WO2008015266A1 - Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments - Google Patents
Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments Download PDFInfo
- Publication number
- WO2008015266A1 WO2008015266A1 PCT/EP2007/058047 EP2007058047W WO2008015266A1 WO 2008015266 A1 WO2008015266 A1 WO 2008015266A1 EP 2007058047 W EP2007058047 W EP 2007058047W WO 2008015266 A1 WO2008015266 A1 WO 2008015266A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mono
- unsubstituted
- substituted
- butyl
- group
- Prior art date
Links
- 0 C*(C)[n]1nccn1 Chemical compound C*(C)[n]1nccn1 0.000 description 6
- UWRLOVDEFTUCHR-IQNGYNMSSA-N CC(C)([C@@H]12)[C@@H](CCN3CCCCC3)C1C2OCc1ccccc1 Chemical compound CC(C)([C@@H]12)[C@@H](CCN3CCCCC3)C1C2OCc1ccccc1 UWRLOVDEFTUCHR-IQNGYNMSSA-N 0.000 description 1
- UDTNUZWGLSDRDS-IQNGYNMSSA-N CC(C)([C@@H]12)[C@@H](CCOCc3ccccc3)C1C2N1CCN(C)CC1 Chemical compound CC(C)([C@@H]12)[C@@H](CCOCc3ccccc3)C1C2N1CCN(C)CC1 UDTNUZWGLSDRDS-IQNGYNMSSA-N 0.000 description 1
- ARKUPRJHFQCYPM-BMHOQUIDSA-N CC(C)([C@@H]12)[C@@H](COCc3ccccc3)C1C2N1CCN(C)CC1 Chemical compound CC(C)([C@@H]12)[C@@H](COCc3ccccc3)C1C2N1CCN(C)CC1 ARKUPRJHFQCYPM-BMHOQUIDSA-N 0.000 description 1
- NIBMRNSSRNZRJZ-IRXDYDNUSA-N CC1(C)[C@H](CN2CCOCC2)C[C@@H]1CC[n](cc1)c(CCC2)c1C2=O Chemical compound CC1(C)[C@H](CN2CCOCC2)C[C@@H]1CC[n](cc1)c(CCC2)c1C2=O NIBMRNSSRNZRJZ-IRXDYDNUSA-N 0.000 description 1
- APOJIILNJVLCQE-UHFFFAOYSA-N CC1OC(C)CN(C)C1 Chemical compound CC1OC(C)CN(C)C1 APOJIILNJVLCQE-UHFFFAOYSA-N 0.000 description 1
- BDMFEFZOOOYCKN-UHFFFAOYSA-N CCC[n]1nccc1 Chemical compound CCC[n]1nccc1 BDMFEFZOOOYCKN-UHFFFAOYSA-N 0.000 description 1
- WQDDXVGJRSTLED-UHFFFAOYSA-N CN(CC1)CCN1c1ccccc1 Chemical compound CN(CC1)CCN1c1ccccc1 WQDDXVGJRSTLED-UHFFFAOYSA-N 0.000 description 1
- KDTVWEHAAISPNW-UHFFFAOYSA-N CN1CCSCC1 Chemical compound CN1CCSCC1 KDTVWEHAAISPNW-UHFFFAOYSA-N 0.000 description 1
- MWZDIEIXRBWPLG-UHFFFAOYSA-N C[n]1ncnc1 Chemical compound C[n]1ncnc1 MWZDIEIXRBWPLG-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/67—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of saturated acids
- C07C69/675—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of saturated acids of saturated hydroxy-carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/12—Antidiarrhoeals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/06—Antimigraine agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
- A61P25/36—Opioid-abuse
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/06—Antiarrhythmics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C215/00—Compounds containing amino and hydroxy groups bound to the same carbon skeleton
- C07C215/02—Compounds containing amino and hydroxy groups bound to the same carbon skeleton having hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C215/04—Compounds containing amino and hydroxy groups bound to the same carbon skeleton having hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being saturated
- C07C215/20—Compounds containing amino and hydroxy groups bound to the same carbon skeleton having hydroxy groups and amino groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being saturated the carbon skeleton being saturated and containing rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/08—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms
- C07D211/10—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with radicals containing only carbon and hydrogen atoms attached to ring carbon atoms
- C07D211/14—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with radicals containing only carbon and hydrogen atoms attached to ring carbon atoms with hydrocarbon or substituted hydrocarbon radicals attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/08—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms
- C07D295/096—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms with the ring nitrogen atoms and the oxygen or sulfur atoms separated by carbocyclic rings or by carbon chains interrupted by carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/10—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by doubly bound oxygen or sulphur atoms
- C07D295/112—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by doubly bound oxygen or sulphur atoms with the ring nitrogen atoms and the doubly bound oxygen or sulfur atoms separated by carbocyclic rings or by carbon chains interrupted by carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/12—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly or doubly bound nitrogen atoms
- C07D295/135—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly or doubly bound nitrogen atoms with the ring nitrogen atoms and the substituent nitrogen atoms separated by carbocyclic rings or by carbon chains interrupted by carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/04—Systems containing only non-condensed rings with a four-membered ring
Definitions
- the present invention relates to substituted dimethylcyclobutyl compounds of general formula I,
- medicaments comprising said substituted dimethylcyclobutyl compounds as well as the use of said substituted dimethylcyclobutyl compounds for the preparation of medicaments, which are particularly suitable for the prophylaxis and/or treatment of disorders or diseases that are at least partially mediated via sigma receptors.
- sigma receptor ligands may be useful in the treatment of psychosis and movement disorders such as dystonia and tardive dyskinesia, and motor disturbances associated with Huntington's chorea or Tourette's syndrome and in Parkinson's disease (Walker, J. M. et al, Pharmacological Reviews, 1990, 42, 355). It has been reported that the known sigma receptor ligand rimcazole clinically shows effects in the treatment of psychosis (Snyder, S. H., Largent, B. L. J. Neuropsychiatry 1989, 1 , 7). The sigma binding sites have preferential affinity for the dextrorotatory isomers of certain opiate benzomorphans, such as (+)SKF 10047, (+)cyclazocine, and
- (+)pentazocine and also for some narcoleptics such as halopehdol.
- the sigma receptor has at least two subtypes, which may be discriminated by stereoselective isomers of these pharmacoactive drugs. Possible sigma- site-mediated drug effects include modulation of glutamate receptor function, neurotransmitter response, neuroprotection, behaviour, and cognition (Quirion, R. et al. Trends Pharmacol. Sci., 1992, 13:85-86). Most studies have implied that sigma binding sites (receptors) are plasmalemmal elements of the signal transduction cascade. Drugs reported to be selective sigma ligands have been evaluated as antipsychotics (Hanner, M. et al. Proc. Natl.
- an object of the present invention was to provide compounds that are particularly suitable as active ingredients in medicaments, especially in medicaments for the prophylaxis and/or treatment of disorders or diseases related to sigma receptors, preferably sigma-1 receptors, such as food intake related disorders or pain.
- the substituted dimethylcyclobutyl compounds of general formula I given below show good to excellent affinity for sigma receptors, in particular they show good to excellent affinity to sigma-1 receptors.
- These compounds are therefore particularly suitable as pharmacologically active agents in a medicament for the prophylaxis and/or treatment of disorders or diseases related to sigma receptors, preferably related to sigma-1 receptors.
