VITAMIN D RECEPTOR MODULATORS
BACKGROUND OF THE INVENTION Vitamin D3 Receptor (VDR) is a ligand dependent transcription factor that belongs to the superfamily of nuclear hormone receptors. The VDR protein is 427 amino acids, with a molecular weight of -50 kDa. The VDR ligand, lα,25-dihydroxyvitamin D3 (the hormonally active form of Vitamin D) has its action mediated by its interaction with the nuclear receptor known as Vitamin D receptor ("VDR"). The VDR ligand, lα,25- dihydroxyvitamin D3 (l ,25(OH)2D3) acts upon a wide variety of tissues and cells both related to and unrelated to calcium and phosphate homeostasis. The activity lα,25-dihydroxyvitamin D3 in various systems suggests wide clinical applications. However, use of conventional VDR ligands is hampered by their associated toxicity, namely hypercalcemia (elevated serum calcium). Cunently, lα,25(OH)2U3 marketed as Rocaltrol® pharmaceutical agent ( product of Hoffmann-La Roche), is administered to kidney failure patients undergoing chronic kidney dialysis to treat hypocalcemia and the resultant metabolic bone disease. Other therapeutic agents, such as Calcipofriol® (synthetic analog of l ,25(OH)2D3 ) show increased separation of binding affinity on VDR from hypercalcemic activity. Chemical modifications of l ,25(OH)2D3 have yielded analogs with attenuated calcium mobilization effects (R. Bouillon et. al., Endocrine Rev. 1995, 16, 200-257). One such analog, Dovonex ® pharmaceutical agent (product of Bristol-Meyers Squibb Co.), is cunently used in Europe and the United States as a topical treatment for mild to moderate psoriasis (K. Kragballe et. al., Br. J. Dermatol. 1988, 119, 223-230). Other Vitamin D3 mimics have been described in the publication, Vitamin D
Analogs: Mechanism of Action of Therapeutic Applications, by Nagpal, S.; Lu, J.; Boehm, M. F., Cun. Med. Chem. 2001, 8, 1661-1679.
Although some degree of separation between the beneficial action and calcium raising (calcemic) effects has been achieved with these VDR ligands, to date the separation has been insufficient to allow for oral administration to treat conditions such as osteoporosis, cancers, leukemias, and severe psoriasis. One example of a major class of disorder that could benefit from VDR mediated biological efficacy in the absence of hypercalcemia is osteoporosis. Osteoporosis is a systemic disorder characterized by decreased bone mass and microarchitectural deterioration of bone tissue leading to bone fragility and increased susceptibility to fractures of the hip, spine, and wrist (World Health Organization WHO 1994). Osteoporosis affects an estimated 75 million people in the United States, Europe, and Japan. Within the past few years, several antiresorptive therapies have been introduced. These include bisphosphonates, hormone replacement therapy (HRT), a selective estrogen receptor modulator (SERM), and calcitonins. These treatments reduce bone resorption, bone formation, and increase bone density. However, none of these treatments increase tme bone volume nor can they restore lost bone architecture. Another major disorder that could benefits from VDR mediated biological activity is psoriasis. Psoriasis is one of the most common dermatologic diseases and is a chronic inflammatory skin condition characterized by erythematous, sharply demarcated papules and rounded plaques, covered by silvery micaceous scale. Synthetic VDR ligands with reduced calcemic potential have been synthesized. For example, a class of bis-phenyl compounds stated to mimic l , 25-dihydroxyvitamin D3 is described in US Patent No. 6,218,430 and the article; "Novel nonsecosteroidal vitamin D mimics exert VDR-modulating activities with less calcium mobilization than lα, 25-Dihydroxyvitamin D3" by Marcus F. Boehm, et. al., Chemistry & Biology 1999.
Vol 6, No. 5, pgs. 265-275. Synthetic VDR ligands having an aryl-thiophene nucleus are described in United States provisional patent application SN 60/384151, filed 29 May 2002. There remains a need for improved treatments using alternative or improved pharmaceutical agents that mimic l , 25-dihydroxyvitamin D3 to stimulate bone
formation, restore bone quality, and treat other diseases without the attendant disadvantage of hypercalcemia.
SUMMARY OF THE INVENTION
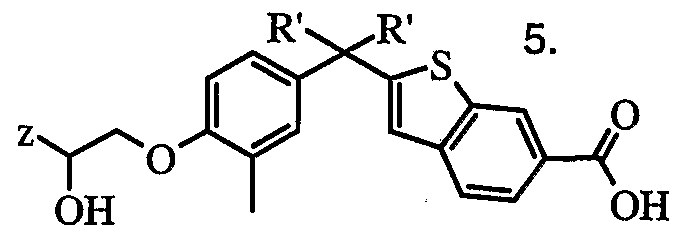
Novel compounds having a nucleus of Formula "(TP)", "(PT)", or (PT6) have been found effective as Vitamin D Receptor modulators (VDRM):
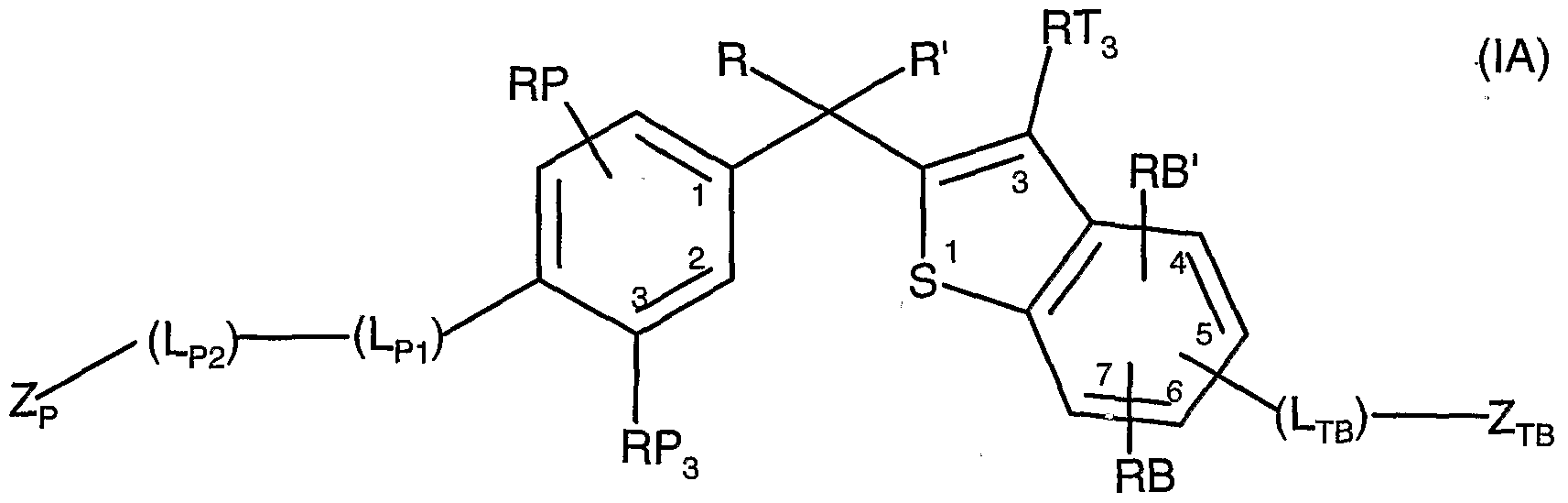
Compounds of the invention with VDR modulating activities are represented by formulae (IA)
wherein the variables R, R', RP, RP3, Lpi, Lp2, Zp, RB, RB', RT , Lrβand Zj/g are as hereinafter defined. Compounds of the invention with VDR modulating activities are also represented by formulae (IB)
wherein the variables R, R', RP, RP
3, L
P1, L
κ, Zp, RB7, RB, RB
4, RT
3, L
BTand ZβT are as hereinafter defined. Compounds of the invention with VDR modulating activities are also represented by formulae (IC)
wherein the variables R, R', RP, RP
3, L
P1, L
P2, Zp, RB
7, RB, RB
4, RT
3, L
Bτand ZβT are as hereinafter defined. In another aspect, the present invention is directed towards pharmaceutical compositions containing pharmaceutically effective amounts of compounds of formulae IA, IB, IC or a pharmaceutically acceptable salt or a prodrug thereof, either singly or in combination, together with pharmaceutically acceptable earners and/or auxiliary agents. Another aspect of the invention is a pharmaceutical formulation for treatment or prevention of osteoporosis containing pharmaceutically effective amounts of the vitamin D receptor modulator compound of formulae IA, IB, or IC alone or together with pharmaceutically effective amounts of co-agents conventionally used for the treatment of osteoporosis. Another aspect of the invention is a pharmaceutical formulation for treatment or prevention of psoriasis containing pharmaceutically effective amounts of the vitamin D receptor modulator compound of formulae IA, IB, or IC alone or together with pharmaceutically effective amounts of co-agents conventionally used for the treatment of psoriasis. Another aspect of the invention is a pharmaceutical formulation for treatment or prevention of prostate cancer containing pharmaceutically effective amounts of the vitamin D receptor modulator compound of formulae IA, IB, IC alone or together with pharmaceutically effective amounts of co-agents conventionally used for the treatment of prostate cancer. Another aspect of the invention is to use the compounds of formulae IA, IB, or IC to treat disease states responsive to Vitamin D receptor ligands. Another aspect of the invention is the prevention and treatment of acne, actinic keratosis, alopecia, Alzheimer's disease, autoimmune induced diabetes, benign prostatic hyperplasia, bladder cancer, bone fracture healing, breast cancer, Crohn's disease, prostate cancer, colon cancer, Type I diabetes, host-graft rejection, hypercalcemia , Type II diabetes, leukemia, multiple sclerosis, insufficient sebum secretion, osteomalacia, osteoporosis, insufficient dermal firmness, insufficient dermal hydration, myelodysplastic syndrome, psoriatic arthritis, psoriasis, renal osteodystrophy, rheumatoid arthritis, scleroderma, sebonheic dermatitis, skin cancer, systemic lupus erythematosis, skin cell
da age from Mustard vesicants, ulcerative colitis, and wrinkles, by administering to a mammal in need thereof a pharmaceutically effective amount of a compound of Formulae IA, IB, or IC.
