WO2008024438A2 - 2-phenoxy pyrimidinone analogues - Google Patents
2-phenoxy pyrimidinone analogues Download PDFInfo
- Publication number
- WO2008024438A2 WO2008024438A2 PCT/US2007/018654 US2007018654W WO2008024438A2 WO 2008024438 A2 WO2008024438 A2 WO 2008024438A2 US 2007018654 W US2007018654 W US 2007018654W WO 2008024438 A2 WO2008024438 A2 WO 2008024438A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- fluorophenyl
- hydrate
- purin
- salt
- Prior art date
Links
- 0 *c1c[s]cc1 Chemical compound *c1c[s]cc1 0.000 description 6
- QGRWLZSIKBOSNE-UHFFFAOYSA-N CCOC(C1=CC=CC=CC1N)=O Chemical compound CCOC(C1=CC=CC=CC1N)=O QGRWLZSIKBOSNE-UHFFFAOYSA-N 0.000 description 1
- QEPWCMCEWOBJEY-UHFFFAOYSA-N CN1[I]=NC(C(N2c(cc3)cnc3Cl)=O)=C1N=C2Oc(cc1F)cc(F)c1F Chemical compound CN1[I]=NC(C(N2c(cc3)cnc3Cl)=O)=C1N=C2Oc(cc1F)cc(F)c1F QEPWCMCEWOBJEY-UHFFFAOYSA-N 0.000 description 1
- SACYJCHHAPQMPK-UHFFFAOYSA-N Cc1c[s]c(C(N2c(cc3)ccc3Cl)=O)c1N=C2Oc(ccc(F)c1)c1F Chemical compound Cc1c[s]c(C(N2c(cc3)ccc3Cl)=O)c1N=C2Oc(ccc(F)c1)c1F SACYJCHHAPQMPK-UHFFFAOYSA-N 0.000 description 1
- RIOAPNVMXKCLEW-UHFFFAOYSA-N Cc1c[s]c(C(N2c(cc3)ccc3F)=O)c1N=C2Oc(c(F)ccc1F)c1F Chemical compound Cc1c[s]c(C(N2c(cc3)ccc3F)=O)c1N=C2Oc(c(F)ccc1F)c1F RIOAPNVMXKCLEW-UHFFFAOYSA-N 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Oc1ccccc1 Chemical compound Oc1ccccc1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/04—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/26—Heterocyclic compounds containing purine ring systems with an oxygen, sulphur, or nitrogen atom directly attached in position 2 or 6, but not in both
- C07D473/32—Nitrogen atom
- C07D473/34—Nitrogen atom attached in position 6, e.g. adenine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/26—Heterocyclic compounds containing purine ring systems with an oxygen, sulphur, or nitrogen atom directly attached in position 2 or 6, but not in both
- C07D473/28—Oxygen atom
- C07D473/30—Oxygen atom attached in position 6, e.g. hypoxanthine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/14—Prodigestives, e.g. acids, enzymes, appetite stimulants, antidyspeptics, tonics, antiflatulents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/14—Antitussive agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/10—Drugs for disorders of the urinary system of the bladder
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/12—Drugs for genital or sexual disorders; Contraceptives for climacteric disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/04—Antipruritics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/06—Antimigraine agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
Definitions
- This invention relates generally to 2-phenoxy pyrimidinone analogues that have useful pharmacological properties.
- the invention further relates to the use of such compounds for treating conditions related to capsaicin receptor activation, for identifying other agents that bind to capsaicin receptor, and as probes for the detection and localization of capsaicin receptors.
- Pain perception, or nociception is mediated by the peripheral terminals of a group of specialized sensory neurons, termed "nociceptors.”
- nociceptors A wide variety of physical and chemical stimuli induce activation of such neurons in mammals, leading to recognition of a potentially harmful stimulus. Inappropriate or excessive activation of nociceptors, however, can result in debilitating acute or chronic pain.
- Neuropathic pain involves pain signal transmission in the absence of stimulus, and typically results from damage to the nervous system. In most instances, such pain is thought to occur because of sensitization. in the peripheral and central nervous systems following initial damage to the peripheral system (e.g., via direct injury or systemic disease). Neuropathic pain is typically burning, shooting and unrelenting in its intensity and can sometimes be more debilitating that the initial injury or disease process that induced it.
- neuropathic pain is frequently non-responsive or only partially responsive to conventional opioid analgesic regimens.
- Treatments employing the N-methyl-D-aspartate antagonist ketamine or the alpha(2)-adrenergic agonist clonidine can reduce acute or chronic pain, and permit a reduction in opioid consumption, but these agents are often poorly tolerated due to side effects.
- Topical treatment with capsaicin has been used to treat chronic and acute pain, including neuropathic pain.
- Capsaicin is a pungent substance derived from the plants of the Solanaceae family (which includes hot chili peppers) and appears to act selectively on the small diameter afferent nerve fibers (A-delta and C fibers) that are believed to mediate pain.
- the response to capsaicin is characterized by persistent activation of nociceptors in peripheral tissues, followed by eventual desensitization of peripheral nociceptors to one or more stimuli. From studies in animals, capsaicin appears to trigger C fiber membrane depolarization by opening cation selective channels for calcium and sodium.
- VRl vanilloid receptor type 1
- capsaicin receptor The first type of vanilloid receptor to be identified is known as vanilloid receptor type 1 (VRl), and the terms “VRl” and “capsaicin receptor” are used interchangeably herein to refer to rat and/or human receptors of this type, as well as mammalian homologies.
- VRl is a nonselective cation channel with a threshold for opening that is lowered in response to elevated temperatures, low pH, and capsaicin receptor agonists. Opening of the capsaicin receptor channel is generally followed by the release of inflammatory peptides from neurons expressing the receptor and other nearby neurons, increasing the pain response. After initial activation by capsaicin, the capsaicin receptor undergoes a rapid desensitization via phosphorylation by cAMP-dependent protein kinase.
- VRl agonist vanilloid compounds Because of their ability to desensitize nociceptors in peripheral tissues, VRl agonist vanilloid compounds have been used as topical anesthetics. However, agonist application may itself cause burning pain, which limits this therapeutic use. Recently, it has been reported that VRl antagonists, including certain nonvanilloid compounds, are also useful for the treatment of pain (see, e.g., PCT International Application Publication Numbers WO 02/08221, WO 03/062209, WO 04/054582, WO 04/055003, WO 04/055004, WO 04/056774, WO 05/007646, WO 05/007648, WO 05/007652, WO 05/009977, WO 05/009980, WO 05/009982, WO 05/049601, WO 05/049613, WO 06/122200 and WO 06/120481).
- the present invention provides 2-phenoxy pyrimidinone analogues of Formula A:
- Formula A as well as pharmaceutically acceptable salts, solvates ⁇ e.g., hydrates) and esters of such compounds.
- (Aj — represents a fused 5- or 6-membered heteroaryl that contains 1, 2 or 3 heteroatoms in the ring, said heteroatoms being independently chosen from O, N and S, with the remaining ring atoms being carbon, wherein the fused heteroaryl is optionally substituted; preferably the fused heteroaryl is substituted with from 0 to 3, or from 0 to 2, substituents independently chosen from amino, hydroxy, Ci-C 6 alkyl, Ci-C ⁇ hydroxyalkyl, (C 3 - C ⁇ cycloalkyl)Co-C 2 alkyl, C,-C 6 haloalkyl, Ci-C 6 alkoxy, C 2 -C 6 alkyl ether, C 1 - C 6 alkanoyloxy, Ci-C ⁇ alkylsulfonylamino, Ci-C 6 alkanonylamino, and mono- or di-(Ci-
- Ar is phenyl or a 5- or 6-membered heteroaryl, each of which is optionally substituted, and each of which is preferably substituted with from 0 to 4 or from 0 to 3 substituents that are independently chosen from halogen, cyano, amino, nitro, Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, Ci-Cehaloalkyl, C-C ⁇ hydroxyalkyl, d-C 6 alkoxy, Q-C ⁇ haloalkoxy, (C 3 -
- R 3 represents from 0 to 4, or from 0 to 3, substituents, which substituents are preferably independently chosen from halogen, hydroxy, cyano, amino, nitro, Ci-C ⁇ alkyl, C 2 - C 6 alkenyl, C 2 -C 6 alkynyl, Ci-Cehaloalkyl, C,-C 6 hydroxyalkyl, C,-C 6 alkoxy, C 1 - C 6 haloalkoxy, (C 3 -C 7 cycloalkyl)Co-C4alkyl, mono- or di-(Ci-C 6 alkyl)amino, and mono- or di-(C]-C 6 alkyl)aminosulfonyl.
- the present invention further provides 2-phenoxy pyrimidinone analogues of Formula I:
- X is N or CH that is optionally substituted with a substituent represented by R 1 ; and Ri represents from 0 to 3 substituents; which substituents are preferably independently chosen from halogen, cyano, amino, nitro, Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, Ci- Cehaloalkyl, C-C ⁇ hydroxyalkyl, Ci-C 6 alkoxy, C-C ⁇ haloalkoxy, (C 3 -C7cycloalkyl)Co- C 4 alkyl, and mono- or di-(Ci-C 6 alkyl)amino.
- compounds of Formula A and Formula I are VRl modulators and exhibit a K; of no greater than 1 micromolar, 500 nanomolar, 100 nanomolar, 50 nanomolar, 10 nanomolar or 1 nanomolar in a capsaicin receptor binding assay and/or have an ECso or IC 50 value of no greater than 1 micromolar, 500 nanomolar, 100 nanomolar, 50 nanomolar, 10 nanomolar or 1 nanomolar in an in vitro assay for determination of capsaicin receptor agonist or antagonist activity.
- VRl modulators are VRl antagonists and exhibit no detectable agonist activity in an in vitro assay of capsaicin receptor activation (e.g., the assay provided in Example 6, herein) at a concentration equal to the IC 50 , 10 times the IC 50 or 100 times the IC 50 -
- compounds provided herein are labeled with a detectable marker (e.g., radiolabeled or fluorescein conjugated).
- the present invention further provides, within other aspects, pharmaceutical compositions comprising at least one 2-phenoxy pyrimidinone analogue in combination with a physiologically acceptable carrier or excipient.
- methods are provided for reducing calcium conductance of a cellular capsaicin receptor, comprising contacting a cell (e.g., neuronal, such as cells of the central nervous system or peripheral ganglia, urothelial or lung) that expresses a capsaicin receptor with at least one VRl modulator as described herein.
- Such contact may occur in vivo or in vitro and is generally performed using a concentration of VRl modulator that is sufficient to alter the binding of vanilloid ligand to VRl in vitro (using the assay provided in Example 5) and/or VRl-mediated signal transduction (using an assay provided in Example 6).
- Methods are further provided for inhibiting binding of vanilloid ligand to a capsaicin receptor.
- the inhibition takes place in vitro.
- Such methods comprise contacting a capsaicin receptor with at least one VRl modulator as described herein, under conditions and in an amount or concentration sufficient to detectably inhibit vanilloid ligand binding to the capsaicin receptor.
- the capsaicin receptor is in a patient.
- Such methods comprise contacting cells expressing a capsaicin receptor in a patient with at least one VRl modulator as described herein in an amount or concentration that would be sufficient to detectably inhibit vanilloid ligand binding to cells expressing a cloned capsaicin receptor in vitro.
- the present invention further provides methods for treating a condition responsive to capsaicin receptor modulation in a patient, comprising administering to the patient a therapeutically effective amount of at least one VRl modulator as described herein.
- methods for treating pain in a patient, comprising administering to a patient suffering from (or at risk for) pain a therapeutically effective amount of at least one VRl modulator as described herein.
- Methods are further provided for treating itch, urinary incontinence, overactive bladder, menopause symptoms, cough and/or hiccup in a patient, comprising administering to a patient suffering from (or at risk for) one or more of the foregoing conditions a therapeutically effective amount of at least one VRl modulator as described herein.
- methods are provided for treating menopause symptoms in a patient, comprising administering to a patient suffering from (or at risk for) such symptoms a therapeutically effective amount of at least one VRl modulator as described herein.
- the present invention further provides methods for promoting weight loss in an obese patient, comprising administering to an obese patient a therapeutically effective amount of at least one VRl modulator as described herein.
- Methods are further provided for identifying an agent that binds to capsaicin receptor, comprising: (a) contacting capsaicin receptor with a labeled compound as described herein under conditions that permit binding of the compound to capsaicin receptor, thereby generating bound, labeled compound; (b) detecting a signal that corresponds to the amount of bound, labeled compound in the absence of test agent; (c) contacting the bound, labeled compound with a test agent; (d) detecting a signal that corresponds to the amount of bound labeled compound in the presence of test agent; and (e) detecting a decrease in signal detected in step (d), as compared to the signal detected in step (b).
- the present invention provides methods for determining the presence or absence of capsaicin receptor in a sample, comprising: (a) contacting a sample with a compound as described herein under conditions that permit binding of the compound to capsaicin receptor; and (b) detecting a signal indicative of a level of the compound bound to capsaicin receptor.
- the present invention also provides packaged pharmaceutical preparations, comprising: (a) a pharmaceutical composition as described herein in a container; and (b) instructions for using the composition to treat one or more conditions responsive to capsaicin receptor modulation, such as pain, itch, urinary incontinence, overactive bladder, menopause symptoms, cough, hiccup and/or obesity.
- the present invention provides methods for preparing the compounds disclosed herein, including the intermediates.
- the present invention provides 2-phenoxy pyrimidinone analogues. Such compounds may be used in vitro or in vivo, to modulate capsaicin receptor activity in a variety of contexts.
- each variable within such a formula is defined independently of any other variable, and any variable that occurs more than one time in a formula is defined independently at each occurrence.
- a “pharmaceutically acceptable salt” of a compound recited herein is an acid or base salt that is suitable for use in contact with the tissues of human beings or animals without excessive toxicity or carcinogenicity, and preferably without irritation, allergic response, or other problem or complication.
- Such salts include mineral and organic acid salts of basic residues such as amines, as well as alkali or organic salts of acidic residues such as carboxylic acids.
- Specific pharmaceutically acceptable anions for use in salt formation include, but are not limited to, acetate, 2-acetoxybenzoate, ascorbate, benzoate, bicarbonate, bromide, calcium edetate, carbonate, chloride, citrate, dihydrochloride, diphosphate, ditartrate, edetate, estolate (ethylsuccinate), formate, fumarate, gluceptate, gluconate, glutamate, glycolate, glycollylarsanilate, hexylresorcinate, hydrabamine, hydrobromide, hydrochloride, hydroiodide, hydroxymaleate, hydroxynaphthoate, iodide, isethionate, lactate, lactobionate, malate, maleate, mandelate, methylbromide, methylnitrate, methylsulfate, mucate, napsylate, nitrate, pamoate, pantothenate, phenylacetate
- pharmaceutically acceptable cations for use in salt formation include, but are not limited to ammonium, benzathine, chloroprocaine, choline, diethanolamine, ethylenediamine, meglumine, procaine, and metals such as aluminum, calcium, lithium, magnesium, potassium, sodium and zinc.
- a pharmaceutically acceptable acid or base salt can be synthesized from a parent compound that contains a basic or acidic moiety by any conventional chemical method.
- such salts can be prepared by reacting the free acid or base forms of these compounds with a stoichiometric amount of the appropriate base or acid in water or in an organic solvent, or in a mixture of the two; generally, the use of nonaqueous media, such as ether, ethyl acetate, ethanol, methanol, isopropanol or acetonitrile, is preferred.
- nonaqueous media such as ether, ethyl acetate, ethanol, methanol, isopropanol or acetonitrile
- each compound provided herein may, but need not, be formulated as a solvate (e.g., hydrate) or non-covalent complex.
- the various crystal forms and polymorphs are within the scope of the present invention.
- prodrugs of the compounds of the recited Formulas are also provided herein.
- a "prodrug” is a compound that may not fully satisfy the structural requirements of the compounds provided herein, but is modified in vivo, following administration to a patient, to produce a compound a formula provided herein.