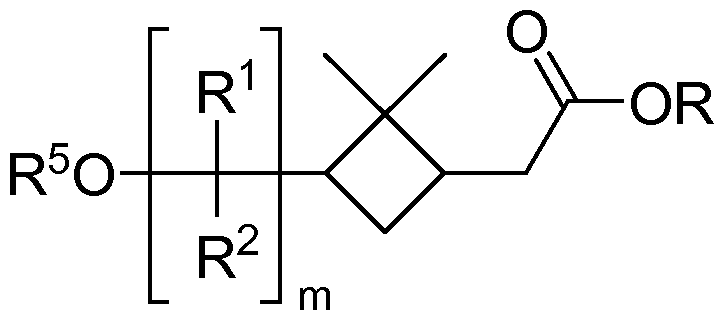
- the present invention relates to a dimethylcyclobutyl compound of general formula I,
- n 1 , 2, 3 or 4;
- X represents a -OR moiety or a -NR 6iR-)7 moiety
- an unsubstituted or at least mono-substituted aryl or heteroaryl radical which may be bonded via a linear or branched, unsubstituted or at least- mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated or unsaturated, but not aromatic, mono- or bicyclic ring system;
- an unsubstituted or at least mono-substituted aryl or heteroaryl radical which may be bonded via a linear or branched, unsubstituted or at least- mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated or unsaturated, but not aromatic, mono- or bicyclic ring system;
- R 1 represents a hydrogen atom or a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical
- R 2 represents a hydrogen atom
- a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical a saturated or unsaturated, unsubstituted or at least mono-substituted, optionally at least one heteroatom as a ring member containing cycloaliphatic radical, which may be bonded via a linear or branched, unsubstituted or at least-mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system;
- an unsubstituted or at least mono-substituted aryl or heteroaryl radical which may be bonded via a linear or branched, unsubstituted or at least- mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated or unsaturated, but not aromatic, mono- or bicyclic ring system;
- R 3 and R 4 independently of one another, each represent a hydrogen atom or a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical;
- R 5 and R 8 independently of one another, each represent a hydrogen atom or a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical;
- an unsubstituted or at least mono-substituted aryl or heteroaryl radical which may be bonded via a linear or branched, unsubstituted or at least-mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated or unsaturated, but not aromatic, mono- or bicyclic ring system;
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a hydrogen atom or a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical;
- an unsubstituted or at least mono-substituted aryl or heteroaryl radical which may be bonded via a linear or branched, unsubstituted or at least- mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated or unsaturated, but not aromatic, mono- or bicyclic ring system;
- R 6 and R 7 together with the bridging nitrogen atom form an unsubstituted or at least mono-substituted, saturated, unsaturated or aromatic heterocyclic ring which may contain one or more additional heteroatom(s) as ring member(s) and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system;
- R 9 and R 10 together with the bridging nitrogen atom form an unsubstituted or at least mono-substituted, saturated, unsaturated or aromatic heterocyclic ring which may contain one or more additional heteroatom(s) as ring member(s) and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system;
- R 11 and R 12 independently of one another, represent a hydrogen atom or a linear or branched, saturated or unsaturated, unsubstituted or at least mono- substituted aliphatic radical;
- an unsubstituted or at least mono-substituted aryl or heteroaryl radical which may be bonded via a linear or branched, unsubstituted or at least- mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated or unsaturated, but not aromatic, mono- or bicyclic ring system;
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- X and Y are different; wherein if R 5 represents hydrogen and m is 1 , R 1 represents a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical; R 2 represents a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical; a saturated or unsaturated, unsubstituted or at least mono-substituted, optionally at least one heteroatom as a ring member containing cycloaliphatic radical, which may be bonded via a linear or branched, unsubstituted or at least-mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system; or an unsubstituted or at least mono-substituted aryl
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a linear or branched, saturated or unsaturated, unsubstituted or at least mono- substituted aliphatic radical;
- R 6 and R 7 together with the bridging nitrogen atom form an unsubstituted or at least mono-substituted, saturated, unsaturated or aromatic heterocyclic ring which may contain one or more additional heteroatom(s) as ring member(s) and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system;
- R 9 and R 10 together with the bridging nitrogen atom form an unsubstituted or at least mono-substituted, saturated, unsaturated or aromatic heterocyclic ring which may contain one or more additional heteroatom(s) as ring member(s) and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system;
- R 11 represents a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical
- R 12 represents a hydrogen atom or a linear or branched, saturated or unsaturated, unsubstituted or at least mono-substituted aliphatic radical; a saturated or unsaturated, unsubstituted or at least mono-substituted, optionally at least one heteroatom as a ring member containing cycloaliphatic radical, which may be bonded via a linear or branched, unsubstituted or at least-mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an unsubstituted or at least mono-substituted saturated, unsaturated or aromatic mono- or bicyclic ring system; or an unsubstituted or at least mono-substituted aryl or heteroaryl radical, which may be bonded via a linear or branched, unsubstituted or at least- mono substituted alkylene, alkenylene or alkinylene group and which may be condensed with an
- any of the substituents in any of the above defined formulae represents or comprises a 3-, A-, 5-, 6-, 7-, 8-, 9-, 10-, 11 -, 12-, 13- or 14-membered heterocyclic ring, 3- to 9-membered (hetero)cycloaliphatic radical, C 3- g cycloalkyl radical or C 4-9 cycloalkenyl radical, said heterocyclic ring, (hetero)cycloaliphatic radical, C 3- g cycloalkyl radical or C 4- 9 cycloalkenyl radical may - if not defined otherwise - be unsubstituted or substituted by one or more substituents, preferably unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s).
- any of the substituents in any of the above defined formulae represents or comprises a cycloaliphatic radical, C 3- 9 cycloalkyl radical or C 4- 9 cycloalkenyl radical which contains one or more, preferably 1 , 2 or 3 heteroatom(s) as ring member(s), unless defined otherwise, each of these heteroatom(s) may preferably be selected independently from the group consisting of N, O and S.
- any of the substituents in any of the above defined formulae represents or comprises a heterocyclic ring which contains one or more, preferably 1 , 2 or
- each of these heteroatom(s) may preferably be selected independently from the group consisting of N, O and S.
- Suitable saturated or unsaturated, optionally at least one heteroatom as ring member containing cycloaliphatic radicals, Cs-g cycloalkyl radicals or C 4- g cycloalkenyl radicals may preferably be selected from the group consisting of cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclopentenyl, cyclohexenyl, cycloheptenyl, cyclooctenyl, pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, morpholinyl, aziridinyl, azetidinyl, imidazolidinyl, thiomorpholinyl, pyrazolidinyl, tetrahydrofuranyl, tetrahydrothiophenyl, azepanyl
- Suitable saturated or unsaturated, optionally at least one heteroatom as ring member containing cycloaliphatic radicals, C 3- 9 cycloalkyl radicals or C 4-9 cycloalkenyl radicals which are condensed with an unsubstituted or at least mono-substituted mono- or bicyclic ring system may preferably be selected from the group consisting of indolinyl, isoindolinyl, decahydronaphthyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)-tetrahydroisoquinolinyl, (1 ,2,3,4)- tetrahydronaphthyl, octahydro-cyclopenta[c]pyrrolyl, (1 , 3,4,7, 9a)-hexahydro- 2H-quinolizinyl, (1 ,2,3,5,6,8a)-hexahydro-indolizinyl, decahydro
- Suitable saturated, unsaturated or aromatic heterocyclic rings may preferably be selected from the group consisting of pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, morpholinyl, aziridinyl, azetidinyl, imidazolidinyl, thiomorpholinyl, pyrazolidinyl, azepanyl, diazepanyl, azocanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyhdinyl, (1 ,2,5,6)-tetrahydropyhdinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyhmidinyl, pyrrolyl, imidazolyl, pyrazolyl and triazolyl.
- Suitable saturated, unsaturated or aromatic heterocyclic rings which are condensed with an unsubstituted or at least mono-substituted mono- or bicyclic ring system may preferably be selected from the group consisting of indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)- tetrahydroisoquinolinyl, octahydro-cyclopenta[c]pyrrolyl, decahydroquinolinyl, dodecahydrocarbazolyl, 9H-carbazolyl, decahydroisoquinolinyl, (2,3)-dihydro- 1 H-cyclopenta[b]indolyl, [1 ,2,3,4]-tetrahydroquinazolinyl, [3,4]-dihydro-2H- benzo[1 ,4]oxazinyl and (4,5,6,7)-tetra
- any of the substituents in any of the above defined formulae represents an alkylene group, preferably an Ci -6 alkylene group, an alkenylene group, preferably an C 2- 6 alkenylene group or an alkinylene group, preferably an C 2- 6 alkinylene group, which may be substituted, said alkylene group, C 2- 6 alkylene group, alkenylene group, C 2- 6 alkenylene group, alkinylene group or C 2- 6 alkinylene group may be unsubstituted or substituted by one or more substituents, preferably unsubstituted or optionally substituted with 1 , 2 or 3 substituent(s).
- Said substituent(s) may preferably be selected independently from the group consisting of -O-Ci -5 -alkyl, -S-Ci -5 -alkyl, -F, Cl, Br, I, -CN, - CF 3 , -OCF 3 , -SCF 3 , -OH, -SH, -NH 2 , -NH(Ci -5 -alkyl) and -N(Ci -5 -alkyl) 2 , whereby in each occurrence Ci -5 -alkyl may be linear or branched.
- An alkenylene group comprises at least one carbon-carbon double bond
- an alkinylene group comprises at least one carbon-carbon triple bond.
- any of the substituents in any of the above defined formulae represents or comprises an aryl radical, including a 6-membered aryl radical such as phenyl or a 10-membered aryl radical such as naphthyl or a 14-membered aryl radical such as anthracenyl, said aryl radical may - if not defined otherwise - be unsubstituted or substituted by one or more substituents, preferably unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituent(s).
- Preferred aryl radicals which may optionally be at least mono-substituted, are phenyl and naphthyl.
- any of the substituents in any of the above defined formulae represents or comprises a heteroaryl radical, including a monocyclic 5- or 6-membered heteroaryl radical or a bi- or tricyclic 8-, 9-, 10-, 11 -, 12-, 13- or 14- membered heteroaryl radical
- said heteroaryl radical may - if not defined otherwise - be unsubstituted or substituted by one or more substituents, preferably unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituent(s).
- substituents may be selected independently from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, sec- butyl, isobutyl, n-pentyl, -O-CH 3 , -0-C 2 H 5 , -0-CH 2 -CH 2 -CH 3 , -O-CH(CH 3 ) 2 , - O-C(CH 3 ) 3 , -S-CH 3 , -S-C 2 H 5 , -S-CH 2 -CH 2 -CH 3 , -S-CH(CH 3 ) 2 , -S-C(CH 3 ) 3 , -
- heteroatom(s), which are present as ring member(s) in the heteroaryl radical may, unless defined otherwise, independently be selected from the group consisting of nitrogen, oxygen and sulphur.
- the heteroaryl radical comprises 1 , 2, 3 or 4 heteroatom(s).
- Suitable bi- or tricyclic heteroaryl radicals may preferably be selected from the group consisting of indolyl, isoindolyl, quinolinyl, isoquinolinyl, benzo[b]furanyl, benzo[b]thiophenyl, benzo[2,1 ,3]thiadiazolyl, [1 ,2,3]-benzothiadiazolyl, [2,1 ,3]-benzoxadiazolyl, [1 ,2,3]-benzoxadiazolyl, benzoxazolyl, benzothiazolyl, benzisoxazolyl, benzisothiazolyl, imidazo[2,1-b]thiazolyl, 2H- chromenyl, indazolyl and quinazolinyl.
- Suitable mono-, bi- or tricyclic heteroaryl radicals which are condensed with an unsubstituted or at least mono-substituted saturated or unsaturated mono- or bicyclic ring system, may preferably be selected from the group consisting of (4,5,6,7)-tetrahydro-1 H-indolyl, [1 ,3]-benzodioxolyl, [1 ,4]-benzodioxanyl, [1 ,2,3,4]-tetrahydronaphthyl, (2,3)-dihydro-1 H-cyclopenta[b]indolyl, [1 ,2,3,4]- tetrahydroquinolinyl, [1 ,2,3,4]-tetrahydroisoquinolinyl, [1 ,2,3,4]- tetrahydroquinazolinyl and [3,4]-dihydro-2H-benzo[1 ,4]oxaziny
- Suitable monocyclic heteroaryl radicals may preferably be selected from the group consisting of pyridinyl, furyl (furanyl), thienyl (thiophenyl), pyrrolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, imidazolyl, pyrazolyl, oxadiazolyl, thiadiazolyl, thazolyl, pyhdazinyl, pyhmidinyl, pyrazinyl and pyranyl.