DETAILED DESCRIPTION OF THE INVENTION Definitions: The term, "abscess" refers to adverse complications often associated with surgery, trama, or diseases that predispose the host to abscess formation from encapsulated bacteria lymphocytes, macrophages, and etc. The term, "adhesion" refers to the adverse and abnormal union of surfaces normally separate by the formation of new fibrous tissue resulting from an inflammatory process. The term, "compound of the invention" refers to a compound represented by Formulae IA, IB, or IC or as set out as products of the Examples or synthesis schemes described herein. The term, "Active Ingredient" means a compound of the invention. The term, "Mustard" is inclusive of both sulfur mustards and nitrogen mustards, either alone or in any combnation. Examplary of such compounds are the vesicants; bis(2-chloroefhyl) sulfide (Chemical Agent Symbol HD), C1(CH2)2S(CH2)2C1 1,2- bis(2-chloroethylthio)ethane (Chemical Agent Symbol Q), C1(CH2)2S(CH2)2S(CH2)2C1; bis(2-chloroethylthioethyl) ether, Cl(CH2)2S(CH2)O(CH2)2S(CH2)2Cl (Chemical Agent
Symbol T); tris(2-chloroethyl) amine (Chemical Agent Symbol HN3) N(CH2CH2C1)3;
N-methyl-2,2'-dichlorodiethylamine (Chemical Agent Symbol NH2); and 2,2'- dichlorotriethylamine, CH3CH2N(CH2CH2C1)2 (Chemical Agent Symbol NH1). The term, "(Acidic Group)" means an organic group that acts as a proton donor capable of hydrogen bonding. Illustrative of an (Acidic Group) is a group selected from the following: -C(O)OH, -5-tetrazolyl,
The term, "-l,3-thiazolidine-2,4-dione-5-ethylidene", refers to the radical represented by the stractural formula:
The term, "-CH2-C(O)-N-pynolidine" refers to the radical represented by the structural formula:
The term, "-CH2-N-pynolidin-2-one" refers to the radical represented by the stractural formula:
The term, "-CH2-(l-methylpynolidin-2-one-3-yl)" refers to the organic radical represented by the stractural formula:
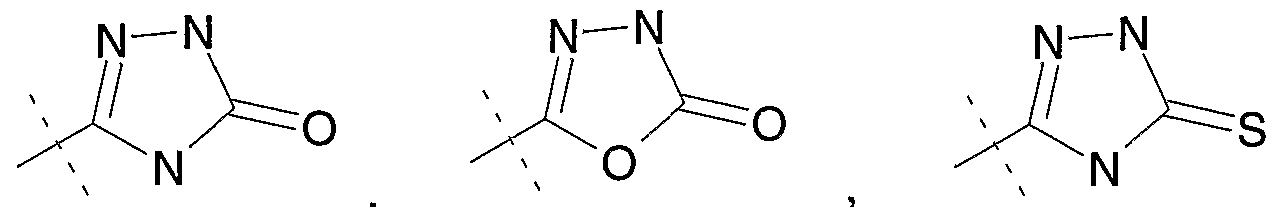
The term, "l,3,4-oxadiazolin-2-one-5-yl" refers to the organic radical represented by the structural formula:
The term, "l,3,4-oxadiazolin-2-thione-5-yl" refers to the organic radical represented by the structural formula:
The terml, "imidazolidine-2,4-dione-5-yl" refers to the organic radical represented by the structural formula:
The term, "isoxazol-3-ol-5-yl" refers to the organic radical represented by the structural formula:
The dotted line symbol crossing a solid line representing a bond
means that the bond so marked is the bond of attachment. The term, "mammal" includes humans. The term "halo" refer to fluorine, chlorine, bromine, and iodine. The term "sulfonate" refers to the group
where R'" is C1-C5 alkyl, C1-C5 fluoroalkyl ,
where R' is -CO
2H, -CO
2R'", -OH, -CF
3, or C1-C5 alkyl.
The term "sulfonamide" refers to the group methyl, ethyl, branched C3-C5 alkyl,
where R" is H, C1-C5 alkyl, C1-C5 fluoroalkyl , or O
■ 3 (C C
5 alkyl)
O or alkyl) halo
where R'" is C1-C5 alkyl, C1-C5 fluoroalkyl ,
where R' is -CO2H, -CO2R'", -OH, -CF3, or C1-C5 alkyl. The term, "Ci .3 alkyl" refers to an alkyl group selected from methyl, ethyl, n- propyl, and isopropyl. The term, "branched C3-C5 alkyl" is an alkyl group selected from 1-methylethyl;
1-mefhylpropyl; 2-methylpropyl; 1,1-dimethylethyl; 1,1-dimethylpropyl; 1,2-
dimethylpropyl; or 2,2-dimethylpropyl. Prefened branched C3-C5 alkyl groups are 2- methylpropyl and 1,1-dimethylethyl, with the 1,1-dimethylethyl group being most prefened. The term "alkenyl" refers to aliphatic groups wherein the point of attachment is a carbon-carbon double bond, for example vinyl, 1 -propenyl, and 1-cyclohexenyl. Alkenyl groups may be straight-chain, branched-chain, cyclic, or combinations thereof, and may be optionally substituted. Suitable alkenyl groups have from 2 to about 20 carbon atoms. The term "C1-C5 alkyl" refers to saturated aliphatic groups including straight- chain, branched-chain, and cyclic groups and any combinations thereof. Examples of C - C5 alkyl groups are methyl, ethyl, n-propyl, from 1-methylethyl; n-butyl, 1-methylpropyl;
2-methylpropyl; 1,1-dimethylethyl; n-amyl, 1,1-dimethylpropyl; 1,2-dimethylpropyl; and 2,2-dimethylpropyl. The term "cycloalkyl" includes organic radicals such as cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl. The term, "cycloalkenyl" includes organic radicals such as cyclopropenyl, cyclobutenyl, cyclopentenyl and cyclohexenyl. The term, "C1-C5 fluoroalkyP'is an alkyl group containing fluorine and includes organic radicals such as -CF3, -CHF2, -CH2F, -CF2CF3, -CHFCF3, -CH2CF3,
-CH2CHF2, and -CH2CH2F, with -CF3 being prefened. The abbreviation, "Me" means methyl. The abbreviation, "Et" means ethyl. The abbreviation, "iPr" means 1-methylethyl. The abbreviation, "tBu" means 1,1-dimethylethyl. The term,"terminal hydroxyalkyl" is a group selected from 3 -methyl-3 -hydroxypentyl , 3-methyl-3-hydroxypentenyl, 3-methyl-3-hydroxypentynyl, 3-ethyl-3-hydroxypentyl, 3-ethyl-3-hydroxypentenyl, 3-ethyl-3-hydroxypentynyl, 3 -ethyl-3 -hydroxy-4-methylpentyl,
3-ethyl-3-hydroxy-4-methylpentenyl, 3-ethyl-3-hydroxy-4-methylpentynyl, 3 -proρyl-3 -hydroxypentyl, 3 -propyl-3 -hydroxypentenyl, 3 -propyl-3 -hydroxypentynyl, 1 -hydroxy-2-methyl- 1 -(methylethyl)propyl, 2-methyl-3-hydroxy-4-dimethylpentyl, 2-methyl-3-hydroxy-3-ethylpentyl, 2-ethyl-3 -hydroxy-3 -ethylpentyl, 2-ethyl-3-hydroxy-4-dimethylpentyl, 1-hydroxycycloalkenyl; and 1 -hydroxycycloalkyl .
The term, "3 -methyl-3 -hydroxypentyl" refers to the radical having the stractural formula:
The term, "3 -methyl-3 -hydroxypentenyl" refers to the radical having the stractural formula (both cis and trans isomers):
The term, "3-methyl-3-hydroxypentynyl" refers to the radical having the stractural formula:
The term, "3-efhyl-3-hydroxypentyl" refers to the radical having the structural formula:
The term, "3 -ethyl-3 -hydroxypentenyl" refers to the radical having the structural formula (both cis and trans isomers):
The term, "3-ethyl-3-hydroxypentynyl" refers to the radical having the structural formula:
The term, "3 -propyl-3 -hydroxypentyl" refers to the radical having the stractural formula:
The term, "3 -propyl-3 -hydroxypentenyl" refers to the radical having the structural formula (both cis and trans isomers):
The term, "3-propyl-3-hydroxypentynyl" refers to the radical having the structural formula:
The term, "3-ethyl-3-hydroxy-4-methylpentyl" refers to the radical having the stractural formula:
The term, "3-ethyl-3-hydroxy-4-methylpentenyl" refers to the radical having the structural formula (both cis and trans isomers):
The term, "3-ethyl-3-hydroxy-4-methylpentynyl" refers to the radical having the stractural formula:
The term, "l-hydroxy-2-mefhyl-l-(methylethyl)propyl" refers to the radical having the stractural formula:
The term, "3-mefhyl-3-hydroxy-4,4-dimethylpentyl" refers to the radical having the stractural formula:
The term, "3-methyl-3-hydroxy-4,4-dimethylpentenyl." refers to the radical having the stractural formula (both cis and trans isomers):
The term, "3-methyl-3-hydroxy-4,4-dimethylpentynyl" refers to the radical having the stractural formula:
The term, "3-ethyl-3-hydroxy-4,4-dimethylpentyl" refers to the radical having the structural formula:
The tem , "3-ethyl-3-hydroxy-4,4-dimethylpentenyl" refers to the radical having the stractural formula (both cis and trans isomers):
The term, "3-ethyl-3-hydroxy-4,4-dimethylpentynyl" refers to the radical having the stractural formula:
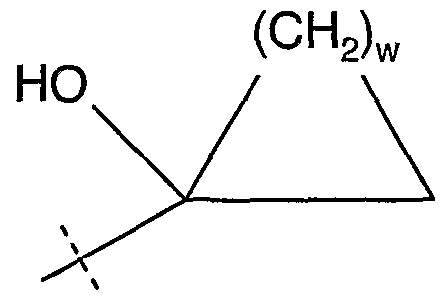
The term, "1-hydroxycycloalkenyl" refers to a radical selected from 1-hydroxycyclopentenyl, 1-hydroxycyclohexenyl, 1-hydroxycycloheptenyl, or 1- hydroxycyclooctenyl. The term "hydroxycycloalkyl" refers to a radical having the general structural formula:
where w is an integer from 1 to 6 and the hydroxyl radical is substituted on any ring carbon atom. The term "1-hydroxycycloalkyl" refers to a radical having the general stractural formula:
Examples of 1-hydroxycycloalkyl radicals are
1 -hydroxycyclopropyl, 1 -hydroxycyclobutyl, 1 -hydroxycyclopentyl,
1-hydroxycyclohexyl, 1-hydroxycycloheptyl, and 1-hydroxycyclooctyl. The abbreviation, "Me" means methyl. The abbreviation, "Et" means ethyl. The abbreviation, "iPr" means 1-methylethyl. The abbreviation, "nPr" means n-propyl. The abbreviation, "3Me3OH-Pentyl" means 3-methyl-3-hydroxypentyl. The abbreviation, "3Me3OH-Pentenyl" means 3 -methyl-3 -hydroxypentenyl The abbreviation, "3Me3OH-Pentynyl" means 3-methyl-3-hydroxypentynyl The abbreviation, "3Et3OH-Pentyl" means 3-ethyl-3-hydroxypentyl. The abbreviation, "3Et3OH-Pentenyl" means 3-ethyl-3-hydroxyρentenyl The abbreviation, "3Et3OH-Pentynyl" means 3-ethyl-3-hydroxypentynyl The abbreviation, "3Pr3OH-Pentyl" means 3 -propyl-3 -hydroxypentyl. The abbreviation, "3Pr3OH-Pentenyl" means 3-propyl-3-hydroxypentenyl. The abbreviation, "3Pr3OH-Pentynyl" means 3 -propyl-3 -hydroxypentynyl. The abbreviation, "3Et3OH4Me-Pentyl" means 3-ethyl-3-hydroxy-4- methylpentyl. The abbreviation, "3Et3OH4Me-Pentenyl" means 3-ethyl-3-hydroxy-4- methylpentenyl, The abbreviation, "3Et3OH4Me-Pentynyl" means 3-ethyl-3-hydroxy-4- methylpentynyl. The abbreviation, "lOH2MelMeEt-Proρyl" means l-hydroxy-2-methyl-l- (methylethyl)propyl.
