WO2010074747A1 - Stable insecticide compositions and methods for producing same - Google Patents
Stable insecticide compositions and methods for producing same Download PDFInfo
- Publication number
- WO2010074747A1 WO2010074747A1 PCT/US2009/006670 US2009006670W WO2010074747A1 WO 2010074747 A1 WO2010074747 A1 WO 2010074747A1 US 2009006670 W US2009006670 W US 2009006670W WO 2010074747 A1 WO2010074747 A1 WO 2010074747A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- composition
- ethyl
- diastereomers
- mmol
- Prior art date
Links
- 0 CC(c1ccc(*)nc1)Br Chemical compound CC(c1ccc(*)nc1)Br 0.000 description 2
- HJTKBEQYNOHPKA-UHFFFAOYSA-N CC(c1ccc(C(Cl)(Cl)Cl)nc1)Cl Chemical compound CC(c1ccc(C(Cl)(Cl)Cl)nc1)Cl HJTKBEQYNOHPKA-UHFFFAOYSA-N 0.000 description 1
- JGRACJXRTYFCRM-UHFFFAOYSA-N CC(c1ccc(C(F)(F)Cl)nc1)O Chemical compound CC(c1ccc(C(F)(F)Cl)nc1)O JGRACJXRTYFCRM-UHFFFAOYSA-N 0.000 description 1
- GZIMRECSYGRPMH-UHFFFAOYSA-N CC(c1ccc(C([F-])(F)Cl)nc1)=O Chemical compound CC(c1ccc(C([F-])(F)Cl)nc1)=O GZIMRECSYGRPMH-UHFFFAOYSA-N 0.000 description 1
- SHCDHIRSCJOUBW-UHFFFAOYSA-N CCc1ccc(C(O)=O)nc1 Chemical compound CCc1ccc(C(O)=O)nc1 SHCDHIRSCJOUBW-UHFFFAOYSA-N 0.000 description 1
- RYJURGZHLALOTJ-UHFFFAOYSA-N CCc1cnc(C(Cl)(Cl)Cl)cc1 Chemical compound CCc1cnc(C(Cl)(Cl)Cl)cc1 RYJURGZHLALOTJ-UHFFFAOYSA-N 0.000 description 1
- ZVQOOHYFBIDMTQ-XNQUEAFESA-N C[C@H](c1cnc(C(F)(F)F)cc1)S(C)(=NC#N)=O Chemical compound C[C@H](c1cnc(C(F)(F)F)cc1)S(C)(=NC#N)=O ZVQOOHYFBIDMTQ-XNQUEAFESA-N 0.000 description 1
- TVZRQJSNEISQFN-UHFFFAOYSA-N N#C/N=S(\CCC1)/C1c1cnc(C(F)(F)F)cc1 Chemical compound N#C/N=S(\CCC1)/C1c1cnc(C(F)(F)F)cc1 TVZRQJSNEISQFN-UHFFFAOYSA-N 0.000 description 1
- KHBLOULTRLDZPX-UHFFFAOYSA-N N#CN=S(CCC1)(C1c1ccc(C(F)(F)F)nc1)=O Chemical compound N#CN=S(CCC1)(C1c1ccc(C(F)(F)F)nc1)=O KHBLOULTRLDZPX-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/22—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom rings with more than six members
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/40—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having a double or triple bond to nitrogen, e.g. cyanates, cyanamides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/28—Radicals substituted by singly-bound oxygen or sulphur atoms
- C07D213/32—Sulfur atoms
- C07D213/34—Sulfur atoms to which a second hetero atom is attached
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/44—Radicals substituted by doubly-bound oxygen, sulfur, or nitrogen atoms, or by two such atoms singly-bound to the same carbon atom
- C07D213/53—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/54—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/57—Nitriles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the invention disclosed in this document is related to the field of pesticides and their use in controlling pests. BACKGROUND OF THE INVENTION
- compositions have been developed over time to destroy pests and alleviate the damages they cause.
- physical and chemical instabilities can lead to a reduction in pesticidal activity of the composition and/or present complications when it comes time to apply the composition to a locus where pest control is necessary or desired.
- physical and chemical instabilities can alter one or more properties of the composition which make it difficult or impossible to prepare appropriate solutions of the composition for use.
- many pesticide compositions are provided in a concentrated formulation from the manufacturer and are subsequently diluted by an end user before their application. During the time between manufacture and application, liquid forms of pesticide compositions can solidify as a result of chemical and physical instabilities of the composition.
- this solidification prevents or substantially impedes the dispersion of the composition into a solution suitable for application, resulting in greater user burden and cost and/or wasted pesticide products.
- physical and chemical instabilities lead to a reduction in pesticidal activity of a composition, an increase in the concentration at which the pesticide is applied and/or more frequent applications of the pesticide composition are often required.
- user costs and the cost to consumers can escalate. Therefore, a need exists for new pesticide compositions that exhibit increased chemical and physical stability properties.
- the present invention concerns novel compositions including an N-substituted (6- haloalkylpyridin-3-yl)alkyl sulfoximine compound and exhibiting increased stability, along with their use in controlling insects and certain other invertebrates, particularly aphids and other sucking insects.
- This invention also includes new synthetic procedures for preparing the compositions and methods of controlling insects using the compositions.
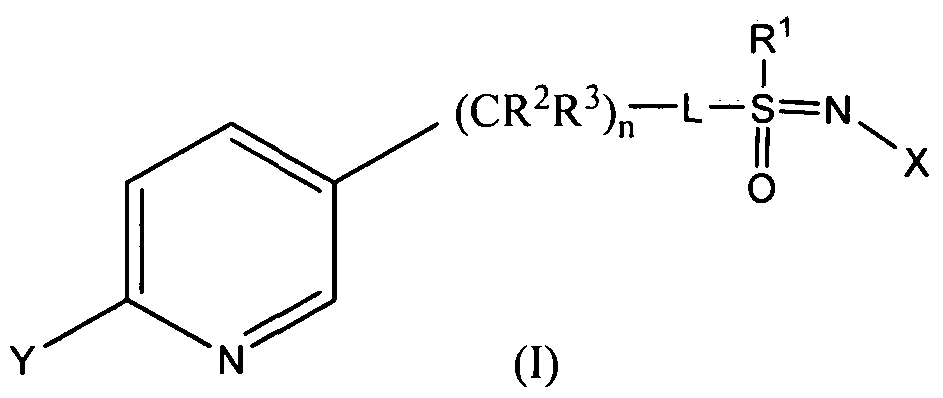
- compositions useful for the control of insects especially useful for the control of aphids and other sucking insects, along with methods for preparing same. More specifically, in one embodiment, a method includes providing a composition including a first ratio between stereoisomers of a compound having the formula (I)
- X represents NO 2 , CN or COOR 4 ;
- L represents a single bond or R 1 , S and L taken together represent a 4-, 5- or 6- membered ring;
- R 1 represents (Ci-C 4 ) alkyl
- R 2 and R 3 are distinct from each other and individually represent hydrogen, methyl, ethyl, flouro, chloro or bromo; n is 1 when L represents a single bond and is 0 when R 1 , S and L taken together represents a 4-, 5- or 6- membered ring;
- Y represents (Ci-C 4 ) haloalkyl, F, Cl, Br, or I;
- R 4 represents (Ci-C 3 ) alkyl.
- the method also includes heating the composition in a manner effective to provide a second, distinct ratio between the stereoisomers.
- the composition includes a compound of formula (I) wherein L represents a single bond, i.e., having the following structure wherein n is 1
- X represents NO 2 , CN or COOR 4 ;
- R 1 represents (C i -C 4 ) alkyl
- R 2 and R 3 are distinct from each other and individually represent hydrogen, methyl, ethyl, flouro, chloro or bromo;
- Y represents (C 1 -C 4 ) haloalkyl, F, Cl, Br, or I;
- R 4 represents (Ci-C 3 ) alkyl.
- the composition includes a compound of formula (I) wherein R 1 , S and L taken together form a saturated 5-membered ring, and n is O, i.e., having the structure
- X represents NO 2 , CN or COOR 4 ; Y represents (Ci-C 4 ) haloalkyl, F, Cl, Br, or I; and R 4 represents (Ci-C 3 ) alkyl.
- the composition includes compounds of formula (I) in one or more of the following classes: (1) Compounds of formula (I) wherein X is NO 2 or CN, most preferably CN.
- the heating is performed at a minimum of about 20 0 C for at least about four hours. In another form, the heating is performed at a minimum of about 50 0 C from about four to about seventy two hours.
- a method includes providing a composition which includes a stereoisomeric mixture of a compound having the following structure:
- X represents NO 2 , CN or COOR 4 ;
- R 1 represents (Ci-C 4 ) alkyl
- R 2 and R 3 are distinct from each other and individually represent hydrogen, methyl, ethyl, flouro, chloro or bromo;
- Y represents (Ci-C 4 ) haloalkyl, F, Cl, Br, or I;
- R 4 represents (C]-C 3 ) alkyl; and the mixture is defined by a first pair of diastereomers and a second pair of diastereomers.
- the method also includes heating the composition to convert at least a portion of the second pair of diastereomers to the first pair of diastereomers.
- a composition in another embodiment, includes a stereoisomeric mixture of ⁇ 1 -[6- (trifluoromethyl)pyridin-3-yl]ethyl ⁇ (methyl)oxido- ⁇ 4 -sulfanylidenecyanamide defined by a first pair of diastereomers and a second pair of diastereomers, wherein the first and second pairs of diastereomers are present at a ratio of at least about 3:1. In one form, the first and second pairs of diastereomers are present at a ratio from about 3:1 to 100:1. In another form, the first and second pairs of diastereomers are present at a ratio from about 3:1 to 40: 1. In yet another embodiment, a method includes applying to a locus where control is desired an insect-inactivating amount of a pesticide composition.
- alkyl (including derivative terms such as alkoxy) means straight chain, branched chain and cyclic groups including, for example, methyl, ethyl, 1-methylethyl, propyl, 1,1- dimethylethyl and cyclopropyl.
- alkoxy means an alkyl further consisting of a carbon-oxygen single bond, for example, methoxy, ethoxy, propoxy, isopropoxy, 1 -butoxy, 2-butoxy, isobutoxy, tert-butoxy, pentoxy, 2-methylbutoxy, 1 ,1-dimethylpropoxy, hexoxy, heptoxy, octoxy, nonoxy, and decoxy.
- aryl means a cyclic, aromatic substituent consisting of hydrogen and carbon, for example, phenyl, naphthyl, and biphenylyl.
- halo means fluoro, chloro, bromo, and iodo.
- haloalkyl means an alkyl group substituted with from one to the maximum possible number of halogen atoms, all combinations of halogens included.
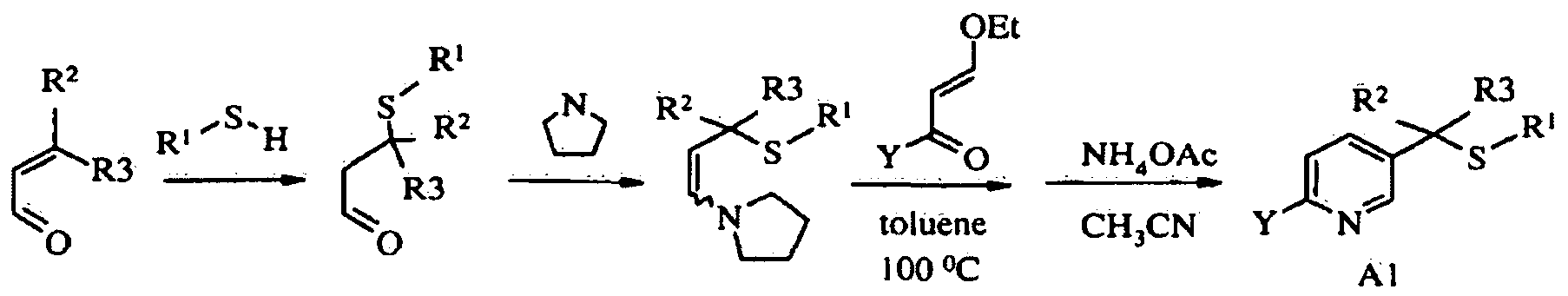
- step a of Scheme A sulfide of formula (A) is oxidized with meta- chloroperoxybenzoic acid (mCPBA) in a polar solvent below O 0 C to provide sulfoxide of formula (B).
- mCPBA meta- chloroperoxybenzoic acid
- step a of Scheme A sulfide of formula (A) is oxidized with meta- chloroperoxybenzoic acid (mCPBA) in a polar solvent below O 0 C to provide sulfoxide of formula (B).
- mCPBA meta- chloroperoxybenzoic acid
- dichloromethane is the preferred solvent for oxidation.
- step b of Scheme A sulfoxide (B) is iminated with sodium azide in the presence of concentrated sulfuric acid in an aprotic solvent under heating to provide sulfoximine of formula (C). In most cases, chloroform is the preferred solvent for this reaction.
- the nitrogen of sulfoximine (C) can be either cyanated with cyanogen bromide in the presence of a base, or nitrated with nitric acid in the presence of acetic anhydride under mildly elevated temperature, or carboxylated with alkyl (R4) chloroformate in the presence of base such as 4-dimethylaminopyridine (DMAP) to provide N-substitutedsulfoximine (Ia).
- Base is required for efficient cyanation and carboxylation and the preferred base is DMAP, whereas sulfuric acid is used as catalyst for efficient nitration reaction.
- step a of Scheme B sulfide is oxidized with iodobenzene diacetate in the presence of cyanamide at O 0 C to give sulfilimine (D).
- the reaction can be carried out in a polar aprotic solvent like CH 2 Cl 2 .
- step b of Scheme B the sulfilimine (D) is oxidized with wCPBA.
- a base such as potassium carbonate is employed to neutralize the acidity of r ⁇ CPBA.
- Protic polar solvents such as ethanol and water are used to increase the solubility of the sulfilimine starting material and the base employed.
- the sulfilimine (D) can also be oxidized with aqueous sodium or potassium periodinate solution in the presence of catalyst ruthenium trichloride hydrate or similar catalyst.
- the organic solvent for this catalysis can be polar aprotic solvent such as CH 2 Cl 2 , chloroform, or acetonitrile.
- KHMDS potassium hexamethyldisilamide
- a base like potassium-Z-butoxide in a polar aprotic solvent such as THF.
- an amine e.g., pyrrolidine
- DAST diethylaminosulfur trifluoride
- Example I Preparation of ri-(6-trifluoromethylpyridin-3-yDethyll(methyl)-oxido- ⁇ 4 - sulfanylidenecyanamide (2).
- Sulfoximine (2) is commonly known as sulfoxaflor, further details of which are available at http://www.alanwood.net/pesticides/index cn frame.html.
- sulfoximine (2) is also referred to as [methyl(oxido) ⁇ l-[6- (trifluoromethyl)-3-pyridyl]ethyl ⁇ - ⁇ 6 -sulfanylidene]cyanamide, and the CAS name given to sulfoximine (2) is N-[methyloxido[l-[6-(trifluoromethyl)-3-pyridinil]ethyl]- ⁇ 4 - sulfanylidenejcyanamide.
- Example VII Preparation of 2-trifluoromethyl-5-(l- ⁇ methyl(oxido ' )[oxido(oxo)hvdrazono]- ⁇ 4 -sulfanyl ⁇ ethvnpyridine (8).
- compositions of this invention can include compounds that can exist as one or more stereoisomers.
- the compositions include a mixture of stereoisomers of a compound according to formula (I).
- compositions of the present invention can include compounds of racemic mixtures, individual stereoisomers and optically active mixtures. It will be appreciated by those skilled in the art that one stereoisomer may be more active than the others. Individual stereoisomers and optically active mixtures may be obtained by selective synthetic procedures, by conventional synthetic procedures using resolved starting materials or by conventional resolution procedures.
- Diastereomer group (A) is defined by ⁇ (/?)- 1 -[6-
- Diastereomer group (B) is defined by ⁇ (/?)-l-[6-(triflouromethyl)pyridin-3-yl]ethyl ⁇ -
- compositions including a mixture of stereoisomers of a compound according to formula (I) conversion between the stereoisomers over time is contemplated, thereby resulting in ratios between the stereoisomers that are distinct from an initial ratio of the stereoisomers following synthesis of the compound.
- diastereomer groups (A) and (B) are present in an approximate 1 :2 mixture.
- a conversion between stereoisomers present in a composition at a first ratio is accelerated by heating the composition in a manner effective to yield a second ratio between the stereoisomers that is distinct from the first ratio.
- the heating is performed at a minimum of about 10 0 C for at least about 4 hours.
- the heating is performed at a minimum of about 20 0 C for at least about 4 hours.
- the heating is performed at a minimum of about 30 0 C for at least about 4 hours.
- the heating is performed at a minimum of about 40 0 C for at least about 4 hours.
- the heating is performed at a minimum of about 50 0 C for at least about 4 hours.
- the heating is performed at a minimum of about 60 0 C for at least about 4 hours. In a further example, the heating is performed at a minimum of about 70 0 C for at least about 4 hours. In yet another example, the heating is performed at a minimum of about 80 0 C for at least about 4 hours. In still another example, the heating is performed at a minimum of about 90 0 C for at least about 4 hours. In another example, the heating is performed at a minimum of about 100 0 C for at least about 4 hours.
- the heating can be performed at one of the temperatures specified above but for an alternative period of time, such as from about 1 to about 100 hours. In a more particular form, the heating is performed at one of the above temperatures from about 2 to about 90 hours. In another form, the heating is performed at one of the above temperatures from about 4 to about 72 hours. In another variant, it is contemplated that the heating could be performed at a temperature up to a point just below the degradation or melting point of the compound according to formula (I) in the composition, thereby avoiding degradation of the compound during heating.
- the heating should be performed at a temperature less than about 128 0 C, which is the lower of the two melting points for diastereomer groups A and B.
- the heating is performed from 23-70 0 C for a period from 4-72 hours. In another example, the heating is performed at 23 0 C for at least about four hours. In yet another example, the heating is performed at about 54 0 C for a period from 24-72 hours. In still another example, the heating is performed at about 70 0 C for a period from 4-72 hours. In a further variant of this example, the period is selected from one of 4, 8, 24 and 72 hours. Still, other variations in the temperature and time at which the heating are performed are contemplated. For example, it is contemplated that as the value for one of the temperature and period of time at which the heating is performed changes, the value for the other of the temperature and period of time at which the heating is performed may also change.
- the heating can be performed before the formulation of the composition is complete.
- a composition that includes a stereoisomeric mixture of a compound according to formula (I) and is substantially free of other materials is heated to obtain the desired conversion between stereoisomers.
- One or more additional materials may then be added to the composition, such as a(n) phytologically-acceptable carrier, wetting agent, thickener, insecticide or antifreeze, just to name a few possibilities.
- a composition that includes a stereoisomeric mixture of a compound according to formula (I) and at least one other insecticide, and is substantially free of other materials is heated to obtain the desired conversion between stereoisomers.
- compositions described herein may be prepared by a phytologically-acceptable carrier, wetting agent, thickener or antifreeze, just to name a few examples.
- the stereoisomers of the compound according to formula (I) define two pairs of diastereomers and the heating converts at least a portion of one pair of the diastereomers to the other pair of the diastereomers, thereby changing the ratio between the diastereomers of the compound present in the composition.
- the ratio between the pairs of diastereomers after heating is at least about 3:1.
- the ratio between the pairs of diastereomers after heating is greater than about 10: 1.
- the ratio between the pairs of diastereomers after heating is greater than about 20: 1.
- the ratio between the pairs of diastereomers after heating is greater than about 30:1.
- the ratio between the pairs of diastereomers after heating is greater than about 40:1. In a further form, the ratio between the pairs of diastereomers after heating is greater than about 50:1. In another form, the ratio between the pairs of diastereomers after heating is greater than about 60: 1. In still another form, the ratio between the pairs of diastereomers after heating is greater than about 70:1. In yet another form, the ratio between the pairs of diastereomers after heating is greater than about 80: 1. In another form, the ratio between the pairs of diastereomers after heating is greater than about 90:1. In still another form, the ratio between the pairs of diastereomers after heating is at least about 100:1.
- the ratio between the pairs of diastereomers after heating is from about 3: 1 to about 100: 1. In yet another form, the ratio between the pairs of diastereomers after heating is from about 3: 1 to about 50: 1. In another form, the ratio between the pairs of diastereomers after heating is from about 3: 1 to about 40: 1. Still, in another form, the ratio between the pairs of diastereomers after heating is from about 3: 1 to about 39: 1. In yet another form, the ratio between the pairs of diastereomers after heating is from about 19: 1 to about 39: 1.
- the ratio between the pairs of diastereomers after heating is from about 25: 1 to about 39: 1. Still, in other forms, the ratio between the pairs of diastereomers after heating is about 39: 1. It should be appreciated however that further variations of the ratio between the pairs of the diastereomers are contemplated.
- a composition in another embodiment, includes a compound according to formula (I) in a stereoisomeric mixture defined by two pairs of diastereomers.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of diastereomers from about 50 to about 98 weight percent and a second pair of diastereomers from about 2 to about 50 weight percent.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of diastereomers from about 60 to about 98 weight percent and a second pair of diastereomers from about 2 to about 40 weight percent.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of diastereomers from about 75 to about 98 weight percent and a second pair of diastereomers from about 2 to about 25 weight percent. In a further form, the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of diastereomers from about 85 to about 98 weight percent and a second pair of diastereomers from about 2 to about 15 weight percent.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of diastereomers from about 90 to about 98 weight percent and a second pair of diastereomers from about 2 to about 10 weight percent.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of diastereomers from about 95 to about 98 weight percent and a second pair of diastereomers from about 2 to about 5 weight percent.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, a first pair of the diastereomers at about 99 weight percent and a second pair of diastereomers at about 1 weight percent.
- the mixture includes, based on the total weight of the stereoisomeric mixture in the composition, about 100 weight percent of a first pair of the diastereomers.
- the compound according to formula (I) is ⁇ l-[6-(trifluoromethyl)pyridin-3-yl]ethyl ⁇ (methyl)oxido- ⁇ 4 -sulfanylidenecyanamide and the first pair of stereoisomers is defined by ⁇ (/?)-l -[6-(triflouromethyl)pyridin-3-yl]ethyl ⁇ -(/?)- (methyl)oxido- ⁇ 4 -sulfanylidenecyanamide and ⁇ (5)-l-[6-(triflouromethyl)pyridin-3-yl]ethyl ⁇ - (5)-(methyl)oxido- ⁇ 4 -suIfanylidenecyanamide and the second pair of diastereomers is defined by ⁇ ( ⁇ -l-f ⁇ -CtriflouromethyOpyridin-S-yOethyll-r ⁇ -CmethyOoxido- ⁇ 4 - sulfanylidene
- the stereoisomeric mixture having one of the foregoing weight percentages of diastereomers is prepared by heating the composition at a temperature from about 20-70 0 C for a period from about 4-72 hours.
- the composition can include a spinosyn, such as spinetoram, spinosad or mixtures thereof.
- the composition can include a ratio, by weight, between ⁇ l -[6-(trifluoromethyl)pyridin-3- yl]ethyl ⁇ (methyl)oxido ⁇ 4 -sulfanylidenecyanamide and the spinosyn from about 1 : 10 to about 10:1.
- the composition can include a ratio, by weight, between ⁇ l-[6- (trifluoromethyl)pyridin-3-yl]ethyl ⁇ (methyl)oxido ⁇ 4 -sulfanylidenecyanamide and the spinosyn from about 1 :5 to about 5:1.
- the composition can include a ratio, by weight, between ⁇ l-[6-(trifluorornethyl)pyridin-3-yl]ethyl ⁇ (methyl)oxido ⁇ 4 - sulfanylidenecyanamide and the spinosyn from about 1 :3 to about 3: 1.
- the composition can include a ratio, by weight, between ⁇ l-[6-(trifluoromethyl)pyridin-3- yl]ethyl ⁇ (methyl)oxido- ⁇ 4 -sulfanylidenecyanamide and the spinosyn from about 2:1 to about 2.4:1.
- compositions can include any compound according to formula (I) and one or more other insecticides.
- these compositions could include a compound according to formula (I) and a spinosyn, such as spinetoram, spinosad or mixtures thereof.
- these compositions can include a ratio by weight between the compound according to formula (I) and the insecticide from about 1 : 10 to about 10: 1.
- compositions can include a ratio by weight between the compound according to formula (I) and the insecticide from about 1 :5 to about 5: 1. In still another form, these compositions can include a ratio by weight between the compound according to formula (I) and the insecticide from about 1 :3 to about 3:1.
- the compositions of this invention may also be provided with a phytologically- acceptable inert carrier in the form of sprays, topical treatments, gels, seed coatings, microcapsulations, systemic uptake, baits, eartags, boluses, foggers, fumigants aerosols, dusts and many others. Typically, formulations are applied as aqueous suspensions or emulsions.
- Such suspensions or emulsions are produced from water-soluble, water suspendable, or emulsifiable formulations which are (1) solids, usually known as wettable powders or water dispersible granules or (2) liquids, usually known as emulsifiable concentrates, aqueous emulsions, suspension concentrates and water suspended capsules containing the composition.
- solids usually known as wettable powders or water dispersible granules or
- liquids usually known as emulsifiable concentrates, aqueous emulsions, suspension concentrates and water suspended capsules containing the composition.
- any material to which the composition can be added may be used, provided they yield the desired utility without significant interference with the activity of the composition as a pesticide.
- Wettable powders which may be compacted, extruded or processed through a dispersion in water followed by spray drying or fluid bed agglomeration to form water dispersible granules, comprise an intimate mixture of the composition, an inert carrier and surfactants.
- concentration of the composition in the wettable powder is usually from 10 percent to 90 percent by weight based on the total weight of the wettable powder, more preferably 25 weight percent to 75 weight percent.
- the composition can be compounded with any finely divided solid, such as prophyllite, talc, chalk, gypsum, Fuller's earth, bentonite, attapulgite, starch, casein, gluten, montmorillonite clays, diatomaceous earths, purified silicates or the like.
- finely divided carrier and surfactants are typically blended with the composition and milled.
- Emulsifiable concentrates of the composition comprise a convenient concentration, such as from 5 weight percent to 75 weight percent of the composition, in a suitable liquid, based on the total weight of the concentrate.
- the composition is dissolved in an inert carrier, which is either water, a water miscible solvent, a water immiscible solvent, or a mixture thereof and emulsifiers.
- the concentrates may be diluted with water and oil to form spray mixtures in the form of oil-in-water emulsions.
- Useful organic solvents include aromatics, especially the high-boiling naphthalenic and olefinic portions of petroleum such as heavy aromatic naphtha. Other organic solvents may also be used, such as, for example, terpenic solvents, including rosin derivatives, aliphatic ketones, such as cyclohexanone, and complex alcohols, such as 2-ethoxyethanol.
- Emulsifiers which can be advantageously employed herein can be readily determined by those skilled in the art and include various nonionic, anionic, cationic and amphoteric emulsifiers, or a blend of two or more emulsifiers.
- nonionic emulsifiers useful in preparing the emulsifiable concentrates include the polyalkylene glycol ethers and condensation products of alkyl and aryl phenols, aliphatic alcohols, aliphatic amines or fatty acids with ethylene oxide, propylene oxides such as the ethoxylated alkyl phenols and carboxylic esters solubilized with the polyol or polyokyalkylene.
- Cationic emulsifiers include quaternary ammonium compounds and fatty amine salts.
- Anionic emulsifiers include the oil-soluble salts (e.g., calcium) of alkylaryl sulphonic acids, oil soluble salts or sulfated polyglycol ethers and appropriate salts of phosphated polyglycol ether.
- aromatic liquids such as xylene, propyl benzene fractions; or mixed naphthalene fractions, mineral oils, substituted aromatic organic liquids such as dioctyl phthalate; kerosene; dialkyl amides of various
- Preferred organic liquids include xylene, and propyl benzene fractions, with propylbenzene fractions being most preferred.
- Surface-active emulsifying agents are typically employed in liquid formulations and in an amount of from 0.1 to 20 percent by weight based on the combined weight of the emulsifying agent with the composition.
- the formulations comprising the composition of the present invention can also contain other compatible additives, for example, miticides, insecticides, plant growth regulators, other fungicides, and other biologically active compounds used in agriculture.
- Aqueous suspensions comprise suspensions of the composition, dispersed in an aqueous vehicle at a concentration in the range from 5 to 50 weight percent, based on the total weight of the aqueous suspension.
- Aqueous suspensions are prepared by vigorously mixing the composition of the present invention, or its solution, into a vehicle comprised of water and surfactants chosen from the same types discussed above. Other components, such as inorganic salts and synthetic or natural gums, may also be added to increase the density and viscosity of the aqueous vehicle.
- aqueous suspensions include suspensions of oil droplets (EWs), solids (SCs), and capsules (CS's).
- the composition can also be applied as granular formulations, which are particularly useful for applications to the soil.
- Granular formulations usually contain from 0.5 to 10 weight percent, based on the total weight of the granular formulation of the composition, dispersed in an inert carrier which consists entirely or in large part of coarsely divided inert material such as attapulgite, bentonite, diatomite, clay or a similar inexpensive substance.
- Such formulations are usually prepared by diluting the composition in a suitable solvent and applying it to a granular carrier which has been preformed to the appropriate particle size, in the range of from 0.5 to 3 mm.
- a suitable solvent is a solvent in which the compound is substantially or completely soluble.
- Such formulations may also be prepared by making a dough or paste of the carrier and the composition and solvent, and crushing and drying to obtain the desired granular particle.
- the composition of the present invention can also be applied as a water dispersible granule, or dry flowable formulation.
- Water dispersible granules typically contain from 10 to 70 percent of the composition, based on the total weight of the formulation.
- Such formulations are typically obtained through mixing and/or spraying the mixture onto a carrier with the addition of a dispersing and/or wetting agent, and combining with water to form a mixture suitable for further processing using well known granulation technologies, such as pan granulation, extrusion, spray-drying, fluid bed agglomeration, and the like.
- Dusts containing the composition can be prepared by intimately mixing the composition with a suitable dusty agricultural carrier, such as, for example, kaolin clay, ground volcanic rock, and the like. Dusts can suitably contain from 1 to 10 weight percent of the composition, based on the total weight of the dust. Dusts may also be prepared by impregnating the composition onto a carrier in a similar manner to that described for granules above.
- a suitable dusty agricultural carrier such as, for example, kaolin clay, ground volcanic rock, and the like. Dusts can suitably contain from 1 to 10 weight percent of the composition, based on the total weight of the dust. Dusts may also be prepared by impregnating the composition onto a carrier in a similar manner to that described for granules above.
- the formulations of the present invention may additionally contain adjuvant surfactants to enhance deposition, wetting and penetration of the composition onto the target crop and organism.
- adjuvant surfactants may optionally be employed as a component of the formulation or as a tank mix.
- the amount of adjuvant surfactant will typically vary from 0.01 to 1.0 percent by volume, based on a spray-volume of water, preferably 0.05 to 0.5 volume percent.
- Suitable adjuvant surfactants include, but are not limited to ethoxylated nonyl phenols, ethoxylated synthetic or natural alcohols, salts of the esters or sulphosuccinic acids, ethoxylated organosilicones, ethoxylated fatty amines and blends of surfactants with mineral or vegetable oils.
- Example IX Preparation of composition including a stereoisomeric mixture of ( l-[6- (trifluoromethyl)pyridin-3-yllethvU(methyl)oxido- ⁇ 4 -sulfanylidenecvanamide.
- a 1 liter total volume ( ⁇ 1 100 g total weight) of a suspension concentrate product having a 240g/L concentration of ⁇ l-[6-(trifluoromethyl)pyridin-3-yl]ethyl ⁇ (methyl)oxido- ⁇ 4 -sulfanylidenecyanamide with an approximate 1 :2 ratio between diastereomer groups A and B was prepared by first adding deionized water to a clean beaker equipped with a mechanical stirrer.
- Van Buren St. Chicago, IL 60607; 40 g of propylene glycol; 1 g of Proxel® GXL, a microbiostat solution commercially available from Arch Chemicals, Inc., 1955 Lake Drive, Suite 100, Smyrna, GA 30080.
- a composition having a 160 mL total volume was prepared by combining 80 mL of the suspension concentrate product described above having a 240 g/L concentration of ⁇ l-[6- (trifluoromethyI)pyridin-3-yl]ethyl ⁇ (methyl)oxido- ⁇ 4 -sulfanylidenecyanamide and 80 mL of Radiant® SC, an insecticide formulation having a 120 g/L concentration of spinetoram and being commercially available from Dow AgroSciences, LLC, 9330 Zionsville Road, Indianapolis, IN 46268. The composition was thoroughly mixed until a homogenous consistency was obtained. Sixteen individual 10 mL samples (samples (i)-(xvi)) of the composition were then heated in an oven in accordance with the time and temperature parameters set forth in Table 2 below.
- compositions disclosed in this document are useful for the control of invertebrates including insects. Therefore, the present invention also is directed to a method for inhibiting an insect which comprises applying an insect-inhibiting amount of the composition to a locus of the insect, to the area to be protected, or directly on the insect to be controlled.
- the compositions of the invention may also be used to control other invertebrate pests such as mites and nematodes.
- insects which eat, damage or contact edible, commodity, ornamental, turf or pasture plants can be controlled by applying the compositions to the seed of the plant before planting, to the seedling, or cutting which is planted, the leaves, stems, fruits, grain, and/or roots, or to the soil or other growth medium before or after the crop is planted. Protection of these plants against virus, fungus or bacterium diseases may also be achieved indirectly through controlling sap-feeding pests such as whitefly, plant hopper, aphid and spider mite. Such plants include those which are bred through conventional approaches and which are genetically modified using modern biotechnology to gain insect-resistant, herbicide-resistant, nutrition-enhancement, and/or any other beneficial traits.
- compositions might also be useful to protect textiles, paper, stored grain, seeds and other foodstuffs, houses and other buildings which may be occupied by humans and/or companion, farm, ranch, zoo, or other animals, by applying an active composition to or riear such objects.
- domesticated animals, buildings or human beings might be protected with the compositions by controlling invertebrate and/or nematode pests that are parasitic or are capable of transmitting infectious diseases.
- pests include, for example, chiggers, ticks, lice, mosquitoes, flies, fleas and heartworms.
- Nonagronomic applications also include invertebrate pest control in forests, in yards, along road sides and railroad right of way.
- insects or other pests which can be inhibited include, but are not limited to:
- Lepidoptera Heliothis spp., Helicoverpa spp., Spodoptera spp., Mythimna unipuncta, Agrotis ipsilon, Earias spp., Euxoa auxiliaris, Trichoplusia ni, Anticarsia gemmatalis, Rachiplusia nu, Plutella xylostella, Chilo spp., Scirpophaga incertulas, Sesamia inferens, Cnaphalocrocis medinalis, Ostrinia nubilalis, Cydia pomonella, Carposina niponensis, Adoxophyes orana, Archips argyrospilus, Pandemis heparana, Epinotia aporema, Eupoecilia ambiguella, Lobesia botrana, Polychrosis viteana, Pectinophora gossypiella,
- Coleoptera ⁇ Diabrotica spp. Leptinotarsa decemlineata, Oulema or ⁇ zae, Anthonomus grandis, Lissorhoptrus oryzophilus, Agriotes spp., Melanotus communis, Popilliajaponica, Cyclocephala spp., Tribolium spp.
- Reticulitermes ⁇ avipes Coptotermes formosanus, Reticulitermes virginicus, Heterotermes aureus, Reticulitermes hesperus, Coptotermes frenchii, Shedorhinotermes spp., Reticulitermes santonensis, Reticulitermes grassei, Reticulitermes banyulensis, Reticulitermes speratus, Reticulitermes hageni, Reticulitermes tibialis, Zootermopsis spp., Incisitermes spp., Marginitermes spp., Macrotermes spp., Microcerotermes spp., Microtermes spp.
- Hymenoptera Iridomyrmex humilis, Solenopsis spp., Monomorium pharaonis, Atta spp., Pogonomyrmex spp., Camponotus spp., Monomorium spp., Tapinoma sessile, Tetramorium spp., Xylocapa spp., Vespula spp., Polistes spp.
- Orthoptera grasshoppers, crickets
- Melanoplus spp. Locusta migratoria
- Schistocerca gregaria Gryllotalpidae (mole crickets).
- Blattoidea (cockroaches) ⁇ Blatta orientalis, Blattella germanica, Periplaneta americana, Supella longipalpa, Periplaneta australasiae, Periplaneta brunnea, Parcoblatta pennsylvanica, Periplaneta fuliginosa, Pycnoscelus surinamensis,
- Siphonaptera ⁇ Ctenophalides spp. Pulex irritans Acari — Tetranychus spp., Panonychus spp., Eotetranychus carpini, Phyllocoptruta oleivora, Aculus pelekassi, Brevipalpus phoencis, Boophilus spp., Dermacentor variabilis, Rhipicephalus sanguineus, Amblyomma americanum, Ixodes spp., Notoedres cati, Sarcoptes scabiei, Dermatophagoides spp.
- Nematoda Dirofilaria immitis, Meloidogyne spp., Heterodera spp., Hoplolaimus columbus, Belonolaimus spp., Pratylenchus spp., Rotylenchus reniformis, Criconemella ornata, Ditylenchus spp., Aphelenchoides besseyi, Hirschmanniella spp.
- compositions to be applied to loci of insects and mites are not critical and can readily be determined by those skilled in the art in view of the examples above. In general, concentrations from 10 ppm to 5000 ppm by weight of compound are expected to provide good control. With many of the compounds, concentrations from 100 to 1500 ppm will suffice.
- the locus to which a composition is applied can be any locus inhabited by an insect or mite, for example, vegetable crops, fruit and nut trees, grape vines, ornamental plants, domesticated animals, the interior or exterior surfaces of buildings, and the soil around buildings.
- compositions of the invention may be utilized to control pests on one portion of the plant by applying the compositions to a different portion of it.
- control of foliar-feeding insects can be controlled by drip irrigation or furrow application, or by treating the seed before planting.
- Seed treatment can be applied to all types of seeds, including those from which plants genetically transformed to express specialized traits will germinate.
- compositions can also be provided as an insecticidal bait formulation including attractants and/or feeding stimulants that may be used to increase efficacy of the compositions against insect pest in a device such as trap, bait station, and the like.
- the bait formulation is usually a solid, semi-solid (including gel) or liquid bait matrix including the stimulants and one or more non-microencapsulated or microencapsulated insecticides in an amount effective to act as kill agents.
- compositions of the present invention are often applied in conjunction with one or more other insecticides or fungicides or herbicides to obtain control of a wider variety of pests diseases and weeds.
- the presently claimed compositions can be formulated with the other insecticides or fungicides or herbicide, tank mixed with the other insecticides or fungicides or herbicides, or applied sequentially with the other insecticides or fungicides or herbicides.
- antibiotic insecticides such
- Some of the herbicides that can be employed in conjunction with the compositions of the present invention include: amide herbicides such as allidochlor, beflubutamid, benzadox, benzipram, bromobutide, cafenstrole, CDEA, chlorthiamid, cyprazole, dimethenamid, dimethenamid-P, diphenamid, epronaz, etnipromid, fentrazamide, flupoxam, fomesafen, halosafen, isocarbamid, isoxaben, napropamide, naptalam, pethoxamid, propyzamide, quinonamid and tebutam; anilide herbicides such as chloranocryl, cisanilide, clomeprop, cypromid, diflufenican, etobenzanid, fenasulam, flufenacet, flufenican, mefenacet, mefi
Abstract
Description
Claims
Priority Applications (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011543496A JP5718249B2 (en) | 2008-12-26 | 2009-12-22 | Stable insecticide composition and method for producing the same |
| EP09795588.4A EP2369935B1 (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
| ES09795588.4T ES2589006T3 (en) | 2008-12-26 | 2009-12-22 | Stable insecticidal compositions and methods to produce them |
| BRPI0923702A BRPI0923702B8 (en) | 2008-12-26 | 2009-12-22 | STABLE INSECTICIDE COMPOSITIONS COMPRISING SPINOSIN AND A STEROISOMERIC MIXTURE OF SULFOXAFLOR, AND THEIR PRODUCTION METHOD |
| MX2011006909A MX2011006909A (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same. |
| UAA201109330A UA112281C2 (en) | 2008-12-26 | 2009-12-22 | STABLE INSECTICID COMPOSITIONS AND METHODS OF PREPARING THEM |
| CN200980152713.7A CN102271516B (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
| RU2011131067/13A RU2523293C2 (en) | 2008-12-26 | 2009-12-22 | Stable insecticidal composition and method of its obtaining (versions) |
| AU2009330658A AU2009330658B2 (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
| MX2013002749A MX343270B (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same. |
| KR1020117017454A KR101736078B1 (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
| CA2748132A CA2748132C (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
| NZ59316809A NZ593168A (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
| DK09795588.4T DK2369935T3 (en) | 2008-12-26 | 2009-12-22 | STABLE insecticidal FORMATIONS AND METHODS OF MAKING THEREOF |
| IL213706A IL213706A0 (en) | 2008-12-26 | 2011-06-21 | Stable insecticide compositions and methods for producing same |
| HK12101504A HK1160728A1 (en) | 2008-12-26 | 2012-02-15 | Stable insecticide compositions and methods for producing same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US20360008P | 2008-12-26 | 2008-12-26 | |
| US61/203,600 | 2008-12-26 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010074747A1 true WO2010074747A1 (en) | 2010-07-01 |
| WO2010074747A8 WO2010074747A8 (en) | 2011-11-03 |
Family
ID=42046455
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2009/006670 WO2010074747A1 (en) | 2008-12-26 | 2009-12-22 | Stable insecticide compositions and methods for producing same |
Country Status (20)
| Country | Link |
|---|---|
| US (3) | US8507532B2 (en) |
| EP (1) | EP2369935B1 (en) |
| JP (1) | JP5718249B2 (en) |
| KR (1) | KR101736078B1 (en) |
| CN (1) | CN102271516B (en) |
| AR (1) | AR075494A1 (en) |
| AU (1) | AU2009330658B2 (en) |
| BR (1) | BRPI0923702B8 (en) |
| CA (1) | CA2748132C (en) |
| CO (1) | CO6390084A2 (en) |
| DK (1) | DK2369935T3 (en) |
| ES (1) | ES2589006T3 (en) |
| HK (1) | HK1160728A1 (en) |
| IL (1) | IL213706A0 (en) |
| MX (2) | MX2011006909A (en) |
| NZ (1) | NZ593168A (en) |
| PL (1) | PL2369935T3 (en) |
| RU (1) | RU2523293C2 (en) |
| UA (1) | UA112281C2 (en) |
| WO (1) | WO2010074747A1 (en) |
Cited By (89)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011138285A1 (en) | 2010-05-05 | 2011-11-10 | Bayer Cropscience Ag | Thiazol derivatives as pest control agents |
| WO2011157778A1 (en) | 2010-06-18 | 2011-12-22 | Bayer Cropscience Ag | Active substance combinations with insecticide and acaricide properties |
| WO2012000896A2 (en) | 2010-06-28 | 2012-01-05 | Bayer Cropscience Ag | Heterocyclic compounds as agents for pest control |
| WO2012000902A1 (en) | 2010-06-29 | 2012-01-05 | Bayer Cropscience Ag | Improved insecticidal compositions comprising cyclic carbonylamidines |
| WO2012004293A2 (en) | 2010-07-08 | 2012-01-12 | Bayer Cropscience Ag | Insecticide and fungicide active ingredient combinations |
| WO2012004326A1 (en) | 2010-07-08 | 2012-01-12 | Bayer Cropscience Ag | Pesticidal pyrroline derivatives |
| WO2012007500A2 (en) | 2010-07-15 | 2012-01-19 | Bayer Cropscience Ag | New heterocyclic compounds as pesticides |
| WO2012034957A1 (en) | 2010-09-15 | 2012-03-22 | Bayer Cropscience Ag | Pesticidal pyrroline n-oxide derivatives |
| WO2012035011A1 (en) | 2010-09-15 | 2012-03-22 | Bayer Cropscience Ag | Pesticidal arylpyrrolidines |
| WO2012045680A2 (en) | 2010-10-04 | 2012-04-12 | Bayer Cropscience Ag | Insecticidal and fungicidal active substance combinations |
| WO2012052490A1 (en) | 2010-10-21 | 2012-04-26 | Bayer Cropscience Ag | N-benzyl heterocyclic carboxamides |
| WO2012052412A1 (en) | 2010-10-22 | 2012-04-26 | Bayer Cropscience Ag | Novel heterocyclic compounds as pesticides |
| WO2012059497A1 (en) | 2010-11-02 | 2012-05-10 | Bayer Cropscience Ag | N-hetarylmethyl pyrazolylcarboxamides |
| WO2012065944A1 (en) | 2010-11-15 | 2012-05-24 | Bayer Cropscience Ag | N-aryl pyrazole(thio)carboxamides |
| WO2012072660A1 (en) | 2010-12-01 | 2012-06-07 | Bayer Cropscience Ag | Use of fluopyram for controlling nematodes in crops and for increasing yield |
| WO2012072489A1 (en) | 2010-11-29 | 2012-06-07 | Bayer Cropscience Ag | Alpha,beta-unsaturated imines |
| WO2012076471A1 (en) | 2010-12-09 | 2012-06-14 | Bayer Cropscience Ag | Insecticidal mixtures with improved properties |
| WO2012076470A1 (en) | 2010-12-09 | 2012-06-14 | Bayer Cropscience Ag | Pesticidal mixtures with improved properties |
| WO2012082548A2 (en) | 2010-12-15 | 2012-06-21 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2012110519A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Cropscience Ag | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy and halogen-substituted spirocyclic ketoenols |
| WO2012110464A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Cropscience Ag | Use of sdhi fungicides on conventionally bred asr-tolerant, stem canker resistant and/or frog-eye leaf spot resistant soybean varieties |
| WO2012116960A1 (en) | 2011-03-01 | 2012-09-07 | Bayer Cropscience Ag | 2-acyloxy-pyrrolin-4-ones |
| WO2012120105A1 (en) | 2011-03-10 | 2012-09-13 | Bayer Cropscience Ag | Use of lipochito-oligosaccharide compounds for safeguarding seed safety of treated seeds |
| WO2012119984A1 (en) | 2011-03-09 | 2012-09-13 | Bayer Cropscience Ag | Indolecarboxamides and benzimidazolecarboxamides as insecticides and acaricides |
| WO2012126766A1 (en) | 2011-03-18 | 2012-09-27 | Bayer Cropscience Ag | N-(3-carbamoylphenyl)-1h-pyrazole-5-carboxamide derivatives and the use thereof for controlling animal pests |
| EP2535334A1 (en) | 2011-06-17 | 2012-12-19 | Bayer CropScience AG | Crystalline modifications of penflufen |
| EP2540163A1 (en) | 2011-06-30 | 2013-01-02 | Bayer CropScience AG | Nematocide N-cyclopropyl-sulfonylamide derivatives |
| WO2013014227A1 (en) | 2011-07-27 | 2013-01-31 | Bayer Intellectual Property Gmbh | Seed dressing for controlling phytopathogenic fungi |
| WO2013014126A1 (en) | 2011-07-26 | 2013-01-31 | Bayer Intellectual Property Gmbh | Etherified lactate esters, method for the production thereof and use thereof for enhancing the effect of plant protecting agents |
| WO2013050433A1 (en) | 2011-10-05 | 2013-04-11 | Bayer Intellectual Property Gmbh | Pesticide preparation and process for producing the same |
| EP2604118A1 (en) | 2011-12-15 | 2013-06-19 | Bayer CropScience AG | Active ingredient combinations having insecticidal and acaricidal properties |
| EP2606726A1 (en) | 2011-12-21 | 2013-06-26 | Bayer CropScience AG | N-Arylamidine-substituted trifluoroethylsulfide derivatives as acaricides and insecticides |
| WO2013092522A1 (en) | 2011-12-20 | 2013-06-27 | Bayer Intellectual Property Gmbh | Novel insecticidal aromatic amides |
| WO2013092519A1 (en) | 2011-12-19 | 2013-06-27 | Bayer Cropscience Ag | Use of anthranilic acid diamide derivatives for pest control in transgenic crops |
| WO2013107785A1 (en) | 2012-01-21 | 2013-07-25 | Bayer Intellectual Property Gmbh | Use of host defense inducers for controlling bacterial harmful organisms in useful plants |
| WO2013135724A1 (en) | 2012-03-14 | 2013-09-19 | Bayer Intellectual Property Gmbh | Pesticidal arylpyrrolidines |
| WO2013171199A1 (en) | 2012-05-16 | 2013-11-21 | Bayer Cropscience Ag | Insecticidal water-in-oil (w/o) formulation |
| WO2013171201A1 (en) | 2012-05-16 | 2013-11-21 | Bayer Cropscience Ag | Insecticidal oil-in water (o/w) formulation |
| WO2013174836A1 (en) | 2012-05-22 | 2013-11-28 | Bayer Cropscience Ag | Active compounds combinations comprising a lipo-chitooligosaccharide derivative and a nematicide, insecticidal or fungicidal compound |
| WO2013182613A1 (en) | 2012-06-08 | 2013-12-12 | Bayer Cropscience Ag | Detection system for the identification of insecticide resistance |
| WO2014019983A1 (en) | 2012-07-31 | 2014-02-06 | Bayer Cropscience Ag | Compositions comprising a pesticidal terpene mixture and an insecticide |
| WO2014026984A1 (en) | 2012-08-17 | 2014-02-20 | Bayer Cropscience Ag | Azaindole carboxylic acid amides and azaindole thiocarboxylic acid amides for use as insecticides and acaricides |
| WO2014053450A1 (en) | 2012-10-02 | 2014-04-10 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| WO2014060381A1 (en) | 2012-10-18 | 2014-04-24 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| WO2014067962A1 (en) | 2012-10-31 | 2014-05-08 | Bayer Cropscience Ag | Novel heterocyclic compounds as pest control agents |
| WO2014072250A1 (en) | 2012-11-06 | 2014-05-15 | Bayer Cropscience Ag | Herbicidal combinations for tolerant soybean cultures |
| WO2014083033A1 (en) | 2012-11-30 | 2014-06-05 | Bayer Cropsience Ag | Binary fungicidal or pesticidal mixture |
| WO2014083088A2 (en) | 2012-11-30 | 2014-06-05 | Bayer Cropscience Ag | Binary fungicidal mixtures |
| WO2014086759A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising biological control agents |
| WO2014086750A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising a biological control agent and an insecticide |
| WO2014086758A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising a biological control agent and an insecticide |
| WO2014086749A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising a biological control agent and an insecticide |
| WO2014086753A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising biological control agents |
| WO2014090765A1 (en) | 2012-12-12 | 2014-06-19 | Bayer Cropscience Ag | Use of 1-[2-fluoro-4-methyl-5-(2,2,2-trifluoroethylsulfinyl)phenyl]-5-amino-3-trifluoromethyl)-1 h-1,2,4 tfia zole for controlling nematodes in nematode-resistant crops |
| WO2014095826A1 (en) | 2012-12-18 | 2014-06-26 | Bayer Cropscience Ag | Binary fungicidal and bactericidal combinations |
| WO2014124375A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising gougerotin and a biological control agent |
| WO2014124379A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising a streptomyces-based biological control agent and an insecticide |
| WO2014122083A1 (en) | 2013-02-06 | 2014-08-14 | Bayer Cropscience Ag | Halogen-substituted pyrazol derivatives as pest-control agents |
| WO2014139897A1 (en) | 2013-03-12 | 2014-09-18 | Bayer Cropscience Ag | Use of dithiine-tetracarboximides for controlling bacterial harmful organisms in useful plants |
| WO2014170364A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Binary insecticidal or pesticidal mixture |
| WO2014170313A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Active compound combinations having insecticidal properties |
| DE202014008418U1 (en) | 2014-02-19 | 2014-11-14 | Clariant International Ltd. | Low foaming agrochemical compositions |
| DE202014008415U1 (en) | 2014-02-19 | 2014-11-25 | Clariant International Ltd. | Aqueous adjuvant composition for increasing the effectiveness of electrolyte active substances |
| WO2014202510A1 (en) | 2013-06-20 | 2014-12-24 | Bayer Cropscience Ag | Aryl sulfide derivatives and aryl sulfoxide derivatives as acaricides and insecticides |
| WO2014202505A1 (en) | 2013-06-20 | 2014-12-24 | Bayer Cropscience Ag | Aryl sulfide derivatives and aryl sulfoxide derivatives as acaricides and insecticides |
| WO2015004028A1 (en) | 2013-07-08 | 2015-01-15 | Bayer Cropscience Ag | Six-membered c-n-linked aryl sulfide derivatives and aryl sulfoxide derivatives as pest control agents |
| DE102014018274A1 (en) | 2014-12-12 | 2015-07-30 | Clariant International Ltd. | Sugar surfactants and their use in agrochemical compositions |
| WO2015160620A1 (en) | 2014-04-16 | 2015-10-22 | Bayer Cropscience Lp | Compositions comprising ningnanmycin and an insecticide |
| WO2015160618A1 (en) | 2014-04-16 | 2015-10-22 | Bayer Cropscience Lp | Compositions comprising ningnanmycin and a biological control agent |
| WO2016001129A1 (en) | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Improved insecticidal compositions |
| DE102014012022A1 (en) | 2014-08-13 | 2016-02-18 | Clariant International Ltd. | Organic ammonium salts of anionic pesticides |
| WO2016106063A1 (en) | 2014-12-22 | 2016-06-30 | Bayer Corpscience Lp | Method for using a bacillus subtilis or bacillus pumilus strain to treat or prevent pineapple disease |
| US9510594B2 (en) | 2011-02-17 | 2016-12-06 | Bayer Intellectual Property Gmbh | Use of SDHI fungicides on conventionally bred ASR-tolerant, stem canker resistant and/or frog-eye leaf spot resistant soybean varieties |
| WO2017186543A2 (en) | 2016-04-24 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Use of fluopyram and/or bacillus subtilis for controlling fusarium wilt in plants of the musaceae family |
| EP3243387A2 (en) | 2012-05-30 | 2017-11-15 | Bayer CropScience Aktiengesellschaft | Compositions comprising a biological control agent and an insecticide |
| WO2018019676A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Active compound combinations and methods to protect the propagation material of plants |
| EP3363289A2 (en) | 2012-05-30 | 2018-08-22 | Bayer CropScience Aktiengesellschaft | Compositions comprising a biological control agent and an insecticide |
| US10772324B2 (en) | 2012-11-03 | 2020-09-15 | Clariant International Ltd. | Aqueous adjuvant-compositions |
| US10813862B2 (en) | 2012-05-30 | 2020-10-27 | Clariant International Ltd. | Use of N-methyl-N-acylglucamines as solubilizers |
| US10864275B2 (en) | 2012-05-30 | 2020-12-15 | Clariant International Ltd. | N-methyl-N-acylglucamine-containing composition |
| US10920080B2 (en) | 2015-10-09 | 2021-02-16 | Clariant International Ltd. | N-Alkyl glucamine-based universal pigment dispersions |
| US10961484B2 (en) | 2015-10-09 | 2021-03-30 | Clariant International Ltd. | Compositions comprising sugar amine and fatty acid |
| US11220603B2 (en) | 2016-05-09 | 2022-01-11 | Clariant International Ltd. | Stabilizers for silicate paints |
| US11425904B2 (en) | 2014-04-23 | 2022-08-30 | Clariant International Ltd. | Use of aqueous drift-reducing compositions |
| US11700852B2 (en) | 2014-12-19 | 2023-07-18 | Clariant International Ltd | Aqueous electrolyte-containing adjuvant compositions, active ingredient-containing compositions and the use thereof |
| US11750794B2 (en) | 2015-03-24 | 2023-09-05 | Augmedics Ltd. | Combining video-based and optic-based augmented reality in a near eye display |
| US11766296B2 (en) | 2018-11-26 | 2023-09-26 | Augmedics Ltd. | Tracking system for image-guided surgery |
| US11801115B2 (en) | 2019-12-22 | 2023-10-31 | Augmedics Ltd. | Mirroring in image guided surgery |
| US11896445B2 (en) | 2021-07-07 | 2024-02-13 | Augmedics Ltd. | Iliac pin and adapter |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI398433B (en) * | 2006-02-10 | 2013-06-11 | Dow Agrosciences Llc | Insecticidal n-substituted (6-haloalkylpyridin-3-yl)alkyl sulfoximines |
| CN102264224B (en) * | 2008-12-26 | 2014-10-08 | 美国陶氏益农公司 | Stable sulfoximine-insecticide compositions |
| NZ593168A (en) | 2008-12-26 | 2013-12-20 | Dow Agrosciences Llc | Stable insecticide compositions and methods for producing same |
| EP2717700A4 (en) * | 2011-06-07 | 2015-02-18 | Dow Agrosciences Llc | Oil dispersion of sulfoximines for the control of insects |
| EP2866564B1 (en) * | 2012-06-30 | 2020-02-12 | Dow AgroSciences LLC | Production of n-substituted sulfoximine pyridine n-oxides |
| JP6242392B2 (en) * | 2012-06-30 | 2017-12-06 | ダウ アグロサイエンシィズ エルエルシー | Pyridine N-oxide and process for producing them |
| BR112014032874B8 (en) * | 2012-06-30 | 2022-08-23 | Dow Agrosciences Llc | SULPHYLIMINE AND N-SUBSTITUTED SULFOXIMINE PYRIDEN-OXIDES INSECTICIDES, COMPOSITION AND METHOD TO CONTROL INSECTS |
| CN103333102B (en) * | 2013-06-08 | 2015-06-17 | 北京格林凯默科技有限公司 | Pyridyl-N-cyano sulfo oxime compound and preparation method thereof |
| CN103333101B (en) * | 2013-06-08 | 2015-06-17 | 北京格林凯默科技有限公司 | Pyridyl sulfoximine compound and preparation method thereof |
| TW201639453A (en) * | 2015-03-31 | 2016-11-16 | 陶氏農業科學公司 | Pesticidal compositions and related methods |
| CN105503672A (en) * | 2015-12-06 | 2016-04-20 | 杨秀娟 | Method for synthesizing aryl sulfone compound as pharmaceutical intermediate |
| CN109688815A (en) | 2016-07-01 | 2019-04-26 | 麦克劳克林戈姆利金公司 | Sabadilla alkaloid and the mixture of Dipel and application thereof |
| CN107254920B (en) * | 2016-11-11 | 2019-11-01 | 新沂市金达木业有限公司 | A kind of preparation method of indoor decoration fire-proof plate that capableing of pre- favorable moth-proof |
| US10925286B2 (en) * | 2017-03-17 | 2021-02-23 | Telluris Biotech India Private Limited | Synergistic chalcone containing composition of a nematicide |
| CN115466208A (en) * | 2022-11-01 | 2022-12-13 | 安徽宁亿泰科技有限公司 | Method for purifying cis-intermediate of spirotetramat |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007095229A2 (en) * | 2006-02-10 | 2007-08-23 | Dow Agrosciences Llc | Insecticidal n-substituted (6-haloalkylpyridin-3-yl)alkyl sulfoximines |
| WO2007149134A1 (en) * | 2006-06-23 | 2007-12-27 | Dow Agrosciences Llc | A method to control insects resistant to common insecticides |
| WO2009134224A1 (en) * | 2008-05-01 | 2009-11-05 | Dow Agrosciences Llc | Synergistic pesticidal mixtures |
| WO2009135613A1 (en) * | 2008-05-07 | 2009-11-12 | Bayer Cropscience Aktiengesellschaft | Synergistic active ingredient combinations |
Family Cites Families (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050038501A1 (en) * | 2003-08-12 | 2005-02-17 | Moore James E. | Dynamic stent |
| UY28506A1 (en) | 2003-09-12 | 2005-04-29 | Basf Ag | 6-HALOGEN- (1,2,4) TRIAZOL (1,5-A) PYRIMIDINS TO COMBAT ANIMAL PESTS. |
| BRPI0509695B8 (en) * | 2004-04-08 | 2022-06-28 | Dow Agrosciences Llc | N-SUBSTITUTED SULFOXYMINES, COMPOSITION AND METHOD TO CONTROL INSECTS |
| TW200626532A (en) | 2004-09-21 | 2006-08-01 | Syngenta Participations Ag | Novel insecticides |
| TWI383973B (en) * | 2006-08-07 | 2013-02-01 | Dow Agrosciences Llc | Process for the preparation of 2-substituted-5-(1-alkylthio)alkylpyridines |
| TWI409256B (en) * | 2006-09-01 | 2013-09-21 | Dow Agrosciences Llc | Insecticidal n-substituted (heteroaryl)cycloalkyl sulfoximines |
| TWI387585B (en) * | 2006-09-01 | 2013-03-01 | Dow Agrosciences Llc | Insecticidal n-substituted (heteroaryl)alkyl sulfilimines |
| TW200820902A (en) * | 2006-11-08 | 2008-05-16 | Dow Agrosciences Llc | Use of N-substituted sulfoximines for control of invertebrate pests |
| TWI395737B (en) * | 2006-11-08 | 2013-05-11 | Dow Agrosciences Llc | Heteroaryl (substituted)alkyl n-substituted sulfoximines as insecticides |
| TWI383970B (en) * | 2006-11-08 | 2013-02-01 | Dow Agrosciences Llc | Multi-substituted pyridyl sulfoximines and their use as insecticides |
| EP2364971B1 (en) * | 2006-11-30 | 2014-04-30 | Dow AgroSciences LLC | 2,5-Disubstituted pyridines for the preparation of 2-substituted 5-(1-alkylthio)-alkylpyridines |
| US7511149B2 (en) * | 2007-02-09 | 2009-03-31 | Dow Agrosciences Llc | Process for the oxidation of certain substituted sulfilimines to insecticidal sulfoximines |
| US7709648B2 (en) * | 2007-02-09 | 2010-05-04 | Dow Agrosciences Llc | Process for the preparation of 2-substituted-5-(1-alkylthio)alkylpyridines |
| EP2114886B1 (en) | 2007-02-26 | 2014-03-26 | Dow AgroSciences LLC | Process for the preparation of certain substituted sulfilimines |
| US8153560B2 (en) | 2007-03-01 | 2012-04-10 | Basf Se | Pesticidal active mixtures comprising aminothiazoline compounds |
| AR066366A1 (en) * | 2007-05-01 | 2009-08-12 | Dow Agrosciences Llc | PESTICIDED SYNERGIC BLENDS |
| US7844777B2 (en) * | 2007-06-26 | 2010-11-30 | Intel Corporation | Cache for a host controller to store command header information |
| US20090006676A1 (en) * | 2007-06-29 | 2009-01-01 | Intel Corporation | Method for Scheduling a Device Notification Time Slot for a Wireless USB System |
| US8604077B2 (en) | 2007-09-04 | 2013-12-10 | Segetis, Inc. | Ketal compounds from polyols and oxocarboxylates |
| CN101909448A (en) | 2007-11-16 | 2010-12-08 | 巴斯夫欧洲公司 | The pesticide combination that comprises cyanosulfoximine compounds |
| TWM332793U (en) * | 2007-11-28 | 2008-05-21 | Cooler Master Co Ltd | Heat radiating structure and the lighting apparatus |
| US20110160054A1 (en) * | 2008-08-28 | 2011-06-30 | Basf Se | Pesticidal Mixtures Comprising Cyanosulfoximine Compounds and Spinetoram |
| NZ593168A (en) | 2008-12-26 | 2013-12-20 | Dow Agrosciences Llc | Stable insecticide compositions and methods for producing same |
-
2009
- 2009-12-22 NZ NZ59316809A patent/NZ593168A/en unknown
- 2009-12-22 AU AU2009330658A patent/AU2009330658B2/en active Active
- 2009-12-22 DK DK09795588.4T patent/DK2369935T3/en active
- 2009-12-22 RU RU2011131067/13A patent/RU2523293C2/en active
- 2009-12-22 ES ES09795588.4T patent/ES2589006T3/en active Active
- 2009-12-22 AR ARP090105030 patent/AR075494A1/en not_active Application Discontinuation
- 2009-12-22 MX MX2011006909A patent/MX2011006909A/en active IP Right Grant
- 2009-12-22 EP EP09795588.4A patent/EP2369935B1/en active Active
- 2009-12-22 MX MX2013002749A patent/MX343270B/en unknown
- 2009-12-22 WO PCT/US2009/006670 patent/WO2010074747A1/en active Application Filing
- 2009-12-22 BR BRPI0923702A patent/BRPI0923702B8/en active IP Right Grant
- 2009-12-22 JP JP2011543496A patent/JP5718249B2/en active Active
- 2009-12-22 PL PL09795588T patent/PL2369935T3/en unknown
- 2009-12-22 KR KR1020117017454A patent/KR101736078B1/en active IP Right Grant
- 2009-12-22 CN CN200980152713.7A patent/CN102271516B/en active Active
- 2009-12-22 CA CA2748132A patent/CA2748132C/en active Active
- 2009-12-22 US US12/653,996 patent/US8507532B2/en active Active
- 2009-12-22 UA UAA201109330A patent/UA112281C2/en unknown
-
2011
- 2011-06-21 IL IL213706A patent/IL213706A0/en active IP Right Grant
- 2011-06-22 CO CO11078100A patent/CO6390084A2/en active IP Right Grant
-
2012
- 2012-02-15 HK HK12101504A patent/HK1160728A1/en unknown
-
2013
- 2013-03-15 US US13/833,069 patent/US8846717B2/en active Active
-
2014
- 2014-07-01 US US14/321,373 patent/US9125412B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007095229A2 (en) * | 2006-02-10 | 2007-08-23 | Dow Agrosciences Llc | Insecticidal n-substituted (6-haloalkylpyridin-3-yl)alkyl sulfoximines |
| US20070203191A1 (en) | 2006-02-10 | 2007-08-30 | Loso Michael R | Insecticidal N-substituted (6-haloalkylpyridin-3-yl)alkyl sulfoximines |
| WO2007149134A1 (en) * | 2006-06-23 | 2007-12-27 | Dow Agrosciences Llc | A method to control insects resistant to common insecticides |
| WO2009134224A1 (en) * | 2008-05-01 | 2009-11-05 | Dow Agrosciences Llc | Synergistic pesticidal mixtures |
| WO2009135613A1 (en) * | 2008-05-07 | 2009-11-12 | Bayer Cropscience Aktiengesellschaft | Synergistic active ingredient combinations |
Cited By (113)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8686156B2 (en) | 2010-05-05 | 2014-04-01 | Bayer Cropscience Ag | Thiazole derivatives as pesticides |
| WO2011138285A1 (en) | 2010-05-05 | 2011-11-10 | Bayer Cropscience Ag | Thiazol derivatives as pest control agents |
| WO2011157778A1 (en) | 2010-06-18 | 2011-12-22 | Bayer Cropscience Ag | Active substance combinations with insecticide and acaricide properties |
| US9968086B2 (en) | 2010-06-18 | 2018-05-15 | Bayer Intellectual Property Gmbh | Active ingredient combinations having insecticidal and acaricidal properties |
| US9198424B2 (en) | 2010-06-18 | 2015-12-01 | Bayer Intellectual Property Gmbh | Active ingredient combinations having insecticidal and acaricidal properties |
| WO2012000896A2 (en) | 2010-06-28 | 2012-01-05 | Bayer Cropscience Ag | Heterocyclic compounds as agents for pest control |
| US9044015B2 (en) | 2010-06-28 | 2015-06-02 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| US8686004B2 (en) | 2010-06-28 | 2014-04-01 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| WO2012000902A1 (en) | 2010-06-29 | 2012-01-05 | Bayer Cropscience Ag | Improved insecticidal compositions comprising cyclic carbonylamidines |
| WO2012004293A2 (en) | 2010-07-08 | 2012-01-12 | Bayer Cropscience Ag | Insecticide and fungicide active ingredient combinations |
| WO2012004326A1 (en) | 2010-07-08 | 2012-01-12 | Bayer Cropscience Ag | Pesticidal pyrroline derivatives |
| WO2012007500A2 (en) | 2010-07-15 | 2012-01-19 | Bayer Cropscience Ag | New heterocyclic compounds as pesticides |
| US9233951B2 (en) | 2010-07-15 | 2016-01-12 | Bayer Intellectual Property Gmbh | Heterocyclic compounds as pesticides |
| WO2012035011A1 (en) | 2010-09-15 | 2012-03-22 | Bayer Cropscience Ag | Pesticidal arylpyrrolidines |
| WO2012034957A1 (en) | 2010-09-15 | 2012-03-22 | Bayer Cropscience Ag | Pesticidal pyrroline n-oxide derivatives |
| US9375000B2 (en) | 2010-09-15 | 2016-06-28 | Bayer Intellectual Property Gmbh | Pesticidal arylpyrrolidines |
| WO2012045680A2 (en) | 2010-10-04 | 2012-04-12 | Bayer Cropscience Ag | Insecticidal and fungicidal active substance combinations |
| WO2012052490A1 (en) | 2010-10-21 | 2012-04-26 | Bayer Cropscience Ag | N-benzyl heterocyclic carboxamides |
| WO2012052412A1 (en) | 2010-10-22 | 2012-04-26 | Bayer Cropscience Ag | Novel heterocyclic compounds as pesticides |
| US9173396B2 (en) | 2010-10-22 | 2015-11-03 | Bayer Intellectual Property Gmbh | Heterocyclic compounds as pesticides |
| WO2012059497A1 (en) | 2010-11-02 | 2012-05-10 | Bayer Cropscience Ag | N-hetarylmethyl pyrazolylcarboxamides |
| WO2012065944A1 (en) | 2010-11-15 | 2012-05-24 | Bayer Cropscience Ag | N-aryl pyrazole(thio)carboxamides |
| US9206137B2 (en) | 2010-11-15 | 2015-12-08 | Bayer Intellectual Property Gmbh | N-Aryl pyrazole(thio)carboxamides |
| WO2012072489A1 (en) | 2010-11-29 | 2012-06-07 | Bayer Cropscience Ag | Alpha,beta-unsaturated imines |
| US9055743B2 (en) | 2010-11-29 | 2015-06-16 | Bayer Intellectual Property Gmbh | Alpha, beta-unsaturated imines |
| WO2012072660A1 (en) | 2010-12-01 | 2012-06-07 | Bayer Cropscience Ag | Use of fluopyram for controlling nematodes in crops and for increasing yield |
| WO2012076470A1 (en) | 2010-12-09 | 2012-06-14 | Bayer Cropscience Ag | Pesticidal mixtures with improved properties |
| WO2012076471A1 (en) | 2010-12-09 | 2012-06-14 | Bayer Cropscience Ag | Insecticidal mixtures with improved properties |
| WO2012082548A2 (en) | 2010-12-15 | 2012-06-21 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| US9510594B2 (en) | 2011-02-17 | 2016-12-06 | Bayer Intellectual Property Gmbh | Use of SDHI fungicides on conventionally bred ASR-tolerant, stem canker resistant and/or frog-eye leaf spot resistant soybean varieties |
| WO2012110464A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Cropscience Ag | Use of sdhi fungicides on conventionally bred asr-tolerant, stem canker resistant and/or frog-eye leaf spot resistant soybean varieties |
| WO2012110518A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Pharma Aktiengesellschaft | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-4-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy |
| WO2012110519A1 (en) | 2011-02-17 | 2012-08-23 | Bayer Cropscience Ag | Substituted 3-(biphenyl-3-yl)-8,8-difluoro-hydroxy-1-azaspiro[4.5]dec-3-en-2-ones for therapy and halogen-substituted spirocyclic ketoenols |
| US9204640B2 (en) | 2011-03-01 | 2015-12-08 | Bayer Intellectual Property Gmbh | 2-acyloxy-pyrrolin-4-ones |
| WO2012116960A1 (en) | 2011-03-01 | 2012-09-07 | Bayer Cropscience Ag | 2-acyloxy-pyrrolin-4-ones |
| US9107411B2 (en) | 2011-03-09 | 2015-08-18 | Bayer Intellectual Property Gmbh | Indolecarboxamides and benzimidazolecarboxamides as insecticides and acaricides |
| WO2012119984A1 (en) | 2011-03-09 | 2012-09-13 | Bayer Cropscience Ag | Indolecarboxamides and benzimidazolecarboxamides as insecticides and acaricides |
| WO2012120105A1 (en) | 2011-03-10 | 2012-09-13 | Bayer Cropscience Ag | Use of lipochito-oligosaccharide compounds for safeguarding seed safety of treated seeds |
| WO2012126766A1 (en) | 2011-03-18 | 2012-09-27 | Bayer Cropscience Ag | N-(3-carbamoylphenyl)-1h-pyrazole-5-carboxamide derivatives and the use thereof for controlling animal pests |
| EP2535334A1 (en) | 2011-06-17 | 2012-12-19 | Bayer CropScience AG | Crystalline modifications of penflufen |
| WO2013010758A1 (en) | 2011-06-30 | 2013-01-24 | Bayer Intellectual Property Gmbh | Nematocide n- cyclopropyl - sulfonylamide derivatives |
| EP2540163A1 (en) | 2011-06-30 | 2013-01-02 | Bayer CropScience AG | Nematocide N-cyclopropyl-sulfonylamide derivatives |
| US9826734B2 (en) | 2011-07-26 | 2017-11-28 | Clariant International Ltd. | Etherified lactate esters, method for the production thereof and use thereof for enhancing the effect of plant protecting agents |
| WO2013014126A1 (en) | 2011-07-26 | 2013-01-31 | Bayer Intellectual Property Gmbh | Etherified lactate esters, method for the production thereof and use thereof for enhancing the effect of plant protecting agents |
| WO2013014227A1 (en) | 2011-07-27 | 2013-01-31 | Bayer Intellectual Property Gmbh | Seed dressing for controlling phytopathogenic fungi |
| WO2013050433A1 (en) | 2011-10-05 | 2013-04-11 | Bayer Intellectual Property Gmbh | Pesticide preparation and process for producing the same |
| WO2013087709A1 (en) | 2011-12-15 | 2013-06-20 | Bayer Intellectual Property Gmbh | Active ingredient combinations having insecticidal and acaricidal properties |
| EP2604118A1 (en) | 2011-12-15 | 2013-06-19 | Bayer CropScience AG | Active ingredient combinations having insecticidal and acaricidal properties |
| WO2013092519A1 (en) | 2011-12-19 | 2013-06-27 | Bayer Cropscience Ag | Use of anthranilic acid diamide derivatives for pest control in transgenic crops |
| WO2013092522A1 (en) | 2011-12-20 | 2013-06-27 | Bayer Intellectual Property Gmbh | Novel insecticidal aromatic amides |
| WO2013092350A1 (en) | 2011-12-21 | 2013-06-27 | Bayer Cropscience Ag | N-arylamidine-substituted trifluoroethyl sulfide derivatives as acaricides and insecticides |
| EP2606726A1 (en) | 2011-12-21 | 2013-06-26 | Bayer CropScience AG | N-Arylamidine-substituted trifluoroethylsulfide derivatives as acaricides and insecticides |
| EP3395171A1 (en) | 2011-12-21 | 2018-10-31 | Bayer CropScience Aktiengesellschaft | N-arylamidine-substituted trifluoroethylsulfide derivatives as acaricides and insecticides |
| WO2013107785A1 (en) | 2012-01-21 | 2013-07-25 | Bayer Intellectual Property Gmbh | Use of host defense inducers for controlling bacterial harmful organisms in useful plants |
| WO2013135724A1 (en) | 2012-03-14 | 2013-09-19 | Bayer Intellectual Property Gmbh | Pesticidal arylpyrrolidines |
| WO2013171201A1 (en) | 2012-05-16 | 2013-11-21 | Bayer Cropscience Ag | Insecticidal oil-in water (o/w) formulation |
| WO2013171199A1 (en) | 2012-05-16 | 2013-11-21 | Bayer Cropscience Ag | Insecticidal water-in-oil (w/o) formulation |
| WO2013174836A1 (en) | 2012-05-22 | 2013-11-28 | Bayer Cropscience Ag | Active compounds combinations comprising a lipo-chitooligosaccharide derivative and a nematicide, insecticidal or fungicidal compound |
| EP3363289A2 (en) | 2012-05-30 | 2018-08-22 | Bayer CropScience Aktiengesellschaft | Compositions comprising a biological control agent and an insecticide |
| US10864275B2 (en) | 2012-05-30 | 2020-12-15 | Clariant International Ltd. | N-methyl-N-acylglucamine-containing composition |
| US10813862B2 (en) | 2012-05-30 | 2020-10-27 | Clariant International Ltd. | Use of N-methyl-N-acylglucamines as solubilizers |
| EP3243387A2 (en) | 2012-05-30 | 2017-11-15 | Bayer CropScience Aktiengesellschaft | Compositions comprising a biological control agent and an insecticide |
| WO2013182613A1 (en) | 2012-06-08 | 2013-12-12 | Bayer Cropscience Ag | Detection system for the identification of insecticide resistance |
| WO2014019983A1 (en) | 2012-07-31 | 2014-02-06 | Bayer Cropscience Ag | Compositions comprising a pesticidal terpene mixture and an insecticide |
| EP3424322A1 (en) | 2012-07-31 | 2019-01-09 | Bayer CropScience Aktiengesellschaft | Compositions comprising a pesticidal terpene mixture and an insecticide |
| WO2014026984A1 (en) | 2012-08-17 | 2014-02-20 | Bayer Cropscience Ag | Azaindole carboxylic acid amides and azaindole thiocarboxylic acid amides for use as insecticides and acaricides |
| WO2014053450A1 (en) | 2012-10-02 | 2014-04-10 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| WO2014060381A1 (en) | 2012-10-18 | 2014-04-24 | Bayer Cropscience Ag | Heterocyclic compounds as pesticides |
| WO2014067962A1 (en) | 2012-10-31 | 2014-05-08 | Bayer Cropscience Ag | Novel heterocyclic compounds as pest control agents |
| US10772324B2 (en) | 2012-11-03 | 2020-09-15 | Clariant International Ltd. | Aqueous adjuvant-compositions |
| WO2014072250A1 (en) | 2012-11-06 | 2014-05-15 | Bayer Cropscience Ag | Herbicidal combinations for tolerant soybean cultures |
| EP3369318A1 (en) | 2012-11-06 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Herbicidal combinations for tolerant soybean cultures |
| EP3387906A1 (en) | 2012-11-06 | 2018-10-17 | Bayer CropScience Aktiengesellschaft | Herbicidal combinations for tolerant soybean cultures |
| WO2014083033A1 (en) | 2012-11-30 | 2014-06-05 | Bayer Cropsience Ag | Binary fungicidal or pesticidal mixture |
| WO2014083088A2 (en) | 2012-11-30 | 2014-06-05 | Bayer Cropscience Ag | Binary fungicidal mixtures |
| WO2014086759A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising biological control agents |
| WO2014086758A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising a biological control agent and an insecticide |
| WO2014086750A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising a biological control agent and an insecticide |
| WO2014086753A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising biological control agents |
| WO2014086749A2 (en) | 2012-12-03 | 2014-06-12 | Bayer Cropscience Ag | Composition comprising a biological control agent and an insecticide |
| WO2014090765A1 (en) | 2012-12-12 | 2014-06-19 | Bayer Cropscience Ag | Use of 1-[2-fluoro-4-methyl-5-(2,2,2-trifluoroethylsulfinyl)phenyl]-5-amino-3-trifluoromethyl)-1 h-1,2,4 tfia zole for controlling nematodes in nematode-resistant crops |
| WO2014095826A1 (en) | 2012-12-18 | 2014-06-26 | Bayer Cropscience Ag | Binary fungicidal and bactericidal combinations |
| WO2014122083A1 (en) | 2013-02-06 | 2014-08-14 | Bayer Cropscience Ag | Halogen-substituted pyrazol derivatives as pest-control agents |
| WO2014124375A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising gougerotin and a biological control agent |
| WO2014124379A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising a streptomyces-based biological control agent and an insecticide |
| WO2014124361A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising a streptomyces-based biological control agent and another biological control agent |
| WO2014124373A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising gougerotin and an insecticide |
| WO2014139897A1 (en) | 2013-03-12 | 2014-09-18 | Bayer Cropscience Ag | Use of dithiine-tetracarboximides for controlling bacterial harmful organisms in useful plants |
| WO2014170313A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Active compound combinations having insecticidal properties |
| WO2014170364A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Binary insecticidal or pesticidal mixture |
| WO2014202510A1 (en) | 2013-06-20 | 2014-12-24 | Bayer Cropscience Ag | Aryl sulfide derivatives and aryl sulfoxide derivatives as acaricides and insecticides |
| WO2014202505A1 (en) | 2013-06-20 | 2014-12-24 | Bayer Cropscience Ag | Aryl sulfide derivatives and aryl sulfoxide derivatives as acaricides and insecticides |
| WO2015004028A1 (en) | 2013-07-08 | 2015-01-15 | Bayer Cropscience Ag | Six-membered c-n-linked aryl sulfide derivatives and aryl sulfoxide derivatives as pest control agents |
| DE202014008418U1 (en) | 2014-02-19 | 2014-11-14 | Clariant International Ltd. | Low foaming agrochemical compositions |
| DE202014008415U1 (en) | 2014-02-19 | 2014-11-25 | Clariant International Ltd. | Aqueous adjuvant composition for increasing the effectiveness of electrolyte active substances |
| WO2015160620A1 (en) | 2014-04-16 | 2015-10-22 | Bayer Cropscience Lp | Compositions comprising ningnanmycin and an insecticide |
| WO2015160618A1 (en) | 2014-04-16 | 2015-10-22 | Bayer Cropscience Lp | Compositions comprising ningnanmycin and a biological control agent |
| US11425904B2 (en) | 2014-04-23 | 2022-08-30 | Clariant International Ltd. | Use of aqueous drift-reducing compositions |
| WO2016001129A1 (en) | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Improved insecticidal compositions |
| DE102014012022A1 (en) | 2014-08-13 | 2016-02-18 | Clariant International Ltd. | Organic ammonium salts of anionic pesticides |
| EP3556211A1 (en) | 2014-08-13 | 2019-10-23 | Clariant International Ltd | Organic ammonium salts of anionic pesticides |
| DE102014018274A1 (en) | 2014-12-12 | 2015-07-30 | Clariant International Ltd. | Sugar surfactants and their use in agrochemical compositions |
| US11700852B2 (en) | 2014-12-19 | 2023-07-18 | Clariant International Ltd | Aqueous electrolyte-containing adjuvant compositions, active ingredient-containing compositions and the use thereof |
| WO2016106063A1 (en) | 2014-12-22 | 2016-06-30 | Bayer Corpscience Lp | Method for using a bacillus subtilis or bacillus pumilus strain to treat or prevent pineapple disease |
| US11750794B2 (en) | 2015-03-24 | 2023-09-05 | Augmedics Ltd. | Combining video-based and optic-based augmented reality in a near eye display |
| US10920080B2 (en) | 2015-10-09 | 2021-02-16 | Clariant International Ltd. | N-Alkyl glucamine-based universal pigment dispersions |
| US10961484B2 (en) | 2015-10-09 | 2021-03-30 | Clariant International Ltd. | Compositions comprising sugar amine and fatty acid |
| WO2017186543A2 (en) | 2016-04-24 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Use of fluopyram and/or bacillus subtilis for controlling fusarium wilt in plants of the musaceae family |
| US11220603B2 (en) | 2016-05-09 | 2022-01-11 | Clariant International Ltd. | Stabilizers for silicate paints |
| WO2018019676A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Active compound combinations and methods to protect the propagation material of plants |
| US11766296B2 (en) | 2018-11-26 | 2023-09-26 | Augmedics Ltd. | Tracking system for image-guided surgery |
| US11801115B2 (en) | 2019-12-22 | 2023-10-31 | Augmedics Ltd. | Mirroring in image guided surgery |
| US11896445B2 (en) | 2021-07-07 | 2024-02-13 | Augmedics Ltd. | Iliac pin and adapter |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9125412B2 (en) | Stable insecticide compositions and methods for producing same | |
| EP2369921B1 (en) | Stable sulfoximine-insecticide compositions | |
| US8669278B2 (en) | Insecticidal N-substituted (6-haloalkylpyridin-3-yl)alkyl sulfoximines | |
| EP2338882B1 (en) | 2-((6-(Haloalkyl substituted))pyridin-3-yl)-tetrahydro-1H-1(lamba)4-thien-1-ylidenecyanamide derivatives and related compounds as pesticides and insecticides | |
| CA2661480C (en) | Insecticidal n-substituted (heteroaryl)cycloalkyl sulfoximines | |
| AU2014256417B2 (en) | Stable sulfoximine-insecticide compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980152713.7 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09795588 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 593168 Country of ref document: NZ |
|
| REEP | Request for entry into the european phase |
Ref document number: 2009795588 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009795588 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 4538/DELNP/2011 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2748132 Country of ref document: CA Ref document number: 13129462 Country of ref document: CO Ref document number: 11078100 Country of ref document: CO |
|
| ENP | Entry into the national phase |
Ref document number: 2009330658 Country of ref document: AU Date of ref document: 20091222 Kind code of ref document: A Ref document number: 2011543496 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2011/006909 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20117017454 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: a201109330 Country of ref document: UA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011131067 Country of ref document: RU |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI0923702 Country of ref document: BR |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: PI0923702 Country of ref document: BR Free format text: APRESENTAR, EM ATE 60 (SESSENTA) DIAS, DOCUMENTOS COMPROBATORIOS QUE EXPLIQUEM E REGULARIZEM A DIVERGENCIA NO NOME DO DEPOSITANTE/INVENTOR CONSTANTE NA PUBLICACAO INTERNACIONAL WO/2010/074747 DE 14/10/2010 COMO RAYMOND E BOUCHER JR E O CONSTANTE NO FORMULARIO 1.03 DA PETICAO INICIAL NO 020110067370 DE 27/06/2011 COMO RAYMOND E BOUCHER. |
|
| ENP | Entry into the national phase |
Ref document number: PI0923702 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110627 |