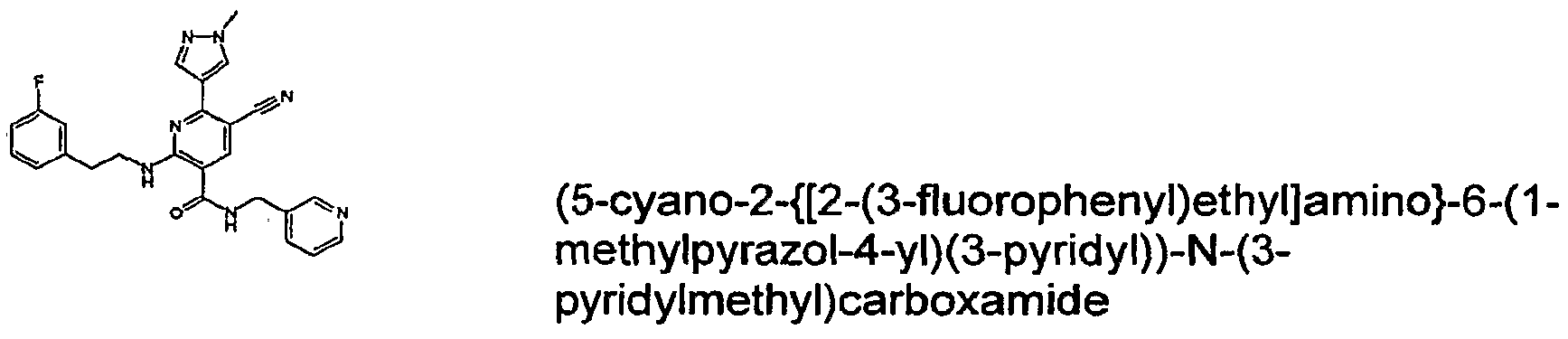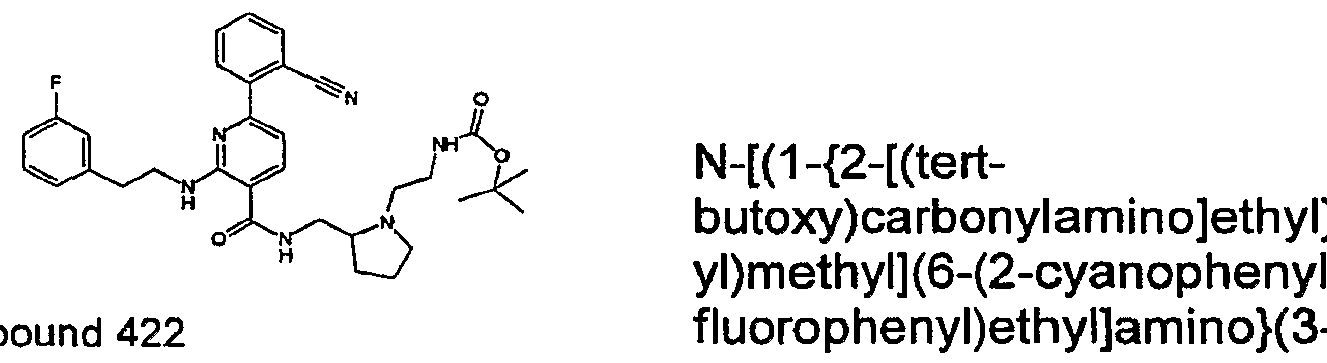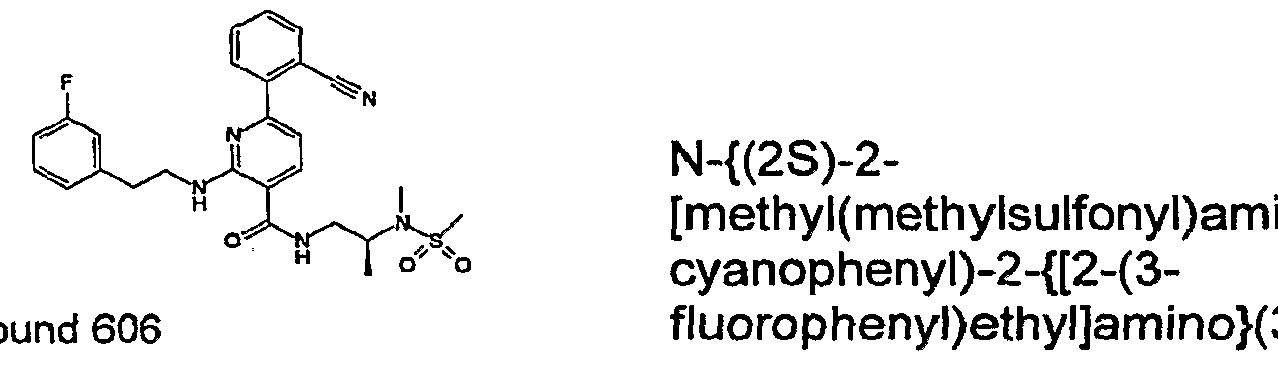WO2008016643A2 - Certain chemical entities, compositions, and methods - Google Patents
Certain chemical entities, compositions, and methods Download PDFInfo
- Publication number
- WO2008016643A2 WO2008016643A2 PCT/US2007/017186 US2007017186W WO2008016643A2 WO 2008016643 A2 WO2008016643 A2 WO 2008016643A2 US 2007017186 W US2007017186 W US 2007017186W WO 2008016643 A2 WO2008016643 A2 WO 2008016643A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- optionally substituted
- ethyl
- methyl
- amino
- pyridin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C[C@@](CC(CNCC=*)=O)[C@](*)CNC(/C(/C(/NCCc1cccc(F)c1)=N\CC1)=C\C=C1\c1ccccc1C#N)=O Chemical compound C[C@@](CC(CNCC=*)=O)[C@](*)CNC(/C(/C(/NCCc1cccc(F)c1)=N\CC1)=C\C=C1\c1ccccc1C#N)=O 0.000 description 41
- IVCBREIIGBKFSE-GORDUTHDSA-N C/C=C/c(cc1)nc(NCCc2cc(F)ccc2)c1C(NCc1cnccc1)=O Chemical compound C/C=C/c(cc1)nc(NCCc2cc(F)ccc2)c1C(NCc1cnccc1)=O IVCBREIIGBKFSE-GORDUTHDSA-N 0.000 description 1
- JRBLZKJLELMAJA-IBBHUPRXSA-N CC(C)(C/C=N/C(c(cc1)c(NCCc2cc(F)ccc2)nc1-c1ccccc1C#N)=O)NC=O Chemical compound CC(C)(C/C=N/C(c(cc1)c(NCCc2cc(F)ccc2)nc1-c1ccccc1C#N)=O)NC=O JRBLZKJLELMAJA-IBBHUPRXSA-N 0.000 description 1
- GFKXSFIHVVNJFL-VHCIKCAMSA-N CC(C1C(NC[C@@H](CC2)NC2=O)=O)C=C(c2ccccc2C#N)N=C1NCCc1cc(F)ccc1 Chemical compound CC(C1C(NC[C@@H](CC2)NC2=O)=O)C=C(c2ccccc2C#N)N=C1NCCc1cc(F)ccc1 GFKXSFIHVVNJFL-VHCIKCAMSA-N 0.000 description 1
- QGMWCDTVHAPFMS-UHFFFAOYSA-N CC1N=CC(c2cc(C(CCC3CC(C)=CCC3)=N)c(CO)cc2C)=C1 Chemical compound CC1N=CC(c2cc(C(CCC3CC(C)=CCC3)=N)c(CO)cc2C)=C1 QGMWCDTVHAPFMS-UHFFFAOYSA-N 0.000 description 1
- TUIDWMOUUIPPCA-UHFFFAOYSA-N CC1NC1C(c(cc1)cc(C(CCC(C=CC2)=CC2F)=C)c1C(NCc1cnccc1)=O)=O Chemical compound CC1NC1C(c(cc1)cc(C(CCC(C=CC2)=CC2F)=C)c1C(NCc1cnccc1)=O)=O TUIDWMOUUIPPCA-UHFFFAOYSA-N 0.000 description 1
- HLJIHIHBDFXKJL-UHFFFAOYSA-N CCCCOCCCCC(C(C(CC(CCc1cc(F)ccc1)N)=CC1)=CC=C1N1CCCCC1)=O Chemical compound CCCCOCCCCC(C(C(CC(CCc1cc(F)ccc1)N)=CC1)=CC=C1N1CCCCC1)=O HLJIHIHBDFXKJL-UHFFFAOYSA-N 0.000 description 1
- JPWOBKNDBLMXQU-RUZDIDTESA-N CCNCCCC(N1[C@@H](CNC(c2c(NCCc3cccc(F)c3)nc(-c3ccccc3C#N)c(F)c2)=O)CCC1)=O Chemical compound CCNCCCC(N1[C@@H](CNC(c2c(NCCc3cccc(F)c3)nc(-c3ccccc3C#N)c(F)c2)=O)CCC1)=O JPWOBKNDBLMXQU-RUZDIDTESA-N 0.000 description 1
- DRSKQHXSDRRWPE-KVUNNZNLSA-N CS(CC1[C@@H](C/N=C/C(c(cc2)c(CNCCc3cccc(F)c3)nc2-c2ccccc2C#N)=O)CC1)(=O)=O Chemical compound CS(CC1[C@@H](C/N=C/C(c(cc2)c(CNCCc3cccc(F)c3)nc2-c2ccccc2C#N)=O)CC1)(=O)=O DRSKQHXSDRRWPE-KVUNNZNLSA-N 0.000 description 1
- LYQGHCHCBWUXMD-KEJDIYNNSA-N C[C@@H](CNC(C1=CC=C(c2ccccc2C#N)NC1NCCc1cc(F)ccc1)=O)NC Chemical compound C[C@@H](CNC(C1=CC=C(c2ccccc2C#N)NC1NCCc1cc(F)ccc1)=O)NC LYQGHCHCBWUXMD-KEJDIYNNSA-N 0.000 description 1
- YOYPSHXARYSXSW-HLHLGKBNSA-N C[C@@](CCC1=CCCN=C1)(C1)C1C(c(cc1)c(C[C@H](C2)/C2=C/Cc2cccc(F)c2)nc1NCc1ccccn1)=O Chemical compound C[C@@](CCC1=CCCN=C1)(C1)C1C(c(cc1)c(C[C@H](C2)/C2=C/Cc2cccc(F)c2)nc1NCc1ccccn1)=O YOYPSHXARYSXSW-HLHLGKBNSA-N 0.000 description 1
- CWRATBLIJOWERP-WLTQESGOSA-N C[C@H](C(N1[C@@H](C[C@@H](CC2)[C@@H]2C(C(C(CCCCCc2cc(F)ccc2)=C(C)C2)=CC=C2c2ccccc2C#N)=O)CCC1)=O)N Chemical compound C[C@H](C(N1[C@@H](C[C@@H](CC2)[C@@H]2C(C(C(CCCCCc2cc(F)ccc2)=C(C)C2)=CC=C2c2ccccc2C#N)=O)CCC1)=O)N CWRATBLIJOWERP-WLTQESGOSA-N 0.000 description 1
- LMHDHHUBQUTCJW-SNVBAGLBSA-N C[C@H]1C=C(CCC=C)CCC1 Chemical compound C[C@H]1C=C(CCC=C)CCC1 LMHDHHUBQUTCJW-SNVBAGLBSA-N 0.000 description 1
- PJJQULXSCKPHHI-UHFFFAOYSA-N C[n]1ncc(-c(c(F)c2)nc(NCCc3cc(F)ccc3)c2C(O)=O)c1 Chemical compound C[n]1ncc(-c(c(F)c2)nc(NCCc3cc(F)ccc3)c2C(O)=O)c1 PJJQULXSCKPHHI-UHFFFAOYSA-N 0.000 description 1
- AUCABHRMIZZYPQ-IERCKHOLSA-N Cc1cc(C(NCC[C@@H](CC2)C3=C(CCCN)C3C2=O)=O)c(CNCCc2cc(F)ccc2)c(C)c1-c(cccc1)c1C#N Chemical compound Cc1cc(C(NCC[C@@H](CC2)C3=C(CCCN)C3C2=O)=O)c(CNCCc2cc(F)ccc2)c(C)c1-c(cccc1)c1C#N AUCABHRMIZZYPQ-IERCKHOLSA-N 0.000 description 1
- GUPWFAIPKGCNJQ-UHFFFAOYSA-N Cc1ccccc1C1=CC=C(C2(C3=NC3CCC(C(N(C)C)=O)N)OC2)C(NCCc2cc(F)ccc2)=NCC1 Chemical compound Cc1ccccc1C1=CC=C(C2(C3=NC3CCC(C(N(C)C)=O)N)OC2)C(NCCc2cc(F)ccc2)=NCC1 GUPWFAIPKGCNJQ-UHFFFAOYSA-N 0.000 description 1
- IIWHEPJKQOXIFA-UUSAFJCLSA-N N#Cc1ccccc1-c(cc1)nc(NCCC(C2)C=CC=C2F)c1C(NC[C@@H](CC1)NC1=O)=O Chemical compound N#Cc1ccccc1-c(cc1)nc(NCCC(C2)C=CC=C2F)c1C(NC[C@@H](CC1)NC1=O)=O IIWHEPJKQOXIFA-UUSAFJCLSA-N 0.000 description 1
- SMBXXCJFRGARKH-UHFFFAOYSA-N N#Cc1ccccc1-c(cc1)nc(NCCc2cccc(F)c2)c1C(O)=O Chemical compound N#Cc1ccccc1-c(cc1)nc(NCCc2cccc(F)c2)c1C(O)=O SMBXXCJFRGARKH-UHFFFAOYSA-N 0.000 description 1
- AFYLJZSLSWNUTF-UHFFFAOYSA-N NC(CC1C=CC=NC1)C(c1c(CCCC(C2)CC=CC2F)nc(C2CCCC2)cc1)=O Chemical compound NC(CC1C=CC=NC1)C(c1c(CCCC(C2)CC=CC2F)nc(C2CCCC2)cc1)=O AFYLJZSLSWNUTF-UHFFFAOYSA-N 0.000 description 1
- GFOAHABINHRDKL-SCSAIBSYSA-N NC[C@@H](CC1)NC1=O Chemical compound NC[C@@H](CC1)NC1=O GFOAHABINHRDKL-SCSAIBSYSA-N 0.000 description 1
- PHRGGDPCXVTHRA-UHFFFAOYSA-N O=C(c(cc1)c(NCCc2cc(F)ccc2)nc1Cl)NCc1cnccc1 Chemical compound O=C(c(cc1)c(NCCc2cc(F)ccc2)nc1Cl)NCc1cnccc1 PHRGGDPCXVTHRA-UHFFFAOYSA-N 0.000 description 1
- TWEOVINTSCVKEW-UHFFFAOYSA-N O=C(c(cc1)c(NCCc2cc(F)ccc2)nc1N1CCCC1)NCc1cnccc1 Chemical compound O=C(c(cc1)c(NCCc2cc(F)ccc2)nc1N1CCCC1)NCc1cnccc1 TWEOVINTSCVKEW-UHFFFAOYSA-N 0.000 description 1
- DBIIXNIOCCWVSH-SSDOTTSWSA-N O=C(c(ccc(Cl)n1)c1Cl)NC[C@@H]1NCCC1 Chemical compound O=C(c(ccc(Cl)n1)c1Cl)NC[C@@H]1NCCC1 DBIIXNIOCCWVSH-SSDOTTSWSA-N 0.000 description 1
- ZYATVYPHMGYEPI-UHFFFAOYSA-N OC(c(c(NCCc1cc(F)ccc1)n1)cc(F)c1Cl)=O Chemical compound OC(c(c(NCCc1cc(F)ccc1)n1)cc(F)c1Cl)=O ZYATVYPHMGYEPI-UHFFFAOYSA-N 0.000 description 1
- LTDGKGCHRNNCAC-UHFFFAOYSA-N OC(c(cc(c(Cl)n1)F)c1Cl)=O Chemical compound OC(c(cc(c(Cl)n1)F)c1Cl)=O LTDGKGCHRNNCAC-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/81—Amides; Imides
- C07D213/82—Amides; Imides in position 3
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/10—Drugs for disorders of the urinary system of the bladder
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/02—Muscle relaxants, e.g. for tetanus or cramps
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/78—Carbon atoms having three bonds to hetero atoms, with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/79—Acids; Esters
- C07D213/80—Acids; Esters in position 3
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
Definitions
- substituted heterocycles including chemical entities that modulate smooth muscle myosin and/or non-muscle myosin, pharmaceutical compositions and methods of treatment of diseases and conditions associated with smooth muscle myosin and/or non- muscle myosin.
- Myosin is present in all muscle and non-muscle cells. Of the ten distinct classes of myosin in human cells, myosin-l! is thought to be the form responsible for contraction of skeletal, cardiac, and smooth muscle. Myosin -Il is also the isoform present in non-muscle myosins, also known as cytoplasmic myosins. The non-muscle myosins are ubiquitously present in eukaryotic cells, where the smooth muscle myosins are generally present in smooth muscle cells.
- Myosin-ll is significantly different in amino acid composition and in overall structure from myosins in the other nine distinct classes.
- Myosin-ll consists of two globular head domains, called Subfragment-1 or S1, linked together by a long alpha-helical coiled-coiled tail.
- S1 contains the ATPase and actin- binding properties of the molecule. S1 has been shown to be sufficient to move actin filaments in vitro, and is therefore likely to be the motor domain of the molecule.
- myosin-ll isoforms from various tissues differ in a number of biological properties, they share the same basic molecular structure as a dimer of two heavy chains (approximately 200 kDa) which are ⁇ oncovalently associated with two pairs of light chains (approximately 20 and 17 kDa).
- the two globular amino-terminal heads are tethered together by the carboxy-terminal alpha-helical coiled-coil that forms a tail.
- the tails are believed to be involved in the assembly of myosin molecules into filaments, whereas the heads are thought to have an actin-activated Mg 2+ -ATPaSe activity.
- Each myosin head can be divided by three protease-sensitive regions into peptides of approximately 25, 50, and 20 kDa. The more amino-terminal 25 kDa - 50 kDa junction is close to the ATP binding region, whereas the actin- binding domain is near the 50 kDa - 20 kDa junction.
- S1 consists of a globular actin binding and nucleotide binding region known as the catalytic domain. This domain is attached at its carboxy-terminus to an alpha-helix that has two light chains of about 20 kDa each wrapped around it. This light-chain binding domain of S1 is known as the lever arm. Upon transitioning from the pre-stroke to the post-stroke state, the lever arm is believed to swing through an angle of about 90 degrees about a fulcrum point in the catalytic domain near the nucleotide-binding site. The "power stroke” is driven by the hydrolysis of ATP.
- the other end of the myosin molecule is an alpha-helical coiled-coiled tail involved in self assembly of myosin molecules into bipolar thick filaments. These thick filaments interdigitate between thinner actin filaments, and the two filament systems slide past one another during contraction of the muscle. This filament sliding mechanism is thought to involve conformational changes in the myosin heads causing them to walk along the thin actin filaments at the expense of ATP hydrolysis. While non-muscle myosins act in a similar manner, they are understood to slide at a slower velocity than the smooth muscle myosins. [008] The complete cDNA of the human smooth muscle myosin has been described.
- the sequence of human smooth muscle myosin is 52% identical to human cardiac myosin in the catalytic S1 region. See, for example, PCT publication No. WO 03/14323. [009] Provided is at least one chemical entity chosen from compounds of Formula X
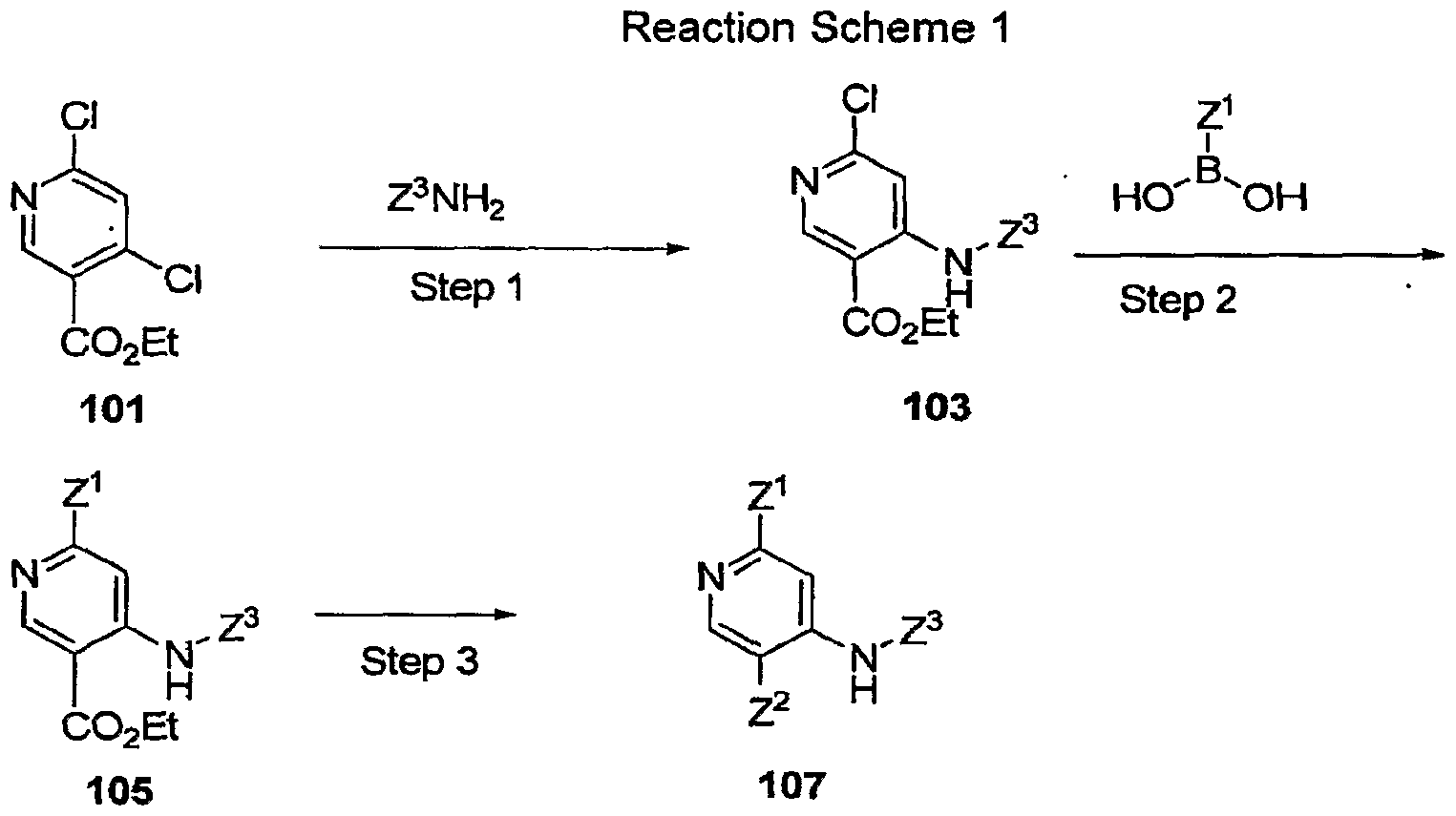
- W 1 is chosen from N and CR 6 and W 2 is chosen from N and CR 4 , provided that both W 1 and W 2 are not N;
- R 1 and R 4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodo
- Z 1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
- Z 2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
- Z 3 is chosen from hydrogen, halogen, and optionally substituted alkyl
- R 3 is chosen from hydrogen and optionally substituted alkyl
- R 6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
- composition comprising at least one chemical entity described herein, together with at least one pharmaceutically acceptable vehicle chosen from carriers, adjuvants, and excipients.
- kits for treatment of one or more diseases associated with smooth muscle myosin, or non-muscle myosin comprise administering a therapeutically effective amount of at least one chemical entity provided herein or a pharmaceutical composition comprising at least one chemical entity described herein, together with at least one pharmaceutically acceptable vehicle chosen from carriers, adjuvants, and excipients.
- Boc tert-butoxycarbonyl cyclo
- DIPEA N,N-diisopropylethylamine
- HATU 0-(7-azabenzotriazol-1 -yl)- ⁇ /, N, N', N -tetramethyluron ium hexafluorophosphate
- HBTU 0-(benzotriazol-1 -yl)- ⁇ /, ⁇ /, ⁇ /', ⁇ /'-tetramethyluronium hexafluorophosphate
- NMP N-Methyl-2-pyrrolidone
- Ph phenyl
- TBAF tetrabutylammonium fluoride
- TBS TBDMS tert-butyldimethylsilyl
- TES triethylsilyl or triethylsilane
- TMS trimethylsilyl or trimethylsilane
- a dash (“-") that is not between two letters or symbols is used to indicate a point of attachment for a substituent.
- -CONH 2 is attached through the carbon atom.
- ATPase refers to an enzyme that is capable of hydrolyzing
- ATPases include proteins comprising molecular motors such as myosins.
- Alkyl encompasses straight chain and branched chain having the indicated number of carbon atoms, usually from 1 to 20 carbon atoms, for example 1 to 8 carbon atoms, such as 1 to 6 carbon atoms.
- C 1 -C 6 alkyl encompasses both straight and branched chain alkyl of from 1 to 6 carbon atoms.
- alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, 2-pentyl, isopentyl, neopentyl, hexyl, 2-hexyl, 3- hexyl, 3-methylpentyl, and the like.
- Alkylene is another subset of alkyl, referring to the same residues as alkyl, but having two points of attachment. Alkylene groups will usually have from 2 to 20 carbon atoms, for example 2 to 8 carbon atoms, such as from 2 to 6 carbon atoms.
- C 0 alkylene indicates a covalent bond and Ci alkylene is a methylene group.
- alkyl residue having a specific number of carbons is named, all geometric combinations having that number of carbons are intended to be encompassed; thus, for example, "butyl” is meant to include n-butyl, sec-butyl, isobutyl and t-butyl; "propyl” includes n-propyl and isopropyl.
- “Lower alkyl” refers to alkyl groups having one to four carbons.
- Alkenyl refers to an unsaturated branched or straight-chain alkyl group having at least one carbon-carbon double bond derived by the removal of one hydrogen atom from a single carbon atom of a parent alkene.
- the group may be in either the cis or trans configuration about the double bond(s).
- Typical alkenyl groups include, but are not limited to, ethenyl; propenyls such as prop-1-en-1-yl, prop-1-en-2-yl, prop-2-en-1-yl (allyl), prop-2-en-2-yl, cycloprop-1-en-1-yl; cyc!oprop-2-en-1-yl; butenyls such as but-1-en-1-yl, but-1 -en-2-yl, 2-methyl-prop-1-en-1-yl, but-2-en-1-yl, but-2-e ⁇ -1-yl, but-2-en-2-yl, buta-1 ,3-dien-1-yl, buta-1,3-dien-2-yl, cyclobut-1-en-1-yl, cyclobut-1-en-3-yl, cyclobuta-1 ,3-dien-1-yl; and the like.
- an alkenyl group has from 2 to 20 carbon atoms and in
- Alkynyl refers to an unsaturated branched or straight-chain alkyl group having at least one carbon-carbon triple bond derived by the removal of one hydrogen atom from a single carbon atom of a parent alkyne.
- Typical alkynyl groups include, but are not limited to, ethynyl; propynyls such as prop-1-yn-1-yl, prop-2-yn-1-yl; butynyls such as but-1 -y ⁇ -1-yl, but-i-yn-3-yl, but-3-yn-1-yl; and the like.
- an alkynyl group has from 2 to 20 carbon atoms and in other embodiments, from 3 to 6 carbon atoms.
- Cycloalkyl indicates a non-aromatic carbocyclic ring, usually having from 3 to 7 ring carbon atoms. The ring may be saturated or have one or more carbon-carbon double bonds.
- Examples of cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, and cyclohexenyl, as well as bridged and caged saturated ring groups such as norbornane.
- alkoxy is meant an alkyl group of the indicated number of carbon atoms attached through an oxygen bridge such as, for example, methoxy, ethoxy, propoxy, isopropoxy, n- butoxy, sec-butoxy, tert-butoxy, pentyloxy, 2-pentyloxy, isopentyloxy, neopentyloxy, hexyloxy, 2- hexyloxy, 3-hexyloxy, 3-methylpentyloxy, and the like.
- Alkoxy groups will usually have from 1 to 7 carbon atoms attached through the oxygen bridge.
- “Lower alkoxy” refers to alkoxy groups having one to four carbons.
- Acyl refers to the groups H-C(O)-; (alkyl)-C(O)-; (cycloalkyl)-C(O)-; (aryl)-C(O)-; (heteroaryl)-C(O)-; and (heterocycloalkyl)-C(O)-, wherein the group is attached to the parent structure through the carbonyl functionality and wherein alkyl, cycloalkyl, aryl, heteroaryl, and heterocycloalkyl are as described herein.
- Acyl groups have the indicated number of carbon atoms, with the carbon of the keto group being included in the numbered carbon atoms.
- Tormyl refers to the group -C(O)H.
- Carboxy and/or “carboxyl” refer to the group -C(O)OH.
- a C 1 -C 6 alkoxycarbony] group is an alkoxy group having from 1 to 6 carbon atoms attached through its oxygen to a carbonyl linker.
- amino is meant the group -NH 2 .
- “Mono- and di-(alkyl)amino” encompasses secondary and tertiary alkyl amino groups, wherein the alkyl groups are as defined above and have the indicated number of carbon atoms. The point of attachment of the alkylamino group is on the nitrogen. Examples of mono- and di- alkylamino groups include ethylamino, dimethylamino, and methyl-propyl-amino.
- aminocarbonyl refers to the group -CONR b R c , where R b is chosen from H, optionally substituted C 1 -C 6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl; and R c is independently chosen from hydrogen and optionally substituted Ci-C 4 alkyl; or R b and R c taken together with the nitrogen to which they are bound, form an optionally substituted 5- to 7-membered nitrogen-containing heterocycloalkyl which optionally includes 1 or 2 additional heteroatoms selected from O, N, and S in the heterocycloalkyl ring; where each substituted group is independently substituted with one or more substituents independently selected from C 1 -C 4 alkyl, aryl, heteroaryl, aryl-C r C 4 alkyl-, heteroaryKVC alkyl-, Ci-C 4 haloalkyl
- 6-membered carbocyclic aromatic rings for example, benzene; bicyclic ring systems wherein at least one ring is carbocyclic and aromatic, for example, naphthalene, indane, and tetralin; and tricyclic ring systems wherein at least one ring is carbocyclic and aromatic, for example, fluorene.
- aryl includes 6-membered carbocyclic aromatic rings fused to a 5- to 7- membered heterocycloalkyl ring containing 1 or more heteroatoms chosen from N, O, and S.
- bicyclic ring systems wherein only one of the rings is a carbocyclic aromatic ring, the point of attachment may be at the carbocyclic aromatic ring or the heterocycloalkyl ring.
- Bivalent radicals formed from substituted benzene derivatives and having the free valences at ring atoms are named as substituted phenylene radicals.
- Bivalent radicals derived from univalent polycyclic hydrocarbon radicals whose names end in "-yl” by removal of one hydrogen atom from the carbon atom with the free valence are named by adding "-idene" to the name of the corresponding univalent radical, e.g., a naphthyl group with two points of attachment is termed naphthylidene.
- Aryl does not encompass or overlap in any way with heteroaryl, separately defined below. Hence, if one or more carbocyclic aromatic rings is fused with a heterocycloalkyl aromatic ring, the resulting ring system is heteroaryl, not aryl, as defined herein.
- aryloxy refers to the group -O-aryl.
- aralkyl refers to the group -alkyl-aryl.
- -R a -OR 6 , optionally substituted amino (including -NR c COR b , -NR 0 CO 2 R 3 , -NR c CONR b R c , - NR b C(NR°)NR b R c , -NR b C(NCN)NR b R°, and -NR 0 SO 2 R 3 ), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -COR b ), optionally substituted alkoxycarbonyl (such as -CO 2 R"), aminocarbonyl (such as -CONR b R c ), -OCOR b , -OCO 2 R 3 , -OCONR b R c , -OP(O)(O R b )OR c , sulfanyl
- R is chosen from H, optionally substituted C- ⁇ -CQ alkyl, optionally substituted aryl, and optionally substituted heteroaryl; and c
- R is independently chosen from hydrogen and optionally substituted C1-C4 alkyl; or b c
- R and R and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C 1 -C 4 alkyl, aryl, heteroaryl, aty ⁇ -C ⁇ C 4 alkyl-, heteroaryl-d-C4 alkyl-, C 1 -C 4 haloalkyl, -OC 1 -C 4 alkyl, -OC 1 -C 4 alkylphenyl, -C 1 -C 4 alkyl-OH, -OC 1 -C 4 haloalkyl, halo, -OH, -NH 2 , -C 1 -C 4 alkyl-NH 2 , -N(C 1 -C 4 8 ⁇ yI)(C 1 -C 4 alkyl), -NH(C 1 -C 4 alkyl), -NH(C 1
- Haloalkyl indicates alkyl as defined above having the specified number of carbon atoms, substituted with 1 or more halogen atoms, up to the maximum allowable number of halogen atoms.
- haloalkyl include, but are not limited to, trifluoromethyl, difluoromethyl, 2-fluoroethyl, and penta-fluoroethyl.
- Heteroaryl encompasses:
- heteroaryl includes a 5- to 7-membered heterocycloalkyl, aromatic ring fused to a 5- to 7-membered cycloalkyl or heterocycloalkyl ring.
- bicyclic heteroaryl ring systems wherein only one of the rings contains one or more heteroatoms, the point of attachment may be at either ring.
- the total number of S and O atoms in the heteroaryl group exceeds 1 , those heteroatoms are not adjacent to one another.
- the total number of S and O atoms in the heteroaryl group is not more than 2.
- the total number of S and O atoms in the aromatic heterocycle is not more than 1.
- heteroaryl groups include, but are not limited to, (as numbered from the linkage position assigned priority 1), 2-pyridyl, 3-pyridyl, 4-pyridyl, 2,3-pyrazinyl, 3,4- pyrazinyl, 2,4-pyrimidinyl, 3,5-pyrimidinyl, 2,3-pyrazolinyl, 2,4-imidazolinyl, isoxazolinyl, oxazolinyl, thiazolinyl, thiadiazolinyl, tetrazolyl, thienyl, benzothiophenyl, furanyl, benzofuranyl, benzoimidazolinyl, indolinyl, pyridazinyl, triazolyl, quinolinyl, pyrazolyl, and 5,6,7,8- tetrahydroisoquinolinyl.
- Bivalent radicals derived from univalent heteroaryl radicals whose names end in "-yl” by removal of one hydrogen atom from the atom with the free valence are named by adding "-idene" to the name of the corresponding univalent radical, e.g., a pyridyl group with two points of attachment is a pyridylidene.
- Heteroaryl does not encompass or overlap with aryl, cycloalkyl, or heterocycloalkyl, as defined herein [043]
- Substituted heteroaryl also includes ring systems substituted with one or more oxide (-O " ) substituents, such as pyridinyl N-oxides.
- heterocycloalkyl is meant a single, non-aromatic ring, usually with 3 to 7 ring atoms, containing at least 2 carbon atoms in addition to 1-3 heteroatoms independently selected from oxygen, sulfur, and nitrogen, as well as combinations comprising at least one of the foregoing heteroatoms.
- the ring may be saturated or have one or more carbon-carbon double bonds.
- Suitable heterocycloalkyl groups include, for example (as numbered from the linkage position assigned priority 1), 2-pyrrolidinyl, 2,4-imidazolidinyl, 2,3-pyrazolidinyl, 2-piperidyl, 3-piperidyl, A- piperidyl, and 2,5-piperizinyl.
- Morpholinyl groups are also contemplated, including 2- morpholinyl and 3-morpholinyl (numbered wherein the oxygen is assigned priority 1).
- Heterocycloalkyl also includes bicyclic ring systems wherein one non-aromatic ring, usually with 3 to 7 ring atoms, contains at least 2 carbon atoms in addition to 1-3 heteroatoms independently selected from oxygen, sulfur, and nitrogen, as well as combinations comprising at least one of the foregoing heteroatoms; and the other ring, usually with 3 to 7 ring atoms, optionally contains 1-3 heteratoms independently selected from oxygen, sulfur, and nitrogen and is not aromatic.
- modulation refers to a change in activity as a direct or indirect response to the presence of a chemical entity as described herein, relative to the activity of in the absence of the chemical entity.
- the change may be an increase in activity or a decrease in activity, and may be due to the direct interaction of the compound with the a target or due to the interaction of the compound with one or more other factors that in turn affect the target's activity.
- the presence of the chemical entity may, for example, increase or decrease the target activity by directly binding to the target, by causing (directly or indirectly) another factor to increase or decrease the target activity, or by (directly or indirectly) increasing or decreasing the amount of target present in the cell or organism.
- sulfanyl includes the groups: -S-(optionally substituted (Ci-C ⁇ )alkyl), -S-(optionally substituted aryl), -S-(optionally substituted heteroaryl), and -S-(optionally substituted heterocycloalkyl).
- sulfanyl includes the group Ci-C 6 alkylsulfanyl.
- sulfinyl includes the groups: -S(O)-(optionally substituted (Ci-C ⁇ )alkyl), -S(0)-optionally substituted aryl), -S(O)-optionally substituted heteroaryl), -S(O)-(optionally substituted heterocycloalkyl); and -S(0)-(optionally substituted amino).
- sulfonyl includes the groups: -S(O 2 )-(optionally substituted (CrC e )alkyl), -S( ⁇ 2 )-optionally substituted aryl), -S( ⁇ 2 )-optionally substituted heteroaryl), -S(O 2 )-(optionally substituted heterocycloalkyl) ,-S(O 2 )-(optio ⁇ ally substituted alkoxy), -S(O 2 )-optionally substituted aryloxy), -S(0 2 )-optionally substituted heteroaryloxy), -S(0 2 )-(optionally substituted heterocyclyloxy); and -S(0 2 )-(optionally substituted amino).
- substituted means that any one or more hydrogens on the designated atom or group is replaced with a selection from the indicated group, provided that the designated atom's normal valence is not exceeded.
- substituents and/or variables are permissible only if such combinations result in stable compounds or useful synthetic intermediates.
- a stable compound or stable structure is meant to imply a compound that is sufficiently robust to survive isolation from a reaction mixture, and subsequent formulation as an agent having at least practical utility.
- substituents are named into the core structure. For example, it is to be understood that when (cycloalkyl)alkyl is listed as a possible substituent, the point of attachment of this substituent to the core structure is in the alkyl portion.
- substituted alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl refer respectively to alkyl, cycloalkyl, aryl, heterocycloalkyi, and heteroaryl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
- -R a -OR
- optionally substituted amino including -NR c COR b , -NR 0 CO 2 R 3 , -NR c CONR b R°, - NR b C(NR c )NR b R c , -NR b C(NCN)NR b R c , and -NR 0 SO 2 R 3 ), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -COR b ), optionally substituted alkoxycarbonyl (such as -CO 2 R b ), aminocarbonyl (such as -CONR b R°), -OCOR b , -OCO 2 R 3 , -OCONR b R c , -OP(O)(OR b )OR°, sulfanyl (such as
- R c is independently chosen from hydrogen and optionally substituted Ci-C 4 alkyl; or
- R b and R c and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C 1 -C 4 alkyl, aryl, heteroaryl, aryl-CrC 4 alkyl-, heteroaryl-Ci-C 4 alkyl-, C 1 -C 4 haloalkyl, -OC 1 -C 4 alkyl,
- substituted acyl refers to the groups (substituted alkyl)-C(O)-; (substituted cycloalkyl)-C(O)-; (substituted aryl)-C(O)-; (substituted heteroaryl)-C(O)-; and (substituted heterocycloalkyl)-C(O)-, wherein the group is attached to the parent structure through the carbonyl functionality and wherein substituted alkyl, cycloalkyl, aryl, heteroaryl, and heterocycloalkyl, refer respectively to alkyl, cycloalkyl, aryl, heteroaryl, and heterocycloalkyl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
- alkoxycarbonyl such as -CO 2 R b
- aminocarbonyl such as
- R a is chosen from optionally substituted Ci-Ce alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl
- R b is chosen from H, optionally substituted CrC 6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl;
- R c is independently chosen from hydrogen and optionally substituted Ci-C 4 alkyl; or R b and R c , and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from Ci-C 4 alkyl, aryl, heteroaryl, aryl-Ci-C 4 alkyl-, heteroaryl-C 1 -C 4 alkyl-, Ci-C 4 haloalkyl, -OC 1 -C 4 alkyl, -OC 1 -C 4 alkylphenyl, -C 1 -C 4 alkyl-OH, -OC 1 -C 4 haloalkyl, halo, -OH, -NH 2 , -C 1 -C 4 alkyl-NH 2 , -N(C 1 -C 4 alkyl)(d-C 4
- substituted alkoxy refers to alkoxy wherein the alkyl constituent is substituted (i.e., - ⁇ -(substituted alkyl)) wherein “substituted alkyl” refers to alkyl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
- -R a , -OR b optionally substituted amino (including -NR c COR b , -NR 0 CO 2 R 8 , -NR c CONR b R c , - NR b C(NR c )NR b R c , -NR b C(NCN)NR b R°, and -NR 0 SO 2 R 3 ), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -COR b ), optionally substituted alkoxycarbonyl (such as -CO 2 R b ), aminocarbonyl (such as -CONR b R°), -OCOR", -OCO 2 R 3 , -OCONR b R°, -OP(O)(OR b )OR c , sulfanyl (such as
- -C 4 alkyl- C 1 -C 4 haloalkyl, -OC 1 -C 4 alkyl, -OC 1 -C 4 alkylphenyl, -C 1 -C 4 alkyl-OH, -OC 1 -C 4 haloalkyl, halo, -OH, -NH 2 , -C 1 -C 4 alkyl-NH 2 , -N(C 1 -C 4 alkyl)(C r C 4 alkyl), -NH(C 1 -C 4 alkyl),
- a substituted alkoxy group is "polyalkoxy" or -O-(optionally substituted alkylene)-(optionally substituted alkoxy), and includes groups such as -OCH 2 CH 2 OCH 3 , and residues of glycol ethers such as polyethyleneglycol, and -0(CH 2 CH 2 O) x CH 3 , where x is an integer of 2-20, such as 2-10, and for example, 2-5.
- Another substituted alkoxy group is hydroxyalkoxy or -OCH 2 (CH 2 ) y OH, where y is an integer of 1-10, such as 1-4.
- substituted alkoxycarbonyl refers to the group (substituted alkyl)-O-C(O)- wherein the group is attached to the parent structure through the carbonyl functionality and wherein substituted refers to alkyl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from: -R a , -OR b , optionally substituted amino (including -NR c COR b , -NR 0 CO 2 R 3 , -NR c CONR b R c , - NR b C(NR°)NR b R c , -NR b C(NCN)NR b R c , and -NR 0 SO 2 R 3 ), halo, cyano, nitro.
- oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -COR b ), optionally substituted alkoxycarbonyl (such as -CO 2 R b ), aminocarbonyl (such as -CONR b R c ), -OCOR b , -OCO 2 R 3 , -OCONR b R°, -OP(O)(OR b )OR c , sulfanyl (such as SR b ), sulfinyl (such as -SOR a ), and sulfonyl (such as -SO 2 R 3 and -SO 2 NR b R°), where R a is chosen from optionally substituted Ci-C 6 alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl; R b
- R c is independently chosen from hydrogen and optionally substituted C 1 -C 4 alkyl; or R b and R c , and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C 1 -C 4 alkyl, aryl, heteroaryl, 817!-C 1 -C 4 alkyl-, heteroaryl-Crd alkyl-, C 1 -C 4 haloalkyl, -OC 1 -C 4 alkyl, -OC 1 -C 4 alkylphenyl, -C 1 -C 4 alkyl-OH, -OC 1 -C 4 haloalkyl, halo, -OH, -NH 2 , -C 1 -C 4 alkyl-NHz, -N(C 1 -C 4 8 ⁇ yI)(
- substituted amino refers to the group -NHR d or -NR d R e wherein R d is chosen from: hydroxy, optionally substituted alkoxy, optionally substituted alkyl, optionally substituted cycloalkyl, optionally substituted acyi, optionally substituted carbamimidoyl, aminocarbonyl, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted alkoxycarbonyl, sulfinyl and sulfonyl, and wherein R ⁇ is chosen from: optionally substituted alkyl, optionally substituted cycloalkyl, optionally substituted aryl, optionally substituted heteroaryl, and optionally substituted heterocycloalkyl, and wherein substituted alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl refer respectively to alkyl, cycloalkyl, aryl, heterocycloalkyl, and
- R a is chosen from optionally substituted C 1 -C 6 alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl;
- R b is chosen from H, optionally substituted CrC 6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl;
- R c is independently chosen from hydrogen and optionally substituted C 1 -C 4 alkyl; or
- R b and R c and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C 1 -C 4 alkyl, aryl, heteroaryl, 8FyI-C 1 -C 4 alkyl-, heteroaryl-CrCj alkyl-, C 1 -C 4 haloalkyl, -OC 1 -C 4 alkyl, -OC 1 -C 4 alkylphenyl, -C 1 -C 4 alkyl-OH, -OC 1 -C 4 haloalkyl, halo, -OH, -NH 2 , -C 1 -C 4 alkyl-NH 2 , -N(C 1 -C 4 alkyl)(Ci-C 4 alkyl), -NH(C 1 -C 4 alkyl),
- substituted amino also refers to N-oxides of the groups -NHR d , and NR d R d each as described above. N-oxides can be prepared by treatment of the corresponding amino group with, for example, hydrogen peroxide or m-chloroperoxybenzoic acid. The person skilled in the art is familiar with reaction conditions for carrying out the N-oxidation.
- Compounds of Formula X include, but are not limited to, optical isomers of compounds of Formula X, racemates, and other mixtures thereof. In those situations, the single enantiomers or diastereomers, i.e., optically active forms, can be obtained by asymmetric synthesis or by resolution of the racemates.
- Racemates can be accomplished, for example, by conventional methods such as crystallization in the presence of a resolving agent, or chromatography, using, for example a chiral high-pressure liquid chromatography (HPLC) column.
- compounds of Formula X include Z- and E- forms (or cis- and trans- forms) of compounds with carbon-carbon double bonds. Where compounds of Formula X exists in various tautomeric forms, chemical entities of the present invention include all tautomeric forms of the compound.
- Chemical entities of the present invention include, but are not limited to compounds of Formula X and all pharmaceutically acceptable forms thereof.
- Pharmaceutically acceptable forms of the chemical entities recited herein include pharmaceutically acceptable salts, solvates, crystal forms (including polymorphs and clathrates), chelates, non-covalent complexes, prodrugs, and mixtures thereof.
- the chemical entities described herein are in the form of pharmaceutically acceptable salts.
- the terms "chemical entity” and “chemical entities” also encompass pharmaceutically acceptable salts, solvates, chelates, non-covalent complexes, prodrugs, and mixtures.
- “Pharmaceutically acceptable salts” include, but are not limited to salts with inorganic acids, such as hydrochloride, phosphate, diphosphate, hydrobromide, sulfate, sulfinate, nitrate, and like salts; as well as salts with an organic acid, such as malate, maleate, fumarate, tartrate, succinate, citrate, lactate, methanesulfonate, p-toluenesulfonate, 2-hydroxyethylsulfonate, benzoate, salicylate, stearate, and alkanoate such as acetate, HOOC-(CH 2 ) n -COOH where n is 0-4, and like salts.
- pharmaceutically acceptable cations include, but are not limited to sodium, potassium, calcium, aluminum, lithium, and ammonium.
- the free base can be obtained by basifying a solution of the acid salt.
- an addition salt particularly a pharmaceutically acceptable addition salt, may be produced by dissolving the free base in a suitable organic solvent and treating the solution with an acid, in accordance with conventional procedures for preparing acid addition salts from base compounds.
- Those skilled in the art will recognize various synthetic methodologies that may be used to prepare non-toxic pharmaceutically acceptable addition salts.
- prodrugs also fall within the scope of chemical entities, for example ester or amide derivatives of the compounds of Formula X.
- prodrugs includes any chemical entities that become compounds of Formula X when administered to a patient, e.g., upon metabolic processing of the prodrug.
- examples of prodrugs include, but are not limited to, acetate, formate, phosphate, and benzoate and like derivatives of functional groups (such as alcohol or amine groups) in the compounds of Formula X.
- solvate refers to the chemical entity formed by the interaction of a solvent and a compound. Suitable solvates are pharmaceutically acceptable solvates, such as hydrates, including monohydrates and hemi-hydrates.
- chelate refers to the chemical entity formed by the coordination of a compound to a metal ion at two (or more) points.
- non-covalent complex refers to the chemical entity formed by the interaction of a compound and another molecule wherein a covalent bond is not formed between the compound and the molecule.
- complexation can occur through van der Waals interactions, hydrogen bonding, and electrostatic interactions (also called ionic bonding).
- active agent is used to indicate a chemical entity which has biological activity.
- an “active agent” is a compound having pharmaceutical utility.
- an active agent may be an anti-cancer therapeutic.
- a chemical entity described herein means an amount effective, when administered to a human or non-human patient, to provide a therapeutic benefit such as amelioration of symptoms, slowing of disease progression, or prevention of disease.
- Treatment means any treatment of a disease in a patient, including: a) preventing the disease, that is, causing the clinical symptoms of the disease not to develop; b) inhibiting the disease; c) slowing or arresting the development of clinical symptoms; and/or d) relieving the disease, that is, causing the regression of clinical symptoms.
- Patient refers to an animal, such as a mammal, that has been or will be the object of treatment, observation or experiment. The methods described herein can be useful in both human therapy and veterinary applications.
- the patient is a mammal; in some embodiments the patient is human; and in some embodiments the patient is chosen from cats and dogs.
- Provided is at least one chemical entity chosen from compounds of Formula X
- W 1 is chosen from N and CR 6 and W 2 is chosen from N and CR 4 , provided that both W 1 and W 2 are not N;
- R 1 and R 4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
- Z 1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
- Z 2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
- Z 3 is chosen from hydrogen, halogen, and optionally substituted alkyl
- R 3 is chosen from hydrogen and optionally substituted alkyl; and R 6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
- W 1 is N and W 2 is CR 4 .
- W 1 is CR ⁇ and W 2 is N.
- R 1 and R 4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
- Z 1 is chosen from optionally substituted aryl and optionally substituted heteroaryl
- Z 2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
- Z 3 is chosen from hydrogen and optionally substituted alkyl
- R 3 is chosen from hydrogen and optionally substituted alkyl.
- Z 1 is optionally substituted aryl.
- Z 1 is optionally substituted heteroaryl.
- Z 3 is optionally substituted alkyl.
- Z 3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl.
- R 1 and R 4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
- Z 1 is optionally substituted aryl
- Z 2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, and optionally substituted aminocarbonyl;
- Z 3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl
- R 3 is chosen from hydrogen and optionally substituted alkyl.
- Z 2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
- Z 2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
- Z 2 is chosen from (2-(N- acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2-
- R 3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula II, R 3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula II, R 3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula II, R 3 is hydrogen.
- R 4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula II, R 4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula II, R 4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl. In certain embodiments of compounds of Formula II, R 4 is hydrogen.
- Z 2 is -C(O)-NR 2 R 5 wherein R 2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R 5 is chosen from hydrogen and optionally substituted alkyl.
- R 2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
- R 2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
- R 2 is chosen from (1-(2- aminoethyl)-1H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-3-yl)methyl; (1 -methyl- 1 H-pyrazol-5-yl)methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl;
- Z 3 is chosen from -(CH 2 ) r R 20 wherein r is chosen from 1 , 2, and 3 and R 20 is chosen from optionally substituted aryl and optionally substituted heteroaryl.
- compounds of Formula M 1 Z 3 is chosen from 2-(3- methylphenyl)ethyl, 2-(1 H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(2,3- difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(
- Z 3 is 2-(pyridin-2-yl)ethyl.
- Z 1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamlmidoyl, and optionally substituted sulfonyl.
- Z 1 is chosen from phenyl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, lower alkenyl, lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, lower acyl, dialkylamino, acetylamino, carbamimidoyl optionally substituted with cyano, aralkyl, and herteroaralkyl, and aminocarbonyl.
- Z 1 is chosen from phenyl, 2-aminocarbonylphenyl, 2-fluorophenyl, 2-chlorophenyl, 2-hydroxyphenyl, 2-methylphenyl, 2-methoxyphenyl, 2-cyanophenyl, 2-cyanomethylphenyl, 2-vinylphenyl, 2- formylphenyl, 3,4-difluorophenyl, 3-methylphenyl, 3-chlorophenyl, 3-fluorophenyl, 3- cyanophenyl, 3-dimethylaminophenyl, 3-methoxyphenyl, 3-methoxymethylphenyl, 3- hydroxyphenyl, 3-trifluoromethylphenyl, 4-aminocarbonylphenyl, 4-fluorophenyl, 4-chlorophenyl, 4-methylphenyl, 4-hydroxyphenyl, 4-cyanophenyl, 4-methoxyphenyl, 4-hydroxymethylphenyl, A- trifiuoromethyl
- R 1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula II, R 1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula II,
- R 1 is chosen from hydrogen and methyl.
- R 1 is hydrogen
- R 1 and R 4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
- Z 1 is optionally substituted heteroaryl
- Z 2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, and optionally substituted aminocarbonyl;
- Z 3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl
- R 3 is chosen from hydrogen and optionally substituted alkyl.
- Z 1 is chosen from heteroaryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyi, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
- Z 1 is chosen from 1 H-pyrazol-3-yl; 1 H-pyrazol-4-yl; 1 H-tetrazol-5-yl; 1-methyl-1 H-pyrazol-4-yl; 1-methyl-1 H-pyrazol-5-yl; 2- (hydroxymethyl)thiophen-3-yl; 2,2'-bipyridine; 2,3'-bipyridi ⁇ e; 2,4'-bipyridine; 2-cyanothiophen-3- yl; 2-formylthiophen-3-yl; 3-(hydroxymethyl)thiophen-2-yl; 3-cyano-1-methyl-1 H-pyrazol-4-yl; 3- formylthiophen-2-yl; 4-cyanothiophen-3-yl; 4-methylthiophen-2-yl; 4-methylthiophen-3-yl; 5-(1- hydroxyethyl)thiophen-2-yl; 5-(aminomethyl)thiophen-2-yl; 5-
- Z 2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
- Z 2 is chosen from 2- (methylamino)-2-oxoethyl, 2-amino-2-oxoethyl, 3-amino-3-oxopropyl, (2-(N- acetylaminomethyl)piperidin-1 -yl)carbonyl; (2-aminomethylpiperidin-1 -yl)carbonyl; 1 -hydroxy-2- amino-ethyl; 2-((methyIsulfonamido)methyl)piperidin-1-ylcarbonyl; 2-
- R 3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula III, R 3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula III, R 3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula III, R 3 is hydrogen.
- R 4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula III, R 4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula III, R 4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxy methyl. In certain embodiments of compounds of Formula III, R 4 is hydrogen.
- Z 2 is -C(O)NR 2 R 5 wherein R 2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R 5 is chosen from hydrogen and optionally substituted alkyl.
- R 2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
- R 2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
- R 2 is chosen from (1-(2- aminoethyl)-1H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1 -acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl;
- Z 3 is chosen from -(CH 2 ) r R 20 wherein r is chosen from 1 , 2, and 3 and R 20 is chosen from optionally substituted aryl and optionally substituted heteroaryl.
- Z 3 is chosen from 2-(3- methylphenyl)ethyl, 2- ⁇ 1H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(2,3- difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2- ⁇
- Z 3 is chosen from 3- fJuorophenethyl, 3,5-difluorophenethyl, and 2-(pyridin-2-yl)ethyl. In certain embodiments of compounds of Formula III, Z 3 is 2-(pyridin-2-yl)ethyl.
- R 1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula III, R 1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula III,
- R 1 is chosen from hydrogen and methyl.
- R 1 is hydrogen
- R 1 and R 4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
- Z 1 is chosen from optionally substituted aryl and optionally substituted heteroaryl
- Z 2 is chosen from hydrogen, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted amidino, and optionally substituted aminocarbonyl;
- Z 3 is optionally substituted alkyl
- R 3 is chosen from hydrogen and optionally substituted alkyl.
- Z 1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl, and heteroaryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alke ⁇ yl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted
- Z 1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted lower acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and amino optionally substituted with alkyl, and heteroaryl optionally substituted by one or two groups selected from optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted lower acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optional
- Z 1 is chosen from phenyl
- Z 2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
- Z 2 is chosen from carboxyl, optionally substituted pyridinylmethylcarbonyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
- Z 2 is -C(O)NR 2 R 5 wherein
- R 2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R 5 is chosen from hydrogen and optionally substituted alkyl.
- R 2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
- R 2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
- R 2 is chosen from ⁇ 1-(2- aminoethyl)-1 H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1-rnethyl-1H-pyrazol-3-yl)methyl; (1-methyl-1H-pyrazol-5-yl)methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)car
- R 2 is pyridin-3-ylmethyl.
- R 5 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula IV, R 5 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula IV, R 5 is hydrogen.
- Z 3 is chosen from optionally substituted cycloalkylalkyl, optionally substituted heterocycloalkylalkyl.
- Z 3 is chosen from -(CH ⁇ J r R 20 wherein r is chosen from 1 , 2, and 3 and R 20 is chosen from optionally substituted heterocycloalkyl and optionally substituted cycloalkyl.
- Z 3 is chosen from
- R 1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula IV, R 1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula IV 1 R 1 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula IV, R 1 is hydrogen.
- R 3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula IV, R 3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula IV, R 3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula IV 1 R 3 is hydrogen.
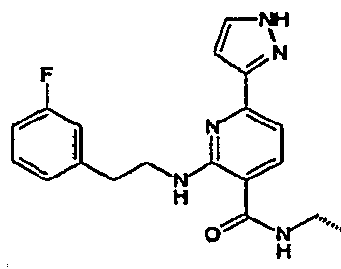
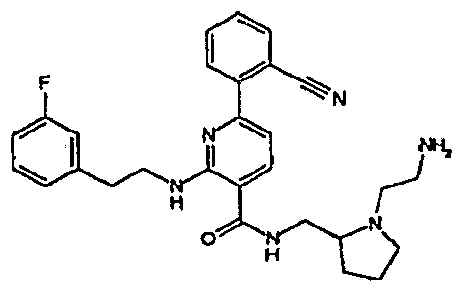
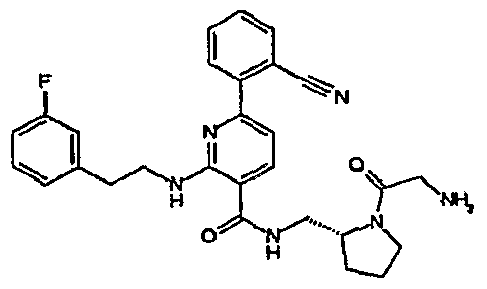
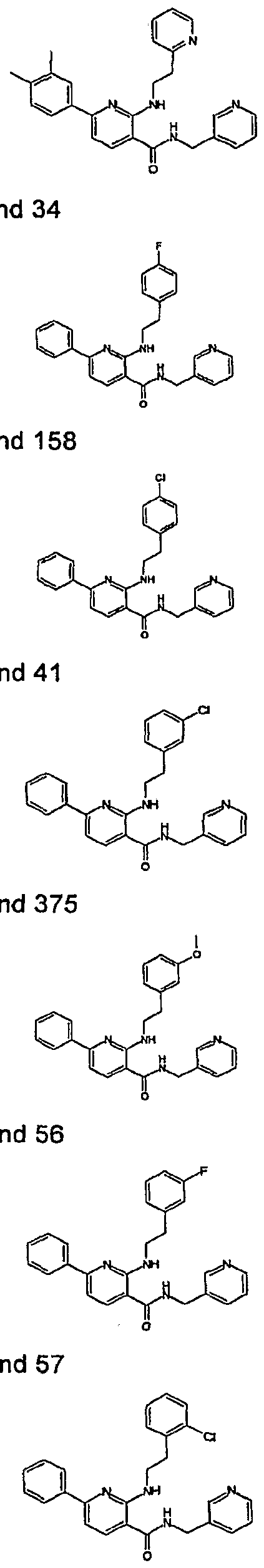
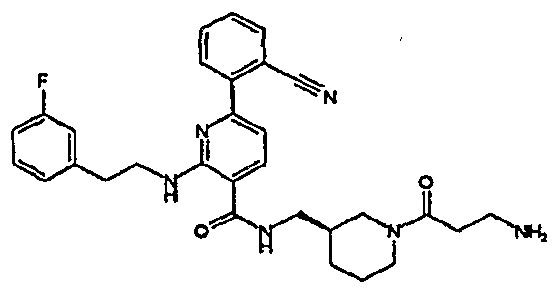
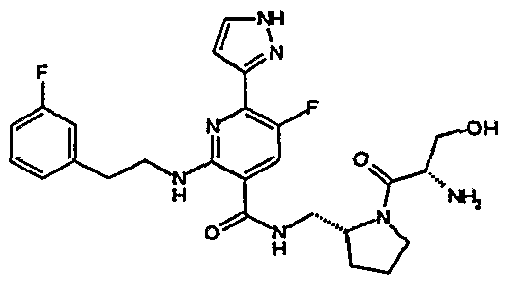
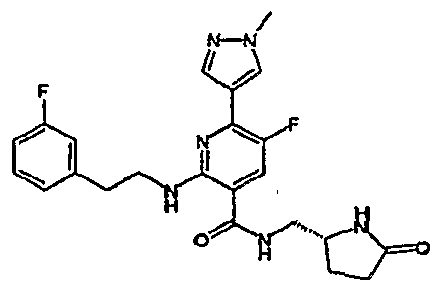
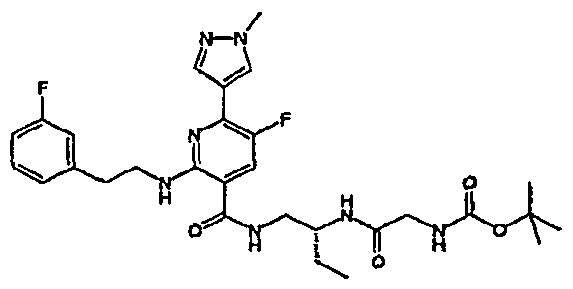
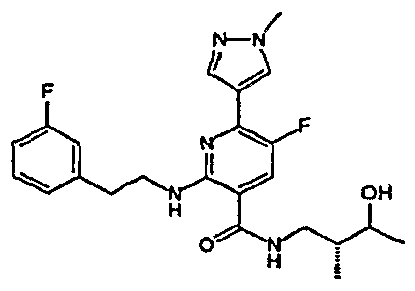
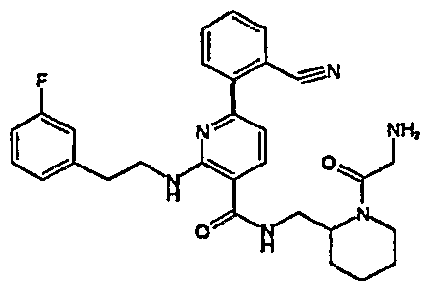
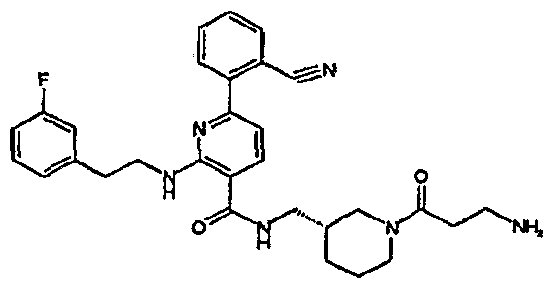
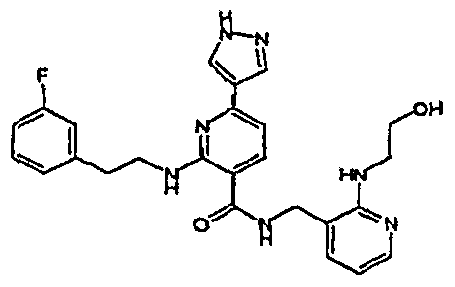
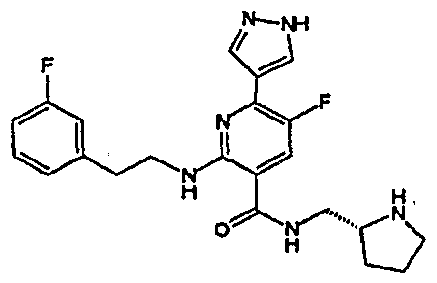
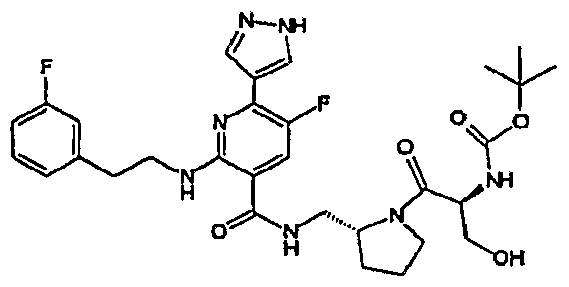
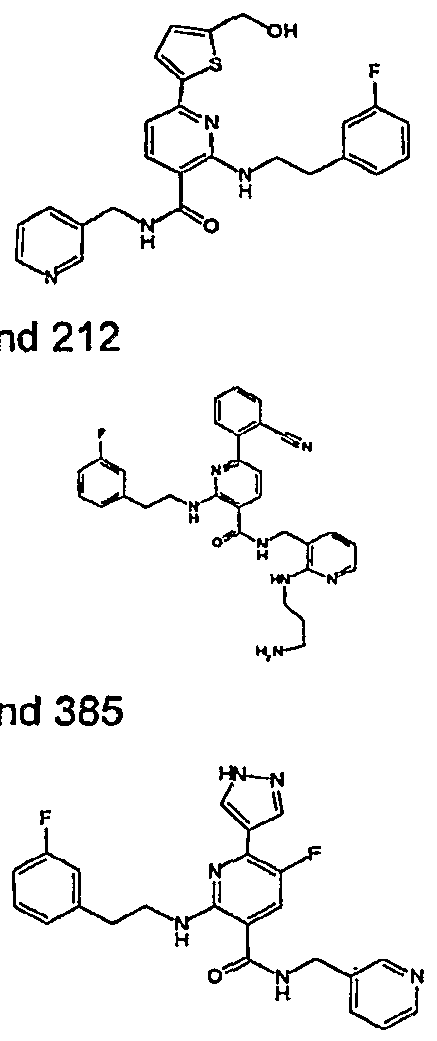
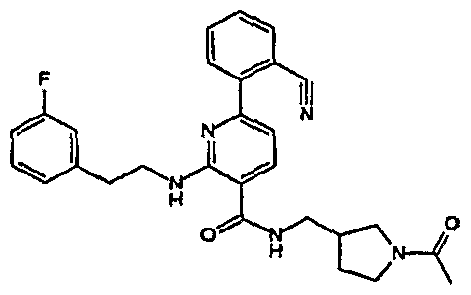
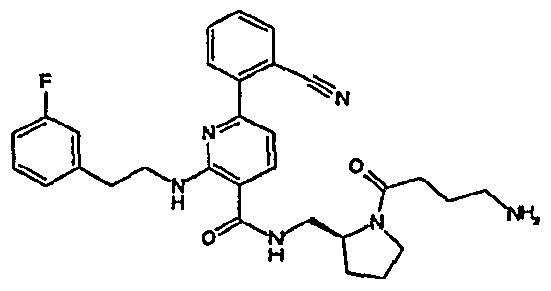
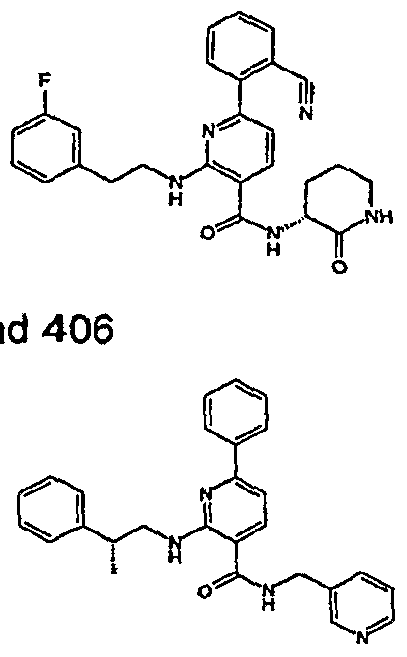
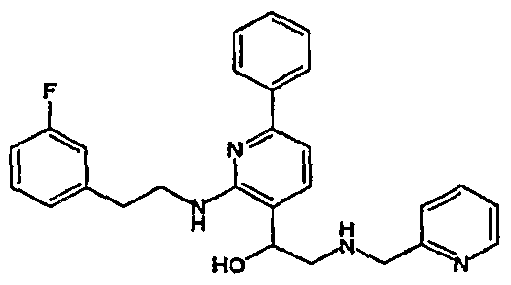
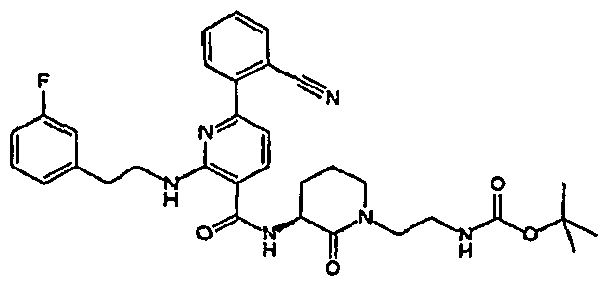
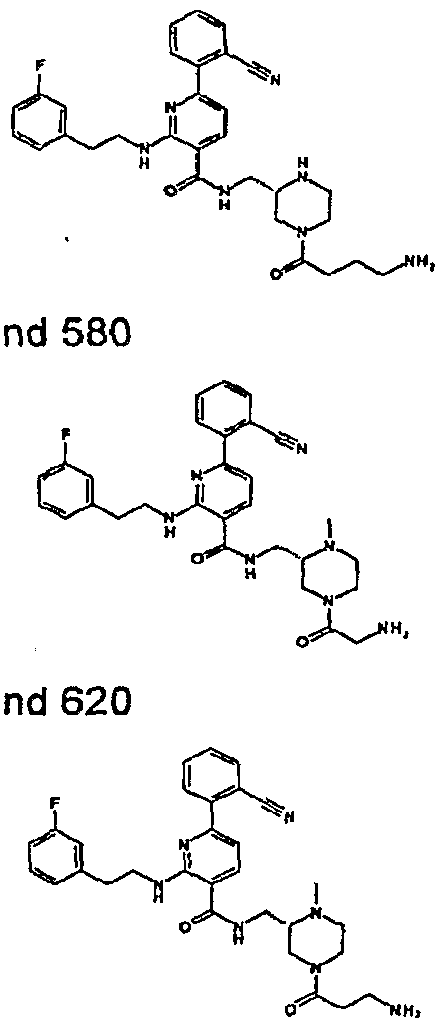
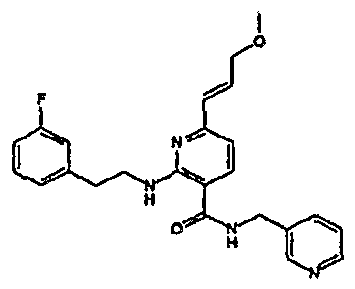
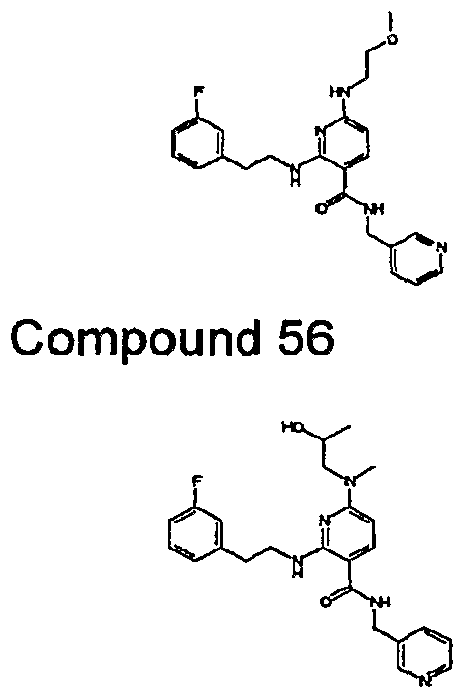
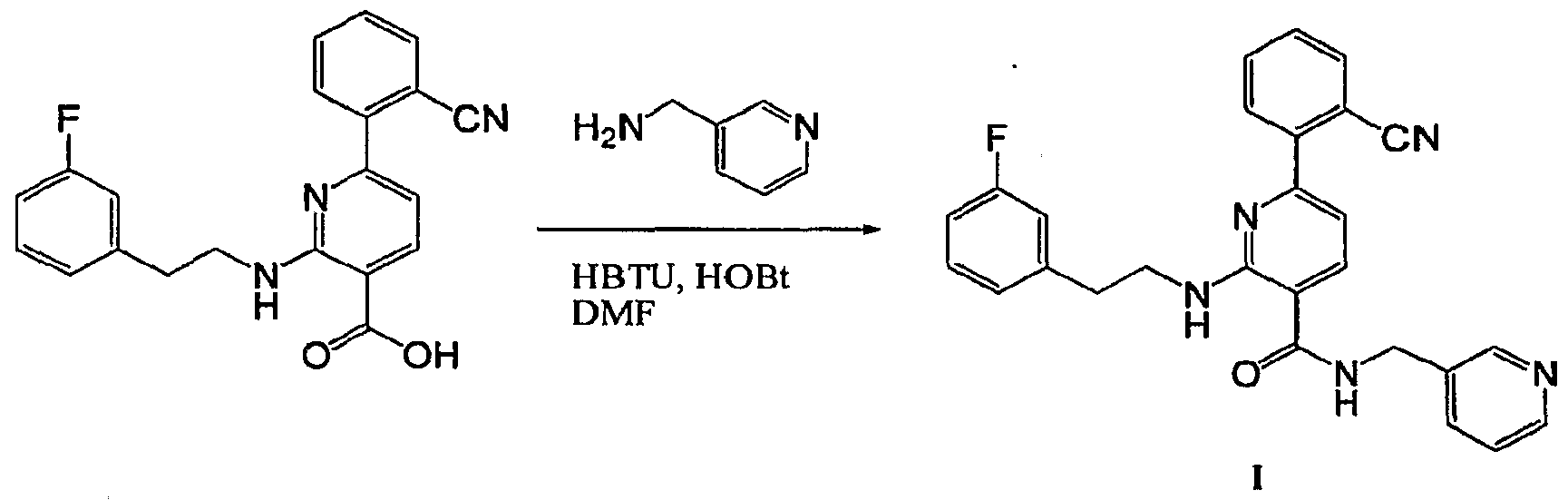
- R 4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula IV, R 4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula IV 1 R 4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl. In certain embodiments of compounds of Formula IV, R 4 is hydrogen. [0124] In certain embodiments, the compound of Formula I is chosen from
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Neurology (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Pulmonology (AREA)
- Pain & Pain Management (AREA)
- Urology & Nephrology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pyridine Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Chemical entities that modulate smooth muscle myosin and/or non-muscle myosin, and chemical entities, pharmaceutical compositions and methods of treatment of diseases and conditions associated with smooth muscle myosin and/or non-muscle myosin are described.
Description
CERTAIN CHEMICAL ENTITIES, COMPOSITIONS, AND METHODS [001] This application claims the benefit of U.S. Provisional Patent Application No. 60/834,903, filed August 1, 2006, U.S. Provisional Patent Application No. 60/835,183, filed August 1, 2006, U.S. Provisional Patent Application No. 60/835,010, filed August 1, 2006, U.S. Provisional Patent Application No. 60/834,904, filed August 1, 2006, U.S. Provisional Patent Application No. 60/834,901, filed August 1, 2006, and U.S. Provisional Patent Application No. 60/835,236, filed August 2, 2006, each of which is incorporated herein by reference for all purposes.
[002] Provided are certain substituted heterocycles, including chemical entities that modulate smooth muscle myosin and/or non-muscle myosin, pharmaceutical compositions and methods of treatment of diseases and conditions associated with smooth muscle myosin and/or non- muscle myosin.
[003] Myosin is present in all muscle and non-muscle cells. Of the ten distinct classes of myosin in human cells, myosin-l! is thought to be the form responsible for contraction of skeletal, cardiac, and smooth muscle. Myosin -Il is also the isoform present in non-muscle myosins, also known as cytoplasmic myosins. The non-muscle myosins are ubiquitously present in eukaryotic cells, where the smooth muscle myosins are generally present in smooth muscle cells.
[004] Myosin-ll is significantly different in amino acid composition and in overall structure from myosins in the other nine distinct classes. Myosin-ll consists of two globular head domains, called Subfragment-1 or S1, linked together by a long alpha-helical coiled-coiled tail. Proteolysis of myosin generates either S1 or heavy meromyosin (HMM, a two-headed form with a truncated tail), depending on the proteolysis conditions. S1 contains the ATPase and actin- binding properties of the molecule. S1 has been shown to be sufficient to move actin filaments in vitro, and is therefore likely to be the motor domain of the molecule. [005] Although myosin-ll isoforms from various tissues differ in a number of biological properties, they share the same basic molecular structure as a dimer of two heavy chains (approximately 200 kDa) which are πoncovalently associated with two pairs of light chains (approximately 20 and 17 kDa). The two globular amino-terminal heads are tethered together by the carboxy-terminal alpha-helical coiled-coil that forms a tail. The tails are believed to be involved in the assembly of myosin molecules into filaments, whereas the heads are thought to have an actin-activated Mg2+-ATPaSe activity. Each myosin head can be divided by three protease-sensitive regions into peptides of approximately 25, 50, and 20 kDa. The more
amino-terminal 25 kDa - 50 kDa junction is close to the ATP binding region, whereas the actin- binding domain is near the 50 kDa - 20 kDa junction.
[006] S1 consists of a globular actin binding and nucleotide binding region known as the catalytic domain. This domain is attached at its carboxy-terminus to an alpha-helix that has two light chains of about 20 kDa each wrapped around it. This light-chain binding domain of S1 is known as the lever arm. Upon transitioning from the pre-stroke to the post-stroke state, the lever arm is believed to swing through an angle of about 90 degrees about a fulcrum point in the catalytic domain near the nucleotide-binding site. The "power stroke" is driven by the hydrolysis of ATP.
[007] The other end of the myosin molecule is an alpha-helical coiled-coiled tail involved in self assembly of myosin molecules into bipolar thick filaments. These thick filaments interdigitate between thinner actin filaments, and the two filament systems slide past one another during contraction of the muscle. This filament sliding mechanism is thought to involve conformational changes in the myosin heads causing them to walk along the thin actin filaments at the expense of ATP hydrolysis. While non-muscle myosins act in a similar manner, they are understood to slide at a slower velocity than the smooth muscle myosins. [008] The complete cDNA of the human smooth muscle myosin has been described. The sequence of human smooth muscle myosin is 52% identical to human cardiac myosin in the catalytic S1 region. See, for example, PCT publication No. WO 03/14323. [009] Provided is at least one chemical entity chosen from compounds of Formula X
Formula X and pharmaceutically acceptable salts thereof wherein
W1 is chosen from N and CR6 and W2 is chosen from N and CR4, provided that both W1 and W2 are not N;
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from hydrogen, halogen, and optionally substituted alkyl;
R3 is chosen from hydrogen and optionally substituted alkyl; and
R6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
[010] Also provided is a pharmaceutical composition comprising at least one chemical entity described herein, together with at least one pharmaceutically acceptable vehicle chosen from carriers, adjuvants, and excipients.
[011] Also provided are methods of treatment of one or more diseases associated with smooth muscle myosin, or non-muscle myosin. The methods of treatment comprise administering a therapeutically effective amount of at least one chemical entity provided herein or a pharmaceutical composition comprising at least one chemical entity described herein, together with at least one pharmaceutically acceptable vehicle chosen from carriers, adjuvants, and excipients.
W
[012] Other aspects and embodiments will be apparent to those skilled in the art from the following detailed description.
[013] As used in the present specification, the following words and phrases are generally intended to have the meanings as set forth below, except to the extent that the context in which they are used indicates otherwise.
[014] As used herein, when any variable occurs more than one time in a chemical formula, its definition on each occurrence is independent of its definition at every other occurrence.
[015] The following abbreviations and terms have the indicated meanings throughout:
PIPES 1 ,4-piperazinediethanesulfonic acid
ATP adenosine 5'-triphosphate
DTT DL-dithiothreitol
BSA bovine serum albumin
NADH nicotinamide adenine dinucleotide
PEP phosphoenolpyruvic acid
EGTA ethylene glycol-bis(2-aminoethylether)-N,N,N',N'-tetraacetic acid
Ac acetyl
APCI atmospheric pressure chemical ionization atm = atomosphere
Boc = tert-butoxycarbonyl cyclo
CBZ carbobenzyloxy = benzyloxycarbonyl
CDI carbonyldiimidazole
DCM dichloromethane = methylene chloride = CH2CI2
DIAD diisopropyl azodicarboxylate
DIEA = DIPEA = N,N-diisopropylethylamine
DMAP 4-(dimethylamino)pyridine
DMF = N.N-dimethylformamide
DMSO = dimethyl sulfoxide
(DPPF)PdCI2 = [1 ,1 '-bis(diphenylphosphino)ferrocene]dichloropalladium(ll)
Et ethyl
EtOAc ethyl acetate
EtOH ethanol
9 - gram
GC = gas chromatograghy h or hr = hour
HATU = 0-(7-azabenzotriazol-1 -yl)-Λ/, N, N', N -tetramethyluron ium hexafluorophosphate
HBTU = 0-(benzotriazol-1 -yl)-Λ/,Λ/,Λ/',Λ/'-tetramethyluronium hexafluorophosphate
HOBT = 1 -hydroxybenzotriazole
HPLC = high pressure liquid chromatography i- = iso kg or Kg = kilogram
L or i liter
LC/MS = LCMS = liquid chromatography-mass spectrometry
LDA = lithium diisopropylamide
LRMS = low resolution mass spectrometry m/z = mass-to-charge ratio
Me = methyl
NMP = N-Methyl-2-pyrrolidone
NMR = nuclear magnetic resonance
MPLC = medium pressure liquid chromatography min = minute mL = milliliter
MW = microwave n- = normal
Ph = phenyl
(Ph3P)4Pd = tetrakis(triphenylphosphine)palladium(0)
(Ph3P)2PdCI2 = dichlorobis(triphenylphosphine)palladium(ll)
RP-HPLC = reverse phase-high pressure liquid chromatography rt or RT = room temperature
S- = sec- = secondary t- = tert- = tertiary
TBAF = tetrabutylammonium fluoride
TBS TBDMS = tert-butyldimethylsilyl
TES = triethylsilyl or triethylsilane
TMS = trimethylsilyl or trimethylsilane
TFA = trifluoroacetic acid
THF = tetrahydrofuran
TLC = thin layer chromatography
UV = ultraviolet vol = volume equivalent in mL/g or L/Kg or the limiting reagent unless otherwise specified
[016] A dash ("-") that is not between two letters or symbols is used to indicate a point of attachment for a substituent. For example, -CONH2 is attached through the carbon atom.
[017] By "optional" or "optionally" is meant that the subsequently described event or circumstance may or may not occur, and that the description includes instances where the event or circumstance occurs and instances in which it does not. For example, "optionally substituted alkyl" encompasses both "alkyl" and "substituted alkyl" as defined below. It will be understood by those skilled in the art, with respect to any group containing one or more substituents, that such groups are not intended to introduce any substitution or substitution patterns that are sterically impractical, synthetically non-feasible and/or inherently unstable.
[018] The term "ATPase," as used herein, refers to an enzyme that is capable of hydrolyzing
ATP. ATPases include proteins comprising molecular motors such as myosins.
[019] "Alkyl" encompasses straight chain and branched chain having the indicated number of carbon atoms, usually from 1 to 20 carbon atoms, for example 1 to 8 carbon atoms, such as 1 to 6 carbon atoms. For example C1-C6 alkyl encompasses both straight and branched chain alkyl of from 1 to 6 carbon atoms. Examples of alkyl groups include methyl, ethyl, propyl, isopropyl, n-butyl, sec-butyl, tert-butyl, pentyl, 2-pentyl, isopentyl, neopentyl, hexyl, 2-hexyl, 3- hexyl, 3-methylpentyl, and the like. Alkylene is another subset of alkyl, referring to the same residues as alkyl, but having two points of attachment. Alkylene groups will usually have from 2 to 20 carbon atoms, for example 2 to 8 carbon atoms, such as from 2 to 6 carbon atoms. For example, C0 alkylene indicates a covalent bond and Ci alkylene is a methylene group. When an alkyl residue having a specific number of carbons is named, all geometric combinations having that number of carbons are intended to be encompassed; thus, for example, "butyl" is meant to include n-butyl, sec-butyl, isobutyl and t-butyl; "propyl" includes n-propyl and isopropyl.
"Lower alkyl" refers to alkyl groups having one to four carbons.
[020] "Alkenyl" refers to an unsaturated branched or straight-chain alkyl group having at least one carbon-carbon double bond derived by the removal of one hydrogen atom from a single
carbon atom of a parent alkene. The group may be in either the cis or trans configuration about the double bond(s). Typical alkenyl groups include, but are not limited to, ethenyl; propenyls such as prop-1-en-1-yl, prop-1-en-2-yl, prop-2-en-1-yl (allyl), prop-2-en-2-yl, cycloprop-1-en-1-yl; cyc!oprop-2-en-1-yl; butenyls such as but-1-en-1-yl, but-1 -en-2-yl, 2-methyl-prop-1-en-1-yl, but-2-en-1-yl, but-2-eπ-1-yl, but-2-en-2-yl, buta-1 ,3-dien-1-yl, buta-1,3-dien-2-yl, cyclobut-1-en-1-yl, cyclobut-1-en-3-yl, cyclobuta-1 ,3-dien-1-yl; and the like. In certain embodiments, an alkenyl group has from 2 to 20 carbon atoms and in other embodiments, from 2 to 6 carbon atoms.
[021] "Alkynyl" refers to an unsaturated branched or straight-chain alkyl group having at least one carbon-carbon triple bond derived by the removal of one hydrogen atom from a single carbon atom of a parent alkyne. Typical alkynyl groups include, but are not limited to, ethynyl; propynyls such as prop-1-yn-1-yl, prop-2-yn-1-yl; butynyls such as but-1 -yπ-1-yl, but-i-yn-3-yl, but-3-yn-1-yl; and the like. In certain embodiments, an alkynyl group has from 2 to 20 carbon atoms and in other embodiments, from 3 to 6 carbon atoms.
[022] "Cycloalkyl" indicates a non-aromatic carbocyclic ring, usually having from 3 to 7 ring carbon atoms. The ring may be saturated or have one or more carbon-carbon double bonds. Examples of cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, and cyclohexenyl, as well as bridged and caged saturated ring groups such as norbornane.
[023] By "alkoxy" is meant an alkyl group of the indicated number of carbon atoms attached through an oxygen bridge such as, for example, methoxy, ethoxy, propoxy, isopropoxy, n- butoxy, sec-butoxy, tert-butoxy, pentyloxy, 2-pentyloxy, isopentyloxy, neopentyloxy, hexyloxy, 2- hexyloxy, 3-hexyloxy, 3-methylpentyloxy, and the like. Alkoxy groups will usually have from 1 to 7 carbon atoms attached through the oxygen bridge. "Lower alkoxy" refers to alkoxy groups having one to four carbons. [024] The term "amidino" refers to the group -C(=NH)-NH2. The term "substituted amidino" refers to the formula -C(=NR')-NR"R" in which each of the R" groups is independently chosen from hydrogen, optionally substituted alkyl, optionally substituted alkoxy, optionally substituted aminocarbonyl, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocyclyl, acyl, alkoxycarbonyl, sulfanyl, sulfinyl and sulfonyl and R1 is chosen from hydrogen, cyano, optionally substituted alkyl, optionally substituted alkoxy, optionally substituted aminocarbonyl, optionally substituted aryl, optionally substituted heteroaryl,
optionally substituted heterocyclyl, acyl, alkoxycarbonyl, sulfanyl, sulfinyl and sulfonyl, provided that at least one R1 or R" group is not hydrogen.
[025] "Mono- and di-alkylcarboxamide" encompasses a group of the formula -(C=O)NR3Rb where Ra and Rb are independently chosen from hydrogen and alkyl groups of the indicated number of carbon atoms, provided that R3 and Rb are not both hydrogen. [026] "Acyl" refers to the groups H-C(O)-; (alkyl)-C(O)-; (cycloalkyl)-C(O)-; (aryl)-C(O)-; (heteroaryl)-C(O)-; and (heterocycloalkyl)-C(O)-, wherein the group is attached to the parent structure through the carbonyl functionality and wherein alkyl, cycloalkyl, aryl, heteroaryl, and heterocycloalkyl are as described herein. Acyl groups have the indicated number of carbon atoms, with the carbon of the keto group being included in the numbered carbon atoms. For example a C2 acyl group is an acetyl group having the formula CH3(C=O)-. [027] Tormyl" refers to the group -C(O)H. [028] "Carboxy" and/or "carboxyl" refer to the group -C(O)OH.
[029] By "alkoxycarbonyl" is meant a group of the formula (alkoxy)(C=0)- attached through the carbonyl carbon wherein the alkoxy group has the indicated number of carbon atoms. Thus a C1-C6 alkoxycarbony] group is an alkoxy group having from 1 to 6 carbon atoms attached through its oxygen to a carbonyl linker. [030] By "amino" is meant the group -NH2.
[031] "Mono- and di-(alkyl)amino" encompasses secondary and tertiary alkyl amino groups, wherein the alkyl groups are as defined above and have the indicated number of carbon atoms. The point of attachment of the alkylamino group is on the nitrogen. Examples of mono- and di- alkylamino groups include ethylamino, dimethylamino, and methyl-propyl-amino. [032] The term "aminocarbonyl" refers to the group -CONRbRc, where Rb is chosen from H, optionally substituted C1-C6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl; and Rc is independently chosen from hydrogen and optionally substituted Ci-C4 alkyl; or Rband Rc taken together with the nitrogen to which they are bound, form an optionally substituted 5- to 7-membered nitrogen-containing heterocycloalkyl which optionally includes 1 or 2 additional heteroatoms selected from O, N, and S in the heterocycloalkyl ring; where each substituted group is independently substituted with one or more substituents independently selected from C1-C4 alkyl, aryl, heteroaryl, aryl-CrC4 alkyl-, heteroaryKVC alkyl-, Ci-C4 haloalkyl, -OC1-C4 alkyl, -OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyf-NH2, -N(C1-C4 alkyl)(CrC4 alkyl),
-NH(C1-C4 alkyl), -N(C1-C4 3IRyI)(C1-C4 alkylphenyl), -NH(C1-C4 alkylphenyl), cyano, nitro, oxo
(as a substitutent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OC1-C4 alkyl,
-CON(C1-C4 alkyl)(CrC4 alkyl), -CONH(C1-C4 alkyl), -CONH2, -NHC(O)(CrC4 alkyl),
-NHC(O)(phenyl), -N(C1-C4 3IkVl)C(O)(C1-C4 alkyl), -N(C1-C4 alkyl)C(O)(phenyl),
-C(O)C1-C4 alkyl, -C(O)C1-C4 alkylphenyl, -C(O)C1-C4 haloalkyl, -OC(O)C1-C4 alkyl, -
SO2(C1-C4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2, -SO2NH(C1-C4 alkyl),
-SO2NH(phenyl), -NHSO2(C1-C4 alkyl), -NHSO2(phenyl), and -NHSO2(C1-C4 haloalkyl).
[033] "Aryl" encompasses:
6-membered carbocyclic aromatic rings, for example, benzene; bicyclic ring systems wherein at least one ring is carbocyclic and aromatic, for example, naphthalene, indane, and tetralin; and tricyclic ring systems wherein at least one ring is carbocyclic and aromatic, for example, fluorene.
[034] For example, aryl includes 6-membered carbocyclic aromatic rings fused to a 5- to 7- membered heterocycloalkyl ring containing 1 or more heteroatoms chosen from N, O, and S.
For such fused, bicyclic ring systems wherein only one of the rings is a carbocyclic aromatic ring, the point of attachment may be at the carbocyclic aromatic ring or the heterocycloalkyl ring. Bivalent radicals formed from substituted benzene derivatives and having the free valences at ring atoms are named as substituted phenylene radicals. Bivalent radicals derived from univalent polycyclic hydrocarbon radicals whose names end in "-yl" by removal of one hydrogen atom from the carbon atom with the free valence are named by adding "-idene" to the name of the corresponding univalent radical, e.g., a naphthyl group with two points of attachment is termed naphthylidene. Aryl, however, does not encompass or overlap in any way with heteroaryl, separately defined below. Hence, if one or more carbocyclic aromatic rings is fused with a heterocycloalkyl aromatic ring, the resulting ring system is heteroaryl, not aryl, as defined herein.
[035] The term "aryloxy" refers to the group -O-aryl.
[036] The term "aralkyl" refers to the group -alkyl-aryl.
[037] "Carbamimidoyl" refers to the group -C(=NH)-NH2-
[038] "Substituted carbamimidoyl" refers to the group -C(=NR )-NR R where R , is chosen from: hydrogen, cyano, optionally substituted alkyl, optionally substituted cycloalkyl, optionally substituted aryl, optionally substituted heteroaryl, and optionally substituted heterocycloalkyl; f g and R and R are independently chosen from: hydrogen optionally substituted alkyl, optionally
substituted cycloalkyl, optionally substituted aryl, optionally substituted heteroaryl, and optionally substituted heterocycloalkyl, provided that at least one of Rβ, R , and R9 is not hydrogen and wherein substituted alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl refer respectively to alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
-Ra, -OR6, optionally substituted amino (including -NRcCORb, -NR0CO2R3, -NRcCONRbRc, - NRbC(NR°)NRbRc, -NRbC(NCN)NRbR°, and -NR0SO2R3), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -CORb), optionally substituted alkoxycarbonyl (such as -CO2R"), aminocarbonyl (such as -CONRbRc), -OCORb, -OCO2R3, -OCONRbRc, -OP(O)(O Rb)ORc, sulfanyl (such as SRb), sulfinyl (such as -SORa), and sulfonyl (such as -SO2R3 and -SO2NRbRc), where R is chosen from optionally substituted C-j-Cβ alkyl, optionally substituted aryl, and optionally substituted heteroaryl; b
R is chosen from H, optionally substituted C-\-CQ alkyl, optionally substituted aryl, and optionally substituted heteroaryl; and c
R is independently chosen from hydrogen and optionally substituted C1-C4 alkyl; or b c
R and R , and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C1-C4 alkyl, aryl, heteroaryl, aty\-C^C4 alkyl-, heteroaryl-d-C4 alkyl-, C1-C4 haloalkyl, -OC1-C4 alkyl, -OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyl-NH2, -N(C1-C4 8^yI)(C1-C4 alkyl), -NH(C1-C4 alkyl),
-N(C1-C4 8^yI)(C1-C4 alkylphenyl), -NH(C1-C4 alkylphenyl), cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OC1-C4 alkyl, -CON(C1-C4 alkyiχCτ-0, alkyl), -CONH(C1-C4 alkyl), -CONH2, -NHC(O)(C1-C4 alkyl), -NHC(OXphenyl), -N(C1-C4 8^yI)C(O)(C1-C4 alkyl), -N(C1-C4 alkyl)C(O)(phenyl), -C(O)C1-C4 alkyl, -C(O)C1-C4 phenyl, -C(O)C1-C4 haloalkyl, -OC(O)C1-C4 alkyl, - SO2(CrC4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2, -SO2NH(C1-C4 alkyl), -SO2 NH(phenyl), -NHSO2(C1-C4 alkyl), -NHSO2(phenyl), and -NHSO2(C1-C4 haloalkyl).
[039] The term "halo" includes fluoro, chloro, bromo, and iodo, and the term "halogen" includes fluorine, chlorine, bromine, and iodine.
[040] "Haloalkyl" indicates alkyl as defined above having the specified number of carbon atoms, substituted with 1 or more halogen atoms, up to the maximum allowable number of halogen atoms. Examples of haloalkyl include, but are not limited to, trifluoromethyl, difluoromethyl, 2-fluoroethyl, and penta-fluoroethyl. [041] "Heteroaryl" encompasses:
5- to 7-membered aromatic, monocyclic rings containing one or more, for example, from 1 to 4, or in certain embodiments, from 1 to 3, heteroatoms chosen from N, O, and S1 with the remaining ring atoms being carbon; bicyclic heterocycloalkyl rings containing one or more, for example, from 1 to 4, or in certain embodiments, from 1 to 3, heteroatoms chosen from N, O, and S, with the remaining ring atoms being carbon and wherein at least one heteroatom is present in an aromatic ring; and tricyclic heterocycloalkyl rings containing one or more, for example, from 1 to 5, or in certain embodiments, from 1 to 4, heteroatoms chosen from N, O, and S, with the remaining ring atoms being carbon and wherein at least one heteroatom is present in an aromatic ring. [042] For example, heteroaryl includes a 5- to 7-membered heterocycloalkyl, aromatic ring fused to a 5- to 7-membered cycloalkyl or heterocycloalkyl ring. For such fused, bicyclic heteroaryl ring systems wherein only one of the rings contains one or more heteroatoms, the point of attachment may be at either ring. When the total number of S and O atoms in the heteroaryl group exceeds 1 , those heteroatoms are not adjacent to one another. In certain embodiments, the total number of S and O atoms in the heteroaryl group is not more than 2. In certain embodiments, the total number of S and O atoms in the aromatic heterocycle is not more than 1. Examples of heteroaryl groups include, but are not limited to, (as numbered from the linkage position assigned priority 1), 2-pyridyl, 3-pyridyl, 4-pyridyl, 2,3-pyrazinyl, 3,4- pyrazinyl, 2,4-pyrimidinyl, 3,5-pyrimidinyl, 2,3-pyrazolinyl, 2,4-imidazolinyl, isoxazolinyl, oxazolinyl, thiazolinyl, thiadiazolinyl, tetrazolyl, thienyl, benzothiophenyl, furanyl, benzofuranyl, benzoimidazolinyl, indolinyl, pyridazinyl, triazolyl, quinolinyl, pyrazolyl, and 5,6,7,8- tetrahydroisoquinolinyl. Bivalent radicals derived from univalent heteroaryl radicals whose names end in "-yl" by removal of one hydrogen atom from the atom with the free valence are named by adding "-idene" to the name of the corresponding univalent radical, e.g., a pyridyl group with two points of attachment is a pyridylidene. Heteroaryl does not encompass or overlap with aryl, cycloalkyl, or heterocycloalkyl, as defined herein
[043] Substituted heteroaryl also includes ring systems substituted with one or more oxide (-O" ) substituents, such as pyridinyl N-oxides.
[044] By "heterocycloalkyl" is meant a single, non-aromatic ring, usually with 3 to 7 ring atoms, containing at least 2 carbon atoms in addition to 1-3 heteroatoms independently selected from oxygen, sulfur, and nitrogen, as well as combinations comprising at least one of the foregoing heteroatoms. The ring may be saturated or have one or more carbon-carbon double bonds. Suitable heterocycloalkyl groups include, for example (as numbered from the linkage position assigned priority 1), 2-pyrrolidinyl, 2,4-imidazolidinyl, 2,3-pyrazolidinyl, 2-piperidyl, 3-piperidyl, A- piperidyl, and 2,5-piperizinyl. Morpholinyl groups are also contemplated, including 2- morpholinyl and 3-morpholinyl (numbered wherein the oxygen is assigned priority 1). Substituted heterocycloalkyl also includes ring systems substituted with one or more oxo (=0) or oxide (-O") substituents, such as piperidinyl N-oxide, morpholinyl-N-oxide, 1-oxo-1- thiomorpholinyl and 1 ,1-dioxo-1-thiomorpholinyl.
[045] "Heterocycloalkyl" also includes bicyclic ring systems wherein one non-aromatic ring, usually with 3 to 7 ring atoms, contains at least 2 carbon atoms in addition to 1-3 heteroatoms independently selected from oxygen, sulfur, and nitrogen, as well as combinations comprising at least one of the foregoing heteroatoms; and the other ring, usually with 3 to 7 ring atoms, optionally contains 1-3 heteratoms independently selected from oxygen, sulfur, and nitrogen and is not aromatic.
[046] As used herein, "modulation" refers to a change in activity as a direct or indirect response to the presence of a chemical entity as described herein, relative to the activity of in the absence of the chemical entity. The change may be an increase in activity or a decrease in activity, and may be due to the direct interaction of the compound with the a target or due to the interaction of the compound with one or more other factors that in turn affect the target's activity. For example, the presence of the chemical entity may, for example, increase or decrease the target activity by directly binding to the target, by causing (directly or indirectly) another factor to increase or decrease the target activity, or by (directly or indirectly) increasing or decreasing the amount of target present in the cell or organism. [047] The term "sulfanyl" includes the groups: -S-(optionally substituted (Ci-Cβ)alkyl), -S-(optionally substituted aryl), -S-(optionally substituted heteroaryl), and -S-(optionally substituted heterocycloalkyl). Hence, sulfanyl includes the group Ci-C6 alkylsulfanyl.
[048] The term "sulfinyl" includes the groups: -S(O)-(optionally substituted (Ci-Cβ)alkyl), -S(0)-optionally substituted aryl), -S(O)-optionally substituted heteroaryl), -S(O)-(optionally substituted heterocycloalkyl); and -S(0)-(optionally substituted amino). [049] The term "sulfonyl" includes the groups: -S(O2)-(optionally substituted (CrCe)alkyl), -S(θ2)-optionally substituted aryl), -S(θ2)-optionally substituted heteroaryl), -S(O2)-(optionally substituted heterocycloalkyl) ,-S(O2)-(optioπally substituted alkoxy), -S(O2)-optionally substituted aryloxy), -S(02)-optionally substituted heteroaryloxy), -S(02)-(optionally substituted heterocyclyloxy); and -S(02)-(optionally substituted amino).
[050] The term "substituted", as used herein, means that any one or more hydrogens on the designated atom or group is replaced with a selection from the indicated group, provided that the designated atom's normal valence is not exceeded. When a substituent is oxo (i.e., =O) then 2 hydrogens on the atom are replaced. Combinations of substituents and/or variables are permissible only if such combinations result in stable compounds or useful synthetic intermediates. A stable compound or stable structure is meant to imply a compound that is sufficiently robust to survive isolation from a reaction mixture, and subsequent formulation as an agent having at least practical utility. Unless otherwise specified, substituents are named into the core structure. For example, it is to be understood that when (cycloalkyl)alkyl is listed as a possible substituent, the point of attachment of this substituent to the core structure is in the alkyl portion.
[051] The terms "substituted" alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl, unless otherwise expressly defined, refer respectively to alkyl, cycloalkyl, aryl, heterocycloalkyi, and heteroaryl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
-Ra, -OR", optionally substituted amino (including -NRcCORb, -NR0CO2R3, -NRcCONRbR°, - NRbC(NRc)NRbRc, -NRbC(NCN)NRbRc, and -NR0SO2R3), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -CORb), optionally substituted alkoxycarbonyl (such as -CO2Rb), aminocarbonyl (such as -CONRbR°), -OCORb, -OCO2R3, -OCONRbRc, -OP(O)(ORb)OR°, sulfanyl (such as SRb), sulfinyl (such as -SORa), and sulfonyl (such as -SO2R3 and -SO2NRbRc), where Ra is chosen from optionally substituted C1-C6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl;
Rb is chosen from hydrogen, optionally substituted C1-Ce alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl; and
Rc is independently chosen from hydrogen and optionally substituted Ci-C4 alkyl; or
Rband Rc, and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C1-C4 alkyl, aryl, heteroaryl, aryl-CrC4 alkyl-, heteroaryl-Ci-C4 alkyl-, C1-C4 haloalkyl, -OC1-C4 alkyl,
-OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyl-NH2,
-N(C1-C4 alkyl)(d-C4 alkyl), -NH(C1-C4 alkyl), -N(C1-C4 alkyl)(d-C4 alkylphenyl),
-NH(C1-C4 alkylphenyl), cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OC1-C4 alkyl, -CON(C1-C4 alkyl)(Ci-C4 alkyl), -CONH(Ci-C4 alkyl),
-CONH2, -NHC(O)(C1-C4 alkyl), -NHC(O)(phenyl), -N(C1-C4 8IkVl)C(O)(C1-C4 alkyl),
-N(C1-C4 alkyl)C(O)(phenyl), -C(O)C1-C4 alkyl, -C(O)C1-C4 alkylphenyl, -C(O)C1-C4 haloalkyl,
-OC(O)C1-C4 alkyl, -SO2(C1-C4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2,
-SO2NH(C1-C4 alkyl), -SO2NH(phenyl), -NHSO2(C1-C4 alkyl), -NHSO2(phenyl), and
-NHSO2(C1-C4 haloalkyl).
[052] The term "substituted acyl" refers to the groups (substituted alkyl)-C(O)-; (substituted cycloalkyl)-C(O)-; (substituted aryl)-C(O)-; (substituted heteroaryl)-C(O)-; and (substituted heterocycloalkyl)-C(O)-, wherein the group is attached to the parent structure through the carbonyl functionality and wherein substituted alkyl, cycloalkyl, aryl, heteroaryl, and heterocycloalkyl, refer respectively to alkyl, cycloalkyl, aryl, heteroaryl, and heterocycloalkyl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
-Ra, -ORb, optionally substituted amino (including -NRcCORb, -NRcCO2Ra, -NRcCONRbRc, -
NRbC(NRc)NRbRc, -NRbC(NCN)NRbRc, and -NR0SO2R3), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as
-CORb), optionally substituted alkoxycarbonyl (such as -CO2Rb), aminocarbonyl (such as
-CONRbRc), -OCORbi -OCO2R3, -OCONRbRc, -OP(O)(ORb)ORc, sulfanyl (such as SRb), sulfinyl
(such as -SORa), and sulfonyl (such as -SO2R3 and -SO2NR6R0), where Ra is chosen from optionally substituted Ci-Ce alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl;
Rb is chosen from H, optionally substituted CrC6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl; and
Rc is independently chosen from hydrogen and optionally substituted Ci-C4 alkyl; or Rb and Rc, and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from Ci-C4 alkyl, aryl, heteroaryl, aryl-Ci-C4 alkyl-, heteroaryl-C1-C4 alkyl-, Ci-C4 haloalkyl, -OC1-C4 alkyl, -OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyl-NH2, -N(C1-C4 alkyl)(d-C4 alkyl), -NH(C1-C4 alkyl),
-N(Ci-C4 alkyl)(Ci-C4 alkylphenyl), -NH(Ci-C4 alkylphenyl), cyano, nitro, oxo (as a substituent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OC1-C4 alkyl, -CON(C1-C4 alkyl)(CrC4 alkyl), -CONH(C1-C4 alkyl), -CONH2, -NHC(O)(C1-C4 alkyl), -NHC(OXphenyl), -N(C1-C4 alkyl)C(O)(CrC4 alkyl), -N(C1-C4 alkyl)C(O)(phenyl), -C(O)C1-C4 alkyl, -C(O)C1-C4 alkylphenyl, -C(O)C1-C4 haloalkyl, -OC(O)C1-C4 alkyl, - SO2(Ci-C4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2, -SO2NH(C1-C4 alkyl), -SO2NH(phenyl), -NHSO2(Ci-C4 alkyl), -NHSO2(phenyl), and -NHSO2(C1-C4 haloalkyl). [053] The term "substituted alkoxy" refers to alkoxy wherein the alkyl constituent is substituted (i.e., -©-(substituted alkyl)) wherein "substituted alkyl" refers to alkyl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from:
-Ra, -ORb, optionally substituted amino (including -NRcCORb, -NR0CO2R8, -NRcCONRbRc, - NRbC(NRc)NRbRc, -NRbC(NCN)NRbR°, and -NR0SO2R3), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -CORb), optionally substituted alkoxycarbonyl (such as -CO2Rb), aminocarbonyl (such as -CONRbR°), -OCOR", -OCO2R3, -OCONRbR°, -OP(O)(ORb)ORc, sulfanyl (such as SRb), sulfinyl (such as -SORa), and sulfonyl (such as -SO2Ra and -SO2NRbR°), where Ra is chosen from optionally substituted C1-C6 alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl; Rb is chosen from H, optionally substituted C1-C6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted, heteroaryl; and
Rc is independently chosen from hydrogen and optionally substituted C1-C4 alkyl; or Rb and Rc, and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C1-C4 alkyl, aryl, heteroaryl, aryl-Ci-C4 alkyl-, heteroaryl-C!-C4 alkyl-, C1-C4 haloalkyl, -OC1-C4 alkyl, -OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyl-NH2, -N(C1-C4 alkyl)(CrC4 alkyl), -NH(C1-C4 alkyl),
-N(C1-C4 8IKyI)(C1-C4 alkylphenyl), -NH(C1-C4 alkylphenyl), cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OC1-C4 alkyl, -CON(C1-C4 8^yI)(C1-C4 alkyl), -CONH(C1-C4 alkyl), -CONH2, -NHC(O)(C1-C4 alkyl), -NHC(O)(phenyl), -N(C1-C4 alkyl)C(O)(CrC4 alkyl), -N(C1-C4 alkyl)C(O)(phenyl), -C(O)C1-C4 alkyl, -C(O)C1-C4 alkylphenyl, -C(O)C1-C4 haloalkyl, -OC(O)C1-C4 alkyl, - SO2(C1-C4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2, -SO2NH(C1-C4 alkyl), -SO2NH(phenyl), -NHSO2(C1-C4 alkyl), -NHSO2(phenyl), and -NHSO2(C1-C4 haloalkyl). In some embodiments, a substituted alkoxy group is "polyalkoxy" or -O-(optionally substituted alkylene)-(optionally substituted alkoxy), and includes groups such as -OCH2CH2OCH3, and residues of glycol ethers such as polyethyleneglycol, and -0(CH2CH2O)xCH3, where x is an integer of 2-20, such as 2-10, and for example, 2-5. Another substituted alkoxy group is hydroxyalkoxy or -OCH2(CH2)yOH, where y is an integer of 1-10, such as 1-4. [054] The term "substituted alkoxycarbonyl" refers to the group (substituted alkyl)-O-C(O)- wherein the group is attached to the parent structure through the carbonyl functionality and wherein substituted refers to alkyl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from: -Ra, -ORb, optionally substituted amino (including -NRcCORb, -NR0CO2R3, -NRcCONRbRc, - NRbC(NR°)NRbRc, -NRbC(NCN)NRbRc, and -NR0SO2R3), halo, cyano, nitro. oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -CORb), optionally substituted alkoxycarbonyl (such as -CO2Rb), aminocarbonyl (such as -CONRbRc), -OCORb, -OCO2R3, -OCONRbR°, -OP(O)(ORb)ORc, sulfanyl (such as SRb), sulfinyl (such as -SORa), and sulfonyl (such as -SO2R3 and -SO2NRbR°), where Ra is chosen from optionally substituted Ci-C6 alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl;
Rb is chosen from H, optionally substituted Ci-C6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl; and
Rc is independently chosen from hydrogen and optionally substituted C1-C4 alkyl; or Rband Rc, and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C1-C4 alkyl, aryl, heteroaryl, 817!-C1-C4 alkyl-, heteroaryl-Crd alkyl-, C1-C4 haloalkyl, -OC1-C4 alkyl, -OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyl-NHz, -N(C1-C4 8^yI)(C1-C4 alkyl), -NH(C1-C4 alkyl),
-N(C1-C4 8^yI)(C1-C4 alkylphenyl), -NH(C1-C4 alkylphenyl), cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OCi-C4 alkyl, -CON(C1-C4 alkyl)(CrC4 alkyl), -CONH(C1-C4 alkyl), -CONH2, -NHC(O)(Ci-C4 alkyl), -NHC(O)(phenyl), -N(C1-C4 alkyl)C(O)(d-C4 alkyl), -N(C1-C4 alkyl)C(O)(phenyl), -C(O)C1-C4 alkyl, -C(O)C1-C4 alkylphenyl, -C(O)C1-C4 haloalkyl, -OC(O)C1-C4 alkyl, - SO2(C1-C4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2, -SO2NH(C1-C4 alkyl), -SO2NH(phenyl), -NHSO2(C1-C4 alkyl), -NHSO2(phenyl), and -NHSO2(C1-C4 haloalkyl). [055] The term "substituted amino" refers to the group -NHRd or -NRdRe wherein Rd is chosen from: hydroxy, optionally substituted alkoxy, optionally substituted alkyl, optionally substituted cycloalkyl, optionally substituted acyi, optionally substituted carbamimidoyl, aminocarbonyl, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted alkoxycarbonyl, sulfinyl and sulfonyl, and wherein Rβ is chosen from: optionally substituted alkyl, optionally substituted cycloalkyl, optionally substituted aryl, optionally substituted heteroaryl, and optionally substituted heterocycloalkyl, and wherein substituted alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl refer respectively to alkyl, cycloalkyl, aryl, heterocycloalkyl, and heteroaryl wherein one or more (such as up to 5, for example, up to 3) hydrogen atoms are replaced by a substituent independently chosen from: -Ra, -ORb, optionally substituted amino (including -NRcCORb, -NR0CO2R3, -NRcCONRbRc, - NRbC(NRc)NRbRc, -NRbC(NCN)NRbRc, and -NR0SO2R3), halo, cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, and heteroaryl), optionally substituted acyl (such as -CORb), optionally substituted alkoxycarbonyl (such as -CO2Rb), aminocarbonyl (such as
-CONRbR°), -OCORb, -OCO2R3, -OCONRbRc, -OP(O)(ORb)ORc, sulfanyl (such as SRb), sulfinyl
(such as -SORa), and sulfonyl (such as -SO2R3 and -SO2NRbRc), where Ra is chosen from optionally substituted C1-C6 alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryl, and optionally substituted heteroaryl;
Rb is chosen from H, optionally substituted CrC6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl, optionally substituted aryl, and optionally substituted heteroaryl; and
Rc is independently chosen from hydrogen and optionally substituted C1-C4 alkyl; or
Rb and Rc, and the nitrogen to which they are attached, form an optionally substituted heterocycloalkyl group; and where each optionally substituted group is unsubstituted or independently substituted with one or more, such as one, two, or three, substituents independently selected from C1-C4 alkyl, aryl, heteroaryl, 8FyI-C1-C4 alkyl-, heteroaryl-CrCj alkyl-, C1-C4 haloalkyl, -OC1-C4 alkyl, -OC1-C4 alkylphenyl, -C1-C4 alkyl-OH, -OC1-C4 haloalkyl, halo, -OH, -NH2, -C1-C4 alkyl-NH2, -N(C1-C4 alkyl)(Ci-C4 alkyl), -NH(C1-C4 alkyl),
-N(C1-C4 alkyl)(CrC4 alkylphenyl), -NH(C1-C4 alkylphenyl), cyano, nitro, oxo (as a substitutent for cycloalkyl, heterocycloalkyl, or heteroaryl), -CO2H, -C(O)OC1-C4 alkyl, -CON(C1-C4 8IKyI)(C1-C4 alkyl), -CONH(C1-C4 alkyl), -CONH2, -NHC(O)(C1-C4 alkyl), -NHC(OXphenyl), -N(C1-C4 alkyl)C(O)(d-C4 alkyl), -N(C1-C4 alkyl)C(O)(phenyl), -C(O)C1-C4 alkyl, -C(O)C1-C4 alkylphenyl, -C(O)C1-C4 haloalkyl, -OC(O)C1-C4 alkyl, - SO2(C1-C4 alkyl), -SO2(phenyl), -SO2(C1-C4 haloalkyl), -SO2NH2, -SO2NH(C1-C4 alkyl), -SO2NH(phenyl), -NHSO2(C1-C4 alkyl), -NHSO2(phenyl), and -NHSO2(C1-C4 haloalkyl); and wherein optionally substituted acyl, optionally substituted alkoxycarbonyl, sulfinyl and sulfonyl are as defined herein.
[056] The term "substituted amino" also refers to N-oxides of the groups -NHRd, and NRdRd each as described above. N-oxides can be prepared by treatment of the corresponding amino group with, for example, hydrogen peroxide or m-chloroperoxybenzoic acid. The person skilled in the art is familiar with reaction conditions for carrying out the N-oxidation. [057] Compounds of Formula X include, but are not limited to, optical isomers of compounds of Formula X, racemates, and other mixtures thereof. In those situations, the single enantiomers or diastereomers, i.e., optically active forms, can be obtained by asymmetric synthesis or by resolution of the racemates. Resolution of the racemates can be accomplished, for example, by conventional methods such as crystallization in the presence of a resolving
agent, or chromatography, using, for example a chiral high-pressure liquid chromatography (HPLC) column. In addition, compounds of Formula X include Z- and E- forms (or cis- and trans- forms) of compounds with carbon-carbon double bonds. Where compounds of Formula X exists in various tautomeric forms, chemical entities of the present invention include all tautomeric forms of the compound.
[058] Chemical entities of the present invention include, but are not limited to compounds of Formula X and all pharmaceutically acceptable forms thereof. Pharmaceutically acceptable forms of the chemical entities recited herein include pharmaceutically acceptable salts, solvates, crystal forms (including polymorphs and clathrates), chelates, non-covalent complexes, prodrugs, and mixtures thereof. In certain embodiments, the chemical entities described herein are in the form of pharmaceutically acceptable salts. Hence, the terms "chemical entity" and "chemical entities" also encompass pharmaceutically acceptable salts, solvates, chelates, non-covalent complexes, prodrugs, and mixtures. [059] "Pharmaceutically acceptable salts" include, but are not limited to salts with inorganic acids, such as hydrochloride, phosphate, diphosphate, hydrobromide, sulfate, sulfinate, nitrate, and like salts; as well as salts with an organic acid, such as malate, maleate, fumarate, tartrate, succinate, citrate, lactate, methanesulfonate, p-toluenesulfonate, 2-hydroxyethylsulfonate, benzoate, salicylate, stearate, and alkanoate such as acetate, HOOC-(CH2)n-COOH where n is 0-4, and like salts. Similarly, pharmaceutically acceptable cations include, but are not limited to sodium, potassium, calcium, aluminum, lithium, and ammonium.
[060] In addition, if the compound of Formula X is obtained as an acid addition salt, the free base can be obtained by basifying a solution of the acid salt. Conversely, if the product is a free base, an addition salt, particularly a pharmaceutically acceptable addition salt, may be produced by dissolving the free base in a suitable organic solvent and treating the solution with an acid, in accordance with conventional procedures for preparing acid addition salts from base compounds. Those skilled in the art will recognize various synthetic methodologies that may be used to prepare non-toxic pharmaceutically acceptable addition salts. [061] As noted above, prodrugs also fall within the scope of chemical entities, for example ester or amide derivatives of the compounds of Formula X. The term "prodrugs" includes any chemical entities that become compounds of Formula X when administered to a patient, e.g., upon metabolic processing of the prodrug. Examples of prodrugs include, but are not limited to, acetate, formate, phosphate, and benzoate and like derivatives of functional groups (such as alcohol or amine groups) in the compounds of Formula X.
[062] The term "solvate" refers to the chemical entity formed by the interaction of a solvent and a compound. Suitable solvates are pharmaceutically acceptable solvates, such as hydrates, including monohydrates and hemi-hydrates.
[063] The term "chelate" refers to the chemical entity formed by the coordination of a compound to a metal ion at two (or more) points.
[064] The term "non-covalent complex" refers to the chemical entity formed by the interaction of a compound and another molecule wherein a covalent bond is not formed between the compound and the molecule. For example, complexation can occur through van der Waals interactions, hydrogen bonding, and electrostatic interactions (also called ionic bonding). [065] The term "active agent" is used to indicate a chemical entity which has biological activity. In certain embodiments, an "active agent" is a compound having pharmaceutical utility. For example an active agent may be an anti-cancer therapeutic.
[066] By "significant" is meant any detectable change that is statistically significant in a standard parametric test of statistical significance such as Student's T-test, where p < 0.05. [067] The term "therapeutically effective amount" of a chemical entity described herein means an amount effective, when administered to a human or non-human patient, to provide a therapeutic benefit such as amelioration of symptoms, slowing of disease progression, or prevention of disease. [068] "Treatment" or "treating" means any treatment of a disease in a patient, including: a) preventing the disease, that is, causing the clinical symptoms of the disease not to develop; b) inhibiting the disease; c) slowing or arresting the development of clinical symptoms; and/or d) relieving the disease, that is, causing the regression of clinical symptoms.
[069] "Patient" refers to an animal, such as a mammal, that has been or will be the object of treatment, observation or experiment. The methods described herein can be useful in both human therapy and veterinary applications. In some embodiments, the patient is a mammal; in some embodiments the patient is human; and in some embodiments the patient is chosen from cats and dogs. [070] Provided is at least one chemical entity chosen from compounds of Formula X
Formula X and pharmaceutically acceptable salts thereof wherein
W1 is chosen from N and CR6 and W2 is chosen from N and CR4, provided that both W1 and W2 are not N;
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from hydrogen, halogen, and optionally substituted alkyl;
R3 is chosen from hydrogen and optionally substituted alkyl; and
R6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
[071] In some embodiments of compounds of Formula X, W1 is N and W2 is CR4. In some embodiments of compounds of Formula X1 W1 is CRβ and W2 is N. [072] Provided is at least one chemical entity chosen from compounds of Formula I
Formula I and pharmaceutically acceptable salts thereof, wherein
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is chosen from optionally substituted aryl and optionally substituted heteroaryl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from hydrogen and optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl. [073] In certain embodiments of compounds of Formula I, Z1 is optionally substituted aryl.
[074] In certain embodiments of compounds of Formula I, Z1 is optionally substituted heteroaryl.
[075] In certain embodiments of compounds of Formula I, Z3 is optionally substituted alkyl.
[076] In certain embodiments of compounds of Formula I, Z3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl.
[077] Provided is at least one chemical entity chosen from compounds of Formula Il
Formula Il
and pharmaceutically acceptable salts thereof wherein
R1 and R4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is optionally substituted aryl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, and optionally substituted aminocarbonyl;
Z3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
[078] In certain embodiments of compounds of Formula II, Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl. In certain embodiments, Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl. In certain
embodiments of compounds of Formula II, Z2 is chosen from (2-(N- acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2-
(methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-yl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl. [079] In certain embodiments of compounds of Formula II, R3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula II, R3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula II, R3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula II, R3 is hydrogen.
[080] In certain embodiments of compounds of Formula II, R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula II, R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula II, R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl. In certain embodiments of compounds of Formula II, R4 is hydrogen. [081] In certain embodiments of compounds of Formula II, Z2 is -C(O)-NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
[082] In certain embodiments of compounds of Formula II, R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[083] In certain embodiments of compounds of Formula II, R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl,
optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[084] In certain embodiments of compounds of Formula II, R2 is chosen from (1-(2- aminoethyl)-1H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-3-yl)methyl; (1 -methyl- 1 H-pyrazol-5-yl)methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1 -(2-(tert-butoxycarbonylamtno)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 1 -(hydroxy)-2-(aminocarbonylmethylamino)eth-1 -y I; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1-(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(arnino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; 1-aminocarbonyl-2-hydroxy-eth-1-yl; 1 -methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyI)-2-(amino)-eth-1 -yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1 -yl; 2-
(methylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbony))-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-yimethyl; 2-(t-butoxycarbonyIaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyιϊdin-3-ylmethyl; 2-methylpyridin-5-ylmethyl; 2-methyipyridin-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-{4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; 3-carbamoylcyclopentyl; 3-carboxy propyl; 3-hydroxypropyl; 3-methoxypropyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-{t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; 4- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; 4- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamiπo)methyleπeamiπo)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1 -yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazo!-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-yl methyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
[085] In certain embodiments of compounds of Formula II, Z3 is chosen from -(CH2)rR20 wherein r is chosen from 1 , 2, and 3 and R20 is chosen from optionally substituted aryl and optionally substituted heteroaryl.
[086] In certain embodiments of compounds of Formula M1 Z3 is chosen from 2-(3- methylphenyl)ethyl, 2-(1 H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(2,3- difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2-
chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1 H-pyrazol-1-yl)ethyl, 2-(3,5- dimethyl-1 H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fluorophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydroxyphenyI)ethyl, 2-(benzo[d][1,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-phenylprop-1-yl, 3-{1H-imidazol-1-yl)prop-1-yI, 3-phenylpropyl, benzyl, pyridin-2-ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
[087] In certain embodiments of compounds of Formula II, Z3 is 2-(pyridin-2-yl)ethyl. [088] In certain embodiments of compounds of Formula II, Z1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamlmidoyl, and optionally substituted sulfonyl. In certain embodiments of compounds of Formula II, Z1 is chosen from phenyl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, lower alkenyl, lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, lower acyl, dialkylamino, acetylamino, carbamimidoyl optionally substituted with cyano, aralkyl, and herteroaralkyl, and aminocarbonyl. In certain embodiments of compounds of Formula II, Z1 is chosen from phenyl, 2-aminocarbonylphenyl, 2-fluorophenyl, 2-chlorophenyl, 2-hydroxyphenyl, 2-methylphenyl, 2-methoxyphenyl, 2-cyanophenyl, 2-cyanomethylphenyl, 2-vinylphenyl, 2- formylphenyl, 3,4-difluorophenyl, 3-methylphenyl, 3-chlorophenyl, 3-fluorophenyl, 3- cyanophenyl, 3-dimethylaminophenyl, 3-methoxyphenyl, 3-methoxymethylphenyl, 3- hydroxyphenyl, 3-trifluoromethylphenyl, 4-aminocarbonylphenyl, 4-fluorophenyl, 4-chlorophenyl, 4-methylphenyl, 4-hydroxyphenyl, 4-cyanophenyl, 4-methoxyphenyl, 4-hydroxymethylphenyl, A- trifiuoromethylphenyl, 4-methoxycarbonylphenyl, 4-dimethylaminophenyl, 4-acetylphenyl, 4- carboxyphenyl, 4-ethylphenyl, 4-acetylaminophenyl, 3,4-dichlorophenyl, 3,4-difluorophenyl, 2,3- dimethylphenyl, 2,5-dimethylphenyl, 2,4-dimethylphenyl, 3,5-dimethylphenyl, 3,4- dimethylphenyl, 2-chloro-4-fluoro-phenyl, 2-cyano-4-fluoro-phenyl, 2-chloro-6-fluoro-phenyl, 2-
chloro-4-methoxy-phenyl, 2-cyano-4-methoxy-phenyl, 2-cyano-6-fluoro-phenyl, 2-chloro-5- fluoro-phenyl, 2-cyano-5-fluoro-phenyl, 2-chloro-4-methylphenyl, 2-chloro-4-hydroxymethyl- phenyl, 2-(N-(pyridin-3-ylmethyl)carbamimidoyl)phenyl, and 2-cyano-4-methylphenyl.
[089] In certain embodiments of compounds of Formula II, R1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula II, R1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula II,
R1 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula II,
R1 is hydrogen.
[090] Provided is at least one chemical entity chosen from compounds of Formula III
Formula III
and pharmaceutically acceptable salts thereof wherein
R1 and R4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is optionally substituted heteroaryl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, and optionally substituted aminocarbonyl;
Z3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
[091] In certain embodiments of compounds of Formula III, Z1 is chosen from heteroaryl optionally substituted by one or two groups selected from cyano, halo, hydroxy,
formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyi, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl. [092] In certain embodiments of compounds of Formula III, Z1 is chosen from 1 H-pyrazol-3-yl; 1 H-pyrazol-4-yl; 1 H-tetrazol-5-yl; 1-methyl-1 H-pyrazol-4-yl; 1-methyl-1 H-pyrazol-5-yl; 2- (hydroxymethyl)thiophen-3-yl; 2,2'-bipyridine; 2,3'-bipyridiπe; 2,4'-bipyridine; 2-cyanothiophen-3- yl; 2-formylthiophen-3-yl; 3-(hydroxymethyl)thiophen-2-yl; 3-cyano-1-methyl-1 H-pyrazol-4-yl; 3- formylthiophen-2-yl; 4-cyanothiophen-3-yl; 4-methylthiophen-2-yl; 4-methylthiophen-3-yl; 5-(1- hydroxyethyl)thiophen-2-yl; 5-(aminomethyl)thiophen-2-yl; 5-(hydroxymethyl)thiopheπ-2-yl; 5- (hydroxymethyl)thiophen-3-yl; 5-acetylthiophen-2-yl; 5-cyano-1 H-pyrazol-4-yl; 5-cyano-1- methyl-1 H-pyrazol-4-yl; 5-cyanothiophen-2-yl; 5-formylthiophen-2-yl; 5-formylthiophen-3-yl; 3- (aminomethyl)thiophen-2-yl; isoxazol-4-yl; thiazol-2-yl; thiophen-2-yl; and thiophen-3-yl. [093] In certain embodiments of compounds of Formula III, Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
[094] In certain embodiments of compounds of Formula III, Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl. [095] In certain embodiments of compounds of Formula III, Z2 is chosen from 2- (methylamino)-2-oxoethyl, 2-amino-2-oxoethyl, 3-amino-3-oxopropyl, (2-(N- acetylaminomethyl)piperidin-1 -yl)carbonyl; (2-aminomethylpiperidin-1 -yl)carbonyl; 1 -hydroxy-2- amino-ethyl; 2-((methyIsulfonamido)methyl)piperidin-1-ylcarbonyl; 2-
(methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-yl; amiπomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl. [096] In certain embodiments of compounds of Formula III, R3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula III, R3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula III, R3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula III, R3 is hydrogen.
[097] In certain embodiments of compounds of Formula III, R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula III, R4 is chosen from hydrogen, cyano, chloro,
bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula III, R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxy methyl. In certain embodiments of compounds of Formula III, R4 is hydrogen. [098] In certain embodiments of compounds of Formula III, Z2 is -C(O)NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
[099] In certain embodiments of compounds of Formula III, R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0100] In certain embodiments of compounds of Formula III, R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0101] In certain embodiments of compounds of Formula III, R2 is chosen from (1-(2- aminoethyl)-1H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1 -acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl;
(1 H-pyrazol-3-yl) methyl; (1 -methyl-1 H-pyrazol-3-yl)methyl; (1 -methyl-1 H-pyrazol-5-yl) methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylarnino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisorndolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2-
aminoethylamiπo)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1-(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazoI-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyt)-3-(amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarbonylmethylamino)eth-1-yl; 1 -(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1 -yl; 1 -(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1 -(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amiπo)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; i-aminocarbonyl-2-hydroxy-eth-i-yl; 1-methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamrno)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1-yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyi; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridiπ-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyrϊdin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylamiπocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; 3-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxypropyl; A-
(aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; A- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; A- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1-yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyI; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarboπylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
[0102] In certain embodiments of compounds of Formula III, Z3 is chosen from -(CH2)rR20 wherein r is chosen from 1 , 2, and 3 and R20 is chosen from optionally substituted aryl and optionally substituted heteroaryl.
[0103] In certain embodiments of compounds of Formula III, Z3 is chosen from 2-(3- methylphenyl)ethyl, 2-{1H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(2,3- difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-{3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1 H-pyrazof-1-yl)ethyl, 2-(3,5- dimethyl-1H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyI)ethyl, 2-(3- cyanopheπyOethyi, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fludrophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluoropheny!)ethyl, 2- (4-hydroxyphenyl)ethyl, 2-(benzo[d][1 ,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-phenylprop-1-yl, 3-(1 H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, pyridin-2-ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
[0104] In certain embodiments of compounds of Formula III, Z3 is chosen from 3- fJuorophenethyl, 3,5-difluorophenethyl, and 2-(pyridin-2-yl)ethyl. In certain embodiments of compounds of Formula III, Z3 is 2-(pyridin-2-yl)ethyl.
[0105] In certain embodiments of compounds of Formula III, R1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula III, R1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula III,
R1 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula III,
R1 is hydrogen.
[0106] Provided is at least one chemical entity chosen from compounds of Formula IV
Formula IV and pharmaceutically acceptable salts thereof wherein
R1 and R4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is chosen from optionally substituted aryl and optionally substituted heteroaryl;
Z2 is chosen from hydrogen, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted amidino, and optionally substituted aminocarbonyl;
Z3 is optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
[0107] In certain embodiments of compounds of Formula IV, Z1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted
aminocarbonyl, optionally substituted amino optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl, and heteroaryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkeπyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl. [0108] In certain embodiments of compounds of Formula IV, Z1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted lower acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and amino optionally substituted with alkyl, and heteroaryl optionally substituted by one or two groups selected from optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted lower acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and amino optionally substituted with alkyl.
[0109] In certain embodiments of compounds of Formula IV, Z1 is chosen from phenyl,
1 H-pyrazol-3-yl; 1H-pyrazol-4-yl; 1 H-tetrazol-5-yl; 1-methyl-1H-pyrazol-4-yl; 1-methyl-1H- pyrazol-5-yl; 2-aminomethylthiophen-5-yl; 2-cyanothiophen-3-yl; 2-formylthiophen-3-yl; 2- formylthiophen-4-yl; 2H-pyrrol-1(5H)-yl; 2-hydroxymethylthiophen-3-yl; 2- hydroxymethylthiophen-4-yl; 2-hydroxymethylthiophen-5-yl; 3-aminomethylthiophen-2yl; 3- cyano-1-methyl-1H-pyrazol-4-yl; 3-cyanothiophen-4-yl; 3-formylthiophen-2-yl; 3- hydroxymethylthiophen-2-yl; 4-methylthiophen-2-yl; 4-methylthiophen-3-yl; 5-(1-hydroxyeth-1- yl)-thiophen-2-yl; 5-acetylthiophen-5-yl; 5-cyano-1H-pyrazol-4-yl; 5-cyano-1-methyl-1H-pyrazol- 4-yl; 5-cyanothiophen-2-yl; 5-formylthiophen-2-yl; 5-hydroxymethylthiophen-2-yl; isoxazol-4-yl; pyridin-2-yl; pyridin-3-yl; pyridin-4-yl; thiazol-2-yl;thiophen-2-yl; and thiophen-3-yl. In certain embodiments of compounds of Formula IV, Z1 is phenyl.
[0110] In certain embodiments of compounds of Formula IV, Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
[0111] In certain embodiments of compounds of Formula IV, Z2 is chosen from carboxyl, optionally substituted pyridinylmethylcarbonyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
[0112] In certain embodiments of compounds of Formula IV1 Z2 is chosen from pyridin-
3-ylmethylcarbonyl, (2-(N-acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1- yl)carbonyl; 1-hydroxy-2-amino-ethyl; 2-({methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2- (methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-ylmethyl; amiπomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ytcarbonyl. [0113] In certain embodiments of compounds of Formula IV, Z2 is -C(O)NR2R5 wherein
R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
[0114] In certain embodiments of compounds of Formula IV, R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0115] In certain embodiments of compounds of Formula IV, R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0116] In certain embodiments of compounds of Formula IV, R2 is chosen from {1-(2- aminoethyl)-1 H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1-rnethyl-1H-pyrazol-3-yl)methyl; (1-methyl-1H-pyrazol-5-yl)methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2-
(hydroxy)ethylamino)carboπylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridiπ-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1-(2-(tert-butoxycarbonylamino)ethyl)-1H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1 -(aminocarbonyl)-4-(amino)-butyl; 1 -(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1 - (aminocarbonyl)-5-(amino)-pentyl; 1~(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarboπylmethylamino)eth-1-yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1-(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; i-aminocarbonyl-2-hydroxy-eth-i-yl; 1 -methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-amiπoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamiπo)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarboπyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1 -yl; 2- (methylamiπocarbony!)-2-(amiπo)-eth-1 -yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-yl methyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2-
aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylamiπocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyI)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; S-carbamoylcyclopentyl; 3-carboxy propyl; 3-hydroxypropyl; 3-methoxypropyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethyfamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; A- cyanobenzyl; 4-fluorobenzyl; 4-methyIamiπopyridin-3-ylmethyl; 4-methylbenzyl; 4- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminornethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyt; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1-yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonyimethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
[0117] In certain embodiments of compounds of Formula IV, R2 is pyridin-3-ylmethyl.
[0118] In certain embodiments of compounds of Formula IV, R5 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula IV, R5 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula IV, R5 is hydrogen.
[0119] In certain embodiments of compounds of Formula IV, Z3 is chosen from optionally substituted cycloalkylalkyl, optionally substituted heterocycloalkylalkyl. In certain embodiments of compounds of Formula IV1 Z3 is chosen from -(CH≥JrR20 wherein r is chosen from 1 , 2, and 3 and R20 is chosen from optionally substituted heterocycloalkyl and optionally substituted cycloalkyl.
[0120] In certain embodiments of compounds of Formula IV, Z3 is chosen from
(tetrahydrofuran-2-yl)methyl, 1-hydroxycyclohexylmethyl, 2-(1 ,3-dioxolan-2-yl)ethyl, 2-(1- methylpiperidin-4-yl)ethyl, 2-(2-oxoimidazolidin-1-yl)ethyl, 2-(2-oxopiperidin-1-yl)ethyl, 2-(2- oxopyrrolidin-1-yl)ethyl, 2-cyclohex-1-enylethyl, 2-cyclohexylethyl, and 2-morpholinoethyl.
[0121] In certain embodiments of compounds of Formula IV, R1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula IV, R1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula IV1 R1 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula IV, R1 is hydrogen.
[0122] In certain embodiments of compounds of Formula IV, R3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula IV, R3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula IV, R3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula IV1 R3 is hydrogen.
[0123] In certain embodiments of compounds of Formula IV, R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula IV, R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula IV1 R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl. In certain embodiments of compounds of Formula IV, R4 is hydrogen. [0124] In certain embodiments, the compound of Formula I is chosen from
COMPOUND CHEMICAL NAME
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-prop-2-enylcarboxamide
N-methyl(2-{[2-(3-methylphenyl)ethyl]amino}-6- phenyl(3-pyridyl))carboxamide
N-methyl{6-phenyl-2-[(2-ρhenylethyl)amino](3- pyridyl)}carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N- prop-2-enylcarboxamide
(2-{[2-(3-methylphenyl)ethyl]amino}-6-phenyl(3- pyridyl))-N-prop-2-enylcarboxamide
N-methyl{6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-benzyl{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3- pyridyl)}carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N- (3-pyridylmethyl)carboxamide
N-(methylethyl){6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
{6-phenyl~2-[(2-(2-pyridyl)ethyl)amino](3-pyιϊdyl)}- N-(4-pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(3-pyridyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(2-py ridyl)ca rboxam ide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(4-pyridyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(2-thienylmethyl)carboxamide
(6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(3-thienylmethyl)carboxamide
N-(oxolan-2-ylmethyl){6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((2S)oxolan-2-yl)methyl]{6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
tert-butyl 2-[({6-phenyl-2-[(2-(2- pyridyl)ethyl)amino]-3- pyridyl}carbonylamino)methyl]morpholine-4- carboxylate
N-[(1-acetyl(4-piperidyl))methyl]{6-phenyl-2-[(2- (2-pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[(1-acetyl(3-piperidyl))methyl]{6-phenyl-2-[(2- (2-py ridyl)ethy l)am i n o](3-pyridy l)}ca rboxa m ide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(4-piperidylmethyl)carboxamide
{2-[(2-cyclohex-1 -enylethyl)amino]-6-phenyl(3-
pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-phenyl-2-[(3-phenylpropyl)amino](3-pyridyl)}- N-(3-pyridy lmethy l)ca rboxam ide
{6-phenyl-2-[benzylamino](3-pyridyl)}-N-(3- pyridy lmethyl)ca rboxam ide
N-(1,3-oxazol-2-ylmethyl){6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-(imidazol-2-ylmethyl){6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((2S)-1-acetylpyrrolidin-2-yl)methyl]{6-phenyl-
2-[(2-(2-pyridyl)ethyl)amino](3- pyridyl)}carboxamide
N-[((3R)-1-acetylpyrrolidin-3-yl)methyl]{6-phenyl-
2-[(2-(2-pyridyl)ethyl)amino](3- pyridyl)}carboxamide
N-[((3S)-1-acetylpyrrolidin-3-yl)methyl]{6-phenyl-
2-[(2-(2-pyridyl)ethyl)amino](3- pyridyl)}carboxamide
methyl 2-({6-phenyl-2-[(2-phenylethyl)amino]-3- pyridyl}carbonylamino)acetate
[6-phenyl-2-(propylamino)(3-pyridyl)]-N-(3- pyridy lmethyl)carboxa m ide
{2-[(3-methylbutyl)amino]-6-phenyl(3-pyridyl)}-N- (3-pyridylmethyl)carboxamide
{2-[(2-cyclohexylethyl)amino]-6-phenyl(3- pyridyl)}-N-(3-pyridylmethyl)carboxamide
N-{[1-(methylsulfonyl)(3-piperidyl)]methylK6- phenyl-2-[(2-(2-pyridyl)ethyl)amino](3- pyridyl)}carboxamide
N-{[1-({6-phenyl-2-[(2-(2-pyridyl)ethyl)amino]-3- pyridyl}carbonyl)-2-piperidyl]methyl}acetamide
2-{[(methylsulfonyl)amino]methyl}piperidyl 6- phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl) ketone
N-[(1-methylpyrazol-5-yl)methyl]{6-phenyl-2-[(2- (2-pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[(1-methylpyrazol-3-yl)methyI]{6-phenyl-2-[(2- (2-pyridyl)ethyl)amino](3-pyridyl)}carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- N-(pyrazol-3-ylmethyl)carboxamide
N-(isoxazol-5-ylmethyl){6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((3S)pyrrolidin-3-yl)methyl]{6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((3R)pyrrolidin-3-yl)methyl]{6-phenyl-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N- (2-(2-pyridyl)ethyl)carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N- (2-(3-pyridyl)ethyl)carboxamide
N-methyl-2-({6-phenyl-2-[(2- phenylethyl)amino](3- pyridyl)}carbonylamino)acetamide
N,N-dimethyl-2-({6-phenyl-2-[(2- phenylethyl)amino](3- pyridyl)}carbonylamino)acetamide
(2-{[2-(2-oxopiperidyl)ethyl]amino}-6-phenyl(3- pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(1-methyl(4-piperidyl))ethyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(2-thienyl)ethyl)amino](3-pyιϊdyl)}- N-(3-pyridylmethyl)carboxamide
(2-{[2-(1-methylimidazol-4-yl)ethyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
{2-[(2-(2-furyl)ethyl)amino]-6-phenyl(3-pyridyl)}-N- (3-pyridylmethyl)carboxamide
{2-[(oxolan-2-ylmethyl)amino]-6-phenyl(3- pyridyl)}-N-(3-pyridylmethyl)carboxamide
{2-[(2-(1,3-dioxolan-2-yl)ethyl)amino]-6-phenyl(3- pyridyl)}-N-(3-pyridylmethyl)carboxamide
{2-[(3-imidazolylpropyl)amino]-6-phenyl(3- pyridyl)}-N-(3-pyridylmethyl)carboxamide
{2-[(2-morpholin-4-ylethyl)amino]-6-phenyl(3- pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-phenyl-2-[(2-phenoxyethyl)amino](3-pyridyl)}- N-(3-pyridylmethyl)carboxamide
{2-[(2-(2H-benzo[3,4-d]1,3-dioxolen-5- yl)ethyl)amino]-6-phenyl(3-pyridyl)}-N-(3- pyridylmethyl)carboxamide
(2-{[2-(2-oxoimidazolidinyl)ethyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
{2-[(2-imidazol-4-ylethyl)amino]-6-phenyl(3- pyridy1)}-N-(3-pyridylmethyl)carboxamide
(2-{[(hydroxycyclohexyl)methyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(2-{[2-(3,5-dimethylpyrazol-4-yl)ethyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(2-{[2-(3,5-dimethylpyrazolyl)ethyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(2-{[2-(2-chlorophenyl)ethyl]amino}-6-(2- cyanophenyl)(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(2-
methylphenyl)ethyl]amino}(3-pyridyl))-N-(3- pyridylmethyljcarboxamide
(6-(2-cyanopheny!)-2-{[2-(2- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(2- cyanophenyl)ethyl]amino}(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
{6-(2-cyanophenyl)-2-[(2-(2- thienyl)ethyl)amino](3-pyridyl)}-N-(3- pyridylmethyl)carboxamide
{6-(2-cyanophenyl)-2-[(2-(2-furyl)ethyl)amino](3- pyridyl)}-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluoro(2-pyridyl))ethyl]amino}-6- phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(2-{[2-(3-methylphenyl)ethyl]amino}-6-phenyl(3- pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[2-(aminornethyl)(3-pyridyl)]methyl}{6-pyrazol-
4-yl-2-[(2-(2-pyridyl)ethyl)amino](3- pyridyl)}carboxamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)-2- hydroxyethyl]amino}-6-pyrazol-4-yl(3- pyridyl))carbonylamino]-isopropyl}-2-[(tert- butoxy)carbonylamino]acetamide
{5-fluoro-6-pyrazol-4-yl-2-[(2- pyrazolylethyl)amino](3-pyridyl)}-N-(3- pyridylmethyl)carboxamide
N-[(1R)-2-({5-fluoro-6-pyrazol-4-yl-2-[(2- pyrazolylethyl)amino](3-pyridyl)}carbonylamino)- isopropyl]-2-aminoacetamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[6-
(trifluoromethyl)(2-pyridyl)]methyl}carboxamide
(trifluoromethyl)(2-pyridyl)]methyl}carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyI)ethyl]amino}(3-pyridyl))-N-{[3-
(trifluoromethyl)(2-pyridyl)]methyl}carboxamide
N-[(6-chloro(3-pyridyl))methyl](6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyano(3-thienyl))-2-{[2-(3- fIuorophenyl)ethyl]amino}(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(2E)-3-(2-{[2-(3-fluorophenyl)ethyl]amino}-6- phenyl(3-pyridyl))-N-methylprop-2-enamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[6-
(methylamino)(3-pyridyl)]methyl}carboxamide
N-{[6-(dimethylamino)(3-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-4-
N-[(5-amino(2-pyrϊdyl))methyl](6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-5-
(hydroxymethyl)-6-phenyl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl-5-[N-
(3-pyridylmethyl)carbamoyl](3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(5-(2-diazo-2-azavinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
(3-pyridylmethyl)carboxamide
(5-(aminomethyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
(3-pyridylrriethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
methylpyrazol-3-yl)(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
N-({6-[3,3-bis(dimethylamino)-2-azaprop-2- enyl](2-pyridyl)}methyl)(6-(2-cyanophenyl)-2-{[2- (3-fl uoropheny l)ethyl]am ino}(3- pyridyl))carboxamide
tert-butyl 2-(6-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3- pyridyl)carbonylamino]methyl}-2-pyridyl)acetate
N-{[4-(aminomethyl)(3-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridy1))carboxamide
N-{[3-(aminomethyl)(4-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(6-(5-cyanopyrazol-4-yl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3- yl(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(3-cyano-1-methylpyrazol-4-yl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
(6-(5-cyano-1-methylpyrazol-4-yl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3- pyridylmethyl)carboxamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-
4-yl(3-pyridyl))carbonylamino]-N- methylacetamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-N- methylacetamide
pyridylmethyl)carboxamide
N-{[6-(aminomethyl)(2-pyridyl)Jmethyl}(5-bromo-
2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3- pyridyl))carboxamide
rboxamide
N-{[1-(2-aminoethyl)pyrazol-3-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-5-yiJmethyl}(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3- pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(5-bromo-
2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-5-oxopyrrolidin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)-5-oxopyrrolidin-2-yl)methyll(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{3-[(5S)-5-(2-diazo-2-azavinyl)-2- oxopyrrolidinyl]propyl}(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3- pyridyl))carboxam id e
N-[((2S)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))carboxamide
N-{[(2S)-1-(2-aminoethyl)-5-oxopyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{3-[(5S)-5-(aminomethyl)-2- oxopyrrolidinyl]propyl}(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3- pyridyl))carboxamide
N-{[(2R)-1-(2-aminoethyl)-5-oxopyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{3-[(5R)-5-(2-diazo-2-azavinyl)-2- oxopyrrolidinyl]propyl}(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3- pyridyl))carboxamide
N-{3-[(5R)-5-(aminomethyl)-2- oxopyrrolidinyl]propyl}(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3- pyridyl))carboxamide
N-{2-[(5S)-5-(aminomethyl)-2- oxopyrrolidinyl]ethyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 412 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 413 pyridyl))carboxamide
N-[((2R)-5-oxopyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compou yl)(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-
Compou (pyrrolidin-2-ylmethyl)carboxamide
Compound 420 pyridy l))carboxa m ide
}pyrrolidin-2- l)-2-{[2-(3-
Compound 424 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 425 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 428 pyridyl))carbonylamino]propyl}amino)acetate
Compound 429 pyridyl))carbonylamino]propyl}amino)acetate
N-[((2S)-6-oxopiperazin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compoun pyridyl))carboxamide
N-[((2S)-6-oxopiperazin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 434
N-[((2R)-4-{3-[(tert-butoxy)carbonylamino]propyl}- 6-oxopiperazin-2-yl)methyl](6-(2-cyanophenyl)-2- {[2-(3-fluorophenyl)ethyl]arnino}(3-
Compoun pyridyl))carboxamide
N-[((2S)-6-oxopiperazin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compoun yl)(3-pyridyl))carboxamide
N-[((2S)-6-oxopiperazin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
Compoun pyridyl)) ca rboxam ide
Compound 438 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 0 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 440 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-4-(2-aminoethyl)-6-oxopiperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compound fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 452 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({(2R)-1 -[4-(1 ,3-dioxobenzo[c]a2olin-2-yl)butyl]- 5-oxopyrrolidin-2-yl}methyl)(6-(2-cyanophenyl)-2- {[2-(3-fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carboxamide
N-({(2R)-1-[5-(1 ,3-dioxobenzo[c]azoIin-2- yi)pentyll-5-oxopyrrolidin-2-yl}methyl)(6-(2-
cyanophenyl)-2-{[2-(3-
Compound 454 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
tert-butyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}pyrrolidinecarboxyl
Compound 455 ate
Compound 1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(5-aminopentyl)-5-oxopyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 2
Compound 3 pyridyl))carboxamide
Compound 459 yl(3-pyridyl))-N-(pyrazol-3-ylmethyl)carboxamide
ΛTΎ (2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
"TΛJ methylpyrazol-4-yl)(3-pyridyl))-N-(pyrazol-3-
Compound 460 ylmethyl)carboxamide
Compound 461 yl(3-pyridyl))-N-(pyrazol-3-ylmethyl)carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2-
yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compound 462 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 464 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-3-yl3methyl}(6-(2-
cyanophenyl)-2-{[2-(3-
Compound 466 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 467 4-yl)(3-pyridyl))carboxamide
Compound 468 pyridyl))carboxamide
Compound 469 4-yl)(3-pyridyl))carboxamide
Compound 470 pyridyl))carboxarn id e
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]ethyl}pyrrolidin-2-
yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compound 471 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert- butoxy)carbonylamino]propyl}pyrrolidin-2- yl)methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 472
Compound 473 fluorophenyl)ethyl]amino}(3-pyridy!))carboxamide
Compound 474 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
o]ethyl}pyrazol-3- orophenyl)ethyl]amino}-6-
Compound 480 pyridyl))carboxamide
Compound 481 pyridyl))carboxamide
Compound 482 pyridyl))carboxamide
N-{[1-<2-aminoethyl)pyrazol-3-yl]methyl}(2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 483 yl)(3-pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-5-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compou yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxopiperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxopiperazin-2- yl]methyl}(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
Compou methylpyrazol-4-yl)(3-pyridyI))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxopiperazin-2- yl]methyl}(2-{[2-(3-fluorophenyl)ethyl]amino}-6-
Compou pyrazol-4-yl(3-pyridyl))carboxamide
Compound 488 pyrazol-3-yl(3-pyridyl))carboxamide
N-[((2R)-1-acetylpyrrolidin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 489
N-{l(2R)-1-(methylsulfonyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-methylpyrrolidin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 492 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{[3-(1 ,3-dioxobenzo[c]azolin-2- yl)propyl]sulfonyl}pyrrolidin-2-yI)methyl](6-(2-
cyanophenyl)-2-{[2-(3-
Compound 494 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 495 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 496 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-3-yl]methyl}(2-{[2-
(3-fluorophenyl)ethyl]amino}-6~pyrazol-4-yl(3-
Compound 497 pyridyl))carboxamide
Compound 498 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidin-2-yl)methy[](2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 520 yl)(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(6-{1-
Compound 522 pyridyl))carboxamide
Compound 523 methylpyrazol-4-yl)(3-pyrldyl))carboxamide -methylpyrazol-4- }pyrrolidinecarboxyl
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(2-{[2-(3-fluorophenyl)ethyl]amino}-6-
pyrazol-4-yl(3-pyridyl))carboxamide
Compound 526
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidϊn-2-yl)methyl](6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 528 pyιϊdyl))carboxamide
N-(2-amino-2-methylpropyl)(6-(2-cyanophenyl)-2-
{[2-(3-fIuorophenyl)ethyl]amino}(3-
Compou pyridyl))carboxamϊde
N-[((2R)pyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amJno}-6-pyrazol-4-yl(3-
Compou pyridyl))carboxamide
N-[((2R)pyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compou yl)(3-pyridyl))carboxamide
Compound 537 2-[(methylsulfoπyl)amiπo]propyl}carboxamide
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}pyrrolidin-2-
yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(1-
Compound 538 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert- butoxy)carbonylamino]propanoyl}pyrrolidin-2-
yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
Compound 539 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2-
yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
Compound 540 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 541 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 542 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 543 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 544 py ridyl))ca rboxam ide
2-[(tert-butoxy)carbonylamino]-N-{2-[(6-(2- cyanophenyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-
pyridyl))carbonylamino]-tert-butyl}acetamide
Compound 545
Compound 546 pyridyl))carbonylamino]-tert-butyl}butanamide
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-4-[(tert-
Compound 547 butoxy)carbonylamino]butanamide
N-[((2R)-1-{5-[(tert- butoxy)carbonylamino]pentanoyl}pyrrolidin-2-
yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compound 548 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 549 pyridyl))carbonylamino]-tert-butyl}acetamide
Compound 550 pyridyl))carbonylamino]-tert-butyl}butanamide
Compound 551 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}pyrrolidinecarboxyl
Compound 552 ate
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-
pyridyl))carbonylamino]-isopropyl>-2-
Compound 555 aminoacetamide
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-3-
Compound 556 aminopropanamide
Compound 557 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 558 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-aminoethyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}pyrrolidinecarboxyl
Compound 602 ate
N-[(2S)-2-(methylamino)propyl](6-(2- cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 603
N-((2R)-2-aminopropyl)(6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carboxamide
Compound 605 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
ino]propyl}(6-(2-
Compound 607 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
ino]propyl}(6-(2-
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-2-[(tert-
Compound 609 butoxy)carbonylamino]acetamide
N-{(1 R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-2-[(tert-
Compound 610 butoxy)carbonylamino]-N-methylacetamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-4-[(tert-
Compound 611 butoxy)carbonylamino]butanamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-4-[(tert-
Compound 612 butoxy)carbonylamino]-N-methylbutanamide
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{t2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-2-amino-N-
Compound 613 methylacetamide
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyljamino}(3-
pyridyl))carbonylamino]-isopropyl}-4-amino-N-
Compound 614 methylbutanamide
N-{(1 R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-2-
Compound 615 aminoacetamide
N-{(1 R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]-isopropyl}-4-
aminobutanamide
Compound 616
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-4-amino-N-
Compound 617 methylbutanamide
N-{(1 R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-isopropyl}-2-amino-N-
Compound 618 methylacetamide
Compound 619 pyridylmethyl)carboxamide
Compound 463 fluorophenyl)ethyl]arnino}(3-pyridyl))carboxamide
Compound 465 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 410 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-
Compoun yl(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
{6-(3-methylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(3-fluorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(3-cyanophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(3-methoxypheny])-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(3-hydroxyphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(3-chlorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
pyridylmethyl)carboxamide
Compound 7
{2-[(2-(2-pyridyl)ethyl)amino]-6-[3-
(trifluoromethyl)phenyl](3-pyridyl)}-N-(3- pyridylmethyl)carboxamide
Compound 11 pyιϊdylmethyl)carboxamide
{6-(2-fluorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(2-chlorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(2-methylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 15 pyridylmethyl)carboxamide
{6-(2-cyanophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-(2-thienyl)(3-
Compoun pyridyl)}-N-(3-pyridylmethyl)carboxamide
Compound 58 pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-(4-hydroxyphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compou pyridylmethyl)carboxamide
{6-(2-hydroxyphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compou pyridylmethyl)carboxamide
{6-(4-ethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-
Compound 26 pyridyl)}-N-(3-pyridylmethyl)carboxamide
N-(4-{6-[(2-(2-pyridyl)ethyl)amino]-5-[N-(3- pyridylmethyl)carbamoyl]-2-
pyridyl}phenyl)acetamide
Compound 861
{6-(4-methyl(2-thienyl))-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(4-methyl(3-thienyl))-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(2,3-dimethylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(2,5-dinnethylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(2,4-dimethylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamtde
Compound 33 pyridylmethyl)carboxamide
{6-(3,4-dimethylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyιϊdyl)}-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(4-fluorophenyl)ethyl]amino}-6-phenyl<3-
Compou pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(4-chlorophenyl)ethyl]amino}-6-phenyl(3-
Compou pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-chlorophenyl)ethyl]amino}-6-phenyl(3-
Compou pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-methoxyphenyl)ethyl]amino}-6-pheny!(3-
Compou pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-
Compou pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(2-chlorophenyl)ethyl]amino}-6-phenyl(3-
pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 70
(2-{[2-(2-methoxyphenyl)ethyl]amino}-6-phenyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(2-methylphenyl)ethyl]amino}-6-phenyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(2-fluorophenyl)ethyl]amino}-6-phenyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(2,6-difluorophenyl)ethyl]amino}-6- phenyl(3-pyridyI))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6- phenyl(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 84 pyridylmethyl)carboxamide
<2-{I2-(2,5-difluorophenyl)ethyl]amino}-6- phenyl(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxarnide
[6-phenyl-2-({2-[3-
(trifluoromethyl)phenyl]ethyl}amino)(3-pyridyl)]-N-
Compoun (3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-methyl(2- thienyl))(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(3- thienyl)(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(2-cyanophenyl)ethyl]amino}-6-phenyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-cyanophenyl)ethyl]amino}-6-phenyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(4-hydroxyphenyl)ethyl]amino}-6-phenyl(3-
pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 91
(2-{[2-(2-hydroxyphenyl)ethyl]amino}-6-phenyi(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(4-aminophenyl)ethyl]amino}-6-phenyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
3-[2-{{6-phenyl-3-[N-(3-pyridylmethyl)carbamoyl]-
Compoun 2-pyridyl}amino)ethyl]benzamlde
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(3- thienyl)(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(4- methyl(2-thieπyl))(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 99 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2- thienyl)(3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(2- thienyl)(3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
methyl 3-[2-({6-phenyl-3-[N-(3- pyridylmethyl)carbamoyl]-2-
Compoun pyridyl}amino)ethyl]benzoate
Compound 108 pyridylmethyl)carbamoyl]-2-pyridyl)benzamide
(6-(4-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
3-[2-({6-phenyl-3-[N-(3-pyridylmethyl)carbamoyl]-
Compoun 2-pyridyl}amino)ethyl]benzoic acid
(6-(2-chloro-4-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
Compound 156
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(4- fluorophenyl)(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(6-(2-cyano-4-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(6-(2-chloro-6-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-(2-chloro-5-methoxyphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-(2-cyano-5-methoxyphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
Compound 206 pyridylmethyl)carboxamide
(6-(2-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(2,3-difluorophenyl)ethyl]amino}-6-(2- cyanophenyl)(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-(2-chloro-5-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridy!))-N-(3-
Compou pyridylmethyl)carboxamide
(6-(2-cyano-5-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-(3-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-[2-(cyanomethyl)phenyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[3-
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(5-formyl(3- thienyl))(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5-
(hydroxymethyl)(3-thienyl)](3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(6-(2-chloro-4-methylphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(6-(2-cyano-4-methylphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 281 pyridylmethyl)carboxamide
Compound 282 pyridylmethyl)carboxamide
(6-(1 H-1 ,2,3,4-tetraazol-5-yl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
Compound 289 pyridylmethyl)carboxamide
Compound 290 pyridyl))carbonylamino]-N-methylbutanamide
Compound 294 pyridyl))carbonylamino]-N-methylbutanamide
Compound 295 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3- boc fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-phenyl(3-
pyridyl))carboxamide
Compound 399
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
Compou pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-3-
Compou yl)(3-pyridyl))carboxamide
N-[((3R)(3-piperidyl))methyl](6-(2-cyanophenyl)-
2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(4-
Compou piperidylmethyl)carboxamide
Compound 505 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 506 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(3S)-1-(2-aminoacetyl)(3-piperidyl)]methyl}(6-
(2-cyanophenyl)-2-{[2-(3-
Compound 507 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 508 fluorophenyl)ethyl]amino}(3-pyridy!))carboxamide
Compound 509 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 510 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 553 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl 4-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3- pyridyl)carbonylamino]methyl}p(peridinecarboxyla
Compou te
N-[((2R)(2-piperidyl))methyl](6-(2-cyanophenyl)-
Compound 560 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 561 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 562 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-acetyl(2-piperidyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}piperidinecarboxyl
Compound 564 ate
Compound 597 fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-[((2R)-1-methyl(2-piperidyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 599 fiuorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 600 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 601 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 626 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 627 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-aminopropanoyl)(2- piperidyl)]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 628
N-{[(2R)-1-((2R)-2-aminopropanoyl)(2- piperidyl)]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-chloroethyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}piperidinecarboxyl
Compound 630 ate
2-aminoethyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}piperidinecarboxyl
Compound 631 ate
Compound 637 6-(2-cyanophenyl)(3-pyridyl))carboxamide
Compound 638 enylethyl)amino](3-pyridyl)}carboxamide
Compound 639 pyridyl)]amino}-N,N-dimethylpropanamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-
yl]methyl}{6-(2-cyanophenyl)-2-[(2-
Compound 673 phenylethyl)amino](3-pyridyl)}carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methylX6-(2-cyanophenyl)-2-[(2-(2H-3,4,5,6-
tetrahydropyran-4-yl)ethyl)amino](3-
Compound 674 pyridyl)}carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}{6-(2-cyanophenyl)-2-[(2-(2H-3,4,5,6-
tetrahydropyran-2-yl)ethyl)amino](3-
Compound 681 pyridyl)}carboxamide
Compound 689 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 690 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 691 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2- {[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
pyridyl))carboxamide
Compound 772
N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
Compound 773 pyridyl))carboxamide
N-{[(2R)-1 -((2S)-2-amino-3- hydroxypropanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-
2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-
Compound 774 yl(3-pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-aminopropanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
Compound 775 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
Compound 776 pyridyl))carboxamide
{6-(2-cyaπophenyl)-2-[(2-oxolan-2- ylethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 778 yl)(3-pyridyl))carboxamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}~2-[(tert-
Compound 779 butoxy)carbonylamino]acetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
Compound 780 aminoacetamide
Compound 787 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{(1 S)-2-[(5-fluoro-2~{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
Compound 808 aminoacetamide
N-{{1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
Compound 812 hydroxyacetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}-4-[(tert-
Compound 813 butoxy)carbonylamino]butanamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methy!pyrazol-4- yl)(3-pyridyl))carbonylamino]-isopropyl}-4-
aminobutanamide
Compound 814
Compound 830 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 831 pyrazol-4-y!(3-pyridyl))carboxamide
Compound 832 pyridyl))carboxamide
6 N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 833 yl)(3-pyridyl))carbonylamino]-isopropyl}acetamide
Compound 834 methy!pyrazol-4-yl)(3-pyridyl))carboxamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]-isopropyl}-2-
Compound 835 aminoacetamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
pyrazol-4-yl(3-pyridyl))-N-(2-
Compound 836 pyridylmethyl)carboxamide
Compound 837 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 847 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
Compound 848 (dimethylamino)acetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4- yl)(3-pyridyl))carbonylamino]-isopropyl}(2S)-2-
[(tert-butoxy)carbonylamino]-3-
Compound 849 hydroxypropanamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}(2S)-2-
Compound 850 amino-3-hydroxypropanamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4- yl)(3-pyridyl))carbonylamino]-isopropyl}-2-amino-
2-methylpropanamide
Compound 851
N-((1 R)-1-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amiπo}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]methyl}propyl)-2-
Compound 871 [(tert-butoxy)carbonylamino]acetamide
N-((1 R)-H[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino3methyl}propyl)-2-
Compound 872 aminoacetamide
N-((1 R)-1-{[(5-ftuoro-2-{[2-(3- fluorophenyi)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}propyl)-2-[(tert-
Compound 873 butoxy)carbonylamino]acetamide
N-((1 R)-1-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}propyl)-2-
Compound 874 aminoacetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3- fluorophenyl)ethylJamino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-isopropyl}(2S)-2-
Compound 877 aminopropanamide
Compound 878 4-yl)(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyιϊdyl)]methyl}(6-(2- cyanophenyI)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compou pyridyl))carboxamide
(6-(2-cyan oph eny l)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-
Compou N-(3-pyridylmethyl)carboxamide
{6-(3,4-difluorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compou pyridy lmethyl)ca rboxam ide
{6-(4-fluorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compou pyridylmethyl)ca rboxam ide
{6-(4-chlorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
pyridylmethyl)carboxamide
Compound 24
{6-(4-methylphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(4-cyanophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
{6-(4-methoxyphenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3- Compoun pyridylmethyl)carboxamide
Compound 18 pyridylmethyl)carboxamide
CwO {6-(3,4-dichlorophenyl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compound 13 pyridylmethyl)carboxamide
Compound 4 pyridylmethyl)carboxamide
methyl 4-{6-[(2~(2-pyridyl)ethyl)amino]-5-[N-(3-
Compound pyridylmethyl)carbamoyl]-2-pyridyl}benzoate
{6-[4-(dimethylamino)phenyl]-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compound pyridylmethyl)carboxamide
Compound 2 pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(3-pyridyl)ethyl)amino](3-pyridyl)}-
Compoun N-(3-pyridylmethyl)carboxamide
4-{6-[(2-(2-pyridyl)ethyl)amino]-5-[N-(3-
Compoun pyridylmethyl)carbamoyl]-2-pyridyl}benzoic acid
N-(3-pyridylmethyl){6-(3-pyridyl)-2-[(2-(2-
Compoun pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-(3-pyridylmethyl){6-(2-pyridyl)-2-[(2-(2-
Compoun pyridyl)ethyl)amino](3-pyridyl)}carboxamide
4-{6-[(2-(2-pyridyl)ethyl)amino]-5-[N-(3-
Compoun pyridyl methyl) ca rbam oy l]-2-pyridyl}benzamide
{2-[(2-hydroxy-2-phenylethyl)amino]-6-phenyl(3-
Compoun pyridyl)}-N-(3-pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-(2-vinylphenyl)(3-
Compoun pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-[2-(methoxymethyl)phenyl]-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 102 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)-2-hydroxyethyl]amino}-6- phenyl(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)-2- hydroxyethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-(1 ,3-thiazol-2-
Compoun yl)(3-pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-(1-methylpyrazol-4-yl)-2-[(2-(2- pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
[3-(aminomethyl)-6-phenyl(2-pyridyl)][2-(3-
Compou fluorophenyl)ethyl]amine
(6-(3,4-difluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{3-
Compou pyridylmethyl)carboxamide
(6-(5-cyano(2-thienyl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
Compound 274
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(5-formyl(2- th ienyl))(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-formyl(2- thienyl))(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-formyl(3- thienyl))(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
[2-{[2-(3-fluorophenyl)ethyl]amino}-6-(6-{[2-(3- fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))(3-pyridyl)]-N-
Compou (3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[2-
(hydroxymethyl)(3-thienyl)](3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
Compound 214 yl(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(5-acetyl(2-thienyl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyιϊdyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-[3-(aminomethyl)(2-thienyl)]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5-
(hydroxyethyl)(2-thienyl)](3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-(4-cyano(3-thienyl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
Compound 241 pyridylmethyl)carboxamide
Compound 306 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[5-
(hydroxymethyl)(3-pyridyl)]rnethyl}carboxamide
Compound 307
N-({5-[3,3-bis(dimethylamino)-2-azaprop-2- enyl](3-pyridyl)}methyl)(6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyljamino}(3-
Compound 314 pyridyl))carboxamide
Compound 315 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(3-{[(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}(2-
Compound 316 pyridyl))methyl]carboxamide
Compound 331 pyridyl))methyl](3-pyridylamino)carboxamide
(5-chloro-6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 368 py ridylmethy l)carboxam ide
(5-cyano-6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
N-[(2-chloro(3-pyridyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({2-[(2-aminoethyl)amino](3-pyridyl)}methyl)(6-
(2-cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 404 pyridylmethyl)carboxamide
Compound 408 piperidyl))methyl]carboxannide
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-6- oxo(2-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-
Compound 430 pyridyl))carboxamide
N-{[1 -(3-aminopropyl)-6-oxo(2- piperidyl)]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
flυorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 431
Compound 432 pyridyl))carboxamide
N-[((2S)-1-{2-[(tert- butoxy)carbonylamino]acetyl}(2-
piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compound 445 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 446 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)-1-{3-[(tert- butoxy)carbonylamino]propanoyl}(2-
piperidyl))nnethyl](6-(2-cyanopheπyl)-2-{[2-(3-
Compound 448 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 450 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 479 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-6-oxo(2-piperidyl))methyl](6-(2-
cyanophenyl)-2-{[2-(3-
Compound 511 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 6-oxo(2-piperidyl))methyl](6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 512 pyridyl))carboxamide
Compound 513 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 516 pyridyl))carboxamide
Compound 518 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl (3R)-3-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}piperidinecarboxyl
Compound 519 ate
N-[((3R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}(3- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 521
N-[((3R)-1-{3-[(tert- butoxy)carbonylamino]propanoyl}(3-
ρiperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compound 524 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((3R)-1-{4-[(tert- butoxy)carboπylamino]butanoyl}(3-
piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compound 527 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 529 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 532 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 533 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({(2R)-1-[4-(1 ,3-dioxobenzo[c]azolin-2-yl)butyl]- 6-oxo(2-piperidyl)}methyl)(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 535 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutyl)-6-oxo(2-
piperidyl)]methyl}(6-(2-cyanophenyl)-2-{j;2-(3-
Compound 536 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((3S,4S,2R,5R)-3,4,516-tetrahydroxy(2H- 3,4,5,6-tetrahydropyran-2-yl))methyl](6-(2-
cyanophenyl)-2-{[2-(3-
Compound 596 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 667 fluorophenyl)ethyl]amino}-2-pyridyl]benzamide
N-[2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3-
HΑrx O fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}pyrrolidinyl)(1 S)-1 ■
Compound 668 methyl-2-oxoethyl](tert-butoxy)carboxamide
Compound 669 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(1R)-2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}pyrrolidiπyl)-1-
Compound 670 methyl-2-oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2R)-2-aminoproρanoyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 671
Compound 672 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 696 yl)(3-pyridyl))carboxamide
N-({2-[(3-aminopropyl)amino](3- pyridyl)}methyl)(2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 699 py ridy I)) ca rboxam ide
N-({2-[(2-aminoethyl)amino](3-pyridyl)}methyl)(2-
{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 711 pyridyl))carboxamide
Compound 712 ate
Compound 713 yl)(3-pyridyl))carboxamide
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 714 yl)(3-pyridyl))carboxamide
Compound 717 pyridyl)}methyl)carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- ρyridyl))methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 745 pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-
{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 746 py ridy I) ) ca rboxa m ide
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 747 pyridyl))carboxamide
N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 748 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert- butoxy)carbonylamino]propanoyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carboxamide
Compound 749
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethylJamino}-6-pyrazol-4-yl(3-
Compound 750 py ridyl))carboxam id e
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-
fluoropheny0ethyl]amino}-6-pyrazol-4-yl(3-
Compound 751 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 752 pyridyl))carboxamide
Compound 753 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 757 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(5-fIuoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 758 py ridyl)) ca rboxam ide
N-(carbamoylmethyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compou pyridyl))carboxamide
Compound 760 methylacetamide
N-[((2R)-1-{(2S)-2-[(tert-butoxy)carbonylamino]-3- hydroxypropanoyl}pyrrolidin-2-yl)methyl](5-fluoro-
2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-
Compound 761 yl(3-pyridyl))carboxamide
N-{[(2R)-1 -((2S)-2-amino-3- hydroxypropanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-
2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-
Compound 762 yl(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(2-pyridyl)(3-
Compound 806 pyridyl))carboxamide
N-([2-(am[nomethyl)(3-pyridyl)]methyl}(5-fluoro-2-
{[2-(3-fluorophenyl)ethyl]amino}-6-(2-pyridyl)(3-
Compou pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))-N-(2-hydroxy-2-
methy lpropyl)carboxam id e
Compound 869
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
methylpyrazol-4-yl)(3-pyridyl))-N-(3-hydroxy-2,2-
Compound 870 dimethylpropyl)carboxamide
Compound 879 yl)(3-pyridyl))carboxamide
Compound 880 methylpyrazol-4-yl)(3-pyridyl))carboxamide
2-((2R)-2-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4- yl)(3-pyridyl))carbonylamino]methyl}azetidinyl)-2- b oxoethyl 2-[(tert-butoxy)carbonylamino]acetate
2-((2R)-2-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]methyl}azetidinyl)-2-
Compound 908 oxoethyl 2-aminoacetate
2-((2R)-2-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]methyl}azetidinyl)-2-
Compound 909 oxoethyl dimethyl phosphate
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5-
(hydroxymethyl)(2-thienyl)](3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
N-({2-[(3-aminopropyl)amino](3- pyridyl)}methyl)(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 695 py ridy lmethy l)ca rboxam ide
N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 715 yl)(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-4-methyl(3-pyridyl))-N-
Compoun (3-pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(6-
Compoun morpholin-4-yl(3-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(6-
pyrrolidinyl(3-pyridyl))methyl]carboxamide
Compound 217
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(6-
Compoun piperazinyl(3-pyridyl))rnethyl]carboxamide
N-{[6-(4-acetylpiperazinyl)(3-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(6-bromo(2-pyridyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridy!))carboxannide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5- (hydroxy methyl) (2-th ienyl)](3-
Compoun pyridyl))carbonylamino]-N-methylacetamide
N-(2-carbamoylethyl)(2-{[2-(3- fluorophenyl)ethyl]amino}-6-[5- (hyd roxym ethy l)(2-thieπy I)] (3-
Compoun pyridyl))carboxamide
Compound 268 pyridylmethyl)carboxamide
N-(carbamoyl methyl) (2-{[2-{3- fluorophenyl)ethyl]amino}-6-[5- (hydroxymethyl)(2-thienyl)](3-
Compou pyridyl))carboxamide
2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]-N-
Compou (methylsulfonyl)acetamide
N-{[3-(aminomethyl)cyclohexyl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 325 pyridyl)carbonylamino]cyclopentanecarboxamide
Compound 339 piperazinylethyl)carboxamide
Compound 340 pyrazin-2-ylethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amlno}(3-pyridyl))-N-(pyrazin-
2-ylm ethyl)ca rboxamide
Compound 341
N-((1S)-5-amino-1-carbamoylpentyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 350 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 351 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(4-amino-1-carbamoylbutyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1 R)-5-amiπo-1 -carbamoylpentyl)(6-(2- cyaπopheπyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 356 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({6-[(3-aminopropyl)amino](2-
pyridyl)}methyl)(6-(2-cyan.ophenyl)-2-{[2-(3-
Compound 357 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(4R)-4-carbamoyl-4-[(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]butyl}(phenylmethoxy)car
Compound 401 boxamide
N-((1 S)-4-amino-1 -carbamoylbutyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1 R)-4-amino-1 -carbamoylbutyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluoropheπyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))-N-
Compoun (pyrrolidin-3-ylmethyl)carboxamide
N-{[1-(2-aminoacetyl)pyrrolidin-3-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropanoyl)pyrrolidin-3- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 442
Compound 443 flυorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 444 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrrolidin-3-yl]methyl}(6-(2- cyanopheny!)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(3-amino-2-carbamoylpropyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 499 oxopiperazin-2-yl)methyl]carboxamide
Compound 500 yl)methyl]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-5- yl(3-pyιϊdyl))-N-[(6-oxopiperazin-2-
Compou yl)methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[4- (methylsulfonyl)-6-oxopiperazin-2-
Compou yl]methyl}carboxarnide
N-[(4-acetyl-6-oxopiperazin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 517 methyl-6-oxopiperazin-2-yl)methyl]carboxamide
Compound 581 [(methylamino)carbonylamino]ethyl}carboxamide
Compound 582 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)pyrrolidiη-2-yl)methyl](6-(2-cyanophenyl)-
Compound 584 pyridyl))carbonylamino]ethyl}acetamide
Compound 585 pyridyl))carbonylamino]ethyl}propanamide
Compound 586 pyridyl))carbonylamino]ethyl}butanamide
Compound 587 pyridyl))carbonylamino]ethyl}-N-methylacetamide
4-[(tert-butoxy)carbonylamino]-N-{2-[(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]ethyl}-N-
Compound 588 methylbutanamide
Compound 589 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropanoyl)pyrrolidin-2-
yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compound 590 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 591 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 592 pyridyl))carbonylamino]ethyl}-N-methylacetamide
3-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]ethyl}-N-
Compound 593 methylpropanamide
4-amino-N-{2-[(6-(2-cyanopheπyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]ethyl}-N-
Compound 594 methylbutanamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{2-
Compo [methyl(methylsulfonyl)amino]ethyl}carboxamide
3-amino-N-(2-aminoethyl)-2-{[(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}propanamide
Compound 632
Compound 633 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 634 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1S)-1-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}-2-hydroxyethyl)-
Compound 635 2-aminoacetamide
Compound 636 pyridyl))carbonylamino]propyl 2-aminoacetate
N-((1 R)-1-{[(6-(2-cyaπophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}-2-methylpropyl)-
Compound 643 2-hyd roxyacetam ide
N-((1R)-H[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carboπylamino]methyl}-2-methylpropyl)-
Compound 644 2-aminoacetamide
N-((2R)-2-amino-4-methylpentyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1R)-1-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}-3-methylbutyl)-2-
Compou hydroxyacetamide
N-((1R)-1-{[(6-(2-cyanophenyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}-3-methylbutyl)-2-
Compound 666 aminoacetamide
N-((1 R)-2-amino-1 -{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}ethyl)-2-
Compound 692 aminoacetamide
N-((1 R)-2-amino-1 -{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}ethyl)-2-
Compound 693 hydroxyacetamide
N-((3R)-2-oxo(3-piperidyl))(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compoun pyridyl))carboxamide
N-(carbamoylethyl)(6-(2-cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1R)-1-carbamoylethyl)(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compoun py ridy l))carboxam ide
methyl (2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]propanoate
methyl (2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]propanoate
Compound 133 pyridyl)carbonylamino]acetate
Compound 134 hydroxyethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(2-
Compou methoxyethyl)carboxamide
Compound 136 pyridyl))carboxamide
N-{2-[(tert-butoxy)-N- methylcarbonylamino]ethyl}(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 137 pyridyl))carboxamide
Compound 138 (methylamino)ethyl]carboxamide
Compound 139 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compoun pyridyl))carbonylamino]-N-methylpropanamide
(2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-N-methylpropanamide
Compound 292
Compound 291 pyridyl)carbonylamino]ethyl}acetamide
Compound 143 (methoxycarbonylamino)ethyljcarboxamide
Compound 144 [(methylsulfonyl)amino]ethyl}carboxamide ethyl 3-[(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}-3-
Compound 145 pyridyl)carbonylamino]propanoate methyl 4-[(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}-3-
Compound 287 pyridyl)carbonylamino]butanoate
Compound 286 pyridyl))carbonylamino]-N-methylpropanamide
4-[(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-
Compound 148 pyridyl))carbonylamino]-N-methylbutanamide
Compound 149 pyridyl))carboxamide
Compound 283 pyridyl)carbonylamiπo]butanoic acid
N-(2-aminoethyl)(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanoρhenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(4-
Compou fluorophenyl)methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(4-
Compou cyanophenyl)methyl]carboxamide
N-[(3,4-difluorophenyl)methyl](6-(2-cyanophenyl)-
Compound 279
Compound 155 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 167 hydroxypropyl)carboxamide
Compound 168 methoxypropyl)carboxamide
methyl (2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compoun pyridyl))carbonylamino]-3-hydroxypropanoate
Compound 170 pyridyl))carbonylamino]-3-hydroxypropanoate
(2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-3-hydroxy-N-
Compound 262 methylpropanamide
(2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]-3-hydroxy-N-
Compoun methylpropanamide
Compound 173 pyridylmethyl)carboxamide
(6-(2-cyanoρhenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(4-
Compou methylphenyl)methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(6-
Compou methyl(3-pyridyl))methyl]carboxamide
Compound 176 methyl(2-pyridyl))methyl]carboxamide
Compound 177 pyridyl)ethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(2-(3-
pyridyl)ethyl)carboxamide
Compound 178
N-[(6-amino(3-pyridyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethy!]amino}(3-pyridyl))-N-[(6-
OMe methoxy(3-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-
Compou methyl(3-pyridyl))methyl]carboxamide
N-[(2-amino(3-pyridyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-
Compou methoxy(3-pyridyl))methyl]carboxamide
Compound 200 hydroxy(2-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(1-
Compound 201 hydroxy(3-pyridyl))methyl]carboxamide
Compound 202 methylpiperazinyl)propyl]carboxamide
Compound 203 (cyclopropylmethyl)carboxamide
Compound 219 pyridyl))carbonylamino]acetamide
Compound 220 hydroxyethyl)acetamide
N-{3-[(tert-butoxy)carbonylamino]propyl}-2-[(6-(2- cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-
Compound 223 pyridyl))carbonylamino]acetamide
Compound 224 pyridyl))carbonylamino]acetamide
Compound 225 hydroxypropyl)acetamide
N-{2-[(tert-butoxy)-N-methylcarbonylamino]ethyl}- 2-[(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-
Compound 232 pyridyl))carbonylamino]acetamide
Compound 233 (methylamino)ethyl]acetamide
Compound 234 pyridyl))carbonylamino]propanoate
N-((1S)-2-amino-1-carbamoylethyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2S)-3-[(tert-butoxy)carbonylamino]-2-[(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]propanoic acid
(2S)-3-[(tert-butoxy)carbonylamino]-2-[(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]-N-methylpropanamide
(2S)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]-N-methylpropanamide
(2S)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]-N,N-
Compou dimethylpropanamide
(2S)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]propanoic acid
methyl (2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-
methyl (2S)-2-(acetylamino)-3-[(6-(2- cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]propanoate
N-((2S)-2-amino-2-carbamoylethyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(1 S)- 1 -carbamoyl-2-[(6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]ethyl}acetamide
(2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]-N-methylpropanamide
(2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]-N, N-
Compou dimethylpropanamide
(2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-
Compound 251 pyridyl))carbonylamino]propanoic acid
(2S)-2-(acetylamino)-3-[(6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-
Compoun pyridyl))carbonylamino]-N-methylpropanamide
methyl (2R)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-
(3-fIuorophenyl)ethyl]amino}(3-
Compoun pyridyl))carbonylamino]propanoate
N-((1 R)-2-amino-1 -carbamoylethyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 257 pyridyl))carbonylamino]-N-methylpropanamide
methyl (2R)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-
Compou pyridyl))carbonylamino]propanoate
N-((2R)-2-amino-2-carbamoylethyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(1 ,3-
oxazol-2-ylmethyl)carboxamide
Compound 260
N-[(6-cyano(3-pyridyl))methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-phenyl(3-
Compou pyridyl))carboxamide
N-{[6-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-phenyl(3-
Compou pyridyl))carboxamide
Compound 288 phenyl(3-pyridyl))ethan-1 -ol
Compound 293 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 297 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 298 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[4-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
fluorophenyl)ethyl]amino}-6-phenyl(3-
Compound 299 pyridyl))carboxamide
Compound 300 pyridyl))carboxamide
Compound 301 5-ylmethyl)carboxamϊde
Compound 302 hydroxyethyl]amino}acetamide
Compound 305 phenyl(3-pyridyl))-2-hydroxyethyl]amino}acetate
Compound 313 pyridyl))-2-[(2-pyridylmethyl)amino]ethan-1-ol
N-(4-aminobutyl)(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 317
Compound 318 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 319 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 320 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{2-[6-(aminomethyl)(2-pyridyl)]ethyl}(2-{[2-(3-
fluorophenyl)ethyl]amino}-6-phenyl(3- Compound 321 pyridyl))carboxamide
Compound 322 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 323 py ridyl))ca rboxam ide
N-[2-(2-aminoethoxy)ethyl](6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 327 pyridyl))carboxamide
Compound 342 (hydroxymethyl)(2-pyridyl)]methyl}carboxamide
Compound 343 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 347 pyridyl))carbonylamino]-N,N-dimethylacetamide
N-{2-[(2-aminoethyl)amino]ethyl}(6-(2- cyanophenyl)-2-{[2-(3-
Comp fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 355 piperidyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyrrdyl))-N-(pyrazol-
3-ylmethyl)carboxam ide
Compound 364
Compound 365 methylpyrazol-3-yl)rnethyl]carboxamide
Compound 366 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 367 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl 6-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3-
pyridyl)carbonylamino]methyl}pyridine-2-
Compound 376 carboxylate
6-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3-
pyridyl)carbonylamino]methyl}pyridine-2-
Compound 377 carboxylic acid
6-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3-
pyridyl)carbonylamino]methyl}pyridine-2-
Compound 378 carboxamide
W
Compound 379 methylpyrazol-5-yl)methyl]carboxamide
Compound 380 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 381 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(2-{2-oxo- ridyl}θthyl)c
Compound 383 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(aminomethyl)phenyl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyf]amino}(3-pyridyl))-N-[(2-
oxopyrrolidin-3-yl)methyl]carboxamide
Compound 397
Compound 415 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(2-aminoethyl)(3-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{2-[3-(aminomethyl)(2-pyridyl)]ethyl}(6-(2- , cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 418 pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[1-(2-
hyd roxyethy l)-2-oxo(3-
Compound 447 piperidyl)]methyl}carboxamide
Compound 449 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[1-(3-
diazo-3-azaprop-3-enyl)-2-oxo(3-
Compound 477 pi peridyl)]methy l}carboxam ide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[3-(3- diazo-3-azaprop-3-enyl)-2-oxo(3-
Compoun piperidyl)]methyl}carboxamide
N-[((2R)piperazin-2-yl)methyl](6-(2-cyaπophenyl)-
2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compoun pyridyl))carboxamide
N-[((2R)-1-methylpiperazin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-acetylpiperazin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 568 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide -(2-cyanophenyl)-2-{[2-(3- ]amino}(3- mino]methyl}piperazinecarboxyl
Compound 569
N-[((2S)-1-acetyl-4-methy1piperazin-2- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl (2S)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}-4-
Compound 571 methylpiperazineca rboxylate
Compound 572 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 573 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 574 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 575 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropanoyl)-4- methylpiperazin-2-yl]methyl}(6-(2-cyanophenyl)-
2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 576 pyridyl))carboxamide
Compound 577 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-4-(2-aminoacetyl)piperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 579 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-4-(4-aminobutanoyl)piperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{{(2S)-4-(2-aminoacetyl)-1-methylpiperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-4-(3-aminopropanoyl)-1- methylpiperazin-2-yl]methyl}(6-(2-cyanophenyl)- 2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carboxam«de
Compound 621
N-{[(2S)-4-(4-aminobutanoyl)-1-methylpiperazin-
2-yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)acetamide
N-[((2S)-1,4-diacetylpiperazin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)-4-methylpiperazin-2-yl)methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluoropheπyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 640 pyridyl))carboxamide
methyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}azetidinecarboxyla
Compound 641 te
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))-N-[(2-
Compou oxopyrrolidin-3-yl)methyl]carboxamide
Compound 646 yl)methyl]carboxamide
Compound 647 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
methyl 4-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}pyrrolidinyl)-4-
Compound 648 oxobutanoate
Compound 649 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 650 methylpyrazol-4-yl)(3-pyridyl))carboxamide
methyl 4-((2R)-2-{[(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4- yl)(3-pyridyl))carbonylamino]methyl}pyrrolidinyl)-
4-oxobutanoate
Compound 651
Compound 652 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 653 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 654 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 655 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 656 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 657 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2-
yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
Compound 658 f luoropheny l)ethyl]a m i no}(3-py ridyl))carboxam ide
methyl 4-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}azetidinyl)-4-
Compound 659 oxobutanoate
Compound 660 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(azaperhydroepin-2-ylmethyl)(6-(2- cyanophβnyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 662 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 663 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(4-aminobutanoyl)azaperhydroepin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-{3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 664
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-2- oxopyrrolidin-3-yl)methyl](6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 675 pyridyl))carboxamide
Compound 676 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-2- oxopyrrolidin-3-yl)methyl](2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 677 pyridyl))carboxamide
Compound 678 pyrazol-4-yl(3-pyridyl))carboxamide
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-2- oxopyrrolidin-3-yl)methyl](2-{[2-(3-
fluorop he ny l)ethy l]amino}-6-( 1 -m ethylpyrazol-4-
Compound 679 y I) (3-pyιϊdyl))car boxam ide
Compound 680 methylpyrazol-4-yl)(3-pyridyl))carboxamide
tert-butyl (2R)-2-{[(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}pyrrolidinecarboxyl
Compound 682 ate
Compound 683 pyrazol-4-yl(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyrϊdyl))-N-[(7-
Compou oxoazaperhydroepin-2-yl)methyl]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4- yl(3-pyridyl))-N-[(7-oxoazaperhydroepin-2-
Compou yl)methyl]carboxamide
Compound 686 oxoazaperhydroepin-2-yl)methyl]carboxamide
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-7- oxoazaperhydroepin-2-yl)methyl](6-(2-
cyanophenyl)-2-{[2-(3-
Compound 687 fluoropheπyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)-7-oxoazaperhydroepin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 688
Compound 694 pyrazol-4-yl(3-pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2- yl]methyl}(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5-
(hydroxymethyl)(2-thienyl)](3-
Compound 697 pyridyl))carboxamide
Compound 698 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
tert-butyl (2R)-2-{[(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}azetidinecarboxyla
Compound 700 te
N-[((2R)azetidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compou pyridyl))carboxamidθ
N-[2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}azetidinyl)(1S)-1-
(hydroxymethyl)-2-oxoethyl](tert-
Compound 702 butoxy)carboxamide
W
Compound 703 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(1R)-2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}azetidinyl)-1-
(hydroxymethyl)-2-oxoethyl](tert-
Compound 704 butoxy)carboxamide
Compound 705 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}azetidinyl)(1S)-1-
(3-aminopropyl)-2-oxoethyl](tert-
Compound 706 butoxy)carboxamide
Compound 707 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 708 pyrazol-4-yl(3-pyridyl))carboxamide
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}azetidin-2- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-
pyrazol-4-yl(3-pyridyl))carboxamide
Compound 709 •
N-{[(2R)-1-(2-aminoacetyl)azetidin-2-yl]methyl}(2-
{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 710 pyridyl))carboxamide
Compound 718 pyridyl))carboxamide
N-({6-[(3-aminopropyl)amino](2- pyridyl)}methyl)(2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 719 pyridyl))carboxamide
N-({6-[(2-a m inoethyl)a m ino] (2-py ridyl)}methyl)(2-
{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 720 pyridyl))carboxamide
Compound 721 pyridyl)}methyl)carboxamide
methyl (2R)-2-{[(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}azetidinecarboxyla
Compound 722 te
N-[((2R)-1-{3-[(tert- butoxy)carbonylamino]propanoyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 723 y I) (3-py ridy l))carboxam ide
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fIuorophenyl)ethyl]amino}-6-(1-methylpyra2oI-4-
Compound 724 yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 725 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 726 yl)(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 727 yl)(3-pyridyl))carboxamide
Compound 728 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrro(idin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carboxamide
Compound 729
N-{[(2 R)-1 -(3-am inopropy l)-5-oxopyrrol id i n-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 730 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(2,3-dihydroxypropanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 731 yl)(3-pyridyl))carboxamide
Compound 732 pyridylmethyl)carboxamide
tert-butyl (2R)-2-{[(6-(2-cyaπopheπyl)-5-fluoro-2- {[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]methyl}pyrrolidinecarboxyl
Compound 733 ate
N-[((2 R)pyrrolid i n-2-yl)methy I] (6-(2-cyanopheny I)-
5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 734 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](2-{[2-(3-
fluorophenyl)ethyl]amino}-6-[1-(2-
Compound 735 hydroxyethyl)pyrazol-4-yl](3-pyridyl))carboxamide
"0V5 N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-[1-(2-
Compound 736 hydroxyethyl)pyrazol-4-yl](3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-
Compound 737 pyridyl))carboxamrde
Compound 738 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}pyrrolidin-2-
yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
Compound 739 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 740 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert- butoxy)carbonylamino]propanoyl}pyrrolidin-2-
yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
Compound 741 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 742
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2-
yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
Compound 743 fluorophenyl)ethyl]amino}(3-pyridyJ))carboxamide
Compound 744 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-5-
fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 754 pyridyl))ca rboxam ide
Compound 755 fiuorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 756 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide luoro-2-{[2-(3- ]amino}-6-(1-methylpyrazol-4- mino]methyl}pyrrolidiπyl)(1S)-1-
Comp
](tert-butoxy)carboxamide
N-{[(2R)-1-((2S)-2-aminopropanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 765 yl)(3-pyridyl))carboxamide
N-[((2R)-H(2S)-2,5-bis[(tert- butoxy)carbonylamino]pentanoyl}pyrrolidin-2- ό~ yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 766 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-((2S)-2,5-diaminopentanoyl)pyrrolidin- 2-yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 767 yl)(3-pyridyl))carboxamide ol-4- 1S)-1-
N-{[(2R)-1-((2S)-2-amino-3- hydroxypropanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-
2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(1-
Compound 769 methylpyrazol-4-yl)(3-pyridyl))carboxamide
methyl 4-((2R)-2-{[(5-fIuoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]methyl}pyrrolidinyl)-
Compound 770 4-oxobutanoate
N-{[(2R)-1-(4-hydroxybutanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carboxamide
Compound 771
N-[(2-{[(tert-butoxy)carbonylaminojmethyl}(3- pyridyl))methyl](5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazo!-4-
Compound 781 yl)(3-pyridyl))carboxamide
Compound 782 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[3-((5R)-5-{[(5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]methyl}-2-
Compound 783 oxopyrrolidinyl)propyl](tert-butoxy)carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 784 yl)(3-pyridyl))carboxamide
l-4- boxyl
Compound 786 yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 788 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}(5-cyano-2-{|;2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 789 yl)(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](2-{[2-(3-
fluorophenyl)ethyl]amino}-5-methyl-6-(1-
Compound 790 methylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 791 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidiπ-2-yl)methyl](2-{[2-(3-
fluorophenyl)ethyl]amino}-5-methyl-6-(1-
Compound 792 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(2-{[2-(3-fluorophenyl)ethyl]amino}-5-
methy l-6-( 1 -methylpyrazol-4-y I) (3-
Compound 795 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyI](2-{[2-(3-fluorophenyl)ethyl]amino}-5-
methyl-6-(1-methylpyrazol-4-yl)(3-
Compound 796 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyI)pyrrolidin-2- yl]methyl}(2-{[2-(3-fluorophenyl)ethyl]amino}-5-
methyl-6-(1-methylpyrazol-4-yl)(3-
Compound 797 pyridyl))carboxamide
tert-butyl (2R)-2-{[(5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}pyrrolidinecarboxyl
Compound 798 ate
Compound 799 pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 800 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}(5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 801 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamiπo]propyl}- 5-oxopyrrolidin-2-yl)methyfJ(5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 802 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 803 pyridyl))carboxam ide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](5-cyano-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compound 804 pyridyl))carboxamide
Compound 805 yl(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}-5-methyl(3-
Compound 809 pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-
5-methyl(3-pyridyl))carboxamide
Compound 810
N-[3-<(5R)-5-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-5-methyl(3-
pyridyl))carbonylamino]methyl}-2-
Compound 811 oxopyrrolidinyl)propyl](tert-butoxy)carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}-5-methyl(3-
Compound 815 pyridyl))carboxamide
Compound 816 yl)pyridine-3-carboxylate
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3- pyridyl))methyl](5-chloro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 817 yl)(3-pyridyl))carboxamide
Compound 818 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}- 5-oxopyrrolidin-2-yl)methyl](5-chloro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 819 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yllmethyl}(5-chloro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 820 yl) (3-pyridyl)) ca rlDoxamide
ylpyrazol-4- lidinecarboxyl
Compound 822 yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamiπo]butanoyl}pyrrolidin-2- yl)methyl](5-chloro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 823 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}(5-chloro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 824 yl)(3-pyridyl))carboxamide
Compound 826 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-(3-aminopropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carboxamide
Compound 827
Compound 828 yl)(3-pyridyl))carbonylamino]-2-methylpropanoate
(2R)-3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-2-methylpropanoic
Compound 829 acid
2-[(tert-butoxy)carbonylamino]-N-{3-[(5-fluoro-2- {[2-(3-fluorophenyl)ethyl]amino}-6-(1-
methylpyrazol-4-yl)(3-
Compound 838 pyridyl))carbonylamino]propyl}acetamide
Compound 839 yl)(3-pyridyl))carbonylamino]propyl}acetamide
Compound 840 pyridyl)carbonylamino]propyl}acetamide
Compound 841 4-yl)(3-pyridyl))carboxamide
N-((2R)-2-carbamoylpropyl)(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 842 yl)(3-pyridyl))carboxamide
(2R)-3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-2-methyl-N-
Compound 843 methylpropanamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))-N-(pyrazol-3-
Compou ylmethyl)carboxamide
Compound 845 ylmethyl)carboxamide
Compound 853 rnethylpyrazol-4-yl)(3-pyridyl))carboxamide
Compound 854 ketone
N-((2S)-3-amino-2-methylpropyl)(5-fluoro-2-{[2-
N-{(2R)-3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-2-methylpropyl}-2-
Compound 856 [(tert-butoxy)carbonylamino]acetamide
N-{(2R)-3-[(5-fluoro-2-{[2-(3- fluoropheny0ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-2-methylpropyl}-2-
Compound 857 aminoacetamide
N-{(2R)-3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carbonylamino]-2-methylpropyl}-2-
Compound 858 hyd roxyaceta m ide
» a pyridyl))carbonylamiπo]methyl}azetidinecarboxyla
Compound 862 te
Compound 863 yl)(3-pyridyl))carboxamide
N-[((2R)-1-{2-[(tert- butoxy)carbonylamino]acetyl}azetidin-2- yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 864 yl)(3-pyridyl))ca rboxam ide
N-{[(2R)-1-(2-aminoacetyl)azetidin-2-yl]methyl}(5-
fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
Compound 865 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert- butoxy)carbonylamino]butanoyl}azetidin-2- yl)methyl](5-fJuoro-2-{[2-(3-
fluorophenyl)ethyl]arnino}-6-(1-methylpyrazol-4-
Compound 866 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)azetidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 867 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 868 yl)(3-pyridyl))carboxamide
Compound 875 yl)(3-pyridyl))carbonylamino]-2-methylpropanoate
Compound 876 4-yl)(3-pyridyl))carboxamide
N-((2R)-3-hydroxy-2-methylpropyl)(5-fluoro-2-{[2-
Compound 882 pyridyl))carbonylamino]-2-methylpropanoate
Compound 883 pyridyl))carbonylamino]-2-methylpropanoate
N-((2R)-3-hydroxy-2-methylpropyl)(6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}{3-pyridyl))carboxamide
N-[(2,2-dimethyl(1,3-dioxan-5-yl))methyl3(5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
Compou methylpyrazol-4-yl)(3-pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluoroρhenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))-N-[3-hydroxy-2-
Compou (hydroxymethyl)propyl]carboxamide
Compound 887 pyrazol-4-yl(3-pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))-N-[3-hydroxy-2-
Compou (hydroxymethyl)propyl]carboxamide
N-[(2,2-dimethyl(1,3-dioxaπ-5-yl))methyl](6-(2- cyanophenyl)-2-{[2-(3-
Compou fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[3-
Compou hydroxy-2-(hydroxymethyl)propyl]carboxamide
methyl (2R)-2-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compou yl)(3-pyridyl))carbonylamino]methyl}i3utanoate
N-((2R)-2-ethyl-3-hydroxypropyl)(5-fluoro-2-{[2-
(3-fluoropheny[)ethyl]amino}-6-(1-methylpyrazol-
Compou 4-yl)(3-pyridyl))carboxamide
methyl (2R)-2-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
Compou pyridyl))carbonylamino]methyl}butanoate
N-((2R)-2-ethyl-3-hydroxypropyl)(5-fluoro-2-{[2-
Compound 894
methyl (2R)-2-[(tert-butoxy)carbonylamino]-3-[(5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-
Compou pyridyl))carbonylamino]propanoate
methyl (2R)-2-amino-3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazoI-4-
Compou yl)(3-pyridyl))carbonylamino]propanoate
N-((1 R)-1-{[(5-fluorp-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4- yl)(3-pyridyl))carbonylamino]methyJ}-2-
Compou hydroxyethyl)(tert-butoxy)carboxamide
N-((2R)-2-amino-3-hydroxypropyl)(5-fluoro-2-{[2- (3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-
Compou 4-yl)(3-pyridyl))carboxamide
N-((1 R)-H[(5-fiuoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3- pyridyl))carbonylamino]methyl}-2-
Compou hydroxyethyl)(tert-butoxy)carboxamide
Compound 900 pyridyl))carboxamide
rr N-(2,3-dihydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
Compound 901 yl)(3-pyridyl))carboxamide
N-(2,3-dihydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyf]amino}-6-pyrazol-4-yl(3-
Compou pyridyl))carboxamide
Compound 903 methylpropanoate
N-{[(2R)-1 -(2-hydroxy-2- methylpropanoyl)azetidin-2-yl]methyl}(5-fluoro-2-
{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
Compound 904 methylpyrazol-4-yl)(3-pyridyl))carboxamide
tert-butyl (2R)-2-{[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carbonylamino]methyl}azetidinecarboxyla
Compound 905 te
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2- yl]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrazoI-4-yl(3-
Compound 906 pyridyl))carboxamide
N-[((2R)azetidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
pyridyl))carboxamide
Compound 907
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyljamino}-6-phenyl(3-
Compoun pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-
Compoun fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 126 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 127 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
Compoun pyridyl))carbonylamino]-N-methylacetamide
Compound 396 pyridyl))carboxam ide
[0125] Provided is at least one chemical entity chosen from compounds of Formula V
Formula V and pharmaceutically acceptable salts thereof wherein
R1 is selected from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl,
Z2 is selected from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is selected from optionally substituted alkyl and optionally substituted heterσaralkyl;
R3 is chosen from hydrogen and optionally substituted alkyl; and
R6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
[0126] In certain embodiments of compounds of Formula V1 Z1 is chosen from phenyl substituted with one, two or three groups chosen from halo, alkyl, alkoxy, cyano, and optionally substituted acyl. In certain embodiments of compounds of Formula V1 Z1 ischosen from 3,4- difluorophenyl and 2-cyanophenyl.
[0127] In certain embodiments of compounds of Formula V, Z2 is chosen from methylaminocarbonyl, methylaminocarbonylmethylaminocarbonyl, ethylaminocarbonyl, propylaminocarbonyl, and isopropylaminocarbonyl, wherein the methyl, ethyl, propyl, and isopropyl group is optionally substituted with one, two or three groups independently selected from optionally substituted methoxy, optionally substituted ethoxy, optionally substituted phenyl, and optionally substituted pyridinyl. In certain embodiments of compounds of Formula V1 Z2 is chosen from methylaminocarbonylmethylaminocarbonyl, 2-methoxyeth-1-ylaminocarbonyl, benzylaminocarbonyl, isopropylaminocarbonyl, pyridin-3-ylmethylaminocarbonyl, and pyridin-4- ylmethylaminocarbonyl.
[0128] In certain embodiments of compounds of Formula V, Z3 is chosen from ethyl, methyl and propyl substituted with one, two or three groups independently selected from optionally substituted aryl and optionally substituted heteroaryl. In certain embodiments of compounds of Formula V, Z3 is chosen from ethyl and methyl substituted with a group selected from optionally substituted phenyl and optionally substituted pyridinyl. In certain embodiments of compounds of Formula V, Z3 is chosen from 2-(3-fluorophenyl)ethyl, 2-(3-methylphenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(3-methylpheny!)ethyl and pyridin-2-ylmethyl.
[0129] In certain embodiments of compounds of Formula V, R1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula V1 R1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula V, R1 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula V1 R1 is hydrogen.
[0130] Mn certain embodiments of compounds of Formula V, R3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula V. R3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula V, R3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula V, R3 is hydrogen.
[0131] In certain embodiments of compounds of Formula V1 Rβ is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula V, Rβ is chosen from hydrogen, cyano,
chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula V, RB is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl. In certain embodiments of compounds of Formula V, R6 is hydrogen. [0132] In certain embodiments, the compound of Formula V is chosen from
COMPOUND CHEMICAL NAME
(6-(2-cyanophenyl)-4-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound 0 pyrϊdylmethyl)carboxamide
2-l(6-(2-cyaπophenyl)-4-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 1 pyridyl))carbonylamino]-N-methylacetarnide
Compound 2 pyridylmethyl)amino]methyl}phenyl)(3-pyridyl)]-N-(3-pyridytmethyl)carboxamide
[0133] Provided is at least one chemical entity chosen from compounds of Formula Vl
Formula Vl
and pharmaceutically acceptable salts thereof wherein
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is chosen from optionally substituted cycloalkyl and optionally substituted heterocycloalkyl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from halo and optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
[0134] In certain embodiments of compounds of Formula Vl1 Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
[0135] In certain embodiments of compounds of Formula Vl1 Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
[0136] In certain embodiments of compounds of Formula Vl, Z2 is chosen from (2-(N- acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1 -ylcarbonyl; 2-
(methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-yl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
[0137] In certain embodiments of compounds of Formula Vl, R3 is chosen from hydrogen and optionally substituted lower alkyl.
[0138] In certain embodiments of compounds of Formula Vl, R3 is chosen from hydrogen and lower alkyl.
[0139] In certain embodiments of compounds of Formula Vl, R3 is chosen from hydrogen and methyl.
[0140] In certain embodiments of compounds of Formula Vl, R3 is hydrogen.
[0141] In certain embodiments of compounds of Formula Vl, Z2 is -C(O)NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
[0142] In certain embodiments of compounds of Formula Vl, R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl.
[0143] In certain embodiments of compounds of Formula Vl, R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
[0144] In certain embodiments of compounds of Formula Vl1 R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl.
[0145] In certain embodiments of compounds of Formula Vl1 R4 is hydrogen.
[0146] In certain embodiments of compounds of Formula Vl, R5 is chosen from hydrogen and lower alkyl.
[0147] In certain embodiments of compounds of Formula Vl, R5 is chosen from hydrogen and methyl
[0148] In certain embodiments of compounds of Formula Vl, R5 is hydrogen.
[0149] In certain embodiments of compounds of Formula Vl, R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0150] In certain embodiments of compounds of Formula Vl, R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl,
optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0151] In certain embodiments of compounds of Formula Vl, R2 is chosen from (1-(2- aminoethyl)-1H-pyrazol-3-yl)methyI; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyt; (1 H-imidazol-2-yl)methyl;
(1 H-pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-3-yl)methyl; (1 -methyl-1 H-pyrazol-5-yl)methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- arninoethylarnino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1 -(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1 -(aminocarbonyl)-4-(amino)-butyl; 1 -(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1 - (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 1 -(hydroxy)-2-(aminocarbonylmethylamino)eth-1 -yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1-(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; 1-aminocarbonyl-2-hydroxy-eth-1 -yl; 1-methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1-yl; 2-
(methylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(methylsulfonamido)ethyl; 2-(rηethyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amiπo-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-mβthoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridiπ-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylamiπocarbonyl)prop-i-yl; 3-(amiπocarboπyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; S-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxypropyl; A- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; A- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; A- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridiπ-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1 -yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
[0152] In certain embodiments of compounds of Formula Vl, Z3 is chosen from halo and optionally substituted lower alkyl.
[0153] In certain embodiments of compounds of Formula Vl1 Z3 is chosen from halo, optionally substituted methyl, optionally substituted ethyl, and optionally substituted propyl wherein the methyl, ethyl and propyl groups are optionally substituted with one or two groups
selected from hydroxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, and optionally substituted aryloxy.
[0154] In certain embodiments of compounds of Formula Vl1 Z3 is chosen from 2-(3- methylphenyl)ethyl, (tetrahydrofuran-2-yl)methyl, 1-hydroxycyclohexylmethyl, 2-(1 ,3-dioxolan-2- yl)ethyl, 2-(1 H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(1-methylpiperidin-4- yl)ethyl, 2-(2,3-difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(2-oxoimidazolidiπ-1-yl)ethyl, 2-(2- oxopiperidin-1-yl)ethyl, 2-(2-oxopyrrolidin-1-yl)ethyl, 2-(3-(trif!uoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1H-pyrazol-1-yl)ethyl, 2-(3,5- dimethyl-1 H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fluorophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydrσxyphenyl)ethyl, 2-(benzo[d][1 ,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-cyclohex-1-enylethyl, 2-cyclohexylethyl, 2-morpholinoethyl, 2-phenoxyethyl, 2-phenylprop-1- yl, 3-(1H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, chloro, isopentyl, propyl, pyridin-2- ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
[0155] In certain embodiments of compounds of Formula Vl, Z3 is chosen from 2-(3- fluorophenyl)ethyl and 2-(pyridin-2-yl)ethyl.
[0156] In certain embodiments of compounds of Formula Vl, Z1 is chosen from cycloalkyl and heterocycloalkyl, where each group is optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
[0157] In certain embodiments of compounds of Formula Vl, Z1 is chosen from cycloalkyl and heterocycloalkyl, where each group is optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl,
optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
[0158] In certain embodiments of compounds of Formula Vl, Z1 is chosen from 1-(2H)-(methoxycarbonyl)-3,4-dihydropyridin-4-yl; 1-(2H)-(methoxycarbonyl)-5,6-dihydropyridin-4-yl; 1-(2H)-(methoxymethyl)-5,6-dihydropyridin4-yl; 1-(2H)-(t-butoxycarbonyl)-5,6-dihydropyridin-4-yl; 1-(2H)-5,6-dihydropyridin-4-yl; 1-(acetyl)-4,5-dihydroimidazo-2-yl; 1-(dimethylcarbamoyl)-1 ,2,3,6-tetrahydropyridin-4-yl; 1-(methoxymethyl)-4,5-dihydroimida2O-2-yl; 1-cyano-4,5-dihydro-1H-imidazol-2-yl; 1 -methyl-1 ,2,3,6-tetrahydropyridin-4-yl; 2-(acetylaminomethyl)pyrrolidin-1-yl; 2-(hydroxymethyl)morpholino; 2-(hydroxymethyl)pyrrolidin-1-yl; 2-(methoxymethyl)pyrrolidiπ-1-yl; 2H-pyrrol-1(5H)-yl; 2-hydroxymorpholino;
2-methyl-4-(t-butoxycarbonyl)-piperazin-1-yl; 2-oxo-5-hydroxy-piperidinyl; 2-oxopiperazinyl; 2-oxopiperidinyl; 2-oxopyrrolidinyl;
3-(3,3-dimethylureido)pyrrolidin-1-yl; 3-(aminomethyl)-pyrrolidin-1-yl; 3-(carboxymethyl)-pyrrolidinyl; 3-(dimethylamino)pyrrolidin-1-yl; 3-(dimethylaminomethyl)-pyrrolidin-1-yl; 3-(hydroxymethyl)pyrrolidin-1-yl; 3-(methoxycarbonylamino)-pyrrolidinyl; 3-(methylsulfonyl)pyrrolidin-1-yl; 3-(N-acetyl-N-methyl)-aminomethyl)-pyrrolidin-1-yl;
-(pyridin-4-yl)pyrrolidin-1 -yl; -(t-butoxycarbonylamino)-pyrrolidinyl;
3-(trifluoromethyl)pyrrolidin-1-yl;
3,3-difluoropyrrolidinyl;
3-acetylaminopyrrolidinyl;
3-aminopyrrolidin-1 -yl;
3-fluoropyrrolidin-1 -yl;
3-hydroxymethylpyrrolidin-1-yl;
3-hydroxypyrrolidin-1 -yl;
3-methoxymethylpyrrolidin-1 -yl;
3-methylamino-pyrrolidinyl;
3-oxopiperazin-1 -yl; -(acetyl)-piperazin-1 -yl; -(aminomethyl)piperidin-1-yl; -(ethoxycarbonyl)-piperazin-1-yl;
4-(ethoxycarbonyl)-piperidinyl;
4-(methoxycarbonyl)-piperazin-1-yl;
4-(methylsulfonyl)cyclohex-1-enyl;
4-(N, N-dimethylaminocarbonyl)-piperazin-1 -yl;
4-(t-butoxycarbonylamino)-piperidin-1-yl;
4-(t-butoxycarbonylaminomethyl)-piperidin-1-yl;
4,5-dihydro-1 H-imidazol-2-yl;
4,5-dihydrooxazol-2-yl;
4-hydroxy-4-phenylpiperidin-1-yl;
4-hydroxyethyl-piperidin-yl;
4-hydroxym ethyl-pi perid i n-yl ;
4-hydroxypiperidinyl;
4-methylaminocarboπyl-piperidiπyl;
4-methylpiperazin-1-yl;
4-methylpiperidinyl;
4-oxocyclohex-1 -enyl;
5,6-dihydropyridin-i (2H)-yl; azetidinyl;
cyclohex-1 -enyl; cyclohexyl; cyclopent-1-enyl; cyclopentyl; morpholino; piperazinyl; piperidinyl; pyrrolidinyl; and thiomorpholino.
[0159] In certain embodiments of compounds of Formula Vl, R1 is chosen from hydrogen and optionally substituted lower alkyl.
[0160] In certain embodiments of compounds of Formula Vl, R1 is chosen from hydrogen and lower alkyl.
[0161] In certain embodiments of compounds of Formula Vl, R1 is chosen from hydrogen and methyl.
[0162] In certain embodiments of compounds of Formula Vl, R1 is hydrogen.
[0163] In certain embodiments, the compound of Formula Vl is chosen from COMPOUND Chemical Name
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-piperidyl(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
N-{[(3S)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
Compou yl]methyl}acetamide
Compound 2 (carbamoylmethyl)carboxamide
W
2-[(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
Compoun methylacetamide
Compound 4 dimethylcarboxamide
N-(carbamoylmethyl)(2-{[2-(3-fluorophenyl)ethyl]amino}-6-
Compou pyrrolidinyl(3-pyridyl))carboxamide
(6-[(3R)-3-(hydroxymethyl)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-{(3S)-3-[(methylamino)methyl]pyrrolidiπyl}-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
N-{[(3R)-1-(6-{[2-(3-fiuorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-yl]methyl}-N-
Compou methylacetamide
(6-{(3S)-3-[(dϊmethylamino)methyl]pyrrolidinyl}-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
Compound 9
(2-{[2-(3-fluorophenyl)ethyljamino}-6-(2-oxopiperidyl)(3-
Compoun pyridyl))-N-(3~pyridylmethyl)carboxamide
Compound 11 pyridylmethyl)carboxamide
Compound 12 pyridyl))carboxamide
)ethyl]amino}-6-
Compound 15 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-pyridyl))-N-
Compou (3-pyridylmethyl)carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridy!))-N-(2-
Compou carbamoylethyl)carboxamide
(6-((3S)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[4-
(hydroxymethyl)piperidyl](3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(5-hydroxy-2-
Compou oxopiperidyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-oxopiperazinyl)(3-
Compou pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-methylpiperazinyl)(3-
pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 22
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[4-(2- hydroxyethyl)piperidyl](3-pyridyl))-N-(3-
Compoun pyridylmethyljcarboxamide
tert-butyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))-3-
Compoun oxopiperazlnecarboxylate
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-oxopiperazinyl)(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-oxopiperidyl)(3-
Compoun pyridyl))-N,N-dimethylcarboxamide
Compound 27 pyridylmethyl)carboxamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compound 28 pyridyl))carbonylamino]-N-methylacetamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-pyrrolinyl)(3-pyridyl))-
Compou N-(3-pyridylmethyl)carboxarnide
(6-((3S)-3-aminopyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
N-[(3R)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
Compou yl](dimethylamino)carboxamide
Compound 32 (3-pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-pyrrolidinyl(3-pyridyl)}-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 34 N-(3-pyridylmethyl)carboxam ide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1,3-oxazolin-2-yl)(3-
pyridyl))-N-(3-pyridy!methyl)carboxamide
Compound 35
<6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-(aminomethyl)(3- Compou pyridyl)]methyl}carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(6-cyclopent-1-eπyl-2-
Compou {[2-(3-fIuorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-((3S)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl)amino}(3-pyridyl))-N-{[2-(aminomethyl)(3-
Compou pyridyl)]methyl}carboxamide
(6-((3R)-3-fluoropyrrolidinyl)-2-{[2-{3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-(aminomethyl)(3-
Compou pyridyl)]methyl}carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3-
Compou fIuorophenyl)ethyl]amino)-6-piperidyl{3-pyridyl))carboxamide
Compound 41 pyridyl))carboxamide
N-[(3S)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
Compoun yljmethoxyca rboxam ide
methyl 4-(6-{[2-( 3-fIuorophenyl)ethyl]amino}-5-{N-[(N- methylcarbamoyl)methyl]carbamoyl}-2-pyridyl)-1 , 2,5,6-
Compoun tetrahydropyridiπecarboxylate
N-[(3R)-1-(6-{[2-(3-fIuorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
Compoun yl]methoxycarboxamide
{6-piperidyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-<3-
Compou pyridylmethyl )ca rboxam ide
(6-((3R)-3-aminopyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-{1 ,2,5,6-
Compoun tetrahydropyridyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(5-chloro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 48
(5-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
methyl 2-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
Compoun pyridylmethyl)carbamoyl]-2-pyridyl)-2-imidazolinecarboxylate
Compound 51 pyridylmethyl)carboxamide
N-[[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(5-
fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compound 52 pyridyl))carboxamide
(6-((3R)-3-fluoropyrrolidinyl)-5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-(aminomethyl)(3-
Compound 53 pyrιdyl)]methyl}carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)-5-fluoro-2-{[2-(3-
fluorophenyl)ethylJamino}(3-pyridyl))-N-[(2-{[(tert-
Compound 54 butoxy)carbonylamino]methyl}(3-pyridyl))methyl]carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)~5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}{3-pyridyl))-N-{[2-(aminomethyl)(3-
Compou pyridyl)]methyl}carboxamide
(6-(1-acetyl(4-1 ,2,5,6-tetrahydropyridyl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-1 ,2,5,6-
Compou tetrahydropyridyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
tert-butyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
pyridylmethyl)carbamoyl]-2-pyridyl)-1 ,2,5,6-
Compound 58 tetrahydropyridinecarboxylate
<6-(1,4-dioxaspiro[4.5]dec-7-en-8-yl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
methyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl]-2-pyridyl)-1,2,5,6-
tetrahydropyridinecarboxylate
Compound 61
(6-cyclopent-1-enyl-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 62 pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
pyrrolidinyl(3-pyridyl))carbonylarnino]-isopropyl}-2-[(tert-
Compound 63 butoxy)carbonylamino]acetatπide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-{4-oxocyclohex-1-
Compound 64 enyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-((3R)-3-fluoropyrrolidinyl)-5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-{[(tert-
Compound 65 butoxy)carbonylamino]methyl}(3-pyridyl))methyl]carboxamide
(6-cyclohexyl-2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))-N-
Compound 66 (3-pyridylmethyl)carboxamide
Compoun
N-[((2R)-1-{3-[(tert-butoxy)carbonylamtno]propyl}-5- oxopyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compound 68 pyridyl))carboxamide
{6-[1-(N,N-dimethylcarbamoyl)(4-1,2,5,6-tetrahydropyridyl)]-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridytmethyl)carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compoun pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1 -methy!(4-1 ,2,5,6-
Compoun tetrahydropyridyl))(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
methyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl]-2-pyridyl)-1 ,4,5,6-
Compou tetrahydropyridinecarboxylate
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-imidazolin-2-yl){3-
Compound 73 pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-2-
2-yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
Compound 75 pyrrolidinyl(3-pyridyl))carboxamide
{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compound 76 pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]acetyl}pyrrolidin-2-
yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
Compound 77 pyrrolidinyl(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
Compound 78 pyridyl))carboxamide
Compound 79 pyridyl))carboxamide
6-pyrrolidinyl(3-
N-{(1R)-2-[(5-fIuoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrrolidinyl(3-pyridyl))carbonylamino]-isopropyl}-2-
Compound 82 aminoacetamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[1-(methylsulfonyl)(4-
1l2,5,6-tetrahydropyridyl)](3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
[4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pipera2inyl]-N,N-
Compound dimethylcarboxamide
methyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
Compound pyridylmethyl)carbamoyl]-2-pyridyl)piperazinecarboxylate
(6-(3-aminopyrrolidinyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{f2-(3-fluorophenyl)ethyl]amiπo}-6-morpholiπ-4-yl(3-
pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 87
<6-[(2R)-2-(hydroxymethyl)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
2-[(6-((3S)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
Compoun methylacetamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-hydroxypiperidyI)(3-
Compoun pyridyl))carbonylamino]-N-methylacetamide
(tert-butoxy)-N-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
Compoun yljcarboxamide
(6-[(3R)-3-(dimethylamino)pyrrolidinyl]-2-{[2-(3- fluoropnenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compoun pyridylmethyl)carboxamide
Compound 93 methylacetamide
2-[(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3-
fluorophenyl)ethyI]amino}(3-pyιϊdyl))carbonylamino]-N-
Compound 94 methylacetamide eft
2-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
Compound pyridylmethyl)carbamoyl]-2-pyridyl)pyrrolidin-3-yl]acetic acid
Compound 96 pyridyl)pyrrolidinyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
2-[(6-(3,3-difluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N- methylacetamide
Compound 98 pyridyl))carboxamide
(2-{[2-{3-fluorophenyl)ethyl]amino}-6-[3-
(trifluoromethyl)pyrrolidinyl](3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
(6-(3,3-difluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethy!]amino}(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
Compound 100
(6-((3S)-3-hydroxypyrrolidinyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[6-(aminomethyl)(2- Compound 102 pyridyl)]methyl}carboxamide
-(aminomethyl)(2-
(6-((3R)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound 1 pyridylmethyl)carboxamϊde
Compound 1 pyridylmethyl)carboxamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-piperidyl(3-
Compound 106 pyridyl))carbonylamino]-N-methylacetamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[3-
(methylsulfonyl)pyrrolidinyl](3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(6-[(2S)-2-(methoxymethyl)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}{3-pyridyl))-N-(3-
Compou pyridylmethyl)carboxamide
(tert-butoxy)-N-{[ 1 -{6-{[2-(3-f luorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl)){4-
Compou piperidyl)]methyl}carboxamide
(tert-butoxy)-N-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
Compou pyridylmethyl)carbamoyl](2-pyridyl))(4-piperidyl)]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-hydroxy-4-
Compou phenylpiperidylJCS-pyridylJJ-N-CS-pyridylmethyQcarboxamide
ethyl 1 -(6-{[2-(3-fIuorophenyl)ethyl]amino}-5-[N-(3-
Compou pyridylmethyl)carbamoyl]-2-pyridyl)piperidine-4-carboxylate
(6-((3S)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridy)))-N-(3-
pyιϊdylmethyl)carboxamide
Compound 113
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[4-(N- methylcarbamoyl)piperidyl](3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-hydroxypiperidyl)(3-
Compound pyridyl))-N-(3-pyridylmethyl)carboxamide
{2-{[2-(3-fluorophenyl)ethyl]amino}-6-piperazinyl(3-pyridyl))-N-
Compound {3-pyridylmethyl)carboxamide
Compound 117 pyridylmethyl)carboxamide
N-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl]-2-pyridyl)pyrrolidin-3-yl]acetamide
(hydroxymethyl)morpholin-4-yl](3-pyridyl))-N-(3-
Compound 119 pyridylmethyl)carboxamide
(6-[(3S)-3-(methylamino)pyrro!idinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound pyridylmethyljcarboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1,4-thiazaperhydroin-4-
Compound yl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(1,1-dioxo(1,4-thiazaperhydroin-4-yl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound pyridylmethyl)carboxamide
(6-(4-acetylpiperazinyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-piperidylpiperidyl)(3-
Compound pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 125 pyridylmethyl)carbamoyl]-2-pyridyl)piperazinecarboxylate
[0164] Provided is at least one chemical entity chosen from compounds of Formula VII
Formula VII
and pharmaceutically acceptable salts thereof wherein
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is chosen from hydrogen, cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
Z2 is chosen from hydrogen, optionally substituted amidiπo, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
[0165] In certain embodiments of compounds of Formula VII, Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
[0166] In certain embodiments of compounds of Formula VII1 Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
[0167] In certain embodiments of compounds of Formula VII, Z2 is chosen from (2-(N- acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2- (methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-ylmethyl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
[0168] In certain embodiments of compounds of Formula VII1 R3 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments, R3 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula VII, R3 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula VII, R3 is hydrogen.
[0169] In certain embodiments of compounds of Formula VII, R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl. In certain embodiments of compounds of Formula VII, R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl. In certain embodiments of compounds of Formula VII1 R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl. In certain embodiments of compounds of Formula VII, R4 is hydrogen.
[0170] In certain embodiments of compounds of Formula VII, R5 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula VII, R5 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula VII, R5 is hydrogen.
[0171] In certain embodiments of compounds of Formula VII, Z2 is -C(O)-NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
[0172] In certain embodiments of compounds of Formula VII, R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally
substituted cyclopropyl, optionally substituted cyclopeπtyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0173] In certain embodiments of compounds of Formula VII, R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
[0174] In certain embodiments of compounds of Formula VII, R2 is chosen from (1-(2- aminoethyl)-1H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin- 3-yl)methyl; (1 -acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-5-yl)methyl; (2- (aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- amiπoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1-(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1 -(aminocarbonyl)-2-(amino)-eth-1 -yl; 1 -(aminocarbonyl)-3-(amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1 -yl; 1 -(methoxycarbonyl)-2-(amino)-eth-1 -yl; 1 -(methoxycarbonyl)-2-(t-butoxycarbonylamino)-
eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylamiπocarbonyl)eth-i-yl; i-amiπocarbonyl-2-hydroxy-eth-i-yl; 1-methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-{acetylamino)ethyl; 2-{amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamiπo)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i -yl; 2-(methoxycarbonyl)-2-(amino)-eth-1 -yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1 -yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2- amiπoethyl)cyclohexyl)methyl; 3-{4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; 3-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxypropyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; 4- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; A- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)ρyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1 -yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl;
isopropyl; isoxazol-5-ylm ethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
[0175] In certain embodiments of compounds of Formula VII, Z3 is chosen optionally substituted lower alkyl.
[0176] In certain embodiments of compounds of Formula VII, Z3 is chosen from optionally substituted methyl, optionally substituted ethyl, and optionally substituted propyl wherein the methyl, ethyl and propyl groups are optionally substituted with one or two groups selected from hydroxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, and optionally substituted arylσxy.
[0177] In certain embodiments of compounds of Formula VII, Z3 is chosen from 2-(3- methylphenyl)ethyl, (tetrahydrofuran-2-yl)methyl, 1-hydroxycyclohexylmethyl, 2-(1 ,3-dioxolan-2- yl)ethyl, 2-(1H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazoI-4-yl)ethyl, 2-(1-methylpiperidin-4- yl)ethyl, 2-(2,3-difludrophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(2-oxoimidazolidin-1-yl)ethyl, 2-(2- oxopiperidin-1 -yl)ethyl, 2-(2-oxopyrrolidin-1 -yl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1H-pyrazol-1-yl)ethyl, 2-(3,5- dimethyl-1 H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-{3-fluorophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydroxyphenyl)ethyl, 2-(benzo[d][1,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-cyclohex-1-enylethyl, 2-cyclohexylethyl, 2-morpholinoethyl, 2-phenoxyethyl, 2-phenylprop-1- yl, 3-(1H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, chloro, isopentyl, propyl, pyridin-2- ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
[0178] In certain embodiments of compounds of Formula VII, Z3 is chosen from 2-(3- fluorophenyl)ethyl and 2-(pyridin-2-yl)ethyl.
[0179] In certain embodiments of compounds of Formula VII, Z1 is chosen from cyano, halo, optionally substituted acyl, optionally substituted alkenyl, optionally substituted
alkoxycarbonyl, optionally substituted alkyl, optionally substituted amino, optionally substituted aminocarbonyl, and optionally substituted carbamimidoyl.
[0180] In certain embodiments of compounds of Formula VII1 Z1 is chosen from acyl, alkenyl, alkoxycarbonyl, alkyl, amino, aminocarbonyl, and carbamimidoyl, where each group is optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
[0181] In certain embodiments of compounds of Formula VII, Z1 is chosen from (5- oxo-tetrahydrofuran-2-yl)methylamino; (E)-N'-cyano-N, N-dimethylcarbamimidoyl; (Z)-4-(N-(2- hydroxyethyl)-N,N'-bis(methoxycarbonyl)carbamimidoyl); (Z)-but-2-en-2-yl; 1-ethoxyeth-1-yl; 1- ethoxyvinyl;2-hydroxyeth-1 -ylamino 2-methoxyeth-1 ylamino;2-methylprop-1 -enyl; 3- (methoxycarbonyl)-prop-2-en-2-yl; 3-(N,N-dimethylamino)carbonyl-prop-1 -ylamino; 3- aminocarbonyl-prop-1 -ylamino; 3-hydroxyprop-1 -ylamino; 3-methoxy-prop-1 -enyl; 3- methoxyprop-1 -ylamino; 3-methoxypropyl; 3-methylbut-2-eπ-2-yl; 4-((pyridin-3- ylmethyl)aminocarbonyl)butylamino; 4-(N, N-dimethylamino)carbonyl-but-1 -ylamino; 4-(N- methylamino)carbonyl-but-1 -ylamino; 4-aminocarbonyl-but-1 -ylamino; acetyl; aminocarbonyl; carbamimidoyl; chloro; cyano; dimethylamino; ethoxycarbonyl; hydrogen; methoxycarbonyl; methylamino; N-(2-hydroxyethyl)-N-(acetyl)-amino; N-(2-hydroxyethyl)-N-(methyl)-amino; N-(2- hydroxyprop-1-yl)-N-(methyl)-amino; N-(methyl)-aminocarbonyl; N-(pyridin-2-ylmethyl)amino; N- (pyridin-3-ylmethyl)amiπo; N-(pyridin-4-ylmethyl)amino; N, N-dimethylcarbamimidoyl; N- cyanocarbamimidoyl; N-methyl-N-acetylamino; and prop-1-enyl.
[0182] In certain embodiments of compounds of Formula VII, R1 is chosen from hydrogen and optionally substituted lower alkyl. In certain embodiments of compounds of Formula VIJ, R1 is chosen from hydrogen and lower alkyl. In certain embodiments of compounds of Formula VII, R1 is chosen from hydrogen and methyl. In certain embodiments of compounds of Formula VII, R1 is hydrogen.
[0183] In certain embodiments, the compound of Formula VII is chosen from Compound Chemical Name
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
fluorophenyl)ethyl]amino}-5-(3-pyridyl)(3-
Compound 0 pyridyl))carboxamide
Compound 1 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
fluorophenyl)ethyl]amino}-5-(1-methylpyrazol-4-yl)(3-
Compound 2 py ridyl))ca rboxam ide
Compound 3 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 4 (2-pyridylmethyl)carboxamide
Compound 5 [(6-methyl(2-pyridyl))methyl]carboxamide
(δ-bromo-δ-chloro-Z-fp-CS-fluorophenylJethyllaminoXS-
Compound 6 pyridyl))-N-(2-pyridylmethyl)carboxamide
Compound 7 carboxylic acid
Compound 8 3-carboxylate
Compound 9 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
C 10 (2-py ridylmethyl)ca rboxam ide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(5-bromo-2-{[2-(3- Compound 11 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
fluorophenyl)ethyl]amino}-5-(2-pyridyl)(3-
Compound 12 pyridyl))carboxamide
Compound 13 fluorophenyl)ethyl]amino}pyridine-3-carboxylate
Compound 14 dimethylpentaπamide
Compound 15 methylpentanam ide
Compound 16 pyridylmethyl)carboxamidθ
Compound 17 dimethylbutanamide
(6-[(3-carbamoylpropyl)amino]-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound 18 pyridylmethyl)carboxamide
Compound 19 pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 20 pyridylmethyl)peπtanamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(methylamino)(3- pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 22 pyridylmethyl)carbamoyl](2-pyridyl))-N-methylacetamide
Compound 24 pyridylmethyl)carbamoyl]pyridine-2-carboxamide
Compound 25 (3-pyridylmethyl)carboxamide
Compound 26 pyridylmethyl)carboxamide
Compound 27 pyridylmethyl)carboxamide
Compound 28 pyridylmethyl)carboxamide
Compound 30 pyridylmethyl)carbamoyl]pyιϊdine-2-carboxylate
Compound 31 pyridylmethyl)carboxamide
Compound 32 pyridylmethyl)carboxamide
Compound 33 pyridylmethyl)carbamoyl](2-pyridyl))but-2-enoate
Compound 34 pyridylmethyl)carboxam»de
Compound 35 pyridylmethyl)carbamoyl]pyridine-2-carboxylate
(6-((1 E)prop-1 -enyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
Compound 36 pVridyl))-N-(3-pyridylmethyl)carboxamide
Compound 37 pyridylmethyl)carboxamide
Compound 38 enyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-[(dimethylamino)iminomethyl]-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3- Compound 39 pyridylmethyl)carboxamide
(6-[(1 E)-1 -(dimethylamino)-2-cyano-2-azavinyl]-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
Compound 40 pyridylmethyl)carboxamide
Compound 41 pyridylmethyl)carbamoyl]pyridine-2-carboxamidine
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-5-phenyl(3-
Compound 42 pyridyl))carbonylamino]-N-methylacetamide
(6-((1 E)prop-1 -enyl)-2-{[2-(3-f luorophenyl)ethyl]amino}(3-
pyridyl))-N-{[2-(aminomethyl)(3- boc pyridyl)]methyl}carboxamide
Compound 43 pyridyl))carboπylamino]-N-methylacetamide
Compound 44 pyridylmethyl)carboxamide
Compound 45 pyridyl))-N-(3-pyridylmethyl)carboxamlde
Compound 46 (3-pyridylmethyl)carboxamide
(6-((1 E)prop-1 -enyl)-5-f luoro-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-
Compound 47 (aminomethyl)(3-pyridyl))methyl}carboxamide
(6-((1E)prop-1-enyl)-5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-{[(tert-
butoxy)carbonylamino]methyl}(3-
Compound 48 pyrid y l))methyl]carboxam ide
Compound 49 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Compound 50 pyridylmethyl)carboxamide
Compound 51 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-methoxypropyl)(3- Compound 52 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(1-ethoxyvinyI)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))-N-(3-pyridylmethyl)carboxamide
Compound 54 pyridylmethyl)carboxamide
Compound 55 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(2- methoxyethyl)amino](3-pyridyl))-N-(3- pyridylmethyl)carboxamide
Compound 57 pyridylmethyl)carboxamide
Compound 58 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(3-
hydroxypropyl)amino](3-pyridyl))-N-(3- Compound 59 pyridylmethyl)carboxamide
[0184] The compounds described herein can be synthesized utilizing techniques well known in the art from commercially available starting materials and reagents. For example, the compounds can be prepared as described below.
[0185] Referring to Reaction Scheme 1, Step 1, to a solution of a compound of Formula 101 and an excess (such as about 1.1 equivalents) of a compound of formula Z3NH2 in a polar solvent such as DMF is added a base such as potassium carbonate at room temperature. The product, a compound of Formula 103, is isolated and optionally purified.
[0186] Referring to Reaction Scheme 1, Step 2, to a mixture of a compound of Formula 103, an excess (such as about 2 equivalents) of a compound of formula Z1B(OH)2, and a base such as potassium carbonate in a polar solvent such as DMF is added about 0.1 equivalent of PdCI2. dppf. The reaction mixture is stirred at about 1000C for about 4 h. The product, a compound of Formula 105, is isolated and optionally purified.
[0187] Referring to Reaction Scheme 1 , Step 2, to a solution of a compound of Formula 105 and an excess (such as about 1.4 equivalents) of a compound of Formula R10NH2 in a nonpolar solvent such as toluene is added an excess (such as about 1.5 equivalents) of AIMe3 at r.t.. The reaction mixture is stirred at about 70 0C overnight until completion. The product, a compound of Formula 107 wherein Z2 is aminocarbonyl, is isolated and optionally purified.
[0188] Unless specified to the contrary, for each gram of the limiting reagent, one cc (or ml.) of solvent constitutes a volume equivalent.
[0189] Isolation and purification of the compounds and intermediates described herein can be effected, if desired, by any suitable separation or purification procedure such as, for example, filtration, extraction, crystallization, column chromatography, thin-layer chromatography or thick-layer chromatography, or a combination of these procedures. Specific illustrations of suitable separation and isolation procedures are provided in the Example. However, other equivalent separation or isolation procedures can, of course, also be used. [0190] When desired, the (R) and (S) isomers may be resolved by methods known to those skilled in the art, for example by formation of diastereoisomeric salts or complexes which may be separated, for example, by crystallization; via formation of diastereoisomeric derivatives which may be separated, for example, by crystallization, gas-liquid or liquid chromatography; selective reaction of one enantiomer with an enantiomer-specific reagent, for example enzymatic oxidation or reduction, followed by separation of the modified and unmodified enantiomers; or gas-liquid or liquid chromatography in a chiral environment, for example on a chiral support, such as silica with a bound chiral ligand or in the presence of a chiral solvent. It will be appreciated that when the desired enantiomer is converted into another chemical entity by one of the separation procedures described above, a further step may be required to liberate the desired enantiomeric form. Alternatively, a specific enantiomer may be synthesized by asymmetric synthesis using optically active reagents, substrates, catalysts and/or solvents, or by converting one enantiomer to the other by asymmetric transformation. [0191] The chemical entities described herein may be useful in a variety of applications involving smooth muscle cells and/or non-muscle cells. In certain embodiments, the chemical entities may be used to inhibit smooth muscle myosin. The chemical entities may be useful to bind to, and/or inhibit the activity of, smooth muscle myosin. In certain embodiments, the smooth muscle myosin is human, although the chemical entities may be used to bind to or inhibit the activity of smooth muscle myosin from other organisms, such as other mammals.
[0192] In certain embodiments, the chemical entities may be used to inhibit non-muscle myosin. The chemical entities may be useful to bind to, and/or inhibit the activity of, non- muscle myosin. In certain embodiments, the non-muscle myosin is human, although the chemical entities may be used to bind to or inhibit the activity of non-muscle myosin from other organisms, such as other mammals.
[0193] The chemical entities described herein may be used to treat disease states associated with smooth muscle and/or non-muscle myosin. Such disease states which can be treated by the chemical entities described herein include, but are not limited to, hypertension, asthma, incontinence, chronic obstructive pulmonary disorder, pre-term labor, and the like. It is appreciated that in some cases the cells may not be in an abnormal state and still require treatment. Thus, in certain embodiments, the chemical entities described herein can be applied to cells or administered to individuals afflicted or subject to impending affliction with any one of these disorders or states.
[0194] More specifically, the cchemical entities described herein may be useful for the treatment of diseases or symptoms related to abnormal increased muscle tone or excessive contraction, or spasm of vascular smooth muscle in systemic, coronary, pulmonary circulation, and micro-circulatory smooth muscle as well, such as systemic hypertension, malignant hypertension, hypertension crisis, symptomatic hypertension, pulmonary hypertension, pulmonary infarction, angina pectoris, cardiac infarction, micro-circulation malfunction under shock condition, and infarction occurred in other location or organs of the human or animal body. Other diseases or symptoms that can be treated with the chemical entities described herein include: spasm of gastro-intestine smooth muscle, including sphincters, such as gastric spasm, pylorospasm, and spasms of biliary tract, pancreatic tract, urinary tract, caused by inflammation, stimulation of stones or parasites; spasm of other visceral organs such as uterus, Fallopian tube, and so on; spasm of trachea-bronchial tree smooth muscle, diaphragm muscle, such as various asthma, breathlessness, dyspnea, diaphragmatic convulsion, and so on; spasm of alimentary canal smooth muscle, including stomach, intestine and colons, biliary and pancreatic duct etc.; and spasm of urinary tract smooth muscle.
[0195] In addition, the chemical entities described herein can be used for control, management and manipulation of labor during pregnancy. The method is particularly useful for inhibition of
spontaneous preterm labor which would, if untreated, result in premature delivery or abortion and for inhibition of surgically induced labor during transuterine fetal surgery. The method is also useful for inducing the labor in overterm pregnancies where the labor does not occur on term and when it is necessary to induce labor in order to assure the normal delivery. [0196] Further, the chemical entities described herein can be used for the treatment of "airway wall remodeling", which is a condition associated with diseases or conditions characterized by airway wall thickening and air obstruction, which may, for example occur in the small airways of patients with certain respiratory disease conditions, such as, chronic obstructive pulmonary disease (COPD).
[0197] Such disease states which can be treated by the chemical entities, compositions and methods provided herein also include, but are not limited to glaucoma and other ocular indications. More specifically, chemical entities described herein may be useful for the treatment of diseases or symptoms related to glaucoma, including increased intraocular pressure, reduced flow of intraocular aqueous humor, and optical nerve damage. Other diseases or symptoms that can be treated with the chemical entities, compositions, and methods described herein including intraocular hypertenstion.
[0198] ATP hydrolysis is employed by myosin to produce force. An increase in ATP hydrolysis would correspond to an increase in the force or velocity of muscle contraction. In the presence of actin, myosin ATPase activity is stimulated more than 100-fold. Thus, the measurement of ATP hydrolysis not only measures myosin enzymatic activity but also its interaction with the actin filament. Assays for such activity may employ smooth muscle myosin from a human source, although myosin from other organisms can also be used. Systems that model the regulatory role of calcium in myosin binding may also be used.
[0199] The in vitro rate of ATP hydrolysis correlates to smooth muscle myosin potentiating activity, which can be determined by monitoring the production of either ADP or phosphate, for example as described in U.S. Patent No. 6,410,254. ADP production can also be monitored by coupling the ADP production to NADH oxidation (using, for example, the enzymes pyruvate kinase and lactate dehydrogenase) and monitoring the NADH level, by example, either by absorbance or fluorescence (Greengard, P., Nature 178 (Part 4534): 632-634 (1956); MoI Pharmacol 1970 Jan;6(1):31-40). Phosphate production can be monitored using purine nucleoside phosphorylase to couple phosphate production to the cleavage of a purine analog, which results in either a change in absorbance (Proc Natl Acad Sci U S A 1992 Jun 1 ;89(11 ):4884-7) or fluorescence (Biochem J 1990 Mar 1;266(2):611-4). While a single
measurement is employed, multiple measurements of the same sample at different times in order may be used to determine the absolute rate of the protein activity; such measurements have higher specificity particularly in the presence of test compounds that have similar absorbance or fluorescence properties with those of the enzymatic readout. [0200] Test compounds may be assayed in a highly parallel fashion using multiwell plates by placing the compounds either individually in wells or testing them in mixtures. Assay components including the target protein complex, coupling enzymes and substrates, and ATP may then be added to the wells and the absorbance or fluorescence of each well of the plate can be measured with a plate reader.
[0201] One method uses a 384 well plate format and a 25 μl_ reaction volume. A pyruvate kinase/lactate dehydrogenase coupled enzyme system (Huang TG and Hackney DD. (1994) J Biol Chem 269(23): 16493-16501) is used to measure the rate of ATP hydrolysis in each well. As will be appreciated by those of skill in the art, the assay components are added in buffers and reagents. Since the methods outlined herein allow kinetic measurements, incubation periods may be optimized to give adequate detection signals over the background. The assay is performed in real time to give the kinetics of ATP hydrolysis to increase the signal-to-noise ratio of the assay.
[0202] Selectivity for smooth muscle myosin may be determined by substituting other myosins in one or more of the above-described assays and comparing the results obtained against those obtained using the cardiac equivalents.
[0203] Chemical entities identified by the methods described herein as smooth muscle myosin modulators may be further tested in an efficacy screen, such as a screen using strips of permeabilized smooth muscle from, e.g., chicken gizzard. Calcium-sensitive smooth muscle strips are prepared by dissecting chicken gizzard tissue, followed by treatment with 1% Triton X-100 to make the strips permeable to exogenous compounds (Barsotti, RJ, et al., Am J Physiol. 1987 May;252(5 Pt 1):C543-54). These strips can be stored in 50% glycerol for several weeks at -200C, allowing multiple experiments to be performed with each batch of muscle strips. Experiments are performed using a solution of 20 mM imidazole pH 7.0, 5.5 mM ATP1 7 mM MgCI2, 55 mM KCI, 1 μM Calmodulin, and 10 mM EGTA. Free calcium will be controlled by addition of various amounts of CaCI2, according to the calculations of MAXChelator (Patton, et al. Cell Calcium. 35/5 pp. 427-431, 2004). An isometric muscle fiber apparatus is used to measure isometric tension, for example using an Aurora Scientific 400A transducer with National Instruments PCI-MIO-16E-4, 16 channels, 12 bit A/D board for data
acquisition. The chemically skinned gizzard fibers are relaxed when bathed in low calcium solutions (pCa 8), but develop isometric tension when the free calcium of the bathing solution is increased to pCa 5. These fibers can be repeatedly contracted and relaxed by switching between high and low calcium bathing solutions.
[0204] Compounds are first tested for their ability to prevent contraction of gizzard strips, by preincubating relaxed fibers with a compound, followed by transfer to high calcium solution containing the compound. Next, compounds are tested for their ability to cause relaxation of contracting fibers by adding the compound to fibers already incubating in high calcium solution. Washout experiments are performed to ensure that the inhibitory effects are reversible, so that the compounds do not cause denaturation or other irreparable damage to the smooth muscle myosin.
[0205] The chemical entities are administered at a therapeutically effective dosage, e.g., a dosage sufficient to provide treatment of the disease states previously described. Generally, a daily dose is from about 0.05 to about 100 mg/kg of body weight, such as from about 0.10 to about 10 mg/kg of body weight or from about 0.15 to about 1 mg/kg of body weight. Thus, for administration to a 70 kg person, the dosage range is from about 3.5 to about 7000 mg per day, such as from about 7 to about 700 mg per day or from about 10 to about 100 mg per day. The amount of active chemical entity administered will, of course, be dependent on the subject and disease state being treated, the severity of the affliction, the manner and schedufe of administration and the judgment of the prescribing physician; for example, a dose range for oral administration may be from about 70 to about 700 mg per day, whereas for intravenous administration the dose range may be from about 700 to about 7000 mg per day. The active agents may be selected for longer or shorter plasma half-lives, respectively. [0206] Administration of the chemical entities described herein can be via any of the accepted modes of administration for agents that serve similar utilities including, but not limited to, orally, subcutaneously, intravenously, intranasally, topically, transdermal^, sublingually, intramucosally, intraperitoneally, intramuscularly, intrapulmonarilly, vaginally, rectally, and intraocularly (including intraocular injection). Oral, topical, parenteral, and intraocular administration are customary in treating many of the indications recited herein. [0207] Pharmaceutically acceptable compositions include solid, semi-solid, liquid and aerosol dosage forms, such as, e.g., tablets, capsules, powders, liquids, suspensions, suppositories, aerosols, and the like. The chemical entities can also be administered in sustained- or controlled-release dosage forms, including depot injections, osmotic pumps, pills, transdermal
(including electrotransport) patches, drops and the like, for prolonged and/or timed, pulsed administration at a predetermined rate. The compositions may be provided in unit dosage forms suitable for single administration of a precise dose.
[0208] The chemical entities may be administered either alone or in combination with a conventional pharmaceutical carrier or the like (e.g., mannitol, lactose, starch, magnesium stearate, sodium saccharine, talcum, cellulose, sodium crosscarmellose, glucose, gelatin, sucrose, magnesium carbonate, and the like). If desired, the pharmaceutical composition may also contain minor amounts of nontoxic auxiliary substances such as wetting agents, emulsifying agents, solubilizing agents, pH buffering agents and the like (e.g., sodium acetate, sodium citrate, cyclodextrine derivatives, sorbitan monolaurate, triethanolamine acetate, triethanolamine oleate. Generally, depending on the intended mode of administration, the pharmaceutical composition may contain from about 0.005% to about 95%, for example, from about 0.5% to about 50%, by weight of at least one chemical entity. Actual methods of preparing such dosage forms are known or will be apparent, to those skilled in this art; for example, see Remington's Pharmaceutical Sciences, Mack Publishing Company, Easton, Pennsylvania. Pharmaceutical compositions are also referred to as pharmaceutical formulations.
[0209] In addition, the chemical entities may be co-administered with, and the pharmaceutical compositions can include, other medicinal agents, pharmaceutical agents, adjuvants, and the like.
[0210] In certain embodiments, the compositions are in the form of a pill or tablet and contain, along with the active ingredient, one or more of a diluent such as lactose, sucrose, dicalcium phosphate, and the like; a lubricant such as magnesium stearate or the like; and a binder such as starch, gum acacia, polyvinylpyrrolidine, gelatin, cellulose, cellulose derivatives and the like. In another solid dosage form, a powder, marume, solution or suspension (e.g., in propylene carbonate, vegetable oils or triglycerides) may be encapsulated in a gelatin capsule. [0211] Liquid pharmaceutical compositions may, for example, be prepared by dissolving, dispersing, etc. at least one chemical entity and one or more optional pharmaceutical adjuvants in a carrier (e.g., water, saline, aqueous dextrose, glycerol, glycols, ethanol and-the like) to form a solution or suspension. Injectables may be prepared in conventional forms, either as liquid solutions or suspensions, as emulsions, or in solid forms suitable for dissolution or suspension in liquid prior to injection. The percentage of chemical entities contained in such parenteral compositions is highly dependent on the specific nature thereof, as well as the activity of the
chemical entities and the needs of the subject. However, percentages of active ingredient ranging from about 0.01% to about 10% in solution may be used, and may be higher if the composition is a solid which will be subsequently diluted to the above percentages. In certain embodiments, the composition has from about 0.2% to about 2% of the active agent in solution. [0212] Compositions comprising at least one chemical entity may be administered intraocularly (including intraocular, periocular, and retrobulbar injection and perfusion). When adminsitered intraocularly the sterile composition is typically aqueous. An appropriate buffer system may be added to prevent pH drift under storage conditions. When administered during intraocular surgical procedures, such as retrobulbar or periocular injection and intraocular perfusion or injection, the use of balanced salt irrigating solutions may be necessary. When used in a multidose form, preservatives may be required to prevent microbial contamination during use. [0213] Compositions comprising at least one chemical entity may also be administered topically as eye drops, eye wash, creams, ointments, gels, and sprays. When administered as eye drops or eye wash, the active ingredients are typically dissolved or suspended in suitable carrier, typically a sterile aqueous solvent. An appropriate buffer system may be added to prevent pH drift under storage conditions. When used in a multidose form, preservatives may be required to prevent microbial contamination during use.
[0214] Compositions comprising at least one chemical entity may also be administered to the respiratory tract as an aerosol or in a solution for a nebulizer, or as a microfine powder for insufflation, alone or in combination with an inert carrier such as lactose. The particles of the composition typically have diameters of less than 50 microns, for example, less than 10 microns.
[0215] Generally, to employ the chemical entities described herein in methods of screening for smooth muscle myosin binding, smooth muscle myosin is bound to a support and at least one chemical entity is added to the assay. Alternatively, the chemical entity may be bound to the support and the smooth muscle myosin added. Classes of compounds among which novel binding agents may be sought include specific antibodies, non-natural binding agents identified in screens of chemical libraries, peptide analogs, etc. Of particular interest are screening assays for candidate agents that have a low toxicity for human cells. A wide variety of assays may be used for this purpose, including labeled in vitro protein-protein binding assays, electrophoretic mobility shift assays, immunoassays for protein binding, functional assays (phosphorylation assays, etc.), and the like. See, e.g., U.S. Patent No. 6,495,337.
EXAMPLES
[0216] The following examples serve to more fully describe the manner of using the invention. These examples are presented for illustrative purposes and should not serve to limit the true scope of the invention.
Example 1 :
6-(2-cyanophenyl)-2-(3-fluorophenethylamino)-iV-(pyridin-3-ylinethyl)nicotinamide
[0217J To a solution of 2,6-dichloronicotinic acid (5Og, 0.26 mol) in methanol (250 ml_) was added thionyl chloride (22.7 mL, 0.312 mol) slowly. The mixture was refluxed for 4 hours and concentrated to give methyl 2,6-dichloronicotinate as a white solid in quantitative yield.
[0218] A mixture of methyl 2,6-dichloronicotinate (53.3g, 0.26mol), 3-fluorophenethyl amine (37.3 ml_, 0.29 mol), K2CO3 (72 g 0.52 mol) and NMP (150 mL) was stirred at 1000C for 10 hrs. The mixture was diluted with ethyl acetate (2 X 300 mL) and washed with brine. The organic layers were combined, dried (MgSO4) and concentrated. The residue was purified by silica gel chromatography using hexane-EtOAc (9:1) as eluant to give methyl 6-chloro-2-(3- fluorophenethylamino)nicotinate as an off-white solid (44 g, 54%). LRMS (M+H+) m/z 309.0.
[0219] A mixture of chloride methyl 6-chloro-2-(3-fluorophenethylamino)nicotinate (30.8 g, 0.1 mol), 2-cyanophenyl boronic acid (17.6 g, 0.12 mol), PdCI2(dppf)2 (3.6 g, 5.0 mmol), K2CO3 (41.4 g, 0.3 mol) and anhydrous DMF (100 mL) was degassed with nitrogen for 30min. The mixture was then stirred at 100°C for 2 hrs. The mixture was filtered through the Celite and washed with EtOAc. The filtrate was diluted with EtOAc, washed with brine and concentrated to dryness. The residue was purified by silica gel flash chromatography using hexane-EtOAc (4:1) as eluant to give the desired product methyl 6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinate as a yellow solid (31 g, 82%). LRMS (M+H+) m/z 376.1.
[0220] To a mixture of methyl ester methyl 6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinate (15 g, 40 mmol), methanol (80 mL) and THF (80 mL) was added 3N NaOH (40 mL). The mixture was stirred at room temperature for 16 hrs. The mixture
was adjusted to pH 7 with 2N HCI. After concentration, the residue in the aqueous solution was acidified to pH 3 with 2N HCI (10 ml_) to give the yellow precipitates. The yellow solid was filtered, washed with water and dried to afford 6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinic acid (14.5 g, 100%). LRMS (M+H+) m/z 362.1.
[0221] To a mixture of 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinic acid (2.8 g, 7.76 mmol) in DMF (28 mL) was added HBTU (5.88 g, 15.51 mmol), HOBt (2.1 g, 15.51 mmol), then 3-aminomethyl pyridine (1.68 g, 15.51 mmol). The reaction mixture was stirred at room temperature for 1 hr. The crude mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O to give 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)-N-(pyridin-3- ylmethyl)nicotinamide as a pale yellow solid (3.0 g, 86%). LRMS (M+H+) m/z 452.2.
Method B:
[0222] A mixture of acid 2,6-dichloronicotinic acid (19.2 g, 0.1 mol), N.N'-carbonyldiimidazole (CDI, 17.8 g, 0.11 mmol) and THF (1 L) was stirred at 6O0C for 30 min and cooled to room temperature. To the mixture was added 3-aminomethyl pyridine (10.2 mL, 0.1 mol) and DIEA
(21 ml_, 0.12 mol). The reaction mixture was stirred at room temperature for 4 hrs, and diluted with EtOAc. The organic layer was washed with H2O, brine, dried over Na2SO4. Concentration of the organic solution gave 2,6-dichloro-N-(pyridin-3-ylmethyl)nicotinamide (20.5 g, 73%). LRMS (M+H+) m/z 282.0; (M+2+H+) m/z 283.9.
[0223] A mixture of 2,6-dichloro-N-(pyridin-3-ylmethyl)nicotinamide (20.4 g, 72.3 mmol), 3- fluorophenethyl amine (11.1 g, 79.5 mmol), K2CO3 (20 g 145 mmol) and DMF (200 ml_) was stirred at 1000C for 10 hours. The mixture was diluted with ethyl acetate (2 X 300 mL) and washed wjth saturated ammonium chloride, sodium bicarbonate and brine. The organic layers were combined, dried (MgSO4) and concentrated. The residue was purified by silica gel chromatography to give 6-chloro-2-(3-fluorophenethyIamino)-N-(pyridin-3-ylmethyl)nicotinamide (12.6 g, 46%). LRMS (M+H+) m/z 385.1.
[0224] A mixture of 6-chloro-2-(3-fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide (1.0 g, 2.6 mmol), 2-cyanophenyl boronic acid (460 mg, 3.12 mmol), PdCI2(dppf)2 (140 mg, 0.26 mmol), K2CO3 (1.08 g, 7.8 mmol) and anhydrous DMF (8 mL) was degassed with nitrogen for 10 min. The mixture was then stirred at 1250C for 1 hr. The mixture was filtered through the silica gel pad and washed with EtOAc. The filtrate was concentrated to dryness and purified by silica gel flash chromatography to give the desired product 6-(2-cyanophenyl)-2-(3-
fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide as a pale yellow solid (740 mg, 63%). LRMS (M+H+) m/z 452.1.
Example 2:
II
/V-((2-(aminomethyl)pyridin-3-yI)methyl)-6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinamide
[0225] To a mixture of 2-bromoπicotiπaldehyde (50 g, 0.27 mol), methanol (250 mL) and THF (250 mL) in a 3-necked round bottom flask at O0C was added sodium borohydride (10.3 g, 0.27 mol) portion wise. The reaction mixture was stirred at O0C for 1 hr and warmed up to room temperature. The reaction mixture was adjusted to pH 7 with 2N HCI (120 mL), and concentrated to remove methanol and THF. The resulting mixture was partitioned between EtOAc and brine three times. The organic layer was combined and washed with brine, dried over Na2SO4, and concentrated to give (2-bromopyridin-3-yl)methanol (51 g), which was used without further purification. LRMS (M+H*) m/z 187.9.
[0226] To a mixture of (2-bromopyridin-3-yl)methanol (51 g, 0.27 mol), phthalimide (47.6 g, 0.32 mol), triphenylphosphine (83.8 g, 0.32 mol) and THF (60 ml_) at O0C was added DIAD (62 ml_, 0.32 mol) dropwise. The reaction mixture was allowed to warm to room temperature and stirred for 1 hr. The formed precipitate was filtered and washed with EtOAc (50 ml_). The obtained solid 2-((2-bromopyridin-3-yl)methyl)isoindoline-1 ,3-dione (50.2 g, 59%) was used for next step without further purification. LRMS (M+H+) m/z 317.1.
[0227] A mixture of bromide 2-((2-bromopyridin-3-yl)methyl)isoindoline-1,3-dione (50 g, 0.16 mol), Zn(CN)2 (37.4 g, 0.32 mol), Pd(PPh3J4 (18.5 g, 16 mmol) and DMF (160 ml_) was degassed with nitrogen for 10 min and then stirred at 1000C for 3 hrs. The mixture was filtered through the Celite and washed with DMF. The combined filtrate was diluted with EtOAc, washed with water, brine, dried (Na2SO4) and concentrated. The residue was triturated with dichloromethane to give the desired product 3-((1,3-dioxoisoindolin-2-yl)methyl)picolinonitrile as an off-white solid (35 g, 83%). LRMS (M+H+) m/z 264.0.
[0228] To a mixture of 3-((1 ,3-dioxoisoindolin-2-yl)methyl)picolinonitrile (10 g, 38 mmol) and methanol (80 ml_) was added cone. HCI (6.5 mL, 76 mmol) and 10% Pd/C (1 g). The mixture was stirred vigorously under 50 Psi H2 for 6 hrs. The mixture was filtered through the Celite pad and washed with methanol. The combined filtrate was concentrated to give the 2-((2- (aminomethyl)pyridin-3-yl)methyl)isoindoline-1 ,3-dione as a white solid in quantitative yield. LRMS (M+H+) m/z 268.1.
[0229] To a mixture of 2-((2-(aminomethyl)pyridin-3-yl)methyl)isoindoline-1 ,3-dione HCI salt (44.5 g, 0.13 mol), THF (230 mL) and H2O (230 mL) at O0C was added NaHCO3 (36 g, 0.43 mol), then Boc2O (31.4 g, 0.144 mol). The mixture was allowed to warm to room temperature and stirred for 2 hrs. The mixture was diluted with ethyl acetate (200 mL), washed with water, brine, dried and concentrated. The residue was triturated with EtOAc (150 mL) to give the desired product as a white solid (30 g, 63%). The EtOAc solution was purified by silica gel flash chromatography using hexane-EtOAc (1 :1) as eluant to give the second portion of tert-butyl (3- ((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methylcarbamate (13.5 g, 28%). LRMS (M+H+) m/z 368.1.
[0230] To a solution of tert-butyl (3-((1,3-dioxoisoindolin-2-yl)methyl)pyridin-2- yl)methylcarbamate (30 g, 81 mmol) in MeOH (450 mL) at 00C was added hydrazine (25 mL, 810 mmol). The mixture was allowed to warm to room temperature and stirred overnight. The precipitate was filtered off, and the filtrate was concentrated to dryness. The residue was triturated with dichloromethane (500 mL) and filtered. Concentration of the filtrate afforded tert- butyl (3-(aminomethyl)pyridin-2-yl)methylcarbamate in quantitative yield (18.7g). LRMS (M+H+) m/z 238.1.
[0231] To a mixture of 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinic acid (0.60 g, 1.66 mmol) in DMF (3 mL) was added DIEA (0.329 mL, 2.53 mmol), HBTU (0.756 g, 2.00 mmol), then tert-butyl (3-(aminomethyl)pyridin-2-yl)methylcarbamate (0.473 g, 2.11 mmol). The reaction mixture was stirred at room temperature for 1 hr. The crude mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O to give tert-butyl (3-((6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamido)methyl)pyridin-2-yl)methylcarbarnate as a pale yellow solid (545 mg, 57%). LRMS (M+H*) m/z 581.3.
II
[0232] To a solution of tert-butyl (3-((6-(2-cyaπophenyl)-2-(3- fluorophenethyIamino)nicotinamido)methyl)pyridin-2-yl)methylcarbamate (737 mg, 1.27 mmol) in methanol (6 ml_) at O0C was added a solution of 4M HCI in 1,4-dioxane (3 ml_). The mixture was warmed to room temperature and stirred for 2 hrs. Concentration of the mixture followed by the purification on RP-HPLC using a mixture of acetonitrile and H2O gave N-((2- (aminomethyl)pyridin-3-yl)methyl)-6-{2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamide as a pale yellow solid (545 mg, 89%). LRMS (M+H+) m/z 481.2.
Example 3:
III
[0233] Compound N-(2-amino-2-oxoethyl)-6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamide was prepared from 6-(2-cyanophenyl)-2-(3- f!uorophenethylamino)nicotinic acid and the appropriate amine 2-aminoacetamide according to the procedure (Method A) of preparation of Example 1. LRMS (M+H+) m/z 418.1.
Example 4:
IV
(/?)-/V-((l-(4-aminobutanoyI)pyrrolidin-2-yl)methyl)-6-(2-cyanophenyl)-2-(3-fluoropheπethylamino)nicotinamide
[0234] To a mixture of 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinic acid (510 mg, 1.41 mmol), (R)-tert-butyl 2-(aminomethyl)pyrrolidine-1-carboxylate (424 mg, 2.11 mmol), HOBT (285 mg, 2.11 mmol), and HBTU (800 mg, 2.11 mg) in RB flask was added DMF and the mixture was stirred at r.t. for 1 h. LC/MS indicated the reaction was complete and the mixture was filtered and purified on RP-HPLC using a mixture of acetonitrile and water to give (R)-tert- butyl 2-((6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinamido)methyl)pyrrolidine-1- carboxylate (695.1 mg, 91%). LRMS (M +H+) m/z 544.2.
[0235] To a solution of (R)-tert-butyl 2-((6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamido)methyl)pyrrolidine-1τcarboxylate (680 mg, 1.25 mmol) in MeOH (20 mL) was added HCI (2.0 mL, 8 mmol, 4 M in dioxane). The reaction was stirred at r.t. for 2 h. LC/MS indicated the reaction was complete. The reaction mixture was concentrated under reduced pressure to give 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)-N- (pyrrolidin-2-ylmethyl)nicotinamide hydrochloride which was used for next step without further purification. LRMS (M +H+) m/z 444.2.
[0236] To a mixture of 6-(2-cyanopheπyl)-2-(3-fluoropheπethylamino)-N-(pyrrolidiπ-2- ylmethyl)nicotinamide hydrochloride (95 mg, 0.2 mmol), 4-(tert-butoxycarbonylamino)butanoic acid {61 mg, 0.3 mmol), HOBT (41 mg, 0.3 mmol), and HBTU (114 mg, 0.3 mmol) were added DMF (1.0 ml_) and DIEA (0.1 ml_, 0.6 mmol). The reaction mixture was stirred at r.t. for 1 h. LC/MS indicated the reaction was complete. The reaction mixture was then filtered and the filtrate was purified on RP-HPLC using a mixture of acetonitrile and water to give tert-butyl 4-(2- ((6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinamido)methyl)pyrrolidin-1-yl)-4- oxobutylcarbamate (81.2 mg, 65%). LRMS (M +H+) m/z 629.2.
IV
[0237] To a solution of tert-butyl 4-(2-((6-(2-cyanophenyl)-2-(3- fluorophenethylaminoJnicotinamidoJmethylJpyrrolidin-i-yO^-oxobutylcarbamate (71 mg, 0.11 mmol) in MeOH (5.0 mL) was added HCI (1.0 mL, 4 mmol, 4 M in dioxane). The reaction mixture was stirred at r.t. for 2 h. LC/MS indicated the reaction was complete. The reaction mixture was concentrated under reduced pressure and the crude was purified on RP-HPLC using a mixture of acetonitrile (0.1% TFA) and water (0.1% TFA) to give N-((i-(4-
aminobutanoyl)pyrrolidin-2-yl)methyl)-6-(2-cyanophenyl)-2-(3- fluorophenethyIamino)nicotinamJde as a pale yellow solid (57.1 mg, 68%). LRMS (M +H+) m/z 529.2.
Example 5:
(R)-6-(2-cyanophenyl)-2-(3-fluorophenethylamino)-Λ/-((5-oxopyrrolidin-2-yl)methyl)nicotinamide
H DIAD, PPh3 HN3, THF N3 X ■■<y°
[0238] To (R)-5-(hydroxymethyl)pyrrolidin-2-one (660 mg, 5.73 mmol), hydrogen azide (10.8 mL, 8.60 mmol, 0.8 M in toluene), and PPh3 (2.25 g, 8.60 mmol) in THF was added DIAD dropwise. The mixture was stirred at rt for 1 h. LCMS indicated the completion of the reaction. The mixture was partially concentrated, filtered, and purified by RP-HPLC to give (R)-5- (azidomethyl)pyrrolidin-2-one (730 mgt 87%, 90% pure). LRMS (M +H+) m/z 141.1.
[0239] To (R)-5-(azidomethyl)pyrrolrdin-2-one (150 mg, 1.07 mmol) in MeOH was added Pd/C. The mixture was the placed into an autoclave, degasses three times, charged with 30 psi of H2 and stirred at rt for 30 min. The mixture was the filtered and the filtrate was concentrated under reduced pressure to give (R)-5-(aminomethyl)pyrrolidin-2-one (92 mg, 75%). LRMS (M +H+) m/z 115.1.
[0240] To a mixture of 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinic acid (110 mg, 0.30 mmol), (R)-5-(aminomethyl)pyrrolidin-2-one (52 mg, 0.46 mmol), HOBT (62 mg, 0.46 mmol), and HBTU (174 mg, 0.46 mg) in RB flask was added DMF and the mixture was stirred at r.t. for 1 h. LC/MS indicated the reaction was complete and the mixture was filtered and purified on RP-HPLC using a mixture of acetonitrile and water to give (R)-6-(2-cyanophenyl)-2- (3-fluorophenethylamiπo)-N-((5-oxopyrrolidiπ-2-yl)methyl)nicotinamide (91.6 mg, 67%). LRMS (M +H+) m/z 458.1.
Example 6:
(^-^((I^S-amiπopropylJ-δ-oxopyrrolidin^-ypmethyO-e^-cyaπophenyO^-fS- fluorophenethylamino)nicotinamide
[0241] To NaH (128 mg, 3.21 mmol) in DMF (5.0 ml_) was added (R)-5-(azidomethyl)pyrrolidin- 2-one (300 mg, 2.14 mmol) in DMF (5.0 ml_) and stirred at rt for 30 min followed by addition of tert-butyl 3-bromopropylcarbamate. The mixture was heated to 60 0C for 2 h and quenched with sat. NH4CI solution. The aqueous layer was extracted with EtOAc (50 ml_ x 2) and the combined organic layer was washed with brine, dried over Na2SO4, filtered, and concentrated under reduced pressure to give (R)-tert-butyl 3-(2-(azidomethyl)-5-oxopyrrolidin-1- yl)propylcarbamate (358 mg of crude), which was used for next step without purification. LRMS (M +H+-BoC) m/z 198.1.
[0242] To (R)-tert-butyl 3-(2-(azidomethyl)-5-oxopyrrolidin-1-yl)propylcarbamate (300 mg, 1.01 mmol) in MeOH was added Pd/C. The mixture was the placed into an autoclave, degasses three times, charged with 30 psi of H2 and stirred at rt for 30 min. The mixture was the filtered and the filtrate was concentrated under reduced pressure to give (R)-tert-butyl 3-(2- (aminomethyl)-5-oxopyrrolidin-1-yl)propylcarbamate (180 mg of crude). LRMS (M +H+) m/z 198.1.
[0243] To a mixture of 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinic acid (110 mg, 0.30 mmol), (R)-tert-butyl 3-(2-(aminomethyl)-5-oxopyrrolidin-1-yl)propylcarbamate (125 mg, 0.46 mmol), HOBT (107 mg, 0.46 mmol), and HBTU (174 mg, 0.46 mg) in RB flask was added DMF and the mixture was stirred at r.t. for 1 h. LC/MS indicated the reaction was complete and the mixture was filtered and purified on RP-HPLC using a mixture of acetonitrile and water to give (R)-tert-butyl 3-(2-((6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinamido)methyl)-5- oxopyrrolidin-1-yl)propylcarbamate (72 mg, 39%). LRMS (M +H+) m/z 615.2.
[0244] To a solution of (R)-tert-butyl 3-(2-((6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamido)methyl)-5-oxopyrrolidin-1-yl)propylcarbamate (72 mg, 0.12 mmol) in MeOH (5.0 mL) was added HCI ( 1.0 ml_, 4 mmol, 4 M in dioxane). The reaction mixture was stirred at r.t. for 2 h. LC/MS indicated the reaction was complete. The reaction mixture was concentrated under reduced pressure to give (R)-N-((1-(3-aminopropyl)-5- oxopyrrolidin-2-yl)methyl)-6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinamide hydrogen chloride (75 mg, quantitative). LRMS (M +H+) m/z 515.1.
Example 7:
VII yV-((6-(3-aminopropylamino)pyridin-2-yl)methyl)-6-(2-cyaπophenyI)-2-(3-fluorophenethyIamino)nicotinamide
[0245]To a solution of 6-(2-cyanophenyl)-2-(3-fluorophenethylamino)nicotinic acid (582mg, 1.61 mmol) and 6-bromo-pyridin-2yl-methylamine (300mg, 1.61 mmol) in DMF was added CDI (261 mg, 1.61 mmol). The reaction mixture was stirred at r.t. for 2 h. The reaction mixture was purified on RP-HPLC to give N-((6-bromopyridin-2-yl)methyl)-6-(2-
cyanophenyl)-2-(3-fluorophenethylamino)nicotinamide (700mg, 82%) LRMS (M+H+) m/z 530.1.
VII
[0246] A solution of N-((6-bromopyridin-2-yl)methyl)-6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamide (50 mg, 0.095 mmol,) and propane-1 ,3-diamine (396ul, 4.75mmol) in NMP was stirred in microwave at 150C for 15min. The reaction mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O gave N-((6-(3- aminopropylamino)pyridin-2-yl)methyl)-6-(2-cyanophenyl)-2-(3- fluorophenethylamino)nicotinamide (28 mg, 56 %). LRMS (M+H+) m/z 524.1.
Example 8:
VIII
2-(3-fluorophenethylamino)-6-(l//-pyrazol-4-yl)-Λ^-(pyridin-3-yImethyl)nicotinamide
[0247] To a mixture of ester methyl 6-chloro-2-(3-fluorophenethylamino)nicotinate (3.08 g, 10 mmol), methanol (20 ml_) and THF (20 mL) was added 3N NaOH (10 ml_, 30 mmol). The reaction mixture was stirred at room temperature for 2 days. The mixture was concentrated to remove methanol and THF and adjusted pH to 3 with 2N HCI. The precipitate was filtered and washed with water to give the 6-chloro-2-(3-fluorophenethylamino)nicotinic acid in quantitative yield. LRMS (M+H+) m/z 295.1.
[0248] A mixture of 6-chloro-2-(3-fluorophenethylamino)nicotinic acid (10.6 g, 36 mmol), borolane (12.72 g, 43 mmol), Pd(PPh3J4 (4.2 g, 3.6 mmol), K2CO3 (14.9 g, 108 mmol) and anhydrous DMF (50 mL) was degassed with nitrogen for 10 min. The mixture was then stirred at 120 0C in microwave for 2 hrs. The mixture was filtered through the Celite and washed with DMF. The filtrate was adjusted to pH 4 with 2N HCI (28 mL), and diluted with EtOAc. The organic layer was washed with brine twice and concentrated to dryness. The residue was triturated with dichloromethane (300 mL). Filtration followed by washed with EtOAc (20 mL) gave 2~(3-fluorophenethylamino)-6-(1 H-pyrazol-4-yl)nicotinic acid as a yellow solid (7.1 g, 61%). LRMS (M+H+) m/z 327.1.
[0249] To a mixture of 2-(3-fluorophenethylamino)-6-(1 H-pyrazol-4-yl)nicotinic acid (prepared according to example 1, method A) (1.0 g, 3.07 mmol) in DMF (10 ml.) was added HBTU (1.75 g, 4.6 mmol), HOBt (0.62 g, 4.6 mmol), then amine 7 (0.66 g, 6.13 mmol). The reaction mixture was stirred at room temperature for 2 hr. The crude mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O to give 2-(3-fluorophenethyIamino)-6-(1H-pyrazol-4-yl)-N- (pyridin-3-ylmethyl)nicotinamide as a pale yellow solid (0.79 g, 62%). LRMS (M+H+) m/z 417.1.
Method B:
VIII
[0250] A mixture of 6-chloro-2-(3-fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide (prepared according to example 1, method B) (152 mg, 0.39 mmol), borolane (139 mg, 0.47 mmol), Pd(PPh3J4, and K2CO3 in DMF was heated at microwave reactor at 1250C for 1 h. The mixture was filtered on a silica gel plug using EtOAc as eluent. The filtrate was concentrated
and purified on RP-HPLC using a mixture of acetonitrile and water to give 2-(3- f luorophenethylamino)-6-(1 H-pyrazol-4-yl)-N-(pyridin-3-ylmethyl)nicotinamide (60.5 mg,
37%). LRMS (M +H+-BoC) m/z 417.1.
Example 9:
Λf-((2-(aminomethyl)pyridin-3-yl)methyl)-2-(3-fluorophenethylamino)-6-(l//-pyrazol-4-yl)nicotinamide
[0251] To a mixture of 6-chloro-2-(3-fluorophenethylamino)nicotinic acid (prepared from example 8, method A) (0.72 g, 2.21 mmol) in DMF (5 mL) was added DIEA (0.417 mL, 2.53 mmol), HBTU (0.84 g, 2.21 mmol), HOBt (0.30 g, 2.21 mmol), then amine tert-butyl (3- (aminornethyl)pyridin-2-yl)methylcarbamate (preparations accordint to example 2) (0.50 g, 2.11 mmol). The reaction mixture was stirred at room temperature for 1 hr. The crude mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O to give tert-butyl (3-((2-(3-
fluorophenethylamino)-6-(1 H-pyrazol-4-yl)nicotinamido)methyl)pyridin-2-yl)methylcarbamate as a pale yellow solid (737 mg, 60%). LRMS (M+H+) m/z 546.3.
[0252] To a solution of tert-butyl (3-((2-(3-fluorophenethylamino)-6-(1H-pyrazol-4- yl)nicotinamido)rnethyl)pyridin-2-yl)methylcarbamate (737 mg, 1.27 mmol) in methanol (6 ml_) at O0C was added a solution of 4M HCI in 1 ,4-dioxane (3 ml_). The mixture was warmed to room temperature and stirred for 2hrs. Concentration of the mixture followed by the purification on RP-HPLC using a mixture of acetonitrile and H2O gave N-((2-(aminomethyl)pyridin-3- yl)methyl)-2-(3-fluorophenethylamino)-6-(1 H-pyrazol-4-yl)nicotinamide compound with methanol (1:1) as a pale yellow solid (545 mg, 89%). LRMS (M+H+) m/z 446.2.
Example 10:
Λ^-((2-(aminomethyl)pyridin-3-yI)methyl)-5-fluoro-2-(3-fluorophenethylamino)-6-(l//-pyrazoI-4-yl)nicotinamide
[0253] A mixture of 2,6-dichloro-5-fluoronicotinic acid (1.26 g, 6.0 mmol), 3-fluorophenethyl amine (1.25 ml_, 9.0 mmol), DIEA (3.14 ml_, 18 mmol) and MeCN (6 ml.) was refluxed for 48 hrs and concentrated. The residue was dissolved in EtOAc (120 ml_) and washed with saturated citric acid, water, brine, dried and concentrated. The organic layers were combined, dried (MgSO4) and concentrated. The residue was purified on RP-HPLC using a mixture of acetonitrile and H2O to give 6-chloro-5-fluoro-2-(3-fluorophenethylamino)nicotinic acid as a solid (920 mg, 49%). LRMS (M+H+) m/z 313.0.
[0254] A mixture of 6-chloro-5-fluoro-2-(3-fluorophenethylamino)nicotinic acid (2.2 g, 7.04 mmol), 4,4,5,5-tetramethyl-2-(1 H-pyrazol-4-yl)-1,3,2-dioxoborolane (2.04 g, 10.56 mmol), Pd(PPh3)4 (0.81 g, 0.71 mmol), K2CO3 (2.92g, 21.1 mmol) and anhydrous DMF (10 mL) was degassed with nitrogen for 10 min. The mixture was then stirred at 150 0C in microwave for 3 hrs. The mixture was filtered through the Celite and washed with DMF (50 mL). The filtrate was adjusted to pH 4 with 2N HCI (5 mL), and diluted with EtOAc (150 mL). The organic layer was washed with saturated ammonium chloride, brine and concentrated to dryness. The residue was triturated with dichloromethane (30 mL). Filtration followed by washed with EtOAc (5 mL) gave 5-fluoro-2-(3-fluorophenethylamino)-6-(1 H-pyrazol-4-yl)nicotinic acid as a yellow solid (2.1 g, 87%). LRMS (M+H+) m/z 345.3.
[0255] Compound X was prepared from 5-fluoro-2-(3-fluorophenethylamino)-6-(1 H-pyrazol-4- yl)nicotinic acid and amine tert-butyl (3-(aminomethyl)pyridin-2-yl)methylcarbamate according to the procedure of preparation of Example 9. LRMS (M+H+) m/z 464.1 ,
Example 11:
Xl
(R)-5-fluoro-2-(3-fluorophenethylamino)-/V-((1-(2-hydro>^acetyl)azetidin-2-yl)methyl)-6-(1-methyl-1H- pyrazol-4-yl)nicotinamide
[0256] A mixture of 6-chloro-5-fluoro-2-(3-fluorophenethylamino)nicotinic acid (see example 10) (15.6g, 50 mmol), borolaπe (12.5g, 60 mmol), Pd(PPh3)4 (5.8 g, 5 mrnol), K2CO3 (20.7g, 150 mmol) and anhydrous DMF (100 ml_) was degassed with nitrogen for 10 min. The mixture was then stirred at 1250C in microwave for 2 hrs. The mixture was filtered through the Celite and washed with DMF. The filtrate was adjusted to pH 4 with 2N HCI, and diluted with EtOAc. The organic layer was washed with brine twice and concentrated to dryness. The residue was triturated with EtOAc (200 ml_). Filtration followed by washed with EtOAc (20 ml_) gave 5-fluoro- 2-(3-fluorophenethylamino)-6-(1-methyl-1H-pyrazol-4-yl)nicotinic acid as a yellow solid (14.5 g, 85%). LRMS (M+H+) m/z 359.0.
[0257] To a mixture of D-azetidine-carboxylic acid (20 g, 200 mmol), H2O (200 mL) and dioxane (200 ml) at 00C was added a solution of BoC2O in dioxane (100 mL) with an additional funnel. The mixture was stirred at r.t for 1 hour. After concentration, 200 mL ether and 200 mL H2O were added to the residue. The aqueous layer was acidified with 2N HCI to pH 3-4, and then extracted with 200 mL EtOAc. The organic layer was dried and concentrated to give (R)-1- (tert-butoxycarbonyl)azetidine-2-carboxylic acid in quantitative yield (4Og). LRMS (M-100+H*) m/z 102.1.
[0258] To a solution of 2M LiBH4 (200 ml, 400mmol) in THF at 00C was added TMSCI (101ml, 800 mmol) slowly. The mixture was warmed up to r.t and stirred at r.t. for 30 min. To the mixture at 0 0C was added a solution of (R)-1-(tert-butoxycarbonyl)azetidine-2-carboxylic acid (4Og, 200 mmol) in 200 ml THF with an additional funnel over 1 hour. The mixture was warmed up to r.t and stirred at r.t. for 2 hours. The mixture was cooled down to 0 0C and then treated with 32 mL MeOH followed by 40 mL H2O. After removal of THF, the residue was extracted with
300 mL EtOAc and washed with 300 ml_ brine. The organic layer was dried and concentrated to give (R)-tert-butyl 2-(hydroxymethyl)azetidine-1-carboxylate as a colorless oil (32.5g, 87%). LRMS (M-100+H+) m/z 132.1.
[0259] To a mixture of (R)-tert-butyl 2-(hydroxymethyl)azetidine-1-carboxylate (32.5g, 173 mmol) and Et3N (27 mL, 191mmol) in THF (650 mL) at 0 0C was added methane sulfonylchloride (14.9 ml, 191 mmol) dropwise. The mixture was stirred for 10 min. The precipitation was filtered off and the filtrate was concentrated. The residue was then dissolved with DMF (200 mL). Sodium azide (34g, 519 mmol) was then added. The mixture was stirred at 65 0C for 6 hours. 300 mL EtOAc and 300 mL brine were then added to the reaction mixture. The organic layer was dried and concentrated. The residue was purified by Biotage silica gel chromatography (EtOAc: Hexane 1 :4) to give (R)-tert-butyl 2-(azidomethyl)azetidine-1- carboxylate as a colorless oil (32g, 87% yield). LRMS (M-100+H*) m/z 113.1.
[0260] A mixture of (R)-tert-butyl 2-(azidomethyl)azetidine-1-carboxylate (17g, 80 mmol), 10% Pd/C (2g) and MeOH (15OmL) was stirred under 45 psi hydrogen for 1 hour. The catalyst was filtered off and the filtrate was concentrated to give (R)-tert-butyl 2-(aminomethyl)azetidine-1- carboxylate (14.9 g, yield 100%). LRMS (M+H+) m/z 187.3.
[0261] To a mixture of 5-fluoro-2-(3-fluorophenethylamino)-6-(1-methyl-1H-pyrazol-4-yl)nicotinic acid (14.3 g, 40 mmol) and DMF (30OmL) was added of HBTU (18.2 g, 48 mmol) and HOBT (6.49 g, 48 mmol). The mixture was cooled down to 00C, 8.93 g (48 mmol) of (R)-tert-butyl 2- (aminomethyl)azetidine-i-carboxylate was added, followed by 13.9 ml of DIEA (80mmol). The mixture was stirred at r.t for 1 hour and extracted with 300 mL EtOAc. The organic layer was washed with 500 mL brine, dried over MgSO4 and then concentrated. The residue was purified by Biotage silica gel chromatography using EtOAc:Hex (1:1) as eluant. (R)-tert-butyl 2-((5- fluoro^-tS-fluorophenethylaminoJ-e-ti-methyl-IH-pyrazoM-ylJnicotinamidoJmethyOazetidine-i- carboxylate was obtained as a yellow solid (16.5 g, yield 80%). LRMS (M+H+) m/z 527.1.
Xl
[0262] To a mixture of (R)-tert-butyl 2-<(5-fluoro-2-(3-fluorophenethylamino)-6-(1-methyl-1H- pyrazol-4-yl)nicotinamido)methyl)azetidine-1-carboxylate (16.5g, 31.4mmol) in 100 mL DCM at 00C was added trifluoroacetic acid (24 mL, 314 mmol) slowly. The mixture was stirred at r.t for 2 hours until the Boc protection group was completely removed. The mixture was then concentrated. The residue was dissolved in 100 mL DMF and cooled down to 0 0C. To the mixture was added a mixture of glycolic acid (2.86g, 38mmol), HBTU (14.3g, 38mmol), HOBT (5.1g, 38mmol) and DIEA (16.4ml, 94mmol). The mixture was warmed up to r.t and stirred at r.t. for 2 hours. The mixture was extracted with EtOAc. The organic layer was further washed with 1N NaOH (300ml) and brine, dried and concentrated. The residue was subjected to a Biotage silica gel chromatography using gradient 0-40% B as eluant (A = dichloromethane, B = 10% MeOH in dichloromethane) to give the crude product. The yellow solid was further tritirated with 100 mL EtOAc. (R)-5-fluoro-2-(3-fluorophenethylamino)-N-((1-(2- hydroxyacetyl)azetidin-2-yl)methyl)-6-(1-methyl-1H-pyrazol-4-yl)nicotinamide was obtained as a pale yellow solid (12.7g, 83% yield). LRMS (M+H+) m/z 485.1.
Example 12:
Λ^-(((Λ)-l-(4-aminobutanoyI)pyiτolidin-2-y])πiethyl)-6-(2-cyanophenyl)-2-(2-(tetrahydro-2//-pyran-2-yl)ethylaniino)
iii. TFA/ CH2CI2
[0263] A mixture of 2,6-dichloronicotinic acid (1.0 g, 5.21 mmol), N,N'-carbonyldiimidazole (CDI, 1.01g, 6.25 mmol) and THF (10 mL) was stirred at 600C for 30 min and cooled to room temperature. To the mixture was added (R)-tert-butyl 2-(aminomethyl)pyrrolidine-1-carboxylate (1.04 g, 6.25 mmol) and DIEA (1.03 mL, 5.21 mmol). The reaction mixture was stirred at room temperature for 4 hrs, and diluted with EtOAc. The organic layer was washed with H2O, brine, dried over Na2SO4. Concentration of the organic solution followed by treatment with 30% TFA in dichloromethane (10 mL) gave (R)-2,6-dichloro-N-(pyrrolidin-2-ylmethyl)nicotinamide TFA salt as a white solid (1.7 g, 84%). LRMS (M+H+) m/z 274.0.
[0264] To a mixture of 4-(benzyloxycarbonylamino)butanoic acid (1.29 g, 5.45 mmol) in DMF (4 mL) was added HBTU (2.07 g, 5.45 mmol), DIEA (3.0 mL, 18.17 mmol), then amine (R)-2,6- dichloro-N-(pyrrolidin-2-ylmethyl)nicotinamide (1.7 g, 4.54 mmol). The reaction mixture was stirred at room temperature for 1 hr. The crude mixture was purified by silica gel chromatography using a mixture of EtOAC-MeOH (19:1) to give (R)-benzyl 4-(2-((2,6- dichloronicotinamidoJmethylJpyrrolidin-i-yO^-oxobutylcarbamate (1.9 g, 85%). LRMS (M+H+) m/z 493.4.
[0265] A mixture of chloride (R)-benzyl 4-(2-((2,6-dichloronicotinamido)methyl)pyrrolidin-1-yl)-4- oxobutylcarbamate (150 mg, 0.304 mmol), 2-(tetrahydro-2H-pyran-2-yl)ethanamine HCI salt (76 mg, 0.459 mmol), K2CO3 (125 mg, 0.905 mmol) and DMF (1 mL) was stirred at 10O0C for 10 hrs. The crude mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O to give benzyl 4-((R)-2-((6-chloro-2-(2-(tetrahydro-2H-pyran-2- yl)ethylamino)nicotinamido)methyl)pyrrolidin-1-yl)-4-oxobutylcarbamate (62 mg, 35%). LRMS (M+H+) m/z 586.3.
[0266] A mixture of benzyl 4-((R)-2-((6-chloro-2-(2-(tetrahydro-2H-pyran-2- yl)ethylamino)nicotinamido)methyl)pyrrolidin-1-yl)-4-oxobutylcarbamate (62 mg, 0.101 mmol), 2- cyanophenyl boronic acid (23 mg, 0.156 mmol), PdCI2(dppf)2 (11 mg, 0.02 mmol), K2CO3 (92 g, 0.667 mmol) and anhydrous DMF (1 ml_) was stirred at 1250C for 1 hr. The mixture was filtered through the silica gel pad and washed with EtOAc. The filtrate was concentrated to dryness and purified on RP-HPLC using a mixture of acetonitrile and H2O to give benzyl 4-((R)-2-((6-(2- cyanophenyl)-2-(2-(tetrahydro-2H-pyran-2-yl)ethyfamino)nicotinamido)methyl)pyrrolidin-1-yl)-4- oxobutylcarbamate (50 mg, 77%). LRMS (M+H+) m/z 653.3.
XII
[0267] To a solution of benzyl 4-((R)-2-((6-(2-cyanophenyl)-2-(2-(tetrahydro-2H-pyran-2- yl)ethylamino)nicotinamido)methyl)pyrrolidin-1-yl)-4-oxobutylcarbamate (47 mg, 0.072 mmol) in MeCN (1 mL) at 00C was added TMSI (72 mg, 0.36 mmol). The mixture was stirred at room temperature for 2 hrs and purified on RP-HPLC using a mixture of acetonitrile and H2O to give N-({(R)-1-(4-aminobutanoyl)pyrrolidin-2-yl)methyl)-6-(2-cyanophenyl)-2-(2-(tetrahydro-2H- pyran-2-yl)ethylamino)nicotinamide (36 mg, 77%). LRMS (M+H+) m/z 519.3.
Example 13
[0268] Using procedures similar to those described herein, the compounds in the following table were synthesized and tested.
IC50
Arithmetic
Mean Ion m/z CHEMICAL NAME
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-prop-2-
3.123 M+H 359.2 enylcarboxamide
N-methyl(2-{[2-(3-methylpheπyl)ethyl]amino}-6-phenyl(3-
6.747 M+H 346.2 pyridyl))carboxamide
N-methyl{6-phenyl-2-[(2-phenylethyl)amino](3-
18.628 M+H 332.2 pyridyl)}carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N-prop-2-
1.062 M+H 358.2 enylcarboxamide
(2-{[2-(3-methylphenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
3.74 M+H 372.3 prop-2-enylcarboxamide
N-methyl{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-
27.729 M+H 333.2 pyridyl)}carboxamide
N-benzyl{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-
1.564 M+H 409.2 pyridyl)}carboxamide
{6-phenyl-2-[(2-phenylethyl)amiπo](3-pyridyl)}-N-(3-
0.151 M+H 409.2 pyridylmethyl)carboxamide
N-(methylethyl){6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-
13.296 M+H 361.3 pyridyl)}carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(2-
0.463 M+H 410.1 pyridylmethyijcarboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(4-
1.29 M+H 410.2 pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyϊ)ethyl)amino](3-pyridyl)}-N-(3-
5.373 M+H 396.2 pyridyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(2-
7.189 M+H 396.2 pyridyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(4-
5.209 M+H 396.2 pyridyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(2-
1.721 M+H 415.2 thienylmethyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(3-
0.683 M+H 415.2 thienylmethyl)carboxamide
N-(oxolan-2-ylmethyl){6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-
11.022 M+H 403.3 pyridyl)}carboxamide
N-[((2S)oxolan-2-yl)methyl]{6-pheπyl-2-[(2-(2-
6.821 M+H 403.2 pyridyl)ethyl)amino](3-pyridyl)}carboxamide tert-butyl 2-[({6-phenyl-2-[(2-(2-pyridyl)ethyl)amino]-3-
65.098 M+H 518.2 pyridyl}carbonylamino)methyl]morpholine-4-carboxylate
N-[(1-acetyl(4-piperidyl))methyl]{6-phenyl-2-I(2-(2-
96.68 M+H 458.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[( 1 -acetyl(3-piperidyl))methy l]{6-phenyl-2-[(2-(2-
48.434 M+H 458.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(4-
43.709 M+H 416.3 piperidylmethyl)carboxamide
^-^-cyclohex-i-enylethyOaminoJ-e-phenylCS-pyridylJJ-N^S-
0.532 M+H 413.2 pyridylmethyl)carboxamide
{6-phenyl-2-[(3-phenylpropyl)amino](3-pyridyl)}-N-(3-
5.689 M+H 423.3 pyridylmethyl)carboxamide
{6-phenyl-2-lbenzylamino](3-pyridyl)}-N-(3-
2.792 M+H 395.2 pyridy!methyl)carboxamide
4.897 M+H 400.2 N-(1 ,3-oxazol-2-ylmethyl){6-pheπyl-2-[(2-(2-
pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-(imidazol-2-ylmethyl){6-phenyl-2-[(2-(2-
6.148 M+H 399.1 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((2S)-1-acetylpyrrolidiπ-2-yl)methyl]{6-phenyl-2-[(2-(2-
15.715 M+H 444.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((3R)-1-acetylpyrrolidin-3-yl)methyl]{6-phenyl-2-[(2-(2-
26.904 M+H 444.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((3S)-1-acetylpyrrolidin-3-yl)methyl]{6-phenyl-2-[(2-(2-
25.159 M+H 444.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide methyl 2-({6-phenyl-2-[(2-phenylethyl)amino]-3-
1.335 M+H 390.2 pyridyljcarbonylami no)acetate
[6-phenyl-2-(propylamino)(3-pyridyl)]-N-(3-
11.377 M+H 347.1 pyridylmethyl)carboxamide
{2-[(3-methylbutyl)amino]-6-phenyl(3-pyridyl)}-N-(3-
2.484 M+H 375.2 pyridylmethyl)carboxamide
{2-[(2-cyclohexylethyl)amino]-6-phenyl(3-pyridyl)}-N-{3-
1.854 M+H 415.3 pyridylmethyl)carboxamide
N-{[1-(methylsulfonyl)(3-piperidyl)]methyl}{6-phenyl-2-[(2-(2-
20.292 M+H 494.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
2-(aminomethyl)piperidyl 6-phenyl-2-[(2-(2-
18.23 M+H 416.3 pyridyl)ethyt)amino](3-pyridyl) ketone
N^[1-({6-phenyl-2-[(2-(2-pyridyl)ethyl)amino]-3-
21.154 M+H 458.3 pyridyl}carbonyl)-2-piperidyl]methyl}acetamide
2-{[(methylsulfonyl)amino]methyl}piperidyl 6-pheπyl-2-[(2-(2-
22.226 M+H 494.3 pyridyl) ethyl)ami no](3-pyridy I) keton e
N-[(1-methylpyrazol-5-yl)methyl]{6-phenyl-2-[(2-(2-
1.298 M+H 413.2 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[(1-methylpyrazol-3-yl)methyl]{6-phenyl-2-[(2-(2-
2.733 M+H 413.2 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(pyrazol-3-
0.711 M+H 399.2 ylmethyl)carboxamide
N-(isoxazol-5-ylmethyl){6-pheny!-2-[(2-(2-
0.836 M+H 400.2 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((3S)pyrrolidin-3-yl)methyl]{6-phenyl-2-[(2-(2-
62.553 M+H 402.3 pyridyl)ethyl)amino](3-pyridyl)}carboxamide
N-[((3R)pyrrolidin-3-yl)methyl]{6-pheπyl-2-[(2-(2-
53.616 M+H 402.3 pyridyl)ethyl)amiπo](3-pyridyl)}carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N-(2-(2-
5.245 M+H 423.3 pyridyl)ethyl)carboxamide
{6-phenyl-2-[(2-phenylethyl)amino](3-pyridyl)}-N-(2-(3-
3.093 M+H 423.2 pyridyl)ethyl)carboxamide
N-methyl-2-({6-phenyl-2-[(2-phenylethyl)amino](3-
0.486 M+H 389.2 pyridyl)}carbonylamino)acetamide
N,N-dimethyl-2-({6-phenyl-2-[(2-phenylethyl)amino](3-
30.309 M+H 403.3 pyridyl)}carbonylamino)acetamide
N-(carbamoylmethyl){6-pheπyl-2-[(2-phenylethyl)amino](3-
0.81 M+H 375.2 pyridyl)}carboxamide
(2-{[2-(2-oxopiperidyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
22.145 M+H 430.3 pyridylmethyl)carboxamide
(2-{[2-(1-methyl(4-piperidyl))ethyl]amino}-6-phenyl(3-pyridyl))-
20.145 M+H 430.3 N-(3-pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(2-thienyl)ethy!)amino](3-pyridyl)}-N-(3-
0.149 M+H 415.2 pyridylmethyl)carboxamide
32.174 M+H 413.2 (2-{t2-(1-methylimidazol-4-yl)ethyl]amino}-6-pheny!(3-pyridyl))-
N-(3-pyridylmethyl)carboxamide
{2-[(2-(2-furyl)ethyl)amiπo]-6-pheπyl(3-pyridyl)}-N-(3- 0.294 M+H 399.2 pyridylmethyljcarboxamide
{2-[(oxolan-2-ylmethyl)amino]-6-phenyl(3-pyridyl)}-N-(3- 4.835 M+H 389.2 pyridylmethyl)carboxamide
{2-[(2-( 1 , 3-dioxolan-2-yl)ethyl)amino]-6-phenyl(3-pyridyl)}-N-(3- 7.122 M+H 405.2 pyridylmethyl)carboxamide
{2-[(3-imidazolylpropyl)amino]-6-phenyl(3-pyridyl)}-N-(3- 30.307 M+H 413.3 pyridylmethyljcarboxamide
{2-[(2-morpholin-4-ylethyl)amino]-6-pheπyl{3-pyridyl)}-N-(3- 37.33 M+H 418.3 pyridylmethyl)carboxamide
{6-phenyl-2-[(2-phenoxyethyl)amino](3-pyridyl)}-N-(3- 20.164 M+H 425.2 pyridylmethyl)carboxamide
{2-[(2-(2H-benzo[3,4-d]1,3-dioxolen-5-yl)ethyl)amino]-6- 28.307 M+H 453.2 phenyl(3-pyridyl)}-N-(3-pyridylmethyl)carboxamide
(2-{[2-(2-oxoimidazolidinyl)ethyl]amino}-6-phenyl(3-pyridyl))-N- 70.65 M+H (3-pyridylmethyl)carboxamide
(2-{[2-(2-oxopyrrolidinyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3- 10.62 M+H 416.2 pyridylmethyl)carboxamide
{2-[(2-imidazol-4-ylethyl)amino]-6-phenyl(3-pyridyl)}-N-(3- 9.766 M+H 399.2 pyridylmethyl)carboxamide
(^-{[(hydroxycyclohexylJmethyllaminoJ-β-phenyKS-pyridyOJ-N- 7.811 M+H 417.3 (3-pyridylmethyl)carboxamide
(2-{[2-(3,5-dimethylpyrazol-4-yl)ethyl]amino}-6-phenyl(3- 5.941 M+H 427.2 pyridyI))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3,5-dimethylpyrazolyI)ethyl]amino}-6-phenyl(3-pyridyl))- 6.833 M+H 427.2 N-(3-pyridylmethyl)carboxamide
(2-{[2-(2-chlorophenyl)ethyl]amino}-6-(2-cyanophenyl)(3- 0.028 M+H 468.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(2-methylphenyl)ethyl]amino}(3- 0.076 M+H 448.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(2-fluorophenyl)ethyl]amino}(3- 0.028 M+H 452.2 pyridyI»-N-(3-pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(2-cyanophenyl)ethyl]amino}(3- 0.044 M+H 458.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
{6-(2-cyanophenyl)-2-[(2-(2-thienyl)ethyl)amiπo](3-pyridyl)}-N- 0.029 M+H 440.2 (3-pyridylmethyl)carboxamide
{6-(2-cyanophenyl)-2-[(2-(2-furyl)ethyl)amino](3-pyridyl)}-N-(3- 0.065 M+H 424.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluoro(2-pyridyl))ethyl]amino}-6-prιenyl(3-pyridyl))-N- 1.479 M+H (3-pyridylmethyl)carboxamide
^-(P-O-methylphenyOethyllaminoJ-θ-phenylCS-pyridyOJ-N-CS- 0.45 M+H 423.3 pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(2-hydroxyphenyl)ethyl]amino}(3- 0.38 M+H 450.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methylK6-pyrazol-4-yl-2-[(2-(2- 1.238 M+H 429.1 pyridyl)ethyl)amino3(3-pyridyl)}carboxamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)-2- hydroxyethyl]amino}-6-pyrazol-4-yl(3-pyridyl))carbonylamino]- 14.214 M+H 574.3 isopropyl}-2-[(tert-butoxy)carbonylamino]acetamide
{5-fluoro-6-pyrazol-4-yl-2-[(2-pyrazolylethyl)amino](3-pyridyl)}- 0.211 M+H 407.3 N-(3-pyridylmethyl)carboxamide
N-[( 1 R)-2-({5-fluoro-6-pyrazol-4-yl-2-[(2- 4.635 M+H 430.3 pyrazolylethyl)amino](3-pyridyl)}carbonylamino)-isopropyl]-2-
aminoacetamide
(6-(2-cyanophenyl)-2-{[2-(3-flubrophenyl)ethyl]amino}(3- 0.604 M+H 520.1 pyridyl))-N-{[6-(trifluoromethyl)(2-pyridyl)]methyl}carboxarriide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 1.497 M+H 520.1 pyridyl))-N-{[5-(trifluoromethyl)(2-pyridyl)]methyl}carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.603 M+H 520.1 pyridyl))-N-{[3-(trifluoromethyl)(2-pyridyl)]methyl}carboxamide
N-[(6-chloro(3-pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3- 1.789 M+H 486.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyano(3-thienyl))-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.033 M+H 458.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2E)-3-(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))- 57.378 M+H 376.1 N-methylprop-2-enamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethy!]amino}(3- 0.298 M+H 481.2 pyridyl))-N-{[6-(methylamino)(3-pyridyl)]methyl}carboxamide
N-{[6-(dimethylamino)(3-pyridyl)]methyl}(6-(2-cyanophenyl)-2- 30.234 M+H 495.2 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2-{[2-(3-fluoropheπyl)ethyl]amino}-4-(hydroxymethyl)-6- 59.114 M+H 457.2 phenyl(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
N-[(5-amino(2-pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3- 0.021 M+H 467.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2-{[2-(3-fIuorophenyl)ethyl]amino}-5-(hydroxymethyl)-6- 0.625 M+H 457.1 phenyl(3-pyridyl))-N-(3-pyridyImethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl-5-[N-(3- pyridylmethyl)carbamoyl](3-pyridyl))-N-(3- 55.988 M+H 561.2 pyridylmethyl)carboxamide
(5-(2-diazo-2-azavinyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-6- 0.428 M+H 482.2 phenyl(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(5-(aminomethyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-6- 8.904 M+H 456.1 phenyl(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-3-yl)(3- 0.04 M+H 431.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
N-({6-[3, 3-bis(dimethylamino)-2-azaprop-2-enyl](2- pyridyl)}methyl)(6-(2-cyanophenyl)-2-{[2-(3- 0.285 M+H 579.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide tert-butyl 2-(6-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3-pyridyI)carboπylamino]methyl}-2- 1.189 M+Na 603.2 pyridyl)acetate
N-{[4-(aminomethyl)(3-pyridyl)]methyl}(6-(2-cyanophenyl)-2- 0.401 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[3-(aminomethyl)(4-pyridyl)]methyl}(6-(2-cyanophenyl)-2- 0.056 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3- 0.021 M+H 497.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(1- 0.023 M+H 509.1 methylpyra2ol-4-yl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
{6-(5-cyanopyrazol-4-yl)-2-{[2-(3-fluorophenyl)ethyl]arnino}(3- 0.105 M+H 442.1 pyridyl))-N-(3-pyridylmethyl)carboxa.mide
(5-cyano-2-[[2-(3-fluorophenyl)ethyl]amino}-6-(1- 0.021 M+H 456.1 methylpyrazol-4-yl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyra∑ol-3-yl(3-pyridyl))-N- 0.124 M+H 417.1 (3-pyridylmethyl)carboxamide
(6-(3-cyano-1-methylpyrazol-4-yl)-2-{[2-(3- 0.047 M+H 456.1 fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
(6-(5-cyano-1-methylpyrazol-4-yl)-2-{[2-(3- fluorophenyl)ethy]]amino}(3-pyridyl))-N-(3-
0.02 M+H 456.1 pyridylmethyl)carboxamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.328 M+H 397.0 pyridyl))carbonylamino]-N-methy1acetamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
0.35 M+H 411.1 yl)(3-pyridyl))carbonylamino]-N-methylacetamide
(δ-chloro^-tP-CS-fluorophenyOethyllaminoJ-δ-CI-
0.021 M+H 465.1 methylpyrazol-4-yl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]tnethyl}(5-bromo-2-{[2-(3-
0.604 M+H 536.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
N-[(1-{2-[(tert-butoxy)carboπylamino]ethyl}pyrazol-3- yl)rnethyl](6-(2-cyanophenyl)-2-{[2-(3-
2.725 M+Na 606.1 fluorophenyl)ethyl]amino}(3-pyridy1))carboxamide
N-[(1-{2-[(tert-butoxy)carbonylamino]ethyl}pyrazol-5- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
2.2 M+H 584.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-{2-aminoethyl)pyrazol-3-yl]methyl}(6-(2-cyanophenyl)-2-
0.296 M+H 484.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-5-yl]methyl}(6-(2-cyanophenyl)-2-
0.032 M+H 484.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.136 M+H 460.1 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.121 M+H 446.1 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(5-bromo-2-{[2-(3- fIuoropheπyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.027 M+H 539.1 pyridyl))carboxamide
N-[((2R)-5-oxopyrro!idin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-
0.013 M+H 458.1 (3-fiuorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)-5-oxopyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-
0.133 M+H 458.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{3-[(5S)-5-(2-diazo-2-azavinyl)-2-oxopyrrolidinyl]propyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
16.552 M+H 541.1 pyridyi))carboxam ide
N-[((2S)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-{{2-(3-
4.516 M+H 615.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{t(2S)-1-(2-aminoethyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-([2-(3-fluorophenyl)ethyl]amino}(3-
0.917 M+H 501.1 pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.431 M+H 515.2 pyridyl))carboxamide
N-{3-[(5S)-5-(aminomethyl)-2-oxopyrrolidinyl]propyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
11.658 M+H 515.1 pyridyl))carboxamide
N-{[(2R)-1-(2-aminoethyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amiπo}(3-
0.11 M+H 501.1 pyridyl))carboxamide
5.621 M+H 541.2 N-{3-[(5R)-5-(2-diazo-2-azavinyl)-2-oxopyrrolidinyl]propyl}(6-(2-
cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))carboxamide
N-{3-[(5R)-5-(aminomethyl)-2-oxopyrrolidinyl]propyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
4.951 M+H 515.1 pyridyl))carboxamide
N-{2-[(5S)-5-(aminomethyl)-2-oxopyrrolidinyl]ethyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
17.87 M+H 501.1 pyridyl))carboxamide
N-{2-[(5R)-5-(aminomethyl)-2-oxopyrrolidinyl]ethyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
19.454 M+H 501.1 pyridyl))carboxamide
N-[((2R)-5-oxopyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
0.355 M+H 423.1 pyridyl))carboxamide
N-[((2R)-5-oxopyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.156 M+H 437.1 pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.439 M+H 444.0 pyridyl))-N-(pyrrolidin-2-ylmethyl)carboxamide N-[((2R)-5-oxopyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.119 M+H 423.1 pyridyl))carboxamide
N-[(1-{2-[(tert-butoxy)carbonylamiπo]ethyl}pyrrolidin-2- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
4.834 M+H 587.1 f luorophenyl)ethyl]amino}(3-pyridyl))carboxamide N-[(1-{3-[(tert-butoxy)carbonyIamino]propyl}pyrrolidin-2- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
2.082 M+H 601.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide N-{[1-(2-aminoethyl)pyrrolidin-2-yl]methyl}(6-(2-cyanophenyl)-
0.758 M+H 487.1 2-{t2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamϊde N-{[1-(4-aminobutyl)pyrrolidin-2-yl]methyl}(6-(2-cyanophenyl)-
3.502 M+H 501.2 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide methyl 2-({(2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
0.913 M+H 505.1 pyridyl))carbonylamino]propyl}amino)acetate methyl 2-({(2S)-2-amino-3-[(2-{[2-(3-fluorophenyl)ethyl]amino}-
M+H 6-(1-methylpyrazol-4-yl)(3-
10.852 OMe 452.3 pyridyl))carbonylamino]propyl}amino)acetate N-[((2S)-6-oxopiperazin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
16.026 M+H 438.2 pyridyl))carboxamide N-[((2S)-6-oxopiperazin-2-yl)methylj(6-(2-cyanophenyl)-2-{[2-
0.819 M+H 473.1 (3-fIuorophenyl)ethyl]amino}(3-pyridyl))carboxamide N-[((2R)-4-{3-[(tert-butoxy)carbonylamino]propyl}-6-
M+H oxopiperazin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
9.536 Boc 530.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide N-[((2S)-6-oxopiperazin-2-yl)methyl](2-{t2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
13.056 M+H 452.1 pyridyl))carboxamide
N-[((2S)-6-oxopiperazin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
6.391 M+H 438.1 pyridyl))carboxamide N-{[(2R)-4-(3-aminopropyl)-6-oxopiperazin-2-yl]methyl}(6-(2-
4.522 M+H 530.2 cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carboxam ide
N-{[(2R)-4-(2-aminoethyl)-6-oxopiperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 8.4722 M+H 516.2 pyridyl))carboxamide
N-[((2R)-6-oxopiperazin-2-yl)methyl]{6-(2-cyanopheπyl)-2-{[2- 0.155 M+H 473.1 (3-fluoropheny1)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-4-(2-aminoethyl)-6-oxopiperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.623 M+H 515.2 pyridyl))carboxamide
N-{[(2S)-4-(3-aminopropyl)-6-oxopiperazirv2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorOphenyl)ethyl]amino}(3- 2.809 M+H 530.2 pyridyl))carboxamide
N-({(2R)-1-[4-(1,3-dioxobenzo[c]azolin-2-yl)butyl]-5- oxαpyrrolidin-2-yl}methyl)(6-(2-cyanophenyl)-2-{[2-(3- 0.376 M+H 659.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({(2R)-1-[5-(1 ,3-dioxobenzo[c]azolin-2-yl)pentyl]-5- oxopyrrolidin-2-yl}methyl)(6-(2-cyanophenyl)-2-{[2-(3- 0.266 M+H 673.2 fluorophenyl)ethyl)amino}(3-pyridyl))carboxamide tert-butyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 1.933 M+H 544.2 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-{[(2R)-1-(4-aminobutyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.0517 M+H 529.2 pyridyl))carboxamide
N-{[(2R)-1-(5-aminopentyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.039 M+H 543.2 pyridyl))carboxamide
N-[((2R)pyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3- 0.1738 M+H 444.1 fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
(2-{[2-{3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N- 0.034 M+H 406.1 (pyrazol-3-ylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.031 M+H 420.1 pyridyl))-N-(pyrazol-3-ylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-pyridyl))-N- 0.092 M+H 406.1 (pyrazol-3-ylmethyl)carboxamide
N-[((2R)-H4-[(tert-butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3- 0.432 M+H 629.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{t(2R)-1-(3-aminopropanoyl)pyrrolidiπ-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.047 M+H 515.2 pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-3-yl]methyl}(6-(2-cyanophenyl)-2- 0.065 M+H 498.2 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[i-(3-aminopropyl)pyrazol-3-yl]methyl}(2-{[2-(3- f luorophenyl)ethyl]amiπo}-6-( 1 -methylpyrazol-4-yl)(3- 2.686 M+H 477.1 pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-3-yl]methyl}(2-{[2-(3- fluorophenyI)ethyl]amiπo}-6-pyrazol-3-yl(3- 3.764 M+H 463.2 pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazo!-5-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazoi-4-yl)(3- 0.228 M+H 477.2 pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-5-yl]methyl}(2-{[2-(3- 0.462 M+H 463.2 fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]ethyl}pyrrolidin-2- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
3.691 M+H 587.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}pyrrolidin-2- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
1.341 M+H 601.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamidβ
N-{[(2R)-1-(2-aminoethyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl )ethy l]am ino}( 3-
0.705 M+H 487.2 py ridy l))carboxam ide
N-{[(2R)-1-(3-aminopropyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.389 M+H 501.2 pyridyl))carboxamide
N-[(1-{2-[(tert-butoxy)carbonylamino]ethyl}pyrazol-3- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
11.966 M+H 549.2 pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-3-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
1.488 M+H 449.1 pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-3-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
5.506 M+H 449.2 pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-5-yl]methyl}(2-{t2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
0.634 M+H 449.2 pyridyl))carboxamide
N-{[1-(2-aminoethyl)pyrazol-3-yl3methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-{1-methylpyrazol-4-yl)(3-
1.862 M+H 463.2 pyridyl))carboxamide
N-{[1-(2-amiπoethyl)pyrazol-5-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-( 1 -methylpyrazol-4-yl)(3-
0.313 M+H 463.3 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxopiperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.318 M+H 530.3 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxopiperazin-2-yl]methyl}(2-{I2-
(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
3.562 M+H 509.3 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxopiperazin-2-yl]methyl}(2-{[2-
(3-fiuorophenyl)ethyi]amino}-6-pyrazol-4-yl(3-
2.343 M+H 495.3 pyridyl))carboxamide
N-{[(2R)-1-(3-amiπopropyl)-6-oxopiperazin-2-yl]methyl}(2-{[2-
(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
9.627 M+H 495.3 pyridyl))carboxamide
N-[((2R)-1-acetylpyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-
0.119 M+H 486.2 {[2-(3-fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-{[(2R)-1-(methylsulfonyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.448 M+H 522.2 pyridyl))carboxamide
N-[((2R)-1-methylpyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-
0.214 M+H 458.3 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1,1-dimethylpyrrolidin-2-yl)nnethyl](6-{2-cyanophenyl)-
2.049 M 472.2 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-{[3-(1 ,3-dioxobenzo[c]azolin-2-
11.341 M+H 695.3 yl)propyl]sulfonyl}pyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({(2R)-1-[(2-aminoethyl)sulfonyl]pyrrolidin-2-yl}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.071 M+H 551.3 pyridyl))carboxamide
N-({(2R)-1-[(3-aminopropyl)sulfonyl]pyrrolidin-2-yl}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.234 M+H 565.3 pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-3-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl<3-
1.406 M+H 463.1 pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrazol-5-yl]methyl}(2-{[2-(3- fluorophenyl)ethy]]amino}-6-pyrazol-4-yl(3-
0.095 M+H 463.1 pyridyl))carboxamide
N-[((2R)-1-{3-[{tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-
1.342 M+H 594.3 (1-methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(6-{1- [(dimethylamino)(dimethylylidene)methyl]pyrazol-4-yl}-2-{[2-(3-
2.127 M+H 578.4 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{t(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(2-{[2- (3-fluorophenyl)ethyl]amino}-6-(1-rrιethylpyrazol-4-yl)(3- 0.29 M+H 494.3 pyridyl))carboxamide tert-butyl (2R)-2-{[(2-{[2-(3-fluoropheny!)ethyl]amino}-6-(1 - methylpyrazol-4-yl){3- 4.64 M+H 523.3 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(2-{[2- (3-fiuorophenyl)ethyl]amino}-6-pyrazo!-4-yl(3-
0.966 M+H 480.2 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrro!idin-2-yl)methyl](6-(2-cyanophenyl)-2-[[2-(3-
0.518 M+H 615.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(2-amino-2-methylpropyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.666 M+H 432.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)pyrrolidir»-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
4.746 M+H 409.2 pyridyl))carboxamide
N-[((2R)pyrrolidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
3.185 M+H 423.2 pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{t2-(3-fIuorophenyl)ethyl]amino}(3- pyridyl))-N-{2-methyl-2-
1.473 M+H 510.3 [(methylsulfonyl)amino]propyl}carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]acetyl}pyrrolidin-2- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
5.532 M+H 580.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carboπyIamino]propanoyl}pyrrolidin- 2-yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
5.026 M+H 594.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
3.651 M+H 608.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{t(2R)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.581 M+H 480.3 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidiπ-2-yl]methyl}(2-{[2-(3- f luorophenyl)ethyl]amino}-6-( 1 -methylpyrazol-4-yl)(3-
0.587 M+H 494.3 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.351 M+H 508.3 pyridyl))carboxamide N-((2S)-2-aminopropyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.515 M+H 418.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide 2-[(tert-butoxy)carbonylamino]-N-(2-[(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-tert-
9.572 M+H 589.4 butyl}acetamide
4-[(tert-butoxy)carbonylamino]-N-{2-[(6-(2-cyanophenyl)-2-{[2- (3-fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-tert-
0.194 M+H 617.4 butyljbutanamide
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{[2-(3. fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
2.019 M+H 603.3 4-[(tert-butoxy)carbonylamino]butanamide N-[((2R)-1-{5-[(tert-butoxy)carbonylamino]pentanoyl}pyrrolidin- 2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
2.94 M+H 643.4 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide 2-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyljamino}(3-pyridyl))carbonylamino]-tert-
0.32 M+H 489.3 butyljacetamide
4-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-tert-
0.277 M+H 517.3 butyl}butanamide
N-{[(2R)-1-(N-methylcarbamoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-flυoropheπyl)ethyI]amino}(3-
0.307 M+H 501.1 pyridyl))carboxamide methyl (2R)-2-{[(6-(2-cyaπophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
0.101 M+H 502.2 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate N-{(1S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-pyridyl))carbonylamino]-isopropyl}-
1.459 M+H 475.2 2-aminoacetamide
N-{(1S)-2-[(6-(2-cyanophenyl)-2-{t2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
3.56 M+H 503.2 3-aminopropanamide
N-{[(2R)-1-(5-aminopeπtanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.099 M+H 543.3 pyridyl))carboxamide N-{(2S)-2-[(methylsulfonyl)amino]propyl}<6-(2-cyanophenyl)-2-
1.215 M+H 496.1 {[2-(3-fluorophenyl)ethyl]amino}(3-py ridyl))carboxamide 2-aminoethyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-
0.027 M+H 531.2 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate M+H N-[{2S)-2-(methylamino)propyl](6-(2-cyanophenyl)-2-{[2-(3- 0.944 Me 432.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide N-((2R)-2-aminopropyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.399 M+H 418.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(2R)-2-(methylamino)propyl](6-(2-cyanophenyl)-2-{[2-(3-
1.89 M+H 432.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(2S)-2-[methyl(methylsulfonyl)amiπo]propyl}(6-(2-
5.84 M+H 510.2 cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]annino}(3-
pyridyl))carboxamide
N-{(2R)-2-[(methylsulfonyl)amino]propyl}(6-(2-cyanophenyl)-2-
1.333 M+H 496.2 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(2R)-2-[methyl(methylsulfonyl)amino]propyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amiπo}(3-
7.071 M+H 510.2 pyridyi))carboxamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
2.312 M+H 575.3 2-[(tert-butoxy)carbonylamino]acetamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
5.916 M+H 589.3 2-[(tert-butoxy)carbonylamino]-N-methylacetamide
N-{( 1 R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
0.049 M+H 603.3 4-[(tert-butoxy)carbonylamino]butanamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-<3- fluorophenyl)ethyl]amiπo}(3-pyridyl))carboπylamiπo]-isopropyl}-
3.677 M+H 617.4 4-[(tert-butoxy)carbonylamino]-N-methylbutanamide
N-{( 1 S)-2-[(6-(2-cyanopheπyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
3.246 M+H 489.2 2-amino-N-methylacetamide
N-{( 1 S)-2-[(6-(2-cyanophenyl)-2-fl2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
6.826 M+H 517.3 4-amino-N-methylbutanamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
0.041 M+H 475.2 2-aminoacetamide
N-{(1R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino)-isopropyl}-
0.204 M+H 503.2 4-aminobutanamide
N-{( 1 R)-2-[(6-(2-cyanophenyl)-2~[[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-isopropyl}-
0.71 M+H 517.2 4-amino-N-methylbutanamide
N-{( 1 R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyljamino}{3-pyridyl))carbonylamino]-isopropyl}-
0.152 M+H 489.2 2-amino-N-methylacetamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-
0.012 M+H 449.1 4-yl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.025 M+H 501.2 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.029 M+H 528.2 pyridyl))carboxamide
N-{[(2R)-1 -(3-aminopropyl)-5-oxopyrrolid in-2-yl]methy l}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.042 M+H 515.2 pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N-
0.044 M+H 417.1 (3-pyridylmethyl)carboxamide
{6-(3-methylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-
6.265 M+H 424.2 (3-pyridylmethyl)carboxamide
{6-(3-fluorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-
0.915 M+H 428.2 (3-pyridylmethyl)carboxamide 2.992 M+H 435.2 {6-(3-cyanophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-
(3-pyridylmethyl)carboxamide
{6-(3-methoxyphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 14.933 M+H 440.2 N-(3-pyridylmethyl)carboxamide
{6-(3-hydroxyphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 0.79 M+H 426.1 (3-pyridylmethyl)carboxamide
{6-(3-chlorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 1.967 M+H 444.1 (3-pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-[3-(trifluoromethyl)phenyl](3- 5.927 M+H 478.1 pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-[3-(dimethylamino)phenyl]-2-[(2-(2-pyridyl)ethyl)amino](3- 12.404 M+H 453.2 pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-(2-fluorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 1.321 M+H 428.1 (3-pyridylmethyl)carboxamide
{6-(2-chlorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 4.438 M+H 444.1 (3-pyridylmethyl)carboxamide
{6-(2-methylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 11.031 M+H 424.2 (3-pyridylmethyl)carboxamide
{6-(2-methoxyphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 17.936 M+H 440.2 N-(3-pyridylmethyl)carboxamide
{6-(2-cyanophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyi)}-N- 0.46 M+H 435.2 (3-pyridylmethyl)carboxamide
{2-t(2-(2-pyridyl)ethyl)amino]-6-(2-thienyl)(3-pyridyl)}-N-(3- 0.797 M+H 416.1 pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-(3-thienyl)(3-pyridyl)}-N-(3- 0.414 M+H 416.1 pyridylmethyl)carboxamide
{6-(4-hydroxyphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 0.586 M+H 426.1 (3-pyridylmethyl)carboxamide
{6-(2-hydroxyphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 2.17 M+H 426.1 (3-pyridylmethyl)carboxamide
{6-(4-ethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(3- 2.792 M+H 438.2 pyridylmethyl)carboxamide
N-(4-{6-[(2-(2-pyridyl)ethyl)amino]-5-[N-(3- 1.221 M+H 467.2 pyridylmethyl)carbamoyl]-2-pyridyl}phenyl)acetamide
{6-(4-methyl(2-thienyl))-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 0.344 M+H 430.1 N-(3-pyridylmethyl)carboxamide
{6-(4-methyl(3-thienyl))-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 4.063 M+H 430.1 N-(3-pyridylmethyl)carboxamide
{6-(2,3-dimethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 6.945 M+H 438.2 N-(3-pyridylmethyl)carboxamide
{6-(2,5-dimethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 13.588 M+H 438.2 N-(3-pyridylmethyl)carboxamide
{6-(2,4-dimethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 3.964 M+H 438.2 N-(3-pyridylmethyl)carboxamide
{6-(3,5-dimethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 10.138 M+H 438.2 N-(3-pyridylmethyl)carboxamide
{6-(3,4-dimethylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 3.38 M+H 438.2 N-(3-pyridylmethyl)carboxamide
(2-{[2-(4-fIuorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3- 1.609 M+H 427.2 pyridylmethyl)carboxamide
(2-{[2-(4-chlorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3- 6.677 M+H 443.1 pyridylmethyl)carboxamide
(2-{[2-(3-chlorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3- 0.349 M+H 443.1 pyridylmethyl)carboxamide
0.269 M+H 439.1 (2-{[2-(3-methoxyphenyl)ethyl]amino}-6-pheny!(3-pyπdyl))-N-
(3-pyridylmethyl)carboxamide
<2-{[2-{3-fluorophenyl)ethylJamino}-6-phenyl(3-pyridyl))-N-(3-
0.063 M+H 427.1 pyιϊdylmethyl)carboxamide
(2-{[2-(2-chlorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
0.16 M+H 443.1 pyridylmethyl)carboxamide
(2-{[2-{2-methoxyphenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
0.98 M+H 439.1 (3-py ridylm ethy l)carboxam ide
(2-{[2-(2-methylphenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
0.326 M+H 423.1 pyridylmethyl)carboxamide
(2-{[2-(2-fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
0.107 M+H 427.1 pyridylmethyl)carboxamide
(2-{[2-(2,6-difluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
0.288 M+H 445.1 (3-pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
0.055 M+H 445.1 (3-pyridylmethyl)carboxamide
(2-{[2-(3,4-difluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
0.521 M+H 445.2 (3-pyridylmethyl)carboxamide
(2-{[2-(2,5-difluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-
0.079 M+H 445.2 (3-pyridylmethyl)carboxam ide
[6-phenyl-2-({2-[3-(trifluoromethyl)phenyl]ethyl}amiπo)(3-
8.254 M+H 477.2 pyιϊdyl)]-N-(3-pyridylmethy1)carboxamide
(2-{[2-(3-fIuorophenyl)ethyl]amino}-6-(4-methyl(2-thienyl))(3-
0.148 M+H 447.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-thienyl)(3-pyridyl))-N-
0.048 M+H 433.1 (3-pyridylmethyl)carboxamide
(2-{[2-(2-cyanophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
0.26 M+H 443.1 pyridylmethyl)carboxamide
(2-{[2-(3-cyanophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
4.649 M+H 434.1 pyridylmethyl)carboxamide
(2-{[2-(4-hydroxyphenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
36.722 M+H 425.1 pyridylmethyl)carboxamide
(2-{[2-(2-hydroxyphenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
2.431 M+H 425.1 pyridylmethyl)carboxamide
(2-{[2-(4-aminophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-N-(3-
27.106 M+H 424.1 pyridylmethyl)carboxamide
3-[2-({6-phenyl-3-[N-(3-pyridylmethyl)carbamoyl]-2-
11.492 M+H 452.1 pyridyl}amino)ethyl]benzamide
(2-{[2-(3,5-difIuorophenyl)ethyl]amino}-6-(3-thienyl)(3-pyridyl))-
0.04 M+H 451.1 N-(3-pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(4-methyl(2-
0.119 M+H 465.1 thienyI))(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(2-cyanophenyl)(3-
0.041 M+H 470.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{t2-(3-fluorophenyl)ethyl]amino}-6-(2-thienyl)(3-pyridyl))-N-
0.137 M+H 433.1 (3-pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(2-thienyl)(3-pyridyl))-
0.097 M+H 451.1 N-(3-pyridylmethyl)carboxamide methyl 3-[2-({6-phenyl-3-[N-(3-pyridylmethyl)carbamoyl]-2-
28.097 M+H 467.2 pyridyl}amino)ethyl]benzoate
2-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
16.384 M+H 470.2 pyridylmethyl)carbamoyl]-2-pyridyl)benzamide
(6-(4-fluorophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.198 M+H 445.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
21.862 M+H 453.2 3-[2-({6-phenyl-3-[N-(3-pyridylmethyl)carbamoyl]-2-
pyridyl}amino)ethyl]benzoic acid
(6-(2-chloro~4-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
1.383 M+H 479.1 pyridylmethyl)carboxamide
(2-{[2-(3,5-difluorophenyl)ethyl]amino}-6-(4-fluorophenyl)(3-
0.138 M+H 463.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(2-cyano-4-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.038 M+H 470.1 pyridylmethyl)carboxamide
(6-(2-chloro-6-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3- 0.54 M+H 479.1 pyridylmethyl)carboxamide
(6-(2-chloro-5-methoxyphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
2.005 M+H 491.1 pyrϊdylmethyl)carboxamide
(6-(2-cyano-5-methoxyphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.041 M+H 482.1 pyridylmethyl)carboxamide
(6-(2-cyano-6-fluorophenyl)-2-{[2-(3- fIuorophenyl)ethyl]arτiino}(3-pyridyl))-N-(3-
0.086 M+H 470.1 pyridylmethyl)carboxamide
(6-(2-fluorophenyl)-2-{t2-(3-fluorophenyl)ethyl]amino}(3-
0.139 M+H 445.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(2,3-difluorophenyl)ethyl]amino}-6-(2-cyanophenyl)(3-
0.022 M+H 470.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(2-chloro-5-fluorophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
1.327 M+H 479.1 pyridylmethyl)carboxamide
(6-(2-cyano-5-fluoropheπyl)-2-{[2-{3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.053 M+H 470.1 pyridylmethyl)carboxamide
(6-(3-fluorophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.157 M+H 445.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-[2-(cyanomethyl)phenyl]-2-{[2-(3- fluorophenyl)ethyl]amir»o}(3-pyridyl))-N-(3- 16.392 M+H 466.2 pyridylmethyl)carboxamide
(2-{t2-(3-fluorophenyl)ethyl]amino}-6-[3-(hydroxymethyl)(2-
2.245 M+H 463.1 thienyl)](3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(5-formyl(3-thienyl))(3-
0.098 M+H 461.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluoropheπyl)ethyl]amino}-6-[5-(hydroxymethyl)(3-
0.073 M+H 463.1 thienyl)](3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(2-chloro-4-methylphenyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))-N-(3-
2.078 M+H 475.1 pyridylmethyl)carboxamide
(6-(2-cyano-4-methylphenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.996 M+H 466.2 pyιϊdylmethyl)carboxamide
(2-{[2-(3-fluoropheπyl)ethyl]amino}-6-[3- (hydroxymethyl)phenyl](3-pyridyl))-N-(3-
0.281 M+H 457.1 pyridylmethyl)carboxamide
(2-{[2-{3-fluorophenyl)ethyl]amino}-6-[3- (methoxymethyl)phenyl](3-pyridyl))-N-(3-
5.719 M+H 471.2 pyridylmethyl)carboxamide
(6-(1 H-1 ,2,3,4-tetraazol-5-yl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
78.064 M+H 419.1 pyridylmethyl)carboxamide
(6-[2-chloro-5-(hydroxymethyl)phenyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
1.185 M+H 491.1 pyridylmethyl)carboxamide
(2R)-2-amino-4-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]~N-
12.904 M+H 475.2 methylbutanamide
(2S)-2-amino-4-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
5.022 M+H 475.2 methyibutaπamide
N-(3-amino-2-hydroxypropyl)(6-(2-cyanophenyl)-2-{[2-(3-
4.812 M+H 434.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.209 M+H 581.2 pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3-
0.008 M+H 456.1 fluorophenyl)ethy!]amino}-6-phenyl(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fIuorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
0.043 M+H 446.1 pyridyl))carboxamide
N-{[2-(amiπomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-3-yl)(3-
0.032 M+H 460.1 pyridyl))carboxamide
N-[((3R)(3-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
2.148 M+H 458.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.336 M+H 458.2 pyridyl))-N-(4-piperidylmethyl)carboxamide
N-{[1-(2-aminoacetyl)(4-piperidyl)]methyl}(6-(2-cyanophenyl)-2-
0.572 M+H 515.2 {[2-(3-fIuorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropanoyl)(4-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
3.874 M+H 529.2 pyridyl))carboxamide
N-{[(3S)-1-(2-aminoacetyl)(3-piperidyl)3methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluoropheny])ethyl]amino}(3-
0.755 M+H 515.2 pyridyi))carboxamide
N-{[(3S)-1-(3-aminopropanoyl)(3-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluoropheπyl)ethyl]amiπo}(3-
1.261 M+H 529.2 pyridyl))carboxamide
N-[(1-acetyl(4-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
1.696 M+H 500.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((3S)-1-acetyl(3-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-
10.729 M+H 500.3 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(4-aminobutanoyl)(4-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
6.009 M+H 543.3 pyridyl))carboxamide methyl 4-{[(6-(2-cyanophenyl)-2-{[2-( 3- fluorophenyl)ethyl]amino}-3-
2.13 M+H 516.3 pyridyl)carbonylamino]methyl}piperidinecarboxylate
N-[((2R)(2-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
1.159 M+H 458.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(2-aminoacetyl)(2-piperidyl)]methyl}(6-(2-
0.092 M+H 515.3 cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyt)ethyl]amino}(3-
0.155 M+H 529.2 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.083 M+H 543.2 pyridyl))carboxamide
N-[((2R)-1-acetyl(2-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-
2.676 M+H 500.2 (3-fluoropheπyl)ethyl]amino}(3-pyridyl))carboxamide methyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
4.282 M+H 516.3 pyridyl))carbonylamino]methyl}piperidinecarboxylate
N-({(2R)-1-[(3-aminopropyl)sulfonylj(2-piperidyl)}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amiπo}(3-
0.37 M+H 579.2 pyridyl))carboxamide
N-[((2R)-1-methyl(2-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-
0.488 M+H 472.2 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-((2R)-2,3-diaminopropanoyl)(2-piperidyl)]methyl}(6-
(2-cyanophenyl)-2-{[2-(3-fIuorophenyl)ethyl]amino}(3-
0.161 M+H 544.3 pyridyl))carboxamide
N-{[(2R)-1-((2R)-2,4-diaminobutanoyl)(2-piperidyl)]methyl}(6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyt]amino}(3-
0.399 M+H 558.3 pyridyl))carboxamide
N-{[(2R)-1-((2R)-2,5-diaminopentanoyl)(2-piperidyl)]methytK6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.456 M+H 572.2 pyridyl))carboxamide
N-({(2R)-1-[(2-aminoethyl)sulfonyl](2-piperidy!)}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.178 M+H 565.2 pyridyl))carboxamide
N-{[(2R)-1-((2S)-2,5-diaminopentanoyl)(2-piperidyI)]methyl}(6-
(2-cyanopheπyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.048 M+H 572.3 pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-aminopropanoyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.052 M+H 529.3 pyridyl))carboxamide
N-{[(2R)-1 -((2 R)-2-aminopropanoyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.261 M+H 529.2 pyridyl))carboxamide
2-chloroethyl (2R)-2-{t(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
2.691 M+H 564.1 pyridyl))carboπylamiπo]methyl}piperidinecarboxylate
2-aminoethyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
0.091 M+H 545.2 pyridyl))carbonylamino]methyl}piperidinecarboxylate
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(2-{[2-(3l5- difluorophenyl)ethyl]amino}-6-(2-cyanophenyl)(3-
0.023 M+H 547.2 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}{6-(2- cyanophenyl)-2-[(2-cyclohex-1-enylethyl)amino](3-
0.133 M+H 515.3 pyridyl)}carboxamide
3-{[3-(N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2- yl]methyl}carbamoyl)-6-(2-cyanophenyl)(2-pyridyl)]amino}-N,N-
3.56 M+H 506.2 dimethylpropanamide 0.062 M+H 511.2 N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methylK6-(2-
cyanophenyl)-2-[(2-phenylethyl)amino](3-pyridyl)}carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methylK6-(2- cyanophenyl)-2-[(2-(2H-3,4,5,6-tetrahydropyran-4-
8.767 M+H 519.2 yl)ethyl)amiπo](3-pyridyl)}carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}{6-(2- cyanophenyl)-2-[(2-(2H-3,4,5,6-tetrahydropyran-2-
2.038 M+H 519.2 yl)ethyl)amino](3-pyridyl)}carboxamide
N-{[(2R)-1-((2S)-2,3-diaminopropanoyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.059 M+H 530.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-amino-3-methoxypropanoyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.193 M+H 545.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-amino-3-hydroxypropanoyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.173 M+H 531.2 fluoropheny!)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
0.014 M+H 464.1 pyridyl))carboxamide
N-([(2R)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(5-fluoro-2-{t2- (3-fluorophenyl)ethyl]arnino}-6-pyrazol-3-yl(3-
0.108 M+H 484.1 pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-amino-3-hydroxypropanoyl)pyrro1idin-2- yl]methyl}(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-
0.269 M+H 514.1 3-yl(3-pyridyl))carboxamide
N%2R)-1-((2S)-2-aminopropanoyl)pyrro1idin-2-yl]methyl}(5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
0.314 M+H 498.2 pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-2- {[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-3-yl(3-
0.169 M+H 512.1 pyridyl))carboxamide
{6-(2-cyanophenyl)-2-[(2-oxolan-2-ylethyl)amino](3-pyridyl)}-N- 0.92 M+H 428.1 (3-pyridylmethyl)carboxamide
N-((2R)-2-aminopropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.398 M+H 415.1 pyridyl))carboxamide
N-{( 1 R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]annino}-6-(1 - methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
1.754 M+H 572.2 [(tert-butoxy)carbonylamino]acetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
0.076 M+H 472.3 aimirtoacetamide
N-[((2R)-5-oxopyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.027 M+H 455.1 pyridyl))carboxamide
N-^IS^-tCS-fluoro^-iP-CS-fluorophenyOethyllamino^θ^i- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
1.804 M+H 472.3 aminoacetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1 - methyl py razol-4-y I) (3-py ridyl))ca rbony lam ino]-isop ropy l}-2-
0.197 M+H 473.1 hydroxyacetamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl)amiπo}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}-4- 0.1 M+H 600.2 [(tert-butoxy)carbonylamino]butanamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1 - methylpyrazo]-4-yl)(3-pyridyl))carbonylamino]-isopropyl}-4-
0.116 M+H 500.3 aminobutanamide
N-{(2R)-2-[(2-methoxyethyl)amino]propyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
2.328 M+H 473.1 pyridyl))carboxamide
N-{(2R)-2-[(tert-butoxy)carbonylamino]propyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
8.211 M+H 501.1 pyridyl))carboxamide
N-((2R)-2-aminopropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl{3-
0.443 M+H 401.1 pyridyl))carboxamide
N-{(1 R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1 - methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-
0.251 M+H 457.1 isopropyl}acetamide
N-{(2R)-2-[(methylsulfonyl)amino]propyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
1.002 M+H 493.1 pyridyl))carboxamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))carbonylamino]-isopropyl}-2-
0.122 M+H 459.1 aminoacetamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.024 M+H 435.1 pyridyl))-N-(2-pyridylmethyl)carboxamide
N-{(2R)-2-[(2-hydroxyethyl)amino]propyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-{1-methylpyrazol-4-yl)(3-
0.648 M+H 459.2 pyridyl))carboxamide
N-{(2R)-2-[(carbamoylmethyl)amino]propyl}(5-fluoro-2-{[2-{3- fluorophenyl)ethyl]amino}-6-{1-methylpyrazol-4-yl)(3-
0.636 M+H 472.1 pyridyl))carboxamide
N-{(1R)-2-[(5-fIuoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}-2-
0.121 M+H 500.2 {dimethylamino)acetamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}(2S)-2-
2.264 M+H 602.2 [(tert-butoxy)carbonylamino]-3-hydroxypropanamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-isopropyl}(2S)-2-
0.115 M+H 502.2 amino-3-hydroxypropanamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamiπo]-isopropyl}-2-
0.285 M+H 500.2 amino-2-methylpropanamide
N-(( 1 R)-1 -{[(5-fluoro-2-{[2-(3-f luorophenyl)ethyl]amino}-6-( 1 - methylpyrazol-4-yl)(3-pyridyl))carbonylamino]methyl}propyl)-2-
3.04 M+H 586.3 [(tert-butoxy)carbonylamino]acetamide
N-((1R)-H[(5-fluoro-2-{t2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]methyl}propyl)-2-
0.131 M+H 486.2 aminoacetamide
N-((1R)-1-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))carbonylamino]methyl}propyl)-2-[(tert-
4.323 M+H 572.3 butoxy)carbonylamino]acetamide
N-((1R)-1-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))carbonylamino]methyl}propyl)-2-
0.16 M+H 472.2 aminoacetamide 0.131 M+H 486.2 N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(1-
methylpyrazol-4-yl)(3-pyridyl))carbonylarniπo]-isopropyl}(2S)-2- aminopropanamide
N-((2R)-3-hydroxy-2-methylbutyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-rnethylpyrazol-4-yl)(3- 0.283 M+H 444.2 pyrϊdyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(6-(2-cyanophenyl)-2- 0.006 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl»carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}<2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3- 0.024 M+H 446.3 pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.024 M+H 452.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(2-pyridyl)ethy1)amino](3-pyridyl)}-N-(3- 0.512 M+H 410.1 pyridylmethyl)carboxamide
{6-(3,4-difluorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyrϊdyl)}- 1.206 M+H 446.1 N-(3-pyridylmethyl)carboxamide
{6-(4-fluorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 1.045 M+H 428.2 (3-pyridylmethyl)carboxamide
{6-(4-chlorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 0.629 M+H 444.1 (3-pyridylmethyl)carboxamide
{6-(4-methylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 0.677 M+H 424.2 (3-pyιϊdylmethy])carboxamide
{6-(4-cyanophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 1.935 M+H 435.1 <3-pyridylmethyl)carboxamide
{6-(4-methoxyphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 1.063 M+H 440.1 N-(3-pyridylmethyl)carboxamide
{6-[4-(hydroxymethyl)phenyl]-2-[(2-(2-pyridyl)ethyl)amino](3- 2.43 M+H 440.2 pyridyl)>-N-(3-pyridylmethyl)carboxamide
{6-(3,4-dichlorophenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}- 3.375 M+H 478.1 N-(3-pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-[4-(trifluoromethyl)pheπyl](3- 2.285 M+H 478.1 pyridyl)}-N-(3-pyridylmethyl)carboxamide methyl 4-{6-[(2-{2-pyridyl)ethyl)amino]-5-[N-(3- 0.989 M+H 468.1 pyridylmethyl)carbamoyl]-2-pyridyl}benzoate
{6-[4-(dimethylamino)phenyl]-2-[(2-<2-pyridyl)ethyl)amino](3- 0.809 M+H 453.2 pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-(4-acetylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N- 2.133 M+H 452.1 (3-pyridylmethyl)carboxamide
{6-phenyl-2-[(2-(3-pyridyl)ethyl)amino](3-pyridyl)}-N-(3- 2.187 M+H 410.1 pyridylmethyl)carboxamide
4-{6-[(2-(2-pyridyl)ethyl)amino]-5-[N-(3- 18.274 M+H 454.1 pyridylmethyl)carbamoyl]-2-pyridyl}ben∑oic acid
N-(3-pyridylmethyl){6-(3-pyridyl)-2-[(2-(2-pyridyl)ethyl)amino](3- 19.129 M+H 411.1 pyridyl)}carboxamide
N-(3-pyridylmethyl){6-(4-pyridyl)-2-[(2-(2-pyridyl)ethyl)amino](3- 6.557 M+H 411.1 pyridyl)}carboxamide
N-(3-pyridylmethyl){6-(2-pyridyl)-2-[(2-{2-pyridyl)ethyl)amino](3- 5.379 M+H 411.2 pyridyl)}carboxamide
4-{6-[(2-(2-pyridyl)ethyl)amino]-5-[N-(3- 2.538 M+H 453.1 pyridylmethyl)carbamoyl]-2-pyridyl}benzannide
^-^-hydroxy^-phenylethylJaminol-β-phenyKS-pyridyl^-N-CS- 1.942 M+H 425.1 pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-(2-vinylphenyl)(3-pyridyl)}-N-(3- 5.342 M+H 436.1 pyridylmethyl)carboxamide
{6-[2-(methoxymethyl)phenyl]-2-[(2-(2-pyridyl)ethyl)amino](3-
84.495 M+H 454.2 pyridyl)}-N-(3-pyridylmethyl)carboxamide
{6-(2-formylphenyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-
8.751 M+H 438.2 (3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)-2-hydroxyethyl]amino}-6-phenyl(3-
0.559 M+H 443.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)-2- nydroxyethyl)amino}(3-pyridyl))-N-(3-
0.339 M+H 468.2 pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-(1,3-thiazol-2-yl)(3-pyridyl)}-N-
8.662 M+H 417.1 (3-pyridylmethyl)carboxamide
{6-(1-methylpyrazol-4-yl)-2-[(2-(2-pyridyl)ethyl)amino](3-
2.53 M+H 414.2 pyridyl)}-N-(3-pyridylmethyl)carboxamide
[3-(aminomethyl)-6-phenyl(2-pyridyl)][2-(3-
39.528 M+H 322.1 fluorophenyl)ethyl]amine
(6-(3,4-difluorophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.534 M+H 463.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(5-cyano(2-thienyl))-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.28 M+H 458.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(5-formyl(2-thieπyl))(3-
0.052 M+H 461.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fIuorophenyl)ethyl]amino}-6-(3-formyl(2-thienyl))(3-
0.783 M+H 461.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]annino}-6-(2-formyl(3-thienyl))(3-
1.482 M+H 461.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
[2-{[2-(3-fluorophenyl)ethyl]amino}-6-(6-{[2-(3- fluorophenyl)ethyl]amino}-5-[N-(3-pyridylmethyl)carbamoylI(2-
30.616 M+H 699.2 pyridyl))(3-pyridyl)]-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[2-(hydroxymethyl)(3-
1.567 M+H 463.1 thienyl)](3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-isoxazol-4-yl(3-pyridyl))-
49.792 M+H 418.1 N-(3-pyridyl methyl)carboxamide
(6-(5-acetyl(2-thienyl))-2-{[2-(3-fluorophenyl)ethyl]atτiino}(3-
0.178 M+H 475.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-[3-(aminomethyl)(2-thienyl)]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
55.694 M+H 462.1 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5-(hydroxyethyl)(2-
0.415 M+H 477.1 thienyl)](3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-(4-cyano(3-thienyl))-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.014 M+H 458.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-[5-(aminomethyl)(2-thienyl)]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
2.698 M+H 462.1 pyridylmethyl)carboxamide
N-{[5-(aminomethyl)(3-pyridyl)]methyl}(6-(2-cyanophenyl)-2-
0.323 M+H 481.2 {[2-(3-fIuorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.081 M+H 482.1 pyridyOJ-N^IS-ChydroxymethylJCS-pyridyOlmethylJcarboxamide
N-({5-[3,3-bis(dimethylannino)-2-azaprop-2-enyl](3- pyridyl)}methyl)(6-(2-cyanophenyl)-2-{[2-(3-
2.715 M+H 579.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[3-(amiπomethyl)(2-pyridyl)]methyl}(6-(2-cyanophenyl)-2-
0.624 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2.437 M+H 824.3 (6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))-N-[(3-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]methyl}(2- pyridyl))methyl]carboxamide N-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-
70.994 M+H 442.1 pyridyl))methyl](3-pyridylamino)carboxamide
(5-chloro-δ-(2-cyanophenyl)-2-{[2-(3- fluoraphenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.512 M+H 486.1 pyιϊdylmethyl)carboxamide
(5-bromo-6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
2.653 M+H 531 pyridylmethyl)carboxamide
(5-cyano-6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.318 M+H 477.1 pyridylmethyl)carboxamide
N-[(2-chloro(3-pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.072 M+H 486.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({2-[(2-aminoethyl)amino](3-pyridyl)}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.013 M+H 510.1 pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-5-oxoindeno[3,2-b]pyridin-
0.088 M+H 453.1 3-yl)-N-(3-pyridylmethyl)carboxamide
{6-(2-cyaπophenyl)-2-([2-(3-fluorophenyl)ethyl]amino}(3-
0.029 M+H 472 pyridyl))-N-[(6-oxo(2-piperidyl))methyl]carboxamide
N-[{1-{3-[(tert-butoxy)carboπylamino]propyl}-6-oxo(2- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.877 M+H 629.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)-6-oxo(2-piperidyl)]methyl}(6-(2- cyanophenyI)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.025 M+H 529.2 pyridyl)}carboxamide
N-[((2S)(2-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
1.024 M+H 458.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)-1-{2-[(tert-butoxy)carbonylamino]acetyl}(2- piperidyl))methyl](6-(2-cyanophenyl)-2-{I2-(3- 16.979 M+H 615.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-1-(2-aminoacetyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.074 M+H 515.2 pyridyl))carboxamide
N-[((2S)-1-{3-[(tert-butoxy)carbonylamino]propanoyl}(2- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
8.636 M+H 629.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropanoyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.109 M+H 529.2 pyridyl))carboxamide
N-{[(2S)-1-(methylsulfonyl)(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.632 M+H 536.3 pytϊdyl))carboxamide
N-[((2R)-6-oxo(2-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.015 M+H 472.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-H[3-[(tert-butoxy)carboπylamino]propyl}-6-oxo(2- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-{3-
0.392 M+H 629.4 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-6-oxo(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.026 M+H 529.2 pyridyl))carboxamide
N-[((3S)(3-piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
1.974 M+H 458.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((3R)-1-acetyl(3-piperidyl))methyl]{6-(2-cyanophenyl)-2-{[2-
0.696 M+H 500.3 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide methyl (3R)-3-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
0.718' M+H 516.3 pyridyl))carbonylamino]methyl}piperidinecarboxylate
N-KPRJ-i-^-tCtert-butoxyJcarbonylaminoJacetylKS- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.199 M+H 615.4 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((3R)-H3-[(tert-butoxy)carbonylamino]propanoyl}(3- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
3.79 M+H 629.4 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((3R)-1-{4-Ktert-butoxy)carbonylamino]butanoyl}(3- piperidyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
6.161 M+H 643.4 fluorophenyl)ethyl]amino}(3-pyridyl))carboxarnide
N-{[(3R)-1-(2-aminoacetyl)(3-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.246 M+H 615.4 pyridyi))carboxamide
N-{[(3R)-1-(3-aminopropanoyl)(3-piperidyl)]methyl}(6-(2- cyaπophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.173 M+H 629.5 py ridyl))carboxam ide
N-{[(3R)-1-(4-amiπobutanoyl)(3-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethy]]amino}(3-
0.512 M+H 543.3 pyridyl))carboxamide
N-({(2R)-1 -[4-(1 ,3-dioxobenzo[c]azolin-2-yl)butyl]-6-oxo(2- piperidyl)}methyl)(6-(2-cyaπophenyl)-2-{[2-(3-
0.791 M+H 673.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutyl)-6-oxo(2-piperidyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.032 M+H 543.2 pyridyl) )carboxam ide
N-[((3S,4S,2R,5R)-3,4,5,6-tetrahydroxy(2H-3,4,5,6- tetrahydropyraπ-2-yl))methyl](6-(2-cyaπophenyl)-2-{[2-(3-
5.537 M+H 523.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-[5-(N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2- yl]methyl}carbamoyl)-6-{[2-(3-fluorophenyl)ethyl]amino}-2-
13.202 M+H 533.2 pyridyljbenzamide
N-[2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}pyrrolidinyl)(1 S)-1 -methyl-2-
3.262 M+H 615.2 oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2S)-2-aminopropanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.053 M+H 515.2 pyridyl))carboxamide
N-[( 1 R)-2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamiπo]methyl}pyrro!idinyl)-1-methyl-2-
3.94 M+H 615.2 oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2R)-2-amiπopropaπoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fIuorophenyl)ethyl]amino}(3-
0.166 M+H 515.2 pyridyl))carboxamide
N-{[(2R)-1-((2S)-2,5-diaminopentanoyl)pyrrolidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.052 M+H 558.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(2-chloro(3-pyridyl))methyl](2-{[2-(3- fiuorophenyl)ethyl]arnino}-6-(1-methylpyrazol-4-yl)(3-
0.046 M+H 451.1 pyridyl))carboxamide
N-({2-[(3-aminopropyl)amino](3-pyridyl)}methyl)(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.129 M+H 489.2 pyridyl))carboxamide
N-({2-[(2-aminoethyl)amino](3-pyridyl)}methyl)(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.122 M+H 475.1 pyridyl))carboxamide tert-butyl (2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}- 6-(1 -methylpyrazol-4-yl)(3-
1.459 M+H 541.3 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-[((2R)pyrralidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.275 M+H 441.1 pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamiπo]acetyl}pyrrolidiπ-2- yl)methyl](5-fluoro-2-{[2-(3-fIuorophenyl)ethyl]amino}-6-(1-
0.625 M+H 598.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N-
0.065 M+H 476.1 ({2-[(2-hydroxyethyl)amino](3-pyridyl)}methyl)carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.488 M+H 564.2 pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.012 M+H 464.1 pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]acetyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-
1.079 M+H 584.2 4-yl(3-pyridyl))carboxamide
N^[[(2R)-1-(2-aπninoacetyl)pyrrolidin-2-yl]methyl}(5-fluoro-2-{[2- (3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.061 M+H 484.1 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propanoyl}pyrrolidin- 2-yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- 0.98 M+H 598.3 pyrazol-4-yl(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidin-2-yl]methyl}(5-fluoro- 2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3- 0.09 M+H 498.2 pyridyl))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-fiuoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol- 0.75 M+H 612.2 4-yl(3-pyridyl))carboxamide
N-[[(2R)-1-(4-amiπobutanoyl)pyrrolidin-2-yl]methyl}(5-fIuoro-2- {[2-(3-fluoropheπyl)ethyl]amiπo}-6-pyrazol-4-yl(3-
0.047 M+H 512.2 pyridyl))carboxamide
N-[((2R)pyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluoropheπyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.394 M+H 427.1 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amiπo}-6-pyrazol-4-yl(3-
0.354 M+H 598.2 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.066 M+H 498.1 pyridyl))carboxamide
N-(carbamoylmethyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.14 M+H 401.1 pyridy l))carboxam ide
2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.065 M+H 415 pyridyl))carbonylamino]-N-methylacetamide
N-[((2R)-H<2S)-2-[(tert-butoxy)carbonylamino]-3- hydroxypropanoyl}pyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
1.001 M+H 614.3 pyridyl))carboxamide
N-{[(2R)-1-((2S)-2-amtno-3-hydroxypropaπoyl)pyrrolidin-2- yl]methyl}(5-fIuoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-
0.059 M+H 514.1 4-y!(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-pyridyl)(3-
3.665 M+H 575.2 pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-{[2-(3-
0.066 M+H 475.1 fluorophenyl)ethyl]amino}-6-(2-pyridyl)(3-pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluoropnenyl)ethyl]amino}-6-(1-methylpyrazol-
0.381 M+H 430.1 4-yl)(3-pyridyl))-N-(2-hydroxy-2-methylpropyl)carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-( 1 -methylpyrazol-
0.224 M+H 444.1 4-yl)(3-pyridyl))-N-(3-hydroxy-2,2-dimethylpropyl)carboxamide
N-((2R)-2-methyl-3-oxobutyl)(5-fluoro-2-{[2-(3- fluoropheπyl)ethyl]amino}-6-(1-methylpyra2θl-4-yl)(3-
0.109 M+H 442.2 pyridyl))carboxamide
N-((2R)-3-hydroxy-2,3-dimethylbutyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
1.225 M+H 458.1 pyridyl))carboxamide
2-((2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3- pyridyl))carbonylamino]methyl}azetidinyl)-2-oxoethyl 2-[(tert-
0.191 M+H 642.2 butoxy)carbonylamino]acetate
2-((2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3- pyridyl))carbonylamino]methyl}azetidinyl)-2-oxoethyl 2-
0.045 M+H 542.2 aminoacetate
2-((2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3- pyridyl))carbonylamino]methyl}azetidinyl)-2-oxoethyl dimethyl
0.4 M+H 593.2 phosphate
(2-{[2-(3-fluoropheπyl)ethylJamino}-6-[5-(hydroxymethyl)(2-
0.041 M+H 463.1 thienyl)](3-pyridyl))-N-(3-pyridylmethyl)carboxamide
N-({2-[(3-amiπopropyl)amino](3-pyridyl)}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.017 M+H pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.018 M+H pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(5-fluoro-2-{[2-
(3-fiuorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.066 M+H pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-4-
1.076 M+H 466.2 methyl(3-pyridyl))-N-(3-pyridy]methyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
16.894 M+H 537.2 pyridyl))-N-[(6-morpholin-4-yl(3-pyridyl))methyl]carboxamide
66.631 M+H 521.2 (6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))-N-[(6-pyrrolidinyl(3-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 1.752 M+H 536.2 pyridyl))-N-[(6-piperazinyl(3-pyridyl))methyl]carboxamide
N-{[6-(4-acetylpiperazinyl)(3-pyridyl)]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 7.747 M+H 578.2 pyridyl))carboxamide
N-[(6-bromo(2-pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3- 0.233 M+H 530.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-[(2-{[2-{3-fluorophenyl)ethyl]amino}-6-[5-(hydroxymethyl)(2- 0.254 M+H 443.1 thienyl)](3-pyridyl))carbonylamiπo]-N-methylacetamide
N-(2-carbamoylethyl)(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5- 0.549 M+H 443.1 {hydroxymethyl)(2-thienyl)](3-pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[5-(hydroxymethyl)(2- 0.045 M+H 463.1 thienyl)](3-pyridyl))-N-(2-pyridylmethyl)carboxamide
N-(carbamoylmethyl)(2-{[2-(3-fluorophenyl)ethyi]amiπo}-6-[5- 0.496 M+H 429.1 (hydroxymethyl)(2-thienyl)](3-pyridyl))carboxamide
2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 24.88 M+H 496.1 pyridyOJcarbonylaminoJ-N-CnnethylsulfonylJacetamide
N-{[3-(aminomethyl)cyclohexyl]methyl}(6-(2-cyanophenyl)-2- 0.352 M+H 486.2 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
3-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-3- 11.631 M+H 472.1 pyridyl)carbonylamino]cyclopentanecarboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 16.178 M+H 473.2 pyridyl))-N-(2-piperaziπylethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.266 M+H 467.1 pyridyl))-N-(2-pyrazin-2-ylethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.026 M+H 453.1 pyridyl))-N-(pyrazin-2-ylmethyl)carboxamide
N-((1S)-5-amino-1-carbamoylpentyl)(6-(2-cyanophenyl)-2-{[2- 4.421 M+H 489.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(3-amino-1-carbamoylpropyl)(6-(2-cyanophenyl)-2-{[2-(3- 1.906 M+H 461.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(1R)-1-carbamoyl-5-
[(phenylmethoxy)carbonylamino]pentyl}(6-(2-cyanophenyl)-2- 58.8 M+H 623.2 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(4-amino-1-carbamoylbutyl)(6-(2-cyanophenyl)-2-{[2-(3- 0.59 M+H 475.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1 R)-5-amiπo-1 -carbamoylpeπtyl)(6-(2-cyanophenyl)-2-([2- 1.28 M+H 489.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-({6-[(2-aminoethyl)amino](2-pyridyl)}methyl)(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.007 M+H 510.1 pyridyl))carboxamide
N-({6-[(3-amiπopropyl)amiπo](2-pyridyl)}methyl){6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.005 M+H 524.2 pyridyl))carboxamide
N-{(4R)-4-carbamoyl-4-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 19.6 M+H 609.1 pyridyl))carbonylamino]butyl}(phenylmethoxy)carboxamide
N-((1S)-4-amino-1-carbamoylbutyl)(6-(2-cyanophenyl)-2-{[2-(3- 5.404 M+H 475.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1 R)-4-amino-1-carbamoylbutyl)(6-(2-cyanophenyl)-2-{[2- 7.413 M+H 475.1 (3-fluoropheπyl)ethyl]amino}(3-pyridyl))carboxamide
(θ^-cyanophenyO^-fP-CS-fluoropheπyOethyllaminoXS- 0.963 M+H 444.1 pyridyl))-N-(pyrrolidin-3-ylmethyl)carboxamide
N-{[1-(2-aminoacetyl)pyrrolidin-3-yl]methyl}(6-(2-cyaπopheπyl)-
0.203 M+H 501.1 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropanoyl)pyrrolidin-3-yl]methyl}(6-{2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.27 M+H 515.1 pyridyl))carboxamide
N-{[1-(4-aminobutanoyl)pyrrolidin-3-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fIuorophenyl)ethyl]amino}(3-
0.433 M+H 529.1 pyridyl))carboxamide
N-[(1-acetylpyrrolidin-3-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.334 M+H 486.1 fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)pyrrolidin-3-yl]methyl}(6-(2-cyanophenyl)-
4.787 M+H 501.2 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(3-amino-2-carbamoylpropyl)(6-(2-cyanophenyl)-2-{[2-(3-
6.654 M+H 460.5 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2-{[2-(3-fluoropheny!)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
1.236 M+H 452.3 pyridyl))-N-[(6-oxopiperazin-2-yl)rnethyl]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N-
0.973 M+H 438.3 [(6-oxopiperazin-2-yl)methyl]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-5-yl(3-pyridyl))-N-
6.954 M+H 438.3 [(6-oxopiperazin-2-yl)methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))-N-{[4-(methyIsulfonyl)-6-oxopiperazin-2-
2.565 M+H 551.1 yl]methyl}carboxamide
N-[(4-acetyl-6-oxopiperazin-2-yl)methyt](6-(2-cyanophenyl)-2-
1.635 M+H 515.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.037 M+H 487.1 pyridyl))-N-[(4-methyl-6-oxopiperazin-2-yl)methyI]carboxamide
{6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.866 M+H 461.2 pyridyl))-N-{2-[(methylamino)carbonylamino]ethyl}carboxamide
N-(2-{[(2-aminoethyl)sulfonyl]amino}ethyl)(6-(2-cyanophenyl)-
1.548 M+H 511.2 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[{(2S)pyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
4.569 M+H 444.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
1.23 M+H 461.2 pyridyl))carbonylamino]ethyl}acetamide
3-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
4.325 M+H 475.2 pyridyl))carbonylamino]ethyl}propanamide
4-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
1.265 M+H 489.2 pyridyl))carbonylamino]ethyl}butanamide
2-[(tert-butoxy)carbonylamino]-N-{2-[(6-(2-cyanophenyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]ethyl}-N-
15.303 M+H 575.3 methylacetamide
4-[(tert-butoxy)carbonylamino]-N-{2-[(6-(2-cyaπopheπyl)-2-{[2-
(3-fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]ethyl}-N-
3.883 M+H 603.3 methylbutanamide
N-{t(2S)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl3amino}(3-
0.995 M+H 501.2 pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluoropnenyl)ethyl]amino}(3-
1.856 M+H 515.2 pyridyl))carboxamide
N-{[(2S)-1-(4-aminobutaπoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 1.27 M+H 529.3 pyridyl))carboxamide
2-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyljamino}(3-pyridyl))carbonylamino]ethyl}-N-
1.374 M+H 475.2 methylacetamide
3~amino-N-{2-[<6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-pyridyl))carboπylamino]ethyl}-N-
1.775 M+H 489.2 methylpropanamide
4-amino-N-{2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]ethyt}-N- 1.2 M+H 503.2 methylbutanamide
(6-(2-cyanophenyl)-2-{[2-(3-fluoropheny!)ethyl]amino}(3-
1.385 M+H 496.2 pyridyl))-N-{2-[methyl(methylsulfonyl)amino]ethyl}carboxamide
3-amino~N-(2-aminoethyl)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 13.725 M+H 504.2 pyridyl))carbonylamino]methyl}propanamide
N-(2-carbonylamino-2-methylpropyl)(6-(2-cyanophenyl)-2-{[2-
0.151 M+H 460.2 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((2S)-2-amino-3-hydroxypropyl)(6-(2-cyanophenyl)-2-{[2-(3-
1.588 M+H 434.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{( 1 S)-1 -{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]methyl}-2-
0.249 M+H 491.1 hydroxyethyl)-2-aminoacetamide
(2S)-2-(2-aminoacetylamino)-3-[(6-(2-cyanophenyl)-2-{t2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propyl 2-
5.705 M+H 548.3 aminoacetate
N-((1R)-1-{t(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]methyl}-2-
0.441 M+H 504.2 methylpropyl)-2-hydroxyacetamide
N-((1R)-1-{[(6-(2-cyanophenyl)-2-{[2-(3- fIuoropheπyl)ethyl]amino}(3-pyridyl))carbonylamino]methyl}-2- 0.939 M+H 503.3 methylpropyl)-2-aminoacetamide
N-((2R)-2-amino-4-methylpentyl)(6-(2-cyanophenyl)-2-{[2-(3- 1.237 M+H 460.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1 R)-1-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]methyl}-3- 1.169 M+H 518.2 methylbutyl)-2-hydroxyacetamide
N-((1R)-1-{t(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]methyl}-3- 0.913 M+H 517.2 methylbutyl)-2-aminoacetamide
N-(( 1 R)-2-amino-1 -{[<6-(2-cyanopheny l)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 12.554 M+H 490.2 pyridyl))carbonylamino]methyl}ethyl)-2-aminoacetamide
N-(( 1 R)-2-amino-1 -{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}{3- 10.561 M+H 491.2 pyridyl))carbonylamino]methyl}ethyl)-2-hydroxyacetamide
N-((3R)-2-oxo(3-piperidyl))(6-(2-cyanophenyl)-2-{[2-(3- 0.352 M+H 458.3 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
{2-[((2R)-2-phenylpropyl)amino]-6-phenyl(3-pyridyl)}-N-(3- 17.307 M+H 423.1 pyridylmethyl)carboxamide
N-(carbamoylethyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.564 M+H 432.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamϊde
0.532 M+H 432.1 N-((1 R)-1-carbamoylethyl)(6-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide methyl (2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3-
6.98 M+H 447.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoate methyl (2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3-
0.743 M+H 447.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoate methyl 2-[(6-(2-cyanophenyl)-2-{[2-(3-
0.101 M+H 433.1 fluorophenyl)ethyl]amino}-3-pyridyl)carbonylamino]acetate
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.083 M+H 405.1 pyridyl))-N-(2-hydroxyethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.238 M+H 419.1 pyridyl))-N-(2-methoxyethyl)carboxamide
N-[2-(dimethylamino)ethyl](6-(2-cyaπophenyl)-2-{[2-(3-
4.222 M+H 432.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{2-[(tert-butoxy)-N-methylcarbonylamino]ethyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
42.246 M+H 518.1 pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
4.455 M+H 418.1 pyridyl))-N-[2-(methylamino)ethyl]carboxamide
N-(3-aminopropyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.994 M+H 418.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fiuorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
0.985 M+H 446.1 methylpropanamide
(2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-pyridyl))carboπylamiπo]-N-
1.273 M+H 446.1 methylpropanamide
N-{2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-3-
0.958 M+H 446.1 pyridyl)carbonylamino]ethyl}acetamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.118 M+H 462.1 pyridyl))-N-[2-(methoxycarbonylamino)ethyl]carboxamide
(6-(2-cyanophenyl)-2-{J2-(3-fluorophenyl)ethyl]amino}(3-
0.326 M+H 482.1 pyridyl))-N-{2-[(methylsulfonyl)amino]ethyl}carboxamide ethyl 3-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-
4.792 M+H 461.1 3-pyridyl)carbonylamino]propanoate methyl 4-[(6-(2-cyanophenyl)-2-{[2-(3-
0.713 M+H 461.1 fluorophenyl)ethyl]amiπo}-3-pyrϊdyl)carbonylamino]butanoate
3-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.34 M+H 446.1 pyridyl))carbonylamino]-N-methylpropanamide
4-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
2.956 M+H 460.1 pyridyl))carbonylamino]-N-rnethylbutanamide
N-(3-carbamoylpropyl)(6-(2-cyaπophenyl)-2-{[2-(3-
0.185 M+H 446.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
4-l(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-3-
0.627 M+H 447.1 pyridyl)carbonylamino]butanoic acid
N-(2-aminoethyl)(6-(2-cyanophenyl)-2-{[2-(3-
2.637 M+H 404.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
2.108 M+H 469.1 pyridyl))-N-[(4-fluorophenyl)methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.968 M+H 476.1 pyridyl))-N-[(4-cyanophenyl)methyl]carboxamide
N-[(3,4-difluorophenyl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
1.981 M+H 487.1 flαorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((1 S)-1 -carbamoyl-2-hydroxyethyl)(6-(2-cyanophenyl)-2-{[2-
1.02 M+H 448.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanopheπyl)-2-{[2-(3-fluoropheπyl)ethyl]amino}(3-
0.318 M+H 419.1 pyridyl))-N-(3-hydroxypropyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.396 M+H 433.1 pyridyl))-N-(3-methoxypropyl)carboxamide methyl (2S)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-3-
4.177 M+H 463.1 hydroxypropanoate methyl (2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-3-
0.513 M+H 463.1 hydroxypropanoate
(2S)-2-[<6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl)amino}(3-pyridyl))carbonylamino]-3-hydroxy-
1.121 M+H 462.1 N-methylpropanamide
(2R)-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-3-hydroxy-
0.544 M+H 462.1 N-methylpropanamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.029 M+H 452.1 pyridyl))-N-(2-pyridylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
24.295 M+H 465.1 pyridyl))-N-[(4-methylphenyl)methyl]carboxamide
(6-(2-cyanopheπyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.426 M+H 466.1 pyridyl))-N-[(6-methyl(3-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.054 M+H 466.1 pyridyl))-N-[(6-methyl(2-pyridyl))methyl]carboxamide
{6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.291 M+H 466.1 pyridyl))-N-(2-(2-pyridyl)ethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.15 M+H 466.1 pyridyl))-N-(2-(3-pyridyl)ethyl)carboxamide
N-[(6-amino(3-pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.026 M+H 467.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amiπo}(3-
8.099 M+H 482.1 pyridyl))-N-[(6-methoxy(3-pyridyl))methyl]carboxamide
(6-(2-cyaπophenyl)-2-{[2-(3-fluoropheπyl)ethyl]aιniπo}(3-
0.022 M+H 466.1 pyridyl))-N-[(2-methyl(3-pyridyl))methyl]carboxamide
N-[(2-amino(3-pyridyl))methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.022 M+H 467.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.087 M+H 482.1 pyridyl))-N-[(2-methoxy(3-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.021 M+H 470.1 pyridyl))-N-[(1-hydroxy(2-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-([2-(3-fluorophenyl)ethyl]amino}(3-
0.019 M+H 470.1 pyridyl))-N-t(1-hydroxy(3-pyridyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
3.064 M+H 501.1 pyridyl))-N-[3-(4-methylpiperazinyl)propyi]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.136 M+H 415.1 pyridyl))-N-(cyclopropylmethyl)carboxamide
N-[2-(dimethylamino)ethyl]-2-[(6-(2-cyanophenyl)-2-{[2-(3-
9.309 M+H 489.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]acetamide
2-[(6-(2-cyanopheπyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
2.07 M+H 462.1 pyridyl))carbonylamino]-N-(2-hydroxyethyl)acetamide
N-(2-carbamoylethyl)-2-[(6-(2-cyanophenyl)-2-{[2-(3-
12.17 M+H 489.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboπylamino]acetamide
3-{2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-3-
9.666 M+H 490.1 pyridyl)carbonylamino]acetylamino}propanoic acid
N-{3-[(tert-butoxy)carbonylamino]propyl}-2-[(6-(2- cyanophenyl)-2-{[2-(3-fluoropheny1)ethyl]amino}(3-
9.876 M+H 575.1 pyridyi))carbonylamino]acetamide
N-[3-(dimethylamino)propyl]-2-[(6-{2-cyanophenyl)-2-{[2-(3-
12.22 M+H 503.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]acetamide
2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
5.836 M+H 476.1 pyridyl))carbonylamino]-N-(3-hydroxypropyl)acetamide
N-{2-[(tert-butoxy)-N-methylcarbonylamino]ethyl}-2-[{6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
65.469 M+H 575.1 pyridyl))carbony!amino]acetamide
2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]annino}{3-
11.096 M+H 475.1 pyridyl))carbonylamino]-N-[2-(methylamino)ethyl]acetamide methyl (2S)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3-
2.318 M+H 462.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylaminojpropanoate
N-((1 S)-2-amino-1 -carbamoylethyl)(6-(2-cyanopheπyl)-2-{[2-(3-
0.816 M+H 447.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2S)-3-[(tert-butoxy)carbonylamino]-2-[(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
30.417 M+H 548.1 pyridyl))carbonylamiπo]propaπoic acid
(2S)-3-[(tert-butoxy)carbonylamino]-2-[(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amiπo}(3-pyridyl))carbonylamino]-N-
43.598 M+H 561.1 methylpropanamide
(2S)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamiπo]-N-
3.454 M+H 461.1 methylpropanamide
(2S)-3-amiπo-2-[(6-(2-cyaπopheπyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N,N-
23.721 M+H 475.1 dimethylpropanamide
(2S)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoic
11.072 M+H 448.1 acid methyl (2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3-
0.619 M+H 462.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoate methyl (2S)-2-(acetylamino)-3-[(6-(2-cyanophenyl)-2-{[2-(3-
1.639 M+H 504.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoate
N-((2S)-2-amino-2-carbamoylethyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.669 M+H 447.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(1S)-1-carbamoyl-2-[(6-(2-cyanophenyl)-2^[2-(3- fluorophenyl)ethyl]amino}(3-
2.629 M+H 489.1 pyridyl))carbonylamino]ethyl}acetamide
(2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
2.318 M+H 461.1 methylpropanamide
(2S)-2-amino-3-[(6-(2-cyanopheπyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N,N-
8.179 M+H 475.1 dimethylpropanamide
(2S)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoic
9.758 M+H 448.1 acid
(2S)-2-(acetylamino)-3-[(6-(2-cyanophenyl)-2-{t2-{3- fluorophenyl)ethyl]amino}(3-pyridyl))carboπylamino]-N-
5.554 M+H 503.1 methylpropanamide methyl (2R)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3-
2.384 M+H 462.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoate
N-((1 R)-2-amino-1-carbamoylethyl)(6-(2-cyanophenyl)-2-{[2-
0.538 M+H 447.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(2R)-3-amino-2-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
0.274 M+H 461.1 methylpropanarnide methyl (2R)-2-amino-3-[(6-(2-cyanophenyl)-2-{[2-(3-
2.29 M+H 462.1 fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]propanoate
N-((2R)-2-amino-2-carbamoylethyl)(6-(2-cyanophenyt)-2-{[2-
0.321 M+H 447.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amiπo}(3-
0.04 M+H 442.1 pyridyl))-N-(1,3-oxazol-2-ylmethyl)carboxamide
N-[(6-cyano(3-pyridyl))methyl](2-{[2-(3-
1.245 M+H 452.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3-
0.42 M+H 456.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
2-amiπo-1-(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-
52.451 M+H 352.1 pyridyl))ethan-1-ol
N-{[4-(aminomethyl)(2-pyridyl)]methyl}(6-(2-cyanophenyl)-2-
0.598 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(5-{[(tert-butoxy)carbonylamino]methyl}(2-pyridyl))methyl](6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
68.638 M+H 581.1 pyridyl))carboxamide
N-{[5-(aminomethyl)(2-pyridyl)]methyl}(6-<2-cyanophenyl)-2-
0.515 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[4-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
1.215 M+H 456.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
N-{[5-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
1.061 M+H 456.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
(6-(2-cyanopheπyl)-2-{[2-(3-fluoropheπy!)ethyl]amino}(3-
0.021 M+H 442.1 pyridyl))-N-(isoxazol-5-ylmethyI)carboxamide
2-{[2-(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-pyridy!))-2-
51.566 M+H 409.1 hydroxyethyl]amino}acetamide ethyl 2-{[2-(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-
15.082 M+H 438.1 pyridyl))-2-hydroxyethyl]amino}acetate
1-(2-{[2-(3-fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))-2-[(2-
19.362 M+H 443.1 pyridylmethyl)amino]ethan-1 -ol
N-(4-aminobutyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.416 M+H 432.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(5-aminopentyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.154 M+H 446.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(6-aminohexyl)(6-(2-cyanophenyl)-2-{[2-(3-
1.244 M+H 460.1 fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-{2-[6-(aminomethyl)(2-pyridyl)]ethyl}(6-(2-cyanophenyl)-2-
0.032 M+H 495.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{2-[6-(aminomethyl)(2-pyridyl)]ethyl}(2^{[2-(3-
0.252 M+H 470.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
N-{[6-(2-amiπoethyl)(2-pyridyl)]methyl}(6-(2-cyanophenyl)-2-
0.011 M+H 495.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[6-(2-aminoethyl)(2-pyridyl)]methyl}(2-{[2-(3-
0.024 M+H 470.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
N-[2-(2-aminoethoxy)ethyl](6-(2-cyaπophenyl)-2-{[2-(3-
1.62 M+H 448.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(β^-cyanophenylJ-Σ^p-CS-fluorophenylJethyllaminoXS-
0.037 M+H 482.1 pyridyl))-N-{[6-(hydroxymethyl)(2-pyridyl)]methyl}carboxamide
N-{[3-(aminomethyl)phenyl]methyl}(6-(2-cyaπopheπyl)-2-{[2-(3-
0.012 M+H 480.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.532 M+H 446.1 pyridyl))carbonylamino]-N,N-dimethylacetamide
N-{2-[(2-aminoethyl)amino]ethyl}(6-(2-cyaπophenyl)-2-{[2-(3-
0.444 M+H 447.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyaπopheπyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.014 M+H 472.1 pyridyl))-N-[(2-oxo(3-piperidyl))methyl]carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.015 M+H 441.1 pyridyl))-N-(pyrazol-3-ylmethyl)carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.032 M+H 455.1 pyridyl))-N-[( 1 -methylpyrazol-3-yl)methyl]carboxa mide
N-{[4-(aminomethyl)phenyl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.277 M+H 480.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxarnide
N-[(6-{[(2-aminoethyl)amino]methyl}(2-pyridyl))methyl](6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.019 M+H 524.1 pyridyi))carboxamide methyl 6-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-3-
0.064 M+H 510.1 pyridyl)carbonylamino]methyl}pyridine-2-carboxylate
6-{[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-3-
0.047 M+H 496.1 pyridyl)carbonylamino]methyl}pyridine-2-carboxylic acid
6-{[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyI]amino}-3-
0.06 M+H 495.1 pyridyl)carbonylamino]methyl}pyridine-2-carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.031 M+H 455.1 pyridyl))-N-[(1-methylpyrazol-5-yl)methyl]carboxamide
N-(1-π[2-[(tert-butoxy)carbonylamino]ethyl}-2-oxo(3- piperidyl))(6-(2-cyanophenyl)-2-{[2-(3-
15.254 M+H 601.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[1-(2-aminoethyl)-2-oxo(3-piperidyl)](6-(2-cyanophenyl)-2-
18.531 M+H 501.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))-N-(2-{2-oxo-3-
11.834 M+H 635.1 [(phenylmethoxy)carbonylamino]piperidyl}ethyl)carboxamide
N-[2-(3-amino-2-oxopiperidyl)ethyl](6-(2-cyanophenyl)-2-{[2-(3-
2.95 M+H 501.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[2-(aminomethyl)phenyl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.024 M+H 480.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.032 M+H 458.1 pyridyl))-N-[(2-oxopyrrolidin-3-yl)methyl]carboxamide
N-[(2-{2-[(tert-butoxy)carbonylamino]ethyl}(3-pyridyl))methyl](6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.47 M+H 595.1 pyridyl))carboxamide
N-{[2-(2-aminoethyl)(3-pyridyl)]nnethyl}(6-(2-cyanophenyl)-2-
0.006 M+H 495.1 {[2-(3-fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-{2-[3-(aminomethyI)(2-pyridyl)]ethyl}(6-(2-cyanophenyl)-2-
0.31 M+H 495.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-((3S)-2-oxopyrrolidin-3-yl)(6-(2-cyanophenyl)-2-{[2-(3-
3.246 M+H 444.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))-N-{[1-(2-hydroxyethyl)-2-oxo(3-
0.059 M+H 516.1 piperidyl)]methyl}carboxamide
N-{[1-(2-aminoethyl)-2-oxo(3-piperidyl)]methyl}(6-(2-
0.031 M+H 515.1 cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- pyridyl))-N-{[1-(3-diazo-3-azaprop-3-enyl)-2-oxo(3- 0.412 M+H 541.1 piperidyl)]methyl}carboxamide
(6-(2-cyaπopheπyl)-2-{[2-(3-fluorophenyl)ethyl]amino}{3- pyridyl))-N-{[3-(3-diazo-3-azaprop-3-enyl)-2-oxo(3- 15.844 M+H 541.1 piperidyl)]methyl}carboxamide
N-[((2R)piperazin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3- 12.204 M+H 459.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-methylpiperazin-2-yl)methyl](6-(2-cyanophenyl)-2- 15.724 M+H 473.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)-1-acetylpiperazin-2-yl)methyl](6-(2-cyanophenyl)-2- 7.437 M+H 501.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(ethylsulfonyl)piperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 16.793 M+H 551.1 pyridyl))carboxamide methyl (2 R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 11.602 M+H 517.1 pyridyl))carbonylamino]methyl}piperazinecarboxylate
N-[((2S)-1-acetyl-4-methylpiperazin-2-yl)methyl](6-(2- cyanophenyl)~2-{[2-(3-fluorophenyl)ethyl]amino}(3- 6.789 M+H 515.1 pyridyl))carboxamide methyl (2S)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]methylH- 10.062 M+H 531.1 methylpiperazinecarboxylate
N-{[(2R)-1-(2-aminoacetyl)piperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
2.775 M+H 516.1 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)piperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
4.893 M+H 530.1 pyridyl))carboxamide
N-{[(2R)-1-(4-amiπobutaπoyl)piperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
5.475 M+H 544.1 pyridyl))carboxamide
N-{[(2S)-1-(2-aminoacetyl)-4-methylpiperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
1.283 M+H 530.1 pyridyl))carboxamide
N-{[(2S)-1-(3-aminopropanoyl)-4-methylpiperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
2.393 M+H 544.1 fluoropheπyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-1-(4-aminobutaπoyl)-4-methylpiperaziπ-2-yl]methyl}(6- (2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
2.322 M+H 558.1 pyridyl))carboxamide
N-{[(2S)-4-(2-aminoacetyl)piperazin-2-yl]methyl}(6-(2- cyaπophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
8.885 M+H 516.1 pyridyl))carboxamide
N-{[(2S)-4-(3-aminopropanoyl)piperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 11.197 M+H 530.1 pyridyl))carboxamide
N-{[(2S)-4-(4-aminobutanoyl)piperazin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyI]amino}(3- 12.397 M+H 544.1 pyridyl))carboxamide
N-{[(2S)-4-(2-aminoacetyl)-1-methylpiperazin-2-yl]methyl}(6-(2- 6.97 M+H 530.1 cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carboxamide
N-{[(2S)-4-(3-aminopropanoyl)-1-methylpiperazin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3- 16.962 M+H 544.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2S)-4-(4-aminobutanoyl)-1-methylpiperazin-2-yl]methyl}(6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 6.717 M+H 558.1 pyridyl))carboxamide
2-(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 13.156 M+H 466.1 pyridyl))-N-(3-pyridylmethyl)acetamide
N-[((2S)-1,4-diacetylpiperazin-2-yl)methyl](6-(2-cyanophenyl)- 17.175 M+H 543.1 2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2S)-4-methylpiperazin-2-yl)methyl](6-(2-cyanophenyl)-2- 2.92 M+H 473.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[((2R)azetidin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3- 0.413 M+H 430.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide methyl (2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 0.073 M+H 488.1 pyridyl))carbonylamino]methyl}azetidinecarboxylate
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.248 M+H 437.1 pyridyl))-N-[(2-oxopyrrolidiπ-3-yl)methyl]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N- 0.18 M+H 423.1 [(2-oxopyrrolidin-3-yl)methyljcarboxamide
N-{[(2R)-1-(2-hydroxyacetyl)pyrrolidin-2-yl]rnethyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.116 M+H 502.1 pyridyl))carboxamide methyl 4-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- 0.657 M+H 558.1 pyridyl))carbonylamino]methyl}pyrrolidinyl)-4-oxobutanoate
N-{[(2R)-1-(4-hydroxybutanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.085 M+H 530.1 pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)pyrrolidiπ-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 1.784 M+H 481.1 pyridyl))carboxamide methyl 4-((2R)-2-{[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3- 3.935 M+H 537.1 pyridyl))carbonylamino]methyl}pyrrolidinyl)-4-oxobutanoate
N-{[(2R)-1-(4-hydroxybutanoyl)pyrrolidin-2-yl]methy!}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 1.456 M+H 509.1 pyridyl))carboxamide
N-[((2R)-1-acetylazetidin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2- 0.043 M+H 472.1 (3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(methyisulfoπyl)azetidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 1.524 M+H 508.1 pyridyl))carboxamide
N-{[(2R)-1-(2-aminoacetyl)azetidin-2-yl]methyl}(6-(2- cyaπophenyl)-2-{[2-(3-fluoropheπyl)ethyl]amiπo}(3- 0.044 M+H 487.1 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)azetidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fIuorophenyl)ethyl]amino}(3- 0.045 M+H 501.1 pyridyl))carboxamide
N-{[(2R)-1-(4-amiπobutanoyl)azetidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.025 M+H 515.1 pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.04 M+H 488.1 pyridyl))carboxamide methyl 4-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
0.519 M+H 544.1 pyridyl))carbonylamino]methyl}azetidinyl)-4-oxobutanoate
N-{[(2R)-1-(4-hydroxybutanoyl)azetidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.06 M+H 516.1 pyridyl))carboxamide
N-(azaperhydroepin-2-ylmethyl)(6-(2-cyanophenyl)-2-{[2-(3-
2.501 M+H 472.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(2-aminoacetyl)azaperhydroepin-2-yl]rnethyl}(6-(2- cyanophenyl)-2-{[2-(3-fluoropnenyl)ethyl]amino}(3-
0.248 M+H 529.1 pyridyl))carb.oxamide
N-{t1-(3-aminopropanoyl)azaperhydroepin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.256 M+H 543.1 pyridyl))carboxamide
N-{[1-(4-amiπobutanoyl)azaperhydroepin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.383 M+H 557.1 pyridyl))carboxamide
N-[(H3-[(tert-butoxy)carbonylamino]propyl}-2-oxopyrrolidin-3- yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
0.104 M+H 615.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxarriide
N-{[1-(3-aminopropyl)-2-oxopyrrolidin-3-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.382 M+H 515.1 pyridyl))carboxamide
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-2-oxopyrrolidin-3- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.589 M+H 580.1 pyridyl))carboxamide
N-{[1-(3-aminopropyl)-2-oxopyrrolidin-3-yl]methyl}(2-{[2-(3- fIuorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
3.125 M+H 480.1 pyridyl))carboxamide
N-[(1-{3-[(tert-butoxy)carbonylamino]propyl}-2-oxopyrrolidin-3- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(1-
0.699 M+H 594.1 methy!pyrazol-4-yl)(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)-2-oxopyrrolidin-3-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
5.504 M+H 494.1 pyridyl))carboxamide tert-butyl (2R)-2-{[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-
3.167 M+H 509.1 4-yl(3-pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-{[(2R)-1-(2-hydroxyacetyl)pyrrolidin-2-yl]methyl}(2-{[2-(3- fluorophenyOethyllamino^'pyrazoM-yKS-
1.185 M+H 467.1 pyridyl))carboxamide
(6-(2-cyanopheπyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.095 M+H 486.1 pyridyl))-N-[(7-oxoazaperhydroepin-2-yl)methyl]carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N-
0.687 M+H 451.1 [(7-oxoazaperhydroepiπ-2-yl)methyl]carboxamide
^-{^-(S-fluorophenyOethyljamino^-CI-methylpyrazoM-yOCS-
0.603 M+H 465.1 pyridyl))-N-[{7-oxoazaperhydroepin-2-yl)methyl]carboxannide
N-[(1-{3-[(tert-butoxy)carboπylamino]propyl}-7- oxoazaperhydroepin-2-yl)methyl](6-(2-cyanophenyl)-2-{[2-(3-
17.15 M+H 643.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[1-(3-aminopropyl)-7-oxoazaperhydroepin-2-yl]methyl}(6-(2-
0.273 M+H 543.1 cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carboxamide
N-{[(2R)-1-(4-hydroxybutanoyl)pyrrolidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.963 M+H 495.1 pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-[5-(hydroxymethyl)(2-thienyl)](3-
2.541 M+H 499.1 pyridyl))carboxamide
N-{[(2R)-1-(2,3-dihydroxypropanoyl)azetidin-2-yl]methyl}(6-(2- cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.051 M+H 518.1 pyridyl))carboxamide tert-butyl (2R)-2-{[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-
0.525 M+H 495.1 4-yl(3-pyridyl))carbonylamino]methyl}azetidiπecarboxylate
N-[((2R)azetidin-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
5.442 M+H 395.1 pyridyl))carboxamide
N-[2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}azetidinyl)(1S)-1-
2.482 M+H 617.1 (hydroxymethyl)-2-oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2S)-2-annino-3-hydroxypropanoyl)azetidin-2- yl)methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.088 M+H 517.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(1R)-2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamiπo]methyl}azetidinyl)-1-(hydroxymethyl)-2-
0.439 M+H 617.1 oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2R)-2-amino-3-hydroxypropanoyl)azetidin-2- yl]methyl}(6-(2-cyanophenyl)-2-{[2-(3-
0.083 M+H 517.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[2-((2R)-2-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]methyl}azetidinyl)(1 S)-1 -(3-
1.349 M+H 644.1 aminopropyl)-2-oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2S)-2,5-diaminopentanoyl)azetidin-2-yl]methyl}(6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.274 M+H 544.1 pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.559 M+H 453.1 pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]acetyl}azetidin-2- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]annino}-6-pyrazol-4-yl(3-
2.989 M+H 552.1 pyridyl))carboxamide
N-{[(2R)-1-(2-aminoacetyl)azetidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.562 M+H 452.1 pyridyl))carboxamide
N-[(6-bromo(2-pyridyl))methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.117 M+H 494.0 pyridyl))carboxamide
N-({6-[(3-aminopropyl)amino](2-pyridyl)}methyl)(2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.018 M+H 489.1 pyridyl))carboxamide
N-({6-[(2-aminoethyl)amino](2-pyridyl)}methyl)(2-{[2-(3- fluorophenyl)ethyl]amiπo}-6-pyrazol-4-yl(3-
0.02 M+H 475.1 pyridyl))carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-pyridyl))-N-
0.039 M+H 476.1 ({6-[(2-hydroxyethyl)amino](2-pyridyl)}methyl)carboxamide methyl (2R)-2-{[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-
0.245 M+H 453.1 yl(3-pyridyl))carbonylamino]methyl}azetidinecarboxylate
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propanoyl}pyrrolidin- 2-yt)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
0.505 M+H 612.1 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidin-2-yl]methyl}(5-fluoro- 2-{[2-(3-fluoropheπyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.07 M+H 512.1 pyridyl))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamino]butanoyl}pyrrolidJn-2- yl)methyl](5-ftuoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
0.483 M+H 626.1 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-2- {[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.087 M+H 526.1 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](5- fluoro-2-{[2-(3-fluorophenyl>ethyl]amino}-6-(1-methylpyrazol-4-
0.388 M+H 578.1 yl)(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-rnethylpyrazol-4-yl)(3-
0.012 M+H 478.1 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-( 1 -methylpy razol-4-yl)(3-
0.322 M+H 612.1 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(5- f)uoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
0.079 M+H 512.1 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(2,3-dihydroxypropanoyl)pyrrolidin-2-yl]methyl}(5- fIuoro-2-{[2-(3-fIuorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
0.143 M+H 529.1 yl)(3-pyridyl))carboxamide
(2-[[2-{3-fluorophenyl)ethyl]amino}-6-[1-(2- hydroxyethyl)pyrazol-4-yl](3-pyridyl))-N-(3-
0.972 M+H 461.1 pyridylmethyl)carboxamide tert-butyl (2R)-2-{[(6-(2-cyanophenyl)-5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}(3-
9.038 M+H 562.1 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-[((2R)pyrrolidin-2-yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-
0.103 M+H 462.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonyIamino]methyl}(3-pyridyl))methyl](2- {[2-(3-fluorophenyl)ethyl]amino}-6-[1-(2-hydroxyethyl)pyrazol-4- 14.993 M+H 590.1 yl](3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{t2-(3- fluorophenyl)ethyl]amino}-6-[1-(2-hydroxyethyl)pyrazol-4-yl](3-
0.707 M+H 490.1 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](6- (2-cyanophenyl)-5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
5.358 M+H 599.1 pyridyl))carboxamide
N^p-CaminomethyOCS-pyridylJJmethylKe^-cyanophenylJ-S- fluoro-2-{[2-(3-fluorophenyl)ethyl]aπfiino}(3-
0.006 M+H 499.1 pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]acetyl}pyrrolidin-2-
2.786 M+H 619.1 yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-5-fluoro~2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.04 M+H 519.1 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propanoyl}pyrrolidin- 2-yl)methyl](6-(2-cyanophenyl)-5-fiuoro-2-{[2-(3-
2.771 M+H 633.1 fluorophenyl)ethyl]amino}(3-pyridy!))carboxamide
N-{[(2R)-1-(3-aminopropanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.042 M+H 533.1 pyridyl))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamiπo]butaπoyl}pyrrolidin-2- yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
1.059 M+H 647.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.038 M+H 547.1 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidiπ-2-yl)methyl](6-(2-cyanophenyl)-5-fluoro-2-{[2-(3-
0.845 M+H 633.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-5-fluoro-24[2-(3-fluorophenyl)ethyl]amino}(3-
0.042 M+H 533.1 pyridyl))carboxamide
N-{[{2R)-1-(2,3-dihydroxypropanoyl)pyrrolidin-2-yl]methyl}(6-(2- cyanophenyl)-5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}(3- 0.11 M+H 550.1 pyridyl))carboxamide
N-[2-((2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3- pyridyl))carbonylamino]methyl}pyrrolidinyl)(1S)-1-methyl-2-
1.953 M+H 612.1 oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2S)-2-aminopropanoyl)pyrrolidin-2-yl]methyl}(5- fluoro-2-{[2-(3-fluorophenyl)ethyl]annino}-6-(1-methylpyrazol-4-
0.099 M+H 512.1 yl)(3-pyridyl))carboxamide
N-[((2R)-1 -{(2S)-2,5-bis[(tert- butoxy)carbonylamino]pentanoyl}pyrrolidin-2-yl)methyl](5- fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4- 19.6 M+H 755.4 yl)(3-pyridyl))carboxamide
N-{[(2R)-1-((2S)-2,5-diaminopentandyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-fIuorophenyl)ethyl]amiπo}-6-(1-
0.078 M+H 555.1 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-[2-((2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3- pyridyl))carbonylamino]methyl}pyrrolidinyl)(1S)-1-
1.195 M+H 628.1 (hydroxymethyl)-2-oxoethyl](tert-butoxy)carboxamide
N-{[(2R)-1-((2S)-2-amino-3-hydroxypropanoyl)pyrrolidin-2- yl]methyl}(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]annino}-6-(1-
0.072 M+H 528.1 methylpyrazol-4-yl)(3-pyridyl))carboxamide methyl 4-((2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}- 6-(1 -methylpyrazol-4-yl)(3-
0.534 M+H 555.1 pyridyl))carbonylamino]methyl}pyrrolidinyl)-4-oxobutanoate
N-{[(2R)-1-(4-hydroxybutanoyl)pyrrolidin-2-yl]methyl}(5-fIuoro- 2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.245 M+H 527.1 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](5- 0.75 M+H 585.1 cyaπo-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
yl)(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methy]}(5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.05 M+H 485.1 pyridyl))carboxamide
N-[3-((5R)-5-{[(5-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}-6-{1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]rnethyl}-2- 1.15 M+H 619.1 oxopyrrolidinyl)propyl](tert-butoxy)carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-y!]methyl}(5- cyano-2-{[2-(3-fluorophenyl)ethyl]amino}-6-( 1 -methylpyrazol-4- 0.31 M+H 519.1 yl)(3-pyridyl))carboxamide tert-butyl (2R)-2-{[(5-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}- 6-(1-methylpyrazol-4-yl)(3-
2.518 M+H 548.1 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-[((2R)pyrrolidin-2-yl)methyl](5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-rnethylpyrazol-4-yl)(3-
0.281 M+H 448.1 pyridyl))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
1.613 M+H 633.2 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-cyano-2- {[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.211 M+H 533.2 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}{3-pyridyl))methyl](2- {[2-(3-fluoropheπyl)ethyl]amino}-5-methyl-6-(1-methylpyrazol-
1.122 M+H 574.1 4-yl)(3-pyridyl))carboxamide
N-{t2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-5-methyl-6-(1-methy]pyrazol-4-yl){3-
0.129 M+H 474.1 pyridyl))carboxamide
N-[((2R)-1-{3-((tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyI](2-{[2-(3-fluorophenyl)ethyl]amino}-5-
1.461 M+H 608.2 methyl-6-(1-methylpyrazol~4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(2-{[2- (3-fiuorophenyl)ethyl]amino}-5-methyl-6-(1-methylpyrazol-4-
0.577 M+H 508.2 yl)(3-pyridyl))carboxamide tert-butyl (2R)-2-{[(2-{[2-(3-fluorophenyl)ethyl]amino}-5-methyl- 6-(1 -methylpy razol-4-yl)(3-
3.702 M+H 537.1 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-[((2R)pyrrolidiπ-2-yl)methyl](2-{[2-(3- fluorophenyl)ethyl]amino}-5-methyl-6-{1-methylpyrazol-4-yl)(3-
0.901 M+H 437.1 pyridyt))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamino]butaπoyl}pyrrolidin-2- yl)methyl](2-{[2-(3-fluorophenyl)ethyl]amino}-5-methyl-6-(1-
2.265 M+H 622.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrotidin-2-yl]methyl}(2-{[2-(3- fluorophenyl)ethyl]amino}-5-methyl-6-(1-methylpyrazo!-4-yl)(3-
0.381 M+H 522.2 pyridyl))carboxamide tert-butyl (2R)-2-{[(5-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}- 6-pyrazol-4-yl(3-
6.407 M+H 534.1 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-[((2R)pyrrolidin-2-yl)methyl](5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.624 M+H 434.1 pyridyl))carboxamide
N-[{(2R)-1-{4-[(tert-butoxy)carbonylamino]butanoyl}pyrro!idiπ-2-
3.421 M+H 619.2 yl)methyl](5-cyano-2-{[2-(3-fluoropheπyl)ethyl]amino}-6-
pyrazol-4-yl(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-cyano-2-
{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.359 M+H 519.2 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
3.506 M+H 605.1 pyridyl ))carboxa mide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(5- cyano-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.735 M+H 505.1 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](5- cyano-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
1.003 M+H 571.1 pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-cyano-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.058 M+H 471.1 pyridyl))carboxamide
N-[(2-{[(tert-butoxy)carbonylamino]methyl}(3-pyridyl))methyl](6-
(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}-5-methyl(3-
4.252 M+H 595.2 pyridyl))carboxamide
N-{[2-(amiπomethyl)(3-pyridyl)]methyl}(6-(2-cyanophenyl)-2-
{[2-(3-fluorophenyl)ethyl]amino}-5-methyl(3-
0.158 M+H 495.2 pyridyl))carboxamide
N-[3-((5R)-5-{[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}-5-methyl(3- pyridyl))carbonylamino]methyl}-2-oxopyrrolidinyl)propyl](tert-
5.372 M+H 629.2 butoxy)carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(6-(2- cyanophenyO^-fP-CS-fluorophenyόethyllamino^δ-methyKS-
1.226 M+H 529.1 pyridyl))carboxamide methyl 5-chloro-2-{t2-(3-fluorophenyl)ethyl]amino}-6-(1-
15.521 M+H 389.0 methylpyrazol-4-yl)pyridine-3-carboxylate
N-[(2-{[(tert-butoxy)carbonylamiπo]methyl}(3-pyridyl))methyl](5- chloro-2-{[2-(3-fIuorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
0.414 M+H 594.1 yl)(3-pyridyl))carboxamide
N-IP-CaminomethylXS-pyridyOlmethylKS-chloro^-IP-CS- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.03 M+H 494.0 pyridyl))carboxamide
N-[((2R)-1-{3-[(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](5-ch!oro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.443 M+H 628.2 pyridyl))carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidiπ-2-yl]methyl}(5- chloro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-
0.16 M+H 528.1 yl)(3-pyridyl))carboxamide tert-butyl (2R)-2-{[(5-chloro-2-{[2-(3-fluorophenyl)ethyl]amino}-
6-( 1 -methylpy razol-4-yl)(3-
1.601 M+H 557.1 pyridyl))carbonylamiπo]methyl}pyrrolidinecarboxylate
N-[((2R)pyrrolidin-2-yl)methyl](5-chloro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.167 M+H 457.1 pyridyl))carboxamide
N-[((2R)-i-{4-[(tert-butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-chloro-2-{[2-(3-fluoropheπyl)ethyl]amino}-6-(1-
0.683 M+H 642.2 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-chloro-2- {[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.047 M+H 542.1 pyridyl))carboxamide
N-{3-[(tert-butoxy)carbonylamino]propyl}(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
4.501 M+H 515.2 pyridyl))carboxamide
N-(3-aminopropyl)(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-
3.957 M+H 415.1 6-(1-methylpyrazol-4-yl)(3-pyridyl))carboxamide methyl (2R)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- (1-methylpyrazo1-4-yl)(3-pyridyl))carbonylamino]-2- 0.1 M+H 458.1 methylpropanoate
(2R)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-2-
3.841 M+H 444.1 methylpropanoic acid
2-[(tert-butoxy)carbonylamino]-N-{3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-rnethylpyrazol-4-yl)(3-
0.768 M+H 572.2 pyridyl))carbonylamino]propyl}acetamide
2-amino-N-{3-[(5-fluoro-2-{[2-(3-fluoropheny])ethyl]amino}-6- (1-methylpyrazol-4-yl)(3-
2.494 M+H 472.1 pyridyl))carbonylamino]propyl}acetamide
N-{3-[(5-fluoro-2-{[2-{3-fluorophenyl)ethyl]amino}-6-(1-
2.617 M+H 457.1 methylpyrazol-4-yl)-3-pyridyl)carbonylamino]propyl}acetamide
N-((2R)-3-hydroxy-2-methylpropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.05 M+H 430.1 pyridyl))carboxamide
N-((2R)-2-carbamoylpropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.089 M+H 443.1 pyridyl))carboxamide
(2R)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-2-methyl-N- 0.13 M+H 457.1 methylpropanamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazo!-4-yl(3-
0.024 M+H 424.1 pyridyl))-N-(pyrazol-3-ylmethyl)carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol- 0.02 M+H 438.1 4-yl)(3-pyridyl))-N-(pyrazol-3-ylmethyl)carboxamide
N-((2R)-4-diazo-2-methyl-4-azabut-4-enyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amiπo}-6-(1-methylpyrazol-4-yl)(3-
0.118 M+H 455.2 pyridyl))carboxamide
5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-nriethylpyrazol- 10.538 M+H 412.1 4-yl)(3-pyridyl) 3-methylazetidinyl ketone
N-((2S)-3-amino-2-methylpropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 1.05 M+H 429.2 pyridyl))carboxamide
N-{(2R)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-2-methylpropyl}-
0.373 M+H 586.5 2-[(tert-butoxy)carbonylamino]acetamide
N^RJ-S-Kδ-fluoro^-CP-CS-fluorophenyOethyllamino^-CI- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-2-methylpropyl}-
0.152 M+H 486.2 2-aminoacetamide
N-{(2R)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-2-methylpropyl}-
0.105 M+H 487.1 2-hydroxyacetamide tert-butyl (2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-
0.525 M+H 527.1 6-(1-methylpyrazol-4-yl)(3-
pyridyl))carbonylamino]methyl}azetidinecarboxylate
N-[((2R)azetidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
1.582 M+H 427.1 pyridyl))carboxamide
N-[((2R)-1 -{2-[(tert-butoxy)carbonylamino]acetyl}azetid in-2- yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
0.436 M+H 584.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(2-aminoacetyl)azetidin-2-yl]methyl}(5-fluoro-2-{[2- (3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.05 M+H 484.1 pyridyl))carboxamide
N-[((2R)-1-{4-[(tert-butoxy)carbonylamino]butaπoyl}azetidin-2- yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-
0.122 M+H 612.3 methylpyrazol-4-yl)(3-pyridyl))carboxamide
N-{[(2R)-1-(4-aminobutanoyl)azetidin-2-yl]methyl}(5-fluoro-2- {[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.04 M+H 512.1 pyridyl))carboxamide
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2-yl]methyl}(5-flυoro-2-{[2- (3-fiuorophenyl)ethyljamino}-6-(1-methylpyrazol-4-yl)(3-
0.047 M+H 485.2 pyridyl))carboxamide methyl (2S)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- (1-methylpyrazol-4-yl)(3-pyridyl))carbonylamino]-2-
0.141 M+H 458.1 methylpropanoate
N-((2S)-3-hydroxy-2-methylpropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- 0.09 M+H 430.1 pyridyl))carboxamide
N-((2R)-3-hydroxy-2-methylpropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.059 M+H 416.1 pyridyl))carboxamide methyl (2R)-3-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
0.073 M+H 444.1 pyrazol-4-yl(3-pyridyl))carbonylamino]-2-methylpropanoate methyl (2R)-3-[(6-(2-cyanophenyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-2-
0.186 M+H 461.1 methylpropanoate
N-((2R)-3-hydroxy-2-methylpropyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.061 M+H 433.2 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-[(2,2-dimethyl(1,3-dioxan-5-yl))methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
2.226 M+H 486.2 pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol- 4-yl)(3-pyridyl))-N-[3-hydroxy-2-
0.134 M+H 446.2 (hydroxymethyl)propyl]carboxamide
N-[(2,2-dimethyl(1,3-dioxan-5-yl))methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.927 M+H 472.2 pyridyl))carboxamide
(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.215 M+H ' 432.2 pyridyl))-N-[3-hydroxy-2-(hydroxymethyl)propyl]carboxamide
N-[(2,2-dimethyl(1,3-dioxaπ-5-yl))methyl)(6-(2-cyanophenyl)-2-
1.023 M+H 489.2 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
(6-(2-cyanophenyl)-2-{[2-(3-fIuorophenyl)ethyl]amino}(3-
0.209 M+H 449.2 pyridyl))-N-[3-hydroxy-2-(hydroxymethyl)propyl]carboxamide methyl (2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- (1 -methylpyrazol-4-yl){3-
0.117 M+H 472.1 pyridyl))carbonylamino]methyl}butanoate
0.039 M+H 444.1 N-((2R)-2-ethyl-3-hydroxypropyl)(5-fluoro-2-{[2-(3-
fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3- pyridy I) )carboxam ide methyl (2R)-2-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
0.117 M+H 458.1 pyrazol-4-yl(3-pyridyl))carboπylamino]methyl}butaπoate
N-((2R)-2-ethy]-3-hydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.052 M+H 430.1 pyridyl))carboxamide methyl (2R)-2-[(tert-butoxy)carbonylamiπo]-3-[(5-fluoro-2-{[2-
(3-fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
90.761 M+H 559.2 pyridyl))carbonylamino]propanoate methyl (2R)-2-amino-3-[(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.568 M+H 459.2 pyridyl))carbonylamino]propanoate
N-tCIR^I-IKδ-fluoro^p-CS-fluorophenylJethylJaminoJ-e-CI- methylpyrazol-4-yl)(3-pyridyl))carbonylamino]rnethyl}-2-
25.925 M+H 531.2 hydroxyethyl)(tert-butoxy)carboxamide
N-((2R)-2-amino-3-hydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
1.258 M+H 431.2 pyridyl))carboxamide
N-((1R)-1-{[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrazol-4-yl(3-pyridyl))carbonylamino]methyl}~2-
57.166 M+H 517.2 hydroxyethyl)(tert-butoxy)carboxamide
N-{(2R)-2-amino-3-hydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
1.825 M+H 417.2 pyridy!))carboxamide
N-(2,3-dihydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-(1-methylpyrazol-4-yl)(3-
0.307 M+H 432.1 pyridyl))carboxamide
N-{2,3-dihydroxypropyl)(5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.448 M+H 418.1 pyridyl))carboxamide methyl (2R)-3-{[2-amino-5-fluoro-6-(1 -methylpyrazol-4-yl)(3-
86.027 M+H 336.1 pyridyl)]carbonylamino}-2-methylpropanoate
N-{[(2R)-1-(2-hydroxy-2-methylpropanoyl)azetidin-2- yl]methyl}(5-fluoro-2H[[2-(3-fluorophenyl)ethyl]amino}-6-(1-
0.141 M+H 513.1 methylpyrazol-4-yl)(3-pyridyl))carboxamide tert-butyl (2R)-2-{[(5-fluoro-2-{[2-{3-fluoropheπyl)ethyl]amiπo}-
6-pyrazol-4-yl(3-
0.289 M+H 513.1 pyridyl))carbonylamino]methyl}azetidinecarboxylate
N-{[(2R)-1-(2-hydroxyacetyl)azetidin-2-yl]methyl}(5-fluoro-2-{[2-
(3-fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.046 M+H 471.1 pyridyl))carboxamide
N-[((2R)azetidin-2-yl)methyl](5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}-6-pyrazol-4-yl(3-
0.466 M+H 413.1 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
0.019 M+H 456.1 fluorophenyl)ethyl]amino}-6-phenyl(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(6-(2-cyanophenyl)-2-
0.009 M+H 481.1 {[2-(3-fluorophenyl)ethyl]amiπo}(3-pyridyl))carboxamide
N-(carbamoylmethyl)(6-(2-cyanophenyl)-2-{E2-(3-
0.104 M+H 418.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-(2-carbamoylethyl)(6-(2-cyanophenyl)-2-{[2-(3-
0.099 M+H 432.1 fluorophenyl)ethy]]amino}(3-pyridyl))carboxamide
0.102 M+H 432.1 2-[(6-(2-cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
pyridyl))carbonylamino]-N-methylacetamide N-((3S)-2-oxo(3-piperidyl))(6-(2-cyanophenyl)-2-{[2-(3-
5.614 M+H 458.1 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
Example 14:
I
2-(3-fluorophenethylamino)-iV-(pyridin-3-ylmethyl)-6-(pyrrolidin-l-yl)nicotinamide
[0269] A mixture of 2,6-dichloronicotinic acid (19.2 g, 0.1 mol), N1N'- carbonyldiimidazole (CDI, 17.8 g, 0.11 mmol) and THF (1 L) was stirred at 6O0C for 30 min and cooled to room temperature. To the mixture was added 3-aminomethyl pyridine (10.2 ml_, 0.1 mol) and DIEA (21 ml_, ,0.12 mol). The reaction mixture was stirred at room temperature for 4 hrs, and diluted with EtOAc. The organic layer was washed with H2O, brine, dried over Na2SO4. Concentration of the organic solution gave 2,6-dichloro-N-(pyridin-3-ylmethyl)picotinamide (20.5 g, 73%). LRMS (M+H+) m/z 282.0; (M+2+H+) m/z 283.9.
[0270] A mixture of 2,6-dichloro-N-(pyridin-3-ylmethyl)nicotinamide (20.4 g, 72.3 mmol), 3-fluorophenethyl amine (11.1 g, 79.5 mmol), K2CO3 (20 g 145 mmol) and DMF (200 mL) was stirred at 1000C for 10 hours. The mixture was diluted with ethyl acetate (2 X 300 mL) and washed with saturated ammonium chloride, sodium bicarbonate and brine. The organic layers were combined, dried (MgSO4) and concentrated. The residue was purified by silica gel chromatography to 6-chloro-2-(3- fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide (12.6 g, 46%). LRMS (M+H+) m/z 385.1.
[0271] A mixture of 6-chloro-2-(3-fluorophenethylamino)-N-(pyridin-3- ylmethyl)nicotinamide (50 mg, 0.13 mmol) and pyrrolidine (94 mg, 1.3 mmol) in a sealed tube was heated to 150 0C for 10 min. LC/MS indicated the reaction was complete. The crude was diluted with MeOH1 filtered, and purified on RP-HPLC using a mixture of acetonitrile and H2O to a give 2-(3-fluorophenethylamino)-N-(pyridin-3-ylmethyl)-6-(pyrrolidiπ-1-yl)nicotinamide (20.2 mg, 37%). LRMS (M +H+) m/z 420.2.
2-(3-fluorophenethylamino)-6-(cyclopentene)-Λ/-(2-methylamino-3-ylmethyl)nicotinamide
[0272] A mixture of the acid 6-chloro-2-(3-fluorophenethylamino)nicotinic acid (1.2 g, 4.2 mmol), DIPEA (0.69 ml_, 4.2 mmol), HBTU (1.6 g, 4.2 mmol) and DMF (7 ml_) was stirred at room temperature. An amine tert-butyl (3-(aminomethyl)pyridin-2-yl)methylcarbamate (0.9 g, 3.8 mmol) dissolved in DMF (3.5 mL) was added to the reaction mixture and allowed to stir at room temperature until complete. The mixture was diluted with EtOAc and washed with H2O1 1 M NaOH, brine and dried (Na2SOi). The organics were concentrated and purified by a silica gel chromatography with a mixture of EtOAc and hexane to give tert-butyl (3-((6-chloro-2-(3- fluorophenethylamino)nicotinamido)methyl)pyridin-2-yl)methylcarbamate (0.9 g, 46%). LC/MS (M+H+) m/z 514.1.
[0273] A mixture of (3-((6-chloro-2-(3- fluorophenethylamino)nicotinarnido)methyl)pyridin-2-yl)rnethylcarbamate (0.10 g, 0.19 mnnol), tetrakis(triphenylphosphine)palladium(0) (0.045 g, 0.039 mmol), K2CO3 (0.081 g, 0.59 mmol), cyclopenten-1 -yl boronic acid (0.026 g, 0.23 mmol) and NMP (0.2 ml_) was sealed in a tube and microwaved at 1500C for 30 min. The reaction mixture was filtered through a silica plug with EtOAc1 washed with H2O, brine, dried (Na2SO4) and concentrated to give tert-butyl (3-((6- cyclopentenyl-2-(3-fluorophenethylamino)nicotinamido)methyl)pyridin-2-yl)methylcarbamate. LC/MS (M+H+) m/z 546.2.
[0274] tert-butyl (3-((6-cyclopentenyl-2-(3- fluorophenethylamino)nicotinamido)methyl)pyridin-2-yl)methylcarbamate was dissolved in MeOH (1 ml_) and allowed to stir at room temperature. HCI/dioxane (4M, 2 mL) was added dropwise and stirred until complete. The reaction mixture was concentrated, diluted with EtOAc, washed with saturated sodium bicarbonate, brine and dried (Na2SO4). The organics were concentrated and purified by RP-HPLC with a mixture of acetonitrile and H2O to give N-((2- (aminomethyl)pyridin-3-yl)methyl)-6-cyclopentenyl-2-(3-fluorophenethylamino)nicotinamide as a solid (0.026 g, 25%). LC/MS (M+H+) m/z 446.2.
Example 16:
methyl 4-(6-(3-fIuorophenethylamino)-5-(pyridin-3-ylmethylcarbamoyl)pyridin-2-yl)-5,6-dihydropyridine-1(2W)- carboxylate
[0275] A mixture of 2,6-dichloro-N-(pyridin-3-ylmethyl)nicotinamide (234 mg, 0.6 mmol), tert-butyl 4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)-5,6-dihydropyridine-1(2H)- carboxylate (234 mg, 0.75 mmol), Pd(PPh3)4 (69 mg. 0.06 mmol), K2CO3 (249 mg, 1.8 mmol) and DMF (6 mL) was stirred at 150 0C in microwave for 1 h, then concentrated to dryness. The residue was dissolved in EtOAc-MeOH (9:1 , 10 mL), filtered through a silica gel pak. Concentration of the mixture followed by the purification on RP-HPLC using a mixture of acetonitrile and H2O gave tert-butyl 4-(6-(3-fluorophenethylamino)-5-(pyridin-3- ylmethylcarbamoyl)pyridin-2-yl)-5,6-dihydropyridine-1 (2H)-carboxylate as a pale yellow solid (240 mg, 75%). LRMS (M+H+) m/z 532.2
[0276] To a solution of tert-butyl 4-(6-(3-fluorophenethylamino)-5-(pyridin-3- ylmethylcarbamoyl)pyridin-2-yl)-5,6-dihydropyridine-1(2H)-carboxylate (200 mg, 0.377 mmol) in MeOH (8 ml_) at 0 0C was added HCI (2.0 rriL, 8 mmol, 4 M in dioxane) under nitrogen. The reaction mixture was warmed to room temperatures stirred at r.t. for 4 h. LC/MS indicated the reaction was complete. The reaction mixture was concentrated under reduced pressure to give 2-(3-fluorophenethylamino)-N-(pyridin-3-ylmethyl)-6-(1 ,2,3,6-tetrahydropyridin-4- yl)nicotinamide which was used for next step without further purification. LRMS (M +H+) m/z 444.2.
[0277] To a mixture of 4 (69 mg, 0.128 mmol), DIEA (111 μL, 0.640 mmol) and DCM (2 ml_) at 0 0C was added methyl chloroformate (12 μL, 0.154 mmol) dropwise. The mixture was stirred at r.t. for 0.5h. Concentration of the mixture followed by the purification on RP-HPLC using a mixture of acetonitrile and H2O gave methyl 4-(6-(3-fluorophenethylamino)-5-(pyridin-3- ylmethylcarbamoyl)pyridin-2-yl)-5,6-dihydropyridine-1(2H)-carboxylate (33 mg, 53%). LRMS (M+H+) m/z 490.2.
Example 17
[0278] Using procedures similar to those described herein, the compounds in the following table were synthesized and tested.
IC50
Arithmetic
Mean ion m/z ChemicalName
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-piperidyl(3-pyridyl))-N-(3-
0.3713 M+H 434.2 pyridylmethyl)carboxamide
N-{[(3S)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidiπ-3-
3.7733 M+H 491.2 yl]methyl}acetamide
(6-((3R)-3-hydroxypyrrolidiny!)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-
11.4467 M+H 402.2 (carbamoylmethyl)carboxamide
2-[(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carboπylamino]-N-
3.4375 M+H 416.2 methylacetamide
(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3-
71.3315 M+H 373.2 fluorophenyl)ethyl]amiπo}(3-pyridyl))-N,N-dimethylcarboxamide
N-(carbamoylmethyl)(2-{[2-(3-fluorophenyl)ethyl]amino}-6-
1.4612 M+H 386.2 pyrrolidinyl(3-pyridyl))carboxamide
(6-[{3R)-3-(hydroxymethyl)pyrrolidinyl]-2-{[2-(3- fiuorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.3107 M+H 450.2 pyridy!methyl)carboxamide
(6-{(3S)-3-[(methylamino)methyl]pyrrolidinyl}-2-{[2-(3- fluorophenyl)ethylJamino}(3-pyridyl))-N-{3-
64.7725 M+H 463.2 pyridylmethyl)carboxamide
N-{t(3R)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-yl]methyl}-N-
47.5598 M+H 505.2 methylacetamide
(6~{(3S)-3-[(dimethylamino)methyl]pyrrolidinyl}-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
75.0803 M+H 477.3 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-oxopiperidyl)(3-pyridyl))-
5.4042 M+H 448.1 N-(3-pyridylmethyl)carboxamide
(6-[(3S)-3-(aminomethyl)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
16.8833 M+H 449.2 pyridylmethyl)carboxamide
N-[(6-amino(3-pyridyl))methyl](2-{[2-(3-
0.243 M+H 435.2 fluorophenyl)ethyl]amiπo}-6-pyrrolidinyl(3-pyridyl))carboxamide
N-(2-carbamoylethyl)(2-{[2-(3-fluorophenyl)ethyl]amino}-6-
1.5893 M+H 400.2 pyrrolidinyl(3-pyridyl))carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.5329 M+H 436.2 pyridylmethyl)carboxamide b-KP-CS-fluoropheπyOethylJamino^θ^-oxopyrrolidinyOCS-
4.5555 M+H 434.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyf)ethyl]amino}-6-pyrrolidinyl(3-pyridyl))-N-
0.1306 M+H 420.2 (3-pyridylmethyl)carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3-
15.9209 M+H 416.2 fIuorophenyl)ethyl]amino}(3-pyridyl))-N-(2-
IC50
Arithmetic
Mean Ion m/z ChemicalName carbamoylethyl)carboxamide
(6-((3S)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))-N-(3-
1.0159 M+H 436.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[4-
(hydroxymethyl)piperidyl](3-pyridyl))-N-(3-
0.3825 M+H 464.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(5-hydroxy-2-
29.6559 M+H 464.2 oxopiperidyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-oxopiperazinyl)(3-
6.9699 M+H 449.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-methylpiperazinyl)(3-
59.8594 M+H 449.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluoropheπyl)ethyl]amino}-6-[4-(2- hydroxyethyl)piperidyl](3-pyridyl))-N-(3-
0.4705 M+H 478.2 pyridylmethyljcarboxamide tert-butyl 4-(6-{[2-{3-fluorophenyl)ethyl]amino}-5-[N-(3-
59.7576 M+H 549.2 pyridylmethyl)carbamoyl](2-pyridyl))-3-oxopiperazinecarboxylate
(2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(2-oxopiperazinyl)(3-
70.7815 M+H 449.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2oxopiperidyl)(3-pyridyl))-
53.3561 M+H 385.1 N , N-dimethylcarboxamide
(6-[(3S)-3-(hydroxymethyl)pyrrolidinyl]-2-{[2-(3- fluoropheπyl)ethyl]amiπo}(3-pyridyl))-N-(3-
0.4654 M+H 450.2 pyridylmethyl)carboxamide
2-[(2-{[2-(3-fluorophenyl)ethyl]annino}-6-pyrrolidinyl(3-
0.3292 M+H 400.2 pyridyl))carboπylamiπo]-N-methylacetamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-pyrrolinyl)(3-pyridyl))-N-
0.1539 M+H 418.1 (3-pyridylmethyl)carboxam ide
(6-((3S)-3-aminopyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
28.4133 M+H 435.2 pyridylmethyl)carboxamide
N-[(3R)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
67.7779 M+H 506.2 yl](dimethylamino)carboxamide
(6-azetidinyl-2-{[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
1.1721 M+H 406.2 pyridylmethyl)carboxamide
{2-[(2-(2-pyridyl)ethyl)amino]-6-pyrrolidinyl(3-pyridyl)}-N-(3-
32.8026 M+H 403.2 pyridylmethyl)carboxamide
{6-(4-methylpiperidyl)-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-
14.3105 M+H 431.2 (3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1,3-oxazolin-2-yl)(3-
1.5042 M+H 420.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-(aminomethyl)(3-
0.3268 M+H 465.2 pyridyl)]methyl}carboxamide
N-{[2-(amiπomethyl)(3-pyridyl)]methyl}(6-cyclopent-1-enyl-2-{[2-
0.0146 M+H 446.2 (3-fiuorophenyl)ethyl]amino}(3-pyridyf))carboxamide
(6-((3S)-3-fIuoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-(aminomethyl)(3-
0.0882 M+H 467.1 pyridyl)]methyl}carboxamide
IC50
Arithmetic Mean Ion m/z ChemicalName
(6-<(3R)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-(aminomethyl)(3-
0.0313 M+H 467.1 pyridyl)]methyl}carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- 0.0656 M+H 463.2 fluorophenyl)ethyl]amino}-6-piperidyl(3-pyridyl))carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(2-{[2-(3- 0.0297 M+H 449.1 fIuorophenyl)ethyl]amino}-6-pyrrolidinyl(3-pyridyl))carboxamide
N-[(3S)-1-(6-{[2-(3-fIuorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
12.277 M+H 493.2 yl]methoxycarboxamide methyl 4-(6-{[2-(3-f luorophenyl)ethyl]amino}-5-{N-[( N- methylcarbamoyl)methyl]carbamoyl}-2-pyridyl)-1 ,2,5,6- 0.6271 M+H 470.2 tetrahydropyridinecarboxylate
N-[(3R)-1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-
4.5022 M+H 493.2 yljmethoxycarboxamide
{6-piperidyl-2-[(2-(2-pyridyl)ethyl)amino](3-pyridyl)}-N-(3- 13.1958 M+H 417.1 pyridylmethyl)carboxamide
(6-((3R)-3-aminopyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
35.5447 M+H 435.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(1,2,5,6- 0.6872 M+H 432.2 tetrahydropyridyl)(3-pyridyI))-N-(3-pyridylmethyl)carboxamide
(5-chloro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3- 0.2341 454.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(έ-cyano^-CP-CS-fluorophenyOethyllaminoJ-β-pyrrolidinyKS- 0.1649 445.1 pyridyl))-N-(3-pyridylmethyl)carboxamide methyl 2-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
21.1565 477.1 pyridylmethyl)carbamoyl]-2-pyridyl)-2-imidazolinecarboxylate
(6-(1-cyano(2-imidazolin-2-yl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.4347 444.1 pyridylmethyl)carboxamide
N-{[(2R)-1-(3-aminopropyl)-5-oxopyrrolidin-2-yl]methyl}(5-fliJoro-
2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3- 0.1186 501.2 pyιϊdyl))carboxamide
(6-((3R)-3-fluoropyrrolidinyl)-5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{i2-(aminomethyl)(3-
0.023 485.1 pyridyl)]methyl}carboxamide
^-((SRJ-S-hydroxypyrrolidinyO-S-fluoro^-fP-CS- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-{[(tert-
6.7754 M+H 583.1 butoxy)carbonylamino]methyl}(3-pyridyl))methyl]carboxamide
(6-((3R)-3-hydroxypyrrolidinyl)-5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{f2-(aminomethyl)(3-
0.1249 M+H 483.1 pyridyl)]methyl}carboxamide
(6-(1-acetyl(4-1 ,2,5,6-tetrahydropyridyl))-2-{[2-(3- fluorophenyl)ethyl]amiπo}(3-pyridyl))-N-(3-
0.4299 M+H 474.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-1,2,5,6-
10.5122 M+H 432.2 tetrahydropyridyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide tert-butyl 4-(6-{{2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
2.148 M+H 532.2 pyridylmethyl)carbamoyl]-2-pyridyl)-1 , 2,5,6-
IC50
Arithmetic
Mean Ion m/z ChemicalName tetrahydrαpyridinecarboxylate
(e-cyclopentyl^-^-β-fluorophenyOethyllaminoKS-pyridyOJ-N-
4.4109 M+H 419.2 (3-pyridylmethyl)carboxamide
(6-(1 ,4-dioxaspiro[4.5]dec-7-en-8-yl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
1.8243 M+H 489.2 pyιϊdylmethyl)carboxamide methyl 4-(6-{[2-(3-fluorophenyI)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl]-2-pyridyl)-1 , 2,5,6-
0.1001 M+H 490.2 tetrahydropyridinecarboxylate
(δ-cyclopent-i-enyl^^-fS-fluorophenylJethyπaminoXS-
0.637 M+H 417.2 pyridyl))-N-(3-pyιϊdylmethyl)carboxamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrrolidinyl(3-pyridyl))carbonylamino]-isopropyl}-2-[(tert-
15.099 M+H 561.2 butoxy)carbonylamino]acetamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-oxocyclohex-1-enyl)(3-
0.6709 M+H 445.1 pyιϊdyl))-N-(3-pyridylmethyl)carboxamide
(6-((3R)-3-fluoropyrrolidinyl)-5-fluoro"-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-{[(tert-
8.243 M+H 585.2 butoxy)carbonylamino]methyl}(3-pyridyl))methyl]carboxamide
(e-cyclohexyl^p-tS-fluorophenylJethylJaminoKS-pyridylJJ-N^S-
4.1699 M+H 433.2 pyridylmethyl)carboxamide
(6-cyclohex-1-enyl-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.1169 M+H 431.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
N-[((2R)-1-{3-i(tert-butoxy)carbonylamino]propyl}-5- oxopyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3-
3.3223 M+H 601.2 fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-pyridyl))carboxamide
{6-[1-(N,N-dimethylcarbamoyl)(4-1,2,5,6-tetrahydropyridyl)]-2-
{[2-(3-fluorophenyl)ethyl]amiπo}(3-pyridyl))-N-(3-
0.4518 M+H 503.2 pyridylmethyl)carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
0.2947 M+H 498.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl)amino}-6-(1-methyl(4-1,2,5,6-
4.3572 M+H 446.2 tetrahydropyridyl))(3-pyridyl))-N-(3-pyridylmethyl)carboxamide methyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl]-2-pyridyl)-1 , 4,5,6-
5.7696 M+H 490.2 tetrahyd ropyridiπ eca rboxylate
(2-{[2-(3-fluorophenyl)ethyI]amino}-6-(2-imidazoliπ-2-yl)(3-
56.4278 M+H 419.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
N-{[(2R)-1-(4-aminobutanoyl)pyrrolidin-2-yl]methyl}(5-fluoro-2-
{[2-(3-fluorophenyl)ethyl]amino}-6-ρyrrolidinyl(3-
0.3084 M+H 515.2 pyridyl))carboxamide
N-[((2R)-1-{4-[{tert-butoxy)carbonylamino]butanoyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6-
8.5545 M+H 615.3 pyrrolidinyl(3-pyridyl))carboxamide
N-{[(2R)-1-(2-aminoacetyl)pyrrolidin-2-yl]methyl}(5-fluoro-2-{[2-
(3-fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-
0.212 M+H 487.2 pyridyl))carboxamide
N-[((2R)-1-{2-[(tert-butoxy)carbonylamino]acetyl}pyrrolidin-2- yl)methyl](5-fluoro-2-{[2-(3-fIuorophenyl)ethyl]amino}-6-
13.6492 M+H 587.3 pyrrolidinyl(3-pyridyl))carboxamide 0.0259 M+H 467.1 N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-{[2-(3-
IC50 Arithmetic Mean Ion m/z ChemicalName fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-pyridyl))carboxamide
N-[((2R)pyrrolidin-2-yl)methyl](5-fluoro-2-{[2-(3-
1.7993 M+H 430 fluorophenyl)ethyl]amiπo}-6-pyrrolidiπyl(3-pyridyl))carboxamide tert-butyl (2R)-2-[[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrrolidinyl(3-
6.8551 M+H 530.2 pyridyl))carbonylamino]methyl}pyrrolidinecarboxylate
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(6-cyclopropyl-5-fluoro-2-
1.3219 M+H 438.1 {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide
N-{(1R)-2-[(5-fluoro-2-{[2-(3-fluorophenyl)ethyl]amino}-6- pyrrolidinyl(3-pyridyl))carbonylamino]-isopropyl}-2-
0.1973 M+H 461.2 amiπoacetamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[1-(methylsulfonyl)(4-
1,2,5,6-tetrahydropyridyl)](3-pyridyl))-N-(3-
6.3961 M+H 510.2 pyridylmethyl)carboxamide
[4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))piperazinyl]-N,N-
48.4841 M+H 506.2 dimethylcarboxamide methyl 4-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
1.5317 M+H 493.2 pyridylmethyl)carbamoyl]-2-pyridyl)piperazinecarboxylate
(6-{3-aminopyrrolidinyl)-2-{[2-(3-fiuorophenyl)ethyl]amino}(3-
23.1246 M+H 435.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-morpholiπ-4-yl(3-pyridyl))-
2.6652 M+H 436.2 N-(3-pyridylmethyl)carboxamide
(6-[(2R)-2-(hydroxymethyl)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
10.5355 M+H 450.2 pyridylmethyl)carboxamide
2-[(6-((3S)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
8.2504 M+H 416.1 methylacetamide
2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-hydroxypiperidyl)(3-
9.4962 M+H 430.1 ρyridyl))carbonylamino]-N-methylacetamide
(tert-butoxy)-N-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
18.4756 M+H 535.2 pyridylmethyl)carbamoyl](2-pyridyl))pyrrolidin-3-yl]carboxamide
(6-[(3R)-3-(dimethylamino)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
75.9885 M+H 463.2 pyridylmethyl)carboxamide
2-[(6-((3S)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
1.062 M+H 418.1 methylacetamide
2-[(6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
3.6861 M+H 416.1 methylacetamide
2-t1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
5.6018 M+H 478.2 pyridylmethyl)carbamoyl]-2-pyridyl)pyrrolidin-3-yl]acetic acid
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-(4-pyridyl)pyrrolidinyl)(3-
2.968 M+H 497.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
2-[(6-(3,3-difluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))carbonylamino]-N-
0.9312 M+H 436.1 methylacetamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
0.2133 M+H 449.2 fluorophenyl)ethyl]amino}-6-pyrrolidinyl(3-pyridyl))carboxamide
IC50
Arithmetic Mean Ion m/z ChemicalName
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[3- (trifluoromethyl)pyrrolidinyl](3-pyridyl))-N-(3-
2.4916 M+H 488.2 pyridylmethyl)carboxamide
(6-(3,3-difluoropyrrolidinyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
0.274 456.1 pyridyl))-N-(3-pyridylmethyl)carboxamide N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
0.3336 M+H 463.2 fluoropheπyl)ethyl]amiπo}-6-piperidyl(3-pyridyl))carboxamide (6-((3S)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[6-(aminomethyl)(2-
3.0712 M+H 465.1 pyridyl)]methyl}carboxamide (6-((3R)-3-hydroxypyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyljamino}(3-pyridyl))-N-{[6-(aminomethyl)(2-
2.2162 M+H 465.2 pyridyl)]methyl}carboxamide (6-((3R)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]arnino}(3-pyridyl))-N-(3-
0.1066 M+H 438.2 pyridylmethyl)carboxamide
(6-[4-(aminomethyl)piperidyl]-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))-N-(3-
39.1112 M+H 463.2 pyridylmethyl)carboxamide
2-[(2-{[2-(3-fIuorophenyOethyl]amino}-6-piperidyl(3-
1.1175 M+H 414.2 pyridyl))carbonylamino]-N-methylacetamide
(2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-[3-
(methylsulfonyl)pyrrolidinyl](3-pyridyl))-N-(3-
11.54 M+H 498.2 pyridylmethyl)carboxamide
(6-[(2S)-2-(methoxymethyl)pyrrolidinyl]-2-{[2-(3- fluoropheπyl)ethyl]amino}(3-pyridyl))-N-(3-
67.3028 M+H 464.2 pyridylmethyl)carboxamide
(tert-butoxy)-N-{[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- pyridylmethyl)carbamoyl](2-pyridyl))(4-
4.5603 M+H 563.3 piperidyl)]methyl}carboxamide (tert-butoxy)-N-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
16.3821 M+H 549.2 pyridylmetnyl)carbamoyl](2-pyridyl))(4-piperidyl)]carboxamide
(2-{[2-(3-fliJorophenyl)ethyl]amiπo}-6-(4-hydroxy-4-
16.6585 M+H 526.2 pheny]piperidyl)(3-pyridyl))-N-(3-pyridylmethyl)carboxamide ethyl 1 -(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
1.5586 M+H 506.2 pyridylmethyl)carbamoyl]-2-pyridyl)piperidine-4-carboxylate
(6-((3S)-3-fluoropyrrolidinyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.4307 M+H 438.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[4-(N- methylcarbamoyl)piperidyl](3-pyridyl))-N-(3-
11.0012 M+H 491.2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(4-hydroxypiperidyl)(3-
0.7562 M+H 450.2 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-piperazinyl(3-pyridyl))-N-
31.7978 M+H 435.2 (3-pyridylmethyl)carboxamide
(6-((3S)-3-hydroxypiperidyl)-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.5153 M+H 450.2 pyridylmethyl)carboxamide
N-[1-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
20.7136 M+H 477.2 pyridylmethyl)carbamoyl]-2-pyridyl)pyrrolidiπ-3-yl]acetamide
IC50
Arithmetic
Mean Ion m/z ChemicalName
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[2-
(hydroxymethyl)morpholin-4-yl](3-pyridyl))-N-(3-
7.2059 M+H 466.2 pyridylmethyl)carboxamide
(6-[(3S)-3-(methylamino)pyrrolidinyl]-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
59.5321 M+H 449.2 pyridylmethyl)carboxamide
(2-{[2-(3-fIuorophenyl)ethyl]amino}-6-(1 ,4-thiazaperhydroin-4-
1.5743 M+H 452.1 yl)(3-pyridy!))-N-(3-pyridylmethyl)carboxamide (6-(1 , 1 -dioxo(1 ,4-thiazaperhydroin-4-yl))-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
29.8736 M+H 484.2 pyridylmethyl)carboxamide (β^-acetylpiperazinylJ^-d∑-CS-fluorophenyOethyllaminoKS-
8.5678 M+H 477.2 pyridyl))-N-(3-pyridylmethyl)carboxamide (2-{[2-(3-fluorophenyl)ethyl]amiπo}-6-(4-piperidylpiperidyl)(3-
29.7341 M+H 517.3 pyridyl))-N-(3-pyridylmethyl)carboxamide ethyl 4-(6-{[2-(3-fluorophenyl)ethyl]annino}-5-[N-(3-
3.1634 M+H 507.2 pyridylmethyl)carbamoylJ-2-pyridyl)piperazinecarboxylate
Example 18:
(£)-2-(3-fluorophenethylamino)-6-(prop-l-enyl)-Λ^-(pyridin-3-ylmethyl)nicotinamide
[0279] A mixture of acid 2,6-dichloronicotinic acid (19.2 g, 0.1 mol), N1N'- carbonyldiimidazole (CDI, 17.8 g, 0.11 mmol) and THF (1 L) was stirred at 60°C for 30 min and cooled to room temperature. To the mixture was added 3-aminomethyl pyridine (10.2 mL, 0.1 mol) and DIEA (21 mL, 0.12 mol). The reaction mixture was stirred at room temperature for 4 hrs, and diluted with EtOAc. The organic layer was washed with H2θ, brine, dried over Na2SO4. Concentration of the organic solution gave the 2,6- dichloro-N-(pyridin-3-ylmethyl)nicotinamide (20.5 g, 73%). LRMS (M+H+) m/z 282.0; (M+2+H+) m/z 283.9.
[0280] A mixture of chloride 2,6-dichloro-N-(pyridin-3-ylmethyl)nicotinamide (20.4 g, 72.3 mmol), 3-fluorophenethyl amine (11.1 g, 79.5 mmol), K2CO3 (20 g 145 mmol) and DMF (200 mL) was stirred at 1000C for 10 hours. The mixture was diluted with ethyl acetate (2 X 300 mL) and washed with saturated ammonium chloride, sodium bicarbonate and brine. The organic layers were combined, dried (MgSO4) and ∞110611^1^. The residue was purified by silica gel chromatography to give the desired product 6- chloro-2-(3-fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide (12.6 g, 46%). LRMS (M+H+) m/z 385.1.
[0281] A mixture of chloride 6-chloro-2-(3-fluorophenethylamino)-N-(pyridin-3- ylmethyl)nicotinamide (100 mg, 0.260 mmol), trans-1-propen-1-yl boronic acid (27 mg, 0.312 mmol), PdCI2(dppf)2 (14 mg, 0.026 mmol), 2N K2CO3 (0.39 mL, 0.78 mmol) and THF (1.5 mL) was stirred at 1250C for 30 min. The mixture was filtered through the silica gel pad and washed with EtOAc. The filtrate was concentrated to dryness and purified on RP-HPLC using a mixture of acetonitrile and H2O to give (E)-2-(3- fluorophenethylamiπo)-6-(prop-1-enyl)-N-(pyridin-3-ylmethyl)nicotinamide as a pale yellow solid (59 mg, 86%). LRMS (M+H+) m/z 391.2.
Example 19:
[0282] A mixture of 6-chloro-2-(3-fluorophenethylamino)-N-(pyridin-3- ylmethy])nicotinamide (1.3 g, 3.3 mmol), NiBr2 (1.1 g, 4.9 mmol), NaCN (0.48 g, 9.8 mmol) and NMP (6 mL) was stirred at 2200C for 1 hour. The reaction mixture was filtered through Celite with EtOAc. The organics were washed with water, brine, dried (Na2SO4) and concentrated to give the nitrile 6-cyano-2-(3-fluorophenethylamino)-N- (pyridin-3-ylmethyl)nicotinamide (0.85 g, 69%). LC/MS (M+H+) m/z 376.1.
[0283] A three neck round bottom flask fixed with a dry ice acetone condenser was charged with the nitrile nitrile 6-cyano-2-(3-fluorophenethylamino)-N-(pyridin-3- ylmethyl)nicotinamide (0.10 g, 0.27 mmol), EtOH (2 mL) and stirred with HCICg)for 20 minutes. The reaction mixture was then concentrated to dryness to give the ethyl 6-(3- fluorophenethylamino)-5-(pyridin-3-ylmethylcarbamoyl)picolinimidate. LC/MS (M+H+) m/z 422.1.
[0284] A mixture of the ethyl 6-(3-fluorophenethylamino)-5-(pyridin-3- ylmethylcarbamoyl)picolinimidate and NH3ZMeOH (7N1 0.76 mL) was stirred at 500C for 1 hour. The reaction mixture was concentrated to dryness to give 6-carbamirnidoyl-2-(3- fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide. LC/MS (M+H+) m/z 394.1.
II
[0285] A mixture of 6-carbamimidoyl-2-(3-fluorophenethylamino)-N-(pyridin-3- ylmethyl)nicotinamide (0.013 g, 0.034 mmol), CNBr (3N, 0.017 mL, 0.051 mmol), DIPEA (0.0085 mL, 0.051 mmol), in DCM (0.10 mL) was stirred at room temperature until the reaction was complete. The reaction mixture was concentrated to dryness and purified by RP-HPLC using a mixture of acetonitrile and H2O to give 6-(N- cyanocarbamimidoyl)-2-(3-fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide as a solid (0.0058 g, 41%). LC/MS (M+H+) m/z 418.1.
Example 20
[0286] Using procedures similar to those described herein, the compounds in the following table were synthesized and tested.
IC50 Ion m/z CHEMICAL NAME
Arithmetic
Mean
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
457. fluorophenyl)ethyl]amino}-5-(3-pyridyl)(3-
7.3929 M+H 1 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
457. fluorophenyl)ethyl]amino}-5-(4-pyridyl)(3-
9.8364 M+H 1 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3-
460. fluorophenyl)ethyl]amino}-5-(1-methylpyrazol-4-
17.7374 M+H 1 yl)(3-pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(5-(2-
481. cyanophenyl)-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
6.0282 M+H 1 pyridyl))carboxamide
376. (5-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
12.683 M+H 1 pyridyl))-N-(2-pyridylrnethyl)carboxamide
(5-bromo-2-{[2-(3-fluorophenyl)ethyl]amiπo}(3- pyridyl))-N-[(6-methyI(2-
17.7243 M+H 445 pyridyl))methyl]carboxamide
(5-bromo-6-chloro-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(2-
17.258 M+H 446655 pyridylmethyl)carboxamide 5-bromo-2-{[2-(3-
11.19 M+H 341 fluorophenyl)ethyl]amino}pyridine-3-carboxylic acid
300. methyl 5-cyaπo-2-{[2-(3-
14.887 M+H 1 fluorophenyl)ethyl]amino}pyridiπe-3-carboxylate N-({6-[(1,3-dioxobenzo[c]azolin-2-yl)methyl](2- pyridyl)}methyl)(5-bromo-2-{[2-(3- /
9.7159 M+H 559900 fluorophenyl)ethyl]amino}(3-pyridyl))carboxamide (5-bromo-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
11.8154 M+H 431 pyridyl))-N-(2-pyridylmethyl)carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(5-bromo-2-
{[2-(3-flυorophenyl)ethyl]amino}(3-
15.0424 M+H 460 pyridyl))carboxamide
N-{[6-(aminomethyl)(2-pyridyl)]methyl}(2-{[2-(3- 457. fluorophenyl)ethyl]amino}-5-(2-pyridyl)(3-
16.0219 M+H 1 pyridyl))carboxamide
389. methyl 6-[(4-carbamoylbutyl)amino]-2-{[2-(3-
44.3072 M+H 2 fluorophenyl)ethyl]amino}pyridine-3-carboxylate 5-[(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
493. pyridylmethyl)carbamoyl](2-pyridyl))amino]-N,N-
55.0688 M+H 2 dimethylpentanamide
5-[(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- 479. pyridylmethyl)carbamoyl](2-pyridyl))amino]-N-
38.046 M+H 2 methylpentanamide
(6-[(4-carbamoylbutyl)amino]-2-{[2-(3- 465. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
6.7378 M+H 2 pyridylmethyl)carboxamide
4-[(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3- 479. pyridylmethyl)carbamoyl](2-pyridyl))amino]-N,N-
70.3134 M+H 2 dimethylbutanamide
(6-[(3-carbamoylpropyl)amino]-2-{[2-(3- 451. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
42.7772 M+H 2 pyridylmethyl)carboxamide (6-(dimethylamino)-2-{[2-(3- 394. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
4.919 M+H 1 pyridylmethyl)carboxamide 5-[(6-{[2-(3-fluorophenyl)ethyl]amino>-5-[N-(3-
556. pyridylmethyl)carbamoyl](2-pyridyl))amino]-N-(3-
49.2162 M+H 2 pyridylmethyl)pentanamide
380. (2-{[2-(3-fluorophenyl)ethyl]amino}-6-
10.883 M+H 2 (methylamino)(3-pyridyl))-N-(3-
pyridylmethyl)carboxamide
N-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
422. pyridylmethyl)carbamoylJ(2-pyridyl))-N-
33.5035 M+H 2 methylacetamide
N-(6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
452. pyridylmethyl)carbamoyl](2-pyridyl))-N-(2-
53.412 M+H 2 hydroxyethyl)acetamide
394. 6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
13.9674 M+H 1 pyridylmethyl)carbamoyl]pyridine-2-carboxamide
376. (6-cyano-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
65.8061 M+H 1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(3-
457. pyridylmethyl)amino]{3-pyridyl))-N-(3-
5.6092 M+H 1 pyridylmethyl)carboxamide
(6-[(cyanoamino)iminomethyl]-2-{[2-(3-
418. fluorophenyl)ethyl3amino}(3-pyridyl))-N-(3-
0.2309 M+H 1 pyridylmethyl)carboxamide
(6-((1 Z)- 1 -methylprop-1 -enyl)-2-{[2-(3-
405. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
10.5082 M+H 1 py ridy lmethyl)carboxa m ide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(N-
408. methylcarbamoyl)(3-pyridyl))-N-(3-
44.1181 M+H 1 pyridylmethyl)carboxamide
409. methyl 6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
4.3507 M+H 1 pyridylmethyl)carbamoyl]pyridine-2-carboxylate
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(2-
457. pyridylmethyl)amino](3-pyridyl))-N-(3-
10.7731 M+H 2 pyridylmethyl)carboxamide
(6-((1 E)prop-1 -enyl)-5-bromo-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.7796 M+H 469 pyridylmethyl)carboxamide
methyl (2E)-3-(6-{[2-(3-fIuorophenyl)ethyl]amino}-
449. 5-[N-(3-pyridylmethyl)carbamoyl](2-pyridyl))but-2-
3.4457 M+H 1 enoate
(2-{[2-(3-fluoropheny!)ethyl]amino}-6-[(4-
457. pyridylmethyl)amino](3-pyridyl))-N-(3-
33.9795 M+H 1 pyridylmethyl)carboxamide
423. ethyl 6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
2.6522 M+H 1 pyridylmethyl)carbamoyl]pyridine-2-carboxylate (6-((1 E)prop-1-enyl)-2-{[2-(3-
391. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.3021 M+H 2 pyridylmethyl)carboxamide (6-((1 E)prop-1-enyl)-5-cyano-2-{[2-(3-
416. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.3608 M+H 1 pyridylmethyl)carboxamide (2-{[2-(3-fluorophenyl)ethyl]amino}-6-(2-
405. methylprop-1-enyl)(3-pyridyl))-N-(3-
0.3473 M+H 2 pyridylmethyl)carboxamide (6-[(dimethylamino)iminomethyl]-2-{[2-(3-
421. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
17.2629 M+H 2 pyridylmethyl)carboxamide
(6-[(1 E)-1 -(dimethylamino)-2-cyano-2-azavinyl]-2-
446. {[2-(3-fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
5.1875 M+H 1 pyridylmethyl)carboxamide
339933.. 6-{[2-(3-fluorophenyl)ethyl]amino}-5-[N-(3-
6.619 M+H 1 pyridylrnethyl)carbamoyl]pyridine-2-carboxamidine
407. 2-[(2-{[2-(3-fluorophenyl)ethyl]amino}-5-phenyl(3-
14.9996 M+H 1 pyridyl))carbonylamino]-N-methylaceta'mide
(6-((1 E)prop-1-enyl)-2-{[2-(3-
420. fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-
0.038 M+H 1 (aminomethyl)(3-pyridyl)]methyl}carboxamide
17.9719 M+H 432. 2-[(5-(2-cyanophenyl)-2-{[2-(3-
fluorophenyl)ethyl]amino}(3- pyridyl))carbonylamino]-N-methylacetamide
(6-((1 E)-3-methoxyprop-1 -enyl)-2-{[2-(3-
421. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.9273 M+H 2 pyridylmethyl)carboxamide
(6-(ethoxyethyl)-2-{[2-(3-
423. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
73.03 M+H 2 pyridylmethyl)carboxamide
393. (6-acetyl-2-{[2-(3-fluorophenyl)ethyl]amino}(3-
24.7905 M+H 1 pyridyl))-N-(3-pyridylmethyl)carboxamide
(6-((1 E)prop-1-enyl)-5-fluoro-2-{[2-(3-
438. fluorophenyl)ethyl]amino}(3-pyridyl))-N-{[2-
0.0805 M+H 1 (aminomethyl)(3-pyridyl)]methyl}carboxamide
(6-((1 E)prop-1 -enyl)-5-fluoro-2-{[2-(3- fluorophenyl)ethyl]amino}(3-pyridyl))-N-[(2-{[(tert-
538. butoxy)carbonylamino]methyl}(3-
11.7073 M+H 2 pyridyl))methyl]carboxamide
N-{[2-(aminomethyl)(3-pyridyl)]methyl}(5-fluoro-2-
{[2-(3-fluorophenyl)ethyl]amino}(3-
2.6897 M+H 398 pyridyl))carboxamide
351. (2-{[2-(3-fluorophenyl)ethyl]amiπo}(3-pyridyl))-N-(3-
26.8336 M+H 1 pyridylmethyl)carboxamide
(6-(1 ,2-dimethylprop-1-enyl)-2-{[2-(3-
419. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
2.7696 M+H 2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-(3-
423. methoxypropyl)(3-pyridyl))-N-(3-
8.1803 M+H 1 pyridylmethyl)carboxamide
(6-(1 -ethoxyvinyl)-2-{[2-(3-
421. fluorophenyl)ethyl]amino}(3-pyridyl))-N-(3-
0.7025 M+H 2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(2-
410. hydroxyethyl)amino](3-pyridyl))-N-(3-
13.0601 M+H 2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(2-
424. hydroxyethyl)methylamino](3-pyridyl))-N-(3-
45.4109 M+H 2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(2-
424. methoxyethyl)amino](3-pyridyl))-N-(3-
7.6223 M+H 2 pyridy!methyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(2-
438. hydroxypropyl)methylamino](3-pyridyl))-N-(3-
10.4821 M+H 2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(3-
438. methoxypropyl)amino](3-pyridyl))-N-(3-
11.0022 M+H 2 pyridylmethyl)carboxamide
(2-{[2-(3-fluorophenyl)ethyl]amino}-6-[(3-
424. hydroxypropyl)amino](3-pyridyl))-N-(3-
8.4659 M+H 2 pyridylmethyl)carboxamide
EXAMPLE 21 : Synthesis of 6-(2-cyanophenyl)-4-(3- fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide
I
6-(2-cyanophenyl)-4-(3-fluorophenethylamino)-Λ/-(pyridin-3-ylmethyl)nicotinamide
[0287] To a solution of ethyl 4,6-dichloronicotinate (2.2 g, 10.0 mmol) and 3- fluoro-phenylethylamine(1.5g, 11.Ommol) in DMF (20 ml_) was added K2CO3 (4.1g, 30 mmol) at r.t.. The reaction mixture was stirred at r.t. overnight. The reaction mixture was filtered and concentrated under reduced pressure. The resulting residue was purified on RP-HPLC using a mixture of acetonitrile and H2O tcgive ethyl 6-chloro-4-(3- fluorophenethylamino)nicotinate (1.1 mg, 34%). LRMS (M+H+) m/z 323.0.
[0288] To a mixture of ethyl 6-chloro-4-(3-fluorophenethylamino)nicotinate (1.2 g, 3.7 mmol), 2-cyano-phenylboronic acid (1.1 g, 7.4mmol) and K2CO3 (1.5 g, 11.1 mmol) in DMF (25 ml.) was added PdCtø dppf (0.2 g, 0.37 mmol). The reaction mixture was stirred at 100 0C for 4 h. LC/MS indicated the reaction was complete. The reaction mixture was filtered and the filtrate was concentrated. The resulting residue was purified on RP-HPLC using a mixture of acetonitrile and H2O to give ethyl 6-(2-cyanophenyl)-4- (3-fluorophenethylamino)nicotinate (500 mg, 34%). LRMS (M+H+) m/z 389.0.
[0289] To a solution of ethyl 6-(2-cyanophenyl)-4-(3- fluorophenethylamino)nicotinate (100 mg, 0.26 mmol) and 3-aminomethylpyridine (39 mg, 0.36mmol) in toluene (1 mL) was added AIMe3 (0.2 mL, 2M in toluene, 0.39 mmol) at r.t.. The reaction mixture was stirred at 70 0C overnight. The reaction was cooled, quenched with sat. NaHCO3, and diluted with EtOAc. The organic layer was separated, washed with brine, dried over Na2SO4, and filtered. The filtrate was concentrated under reduced pressure to give a crude oil. The crude mixture was purified on RP-HPLC using a mixture of acetonitrile and H2O to give 6-(2-cyanophenyl)-4-(3~ fluorophenethylamino)-N-(pyridin-3-ylmethyl)nicotinamide (10 mg, 8.6%). LRMS (M+H+) m/z 452.0.
Example 22
[0290] Using procedures similar to those described herein, the compounds in the following table were synthesized and tested.
IC50
Arithmetic
Mean Ion m/z Object_CH EMICALNAM E
(6-(2-cyanophenyl)-4-{[2-(3-fluorophenyl)ethyl]amino}(3- 2.02 M+H 452.1 pyridyl))-N-(3-pyridylmethyl)carboxamide
2-[(6-(2-cyanophenyl)-4-{[2-(3-fluorophenyl)ethyl]amino}(3- 48.426 M+H 432.1 pyridyl))carbonylamino]-N-methylacetamide
[4-{[2-{3-fluorophenyl)ethyl]amino}-6-(2-{imino[(3- pyridylmethyl)amino]methyl}phenyl)(3-pyridyl)]-N-(3- 61.236 M+H 560.2 pyridylmethyl)carboxamide
Example 23: In vitro Model of Dose Dependent Smooth Muscle Myosin ATPase Modulation
[0291] Screening assays were performed using a pyruvate kinase and lactate dehydrogenase-coupled ATPase assay containing the following reagents: Potassium PIPES (50 mM), MgCI2 (3 mM), KCI (100 mM), ATP (0.15 mM), DTT (1 mM), BSA (0.1 mg/ml), NADH (0.5 mM), PEP (1.5 mM), pyruvate kinase (4 U/ml), lactate dehydrogenase (8 U/ml), and antifoam (50 ppm) (concentrations expressed are final assay concentrations). The pH was adjusted to 6.80 at 22 0C by addition of potassium hydroxide. Lead optimization assays were performed with a more sensitive pyruvate kinase / horseradish peroxidase / pyruvate oxidase-coupled ATPase assay containing the following reagents: Potassium PIPES (12 mM), MgCI2 (2 mM), KCI (100 mM), ATP (0.15 mM), BSA (0.05 mg/ml), potassium phosphate (2 mM), amplex red (0.1 mM), PEP (0.1 mM), pyruvate kinase (4 U/ml), horseradish peroxidase (0.5 U/ml), pyruvate oxidase (0.5 U/ml), and antifoam (50 ppm) (concentrations expressed are final assay concentrations). The pH was adjusted to 7.00 at 22°C by addition of potassium hydroxide.
[0292] The protein components specific to this assay are chicken gizzard smooth muscle myosin subfragment-1 that has been chemically crosslinked to either cardiac or skeletal actin using an excess of 1-Ethyl-3-[3- dimethylaminopropyl]carbodiimide hydrochloride and N-hydroxysuccinimide. The exact concentration of the crosslinked smooth muscle myosin in the assay is determined empirically, by titration to achieve a desired rate of ATP hydrolysis. The concentration varies between protein preparations due to variations in the fraction of active molecules in each preparation.
[0293] Compound dose response assays are performed by first preparing a dilution series of test compound, each with an assay mixture containing potassium PIPES, MgCI2, KCI, ATP, BSA, potassium phosphate, amplex red, PEP1 crosslinked smooth muscle actomyosin (subfragment-1), antifoam, and water. The assay is started by adding an equal volume of solution containing potassium Pipes, MgCb, KCI, BSA, potassium phosphate, pyruvate kinase, horseradish peroxidase, pyruvate oxidase, antifoam, and water. ATP hydrolysis is monitored by measuring the fluorescence of amplex red (excitation at 480 nm, emission at 615 nm). The resulting dose response curve is fit by the 4 parameter equation y = Bottom + ((Top-Bottom)/(1+((IC5o/X)ΛHill))). The IC50 is defined as the concentration at which ATPase activity is midway between the top and bottom of the dose response curve.
[0294] Certain chemical entities described herein have an IC50 less than 10 μM; for example, less than 1 μM.
[0295] While the present invention has been described with reference to the specific embodiments thereof, it should be understood by those skilled in the art that various changes may be made and equivalents may be substituted without departing from the true spirit and scope of the invention. In addition, many modifications may be made to adapt a particular situation, material, composition of matter, process, process step or steps, to the objective, spirit and scope of the present invention. All such modifications are intended to be within the scope of the claims appended hereto.
Claims
1. At least one chemical entity chosen from compounds of Formula X
Formula X and pharmaceutically acceptable salts thereof wherein
W1 is chosen from N and CR6 and W2 is chosen from N and CR4, provided that both W1 and W2 are not N;
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocycloalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from hydrogen, halogen, and optionally substituted alkyl;
R3 is chosen from hydrogen and optionally substituted alkyl; and
R6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
2. At least one chemical entity of claim 1 wherein the compound of Formula X is chosen from compounds of Formula I
Formula I wherein
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted aikyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is chosen from optionally substituted aryl and optionally substituted heteroaryl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alky I, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from hydrogen and optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
3. At least one chemical entity of claim 2 wherein the compound of Formula I is chosen from compounds of Formula Il
Formula Il wherein
R1 and R4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, suifinyl, and sulfanyl;
Z1 is optionally substituted aryl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, and optionally substituted aminocarbonyl;
Z3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
4. At least one chemical entity of Claim 3 wherein Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
5. At least one chemical entity of Claim 4 wherein Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
6. At least one chemical entity of Claim 5 wherein Z2 is chosen from (2-(N- acety laminomethyl)piperidin-1 -yl)carbonyl; (2-aminomethylpiperidin-1 -yl)carbonyl; 1 -hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1 -ylcarbonyl; 2-
(methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-yl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
7. At least one chemical entity of any one of Claims 3 to 6 wherein R3 is chosen from hydrogen and optionally substituted lower alkyl.
8. At least one chemical entity of Claim 7 wherein R3 is chosen from hydrogen and lower alkyl.
9. At least one chemical entity of Claim 8 wherein R3 is chosen from hydrogen and methyl.
10. At least one chemical entity of Claim 9 wherein R3 is hydrogen.
11. At least one chemical entity of Claim 2 wherein Z2 is -C(O)NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
12. At least one chemical entity of any one of Claims 3 to 11 wherein R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl.
13. At least one chemical entity of Claim 12 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
14. At least one chemical entity of Claim 13 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-STylmethylaminocarbonyl, aminomethyl, and hydroxy methyl.
15. At least one chemical entity of Claim 14 wherein R4 is hydrogen.
16. At least one chemical entity of any one of Claims 11 to 15 wherein R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
17. At least one chemical entity of Claim 16 wherein R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
18. At least one chemical entity of Claim 17 wherein R2 is chosen from (1-(2-aminoethyl)-
1 H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin-3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1-methyl-1 H- pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-5-yl)methyl; (2-
(aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarboπyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)rnethyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridiπ-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1 -(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1 -(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-{amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1-(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; i-aminocarbonyl-2-hydroxy-eth-i-yl; 1-methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i -yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1-yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(methylsulfonamido)ethyl; 2-{methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-y!methyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yI; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-yImethyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1-yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)beπzyl; 3- (amiπomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; 3-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxypropyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamiπo)-morpholinomethyl; 4-aminobutyl; 4-amiπomethylpyridin-3-ylmethyl; 4- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; 4- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethy!; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarboπylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyt; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1 -yl)pyridiπ-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
19. At least one chemical entity of any one of Claims 3 to 18 wherein Z3 is chosen from - (CH2)rR20 wherein r is chosen from 1 , 2, and 3 and R20 is chosen from optionally substituted aryl and optionally substituted heteroaryl.
20. At least one chemical entity of Claim 19 wherein Z3 is chosen from 2-(3- methylphenyl)ethyl, 2-(1H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(2,3- difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyI, 2-(2-methylphenyl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1 H-pyrazol-1-yl)ethyl, 2-(3,5- dimethyl-1 H-pyrazol-4-yi)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fluorophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyI, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydroxyphenyl)ethyl, 2-(benzo[d][1 ,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-phenylprop-1-yl, 3-(1 H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, pyridin-2-ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
21. At least one chemical entity of Claim 20 wherein Z3 is 2-(pyridin-2-yl)ethyl.
22. At least one chemical entity of any one of Claims 3 to 21 wherein Z1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
23. At least one chemical entity of Claim 22 wherein Z1 is chosen from phenyl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, lower alkenyl, lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, lower acyl, dialkylamino, acetylamino, carbamimidoyl optionally substituted with cyano, aralkyl, and herteroaralkyl, and aminocarbonyl.
24. At least one chemical entity of Claim 23 wherein Z1 is chosen from phenyl, 2- aminocarbonylphenyl, 2-fluorophenyl, 2-chlorophenyl, 2-hydroxyphenyl, 2-methylphenyl, 2- methoxyphenyl, 2-cyanophenyl, 2-cyanomethylphenyl, 2-vinylphenyl, 2-formylphenyl, 3,4- difluorophenyl, 3-methylphenyl, 3-chlorophenyl, 3-fluorophenyl, 3-cyanophenyl, 3- dimethylaminophenyl, 3-methoxyphenyl, 3-methoxymethylphenyl, 3-hydroxyphenyl, 3- trifluoromethylphenyl, 4-aminocarbonylphenyl, 4-fluorophenyl, 4-chlorophenyl, 4-methylphenyl, 4-hydroxyphenyl, 4-cyanophenyl, 4-methoxyphenyl, 4-hydroxymethylphenyl, 4- trifluoromethylphenyl, 4-methoxycarbonylphenyl, 4-dimethylaminophenyl, 4-acetylphenyl, 4- carboxyphenyl, 4-ethylphenyl, 4-acetylaminophenyl, 3,4-dichlorophenyl, 3,4-difluorophenyl, 2,3- dimethylphenyl, 2,5-dimethylphenyl, 2,4-dimethylphenyl, 3,5-dimethylphenyl, 3,4- dimethylphenyl, 2-chloro-4-fluoro-phenyl, 2-cyano-4-fluoro-phenyl, 2-chloro-6-fluoro-phenyl, 2- chloro-4-methoxy-phenyl, 2-cyano-4-methoxy-phenyl, 2-cyano-6-fluoro-phenyl, 2-chloro-5- fluoro-phenyl, 2-cyano-5-fluoro-phenyl, 2-chloro-4-methylphenyl, 2-chloro-4-hydroxymethyl- phenyl, 2-(N-(pyridin-3-ylmethyl)carbamimidoyl)pheπyl, and 2-cyano-4-methylphenyl.
25. At least one chemical entity of any one of Claims 3 to 24 wherein R1 is chosen from hydrogen and optionally substituted lower alkyl.
26. At least one chemical entity of Claim 25 wherein R1 is chosen from hydrogen and lower alkyl.
27. At least one chemical entity of Claim 26 wherein R1 is chosen from hydrogen and methyl.
28. At least one chemical entity of claim 2 wherein the compound of Formula I is chosen from compounds of Formula III
Formula III wherein
R1 and R4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is optionally substituted heteroaryl; /
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, and optionally substituted aminocarbonyl;
Z3 is chosen from optionally substituted aralkyl and optionally substituted heteroaralkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
29. At least one chemical entity of Claim 28 wherein Z1 is chosen from 1 H-pyrazol-3-yl; 1 H- pyrazol-4-yl; 1 H-tetrazol-5-yl; 1-methyl-1H-pyrazol~4-yl; 1-methyl-1 H-pyrazol-5-yl; 2- aminomethylthiophen-5-yl; 2-cyanothiophen-3-yl; 2-formylthiophen-3-yl; 2-formylthiophen-4-yl; 2H-pyrrol-1 (5H)-yl; 2-hydroxymethylthiophen-3-yl; 2-hydroxymethylthiopheπ-4-yl; 2- hydroxymethylthiophen-5-yl; 3-aminomethylthiophen-2yl; 3-cyano-1 -methyl-1 H-pyrazol-4-yl; 3- cyanothiophen-4-yl; 3-formylthiophen-2-yl; 3-hydroxymethylthiophen-2-yl; 4-methylthiophen-2- yl; 4-methylthiophen-3-yl; 5-(1-hydroxyeth-1-yl)-thiophen-2-yl; 5-acetylthiophen-5-yl; 5-cyano- 1 H-pyrazol-4-yl; 5-cyano-1 -methyl-1 H-pyrazol-4-yl; 5-cyanothiophen-2-yl; 5-formylthiophen-2-yl; 5-hydroxymethylthiophen-2-yl; isoxazol-4-yl; pyridin-2-yl; pyridin-3-yl; pyridin-4-yl; thiazol-2- yl;thiophen-2-yl; and thiophen-3-yl.
30. At least one chemical entity of Claim 29 wherein Z1 is chosen from 1 H-pyrazol-3-yl; 1H- pyrazol-4-yl; 1 H-tetrazol-5-yl; 1 -methyl-1 H-pyrazol-4-yl; 1 -methyl-1 H-pyrazol-5-yl; 2- (hydroxymethyl)thiophen-3-yl; 2,2'-bipyridine; 2,3'-bipyridine; 2,4 -bipyridine; 2-cyanothiophen-3- yl; 2-formylthiophen-3-yl; 3-(hydroxymethyl)thiophen-2-yl; 3-cyano-1 -methyl-1 H-pyrazol-4-yl; 3- formylthiophen-2-yl; 4-cyanothiophen-3-yl; 4-methylthiophen-2-yl; 4-methylthiophen-3-yl; 5-(1- hydroxyethyl)thiophen-2-yl; 5-(aminomethyl)thiophen-2-yl; 5-(hydroxymethyl)thiophen-2-yl; 5- (hydroxymethyl)thiophen-3-yl; 5-acetylthiophen-2-yl; 5-cyano-1 H-pyrazol-4-yl; 5-cyano-1- methyl-1 H-pyrazol-4-yl; 5-cyanothiophen-2-yl; 5-formylthiophen-2-yl; 5-formylthiophen-3-yl; 3- (aminomethyl)thiophen-2-yl; isoxazol-4-yl; thiazol-2-yl; thiophen-2-yl; and thiophen-3-yl.
31. At least one chemical entity of any one of Claims 28 to 30 wherein Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
32. At least one chemical entity of Claim 31 wherein Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
33. At least one chemical entity of Claim 32 wherein Z2 is chosen from 2-(methylamino)-2- oxoethyl, 2-amino-2-oxoethyl, 3-amiπo-3-oxopropyl, (2-(N-acetylaminomethyl)piperidin-1- yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2-amino-ethyl; 2- ((methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2-(methylaminocarbonyl)ethenyl; 2-(pyridin- 3-ylmethyl)-2H-tetrazol-5-yl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
34. At least one chemical entity of any one of Claims 28 to 33 wherein R3 is chosen from hydrogen and optionally substituted lower alkyl.
35. At least one chemical entity of Claim 34 wherein R3 is chosen from hydrogen and lower alkyl.
36. At least one chemical entity of Claim 35 wherein R3 is chosen from hydrogen and methyl.
37. At least one chemical entity of Claim 36 wherein R3 is hydrogen.
38. At least one chemical entity of Claim 28 wherein Z2 is -C(O)NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
39. At least one chemical entity of any one of Claims 28 to 38 wherein R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl.
40. At least one chemical entity of Claim 39 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
41. At least one chemical entity of Claim 40 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl.
42. At least one chemical entity of Claim 41 wherein R4 is hydrogen.
43. At least one chemical entity of any one of Claims 38 to 42 wherein R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
44. At least one chemical entity of Claim 43 wherein R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
45. At least one chemical entity of Claim 44 wherein R2 is chosen from (1-(2-aminoethyl)- 1H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin-3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1H-imidazol-2-yl)methyl; (1H-pyrazol-3-yl)methyl; (1-methyl-1 H- pyrazol-3-yl)methyl; (1 -methyl-1 H-pyrazol-5-yl)methyl; (2-
(aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylrnethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1-(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(ethoxycarbonylrnethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamiπo)eth- 1-yl; 1-(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; i-aminocarbonyl-2-hydroxy-eth-i-yl; 1-methoxycarbonyl-2- hydroxy-eth-1 -yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(ethoxycarbσnyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1 -yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridiπ-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridfn-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1-yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylamiπocarboπyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; 3-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxypropyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(NlN-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; A- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; 4- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methyIamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridrn-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1-yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-yl methyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
46. At least one chemical entity of any one of Claims 28 to 45 wherein Z3 is chosen from - (CH2)rR20 wherein r is chosen from 1 , 2, and 3 and R20 is chosen from optionally substituted aryl and optionally substituted heteroaryl.
47. At least one chemical entity of Claim 46 wherein Z3 is chosen from 2-{3- methylphenyl)ethyl, 2-(1H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imidazol-4-yl)ethyl, 2-(2,3- difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1 H-pyrazol-1-yl)ethyl, 2-(3,5- dimethyl-1H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fluorophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydroxyphenyl)ethyl, 2-(benzo[d][1,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-phenylprop-1-yl, 3-(1H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, pyridin-2-ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
48. At least one chemical entity of Claim 47 wherein Z3 is chosen from 3-fluorophenethyl, 3,5-difluorophenethyl, and 2-(pyridin-2-yl)ethyl.
49. At least one chemical entity of any one of Claims 28 to 48 wherein R1 is chosen from hydrogen and optionally substituted lower alkyl.
50. At least one chemical entity of Claim 49 wherein R1 is chosen from hydrogen and lower alkyl.
51. At least one chemical entity of Claim 50 wherein R1 is chosen from hydrogen and methyl.
52. At least one chemical entity of Claim 51 wherein R1 is hydrogen.
53. At least one chemical entity of Claim 2 wherein the compound of Formula I is chosen from compounds of Formula IV
Formula IV
wherein
R1 and R4 are independently chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is chosen from optionally substituted aryl and optionally substituted heteroaryl;
Z2 is chosen from hydrogen, carboxyl, optionally substituted alkoxy carbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted amidino, and optionally substituted aminocarbonyl;
Z3 is optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
54. At least one chemical entity of Claim 53 wherein Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
55. At least one chemical entity of Claim 54 wherein Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted pyridinylmethylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
56. At least one chemical entity of Claim 55 wherein Z2 is chosen from pyridin-3- ylmethylcarbonyl, (2-(N-acetylaminomethyl)piperidin-1 -yl)carbonyl; (2-aminomethylpiperidin-1 - yl)carbonyl; 1-hydroxy-2-amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2- (methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-ylmethyl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
57. At least one chemical entity of any one of Claims 53 to 56 wherein R3 is chosen from hydrogen and optionally substituted lower alkyl.
58. At least one chemical entity of Claim 57 wherein R3 is chosen from hydrogen and lower alkyl.
59. At least one chemical entity of Claims 58 wherein R3 is chosen from hydrogen and methyl.
60. At least one chemical entity of Claim 59 wherein R3 is hydrogen.
61. At least one chemical entity of Claim 53 wherein Z2 is -C(O)NR2R5 wherein R2 is chosen from optionally substituted alkyl and optionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
62. At least one chemical entity of any one of Claims 53 to 61 wherein R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl.
63. At least one chemical entity of Claim 62 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
64. At least one chemical entity of Claim 63 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl.
65. At least one chemical entity of Claim 64 wherein R4 is hydrogen.
66. At least one chemical entity of any one of Claims 61 to 65 wherein R5 is chosen from hydrogen and lower alkyl.
67. At least one chemical entity of Claim 66 wherein R5 is chosen from hydrogen and methyl.
68. At least one chemical entity of Claim 67 wherein R5 is hydrogen.
69. At least one chemical entity of any one of Claims 61 to 68 wherein R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
70. At least one chemical entity of Claim 69 wherein R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
71. At least one chemical entity of Claim 70 wherein R2 is chosen from (1-(2-aminoethyl)- 1 H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; <1-acetylpiperidin-3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl) methyl; (1-methyl-1 H- pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-5-yl)methyl; (2-
(aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- <hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonyImethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1 ,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)rnethyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1 -(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1 -(aminocarbonyl)eth-i -yl; 1 -(carboxy)-2-(am ino)-eth-1 -yl; 1 - (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1-(methoxycarbonyl)-2-(amino)-eth-1-yl; 1-(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; 1-aminocarbonyl-2-hydroxy-eth-1-yl; 1-methoxycarbonyl-2- hydroxy-eth-1 -yl; 2-(2-aminoethoxy)ethyl; 2-{2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1 -yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyI)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-yImethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyI; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; 3-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxy propyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; 4- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; A- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyr, 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethy!)-pyridin-2-ylmethyl; 5- aminopentyl; 5-amiπopyridiπ-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1 -yl)pyridin-3-ylmethyl); 6-(amiπomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylamiπocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
72. At least one chemical entity of Claim 71 wherein R2 is pyridin-3-ylmethyl.
73. At least one chemical entity of any one of Claims 53 to 72 wherein Z3 is chosen from - (CH2)rR20 wherein r is chosen from 1 , 2, and 3 and R20 is chosen from optionally substituted heterocycloalkyl and optionally substituted cycloalkyl.
74. At least one chemical entity of Claim 73 wherein Z3 is chosen from (tetrahydrofuran-2- yl)methyl, 1-hydroxycyclohexylmethyl, 2-(1 ,3-dioxoIan-2-yl)ethyl, 2-(1 -methylpiperidin-4-yl)ethyl, 2-(2-oxoimidazolidin-1-yl)ethyl, 2-(2-oxopiperidin-1-yl)ethyl, 2-(2-oxopyrrolidin-1-yl)ethyl, 2- cyclohex-1-enylethyl, 2-cyclohexylethyl, and 2-morpholinoethyl.
75. At least one chemical entity of any one of Claims 53 to 74 wherein Z1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl, and heteroaryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
76. At least one chemical entity of Claim 75 wherein Z1 is chosen from aryl optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted lower acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and amino optionally substituted with alkyl, and heteroaryl optionally substituted by one or two groups selected from optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted lower acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and amino optionally substituted with alkyl.
77. At least one chemical entity of Claim 76 wherein Z1 is chosen from phenyl, 1H-pyrazol-3- yl; 1 H-pyrazol-4-yl; 1 H-tetrazol-5-yl; 1-methyl-1 H-pyrazol-4-yl; 1-methyl-1H-pyrazol-5-yl; 2- aminomethylthiophen-5-yl; 2-cyanothiophen-3-yl; 2-formylthiophen-3-yl; 2-formylthiophen-4-yl; 2H-pyrrol-1 (5H)-yl; 2-hydroxymethylthiophen-3-yl; 2-hydroxymethylthiophen-4-yl; 2- hydroxymethylthiophen-5-yl; 3-aminomethylthiophen-2yl; 3-cyano-1-methyl-1H-pyrazol-4-yl; 3- cyanothiophen-4-yl; 3-formylthiophen-2-yl; 3-hydroxymethylthiophen-2-yl; 4-methylthiophen-2- yl; 4-methylthiopheπ-3-yl; 5-(1-hydroxyeth-1-yl)-thiophen-2-yl; 5-acetylthiophen-5-yl; 5-cyano- 1 H-pyrazol-4-yl; 5-cyano-1-methyl-1H-pyrazol-4-yl; 5-cyanothiophen-2-yl; 5-formylthiophen-2-yl; 5-hydroxymethylthiophen-2-yl; isoxazol-4-yl; pyridin-2-yl; pyridin-3-yl; pyridin-4-yl; thiazol-2- yl;thiophen-2-yl; and thiophen-3-yl.
78. At least one chemical entity of any one of Claims 53 to 77 wherein R1 is chosen from hydrogen and optionally substituted lower alkyl.
79. At least one chemical entity of Claim 78 wherein R1 is chosen from hydrogen and methyl.
80. At least one chemical entity of Claim 79 wherein R1 is hydrogen.
81. At least one chemical entity of claim 1 wherein the compound of Formula X is chosen from compounds of Formula V
Formula V wherein R1 is selected from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl;
Z1 is selected from hydrogen, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, optionally substituted heterocydoalkyl , cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl; optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl,
Z2 is selected from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocydoalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is selected from optionally substituted alkyl and optionally substituted heteroaralkyl;
R3 is chosen from hydrogen and optionally substituted alkyl; and
R6 is chosen from hydrogen, optionally substituted acyl, optionally substituted alkyl, cyano, halo, azido, optionally substituted amino, optionally substituted alkoxycarbonyl, optionally substituted aminocarbonyl, sulfonyl, sulfinyl, and sulfanyl.
82. At least one chemical entity of Claim 81 wherein Z1 is chosen from phenyl substituted with one, two or three groups chosen from halo, alkyl, alkoxy, cyano, and optionally substituted acyl.
83. At least one chemical entity of Claim 82 wherein Z1 is chosen from 3,4-difluorophenyl and 2-cyanophenyl.
84. At least one chemical entity of any one of Claims 81 to 83 wherein Z2 is chosen from methylaminocarbonyl, methylaminocarbonylmethylaminocarbonyl, ethylaminocarbonyl, propylaminocarbonyl, and isopropylaminocarbonyl, wherein the methyl, ethyl, propyl, and isopropyl group is optionally substituted with one, two or three groups independently selected from optionally substituted methoxy, optionally substituted ethoxy, optionally substituted phenyl, and optionally substituted pyridinyl.
85. At least one chemical entity of Claim 84 wherein Z2 is chosen from 2-methoxyeth-1- ylaminocarbonyl, benzylaminocarbonyl, isopropylaminocarbonyl, pyridin-3- ylmethylaminocarbonyl, and pyridin-4-ylmethylaminocarbonyl.
86. At least one chemical entity of any one of Claims 81 to 85 wherein Z3 is chosen from ethyl, methyl and propyl substituted with one, two or three groups independently selected from optionally substituted aryl and optionally substituted heteroaryl.
87. At least one chemical entity of Claim 86 wherein Z3 is chosen from ethyl and methyl substituted with a group selected from optionally substituted phenyl and optionally substituted pyridinyl.
88. At least one chemical entity of Claim 87 wherein Z3 is chosen from 2-(3- fluorophenyl)ethyl, 2-(3-methylphenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(3-methylphenyl)ethyl and pyridin-2-ylmethyl.
89. At least one chemical entity of any one of Claims 81 to 88 wherein R1 is chosen from hydrogen and optionally substituted lower alkyl.
90. At least one chemical entity of Claim 89 wherein R1 is chosen from hydrogen and lower alkyl.
91. At least one chemical entity of Claim 90 wherein R1 is chosen from hydrogen and methyl.
92. At least one chemical entity of Claim 91 wherein R1 is hydrogen.
93. At least one chemical entity of any one of Claims 81 to 92 wherein R3 is chosen from hydrogen and optionally substituted lower alkyl.
94. At least one chemical entity of Claim 93 wherein R3 is chosen from hydrogen and lower alkyl.
95. At least one chemical entity of Claims 94 wherein R3 is chosen from hydrogen and methyl.
96. At least one chemical entity of Claim 95 wherein R3 is hydrogen.
97. At least one chemical entity of any one of claims 81 to 96 wherein Rβ is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alky I.
98. At least one chemical entity of Claim 97 wherein Rδ is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
99. At least one chemical entity of Claim 98 wherein R6 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl.
100. At least one chemical entity of Claim 99 wherein R6 is hydrogen.
101. At least one chemical entity of claim 1 wherein the compound of Formula X is chosen from compounds of Formula Vl
Formula Vl
wherein R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, sulfanyl, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted carbaminodoyl;
Z1 is chosen from optionally substituted cycloalkyl and optionally substituted heterocycloalkyl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from halo and optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
102. At least one chemical entity of Claim 101 wherein Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
103. At least one chemical entity of Claim 102 wherein Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
104. At least one chemical entity of Claim 103 wherein Z2 is chosen from (2-(N- acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1-ylcarbonyl; 2-
(methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-yl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
105. At least one chemical entity of any one of Claims 101 to 104 wherein R3 is chosen from hydrogen and optionally substituted lower alkyl.
106. At least one chemical entity of Claim 105 wherein R3 is chosen from hydrogen and lower alkyl.
107. At least one chemical entity of Claims 106 wherein R3 is chosen from hydrogen and methyl.
108. At least one chemical entity of Claim 107 wherein R3 is hydrogen.
109. At least one chemical entity of Claim 101 wherein Z2 is -C(O)NR2R5 wherein R2 is cchhoosseenn ffrroomm ooppttiioonnaallllyy ssuubbssttiittuutteedd aallkkyyll aanndd oopp'tionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
110. At least one chemical entity of any one of Claims 101 to 109 wherein R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl.
111. At least one chemical entity of Claim 110 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
112. At least one chemical entity of Claim 1 11 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl.
113. At least one chemical entity of Claim 12 wherein R4 is hydrogen.
114. At least one chemical entity of any one of Claims 109 to 113 wherein R5 is chosen from hydrogen and lower alkyl.
115. At least one chemical entity of Claim 114 wherein R5 is chosen from hydrogen and methyl.
116. At least one chemical entity of Claim 115 wherein R5 is hydrogen.
117. At least one chemical entity of any one of Claims 109 to 116 wherein R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
118. At least one chemical entity of Claim 117 wherein R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
119. At least one chemical entity of Claim 118 wherein R2 is chosen from (1-(2-aminoethyl)- 1 H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1 -acetylpiperidin-3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl; (1H-pyrazol-3-yl)methyl; (1-methyl-1H- pyrazol-3-yl)methyl; (1-methyl-1 H-pyrazol-5-yl)methyl; (2-
(arninocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylrnethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylarnino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1-(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1-(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amiπo)-pentyl; 1 -(aminocarbonyl)eth-i -yl; 1 -(carboxy)-2-(amino)-eth-1 -yl; 1 - (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 1 -(hydroxy)-2-(aminocarbonylmethylamino)eth-1 -yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1-yl; 1-(methoxycarbonyl)-2-(amiπo)-eth-1 -yl; 1-(methoxycarbonyl)-2-(t-butoxycarboπylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; 1-aminocarbonyl-2-hydroxy-eth-1 -yl; 1-methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-{2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i -y I; 2-(methoxycarbonyl)-2-(amino)-eth-1 -yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1 -yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1-yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amiπo)-eth-i-yl; 2-(N-(t-butoxycarbonyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1-yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethyl; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyl; 2-chloropyridin-5-ylmethyl; 2-cyanopyridiπ-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyridin-5-ylmethyl; 2-methylpyridin-6-ylmethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1-yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyl; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; S-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxy propyl; 3-methoxypropyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylannino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; 4- cyanobenzyl; 4-fluorobeπzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; 4- morphoIinopyπdin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5- ((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridiπ-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis{dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1-yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridiπ-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
120. At least one chemical entity of any one of Claims 101 to 119 wherein Z3 is chosen from halo and optionally substituted lower alkyl.
121. At least one chemical entity of Claim 120 wherein Z3 is chosen from halo, optionally substituted methyl, optionally substituted ethyl, and optionally substituted propyl wherein the methyl, ethyl and propyl groups are optionally substituted with one or two groups selected from hydroxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, and optionally substituted aryloxy.
122. At least one chemical entity of Claim 121 wherein Z3 is chosen from 2-{3- methylphenyl)ethyl, (tetrahydrofuran-2-yl)methyl, 1-hydroxycyclohexylmethyl, 2-(1 ,3-dioxolan-2- yl)ethyl, 2-(1 H-imidazol-4-yl)ethyl, 2-(1-methyl-1 H-imidazol-4-yl)ethyl, 2-(1-methylpiperidin-4- yl)ethyl, 2-(2,3-difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyi, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(2-oxoimidazolidin-1-yl)ethyl, 2-(2- oxopiperidin-1 -yl)ethyl, 2-(2-oxopyrrolidin-1 -yl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dimethyl-1H-pyrazσl-1-yl)ethyl, 2-(3,5- dimethyl-1H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fluorophenyl)ethyl, 2-(3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyl)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydroxyphenyl)ethyl, 2-(benzo[d][1,3]dioxol-5-yl)ethyl, 2-{furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-cyclohex-1-enylethyl, 2-cyclohexylethyl, 2-morpholinoethyl, 2-phenoxyethyl, 2-phenylprop-1- yl, 3-{1H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, chloro, isopentyl, propyl, pyridin-2- ylmethyl, pyridin-3-ylmethyl, and thiophen-3-ylmethyl.
123. At least one chemical entity of Claim 122 wherein Z3 is chosen from 2-(3- fluorophenyl)ethyl and 2-(pyridin-2-yl)ethyl.
124. At least one chemical entity of any one of Claims 101 to 123 wherein Z1 is chosen from cycloalkyl and heterocycloalkyl, where each group is optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted amino, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
125. At least one chemical entity of Claim 124 wherein Z1 is chosen from cycloalkyl and heterocycloalkyl, where each group is optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, optionally substituted lower alkoxy, carboxyl, optionally substituted lower alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
126. least one chemical entity of Claim 125 wherein Z1 is chosen from 1-(2H)- (methoxycarbonyl)-3,4-dihydropyridin-4-yl; 1 -(2H)-(methoxycarbonyl)-5,6-dihydropyridin-4-yl; 1 - (2H)-(methoxymethyl)-5,6-dihydropyridin4-yl; 1-(2H)-(t-butoxycarbonyl)-5,6-dihydropyridin-4-yl; 1-(2H)-5,6-dihydropyridin-4-yl; 1-(acetyl)-4,5-dihydroimidazo-2-yl; i-(dimethylcarbamoyl)-
1 ^.S.θ-tetrahydropyridin^-yl; 1-(methoxymethyl)-4,5-dihydroimidazo-2-yl; 1-cyano-4,5-dihydro- 1 H-imidazol-2-yl; 1-methyl-1 ^^.δ-tetrahydropyridin^-yl; 2-{acetylaminomethyl)pyrrolidin-1-yl; 2-(hydroxymethyl)morpholino; 2-(hydroxymethyl)pyrrolidin-1 -yl; 2-(methoxymethyl)pyrrolidin-1 - yl; 2H-pyrrol-1 (5H)-yl; 2-hydroxymorpholino; 2-methyl-4-(t-butoxycarbonyl)-piperaziπ-1-yl; 2- oxo-5-hydroxy-piperidinyl; 2-oxopiperazinyl; 2-oxopiperidinyl; 2-oxopyrrolidinyl; 3-(3,3- dimethylureido)pyrrolidin-1-yl; 3-(aminomethyl)-pyrrolidin-1-yl; 3-(carboxymethyl)-pyrrolidinyl; 3- (dimethylamino)pyrrolidin-i -yl; 3-(dimethylaminomethyl)-pyrrolidin-1-yl; 3- (hydroxymethyl)pyrrolidin-i -yl; 3-(methoxycarbonylamino)-pyrrolidinyl; 3- (methylsulfonyl)pyrrolidin-i-yl; 3-(N-acetyl-N-methyl)-aminomethyl)-pyrrolidin-1-yl; 3-(pyridin-4- yl)pyrrolidin-1 -yl; 3-(t-butoxycarboπylamino)-pyrrolidinyl; 3-(trifluoromethyl)pyrrolidin-1 -yl; 3,3- difluoropyrrolidinyl; 3-acetylaminopyrrolidinyl; 3-aminopyrrolidin-1-yl; 3-fluoropyrrolidin-1-yl; 3- hydroxymethylpyrrolidin-1-yl; 3-hydroxypyrrolidin-1-yl;3-methoxymethylpyrrolidin-1-yl; 3- methylamino-pyrrolidinyl; 3-oxopiperazin-1-yl; 4-(acetyl)-piperazin-1-yl; 4- (aminomethyl)piperidin-i-yl; 4-(ethoxycarbonyl)-piperazin-1-yl; 4-(ethoxycarbonyl)-piperidinyl; 4- (methoxycarbonyl)-piperazin-i-yl; 4-(methylsulfonyl)cyclohex-1-enyl; 4-(N1N- dimethylaminocarbonyl)-piperazin-1 -yl; 4-(t-butoxycarbonylamino)-piperidin-1 -yl; 4-(t- butoxycarbonylaminomethyl)-piperidin-1 -yl; 4,5-dihydro-1 H-imidazol-2-yl; 4,5-dihydrooxazol-2- yl;4-hydroxy-4-phenylpiperidin-1-yl;4-hydroxyethyl-piperidin-yl;4-hydroxyrnethyl-piperidin-yl;4- hydroxypiperidinyl; 4-methylaminocarbonyl-piperidinyl; 4-methylpiperazin-1-yl; 4- methylpiperidinyl; 4-oxocyclohex-1-enyl; 5,6-dihydropyridin-1(2H)-yl; azetidinyl; cyclohex-1-enyl; cyclohexyl; cyclopent-1 -enyl; cyclopentyl; morpholino; piperazinyl; piperidinyl.pyrrolidinyl; and thiomorpholino.
127. At least one chemical entity of any one of Claims 101 to 126 wherein R1 is chosen from hydrogen and optionally substituted lower alkyl.
128. At least one chemical entity of Claim 127 wherein R1 is chosen from hydrogen and lower alkyl.
129. At least one chemical entity of Claim 128 wherein R1 is chosen from hydrogen and methyl
130. At least one chemical entity of Claim 129 wherein R1 is hydrogen.
131. At least one chemical entity of claim 1 wherein the compound of Formula X is chosen from compounds of Formula VII
Formula VII
wherein
R1 and R4 are independently chosen from hydrogen, cyano, halo, hydroxy, azido, nitro, sulfonyl, sulfinyl, su If any I, optionally substituted alkoxy, optionally substituted acyloxy, optionally substituted aminocarbonyloxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted aryloxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted amino, optionally substituted acyl, optionally substituted aminocarbonyl, and optionally substituted carbaminodoyl;
Z1 is chosen from hydrogen, cyano, optionally substituted alkyl, optionally substituted acyl, optionally substituted alkoxycarbonyl, optionally substituted amino, optionally substituted aminocarbonyl, optionally substituted carbamimidoyl, optionally substituted alkoxy, optionally substituted aryloxy, optionally substituted heteroaryloxy, optionally substituted heterocycloalkyloxy, optionally substituted alkenyl, optionally substituted alkynyl, sulfonyl, sulfinyl, and sulfanyl;
Z2 is chosen from hydrogen, optionally substituted amidino, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, and optionally substituted aminocarbonyl;
Z3 is chosen from optionally substituted alkyl; and
R3 is chosen from hydrogen and optionally substituted alkyl.
132. At least one chemical entity of Claim 131 wherein Z2 is chosen from carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted alkenyl, and optionally substituted alkyl.
133. At least one chemical entity of Claim 132 wherein Z2 is chosen from carboxyl, optionally substituted piperidinylcarbonyl, optionally substituted pyridinylcarbonyl, optionally substituted lower alkyl, optionally substituted lower alkenyl, and lower alkoxycarbonyl.
134. At least one chemical entity of Claim 133 wherein Z2 is chosen from (2-(N- acetylaminomethyl)piperidin-1-yl)carbonyl; (2-aminomethylpiperidin-1-yl)carbonyl; 1-hydroxy-2- amino-ethyl; 2-((methylsulfonamido)methyl)piperidin-1 -ylcarbonyl; 2- (methylaminocarbonyl)ethenyl; 2-(pyridin-3-ylmethyl)-2H-tetrazol-5-ylmethyl; aminomethyl; carboxyl; methoxycarbonyl; pyridin-2-ylcarbonyl; pyridin-3-ylcarbonyl; and pyridin-4-ylcarbonyl.
135. At least one chemical entity of any one of Claims 131 to 134 wherein R3 is chosen from hydrogen and optionally substituted lower alkyl.
136. At least one chemical entity of Claim 135 wherein R3 is chosen from hydrogen and lower alkyl.
137. At least one chemical entity of Claims 136 wherein R3 is chosen from hydrogen and methyl.
138. At least one chemical entity of Claim 137 wherein R3 is hydrogen.
139. At least one chemical entity of Claim 131 wherein Z2 is -C(O)NR2R5 wherein R2 is cchhoosseenn ffrroomm ooppttiioonnaallllyy ssuubbssttiittuutteedd aallkkyyll aanndd oopptionally substituted cycloalkyl; and R5 is chosen from hydrogen and optionally substituted alkyl.
140. At least one chemical entity of any one of Claims 131 to 139 wherein R4 is chosen from hydrogen, cyano, halo, azido, optionally substituted aminocarbonyl, and optionally substituted alkyl.
141. At least one chemical entity of Claim 140 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, fluoro, azido, optionally substituted alkylaminocarbonyl, and optionally substituted methyl.
142. At least one chemical entity of Claim 141 wherein R4 is chosen from hydrogen, cyano, chloro, bromo, azido, pyridin-3-ylmethylaminocarbonyl, aminomethyl, and hydroxymethyl.
143. At least one chemical entity of Claim 142 wherein R4 is hydrogen.
144. At least one chemical entity of any one of Claims 139 to 143 wherein R5 is chosen from hydrogen and lower alkyl.
145. At least one chemical entity of Claim 144 wherein R5 is chosen from hydrogen and methyl.
146. At least one chemical entity of Claim 145 wherein R5 is hydrogen.
147. At least one chemical entity of any one of Claims 139 to 146 wherein R2 is chosen from optionally substituted methyl, optionally substituted ethyl, optionally substituted propyl, optionally substituted butyl, optionally substituted pentyl, optionally substituted hexyl, optionally substituted cyclopropyl, optionally substituted cyclopentyl, and optionally substituted cyclohexyl where each optionally substituted group is optionally substituted with one, two or three groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
148. At least one chemical entity of Claim 147 wherein R2 is chosen from methyl and ethyl, where the methyl and ethyl groups are optionally substituted with one or two groups selected from optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, optionally substituted aminocarbonyl, optionally substituted amino, hydroxy, carboxyl, optionally substituted alkoxycarbonyl, and optionally substituted alkoxy.
149. At least one chemical entity of Claim 148 wherein R2 is chosen from (1-(2-aminoethyl)- 1 H-pyrazol-3-yl)methyl; (1-(methylsulfonyl)piperidin-3-yl)methyl; (1-acetylpiperidin-3-yl)methyl; (1-acetylpyrrolidin-2-yl)methyl; (1 H-imidazol-2-yl)methyl; (1 H-pyrazol-3-yl)methyl; (1 -methyl-1 H- pyrazol-3-yl)methyl; (1 -methyl-1 H-pyrazol-5-yl)methyl; (2-
(aminocarbonyl)ethylamino)carbonylmethyl; (2-(aminomethyl)pyridin-3-yl)methyl; (2- (carboxy)ethylamino)carbonylmethyl; (2-(dimethylamino)ethylamino)carbonylmethyl; (2- (hydroxy)ethylamino)carbonylmethyl; (2-(methylamino)ethylamino)carbonylmethyl; (2-(N- methyl-N-(t-butoxycarbonyl)-amino)ethylamino)carbonylmethyl; (2-oxopiperidin-3-yl)methyl; (3- (dimethylamino)propylamino)carbonylmethyl; (3-(hydroxy)ethylamino)carbonylmethyl; (3-(t- butoxycarbonylamino)propylamino)carbonylmethyl; (4-(aminomethyl)pyridin-2-yl)methyl; (5- (aminomethyl)pyridin-2-yl)methyl; (6-((1,3-dioxoisoindolin-2-yl)methyl)pyridin-2-yl)methyl; (6-(2- aminoethylamino)pyridin-2-yl)methyl; (6-(3-aminopropylamino)pyridin-2-yl)methyl; (6- (aminomethyl)pyridin-2-yl)methyl; (6-(hydroxymethyl)pyridin-2-yl)methyl; (6-bromopyridin-2- yl)methyl; (methylsulfonamido)carbonylmethyl; (pyridin-2-yl)ethylamino; 1-(2-(tert- butoxycarbonylamino)ethyl)-1 H-pyrazol-3-ylmethyl; 1-(2-(tert-butoxycarbonylamino)ethyl)-1 H- pyrazol-5-ylmethyl; 1-(aminocarbonyl)-2-(amino)-eth-1-yl; 1-(aminocarbonyl)-3-(amino)-propyl; 1 -(aminocarbonyl)-4-(amino)-butyl; 1-(aminocarbonyl)-4-(benzyloxycarbonylamino)-pentyl; 1- (aminocarbonyl)-5-(amino)-pentyl; 1-(aminocarbonyl)eth-1-yl; 1-(carboxy)-2-(amino)-eth-1-yl; 1- (dimethylaminocarbonyl)-2-(amino)-eth-1-yl; 1-(hydroxy)-2-(aminocarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(ethoxycarbonylmethylamino)eth-1-yl; 1-(hydroxy)-2-(pyridin-2-ylmethylamino)eth- 1 -yl; 1 -(methoxycarbonyl)-2-(amino)-eth-1 -yl; 1 -(methoxycarbonyl)-2-(t-butoxycarbonylamino)- eth-1-yl; 1-(methoxycarbonyl)eth-1-yl; 1-(methylaminocarbonyl)-2-(amino)-eth-1-yl (2 occ); 1- (methylaminocarbonyl)eth-i-yl; 1-aminocarbonyl-2-hydroxy-eth-1-yl; 1-methoxycarbonyl-2- hydroxy-eth-1-yl; 2-(2-aminoethoxy)ethyl; 2-(2-aminoethyl)-pyridin-6-ylmethyl; 2-(2- aminoethylamino)ethyl; 2-(3-fluorophenyl)ethyl; 2-(3-methoxycarbonyl)ethyl; 2-(6- (aminomethyl)pyridin-2-yl)ethyl; 2-(acetylamino)ethyl; 2-(amino)ethyl; 2-(aminocarbonyl)-2- (acetylamino)-eth-i-yl; 2-(aminocarbonyl)-2-(amino)-eth-1-yl; 2-(aminocarbonyl)ethyl; 2- (aminomethyl)pyridin-5-ylmethyl; 2-(carboxy)-2-(amino)-eth-1-yl; 2-(dimethylamino)ethyl; 2- (dimethylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(ethoxycarbonyl)ethyl; 2-(methoxycarbonyl)-2- (acetylamino)-eth-i-yl; 2-(methoxycarbonyl)-2-(amino)-eth-1-yl; 2-(methoxycarbonylamino)ethyl; 2-(methylamino)ethyl; 2-(methylaminocarbonyl)-2-(acetylamino)-eth-1-yl; 2- (methylaminocarbonyl)-2-(amino)-eth-1 -yl; 2-(methylsulfonamido)ethyl; 2-(methyoxycarbonyl)-2- (amino)-eth-i-yl; 2-(N-(t-butoxycarboπyl)-N-(methyl)-amino)ethyl; 2-(piperazin-1 -yl)ethyl; 2- (pyrazin-2-yl)ethyl; 2-(pyridin-2-yl)ethyl; 2-(pyridin-3-yl)ethyl; 2-(t- butoxycarbonylaminomethyl)pyridin-3-ylmethyl; 2-(t-butoxycarbonylaminomethyl)pyridin-6- ylmethy!; 2-(trifluoromethyl)-pyridin-6-ylmethyl; 2-aminopyridin-3-ylmethyl; 2-aminopyridin-5- ylmethyt; 2-chloropyridin-5-ylmethyl; 2-cyanopyridin-5-ylmethyl; 2-hydroxy-3-amino-prop-1-yl; 2- hydroxyethyl; 2-methoxyeth-1-yl; 2-methoxypyridin-3-ylmethyl; 2-methoxypyridin-5-ylmethyl; 2- methylpyridin-3-ylmethyl; 2-methylpyridin-5-yl methyl; 2-methylpyridin-6-yImethyl; 3-(2- aminoethyl)cyclohexyl)methyl; 3-(4-methylpiperazin-1 -yl)propyl; 3-(amino)-3- (methylaminocarbonyl)prop-i-yl; 3-(aminocarbonyl)propyl; 3-(aminomethyl)benzyl; 3- (aminomethyl)pyridin-2-ylmethyI; 3-(methoxycarbonyl)propyl; 3-(methylaminocarbonyl)propyl; 3- (trifluoromethyl)-pyridin-2-ylmethyl; 3,4-difluorobenzyl; 3-aminomethylpyridin-4-ylmethyl; 3- aminopropyl; S-carbamoylcyclopentyl; 3-carboxypropyl; 3-hydroxypropyl; 3-methoxy propyl; 4- (aminomethyl)-pyridin-2-ylmethyl; 4-(N,N-dimethylamino)pyridin-3-ylmethyl; 4-(t- butoxycarbonylamino)-morpholinomethyl; 4-aminobutyl; 4-aminomethylpyridin-3-ylmethyl; 4- cyanobenzyl; 4-fluorobenzyl; 4-methylaminopyridin-3-ylmethyl; 4-methylbenzyl; 4- morpholinopyridin-3-ylmethyl; 4-piperazinylpyridin-3-ylmethyl; 5-
((bis(dimethylamino)methylamino)methyl)pyridin-3-ylmethyl; 5-(aminomethyl)pyridin-2-ylmethyl; 5-(aminomethyl)pyridin-3-ylmethyl; 5-(hydroxymethyl)pyridin-3-ylmethyl; 5-(t- butoxycarbonylaminomethyl)-pyridin-2-ylmethyl; 5-(trifluoromethyl)-pyridin-2-ylmethyl; 5- aminopentyl; 5-aminopyridin-2-ylmethyl; 6-((bis(dimethylamino)methyleneamino)methyl)pyridin- 2-yl)methyl; 6-(4-acetylpiperazin-1 -yl)pyridin-3-ylmethyl); 6-(aminomethyl)pyridin-2-yl)methyl; 6- aminohexyl; aminocarbonylmethyl; benzyl; cyclopropylmethyl; dimethylaminocarbonylmethyl; isopropyl; isoxazol-5-ylmethyl; methoxycarbonylmethyl; methyl; methylaminocarbonylmethyl; oxazol-2-ylmethyl; piperidin-4-ylmethyl; prop-2-en-1-yl; pyrazin-2-ylmethyl; pyridin-2-ylmethyl; pyridin-2-ylmethyl -N-oxide; pyridin-3-ylmethyl; pyridin-3-ylmethyl -N-oxide; pyridin-4-ylmethyl; thiophen-2-ylmethyl; and thiophen-3-ylmethyl.
150. At least one chemical entity of any one of Claims 131 to 149 wherein Z3 is chosen from optionally substituted lower alkyl.
151. At least one chemical entity of Claim 150 wherein Z3 is chosen from optionally substituted methyl, optionally substituted ethyl, and optionally substituted propyl wherein the methyl, ethyl and propyl groups are optionally substituted with one or two groups selected from hydroxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted heterocycloalkyl, optionally substituted cycloalkyl, and optionally substituted aryloxy.
152. At least one chemical entity of Claim 151 wherein Z3 is chosen from 2-(3- methylphenyl)ethyl, (tetrahydrofuran-2-yl)methyl, 1-hydroxycyclohexylmethyl, 2-(1 ,3-dioxolan-2- yl)ethyl, 2-(1 H-imidazol-4-yl)ethyl, 2-(1-methyl-1H-imida2ol-4-yl)ethyl, 2-(1-methylpiperidin-4- yl)ethyl, 2-(2,3-difluorophenyl)ethyl, 2-(2,5-difluorophenyl)ethyl, 2-(2,6-difluorophenyl)ethyl, 2-(2- chlorophenyl)ethyl, 2-(2-cyanophenyl)ethyl, 2-(2-fluorophenyl)ethyl, 2-(2-hydroxyphenyl)ethyl, 2- (2-methoxyphenyl)ethyl, 2-(2-methylphenyl)ethyl, 2-(2-oxoimidazolidin-1-yl)ethyl, 2-(2- oxopiperidin-1-yl)ethyl, 2-(2-oxopyrrolidin-1-yl)ethyl, 2-(3-(trifluoromethyl)phenyl)ethyl, 2-(3,4- difluorophenyl)ethyl, 2-(3,5-difluorophenyl)ethyl, 2-(3,5-dim ethyl- 1 H-pyrazol-1-yl)ethyl, 2-(3,5- dimethyl-1H-pyrazol-4-yl)ethyl, 2-(3-carbamoylphenyl)ethyl, 2-(3-carboxyphenyl)ethyl, 2-(3- cyanophenyl)ethyl, 2-(3-fluorophenyl)-2-(hydroxy)-ethyl, 2-(3-fluorophenyl)ethyl, 2-{3- fluoropyridin-2-yl)ethyl, 2-(3-methoxycarbonylphenyl)ethyl, 2-(3-methoxyphenyl)ethyl, 2-(3- methylphenyl)ethyl, 2-(4-aminophenyi)ethyl, 2-(4-chlorophenyl)ethyl, 2-(4-fluorophenyl)ethyl, 2- (4-hydroxyphenyl)ethyl, 2-(benzo[d][1 ,3]dioxol-5-yl)ethyl, 2-(furan-2-yl)ethyl, 2-(hydroxy)-2- (phenyl)ethyl, 2-(phenyl)ethyl, 2-(pyridin-2-yl)ethyl, 2-(pyridin-3-yl)ethyl, 2-(thiophen-2-yl)ethyl, 2-cyclohex-1-enylethyl, 2-cyclohexylethyl, 2-morpholinoethyl, 2-phenoxyethyl, 2-phenylprop-1- yl, 3-(1 H-imidazol-1-yl)prop-1-yl, 3-phenylpropyl, benzyl, chloro, isopentyl, propyl, pyridin-2- ylmethyl, pyridin-3-yl methyl, and thiophen-3-ylmethyl.
153. At least one chemical entity of Claim 152 wherein Z3 is chosen from 2-(3- fluorophenyl)ethyl and 2-(pyridin-2-yl)ethyl.
154. At least one chemical entity of any one of Claims 131 to 153 wherein Z1 is chosen from cyano, halo, optionally substituted acyl, optionally substituted alkenyl, optionally substituted alkoxycarbonyl, optionally substituted alkyl, optionally substituted amino, optionally substituted aminocarbonyl, and optionally substituted carbamimidoyl.
155. At least one chemical entity of Claim 154 wherein Z1 is chosen from acyl, alkenyl, alkoxycarbonyl, alkyl, amino, aminocarbonyl, and carbamimidoyl, where each group is optionally substituted by one or two groups selected from cyano, halo, hydroxy, formyl, optionally substituted alkyl, optionally substituted alkenyi, optionally substituted alkoxy, carboxyl, optionally substituted alkoxycarbonyl, optionally substituted acyl, optionally substituted aminocarbonyl, optionally substituted heterocycloalkyl, optionally substituted heteroaryl, optionally substituted carbamimidoyl, and optionally substituted sulfonyl.
156. At least one chemical entity of Claim 155 wherein Z1 is chosen from (5-oxo- tetrahydrofuran-2-yl)methylamino; (E)-N'-cyano-N,N-dimethylcarbamimidoyl; (Z)-4-(N-(2- hydroxyethyl)-N,N'-bis(methoxycarbonyl)carbamimidoyl); (Z)-but-2-en-2-yl; 1-ethoxyeth-1-yl; 1- ethoxyvinyl;2-hydroxyeth-1-ylamino 2-methoxyeth-1ylamino;2-methylprop-1-enyl; 3- (methoxycarbonyl)-prop-2-en-2-yl; 3-(N,N-dimethylamino)carbonyl-prop-1-ylamino; 3- aminocarbonyl-prop-1-ylamino; 3-hydroxyprop-1-ylamino; 3-methoxy-prop-1-enyl; 3- methoxyprop-1-ylamino; 3-methoxypropyl; 3-methylbut-2-en-2-yl; 4-((pyridin-3- ylmethyl)aminocarbonyl)butylamino; 4-(N,N-dimethylamino)carbonyl-but-1-ylamino; 4-(N- methylamino)carbonyl-but-1-ylamino; 4-aminocarbonyl-but-1-ylamino; acetyl; aminocarbonyl; carbamimidoyl; chloro; cyano; dimethylamino; ethoxycarbonyl; hydrogen; methoxycarbonyl; methylamino; N-(2-hydroxyethyl)-N-(acetyl)-amino; N-(2-hydroxyethyl)-N-(methyl)-amino; N-(2- hydroxyprop-1-yl)-N-(methyl)-amino; N-(methyl)-aminocarbonyI; N-(pyridin-2-ylmethyl)amino; N- (pyridin-3-ylmethyl)amino; N-(pyridin-4-ylmethyl)amino; N,N-dimethylcarbamimidoyl; N- cyanocarbamimidoyl; N-methyl-N-acetylamino; and prop-1-enyl.
157. At least one chemical entity of any one of Claims 131 to 156 wherein R1 is chosen from hydrogen and optionally substituted lower alkyl.
158. At least one chemical entity of Claim 157 wherein R1 is chosen from hydrogen and lower alkyl.
159. At least one chemical entity of Claim 158 wherein R1 is chosen from hydrogen and methyl.
160. At least one chemical entity of Claim 159 wherein R1 is hydrogen.
161. A pharmaceutically acceptable composition comprising a pharmaceutically acceptable carrier and at least one chemical entity of any one of Claims 1 to 160.
162. A pharmaceutical composition of Claim 161 wherein the composition is formulated in a form chosen from tablets, capsules, powders, liquids, suspensions, suppositories and aerosols.
163. A packaged pharmaceutical composition comprising a pharmaceutical composition of Claim 161 or 162 and instructions for using the composition to treat a patient suffering from a disease associated with smooth muscle myosin or non-muscle myosin.
164. The packaged pharmaceutical composition of Claim 163 wherein the disease associated with smooth muscle myosin is selected from hypertension, asthma, chronic obstructive pulmonary disease (copd) asthma, bronchoconstrictive disease, glaucoma and other ocular indications, incontinence and other bladder disfunctions, irritable bowel syndrome, pre-term labor, esophogial dysmotility, strokes, subarachnoid hemmorhages, pre-menstrual cramps, erectile dysfunction and other acute and chronic diseases and conditions associated with smooth muscle myosin and/or non-muscle myosin.
165. A method of treating or ameliorating a disease associated with smooth muscle myosin or non-muscle myosin in a mammal which method comprises administering to a mammal in need thereof a therapeutically effective amount of at least one chemical entity of any one of Claims 1 to 160.
166. A method of Claim 165 wherein said diseases associated with smooth muscle myosin is selected from hypertension, asthma, chronic obstructive pulmonary disease (copd) asthma, bronchoconstrictive disease, glaucoma and other ocular indications, incontinence and other bladder disfunctions, irritable bowel syndrome, pre-term labor, esophogial dysmotility, strokes, subarachnoid hemmorhages, pre-menstrual cramps, erectile dysfunction and other acute and chronic diseases and conditions associated with smooth muscle myosin and/or non-muscle myosin.
Applications Claiming Priority (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US83490106P | 2006-08-01 | 2006-08-01 | |
| US83490406P | 2006-08-01 | 2006-08-01 | |
| US83501006P | 2006-08-01 | 2006-08-01 | |
| US83490306P | 2006-08-01 | 2006-08-01 | |
| US83518306P | 2006-08-01 | 2006-08-01 | |
| US60/835,183 | 2006-08-01 | ||
| US60/834,901 | 2006-08-01 | ||
| US60/835,010 | 2006-08-01 | ||
| US60/834,904 | 2006-08-01 | ||
| US60/834,903 | 2006-08-01 | ||
| US83523606P | 2006-08-02 | 2006-08-02 | |
| US60/835,236 | 2006-08-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008016643A2 true WO2008016643A2 (en) | 2008-02-07 |
| WO2008016643A3 WO2008016643A3 (en) | 2008-06-19 |
Family
ID=38997696
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/017186 Ceased WO2008016643A2 (en) | 2006-08-01 | 2007-07-31 | Certain chemical entities, compositions, and methods |
Country Status (4)
| Country | Link |
|---|---|
| AR (1) | AR062176A1 (en) |
| PE (1) | PE20080432A1 (en) |
| TW (1) | TW200823188A (en) |
| WO (1) | WO2008016643A2 (en) |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009051112A1 (en) * | 2007-10-15 | 2009-04-23 | Takeda Pharmaceutical Company Limited | Amide compounds and use of the same |
| US7919511B2 (en) | 2006-08-01 | 2011-04-05 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| WO2011042797A1 (en) * | 2009-10-08 | 2011-04-14 | Icozen Therapeutics Pvt. Ltd. | Pyrazole derivatives as modulators of calcium release -activated calcium channel |
| US7932270B2 (en) | 2006-08-01 | 2011-04-26 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| US7939548B2 (en) | 2006-08-02 | 2011-05-10 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| US7939534B2 (en) | 2006-08-01 | 2011-05-10 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| JP2013506715A (en) * | 2009-10-06 | 2013-02-28 | ミレニアム ファーマシューティカルズ, インコーポレイテッド | Heterocyclic compounds useful as PDK1 inhibitors |
| US8586751B2 (en) | 2009-06-12 | 2013-11-19 | Bristol-Myers Squibb Company | Nicotinamide compounds useful as kinase modulators |
| WO2015010832A1 (en) * | 2013-07-22 | 2015-01-29 | Syngenta Participations Ag | Microbiocidal heterocyclic derivatives |
| US8993612B2 (en) | 2009-10-08 | 2015-03-31 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel and methods for treatment of non-small cell lung cancer |
| WO2016039448A1 (en) * | 2014-09-12 | 2016-03-17 | 田辺三菱製薬株式会社 | Aromatic carboxylic acid amide compound |
| US9458110B2 (en) | 2013-02-28 | 2016-10-04 | Bristol-Myers Squibb Company | Phenylpyrazole derivatives as potent ROCK1 and ROCK2 inhibitors |
| US9828345B2 (en) | 2013-02-28 | 2017-11-28 | Bristol-Myers Squibb Company | Phenylpyrazole derivatives as potent ROCK1 and ROCK2 inhibitors |
| US9951069B1 (en) | 2017-01-11 | 2018-04-24 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10336762B2 (en) | 2017-02-16 | 2019-07-02 | Gilead Sciences, Inc. | Pyrrolo[1,2-b]pyridazine derivatives |
| US10421756B2 (en) | 2015-07-06 | 2019-09-24 | Rodin Therapeutics, Inc. | Heterobicyclic N-aminophenyl-amides as inhibitors of histone deacetylase |
| CN111393376A (en) * | 2020-05-11 | 2020-07-10 | 安徽赛迪生物科技有限公司 | A kind of synthetic method of 2-chloropyrimidine-4-carboxylic acid |
| US10875866B2 (en) | 2018-07-13 | 2020-12-29 | Gilead Sciences, Inc. | Pyrrolo[1,2-B]pyridazine derivatives |
| US10919902B2 (en) | 2015-07-06 | 2021-02-16 | Alkermes, Inc. | Hetero-halo inhibitors of histone deacetylase |
| WO2021092322A1 (en) * | 2019-11-06 | 2021-05-14 | Remedy Plan, Inc. | Cancer treatments targeting cancer stem cells |
| EP3868749A1 (en) | 2016-03-23 | 2021-08-25 | Syngenta Participations Ag | Herbicidal compounds |
| US11225475B2 (en) | 2017-08-07 | 2022-01-18 | Alkermes, Inc. | Substituted pyridines as inhibitors of histone deacetylase |
| US12522587B2 (en) | 2018-05-04 | 2026-01-13 | Remedy Plan, Inc. | Cancer treatments targeting cancer stem cells |
| US12612368B2 (en) | 2020-11-06 | 2026-04-28 | Remedy Plan, Inc. | Cancer treatments targeting cancer stem cells |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4140853A (en) * | 1975-04-18 | 1979-02-20 | Schering Aktiengesellschaft | Process for the preparation of aminopyridines |
| WO2005108358A2 (en) * | 2004-04-22 | 2005-11-17 | Eli Lilly And Company | Pyrrolidine derivatives useful as bace inhibitors |
-
2007
- 2007-07-31 WO PCT/US2007/017186 patent/WO2008016643A2/en not_active Ceased
- 2007-08-01 PE PE2007000999A patent/PE20080432A1/en not_active Application Discontinuation
- 2007-08-01 TW TW096128145A patent/TW200823188A/en unknown
- 2007-08-01 AR ARP070103397A patent/AR062176A1/en unknown
Cited By (54)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7919511B2 (en) | 2006-08-01 | 2011-04-05 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| US7932270B2 (en) | 2006-08-01 | 2011-04-26 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| US7939534B2 (en) | 2006-08-01 | 2011-05-10 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| US7939548B2 (en) | 2006-08-02 | 2011-05-10 | Cytokinetics, Inc. | Certain chemical entities, compositions, and methods |
| WO2009051112A1 (en) * | 2007-10-15 | 2009-04-23 | Takeda Pharmaceutical Company Limited | Amide compounds and use of the same |
| US8329691B2 (en) | 2007-10-15 | 2012-12-11 | Takeda Pharmaceutical Company Limited | Amide compounds and use of the same |
| US8586751B2 (en) | 2009-06-12 | 2013-11-19 | Bristol-Myers Squibb Company | Nicotinamide compounds useful as kinase modulators |
| US10030016B2 (en) | 2009-10-06 | 2018-07-24 | Sunesis Pharmaceuticals, Inc. | Heterocyclic compounds useful as PDK1 inhibitors |
| US9546165B2 (en) | 2009-10-06 | 2017-01-17 | Sunesis Pharmaceuticals, Inc. | Heterocyclic compounds useful as PDK1 inhibitors |
| JP2013506715A (en) * | 2009-10-06 | 2013-02-28 | ミレニアム ファーマシューティカルズ, インコーポレイテッド | Heterocyclic compounds useful as PDK1 inhibitors |
| US10246450B2 (en) | 2009-10-08 | 2019-04-02 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel |
| US10174034B2 (en) | 2009-10-08 | 2019-01-08 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel and methods for treatment of non-small cell lung cancer |
| US10668051B2 (en) | 2009-10-08 | 2020-06-02 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel |
| US8993612B2 (en) | 2009-10-08 | 2015-03-31 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel and methods for treatment of non-small cell lung cancer |
| CN104788430A (en) * | 2009-10-08 | 2015-07-22 | 理森制药股份公司 | Pyrazoles derivatives modulators of calcium release-activated calcium channel and methods for treatment of non-small cell lung cancer |
| CN110627724A (en) * | 2009-10-08 | 2019-12-31 | 理森制药股份公司 | Pyrazole derivative calcium release activated calcium channel modulator and treatment method for non-small cell lung cancer |
| CN102834382B (en) * | 2009-10-08 | 2016-08-03 | 理森制药股份公司 | The pyrazole derivatives of calcium channel modulators is activated as calcium release |
| CN104788430B (en) * | 2009-10-08 | 2019-10-22 | 理森制药股份公司 | Pyrazole derivative calcium release activated calcium channel modulator and treatment method for non-small cell lung cancer |
| EA024630B1 (en) * | 2009-10-08 | 2016-10-31 | Ризен Фармасьютикалз С.А. | Pyrazole derivatives as calcium release-activated calcium (crac) channel inhibitors |
| US8377970B2 (en) | 2009-10-08 | 2013-02-19 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel |
| US9758514B2 (en) | 2009-10-08 | 2017-09-12 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel |
| US8921364B2 (en) | 2009-10-08 | 2014-12-30 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel |
| US9944631B2 (en) | 2009-10-08 | 2018-04-17 | Rhizen Pharmaceuticals Sa | Modulators of calcium release-activated calcium channel |
| WO2011042797A1 (en) * | 2009-10-08 | 2011-04-14 | Icozen Therapeutics Pvt. Ltd. | Pyrazole derivatives as modulators of calcium release -activated calcium channel |
| CN102834382A (en) * | 2009-10-08 | 2012-12-19 | 理森制药股份公司 | Pyrazole derivatives as modulators of calcium release-activated calcium channel |
| US9828345B2 (en) | 2013-02-28 | 2017-11-28 | Bristol-Myers Squibb Company | Phenylpyrazole derivatives as potent ROCK1 and ROCK2 inhibitors |
| US9458110B2 (en) | 2013-02-28 | 2016-10-04 | Bristol-Myers Squibb Company | Phenylpyrazole derivatives as potent ROCK1 and ROCK2 inhibitors |
| WO2015010832A1 (en) * | 2013-07-22 | 2015-01-29 | Syngenta Participations Ag | Microbiocidal heterocyclic derivatives |
| WO2016039448A1 (en) * | 2014-09-12 | 2016-03-17 | 田辺三菱製薬株式会社 | Aromatic carboxylic acid amide compound |
| US10919902B2 (en) | 2015-07-06 | 2021-02-16 | Alkermes, Inc. | Hetero-halo inhibitors of histone deacetylase |
| US11858939B2 (en) | 2015-07-06 | 2024-01-02 | Alkermes, Inc. | Hetero-halo inhibitors of histone deacetylase |
| US10421756B2 (en) | 2015-07-06 | 2019-09-24 | Rodin Therapeutics, Inc. | Heterobicyclic N-aminophenyl-amides as inhibitors of histone deacetylase |
| EP3868749A1 (en) | 2016-03-23 | 2021-08-25 | Syngenta Participations Ag | Herbicidal compounds |
| US10696673B2 (en) | 2017-01-11 | 2020-06-30 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US11987580B2 (en) | 2017-01-11 | 2024-05-21 | Alkermes, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10793567B2 (en) | 2017-01-11 | 2020-10-06 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US11286256B2 (en) | 2017-01-11 | 2022-03-29 | Alkermes, Inc. | Bicyclic inhibitors of histone deacetylase |
| US9951069B1 (en) | 2017-01-11 | 2018-04-24 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US11225479B2 (en) | 2017-01-11 | 2022-01-18 | Alkermes, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10519149B2 (en) | 2017-01-11 | 2019-12-31 | Rodin Therapeutics, Inc. | Bicyclic inhibitors of histone deacetylase |
| US10336762B2 (en) | 2017-02-16 | 2019-07-02 | Gilead Sciences, Inc. | Pyrrolo[1,2-b]pyridazine derivatives |
| US11225475B2 (en) | 2017-08-07 | 2022-01-18 | Alkermes, Inc. | Substituted pyridines as inhibitors of histone deacetylase |
| US11912702B2 (en) | 2017-08-07 | 2024-02-27 | Alkermes, Inc. | Substituted pyridines as inhibitors of histone deacetylase |
| US12522587B2 (en) | 2018-05-04 | 2026-01-13 | Remedy Plan, Inc. | Cancer treatments targeting cancer stem cells |
| US12415808B2 (en) | 2018-07-13 | 2025-09-16 | Gilead Sciences, Inc. | Pyrrolo[1,2-b]pyridazine derivatives |
| US10875866B2 (en) | 2018-07-13 | 2020-12-29 | Gilead Sciences, Inc. | Pyrrolo[1,2-B]pyridazine derivatives |
| US11535622B2 (en) | 2018-07-13 | 2022-12-27 | Gilead Sciences, Inc. | Pyrrolo[1,2-b] pyridazine derivatives |
| WO2021092322A1 (en) * | 2019-11-06 | 2021-05-14 | Remedy Plan, Inc. | Cancer treatments targeting cancer stem cells |
| CN114929672A (en) * | 2019-11-06 | 2022-08-19 | 治疗方案股份有限公司 | Cancer therapy targeting cancer stem cells |
| JP2022553865A (en) * | 2019-11-06 | 2022-12-26 | リメディー プラン,インコーポレーテッド | Cancer therapy targeting cancer stem cells |
| CN114929672B (en) * | 2019-11-06 | 2025-01-21 | 治疗方案股份有限公司 | Cancer therapy targeting cancer stem cells |
| CN111393376B (en) * | 2020-05-11 | 2022-05-13 | 安徽赛迪生物科技有限公司 | Synthetic method of 2-chloropyrimidine-4-formic acid |
| CN111393376A (en) * | 2020-05-11 | 2020-07-10 | 安徽赛迪生物科技有限公司 | A kind of synthetic method of 2-chloropyrimidine-4-carboxylic acid |
| US12612368B2 (en) | 2020-11-06 | 2026-04-28 | Remedy Plan, Inc. | Cancer treatments targeting cancer stem cells |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2008016643A3 (en) | 2008-06-19 |
| PE20080432A1 (en) | 2008-05-29 |
| AR062176A1 (en) | 2008-10-22 |
| TW200823188A (en) | 2008-06-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2008016643A2 (en) | Certain chemical entities, compositions, and methods | |
| EP0783500B1 (en) | Aminoheterocyclic derivatives as antithrombotic or anticoagulant agents | |
| AU2006282456B2 (en) | Novel pyridine derivative and pyrimidine derivative (3) | |
| JP6759100B2 (en) | Therapeutic inhibitor compound | |
| US7939534B2 (en) | Certain chemical entities, compositions, and methods | |
| CN102227409B (en) | Pyridine-3-carboxyamide derivative | |
| EP1831198B1 (en) | Pyridine carboxamide derivatives for use as anticancer agents | |
| CA2736955C (en) | Multiheteroaryl compounds as inhibitors of h-pgds and their use for treating prostaglandin d2 mediated diseases | |
| KR101546595B1 (en) | Heteroarylmethyl amides | |
| TW200306180A (en) | New compounds | |
| NZ576501A (en) | Pyridine carboxamides as 11-beta-hsd1 inhibitors | |
| EP2059516A1 (en) | Pyridine compounds for treating gpr119 related disorders | |
| AU2010291212A1 (en) | Heteroaryl compounds as kinase inhibitors | |
| MX2013000575A (en) | Ampk-activating heterocyclic compounds and methods for using the same. | |
| WO2005063743A1 (en) | Thiazole derivative | |
| EP2195328A1 (en) | Certain chemical entities, compositions, and methods | |
| US7939548B2 (en) | Certain chemical entities, compositions, and methods | |
| JP2012521396A (en) | Nicotinamide derivatives, their preparation and their therapeutic use as anticancer drugs | |
| US7932270B2 (en) | Certain chemical entities, compositions, and methods | |
| JP7386576B2 (en) | N-(1H-imidazol-2-yl)benzamide compounds and pharmaceutical compositions containing the compounds as active ingredients | |
| CA2551604A1 (en) | Amidopyrazole derivative | |
| US7919511B2 (en) | Certain chemical entities, compositions, and methods | |
| JP2009501216A (en) | New pyridine analogues | |
| WO2008016676A2 (en) | Certain chemical entities, compositions, and methods | |
| US20090198057A1 (en) | Certain Chemical Entities, Compositions, and Methods |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07836402 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 07836402 Country of ref document: EP Kind code of ref document: A2 |