Compounds of the Invention: The compounds of the invention with vitamin receptor modulating (VDRM) activity are represented by formula (IA) or a pharmaceutically acceptable salt or a prodrug derivative thereof:
wherein R and R' are independently C1-C5 alkyl, C1-C5 fluoroalkyl, or together R and R' form a substituted or unsubstituted, saturated or unsaturated carbocyclic ring having from 3 to 8 carbon atoms; RP
3 and RB are independently selected from the group consisting of hydrogen, halo, C1-C5 alkyl, C1-C5 fluoroalkyl, -O-C1-C5 alkyl, -S-C1-C5 alkyl, -O-C1-C5 fluoroalkyl, -CN, -NO2, acetyl, -S-C1-C5 fluoroalkyl, C2-C5 alkenyl, C3-C5 cycloalkyl, and C3-C5 cycloalkenyl; RP, RT
3, and RB' are independently selected from hydrogen, halo, Ci -C5 alkyl,
Ci-C5 fluoroalkyl, -O-C1-C-5 alkyl, -S-C1-C5 alkyl, -O-C1-C5 fluoroalkyl, -CN, -NO2, acetyl, -S-C1-C5 fluoroalkyl, C2-C5 alkenyl, C3-C5 cycloalkyl, or C3-C5 cycloalkenyl; (LP , (Lγι), and (LTB) are divalent linking groups independently selected from the group consisting of
a bond
-(CH2)— o- -(CH2)— S -
(CH
2)
m— CH =CH- and — (CH
2)
m— C≡C— where m is 0, 1, or 2, and each R40 is independently hydrogen, C1-C5 alkyl, or C1-C5 fluoroalkyl; Zp is branched C3-C5 alkyl, 3 -methyl-3 -hydroxypentyl ,
3 -methyl-3 -hydroxypentenyl,
3 -methyl-3 -hydroxypentynyl,
3-ethyl-3-hydroxypentyl,
3 -ethyl-3 -hydroxypentenyl,
3 -ethyl-3 -hydroxypentynyl,
3-ethyl-3-hydroxy-4-methylpentyl,
3-ethyl-3-hydroxy-4-methylpentenyl,
3-ethyl-3-hydroxy-4-methylpentynyl,
3 -propyl-3 -hydroxypentyl,
3 -propyl-3 -hydroxypentenyl,
3-ρropyl-3-hydroxypentynyl,
1 -hydroxy-2-methyl- 1 -(methylethyl)propyl,
2-methyl-3-hydroxy-4-dimethylpentyl, .
2-methyl-3-hydroxy-3~ethylpentyl,
2-ethyl-3 -hydroxy-3 -ethylpentyl,
2-ethyl-3-hydroxy-4-dimethylpentyl,
3-methyl-3-hydroxy-4,4-dimethylpentyl,
3-methyl-3-hydroxy-4,4-dimethylpentenyl,
3 -methyl-3 -hydroxy-4,4-dimethylpentyl,
3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
3-ethyl-3-hydroxy-4,4-dimethylpentenyl,
3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
1 -hydroxycycyclopentenyl,
1 -hydroxycyclohexenyl,
1 -hydroxycycloheptenyl,
1 -hydroxycyclooctenyl,
1 -hydroxycyclopropyl,
1 -hydroxycyclobutyl,
1-hydroxycyclopentyl,
1 -hydroxycyclohexyl,
2-oxocyclohexyloxy
2-oxocyclohexylmethyl
3 -methyl-2-oxocyclohexyloxy, 3-methyl-2-oxocyclohexylmethyl, 3 ,3 -dimethyl-2-oxocyclohexyloxy, 3,3-dimethyl-2-oxocyclohexylmethyl, 2-hydroxycyclohexyloxy, 2-hydroxycyclohexylmethyl, 3 -methyl-2-hydroxycyclohexyloxy , 3-methyl-2-hydroxycyclohexylmethyl, 3 ,3 -dimethyl-2-hydroxycyclohexyloxy, 3 ,3 -dimethyl-2-hydroxycyclohexylmethyl , 1-hydroxycycloheptyl, or 1 -hydroxycyclooctyl; provided, however, that when Zp is 3-methyl-3-hydroxypentyl, 3 -methyl-3 -hydroxypentenyl, 3 -methyl-3 -hydroxypentynyl, 3 -ethyl-3 -hydroxypentyl, 3-ethyl-3-hydroxypentenyl, 3 -ethyl-3 -hydroxypentynyl , 3-ethyl-3-hydroxy-4-methylpentyl, 3-ethyl-3-hydroxy-4-methylpentenyl, 3-ethyl-3-hydroxy-4-methylpentynyl, 3-propyl-3-hydroxypentyl, 3-propyl-3-hydroxypentenyl, 3-propyl-3-hydroxypentynyl, 3-methyl-3-hydroxy-4,4-dimethylpentyl, 3-methyl-3-hydroxy-4,4-dimethylpentenyl, 3-methyl-3-hydroxy-4,4-dimethylpentyl, 3-ethyl-3-hydroxy-4,4-dimethylpentynyl, 3-ethyl-3-hydroxy-4,4-dimethylpentenyl, 3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
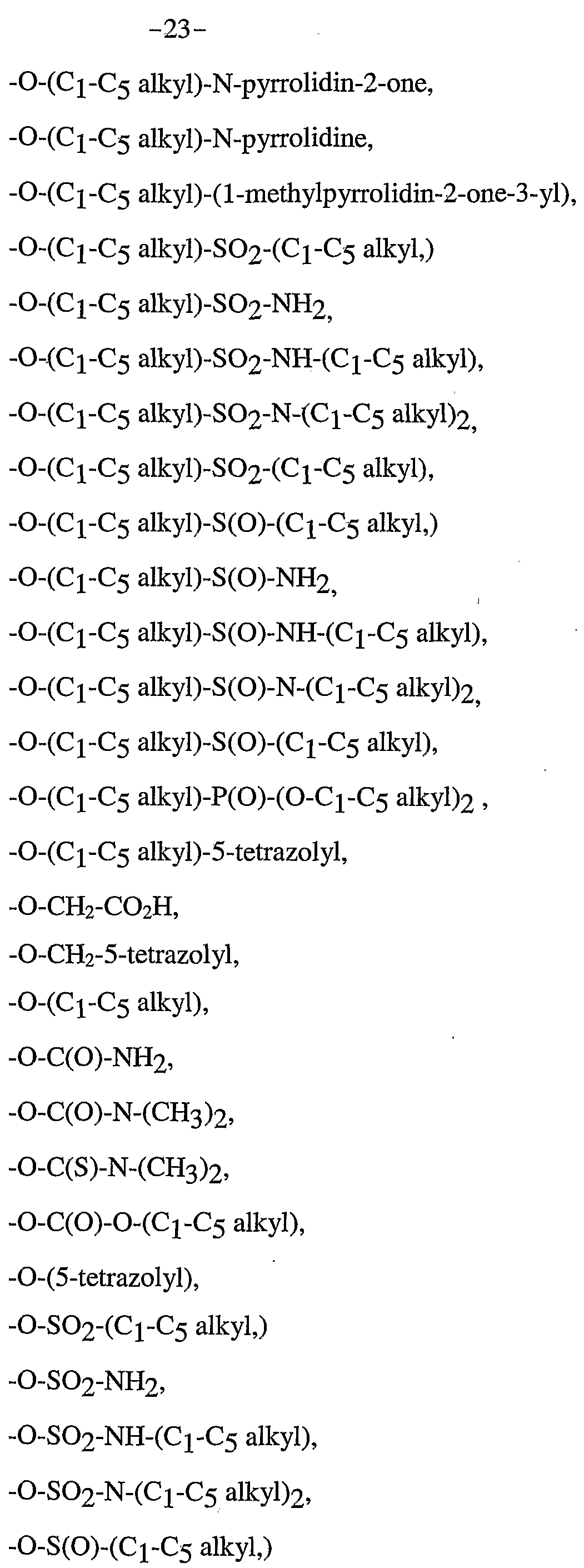
2-methyl-3-hydroxy-4-dimethylρentyl, 2-methyl-3-hydroxy-3-ethylpentyl, 2-ethyl-3-hydroxy-3-ethylpentyl, 2-ethyl-3-hydroxy-4-dimethylpentyl, or 1 -hydroxy-2-methyl- 1 -(methylethyl)propyl; then (LP and (Li^) combine as a bond; Z /B is selected from -O-(Cχ-C5 alkyl), -O-(C2-C5 alkenyl), -O-(C3-C5 cycloalkyl), -O-(C3-C5 cycloalkenyl), -O-(Cχ-C5 hydroxyalkyl), -O-(Cχ-C5 fluoroalkyl), -O-(Cχ-C5 alkyl)-ρhenyl, -O-(Cι -C5 alkyl)-(O)-(Cι-C5 alkyl), -O-(Cχ-C5 alkyl) NH2, -O-(C!-C5 alkyl)-NH-(C!-C5 alkyl)2 , -O-(Cι-C5 alkyl)-C(O)-NH2, -O-(C!-C5 alkyl)-C(O)-NH-(C1-C5 alkyl), -O-(C!-C5 alkyl)-C(O)-N-(C1-C5 alkyl)2, -O-(C!-C5 alkyl)-C(O)-OH, -O-(C1-C5 alkyl)-C(O)-NH-5-tetrazolyl, -O-(C!-C5 alkyl)-C(O)-(C1-C5 alkyl), -O-(C!-C5 alkyl)-C(O)-(O-C1-C5 alkyl), -O-(C!-C5 alkyl)-NH2, -O-(Cχ-C5 alkyl)-NH-(Cι-C5 alkyl), -O-(C!-C5 alkyl)-N-(Cι-C5 alkyl)2, -O-(Cχ-C5 alkyl NH-SO2-(C1-C5 alkyl),
-O-S(O)-NH2, -O-S(O)-NH-(C!-C5 alkyl), -O-S(O)-N-(Cι-C5 alkyl)2, -S-(Cχ-C5 alkyl), -S-(C2-C5 alkenyl), -S-(C3-C5 cycloalkyl), -S-(C3-C5 cycloalkenyl),
-S- C1 -C5 fluoroalkyl),
-S- C -C5 hydroxyalkyl),
-S- C -C5 alkyl)-phenyl,
-S-< C -C5 alkyl)-O-(Cι-C5 alkyl),
-S C -C5 alkyl)-C(O)-OH,
-S C -C5 alkyl)-C(O)-(Cι-C5 alkyl),
-S (C -C5 alkyl)-C(O)-NH2,
-S- C -C5 alkyl)-C(O)-NH-(Cι-C5 alkyl),
-S- C -C5 alkyl)-C(O)-N-(C1-C5 alkyl)2,
-S- C -C5 alkyl) NH2,
-S- C -C5 alkyl)-NH-(C1-C5 alkyl),
-s- C -C5 alkyl)-N-(Cι-C5 alkyl)2)
-s- C -C
5 alkyl)-NH-SO
2-(C
1-C
5 alkyl), -s- C -C5 alkyl)-N-pynolidin-2-one, -s- C -C5 alkyl)-N-pynolidine, -s- C -C5 alkyl)-( 1 -methylpynolidin-2-one-3 -yl), -s- C -C
5 alkyl)-SO
2-(C
1-C5 alkyl), -s- C -C
5 alkyl)-SO
2-NH
2, -s- C -C
5 alkyl)-SO
2-NH-(C
1-C
5 alkyl), -s- C -C
5 alkyl)-SO
2-N-(C
1-C
5 alkyl)
2,
-NHC(S)NH-(C!-C5 alkyl)-N-pynolidin-2-one, -NHC(S)NH-(C1-C5 alkyl)-N-pynolidine, -NHC(S)NH-(C1-C5 alkyl)-(l-methylpynolidin-2-one- 3-yl), -NHC(S)NH-(C1-C5 alkyl)-5-tetrazolyl,
-NHC(S)NH-(C!-C5 alkyl)-SO2-(C1-C5 alkyl), -NHC(S)NH-(C1-C5 alkyl)-SO2-NH2, -NHC(S)NH-(C!-C5 alkyl)-SO2-NH-(Cι-C5 alkyl), -NHC(S)NH-(C!-C5 alkyl)-SO2-N-(C1-C5 alkyl)2, -NHC(S)NH-(Cι-C5 alkyl)-S(O)-(C1-C5 alkyl), -NHC(S)NH-(C1-C5 alkyl)-S(O)-NH2, -NHC(S)NH-(Cι-C5 alkyl)-S(O)-NH-(C!-C5 alkyl), -NHC(S)NH-(C!-C5 alkyl)-S(O)-N-(C!-C5 alkyl)2, -NHC(S)NH-(C!-C5 alkyl)-P(O)-(O-Cι-C5 alkyl)2 , -NHC(O)NH2, -NHC(O)NH-(Cι-C5 alkyl), -NHC(O)N-(Cι-C5 alkyl)2, -NHC(O)NH-(C2-C5 alkenyl), -NHC(O)NH-(C3-C5 cycloalkyl), -NHC(O)NH-(C3-C5 cycloalkenyl), -NHC(O)NH-(Cι-C5 hydroxyalkyl), -NHC(O)NH-(C!-C5 fluoroalkyl), -NHC(O)NH-phenyl, -NHC(O)NH-(C!-C5 alkyl)-NH2, -NHC(O)NH-(Cι-C5 alkyl)-NH-(Cχ-C5 alkyl), -NHC(O)NH-(Cι-C5 alkyl)-N-(Cl-C5 alkyl)2j -NHC(O)NH-(Cl-C5 alkyl)-O-<Cι-C5 alkyl), -NHC(O)NH-(Cι-C5 alkyl)-NH2,
-NHC(O)NH-(C1-C5 alkyl NH-CCx-Cs alkyl),
-NHC(O)NH-(Cι-C5 alkyl)-N-(C1-C5 alkyl)2,
-NHC(O)NH-(Cχ-C5 alkyl)-C(O)-NH2,
-NHC(O)NH-(C!-C5 alkyl)-C(O)-NH-(C1-C5 alkyl),
-NHC(O)NH-(C1-C5 alkyl)-C(O)-N-(C1-C5 alkyl)2,
-NHC(O)NH-(C!-C5 alkyl)-NH-SO2-(C1-C5 alkyl),
-NHC(O)NH-(C1-C5 alkyl)-N-pynolidin-2-one,
-NHC(O)NH-(Cι-C5 alkyl)-N-pyrrolidine,
-NHC(O)NH-(C!-C5 alkyl)- (l-methylpyrrolidin-2-one-3-yl), -NHC(O)NH-(C!-C5 alkyl)-C(O)-OH,
-NHC(O)NH-(Cι-C5 alkyl)-C(O)-O-(C1-C5 alkyl),
-NHC(O)NH-(C1-C5 alkyl)-5-tetrazolyl,
-NHC(O)NH-(C!-C5 alkyl)-SO2-(C1-C5 alkyl),
-NHC(O)NH-(Cι -C5 alkyl)-SO2-NH2,
-NHC(O)NH-(C!-C5 alkyl)-SO2-NH-(C1-C5 alkyl),
-NHC(O)NH-(C!-C5 alkyl)-SO2-N-(C1-C5 alkyl)2,
-NHC(O)NH-(C1-C5 alkyl)-P(O)-O-(C1-C5 alkyl)2 ,
-NH2,
-NH-(C!-C5 alkyl),
-NH-CH2-C(O)OH,
-N-(Cι-C5 alkyl)2,
-NH-C(O)-NH2,
-NH-C(O)-NH-(C1-C5 alkyl),
-NH-C(O)-N-(Cι-C5 alkyl)2,
-NH-C(O)-(Cι -C5 alkyl),
-NH-SO2-(Cι -C5 alkyl),
-NH-S(0)-(Cχ-C5 alkyl),
-N(CH3)(OCH3),
-N(OH)(CH3),
-N-pynolidin-2-one,
-N-pynolidine,
-( 1 -methylpynolidin-2-one-3 -yl),
-CO2H,
-CO2Me,
-CO2Et,
-C(O)CH2S(O)Me,
-C(O)CH2S(O)Et,
-C(O)CH2S(O)2Me,
-C(O)CH2S(O)2Et,
-C(O)CH2CH2S(O)Me,
-C(O)CH2CH2S(O)Et,
-C(O)CH2CH2S(O)2Me,
-C(O)CH2CH2S(O)2Et,
-C(O)CH(Me)CH2CO2H,
-C(O)CH(Me)CH2CO2Me,
-C(O)CH(Me)CH2CO2Et,
-C(O)CH(Me)CH2CO2iPr,
-C(O)CH(Me)CH2CO2tBu,
-C(O)CH(Me)CH(Me)CO2H,
-C(O)CH(Me)CH(Me)CO2Me,
-C(O)CH(Me)CH(Me)CO2Et,
-C(O)CH(Me)CH(Me)CO2iPr,
-C(O)CH(Me)CH(Me)CO2tBu,
-C(O)CH(Me)C(Me) 2CO2H,
-C(O)CH(Me)C(Me) 2CO2Me, -C(O)CH(Me)C(Me) 2CO2Et, -C(O)CH(Me)C(Me) 2CO2iPr, -C(O)CH(Me)C(Me) 2CO2tBu, -C(O)CH(Me)CH(Et)CO2H, -C(O)CH(Me)CH(Et)CO2Me, -C(O)CH(Me)CH(Et)CO2Et, -C(O)CH(Me)CH(Et)CO2iPr, -C(O)CH(Me)CH(Et)CO2tBu, -C(O)C(O)OH, -C(O)C(O)NH2, -C(O)C(O)NHMe, -C(O)C(O)NMe2, -C(O)NH2, -C(O)NMe2, -C(O)NH-CH2-C(O)OH, -C(O)NH-CH2-C(O)OMe, -C(O)NH-CH2-C(O)OEt, -C(O)NH-CH2-C(O)OiPr, -C(O)NH-CH2-C(O)OtBu, -C(O)NH-CH(Me)-C(O)OH, -C(O)NH-CH(Me)-C(O)OMe, -C(O)NH-CH(Me)-C(O)OEt, -C(O)NH-CH(Me)-C(O)iPr, -C(O)NH-CH(Me)-C(O)tBu, -C(O)NH-CH(Et)-C(O)OH, -C(O)NH-C(Me)2-C(O)OH, -C(O)NH-C(Me)2-C(O)OMe, -C(O)NH-C(Me)2-C(O)OEt, -C(O)NH-C(Me)2-C(O)iPr,
-C(O)NH-C(Me)2-C(O)tBu, -C(O)NH-CMe(Et)-C(O)OH, -C(O)NH-CH(F)-C(O)OH, -C(O)NH-CH(CF3)-C(O)OH, -C(O)NH-CH(OH)-C(O)OH, -C(O)NH-CH(cyclopropyl)-C(O)OH, -C(O)NH-C(Me)2-C(O)OH, -C(O)NH-C(Me)2-C(O)OH, -C(O)NH-CF(Me)-C(O)OH, -C(O)NH-C(Me)(CF3)-C(O)OH, -C(O)NH-C(Me)(OH)-C(O)OH, -C(O)NH-C(Me)(cyclopropyl)CO2H
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH2-C(O)OMe,
-C(O)NMe-CH2-C(O)OEt,
-C(O)NMe-CH2-C(O)OiPr,
-C(O)NMe-CH2-C(O)tBu,
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH(Me)-C(O)OH,
-C(O)NMe-CH(F)-C(O)OH,
-C(O)NMe-CH(CF3)-C(O)OH,
-C(O)NMe-CH(OH)-C(O)OH,
-C(O)NMe-CH(cyclopropyl)-C(O)OH,
-C(O)NMe-C(Me)2-C(O)OH,
-C(O)NMe-CF(Me)-C(O)OH,
-C(O)NMe-C(Me)(CF3)-C(O)OH,
-C(O)NMe-C(Me)(OH)-C(O)OH,
-C(O)NMe-C(Me)(cyclopropyl)-C(O)OH,
-C(O)NHS(O)Me,
-C(O)NHSO2Me,
-C(O)-NH-5-tetrazolyl, -C(O)NHS(O)Me,
-C(O)NHS(O)Et, -C(O)NHSO2Me,
-C(O)NHSO2Et,
-C(O)NHS(O)iPr, -C(O)NHSO2iPr,
-C(O)NHS(O)tBu, -C(O)NHSO2tBu,
-C(O)NHCH2S(O)Me,
-C(O)NHCH2S(O)Et,
-C(O)NHCH2SO2Me,
-C(O)NHCH2SO2Et,
-C(O)NHCH2CH2S(O)Me,
-C(O)NHCH2CH2S(O)Et,
-C(O)NHCH2CH2SO2Me,
-C(O)NHCH2CH2SO2Et,
-C(O)N(Me)S(O)Me, -C(O)N(Me)SO2Me,
-C(O)-N(Me)-5-tetrazolyl, -C(O)N(Me)S(O)Me, -C(O)N(Me)S(O)Et, -C(O)N(Me)SO2Me,
-C(O)N(Me)SO2Et,
-C(O)N(Me)S(O)iPr,
-C(O)N(Me))SO2iPr,
-C(O)N(Me))S(O)tBu,
-C(O)N(Me)SO2tBu,
-C(O)N(Me)CH2S(O)Me,
-C(O)N(Me)CH2S(O)Et,
-C(O)N(Me)CH2SO2Me,
-C(O)N(Me)CH2SO2Et, -C(O)N(Me)CH2CH2S(O)Me, -C(O)N(Me)CH2CH2S(O)Et, -C(O)N(Me)CH2CH2SO2Me, -C(O)N(Me)CH2CH2SO2Et, -CH2CO2H, -CH2-5-tetrazolyl, -CH2CO2Me, -CH2CO2Et, -CH2NHS(O)Me, -CH2NHS(O)Et, -CH2NHSO2Me, -CH2NHSO2Et, -CH2NHS(O)iPr, -CH2NHSO2iPr, -CH2NHS(O)tBu, -CH2NHSO2tBu, -CH2NHCH2CH2SO2CH3, -CH2NH(CH2CO2H), -CH2N(C(O)Me)(CH2CO2H), -CH2-N-pynolidin-2-one, -CH2-(l-methylpyrrolidin-2-one-3-yl), -CH2S(O)Me, -CH2S(O)Et, -CH2S(O)2Me, -CH2S(O)2Et, -CH2S(O)iPr, -CH2S(O)2iPr,
-CH2S(O)tBu,
-CH2S(O)2tBu,
-CH2CO2H, CH2C(O)NH2,
-CH2C(O)NMe2,
-CH2C(O)NHMe,
-CH2C(O)-N-pynolidine,
-CH2S(O)2Me, CH2S(O)Me,
-CH(OH) CO2H,
-CH(OH)C(O)NH2,
-CH(OH)C(O)NHMe,
-CH(OH)C(O)NMe2,
-CH(OH)C(O)NEt2,
-CH2CH2CO2H,
-CH2CH2CO2Me,
-CH2CH2CO2Et,
-CH2CH2C(O)NH2,
-CH2CH2C(O)NHMe,
-CH2CH2C(O)NMe2,
-CH2CH2-5-tetrazolyl,
-CH2CH2S(O)2Me,
-CH2CH2S(O)Me,
-CH2CH2S(O)2Et,
-CH2CH2S(O) Et,
-CH2CH2S(O)iPr,
-CH2CH2S(O)2iPr,
-CH2CH2S(O)tBu,
-CH2CH2S(O)2tBu,
IΛ © IΛ © © o
-l,3,4-oxadiazolin-2-one-5-yl, -imidazolidine-2,4-dione-5-yl, -isoxazol-3-ol-yl, or -l,3,4-oxadiazolin-2-thione-5-yl; provided that RB is substituted at either the 6 or 7 position of the benzothiophene ring, except that RB is substituted only at the 7 position of the benzothiophene ring when Z
B is at the 6 position.; and provided that -(L
TB)-Z
TB is substituted at either the 5 or 6 position of the benzothiophene ring; and
provided that RB is substituted at either the 6 or 7 position of the benzot -hi -op -h
ei
ne ring, except that RB is substituted only at the 7 position of the benzothiophene ring when the group -(LTB)-ZTB is at the 6 position.; and provided that RB' is substituted at either the 4 or 5 position of the benzothiophene ring, except that RB' is substituted only at the 5 position of the benzothiophene ring when the group -(LTB)-ZTB is at the 6 position of the phenyl ring; and provided that RP is substituted at either the 2, or 5 or 6 position of the phenyl ring. The compounds of the invention with vitamin receptor modulating (VDRM) activity are represented by formula (IB) or a pharmaceutically acceptable salt or a prodrug derivative thereof:
wherein the variables R, R', RP, RP
3, Zp, RBγ, RB, RB
4, RT
3, and Zg- are as hereinafter defined, wherein R and R' are independently C1-C5 alkyl, C1-C5 fluoroalkyl, or together R and R' form a substituted or unsubstituted, saturated or unsaturated carbocyclic ring having from 3 to 8 carbon atoms; RP, RB
4, RT
3 and RB are independently selected from the group consisting of hydrogen, halo, C1-C5 alkyl, Cχ-C
5 fluoroalkyl, -O-Cχ-C5 alkyl, -S-C1-C5 alkyl, -O-Cχ-
C5 fluoroalkyl, -CN, -NO2, acetyl, -S-C1-C5 fluoroalkyl, C2-C5 alkenyl, C3-C5 cycloalkyl, and C3-C5 cycloalkenyl; RP3 and RB7 are independently selected from hydrogen, halo, C1-C5 alkyl, C1-C5 fluoroalkyl, -O-C1-C5 alkyl, -S-Cχ-C5 alkyl, -O-Cχ-C5 fluoroalkyl, -CN, -NO2, acetyl, -
S-C1-C5 fluoroalkyl, C2-C5 alkenyl, C3-C5 cycloalkyl, or C3-C5 cycloalkenyl;
(Lpι), (LP2), and (LBτ) are divalent linking groups independently selected from the group consisting of a bond
-« H2)— O-
(CH
2)
m— CH=CH— and — (CH
2)
m— C≡C- where m is 0, 1, or 2, and each R40 is independently hydrogen, C1-C5 alkyl, or C1-C5 fluoroalkyl; Zp is
branched C3-C5 alkyl,
3-methyl-3-hydroxypentyl,
3-methyl-3-hydroxypentenyl,
3 -methyl-3 -hydroxypentynyl,
3 -ethyl-3 -hydroxypentyl,
3 -ethyl-3-hydroxypentenyl ,
3 -ethyl-3 -hydroxypentynyl,
3-ethyl-3-hydroxy-4-methylpentyl,
3-ethyl-3-hydroxy-4-methylpentenyl,
3-ethyl-3-hydroxy-4-methylpentynyl,
3 -propyl-3 -hydroxypentyl,
3 -propyl-3 -hydroxypentenyl,
3 -propyl-3 -hydroxypentynyl,
1 -hydroxy-2-methyl- 1 -(methylethyl)propyl,
2-methyl-3-hydroxy-4-dimethylpentyl,
2-methyl-3-hydroxy-3-ethylpentyl,
2-ethyl-3 -hydroxy-3 -ethylpentyl,
2-ethyl-3-hydroxy-4-dimethylpentyl,
3-methyl-3-hydroxy-4,4-dimethylpentyl,
3-methyl-3-hydroxy-4,4-dimethylpentenyl,
3 -methyl-3 -hydroxy-4,4-dimethylpentyl,
3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
3-ethyl-3-hydroxy-4,4-dimethylpentenyl,
3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
1 -hydroxycycyclopentenyl ,
1-hydroxycyclohexenyl,
1-hydroxycycloheptenyl,
1 -hydroxycyclooctenyl,
1 -hydroxycyclopropyl,
1 -hydroxycyclobutyl,
1 -hydroxycyclopentyl,
1 -hydroxycyclohexyl,
2-oxocyclohexyloxy, 2-oxocyclohexylmexhyl, 3 -methyl-2-oxocyclohexyloxy, 3-methyl-2-oxocyclohexylmethyl, 3 ,3-dimethyl-2-oxocyclohexyloxy, 3,3-dimethyl-2-oxocyclohexylmethyl, 2-hydroxycyclohexyloxy, 2-hydroxycyclohexylmethyl, 3 -methyl-2-hydroxycyclohexyloxy, 3-methyl-2-hydroxycyclohexylmethyl, 3 ,3 -dimethyl-2-hydroxycyclohexyloxy, 3 ,3-dimethyl-2-hydroxycyclohexylmethyl , 1-hydroxycycloheptyl, or 1 -hydroxycyclooctyl ; provided, however, that when Zp is 3 -methyl-3 -hydroxypentyl , 3 -methyl-3 -hydroxypentenyl, 3-methyl-3-hydroxypentynyl, 3-ethyl-3-hydroxypentyl, 3-ethyl-3-hydroxypentenyl, 3-ethyl-3-hydroxypentynyl, 3-ethyl-3-hydroxy-4-methylpentyl, 3-ethyl-3-hydroxy-4-methylpentenyl, 3-ethyl-3-hydroxy-4-methylpentynyl, 3 -propyl-3 -hydroxypentyl, 3 -propyl-3 -hydroxypentenyl, 3-propyl-3-hydroxypentynyl, 3-methyl-3-hydroxy-4,4-dimethylpentyl, 3-methyl-3-hydroxy-4,4-dimethylpentenyl, 3-methyl-3-hydroxy-4,4-dimethylpentyl, 3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
3-ethyl-3-hydroxy-4,4-dimethylpentenyl, 3-ethyl-3-hydroxy-4,4-dimethylpentynyl, 2-methyl-3-hydroxy-4-dimethylpentyl, 2-methyl-3-hydroxy-3-ethylpentyl, 2-ethyl-3 -hydroxy-3 -ethylpentyl, 2-ethyl-3-hydroxy-4-dimetlιylpentyl, or 1 -hydroxy-2-methyl- 1 -(methylethyl)propyl ; then (Lp and (Lp
2) combine as a bond; Zj χ is selected from -O-(Cι-C
5 alkyl), -O-(C2~C5 alkenyl), -O-(C3-C5 cycloalkyl), -O-(C3~C5 cycloalkenyl), -O-(C -C5 hydroxyalkyl), -O-(C I-C5 fluoroalkyl), -O-(C -C5 alkyl)-phenyl, -O-(C
: l-C
5 alkyl)-(O)-(C
1-C
5 alkyl), -O-(C
] L-C
5 alkyl) NH
2, -O-(C
] L-C
5 alkyl)-NH-(Cι-C
5 alkyl)
2 ) -O-(C
] L-C
5 alkyl)-C(O)-NH
2, -O-(C
] L-C
5 alkyl)-C(O)-NH-(C
1-C
5 alkyl), -O-(C] L-C
5 alkyl)-C(O)-N-(Cι-C
5 alkyl)
2, -O-(C] L-C5 alkyl)-C(O)-OH, -O-(C]
L-C
5 alkyl)-C(O)-NH-5-tetrazolyl, -O-(C] L-C
5 alkyl)-C(O)-(Cι-C
5 alkyl), -O-(C
] L-C5 alkyl)-C(O)-(O-Cι-C5 alkyl), -O-(C] L-C5 alkyl)-NH
2; -O-(C] [-C5 alkyl)-NH-(Cι-C5 alkyl), -O-(C]
L-C
5 alky^-N-^x-Cs alkyl)
2,
-O-S(O)-(Cι-C
5 alkyl,) -O-S(O)-NH
2, -O-S(O)-NH-(C!-C
5 alkyl), -O-S(O)-N-(C!-C
5 alkyl)
2, -S-(C!-C
5 alkyl), -S-(C
2-C5 alkenyl), -S-(C -C5 cycloalkyl), -S-(C3~C5 cycloalkenyl), -S-(C!-C
5 fluoroalkyl), -S-(Cχ-C
5 hydroxyalkyl), -S-(Ci-C5 alkyl)-phenyl, -S-(Cι-C
5
alkyl), -S-(C!-C5 alkyl)-C(O)-OH, -S-(C!-C
5 alkyl)-C(O)-(C
1-C
5 alkyl), -S-(C!-C
5
alkyl), -S-(Cι-C
5 alkyl)-C(O)-NH
2, -S-(Cχ-C
5 alkyl)-C(O)-NH-(Cχ-C5 alkyl), -S-(C!-C
