WO2007130047A1 - Oled electron-injecting layer - Google Patents
Oled electron-injecting layer Download PDFInfo
- Publication number
- WO2007130047A1 WO2007130047A1 PCT/US2006/017555 US2006017555W WO2007130047A1 WO 2007130047 A1 WO2007130047 A1 WO 2007130047A1 US 2006017555 W US2006017555 W US 2006017555W WO 2007130047 A1 WO2007130047 A1 WO 2007130047A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- electron
- oled
- injecting layer
- substituted
- organic
- Prior art date
Links
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/17—Carrier injection layers
- H10K50/171—Electron injection layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
- H10K50/165—Electron transporting layers comprising dopants
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/50—Oxidation-reduction potentials, e.g. excited state redox potentials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2102/00—Constructional details relating to the organic devices covered by this subclass
- H10K2102/10—Transparent electrodes, e.g. using graphene
- H10K2102/101—Transparent electrodes, e.g. using graphene comprising transparent conductive oxides [TCO]
- H10K2102/103—Transparent electrodes, e.g. using graphene comprising transparent conductive oxides [TCO] comprising indium oxides, e.g. ITO
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/321—Metal complexes comprising a group IIIA element, e.g. Tris (8-hydroxyquinoline) gallium [Gaq3]
- H10K85/324—Metal complexes comprising a group IIIA element, e.g. Tris (8-hydroxyquinoline) gallium [Gaq3] comprising aluminium, e.g. Alq3
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/621—Aromatic anhydride or imide compounds, e.g. perylene tetra-carboxylic dianhydride or perylene tetracarboxylic di-imide
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
Definitions
- the present invention relates to an organic light-emitting device (OLED) in which an organic material having a strong oxidizing property is used in an electron-injecting layer of the device.
- OLED organic light-emitting device
- a typical OLED includes two electrodes and one organic EL unit disposed between the two electrodes.
- the organic EL unit commonly includes an organic hole-transporting layer (HTL), organic light-emitting layer (LEL), and an organic electron-transporting layer (ETL).
- One of the electrodes is the anode, which is capable of injecting positive charges (holes) into the HTL of the EL unit, and the other electrode is the cathode, which is capable of injecting negative charges (electrons) into the ETL of the EL unit.
- the anode is biased with a certain positive electrical potential relative to the cathode, holes injected from the anode and electrons injected from the cathode can recombine and emit light from the LEL.
- At least one of the electrodes is optically transmissive, and the emitted light can be seen through the transmissive electrode.
- HIL hole-injecting layer
- EIL electron-injecting layer
- EBL electron-blocking layer
- HBL hole-blocking layer
- the commonly used ETL or EIL in OLEDs includes the ETM of tris(8-hydroxyquinoline)aluminum (AIq), 4,7-diphenyl-l,10 ⁇ phenanthroline (Bphen), 2,9-dimethyl-4,7-diphenyl- 1 , 10-phenanthroline (BCP), and 2,2'-[l,l '-biphenyl]-4,4'-diylbis[4,6-(p-tolyl)-l,3,5-triazine] (TRAZ).
- AIq tris(8-hydroxyquinoline)aluminum
- Bphen 4,7-diphenyl-l,10 ⁇ phenanthroline
- BCP 2,9-dimethyl-4,7-diphenyl- 1 , 10-phenanthroline
- TRAZ 2,2'-[l,l '-biphenyl]-4,4'-diylbis[4,6-(p-tolyl)-l,3,5-triazine
- the selection of practically useful organic ETMs in OLEDs is limited to those having a reduction potential less than -1.0 V vs. a Saturated Calomel Electrode (SCE) or having a LUMO less than 3.3 eV below the vacuum energy level (E vac ).
- SCE Saturated Calomel Electrode
- E vac vacuum energy level
- an organic light-emitting device comprising: a) an anode; b) a light-emitting layer disposed over the anode; c) a first electron-injecting layer disposed over the light- emitting layer, wherein the first electron-injecting layer includes at least one organic host material having a reduction potential less than -1.0 V vs. a Saturated Calomel Electrode and at least one dopant material capable of reducing the organic host material; d) a second electron-injecting layer disposed in contact with the first electron-injecting layer, wherein the second electron-injecting layer includes at least one organic material having a reduction potential greater than -1.0 V vs.
- OLED organic light-emitting device
- the present invention makes use of a second electron-injecting layer including an organic material having a reduction potential greater than -1.0 eV in direct contact with the cathode and forming a stable interface between the cathode and this layer with no electron injection barrier from the cathode into the electron-injecting layer. It is an advantage of the present invention that the OLED with the second electron-injecting layer can not only increase the lifetime but also reduce the voltage rise during operation, which is useful for making high quality active matrix OLED display and other electronic devices. It is another advantage of the present invention that a stable metal, such as Ag, Au, Al, and Cu, can be used to form the cathode. These stable metals do not readily react with water or oxygen to form dark spots in the OLEDs.
- FIG. 1 shows a cross-sectional view of a prior art OLED
- FIG. 2 shows a cross-sectional view of one embodiment of an
- FIG. 3 shows a cross-sectional view of another embodiment of an OLED prepared in accordance with the present invention.
- FIG. 4 shows a cross-sectional view of yet another embodiment of an OLED prepared in accordance with the present invention.
- FIG. 5 shows a cross-sectional view of yet another embodiment of an OLED prepared in accordance with the present invention.
- FIG. 6 is a graph showing the normalized luminance vs. operational time of a group of OLEDs tested at room temperature and at 80 mA/cm
- FIG. 7 is a graph showing the drive voltage vs. operational time of the group of OLEDs tested at room temperature and at 80 mA/cm 2 ;
- FIG. 8 is a graph showing the normalized luminance vs. operational time of another group of OLEDs tested at room temperature and at 80 mA/cm ; and FIG. 9 is a graph showing the drive voltage vs. operational time of the other group of OLEDs tested at room temperature and at 80 mA/cm 2 .
- FIGS. 1-5 are not to scale since the thickness differences of various layers are too great to permit depiction to scale.
- OLED 100 includes substrate 110, anode 120, HIL 130, HTL 140, LEL 150, ETL 160, EIL 170, and cathode 180 (HIL 130, HTL 140, LEL 150, ETL 160, EIL 170, and cathode 180 (HIL 130, HTL 140, LEL 150, ETL 160, and EIL 170 form an organic EL unit in between the anode 120 and cathode 180).
- OLED 100 is externally connected to a voltage/current source 192 through electrical conductors 191.
- OLED 100 is operated by applying an electric potential produced by the voltage/current source 192 between the pair of contact electrodes, anode 120, and cathode 180.
- OLED 200 includes substrate 110, anode 120, HIL 130, HTL 140, LEL 150, ETL 160, the 1 st EIL 271, the 2 nd EIL 272, and cathode 180.
- OLED 200 is the same as OLED 100 except that OLED 200 has two EILs (EIL 271 and EIL 272).
- OLED 200 is also externally connected to a voltage/current source 192 through electrical conductors 191. Shown in FIGS. 3, 4, and 5 are OLED 300, OLED 400, and OLED 500, respectively, which are some other embodiments of OLEDs prepared in accordance with the present invention.
- OLED 300 is the same as OLED 200 except that there is no HIL 130 in OLED 300
- OLED 400 in FIG. 4 is the same as OLED 200 except that there is no ETL 160 in OLED 400
- OLED 500 in FIG. 5 is the same as OLED 200 except that there is no HIL 130 nor ETL 160 in OLED 500.
- the substrate can alternatively be located adjacent to the cathode, or the substrate can actually constitute the anode or cathode.
- the total combined thickness of the organic layers is preferably less than 500 ran.
- the present invention can be employed in the making of most OLED device configurations. These include very simple structures including a single anode and cathode to more complex devices, such as passive matrix displays including orthogonal arrays of anodes and cathodes to form pixels, and active matrix displays where each pixel is controlled independently, for example, with thin film transistors (TFTs).
- TFTs can be constructed using amorphous silicon, low temperature polycrystalline silicon, single crystal silicon, other inorganic semiconductors, or organic semiconductor materials.
- Substrate 110 can be an organic solid, an inorganic solid, or includes organic and inorganic solids that provides a supporting backplane to hold the OLED.
- Substrate 110 can be rigid or flexible and can be processed as separate individual pieces, such as sheets or wafers, or as a continuous roll.
- Typical substrate materials include glass, plastic, metal, ceramic, semiconductor, metal oxide, semiconductor oxide, or semiconductor nitride, or combinations thereof.
- Substrate 110 can be a homogeneous mixture of materials, a composite of materials, or multiple layers of materials.
- Substrate 110 can also be a backplane containing thin-film transistor (TFT) circuitry commonly used for preparing OLED display, e.g. an active matrix low temperature polysilicon TFT substrate.
- TFT thin-film transistor
- the substrate 110 can either be light transmissive or opaque, depending on the intended direction of light emission.
- the light transmissive property is desirable for viewing the EL emission through the substrate.
- Transparent glass or plastic is commonly employed in such cases.
- the transmissive characteristic of the bottom support is immaterial, and therefore can be light transmissive, light absorbing, or light reflective.
- Substrates for use in this case include, but are not limited to, glass, plastic, semiconductor materials, ceramics, and circuit board materials, or any others commonly used in the formation of OLEDs, which can be either passive matrix devices or active matrix devices.
- Anode 120 is formed over substrate 110.
- the anode should be transparent or substantially transparent to the emission of interest.
- the transmissive characteristics of the anode material are immaterial and any conducting or semiconducting material can be used, regardless if it is transparent, opaque, or reflective.
- Desired anode materials can be deposited by any suitable way such as thermal evaporation, sputtering, chemical vapor deposition, or electrochemical means. Anode materials can be patterned using well known photolithographic processes.
- the material for use to form anode 120 can be selected from inorganic materials, or organic materials, or combination thereof.
- the anode 120 can contain the element material selected from aluminum, silver, gold, copper, zinc, indium, tin, titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, indium, nickel, palladium, platinum, silicon, or germanium, or combinations thereof.
- the anode 120 can also contain a compound material, such as a conducting or semiconducting compound.
- the conducting or semiconducting compound can be selected from the oxides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof.
- the conducting or semiconducting compound can be selected from the sulfides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof.
- the conducting or semiconducting compound can be selected from the selenides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof.
- the conducting or semiconducting compound can be selected from the tellurides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof.
- the conducting or semiconducting compound can be selected from the nitrides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof.
- the conducting or semiconducting compound can be selected from indium tin oxide, tin oxide, aluminum-doped zinc oxide, indium-doped zinc oxide, magnesium indium oxide, nickel tungsten oxide, zinc sulfide, zinc selenide, or gallium nitride, or the combination thereof.
- hole-injecting layer 130 between anode 120 and hole-transporting layer 140.
- the hole-injecting material can serve to improve the film formation property of subsequent organic layers and to facilitate injection of holes into the hole-transporting layer.
- Suitable materials for use in the hole-injecting layer include, but are not limited to, porphyrinic compounds as described in U.S. Patent 4,720,432, plasma-deposited fluorocarbon polymers as described in U.S.
- Patents 6,127,004, 6,208,075 and 6,208,077 some aromatic amines, for example, m-MTDATA (4,4',4"-tris[(3-methylphenyl)phenyl- amino]triphenylamine), and inorganic oxides including vanadium oxide (VOx), molybdenum oxide (MoOx), and nickel oxide (NiOx).
- m-MTDATA (4,4',4"-tris[(3-methylphenyl)phenyl- amino]triphenylamine
- inorganic oxides including vanadium oxide (VOx), molybdenum oxide (MoOx), and nickel oxide (NiOx).
- Alternative hole-injecting materials reportedly useful in organic EL devices are described in EP 0 891 121 Al and EP 1 029 909 Al.
- Hexaazatriphenylene derivatives are also useful HIL materials, as described in U.S. Patent 6,720,573.
- a p-type doped organic material typically includes a hole-transporting material such as an aromatic amine (see below) that is doped with an electron accepting dopant.
- Such dopants can include, for example, 2,3,5,6-tetrafluoro-7,7,8,8-tetracyanoquinodimethane (F4-TCNQ) and other derivatives of 7,7,8, 8-tetracyanoquinodimethane (TCNQ), and inorganic oxidizing agents such as iodine, FeC13, FeF3, SbCl5, some other metal chlorides, and some other metal fluorides.
- the p-type doped concentration is preferably in the range of 0.01-20 vol. %.
- Such layers materials are further described in, for example, U.S. Patents 5,093,698, 6,423,429, and 6,597,012.
- the thickness of the HIL can be in the range of from 0.1 nm to 200 nm, preferably, in the range of from 0.5 nm to 150 nm.
- the HTL 140 contains at least one hole-transporting material such as an aromatic tertiary amine, where the aromatic tertiary amine is understood to be a compound containing at least one trivalent nitrogen atom that is bonded only to carbon atoms, at least one of which is a member of an aromatic ring.
- the aromatic tertiary amine can be an arylamine, such as a monoarylamine, diarylamine, triarylamine, or a polymeric arylamine. Exemplary monomeric triarylamines are illustrated by Klupfel et al. in U.S. Patent 3,180,730.
- Suitable triarylamines substituted with one or more vinyl radicals or at least one active hydrogen-containing group are disclosed by Brantley, et al. in U.S. Patents 3,567,450 and 3,658,520.
- a more preferred class of aromatic tertiary amines includes compounds that include at least two aromatic tertiary amine moieties as described by Van Slyke and Tang et al. in U.S. Patents 4,720,432 and 5,061,569.
- the HTL can be formed of a single or a mixture of aromatic tertiary amine compounds.
- Illustrative of useful aromatic tertiary amines are the following:
- NPB 4,4'-Bis[N-(l -naphthyl)-N-phenylamino]biphenyl
- Another class of useful hole-transporting materials includes polycyclic aromatic compounds as described in EP 1 009 041. Some hole-injecting materials described in EP 0 891 121 Al and EP 1 029 909 Al can also make useful hole-transporting materials.
- polymeric hole-transporting materials can be used including poly(N-vinylcarbazole) (PVK), polythiophenes, polypyrrole, polyaniline, and copolymers including poly(3, 4-ethylenedioxy- thiophene)/poly(4-styrenesulfonate) also called PEDOT/PSS.
- the thickness of the HTL can be in the range of from 5 nm to 200 nm, preferably, in the range of from 10 nm to 150 nm.
- the LEL 150 can include a luminescent fluorescent or phosphorescent material where electroluminescence is produced as a result of electron-hole pair recombination in this layer.
- the light-emitting layer can include a single material, but more commonly contains at least one host material doped with at least one guest emitting material or materials where light emission comes primarily from the emitting materials and can be of any color. This guest emitting material is often referred to as a luminescent dopant material.
- the host materials in the light-emitting layer can be an electron-transporting material as defined below, a hole-transporting material as defined above, or another material or combination of materials that support hole-electron recombination.
- the emitting material is typically chosen from highly fluorescent dyes and phosphorescent compounds, e.g., transition metal complexes as described in WO 98/55561 Al, WO 00/18851 Al, WO 00/57676 Al, and WO 00/70655.
- Emitting materials are typically incorporated at 0.01 to 20 % by volume of the host material.
- the host and emitting materials can be small nonpolymeric molecules or polymeric materials including polyfluorenes and polyvinylarylenes, e.g., poly(p-phenylenevinylene), PPV.
- small molecule emitting materials can be molecularly dispersed into a polymeric host, or the emitting materials can be added by copolymerizing a minor constituent into a host polymer.
- An important relationship for choosing an emitting material is a comparison of the electron energy bandgap that is defined as the energy difference between the highest occupied molecular orbital and the lowest unoccupied molecular orbital of the molecule.
- the bandgap of the dopant is smaller than that of the host material.
- phosphorescent emitters including materials that emit from a triplet excited state, i.e., so-called "triplet emitters" it is also important that the triplet energy level of the host be high enough to enable energy transfer from host to emitting material.
- Host and emitting materials known to be of use include, but are not limited to, those disclosed in U.S. Patents 4,768,292, 5,141,671, 5,150,006, 5,151,629, 5,405,709, 5,484,922, 5,593,788, 5,645,948, 5,683,823, 5,755,999, 5,928,802, 5,935,720, 5,935,721, 6,020,078, 6,475,648, 6,534,199, 6,661,023, U.S. Patent Application Publications 2002/0127427 Al, 2003/0198829 Al, 2003/0203234 Al, 2003/0224202 Al, and 2004/0001969 Al.
- oxine 8-hydroxyquinoline
- chelated oxinoid compound metal complexes of 8-hydroxyquinoline (oxine), otherwise known as chelated oxinoid compound, and similar derivatives constitute one class of useful host compounds capable of supporting electroluminescence.
- useful chelated oxinoid compounds are the following:
- CO-I Aluminum trisoxine [alias, tris(8-qumolmolato)aluminum(IIi)]
- CO-2 Magnesium bisoxine [alias, bis(8-quinolinolato)magnesium(II)];
- CO-3 Bis[benzo ⁇ f ⁇ -8-quinolinolato]zinc (II);
- CO-4 Bis(2-methyl-8-quinolinolato)aluminum(III)- ⁇ -oxo-bis(2-methyl-8- quinolinolato) aluminum(III);
- CO-5 Indium trisoxine [alias, tris(8-quinolinolato)indium]
- CO-6 Aluminum tris(5-methyloxine) [alias, tris(5-methyl-8-quinolinolato) aluminum(III)]
- CO-7 Lithium oxine [alias, (8-quinolinolato)lithmm(I)]
- CO-8 Gallium oxine [alias, tris(8-quinolinolato)gallium(III)]
- CO-9 Zirconium oxine [alias, tetra(8-quinolinolato)zirconium(IV)]
- Another class of useful host materials includes derivatives of anthracene, such as those described in U.S.
- Some examples include derivatives of 9,10-dinaphthylanthracene and 9-naphthyl-10- phenylanthracene.
- Other useful classes of host materials include distyrylarylene derivatives as described in U.S. Patent 5,121,029, and benzazole derivatives, for example, 2, 2', 2"-(l,3,5-phenylene)tris[l-phenyl-lH-benzimidazole],
- Suitable host materials for phosphorescent dopants should be selected so that a triplet exciton can be transferred efficiently from the host material to the phosphorescent material. For this transfer to occur, it is a highly desirable condition that the excited state energy of the phosphorescent material be lower than the difference in energy between the lowest triplet state and the ground state of the host. However, the bandgap of the host should not be chosen so large as to cause an unacceptable increase in the drive voltage of the OLEDs. Suitable host materials are described in WO 00/70655 A2, WO 01/39234 A2, WO 01/93642 Al, WO 02/074015 A2, WO 02/15645 Al, and U.S. Patent Application Publication 2002/0117662 Al .
- Suitable hosts include certain aryl amines, triazoles, indoles and carbazole compounds.
- Examples of desirable hosts are 4,4'-N,N'-dicarbazole-bi ⁇ henyl (CBP), 2,2'-dimethyl-4,4'-N,N'-dicarbazole- biphenyl, m-(N,N'-dicarbazole)benzene, and poly(N-vinylcarbazole), including their derivatives.
- Desirable host materials are capable of forming a continuous film.
- the light-emitting layer can contain more than one host material in order to improve the device's film morphology, electrical properties, light emission efficiency, and lifetime.
- Mixtures of electron-transporting and hole-transporting materials are known as useful hosts.
- mixtures of the above listed host materials with hole-transporting or electron-transporting materials can make suitable hosts.
- Useful fluorescent dopants include, but are not limited to, derivatives of anthracene, tetracene, xanthene, perylene, rubrene, coumarin, rhodamine, and quinacridone, dicyanomethylenepyran compounds, thiopyran compounds, polymethine compounds, pyrylium and thiapyrylium compounds, fluorene derivatives, periflanthene derivatives, indenoperylene derivatives, bis(azinyl)amine boron compounds, bis(azinyl)methane boron compounds, derivatives of distryrylbenzene and distyrylbiphenyl, and carbostyryl compounds.
- derivatives of distyrylbenzene particularly useful are those substituted with diarylamino groups, informally known as distyrylamines.
- Examples of useful phosphorescent dopants that can be used in light-emitting layers of this invention include, but are not limited to, those described in WO 00/57676 Al, WO 00/70655, WO 01/41512 Al, WO 02/15645 Al, WO 01/93642 Al, WO 01/39234 A2, WO 02/071813 Al, WO 02/074015 A2, U.S.
- Useful phosphorescent dopants include transition metal complexes, such as iridium and platinum complexes. hi some cases it is useful for one or more of the LELs within an EL unit to emit broadband light, for example white light. Multiple dopants can be added to one or more layers in order to produce a white-emitting OLED, for example, by combining blue- and yellow-emitting materials, cyan- and red- emitting materials, or red-, green-, and blue-emitting materials.
- White-emitting devices are described, for example, in EP 1 187 235, EP 1 182 244, U.S.
- the host for one light-emitting layer is a hole-transporting material.
- the host for one light-emitting layer is a hole-transporting material.
- emitting dopants can be added to the HTL 140, thereby enabling HTL 140 to serve as a host.
- the thickness of each LEL can be in the range of from 5 nm to 80 run, preferably, in the range of from 10 nm to 40 nm.
- Preferred organic materials for use in forming the ETL 160 are metal chelated oxinoid compounds, including chelates of oxine itself, also commonly referred to as 8-quinolinol or 8-hydroxyquinoline. Such compounds help to inject and transport electrons, exhibit high levels of performance, and are readily deposited to form thin films.
- Exemplary oxinoid compounds have been listed above from CO-I to CO-9 (the oxinoid compounds can be used as both the host material in LEL 150 and the electron-transporting material in ETL 160).
- electron-transporting materials include various butadiene derivatives as disclosed in U.S. Patent 4,356,429 and various heterocyclic optical brighteners as described in U.S. Patent 4,539,507. Benzazoles, oxadiazoles, triazoles, pyridinethiadiazoles, triazines, phenanthrolme derivatives, and some silole derivatives are also useful electron-transporting materials.
- the 1 st EIL 271 as shown in FIGS. 2-5 is an n-type doped layer containing at least one electron-transporting material as a host material and at least one n-type dopant material (this EIL can also be called an n-type doped 1 st EIL 271).
- the reduction potential of one of the organic host materials in the 1 st EIL 271 is less than -1.0 V vs. SCE and one of the dopant materials is capable of reducing one of the organic host materials.
- the term " n-type doped layer" means that this layer has semiconducting properties after doping, and the electrical current through this layer is substantially carried by the electrons.
- the host material is capable of supporting electron injection and electron transport.
- the electron-transporting materials used in ETL 160 represent a useful class of host materials for the n-type doped 1 st EIL 271.
- Preferred materials are metal chelated oxinoid compounds, including chelates of oxine itself (also commonly referred to as 8-quinolinol or 8-hydroxyquinoline), such as tris(8-hydroxyquinoline)- aluminum (AIq).
- Other materials include various butadiene derivatives as disclosed by Tang in U.S. Patent 4,356,429, various heterocyclic optical brighteners as disclosed by Van Slyke and Tang et al. in U.S. Patent 4,539,507, triazines, hydroxyquinoline derivatives, benzazole derivatives, and phenanthroline derivatives.
- Silole derivatives such as 2,5-bis(2',2"-bipridin-6-yl)-l,l-dimethyl- 3,4-diphenyl silacyclopentadiene are also useful host organic materials.
- the combination of the aforementioned host materials is also useful to form the n-typed doped EIL 170.
- the host material in the n-type doped EIL 170 includes AIq, 4,7-di ⁇ henyl-l,10- ⁇ henanthroline (Bphen), 2,9-dimethyl- 4,7-diphenyl-l,10-phenanthroline (BCP), 2,2'-[l,l '-biphenyl]-4,4'-diylbis[4,6- (p-tolyl)-l,3,5-triazine] (TRAZ), or rubrene, or combinations thereof.
- Bphen 4,7-di ⁇ henyl-l,10- ⁇ henanthroline
- BCP 2,9-dimethyl- 4,7-diphenyl-l,10-phenanthroline
- TRAZ 2,2'-[l,l '-biphenyl]-4,4'-diylbis[4,6- (p-tolyl)-l,3,5-triazine]
- rubrene or combinations thereof.
- Both 1 st EIL 271 and ETL 160 in the OLEDs can use the same or different material.
- the n-type dopant in the n-type doped 1 st EIL 271 is selected from alkali metals, alkali metal compounds, alkaline earth metals, or alkaline earth metal compounds, or combinations thereof.
- the term "metal compounds" includes organometallic complexes, metal organic salts, and inorganic salts, oxides and halides.
- metal containing n-type dopants Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Sm, Eu, Tb, Dy, or Yb, and their compounds, are particularly useful.
- the materials used as the n-type dopants in the n-type doped EIL 170 also include organic reducing agents with strong electron-donating properties.
- strong electron-donating properties it is meant that the organic dopant should be able to donate at least some electronic charge to the host to form a charge transfer complex with the host.
- organic molecules include bis(ethylenedithio)-tetrathiafulvalene (BEDT-TTF), tefrathiafurvalene (TTF), and their derivatives.
- the dopant can be any of the above or also a material molecularly dispersed or copolymerized with the host as a minor component.
- the n-type dopant in the n-type doped 1 st EIL 271 includes Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Nd, Sm, Eu, Tb, Dy, or Yb, or combinations thereof.
- the n-type doped concentration is preferably in the range of 0.01 -20% by volume of this layer.
- the thickness of the n-type doped 1 st EIL 271 is typically less than 200 nm, and preferably in the range of 2 to 150 nm.
- the 2 nd EIL 272 in the OLEDs as shown in FIGS. 2-5 is disposed in contact with the 1 st EIL 271, and includes at least one organic material having a reduction potential greater than -1.0 V vs. SCE, preferably, greater than -0.5 Vs. SCE.
- the organic materials having a reduction potential greater than -1.0 V vs. SCE, especially greater than -0.5 V vs. SCE, are promising n-type semiconducting materials with electron accepting property.
- the materials can provide effective electron transport in OLEDs.
- the reduction potential of the materials is greater than -0.5 V vs. SCE, the LUMO position of the material will be lower than 3.8 eV from the E vac . Therefore, it is possible to select the 2 nd EIL 272 according to the work function of the cathode 180 such that the LUMO position of the 2 nd EIL 272 is below or near the work function of the cathode 180.
- the 2 nd EIL 272 can also react with the first several monolayers of the cathode when the cathode layer is formed on top of the 2 n EIL 272.
- cathode 180 can have an improved contact with the 2 nd EIL 272 forming a stable interface between the 2 nd EIL 272 and the cathode 180.
- the 2 nd EIL 272 can be as thin as 1 nm. However, it can also be as thick as 200 nm. Preferably, the thickness of 2 nd EIL 272 is in the range of from 1 to 150 nm.
- the reduction potential of a substance can be conveniently obtained by cyclic voltammetry (CV) and it is measured vs. SCE.
- the measurement of the reduction potential of a substance can be as following: An electrochemical analyzer (for instance, a CHI660 electrochemical analyzer, made by CH Instruments, Inc., Austin, TX) is employed to carry out the electrochemical measurements. Both CV and Osteryoung square-wave voltammetry (SWV) can be used to characterize the redox properties of the substance.
- the GC electrode is polished with 0.05 ⁇ m alumina slurry, followed by sonication cleaning in deionized water twice and rinsed with acetone between the two water cleanings. The electrode is finally cleaned and activated by electrochemical treatment prior to use.
- a platinum wire can be used as the counter electrode and the SCE is used as a quasi-reference electrode to complete a standard 3 -electrode electrochemical cell.
- a mixture of acetonitrile and toluene (1:1 MeCN/toluene) or methylene chloride (MeCl 2 ) can be used as organic solvent systems.
- the testing solution is purged with high purity nitrogen gas for approximately 15 minutes to remove oxygen and a nitrogen blanket is kept on the top of the solution during the course of the experiments. All measurements are performed at an ambient temperature of 25 + 1°C. If the compound of interest has insufficient solubility, other solvents can be selected and used by those skilled in the art. Alternatively, if a suitable solvent system cannot be identified, the electron accepting material can be deposited onto the electrode and the reduction potential of the modified electrode can be measured. Several types of organic materials having a reduction potential greater than -1.0 V vs. SCE, especially greater than -0.5 V vs. SCE, can be used to form the 2 nd EIL 272 in the present invention.
- the organic material used in the 2 nd EIL 272 can be a chemical compound of Formula I wherein R 1 -R 4 represent hydrogen or substituents independently selected from the group including halo, nitrile (-CN), nitro (-NO 2 ), sulfonyl (-SO 2 R), sulfoxide (-SOR), trifluoromethyl (-CF 3 ), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein R 1 and R 2 , or R 3 and R 4 , combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
- the organic material used in the 2 nd EIL 272 can be a chemical compound of Formula Ia (2,3,5,6-tetrafluoro-7,7,8,8-tetracyano- quinodimethane [F 4 -TCNQ])
- the organic material used in the 2 nd EIL 272 can be a chemical compound of
- the organic material used in the 2 nd EIL 272 can be a chemical compound of Formula III
- R 1 -R 6 represent hydrogen or a substituent independently selected from the group including halo, nitrile (-CN), nitro (-NO 2 ), sulfonyl (-SO 2 R), sulfoxide (-SOR), trifiuoromethyl (-CF 3 ), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein R 1 and R 2 , R 3 and R 4 , or R 5 and R 6 , combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
- the organic material used in the 2 nd EIL 272 can be a chemical compound of Formula Ilia (hexaazatriphenylene hexacarbonitrile)
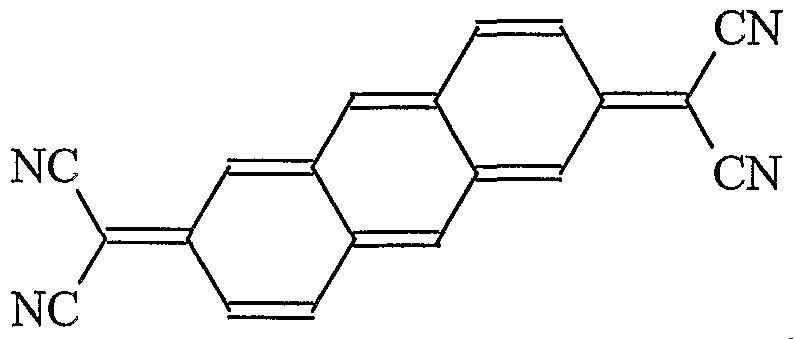
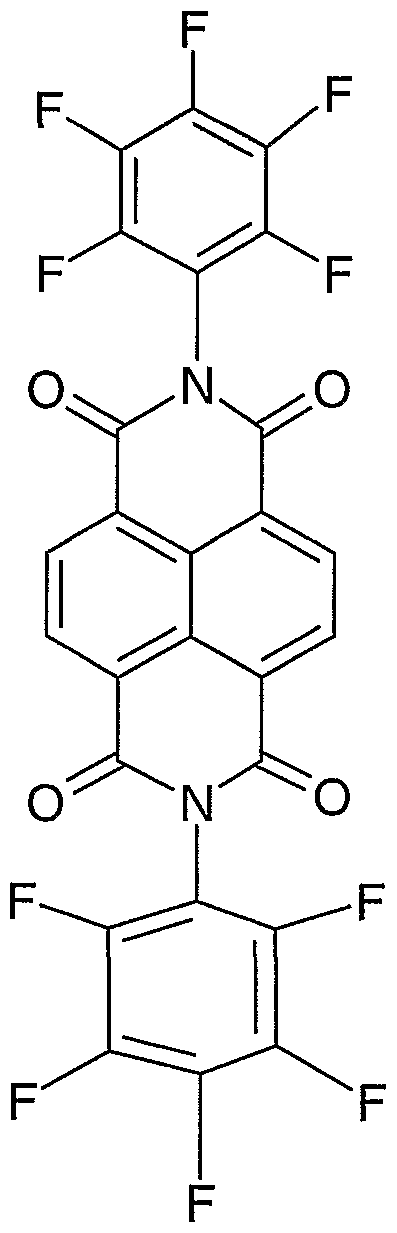
- Additional organic materials that can be used in the 2 nd EIL include diimide derivative of naphthalene tetracarboxylic diimide and of perylene tetracarboxylic diimide that are substituted with two or more fluoro groups, such as a chemical compound of
- organic materials suitable for use in the 2 nd EIL 272 not only include the compounds containing at least carbon and hydrogen, but also include metal complexes, e.g., transition metal complexes having organic ligands and organometallic compounds, as long as their reduction potentials are in the appropriate range.
- metal complexes e.g., transition metal complexes having organic ligands and organometallic compounds, as long as their reduction potentials are in the appropriate range.
- the organic materials used in the 2 n EIL 272 should preferably have an optical bandgap greater than 1.6 eV.
- the 2 nd EIL 272 can also be doped with at least one n-type dopant material having a work function less than 4.0 eV into the organic host material.
- the n-type dopant for use in the 2 nd EIL 272 is selected from alkali metals, alkali metal compounds, alkaline earth metals, or alkaline earth metal compounds, or combinations thereof.
- the class of metal containing n-type dopants Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Sm, Eu, Tb, Dy, or Yb, and their compounds, are particularly useful.
- the n-type doped concentration is preferably in the range of 0.01 -20% by volume of this layer.
- the n-type dopant used in the 2 nd EIL 272 can be the same or different from that used in the 1 st EIL 271.
- Additional layers such as exciton, electron and hole-blocking layers as taught in the art can be employed in devices of this invention.
- Hole-blocking layers are commonly used to improve efficiency of phosphorescent emitter devices, for example, as in U.S. Patent Application Publications
- Each of the layers in the organic EL units in the OLEDs can be formed from small molecule (or nonpolymeric) materials (including fluorescent materials and phosphorescent materials), polymeric LED materials, or inorganic materials, or combinations thereof.
- the cathode 180 can include nearly any conductive material. Desirable materials have effective film forming properties to ensure effective contact with the underlying organic layer, promote electron injection at low voltage, and have effective stability. Commonly used cathodes include a Mg: Ag alloy as described in U.S. Patent 4,885,221, a LiF/Al bilayer as described in U.S. Patent 5,677,572, Al, Ag, Au, Cu, or a combination thereof. Other useful cathodes include, but are not limited to, those disclosed in U.S. Patents 5,059,861, 5,059,862, and 6,140,763.
- the 2 nd EIL 272 includes the organic material having a reduction potential greater than -1.0 V vs. SCE, the deposited cathode can form a stable interface with the 2 nd EIL 272 resulting in less interface degradation during operation as well as less dark spots due to less reaction with water or oxygen during storage. It is preferable that the barrier to injection of electrons from the cathode material into the 2 nd EIL be no greater than 0.3 eV. For example, if the 2 nd EIL 272 includes an organic material having a reduction potential of -0.5 V vs. SCE, which means that the LUMO position is about -3.8 eV, the cathode can be then selected from materials whose work functions are equal to or smaller than 4.1 eV.
- the cathode When light emission is viewed through the cathode, the cathode should be transparent or nearly transparent. For such applications, metals should be thin or one should use transparent conductive oxides, or include these materials.
- Optically transparent cathodes have been described in more detail in U.S. Patents 4,885,211, 5,247,190, 5,703,436, 5,608,287, 5,837,391, 5,677,572, 5,776,622, 5,776,623, 5,714,838, 5,969,474, 5,739,545, 5,981,306, 6,137,223, 6,140,763, 6,172,459, 6,278,236, 6,284,393, and EP 1 076 368.
- Cathode materials are typically deposited by thermal evaporation, electron beam evaporation, ion sputtering, or chemical vapor deposition. When needed, patterning can be achieved through many well known methods including, but not limited to, through-mask deposition, integral shadow masking, for example as described in U.S. Patent 5,276,380 and EP 0 732 868, laser ablation, and selective chemical vapor deposition.
- tandem devices can be used in a so-called tandem device architecture, for example, as taught in U.S. Patents 6,337,492, 6,717,358, and U.S. Patent Application Publication 2003/0170491 Al .
- tandem devices have multiple electroluminescent units provided between an anode and a cathode, typically with connector layer between units to promote charge production and injection into the electroluminescent units.
- the organic materials mentioned above are suitably deposited through a vapor-phase method such as sublimation, but can be deposited from a fluid, for example, from a solvent with an optional binder to improve film formation. If the material is a polymer, solvent deposition is useful but other methods can be used, such as sputtering or thermal transfer from a donor sheet.
- the material to be deposited by sublimation can be vaporized from a sublimation "boat" often including a tantalum material, e.g., as described in U.S. Patent 6,237,529, or can be first coated onto a donor sheet and then sublimed in closer proximity to the substrate.
- Layers with a mixture of materials can use separate sublimation boats or the materials can be premixed and coated from a single boat or donor sheet. Patterned deposition can be achieved using shadow masks, integral shadow masks (U.S. Patent 5,294,870), spatially defined thermal dye transfer from a donor sheet (U.S. Patents 5,688,551, 5,851,709 and 6,066,357), and inkjet method (U.S. Patent 6,066,357).
- Most OLED devices are sensitive to moisture or oxygen, or both, so they are commonly sealed in an inert atmosphere such as nitrogen or argon.
- a protective cover can be attached using an organic adhesive, a metal solder, or a low melting temperature glass.
- a getter or desiccant is also provided within the sealed space.
- Useful getters and desiccants include, alkali and alkaline metals, alumina, bauxite, calcium sulfate, clays, silica gel, zeolites, alkaline metal oxides, alkaline earth metal oxides, sulfates, or metal halides and perchlorates.
- Methods for encapsulation and desiccation include, but are not limited to, those described in U.S. Patent 6,226,890.
- barrier layers such as SiOx, Teflon, and alternating inorganic/polymeric layers are known in the art for encapsulation.
- OLED devices of this invention can employ various well known optical effects in order to enhance its properties if desired. This includes optimizing layer thicknesses to yield maximum light transmission, providing dielectric mirror structures, replacing reflective electrodes with light-absorbing elect-odes, providing anti glare or antireflection coatings over the display, providing a polarizing medium over the display, or providing colored, neutral density, or color conversion filters in functional relationship with the light emitting areas of the display. Filters, polarizers, and antiglare or antireflection coatings can also be provided over a cover or as part of a cover.
- the OLED device can have a microcavity structure. In one useful example, one of the metallic electrodes is essentially opaque and reflective; the other one is reflective and semitransparent.
- the reflective electrode is preferably selected from Au, Ag, Mg, Ca, or alloys thereof. Because of the presence of the two reflecting metal electrodes, the device has a microcavity structure. The strong optical interference in this structure results in a resonance condition. Emission near the resonance wavelength is enhanced and emission away from the resonance wavelength is depressed.
- the optical path length can be tuned by selecting the thickness of the organic layers or by placing a transparent optical spacer between the electrodes.
- an OLED device of this invention can have ITO spacer layer placed between a reflective anode and the organic EL media, with a semitransparent cathode over the organic EL media.
- OLED displays or the other electronic devices can include a plurality of the OLEDs as described above.
- EXAMPLES The following examples are presented for a further understanding of the present invention.
- the reduction potentials of the materials were measured using an electrochemical analyzer (CHI660 electrochemical analyzer, made by CH Instruments, Inc., Austin, TX) with the method as discussed before.
- CHI660 electrochemical analyzer made by CH Instruments, Inc., Austin, TX
- the thickness of the organic layers and the doping concentrations were controlled and measured in situ using calibrated thickness monitors (INFICON IC/5 Deposition Controller, made by Inf ⁇ con Inc., Syracuse, NY).
- Example 1 (Comparative) The preparation of a conventional OLED is as follows: A ⁇ 1.1 mm thick glass substrate coated with a transparent ITO conductive layer was cleaned and dried using a commercial glass scrubber tool.
- the thickness of ITO is about 42 nm and the sheet resistance of the ITO is about 68 ⁇ /square.
- the ITO surface was subsequently treated with oxidative plasma to condition the surface as an anode.
- a layer of CFx, 1 nm thick, was deposited on the clean ITO surface as the anode buffer layer by decomposing CHF 3 gas in an RF plasma treatment chamber.
- the substrate was then transferred into a vacuum deposition chamber for deposition of all other layers on top of the substrate.
- the following layers were deposited in the following sequence by evaporation from a heated boat under a vacuum of approximately 10 "6 Torn 1.
- HIL hexaazatriphenylene hexacarbonitrile
- HTL 65 nm thick, composed of 4,4'-bis[N-(l -na ⁇ hthyl)-N- phenylamino]biphenyl
- a LEL 30 nm thick, composed of tris(8-hydroxyquinoline)- aluminum (AIq) doped with 1.0 vol% 10-(2-benzothiazolyl)-l,l,7,7-tetramethyl- 2,3,6,7-tetrahydro-lH,5H,l lH(l)benzopyrano(6,7,8-ij)quinolizin-l 1-one (C545T); and d) an EIL, 30 nm thick, composed of AIq doped with about 1.2 vol% lithium.
- Cathode approximately 210 nm thick, composed of Mg:Ag (formed by co- evaporation of about 95 vol% Mg and 5 vol% Ag).
- the device was transferred from the deposition chamber into a dry box (made by VAC Vacuum Atmosphere Company, Hawthorne, CA) for encapsulation.
- the OLED has an emission area of 10 mm 2 .
- the reduction potentials of HAT-CN and AIq were measured as about -0.01 V and -1.77 V vs. SCE in the 1:1 MeCN/toluene organic solvent system, respectively.
- This conventional OLED requires a drive voltage of about 5.0 V to pass 20 mA/cm . Under this test condition, the device has a luminance of
- Example 2 Another OLED was constructed as the same as that in Example 1, except that the 30-nm-thick Li-doped AIq layer (the EIL formed as layer d) in Example 1 was replaced by a 30-nm-thick HAT-CN layer as an EIL Example 2.
- This OLED requires a drive voltage of about 5.4 V to pass 20 mA/cm 2 .
- the HAT-CN layer having a reduction potential greater than -1.0 V vs. SCE cannot be effectively used as an EIL in this device configuration.
- Example 3 Another OLED was constructed as the same as that in Example I 5 except that the 30-nm-thick Li-doped AIq layer (the EIL, layer d) in Example 1 was replaced by a 30-nm-thick HAT-CN layer doped with about 1.2 vol% lithium as an EIL in Example 3.
- the 30-nm-thick Li-doped AIq layer (the EIL, layer d) in Example 1 was replaced by a 30-nm-thick HAT-CN layer doped with about 1.2 vol% lithium as an EIL in Example 3.
- the layer structure of the EL unit is as follows: a) an HTL, 75 nm thick, composed of NPB; b) a LEL, 30 nm thick, composed of AIq doped with 1.0 vol% C545T; and c) an ETL, 30 nm thick, composed of AIq.
- This conventional OLED requires a drive voltage of about 6.6 V to pass 20 mA/cm .
- the device has a luminance of 1977 cd/m , and a luminous efficiency of about 9.9 cd/A. Its emission peak is at 522 nm.
- the operational lifetime was measured as T 50 (RT@80 mA/cm 2 ) (i.e. the time at which the luminance has fallen to 50% of its initial value after being operated at room temperature and at 80 mA/cm 2 ). Its T 50 (RT@80 mA/cm 2 ) is about 130 hours.
- the average rate of the voltage rise during the lifetime testing is about 17 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm 2 , are shown in FIGS. 6 and 7, respectively.
- Example 5 Comparative
- Example 4 Another OLED was constructed as the same as that in Example 4, except that the layer c in Example 4 was replaced by: c) an EIL, 30 nm thick, composed of AIq doped with about 1.2 vol% lithium.
- This OLED requires a drive voltage of about 4.6 V to pass 20 mA/cm .
- the device has a luminance of 2008 cd/m , and a luminous efficiency of about 10.0 cd/A. Its emission peak is at 522 nm.
- the operational lifetime, measured as T 50 (RT@80 mA/cm 2 ), is about 246 hours.
- the average rate of the voltage rise during the lifetime testing is about 5.0 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm 2 , are shown in FIGS. 6 and 7, respectively.
- Example 6 (Inventive) An OLED in accordance with the present invention was constructed as the same as that in Example 4, except that the layer c in Example 4 was replaced by: c) the 1 st EIL, 29 nm thick, composed of AIq doped with about 1.2 vol% lithium; and d) the 2 nd EIL, 1 nm thick, composed of HAT-CN.
- This OLED having the 2 nd EIL in direct contact with the 1 st EIL, requires a drive voltage of about 5.0 V to pass 20 mA/cm 2 .
- the device has a luminance of 1891 cd/m 2 , and a luminous efficiency of about 9.5 cd/A. Its emission peak is at 521 nm.
- the operational lifetime measured as T 50 (RT@80 mA/cm 2 ), is about 312 hours.
- the average rate of the voltage rise during the lifetime testing is about 3.5 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm 2 , are shown in FIGS. 6 and 7, respectively.
- FIGS. 6 and 7 demonstrate that the OLED constructed in accordance with the present invention can have improved lifetime and improved voltage rise.
- the following examples will show how one can further reduce the drive voltage.
- Example 7 (Inventive)
- the layer structure of the EL unit is as follows: a) an HIL, 10 nm thick, composed of HAT-CN; b) an HTL, 65 nm thick, composed of NPB; c) a LEL , 30 nm thick, composed of AIq doped with 1.0 vol% C545T; d) the 1 st EIL, 20 nm thick, composed of AIq doped with about 1.2 vol% lithium; and e) the 2 nd EIL, 10 nm thick, composed of HAT-CN.
- This OLED requires a drive voltage of about 5.1 V to pass 20 mA/cm .
- the device has a luminance of 1823 cd/m , and a luminous efficiency of about 9.1 cd/A. Its emission peak is at 521 nm.
- the operational lifetime, measured as T 50 (RT@80 mA/cm 2 ), is about 350 hours.
- the average rate of the voltage rise during the lifetime testing is about 2.9 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm 2 , are shown in FIGS. 8 and 9, respectively.
- Example 8 (Inventive)
- OLED was constructed in accordance with the present invention.
- This OLED is the same as that in Example 7, except that layers d and e were changed as follows: d) the 1 st EIL, 5 nm thick, composed of AIq doped with about 1.2 vol% lithium; and e) the 2 nd EIL, 25 nm thick, composed of HAT-CN doped with about 1.2 vol% lithium.
- This OLED requires a drive voltage of about 4.6 V to pass
- the device has a luminance of 1898 cd/m , and a luminous efficiency of about 9.5 cd/A. Its emission peak is at 521 nm.
- the operational lifetime measured as Tso(RT@8O mA/cm 2 ), is about 330 hours.
- the average rate of the voltage rise during the lifetime testing is about 2.1 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm 2 , are shown in FIGS. 8 and 9, respectively.
- the OLEDs having a Li-doped HAT-CN layer as the 2 nd EIL can further reduce both the drive voltage and the voltage rise during operation.
- HIL hole-injecting layer
- HTL hole-transporting layer
- ETL 160 electron-transporting layer
- EIL electron-injecting layer
Landscapes
- Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
An OLED includes an anode (120) , a light emitting layer (150) disposed over the anode (120), and a first electron injecting layer disposed over the light- emitting layer, wherein the first electron- injecting layer (271) includes at least one organic host material having a reduction potential less than -1.0 V vs. a Saturated Calomel Electrode and at least one dopant material capable of reducing the organic host material. The OLED also includes a second electron-injecting layer (272) disposed in contact with the first electron- injecting layer (271) , wherein the second electron- injecting layer (272) includes at least one organic material having a reduction potential greater than -1.0 V vs . a Saturated Calomel Electrode, and a cathode (180) disposed over the second electron- injecting layer (272) .
Description
OLED ELECTRON-INJECTING LAYER FIELD OF INVENTION
The present invention relates to an organic light-emitting device (OLED) in which an organic material having a strong oxidizing property is used in an electron-injecting layer of the device.
BACKGROUND OF THE INVENTION OLEDs, as described by Tang in commonly assigned U.S. Patent 4,356,429, are commercially attractive because they offer the promise of low cost fabrication of high density pixel displays exhibiting bright electroluminance (EL) with long lifetime, high luminous efficiency, low drive voltage, and wide color range.
A typical OLED includes two electrodes and one organic EL unit disposed between the two electrodes. The organic EL unit commonly includes an organic hole-transporting layer (HTL), organic light-emitting layer (LEL), and an organic electron-transporting layer (ETL). One of the electrodes is the anode, which is capable of injecting positive charges (holes) into the HTL of the EL unit, and the other electrode is the cathode, which is capable of injecting negative charges (electrons) into the ETL of the EL unit. When the anode is biased with a certain positive electrical potential relative to the cathode, holes injected from the anode and electrons injected from the cathode can recombine and emit light from the LEL. At least one of the electrodes is optically transmissive, and the emitted light can be seen through the transmissive electrode.
Significant efforts have been made in selecting suitable materials and forming different layer structures in OLEDs to achieve improved EL performance. Numerous OLEDs with alternative layer structures have been disclosed. For example, in addition to the three layer OLEDs that contain a LEL between the HTL and the ETL (denoted as HTL/LEL/ETL), there are other multilayer OLEDs that contain additional functional layers in the EL unit, such as a hole-injecting layer (HIL), an electron-injecting layer (EIL), an electron-blocking layer (EBL), or a hole-blocking layer (HBL), or the combination thereof. These
new layer structures with new materials have indeed resulted in improved device performance.
As is known, electron transport in OLED is generally less efficient than hole transport. As a result, the electron-hole recombination in LEL is sometimes unbalanced with insufficient electrons. In order to achieve more efficient electron-hole recombination, much attention has been paid in forming an effective ETL or EIL using suitable electron-transporting material (ETM) in OLEDs. For example, the commonly used ETL or EIL in OLEDs includes the ETM of tris(8-hydroxyquinoline)aluminum (AIq), 4,7-diphenyl-l,10~ phenanthroline (Bphen), 2,9-dimethyl-4,7-diphenyl- 1 , 10-phenanthroline (BCP), and 2,2'-[l,l '-biphenyl]-4,4'-diylbis[4,6-(p-tolyl)-l,3,5-triazine] (TRAZ).
According to prior art, the selection of practically useful organic ETMs in OLEDs is limited to those having a reduction potential less than -1.0 V vs. a Saturated Calomel Electrode (SCE) or having a LUMO less than 3.3 eV below the vacuum energy level (Evac). This is because the LEL in an OLED typically has a reduction potential less than -1.0 V vs. SCE. Therefore if any organic material having a reduction potential greater than -1.0 V vs. SCE were used as an ETL in the OLED, it would produce an electron injection barrier between the ETL and the LEL. Especially, if any organic material having a reduction potential greater than -0.5 V vs. SCE were used as an ETL in the OLED, it would produce an electron injection barrier greater than 0.5 eV. Electron injection from the ETL into the LEL would be very difficult or impossible, resulting in little or no EL emission from the OLED. However, the commonly used ETMs, although forming a low or no barrier for electron injection at LEL/ETL interface, exhibit a relatively high barrier for electron injection at the interface between the ETL and the cathode, especially when the cathode material has a work function higher than 4.0 eV. This high barrier interface between the ETL and the cathode can be modified by inserting a thin insulating layer or by doping the ETL with a material having a work function lower than 3.0 eV, but this interface will be degraded eventually during operation.
- 9 -
Therefore, it would be advantageous to find a way to broaden the material selection for ETL (EIL)5 to use the material having a reduction potential greater than that of commonly used ETMs, and to produce a stable interface between organic layer and the cathode in OLEDs. SUMMARY OF THE INVENTION
It is therefore an object of the present invention to improve the EL performance of the OLED.
The object is achieved by an organic light-emitting device (OLED) comprising: a) an anode; b) a light-emitting layer disposed over the anode; c) a first electron-injecting layer disposed over the light- emitting layer, wherein the first electron-injecting layer includes at least one organic host material having a reduction potential less than -1.0 V vs. a Saturated Calomel Electrode and at least one dopant material capable of reducing the organic host material; d) a second electron-injecting layer disposed in contact with the first electron-injecting layer, wherein the second electron-injecting layer includes at least one organic material having a reduction potential greater than -1.0 V vs. a Saturated Calomel Electrode; and e) a cathode disposed over the second electron-injecting layer. The present invention makes use of a second electron-injecting layer including an organic material having a reduction potential greater than -1.0 eV in direct contact with the cathode and forming a stable interface between the cathode and this layer with no electron injection barrier from the cathode into the electron-injecting layer. It is an advantage of the present invention that the OLED with the second electron-injecting layer can not only increase the lifetime but also reduce the voltage rise during operation, which is useful for making high quality active matrix OLED display and other electronic devices. It is another advantage of the present invention that a stable metal, such as Ag, Au, Al, and Cu,
can be used to form the cathode. These stable metals do not readily react with water or oxygen to form dark spots in the OLEDs.
BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1 shows a cross-sectional view of a prior art OLED; FIG. 2 shows a cross-sectional view of one embodiment of an
OLED prepared in accordance with the present invention;
FIG. 3 shows a cross-sectional view of another embodiment of an OLED prepared in accordance with the present invention;
FIG. 4 shows a cross-sectional view of yet another embodiment of an OLED prepared in accordance with the present invention;
FIG. 5 shows a cross-sectional view of yet another embodiment of an OLED prepared in accordance with the present invention;
FIG. 6 is a graph showing the normalized luminance vs. operational time of a group of OLEDs tested at room temperature and at 80 mA/cm ; FIG. 7 is a graph showing the drive voltage vs. operational time of the group of OLEDs tested at room temperature and at 80 mA/cm2;
FIG. 8 is a graph showing the normalized luminance vs. operational time of another group of OLEDs tested at room temperature and at 80 mA/cm ; and FIG. 9 is a graph showing the drive voltage vs. operational time of the other group of OLEDs tested at room temperature and at 80 mA/cm2.
It will be understood that FIGS. 1-5 are not to scale since the thickness differences of various layers are too great to permit depiction to scale.
DETAILED DESCRIPTION OF THE INVENTION There is shown a cross-sectional view of a prior art OLED in
FIG. 1. OLED 100 includes substrate 110, anode 120, HIL 130, HTL 140, LEL 150, ETL 160, EIL 170, and cathode 180 (HIL 130, HTL 140, LEL 150, ETL 160, and EIL 170 form an organic EL unit in between the anode 120 and cathode 180). OLED 100 is externally connected to a voltage/current source 192 through electrical conductors 191. OLED 100 is operated by applying an electric potential
produced by the voltage/current source 192 between the pair of contact electrodes, anode 120, and cathode 180.
There is shown a cross-sectional view of one embodiment of an OLED in accordance with the present invention in FIG. 2. OLED 200 includes substrate 110, anode 120, HIL 130, HTL 140, LEL 150, ETL 160, the 1st EIL 271, the 2nd EIL 272, and cathode 180. OLED 200 is the same as OLED 100 except that OLED 200 has two EILs (EIL 271 and EIL 272). OLED 200 is also externally connected to a voltage/current source 192 through electrical conductors 191. Shown in FIGS. 3, 4, and 5 are OLED 300, OLED 400, and OLED 500, respectively, which are some other embodiments of OLEDs prepared in accordance with the present invention. OLED 300 in FIG. 3 is the same as OLED 200 except that there is no HIL 130 in OLED 300; OLED 400 in FIG. 4 is the same as OLED 200 except that there is no ETL 160 in OLED 400; and OLED 500 in FIG. 5 is the same as OLED 200 except that there is no HIL 130 nor ETL 160 in OLED 500. Note that the substrate can alternatively be located adjacent to the cathode, or the substrate can actually constitute the anode or cathode. The total combined thickness of the organic layers is preferably less than 500 ran.
The present invention can be employed in the making of most OLED device configurations. These include very simple structures including a single anode and cathode to more complex devices, such as passive matrix displays including orthogonal arrays of anodes and cathodes to form pixels, and active matrix displays where each pixel is controlled independently, for example, with thin film transistors (TFTs). Such TFTs can be constructed using amorphous silicon, low temperature polycrystalline silicon, single crystal silicon, other inorganic semiconductors, or organic semiconductor materials.
The following will be the description of the device structure, material selection, and fabrication process of the OLEDs in accordance with the present invention.
Substrate 110 can be an organic solid, an inorganic solid, or includes organic and inorganic solids that provides a supporting backplane to hold the OLED. Substrate 110 can be rigid or flexible and can be processed as separate
individual pieces, such as sheets or wafers, or as a continuous roll. Typical substrate materials include glass, plastic, metal, ceramic, semiconductor, metal oxide, semiconductor oxide, or semiconductor nitride, or combinations thereof. Substrate 110 can be a homogeneous mixture of materials, a composite of materials, or multiple layers of materials. Substrate 110 can also be a backplane containing thin-film transistor (TFT) circuitry commonly used for preparing OLED display, e.g. an active matrix low temperature polysilicon TFT substrate. The substrate 110 can either be light transmissive or opaque, depending on the intended direction of light emission. The light transmissive property is desirable for viewing the EL emission through the substrate. Transparent glass or plastic is commonly employed in such cases. For applications where the EL emission is viewed through the top electrode, the transmissive characteristic of the bottom support is immaterial, and therefore can be light transmissive, light absorbing, or light reflective. Substrates for use in this case include, but are not limited to, glass, plastic, semiconductor materials, ceramics, and circuit board materials, or any others commonly used in the formation of OLEDs, which can be either passive matrix devices or active matrix devices.
Anode 120 is formed over substrate 110. When EL emission is viewed through the substrate 110, the anode should be transparent or substantially transparent to the emission of interest. For applications where EL emission is viewed through the top electrode, the transmissive characteristics of the anode material are immaterial and any conducting or semiconducting material can be used, regardless if it is transparent, opaque, or reflective. Desired anode materials can be deposited by any suitable way such as thermal evaporation, sputtering, chemical vapor deposition, or electrochemical means. Anode materials can be patterned using well known photolithographic processes.
The material for use to form anode 120 can be selected from inorganic materials, or organic materials, or combination thereof. The anode 120 can contain the element material selected from aluminum, silver, gold, copper, zinc, indium, tin, titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, indium, nickel, palladium,
platinum, silicon, or germanium, or combinations thereof. The anode 120 can also contain a compound material, such as a conducting or semiconducting compound. The conducting or semiconducting compound can be selected from the oxides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof. The conducting or semiconducting compound can be selected from the sulfides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof. The conducting or semiconducting compound can be selected from the selenides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof. The conducting or semiconducting compound can be selected from the tellurides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof. The conducting or semiconducting compound can be selected from the nitrides of titanium, zirconium, hafnium, niobium, tantalum, molybdenum, tungsten, manganese, iron, ruthenium, rhodium, iridium, nickel, palladium, platinum, copper, zinc, indium, tin, silicon, or germanium, or combinations thereof. Preferably, the conducting or semiconducting compound can be selected from indium tin oxide, tin oxide, aluminum-doped zinc oxide, indium-doped zinc oxide, magnesium indium oxide, nickel tungsten oxide, zinc sulfide, zinc selenide, or gallium nitride, or the combination thereof.
It is often useful to provide a hole-injecting layer 130 between anode 120 and hole-transporting layer 140. The hole-injecting material can serve to improve the film formation property of subsequent organic layers and to facilitate injection of holes into the hole-transporting layer. Suitable materials for use in the hole-injecting layer include, but are not limited to, porphyrinic
compounds as described in U.S. Patent 4,720,432, plasma-deposited fluorocarbon polymers as described in U.S. Patents 6,127,004, 6,208,075 and 6,208,077, some aromatic amines, for example, m-MTDATA (4,4',4"-tris[(3-methylphenyl)phenyl- amino]triphenylamine), and inorganic oxides including vanadium oxide (VOx), molybdenum oxide (MoOx), and nickel oxide (NiOx). Alternative hole-injecting materials reportedly useful in organic EL devices are described in EP 0 891 121 Al and EP 1 029 909 Al. Hexaazatriphenylene derivatives are also useful HIL materials, as described in U.S. Patent 6,720,573.
Another class of suitable materials for use in the HIL includes p-type doped organic materials. A p-type doped organic material typically includes a hole-transporting material such as an aromatic amine (see below) that is doped with an electron accepting dopant. Such dopants can include, for example, 2,3,5,6-tetrafluoro-7,7,8,8-tetracyanoquinodimethane (F4-TCNQ) and other derivatives of 7,7,8, 8-tetracyanoquinodimethane (TCNQ), and inorganic oxidizing agents such as iodine, FeC13, FeF3, SbCl5, some other metal chlorides, and some other metal fluorides. The p-type doped concentration is preferably in the range of 0.01-20 vol. %. Such layers materials are further described in, for example, U.S. Patents 5,093,698, 6,423,429, and 6,597,012.
The thickness of the HIL can be in the range of from 0.1 nm to 200 nm, preferably, in the range of from 0.5 nm to 150 nm.
The HTL 140 contains at least one hole-transporting material such as an aromatic tertiary amine, where the aromatic tertiary amine is understood to be a compound containing at least one trivalent nitrogen atom that is bonded only to carbon atoms, at least one of which is a member of an aromatic ring. In one form the aromatic tertiary amine can be an arylamine, such as a monoarylamine, diarylamine, triarylamine, or a polymeric arylamine. Exemplary monomeric triarylamines are illustrated by Klupfel et al. in U.S. Patent 3,180,730. Other suitable triarylamines substituted with one or more vinyl radicals or at least one active hydrogen-containing group are disclosed by Brantley, et al. in U.S. Patents 3,567,450 and 3,658,520.
A more preferred class of aromatic tertiary amines includes compounds that include at least two aromatic tertiary amine moieties as described by Van Slyke and Tang et al. in U.S. Patents 4,720,432 and 5,061,569. The HTL can be formed of a single or a mixture of aromatic tertiary amine compounds. Illustrative of useful aromatic tertiary amines are the following:
1 , 1 -Bis(4-di-p-tolylaminophenyl)cyclohexane;
1 , 1 -Bis(4-di-p-tolylaminophenyl)-4-phenylcyclohexane;
N,N,Nl,Nt-tetraphenyl-4,4m-diamino- 1 , 1' :4', 1" :4", 1 '"-quaterphenyl;
Bis(4-dimethylamino-2-methylphenyl)phenylmethane; 1 ,4-bis[2-[4-[N,N-di(p-toly)amino]phenyl]vinyl]benzene (BDTAPVB);
N,N,N',N'-Tetra-p-tolyl-4,4'-diaminobiphenyl;
N,N,N',N'-Tetraphenyl-4,4'-diaminobiphenyl;
N,N,N',N'-tetra-l-naphthyl-4,4'-diaminobiphenyl;
N,N,N',N'-tetra-2-naphthyl-4,4'-diaminobiphenyl; N-Phenylcarbazole;
4,4'-Bis[N-(l -naphthyl)-N-phenylamino]biphenyl (NPB);
4,4'-Bis[N-(l -naρhthyl)-N-(2-naρhthyl)amino]biphenyl (TNB);
4,4'-Bis [N-( 1 -naphthyl)-N-phenylamino]p-terphenyl;
4,4'-Bis[N-(2-naphthyl)-N-phenylammo]biphenyl; 4,4'-Bis [N-(3 -acenaphthenyl)-N-phenylamino]biphenyl;
1 ,5-Bis[N-(l -naphthyl)-N-phenylamino]naphthalene;
4,4'-Bis[N-(9-anthryl)-N-phenylamino]biphenyl;
4,4'-Bis[N-(l-anthryl)-N-phenylamino]-p-terphenyl;
4,4'-Bis[N-(2-phenanthryl)-N-phenylamino]biphenyl; 4,4'-Bis[N-(8-fluoranthenyl)-N-phenylamino]biphenyl;
4,4'-Bis[N-(2-pyrenyl)-N-phenylamino]biphenyl;
4,4'-Bis[N-(2-naphthacenyl)-N-phenylamino]biphenyl;
4,4'-Bis[N-(2-perylenyl)-N-phenylamino]biphenyl;
4,4'-Bis[N-(l-coronenyl)-N-phenylamino]biphenyl; 2,6-Bis(di-p-tolylamino)naphthalene;
2,6-Bis[di-(l-naphthyl)amino]naphthalene;
2,6-Bis[N-(l-naphthyl)-N-(2-naphthyl)amino]naphthalene; N,N,N',N'-Tetra(2-naphthyl)-4,4"-diamino-p-terplienyl; 4,4'-Bis {N-phenyl-N-[4-(l -naphthyl)-phenyl] amino }biphenyl; 2, 6-Bis [N,N-di(2-naphthyi)amino] fluorene; 4,4l,4"-tris[(3-methylphenyl)phenylamino]triphenylamine (MTDATA); and
4,4'-Bis[N-(3-methylρlienyl)-N-ρhenylamino]biρlienyl (TPD).
Another class of useful hole-transporting materials includes polycyclic aromatic compounds as described in EP 1 009 041. Some hole-injecting materials described in EP 0 891 121 Al and EP 1 029 909 Al can also make useful hole-transporting materials. In addition, polymeric hole-transporting materials can be used including poly(N-vinylcarbazole) (PVK), polythiophenes, polypyrrole, polyaniline, and copolymers including poly(3, 4-ethylenedioxy- thiophene)/poly(4-styrenesulfonate) also called PEDOT/PSS.
The thickness of the HTL can be in the range of from 5 nm to 200 nm, preferably, in the range of from 10 nm to 150 nm.
The LEL 150 can include a luminescent fluorescent or phosphorescent material where electroluminescence is produced as a result of electron-hole pair recombination in this layer. The light-emitting layer can include a single material, but more commonly contains at least one host material doped with at least one guest emitting material or materials where light emission comes primarily from the emitting materials and can be of any color. This guest emitting material is often referred to as a luminescent dopant material. The host materials in the light-emitting layer can be an electron-transporting material as defined below, a hole-transporting material as defined above, or another material or combination of materials that support hole-electron recombination. The emitting material is typically chosen from highly fluorescent dyes and phosphorescent compounds, e.g., transition metal complexes as described in WO 98/55561 Al, WO 00/18851 Al, WO 00/57676 Al, and WO 00/70655. Emitting materials are typically incorporated at 0.01 to 20 % by volume of the host material. The host and emitting materials can be small nonpolymeric molecules or polymeric materials including polyfluorenes and polyvinylarylenes,
e.g., poly(p-phenylenevinylene), PPV. In the case of polymers, small molecule emitting materials can be molecularly dispersed into a polymeric host, or the emitting materials can be added by copolymerizing a minor constituent into a host polymer. An important relationship for choosing an emitting material is a comparison of the electron energy bandgap that is defined as the energy difference between the highest occupied molecular orbital and the lowest unoccupied molecular orbital of the molecule. For efficient energy transfer from the host to the emitting material, a necessary condition is that the bandgap of the dopant is smaller than that of the host material. For phosphorescent emitters (including materials that emit from a triplet excited state, i.e., so-called "triplet emitters") it is also important that the triplet energy level of the host be high enough to enable energy transfer from host to emitting material.
Host and emitting materials known to be of use include, but are not limited to, those disclosed in U.S. Patents 4,768,292, 5,141,671, 5,150,006, 5,151,629, 5,405,709, 5,484,922, 5,593,788, 5,645,948, 5,683,823, 5,755,999, 5,928,802, 5,935,720, 5,935,721, 6,020,078, 6,475,648, 6,534,199, 6,661,023, U.S. Patent Application Publications 2002/0127427 Al, 2003/0198829 Al, 2003/0203234 Al, 2003/0224202 Al, and 2004/0001969 Al. Metal complexes of 8-hydroxyquinoline (oxine), otherwise known as chelated oxinoid compound, and similar derivatives constitute one class of useful host compounds capable of supporting electroluminescence. Illustrative of useful chelated oxinoid compounds are the following:
CO-I : Aluminum trisoxine [alias, tris(8-qumolmolato)aluminum(IIi)]; CO-2: Magnesium bisoxine [alias, bis(8-quinolinolato)magnesium(II)];
CO-3: Bis[benzo{f}-8-quinolinolato]zinc (II); CO-4: Bis(2-methyl-8-quinolinolato)aluminum(III)-μ-oxo-bis(2-methyl-8- quinolinolato) aluminum(III);
CO-5: Indium trisoxine [alias, tris(8-quinolinolato)indium]; CO-6: Aluminum tris(5-methyloxine) [alias, tris(5-methyl-8-quinolinolato) aluminum(III)];
CO-7: Lithium oxine [alias, (8-quinolinolato)lithmm(I)]; CO-8: Gallium oxine [alias, tris(8-quinolinolato)gallium(III)]; and CO-9: Zirconium oxine [alias, tetra(8-quinolinolato)zirconium(IV)] Another class of useful host materials includes derivatives of anthracene, such as those described in U.S. Patents 5,935,721, 5,972,247, 6,465,115, 6,534,199, 6,713,192, U.S. Patent Application Publications 2002/0048687 Al, 2003/0072966 Al, and WO 2004/018587. Some examples include derivatives of 9,10-dinaphthylanthracene and 9-naphthyl-10- phenylanthracene. Other useful classes of host materials include distyrylarylene derivatives as described in U.S. Patent 5,121,029, and benzazole derivatives, for example, 2, 2', 2"-(l,3,5-phenylene)tris[l-phenyl-lH-benzimidazole],
Suitable host materials for phosphorescent dopants should be selected so that a triplet exciton can be transferred efficiently from the host material to the phosphorescent material. For this transfer to occur, it is a highly desirable condition that the excited state energy of the phosphorescent material be lower than the difference in energy between the lowest triplet state and the ground state of the host. However, the bandgap of the host should not be chosen so large as to cause an unacceptable increase in the drive voltage of the OLEDs. Suitable host materials are described in WO 00/70655 A2, WO 01/39234 A2, WO 01/93642 Al, WO 02/074015 A2, WO 02/15645 Al, and U.S. Patent Application Publication 2002/0117662 Al . Suitable hosts include certain aryl amines, triazoles, indoles and carbazole compounds. Examples of desirable hosts are 4,4'-N,N'-dicarbazole-biρhenyl (CBP), 2,2'-dimethyl-4,4'-N,N'-dicarbazole- biphenyl, m-(N,N'-dicarbazole)benzene, and poly(N-vinylcarbazole), including their derivatives.
Desirable host materials are capable of forming a continuous film. The light-emitting layer can contain more than one host material in order to improve the device's film morphology, electrical properties, light emission efficiency, and lifetime. Mixtures of electron-transporting and hole-transporting materials are known as useful hosts. In addition, mixtures of the above listed host
materials with hole-transporting or electron-transporting materials can make suitable hosts.
Useful fluorescent dopants include, but are not limited to, derivatives of anthracene, tetracene, xanthene, perylene, rubrene, coumarin, rhodamine, and quinacridone, dicyanomethylenepyran compounds, thiopyran compounds, polymethine compounds, pyrylium and thiapyrylium compounds, fluorene derivatives, periflanthene derivatives, indenoperylene derivatives, bis(azinyl)amine boron compounds, bis(azinyl)methane boron compounds, derivatives of distryrylbenzene and distyrylbiphenyl, and carbostyryl compounds. Among derivatives of distyrylbenzene, particularly useful are those substituted with diarylamino groups, informally known as distyrylamines.
Examples of useful phosphorescent dopants that can be used in light-emitting layers of this invention include, but are not limited to, those described in WO 00/57676 Al, WO 00/70655, WO 01/41512 Al, WO 02/15645 Al, WO 01/93642 Al, WO 01/39234 A2, WO 02/071813 Al, WO 02/074015 A2, U.S. Patent Application Publications 2003/0017361 Al, 2002/0197511 Al, 2003/0072964 Al, 2003/0068528 Al, 2003/0059646 Al, 2003/0054198 Al, 2003/0124381 Al, 2002/0100906 Al, 2003/0068526 Al, 2003/0068535 Al, 2003/0141809Al, 2003/0040627 Al, 2002/0121638 Al, U.S. Patents 6,458,475, 6,573,651, 6,451,455, 6,413,656, 6,515,298, 6,451,415, 6,097,147, EP 1 239 526 A2, EP 1 238 981 A2, and EP 1 244 155 A2. Useful phosphorescent dopants include transition metal complexes, such as iridium and platinum complexes. hi some cases it is useful for one or more of the LELs within an EL unit to emit broadband light, for example white light. Multiple dopants can be added to one or more layers in order to produce a white-emitting OLED, for example, by combining blue- and yellow-emitting materials, cyan- and red- emitting materials, or red-, green-, and blue-emitting materials. White-emitting devices are described, for example, in EP 1 187 235, EP 1 182 244, U.S. Patents 5,683,823, 5,503,910, 5,405,709, 5,283,182, 6,627,333, 6,696,177, 6,720,092, U.S. Patent Application Publications 2002/0186214 Al, 2002/0025419 Al, and
2004/0009367 Al. In some of these systems, the host for one light-emitting layer is a hole-transporting material. For example, it is known in the art that emitting dopants can be added to the HTL 140, thereby enabling HTL 140 to serve as a host. The thickness of each LEL can be in the range of from 5 nm to 80 run, preferably, in the range of from 10 nm to 40 nm.
Preferred organic materials for use in forming the ETL 160 are metal chelated oxinoid compounds, including chelates of oxine itself, also commonly referred to as 8-quinolinol or 8-hydroxyquinoline. Such compounds help to inject and transport electrons, exhibit high levels of performance, and are readily deposited to form thin films. Exemplary oxinoid compounds have been listed above from CO-I to CO-9 (the oxinoid compounds can be used as both the host material in LEL 150 and the electron-transporting material in ETL 160).
Other electron-transporting materials include various butadiene derivatives as disclosed in U.S. Patent 4,356,429 and various heterocyclic optical brighteners as described in U.S. Patent 4,539,507. Benzazoles, oxadiazoles, triazoles, pyridinethiadiazoles, triazines, phenanthrolme derivatives, and some silole derivatives are also useful electron-transporting materials.
The 1st EIL 271 as shown in FIGS. 2-5 is an n-type doped layer containing at least one electron-transporting material as a host material and at least one n-type dopant material (this EIL can also be called an n-type doped 1st EIL 271). The reduction potential of one of the organic host materials in the 1st EIL 271 is less than -1.0 V vs. SCE and one of the dopant materials is capable of reducing one of the organic host materials. The term " n-type doped layer" means that this layer has semiconducting properties after doping, and the electrical current through this layer is substantially carried by the electrons. The host material is capable of supporting electron injection and electron transport. The electron-transporting materials used in ETL 160 represent a useful class of host materials for the n-type doped 1st EIL 271. Preferred materials are metal chelated oxinoid compounds, including chelates of oxine itself (also commonly referred to as 8-quinolinol or 8-hydroxyquinoline), such as tris(8-hydroxyquinoline)- aluminum (AIq). Other materials include various butadiene derivatives as
disclosed by Tang in U.S. Patent 4,356,429, various heterocyclic optical brighteners as disclosed by Van Slyke and Tang et al. in U.S. Patent 4,539,507, triazines, hydroxyquinoline derivatives, benzazole derivatives, and phenanthroline derivatives. Silole derivatives, such as 2,5-bis(2',2"-bipridin-6-yl)-l,l-dimethyl- 3,4-diphenyl silacyclopentadiene are also useful host organic materials. The combination of the aforementioned host materials is also useful to form the n-typed doped EIL 170. More preferably, the host material in the n-type doped EIL 170 includes AIq, 4,7-diρhenyl-l,10-ρhenanthroline (Bphen), 2,9-dimethyl- 4,7-diphenyl-l,10-phenanthroline (BCP), 2,2'-[l,l '-biphenyl]-4,4'-diylbis[4,6- (p-tolyl)-l,3,5-triazine] (TRAZ), or rubrene, or combinations thereof.
Both 1st EIL 271 and ETL 160 in the OLEDs can use the same or different material.
The n-type dopant in the n-type doped 1st EIL 271 is selected from alkali metals, alkali metal compounds, alkaline earth metals, or alkaline earth metal compounds, or combinations thereof. The term "metal compounds" includes organometallic complexes, metal organic salts, and inorganic salts, oxides and halides. Among the class of metal containing n-type dopants, Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Sm, Eu, Tb, Dy, or Yb, and their compounds, are particularly useful. The materials used as the n-type dopants in the n-type doped EIL 170 also include organic reducing agents with strong electron-donating properties. By "strong electron-donating properties" it is meant that the organic dopant should be able to donate at least some electronic charge to the host to form a charge transfer complex with the host. Nonlimiting examples of organic molecules include bis(ethylenedithio)-tetrathiafulvalene (BEDT-TTF), tefrathiafurvalene (TTF), and their derivatives. In the case of polymeric hosts, the dopant can be any of the above or also a material molecularly dispersed or copolymerized with the host as a minor component. Preferably, the n-type dopant in the n-type doped 1st EIL 271 includes Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Nd, Sm, Eu, Tb, Dy, or Yb, or combinations thereof. The n-type doped concentration is preferably in the range of 0.01 -20% by volume of this layer. The
thickness of the n-type doped 1st EIL 271 is typically less than 200 nm, and preferably in the range of 2 to 150 nm.
The 2nd EIL 272 in the OLEDs as shown in FIGS. 2-5 is disposed in contact with the 1st EIL 271, and includes at least one organic material having a reduction potential greater than -1.0 V vs. SCE, preferably, greater than -0.5 Vs. SCE.
The organic materials having a reduction potential greater than -1.0 V vs. SCE, especially greater than -0.5 V vs. SCE, are promising n-type semiconducting materials with electron accepting property. The materials can provide effective electron transport in OLEDs. Moreover, when the reduction potential of the materials is greater than -0.5 V vs. SCE, the LUMO position of the material will be lower than 3.8 eV from the Evac. Therefore, it is possible to select the 2nd EIL 272 according to the work function of the cathode 180 such that the LUMO position of the 2nd EIL 272 is below or near the work function of the cathode 180. As a result, there is no electron injection barrier at the interface of the 2nd EIL 272 and the cathode 180. Furthermore, by "electron accepting property" it is meant that the organic material has the capability or tendency to accept at least some electronic charge from other types of material that it is adjacent to. Having electron accepting property also means having a strong oxidizing property. Therefore, the 2nd EIL 272 can also react with the first several monolayers of the cathode when the cathode layer is formed on top of the 2n EIL 272. As a result, cathode 180 can have an improved contact with the 2nd EIL 272 forming a stable interface between the 2nd EIL 272 and the cathode 180. The 2nd EIL 272 can be as thin as 1 nm. However, it can also be as thick as 200 nm. Preferably, the thickness of 2nd EIL 272 is in the range of from 1 to 150 nm.
The term "reduction potential", expressed in volts and abbreviated Eie , measures the affinity of a substance for an electron: the larger (more positive) the number, the greater the affinity. The reduction potential of a substance can be conveniently obtained by cyclic voltammetry (CV) and it is measured vs. SCE. The measurement of the reduction potential of a substance can be as following: An electrochemical analyzer (for instance, a CHI660 electrochemical analyzer, made
by CH Instruments, Inc., Austin, TX) is employed to carry out the electrochemical measurements. Both CV and Osteryoung square-wave voltammetry (SWV) can be used to characterize the redox properties of the substance. A glassy carbon (GC) disk electrode (A=0.071 cm2) is used as working electrode. The GC electrode is polished with 0.05 μm alumina slurry, followed by sonication cleaning in deionized water twice and rinsed with acetone between the two water cleanings. The electrode is finally cleaned and activated by electrochemical treatment prior to use. A platinum wire can be used as the counter electrode and the SCE is used as a quasi-reference electrode to complete a standard 3 -electrode electrochemical cell. A mixture of acetonitrile and toluene (1:1 MeCN/toluene) or methylene chloride (MeCl2) can be used as organic solvent systems. All solvents used are ultra low water grade (<10 ppm water). The supporting electrolyte, tetrabutylammonium tetrafmoroborate (TBAF) is recrystallized twice in isopropanol and dried under vacuum for three days. Ferrocene (Fc) can be used as an internal standard (Ered Fo=0.50 V vs. SCE in 1 : 1 MeCN/toluene, Ered Fc=0.55 V vs. SCE in MeCl2, 0.1 M TBAF, both values referring to the reduction of the ferrocenium radical anion). The testing solution is purged with high purity nitrogen gas for approximately 15 minutes to remove oxygen and a nitrogen blanket is kept on the top of the solution during the course of the experiments. All measurements are performed at an ambient temperature of 25 + 1°C. If the compound of interest has insufficient solubility, other solvents can be selected and used by those skilled in the art. Alternatively, if a suitable solvent system cannot be identified, the electron accepting material can be deposited onto the electrode and the reduction potential of the modified electrode can be measured. Several types of organic materials having a reduction potential greater than -1.0 V vs. SCE, especially greater than -0.5 V vs. SCE, can be used to form the 2nd EIL 272 in the present invention.
The organic material used in the 2nd EIL 272 can be a chemical compound of Formula I
wherein R1-R4 represent hydrogen or substituents independently selected from the group including halo, nitrile (-CN), nitro (-NO2), sulfonyl (-SO2R), sulfoxide (-SOR), trifluoromethyl (-CF3), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein R1 and R2, or R3 and R4, combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
Specifically, the organic material used in the 2nd EIL 272 can be a chemical compound of Formula Ia (2,3,5,6-tetrafluoro-7,7,8,8-tetracyano- quinodimethane [F4-TCNQ])
or can be a chemical compound of Formula Ib
Formula Ha
or can be a chemical compound of
Formula lib
or can be a chemical compound of
Formula Hc
or can be a chemical compound of
Formula Hd
or can be a chemical compound of
Formula Hf
or a derivative any of these compounds resulting from replacement of one or more hydrogen atoms by substituents including of halo, nitrile (-CN), nitro (-NO2), sulfonyl (-SO2R), sulfoxide (-SOR), trifluoromethyl (-CF3), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein two or more substituents combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
The organic material used in the 2nd EIL 272 can be a chemical compound of Formula III
wherein R1-R6 represent hydrogen or a substituent independently selected from the group including halo, nitrile (-CN), nitro (-NO2), sulfonyl (-SO2R), sulfoxide (-SOR), trifiuoromethyl (-CF3), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein R1 and R2, R3 and R4, or R5 and R6, combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
Specifically, the organic material used in the 2nd EIL 272 can be a chemical compound of Formula Ilia (hexaazatriphenylene hexacarbonitrile)
Additional organic materials that can be used in the 2nd EIL include diimide derivative of naphthalene tetracarboxylic diimide and of perylene tetracarboxylic diimide that are substituted with two or more fluoro groups, such as a chemical compound of
Formula IVa
Formula IVb
Formula IVc
or a chemical compound of
Formula IVd
It should be noted that in addition to the aforementioned types of materials, other materials useful to form the 2nd EIL 272 can be selected from any material having the reduction potential is greater than -1.0 V. vs. SCE. It should also be noted that organic materials suitable for use in the 2nd EIL 272 not only include the compounds containing at least carbon and hydrogen, but also include metal complexes, e.g., transition metal complexes having organic ligands and organometallic compounds, as long as their reduction potentials are in the appropriate range. hi order to have effective optical transparency and not absorb light produced by the device, the organic materials used in the 2n EIL 272 should preferably have an optical bandgap greater than 1.6 eV.
The 2nd EIL 272 can also be doped with at least one n-type dopant material having a work function less than 4.0 eV into the organic host material. The n-type dopant for use in the 2nd EIL 272 is selected from alkali metals, alkali
metal compounds, alkaline earth metals, or alkaline earth metal compounds, or combinations thereof. Among the class of metal containing n-type dopants, Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Sm, Eu, Tb, Dy, or Yb, and their compounds, are particularly useful. The n-type doped concentration is preferably in the range of 0.01 -20% by volume of this layer. The n-type dopant used in the 2nd EIL 272 can be the same or different from that used in the 1st EIL 271.
Additional layers such as exciton, electron and hole-blocking layers as taught in the art can be employed in devices of this invention. Hole-blocking layers are commonly used to improve efficiency of phosphorescent emitter devices, for example, as in U.S. Patent Application Publications
2002/0015859 Al, 2003/0068528 Al, 2003/0175553 Al, WO 00/70655 A2, and WO 01/93642 Al.
Each of the layers in the organic EL units in the OLEDs can be formed from small molecule (or nonpolymeric) materials (including fluorescent materials and phosphorescent materials), polymeric LED materials, or inorganic materials, or combinations thereof.
When light emission is viewed solely through the anode, the cathode 180 can include nearly any conductive material. Desirable materials have effective film forming properties to ensure effective contact with the underlying organic layer, promote electron injection at low voltage, and have effective stability. Commonly used cathodes include a Mg: Ag alloy as described in U.S. Patent 4,885,221, a LiF/Al bilayer as described in U.S. Patent 5,677,572, Al, Ag, Au, Cu, or a combination thereof. Other useful cathodes include, but are not limited to, those disclosed in U.S. Patents 5,059,861, 5,059,862, and 6,140,763. Since the 2nd EIL 272 includes the organic material having a reduction potential greater than -1.0 V vs. SCE, the deposited cathode can form a stable interface with the 2nd EIL 272 resulting in less interface degradation during operation as well as less dark spots due to less reaction with water or oxygen during storage. It is preferable that the barrier to injection of electrons from the cathode material into the 2nd EIL be no greater than 0.3 eV. For example, if the 2nd EIL 272 includes an organic material having a reduction potential of -0.5 V vs. SCE, which means that
the LUMO position is about -3.8 eV, the cathode can be then selected from materials whose work functions are equal to or smaller than 4.1 eV.
When light emission is viewed through the cathode, the cathode should be transparent or nearly transparent. For such applications, metals should be thin or one should use transparent conductive oxides, or include these materials. Optically transparent cathodes have been described in more detail in U.S. Patents 4,885,211, 5,247,190, 5,703,436, 5,608,287, 5,837,391, 5,677,572, 5,776,622, 5,776,623, 5,714,838, 5,969,474, 5,739,545, 5,981,306, 6,137,223, 6,140,763, 6,172,459, 6,278,236, 6,284,393, and EP 1 076 368. Cathode materials are typically deposited by thermal evaporation, electron beam evaporation, ion sputtering, or chemical vapor deposition. When needed, patterning can be achieved through many well known methods including, but not limited to, through-mask deposition, integral shadow masking, for example as described in U.S. Patent 5,276,380 and EP 0 732 868, laser ablation, and selective chemical vapor deposition.
This invention can be used in a so-called tandem device architecture, for example, as taught in U.S. Patents 6,337,492, 6,717,358, and U.S. Patent Application Publication 2003/0170491 Al . Such tandem devices have multiple electroluminescent units provided between an anode and a cathode, typically with connector layer between units to promote charge production and injection into the electroluminescent units.
The organic materials mentioned above are suitably deposited through a vapor-phase method such as sublimation, but can be deposited from a fluid, for example, from a solvent with an optional binder to improve film formation. If the material is a polymer, solvent deposition is useful but other methods can be used, such as sputtering or thermal transfer from a donor sheet. The material to be deposited by sublimation can be vaporized from a sublimation "boat" often including a tantalum material, e.g., as described in U.S. Patent 6,237,529, or can be first coated onto a donor sheet and then sublimed in closer proximity to the substrate. Layers with a mixture of materials can use separate sublimation boats or the materials can be premixed and coated from a single boat
or donor sheet. Patterned deposition can be achieved using shadow masks, integral shadow masks (U.S. Patent 5,294,870), spatially defined thermal dye transfer from a donor sheet (U.S. Patents 5,688,551, 5,851,709 and 6,066,357), and inkjet method (U.S. Patent 6,066,357). Most OLED devices are sensitive to moisture or oxygen, or both, so they are commonly sealed in an inert atmosphere such as nitrogen or argon. In sealing an OLED device in an inert environment, a protective cover can be attached using an organic adhesive, a metal solder, or a low melting temperature glass. Commonly, a getter or desiccant is also provided within the sealed space. Useful getters and desiccants include, alkali and alkaline metals, alumina, bauxite, calcium sulfate, clays, silica gel, zeolites, alkaline metal oxides, alkaline earth metal oxides, sulfates, or metal halides and perchlorates. Methods for encapsulation and desiccation include, but are not limited to, those described in U.S. Patent 6,226,890. In addition, barrier layers such as SiOx, Teflon, and alternating inorganic/polymeric layers are known in the art for encapsulation.
OLED devices of this invention can employ various well known optical effects in order to enhance its properties if desired. This includes optimizing layer thicknesses to yield maximum light transmission, providing dielectric mirror structures, replacing reflective electrodes with light-absorbing elect-odes, providing anti glare or antireflection coatings over the display, providing a polarizing medium over the display, or providing colored, neutral density, or color conversion filters in functional relationship with the light emitting areas of the display. Filters, polarizers, and antiglare or antireflection coatings can also be provided over a cover or as part of a cover. The OLED device can have a microcavity structure. In one useful example, one of the metallic electrodes is essentially opaque and reflective; the other one is reflective and semitransparent. The reflective electrode is preferably selected from Au, Ag, Mg, Ca, or alloys thereof. Because of the presence of the two reflecting metal electrodes, the device has a microcavity structure. The strong optical interference in this structure results in a resonance condition. Emission near the resonance wavelength is enhanced and emission away from the resonance
wavelength is depressed. The optical path length can be tuned by selecting the thickness of the organic layers or by placing a transparent optical spacer between the electrodes. For example, an OLED device of this invention can have ITO spacer layer placed between a reflective anode and the organic EL media, with a semitransparent cathode over the organic EL media.
The aforementioned OLEDs prepared in accordance with the present invention are useful in applications. OLED displays or the other electronic devices can include a plurality of the OLEDs as described above. EXAMPLES The following examples are presented for a further understanding of the present invention. In the following examples, the reduction potentials of the materials were measured using an electrochemical analyzer (CHI660 electrochemical analyzer, made by CH Instruments, Inc., Austin, TX) with the method as discussed before. During the fabrication of OLEDs, the thickness of the organic layers and the doping concentrations were controlled and measured in situ using calibrated thickness monitors (INFICON IC/5 Deposition Controller, made by Infϊcon Inc., Syracuse, NY). The EL characteristics of all the fabricated devices were evaluated using a constant current source (KEITHLEY 2400 SourceMeter, made by Keithley Instruments, hie, Cleveland, Ohio) and a photometer (PHOTO RESEARCH SpectraScan PR 650, made by Photo Research, Inc., Chatsworth, CA) at room temperature. Operational stabilities of the devices were tested at room temperature by driving a direct current of 80 mA/cm2 through the devices. Example 1 (Comparative) The preparation of a conventional OLED is as follows: A ~1.1 mm thick glass substrate coated with a transparent ITO conductive layer was cleaned and dried using a commercial glass scrubber tool. The thickness of ITO is about 42 nm and the sheet resistance of the ITO is about 68 Ω/square. The ITO surface was subsequently treated with oxidative plasma to condition the surface as an anode. A layer of CFx, 1 nm thick, was deposited on the clean ITO surface as the anode buffer layer by decomposing CHF3 gas in an RF plasma treatment chamber.
The substrate was then transferred into a vacuum deposition chamber for deposition of all other layers on top of the substrate. The following layers were deposited in the following sequence by evaporation from a heated boat under a vacuum of approximately 10"6 Torn 1. EL Unit: a) an HIL, 10 run thick, composed of hexaazatriphenylene hexacarbonitrile (HAT-CN); b) an HTL, 65 nm thick, composed of 4,4'-bis[N-(l -naρhthyl)-N- phenylamino]biphenyl (NPB); c) a LEL, 30 nm thick, composed of tris(8-hydroxyquinoline)- aluminum (AIq) doped with 1.0 vol% 10-(2-benzothiazolyl)-l,l,7,7-tetramethyl- 2,3,6,7-tetrahydro-lH,5H,l lH(l)benzopyrano(6,7,8-ij)quinolizin-l 1-one (C545T); and d) an EIL, 30 nm thick, composed of AIq doped with about 1.2 vol% lithium.
2. Cathode: approximately 210 nm thick, composed of Mg:Ag (formed by co- evaporation of about 95 vol% Mg and 5 vol% Ag).
After the deposition of these layers, the device was transferred from the deposition chamber into a dry box (made by VAC Vacuum Atmosphere Company, Hawthorne, CA) for encapsulation. The OLED has an emission area of 10 mm2. The reduction potentials of HAT-CN and AIq were measured as about -0.01 V and -1.77 V vs. SCE in the 1:1 MeCN/toluene organic solvent system, respectively.
This conventional OLED requires a drive voltage of about 5.0 V to pass 20 mA/cm . Under this test condition, the device has a luminance of
1905 cd/m2, and a luminous efficiency of about 9.5 cd/A. Its emission peak is at
521 nm.
Example 2 (Comparative)
Another OLED was constructed as the same as that in Example 1, except that the 30-nm-thick Li-doped AIq layer (the EIL formed as layer d) in Example 1 was replaced by a 30-nm-thick HAT-CN layer as an EIL Example 2.
This OLED requires a drive voltage of about 5.4 V to pass 20 mA/cm2. However, there is no light emission from this device. This observation indicates that the HAT-CN layer having a reduction potential greater than -1.0 V vs. SCE cannot be effectively used as an EIL in this device configuration.
Example 3 (Comparative)
Another OLED was constructed as the same as that in Example I5 except that the 30-nm-thick Li-doped AIq layer (the EIL, layer d) in Example 1 was replaced by a 30-nm-thick HAT-CN layer doped with about 1.2 vol% lithium as an EIL in Example 3.
This OLED requires a drive voltage of about 5.2 V to pass 20 mA/cm2. However, there is no light emission from this device. This observation indicates that the Li-doped HAT-CN layer also cannot be effectively used as an EIL in this device configuration. Example 4 (Comparative)
Another OLED was constructed in the same manner as that in Example 1. However, the layer structure of the EL unit is as follows: a) an HTL, 75 nm thick, composed of NPB; b) a LEL, 30 nm thick, composed of AIq doped with 1.0 vol% C545T; and c) an ETL, 30 nm thick, composed of AIq.
This conventional OLED requires a drive voltage of about 6.6 V to pass 20 mA/cm . Under this test condition, the device has a luminance of 1977 cd/m , and a luminous efficiency of about 9.9 cd/A. Its emission peak is at 522 nm. The operational lifetime was measured as T50(RT@80 mA/cm2) (i.e. the time at which the luminance has fallen to 50% of its initial value after being operated at room temperature and at 80 mA/cm2). Its T50(RT@80 mA/cm2) is about 130 hours. The average rate of the voltage rise during the lifetime testing is about 17 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm2, are shown in FIGS. 6 and 7, respectively.
Example 5 (Comparative)
Another OLED was constructed as the same as that in Example 4, except that the layer c in Example 4 was replaced by: c) an EIL, 30 nm thick, composed of AIq doped with about 1.2 vol% lithium.
This OLED, requires a drive voltage of about 4.6 V to pass 20 mA/cm . Under this test condition, the device has a luminance of 2008 cd/m , and a luminous efficiency of about 10.0 cd/A. Its emission peak is at 522 nm. The operational lifetime, measured as T50(RT@80 mA/cm2), is about 246 hours. The average rate of the voltage rise during the lifetime testing is about 5.0 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm2, are shown in FIGS. 6 and 7, respectively. Example 6 (Inventive) An OLED in accordance with the present invention was constructed as the same as that in Example 4, except that the layer c in Example 4 was replaced by: c) the 1st EIL, 29 nm thick, composed of AIq doped with about 1.2 vol% lithium; and d) the 2nd EIL, 1 nm thick, composed of HAT-CN.
This OLED, having the 2nd EIL in direct contact with the 1st EIL, requires a drive voltage of about 5.0 V to pass 20 mA/cm2. Under this test condition, the device has a luminance of 1891 cd/m2, and a luminous efficiency of about 9.5 cd/A. Its emission peak is at 521 nm. The operational lifetime, measured as T50(RT@80 mA/cm2), is about 312 hours. The average rate of the voltage rise during the lifetime testing is about 3.5 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm2, are shown in FIGS. 6 and 7, respectively.
FIGS. 6 and 7 demonstrate that the OLED constructed in accordance with the present invention can have improved lifetime and improved
voltage rise. The following examples will show how one can further reduce the drive voltage. Example 7 (Inventive)
Another OLED in accordance with the present invention was constructed in the same general manner as that in Example 1. But, the layer structure of the EL unit is as follows: a) an HIL, 10 nm thick, composed of HAT-CN; b) an HTL, 65 nm thick, composed of NPB; c) a LEL , 30 nm thick, composed of AIq doped with 1.0 vol% C545T; d) the 1st EIL, 20 nm thick, composed of AIq doped with about 1.2 vol% lithium; and e) the 2nd EIL, 10 nm thick, composed of HAT-CN.
This OLED requires a drive voltage of about 5.1 V to pass 20 mA/cm . Under this test condition, the device has a luminance of 1823 cd/m , and a luminous efficiency of about 9.1 cd/A. Its emission peak is at 521 nm. The operational lifetime, measured as T50(RT@80 mA/cm2), is about 350 hours. The average rate of the voltage rise during the lifetime testing is about 2.9 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm2, are shown in FIGS. 8 and 9, respectively. Example 8 (Inventive)
Another OLED was constructed in accordance with the present invention. This OLED is the same as that in Example 7, except that layers d and e were changed as follows: d) the 1st EIL, 5 nm thick, composed of AIq doped with about 1.2 vol% lithium; and e) the 2nd EIL, 25 nm thick, composed of HAT-CN doped with about 1.2 vol% lithium. This OLED requires a drive voltage of about 4.6 V to pass
20 mA/cm . Under this test condition, the device has a luminance of 1898 cd/m ,
and a luminous efficiency of about 9.5 cd/A. Its emission peak is at 521 nm. The operational lifetime, measured as Tso(RT@8O mA/cm2), is about 330 hours. The average rate of the voltage rise during the lifetime testing is about 2.1 mV/hr. Its normalized luminance vs. operational time and its drive voltage vs. operational time, tested at room temperature and at 80 mA/cm2, are shown in FIGS. 8 and 9, respectively.
It is evident from FIGS. 8 and 9 that the OLEDs having a Li-doped HAT-CN layer as the 2nd EIL can further reduce both the drive voltage and the voltage rise during operation.
PARTS LIST
100 OLED
110 substrate
120 anode
130 hole-injecting layer (HIL)
140 hole-transporting layer (HTL)
150 light-emitting layer (LEL)
160 electron-transporting layer (ETL)
170 electron-injecting layer (EIL)
180 cathode
191 electrical conductors
192 voltage/current source
200 OLED
271 1st electron-injecting layer (1st EIL)
272 2nd electron-injecting layer (2nd EIL)
300 OLED
400 OLED
500 OLED
Claims
1. An organic light-emitting device (OLED) comprising: a) an anode; b) a light-emitting layer disposed over the anode; c) a first electron-injecting layer disposed over the light- emitting layer, wherein the first electron-injecting layer includes at least one organic host material having a reduction potential less than -1.0 V vs. a Saturated Calomel Electrode and at least one dopant material capable of reducing the organic host material; d) a second electron-injecting layer disposed in contact with the first electron-injecting layer, wherein the second electron-injecting layer includes at least one organic material having a reduction potential greater than -1.0 V vs. a Saturated Calomel Electrode; and e) a cathode disposed over the second electron-injecting layer.
2. The OLED of claim 1 wherein the at least one organic material in the second electron-injecting layer has a reduction potential greater than -0.5 V vs. a Saturated Calomel Electrode and has an optical band gap greater than 1.6 eV.
3. The OLED of claim 1 wherein the first electron-injecting layer has a thickness in a range of from 2 to 150 nm.
4. The OLED of claim 1 wherein the second electron-injecting layer has a thickness in a range of from 1 to 150 nm.
5. The OLED of claim 1 wherein the at least one organic host material in the first electron-injecting layer is selected from metal chelated oxinoid compounds, 8-hydroxy-quinoline derivatives, butadiene derivatives, heterocyclic optical brighteners, triazines, benzazoles, phenanthroline derivatives, or silole derivatives.
6. The OLED of claim 1 wherein the at least one organic host material in the first electron-injecting layer includes tris(8-hydroxyquinoline)- aluminum (AIq), 4,7-diphenyl-l,10-phenanthroline (Bphen), 2,9-dimethyl-4,7- diphenyl-l,10-ρhenantliroline (BCP), or 2,2'-[l,l'-biphenyl]-4,4'-diylbis[4,6-(p- toryl)-l,3,5-triazine] (TRAZ).
7. The OLED of claim 1 wherein the at least one dopant material in the first electron-injecting layer includes an alkali metal, alkali metal compound, alkaline earth metal, or alkaline earth metal compound.
8. The OLED of claim 1 wherein the at least one dopant material in the first electron-injecting layer includes Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Nd, Sm, Eu, Tb, Dy, or Yb.
9. The OLED of claim 1 wherein the at least one organic material in the second electron-injecting layer is selected from the chemical compounds having the structure of
wherein R1-R4 represent hydrogen, or substituents independently selected from the group including halo, nitrile (-CN), nitro (-NO2), sulfonyl (-SO2R), sulfoxide (-SOR), trifluoromethyl (-CF3), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein R1 and R2, or R3 and R4, combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
10. The OLED of claim 9 wherein the at least one organic material in the second electron-injecting layer includes a chemical compound
or
11. The OLED of claim 1 wherein the at least one organic material in the second electron-injecting layer includes a chemical compound
or a derivative any of these compounds resulting from replacement of one or more hydrogen atoms by substituents including of halo, nitrile (-CN), nitro (-NO2), sulfonyl (-SO2R), sulfoxide (-SOR), trifluoromethyl (-CF3), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein two or more substituents combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
12. The OLED of claim 1 wherein the at least one organic material in the second electron-injecting layer is selected from the chemical compounds having the structure of wherein R1-R6 represent hydrogen or a substituent independently selected from the group including halo, nitrile (-CN), nitro (-NO2), sulfonyl (-SO2R), sulfoxide (-SOR), trifluoromethyl (-CF3), ester (-CO-OR), amide (-CO-NHR or -CO-NRR'), substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, and substituted or unsubstituted alkyl, where R and R' include substituted or unsubstituted alkyl or aryl; or wherein R1 and R2, R3 and R4, or R5 and R6, combine form a ring structure including an aromatic ring, a heteroaromatic ring, or a nonaromatic ring, and each ring is substituted or unsubstituted.
13. The OLED of claim 12 wherein the at least one organic material in the second electron-injecting layer includes a chemical compound
14. The OLED of claim 1 wherein the at least one organic material in the second electron-injecting layer includes a chemical compound
15. The OLED of claim 1 wherein the second electron-injecting layer includes at least one organic material having a reduction potential greater than -1.0 V vs. SCE and at least one dopant material having a work function less than 4.0 eV.
16. The OLED of claim 14 wherein the at least one dopant material in the second electron-injecting layer includes an alkali metal, alkali metal compound, alkaline earth metal, or alkaline earth metal compound.
17. The OLED of claim 14 wherein the at least one dopant material in the second electron-injecting layer includes Li, Na, K, Rb, Cs, Mg, Ca, Sr, Ba, La, Ce, Nd, Sm, Eu, Tb, Dy, or Yb.
18. The OLED of claim 14 wherein the concentration of the at least one dopant material in the second electron-injecting layer is in the range of from 0.01% to 20% by volume.
19. The OLED of claim 1 wherein the device emits a red, green, blue, or white color.
20. An OLED display including a plurality of OLEDs according to claim 1.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/US2006/017555 WO2007130047A1 (en) | 2006-05-08 | 2006-05-08 | Oled electron-injecting layer |
| EP11003715.7A EP2355198B1 (en) | 2006-05-08 | 2006-05-08 | OLED electron-injecting layer |
| EP06759222A EP2016633A1 (en) | 2006-05-08 | 2006-05-08 | Oled electron-injecting layer |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/US2006/017555 WO2007130047A1 (en) | 2006-05-08 | 2006-05-08 | Oled electron-injecting layer |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007130047A1 true WO2007130047A1 (en) | 2007-11-15 |
Family
ID=37568955
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2006/017555 WO2007130047A1 (en) | 2006-05-08 | 2006-05-08 | Oled electron-injecting layer |
Country Status (2)
| Country | Link |
|---|---|
| EP (2) | EP2016633A1 (en) |
| WO (1) | WO2007130047A1 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2192634A1 (en) * | 2008-12-01 | 2010-06-02 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| WO2010073348A1 (en) * | 2008-12-25 | 2010-07-01 | 富士電機ホールディングス株式会社 | Organic el element having cathode buffer layer |
| US20110042662A1 (en) * | 2008-03-31 | 2011-02-24 | Sumitomo Chemical Company, Limited | Organic electroluminescence element, and method for production thereof |
| GB2485050A (en) * | 2010-10-25 | 2012-05-02 | Lg Display Co Ltd | Organic light emitting diode and method of fabricating the same |
| US8389979B2 (en) | 2009-05-29 | 2013-03-05 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US8431940B2 (en) | 2008-12-01 | 2013-04-30 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, lighting device, and electronic device |
| WO2013145667A1 (en) * | 2012-03-29 | 2013-10-03 | ソニー株式会社 | Organic electroluminescence element |
| US8993126B2 (en) | 2010-06-25 | 2015-03-31 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element including a layer having a concentration gradient, light-emitting device, display, and electronic device |
| US9012041B2 (en) | 2010-03-08 | 2015-04-21 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US9023491B2 (en) | 2010-03-23 | 2015-05-05 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US9048439B2 (en) | 2010-03-31 | 2015-06-02 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device comprising a metal complex having a metal-oxygen bond and an aromatic ligand |
| US9276221B2 (en) | 2010-03-31 | 2016-03-01 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device comprising a phthalocyanine-based material |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4356429A (en) | 1980-07-17 | 1982-10-26 | Eastman Kodak Company | Organic electroluminescent cell |
| EP1099744A2 (en) | 1999-11-12 | 2001-05-16 | Canon Kabushiki Kaisha | Organic electroluminescent device |
| US20050104511A1 (en) | 2003-11-14 | 2005-05-19 | Eastman Kodak Company | Organic electroluminescent devices having a stability-enhancing layer |
| WO2006091560A1 (en) | 2005-02-23 | 2006-08-31 | Eastman Kodak Company | Tandem oled having an organic intermediate connector |
Family Cites Families (119)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NL124075C (en) | 1959-04-09 | |||
| US3658520A (en) | 1968-02-20 | 1972-04-25 | Eastman Kodak Co | Photoconductive elements containing as photoconductors triarylamines substituted by active hydrogen-containing groups |
| US3567450A (en) | 1968-02-20 | 1971-03-02 | Eastman Kodak Co | Photoconductive elements containing substituted triarylamine photoconductors |
| US4539507A (en) | 1983-03-25 | 1985-09-03 | Eastman Kodak Company | Organic electroluminescent devices having improved power conversion efficiencies |
| DE3518323A1 (en) | 1985-05-22 | 1986-11-27 | SEVAR Entsorgungsanlagen GmbH, 8590 Marktredwitz | METHOD AND DEVICE FOR DRYING CLEANING SLUDGE |
| US5283182A (en) | 1986-09-17 | 1994-02-01 | Beecham Group Plc | Preparation of immobilized hydantoinase stabilized with divalent metal ions |
| US4885221A (en) | 1986-12-06 | 1989-12-05 | Kabushiki Kaisha Toshiba | Electrophotography apparatus and electrophtographic process for developing positive image from positive or negative film |
| US4885211A (en) | 1987-02-11 | 1989-12-05 | Eastman Kodak Company | Electroluminescent device with improved cathode |
| US4720432A (en) | 1987-02-11 | 1988-01-19 | Eastman Kodak Company | Electroluminescent device with organic luminescent medium |
| US5121029A (en) | 1987-12-11 | 1992-06-09 | Idemitsu Kosan Co., Ltd. | Electroluminescence device having an organic electroluminescent element |
| GB8909011D0 (en) | 1989-04-20 | 1989-06-07 | Friend Richard H | Electroluminescent devices |
| US5061569A (en) | 1990-07-26 | 1991-10-29 | Eastman Kodak Company | Electroluminescent device with organic electroluminescent medium |
| US5059861A (en) | 1990-07-26 | 1991-10-22 | Eastman Kodak Company | Organic electroluminescent device with stabilizing cathode capping layer |
| US5059862A (en) | 1990-07-26 | 1991-10-22 | Eastman Kodak Company | Electroluminescent device with improved cathode |
| US5093698A (en) | 1991-02-12 | 1992-03-03 | Kabushiki Kaisha Toshiba | Organic electroluminescent device |
| US5151629A (en) | 1991-08-01 | 1992-09-29 | Eastman Kodak Company | Blue emitting internal junction organic electroluminescent device (I) |
| US5141671A (en) | 1991-08-01 | 1992-08-25 | Eastman Kodak Company | Mixed ligand 8-quinolinolato aluminum chelate luminophors |
| US5150006A (en) | 1991-08-01 | 1992-09-22 | Eastman Kodak Company | Blue emitting internal junction organic electroluminescent device (II) |
| US5294870A (en) | 1991-12-30 | 1994-03-15 | Eastman Kodak Company | Organic electroluminescent multicolor image display device |
| US5276380A (en) | 1991-12-30 | 1994-01-04 | Eastman Kodak Company | Organic electroluminescent image display device |
| JP3445315B2 (en) | 1992-07-13 | 2003-09-08 | イーストマン コダック カンパニー | Aluminum chelate compound and internal junction type organic electroluminescent device |
| US5405709A (en) | 1993-09-13 | 1995-04-11 | Eastman Kodak Company | White light emitting internal junction organic electroluminescent device |
| JP3813217B2 (en) | 1995-03-13 | 2006-08-23 | パイオニア株式会社 | Method for manufacturing organic electroluminescence display panel |
| US5503910A (en) | 1994-03-29 | 1996-04-02 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device |
| US5703436A (en) | 1994-12-13 | 1997-12-30 | The Trustees Of Princeton University | Transparent contacts for organic devices |
| US5608287A (en) | 1995-02-23 | 1997-03-04 | Eastman Kodak Company | Conductive electron injector for light-emitting diodes |
| US5593788A (en) | 1996-04-25 | 1997-01-14 | Eastman Kodak Company | Organic electroluminescent devices with high operational stability |
| US5688551A (en) | 1995-11-13 | 1997-11-18 | Eastman Kodak Company | Method of forming an organic electroluminescent display panel |
| JP4477150B2 (en) | 1996-01-17 | 2010-06-09 | 三星モバイルディスプレイ株式會社 | Organic thin film EL device |
| US5683823A (en) | 1996-01-26 | 1997-11-04 | Eastman Kodak Company | White light-emitting organic electroluminescent devices |
| US5776623A (en) | 1996-07-29 | 1998-07-07 | Eastman Kodak Company | Transparent electron-injecting electrode for use in an electroluminescent device |
| US5677572A (en) | 1996-07-29 | 1997-10-14 | Eastman Kodak Company | Bilayer electrode on a n-type semiconductor |
| US5776622A (en) | 1996-07-29 | 1998-07-07 | Eastman Kodak Company | Bilayer eletron-injeting electrode for use in an electroluminescent device |
| US5645948A (en) | 1996-08-20 | 1997-07-08 | Eastman Kodak Company | Blue organic electroluminescent devices |
| US5714838A (en) | 1996-09-20 | 1998-02-03 | International Business Machines Corporation | Optically transparent diffusion barrier and top electrode in organic light emitting diode structures |
| JPH10125469A (en) | 1996-10-24 | 1998-05-15 | Tdk Corp | Organic electroluminescent element |
| DE69723538T2 (en) | 1996-11-29 | 2004-06-09 | Idemitsu Kosan Co. Ltd. | Organic electroluminescent device |
| US5981306A (en) | 1997-09-12 | 1999-11-09 | The Trustees Of Princeton University | Method for depositing indium tin oxide layers in organic light emitting devices |
| JP3654909B2 (en) | 1996-12-28 | 2005-06-02 | Tdk株式会社 | Organic EL device |
| US5739545A (en) | 1997-02-04 | 1998-04-14 | International Business Machines Corporation | Organic light emitting diodes having transparent cathode structures |
| US5935720A (en) | 1997-04-07 | 1999-08-10 | Eastman Kodak Company | Red organic electroluminescent devices |
| US5928802A (en) | 1997-05-16 | 1999-07-27 | Eastman Kodak Company | Efficient blue organic electroluminescent devices |
| US5755999A (en) | 1997-05-16 | 1998-05-26 | Eastman Kodak Company | Blue luminescent materials for organic electroluminescent devices |
| GB9711237D0 (en) | 1997-06-02 | 1997-07-23 | Isis Innovation | Organomettallic Complexes |
| US6337492B1 (en) | 1997-07-11 | 2002-01-08 | Emagin Corporation | Serially-connected organic light emitting diode stack having conductors sandwiching each light emitting layer |
| US6451455B1 (en) | 1998-04-01 | 2002-09-17 | The Trustees Of Princeton University | Metal complexes bearing both electron transporting and hole transporting moieties |
| US6413656B1 (en) | 1998-09-14 | 2002-07-02 | The University Of Southern California | Reduced symmetry porphyrin molecules for producing enhanced luminosity from phosphorescent organic light emitting devices |
| US5851709A (en) | 1997-10-31 | 1998-12-22 | Eastman Kodak Company | Method for selective transfer of a color organic layer |
| JPH11251067A (en) | 1998-03-02 | 1999-09-17 | Junji Kido | Organic electroluminescence element |
| US5972247A (en) | 1998-03-20 | 1999-10-26 | Eastman Kodak Company | Organic electroluminescent elements for stable blue electroluminescent devices |
| US5935721A (en) | 1998-03-20 | 1999-08-10 | Eastman Kodak Company | Organic electroluminescent elements for stable electroluminescent |
| US6172459B1 (en) | 1998-07-28 | 2001-01-09 | Eastman Kodak Company | Electron-injecting layer providing a modified interface between an organic light-emitting structure and a cathode buffer layer |
| US6137223A (en) | 1998-07-28 | 2000-10-24 | Eastman Kodak Company | Electron-injecting layer formed from a dopant layer for organic light-emitting structure |
| US6140763A (en) | 1998-07-28 | 2000-10-31 | Eastman Kodak Company | Interfacial electron-injecting layer formed from a doped cathode for organic light-emitting structure |
| US6451415B1 (en) | 1998-08-19 | 2002-09-17 | The Trustees Of Princeton University | Organic photosensitive optoelectronic device with an exciton blocking layer |
| EP1029909A4 (en) | 1998-09-09 | 2007-01-10 | Idemitsu Kosan Co | Organic electroluminescence device and phenylenediamine derivative |
| US6830828B2 (en) | 1998-09-14 | 2004-12-14 | The Trustees Of Princeton University | Organometallic complexes as phosphorescent emitters in organic LEDs |
| US6097147A (en) | 1998-09-14 | 2000-08-01 | The Trustees Of Princeton University | Structure for high efficiency electroluminescent device |
| GB9820805D0 (en) | 1998-09-25 | 1998-11-18 | Isis Innovation | Divalent lanthanide metal complexes |
| US6208075B1 (en) | 1998-11-05 | 2001-03-27 | Eastman Kodak Company | Conductive fluorocarbon polymer and method of making same |
| US6208077B1 (en) | 1998-11-05 | 2001-03-27 | Eastman Kodak Company | Organic electroluminescent device with a non-conductive fluorocarbon polymer layer |
| US6361886B2 (en) | 1998-12-09 | 2002-03-26 | Eastman Kodak Company | Electroluminescent device with improved hole transport layer |
| US6465115B2 (en) | 1998-12-09 | 2002-10-15 | Eastman Kodak Company | Electroluminescent device with anthracene derivatives hole transport layer |
| US6020078A (en) | 1998-12-18 | 2000-02-01 | Eastman Kodak Company | Green organic electroluminescent devices |
| US6066357A (en) | 1998-12-21 | 2000-05-23 | Eastman Kodak Company | Methods of making a full-color organic light-emitting display |
| KR20050084516A (en) | 1998-12-28 | 2005-08-26 | 이데미쓰 고산 가부시키가이샤 | Organic electroluminescent element |
| US6127004A (en) | 1999-01-29 | 2000-10-03 | Eastman Kodak Company | Forming an amorphous fluorocarbon layer in electroluminescent devices |
| WO2000057676A1 (en) | 1999-03-23 | 2000-09-28 | The University Of Southern California | Cyclometallated metal complexes as phosphorescent dopants in organic leds |
| KR100913568B1 (en) | 1999-05-13 | 2009-08-26 | 더 트러스티즈 오브 프린스턴 유니버시티 | Very high efficiency organic light emitting devices based on electrophosphorescence |
| US6521360B2 (en) | 1999-06-08 | 2003-02-18 | City University Of Hong Kong | White and colored organic electroluminescent devices using single emitting material by novel color change technique |
| US6310360B1 (en) | 1999-07-21 | 2001-10-30 | The Trustees Of Princeton University | Intersystem crossing agents for efficient utilization of excitons in organic light emitting devices |
| EP1076368A2 (en) | 1999-08-11 | 2001-02-14 | Eastman Kodak Company | A surface-emitting organic light-emitting diode |
| US6278236B1 (en) | 1999-09-02 | 2001-08-21 | Eastman Kodak Company | Organic electroluminescent devices with electron-injecting layer having aluminum and alkali halide |
| KR100790663B1 (en) | 1999-09-21 | 2008-01-03 | 이데미쓰 고산 가부시키가이샤 | Organic electroluminescence and organic luminous medium |
| US6458475B1 (en) | 1999-11-24 | 2002-10-01 | The Trustee Of Princeton University | Organic light emitting diode having a blue phosphorescent molecule as an emitter |
| CN1840607B (en) | 1999-12-01 | 2010-06-09 | 普林斯顿大学理事会 | Complexes of form l2mx as phosphorescent dopants for organic LEDs |
| JP4255610B2 (en) | 1999-12-28 | 2009-04-15 | 出光興産株式会社 | White organic electroluminescence device |
| KR100377321B1 (en) | 1999-12-31 | 2003-03-26 | 주식회사 엘지화학 | Electronic device comprising organic compound having p-type semiconducting characteristics |
| US6237529B1 (en) | 2000-03-03 | 2001-05-29 | Eastman Kodak Company | Source for thermal physical vapor deposition of organic electroluminescent layers |
| DE60140720D1 (en) | 2000-03-29 | 2010-01-21 | Idemitsu Kosan Co | ANTHRACEN DERIVATIVES AND ORGANIC ELECTROLUMINESCENT DEVICE PRODUCED BY USING THESE DERIVATIVES |
| JP4094203B2 (en) | 2000-03-30 | 2008-06-04 | 出光興産株式会社 | Organic electroluminescence device and organic light emitting medium |
| JP4037033B2 (en) | 2000-03-31 | 2008-01-23 | パイオニア株式会社 | Organic electroluminescence device |
| US6226890B1 (en) | 2000-04-07 | 2001-05-08 | Eastman Kodak Company | Desiccation of moisture-sensitive electronic devices |
| US6645645B1 (en) | 2000-05-30 | 2003-11-11 | The Trustees Of Princeton University | Phosphorescent organic light emitting devices |
| US6475648B1 (en) | 2000-06-08 | 2002-11-05 | Eastman Kodak Company | Organic electroluminescent devices with improved stability and efficiency |
| US20020121638A1 (en) | 2000-06-30 | 2002-09-05 | Vladimir Grushin | Electroluminescent iridium compounds with fluorinated phenylpyridines, phenylpyrimidines, and phenylquinolines and devices made with such compounds |
| CN100505375C (en) | 2000-08-11 | 2009-06-24 | 普林斯顿大学理事会 | Organometallic compounds and emission-shifting organic electrophosphorescence |
| US6696177B1 (en) | 2000-08-30 | 2004-02-24 | Eastman Kodak Company | White organic electroluminescent devices with improved stability and efficiency |
| JP4086498B2 (en) | 2000-11-29 | 2008-05-14 | キヤノン株式会社 | Metal coordination compound, light emitting device and display device |
| AU2002222565A1 (en) | 2000-11-30 | 2002-06-11 | Canon Kabushiki Kaisha | Luminescent element and display |
| KR100825183B1 (en) | 2000-11-30 | 2008-04-24 | 캐논 가부시끼가이샤 | Luminescent Element and Display |
| JP4154145B2 (en) | 2000-12-01 | 2008-09-24 | キヤノン株式会社 | Metal coordination compound, light emitting device and display device |
| US6573651B2 (en) | 2000-12-18 | 2003-06-03 | The Trustees Of Princeton University | Highly efficient OLEDs using doped ambipolar conductive molecular organic thin films |
| US6693295B2 (en) | 2000-12-25 | 2004-02-17 | Fuji Photo Film Co., Ltd. | Indole derivative, material for light-emitting device and light-emitting device using the same |
| US6720090B2 (en) | 2001-01-02 | 2004-04-13 | Eastman Kodak Company | Organic light emitting diode devices with improved luminance efficiency |
| WO2002071813A1 (en) | 2001-03-02 | 2002-09-12 | The Trustees Of Princeton University | Double doped-layer, phosphorescent organic light emitting devices |
| JP4307000B2 (en) | 2001-03-08 | 2009-08-05 | キヤノン株式会社 | Metal coordination compound, electroluminescent element and display device |
| JP4438042B2 (en) | 2001-03-08 | 2010-03-24 | キヤノン株式会社 | Metal coordination compound, electroluminescent element and display device |
| KR100916231B1 (en) | 2001-03-14 | 2009-09-08 | 더 트러스티즈 오브 프린스턴 유니버시티 | Materials and devices for blue phosphorescence based organic light emitting diodes |
| JP4307001B2 (en) | 2001-03-14 | 2009-08-05 | キヤノン株式会社 | Metal coordination compound, electroluminescent element and display device |
| US6597012B2 (en) | 2001-05-02 | 2003-07-22 | Junji Kido | Organic electroluminescent device |
| KR100888424B1 (en) | 2001-05-16 | 2009-03-11 | 더 트러스티즈 오브 프린스턴 유니버시티 | High efficiency multi-color electro- phosphorescent oleds |
| US7012588B2 (en) | 2001-06-05 | 2006-03-14 | Eastman Kodak Company | Method for saving power in an organic electroluminescent display using white light emitting elements |
| EP1399002A4 (en) | 2001-06-15 | 2007-11-21 | Canon Kk | Organic electroluminescnece device |
| US7067202B2 (en) | 2001-06-15 | 2006-06-27 | Sanyo Electric Co., Ltd. | Luminescent organometallic compound and light emitting device |
| US6627333B2 (en) | 2001-08-15 | 2003-09-30 | Eastman Kodak Company | White organic light-emitting devices with improved efficiency |
| KR100917347B1 (en) | 2001-08-29 | 2009-09-16 | 더 트러스티즈 오브 프린스턴 유니버시티 | Organic light emitting devices having carrier blocking layers comprising metal complexs |
| US6835469B2 (en) | 2001-10-17 | 2004-12-28 | The University Of Southern California | Phosphorescent compounds and devices comprising the same |
| US6869695B2 (en) | 2001-12-28 | 2005-03-22 | The Trustees Of Princeton University | White light emitting OLEDs from combined monomer and aggregate emission |
| US6863997B2 (en) | 2001-12-28 | 2005-03-08 | The Trustees Of Princeton University | White light emitting OLEDs from combined monomer and aggregate emission |
| US6872472B2 (en) | 2002-02-15 | 2005-03-29 | Eastman Kodak Company | Providing an organic electroluminescent device having stacked electroluminescent units |
| US6824893B2 (en) | 2002-02-28 | 2004-11-30 | Eastman Kodak Company | Organic element for electroluminescent devices |
| US6661023B2 (en) | 2002-02-28 | 2003-12-09 | Eastman Kodak Company | Organic element for electroluminescent devices |
| US6680132B2 (en) | 2002-03-25 | 2004-01-20 | Eastman Kodak Company | Red organic electroluminescent devices |
| US6841267B2 (en) | 2002-04-24 | 2005-01-11 | Eastman Kodak Company | Efficient electroluminescent device |
| US20040001969A1 (en) | 2002-06-27 | 2004-01-01 | Eastman Kodak Company | Device containing green organic light-emitting diode |
| US6720092B2 (en) | 2002-07-08 | 2004-04-13 | Eastman Kodak Company | White organic light-emitting devices using rubrene layer |
| KR100946476B1 (en) | 2002-08-23 | 2010-03-10 | 이데미쓰 고산 가부시키가이샤 | Organic electroluminescence device and anthracene derivative |
| US6717358B1 (en) | 2002-10-09 | 2004-04-06 | Eastman Kodak Company | Cascaded organic electroluminescent devices with improved voltage stability |
-
2006
- 2006-05-08 WO PCT/US2006/017555 patent/WO2007130047A1/en active Application Filing
- 2006-05-08 EP EP06759222A patent/EP2016633A1/en not_active Withdrawn
- 2006-05-08 EP EP11003715.7A patent/EP2355198B1/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4356429A (en) | 1980-07-17 | 1982-10-26 | Eastman Kodak Company | Organic electroluminescent cell |
| EP1099744A2 (en) | 1999-11-12 | 2001-05-16 | Canon Kabushiki Kaisha | Organic electroluminescent device |
| US20050104511A1 (en) | 2003-11-14 | 2005-05-19 | Eastman Kodak Company | Organic electroluminescent devices having a stability-enhancing layer |
| WO2006091560A1 (en) | 2005-02-23 | 2006-08-31 | Eastman Kodak Company | Tandem oled having an organic intermediate connector |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110042662A1 (en) * | 2008-03-31 | 2011-02-24 | Sumitomo Chemical Company, Limited | Organic electroluminescence element, and method for production thereof |
| JP2017175167A (en) * | 2008-12-01 | 2017-09-28 | 株式会社半導体エネルギー研究所 | Light-emitting device, light-emitting apparatus, electronic apparatus and illumination device |
| US8158991B2 (en) | 2008-12-01 | 2012-04-17 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US11139445B2 (en) | 2008-12-01 | 2021-10-05 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| EP2192634A1 (en) * | 2008-12-01 | 2010-06-02 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US8431940B2 (en) | 2008-12-01 | 2013-04-30 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, lighting device, and electronic device |
| US10403843B2 (en) | 2008-12-01 | 2019-09-03 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element comprising the first to third layer and EL layer |
| US8581266B2 (en) | 2008-12-01 | 2013-11-12 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, lighting device, and electronic device |
| US8835973B2 (en) | 2008-12-01 | 2014-09-16 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US8952394B2 (en) | 2008-12-01 | 2015-02-10 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, lighting device, and electronic device |
| WO2010073348A1 (en) * | 2008-12-25 | 2010-07-01 | 富士電機ホールディングス株式会社 | Organic el element having cathode buffer layer |
| US8389979B2 (en) | 2009-05-29 | 2013-03-05 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US9012041B2 (en) | 2010-03-08 | 2015-04-21 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US9023491B2 (en) | 2010-03-23 | 2015-05-05 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device |
| US9048439B2 (en) | 2010-03-31 | 2015-06-02 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device comprising a metal complex having a metal-oxygen bond and an aromatic ligand |
| US9276221B2 (en) | 2010-03-31 | 2016-03-01 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element, light-emitting device, electronic device, and lighting device comprising a phthalocyanine-based material |
| US8993126B2 (en) | 2010-06-25 | 2015-03-31 | Semiconductor Energy Laboratory Co., Ltd. | Light-emitting element including a layer having a concentration gradient, light-emitting device, display, and electronic device |
| GB2485050A (en) * | 2010-10-25 | 2012-05-02 | Lg Display Co Ltd | Organic light emitting diode and method of fabricating the same |
| US9978975B2 (en) | 2012-03-29 | 2018-05-22 | Joled Inc | Organic electroluminescence device |
| WO2013145667A1 (en) * | 2012-03-29 | 2013-10-03 | ソニー株式会社 | Organic electroluminescence element |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2355198B1 (en) | 2015-09-09 |
| EP2355198A1 (en) | 2011-08-10 |
| EP2016633A1 (en) | 2009-01-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7629741B2 (en) | OLED electron-injecting layer | |
| EP1851801B1 (en) | Tandem oled having an organic intermediate connector | |
| EP2355198B1 (en) | OLED electron-injecting layer | |
| EP1704604B1 (en) | Using a crystallization-inhibitor in organic electroluminescent devices | |
| US8057916B2 (en) | OLED device with improved performance | |
| US7126267B2 (en) | Tandem OLED having stable intermediate connectors | |
| US6881502B2 (en) | Blue organic electroluminescent devices having a non-hole-blocking layer | |
| US20060240277A1 (en) | Tandem OLED device | |
| US20090004365A1 (en) | Contaminant-scavenging layer on oled anodes | |
| US7799439B2 (en) | Fluorocarbon electrode modification layer | |
| US20060240280A1 (en) | OLED anode modification layer | |
| US7075231B1 (en) | Tandem OLEDs having low drive voltage | |
| EP1668717B1 (en) | Metal sub-layer within a hole-transporting region | |
| US7138763B2 (en) | Organic electroluminescent devices having a stability-enhancing layer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 06759222 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006759222 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009509514 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |