WO2010066840A1 - SPIRO AZEPANE-OXAZOLIDINONES AS Kv1.3 POTASSIUM CHANNEL BLOCKERS - Google Patents
SPIRO AZEPANE-OXAZOLIDINONES AS Kv1.3 POTASSIUM CHANNEL BLOCKERS Download PDFInfo
- Publication number
- WO2010066840A1 WO2010066840A1 PCT/EP2009/066836 EP2009066836W WO2010066840A1 WO 2010066840 A1 WO2010066840 A1 WO 2010066840A1 EP 2009066836 W EP2009066836 W EP 2009066836W WO 2010066840 A1 WO2010066840 A1 WO 2010066840A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- compounds
- diazaspiro
- oxa
- Prior art date
Links
- 108010027296 Kv1.3 Potassium Channel Proteins 0.000 title abstract description 11
- 102000018706 Kv1.3 Potassium Channel Human genes 0.000 title abstract description 11
- 239000003450 potassium channel blocker Substances 0.000 title abstract description 6
- 150000001875 compounds Chemical class 0.000 claims abstract description 117
- 238000000034 method Methods 0.000 claims abstract description 32
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 18
- -1 spiro azepane-oxazolidinones Chemical class 0.000 claims abstract description 17
- 238000003786 synthesis reaction Methods 0.000 claims abstract description 13
- 230000015572 biosynthetic process Effects 0.000 claims abstract description 10
- 208000023275 Autoimmune disease Diseases 0.000 claims abstract description 6
- 208000008589 Obesity Diseases 0.000 claims abstract description 6
- 201000004681 Psoriasis Diseases 0.000 claims abstract description 6
- 210000001744 T-lymphocyte Anatomy 0.000 claims abstract description 6
- 206010052779 Transplant rejections Diseases 0.000 claims abstract description 6
- 206010012601 diabetes mellitus Diseases 0.000 claims abstract description 6
- 230000002757 inflammatory effect Effects 0.000 claims abstract description 6
- 230000001404 mediated effect Effects 0.000 claims abstract description 6
- 201000006417 multiple sclerosis Diseases 0.000 claims abstract description 6
- 230000007823 neuropathy Effects 0.000 claims abstract description 6
- 201000001119 neuropathy Diseases 0.000 claims abstract description 6
- 235000020824 obesity Nutrition 0.000 claims abstract description 6
- 206010039073 rheumatoid arthritis Diseases 0.000 claims abstract description 6
- 150000003839 salts Chemical class 0.000 claims description 32
- 125000000217 alkyl group Chemical group 0.000 claims description 19
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 17
- 229910052736 halogen Inorganic materials 0.000 claims description 15
- 239000003814 drug Substances 0.000 claims description 13
- 150000002367 halogens Chemical class 0.000 claims description 13
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 11
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 11
- 239000000126 substance Substances 0.000 claims description 10
- 229910052799 carbon Inorganic materials 0.000 claims description 9
- 229910052739 hydrogen Inorganic materials 0.000 claims description 9
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 claims description 8
- 239000001257 hydrogen Substances 0.000 claims description 8
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 7
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 claims description 6
- 125000001153 fluoro group Chemical group F* 0.000 claims description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 6
- 125000001624 naphthyl group Chemical group 0.000 claims description 6
- 229910052757 nitrogen Inorganic materials 0.000 claims description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 6
- 125000006239 protecting group Chemical group 0.000 claims description 6
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 5
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 5
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 claims description 5
- 229910052805 deuterium Inorganic materials 0.000 claims description 5
- 229910052731 fluorine Inorganic materials 0.000 claims description 5
- 239000011737 fluorine Substances 0.000 claims description 5
- 230000008569 process Effects 0.000 claims description 5
- 150000001412 amines Chemical class 0.000 claims description 4
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 claims description 4
- 239000003795 chemical substances by application Substances 0.000 claims description 4
- 239000003937 drug carrier Substances 0.000 claims description 4
- 150000002576 ketones Chemical class 0.000 claims description 4
- 229960000549 4-dimethylaminophenol Drugs 0.000 claims description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 3
- 125000003277 amino group Chemical group 0.000 claims description 3
- 238000007098 aminolysis reaction Methods 0.000 claims description 3
- GMHPWGYTSXHHPI-UHFFFAOYSA-N azepan-4-one Chemical compound O=C1CCCNCC1 GMHPWGYTSXHHPI-UHFFFAOYSA-N 0.000 claims description 3
- 150000001721 carbon Chemical group 0.000 claims description 3
- 238000006798 ring closing metathesis reaction Methods 0.000 claims description 3
- MKJKAAXDFKYVOJ-QMHKHESXSA-N (5r)-9-[3,5-bis(trifluoromethyl)benzoyl]-3-[(1s)-1-phenylethyl]-1-oxa-3,9-diazaspiro[4.6]undecan-2-one Chemical compound C([C@]1(CN(C(O1)=O)[C@@H](C)C=1C=CC=CC=1)CC1)CCN1C(=O)C1=CC(C(F)(F)F)=CC(C(F)(F)F)=C1 MKJKAAXDFKYVOJ-QMHKHESXSA-N 0.000 claims description 2
- MKJKAAXDFKYVOJ-HJPURHCSSA-N (5s)-9-[3,5-bis(trifluoromethyl)benzoyl]-3-[(1s)-1-phenylethyl]-1-oxa-3,9-diazaspiro[4.6]undecan-2-one Chemical compound C([C@@]1(CN(C(O1)=O)[C@@H](C)C=1C=CC=CC=1)CC1)CCN1C(=O)C1=CC(C(F)(F)F)=CC(C(F)(F)F)=C1 MKJKAAXDFKYVOJ-HJPURHCSSA-N 0.000 claims description 2
- IJLAKLCXGHCORN-MHECFPHRSA-N 3-[(5r)-2-oxo-3-[(1s)-1-phenylethyl]-1-oxa-3,9-diazaspiro[4.6]undecane-9-carbonyl]benzonitrile Chemical compound C([C@]1(CN(C(O1)=O)[C@@H](C)C=1C=CC=CC=1)CC1)CCN1C(=O)C1=CC=CC(C#N)=C1 IJLAKLCXGHCORN-MHECFPHRSA-N 0.000 claims description 2
- UPULOMQHYQDNNT-UHFFFAOYSA-N 5h-1,3-oxazol-2-one Chemical group O=C1OCC=N1 UPULOMQHYQDNNT-UHFFFAOYSA-N 0.000 claims description 2
- 238000010511 deprotection reaction Methods 0.000 claims description 2
- 150000002431 hydrogen Chemical class 0.000 claims 2
- KYWIYKKSMDLRDC-UHFFFAOYSA-N methyl n-nonyl ketone Natural products CCCCCCCCCC(C)=O KYWIYKKSMDLRDC-UHFFFAOYSA-N 0.000 claims 1
- 239000000203 mixture Substances 0.000 abstract description 46
- 239000000543 intermediate Substances 0.000 abstract description 8
- 238000011282 treatment Methods 0.000 abstract description 5
- 230000001225 therapeutic effect Effects 0.000 abstract description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 22
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 18
- 239000000243 solution Substances 0.000 description 18
- 239000004480 active ingredient Substances 0.000 description 15
- 239000004615 ingredient Substances 0.000 description 15
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 13
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- 238000009472 formulation Methods 0.000 description 12
- 239000002904 solvent Substances 0.000 description 11
- 239000003643 water by type Substances 0.000 description 11
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- 239000013078 crystal Substances 0.000 description 8
- 125000001424 substituent group Chemical group 0.000 description 8
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 239000002585 base Substances 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- 239000007787 solid Substances 0.000 description 7
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 239000003153 chemical reaction reagent Substances 0.000 description 6
- 239000007789 gas Substances 0.000 description 6
- 125000004435 hydrogen atom Chemical class [H]* 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 6
- 238000000926 separation method Methods 0.000 description 6
- 239000003826 tablet Substances 0.000 description 6
- 238000004809 thin layer chromatography Methods 0.000 description 6
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 5
- 102000004257 Potassium Channel Human genes 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 239000003480 eluent Substances 0.000 description 5
- 230000005764 inhibitory process Effects 0.000 description 5
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 5
- 239000003921 oil Substances 0.000 description 5
- 235000019198 oils Nutrition 0.000 description 5
- 239000012044 organic layer Substances 0.000 description 5
- 239000011591 potassium Substances 0.000 description 5