5 alkyl)-C(O)-N-(C
1-C
5 alkyl)
2, -S-(Cι-C
5 alkyl) NH
2, -S-(Cι-C
5 alkyl), -S-(C!-C
5
lkyl)
2, -S-(Cι-C
5 alkyl)-NH-SO
2-(C
1-C
5 alkyl), -S-(Cι-C5 alkyl)-N-pynolidin-2-one, -S-(Cχ-C5 alkyl)-N-ρynolidine, -S-(Cι-C5 alkyl)-(l-methylρynolidin-2-one-3-yl), -S-(Cι-C
5 alkyl)-SO
2-(C
1-C5 alkyl), -S-(Cχ-C5 alkyl)-SO2-NH
2, -S-(Cι-C
5 alkyl)-SO
2-NH-(C
1-C
5 alkyl),
-S-(Cι -C
5 alkyl)-SO
2-N-(C
1-C
5 alkyl)
2, -S-(C -C
5 alkyl)-SO
2-(C
1-C
5 alkyl), -S-(C!-C
5 alkyl)-P(O)-(O-C
1-C
5 alkyl)
2 , -S-(Cχ-C5 alkyl)-5-tetrazolyl, -S-(Cχ-C5 alkyl)-S(O)-(Cι-C5 alkyl), -S-(C!-C5 alkyl)-S(O)-NH
2, -S-(C!-C5
alkyl), -S-(Ci-C
5 alkyl)-S(O)-N-<Cι -C
5 alkyl)
2, -S-(C!-C
5 alkyl)-S(O)-(Cι-C
5 alkyl), -SO
2-(Cι-C
5 alkyl), -SO
2-(C
2-C
5 alkenyl), -SO2-(C3-C5 cycloalkyl), -SO2-(C3-C5 cycloalkenyl), -Sθ2-(Cχ-C5 hydroxyalkyl), -Sθ2-(Cχ-C5 fluoroalkyl), -SO
2-(Cι-C
5)-phenyl, -SO
2-NH
2,
-SO2-NH-(C!-C
5 alkyl), -SO
2-NH-CH
2-C(O)OH, -Sθ2-NH-CH2-C(O)(O-Cχ-C
5 alkyl), -SO2-NH-(C!-C5 alkyl)-C(O)OH, -SO
2-NH-(Cι-C
5 alkyl)-C(O)(O-C
1-C
5 alkyl), -SO
2-NHC(O)-(C
3-C
6 cycloalkyl), -SO
2-NH-C(O)-(C
1-C
5 alkyl), -SO
2-N-(C
1-C
5 alkyl)
2, -SO
2-(Cι-C
5 alkyl)-O-(Cι-C
5 alkyl), -SO2-(C!-C
5 alkyl)-C(O)-(C
1-C
5 alkyl), -SO
2-(C
1-C
5 alkyl) NH
2,
-SO
2-(C
1-C
5 alkyl)- H-(Cι-C5 alkyl), -SO
2-(C
1-C
5 alkyl)-N-(Cι-C
5 alkyl)
2, -SO
2-(C
1-C
5 alkyl)-C(O)-NH
2, -SO
2-(C
1-C
5 alkyl)-C(O)-NH-(Cχ-C5 alkyl), -SO
2-(C!-C
5 alkyl)-C(O)-N-(Cι-C
5 alkyl)
2, -SO2-(C!-C5 alkyl)-NH-S02-(Cχ-C5 alkyl), -SO2-(Ci-C5 alkyl)-N-pynolidin-2-one, -Sθ2-(Cχ-C5 alkyl)-N-pynolidine, -SO2-(C i -C5 alkyl)-( 1 -methylρynolidin-2-one-3-yl), -SO
2-(C
1-C
5 alkyl)-C(O)-O-(Cι-C
5 alkyl), -SO
2-(C
1-C
5 alkyl)-C(O)-OH, -Sθ2-(Cχ-C5 alkyl)-5-teteazolyl, -SO
2-(C
1-C
5 alkyl)-SO
2-(C
1-C
5 alkyl), -SO
2-(Cι-C
5 alkyl)-SO
2-NH
2, -SO
2-(Cι-C
5 alkyl)-S02-NH-(Cχ-C5 alkyl), -SO
2-(Cι -C5 alkyl)-SO
2-N-(Cι-C5 alkyl)
2, -Sθ2-(Cχ-C5 alkyl)-SO
2-(C
1-C
5 alkyl), -SO
2-(Ci-C
5 alkyl)-P(O)-(O-Cι-C
5 alkyl)
2 , -S0
2-(C
1-C
5 alkyl), -SO
2-(C
2-C
5 alkenyl), ' -SO
2-(C
3-C
5 cycloalkyl), -SO2-(C3-C5 cycloalkenyl), -Sθ2-(Cχ-C5 hydroxyalkyl), -Sθ2-(Cι-C5 fluoroalkyl), -SO
2-(Cι-C
5)-phenyl,
alkyl)
2, -S(O)-NH
2, -S(0)-NH-(Cι-C5 alkyl),
-S(O)-(Cι-C5 alkyl)-S(O)-NH-(C!-C5 alkyl),
-S(O)-(Cχ-C5 alkyl)-SO2-N-(C1-C5 alkyl)2,
-S(O)-(Cχ-C5 alkyl)-S(O)-N-(C1-C5 alkyl)2,
-S(O)-(C!-C5 alkyl)-SO2-(C1-C5 alkyl),
-S(O)-(Cχ-C5 alkyl)-S(O)-(C1-C5 alkyl),
-S(O)-(Cι-C5 alkyl)-P(O)-(O-C1-C5 alkyl)2 ,
-S(O)-N=CHN(Cι-C5 alkyl) 2,
-NHC(S)NH2>
-NHC(S)NH-(Cχ-C5 alkyl),
-NHC(S)N-(Cχ-C5 alkyl)2,
-NHC(S)NH-(C2-C5 alkenyl),
-NHC(S)NH-(C3-C5 cycloalkyl),
-NHC(S)NH-(C3-C5 cycloalkenyl),
-NHC(S)NH-(Cχ-C5 fluoroalkyl),
-NHC(S)NH-Cχ-C5 hydroxyalkyl,
-NHC(S)NH-(Cι-C5 fluoroalkyl)
-NHC(S)NH-phenyl, -NHC(S)NH-(Cι-C5 alkyl)-C(O)-OH,
-NHC(S)NH-(Cι-C5 alkyl)-C(O)-(Cι -C5 alkyl),
-NHC(S)NH-(C!-C5 alky -CtOMO-Cx-Cs alkyl),
-NHC(S)NH-(C!-C5 alkyl)-NH2,
-NHC(S)NH-(C!-C5 alkyl)-NH-(Cχ-C5 alkyl),
-NHC(S)NH-(C!-C5 alkyl)-N-(Cχ-C5 alkyl)2,
-NHC(S)NH-(C1-C5 alkyl)-C(O)-NH2>
-NHC(S)NH-(Cχ-C5 alkyl)-C(O)-NH-(C1-C5 alkyl),
-NHC(S)NH-(Cχ-C5 alkyl)-C(O)-N-(C1-C5 alkyl)2,
-NHC(S)NH-(C1-C5 alkyl)-NH-SO2-(C1-C5 alkyl),
-NHC(S)NH-(Cι-C5 alkyl)-NH-S(O)-(Cι-C5 alkyl), -NHC(S)NH-(Cι-C5 alkyl)-N-pynolidin-2-one, -NHC(S)NH-(C1-C5 alkyl)-N-pynolidine, -NHC(S)NH-(Cχ-C5 alkyl)-(l-methylpynolidin-2-one- 3-yl), -NHC(S)NH-(Cι-C5 alkyl)-5-tetrazolyl, -NHC(S)NH-(Cι-C5 alkyl)-SO2-(C1-C5 alkyl), -NHC(S)NH-(Cι-C5 alkyl)-SO2-NH2) -NHC(S)NH-(Cι-C5 alkyl)-SO2-NH-(C1-C5 alkyl), -NHC(S)NH-(Cχ-C5 alkyl)-SO2-N-(C1-C5 alkyl)2, ' -NHC(S)NH-(Cι-C5 alkyl)-S(O)-(Cχ-C5 alkyl), -NHC(S)NH-(C!-C5 alkyl)-S(O)-NH2, -NHC(S)NH-(Cχ-C5 alkyl)-S(O)-NH-(Cι-C5 alkyl), -NHC(S)NH-(C!-C5 alkyl)-S(O)-N-(C1-C5 alkyl)2, -NHC(S)NH-(C!-C5 alkyl)-P(O)-(O-C!-C5 alkyl)2 , -NHC(O)NH2, -NHC(O)NH-(Cχ-C5 alkyl), -NHC(O)N-(C!-C5 alkyl)2, -NHC(O)NH-(C2-C5 alkenyl), -NHC(O)NH-(C3-C5 cycloalkyl), -NHC(O)NH-(C3-C5 cycloalkenyl), -NHC(O)NH-(Cχ-C5 hydroxyalkyl), -NHC(O)NH-(Cχ-C5 fluoroalkyl), -NHC(O)NH-phenyl, -NHC(O)NH-(Cχ-C5 alkyl)-NH2, -NHC(O)NH-(Cχ-C5 alkyl)-NH-(C1-C5 alkyl), -NHC(O)NH-(Cχ-C5 alkyl)-N-(Cl-C5 alkyl)2? -NHC(O)NH-(Cl-C5 alkyl)-O-(Cχ-C5 alkyl),
-NHC(O)NH-(Cι-C5 alkyl)-NH2, -NHC(O)NH-(C!-C5 alkyl)-NH-(C!-C5 alkyl), -NHC(O)NH-(Cχ-C5 alkyl)-N-(C1-C5 alkyl)2, -NHC(O)NH-(Cχ-C5 alkyl)-C(O)-NH2? -NHC(O)NH-(Cχ-C5 alkyl)-C(O)-NH-(C1-C5 alkyl), -NHC(O)NH-(Cχ-C5 alkyl)-C(O)-N-(Cι-C5 alkyl)2, -NHC(O)NH-(Cχ-C5 alkyl)-C(O)-(C1-C5 alkyl), -NHC(O)NH-(Cχ-C5 alkyl)-NH-SO2-(C1-C5 alkyl), -NHC(O)NH-(C1-C5 alkyl)-N-pynolidin-2-one, -NHC(O)NH-(C χ-C5 alkyl)-N-pynolidine, -NHC(O)NH-(Cχ-C5 alkyl)- (l-methylpynolidin-2-one-3-yl), -NHC(O)NH-(Cχ-C5 alkyl)-C(O)-OH,
-NHC(O)NH-(C!-C5 alkyl)-C(O)-O-(Cι-C5 alkyl),
-NHC(O)NH-(C1-C5 alkyl)-5-tetrazolyl,
-NHC(O)NH-(C!-C5 alkyl)-SO2-(C1-C5 alkyl),
-NHC(O)NH-(Cχ-C5 alkyl)-SO2-NH2,
-NHC(O)NH-(Cι -C5 alkyl)-SO2-NH-(C1-C5 alkyl),
-NHC(O)NH-(C!-C5 alkyl)-SO2-N-(C!-C5 alkyl)2,
-NHC(O)NH-(C1-C5 alkyl)-P(O)-O-(Cι-C5 alkyl)2 ,
-NH2,
-NH-(Cχ-C5 alkyl),
-NH-CH2-C(O)OH,
-N-(Ci-C5 alkyl)2,
-NH-C(O)-NH2,
-NH-C(O)-NH-(C!-C5 alkyl),
-NH-C(O)-N-(C!-C5 alkyl)2,
-NH-C(O)-(C!-C5 alkyl),
-NH-SO2-(C!-C5 alkyl),
-NH-S(0)-(Cι-C"5 alkyl),
-N(CH3)(OCH3),
-N(OH)(CH3),
-N-pynolidin-2-one,
-N-pynolidine,
-( 1 -methylpynolidin-2-one-3-yl),
-CO2H,
-CO2Me,
-CO2Et,
-C(O)CH2S(O)Me,
-C(O)CH2S(O)Et,
-C(O)CH2S(O)2Me,
-C(O)CH2S(O)2Et,
-C(O)CH2CH2S(O)Me,
-C(O)CH2CH2S(O)Et,
-C(O)CH2CH2S(O)2Me,
-C(O)CH2CH2S(O)2Et,
-C(O)CH(Me)CH2CO2H,
-C(O)CH(Me)CH2CO2Me,
-C(O)CH(Me)CH2CO2Et,
-C(O)CH(Me)CH2CO2iPr,
-C(O)CH(Me)CH2CO2tBu,
-C(O)CH(Me)CH(Me)CO2H,
-C(O)CH(Me)CH(Me)CO2Me,
-C(O)CH(Me)CH(Me)CO2Et,
-C(O)CH(Me)CH(Me)CO2iPr,
-C(O)CH(Me)CH(Me)CO2tBu,
-C(O)CH(Me)C(Me) 2CO2H, -C(O)CH(Me)C(Me) 2CO2Me, -C(O)CH(Me)C(Me) 2CO2Et, -C(O)CH(Me)C(Me) 2CO2iPr, -C(O)CH(Me)C(Me) 2CO2tBu, -C(O)CH(Me)CH(Et)CO2H, -C(O)CH(Me)CH(Et)CO2Me, -C(O)CH(Me)CH(Et)CO2Et, -C(O)CH(Me)CH(Et)CO2iPr, -C(O)CH(Me)CH(Et)CO2tBu, -C(O)C(O)OH, -C(O)C(O)NH2, -C(O)C(O)NHMe, -C(O)C(O)NMe2,
-C(O)NH2,
-C(O)NMe2,
-C(O)NH-CH2-C(O)OH,
-C(O)NH-CH2-C(O)OMe,
-C(O)NH-CH2-C(O)OEt,
-C(O)NH-CH2-C(O)OiPr,
-C(O)NH-CH2-C(O)OtBu,
-C(O)NH-CH(Me)-C(O)OH,
-C(O)NH-CH(Me)-C(O)OMe,
-C(O)NH-CH(Me)-C(O)OEt,
-C(O)NH-CH(Me)-C(O)iPr,
-C(O)NH-CH(Me)-C(O)tBu,
-C(O)NH-CH(Et)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OMe,
-C(O)NH-C(Me)2-C(O)OEt,
-C(O)NH-C(Me)2-C(O)iPr, -C(O)NH-C(Me)2-C(O)tBu, -C(O)NH-CMe(Et)-C(O)OH, -C(O)NH-CH(F)-C(O)OH, -C(O)NH-CH(CF3)-C(O)OH, -C(O)NH-CH(OH)-C(O)OH, -C(O)NH-CH(cyclopropyl)-C(O)OH, -C(O)NH-C(Me)2-C(O)OH, -C(O)NH-C(Me)2-C(O)OH, -C(O)NH-CF(Me)-C(O)OH, -C(O)NH-C(Me)(CF3)-C(O)OH, -C(O)NH-C(Me)(OH)-C(O)OH, -C(O)NH-C(Me)(cyclopropyl)CO2H
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH2-C(O)OMe,
-C(O)NMe-CH2-C(O)OEt,
-C(O)NMe-CH2-C(O)OiPr,
-C(O)NMe-CH2-C(O)tBu,
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH(Me)-C(O)OH,
-C(O)NMe-CH(F)-C(O)OH,
-C(O)NMe-CH(CF3)-C(O)OH,
-C(O)NMe-CH(OH)-C(O)OH,
-C(O)NMe-CH(cyclopropyl)-C(O)OH,
-C(O)NMe-C(Me)2-C(O)OH,
-C(O)NMe-CF(Me)-C(O)OH,
-C(O)NMe-C(Me)(CF3)-C(O)OH,
-C(O)NMe-C(Me)(OH)-C(O)OH,
-C(O)NMe-C(Me)(cyclopropyl)-C(O)OH,
-C(O)NHS(O)Me,
-C(O)NHSO2Me,
-C(O)-NH-5-tetrazolyl,
-C(O)NHS(O)Me,
-C(O)NHS(O)Et,
-C(O)NHSO2Me,
-C(O)NHSO2Et,
-C(O)NHS(O)iPr, -C(O)NHSO2iPr,
-C(O)NHS(O)tBu, -C(O)NHSO2tBu,
-C(O)NHCH2S(O)Me,
-C(O)NHCH2S(O)Et,
-C(O)NHCH2SO2Me,
-C(O)NHCH2SO2Et,
-C(O)NHCH2CH2S(O)Me,
-C(O)NHCH2CH2S(O)Et,
-C(O)NHCH2CH2SO2Me,
-C(O)NHCH2CH2SO2Et,
-C(O)N(Me)S<O)Me, -C(O)N(Me)S 2Me,
-C(O)-N(Me)-5-tetrazolyl, -C(O)N(Me)S(O)Me, -C(O)N(Me)S(O)Et, -C(O)N(Me)SO2ϊvle,
-C(O)N(Me)SO2Et,
-C(O)N(Me)S(O)iPr, -C(O)N(Me))SO2iPr, -C(O)N(Mc))S(O)tBu, -C(O)N(Me)SO2tBu,
-C(O)N(Me)CH2S(O)Me, -C(O)N(Me)CH2S<O)Et,
-C(O)N(Me)CH2SO2Me, -C(O)N(Me)CH2SO2Et, -C(O)N(Me)CH2CH2S(O)Me, -C(O)N(Me)CH2CH2S(O)Et, -C(O)N(Me)CH2CH2SO2Me, -C(O)N(Me)CH2CH2SO2Et, -CH2CO2H, -CH2-5-tetrazolyl, -CH2CO2Me, -CH2CO2Et, -CH2NHS(O)Me, -CH2NHS(O)Et, -CH2NHSO2Me, -CH2NHSO2Et, -CH2NHS(O)iPr, -CH2NHSO2iPr, -CH2NHS(O)tBu, -CH2NHS02tBu, -CH2NHCH2CH2SO2CH3 , -CH2NH(CH2CO2H), -CH2N(C(O)Me)(CH2CO2H), -CH2-N-pynolidin-2-one, -CH2-(l-methylpynolidin-2-one-3-yl), -CH2S(O)Me, -CH2S(O)Et, -CH2S(O)2Me, -CH2S(O)2Et, -CH2S(O)iPr,
-CH2S(O)2iPr, -CH2S(O)tBu, -CH2S(O)2tBu, -CH2CO2H, CH2C(O)NH2, -CH2C(O)NMe2, -CH2C(O)NHMe, -CH2C(O)-N-pynolidine, -CH2S(O)2Me, CH2S(O)Me, -CH(OH) CO2H, -CH(OH)C(O)NH2, -CH(OH)C(O)NHMe, -CH(OH)C(O)NMe2, -CH(OH)C(O)NEt2, -CH2CH2CO2H, -CH2CH2CO2Me, -CH2CH2CO2Et, -CH2CH2C(O)NH2, -CH2CH2C(O)NHMe, -CH2CH2C(O)NMe2, -CH2CH2-5-tetrazolyl, -CH2CH2S(O)2Me, -CH2CH2S(O)Me, -CH2CH2S(O)2Et, ~CH2CH2S(O) Et, -CH2CH2S(O)iPr, -CH2CH2S O)2iPr, -CH2CH2S(O)tBu, -CH2-CH2S<O)2tBu,
-CH2CH2S(O)NH2, -CH2CH2S(O)NHMe,
-CH2CH2S(O)NMe2,
-CH2CH2S(O)2NH2,
-CH2CH2S(O)2NHMe
-CH2CH2S(O)2NMe2,
-CH2CH2CH2S(O)Me,
-CH2CH2CH2S(O)Et,
-CH2CH2CH2S(O)2Me,
-CH2CH2CH2S(O)2Et,
-CH(Me)CH2C(O)OH, -C(Me)2CH2C(O)OH, -5-tetrazolyl,
- 1 ,3 ,4-oxadiazolin-2-one-5-yl, -imidazolidine-2,4-dione-5-yl, -isoxazol-3-ol-yl, or -l,3,4-oxadiazolin-2-thione-5-yl; provided that RP is substituted at either the 2, 5, or 6 position of the phenyl ring. The compounds of the invention with vitamin receptor modulating (VDRM) activity are represented by formula (IC) or a pharmaceutically acceptable salt or a prodrug derivative thereof:
wherein R and R' are independently C1-C5 alkyl, C -C5 fluoroalkyl, or together R and R' form a substituted or unsubstituted, saturated or unsaturated carbocyclic ring having from 3 to 8 carbon atoms; RP, RB
4, RT
3 and RB are independently selected from the group consisting of hydrogen, halo, C1-C5 alkyl, Cχ-C5 fluoroalkyl, -O-C1-C5 alkyl, -S-Cχ-C
5 alkyl, -O-Cχ-
C5 fluoroalkyl, -CN, -NO2, acetyl, -S-C1-C5 fluoroalkyl, C2-C5 alkenyl, C3-C5 cycloalkyl, and C3-C5 cycloalkenyl; RP3 and RB7 are independently selected from hydrogen, halo, C1-C5 alkyl, C1-C5 fluoroalkyl, -O-C -C5 alkyl, -S-Cχ-C5 alkyl, -O-Cχ-C5 fluoroalkyl, -CN, -NO2, acetyl, -
S-Cχ-C5 fluoroalkyl, C2-C5 alkenyl, C3-C5 cycloalkyl, or C3-C5 cycloalkenyl; (Lpj.), (Lp2), and (LBT) are divalent linking groups independently selected from the group consisting of a bond
(CH
2)
m— CH =CH- and
where m is 0, 1, or 2, and each R40 is independently hydrogen, C1-C5 alkyl, or C1-C5 fluoroalkyl; Zp is branched C3-C5 alkyl, 3-mefhyl-3-hydroxypentyl, 3-methyl-3-hydroxypentenyl, 3 -methyl-3 -hydroxypentynyl, 3 -ethyl-3 -hydroxypentyl, 3-ethyl-3-hydroxypentenyl, 3-ethyl-3-hydroxypentynyl, 3-ethyl-3-hydroxy-4-methylpentyl,
3-ethyl-3-hydroxy-4-methylpentenyl,
3-ethyl-3-hydroxy-4-methylpentynyl,
3 -propyl-3 -hydroxypentyl,
3 -propyl-3 -hydroxypentenyl,
3-propyl-3-hydroxypentynyl,
1 -hydroxy-2-methyl- 1 -(methylethyl)propyl,
2-methyl-3-hydroxy-4-dimethylpentyl,
2-methyl-3-hydroxy-3-ethylpentyl,
2-ethyl-3-hydroxy-3-ethylpentyl,
2-ethyl-3-hydroxy-4-dimethylpentyl,
3-methyl-3-hydroxy-4,4-dimethylpentyl,