- a mono- or bicyclic ring system according to the present invention - if not defined otherwise - means a mono- or bicyclic hydrocarbon ring system that may be saturated, unsaturated or aromatic. Each of its different rings may show a different degree of saturation, i.e. it may be saturated, unsaturated or aromatic.
- each of the rings of the mono- or bicyclic ring system may contain one or more, preferably 1 , 2 or 3, heteroatom(s) as ring member(s), which may be identical or different and which can preferably be selected from the group consisting of N, O and S.
- the rings of the mono- or bicyclic ring system are preferably 5-, 6- or 7-membered.
- a mono-or bicyclic ring system according to the present invention is a phenyl or naphthyl ring system.
- condensed means that a ring or ring system is attached to another ring or ring system, whereby the terms “annulated” or “annelated” are also used by those skilled in the art to designate this kind of attachment.
- Such a mono- or bicyclic ring system may - if not defined otherwise - be unsubstituted or substituted by one or more substituents, preferably unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituent(s).
- cyclic substituents may be unsubstituted or substituted by 1 , 2 or 3 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, methoxy, ethoxy, F, Cl, Br, -CN, -CF 3 , -OCF 3 , -SCF 3 , -OH, -SH, -NH 2 and -
- any of the substituents in any of the above defined formulae represents a saturated or unsaturated aliphatic radical, i.e. an alkyl radical, preferably an Ci-io alkyl radical; an alkenyl radical, preferably an C 2- - I0 alkenyl radical or an alkinyl radical, preferably an C2-10 alkinyl radical; said aliphatic radical may - if not defined otherwise - be unsubstituted or substituted by one or more substituents, preferably unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituent(s).
- an alkyl radical preferably an Ci-io alkyl radical
- an alkenyl radical preferably an C 2- - I0 alkenyl radical or an alkinyl radical, preferably an C2-10 alkinyl radical
- said aliphatic radical may - if not defined otherwise - be unsubstituted or substituted by one or more substituents, preferably unsubstituted
- Said substituent(s) may preferably be selected independently from the group consisting of -O-Ci -5 -alkyl, -S-Ci -5 -alkyl, F, Cl, Br, I, -CN, -CF 3 ,
- Ci -5 -alkyl may be linear or branched.
- said substituent(s) may preferably be selected independently from the group consisting Of -O-CH 3 , -0-C 2 H 5 , -0-CH 2 -CH 2 -CH 3 , -O-CH(CH 3 ) 2 , -O-C(CH 3 ) 3 , - S-CH 3 , -S-C 2 H 5 , -S-CH 2 -CH 2 -CH 3 , -S-CH(CH 3 ) 2 , -S-C(CH 3 ) 3 , F, Cl, Br, I, -CN,
- -CF 3 -OCF 3 , -SCF 3 , -OH, -SH, -NH 2 , NH-CH 3 , -NH-C 2 H 5 , -NH-CH 2 -CH 2 -CH 3 , -NH-CH(CHs) 2 , -NH-C(CHs) 3 , -N(CH 3 ) 2 , -N(C 2 H 5 ) 2 .
- An alkenyl radical comprises at least one carbon-carbon double bond
- an alkinyl radical comprises at least one carbon-carbon triple bond
- Suitable alkyl radicals which may be substituted by one or more substituents, may preferably be selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, neo- pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl and n-decyl.
- Suitable alkenyl radicals which may be substituted by one or more substituents, may preferably be selected from the group consisting of vinyl, 1- propenyl, 2-propenyl, 1 -butenyl, 2-butenyl and 3-butenyl.
- Suitable alkinyl radicals which may be substituted by one or more substituents, may preferably be selected from the group consisting of ethinyl, 1 -propinyl, 2-propinyl, 1 -butinyl, 2-butinyl and 3-butinyl.
- Preferred is a substituted dimethylcyclobutyl compound of general formula I, wherein the stereoisomers have the general formulae Ia or Ib or Ic or Id,
- n have the above defined meanings.
- a dimethylcyclobutyl compound of general formula I wherein n is 0 or 1 ; and the other substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- dimethylcyclobutyl compound of general formula I wherein m is 1 ;
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- X represents a -OR 5 moiety or a -NR 6 R 7 moiety; a (hetero)cycloaliphatic radical selected from the group consisting of cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, imidazolidinyl, aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, pyrazolidinyl, azepanyl (1 ,2,3,6)-tetrahydropyridinyl, (1 ,2,3,4)- tetrahydropyridinyl, (1 ,2,5,6)-tetrahydropyhdinyl, hexahydropyhmidinyl, (5,6)- dihydro-4H-pyrimidinyl, in
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- (hetero)cycloaliphatic radical selected from the group consisting of cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, imidazolidinyl, aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, pyrazolidinyl, azepanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyridinyl, (1 ,2,5,6)-tetrahydropyridinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyrinnidinyl, indolinyl, isoindolinyl,
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- R 1 represents a hydrogen atom or a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, neo-pentyl, n-hexyl, vinyl, allyl, ethinyl, -CF 3 , -CFH 2 , - CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 -CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH, -CH 2 -CH 2 -SH, -CH 2 -NH 2 , -CH 2 -NH-CH 3 , -CH 2 -N(CHs) 2 , -CH 2 -
- N(C 2 Hs) 2 -CH 2 -NH-C 2 H 5 , -CH 2 -CH 2 -NH 2 , -CH 2 -CH 2 -NH-CH 3 , -CH 2 -CH 2 - N(CH 3 ) 2 , -CH 2 -CH 2 -N(C 2 Hs) 2 , -CH 2 -CH 2 -NH-C 2 H 5 , -CH 2 -CH 2 -CH 2 -NH-CH 3 , - CH 2 -CH 2 -CH 2 -N(CHs) 2 , -CH 2 -CH 2 -CH 2 -N(C 2 Hs) 2 and -CH 2 -CH 2 -CH 2 -NH- C 2 Hs;
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- R 2 represents a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n- pentyl, neo-pentyl, n-hexyl, vinyl, allyl, ethinyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 -CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH, -CH 2 - CH 2 -SH, -CH 2 -NH 2 , -CH 2 -NH-CH 3 , -CH 2 -N(CHs) 2 , -CH 2 -N(C 2 Hs
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- dimethylcyclobutyl compound of general formula I wherein R 3 and R 4 both represent a hydrogen atom;
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- R 5 and R 8 independently of one another, each represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, neo-pentyl, n-hexyl, vinyl, allyl, ethinyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 - CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH, -CH 2 -CH 2 -SH, -CH 2 -NH 2 , -CH 2 - NH-CH 3 , -CH 2 -N(CHs) 2 , ,
- a (hetero)cycloaliphatic radical selected from the group consisting of imidazolidinyl, aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, pyrazolidinyl, azepanyl, (1 ,2,3,6)-tetrahydropyhdinyl, (1 ,2,3,4)-tetrahydropyridinyl, (1 ,2,5,6)- tetrahydropyridinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyhmidinyl, indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)- tetrahydroisoquinolinyl and
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, neo-pentyl, n-hexyl, vinyl, allyl, ethinyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 - CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH, -CH 2 -CH 2 -SH, -CH 2 -NH 2 ,
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- a dimethylcyclobutyl compound of general formula I wherein
- R 6 and R 7 together with the bridging nitrogen atom form a heterocyclic ring selected from the group consisting of pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, morpholinyl, aziridinyl, azetidinyl, imidazolidinyl, thiomorpholinyl, pyrazolidinyl, azepanyl, diazepanyl, azocanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyhdinyl, (1 ,2,5,6)-tetrahydropyhdinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyrimidinyl, indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)-tetrahydroisoquino
- R 9 and R 10 together with the bridging nitrogen atom form a heterocyclic ring selected from the group consisting of pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, morpholinyl, aziridinyl, azetidinyl, imidazolidinyl, thiomorpholinyl, pyrazolidinyl, azepanyl, diazepanyl, azocanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyhdinyl, (1 ,2,5,6)-tetrahydropyhdinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyrimidinyl, indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)-tetrahydroisoquino
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- R 11 and R 12 independently of one another, represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, neo-pentyl, n-hexyl, vinyl, allyl, ethinyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 - CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH, -CH 2 -CH 2 -SH, -CH 2 -NH 2 , -CH 2 - NH-CH 3 , -CH 2 -N(CHs) 2 , -CH
- a (hetero)cycloaliphatic radical selected from the group consisting of cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, imidazolidinyl, aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, pyrazolidinyl and azepanyl, which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent
- substituents have any of the above defined meanings, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding solvate thereof.
- n is 0 or 1 ;
- X represents a -OR 5 moiety or a -NR 6 R 7 moiety; or a (hetero)cycloaliphatic radical selected from the group consisting of pyrrolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, pyrazolidinyl, azepanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyhdinyl, (1 ,2,5,6)-tetrahydropyhdinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyrimidinyl, indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)-tetrahydroisoquinolinyl and octahydro- cyclopenta[c]pyrrolyl, which may
- R 1 represents a hydrogen atom or a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 - CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH and -CH 2 -CH 2 -SH;
- R 2 represents a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n- pentyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 -CH 2 -CN, - CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 -SH and -CH 2 -CH 2 -SH; or an aryl or heteroaryl radical selected from the group consisting of phenyl, naphthyl, furyl (furanyl), thienyl (thiophenyl), pyrrolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl
- R 3 and R 4 both represent a hydrogen atom
- R 5 and R 8 independently of one another, each represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, -CF 3 , -CFH 2 , -CF 2 H,
- aryl or heteroaryl radical selected from the group consisting of phenyl, naphthyl, furyl (furanyl), thienyl (thiophenyl), pyrrolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, imidazolyl, pyrazolyl, oxadiazolyl, thiadiazolyl, triazolyl, pyridinyl, pyridazinyl, pyrimidinyl, pyrazinyl, quinolinyl, isoquinolinyl, benzo[b]furanyl, benzo[b]thiophenyl, benzothiadiazolyl and imidazo[
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 -CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 - SH and -CH 2 -CH 2 -SH, ; or an aryl or heteroaryl radical selected from the group consisting of phenyl, naphthyl, furyl (furanyl), thienyl (thiophenyl), pyrrolyl, oxazolyl,
- R 6 and R 7 together with the bridging nitrogen atom form a heterocyclic ring selected from the group consisting of pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, morpholinyl, aziridinyl, azetidinyl, imidazolidinyl, thiomorpholinyl, pyrazolidinyl, azepanyl, diazepanyl, azocanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyridinyl, (1 ,2,5,6)-tetrahydropyridinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyrimidinyl, indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)-tetrahydroisoquinolin
- R 9 and R 10 together with the bridging nitrogen atom form a heterocyclic ring selected from the group consisting of pyrrolidinyl, piperidinyl, piperazinyl, homopiperazinyl, morpholinyl, aziridinyl, azetidinyl, imidazolidinyl, thiomorpholinyl, pyrazolidinyl, azepanyl, diazepanyl, azocanyl, (1 ,2,3,6)- tetrahydropyridinyl, (1 ,2,3,4)-tetrahydropyhdinyl, (1 ,2,5,6)-tetrahydropyhdinyl, hexahydropyrimidinyl, (5,6)-dihydro-4H-pyrimidinyl, indolinyl, isoindolinyl, (1 ,2,3,4)-tetrahydroquinolinyl, (1 ,2,3,4)-tetrahydroisoquino
- R 11 and R 12 independently of one another, represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, -CF 3 , -CFH 2 , -CF 2 H, -CH 2 -CF 3 , -CF 2 -CF 3 , -CH 2 -CN, -CH 2 -CH 2 -CN, -CH 2 -OH, -CH 2 -CH 2 -OH, -CH 2 - SH and -CH 2 -CH 2 -SH; or an aryl or heteroaryl radical selected from the group consisting of phenyl, naphthyl, furyl (furanyl), thienyl (thiophenyl), pyrrolyl, and pyridinyl, which may be unsubstitute
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- n is 0 or 1 ;
- X represents a -OR 5 moiety or a -NR 6 R 7 moiety
- R 1 represents a hydrogen atom or a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl;
- R 3 and R 4 both represent a hydrogen atom
- R 5 and R 8 independently of one another, each represent a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; an aryl radical selected from the group consisting of phenyl and naphthyl, which may be bonded via a -(CH 2 )i, 2or3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, tert-butyl, -O-CH 3 , -0-C 2 H 5 -F, CI, Br, I, -CN, -CF 3 , -OCF 3 , -SCF 3 ,
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; or an aryl or heteroaryl radical selected from the group consisting of phenyl and naphthyl, which may be bonded via a -(CH 2 )i, 2 or 3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, tert-butyl, sec-butyl, isobutyl, -O-CH 3 , -0-C 2 H 5 , -S-CH 3 , -S-C 2
- R 6 and R 7 together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 9 and R 10 together with the bridging nitrogen atom form a moiety selected from the group consisting of
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- n is 0 or 1 ;
- X represents a -OR 5 moiety or a -NR 6 R 7 moiety
- R 1 represents a hydrogen atom or a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl;
- R 2 represents a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; or a phenyl radical which may be unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, sec-butyl, isobutyl, n- pentyl, -O-CH 3 , -0-C 2 H 5 , F, CI, Br, I, -CN, -CF 3 , -OCF 3 and -SCF 3 ;
- R 3 and R 4 both represent a hydrogen atom;
- R 5 and R 8 independently of one another, each represent a hydrogen atom;
- a phenyl radical which may be bonded via a -(CH 2 )i, 2 or 3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, -0-CH 3 , -0-C 2 H 5 , F, Cl, Br, I, -CN, -CF 3 and -
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; or a phenyl radical, which may be bonded via a -(CH 2 )i, 2or3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, tert-butyl, sec-butyl, isobutyl, -O-CH 3 , -0-C 2 H 5 , F, CI, Br, I, -CN, -CF 3 , - OCF 3 and -SCF 3 ;
- R 6 and R 7 together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 9 and R 10 together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 11 and R 12 independently of one another, represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec- butyl, isobutyl, tert-butyl and n-pentyl;
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- n is 0 or 1 ;
- X represents a -OR 5 moiety or a -NR 6 R 7 moiety
- R 1 represents a hydrogen atom or a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl;
- R 2 represents a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; or a phenyl radical which may be unsubstituted or substituted with
- substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, sec-butyl, isobutyl, n- pentyl, -O-CH 3 , -0-C 2 H 5 , F, CI, Br, I, -CN, -CF 3 , -OCF 3 and -SCF 3 ;
- R 3 and R 4 both represent a hydrogen atom
- R 6 , R 7 , R 9 and R 10 independently of one another, each represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; or a phenyl radical, which is bonded via a -(CH 2 )i, 2or3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, tert-butyl, sec-butyl, isobutyl, -O-CH 3 , -0-C 2 H 5 , F, CI, Br, I, -CN, -CF 3 , -
- R 6 and R 7 together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 9 and R 10 together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 11 and R 12 independently of one another, represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec- butyl, isobutyl, tert-butyl and n-pentyl;
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- ne is O or i ;
- R 6e and R 7e independently of one another, each represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec- butyl, isobutyl, tert-butyl and n-pentyl; or an aryl or heteroaryl radical selected from the group consisting of phenyl and naphthyl, which may be bonded via a -(CH 2 )i, 2or3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, sec-butyl, isobutyl, -O-CH 3 , -0-C 2 H 5 , -S-CH 3 , -S-C 2 H 5 , F
- R 6e and R 7e together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 5 8e represents a hydrogen atom; a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; an aryl radical selected from the group consisting of phenyl and naphthyl, which may be bonded via a -(CH 2 )i, 2 or3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, tert-butyl, -0-CH 3 , -0-C 2 H 5 -F, Cl, Br, I, -CN, -CF 3 , -OCF 3 , SCF 3 , -OH, -SH, -NH
- R 12e represents a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl;
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- Preferred is a substituted dimethylcyclobutyl compound of general formula Ie, wherein the stereoisomers have the general formulae If or Ig or Ih or Ij,
- ne, R 6e , R 7e and R 8e have the above defined meanings.
- nk O or i ;
- R 6k , R 7k , R 9k and R 1Ok independently of one another, each represent a radical selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, sec-butyl, isobutyl, tert-butyl and n-pentyl; or an aryl or heteroaryl radical selected from the group consisting of phenyl and naphthyl, which may be bonded via a -(CH 2 )i, 2 o r 3-group and which may be unsubstituted or optionally substituted with 1 , 2, 3, 4 or 5 substituent(s) independently selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n- butyl, tert-butyl, sec-butyl, isobutyl, -0-CH 3 , -0-C 2 H 5 , -S-CH 3
- R 6k and R 7k together with the bridging nitrogen atom form a moiety selected from the group consisting of
- R 9k and R 1Ok together with the bridging nitrogen atom form a moiety selected from the group consisting of
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
- Preferred is a substituted dimethylcyclobutyl compound of general formula Ik, wherein the stereoisomers have the general formulae Im or In or Io or Ip,
- nk, R 6k , R 7k , R 9k and R 1Ok have the above defined meanings.
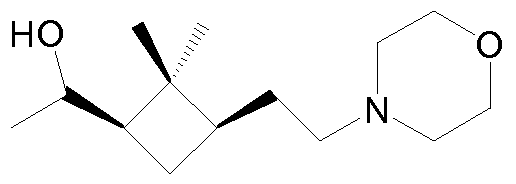
- dimethylcyclobutyl compound selected from the group consisting of
- stereoisomers optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two of its stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a salt thereof, or a corresponding solvate thereof.
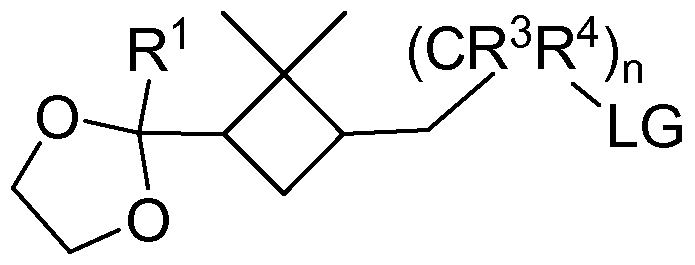
- the present invention relates to a process for the preparation of a substituted dimethylcyclobutyl compound of general formula
- m, R 1 and R 2 have the meaning given above and R represents a linear or branched Ci -5 -alkyl radical, is reacted with methane sulfonylchloride, p-toluene sulfonylchloride, trifluormethane sulfonylchloride, thionyl chloride or tetrabromethane, preferably in a reaction medium selected from the group consisting of diethylether, tetrahydrofuran, tert-butylmethylether and dichloromethane or a mixture thereof, more preferably in dichloromethane, preferably in the presence of at least one base selected from the group consisting of pyridine, triethylamine, diisopropylamine, diisopropylethylamine, 4-methylmorpholine and morpholine, to yield at least one compound of general formula III,
- R 1 , R 2 , R 6 and R 7 have the meaning given above and R represents a linear or branched Ci -5 -alkyl radical, which is optionally purified and/or isolated,
- At least one compound of general formula IV is reacted with at least one reducing agent selected from the group consisting of lithium borohydride, sodium borohydride, lithium aluminium hydride and diborane, preferably with lithium borohydride, preferably in a reaction medium selected from the group consisting of methanol, ethanol, hexane and tetrahydrofuran or a mixture thereof, more preferably in methanol and/or tetrahydrofuran, to yield at least one compound of general formula V,
- M represents a monovalent kation selected from the group consisting of sodium, magnesium, potassium and lithium, preferably M represents a lithium kation, preferably in a reaction medium selected from the group consisting of dimethylformamide, dichloromethane and tetrahydrofuran or a mixture thereof, more preferably in tetrahydrofuran, preferably in the presence of at least one base selected from the group consisting of pyridine, triethylamine, diisopropylamine, diisopropylethylamine, 4-methylmorpholine and morpholine, to yield at least one compound of general formula VIII,
- R 8 represents hydrogen
- the present invention relates to a process for the preparation of a substituted dimethylcyclobutyl compound of general formula I, wherein at least one compound of general formula II,
- R 1 , R 2 and R 5 have the meaning given above and R represents a linear or branched Ci -5 -alkyl radical, which is optionally purified and/or isolated,
- At least one compound of general formula IX is reacted with at least one reducing agent selected from the group consisting of lithium borohydhde, lithium aluminium hydride and diborane, sodium borohydride, preferably with lithium borohydride, preferably in a reaction medium selected from the group consisting of methanol, ethanol, hexane and tetrahydrofuran or a mixture thereof, more preferably in methanol and/or tetrahydrofuran, to yield at least one compound of general formula X,
- At least one compound of general formula Xl is reacted with at least one compound of general formula HNR 9 R 10 , wherein R 9 and R 10 have the meaning given above, preferably in a reaction medium selected from the group consisting of dimethylformamide, acetonitrile, dichloromethane and tetrahydrofuran or a mixture thereof, preferably in the presence of at least one base selected from the group consisting of pyridine, triethylamine, diisopropylamine, diisopropylethylamine, 4-methylmorpholine and morpholine, to yield at least one compound of general formula XII,
- the present invention relates to a process for the preparation of a substituted dimethylcyclobutyl compound of general formula I, wherein at least one compound of general formula XIII,
- R 1 , R 3 and R 4 have the meaning given above, is reacted with methane sulfonylchloride, p-toluene sulfonylchloride, trifluormethane sulfonylchloride, thionyl chloride or tetrabromethane, preferably in a reaction medium selected from the group consisting of diethylether, tetrahydrofuran, tert-butylmethylether and dichloromethane or a mixture thereof, more preferably in dichloromethane, preferably in the presence of at least one base selected from the group consisting of pyridine, thethylamine, diisopropylamine, diisopropylethylamine, 4-methylmorpholine and morpholine,to yield at least one compound of general formula XIV,
- At least one compound of general formula XIV is reacted with at least one compound of general formula HNR 9 R 10 , wherein R 9 and R 10 have the meaning given above, preferably in a reaction medium selected from the group consisting of dimethylformamide, acetonitrile, dichloromethane and tetrahydrofuran or a mixture thereof, preferably in the presence of at least one base selected from the group consisting of pyridine, triethylamine, diisopropylamine, diisopropylethylamine, 4-methylmorpholine and morpholine, to yield at least one compound of general formula XV,
- R 1 , R 3 , R 4 , R 9 and R 10 have the meaning given above, which is optionally purified and/or isolated,
- At least one compound of general formula XV is reacted with at least one reagent selected from the group consisting of hydrochloric acid, pyhdinium p- toluenesulfonate, sulfonic acid and trifluoroacetic acid, preferably with pyhdinium p-toluenesulfonate, preferably in a reaction medium selected from the group consisting of acetone and water or a mixture thereof, to yield at least one compound of general formula XVI,
- R 1 , R 3 , R 4 , R 9 and R 10 have the meaning given above, which is optionally purified and/or isolated, and at least one compound of general formula XVI is reacted with at least one compound of general formula R 2 -Li, wherein R 2 has the meaning given above, or R 2 -Mg-Z, wherein R 2 has the meaning given above, and Z represents an anion selected from the group consisting of bromide and chloride, preferably in a reaction medium, selected from the group consisting of ether and tetrahydrofurane, to yield at least one compound of general formula XVII,
- R 1 , R 2 , R 3 , R 4 , R 9 and R 10 have the meaning given above, which is optionally purified and/or isolated,
- R 1 , R 2 , R 3 , R 4 , R 5 , R 9 and R 10 have the meaning given above, which is optionally purified and/or isolated.
- Ic, Id, Ie If, Ig, Ih, Ij, Ik, Im, In, Io and Ip are obtained in form of a mixture of stereoisomers, particularly enantiomers or diastereomers, said mixtures may be separated by standard procedures known to those skilled in the art, e.g. chromatographic methods or crystallization with chiral reagents.
- Suitable reaction media include, for example, any of the ones given above.
- Suitable inorganic acids include but are not limited to hydrochloric acid, hydrobromic acid, phosphoric acid, oxalic acid, sulfuric acid, nitric acid
- suitable organic acids include but are not limited to citric acid, maleic acid, fumaric acid, tartaric acid, or derivatives thereof, p- toluenesulfonic acid, methanesulfonic acid or camphersulfonic acid.
- salt is to be understood as meaning any form of the substituted dimethylcyclobutyl compounds in which they assume an ionic form or are charged and are coupled with a counter-ion (a cation or anion) or are in solution.
- a counter-ion a cation or anion
- complexes of the active compound with other molecules and ions in particular complexes which are complexed via ionic interactions.
- physiologically acceptable salt is understood in particular, in the context of this invention, as salt (as defined above) formed either with a physiologically tolerated acid, that is to say salts of the particular active compound with inorganic or organic acids which are physiologically tolerated - especially if used on humans and/or mammals - or with at least one, preferably inorganic, cation which are physiologically tolerated - especially if used on humans and/or mammals.
- physiologically tolerated salts of particular acids are salts of: hydrochloric acid, hydrobromic acid, sulfuric acid, hydrobromide, monohydrobromide, monohydrochlohde or hydrochloride, methiodide, methanesulfonic acid, formic acid, acetic acid, oxalic acid, succinic acid, malic acid, tartaric acid, mandelic acid, fumaric acid, lactic acid, citric acid, glutamic acid, hippuric acid picric acid and/or aspartic acid.
- physiologically tolerated salts of particular bases are salts of alkali metals and alkaline earth metals and with NH 4 .
- solvate is to be understood as meaning any form of the substituted dimethylcyclobutyl compounds in which they have attached to it via non-covalent binding another molecule (most likely a polar solvent) especially including hydrates and alcoholates, e.g. ethanolate.
- a further aspect of the present invention relates to a medicament comprising at least one substituted dimethylcyclobutyl compound of general formulae I,
- Ia, Ib, Ic, Id, Ie If, Ig, Ih, Ij, Ik, Im, In, Io and Ip given above, optionally in form of one of its stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a physiologically acceptable salt thereof, or a corresponding solvate thereof, and optionally at least one physiologically acceptable auxiliary agent.
- one of its stereoisomers preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a physiologically acceptable salt thereof, or a corresponding solvate thereof, and optionally at least one physiologically acceptable auxiliary agent.
- Said medicament is particularly suitable for sigma receptor regulation, preferably for sigma-1 receptor regulation, and can therefore be used for the prophylaxis and/or treatment of a disorder or a disease that is least partially mediated via sigma receptors, preferably sigma-1 receptors.
- said medicament is suitable for the prophylaxis and/or treatment of a disorder or disease related to food intake, preferably for the regulation of appetite, for the maintenance, increase or reduction of body weight, for the prophylaxis and/or treatment of obesity, bulimia, anorexia, cachexia, type Il diabetes (non insulin dependent diabetes mellitus), preferably type Il diabetes that is caused by obesity; for the prophylaxis and/or treatment of diarrhoea; lipoprotein disorders, preferably selected from the group consisting of hypercholesterolemia (type Il hyperlipoproteinemia); hypertriglyceridemia; hypoalphalipoproteinemia and high lipoprotein(a) levels; arthritis; hypertension; arrhythmia; ulcer; tardive dyskinesia; stress; cancer; stroke; ischemic stroke; migraine; epilepsy; anxiety; panic attacks; depression; cognitive disorders; cognitive dysfunction associated with psychiatric diseases; memory disorders; psychosis; schizophrenia; for the prophylaxis and/or treatment of drug addiction and
- said medicament is suitable for the prophylaxis and/or treatment of pain, preferably neuropathic pain, allodynia, analgesia, causalgia, central pain, dysesthesia, hyperesthesia, hyperalgesia, hypoalgesia, hypoesthesia, or neuralgia, more preferably neuropathic pain, hyperalgesia or allodynia.
- the present invention relates to the use of at least one substituted dimethylcyclobutyl compound of general formulae I, Ia, Ib, Ic, Id, Ie, If, Ig, Ih, Ij, Ik, Im, In, Io and Ip given above, optionally in form of one of their stereoisomers, preferably enantiomers or diasteromers, a racemate or in form of a mixture of at least two stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a physiologically acceptable salt thereof, or a corresponding solvate thereof, for the preparation of a medicament suitable for sigma receptor regulation, preferably for sigma-1 receptor regulation and/or preferably for the prophylaxis and/or treatment of a disorder or a disease that is least partially mediated via sigma receptors, preferably sigma-1 receptors.
- Any medicament according to the present invention may be in any form suitable for the application to humans and/or animals, preferably humans including infants, children and adults.
- the medicament can be produced by standard procedures known to those skilled in the art, e.g. from the table of contents of
- composition of the medicament may vary depending on the route of administration.
- the medicament of the present invention may, for example, be administered parentally in combination with conventional injectable liquid carriers, such as water or suitable alcohols.
- conventional pharmaceutical excipients for injection such as stabilizing agents, solubilizing agents, and buffers, may be included in such injectable compositions.
- These medicaments may for example be injected intramuscularly, intraperitoneally, or intravenously.
- Medicaments according to the present invention may also be formulated into orally administrable compositions containing one or more physiologically compatible carriers or excipients, in solid or liquid form.
- These compositions may contain conventional ingredients such as binding agents, fillers, lubricants, and acceptable wetting agents.
- the compositions may take any convenient form, such as tablets, pellets, granules, capsules, lozenges, aqueous or oily solutions, suspensions, emulsions, or dry powdered forms suitable for reconstitution with water or other suitable liquid medium before use, for immediate or retarded release.
- the multiparticulate forms, such as pellets or granules may e.g. be filled into a capsule, compressed into tablets or suspended in a suitable liquid.
- Suitable controlled release formulations, materials and methods for their preparation are known from the prior art, e.g. from the table of contents of "Modified-Release Drug Delivery Technology", Rathbone, M.J. Hadgraft, J. and Roberts, M.S. (Eds.), Marcel Dekker, Inc., New York (2002); "Handbook of Pharmaceutical Controlled Release Technology”, Wise, D. L. (Ed.), Marcel Dekker, Inc. New York, (2000); "Controlled Drug Delivery", VoI, I, Basic
- Medicaments according to the present invention may also comprise an enteric coating, so that their dissolution is dependent on pH-value. Due to said coating the medicament can pass the stomach undissolved and the respective dimethylcyclobutyl compound is liberated in the intestinal tract.
- the enteric coating is soluble at a pH value of 5 to 7.5. Suitable materials and methods for the preparation are known from the prior art.
- the medicaments according to the present invention may contain 1 -
- liquid oral forms for administration may also contain certain additives such as sweeteners, flavoring, preservatives, and emulsifying agents.
- Nonaqueous liquid compositions for oral administration may also be formulated, containing edible oils. Such liquid compositions may be conveniently encapsulated in e.g., gelatin capsules in a unit dosage amount.
- compositions of the present invention may also be administered topically or via a suppository.
- the daily dosage for humans and animals may vary depending on factors that have their basis in the respective species or other factors, such as age, sex, weight or degree of illness and so forth.
- the daily dosage for humans may preferably be in the range fromi to 2000 mg, preferably 1 to 1500 mg, more preferably 1 to 1000, even more preferably 1 to 500 mg, most preferably 1 to 100 mg of active substance to be administered during one or several intakes per day.
- Brain membrane preparation and binding assays for the sigma-1 receptor were performed as described (DeHaven-Hudkins et al., Characterization of the binding of [ 3 H]-(+)-pentazocine to recognition sites in guinea pig brain, Eur. J. Pharmacol. 227, 371-378) with some modifications.
- guinea pig brains were homogenized in 10 vols. (w/v) of Tris-HCI 50 mM, 0.32 M sucrose, pH 7.4, with a Kinematica Polytron PT 3000 at 15000 r.p.m. for 30 s.
- the homogenate was centhfuged at 1000 g for 10 min at 4 0 C and the supernatants collected and centrifuged again at 48000 g for 15 min at 4 0 C.
- the pellet was resuspended in 10 volumes of Tris-HCI buffer (50 mM, pH 7.4), incubated at 37 0 C for 30 min, and centrifuged at 48000 g for 20 min at 4 0 C. Following this, the pellet was resuspended in fresh Tris-HCI buffer (50 mM, pH 7.4) and stored on ice until use.
- Each assay tube contained 10 ⁇ L of [ 3 H]-(+)-pentazocine (final concentration of 0.5 nM), 900 ⁇ L of the tissue suspension to a final assay volume of 1 mL and a final tissue concentration of approximately 30 mg tissue net weight/mL.
- Non-specific binding was defined by addition of a final concentration of 1 M haloperidol.
- All tubes were incubated at 37 0 C for 150 min before termination of the reaction by rapid filtration over Schleicher & Schuell GF 3362 glass fibre filters [previously soaked in a solution of 0,5% polyethylenimine for at least 1 h]. Filters were then washed with four times with 4 ml_ of cold Tris-HCI buffer (50 mM, pH 7.4).
- Binding studies for sigma-2 receptor were performed as described (Radesca et al., Synthesis and Receptor Binding of Enantiomeric N-Substituted cis-N- [2-(3,4-Dichlorophenyl)ethyl]-2-(1 -pyrrolidinyl)ciclohexylamines as High- Affinity Receptor Ligands, J. Med. Chem. 34, 3065-3074) with some modifications.
- brains from sigma receptor type I (sigma-1 ) knockout mice (Langa, F., Codony X., Tovar V., Lavado A., Gimenez E., Cozar P., Cantero M., Dordal A., Hernandez E., Perez R., Monroy X., Zamanillo D., Guitart X., Montoliu LL, 2003, Generation and phenotypic analysis of sigma receptor type I (Sigmal ) knockout mice, European Journal of Neuroscience, Vol.
- the assay tubes contained 10 ⁇ L of [ 3 H]-DTG (final concentration of 3 nM), 400 ⁇ L of the tissue suspension (5.3 mL/g in 50 mM Tris-HCI, pH 8.0) to a final assay volume of 0.5 mL. Non-specific binding was defined by addition of a final concentration of 1 M halopehdol. All tubes were incubated at 25 0 C for
- a compound of general formula II wherein both R 1 and R 2 represent hydrogen and R represents tert-butyl, can be prepared starting from (-)- ⁇ - pinene as depicted in scheme 1. below.
- Aqueous NaOBr prepared from bromine (9.4 ml_, 183.2 mmol), NaOH (28.4 g, 710.9 mmol) and water (237 ml_) was added to a solution of tert-butyl 2- ((1 R,3R)-3-acetyl-2,2-dimethylcyclobutyl)acetate (5.5 g, 22.9 mmol) in 1 ,4- dioxane (138 ml_) and water (38 ml_).
- the reaction mixture was cooled to -5 0 C and stirred at temperature between -5 and 0 0 C for 5 h.
- the reaction mixture was washed with dichloromethane (4 x 200 ml_).
- Triethylamine (1.2 ml_, 8.7 mmol) and mesyl chloride 0.6 ml_, 8.1 mmol) were added to an ice-cooled solution of alcohol 2-((1 R,3R)-2,2-dimethyl-3- (morpholinomethyl)cyclobutyl)ethanol (1.4 g, 6.2 mmol) under nitrogen atmosphere. After 1 h stirring the reaction mixture was washed with saturated aqueous NaHCO 3 (4x50 ml_), and the organic phase was dried over MgSO 4 .
- example compound 46 i ⁇ -UI R ⁇ RJ ⁇ -dimethyl-S ⁇ morpholinomethyOcyclobutyOethyO- ⁇ J- dihydro-1 H-indol-4(5H)-one
- Example compound 53 6,7-Dihydro-1-(2-((1/?,3/?)-2,2-dimethyl-3- ((piperidin-1-yl)methyl)cyclobutyl)ethyl)-1H-indol-4(5H)-one
- Example compound 42 4-(((1R,3R)-3-(2-(4-Benzylpiperidin-1-yl)ethyl)- 2,2-dimethylcyclobutyl)methyl)morpholine
- Example compound 52 4-Benzyl-1-(2-((1/?,3/?)-2,2-dimethyl-3- ((piperidin-1-yl)methyl)cyclobutyl)ethyl)piperidine
- Example compound 41 4-(((1/?,3/?)-2,2-Dimethyl-3-(2-(4- phenylpiperidin-1-yl)ethyl)cyclobutyl)methyl)morpholine
- Example compound 51 1-(2-((1/?,3/?)-2,2-Dimethyl-3-((piperidin-1- yl)methyl)cyclobutyl)ethyl)-4-phenylpiperidine
- Example compound 40 4-(((1R,3R)-2,2-Dimethyl-3-(2-(3- phenylpiperidin-1-yl)ethyl)cyclobutyl)methyl)morpholine
- Example compound 43 4-(((1R,3R)-3-(2-(1H-lmidazol-1-yl)ethyl)-2,2- dimethylcyclobutyl)methyl)morpholine
- Example compound 48 1-(((1R,3R)-3-(2-(1H-lmidazol-1-yl)ethyl)-2,2- dimethylcyclobutyl)methyl)piperidine
- Example compound 44 4-(((7R,3R)-3-(2-(1H-Pyrazol-1-yl)ethyl)-2,2- dimethylcyclobutyl)methyl)morpholine
- Example compound 49 1-(((1R,3R)-3-(2-(1H-Pyrazol-1-yl)ethyl)-2,2- dimethylcyclobutyl)methyl)piperidine
- Example compound 45 4-(((1R,3R)-3-(2-(1H-1,2,4-Triazol-1-yl)ethyl)-2,2- dimethylcyclobutyl)methyl)morpholine
- Example compound 50 1-(((1R,3R)-3-(2-(1H-1,2,4-Triazol-1-yl)ethyl)-2,2- dimethylcyclobutyl)methyl)piperidine
- Example compound 47 1,2,3,4-Tetrahydro-2-(2-((1R,3R)-2,2-dimethyl-3- (morpholinomethyl)cyclobutyl)ethyl) isoquinoline
- Example compound 54 1,2,3,4-Tetrahydro-2-(2-((1R,3R)-2,2-dimethyl-3- ((piperidin-1-yl)methyl)cyclobutyl)ethyl)isoquinoline
- example compound 15 4-(((1 R,3R)-2,2-Dimethyl-3-(2- phenoxyethyl)cyclobutyl)methyl)morpholine)
- Example compound 16 1 (((1 R,3R)-2,2-Dimethyl-3-(2- phenoxyethyl)cyclobutyl)methyl)piperidine was prepared in a similar manner.
- Thethylamine (0.8 ml_, 5.7 mmol) and pivaloyl chloride (0.6 ml_, 5.2 mmol) were added to an ice-cooled solution of 2-((1 R,3R)-2,2-dimethyl-3-(piperidin- 1 -ylmethyl)cyclobutyl)ethanol (0.9 g, 4.0 mmol) in anhydrous dichloromethane (50 ml_) under nitrogen atmosphere. After stirring at O 0 C for 1 h, the reaction mixture was washed with saturated aqueous NaHCO3 (4 x 25 ml_) and dried over MgSO 4 .
