WO2005077897A1 - Substituted azetidine compounds, their preparation and use as medicaments - Google Patents
Substituted azetidine compounds, their preparation and use as medicaments Download PDFInfo
- Publication number
- WO2005077897A1 WO2005077897A1 PCT/EP2005/001658 EP2005001658W WO2005077897A1 WO 2005077897 A1 WO2005077897 A1 WO 2005077897A1 EP 2005001658 W EP2005001658 W EP 2005001658W WO 2005077897 A1 WO2005077897 A1 WO 2005077897A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- optionally
- substituted
- mono
- linear
- Prior art date
Links
- 239000003814 drug Substances 0.000 title claims abstract description 49
- 150000001539 azetidines Chemical class 0.000 title claims abstract description 24
- 238000002360 preparation method Methods 0.000 title claims abstract description 23
- 150000001875 compounds Chemical class 0.000 claims abstract description 125
- 238000000034 method Methods 0.000 claims abstract description 32
- 238000011282 treatment Methods 0.000 claims abstract description 24
- -1 mono-substituted phenyl group Chemical group 0.000 claims description 241
- 125000001424 substituent group Chemical group 0.000 claims description 156
- 229910052801 chlorine Inorganic materials 0.000 claims description 154
- 229910052731 fluorine Inorganic materials 0.000 claims description 137
- 125000000217 alkyl group Chemical group 0.000 claims description 135
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 129
- 229910052794 bromium Inorganic materials 0.000 claims description 124
- 125000002950 monocyclic group Chemical class 0.000 claims description 124
- 229920006395 saturated elastomer Polymers 0.000 claims description 118
- 239000000460 chlorine Substances 0.000 claims description 115
- 125000001072 heteroaryl group Chemical group 0.000 claims description 114
- 125000003118 aryl group Chemical group 0.000 claims description 95
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 86
- 125000002947 alkylene group Chemical group 0.000 claims description 79
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 76
- 125000005842 heteroatom Chemical group 0.000 claims description 76
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 75
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 71
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 62
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 57
- 208000035475 disorder Diseases 0.000 claims description 57
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 53
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 claims description 51
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 49
- 239000000203 mixture Substances 0.000 claims description 47
- 125000001544 thienyl group Chemical class 0.000 claims description 44
- 125000002541 furyl group Chemical group 0.000 claims description 43
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 42
- 150000003839 salts Chemical class 0.000 claims description 38
- 125000001153 fluoro group Chemical group F* 0.000 claims description 36
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 35
- 150000001204 N-oxides Chemical class 0.000 claims description 34
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 31
- 239000012453 solvate Substances 0.000 claims description 31
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 30
- 239000012429 reaction media Substances 0.000 claims description 29
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 27
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 25
- 229910052757 nitrogen Inorganic materials 0.000 claims description 25
- 125000001931 aliphatic group Chemical class 0.000 claims description 24
- 238000002156 mixing Methods 0.000 claims description 24
- 229910052760 oxygen Inorganic materials 0.000 claims description 24
- PDQRQJVPEFGVRK-UHFFFAOYSA-N 2,1,3-benzothiadiazole Chemical compound C1=CC=CC2=NSN=C21 PDQRQJVPEFGVRK-UHFFFAOYSA-N 0.000 claims description 23
- 125000001624 naphthyl group Chemical group 0.000 claims description 23
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 claims description 22
- 125000005843 halogen group Chemical group 0.000 claims description 22
- UFBBWLWUIISIPW-UHFFFAOYSA-N imidazo[2,1-b][1,3]thiazole Chemical compound C1=CSC2=NC=CN21 UFBBWLWUIISIPW-UHFFFAOYSA-N 0.000 claims description 22
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 22
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 claims description 21
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 21
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 21
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 20
- 239000005864 Sulphur Chemical group 0.000 claims description 20
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 20
- 125000000623 heterocyclic group Chemical group 0.000 claims description 20
- 239000001301 oxygen Chemical group 0.000 claims description 20
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 19
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 19
- 125000002619 bicyclic group Chemical group 0.000 claims description 18
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 18
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 18
- 238000011321 prophylaxis Methods 0.000 claims description 18
- 102000018208 Cannabinoid Receptor Human genes 0.000 claims description 16
- 108050007331 Cannabinoid receptor Proteins 0.000 claims description 16
- 229930003827 cannabinoid Natural products 0.000 claims description 15
- 239000003557 cannabinoid Substances 0.000 claims description 15
- 208000011117 substance-related disease Diseases 0.000 claims description 13
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 12
- CIUQDSCDWFSTQR-UHFFFAOYSA-N [C]1=CC=CC=C1 Chemical compound [C]1=CC=CC=C1 CIUQDSCDWFSTQR-UHFFFAOYSA-N 0.000 claims description 12
- 229910052739 hydrogen Inorganic materials 0.000 claims description 12
- 239000001257 hydrogen Substances 0.000 claims description 12
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 claims description 12
- 102000005962 receptors Human genes 0.000 claims description 12
- 108020003175 receptors Proteins 0.000 claims description 12
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 11
- 125000001246 bromo group Chemical group Br* 0.000 claims description 10
- 125000004043 oxo group Chemical group O=* 0.000 claims description 10
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 claims description 9
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 9
- 206010013663 drug dependence Diseases 0.000 claims description 9
- 230000008569 process Effects 0.000 claims description 9
- 208000008589 Obesity Diseases 0.000 claims description 8
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 8
- 235000020824 obesity Nutrition 0.000 claims description 8
- 230000035939 shock Effects 0.000 claims description 8
- 208000007848 Alcoholism Diseases 0.000 claims description 7
- 210000000748 cardiovascular system Anatomy 0.000 claims description 7
- 210000003169 central nervous system Anatomy 0.000 claims description 7
- 210000001035 gastrointestinal tract Anatomy 0.000 claims description 7
- 210000000987 immune system Anatomy 0.000 claims description 7
- 210000002345 respiratory system Anatomy 0.000 claims description 7
- 208000028017 Psychotic disease Diseases 0.000 claims description 6
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 6
- 206010012601 diabetes mellitus Diseases 0.000 claims description 6
- 208000032841 Bulimia Diseases 0.000 claims description 5
- 206010006550 Bulimia nervosa Diseases 0.000 claims description 5
- 206010006895 Cachexia Diseases 0.000 claims description 5
- 206010012335 Dependence Diseases 0.000 claims description 5
- 206010012735 Diarrhoea Diseases 0.000 claims description 5
- 208000003251 Pruritus Diseases 0.000 claims description 5
- 230000000202 analgesic effect Effects 0.000 claims description 5
- 208000022531 anorexia Diseases 0.000 claims description 5
- 206010061428 decreased appetite Diseases 0.000 claims description 5
- 230000001419 dependent effect Effects 0.000 claims description 5
- 230000037406 food intake Effects 0.000 claims description 5
- 235000012631 food intake Nutrition 0.000 claims description 5
- 208000024827 Alzheimer disease Diseases 0.000 claims description 4
- 208000019901 Anxiety disease Diseases 0.000 claims description 4
- 208000020925 Bipolar disease Diseases 0.000 claims description 4
- 208000015879 Cerebellar disease Diseases 0.000 claims description 4
- 208000014094 Dystonic disease Diseases 0.000 claims description 4
- 208000010412 Glaucoma Diseases 0.000 claims description 4
- 208000001953 Hypotension Diseases 0.000 claims description 4
- 208000026139 Memory disease Diseases 0.000 claims description 4
- 208000019695 Migraine disease Diseases 0.000 claims description 4
- 208000016285 Movement disease Diseases 0.000 claims description 4
- 206010028980 Neoplasm Diseases 0.000 claims description 4
- 208000002193 Pain Diseases 0.000 claims description 4
- 206010033664 Panic attack Diseases 0.000 claims description 4
- 208000018737 Parkinson disease Diseases 0.000 claims description 4
- 208000003782 Raynaud disease Diseases 0.000 claims description 4
- 208000012322 Raynaud phenomenon Diseases 0.000 claims description 4
- 206010039966 Senile dementia Diseases 0.000 claims description 4
- 208000013738 Sleep Initiation and Maintenance disease Diseases 0.000 claims description 4
- 208000011641 Spinocerebellar disease Diseases 0.000 claims description 4
- 206010043118 Tardive Dyskinesia Diseases 0.000 claims description 4
- 206010047700 Vomiting Diseases 0.000 claims description 4
- 229910021529 ammonia Inorganic materials 0.000 claims description 4
- 239000000730 antalgic agent Substances 0.000 claims description 4
- 230000036506 anxiety Effects 0.000 claims description 4
- 208000006673 asthma Diseases 0.000 claims description 4
- 201000011510 cancer Diseases 0.000 claims description 4
- 208000010877 cognitive disease Diseases 0.000 claims description 4
- 208000010118 dystonia Diseases 0.000 claims description 4
- 231100000284 endotoxic Toxicity 0.000 claims description 4
- 230000002346 endotoxic effect Effects 0.000 claims description 4
- 206010015037 epilepsy Diseases 0.000 claims description 4
- 230000036543 hypotension Effects 0.000 claims description 4
- 230000001900 immune effect Effects 0.000 claims description 4
- 208000014674 injury Diseases 0.000 claims description 4
- 206010022437 insomnia Diseases 0.000 claims description 4
- 230000000968 intestinal effect Effects 0.000 claims description 4
- 206010027599 migraine Diseases 0.000 claims description 4
- 230000003533 narcotic effect Effects 0.000 claims description 4
- 208000015122 neurodegenerative disease Diseases 0.000 claims description 4
- 230000007823 neuropathy Effects 0.000 claims description 4
- 201000001119 neuropathy Diseases 0.000 claims description 4
- 230000036407 pain Effects 0.000 claims description 4
- 208000019906 panic disease Diseases 0.000 claims description 4
- 208000033808 peripheral neuropathy Diseases 0.000 claims description 4
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 4
- 230000001850 reproductive effect Effects 0.000 claims description 4
- 201000000980 schizophrenia Diseases 0.000 claims description 4
- 230000002784 sclerotic effect Effects 0.000 claims description 4
- 230000002992 thymic effect Effects 0.000 claims description 4
- 230000008733 trauma Effects 0.000 claims description 4
- 230000008673 vomiting Effects 0.000 claims description 4
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 claims description 3
- BNBGRXOGPFOPLL-QRQCRPRQSA-N n-[(2r,3s)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-fluorobenzenesulfonamide Chemical compound C([C@@H]([C@H]1C)NS(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 BNBGRXOGPFOPLL-QRQCRPRQSA-N 0.000 claims description 3
- BNBGRXOGPFOPLL-OYHNWAKOSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-fluorobenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 BNBGRXOGPFOPLL-OYHNWAKOSA-N 0.000 claims description 3
- NMZREQPYUSVIIX-MEDUHNTESA-N (2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-amine Chemical compound C[C@H]1[C@H](N)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 NMZREQPYUSVIIX-MEDUHNTESA-N 0.000 claims description 2
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical group C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 claims description 2
- XODIKZYDMAHSPS-UPCLLVRISA-N 4-acetyl-n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]benzenesulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(=CC=2)C(C)=O)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 XODIKZYDMAHSPS-UPCLLVRISA-N 0.000 claims description 2
- LFLSATHZMYYIAQ-UHFFFAOYSA-N 4-flourobenzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=C(F)C=C1 LFLSATHZMYYIAQ-UHFFFAOYSA-N 0.000 claims description 2
- WVSYONICNIDYBE-UHFFFAOYSA-N 4-fluorobenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(F)C=C1 WVSYONICNIDYBE-UHFFFAOYSA-N 0.000 claims description 2
- LNLYQVGIHBTFKG-QMHKHESXSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C2=C(C3=CC(Cl)=CC=C3S2)C)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C2=C(C3=CC(Cl)=CC=C3S2)C)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 LNLYQVGIHBTFKG-QMHKHESXSA-N 0.000 claims description 2
- KBZYHQABKNEVJK-AVRWGWEMSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C2=C(N(N=C2)C=2C=CC=CC=2)C)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C2=C(N(N=C2)C=2C=CC=CC=2)C)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 KBZYHQABKNEVJK-AVRWGWEMSA-N 0.000 claims description 2
- AJQTYRCYYGKDJI-KSFYIVLOSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C=2C3=CC=CC=C3SC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C3=CC=CC=C3SC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 AJQTYRCYYGKDJI-KSFYIVLOSA-N 0.000 claims description 2
- NQQZSKNYXUNFLL-HFJWLAOPSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=C3C=CC=CC3=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=C3C=CC=CC3=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 NQQZSKNYXUNFLL-HFJWLAOPSA-N 0.000 claims description 2
- JNQMSYHAWDHJPA-WTYVLRPYSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(=CC=2)C=2C=CC=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(=CC=2)C=2C=CC=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 JNQMSYHAWDHJPA-WTYVLRPYSA-N 0.000 claims description 2
- DZKBBMVMJPRBKE-SCLBCKFNSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C=2N3C=CSC3=NC=2Cl)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2N3C=CSC3=NC=2Cl)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 DZKBBMVMJPRBKE-SCLBCKFNSA-N 0.000 claims description 2
- GKTDYNGGZFLUNK-WTYVLRPYSA-N C([C@H]([C@@H]1C)NS(=O)(=O)CCC=2C3=CC=CC=C3C=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)CCC=2C3=CC=CC=C3C=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 GKTDYNGGZFLUNK-WTYVLRPYSA-N 0.000 claims description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 2
- 125000002393 azetidinyl group Chemical group 0.000 claims description 2
- 229910052799 carbon Inorganic materials 0.000 claims description 2
- 125000004432 carbon atom Chemical group C* 0.000 claims description 2
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 claims description 2
- 239000011737 fluorine Substances 0.000 claims description 2
- 229910052740 iodine Inorganic materials 0.000 claims description 2
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 claims description 2
- OLELREMYQQFANK-VBKZILBWSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-2,1,3-benzothiadiazole-4-sulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C3=NSN=C3C=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 OLELREMYQQFANK-VBKZILBWSA-N 0.000 claims description 2
- RITBMBQQADQHAZ-MEDUHNTESA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-2,2,2-trifluoroacetamide Chemical compound C[C@H]1[C@H](NC(=O)C(F)(F)F)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 RITBMBQQADQHAZ-MEDUHNTESA-N 0.000 claims description 2
- BGBDNSFFONZWAD-RCDICMHDSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-3,5-difluorobenzamide Chemical compound C([C@H]([C@@H]1C)NC(=O)C=2C=C(F)C=C(F)C=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 BGBDNSFFONZWAD-RCDICMHDSA-N 0.000 claims description 2
- HJXUSRGNAFOFFM-HMILPKGGSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-(4-bromophenoxy)benzenesulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(OC=3C=CC(Br)=CC=3)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 HJXUSRGNAFOFFM-HMILPKGGSA-N 0.000 claims description 2
- NGNVYVKNYJPDOF-AFJIDDCJSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-fluoro-n-[(4-fluorophenyl)methyl]benzenesulfonamide Chemical compound C([C@H]([C@@H]1C)N(CC=2C=CC(F)=CC=2)S(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 NGNVYVKNYJPDOF-AFJIDDCJSA-N 0.000 claims description 2
- OPNLETKMWVQXOA-QMHKHESXSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-fluoro-n-methylbenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)N(C)S(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 OPNLETKMWVQXOA-QMHKHESXSA-N 0.000 claims description 2
- HWECFSOUORLJIW-QMHKHESXSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-fluoro-n-methylsulfonylbenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)N(S(C)(=O)=O)S(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 HWECFSOUORLJIW-QMHKHESXSA-N 0.000 claims description 2
- WIGGIEDOQRQISD-AVRWGWEMSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-fluoro-n-propylbenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)N(CCC)S(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 WIGGIEDOQRQISD-AVRWGWEMSA-N 0.000 claims description 2
- SFXTXMOHUAZTJJ-QMHKHESXSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-methylsulfonylbenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(=CC=2)S(C)(=O)=O)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 SFXTXMOHUAZTJJ-QMHKHESXSA-N 0.000 claims description 2
- OWTBFARKWJWPTA-WTYVLRPYSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-4-phenoxybenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=CC(OC=3C=CC=CC=3)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 OWTBFARKWJWPTA-WTYVLRPYSA-N 0.000 claims description 2
- BUKQKDHNFJMMEN-SDDQJFBMSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-n-(7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl)methanesulfonamide Chemical compound C([C@H]([C@@H]1C)N(C23C(CC(CC2)C3(C)C)=O)S(C)(=O)=O)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 BUKQKDHNFJMMEN-SDDQJFBMSA-N 0.000 claims description 2
- SAQXOYXLZPGDGO-GAJHUEQPSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-n-phenylmethanesulfonamide Chemical compound C([C@H]([C@@H]1C)N(C=2C=CC=CC=2)S(C)(=O)=O)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 SAQXOYXLZPGDGO-GAJHUEQPSA-N 0.000 claims description 2
- YZVQUUKBYFDMQY-MGPUTAFESA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]butane-1-sulfonamide Chemical compound C[C@H]1[C@H](NS(=O)(=O)CCCC)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 YZVQUUKBYFDMQY-MGPUTAFESA-N 0.000 claims description 2
- FBCPNMOKIIQGED-KSFYIVLOSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]cyclohexanecarboxamide Chemical compound C([C@H]([C@@H]1C)NC(=O)C2CCCCC2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 FBCPNMOKIIQGED-KSFYIVLOSA-N 0.000 claims description 2
- BSXLLFUSNQCWJP-UHFFFAOYSA-N thiophene-2-sulfonic acid Chemical compound OS(=O)(=O)C1=CC=CS1 BSXLLFUSNQCWJP-UHFFFAOYSA-N 0.000 claims description 2
- 206010013654 Drug abuse Diseases 0.000 claims 4
- 206010057852 Nicotine dependence Diseases 0.000 claims 2
- 208000025569 Tobacco Use disease Diseases 0.000 claims 2
- 206010043903 Tobacco abuse Diseases 0.000 claims 2
- 206010001584 alcohol abuse Diseases 0.000 claims 2
- 208000025746 alcohol use disease Diseases 0.000 claims 2
- 201000009032 substance abuse Diseases 0.000 claims 2
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 claims 1
- PPZFWLBZGQWMEP-UHFFFAOYSA-N 1,3,5-trimethylpyrazole-4-sulfonic acid Chemical compound CC1=NN(C)C(C)=C1S(O)(=O)=O PPZFWLBZGQWMEP-UHFFFAOYSA-N 0.000 claims 1
- VHMPGAJRLKGKLQ-AVRWGWEMSA-N C([C@H]([C@@H]1C)NS(=O)(=O)C=2C3=CC=CC=C3C=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C3=CC=CC=C3C=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 VHMPGAJRLKGKLQ-AVRWGWEMSA-N 0.000 claims 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-N Caprylic acid Natural products CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 claims 1
- 150000001408 amides Chemical class 0.000 claims 1
- GONOPSZTUGRENK-UHFFFAOYSA-N benzyl(trichloro)silane Chemical compound Cl[Si](Cl)(Cl)CC1=CC=CC=C1 GONOPSZTUGRENK-UHFFFAOYSA-N 0.000 claims 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N n-hexanoic acid Natural products CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 claims 1
- 241001465754 Metazoa Species 0.000 abstract description 9
- 238000006243 chemical reaction Methods 0.000 description 53
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 33
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 30
- 239000000243 solution Substances 0.000 description 27
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 24
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 24
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 18
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 15
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 12
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 12
- 239000002904 solvent Substances 0.000 description 12
- 239000000126 substance Substances 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 230000000694 effects Effects 0.000 description 11
- 239000011541 reaction mixture Substances 0.000 description 11
- 238000005160 1H NMR spectroscopy Methods 0.000 description 10
- 208000009132 Catalepsy Diseases 0.000 description 10
- 206010047853 Waxy flexibility Diseases 0.000 description 10
- 239000002585 base Substances 0.000 description 10
- 238000007796 conventional method Methods 0.000 description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 9
- 229960004132 diethyl ether Drugs 0.000 description 9
- 206010039897 Sedation Diseases 0.000 description 8
- 238000002844 melting Methods 0.000 description 8
- 230000008018 melting Effects 0.000 description 8
- 230000036280 sedation Effects 0.000 description 8
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 7
- 241000699670 Mus sp. Species 0.000 description 7
- 230000036592 analgesia Effects 0.000 description 7
- 239000007788 liquid Substances 0.000 description 7
- 239000003960 organic solvent Substances 0.000 description 7
- RVJIUWJMJDLQIP-XJKSGUPXSA-N (2s,3r)-1-benzhydryl-2-methylazetidin-3-ol Chemical compound C[C@H]1[C@H](O)CN1C(C=1C=CC=CC=1)C1=CC=CC=C1 RVJIUWJMJDLQIP-XJKSGUPXSA-N 0.000 description 6
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 6
- 230000035484 reaction time Effects 0.000 description 6
- SPEUIVXLLWOEMJ-UHFFFAOYSA-N 1,1-dimethoxyethane Chemical compound COC(C)OC SPEUIVXLLWOEMJ-UHFFFAOYSA-N 0.000 description 5
- OILLHIQWPYRWOI-WMLDXEAASA-N [(2s,3r)-1-benzhydryl-2-methylazetidin-3-yl] methanesulfonate Chemical compound C[C@H]1[C@H](OS(C)(=O)=O)CN1C(C=1C=CC=CC=1)C1=CC=CC=C1 OILLHIQWPYRWOI-WMLDXEAASA-N 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 229940065144 cannabinoids Drugs 0.000 description 5
- 150000007529 inorganic bases Chemical class 0.000 description 5
- 239000010410 layer Substances 0.000 description 5
- 150000007530 organic bases Chemical class 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- CJCQBRZYAQTEOO-XJKSGUPXSA-N (2s,3r)-1-benzhydryl-2-methylazetidin-3-amine Chemical compound C[C@H]1[C@H](N)CN1C(C=1C=CC=CC=1)C1=CC=CC=C1 CJCQBRZYAQTEOO-XJKSGUPXSA-N 0.000 description 4
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical compound N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 description 4
- 206010003591 Ataxia Diseases 0.000 description 4
- 229940122820 Cannabinoid receptor antagonist Drugs 0.000 description 4
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 4
- 239000013543 active substance Substances 0.000 description 4
- 238000009835 boiling Methods 0.000 description 4
- 239000003536 cannabinoid receptor antagonist Substances 0.000 description 4
- ZPUCINDJVBIVPJ-LJISPDSOSA-N cocaine Chemical compound O([C@H]1C[C@@H]2CC[C@@H](N2C)[C@H]1C(=O)OC)C(=O)C1=CC=CC=C1 ZPUCINDJVBIVPJ-LJISPDSOSA-N 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 230000002631 hypothermal effect Effects 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 229910052717 sulfur Inorganic materials 0.000 description 4
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 4
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 4
- NMZREQPYUSVIIX-BZNIZROVSA-N (2r,3s)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-amine Chemical compound C[C@@H]1[C@@H](N)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 NMZREQPYUSVIIX-BZNIZROVSA-N 0.000 description 3
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 3
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 3
- 244000025254 Cannabis sativa Species 0.000 description 3
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- 229920001774 Perfluoroether Polymers 0.000 description 3
- 229910006074 SO2NH2 Inorganic materials 0.000 description 3
- 239000000556 agonist Substances 0.000 description 3
- 201000007930 alcohol dependence Diseases 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 125000003368 amide group Chemical group 0.000 description 3
- 238000012512 characterization method Methods 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 150000004292 cyclic ethers Chemical class 0.000 description 3
- 125000004122 cyclic group Chemical group 0.000 description 3
- CYQFCXCEBYINGO-IAGOWNOFSA-N delta1-THC Chemical compound C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@@H]21 CYQFCXCEBYINGO-IAGOWNOFSA-N 0.000 description 3
- 229940093499 ethyl acetate Drugs 0.000 description 3
- 235000019439 ethyl acetate Nutrition 0.000 description 3
- 238000007327 hydrogenolysis reaction Methods 0.000 description 3
- AQYSYJUIMQTRMV-UHFFFAOYSA-N hypofluorous acid Chemical compound FO AQYSYJUIMQTRMV-UHFFFAOYSA-N 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 238000001727 in vivo Methods 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- RITBMBQQADQHAZ-BZNIZROVSA-N n-[(2r,3s)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-2,2,2-trifluoroacetamide Chemical compound C[C@@H]1[C@@H](NC(=O)C(F)(F)F)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 RITBMBQQADQHAZ-BZNIZROVSA-N 0.000 description 3
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 3
- 239000003921 oil Substances 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 229910000027 potassium carbonate Inorganic materials 0.000 description 3
- NLKNQRATVPKPDG-UHFFFAOYSA-M potassium iodide Chemical compound [K+].[I-] NLKNQRATVPKPDG-UHFFFAOYSA-M 0.000 description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 229910052938 sodium sulfate Inorganic materials 0.000 description 3
- 235000011152 sodium sulphate Nutrition 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 238000010561 standard procedure Methods 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- VXNZUUAINFGPBY-UHFFFAOYSA-N 1-Butene Chemical group CCC=C VXNZUUAINFGPBY-UHFFFAOYSA-N 0.000 description 2
- LTVSBZFRNCOETR-UHFFFAOYSA-N 1-[bromo-(4-chlorophenyl)methyl]-4-chlorobenzene Chemical compound C1=CC(Cl)=CC=C1C(Br)C1=CC=C(Cl)C=C1 LTVSBZFRNCOETR-UHFFFAOYSA-N 0.000 description 2
- JVSFQJZRHXAUGT-UHFFFAOYSA-N 2,2-dimethylpropanoyl chloride Chemical compound CC(C)(C)C(Cl)=O JVSFQJZRHXAUGT-UHFFFAOYSA-N 0.000 description 2
- WCASXYBKJHWFMY-NSCUHMNNSA-N 2-Buten-1-ol Chemical compound C\C=C\CO WCASXYBKJHWFMY-NSCUHMNNSA-N 0.000 description 2
- 241000218236 Cannabis Species 0.000 description 2
- 235000012766 Cannabis sativa ssp. sativa var. sativa Nutrition 0.000 description 2
- 235000012765 Cannabis sativa ssp. sativa var. spontanea Nutrition 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N DMSO Substances CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 2
- 239000005977 Ethylene Substances 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- XADCESSVHJOZHK-UHFFFAOYSA-N Meperidine Chemical compound C=1C=CC=CC=1C1(C(=O)OCC)CCN(C)CC1 XADCESSVHJOZHK-UHFFFAOYSA-N 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- KFSLWBXXFJQRDL-UHFFFAOYSA-N Peracetic acid Chemical compound CC(=O)OO KFSLWBXXFJQRDL-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- CYQFCXCEBYINGO-UHFFFAOYSA-N THC Natural products C1=C(C)CCC2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3C21 CYQFCXCEBYINGO-UHFFFAOYSA-N 0.000 description 2
- HRQHJTJCRMLNPP-OYHNWAKOSA-N [(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl] 4-fluorobenzenesulfonate Chemical compound C([C@H]([C@@H]1C)OS(=O)(=O)C=2C=CC(F)=CC=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 HRQHJTJCRMLNPP-OYHNWAKOSA-N 0.000 description 2
- FXXACINHVKSMDR-UHFFFAOYSA-N acetyl bromide Chemical compound CC(Br)=O FXXACINHVKSMDR-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 230000001270 agonistic effect Effects 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 150000001340 alkali metals Chemical class 0.000 description 2
- 230000003042 antagnostic effect Effects 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 229940125717 barbiturate Drugs 0.000 description 2
- 229940049706 benzodiazepine Drugs 0.000 description 2
- 125000003310 benzodiazepinyl group Chemical class N1N=C(C=CC2=C1C=CC=C2)* 0.000 description 2
- 238000004166 bioassay Methods 0.000 description 2
- 229950005499 carbon tetrachloride Drugs 0.000 description 2
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 239000003638 chemical reducing agent Substances 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 229960003920 cocaine Drugs 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 239000013058 crude material Substances 0.000 description 2
- 238000002425 crystallisation Methods 0.000 description 2
- 230000008025 crystallization Effects 0.000 description 2
- 229960004242 dronabinol Drugs 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- WCASXYBKJHWFMY-UHFFFAOYSA-N gamma-methylallyl alcohol Natural products CC=CCO WCASXYBKJHWFMY-UHFFFAOYSA-N 0.000 description 2
- 239000000380 hallucinogen Substances 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 229940005483 opioid analgesics Drugs 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 239000007800 oxidant agent Substances 0.000 description 2
- JTJMJGYZQZDUJJ-UHFFFAOYSA-N phencyclidine Chemical compound C1CCCCN1C1(C=2C=CC=CC=2)CCCCC1 JTJMJGYZQZDUJJ-UHFFFAOYSA-N 0.000 description 2
- 229950010883 phencyclidine Drugs 0.000 description 2
- 125000003367 polycyclic group Chemical group 0.000 description 2
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 239000012047 saturated solution Substances 0.000 description 2
- 230000035807 sensation Effects 0.000 description 2
- 239000012279 sodium borohydride Substances 0.000 description 2
- 229910000033 sodium borohydride Inorganic materials 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- HJUGFYREWKUQJT-UHFFFAOYSA-N tetrabromomethane Chemical compound BrC(Br)(Br)Br HJUGFYREWKUQJT-UHFFFAOYSA-N 0.000 description 2
- VXUYXOFXAQZZMF-UHFFFAOYSA-N titanium(IV) isopropoxide Chemical compound CC(C)O[Ti](OC(C)C)(OC(C)C)OC(C)C VXUYXOFXAQZZMF-UHFFFAOYSA-N 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- OKZNPPMXPUCIDX-UHFFFAOYSA-N (2-methylazetidin-3-yl) 2,2-dimethylpropanoate;hydrochloride Chemical compound Cl.CC1NCC1OC(=O)C(C)(C)C OKZNPPMXPUCIDX-UHFFFAOYSA-N 0.000 description 1
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 description 1
- 125000003161 (C1-C6) alkylene group Chemical group 0.000 description 1
- 0 *C(*)N1C(*)C(*)(*)C1 Chemical compound *C(*)N1C(*)C(*)(*)C1 0.000 description 1
- QRABWFYVCUFAAB-UHFFFAOYSA-N 1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-ol Chemical compound CC1C(O)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 QRABWFYVCUFAAB-UHFFFAOYSA-N 0.000 description 1
- AZHWVHNIAGJINK-UHFFFAOYSA-N 1-benzhydrylazetidine Chemical class C1CCN1C(C=1C=CC=CC=1)C1=CC=CC=C1 AZHWVHNIAGJINK-UHFFFAOYSA-N 0.000 description 1
- AFFLGGQVNFXPEV-UHFFFAOYSA-N 1-decene Chemical group CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 1
- LOHMOYDNSULEAJ-DMTCNVIQSA-N 2,2,2-trifluoro-n-[(2r,3s)-2-methylazetidin-3-yl]acetamide Chemical compound C[C@H]1NC[C@@H]1NC(=O)C(F)(F)F LOHMOYDNSULEAJ-DMTCNVIQSA-N 0.000 description 1
- LOHMOYDNSULEAJ-IUYQGCFVSA-N 2,2,2-trifluoro-n-[(2s,3r)-2-methylazetidin-3-yl]acetamide Chemical compound C[C@@H]1NC[C@H]1NC(=O)C(F)(F)F LOHMOYDNSULEAJ-IUYQGCFVSA-N 0.000 description 1
- RZUXIYDMQGCCDD-UHFFFAOYSA-N 2,3-dibromobutan-1-ol Chemical compound CC(Br)C(Br)CO RZUXIYDMQGCCDD-UHFFFAOYSA-N 0.000 description 1
- CPZUINYGJOGDQX-UHFFFAOYSA-N 2-[(3-chlorophenyl)-imidazol-1-ylmethyl]adamantan-2-ol Chemical compound C1C2CC(C3)CC1CC3C2(O)C(N1C=NC=C1)C1=CC=CC(Cl)=C1 CPZUINYGJOGDQX-UHFFFAOYSA-N 0.000 description 1
- MRHUWANOQDXKAG-WMLDXEAASA-N 3-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-2,2-dimethylpropanoic acid Chemical compound C[C@H]1[C@H](CC(C)(C)C(O)=O)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 MRHUWANOQDXKAG-WMLDXEAASA-N 0.000 description 1
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 1
- PHUYGURFBULKPA-UHFFFAOYSA-N 4,4'-dichlorobenzhydrol Chemical compound C=1C=C(Cl)C=CC=1C(O)C1=CC=C(Cl)C=C1 PHUYGURFBULKPA-UHFFFAOYSA-N 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 102000009132 CB1 Cannabinoid Receptor Human genes 0.000 description 1
- 108010073366 CB1 Cannabinoid Receptor Proteins 0.000 description 1
- 102000009135 CB2 Cannabinoid Receptor Human genes 0.000 description 1
- 108010073376 CB2 Cannabinoid Receptor Proteins 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- OKZNPPMXPUCIDX-UOERWJHTSA-N Cl.C[C@@H]1NC[C@H]1OC(=O)C(C)(C)C Chemical compound Cl.C[C@@H]1NC[C@H]1OC(=O)C(C)(C)C OKZNPPMXPUCIDX-UOERWJHTSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 238000003747 Grignard reaction Methods 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical class Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- 206010021113 Hypothermia Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010022998 Irritability Diseases 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 1
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 1
- 238000011785 NMRI mouse Methods 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 229920003776 Reny® Polymers 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- HQVHOQAKMCMIIM-HXUWFJFHSA-N WIN 55212-2 Chemical compound C([C@@H]1COC=2C=CC=C3C(C(=O)C=4C5=CC=CC=C5C=CC=4)=C(N1C3=2)C)N1CCOCC1 HQVHOQAKMCMIIM-HXUWFJFHSA-N 0.000 description 1
- NRJVSTWYKYKNBK-QFBILLFUSA-N [(2s,3r)-1-benzhydryl-2-methylazetidin-3-yl] 2,2-dimethylpropanoate Chemical compound C[C@H]1[C@H](OC(=O)C(C)(C)C)CN1C(C=1C=CC=CC=1)C1=CC=CC=C1 NRJVSTWYKYKNBK-QFBILLFUSA-N 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 125000004450 alkenylene group Chemical group 0.000 description 1
- 150000004703 alkoxides Chemical class 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 230000002082 anti-convulsion Effects 0.000 description 1
- 230000001430 anti-depressive effect Effects 0.000 description 1
- 239000000935 antidepressant agent Substances 0.000 description 1
- 229940005513 antidepressants Drugs 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- GMWFCJXSQQHBPI-UHFFFAOYSA-N azetidin-3-ol Chemical class OC1CNC1 GMWFCJXSQQHBPI-UHFFFAOYSA-N 0.000 description 1
- 239000003637 basic solution Substances 0.000 description 1
- 230000003542 behavioural effect Effects 0.000 description 1
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 229940121376 cannabinoid receptor agonist Drugs 0.000 description 1
- 239000003537 cannabinoid receptor agonist Substances 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 150000001728 carbonyl compounds Chemical class 0.000 description 1
- 230000002903 catalepsic effect Effects 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000012069 chiral reagent Substances 0.000 description 1
- DERZBLKQOCDDDZ-JLHYYAGUSA-N cinnarizine Chemical compound C1CN(C(C=2C=CC=CC=2)C=2C=CC=CC=2)CCN1C\C=C\C1=CC=CC=C1 DERZBLKQOCDDDZ-JLHYYAGUSA-N 0.000 description 1
- 229960000876 cinnarizine Drugs 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001162 cycloheptenyl group Chemical group C1(=CCCCCC1)* 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000000522 cyclooctenyl group Chemical group C1(=CCCCCCC1)* 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 229940080861 demerol Drugs 0.000 description 1
- 125000004177 diethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- XEBCWEDRGPSHQH-UHFFFAOYSA-N diisopropyl tartrate Chemical compound CC(C)OC(=O)C(O)C(O)C(=O)OC(C)C XEBCWEDRGPSHQH-UHFFFAOYSA-N 0.000 description 1
- MGHPNCMVUAKAIE-UHFFFAOYSA-N diphenylmethanamine Chemical compound C=1C=CC=CC=1C(N)C1=CC=CC=C1 MGHPNCMVUAKAIE-UHFFFAOYSA-N 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000008157 edible vegetable oil Substances 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000006735 epoxidation reaction Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- SMANXXCATUTDDT-QPJJXVBHSA-N flunarizine Chemical compound C1=CC(F)=CC=C1C(C=1C=CC(F)=CC=1)N1CCN(C\C=C\C=2C=CC=CC=2)CC1 SMANXXCATUTDDT-QPJJXVBHSA-N 0.000 description 1
- 229960000326 flunarizine Drugs 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 230000002140 halogenating effect Effects 0.000 description 1
- 230000026030 halogenation Effects 0.000 description 1
- 238000005658 halogenation reaction Methods 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000011261 inert gas Substances 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 229940125425 inverse agonist Drugs 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000009191 jumping Effects 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- PGQDAWJAYJIGTH-RCDICMHDSA-N n-[(2s,3r)-1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]-3,5-dichlorobenzenesulfonamide Chemical compound C([C@H]([C@@H]1C)NS(=O)(=O)C=2C=C(Cl)C=C(Cl)C=2)N1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 PGQDAWJAYJIGTH-RCDICMHDSA-N 0.000 description 1
- SCKIGOBSACJIRK-XJKSGUPXSA-N n-[(2s,3r)-1-benzhydryl-2-methylazetidin-3-yl]-2,2,2-trifluoroacetamide Chemical compound C[C@H]1[C@H](NC(=O)C(F)(F)F)CN1C(C=1C=CC=CC=1)C1=CC=CC=C1 SCKIGOBSACJIRK-XJKSGUPXSA-N 0.000 description 1
- ZKNYVNQXGPDKFH-MELYUZJYSA-N n-[(2s,3r)-1-benzhydryl-2-methylazetidin-3-yl]-2,2,2-trifluoroacetamide;hydrochloride Chemical compound Cl.C[C@H]1[C@H](NC(=O)C(F)(F)F)CN1C(C=1C=CC=CC=1)C1=CC=CC=C1 ZKNYVNQXGPDKFH-MELYUZJYSA-N 0.000 description 1
- WBIYZICLJFECRW-UHFFFAOYSA-N n-[1-[bis(4-chlorophenyl)methyl]-2-methylazetidin-3-yl]hexanamide Chemical compound CC1C(NC(=O)CCCCC)CN1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 WBIYZICLJFECRW-UHFFFAOYSA-N 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 230000036403 neuro physiology Effects 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000002892 organic cations Chemical class 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- NXJCBFBQEVOTOW-UHFFFAOYSA-L palladium(2+);dihydroxide Chemical compound O[Pd]O NXJCBFBQEVOTOW-UHFFFAOYSA-L 0.000 description 1
- 230000035778 pathophysiological process Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 229960000482 pethidine Drugs 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 229940124531 pharmaceutical excipient Drugs 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- IPNPIHIZVLFAFP-UHFFFAOYSA-N phosphorus tribromide Chemical compound BrP(Br)Br IPNPIHIZVLFAFP-UHFFFAOYSA-N 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 239000002287 radioligand Substances 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 230000003893 regulation of appetite Effects 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000037380 skin damage Effects 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000012321 sodium triacetoxyborohydride Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 230000000707 stereoselective effect Effects 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- HFRXJVQOXRXOPP-UHFFFAOYSA-N thionyl bromide Chemical compound BrS(Br)=O HFRXJVQOXRXOPP-UHFFFAOYSA-N 0.000 description 1
- QAEDZJGFFMLHHQ-UHFFFAOYSA-N trifluoroacetic anhydride Chemical compound FC(F)(F)C(=O)OC(=O)C(F)(F)F QAEDZJGFFMLHHQ-UHFFFAOYSA-N 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/08—Drugs for disorders of the alimentary tract or the digestive system for nausea, cinetosis or vertigo; Antiemetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/12—Antidiarrhoeals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/14—Prodigestives, e.g. acids, enzymes, appetite stimulants, antidyspeptics, tonics, antiflatulents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/04—Antipruritics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/06—Antimigraine agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/20—Hypnotics; Sedatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
- A61P25/32—Alcohol-abuse
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D205/00—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom
- C07D205/02—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings
- C07D205/04—Heterocyclic compounds containing four-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
Definitions
- the present invention relates to substituted Azetidine compounds of general formula (I), methods for their preparation, medicaments comprising these compounds as well as their use for the preparation of a medicament for the treatment of humans and animals.
- Cannabinoids are compounds, which are derived from the cannabis sativa plant which is commonly known as marijuana.
- the most active chemical compound of the naturally occurring cannabinoids is tetrahydrocannabinol (THC), particularly ⁇ 9 -THC.
- cannabinoids as well as their synthetic analogues promote their physiological effects via binding to specific G-coupled receptors, the so-called cannabinoid-receptors.
- CB ⁇ and CB 2 are involved in a variety of physiological or pathophysiological processes in humans and animals, e.g. processes related to the central nervous system, immune system, cardiovascular system, endocrinous system, respiratory system, the gastrointestinal tract or to reproduction, as described for example, in Hollister, Pharm. Rev. 38, 1986, 1-20; Reny and Singha, Prog. Drug. Res., 36, 71-114, 1991; Consroe and Sandyk, in Marijuana/Cannabinoids, Neurobiology and Neurophysiology, 459, Murphy L and Barthe A. Eds., CRC Press, 1992.
- medicaments preferably in medicaments for the modulation of Cannabinoid receptors, particularly Cannabinoid 1 (CB-i) receptors.
- said medicaments should be suitable for the prophylaxis and/or treatment of disorders related to the central nervous system, the immune system, the cardiovascular system, the endocrinous system, the respiratory system, the gastrointestinal tract or reproduction in humans and/or animals.
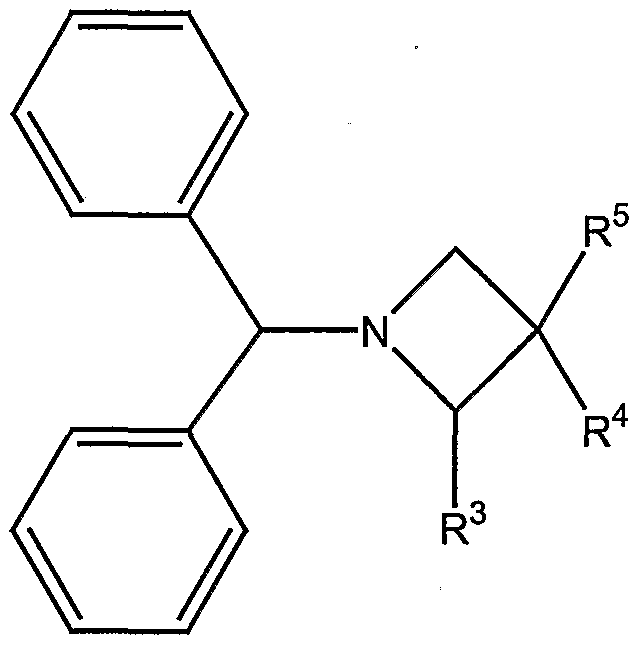
- the present invention relates to substituted Azetidine compounds of general formula I,
- R 1 represents an optionally at least mono-substituted phenyl group
- R 2 represents a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with an optionally at least mono-substituted mono- or polycyclic ring system, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with an optionally at least mono-substituted mono- or polycyclic ring system
- R 3 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or branched alkylene group, or an optionally at least mono-substituted
- R 4 represents a hydrogen atom, a cyano group, a carboxy group, a linear or branched alkyl group, or an optionally at least mono-substituted aryl group,
- R 5 represents an -O-SO 2 -R 6 -moiety, an -NH-CO-R 7 -moiety, an -Nh -moiety, an - NH-SO 2 -R 8 moiety, an -NR 9 -SO 2 -R 10 -moiety or an -O-CO-R 11 -moiety,
- R 6 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or branched alkylene group, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched alkylene group,
- R 7 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or branched alkylene group, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched alkylene group
- R 8 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may
- R 9 represents an -S0 2 -R 12 -moiety, a -CO-R 13 -moiety, a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring-system and/or which may be bonded via a linear or branched alkylene group and/or bridged by a linear or branched alkylene group, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via an alkylene group,
- R 10 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or bridged alkylene group, and/or which may be bridged by a linear or branched alkylene group, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched alkylene group
- R 11 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at
- R 12 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or bridged alkylene group, and/or which may be bridged by a linear or branched alkylene group, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched alkylene group,
- R 13 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or bridged alkylene group, and/or which may be bridged by a linear or branched alkylene group, or an optionally at least mono-substituted aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched alkylene group, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/
- a mono- or polycyclic ring-system means a mono- or polycyclic hydrocarbon ring-system that may be saturated, unsaturated or aromatic. If the ring system is polycyclic, each of its different rings may show a different degree of saturation, i.e. it may be saturated, unsaturated or aromatic. Optionally each of the rings of the mono- or polycyclic ring system may contain one or more, e.g. 1 , 2 or 3, heteroatoms as ring members, which may be identical or different and which can preferably be selected from the group consisting of N, O, S and P, more preferably be selected from the group consisting of N, O and S. Preferably the polycyclic ring-system may comprise two rings that are condensed.
- the rings of the mono- or polycyclic ring-sytem are preferably 5- or 6-membered.
- condensed means that a ring or ring- system is attached to another ring or ring-system, whereby the terms “annulated” or “annelated” are also used by those skilled in the art to designate this kind of attachment.
- each of the substituents may be independently selected from the group consisting of hydroxy, fluorine, chlorine, bromine, branched or unbranched C ⁇ .
- residues R 2 , R 3 and R 6 -R 13 represents or comprises a cycloaliphatic group, which contains one or more heteroatoms as ring members, unless defined otherwise, each of these heteroatoms may preferably be selected from the group consisting of N, O and S.
- a cycloaliphatic group may contain 1 , 2 or 3 heteroatoms independently selected from the group consisting of N, O and S.
- Suitable saturated or unsaturated, optionally at least heteroatom as ring member containing, optionally at least mono-substituted cycloaliphatic groups may preferably be selected from the group consisting of Cyclopropyl, Cyclobutyl, Cyclopentyl, Cyclohexyl, Cycloheptyl, Cyclooctyl, Cyclopentenyl, Cyclohexenyl, Cycloheptenyl, Cyclooctenyl, Pyrrolidinyl, Piperidinyl, Piperazinyl, Homo-Piperazinyl and Morpholinyl.
- each of the substituents may be independently selected from the group consisting of hydroxy, fluorine, chlorine, bromine, branched or unbranched C-i- 4 -alkoxy, branched or unbranched C ⁇ -alkyl, branched or unbranched C 1 - 4 - perfluoroalkoxy, branched or unbranched C ⁇ - 4 -perfluoroalkyl, amino, carboxy, oxo, amido, cyano, nitro, -SO 2 NH 2 , -CO-C- M -alkyl, -SO-C ⁇ -alkyl, -SO 2 -d ⁇ -alkyl, -NH- SO 2 -C ⁇ - 4 -alkyl , wherein the C
- each of the substituents may be independently selected from the group consisting of a halogen atom, a linear or branched C ⁇ _ 6 -alkyl group, a linear or branched C ⁇ . 6 alkoxy group, a formyl group, a hydroxy group, a trifluoromethyl group, a trifluoromethoxy group, a -CO-C-
- 6 -alkyl group a cyano group, a carboxy group, a - CO-O-C- ⁇ - 6 -alkyl group, a -CO-NR A R B - moiety, a -CO-NH-NR c R D -moiety, an -S-d-e- alkyl group, an -SO-d. 6 -alkyl group, an -S0 2 -C ⁇ . 6 -alkyl group, a -C ⁇ . 6 -alkylene-S-C ⁇ . 6 - alkyl group, a -C ⁇ . 6 -alkylene-SO-C ⁇ .
- R A , R B identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R A and R B together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different, C-i-e alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- R c , R D identical or different, represent a hydrogen atom, a C ⁇ - 6 -alkyl group, a -CO-O-C ⁇ - 6 -alkyl group, a C 3 - 8 -cycloalkyl group, a C ⁇ - 6 -alkylene-C 3 .
- R E , R F identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R E and R F together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different Ci- ⁇ alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member.
- Preferred aryl groups which may optionally be at least mono-substituted, are phenyl and naphthyl. If one or more of the residues R 2 , R 3 and R 6 -R 13 represents or comprises a heteroaryl group, which is substituted by one or more, e.g.
- each of the substituents may be independently selected from the group consisting of a halogen atom, a linear or branched Ci- 6 -alkyl group, a linear or branched C-i- 6 alcoxy group, a formyl group, a hydroxy group, a trifluoromethyl group, a trifluoromethoxy group, a -CO-C ⁇ . 6 -alkyl group, a cyano group, a carboxy group, a - CO-O-d.
- 6 -alkyl group a -CO-NR A R B - moiety, a -CO-NH-NR c R D -moiety, an -S-d_ 6 - alkyl group, an -SO-d-6-alkyl group, an -SO 2 -C ⁇ . ⁇ -alkyl group, a -C ⁇ . 6 -alkylene-S-C ⁇ . 6 - alkyl group, a -C ⁇ . 6 -alkylene-SO-C ⁇ . 6 -alkyl group, a -C ⁇ - 6 -alkylene-SO 2 -C ⁇ . 6 -alkyl group, a d- 6 -alkyl group substituted by one or more hydroxy groups and a -d- ⁇ - alkylene-NR E R F group,
- R A , R B identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R A and R B together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different, C ⁇ _ 6 alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- R c , R D identical or different, represent a hydrogen atom, a C ⁇ . 6 -alkyl group, a -CO-O-d- 6 -alkyl group, a C 3 . 8 -cycloalkyl group, a C ⁇ . 6 -alkylene-C 3 . 8 -cycloalkyl group, C ⁇ - 6 -alkylene-0-C ⁇ - 6 -alkyl group or a C ⁇ .
- 6 -alkyl group substituted with one or more hydroxy groups, or R c , R D together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more substituents independently selected from the group consisting of d- 6 alkyl group, a -CO-Ci- 6 -alkyl group, a -CO-O- Ci- ⁇ -alkyl group, a - CO-NH- C ⁇ _ 6 -alkyl group, a -CS-NH- C ⁇ . 6 -alkyl group, an oxo group, a C ⁇ .
- 6 -alkyl group substituted with one or more hydroxy groups, a C ⁇ . 6 -alkylene-O-C ⁇ . 6 -alkyl group and a -CO-NH 2 group and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member, and
- R E , R F identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R E and R F together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different C ⁇ _ 6 alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- heteroatoms which are present as ring members in the heteroaryl radical, may, unless defined otherwise, independently be selected from the group consisting of nitrogen, oxygen and sulphur.
- a heteroaryl radical may contain 1, 2 or 3 heteroatoms independently selected from the group consisting of nitrogen, oxygen and sulphur.
- Suitable heteroaryl groups may preferably be selected from the group consisting of thienyl, furyl, pyrrolyl, pyridinyl, imidazolyl, pyrimidinyl, pyrazinyl, indolyl, chinolinyl, isochinolinyl, benzo[1 ,2,5]- thiodiazolyl, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2,1-b]thiazolyl and pyrazolyl, more preferably be selected from the group consisting of thienyl-, benzo[1,2,5]-thiodiazolyl, benzo[b]thiophenyl, imidazo[2,1-b]thiazolyl and pyrazolyl.
- each of the substituents may be independently selected from the group consisting of hydroxy, fluorine, chlorine, bromine, branched or unbranched C ⁇ - 4 -alkoxy, branched or unbranched Ci- 4 -perfluoroalkoxy, branched or unbranched d- 4 -perfluoroalkyl, amino, carboxy, amido, cyano, nitro, -SO 2 NH 2 , -CO-d- 4 -alkyl, -SO-C ⁇ .
- d. 4 -alkyl may in each case be branched or unbranched, and a phenyl group, more preferably be selected from the group consisting of hydroxy, F, Cl, Br, methoxy, ethoxy, CF 3 and a phenyl group.
- Preferred linear or branched, saturated or unsaturated aliphatic groups which may be substituted by one or more substituents, may preferably be selected from the group consisting of methyl, ethyl, n-propyl, isopropyl, n-butyl, iso-butyl, sec-butyl, tert- butyl, n-pentyl, n-hexyl, n-heptyl, n-octyl, n-nonyl, n-decyl, vinyl, ethinyl, propenyl, propinyl, butenyl and butinyl.
- any of the residues R 3 and R 6 -R 13 comprises a linear or branched alkylene group
- said alkylene group may preferably be selected from the group consisting of methylene (-(CH 2 )-), ethylene (-(CH 2 ) 2 -), n-propylene (-(CH 2 ) -), isopropylene (-(C(CH )2-), n-butylene (-(CH 2 ) 4 -), n-pentylene (-(CH 2 ) 5 -), n-hexylene ( -(CH 2 ) 6 -), n-heptylene (-(CH 2 ) 7 -), n-octylene (-(CH 2 ) 8 -), n-nonylene( -(CH 2 ) 9 -) and n-decylene (-(CH 2 ) ⁇ 0 -), more preferably from the group consisting of (-(CH 2 )-), ethylene (-(
- R 1 represents a phenyl group, which is optionally substituted by one or more substituents independently selected from the group consisting of a halogen atom, a linear or branched Ci- 6 -alkyl group, a linear or branched C- ⁇ - 6 alkoxy group, a formyl group, a hydroxy group, a trifluoromethyl group, a trifluoromethoxy group, a -CO-C ⁇ .
- 6 -alkyl group a cyano group, a carboxy group, a -CO-O-C ⁇ - 6 -alkyl group, a -CO-NR A R B - moiety, a -CO-NH-NR c R D -moiety, an -S-C ⁇ . 6 -alkyl group, an -SO-C ⁇ . 6 -alkyl group, an -S ⁇ 2 -C ⁇ - 6 -alkyl group, a -C ⁇ _6-alkylene-S-C ⁇ -6-alkyl group, a -C ⁇ . 6 -alkylene-SO-C ⁇ - 6 - alkyl group, a -C ⁇ . 6 -alkylene-SO 2 -C ⁇ - 6 -alkyl group, a Ci- ⁇ -alkyl group substituted by one or more hydroxy groups and a -C ⁇ . 6 -alkylene-NR E R F group,
- R A , R B identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R A and R B together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different, C 1 - 6 alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- R c , R D identical or different, represent a hydrogen atom, a C ⁇ . 6 -alkyl group, a -CO-O-Ci- 6 -alkyl group, a C 3 . 8 -cycloalkyl group, a C ⁇ - 6 -alkylene-C 3 . 8 -cycloalkyl group, C-
- R E , R F identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R E and R F together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different C-i-e alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- R 1 represents a phenyl group, which is optionally substituted by one or more substituents independently selected from the group consisting of a fluorine atom, a chlorine atom, a bromine atom, a linear or branched C ⁇ _ 6 -alkyl group, a linear or branched C ⁇ _ 6 alkoxy group, a formyl group, a hydroxy group, a trifluoromethyl group, a trifluoromethoxy group, a cyano group and a carboxy group,
- R 1 represents a phenyl group, which is optionally substituted by one or more substituents independently selected from the group consisting of a fluorine atom, a chlorine atom, a bromine atom, a methyl group, a methoxy group, a trifluoromethyl group and a trifluoromethoxy group, most preferably R 1 represents a phenyl group, which is substituted by a chlorine atom in the 4-position,
- R 2 -R 13 have the meaning given above, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof.
- R 2 represents a saturated or unsaturated, optionally at least mono-substituted, optionally at least heteroatom as ring member containing C 3 . 8 cycloaliphatic group, which may be condensed with an optionally at least mono-substituted mono- or polycyclic ring system, or an optionally at least mono-substituted, 5- or 6-membered aryl or heteroaryl group, which may be condensed with an optionally at least mono- substituted mono- or polycyclic ring system,
- R 2 represents a phenyl group, which is optionally substituted by one or more substituents independently selected from the group consisting of a halogen atom, a linear or branched C ⁇ _ 6 -alkyl group, a linear or branched C ⁇ _ 6 alcoxy group, a formyl group, a hydroxy group, a trifluoromethyl group, a trifluoromethoxy group, a - CO-Ci- 6 -alkyl group, a cyano group, a carboxy group, a -CO-O-C ⁇ .6-alkyl group, a - CO-NR A R B - moiety, a -CO-NH-NR c R D -moiety, an -S-d.
- substituents independently selected from the group consisting of a halogen atom, a linear or branched C ⁇ _ 6 -alkyl group, a linear or branched C ⁇ _ 6 alcoxy group, a formy
- 6 -alkyl group an -SO-C 1 - 6 - alkyl group, an -SO 2 -C ⁇ . 6 -alkyl group, a -C ⁇ . 6 -alkylene-S-C ⁇ - 6 -alkyl group, a -d. 6 - alkylene-SO-C ⁇ - 6 -alkyl group, a -C ⁇ - 6 -alkylene-SO 2 -C ⁇ _ 6 -alkyl group, a C ⁇ . 6 -alkyl group substituted by one or more hydroxy groups and a -d. 6 -alkylene-NR E R F group,
- R A , R B identical or different, represent hydrogen or a C ⁇ - 6 -alkyl group, or R A and R B together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different, C ⁇ _ 6 alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- R c , R D identical or different, represent a hydrogen atom, a d- 6 -alkyl group, a -CO-O-Ci_ 6 -alkyl group, a C 3 . 8 -cycloalkyl group, a C ⁇ - 6 -alkylene-C 3 - 8 -cycloalkyl group, C ⁇ - 6 -alkylene-O-C ⁇ - 6 -alkyl group or a C ⁇ .
- 6 -alkyl group substituted with one or more hydroxy groups, or R c , R D together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more substituents independently selected from the group consisting of C 1 . 6 alkyl group, a -CO-C ⁇ . 6 -alkyl group, a -CO-O- C ⁇ _ 6 -alkyl group, a - CO-NH- Ci- 6 -alkyl group, a -CS-NH- C ⁇ profession 6 -alkyl group, an oxo group, a C ⁇ .
- R E , R F identical or different, represent hydrogen or a C ⁇ . 6 -alkyl group, or R ⁇ and R F together with the bridging nitrogen atom form a saturated, mono- or bicyclic, 3-10 membered heterocyclic ring system, which may be at least mono-substituted by one or more, identical or different C-j- ⁇ alkyl groups and/or which may contain at least one further heteroatom selected from the group consisting of nitrogen, oxygen and sulphur as a ring member,
- R 2 represents a phenyl group, which is optionally substituted by one or more substituents independently selected from the group consisting of a fluorine atom, a chlorine atom, a bromine atom, a methyl group, a methoxy group, a trifluoromethyl group and a trifluoromethoxy group,
- R 2 represents a phenyl group, which is substituted by a chlorine atom in the 4-position
- R 1 and R 3 -R 13 have the meaning given above, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof.
- R 3 represents a linear or branched, saturated or unsaturated, optionally at least mono- substituted C ⁇ _ ⁇ o-aliphatic group, a saturated or unsaturated, optionally at least mono- substituted, optionally at least one heteroatom as ring member containing C 3 .
- R 3 represents a linear or branched, optionally at least mono-substituted C ⁇ - 10 -alkyl group, or an optionally at least mono-substituted, 5- or 6- membered aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched C ⁇ _ 6 -alkylene group, more preferably R 3 represents a linear or branched, unsubstituted C ⁇ - ⁇ 0 -alkyl group, most preferably R 3 represents a methyl group, and R 1 , R 2 and R 4 -R 13 have the meaning given above, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof,
- R 4 represents a hydrogen atom, a cyano group, a carboxy group, a linear or branched C ⁇ - ⁇ 0 -alkyl group, or an optionally at least mono-substituted, 5- or 6- membered aryl group
- R 4 represents a hydrogen atom, a linear or branched Ci- 3 -alkyl group, or an optionally at least mono-substituted phenyl group, more preferably R 4 represents a hydrogen atom or a linear or branched C ⁇ _ 3 -alkyl group, most preferably R 4 represents a hydrogen atom
- R 1 -R 3 and R 5 -R 13 have the meaning given above, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers,
- R 5 represents an -O-SO 2 -R 6 -moiety, an -NH-CO-R 7 -moiety, an -NH 2 -moiety, an -NH- SO 2 -R 8 moiety or an -NR 9 -SO 2 -R 10 -moiety
- R 5 represents an -O-SO 2 -R 6 - moiety, an -NH-S0 2 -R 8 moiety or an -NR 9 -SO 2 -R 10 -moiety
- R 1 -R 4 and R 6 -R 13 have the meaning given above, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof,
- R 6 represents a linear or branched, saturated or unsaturated, optionally at least mono- substituted C 1 - 10 aliphatic group, a saturated or unsaturated, optionally at least mono- substituted, optionally at least one heteroatom as ring member containing C 3 . 8 - cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or branched C ⁇ .
- R 6 represents an optionally at least mono-substituted C 3 -s-cycloaliphatic group or an optionally at least mono-substituted phenyl group, wherein the respective substituents are independently selected from the group consisting of a fluorine atom, a chlorine atom, a bromine atom, a linear or branched C ⁇ _ 6 -alkyl group, a linear or branched C ⁇ - ⁇ alcoxy group, a formyl group, a hydroxy group, a trifluoromethyl group, a trifluoromethoxy group, a cyano group and a carboxy group, more preferably R 6 represents a pheny
- R 7 represents a linear or branched, saturated or unsaturated, optionally at least mono- substituted C ⁇ - ⁇ o-aliphatic group, a saturated or unsaturated, optionally at least mono- substituted, optionally at least one heteroatom as ring member containing C 3 . 8 - cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or branched C ⁇ .
- R 7 represents a linear or branched, optionally at least mono-substituted C ⁇ - 5 -alkyl group, a saturated, optionally at least mono-substituted C 5tician 6 -cycloaliphatic group, or an optionally at least mono-substituted phenyl group, wherein in each case the substituents are independently from one another selected from the group consisting of group consisting of a fluorine atom, a chlorine atom, a bromine atom, a methyl group, a methoxy group, a trifluoromethyl group and a trifluoromethoxy group, and R 1 -R 6 and R 8 -R 13 have the meaning given above, optionally in form of one of the stereoisomers, preferably enantiomers
- R 8 represents a linear or branched, saturated or unsaturated, optionally at least mono- substituted C ⁇ - ⁇ o-aliphatic group, a saturated or unsaturated, optionally at least mono- substituted, optionally at least one heteroatom as ring member containing C 3 .
- R 8 represents a linear or branched C ⁇ - 10 -alkyl group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing C 5 .
- R 8 represents a methyl group, an ethyl group, an n-propyl group, an n-butyl group, an optionally at least mono-substituted phenyl group, an optionally at least mono-substituted benzyl group, an optionally at least mono-substituted naphthyl group, which may be bonded via a
- R 9 represents an -SO 2 -R 2 -moiety, a -CO-R 13 -moiety, a linear or branched, saturated or unsaturated, optionally at least mono-substituted d-10 aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least heteroatom as ring member containing C3-8 cycloaliphatic group, which may be condensed with a mono- or polycyclic ring-system and/or which may be bonded by a linear or branched C 1 .
- R 9 represents an -SO 2 -R 12 -moiety, a linear or branched C 1 - 10 alkyl group, or an optionally at least mono-substituted phenyl group, which may be bonded via a C ⁇ _ 2 alkylene group, more preferably R 9 represents an - S0 2 -R 12 -moiety, a linear or branched d_ 3 alkyl group, or a phenyl group, which may be bonded via a C ⁇ _ 2 alkylene group and/or substituted with one or more substituents independently selected from the group consisting of a fluorine atom, a chlorine atom and a bromine atom, and R 1 -R 8 and R 10 -R 13 have the meaning given above optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably en
- R 10 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted C ⁇ . 10 -aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing C 3 _ 8 -cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or bridged Ci- 10 -alkylene group and/or which may be bridged by a linear or branched C ⁇ _ 5 -alkylene group, or an optionally at least mono-substituted 5- or 6-membered aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched Ci-10-alkylene group, preferably R 10 represents a linear or branched
- R 10 represents a methyl group, an ethyl group, an n-propyl group, an n-butyl group, an optionally at least mono-substituted phenyl group, an optionally at least mono-substituted benzyl group, an optionally at least mono-substituted naphthyl group, which may be bonded via
- R 11 represents a linear or branched, saturated or unsaturated, optionally at least mono- substituted C ⁇ - ⁇ o-aliphatic group, a saturated or unsaturated, optionally at least mono- substituted, optionally at least one heteroatom as ring member containing C 3 - 8 - cycloaliphatic group, which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or bridged C ⁇ - ⁇ o-alkylene group and/or which may be bridged by a linear or branched C ⁇ - 5 -alkylene group, or an optionally at least mono-substituted 5- or 6-membered aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched C ⁇ _ ⁇ o-alkylene group, preferably R 11 represents a -linear or-branched C
- ring member containing C 5 - 6 - cycloaliphatic group which may be condensed with a mono- or polycyclic ring- system and/or which may be bonded via a linear or bridged C ⁇ .
- R 11 represents a methyl group, an ethyl group, an n-propyl group, an n-butyl group, an optionally at least mono-substituted phenyl group, an optionally at least mono-substituted benzyl group, an optionally at least mono-substituted naphthyl group, which may be bonded via a C ⁇ - 3 -alkylene group, an optionally at least mono-substituted thienyl group, an optionally at least mono-substituted 2,1 ,3-Benzothiadiazole group, an optionally at least mono-substituted Benzo[b]thiophenyl group, an optionally at least mono- substituted lmidazo[2.1-b]thiazole group, an optionally at least mono-substituted 1 H- pyrazole group or a 7,7-Dimethyl-2-oxo-bicyclo-[2.2.1
- R 12 represents a linear or branched, saturated or unsaturated, optionally at least mono- substituted Ci- 10 -aliphatic group, a saturated or unsaturated, optionally at least mono- substituted, optionally at least one heteroatom as ring member containing C 3 .
- R 12 represents a linear or branched C ⁇ _ ⁇ o-alkyl group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing C 5 .
- R 12 represents a methyl group, an ethyl group, an n-propyl group, an n-butyl group, an optionally at least mono-substituted phenyl group, an optionally at least mono-substituted benzyl group, an optionally at least mono-substituted naphthyl group, which may be bonded via a d- 3 -alkylene group, an optionally at least mono-substituted thienyl group, an optionally at least mono-substituted 2,1 ,3-Benzothiadiazole group, an optionally at least mono-substituted Benzo[b]thiophenyl group, an optionally at least mono- substituted lmidazo[2.1-b]thiazole group, an optionally at least mono-substituted 1 H- pyrazole group or a 7,7-Dimethyl-2-oxo-bicyclo-[2.2.1
- R 13 represents a linear or branched, saturated or unsaturated, optionally at least mono-substituted C 1 - 10 - aliphatic group, a saturated or unsaturated, optionally at least mono-substituted, optionally at least one heteroatom as ring member containing C 3 - 8 -cycloaliphatic group, which may be condensed with a mono- or polycyclic ring-system and/or which may be bonded via a linear or bridged C ⁇ _ ⁇ o-alkylene group and/or which may be bridged by a linear or branched C ⁇ - 5 -alkylene group, or an optionally at least mono- substituted 5- or 6-membered aryl or heteroaryl group, which may be condensed with a mono- or polycyclic ring system and/or which may be bonded via a linear or branched C- M o-alkylene group, preferably R 13 represents a linear or branched C-MO
- R 13 represents a methyl group, an ethyl group, an n-propyl group, an n-butyl group, an optionally at least mono-substituted phenyl group, an optionally at least mono- substituted benzyl group, an optionally at least mono-substituted naphthyl group, which may be bonded via a C ⁇
- R 7 represents a linear or branched C 1 - 5 alkyl group, said alkyl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, -OH, -SH, linear or branched C 1 .
- -R 8 represents a linear or branched C 1 . 5 alkyl group; an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, linear or branched C 1 -6 alkyl. linear or branched C ⁇ .
- phenyl optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C ⁇ _ 6 alkyl and linear or branched C 1 - 6 - alcoxy
- phenoxy optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched d- 6 alkyl and linear or branched C ⁇ .
- R 10 represents a linear or branched C 1 - 5 alkyl group; an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, linear or branched C1- 6 alkyl, linear or branched d-6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C ⁇ _ 6 alkyl and linear or branched d.
- R 11 represents a linear or branched C 1 - 5 alkyl group; an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3,
- substituents independently selected from the group consisting of F, Cl, Br, linear or branched C 1 - 6 alkyl, linear or branched d- 6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C 1 - 6 alkyl and linear or branched C 1 - 6 - alcoxy), phenoxy (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C 1 . 6 alkyl and linear or branched d.
- R 12 represents a linear or branched C 1 - 5 alkyl group, an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, linear or branched C ⁇ - 6 alkyl, linear or branched C 1 - 6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C 1 - 6 alkyl and linear or branched C 1 - 6 - alcoxy), phenoxy
- R 13 represents a linear or branched C1. 5 alkyl group, an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3,
- substituents independently selected from the group consisting of F, Cl, Br, linear or branched C 1 .6 alkyl, linear or branched C1-6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C 1 - 6 alkyl and linear or branched d.
- stereoisomers optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof, especially to compounds of the following formula lb,
- X represents a halogen atom, preferably a chlorine atom
- alkyl group said alkyl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, -OH, -SH, linear or branched C- ⁇ - 6 alcoxy, -CF 3 and -OCF 3 ; a cyclopentyl or cyclohexyl group, said cyclopentyl or cyclohexyl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, -OH, -SH, linear or branched C 1 - 6 alcoxy, -CF 3 and -OCF 3 ; or a phenyl group, said phenyl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, linear or branched C ⁇ . 6 alkyl, linear or branched C
- R 8 represents a linear or branched C 1 -5 alkyl group; an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1, 2, 3,
- R 10 represents a linear or branched C 1 .5 alkyl group; an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, linear or branched C-i-e alkyl, linear or branched C 1 - 6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched d_ 6 alkyl and linear or branched d.
- R 11 represents a linear or branched C 1 - 5 alkyl group; an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3,
- substituents independently selected from the group consisting of F, Cl, Br, linear or branched C ⁇ - 6 alkyl, linear or branched C 1 - 6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched d_ 6 alkyl and linear or branched C 1 - 6 - alcoxy), phenoxy (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C1- 6 alkyl and linear or branched d.
- R 12 represents a linear or branched C1.
- an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3,
- substituents independently selected from the group consisting of F, Cl, Br, linear or branched C ⁇ . 6 alkyl, linear or branched C ⁇ . 6 alcoxy, phenyl (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched C ⁇ _ 6 alkyl and linear or branched C ⁇ - 6 - alcoxy), phenoxy (optionally substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, OH, linear or branched d- 6 alkyl and linear or branched d.
- R 13 represents a linear or branched C1. 5 alkyl group, an aryl or heteroaryl group selected from the group consisting of phenyl, naphthyl, thienyl, furanyl (furyl), 2,1 ,3- benzothiadiazole, benzo[b]thiophenyl, benzo[b]furanyl, imidazo[2.1-b]thiazole and pyrazole, said aryl or heteroaryl group being unsubstituted or substituted with 1 , 2, 3, 4 or 5 substituents independently selected from the group consisting of F, Cl, Br, linear or branched C ⁇ .
- stereoisomers optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof.
- R 1 represents a phenyl group, which is mono-substituted with a halogen atom, preferably a chlorine atom, in the 4-position of the phenyl ring,
- R 2 represents a phenyl group, which is mono-substituted with a halogen atom, preferably a chlorine atom, in the 4-position of the phenyl ring,
- R 3 represents a linear or branched, unsubstituted C ⁇ _ 6 alkyl group, preferably a methyl group,
- R 4 represents a hydrogen atom
- R 5 represents an -O-SO 2 -R 6 -moiety, an -NH-CO-R 7 -moiety, an -NH 2 -moiety, an NH-SO 2 -R 8 moiety, or an -NR 9 -SO 2 -R 10 -moiety
- R 6 represents a phenyl ring, which is optionally substituted with one or more halogen atoms, preferably one or more fluorine and/or one or more chlorine atoms,
- R 7 represents a linear or branched C 1 -5 alkyl group, a linear or branched C 1 - 5 alkyl group, which is at least partially fluorinated, a C 3 . 8 cycloalkyl group, or a phenyl group, which is optionally substituted with one or more halogen atoms, preferably one or more fluorine atoms,
- R 8 represents a linear or branched C 1 . 5 alkyl group
- a phenyl group which is optionally substituted with one or more substituents independently selected from the group consisting of a fluorine atom, a chlorine atom, an unsubstituted phenyl group, a formyl group, a methylsulfonyl group, a benzyl group and a phenoxy group, which is optionally mono-substituted by a bromine atom in its 4-position,
- a naphthyl group which may be bonded via a methylene or ethylene group
- Benzo[b]thiophene group which is optionally substituted with one or more methyl groups and/or one or more chlorine atoms,
- a pyrazole group which is optionally substituted with one or more substituents independently selected from the group consisting of a methyl group, an ethyl group and a phenyl group,
- thienyl group a furyl group, a.2,1,3-Benzothiadiazole group, a 7,7-Dimethyl-2-oxo- bicyclo-[2.2.1]-hept-1-yl group, or a benzyl group,
- R 9 represents a C 1 . 5 alkyl group, preferably a methyl group, a phenyl group, which is optionally substituted with one or more fluorine atoms and/or one or more chlorine atoms, a benzyl group, wherein the ring is optionally substituted with one or more fluorine atoms and/or one or more chlorine atoms, or a -S0 2 -R 12 -moiety,
- R 10 represents a phenyl group, which is optionally substituted with one or more fluorine atoms and/or one or more chlorine atoms,
- R 12 represents a C 1 . 5 alkyl group, preferably a methyl group, or a phenyl group, which is optionally substituted with one or more fluorine atoms and/or one or more chlorine atoms,
- stereoisomers optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof.
- substituted azetidine compounds of general formula I given above selected from the group consisting of
- the present invention provides a process for the preparation of substituted azetidine compounds of general formula I given above and corresponding stereoisomers thereof, according to which at least one compound of general formula
- R 1 to R 4 have the meaning given above, is reacted with at least one compound of general formula X 1 -SO 2 -R 6 or X 2 -CO-R 11 , wherein R 6 and R 11 have the meaning given above and X 1 and X 2 represent suitable leaving groups, in a suitable reaction medium, optionally in the presence of at least one base, to yield at least one compound of general formula I given above, wherein R 5 represents an -O-S0 2 -R 6 - moiety or an -0-CO-R 11 -moiety, and optionally purifying and/or optionally isolating said compound(s),
- R 5 represents an -O-SO 2 -R 6 or an -O-CO-R 11 moiety
- ammonia is reacted with ammonia, to yield a compound of general formula I, wherein R 5 represents an -NH 2 -moiety, and optionally purifying and/or optionally isolating said compound(s),
- R 5 represents an -NH 2 -moiety
- R 7 , R 8 and R 10 have the meaning given above and X 3 , X 4 and X 5 are suitable leaving groups, in a reaction medium, optionally in the presence of at least one base, to yield a compound of general formula I given above, wherein R 5 represents an -NH-CO-R 7 -moiety, an -NH-SO 2 -R 8 -moiety or an - NR 9 -S ⁇ 2 -R 10 -moiety with R 9 representing a hydrogen atom, and optionally purifying and/or optionally isolating said compound(s),
- R 5 represents an -NR 9 -SO 2 -R 10 -moiety with R 9 representing a hydrogen atom is reacted with at least one compound of general formula X 6 -R 9 , wherein R 9 has the meaning given above except for a hydrogen atom and X 6 is a leaving group, to yield at least one compound of general formula I given above, wherein R 5 represents an -NR 9 -SO 2 -R 10 -moiety, and optionally purifying and/or optionally isolating said compound(s),
- R 4 represents a hydrogen atom and R 1 -R 3 have the meaning given above, is oxidized to yield at least one compound of general formula IV,
- R 1 -R 3 have the meaning given above, which is optionally purified and/or optionally isolated, and reacted with at least one compound of general formula R 5a H, wherein R 5a represents an -NH 2 -moiety or an -NHR 9 -moiety, wherein R 9 has the meaning given above, the resulting compound is optionally purified and/or optionally isolated and optionally reacted with at least one compound of general formula X 3 -CO-R 7 , X 4 -SO 2 -R 8 or X 5 -SO 2 -R 10 , wherein R 7 , R 8 and R 10 have the meaning given above and X 3 , X 4 and X 5 represent a leaving group, in a reaction medium, optionally in the presence of at least one base, to yield a compound of general formula I given above, wherein R 5 represents an -NH 2 -moiety, an -NH-CO-R 7 -moiety, an -NH-SO2- R 8 -m
- step (a) according to scheme 1 is carried out in one or more organic solvents as reaction medium.
- Suitable solvents include but are not limited to halogenated, preferably chlorinated, organic solvents such as dichloromethane or chloroforme and linear or cyclic ethers, such as tetrahydrofurane, 1 ,4-dioxane, or 1,1- dimethoxyethane.
- Reaction temperature as well as the duration of the reaction may vary over a broad range. The optimum reaction temperature and duration of reaction for a given reaction may be determined by conventional methods known to those skilled in the art.
- Preferred reaction temperatures are 0-30 °C, preferably 15-25 °C. Suitable reaction times may vary from approximately 10 minutes to 3 days.
- Suitable bases for use in step (a) include organic bases such as triethylamine or pyridine as well as inorganic bases such as sodium hydroxide or potassium hydroxide. Mixtures of one or more organic bases and/or one or more inorganic bases may also be used.
- the leaving groups X 1 and X 2 may be of any type known to those skilled in the art for this type of reaction.
- the leaving group is a halogen atom, more preferably a chlorine or bromine atom.
- Suitable reaction media for the reaction with ammonia according to step (b) of scheme 1 include, for example, alcohols such as methanol, ethanol, isopropanol or mixtures of at least two of these alcohols.
- the ammonia is preferably added as a concentrated, preferably aqueous solution.
- Reaction temperature, pressure as well as the duration of the reaction may vary over a broad range.
- the optimum conditions may be determined by conventional methods known to those skilled in the art.
- Preferred reaction temperatures range from ambient temperature, i.e. approximately 15-25 °C to the boiling point of the reaction medium. Suitable reaction times may vary, for example from approximately 10 minutes to 3 days. Obviously the reaction may also be carried out in a reactor at elevated temperatures and pressure.
- Preferably compounds of general formula I, wherein R 5 represents a -0-S0 2 -R 6 -moiety with R 6 representing a methyl group, are used in step (b).
- the reaction steps (c) and (d) according to scheme 1 may also be carried out under conventional conditions known to those skilled in the art.
- a suitable reaction medium used for these reaction steps is preferably comprises one or more organic solvents.
- Suitable solvents include but are not limited to halogenated, preferably chlorinated, organic solvents such as dichloromethane or chloroforme and linear or cyclic ethers, such as tetrahydrofurane, 1,4-dioxane, or 1,1-dimethoxyethane.
- Reaction temperature as well as the duration of the reaction may vary over a broad range. The optimum conditions for a given reaction may be determined by conventional methods known to those skilled in the art. Preferred reaction temperatures are 0-30 °C, preferably 15-25 °C. Suitable reaction times vary, e.g. from approximately 10 minutes to 3 days.
- Suitable bases that may be present during reaction steps (c) and (d) include organic bases such as triethylamine or pyridine as well as inorganic bases such as sodium hydroxide or potassium hydroxide. Mixtures of one or more organic bases and/or one or more inorganic bases may also be used.
- the leaving groups X 3 , X 4 , X 5 and X 6 may be of any type known to those skilled in the art for this type of reaction.
- the leaving group is a halogen atom, more preferably a chlorine or bromine atom.
- step (e) of scheme 1 may preferably be carried out according to the literature publications of Katritzky et al., J. Heterocycl. Chem., 1994, 271-275; P.R. Dave et al., J. Org. Chem., 1996, 61(16), 5453, Synlett. 1991, (11), 783-784 and Axenrod et al., Tetrahedron Lett., 1993, 6677-6680.
- the respective descriptions are hereby incorporated by reference and form part of the present disclosure.
- step (f) according to scheme 1 is carried out in one or more organic solvents as reaction medium such as chlorinated, organic solvents like dichloromethane or chloroforme and preferably in the presence of sodium triacetoxyborohydride and acetic acid, or in the presence of H 2 and an alcohol such as methanol and/or ethanol as a reaction medium.
- organic solvents as reaction medium such as chlorinated, organic solvents like dichloromethane or chloroforme and preferably in the presence of sodium triacetoxyborohydride and acetic acid, or in the presence of H 2 and an alcohol such as methanol and/or ethanol as a reaction medium.
- Reaction temperature as well as the duration of the reaction may vary over a broad range.
- the optimum reaction temperature and duration of reaction for a given reaction may be determined by conventional methods known to those skilled in the art. Preferred reaction temperatures are 0-30 °C, preferably 15-25 °C.
- the substituents R 1 -R 4 have the same meaning as definied above and X represents a halogen atom, preferably a bromine or chlorine atom.
- X represents a halogen atom, preferably a bromine or chlorine atom.
- the substituted hydroxy-alkenylene compounds used as a starting material for reaction steps A and A1 according to scheme 2 are commercially available and/or may be prepared according to methods well known to those skilled in the art, e.g. by reduction of the corresponding carbonyl compound with a suitable reducing agent such as aluminium lithium hydride, sodium borohydride or catalysed hydrogenation as described, for example, in Catalysis Letters, 1999, 62(2-4), 175-177; Tetrahedron, 1984, 40(7), 1195-1198; J. Org.
- halogenenated alkenylene compounds used as starting material for reaction step B and B1 are commercially available and/or may be prepared from the corresponding hydroxy-alkenylene compounds using a suitable halogenation agent such as thionyl chloride, triphenyl phosphin/carbontetrachloride, bromine, hydrobromic acid, triphenyl phosphin/carbontetrabromide, phosphor tribromide and others described in the literature, for example, in Chem. Ber. 123(12), 2387-94, 1990; J. Am. Chem. Soc, 111(9), 3363-3368, 1989; J. Org.
- a suitable halogenation agent such as thionyl chloride, triphenyl phosphin/carbontetrachloride, bromine, hydrobromic acid, triphenyl phosphin/carbontetrabromide, phosphor tribromide and others described in the literature, for example, in Chem. Ber. 123
- reaction steps A, B and B1 may be carried out as described in Higgins et al., J.
- Reaction step A is preferably carried out in an organic solvent such as dichloromethane, chlorofom, carbontetrachloride or mixtures thereof as reaction medium.
- Reaction temperature as well as the duration of the reaction may vary over a broad range.
- the optimum conditions for a given reaction may be determined by conventional methods known to those skilled in the art.
- Preferred reaction temperatures are from -10°C to +30 °C, preferably from 0°C to 25 °C, more preferably from 0 °C to 10°C.
- Suitable reaction times vary from a few minutes to several hours, preferably from 3 to 8 hours.
- Reaction step B is preferably carried out by dissolving the educt in a suitable reaction medium, preferably a linear ether such as diethylether, or a cyclic ether, such as tetrahydrofuran, 1 ,4-dioxane or 1 ,1-dimethoxyethan, and in the presence of an aqueous solution of a base, preferably selected from the group consisting of an alkali metal such as lithium, sodium, or potassium, under vigorous stirring.
- a suitable reaction medium preferably a linear ether such as diethylether, or a cyclic ether, such as tetrahydrofuran, 1 ,4-dioxane or 1 ,1-dimethoxyethan
- a base preferably selected from the group consisting of an alkali metal such as lithium, sodium, or potassium, under vigorous stirring.
- Reaction temperature as well as the duration of the reaction may vary over a broad range.
- the optimum reaction conditions for a given reaction may be determined by conventional methods known to those skilled in the art.
- Preferred reaction temperatures range from 10°C to the boiling point of the reaction mixture, preferably from 15°C to 25 °C.
- Suitable reaction times vary from a few minutes to several hours, preferably from 3 to 8 hours
- Reaction step B1 is preferably carried out in the presence of an oxidizing agent such as peracetic acid, m-chloro perbenzoic acid, N-Bromsuccinimide or the like.
- an oxidizing agent such as peracetic acid, m-chloro perbenzoic acid, N-Bromsuccinimide or the like.
- the 3-azetidinol compound is obtained by carrying out reaction step (c) according to the methods described in the literature publications of M.E. Jung, J. Org. Chem., 1991 , 56(24), 6729-6730; V.R. Gaertner, J. Org. Chem., 1967, 32, 2972, Katrizky et al., J. Heterocycl. Chem. 1994, 271-275, P.R. Dave et al, J. Org. Chem., 1996, 61 (16), 5453 and US 5,073,646.
- the corresponding descriptions are incorporated by reference and form part of the present disclosure.
- reaction steps A2, B2, C2a and C2b may be carried out according to conventional methods well known to those skilled in the art.
- Step A2 is for example described in Hajipour et al., Synth. Commun. 1999, 29(10), 1697-1701 , A. Sasse et al., Arch. Pharm., 2001 , 334(2), 45-52.
- Step B2 is for example described in the publication of Fernandez et al., Synthesis 2001 , (2), 239-242; Baruah et al., Synlett, 1999, (4), 409-410.
- Steps C2a and C2b are for example disclosed in the publications of M. Grisar et al., J. Med.
- Another aspect the present invention is a process for the preparation of substituted azetidine compounds of general formula I given above and corresponding stereoisomers thereof, according to which at least one compound of general formula
- R and R 2 have the meaning given above and Y represents a halogen atom, preferably a chlorine atom or a bromine atom, is reacted with at least one compound of general formula VI,
- azetidine compound(s) optionally in form of a salt, wherein R 3 , R 4 and R 5 have the meaning given above, in a suitable reaction medium, optionally in the presence of a base, and the resulting azetidine compound(s) is/are optionally purified and/or optionally isolated.
- the compounds of general formula (Va) may, for example, be obtained according to step A3 of scheme 4 by reduction of a corresponding ketone compound with a suitable reducing agent such as sodium borohydride in a suitable reaction medium such as alcohol, preferably methanol, whereby the reaction temperature is preferably kept in the range of 0°C and the boling point of the reaction medium.
- a suitable reducing agent such as sodium borohydride
- a suitable reaction medium such as alcohol, preferably methanol
- the compounds of general formula (Va) may, for example, also be obtained according to step B3 of scheme 4 by a Grignard reaction in a suitable inert reaction medium such as diethylether or tetrahydrofuran, whereby the reation temperature is preferably kept in the range of 0 °C and the boiling point of the reaction medium.
- a suitable inert reaction medium such as diethylether or tetrahydrofuran
- Reaction step C3 of scheme 4, in which X represents a halogen atom, preferably a chlorine or bromine atom, may be carried out using a suitable halogenating agent such as hydrobromic acid, thionyl bromide, thionyl chloride, acetyl bromide, in a suitable reaction medium such as acetic acid, benzene, toluene, dichloromethane or chloroform, whereby the reaction temperature is preferably kept in the range of 0°C to the boling point of the reaction medium.
- a suitable halogenating agent such as hydrobromic acid, thionyl bromide, thionyl chloride, acetyl bromide
- a suitable reaction medium such as acetic acid, benzene, toluene, dichloromethane or chloroform
- the reaction of at least one compound of general formula V given above with at least one compound of general formula VI may preferably be carried out in an inert reaction medium, whereby acetonitrile, tetrahydrofuran or mixtures thereof are preferred.
- Suitable bases include organic bases such as triethylamine and inorganic bases such as carbonates of alkali metals, preferably potassium carbonate, or potassium iodide.
- the reaction temperature is preferably in the range of ambient temperature to the boling point of the reaction medium. The reaction times may vary over a broad range.
- the compounds of general formula VI may be prepared from the corresponding substituted benzhydrylazetidine compounds of general formula VII,
- compounds of general formula VI are preferably isolated in form of a corresponding salt such as a hydrochloride or hydrobromide salt.
- the compounds of general formula VII may be prepared according to the methods described, for example, in US 5,073,646 and J. Frigola, J. Med. Chem., 1993, 36, 801-810.
- the compounds of general formula VI are usually obtained in form of a mixture of diastereoisomers.
- the respective enantiomers may be obtained by racemic resolution with HPLC using chiral columns or by crystallization with chiral derivatives obtained from the reaction of the corresponding racemate with chiral agents as described in the publication of J. Frigola, J. Med. Chem. 1994, 37, 4195- 4210 and J. Frigola, J. Med. Chem., 1995, 38, 1203-1215.
- the corresponding enantiomers may also be obtained by asymmetric epoxidation in a corresponding reaction as described in scheme 2, which is carried out according to the method described in the publication of Sharpless et al., J. Am. Chem. Soc, 1980, 102, 5974- 5976; J. A. Marshall et al., Org. Lett., 2000 2(18), 2897-2900 using tert.-Butyl hydroperoxide in the presence of titanium isopropoxide and diethyl or diisopropyl tartrate as oxidant.
- substituted azetidine compounds of general formula (I) themselves are obtained in form of a mixture of stereoisomers, particularly enantiomers or diastereomers, said mixtures may be separated by standard procedures known to those skilled in the art, e.g. chromatographic methods or fractunalized crystallization with chiral reagents. It is also possible to obtain pure stereoisomers via stereoselective synthesis.
- Substituted azetidine compounds of general formula I which comprise nitrogen-atom containing saturated, unsaturated or aromatic rings may also be obtained in the form of their N-oxides by methods well known to those skilled in the art.
- the present invention also provides a process for the preparation of salts of substituted azetidine compounds of general formula (I), stereoisomers thereof or N-oxides thereof, wherein at least one compound of general formula (I) having at least one basic group is reacted with at least one inorganic and/or organic acid, preferably in the presence of a suitable reaction medium.
- Suitable reaction media include, for example, any of the ones given above.
- Suitable inorganic acids include hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid, nitric acid
- suitable organic acids are e.g. citric acid, maleic acid, fumaric acid, tartaric acid, or derivatives thereof, p-toluenesulfonic acid, methanesulfonic acid or camphersulfonic acid.
- the present invention also provides a process for the preparation of salts of substituted azetidine compounds of general formula (I), stereoisomers thereof or N-oxides thereof, wherein at least one compound of general formula (I) having at least one acidic group is reacted with one or more suitable bases, preferably in the presence of a suitable reaction medium.
- suitable bases are e.g. hydroxides, carbonates or alkoxides, which include suitable cations, derived e.g. from alkaline metals, alkaline earth metals or organic cations, e.g.
- Suitable reaction media are, for example, any of the ones given above.
- Solvates, preferably hydrates, of the substituted azetidine compounds of general formula (I), of corresponding stereoisomers, corresponding N-oxides thereof, or of corresponding salts thereof may also be obtained by standard procedures known to those skilled in the art.
- the purification and isolation of the inventive substituted azetidine compounds of general formula (I), of a corresponding stereoisomer, or salt, or solvate or any intermediate thereof may, if required, be carried out by conventional methods known to those skilled in the art, e.g. chromatographic methods or recrystallization.
- substituted azetidine compounds of general formula (I), their N-oxides, their stereoisomers, corresponding salts thereof and corresponding solvates are toxicologically acceptable and are therefore suitable as pharmaceutical active substances for the preparation of medicaments.
- an other aspect of the present invention is a medicament comprising at least one substituted azetidine compound of general formula I given above including the afore disclaimed compounds, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof, and optionally one or more pharmaceutically acceptable excipients.
- Said medicament may comprise any combination of one or more of the substituted azetidine compounds of general formula I given above, stereoisomers thereof, corresponding N-oxides thereof, physiologically acceptable salts thereof or physiologically acceptable solvates thereof.
- said medicament is suitable for the modulation (regulation) of cannabinoid-receptors, preferably cannabinoid 1 (CB-i) receptors, for the prophylaxis and/or treatment of disorders of the central nervous system, disorders of the immune system, disorders of the cardiovascular system, disorders of the endocrinous system, disorders of the respiratory system, disorders of the gastrointestinal tract or reproductive disorders.
- cannabinoid-receptors preferably cannabinoid 1 (CB-i) receptors
- said medicament is suitable for the prophylaxis and/or treatment of one or more disorders selected from the group consisting of psychosis, schizophrenia, anxiety, depression, epilepsy, neurodegenerative disorders, cerebellar disorders, spinocerebellar disorders, cognitive disorders, cranial trauma, panic attacks, peripheric neuropathy, inflammation, glaucoma, migraine, Morbus Parkinson, Morbus Huntington, Morbus Alzheimer, Raynaud's disease, tremblement disorders, compulsive disorders, senile dementia, thymic disorders, tardive dyskinesia, bipolar disorders, cancer, medicament-induced movement disorders, dystonia, endotoxemic shock, hemorragic shock, hypotension, insomnia, immunologic disorders, sclerotic plaques, vomiting, diarrhea, asthma, food intake disorders, preferably bulimia, anorexia, cachexia, obesity, type II diabetus mellitus (non-insuline dependent diabetes mellitus), memory disorders, pruritus, alcoholism, drug addiction, medicament
- the inventive medicament is suitable for the prophylaxis and/or treatment of pain, of food intake disorders, preferably bulimia, anorexia, cachexia, obesity or type II diabetus mellitus (non-insuline dependent diabetes mellitus), preferably diabetes, psychosis, alcoholism, drug addiction and/or medicament addiction, preferably drug addiction, diarrhea and/or pruritus.
- food intake disorders preferably bulimia, anorexia, cachexia, obesity or type II diabetus mellitus (non-insuline dependent diabetes mellitus), preferably diabetes, psychosis, alcoholism, drug addiction and/or medicament addiction, preferably drug addiction, diarrhea and/or pruritus.
- the inventive medicament is suitable for the prophylaxis and/or treatment of one or more disorders selected from the group consisting of obesity, psychosis and/or drug addiction.
- Another aspect of the present invention is the use of at least one substituted azetidine compound of general formula I including the afore disclaimed compounds, optionally in form of one of the stereoisomers, preferably enantiomers or diastereomers, a racemate or in form of a mixture of at least two of the stereoisomers, preferably enantiomers and/or diastereomers, in any mixing ratio, or a corresponding N-oxide thereof, a corresponding salt thereof, or a corresponding solvate thereof, and optionally one or more pharmaceutically acceptable excipients, for the preparation of a medicament for the modulation of cannabinoid-receptors, preferably cannabinoid 1 (CB ⁇ ) receptors, for the prophylaxis and/or treatment of disorders of the central nervous system, disorders of the immune system, disorders of
- the medicament according to the present invention may be in any form suitable for the application to humans and/or animals, preferably humans including infants, children and adults and can be produced by standard procedures known to those skilled in the art.
- the composition of the medicament may vary depending on the route of administration.
- the medicament of the present invention may for example be administered parentally in combination with conventional injectable liquid carriers, such as water or suitable alcohols.
- conventional pharmaceutical excipients for injection such as stabilizing agents, solubilizing agents, and buffers, may be included in such injectable compositions.
- These medicaments may for example be injected intramuscularly, intraperitoneally, or intravenously.
- Medicaments according to the present invention may also be formulated into orally administrable compositions containing one or more physiologically compatible carriers or excipients, in solid or liquid form. These compositions may contain conventional ingredients such as binding agents, fillers, lubricants, and acceptable wetting agents.
- the compositions may take any convenient form, such as tablets, pellets, capsules, lozenges, aqueous or oily solutions, suspensions, emulsions, or dry powdered forms suitable for reconstitution with water or other suitable liquid medium before use, for immediate or retarded release.
- liquid oral forms for administration may also contain certain additives such as sweeteners, flavoring, preservatives, and emulsifying agents.
- Non-aqueous liquid compositions for oral administration may also be formulated, containing edible oils. Such liquid compositions may be conveniently encapsulated in e.g., gelatin capsules in a unit dosage amount.
- the compositions of the present invention may also be administered topically or via a suppository.
- the daily dosage for humans and animals may vary depending on factors that have their basis in the respective species or other factors, such as age, sex, weight or degree of illness and so forth.
- the daily dosage for humans may preferably be in the range froml to 2000, preferably 1 to 1500, more preferably 1 to 1000 milligrams of active substance to be administered during one or several intakes per day.
- Substances with affinity for cannabinoid receptors are known to produce a wide range of pharmacological effects. It is also known that intravenous administration of a substance with affinity for cannabinoid receptors in mice produces analgesia , hypothermia, sedation and catalepsy. Individually, none of these effects can be considered as proof that a tested substance has affinity for cannabinoid-receptors, since all of these effects are common for various classes of centrally active agents. However, substances, which show all of these effects, i.e. substances that are active in this so-called tetrad model are considered to have affinity for the cannabinoid receptors. It has further been shown that cannabinoid receptor antagonists are higly effective in blocking the effects of a cannabinoid agonist in the mouse tetrad model.
- mice with a weight of 20-30 g Male NMRI mice with a weight of 20-30 g (Harlan, Barcelona, Spain) are used in all of the following experiments.
- mice are acclimatized to the experimental setting.
- Pre-Treatment control values are determined for analgesia hot plate latency (in seconds), rectal temperature, sedation and catalepsy.
- the mice are injected intravenously with the substance to be tested or the vehicle alone. 15 minutes after injection, latency in hot plate analgesia is measured. Rectal temperature, sedation and catalepsy are measured 20 minutes after injection.
- the hot plate analgesia is determined according to the method described in Woolfe D. et al. butThe evaluation of analgesic action of pethidine hydrochloride (Demerol)", J. Pharmacol. Exp. Ther. 80, 300-307,1944. The respective description is hereby incorporated by reference and forms part of the present disclosure.
- mice are placed on a hot plate (Harvard Analgesimeter) at 55 ⁇ 0.5 °C until they show a painful sensation by licking their paws or jumping and the time for these sensations to occur is recorded. This reading is considered the basal value (B).
- B basal value
- PC cut-off time
- mice Fifteen minuts after the administration of the substance to be tested, the mice are again placed on the hot plate and the afore described procedure is repeated. This period is called the post-treatment reading (PT).
- PT post-treatment reading
- the degree of analgesia is calculated from the formula :
- % MPE of Analgesia ( PT- B) / (PC-B) x 100
- Sedation and ataxia is determined according to the method described in Desmet L. K. C. et al. perennial Anticonvulsive properties of Cinarizine and Flunarizine in Rats and Mice", Arzneim. -Forsch. (Frug Res) 25, 9, 1975.
- the respective description is hereby incorporated by reference and forms part of the present disclosure.
- the chosen scoring system is
- the percentage of sedation is determined according to the formula:
- % of sedation arithmetic mean / 3 X 100
- the base-line rectal temperatures are determined with a thermometer (Yello Springs Instruments Co., Panlabs) and a thermistor probe inserted to 25mm before the administration of the substance to be tested. Rectal temperature is again measured 20 minutes after the administration of the substances to be tested. The temperature difference is calculated for each animal, whereby differences of >-2 °C are considered to represent activity.
- Catalepsy is determined according to the method described in Alpermann H. G. et al. -Pharmacological effets of Hoe 249: A new potential antidepressant", Drugs Dev. Res. 25, 267-282. 1992. The respective description is hereby incorporated by reference and forms part of the present disclosure.
- the cataleptic effect of the substance to be tested is evaluated according to the duration of catalepsy, whereby the animals are placed head downwards with their kinlegs upon the top of the wooden block.
- the chosen scoring system is:
- the percentage of catalepsy is determined according ot the following formula:
- % Catalepsy arithmetic mean / 6 X 100
- the etheric layer of the first extraction was acidified with dilute 5 % (volume/volume) acetic acid, the acidic layer alkalinized with sodium hydroxide and extracted wilh dichloromethane, to yield 6.3 g of the compound giving a total of 16.3 g (70% of theory) of trans-3-amino-1-diphenylmethyl-2-methylazetidine having a melting point of 68-69° C.
- the enantiomer (2S, 3R)-1-diphenylmethyl-2-methyl-3-methylsulphonyloxyazetidine was obtained by the same procedure as described above for the preparation of the trans-racemate but using (2S, 3R)-1-diphenylmethyl-3-hydroxy-2-methylazetidine.
- the compound (2S, 3R)-1-diphenylmethyl-3-hydroxy-2-methylazetidine was obtained via the optical resolution of trans-1 -diphenylmethyl-3-hydroxy-2-methylazetidine with (+)-(1S)-camphersulfonic acid, as described in the literature publication of J. Frigola et al., J. Med. Chem., 1995, 38, 1203-1215.
- the crude material is dissolved in 15 ml of ethanol, and an ethanolic solution saturated with hydrochloride gas is added to precipitate the corresponding hydrochloride salt, which is filtered off and washed with diethylether to yield 3.95 g (yield 85% of theory) of the hydrochloride salt having a melting point of 163 - 166 °C.
- This compound was obtained via the hydrolysis of the ester prepared according to step (1c) given above, which was dissolved in ethanol comprising 10 % by weight of sodium hydroxide. The reaction mixture was kept at room temperature (approximately 25 °C) overnight. 1-[Bis-(4-chloro-phenyi)-methyl]-2-methyl-azetidin- 3-ol was obtained in a yield of 89%.
- inventive azetidine compounds act as cannabinoid receptor antagonists, particularly for the CB-i-receptors.
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| RU2006133258/04A RU2006133258A (en) | 2004-02-17 | 2005-02-16 | SUBSTITUTED AZETIDINE DERIVATIVES, THEIR PRODUCTION AND APPLICATION AS MEDICINES |
| AU2005212835A AU2005212835A1 (en) | 2004-02-17 | 2005-02-16 | Substituted Azetidine compounds, their preparation and use as medicaments |
| BRPI0507801-6A BRPI0507801A (en) | 2004-02-17 | 2005-02-16 | substituted azetidine compounds, process for preparing substituted azetidine compounds, medicament and use of at least one substituted azetidine compound |
| CA002556565A CA2556565A1 (en) | 2004-02-17 | 2005-02-16 | Substituted azetidine compounds, their preparation and use as medicaments |
| JP2006553539A JP2007522255A (en) | 2004-02-17 | 2005-02-16 | Substituted azetidine compounds, their manufacture and use as pharmaceuticals |
| EP05715383A EP1718609A1 (en) | 2004-02-17 | 2005-02-16 | Substituted azetidine compounds, their preparation and use as medicaments |
| IL177456A IL177456A0 (en) | 2004-02-17 | 2006-08-10 | Substituted azetidine compounds, their preparation and use as medicaments |
| US11/505,463 US20070066587A1 (en) | 2004-02-17 | 2006-08-17 | Substituted azetidine compounds, their preparation and use as medicaments |
| NO20064078A NO20064078L (en) | 2004-02-17 | 2006-09-11 | Substituted azetidine compositions, their preparation and use as drugs |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| ES200400379 | 2004-02-17 | ||
| ES200400379A ES2244314B1 (en) | 2004-02-17 | 2004-02-17 | SUBSTITUTED AZETIDINIC COMPOUNDS, THEIR PREPARATION AND THEIR APPLICATION AS MEDICATIONS. |
| US10/804,558 | 2004-03-19 | ||
| US10/804,558 US20050187208A1 (en) | 2004-02-17 | 2004-03-19 | Substituted Azetidine compounds, their preparation and use as medicaments |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/505,463 Continuation-In-Part US20070066587A1 (en) | 2004-02-17 | 2006-08-17 | Substituted azetidine compounds, their preparation and use as medicaments |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005077897A1 true WO2005077897A1 (en) | 2005-08-25 |
Family
ID=34863169
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2005/001658 WO2005077897A1 (en) | 2004-02-17 | 2005-02-16 | Substituted azetidine compounds, their preparation and use as medicaments |
Country Status (9)
| Country | Link |
|---|---|
| EP (1) | EP1718609A1 (en) |
| JP (1) | JP2007522255A (en) |
| KR (1) | KR20060124756A (en) |
| AU (1) | AU2005212835A1 (en) |
| BR (1) | BRPI0507801A (en) |
| CA (1) | CA2556565A1 (en) |
| NO (1) | NO20064078L (en) |
| RU (1) | RU2006133258A (en) |
| WO (1) | WO2005077897A1 (en) |
Cited By (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007169270A (en) * | 2005-11-25 | 2007-07-05 | Tanabe Seiyaku Co Ltd | Medicinal composition |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| US7485732B2 (en) | 2003-06-11 | 2009-02-03 | Merck & Co., Inc. | Substituted 3-alkyl and 3-alkenyl azetidine derivatives |
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| FR2928149A1 (en) * | 2008-02-29 | 2009-09-04 | Sanofi Aventis Sa | AZETIDINE-DERIVED COMPOUNDS, THEIR PREPARATION AND THEIR THERAPEUTIC USE |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| FR2946650A1 (en) * | 2009-06-16 | 2010-12-17 | Sanofi Aventis | ESTERS DERIVED FROM AZETIDINES, THEIR PREPARATION AND THEIR USE IN THERAPEUTICS. |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| US7906652B2 (en) | 2005-11-28 | 2011-03-15 | Merck Sharp & Dohme Corp. | Heterocycle-substituted 3-alkyl azetidine derivatives |
| WO2011157827A1 (en) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-one derivatives as inhibitors of lipases and phospholipases |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2013001505A3 (en) * | 2011-06-29 | 2013-03-07 | Adamed Sp. Z O.O. | Sulphonamide derivatives of alicyclic amines for the treatment of central nervous system diseases |
| EP2754653A2 (en) * | 2011-09-05 | 2014-07-16 | Korea Institute of Science and Technology | Azetidine derivative and antidepressant composition containing same |
| US9440948B2 (en) | 2010-09-03 | 2016-09-13 | University Of Florida Research Foundation, Inc. | Nicotine compounds and analogs thereof, synthetic methods of making compounds, and methods of use |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0406112A1 (en) * | 1989-06-29 | 1991-01-02 | Laboratorios Del Dr. Esteve, S.A. | 1-Benzhydrylazetidines, their preparation and their use as intermediates for the preparation of compounds with antimicrobial activity |
| WO2001064633A1 (en) * | 2000-03-03 | 2001-09-07 | Aventis Pharma S.A. | Pharmaceutical compositions containing 3-amino-azetidine derivatives, novel derivatives and preparation thereof |
| WO2003020314A1 (en) * | 2001-08-29 | 2003-03-13 | Aventis Pharma S.A. | Compositions for the treatment of parkinson's disease containing a cb1 receptor antagonist and a product that activates dopaminergic neurotransmission in the brain |
| WO2003053431A2 (en) | 2001-12-21 | 2003-07-03 | Aventis Pharma S.A. | Pharmaceutical compositions based on azetidine derivatives |
-
2005
- 2005-02-16 KR KR1020067018702A patent/KR20060124756A/en not_active Application Discontinuation
- 2005-02-16 BR BRPI0507801-6A patent/BRPI0507801A/en not_active Application Discontinuation
- 2005-02-16 EP EP05715383A patent/EP1718609A1/en not_active Withdrawn
- 2005-02-16 WO PCT/EP2005/001658 patent/WO2005077897A1/en active Application Filing
- 2005-02-16 JP JP2006553539A patent/JP2007522255A/en not_active Withdrawn
- 2005-02-16 AU AU2005212835A patent/AU2005212835A1/en not_active Abandoned
- 2005-02-16 CA CA002556565A patent/CA2556565A1/en not_active Abandoned
- 2005-02-16 RU RU2006133258/04A patent/RU2006133258A/en unknown
-
2006
- 2006-09-11 NO NO20064078A patent/NO20064078L/en not_active Application Discontinuation
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0406112A1 (en) * | 1989-06-29 | 1991-01-02 | Laboratorios Del Dr. Esteve, S.A. | 1-Benzhydrylazetidines, their preparation and their use as intermediates for the preparation of compounds with antimicrobial activity |
| US5073646A (en) | 1989-06-29 | 1991-12-17 | Laboratorios Del Dr. Esteve, S.A. | Substituted 1-diphenylmethyl azetidines |
| WO2001064633A1 (en) * | 2000-03-03 | 2001-09-07 | Aventis Pharma S.A. | Pharmaceutical compositions containing 3-amino-azetidine derivatives, novel derivatives and preparation thereof |
| WO2003020314A1 (en) * | 2001-08-29 | 2003-03-13 | Aventis Pharma S.A. | Compositions for the treatment of parkinson's disease containing a cb1 receptor antagonist and a product that activates dopaminergic neurotransmission in the brain |
| WO2003053431A2 (en) | 2001-12-21 | 2003-07-03 | Aventis Pharma S.A. | Pharmaceutical compositions based on azetidine derivatives |
Non-Patent Citations (47)
| Title |
|---|
| A. C. HOWLETT ET AL.: "International Union of Pharmacology XXVII. Classification of Cannabinoid Receptors", PHARMACOL REV, vol. 54, 2002, pages 161 - 202 |
| A. SASSE ET AL., ARCH. PHARM., vol. 334, no. 2, 2001, pages 45 - 52 |
| ALPERMANN H. G. ET AL.: "Pharmacological effets of Hoe 249: A new potential antidepressant", DRUGS DEV. RES., vol. 25, 1992, pages 267 - 282 |
| AXENROD ET AL., TETRAHEDRON LETT., 1993, pages 6677 - 6680 |
| BARUAH ET AL., SYNLETT, no. 4, 1999, pages 409 - 410 |
| BULL. SOC. CHIM. FRANCE, vol. 132, no. 5-6, 1995, pages 522 - 30 |
| CATALYSIS LETTERS, vol. 62, no. 2-4, 1999, pages 175 - 177 |
| CHEM. BER., vol. 123, no. 12, 1990, pages 2387 - 94 |
| CONSROE; SANDYK: "Neurobiology and Neurophysiology", vol. 459, 1992, CRC PRESS, article "Marijuana/Cannabinoids" |
| DAVID R. COMPTON ET AL.: "In-vivo Characterization of a Specific Cannabinoid Receptor Antagonist (SR141716A) :Inhibition of Tetrahvdrocannbinol- induced Responses and Apparent Aaonist Activity", J. PHARMACOL. EXP. THER., vol. 277, no. 2, 1996, pages 586 - 594 |
| DAVID R. COMPTON ET AL.: "In-vivo Characterization of a Specific Cannabinoid Receptor Antagonist (SR141716A) Inhibition of Tetrahydrocannbinol- induced Responses and Apparent Agonist Activity", J. PHARMACOL EXP THER., vol. 277, no. 2, 1996, pages 586 - 594 |
| DEJAEGHER ET AL., SYNLETT., 2002, pages 113 - 115 |
| DESMET L. K. C. ET AL.: "Anticonvulsive properties of Cinarizine and Flunarizine in Rats and Mice", ARZNEIM. -FORSCH. (FRUG RES), vol. 25, 1975, pages 9 |
| FARMACO, vol. 44, no. 12, 1989, pages 1167 - 91 |
| FERNANDEZ ET AL., SYNTHESIS, no. 2, 2001, pages 239 - 242 |
| FRIGOLA J ET AL: "7-Azetidinylquinolones as Antibacterial Agents. 2. Synthesis and Biological Activity of 7-(2,3-Disubstituted-1-azetidinyl)-4-oxoquino line- and -1,8-naphthyridine-3-carboxylic Acids. Properties and Structure-Activity Relationships of Quinolones with an Azetidine Moiety", JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 37, no. 24, 1994, pages 4195 - 4210, XP002115349, ISSN: 0022-2623 * |
| FRIGOLA J ET AL: "7-Azetidinylquinolones as Antibacterial Agents. 3. Synthesis, Properties and Structure-Activity Relationships of the Stereoisomers Containing a 7-(3-Amino-2-methyl-1-azetidinyl) Moiety", JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 38, no. 7, 1995, pages 1203 - 1215, XP002115345, ISSN: 0022-2623 * |
| HAJIPOUR ET AL., SYNTH. COMMUN., vol. 29, no. 10, 1999, pages 1697 - 1701 |
| HETEROCYCLES, vol. 32, no. 5, 1991, pages 965 - 73 |
| HIGGINS ET AL., J. HETEROCYCLIC CHEM., vol. 8, 1971, pages 1059 - 1062 |
| HOLLISTER, PHARM. REV., vol. 38, 1986, pages 1 - 20 |
| J. A. MARSHALL ET AL., ORG. LETT., vol. 2, no. 18, 2000, pages 2897 - 2900 |
| J. AM. CHEM. SOC., 2001, pages 12168 - 12175 |
| J. AM. CHEM. SOC., vol. 111, no. 9, 1989, pages 3363 - 3368 |
| J. FRIGOLA ET AL., J. MED. CHEM., vol. 38, 1995, pages 1203 - 1215 |
| J. FRIGOLA, J. MED. CHEM., vol. 36, 1993, pages 801 - 810 |
| J. FRIGOLA, J. MED. CHEM., vol. 37, 1994, pages 4195 - 4210 |
| J. FRIGOLA, J. MED. CHEM., vol. 38, 1995, pages 1203 - 1215 |
| J. MED. CHEM., vol. 33, no. 3, 1990, pages 908 - 18 |
| J. NATURAL PRODUCTS, vol. 65, no. 6, 2002, pages 902 - 908 |
| J. ORG. CHEM., vol. 63, no. 25, 1998, pages 9565 - 68 |
| J. ORG. CHEM., vol. 64, no. 7, 1999, pages 2582 - 2589 |
| KATRITZKY ET AL., J. HETEROCYCL. CHEM., 1994, pages 271 - 275 |
| KATRIZKY ET AL., J. HETEROCYCL. CHEM., 1994, pages 271 - 275 |
| M. GRISAR ET AL., J. MED. CHEM., 1973, pages 885 |
| M.E. JUNG, J. ORG. CHEM., vol. 56, no. 24, 1991, pages 6729 - 6730 |
| P.R. DAVE ET AL., J. ORG. CHEM., vol. 61, no. 16, 1996, pages 5453 |
| RENY; SINGHA, PROG. DRUG. RES., vol. 36, 1991, pages 71 - 114 |
| RUTH A. ROSS; HEATHER C. BROCKIE ET AL.: "Agonist-inverse agonist characterization at CB and CB2 cannabinoid receptors of L-759633, L759656 and AM630", BRITISH JOURNAL OF PHARMACOLOGY, vol. 126, 1999, pages 665 - 672 |
| SHARPLESS ET AL., J. AM. CHEM. SOC., vol. 102, 1980, pages 5974 - 5976 |
| SYNLETT., 1991, pages 783 - 784 |
| SYNLETT., 1999, pages 1763 - 65 |
| SYNTHESIS, vol. 8, 1989, pages 598 - 603 |
| TETRAHEDRON LETT., vol. 43, no. 49, 2002, pages 8893 - 8896 |
| TETRAHEDRON, vol. 40, no. 7, 1984, pages 1195 - 1198 |
| V.R. GAERTNER, J. ORG. CHEM., vol. 32, 1967, pages 2972 |
| WOOLFE D. ET AL.: "The evaluation of analgesic action of pethidine hydrochloride (Demerol)", J. PHARMACOL. EXP. THER., vol. 80, 1944, pages 300 - 307 |
Cited By (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7485732B2 (en) | 2003-06-11 | 2009-02-03 | Merck & Co., Inc. | Substituted 3-alkyl and 3-alkenyl azetidine derivatives |
| US7906503B2 (en) | 2003-06-11 | 2011-03-15 | Merck Sharp & Dohme Corp. | Substituted 3-alkyl and 3-alkenyl azetidine derivatives |
| JP2007169270A (en) * | 2005-11-25 | 2007-07-05 | Tanabe Seiyaku Co Ltd | Medicinal composition |
| US7906652B2 (en) | 2005-11-28 | 2011-03-15 | Merck Sharp & Dohme Corp. | Heterocycle-substituted 3-alkyl azetidine derivatives |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2009118473A3 (en) * | 2008-02-29 | 2009-11-19 | Sanofi-Aventis | Azetidine-derived compounds, preparation method therefor and therapeutic use of same |
| WO2009118473A2 (en) | 2008-02-29 | 2009-10-01 | Sanofi-Aventis | Azetidine-derived compounds, preparation method therefor and therapeutic use of same |
| FR2928149A1 (en) * | 2008-02-29 | 2009-09-04 | Sanofi Aventis Sa | AZETIDINE-DERIVED COMPOUNDS, THEIR PREPARATION AND THEIR THERAPEUTIC USE |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| FR2946650A1 (en) * | 2009-06-16 | 2010-12-17 | Sanofi Aventis | ESTERS DERIVED FROM AZETIDINES, THEIR PREPARATION AND THEIR USE IN THERAPEUTICS. |
| WO2010146299A1 (en) * | 2009-06-16 | 2010-12-23 | Sanofi-Aventis | Azetidine-derived esters, preparation thereof, and therapeutic use thereof as cannabinoid receptor modulators |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| WO2011157827A1 (en) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-one derivatives as inhibitors of lipases and phospholipases |
| US9440948B2 (en) | 2010-09-03 | 2016-09-13 | University Of Florida Research Foundation, Inc. | Nicotine compounds and analogs thereof, synthetic methods of making compounds, and methods of use |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2013001505A3 (en) * | 2011-06-29 | 2013-03-07 | Adamed Sp. Z O.O. | Sulphonamide derivatives of alicyclic amines for the treatment of central nervous system diseases |
| EP2754653A2 (en) * | 2011-09-05 | 2014-07-16 | Korea Institute of Science and Technology | Azetidine derivative and antidepressant composition containing same |
| EP2754653A4 (en) * | 2011-09-05 | 2015-02-18 | Korea Inst Sci & Tech | Azetidine derivative and antidepressant composition containing same |
| US9120771B2 (en) | 2011-09-05 | 2015-09-01 | Korea Institute Of Science And Technology | Azetidine derivative and antidepressant composition including the same |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2007522255A (en) | 2007-08-09 |
| CA2556565A1 (en) | 2005-08-25 |
| EP1718609A1 (en) | 2006-11-08 |
| BRPI0507801A (en) | 2007-07-10 |
| KR20060124756A (en) | 2006-12-05 |
| NO20064078L (en) | 2006-09-11 |
| AU2005212835A1 (en) | 2005-08-25 |
| RU2006133258A (en) | 2008-04-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2005077897A1 (en) | Substituted azetidine compounds, their preparation and use as medicaments | |
| US7524868B2 (en) | Substituted pyrazoline compounds, their preparation and use as medicaments | |
| UA73554C2 (en) | A pharmaceutical composition containing azetidine derivatives, azetidine derivatives and preparation thereof | |
| US20070066587A1 (en) | Substituted azetidine compounds, their preparation and use as medicaments | |
| EP1757587A1 (en) | Substituted pyrazoline compounds, their preparation and use as medicaments | |
| EP1743890A1 (en) | 4,5-Dihydro-1H-pyrazole derivatives, their preparation and use as medicaments | |
| WO2006069807A1 (en) | Substituted phenyl-piperazine compounds, their preparation and use in medicaments | |
| JP2008525388A (en) | N-[(4,5-diphenyl-3-alkyl-2-thienyl) methyl] amine (amide, sulfonamide, carbamic acid and urea) derivatives as cannabinoid CB1 receptor antagonists | |
| WO2007017124A1 (en) | Amorphous phase of a substituted pyrazoline, its preparation and use as medicament | |
| WO2007009705A1 (en) | (rac)-n-piperidinyl-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4,5-dihydr0-1h-pyrazole-3-carboxamide hydrates | |
| WO2007017126A2 (en) | POLYMORPH OF N-PIPERIDINYL-5- (4-CHLOROPHENYL) -1- (2, 4-DICHLOROPHENYL) -4, 5-DIHYDRO-lH-PYRAZOLE- 3 -CARBOXAMIDE AND ITS USE AS A CAMNABINOID RECEPTOR MODULATOR | |
| EP1743892A1 (en) | Substituted pyrazoline compounds, their preparation and use as medicaments | |
| EP1743888A1 (en) | Carbonyl substituted pyrazoline compounds, their preparation and use as CB1 receptor modulators | |
| US7790727B2 (en) | Benzenesulfonanilide compounds suitable for treating disorders that respond to modulation of the serotonin 5-HT6 receptor | |
| JP2008088057A (en) | N-substituted-n-(4-piperidinyl)amide derivative | |
| EP1743894A1 (en) | Heterocyclyl-substituted pyrazoline compounds, their preparation and use as medicaments | |
| MXPA06009336A (en) | Substituted azetidine compounds, their preparation and use as medicaments | |
| US20090149472A1 (en) | Salts of substitutted pyrazoline compounds, their preparation and use and medicaments | |
| MX2011001669A (en) | Azetidine polysubstituted compounds, preparation thereof, and therapeutic application thereof. | |
| EP1749821A1 (en) | Quaternary ammonium salts of substituted pyrazoline compounds, their preparation and use as medicaments | |
| MXPA06009334A (en) | Substituted pyrazoline compounds, their preparation and use as medicaments | |
| EP1743887A1 (en) | Thiocarbonyl-substituted pyrazoline compounds, their preparation and use as CB1 modulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 177456 Country of ref document: IL Ref document number: 4609/DELNP/2006 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006/06732 Country of ref document: ZA Ref document number: 200606732 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2556565 Country of ref document: CA Ref document number: PA/a/2006/009336 Country of ref document: MX Ref document number: 549201 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006553539 Country of ref document: JP Ref document number: 11505463 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005212835 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005715383 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2005212835 Country of ref document: AU Date of ref document: 20050216 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005212835 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020067018702 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006133258 Country of ref document: RU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580011514.6 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005715383 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067018702 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 11505463 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0507801 Country of ref document: BR |