WO2008024398A2 - Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators - Google Patents
Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators Download PDFInfo
- Publication number
- WO2008024398A2 WO2008024398A2 PCT/US2007/018571 US2007018571W WO2008024398A2 WO 2008024398 A2 WO2008024398 A2 WO 2008024398A2 US 2007018571 W US2007018571 W US 2007018571W WO 2008024398 A2 WO2008024398 A2 WO 2008024398A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- isoquinolin
- dihydro
- dimethylbutanamide
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
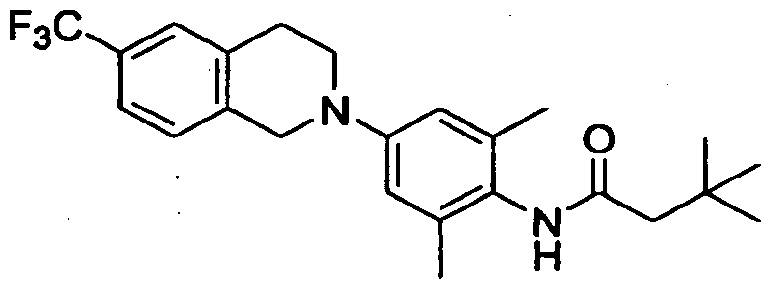
- 0 *CCNC(Nc(c(*)c1)c(*)cc1N1Cc2ccc(CCNC3)c3c2CC1)=* Chemical compound *CCNC(Nc(c(*)c1)c(*)cc1N1Cc2ccc(CCNC3)c3c2CC1)=* 0.000 description 7
- HKUZIWRBRAOLCN-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C(F)(F)F)c1)ccc1Br)=O Chemical compound CC(C)(C)CC(Nc(c(C(F)(F)F)c1)ccc1Br)=O HKUZIWRBRAOLCN-UHFFFAOYSA-N 0.000 description 1
- ABTRCHMHHBOXFV-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C(F)(F)F)c1)ccc1N(CC1)Cc(cc2)c1cc2F)=O Chemical compound CC(C)(C)CC(Nc(c(C(F)(F)F)c1)ccc1N(CC1)Cc(cc2)c1cc2F)=O ABTRCHMHHBOXFV-UHFFFAOYSA-N 0.000 description 1
- WJLOXTPZZKOJPH-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C(F)(F)F)c1)ccc1N1Cc2cc(C(F)(F)F)ccc2CC1)=O Chemical compound CC(C)(C)CC(Nc(c(C(F)(F)F)c1)ccc1N1Cc2cc(C(F)(F)F)ccc2CC1)=O WJLOXTPZZKOJPH-UHFFFAOYSA-N 0.000 description 1
- QRHHSFCJTGZUMH-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C(F)(F)F)cc(Br)c1)c1Cl)=O Chemical compound CC(C)(C)CC(Nc(c(C(F)(F)F)cc(Br)c1)c1Cl)=O QRHHSFCJTGZUMH-UHFFFAOYSA-N 0.000 description 1
- JURDKTFYHWIOLE-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C(F)(F)F)cc(N1Cc2ccccc2CC1)c1)c1Cl)=O Chemical compound CC(C)(C)CC(Nc(c(C(F)(F)F)cc(N1Cc2ccccc2CC1)c1)c1Cl)=O JURDKTFYHWIOLE-UHFFFAOYSA-N 0.000 description 1
- XEVJQEIJAXJBKV-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C)c1)c(C)cc1Br)=O Chemical compound CC(C)(C)CC(Nc(c(C)c1)c(C)cc1Br)=O XEVJQEIJAXJBKV-UHFFFAOYSA-N 0.000 description 1
- FJNPZKZPWVVSON-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C)c1)c(C)cc1N(CCc1c2)Cc1ccc2F)=O Chemical compound CC(C)(C)CC(Nc(c(C)c1)c(C)cc1N(CCc1c2)Cc1ccc2F)=O FJNPZKZPWVVSON-UHFFFAOYSA-N 0.000 description 1
- NWQBIMDKXBZSID-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(C)c1)c(C)cc1N1Cc2ccc(C(F)(F)F)cc2CC1)=S Chemical compound CC(C)(C)CC(Nc(c(C)c1)c(C)cc1N1Cc2ccc(C(F)(F)F)cc2CC1)=S NWQBIMDKXBZSID-UHFFFAOYSA-N 0.000 description 1
- JLKZQPXSHYGHBK-UHFFFAOYSA-N CC(C)(C)CC(Nc(c(Cl)c1)ccc1N1Cc2ccc(C(F)(F)F)cc2CC1)=O Chemical compound CC(C)(C)CC(Nc(c(Cl)c1)ccc1N1Cc2ccc(C(F)(F)F)cc2CC1)=O JLKZQPXSHYGHBK-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/22—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the nitrogen-containing ring
- C07D217/24—Oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/12—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring
- C07D217/14—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring other than aralkyl radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/12—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/12—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring
- C07D217/14—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring other than aralkyl radicals
- C07D217/16—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring other than aralkyl radicals substituted by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/12—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with radicals, substituted by hetero atoms, attached to carbon atoms of the nitrogen-containing ring
- C07D217/18—Aralkyl radicals
Definitions
- This invention concerns novel compounds that activate or otherwise modulate voltage-gated potassium channels.
- the compounds are useful for the treatment and prevention of diseases and disorders which are affected by modulation of potassium ion channels.
- One such condition is seizure disorders.
- Epilepsy is a well-known neurological disease, found in about 3% of the population. Approximately 30% of patients with epilepsy do not respond to currently available therapies. Such unfortunate patients — who number hundreds of thousands of people world-wide — must contend with both uncontrolled seizures and the resulting narrowing of their options in such crucial areas of life as health insurance, employment, and driving.
- Retigabine N-[2-amino-4-(4-fluorobenzylamino)phenyl]carbamic acid, ethyl ester
- U.S. Patent No. 5,384,330 has been found to be an effective treatment of seizure disorders and has also been found useful in treating pain.
- Retigabine has been found to be particularly potent in models for the drug-refractory types of epilepsy. Bialer, M. et al., Epilepsy Research 1999, 34, 1-41; Blackburn-Munro and Jensen, Eur. J. Pharmacol. 2003, 460, 109-116; Wickenden, A.D. et al., Expert Opin. Ther. Patents, 2004, 14(4).
- Retigabine has been shown to increase the conductance of the channels at the resting membrane potential, with a possible mechanism involving binding of the activation gate of the KCNQ 2/3 channel. Wuttke, T. V., et al., MoI. Pharmacol. 2005. Additionally, retigabine has been shown to increase neuronal M currents and to increase the channel open probability of KCNQ 2/3 channels. Delmas, P. and Brown, D.A. Nat. Revs Neurosci., vol. 6, 2005, 850-62; Tatulian, L. and Brown, D.A., ./. Physiol, (2003) 549, 57-63.
- the seizure type that has been most resistant to therapy is the so-called "complex partial seizure.”
- Retigabine is active in several seizure models, including, as indicated above, models for drug-refractory epilepsy. Because of retigabine's broad spectrum of activity and its unusual molecular mechanism, there is hope that retigabine will be effective in management of several seizure types, including the complex partial seizure, which have been resistant to treatment.
- Porter, R. J., Nohria, V., and Rundfeldt, C Neurotherapeutics, 2007, vol. 4, 149-154.
- retigabine as a potassium channel opener has inspired a search among compounds with structural features in common with retigabine for other compounds which can affect the opening of, or otherwise modulate, potassium ion channels.
- tetrahydroisoquinoline derivatives are, of course, benzyl amines which are restricted to particular conformations because the benzylic nitrogen is a member of a second ring fused to the phenyl ring.
- the present inventors have further discovered that replacement of the primary amino group of retigabine with substituents like halogen, C 1 -C 3 alkyl, OC 1 -C 3 alkyl, and trifluoromethyl also confers surprising and desirable properties.
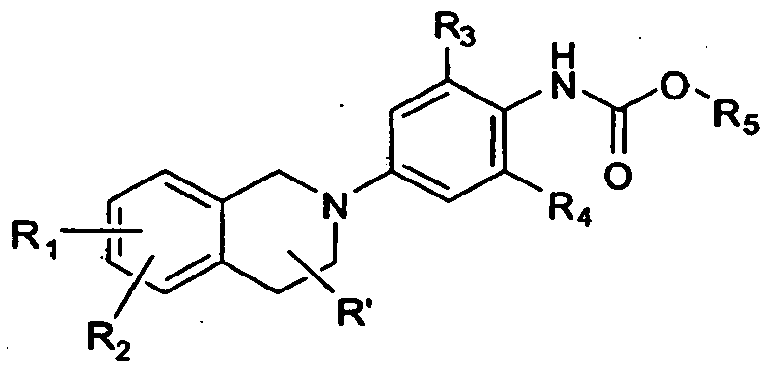
- this invention provides or contemplates a compound of formula IA
- this invention provides or contemplates a composition
- a pharmaceutically acceptable carrier or diluent and at least one of the following: a pharmaceutically effective amount of a compound of formula IA, a pharmaceutically acceptable salt of a compound of formula IA, a pharmaceutically acceptable solvate of a compound of formula IA, and a pharmaceutically acceptable ester of a compound of formula IA.
- this invention provides or contemplates a pediatric pharmaceutical composition
- a pediatric pharmaceutical composition comprising a pharmaceutically acceptable carrier or diluent, a syrup for pediatric use, and at least one of the following: a pharmaceutically effective amount of a compound of formula IA, a pharmaceutically acceptable salt of a compound of formula IA, a pharmaceutically acceptable ester of a compound of formula IA, and a pharmaceutically acceptable solvate of a compound of formula IA.
- this invention provides or contemplates a chewable tablet, suitable for pediatric pharmaceutical use, comprising a pharmaceutically acceptable carrier or diluent, and at least one of the following: a pharmaceutically effective amount of a compound of formula IA, a pharmaceutically acceptable salt of a compound of formula IA, a pharmaceutically acceptable solvate of a compound of formula IA, and a pharmaceutically acceptable ester of a compound of formula IA.
- this invention provides or contemplates a method of preventing or treating a disease or disorder which is affected by activation voltage-gated potassium channels, comprising administering to a patient in need thereof a therapeutically effective amount of a compound of formula IA or a salt or ester or solvate thereof.
- This invention includes all tautomers and salts of compounds of this invention. This invention also includes all compounds of this invention where one or more atoms are replaced by a radioactive isotope thereof.
- this invention provides or contemplates a compound of formula IA, where q is zero and Rs is Ci-C ⁇ alkyl, or CCHR ⁇ VCj-C ⁇ cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R' is H, methyl, ethyl, or halogen.
- this invention provides or contemplates a compound of formula IA, where R' is phenyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where R 1 is OC 1 -C 3 alkyl.
- this invention provides or contemplates a compound of formula IA, where R' is 2-dimethylaminoethyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H.
- this invention provides or contemplates a compound of formula IA, where R 1 halogen.
- this invention provides or contemplates a compound of formula IA, where R 1 is methyl or ethyl.
- Ri is located as shown below
- Ri is located as shown below
- Ri is located as shown below
- Ri is located as shown below
- this invention provides or contemplates a compound of the structure shown below.
- this invention provides or contemplates a compound of the structure shown below.
- this invention provides or contemplates a compound of the structure shown below, where R 5 is Cs-Ce alkyl or (CH 2 )wC5-C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 5 is Cs-Ce alkyl or (CH 2 )wC 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, or methoxy.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, or methoxy, and R 5 is C5-C6 alkyl or (CH 2 )WCs-Ce cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R5 is Cs-Ce alkyl or (CH 2 )w C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, or methoxy.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, or methoxy.
- this invention provides or contemplates a compound of the structure shown below, where Rs is C 5 -C 6 alkyl or (CH 2 ) * C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R4 are, independently, H, methyl, Cl, CF 3 , OCF 3 , or methoxy.
- this invention provides or contemplates a compound of the structure shown below, where R 5 is Cs-C 6 alkyl or (CH 2 )w C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, Cl, CF3, OCF 3 , or methoxy.
- this invention provides or contemplates a compound of the structure shown below, where R 5 is C 5 -C 6 alkyl or (CH 2 )w Cs-C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where Rs is Cs-C 6 alkyl or (CH 2 )w C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 5 is'(CH 2 ) w Ar or C 3 -C 6 alkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, Cl, CF 3 , OCF 3 , or methoxy.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, Cl, CF 3 , OCF 3 , or methoxy and R 5 is (CH 2 ) w Ar or C 3 -C 6 alkyl.
- this invention provides or contemplates a compound of the structure shown below, where Rs is (CH 2 ) w Ar or C 3 -C 6 alkyl.
- this invention provides or contemplates a compound of the structure shown below, where R 3 and R 4 are, independently, H, methyl, Cl, CF 3 , OCF 3 , or methoxy and where R 5 is (CH 2 ) w Ar or C 3 -Ce alkyl.
- this invention provides or contemplates a compound of formula IA, where R 2 is H.
- this invention provides or contemplates a compound of formula IA, where R 2 is halogen.
- this invention provides or contemplates a compound of formula IA, where R 2 is Cl or F.
- this invention provides or contemplates a compound of formula IA, where R 2 is trifluoromethyl.
- this invention provides or contemplates a compound of formula IA, where R 3 and R 4 are, independently, H, Cl, methyl, ethyl, trifluoromethyl, or methoxy.
- this invention provides or contemplates a compound of formula IA, where q is zero and R3 and R4 are Cl, ethyl, methoxy, or methyl.
- this invention provides or contemplates a compound of formula IA, where q is zero and R 3 and R4 are both methyl.
- this invention provides or contemplates a compound of formula IA, where R' is methyl, halogen, or H; and R 3 and R 4 are, independently, H, Cl, ethyl, methoxy, or methyl.
- this invention provides or contemplates a compound of formula IA, where R' is methoxy; and R 3 and R4 are, independently, H, Cl, ethyl, methoxy, or methyl.
- this invention provides or contemplates a compound of formula IA, where R' is H; and R 3 and R4 are, independently, H, Cl, ethyl, or methyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H, q is zero, and R 5 is Ci-Ce alkyl, or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H; q is 1 ; Y is O; and R 5 is Q-C 6 alkyl, or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H; q is 1 ; Y is S; and R5 is Ci-C 6 alkyl, or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R' and R 2 are H and R 5 is Ci-C 6 alkyl, or (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R' and R 2 are H and R 5 is Ar, (CHR 6 )WAr, CH 2 (CHR 6 ) W Ar, or (CHR 6 ) W CH 2 Ar.
- this invention provides or contemplates a compound of formula IA, where R 1 and R 2 are H and R 5 is (CHR 6 ) W Cs-C 6 cycloalkenyl, CH 2 (CHR 6 ) W C 5 -C 6 cycloalkenyl, C 2 -C 6 alkenyl, or C 2 -C 6 alkynyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is halogen; and R 3 and R 4 are H, Cl, ethyl, or methyl. In another more specific subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where R 1 is Cl or F; and R 3 and R 4 are H, Cl, ethyl, or methyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is Cl or F; R 3 and R 4 are H, Cl, ethyl, or methyl; and R 5 is CpC 6 alkyl, or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is phenyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA, where R 1 is 1 -phenyl, optionally substituted. In another more specific subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where R 1 is 4-phenyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA, where R 1 is phenyl, optionally substituted, and R 5 is Ci-Ce alkyl, or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where Ri is NH-Ci-C 6 alkyl, N(Ci-C 6 alkyl )-Cr C 6 alkyl, C(O)NH-C, -C 6 alkyl, NH-C(O)C 1 -C 6 alkyl; 0-C 1 -C 6 alkyl, CeO)-C 1 -C 6 alkyl, C(O)-OCi-C 6 alkyl, or OC(O)Ci-C 6 alkyl; R 1 is phenyl, optionally substituted, and R 5 is C 1 -C 6 alkyl, or (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R' is H, methyl, or ethyl; and R 1 is NH-Ci-C 6 alkyl, N(Ci-C 6 alkyl )-C,-C 6 alkyl, C(O)NH-Ci -C 6 alkyl, or NH-C(O)C 1 -C 6 alkyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H, methyl, or ethyl; and R 1 is C(O)OCi -C 6 alkyl, OC(O)C 1 -C 6 alkyl, or OC 1 -C 6 alkyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H, methyl, methoxy, or halogen, and R' is methyl or ethyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H, methyl, methoxy, or halogen, and R' is phenyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is H, methyl, methoxy, or halogen, and R 1 is F.
- this invention provides or contemplates a compound of formula IA, where Ri is methoxy, methoxymethyl, ethoxymethyl, or methoxyethyl.
- this invention provides or contemplates a compound of formula IA, where Ri is methoxy, methoxymethyl, ethoxymethyl, or methoxyethyl; R 2 is H, methyl, or halogen; and R 3 is methyl or Cl.
- this invention provides or contemplates a compound of formula IA, where R' is 4-phenyl, optionally substituted, and R 2 is H, methyl, methoxy, or halogen.
- this invention provides or contemplates a compound of formula IA, where R 1 is CF 3 or C 1 -C3 alkyl, and R2 is H, methyl, methoxy, or halogen.
- this invention provides or contemplates a compound of formula IA, where R' is methoxy, and R2 is H, methyl, methoxy, or halogen.
- this invention provides or contemplates a compound of formula IA, where R 1 is 2-dimethylamino ethyl, and R 2 is H, methyl, methoxy, or halogen.
- this invention provides or contemplates a compound of formula IA, where q is zero, R 2 is H, methyl, methoxy, or halogen, R' is 1 -phenyl, optionally substituted; and R 3 and R4 are H, Cl, ethyl, or methyl.
- this invention provides or contemplates a compound of formula IA, where q is zero, R 2 is H, methyl, methoxy, or halogen, R 1 is 4-phenyl, optionally substituted; and R 3 and R 4 are H, Cl, ethyl, or methyl.
- this invention provides or contemplates a compound of formula IA, where q is zero, R2 is H, methyl, methoxy, or halogen; R' is CF 3 or C1-C3 alkyl; and R 3 and R4 are H, Cl, ethyl, or methyl.
- this invention provides or contemplates a compound of formula IA, where q is zero, R 2 is H, methyl, methoxy, or halogen; R' is methoxy; and R 3 and R 4 are H, Cl, ethyl, or methyl.
- this invention provides or contemplates a compound of formula IA, where q is zero; R' is (2-dimethylamino) ethyl; R 2 is H, methyl, methoxy, or halogen; and R 3 and R 4 are H, Cl, ethyl, or methyl.
- the invention provides or contemplates a compound of formula IA-I below.
- this invention provides or contemplates a compound of formula IA-2 below.
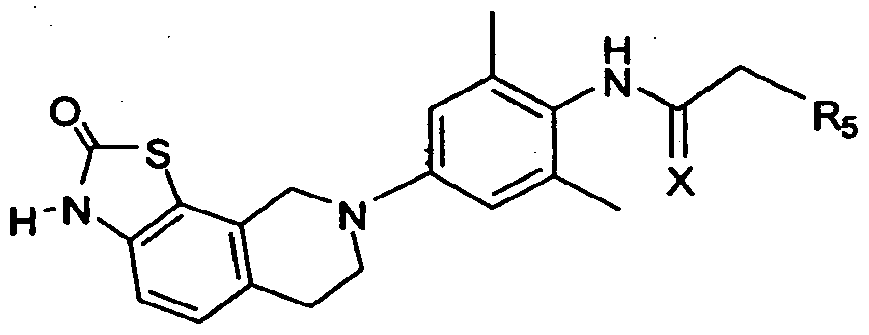
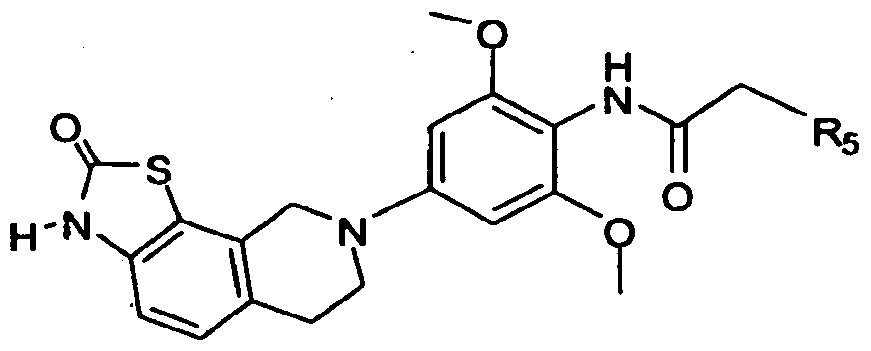
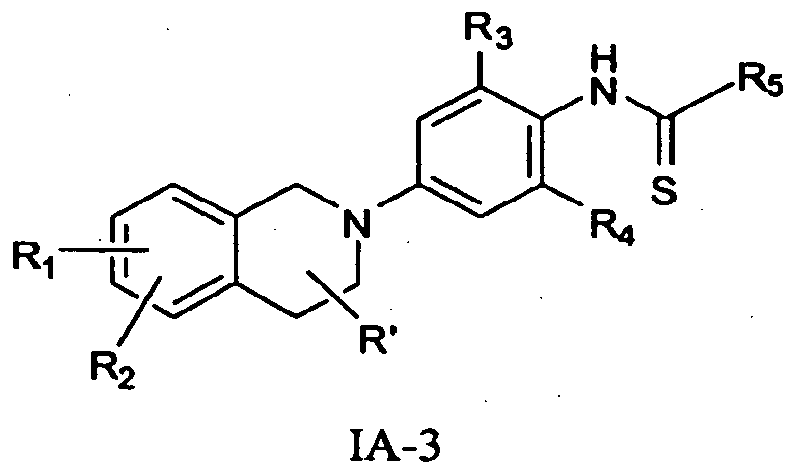
- this invention provides or contemplates a compound of formula IA-3 below.
- this invention provides or contemplates a compound of formula IA-4 below.
- this invention provides or contemplates a compound of formula IA-5 below.
- this invention provides or contemplates a compound of formula IA-I or formula IA-3, where R 2 is H, alkyl, or halogen; and R 5 is
- this invention provides or contemplates a compound of formula IA-I or formula IA-3, where R
- this invention provides or contemplates a compound of formula IA-I or formula IA-3, where Ri is methoxy, methoxymethyl, or methoxyethyl; R 2 is H, alkyl, or halogen; and R 5 is C]-C 6 alkyl,
- this invention provides or contemplates a compound of formula IA-2, where R5 is Ci-C 6 alkyl, (CHR 6 )WC 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is Ar, (CHR 6 ) W Ar, CH 2 (CHR 6 ) w Ar, or (CHR 6 ) W CH 2 Ar.
- this invention provides or contemplates a compound of formula IA-3, where R 2 and R 1 are H; R 3 is methyl; and R 5 is C 1 -C 6 alkyl, (CHR 6 )WC 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 - Ce cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is H, F, Cl, Br, methoxy, methoxymethyl, ethoxymethyl, methoxyethyl, or trifluoromethyl; R 3 is methyl; and R 5 is C 4 -C 6 alkyl, (CHR 6 ) W CH 2 C 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is C 4 -C 6 alkyl, (CHR 6 )WCs-C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, methoxymethyl, ethoxymethyl, methoxyethyl, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri is H, F, Cl, Br, methoxy, methoxymethyl, ethoxymethyl, methoxyethyl, or trifluoromethyl; R 2 is H, methyl, or F; R 1 is H or methyl; R 3 is methyl; and R 5 is C 4 -C 6 alkyl, (CHR 6 ) W C 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 1 is (CH 2 ) m C 3 -C ⁇ cycloalkyl, C 3 -C 6 cycloalkenyl, or (CH 2 ) m C 3 -C 6 cycloalkenyl; R' is halogen; and R 3 is methyl or Cl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is (CH 2 ) m C 3 -C 6 cycloalkyl, C3-C6 cycloalkenyl, or (CH 2 ) m C 3 -C6 cycloalkenyl; and R 1 is F or Cl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is methoxy, methoxymethyl, ethoxymethyl; or methoxyethyl; R 2 is H or F; R 3 is methyl; R4 is methyl or Cl; and R5 is (CHR 6 )WC 5 -C 6 cycloalkenyl or (CHR 6 )WAr.
- this invention provides or contemplates a compound of formula IA-I, where Rj is phenyl, optionally substituted. In another more specific subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where Ri is methyl, halomethyl, ethyl, or haloethyl.
- this invention provides or contemplates a compound of formula IA-I, where R 1 is 2-(dimethylamino) ethyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where R 1 is 1 -methyl or 1 -ethyl.
- this invention provides or contemplates a compound of formula IA-I, where R 1 is 1-fluoro, R 5 is C 4 -C 6 alkyl, (CHR 6 )WCs-C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and R, is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is 4-fluoro, R 5 is (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is (CH 2 ) m ⁇ dazolyl, (CH 2 ) m pyrazyl, (CH 2 )m furyl, (CH 2 ) m thienyl, (CH 2 ) m oxazolyl, (CH 2 ) m isoxazolyl, (CH 2 ) m thiazolyl, (CH 2 ) m isothiazolyl, (CH 2 ) m phenyl, (CH 2 ) m ⁇ yrrolyl, (C ⁇ pyridyl, or (CH 2 ) m ⁇ yrimidyl; and R 2 and R' are H.
- this invention provides or contemplates a compound of formula IA-I, where Ri is (CH 2 ) m imidazolyl, (CH 2 ) ⁇ ,pyrazyl, (CH 2 ) m furyl, (CH 2 ) m thienyl, (CH 2 ) m oxazolyl, (CH 2 ) m isoxazolyl, (CH 2 ) m thiazolyl, (CH 2 ) m isothiazolyl, (CH 2 ) m phenyl, (CH 2 ) m pyrrolyl, (CH 2 ) m pyridyl, or (CH 2 ) m pyrimidyl; and R' is 4-phenyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA-I, where R 1 is CF 3 or C 1 -C 3 alkyl; R 5 is G t -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and R t is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is 4-methyl or 4-ethyl; and R 5 is C 4 - C 6 alkyl, (CHR 6 ) W C 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Rj is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I , where R' is methoxy or ethoxy; and R s is C 4 - C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is 1 -phenyl, optionally substituted; R 5 is C 4 -C 6 alkyl, (CHR 6 ) W C 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is 4-phenyl, optionally substituted; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and R 1 is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is CF 3 or C 1 -C 3 alkyl; R 5 is C 4 -C 6 alkyl, (CHR 6 ) ⁇ C 5 -C 6 cycloalkyl, or CH 2 (CHR ⁇ ;) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is 4-methyl or 4-ethyl; R 5 is C4-C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cydoalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-I, where R' is methoxy or ethoxy, R 5 is C-J-C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-4, where Ri is H, F, Cl, Br, methoxy, or trifluoromethyl; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Rj is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-4, where R 2 is H, F, or methyl; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-2, where R' is H.
- this invention provides or contemplates a compound of formula IA-2, where R 1 is halogen.
- this invention provides or contemplates a compound of formula IA-2, where R* is F.
- this invention provides or contemplates a compound of formula IA-2, where R 1 is methyl or ethyl.
- this invention provides or contemplates a compound of formula IA-2, where R 1 is methyl or ethyl; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and R
- this invention provides or contemplates a compound of formula IA-2, where R' is halogen; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR6) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-2, where R' is H; R 5 is C 4 -C 6 alkyl, (CHR 6 ) W C 5 - C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-2, where R' is 1 -phenyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA-2, where R 1 is 4-phenyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA-2, where R' is CF 3 or Ci -Cj alkyl.
- this invention provides or contemplates a compound of formula IA-3, where R 1 is H; R 5 is C 4 -QaIkVl, (CHR 6 ) W C 5 - C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-3, where R' is F; R 5 is G t -C 6 alkyl, (CHR 6 ) W C 5 - C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-3, where R' is 1 -phenyl, optionally substituted; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-3, where R' is 4-phenyl, optionally substituted; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and R t is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA-3, where R' is CF 3 or C 1 -C 3 alkyl; R 5 is C 4 -C 6 alkyl, (CHR 6 )WC 5 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 5 -C 6 cycloalkyl; and Ri is H, F, Cl, Br, methoxy, or trifluoromethyl.
- this invention provides or contemplates a compound of formula IA, where R 5 is Ar, (CHR 6 )WAr 5 CH 2 (CHR 6 ) W Ar, or In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-2, where R 5 is Ar, (CHR 6 ) W Ar, CH 2 (CHR 6 ) W Ar, or
- this invention provides or contemplates a compound of formula IA-3, where R 5 is Ar, (CHR 6 ) W Ar, CH 2 (CHR 6 ) W Ar, or
- this invention provides or contemplates compounds of formula IA-I, IA-2, IA-3, IA-4, or IA-5, where Ri and R 2 , are, independently, methyl, ethyl, F, Cl, CF 3 , methoxy or methoxymethyl, R 1 is methyl, and R 5 is C 4 -C 6 alkyl, (CHR 6 ) W C 5 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where R 5 is haloalkyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where R 5 is haloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is haloalkyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-3, where R5 is haloalkyl.
- this invention provides or contemplates a compound of formula IA, where Rs is methoxy alkyl.
- this invention provides or contemplates a compound of formula IA, where R5 is cyano alkyl.
- the invention provides or contemplates a compound of formula IA-4, where Rs is halo alkyl.
- the invention provides or contemplates a compound of formula IA, where R 5 is CH 2 -cycloalkyl or CH 2 CH 2 - cycloalkyl.
- the invention provides or contemplates a compound of formula IA-4, where R 5 is CH2-cycloalkyl or CH 2 CH 2 - cycloalkyl.
- the invention provides or contemplates a compound of formula IA-5, where R 5 is CH 2 -cycloalkyl or CH 2 CH 2 - cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I , where R 3 and R 4 are chloro, methoxy, or methyl and R 5 is CH 2 -cycloalkyl .
- this invention provides or contemplates a compound of formula IA-I, where R 3 and R4 are chloro, methoxy, or methyl and Rs is haloalkyl, hydroxyalkyl, or methoxyalkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 3 and R 4 are chloro, methoxy, or methyl and R 5 is methoxy alkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 3 and R 4 are chloro, methoxy, or methyl and R 5 is chloroalkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 3 and R4 are chloro, methoxy, or methyl and R 5 is methoxyalkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 3 and R4 are both methyl and R 5 is 2-(2-halo cyclopentyl) ethyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where R 3 and R4 are both methyl and R 5 is 2-(2-furyl) ethyl.
- this invention provides or contemplates a compound of formula IA-I, where R 3 and R 4 are both methyl and R 5 is 2-(2- tetrahydrofuryl) ethyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where R 3 and R 4 are both methyl and R 5 is 2-phenyl ethyl.
- this invention provides or contemplates a compound of formula IA-I, where R 3 and R4 are both methyl and R 5 is 3-phenyl propyl.
- this invention provides or contemplates a compound of formula IA-I , where R 3 and R4 are both methyl and R 5 is 2-phenyl propyl.
- this invention provides or contemplates a compound of formula IA-I, where R 5 is Ci-C 6 alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl; R' is halogen or C1-C 3 alkyl; and Ri is halogen.
- this invention provides or contemplates a compound of formula IA-I, where R5 is Ci-C 6 alkyl, (CHR6) W C 3 -C 6 cycloalkyl, (CHR 6 ) W CH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl; R' is halogen or Ci-C 3 alkyl; R 2 is H or halogen; and Ri is halogen.
- this invention provides or contemplates a compound of formula IA- 1 , where R 5 is Ci-C 6 alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl; R 1 is phenyl, optionally substituted; R 2 is H or halogen; and Ri is halogen.
- this invention provides or contemplates a compound of formula IA-I, where R 5 is C)-C 6 alkyl, (CHR 6 )WC 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl; R 1 is halogen or Ci-C 3 alkyl; R 2 is H or halogen; and Ri is halogen.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is Ci-C 6 alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 ) W CH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 3 -C 6 cycloalkyl; R 1 is halogen or Cj-C 3 alkyl; and Ri is halogen.
- this invention provides or contemplates a compound of formula IA-3, where R 5 is Ci-C 6 alkyl, (CHR 6 )WC 3 -C 6 cycloalkyl, (CHR 6 ) W CH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 3 -C 6 cycloalkyl; R 1 is halogen or Ci-C 3 alkyl; and Rj is halogen.
- this invention provides or contemplates a compound of formula IA, where R 5 is Ar, (CHR 6 ) W Ar, CH 2 (CHR 6 ) W Ar, or (CHR 6 ) W CH 2 Ar 1 .
- this invention provides or contemplates a compound of formula IA-3, where Ri is haloalkyl; R 2 is H or F; R 3 and R 4 are Cl, methoxy, or methyl; and R 5 is Cj-C 6 alkyl, (CHR 6 )WC 3 -C 6 cycloalkyl, (CHR 6 ) W CH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is Cj-C 3 alkyl, halogen, or haloalkyl; R 2 is H or F; R 3 and R 4 are H, methyl, or Cl; and R 5 is CH 2 CR 6 -C 3 -C 6 cycloalkyl, or C 2 -C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri is Ci-C 3 alkyl, halogen, or haloalkyl; R 2 is H or F; R 3 and R 4 are H, methyl, or Cl; and R 5 is CH 2 CR 6 -C 3 -C 6 cycloalkyl, (CHR 6 ) W C 5 -C 6 cycloalkenyl, CH 2 (CHR 6 ) W C 5 -C 6 cycloalkenyl, C 2 -C 6 alkenyl, or C 2 -C 6 alkynyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri is halogen or haloalkyl; R 2 is H or F; R 3 and R 4 are Cl, methoxy, or methyl; and R 5 is Ci-C 6 alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri is methyl, fluoro, or fluoroalkyl; R 2 is H or F; and R 5 is Ci-C 6 alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 ) W CH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri is Cl, F, or CF 3 ; R 2 is H or F; R' is H or CH 3 ; and R 5 is Ar, (CHR 6 )WAr, CH 2 (CHR 6 )WAr, or (CHR 6 )WCH 2 Ar.
- this invention provides or contemplates a compound of formula I A-4, where R 3 and R4 are H, methyl, or Cl; and R 5 is Ci-C 6 alkyl, (CHR 6 )WC 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 3 - C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 3 and R 4 are H, methyl, or Cl; and where Ri and R 2 , on adjacent carbons, form a six-membered ring.
- this invention provides or contemplates a compound of formula IA- 1 , where R' is F, methyl, or H; R 3 and R 4 are H, methyl, or Cl; and R 5 is Ci-C 6 alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 ) W CH 2 C 3 -C 6 cycloalkyl, or CH 2 (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 1 is halogen and R 5 is Ar, (CHR 6 ) W Ar, .
- this invention provides or contemplates a compound of formula IA-I , where Ri and R 2 are on adjacent carbon atoms and are both other than H.
- this invention provides or contemplates a compound of formula IA-I, where Ri and R 2 , on adjacent carbon atoms are, independently trifluoromethyl or halogen; and where Rs is Ci-Ce alkyl, (CHR 6 ) W C 3 -C 6 cycloalkyl, (CHR 6 )WCH 2 C 3 -C 6 cycloalkyi, or CH 2 (CHR 6 )WC 3 -C 6 cycloalkyl-
- this invention provides or contemplates a compound of formula IA-I, where Rs is (CHR 6 )WCs-C 6 cycloalkenyl, CH 2 (CHR 6 )WCs-C 6 cycloalkenyl, C 2 -C 6 alkenyl, or C 2 -C 6 alkynyl.
- this invention provides or contemplates a compound of fo ⁇ nula IA-I, where R 5 is Ar, (CHR ⁇ JwAr, CH 2 (CHR 6 ) W Ar, or
- this invention provides or contemplates a compound of formula IA-I, where Ri is halogen or trifluoromethyl and R 2 is H, or Ri and R 2 , on adjacent carbon atoms are, independently trifluoromethyl or halogen; and where R 5 is Ar, (CHR 6 ) W Ar, CH 2 (CHR 6 ) W Ar, or (CHR 6 ) W CH 2 Ar.
- this invention provides or contemplates a compound of formula IA -2 where R 5 is Ci-C 6 alkyl or (CHR 6 ) W Cs-C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-3, where R 5 is C]-C 6 alkyl or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where Ri is halogen or trifluoromethyl and R 2 is H or Ri and R 2 , on adjacent carbon atoms, are, independently, halogen or trifluoromethyl; and R 5 is Ci-C 6 alkyl or (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri is halogen or trifluoromethyl and R 2 is H or Ri and R2, on adjacent carbon atoms, are, independently, halogen or trifluoromethyl; and R 5 is Ci-C 6 alkyl or (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where Ri and R 2 are, independently, methyl, methoxy, trifluoromethyl, F, Cl, or H; and R 5 is Ci-C 6 alkyl or (CHR 6 ) W C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-3, where Rj and R 2 are, independently, methyl, methoxy, trifluoromethyl, F, Cl, or H; R' is H; and R 5 is Q-C 6 alkyl or (CHR 6 )WC 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I or IA-2 or IA-3, where Rj is halogen, Ci-C 6 alkyl, mono-halo Ci-C 6 alkyl, CN, di-halo C r C 6 alkyl, CF 3 , CN, or O-Ci-C 6 alkyl; R' is methyl or ethyl; and R 5 is C 5 -C 6 alkyl or CH 2 -C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I or IA-2 or IA-3, where R
- this invention provides or contemplates a compound of formula IA, where Ri is F, Cl, or CF 3 ; R 2 is H; and R 1 is halogen, methyl, ethyl, or methoxy; R 3 and R 4 are H, methyl, or Cl; and R5 is C 5 -C6 alkyl or CH2-C 3 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where Ri is halogen or CF 3 ; R 2 is H, F, or methyl, R' is phenyl; R 3 and R 4 are H, methyl, or Cl; and R 5 is C 5 -C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where Ri is halogen or CF 3 ; R2 is H, F, or methyl, R' is halophenyl; R 3 and R4 are H, methyl, or Cl; and R 5 is C 5 -C 6 alkyl or CFb-C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA where Rj is NH 2 , NH-Ci -C 6 alkyl; or N(CpC 6 alkyl)-Ci -C 6 alkyl; and R 2 is H or halogen.
- this invention provides or contemplates a compound of formula IA-I where Ri is NHC(O)Ci -C 6 alkyl, C(O)N(CHj) 2 , C(O)N(Et) 2 , C(O)NH 2 , or C(O)NH-Ci-C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-I where Ri is SO 2 NH 2 or NHSO 2 -Ci-C 6 alkyl. In another more specific subgeneric embodiment, this invention provides or contemplates a compound of formula IA-2 where R 1 is SO 2 NH 2 or NHSO 2 -Ci-Ce alkyl.
- this invention provides or contemplates a compound of formula IA, where R 1 is (CH 2 ) m C 3 -C 6 cycloalkenyl, C2-C 6 alkenyl, or C2-C6 alkynyl.
- this invention provides or contemplates a compound of formula IA, where Ri is CH 2 OCH 3 , CH 2 OCH 2 CH 3 , OC 1 -C 6 alkyl, or SC r C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 1 is CH 2 OCH 3 , CH 2 OCH 2 CH 3 , OCi-C 6 alkyl, or SC 1 - C 6 alkyl.
- -C 6 alkyl, OCC O)C 1 - C 6 alkyl, OCi-C 6 alkyl, or SC 1 -C 6 alkyl; R 2 is H, F, or methyl, R' is halogen or methyl; and R 5 is C 5 -C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is NH 2 , NH-Ci-C 6 alkyl; or N(Ci-C 6 alkyl)-Ci -C 6 alkyl; R 2 is H, F, or methyl, R 1 is halogen or methyl; and R5 is Cs-C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where Ri is C 2 -Ce alkynyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA, where Rj and R 2 form a fused, nitrogen-containing ring. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where Ri and R 2 form a fused, oxygen-containing ring.
- this invention provides or contemplates a compound of formula IA, where Ri and R 2 form a fused thiazolo or isothiazolo group.
- this invention provides or contemplates a compound of formula IA, where Ri and R 2 form a fused cyclopentane, optionally substituted.
- this invention provides or contemplates a compound of formula IA, where R] and R 2 form a fused cyclohexane, optionally substituted.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused, nitrogen-containing ring.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused, oxygen-containing ring.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused thiazolo or isothiazolo group.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused cyclopentane, optionally substituted.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused cyclohexane, optionally substituted.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused, nitrogen-containing ring; and R 5 is Cs-Ce alkyl or CH 2 -Cs-Ce cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Rj and R 2 form a fused, oxygen-containing ring; and R 5 is Cs-Q alkyl or CH2-C5-C6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused thiazolo or isothiazolo group; and R 5 is C 5 -C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused cyclopentane, optionally substituted; and R 5 is C 5 -C 6 alkyl or CH 2 -Cs-Ce cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I or IA-2, where Ri and R 2 form a fused cyclohexane, optionally substituted; and R 5 is C 5 -C 6 alkyl or CH 2 -Cs-C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is halogen; R 2 is H, F, or methyl, R' is halogen or methyl; and R 5 is C 5 -Ce alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is halogen; R 2 is H, F, or methyl, R 1 is 2-(dimethylamino) ethyl; and R 5 is C 5 -C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is halogen; R 2 is H, halogen, or methyl, R' is H; and R 5 is C 5 -C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where Ri is halogen; R 2 is H or methyl, R' is halogen or methyl; and R 5 is C 5 -C 6 alkyl or CH 2 -C 5 -C 6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-I .
- this invention provides or contemplates a compound of formula IA-I, where R 1 is trifluoromethyl; R2 is H or methyl, R' is halogen or methyl; and R 5 is C 5 -C 6 alkyl or CH 2 -C S -C O cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where Ri where Ri is trifluoromethyl; R 2 is H or methyl, R' is halogen or methyl; and R 5 is Cs-C ⁇ alkyl or CH2-C5-C6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-3, where Ri where Ri is trifluoromethyl; R 2 is H or methyl, R' is halogen or methyl; and R 5 is Cs-C ⁇ alkyl or CH2-C5-C6 cycloalkyl.
- this invention provides or contemplates a compound of formula IA-4 or IA-5, where Rj where Ri is trifluoromethyl; R 2 is H or methyl, R* is halogen or methyl; and R 5 is C 5 -C 6 alkyl or CH 2 -Cs-Cg cycloalkyl.
- this invention provides or contemplates a compound of formula IA-2, where Ri is trifluoromethyl; R 2 is F; R 1 is halogen or methyl; and Rs is Cs-Q alkyl or CH 2 -C S -C O cycloalkyl.
- this invention provides or contemplates a compound of formula IA, where Ri or R 5 is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is phenyl, pyridyl, pyrrolyl, imidazolyl, oxazolyl, or thiazolyl.
- this invention provides or contemplates a compound of formula IA, where Ri is F. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where Ri is Cl.
- this invention provides or contemplates a compound of formula IA, where Ri is Br.
- this invention provides or contemplates a compound of formula IA- 1 , where Ri is F.
- this invention provides or contemplates a compound of formula IA-I, where Ri is Cl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is Br. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where Ri is F and R 2 is H, OCH 3 , or F.
- this invention provides or contemplates a compound of formula IA-I, where Ri is F; R 3 and R 4 are both methyl; and R 1 is H. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-I, where Ri is CF 3 ; R 3 and R 4 are both methyl; and R' is H.
- this invention provides or contemplates a compound of formula IA, where Ri and R 2 are both F.
- this invention provides or contemplates a compound of formula IA, where Ri is mono-, di-, or tri-halomethyl.
- this invention provides or contemplates a compound of formula IA, where Ri is CH 2 F, CHF 2 , or CF 3 .
- this invention provides or contemplates a compound of formula IA, where Ri is CH 2 CI. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where Ri is CH 2 Br.
- this invention provides or contemplates a compound of formula IA-I, where Ri and R 2 are both F; R 3 and R4 are both methyl; and R 1 is H. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA-2, where Ri is F.
- this invention provides or contemplates a compound of formula IA-2, where Ri and R 2 are both F.
- this invention provides or contemplates a compound of formula IA-3, where Ri is F.
- this invention provides or contemplates a compound of formula IA-3, where Ri and R 2 are both F.
- this invention provides or contemplates a compound of formula IA, where Ri or R 5 is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is isoxazolyl or isothiazolyl. In another subgeneric embodiment, this invention provides or contemplates a compound of formula IA, where R
- this invention provides or contemplates a compound of formula IA, where Ri or Rs is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is pyrimidyl or purinyl.
- this invention provides or contemplates a compound of formula IA, where Ri or R5 is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is indolyl, isoindolyl, or benzimidazolyl.
- this invention provides or contemplates a compound of formula IA, where Ri or Rs is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is halo phenyl.
- this invention provides or contemplates a compound of formula IA, where Ri or R 5 is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is dihalophenyl or dihalopyridyl.
- invention provides or contemplates a compound of formula IA, where Ri or Rs is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is mono- or di-halothienyl, mono- or di-halofiiryl, mono- or di-halobenzothienyl, or mono- or di- halobenzofuryl.
- this invention provides or contemplates a compound of formula IA, where Ri or R 5 is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is o-, m-, or p- xylyl or o-, m-, orp-anisyl.
- this invention provides or contemplates a compound of formula IA, where Ri or Rs is CH 2 Ar or CH 2 CH 2 -Ar, where Ar is m- o ⁇ p- cyanophenyl or m- or p-cyanom ethyl phenyl.
- this invention provides or contemplates a compound of formula IA, in which R 3 and R 4 are halogen, CF 3 , or C 1 -C 3 alkyl and R 5 is C 1 -C 6 alkyl, where the alkyl group is substituted with one or two groups selected, independently, from OH, OMe, OEt, F, CF 3 , Cl, or CN.
- this invention provides or contemplates a compound of formula IA, in which R 3 and R 4 are halogen, CF 3 , OCF 3 , C 1 -C 3 alkyl, or OCi-C 3 alkyl, and R 5 is (CH2) W C 3 -C 6 cycloalkyl, where w is 1 or 2, where the cycloalkyl group is substituted with Me, OH, OMe, OEt, F, CF 3 , Cl, or CN.
- this invention provides or contemplates a compound of formula IA-I, in which R3 and R4 are halogen, CF 3 , or Ci- C 3 alkyl, and R 5 is (CH 2 ) W -Cs-C 6 cycloalkyl, optionally substituted, or (CH 2 ) W -Cs-C 6 heterocycloalkyl, optionally substituted.
- this invention provides or contemplates a compound of formula IA-I , where Ri is CH 2 phenyl or CH2CH2-phenyl.
- this invention provides or contemplates a compound of formula IA-I, where Ri is Ar, CH 2 Ar or CH 2 CH 2 -Ar, where Ar is 3,5- dichlorophenyl or 3,5-difluorophenyl.
- this invention provides or contemplates a compound of formula IA-I, where Rs is Ar, (CHRe) w Ar, CH 2 (CHR ⁇ j) w Ar, or (CHR 6 )WCH 2 Ar, where Ar is phenyl or pyridyl; R 3 and R4 are H or Ci-Ce alkyl, unsubstituted or substituted with one or two groups selected from OH, OMe; and Re is CN, CH 2 CN, or halogen.
- this invention provides or contemplates a compound of formula IA-I , where R 5 is Ar, (CHR 6 ) w Ar, CH 2 (CHR 6 ) w Ar, or
- this invention provides or contemplates a compound of formula IA-I, where R 5 is Ar, (CHR ⁇ ) w Ar, CH 2 (CHRe) W Ar, or (CHR 6 )WCH 2 Ar, where Ar is phenyl or pyridyl, and Ri is OCi-C 6 alkyl or C(O)Cj-C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 5 is Ar, (CHR ⁇ ) w Ar, CH 2 (CHR 6 ) w Ar, or (CHR6) w CH 2 Ar, where Ar is phenyl or pyridyl, and Ri is C(O)OCi-C 6 alkyl or OC(O)Ci-C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-I, where R5 is Ar, (CHR6) w A ⁇ , CH 2 (CHR 6 )WAr, or (CHR 6 )WCH 2 Ar, where Ar is phenyl or pyridyl, Ri is C2-C6 alkenyl or C2-C6 alkynyl, q is 1, and X and Y are both O.
- this invention provides or contemplates a compound of formula IA-I, where R5 is Ar, (CHR 6 )WAr, CH 2 (CHRe)WAr, or (CHRe) Vv CH 2 Ar, Ar is phenyl or pyridyl, and Ri is SCi-Ce alkyl.
- this invention provides or contemplates a compound of formula IA-I, where R 5 is Ar, (CHR 6 )V 1 Ar, CH 2 (CHR 6 )WAr, or (CHR 6 ) W CH 2 Ar, where Ar is phenyl or pyridyl, R 3 and R 4 are H, Cl, methoxy, or C 1 -C 3 alkyl, and Ri is CpC 6 alkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is Ar, (CHR 6 ) w Ar, CH 2 (CHR 6 ) W Ar, or (CHR 6 ) w CH 2 Ar, where Ar is phenyl or pyridyl; R3 and R 4 are H, Cl, methoxy, or Ci-C 2 alkyl, unsubstituted or substituted with one or two groups selected from OH, OMe; and Ri is CN, CH 2 CN, or halogen.
- this invention provides or contemplates a compound of formula IA-2, where R5 is Ar, (CHR6) w Ar, CH 2 (CHR 6 )WAr, or (CHR 6 )WCH 2 Ar, where Ar is phenyl or pyridyl; and Ri is F, CH 2 F, CHF 2 , CF 3 , or CF 2 CF 3 .
- this invention provides or contemplates a compound of formula IA-I, where R s is Ar, (CHR 6 )WAr, CH 2 (CHR 6 )WAr, or (CHR 6 )WCH 2 Ar, where Ar is phenyl or pyridyl, and Ri is OCi-C 6 alkyl or C(O)Ci-C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is Ar, (CHRe) w Ar, CH 2 (CHR 6 ) W Ar, or
- this invention provides or contemplates a compound of formula IA-3, where R 5 is Ar, (CHR 6 )WAr, CH 2 (CHR 6 ) W Ar, or (CHR 6 ) w CH 2 Ar, where Ar is phenyl or pyridyl, and Ri is OC 1 -C 6 alkyl or C(K))Ci-C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is Ar, (CHR ⁇ Ar, CH 2 (CHR 6 ) w Ar, or (CHR 6 ) W CH 2 Ar, Ar is phenyl or pyridyl, and Rj is SCi -C 6 alkyl.
- this invention provides or contemplates a compound of formula IA-2, where R 5 is Ar, (CHR 6 )WAr, CH 2 (CHR 6 ) W Ar, or (CHR 6 ) W CH 2 Ar, where Ar is phenyl or pyridyl, R 3 and R 4 are H or C 1 -C 3 alkyl, and Ri is Ci-C 6 alkyl.
- this invention provides or contemplates a method of treating or preventing a disease, disorder, or condition that is affected by modulation of potassium ion channels in a patient comprising administration of a compound of formula LA in an amount of up to 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a disease, disorder, or condition that is affected by modulation of potassium ion channels in a patient comprising administration of a compound of formula IA in an amount of from about 10 mg to about 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a disease, disorder, or condition that is affected by modulation of potassium ion channels in a patient comprising administration of a compound of formula IA-I in an amount of up to about 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA in an amount of up to about 2000 mg per day. In another embodiment, this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA in an amount of from about 10 mg per day to about 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA in an amount of from about 300 mg per day to about 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA in an amount of from about 300 mg per day to about 1200 mg per day. In another more specific embodiment, this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA-I in an amount of up to 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA- 1 in an amount of from about 10 mg per day to about 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA-I in an amount of from about 300 mg per day to about 2000 mg per day.
- this invention provides or contemplates a method of treating or preventing a seizure disorder in a patient comprising administration of a compound of formula IA-I in an amount of from about 300 mg per day to about 1200 mg per day.
- compounds of formula IA are designed for oral or intravenous dosing of up to 2000 mg per day. Yet the high activities of many of these compounds indicate that dosing of less than 1200 mg per day - the current anticipated dosing level of retigabine in adults — is possible.
- this invention comprises tablets, capsules, solutions, and suspensions of compounds of formula IA which are formulated for oral administration.
- solutions and suspensions suitable for oral pediatric administration comprising, in addition to compounds of formula IA, a syrup such as sorbitol or propylene glycol, among many other examples, are also contemplated.
- solutions and suspensions comprising, in addition to compounds of formula IA, a syrup such as sorbitol or propylene glycol, along with colorants and flavorings suitable for oral pediatric administration, are also contemplated.
- a syrup such as sorbitol or propylene glycol
- colorants and flavorings suitable for oral pediatric administration are also contemplated.
- both chewable and non- chewable tablets comprising compounds of formula IA, along with pharmaceutically acceptable tabletting agents and other pharmaceutically acceptable carriers and excipients, are also contemplated.
- pharmaceutically acceptable carrier comprises such excipients, binders, lubricants, tabletting agents, disintegrants, preservatives, anti-oxidants, flavours and colourants as are typically used in the art of formulation of pharmaceuticals.
- Such agents include — but are not limited to — starch, calcium carbonate, dibasic calcium phosphate, dicalcium phosphate, microcrystalline cellulose, hydroxypropylcellulose, hydroxypropylmethylcellulose lactose, polyethylene glycols, polysorbates, glycols, safflower oil, sesame oil, soybean oil, and Povidone. Additionally, disintegrants such as sodium starch glycolate; lubricants such as magnesium stearate, stearic acid, and SiO 2 ; and solubility enhancers such as cyclodextrins, among a great many other examples for each group, are contemplated. ' Such materials and the methods of using them are well known in the pharmaceutical art.
- the term "pharmaceutically acceptable acid salts” refers to acid addition salts formed from acids which provide non-toxic anions.
- the pharmaceutically acceptable anions include, but are not limited to, acetate, aspartate, benzoate, bicarbonate, carbonate, bisulfate, sulfate, chloride, bromide, benzene sulfonate, methyl sulfonate, phosphate, acid phosphate, lactate, maleate, malate, malonate, fumarate, lactate, tartrate, borate, camsylate, citrate, edisylate, esylate, formate, fumarate, gluceptate, glucuronate, gluconate oxalate, palmitate, pamoate, saccharate, stearate, succinate, tartrate, tosylate and trifluoroacetate salts, among a great
- salts of compounds of formula I maybe prepared by reaction of a compound of formula I with the desired acid; by removal of a protecting group from a suitable precursor of the compound of formula I or by ring-opening a suitable cyclic precursor, for example, a lactone or lactam, using the desired acid or base; and by conversion of one salt of the compound of formula I to another by reaction with an appropriate acid or base or by passage through an appropriate ion-exchange column.
- solvate refers to describe a molecular complex comprising the compound of the invention and a stoichiometric amount of one or more pharmaceutically acceptable solvent molecules, including but not limited to water and ethanol.
- solvate includes a hydrate as one example and an ethanolate as another example.
- modulation of ion channels refers to activating the ion channels, to affecting the kinetics of opening and closing of the ion channels, or to causing any change in the channel open probability of the ion channels.
- Such substituted tetrahydroisoquinolines are either commercially available or are prepared from commercially available materials.
- a great many substituted tetrahydroisoquinolines are known, including many fused isothiazole, piperidino and pyrrolidine derivatives.
- compounds of formula IA where Ri is 5- fluoro- can be prepared starting with 5-fluoro-l,2,3,4-tetrahydroisoquinoline.
- compounds of formula IA where Rj or R 2 is 6-methyl- can be prepared starting with 6-methyl-l ,2,3,4-tetrahydroisoquinoline.
- compounds of formula IA where Ri and R 2 are 6- and 7- chloro, respectively can be prepared starting with 6-, 7- dichloro-1,2,3,4- tetrahydroisoquinoline; and compounds with a substituent in the 9-position can be prepared starting with the appropriate 9-substitued tetrahydroisoquinoline.
- compounds with R' other than H can be prepared starting with the appropriate 1-, 3-, or 4- substituted tetrahydroisoquinolines.
- R' is phenyl, methoxy, ethyl, methyl, F, or 2-(N-, N-dimethylamino)ethyl are accessible via the commercially available 1- and 4-substituted tetrahydroisoquinolines.
- the aromatic amine I is brominated according to standard procedures, including but not limited to the reaction with such reagents as N- bromosuccinimide in an aprotic solvent such as acetonitrile.
- the reaction mixture is typically heated under reflux for a period of from approximately 8 to approximately 48 hours.
- the resulting bromo derivative II is purified by filtration of the crude reaction mixture through Celite. If desired, other standard purification techniques, including flash chromatography, can be used.
- the reaction of a compound II with the appropriate acyl chloride III in an aprotic solvent such as acetonitrile produces the amide of general formula IV.
- This reaction is typically conducted at room temperature for a period of from approximately 4 to approximately 48 hours.
- the resulting amide of general formula IV can be purified by a standard chromatographic technique such as flash chromatography or thin layer chromatography.
- the next step of the reaction sequence is to prepare the desired product of general Formula VI using the well-known palladium coupling reaction, employing a phosphine ligand such as the commercially available dicyclohexyl phosphino-2'-(7/, ⁇ ';- dimethylamino)biphenyl.
- a phosphine ligand such as the commercially available dicyclohexyl phosphino-2'-(7/, ⁇ ';- dimethylamino)biphenyl.
- the amine of general formula V can be coupled to the bromine derivative of general formula IV using a palladium derivative such as, for example, bis(dibenzylidineacetone)palladium, a base such as potassium t ⁇ r/-butoxide and the ligand dicyclohexyl phosphino-2'-(JV,A' r , -dimethyl amino)biphenyl in an aprotic solvent.
- the reaction mixture is typically heated in an oil bath at 90 0 C for a period of from approximately 8 to approximately 48 hours, or it can be heated using a microwave 5 apparatus (Horizon unit, Biotage) at a temperature range of from approximately 90° to approximately 250 0 C.
- the desired compound of general formula VI is purified by standard chromatographic techniques, such as flash chromatography or thin layer chromatography. It can also be recrystallized from toluene.
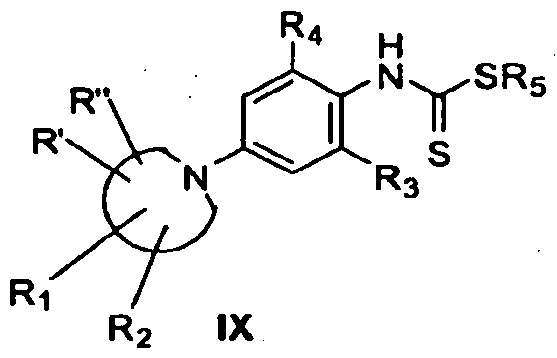
- the compounds of general Formula IX are prepared in a way similar to that employed in section I.
- the aniline derivative II (section I) is combined with the haloalkyl compound VII under standard conditions to produce the desired thioester of general formula VIII.
- the reaction is typically conducted at a temperature of from approximately 20° to approximately 9O 0 C for a period of from
- thioester can be purified by standard chromatographic techniques such as flash chromatography or thin layer chromatography.
- a palladium coupling reaction to produce the compound of general Formula IX is identical to that described in the corresponding step in Section I.
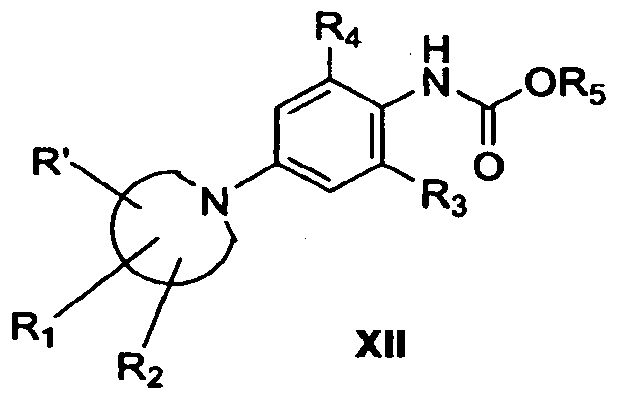
- the aniline is allowed to react with an anhydride derivative of general Formula X in the presence of a base such as triethylamine or diisopropyl ethylamine in an aprotic solvent such as methylene chloride.
- a base such as triethylamine or diisopropyl ethylamine
- an aprotic solvent such as methylene chloride.
- the reaction is conducted at a temperature in the range of from approximately -20° to approximately 4O 0 C for a period of from approximately 30 min to approximately 48 hours, depending on the particular substrates.
- the resulting carbamate derivative of general Formula XI can be purified by the usual chromatographic techniques, such as flash chromatography or thin layer chromatography. As in sections I and II, the final step is a palladium coupling.
- a compound of general Formula XII obtained as in section III, reacts with Lawesson's reagent in an aprotic solvent such as methylene chloride to produce the 5 thiocarbamate.
- an aprotic solvent such as methylene chloride
- the reaction is stirred at room temperature or is heated under reflux for a period of from approximately 2 to approximately 48 hours.
- the resulting compound XIII can be purified by the usual chromatographic techniques, such as flash chromatography or thin layer chromatography.
- the compound of general Formula XIV is obtained under the same conditions 15 described in section IV.
- the reaction is typically heated under reflux or stirred at room temperature for a period of from approximately 2 to approximately 48 hours.
- the resulting derivative of general Formula XIV can be purified by the usual chromatographic techniques, such as flash chromatography or thin layer chromatography.
- bromodimethylaniline was obtained from either Alfa Aesar or Sigma Aldrich.
- Step B N-(4-bromo-2-chloro-6-(trifluoromethyl)phenyl)-3.3- dimethylbutanamide:
- Step A N-(4-Bromo-2.6-dimethyl-phenyl ' )-3,3-dimethyl-butanamide:
- Step A 4-bromo-2-chloro-6-(trifluoromethyl)aniline:
- N-bromosuccinimide (910 mg, 5.1 mmol) was added to a solution of 2-chloro-6- (trifluoromethyl)aniline (1.0 g, 5.1 mmol) and acetic acid (3 mL) in acetonitrile (10 mL) at room temperature. The mixture was stirred at reflux for 18h. The reaction mixture was then filtered through Celite and concentrated to give the title compound, which was used in the next step without further purification.
- Step B N-(4-Bromo-2-chloro-6-trifluoromethyl-phenv ⁇ -3-cvclopentyl- propionamide:
- Step C N-(2-chloro-4-(3,4-dihydroisoquinolin-2(7HVyl)-6- (trifluoromethyl)phenyl)-3 -cvclopentyl propanamide:
- Step B 6-fluoro-1.2.3.4-tetrahydroisoquinoline:
- Step C N-(2-chloro-4-(6-fluoro-3,4-dihvdroisoquinolin-2(7i : /)-yl)-6- (trifluorornethyl)phenv1)-3,3-dirnethylbutanamide:
- Step A N-(4-Bromo-2-chloro-6-methylphenyl)-3.3-dimethyl butanamide:
- Step B N-[2-Chloro-4-(3.4-dihydro-7//-isoquinolin-2-yl)-6-methyl-phenyll-3.3- dimethyl-butanamide :
- Step A 4-bromo-2-chloro-6-(trifluoromethyl ' )aniline:
- N-bromosuccinimide (910 mg, 5.1 ⁇ unol) was added at room temperature to a solution of 2-chloro-6-(trifluoromethyl)aniline (1.0 g, 5.1 mmol) and acetic acid (3 mL) in acetonitrile ( 10 mL). The mixture was stirred at reflux to 18h. The reaction mixture was then filtered through celite and concentrated to give the title compound, which was used in the next step without further purification.
- Step B N-(4-Bromo-2-chloro-6-trifluoromethyl-phenyl)-3-cvclopentyl propionamide:
- Step C N-r2-Chloro-4-f6-fluoro-3.4-dihvdro-7.H-isoquinolin-2-vn-6- trifluoromethyl- ⁇ henyli-3-cvclopentyl propionamide:
- Step A N-f4-Bromo-2.6-dimethyl-phenvD-3.3-dimethyl-butanamide:
- Step B N-r2.6-Dimethyl-4-(6-trifluoromethyl-3.4-dihydro-7H-isoquinolin-2-yl)- phenv ⁇ -3.3-dimethyl butanamide:
- Step A 4-bromo-2-chloro-6-ftrifluoromethyl ' ) aniline:
- Step B N-(4-bromo-2-chloro-6-(trifluoromethyl)phenyl)-3.3- dimethylbutanamide:
- Step C N-[2-Chloro-6-trifluoromethyl-4-f6-trifluoromethyl-3.4-dihvdro-7.H- isoquinolin-2-yl)-phenyll-3.3-dimethyl butanamide:
- Step A 4-bromo-2-chloro-6-(trifluoromethyl)aniline:
- Step B N-(4-bromo-2-chloro-6-(trifluoromethv ⁇ phenvI)-3.3- dimethylbutanamide:
- Step C N-[2-Chloro-4-(6-chloro-3,4-dihvdro-7H-isoquinolin-2-ylV6- trifluoromethyl- ⁇ henyll-3.3-dimethyl-butanamide:
- Step A N-(4-Bromo-2.6-dimethyl-phenyl)-3.3-dimethyl-butanamide:
- Step B N-r4-f6-Chloro-3.4-dihvdro-7//-isoquinolin-2-yl)-2.6-dimethyl-phenyll- 3.3 -dimethyl butanamide:
- Step A N-(4-Bromo-2,6-dimethyl-phenyl)-3.3-dimethyl-butanamide:
- Step B N-[4-(6-Fluoro-3.4-dmydro-7//-isoquinolin-2-ylV2.6-dimethylphenyl '
- Step A 4-bromo-2-chloro-6-(trifluoromethyl)aniline:
- N-bromosuccinimide (910 mg, 5.1 mmol) was added to a solution of 2-chloro-6- (trifluoromethyl)aniline (1.0 g, 5.1 mmol) in acetonitrile (10 mL) and acetic acid (3 mL) at room temperature. The mixture was stirred at reflux for 18h. The reaction mixture was then filtered through Celite and concentrated to give the title compound, which was used in the next step without further purification.
- Step B N-(4-bromo-2-chloro-6-(trifluoromethyl)phenyl)-3,3- dimethylbutanamide:
- Step C N-f2-Chloro-4-f7-fluoro-3.4-dihvdro-7//-isoquinolin-2-vn-6- trifluoromethyl-phenvn-3,3-dimethylbutanamide:
- Step A N-(4-Bromo-2.6-dimethyl-phenv ⁇ -3.3-dimethyl-butanamide:
- Step B N44-(7-Fluoro-3,4-dihvdro-7H-isoquinolin-2-yl)-2.6-dimethyl-phenyll- 3.3 -dimeth yl-butanamide:
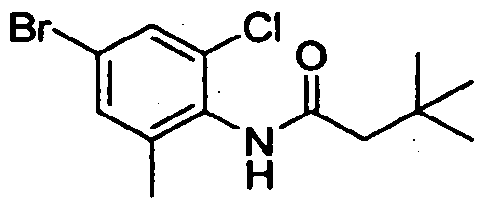
- Step A N-(4-Bromo-2-chloro-6-methyl-phenyl)-3.3-dimethyl-butanamide:
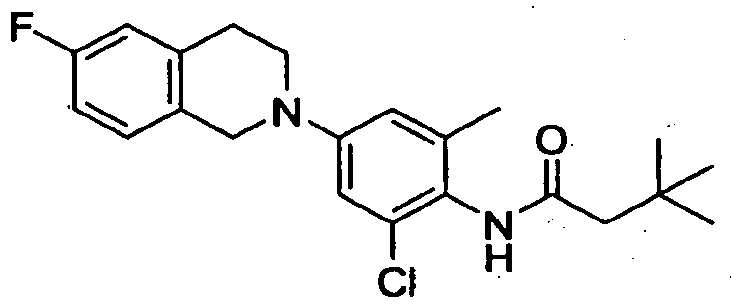
- Step B N-r2-Chloro-4-f6-fluoro-3.4-dihvdro-7H-isoquinolin-2-ylV6- methylphenyl "
- Step B N-[2-Chloro-4-(7-fluoro-3.4-dihvdro-7//-isoquinolin-2-yl)-6- methylphenyl1-3.3-dimethylbutanamide:
- Step A N-(4-Bromo-2-chloro-6-methyl-phenyl)-3,3-dimethyl-butanamide:
- Step B N-[2-Chloro-6-methyl-4-(6-trifluoromethyl-3.4-dihvdro-7H-isoquinolin- 2-ylVphenyll-3.3-dimethylbutanamide:
- Step A N-(4-Bromo-2-chloro-6-methyl-phenyl)-3.3-dimethvIbutanamide:
- Step A N-(4-Bromo-2-chloro-phenylV3.3-dimethyl-butanamide:
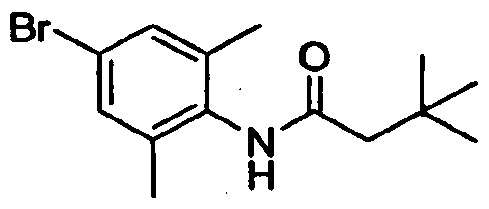
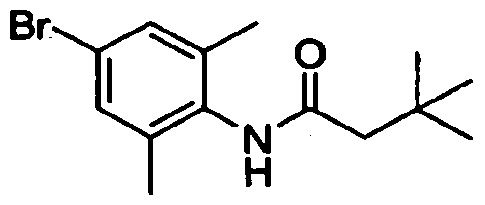
- Step A N-(4-Bromo-2-methyl-phenyl)-3.3-dimethylbutanamide:
- Step B N-r4-(6-Fluoro-3.4-dihvdro-7H-isoquinolin-2-yl)-2-methyl-phenyl]-3.3- dimethylbutanamide:
- Step B N-.
- Step A N-(4-Bromo-2-chloro-phenylV3.3-dimethylbutanamide:
- Step B N-r2-Chloro-4-(6-trifluoromethyl-3.4-dihvdro-7//-isoquinolin-2-yl')- phenyl]-3,3-dimethylbutanamide:
- Step A N-(4-Bromo-2-trifluoromethyl-phenyl)-3,3-dimethyl-butanamide:
- Step B N-[4-(7-Fluoro-3.4-dihvdro-///-isoquinolin-2-ylV2-trifluoromethyl- phenyl] -3.3 -dimeth ylbutanamide:
- Step A N-f4-Bromo-2-trifluoromemyl-phenvD-3.3-dimethylbutanamide:
- Step A N-(4-Bromo-2.6-dimethyl-phenyl)-3.3-dimethyl-butanamide:
- Step B N-[4-(6-Metho ⁇ y-3.4-dihvdro-/H-isoquinolin-2-yl)-2.6-dimethyl- phenyli-3 ,3-dimethylbutanamide:
- Step A N-(4-Bromo-2.6-dimethyl-phenyl)-3,3-dimethylbutanamide:
- Step B N-[2.6-Dimethyl-4-(7-trifluoromethyl-3.4-dihvdro-/H-isoquinolin-2-yl " )- phenyl1-33-dimethyl-butanamide:
- Step A 4-bromo-2-methoxy-6-methyl-aniline:
- Step B (4-Bromo-2-methoxy-6-methyl-phenylV3.3-dimethyl butanamide:
- Step C N-[4-(3.4-Dihvdro-7H-isoquinolin-2-yl')-2-methoxy-6-methyl-phenyl]- 3.3 -dimethyl-butanamide:
- reaction tube was sealed and reacted in microwave at 100 0 C for 2 hours.
- the reaction mixture was purified by silica gel column (ISCO, hexane/EtOAc, 0- 40%, 40 min) to give pure compound as a white solid.
- Step A N-(4-Bromo-2-chloro-6-trifluoromethoxy-phenyl)-3.3-dimethyl- butanamide:
- Step B N-f2-Chloro-4-f3.4-dihvdro-7//-isoquinolin-2-yl)-6-trifluoromethoxy- phenvn-3.3-dimethylbutanamide:
- Step B 5-Bromo-l .3-dimethoxy-2-amino-benzene:
- Step D N-[4-(3.4-Dihvdro-/H-isoquinolin-2-yl)-2,6-dimethoxy-phenyl]-3.3- dimethyl-butanamide:
- Step A N-(4-Bromo-2.6-dimethyl-phenv ⁇ -3.3-dimethyl-butyramide:
- Step B N-r2.6-Dimethyl-4-(6-trifluoromethyl-3.4-dihvdro-lH-isoquinolin-2-yl)- phenyll-3.3-dimethyl-butanamide:
- Step C N-[2,6-Dimethyl-4-(6-trifluoromethyl-3,4-dihydro-7//-isoquinolin-2-yl)- phenyl]-3.3-dimethyl-thiobutanamide:
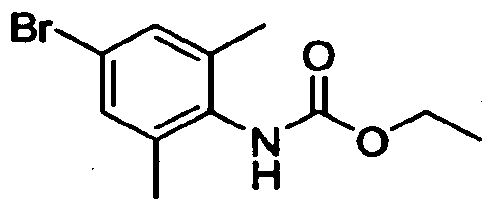
- Step A (4-Bromo-2.6-dimethyl-phenyl)-carbamic acid ethyl ester:
- Step B r2,6-Dimethyl-4-(6-trifluoroniethyl-3.4-dihydro-lH-isoquinoIin-2-v0- phenyli-carbamic acid ethyl ester:
- PC-12 cells were grown at 37 0 C and 5 % CO 2 in DMEM/F12 Medium (Dulbecco's Modified Eagle Medium with Nutrient Mix F-12, available from Invitrogen of Carlsbad, CA), supplemented with 10 % horse serum, 5 % fetal bovine serum, 2 mM glutamine, 100 U/ml penicillin, and 100 U/ml streptomycin. They were plated in poly-D- lysine-coated 96-well cell culture microplates at a density of 40,000 cells/well and differentiated with 100 ng/ml NGF-7s for 2-5 days.
- DMEM/F12 Medium Dulbecco's Modified Eagle Medium with Nutrient Mix F-12, available from Invitrogen of Carlsbad, CA
- the medium was aspirated, and the cells were washed once with 0.2 ml in wash buffer (25 mM HEPES, pH 7.4, 150 mM NaCl, 1 mM MgCl 2 , 0.8 mM NaH 2 PO 4 , 2 mM CaCl 2 ).
- the cells were then loaded with 0.2 ml Rb + loading buffer (wash buffer plus 5.4 mM RbCl 2 , 5 mM glucose) and incubated at 37 0 C for 2 h. Attached cells were quickly washed three times with buffer (same as Rb + loading buffer, but containing 5.4 mM KCl instead of RbCl) to remove extracellular Rb + .
- Rb + Ly S were quantified using an ICR8000 flame atomic absorption spectrometer (Aurora Biomed Inc., Vancouver, B.C.) under conditions defined by the manufacturer. Samples 0.05 ml in volume were processed automatically from microtiter plates by dilution with an equal volume OfRb + sample analysis buffer and injection into an air- acetylene flame. The amount OfRb + in the sample was measured by absorption at 780 nm using a hollow cathode lamp as light source and a PMT detector. A calibration curve covering the range 0-5 mg/L Rb + in sample analysis buffer was generated with each set of plates. The percent Rb + efflux (F) was defined by
- F [Rb + SUp / (Rb + SUp + Rb + Ly 5 )] X 100 %.
- F c is the efflux in the presence of compound in depolarization buffer
- F b is the efflux in basal buffer
- F s is the efflux in depolarization buffer
- F c is the efflux in the presence of compound in depolarization buffer.
- the efflux (F) and compound concentration relationship was plotted to calculate an EC 50 value, a compound's concentration for 50% of maximal Rb + efflux. The results are shown below.
- the MES testing protocol is based on procedures established at the National Institute of Neurological Disorders and Stroke in conjunction with the Anticonvulsant Screening Program (ASP) at the University of Utah (White, H.S., Woodhead, J.H., Wilcox, K.S., Stables, J.P., Kupferberg, HJ and Wolf, H.H. 2002. "General Principles: Discovery and Preclinical Development of Antiepileptic Drugs, " in Antiepileptic Drugs, 5 th Edition, R.H. Levy, ed.; R.H. Mattson, B.S. Meldrum, and E. Perucca. Philadelphia, Lippincott Williams & Wilkins.), The goal of the test rapid identification and characterization of the in vivo anticonvulsant activity of any compounds that have been shown active in PC- 12 cellular based Rb + efflux assay.
- mice Adult male CF-I albino mice (18-25 g, Charles River Laboratories) are exclusively used for in-house MES screen of compounds. Male Sprague-Dawley albino rats (100-125g, Charles River Laboratories) are also used to test anticonvulsant compounds. Variability of test outcomes is reduced by using animals of the same sex, age, and weight. Animals are permitted to rest and recover from transit for at least 48 hr prior to experimentation. Animals are used for AED testing only once. In some instances, the animals may be anesthetized prior to blood collection and/or whole brain extraction for pharmacokinetic assay. All animals are maintained and handled as outlined in standard animal care guidelines.
- testing compounds are prepared as suspensions in 0.5% methyl cellulose (Sigma, Cat # M0512, Viscosity 4000 cP at 20 0 C) in water, regardless of solubility. Dry powder compounds are initially ground with a glass rod in a test tube in several drops of methyl cellulose to create a paste and to break down any large chunks. After several minutes of grinding, the volume of the suspension is increased to the final concentration desired. The suspension is then sonicated using a Branson sonicator model 3510 in a water bath at room temperature for 15 minutes. Compound suspensions are further vortexed prior to animal dosing.
- DMSO is used to initially solubilize compounds in small volumes and then this solution is added to the 0.5% methyl cellulose solution, in order to create more even and less aggregated compound suspensions.
- the final concentration of DMSO is 3.75%, an amount with no apparent toxicity or neuroprotective effects in our usual rotarod and MES tests.
- Methyl cellulose/DMSO compound suspensions are identically prepared for intraperitoneally (i.p.) to mice or orally (p.o.) to rat dosing. Initially the animals are weighed with an electronic scale and then marked. Data recording sheets are generated for each compound assessment. Mice or rats are dosed with the compound suspension at 0.01 mL/g of body weight.
- mice The typical injection volume range is between 180-250 ⁇ l for mice.
- Compounds are dosed by i.p. to mice using a 25 or 22 gauge needle, depending on the viscosity of the suspension. Rats are p.o. dosed using a flexible feeding tube, typically starting at a compound dose of 5 mg/kg.
- a Rodent Electroconvulsive Stimulator (Model 200, Hamit-Darvin-Freesh, Snow Canyon Clinic, Ivins, UT) is used for MES testing.
- a 60-Hz alternating current (50 mA for mice; 150 mA for rats) is delivered for 0.2 seconds through corneal electrodes to the mice.
- a drop of 0.5% tetracaine (Sigma, Cat. # T-7508) solution is placed on the eye prior to current delivery.
- the electrodes are subsequently placed gently onto the eyes of the animal and the electrical shock is initiated by triggering through a foot-pedal activator.
- the animals are restrained by hand and gently released as the shock is delivered and the seizure commences. Animals are monitored for hind limb tonic extension as the end point for this test.
- time-to-peak effect studies are initially performed using 0.5, 1, 2, 4, 8 and 24 hr time points, typically using a single 5 or 25 mg/kg dose.
- the determined time-to-peak effect is used for further titration of a compound's potency (ED50, the dose of a drug that protects 50% of animals from electrical induced seizure) in both mouse and rat models.
- ED50 the dose of a drug that protects 50% of animals from electrical induced seizure
- 8 animals are used per concentration and dose (normal 5 concentrations) is varied until a full dose response curve can be obtained.
- Probit analysis ASP method
- non-linear regression analysis on Graph Pad is used to calculate an ED 50 value for the test compound.
- rats are visually observed for acute toxicity signs for approximately one minute in the open field test.
- rats are gently placed into a plexiglass enclosure and are monitored for behavior consistent with toxicity including ataxia, trembling, hypoactivity (including failure to seek the walls), hypersensitivity, lack of exploratory behavior and lack of avoidance of the open area.
- toxicity including ataxia, trembling, hypoactivity (including failure to seek the walls), hypersensitivity, lack of exploratory behavior and lack of avoidance of the open area.
- hypoactivity including failure to seek the walls
- hypersensitivity including failure to seek the walls
- lack of exploratory behavior and lack of avoidance of the open area.
- the rats typically if the rats exhibits two or more of these abnormal behaviors they are scored as toxic.
- A EC 50 ⁇ 1 nM
- C 10 nM ⁇ EC 50 ⁇ 50 nM
- D 50 nM ⁇ EC 50 ⁇ 500 nM ⁇ :0.12 ⁇ ED50 ⁇ 1.2 ⁇ : 1.2 ⁇ ED50 ⁇ 12 ⁇ :12 ⁇ ED50
- Xenopus laevis extracted ovaries were purchased from eNASCO (LM00935MX, eNASCO Fort Atkinson, WI). Following manual dissection of the oocytes into smaller groups, the oocytes were defolliculated by enzymatic treatment with collagenase type 2 (LS004177, Worthington, Lakewood, NJ) for 1-1/2 hour in the presence of calcium-free Culture Bath solution (88 mM NaCl, 1 mM KCl, 0.82 mM MgSO 4 , 2.4 mM NaHCO 3 , and 5 mM HEPES, pH 7.5).
- calcium-free Culture Bath solution 88 mM NaCl, 1 mM KCl, 0.82 mM MgSO 4 , 2.4 mM NaHCO 3 , and 5 mM HEPES, pH 7.5.
- Oocytes were then kept in supplemented Culture Bath solution (88 mM NaCl, 1 mM KCl, 0.82 mM MgSO 4, 0.9 mM CaCl 2 , 2.4 mM NaHCO 3 , 1 mM sodium pyruvate, 0.05 mg/ml Geneticin, 100 U/ml penicillin, 0.1 mg/ml streptomycin and 5 mM HEPES, pH 7.5) at 19 0 C for 24 hours before injection of cRNA. Approximately 50 nl cRNA (about 50 ng) was injected for KCNQl , KCNQ4, and KCNQ5 using a Nanoject microinjector (Drummond, Broomall, PA, USA).
- a Nanoject microinjector (Drummond, Broomall, PA, USA).
- cRNA's were mixed in equal molar ratios before injection of approximately 50 nl.
- the mixtures contained about 10 + 10 ng and 12.5 + 2.5 ng cRNA, respectively.
- the smaller amounts are needed because larger currents arise when KCNQ2/KCNQ3 and KCNQ1/KCNE1 are co-expressed.
- Oocytes were kept in Culture Barth solution at 19°C which was changed daily and currents were recorded after 3 to 5 days.
- Electrophysiology KCNQ channel currents expressed in Xenopus laevis oocytes were recorded using a two-electrode voltage-clamp.
- the recordings were made at room temperature in recording solution (96 mM NaCl, 2 mM KCl, 1 mM MgCl 2 , 1.8 mM CaCl 2 , and 5 mM HEPES, pH 7.5) using a two-electrode voltage-clamp amplifier (OC T 725C, Warner Instrument, Hamden, CT, USA).
- the oocytes were placed in custom built perfusion chambers connected to a continuous flow system and impaled with a current electrode and a voltage-clamp electrode pulled from borosilicate glass on a Flaming/Brown Micropipette Puller (Sutter Instruments Co, Novato, CA, USA). Recording electrodes were filled with 3 M KCl and had a resistance of 0.5 to 2.5 M ⁇ .
- C2 is the recorded steady current in the presence of follow-on compound at its EC 7s and Cl is the recorded steady current in the presence of Retigabine at its EC 75 .
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pain & Pain Management (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Other In-Based Heterocyclic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
Claims
Priority Applications (13)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NZ575652A NZ575652A (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| DK07837199.4T DK2061465T3 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4- (N-azacycloalkyl) anilides as potassium channel modulators |
| AU2007288253A AU2007288253B2 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(N-azacycloalkyl) anilides as potassium channel modulators |
| MX2009002002A MX2009002002A (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators. |
| JP2009525618A JP5419691B2 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4- (N-azacycloalkyl) anilide as potassium channel modulators |
| EP07837199.4A EP2061465B1 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| BRPI0716715-6A BRPI0716715B1 (en) | 2006-08-23 | 2007-08-22 | 4-(N-AZACICLOALKYL) DERIVATIVES AS POTASSIUM CHANNEL MODULATORS, THEIR USES, PRODUCT, COMPOSITION, TABLET AND CAPSULE |
| CA2661462A CA2661462C (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| ES07837199T ES2420960T3 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4- (N-azacycloalkyl) anilides as modulators of potassium channels |
| PL07837199T PL2061465T3 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| IL197161A IL197161A (en) | 2006-08-23 | 2009-02-22 | Derivatives of 4-(n-azacycloalkyl)anilides, compositions containing the same and uses thereof |
| ZA2009/01977A ZA200901977B (en) | 2006-08-23 | 2009-03-20 | Derivatives of 4-(in-azacycloalkyl) anilides as potassium channel modulators |
| NO20091183A NO343575B1 (en) | 2006-08-23 | 2009-03-20 | Derivatives of 4- (N-azacycloalkyl) anilides, process for the preparation of such compounds, preparations comprising such compounds and the use of such compounds for the prevention or treatment of disease |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US83994106P | 2006-08-23 | 2006-08-23 | |
| US60/839,941 | 2006-08-23 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2008024398A2 true WO2008024398A2 (en) | 2008-02-28 |
| WO2008024398A3 WO2008024398A3 (en) | 2008-06-26 |
| WO2008024398B1 WO2008024398B1 (en) | 2008-08-14 |
Family
ID=39107384
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/018571 Ceased WO2008024398A2 (en) | 2006-08-23 | 2007-08-22 | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
Country Status (23)
| Country | Link |
|---|---|
| US (1) | US8293911B2 (en) |
| EP (1) | EP2061465B1 (en) |
| JP (1) | JP5419691B2 (en) |
| KR (1) | KR20090079191A (en) |
| CN (1) | CN101563085A (en) |
| AR (1) | AR062508A1 (en) |
| AU (1) | AU2007288253B2 (en) |
| BR (1) | BRPI0716715B1 (en) |
| CA (1) | CA2661462C (en) |
| CL (1) | CL2007002455A1 (en) |
| DK (1) | DK2061465T3 (en) |
| ES (1) | ES2420960T3 (en) |
| IL (1) | IL197161A (en) |
| MX (1) | MX2009002002A (en) |
| NO (1) | NO343575B1 (en) |
| NZ (1) | NZ575652A (en) |
| PL (1) | PL2061465T3 (en) |
| PT (1) | PT2061465E (en) |
| RU (1) | RU2009110172A (en) |
| SG (1) | SG174095A1 (en) |
| TW (1) | TW200827339A (en) |
| WO (1) | WO2008024398A2 (en) |
| ZA (1) | ZA200901977B (en) |
Cited By (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009018466A1 (en) * | 2007-08-01 | 2009-02-05 | Valeant Pharmaceuticals International, Inc. | Naphthyridine derivatives as potassium channel modulators |
| WO2008157404A3 (en) * | 2007-06-13 | 2009-02-19 | Valeant Pharmaceuticals Int | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| US7786146B2 (en) | 2007-08-13 | 2010-08-31 | Valeant Pharmaceuticals International | Derivatives of 5-amino-4,6-disubstituted indole and 5-amino-4,6-disubstituted indoline as potassium channel modulators |
| US7799832B2 (en) | 2003-10-23 | 2010-09-21 | Valeant Pharmaceuticals North America | Combinations of retigabine and sodium channel inhibitors or sodium channel-influencing active compounds for treating pains |
| US7960436B2 (en) | 2006-06-05 | 2011-06-14 | Valeant Pharmaceuticals International | Substituted arylamino-1,2,3,4-tetrahydro naphthalenes and-2,3-dihydro-1H-indenes as potassium channel modulators |
| WO2010130260A3 (en) * | 2009-05-11 | 2011-06-16 | H. Lundbeck A/S | Stable forms of n-(2,6-dimethyl-4-morpholin-4-yl-phenyl)-3,3-dimethyl-butyramide |
| WO2011094186A1 (en) * | 2010-02-01 | 2011-08-04 | Valeant Pharmaceuticals International | Derivatives of 4-(n-azacycloalkyl) aniilides as potassium channel modulators |
| US8030518B2 (en) | 2006-11-28 | 2011-10-04 | Valeant Pharmaceuticals International | 1,4 diamino bicyclic retigabine analogues as potassium channel modulators |
| WO2012004698A1 (en) * | 2010-07-08 | 2012-01-12 | Pfizer Inc. | Piperidinyl pyrimidine amides as kv7 potassium channel openers |
| US8293911B2 (en) | 2006-08-23 | 2012-10-23 | Valeant Pharmaceuticals International | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| WO2013055984A1 (en) | 2011-10-14 | 2013-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| WO2013056060A1 (en) | 2011-10-14 | 2013-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| WO2013056034A1 (en) | 2011-10-14 | 2013-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| US8741901B2 (en) | 2004-07-15 | 2014-06-03 | Albany Molecular Research, Inc. | Aryl- and heteroaryl-substituted tetrahydroisoquinolines and use thereof to block reuptake of norepinephrine, dopamine, and serotonin |
| US8802696B2 (en) | 2009-05-12 | 2014-08-12 | Albany Molecular Research, Inc. | 7-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydroisoqu inoli and use thereof |
| US8815894B2 (en) | 2009-05-12 | 2014-08-26 | Bristol-Myers Squibb Company | Crystalline forms of (S)-7-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydroisoquinoline and use thereof |
| WO2014160668A1 (en) | 2013-03-25 | 2014-10-02 | Bristol-Myers Squibb Company | Tetrahydroisoquinolines containing substituted azoles as factor xia inhibitors |
| US9034899B2 (en) | 2009-05-12 | 2015-05-19 | Albany Molecular Research, Inc. | Aryl, heteroaryl, and heterocycle substituted tetrahydroisoquinolines and use thereof |
| US9498476B2 (en) | 2008-06-04 | 2016-11-22 | Albany Molecular Research, Inc. | Crystalline form of 6-[(4S)-2-methyl-4-(2-naphthyl)-1,2,3,4-tetrahydroisoquinolin-7-yl]pyridazin-3-amine |
| WO2019118657A1 (en) * | 2017-12-13 | 2019-06-20 | Scifluor Life Sciences, Inc. | Fluorinated 4-(substituted amino)phenyl carbamate derivatives |
| WO2019217924A1 (en) * | 2018-05-11 | 2019-11-14 | Xenon Pharmaceuticals Inc. | Methods for enhancing the bioavailability and exposure of a voltage-gated potassium channel opener |
| EP3468947A4 (en) * | 2016-06-10 | 2020-01-08 | SciFluor Life Sciences, Inc. | FLUORINATED 2-AMINO-4- (SUBSTITUTED AMINO) PHENYLCARBAMATE DERIVATIVES |
| WO2020157126A1 (en) | 2019-01-29 | 2020-08-06 | Università degli Studi di Salerno | Modulators of potassium ion channels and uses thereof |
| WO2021072307A1 (en) * | 2019-10-10 | 2021-04-15 | Xenon Pharmaceuticals Inc. | Solid state crystalline forms of a selective potassium channel modulator |
| WO2021092439A1 (en) * | 2019-11-08 | 2021-05-14 | Xenon Pharmaceuticals Inc. | Methods of treating depressive disorders |
| US11365181B2 (en) | 2018-05-22 | 2022-06-21 | Shanghai Zhimeng Biopharma Inc. | P-phenylenediamine derivative as potassium channel regulator and preparation method and medical application thereof |
| WO2022174667A1 (en) | 2021-10-27 | 2022-08-25 | 上海挚盟医药科技有限公司 | Compound as potassium channel regulator, and preparation and use thereof |
| AU2021359042B1 (en) * | 2021-10-27 | 2023-04-13 | Shanghai Zhimeng Biopharma, Inc. | Compound as potassium channel regulator and preparation and use thereof |
| RU2798327C1 (en) * | 2021-10-27 | 2023-06-21 | Шанхай Чжимэн Биофарма, Инк. | Compound as a potassium channels regulator, its production and application |
| WO2023134673A1 (en) | 2022-01-14 | 2023-07-20 | 中国科学院上海药物研究所 | High-selectivity kcnq4 potassium channel agonist, preparation method therefor and use thereof |
| WO2023239729A1 (en) * | 2022-06-08 | 2023-12-14 | Xenon Pharmaceuticals Inc. | Pyridinamine derivatives and their use as potassium channel modulators |
| US11957675B2 (en) | 2021-02-09 | 2024-04-16 | Xenon Pharmaceuticals Inc. | Methods and uses for treating anhedonia |
| US11993586B2 (en) | 2018-10-22 | 2024-05-28 | Novartis Ag | Crystalline forms of potassium channel modulators |
| CN118530144A (en) * | 2023-02-21 | 2024-08-23 | 中国医学科学院药物研究所 | A preparation method of pinegabine |
| WO2025038930A1 (en) | 2023-08-17 | 2025-02-20 | Xenon Pharmaceuticals, Inc. | Azepane and pyrrolidine compounds and uses thereof |
| WO2025038920A1 (en) | 2023-08-17 | 2025-02-20 | Xenon Pharmaceuticals, Inc. | Heteroaryl amine compounds and uses thereof |
| WO2026006669A1 (en) * | 2024-06-28 | 2026-01-02 | Xenon Pharmaceuticals Inc. | Prodrugs of n-[4-(6-fluoro-3,4-dihydro-2(1 h)-isoquinolinyl)-2,6-dimethylphe nyl]-3,3-dimethyl- butanamide |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080045534A1 (en) * | 2006-08-18 | 2008-02-21 | Valeant Pharmaceuticals North America | Derivatives of 1,3-diamino benzene as potassium channel modulators |
| US8722929B2 (en) * | 2006-10-10 | 2014-05-13 | Valeant Pharmaceuticals International | N-[2-amino-4-(phenylmethoxy)phenyl] amides and related compounds as potassium channel modulators |
| AU2009271302A1 (en) * | 2008-06-24 | 2010-01-21 | Valeant Pharmaceuticals International | Benzyloxy anilide derivatives useful as potassium channel modulators |
| CN103073455B (en) | 2011-10-25 | 2015-08-19 | 中国科学院上海药物研究所 | Kcnq potassium channel agonist, Preparation Method And The Use that one class is novel |
| CN103694163A (en) * | 2012-09-27 | 2014-04-02 | 上海先声药物研究有限公司 | Compounds as Potassium Channel Modulators |
| US10329516B2 (en) * | 2016-12-20 | 2019-06-25 | International Flavors & Fragrances Inc. | Organoleptic compounds |
| DK3737677T3 (en) | 2018-01-10 | 2022-01-10 | Allinky Biopharma | TETRAHYDROISOQUINOLINE COMPOUNDS |
| BR112022010733A2 (en) * | 2019-12-06 | 2022-08-23 | Xenon Pharmaceuticals Inc | METHODS TO TREAT PAIN |
| CN114057641B (en) * | 2020-08-07 | 2025-02-18 | 上海挚盟医药科技有限公司 | Tetrahydroisoquinoline compounds as potassium channel regulators and their preparation and use |
| EP4291185A1 (en) | 2021-02-09 | 2023-12-20 | Xenon Pharmaceuticals Inc. | Conjoint therapy for treating seizure disorders |
| JP7681132B2 (en) * | 2021-08-26 | 2025-05-21 | 紐欧申医薬(上海)有限公司 | Potassium channel modulators, compositions and uses |
| WO2023125935A1 (en) * | 2021-12-31 | 2023-07-06 | 上海翰森生物医药科技有限公司 | Benzopyridine derivative regulator, and preparation method therefor and use thereof |
| CN116535353A (en) * | 2022-01-25 | 2023-08-04 | 上海挚盟医药科技有限公司 | Amide compounds as potassium channel regulators and their preparation and application |
| WO2024050389A1 (en) * | 2022-08-30 | 2024-03-07 | Biohaven Therapeutics Ltd. | Combination therapies including metal channel activators and nmda receptor antagonists |
| TW202430510A (en) * | 2022-09-30 | 2024-08-01 | 大陸商上海翰森生物醫藥科技有限公司 | Piperidine polycyclic derivative regulator, preparation method therefor and application thereof |
| AU2024344798A1 (en) * | 2023-09-19 | 2026-03-19 | Xenon Pharmaceuticals Inc. | Deuterium and aryl amine compounds and uses thereof |
| TW202540067A (en) * | 2024-02-08 | 2025-10-16 | 大陸商上海摯盟醫藥科技有限公司 | Crystal form of tetrahydroisoquinoline compound and its application |
| WO2025179128A1 (en) | 2024-02-22 | 2025-08-28 | Xenon Pharmaceuticals Inc. | Azetukalner for use in the treatment of depression, anhedonia or a seizure disorder such as epilepsy in pediatric subjects |
| WO2026037240A1 (en) * | 2024-08-12 | 2026-02-19 | 江西科睿药业有限公司 | Potassium channel modulator and preparation method therefor, pharmaceutical composition and use |
Family Cites Families (83)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4181803A (en) * | 1973-12-14 | 1980-01-01 | Eisai Co., Ltd. | Propiophenone derivatives and preparation thereof |
| DE3369315D1 (en) * | 1982-10-27 | 1987-02-26 | Degussa | Derivatives of 2-amino-3-acylamino-6-benzylamino pyridine with an anti-epileptic activity |
| DE3337593A1 (en) | 1982-10-27 | 1984-05-03 | Degussa Ag, 6000 Frankfurt | 2-Amino-3-acylamino-6-benzylaminopyridine derivatives having antiepileptic action |
| DE3665538D1 (en) | 1985-01-23 | 1989-10-19 | Asta Pharma Ag | Synergistic combination of flupirtin and non-steroidal anti-phlogistics |
| DE3604575A1 (en) | 1985-02-23 | 1986-08-28 | Degussa Ag, 6000 Frankfurt | Combination of flupirtine and spasmolytics with anticholinergic activity |
| DE3663775D1 (en) * | 1985-02-23 | 1989-07-13 | Asta Pharma Ag | Combination of flupirtin with spasmolytics having an anticholinergic effect |
| JP2583067B2 (en) * | 1987-08-04 | 1997-02-19 | 住友化学工業株式会社 | Monoazo compound and method for dyeing or printing hydrophobic fiber material using the same |
| GB8800199D0 (en) * | 1988-01-06 | 1988-02-10 | Beecham Group Plc | Pharmaceutical preparation |
| MC2029A1 (en) | 1988-05-16 | 1990-04-25 | Asta Pharma Ag | (N-HETEROCYCLYL) -3 DIAMINO-2,6 SUBSTITUTED PYRIDINES AND THEIR N-OXIDES, PREPARATION OF THESE COMPOUNDS AND THEIR APPLICATION AS MEDICAMENTS |
| US5629307A (en) * | 1989-10-20 | 1997-05-13 | Olney; John W. | Use of ibogaine in reducing excitotoxic brain damage |
| US6004945A (en) * | 1990-05-10 | 1999-12-21 | Fukunaga; Atsuo F. | Use of adenosine compounds to relieve pain |
| IN172468B (en) * | 1990-07-14 | 1993-08-14 | Asta Medica Ag | |
| US5643921A (en) * | 1990-09-26 | 1997-07-01 | E.R. Squibb & Sons, Inc. | Cardiopulmonary bypass and organ transplant using a potassium channel activator |
| US5234947A (en) * | 1991-11-07 | 1993-08-10 | New York University | Potassium channel activating compounds and methods of use thereof |
| DE4200259A1 (en) * | 1992-01-08 | 1993-07-15 | Asta Medica Ag | NEW 1,2,4-TRIAMINOBENZOL DERIVATIVES AND METHOD FOR THE PRODUCTION THEREOF |
| US5262419A (en) * | 1992-06-11 | 1993-11-16 | E. R. Squibb & Sons, Inc. | Method for the prophylaxis and/or treatment of ulcerative gastrointestinal conditions using a potassium channel activator |
| CA2115792C (en) * | 1993-03-05 | 2005-11-01 | David J. Mayer | Method for the treatment of pain |
| US5428039A (en) * | 1994-02-20 | 1995-06-27 | The Center For Innovative Technology | Method for electively achieving reversible hyperpolarized cardiac arrest |
| AU8071894A (en) * | 1994-09-22 | 1996-04-09 | Jonathan M Licht | Compositions useful for the preparation of medicines for treating a variety of intractable disorders |
| US5679706A (en) * | 1994-09-30 | 1997-10-21 | Bristol-Myers Squibb Company | Combination of a potassium channel activator and an antiarrhythmic agent |
| MX9703988A (en) * | 1994-12-12 | 1998-02-28 | Omeros Med Sys Inc | SOLUTION AND IRRIGATION METHOD FOR THE INHIBITION OF PAIN, INFLAMMATION AND IT IS SPASM. |
| DE19539861A1 (en) * | 1995-10-26 | 1997-04-30 | Asta Medica Ag | Use of 4-amino-4- (4-fluorobenzylamino) -1-ethoxy-carbonylaminobenzen for the prophylaxis and treatment of the consequences of acute and chronic cerebral low blood circulation and neurodegenerative diseases |
| DE19701694A1 (en) * | 1997-01-20 | 1998-07-23 | Asta Medica Ag | New modifications of the 2-amino-4- (4-fluorobenzylamino) -l-ethoxycarbonyl-aminobenzen and processes for their preparation |
| US5760007A (en) * | 1997-07-16 | 1998-06-02 | Ortho Pharmaceutical Corporation | Anticonvulsant derivatives useful in treating neuropathic pain |
| US6218411B1 (en) * | 1997-08-08 | 2001-04-17 | Chugai Seiyaku Kabushiki Kaisha | Therapeutics for diabetic complications |
| US6265417B1 (en) * | 1997-12-18 | 2001-07-24 | Abbott Laboratories | Potassium channel openers |
| US6593335B1 (en) * | 1997-12-18 | 2003-07-15 | Abbott Laboratories | Potassium channel openers |
| US6333337B1 (en) * | 1998-01-27 | 2001-12-25 | Icagen, Inc. | Potassium channel inhibitors |
| US6211171B1 (en) * | 1998-05-19 | 2001-04-03 | Dalhousie University | Use of antidepressants for local analgesia |
| JP3441970B2 (en) | 1998-06-30 | 2003-09-02 | 株式会社サミー | Method and apparatus for producing tofu |
| JP2000143510A (en) | 1998-11-16 | 2000-05-23 | Taisho Pharmaceut Co Ltd | External composition |
| US6395736B1 (en) * | 1998-12-14 | 2002-05-28 | Cellegy Pharmaceuticals, Inc. | Compositions and methods for the treatment of anorectal disorders |
| US6281211B1 (en) * | 1999-02-04 | 2001-08-28 | Euro-Celtique S.A. | Substituted semicarbazides and the use thereof |
| GB9903476D0 (en) * | 1999-02-17 | 1999-04-07 | Zeneca Ltd | Therapeutic agents |
| US6451857B1 (en) * | 1999-03-10 | 2002-09-17 | Warner-Lambert Company | Analgesic compositions comprising anti-epileptic compounds and methods of using same |
| WO2000055137A1 (en) | 1999-03-17 | 2000-09-21 | Signal Pharmaceuticals, Inc. | Compounds and methods for modulation of estrogen receptors |
| AT409083B (en) * | 1999-04-01 | 2002-05-27 | Sanochemia Pharmazeutika Ag | PHARMACEUTICAL PREPARATION CONTAINING TOLPERISON FOR ORAL ADMINISTRATION |
| US6610324B2 (en) | 1999-04-07 | 2003-08-26 | The Mclean Hospital Corporation | Flupirtine in the treatment of fibromyalgia and related conditions |
| GB9915414D0 (en) | 1999-07-01 | 1999-09-01 | Glaxo Group Ltd | Medical use |
| EP1200073B1 (en) | 1999-07-06 | 2007-01-10 | Eli Lilly And Company | SELECTIVE iGluR 5? RECEPTOR ANTAGONISTS FOR THE TREATMENT OF MIGRAINE |
| US6472165B1 (en) * | 1999-08-03 | 2002-10-29 | Arzneimittelwerk Dresden Gmbh | Modulatory binding site in potassium channels for screening and finding new active ingredients |
| BR0012934A (en) * | 1999-08-04 | 2003-07-29 | Icagen Inc | Processes for reducing pain and anxiety |
| US6495550B2 (en) * | 1999-08-04 | 2002-12-17 | Icagen, Inc. | Pyridine-substituted benzanilides as potassium ion channel openers |
| EP1208085B1 (en) * | 1999-08-04 | 2007-12-05 | Icagen, Inc. | Benzanilides as potassium channel openers |
| US6117900A (en) | 1999-09-27 | 2000-09-12 | Asta Medica Aktiengesellschaft | Use of retigabine for the treatment of neuropathic pain |
| US6383511B1 (en) * | 1999-10-25 | 2002-05-07 | Epicept Corporation | Local prevention or amelioration of pain from surgically closed wounds |
| US6538004B2 (en) * | 2000-03-03 | 2003-03-25 | Abbott Laboratories | Tricyclic dihydropyrazolone and tricyclic dihydroisoxazolone potassium channel openers |
| US20020015730A1 (en) * | 2000-03-09 | 2002-02-07 | Torsten Hoffmann | Pharmaceutical formulations and method for making |
| US6348486B1 (en) * | 2000-10-17 | 2002-02-19 | American Home Products Corporation | Methods for modulating bladder function |
| US6589986B2 (en) * | 2000-12-20 | 2003-07-08 | Wyeth | Methods of treating anxiety disorders |
| US6469042B1 (en) * | 2001-02-20 | 2002-10-22 | Bristol-Myers Squibb Company | Fluoro oxindole derivatives as modulators if KCNQ potassium channels |
| AR033095A1 (en) * | 2001-04-04 | 2003-12-03 | Wyeth Corp | METHODS FOR THE TREATMENT OF HYPERACTIVE GASTRIC MOTILITY |
| EP1392644A1 (en) * | 2001-05-31 | 2004-03-03 | Bristol-Myers Squibb Company | Cinnamide derivatives as kcnq potassium channel modulators |
| GB0121214D0 (en) * | 2001-08-31 | 2001-10-24 | Btg Int Ltd | Synthetic method |
| WO2003068769A1 (en) | 2002-02-12 | 2003-08-21 | Pfizer Inc. | Non-peptide compounds affecting the action of gonadotropin-releasing hormone (gnrh) |
| AUPS110302A0 (en) * | 2002-03-14 | 2002-04-18 | Biomolecular Research Institute Limited | Novel chalcone derivatives and uses thereof |
| US6984647B2 (en) | 2002-05-17 | 2006-01-10 | Janssen Pharmaceutica N.V. | Aminotetralin-derived urea modulators of vanilloid VR1 receptor |
| WO2003106454A1 (en) * | 2002-06-12 | 2003-12-24 | Orchid Chemicals & Pharmaceuticals Ltd | 1h-isoquinoline-oxazolidinone derivaties and their use as antibacterial agents |
| AUPS312602A0 (en) * | 2002-06-21 | 2002-07-18 | James Cook University | Organ arrest, protection, preservation and recovery |
| US7419981B2 (en) * | 2002-08-15 | 2008-09-02 | Pfizer Inc. | Synergistic combinations of an alpha-2-delta ligand and a cGMP phosphodieterse 5 inhibitor |
| AU2003294441A1 (en) * | 2002-11-22 | 2004-06-18 | Bristol-Myers Squibb Company | 1-aryl-2-hydroxyethyl amides as potassium channel openers |
| AU2003303484C1 (en) * | 2002-12-23 | 2012-06-14 | Icagen, Inc. | Quinazolinones as potassium channel modulators |
| PT1578740E (en) | 2002-12-27 | 2007-05-31 | Lundbeck & Co As H | 1,2,4-triaminobenzene derivatives useful for treating disorders of the central nervous system |
| WO2004080950A1 (en) | 2003-03-14 | 2004-09-23 | H. Lundbeck A/S | Substituted aniline derivatives |
| SG157231A1 (en) | 2003-03-21 | 2009-12-29 | Lundbeck & Co As H | Substituted p-diaminobenzene derivatives |
| WO2004096767A1 (en) | 2003-04-25 | 2004-11-11 | H. Lundbeck A/S | Sustituted indoline and indole derivatives |
| AU2004243444A1 (en) | 2003-05-27 | 2004-12-09 | Altana Pharma Ag | Pharmaceutical combinations of a proton pump inhibitor and a compound which modifies gastrointestinal motility |
| US20050070570A1 (en) * | 2003-06-18 | 2005-03-31 | 4Sc Ag | Novel potassium channels modulators |
| JP2007505142A (en) * | 2003-09-10 | 2007-03-08 | セダーズ−シナイ メディカル センター | Potassium channel-mediated delivery of drugs across the blood brain barrier |
| WO2005046578A2 (en) * | 2003-09-23 | 2005-05-26 | Merck & Co., Inc. | Isoquinolinone potassium channel inhibitors |
| US7799832B2 (en) * | 2003-10-23 | 2010-09-21 | Valeant Pharmaceuticals North America | Combinations of retigabine and sodium channel inhibitors or sodium channel-influencing active compounds for treating pains |
| DE10359335A1 (en) | 2003-10-23 | 2005-05-25 | Viatris Gmbh & Co. Kg | Combinations of potassium channel openers and sodium channel inhibitors or sodium channel influencing agents for the treatment of pain |
| ES2235626B1 (en) | 2003-11-10 | 2006-11-01 | Almirall Prodesfarma, S.A. | MASTICABLE ADMINISTRATION FORMS, NOT INDIVIDUALLY DOSED COMPRESSED. |
| TWI357901B (en) | 2004-03-12 | 2012-02-11 | Lundbeck & Co As H | Substituted morpholine and thiomorpholine derivati |
| WO2005100349A2 (en) | 2004-04-13 | 2005-10-27 | Icagen, Inc. | Polycyclic pyridines as potassium ion channel modulators |
| BRPI0510593A (en) * | 2004-05-03 | 2007-11-20 | Univ Duke | compositions to affect weight loss |
| UA89503C2 (en) | 2004-09-13 | 2010-02-10 | Х. Луннбек А/С | Substituted aniline derivatives |
| WO2006054513A1 (en) | 2004-11-19 | 2006-05-26 | Kissei Pharmaceutical Co., Ltd. | Preventive or therapeutic agent for neuropathic pain |
| EP1688141A1 (en) * | 2005-01-31 | 2006-08-09 | elbion AG | The use of flupirtine for the treatment of overactive bladder and associated diseases, and for the treatment of irritable bowel syndrome |
| WO2006092143A1 (en) | 2005-03-03 | 2006-09-08 | H. Lundbeck A/S | Substituted pyridine derivatives |
| US7683058B2 (en) * | 2005-09-09 | 2010-03-23 | H. Lundbeck A/S | Substituted pyrimidine derivatives |
| US8293911B2 (en) | 2006-08-23 | 2012-10-23 | Valeant Pharmaceuticals International | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| WO2008066900A1 (en) | 2006-11-28 | 2008-06-05 | Valeant Pharmaceuticals International | 1,4 diamino bicyclic retigabine analogues as potassium channel modulators |
-
2007
- 2007-08-22 US US11/894,877 patent/US8293911B2/en active Active
- 2007-08-22 CN CNA2007800392650A patent/CN101563085A/en active Pending
- 2007-08-22 EP EP07837199.4A patent/EP2061465B1/en active Active
- 2007-08-22 JP JP2009525618A patent/JP5419691B2/en active Active
- 2007-08-22 KR KR1020097005943A patent/KR20090079191A/en not_active Withdrawn
- 2007-08-22 BR BRPI0716715-6A patent/BRPI0716715B1/en active IP Right Grant
- 2007-08-22 SG SG2011060944A patent/SG174095A1/en unknown
- 2007-08-22 WO PCT/US2007/018571 patent/WO2008024398A2/en not_active Ceased
- 2007-08-22 MX MX2009002002A patent/MX2009002002A/en active IP Right Grant
- 2007-08-22 ES ES07837199T patent/ES2420960T3/en active Active
- 2007-08-22 DK DK07837199.4T patent/DK2061465T3/en active
- 2007-08-22 CA CA2661462A patent/CA2661462C/en active Active
- 2007-08-22 PT PT78371994T patent/PT2061465E/en unknown
- 2007-08-22 PL PL07837199T patent/PL2061465T3/en unknown
- 2007-08-22 RU RU2009110172/15A patent/RU2009110172A/en not_active Application Discontinuation
- 2007-08-22 NZ NZ575652A patent/NZ575652A/en unknown
- 2007-08-22 AU AU2007288253A patent/AU2007288253B2/en active Active
- 2007-08-23 AR ARP070103756A patent/AR062508A1/en unknown
- 2007-08-23 CL CL200702455A patent/CL2007002455A1/en unknown
- 2007-08-23 TW TW096131287A patent/TW200827339A/en unknown
-
2009
- 2009-02-22 IL IL197161A patent/IL197161A/en active IP Right Grant
- 2009-03-20 ZA ZA2009/01977A patent/ZA200901977B/en unknown
- 2009-03-20 NO NO20091183A patent/NO343575B1/en unknown
Non-Patent Citations (2)
| Title |
|---|
| None |
| See also references of EP2061465A4 |
Cited By (78)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7799832B2 (en) | 2003-10-23 | 2010-09-21 | Valeant Pharmaceuticals North America | Combinations of retigabine and sodium channel inhibitors or sodium channel-influencing active compounds for treating pains |
| US9085531B2 (en) | 2004-07-15 | 2015-07-21 | Albany Molecular Research, Inc. | Aryl- and heteroaryl-substituted tetrahydroisoquinolines and use thereof to block reuptake of norepinephrine, dopamine, and serotonin |
| US9499531B2 (en) | 2004-07-15 | 2016-11-22 | Albany Molecular Research, Inc. | Aryl- and heteroaryl-substituted tetrahydroisoquinolines and use thereof to block reuptake of norepinephrine, dopamine, and serotonin |
| US8741901B2 (en) | 2004-07-15 | 2014-06-03 | Albany Molecular Research, Inc. | Aryl- and heteroaryl-substituted tetrahydroisoquinolines and use thereof to block reuptake of norepinephrine, dopamine, and serotonin |
| US7960436B2 (en) | 2006-06-05 | 2011-06-14 | Valeant Pharmaceuticals International | Substituted arylamino-1,2,3,4-tetrahydro naphthalenes and-2,3-dihydro-1H-indenes as potassium channel modulators |
| US8338487B2 (en) | 2006-06-05 | 2012-12-25 | Valeant Pharmaceuticals International, Inc. | Substituted arylamino-1,2,3,4-tetrahydro naphthalenes and -2,3-dihydro-1H-indenes as potassium channel modulators |
| US8293911B2 (en) | 2006-08-23 | 2012-10-23 | Valeant Pharmaceuticals International | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| US8993593B2 (en) | 2006-08-23 | 2015-03-31 | Valeant Pharmaceuticals International | N-(4-(6-fluoro-3,4-dihydroisoquinolin-2(1H)-yl)-2,6-dimethylphenyl)-3,3-dimethylbutanamide as potassium channel modulators |
| US8030518B2 (en) | 2006-11-28 | 2011-10-04 | Valeant Pharmaceuticals International | 1,4 diamino bicyclic retigabine analogues as potassium channel modulators |
| JP2010530002A (en) * | 2007-06-13 | 2010-09-02 | バレアント ファーマシューティカルズ インターナショナル | Derivatives of 4- (N-azacycloalkyl) anilide as potassium channel modulators |
| US8367684B2 (en) | 2007-06-13 | 2013-02-05 | Valeant Pharmaceuticals International | Derivatives of 4-(N-azacycloalkyl) anilides as potassium channel modulators |
| WO2008157404A3 (en) * | 2007-06-13 | 2009-02-19 | Valeant Pharmaceuticals Int | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
| WO2009018466A1 (en) * | 2007-08-01 | 2009-02-05 | Valeant Pharmaceuticals International, Inc. | Naphthyridine derivatives as potassium channel modulators |
| EP2311836A1 (en) * | 2007-08-01 | 2011-04-20 | Valeant Pharmaceuticals International, Inc. | Process for preparing naphthyridine derivatives. |
| US8211918B2 (en) | 2007-08-13 | 2012-07-03 | Valeant Pharmaceuticals International | Derivatives of 5-amino-4,6-disubstituted indole and 5-amino-4,6-disubstituted indoline as potassium channel modulators |
| US7786146B2 (en) | 2007-08-13 | 2010-08-31 | Valeant Pharmaceuticals International | Derivatives of 5-amino-4,6-disubstituted indole and 5-amino-4,6-disubstituted indoline as potassium channel modulators |
| US9498476B2 (en) | 2008-06-04 | 2016-11-22 | Albany Molecular Research, Inc. | Crystalline form of 6-[(4S)-2-methyl-4-(2-naphthyl)-1,2,3,4-tetrahydroisoquinolin-7-yl]pyridazin-3-amine |
| WO2010130260A3 (en) * | 2009-05-11 | 2011-06-16 | H. Lundbeck A/S | Stable forms of n-(2,6-dimethyl-4-morpholin-4-yl-phenyl)-3,3-dimethyl-butyramide |
| US8802696B2 (en) | 2009-05-12 | 2014-08-12 | Albany Molecular Research, Inc. | 7-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydroisoqu inoli and use thereof |
| US9173879B2 (en) | 2009-05-12 | 2015-11-03 | Bristol-Myers Squibb Company | Crystalline forms of (S)-7-([1,2,4]triazolo[1,5-a ]pyridin-6-yl)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydroisoquinoline and use thereof |
| US8815894B2 (en) | 2009-05-12 | 2014-08-26 | Bristol-Myers Squibb Company | Crystalline forms of (S)-7-([1,2,4]triazolo[1,5-a]pyridin-6-yl)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydroisoquinoline and use thereof |
| US9604960B2 (en) | 2009-05-12 | 2017-03-28 | Albany Molecular Research, Inc. | Aryl, heteroaryl, and heterocycle substituted tetrahydroisoquinolines and use thereof |
| US9034899B2 (en) | 2009-05-12 | 2015-05-19 | Albany Molecular Research, Inc. | Aryl, heteroaryl, and heterocycle substituted tetrahydroisoquinolines and use thereof |
| WO2011094186A1 (en) * | 2010-02-01 | 2011-08-04 | Valeant Pharmaceuticals International | Derivatives of 4-(n-azacycloalkyl) aniilides as potassium channel modulators |
| KR101841989B1 (en) * | 2010-02-01 | 2018-03-26 | 제논 파마슈티칼스 인크. | Derivatives of 4-(N-Azacycloalkyl) Aniilides as Potassium Channel Modulators |
| US8883812B2 (en) | 2010-07-08 | 2014-11-11 | Pfizer Inc. | Piperidinyl pyrimidine amides as Kv7 potassium channel openers |
| WO2012004698A1 (en) * | 2010-07-08 | 2012-01-12 | Pfizer Inc. | Piperidinyl pyrimidine amides as kv7 potassium channel openers |
| AU2011275393B2 (en) * | 2010-07-08 | 2014-04-10 | Pfizer Inc. | Piperidinyl pyrimidine amides as Kv7 potassium channel openers |
| WO2013055984A1 (en) | 2011-10-14 | 2013-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| EP2899183A1 (en) | 2011-10-14 | 2015-07-29 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| WO2013056060A1 (en) | 2011-10-14 | 2013-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| WO2013056034A1 (en) | 2011-10-14 | 2013-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| EP3309148A1 (en) | 2011-10-14 | 2018-04-18 | Bristol-Myers Squibb Company | Substituted tetrahydroisoquinoline compounds as factor xia inhibitors |
| WO2014160668A1 (en) | 2013-03-25 | 2014-10-02 | Bristol-Myers Squibb Company | Tetrahydroisoquinolines containing substituted azoles as factor xia inhibitors |
| EP3468947A4 (en) * | 2016-06-10 | 2020-01-08 | SciFluor Life Sciences, Inc. | FLUORINATED 2-AMINO-4- (SUBSTITUTED AMINO) PHENYLCARBAMATE DERIVATIVES |
| US11858900B2 (en) | 2016-06-10 | 2024-01-02 | Ocuterra Therapeutics, Inc. | Fluorinated 2-amino-4-(substituted amino)phenyl carbamate derivatives |
| AU2017278093B8 (en) * | 2016-06-10 | 2021-12-23 | Ocuterra Therapeutics, Inc. | Fluorinated 2-amino-4-(substituted amino)phenyl carbamate derivatives |
| AU2017278093B2 (en) * | 2016-06-10 | 2021-12-16 | Ocuterra Therapeutics, Inc. | Fluorinated 2-amino-4-(substituted amino)phenyl carbamate derivatives |
| WO2019118657A1 (en) * | 2017-12-13 | 2019-06-20 | Scifluor Life Sciences, Inc. | Fluorinated 4-(substituted amino)phenyl carbamate derivatives |
| EP3724163A4 (en) * | 2017-12-13 | 2021-07-28 | SciFluor Life Sciences, Inc. | FLUORINE DERIVATIVES 4- (AMINO SUBSTITUTE) PHENYL CARBAMATE |
| US10676437B2 (en) | 2017-12-13 | 2020-06-09 | Scifluor Life Sciences, Inc. | Fluorinated 4-(substituted amino)phenyl carbamate derivatives |
| EP4279133A3 (en) * | 2018-05-11 | 2024-02-21 | Xenon Pharmaceuticals Inc. | Methods for enhancing the bioavailability and exposure of a voltage-gated potassium channel opener |
| IL278577B1 (en) * | 2018-05-11 | 2025-03-01 | Xenon Pharmaceuticals Inc | Methods for enhancing the bioavailability and exposure of a voltage-gated potassium channel opener |
| US11135214B2 (en) | 2018-05-11 | 2021-10-05 | Xenon Pharmaceuticals Inc. | Methods for enhancing the bio availability and exposure of a voltage-gated potassium channel opener |
| WO2019217924A1 (en) * | 2018-05-11 | 2019-11-14 | Xenon Pharmaceuticals Inc. | Methods for enhancing the bioavailability and exposure of a voltage-gated potassium channel opener |
| AU2019265002C1 (en) * | 2018-05-11 | 2025-01-23 | Xenon Pharmaceuticals Inc. | Methods for enhancing the bioavailability and exposure of a voltage-gated potassium channel opener |
| IL278577B2 (en) * | 2018-05-11 | 2025-07-01 | Xenon Pharmaceuticals Inc | Methods for improving bioavailability and exposure of voltage-gated potassium channel openers |
| EP3798220B1 (en) * | 2018-05-22 | 2023-08-23 | Shanghai Zhimeng Biopharma, Inc. | P-phenylenediamine derivative as potassium channel regulator and preparation method and medical application thereof |
| US12157726B2 (en) | 2018-05-22 | 2024-12-03 | Shanghai Zhimeng Biopharma Inc. | P-phenylenediamine derivative as potassium channel regulator and preparation method and medical application thereof |
| US11365181B2 (en) | 2018-05-22 | 2022-06-21 | Shanghai Zhimeng Biopharma Inc. | P-phenylenediamine derivative as potassium channel regulator and preparation method and medical application thereof |
| US11993586B2 (en) | 2018-10-22 | 2024-05-28 | Novartis Ag | Crystalline forms of potassium channel modulators |
| WO2020157126A1 (en) | 2019-01-29 | 2020-08-06 | Università degli Studi di Salerno | Modulators of potassium ion channels and uses thereof |
| IL292005B2 (en) * | 2019-10-10 | 2025-05-01 | Xenon Pharmaceuticals Inc | Solid state crystalline form of n-[4-(6-fluoro-3,4-dihydro-1h-isoquinolin-2-yl)-2,6-dimethylphenyl]-3,3-dimethylbutanamide |
| WO2021072307A1 (en) * | 2019-10-10 | 2021-04-15 | Xenon Pharmaceuticals Inc. | Solid state crystalline forms of a selective potassium channel modulator |
| IL292005B1 (en) * | 2019-10-10 | 2025-01-01 | Xenon Pharmaceuticals Inc | Solid state crystalline form of n-[4-(6-fluoro-3,4-dihydro-1h-isoquinolin-2-yl)-2,6-dimethylphenyl]-3,3-dimethylbutanamide |
| EP4523687A3 (en) * | 2019-10-10 | 2025-05-21 | Xenon Pharmaceuticals Inc. | Solid state crystalline forms of a selective potassium channel modulator |
| US12319657B1 (en) | 2019-10-10 | 2025-06-03 | Xenon Pharmaceuticals Inc. | Solid state crystalline forms of a selective potassium channel modulator |
| US11091441B2 (en) | 2019-10-10 | 2021-08-17 | Xenon Pharmaceuticals Inc. | Solid state crystalline forms of a selective potassium channel modulator |
| TWI886158B (en) * | 2019-10-10 | 2025-06-11 | 加拿大商再諾製藥公司 | Solid state crystalline forms of a selective potassium channel modulator |
| WO2021092439A1 (en) * | 2019-11-08 | 2021-05-14 | Xenon Pharmaceuticals Inc. | Methods of treating depressive disorders |
| US12178811B2 (en) | 2019-11-08 | 2024-12-31 | Xenon Pharmaceuticals Inc. | Methods of treating depressive disorders |
| US12171759B1 (en) | 2021-02-09 | 2024-12-24 | Xenon Pharmaceuticals Inc. | Methods and uses for treating anhedonia |
| US11957675B2 (en) | 2021-02-09 | 2024-04-16 | Xenon Pharmaceuticals Inc. | Methods and uses for treating anhedonia |
| KR102694996B1 (en) | 2021-10-27 | 2024-08-13 | 상하이 지멍 바이오파마 아이엔씨 | Compounds used as potassium channel modulators and their preparation and applications |
| JP2023516838A (en) * | 2021-10-27 | 2023-04-21 | シャンハイ ジムン バイオファーマ,インコーポレーテッド | Compounds as potassium channel modulators and their preparation and application |
| WO2022174667A1 (en) | 2021-10-27 | 2022-08-25 | 上海挚盟医药科技有限公司 | Compound as potassium channel regulator, and preparation and use thereof |
| AU2021359042B1 (en) * | 2021-10-27 | 2023-04-13 | Shanghai Zhimeng Biopharma, Inc. | Compound as potassium channel regulator and preparation and use thereof |
| US12319656B2 (en) | 2021-10-27 | 2025-06-03 | Shanghai Zhimeng Biopharma, Inc. | Compound as potassium channel regulator and preparation and use thereof |
| KR20230060487A (en) * | 2021-10-27 | 2023-05-04 | 상하이 지멍 바이오파마 아이엔씨 | Compounds used as potassium channel modulators and their preparation and application |
| JP7294734B2 (en) | 2021-10-27 | 2023-06-20 | シャンハイ ジムン バイオファーマ,インコーポレーテッド | Compounds as potassium channel modulators and their preparation and application |
| RU2798327C1 (en) * | 2021-10-27 | 2023-06-21 | Шанхай Чжимэн Биофарма, Инк. | Compound as a potassium channels regulator, its production and application |
| WO2023134673A1 (en) | 2022-01-14 | 2023-07-20 | 中国科学院上海药物研究所 | High-selectivity kcnq4 potassium channel agonist, preparation method therefor and use thereof |
| WO2023239729A1 (en) * | 2022-06-08 | 2023-12-14 | Xenon Pharmaceuticals Inc. | Pyridinamine derivatives and their use as potassium channel modulators |
| CN118530144A (en) * | 2023-02-21 | 2024-08-23 | 中国医学科学院药物研究所 | A preparation method of pinegabine |
| WO2025038917A1 (en) | 2023-08-17 | 2025-02-20 | Xenon Pharmaceuticals Inc. | Heteroaryl amine compounds and uses thereof |
| WO2025038920A1 (en) | 2023-08-17 | 2025-02-20 | Xenon Pharmaceuticals, Inc. | Heteroaryl amine compounds and uses thereof |
| WO2025038930A1 (en) | 2023-08-17 | 2025-02-20 | Xenon Pharmaceuticals, Inc. | Azepane and pyrrolidine compounds and uses thereof |
| WO2026006669A1 (en) * | 2024-06-28 | 2026-01-02 | Xenon Pharmaceuticals Inc. | Prodrugs of n-[4-(6-fluoro-3,4-dihydro-2(1 h)-isoquinolinyl)-2,6-dimethylphe nyl]-3,3-dimethyl- butanamide |
Also Published As
| Publication number | Publication date |
|---|---|
| IL197161A (en) | 2015-07-30 |
| CN101563085A (en) | 2009-10-21 |
| EP2061465A4 (en) | 2011-03-16 |
| AU2007288253A1 (en) | 2008-02-28 |
| BRPI0716715B1 (en) | 2021-07-06 |
| PL2061465T3 (en) | 2014-03-31 |
| US8293911B2 (en) | 2012-10-23 |
| PT2061465E (en) | 2013-07-15 |
| TW200827339A (en) | 2008-07-01 |
| NO20091183L (en) | 2009-05-08 |
| NO343575B1 (en) | 2019-04-08 |
| ZA200901977B (en) | 2010-02-24 |
| CL2007002455A1 (en) | 2008-04-18 |
| CA2661462C (en) | 2015-09-29 |
| AR062508A1 (en) | 2008-11-12 |
| AU2007288253B2 (en) | 2013-05-02 |
| EP2061465B1 (en) | 2013-04-10 |
| CA2661462A1 (en) | 2008-02-28 |
| RU2009110172A (en) | 2010-09-27 |
| IL197161A0 (en) | 2009-12-24 |
| KR20090079191A (en) | 2009-07-21 |
| US20080139610A1 (en) | 2008-06-12 |
| ES2420960T3 (en) | 2013-08-28 |
| JP5419691B2 (en) | 2014-02-19 |
| JP2010501568A (en) | 2010-01-21 |
| DK2061465T3 (en) | 2013-07-08 |
| BRPI0716715A2 (en) | 2013-09-03 |
| WO2008024398A3 (en) | 2008-06-26 |
| NZ575652A (en) | 2011-08-26 |
| SG174095A1 (en) | 2011-09-29 |
| MX2009002002A (en) | 2009-07-22 |
| EP2061465A2 (en) | 2009-05-27 |
| WO2008024398B1 (en) | 2008-08-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2061465B1 (en) | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators | |
| CA2788712C (en) | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators | |
| JP2010501568A5 (en) | ||
| EP2086925B1 (en) | 1,4 diamino bicyclic retigabine analogues as potassium channel modulators | |
| US8492588B2 (en) | Benzyloxy anilide derivatives useful as potassium channel modulators | |
| AU2013211531A1 (en) | Derivatives of 4-(n-azacycloalkyl) anilides as potassium channel modulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780039265.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07837199 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2661462 Country of ref document: CA Ref document number: 2009525618 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 197161 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2009/002002 Country of ref document: MX Ref document number: 12009500357 Country of ref document: PH |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007288253 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 575652 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1085/KOLNP/2009 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2009110172 Country of ref document: RU Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: A20090426 Country of ref document: BY Ref document number: 2007837199 Country of ref document: EP Ref document number: 1020097005943 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: 2007288253 Country of ref document: AU Date of ref document: 20070822 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: PI0716715 Country of ref document: BR Kind code of ref document: A2 Effective date: 20090225 |