- 229910052700 potassium Inorganic materials 0.000 description 5
- 108020001213 potassium channel Proteins 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 238000001228 spectrum Methods 0.000 description 5
- 238000005160 1H NMR spectroscopy Methods 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 4
- BDAGIHXWWSANSR-UHFFFAOYSA-N Formic acid Chemical compound OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 4
- 108010010803 Gelatin Proteins 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 4
- 102100034355 Potassium voltage-gated channel subfamily A member 3 Human genes 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 230000000903 blocking effect Effects 0.000 description 4
- 239000002775 capsule Substances 0.000 description 4
- 125000004432 carbon atom Chemical group C* 0.000 description 4
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 description 4
- 238000004807 desolvation Methods 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 229920000159 gelatin Polymers 0.000 description 4
- 239000008273 gelatin Substances 0.000 description 4
- 239000007903 gelatin capsule Substances 0.000 description 4
- 235000019322 gelatine Nutrition 0.000 description 4
- 235000011852 gelatine desserts Nutrition 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 230000003287 optical effect Effects 0.000 description 4
- 238000002953 preparative HPLC Methods 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 4
- 238000001851 vibrational circular dichroism spectroscopy Methods 0.000 description 4
- IGVKWAAPMVVTFX-BUHFOSPRSA-N (e)-octadec-5-en-7,9-diynoic acid Chemical compound CCCCCCCCC#CC#C\C=C\CCCC(O)=O IGVKWAAPMVVTFX-BUHFOSPRSA-N 0.000 description 3
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 3
- VRJHQPZVIGNGMX-UHFFFAOYSA-N 4-piperidinone Chemical compound O=C1CCNCC1 VRJHQPZVIGNGMX-UHFFFAOYSA-N 0.000 description 3
- 230000005526 G1 to G0 transition Effects 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 239000007995 HEPES buffer Substances 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 238000004587 chromatography analysis Methods 0.000 description 3
- 238000004440 column chromatography Methods 0.000 description 3
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 238000002425 crystallisation Methods 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- 125000005843 halogen group Chemical group 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 230000014759 maintenance of location Effects 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 150000005846 sugar alcohols Chemical class 0.000 description 3
- DLRRAILFCIAPOA-OQHSHRKDSA-N tert-butyl 3-[(1r)-1-phenylethyl]-2-oxa-3,9-diazaspiro[4.6]undecane-9-carboxylate Chemical compound C1ON([C@H](C)C=2C=CC=CC=2)CC21CCCN(C(=O)OC(C)(C)C)CC2 DLRRAILFCIAPOA-OQHSHRKDSA-N 0.000 description 3
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- 229940124597 therapeutic agent Drugs 0.000 description 3
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 description 3
- 239000008158 vegetable oil Substances 0.000 description 3
- ZGBXGJPUSCAJEA-UHFFFAOYSA-N 1-oxa-3,9-diazaspiro[4.6]undecan-2-one Chemical class O1C(=O)NCC11CCNCCC1 ZGBXGJPUSCAJEA-UHFFFAOYSA-N 0.000 description 2
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical class CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 2
- WZOBGZLLNVPBGT-JBZHPUCOSA-N 3-[(1r)-1-phenylethyl]-1-oxa-3,9-diazaspiro[4.6]undecan-2-one Chemical compound O1C(=O)N([C@H](C)C=2C=CC=CC=2)CC21CCCNCC2 WZOBGZLLNVPBGT-JBZHPUCOSA-N 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- 229920000945 Amylopectin Polymers 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 238000003775 Density Functional Theory Methods 0.000 description 2
- 241000792859 Enema Species 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 102000004310 Ion Channels Human genes 0.000 description 2
- 108090000862 Ion Channels Proteins 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 239000008186 active pharmaceutical agent Substances 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 238000003556 assay Methods 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 2
- 229940125782 compound 2 Drugs 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 230000002939 deleterious effect Effects 0.000 description 2
- 235000013681 dietary sucrose Nutrition 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000007920 enema Substances 0.000 description 2
- 229940095399 enema Drugs 0.000 description 2
- 150000002118 epoxides Chemical class 0.000 description 2
- 238000000105 evaporative light scattering detection Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 125000004404 heteroalkyl group Chemical group 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 238000002329 infrared spectrum Methods 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 238000013160 medical therapy Methods 0.000 description 2
- 230000028161 membrane depolarization Effects 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 230000007935 neutral effect Effects 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 125000004430 oxygen atom Chemical group O* 0.000 description 2
- 238000007911 parenteral administration Methods 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 238000005070 sampling Methods 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 229910000104 sodium hydride Inorganic materials 0.000 description 2
- 239000012453 solvate Substances 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 229960004793 sucrose Drugs 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- PZWHCFGHKOCPQN-QRIPLOBPSA-N tert-butyl 4-hydroxy-4-[[[(1r)-1-phenylethyl]amino]methyl]azepane-1-carboxylate Chemical compound N([C@H](C)C=1C=CC=CC=1)CC1(O)CCCN(C(=O)OC(C)(C)C)CC1 PZWHCFGHKOCPQN-QRIPLOBPSA-N 0.000 description 2
- BPLKQGGAXWRFOE-UHFFFAOYSA-M trimethylsulfoxonium iodide Chemical compound [I-].C[S+](C)(C)=O BPLKQGGAXWRFOE-UHFFFAOYSA-M 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- RQEUFEKYXDPUSK-SSDOTTSWSA-N (1R)-1-phenylethanamine Chemical compound C[C@@H](N)C1=CC=CC=C1 RQEUFEKYXDPUSK-SSDOTTSWSA-N 0.000 description 1
- RQEUFEKYXDPUSK-ZETCQYMHSA-N (1S)-1-phenylethanamine Chemical compound C[C@H](N)C1=CC=CC=C1 RQEUFEKYXDPUSK-ZETCQYMHSA-N 0.000 description 1
- SHAHPWSYJFYMRX-GDLCADMTSA-N (2S)-2-(4-{[(1R,2S)-2-hydroxycyclopentyl]methyl}phenyl)propanoic acid Chemical compound C1=CC([C@@H](C(O)=O)C)=CC=C1C[C@@H]1[C@@H](O)CCC1 SHAHPWSYJFYMRX-GDLCADMTSA-N 0.000 description 1
- VUDZSIYXZUYWSC-DBRKOABJSA-N (4r)-1-[(2r,4r,5r)-3,3-difluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-4-hydroxy-1,3-diazinan-2-one Chemical compound FC1(F)[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)N[C@H](O)CC1 VUDZSIYXZUYWSC-DBRKOABJSA-N 0.000 description 1
- 0 **(CCC1)CCC1(CN1*)OC1=O Chemical compound **(CCC1)CCC1(CN1*)OC1=O 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- KQZLRWGGWXJPOS-NLFPWZOASA-N 1-[(1R)-1-(2,4-dichlorophenyl)ethyl]-6-[(4S,5R)-4-[(2S)-2-(hydroxymethyl)pyrrolidin-1-yl]-5-methylcyclohexen-1-yl]pyrazolo[3,4-b]pyrazine-3-carbonitrile Chemical compound ClC1=C(C=CC(=C1)Cl)[C@@H](C)N1N=C(C=2C1=NC(=CN=2)C1=CC[C@@H]([C@@H](C1)C)N1[C@@H](CCC1)CO)C#N KQZLRWGGWXJPOS-NLFPWZOASA-N 0.000 description 1
- 238000004293 19F NMR spectroscopy Methods 0.000 description 1
- WZOBGZLLNVPBGT-KNVGNIICSA-N 3-[(1s)-1-phenylethyl]-1-oxa-3,9-diazaspiro[4.6]undecan-2-one Chemical compound O1C(=O)N([C@@H](C)C=2C=CC=CC=2)CC21CCCNCC2 WZOBGZLLNVPBGT-KNVGNIICSA-N 0.000 description 1
- IJLAKLCXGHCORN-UUOWRZLLSA-N 3-[(5s)-2-oxo-3-[(1s)-1-phenylethyl]-1-oxa-3,9-diazaspiro[4.6]undecane-9-carbonyl]benzonitrile Chemical compound C([C@@]1(CN(C(O1)=O)[C@@H](C)C=1C=CC=CC=1)CC1)CCN1C(=O)C1=CC=CC(C#N)=C1 IJLAKLCXGHCORN-UUOWRZLLSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 244000105624 Arachis hypogaea Species 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- PTHCMJGKKRQCBF-UHFFFAOYSA-N Cellulose, microcrystalline Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC)C(CO)O1 PTHCMJGKKRQCBF-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- 244000020551 Helianthus annuus Species 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- RSJKGSCJYJTIGS-UHFFFAOYSA-N N-undecane Natural products CCCCCCCCCCC RSJKGSCJYJTIGS-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 239000007832 Na2SO4 Substances 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000005662 Paraffin oil Substances 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 238000001237 Raman spectrum Methods 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 235000019486 Sunflower oil Nutrition 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000012345 acetylating agent Substances 0.000 description 1
- 238000005903 acid hydrolysis reaction Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000001464 adherent effect Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 238000011949 advanced processing technology Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 150000001414 amino alcohols Chemical class 0.000 description 1
- 239000010775 animal oil Substances 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- WFTRLIZPJMFJER-UHFFFAOYSA-N azepan-1-ium-4-one;chloride Chemical compound Cl.O=C1CCCNCC1 WFTRLIZPJMFJER-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- JUHORIMYRDESRB-UHFFFAOYSA-N benzathine Chemical compound C=1C=CC=CC=1CNCCNCC1=CC=CC=C1 JUHORIMYRDESRB-UHFFFAOYSA-N 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- PASDCCFISLVPSO-UHFFFAOYSA-N benzoyl chloride Chemical compound ClC(=O)C1=CC=CC=C1 PASDCCFISLVPSO-UHFFFAOYSA-N 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000012267 brine Substances 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 125000004181 carboxyalkyl group Chemical group 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- PBAYDYUZOSNJGU-UHFFFAOYSA-N chelidonic acid Natural products OC(=O)C1=CC(=O)C=C(C(O)=O)O1 PBAYDYUZOSNJGU-UHFFFAOYSA-N 0.000 description 1
- 239000013626 chemical specie Substances 0.000 description 1
- 239000007910 chewable tablet Substances 0.000 description 1
- 238000010568 chiral column chromatography Methods 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 238000011097 chromatography purification Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 238000010668 complexation reaction Methods 0.000 description 1
- 229940125773 compound 10 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 229940125877 compound 31 Drugs 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 239000002178 crystalline material Substances 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 229940097362 cyclodextrins Drugs 0.000 description 1
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- YMWUJEATGCHHMB-DICFDUPASA-N deuterated dichloromethane Substances [2H]C([2H])(Cl)Cl YMWUJEATGCHHMB-DICFDUPASA-N 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- MVPICKVDHDWCJQ-UHFFFAOYSA-N ethyl 3-pyrrolidin-1-ylpropanoate Chemical compound CCOC(=O)CCN1CCCC1 MVPICKVDHDWCJQ-UHFFFAOYSA-N 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 235000019197 fats Nutrition 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000003818 flash chromatography Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 238000001640 fractional crystallisation Methods 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 230000014509 gene expression Effects 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 229940049906 glutamate Drugs 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 229940060367 inert ingredients Drugs 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 229940030980 inova Drugs 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 238000003819 low-pressure liquid chromatography Methods 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 125000002960 margaryl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 229960003194 meglumine Drugs 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000003094 microcapsule Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 230000009251 neurologic dysfunction Effects 0.000 description 1
- 230000007658 neurological function Effects 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-M octanoate Chemical compound CCCCCCCC([O-])=O WWZKQHOCKIZLMA-UHFFFAOYSA-M 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 229940049964 oleate Drugs 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 125000005429 oxyalkyl group Chemical group 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000003182 parenteral nutrition solution Substances 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- 235000019371 penicillin G benzathine Nutrition 0.000 description 1
- 125000002958 pentadecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 230000003285 pharmacodynamic effect Effects 0.000 description 1
- 238000001050 pharmacotherapy Methods 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 239000002985 plastic film Substances 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000000634 powder X-ray diffraction Methods 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000002685 pulmonary effect Effects 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000000452 restraining effect Effects 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 229910052701 rubidium Inorganic materials 0.000 description 1
- IGLNJRXAVVLDKE-UHFFFAOYSA-N rubidium atom Chemical compound [Rb] IGLNJRXAVVLDKE-UHFFFAOYSA-N 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- YGSDEFSMJLZEOE-UHFFFAOYSA-M salicylate Chemical compound OC1=CC=CC=C1C([O-])=O YGSDEFSMJLZEOE-UHFFFAOYSA-M 0.000 description 1
- 229960001860 salicylate Drugs 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 238000007493 shaping process Methods 0.000 description 1
- 238000002603 single-photon emission computed tomography Methods 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229940045902 sodium stearyl fumarate Drugs 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 238000000279 solid-state nuclear magnetic resonance spectrum Methods 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 229940114926 stearate Drugs 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000000707 stereoselective effect Effects 0.000 description 1
- 239000008223 sterile water Substances 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 239000002600 sunflower oil Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 238000011285 therapeutic regimen Methods 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000004001 thioalkyl group Chemical group 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 125000002889 tridecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000019871 vegetable fat Nutrition 0.000 description 1
- 238000002460 vibrational spectroscopy Methods 0.000 description 1
- 230000004304 visual acuity Effects 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 238000002424 x-ray crystallography Methods 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/10—Spiro-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
Definitions
- Embodiments of the present invention relate to, and provide spiro azepane-oxazolidinones (1-oxa-3,8-diazaspiro- [4.6]undecan-2-ones) as voltage gated Kv1.3 potassium channel blockers, and intermediates, formulations and methods.
- Blockers of voltage gated Kv1.3 potassium channels based on the benzamidomethylene- cyclohexyl scaffold were disclosed in WO 00/25786 ⁇ Merck & Co., 2000), by Schmalhofer et al. ⁇ Biochemistry, 41, 7781-7794, 2002), by Baell ⁇ Expert Opin. Ther. Patents, 15(9), 1209-1220, 2005) and Harvey ⁇ J. Med. Chem., 49(4), 1433-1441, 2006).
- substituted spiro azepane-oxazolidinones (1-oxa-3,8-diazaspiro[4.6]undecan- 2-ones) are novel voltage gated Kv1.3 potassium channel blockers.
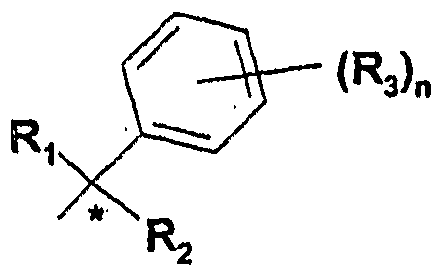
- This invention relates to a compound of formula (1 ), or a tautomer, stereoisomer, or a pharmacologically acceptable salt of any of the foregoing,
- R 1 and R 2 independently are hydrogen, deuterium, fluorine, CF 3 or alkyl(Ci -3 ), unsubstituted or substituted with one or more fluorine atoms, n is 0 (zero), 1 or 2,
- R 3 is chosen from halogen, alkyl(C 1-3 ), CF 3 , CN, NH 2 , NHAc, OH, OCH 3 , or OCF 3 - m is O (zero), 1 , 2 or 3,
- R 4 is chosen from halogen, alkyl(C 1-3 ), CF 3 , CN, NH 2 , NHAc, OH, OCH 3 , or OCF 3 , or
- the invention also relates, in some embodiments, to a compound of formula (1 ), or a tautomer, stereoisomer, or a pharmacologically acceptable salt of any of the foregoing, wherein R 1 and R 2 independently are hydrogen or methyl, n is 0 (zero) or 1 , R 3 is halogen, m is 1 or 2, and R 4 is chosen from halogen, CF 3 , CN, OCH 3, or OCF 3 , or (R 4 ) m and the phenyl ring to which it is attached form a naphthyl group.
- R 1 and R 2 independently are hydrogen, deuterium, fluorine, CF 3 or alkyl(C 1-3 ), unsubstituted or substituted with on or more fluorine atoms; n is 0 (zero), 1 or 2; R 3 is chosen from halogen, alkyl(C 1-3 ), CF 3 , CN, NH 2 , NHAc, OH, OCH 3 , or OCF 3 ; m is 0 (zero), 1 , 2 or 3; R 4 is chosen from halogen, alkyl(C 1-3 ), CF 3 , CN, NH 2 , NHAc, OH, OCH 3, or OCF 3 , or (R 4 ) m and the phenyl ring to which it is attached form a naphthyl group.
- the invention also relates, in some embodiments, to a compound of formula (1 ), or a tautomer, stereoisomer, or a pharmacologically acceptable salt of any of the foregoing, wherein R 1 and R 2 independently are hydrogen or methyl, n is O (zero) or 1 , R 3 is halogen, m is 1 or 2, and R 4 is chosen from halogen, CF 3 , CN, OCH 3, or OCF 3 , or (R 4 ) m and the phenyl ring to which it is attached form a naphthyl group.
- the invention also relates, in some embodiments, to a compound of formula (1 ) or a pharmacologically acceptable salt thereof, wherein the carbon atom attached to the nitrogen atom of the oxazolinone ring is the (R) or (S) enantiomer.
- the invention also relates, in some embodiments, to a compound of formula (1 ) or a pharmaco- logically acceptable salt thereof, wherein the central quaternary spiro-carbon atom is the (R) or (S) enantiomer.
- R 1 , R 2 , n and R3 have the meanings as given above, such compounds being useful in the synthesis of compounds of formula (1 ).
- Another embodiment provides a process to prepare compounds of formula (1 ), comprising the steps of:
- R 1 and R 2 independently are hydrogen, deuterium, fluorine, CF 3 or alkyl(Ci_ 3 ), unsubstituted or substituted with on or more fluorine atoms; n is 0 (zero), 1 or 2; R 3 is chosen from halogen, alkyl(C 1-3 ), CF 3 , CN, NH 2 , NHAc, OH, OCH 3 , or OCF 3 ; m is 0 (zero), 1 , 2 or 3, to yield an aminoalcohol of formula 5:
- a medicament comprising a compound of formula (1 ), or a pharmacologically acceptable salt thereof.
- the invention also relates, in some embodiments, to a pharmaceutical composition
- a pharmaceutical composition comprising, at least one pharmaceutically acceptable carrier, or at least one pharmaceutically acceptable auxiliary substance, or a combination of two or more thereof; and a pharmacologically active amount of at least one compound of formula (1 ), or a pharmacologically acceptable salt thereof.
- Further embodiments provide one the use of a compound of formula (1 ), to prepare a pharmaceutical composition for treating diabetes, psoriasis, obesity, transplant rejection, and inflammatory neuropathies, including T-cell mediated autoimmune diseases such as rheumatoid arthritis and multiple sclerosis.
- inventions include: methods for treating diabetes, psoriasis, obesity, transplant rejection, and inflammatory neuropathies, including T-cell mediated autoimmune diseases such as rheumatoid arthritis and multiple sclerosis, the methods comprising administering to a patient in need of such treating a compound of formula (1 ).
- methods of blocking Kv1.3 potassium channels comprising administering to a subject in need thereof, an pharmaceutically effective amount of a compound of formula (1 );
- the invention further relates to combination therapies comprising a compound of formula (1 ), or a pharmaceutical composition or formulation comprising a compound of formula (1 ), is administered concurrently or sequentially or as a combined preparation with another therapeutic agent or agents, for treating one or more of the conditions listed above.
- Such other therapeutic agent(s) may be administered prior to, simultaneously with, or following the administration of the compounds of the invention.
- the invention also provides compounds, pharmaceutical compositions, kits and methods for treating diabetes, psoriasis, obesity, transplant rejection, and inflammatory neuropathies, including T-cell mediated autoimmune diseases such as rheumatoid arthritis and multiple sclerosis, the method comprising administering to a patient in need of such treating a compound of formula (1 ).
- the compounds of the invention possess Kv1.3 potassium channel blocking activity.
- the inhibiting activities of the compounds of the invention can be readily demonstrated, for example, using one or more of the assays described herein or known in the art.
- the invention also provides methods of preparing the compounds of the invention and the intermediates used in those methods.
- Isolation and purification of the compounds and intermediates described herein can be affected, if desired, by any suitable separation or purification procedure such as, for example, filtration, extraction, crystallization, column chromatography, thin-layer chromatography, thick- layer chromatography, preparative low or high-pressure liquid chromatography, or a combination of these procedures.
- suitable separation and isolation procedures can be taken from the preparations and examples. However, other equivalent separation or isolation procedures could, of course, also be used.
- the compounds of the present invention may contain one or more asymmetric centers and can thus occur as racemates and racemic mixtures, single enantiomers, diastereomeric mixtures and individual diastereomers.
- All compounds of the present invention do contain at least one chiral center at their quaternary spiro-carbon atom.
- a chiral with unknown absolute chirality is named 'conf 1 ' or 'configuration 1 ', and the other enantiomer 'conf 2' or 'configuration 2'.
- 'Conf(iguration) V and 'conf(iguration) 2' can correlate to the (R) or the (S) assignments respectively, on conversely to the (S) or (R).
- the molecule can have additional asymmetric centers. Each such asymmetric center will independently produce two optical isomers. All of the possible optical isomers, enantiomers and diastereomers, in mixtures and as pure or partially purified compounds, belong to this invention. The present invention comprehends all such isomeric forms of these compounds.
- Formula (1 ) shows the structure of the class of compounds without preferred stereochemistry. The independent syntheses of these optical isomers, or their chromatographic separations, may be achieved as known in the art by appropriate modification of the methodology disclosed therein.
- Racemic mixtures of the compounds can be separated into the individual enantiomers by methods well-known in the art, such as the coupling of a racemic mixture of compounds to an enantiomerically pure compound to form a diastereomeric mixture, followed by separation of the individual diastereomers by standard methods, such as fractional crystallization or chromatography.
- the racemic mixture of the compounds can also be separated directly by chromatographic methods utilizing chiral stationary phases, which are well-known in the art.
- any enantiomer of a compound may be obtained by stereoselective synthesis using optically pure starting materials or reagents of known configuration by methods well-known in the art.
- Cis and trans isomers of the compound of formula (1 ), or a pharmaceutically acceptable salt thereof, also belong to the invention, and this also applies to tautomers of the compounds of formula (1 ).
- the compounds of the invention may also be used as reagents or standards in the biochemical study of neurological function, dysfunction and disease.
- alkyl denotes a univalent saturated, branched or straight, hydrocarbon chain. Unless otherwise stated, such chains can contain from 1 to 18 carbon atoms.
- alkyl groups are methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, te/f-butyl, pentyl, isopentyl, neopentyl, hexyl, isohexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, etc.
- the alkyl group When qualified 'lower', the alkyl group will contain from 1 to 6 carbon atoms. The same carbon content applies to the parent term 'alkane', and to derivative terms such as 'alkoxy'.
- the carbon content of various hydrocarbon containing moieties is indicated by a prefix designating the minimum and maximum number of carbon atoms in the moiety, i.e., the prefix C x-y defines the number of carbon atoms present from the integer "x" to the integer "y” inclusive.
- ⁇ lkyl(Ci -3 )' for example, includes methyl, ethyl, n-propyl or isopropyl
- 'alkyl(Ci -4 )' includes 'methyl, ethyl, n-propyl, isopropyl, n-butyl, 2-butyl, isobutyl or tert-butyl'.
- 'Halo' or 'Halogen' refers to chloro, fluoro, bromo or iodo;
- 'hetero' as in 'heteroalkyl, heteroaromatic', etc. includes containing one or more N, O or S atoms, 'heteroalkyl' includes alkyl groups with heteroatoms in any position, thus including N-bound O-bound or S-bound alkyl groups.
- substituted means that the specified group or moiety bears one or more substituents. Where any group may carry multiple substituents, and a variety of possible substituents can be provided, the substituents are independently selected, and need not to be the same.
- unsubstituted means that the specified group bears no substituents.
- substituents the term “independently” means that when more than one of such substituents are possible, they may be the same or different from each other.
- amino refers to a nitrogen atom that may be either terminal, or a linker between two other groups, wherein the group may be a primary, secondary or tertiary (two hydrogen atoms bonded to the nitrogen atom, one hydrogen atom bonded to the nitrogen atom and no hydrogen atoms bonded to the nitrogen atom, respectively) amine.
- the terms 'compound' or 'compounds' include tautomers, stereoisomers, or pharmacologically acceptable salts, also when not explicitly mentioned.
- 'Form' is a term encompassing all solids: polymorphs, solvates, amorphous forms.
- 'Crystal form' refers to various solid forms of the same compound, for example polymorphs, solvates and amorphous forms.
- 'Cocrystals' are multicomponent crystals with a unique lattice: new chemical species produced with neutral compounds.
- 'Amorphous forms' are non-crystalline materials with no long range order, and generally do not give a distinctive powder X-ray diffraction pattern.
- Different polymorphs usually have different X-ray diffraction patterns, solid state NMR spectra, infrared or Raman spectra, melting points, density, hardness, crystal shape, optical and electrical properties, stability, and solubility. Recrystallization solvent, rate of crystallization, storage temperature, and other factors may cause one crystal form to dominate.
- the present invention provides a pharmaceutical composition comprising at least one compound of formula (1 ), at least one pharmaceutically acceptable salt thereof, or a mixture of any of the foregoing, together with one or more pharmaceutically acceptable carriers thereof, and with or without one or more other therapeutic ingredients.
- the carrier(s) must be 'acceptable' in the sense of being compatible with the other ingredients of the formulation and not deleterious to the recipient thereof.
- composition encompasses a product comprising specified ingredients in predetermined amounts or proportions, as well as any product that results, directly or indirectly, from combining specified ingredients in specified amounts.
- this term encompasses a product comprising one or more active ingredients, and an optional carrier comprising inert ingredients, as well as any product that results, directly or indirectly, from combination, complexation or aggregation of any two or more of the ingredients, or from dissociation of one or more of the ingredients, or from other types of reactions or interactions of one or more of the ingredients.
- compositions are prepared by uniformly and intimately bringing the active ingredient into association with a liquid carrier or a finely divided solid carrier or both, and then, if necessary, shaping the product into the desired formulation.
- the pharmaceutical composition includes enough of the active object compound to produce the desired effect upon the progress or condition of diseases.
- the pharmaceutical compositions of the present invention encompass any composition made by admixing a compound of the present invention and a pharmaceutically acceptable carrier.
- pharmaceutically acceptable it is meant the carrier, diluent or excipient must be compatible with the other ingredients of the formulation and not deleterious to the recipient thereof.
- the term 'combination preparation' comprises both true combinations, meaning a compound of formula (1 ) and one or more other medicaments physically combined in one preparation such as a tablet or injection fluid, as well as 'kit-of-parts', comprising a compound of formula (1 ) and one or more other medicaments in separate dosage forms, together with instructions for use, with or without further means for facilitating compliance with the administration of the component compounds, e.g. label or drawings.
- the pharmacotherapy by definition is simultaneous.
- the contents of 'kit-of-parts' can be administered either simultaneously or at different time intervals.
- the potency of the compounds of the invention as inhibitors of voltage gated Kv1.3 channels was determined below. From the potency measured for a given compound of formula (1 ), one can estimate a theoretical lowest effective dose. At a concentration of the compound equal to twice the measured inhibition constant, nearly 100% of the Kv1.3 channels will be blocked by the compound. By converting that concentration to mg of compound per kg of patient one obtains a theoretical lowest effective dose, assuming ideal bioavailability.
- the typical daily dose of the active ingredients varies within a wide range and will depend on various factors such as the relevant indication, the route of administration, the age, weight and sex of the patient, and may be determined by a physician.
- total daily dose administration to a patient in single or individual doses may be in amounts, for example, from 0.001 to 10 mg/kg body weight daily, and more usually from 0.01 to 1 ,000 mg per day, of total active ingredients.
- Such dosages will be administered to a patient in need of treatment from one to three times each day, or as often as needed for efficacy, and for periods of at least two months, more typically for at least six months, or chronically.
- terapéuticaally effective amount refers to an amount of a therapeutic agent to treat a condition treatable by administrating a composition of the invention. That amount includes the amount sufficient to exhibit a detectable therapeutic or ameliorative response in a tissue system, animal or human. The effect may include, for example, treating the conditions listed herein.
- the precise pharmaceutically effective amount for a subject will depend upon the subject's size and health, the nature and extent of the condition being treated, recommendations of the treating physician (researcher, veterinarian, medical doctor or other clinician), and the therapeutics, or combination of therapeutics, selected for administration. Thus, it is not useful to specify an exact pharmaceutically effective amount in advance.
- a “pharmaceutical salt' refers to an acid:base complex containing an active pharmaceutical ingredient (API) along with additional non-toxic molecular species in the same crystal structure.
- pharmaceutically acceptable salt refers to those salts that are, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and lower animals without undue toxicity, irritation, allergic response, etc., and are commensurate with a reasonable benefit/risk ratio.
- Pharmaceutically acceptable salts are well-known in the art. They can be prepared in situ when finally isolating and purifying the compounds of the invention, or separately by reacting them with pharmaceutically acceptable non-toxic bases or acids, including inorganic or organic bases and inorganic or organic acids (Berge, S.
- Common cations used as counterions in pharmaceutically acceptable salts include: sodium, potassium, calcium, magnesium, lithium, zinc, aluminum, arginine, lysine, histidine, triethylamine, ethanolamine, triethanolamine, ethilenediamine, meglumine, procaine and benzathine.
- the 'free base' form may be regenerated by contacting the salt with a base or acid, and isolating the parent compound in the conventional matter.
- the parent form of the compound differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but otherwise the salts are equivalent to the parent form of the compound for the purposes of the present invention.
- treatment refers to any treatment of a human condition or disease, and includes: (1 ) inhibiting the disease or condition, i.e., arresting its development, (2) relieving the disease or condition, i.e., causing the condition to regress, or (3) stopping the symptoms of the disease.
- the term 'inhibit' includes its generally accepted meaning which includes restraining, alleviating, ameliorating, and slowing, stopping or reversing progression, severity, or a resultant symptom.
- medical therapy intendeds to include diagnostic and therapeutic regimens carried out in vivo or ex vivo on humans.
- Nuclear magnetic resonance spectra ( 1 H NMR and 13 C NMR, APT) were determined in the indicated solvent using a Bruker ARX 400 ( 1 H: 400 MHz, 13 C: 100 MHz) at 300 K, unless indicated otherwise.
- 19 F NMR and 13 C NMR experiments were carried out on a Varian Inova 500 spectrometer operating at 1 1.74 T (499.9 MHz for 1 H; 125.7 MHz for 13 C; 50.7 Mhz, 470.4 MHz for 19 F) using a 5 mm SW probe.
- the spectra were determined in deuterated chloroform or dichloromethane obtained from Cambridge Isotope Laboratories Ltd.
- Flash chromatography refers to purification using the indicated eluent and silica gel (either
- Reactions were monitored by using thin-layer chromatography (TLC) on silica coated plastic sheets (Merck precoated silica gel 60 F254) with the indicated eluent. Spots were visualised by UV light (254 nm) or I 2 .
- TLC thin-layer chromatography
- Mass spectra and accurate masses were measured with a JEOL JMS-SX/SX 102 A Tandem Mass Spectrometer using Fast Atom Bombardement (FAB). A resolving power of 10,000 (10% valley definition) for high resolution FAB mass spectrometry was used.
- FAB Fast Atom Bombardement
- the LC-MS system consisted of a Waters 1525 ⁇ pump, connected to a Waters 2777 auto sampler.
- the LC methode was:
- the auto sampler had a 10 ⁇ l injection loop, the injection volume was 10 ⁇ l.
- the auto sampler was connected to a Waters Sunfire C18 30 * 4.6 mm column with 2.5 urn particles.
- the column was thermostated at room temperature (about 23 0 C).
- the column was connected to a Waters 2996 PDA.
- the wavelength was scanned from 240 to 320 nm.
- the resolution was 1.2 nm and the sampling rate was 20 Hz.
- the flow was split 1 :1 and connected to a Waters 2424 ELSD, having the following parameters: gas pressure: 40 psi; data rate 20 points/sec; gain 500; time constant 0.2 sec; nebulizer mode cooling; drift tube 50 0 C.
- V Multiplier
- CHO cells expressing either Kv1.3, Kv1.5 or hERG ion channels were cultured at 37°C/5% CO 2 . Prior to the experiment cells were seeded in 96 well plates (Corning, New York, USA) and cultured for 24h. Medium was discarded and replaced by 10O ⁇ l Rb buffer (1 OmM HEPES pH 7.4, 5mM Glucose, 5mM RbCI, 14OmM NaCI , 2mM CaCI 2 , 1 mM MgSO 4 ) per well.
- the supernatant was transferred in a 96 well plate and the Rb concentration was measured using a ZEEnit Atomic Absorption Spectrometer (Analytik Jena, Germany). By comparing control vs. depolarized w/o compound, the ion channel inhibition was determined. Measurements were done in triplicate.
- Hexahydro-(1 H)-azepin-4-one 1 can be synthesized from the commercially available piperidin-4- one as described (Roglans, A. et al., Synth. Commun. 22, 1249-1258, 1992; Ashwood, M.S. et al., J. Chem. Soc. Perkin Trans. I, 641-644, 1995).
- the amino group in 1 can be protected with a protecting group, such as the te/f-Butyloxycarboyl (t-Boc) or benzyloxycarbonyl (Cbz) group to give a compound of formula 2.
- such a protection is carried out in the presence of a base, such as aqueous sodium hydroxide or triethylamine in an inert organic solvent such as dichloromethane or methanol.
- a base such as aqueous sodium hydroxide or triethylamine
- the carbonyl moiety in compound 2 can be epoxidized to afford the epoxide 3, applying trimethyl sulfoxonium iodide in the presence of a base, such as sodium hydride.
- a base such as sodium hydride
- the title compounds can be prepared by aminolysis of the spiro-epoxide 3 with amines of general formula 4, followed by ring-closure of the formed aminoalcohols 5 using a carbonylating agent such as carbonyldiimidazole (CDI).
- a carbonylating agent such as carbonyldiimidazole (CDI).
- CDI carbonyldiimidazole
- This ring-closure reaction wherein a [4,6]diazaspiro- undecane spirocycle is formed — in general proceeds more sluggishly in comparison with the analogous reaction for the formation of [4.5]diazaspirodecanes ⁇ Caroon, J. M. et al., J. Med. Chem. 1981, 24, 1320-1328).
- traces of remaining starting material 4 are removed from the aminoalcohol 5 in order to increase the yields of the conversion of 5 to 6.
- the resulting spiro oxazolidinones 6 can be deprotected by acidic hydrolysis of the f-butyloxy carbonyl group, a or ng compoun s o genera ormu a .
- ompoun o genera ormu a can e reac e with a acetylating agent such as an acid chloride derivative of general formula 8 to give a compound of formula 9.
- a reaction is preferably carried out in an inert organic solvent such as acetonitrile in the presence of a base such as DIPEA or triethylami ⁇ e in order to scavenge the liberated hydrochloric acid.
- compositions may be obtained using procedures well-known in the art, for example by mixing a compound of the present invention with a suitable acid, for instance an inorganic acid or an organic acid.
- a suitable acid for instance an inorganic acid or an organic acid.
- EXAMPLE 4 SYNTHESES OF INTERMEDIATES t-Butyl hexahydr ⁇ ' 4-oxo-(4H)-azepine-1-carboxylate (2): hexahydro-(4H)-azepin-4-one monohydrochloride (30 g, 200 mmol) was suspended in methanol (200 mL) and cooled to. O 0 C. Sodium hydroxide (8.02g, 200 mmol) dissolved in water (20 ml) was added dropwise. Di-tert- butyl dicarbonate (Boc anhydride) (43.76g, 200 mmol) was added portionwise and the resulting solution was stirred for 16 hours.
- Boc anhydride Di-tert- butyl dicarbonate
- Compounds 6a and 6b are mixtures of two diastereoisomers that were separated by chiral column chromatography.
- the protocol (“Method A”) for the R-mixture 6a was different from the protocol (“Method B”) used for the S-mixture 6b.
- Detection was performed by using UV at a ⁇ of 220 nm. The separation was checked by a chiral analytical system based on Chiralpak AD-H ® , 5 ⁇ (250*4.6 mm) and the same mobile phase (detection UV, ⁇ 210 nm) @ 1 ml/min and room temperature. The retention times from the analytical system were 5.2 and 8.2 min respectively
- VCD Vibrational Circular Dichroism
- EXAMPLE 6 PHARMACEUTICAL PREPARATIONS
- compounds of formula (1 ) are formulated into pharmaceutical compositions, which are novel embodiments of the invention because they contain the compounds, more particularly specific compounds disclosed herein.
- Types of pharmaceutical compositions that may be used include: tablets, chewable tablets, capsules (including microcapsules), solutions, parenteral solutions, ointments (creams and gels), suppositories, suspensions, and other types disclosed herein, or are apparent to a person skilled in the art from the specification and general knowledge in the art.
- the active ingredient for instance, may also be in the form of an inclusion complex in cyclodextrins, their ethers or their esters.
- compositions are used for oral, intravenous, subcutaneous, tracheal, bronchial, intranasal, pulmonary, transdermal, buccal, rectal, parenteral or other ways to administer.
- the pharmaceutical formulation contains at least one compound of formula (1 ) in admixture with at least one pharmaceutically acceptable adjuvant, diluent and/or carrier.
- the total amount of active ingredients can be in the range of from about 0.1% (w/w) to about 95% (w/w) of the formulation, such as from 0.5% to 50% (w/w) and preferably from 1 % to 25% (w/w). In some embodiments, the amount of active ingredient can be greater than about 95% (w/w) or less than about 0.1 % (w/w).
- the compounds of the invention can be brought into forms suitable for administration by means of usual processes using auxiliary substances such as liquid or solid, powdered ingredients, such as the pharmaceutically customary liquid or solid fillers and extenders, solvents, emulsifiers, lubricants, flavorings, colorings and/or buffer substances.
- auxiliary substances such as liquid or solid, powdered ingredients, such as the pharmaceutically customary liquid or solid fillers and extenders, solvents, emulsifiers, lubricants, flavorings, colorings and/or buffer substances.
- auxiliary substances include magnesium carbonate, titanium dioxide, lactose, saccharose, sorbitol, mannitol and other sugars or sugar alcohols, talc, lactoprotein, gelatin, starch, amylopectin, cellulose and its derivatives, animal and vegetable oils such as fish liver oil, sunflower, groundnut or sesame oil, polyethylene glycol and solvents such as, for example, sterile water and mono- or polyhydric alcohols such as glycerol, as well as with disintegrating agents and lubricating agents such as magnesium stearate, calcium stearate, sodium stearyl fumarate and polyethylene glycol waxes.
- the mixture may then be processed into granules or pressed into tablets.
- a tablet can be prepared using the ingredients below:
- the components are blended and compressed to form tablets each weighing 230 mg.
- the active ingredients may be separately premixed with the other non-active ingredients, before being mixed to form a formulation.
- the active ingredients may also be mixed with each other, before being mixed with the non-active ingredients to form a formulation.
- Soft gelatin capsules may be prepared with capsules containing a mixture of the active ingredients of the invention, vegetable oil, fat, or other suitable vehicle for soft gelatin capsules.
- Hard gelatin capsules may contain granules of the active ingredients.
- Hard gelatin capsules may also contain the active ingredients together with solid powdered ingredients such as lactose, saccharose, sorbitol, mannitol, potato starch, corn starch, amylopectin, cellulose derivatives or gelatin.
- Dosage units for rectal administration may be prepared (i) in the form of suppositories that contain the active substance mixed with a neutral fat base; (ii) in the form of a gelatin rectal capsule that contains the active substance in a mixture with a vegetable oil, paraffin oil or other suitable vehicle for gelatin rectal capsules; (iii) in the form of a ready-made micro enema; or (iv) in the form of a dry micro enema formulation to be reconstituted in a suitable solvent just prior to administration.
- Liquid preparations may be prepared in the form of syrups, elixirs, concentrated drops or suspensions, e.g. solutions or suspensions containing the active ingredients and the remainder consisting, for example, of sugar or sugar alcohols and a mixture of ethanol, water, glycerol, propylene glycol and polyethylene glycol. If desired, such liquid preparations may contain coloring agents, flavoring agents, preservatives, saccharine and carboxymethyl cellulose or other thickening agents. Liquid preparations may also be prepared in the form of a dry powder, reconstituted with a suitable solvent prior to use. Solutions for parenteral administration may be prepared as a solution of a formulation of the invention in a pharmaceutically acceptable solvent.
- solutions for parenteral administration may also be prepared as a dry preparation, reconstituted with a suitable solvent before use.
- formulations and 'kits of parts' comprising one or more containers filled with one or more of the ingredients of a pharmaceutical composition of the invention, for use in medical therapy.
- container(s) can be various written materials such as instructions for use, or a notice in the form prescribed by a governmental agency regulating the manufacture, use or sale of pharmaceuticals products, which notice reflects approval by the agency of manufacture, use, or sale for human or veterinary administration.
- formulations of the present invention in the manufacture of medicaments for use in treating a condition in which blocking of voltage gated Kv1.3 potassium channel is required or desired, and methods of medical treatment, comprise the administration of a therapeutically effective total amount of at least one compound of formula (1 ) to a patient suffering from, or susceptible to, a condition in which blocking of voltage gated Kv1.3 potassium channel is required or desired.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Diabetes (AREA)
- Rheumatology (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Pain & Pain Management (AREA)
- Transplantation (AREA)
- Child & Adolescent Psychology (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Physical Education & Sports Medicine (AREA)
- Dermatology (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2009324358A AU2009324358A1 (en) | 2008-12-12 | 2009-12-10 | Spiro azepane-oxazolidinones as Kv1.3 potassium channel blockers |
| US13/133,042 US8575148B2 (en) | 2008-12-12 | 2009-12-10 | Spiro azepane-oxazolidinones as Kv1.3 potassium channel blockers |
| BRPI0922714A BRPI0922714A2 (en) | 2008-12-12 | 2009-12-10 | compound, process for preparing a compound, drug, use of a compound, and pharmaceutical composition |
| EP09765127A EP2376501A1 (en) | 2008-12-12 | 2009-12-10 | SPIRO AZEPANE-OXAZOLIDINONES AS Kv1.3 POTASSIUM CHANNEL BLOCKERS |
| CA2743557A CA2743557A1 (en) | 2008-12-12 | 2009-12-10 | Spiro azepane-oxazolidinones as kv1.3 potassium channel blockers |
| JP2011540106A JP2012511546A (en) | 2008-12-12 | 2009-12-10 | Spiroazepan-oxazolidinone as a Kv1.3 potassium channel blocker |
| MX2011006221A MX2011006221A (en) | 2008-12-12 | 2009-12-10 | SPIRO AZEPANE-OXAZOLIDINONES AS Kv1.3 POTASSIUM CHANNEL BLOCKERS. |
| EA201170801A EA201170801A1 (en) | 2008-12-12 | 2009-12-10 | SPYROAZEPANOXAZOLIDINONES AS KALIUS CHANNEL BLOCKERS Kv1.3 |
| CN2009801494792A CN102333782A (en) | 2008-12-12 | 2009-12-10 | Spiro azepane-oxazolidinones as kv1.3 potassium channel blockers |
| IL212948A IL212948A0 (en) | 2008-12-12 | 2011-05-17 | SPIRO AZEPANE-OXAZOLIDINONES AS Kv1.3 POTASSIUM CHANNEL BLOCKERS |
| ZA2011/04041A ZA201104041B (en) | 2008-12-12 | 2011-05-31 | Spiro azepane-oxazolidinones as kv1.3 potassium channel blockers |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12191308P | 2008-12-12 | 2008-12-12 | |
| EP08171421 | 2008-12-12 | ||
| EP08171421.4 | 2008-12-12 | ||
| US61/121,913 | 2008-12-12 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010066840A1 true WO2010066840A1 (en) | 2010-06-17 |
Family
ID=40513443
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/066836 WO2010066840A1 (en) | 2008-12-12 | 2009-12-10 | SPIRO AZEPANE-OXAZOLIDINONES AS Kv1.3 POTASSIUM CHANNEL BLOCKERS |
Country Status (20)
| Country | Link |
|---|---|
| US (1) | US8575148B2 (en) |
| EP (1) | EP2376501A1 (en) |
| JP (1) | JP2012511546A (en) |
| KR (1) | KR20110110165A (en) |
| CN (1) | CN102333782A (en) |
| AR (1) | AR074558A1 (en) |
| AU (1) | AU2009324358A1 (en) |
| BR (1) | BRPI0922714A2 (en) |
| CA (1) | CA2743557A1 (en) |
| CO (1) | CO6390036A2 (en) |
| CR (1) | CR20110302A (en) |
| DO (1) | DOP2011000164A (en) |
| EA (1) | EA201170801A1 (en) |
| EC (1) | ECSP11011118A (en) |
| IL (1) | IL212948A0 (en) |
| MX (1) | MX2011006221A (en) |
| PE (1) | PE20110772A1 (en) |
| TW (1) | TW201026708A (en) |
| WO (1) | WO2010066840A1 (en) |
| ZA (1) | ZA201104041B (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013052507A1 (en) * | 2011-10-03 | 2013-04-11 | The Regents Of The University Of California | TREATMENT OF OBESITY AND OBESITY-RELATED DISORDERS BY PHARMACOLOGICAL TARGETING OF Kv1.3 POTASSIUM CHANNELS |
| WO2016146583A1 (en) | 2015-03-13 | 2016-09-22 | 4Sc Discovery Gmbh | Kv1.3 inhibitors and their medical application |
| WO2016146575A1 (en) | 2015-03-13 | 2016-09-22 | 4Sc Discovery Gmbh | Kv1.3 inhibitors and their medical application |
| US9611232B2 (en) | 2012-10-29 | 2017-04-04 | Hoffmann-La Roche Inc. | Oxazolidinone and imidazolidinone compounds |
| WO2020252130A1 (en) * | 2019-06-12 | 2020-12-17 | Lieber Institute, Inc. | Kv11.1-3.1 inhibiting methods and compositions |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN109053751A (en) * | 2018-08-30 | 2018-12-21 | 成都海博锐药业有限公司 | FXR regulator with spirane structure |
| CN111574537B (en) * | 2020-05-20 | 2022-11-15 | 成都药明康德新药开发有限公司 | Synthesis method of tert-butyl-8-oxa-3,11-diazaspiro [5.6] dodecane-3-formylate |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000025786A1 (en) * | 1998-10-30 | 2000-05-11 | Merck & Co., Inc. | Heterocyclic potassium channel inhibitors |
-
2009
- 2009-12-09 AR ARP090104760A patent/AR074558A1/en unknown
- 2009-12-09 TW TW098142079A patent/TW201026708A/en unknown
- 2009-12-10 JP JP2011540106A patent/JP2012511546A/en active Pending
- 2009-12-10 US US13/133,042 patent/US8575148B2/en not_active Expired - Fee Related
- 2009-12-10 PE PE2011001198A patent/PE20110772A1/en not_active Application Discontinuation
- 2009-12-10 AU AU2009324358A patent/AU2009324358A1/en not_active Abandoned
- 2009-12-10 EP EP09765127A patent/EP2376501A1/en not_active Withdrawn
- 2009-12-10 KR KR1020117016047A patent/KR20110110165A/en not_active Application Discontinuation
- 2009-12-10 CA CA2743557A patent/CA2743557A1/en not_active Abandoned
- 2009-12-10 WO PCT/EP2009/066836 patent/WO2010066840A1/en active Application Filing
- 2009-12-10 EA EA201170801A patent/EA201170801A1/en unknown
- 2009-12-10 BR BRPI0922714A patent/BRPI0922714A2/en not_active IP Right Cessation
- 2009-12-10 MX MX2011006221A patent/MX2011006221A/en not_active Application Discontinuation
- 2009-12-10 CN CN2009801494792A patent/CN102333782A/en active Pending
-
2011
- 2011-05-17 IL IL212948A patent/IL212948A0/en unknown
- 2011-05-31 DO DO2011000164A patent/DOP2011000164A/en unknown
- 2011-05-31 ZA ZA2011/04041A patent/ZA201104041B/en unknown
- 2011-06-03 CR CR20110302A patent/CR20110302A/en not_active Application Discontinuation
- 2011-06-08 CO CO11070889A patent/CO6390036A2/en not_active Application Discontinuation
- 2011-06-09 EC EC2011011118A patent/ECSP11011118A/en unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000025786A1 (en) * | 1998-10-30 | 2000-05-11 | Merck & Co., Inc. | Heterocyclic potassium channel inhibitors |
Non-Patent Citations (3)
| Title |
|---|
| BAEL J B: "Potassium channel blockers as immunosuppressants", EXPERT OPINION ON THERAPEUTIC PATENTS 200509 GB, vol. 15, no. 9, September 2005 (2005-09-01), pages 1209 - 1220, XP002523188, ISSN: 1354-3776 * |
| HARVEY ANDREW J ET AL: "A new class of blockers of the voltage-gated potassium channel Kv1.3 via modification of the 4- or 7-position of khellinone.", JOURNAL OF MEDICINAL CHEMISTRY 23 FEB 2006, vol. 49, no. 4, 23 February 2006 (2006-02-23), pages 1433 - 1441, XP002523187, ISSN: 0022-2623 * |
| SCHMALHOFER WILLIAM A ET AL: "Identification of a new class of inhibitors of the voltage-gated potassium channel, Kv1.3, with immunosuppressant properties.", BIOCHEMISTRY 18 JUN 2002, vol. 41, no. 24, 18 June 2002 (2002-06-18), pages 7781 - 7794, XP002523189, ISSN: 0006-2960 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013052507A1 (en) * | 2011-10-03 | 2013-04-11 | The Regents Of The University Of California | TREATMENT OF OBESITY AND OBESITY-RELATED DISORDERS BY PHARMACOLOGICAL TARGETING OF Kv1.3 POTASSIUM CHANNELS |
| CN103987408A (en) * | 2011-10-03 | 2014-08-13 | 加州大学评议会 | Treatment of obesity and obesity-related disorders by pharmacological targeting of Kv1.3 potassium channels |
| US9878006B2 (en) | 2011-10-03 | 2018-01-30 | The Regents Of The University Of California | Treatment of obesity and obesity related disorders by pharmalogical targeting of Kv1.3 potassium channels |
| US9611232B2 (en) | 2012-10-29 | 2017-04-04 | Hoffmann-La Roche Inc. | Oxazolidinone and imidazolidinone compounds |
| WO2016146583A1 (en) | 2015-03-13 | 2016-09-22 | 4Sc Discovery Gmbh | Kv1.3 inhibitors and their medical application |
| WO2016146575A1 (en) | 2015-03-13 | 2016-09-22 | 4Sc Discovery Gmbh | Kv1.3 inhibitors and their medical application |
| WO2020252130A1 (en) * | 2019-06-12 | 2020-12-17 | Lieber Institute, Inc. | Kv11.1-3.1 inhibiting methods and compositions |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2012511546A (en) | 2012-05-24 |
| CR20110302A (en) | 2011-10-27 |
| IL212948A0 (en) | 2011-07-31 |
| AR074558A1 (en) | 2011-01-26 |
| EP2376501A1 (en) | 2011-10-19 |
| CN102333782A (en) | 2012-01-25 |
| CO6390036A2 (en) | 2012-02-29 |
| AU2009324358A1 (en) | 2011-06-23 |
| KR20110110165A (en) | 2011-10-06 |
| CA2743557A1 (en) | 2010-06-17 |
| BRPI0922714A2 (en) | 2016-01-05 |
| US8575148B2 (en) | 2013-11-05 |
| TW201026708A (en) | 2010-07-16 |
| ECSP11011118A (en) | 2011-07-29 |
| PE20110772A1 (en) | 2011-10-19 |
| ZA201104041B (en) | 2012-11-28 |
| EA201170801A1 (en) | 2011-12-30 |
| US20110237569A1 (en) | 2011-09-29 |
| DOP2011000164A (en) | 2011-06-30 |
| MX2011006221A (en) | 2011-06-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8575148B2 (en) | Spiro azepane-oxazolidinones as Kv1.3 potassium channel blockers | |
| US9139594B2 (en) | Fused aminodihydropyrimidone derivatives | |
| JP5651692B2 (en) | Condensed aminodihydro-oxazine derivatives | |
| JP2022549171A (en) | Fused pyridone compounds, and methods of preparation and uses thereof | |
| US20080300241A9 (en) | Tricyclic quinolinone and tricyclic quinoline androgen receptor modulator compounds and methods | |
| JP2016537369A (en) | Substituted 4,5,6,7-tetrahydropyrazolo [1,5-A] pyrazine derivatives as casein kinase 1D / E inhibitors | |
| US10851116B2 (en) | Bicyclic amines as novel JAK kinase inhibitors | |
| AU2017254266B2 (en) | Prodrug of amino acid derivative | |
| CA2959747C (en) | Compounds as crth2 antagonist and uses thereof | |
| AU2015315687A1 (en) | P2X7 modulators | |
| US20220087996A1 (en) | Histone acetylase p300 inhibitor and use thereof | |
| AU2018208516B2 (en) | Novel amino-imidazopyridine derivatives as Janus kinase inhibitors and pharmaceutical use thereof | |
| US20210253573A1 (en) | Method for preparing 2-[(3r)-3-methylmorpholin-4-yl]-4-[1-methyl-1h-pyrazol-5-yl)-8-(1h-pyrazol-5-yl)-1,7-naphthyridine | |
| EP3720855B1 (en) | Imidazopyridine derivatives and the use thereof as medicament | |
| US8729095B2 (en) | 3-(biaryloxy)propionic acid derivatives for prevention and/or treatment of thromboembolic diseases | |
| WO2022117678A1 (en) | Octahydroisoquinolinyl derivatives | |
| ES2968733T3 (en) | Morpholinyl, piperazinyl, oxazepanil and diazepanil derivatives useful as o-glycoprotein-2-acetamido-2-deoxy-3-d-glucopyranosidase inhibitors | |
| IL303036A (en) | Malt-1 modulators | |
| WO2011070298A1 (en) | Di-substituted 9h-pyrido[3,4-b]indole derivatives, preparation of same and therapeutic use thereof | |
| US20220213089A1 (en) | Inhibiting cyclic amp-responsive element-binding protein (creb) | |
| RU2803284C1 (en) | Benzimidazole derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980149479.2 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09765127 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12011500804 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 592496 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2743557 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 212948 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011540106 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2011-000302 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13133042 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11070889 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011001408 Country of ref document: CL Ref document number: 4001/CHENP/2011 Country of ref document: IN Ref document number: 001198-2011 Country of ref document: PE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2011/006221 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009765127 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2009324358 Country of ref document: AU Date of ref document: 20091210 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP2011000439 Country of ref document: DZ |
|
| ENP | Entry into the national phase |
Ref document number: 20117016047 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201170801 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: a201108790 Country of ref document: UA |
|
| ENP | Entry into the national phase |
Ref document number: PI0922714 Country of ref document: BR Kind code of ref document: A2 Effective date: 20110606 |