- the example compound 56 2-((1 R,3R)-2,2-Dimethyl-3-((piperidin-1 - yl)methyl)cyclobutyl)ethyl acetate was prepared accordingly.
- Example compound 3 1-(((1 S,3S)- 3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-phenylpiperidine
- Example compound 27 1-(((1R,3/?)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)piperidine
- Example compound 31 4-(((1R,3/?)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)morpholine
- Example compound 1 4-(((1S,3S)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)morpholine
- Example compound 30 4-(((1R,3R)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)thiomorpholine
- Example compound 33 1-(((1R,3/?)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)pyrrolidine - 1 H-NMR (250 MHz, CDCI 3 )
- Example compound 28 1-(((1/?,3/?)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-methylpiperidine - 1 H-NMR (250 MHz, CDCI 3 )
- Example compound 34 1-(((1R,3R)- 3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-methylpiperazine
- Example compound 5 1-(((1 S,3S)- 3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-methylpiperazine
- Example compound 36 ' ⁇ - ⁇ ' ⁇ R,3R)- 3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-phenylpiperazine
- Example compound 4 1-(((1S,3S)- 3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-phenylpiperazine
- Example compound 7 1-(((1R,3S)-3-((Benzyloxy)methyl)-2,2- dimethylcyclobutyl)methyl)piperidine
- Example compound 10 1-(((1R,3S)-3-((Benzyloxy)methyl)-2,2- dimethylcyclobutyl)methyl)-4-methylpiperazine
- Example compound 9 1-(((1R,3S)-3-((Benzyloxy)methyl)-2,2- dimethylcyclobutyl)methyl)-4-phenylpiperazine
- Example compound 8 1-(((1R,3S)-3-((Benzyloxy)methyl)-2,2- dimethylcyclobutyl)methyl)-4-phenylpiperidine
- Example compound 35 1-(((1/?,3/?)-3-(2-(Benzyloxy)ethyl)-2,2- dimethylcyclobutyl)methyl)-4-methylpiperazine oxalate
- a compound of general formula XIII, wherein R 1 , R 3 and R 4 represent hydrogen and n is 1 , can be prepared starting from (-)- ⁇ -pinene as depicted in scheme 2. below.
- Example compound 13 1-((1/?,3/?)-2,2-Dimethyl-3-(2- morpholinoethyl)cyclobutyl)-1-phenylethanol
- Phenyllithium [1.9 M in ether (0.8 ml_, 1.5 mmol)] was added to an ice-cooled solution of 1-((1 R,3R)-2,2-dimethyl-3-(2-nnorpholinoethyl)cyclobutyl)ethanone (170 mg, 0.7 mmol) in ahydrous THF (8 ml_). The mixture was allowed to warm to room temperature and stirred overnight under nitrogen atmosphere. Excess of Phenyllithium was destroyed with wet ether and then water was added. The layers were separated and the solvent was evaporated from the organic phase. The residue was poured into EtOAc, washed with brine and dried over Mg SO4.
- Example compound 14 1-((1/?,3/?)-2,2-Dimethyl-3-(2-(piperidin-1- yl)ethyl)cyclobutyl)-1 -phenylethanol
- Example compound 12 1-((1R,3R)-2,2-Dimethyl-3-(2-(piperidin-1- yl)ethyl)cyclobutyl)ethanol
- Example compound 11 1-((1R,3R)-2,2-Dimethyl-3-(2-morpholinoethyl)- cyclobutyl)ethanol )
- the substituted dimethylcyclobutyl compounds of general formula I show a high affinity to sigmal receptors (table 1 ).
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07788185.2A EP2066648B1 (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments |
| CA002660028A CA2660028A1 (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments |
| JP2009523253A JP2009545625A (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicine |
| MX2009001315A MX2009001315A (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments. |
| ES07788185.2T ES2632610T3 (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicines |
| US12/376,283 US20100105680A1 (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP06384013.6 | 2006-08-04 | ||
| EP06384013 | 2006-08-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008015266A1 true WO2008015266A1 (en) | 2008-02-07 |
Family
ID=37395822
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2007/058047 WO2008015266A1 (en) | 2006-08-04 | 2007-08-03 | Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20100105680A1 (en) |
| EP (1) | EP2066648B1 (en) |
| JP (1) | JP2009545625A (en) |
| CN (1) | CN101563332A (en) |
| CA (1) | CA2660028A1 (en) |
| ES (1) | ES2632610T3 (en) |
| MX (1) | MX2009001315A (en) |
| WO (1) | WO2008015266A1 (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2353598A1 (en) * | 2010-02-04 | 2011-08-10 | Laboratorios Del. Dr. Esteve, S.A. | Sigma ligands for use in the prevention and/or treatment of postoperative pain |
| EP2567959A1 (en) | 2011-09-12 | 2013-03-13 | Sanofi | 6-(4-Hydroxy-phenyl)-3-styryl-1H-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
| EP2832720A1 (en) | 2013-07-30 | 2015-02-04 | Laboratorios Del. Dr. Esteve, S.A. | 1,2-disubstituted cyclobutyl compounds |
| US9757358B2 (en) | 2010-02-04 | 2017-09-12 | Laboratorios Del Dr. Esteve, S.A. | Sigma ligands for potentiating the analgesic effect of opioids and opiates in post-operative pain and attenuating the dependency thereof |
| US9782483B2 (en) | 2010-05-21 | 2017-10-10 | Laboratories Del Dr. Esteve, S.A. | Sigma ligands for the prevention and/or treatment of emesis induced by chemotherapy or radiotherapy |
| US9789117B2 (en) | 2011-05-18 | 2017-10-17 | Laboratorios Del Dr. Esteve, S.A. | Use of sigma ligands in diabetes type-2 associated pain |
| US9789115B2 (en) | 2010-08-03 | 2017-10-17 | Laboratorios Del Dr. Esteve, S.A. | Use of sigma ligands in opioid-induced hyperalgesia |
| US9914705B2 (en) | 2008-04-25 | 2018-03-13 | Laboratorios Del Dr. Esteve, S.A. | 1-aryl-3-aminoalkoxy pyrazoles as sigma ligands enhancing analgesic effect of opioids and attenuating the dependency thereof |
| US9931346B2 (en) | 2013-12-17 | 2018-04-03 | Laboratorios Del Dr. Esteve S.A. | Serotonin-norepinephrine reuptake inhibitors (SNRIs) and Sigma receptor ligands combinations |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3387021A (en) * | 1965-04-08 | 1968-06-04 | Rohm & Haas | Amino hydroxyethyl cyclobutanes |
| US3453327A (en) * | 1965-01-26 | 1969-07-01 | Rohm & Haas | Pinene diamines and method of preparation |
-
2007
- 2007-08-03 EP EP07788185.2A patent/EP2066648B1/en not_active Not-in-force
- 2007-08-03 ES ES07788185.2T patent/ES2632610T3/en active Active
- 2007-08-03 WO PCT/EP2007/058047 patent/WO2008015266A1/en active Application Filing
- 2007-08-03 US US12/376,283 patent/US20100105680A1/en not_active Abandoned
- 2007-08-03 CA CA002660028A patent/CA2660028A1/en not_active Abandoned
- 2007-08-03 JP JP2009523253A patent/JP2009545625A/en active Pending
- 2007-08-03 CN CNA2007800366779A patent/CN101563332A/en active Pending
- 2007-08-03 MX MX2009001315A patent/MX2009001315A/en not_active Application Discontinuation
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3453327A (en) * | 1965-01-26 | 1969-07-01 | Rohm & Haas | Pinene diamines and method of preparation |
| US3387021A (en) * | 1965-04-08 | 1968-06-04 | Rohm & Haas | Amino hydroxyethyl cyclobutanes |
Non-Patent Citations (34)
| Title |
|---|
| AVOTINS ET AL., LATVIJAS PSR ZINATNU AKADEMIJAS VESTIS, KIMIJAS SERIJA, vol. 4, 1983, RUSSIA, pages 465 - 469, XP001248169 * |
| BERICHTE DER DEUTSCHEN CHEMISCHEN GESELLSCHAFT [ABTEILUNG] B: ABHANDLUNGEN , 70B, 1505-12 CODEN: BDCBAD; ISSN: 0365-9488, 1937 * |
| BLANCO ET AL., CHEM. PHARM. BULL., vol. 47, no. 9, 1999, pages 1314 - 1317, XP001248179 * |
| BULLETIN DE LA SOCIETE CHIMIQUE DE FRANCE , (2), 535-9 CODEN: BSCFAS; ISSN: 0037-8968, 1967 * |
| BULLETIN DE LA SOCIETE CHIMIQUE DE FRANCE , (2), 542-4 CODEN: BSCFAS; ISSN: 0037-8968, 1967 * |
| COLLET ET AL, TETRAHEDRON, vol. 31, 1975, pages 2243 - 2246, XP002408409 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; FRANCOIS, HUGUETTE ET AL: "(.+-.)-cis-Pinic acid derivatives. I. Pinic amino acids", XP002409795, retrieved from STN Database accession no. 1967:115820 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; FRANCOIS, HUGUETTE ET AL: "(.+-.)-cis-Pinic acid derivatives. III. Acidolysis of urea by (.+-.)-cis-pinic acid. Pinic diamine", XP002409796, retrieved from STN Database accession no. 1967:115822 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; GUHA, P. C. ET AL: "Synthetic experiments in the pinane group. III. Synthesis and configuration of pinic acid", XP002409787, retrieved from STN Database accession no. 1937:47839 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; JAOUI, MOHAMMED ET AL: "Mass balance of gaseous and particulate products analysis from .alpha.-pinene/NOx/air in the presence of natural sunlight", XP002409793, retrieved from STN Database accession no. 2001:533378 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; MINACHEV, KH. ET AL.: "Effect of thiophene on the properties of alumina-platinum catalyst in the conditions of methylcyclopentane dehydroisomerization", XP002409788, retrieved from STN Database accession no. 1965:462522 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; MOGLIONI, ALBERTINA G. ET AL: "Stereoselective synthesis of novel cyclobutane dehydro amino acids from (+)-.alpha.-pinene", XP002409790, retrieved from STN Database accession no. 1998:330842 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; OSTLING, G. J.: "Preparation of glycols corresponding with pinic, norpinic, and d-camphoric acids and their derivatives", XP002409791, retrieved from STN Database accession no. 1922:13382 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; PARK, JOSEPH D. ET AL: "Hydroxyalkyl and olefinic substituted gem-dimethylcyclobutanes", XP002409789, retrieved from STN Database accession no. 1965:462521 * |
| DATABASE CAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; THOMAS, ALAN F. ET AL: "The Baeyer-Villiger reaction of pinanones (bicyclo[3.1.1]heptanones)", XP002409792, retrieved from STN Database accession no. 1992:235889 * |
| DATABASE HCAPLUS [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; HERGUETA, ANTONIO R. ET AL: "Synthesis of two enantiomerically pure precursors of cyclobutane carbocyclic nucleosides", XP002409794, retrieved from STN Database accession no. 2003:928830 * |
| FERANDEZ ET AL, NUCLEOSIDES, NUCLEOTIDES AND NUCLEIC ACIDS, vol. 20, no. 4-7, 2001, pages 1129 - 1131, XP009075127 * |
| FIGUEIRA ET AL., SYNTHESIS, no. 10, 2000, pages 1459 - 1463, XP002408414 * |
| HARISPE M ET AL, COMPTES RENDUS DES SEANCES DE L'ACADEMIE DES SCIENCES. SERIE I: MATHEMATIQUES, EDITIONS SCIENTIFIQUES & MEDICALES ELSEVIER, FR, 1957, pages 1550 - 1552, XP009075140, ISSN: 0764-4442 * |
| HERGUETA ET AL., CHEM. PHARM. BULL., vol. 49, no. 9, 2001, pages 1174 - 1177, XP001247962 * |
| INDUSTRIAL & ENGINEERING CHEMISTRY PRODUCT RESEARCH AND DEVELOPMENT , 4(3), 149-53 CODEN: IEPRA6; ISSN: 0196-4321, 1965 * |
| INOKUCHI ET AL., J. ORG. CHEM., vol. 56, no. 7, 1991, pages 2416 - 2421, XP002408410 * |
| JOURNAL OF GEOPHYSICAL RESEARCH, [ATMOSPHERES] , 106(D12), 12541-12558 CODEN: JGRDE3; ISSN: 0148-0227, 2001 * |
| KULA ET AL., POLISH JOURNAL OF CHEMISTRY, vol. 68, 1994, Poland, pages 1699 - 1706, XP001247964 * |
| LEWIS ET AL, JOURNAL OF CHEMICAL AND ENGINEERING DATA, vol. 14, no. 3, 1969, pages 401 - 402, XP002409743 * |
| LIU ET AL., OPPI BRIEFS, vol. 29, no. 4, 1997, pages 473 - 476, XP001248167 * |
| LIU ET AL., TETRAHEDRON LETTERS, vol. 45, 2004, pages 6097 - 6100, XP002408411 * |
| MOGLIONI ET AL., J. ORG. CHEM., vol. 65, 2000, pages 3934 - 3940, XP002408413 * |
| OVERSIKI FINSKA VETENSKAPS SOC. , 57 [A](NO. 7), 19 PP., 1914 * |
| SCHENONE ET AL, BOLL. CHIM. FARM., vol. 110, 1971, Italy, pages 380 - 384, XP009075215 * |
| SCHENONE, P. ET AL.: "Aminoalcoli derivati dal ciclobutano", IL FARMACO, vol. 25, no. 7, 1970, Italy, pages 533 - 541, XP009075142 * |
| TETRAHEDRON , 48(10), 1927-42 CODEN: TETRAB; ISSN: 0040-4020, 1992 * |
| TETRAHEDRON LETTERS , 39(21), 3593-3596 CODEN: TELEAY; ISSN: 0040-4039, 1998 * |
| TETRAHEDRON: ASYMMETRY , 14(23), 3773-3778 CODEN: TASYE3; ISSN: 0957-4166, 2003 * |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9914705B2 (en) | 2008-04-25 | 2018-03-13 | Laboratorios Del Dr. Esteve, S.A. | 1-aryl-3-aminoalkoxy pyrazoles as sigma ligands enhancing analgesic effect of opioids and attenuating the dependency thereof |
| CN106924264A (en) * | 2010-02-04 | 2017-07-07 | 埃斯特韦实验室有限公司 | Sigma ligands for preventing and/or treating postoperative pain |
| WO2011095584A1 (en) * | 2010-02-04 | 2011-08-11 | Laboratorios Del Dr. Esteve, S.A. | Sigma ligands for use in the prevention and/or treatment of postoperative pain |
| US9844516B2 (en) | 2010-02-04 | 2017-12-19 | Laboratorios De Dr. Esteve | Sigma ligands for use in the prevention and/or treatment of post-operative pain |
| EP2353598A1 (en) * | 2010-02-04 | 2011-08-10 | Laboratorios Del. Dr. Esteve, S.A. | Sigma ligands for use in the prevention and/or treatment of postoperative pain |
| AU2011212389B2 (en) * | 2010-02-04 | 2016-05-12 | Laboratorios Del Dr. Esteve, S.A. | Sigma ligands for use in the prevention and/or treatment of postoperative pain |
| US9757358B2 (en) | 2010-02-04 | 2017-09-12 | Laboratorios Del Dr. Esteve, S.A. | Sigma ligands for potentiating the analgesic effect of opioids and opiates in post-operative pain and attenuating the dependency thereof |
| US9782483B2 (en) | 2010-05-21 | 2017-10-10 | Laboratories Del Dr. Esteve, S.A. | Sigma ligands for the prevention and/or treatment of emesis induced by chemotherapy or radiotherapy |
| US9789115B2 (en) | 2010-08-03 | 2017-10-17 | Laboratorios Del Dr. Esteve, S.A. | Use of sigma ligands in opioid-induced hyperalgesia |
| US9789117B2 (en) | 2011-05-18 | 2017-10-17 | Laboratorios Del Dr. Esteve, S.A. | Use of sigma ligands in diabetes type-2 associated pain |
| EP2567959A1 (en) | 2011-09-12 | 2013-03-13 | Sanofi | 6-(4-Hydroxy-phenyl)-3-styryl-1H-pyrazolo[3,4-b]pyridine-4-carboxylic acid amide derivatives as kinase inhibitors |
| US9464069B2 (en) | 2013-07-30 | 2016-10-11 | Laboratorios Del Dr. Esteve S.A. | 1,2-disubstituted cyclobutyl compounds |
| CN105408304A (en) * | 2013-07-30 | 2016-03-16 | 埃斯蒂维实验室股份有限公司 | 1,2-disubstituted cyclobutyl compounds |
| WO2015014816A1 (en) * | 2013-07-30 | 2015-02-05 | Laboratorios Del Dr. Esteve, S.A. | 1,2-disubstituted cyclobutyl compounds |
| EP2832720A1 (en) | 2013-07-30 | 2015-02-04 | Laboratorios Del. Dr. Esteve, S.A. | 1,2-disubstituted cyclobutyl compounds |
| US9931346B2 (en) | 2013-12-17 | 2018-04-03 | Laboratorios Del Dr. Esteve S.A. | Serotonin-norepinephrine reuptake inhibitors (SNRIs) and Sigma receptor ligands combinations |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2009545625A (en) | 2009-12-24 |
| EP2066648B1 (en) | 2017-04-12 |
| US20100105680A1 (en) | 2010-04-29 |
| CN101563332A (en) | 2009-10-21 |
| MX2009001315A (en) | 2009-05-28 |
| CA2660028A1 (en) | 2008-02-07 |
| ES2632610T3 (en) | 2017-09-14 |
| EP2066648A1 (en) | 2009-06-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2066648A1 (en) | Substituted dimethylcyclobutyl compounds, their preparation and use in medicaments | |
| RU2582338C2 (en) | Pyrazole compounds as sigma receptor inhibitors | |
| US8202872B2 (en) | Pyrazole derivatives as sigma receptor inhibitors | |
| US8293917B2 (en) | Pyrazole compounds as CCR1 antagonists | |
| EP1996580B1 (en) | Pyrrazole derivatives as sigma receptors antagonists | |
| KR20090087031A (en) | 1,2,4-triazole derivatives as sigma receptor inhibitors | |
| US20060106068A1 (en) | Sigma receptor inhibitors | |
| US7989485B2 (en) | Imidazole compounds having pharmaceutical activity towards the sigma receptor | |
| EP2941424B1 (en) | 1,2,3-triazole-4-amine derivatives for the treatment of sigma receptor related diseases and disorders | |
| EP1829875A1 (en) | Pyrazole derivatives as sigma receptor inhibitors | |
| US9617281B2 (en) | Imidazo[2,1-b]thiazole derivatives, their preparation and use as medicaments | |
| EP3365338B1 (en) | Substituted morpholine derivatives having activity against pain | |
| KR20170024026A (en) | Tricyclic triazolic compounds | |
| EP2817309A1 (en) | Substituted pyrazolo[1,5-a]pyridines, their preparation and use as medicaments | |
| EP1996548B1 (en) | Sigma receptor compounds | |
| JP2024028642A (en) | Indazole derivatives as cannabinoid receptor partial agonists | |
| WO2012072791A2 (en) | 2-(1-phenyl-4,5,6,7-tetrahydro-1h-indazol-4-yl)ethylamine derivatives useful as sigma receptor inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780036677.9 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07788185 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2660028 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009523253 Country of ref document: JP Ref document number: MX/A/2009/001315 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2007788185 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007788185 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |