclic ring having from 1 to 4, preferably from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, to which is attached an aππυiated ring; in addition said fused bicycHc system is further unsubstituted or substituted by one or more subsiituente as defsned before for Q deluding the preferences given. Those rings can be saturated sing or unsaturated rings.
Examples of fused heterσ-b&cyclic ring systems Q are illustrated in Exhibit 3 below. Exhibit si
wherein for (R)r each the above-given meanings and prefernces apply. In the above Exhibit 3, when the attachment point on the Q group is illustrated as floating, the Q group can be attached to the remainder of the formula (\) through any available carbon or nitrogen of the
Q group by replacement of a hydrogen atom. In addition, when the attachment point between (R)r and the Q group is illustrated as floating, (R)1. can be attached to any available carbon atom or nitrogen atom of the Q group.
Q is even more preferred the unsubstituted radical G-105, G106, Q-107, Q-108, Q-109, Q- 110 or G-111 , wherein r is 0 in each case. Particuiariy preferred fused bicyclic stαscfurøs Q are of formula
According to a preferred embodiment of the invention there is provided a compound of formula
including all geometric anά stereoisomers, N~oxides, and salts thereof, wherein for R1, R2, R3, Q and π each the above-given meanings and preferences apply, and m is an integer from 0 to 3.
In particular, n is an integer from 1 to 3, Rt is CrCa-haioalkyi, each Rg and R3 is independently selected from the group consisting of halogen, CrC2-aikyl, CrCz-hsioaikyl, Ct-C2- alkoxy, Cr-C2~haloaikoxy, C-rCa-haioalkyithio, -CU and -NO2, m is an integer from 0 to 2, in particular 0, and Q is either
(i) phenyl, which is unsubstituied or substituted by 1 to 3, Sn particular 1 or 2, same or different substituents selected from the group consisting of halogen, CrCraikyl, CrC2- haloalkyi, C,-C2-aikoxy, CrC2-ha!oaikoxy, CrQrhaloalkyiihio, -CN, -NO2, and υnsυbststuted or halogen-, CrC2~aikyh GrCrha!oaSkyS-, CrC2-aikoxy-, Ci-CVhaiαatkoxy-, -CN or -NO2- substituted phenyl or phenoxy, or is
(ii) a 5- or ø-membered heterocyclic ring having from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, which is further unsubstituted or substituted by halogen, CrCa-aikyi, Ci-C2-haioa!kyi, d-Cj-aikoxy, d-Cz-haloalkoxy, C1-C2- haioaikylthio, -CN, -NO2 or Ci-C^aikoxycarbonyi, or is
(iii) a fused bicyelic ring system comprising a 5- or 6-membered heterocyclic ring having from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, to which is attached an annulatβd ring, and which is further unsubsiituted or substituted by haiogβn, d-Craikyf, Ci-C2-haioaikyi, Ci~C2~alkoxy, Ci-C2-ha!oaikoxy, Ci-Ca-haioaikyithio, -CN, -NO2 or CrC4~alkoxycarbonyi.
A particuiariy prefeπeά embodiment of the invention relates to a compound of formula (Sa) above, wherein n is an integer from 1 to 3, Ri is CF3, each R2 is independently selected from the group consisting of halogen, CrCa-haioaikyl, Ci-C2-haloalkoxy and -CH, m is 0 and Q is either
(ii) a 5- or δ-mβmbδred hetsroaromatic ring selected from the group consisting of the above- given radicals Q-S1 Q-6, Q~7, Q-14, CM5, Q»16, Q-17, Q-24, Q-2β, Q-30, Q-31 , 0-32, Q-33, Q-34, Q-43, Q-47, Q-49, Q-50, Q-52 and Q-54, wherein r is an integer from 0 to 3 and R is independently selected from the group consisting of halogen, CrQralkyi, CrC2~ha!oa!kyi, d-C'2-aikoxy, CrC2-haioaikoxy, Ci-Crhaloalkyithiot -CN, -NO2 ^nύ CrC^alkoxycarbonyl, or is (iii) a fused bicycfic ring system of formula Q-107a, Q-107b or Q- 111a.
Another pr&iemύ embodiment of the invention relates to a compound of formula (ia) above, wherein n is an integer from 1 to 3, Ri is CF3, each R2 is independently selected from the group consisting of haiogβn, CrQrhaloalkyS, CrC2-ria!oaikoxy and -CN, m is O, and Q is a radical Q~5, Q-14, Q-24, G-25, Q-26, Q-30, Q-32, Q-34, Q-43, Q-44, Q-47, Q-49, Q-50 or Q-75, which is each unsubsfituted or substituted by one or more substifuents selected from the group consisting of CrC2~alkyi, halo- CrC2~aikyi, halo- Ct-C2- alkylcarbonyl, cyaπo, CrCralkoxycarbonyi, CrC4-alkoxycarbony1methyl, carboxy-Ci-Ca-
alkyi, amtnocarbonyl, N-mono- or N,N»di-Ct- C2-a!kylaminocaitsony!, haio- Crθ2- aikySaminocarbonyimβthyi, haio-CrCa-aikyiaminocarbonylmethylaminocarboπyl, halovinyi, ~ C(O)N~CHN{CH3)2, -C(O)N=CHNHOCH3, unβυbstituted or halogen or C1-C2-SIkOXy- substituted phenyl, benzyl and phenyieihyl, unsubstituted and halogen-substituted 2-, 3- and 4-pyridyl and pyridyimethyi, pyridylcarbonyiamsnomethyl, pyrrolidinecarbonyl, and 2~ tθtrahydrofuraπyimβthyi, or Q is a radical Q-10?a, Q-IOTb5 Q-111a or Q-134.
Another preferred embodiment of the invention relates to a compound of formula {ia} above, wherein n is an integer from 1 to 3, R1 is CF3, each R2 is independently halogen, rn is 0, and Q Is a radical Q-24a, Q-26a, Q-SOa, Q-32a, Q-32b, Q-34a, Q~44a or Q-47a, wherein R is hydrogen or d-Cralkyi, haio- Ci-C2 »alky1, haio- CrC∑-alkyicarbonyl, cyano, CrCV alkoxycarbonyl, CrC«~alkoxycarbony!methyi, carboxy-CrCz-alkyi, aminocarbony!, M-roono- or N.N-di-CrCa-alkylsrninocarbonyl, halo- CrCa-alkylaminocarboπylmethyl, ha!o~CrC2-alkyi- aminocarbonyimethylaminocarbonyl, haiovinyl, -C(O)N~CHN(CH3)a, -C(Q)N-CHNHOCH3, unsubstituted or haiogen or Ci-C2-aikoxy-substituted phenyl, benzyl or phenyiethyi, unsubstituted or haiαgen~$ub$ti£uted 2-, 3- and 4-pyridyi and pyridyimethyl, pyridyicarbonyi- aminomethy!, pyrrolidinecarbonyl, or 2~tøtrahydrofυranylmθthyi.
Compounds of this invention can exist as one or more stereoisomers. The various stereoisomers include enantlomers, diastereorners, atropisomers and geomeiήc isomers. One skilled in the art wiii appreciate that one stereoisomer may h® more active and/or may exhibit beneficial effects when enriched relative to the other stereoisomers) or when separated from the other stereoisomer(s). Additionally, the skilled artisan knows how to separate, enrich, and/or to selectively prepare said stereoisomers. The compounds of the invention may be present as a mixture of stereoisomers, individual stereoisomers, or as an optically active forot.
One skilled in the art wiH appreciate that not ail nitrogen containing heterocyclic rings can form N-oxides since the nitrogen requires an available ione pair for oxidation to the oxide; one skilled in the art wiii recognize those nitrogen containing heterocyclic rings which can form N-oxides. One skilled in the art will also recognize that tertiary amines can form N- oxides. Synthetic methods for the preparation of N-oxides of heterocyclic rings and tertiary amines are very well known by one skilled m the &ύ including the oxidation of heterocyclic rings <mή tertiary amines with peroxy acids such as peracetic and m-chloroperberszoic acid (MCPBA), hydrogen peroxide, alky! hydroperoxides such as t-butyl hydroperoxide, sodium
perborate, and dioxiranes such as dimethyl dioxirane These methods for the preparation of iV-oxides have been extensively described anύ reviewed In the literature.
One skiiied in the art recognizes that because of the environment and under physiological conditions salts of chemical compounds are in equilibrium with their corresponding nonsalt forms, salts share the biological utility of the nonsalt forms, Thus a wide variety of salts of the compounds of formula (i) are useful for control of invertebrate pests (i.e. are veterinariiy or agriculturally suitable). The salts of the compounds of formula (I) include acid-addition salts with inorganic or organic acids such as hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric, fumarie, lactic, maiβic, maionic, oxalic, propionic, salicylic, tartaric, 4~ toluenesuifonic or valeric acids. When a compound of formula (i) contains an acidic moiety such as a carbøxyisc acid or phenol, salts also inciude those formed with organic or inorganic bases such as pyridine, tπethylamine or ammonia, or amides, hydrides, hydroxides or carbonates of sodium, potassium, lithium, caicium, magnesium or barium. Accordingly, the present invention comprises compounds selected from formula (I), N-oxides and veterinary acceptable and agriculturally suitable salts thereof.
The compounds of the present invention made be prepared, for exarπpie, in analogy to the processes as outined in WO 2007/75459 on pages 29-31. Accordingly, the compounds of formula (!) or (Sa) may be prepared, for example, by cycioaddition of a compound of formula
with a nitriie oxide derived from an oximβ of fαrmuia
wherein A1-A6, B1-B3, R1, R2, Q and n each have the above-given meaning.
The reaction typicaily proceeds through the intβtrnediacy of an in situ generated hydroxamyi chloride, in a typicai procedure a chlorinating reagent such as sodium hypochlorite, N- chiorosuccinimide, or chioraroine-T is combined with the oxime in the presence of the styrene. Depending on the conditions amine bases such as pyridine or triethylamiπe may be necessary. The reaction can be run in a wide variety of solvents including tetrahydrofuran, diethyl ether, methylene chloride, dioxane, and toluene with optimum temperatures ranging from room temperature to the reflux temperature of the solvent.
The compounds of formula (!) or (Ia), wherein Q is a 5-membered N-linked heterocyclic ring can also be prepared by direct displacement of an aromatic leaving group of formuia
wherein ApAe, BrB3, R5, R2 and n each have the above-given meaning and Y is halogen, for example Br or F1 tosyiate, trifiate or nitro, with a compound of the formula
Q' ~ H (V), wherein Q' is m azoie heterocyclic ring, in the presence of a base, Typical azole heterocyclic rings of formuia (V) include optionally substituted pyrazoiβs, imidazoles, triazoles and teirazoles. Bromides can be displaced with the use of copper iodide and a palladium catalyst, see for example Kanemasa et a!., European Journal of Organic Chemistry, 2004, 685-709. For direct fluorine displacement the reaction is typically run m a polar aprotic solvent such as N.N-dirnethyiformamide or N,N-dimethyiacetamide and in the presence of an inorganic base such as sodium or potassium carbonate.
Another process for the preparation of compounds of the formula (I) or (ia), wherein Q is a phenyl, naphthyl or heteroaramatic ring, includes the Suzuki reaction via Pd~catalyzed cross coupling of an aromatic iodide or bromide of formuia (IV) above, wherein Y is Br or J1 with an aryi or heteroary! borσnic acid of formula
Q" - B(OH)2 (Vi).
Many catalysts are useful for this type of transformation; a typical catalyst is tetrakis- (triphenylphosphine)paiiadium(O). Solvents such as teirahydrofuran, acetonitriie, diethyl
ether and dioxaπe are suitable. The boroπic acids of Formula 6 &r& either commercially avaiiabie or can be prepared by known methods. Other methods including the Heck, StiHe, Kυroada and Buchwald-Hartwig coupling procedures offer many alternatives for introduction of Q heterocyclic groups. For leading references see for example Zificsak, Craig A and Hiasta, Dennis J., Tetrahedron, 2004, 60, 8991-9016.
The compounds of of formulae (Ii) and (SV) are known, for example, from WO 2008/49459 or may be prepared in analogy to the methods disclosed therein. The compounds of formula (HI) may be prepared, for example, in analogy to the processes disclosed in WO 2007/75459 for the preparation of compounds of formuis (3),
Th© compounds of the formula (i) according to the Invention are notable for their broad activity spectrum and are valuable active ingredients for use in pest control They are particularly suitable in the control of ectoparasites and to a certain extent also for controlling endoparasϊtes on and in animals and in the hygiene field, whilst being weli tolerated by warm-blooded animals, in the context of the present invention, ectoparasites are understood to be in particular insects, acari (mites and ticks), and crustaceans (sea lice). These include insects of the following orders: Lepidoptera, Coleoptera, Homoptera, Herniptera, Heteroptera, Diptera, Oictyoptera, Thysanσptera, Orthopiera, Anoplura, Siphonaptera, Mallophaga, Thysanura, isoptera, Psocoptera and Hymenoptera. However, the ectoparasites which may be mentioned in particular are those which trouble humans or animais and carry pathogens, for example Hies such as Musca domestica, Musca vetustissima, Musca autumnaiϊs, Fannia oanicuiaris, Sarcophaga carnaria, lυcilia cuprina, LuciHa sericata, Hypoderrna bovϊs, Hypoderma lineatum, Chrysomyia chloropyga, Dermatobia hominis, Cochiiomyia horninivorax, Gasterophiius intestinalis, Oestrus ovis, biting flies such as Haematobia irrilans irritans, Haβmatobia irritans exigua, Stomoxys calcitrans, horse-flies (Tabanids) with the subfamilies of Tabanidae such as Haematopαta spp. (e.g. Haematopota piuviaiis) and Tabanus spp, (e.g.Tabanus nigrovittatus) and Chrysopsiπaβ such as Chrysops spp. (e.g. Chrysops caβcutiens); Hippoboscids such as Melophagus ovinus (sheep Red); tsetse flies, such as Glossinia spp,; other biting insects like midges, such as Ceratopogonidae (biting midges), Simuisidae (Biackfiies), Psychodidae (Sandflies); but also biood-sucksng insects, for example mosquitoes, such as Anopheles spp, Aedes spp and Culex spp, fleas, such as Ctenocephalides fells and Cienocephaiides canis (cat and dog fleas), Xenopsyila cheopis,
Pυiex irritaπs, CeratopftyOus gafSlnae, Dermatophiius penetrans, blood-sucking lice (Aπoplυra) such as Linognathus spp, Haematopinus spp, Solenopotes spp, Pedicuius humaπis; but also chewing lice (Mafiophaga) such as Bovicoia (Damatinia) ovis, Bovicoia (Damalinia) bovis and other Bovicoia spp. . Ectoparasites also include members of the order Acarina, such as mites (e.g. Chorioptes bovis, Cheyietieiia spp., Dermanyssυs gafifnae, Ortnithonyssus spp., Demodex cams, Sarcoptβs scabiei, Psorαptes ovis and Psorβrgatøs spp. and ticks. Known representatives of ticks are, for example, Boopfciius, Arnblyomma, Anocenior, Derroaeentor, Haemaphysalis, Hyaiomma, Ixodes, Rhipicentor, Margaropus, Rhipicephalus, Argas, Otobius and Qrnithodoros and the like, which preferably infest warmblooded animals inciuding farm animais. such as cattle, horses, pigs, sheep and goats, poultry such as chickens, turkeys, guineafowls and geese, fur-bearing animais such as mink, foxes, chinchillas, rabbits and the like, as well as companion animals such as ferrets, guinea pigs, rats, hamster, cats and dogs, but aiso humans.
The compounds of the formula (!) according to the invention are also active against all or individual development stages of animal pests showing norma! sensitivity, as well as those showing resistance to widely used parasiticides. This is especiaily true for resistant insects and members of the order Acarina. The insecticldai, ovicidai and/or acaricidai effect of the active substances of the invention can manifest itself directly, i.e. killing the pests either immediately or after some time has elapsed, for example when moulting occurs, or by destroying their eggs, or indirectly, e.g. reducing the number of eggs laid ^nό/ot the hatching rate, good efficacy corresponding to a pestiddaf rate (mortality) of at least 50 to 60%.
Compounds of the formula (I) can afso be used against hygiene pests, especially of the order Diptera of the families Muscidae, Sarcophagidae, Anophiiidae and Cuticidae; the orders Orthoptera, Dictyoptera (e.g. the family Btøttidae (cockroaches), such as Blateiia gerrrsansca, Biaita oπentaiis, Peripianeta americana) and Hymenoptera (e.g. the families Formicidaø (ants) and Vespidae (wasps).
Surprisingly, the compounds of formula (I) are also effective against ectoparasites of fishes, especially the sub-class of Copepoda (e.g. order of Siphonostomatoidae (sea lice), whiist being we!i tolerated by fish.
Compounds of the formula (I) also have sustainable efficacy on parasitic mites ®nά insects of plants. In the case of spider mites of the order Acarina, they are effective against eggs, nymphs and adults of Tetranychidae (Tetmnychus spp. and Pmonychus spp,).
They have high activity against sucking insects of the order Homopters, especially against pests of the families Aphididaβ, DΘlphaάdaβ, Cicad&Uidae, Psyilidae, Loccidae, Diaspidiάae and Eriophydidae (e.g. rυst mite on citrus fruits); the orders Hemipt&ra, Hetβroptera and Thysanoptem, and on the plant-eating insects of the orders Lepidoptera, Coleoptera, Diptem and Qriboptera
They are similarly suitable as a soil insecticide against pests in the soil.
The compounds of forrouia (1) are therefore effective against aii stages of development of sucking insects and eating insects on crops such as cereals, cotton, rice, maize, soya, potatoes, vegetables, fruit, tobacco, hops, citrus, avocados and other crops.
The compounds of formula i are also effective against piant nematodes of the species Meioidogyne, Heterodera, Pratyienchus, Dityi&nchus, Radophoius, Rizoglyphus etc.
Certain compounds of the formuia (!) seem to be also effective against certain species of helminths.
Helminths are commercially important because they cause serious diseases in mammals and pouitry, e.g. in sheep, pigs, goats, cattle, horses, donkeys, camels, dogs, cats, rabbits, guinea-pigs, hamsters, chicken, turkeys, guinea fowis and other farmed birds, as weϊi as exotic birds. Typical nematodes are; Haemonchus, Trichostrcngyius, Qstertagia, Nematotiirus, Cooperia, Ascaris, Bunostonum, Gesophagostonum, Charbertia, Trichuris, Strongyius, Trϊchonema, Dictyocaulus, Capsliarsa, Heterakis, Toxocara, Ascaridia, Oxyuris, Ancyiostoma, Uncinaria, Toxascaris and Parascaris. The trematodes include, in particular, the family of FasciαikJeae, especiaify Fasciola hepatica.
The good pesticida! activity of the compounds of formula (!) according to the invention corresponds to a mortality rate of at least 50-60% of the pests mentioned, more preferably to a mortality rate over 90%, most preferably to 95-100%. The compounds of formula (I) are preferably employed internally and externally in unmodified form or preferably together with the adjuvants conventionally used in the art of formulation and may therefore be processed in a known manner to give, for example, liquid formulations (e.g. spot-on, pour-on, spray-on, emulsions, suspensions, solutions, emulssfiable concentrates, solution concentrates), semisolid formulations (e.g. creams, ointments, pastes, gels, liposomal preparations) and solid
preparations (e.g. food additives tablets including e. g, capsules, powders including soluble powders, granules, or embβddings of the active ingredient In polymeric substances, like implants and microparticies). As with the compositions, the methods of application ar® selected in accordance with the intended objectives anύ the prevailing circumstances. The formulation, Ke. preparations containing the active ingredient of formula {!}, or combinations of these active ingredients with other active ingredients, and optionally a soiid, semi-solid or liquid adjuvant, are produced in a manner known per se, for example by intimately mixing, kneading or dispersing the active ingredients with compositions of excipients, whereby the physiological compatibility of the formulation excipients must be taken into consideration.
The solvents in question may be: alcohols (aliphatic and aromatic), such as benzylaicohol, ethanof, propanol, isopropanol or butanol, fatty aieohois, such as oieyl alcohol and glycols and their ethers anό esters, such as glycerin, propylene glycol, dipropylene glycol ether, ethylene glycol, ethylene glycol monomethyi or -ethyl ether and butyl dloxytoi, carbonates, such as propylene carbonate, ketones, such as cydohexanone, isophorone or diacetanoS aicohoi and polyethylene glycols, such as PEG 300. In addition, the compositions may comprise strong polar solvents, such as N-rnethyi-2-pyrroiidone, dimethyl sulfoxide or dimefhyiforroamide, or water, fatty acid esters, such as ethyl oleate or isopropyf pa imitate, vegetable oils, such as rape, castor, coconut, or soybean oil, synthetic mono-, di-, triglycerides like e.g. glyceryl monostearafe and medium chain triglycerides and also, if appropriate, silicone oils. The mentioned ingredients may also serve as carrier for particulate application froms.
As ointment base rβsp. structure building ingredients the following excipients may be used: Petroleum based substances, such as Vaseline or paraffines, bases made from wool fat, like e.g. lanolin or lanolin alcohols, polyethylene glycols like e.g. macrogols and Upiύ bases like e.g. phospholipids or triglyceride, such as hydrogenated vegetable oils. The use of emuisifiers, wetting agents and spreading agents may also be required, in general, lecithins like soy lecithin, salts of fatty acids with alkaline earth and alkali metals, aikyl sulfates like sodium cetylstearyl sulphate, chelates, fatty alcohols like cetyi alcohol, sterols like cholestesterol, pofyoxyeihylene sorbitan fatty acid esters like poiysorbate 20, sorbitan fatty acid esters like sorbitan mono laureate, fatty acid esters and fatty aicohoi ethers of polyoxyethyiene like poloxyl olβyl eih&r, polyoxypropyiene pdyoxyβthytene block copolymers as e.g. Piuronic™ , saccharose esters like saccharose distearate, poiygiyceryl
fatty acid esters like poiygiycβrøi øleate and fatty acid esters like e.g. ethyl oleate or isopropyimyristate.
The formulations may aiso include gelifying and stiffening agents, iikø e.g. polyacryiϊc acid derivatives, cellulose ethers, polyvinyl alcohols, polyvinylpyrroiidons and fine disperse siliciurn dioxide.
As polymeric agents with controlled release properties, may be applied derivatives made by e.g. pofyiactte acid, poiylactic cogiycoiic acid, poly orthoester, poiyethylene carbonate, poly anhydrids and starch and PVC based matrices,
The addition of penetration enhancers like ketones, sulfoxides, amides, fatty acid esters and fatty alcohols may be necessary.
Aiso preservatives like sorbic acid, benzyl alcohol and parabenes, and antioxidants as e.g. alpha tocopherol may be added.
The active ingredient or combinations of the active ingredient may aiso applied in capsules, like hard gelatine capsules or soft capsules.
The binders for tablets and boli may be chemically modified polymeric natural substances that are soluble in water or in alcohol, such as starch, ceiluiose or protein derivatives (e.g. methyl cellulose, carboxymethy! cellulose, ethyihydroxyethyl celiuiose, proteins such as zein, gelatin and the like), as well as synthetic polymers, such as polyvinyl alcohol, polyvinyl pyrroHdone etc. The tablets also contain fillers (e.g. starch, microcrystalline cellulose, sugar, lactose etc.), lubricants (e.g. magnesium stearate), giidants (e.g. colloidal silicon dioxide) and disintegrants (β,g. cellulose derivatives) and acid resistant coatings, like e.g. acrylic acid esters.
The compounds of formula (I) according to the invention may be used alone or in combination with other biocides. They may be combined with pesticides having the same sphere of activity e.g. to increase activity, or with substances having another sphere of activity e.g. to broaden the range of activity. It can also be sensibi© to add so-called repellents. For example, an case of a compound of formula (!) having a particular efficacy as aduiticlde, e.g. since the compounds of formula (!) are effective in particular against the adult stage of the target parasites, the addition of pesticides which instead attack the juveniie stages of the parasites may be very advantageous. In this way, the greatest part of those parasites that produce great economic damage will be covered. Moreover, this action will contribute substantially to avoiding the formation of resistance. Many combinations may aiso lead to synergistic effects, i.e. the total amount of active ingredient can be reduced, which is desirable from an ecological point of view. Preferred groups of combination partners and
especially preferred combination partners are named in the following, whereby combinations may contain one or more of these partners in addition to a compound of formula (i). Suitable partners in the mixture may be biocidβs, e.g. the insecticides anά acarieides with a varying mechanism of activity, which are named in the foiiowiπg and have been known to the person skilled in the art for a long feme, e.g. chitin synthesis inhibitors, growth regulators; active ingredients which act as juvenile hormones; active ingredients which act as aduitiddes; broad-band insecticides, broad-band acaricides and nematieides; and also the well known antheSminthies and insect- and/or aearrci-deterring substances, said repellents or detachers. Non-Hmitative examples of suitable insecticides and acaricides are mentioned in WO 2009/071500, compounds Nos. 1-284 on pages 18-21. Non-limitative examples of suitable anthelmintics are mentioned in WO 2009/071500, compounds (A1 ) - (A31 ) on page 21. Non-iimitative examples of suitable repellents and detachers are mentioned in WO 2009/071500, compounds {R1 } ~{R3) on page 21 and 22. Non-limitative examples of suiiabie synergists are mentioned in WO 2009/071500, compounds (S1 ) -(S3) on page 22. The said partners in the mixture are best known to specialists in this field. Most ar& described in various editions of the Pesticide Manual, The British Crop Protection Council, London, and others in the various editions of The Merck Index, Merck & Co., Inc., Rahway, New Jersey, USA or in patent literature. A iist of suitable partners including a reference is disclosed in WO 2005/058802 on pages 16-21, No. (!) to (CLXXXIH). As a consequence of the above details, a further aspect of the present invention relates to a combination preparation for the control of parasites on warm-biooded animals, characterised in that It contains, in addition to a compound of formula (!), at least one further active ingredient having the same or different sphere of activity mό at ieast one physiologically acceptable carrier. The present invention is not restricted to two-fold combinations. As a rule, the insecticidai and acaricidai compositions according to the invention contain 0.1 to 99 % by weight, especially 0.1 to 95 % by weight of one or more active ingredients of formuia (i), 99.9 to 1 % by weight, especially 99.8 to 5 % by weight of a solid or liquid admixture, including 0 to 25 % by weight, especially 0.1 to 25 % by weight of a surfactant Application of the compositions according to the invention to \h& animals to be treated may take piace topically, peroraily, parenteral^ or subcutaneoυsiy, the composition being present, for example, in the form of solutions, emuisions, suspensions, (drenches), powders, tablets, boJi, capsules, collars, eartags and poυr-on formulations. Preferred topicaS formulations are understood to refer to a ready-to-use solution in form of a spot-on, pour-on or spray-on forrnuiation often consisting of a dispersion or suspoemulsion
or a combination of active ingredient and spreading auxiliaries. The expression spot-on or pour-on method is understood to refer to a ready-to-use concentrate intended to be applied topically &nά locally on the animal This sort of formulation is intended to be applied directly to a relatively smaii area of lhe animal, preferably on the animal's back and breech or at one or several points along the line of the back and breech. !t is applied as a iow volume of about 0.05 to 1 mi per kg, preferably about 0.1 mi per kg, with a total volume from 0.1 to 100 ml per animal, preferably limited to a maximum of about 50 mi. However, it goes without saying that the total volume has to be adapted to the animal that is in ne&ά of the treatment and will clearly be different, for example, in young cats and in cattle. These pour-on and spot-on formulations are designed to spread ai! around the animal giving protection or treatment to almost any part of the animal. Even so the administration Is carried out by applying a swab or spray of the pour~on or spot-on formulation to a relatively smaii area of the coat, one observes that from the active substance is dispensed almost automatically over wide areas of the fur owing to the spreading nature of the components in the formulation and assisted by the animal's movements.
Pour~on or spot-on formulations suitably contain carriers, which promote rapid dispersement over the skin surface or in the coat of the host animal, and are generally regarded as spreading oils. Suitable carriers are e.g. oily solutions; alcoholic mά isopropanoiic solutions such as solutions of 2-octyidodecanoi or oleyi aicohol; solutions in esters of monocarboxylic adds, such as isopropyi myhstate, isopropyi paSmitate, lauric acid oxalate, oleic acid oley! ester, oleic acid decyi ester, hexyl iaurate, oleyi oieate, decyi oieaie, capric acid esters of saturated fat aϊcohois of chain length Ci2-C18; solutions of esters of dicarboxyiie acids, such as dibutyi phthalate, diisopropyl isophthaiate, adipic acid diisopropyi ester, di-n-butyf adipate or aiso soiutions of esters of aliphatic acids, e.g. glycols. It may be advantageous for a dispersing agent to be additionally pmsβnt, such as one known from the pharmaceutical or cosmetic industry. Examples are 2-pyrroiidone, 2-(N-a!ky!)pyrroϋdone, acetone, polyethylene glycol and the ethers and esters thereof, propylene glycol or synthetic triglycerides. The oily soiutions include e.g. vegetable oils such as olive oil, groundnut oil, sesame oil, pine oil, linseed oil or castor oii, The vegetable oils may aiso be present in epoxidised form. Paraffins and silicone oils may also be used.
A pour-on or spot-on formulation generally contains 1 to 98.9 % by weight of a compound of formula (I), 0.1 to 80 % by weight of dispersing agent anύ 1 to 98.9 % by weight of solvent. The pour-on or spot-on method is especially advantageous for use on herd animals such as cattle, horses, sheep or pigs, in which it is difficult or time-consuming to treat all the animals
orally or by injection. Because of its simplicity, this method can of course also be used for ail other animals, including individual domestic animals or pets, and is greatly favoured by the keepers of the animals, as it can often be carried out witliout the specialist presence of the veterinarian.
Whereas it is preferred to formulate commercial products as concentrates, the end user will often use dilute formulations. However, this depends on the mode of administration. Oraϋy administered products are most often used in diluted form or as feed additives, whereas commercial pour-on and spot-on formulations are normally ready-to~use concentrates,
Such compositions may also contain further additives, such as stabilisers, anti-foaming agents, viscosity regulators, binding agents or tackifiers, as weii as other active ingredients, in order to achieve special effects. lnseeticidal and acaricida! compositions of this type, which are used by the ®nά user, similarly form a constituent of the present invention.
In each of the processes according to the invention for pest control or in each of the pest control compositions according to the invention, the active ingredients of formula (!) can be used in ail of their sterie configurations or in mixtures thereof.
The invention also includes a method of prophyfacticaiiy protecting animals, especially productive livestock, domestic animals s>nύ pets, against parasitic helminths, which is characterised in that the active ingredients of formula (I) or the active ingredient formulations prepared therefrom are administered to the animals as an additive to the feed, or to the drinks or also in solid or liquid form, orally or by injection or parenteraliy. The invention also includes the compounds of formula (I) according to the invention for usage in one of the said processes
The following examples serve merely to illustrate the invention without restricting it, the term active ingredient representing any substance as described in the preparation examples,
In particular, preferred formulations are made up as follows:
{% - percent by weight)
The active ingredient is dissolved in methylene chloride, sprayed onto the carrier and the solvent subsequently concentrated by evaporation under vacuum. Granulates of this kind can be mixed with the animai feed, (ii) active ingredient 3 % poiyethyiene glycol (mw 200) 3 % kaolin 94 %
{mw - molecular weight)
The fineiy ground active Ingredient is evenly applied in a mixer to the kaolin which has been moistened with poiyethyiene glycol. In this way, dust-free coated granules Site obtained.
2. Tablets or boii
3 active ingredient 33.00 % methyiceliuiose 0.80 % silicic acid, highly dispersed 0,80 % com starch 8.40 %
Ii lactose, cryst. 22.50 % corn starch 17.00 % microcryst. cellulose 16.50 % magnesium stearate 1.00 % i Methyl cellulose is stirred into water. After the material has swollen, silicic acid is stirred in anά the mixture homogeneously suspended. The active ingredient and the corn starch are mixed. The aqueous suspension Is worked into this mixture and kneaded to a dough. The resulting mass is granulated through a 12 M sieve and dried. M Ail 4 exdpients are mixed thoroughly.
IiI The preliminary mixes obtained according to I mά Si are mixed and pressed into tablets or boli.
3. fnjeetabies
A. Oily vehicle (slow release) (i) active ingredient 0,1-1 ,0 g groundnut oil »ύ 100 ml (ii) active ingredient 0.1-1.0 g sesame oil ad 100 mi
Preparation: The active ingredient is dissolved in part of the oil whilst stirring and, if required, with gentle heating, then after cooling made up to the desired volume and sterile-filtered through a suitable membrane filter with a pore size of 0.22 μrn.
B Water-miseibie solvent (average rate of release) (i) active ingredient 0.1-1.0 g
4~hydroxymethyi~1 ,3-dioxolanβ {glycerol format) 40 g
1 ,2-propanediol ad 100 ml (Ii) active ingredient 0.1-1.0 g glycerol dimethyl keiai 40 g
1 ,2-ρropanedioi ad 100 ml
Preparation; The active ingredient is dissolved in part of the solvent whilst stirring, msάe up to the desired volume and sterile-fiftered through a suitable membrane fitter with a pore size of 0.22 μm.
C. Aqueous solubilisate (rapid release) (i) active ingredient 0,1-1 ,0 g pσiyethoxylated castor oil (40 ethylene oxide units) 10 g
1 ,2-propanediol 20 g benzyl alcohol 1 g aqua ad inject, ad 100 ml (ii) active ingredient 0.1-1.0 g poiyethoxyiated sorbitan monooieate (20 ethylene oxide units) 8 g
4~hydroxymβthyi-1 ,3-dioxoiane (glycerol formal) 20 g benzyl alcohol 1 g aqua ad inject, ad 100 mi
Preparation: The active ingredient is dissolved in the solvents anά the surfactant, and made up with water to the desired volume. Sterile filtration through an appropriate membrane filter of 0,22 μm pore size. 4. Pour on
(i) active ingredient 5 g isopropyi myristate 10 g isopropanoi ad 100 m!
(ii) active ingredient 2 g hexyi Ia urate 5 g medium-chained triglyceride 15 g ethanof ad 100 ml
(ill) active ingredient 2 g otey! oieate 5 g
N~meihy!-pyrroϋdone 40 g ssopropanoi aύ 100 mi
5. Spot on
(i) active ingredient 10-15 g diethySeneglycoi monøethyietber ad 100 mi
(ii) active ingredient 10-15 g octyl palrnitate 10 g isopropanol ad 100 ml
(iii) active ingredient 10-15 g isopropano! 20 g benzyl alcohol ad 100 m!
6. Spray on
(i) active ingredient 1 g isopropano! 40 g propylene carbonate aύ 100 mi
(ii) active ingredient 1 g propylene glycol 10 g ad 100 mi
The aqueous systems may also preferably be used for orai and/or intraluminal application. The compositions may also contain further additives, such as stabilisers, e.g. where appropriate epoxidised vegetable oiis {epoxidised coconut oil, rapeseed oil, or soybean oil); antifoams, e.g. silicone oil, preservatives, viscosity regulators, binders, tackifiers, as weif as fertilisers or other active ingredients to achieve special effects.
Further biologically active substances or additives, which are neutral towards the compounds of formula (!) and do not have a harmful effect on the host animal to be treated, as well as mineral salts or vitamins, may also be added to the described compositions.
The following examples serve to illustrate the invention. They do not limit the invention. The letter 'h' stands for hour. The starting materials am known and partially commercially available or may be produced in analogy to methods known per se.
Analysis of the purified samples is in each case done using a Waters Autopurificatioπ (HPLC/MS) system with a reversed phase column (Daisogel SP-120-ODS-AP 5μm, 150X3mm) from Bischoff, Lβonbβrg, Germany. The samples are characterized by m/s and retention time. The above-given retention times relate in each case to the use of a soivent system comprising two different solvents, solvent A: H2O + 0.01% HCOOH, and solvent 8: CH3CN + 0.01% HCOOH). Said two solvents A and B are employed at a flow rate of 2.00 ml/mjπ with a time-dependent gradient as given in the Table:
Preparation of 1 -{4-[5-(3)5-dichioro-pheπyl)-5-tnfiuoromethy1~4<5-dihydro4soxazol-3'"yi]- naphthaien-1-y!}-1H-[1 ,2,4]triazole (compound 1.1, Table 1).
Sjβg A; 3,3,3-TrifluoroprOpene (3.2 g), potassium carbonate (4.6 g) and bis- (tπphenyiphGsphine)-palladium chloride (0.2 Q) Bm added to a solution of 3,5- dichlorophenylooronic acid in THF (20 mi) under nitrogen. After 3 hours at reflux, the reaction is quenched with ethyl acetate (δO mi) and water {50 ml). The organic phase is then extracted with water and with s saturated aqueous solution of NaCi1 dried over Na2SO4 and concentrated in vacuo. The crude product is purified on silica ge! (35x45 mm) using heptane (150 ml) as eiuanf Io yield 1,3-dich!orθ"5~(1-trif!uoromethyi-viπyi>'benz8nβ (2.7 g), as a colorless oii. MS (HPLC/MS): no ionisatloπ. Retention time: 5.10 rnsn.
.Stesϋi SnCU (13 mi, 1M solution In dichiorcmethane) is added to a solution of djchtormethyi-πiethyiethβr in dichioromethane (6 m!) at O0C. After 1 hour at 00C, a solution of
1-fiuoro-naphtatene (1.46 g) in dtohioromethane (4 ml) is added over 10 minutes. The reaction is stirred overnight at room temperature and then quenched with cøict water (30 ml). The organic phase is separated anά the aqueous phase is extracted 3 times with dichiorømβthane. The combined organic phases are dried over Na3SO4 anά concentrated in vacuo to yield 44luoro-naphihatene~1-carbaidehyde (1.73 Q1 dark brown crystals). The crude product obtained is used without further purification. MS (HPLC/MS): no ionisatioπ. Retention time: 3.88 min.
StejLCj, 1,2,4-triazoie (248 rog) is aύάβό to a solution of 4-fluoro~naphthalene-1- carbaldehyde (522 mg) and potassium carbonate (49? mg} in DMF (4 ml), After 3 hours at room temperature, the reaction is quenched with water {40 ml) and ethyi acetate (50 mi). The organic phase is separated and the aqueous phase is extracted two times with ethyi acetate. The organic phases are combined, extracted with water and with a saturated aqueous solution of NaCI, dried over Na2SO4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yieid 4~[1,2,4]triazoi-1-ykιaphthaiene-1- carbaktehyde (480 mg) as beige crystals. MS (HPUC/MS): 224 (MH+), Retention ϊime: 2.97 min.
Step D: Sodium acetate {282 mg) is added to a solution of hydrαxyiamtne hydrochloride (193 mg) anύ 4~(1 >2,4]tdazoi~1-yi~naphtha!ene-1-carbaidehydβ (4S5 mg) in THF (18 mi), water (2 ml) anά DiVISO (2 ml). After 5 hours at room temperature, the reaction is quenched with water and ethyi acetate. The organic phase is separated and the water phase is extracted with ethyi acetate. The combined organic phases are extracted with water and with a saturated aqueous solution of NaCI, dried over Na2SO4 and concentrated in vacuo to yieid 4-[1,2,4]triazol~1~yl~naphthaiene-1-carbaldehyde oximβ. The crude product obtained (390 mg, beige crystais) is used without further purification. MS (HPLC/MS): 239 (MH*). Retention time 2.78 min.
StepjE: tø-chiσrøsuccinirred (220 mg} is added to a solution of 4~[1 ,2,43tria2θl~1~yi~ naphtha!ene-1-carbaldehyde ©xfrne (390 mg) in DMF (5 mi). After 45 minutes at 400C, the reaction is cold down to 00C anύ 1 ,3~dichioro~5~(14rifluoromethyl-vinyi)-benzene (394 mg, Example 1, step A) and tπetriylarnine (0.25 ml) are added. The reaction is then further reacted at room temperature for 3 hours. Water is added (SO ml) and the reaction mixture is extracted two times with ethyi acetate. The organic phase is extracted with a saturated aqueous solution of NaCi, dried over Na2SO4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yieid 1-{4-[5-{3,5~dichk>ro-phenyi)-S-
trifiuoromethyi-4,5-dihydro-isoxazo["3-yl]~naphthalen-1-yl}-1H-[1,2,4]triazofe (SOO mg) as beige crystals. MS (HPLC/MS); 47? (MH+), Retention time: 5.20 min.
Example 2
Preparation of 5-{3,5-dich!oro~ρheπy3)-3-[4~{1f/~pyrazol-4-yJ}-naphthaien-1-yl]~5- trifluoromethyl-4>5~dihydro-isoxazøle (compound 1.17, Table 1 )
Step A: A solution of benzoyl peroxide {87 mg) in trifiuorotoluene (40 mi) is refiuxed for 30 min under nitrogen. The reaction mixture is cooled to 78 0C and 1-bromo-4-methyl- naphthaiene (1.6 mi) anά W-bromosuccinimide (2.0 g) are added. The reaction is stirred for 1.5 hours at 78 0C and filtered. The filtrate is concentrated in vacuo and the residue crystallized in heptane to yield 1~bromø-4-bromometnyi-naphfchaien8. The crude product is used in the next step without further purification.
Step B: Sodium bicarbonate {1.4 g) is added to a solution of crude 1-bromo-4~bromomethyl~ naphthalene (2.5 g) in DMSO (25 rn!). The reaction is stirred for 3h at 95 "C and cooled down to room temperature. Water is added and the mixture is extracted with ethyi acetate. The organic phase is dried over MgSO4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield 4~bromo~naphtha!ene-1-carbafdehydθ as colorless crystals.
Step C: Hydroxyiamin© (50% aqueous, 0.37 ml) is added to a solution of 4-bromo- naphthalene-1-earbaidehyde (0,7 g) in methanol (8 ml). After 1δ hours at room temperature the reaction is concentrated in vacuo to yield crude 4-bromo-πaphthalenδ-i-carbaidehyde oxime, which Is used in the next step without further purification.
Step D: W-chiorosucclnirnid (134 mg) is added to a solution of crude 4~bromo~naphthalene-1- carbaidehyde oxime (250 rog) in DMF (3 mi). After 1 hour at 40 0C, the reaction is cooled down to 0 "C and 1,3-dichlorα-5-{1-trifiuoromethy!-vinyl}-bβnzene (240 mg) and trieihyiamine (0.15 m!) are added. The reaction is further reacted at room temperature for 16 hours. Water is added and the reaction mixture is extracted with ethyl acetate. The organic phase is washed with a saturated aqueous solution of NaCt, dried over MgSO4 and concentrated in vacuo. The catde product is purified on a semi-preparative HPLC to yield 3~(4-brømθ" naphthalen-i-yi^δ-CS.S-dichloro-phenyO-S-triflυoromethyl^.δ-dihydro-isoxazole as coiorless crystals: m.p. 127-130 0C.
Ste£^Li-rerf-butoxycafbonyi~4-W-pyrazoteboronic acid pinacxsi ester (126 mg), Tris(dibenzyltd8neacetone}dipa[iadium{G) [Pd2(dba}3 (7mg)], KiPO4 (185 mg) and 2- dicyclohexyiphosphiπno-2'(6'-d!m8thoxybipheπy! {12 mg) are added to a solution of 3-{4- bromo~naphtha!en~1 -y^δ^S.S-dichloro-pheπyO-δ-trifiuoromethyi^.δ-dihydfo-isoxazole (140 mg) in dry toluene (2.4 ml). After 18 hours at 75 0C the reaction is cøoied down to room temperature and fiitraied. The filtrate is concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield 4~{4"[5-(3>5-dichiorθ'phenyI)-54rifiuoromethy!~415~ dihydro-isoxazol-S-yil-naphthalβn-i-yi^pyrazoie-i-carboxyiic acid terf-butyl ester (compound 1.10, Table 1) as a colorless oii.
Step F: Trifiuoroacetic acid anhydride (0.18 mi) is added to a soiution of_4-{4-[5-(3,5- dfChioro-phenyO-S-trifluoromethyi^.S-dshydro-isoxazol-S-y^naphthalen'i-yl^pyrazoie-i- carboxyiie acid t&rt~buty\ ester (135 mg) in didiloromethane (1 ml). After 1 hour at room temperature an aqueous soiution of sodium hydroxide (30%) is added. The aqueous phase is extracted with dϊchiorornethane and the combined organic phases are dried over MgSQ* and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield 5-(3,5»dichioro~ρheny1)-3-f4-(1H-pyra2θi-4-yi)-naphthalen-1-yi]-5-triflυoromethyi-4,5- dihydro-ssoxazoie as a coloriess solid: m.p. 117-120 0C (compound 1.17, table 1 ).
Example 3
Preparation of 5--(3,5-dichioro-pheny1)-3-{4-[1 -(2,2,2-trifluoro-ethy1)-1 H-pyrazoi-4-yt]- naphthaiβn-i-yl^δ-triflυoromethyW.δ-dihydro-isoxazote (compound 1.18, Table 1)
Sodium hydride (2 mg) is added to a solution of 5-(3,5-dichloro-pheπyi)"3-[4»(1H~pyrazol-4- yi)-naphthaien-1-yll-5-trif!uQramethyf-4,5-dihydro-isoxazoie (32 mg, Example 2) in DMF (0.4 ml) at 0 'C. After 1 hour at room temperature 2-iodo-1 ,1 ,1,-trέfluoroethane (18 mg) is added and the reaction mixture is further stirred for 18 hours at room temperature. Water is added and the reaction mixture is extracted with ethy! acetate. The organic phase is washed with a saturated aqueous solution of NaCI, dried over MgSC^ and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield 5-{3(5-dichloro-phenyf)-3-{4-[1~ (2,2,2-trifluorø-βthyl)-1H-pyra2θi-4-yl3-r»aphthal8π-1-yi}-5-trifluoromethyl-4,5~dihydro~ isoxazole (compound 1.18, Tabie 1} as a coioriess wax.
Example 4
Preparation of 2-{4-{4-[5-(3>5-dichioro»ph8ny!)-5-trifluorOm8t1iyW,δ-dihyciro-isoxa2θS-'3-yi]- naphthalen-1-yl}-[1(2,3]triazol-1-yim8thyl)-pyridfne (compound 1.27, Table 1)
SIULAI Trimethylsilyiacetylene (0.42 ml), triethyiamine (0.42 mi), Cui (38 rng) and Pd(PPSIa)2Ci2 (98 mg) are added to a solution of 3~(4~&rorno-naphthaien-1-yiHH3,5~ dich!oro-pheny!)-5-trifluoromethyM,5-dihydro-isoxa2:oie (1.0 g, example 2 step D) in DMF (10 mi). The reaction is stirred for 16 hours at room temperature. Water is added ars4 the reaction mixture is extracted with ethyi acetate. The organic phase is washed with a saturated aqueous solution of NaCi, dried over MgSO4 and concentrated in vacuo. The crude 5-(3,5-dicri!oro~pheny!)"54rifiuoromethyl-3-(4-tπmethylsiiany!ethynyi~naphthaieπ-1-yl)- 4,5~dihydrQ~isoxa2αie is used in the n&xi step without further purification.
Step 8: A soiution of tβtrabutyiamrπonium chloride (1 M in THF, 2.25 ml) is siowly added to a solution of crude 5"(3,5-dichloro-phenyi>5'triffuoromethy!-3-(4-tπmethyisiiany!8thyπyi- naphtbaien-1-yi)~4.5~dihydro-isoxazole (step A) in THF (25ml) at δ°C. After 3 hours at 5'C a saturated soiution of ammonium ehioride and ethyl acetate are added. The organic phase is washed with a saturated aqueous solution of NaC!, dried over MgSO4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield S-(3,5-dtch!oro- phenyi}~3-(4-ethynyl-πaphthaien-1 -yi)-5-tπfiuϋrømethyl-4,5-dihydrø-isoxazole as a brown soisd.
StejHC: Cu! (88 mg) is added to a soiution of 2-azidomethyl~pyrϊmidine (62 mg), N,N- dlfsopropyiethylamine (DtPEA, 2.0 m!) and 5"(3,5-dichloro-phenyi}-3-(4-ethyny!-naphtha!en- 1~y!)-5-trifluoromethyi-4,δ-dihydro-lsoxazole (100 rng) in OMF (1.0 rni). After 16 hours at room temperature, the reaction is quenched with a saturated soiution of NaHCO3 and extracted with dichforomethanø. The combined organic phases &r& dried over MgSO4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield 2- (4-{4~{5-(3,5-dich!oro-phenyi)~5~tnfluoromethyl~4<5-c!ihydro~isoxasoi-3-ylJ-naphthalen-1~yl}~ [I^.SJtriazoi-i-ylmethyi^pyridine as a beige foam, (compound 1.27, TabSe 1 ),
Example 5
Preparation of N-{1 -{4-[5-(3,5»diGhioro«phen^)-5-tr!f!uoromethy!-4>5-dihydro-isoxazol-3-yi]~ naphthalen-i-yiJ-IH^I^.SItriazoW-ylmethyO-isonicotinamide (compound 1.61 , Table 1 )
Step A: Sodium acetate (4.8 g) is added to a soiution of hydroxyiamine hydrochloride (2.9 g) and 4-ftuoro~naphthaiene~1~carbaicfehyde (6,1 g, example 1 step B) In THF (40 ml) and
water (10 mi). After 3 hours at room temperature, the reaction is quenched with water and ethy! acetate. The organic phase Is separated and the water phase is extracted with ethy! acetate. The combined organic phases are washed with water anά with a saturated aqueous solution of NaC!, dried over Mg2SO4 and concentrated in vacuo to yield 4~fiuoro~ naphthafene-i-earbaidehyde oxime. The crude product obtained is used without further purification,
StejtBi Λ/~chlorosuccinirnid (4.? q) is added to a solution of 4«fiuoro-naphthaiene-1- carbaldehyde oxime (8.6 g, example 5 step A) in DMF (50 mi). After 45 minutes at 400C, the reaction is cooied down to 00C and 1«3-dichk3ro-5-(1-trif!uoromethy!-vinyi)-benzeπe (8.4 g, example 1 step A) and iriethySamine (5-4 ml) are added. The reaction is then further reacted at room temperature for 3 hours. Water is added (50 mi) and the reaction mixture is extracted with ethyl acetate. The organic phase is washed with a saturated aqueous soiution of NaCi, dried over MgSO4 and concentrated in vacuo. The crude product is dissolved in dicNorometnane and filtered over a pad of silica gei to yield 5~{3,5-dichlQrα-pheπyl}~3-(4- fiυoro-naphthafeπ-1-y!)-5-trif!uoromethyl-4.5-dihydro~isoxa£θle as a beige solid.
Step C: Sodium azide (3.3 g) is aόdeά to a soiution otδ-(3,5~dichioro~phenyi)-3-(4-fiuoro~ πaphihaieπ-1"yi}-5-trifiuorornethyi»4,5-d3hydro-isoxa2θle (10.7 g) in OUF (50 ml). After 20 hours at 80δC another portion of sodium azide is added (3.3 g) and the reaction mixture is stirred for 20 hours at 8O0C. The soiution is cooiβd to room temperature and the volume concentrated to 10 mi in vacuo. Water anά ethyl acetate am added and the precipitate is collected after filtration to yield 3-{4~azido-πaphthaien-1-yl)»5-{3l5-dichioro-ph8nyi)-5- trif!uoromethy!~4,5-dibydro~isQxazofe as a beige soHd: m.p, 183-185 0C.
SMβ»Sl Cut (211 mg) is added to a soiution of 3~(4~azido-naphthalen-1-yl)-5-{3,5-dichioro- phenyi>-54rifluoromethyi-4f5-dihydro-isoxazoie (500 mg), DlPEA (3.3 rni) and Λ/-pfop~2~ynyi~ isonicotiπamide (1SO rπg) in DMF (2.0 ml). After 5 hours at room temperature, the reaction is quenched with a saturated solution of NaHCO3 and extracted with dschbromethane. The combined organic phases are dried ovef Na2SO4 and concentrated in vacuo. The crude product is purified on a semi-preparative HPLC to yield A/-(1~{4~[5-(3,5-dsch!oro«phenyi)~5~ trifluoromethyi-4,5-dfhydro-isoxazo!-3-yl]-naphthalen-1 ~y!}-1 W-[1 ,2,3|tria2øi-4-yimethy1)- lsonicoiinamide as a coioriess oil.
Tabie 1 provides further compounds of formula
wherein the meaning of Q is given Tabie 1. The substances are prepared analogously to the above-described methods. The following physical data are obtained according to the above- described HPLC/MS characterization process. The values of the meiting point are indicated in "C. Table 1:
I- :.. Activity jn ylt/o against Gtenoceøhalidβsfβlis XCatJjea).
A mixed adult population of fleas is placed in a suitably formatted 96-well plate allowing fleas to access and feed on treated biood via an artificial feeding system. Fleas are fed on treated blood for 24 hours, after which the compound's effect is recorded, insecticidal acliviiy is determined on the basis of the number of dead ffeas recovered from the feeding system- Compound 1 ,1, 1.2, 1.5. 1.6, 1.8, 1.10, 1.12, 1.14, 1.17, 1.18, 1.23-1.25, 1.27-1.30, 1.32- 1.35, 1.37-1.39, 1.41 , 1.45, 1 ,47-1 ,50, 1.55, 1.56, 1.58-1.64, 1.87, 1.69-1.74 showed more than 80% (EC80) efficacy at
Z..AcMto..in.vitro against Rhipic&βhalυs sanguineus (Dog tick),
A clean aduit teck population is used to seed a suitably formatted 9β~weii plate containing the test substances to be evaluated for antiparasitic activity. Each compound is tested by serial άllutlon in order to determine its minimal affective dose (MED). Ticks ate left in contact with the test compound for 10 minutes and are then incubated at 28"C and 80% relative humidity for 7 days, during which the test compound's effect is monitored. Acaricidai activity is confirmed if adult ticks are dead.
In this test the following examples showed more than 80% (ECβo) efficacy at 640pprn: 1.1,
1.5, 1.6, 114, 1.17, 1.18, 1.27-1.30, 1.33-1.35, 1.45, 1.55. 1.56, 1.59-1.61, 1.83, 1.74.
3. Activity in yivo BQdΛnsϊβhipioβphaius sanguineus nymphs on MongoJian αerbils (M&riones
On day 0, gerbiSs are treated with the test compound at a given dose by spray application. On day +1 , the animals &ΓB infested with nymphs of R sanguineus. Ticks are left on the
animate until fυii repletion. Seven days after infestation nymphs dropped off fully engorged are collected and counted. Efficacy in killing is expressed as a tick number reduction in comparison with a placebo treated group, using the Abbot's formula.
Results of some compounds from Tabte 1 are shown in Tabiδ 2
Claims
1. A compound of formula
Ai, A2, A3, A4, A5 and A6 are each independently selected from lhe group consisting of CR3" and N; π is an integer from 0 to 4;
8!4 B2 and Bs are each independently selected from the group consisting of CRa' and N; each R2 1 is independently of the other H or R2; each R3' is independently of the other H or R3;
R, is CrCe-alkyl, QrCβ-aikβnyl, QrCValkynyi, GyCVeycioalkyi, C4-C7-alky!cyc!oaikyi or d-
CrcycioalkyiaiKyi, each uπsubstituted or substituted with one or more subststuents independently selected from RA) each R2 is independently halogen, d-Ce-alkyl, CrGs-ha!oalkyl, CrQj-alkoxy, CrQ- haloalkoxy, CrCβ-alkylthio, CrQ-haloaikylthio, d-Ce-aikylsuffinyl, CrCe-haioalkyisu!finyi,
Ci-Ce-aikylsuifonyi, CrC6-haloafkylsulfonyl, N-moπo- or N,N-di-CrCra!ky]amfno, C2-C6 alkoxycarbonyi, cyano (-CN) or nitro {-NO2}; each R3 is independently halogen, CrC6-alkyS, Ct-Qrhalαaiky!, C3~Cs~cycioalkyi, C3-C6- halocycioalkyi, C^Ce-alkoxy, CrC6-haioaikoxy, CrCβ-a[kytthio, Ci-Ce-haioaikylthio, CrC6 a!kyi-$uifinylτ CrCe-ha!oaikyisuifinyl, Ci-Cs-aikylsυifonyi, CrCg-haioaikyisuifonyi, amino,
N-mono- or N.N-di-CrCe-aJkylamino, CrCe-aikoxycarbonyl, -CN or -NO2;
R4 is halogen, CrC6-alkyl( CrCδ-aIkoxy, Ci-Craikyithiot CrCe-aikyisulfinyi, C-Cs-aikyi- sυlfonyi, -CN or -NO2;
Q is a 5- or 8~roembered heterocyclic ring, or a Cβ-Cio-carbocyciic ring system or a 8-, 9- or
10-mernbered fused hetero-bicyciic ring system, each of them being unsυbstituted or substituted with one or more substituents independently selected from halogen, d-Ce-alkyl,
CrQrhaioaikyS, CrCe-cycioalkyi, C3-C6-haiocyc!oalkyf( CrCe-aikoxy, CrCβ-cyctoalkoxy, Cr
Cs-ha!oaikoxy, C2-C6-aikenyi, C2"C6 «haloalkenyi, CrC6-alkyithto, CrCθ-haloaikyithk}, C1-C6- aSkyisuifinyi, CrC6-haioaikytsu!finyi, CrCδ-afkyisutfony!t CrCs-hafoalkylsuifonyi, -CN, -NO2, amino, N-mono- or N.N-di-CrCs-sikyiamino, COaH, Cj~C5~alkoxycarbonyi, sulfonamide, N-
monø- or HM, di~CrCδ-aikyisu!fonamido, CONH2, N-mono- or H,U> di~CrCβ-aikyiamino~ carfoonyi, N-moπo- or NSN, di-CrCs-haio^lkyfarninocarbony!, N-CrCrhaioalkyiamino~ caroonyi-CiCralkyiarninocarbony!, CrCβ-alkyfcarbonyiamino, QrCVaikanoyl, C2-Cs-haio- alkanoyi, CH2R5 , a group CCO}N*CHN{CH3}a, a group C(O)N=CHNHOCH3, pyrroSidonyi- carbonyl, anό unsubstituted or haiogen-, Cf-C6-alky1~, CrC6~ha!øaikyh Ci-Cδ-afkox>s C1-C6- haSoalkcxy-, πitro- or eyano-substituted phenyl, benzyl, phenethyl, pyridyi. pyridinylmethyl, pyrimjdinyi, pyrimidlnyimethyl, benzoyl or pheπoxy; and
R5 is CO2H1 Ct-Cfj-alkoxycarboπyf, CONH2, N-rnoπo- or N,N, di-CrCβ-aikylaminocarbonyl, N-mono- or N,N, di-CrCs-haloaikylamiπocarbonyl, CrCs-aikyicarbonyiamino, pyridiπyiaminocarbortyl or tetrahydrofuranyl.
2. A compound of formula (i) according to claim 1 , wherein
Af, A;, A3, A*, A5 and A$ are each independently selected from the group consisting of CR3'
n is Bn integer from O to 4;
Bi, B2 and B3 are each independently selected from the group consisting of CR2 ' snό N; each R2' is independently of the other H or R2; each R3' is independently of the other H or R3;
R1 is d-Ce-aikyf, QrtV-aikeny!, C2-Cδ-aikyny1, C3-C6-eydoa!kyl< Cj-Cy-aikyicyciøaikyf or C4-
C?-cycloalkyialky), each unsubsiituted or substituted with one or more substituents independently selected from R*; each R2 is independently hafogen, CrCβ-alkyi, CrC3-haioalkyi, CrCg~aikoxy, C1-C6- haloalkoxy, CrCδ-a!kyithio, CrCβ-haloalkylthio, CrCδ-aikyisuffinyi, Ci-C8-πafoalkylsulfϊny!,
CrCδ-aikyisulfony1, C^Cβ-haloaikylsuifonyi, N-mono- or N.N-di-Gi-CVaikylamino, C2-C6- aikoxycarbonyi, cyano (-CN) or nitro (-NO2); each R3 is independently haiogen, C,-Cδ-aiky1( CrCe~ha!oa!kyl, C3-C3-cycioaiky{, C3-C9- haiocycloalkyl, CrC6-a!koxy, CrQrftaloaikoxy, CrCe-alkylthio, CrCe-haioaikyithio, C1-C6 aikyi-sυlfinyi, CrCs-haloaikylsulfinyl, CrCg-aikylsuifonyl, CrCe-haioalkyisulfonyi, at^ino,
N-rnono~ or KN-di-CrQ-alkylamino, CrCralkoxycarbonyi, -CN or -NO2;
R4 is halogen, CrC6~alkyf, d-Cβ-alkoxy, CrCδ~alkyithio, CrCδ-a!kylsu!finy!, CrC6-afkyl- suifonyl, -CN or -NO2; &nd
Q is a 5- or 8-memfaβred heterocyclic ring, or a Ce-Cw-carhooyciie ring system or a 8-, 9- or
10-membered fused hetero-bicydic ring system, each of them being unsubstituted or substituted with oαβ or more substituents independently selected from halogen, CrCe-slkyi
CrCδ~halostikyl, C3-C6-cyc!oaikyi C3<Vna!ocyclQalkyl, CrCδ-alkoxy, Ci-Ce-haloaikoxy, Cr C6-alkyithio. CrCs-haioaikyithio, d-CVaikyisuifiny), CrCδ~ha[oalkyfsuifinyi, Ci-C8- alkyisujfonyj, CrCe-haloalky!suifonyt -CN, -NO2, amino, N-moπo- or N,N~di-CrCδ- alkyiamino, CrCe-alkoxycarbonyS, sulfonamide, N-rnono or N1N, di-C-Cra&yϊsulfonamido, N~mono- or N.N-di-d-Ce-alkyicarbonyiamino, CrCδ-alkanøyi and unsubstituted or halogen-, CrCβ-alkyi-, CrCe-haioaikyi-, CrC3-aIkøxy~, C1-C6-Ha(OaIkOXy-, nitro- or cya no-substituted phenyi, benzyl, benzoyl or pheπoxy.
3. A compound according to claim 1 or 2 of formula (i), wherein Bn S2 and B3 are each CR2 ".
4. A compound according to any one of claims 1 to 3, wherein A1, A2. A3, A4, A5 and Ag are each CH.
5. A compound according to a?iy one of claims 1 to 4, wherein Rt is CrQrhatoalkyi, in particular CF3.
6. A compound according to ciaim 1 of formula
7. A compound according anyone of claims 1 to 6, wherein Q is a δ- or δ-merobereel heterocyclic ring having from 1 to 4, preferably from 1 io 3 same or different heieroatoms selected from the group consisting of N1 O and S, which is further unsubstituted or substituted by one or more sυbstituents selected from the group consisting of Ci-C4-alkyJ, Crαrhaioaikyl, CrCj-aikoxy, C^-haloaikoxy, CrC4~aikylthiGi CrCrhaloalkyHhio, Ci-Gr alkylsυifinyi, CrC^haioalkylsuirlnyi, CrC,ralkylsuifonyi, Ci-Crhafoaikytsuifonyi, -CN, -NO2, Ct-Cralkoxycarbonyi, sulfonamide, N-mono- or N.N-di-€rC^aikylcarbonyiarnino, C2-C3- alkanoy! and υnsubstttuted or halogen- or CrC4~a!kyi-substituted phenyl, benzyl, benzoyl and phenoxy.
3. A compound according to anyone of claims 1 to ?, wherein Q is a radical of formula
9. A compound according to claim 8, wherein Q is the radical Q-34, Q-43 or Q-47, wherein r is in each case 0.
10. A compound of formuia (Ia) according to claim 6, wherein n is an integer from 1 to 3, Ri is CrCyhaloalkyi, each R2 and R3 is independently selected from the group consisting of halogen, d-Cz-aikyl, CrC2~hatoalky!, CrCralkoxy, CrG2-ha!o- aikoxy, CrC2-hafoaikyithio( -CN and -NO2, m is an integer from 0 to 2, m particular 0, and Q is either
(i) phenyl, which is unsubstltuted or substituted by 1 to 3, in particular 1 or 2, same or different subsϋtuenis selected from the group consisting of halogen, CrCj-alkyl, CrC2- haioaikyi, Ci-Qrβlkoxy, Ci-C2-haloaikoxy, CrC2-haioaikyithio, -CN, -NO2, and unsubsiituted or halogen-, CrCVaJkyh CrC2-haioalky!-, C1 -C2^aIkOXy-, CrCr-haioalkoxy-, -CN and
~NO2-substituted phenyl or phenoxy, or is
(H) a 5- or 8-mβmbβred heterocyclic ring having from 1 to 3 same or different heteroaioms selected from the group consisting of N, O anά S, which is further unsubstiiuted or substituted by halogen, CrCz-aikyi, CrCrhaloaikyl, CrC2-alkoxy, d-Crhaloaikoxy, CrC2- haioalkyfthio, -CN, -NG2 or CrC^a ikoxycarbonyl, or is
(its) a fused hetero-bicyclic ring system comprising a 5» or δ-membβred heterocyclic ring having from 1 to 3 same or different heteroatoms selected from the group consisting of M1 O and S1 io which is attached an anπuiated ring, and which is further υnsubstitυted or substituted by halogen, CrQralkyl, CrC2-haioaikyl, CrC2-a!koxy, CrC2-haioa{koxy, C1-C2- haioalkylthio, -CN, -NO2 or CrC^aikoxycarbonyi
11. A compound of formula (fa) according to claim 8, wherein n is an integer from 1 to 3, Ri is CF3, each R2 is independently selected from the group consisting of haiogen, CrC2-hak>alkyi, C-Ca-haioafkoxy and -CN, m is 0 and Q is either
(ii) a 5- or 6-membered heteroarømatic ring seiected from the group consisting of the radicals given in claim 7, or is
(iii) a fused hetero-bJcyclic ring system selected from the group consisting of the radicals
12. A compound of formula (Ia) according to claim β, wherein n is an integer from 1 to 3, Ri is CF3, each R2 is independently seiected from the group consisting of halogen, CrC2-haloa!kyi( CrC2-haioaikoxy and -CH, m is 0 and Q is a radical
iπ particular a radical Q-24, Q-28, G-30, Q-32, Q-34, Q-44 or Q-47, wherein r Is an integer of 0 or 1 , and R is independently selected from the group consisting of CrC2-aikylt halo- CrC2- aikyl, halo- CrC2-alkylcarbonyl( cyaπo, Ct-Cralkoxycarbonyi, CrC^aikoxycarbonyimethy!, carboxy-CrCralkyi, aminocarbonyl, N-mono- or N.N-di-CrCa-afkyiaminocarboπyl, halo- C1- Ca-alkyfaminocarbonyirnethyl, halo-CrCraikylaminorørbønylmethylaminocarbonyi, hakwinyi , »C(O)N=CHN(CH3)2( -C(O)N-CHNHOCH3, unsubstltuted or halogen- or CrC2~aikoxy- substituted phenyl, benzy! and pfienylβthyl, unsubstitυtβd anά halogen-substituted 2-, 3- arxi 4-ρyridyl snύ pyridylmethyl, pyridylcarbonylarninomethy!, pyrrolidsnecarbonyi, and 2- tθtrahydrofuranyimβthyi.
13. Composition for the control of parasites, comprising as active ingredient at ieast one compound of the formula (1 ) according to any one of claims 1 to 12, in addition to a carrier and/or a dispersant
14. Method of controiiing parasites in and on warm-blooded animals, which comprises applying to the animals a pharmaceutical effective amount of at ieast one compound of formula {1} according to any one of claims 1 to 12.
15. Use of a compound of formula (1 } according to any one of claims 1 to 12 in the control sit
16. Use of a compound of formula (1) according to any one of claims 1 to 12 in a process for controiiing parasites in and on warm-blooded animals.
17. Use of a compound of formula (1 ) according to any one of claims 1 to 12 in the preparation of a pharmaceutical composition against parasites in &nά on warm-blooded animals.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09793526.6A EP2379544B1 (en) | 2008-12-18 | 2009-12-17 | Isoxazolines derivatives and their use as pesticide |
| US13/133,432 US8377942B2 (en) | 2008-12-18 | 2009-12-17 | Isoxazolines derivatives and their use as pesticide |
| CA2747060A CA2747060A1 (en) | 2008-12-18 | 2009-12-17 | Isoxazolines derivatives and their use as pesticide |
| ES09793526.6T ES2442342T3 (en) | 2008-12-18 | 2009-12-17 | Isoxazoline derivatives and their use as a pesticide |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP08172156 | 2008-12-18 | ||
| EP08172156.5 | 2008-12-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010079077A1 true WO2010079077A1 (en) | 2010-07-15 |
Family
ID=40647080
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/067437 WO2010079077A1 (en) | 2008-12-18 | 2009-12-17 | Isoxazolines derivatives and their use as pesticide |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8377942B2 (en) |
| EP (1) | EP2379544B1 (en) |
| CA (1) | CA2747060A1 (en) |
| CL (1) | CL2011001389A1 (en) |
| ES (1) | ES2442342T3 (en) |
| WO (1) | WO2010079077A1 (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011075591A1 (en) | 2009-12-17 | 2011-06-23 | Merial Limited | Anti parasitic dihydroazole compounds and compositions comprising same |
| CN102219753A (en) * | 2011-04-21 | 2011-10-19 | 山东大学 | Triazole compounds as well as preparation method and application thereof |
| WO2011157733A3 (en) * | 2010-06-18 | 2012-04-19 | Novartis Ag | Use of substituted heterocyclic compounds to control sea lice on fish |
| WO2012089623A1 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| WO2012089622A2 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation |
| WO2012158396A1 (en) * | 2011-05-19 | 2012-11-22 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| US8415310B2 (en) | 2010-08-05 | 2013-04-09 | Ah Usa 42 Llc | Isoxazoline derivatives as antiparasitic agents |
| WO2013150052A1 (en) | 2012-04-04 | 2013-10-10 | Intervet International B.V. | Soft chewable pharmaceutical products |
| US8686014B2 (en) | 2008-09-04 | 2014-04-01 | Syngenta Limited | Insecticidal compounds |
| JP2014510095A (en) * | 2011-03-22 | 2014-04-24 | ゾエティス・エルエルシー | Isoxazoline derivatives as antiparasitic agents |
| US8735362B2 (en) | 2009-12-01 | 2014-05-27 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| US8822502B2 (en) | 2008-08-22 | 2014-09-02 | Syngenta Crop Protection Llc | Insecticidal compounds |
| EP2585443B1 (en) * | 2010-06-23 | 2015-02-18 | Basf Se | Process for producing imine compounds for combating invertebrate pests |
| WO2015066277A1 (en) | 2013-11-01 | 2015-05-07 | Merial Limited | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015086551A1 (en) | 2013-12-10 | 2015-06-18 | Intervet International B.V. | Antiparasitic use of isoxazoline compounds |
| WO2015091900A1 (en) | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| GB2523811A (en) * | 2014-03-06 | 2015-09-09 | Norbrook Lab Ltd | Novel Isoxazolines and their uses |
| US9180121B2 (en) | 2011-09-12 | 2015-11-10 | Merial, Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US9216973B2 (en) | 2008-08-22 | 2015-12-22 | Syngenta Participations Ag | Insecticidal compounds |
| US9233100B2 (en) | 2012-02-06 | 2016-01-12 | Merial, Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| CN106366018A (en) * | 2016-08-29 | 2017-02-01 | 上海雅本化学有限公司 | Synthesis method of 4-bromonaphthalene-1-carbonitrile |
| WO2019091940A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Process for preparing large size isoxazoline particles |
| WO2019091936A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Injectable isoxazoline pharmaceutical compositions and their use against parasite infestation |
| WO2019115492A1 (en) | 2017-12-12 | 2019-06-20 | Intervet International B.V. | Implantable isoxazoline pharmaceutical compositions and uses thereof |
| WO2019122324A1 (en) | 2017-12-21 | 2019-06-27 | Intervet International B.V. | Antiparasitic pour-on compositions |
| US10456358B2 (en) | 2013-12-20 | 2019-10-29 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| US10799483B2 (en) | 2014-12-22 | 2020-10-13 | Intervet Inc. | Use of isoxazoline compounds for treating demodicosis |
| WO2020225143A1 (en) | 2019-05-03 | 2020-11-12 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| WO2021013825A1 (en) | 2019-07-22 | 2021-01-28 | Intervet International B.V. | Soft chewable veterinary dosage form |
| US10973759B2 (en) | 2012-11-20 | 2021-04-13 | Intervet Inc. | Manufacturing of semi-plastic pharmaceutical dosage units |
| WO2021122515A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Parasite control in ruminants |
| WO2021122513A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control |
| WO2021122521A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control in poultry |
| US11179372B2 (en) | 2015-06-23 | 2021-11-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| WO2021233967A1 (en) | 2020-05-20 | 2021-11-25 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| WO2024047241A1 (en) | 2022-09-02 | 2024-03-07 | Intervet International B.V. | Treatment of fasciolosis |
Families Citing this family (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JO3626B1 (en) | 2012-02-23 | 2020-08-27 | Merial Inc | Topical compositions comprising fipronil and permethrin and methods of use |
| MX363173B (en) * | 2012-12-12 | 2019-03-12 | Zoetis Services Llc | Phenylpyrazole injectable compositions. |
| AR094112A1 (en) * | 2012-12-20 | 2015-07-08 | Novartis Tiergesundheit Ag | (HETERO) USEFUL ARILACRILAMIDS TO CONTROL ECTOPARASITOS |
| WO2015161224A1 (en) | 2014-04-17 | 2015-10-22 | Merial, Inc. | Use of malononitrile compounds for protecting animals from parasites |
| UY36570A (en) | 2015-02-26 | 2016-10-31 | Merial Inc | INJECTABLE FORMULATIONS OF PROLONGED ACTION THAT INCLUDE AN ISOXAZOLINE ACTIVE AGENT, METHODS AND USES OF THE SAME |
| SG11201708068PA (en) * | 2015-04-08 | 2017-10-30 | Merial Inc | Extended release injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| AR104714A1 (en) | 2015-05-20 | 2017-08-09 | Merial Inc | DEPSIPEPTIDE COMPOUNDS AS ANTIHELMINICS |
| UY37137A (en) | 2016-02-24 | 2017-09-29 | Merial Inc | ANTIPARASITARY COMPOUNDS OF ISOXAZOLINE, INJECTABLE FORMULATIONS OF PROLONGED ACTION THAT INCLUDE THEM, METHODS AND USES OF THE SAME |
| CA3023675C (en) | 2016-05-10 | 2021-03-30 | Elanco Tiergesundheit Ag | Dihydroisoxazole compound for use in controlling sea lice |
| BR112019007605A2 (en) | 2016-10-14 | 2019-09-17 | Boehringer Ingelheim Animal Health Usa Inc | pesticide and parasiticide compounds of vinyl isoxazoline |
| CA3044038A1 (en) | 2016-11-16 | 2018-05-24 | Boehringer Ingelheim Animal Health USA Inc. | Anthelmintic depsipeptide compounds |
| EP3668866B1 (en) | 2017-08-14 | 2023-01-18 | Boehringer Ingelheim Animal Health USA Inc. | Pesticidal and parasiticidal pyrazole-isoxazoline compounds |
| WO2020014068A1 (en) | 2018-07-09 | 2020-01-16 | Boehringer Ingelheim Animal Health USA Inc. | Anthelminthic heterocyclic compounds |
| US11773066B2 (en) | 2018-11-20 | 2023-10-03 | Boehringer Ingelheim Animal Health USA Inc. | Indazolylcyanoethylamino compound, compositions of same, method of making, and methods of using thereof |
| CN113825543A (en) | 2019-03-19 | 2021-12-21 | 勃林格殷格翰动物保健美国公司 | Anthelmintic aza-benzothiophene and aza-benzofuran compounds |
| MX2022015038A (en) | 2020-05-29 | 2023-01-04 | Boehringer Ingelheim Animal Health Usa Inc | Anthelmintic heterocyclic compounds. |
| BR112023015168A2 (en) | 2021-01-27 | 2023-10-03 | Corteva Agriscience Llc | CYCLOPROPYLAMIDE COMPOUNDS AGAINST PARASITES IN FISH |
| US20240116854A1 (en) | 2021-01-27 | 2024-04-11 | Intervet Inc. | Cyclopropylamide compounds against parasites in fish |
| US20230312207A1 (en) | 2022-02-17 | 2023-10-05 | Boehringer Ingelheim Vetmedica Gmbh | Method and system for providing a fluid product mailer |
| CN115073454B (en) * | 2022-08-09 | 2024-03-08 | 贵州大学 | Imidazo pyridine-2-oxazoline compound and preparation method and application thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007075459A2 (en) * | 2005-12-16 | 2007-07-05 | E. I. Du Pont De Nemours And Company | 5-aryl isoxazolines for controlling invertebrate pests |
| WO2007079162A1 (en) * | 2005-12-30 | 2007-07-12 | E. I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| WO2008154528A2 (en) * | 2007-06-13 | 2008-12-18 | E. I. Du Pont De Nemours And Company | Isoxazoline insecticides |
-
2009
- 2009-12-17 CA CA2747060A patent/CA2747060A1/en not_active Abandoned
- 2009-12-17 EP EP09793526.6A patent/EP2379544B1/en not_active Not-in-force
- 2009-12-17 ES ES09793526.6T patent/ES2442342T3/en active Active
- 2009-12-17 US US13/133,432 patent/US8377942B2/en not_active Expired - Fee Related
- 2009-12-17 WO PCT/EP2009/067437 patent/WO2010079077A1/en active Application Filing
-
2011
- 2011-06-08 CL CL2011001389A patent/CL2011001389A1/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007075459A2 (en) * | 2005-12-16 | 2007-07-05 | E. I. Du Pont De Nemours And Company | 5-aryl isoxazolines for controlling invertebrate pests |
| WO2007079162A1 (en) * | 2005-12-30 | 2007-07-12 | E. I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| WO2008154528A2 (en) * | 2007-06-13 | 2008-12-18 | E. I. Du Pont De Nemours And Company | Isoxazoline insecticides |
Cited By (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9216973B2 (en) | 2008-08-22 | 2015-12-22 | Syngenta Participations Ag | Insecticidal compounds |
| US8822502B2 (en) | 2008-08-22 | 2014-09-02 | Syngenta Crop Protection Llc | Insecticidal compounds |
| US8686014B2 (en) | 2008-09-04 | 2014-04-01 | Syngenta Limited | Insecticidal compounds |
| US9282742B2 (en) | 2008-09-04 | 2016-03-15 | Syngenta Participations Ag | Insecticidal compounds |