- a prodrug may be an acylated derivative of a compound as provided herein.
- Prodrugs include compounds wherein hydroxy, amine or sulfhydryl groups are bonded to any group that, when administered to a mammalian subject, cleaves to form a free hydroxy, amino, or sulfhydryl group, respectively.
- Examples of prodrugs include, but are not limited to, acetate, formate and benzoate derivatives of alcohol and amine functional groups within the compounds provided herein.
- Prodrugs of the compounds provided herein may be prepared by modifying functional groups present in the compounds in such a way that the modifications are cleaved in vivo to yield the parent compounds.
- alkyl refers to a straight or branched chain saturated aliphatic hydrocarbon.
- Alkyl groups include groups having from 1 to 8 carbon atoms (Ci - Cgalkyl), from 1 to 6 carbon atoms (Ci-C 6 alkyl) and from 1 to 4 carbon atoms (Ci-C 4 alkyl), such as methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-b ⁇ tyl, pentyl, 2-pentyl, isopentyl, neopentyl, hexyl, 2-hexyl, 3-hexyl and 3-methyl ⁇ entyl.
- C 0 -C n alkyl refers to a single covalent bond (C 0 ) or an alkyl group having from 1 to n carbon atoms; for example "Co-C 4 alkyl” refers to a single covalent bond or a Ci-C 4 alkyl group. In some instances, a substituent of an alkyl group is specifically indicated. For example, “hydroxyalkyl” refers to an alkyl group substituted with at least one hydroxy substituent.
- Alkylene refers to a divalent alkyl group, as defined above. Ci-C 2 alkylene is methylene or ethylene; C 0 -C 4 alkylene is a single covalent bond or an alkylene group having 1, 2, 3 or carbon atoms; C 0 -C 2 alkylene is a single covalent bond or an alkylene group having 1 or 2 carbon atoms.
- AlkenyP refers to straight or branched chain alkene groups, which comprise at least one unsaturated carbon-carbon double bond.
- Alkenyl groups include C 2 -C 8 alkenyl, C 2 - C 6 alkenyl and C 2 -C 4 alkenyl groups, which have from 2 to 8, 2 to 6 or 2 to 4 carbon atoms, respectively, such as ethenyl, allyl or isopropenyl.
- Alkynyl refers to straight or branched chain alkyne groups, which have one or more unsaturated carbon-carbon bonds, at least one of which is a triple bond.
- Alkynyl groups include C 2 -Cgalkynyl, C 2 -C 6 alkynyl and C 2 - C 4 alkynyl groups, which have from 2 to 8, 2 to 6 or 2 to 4 carbon atoms, respectively.
- a “cycloalkyl” is a group that comprises one or more saturated and/or partially saturated rings in which all ring members are carbon, such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, adamantyl, and partially saturated variants of the foregoing, such as cyclohexenyl. Cycloalkyl groups do not comprise an aromatic ring or a heterocyclic ring. Certain cycloalkyl groups are C 3 -C 7 CyClOaIlCyI, in which the cycloalkyl group contains a single ring having from 3 to 7 ring members, all of which are carbon.
- a "(C 3 -C 8 cycloalkyl)Co-C 4 alkyl” is a C 3 -C 8 cycloalkyl group linked via a single covalent bond or a Ci-C 4 alkylene group.
- alkoxy is meant an alkyl group as described above attached via an oxygen bridge.
- Alkoxy groups include Ci-C 6 alkoxy and C ) -C 4 alkoxy groups, which have from 1 to 6 or from 1 to 4 carbon atoms, respectively.
- Methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, sec-butoxy, rert-butoxy, n-pentoxy, 2-pentoxy, 3-pentoxy, isopentoxy, neopentoxy, hexoxy, 2-hexoxy, 3-hexoxy, and 3-methylpentoxy are representative alkoxy groups.
- alkylthio refers to an alkyl group as described above attached via a sulfur bridge.
- Alkyl ether refers to a linear or branched ether substituent (i.e., an alkyl group that is substituted with an alkoxy group).
- Alkyl ether groups include C 2 -C 8 alkyl ether, C 2 -C 6 alkyl ether and C 2 -C 4 alkyl ether groups, which have 2 to 8, 6 or 4 carbon atoms, respectively.
- a C 2 alkyl ether has the structure -CH 2 -CMTH 3 .
- Alkanoyl groups have the indicated number of carbon atoms, with the carbon of the keto group being included in the numbered carbon atoms.
- Q-C ⁇ alkanoyl groups contain from 1 to 6 carbon atoms.
- Alkanoyloxy groups include, for example, Q-C ⁇ alkanoyloxy groups, which have from one to six carbon atoms.
- Alkanoylamino groups include, for example, Ci-C 6 alkanoylamino groups, which have from 1 to 6 carbon atoms in the "alkyl" portion (i.e., the carbon of the keto bridge is not included in the indicated number of carbon atoms).
- Alkylsulfonyl refers to groups of the formula — (SO 2 )-alkyl, in which the sulfur atom is the point of attachment. Alkylsulfonyl groups include Ci-C 6 alkylsulfonyl and Q- C 4 alkylsulfonyl groups, which have from 1 to 6 or from 1 to 4 carbon atoms, respectively. Methylsulfonyl is one representative alkylsulfonyl group.
- Alkylsulfonylamino refers to an alkylsulfonyl group attached through an amino linker (i.e., a group having the general structure -N(R)-(SO 2 )-alkyl), in which R is hydrogen or Ci-Cgalkyl).
- Alkylsulfonylamino groups include, for example, C
- Aminosulfonyl refers to groups of the formula -(SO 2 )-NH 2 , in which the sulfur atom is the point of attachment.
- di-(Ci-C 6 alkyl)aminosulfonyl refers to groups that satisfy the formula — (SO 2 )-NR 2 , in which the sulfur atom is the point of attachment, and in which one R is Ci-C 6 alkyl and the other R is hydrogen or an independently chosen C
- Alkylamino refers to a secondary or tertiary amine that has the general structure -
- each alkyl is selected independently from alkyl, cycloalkyl and (cycloalkyl)alkyl groups.
- groups include, for example, mono- and di- (Ci-C 6 alkyl)amino groups, in which each Ci-C 6 alkyl may be the same or different.
- Alkylaminoalkyl refers to an alkylamino group linked via an alkylene group (i.e., a group having the general structure —alkylene— NH-alkyl or —alkylene— N(alkyl)(alkyl)) in which each alkyl is selected independently from alkyl, cycloalkyl and (cycloalkyl)alkyl groups.
- Alkylaminoalkyl groups include, for example, mono- and
- alkyl as used in the terms “alkylamino” and “alkylaminoalkyl” differs from the definition of "alkyl” used for all other alkyl-containing groups, in the inclusion of cycloalkyl and (cycloalkyl)alkyl groups (e.g., (C 3 -C ⁇ cycloalkyl)Co- C 4 alkyl).
- halogen refers to fluorine, chlorine, bromine or iodine.
- haloalkyl is an alkyl group that is substituted with 1 or more independently chosen halogens (e.g., "Ci-C ⁇ haloalkyl” groups have from 1 to 6 carbon atoms).
- haloalkyl groups include, but are not limited to, mono-, di- or tri-fluoromethyl; mono-, di- or tri-chloromethyl; mono-, di-, tri-, tetra- or penta-fluoroethyl; mono-, di-, tri-, tetra- or penta- chloroethyl; and 1,2,2,2-tetrafluoro-l-trifluoromethyl-ethyl.
- Typical haloalkyl groups are trifluoromethyl and difluoromethyl.
- haloalkoxy refers to a haloalkyl group as defined above that is linked via an oxygen bridge.
- a dash (“-") that is not between two letters or symbols is used to indicate a point of attachment for a substituent.
- -CONH 2 is attached through the carbon atom.
- a "heteroaryl” is an aromatic group in which at least one aromatic ring comprises at least one heteroatom selected from N, O and S. Heteroaryls include, for example, 5- and 6- membered heteroaryls such as imidazole, furan, furazan, isothiazole, isoxazole, oxadiazole, oxazole, pyrazine, pyrazole, pyridazine, pyridine, pyrimidine, tetrazole, thiazole and thiophene.
- a “substituent,” as used herein, refers to a molecular moiety that is covalently bonded to an atom within a molecule of interest.
- a ring substituent may be a moiety such as a halogen, alkyl group, haloalkyl group or other group that is covalently bonded to an atom (preferably a carbon or nitrogen atom) that is a ring member.
- Substituents of aromatic groups are generally covalently bonded to a ring carbon atom.
- substitution refers to replacing a hydrogen atom in a molecular structure with a substituent, such that the valence on the designated atom is not exceeded, and such that a chemically stable compound (i.e., a compound that can be isolated, characterized, and tested for biological activity) results from the substitution.
- Groups that are "optionally substituted” are unsubstituted or are substituted by other than hydrogen at one or more available positions, typically 1, 2, 3, 4 or 5 positions, by one or more suitable groups (which may be the same or different).
- Optional substitution is also indicated by the phrase "substituted with from 0 to X substituents," where X is the maximum number of possible substituents.
- optionally substituted groups are substituted with from 0 to 2, 3 or 4 independently selected substituents (i.e., are unsubstituted or substituted with up to the recited maximum number of substituents).
- Other optionally substituted groups are substituted with at least one substituent (e.g., substituted with from 1 to 2, 3 or 4 independently selected substituents).
- VRl and “capsaicin receptor” are used interchangeably herein to refer to a type 1 vanilloid receptor. Unless otherwise specified, these terms encompass both rat and human VRl receptors (e.g., GenBank Accession Numbers AF327067, AJ277028 and NM O 18727; sequences of certain human VRl cDNAs and the encoded amino acid sequences are provided in U.S. Patent No. 6,482,611), as well as homologues thereof found in other species.
- a “VRl modulator,” also referred to herein as a “modulator,” is a compound that modulates VRl activation and/or VRl-mediated signal transduction.
- VRl modulators specifically provided herein are compounds of Formula A, and pharmaceutically acceptable salts, hydrates and esters thereof. Certain preferred VRl modulators are not vanilloids.
- a VRl modulator may be a VRl agonist or antagonist.
- Certain modulators bind to VRl with a Ki that is less than 1 micromolar, preferably less than 500 nanomolar, 100 nanomolar, 10 nanomolar or 1 nanomolar.
- a representative assay for determining K ; at VRl is provided in Example 5, herein.
- a modulator is considered an "antagonist" if it detectably inhibits vanilloid ligand binding to VRl and/or VRl-mediated signal transduction (using, for example, the representative assay provided in Example 6); in general, such an antagonist inhibits VRl activation with a IC S0 value of less than 1 micromolar, preferably less than 500 nanomolar, and more preferably less than 100 nanomolar, 10 nanomolar or 1 nanomolar within the assay provided in Example 6.
- VRl antagonists include neutral antagonists and inverse agonists.
- An "inverse agonist" of VRl is a compound that reduces the activity of VRl below its basal activity level in the absence of added vanilloid ligand. Inverse agonists of VRl may also inhibit the activity of vanilloid ligand at VRl and/or binding of vanilloid ligand to VRl .
- the basal activity of VRl, as well as the reduction in VRl activity due to the presence of VRl antagonist, may be determined from a calcium mobilization assay, such as the assay of Example 6.
- a "neutral antagonist" of VRl is a compound that inhibits the activity of vanilloid ligand at VRl, but does not significantly change the basal activity of the receptor ⁇ i.e., within a calcium mobilization assay as described in Example 6 performed in the absence of vanilloid ligand, VRl activity is reduced by no more than 10%, preferably by no more than 5%, and more preferably by no more than 2%; most preferably, there is no detectable reduction in activity).
- Neutral antagonists of VRl may inhibit the binding of vanilloid ligand to VRl.
- Capsaicin receptor agonist or “VRl agonist” is a compound that elevates the activity of the receptor above the basal activity level of the receptor (i.e., enhances VRl activation and/or VRl -mediated signal transduction). Capsaicin receptor agonist activity may be identified using the representative assay provided in Example 6. In general, such an agonist has an EC 50 value of less than 1 micromolar, preferably less than 500 nanomolar, and more preferably less than 100 nanomolar or 10 nanomolar within the assay provided in Example 6.
- a “vanilloid” is any compound that comprises a phenyl ring with two oxygen atoms bound to adjacent ring carbon atoms (one of which carbon atom is located para to the point of attachment of a third moiety that is bound to the phenyl ring).
- Capsaicin is a representative vanilloid.
- a “vanilloid ligand” is a vanilloid that binds to VRl with a K; (determined as described herein) that is no greater than 10 ⁇ M.
- Vanilloid ligand agonists include capsaicin, olvanil, N-arachidonoyl-dopamine and resiniferatoxin (RTX).
- Vanilloid ligand antagonists include capsazepine and iodo-resiniferatoxin.
- a “therapeutically effective amount” is an amount that, upon administration to a patient, results in a discernible patient benefit (e.g., provides detectable relief from at least one condition being treated). Such relief may be detected using any appropriate criteria, including alleviation of one or more symptoms such as pain.
- a therapeutically effective amount or dose generally results in a concentration of compound in a body fluid (such as blood, plasma, serum, CSF, synovial fluid, lymph, cellular interstitial fluid, tears or urine) that is sufficient to alter the binding of vanilloid ligand to VRl in vitro (using the assay provided in Example 5) and/or VRl -mediated signal transduction (using an assay provided in Example 6). It will be apparent that the discernible patient benefit may be apparent after administration of a single dose, or may become apparent following repeated administration of the therapeutically effective dose according to a predetermined regimen, depending upon the indication for which the compound is administered.
- statically significant results varying from control at the p ⁇ 0.1 level of significance as measured using a standard parametric assay of statistical significance such as a student's T test.
- a "patient” is any individual treated with a compound provided herein. Patients include humans, as well as other animals such as companion animals (e.g., dogs and cats) and livestock. Patients may be experiencing one or more symptoms of a condition responsive to capsaicin receptor modulation (e.g., pain, exposure to vanilloid ligand, itch, urinary incontinence, overactive bladder, menopause symptoms, respiratory disorders, cough and/or hiccup), or may be free of such symptom(s) (i.e., treatment may be prophylactic in a patient considered at risk for the development of such symptoms).
- capsaicin receptor modulation e.g., pain, exposure to vanilloid ligand, itch, urinary incontinence, overactive bladder, menopause symptoms, respiratory disorders, cough and/or hiccup
- treatment may be prophylactic in a patient considered at risk for the development of such symptoms.
- VRl modulators that may be used in a variety of contexts, including in the treatment of pain (e.g., neuropathic or peripheral nerve- mediated pain); exposure to capsaicin; exposure to acid, heat, light, tear gas, air pollutants (such as, for example, tobacco smoke), infectious agents (including viruses, bacteria and yeast), pepper spray or related agents; respiratory conditions such as asthma or chronic obstructive pulmonary disease; itch; urinary incontinence or overactive bladder; menopause symptoms; cough or hiccup; and/or obesity.
- pain e.g., neuropathic or peripheral nerve- mediated pain
- capsaicin e.g., neuropathic or peripheral nerve- mediated pain
- respiratory conditions such as asthma or chronic obstructive pulmonary disease
- itch urinary incontinence or overactive bladder
- menopause symptoms e.g
- Such compounds may also be used within in vitro assays (e.g., assays for receptor activity), as probes for detection and localization of VRl and as standards in ligand binding and VRl -mediated signal transduction assays. It has been found, within the context of the present invention, that the 2-phenoxy pyrimidinone analogues provided herein exhibit an unexpectedly high VRl -modulating activity due, at least in part, to the phenoxy moiety of Formula A and Formula I.
- ⁇ is substituted with from 0 to 2 substituents independently chosen from Ci-C 6 alkyl, (C 3 -
- C7cycloalkyl)Co-C 2 alkyl and Q-C ⁇ haloalkyl is substituted with from O to 2 substituents independently chosen from Ci-C 4 alkyl, (C 3 - C 5 cycloalkyl)C 0 -C 2 alkyl and Q-C ⁇ aloalkyl.
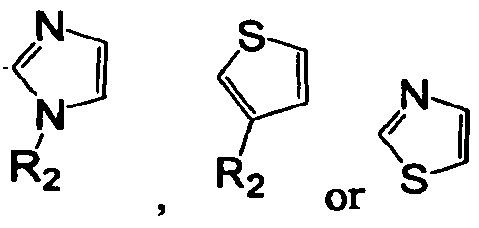
- — is a 5-membered heteroaryl represented by any of the formulae:
- R' 4 is hydrogen, C,-C 4 alkyl, (C 3 -C 3 cycloalkyl)Co-C 2 alkyl, C,-C 4 haloalkyl, Ci-C 4 hydroxyalkyl, C ⁇ -C 4 alkoxy, Ci-C 4 alkanoylamino or Ci-C 4 alkylsulfonylamino.
- R 2 is, for example, hydrogen, cyano, aryl, heteroaryl, halogen, Ci-
- ⁇ ⁇ -""J ⁇ is 0* 2 or ° . It will be apparent that the orientation of such moieties is intended to
- ⁇ - ⁇ is a 6-membered heteroaryl that is substituted with from 0 to 3 substituents independently chosen from hydroxy, Ci-C 6 alkyl, (C 3 - C 7 cycloalkyl)C 0 -C 2 alkyl, Ci-C 6 haloalkyl, C t -C 6 hydroxyalkyl, Ci-C 6 alkoxy, mono-(Ci- C 6 alkyl)amino, Ci-C ⁇ alkanoylamino or Ci-C ⁇ alkylsulfonylamino.
- R 4 represents from 0 to 3, preferably from 1 to 3, substituents independently chosen from hydroxy, Ci-C 4 alkyl, (C 3 -C 5 cycloalkyl)C 0 -C 2 alkyl, Ci-C 4 haloalkyl, Ci-C 4 hydroxyalkyl, Ci-C 4 alkoxy, mono-(Ci-C 4 alkyl)amino, Q- C4alkanoylamino or Ci-C 4 alkylsulfonylamino.
- the variable R 1 within certain embodiments, represents from 0 to 3, preferably from 1 to 3, substituents independently chosen from halogen, cyano, d-C 4 alkyl and Q- C 4 haloalkyl.
- Ri represents exactly one substituent ⁇ e.g., at the para position of the ring Ar) within certain such compounds.
- at least one substituent represented by Ri is a halogen or CN; such substituent is located at the para position of a 6-membered Ar moiety within certain such compounds.
- the para position refers to the position para to the point of attachment of the Ar moiety to the pyrimidinone core; that is, the 4-position of the phenyl ring that results when X is CH, and the 6-position of the pyridin-3-yl ring that results when X is N.
- R 3 represents from 1 to 3 substituents independently chosen from halogen, cyano, Ci-C 4 alkyl, Ci-C 4 haloalkyl and Ci-C 4 alkoxy.
- compounds of Formula I further satisfy one of Formulas II- VII:
- R 2 is hydrogen, Ci-C 4 alkyl, C
- R 3 represents from 1 to 3 substituents independently chosen from halogen, cyano, Ci-C 4 alkyl, Ci -G»haloalkyl and C 1 -CaIkOXy
- R' 4 is hydrogen, C 1 -C 4 alkyl, (C 3 -C 5 cycloalkyl)Co-C 2 alkyl, Q-Qhaloalkyl, C 1 - C 4 hydroxyalkyl, Ci-C 4 alkoxy, Ci-C 4 alkanoylamino or Ci-C 4 alkylsulfonylamino
- R 5 is halogen or CN.
- R' 4 is H (i.e., such compounds further satisfy one of Formulas III -Vila: Formula IIa Formula HIa Formula IVa
- Formula VUI Formula DC in which R 3 represents from 1 to 3 substituents independently chosen from halogen, cyano, Ci-C 4 alkyl, Ci-G t haloalkyl and C
- 2-phenoxy pyrimidinone analogues and intermediates include, but are not limited to, those specifically described in Examples 1-3. It will be apparent that the specific compounds recited herein are representative only, and are not intended to limit the scope of the present invention. Further, as noted above, all compounds of the present invention may be present as a free acid or base, or as a pharmaceutically acceptable salt. In addition, other forms such as hydrates and prodrugs of such compounds are specifically contemplated by the present invention.
- 2-phenoxy pyrimidinone analogues provided herein detectably alter (modulate) VRl activity, as determined using an in vitro VRl functional assay such as a calcium mobilization assay.
- a VRl ligand binding assay may be used as an initial screen for such activity.
- References herein to a "VRl ligand binding assay” are intended to refer to a standard in vitro receptor binding assay such as that provided in Example 5, and a "calcium mobilization assay” (also referred to herein as a “signal transduction assay”) may be performed as described in Example 6.
- a competition assay may be performed in which a VRl preparation is incubated with labeled (e.g., 125 I or 3 H) compound that binds to VRl (e.g., a capsaicin receptor agonist such as RTX) and unlabeled test compound.
- the VRl used is preferably mammalian VRl, more preferably human or rat VRl .
- the receptor may be recombinantly expressed or naturally expressed.
- the VRl preparation may be, for example, a membrane preparation from HEK293 or CHO cells that recombinantly express human VRl .
- Incubation with a compound that detectably modulates vanilloid ligand binding to VRl results in a decrease or increase in the amount of label bound to the VRl preparation, relative to the amount of label bound in the absence of the compound. This decrease or increase may be used to determine the K 4 at VRl as described herein.
- compounds that decrease the amount of label bound to the VRl preparation within such an assay are preferred.
- VRl modulators detectably modulate VRl activity at nanomolar (i.e., submicromolar) concentrations, at subnanomolar concentrations, or at concentrations below 100 picomolar, 20 picomolar, 10 picomolar or 5 picomolar.
- compounds that are VRl antagonists are preferred within certain embodiments.
- IC 50 values for such compounds may be determined using a standard in vitro VRl-mediated calcium mobilization assay, as provided in Example 6.
- cells expressing capsaicin receptor are contacted with a compound of interest and with an indicator of intracellular calcium concentration (e.g., a membrane permeable calcium sensitivity dye such as Fluo-3 or Fura-2 (Molecular Probes, Eugene, OR), each of which produce a fluorescent signal when bound to Ca +* ).
- an indicator of intracellular calcium concentration e.g., a membrane permeable calcium sensitivity dye such as Fluo-3 or Fura-2 (Molecular Probes, Eugene, OR)
- Such contact is preferably carried out by one or more incubations of the cells in buffer or culture medium comprising either or both of the compound and the indicator in solution. Contact is maintained for an amount of time sufficient to allow the dye to enter the cells (e.g., 1-2 hours).
- a vanilloid receptor agonist e.g., capsaicin, RTX or olvanil
- a fluorescence response is measured.
- agonist-contacted cells are contacted with a compound that is a VRl antagonist the fluorescence response is generally reduced by at least 20%, preferably at least 50% and more preferably at least 80%, as compared to cells that are contacted with the agonist in the absence of test compound.
- the IC 50 for VRl antagonists provided herein is preferably less than 1 micromolar, less than 100 nM, less than 10 nM or less than 1 nM.
- VRl antagonists provided herein exhibit no detectable agonist activity an in vitro assay of capsaicin receptor agonism at a concentration of compound equal to the IC 50 . Certain such antagonists exhibit no detectable agonist activity an in vitro assay of capsaicin receptor agonism at a concentration of compound that is 100- fold higher than the ICs 0 . In other embodiments, compounds that are capsaicin receptor agonists are preferred. Capsaicin receptor agonist activity may generally be determined as described in Example 6. When cells are contacted with 1 micromolar of a compound that is a VRl agonist, the fluorescence response is generally increased by an amount that is at least 30% of the increase observed when cells are contacted with 100 nM capsaicin.
- the EC 50 for VRl agonists provided herein is preferably less than 1 micromolar, less than 100 nM or less than 10 nM.
- VRl modulating activity may also, or alternatively, be assessed using a cultured dorsal root ganglion assay as provided in Example 7 and/or an in vivo pain relief assay as provided in Example 8.
- VRl modulators provided herein preferably have a statistically significant specific effect on VRl activity within one or more functional assays provided herein.
- VRl modulators provided herein do not substantially modulate ligand binding to other cell surface receptors, such as EGF receptor tyrosine kinase or the nicotinic acetylcholine receptor.
- such modulators do not substantially inhibit activity of a cell surface receptor such as the human epidermal growth factor (EGF) receptor tyrosine kinase or the nicotinic acetylcholine receptor (e.g., the IC 50 or IC 40 at such a receptor is preferably greater than 1 micromolar, and most preferably greater than 10 micromolar).
- EGF human epidermal growth factor
- a modulator does not detectably inhibit EGF receptor activity or nicotinic acetylcholine receptor activity at a concentration of 0.5 micromolar, 1 micromolar or more preferably 10 micromolar.
- Assays for determining cell surface receptor activity are commercially available, and include the tyrosine kinase assay kits available from Panvera (Madison, WI).
- preferred VRl modulators are non-sedating.
- a dose of VRl modulator that is twice the minimum dose sufficient to provide analgesia in an animal model for determining pain relief causes only transient (i.e., lasting for no more than '/_ the time that pain relief lasts) or preferably no statistically significant sedation in an animal model assay of sedation (using the method described by Fitzgerald et al. (1988) Toxicology 49(2-3):433-9).
- a dose that is five times the minimum dose sufficient to provide analgesia does not produce statistically significant sedation.
- a VRl modulator provided herein does not produce sedation at intravenous doses of less than 25 mg/kg (preferably less than 10 mg/kg) or at oral doses of less than 140 mg/kg (preferably less than 50 mg/kg, more preferably less than 30 mg/kg).
- compounds provided herein may be evaluated for certain pharmacological properties including, but not limited to, oral bioavailability (preferred compounds are orally bioavailable to an extent allowing for therapeutically effective concentrations of the compound to be achieved at oral doses of less than 140 mg/kg, preferably less than 50 mg/kg, more preferably less than 30 mg/kg, even more preferably less than 10 mg/kg, still more preferably less than 1 mg/kg and most preferably less than 0.1 mg/kg), toxicity (a preferred compound is nontoxic when a therapeutically effective amount is administered to a subject), side effects (a preferred compound produces side effects comparable to placebo when a therapeutically effective amount of the compound is administered to a subject), serum protein binding and in vitro and in vivo half-life (a preferred compound exhibits an in vivo half-life allowing for Q.I.D.
- assays used to predict bioavailability include transport across human intestinal cell monolayers, including Caco-2 cell monolayers.
- Penetration of the blood brain barrier of a compound in humans may be predicted from the brain levels of the compound in laboratory animals given the compound ⁇ e.g., intravenously).
- Serum protein binding may be predicted from albumin binding assays.
- Compound half-life is inversely proportional to the frequency of dosage of a compound.
- In vitro half-lives of compounds may be predicted from assays of microsomal half-life as described, for example, within Example 7 of U.S. Patent Application Publication Number 2005/0070547.
- nontoxic shall be understood in a relative sense and is intended to refer to any substance that has been approved by the United States Food and Drug Administration (“FDA”) for administration to mammals (preferably humans) or, in keeping with established criteria, is susceptible to approval by the FDA for administration to mammals (preferably humans).
- FDA United States Food and Drug Administration
- a highly preferred nontoxic compound generally satisfies one or more of the following criteria: (1) does not substantially inhibit cellular ATP production; (2) does not significantly prolong heart QT intervals; (3) does not cause substantial liver enlargement, or (4) does not cause substantial release of liver enzymes.
- a compound that does not substantially inhibit cellular ATP production is a compound that satisfies the criteria set forth in Example 8 of U.S. Patent Application Publication Number 2005/0070547.
- cells treated as described therein with 100 ⁇ M of such a compound exhibit ATP levels that are at least 50% of the ATP levels detected in untreated cells.
- such cells exhibit ATP levels that are at least 80% of the ATP levels detected in untreated cells.
- a compound that does not significantly prolong heart QT intervals is a compound that does not result in a statistically significant prolongation of heart QT intervals (as determined by electrocardiography) in guinea pigs, minipigs or dogs upon administration of a dose that yields a serum concentration equal to the EC 50 or IC 50 for the compound.
- a dose of 0.01, 0.05, 0.1, 0.5, 1, 5, 10, 40 or 50 mg/kg administered parenterally or orally does not result in a statistically significant prolongation of heart QT intervals.
- a compound does not cause substantial liver enlargement if daily treatment of laboratory rodents (e.g., mice or rats) for 5-10 days with a dose that yields a serum concentration equal to the EC 50 or IC 50 for the compound results in an increase in liver to body weight ratio that is no more than 100% over matched controls. In more highly preferred embodiments, such doses do not cause liver enlargement of more than 75% or 50% over matched controls. If non-rodent mammals (e.g., dogs) are used, such doses should not result in an increase of liver to body weight ratio of more than 50%, preferably not more than 25%, and more preferably not more than 10% over matched untreated controls. Preferred doses within such assays include 0.01, 0.05. 0.1, 0.5, 1, 5, 10, 40 or 50 mg/kg administered parenterally or orally.
- a compound does not promote substantial release of liver enzymes if administration of twice the minimum dose that yields a serum concentration equal to the EC 50 or IC 50 at VRl for the compound does not elevate serum levels of ALT, LDH or AST in laboratory animals (e.g.. rodents) by more than 100% over matched mock-treated controls. In more highly preferred embodiments, such doses do not elevate such serum levels by more than 75% or 50% over matched controls.
- a compound does not promote substantial release of liver enzymes if, in an in vitro hepatocyte assay, concentrations (in culture media or other such solutions that are contacted and incubated with hepatocytes in vitro) that are equal to the EC 50 or IC 50 for the compound do not cause detectable release of any of such liver enzymes into culture medium above baseline levels seen in media from matched mock-treated control cells. In more highly preferred embodiments, there is no detectable release of any of such liver enzymes into culture medium above baseline levels when such compound concentrations are five-fold, and preferably ten-fold the EC S0 or IC S0 for the compound.
- certain preferred compounds do not inhibit or induce microsomal cytochrome P450 enzyme activities, such as CYP1A2 activity, CYP2A6 activity, CYP2C9 activity, CYP2C19 activity, CYP2D6 activity, CYP2E1 activity or CYP3A4 activity at a concentration equal to the EC S o or IC 30 at VRl for the compound.
- microsomal cytochrome P450 enzyme activities such as CYP1A2 activity, CYP2A6 activity, CYP2C9 activity, CYP2C19 activity, CYP2D6 activity, CYP2E1 activity or CYP3A4 activity at a concentration equal to the EC S o or IC 30 at VRl for the compound.
- Certain preferred compounds are not clastogenic (e.g., as determined using a mouse erythrocyte precursor cell micronucleus assay, an Ames micronucleus assay, a spiral micronucleus assay or the like) at a concentration equal the EC 50 or IC50 for the compound.
- certain preferred compounds do not induce sister chromatid exchange (e.g., in Chinese hamster ovary cells) at such concentrations.
- VRl modulators provided herein may be isotopically-labeled or radiolabeled.
- compounds may have one or more atoms replaced by an atom of the same element having an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
- isotopes that can be present in the compounds provided herein include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, fluorine and chlorine, such as 2 H, 3 H, ' 1 C, ' 3 C, 14 C, 15 N, 18 O, ' 7 O, 31 P, 32 P, 35 S, 18 F and 36 Cl.
- substitution with heavy isotopes such as deuterium (i.e., 2 H) can afford certain therapeutic advantages resulting from greater metabolic stability, for example increased in vivo half-life or reduced dosage requirements and, hence, may be preferred in some circumstances.
- 2-Phenoxy pyrimidinone analogues may- generally be prepared using standard synthetic methods. Starting materials are commercially available from suppliers such as
- a compound provided herein may contain one or more asymmetric carbon atoms, so that the compound can exist in different stereoisomeric forms.
- Such forms can be, for example, racemates or optically active forms.
- All stereoisomers are encompassed by the present invention. Nonetheless, it may be desirable to obtain single enantiomers ⁇ i.e., optically active forms).
- Standard methods for preparing single enantiomers include asymmetric synthesis and resolution of the racemates. Resolution of the racemates can be accomplished, for example, by conventional methods such as crystallization in the presence of a resolving agent, or chromatography using, for example a chiral HPLC column.
- Compounds may be radiolabeled by carrying out their synthesis using precursors comprising at least one atom that is a radioisotope.
- Each radioisotope is preferably carbon (e.g., 14 C), hydrogen (e.g., 3 H), sulfur (e.g., 35 S), or iodine (e.g., 125 I).
- Tritium labeled compounds may also be prepared catalytically via platinum-catalyzed exchange in tritiated acetic acid, acid-catalyzed exchange in tritiated trifluoroacetic acid, or heterogeneous- catalyzed exchange with tritium gas using the compound as substrate.
- certain precursors may be subjected to tritium-halogen exchange with tritium gas, tritium gas reduction of unsaturated bonds, or reduction using sodium borotritide, as appropriate.
- Preparation of radiolabeled compounds may be conveniently performed by a radioisotope supplier specializing in custom synthesis of radiolabeled probe compounds.
- compositions comprising one or more compounds provided herein, together with at least one physiologically acceptable carrier or excipient.
- Pharmaceutical compositions may comprise, for example, one or more of water, buffers (e.g., neutral buffered saline or phosphate buffered saline), ethanol, mineral oil, vegetable oil, dimethylsulfoxide, carbohydrates (e.g., glucose, mannose, sucrose or dextrans), mannitol, proteins, adjuvants, polypeptides or amino acids such as glycine, antioxidants, chelating agents such as EDTA or glutathione and/or preservatives.
- other active ingredients may (but need not) be included in the pharmaceutical compositions provided herein.
- compositions may be formulated for any appropriate manner of administration, including, for example, topical, oral, nasal, rectal or parenteral administration.
- parenteral as used herein includes subcutaneous, intradermal, intravascular (e.g., intravenous), intramuscular, spinal, intracranial, intrathecal and intraperitoneal injection, as well as any similar injection or infusion technique.
- compositions suitable for oral use are preferred. Such compositions include, for example, tablets, troches, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsion, hard or soft capsules, or syrups or elixirs.
- pharmaceutical compositions may be formulated as a lyophilizate.
- Formulation for topical administration may be preferred for certain conditions (e.g., in the treatment of skin conditions such as burns or itch).
- Formulation for direct administration into the bladder may be preferred for treatment of urinary incontinence and overactive bladder.
- Compositions intended for oral use may further comprise one or more components such as sweetening agents, flavoring agents, coloring agents and/or preserving agents in order to provide appealing and palatable preparations.
- Tablets contain the active ingredient in admixture with physiologically acceptable excipients that are suitable for the manufacture of tablets.
- excipients include, for example, inert diluents (e.g., calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate), granulating and disintegrating agents (e.g., corn starch or alginic acid), binding agents (e.g., starch, gelatin or acacia) and lubricating agents (e.g., magnesium stearate, stearic acid or talc). Tablets may be formed using standard techniques, including dry granulation, direct compression and wet granulation. The tablets may be uncoated or they may be coated by known techniques.
- Formulations for oral use may also be presented as hard gelatin capsules wherein the active ingredient is mixed with an inert solid diluent (e.g., calcium carbonate, calcium phosphate or kaolin), or as soft gelatin capsules wherein the active ingredient is mixed with water or an oil medium (e.g., peanut oil, liquid paraffin or olive oil).
- an inert solid diluent e.g., calcium carbonate, calcium phosphate or kaolin
- an oil medium e.g., peanut oil, liquid paraffin or olive oil
- Aqueous suspensions contain the active material(s) in admixture with suitable excipients, such as suspending agents (e.g., sodium carboxymethylcellulose, methylcellulose, hydropropylmethylcellulose, sodium alginate, polyvinylpyrrolidone, gum tragacanth and gum acacia); and dispersing or wetting agents (e.g., naturally-occurring phosphatides such as lecithin, condensation products of an alkylene oxide with fatty acids such as polyoxyethylene stearate, condensation products of ethylene oxide with long chain aliphatic alcohols such as heptadecaethyleneoxycetanol, condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide with partial esters derived from fatty acids and hexitol anhydrides such as polyethylene sorbitan monooleate).
- suspending agents e.g., sodium carb
- Aqueous suspensions may also comprise one or more preservatives, such as ethyl or n-propyl p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and/or one or more sweetening agents, such as sucrose or saccharin.
- Oily suspensions may be formulated by suspending the active ingredient(s) in a vegetable oil (e.g., arachis oil, olive oil, sesame oil or coconut oil) or in a mineral oil such as liquid paraffin.
- the oily suspensions may contain a thickening agent such as beeswax, hard paraffin or cetyl alcohol.
- Sweetening agents such as those set forth above, and/or flavoring agents may be added to provide palatable oral preparations.
- Such suspensions may be preserved by the addition of an anti-oxidant such as ascorbic acid.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water provide the active ingredient in admixture with a dispersing or wetting agent, a suspending agent and one or more preservatives.
- a dispersing or wetting agent e.g., a suspending agent
- Suitable dispersing or wetting agents and suspending agents are exemplified by those already mentioned above. Additional excipients, such as sweetening, flavoring and coloring agents, may also be present.
- compositions may also be formulated as oil-in-water emulsions.
- the oily phase may be a vegetable oil (e.g., olive oil or arachis oil), a mineral oil (e.g., liquid paraffin) or a mixture thereof.
- Suitable emulsifying agents include naturally-occurring gums
- An emulsion may also comprise one or more sweetening and/or flavoring agents.
- naturally-occurring phosphatides e.g., soy bean lecithin, and esters or partial esters derived from fatty acids and hexitol
- anhydrides e.g., sorbitan monoleate
- condensation products of partial esters derived from fatty acids and hexitol with ethylene oxide e.g., polyoxyethylene sorbitan monoleate.
- An emulsion may also comprise one or more sweetening and/or flavoring agents.
- Syrups and elixirs may be formulated with sweetening agents, such as glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also comprise one or more demulcents, preservatives, flavoring agents and/or coloring agents.
- sweetening agents such as glycerol, propylene glycol, sorbitol or sucrose.
- Such formulations may also comprise one or more demulcents, preservatives, flavoring agents and/or coloring agents.
- Formulations for topical administration typically comprise a topical vehicle combined with active agent(s), with or without additional optional components.
- Suitable topical vehicles and additional components are well known in the art, and it will be apparent that the choice of a vehicle will depend on the particular physical form and mode of delivery.
- Topical vehicles include water; organic solvents such as alcohols (e.g., ethanol or isopropyl alcohol) or glycerin; glycols (e.g., butylene, isoprene or propylene glycol); aliphatic alcohols (e.g., lanolin); mixtures of water and organic solvents and mixtures of organic solvents such as alcohol and glycerin; lipid-based materials such as fatty acids, acylglycerols (including oils, such as mineral oil, and fats of natural or synthetic origin), phosphoglycerides, sphingolipids and waxes; protein-based materials such as collagen and gelatin; silicone-based materials (both non-volatile and volatile); and hydrocarbon-based materials such as microsponges and polymer matrices.
- organic solvents such as alcohols (e.g., ethanol or isopropyl alcohol) or glycerin
- glycols e.g., butylene, isoprene or
- a composition may further include one or more components adapted to improve the stability or effectiveness of the applied formulation, such as stabilizing agents, suspending agents, emulsifying agents, viscosity adjusters, gelling agents, preservatives, antioxidants, skin penetration enhancers, moisturizers and sustained release materials.
- stabilizing agents such as hydroxymethylcellulose or gelatin-microcapsules, liposomes, albumin microspheres, microemulsions, nanoparticles or nanocapsules.
- a topical formulation may be prepared in any of a variety of physical forms including, for example, solids, pastes, creams, foams, lotions, gels, powders, aqueous liquids and emulsions.
- the physical appearance and viscosity of such pharmaceutically acceptable forms can be governed by the presence and amount of emulsif ⁇ er(s) and viscosity adjusters) present in the formulation.
- Solids are generally firm and non-pourable and commonly are formulated as bars or sticks, or in particulate form; solids can be opaque or transparent, and optionally can contain solvents, emulsif ⁇ ers, moisturizers, emollients, fragrances, dyes/colorants, preservatives and other active ingredients that increase or enhance the efficacy of the final product.
- Creams and lotions are often similar to one another, differing mainly in their viscosity; both lotions and creams may be opaque, translucent or clear and often contain emulsif ⁇ ers, solvents, and viscosity adjusting agents, as well as moisturizers, emollients, fragrances, dyes/colorants, preservatives and other active ingredients that increase or enhance the efficacy of the final product.
- Gels can be prepared with a range of viscosities, from thick or high viscosity to thin or low viscosity.
- These formulations may also contain solvents, emulsifiers, moisturizers, emollients, fragrances, dyes/colorants, preservatives and other active ingredients that increase or enhance the efficacy of the final product.
- Liquids are thinner than creams, lotions, or gels and often do not contain emulsifiers.
- Liquid topical products often contain solvents, emulsifiers, moisturizers, emollients, fragrances, dyes/colorants, preservatives and other active ingredients that increase or enhance the efficacy of the final product.
- Suitable emulsifiers for use in topical formulations include, but are not limited to, ionic emulsifiers, cetearyl alcohol, non-ionic emulsifiers like polyoxyethylene oleyl ether, PEG-40 stearate, ceteareth-12, ceteareth-20, ceteareth-30, ceteareth alcohol, PEG-100 stearate and glyceryl stearate.
- Suitable viscosity adjusting agents include, but are not limited to, protective colloids or non-ionic gums such as hydroxyethylcellulose, xanthan gum, magnesium aluminum silicate, silica, microcrystalline wax, beeswax, paraffin, and cetyl pahnitate.
- a gel composition may be formed by the addition of a gelling agent such as chitosan, methyl cellulose, ethyl cellulose, polyvinyl alcohol, polyquaterniums, hydroxyethylcellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose, carbomer or ammoniated glycyrrhizinate.
- a gelling agent such as chitosan, methyl cellulose, ethyl cellulose, polyvinyl alcohol, polyquaterniums, hydroxyethylcellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose, carbomer or ammoniated glycyrrhizinate.
- Suitable surfactants include, but are not limited to, nonionic, amphoteric, ionic and anionic surfactants.
- dimethicone copolyol polysorbate 20, polysorbate 40, polysorbate 60, polysorbate 80, lauramide DEA, cocamide DEA, and cocamide MEA, oleyl betaine, cocamidopropyl phosphatidyl PG- dimonium chloride, and ammonium laureth sulfate may be used within topical formulations.
- Suitable preservatives include, but are not limited to, antimicrobials such as methylparaben, propylparaben, sorbic acid, benzoic acid, and formaldehyde, as well as physical stabilizers and antioxidants such as vitamin E, sodium ascorbate/ascorbic acid and propyl gallate.
- Suitable moisturizers include, but are not limited to, lactic acid and other hydroxy acids and their salts, glycerin, propylene glycol, and butylene glycol.
- Suitable emollients include lanolin alcohol, lanolin, lanolin derivatives, cholesterol, petrolatum, isostearyl neopentanoate and mineral oils.
- Suitable fragrances and colors include, but are not limited to, FD&C Red No. 40 and FD&C Yellow No. 5.
- Suitable additional ingredients include, but are not limited to, abrasives, absorbents, anti-caking agents, anti-foaming agents, anti-static agents, astringents (e.g., witch hazel, alcohol and herbal extracts such as chamomile extract), binders/excipients, buffering agents, chelating agents, film forming agents, conditioning agents, propellants, opacifying agents, pH adjusters and protectants.
- An example of a suitable topical vehicle for formulation of a gel is: hydroxypropylcellulose (2.1%); 70/30 isopropyl alcohol/water (90.9%); propylene glycol (5.1%); and Polysorbate 80 (1.9%).
- An example of a suitable topical vehicle for formulation as a foam is: cetyl alcohol (1.1%); stearyl alcohol (0.5%; Quaternium 52 (1.0%); propylene glycol (2.0%); ethanol 95 PGF3 (61.05%); deionized water (30.05%); P75 hydrocarbon propellant (4.30%). All percents are by weight.
- Typical modes of delivery for topical compositions include application using the fingers; application using a physical applicator such as a cloth, tissue, swab, stick or brush; spraying (including mist, aerosol or foam spraying); dropper application; sprinkling; soaking; and rinsing.
- a pharmaceutical composition may be prepared as a sterile injectible aqueous or oleaginous suspension.
- the compound(s) provided herein, depending on the vehicle and concentration used, can either be suspended or dissolved in the vehicle.
- Such a composition may be formulated according to the known art using suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable dispersing, wetting agents and/or suspending agents such as those mentioned above.
- suitable vehicles and solvents that may be employed are water, 1,3-butanediol, Ringer's solution
- compositions may also be formulated as suppositories (e.g., for rectal administration). Such compositions can be prepared by mixing the drug with a suitable non- irritating excipient that is solid at ordinary temperatures but liquid at the rectal temperature and will therefore melt in the rectum to release the drug. Suitable excipients include, for example, cocoa butter and polyethylene glycols.
- compositions for inhalation typically can be provided in the form of a solution, suspension or emulsion that can be administered as a dry powder or in the form of an aerosol using a conventional propellant ⁇ e.g., dichlorodifluoromethane or trichlorofluoromethane).
- a conventional propellant e.g., dichlorodifluoromethane or trichlorofluoromethane
- compositions may be formulated for release at a pre-determined rate.
- Instantaneous release may be achieved, for example, via sublingual administration (i.e., administration by mouth in such a way that the active ingredient(s) are rapidly absorbed via the blood vessels under the tongue rather than via the digestive tract).
- Controlled release formulations i.e., formulations such as a capsule, tablet or coated tablet that slows and/or delays release of active ingredient(s) following administration
- a controlled release formulation comprises a matrix and/or coating that delays disintegration and absorption in the gastrointestinal tract (or implantation site) and thereby provides a delayed action or a sustained action over a longer period.
- One type of controlled- release formulation is a sustained-release formulation, in which at least one active ingredient is continuously released over a period of time at a constant rate.
- the therapeutic agent is released at such a rate that blood (e.g., plasma) concentrations are maintained within the therapeutic range, but below toxic levels, over a period of time that is at least 4 hours, preferably at least 8 hours, and more preferably at least 12 hours.
- Such formulations may generally be prepared using well known technology and administered by, for example, oral, rectal or subcutaneous implantation, or by implantation at the desired target site.
- Carriers for use within such formulations are biocompatible, and may also be biodegradable; preferably the formulation provides a relatively constant level of modulator release.
- the amount of modulator contained within a sustained release formulation depends upon, for example, the site of implantation, the rate and expected duration of release and the nature of the condition to be treated or prevented.
- Controlled release may be achieved by combining the active ingredient(s) with a matrix material that itself alters release rate and/or through the use of a controlled-release coating.
- the release rate can be varied using methods well known in the art, including (a) varying the thickness or composition of coating, (b) altering the amount or manner of addition of plasticizer in a coating, (c) including additional ingredients, such as release-modifying agents, (d) altering the composition, particle size or particle shape of the matrix, and (e) providing one or more passageways through the coating.
- the amount of modulator contained within a sustained release formulation depends upon, for example, the method of administration (e.g., the site of implantation), the rate and expected duration of release and the nature of the condition to be treated or prevented.
- the matrix material which itself may or may not serve a controlled-release function, is generally any material that supports the active i ⁇ gredient(s).
- a time delay material such as glyceryl monosterate or glyceryl distearate may be employed.
- Active ingredient(s) may be combined with matrix material prior to formation of the dosage form (e.g., a tablet).
- active ingredient(s) may be coated on the surface of a particle, granule, sphere, microsphere, bead or pellet that comprises the matrix material. Such coating may be achieved by conventional means, such as by dissolving the active ingredient(s) in water or other suitable solvent and spraying.
- a controlled release is achieved through the use of a controlled release coating (i.e., a coating that permits release of active ingredient(s) at a controlled rate in aqueous medium).
- the controlled release coating should be a strong, continuous film that is smooth, capable of supporting pigments and other additives, non-toxic, inert and tack-free.
- Coatings that regulate release of the modulator include pH-independent coatings, pH-dependent coatings (which may be used to release modulator in the stomach) and enteric coatings (which allow the formulation to pass intact through the stomach and into the small intestine, where the coating dissolves and the contents are absorbed by the body). It will be apparent that multiple coatings may be employed (e.g., to allow release of a portion of the dose in the stomach and a portion further along the gastrointestinal tract). For example, a portion of active ingredient(s) may be coated over an enteric coating, and thereby released in the stomach, while the remainder of active ingredient(s) in the matrix core is protected by the enteric coating and released further down the GI tract.
- pH dependent coatings include, for example, shellac, cellulose acetate phthalate, polyvinyl acetate phthalate, hydroxypropylmethylcellulose phthalate, methacrylic acid ester copolymers and zein.
- the coating is a hydrophobic material, preferably used in an amount effective to slow the hydration of the gelling agent following administration.
- Suitable hydrophobic materials include alkyl celluloses (e.g., ethylcellulose or carboxymethylcellulose), cellulose ethers, cellulose esters, acrylic polymers (e.g., poly(acrylic acid), poly(methacrylic acid), acrylic acid and methacrylic acid copolymers, methyl methacrylate copolymers, ethoxy ethyl methacrylates, cyanoethyl methacrylate, methacrylic acid alkamide copolymer, poly(methyl methacrylate), polyacryl amide, ammonio methacrylate copolymers, aminoalkyl methacrylate copolymer, poly(methacrylic acid anhydride) and glycidyl methacrylate copolymers) and mixtures of the foregoing.
- Representative aqueous dispersions of ethylcellulose include, for example, AQUACOAT® (FMC Corp., Philadelphia, PA) and SURELEASE® (Colorcon, Inc., West Point, PA), both of which can be applied to the substrate according to. the manufacturer's instructions.
- Representative acrylic polymers include, for example, the various EUDRAGIT® (Rohm America, Piscataway, NJ) polymers, which may be used singly or in combination depending on the desired release profile, according to the manufacturer's instructions.
- Suitable plasticizers for alkyl celluloses include, for example, dibutyl sebacate, diethyl phthalate, triethyl citrate, tributyl citrate and triacetin.
- Suitable plasticizers for acrylic polymers include, for example, citric acid esters such as triethyl citrate and tributyl citrate, dibutyl phthalate, polyethylene glycols, propylene glycol, diethyl phthalate, castor oil and triacetin.
- Controlled-release coatings are generally applied using conventional techniques, such as by spraying in the form of an aqueous dispersion.
- the coating may comprise pores or channels or to facilitate release of active ingredient. Pores and channels may be generated by well known methods, including the addition of organic or inorganic material that is dissolved, extracted or leached from the coating in the environment of use.
- pore-forming materials include hydrophilic polymers, such as hydroxyalkylcelluloses ⁇ e.g., hydroxypropylmethylcellulose), cellulose ethers, synthetic water-soluble polymers (e.g., polyvinylpyrrolidone, cross-linked polyvinylpyrrolidone and polyethylene oxide), water- soluble polydextrose, saccharides and polysaccharides and alkali metal salts.
- a controlled release coating may include one or more orifices, which may be formed my methods such as those described in US Patent Nos. 3,845,770; 4,034,758; 4,077,407; 4,088,864; 4,783,337 and 5,071,607. Controlled-release may also be achieved through the use of transdermal patches, using conventional technology (see, e.g., US Patent No. 4,668,232).
- controlled release formulations may be found, for example, in US Patent Nos. 5,524,060; 4,572,833; 4,587,117; 4,606,909; 4,610,870; 4,684,516; 4,777,049; 4,994,276; 4,996,058; 5,128,143; 5,202,128; 5,376,384; 5,384,133; 5,445,829; 5,510,119; 5,618,560; 5,643,604; 5,891,474; 5,958,456; 6,039,980; 6,143,353; 6,126,969; 6,156,342; 6,197,347; 6,387,394; 6,399,096; 6,437,000; 6,447,796; 6,475,493; 6,491,950; 6,524,615; 6,838,094; 6,905,709; 6,923,984; 6,923,988; and 6,911,217; each of which is hereby incorporated by reference for its teaching of the
- a compound provided herein may be conveniently added to food or drinking water (e.g., for administration to non-human animals including companion animals (such as dogs and cats) and livestock).
- Animal feed and drinking water compositions may be formulated so that the animal takes in an appropriate quantity of the composition along with its diet. It may also be convenient to present the composition as a premix for addition to feed or drinking water.
- Compounds are generally administered in a therapeutically effective amount.
- Preferred systemic doses are no higher than 50 mg per kilogram of body weight per day (e.g., ranging from about 0.001 mg to about 50 mg per kilogram of body weight per day), with oral doses generally being about 5-20 fold higher than intravenous doses (e.g., ranging from 0.01 to 40 mg per kilogram of body weight per day).
- the amount of active ingredient that may be combined with the carrier materials to produce a single dosage unit will vary depending, for example, upon the patient being treated, the particular mode of administration and any other co-administered drugs. Dosage units generally contain between from about 10 ⁇ g to about 500 mg of active ingredient. Optimal dosages may be established using routine testing, and procedures that are well known in the art.
- compositions may be packaged for treating conditions responsive to VRl modulation (e.g., treatment of exposure to vanilloid ligand or other irritant, pain, itch, obesity or urinary incontinence).
- Packaged pharmaceutical compositions generally include (i) a container holding a pharmaceutical composition that comprises at least one VRl modulator as described herein and (ii) instructions (e.g., labeling or a package insert) indicating that the contained composition is to be used for treating a condition responsive to VRl modulation in the patient.
- VRl modulators provided herein may be used to alter activity and/or activation of capsaicin receptors in a variety of contexts, both in vitro and in vivo.
- VRl antagonists may be used to inhibit the binding of vanilloid ligand agonist (such as capsaicin and/or RTX) to capsaicin receptor in vitro or in vivo.
- such methods comprise the step of contacting a capsaicin receptor with one or more VRl modulators provided herein, in the presence of vanilloid ligand in aqueous solution and under conditions otherwise suitable for binding of the ligand to capsaicin receptor.
- the VRl modulator(s) are generally present at a concentration that is sufficient to alter the binding of vanilloid ligand to VRl in vitro (using the assay provided in Example 5) and/or VRl-mediated signal transduction (using an assay provided in Example 6).
- the capsaicin receptor may be present in solution or suspension (e.g., in an isolated membrane or cell preparation), or in a cultured or isolated cell. Within certain embodiments, the capsaicin receptor is expressed by a neuronal cell present in a patient, and the aqueous solution is a body fluid.
- one or more VRl modulators are administered to an animal in an amount such that the VRl modulator is present in at least one body fluid of the animal at a therapeutically effective concentration that is 1 micromolar or less; preferably 500 nanomolar or less; more preferably 100 nanomolar or less, 50 nanomolar or less, 20 nanomolar or less, or 10 nanomolar or less.
- a therapeutically effective dose that is less than 20 mg/kg body weight, preferably less than 5 mg/kg and, in some instances, less than 1 mg/kg.
- modulation may be achieved by contacting a capsaicin receptor (either in vitro or in vivo) with one or more VRl modulators provided herein under conditions suitable for binding of the modulator(s) to the receptor.
- the VRl modulator(s) are generally present at a concentration that is sufficient to alter the binding of vanilloid ligand to VRl in vitro and/or VRl-mediated signal transduction as described herein.
- the receptor may be present in solution or suspension, in a cultured or isolated cell preparation or in a cell within a patient.
- the cell may be a neuronal cell that is contacted in vivo in an animal.
- the cell may be an epithelial cell, such as a urinary bladder epithelial cell (urothelial cell) or an airway epithelial cell that is contacted in vivo in an animal.
- Modulation of signal tranducing activity may be assessed by detecting an effect on calcium ion conductance (also referred to as calcium mobilization or flux).
- Modulation of signal transducing activity may alternatively be assessed by detecting an alteration of a symptom (e.g., pain, burning sensation, broncho-constriction, inflammation, cough, hiccup, itch, menopause symptoms, urinary incontinence or overactive bladder) of a patient being treated with one or more VRl modulators provided herein.
- a symptom e.g., pain, burning sensation, broncho-constriction, inflammation, cough, hiccup, itch, menopause symptoms, urinary incontinence or overactive bladder
- VRl modulator(s) provided herein are preferably administered to a patient (e.g., a human) orally or topically, and are present within at least one body fluid of the animal while modulating VRl signal-transducing activity.
- Preferred VRl modulators for use in such methods modulate VRl signal-transducing activity in vitro at a concentration of 1 nanomolar or less, preferably 100 picomolar or less, more preferably 20 picomolar or less, and in vivo at a concentration of 1 micromolar or less, 500 nanomolar or less, or 100 nanomolar or less in a body fluid such as blood.
- the present invention further provides methods for treating conditions responsive to
- the term “treatment” encompasses both disease-modifying treatment and symptomatic treatment, either of which may be prophylactic (i.e., before the onset of symptoms, in order to prevent, delay or reduce the severity of symptoms) or therapeutic (i.e., after the onset of symptoms, in order to reduce the severity and/or duration of symptoms).
- a condition is "responsive to VRl modulation” if it is characterized by inappropriate activity of a capsaicin receptor, regardless of the amount of vanilloid ligand present locally, and/or if modulation of capsaicin receptor activity results in alleviation of the condition or a symptom thereof.
- Such conditions include, for example, symptoms resulting from exposure to VRl -activating stimuli, pain, respiratory disorders (such as cough, asthma, chronic obstructive pulmonary disease, chronic bronchitis, cystic fibrosis and rhinitis, including allergic rhinitis, such as seasonal an perennial rhinitis, and non- allergic rhinitis), depression, itch, menopause symptoms, urinary incontinence, overactive bladder, acoustic injury (e.g., of the cochlea), tinnitus, hyperacusis, diabetes and prediabetic conditions (e.g., insulin resistance or glucose tolerance), hiccup and obesity, as described in more detail below.
- Such conditions may be diagnosed and monitored using criteria that have been established in the art. Patients may include humans, domesticated companion animals and livestock, with dosages as described above.
- Treatment regimens may vary depending on the compound used and the particular condition to be treated; however, for treatment of most disorders, a frequency of administration of 4 times daily or less is preferred. In general, a dosage regimen of 2 times daily is more preferred, with once a day dosing particularly preferred. For the treatment of acute pain, a single dose that rapidly reaches effective concentrations is desirable. It will be understood, however, that the specific dose level and treatment regimen for any particular patient will depend upon a variety of factors including the activity of the specific compound employed, the age, body weight, general health, sex, diet, time of administration, route of administration, and rate of excretion, drug combination and the severity of the particular disease undergoing therapy. In general, the use of the minimum dose sufficient to provide effective therapy is preferred. Patients may generally be monitored for therapeutic effectiveness using medical or veterinary criteria suitable for the condition being treated or prevented.
- Patients experiencing symptoms resulting from exposure to capsaicin receptor- activating stimuli include individuals with burns caused by heat, light, tear gas or acid and those whose mucous membranes are exposed (e.g., via ingestion, inhalation or eye contact) to capsaicin (e.g., from hot peppers or in pepper spray) or a related irritant such as acid, tear gas, infectious agent(s) or air pollutant(s).
- the resulting symptoms may include, for example, pain, broncho-constriction and inflammation.
- Pain that may be treated using the VRl modulators provided herein may be chronic or acute and includes, but is not limited to, peripheral nerve-mediated pain (especially neuropathic pain).
- Compounds provided herein may be used in the treatment of, for example, postmastectomy pain syndrome, stump pain, phantom limb pain, oral neuropathic pain, toothache (dental pain), denture pain, postherpetic neuralgia, diabetic neuropathy, chemotherapy-induced neuropathy, reflex sympathetic dystrophy, trigeminal neuralgia, osteoarthritis, rheumatoid arthritis, fibromyalgia, Guillain-Barre syndrome, meralgia paresthetica, burning-mouth syndrome and/or pain associated with nerve and root damage, including as pain associated with peripheral nerve disorders (e.g., nerve entrapment and brachial plexus avulsions, amputation, peripheral neuropathies including bilateral peripheral neuropathy, tic douloureux, atypical facial pain, nerve root damage, and a
- Additional neuropathic pain conditions include causalgia (reflex sympathetic dystrophy - RSD, secondary to injury of a peripheral nerve), neuritis (including, for example, sciatic neuritis, peripheral neuritis, polyneuritis, optic neuritis, postfebrile neuritis, migrating neuritis, segmental neuritis and Gombault's neuritis), neuronitis, neuralgias (e.g., those mentioned above, cervicobrachial neuralgia, cranial neuralgia, geniculate neuralgia, glossopharyngial neuralgia, migranous neuralgia, idiopathic neuralgia, intercostals neuralgia, mammary neuralgia, mandibular joint neuralgia, Morton's neuralgia, nasociliary neuralgia, occipital neuralgia, red neuralgia, Sluder's neuralgia, splenopalatine neuralg
- Headache including headaches involving peripheral nerve activity may also be treated as described herein.
- pain includes, for example, such as sinus, cluster (i.e., migrainous neuralgia) and tension headaches, migraine, temporomandibular pain and maxillary sinus pain.
- migraine headaches may be prevented by administration of a compound provided herein as soon as a pre-migrainous aura is experienced by the patient.
- Charcot's pains intestinal gas pains, ear pain, heart pain, muscle pain, eye pain, orofacial pain (e.g., odontalgia), abdominal pain, gynaecological pain (e.g., menstrual pain, dysmenorrhoea, pain associated with cystitis, labor pain, chronic pelvic pain, chronic prostitis and endometriosis), acute and chronic back pain (e.g., lower back pain), gout, scar pain, hemorrhoidal pain, dyspeptic pains, angina, nerve root pain, "non-painful" neuropathies, complex regional pain syndrome, homotopic pain and heterotopic pain — including pain associated with carcinoma, often referred to as cancer pain (e.g., in patients with bone cancer), pain (and inflammation) associated with venom exposure (e.g., due to snake bite, spider bite, or insect sting) and trauma associated pain (e.g., post-surgical pain, episi
- Additional pain conditions that may be treated as described herein include pain associated with respiratory disorders as described above, autoimmune diseases, immunodeficiency disorders, hot flashes, inflammatory bowel disease, gastroesophageal reflux disease (GERD), irritable bowel syndrome and/or inflammatory bowel disease.
- VRl modulators provided herein may be used for the treatment of mechanical pain.
- mechanical pain refers to pain other than headache pain that is not neuropathic or a result of exposure to heat, cold or external chemical stimuli.
- Mechanical pain includes physical trauma (other than thermal or chemical burns or other irritating and/or painful exposures to noxious chemicals) such as post-surgical pain and pain from cuts, bruises and broken bones; toothache; denture pain; nerve root pain; osteoarthritis; rheumatoid arthritis; fibromyalgia; meralgia paresthetica; back pain; cancer- associated pain; angina; carpel tunnel syndrome; and pain resulting from bone fracture, labor, hemorrhoids, intestinal gas, dyspepsia, and menstruation.
- Itching conditions that may be treated include psoriatic pruritus, itch due to hemodialysis, aguagenic pruritus, and itching associated with vulvar vestibulitis, contact dermatitis, insect bites and skin allergies.
- Urinary tract conditions that may be treated as described herein include urinary incontinence (including overflow incontinence, urge incontinence and stress incontinence), as well as overactive or unstable bladder conditions (including bladder detrusor hyper-reflexia, detrusor hyper-reflexia of spinal origin and bladder hypersensitivity).
- VRl modulator is administered via a catheter or similar device, resulting in direct injection of VRl modulator into the bladder.
- Compounds provided herein may also be used as anti-tussive agents (to prevent, relieve or suppress coughing, including cough induced by medications such as ACE inhibitors) and for the treatment of hiccup, for the treatment of menopause symptoms such as hot flashes, and to promote weight loss in an obese patient.
- VRl modulators provided herein may be used within combination therapy for the treatment of conditions involving pain and/or inflammatory components.
- Such conditions include, for example, autoimmune disorders and pathologic autoimmune responses known to have an inflammatory component including, but not limited to, arthritis (especially rheumatoid arthritis), psoriasis, Crohn's disease, lupus erythematosus, irritable bowel syndrome, tissue graft rejection, and hyperacute rejection of transplanted organs.
- Other such conditions include trauma (e.g., injury to the head or spinal cord), cardio- and cerebro-vascular disease and certain infectious diseases.
- Suitable dosages for VRl modulator within such combination therapy are generally as described above. Dosages and methods of administration of anti-inflammatory agents can be found, for example, in the manufacturer's instructions in the Physician's Desk Reference.
- the combination administration of a VRl modulator with an antiinflammatory agent results in a reduction of the dosage of the anti-inflammatory agent required to produce a therapeutic effect (i.e., a decrease in the minimum therapeutically effective amount).
- the dosage of anti-inflammatory agent in a combination or combination treatment method is less than the maximum dose advised by the manufacturer for administration of the anti-inflammatory agent without combination administration of a VRl antagonist.
- this dosage is less than 3 A, even more preferably less than 1 A, and highly preferably, less than % of the maximum dose, while most preferably the dose is less than 10% of the maximum dose advised by the manufacturer for administration of the anti-inflammatory agent(s) when administered without combination administration of a VRl antagonist. It will be apparent that the dosage amount of VRl antagonist component of the combination needed to achieve the desired effect may similarly be affected by the dosage amount and potency of the anti-inflammatory agent component of the combination.
- the combination administration of a VRl modulator with an anti-inflammatory agent is accomplished by packaging one or more VRl modulators and one or more anti-inflammatory agents in the same package, either in separate containers within the package or in the same contained as a mixture of one or more VRl antagonists and one or more anti-inflammatory agents.
- Preferred mixtures are formulated for oral administration (e.g., as pills, capsules, tablets or the like).
- the package comprises a label bearing indicia indicating that the one or more VRl modulators and one or more anti-inflammatory agents are to be taken together for the treatment of an inflammatory pain condition.
- VRl modulators provided herein may be used in combination with one or more additional pain relief medications.
- Certain such medications are also anti- inflammatory agents, and are listed above.
- Other such medications are analgesic agents, including narcotic agents which typically act at one or more opioid receptor subtypes (e.g., ⁇ , K and/or ⁇ ), preferably as agonists or partial agonists.
- opioid receptor subtypes e.g., ⁇ , K and/or ⁇
- Such agents include opiates, opiate derivatives and opioids, as well as pharmaceutically acceptable salts and hydrates thereof.
- narcotic analgesics include, within preferred embodiments, alfentanil, alphaprodine, anileridine, bezitramide, buprenorphine, butorphanol, codeine, diacetyldihydromorphine, diacetylmorphine, dihydrocodeine, diphenoxylate, ethylmorphine, fentanyl, heroin, hydrocodone, hydromorphone, isomethadone, levomethorphan, levorphane, levorphanol, meperidine, metazocine, methadone, methorphan, metopon, morphine, nalbuphine, opium extracts, opium fluid extracts, powdered opium, granulated opium, raw opium, tincture of opium, oxycodone, oxymorphone, paregoric, pentazocine, pethidine, phenazocine, piminodine, propoxyphene, racemethorphan
- narcotic analgesic agents include acetorphine, acetyldihydrocodeine, acetylmethadol, allylprodine, alphracetylmethadol, alphameprodine, alphamethadol, benzethidine, benzylmorphine, betacetylmethadol, betameprodine, betamethadol, betaprodine, clonitazene, codeine methylbromide, codeine-N-oxide, cyprenorphine, desomorphine, dextromoramide, diampromide, diethylthiambutene, dihydromorphine, dimenoxadol, dimepheptanol, dimethylthiamubutene, dioxaphetyl butyrate, dipipanone, drotebanol, ethanol, ethylmethylthiambutene, etonitazene, e
- analgesic agents include, for example acetaminophen (paracetamol); ibuprofen; aspirin and other NSAIDs described above; NR2B antagonists; bradykinin antagonists; anti-migraine agents; anticonvulsants such as oxcarbazepine and carbamazepine; antidepressants (such as TCAs, SSRIs, SNRIs, substance P antagonists, etc.); spinal blocks; pentazocine/naloxone; meperidine; levo ⁇ hanol; bupreno ⁇ hine; hydromo ⁇ hone; fentanyl; sufentanyl; oxycodone; oxycodone/acetaminophen, nalbuphine and oxymorphone.
- analgesic agents include CB2-receptor agonists, such as AM 1241, capsaicin receptor antagonists and compounds that bind to the ⁇ 2 ⁇ subunit of voltage-gated calcium channels, such as gabapentin and pregabalin.
- Representative anti-migraine agents for use in combination with a VRl modulator provided herein include CGRP antagonists, ergotamines and 5-HTj agonists, such as sumatripan, naratriptan, zolmatriptan and rizatriptan.
- modulators provided herein may be used, for example, in the treatment of pulmonary disorders such as asthma, in combination with one or more beta(2)-adrenergic receptor agonists or leukotriene receptor antagonists (e.g., agents that inhibits the cysteinyl leukotriene CysLTi receptor).
- CysLTi antagonists include montelukast, zaf ⁇ rlukast, and pranlukast.
- a VRl modulator as provided herein may be used in combination with other medication designed to treat this condition, such as antibiotics, anti-inflammatory agents, cystinyl leukotrienes, histamine antagonists, corticosteroids, opioids, NMDA antagonists, proton pump inhibitors, nociceptin, neurokinin (NKl, NK2 and NK3) and bradykinin (BKl and BK2) receptor antagonists, cannabinoids, blockers of Na+-dependent channels and large conductance Ca +2 -dependent K + -channel activators.
- other medication designed to treat this condition such as antibiotics, anti-inflammatory agents, cystinyl leukotrienes, histamine antagonists, corticosteroids, opioids, NMDA antagonists, proton pump inhibitors, nociceptin, neurokinin (NKl, NK2 and NK3) and bradykinin (BKl and BK2) receptor antagonists, cannabinoids, blockers of Na+-dependent channels and large conductance Ca
- Specific agents include dexbrompheniramine plus pseudoephedrine, loratadine, oxymetazoline, ipratropium, albuterol, beclomethasone, morphine, codeine, pholcodeine and dextromethorphan.
- the present invention further provides combination therapy for the treatment of urinary incontinence.
- a VRl modulator provided herein may be used in combination with other medication designed to treat this condition, such as estrogen replacement therapy, progesterone congeners, electrical stimulation, calcium channel blockers, antispasmodic agents, cholinergic antagonists, antimuscarinic drugs, tricyclic antidepressants, SNRIs, beta adrenoceptor agonists, phosphodiesterase inhibitors, potassium channel openers, nociceptin/orphanin FQ (OP4) agonists, neurokinin (NKl and NK2) antagonists, P2X3 antagonists, musculotrophic drugs and sacral neuromodulation.
- other medication designed to treat this condition, such as estrogen replacement therapy, progesterone congeners, electrical stimulation, calcium channel blockers, antispasmodic agents, cholinergic antagonists, antimuscarinic drugs, tricyclic antidepressants, SNRIs, beta adrenoceptor agonists,
- Specific agents include oxybutinin, emepronium, tolterodine, flavoxate, flurbiprofen, tolterodine, dicyclomine, propiverine, propantheline, dicyclomine, imipramine, doxepin, duloxetine, 1- deamino-8-D-arginine vasopressin, muscarinic receptor antagonists such as tolterodine and anticholinergic agents such as oxybutynin.
- Suitable dosages for VRl modulator within such combination therapy are generally as described above. Dosages and methods of administration of other pain relief medications can be found, for example, in the manufacturer's instructions in the Physician's Desk Reference. In certain embodiments, the combination administration of a VRl modulator with one or more additional pain medications results in a reduction of the dosage of each therapeutic agent required to produce a therapeutic effect (e.g., the dosage or one or both agent may less than %, less than Vi, less than % or less than 10% of the maximum dose listed above or advised by the manufacturer).
- compositions as described above may further comprise one or more additional medications as described above.
- the additional medication is an analgesic.
- packaged pharmaceutical preparations comprising one or more VRl modulators and one or more additional medications (e.g., analgesics) in the same package.
- Such packaged pharmaceutical preparations generally include (i) a container holding a pharmaceutical composition that comprises at least one VRl modulator as described herein; (ii) a container holding a pharmaceutical composition that comprises at least one additional medication (such as a pain relief and/or anti-inflammatory medication) as described above and (iii) instructions (e.g., labeling or a package insert) indicating that the compositions are to be used simultaneously, separately or sequentially for treating or preventing a condition responsive to VRl modulation in the patient (such as a condition in which pain and/or inflammation predominates).
- a condition responsive to VRl modulation in the patient such as a condition in which pain and/or inflammation predominates.
- VRl agonists may further be used, for example, in crowd control (as a substitute for tear gas) or personal protection (e.g., in a spray formulation) or as pharmaceutical agents for the treatment of pain, itch, menopause symptoms, urinary incontinence or overactive bladder via capsaicin receptor desensitization.
- crowd control as a substitute for tear gas
- personal protection e.g., in a spray formulation
- pharmaceutical agents for the treatment of pain, itch, menopause symptoms, urinary incontinence or overactive bladder via capsaicin receptor desensitization are formulated and used according to conventional tear gas or pepper spray technology.
- the present invention provides a variety of non- pharmaceutical in vitro and in vivo uses for the compounds provided herein.
- such compounds may be labeled and used as probes for the detection and localization of capsaicin receptor (in samples such as cell preparations or tissue sections, preparations or fractions thereof).
- compounds provided herein that comprise a suitable reactive group such as an aryl carbonyl, nitro or azide group
- a suitable reactive group such as an aryl carbonyl, nitro or azide group
- compounds provided herein may be used as positive controls in assays for receptor activity, as standards for determining the ability of a candidate agent to bind to capsaicin receptor, or as radiotracers for positron emission tomography (PET) imaging or for single photon emission computerized tomography (SPECT).
- PET positron emission tomography
- SPECT single photon emission computerized tomography
- Such methods can be used to characterize capsaicin receptors in living subjects.
- a VRl modulator may be labeled using any of a variety of well known techniques (e.g., radiolabeled with a radionuclide such as tritium, as described herein), and incubated with a sample for a suitable incubation time (e.g., determined by first assaying a time course of binding).
- unbound compound is removed (e.g., by washing), and bound compound detected using any method suitable for the label employed (e.g., autoradiography or scintillation counting for radiolabeled compounds; spectroscopic methods may be used to detect luminescent groups and fluorescent groups).
- any method suitable for the label employed e.g., autoradiography or scintillation counting for radiolabeled compounds; spectroscopic methods may be used to detect luminescent groups and fluorescent groups.
- a matched sample containing labeled compound and a greater (e.g., 10-fold greater) amount of unlabeled compound may be processed in the same manner. A greater amount of detectable label remaining in the test sample than in the control indicates the presence of capsaicin receptor in the sample.
- Detection assays including receptor autoradiography (receptor mapping) of capsaicin receptor in cultured cells or tissue samples may be performed as described by Kuhar in sections 8.1.1 to 8.1.9 of Current Protocols in Pharmacology (1998) John Wiley & Sons, New York.
- Compounds provided herein may also be used within a variety of well known cell separation methods.
- modulators may be linked to the interior surface of a tissue culture plate or other support, for use as affinity ligands for immobilizing and thereby isolating, capsaicin receptors (e.g., isolating receptor-expressing cells) in vitro.
- a modulator linked to a fluorescent marker such as fluorescein, is contacted with the cells, which are then analyzed (or isolated) by fluorescence activated cell sorting (FACS).
- FACS fluorescence activated cell sorting
- VRl modulators provided herein may further be used within assays for the identification of other agents that bind to capsaicin receptor.
- assays are standard competition binding assays, in which bound, labeled VRl modulator is displaced by a test compound.
- such assays are performed by: (a) contacting capsaicin receptor with a radiolabeled VRl modulator as described herein, under conditions that permit binding of the VRl modulator to capsaicin receptor, thereby generating bound, labeled VRl modulator; (b) detecting a signal that corresponds to the amount of bound, labeled VRl modulator in the absence of test agent; (c) contacting the bound, labeled VRl modulator with a test agent; (d) detecting a signal that corresponds to the amount of bound labeled VRl modulator in the presence of test agent; and (e) detecting a decrease in signal detected in step (d), as compared to the signal detected in step (b).
- Mass Spectroscopy data provide in the following examples is Electrospray MS, obtained in positive ion mode with a 15V or 30V cone voltage, using a Micromass Time-of- Flight LCT, equipped with a Waters 600 pump, Waters 996 photodiode array detector, Gilson 215 autosampler, and a Gilson 841 microinjector. MassLynx (Advanced Chemistry Development, Inc; Toronto, Canada) version 4.0 software is used for data collection and analysis. Sample volume of 1 microliter is injected onto a 50 * 4.6 mm Chromolith • SpeedROD Cl 8 column, and eluted using a 2-phase linear gradient at 6ml/min flow rate. Sample is detected using total absorbance count over the 220-340nm UV range. The elution conditions are: Mobile Phase A- 95/5/0.05 Water/Methanol/TFA; Mobile Phase B-5/95/0.025 Water/Methanol/TFA.
- the total run time is 2 minutes inject to inject.
- This Example illustrates the preparation of representative intermediates useful in the synthesis of 2-phenoxy pyrimidinone derivatives.
- reaction mixture is concentrated under vacuum and diluted by the addition of ice cold water.
- the reaction mixture is extracted with CH 2 Cl 2 (2 * 250 mL), washed with water
- l-(4-Fluorophenyl)-9-methyl-2-thioxo-l,2,3,9-tetrahydro-6//-purin-6-one hydrochloride (6.5 g, 0.021 mol) is suspended in a large excess of phosphorous oxychloride (150 mL) and heated to 135 0 C for 40 h. The reaction mixture is cooled, evaporated in vacuo, and azeotroped twice with toluene. The resulting sticky brown oil is dissolved in DCM (200 mL) then neutralized with saturated NaHCO 3 (aqueous). The aqueous layer is extracted with DCM (2 * 200 mL) and dried (MgSO 4 ).
- a mixture of 3-aminopyridine-2-carboxylic acid (6.4 g, 46.3 mmol) in 26 mL of EtOH and 8 mL of concentrated sulfuric acid is heated to reflux for 2 days. After cooling, the mixture is concentrated to about 15-20 mL and poured into 20 g of ice. The mixture is basified to pH 8-9 with concentrated NH 4 OH while cooling in an ice bath. The resulting brown precipitate is filtered off, and the filtrate is extracted with ether (4 x 60 mL). The combined ether extracts are washed with brine (4 * 60 mL), dried (Na 2 SO 4 ), filtered, and evaporated to give a yellow/brown solid.
- Ethyl 3-nitriloalaninate (26.9 g, 210 mmol) is dissolved in anhydrous ether (200 mL), and cooled in an ice/water bath.
- Acetic formic anhydride (prepared as a mixture as described above) is added dropwise. When the addition is finished, the reaction mixture is allowed to warm to RT and stirred at RT overnight. Most volatiles are removed in vacuo, and the residue solvents are removed by co-evaporation with toluene (100 mL x 4). The red oil obtained precipitates upon scratching in ether, and the resulting solids are recrystallized in ether to give the title compound as a white solid.
- Ethyl 5-amino-l,3-thiazole-4-carboxylate (1.05 g, 6.10 mmol) and 4-fluorophenyl isothiocyanate (0.93 g, 6.10 mmol) are added to pyridine (3.5 mL) and heated at 45 0 C for 15 h. Most of the solvent is removed under vacuum, and the resulting yellow solids are dissolved in CH 2 Cl 2 (150 mL) and washed with H 2 O (20 mL * 2) and brine (20 mL x 2). The CH 2 Cl 2 phase is dried over MgSO 4 , and the solvent is removed under reduced pressure.
- 6-(4-Fluorophenyl)-5-mercapto[l,3]thiazolo[5,4-d]pyrimidin-7(6H)-one hydrochloride (0.2 g, 0.633 mmol) is added to POCl 3 (10 mL), and the resulting solution is refluxed at 135 0 C for 41 h. Most of the volatiles are removed under reduced pressure and the residual solvent is co-evaporated with toluene (50 mL * 3). The dark solid obtained is dissolved in CH 2 Cl 2 (200 mL) and washed with saturated NaHCO 3 solution (100 mL * 5), brine (50 mL * T), and dried over MgSO 4 .
- This Example illustrates the preparation of VRl-transfected cells and VRl -containing membrane preparations for use in capsaicin binding assays (Example 5).
- a cDNA encoding full length human capsaicin receptor (SEQ ID NO:1, 2 or 3 of U.S. Patent No. 6,482,611) is subcloned in the plasmid pBK-CMV (Stratagene, La Jolla, CA) for recombinant expression in mammalian cells.
- Human embryonic kidney (HEK293) cells are transfected with the pBK-CMV expression construct encoding the full length human capsaicin receptor using standard methods. The transfected cells are selected for two weeks in media containing G418 (400 ⁇ g/ml) to obtain a pool of stably transfected cells. Independent clones are isolated from this pool by limiting dilution to obtain clonal stable cell lines for use in subsequent experiments.
- cells are seeded in T 175 cell culture flasks in media without antibiotics and grown to approximately 90% confluency. The flasks are then washed with PBS and harvested in PBS containing 5 mM EDTA. The cells are pelleted by gentle centrifugation and stored at -80 0 C until assayed. Previously frozen cells are disrupted with the aid of a tissue homogenizer in ice-cold
- HEPES homogenization buffer (5mM KCl 5, 5.8mM NaCl, 0.75mM CaCl 2 , 2mM MgCl 2 , 320 mM sucrose, and 10 mM HEPES pH 7.4). Tissue homogenates are first centrifuged for 10 minutes at 1000 * g (4°C) to remove the nuclear fraction and debris, and then the supernatant from the first centrifugation is further centrifuged for 30 minutes at 35,000 x g (4°C) to obtain a partially purified membrane fraction. Membranes are resuspended in the HEPES homogenization buffer prior to the assay. An aliquot of this membrane homogenate is used to determine protein concentration via the Bradford method (BIO-RAD Protein Assay Kit, #500-0001, BIO-RAD, Hercules, CA).
- This Example illustrates a representative assay of capsaicin receptor binding that may be used to determine the binding affinity of compounds for the capsaicin (VRl ) receptor.
- RTX Resiniferatoxin
- RTX (37 Ci/mmol) is synthesized by and obtained from the Chemical Synthesis and Analysis Laboratory, National Cancer Institute-Frederick Cancer Research and Development Center, Frederick, MD. [ 3 H] RTX may also be obtained from commercial vendors (e.g., Amersham Pharmacia Biotech, Inc.; Piscataway, NJ).
- the membrane homogenate of Example 4 is centrifuged as before and resuspended to a protein concentration of 333 ⁇ g/ml in homogenization buffer. Binding assay mixtures are set up on ice and contain [ 3 H]RTX (specific activity 2200 mCi/ml), 2 ⁇ l non-radioactive test compound, 0.25 mg/ml bovine serum albumin (Cohn fraction V), and 5 * 10 4 - 1 x 10 s VRl- transfected cells. The final volume is adjusted to 500 ⁇ l (for competition binding assays) or 1,000 ⁇ l (for saturation binding assays) with the ice-cold HEPES homogenization buffer solution (pH 7.4) described above.
- Non-specific binding is defined as that occurring in the presence of 1 ⁇ M non-radioactive RTX (Alexis Corp.; San Diego, CA).
- RTX non-radioactive RTX
- [ 3 H]RTX is added in the concentration range of 7-1,000 pM, using 1 to 2 dilutions. Typically 11 concentration points are collected per saturation binding curve.
- Equilibrium binding parameters are determined by fitting the allosteric Hill equation to the measured values with the aid of the computer program FIT P (Biosoft, Ferguson, MO) as described by Szallasi, et al. (1993) J. Pharmacol. Exp. Ther. 266:678-683.
- Compounds provided herein generally exhibit Kj values for capsaicin receptor of less than 1 ⁇ M, 100 nM, 50 nM, 25 nM, 10 nM, or InM in this assay.
- This Example illustrates representative calcium mobilization assays for use in evaluating test compounds for agonist and antagonist activity.
- the culture medium is emptied from the 96 well plates and FLUO-3 AM calcium sensitive dye (Molecular Probes, Eugene, OR) is added to each well (dye solution: 1 mg FLUO-3 AM, 440 ⁇ L DMSO and 440 ⁇ l 20% pluronic acid in DMSO, diluted 1:250 in Krebs-Ringer HEPES (KRH) buffer (25 mM HEPES, 5 mM KCl, 0.96 mM NaH 2 PO 4 , 1 mM MgSO 4 , 2 mM CaCl 2 , 5 mM glucose, 1 mM probenecid, pH 7.4), 50 ⁇ l diluted solution per well).
- KRH Krebs-Ringer HEPES
- capsaicin in KRH buffer 100 ⁇ l capsaicin in KRH buffer is automatically transferred by the FLIPR instrument to each well.
- Capsaicin-induced calcium mobilization is monitored using either FLUOROSKAN ASCENT (Labsystems; Franklin, MA) or FLEPR (fluorometric imaging plate reader system; Molecular Devices, Sunnyvale, CA) instruments. Data obtained between 30 and 60 seconds after agonist application are used to generate an 8-point concentration response curve, with final capsaicin concentrations of 1 nM to 3 ⁇ M.
- y is the maximum fluorescence signal
- x is the concentration of the agonist or antagonist (in this case, capsaicin)
- a is the E n ⁇ x
- b corresponds to the EC 50 value
- c is the Hill coefficient.
- Test compounds are dissolved in DMSO, diluted in KRH buffer, and immediately added to cells prepared as described above. 100 nM capsaicin (an approximate EC 90 concentration) is also added to cells in the same 96-well plate as a positive control. The final concentration of test compounds in the assay wells is between 0.1 nM and 5 ⁇ M.
- the ability of a test compound to act as an agonist of the capsaicin receptor is determined by measuring the fluorescence response of cells expressing capsaicin receptors elicited by the compound as function of compound concentration. This data is fit as described above to obtain the EC 50 , which is generally less than 1 micromolar, preferably less than 100 nM, and more preferably less than 10 nM.
- the extent of efficacy of each test compound is also determined by calculating the response elicited by a concentration of test compound (typically 1 ⁇ M) relative to the response elicited by 100 nM capsaicin. This value, called Percent of Signal (POS), is calculated by the following equation:
- Agonists of the human capsaicin receptor generally elicit detectable responses at concentrations less than 100 ⁇ M, or preferably at concentrations less than 1 ⁇ M, or most preferably at concentrations less than 10 nM.
- Extent of efficacy at human capsaicin receptor is preferably greater than 30 POS, more preferably greater than 80 POS at a concentration of 1 ⁇ M.
- Certain agonists are essentially free of antagonist activity as demonstrated by the absence of detectable antagonist activity in the assay described below at compound concentrations below 4 nM, more preferably at concentrations below 10 ⁇ M and most preferably at concentrations less than or equal to 100 ⁇ M.
- Test compounds are dissolved in DMSO, diluted in 20 ⁇ l KRH buffer so that the final concentration of test compounds in the assay well is between 1 ⁇ M and 5 ⁇ M, and added to cells prepared as described above.
- the 96 well plates containing prepared cells and test compounds are incubated in the dark, at room temperature for 0.5 to 6 hours. It is important that the incubation not continue beyond 6 hours.
- 100 ⁇ l capsaicin in KRH buffer at twice the EC 50 concentration determined as described above is automatically added by the FLIPR instrument to each well of the 96 well plate for a final sample volume of 200 ⁇ l and a final capsaicin concentration equal to the EC 50 .
- the final concentration of test compounds in the assay wells is between 1 ⁇ M and 5 ⁇ M.
- Antagonists of the capsaicin receptor decrease this response by at least about 20%, preferably by at least about 50%, and most preferably by at least 80%, as compared to matched control (i.e., cells treated with capsaicin at twice the EC 50 concentration in the absence of test compound), at a concentration of 10 micromolar or less, preferably 1 micromolar or less.
- the concentration of antagonist required to provide a 50% decrease, relative to the response observed in the presence of capsaicin and without antagonist, is the IC 50 for the antagonist, and is preferably below 1 micromolar, 100 nanomolar, 10 nanomolar or 1 nanomolar.
- the data is analyzed as follows. First, the average maximum relative fluorescent unit (RFU) response from the negative control wells (no agonist) is subtracted from the maximum response detected for each of the other experimental wells. Second, average maximum RFU response is calculated for the positive control wells (agonist wells). Then, percent inhibition for each compound tested is calculated using the equation:
- the % inhibition data is plotted as a function of test compound concentration and test compound IC 50 is determined using, for example, KALEID AGRAPH software (Synergy
- test compound IC 50 the concentration required to provide a 50% decrease, relative to the response observed in the presence of agonist and without antagonist
- m 3 is the Hill coefficient.
- test compound IC 50 is determined using a linear regression in which x is ln(concentration of test compound) and y is ln(percent inhibition/(100 - percent inhibition). Data with a percent inhibition that is greater than 90% or less than 15% are rejected and are not used in the regression. The IC 50 calculated in this fashion is e (' intercept/slope)
- Certain preferred VRl modulators are antagonists that are essentially free of agonist activity as demonstrated by the absence of detectable agonist activity in the assay described above at compound concentrations below 4 nM, more preferably at concentrations below 10 uM and most preferably at concentrations less than or equal to 100 ⁇ M.
- This Example illustrates a representative dorsal root ganglian cell assay for evaluating VRl antagonist or agonist activity of a compound.
- DRG are dissected from neonatal rats, dissociated and cultured using standard methods (Aguayo and White (1992) Brain Research 570:61-67). After 48 hour incubation, cells are washed once and incubated for 30-60 minutes with the calcium sensitive dye Fluo 4 AM (2.5-10 ug/ml; TefLabs, Austin, TX). Cells are then washed once. Addition of capsaicin to the cells results in a VRl -dependent increase in intracellular calcium levels which is monitored by a change in Fluo-4 fluorescence with a fluorometer. Data are collected for 60- 180 seconds to determine the maximum fluorescent signal.
- Antagonists of the capsaicin receptor preferably have an IC 50 below 1 micromolar, 100 nanomolar, 10 nanomolar or 1 nanomolar.
- EC 50 or concentration required to achieve 50% of the maximum signal for a capsaicin-activated response, is preferably below 1 micromolar, below 100 nanomolar or below 10 nanomolar.
- This Example illustrates representative methods for assessing the degree of pain relief provided by a compound.
- the following methods may be used to assess pain relief.
- Testing is initiated with the middle hair of the series with subsequent filaments being applied in consecutive fashion, ascending or descending, depending on whether a negative or positive response, respectively, is obtained with the initial filament.
- Compounds are effective in reversing or preventing mechanical allodynia-like symptoms if rats treated with such compounds require stimulation with a Von Frey filament of higher rigidity strength to provoke a positive allodynic response as compared to control untreated or vehicle treated rats.
- testing of an animal in chronic pain may be done before and after compound administration.
- an effective compound results in an increase in the rigidity of the filament needed to induce a response after treatment, as compared to the filament that induces a response before treatment or in an animal that is also in chronic pain but is left untreated or is treated with vehicle.
- Test compounds are administered before or after onset of pain. When a test compound is administered after pain onset, testing is performed 10 minutes to three hours after administration.
- Test compound produces a reduction in mechanical hyperalgesia if there is a statistically significant decrease in the duration of hindpaw withdrawal.
- Test compound may be administered before or after onset of pain. For compounds administered after pain onset, testing is performed 10 minutes to three hours after administration.
- Thermal hyperalgesia an exaggerated response to noxious thermal stimulus
- Test compound produces a reduction in thermal hyperalgesia if there is a statistically significant increase in the time to hindpaw withdrawal ⁇ i.e., the thermal threshold to response or latency is increased).
- Test compound may be administered before or after onset of pain.
- testing is performed 10 minutes to three hours after administration.
- Pain Models Pain may be induced using any of the following methods, to allow testing of analgesic efficacy of a compound.
- compounds provided herein result in a statistically significant reduction in pain as determined by at least one of the previously described testing methods, using male SD rats and at least one of the following models.
- ACUTE INFLAMMATORY PAIN MODEL Acute inflammatory pain is induced using the carrageenan model essentially as described by Field et al. (1997) Br. J. Pharmacol. 121(8):1513-1522. 100-200 ⁇ l of 1-2% carrageenan solution is injected into the rats' hind paw. Three to four hours following injection, the animals' sensitivity to thermal and mechanical stimuli is tested using the methods described above. A test compound (0.01 to 50 mg/kg) is administered to the animal, prior to testing, or prior to injection of carrageenan. The compound can be administered orally or through any parenteral route, or topically on the paw. Compounds that relieve pain in this model result in a statistically significant reduction in mechanical allodynia and/or thermal hyperalgesia.
- CHRONIC INFLAMMATORY PA ⁇ N MODEL Chronic inflammatory pain is induced using one of the following protocols: 1. Essentially as described by Bertorelli et al. (1999) Br. J. Pharmacol. 128(6):1252- 1258, and Stein et al. (1998) Pharmacol. Biochem. Behav. 31(2):455-51, 200 ⁇ l Complete Freund's Adjuvant (0.1 mg heat killed and dried M. Tuberculosis) is injected to the rats' hind paw: 100 ⁇ l into the dorsal surface and 100 ⁇ l into the plantar surface.
- rats are tested for thermal hyperalgesia, mechanical allodynia and mechanical hyperalgesia as described above.
- rats are tested on days 5, 6, and 7 following CFA injection.
- animals are treated with a test compound, morphine or vehicle.
- An oral dose of morphine of 1-5 mg/kg is suitable as positive control.
- a dose of 0.01-50 mg/kg of test compound is used.
- Compounds can be administered as a single bolus prior to testing or once or twice or three times daily, for several days prior to testing. Drugs are administered orally or through any parenteral route, or applied topically to the animal.
- MPE Percent Maximum Potential Efficacy
- Chronic neuropathic pain is induced using the chronic constriction injury (CCI) to the rat's sciatic nerve essentially as described by Bennett and Xie (1988) Pain 33:87-107.
- Rats are anesthetized (e.g. with an intraperitoneal dose of 50-65 mg/kg pentobarbital with additional doses administered as needed).
- the lateral aspect of each hind limb is shaved and disinfected.
- aseptic technique an incision is made on the lateral aspect of the hind limb at the mid thigh level.
- the biceps femoris is bluntly dissected and the sciatic nerve is exposed.
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009525630A JP5421108B2 (en) | 2006-08-23 | 2007-08-22 | 2-phenoxypyrimidinone analogs |
| ES07811503T ES2399928T3 (en) | 2006-08-23 | 2007-08-22 | 2-phenoxypyrimidinone analogues |
| EP07811503A EP2061794B1 (en) | 2006-08-23 | 2007-08-22 | 2-phenoxy pyrimidinone analogues |
| CA2660957A CA2660957C (en) | 2006-08-23 | 2007-08-22 | 2-phenoxy pyrimidinone analogues |
| MX2009002066A MX2009002066A (en) | 2006-08-23 | 2007-08-22 | 2-phenoxy pyrimidinone analogues. |
| EA200900330A EA200900330A1 (en) | 2006-08-23 | 2007-08-22 | ANALOGY 2-PHENOXYPYRIMIDINONE |
| AU2007288203A AU2007288203B2 (en) | 2006-08-23 | 2007-08-22 | 2-phenoxy pyrimidinone analogues |
| BRPI0716582-0A2A BRPI0716582A2 (en) | 2006-08-23 | 2007-08-22 | 2-PHENOXY PYRIMIDINONE ANALOGS |
| IL197043A IL197043A0 (en) | 2006-08-23 | 2009-02-15 | 2-phenoxy pyrimidinone analogues |
| NO20091182A NO20091182L (en) | 2006-08-23 | 2009-03-20 | 2 fenoksypyrimidinonanaloger |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US82325806P | 2006-08-23 | 2006-08-23 | |
| US60/823,258 | 2006-08-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008024438A2 true WO2008024438A2 (en) | 2008-02-28 |
| WO2008024438A3 WO2008024438A3 (en) | 2008-11-27 |
Family
ID=39107411
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/018654 WO2008024438A2 (en) | 2006-08-23 | 2007-08-22 | 2-phenoxy pyrimidinone analogues |
Country Status (22)
| Country | Link |
|---|---|
| US (2) | US8003656B2 (en) |
| EP (1) | EP2061794B1 (en) |
| JP (1) | JP5421108B2 (en) |
| KR (1) | KR20090043583A (en) |
| CN (2) | CN104292229A (en) |
| AU (1) | AU2007288203B2 (en) |
| BR (1) | BRPI0716582A2 (en) |
| CA (1) | CA2660957C (en) |
| CO (1) | CO6150167A2 (en) |
| CR (1) | CR10675A (en) |
| EA (1) | EA200900330A1 (en) |
| ES (1) | ES2399928T3 (en) |
| GT (1) | GT200900040A (en) |
| IL (1) | IL197043A0 (en) |
| MA (1) | MA30720B1 (en) |
| MX (1) | MX2009002066A (en) |
| NI (1) | NI200900023A (en) |
| NO (1) | NO20091182L (en) |
| SV (1) | SV2009003173A (en) |
| TW (1) | TW200825089A (en) |
| WO (1) | WO2008024438A2 (en) |
| ZA (1) | ZA200901163B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0509573D0 (en) * | 2005-05-11 | 2005-06-15 | Merck Sharp & Dohme | Therapeutic compounds |
| WO2008024438A2 (en) | 2006-08-23 | 2008-02-28 | Neurogen Corporation | 2-phenoxy pyrimidinone analogues |
| RS58960B1 (en) | 2013-09-09 | 2019-08-30 | Peloton Therapeutics Inc | Aryl ethers and uses thereof |
| WO2016023830A1 (en) * | 2014-08-11 | 2016-02-18 | Hydra Biosciences, Inc. | Pyrido[2,3-d]pyrimidine-2,4(1h,3h)-dione derivatives |
| US10807948B2 (en) | 2015-03-11 | 2020-10-20 | Peloton Therapeutics, Inc. | Aromatic compounds and uses thereof |
| US10512626B2 (en) | 2015-03-11 | 2019-12-24 | Peloton Therapeautics, Inc. | Compositions for use in treating glioblastoma |
| US10155726B2 (en) | 2015-03-11 | 2018-12-18 | Peloton Therapeutics, Inc. | Substituted pyridines and uses thereof |
| WO2016145032A1 (en) | 2015-03-11 | 2016-09-15 | Peloton Therapeutics, Inc. | Compositions for use in treating pulmonary arterial hypertension |
| WO2016168510A1 (en) | 2015-04-17 | 2016-10-20 | Peloton Therapeutics, Inc. | Combination therapy of a hif-2-alpha inhibitor and an immunotherapeutic agent and uses thereof |
| WO2020146449A1 (en) * | 2019-01-08 | 2020-07-16 | Texas Tech University System | Small molecule analogs of the protein e4orf1 in the treatment and prevention of metabolic disorders |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5124331A (en) | 1988-03-02 | 1992-06-23 | Yoshitomi Pharmaceutical Industries, Ltd. | 3,4-dihydrothieno[2,3-d]pyrimidine compounds and their pharmaceutical use |
| US5202328A (en) | 1991-03-06 | 1993-04-13 | Merck & Co., Inc. | Substituted fused pyrimidinones |
| ATE227293T1 (en) | 1996-05-15 | 2002-11-15 | Pfizer | 2,3-DISUBSTITUTED-(5,6)-HETEROARYL CONDENSED - PYRIMIDINE-4-ONE |
| JP2001524121A (en) | 1997-05-08 | 2001-11-27 | アグレボ・ユー・ケイ・リミテツド | Fungicide |
| US6627755B1 (en) | 1997-06-09 | 2003-09-30 | Pfizer Inc | Quinazolin-4-one AMPA antagonists |
| IL125950A0 (en) | 1997-09-05 | 1999-04-11 | Pfizer Prod Inc | Methods of administering ampa receptor antagonists to treat dyskinesias associated with dopamine agonist therapy |
| US6323208B1 (en) | 1997-09-05 | 2001-11-27 | Pfizer Inc | Atropisomers of 2,3-disubstituted-(5.6)-heteroaryl fused-pyrimidin-4-ones |
| WO2002026718A2 (en) | 2000-09-29 | 2002-04-04 | Millennium Pharmaceutical, Inc. | Bicyclic pyrimidin-4-one based inhibitors of factor xa |
| MXPA03004926A (en) | 2001-02-14 | 2005-02-14 | Warner Lambert Co | Thieno 2,3-d pyrimidindione derivatives as matrix metalloproteinase inhibitors. |
| HUP0303623A2 (en) | 2001-03-26 | 2004-03-01 | Novartis Ag. | Fused pyridine derivatives, process for their preparation, pharmaceutical compositions containing them for treating pain as vanilloid receptor antagonist |
| ES2242899T3 (en) * | 2001-09-28 | 2005-11-16 | Daimlerchrysler Ag | DOUBLE / TRIPLE LIGHT CONSTRUCTION STEEL OF HIGH RESISTANCE AND ITS USE. |
| EP1461311A2 (en) | 2001-12-26 | 2004-09-29 | Bayer HealthCare AG | Urea derivatives as vr1-antagonists |
| ATE533743T1 (en) | 2002-05-17 | 2011-12-15 | Janssen Pharmaceutica Nv | UREA DERIVATIVES OF AMINOTETRALIN AS MODULATORS OF THE VANILLOID RECEPTOR VR1 |
| AU2003238157A1 (en) | 2002-06-18 | 2003-12-31 | Sankyo Company, Limited | Fused-ring pyrimidin-4(3h)-one derivatives, processes for the preparation and uses thereof |
| WO2004028440A2 (en) | 2002-09-24 | 2004-04-08 | Bayer Healthcare Ag | Vr1 antagonists for the treatment of urological disorders |
| GB0223730D0 (en) | 2002-10-11 | 2002-11-20 | Novartis Ag | Organic compounds |
| AU2003301662A1 (en) | 2002-10-21 | 2004-05-13 | Bristol-Myers Squibb Company | Quinazolinones and derivatives thereof as factor xa inhibitors |
| US7687625B2 (en) | 2003-03-25 | 2010-03-30 | Takeda Pharmaceutical Company Limited | Dipeptidyl peptidase inhibitors |
| WO2005049613A1 (en) * | 2003-11-14 | 2005-06-02 | Merck Sharp & Dohme Limited | Bicyclic pyrimidin-4-(3h)-ones and analogues and derivatives thereof which modulate the function of the vanilloid-1 receptor (vr1) |
| GB0326633D0 (en) | 2003-11-14 | 2003-12-17 | Merck Sharp & Dohme | Therapeutic agents |
| US7544803B2 (en) * | 2004-01-23 | 2009-06-09 | Amgen Inc. | Vanilloid receptor ligands and their use in treatments |
| GB0412769D0 (en) | 2004-06-08 | 2004-07-07 | Novartis Ag | Organic compounds |
| AR051596A1 (en) * | 2004-10-26 | 2007-01-24 | Irm Llc | CONDENSED HETEROCICLIC COMPOUNDS NITROGENATED AS INHIBITORS OF THE ACTIVITY OF THE CANABINOID RECEIVER 1; PHARMACEUTICAL COMPOSITIONS THAT CONTAIN THEM AND THEIR EMPLOYMENT IN THE PREPARATION OF MEDICINES FOR THE TREATMENT OF FOOD DISORDERS |
| WO2006122200A1 (en) * | 2005-05-11 | 2006-11-16 | Merck Sharp & Dohme Limited | 2,3-substituted fused bicyclic pyrimidin-4(3h)-ones modulating the function of the vanilloid-1 receptor (vr1) |
| GB0509573D0 (en) | 2005-05-11 | 2005-06-15 | Merck Sharp & Dohme | Therapeutic compounds |
| CN1318429C (en) * | 2005-07-25 | 2007-05-30 | 华中师范大学 | Substitution thieno[3',2':5,6]-pyridino[4,3-d]-pyrimidine-4(3H)-ketone and preparation method |
| US7893260B2 (en) | 2005-11-04 | 2011-02-22 | Hydra Biosciences, Inc. | Substituted quinazolin-4-one compounds for antagonizing TRPV3 function |
| WO2008024438A2 (en) | 2006-08-23 | 2008-02-28 | Neurogen Corporation | 2-phenoxy pyrimidinone analogues |
| AU2007288198A1 (en) | 2006-08-23 | 2008-02-28 | Neurogen Corporation | Haloalkyl-substituted pyrimidinone derivatives |
| AU2007325940A1 (en) | 2006-11-06 | 2008-06-05 | Neurogen Corporation | Cis-cyclohexyl substituted pyrimidinone derivatives |
| WO2008156607A1 (en) | 2007-06-12 | 2008-12-24 | Neurogen Corporation | Substituted pyrimidinones |
| WO2009100403A1 (en) | 2008-02-07 | 2009-08-13 | Neurogen Corporation | Substituted aryl pyrimidinones |
| WO2009121036A2 (en) | 2008-03-27 | 2009-10-01 | Neurogen Corporation | Substituted aryl pyrimidinone derivatives |
-
2007
- 2007-08-22 WO PCT/US2007/018654 patent/WO2008024438A2/en active Application Filing
- 2007-08-22 US US11/895,286 patent/US8003656B2/en active Active
- 2007-08-22 JP JP2009525630A patent/JP5421108B2/en not_active Expired - Fee Related
- 2007-08-22 CA CA2660957A patent/CA2660957C/en not_active Expired - Fee Related
- 2007-08-22 EP EP07811503A patent/EP2061794B1/en active Active
- 2007-08-22 CN CN201410468406.2A patent/CN104292229A/en active Pending
- 2007-08-22 CN CNA2007800309530A patent/CN101506210A/en active Pending
- 2007-08-22 BR BRPI0716582-0A2A patent/BRPI0716582A2/en not_active IP Right Cessation
- 2007-08-22 ES ES07811503T patent/ES2399928T3/en active Active
- 2007-08-22 TW TW096131019A patent/TW200825089A/en unknown
- 2007-08-22 MX MX2009002066A patent/MX2009002066A/en unknown
- 2007-08-22 AU AU2007288203A patent/AU2007288203B2/en not_active Ceased
- 2007-08-22 ZA ZA200901163A patent/ZA200901163B/en unknown
- 2007-08-22 KR KR1020097005810A patent/KR20090043583A/en not_active Application Discontinuation
- 2007-08-22 EA EA200900330A patent/EA200900330A1/en unknown
-
2009
- 2009-02-15 IL IL197043A patent/IL197043A0/en unknown
- 2009-02-20 NI NI200900023A patent/NI200900023A/en unknown
- 2009-02-20 SV SV2009003173A patent/SV2009003173A/en not_active Application Discontinuation
- 2009-02-23 GT GT200900040A patent/GT200900040A/en unknown
- 2009-03-18 CO CO09028167A patent/CO6150167A2/en unknown
- 2009-03-20 NO NO20091182A patent/NO20091182L/en not_active Application Discontinuation
- 2009-03-20 CR CR10675A patent/CR10675A/en not_active Application Discontinuation
- 2009-03-23 MA MA31732A patent/MA30720B1/en unknown
-
2011
- 2011-08-22 US US13/215,195 patent/US8759361B2/en active Active
Non-Patent Citations (2)
| Title |
|---|
| None |
| See also references of EP2061794A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
Also Published As
| Publication number | Publication date |
|---|---|
| IL197043A0 (en) | 2009-11-18 |
| CR10675A (en) | 2009-04-17 |
| CN101506210A (en) | 2009-08-12 |
| US8759361B2 (en) | 2014-06-24 |
| KR20090043583A (en) | 2009-05-06 |
| AU2007288203A1 (en) | 2008-02-28 |
| MX2009002066A (en) | 2009-03-06 |
| EP2061794B1 (en) | 2012-12-05 |
| SV2009003173A (en) | 2010-08-10 |
| AU2007288203B2 (en) | 2013-01-17 |
| NO20091182L (en) | 2009-05-14 |
| CA2660957C (en) | 2016-10-11 |
| CA2660957A1 (en) | 2008-02-28 |
| US8003656B2 (en) | 2011-08-23 |
| NI200900023A (en) | 2010-03-05 |
| CN104292229A (en) | 2015-01-21 |
| MA30720B1 (en) | 2009-09-01 |
| EP2061794A2 (en) | 2009-05-27 |
| JP5421108B2 (en) | 2014-02-19 |
| GT200900040A (en) | 2010-03-16 |
| US20120208829A1 (en) | 2012-08-16 |
| TW200825089A (en) | 2008-06-16 |
| WO2008024438A3 (en) | 2008-11-27 |
| JP2010502576A (en) | 2010-01-28 |
| ZA200901163B (en) | 2010-08-25 |
| CO6150167A2 (en) | 2010-04-20 |
| BRPI0716582A2 (en) | 2013-10-08 |
| US20090069347A1 (en) | 2009-03-12 |
| ES2399928T3 (en) | 2013-04-04 |
| EP2061794A4 (en) | 2010-09-08 |
| EA200900330A1 (en) | 2009-08-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2061794B1 (en) | 2-phenoxy pyrimidinone analogues | |
| US20080090845A1 (en) | Haloalkyl-substituted pyrimidinone derivatives | |
| AU2006206246A1 (en) | Heteroaryl substituted piperazinyl-pyridine analogues | |
| US20080175794A1 (en) | Substituted Pyridazinyl- and Pyrimidinyl-Quinolin-4-Ylamine Analogues | |
| US20080085901A1 (en) | Heteroaryl Substituted Quinolin-4-Ylamine Analogues | |
| US20100008866A1 (en) | Cis-cyclohexyl substituted pyrimidinone derivatives | |
| EP1811845A2 (en) | Substituted biaryl quinolin-4-ylamine analogues | |
| US20070219203A1 (en) | Arylalkylamino-substituted quinazoline analogues | |
| WO2005023807A2 (en) | 4 - heterobicyclyamino - substituted quinazolines and analogues therof as capsaicin - antagonists | |
| CA2557667A1 (en) | Aryl substituted purine analogues | |
| WO2008156607A1 (en) | Substituted pyrimidinones | |
| AU2005317176A1 (en) | Piperazinyl-pyridine analogues | |
| CA2557545A1 (en) | Substituted 5,12-diaza-benzoanthracene analogues | |
| WO2009121036A2 (en) | Substituted aryl pyrimidinone derivatives | |
| WO2009100403A1 (en) | Substituted aryl pyrimidinones | |
| AU2004259676A1 (en) | Aryl-substituted benzo(d)isothiazol-3-ylamine analogues as capsaicin receptor modulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780030953.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07811503 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 197043 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2660957 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12009500315 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007288203 Country of ref document: AU Ref document number: 575006 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009525630 Country of ref document: JP Ref document number: MX/A/2009/002066 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09028167 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1834/DELNP/2009 Country of ref document: IN Ref document number: 200900330 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: CR2009-010675 Country of ref document: CR Ref document number: 1020097005810 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: DZP2009000159 Country of ref document: DZ |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007811503 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: PI0716582 Country of ref document: BR Kind code of ref document: A2 Effective date: 20090225 |