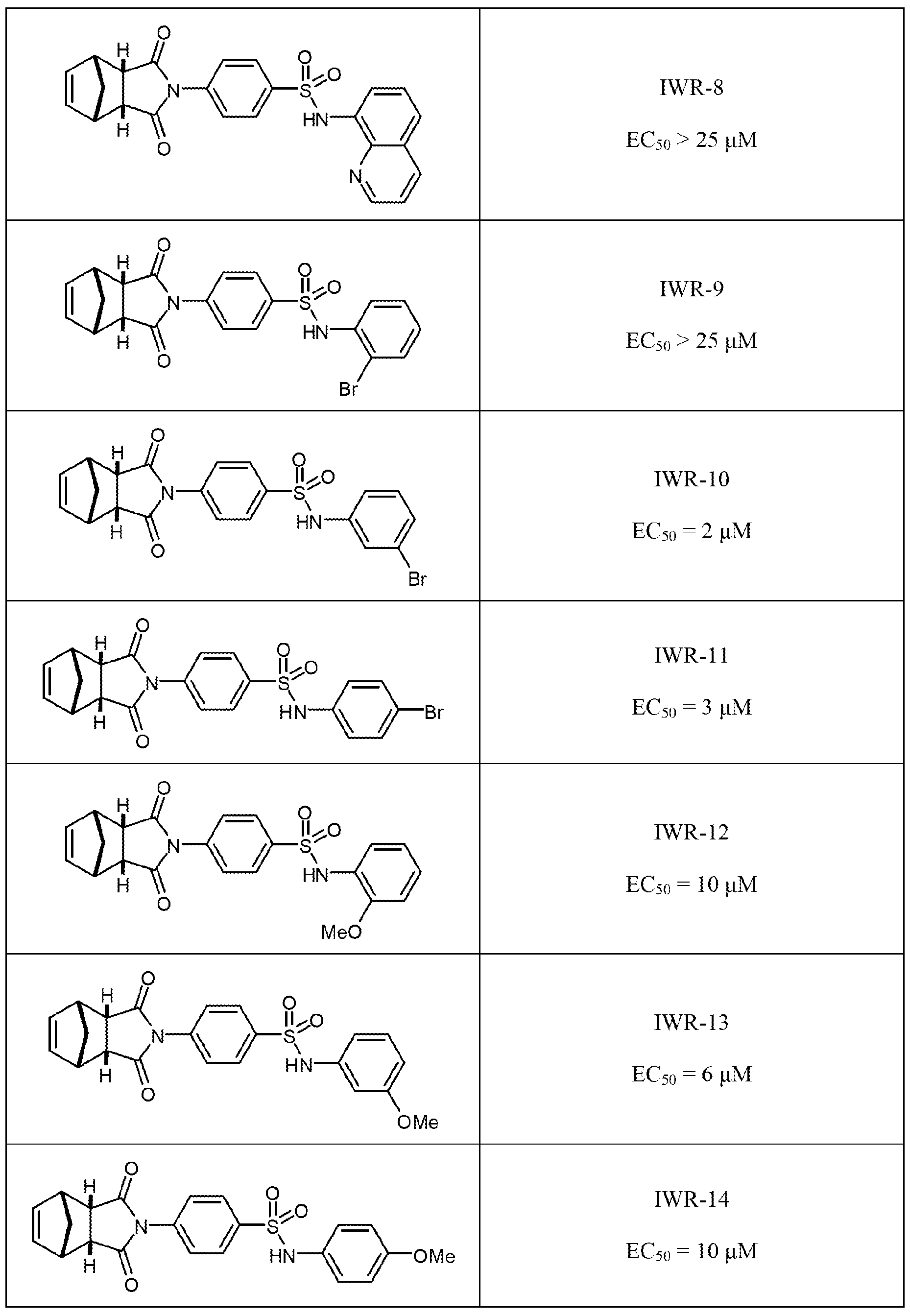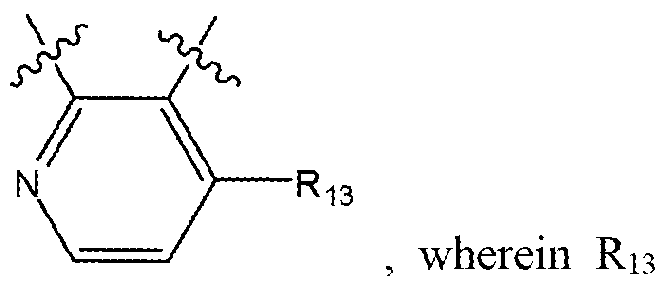WO2009155001A2 - Wnt protein signalling inhibitors - Google Patents
Wnt protein signalling inhibitors Download PDFInfo
- Publication number
- WO2009155001A2 WO2009155001A2 PCT/US2009/045340 US2009045340W WO2009155001A2 WO 2009155001 A2 WO2009155001 A2 WO 2009155001A2 US 2009045340 W US2009045340 W US 2009045340W WO 2009155001 A2 WO2009155001 A2 WO 2009155001A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkoxy
- group
- alkyl
- substituted
- hydrogen
- Prior art date
Links
- 0 C/I(=*/C(CCCCC1NCC(*C2=O)C1*2=C)=O)/C Chemical compound C/I(=*/C(CCCCC1NCC(*C2=O)C1*2=C)=O)/C 0.000 description 8
- JTSGEWHPQPAXOR-UHFFFAOYSA-N CC(NC1CCCC1)=O Chemical compound CC(NC1CCCC1)=O JTSGEWHPQPAXOR-UHFFFAOYSA-N 0.000 description 2
- YPKZJRGKHXINPU-UHFFFAOYSA-N CC(CCCCC(C1N2)SCC1NC2=O)=O Chemical compound CC(CCCCC(C1N2)SCC1NC2=O)=O YPKZJRGKHXINPU-UHFFFAOYSA-N 0.000 description 1
- WHRYLXXHLFPFNZ-UHFFFAOYSA-N CN(C(C1C2C3C=CC1C3)=O)C2=O Chemical compound CN(C(C1C2C3C=CC1C3)=O)C2=O WHRYLXXHLFPFNZ-UHFFFAOYSA-N 0.000 description 1
- MDTWUVBRVLBXCO-UHFFFAOYSA-N COc1cc2cnc(-c(cc3)ccc3N(C(C3C4C5C=CC3C5)=O)C4=O)nc2cc1 Chemical compound COc1cc2cnc(-c(cc3)ccc3N(C(C3C4C5C=CC3C5)=O)C4=O)nc2cc1 MDTWUVBRVLBXCO-UHFFFAOYSA-N 0.000 description 1
- JWGUCOIJCISIAS-UHFFFAOYSA-N O=C(C(C1C=CC2C1)C2C1=O)N1c(cc1)ccc1-c1ncc(cccc2)c2n1 Chemical compound O=C(C(C1C=CC2C1)C2C1=O)N1c(cc1)ccc1-c1ncc(cccc2)c2n1 JWGUCOIJCISIAS-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/50—Pyridazines; Hydrogenated pyridazines
- A61K31/502—Pyridazines; Hydrogenated pyridazines ortho- or peri-condensed with carbocyclic ring systems, e.g. cinnoline, phthalazine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/44—Iso-indoles; Hydrogenated iso-indoles
- C07D209/48—Iso-indoles; Hydrogenated iso-indoles with oxygen atoms in positions 1 and 3, e.g. phthalimide
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/80—[b, c]- or [b, d]-condensed
- C07D209/94—[b, c]- or [b, d]-condensed containing carbocyclic rings other than six-membered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/46—Two or more oxygen, sulphur or nitrogen atoms
- C07D239/52—Two oxygen atoms
- C07D239/54—Two oxygen atoms as doubly bound oxygen atoms or as unsubstituted hydroxy radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Definitions
- the present invention generally relates to the fields of molecular biology and medicine. More particularly, it concerns the discovery of compounds that inhibit Wnt- mediated signal transduction pathways, including the Wnt/ ⁇ -catenin pathway.
- Wnt signalling proteins are deployed in almost all aspects of embryonic development in vertebrates (Clevers, 2006). In post-embryonic animals, their functions are essential to homeostatic tissue renewal and regeneration (Reya and Clevers, 2005). Similar to that of several other signal transduction pathways that have been shown to be important to cell fate decision-making, activity of the Wnt/ ⁇ -catenin pathway maintains transcriptional programs that enable stem cells to retain their multi-potency (Cole et ah, 2008; Van der Flier et ah, 2007).
- identification of methods and compounds that modulate the Wnt-dependent cellular responses may offer an avenue for therapeutic treatment of diseases associated with aberrant activity of these pathways.
- the present invention generally provides compounds and their use as Wnt protein signalling inhibitors. Also provided are methods of synthesis of these compounds and pharmaceutical compositions thereof.
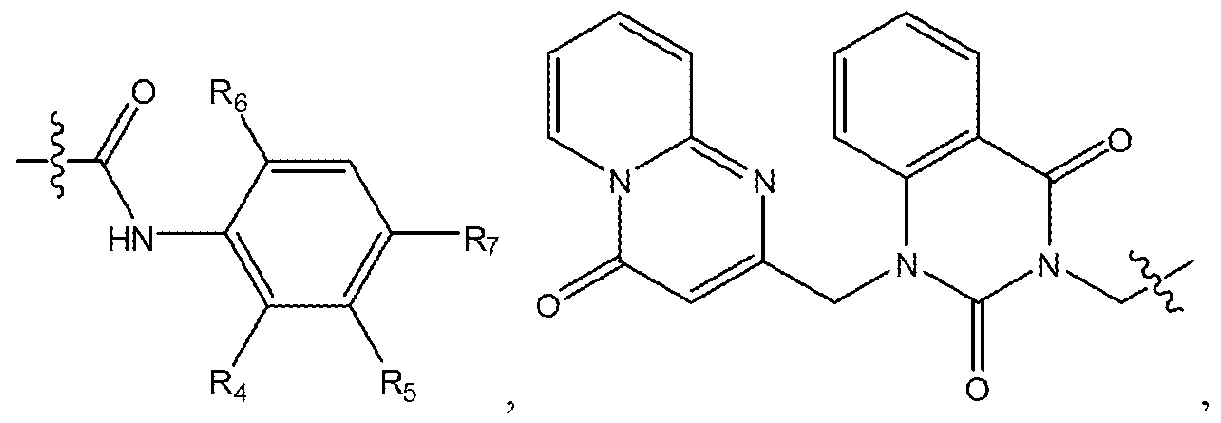
- the present invention provides a method of inhibiting Wnt protein signalling in a cell comprising administering to the cell an effective amount of a compound of formula (A):
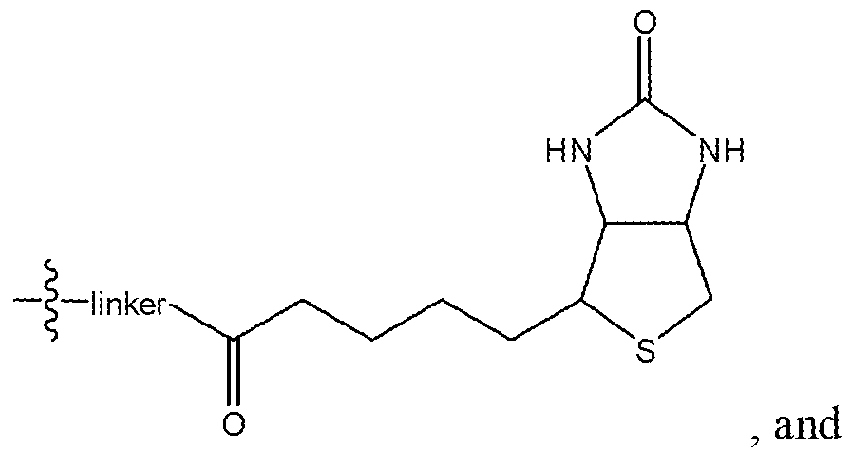
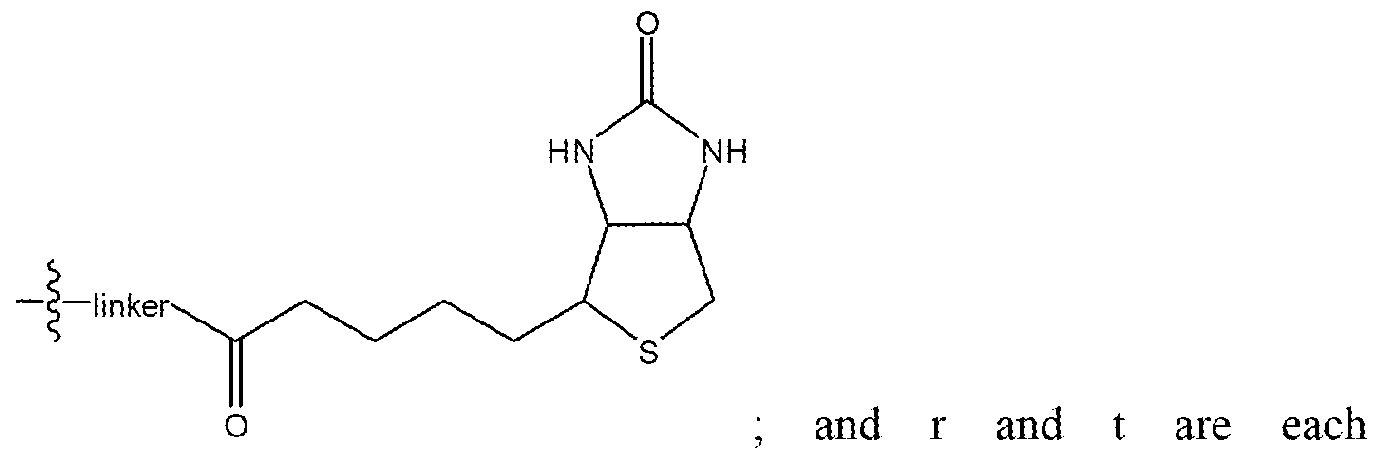
- Ri is selected from the group consisting of
- R 4 and R 5 are each independently selected from the group consisting of hydrogen, alkyl (C ⁇ 4) , substituted alkyl (C ⁇ 4), alkoxy( C ⁇ 4), substituted alkoxy (C ⁇ 4) and
- r and t are each independently 0 or 1 ; or
- Ri 3 is selected from the group consisting of hydrogen, halogen, alkyl ( c ⁇ 4 ), substituted alkyl (C ⁇ 4), alkoxy(c ⁇ 4), substituted alkoxy(c ⁇ 4) and a label;
- R ⁇ , R 7 and R 9 -R 11 are each independently selected from the group consisting of hydrogen, halogen, alkyl (C ⁇ 4 ), substituted alkyl (C ⁇ 4 ), alkoxy (C ⁇ 4 ), substituted alkoxy ( c ⁇ 4) and
- R 12 is selected from the group consisting of
- R 14 is selected from the group consisting of hydrogen, alkyl (C ⁇ 4) , substituted alkyl (C ⁇ 4) , alkoxy(c ⁇ 4) and substituted alkoxy ( c ⁇ 4)
- R- 2 is selected from the group consisting of hydrogen, alkyl ( e ⁇ 4 ), substituted alkyl ( c ⁇ 4 ), alkoxy ( c ⁇ 4) and substituted alkoxy(c ⁇ 4>
- R 3 is selected from the group consisting of hydrogen, halogen, alkyl (C ⁇ 4 ), substituted alkyl (C ⁇ 4), alkoxy (C ⁇ 4), substituted
- the compound is:
- the compound is any of the compounds disclosed in section III below, entitled “Wnt Protein Signalling Inhibitors.”
- the present invention provides a method of inhibiting Wnt protein signalling in a cell comprising administering to the cell an effective amount of a compound of formula (I):
- R 1 is selected from the group consisting of
- R 4 and R 5 are each independently selected from the group consisting of hydrogen, alkyl (C ⁇ 4) , substituted alkyl (C ⁇ 4 ), alkoxy (C ⁇ 4 ), substituted alkoxy ⁇ r ⁇ 4) and
- r and t are each independently 0 or 1 , or
- Ri 3 is selected from the group consisting of hydrogen, halogen, alkyl( C ⁇ 4), substituted alkyl ( c ⁇ 4), alkoxy(c ⁇ 4), substituted alkoxy ( c ⁇ 4) and a label;
- R 6 , R7 and R9- R ⁇ are each independently selected from the group consisting of hydrogen, halogen, alkyl(c ⁇ 4), substituted alkyl (C ⁇ 4), alkoxy ( c ⁇ 4), substituted alkoxy(c ⁇ 4) and
- Ri 2 is selected from the group consisting of and , wherein R H is selected from the group consisting of hydrogen, alkyl(c ⁇ 4i, substituted alkyl(c ⁇ 4>, alkoxy(c ⁇ 4) and substituted alkoxy ( c ⁇ 4); R2 is selected from the group consisting of hydrogen, alkyl (C ⁇ 4), substituted alkyl lC i4), alkoxy ( c ⁇ 4) and substituted alkoxy ( c ⁇ 4 >; and R 3 is selected from the group consisting of hydrogen, halogen, alkyl( C ⁇ 4), substituted alkyl( C ⁇ 4), alkoxv( C ⁇ 4), substituted alkoxy ( c ⁇ 4),
- this or any other method regarding a cell may take place wherein the cell is in vitro, or wherein the cell is in vivo.
- a method of the present invention e.g., a method of inhibiting Wnt protein signalling in a cell
- a method of inhibiting Wnt response may be further defined as a method of inhibiting Wnt response.
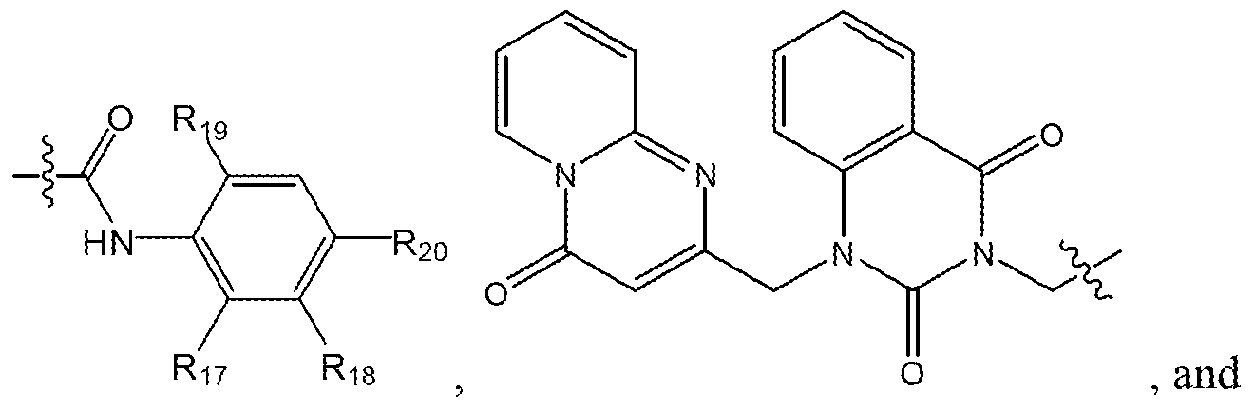
- a compound of formula (I) may be further defined as a compound of formula (II):
- Ri 5 is selected from the group consisting of wherein: Ri 7 and Rig are each independently selected from the group consisting of hydrogen, alkyl (C ⁇ 4) , substituted alkyl (C ⁇ 4 ), alkoxy (C ⁇ 4) , substituted alkoxy ( c ⁇ 4) and
- R 24 is selected from the group consisting of hydrogen, halogen, alkyl( C ⁇ 4), substituted alkyl (C ⁇ 4), alkoxy (C ⁇ 4), substituted alkoxy ( c ⁇ 4), and a label;
- Ri 9 and R 2 o are each independently selected from the group consisting of hydrogen, halogen, alkyl (C ⁇ 4) , substituted alkyl (C ⁇ 4), alkoxy ( c ⁇ 4), substituted alkoxy (C ⁇ 4) and
- R23 is selected from the group consisting of and 5 wherein R 24 is selected from the group consisting of hydrogen, alkyl (C ⁇ 4), substituted alkyl (C ⁇ 4), alkoxy ( c ⁇ 4), substituted alkoxy (C ⁇ 4); and Ri 6 is selected from the group consisting of hydrogen, alkyl( C ⁇ 4 i, substituted alkyl ( c ⁇ 4 ),
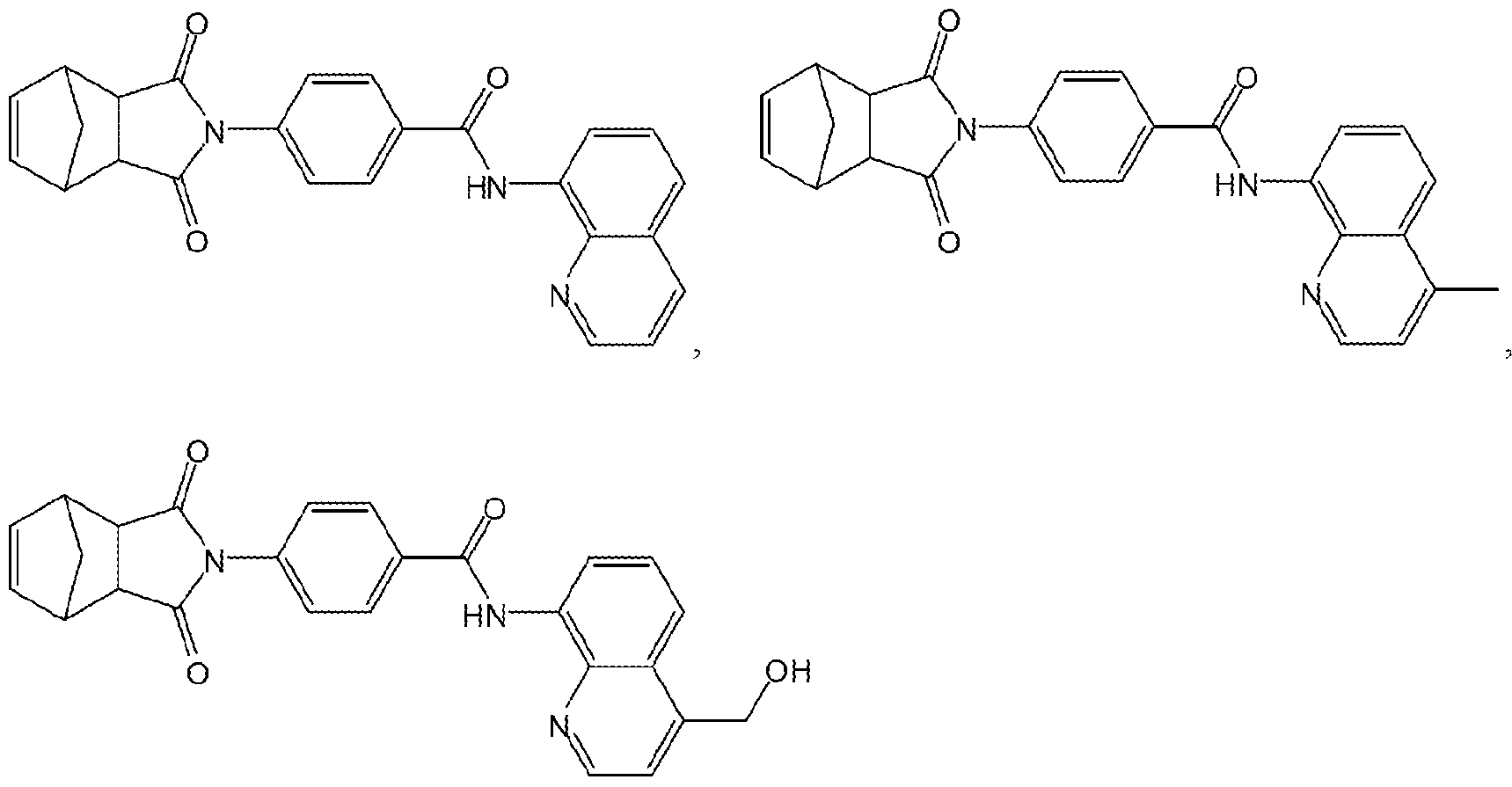
- the compound of formula (II) may be further defined as any one or more of the following:
- a method of the present invention may be further defined as a method of inhibiting Wnt protein production.
- a method of inhibiting Wnt protein production in a cell may comprise administering a compound of formula (I) to the cell.
- the compound of formula (I) may be further defined as a compound of formula (III):
- R25 is alkyl(c ⁇ 4), substituted alkyl ( c ⁇ 4), alkoxy(c ⁇ 4), or substituted alkoxy(c ⁇ 4); and R26 is selected from the group consisting of
- R27-R30, R 27a and R 2 Sa are each independently selected from the group consisting of hydrogen, halogen, alkyl(c ⁇ 4>, substituted alkyl ( c ⁇ 4), alkoxy(c ⁇ 4), substituted alkoxy (C ⁇ 4) and , and r and t are each independently 0 or 1.
- the compound of formula (III) is further defined as any one or more of the following:
- the compound of formula (I) is further defined as a compound of formula (IV):
- R 31 is selected from the group consisting of
- R 33 -R 35 are selected from the group consisting of hydrogen, halogen, alkyl (C ⁇ 4 ), substituted alkyl (C ⁇ 4 ), alkoxy (C ⁇ 4 ), substituted alkoxy (C ⁇ 4 ⁇ and
- R 32 is selected from the group consisting of
- R 36 -R 38 are selected from the group consisting of hydrogen, halogen, alkyl( C ⁇ 4> substituted alkyl (C ⁇ 4), alkoxy ( c ⁇ 4) and substituted alkoxy( C ⁇ 4>
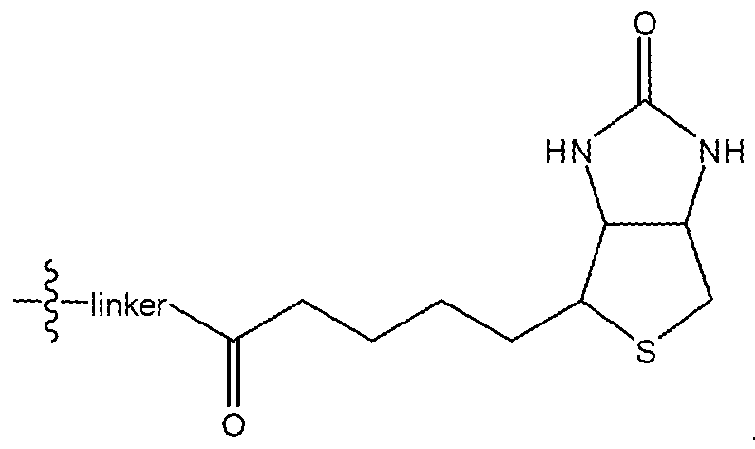
- Labels employed in methods, compounds and compositions of the present invention may be any type known to those of skill in the art.
- a label may be further defined as comprising biotin, such as the following:
- the label may comprise a fluorophore, such as the followin.
- the present invention provides the compounds:
- Ri and R 2 when taken alone, are each independently: hydrogen, hydroxy, halo, amino, nitro, hydroxyamino, cyano, azido or mercapto; or alkyl (C ⁇ i2), alkenyl (C ⁇ i2), alkynyl (C ⁇ i2), aryl (C ⁇ i2), aralkyl (C ⁇ i2), heteroaryl (C ⁇ i2), heteroaralkyl ( c ⁇ i2), alkoxy(c ⁇ i2), alkenyloxy ( c ⁇ i2), alkynyloxy ( c ⁇ i2), aryloxy ( c ⁇ i2), aralkoxy(c ⁇ i2), heteroaryloxy(c ⁇ i2), heteroaralkoxy(c ⁇ i2), acyloxy(c ⁇ i2), alkylamin ⁇ (c ⁇ i2), dialkylamm ⁇ (c ⁇ i2), alkoxyamin ⁇ (c ⁇ i2), alkenylamin ⁇ (c ⁇ i
- R] and R2 when taken together, are alkanediyl(C2-i2), alkenediyl ( c2-i2), or a substituted version of either of these groups.
- Ri and R 2 when taken together are:
- Ri or R 2 is halo. In some varations, Ri or R 2 is bromo. In other embodiments, Ri or R 2 is alkoxy (C ⁇ 6), for example, Ri or R 2 can be methoxy. Examples of compounds provided by the present invention include.
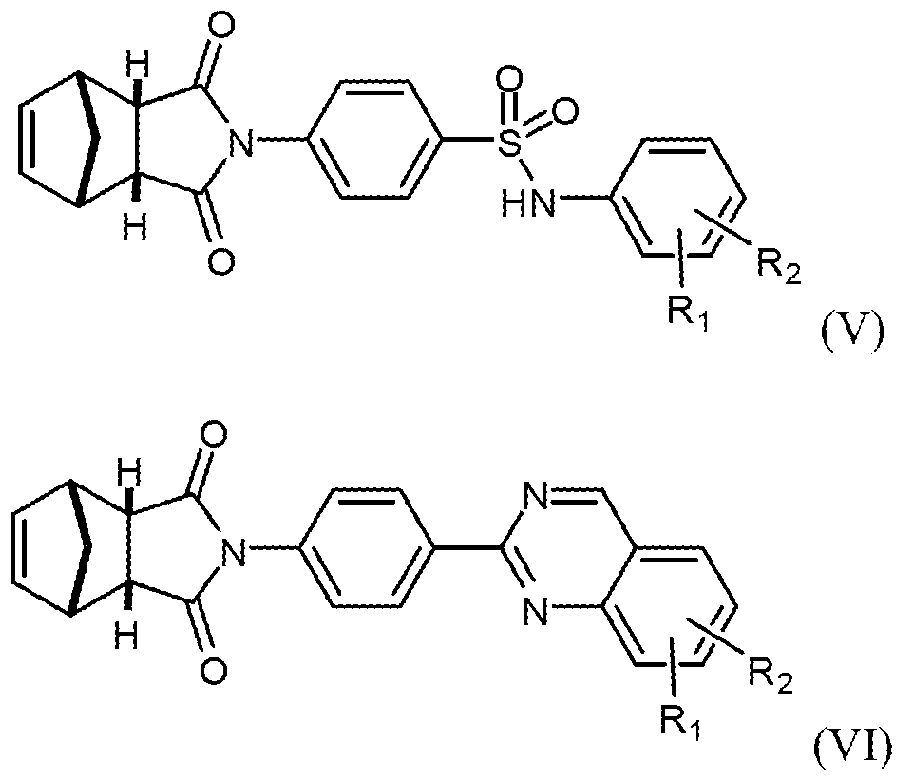
- the invention provides a method of inhibiting Wnt protein signalling in a cell comprising administering to the cell an effective amount of a compound of either formula (V) or formula (VI):
- Ri and R 2 when taken alone, are each independently: hydrogen, hydroxy, halo, amino, nitro, hydroxyamino, cyano, azido or mercapto; or alkyl (C ⁇ i2), alkenyl (C ⁇ i2), alkynyl (C ⁇ i2), aryl (C ⁇ i2), aralkyl (C ⁇ i2), heteroaryl (C ⁇ i2), heteroaralkyl(c ⁇ i2), alkoxy ( c ⁇ i2), alkenyloxy(c ⁇ i2), alkynyloxy ( c ⁇ i2), aryloxy(c ⁇ i2-), aralkoxy(c ⁇ i2), heteroaryloxy(c ⁇ i2), heteroaralkoxy(c ⁇ i2), acyloxy ( c ⁇ i2), alkylamin ⁇ (c ⁇ i2), dialkylamm ⁇ (c ⁇ i2), alkoxyamin ⁇ (c ⁇ i2), alkenylamin ⁇ (c ⁇ i2)
- Ri and R2 when taken together, are alkanediyl(C2-i2), alkenediyl(C2-i2), or a substituted version of either of these groups.
- the cell is in vitro. In other embodiments, the cell is in vivo.
- the method of inhibiting Wnt protein signalling is further defined as a method of inhibiting Wnt response. In some embodiments, the method further comprises one of the specific compounds described above.
- Methods of treatment are also contemplated by the present invention. Such methods may employ any compounds of the compounds described herein. For example, such methods may employ compounds of formulas (A) and (I)-(VI), described above and below.
- the present invention contemplates a method of treating cancer in a patient comprising administering to the patient an effective amount of a compound of formula (A) or any of its sub-generic formulas (I), (II), (III), and/or (IV).
- the present invention contemplates a method of treating cancer in a patient comprising administering to the patient an effective amount of a compound of formula (V) or (VI) or any of their sub-generic formulas.
- the specific compounds described herein are also contemplated in methods of treating cancer.
- the cancer may be colorectal, breast, liver, lung, or prostate cancer.
- Methods of treating cancer may also further comprise administration of a chemotherapeutic, radiation therapy, immunotherapy, hormone therapy, toxin therapy, or gene therapy: such additional methodologies are well-known in the art.
- Methods of administration may include intravenously, intradermally, intraarterially, intraperitoneally, intralesionally, intracranially, intraarticular ⁇ , intraprostaticaly, intrapleurally, intratracheally, intranasally, intravitreally, intravaginally, intrarectally, topically, intratumorally, intramuscularly, subcutaneously, subconjunctival, intravesicularlly, mucosally, intrapericardially, intraumbilically, intraocularally, orally, locally, via inhalation, via injection, via infusion, via continuous infusion, via localized perfusion bathing target cells directly, via a catheter, via a lavage, in cremes, in lipid compositions, or any combination thereof.
- Dosages may include, e.g., about 1 ⁇ g/kg to about 100 mg/kg, or any range derivable therein.
- the compounds disclosed herein may be combined with a pharmaceutically acceptable carrier, diluent, and/or excipient in a pharmaceutical composition.
- compositions are contemplated by the present invention.
- a pharmaceutical composition comprising a pharmaceutically acceptable carrier, diluent, and/or excipient and any one or more of the following compounds is contemplated:
- Another general aspect of the present invention contemplates a method of treating or preventing osteopetrosis in a patient comprising administering to the patient an effective amount of a compound disclosed herein. Such methods may further comprise administration of a second osteopetrosis-treating agent or a second osteopetrosis-preventing agent.
- Administration of the compound of interest may take place via a route selected from the group consisting of intravenously, intradermally, intraarterially, intraperitoneally, intralesionally, intracranially, intraarticularly, intranasally, topically, intramuscularly, subcutaneously, intraumbilically, orally, locally, via inhalation, via injection, via infusion, via continuous infusion, via localized perfusion bathing target cells directly, via a catheter, in cremes, in lipid compositions, or any combination thereof.
- Dosage amounts may range between, for example, about 1 ⁇ g/kg to about 100 mg/kg, or any range derivable therein.
- Also contemplated by the present invention are methods of treating a degenerative disease in a patient comprising administering to the patient an effective amount of a compound disclosed herein.
- the degenerative disease may be, for example, type II diabetes or age-related impairment of tissue repair.
- Methods may further comprise administration of a second agent to treat the degenerative disease.
- Methods of administration may be selected from the group consisting of intravenously, intradermally, intraarterially, intraperitoneally, intralesionally, intracranially, intraaiticularly, mtraprostaticaly, intrapleurally, intratracheally, intranasally, intravitreally, intravaginally, intrarectally, topically, intramuscularly, subcutaneously, subconjunctival, intra vesicularlly, mucosally, intrapericardially, intraumbilically, intraocularally, orally, locally, via inhalation, via injection, via infusion, via continuous infusion, via localized perfusion bathing target cells directly, via a catheter, via a lavage, in cremes, in lipid compositions, or any combination thereof. Dosage amounts may range between, for example, about 1 ⁇ g/kg to about 100 mg/kg, or any range derivable therein.
- Also disclosed herein are methods of treating type II diabetes in a patient comprising administering to the patient an effective amount of a compound disclosed herein. Such methods may further comprise administration of a second agent to treat diabetes.
- Methods of administration may be selected from the group consisting of intravenously, intradermally, intraarterially, intraperitoneally, intralesionally, intracranially, intraarticularly, intraprostaticaly, intrapleurally, intratracheally, intranasally, intravitreally, intravaginally, intrarectally, topically, intramuscularly, subcutaneously, subconjunctival, intravesicularlly, mucosally, intrapericardially, intraumbilically, intraocularally, orally, locally, via inhalation, via injection, via infusion, via continuous infusion, via localized perfusion bathing target cells directly, via a catheter, via a lavage, in cremes, in lipid compositions, or any combination thereof. Dosage amounts may range between, for example, about 1 ⁇ g
- any limitation discussed with respect to one embodiment of the invention may apply to any other embodiment of the invention.
- any composition of the invention may be used in any method of the invention, and any method of the invention may be used to produce or to utilize any composition of the invention.
- FIG. 1 Identification of small molecule antagonists of the Wnt/ ⁇ -catenin signal transduction pathway.
- a -200K chemical library from U.T. Southwestern (Dallas, TX) (UTSW) was screened using a cell line with constitutive Wnt/ ⁇ -catenin pathway activity maintained by cell-autonomous WntSA protein production (L-Wnt-STF cells; Primary screen).
- Potential Wnt/ ⁇ -catenin pathway antagonists were identified using a stably transfected Wnt-responsive firefly luciferase (FL) and control Renilla luciferase (RL) reporters.
- FL stably transfected Wnt-responsive firefly luciferase
- RL Renilla luciferase
- Hh and Notch pathways were tested for effects on two other signal transduction pathways (the Hh and Notch pathways) using cultured cell-based assays similar to those used to identify Wnt/ ⁇ -catenin pathway antagonists (Hh and Notch pathway tests). Hh and Notch pathways were activated using either Shh or an activated Notch (NICD) cDNA constructs, respectively. Those compounds that minimally impacted these two pathways were considered to have specific activity for the Wnt/ ⁇ -catenin pathway.
- Hh and Notch pathways were activated using either Shh or an activated Notch (NICD) cDNA constructs, respectively.
- IWPs were directly tested for their ability to inhibit Wnt3A protein secretion (Wnt secretion test; see FIG. 9). Criteria for selecting hits are provided in FIG. 1. In the end, five IWRs and four IWPs with high specificity for attacking the Wnt/ ⁇ - catenin pathway were selected for further analysis (FIG. 9). Concentration of compounds used in each test is noted. Insets show schematics of assays used in the screen and secondary tests with the utility of each luciferase read-out.
- FIG. 2 Chemical structure and potency of IWR and IWP compounds.
- FIG. 2A Chemical structure and potency of IWR and IWP compounds.
- IWR compounds Two IWR compounds that differ by only a single methyl group and that share similar IC50's (as determined in L-Wnt-STF cells; upper right of graphs) were designated Class I compounds. The remaining three IWRs which share structural similarity (see also FIG. 10) were designated Class II compounds.
- FIG. 2B Two IWR compounds that differ by only a single methyl group and that share similar IC50's (as determined in L-Wnt-STF cells; upper right of graphs) were designated Class I compounds. The remaining three IWRs which share structural similarity (see also FIG. 10) were designated Class II compounds.
- FIG. 2B Two IWR compounds that differ by only a single methyl group and that share similar IC50's (as determined in L-Wnt-STF cells; upper right of graphs) were designated Class I compounds.
- the remaining three IWRs which share structural similarity were designated Class II compounds.
- FIG. 2B Two IWR compounds that differ by only a single methyl group and that
- IWP-2 IC50's with IWPs 2-4 sharing the same core structure (IWP-2) and differing only by either the presence of an additional fluoro or methoxy adduct (IWP-3 and IWP -4, respectively).
- FIG. 3 Biochemical evidence for Wnt/ ⁇ -catenin pathway inhibition by IWR and IWP compounds.
- L-Wnt-STF cells that exhibit constitutive Wnt pathway activation were incubated with IWR (10 mM) and IWP (5 mM) compounds for 24 hrs prior to lysis.
- Cellular lysates were subjected to Western blot analysis to determine levels of LRP6 and Dvl2 phosphorylation, and ⁇ -catenin accumulation, all biochemical events associated with Wnt/ ⁇ - catenin pathway activity.
- IWPs blocked all three biochemical events, whereas IWRs appear to block ⁇ -catenin accumulation without affecting LRP6 and Dvl2 phosphorylation.
- KiBA and tubulin serve as loading controls. Wild-type L-cells stimulated with exogenous Wnt3A protein provided in conditioned medium exhibit similar biochemical changes in Wnt pathway components as that observed in the L-Wnt-STF cells.
- FIGS. 4A-G IWP compounds target the Porcupine O-acyltransferase.
- FIG. 4A Overexpression of the O-acyltransferase Pore but not the Wnt chaperone Evi counters the effects of IWP compounds on Wnt/ ⁇ -catenin pathway activity in HEK293 cells. Wnt/ ⁇ - catenin pathway activity was measured using the STF reporter as before in a 24 hr assay.
- FIG. 4B Overexpression of Pore reverses the block in Wnt protein secretion induced by IWP compounds.
- FIG. 4C IWP compounds inhibit lipidation of Wnt3A in a Pore-dependent manner. Lipidated Wnt3A protein, found in the detergent fraction of an established phase-separation assay used to detect modified Wnt proteins, is absent in IWP -treated cells. In cells overexpressing Pore, detergent-soluble Wnt3A is retained even in the presence of IWP -2.
- FIG. 4D IWP compounds inhibit lipidation of Wnt3A in a Pore-dependent manner. Lipidated Wnt3A protein, found in the detergent fraction of an established phase-separation assay used to detect modified Wnt proteins, is absent in IWP -treated cells. In cells overexpressing Pore, detergent-soluble Wnt3A is retained even in the presence of IWP -2.
- FIG. 4D IWP compounds inhibit lipidation of Wnt3A in a Pore-dependent manner. Lipidated Wnt3A protein, found in the detergent fraction of an established phase-s
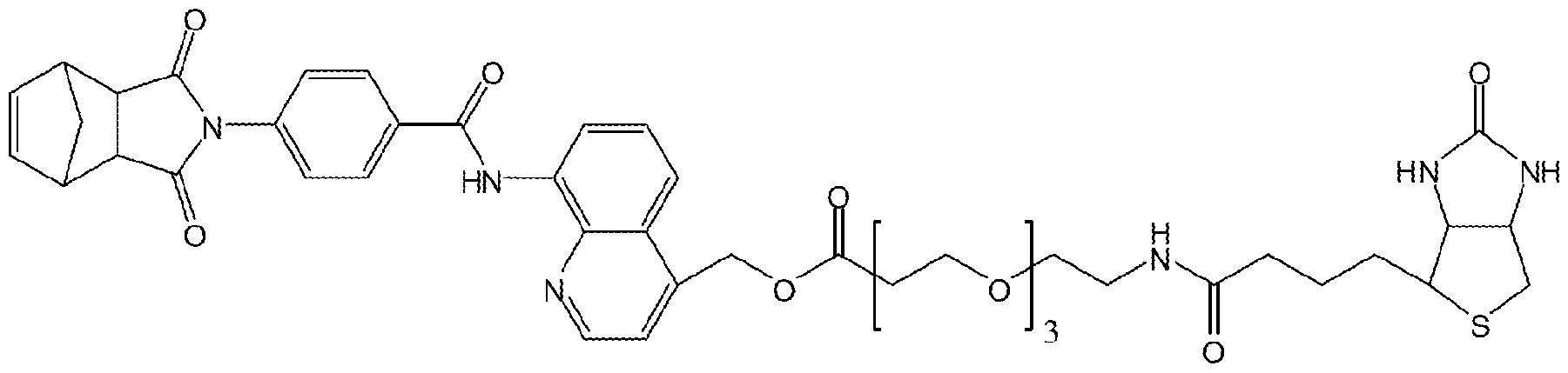
- FIG. 4F Structure of biotinylated IWP-2 and its association with Pore. In order to generate an IWP-2 compound that could be affixed to a streptavidin- based solid support for biochemical studies (IWP-PEG-Biotin; see FIG.
- FIG. 4G Model of IWP action. IWPs inhibit Pore function thereby rendering Wnt proteins non- functional.
- FIGS. 5A-H Stabilization of the Axin2 destruction complex by IWR compounds.
- FIG. 5 A IWR compounds block ⁇ -catenin accumulation induced by loss of APC tumor suppressor. Accumulation of ⁇ -catenin protein in mouse L-cells treated with APC siRNAs can be blocked by IWR-I.
- FIG. 5B IWRs block aberrant Wnt/ ⁇ -catenin pathway activity in the colorectal cancer (CRC) cells. Aberrant Wnt/ ⁇ -catenin pathway activity in DLD-I cells, CRC cells which harbor a loss-of-function mutation in APC, is abrogated by IWR compounds. Pathway activity was monitored using the STF reporter and normalized to RL control reporter as before.
- FIG. 5C IWRs induce accumulation of Axin2 protein.
- FIG. 5D Levels of ⁇ -catenin in CRC cells available for Wnt response are decreased in the presence of IWR compounds. Depletion of ⁇ -catenin bound to E-cadherin receptor proteins reveals levels of ⁇ -catenin available for Wnt pathway response in cells are decreased in cell treated with IWR.
- FIG. 5E IWR stabilizes Axin2 protein.
- FIG. 5F Structure of biotmylated IWR-I (IWR-I-PEG-B).
- FIG. 5G Axin2 interacts with IWR-I-PEG-B.
- Lysates derived from cells transfected with either a control, Axin2, or Axin2 lacking the DAX C-terminal domain (Axin2 ⁇ DAX) expression construct were incuabed with IWR-I-PEG-B, strepavidin agarose beads, and either DMSO or IWR-I .
- FIG. 5H A proposed model of IWR action. Addition of IWR to cells induces stabilization of Axin2 protein with consequential increase in ⁇ -catenin destruction.
- FIGS. 6A-C Chemical inhibition of the Wnt/ ⁇ -catenin pathway in regeneration and cancer.
- FIG. 6A IWR-I prevents caudal fin regeneration in zebrafish. Adult zebrafish with resected caudal fins were placed in water containing DMSO carrier or IWR-I (10 ⁇ M) for four days with replenishment of water and compounds every day. Consistent with inhibition of Wnt/ ⁇ -catenin pathway response by IWR-I, fish treated with IWR-I but not DMSO failed to regenerate fin tissue. Length of regenerated tissue is indicated by bar.
- FIG. 6B IWR-I blocks normal homeostatic renewal of the GI tract.
- FIG. 6C Quantification of BrudU-labeled cells in the intestinal tract of control or IWR-1-treated fish. Histological sections as seen in FIG. 5B (middle column) were scored for the percentage of intestinal folds that contain BrdU-labeled cells. Four independent scorers analyzed sections from eight fish either from control and IWR-I treated groups. Ratios provided represent the number of BrdU-labeled cells in the numerator and the number of intestinal folds scored in the denominator.
- FIGS. 7A-D Chemical inhibition of Wnt-mediated cellular responses in cancer.
- FIG. 7A Growth-inhibitory effects of IWR and IWP compounds on cancerous cells.
- FIG. 7B Biochemical changes of Wnt pathway components in cancerous cells treated with either IWR or IWP compounds. Lysates from H460, or DLD-I cells were treated with IWR or IWP and Western blotted for either Dvl2, Axin2, Axinl, or Actin.
- FIG. 7C Genetic evidence for non- ⁇ -catenin dependent Wnt-mediated signalling in cancerous cells. Targeting of Pore using RNAi in lung cancer or CRC cell lines results in loss of clonal density cell growth whereas targeting ⁇ -catenin with a similar approach altered growth in only the DLD-I cells.
- FIG. 7D Inhibition of ligand-dependent and -independent Wiit pathway activites with IWR and IWP compounds: a proposed mechanism. Based on the proposed mechanism of action as shown in FIG. 7D, IWP and IWR compounds may inhibit pathway response that is driven in a ligand-dependent manner. Additionally, IWR compounds may block ligand-independent pathway responses such as those induced by loss of APC in colorectal cancer cells.
- FIG. 8 Criteria used to identify hits in the screen. Flow diagram of screening process to identify chemical inhibitors of Wnt/ ⁇ -catenin pathway activity as shown in FIG. 1 except with criteria used to identify compounds of interest noted in each step.
- FIGS. 9A-C IWR and IWP compounds specifically inhibit the Wnt/ ⁇ -catenin pathway.
- FIG. 9A Summary of results relating to IWR and IWP compounds from the screening process. Wnt pathway tests were performed in either cells responding to autonomously-produced Wnt protein (L-Wnt-STF cells) or exogenously provided Wnt in conditioned medium (HEK293 cells).
- FIG. 9B IWP compounds inhibit Wnt3A secretion. Left: schematic of Wnt-Gaussia luciferase (Wnt-GL) fusion protein used to monitor levels of secreted Wnt protein in the cell medium.
- Wnt-GL Wnt-Gaussia luciferase
- FIG. 9C IWR and IWP compounds generally inhibit Wnt/ ⁇ - catenin pathway response induced by Wnt proteins. Pathway activity induced by Wntl, Wnt2, or Wnt3a, and monitored using the STF reporter, is decreased in cells treated with either IWR-I or IWP-2. FL activity was normalized to control RL activity as before.
- FIG. 10 IWR 3-5 share structural similarity. Three dimensional representation of IWRs 3-5 in equilibrium geometry using AMI semi-empirical methods reveals similarities in structure. All three structures are superimposed on the right.
- FIGS. 11A-B Synthetic scheme for IWP-2 and IWP-PE G-Biotin. Synthetic routes for IWP-2 (FIG. 1 IA) and IWP-PEG-Biotin (FIG. 1 IB) are shown.
- FIGS. 12A-G Synthetic scheme for IWR-I, IWR-I-PEG-B, and IWR-Cy3 compound.
- FIG. 12A Synthetic route for IWR-I. Endo and exo diastereomers result depending on the starting material.
- FIG. 12B Diagram of endo and exo IWR-I structures.
- FIG. 12C Normalized data regarding endo and exo IWR-I on Wnt pathway response.
- FIG. 12D Synthetic scheme for IWR-Cy3.
- FIG. 12E Synthetic scheme for IWR-I-PEG-B.
- FIG. 12F IWR-Cy3 and IWR-I-PEG-B retains activity against the Wnt/ ⁇ -catenin pathway as measured using L-Wnt-STF cells.
- FIG. 12G IWR-Cy3 and IWR-I-PEG B, like their parental compound IWR-I, inhibit accumulation of ⁇ -catenin in L- Wnt cells.
- FIGS. 13A-C A second-generation IWR compound with increased metabolic stability.
- IWR-related compounds with either greater potency or more favorable pharmacokinetic parameters that those observed for IWR-I
- two IWR-6 and IWR- 7; FIG. 13A
- FIG. 13B One of these (IWR-7) also has a greater half-life than IWR-I as measured using a hepatocyte co-culture assay (FIG. 13C).
- FIGS. 14A-C Characterization of IWP action and specificity.
- FIG. 14A IWP-2 does not induce destruction of Porcn. Levels of overexpressed Porcn increase in the presence of IWP-2.
- FIG. 14B IWP-2 does not appear to alter localization of Porcn.
- FIG. 14C Chemical structure and activity of several compounds related to IWP-2. IWP-2-v2 retains activity against the Wnt/ ⁇ -catenin pathway as measured using the STF reporter (right), whereas IWP-2-vl and -v3 do not.
- FIGS. 15-24 1 H-NMR Spectra for Inhibitors of Wnt Response.
- FIG. 15 corresponds to compound IWR-8.
- FIG. 16 corresponds to compound IWR-9
- FIG 17 corresponds to compound IWR-10.
- FIG. 18 corresponds to compound IWR-I l.
- FIG. 19 corresponds to compound IWR- 12.
- FIG. 20 corresponds to compound IWR-13.
- FIG. 21 corresponds to compound IWR-14.
- FIG. 22 corresponds to compound IWR-15.
- FIG. 23 corresponds to compound IWR- 18.
- FIG. 24 corresponds to compound IWR-19.
- FIG. 25 Inhibition of Fin Regeneration in Zebrafish by IWRs. Arrows indicate the points of resection. The minimum inhibitory concentration of IWR-I is 0.5 ⁇ M. Only partial inhibition of fin regeneration was observed with moderate inhibitors 13 and 43. The weak inhibitor 17 only retarded the growth of the tail fin (picture not shown). DESCRIPTION OF ILLUSTRATIVE EMBODIMENTS
- Small molecules that target Wnt-dependent signal transduction pathways such as the Wnt/ ⁇ -catenin pathway, have been identified. These small molecules reveal chemically- sensitive regulatory mechanisms within these signal transduction pathway that may be exploited by pharmacological means for medical use, such as regenerative and anti-cancer therapy.
- the Wnt gene family encodes secreted ligand proteins that serve key roles in differentiation and development
- This family comprises at least 15 vertebrate and invertebrate genes including the Drosophila segment polarity gene wingless and one of its vertebrate homologues, integrated from which the Wnt name derives.
- the Wnt proteins appear to facilitate a number of developmental and homeostatic processes.
- the Wnt signalling pathways comprises a number of proteins involved in the transduction of cellular responses to secreted Wnt/wingless signalling proteins.
- Wnt proteins that control "non-canonical" pathways such as the Wnt/calcium and planar cell polarity pathways, induce cellular responses that are not-dependent upon ⁇ -catenin.
- the Frizzled receptor activates Disheveled protein, which blocks the inhibiting action of Zeste-white-3 kinase (or GSK3 ⁇ in vertebrates, Glycogen Synthase Kinase-3 ⁇ ) upon the Armadillo protein (a ⁇ -catenin protein).
- the ⁇ -catenin protein transduces the Wnt signal from the cytoplasm to the nucleus.
- ⁇ -catenin is constitutively degraded by the proteasome and can be found in a multimeric complex with conductin (or axin), APC (Adenomatous Polyposis CoIi) and GSK3 ⁇ .
- APC mediates the binding of ⁇ -catenin to conductin and serves to activate the conductin protein.
- Conductin acts as a scaffold to assemble the components of the degradation pathway of ⁇ -catenin.
- GSK3 ⁇ a serine/threonine kinase, phosphorylates ⁇ - catenin, thus stimulating its degradation by the proteasome.
- ⁇ -Catenin Upon Wnt signalling, GSK3 ⁇ kinase is inactivated, leading to stabilization of the ⁇ - catenin protein. ⁇ -Catenin is then released from the multimeric complex and translocates into the nucleus. Once in the nucleus, ⁇ -catenin interacts with the LEF/TCF (Lymphoid Enhancer Factor/T-Cell Factor) family of HMG (High Mobility Group) box transcription factors. The LEF/TCF factors are stimulated through interaction with ⁇ -catenin to become potent transactivators of a number of genes including c-myc and cyclin Dl . II. Therapeutic Implications of Wn t-contr oiled Signal Transduction Pathways
- Wnt-mediated pathway responses sustained by genetic changes that result either in altered Wnt ligand activity or in altered functioning of pathway regulators, have been associated with a broad range of cancers. See Clevers, 2006 and Polakis, 2007, both of which are incorporated herein by reference. Notably, more than 90% of colorectal cancer (CRC) tumors harbor a loss-of-function mutation in APC, a suppressor of the Wnt/b-catenin pathway. See Sjoblom et al, 2006, which is incorporated herein by reference. The ability of IWR compounds to stabilize Axin proteins and induce ⁇ -catenin destruction even in the absence of normal APC protein function suggests that they may block aberrant cell growth supported by hyperactivation of Wnt/ ⁇ -catenin responses.
- CRC colorectal cancer
- IWR compounds are able to inhibit aberrant Wnt/ ⁇ -catenin activity as a consequence of Ape loss in both mouse L cells (using Ape small interfering RNAs) and DLD-I colorectal cancer cells (that harbor a loss-of- function mutation in APC).
- Ape small interfering RNAs a small interfering RNA that was used to inhibit Wnt/ ⁇ -catenin activity.
- DLD-I colorectal cancer cells that harbor a loss-of- function mutation in APC.
- IWR-3 The ability of IWR-3 to mimic the cell growth effects of ⁇ -catenin siRNAs in several cancer cell lines that exhibit differences in growth dependency on Wnt/ ⁇ -catenin pathway activity was also tested.
- IWR-3 mimicked the effects of b-catenin siRNAs on the growth of cells derived from cancers of the colon (DLD-I) and prostate (DU145) but not lung (H460), which suggests that IWR-3 successfully targeted the Wnt/b-catenin pathway in these cells.
- overexpression of b-catenin can overcome the effects of IWR-3 on DLD-I cell growth.
- the present invention provides small molecules that inhibit the Wnt protein signalling pathway.
- Wnt Protein Signalling Inhibitors include IWR-I and IWR-2, as shown here along with their respective in vitro activities:
- R H: IWR-1 (1), EC 50 0.2 ⁇ M Me: IWR-2 (2), EC 50 0.2 ⁇ M
- Tables 1 and 2 provides further examples of such compounds, as well as their activities as determined by a luciferase-based reporter assay that measures Wnt/ ⁇ -catenin-dependent transcriptional response.
- able 1 Examples of Small Molecule Wnt Inhibitors.
- Wnt signaling inhibitors are compounds 50, 51 and 52: Yet still further examples include:
- Wnt protein signalling pathway refers to the pathways by which binding of the Wnt protein to extracellular receptors is either translated into the nucleus and results in transcriptional activation of a variety of genes, or otherwise results in biochemical changes that influence cell behavior.
- the Wnt protein signalling pathways involve a variety of proteins including Frizzled, Disheveled, Axin, APC, GSK3 ⁇ , ⁇ -catenin, LEF/TCF transcription factors, etc.
- C ells from many different species express homologs of the proteins involved in Wnt protein signalling pathways and accordingly have functionally equivalent Wnt protein signalling pathways.
- Wnt protein signalling inhibitor is an organopharmaceutical (that is, a small organic molecule) that inhibits Wnt protein signalling activity.
- Wnt protein signalling inhibitors typically have a molecular weight of about 1000 g/mol or less.
- a method of inhibiting Wnt response refers to methods of inhibiting known biochemical events associated with production of functional Wnt proteins or with cellular responses to Wnt proteins.
- small organic molecules may inhibit Wnt response in accordance with this definition.
- (Cn) defines the exact number (n) of carbon atoms in the group.
- (C ⁇ n) defines the maximum number (n) of carbon atoms that can be in the group, with the minimum number of carbon atoms in such at least one, but otherwise as small as possible for the group in question.
- the minimum number of carbon atoms in the group “alkenyl ⁇ c ⁇ 8)" is 2.
- alkoxy (C ⁇ io) designates those alkoxy groups having from 1 to 10 carbon atoms (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10, or any range derivable therein (e.g., 3- 10 carbon atoms)).
- Cn-n' defines both the minimum (n) and maximum number (n') of carbon atoms in the group.
- alkyl ( c 2 -io ) designates those alkyl groups having from 2 to 10 carbon atoms (e.g., 2, 3, 4, 5, 6, 7, 8, 9, or 10, or any range derivable therein (e.g. , 3- 10 carbon atoms)).
- alkyl when used without the “substituted” modifier refers to a non- aromatic monovalent group with a saturated carbon atom as the point of attachment, a linear or branched, cyclo, cyclic or acyclic structure, no carbon-carbon double or triple bonds, and no atoms other than carbon and hydrogen.
- substituted alkyl refers to a non-aromatic monovalent group with a saturated carbon atom as the point of attachment, a linear or branched, cyclo, cyclic or acyclic structure, no carbon-carbon double or triple bonds, and at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- the following groups are non-limiting examples of substituted alkyl groups: -CH 2 OH, -CH 2 Cl, -CH 2 Br, -CH 2 SH, -CF 3 , -CH 2 CN, -CH 2 C(O)H, -CH 2 C(O)OH, -CH 2 C(O)OCH 3 , -CH 2 C(O)NH 2 , -CH 2 C(O)NHCH 3 , -CH 2 C(O)CH 3 , -CH 2 OCH 3 , -CH 2 OCH 2 CF 3 , -CH 2 OC(O)CH 3 , -CH 2 NH 2 , -CH 2 NHCH 3 , -CH 2 N(CH 3 ) 2 , -CH 2 CH 2 Cl, -CH 2 CH 2 OH, -CH 2 CF 3 , -CH 2 CH 2 OC(O)CH 3 , -CH 2 CH 2 NHCO 2 C(CH 3 )S, and -CH 2
- alkanediyl when used without the "substituted” modifier refers to a non- aromatic divalent group, wherein the alkanediyl group is attached with two ⁇ -bonds, with one or two saturated carbon atom(s) as the point(s) of attachment, a linear or branched, cyclo, cyclic or acyclic structure, no carbon-carbon double or triple bonds, and no atoms other than carbon and hydrogen.
- the groups -CH 2 - (methylene), -CH 2 CH 2 -, -CH 2 C(CH 3 ) 2 CH 2 -,
- alkanediyl groups are non-limiting examples of alkanediyl groups.
- substituted alkanediyl refers to a non-aromatic monovalent group, wherein the alkynediyl group is attached with two ⁇ -bonds, with one or two saturated carbon atom(s) as the point(s) of attachment, a linear or branched, cyclo, cyclic or acyclic structure, no carbon- carbon double or triple bonds, and at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- the following groups are non-limiting examples of substituted alkanediyl groups: -CH(F)-, -CF 2 -, -CH(Cl)-, -CH(OH)-, -CH(OCH 3 )-, and -CH 2 CH(Cl)-.
- alkenyl when used without the "substituted” modifier refers to a monovalent group with a nonaromatic carbon atom as the point of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one nonaromatic carbon-carbon double bond, no carbon-carbon triple bonds, and no atoms other than carbon and hydrogen.
- substituted alkenyl refers to a monovalent group with a nonaromatic carbon atom as the point of attachment, at least one nonaromatic carbon-carbon double bond, no carbon-carbon triple bonds, a linear or branched, cyclo, cyclic or acyclic structure, and at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- alkenediyl when used without the "substituted” modifier refers to a nonaromatic divalent group, wherein the alkenediyl group is attached with two ⁇ -bonds, with two carbon atoms as points of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one nonaromatic carbon-carbon double bond, no carbon-carbon triple bonds, and no atoms other than carbon and hydrogen.
- substituted alkenediyl refers to a non-aromatic divalent group, wherein the alkenediyl group is attached with two ⁇ -bonds, with two carbon atoms as points of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one nonaromatic carbon-carbon double bond, no carbon-carbon triple bonds, and at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I 7 Si 7 P, and S.
- alkynyl when used without the “substituted” modifier refers to a monovalent group with a nonaromatic carbon atom as the point of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one carbon-carbon triple bond, and no atoms other than carbon and hydrogen.
- the groups, -C ⁇ CH, -C ⁇ CCHj, -C ⁇ CC ⁇ Hs and -CH 2 C ⁇ CCH 3 are non-limiting examples of alkynyl groups.
- substituted alkynyl refers to a monovalent group with a nonaromatic carbon atom as the point of attachment and at least one carbon-carbon triple bond, a linear or branched, cyclo, cyclic or acyclic structure, and at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- the group, -C ⁇ CSi(CH,) 3 is a non-limiting example of a substituted alkynyl group.
- alkynediyl when used without the "substituted” modifier refers to a non- aromatic divalent group, wherein the alkynediyl group is attached with two ⁇ -bonds, with two carbon atoms as points of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one carbon-carbon triple bond, and no atoms other than carbon and hydrogen.
- the groups, -C ⁇ C ⁇ , -C ⁇ CCH 2 -, and -C ⁇ CCH(CH 3 )- are non-limiting examples of alkynediyl groups.
- substituted alkynediyl refers to a non-aromatic divalent group, wherein the alkynediyl group is attached with two ⁇ -bonds, with two carbon atoms as points of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one carbon-carbon triple bond, and at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- the groups -C ⁇ CCFH- and -C ⁇ CHCH(Cl)- are non-limiting examples of substituted alkynediyl groups.
- aryl when used without the "substituted” modifier refers to a monovalent group with an aromatic carbon atom as the point of attachment, said carbon atom forming part of a six-membered aromatic ring structure wherein the ring atoms are all carbon, and wherein the monovalent group consists of no atoms other than carbon and hydrogen.
- substituted aryl refers to a monovalent group with an aromatic carbon atom as the point of attachment, said carbon atom forming part of a six- membered aromatic ring structure wherein the ring atoms are all carbon, and wherein the monovalent group further has at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- Non-limiting examples of substituted aryl groups include the groups: -C 6 H 4 F, -C 6 H 4 Cl, -C 6 H 4 Br, -C 6 H 4 I, -C 6 H 4 OH, -C 6 H 4 OCH 3 , -C 6 H 4 OCH 2 CH 3 , -C 6 H 4 OC(O)CH 3 , -C 6 H 4 NH 2 , -C 6 H 4 NHCH 3 , -C 6 H 4 N(CH 3 ) 2 , -C 5 H 4 CH 2 OH, -C 6 H 4 CH 2 OC(O)CH 3 , -C 6 H 4 CH 2 NH 2 , -C 6 H 4 CF 3 , -C 6 H 4 CN, -C 6 H 4 CHO, -C 6 H 4 CHO, -C 6 H 4 C(O)CH,, -C 6 H 4 C(O)C 6 H 5 , -C 6 H 4 CO 2 H, -C
- arenediyl when used without the "substituted” modifier refers to a divalent group, wherein the arenediyl group is attached with two ⁇ -bonds, with two aromatic carbon atoms as points of attachment, said carbon atoms forming part of one or more six-membered aromatic ring structure(s) wherein the ring atoms are all carbon, and wherein the monovalent group consists of no atoms other than carbon and hydrogen.
- arenediyl groups include:
- substituted arenediyl refers to a divalent group, wherein the arenediyl group is attached with two ⁇ -bonds, with two aromatic carbon atoms as points of attachment, said carbon atoms forming part of one or more six-membered aromatic rings structure(s), wherein the ring atoms are all carbon, and wherein the divalent group further has at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- aralkyl when used without the "substituted” modifier refers to the monovalent group -alkanediyl-aryl, in which the terms alkanediyl and aryl are each used in a manner consistent with the definitions provided above.
- Non-limiting examples of aralkyls are: phenylmethyl (benzyl, Bn), 1 -phenyl -ethyl, 2-phenyl-ethyl, indenyl and 2,3-dihydro- indenyl, provided that indenyl and 2,3-dihydro-indenyl are only examples of aralkyl in so far as the point of attachment in each case is one of the saturated carbon atoms.
- aralkyl When the term “aralkyl” is used with the “substituted” modifier, either one or both the alkanediyl and the aryl is substituted.
- substituted aralkyls are: (3-chlorophenyl)- methyl, 2-oxo-2-phenyl-ethyl (phenylcarbonylmethyl), 2-chloro-2-phenyl-ethyl, chromanyl where the point of attachment is one of the saturated carbon atoms, and tetrahydroquinolinyl where the point of attachment is one of the saturated atoms.
- heteroaryl when used without the “substituted” modifier refers to a monovalent group with an aromatic carbon atom or nitrogen atom as the point of attachment, said carbon atom or nitrogen atom forming part of an aromatic ring structure wherein at least one of the ring atoms is nitrogen, oxygen or sulfur, and wherein the monovalent group consists of no atoms other than carbon, hydrogen, aromatic nitrogen, aromatic oxygen and aromatic sulfur.
- Non-limiting examples of aryl groups include acridinyl, furanyl, imidazoimidazolyl, imidazopyrazolyl, imidazopyridinyl, imidazopyrimidinyl, indolyl, indazolinyl, methylpyridyl, oxazolyl, phenylimidazolyl, pyridyl, pyrrolyl, pyrimidyl, pyrazinyl, quinolyl, quinazolyl, quinoxalinyl, tetrahydroquinolinyl, thienyl, triazinyl, pyrrolopyridinyl, pyrrolopyrimidinyl, pyrrolopyrazinyl, pyrrolotriazinyl, pyrroloimidazolyl, chromeiiyl (where the point of attachment is one of the aromatic atoms), and chromanyl (where the point of attachment is one of the aromatic atoms
- substituted heteroaryl refers to a monovalent group with an aromatic carbon atom or nitrogen atom as the point of attachment, said carbon atom or nitrogen atom forming part of an aromatic ring structure wherein at least one of the ring atoms is nitrogen, oxygen or sulfur, and wherein the monovalent group further has at least one atom independently selected from the group consisting of non-aromatic nitrogen, non-aromatic oxygen, non aromatic sulfur F, Cl, Br, I, Si, and P.
- heteroarenediyl when used without the “substituted” modifier refers to a divalent group, wherein the heteroarenediyl group is attached with two ⁇ -bonds, with an aromatic carbon atom or nitrogen atom as the point of attachment, said carbon atom or nitrogen atom two aromatic atoms as points of attachment, said carbon atoms forming part of one or more six-membered aromatic ring structure(s) wherein the ring atoms are all carbon, and wherein the monovalent group consists of no atoms other than carbon and hydrogen.
- heteroarenediyl groups include:
- substituted heteroarenediyl refers to a divalent group, wherein the heteroarenediyl group is attached with two ⁇ -bonds, with two aromatic carbon atoms as points of attachment, said carbon atoms forming part of one or more six-membered aromatic rings structure(s), wherein the ring atoms are all carbon, and wherein the divalent group further has at least one atom independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- heteroarylkyl when used without the “substituted” modifier refers to the monovalent group -alkanediyl-heteroaryl, in which the terms alkanediyl and heteroaryl are each used in a manner consistent with the definitions provided above.
- Non-limiting examples of aralkyls are: pyridylmethyl, and thienylmethyl.
- acyl when used without the "substituted” modifier refers to a monovalent group with a carbon atom of a carbonyl group as the point of attachment, further having a linear or branched, cyclo, cyclic or acyclic structure, further having no additional atoms that are not carbon or hydrogen, beyond the oxygen atom of the carbonyl group.
- the groups, -CHO, -C(O)CH 3 (acetyl, Ac), -C(O)CH 2 CH 3 , -C(O)CH 2 CH 2 CH 3 , -C(O)CH(CH 3 ) 2 , -C(O)CH(CH 2 ) 2 , -C(O)C 6 H 5 , -C(O)C 6 H 4 CH 3 , -C(O)C 5 H 4 CH 2 CH 3 , -COC 6 H 3 (CH 3 ) 2 , and -C(O)CH 2 C 6 H 5 are non-limiting examples of acyl groups.
- acyl therefore encompasses, but is not limited to groups sometimes referred to as “alkyl carbonyl” and “aryl carbonyl” groups.
- substituted acyl refers to a monovalent group with a carbon atom of a carbonyl group as the point of attachment, further having a linear or branched, cyclo, cyclic or acyclic structure, further having at least one atom, in addition to the oxygen of the carbonyl group, independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- substituted acyl encompasses, but is not limited to, "heteroaryl carbonyl” groups.
- alkoxy when used without the "substituted” modifier refers to the group -OR, in which R is an alkyl, as that term is defined above.
- alkoxy groups include: -OCH 3 , -OCH 2 CH 3 , -OCH 2 CH 2 CH 3 , -OCH(CH 3 ) 2 , -OCH(CH 2 ) 2 , -O-cyclopentyl, and -O-cyclohexyl.
- substituted alkoxy refers to the group -OR, in which R is a substituted alkyl, as that term is defined above. For example, -OCH 2 CF 3 is a substituted alkoxy group.
- heteroaryloxy when used without the “substituted” modifier, refers to groups, defined as -OR, in which R is alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and acyl, respectively, as those terms are defined above.
- alkenyloxy, alkynyloxy, aryloxy, aralkyloxy and acyloxy is modified by "substituted,” it refers to the group -OR, in which R is substituted alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and acyl, respectively.
- alkylamino when used without the "substituted” modifier refers to the group -NHR, in which R is an alkyl, as that term is defined above.
- alkylamino groups include: -NHCH 3 , -NHCH 2 CH.,, -NHCH 2 CH 2 CH 3 , -NHCH(CH 3 ) 2 , -NHCH(CH 2 ) 2 , -NHCH 2 CH 2 CH 2 CH 3 , -NHCH(CH 3 )CH 2 CH 3 , -NHCH 2 CH(CH 3 ) 2 , -NHC(CH 3 ) 3 , -NH-cyclopentyl, and -NH-cyclohexyl.
- substituted alkylamino refers to the group -NHR, in which R is a substituted alkyl, as that term is defined above.
- R is a substituted alkyl
- -NHCH 2 CF 3 is a substituted alkylamino group.
- dialkylammo when used without the "substituted” modifier refers to the group -NRR', in which R and R' can be the same or different alkyl groups, or R and R' can be taken together to represent an alkanediyl having two or more saturated carbon atoms, at least two of which are attached to the nitrogen atom.
- Non-limiting examples of dialkylammo groups include: -NHC(CH 3 ) 3 , -N(CH 3 )CH 2 CH 3 , -N(CH 2 CH 3 ) 2 , N-pyrrolidinyl, and N- piperidinyl.
- substituted dialkylammo refers to the group -NRR', in which R and R' can be the same or different substituted alkyl groups, one of R or R' is an alkyl and the other is a substituted alkyl, or R and R' can be taken together to represent a substituted alkanediyl with two or more saturated carbon atoms, at least two of which are attached to the nitrogen atom.
- alkoxyamino refers to groups, defined as -NHR, in which R is alkoxy, alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and alkylsulfonyl, respectively, as those terms are defined above.
- R is alkoxy, alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and alkylsulfonyl, respectively, as those terms are defined above.
- a non-limiting example of an arylamino group is -NHC 6 H 5 .
- alkoxyamino, alkenylamino, alkynylamino, arylamino, aralkylamino, heteroarylamino, heteroaralkylamino and alkylsulfonylamino is modified by "substituted,” it refers to the group -NHR, in which R is substituted alkoxy, alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and alkylsulfonyl, respectively.
- amido (acylamino), when used without the “substituted” modifier, refers to the group -NHR, in which R is acyl, as that term is defined above.
- a non-limiting example of an acylamino group is -NHC(O)CH 3 .
- amido refers to groups, defined as -NHR, in which R is substituted acyl, as that term is defined above.
- the groups -NHC(O)OCH 3 and -NHC(O)NHCH 3 are non- limiting examples of substituted amido groups.
- alkenylimino alkynylimino
- arylimino aralkylimino
- fluoroalkyl when used without the "substituted” modifier refers to an alkyl, as that term is defined above, in which one or more fluorines have been substituted for hydrogens.
- the groups, -CH 2 F, -CF 3 , and -CH 2 CF 3 are non-limiting examples of fluoroalkyl groups.
- substituted fluoroalkyl refers to a non-aromatic monovalent group with a saturated carbon atom as the point of attachment, a linear or branched, cyclo, cyclic or acyclic structure, at least one fluorine atom, no carbon-carbon double or triple bonds, and at least one atom independently selected from the group consisting of N, O, Cl, Br, I, Si, P, and S.
- the following group is a non-limiting example of a substituted fluoroalkyl: -CFHOH.
- alkylphosphate when used without the "substituted” modifier refers to the group -OP(O)(OH)(OR), in which R is an alkyl, as that term is defined above.
- alkylphosphate groups include: -OP(O)(OH)(OMe) and -OP(O)(OH)(OEt).
- substituted alkylphosphate refers to the group -OP(O)(OH)(OR), in which R is a substituted alkyl, as that term is defined above.
- dialkylphosphate when used without the "substituted” modifier refers to the group -OP(O)(OR)(OR'), in which R and R' can be the same or different alkyl groups, or R and R' can be taken together to represent an alkanediyl having two or more saturated carbon atoms, at least two of which are attached via the oxygen atoms to the phosphorus atom.
- Non-limiting examples of dialkylphosphate groups include: -0P(0)(0Me) 2 , -OP(O)(OEt)(OMe) and -OP(O)(OEt) 2 .
- substituted dialkylphosphate refers to the group -OP(O)(OR)(OR'), in which R and R' can be the same or different substituted alkyl groups, one of R or R' is an alkyl and the other is a substituted alkyl, or R and R' can be taken together to represent a substituted alkanediyl with two or more saturated carbon atoms, at least two of which are attached via the oxygen atoms to the phosphorous.
- alkylthio when used without the "substituted” modifier refers to the group -SR, in which R is an alkyl, as that term is defined above.
- alkylthio groups include: -SCH 3 , -SCH 2 CH 3 , -SCH 2 CH 2 CH 3 , -SCH(CH 3 ) 2 , -SCH(CH 2 ) 2 , -S-cyclopentyl, and -S-cyclohexyl.
- substituted alkylthio refers to the group -SR, in which R is a substituted alkyl, as that term is defined above.
- -SCH 2 CF 3 is a substituted alkylthio group.
- alkenylthio when used without the “substituted” modifier, refers to groups, defined as -SR, in which R is alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and acyl, respectively, as those terms are defined above.
- alkenylthio, alkynylthio, arylthio, aralkylthio, heteroarylthio, heteroaralkylthio, and acylthio is modified by "substituted,” it refers to the group -SR, in which R is substituted alkenyl, alkynyl, aryl, aralkyl, heteroaryl, heteroaralkyl and acyl, respectively.
- thioacyl when used without the "substituted” modifier refers to a monovalent group with a carbon atom of a thiocarbonyl group as the point of attachment, further having a linear or branched, cyclo, cyclic or acyclic structure, further having no additional atoms that are not carbon or hydrogen, beyond the sulfur atom of the carbonyl group.
- the groups, -CHS, -C(S)CH 3 , -C(S)CH 2 CH 3 , -C(S)CH 2 CH 2 CH 3 , -C(S)CH(CH 3 ) 2 , -C(S)CH(CH 2 );, -C(S)C 6 H 5 , -C(S)C 6 H 4 CH 3 , -C(S)C 6 H 4 CH 2 CH 3 , -C(S)C 6 H 3 (CHs) 2 , and -C(S)CH 2 C6H5, are non-limiting examples of thioacyl groups.
- thioacyl therefore encompasses, but is not limited to, groups sometimes referred to as “alkyl thiocarbonyl” and “aryl thiocarbonyl” groups.
- substituted thioacyl refers to a radical with a carbon atom as the point of attachment, the carbon atom being part of a thiocarbonyl group, further having a linear or branched, cyclo, cyclic or acyclic structure, further having at least one atom, in addition to the sulfur atom of the carbonyl group, independently selected from the group consisting of N, O, F, Cl, Br, I, Si, P, and S.
- substituted thioacyl encompasses, but is not limited to, "heteroaryl thiocarbonyl” groups.
- alkylsulfonyl when used without the "substituted” modifier refers to the group -S(O) 2 R, in which R is an alkyl, as that term is defined above.
- alkylsulfonyl groups include: -S(O) 2 CH 3 , -S(O) 2 CH 2 CH 3 , -S(O) 2 CH 2 CH 2 CH 3 , -S(O) 2 CH(CH 3 ) 2 , -S(O) 2 CH(CH 2 ) 2 , -S(O) 2 -cyclopentyl, and -S(0) 2 -cyclohexyl,
- -S(O) 2 CH 3 -S(O) 2 CH 2 CH 3
- -S(O) 2 CH(CH 3 ) 2 -S(O) 2 CH(CH 2 ) 2
- -S(O) 2 -cyclopentyl -S(0) 2 -cyclohexyl
- substituted alkylsulfonyl refers to the group -S(O) 2 R, in which R is a substituted alkyl, as that term is defined above.
- R is a substituted alkyl
- -S(O) 2 CH 2 CF 3 is a substituted alkylsulfonyl group.
- alkenylsulfonyl alkynylsulfonyl
- arylsulfonyl alkenylsulfonyl
- aralkylsulfonyl when used without the “substituted” modifier, refers to groups, defined as -S(O) 2 R, in which R is alkenyl, alkynyl, aryl, aralkyl, heteroaryl, and heteroaralkyl, respectively, as those terms are defined above.
- alkenylsulfonyl, alkynylsulfonyl, arylsulfonyl, aralkylsulfonyl, heteroarylsulfonyl, and heteroaralkylsulfonyl is modified by "substituted,” it refers to the group -S(O) 2 R, in which R is substituted alkenyl, alkynyl, aryl, aralkyl, heteroaryl and heteroaralkyl, respectively.
- alkylsulfinyl when used without the "substituted” modifier refers to the group -S(O)R, in which R is an alkyl, as that term is defined above.
- alkylsulfinyl groups include: -S(O)CH 3 , -S(O)CH 2 CH 3 , -S(O)CH 2 CH 2 CH 3 , -S(O)CH(CH 3 ) 2 , -S(O)CH(CH 2 ) 2 , -S(0)-cyclopentyl, and -S(O)-cyclohexyl.
- substituted alkylsulfinyl refers to the group -S(O)R, in which R is a substituted alkyl, as that term is defined above.
- R is a substituted alkyl
- -S(O)CH 2 CF 3 is a substituted alkylsulfinyl group.
- alkenylsulfmyl refers to groups, defined as -S(O)R, in which R is alkenyl, alkynyl, aryl, aralkyl, heteroaryl, and heteroaralkyl, respectively, as those terms are defined above.
- alkenylsulfmyl, alkynylsulfinyl, arylsulfinyl, aralkylsulfmyl, heteroarylsulfinyl, and heteroaralkylsulfinyl is modified by "substituted,” it refers to the group -S(O)R, in which R is substituted alkenyl, alkynyl, aryl, aralkyl, heteroaryl and heteroaralkyl, respectively.
- alkylammonium when used without the "substituted” modifier refers to a group, defined as -NH 2 R + , -NHRR' " , or -NRRTf + , in which R, R' and R" are the same or different alkyl groups, or any combination of two of R, R' and R" can be taken together to represent an alkanediyl.
- Non-limiting examples of alkylammonium cation groups include: -NH 2 (CH 3 ) " , -NH 2 (CH 2 CH 3 )+, -NH 2 (CH 2 CH 2 CH 3 )+, -NH(CH 3 V, -NH(CH 2 CH 3 V, -NH(CH 2 CH 2 CH 3 V, -N(CH 3 V, -N(CH 3 )(CH 2 CH 3 V, "N(CH 3 ), (CH 2 CH 3 ) , -NH 2 C(CH 3 ) 3 ⁇ , -NH(cyclopentyl) 2 + , and -NH 2 (cyclohexylf .
- substituted alkylammonium refers -NH 2 R + , -NHRR'", or -NRR'R” + , in which at least one of R, R' and R" is a substituted alkyl or two of R, R' and R" can be taken together to represent a substituted alkanediyl. When more than one of R, R' and R" is a substituted alkyl, they can be the same of different.
- R, R' and R" that are not either substituted alkyl or substituted alkanediyl, can be either alkyl, either the same or different, or can be taken together to represent a alkanediyl with two or more carbon atoms, at least two of which are attached to the nitrogen atom shown in the formula.
- alkylsulfonium when used without the "substituted” modifier refers to the group -SRR' " , in which R and R' can be the same or different alkyl groups, or R and R' can be taken together to represent an alkanediyl.
- Non-limiting examples of alkylsulfonium groups include: -SH(CH 3 V -SH(CH 2 CH 3 ) + , -SH(CH 2 CH 2 CH 3 V -S(CH 3 V, -S(CH 2 CH 3 V, -S(CH 2 CH 2 CH 3 V, " SH(cyclopentyl) + , and -SH(cyclohexylV
- substituted alkylsulfonium refers to the group -SRR' * , in which R and R' can be the same or different substituted alkyl groups, one of R or R' is an alkyl and the other is a substituted alkyl, or R and R' can be taken together to represent a substituted alkanediyl.
- -SH(CH 2 CF 3 ) is a substituted alkylsulfonium group.
- alkylsilyl when used without the "substituted” modifier refers to a monovalent group, defined as -SiH 2 R, -SiHRR', or -SiRR'R", in which R, R' and R" can be the same or different alkyl groups, or any combination of two of R, R' and R" can be taken together to represent an alkanediyl.
- the groups, -SiH 2 CH 3 , -SiH(CH 3 V -Si(CH 3 ) 3 and -Si(CH 3 ) 2 C(CH 3 ) 3 are non-limiting examples of unsubstituted alkylsilyl groups.
- substituted alkylsilyl refers -SiH 2 R, -SiHRR', or -SiRR'R", in which at least one of R, R' and R" is a substituted alkyl or two of R, R' and R" can be taken together to represent a substituted alkanediyl. When more than one of R, R' and R" is a substituted alkyl, they can be the same of different.
- R, R' and R" that are not either substituted alkyl or substituted alkanediyl can be either alkyl, either the same or different, or can be taken together to represent a alkanediyl with two or more saturated carbon atoms, at least two of which are attached to the silicon atom.
- atoms making up the compounds of the present invention are intended to include all isotopic forms of such atoms.
- Isotopes include those atoms having the same atomic number but different mass numbers.
- isotopes of hydrogen include tritium and deuterium
- isotopes of carbon include 13 C and 14 C.
- one or more carbon atom(s) of a compound of the present invention may be replaced by a silicon atom(s).
- one or more oxygen atom(s) of a compound of the present invention may be replaced by a sulfur or selenium atom(s).
- a compound having a formula that is represented with a dashed bond is intended to include the formulae optionally having zero, one or more double bonds.
- the structure includes the structures , , , and As will be understood by a person of skill in the art, no one such ring atom forms part of more than one double bond.
- Any undefined valency on an atom of a structure shown in this application implicitly represents a hydrogen atom bonded to the atom.
- a ring structure shown with an unconnected "R" group indicates that any implicit hydrogen atom on that ring can be replaced with that R group.
- R group e.g., oxo, imino, thio, alkylidene, etc.
- any pair of implicit hydrogen atoms attached to one atom of that ring can be replaced by that R group.
- a "chiral auxiliary” refers to a removable chiral group that is capable of influencing the stereoselectivity of a reaction. Persons of skill in the art are familiar with such compounds, and many are commercially available.
- a "label” is any composition or moiety detectable by spectroscopic, photochemical, biochemical, immunochemical, or chemical means. Labels that may be employed in the present invention include radioactive labels (e.g., 32 P, 125 I, 14 C, 3 H, and 35 S) and fluorescent dyes (e.g., Cy3). An examples of a label that is not directly detected but is detected through the use of indirect methods is biotin.
- the term “about” is used to indicate that a value includes the inherent variation of error for the device, the method being employed to determine the value, or the variation that exists among the study subjects.
- the terms “comprise,” “have” and “include” are open-ended linking verbs. Any forms or tenses of one or more of these verbs, such as “comprises,” “comprising,” “has,” “having,” “includes” and “including,” are also open-ended. For example, any method that "comprises,” “has” or “includes” one or more steps is not limited to possessing only those one or more steps and also covers other unlisted steps.
- the term “effective,” as that term is used in the specification and/or claims, means adequate to accomplish a desired, expected, or intended result.
- hydrate when used as a modifier to a compound means that the compound has less than one (e.g., hemihydrate), one (e.g., monohydrate), or more than one (e.g. , dihydrate) water molecules associated with each compound molecule, such as in solid forms of the compound.
- IC50 refers to an inhibitory dose which is 50% of the maximum response obtained.
- An "isomer" of a first compound is a separate compound in which each molecule contains the same constituent atoms as the first compound, but where the configuration of those atoms in three dimensions differs.
- the term "patient” or “subject” refers to a living mammalian organism, such as a human, monkey, cow, sheep, goat, dog, cat, mouse, rat, guinea pig, or transgenic species thereof.
- the patient or subject is a primate.
- Non-limiting examples of human subjects are adults, juveniles, infants and fetuses.
- “Pharmaceutically acceptable” means that which is useful in preparing a pharmaceutical composition that is generally safe, non-toxic and neither biologically nor otherwise undesirable and includes that which is acceptable for veterinary use as well as human pharmaceutical use.
- “Pharmaceutically acceptable salts” means salts of compounds of the present invention which are pharmaceutically acceptable, as defined above, and which possess the desired pharmacological activity. Such salts include acid addition salts formed with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like; or with organic acids such as 1 ,2-ethanedisulfonic acid, 2-hydroxyethanesulfonic acid, 2-naphthalenesulfonic acid, 3-phenylpropionic acid,
- Pharmaceutically acceptable salts also include base addition salts which may be formed when acidic protons present are capable of reacting with inorganic or organic bases.
- Acceptable inorganic bases include sodium hydroxide, sodium carbonate, potassium hydroxide, aluminum hydroxide and calcium hydroxide.
- Acceptable organic bases include ethanolamine, diethanolamine, triethanolamine, tromethamine, N-methylglucamine and the like. It should be recognized that the particular anion or cation forming a part of any salt of this invention is not critical, so long as the salt, as a whole, is pharmacologically acceptable. Additional examples of pharmaceutically acceptable salts and their methods of preparation and use are presented in Handbook of Pharmaceutical Salts: Properties, and Use (P. H. Stahl & C. G. Wermuth eds., Verlag Helvetica Chimica Acta, 2002),
- progenitantly one enantiomer means that a compound contains at least about 85% of one enantiomer, or more preferably at least about 90% of one enantiomer, or even more preferably at least about 95% of one enantiomer, or most preferably at least about 99% of one enantiomer.
- the phrase "substantially free from other optical isomers” means that the composition contains at most about 15% of another enantiomer or diastereomer, more preferably at most about 10% of another enantiomer or diastereomer, even more preferably at most about 5% of another enantiomer or diastereomer, and most preferably at most about 1% of another enantiomer or diastereomer.
- Prevention includes: (1) inhibiting the onset of a disease in a subject or patient which may be at risk and/or predisposed to the disease but does not yet experience or display any or all of the pathology or symptomatology of the disease, and/or (2) slowing the onset of the pathology or symptomatology of a disease in a subject or patient which may be at risk and/or predisposed to the disease but does not yet experience or display any or all of the pathology or symptomatology of the disease.
- Prodrug means a compound that is convertible in vivo metabolically into an inhibitor according to the present invention.
- the prodrug itself may or may not also have activity with respect to a given target protein.
- a compound comprising a hydroxy group may be administered as an ester that is converted by hydrolysis in vivo to the hydroxy compound.
- esters that may be converted in vivo into hydroxy compounds include acetates, citrates, lactates, phosphates, tartrates, malonates, oxalates, salicylates, propionates, succinates, fumarates, maleates, methylene-bis- ⁇ -hydroxynaphthoate, gentisates, isethionates, di-/>-toluoyltartrates, methanesulfonates, ethanesulfonates, benzenesulfonates, p-toluenesulfonates, cyclohexylsulfamates, quinates, esters of amino acids, and the like.
- a compound comprising an amine group may be administered as an amide that is converted by hydrolysis in vivo to the amine compound.
- saturated when referring to an atom means that the atom is connected to other atoms only by means of single bonds.
- a “stereoisomer” or “optical isomer” is an isomer of a given compound in which the same atoms are bonded to the same other atoms, but where the configuration of those atoms in three dimensions differs.
- “Enantiomers” are stereoisomers of a given compound that are mirror images of each other, like left and right hands.
- Diastereomers are stereoisomers of a given compound that are not enantiomers.
- stereocenter or axis of chirality for which stereochemistry has not been defined, that stereocenter or axis of chirality can be present in its R form, S form, or as a mixture of the R and S forms, including racemic and non-racemic mixtures.
- Substituent convertible to hydrogen in vivo means any group that is convertible to a hydrogen atom by enzymological or chemical means including, but not limited to, hydrolysis and hydrogenolysis.
- hydrolyzable groups such as acyl groups, groups having an oxycarbonyl group, amino acid residues, peptide residues, onitrophenylsulfenyl, trimethylsilyl, tetrahydro-pyranyl, diphenylphosphinyl, and the like.
- acyl groups include formyl, acetyl, trifluoroacetyl, and the like.
- groups having an oxycarbonyl group include ethoxycarbonyl, /er/-butoxycarbonyl (-C(O)OC(CH 3 ) . ⁇ ), benzyloxycarbonyl, /7-methoxybenzyloxycarbonyl, vinyloxycarbonyl, ⁇ -(p- toluenesulfonyl)ethoxycarbonyl, and the like.
- Suitable amino acid residues include, but are not limited to, residues of GIy (glycine), Ala (alanine), Arg (arginine), Asn (asparagine), Asp (aspartic acid), Cys (cysteine), GIu (glutamic acid), His (histidine), He (isoleucine), Leu (leucine), Lys (lysine), Met (methionine), Phe (phenylalanine), Pro (proline), Ser (serine), Thr (threonine), Trp (tryptophan), Tyr (tyrosine), VaI (valine), Nva (norvaline), Hse (homoserine), 4-Hyp (4-hydroxyproline), 5-Hyl (5-hydroxylysine), Orn (ornithine) and ⁇ -Ala.
- suitable amino acid residues also include amino acid residues that are protected with a protecting group.
- suitable protecting groups include those typically employed in peptide synthesis, including acyl groups (such as formyl and acetyl), arylmethyloxycarbonyl groups (such as benzyloxycarbonyl and p-nitrobenzyloxycarbonyl), f ⁇ r/-butoxycarbonyl groups (-C(O)OC(CH 3 ) 3 ), and the like.
- Suitable peptide residues include peptide residues comprising two to five, and optionally amino acid residues. The residues of these amino acids or peptides can be present in stereochemical configurations of the D-form, the L-form or mixtures thereof.
- amino acid or peptide residue may have an asymmetric carbon atom.
- suitable amino acid residues having an asymmetric carbon atom include residues of Ala, Leu, Phe, Trp, Nva, VaI, Met, Ser, Lys, Thr and Tyr.
- Peptide residues having an asymmetric carbon atom include peptide residues having one or more constituent amino acid residues having an asymmetric carbon atom.
- suitable amino acid protecting groups include those typically employed in peptide synthesis, including acyl groups (such as formyl and acetyl), arylmethyloxycarbonyl groups (such as benzyloxycarbonyl and p-nitrobenzyloxycarbonyl), /ert-butoxycarbonyl groups (-C(O)OC(CH 3 ) 3 ), and the like.
- acyl groups such as formyl and acetyl
- arylmethyloxycarbonyl groups such as benzyloxycarbonyl and p-nitrobenzyloxycarbonyl
- /ert-butoxycarbonyl groups /ert-butoxycarbonyl groups (-C(O)OC(CH 3 ) 3 ), and the like.
- substituents "convertible to hydrogen in vivo" include reductively eliminable hydrogenolyzable groups.
- Suitable reductively eliminable hydrogenolyzable groups include, but are not limited to, arylsulfonyl groups (such as o-toluenesulfonyl); methyl groups substituted with phenyl or benzyloxy (such as benzyl, trityl and benzyloxymethyl); arylmethoxycarbonyl groups (such as benzyloxycarbonyl and o-methoxy-benzyloxycarbonyl); and haloethoxycarbonyl groups (such as ⁇ , ⁇ , ⁇ -trichloroethoxycarbonyl and ⁇ -iodoethoxycarbonyl).
- arylsulfonyl groups such as o-toluenesulfonyl
- methyl groups substituted with phenyl or benzyloxy such as benzyl, trityl and benzyloxymethyl
- arylmethoxycarbonyl groups such as benzyloxy
- “Therapeutically effective amount” or “pharmaceutically effective amount” means that amount which, when administered to a subject or patient for treating a disease, is sufficient to effect such treatment for the disease.
- therapeutic benefit or “therapeutically effective” as used throughout this application refers to anything that promotes or enhances the well-being of the subject with respect to the medical treatment of a condition. This includes, but is not limited to, a reduction in the onset, frequency, duration, or severity of the signs or symptoms of a disease.
- a therapeutically effective amount of a compound of the present invention that is, a Wnt protein signalling inhibitor
- inhibiting includes any measurable decrease or complete inhibition to achieve a desired result. For example, there may be a decrease of 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 99%, or more, or any range derivable therein, reduction of activity compared to normal.
- a cancer patient may experience a reduction in tumor size.
- Treatment includes (1) inhibiting a disease in a subject or patient experiencing or displaying the pathology or symptomatology of the disease (e.g., arresting further development of the pathology and/or symptomatology), (2) ameliorating a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease (e.g., reversing the pathology and/or symptomatology), and/or (3) effecting any measurable decrease in a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease.
- contacted and “exposed,” when applied to a cell are used herein to describe the process by which a compound of the present invention is administered or delivered to a target cell, or are placed in direct juxtaposition with the target cell.
- administered and “delivered” are used interchangeably with “contacted” and “exposed.”
- water soluble means that the compound dissolves in water at least to the extent of 0.010 mole/liter or is classified as soluble according to literature precedence.
- DMSO dimethyl sulfoxide
- NO nitric oxide
- iNOS inducible nitric oxide synthase
- COX-2 cyclooxygenase-2
- NGF nerve growth factor
- IBMX isobutylmethylxanthine
- FBS fetal bovine serum
- GPDH glycerol 3- phosphate dehydrogenase
- RXR retinoid X receptor
- TGF- ⁇ transforming growth factor- ⁇
- IFN ⁇ or IFN- ⁇ interferon- ⁇
- LPS bacterial endotoxic lipopolysaccharide
- TNF ⁇ or TNF- ⁇ tumor necrosis factor- ⁇
- IL- l ⁇ interleukin-l ⁇
- GAPDH glyceraldehyde-3-phosphate dehydrogenase
- MTT 3-[4,5-dimethylmiazol-2-yl]-2,5-diphenylt
- “derivative” refers to a chemically-modified compound that still retains the desired effects of the compound prior to the chemical modification.
- Non-limiting examples of the types of modifications that can be made to the compounds and structures disclosed herein include the addition or removal of lower unsubstituted alkyls such as methyl, ethyl, propyl, or substituted lower alkyls such as hydroxymethyl or aminomethyl groups; carboxyl groups and carbonyl groups; hydroxyls; nitro, amino, amide, imide, and azo groups; sulfate, sulfonate, sulfono, sulfhydryl, sulfenyl, sulfonyl, sulfoxido, sulfonamide, phosphate, phosphono, phosphoryl groups, and halide substituents.
- lower unsubstituted alkyls such as methyl, ethyl, propyl, or substituted lower alkyls such as hydroxymethyl or aminomethyl groups
- carboxyl groups and carbonyl groups hydroxyls; nitro, amino, amide, imide,
- Additional modifications can include an addition or a deletion of one or more atoms of the atomic framework, for example, substitution of an ethyl by a propyl, or substitution of a phenyl by a larger or smaller aromatic group.
- heteroatoms such as N, S, or O can be substituted into the structure instead of a carbon atom.
- Prodrugs and solvates of the compounds of the present invention are also contemplated herein.
- the term "prodrug,” as used herein, is understood as being a compound which, upon administration to a subject, such as a mammal, undergoes chemical conversion by metabolic or chemical processes to yield a compound any of the formulas herein, or a salt and/or solvate thereof.
- Solvates of the compounds of the present invention are preferably hydrates.
- protecting group refers to a moiety attached to a functional group to prevent an otherwise unwanted reaction of that functional group.
- the term “functional group” generally refers to how persons of skill in the art classify chemically reactive groups.
- Examples of functional groups include hydroxyl, amine, sulfhydryl, amide, carboxyl, carbonyl, etc.
- Protecting groups are well-known to those of skill in the art. Non-limiting exemplary protecting groups fall into categories such as hydroxy protecting groups, amino protecting groups, sulfhydryl protecting groups and carbonyl protecting groups. Such protecting groups may be found in Greene and Wuts, 1999, incorporated herein by reference in its entirety.
- the Wnt protein signalling inhibitors described herein are also contemplated as protected by one or more protecting groups — that is, the inhibitors are contemplated in their "protected form.”
- Compounds of the present invention may contain one or more asymmetric centers and thus can occur as racemates and racemic mixtures, single enantiomers, diastereomeric mixtures and individual diastereomers. In certain embodiments, a single diastereomer is present. All possible stereoisomers of the compounds of the present invention are contemplated as being within the scope of the present invention. However, in certain aspects, particular diastereomers are contemplated.
- the chiral centers of the compounds of the present invention can have the S- or the ⁇ -configuration, as defined by the IUPAC 1974 Recommendations. In certain aspects, certain compounds of the present invention may comprise S- or R -configurations at particular carbon centers.
- Synthetic techniques that may be used to prepare certain compounds of the present invention are provided in the Examples section. Other synthetic techniques to prepare compounds of the present invention as well as derivatives are well-known to those of skill in the art. For example, Smith and March, 2001 discuss a wide variety of synthetic transformations, reaction conditions, and possible pitfalls relating thereto. Methods discussed therein may be adapted to prepare compounds of the present invention from commerically available starting materials.
- Solvent choices for preparing compounds of the present invention will be known to one of ordinary skill in the art. Solvent choices may depend, for example, on which one(s) will facilitate the solubilizing of all the reagents or, for example, which one(s) will best facilitate the desired reaction (particularly when the mechanism of the reaction is known). Solvents may include, for example, polar solvents and non-polar solvents. Solvents choices include, but are not limited to, tetrahydrofuran, dimethylformamide, dimethylsulfoxide, dioxane, methanol, ethanol, hexane, methylene chloride and acetonitrile. More than one solvent may be chosen for any particular reaction or purification procedure. Water may also be admixed into any solvent choice. Further, water, such as distilled water, may constitute the reaction medium instead of a solvent.
- compositions of the present invention comprise an effective amount of one or more candidate substances (e.g., a Wnt protein signalling inhibitor) or additional agents dissolved or dispersed in a pharmaceutically acceptable carrier.
- candidate substances e.g., a Wnt protein signalling inhibitor
- additional agents dissolved or dispersed in a pharmaceutically acceptable carrier.
- pharmaceutically acceptable refers to molecular entities and compositions that do not produce an adverse, allergic or other untoward reaction when administered to an animal, such as, for example, a human, as appropriate.
- the preparation of a pharmaceutical composition that contains at least one candidate substance or additional active ingredient will be known to those of skill in the art in light of the present disclosure, as exemplified by Remington's Pharmaceutical Sciences, 18th Ed. Mack Printing Company, 1990, incorporated herein by reference.
- pharmaceutically acceptable carrier includes any and all solvents, dispersion media, coatings, surfactants, antioxidants, preservatives (e.g., antibacterial agents, antifungal agents), isotonic agents, absorption delaying agents, salts, preservatives, drugs, drug stabilizers, gels, binders, excipients, disintegration agents, lubricants, sweetening agents, flavoring agents, dyes, such like materials and combinations thereof, as would be known to one of ordinary skill in the art (see, for example, Remington's Pharmaceutical Sciences, pp 1289-1329, 1990).
- the candidate substance may comprise different types of carriers depending on whether it is to be administered in solid, liquid or aerosol form, and whether it needs to be sterile for such routes of administration as injection.
- Compounds of the present invention may be administered orally, intraadiposally, intraarterially, intraarticularly, intracranially, intradermally, intralesionally, intramuscularly, intranasally, intraocularally, intrapericardially, intraperitoneally, intrapleurally, intraprostaticaly, intrarectally, intrathecally, intratracheally, intratumorally, intraumbilically, intravaginally, intravenously, intravesicularlly, intravitreally, liposomally, locally, mucosally, orally, parenterally, rectally, subconjunctival, subcutaneously, sublingually, topically, transbuccally, transdermally, vaginally, in cremes, in lipid compositions, via a catheter, via a lavage, via continuous infusion, via infusion, via inhalation, via injection, via local delivery, via localized perfusion, bathing target cells directly, or by other method or any combination of the foregoing as would be known to one of
- the composition is administered to a subject using a drug delivery device.
- Any drug delivery device is contemplated for use in delivering a pharmaceutically effective amount of a Wnt protein signalling inhibitor.
- the actual dosage amount of a composition of the present invention administered to an animal patient can be determined by physical and physiological factors such as body weight, severity of condition, the type of disease being treated, previous or concurrent therapeutic interventions, idiopathy of the patient and on the route of administration. The practitioner responsible for administration will typically determine the concentration of active ingredient(s) in a composition and appropriate dose(s) for the individual subject.
- the dose can be repeated as needed as determined by those of ordinary skill in the art.
- a single dose is contemplated.
- two or more doses are contemplated.
- the time interval between doses can be any time interval as determined by those of ordinary skill in the art.
- the time interval between doses may be about 1 hour to about 2 hours, about 2 hours to about 6 hours, about 6 hours to about 10 hours, about 10 hours to about 24 hours, about 1 day to about 2 days, about 1 week to about 2 weeks, or longer, or any time interval derivable within any of these recited ranges.
- compositions may comprise, for example, at least about 0.1% of a Wnt protein signalling inhibitor.
- the Wnt protein signalling inhibitor may comprise between about 2% to about 75% of the weight of the unit, or between about 25% to about 60%, for example, and any range derivable therein.
- a dose may also comprise from about 1 microgram/kg/body weight, about 5 microgram/kg/body weight, about 10 microgram/kg/body weight, about 50 microgram/kg/body weight, about 100 microgram/kg/body weight, about 200 microgram/kg/body weight, about 350 microgram/kg/body weight, about 500 microgram/kg/body weight, about 1 milligram/kg/body weight, about 5 milligram/kg/body weight, about 10 milligram/kg/body weight, about 50 milligram/kg/body weight, about 100 milligram/kg/body weight, about 200 milligram/kg/body weight, about 350 milligram/kg/body weight, about 500 milligram/kg/body weight, to about 1000 mg/kg/body weight or more per administration, and any range derivable therein, hi non-limiting examples of a derivable range from the numbers listed herein, a range of about 5 mg/kg/body weight to about 100 mg/kg/body weight, about 5 microgram/kg/body weight
- the composition may comprise various antioxidants to retard oxidation of one or more component. Additionally, the prevention of the action of microorganisms can be brought about by preservatives such as various antibacterial and antifungal agents, including but not limited to parabens (e.g., methylparabens, propylparabens), chlorobutaiiol, phenol, sorbic acid, thimerosal, or combinations thereof.
- the Wnt protein signalling inhibitor may be formulated into a composition, such as a pharmaceutical composition, in a free base, neutral, or salt form. Pharmaceutically acceptable salts are described herein.
- a carrier can be a solvent or dispersion medium comprising but not limited to, water, ethanol, polyol (e.g., glycerol, propylene glycol, liquid polyethylene glycol, etc.), lipids (e.g. , triglycerides, vegetable oils, liposomes) and combinations thereof.
- the proper fluidity can be maintained, for example, by the use of a coating, such as lecithin; by the maintenance of the required particle size by dispersion in carriers such as, for example liquid polyol or lipids; by the use of surfactants such as, for example hydroxypropylcellulose; or combinations thereof such methods.
- isotonic agents such as, for example, sugars, sodium chloride, or combinations thereof.
- nasal solutions are usually aqueous solutions designed to be administered to the nasal passages in drops or sprays.
- Nasal solutions are prepared so that they are similar in many respects to nasal secretions, so that normal ciliary action is maintained.
- the aqueous nasal solutions usually are isotonic or slightly buffered to maintain a pH of about 5.5 to about 6.5.
- antimicrobial preservatives similar to those used in ophthalmic preparations, drugs, or appropriate drug stabilizers, if required, may be included in the formulation.
- various commercial nasal preparations are known and include drugs such as antibiotics or antihistamines.
- the candidate substance is prepared for administration by such routes as oral ingestion.
- the solid composition may comprise, for example, solutions, suspensions, emulsions, tablets, pills, capsules (e.g., hard or soft shelled gelatin capsules), sustained release formulations, buccal compositions, troches, elixirs, suspensions, syrups, wafers, or combinations thereof.
- Oral compositions may be incorporated directly with the food of the diet.
- carriers for oral administration comprise inert diluents (e.g., glucose, lactose, or mannitol), assimilable edible carriers or combinations thereof.
- the oral composition may be prepared as a syrup or elixir.
- a syrup or elixir may comprise, for example, at least one active agent, a sweetening agent, a preservative, a flavoring agent, a dye, a preservative, or combinations thereof.
- an oral composition may comprise one or more binders, excipients, disintegration agents, lubricants, flavoring agents, or combinations thereof.
- a composition may comprise one or more of the following: a binder, such as, for example, gum tragacanth, acacia, cornstarch, gelatin or combinations thereof; an excipient, such as, for example, dicalcium phosphate, mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, magnesium carbonate or combinations thereof; a disintegrating agent, such as, for example, corn starch, potato starch, alginic acid or combinations thereof; a lubricant, such as, for example, magnesium stearate; a sweetening agent, such as, for example, sucrose, lactose, saccharin or combinations thereof; a flavoring agent, such as, for example peppermint, oil of wintergreen, cherry flavoring, orange flavoring, etc.; or combinations thereof the foregoing.
- a binder such as, for example, gum tragacanth, acacia, cornstarch, gelatin or combinations thereof
- an excipient such as
- the dosage unit form When the dosage unit form is a capsule, it may contain, in addition to materials of the above type, carriers such as a liquid carrier. Various other materials may be present as coatings or to otherwise modify the physical form of the dosage unit. For instance, tablets, pills, or capsules may be coated with shellac, sugar, or both.
- Sterile injectable solutions may be prepared by incorporating a compound of the present invention in the required amount in the appropriate solvent with various of the other ingredients enumerated above, as required, followed by filtered sterilization.
- dispersions are prepared by incorporating the various sterilized active ingredients into a sterile vehicle which contains the basic dispersion medium and/or the other ingredients.
- certain methods of preparation may include vacuum-drying or freeze-drying techniques which yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered liquid medium thereof.
- the liquid medium should be suitably buffered if necessary and the liquid diluent ⁇ e.g., water) first rendered isotonic prior to injection with sufficient saline or glucose.
- the liquid diluent e.g., water
- the preparation of highly concentrated compositions for direct injection is also contemplated, where the use of DMSO as solvent is envisioned to result in extremely rapid penetration, delivering high concentrations of the active agents to a small area.
- the composition should be stable under the conditions of manufacture and storage, and preserved against the contaminating action of microorganisms, such as bacteria and fungi. It will be appreciated that endotoxin contamination should be kept minimally at a safe level, for example, less that 0.5 ng/mg protein.
- prolonged absorption of an injectable composition can be brought about by the use in the compositions of agents delaying absorption, such as, for example, aluminum monostearate, gelatin, or combinations thereof.
- agents delaying absorption such as, for example, aluminum monostearate, gelatin, or combinations thereof.
- the inhibitor may be combined with another therapy, such as another agent that combats and/or prevents cancer, osteopetrosis, a degenerative disease, or type II diabetes.
- Wnt protein signalling inhibitors of the present invention may be provided in a combined amount with an effective amount another agent that is known to reduce tumor size.
- combination therapy of the present invention may be used in vitro or in vivo. These processes may involve administering the agents at the same time or within a period of time wherein separate administration of the substances produces a desired therapeutic benefit. This may be achieved by contacting the cell, tissue, or organism with a single composition or pharmacological formulation that includes two or more agents, or by contacting the cell with two or more distinct compositions or formulations, wherein one composition includes one agent and the other includes another.
- the compounds of the present invention may precede, be co-current with and/or follow the other agents by intervals ranging from minutes to weeks.
- the agents are applied separately to a cell, tissue or organism, one would generally ensure that a significant period of time did not expire between the time of each delivery, such that the agents would still be able to exert an advantageously combined effect on the cell, tissue or organism.
- one may contact the cell, tissue or organism with two, three, four or more modalities substantially simultaneously (i.e., within less than about a minute) as the candidate substance.
- one or more agents may be administered about 1 minute, about 5 minutes, about 10 minutes, about 20 minutes about 30 minutes, about 45 minutes, about 60 minutes, about 2 hours, about 3 hours, about 4 hours, about 5 hours, about 6 hours, about 7 hours about 8 hours, about 9 hours, about 10 hours, about 11 hours, about 12 hours, about 13 hours, about 14 hours, about 15 hours, about 16 hours, about 17 hours, about 18 hours, about 19 hours, about 20 hours, about 21 hours, about 22 hours, about 22 hours, about 23 hours, about 24 hours, about 25 hours, about 26 hours, about 27 hours, about 28 hours, about 29 hours, about 30 hours, about 31 hours, about 32 hours, about 33 hours, about 34 hours, about 35 hours, about 36 hours, about 37 hours, about 38 hours, about 39 hours, about 40 hours, about 41 hours, about 42 hours, about 43 hours, about 44 hours, about 45 hours, about 46 hours, about 47 hours, about 48 hours, about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days, about 7 hours about 8
- an anti-cancer agent may be used in combination therapy with Wnt protein signalling inhibitors of the present invention.
- an "anti-cancer" agent is capable of negatively affecting cancer in a subject, for example, by killing one or more cancer cells, inducing apoptosis in one or more cancer cells, reducing the growth rate of one or more cancer cells, reducing the incidence or number of metastases, reducing tumor size, inhibiting tumor growth, reducing the blood supply to a tumor or one or more cancer cells, promoting an immune response against one or more cancer cells or a tumor, preventing or inhibiting the progression of a cancer, or increasing the lifespan of a subject with a cancer.
- Anti-cancer agents are well-known in the art and include, for example, chemotherapy agents (chemotherapy), radiotherapy agents (radiotherapy), a surgical procedure, immune therapy agents (immunotherapy), genetic therapy agents (gene therapy), reoviral therapy, hormonal therapy, other biological agents (biotherapy), and/or alternative therapies.
- Osteopetrosis therapy Osteopetrosis, also known as marble bone disease and Albers-Schonberg disease, is an extremely rare inherited disorder whereby the bones harden, becoming denser, in contrast to the more prevalent osteomalacia, in which the bones soften.
- Bone marrow transplant therapy may be combined with administration of Wnt protein signalling inhibitors of the present invention to treat or prevent osteopetrosis.
- Other treatments targeting osteopetrosis that may be combined with Wnt protein signalling inhibitors described herein include those disclosed in the following documents, each of which is incorporated herein by reference: U.S. Patent Nos.
- degenerative diseases may be treated using Wnt protein signalling inhibitors of the present invention. Accordingly, other treatments that target degenerative diseases may be combined with administration of the Wnt protein signalling inhibitors.
- degenerative diseases include type II diabetes and age-related impairment of tissue repair.
- Type II diabetes therapy Type II diabetes is a chronic, progressive disease that has no clearly established cure.
- Treatment options that may be combined with Wnt protein signalling inhibitor administration include exercise, diet management to control the intake of glucose, and use of anti-diabetic drugs ⁇ e.g., metformin, phenformin, repaglinide, nateglinide, rosiglitazone, pioglitazone or miglitol).
- anti-diabetic drugs e.g., metformin, phenformin, repaglinide, nateglinide, rosiglitazone, pioglitazone or miglitol.
- Wnt protein signalling inhibition has been implicated in, for example, muscle regeneration (Brack et al, 2007).
- Therapies pertaining to age-related impairment of tissue repair include, for example, gene therapy, such as described by Barton- Davis et al. (1998; incorporated herein by reference) and drugs described by Lynch (2004; incorporated herein by reference).
- L-Wnt-STF cells were generated by transfecting L-Wnt cells (ATCC) with SuperTopFlash (STF; provided by R. Moon) and SV- 40 Renilla luciferase plasmids and selecting for clones resistant to G418 and Zeocin.
- Constructs for expression of Shh and Wnt3A were provided by P. Beachy, and Notch intracellular domain (NICD) by R. Kopan.
- Notch reporter construct was provided by J. LaBorda, Wnt 1 and Wnt 2 expression constructs were purchased from OpenBiosystems and are part of the MGC Clone collection.
- the Wnt-GL expression construct was generated by ligating Wnt3A coding sequence to GL (lacking the signal sequence; AA15-185) using an engineered Xbal site.
- CMV-GL was generated by inserting the CMV promoter into the pGluc-Basic vector (New England Biolabs).
- Expression constructs for mPorc-myc, hAxin2- myc and hAxin ⁇ DIX were engineered using PCR-based cloning and mutagenesis strategies.
- RNAi experiments were purchased from either Qiagen (GS K3 ⁇ , CKl ⁇ , PPl, PP2CA, PP2CB, KAP, WTX, ASEF, DLGl) or from Dharmacon (APC, Axin2). Biochemical studies .
- Biochemical studies involving L-Wnt-STF or DLD-I cells were performed in either 48- or 6-well format with IWR (10 ⁇ M) or IWP (5 ⁇ M) compounds and/or cycloheximide (100 ⁇ M) in a 48 hr assay period.
- Targeting of APC using RNAi in L- cells was achieved by transfecting cells with SMARTPool APC siRNAs (50 nM; Dharmacon).
- E-cadherin depletion studies were performed at 4 0 C using DLD-I cells lysed in PBS/1% NP-40/protease inhibitors
- murine Porcupine (C isoform) and human Wnt3A-myc were transfected into HEK 293 cells (6 well format, 400K cells/well) as appropriate using Effectene transfection reagent (QIAGEN). After 48 hrs of incubation, cells were lysed for 15 min, RT with PL buffer (distilled water, 10 mM tris-HCl, 150 mM NaCl)/l% TritionX-114.
- PL buffer distilled water, 10 mM tris-HCl, 150 mM NaCl
- Lysate was briefly chilled on ice, pelleted for 10 min 4 0 C, and the supernatant combined with an equal volume of PL buffer/3.5% TX-114. Solutions were rotated for 15 min at 4°C, placed at 37°C for 5 min followed by an additional centrifugation for 5 min at 200Og, RT. Distinct phases were collected and combined with PL buffer to a total volume of 1 mL. Samples were chilled on ice, ConA sepharose (GE Healthcare) added, and samples rotated for 2 hrs at 4°C. Beads washed 2x with PL buffer, and a Western blot performed with eluted proteins using an anti-c-myc antibody.
- cell lysate (PBS/1 %NP-40) derived from HEK293 cells transfected with the Porc-myc construct was incubated with either DMSO, linker (165 ⁇ M), IWP-biotin (165 ⁇ M), or IWP-biotin (165 ⁇ M) + IWP3 (585 ⁇ M) and rotated for 30 min prior to addition of NeutrAvidin agarose resin (Pierce) and an additional 20 in rotation at RT. Resin was then washed lysis buffer and protein eluted with sample loading buffer.
- Example 2 Identification of Wnt Protein Signalling Inhibitors
- a high stringency cell-based screening strategy identified small molecular modulators of the Wnt/ ⁇ -catenin pathway from a -200K synthetic chemical library from U.T. Southwestern (Dallas, TX) (UTSW) (FIG. 1). This assay may be employed to identify Wnt- protein signalling inhibitors as well as chemicals that can increase Wnt/ ⁇ -catenin activity.
- Wnt3A-containing conditioned medium prepared following the protocol provided ATCC, was applied to HEK293 cells transiently transfected with STF and control reporters.
- NIH- 3T3 cells or L-cells, respectively were transiently transfected with indicated reporter constructs and immediately incubated with compounds. Luciferase activities were measured 24 hrs later.
- the "Wnt secretion test” was performed in L-cells transiently transfected with the Wnt-GL expression construct and immediately incubated with compounds. Culture medium and cellular lysate were analyzed 48 hrs later for GL activity. Assays used to calculate IC50's for compounds were performed as before in L-Wnt-STF cells.
- FIG. 9A four inhibited Wnt secretion as determined using a Wnt-lucif erase fusion protein (FIG. 1 and FIG. 9). Based on the results from these secondary tests, seven compounds were identified that act as inhibitors of Wnt response (IWRs) and four compounds were identified that act as inhibitors of Wnt production (IWPs; FIG. 1).
- IWPs all share the same core chemical structure
- two different classes of IWRs could be identified based on structural similarities (FIG. 2A and FIG. 2B).
- the IWPs are more potent pathway antagonists than those in the strongest class of IWRs (-40 nM vs —200 iiM, respectively).
- the site of action for each compound was generally localized (FIG. 3).
- IWPs blocked all Wnt-dependent biochemical changes that were assayed (phosphorylation of the LRP6 receptor and Dvl2, and ⁇ -catenin accumulation; FIG. 3).
- IWR compounds appear to only affect ⁇ - catenin levels suggesting they target regulatory events downstream of LRP6 and Dvl2.
- Example 3 Synthesis and Characterization Synthesis of IWR-I, IWP-2, IWR-1-PEG-Biotin, IWP-PEG-Biotin, and IWR-Cy3 were carried out as described in FIG. 12 and FIG. 13. Exemplarary characterization data of some of the compounds disclosed herein is provided below. 1 H-NMR spectra for compounds IWR-8, 9, 10, 1 1, 12, 13, 14, 15, 18 and 19 are provided in FIGS. 15-24, respectively.
- Example 4 Inhibitors of Wnt Production (IWPs) Target the Porcupine Acyltransferase
- IWR compounds Down-regulate ⁇ -catenin Protein Levels By Stabilizing the Axin2 Destruction Complex Based on biochemical evidence, IWR compounds likely inhibit Wnt-induced accumulation of ⁇ -catenin by targeting a pathway component that functions downstream of LRP6 and Dvl2 (see FIG. 3).
- the ability of IWR-I to block ⁇ -catenin accumulation in mouse L-cells treated with siRNAs targeting the APC tumor suppressor was tested (FIG. 5A).
- the effectiveness of IWR-I in this context prompted testing of the IWR compounds in colorectal cancer (CRC) cells that frequently harbor loss-of- function mutations in APC (Sjoblom et ah, 2006). Indeed, the IWR compounds were able to abrogate to varying degrees aberrant Wnt pathway activity exhibited in DLD- 1 cells, a CRC line that expresses a truncated form of APC (FIG. 5B).
- the ⁇ -catenin destruction complex which consists of APC, Axin, CKl, and GSK3 ⁇ , promotes proteasome-mediated proteolysis of phosphorylated ⁇ -catenin (Huang and He, 2008).
- a concomitant decrease in ⁇ -catenin levels was not observed, as would be expected based on the reporter assay results (see FIG. 5B) and the understanding of Axin2 function.
- IWR compounds Interaction of Axin2 with a biotinylated IWR compound in vitro suggests that IWR compounds interact either directly target Axin2 or an Axin2 associated protein (FIG. 5 F 5 G). Without being bound by theory regarding how IWR compounds alter Axin2 protein turnover, the inventors postulate that their effectiveness for inhibiting Wnt/ ⁇ -catenin pathway response can in part be explained by the rate-limiting role that Axin2 occupies in pathway response (Lee et al., 2003). Taken together, the IWR compounds have revealed a chemically tractable regulatory mechanism within this pathway that could be exploited to control levels of Wnt/ ⁇ - catenin pathway response (FIG. 5H).
- Sections were stained with Hematoxylin and Eosin or processed for BrdU immunohistochemistry as described (Shepard et ah, 2005). Sections from 8 animals each from IWR and DMSO groups were independently scored by four blinded observers. The total number of intestinal folds and total number of BrdU-positive nuclei in the mid and distal sections of the intestine were counted from each section. For caudal fin regeneration assays, zebrafish, 3-6 months of age, were anaesthetized in 0.2% Tricaine and half of the fin was resected using a razor blade to remove.
- Wnt pathway activity sustained either by genetic changes that result in altered Wnt ligand activity or the function of pathway regulators, has been associated with a broad range of cancers (Clevers, 2006; Polakis, 2007).
- Many colorectal- and some lung cancer-derived cells harbor molecular changes that result in aberrant activation of Wnt-mediated cellular responses.
- the DLD-I cells like many other CRC cells, harbor a mutation in APC whereas the selected lung cancer cell lines (A549, H 1299, H460 cells) have been shown to aberrantly express excessive levels of Pore that contributes to their tumorigenic behavior (Chen et ah, 2008; Polakis, 2007).
- aberrant pathway activity can be influenced by inhibition of normal Wnt protein function (Clevers, 2006).
- Cancer cell growth studies hi experiments involving IWR and IWP treatments, cancer cells were seeded into a 24 well format (2.5K cells/well) in the presence of the noted Wnt pathway inhibitors (0.5% DMSO final). Media and compound changed every 24 hours for 5 days. On day 6, ATP levels were quantitated via cell titer glo assay (Promega).
- siRNA transfections cells were transfected with 50 nM control, Ctnnbl, or Porcn siRNAs (SMARTPools, Dharmacon) using Effectene transfection reagent (Qiagen) and seeded into a 96 well plate, 7.5K cells/well, in triplicate. After 48 hrs, 2.5K cells were transferred from each well into a six well format. ATP levels were measured 120 hrs later via Cell Titer GIo assay (Promega).
- Van der Flier et al Gastroenterology, 132:628-632, 2007. Veeman et al , Developmental Cell, 5:367, 2003.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Physical Education & Sports Medicine (AREA)
- Diabetes (AREA)
- Neurosurgery (AREA)
- Rheumatology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Endocrinology (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Obesity (AREA)
- Emergency Medicine (AREA)
- Hematology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Indole Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/994,661 US8445491B2 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
| EP09767313.1A EP2303862B1 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
| JP2011511787A JP2011521958A (en) | 2008-05-27 | 2009-05-27 | Wnt protein signaling inhibitor |
| CA2726164A CA2726164A1 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
| AU2009260503A AU2009260503B2 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
| CN2009801294300A CN102105464A (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
| BRPI0913119-1A BRPI0913119A2 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signaling inhibitors |
| IL209556A IL209556A0 (en) | 2008-05-27 | 2010-11-24 | Wnt PROTEIN SIGNALLING INHIBITORS |
| US13/875,170 US9045416B2 (en) | 2008-05-27 | 2013-05-01 | WNT protein signalling inhibitors |
| US14/728,105 US20150374696A1 (en) | 2008-05-27 | 2015-06-02 | Wnt protein signalling inhibitors |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13014908P | 2008-05-27 | 2008-05-27 | |
| US61/130,149 | 2008-05-27 | ||
| US20427909P | 2009-01-02 | 2009-01-02 | |
| US61/204,279 | 2009-01-02 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/994,661 A-371-Of-International US8445491B2 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
| US13/875,170 Division US9045416B2 (en) | 2008-05-27 | 2013-05-01 | WNT protein signalling inhibitors |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2009155001A2 true WO2009155001A2 (en) | 2009-12-23 |
| WO2009155001A3 WO2009155001A3 (en) | 2010-02-25 |
Family
ID=41050590
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2009/045340 WO2009155001A2 (en) | 2008-05-27 | 2009-05-27 | Wnt protein signalling inhibitors |
Country Status (10)
| Country | Link |
|---|---|
| US (3) | US8445491B2 (en) |
| EP (1) | EP2303862B1 (en) |
| JP (1) | JP2011521958A (en) |
| KR (1) | KR20110014229A (en) |
| CN (1) | CN102105464A (en) |
| AU (1) | AU2009260503B2 (en) |
| BR (1) | BRPI0913119A2 (en) |
| CA (1) | CA2726164A1 (en) |
| IL (1) | IL209556A0 (en) |
| WO (1) | WO2009155001A2 (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011159726A2 (en) | 2010-06-14 | 2011-12-22 | The Scripps Research Institute | Reprogramming of cells to a new fate |
| CN102558173A (en) * | 2010-12-31 | 2012-07-11 | 广州源生医药科技有限公司 | Compound inhibiting conduction of WNT signal, and composition and application thereof |
| US8273900B2 (en) | 2008-08-07 | 2012-09-25 | Novartis Ag | Organic compounds |
| US20130177535A1 (en) * | 2011-07-02 | 2013-07-11 | John Cashman | Wnt inhibitors for human stem cell differentiation |
| JP2013533879A (en) * | 2010-06-29 | 2013-08-29 | アイアールエム・リミテッド・ライアビリティ・カンパニー | Compositions and methods for WNT signaling pathway modulation |
| WO2013064913A3 (en) * | 2011-10-19 | 2013-09-12 | Hiroyuki Kouji | Method for treating type i and type ii diabetes |
| WO2013184806A1 (en) * | 2012-06-05 | 2013-12-12 | University Of Kansas | Inhibitors of respiratory syncytial virus |
| US20140323503A1 (en) * | 2011-11-30 | 2014-10-30 | Trustees Of Boston College | Inhibitors of phospodiesterases 11 (pde11) and methods of use to elevate cortisol production |
| CN104530042A (en) * | 2010-12-31 | 2015-04-22 | 广州源生医药科技有限公司 | Compound inhibiting WNT signal conduction, composition, and application of two |
| US9783550B2 (en) | 2013-05-14 | 2017-10-10 | The Board Of Regents Of The University Of Texas System | Highly potent inhibitors of porcupine |
| EP3498824A1 (en) | 2013-04-26 | 2019-06-19 | Memorial Sloan-Kettering Cancer Center | Cortical interneurons and other neuronal cells produced by the directed differentiation of pluripotent and multipotent cells |
| US10815250B2 (en) | 2018-02-06 | 2020-10-27 | Ideaya Biosciences, Inc. | AhR modulators |
| WO2021173458A1 (en) * | 2020-02-28 | 2021-09-02 | Purdue Research Foundation | Generating aorta-gonad-mesonephros-like hematopoietic cells from human pluripotent stem cells under a defined condition |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IN2014CN02646A (en) | 2011-09-14 | 2015-08-07 | Samumed Llc | |
| BR112014018341A8 (en) * | 2012-01-28 | 2017-07-11 | Merck Patent Gmbh | AZA-HETEROCYCLIC COMPOUNDS |
| US9340549B2 (en) | 2012-03-05 | 2016-05-17 | Amgen Inc. | Oxazolidinone compounds and derivatives thereof |
| PH12017500997A1 (en) * | 2012-04-04 | 2018-02-19 | Samumed Llc | Indazole inhibitors of the wnt signal pathway and therapeutic uses thereof |
| WO2013169631A2 (en) * | 2012-05-11 | 2013-11-14 | The Board Of Regents Of The University Of Texas System | Wnt protein signalling inhibitors |
| WO2013177349A2 (en) * | 2012-05-25 | 2013-11-28 | Glaxosmithkline Llc | Quinazolinediones as tankyrase inhibitors |
| KR101418168B1 (en) * | 2012-11-21 | 2014-07-09 | 부경대학교 산학협력단 | Novel tetrahydropyridinol derivative compound and pharmaceutical compositions for prevention or treatment of cancer comprising the same |
| CN103113353B (en) * | 2013-03-13 | 2014-09-10 | 中国科学院昆明植物研究所 | Triazole compounds, medical composition thereof and preparation and application of triazole compounds |
| WO2015016195A1 (en) * | 2013-07-29 | 2015-02-05 | 協和発酵キリン株式会社 | Wnt SIGNALING INHIBITOR |
| US9512406B2 (en) | 2013-12-20 | 2016-12-06 | The J. David Gladstone Institute, a testamentary trust established under the Will of J. David Gladstone | Generating hepatocytes |
| US9914774B2 (en) | 2014-07-11 | 2018-03-13 | Genentech, Inc. | Notch pathway inhibition |
| MX2017004639A (en) | 2014-10-08 | 2017-07-17 | Redx Pharma Plc | N-pyridinyl acetamide derivatives as wnt signalling pathway inhibitors. |
| US10420758B2 (en) | 2015-05-02 | 2019-09-24 | The Board Of Regents Of The University Of Texas System | Indolinyl-sulfonamide inhibitors of tankyrase and methods of use thereof |
| US10624949B1 (en) | 2015-07-27 | 2020-04-21 | National Technology & Engineering Solutions Of Sandia, Llc | Methods for treating diseases related to the wnt pathway |
| WO2017040579A1 (en) * | 2015-08-31 | 2017-03-09 | The Trustees Of Columbia University In The City Of New York | Methods for cardiac differentiation of human induced pluripotent stem cells |
| EP3370721A4 (en) | 2015-11-06 | 2019-05-22 | Samumed, LLC | Treatment of osteoarthritis |
| CN109867661B (en) * | 2017-12-05 | 2022-07-19 | 中国医学科学院药物研究所 | Amide compounds for regulating WNT signal pathway and application thereof |
| WO2019124603A1 (en) * | 2017-12-22 | 2019-06-27 | 경상대학교병원 | Composition for preventing or treating keloid, containing iwr-1 as active ingredient |
| US20240075089A1 (en) * | 2020-12-30 | 2024-03-07 | Arbor Life Holdings, Ltd. | Botanicals as wnt/beta-catenin activators, molecular pathway regulators, tissue regenerators and health biomarker regulators |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2002217970A1 (en) | 2000-11-28 | 2002-06-11 | Curis, Inc. | Wnt signalling assay, methods and uses thereof |
| PA8539301A1 (en) | 2001-02-14 | 2002-09-30 | Warner Lambert Co | INHIBITORS OF THE METALOPROTEINASE OF THE MATRIX |
| PA8539401A1 (en) | 2001-02-14 | 2002-10-28 | Warner Lambert Co | QUINAZOLINAS AS INHIBITORS OF MMP-13 |
| US20040087548A1 (en) | 2001-02-27 | 2004-05-06 | Salvati Mark E. | Fused cyclic succinimide compounds and analogs thereof, modulators of nuclear hormone receptor function |
| US6897220B2 (en) * | 2001-09-14 | 2005-05-24 | Methylgene, Inc. | Inhibitors of histone deacetylase |
| WO2003024448A2 (en) | 2001-09-14 | 2003-03-27 | Methylgene, Inc. | Inhibitors of histone deacetylase |
-
2009
- 2009-05-27 CN CN2009801294300A patent/CN102105464A/en active Pending
- 2009-05-27 US US12/994,661 patent/US8445491B2/en active Active
- 2009-05-27 AU AU2009260503A patent/AU2009260503B2/en not_active Ceased
- 2009-05-27 KR KR1020107029281A patent/KR20110014229A/en not_active Application Discontinuation
- 2009-05-27 WO PCT/US2009/045340 patent/WO2009155001A2/en active Application Filing
- 2009-05-27 JP JP2011511787A patent/JP2011521958A/en not_active Withdrawn
- 2009-05-27 EP EP09767313.1A patent/EP2303862B1/en not_active Not-in-force
- 2009-05-27 CA CA2726164A patent/CA2726164A1/en not_active Abandoned
- 2009-05-27 BR BRPI0913119-1A patent/BRPI0913119A2/en not_active IP Right Cessation
-
2010
- 2010-11-24 IL IL209556A patent/IL209556A0/en unknown
-
2013
- 2013-05-01 US US13/875,170 patent/US9045416B2/en active Active
-
2015
- 2015-06-02 US US14/728,105 patent/US20150374696A1/en not_active Abandoned
Non-Patent Citations (2)
| Title |
|---|
| BARKER NICK; CLEVERS HANS: "Mining the Wnt pathway for cancer therapeutics" NATURE REVIEWS. DRUG DISCOVERY, vol. 5, no. 12, December 2006 (2006-12), pages 997-1014, XP002545380 * |
| VEEMAN MICHAEL T; AXELROD JEFFREY D; MOON RANDALL T: "A second canon: Functions and mechanisms of beta-catenin-independent Wnt signaling" DEVELOPMENTAL CELL, vol. 5, no. 3, September 2003 (2003-09), pages 367-377, XP002545381 * |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8273900B2 (en) | 2008-08-07 | 2012-09-25 | Novartis Ag | Organic compounds |
| US8614213B2 (en) | 2008-08-07 | 2013-12-24 | Novartis Ag | Cyclohexyl amide derivatives and their use as CRF-1 receptor antagonists |
| WO2011159726A2 (en) | 2010-06-14 | 2011-12-22 | The Scripps Research Institute | Reprogramming of cells to a new fate |
| JP2013533879A (en) * | 2010-06-29 | 2013-08-29 | アイアールエム・リミテッド・ライアビリティ・カンパニー | Compositions and methods for WNT signaling pathway modulation |
| CN104530042A (en) * | 2010-12-31 | 2015-04-22 | 广州源生医药科技有限公司 | Compound inhibiting WNT signal conduction, composition, and application of two |
| CN102558173A (en) * | 2010-12-31 | 2012-07-11 | 广州源生医药科技有限公司 | Compound inhibiting conduction of WNT signal, and composition and application thereof |
| US20130177535A1 (en) * | 2011-07-02 | 2013-07-11 | John Cashman | Wnt inhibitors for human stem cell differentiation |
| US9023340B2 (en) | 2011-07-02 | 2015-05-05 | Chemregen, Inc. | Wnt inhibitors for human stem cell differentiation |
| WO2013064913A3 (en) * | 2011-10-19 | 2013-09-12 | Hiroyuki Kouji | Method for treating type i and type ii diabetes |
| US20140323503A1 (en) * | 2011-11-30 | 2014-10-30 | Trustees Of Boston College | Inhibitors of phospodiesterases 11 (pde11) and methods of use to elevate cortisol production |
| US9173884B2 (en) * | 2011-11-30 | 2015-11-03 | Trustees Of Boston College | Inhibitors of phosphodiesterase 11 (PDE11) |
| WO2013184806A1 (en) * | 2012-06-05 | 2013-12-12 | University Of Kansas | Inhibitors of respiratory syncytial virus |
| US9499496B2 (en) | 2012-06-05 | 2016-11-22 | University Of Kansas | Inhibitors of respiratory syncytial virus |
| EP3498824A1 (en) | 2013-04-26 | 2019-06-19 | Memorial Sloan-Kettering Cancer Center | Cortical interneurons and other neuronal cells produced by the directed differentiation of pluripotent and multipotent cells |
| US9783550B2 (en) | 2013-05-14 | 2017-10-10 | The Board Of Regents Of The University Of Texas System | Highly potent inhibitors of porcupine |
| US10815250B2 (en) | 2018-02-06 | 2020-10-27 | Ideaya Biosciences, Inc. | AhR modulators |
| US11407764B2 (en) | 2018-02-06 | 2022-08-09 | Ideaya Biosciences, Inc. | AhR modulators |
| WO2021173458A1 (en) * | 2020-02-28 | 2021-09-02 | Purdue Research Foundation | Generating aorta-gonad-mesonephros-like hematopoietic cells from human pluripotent stem cells under a defined condition |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2009260503B2 (en) | 2014-03-27 |
| CA2726164A1 (en) | 2009-12-23 |
| US20150374696A1 (en) | 2015-12-31 |
| US9045416B2 (en) | 2015-06-02 |
| WO2009155001A3 (en) | 2010-02-25 |
| EP2303862B1 (en) | 2016-08-24 |
| EP2303862A2 (en) | 2011-04-06 |
| US20140038922A1 (en) | 2014-02-06 |
| JP2011521958A (en) | 2011-07-28 |
| AU2009260503A1 (en) | 2009-12-23 |
| US20110136813A1 (en) | 2011-06-09 |
| US8445491B2 (en) | 2013-05-21 |
| IL209556A0 (en) | 2011-01-31 |
| CN102105464A (en) | 2011-06-22 |
| KR20110014229A (en) | 2011-02-10 |
| BRPI0913119A2 (en) | 2015-07-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2303862B1 (en) | Wnt protein signalling inhibitors | |
| TWI751585B (en) | Glucagon-like peptide 1 receptor agonists | |
| TWI582073B (en) | Sphingosine-1-phosphate receptor agonists, methods of preparing the same, and pharmaceutical compositions containing the same as an active agent | |
| EP1841426B1 (en) | Tertiary carbinamines having substituted heterocycles, which are active as inhibitors of beta-secretase, for the treatment of alzheimer's disease | |
| EP1855679B1 (en) | Aminomethyl beta-secretase inhibitors for the treatment of alzheimer's disease | |
| WO2013169631A2 (en) | Wnt protein signalling inhibitors | |
| TWI557110B (en) | Arylsulfonyl pyrazoline carboxamidine derivatives as 5-ht6 antagonists | |
| KR20200115448A (en) | Selective inhibitor of NLRP3 inplasmasome | |
| TW202112761A (en) | SHP2 Phosphatase Allosteric Inhibitor | |
| CN102791708B (en) | The inhibitor of AKT activity | |
| CN113993845A (en) | N- (phenyl) -indole-3-sulfonamide derivatives and related compounds as GPR17 modulators for the treatment of CNS disorders, such as multiple sclerosis | |
| TW201102375A (en) | Hedgehog pathway antagonists and therapeutic applications thereof | |
| WO2022135610A1 (en) | Tetracyclic compound, pharmaceutical composition thereof and use thereof | |
| FR2920023A1 (en) | New indol-2-one derivatives are growth hormone secretagogue receptor-1a antagonists, useful for the preparation of a medicament to treat or prevent obesity, diabetes and appetite disorders | |
| KR20230104151A (en) | Acetamido-phenyltetrazole derivatives and methods of use thereof | |
| WO2023031440A1 (en) | 2-amino-5,5-dimethylhexanoic acid derivatives as sortilin modulators for use in the treatment of disease of the central nervous system | |
| WO2020063968A1 (en) | Ar and bet dual inhibitor and use thereof | |
| CN111683945B (en) | Crystalline forms of CXCR7 receptor antagonists | |
| CA3133870A1 (en) | Quinoline derivatives, processes for their preparation and uses thereof for the treatment of cancer | |
| CA2478942A1 (en) | An optically active pyridine derivative and a medicament containing the same | |
| CN114075141A (en) | Opioid receptor 'bias' ligand, preparation method and application thereof in medicine | |
| WO2023147063A2 (en) | Compounds and methods for yap/tead modulation and indications therefor | |
| AU2022272302A1 (en) | Compounds and methods for yap/tead modulation and indications therefor | |
| WO2022240966A1 (en) | Compounds and methods for yap/tead modulation and indications therefor | |
| EP3766544A1 (en) | Novel pyrazolone derivatives as pd-1/pd-l1 interaction inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980129430.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09767313 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009260503 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011511787 Country of ref document: JP Ref document number: 2726164 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2009260503 Country of ref document: AU Date of ref document: 20090527 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 9223/DELNP/2010 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 20107029281 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2009767313 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009767313 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12994661 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: PI0913119 Country of ref document: BR Kind code of ref document: A2 Effective date: 20101129 |