WO2009086129A1 - Pyrazolo [1,5-a] pyrimidine compounds - Google Patents
Pyrazolo [1,5-a] pyrimidine compounds Download PDFInfo
- Publication number
- WO2009086129A1 WO2009086129A1 PCT/US2008/087717 US2008087717W WO2009086129A1 WO 2009086129 A1 WO2009086129 A1 WO 2009086129A1 US 2008087717 W US2008087717 W US 2008087717W WO 2009086129 A1 WO2009086129 A1 WO 2009086129A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- optionally substituted
- hydrogen
- alkyl
- atoms
- Prior art date
Links
- 0 *c1c2nc(*)c(*)c(*)[n]2nc1* Chemical compound *c1c2nc(*)c(*)c(*)[n]2nc1* 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/16—Emollients or protectives, e.g. against radiation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/18—Antioxidants, e.g. antiradicals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/04—Drugs for skeletal disorders for non-specific disorders of the connective tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Definitions
- This invention relates generally to pyrazolo [1,5-a] pyrimidine-based modulators of Liver X receptors (LXRs) and related methods.
- Atherosclerosis is among the leading causes of death in developed countries. Some of the independent risk factors associated with atherosclerosis include the presence of relatively high levels of serum LDL cholesterol and relatively low levels of serum HDL cholesterol in affected patients. As such, some anti-atherosclerotic therapy regimens include the administration of agents (e.g., statins) to reduce elevated serum LDL cholesterol levels.
- agents e.g., statins
- HDL cholesterol is believed to play a major role in the transport of cholesterol from peripheral tissues to the liver for metabolism and excretion (this process is sometimes referred to as "reverse cholesterol transport").
- ABCAl is a transporter gene involved in HDL production and reverse cholesterol transport. Upregulation of ABCAl can therefore result in increased reverse cholesterol transport as well as inhibition of cholesterol absorption in the gut.
- HDL is also believed to inhibit the oxidation of LDL cholesterol, reduce the inflammatory response of endothelial cells, inhibit the coagulation pathway, and promote the availability of nitric oxide.
- LXRs Liver X receptors
- LXRs are members of the nuclear hormone receptor super family and are believed to be involved in the regulation of cholesterol and lipid metabolism.
- LXRs are ligand- activated transcription factors and bind to DNA as obligate heterodimers with retinoid X receptors. While LXR ⁇ is generally found in tissues such as liver, kidney, adipose tissue, intestine and macrophages, LXR ⁇ displays a ubiquitous tissue distribution pattern.
- Activation of LXRs by oxysterols (endogenous ligands) in macrophages results in the expression of several genes involved in lipid metabolism and reverse cholesterol transport including the aforementioned ABCAl; ABCGl; and ApoE. See, e.g., Koldamova, et al, J. Biol. Chem. 2003, 278, 13244.
- LXR ⁇ knock-out mice Studies have been conducted in LXR ⁇ knock-out (k/o), LXR ⁇ k/o and double k/o mice to determine the physiological role of LXRs in lipid homeostasis and atherosclerosis.
- the increased cholesterol accumulation was believed to be associated with the presence of reduced serum HDL cholesterol and increased LDL cholesterol, even though the total cholesterol levels in the mice were about normal.
- LXR ⁇ k/o mice did not appear to show significant changes in hepatic gene expression, LXR ⁇ k/o mice showed 58% decrease in hepatic ABCAl expression and 208% increase in SREBPIc expression suggesting that LXR ⁇ may be involved in the regulation of liver SREBPIc expression.
- LXRs activation of LXRs results in the inhibition of inflammation and proinflammatory gene expression. This hypothesis is based on data obtained from studies employing three different models of inflammation (LPS-induced sepsis, acute contact dermatitis of the ear and chronic atherosclerotic inflammation of the artery wall). These data suggest that LXR modulators can mediate both the removal of cholesterol from the macrophages and the inhibition of vascular inflammation.
- This invention relates generally to pyrazolo [1,5-a] pyrimidine-based modulators of Liver X receptors (LXRs) and related methods.
- this invention features a compound having formula (I):
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-10 R a ;
- R 2 is C 6 -Ci O aryl or heteroaryl including 5-10 atoms, each of which is:
- R .6 is WA, wherein: W at each occurrence is, independently, a bond; -O-; -NR 7 - wherein R 7 is hydrogen or Ci-C 6 alkyl; Ci_6 alkylene, C2-6 alkenylene, or C2-6 alkynylene; -W ⁇ C 1-6 alkylene)-; or -(C 1-6 alkylene)W 1 -;
- W 1 at each occurrence is, independently, -O- or -NR 7 -;
- a at each occurrence is, independently, C 6 -CiO aryl or heteroaryl including 5-10 atoms, each of which is:
- R 8 at each occurrence is, independently:
- W 2 at each occurrence is, independently, a bond; Ci_ 6 alkylene; C 2 - 6 alkenylene; C2-6 alkynylene; C3-6 cycloalkylene; -0(C 1-6 alkylene)-, or -NR 7 (Ci_ 6 alkylene)-;
- n at each occurrence is, independently, 1 or 2;
- R 9 at each occurrence is, independently:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-5 R a ; or
- C 2 -C 6 alkenyl or C 2 -C 6 alkynyl each of which is optionally substituted with from 1-5 R b ; or
- R 10 and R 11 are each, independently, hydrogen; R 9 ; or heterocyclyl including 3-10 atoms or a heterocycloalkenyl including 3-10 atoms, each of which is optionally substituted with from 1-5 R c ; or
- R 10 and R 11 together with the nitrogen atom to which they are attached form a heterocyclyl including 3-10 atoms or a heterocycloalkenyl including 3-10 atoms, each of which is optionally substituted with from 1-5 R c ;
- R 12 at each occurrence is, independently, hydrogen or R 9 ;
- one of R 13 and R 14 is hydrogen or C1-C3 alkyl; and the other of R 13 and R 14 is: (i) -S(O) n R 9 ; or (ii) -C(O)OR 12 ; or (iii) -C(O)NR 10 R 11 ; or
- each of R 3 and R 4 is, independently: (i) hydrogen; or (ii) halo; or
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-3 R a ;
- R a at each occurrence is, independently:
- R b at each occurrence is, independently:
- halo NR m R n ; hydroxy; Ci-C 6 alkoxy or Ci-C 6 haloalkoxy; C 6 -CiO aryloxy or heteroaryloxy including 5-10 atoms, each of which is optionally substituted with from 1- 5 R d ; C 7 -C 11 aralkoxy, heteroaralkoxy including 6-11 atoms, C3-C10 cycloalkoxy, C3-C10 cycloalkenyloxy, heterocyclyloxy including 3-10 atoms, or heterocycloalkenyloxy including 3-10 atoms, each of which is optionally substituted with from 1-5 R c ; cyano; or
- R c at each occurrence is, independently: (i) halo; NR m R n ; hydroxy; Ci-C 6 alkoxy or Ci-C 6 haloalkoxy; cyano; or
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-5 R a ;
- R d at each occurrence is, independently:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -5 R a ;
- R e at each occurrence is, independently, Ci-C 6 alkyl; Ci-C 6 haloalkyl; halo; hydroxyl; NR m R n ; Ci-C 6 alkoxy; Ci-C 6 haloalkoxy; or cyano;
- R g at each occurrence is, independently:
- R h at each occurrence is, independently, hydroxyl, Ci-C 6 alkoxy, or Ci-C 6 haloalkoxy; C3-C10 cycloalkoxy or C3-C10 cycloalkenyloxy, each of which is optionally substituted with from 1-5 R c ; or C 6 -Ci O aryloxy or heteroaryloxy including 5-10 atoms, each of which is optionally substituted with from 1-5 R d ;
- each of R m and R n at each occurrence is, independently, hydrogen; Ci-C 6 alkyl; or Ci-C 6 haloalkyl;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 at each occurrence is, independently:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and R 8 is:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 is:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 is:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 is:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 is:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 at each occurrence is, independently: (v) C1-C12 alkyl or C1-C12 haloalkyl, each of which is:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 is:
- R 5 is:
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 at each occurrence is, independently:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 at each occurrence is, independently:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention features a compound having formula (I), in which R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 9 , R 10 , R 11 , R 12 , R 13 , R 14 , W, W 1 , W 2 , A, R a , R b , R c , R d , R e , R g , R h , R m , R n , and n, can each be, independently, as defined anywhere herein, and
- R 8 at each occurrence is, independently:
- R 5 is:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- this invention relates to any subgenera of formula (I) described herein.
- this invention relates to any of the specific pyrazolo [1,5 -a] pyrimidine compounds delineated herein.
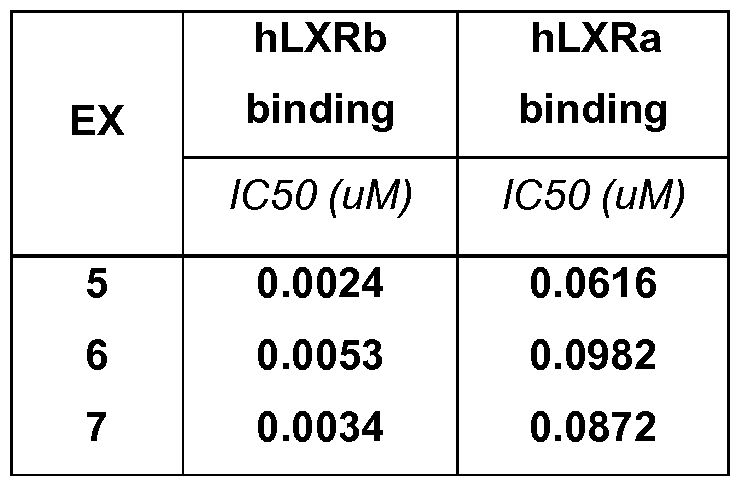
- the compound of formula (I) can be selected from the title compounds of Examples 5-7; or a pharmaceutically acceptable salt and/or N-oxide thereof.
- this invention features a composition (e.g., a pharmaceutical composition), which includes a compound of formula (I) (including any subgenera or specific compounds thereof) or a salt (e.g., a pharmaceutically acceptable salt) or a prodrug thereof and a pharmaceutically acceptable adjuvant, carrier or diluent.
- a composition e.g., a pharmaceutical composition
- the composition can include an effective amount of the compound or the salt thereof.
- the composition can further include an additional therapeutic agent.
- this invention features a dosage form, which includes from about 0.05 milligrams to about 2,000 milligrams (e.g., from about 0.1 milligrams to about 1,000 milligrams, from about 0.1 milligrams to about 500 milligrams, from about 0.1 milligrams to about 250 milligrams, from about 0.1 milligrams to about 100 milligrams, from about 0.1 milligrams to about 50 milligrams, or from about 0.1 milligrams to about 25 milligrams) of formula (I) (including any subgenera or specific compounds thereof), or a salt (e.g., a pharmaceutically acceptable salt), or an N-oxide, or a prodrug thereof.
- the dosage form can further include a pharmaceutically acceptable carrier and/or an additional therapeutic agent.
- the invention also relates generally to modulating (e.g., activating) LXRs with the pyrazolo [1,5 -a] pyrimidine compounds described herein.
- the methods can include, e.g., contacting an LXR in a sample (e.g., a tissue, a cell free assay medium, a cell-based assay medium) with a compound of formula (I) (including any subgenera or specific compounds thereof).
- the methods can include administering a compound of formula (I) (including any subgenera or specific compounds thereof) to a subject (e.g., a mammal, e.g., a human, e.g., a human having or at risk of having one or more of the diseases or disorders described herein).
- a subject e.g., a mammal, e.g., a human, e.g., a human having or at risk of having one or more of the diseases or disorders described herein.
- this invention also relates generally to methods of treating (e.g., controlling, ameliorating, alleviating, slowing the progression of, delaying the onset of, or reducing the risk of developing) or preventing one or more LXR-mediated diseases or disorders in a subject (e.g., a subject in need thereof).
- the methods include administering to the subject an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- LXR-mediated diseases or disorders can include, e.g., cardiovascular diseases (e.g., acute coronary syndrome, restenosis), atherosclerosis, atherosclerotic lesions, type I diabetes, type II diabetes, Syndrome X, obesity, lipid disorders (e.g., dyslipidemia, hyperlipidemia, hypertriglyceridemia, hypercholesterolemia, low HDL and high LDL), cognitive disorders (e.g., Alzheimer's disease or dementia), inflammatory diseases (e.g., multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease, Crohn's disease, endometriosis, LPS-induced sepsis, acute contact dermatitis of the ear, chronic atherosclerotic inflammation of the artery wall), celiac, thyroiditis, skin aging or connective tissue diseases.
- cardiovascular diseases e.g., acute coronary syndrome, restenosis
- atherosclerosis e.g., atherosclerosis, atherosclerotic lesions
- this invention relates to methods of modulating (e.g., increasing) serum HDL cholesterol levels in a subject (e.g., a subject in need thereof), which includes administering to the subject an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of modulating (e.g., decreasing) serum LDL cholesterol levels in a subject (e.g., a subject in need thereof), which includes administering to the subject an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of modulating (e.g., increasing) reverse cholesterol transport in a subject (e.g., a subject in need thereof), which includes administering to the subject an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of modulating (e.g., decreasing or inhibiting) cholesterol absorption in a subject (e.g., a subject in need thereof), which includes administering to the subject an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating a cardiovascular disease (e.g., acute coronary syndrome, restenosis, or coronary artery disease), which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- a cardiovascular disease e.g., acute coronary syndrome, restenosis, or coronary artery disease
- this invention relates to methods of preventing or treating a atherosclerosis and/or atherosclerotic lesions, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating diabetes (e.g., type I diabetes or type II diabetes), which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- diabetes e.g., type I diabetes or type II diabetes
- administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating Syndrome X, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating obesity, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating a lipid disorder (e.g., dyslipidemia, hyperlipidemia, hypertriglyceridemia, hypercholesterolemia, low HDL and/or high LDL), which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- a lipid disorder e.g., dyslipidemia, hyperlipidemia, hypertriglyceridemia, hypercholesterolemia, low HDL and/or high LDL
- this invention relates to methods of preventing or treating a cognitive disorder (e.g., Alzheimer's disease or dementia), which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- a cognitive disorder e.g., Alzheimer's disease or dementia
- administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating dementia, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating Alzheimer's disease, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating an inflammatory disease (e.g., multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease, Crohn's disease, endometriosis, LPS-induced sepsis, acute contact dermatitis of the ear, chronic atherosclerotic inflammation of the artery wall), which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- an inflammatory disease e.g., multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease, Crohn's disease, endometriosis, LPS-induced sepsis, acute contact dermatitis of the ear, chronic atherosclerotic inflammation of the artery wall
- this invention relates to methods of preventing or treating rheumatoid arthritis, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating celiac, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of preventing or treating thyroiditis, which includes administering to a subject in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- this invention relates to methods of treating a connective tissue disease (e.g., osteoarthritis or tendonitis), which includes administering to a subject (e.g., a mammal, e.g., a human) in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- a connective tissue disease e.g., osteoarthritis or tendonitis
- the compound of formula (I) inhibits (e.g., reduces or otherwise diminishes) cartilage degradation. In embodiments, the compound of formula (I) induces (e.g., increases or otherwise agments) cartilage regeneration. In embodiments, the compound of formula (I) inhibits (e.g., reduces or otherwise diminishes) cartilage degradation and induces (e.g., increases or otherwise agments) cartilage regeneration. In embodiments, the compound of formula (I) inhibits (e.g., reduces or otherwise diminishes) aggrecanase activity. In embodiments, the compound of formula (I) inhibits (e.g., reduces or otherwise diminishes) elaboration of pro-inflammatory cytokines in osteoarthritic lesions.
- this invention relates to methods of treating or preventing skin aging, the method comprising administering (e.g., topically administering) to a subject (e.g., a mammal, e.g., a human) in need thereof an effective amount of a compound of formula (I) (including any subgenera or specific compounds thereof) or a pharmaceutically acceptable salt or prodrug thereof.
- a subject e.g., a mammal, e.g., a human
- an effective amount of a compound of formula (I) including any subgenera or specific compounds thereof
- a pharmaceutically acceptable salt or prodrug thereof e.g., a pharmaceutically acceptable salt or prodrug thereof.
- the skin aging can be derived from chronological aging, photoaging, steroid-induced skin thinning, or a combination thereof.
- skin aging includes conditions derived from intrinsic chronological aging (for example, deepened expression lines, reduction of skin thickness, inelasticity, and/or unblemished smooth surface), those derived from photoaging (for example, deep wrinkles, yellow and leathery surface, hardening of the skin, elastosis, roughness, dyspigmentations (age spots) and/or blotchy skin), and those derived from steroid- induced skin thinning.
- another aspect is a method of counteracting UV photodamage, which includes contacting a skin cell exposed to UV light with an effective amount of a compound of formula (I).
- the compound of formula (I) (including any subgenera or specific compounds thereof) does not substantially increase serum and/or hepatic triglyceride levels of the subject.
- the administered compound of formula (I) (including any subgenera or specific compounds thereof) can be an LXR agonist (e.g., an LXR ⁇ agonist or an LXR ⁇ agonist, e.g., an LXR ⁇ agonist).
- the subject can be a subject in need thereof (e.g., a subject identified as being in need of such treatment). Identifying a subject in need of such treatment can be in the judgment of a subject or a health care professional and can be subjective (e.g. opinion) or objective (e.g. measurable by a test or diagnostic method).
- the subject can be a mammal. In certain embodiments, the subject is a human.
- this invention also relates to methods of making compounds described herein.
- the method includes taking any one of the intermediate compounds described herein and reacting it with one or more chemical reagents in one or more steps to produce a compound described herein.
- this invention relates to a packaged product.
- the packaged product includes a container, one of the aforementioned compounds in the container, and a legend (e.g., a label or an insert) associated with the container and indicating administration of the compound for treatment and control of the diseases or disorders described herein.
- a legend e.g., a label or an insert
- any compound, composition, or method can also include any one or more of the features (alone or in combination) delineated in the detailed description and/or in the claims.
- R 1 can be hydrogen
- R 1 can be C 1 -C 3 alkyl or C 1 -C 3 haloalkyl (e.g., CF 3 ).
- R 1 can be CH 3 (i.e., methyl), CH 3 CH 2 (i.e., ethyl), or (CH 3 ) 2 CH (i.e., isopropyl).
- R 1 can be C 6 -CiO aryl or heteroaryl including 5-10 atoms, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, or 1) R d .
- R 1 can be phenyl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, or 1) R d .
- R 1 can be C 7 -C 11 aralkyl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, or 1) R c .
- R 1 can be benzyl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, or 1) R c .
- R 1 can be C 3 -Cs cycloalkyl or heterocyclyl including 3-8 atoms, each of which is optionally substituted with from 1-3 R c .
- R 2 can be C 6 -CiO aryl, which is (a) substituted with 1 R 6 ; and (b) optionally substituted with from 1-2 R e .
- R 2 can be phenyl, which is (a) substituted with 1 R 6 ; and (b) optionally substituted with from 1 R e .
- R 2 can be phenyl, which is substituted with 1 R 6 .
- R 2 can have formula (A-2):
- each of R 22 , R 23 , and R 24 can be, independently, hydrogen or R e .
- R e can be as defined anywhere herein.
- each of R , R , and R is hydrogen; or (ii) one of R , R 23 , and R 24 is R e , and the other two are hydrogen.
- each of R 22 , R 23 , and R 24 can be hydrogen.
- one of R 22 , R 23 , and R 24 can be R e , and the other two are hydrogen.
- R 22 can be R e (e.g., halo, e.g., chloro), and each of R 23 and R 24 can be hydrogen.
- W can be -0-.
- W can be a bond.
- W can be -W ⁇ C 1-6 alkylene)-; in embodiments, W 1 can be -O-, and W can be, for example, -OCH 2 -.
- A can be C 6 -CiO aryl, which is (a) substituted with 1 R 8 ; and (b) optionally substituted with from 1-4 R g .
- A can be phenyl, which is (a) substituted with 1 R 8 ; and (b) optionally substituted with from 1-4 R g .
- A can have formula (B-I):
- each of R 8 and R g can be, independently, as defined anywhere herein.
- R 8 can be -W 2 -S(O) n R 9 .
- W 2 can be a bond, n can be 2.
- W 2 can be a bond, and n can be 2.
- R 9 can be Ci-Cio alkyl, optionally substituted with from 1-2 R a .
- R 9 can be Ci-C 5 alkyl (e.g., CH 3 , CH 3 CH 2 , or (CH 3 ) 2 CH), e.g., CH 3 or CH 3 CH 2 ).
- R 9 can be C 2 -C 8 alkyl substituted with 1 R a .
- R a can be hydroxyl or Ci-C 3 alkoxy.
- R 8 can be -W 2 -C(O)OR 12 .
- R 2 can have formula (C-I):
- each of R 22 , R 23 , and R 24 is, independently, hydrogen or R e ; and one of R A2 , R A3 , R A4 , R A5 , and R A6 is R 8 , and the others are each, independently, hydrogen or R g .
- each of R 22 , R 23 , and R 24 is hydrogen;
- R 22 , R 23 , and R 24 is R e , and the other two are hydrogen; and one of R A2 , R A3 , R A4 , R A5 , and R A6 is R 8 , and the others are each, independently, hydrogen or R g .
- each of W, R 8 , R e and R g can be, independently, as defined anywhere herein.
- Embodiments can include, for example, one or more of the following features (and/or any one or more other features described anywhere herein).
- each of R 22 , R 23 , and R 24 can be hydrogen. In other embodiments, one of R 22 , R 23 , and R 24 can be R e , and the other two are hydrogen.
- R 22 can be R e (e.g., halo, e.g., chloro), and each of R 23 and R 24 is hydrogen.
- W can be -O-.
- W can be a bond.
- W can be -OCH 2 -.
- R A3 and R A4 can be R 8 , and the other of R A3 and R A4 can be hydrogen; and each of R ⁇ , R A5 , and R A6 can be, independently, hydrogen or R g .
- R A3 can be -W 2 -S(O) n R 9 .
- Each of R A2 , R A5 , and R A6 can be hydrogen.
- W 2 can be a bond
- n can be 2.
- W 2 can be a bond
- n can be 2.
- R 9 can be Ci-C 6 alkyl, optionally substituted with from 1-2 R a .
- R 9 can be C 1 - C 3 alkyl (e.g., CH 3 , CH 3 CH 2 , or (CH 3 ) 2 CH)).
- R 9 can be C 2 -C 6 alkyl substituted with 1 R a .
- R a can be hydroxyl or Ci-C 3 alkoxy.
- R A5 can be hydrogen or R g
- each of R A2 and R A6 can be hydrogen.
- R A4 can be -W 2 -C(O)OR 12 .
- R 12 can be hydrogen.
- R 12 can be Ci-C 3 alkyl.
- W 2 can be Ci-C 3 alkylene (e.g., CH 2 ).
- W 2 can be a bond.
- R A2 , R A5 , and R A6 can be hydrogen.
- R 3 and R 4 can be, independently: (i) hydrogen; or (ii) halo.
- Each of R 3 and R 4 can be hydrogen.
- R 5 can be: (ii) halo; or (iii) Ci-C 6 alkyl or Ci-C 6 haloalkyl, each of which is optionally substituted with from 1-3 R a ; or (iv) cyano.
- R 5 can be Ci-C 6 haloalkyl.
- R 5 can be C 1 -C 3 perfluoroalkyl (e.g., CF 3 )
- R 5 can be halo (e.g., chloro).
- R 1 , R 3 , R 4 , and R 5 can be a substituent other than hydrogen.
- the compound can have formula (VI):
- each of R 3 and R 4 is, independently: (i) hydrogen; or (ii) halo; or
- R 5 is:
- each of R 22 , R 23 , and R 24 is, independently, hydrogen or R e
- Embodiments can include one or more of the following features (and/or any one or more other features described anywhere herein).
- R 1 can be hydrogen.
- R 1 can be CH 3 , CH 3 CH 2 , or (CH 3 ) 2 CH.
- R 1 can be phenyl or thienyl, each of which is optionally substituted with from 1-5 R d .
- R 1 can be benzyl, which is optionally substituted with from 1-5 R d .
- W can be -O-.
- W can be a bond.
- W can be -OCH 2 -.
- R A can have formula (B-I), in which one of R ⁇ and R A4 is R 8 , and the other of R A3 and R A4 is hydrogen; and each of R A2 , R A5 , and R A6 is, independently, hydrogen or R g .
- R A3 can be -W 2 -S(O) n R 9 , in which W 2 can be a bond, and n can be 2.
- R 9 can be Ci-C 6 alkyl, optionally substituted with from 1-2 R a .
- R 9 can be CH 3 , CH 2 CH 3 , or ⁇ opropyl.
- R 9 can be C 2 -Cs alkyl substituted with 1 R a .
- R a can be hydroxyl or Ci-C 3 alkoxy.
- R A5 can be hydrogen or R c , and each of R ⁇ and R A6 can be hydrogen.
- R A4 can be -W 2 - C(O)OR 12 .
- R 12 can be hydrogen or Ci-C 3 alkyl.
- W 2 can be CH 2 .
- Each of R A2 , R A5 , and R A6 can be hydrogen.
- Each of R 3 and R 4 can be hydrogen.
- Each of R 22 , R 23 , and R 24 can be hydrogen.
- One of R , R , and R can be R , and the other two can be hydrogen.
- R 22 can be R e (e.g., chloro) and each of R 23 and R 24 can be hydrogen.
- R 5 can be CF 3 .
- R 5 can be chloro.
- mammal includes organisms, which include mice, rats, cows, sheep, pigs, rabbits, goats, horses, monkeys, dogs, cats, and humans.
- an effective amount refers to an amount of a compound that confers a therapeutic effect (e.g., treats, controls, ameliorates, alleviates, slows the progression of, prevents, delays the onset of, or reduces the risk of developing a disease, disorder, or condition or symptoms thereof) on the treated subject.
- the therapeutic effect may be objective (i.e., measurable by some test or marker) or subjective (i.e., subject gives an indication of or feels an effect).
- An effective amount of the compound described above may range from about 0.01 mg/Kg to about 1000 mg/Kg, (e.g., from about 0.1 mg/Kg to about 100 mg/Kg, from about 1 mg/Kg to about 100 mg/Kg). Effective doses will also vary depending on route of administration, as well as the possibility of co-usage with other agents.
- halo or halogen refers to any radical of fluorine, chlorine, bromine or iodine.
- substituent (radical) prefix names are derived from the parent hydride by either (i) replacing the "ane” in the parent hydride with the suffixes "yl,” “diyl,” “triyl,” “tetrayl,” etc.; or (ii) replacing the "e” in the parent hydride with the suffixes "yl,” “diyl,” “triyl,” “tetrayl,” etc. (here the atom(s) with the free valence, when specified, is (are) given numbers as low as is consistent with any established numbering of the parent hydride).
- Accepted contracted names e.g., adamantyl, naphthyl, anthryl, phenanthryl, furyl, pyridyl, isoquinolyl, quinolyl, and piperidyl, and trivial names, e.g., vinyl, allyl, phenyl, and thienyl are also used herein throughout.
- Conventional numbering/lettering systems are also adhered to for substituent numbering and the nomenclature of fused, bicyclic, tricyclic, polycyclic rings.
- alkyl refers to a saturated hydrocarbon chain that may be a straight chain or branched chain, containing the indicated number of carbon atoms.
- C1-C20 alkyl indicates that the group may have from 1 to 20 (inclusive) carbon atoms in it. Any atom can be optionally substituted, e.g., by one or more substituents.
- alkyl groups include without limitation methyl, ethyl, n-propyl, ⁇ opropyl, and tert- butyl.
- cycloalkyl refers to saturated monocyclic, bicyclic, tricyclic, or other polycyclic hydrocarbon groups. Any atom can be optionally substituted, e.g., by one or more substituents. A ring carbon serves as the point of attachment of a cycloalkyl group to another moiety. Cycloalkyl groups can contain fused rings. Fused rings are rings that share a common carbon atom. Cycloalkyl moieties can include, e.g., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, adamantyl, and norbornyl (bicycle[2.2.1]heptyl).
- alkylene alkenylene
- alkynylene alkynylene
- haloalkyl refers to an alkyl group, in which at least one hydrogen atom is replaced by halo.
- more than one hydrogen atom (2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26,etc. hydrogen atoms) on a alkyl group can be replaced by more than one halogen (e.g., 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, etc. halogen atoms).
- the hydrogen atoms can each be replaced by the same halogen (e.g., fluoro) or the hydrogen atoms can be replaced by a combination of different halogens (e.g., fluoro and chloro).
- Haloalkyl also includes alkyl moieties in which all hydrogens have been replaced by halo (e.g., perhaloalkyl, e.g., perfluoroalkyl, such as trifluoromethyl). Any atom can be substituted, e.g., by one or more substituents.
- aralkyl refers to an alkyl moiety in which an alkyl hydrogen atom is replaced by an aryl group. One of the carbons of the alkyl moiety serves as the point of attachment of the heteroaralkyl group to another moiety.
- Aralkyl includes groups in which more than one hydrogen atom on an alkyl moiety has been replaced by an aryl group. Any ring or chain atom can be optionally substituted, e.g., by one or more substituents.
- Non-limiting examples of "aralkyl” include benzyl, 2-phenylethyl, 3- phenylpropyl, benzhydryl (diphenylmethyl), and trityl (triphenylmethyl) groups.
- heteroarylkyl refers to an alkyl moiety in which an alkyl hydrogen atom is replaced by a heteroaryl group. One of the carbons of the alkyl moiety serves as the point of attachment of the aralkyl group to another moiety.
- Heteroaralkyl includes groups in which more than one hydrogen atom on an alkyl moiety has been replaced by a heteroaryl group. Any ring or chain atom can be optionally substituted, e.g., by one or more substituents.
- Heteroaralkyl can include, for example, 2-pyridylethyl.
- alkenyl refers to a straight or branched hydrocarbon chain containing 2-20 carbon atoms and having one or more double bonds. Any atom can be optionally substituted, e.g., by one or more substituents. Alkenyl groups can include, e.g., allyl, 1- butenyl, 2-hexenyl and 3-octenyl groups. One of the double bond carbons can optionally be the point of attachment of the alkenyl substituent.
- alkynyl refers to a straight or branched hydrocarbon chain containing 2-20 carbon atoms and having one or more triple bonds. Any atom can be optionally substituted, e.g., by one or more substituents. Alkynyl groups can include, e.g., ethynyl, propargyl, and 3-hexynyl. One of the triple bond carbons can optionally be the point of attachment of the alkynyl substituent.
- alkoxy refers to an -O-alkyl radical.
- mercapto refers to an SH radical.
- thioalkoxy refers to an -S-alkyl radical.
- aryloxy and heteroaryloxy refer to an -O-aryl radical and -O-heteroaryl radical, respectively.
- thioaryloxy and thioheteroaryloxy refer to an -S-aryl radical and -S-heteroaryl radical, respectively.
- aralkoxy and “heteroaralkoxy” refer to an -O-aralkyl radical and -O- heteroaralkyl radical, respectively.
- thioaralkoxy and “thioheteroaralkoxy” refer to an -S-aralkyl radical and -S -heteroaralkyl radical, respectively.
- cycloalkoxy refers to an -O-cycloalkyl radical.
- cycloalkenyloxy and “heterocycloalkenyloxy” refer to an -O-cycloalkenyl radical and -O-heterocycloalkenyl radical, respectively.
- heterocyclyloxy refers to an -O-heterocyclyl radical.
- thiocycloalkoxy refers to an -S-cycloalkyl radical.
- thiocycloalkenyloxy and “thioheterocycloalkenyloxy” refer to an -S-cycloalkenyl radical and -S-heterocycloalkenyl radical, respectively.
- thioheterocyclyloxy refers to an -S-heterocyclyl radical.
- heterocyclyl refers to a saturated monocyclic, bicyclic, tricyclic or other polycyclic ring system having 1-4 heteroatoms if monocyclic, 1-8 heteroatoms if bicyclic, or 1-10 heteroatoms if tricyclic, said heteroatoms selected from O, N, or S (and mono and dioxides thereof, e.g., N ⁇ O " , S(O), SO 2 ).
- a heterocyclyl ring includes carbon atoms and 1-4, 1-8, or 1-10 heteroatoms selected from N, O, or S if monocyclic, bicyclic, or tricyclic, respectively.
- a ring heteroatom or ring carbon is the point of attachment of the heterocyclyl substituent to another moiety.
- heterocyclyl groups can contain fused rings. Fused rings are rings that share a common carbon or nitrogen atom. Heterocyclyl groups can include, e.g., tetrahydrofuryl, tetrahydropyranyl, piperidyl (piperidino), piperazinyl, morpholinyl (morpholino), pyrrolinyl, and pyrrolidinyl.
- cycloalkenyl refers to partially unsaturated monocyclic, bicyclic, tricyclic, or other polycyclic hydrocarbon groups.
- a ring carbon e.g., saturated or unsaturated is the point of attachment of the cycloalkenyl substituent. Any atom can be optionally substituted, e.g., by one or more substituents.
- the cycloalkenyl groups can contain fused rings. Fused rings are rings that share a common carbon atom. Cycloalkenyl moieties can include, e.g., cyclohexenyl, cyclohexadienyl, or norbornenyl.
- heterocycloalkenyl refers to partially unsaturated monocyclic, bicyclic, tricyclic, or other polycyclic hydrocarbon groups having 1-4 heteroatoms if monocyclic, 1-8 heteroatoms if bicyclic, or 1-10 heteroatoms if tricyclic, said heteroatoms selected from O, N, or S (and mono and dioxides thereof, e.g., N ⁇ O " , S(O), SO 2 ) (e.g., carbon atoms and 1-4, 1-8, or 1-10 heteroatoms of N, O, or S if monocyclic, bicyclic, or tricyclic, respectively).
- a ring carbon (e.g., saturated or unsaturated) or heteroatom is the point of attachment of the heterocycloalkenyl substituent. Any atom can be optionally substituted, e.g., by one or more substituents.
- the heterocycloalkenyl groups can contain fused rings. Fused rings are rings that share a common carbon or nitrogen atom.
- Heterocycloalkenyl groups can include, e.g., tetrahydropyridyl, dihydropyranyl, 4,5-dihydrooxazolyl, 4,5-dihydro-lH-imidazolyl, 1,2,5,6-tetrahydro- pyrimidinyl, and 5,6-dihydro-2H-[l,3]oxazinyl.
- aryl refers to a fully unsaturated, aromatic monocyclic, bicyclic, or tricyclic, hydrocarbon ring system, wherein any ring atom can be optionally substituted, e.g., by one or more substituents.
- Aryl groups can contain fused rings. Fused rings are rings that share a common carbon atom.
- Aryl moieties can include, e.g., phenyl, naphthyl, anthracenyl, and pyrenyl.
- heteroaryl refers to a fully unsaturated, aromatic monocyclic, bicyclic, tricyclic, or other poly eye lie hydrocarbon groups having 1-4 heteroatoms if monocyclic, 1-8 heteroatoms if bicyclic, or 1-10 heteroatoms if tricyclic, said heteroatoms independently selected from O, N, or S (and mono and dioxides thereof, e.g., N ⁇ O ⁇ , S(O), SO 2 ) (e.g., carbon atoms and 1-4, 1-8, or 1-10 heteroatoms of N, O, or S if monocyclic, bicyclic, or tricyclic, respectively). Any atom can be optionally substituted, e.g., by one or more substituents.
- Heteroaryl groups can contain fused rings. Fused rings are rings that share a common carbon or nitrogen atom. Heteroaryl groups can include, e.g., pyridyl, thienyl, furyl (furanyl), imidazolyl, indolyl, isoquinolyl, quinolyl and pyrrolyl.
- the descriptor C(O) refers to a carbon atom that is doubly bonded to oxygen.
- substituted refers to a group “substituted” on, e.g., an alkyl, haloalkyl, cycloalkyl, alkenyl, alkynyl, aralkyl, heteroaralkyl, heterocyclyl, heterocycloalkenyl, cycloalkenyl, aryl, or heteroaryl group at any atom of that group.
- the substituent(s) (e.g., R d ) on a group are independently any one single, or any combination of two or more of the permissible atoms or groups of atoms delineated for that substituent.
- a substituent may itself be substituted with any one of the above substituents.
- Descriptors such as "Ci-C 6 alkyl which is optionally substituted with from 1-2 R a " (and the like) is intended to include as alternatives both unsubstituted Ci-C 6 alkyl and Ci-C 6 alkyl that is substituted with from 1-2 R a .
- the use of a substituent (radical) prefix names such as alkyl without the modifier "optionally substituted” or “substituted” is understood to mean that the particular substituent is unsubstituted.
- haloalkyl without the modifier "optionally substituted” or “substituted” is still understood to mean an alkyl group, in which at least one hydrogen atom is replaced by halo.
- the compounds have agonist activity for genes involved with HDL production and cholesterol efflux (e.g., ABCAl) and antagonist activity for genes involved with triglyceride synthesis (e.g., SREBP-Ic).
- This invention relates generally to pyrazolo [1,5-a] pyrimidine-based modulators of Liver X receptors (LXRs) and related methods.
- pyrazolo [1,5-a] pyrimidine-based LXR modulators have the general formula
- R 1 can be: (1-i) hydrogen; or
- Ci-C 6 e.g., C 1 -C 3 alkyl or C 1 -C 6 (e.g., C 1 -C 4 or C 1 -C 3 ) haloalkyl, each of which is optionally substituted with from 1-10 (e.g., 1-5, 1-4, 1-3, 1-2, 1) R a ; or
- C 3 -Ci 0 (e.g., C 3 -C 8 or C 3 -C 6 ) cycloalkyl, C 3 -Ci 0 (e.g., C 3 -C 8 or C 3 -C 6 ) cycloalkenyl, heterocyclyl including 3-10 (e.g., 3-8 or 3-6) atoms, heterocycloalkenyl including 3-10 (e.g., 3-8 or 3-6) atoms, C 7 -C 11 (e.g., C 7 -C 10 ) aralkyl, or heteroaralkyl including 6-11 (e.g., 6-10) atoms, each of which is optionally substituted with from 1-10 (e.g., 1-5, 1-4, 1-3, 1-2, 1) R c ; or
- C 6 -CiO e.g., phenyl
- heteroaryl including 5-10 (e.g., 5-6) atoms, each of which is optionally substituted with from 1-10 (e.g., 1-5, 1-4, 1-3, 1-2, 1) R d .
- R 1 can be:
- Ci-C 6 e.g., C 1 -C 3 alkyl or C 1 -C 6 (e.g., C 1 -C 4 ) haloalkyl, each of which is optionally substituted with from 1-10 (e.g., 1-5, 1-4, 1-3, 1-2, 1) R a ; or
- C 7 -Cn e.g., C 7 -Ci 0
- heteroaralkyl including 6-11 (e.g., 6-10) atoms, each of which is optionally substituted with from 1-10 (e.g., 1-5, 1-4, 1-3, 1-2, 1) R c ; or
- C 6 -Ci 0 e.g., phenyl
- heteroaryl including 5-10 (e.g., 5-6) atoms, each of which is optionally substituted with from 1-10 (e.g., 1-5, 1-4, 1-3, 1-2, 1) R d .
- R 1 can be any one of: (1-i), (1- ⁇ ), (1-iv), (1-iv'), and (1- v). In certain embodiments, R 1 can be hydrogen. In other embodiments, R 1 can be a substituent other than hydrogen.
- R 1 can be any two of: (1-i), (1-ii), (1-iv), (1-iv'), and (1- v). In certain embodiments, R 1 can be hydrogen and any one of (1-ii), (1-iv), (1-iv'), and (1-v). In other embodiments, R 1 can be any two of (1-ii), (1-iv), (1-iv'), and (1-v), e.g., R 1 can be (1-ii) and (1- iv'). In some embodiments, R 1 can be any three of: (1-i), (1- ⁇ ), (1-iv), (1-iv'), and (1- v).
- R 1 can be hydrogen and any two of (1-ii), (1-iv), (1-iv'), and (1-v), e.g., R 1 can be (1-ii) and (1- iv'). In other embodiments, R 1 can be any three of (1- ii), (1-iv), (1-iv'), and (1-v), e.g., (1-ii), (1-iv'), and (1-v).
- R 1 can be Ci-C 6 (e.g., C 1 -C 5 or C 1 -C 3 ) alkyl.
- R 1 can be methyl (CH 3 ), ethyl (CH 2 CH 3 ), or isopropyl (CH(CH 3 ) 2 ).
- R 1 can be Ci-C 6 (e.g., C 1 -C 4 or C 1 -C 3 ) haloalkyl (e.g., perhaloalkyl).
- R 1 can be CF 3 .
- R 1 can be C 7 -C 11 (e.g., C 7 -C 10 ) aralkyl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R c .
- R 1 can be benzyl or 2- phenylethyl, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R c .
- R 1 can be benzyl.
- R 1 can be heteroaralkyl including 6-10 atoms, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R c .
- the alkyl portion can be Ci-C 2 alkylene
- the heteroaryl portion can be thienyl, furyl, pyrrolyl, or pyridinyl, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1- 3, 1-2, 1) R C .
- R 1 can be C 6 -CiO aryl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R d .
- R 1 can be phenyl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R d .
- R 1 can be heteroaryl including 5-10 (e.g., 5-6) atoms, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R d .
- R 1 can be thienyl, furyl, pyrrolyl, or pyridinyl, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R d .
- R 1 can be other than C 3 -C 6 cycloalkyl.
- R 2 can be other than C 3 -C 6 cycloalkyl.
- R > 2 can be C 6 -Ci 0 (e.g., phenyl) aryl, which is (i) substituted with 1 R > 6 and (ii) optionally substituted with from 1-5 (e.g., 1-3, 1-2, 1) R e .
- each R e can be independently of one another: halo (e.g., chloro); C1-C3 alkyl; C1-C3 haloalkyl (e.g., C 1 - C 3 fluoroalkyl, e.g., 1-5 fluorines can be present; or C 1 -C 3 perfluoroalkyl); CN; hydroxyl; NR m R n (e.g., NH 2 , monoalkylamino, or dialkylamino); C1-C3 alkoxy; or C1-C3 haloalkoxy.
- halo e.g., chloro
- C1-C3 alkyl e.g., C 1 - C 3 fluoroalkyl, e.g., 1-5 fluorines can be present
- C 1 -C 3 perfluoroalkyl CN
- hydroxyl NR m R n (e.g., NH 2 , monoalkylamin
- each R e can be independently of one another: Ci-C 3 alkyl; Ci-C 3 haloalkyl, e.g., Ci-C 3 perfluoroalkyl; halo (e.g., chloro); or CN.
- each R e can be independently of one another: Ci-C 3 alkyl; Ci-C 3 haloalkyl, e.g., Ci-C 3 perfluoroalkyl; halo (e.g., chloro).
- each R e can be independently of one another halo (e.g., chloro).
- R 2 can be C 6 -Ci O aryl, which is (i) substituted with 1 R 6 and (ii) optionally substituted with from 1-5 (e.g., 1-3, 1-2, 1) R e .
- R 2 can be C 6 -CiO aryl, which is (i) substituted with 1 R 6 and (ii) optionally substituted with 1 or 2 R e .
- R 2 can be phenyl, which is (i) substituted with 1 R 6 and (ii) optionally substituted with 1 or 2 (e.g., 1) R e (e.g., halo, e.g., chloro). In other embodiments, R 2 can be phenyl, which is substituted with I R 6 .
- R 2 can have formula (A), in which R 6 (i.e., the moiety -WA) can be attached to a ring carbon that is ortho, meta, ox para (e.g., meta) with respect to the ring carbon that connects the phenyl ring to the 3-position of the pyrazolo[l,5-a] pyrimidine ring, and R e , when present can be connected to ring carbons that are not occupied by WA.
- R 6 i.e., the moiety -WA
- R e when present can be connected to ring carbons that are not occupied by WA.
- R 2 can have formula (A-I), in which R 6 (WA) is attached to the ring carbon that is meta with respect to the ring carbon that connects the phenyl ring to the 3-position of the pyrazolo[l,5-a] pyrimidine ring in formula (I).
- R 2 can have formula (A-2):
- each of R 22 , R 23 , and R 24 can be, independently of one another, hydrogen or R e .
- R e can be as defined anywhere herein.
- each of R , R , and R is hydrogen; or (ii) one of R , R 23 , and R 24 is R e , and the other two are hydrogen.
- each of R 22 , R 23 , and R 24 can be hydrogen. In other embodiments, each of R 22 , R 23 , and R 24 can be a substituent other than hydrogen. In still other embodiments, one or two of R 22 , R 23 , and R 24 can be R e , and the other(s) are hydrogen.
- R 22 , R 23 , and R 24 can be R e , and the other two are hydrogen.
- R 22 can be R e
- each of R 23 and R 24 can be hydrogen.
- R e can be: halo (e.g., chloro); C1-C3 alkyl; or C1-C3 haloalkyl (e.g., C 1 -C 3 fluoroalkyl, e.g., 1-5 fluorines can be present; or C 1 -C 3 perfluoroalkyl).
- R e can be halo (e.g., chloro).
- R 2 can be heteroaryl including 5-10 (e.g., 5-6) atoms, which is (i) substituted with 1 R 6 and (ii) optionally substituted with from 1-5 (e.g., 1-3, 1-2, l) R e .
- each R e when R 2 is heteroaryl and substituted with R e , each R e can be independently as defined anywhere herein.
- each R e can be independently of one another: C1-C3 alkyl; C1-C3 haloalkyl, e.g., C1-C3 perfluoroalkyl; halo (e.g., chloro); e.g., each R e can be halo (e.g., chloro).
- R 2 can be heteroaryl including 5-10 atoms, which is (i) substituted with 1 R 6 and (ii) optionally substituted with from 1-5 (e.g., 1-3, 1-2, 1) R e .
- R 2 can be heteroaryl including 5-10 atoms, which is (i) substituted with 1 R 6 and (ii) optionally substituted with 1 or 2 R e .
- R 2 can be heteroaryl including 5-6 atoms, which is (i) substituted with 1 R 6 and (ii) optionally substituted with 1 or 2 R e .
- R 2 can be heteroaryl including 8-10 atoms, which is (i) substituted with 1 R 6 and (ii) optionally substituted with 1 or 2 R e .
- R 2 can be pyridyl, pyrimidinyl, thienyl, furyl, quinolinyl, oxazolyl, thiazolyl, imidazolyl, isoxazolyl, indolyl, benzo[l,3]-dioxolyl, benzo[ 1,2,5]- oxadiazolyl, isochromenyl-1-one, 3-H-isobenzofuranyl-l-one (e.g., pyridyl, thienyl, or indolyl, e.g., pyridyl or indolyl, e.g., pyridyl), each of which is (i) substituted with 1 R 6 and (ii) optionally substituted with 1 or 2 R e .
- R 2 can be pyridyl substituted with 1 R 6 .
- W can be -O- .
- W can be a bond
- W can be -W ⁇ C 1-6 alkylene)-.
- W 1 can be -O-.
- W can be -O(Ci_ 3 alkylene)- (e.g., -OCH 2 -, -OCH 2 CH 2 , or -OCH 2 CH 2 CH 2 -, e.g., -OCH 2 -)).
- W can be -NR 7 - (e.g., -NH-). In some embodiments, W can be -(C 1-6 alkylene)W 1 -. In certain embodiments, W 1 is -NR 7 -, in which R 7 can be hydrogen; or W 1 can be -O-. In certain embodiments, W can be -(C 1-3 alkylene)NH- (e.g., -CH 2 NH-). In certain embodiments, W can be -(C 1-3 alkylene)O- (e.g., -CH 2 O-).
- A is an aromatic or heteroaromatic ring system that is (a) substituted with one R 8 ; and (b) optionally substituted with one or more R g .
- A can be C 6 -CiO (e.g., phenyl) aryl, which is (a) substituted with 1 R 8 ; and (b) optionally further substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1, e.g., 1-2) R g , in which R g can be as defined anywhere herein.
- R g can be as defined anywhere herein.
- each R g can be independently of one another:
- Ci-C 6 e.g., Ci-C 3 alkoxy or Ci-C 6 (e.g., Ci-C 3 ) haloalkoxy; or cyano; or (ii) Ci-C 6 (e.g., Ci-C 3 ) alkyl or C r C 6 (e.g., C r C 3 ) haloalkyl.
- each R g can be independently of one another:
- halo e.g., chloro or fluoro
- Ci-C 6 e.g., Ci-C 3 ) haloalkoxy
- Ci-C 6 e.g., Ci-C 3 alkoxy or NR m R n ; or
- Ci-C 6 e.g., Ci-C 3 alkyl or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl.
- A can be C 6 -CiO aryl, which is (i) substituted with 1 R 8 and (ii) optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1, e.g., 1-2) R g .
- A can be phenyl, which is (i) substituted with 1 R 8 and (ii) optionally substituted with from 1-4 (e.g., 1-3, 1-2, 1) R g .
- R 8 can be attached to a ring carbon that is ortho, meta, or para (e.g., meta ox para) with respect to the ring carbon that connects the phenyl ring to W.
- A can have formula (B-I):
- R A3 and R A4 is R 8
- the other of R ⁇ and R A4 and each of R A2 , R A5 , and R A6 is, independently, hydrogen or R g
- R g can be as defined anywhere herein.
- R 8 can be as defined anywhere herein.
- one of R A3 and R A4 can be R 8 , the other of R A3 and R A4 can be hydrogen; and each of R ⁇ , R A5 , and R A6 can be, independently, hydrogen or R g .
- R A3 can be R 8 .
- R A3 can be R 8
- R A4 can be hydrogen
- each of R A2 , R A5 , and R A6 can be hydrogen.
- R A3 can be R 8 ;
- R A4 can be hydrogen; one of R ⁇ , R A5 , and R A6 (e.g., R A5 ) can be R g (e.g., halo) and the other two of R A2 , R A5 , and R A6 can be hydrogen.
- R A4 can be R 8 .
- R A4 can be R 8
- R ⁇ can be hydrogen
- each of R A2 , R A5 , and R A6 can be hydrogen.
- R A3 can be R 8 ;
- R A4 can be hydrogen; one of R ⁇ , R A5 , and R A6 can be R g (e.g., halo) and the other two of R A2 , R A5 , and R A6 can be hydrogen.
- A can be heteroaryl including 5-10 atoms, which is (a) substituted with 1 R 8 ; and (b) is optionally substituted with from 1-3 (e.g., 1-2, 1) R g , in which R g can be as defined anywhere herein.
- A can be heteroaryl including 5-10 atoms, which is (a) substituted with 1 R 8 ; and (b) is optionally substituted with from 1-3 (e.g., 1-2, 1) R g .
- A can be pyrrolyl, pyridyl, pyridyl-N-oxide, pyrazolyl, pyrimidinyl, thienyl, furyl, quinolinyl, oxazolyl, thiazolyl, imidazolyl, isoxazolyl, indolyl, benzo[l,3]-dioxolyl, benzo[l,2,5]-oxadiazolyl, isochromenyl-1-one, 3-H- isobenzofuranyl-1-one (e.g., pyridyl, thienyl, or indolyl, e.g., pyridyl), which is (i) substituted with 1 R 8 and (ii) optionally substituted with 1-3 (e.g., 1-2, 1) R g .
- A can be pyrrolyl, pyridyl, pyrimidinyl, pyrazolyl, thienyl, furyl, quinolyl, oxazolyl, thiazolyl, imidazolyl, or isoxazolyl, each of which is (a) substituted with 1 R 8 ; and (b) is optionally substituted with from 1-3 (e.g., 1-2, 1) R g .
- A can be pyridyl, pyrimidinyl, thienyl, furyl, oxazolyl, thiazolyl, imidazolyl, or isoxazolyl, each of which is (a) substituted with 1 R 8 ; and (b) is optionally substituted with from 1-3 (e.g., 1-2, 1) R g .
- A can be pyridyl in which W is attached to the 2- or 3- position of the pyridiyl ring.
- A can be pyridyl in which W is attached to the 2-position of the pyridyl ring, and R 8 is attached to the 4- or the 6-position of the pyridyl ring.
- Such rings can be further substituted with 1, 2 or 3 R g (e.g., halo, e.g., chloro; or NR g R h , e.g., NH 2 ).
- R 8 can be:
- R can be:
- R can be
- R 8 can be any one of: (8-i), (8-i'), (8-ii), (8-iii), (8-iv), (8- v), or (8-vi) or any subset delineated above.
- R 8 can be -W 2 - S(O) n R 9 or -W ⁇ S(O) n NR 10 R 11 (e.g., -W 2 -S(O) n R 9 ).
- R 8 can be - W 2 -C(O)OR 12 .
- R 8 can be any two of: (8-i), (8-i'), (8-ii), (8-iii), (8-iv), (8- v), or (8-vi) or any subset delineated above.
- R 8 can be -W 2 - S(O) n R 9 or -W ⁇ S(O) n NR 10 R 1 ⁇ (e.g., -W 2 -S(O) n R 9 ) and any one of (8-ii), (8-ii), (8-iv), (8-v), or (8-vi) or any subset delineated above.
- R 8 can be:
- R 8 can be any two of (8-ii), (8-iii), (8-iv), (8-v), or (8-vi) or any subset delineated above.
- R 8 can be any three of: (8-i), (8-i'), (8-ii), (8-iii), (8-iv), (8-v), or (8-vi) or any subset delineated above.
- R 8 can be -W 2 -S(O) n R 9 , -W ⁇ S(O) n NR 10 R 1 ⁇ and -W 2 - C(O)OR 12 .
- R 8 can be: • -W 2 -S(O) n R 9 or -W ⁇ S(O) n NR 10 R 1 J (e.g., -W 2 -S(O) n R 9 ); and
- R 8 can be any three of (8-iii), (8-iv), (8-v), or (8-vi) or any subset delineated above.
- R 8 can be -W 2 -S(O) n R 9 (e.g., -W 2 -S(O) 2 R 9 , in which n is 2).
- W 2 can be a bond
- R 8 is connected to variable A by the sulfur (S) atom.
- R 9 can be Ci-C 6 (e.g., C 1 -C 5 or C 2 -C 6 ) alkyl or Ci-C 6 (e.g., C 1 -C 5 or C 1 -C 3 ) haloalkyl, optionally substituted with from 1-2 R a .
- R 9 can be Ci-C 6 (e.g., C1-C5 or C 2 -Cg) alkyl, optionally substituted with from 1-2 (e.g., 1) R a .
- R 9 can be unsubstituted branched or unbranched Ci-C
- R 9 can be methyl (CH3).
- R 9 can be ethyl (CH 2 CHs).
- R 9 can be ⁇ opropyl (CH(CHs) 2 ).
- R 9 can be branched or unbranched C 2 -C 6 (e.g., C 3 -C 6 , or C3-C5) alkyl, which is substituted with 1 R a .
- R a can be: hydroxyl; C 1 - C 6 (e.g., C1-C3) alkoxy; C3-C7 cycloalkoxy or C 6 -CiO aryloxy, each of which can be optionally substituted with R c and R d , respectively; NR m R n ; halo; or heterocyclyl including 3-8 atoms, which is optionally substituted with from 1-5 R c .
- R a can be hydroxyl, Ci-C 6 (e.g., Ci-C 3 ) alkoxy, or NR m R n .
- R a e.g., hydroxyl
- R 9 can be hydroxyl substituted C 3 - C 6 (e.g., C3-C5) alkyl.
- R 9 can be 3-hydroxypropyl or 2,2- dimethy 1-3 -hy droxypropy 1.
- R 9 can be C 7 -C 11 aralkyl (e.g., benzyl), optionally substituted with from 1-3 (e.g., 1-2, 1) R c .
- R > 9 can be C 6 -CiO aryl, optionally substituted with from 1-
- W 2 can be a bond.
- W 2 can be C1-C3 alkylene.
- R 8 can be -W ⁇ S(O) n NR 10 R 11 (e.g., -W ⁇ S(O) 2 NR 10 R 1 ⁇ in which n is 2).
- W 2 can be a bond, and R 8 is connected to variable A by the sulfur (S) atom.
- R 10 and R 11 can be hydrogen.
- R 8 can be -S(O) 2 NH 2 .
- one of R 10 and R 11 can be hydrogen, and the other of R 10 and R 11 can be:
- Ci-C 6 e.g., Ci-C 3 alkyl or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1)
- R a e.g., R a can be: hydroxyl; Ci-C 6 (e.g., C1-C3) alkoxy; C3-C7 cycloalkoxy or C 6 -CiO aryloxy, each of which can be optionally substituted with R c and R d , respectively; NR m R n ; or heterocyclyl including 3-8 atoms, which is optionally substituted with from 1-5 R c ); or
- R 10 and R 11 can each be, independently of one another:
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -5 R a ;
- R 10 and R 11 can each be, independently of one another:
- Ci-C 6 e.g., Ci-C 3 alkyl or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl, each of which is optionally substituted with from 1-5 (e.g., 1-5, 1-4, 1-3, 1-2, 1)
- R a e.g., R a can be: hydroxyl; Ci-C 6 (e.g., Ci-C 3 ) alkoxy; C 3 -C 7 cycloalkoxy or C 6 -CiO aryloxy, each of which can be optionally substituted with R c and R d , respectively; NR m R n ; or heterocyclyl including 3-8 atoms, which is optionally substituted with from 1-5 R c ); or
- R 10 and R 11 together with the nitrogen atom to which they are attached can form a heterocyclyl including 3-10 (e.g., 3-8, or 3-6) atoms or a heterocycloalkenyl including 3-10 (e.g., 3-8, or 3-6) atoms, each of which is optionally substituted with from 1-5 (1-4, 1-3, 1-2, 1) R c .
- the heterocyclyl can further include one or more additional ring heteroatoms (e.g., N, O, or S).
- R 10 and R 11 together with the nitrogen atom to which they are attached can form a heterocyclyl including 3-10 (e.g., 3-8, 3-6, or 5-6) atoms, which is optionally substituted with from 1-5 (1-4, 1-3, 1-2, 1) R c .
- R 10 and R 11 together with the nitrogen atom to which they are attached can form a morpholinyl, piperidyl, pyrrolidinyl, or piperazinyl ring, each of which is optionally substituted with from 1-5 (1-4, 1-3, 1-2, 1) R c .
- R 8 can be -W 2 -C(O)OR 12 .
- R 12 can be:
- Ci-C 6 alkyl which is optionally substituted with from 1-3 (e.g., 1-2, 1) R a ; or (iii) C3-C7 cycloalkyl or C 7 -C 11 aralkyl, each of which is optionally substituted with from 1-5 R c ; or
- R 12 can be hydrogen. In other embodiments, R 12 can be a substituent other than hydrogen.
- W 2 can be Ci-C 6 alkylene; or a bond. In certain embodiments, W 2 can be Ci-C 6 alkylene. For example, W 2 can be C 1 - C 3 alkylene, such as CH 2 or CH 2 CH 2 .
- W 2 can be a bond.
- R 8 can be -W ⁇ C(O)NR 10 R 1 ⁇
- Embodiments can include, for example, any one or more of the features described above in conjunction with -W ⁇ S(O) n NR 10 R 1 ⁇
- R 8 can be -W 2 -CN.
- R 8 can be Ci-Ci 2 alkyl or Ci-Ci 2 haloalkyl, each of which is (a) substituted with from 1 R h , and (b) optionally further substituted with from 1 or 2 R a (e.g., R a can be C3-C7 cycloalkyl, which is optionally substituted with from 1-5 R c ).
- R h at each occurrence can be, independently, hydroxyl, Ci-C 6 alkoxy, Ci-C 6 haloalkoxy; C3-C10 cycloalkoxy, which is optionally substituted with from 1-5 R c ; or C 6 -CiO aryloxy or heteroaryloxy including 5-10 atoms, each of which is optionally substituted with from 1-5 R d .
- R can have the following formula: -C(R )(R )(R ), in which each of R 81 and R 82 is, independently,Ci-Ci 2 alkyl or Ci-Ci 2 haloalkyl, each of which is optionally further substituted with from 1 or 2 R a (e.g., R a can be C 3 -C 7 cycloalkyl, which is optionally substituted with from 1-5 R c ); C 3 -C 7 cycloalkyl, which is optionally substituted with from 1-5 R c ; or C 6 -CiO aryl, which is optionally substituted with from 1-10 R d ; and R h can be as defined anywhere herein.
- R 8 can be -NR 13 R 14 , one of R 13 and R 14 is hydrogen or Ci- C3 alkyl (e.g., hydrogen); and the other of R 13 and R 14 can be: (i) -S(O) n R 9 ; or (ii) -C(O)OR 12 ; or (iii) -C(O)NR 10 R 11 ; or (iv) C 1 -C 12 alkyl or C 1 -C 12 haloalkyl, each of which is:
- each of n, R 9 , R 10 , R 11 , R 12 , R h , R a , and R d can be, independently, as defined anywhere herein.
- R 12 can be other than hydrogen.
- R 8 can be other than -NR 13 R 14 (e.g., NHSO 2 R 9 ) and/or - C(O)OR 12 (e.g., COOH).
- each of R 3 and R 4 can be, independently: (i) hydrogen; or (ii) halo; or
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1-3 R a .
- each of R 3 and R 4 can be, independently: (i) hydrogen; or (ii) halo; or
- each of R 3 and R 4 can be independently hydrogen or halo (e.g., fluoro).
- each of R 3 and R 4 can be hydrogen.
- each of R 3 and R 4 can be a substituent other than hydrogen (e.g., halo, e.g., fluoro).
- one of R 3 and R 4 can be hydrogen, and the other can be:
- Ci-C 6 e.g., Ci-C 3 alkyl or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl (e.g., perhaloalkyl, e.g., perfluoroalkyl), each of which is optionally substituted with from 1-3 R a .
- Ci-C 6 e.g., Ci-C 3 alkyl
- Ci-C 6 e.g., Ci-C 3
- haloalkyl e.g., perhaloalkyl, e.g., perfluoroalkyl
- R 5 can be: (i) halo; or
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R a ;
- R 5 can be halo, cyano, Ci-C 6 (e.g., Ci-C 3 ) alkyl, or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be chloro or bromo (e.g., chloro), cyano, Ci-C 6 (e.g., Ci-C 3 ) alkyl, or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be halo, Ci-C 6 (e.g., Ci-C 3 ) alkyl, or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be chloro or bromo (e.g., chloro), Ci-C 6 (e.g., Ci- C 3 ) alkyl, or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be halo (e.g., chloro) or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl (e.g., CF 3 ).
- R 5 can be chloro or bromo (e.g., chloro) or Ci-C 6 (e.g., Ci- C 3 ) haloalkyl.
- R 5 can be chloro; cyano; CH 3 ; or CF 3 .
- R 5 can be chloro; CH 3 ; or CF 3 .
- R 5 can be chloro or CF 3 .
- R 5 can be hydrogen
- R 5 can be hydrogen, halo, cyano, Ci-C 6 (e.g., Ci-C 3 ) alkyl, or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be hydrogen, chloro or bromo (e.g., chloro), cyano, Ci-C 6 (e.g., Ci-C 3 ) alkyl, or Ci-C 6 (e.g., d-C 3 ) haloalkyl.
- R 5 can be hydrogen, halo, Ci-C 6 (e.g., Ci-C 3 ) alkyl, or C 1 - C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be hydrogen, chloro or bromo (e.g., chloro), Ci-C 6 (e.g., Ci-C 3 ) alkyl, or Ci-C 6 (e.g., C r C 3 ) haloalkyl.
- R 5 can be hydrogen, halo (e.g., chloro), or Ci-C 6 (e.g., C 1 - C 3 ) haloalkyl (e.g., CF 3 ).
- halo e.g., chloro
- Ci-C 6 e.g., C 1 - C 3
- haloalkyl e.g., CF 3
- R 5 can be hydrogen, chloro or bromo (e.g., chloro), or C 1 - C 6 (e.g., Ci-C 3 ) haloalkyl.
- R 5 can be hydrogen, chloro, cyano, CH 3 , or CF 3 . In certain embodiments, R 5 can be hydrogen, chloro, CH 3 , or CF 3 . In certain embodiments, R 5 can be hydrogen, chloro, or CF 3 .
- R 5 can be Ci-C 6 (e.g., Ci-C 3 ) haloalkyl (e.g., perfluoroalkyl). In certain embodiments, R 5 can be CF 3 .
- R 5 can be halo (e.g., chloro).
- R 5 can be Ci-C 6 (e.g., Ci-C 3 ) alkyl (e.g., CH 3 ).
- R 5 can be cyano.
- R 8 when R 8 is -W 2 -S(O) n R 9 or -W ⁇ S(O) n NR 10 R 1 ⁇ then R 5 can be hydrogen or hydrogen and any one or more of the permissible non-hydrogen substitutents delineated above for R 5 .
- R 5 when R 8 is other than -W 2 -S(O) n R 9 or -W ⁇ S(O) n NR 10 R 1 ⁇ then R 5 can be other than hydrogen.
- a subset of compounds includes those in which R 2 has formula (C-I):
- each of R , 22 , ⁇ R23 , and . ⁇ R24 is, independently, hydrogen or R e ; and one of R A2 , R A3 , R A4 , R A5 , and R A6 is R 8 , and the others are each, independently, hydrogen or R g ; and
- W can be as defined anywhere herein.
- each of R 22 , R 23 , and R 24 is hydrogen;
- R 22 , R 23 , and R 24 is R e , and the other two are hydrogen; one of R A2 , R A3 , R A4 , R A5 , and R A6 is R 8 , and the others are each, independently, hydrogen or R g ; and
- W can be as defined anywhere herein.
- Embodiments can include, for example, one or more of the following features (and/or any one or more other features described anywhere herein).
- W can be -O-, a bond, -OCH 2 -, or -NH- (e.g., -O-, a bond, or -OCH 2 -).
- R e , R 8 , and R g can each be, independently, as defined anywhere herein.
- Each of R 22 , R 23 , and R 24 can be hydrogen; or each of R 22 , R 23 , and R 24 can be a substituent other than hydrogen; or one or two of R 22 , R 23 , and R 24 can be R e , and the other(s) can be hydrogen.
- R , R , and R can be R , and the other two can be hydrogen.
- R 22 can be R e
- each of R 23 and R 24 can be hydrogen.
- R e can be: halo (e.g., chloro); C1-C3 alkyl; or C1-C3 haloalkyl (e.g., C1-C3 fluoroalkyl, e.g., 1-5 fluorines can be present; or C 1 -C 3 perfluoroalkyl).
- R e can be halo (e.g., chloro).
- R A3 and R A4 can be R 8 , the other of R A3 and R A4 can be hydrogen; and each of R ⁇ , R A5 , and R A6 can be, independently, hydrogen or R g .
- R A3 can be R 8 , R A4 can be hydrogen, and each of R ⁇ , R A5 , and R A6 can be hydrogen; or R A3 can be R 8 ; R A4 can be hydrogen; one of R A2 , R A5 , and R A6 (e.g., R A5 ) can be R g (e.g., halo, e.g., fluoro) and the other two of R A2 , R A5 , and R A6 can be hydrogen.
- R g e.g., halo, e.g., fluoro
- R A4 can be R 8 , R ⁇ can be hydrogen, and each of R ⁇ , R A5 , and R A6 can be hydrogen.
- R ⁇ can be R 8 ;
- R A4 can be hydrogen; one of R A2 , R A5 , and R A6 can be R g (e.g., halo) and the other two of R ⁇ , R A5 , and R A6 can be hydrogen.
- R 8 can be -W 2 -S(O) n R 9 , in which n is 2, and each of W 2 and R 9 can be as defined anywhere herein.
- W 2 can be a bond.
- R 9 can be C1-C10 alkyl, optionally substituted with from 1-2 R a .
- R 9 can be CH 3 , CH 2 CH 3 , or ⁇ opropyl.
- R A3 can be -W 2 -S(O) n R 9 .
- n can be 2.
- W 2 can be a bond.
- R 9 can be C1-C10 alkyl, optionally substituted with from 1-2 R a .
- R 9 can be C1-C3 alkyl (e.g., CH 3 ).
- R 9 can be C 2 -C 8 alkyl substituted with 1 R a (e.g., R a can be hydroxyl or Ci-C 3 alkoxy).
- Each of R A2 , R A4 , R A5 , and R A6 can be hydrogen.
- R A5 can be R g
- each of R A2 , R A4 , and R A6 can be hydrogen.
- R 8 can be -W -C(O)OR , 12.
- W and R 12 can be as defined anywhere herein.
- W 2 can be a bond or Ci-C 6 alkylene.
- R 12 can be hydrogen or Ci-C 6 alkyl.
- R A4 can be -W 2 -C(O)OR 12 .
- W 2 can be a bond or Ci-C 6 alkylene (e.g., CH 2 ).
- R 12 can be hydrogen or Ci-C 3 alkyl.
- Each of R A2 , R A3 , R A5 , and R A6 can be hydrogen.
- R 8 can be CN.
- the compounds can have formula (II):
- each of R 1 , R 2 , R 3 , and R 4 can be, independently, as defined anywhere herein (generically, subgenerically, or specifically).
- the compounds can have formula (III):
- each of R 1 , R 2 , and R 5 can be, independently, as defined anywhere herein (generically, subgenerically, or specifically).
- the compounds can have formula (IV):
- each of R 1 and R 2 can be, independently, as defined anywhere herein (generically, subgenerically, or specifically).
- the compounds can have formula (V):
- each of R 1 , R 3 , R 4 , R 5 , R e , W, and A can be, independently, as defined anywhere herein (generically, subgenerically, or specifically).
- the compounds can have formula (VI):
- each of R 1 , R 3 , R 4 , R 5 , R 22 , R 23 , R 24 , W, and A can be, independently, as defined anywhere herein (generically, subgenerically, or specifically).
- the compounds can have formula (VII):
- each of R 1 , R 3 , R 4 , R 5 , R 22 , R 23 , R 24 , R ⁇ , R ⁇ , R A4 , R A5 , R A6 , W, and A can be, independently, as defined anywhere herein (generically, subgenerically, or specifically).
- the compounds of formulas (II), (III), (IV), (V), (VI), and (VII) can include any one or more of the following features.
- R 1 can be:
- Ci-C 6 e.g., Ci-C 3 or Ci-C 2 alkyl or Ci-C 6 (e.g., Ci-C 3 or Ci-C 2 ) haloalkyl; or
- C 6 -CiO e.g., phenyl
- aryl or heteroaryl including 5-10 (e.g., 5-6 atoms), each of which is optionally substituted with from 1-5 R d ; or
- C 7 -C 11 e.g., C 7 -C 10
- heteroaralkyl including 6-11 (e.g., 6-10) atoms, each of which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R c .
- R 1 can be hydrogen
- R 1 can be:
- Ci-C 6 e.g., Ci-C 3 or Ci-C 2 alkyl or Ci-C 6 (e.g., Ci-C 3 or Ci-C 2 ) haloalkyl; or (iii) C 6 -Ci 0 (e.g., phenyl) aryl, which is optionally substituted with from 1-5 R d ; or (iv) C 7 -Cn (e.g., C 7 -CiO, benzyl) aralkyl, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R c .
- C 6 -Ci 0 e.g., phenyl
- C 7 -Cn e.g., C 7 -CiO, benzyl
- R 1 can be:
- heteroaryl including 5-10 (e.g., 5-6 atoms), which is optionally substituted with from 1-5 R d ; or
- heteroaralkyl including 6-11 (e.g., 6-10) atoms, which is optionally substituted with from 1-5 (e.g., 1-4, 1-3, 1-2, 1) R c .
- R 1 can be: H; CH 3 , CH 2 CH 3 , or CH(CH 3 ) 2 ; CF 3 ; phenyl, which is optionally substituted with from 1-5 R d ; or benzyl, which is optionally substituted with from 1-5 R c .
- R 2 can have formula (A), (A-I), (A-2), or (C-I) as defined anywhere herein.
- W can be -O- .
- W can be a bond.
- W can be -W ⁇ C 1-6 alkylene)-.
- W 1 can be -O-.
- W can be -O(Ci_ 3 alkylene)- (e.g., -OCH 2 -).
- W can be -(C 1-6 alkylene)W 1 -.
- W 1 is -NR 9 -, in which R 9 can be hydrogen; or W 1 can be -O-.
- W can be -(C 1-3 alkylene)NH- (e.g., -CH 2 NH-).
- W can be -(C 1-3 alkylene)O- (e.g., -CH 2 O-).
- W can be -NR 7 -, (e.g., -NH-).
- A can be phenyl, which is (i) substituted with 1 R 8 and (ii) optionally substituted with from 1-5 (e.g., 1-3, 1-2, 1) R g , in which R g can be as defined anywhere herein.
- R ⁇ and R A4 are R 8 , and the other of R A3 and R A4 is hydrogen; and each of R A2 , R A5 , and R A6 is, independently, hydrogen or R g , in which R 8 and R g can be as defined anywhere herein.
- A can be heteroaryl including 5-10 atoms, which is (a) substituted with 1 R 8 ; and (b) is optionally substituted with from 1-3 (e.g., 1-2, 1) R g , in which R g can be as defined anywhere herein.
- R e , R 8 , and R g can be, independently, as defined anywhere herein.
- R 8 can be:
- R 9 , R 10 , R 11 , and R 12 can be, independently, as defined anywhere herein (e.g., as defined in conjunction with formula (C-I)).
- W 2 , n, R 22 , R 23 , R 24 , R A2 , R A3 , R A4 , R A5 , and R A6 can be as defined in conjunction with formula (C-I).
- Each of R 3 and R 4 can be hydrogen.
- R 5 can be: (ii) halo; or
- Ci-C 6 alkyl or Ci-C 6 haloalkyl each of which is optionally substituted with from 1 -3 R e ;
- R 5 can be halo (e.g., chloro) or Ci-C 6 (e.g., Ci-C 3 ) haloalkyl (e.g., CF 3 ).
- R 1 , R 3 , R 4 , and R 5 can be a substituent other than hydrogen.
- the actual electronic structure of some chemical entities cannot be adequately represented by only one canonical form (i.e. Lewis structure). While not wishing to be bound by theory, the actual structure can instead be some hybrid or weighted average of two or more canonical forms, known collectively as resonance forms or structures.
- Resonance structures are not discrete chemical entities and exist only on paper. They differ from one another only in the placement or "localization" of the bonding and nonbonding electrons for a particular chemical entity. It can be possible for one resonance structure to contribute to a greater extent to the hybrid than the others.
- the written and graphical descriptions of the embodiments of the present invention are made in terms of what the art recognizes as the predominant resonance form for a particular species.
- the compounds described herein can be synthesized according to methods described herein (or variations thereof) and/or conventional, organic chemical synthesis methods from commercially available starting materials and reagents or from starting materials and reagents that can be prepared according to conventional organic chemical synthesis methods.
- the compounds described herein can be separated from a reaction mixture and further purified by a method such as column chromatography, high-pressure liquid chromatography, or recrystallization.
- a method such as column chromatography, high-pressure liquid chromatography, or recrystallization.
- further methods of synthesizing the compounds of the formulae herein will be evident to those of ordinary skill in the art. Additionally, the various synthetic steps may be performed in an alternate sequence or order to give the desired compounds.
- Synthetic chemistry transformations and protecting group methodologies useful in synthesizing the compounds described herein are known in the art and include, for example, those such as described in R. Larock, Comprehensive Organic Transformations, VCH Publishers (1989); T.W. Greene and P.G.M. Wuts, Protective Groups in Organic Synthesis, 2d. Ed., John Wiley and Sons (1991); L. Fieser and M. Fieser, Fieser and Fieser's Reagents for Organic Synthesis, John Wiley and Sons (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995), and subsequent editions thereof.
- compounds of formula (I) can be prepared according to Scheme 1.
- the compounds of formula (I) can be prepared by reacting phenylacetonitriles (1) with esters (2), typically in the presence of a base such as sodium hydride in an aprotic solvent such as THF at ambient temperatures, for several hours. Reaction of the resulting beta-keto-nitrile (3) with hydrazine in a solvent such as ethanol at elevated temperatures, typically reflux, results in aminopyrazoles (4). The aminopyrazoles (4) are reacted with enamines (5) in refluxing acetic acid for several hours to afford the pyrazolo[l,5-a]pyrimidines (6).
- T a protected hydroxyl group such as a methoxy or benzyloxy
- Typical conditions for deprotection when T is a methoxy include treatment with pyridine hydrochloride at 200 0 C for 0.5 - 2 h or treatment with BBr 3 or other methods known to those skilled in the art.
- compounds of formula (I) can be prepared according to Scheme 2.
- the formation of the biarylether of formula (I) is accomplished by treatment with a base such as potassium carbonate, typically in a polar solvent such as dimethylformamide or dimethylsulfoxide, at elevated temperatures, typically 100 0 C to 150 0 C for several hours.
- a base such as potassium carbonate
- a polar solvent such as dimethylformamide or dimethylsulfoxide
- the formation of the biarylether (I) is accomplished with a coupling reaction using a metal catalyst such as a copper salt or a palladium salt in the presence of a base and a solvent such as 1,4-dioxane at elevated temperatures.
- compounds of formula (I) can be prepared according to Scheme 3.
- compounds of formula (I) can be prepared according to Scheme 4.
- certain compounds prepared by Scheme 1 contain a free NH 2 moiety on the phenyl ring that is attached to the 3-position of the pyrazolo[l,5- ajpyrimidine ring system.
- compounds of formula (I) can be prepared according to Scheme 5.
- the compounds of this invention may contain one or more asymmetric centers and thus occur as racemates and racemic mixtures, single enantiomers, individual diastereomers and diastereomeric mixtures. All such isomeric forms of these compounds are expressly included in the present invention.
- the compounds of this invention may also contain linkages (e.g., carbon-carbon bonds, carbon-nitrogen bonds such as amide bonds) wherein bond rotation is restricted about that particular linkage, e.g. restriction resulting from the presence of a ring or double bond. Accordingly, all cis/trans and E/Z isomers and rotational isomers are expressly included in the present invention.
- the compounds of this invention may also be represented in multiple tautomeric forms, in such instances, the invention expressly includes all tautomeric forms of the compounds described herein, even though only a single tautomeric form may be represented (e.g., alkylation of a ring system may result in alkylation at multiple sites, the invention expressly includes all such reaction products). All such isomeric forms of such compounds are expressly included in the present invention.
- the compounds of this invention include the compounds themselves, as well as their salts and their prodrugs, if applicable.
- a salt for example, can be formed between an anion and a positively charged substituent (e.g., amino) on a compound described herein. Suitable anions include chloride, bromide, iodide, sulfate, nitrate, phosphate, citrate, methanesulfonate, trifluoroacetate, and acetate.
- a salt can also be formed between a cation and a negatively charged substituent (e.g., carboxylate) on a compound described herein.
- Suitable cations include sodium ion, potassium ion, magnesium ion, calcium ion, and an ammonium cation such as tetramethylammonium ion.
- Examples of prodrugs include esters and other pharmaceutically acceptable derivatives, which, upon administration to a subject, are capable of providing active compounds.
- Pharmaceutically acceptable salts of the compounds of this invention include those derived from pharmaceutically acceptable inorganic and organic acids and bases.
- suitable acid salts include acetate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, citrate, camphorate, camphorsulfonate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptanoate, glycolate, hemisulfate, heptanoate, hexanoate, hydrochloride, hydrobromide, hydroiodide, 2- hydroxyethanesulfonate, lactate, maleate, malonate, methanesulfonate, 2- naphthalenesulfonate, nicotinate, nitrate, palmoate, pectinate, persulfate, 3- phenylpropionate, phosphate,
- Salts derived from appropriate bases include alkali metal (e.g., sodium), alkaline earth metal (e.g., magnesium), ammonium and N-(alkyl) 4 salts.
- alkali metal e.g., sodium
- alkaline earth metal e.g., magnesium
- ammonium e.g., ammonium
- N-(alkyl) 4 salts e.g., ammonium
- This invention also envisions the quaternization of any basic nitrogen-containing groups of the compounds disclosed herein. Water or oil-soluble or dispersible products may be obtained by such quaternization.
- Salt forms of the compounds of any of the formulae herein can be amino acid salts of carboxy groups (e.g. L-arginine, -lysine, -histidine salts).
- pharmaceutically acceptable carrier or adjuvant refers to a carrier or adjuvant that may be administered to a subject (e.g., a patient), together with a compound of this invention, and which does not destroy the pharmacological activity thereof and is nontoxic when administered in doses sufficient to deliver a therapeutic amount of the compound.
- compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, self-emulsifying drug delivery systems (SEDDS) such as d- ⁇ -tocopherol polyethyleneglycol 1000 succinate, surfactants used in pharmaceutical dosage forms such as Tweens or other similar polymeric delivery matrices, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-polylene glycol, glycine, sorbic acid, potassium
- Cyclodextrins such as ⁇ -, ⁇ -, and ⁇ -cyclodextrin, or chemically modified derivatives such as hydroxyalkylcyclodextrins, including 2- and 3-hydroxypropyl- ⁇ -cyclodextrins, or other solubilized derivatives may also be advantageously used to enhance delivery of compounds of the formulae described herein.
- the compounds described herein can be used for treating (e.g., controlling, ameliorating, alleviating, slowing the progression of, delaying the onset of, or reducing the risk of developing) or preventing one or more diseases, disorders, conditions or symptoms mediated by LXRs (e.g., cardiovascular diseases (e.g., acute coronary syndrome, restenosis), atherosclerosis, atherosclerotic lesions, type I diabetes, type II diabetes, Syndrome X, obesity, lipid disorders (e.g., dyslipidemia, hyperlipidemia, hypertriglyceridemia, hypercholesterolemia, low HDL and high LDL), cognitive disorders (e.g., Alzheimer's disease, dementia), inflammatory diseases (e.g., multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease, Crohn's disease, endometriosis, LPS-induced sepsis, acute contact dermatitis of the ear, chronic atherosclerotic inflammation of the artery wall), celiac,
- a disorder or physiological condition that is mediated by LXR refers to a disorder or condition wherein LXR can trigger the onset of the condition, or where inhibition of a particular LXR can affect signaling in such a way so as to treat, control, ameliorate, alleviate, prevent, delay the onset of, slow the progression of, or reduce the risk of developing the disorder or condition.
- cardiovascular diseases e.g., acute coronary syndrome, restenosis
- atherosclerosis atherosclerotic lesions
- type I diabetes type II diabetes
- Syndrome X obesity
- lipid disorders e.g., dyslipidemia, hyperlipidemia, hypertriglyceridemia, hypercholesterolemia, low HDL and high LDL
- cognitive disorders e.g., Alzheimer's disease, dementia
- inflammatory diseases e.g., multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease, Crohn's disease, endometriosis, LPS-induced sepsis, acute contact dermatitis of the ear, chronic atherosclerotic inflammation of the artery wall
- celiac thyroiditis
- skin aging e.g., skin aging is derived from chronological aging, photoaging, steroid-induced skin thinning, or a combination thereof
- connective tissue disease e.g., osteoarthritis or tendonitis
- LXR modulators that activate cholesterol efflux e.g., upregulate ABCAl
- SREBP-Ic can both reduce atherosclerotic risk and minimize the likelihood of concommitantly increasing serum and hepatic triglyceride levels.
- Candidate compounds having differential activity for regulating ABCAl (ABCGl) vs. SREBP-Ic can be can be evaluated using conventional pharmacological test procedures, which measure the affinity of a candidate compound to bind to LXR and to upregulate the gene ABCAl .
- LXR ligands can be identified initially in cell-free LXR beta and LXR alpha competition binding assays. LXR ligands can be further characterized by gene expression profiling for tissue selective gene regulation.
- the compounds described herein have agonist activity for ABCAl transactivation but do not substantially affect (e.g., inhibit) SREBP-Ic gene expression in differentiated THP-I macrophages. Gene expression analysis in an antagonist mode can be used to further delineate differential regulation of ABCAl and SREBP-Ic gene expression.
- the compounds described herein preferentially antagonize SREBP-Ic activation (a marker for genes involved in cholesterol and fatty acid homeostasis) but do not substantially affect (e.g., have relatively minimal or additive effects) on ABCAl gene expression or genes known to enhance HDL biogenesis (based on a competition assay with known potent synthetic LXR agonists).
- Cell type or tissue specificity may be further evaluated in additional cell lines, intestinal, CaCo2 or liver, HepG2 and Huh-7 cells where ABCAl activity is believed to influence net cholesterol absorption and reverse cholesterol transport.
- additional cell lines intestinal, CaCo2 or liver, HepG2 and Huh-7 cells where ABCAl activity is believed to influence net cholesterol absorption and reverse cholesterol transport.
- the compounds described herein have agonist activity for ABCAl and antagonist activity for SREBP-Ic (e.g., as determined by gene specific modulation in cell based assays).
- the compounds described herein (in the agonist mode) have at least about 20% efficacy for ABCAl activation by LXR and do not substantially agonize SREBP-Ic (at most about 25% efficacy relative to a reference compound N-(2,2,2-trifluoro-ethyl)-N-[4-(2,2,2-trifluoro-l -hydroxy- 1- trifluoromethyl-ethyl)-phenyl]-benzenesulfonamide (Schultz, Joshua R., Genes & Development (2000), 14(22), 2831-2838)).
- the compounds described herein (in the antagonist mode) do not substantially antagonize ABCAl gene expression. While not wishing to be bound by theory, it is believed that there may be an additive effect on ABCAl gene expression relative to the reference compound at their EC50 concentration. In certain embodiments, the compounds described herein (in the antagonist mode) inhibited agonist-mediated SREBP-Ic gene expression in a dose dependent fashion.
- cells can be isolated and RNA prepared and analyzed for the levels of expression of TIMPl, ABCA12, decorin, TNF ⁇ , MMPl, MMP3, and/or IL-8.
- the levels of gene expression i.e., a gene expression pattern
- the levels of gene expression can be quantified, for example, by Northern blot analysis or RT-PCR, by measuring the amount of protein produced, or by measuring the levels of activity of TIMPl, ABCA12, decorin, TNF ⁇ , MMPl, MMP3, and/or IL-8, all by methods known to those of ordinary skill in the art.
- the gene expression pattern can serve as a marker, indicative of the physiological response of the cells to the compounds of formula (I). Accordingly, this response state may be determined before, and at various points during, treatment of the individual with the compounds of formula (I).
- expression levels of cytokines and metalloproteases described herein can be used to facilitate design and/or identification of compounds that treat skin aging through an LXR-based mechanism.
- the invention provides methods (also referred to herein as "screening assays") for identifying modulators, i.e., LXR modulators, that have a stimulatory or inhibitory effect on, for example, TIMPl, ABCA12, decorin, TNF ⁇ , MMPl, MMP3, and/or IL-8 expression.
- An exemplary screening assay is a cell-based assay in which a cell that expresses LXR is contacted with a test compound, and the ability of the test compound to modulate TIMPl, ABCA12, decorin, TNF ⁇ , MMPl, MMP3, and/or IL-8 expression through an LXR-based mechanism.
- Determining the ability of the test compound to modulate TIMPl, ABCA12, decorin, TNF ⁇ , MMPl, MMP3, and/or IL-8 expression can be accomplished by monitoring, for example, DNA, mRNA, or protein levels, or by measuring the levels of activity of TIMPl, ABCA12, decorin, TNF ⁇ , MMPl, MMP3, and/or IL-8, all by methods known to those of ordinary skill in the art.
- the cell for example, can be of mammalian origin, e.g., human.
- cells can be isolated and RNA prepared and analyzed for the levels of expression of ApoD and other genes implicated in osteoarthritis (for example, TNF ⁇ ).
- the levels of gene expression i.e., a gene expression pattern
- a gene expression pattern can be quantified by Northern blot analysis or RT-PCR, by measuring the amount of protein produced, or by measuring the levels of activity of ApoD or other genes, all by methods known to those of ordinary skill in the art.
- the gene expression pattern can serve as a marker, indicative of the physiological response of the cells to the LXR modulator. Accordingly, this response state may be determined before, and at various points during, treatment of the individual with the LXR modulator.
- An exemplary screening assay is a cell-based assay in which a cell that expresses LXR is contacted with a test compound, and the ability of the test compound to modulate ApoD expression and/or aggrecanase activity and/or cytokine elaboration through an LXR-based mechanism. Determining the ability of the test compound to modulate ApoD expression and/or aggrecanase activity and/or cytokine elaboration can be accomplished by monitoring, for example, DNA, mRNA, or protein levels, or by measuring the levels of activity of ApoD, aggrecanase, and/or TNF ⁇ , all by methods known to those of ordinary skill in the art.
- the cell for example, can be of mammalian origin, e.g., human.
- the compounds described herein can be coadministered with one or more other threapeutic agents.
- the additional agents may be administered separately, as part of a multiple dose regimen, from the compounds of this invention (e.g., sequentially, e.g., on different overlapping schedules with the administration of one or more compounds of formula (I) (including any subgenera or specific compounds thereof)).
- these agents may be part of a single dosage form, mixed together with the compounds of this invention in a single composition.
- these agents can be given as a separate dose that is administered at about the same time that one or more compounds of formula (I) (including any subgenera or specific compounds thereof) are administered (e.g., simultaneously with the administration of one or more compounds of formula (I) (including any subgenera or specific compounds thereof)).
- compositions of this invention include a combination of a compound of the formulae described herein and one or more additional therapeutic or prophylactic agents, both the compound and the additional agent can be present at dosage levels of between about 1 to 100%, and more preferably between about 5 to 95% of the dosage normally administered in a monotherapy regimen.
- the compounds and compositions described herein can, for example, be administered orally, parenterally (e.g., subcutaneously, intracutaneously, intravenously, intramuscularly, intraarticularly, intraarterially, intrasynovially, intrasternally, intrathecally, intralesionally and by intracranial injection or infusion techniques), by inhalation spray, topically, rectally, nasally, buccally, vaginally, via an implanted reservoir, by injection, subdermally, intraperitoneally, transmucosally, or in an ophthalmic preparation, with a dosage ranging from about 0.01 mg/Kg to about 1000 mg/Kg, (e.g., from about 0.01 to about 100 mg/kg, from about 0.1 to about 100 mg/Kg, from about 1 to about 100 mg/Kg, from about 1 to about 10 mg/kg) every 4 to 120 hours, or according to the requirements of the particular drug.
- parenterally e.g., subcutaneously, intracutaneously, intraven
- compositions are administered by oral administration or administration by injection.
- the methods herein contemplate administration of an effective amount of compound or compound composition to achieve the desired or stated effect.
- the pharmaceutical compositions of this invention will be administered from about 1 to about 6 times per day or alternatively, as a continuous infusion. Such administration can be used as a chronic or acute therapy.
- the amount of active ingredient that may be combined with the carrier materials to produce a single dosage form will vary depending upon the host treated and the particular mode of administration.
- a typical preparation will contain from about 5% to about 95% active compound (w/w).
- such preparations contain from about 20% to about 80% active compound.
- a maintenance dose of a compound, composition or combination of this invention may be administered, if necessary. Subsequently, the dosage or frequency of administration, or both, may be reduced, as a function of the symptoms, to a level at which the improved condition is retained when the symptoms have been alleviated to the desired level. Patients may, however, require intermittent treatment on a long-term basis upon any recurrence of disease symptoms.
- compositions of this invention may contain any conventional non-toxic pharmaceutically-acceptable carriers, adjuvants or vehicles.
- pH of the formulation may be adjusted with pharmaceutically acceptable acids, bases or buffers to enhance the stability of the formulated compound or its delivery form.
- the compositions may be in the form of a sterile injectable preparation, for example, as a sterile injectable aqueous or oleaginous suspension.
- This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a nontoxic parenterally acceptable diluent or solvent, for example, as a solution in 1,3- butanediol.
- a nontoxic parenterally acceptable diluent or solvent for example, as a solution in 1,3- butanediol.
- acceptable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- Fatty acids such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions.
- These oil solutions or suspensions may also contain a long-chain alcohol diluent or dispersant, or carboxymethyl cellulose or similar dispersing agents which are commonly used in the formulation of pharmaceutically acceptable dosage forms such as emulsions and or suspensions.
- Other commonly used surfactants such as Tweens or Spans and/or other similar emulsifying agents or bioavailability enhancers which are commonly used in the manufacture of pharmaceutically acceptable solid, liquid, or other dosage forms may also be used for the purposes of formulation.
- compositions of this invention may be orally administered in any orally acceptable dosage form including, but not limited to, capsules, tablets, emulsions and aqueous suspensions, dispersions and solutions.
- carriers which are commonly used include lactose and corn starch.
- Lubricating agents such as magnesium stearate, are also typically added.
- useful diluents include lactose and dried corn starch.
- compositions of this invention may also be administered in the form of suppositories for rectal administration.
- These compositions can be prepared by mixing a compound of this invention with a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components.
- suitable non-irritating excipient include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
- Topical administration of the compositions of this invention is useful when the desired treatment involves areas or organs readily accessible by topical application.
- the composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier.
- Carriers for topical administration of the compounds of this invention include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax and water.
- the composition can be formulated with a suitable lotion or cream containing the active compound suspended or dissolved in a carrier with suitable emulsifying agents.
- Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol and water.
- the compositions of this invention may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation.
- topical administration of the compounds and compositions described herein may be presented in the form of an aerosol, a semi-solid pharmaceutical composition, a powder, or a solution.
- a semi-solid composition is meant an ointment, cream, salve, jelly, or other pharmaceutical composition of substantially similar consistency suitable for application to the skin. Examples of semi-solid compositions are given in Chapter 17 of The Theory and Practice of Industrial Pharmacy, Lachman, Lieberman and Kanig, published by Lea and Febiger (1970) and in Remington: The Science and Practice of Pharmacy by University of the Sciences in Philadelphia (Editor); Publisher: Lippincott Williams & Wilkins; Twenty first Edition ( May 1, 2005), which is incorporated herein by reference in its entirety.
- Topically-transdermal patches are also included in this invention. Also within the invention is a patch to deliver active chemotherapeutic combinations herein.
- a patch includes a material layer (e.g., polymeric, cloth, gauze, bandage) and the compound of the formulae herein as delineated herein. One side of the material layer can have a protective layer adhered to it to resist passage of the compounds or compositions.
- the patch can additionally include an adhesive to hold the patch in place on a subject.
- An adhesive is a composition, including those of either natural or synthetic origin, that when contacted with the skin of a subject, temporarily adheres to the skin. It can be water resistant. The adhesive can be placed on the patch to hold it in contact with the skin of the subject for an extended period of time.
- the adhesive can be made of a tackiness, or adhesive strength, such that it holds the device in place subject to incidental contact, however, upon an affirmative act (e.g., ripping, peeling, or other intentional removal) the adhesive gives way to the external pressure placed on the device or the adhesive itself, and allows for breaking of the adhesion contact.
- the adhesive can be pressure sensitive, that is, it can allow for positioning of the adhesive (and the device to be adhered to the skin) against the skin by the application of pressure (e.g., pushing, rubbing,) on the adhesive or device.
- compositions of this invention may be administered by nasal aerosol or inhalation.
- Such compositions are prepared according to techniques well-known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art.
- a composition having the compound of the formulae herein and an additional agent can be administered using any of the routes of administration described herein.
- a composition having the compound of the formulae herein and an additional agent can be administered using an implantable device.
- Implantable devices and related technology are known in the art and are useful as delivery systems where a continuous, or timed- release delivery of compounds or compositions delineated herein is desired. Additionally, the implantable device delivery system is useful for targeting specific points of compound or composition delivery (e.g., localized sites, organs). Negrin et al., Biomaterials, 22(6): 563 (2001). Timed-release technology involving alternate delivery methods can also be used in this invention. For example, timed-release formulations based on polymer technologies, sustained-release techniques and encapsulation techniques (e.g., polymeric, liposomal) can also be used for delivery of the compounds and compositions delineated herein.
- Representative compounds of this invention were evaluated in conventional pharmacological test procedures which measured their affinity to bind to LXR and to upregulate the gene ABCAl, which causes cholesterol efflux from atherogenic cells, such as macrophages.
- LXR activation can be critical for maintaining cholesterol homeostasis, but its coincident regulation of fatty acid metabolism may lead to increased serum and hepatic triglyceride levels.
- Selective LXR modulators that activate cholesterol efflux with minimal impact on SREBP-Ic expression and triglyceride synthesis in liver would be expected to reduce atherosclerotic risk with an improved therapeutic index and minimize the potential for deleterious effects on metabolic balance.
- LXR ligands were identified initially in cell- free LXR beta and LXR alpha competition binding assays. LXR ligands were further characterized by gene expression profiling for tissue selective gene regulation. Selective LXR modulators demonstrate agonist activity for ABCAl transactivation.
- Ligand-binding to the human LXR ⁇ was demonstrated for representative compounds of this invention by the following procedure.
- Buffer 10OmM KCl, 10OmM TRIS (pH 7.4 at +4 0 C), 8.6%glycerol, O.lmM PMSF*,
- Receptor source E.coli extract from cells expressing biotinylated hLXR ⁇ . Extract was made in a similar buffer as above, but with 5OmM TRIS.
- Washed streptavidin and coated flash plates with wash buffer Washed streptavidin and coated flash plates with wash buffer.
- Diluted receptor extract to give Bmax ⁇ 4000 cpm and add to the wells.
- Ligand-binding to the human LXR ⁇ was demonstrated for representative compounds of this invention by the following procedure.
- Buffer 10OmM KCl, 10OmM TRIS (pH 7.4 at +4 0 C), 8.6%glycerol, O.lmM PMSF*,
- Receptor source E.coli extract from cells expressing biotinylated hLXR ⁇ . Extract was made in a similar buffer as above, but with 5OmM TRIS.
- Washed streptavidin and coated flash plates with wash buffer Washed streptavidin and coated flash plates with wash buffer.
- Diluted receptor extract to give Bmax ⁇ 4000 cpm and add to the wells.
- the THP-I monocytic cell line (ATCC # TIB-202) was obtained from American Type Culture Collection (Manassas, VA) and cultured in RPMI 1640 medium (Gibco, Carlsbad, Ca) containing 10% FBS, 2 mM L-glutamine, and 55 uM beta- Mercaptoethanol (BME). Cells were plated in 96-well format at a density of 7.5 X 10 4 in complete medium containing 50-100 ng/ml phorbal 12,13-dibutyrate (Sigma, St.Louis, Mo) for three days to induce differentiation into adherent macrophages.
- THP-I cells were treated with test compounds or ligands dissolved in DMSO (Sigma, D- 8779) in culture medium lacking phorbal ester. Final concentrations of DMSO did not exceed 0.3% of the media volume. Dose response effects were measured in duplicate, in the range of 0.001 to 30 micromolar concentrations and treated cells were incubated for an additional 18 hrs prior to RNA isolation. Unstimulated cells treated with vehicle were included as negative controls on each plate.
- RNA isolation and quantitation Total cellular RNA was isolated from treated cells cultured in 96-well plates using PrepStation 6100 (Applied Biosystems, Foster City, Ca), according to the manufacturer's recommendations. RNA was resuspended in ribonuclease-free water and stored at -7O 0 C prior to analysis. RNA concentrations were quantitated with RiboGreen test procedure, #R-11490 (Molecular Probes, Eugene, OR). Gene expression analysis: Gene-specific mRNA quantitation was performed by real-time PCR with the Perkin Elmer Corp. chemistry on an ABI Prism 7700 Sequence detection system (Applied Biosystems, Foster City, CA) according to the manufacturer's instructions.
- RNA samples 50-100 ng were assayed in duplicate or triplicate in 50 ul reactions using one-step RT-PCR and the standard curve method to estimate specific mRNA concentrations.
- Sequences of gene-specific primer and probe sets were designed with Primer Express Software (Applied Biosystems, Foster City, CA). The human ABCAl primer and probe sequences are: forward,
- RT and PCR reactions were performed according to PE Applied Biosystem's protocol for Taqman Gold RT-PCR or Qiagen's protocl for Quantitect probe RT-PCR. Relative levels of ABCAl mRNA are normalized using GAPDH mRNA or 18S rRNA probe/primer sets purchased commercially (Applied Biosystems, Foster City, CA). Statistics:
- the compounds of this invention can be useful in treating or inhibiting LXR mediated diseases.
- the compounds of this invention can be useful in the treatment and inhibition of atherosclerosis and atherosclerotic lesions, lowering LDL cholesterol levels, increasing HDL cholesterol levels, increasing reverse cholesterol transport, inhibiting cholesterol absorption, treatment or inhibition of cardiovascular diseases (e.g., acute coronary syndrome, restenosis), atherosclerosis, atherosclerotic lesions, type I diabetes, type II diabetes, Syndrome X, obesity, lipid disorders (e.g., dyslipidemia, hyperlipidemia, hypertriglyceridemia, hypercholesterolemia, low HDL and high LDL), cognitive disorders (e.g., Alzheimer's disease, dementia), inflammatory diseases (e.g., multiple sclerosis, rheumatoid arthritis, inflammatory bowel disease, Crohn's disease, endometriosis, LPS-induced sepsis, acute contact dermatitis of the ear
- cardiovascular diseases e.g., acute coronary syndrome,
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Biomedical Technology (AREA)
- Physical Education & Sports Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Neurosurgery (AREA)
- Cardiology (AREA)
- Dermatology (AREA)
- Neurology (AREA)
- Rheumatology (AREA)
- Hospice & Palliative Care (AREA)
- Gastroenterology & Hepatology (AREA)
- Pain & Pain Management (AREA)
- Vascular Medicine (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Child & Adolescent Psychology (AREA)
- Psychiatry (AREA)
- Toxicology (AREA)
- Urology & Nephrology (AREA)
- Biochemistry (AREA)
- Immunology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP08867591A EP2235020A1 (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-a] pyrimidine compounds |
| CA2710454A CA2710454A1 (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-a] pyrimidine compounds |
| AU2008345687A AU2008345687A1 (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-a] pyrimidine compounds |
| CN2008801273723A CN101952293A (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-A] pyrimidine compounds |
| BRPI0822239-8A BRPI0822239A2 (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-a] pyrimidine compound |
| JP2010539880A JP2011507901A (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-a] pyrimidine compounds |
| US12/809,851 US20100324073A1 (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-A] Pyrimidine Compounds |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US1586207P | 2007-12-21 | 2007-12-21 | |
| US61/015,862 | 2007-12-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009086129A1 true WO2009086129A1 (en) | 2009-07-09 |
Family
ID=40456929
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2008/087717 WO2009086129A1 (en) | 2007-12-21 | 2008-12-19 | Pyrazolo [1,5-a] pyrimidine compounds |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20100324073A1 (en) |
| EP (1) | EP2235020A1 (en) |
| JP (1) | JP2011507901A (en) |
| CN (1) | CN101952293A (en) |
| AU (1) | AU2008345687A1 (en) |
| BR (1) | BRPI0822239A2 (en) |
| CA (1) | CA2710454A1 (en) |
| WO (1) | WO2009086129A1 (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013138565A1 (en) * | 2012-03-16 | 2013-09-19 | Vitae Pharmaceuticals, Inc. | Liver x receptor modulators |
| WO2013138568A1 (en) * | 2012-03-16 | 2013-09-19 | Vitae Pharmaceuticals, Inc. | Liver x reception modulators |
| WO2014028461A2 (en) | 2012-08-13 | 2014-02-20 | The Rockefeller University | Treatment and diagnosis of melanoma |
| US9096558B2 (en) | 2010-07-09 | 2015-08-04 | Pfizer Limited | N-sulfonylbenzamide compounds |
| WO2017123568A2 (en) | 2016-01-11 | 2017-07-20 | The Rockefeller University | Methods for the treatment of myeloid derived suppressor cells related disorders |
| US10280170B2 (en) | 2014-03-27 | 2019-05-07 | Janssen Pharmaceutica Nv | Substituted 4,5,6,7-tetrahydro-pyrazolo[1,5-a]pyrazine derivatives and 5,6,7,8-tetrahydro-4H-pyrazolo[1,5-a][1,4]diazepine derivatives as Ros1 inhibitors |
| US10342795B2 (en) | 2014-03-27 | 2019-07-09 | Janssen Pharmaceutica Nv | Substituted 4,5,6,7-tetrahydro-pyrazolo[1,5-a]pyrimidine derivatives and 2,3-dihydro-1H-imidazo[1,2-b]pyrazole derivatives as ROS1 inhibitors |
| US10669296B2 (en) | 2014-01-10 | 2020-06-02 | Rgenix, Inc. | LXR agonists and uses thereof |
| US11174220B2 (en) | 2019-12-13 | 2021-11-16 | Inspirna, Inc. | Metal salts and uses thereof |
| US11214536B2 (en) | 2017-11-21 | 2022-01-04 | Inspirna, Inc. | Polymorphs and uses thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001023387A2 (en) * | 1999-09-30 | 2001-04-05 | Neurogen Corporation | CERTAIN ALKYLENE DIAMINE-SUBSTITUTED PYRAZOLO[1,5,-a]-1,5-PYRIMIDINES AND PYRAZOLO[1,5-a]-1,3,5-TRIAZINES |
| US20060030612A1 (en) * | 2004-08-03 | 2006-02-09 | Wyeth | Indazoles useful in treating cardiovascular diseases |
| US20060252757A1 (en) * | 2005-03-01 | 2006-11-09 | Baihua Hu | Cinnoline compounds |
| WO2007039439A1 (en) * | 2005-09-27 | 2007-04-12 | F.Hoffmann-La Roche Ag | Oxadiazolyl pyrazolo-pyrimidines as mglur2 antagonists |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040259948A1 (en) * | 2003-01-10 | 2004-12-23 | Peter Tontonoz | Reciprocal regulation of inflammation and lipid metabolism by liver X receptors |
| US20080070883A1 (en) * | 2006-09-19 | 2008-03-20 | Wyeth | Use of LXR modulators for the prevention and treatment of skin aging |
-
2008
- 2008-12-19 JP JP2010539880A patent/JP2011507901A/en not_active Withdrawn
- 2008-12-19 AU AU2008345687A patent/AU2008345687A1/en not_active Abandoned
- 2008-12-19 EP EP08867591A patent/EP2235020A1/en not_active Withdrawn
- 2008-12-19 CA CA2710454A patent/CA2710454A1/en not_active Abandoned
- 2008-12-19 WO PCT/US2008/087717 patent/WO2009086129A1/en active Application Filing
- 2008-12-19 BR BRPI0822239-8A patent/BRPI0822239A2/en not_active IP Right Cessation
- 2008-12-19 CN CN2008801273723A patent/CN101952293A/en active Pending
- 2008-12-19 US US12/809,851 patent/US20100324073A1/en not_active Abandoned
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001023387A2 (en) * | 1999-09-30 | 2001-04-05 | Neurogen Corporation | CERTAIN ALKYLENE DIAMINE-SUBSTITUTED PYRAZOLO[1,5,-a]-1,5-PYRIMIDINES AND PYRAZOLO[1,5-a]-1,3,5-TRIAZINES |
| US20060030612A1 (en) * | 2004-08-03 | 2006-02-09 | Wyeth | Indazoles useful in treating cardiovascular diseases |
| US20060252757A1 (en) * | 2005-03-01 | 2006-11-09 | Baihua Hu | Cinnoline compounds |
| WO2007039439A1 (en) * | 2005-09-27 | 2007-04-12 | F.Hoffmann-La Roche Ag | Oxadiazolyl pyrazolo-pyrimidines as mglur2 antagonists |
Cited By (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9096558B2 (en) | 2010-07-09 | 2015-08-04 | Pfizer Limited | N-sulfonylbenzamide compounds |
| EA026907B1 (en) * | 2012-03-16 | 2017-05-31 | Вайтаи Фармасьютиклз, Инк. | Liver x receptor modulators |
| CN104203950A (en) * | 2012-03-16 | 2014-12-10 | 生命医药公司 | Liver X reception modulators |
| AU2013232066B2 (en) * | 2012-03-16 | 2017-07-06 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| KR20140139555A (en) * | 2012-03-16 | 2014-12-05 | 비타이 파마슈티컬즈, 인코포레이티드 | Liver x receptor modulators |
| AU2013232069B2 (en) * | 2012-03-16 | 2017-07-13 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| US9006245B2 (en) | 2012-03-16 | 2015-04-14 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| US9006244B2 (en) | 2012-03-16 | 2015-04-14 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| US9073931B2 (en) | 2012-03-16 | 2015-07-07 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| WO2013138568A1 (en) * | 2012-03-16 | 2013-09-19 | Vitae Pharmaceuticals, Inc. | Liver x reception modulators |
| US9388190B2 (en) | 2012-03-16 | 2016-07-12 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| US9416135B2 (en) | 2012-03-16 | 2016-08-16 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| US9707232B2 (en) | 2012-03-16 | 2017-07-18 | Vitae Pharmaceuticals, Inc. | Liver X receptor modulators |
| EA026431B1 (en) * | 2012-03-16 | 2017-04-28 | Вайтаи Фармасьютиклз, Инк. | Liver x receptor modulators |
| WO2013138565A1 (en) * | 2012-03-16 | 2013-09-19 | Vitae Pharmaceuticals, Inc. | Liver x receptor modulators |
| KR20140135829A (en) * | 2012-03-16 | 2014-11-26 | 비타이 파마슈티컬즈, 인코포레이티드 | Liver x receptor modulators |
| KR102069913B1 (en) | 2012-03-16 | 2020-01-23 | 비타이 파마슈티컬즈, 엘엘씨 | Liver x receptor modulators |
| CN104203950B (en) * | 2012-03-16 | 2016-08-24 | 生命医药公司 | Liver X receptor modifier |
| KR102069912B1 (en) | 2012-03-16 | 2020-01-23 | 비타이 파마슈티컬즈, 엘엘씨 | Liver x receptor modulators |
| US9814715B2 (en) | 2012-03-16 | 2017-11-14 | Vitae Pharamceuticals, Inc. | Liver X receptor modulators |
| WO2014028461A2 (en) | 2012-08-13 | 2014-02-20 | The Rockefeller University | Treatment and diagnosis of melanoma |
| EP3626309A1 (en) | 2012-08-13 | 2020-03-25 | The Rockefeller University | Lxrbeta agonist for the treatment of cancer |
| US10669296B2 (en) | 2014-01-10 | 2020-06-02 | Rgenix, Inc. | LXR agonists and uses thereof |
| US10280170B2 (en) | 2014-03-27 | 2019-05-07 | Janssen Pharmaceutica Nv | Substituted 4,5,6,7-tetrahydro-pyrazolo[1,5-a]pyrazine derivatives and 5,6,7,8-tetrahydro-4H-pyrazolo[1,5-a][1,4]diazepine derivatives as Ros1 inhibitors |
| US10342795B2 (en) | 2014-03-27 | 2019-07-09 | Janssen Pharmaceutica Nv | Substituted 4,5,6,7-tetrahydro-pyrazolo[1,5-a]pyrimidine derivatives and 2,3-dihydro-1H-imidazo[1,2-b]pyrazole derivatives as ROS1 inhibitors |
| WO2017123568A2 (en) | 2016-01-11 | 2017-07-20 | The Rockefeller University | Methods for the treatment of myeloid derived suppressor cells related disorders |
| US11214536B2 (en) | 2017-11-21 | 2022-01-04 | Inspirna, Inc. | Polymorphs and uses thereof |
| US11174220B2 (en) | 2019-12-13 | 2021-11-16 | Inspirna, Inc. | Metal salts and uses thereof |
| US11459292B2 (en) | 2019-12-13 | 2022-10-04 | Inspirna, Inc. | Metal salts and uses thereof |
| US11878956B2 (en) | 2019-12-13 | 2024-01-23 | Inspirna, Inc. | Metal salts and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101952293A (en) | 2011-01-19 |
| CA2710454A1 (en) | 2009-07-09 |
| BRPI0822239A2 (en) | 2015-06-30 |
| JP2011507901A (en) | 2011-03-10 |
| EP2235020A1 (en) | 2010-10-06 |
| US20100324073A1 (en) | 2010-12-23 |
| AU2008345687A1 (en) | 2009-07-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2235019A1 (en) | Imidazo [1,2-b] pyridazine compounds as modulators of liver x receptors | |
| EP2235020A1 (en) | Pyrazolo [1,5-a] pyrimidine compounds | |
| WO2009086123A1 (en) | Imidazo [1,2-a] pyridine compounds | |
| EP2231617A1 (en) | Benzimidazole compounds | |
| JP7224102B2 (en) | Fused Bicyclic Compounds for Disease Treatment | |
| AU2008284224A1 (en) | Quinazoline compounds | |
| WO2010054229A1 (en) | Quinoxaline-based lxr modulators | |
| US20100184786A1 (en) | Polar Quinazolines | |
| US20090069373A1 (en) | Quinoline Acids |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200880127372.3 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08867591 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008345687 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2710454 Country of ref document: CA Ref document number: 2010539880 Country of ref document: JP Ref document number: MX/A/2010/007017 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2008345687 Country of ref document: AU Date of ref document: 20081219 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008867591 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12809851 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0822239 Country of ref document: BR Kind code of ref document: A2 Effective date: 20100621 |