| US9609869B2 (en) | 2009-12-01 | 2017-04-04 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| US11357231B2 (en) | 2009-12-01 | 2022-06-14 | Syngenta Crop Protection Llc | Insecticidal compounds based on isoxazoline derivatives |
| US10750745B2 (en) | 2009-12-01 | 2020-08-25 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| US10206400B2 (en) | 2009-12-01 | 2019-02-19 | Syngenta Participations Ag | Insecticidal compounds based on isoxazoline derivatives |
| US8735362B2 (en) | 2009-12-01 | 2014-05-27 | Syngenta Crop Protection, Llc | Insecticidal compounds based on isoxazoline derivatives |
| WO2011075591A1 (en) | 2009-12-17 | 2011-06-23 | Merial Limited | Anti parasitic dihydroazole compounds and compositions comprising same |
| US9776999B2 (en) | 2009-12-17 | 2017-10-03 | Merial Inc. | Antiparisitic dihydroazole compounds and compositions comprising same |
| US9376434B2 (en) | 2009-12-17 | 2016-06-28 | Merial Inc. | Antiparisitic dihydroazole compounds and compositions comprising same |
| US8618126B2 (en) | 2009-12-17 | 2013-12-31 | Merial Limited | Antiparisitic dihydroazole compounds and compositions comprising same |
| EP3078664A1 (en) | 2009-12-17 | 2016-10-12 | Merial Inc. | Antiparasitic dihydroazole compositions |
| EP3560923A1 (en) | 2009-12-17 | 2019-10-30 | Boehringer Ingelheim Animal Health USA Inc. | Anti parasitic dihydroazole compounds and compositions comprising same |
| WO2011157733A3 (en) * | 2010-06-18 | 2012-04-19 | Novartis Ag | Use of substituted heterocyclic compounds to control sea lice on fish |
| US9023836B2 (en) | 2010-06-18 | 2015-05-05 | Novartis Tiergesundheit Ag | Use of substituted heterocyclic compounds to control sea lice on fish |
| EP2585443B1 (en) * | 2010-06-23 | 2015-02-18 | Basf Se | Process for producing imine compounds for combating invertebrate pests |
| US8415310B2 (en) | 2010-08-05 | 2013-04-09 | Ah Usa 42 Llc | Isoxazoline derivatives as antiparasitic agents |
| US10864195B2 (en) | 2010-12-27 | 2020-12-15 | Intervet Inc. | Topical localized isoxazoline formulation |
| WO2012089622A2 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation |
| WO2012089623A1 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| EP2658542B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation |
| US9532978B2 (en) | 2010-12-27 | 2017-01-03 | Intervet Inc. | Topical localized isoxazoline formulation |
| US9173870B2 (en) | 2010-12-27 | 2015-11-03 | Intervet Inc. | Topical localized isoxazoline formulation |
| EP2658541B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| JP2014510095A (en) * | 2011-03-22 | 2014-04-24 | ゾエティス・エルエルシー | Isoxazoline derivatives as antiparasitic agents |
| CN102219753A (en) * | 2011-04-21 | 2011-10-19 | 山东大学 | Triazole compounds as well as preparation method and application thereof |
| US9255111B2 (en) | 2011-05-19 | 2016-02-09 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| EP2918583A1 (en) * | 2011-05-19 | 2015-09-16 | Eli Lilly And Co. | Parasiticidal dihydroisoxazole compounds |
| US8889710B2 (en) | 2011-05-19 | 2014-11-18 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| WO2012158396A1 (en) * | 2011-05-19 | 2012-11-22 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| CN103547576B (en) * | 2011-05-19 | 2016-06-08 | 伊莱利利公司 | Parasiticidal dihydro isoxazole compound |
| JP2014515347A (en) * | 2011-05-19 | 2014-06-30 | イーライ リリー アンド カンパニー | Dihydroisoxazole compounds for parasite control |
| EA023711B1 (en) * | 2011-05-19 | 2016-07-29 | Эли Лилли Энд Компани | Parasiticidal compounds, methods and formulations |
| US8648081B2 (en) | 2011-05-19 | 2014-02-11 | Eli Lilly And Company | Parasiticidal dihydroisoxazole compounds |
| CN103547576A (en) * | 2011-05-19 | 2014-01-29 | 伊莱利利公司 | Parasiticidal dihydroisoxazole compounds |
| US11464763B2 (en) | 2011-09-12 | 2022-10-11 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US10383854B2 (en) | 2011-09-12 | 2019-08-20 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US9877950B2 (en) | 2011-09-12 | 2018-01-30 | Merial Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US9180121B2 (en) | 2011-09-12 | 2015-11-10 | Merial, Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US10786487B2 (en) | 2011-09-12 | 2020-09-29 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US9931320B2 (en) | 2012-02-06 | 2018-04-03 | Merial Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| US10596156B2 (en) | 2012-02-06 | 2020-03-24 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| US9259417B2 (en) | 2012-02-06 | 2016-02-16 | Merial, Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| US9233100B2 (en) | 2012-02-06 | 2016-01-12 | Merial, Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| WO2013150055A1 (en) | 2012-04-04 | 2013-10-10 | Intervet International B.V. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| US11712416B2 (en) | 2012-04-04 | 2023-08-01 | Intervet Inc. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| US11285101B2 (en) | 2012-04-04 | 2022-03-29 | Intervet Inc. | Soft chewable pharmaceutical products |
| WO2013150052A1 (en) | 2012-04-04 | 2013-10-10 | Intervet International B.V. | Soft chewable pharmaceutical products |
| US11337917B2 (en) | 2012-04-04 | 2022-05-24 | Intervet Inc. | Soft chewable pharmaceutical products |
| US9770440B2 (en) | 2012-04-04 | 2017-09-26 | Intervet Inc. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| EP3964198A1 (en) | 2012-11-20 | 2022-03-09 | Intervet International B.V. | Manufacturing of semi-plastic pharmaceutical dosage units |
| US11911503B2 (en) | 2012-11-20 | 2024-02-27 | Intervet Inc. | Manufacturing of semi-plastic pharmaceutical dosage units |
| US10973759B2 (en) | 2012-11-20 | 2021-04-13 | Intervet Inc. | Manufacturing of semi-plastic pharmaceutical dosage units |
| WO2015066277A1 (en) | 2013-11-01 | 2015-05-07 | Merial Limited | Antiparisitic and pesticidal isoxazoline compounds |
| EP3733664A1 (en) | 2013-11-01 | 2020-11-04 | Boehringer Ingelheim Animal Health USA Inc. | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015086551A1 (en) | 2013-12-10 | 2015-06-18 | Intervet International B.V. | Antiparasitic use of isoxazoline compounds |
| US9655884B2 (en) | 2013-12-10 | 2017-05-23 | Intervet Inc. | Antiparasitic use of isoxazoline compounds |
| EP4306168A2 (en) | 2013-12-20 | 2024-01-17 | Intervet International B.V. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| WO2015091900A1 (en) | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| US11883530B2 (en) | 2013-12-20 | 2024-01-30 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| US10456358B2 (en) | 2013-12-20 | 2019-10-29 | Intervet Inc. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| US10272071B2 (en) | 2013-12-20 | 2019-04-30 | Intervet Inc. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| US11304934B2 (en) | 2013-12-20 | 2022-04-19 | Intervet Inc. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| US10653675B2 (en) | 2013-12-20 | 2020-05-19 | Intervet Inc. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| EP3878281A1 (en) | 2013-12-20 | 2021-09-15 | Intervet International B.V. | Use of isoxazoline compounds in poultry |
| GB2523811A (en) * | 2014-03-06 | 2015-09-09 | Norbrook Lab Ltd | Novel Isoxazolines and their uses |
| EP4008329A1 (en) | 2014-12-22 | 2022-06-08 | Intervet International B.V. | Isoxazoline compounds for use in treating demodicosis |
| US10799483B2 (en) | 2014-12-22 | 2020-10-13 | Intervet Inc. | Use of isoxazoline compounds for treating demodicosis |
| US11179372B2 (en) | 2015-06-23 | 2021-11-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| US11878006B2 (en) | 2015-06-23 | 2024-01-23 | Intervet Inc. | Isoxazoline solution containing vitamin E for use with sanitized drinking water |
| CN106366018A (en) * | 2016-08-29 | 2017-02-01 | 上海雅本化学有限公司 | Synthesis method of 4-bromonaphthalene-1-carbonitrile |
| WO2019091940A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Process for preparing large size isoxazoline particles |
| WO2019091936A1 (en) | 2017-11-07 | 2019-05-16 | Intervet International B.V. | Injectable isoxazoline pharmaceutical compositions and their use against parasite infestation |
| EP4316522A2 (en) | 2017-11-07 | 2024-02-07 | Intervet International B.V. | Injectable isoxazoline pharmaceutical compositions and their use against parasite infestation |
| US11858904B2 (en) | 2017-11-07 | 2024-01-02 | Intervet Inc. | Process for preparing large size isoxazoline particles |
| WO2019115492A1 (en) | 2017-12-12 | 2019-06-20 | Intervet International B.V. | Implantable isoxazoline pharmaceutical compositions and uses thereof |
| US11648238B2 (en) | 2017-12-12 | 2023-05-16 | Intervet Inc. | Implantable isoxazoline pharmaceutical compositions and uses thereof |
| WO2019122324A1 (en) | 2017-12-21 | 2019-06-27 | Intervet International B.V. | Antiparasitic pour-on compositions |
| WO2020225143A1 (en) | 2019-05-03 | 2020-11-12 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| WO2021013825A1 (en) | 2019-07-22 | 2021-01-28 | Intervet International B.V. | Soft chewable veterinary dosage form |
| WO2021122515A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Parasite control in ruminants |
| WO2021122513A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control |
| WO2021122521A1 (en) | 2019-12-16 | 2021-06-24 | Intervet International B.V. | Composition for lice control in poultry |
| WO2021233967A1 (en) | 2020-05-20 | 2021-11-25 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| WO2024047241A1 (en) | 2022-09-02 | 2024-03-07 | Intervet International B.V. | Treatment of fasciolosis |
Also Published As
| Publication number | Publication date |
|---|---|
| US8377942B2 (en) | 2013-02-19 |
| ES2442342T3 (en) | 2014-02-11 |
| CA2747060A1 (en) | 2010-07-15 |
| US20110245274A1 (en) | 2011-10-06 |
| EP2379544A1 (en) | 2011-10-26 |
| EP2379544B1 (en) | 2013-10-16 |
| CL2011001389A1 (en) | 2011-09-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2379544A1 (en) | Isoxazolines derivatives and their use as pesticide | |
| DK2379537T5 (en) | Isoxazoline derivatives and their use as pesticides | |
| AU2012215440B2 (en) | Isoxazoline derivatives for controlling invertebrate pests | |
| AU2012224521B2 (en) | Isoxazole derivatives | |
| EP2582696B1 (en) | 5-aryl isoxazolines for controlling pests | |
| ES2528368T3 (en) | 5-aryl isoxazolines to control pests | |
| NZ613191B2 (en) | Isoxazoline derivatives for controlling invertebrate pests |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09793526 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009793526 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13133432 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2747060 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |