WO2009106982A1 - Indazole derivatives - Google Patents
Indazole derivatives Download PDFInfo
- Publication number
- WO2009106982A1 WO2009106982A1 PCT/IB2009/000432 IB2009000432W WO2009106982A1 WO 2009106982 A1 WO2009106982 A1 WO 2009106982A1 IB 2009000432 W IB2009000432 W IB 2009000432W WO 2009106982 A1 WO2009106982 A1 WO 2009106982A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- carboxamide
- indazole
- dimethylpropyl
- carbonyl
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*C(C(C)C)C(N)=O Chemical compound C*C(C(C)C)C(N)=O 0.000 description 4
- ZGGBHZGLQCFMOU-UHFFFAOYSA-N CC(C(C)(C)C)C(N)=O Chemical compound CC(C(C)(C)C)C(N)=O ZGGBHZGLQCFMOU-UHFFFAOYSA-N 0.000 description 1
- IVWIZTZOWWOLDN-UHFFFAOYSA-N CC(C(C)(C)C)C(NCC(CO)O)=O Chemical compound CC(C(C)(C)C)C(NCC(CO)O)=O IVWIZTZOWWOLDN-UHFFFAOYSA-N 0.000 description 1
- RGTDKECCYWTWGW-UHFFFAOYSA-N CC(C(C)(C)C)C(NCCCO)=O Chemical compound CC(C(C)(C)C)C(NCCCO)=O RGTDKECCYWTWGW-UHFFFAOYSA-N 0.000 description 1
- BWIPDEJTFOCOEM-HSZRJFAPSA-N CC(C)(C)[C@@H](C(N)=O)NC(c(c1c2)n[n](Cc3ccccc3)c1ccc2-c1cnccc1)=O Chemical compound CC(C)(C)[C@@H](C(N)=O)NC(c(c1c2)n[n](Cc3ccccc3)c1ccc2-c1cnccc1)=O BWIPDEJTFOCOEM-HSZRJFAPSA-N 0.000 description 1
- KVLGUDDIVYCQNE-MRXNPFEDSA-N CC(C)(C)[C@@H](C(NCC(OCc1ccccc1)=O)=O)NC(OC(C)(C)C)=O Chemical compound CC(C)(C)[C@@H](C(NCC(OCc1ccccc1)=O)=O)NC(OC(C)(C)C)=O KVLGUDDIVYCQNE-MRXNPFEDSA-N 0.000 description 1
- KRAOMZCZIGDYLW-HXUWFJFHSA-N CC(C)(C)[C@@H](C(NCc1nnc(C(N)=O)[o]1)=O)NC(c1n[n](Cc(cc2)ccc2F)c2ccccc12)=O Chemical compound CC(C)(C)[C@@H](C(NCc1nnc(C(N)=O)[o]1)=O)NC(c1n[n](Cc(cc2)ccc2F)c2ccccc12)=O KRAOMZCZIGDYLW-HXUWFJFHSA-N 0.000 description 1
- RMWQEORAAZDKHH-SCSAIBSYSA-N CC(C)(C)[C@@H](c1n[nH]nn1)N Chemical compound CC(C)(C)[C@@H](c1n[nH]nn1)N RMWQEORAAZDKHH-SCSAIBSYSA-N 0.000 description 1
- SJWKFDVGPCYFDP-SCSAIBSYSA-N CC(C)(C)[C@@H](c1nnc(NC(N)=O)[o]1)N Chemical compound CC(C)(C)[C@@H](c1nnc(NC(N)=O)[o]1)N SJWKFDVGPCYFDP-SCSAIBSYSA-N 0.000 description 1
- FROCGBFQIWUEGD-UHFFFAOYSA-N CC(CC1CCCCC1)CO Chemical compound CC(CC1CCCCC1)CO FROCGBFQIWUEGD-UHFFFAOYSA-N 0.000 description 1
- RNDNSYIPLPAXAZ-UHFFFAOYSA-N CC(CO)c1ccccc1 Chemical compound CC(CO)c1ccccc1 RNDNSYIPLPAXAZ-UHFFFAOYSA-N 0.000 description 1
- SVHNHZQUPDPPIY-UHFFFAOYSA-N CCCC(C(C)(C)C)C(NC(CO)CO)=O Chemical compound CCCC(C(C)(C)C)C(NC(CO)CO)=O SVHNHZQUPDPPIY-UHFFFAOYSA-N 0.000 description 1
- CRXLGUCUIORPDM-UHFFFAOYSA-N CCc(cc1)ccc1P Chemical compound CCc(cc1)ccc1P CRXLGUCUIORPDM-UHFFFAOYSA-N 0.000 description 1
- XDCCUETYAWEEOS-UHFFFAOYSA-N CCc1c(C)[o]c(C)c1 Chemical compound CCc1c(C)[o]c(C)c1 XDCCUETYAWEEOS-UHFFFAOYSA-N 0.000 description 1
- ZQJXTODJWIFLFX-UHFFFAOYSA-N COC(c(c1ccc2)n[n](Cc(cc3)ccc3C#N)c1c2F)=O Chemical compound COC(c(c1ccc2)n[n](Cc(cc3)ccc3C#N)c1c2F)=O ZQJXTODJWIFLFX-UHFFFAOYSA-N 0.000 description 1
- ILIKHLPMOCBIRX-UHFFFAOYSA-N COC(c1n[nH]c2c1cccc2F)=O Chemical compound COC(c1n[nH]c2c1cccc2F)=O ILIKHLPMOCBIRX-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/54—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings condensed with carbocyclic rings or ring systems
- C07D231/56—Benzopyrazoles; Hydrogenated benzopyrazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/08—Bridged systems
Definitions
- the present invention provides pharmaceutically active indazole compounds and analogues. Such compounds have cannabinoid (CB)1 receptor binding activity.
- CB cannabinoid
- the present invention also relates to pharmaceutical compositions, methods of treatment and use, comprising the above derivatives for the treatment of disease conditions mediated by CB 1 receptor binding activity.
- Cannabinoid receptors endogenous cannabinoids and the enzymes that synthesize and degrade endocannabinoids make up the endocannabinoid system.
- CB1 and CB2 are two subtypes of cannabinoid receptors.
- CB1 and CB2 are both G protein coupled receptors.
- CB 1 receptors primarily exist in the central nervous system, but are also found in some peripheral tissues including pituitary gland, immune cells, reproductive tissues, gastrointestinal tissues, sympathetic ganglia, heart, lung, urinary bladder and adrenal gland.
- CB2 receptors primarily exist in immune cells.
- Cannabinoid agonists are believed to be useful in the treatment of pain and several other indications.
- the present invention is directed to pharmaceutically active indazole compounds. Such compounds are useful for as CB1 agonists.
- This invention is directed, in part, to compounds that generally fall within the structure of Formula I:
- R 11 and R 12 are independently H, OH, d-C 6 alkyl, Ci-C 6 haloalkyl, OH-CrC 6 alkyl, (OH) 2 -C 1 -C 6 alkyl, (OH) 3 -C 4 -C 6 alkyl, C r C 6 alkoxy-(CH 2 ) n -, C 3 -C 7 cycloalkyl, benzo-fused C 3 -C 7 cycloalkyl, cyano-Ci-C 6 alkyl, NH 2 -C(NH)-CrC 6 alkyl, (OH-CrC 6 alkyl)rC r C ⁇ alkylene, OH-C 3 -C 7 cycloalkyl-(CH 2 )n-, OH-(CH 2 ) n -C 3 -C 7 cycloalkyl-, OH-C 3 -C 7 cycloalkyl-, CrC 6 alkoxy-C(O
- R 13 is H, CrC 6 alkyl, OH-CrC 6 alkyl, aryl, aryl-(CH 2 ) n -, or C 3 -C 7 cycloalkyl;
- R 14 is (CrC 6 alkyl) 2 N-, aryl, CrC 6 alkyl, or C 3 -C 7 cycoalkyl;
- R 15 R 21 R 29 R3 I R34 an(J R 4 o are j ndep endently H Or C 1 -C 6 alkyl;
- R 16 is OH or CrC 6 alkoxy
- R 17 and R 18 are independently H, CrC 6 alkyl, C 3 -C 7 cycoalkyl,
- each R 19 is independently H or C 1 -C 6 alkyl
- R 20 is C 1 -C 6 alkyl, C 1 -C 6 haloalkyl, or (C 1 -C 6 alkyl) 2 N-
- R 22 and R 23 are independently C 1 -C 6 alkyl, C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH-C 1 -C 6 alkyl, aryl, or aryl-OH-CrC 6 alkylene
- each R 24 is independently H, C 1 -C 6 alkyl, C 3 -C 7 cycloalkyl, CrC 6 haloalkyl, oxo, OH, NH 2 , C 1 -C 6 alkoxy-C(O)-, NH 2 -C(O)-(CH
- R 27 and R 28 independently are H, NH 2 -C(O)-, C 3 -C 7 cycloalkyl-C(O)-, or R 24 i -5 -heteroaryl-;
- R 30 is C 1 -C 6 alkyl, C 3 -C 7 cycloalkyl, NH 2 , C 1 -C 6 alkyl-NH-, C 3 -C 7 cycloalkyl-(CH 2 )n-NH-, morpholin-4-yl, or R 38 1-5 -phenyl;
- R 42 is H, C 1 -C 6 alkyl, OH-C 1 -C 6 alkyl, aryl, aryl-(CH 2 ) n - or NH 2 -C(O)-CH 2 ;
- R 43 is OH-C(O)-, C 1 -C 6 alkoxy-C(O)-, NH 2 -C(O)- or R 44 R 45 NCH 2 -; and R 44 and R 45 are independently Ci-C 6 alkyl or OH-CrC 6 alkyl, or R 44 and R 45 together with the nitrogen atom to which they are attached form a pyrrolidine, piperidine or morpholine ring; n is an integer from 1 to 6; and each R 3 is independently H 1 halo, C r C 6 alkyl, aryl, NH 2 -C(O)-, C 1 -C 6 alkoxy or heteroaryl.
- This invention also includes pharmaceutically acceptable salts, solvates and hydrates. This invention also includes all tautomers and stereochemical isomers of these compounds.
- This invention also is directed, in part, to a method for treating a CB1 mediated disorder in a mammal.
- CB1 mediated disorders include pain, rheumatoid arthritis and osteoarthritis.
- the method comprises administering an above- described compound or pharmaceutically acceptable salt thereof, to the mammal in an amount that is therapeutically-effective to treat the condition.
- the present invention is directed to a class of indazole compounds.
- the present invention is directed to indazole compounds useful as CB1 agonists. While the present invention is not so limited, an appreciation of various aspects of the invention will be gained through the following discussion and the examples provided below.
- alkane refers to a saturated acyclic hydrocarbon which can be either a straight chain or branched chain.
- alkyl refers to a straight or branched chain univalent radical derived from an alkane by removal of one hydrogen.
- alkyl radicals are methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, t-butyl, pentyl, neopentyl, hexyl, isohexyl, and the like.
- alkylene refers to a straight chain or branched bivalent radical derived from alkane by the removal of H from each of the two terminal carbons.
- Examples include methylene: i CH2 ⁇ ! , ethylene: « c ⁇ c « , propylene:
- alkoxy means alkyl-O-, wherein alkyl is as defined above. Examples of such a substituent include methoxy (CH 3 -O-), ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, sec-butoxy, and tert-butoxy.
- cycloalkyl means a saturated carbocyclyl substituent containing from 3 to about 20 carbon atoms.
- a cycloalkyl may be a single cyclic ring or multiple condensed rings.
- Such cycloalkyl groups include, by way of example, single ring structures such as cyclopropyl, cyclobutyl, cyclopentyl, cyclooctyl, and the like, or multiple ring structures such as adamantanyl, and the like.
- aryl means an aromatic carbocyclyl containing from 6 to 14 carbon ring atoms.
- aryl embraces both single and multiple rings. Examples of aryls include phenyl, naphthalenyl, and indenyl.
- arylalkyl means alkyl substituted with aryl, wherein alkyl and aryl are as defined above.
- hydroxy or “hydroxyl” means OH-.
- hydroxyalkyl means alkyl substituted with one more hydroxyl, wherein hydroxyl and alkyl are as defined above.
- halo or halogen refers to bromo, chloro, fluoro or iodo.
- oxy means an ether substituent, and may be depicted as -O-.
- sulfonyl means SO 2 -.
- alkylthio is an alkyl substituted thio, which is also depicted as:
- heterocyclyl means a saturated or partially saturated ring structure containing a total of 3 to 14 ring atoms. At least one of the ring atoms is a heteroatom (i.e., oxygen, nitrogen, or sulfur), with the remaining ring atoms being independently selected from the group consisting of carbon, oxygen, nitrogen, and sulfur.
- a heterocyclyl may be a single ring, which typically contains from 3 to 7 ring atoms, more typically from 3 to 6 ring atoms, and even more typically 5 to 6 ring atoms.
- heterocyclyls include piperidinyl, morpholinyl, thiomorpholinyl, tetrahydrofuranyl, tetrahydropyranyl, pyrrolidinyl, piperazinyl and diazepanyl.
- heteroaryl means an aromatic heterocyclyl containing from 5 to 14 ring atoms.
- a heteroaryl may be a single ring or 2 or 3 fused rings.
- heteroaryl substituents include isoxazolyl, pyridinyl, furyl, oxadiazolyl, tetrazolyl, dihydroimidazolyl, thiadiazolyl, oxazolyl, triazolyl and dihydroisoxazolyl.
- R 5 i.5-heteroaryl-(CH 2 )n-; wherein each R 4 is independently H, halo, cyano, NH 2 -C(O)-, Ci-C ⁇ alkoxy-, trifluoromethyl or Ci-C ⁇ alkoxy-C(O)-; each R 5 is independently H or CrC 6 alkyl; R 2 is
- R 24 i. 5 -heteroaryl-NR 15 -C(O)-R 13 CH-,
- R 11 and R 12 are independently H, OH, Ci-C 6 alkyl, C r C 6 haloalkyl, OH-CrC 6 alkyl, (OH) 2 -CrC 6 alkyl, (OH) 3 -C 4 -C 6 alkyl, C 1 -C 6 alkoxy-(CH 2 ) n -, C 3 -C7 cycloalkyl, benzo-fused C 3 -C 7 cycloalkyl, cyano-CrC 6 alkyl, NH 2 -C(NH)-CrC 6 alkyl, (OH-CrC 6 alkyl) 2 -CrC 6 alkylene, OH-C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH-(CHz) n -C 3 -C 7 cycloalkyl-, OH-C 3 -C 7 cycloalkyl-, CrC 6 alkoxy-C(O
- R 14 is (CrC 6 alkyl) 2 N-, aryl, CrC 6 alkyl, or C 3 -C 7 cycoalkyl;
- R 15 , R 21 , R 29 , R 31 , R 34 , and R 40 are independently H or C r C 6 alkyl;
- R 16 is OH or CrC 6 alkoxy;
- R 17 and R 18 are independently H, CrC 6 alkyl, C 3 -C 7 cycoalkyl, OH-C 1 -C 6 alkyl, (OH) 2 -C 1 -C 6 alkyl, or R 24 1-5 -heteroaryl-; each R 19 is independently H or C 1 -C 6 alkyl; R 20 is C 1 -C 6 alkyl, C 1 -C 6 haloalkyl, or (C 1 -C 6 alkyl) 2 N-; R 22 and R 23 are independently CrC 6 alkyl, C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH-Ci-C 6 alkyl, aryl, or aryl-OH-Ci-C 6 alkylene; each R 24 is independently H, C 1 -C 6 alkyl, C 3 -C 7 cycloalkyl, Ci-C 6 haloalkyl, oxo, OH, NH 2
- R 27 and R 28 independently are H, NH 2 -C(O)-, C 3 -C 7 cycloalkyl-C(O)-, or R 24 i -5 -heteroaryl-;
- R 30 is CrC 6 alkyl, C 3 -C 7 cycloalkyl, NH 2 , C r C 6 alkyl-NH-, C 3 -C 7 cycloalkyl-(CH 2 )n-NH-, morpholin-4-yl, or R 38 i -5 -phenyl;
- R 42 is H, CrC 6 alkyl, OH-C 1 -C 6 alkyl, aryl, aryl-(CH 2 ) n - or NH 2 -C(O)-CH 2 ;
- R 43 is OH-C(O)-, CrC 6 alkoxy-C(O)-, NH 2 -C(O)- or R 44 R 45 NCH 2 -;
- R 44 and R 45 are independently Ci-C 6 alkyl or OH-Ci-C 6 alkyl, or
- R 44 and R 45 together with the nitrogen atom to which they are attached form a pyrrolidine, piperidine or morpholine ring; n is an integer from 1 to 6; and each R 3 is independently H, halo, CrC 6 alkyl, aryl, NH 2 -C(O)-, d-C 6 alkoxy or heteroaryl.
- the present invention includes compounds or pharmaceutically acceptable salts thereof, having a structure according to Formula I:
- R 1 is R 4 i-5-aryl-(CH 2 ) n - or R 5 i -5 -heteroaryl-(CH 2 ) n -; wherein each R 4 is independently H, halo, cyano or NH 2 -C(O)-; each R 5 is independently H or Ci-C 6 alkyl;
- R 2 is NR 11 R 12 -C(O)-R 13 CH-, R 14 -C(O)-NR 15 -(CH 2 ) n -R 13 CH-, R 16 -C(O)-R 13 CH-, CrC 6 alkoxy-C(O)-(CH 2 )n-NR 15 -C(O)-R 13 CH-, NR 17 R 18 -C(O)-(CH 2 ) n -NR 19 -C(O)-R 13 CH- , R 20 -SO 2 -NR 21 -(CH 2 )n-R 13 CH-, R 22 R 23 CH-, R 24 i -5 -heteroaryl,
- R 11 and R 12 are independently H, C r C 6 alkyl, OH-CrC 6 alkyl, (OH) 2 -C 1 - C 6 alkyl, CrC 6 alkoxy-(CH 2 ) n -, C 3 -C 7 cycloalkyl, cyano-C r C 6 alkyl, (OH-CrC 6 alkyl) 2 -C r C 6 alkylene, OH-C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH-(CH 2 ) n -C 3 -C 7 cycloalkyl- , or OH-aryl;
- R 13 is H, CrC 6 alkyl, OH-CrC 6 alkyl, aryl, aryl-(CH 2 ) n -, or C 3 -C 7 cycloalkyl;
- R 14 is (CrC 6 alkyl) 2 N-, aryl, CrC 6 alkyl, or C 3 -C 7 cycoalkyl;
- R 15 , R 21 , R 29 , R 31 , R 33 , R 34 , R 36 , R 39 and R 40 are independently H or C 1 -C 6 alkyl;
- R 16 is OH or CrC 6 alkoxy
- R 17 , R 18 and R 19 are independently H or CrC 6 alkyl
- R 20 is CrC 6 alkyl, C r C 6 haloalkyl, or (C r C 6 alkyl) 2 N-;
- R 22 and R 23 are independently CrC 6 alkyl, C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH- CrC 6 alkyl, aryl, or aryl-OH-CrC 6 alkylene; each R 24 is independently H, CrC 6 alkyl, C 3 -C 7 cycloalkyl, d-C 6 haloalkyl, oxo, NH 2 , CrC 6 alkoxy-C(O)-, NH 2 -C(O)-(CH 2 ) n -, NH 2 -C(O)-, NH 2 - C(O)-NH-, OH-C(O)-, NH 2 -C(O)-(CH 2 ) n -NH-C(O)-, (OH) 2 -CrC 6 alkyl-NH-C(O)-, or OH-CrC 6 alkyl-NH-C(O)-; each R 25 is
- R 27 and R 28 independently are H, NH 2 -C(O)-, or C 3 -C 7 cycloalkyl-C(O)-;
- R 30 is CrC 6 alkyl, C 3 -C 7 cycloalkyl or NH 2 ;
- R 32 is OH
- R 35 is independently H, CrC 6 alkyl, NH 2- C(O)-, CrC 6 alkoxy-C(O)- or C 3 - C 7 cycloalkyl; each R 37 is independently H, NH 2 C(O)- or OH; each R 38 is independently H, NH 2 SO 2 -, cyano, heteroaryl, OH, halo, C 1 - C 6 alkoxy, OH-C(O)-, or C 1 -C 6 alkoxy-C(O)-; each R 41 independently from H, C 1 -C 6 alkoxy or halo; n is an integer from 1 to 6; and each R 3 is independently H, halo, C 1 -C 6 alkyl, aryl, NH 2 -C(O)-, Ci-C 6 alkoxy or heteroaryl.
- X is CH or N
- R 1 is R 4 i -5 -benzyl, R 5 i -5 -isoxazolyl- CH 2 - or R 5 1-5 -pyridinyl- CH 2 -; wherein each R 4 is H, fluoro, cyano, NH 2 -C(O)-; each R 5 is independently H or CH 3 ;
- R 2 is NR 11 R 12 -C(O)-R 13 CH-, R 14 -C(O)-NR 15 - CH 2 -R 13 CH-, R 16 -C(O)-R 13 CH-, (CH 3 ) 3 C-O-C(O)-CH 2 -NR 15 -C(O)-R 13 CH-, NR 17 R 18 -C(O)-CH 2 -NR 19 -C(O)-R 13 CH-, NR 17 R 18 -C(O)- (CH 2 ) 2 -NR 19 -C(O)-R 13 CH-, R 20 -SO 2 -NR 21 -CH 2 -R 13 CH-, R 22 R 23 CH-, R 24 1-5 -dihydroimidazolyl, R 2 V 5 -isoxazolyl, R 24 i.
- R 11 and R 12 independently are H, CH 3 , (CH 3 ) 2 CH-, cyclobutyl, cyclopropyl, CH 3 O(CH 2 ) 2 -, OH-ethyl, OH-propyl, (OH) 2 -propyl, cyano-CH 2 -,
- R 13 is H, (CHa) 3 C-, (CHa) 2 CHCH 2 -, (CH 3 ) 2 CH-, OH-ethyl, benzyl, phenyl, or cyclohexyl;
- R 14 is (CH 3 CH 2 ) 2 N-, phenyl, (CH 3 ) 3 C-, or cyclopropyl;
- R 15 R 21 R2 9 R3 I R33 R S4 R36 R3 9 and R 40 are j nde pendently H or CH 3 ;
- R 16 is OH or CH 3 O
- R 17 , R 18 and R 19 are independently H or CH 3 ;
- R 20 is (CHa) 2 CH-, CH 3 , CF 3 , or (CH 3 ) 2 N-;
- R 22 and R 23 are independently (CH 3 ) 3 C-, (CH 3 ) 2 CH-, cyclohexyl- CH 2 -, OHCH 2 , phenyl, OH-isopropyl, OH-ethyl, or phenyl-OHCH-; each R 24 is independently H, CH 3 , CH 3 CH 2 -, (CH 3 ) 3 C-, cyclopropyl, CF 3 , oxo, NH 2 , CH 3 CH 2 -O-C(O)-, NH 2 -C(O)-CH 2 -, NH 2 -C(O)-, NH 2 -C(O)-NH-, OH- C(O)-, NH 2 -C(O)-CH 2 -NH-C(O)-, (OH ) 2 -propyl-N H-C(O)- or OH-ethyl-NH-C(O)-; each R 25 is independently H or o
- R 27 and R 28 are independently H, NH 2 -C(O)-, or cyclopropyl-C(O)-;
- R 30 is CH 3 , cyclopropyl or NH 2 ;
- X is CH or N; R 1 is each R 3 is independently H, CH 3 , chloro, bromo, fluoro, phenyl, NH 2 -C(O)-, CH 3 O-, 3-pyridinyl, 4-pyridinyl, or 2-oxazolyl.
- a compound of formula I or a pharmaceutically acceptable salt thereof wherein X is CH or N; R 1 is R 4 i-5-aryl-(CH 2 )n- or R 5 i. 5 -heteroaryl-(CH 2 )n-; wherein each R 4 is independently H, halo, cyano or NH 2 -C(O)-; each R 5 is independently H or Ci-C 6 alkyl;
- R 2 is NR 11 R 12 -C(O)-R 13 CH-, R 16 -C(O)-R 13 CH-, NR 17 R 18 -C(O)-(CH 2 ) n -NR 19 -C(O)- R 13 CH-, R 22 R 23 CH-, R 24 1-5 -heteroaryl-R 13 CH-, R 26 I-5 -C 3 -C 7 cycloalkyl, NR 27 R 28 -(CH 2 ) n - NR 29 -C(O)-R 13 CH-, R 30 -SO 2 -NR 31 -(CH 2 )n-NR 19 -C(O)-R 13 CH-, R 30 -SO 2 -(CH 2 )n-NR 31 - C(O)-R 13 CH-, R 32 -C(O)-R 33 CH-NR 34 -C(O)-R 13 CH-, R 35 1-5 -heteroaryl-(CH 2 )n-NR 36
- R 11 and R 12 are independently H, Ci-C 6 alkyl, OH-CrC 6 alkyl, (OH) 2 -C 1 - C 6 alkyl, C r C 6 alkoxy-(CH 2 ) n -, C 3 -C 7 cycloalkyl, (OH-Ci-C 6 alkyl) 2 -C r C 6 alkylene, OH-C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH -(CH 2 ) n -C 3 -C 7 cycloalkyl, OH-aryl,
- R 13 is H, CrC 6 alkyl, OH-C 1 -C 6 alkyl, aryl, aryl-(CH 2 ) n -, or C 3 -C 7 cycloalkyl;
- R 16 is OH or C r C 6 alkoxy
- R 17 , R 18 and R 19 are independently H or C r C 6 alkyl;
- R 22 and R 23 are independently Ci-C 6 alkyl, C 3 -C 7 cycloalkyl-(CH 2 ) n -, OH-Ci-C 6 alkyl, or aryl;
- each R 24 is independently H, C 1 -C 6 alkyl, NH 2 , NH 2 -C(O)-NH-, NH 2 -C(O)-, NH 2 -C(O)-(CH 2 V, OH-C(O)-, NH 2 -C(O)-(CH 2 ) n -NH-C(O)-, (OH) 2 -C 1 -C 6 alkyl- NH-C(O)-, or OH-C 1 -C 6 alkyl-NH-C(O)-;
- each R 26 is independently H, OH, OH-C 1 -C 6 alkyl, aryl-
- R 27 and R 28 independently are H or NH 2 -C(O)-;
- R 29 R 33 , R 34 , R 36 and R 38 are independently H or C 1 -C 6 alkyl
- R 30 is C 1 -C 6 alkyl, C 3 -C 7 cycloalkyl or NH 2 ;
- R 31 is H
- X is CH or N
- R 1 is R 4 i- 5 -benzyl, R 5 i -5 -isoxazolyl- CH 2 - or R 5 i -5 -pyridinyl- CH 2 -; wherein each R 4 is H, fluoro, cyano, NH 2 -C(O)-; each R 5 is independently H or CH 3 ;
- R 2 is NR 11 R 12 -C(O)-R 13 CH-, R 16 -C(O)-R 13 CH-, NR 17 R 18 -C(O)-CH 2 -NR 19 -C(O)- R 13 CH-, NR 17 R 18 -C(O)- (CH 2 ) 2 -NR 19 -C(O)-R 13 CH-, R 22 R 23 CH-, R 24 i -5 -furyl-R 13 CH-, R 24 i -5 -oxadiazolyl-R 13 CH-, R 24 1-5 -tetrazolyl-R 13 CH-, R 26 1-5 -cyclohexyl, R 2 V 5 - tetrahydronapthyl,
- R 11 and R 12 are independently H, CH 3 , (CH 3 ) 2 CH-, cyclobutyl, cyclopropyl,
- R 13 is H, (CH 3 ) 3 C, (CHs) 2 CHCH 2 -, (CH 3 ) 2 CH-, OH-ethyl, benzyl, phenyl, or cyclohexyl;
- R 16 is OH or CH 3 O;
- R 17 , R 18 and R 19 are independently H or CH 3 ;
- R 22 and R 23 are independently (CH 3 ) 3 C-, (CH 3 ) 2 CH-, cyclohexyl-CH 2 -, OHCH 2 , phenyl, OH-isopropyl, or OH-ethyl;
- each R 24 is independently H, CH 3 , NH 2 , NH 2 -C(O)-NH-, NH 2 -C(O)-, NH 2 -C(O)-CH 2 -, OH-C(O)-, NH 2 -C(O)-CH 2 -NH-C(O)-, (OH) 2 -propyl-NH-C(O)-, or OH-ethyl-NH-C(O)-;
- each R 26 is independently H, OH, OHCH 2 , benzyl-O-, NH 2 -C(O)- or CH 3 CH 2 -O-
- R j2'9 a D R3 J 3 J , o R3 M 4, D R3 J 6 b and R ,3 J 8 B are independently H or CH 3 ;
- R j 3 J 0 U is CH 3 , cyclopropyl or NH 2 ;
- R 31 is H
- X is CH or N; R 1 is
- each R 3 is independently H, CH 3 , chloro, bromo, fluoro, phenyl, NH 2 -C(O)-, CH 3 O, 3-pyridinyl, 4-pyridinyl, or 2-oxazolyl.
- X is CH.
- R 1 is R 4 i. 5 -aryl-(CH 2 )n- or R 5 1-5 -heteroaryl-(CH2)n-; wherein each R 4 is independently H, halo, cyano, or NH 2 -C(O)-; each R 5 is independently H or CrC 6 alkyl;
- R 2 is NR 11 R 12 -C(O)-R 13 CH-, NR 17 R 18 -C(O)-(CH 2 )n-NR 19 -C(O)-R 13 CH-, R 22 R 23 CH-, R 2 V 5 -heteroaryl-R 13 CH, R 30 -SO 2 -NR 31 -(CH 2 ) n -NR 19 -C(O)-R 13 CH-, R 30 - SO 2 -(CH 2 ) n -NR 31 -C(O)-R 13 CH- or R 32 -C(O)-R 33 CH-NR 34 -C(O)-R 13 CH-; wherein R 11 and R 12 are independently H, OH-CrC 6 alkyl, (OH) 2 -CrC 6 alkyl, C 3 - C 7 cycloalkyl or (OH-CrC 6 alkyl) 2 -(CH 2 ) n -;
- R 13 is CrC 6 alkyl
- R 17 , R 18 and R 19 are independently H;
- R 22 and R 23 are independently C 1 -C 6 alkyl or OH-C 1 -C 6 alkyl; each R 24 is independently Hor NH 2 ;
- R 30 is C 3 -C 7 cycloalkyl or NH 2 ;
- R 31 is H
- R 32 is OH
- R 33 is H
- R 34 is H; n is an integer from 1 to 6; and R 3 is H, halo or C 1 -C 6 alkyl;
- X is CH; R 1 is
- R 3 is H, F, Cl or CH 3 ;
- X is N
- R 1 is R 4 i.5-aryl-(CH 2 )n- or R 5 1-5 -heteroaryl-(CH 2 ) n -; wherein each R 4 is independently H, halo, cyano, or NH 2 -C(O)-; each R 5 is independently H; R 2 is NR 11 R 12 -C(O)-R 13 CH-, R 22 R 23 CH- or R 16 -C(O)-R 13 CH-; wherein
- R 11 and R 12 are independently H;
- R 13 is C 1 -C 6 alkyl or OH-CrC 6 alkyl
- R 22 and R 23 are independently C r C 6 alkyl or OH-Ci-C 6 alkyl; n is an integer from 1to 6; and R 3 is H.
- X is N
- R 1 is R 4 i- 5 -benzyl or R 5 1-5 -pyridinyl-CH 2 -; wherein each R 4 is H or fluoro; each R 5 is independently H; R 2 is NR 11 R 12 -C(O)-R 13 CH-, R 22 R 23 CH- or R 16 -C(O)-R 13 CH-; wherein
- R 11 and R 12 are independently H;
- R 13 is (CHa) 3 C, (CHs) 2 CHCH 2 , (CH 3 ) 2 CH,OH-ethyl;
- R 16 is OH
- R 22 and R 23 are independently (CH 3 ) 3 C or OHCH 2 ; and R 3 is H.
- X is N
- R 3 is H.
- R 2 * is selected from
- R 11 and R 12 are independently H, Ci-C 6 alkyl, OH-CrC 6 alkyl,
- R 13 is H, C 1 -C 6 alkyl, OH-CrC 6 alkyl, aryl, aryl-(CH 2 ) n -, or C 3 -C 7 cycloalkyl;
- R 15 , R 29 , R 31 , R 33 , R 34 , R 36 , R 39 and R 40 are independently H or
- R 17 , R 18 and R 19 are independently H or C r C 6 alkyl; each R 24 is independently H, CrC 6 alkyl, C 3 -C 7 cycloalkyl, CrC 6 haloalkyl, oxo, NH 2 , CrC 6 alkoxy-C(O)-, NH 2 -C(O)-(CH 2 ) n -,
- each R 25 is independently H or oxo;
- R 27 and R 28 independently are H, NH 2 -C(O)-, or C 3 -C 7 cycloalkyl-C(O)-;
- R 30 is CrC 6 alkyl, C 3 -C 7 cycloalkyl or NH 2 ;
- R 32 is OH
- R 3A and R 3B are independently selected from H and halo; R 4A is selected from F and CN; and R 4B is selected from H and F.
- R 13 is C 1 -C6 alkyl. More preferably it is branched C 3 -C 6 alkyl. Most preferably it is tert-butyl.
- R 3A is selected from H, F and Cl
- R 4A is selected from F and CN
- R 4B is selected from H and F
- R 11A is selected from H, OH-Ci-C 6 alkyl and (OH) 2 -Ci-C 6 alkyl.
- R 3A is selected from H, F and Cl
- R 4A is selected from F and CN
- R 4B is selected from H and F
- R 11A is selected from H, 2-hydroxyethyl and 2,3- dihydroxypropyl.
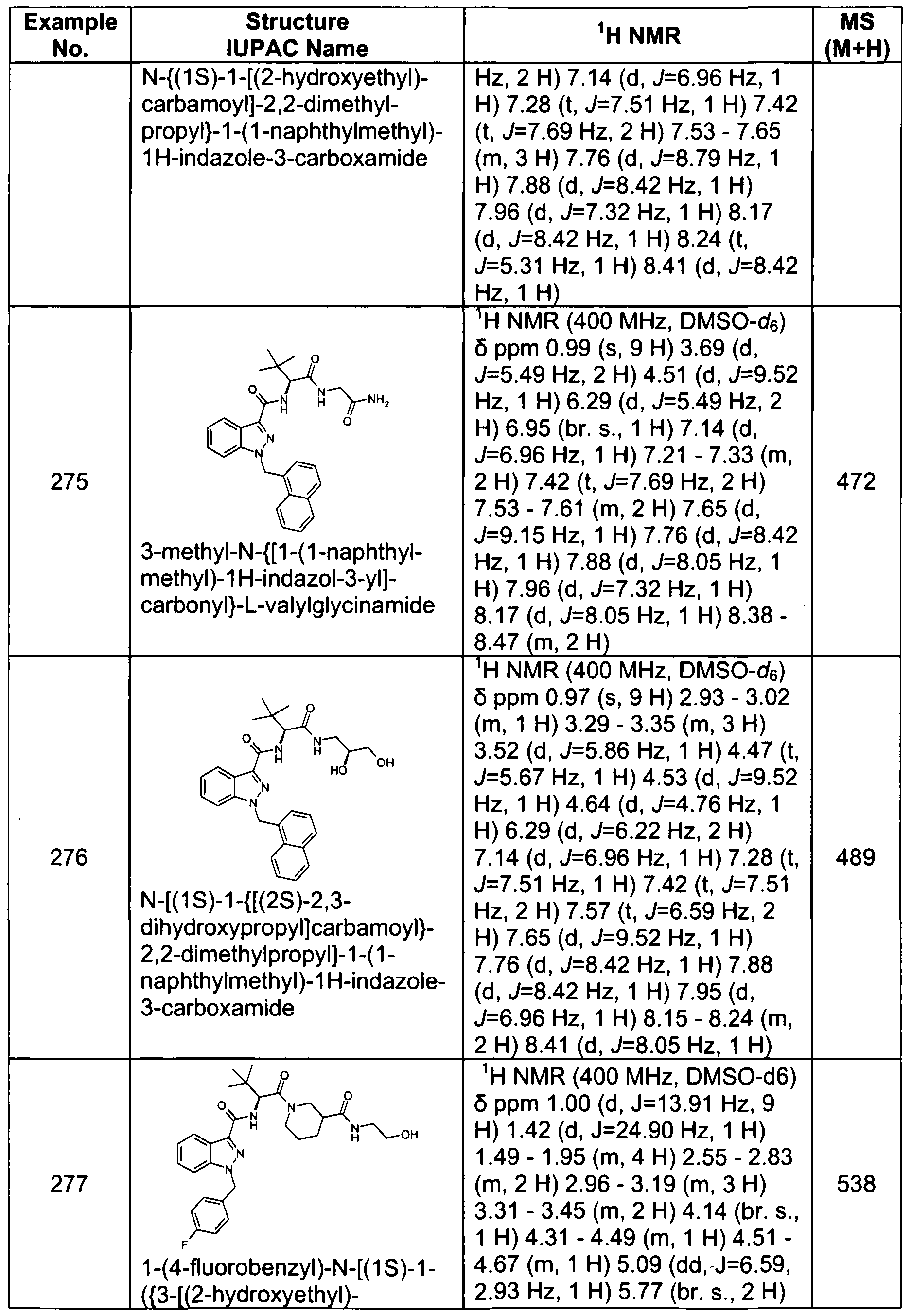
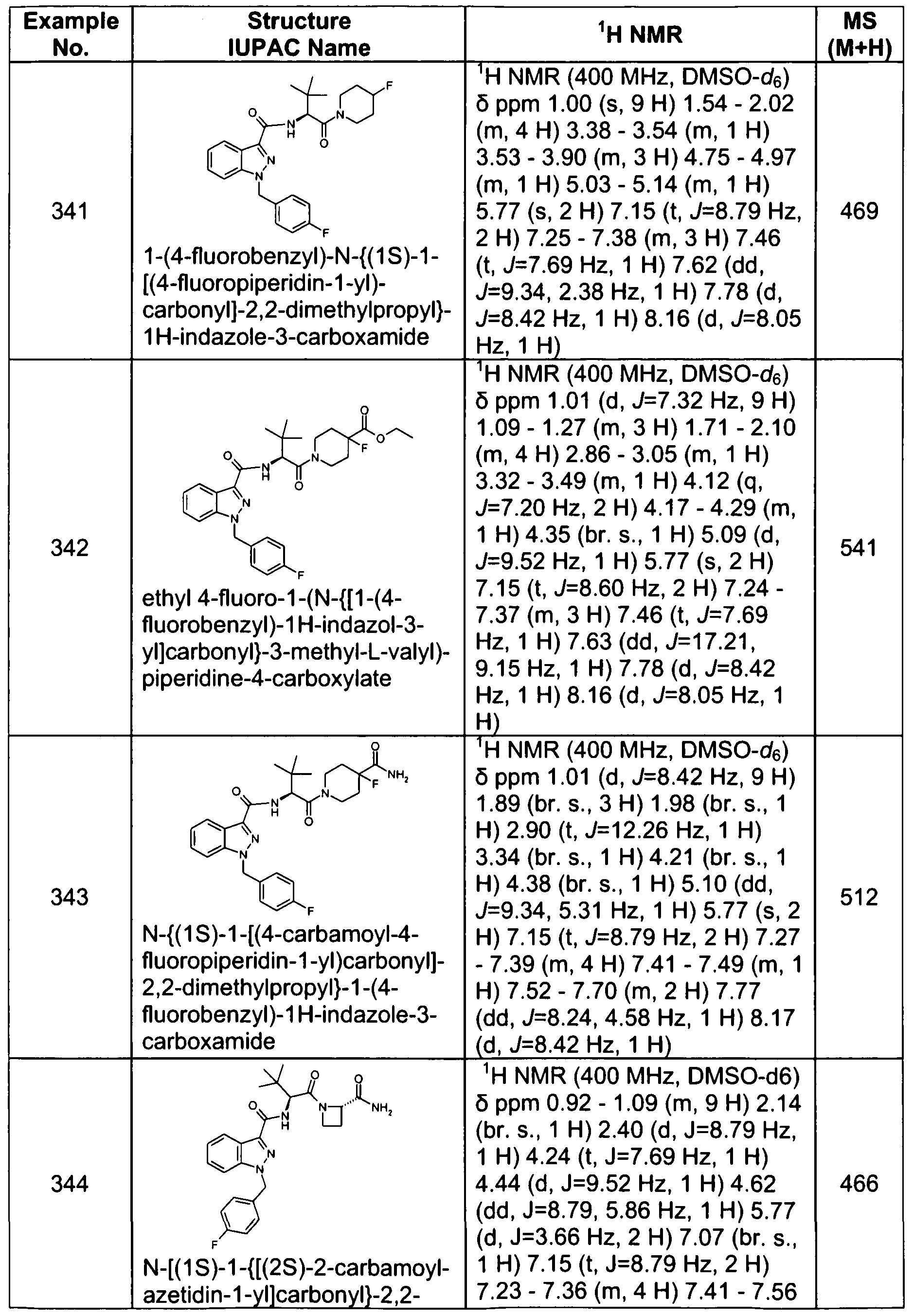
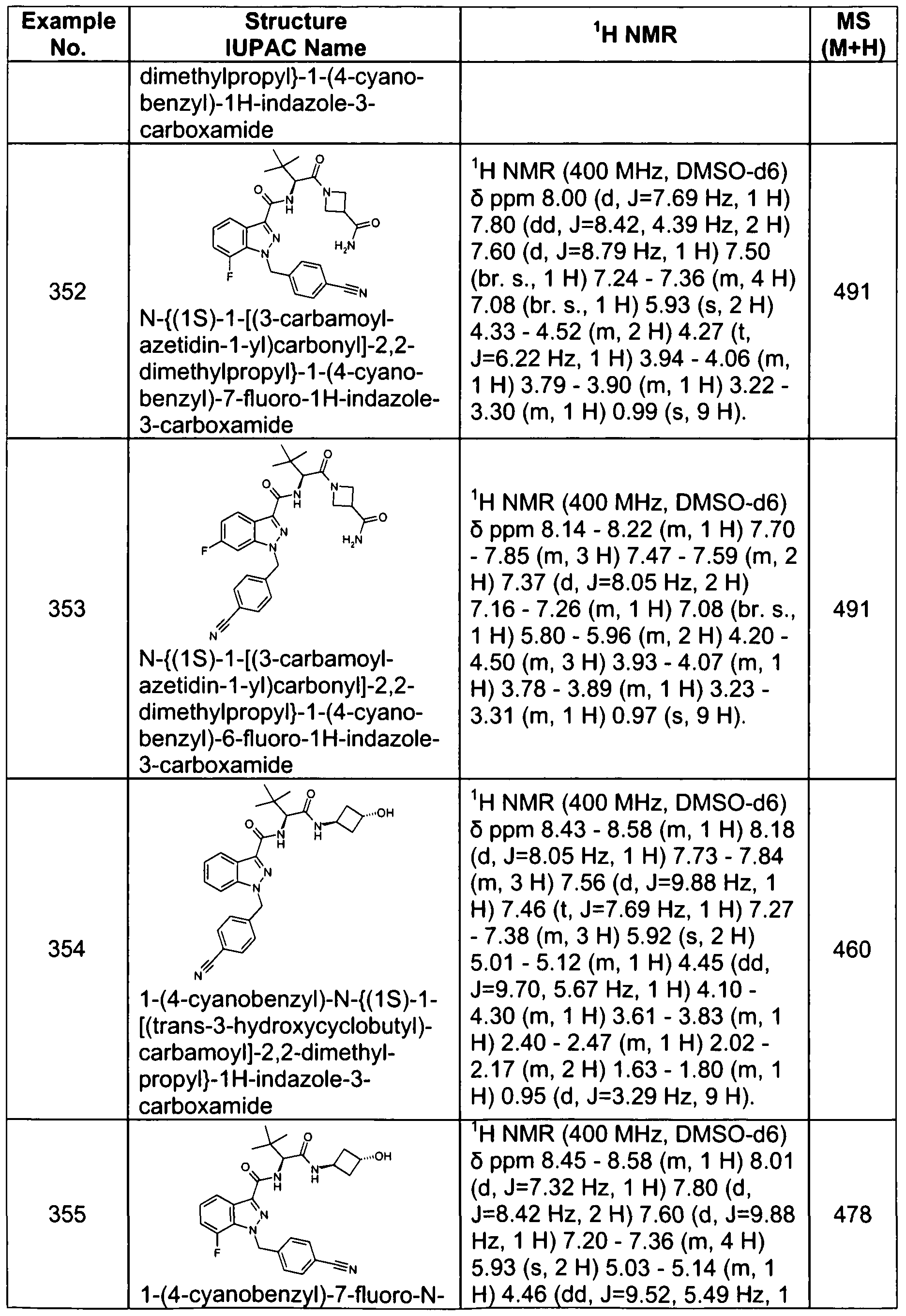
- the compound, or a pharmaceutically acceptable salt thereof is selected from the group consisting of
- the compound, or a pharmaceutically acceptable salt thereof is selected from the group consisting of
- the present invention is a pharmaceutical composition
- a pharmaceutical composition comprising a compound of Formula I or a pharmaceutically acceptable salt, enantiomer, or racemate thereof.
- the present invention is a method for the treatment of a CB1 mediated disorder in a subject in need of such treatment or prevention, wherein the method comprises administering to the subject an amount of a compound of Formula I or a pharmaceutically acceptable salt, enantiomer, or racemate thereof, wherein the amount of the compound is effective for the treatment or prevention of the CB 1 mediated disorder.
- the CB1 mediated disorder is pain.
- the compounds of this invention may be used in the form of salts derived from inorganic or organic acids.
- a salt of the compound may be advantageous due to one or more of the salt's physical properties, such as enhanced pharmaceutical stability in differing temperatures and humidities, or a desirable solubility in water or oil.
- a salt of a compound also may be used as an aid in the isolation, purification, and/or resolution of the compound.
- salts are intended to be administered to a patient (as opposed to, for example, being used in an in vitro context)
- the salt preferably is pharmaceutically acceptable.
- Pharmaceutically acceptable salts include salts commonly used to form alkali metal salts and to form addition salts of free acids or free bases. In general, these salts typically may be prepared by conventional means with a compound of this invention by reacting, for example, the appropriate acid or base with the compound.
- Pharmaceutically-acceptable acid addition salts of the compounds of this invention may be prepared from an inorganic or organic acid.
- suitable inorganic acids include hydrochloric, hydrobromic acid, hydroionic, nitric, carbonic, sulfuric, and phosphoric acid.
- Suitable organic acids generally include, for example, aliphatic, cycloaliphatic, aromatic, araliphatic, heterocyclyl, carboxyic, and sulfonic classes of organic acids.
- suitable organic acids include acetate, trifluoroacetate, formate, propionate, succinate, glycolate, gluconate, digluconate, lactate, malate, tartaric acid, citrate, ascorbate, glucuronate, maleate, fumarate, pyruvate, aspartate, glutamate, benzoate, anthranilic acid, mesylate, stearate, salicylate, p-hydroxybenzoate, phenylacetate, mandelate, embonate (pamoate), methanesulfonate, ethanesulfonate, benzenesulfonate, pantothenate, toluenesulfonate, 2-hydroxyethanesulfonate, sufanilate, cyclohexylaminosulfonate, algenic acid, b-hydroxybutyric acid, galactarate, galacturonate, adipate, alginate, bisulfate, buty
- Pharmaceutically-acceptable base addition salts of the compounds of this invention include, for example, metallic salts and organic salts.
- Preferred metallic salts include alkali metal (group Ia) salts, alkaline earth metal (group Ma) salts, and other physiological acceptable metal salts. Such salts may be made from aluminum, calcium, lithium, magnesium, potassium, sodium, and zinc.
- Preferred organic salts may be made from tertiary amines and quaternary amine salts, such as tromethamine, diethylamine, N,N'-dibenzylethylenediamine, chloroprocaine, choline, diethanolamine, ethylenediamine, meglumine (N-methylglucamine), and procaine.
- Basic nitrogen-containing groups may be quaternized with agents such as lower alkyl (CrC 6 ) halides (e.g., methyl, ethyl, propyl, and butyl chlorides, bromides, and iodides), dialkyl sulfates (e.g., dimethyl, diethyl, dibuytl, and diamyl sulfates), long chain halides (e.g., decyl, lauryl, myristyl, and stearyl chlorides, bromides, and iodides), arylalkyl halides (e.g., benzyl and phenethyl bromides), and others.
- lower alkyl (CrC 6 ) halides e.g., methyl, ethyl, propyl, and butyl chlorides, bromides, and iodides
- dialkyl sulfates e.g., dimethyl, die
- 'prodrugs' of the compounds of formula (I) are so-called 'prodrugs' of the compounds of formula (I).
- certain derivatives of compounds of formula (I) which may have little or no pharmacological activity themselves can, when administered into or onto the body, be converted into compounds of formula (I) having the desired activity, for example, by hydrolytic cleavage.
- Such derivatives are referred to as 'prodrugs'.
- Further information on the use of prodrugs may be found in 'Pro-drugs as Novel Delivery Systems, Vol. 14, ACS Symposium Series (T Higuchi and W Stella) and 'Bioreversible Carriers in Drug Design', Pergamon Press, 1987 (ed. E B Roche, American Pharmaceutical Association).
- Prodrugs in accordance with the invention can, for example, be produced by replacing appropriate functionalities present in the compounds of formula (I) with certain moieties known to those skilled in the art as 'pro-moieties' as described, for example, in “Design of Prodrugs” by H Bundgaard (Elsevier, 1985).
- Some examples of prodrugs in accordance with the invention include:
- Compounds of formula (I) containing one or more asymmetric carbon atoms can exist as two or more stereoisomers. Where the compound contains, for example, a keto or oxime group or an aromatic moiety, tautomeric isomerism ('tautomerism') can occur. It follows that a single compound may exhibit more than one type of isomerism.
- racemate (or a racemic precursor) may be reacted with a suitable optically active compound, for example, an alcohol, or, in the case where the compound of formula (I) contains an acidic or basic moiety, an acid or base such as tartaric acid or 1-phenylethylamine.
- a suitable optically active compound for example, an alcohol, or, in the case where the compound of formula (I) contains an acidic or basic moiety, an acid or base such as tartaric acid or 1-phenylethylamine.
- the resulting diastereomeric mixture may be separated by chromatography and/or fractional crystallization and one or both of the diastereoisomers converted to the corresponding pure enantiomer(s) by means well known to a skilled person.
- Chiral compounds of the invention may be obtained in enantiomerically-enriched form using chromatography, typically HPLC, on an asymmetric resin with a mobile phase consisting of a hydrocarbon, typically heptane or hexane, containing from 0 to 50% isopropanol, typically from 2 to 20%, and from 0 to 5% of an alkylamine, typically 0.1% diethylamine. Concentration of the eluate affords the enriched mixture.
- Stereoisomeric conglomerates may be separated by conventional techniques known to those skilled in the art - see, for example, "Stereochemistry of Organic Compounds” by E L Eliel (Wiley, New York, 1994).
- the present invention includes all pharmaceutically acceptable isotopically- labelled compounds of formula (I) wherein one or more atoms are replaced by atoms having the same atomic number, but an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
- isotopes suitable for inclusion in the compounds of the invention include isotopes of hydrogen, such as 2 H and 3 H, carbon, such as 11 C, 13 C and 14 C, chlorine, such as 36 CI, fluorine, such as 18 F, iodine, such as 123 I and 125 I, nitrogen, such as 13 N and 15 N, oxygen, such as 15 0, 17 O and 18 O, phosphorus, such as 32 P, and sulphur, such as 35 S.
- isotopically-labelled compounds of formula (I), for example, those incorporating a radioactive isotope, are useful in drug and/or substrate tissue distribution studies.
- the radioactive isotopes tritium, i.e. 3 H, and carbon-14, i.e. 14 C, are particularly useful for this purpose in view of their ease of incorporation and ready means of detection.
- substitution with heavier isotopes such as deuterium, i.e. 2 H, may afford certain therapeutic advantages resulting from greater metabolic stability, for example, increased in vivo half-life or reduced dosage requirements, and hence may be preferred in some circumstances.
- Isotopically-labeled compounds of formula (I) can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying Examples and Preparations using an appropriate isotopically-labeled reagents in place of the non-labeled reagent previously employed. All of the compounds of the formula (I) can be prepared by the procedures described in the general methods presented below or by the specific methods described in the Examples section and the Preparations section, or by routine modifications thereof. The present invention also encompasses any one or more of these processes for preparing the compounds of formula (I), in addition to any novel intermediates used therein.
- the method of the present invention is useful for, but not limited to, the treatment of disorders that are mediated by CB 1 in a subject.
- the compounds described herein would be useful for the treatment of any symptoms associated with a CB1 meditated disorder described below.
- Treating includes palliative treatment, preventive treatment and restorative treatment.
- Palliative treatment includes alleviation, elimination of causation of pain and/or inflammation associated with a CB1 mediated disorder.
- Preventaive treatment means to prevent or to slow the appearance of symptoms associated with a CB1 mediated disorder.
- the subject is any subject, and preferably is a subject that is in need of prevention of a CB1 mediated disorder.
- subject for purposes of treatment includes any human or animal subject who is in need of the prevention of, or who has a TNF ⁇ -mediated inflammatory disease or disorder.
- the subject is typically a mammal.
- the methods and compositions of the present invention encompass the treatment of conditions including pain and neurodegenerative disorders.
- pain and neurodegenerative disorders See Annu. Rev. Pharmacol. Toxicol. (2006) 46:101-22; Clinical Neuroscience Research (2005)5 185-199; Prostaglandins, Leukotrienes and Essential Fatty Acids (2002) 66(2&3), 101-121.
- the methods and compositions of the present invention encompass the treatment of pain, including but not limited to chronic pain, acute pain, joint pain, nociceptive pain, neuropathic pain, allodynia, hyperalgesia, burn pain, menstrual cramps, kidney stones, headache, migraine headache, sinus headaches, tension headaches, dental pain, myasthenia gravis, rheumatoid arthritic pain, osteoarthritic pain, back pain, cancer pain, multiple sclerosis, sarcoidosis, Behcet's syndrome, myositis, polymyositis, gingivitis, hypersensitivity, swelling occurring after injury, closed head injury, endometriosis, stroke, and the like.
- pain including but not limited to chronic pain, acute pain, joint pain, nociceptive pain, neuropathic pain, allodynia, hyperalgesia, burn pain, menstrual cramps, kidney stones, headache, migraine headache, sinus headaches, tension headaches, dental pain, myasthenia gravis,
- the methods and compositions of the present invention encompass the treatment of the connective tissue and joint disorders selected from the group consisting of osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, fibromyalgia, spondyloarthopathies, gouty arthritis, lumbar spondylarthrosis, carpal tunnel syndrome, psoriatic arthritis, sclerodoma , canine hip dysplasia, systemic lupus erythematosus, juvenile arthritis, osteoarthritis, tendonitis and bursitis.
- connective tissue and joint disorders selected from the group consisting of osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, fibromyalgia, spondyloarthopathies, gouty arthritis, lumbar spondylarthrosis, carpal tunnel syndrome, psoriatic arthritis, sclerodoma , canine hip dysplasia,
- the methods and compositions of the present invention encompass the treatment of neurological dosirders including neuroinflammation and neurodegenerative disorders selected from the group consisting of neuritis, Alzheimer's disease, multiple sclerosis (MS), Parkinson's disease, Tourette's syndrome, spasticity and epilepsy.
- neurological dosirders including neuroinflammation and neurodegenerative disorders selected from the group consisting of neuritis, Alzheimer's disease, multiple sclerosis (MS), Parkinson's disease, Tourette's syndrome, spasticity and epilepsy.
- the methods and compositions of the present invention encompass the treatment of neuropathies including HIV related neuropathy, nerve injury, spinal cord injury, sciatica, neuralgia, diabetic neuropathy, nerve pain, and some peripheral neuropathies and neurodegenerative disorders.
- the methods and compositions of the present invention encompass the treatment of the respiratory disorders selected from the group consisting of cough, asthma, bronchitis, chronic obstructive pulmonary disease (COPD), broncho constriction, cystic fibrosis, pulmonary edema, pulmonary embolism, pneumonia, pulmonary sarcoisosis, silicosis, pulmonary fibrosis, respiratory failure, acute respiratory distress syndrome, seasonal allergic rhinitis, reversible airway obstruction, adult respiratory disease syndrome, cryptogenic fibrosing alveolitis and emphysema.
- COPD chronic obstructive pulmonary disease
- the methods and compositions of the present invention encompass the treatment of the dermatological disorders selected from the group consisting of acne, psoriasis, eczema, burns, poison ivy, poison oak and dermatitis.
- the methods and compositions of the present invention encompass the treatment of the surgical disorders selected from the group consisting of pain and swelling following surgery, infection following surgery and inflammation following surgery.
- the methods and compositions of the present invention encompass the treatment of the gastrointestinal disorders selected from the group consisting of colitis, inflammatory bowel disease, irritable bowel syndrome, Crohn's disease, gastritis, irritable bowel syndrome, diarrhea, constipation, dysentery, ulcerative colitis, gastric esophageal reflux, gastric ulcers, gastric varices, ulcers, functional gastrointestinal disorder, and heartburn.
- the methods and compositions of the present invention encompass the treatment of the ophthalmic disorders selected from the group consisting of retinopathies, uveitis, ocular photophobia, acute injury to the eye tissue, conjunctivitis, age-related macular degeneration diabetic retinopathy, detached retina, glaucoma, vitelliform macular dystrophy type 2, gyrate atrophy of the choroid and retina, conjunctivitis, corneal infection, fuchs' dystrophy, iridocorneal endothelial syndrome, keratoconus, lattice dystrophy, map-dot-fingerprint dystrophy, ocular herpes, pterygium, myopia, hyperopia, and cataracts.
- the ophthalmic disorders selected from the group consisting of retinopathies, uveitis, ocular photophobia, acute injury to the eye tissue, conjunctivitis, age-related macular degeneration diabetic retinopathy, detached
- Cannabinoid agonists are believed to be useful in the treatment of other disorders including acute cerebral ischemia, neuroprotection, anxiety, cerebrovascular ischemia, cachexia, nausea, emesis, chemotherapy-induced emesis, cutaneous T cell lymphoma, diabetes, osteoporosis, glomerulonephritis, renal ischemia, nephritis, hepatitis, cerebral stroke, vasodialation, hypertension, vasculitis, myocardial infarction and cerebral ischemia.
- compositions Containing the Compounds of this Invention
- This invention also is directed to pharmaceutical compositions (or “medicaments") comprising the compounds described above (including tautomers of the compounds, and pharmaceutically-acceptable salts of the compounds and tautomers), and to methods for making pharmaceutical compositions comprising those compounds in combination with one or more conventional non-toxic, pharmaceutically- acceptable carriers, diluents, wetting or suspending agents, vehicles, and/or adjuvants (the carriers, diluents, wetting or suspending agents, vehicles, and adjuvants sometimes being collectively referred to in this specification as "carrier materials”); and/or other active ingredients.
- carrier materials the carriers, diluents, wetting or suspending agents, vehicles, and adjuvants sometimes being collectively referred to in this specification as "carrier materials”
- carrier materials the carriers, diluents, wetting or suspending agents, vehicles, and adjuvants sometimes being collectively referred to in this specification as "carrier materials”
- the pharmaceutical composition is made in the form of a dosage unit containing a particular amount of the active ingredient.
- the pharmaceutical composition contains from about 0.1 to 1000 mg (and more typically, 7.0 to 350 mg) of the compound.
- the compounds of the invention can also be administered intranasally or by inhalation, typically in the form of a dry powder (either alone, as a mixture, for example, in a dry blend with lactose, or as a mixed component particle, for example, mixed with phospholipids, such as phosphatidylcholine) from a dry powder inhaler or as an aerosol spray from a pressurised container, pump, spray, atomiser (preferably an atomiser using electrohydrodynamics to produce a fine mist), or nebuliser, with or without the use of a suitable propellant, such as 1 ,1 ,1 ,2-tetrafluoroethane or 1 ,1,1 ,2,3,3,3-heptafluoropropane.
- the powder may comprise a bioadhesive agent, for example, chitosan or cyclodextrin.
- the pressurised container, pump, spray, atomizer, or nebuliser contains a solution or suspension of the compound(s) of the invention comprising, for example, ethanol, aqueous ethanol, or a suitable alternative agent for dispersing, solubilising, or extending release of the active, a propellant(s) as solvent and an optional surfactant, such as sorbitan trioleate, oleic acid, or an oligolactic acid.
- a solution or suspension of the compound(s) of the invention comprising, for example, ethanol, aqueous ethanol, or a suitable alternative agent for dispersing, solubilising, or extending release of the active, a propellant(s) as solvent and an optional surfactant, such as sorbitan trioleate, oleic acid, or an oligolactic acid.
- the drug product Prior to use in a dry powder or suspension formulation, the drug product is micronised to a size suitable for delivery by inhalation (typically less than 5 microns). This may be achieved by any appropriate comminuting method, such as spiral jet milling, fluid bed jet milling, supercritical fluid processing to form nanoparticles, high pressure homogenisation, or spray drying.
- comminuting method such as spiral jet milling, fluid bed jet milling, supercritical fluid processing to form nanoparticles, high pressure homogenisation, or spray drying.
- Capsules made, for example, from gelatin or hydroxypropylmethylcellulose
- blisters and cartridges for use in an inhaler or insufflator may be formulated to contain a powder mix of the compound of the invention, a suitable powder base such as lactose or starch and a performance modifier such as l-leucine, mannitol, or magnesium stearate.
- the lactose may be anhydrous or in the form of the monohydrate, preferably the latter.
- Other suitable excipients include dextran, glucose, maltose, sorbitol, xylitol, fructose, sucrose and trehalose.
- a suitable solution formulation for use in an atomiser using electrohydrodynamics to produce a fine mist may contain from 1 ⁇ g to 20mg of the compound of the invention per actuation and the actuation volume may vary from 1 ⁇ l to 100 ⁇ l.
- a typical formulation may comprise a compound of the invention, propylene glycol, sterile water, ethanol and sodium chloride.
- Alternative solvents which may be used instead of propylene glycol include glycerol and polyethylene glycol.
- Suitable flavours such as menthol and levomenthol, or sweeteners, such as saccharin or saccharin sodium, may be added to those formulations of the invention intended for inhaled/intranasal administration.
- Formulations for inhaled/intranasal administration may be formulated to be immediate and/or modified release using, for example, PGLA.
- Modified release formulations include delayed-, sustained-, pulsed-, controlled-, targeted and programmed release.
- the dosage unit is determined by means of a valve which delivers a metered amount.
- Units in accordance with the invention are typically arranged to administer a metered dose or "puff' containing from 0.001 mg to 10mg of the compound of the invention.
- the overall daily dose will typically be in the range 0.001 mg to 40mg which may be administered in a single dose or, more usually, as divided doses throughout the day.
- Solid dosage forms for oral administration include, for example, hard or soft capsules, tablets, pills, powders, and granules.
- the compounds are ordinarily combined with one or more adjuvants.
- the compounds may be mixed with lactose, sucrose, starch powder, cellulose esters of alkanoic acids, cellulose alkyl esters, talc, stearic acid, magnesium stearate, magnesium oxide, sodium and calcium salts of phosphoric and sulfuric acids, gelatin, acacia gum, sodium alginate, polyvinylpyrrolidone, and/or polyvinyl alcohol, and then tableted or encapsulated for convenient administration.
- Such capsules or tablets may contain a controlled-release formulation, as may be provided in a dispersion of the compound of this invention in hydroxypropylmethyl cellulose.
- the dosage forms also may comprise buffering agents, such as sodium citrate, or magnesium or calcium carbonate or bicarbonate. Tablets and pills additionally may be prepared with enteric coatings.
- Liquid dosage forms for oral administration include, for example, pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs containing inert diluents commonly used in the art (e.g., water). Such compositions also may comprise adjuvants, such as wetting, emulsifying, suspending, flavoring (e.g., sweetening), and/or perfuming agents.
- adjuvants such as wetting, emulsifying, suspending, flavoring (e.g., sweetening), and/or perfuming agents.
- Parenter administration includes subcutaneous injections, intravenous injections, intramuscular injections, intrasternal injections, and infusion.
- injectable preparations e.g., sterile injectable aqueous or oleaginous suspensions
- suitable dispersing, wetting agents, and/or suspending agents may be formulated according to the known art using suitable dispersing, wetting agents, and/or suspending agents.
- Acceptable carrier materials include, for example, water, 1 ,3-butanediol, Ringer's solution, isotonic sodium chloride solution, bland fixed oils (e.g., synthetic mono- or diglycerides), dextrose, mannitol, fatty acids (e.g., oleic acid), dimethyl acetamide, surfactants (e.g., ionic and non-ionic detergents), and/or polyethylene glycols (e.g., PEG 400).
- suitable fixed oils e.g., synthetic mono- or diglycerides
- dextrose e.g., mannitol
- fatty acids e.g., oleic acid
- dimethyl acetamide e.g., dimethyl acetamide
- surfactants e.g., ionic and non-ionic detergents
- polyethylene glycols e.g., PEG 400
- Formulations for parenteral administration may, for example, be prepared from sterile powders or granules having one or more of the carriers materials mentioned for use in the formulations for oral administration.
- the compounds may be dissolved in water, polyethylene glycol, propylene glycol, ethanol, com oil, cottonseed oil, peanut oil, sesame oil, benzyl alcohol, sodium chloride, and/or various buffers.
- the pH may be adjusted, if necessary, with a suitable acid, base, or buffer.
- Starting compound 1 wherein X is either carbon or nitrogen and R* is a carboxyl protecting group such as alkyl or aralkyl, can be treated with a base and an alkylating agent.
- bases include sodium hydride, potassium tert-butoxide, sodium hexamethyldisilazide, and potassium carbonate
- exemplary alkylating agents include R 1 -L where L is a leaving group, such as a halogen, or a mesylate, or a tosylate, and R 1 is as described in the description of general formula (I).
- the reaction generally produces a mixture of regioisomers wherein the alkylation occurs either on N1 or N2 position of the indazole ring, depending upon the base and the alkylating agent.
- the desired N 1 -alkylated regioisomer is isolated in pure form by either chromatographic separation, or recrystallization of the crude product mixture. Saponification of the alkylated product with an aqueous base such as sodium hydroxide, potassium hydroxide, or lithium hydroxide gives compound 2.
- Compound 2 may be coupled with an amine 3 by using reaction conditions well known in the art for peptide bond synthesis [see, for example, Bodanszky and Bodanszky, The Practice of Peptide Chemistry. Springer- Verlag (1984); Bodanszky, Principles of Peptide Synthesis. Springer-Verlag (1984); Han, S-Y and Kim, Y-A, Tetrahedron, vol. 60, pp 2447-2467 (2004)] to give a compound of formula (I).
- Exemplary reagents for activating the carboxyl group of compound 2 for reacting with the amine 3 include carbodiimide reagents such as N.N'-dicyclohexylcarbodiimide (DCC) and 1-[3-(dimethylamino)propyl]-3-ethylcarbodimide (EDC), either alone or in combination with 1-hydroxybenzotriazole (HOBt), and uronium reagents such as O-(7- azabenzotriazol-1-yl)-1 ,1 ,3,3-tetramethyluronium hexafluorophosphate (HATU), O- (benzotriazol-1-yl)-1 ,1 ,3,3-tetramethyluronium hexafluorophosphate (HBTU), and O- (benzotriazol-1-yl)-1 ,1 ,3,3-tetramethyluronium tetrafluoroborate (TBTU).
- Starting compound 1 wherein X is a nitrogen and R* is a carboxyl protecting group such as alkyl or aralkyl, can be prepared according to known methods in the literature [see, for example, Lynch, B. M. et al, Canadian Journal of Chemistry, vol. 66, pp 420-428 (1988); Huang, S. et al, Bioor ⁇ anic & Medicinal Chemistry Letters, vol. 17, pp1243-1245 (2007); Lin, R. et al, Bioor ⁇ anic & Medicinal Chemistry Letters, vol. 17, pp 4297-4302 (2007)].
- Amine compounds 3 (R 2 -NH 2 ) are either commercially available, or readily prepared according to methods known in the art as depicted in the protocols for representative Preparations herein.
- the compounds, salts and solvates (including hydrates) of the invention may be separated and purified by conventional methods.
- Separation of diastereomers may be achieved by conventional techniques, e.g. by chromatography or HPLC of a stereoisomeric mixture of a compound of formula (I) or a suitable salt or derivative thereof.
- An individual enantiomer of a compound of formula (I) may also be prepared from a corresponding optically pure intermediate or by resolution, such as by chromatography of the corresponding racemate using a suitable chiral support or by fractional crystallization of the diastereomeric salts formed by a reaction of the corresponding racemate with a suitable optically active acid or base.
- the Human CB1 receptor binding affinity and other biological activities of the compounds of this invention are determined by the following procedures.
- Membrane preparation Human Embryonic Kidney (HEK) Cells expressing the human CB 1 receptor under transcriptional regulation of a tetracycline inducible promoter were grown in Dulbecco's Modified Essential Medium with sodium pyruvate (Invitrogen, Carlsbad, CA) containing 10% tetracycline free fetal bovine serum (Clonetech, Mountain View, CA) 100 ⁇ g/ml hygromycin (Calbiochem, San Diego, CA), 5 ug/ml blasticidin (Invitrogen).
- HEK Human Embryonic Kidney
- CB1 receptor expression was induced by addition of 1 ⁇ g/ml doxycycline (Calbiochem) and incubation for an additional 24 hours.
- Cells were released from flasks using Cell Dissociation Buffer (Invitrogen). Cells were pelleted by centrifugation at 500 X G for 5 minutes.
- Membranes were prepared by resuspending cells in ice cold TEE Buffer (25mM Tris pH 7.4, 5mM EDTA, 5mM EGTA, Complete Protease Inhibitor (Roche, Basel, Switzerland)). Cells were lysed with 12 strokes of a dounce homogenizer. Unlysed cells were pelleted by centrifugation at 500 X G for 5 minutes.
- Membranes were pelleted by centrifugation at 25,000 X G for 30 minutes. Membranes were resuspended in TEE 1 dounced 12 strokes, and pelleted a second time at 25,000 X G for 30 minutes. Membrane pellet was resuspended in 5OmM Tris pH 7.4, 10OmM NaCI, 3mM MgCI 2 , 0.2mM EGTA, Complete Protease Inhibitor (Roche). Protein concentration was determined using the Micro-BCA Protein Assay Kit (Pierce, Rockford, IL) using BSA as a standard. Membranes were quick frozen and stored at -80 degrees Celsius until use.
- Membrane preparation CHO cells expressing the human CB1 receptor were grown to 80% confluence in Ham's F-12 Nutrient Medium (Invitrogen) containing 10% fetal bovine serum (Invitrogen), 1% pen/strep (Invitogen), 1% Nonessential amino acids (Invitrogen) and 500 ⁇ g/ml G418 (Invitrogen). Cells were released from flasks using Cell Dissociation Buffer (Invitrogen). Cells were pelleted by centrifugation at 500 X G for 5 minutes.
- Membranes were prepared by resuspending cells in ice cold Assay Buffer (25mM Tris pH 7.4, 5mM EDTA, 5mM EGTA, Complete Protease Inhibitor (Roche)). Cells were lysed with 12 strokes of a dounce homogenizer. Unlysed cells were pelleted by centrifugation at 500 X G for 5 minutes. Membranes were pelleted by centrifugation at 25,000 X G for 30 minutes. Membranes were resuspended in TEE, dounced 12 strokes, and pelleted a second time at 25,000 X G for 30 minutes.
- Membrane pellet was resuspended in 5OmM Tris pH 7.4, 10OmM NaCI, 3mM MgCb, 0.2mM EGTA, Complete Protease Inhibitor (Roche). Protein concentration was determined using the Micro-BCA Protein Assay Kit (Pierce) using BSA as a standard. Membranes were frozen and stored at -80 degrees Celsius until use.
- GTPvS Binding 40 ⁇ l of test compound was incubated with 20 ⁇ l of [ 35 S] GTPyS (Perkin Elmer) (1250 Ci/millimole) and 140 ⁇ l of membrane homogenate (5 ug/well) in polypropylene 96-well plates (Corning).
- the above protocol assays were used to determine biological activity.
- the Ki towards human CB1 receptors for certain compounds of the invention are measured to be 0.01-1000 nM.
- the EC50 towards human CB1 receptors in the GTPyS assay for certain compounds of the invention are measured to be 0.1-5000 nM.
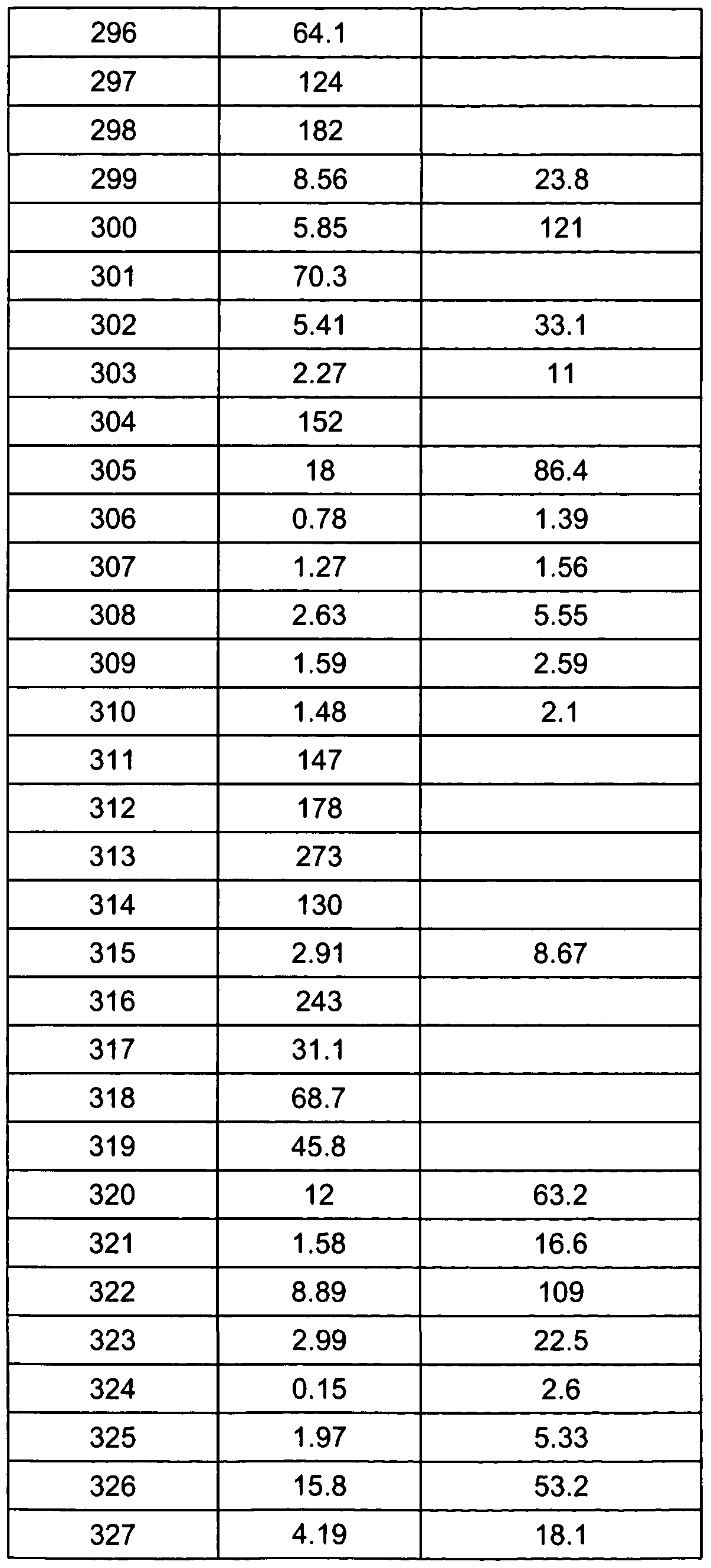
- Table 1 shows certain biological activities for some of the exemplified compounds.
- the invention is illustrated in the following non-limiting examples and preparations in which, unless stated otherwise: all operations were carried out at room or ambient temperature, that is, in the range of 18-25 degrees Celsius; evaporation of solvent was carried out using a rotary evaporator under reduced pressure with a bath temperature of up to 60 degrees Celsius; reactions were monitored by thin layer chromatography (TLC) and reaction times are given for illustration only; melting points (mp) given are uncorrected (polymorphism may result in different melting points); the structure and purity of all isolated compounds were assured by at least one of the following techniques: TLC (Merck silica gel 60 F 25 4 precoated TLC plates or Merck NH 2 gel (an amine coated silica gel) F 2 &4s precoated TLC plates), mass spectrometry, nuclear magnetic resonance spectra (NMR), infrared absorption spectra (IR) or microanalysis.
- TLC Merck silica gel 60 F 25 4 precoated TLC plates or Merck NH 2 gel (an
- Low-resolution mass spectral data (El) were obtained on an Integrity (Waters) mass spectrometer.
- Low-resolution mass spectral data (ESI) were obtained on ZMDTM or ZQTM (Waters) and mass spectrometer.
- IR spectra were measured by a Fourier transform infrared spectrophotometer (Shimazu FTIR-8300). Chemical symbols have their usual meanings; bp (boiling point), mp (melting point), rt (room temperature), L (liter(s)), ml_ (milliliter(s)), g (gram(s)), mg (milligram(s)), mol (moles), mmol (millimoles), eq. (equivalent(s)), quant, (quantitative yield).
- CDI N 1 N'- carbonyldiimidazole
- DMF N,N-dimethylformamide
- DMSO dimethylsulfoxide
- EDCHCI 1-ethyl-3-(3- dimethylaminopropyl)carbodiimide hydrochloride
- HATU 2-(7-aza-1 H-benzotriazol-1-yl)-1 ,1 ,3,3-tetramethyluronium hexafluorophosphate]
- TBTU 2-(1 H-benzotriazol-1-yl)-1 ,1 ,3,3-tetramethyluronium tetrafluoroborate]
- EtOH ethanol
- HOBt 1-Hydroxy-1 H-benzotriazole
- MeOH methanol
- THF tetrahydrofuran
- TFA trifluoroacetic acid
- Step 3 N-[(1 S)-1 -(aminocarbonyl)-2,2-dimethylpropyl]-1 -(4-f luorobenzyl)-1 H- indazole-3-carboxamide
- Step 1 Methyl i-KS-methylisoxazol-S-ylJmethyll-IH-indazole-S-carboxylate
- Step 2 i-Benzyl- ⁇ -bromo-IH-indazole-S-carboxylic acid
- the mixture contains some tetramethyl urea from the HATU.
- the residue was dissolved in dichloromethane and washed 6 times with brine, dried over anhydrous magnesium sulfate and concentrated under reduced pressure.
- Example 8 N-[(1 S)-1 -(Aminocarbonyl)-2,2-dimethylpropyl]-1 -benzyl-5-pyridin-3- yl-1 H-indazole-3-carboxamide
- Nitrogen gas was bubbled through the mixture for 5 minutes at which time 1 ,1'-bis(diphenylphosphino)ferrocene palladium dichloride (0.018 g, 0.025 mmol) was added and the mixture heated to 80 0 C under nitrogen atmosphere overnight. The mixture was removed from heat and cooled to room temperature. The mixture was partitioned between brine and ethyl acetate, the layers were separated and the aqueous phase extracted with ethyl acetate. The combined ethyl acetate extracts were washed four times with brine, dried over anhydrous magnesium sulfate and concentrated under reduced pressure.
- Step 1 ((S)-2- ⁇ [1-(4-fluorobenzyl)-1H-indazole-3-carbonyl]-amino ⁇ -3,3- dimethylbutyryl-amino)acetic acid benzyl ester
- This compound was prepared following the procedure of Johnson, B. L.; Rodgers, J. D. Syn. Comm. 2005, 35, 2681-2684.
- a suspension of 5.28 g 7-fluoroisatin in 30 ml_ of water was added 1.30 g NaOH, in 10 mL water with stirring.
- the resulting dark red solution was stirred until all of the solids dissolved and was then cooled in an ice water bath.
- the solution was then slowly added a cooled (ice bath) solution of 2.21 g NaNO 2 in 10 mL water.
- These combined solutions were then added slowly to cooled (ice bath) to solution of aqueous sulfuric acid (3.4 mL H 2 SO 4 in 60 mL water). Ice was added to maintain a temperature of approximately O 0 C.
- Step 4 1 -(4-cyanobenzyl)-7-f Iuoro-N-[(1 S)-1 ⁇ [(2-hydroxyethyl)amino]carbonyl ⁇ - 2,2-dimethylpropyl]-1H-indazole-3-carboxamide
- the reaction was quenched with water and the biphasic solution was filtered through a phase separator tube.
- the resulting organic solution was concentrated to provide the crude product as an oil.
- the crude material was purified using chromatography over silica gel (heptane/ethyl acetate) to provide N- ⁇ (ISJ-i- ⁇ aminocarbonylJ-I .S ⁇ -oxadiazol ⁇ -yllmethyllaminoJcarbonyl] ⁇ - dimethylpropyl ⁇ -1-(4-fluoro-benzyl)-1H-indazole-3-carboxamide as a colorless oil (95 mg, 25% yield).
- Step 4 N-[(1S)-1-(aminocarbonyl)-2,2-dimethylpropyl]-1-benzyl-1H-pyrazolo[3,4- b]pyridine-3-carboxamide
- Step 1 Benzyl [(1S)-1-(aminocarbonyl)-2,2-dimethylpropyl]carbamate o
- Step 1 [(S)-I -(Carbamoylmethylcarbamoyl)-2,2-dimethylpropyl]carbamic acid tert-butyl ester
- N-Boc-L-tert-leucine 1.0 g, 4.327 mmol
- N 1 N- diisopropylethyl amine 5.1 ml, 30.3 mmol
- EDCHCI 5.1 ml, 30.3 mmol
- HOBT 880 mg, 6.5 mmol
- Glycinamide hydrochloride 720 mg, 6.5 mmol was then added to it and stirring was continued for 18 h at rt.
- Step 1 ((S)-2-tert-Butoxycarbonylamino-3,3-dimethylbutyrylamino)acetic acid benzyl ester:
- N-Boc-L-tert-leucine 1.5 g, 6.48 mmol
- dry DMF 40 ml_
- N 1 N- diisopropylethylamine 8.0 mL, 45.34 mmol
- EDCHCI 1.89 g, 9.89 mmol
- HOBt 1.34 g, 9.89 mmol
- glycine benzyl ester 3.33 g, 9.89 mmol
- Step 2 ((S)-2-Amino-3,3-dimethylbutyrylamino)acetic acid benzyl ester hydrochloride
- Step 1 (S)-5-((2-(tert-butoxycarbonylamino)-3,3-dimethylbutanamido)methyl)- 1 ,3,4-oxadiazole-2-carboxylic acid ethyl ester
- Step 1 ((S)-I -Hydrazinocarbonyl ⁇ -dimethylpropyljcarbamic acid tert-butyl ester
- N-Boc-L-tert-leucine 2.0 g, 8.647 mmol
- dry THF 20 mL
- N 1 N- carbonyl diimidazole CDI
- Step 3 5-((S)-1 -Amino-2,2-dimethylpropyl)-[1 ,3,4]oxadiazol-2-ylamine dihydrochloride
- Step 1 tert-butyl [(1S)-1- ⁇ 5-[(cyclopropylcarbonyl)amino]-1 l 3,4-oxadiazol-2-yl ⁇ - 2,2-dimethylpropyl]carbamate
- Step 2 N- ⁇ 5-[(1S)-1-amino-2,2-dimethylpropyl]-1,3,4-oxadiazol-2- yl ⁇ cyclopropane-carboxamide hydrochloride
- Step 1 tert-Butyl [(1S)-1- ⁇ 5-[(aminocarbonyl)amino]-1,3,4-oxadiazol-2-yl ⁇ -2,2- dimethylpropyl]carbamate
- Step 1 [N'-((S)-2-tert-Butoxycarbonylamino-3,3-dimethyl-butyryl)-hydrazino]- oxo-acetic acid ethyl ester
- Step 1 Benzyl [(1S)-1-cyano-2,2-dimethylpropyl]carbamate
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims
Priority Applications (13)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/918,914 US20110028447A1 (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
| JP2010548208A JP2011513295A (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
| EA201001094A EA201001094A1 (en) | 2008-02-29 | 2009-02-26 | Derivatives indazols |
| MX2010009462A MX2010009462A (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives. |
| AU2009219800A AU2009219800A1 (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
| BRPI0907963-7A BRPI0907963A2 (en) | 2008-02-29 | 2009-02-26 | Indazole Derivatives |
| CN2009801070761A CN101977655A (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
| EP09713687A EP2265335A1 (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
| CA2714573A CA2714573A1 (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
| AP2010005345A AP2010005345A0 (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives. |
| IL207225A IL207225A0 (en) | 2008-02-29 | 2010-07-26 | Indazole derivatives |
| TNP2010000387A TN2010000387A1 (en) | 2009-02-26 | 2010-08-20 | INDAZOLE DERIVATIVES |
| MA33122A MA32108B1 (en) | 2008-02-29 | 2010-08-26 | Indendazole derivatives |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US3265708P | 2008-02-29 | 2008-02-29 | |
| US61/032,657 | 2008-02-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009106982A1 true WO2009106982A1 (en) | 2009-09-03 |
Family
ID=40670272
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2009/000432 Ceased WO2009106982A1 (en) | 2008-02-29 | 2009-02-26 | Indazole derivatives |
Country Status (18)
| Country | Link |
|---|---|
| US (1) | US20110028447A1 (en) |
| EP (1) | EP2265335A1 (en) |
| JP (1) | JP2011513295A (en) |
| KR (1) | KR20100126706A (en) |
| CN (1) | CN101977655A (en) |
| AP (1) | AP2010005345A0 (en) |
| AU (1) | AU2009219800A1 (en) |
| BR (1) | BRPI0907963A2 (en) |
| CA (1) | CA2714573A1 (en) |
| CO (1) | CO6331308A2 (en) |
| CR (1) | CR11600A (en) |
| DO (1) | DOP2010000232A (en) |
| EA (1) | EA201001094A1 (en) |
| EC (1) | ECSP10010428A (en) |
| IL (1) | IL207225A0 (en) |
| MA (1) | MA32108B1 (en) |
| MX (1) | MX2010009462A (en) |
| WO (1) | WO2009106982A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3564214A1 (en) * | 2018-05-04 | 2019-11-06 | Universita' Degli Studi G. D Annunzio Chieti - Pescara | Indazole derivatives as modulators of the cannabinoid system |
| WO2021155227A1 (en) * | 2020-01-30 | 2021-08-05 | Rti International, Inc. | Indazole derivatives as cannabanoid receptor partial agonists |
| US11084789B2 (en) | 2016-01-14 | 2021-08-10 | Beth Israel Deaconess Medical Center, Inc. | Mast-cell modulators and uses thereof |
| EP4213839A4 (en) * | 2020-09-17 | 2024-08-07 | Escient Pharmaceuticals, Inc. | Modulators of mas-related g-protein receptor x4 and related products and methods |
| WO2025172494A1 (en) | 2024-02-16 | 2025-08-21 | F. Hoffmann-La Roche Ag | Novel reversible fluorescent probes for cb1 |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BRPI0910034B1 (en) * | 2008-03-25 | 2022-02-08 | Affectis Pharmaceuticals Ag | P2X7R ANTAGONISTS AND PHARMACEUTICAL COMPOSITIONS THAT COMPRISE THEM |
| UY31984A (en) * | 2008-07-16 | 2010-02-26 | Boehringer Ingelheim Int | DERIVATIVES OF N-substituted 1- (3,4-difluorobenzyl) -6-oxo-1,6-dihydropyrimidin-5-carboxamides and 2- (3,4-difluorobenzyl) -3-oxo-2,3-dihydro- N-substituted 1H-pyrazol-4-carboxamides. |
| AU2011296078B2 (en) * | 2010-09-01 | 2015-06-18 | Ambit Biosciences Corporation | An optically active pyrazolylaminoquinazoline, and pharmaceutical compositions and methods of use thereof |
| CN106535890A (en) | 2014-03-20 | 2017-03-22 | 萨穆梅德有限公司 | 5-substituted indazole-3-carboxamides and preparation and use thereof |
| US10449133B1 (en) | 2018-08-23 | 2019-10-22 | L'oreal | Cosmetic compositions comprising acetyl trifluoromethylphenyl valylglycine |
| KR102048050B1 (en) * | 2019-08-29 | 2020-01-22 | 대한민국 | Synthetic method of metabolites of adb-fubinaca |
| CN117986183A (en) * | 2022-10-28 | 2024-05-07 | 浙江友宁生物医药科技有限公司 | A GPR139 receptor agonist, preparation method and application thereof |
| WO2024108147A1 (en) * | 2022-11-17 | 2024-05-23 | Denali Therapeutics Inc. | Compounds, compositions, and methods |
| WO2026020111A1 (en) * | 2024-07-18 | 2026-01-22 | Washington University | Compositions of novel cannabinoid receptor modulators and methods of use thereof |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0558923A1 (en) * | 1992-01-31 | 1993-09-08 | Nisshin Flour Milling Co., Ltd. | Diazabicyclo derivatives as 5-HT3 antagonists |
| WO2001058869A2 (en) * | 2000-02-11 | 2001-08-16 | Bristol-Myers Squibb Company | Cannabinoid receptor modulators, their processes of preparation, and use of cannabinoid receptor modulators in treating respiratory and non-respiratory diseases |
| WO2003035005A2 (en) * | 2001-10-26 | 2003-05-01 | University Of Connecticut | Heteroindanes: a new class of potent cannabimimetic ligands |
| WO2005092890A2 (en) * | 2004-03-25 | 2005-10-06 | Memory Pharmaceuticals Corporation | Indazoles, benzothiazoles, benzoisothiazoles, benzisoxazoles, and preparation and uses thereof |
| WO2006015263A2 (en) * | 2004-07-29 | 2006-02-09 | Threshold Pharmaceuticals, Inc. | Lonidamine analogs |
| WO2006101456A1 (en) * | 2005-03-21 | 2006-09-28 | S*Bio Pte Ltd | Bicyclic heterocycles hydroxamate compounds useful as histone deacetylase (hdac) inhibitors |
| WO2008052898A1 (en) * | 2006-11-01 | 2008-05-08 | F. Hoffmann-La Roche Ag | Indazole derivatives useful as l-cpt1 inhibitors |
-
2009
- 2009-02-26 MX MX2010009462A patent/MX2010009462A/en not_active Application Discontinuation
- 2009-02-26 CN CN2009801070761A patent/CN101977655A/en active Pending
- 2009-02-26 EA EA201001094A patent/EA201001094A1/en unknown
- 2009-02-26 AU AU2009219800A patent/AU2009219800A1/en not_active Abandoned
- 2009-02-26 BR BRPI0907963-7A patent/BRPI0907963A2/en not_active IP Right Cessation
- 2009-02-26 EP EP09713687A patent/EP2265335A1/en not_active Withdrawn
- 2009-02-26 KR KR1020107018891A patent/KR20100126706A/en not_active Ceased
- 2009-02-26 US US12/918,914 patent/US20110028447A1/en not_active Abandoned
- 2009-02-26 WO PCT/IB2009/000432 patent/WO2009106982A1/en not_active Ceased
- 2009-02-26 JP JP2010548208A patent/JP2011513295A/en not_active Withdrawn
- 2009-02-26 CA CA2714573A patent/CA2714573A1/en not_active Abandoned
- 2009-02-26 AP AP2010005345A patent/AP2010005345A0/en unknown
-
2010
- 2010-07-26 IL IL207225A patent/IL207225A0/en unknown
- 2010-07-27 CR CR11600A patent/CR11600A/en not_active Application Discontinuation
- 2010-07-29 DO DO2010000232A patent/DOP2010000232A/en unknown
- 2010-08-13 CO CO10099995A patent/CO6331308A2/en not_active Application Discontinuation
- 2010-08-26 MA MA33122A patent/MA32108B1/en unknown
- 2010-08-27 EC EC2010010428A patent/ECSP10010428A/en unknown
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0558923A1 (en) * | 1992-01-31 | 1993-09-08 | Nisshin Flour Milling Co., Ltd. | Diazabicyclo derivatives as 5-HT3 antagonists |
| WO2001058869A2 (en) * | 2000-02-11 | 2001-08-16 | Bristol-Myers Squibb Company | Cannabinoid receptor modulators, their processes of preparation, and use of cannabinoid receptor modulators in treating respiratory and non-respiratory diseases |
| WO2003035005A2 (en) * | 2001-10-26 | 2003-05-01 | University Of Connecticut | Heteroindanes: a new class of potent cannabimimetic ligands |
| WO2005092890A2 (en) * | 2004-03-25 | 2005-10-06 | Memory Pharmaceuticals Corporation | Indazoles, benzothiazoles, benzoisothiazoles, benzisoxazoles, and preparation and uses thereof |
| WO2006015263A2 (en) * | 2004-07-29 | 2006-02-09 | Threshold Pharmaceuticals, Inc. | Lonidamine analogs |
| WO2006101456A1 (en) * | 2005-03-21 | 2006-09-28 | S*Bio Pte Ltd | Bicyclic heterocycles hydroxamate compounds useful as histone deacetylase (hdac) inhibitors |
| WO2008052898A1 (en) * | 2006-11-01 | 2008-05-08 | F. Hoffmann-La Roche Ag | Indazole derivatives useful as l-cpt1 inhibitors |
Non-Patent Citations (1)
| Title |
|---|
| SHIM J-Y ET AL: "Molecular interaction of the antagonist N-(piperidin-1-yl)-5-(4-chlor ophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide with the CB1 cannabinoid receptor", JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY, WASHINGTON, US, vol. 45, no. 7, 1 March 2002 (2002-03-01), pages 1447 - 1459, XP002968557, ISSN: 0022-2623 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11084789B2 (en) | 2016-01-14 | 2021-08-10 | Beth Israel Deaconess Medical Center, Inc. | Mast-cell modulators and uses thereof |
| EP3564214A1 (en) * | 2018-05-04 | 2019-11-06 | Universita' Degli Studi G. D Annunzio Chieti - Pescara | Indazole derivatives as modulators of the cannabinoid system |
| WO2021155227A1 (en) * | 2020-01-30 | 2021-08-05 | Rti International, Inc. | Indazole derivatives as cannabanoid receptor partial agonists |
| EP4097088A4 (en) * | 2020-01-30 | 2024-01-24 | Research Triangle Institute | INDAZOL DERIVATIVES AS PARTIAL CANNABANOID RECEPTOR AGONISTS |
| US12577207B2 (en) | 2020-01-30 | 2026-03-17 | Research Triangle Institute | Indazole derivatives as cannabinoid receptor partial agonists |
| EP4213839A4 (en) * | 2020-09-17 | 2024-08-07 | Escient Pharmaceuticals, Inc. | Modulators of mas-related g-protein receptor x4 and related products and methods |
| WO2025172494A1 (en) | 2024-02-16 | 2025-08-21 | F. Hoffmann-La Roche Ag | Novel reversible fluorescent probes for cb1 |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2009219800A1 (en) | 2009-09-03 |
| KR20100126706A (en) | 2010-12-02 |
| AP2010005345A0 (en) | 2010-08-31 |
| MX2010009462A (en) | 2010-09-24 |
| MA32108B1 (en) | 2011-02-01 |
| CA2714573A1 (en) | 2009-09-03 |
| CR11600A (en) | 2010-09-03 |
| DOP2010000232A (en) | 2010-08-15 |
| CN101977655A (en) | 2011-02-16 |
| JP2011513295A (en) | 2011-04-28 |
| CO6331308A2 (en) | 2011-10-20 |
| US20110028447A1 (en) | 2011-02-03 |
| BRPI0907963A2 (en) | 2015-08-04 |
| IL207225A0 (en) | 2010-12-30 |
| ECSP10010428A (en) | 2010-09-30 |
| EP2265335A1 (en) | 2010-12-29 |
| EA201001094A1 (en) | 2011-04-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2009106980A2 (en) | Indazole derivatives | |
| EP2265335A1 (en) | Indazole derivatives | |
| JP5735526B2 (en) | Tricyclic pyrazoleamine derivatives | |
| JP5997265B2 (en) | Pyridine-2-amide useful as a CB2 agonist | |
| JP5449540B2 (en) | New compounds | |
| US9187464B2 (en) | TRPV4 antagonists | |
| JP5451633B2 (en) | Triazole oxadiazole derivatives | |
| JP5837482B2 (en) | Pyrazole oxadiazole derivative | |
| JP2005523310A (en) | Substituted indole | |
| EP0977751A1 (en) | Somatostatin agonists | |
| JP2008525408A (en) | 1,6-Substituted (3R, 6R) -3- (2,3-dihydro-1H-inden-2-yl) -2 as an oxytocin receptor antagonist for the treatment of preterm birth, dysmenorrhea and endometriosis , 5-Piperazinedione derivatives | |
| CN1451007A (en) | Adenosine A2a receptor antagonist | |
| JP2002504929A (en) | Aroylpiperazine derivatives, their preparation and their use as tachykinin antagonists | |
| EP2617715A1 (en) | Glycine transporter inhibitor | |
| HUP0203496A2 (en) | Novel cyclopropane derivatives, pharmaceutical compositions containing them and method for the production thereof | |
| CA2617654A1 (en) | Piperidinoyl-pyrrolidine and piperidinoyl-piperidine compounds | |
| JP2013512952A (en) | Heterocyclic compounds containing indole cores | |
| WO2012153729A1 (en) | Heteroaromatic ring derivative | |
| US20040162278A1 (en) | Triazole compounds useful in therapy | |
| JP2008540500A (en) | Quinoline derivatives as neurokinin receptor antagonists | |
| WO2011091410A1 (en) | Trpv4 antagonists | |
| WO2007109615A2 (en) | β -LACTAMYL VASOPRESSIN V2 ANTAGONISTS | |
| EP3024825A1 (en) | Imidazolecarboxamides and their use as faah inhibitors | |
| JP2014111586A (en) | Pharmaceutical containing heteroaromatic ring derivative | |
| AU2020338971B2 (en) | Substituted 1,3-phenyl heteroaryl derivatives and their use in the treatment of disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980107076.1 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09713687 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009219800 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 207225 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 587018 Country of ref document: NZ Ref document number: 201011600 Country of ref document: CR Ref document number: CR2010-011600 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201001094 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 2714573 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2009219800 Country of ref document: AU Date of ref document: 20090226 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10099995 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12010501863 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PI 2010003491 Country of ref document: MY |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12918914 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20107018891 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: D2010173 Country of ref document: CU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010548208 Country of ref document: JP Ref document number: 11932 Country of ref document: GE Ref document number: MX/A/2010/009462 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP2010000531 Country of ref document: DZ |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009713687 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 6311/DELNP/2010 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: A201010441 Country of ref document: UA |
|
| ENP | Entry into the national phase |
Ref document number: PI0907963 Country of ref document: BR Kind code of ref document: A2 Effective date: 20100830 |