3-methyl-3-hydroxy-4,4-dimethylpentenyl,
3-methyl-3-hydroxy-4,4-dimethylpentyl,
3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
3-ethyl-3-hydroxy-4,4-dimethylpentenyl,
3-ethyl-3-hydroxy-4,4-dimethylpentynyl,
1 -hydroxycycyclopentenyl,
1 -hydroxycyclohexenyl,
1 -hydroxycycloheptenyl,
1 -hydroxycyclooctenyl,
1 -hydroxycyclopropyl ,
1 -hydroxycyclobutyl,
1 -hydroxycyclopentyl,
1 -hydroxycyclohexyl,
2-oxocyclohexyloxy,
2-oxocyclohexylmethyl,
3-methyl-2-oxocyclohexyloxy,
3-methyl-2-oxocyclohexylmethyl,
3,3-dimethyl-2-oxocyclohexyloxy,
3 ,3 -dimethyl-2-oxocyclohexylmethyl,
2-hydroxycyclohexyloxy,
2-hydroxycyclohexylmethyl,
3 -methyl-2-hydroxycyclohexyloxy, 3-methyl-2-hydroxycyclohexylmethyl, 3,3-dimethyl-2-hydroxycyclohexyloxy, 3,3-dimethyl-2-hydroxycyclohexylmethyl , 1-hydroxycycloheptyl, or 1-hydroxycyclooctyl; provided, however, that when Zp is 3 -methyl-3 -hydroxypentyl , 3 -methyl-3 -hydroxypentenyl , 3 -methyl-3 -hydroxypentynyl, 3 -ethyl-3 -hydroxypentyl, 3-ethyl-3-hydroxypentenyl, 3-ethyl-3-hydroxypentynyl, 3-ethyl-3-hydroxy-4-methylpentyl, 3-ethyl-3-hydroxy-4-methylpentenyl, 3-ethyl-3-hydroxy-4-methylpentynyl, 3-propyl-3-hydroxypentyl, 3 -propyl-3 -hydroxypentenyl, 3-propyl-3-hydroxypentynyl, 3-methyl-3-hydroxy-4,4-dimethylpentyl, 3-methyl-3-hydroxy-4,4-dimethylpentenyl, 3-methyl-3-hydroxy-4,4-dimethylpentyl, 3-ethyl-3-hydroxy-4,4-dimethylpentynyl, 3-ethyl-3-hydroxy-4,4-dimethylpentenyl, 3-ethyl-3-hydroxy-4,4-dimethylpentynyl, 2-methyl-3-hydroxy-4-dimethylpentyl, 2-methyl-3-hydroxy-3-ethylpentyl, 2-ethyl-3-hydroxy-3-ethylp'entyl, 2-ethyl-3-hydroxy-4-dirnethylpentyl, or 1 -hydroxy-2-methyl- 1 -(methylethyl)propyl ; then (Lpi) and Ln) combine as a bond;

i o o 9 o 9 O O O O o o O o o o O o O O O o o O o o o O Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω to Ω Ω
U Ωl U Ωi U Ωl U Ωl U Ωl U Ωl U Ωi U Ωl U Ωl U Ωl U Ωl U Ωl U Ωl U Ωl U Ωi U Ωl U Ωi U Ωl U Ωi U Ωl U Ωl U Ωl U Ωl U Ωl U Ωl U Ωl U Ωι
-O-(Cι-C5 alkyl)-SO2-N-(C1-C5 alkyl)2; -O-(C!-C5 alkyl)-SO2-(C1-C5 alkyl), -O-(Cχ-C5 alkyl)-S(O)-(C1-C5 alkyl,)
-O-(C -C5 alkyl)-S(O)-NH2, -O-(Cι-C5 alkyl)-S(O)-NH-(C -C5 alkyl), -O-(Cχ-C5 alkyl)-S(O)-N-(C1-C5 alkyl)2, -O-(C!-C5 alkyl)-S(O)-(C1-C5 alkyl), -O-(C!-C5 alkyl)-P(O)-(O-C1-C5 alkyl)2 , -O-(Cχ-C5 alkyl)-5-tetrazolyl,
-O-CH2-CO2H, -O-CH2-5-tetrazolyl, -O-(Cι-C5 alkyl),
-O-C(O)-NH2, -O-C(O)-N-(CH3)2, -O-C(S)-N-(CH3)2, -O-C(O)-O-(C!-C5 alkyl),
-O-(5-tetrazolyl), -O-SO2-(Cι -C5 alkyl,)
-O-SO2-NH2)
-O-SO2-NH-(C!-C5 alkyl),
-O-SO2-N-(Cι-C5 alkyl)2,
-O-S(O)-(C!-C5 alkyl,)
-O-S(O)-NH2,
-O-S(O)-NH-(Cι -C5 alkyl),
-O-S(O)-N-(Cι-C5 alkyl)2,
-S-(C!-C5 alkyl),
-S-(C2-C5 alkenyl),
CO 00 00 00 00 00 GO 00 GO GO GO 00 GO 00 00 GO GO en 00 00 GO oo GO GO GO GO 00 00 Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω Ω
Ui Ul Ul Ul Ul Ul Ul Uι Ul Ul Uι Ul Ul Ul Ul Ui Ul Ul Ul Ul Ul Ul Ul Ul Ul Ul ui Ul
-S-(Cι-C5 alkyl)-S(O)-N-(Cι-C5 alkyl)2, -S-(C!-C5 alkyl)-S(O)-(C1-C5 alkyl), -SO2-(C!-C5 alkyl), -Sθ2-(C2-C5 alkenyl), -SO2-(C3-C5 cycloalkyl), -Sθ2-(C3-C5 cycloalkenyl), -SO2-(Cι-C5 hydroxyalkyl), -Sθ2-(Cχ-C5 fluoroalkyl), -SO2-(C1-C5)-phenyl, -SO2-NH2?
-SO
2-NH-(C
1-C
5 alkyl), -SO
2-NH-CH
2-C(O)OH, -SO2-NH-CH
2-C(O)(O-C
1-C
5 alkyl), -SO
2-NH-(C
1-C
5 alkyl)-C(O)OH, -SO2-NH-(C!-C
5 alkyl)-C(O)(O-Cι-C5 alkyl), -SO
2-NHC(O)-(C
3-C
6 cycloalkyl), -SO
2-NH-C(O)-(Cι-C
5 alkyl), -SO
2-N-(C
1-C
5 alkyl)2, -SO
2-(C
1-C
5 alkyl)-O-(C
1-C
5 alkyl), -SO
2-(Cι-C
5 alkyl)-C(O)-(C
1-C
5 alkyl), -SO
2-(C -C
5 alkyl) NH
2? -SO -(C!-C
5 alkyl)-NH-(C!-C5 alkyl), -SO
2-(Cι-C
5 alkyl)-N-(Cι-C
5 alkyl)
2, -SO
2-(Cι-C
5 alkyl)-C(O)-NH
2,
alkyl)-C(O)-NH-(Cι-C
5 alkyl), -SO
2-(Cι-C
5 alkyl)-C(O)-N-(C
1-C
5 alkyl)
2, -SO
2-(Cι-C
5 alkyl)-NH-SO
2-(C
1-C
5 alkyl), -SO
2-(Cι-C5 alkyl)-N-pynolidin-2-one,
-NHC(S)NH2, -NHC(S)NH-(C!-C5 alkyl), -NHC(S)N-(Cι-C5 alkyl)2, -NHC(S)NH-(C2-C5 alkenyl), -NHC(S)NH-(C3-C5 cycloalkyl), -NHC(S)NH-(C -C5 cycloalkenyl), -NHC(S)NH-(Cι-C5 fluoroalkyl), -NHC(S)NH-C!-C5 hydroxyalkyl, -NHC(S)NH-(Cχ-C5 fluoroalkyl)
-NHC(S)NH-phenyl, -NHC(S)NH-(C -C5 alkyl)-C(O)-OH,
-NHC(S)NH-(C!-C5 alkyl)-O-(C -C
5 alkyl), -NHC(S)NH-(C!-C5 alkyl)-C(O)-(C
1-C
5 alkyl), -NHC(S)NH-(Cι-C
5
alkyl), -NHC(S)NH-(Cι-C5 alkyl)-NH
2, -NHC(S)NH-(C!-C5 alkyl)-NH-(C -C
5 alkyl), -NHC(S)NH-(Cχ-C
5 alkyl)-N-(Cι-C
5 alkyl)
2, -NHC(S)NH-(Cι-C5 alkyl)-C(O)-NH
2, -NHC(S)NH-(Cχ-C5 alkyl)-C(O)-NH-(C
1-C
5 alkyl), -NHC(S)NH-(C!-C5 alkyl)-C(O)-N-(C -C
5 alkyl)
2, -NHC(S)NH-(Cχ-C5 alkyl)-NH-S02-(Cχ-C5 alkyl), -NHC(S)NH-(Cι-C
5 alkyl)-NH-S(O)-(Cχ-C
5 alkyl), -NHC(S)NH-(C
1-C
5 alkyl)-N-pyrrolidin-2-one, -NHC(S)NH-(C χ-C
5 alkyl)-N-pynolidine, -NHC(S)NH-(C
1-C
5 alkyl)-(l-methylpynolidin-2-one- 3-yl), -NHC(S)NH-(C
1-C
5 alkyl)-5-tetrazolyl,
-NHC(S)NH-(Cι-C5 alkyl)-SO2-(C1-C5 alkyl),
-NHC(S)NH-(Cι-C5 alkyl)-SO2-NH2; -NHC(S)NH-(Cι-C5 alkyl)-SO2-NH-(Cι-C5 alkyl), -NHC(S)NH-(C1-C5 alkyl)-SO2-N-(C1-C5 alkyl)2, -NHC(S)NH-(Cι-C5 alkyl)-S(O)-(Cχ-C5 alkyl), -NHC(S)NH-(C1-C5 alkyl)-S(O)-NH2; -NHC(S)NH-(Cι-C5 alkyl)-S(O)-NH-(C1-C5 alkyl), -NHC(S)NH-(Cι-C5 alkyl)-S(O)-N-(Cι-C5 alkyl)2, -NHC(S)NH-(Cχ-C5 alkyl)-P(O)-(O-C -C5 alkyl)2 , -NHC(O)NH2, -NHC(O)NH-(C!-C5 alkyl), -NHC(O)N-(C!-C5 alkyl)2, -NHC(O)NH-(C2-C5 alkenyl), -NHC(O)NH-(C3-C5 cycloalkyl), -NHC(O)NH-(C3-C5 cycloalkenyl), -NHC(O)NH-(C1-C5 hydroxyalkyl), -NHC(O)NH-(C -C5 fluoroalkyl), -NHC(O)NH-phenyl, -NHC(O)NH-(C!-C5 alkyl)-NH2? -NHC(O)NH-(C!-C5 alkyl)-NH-(C1-C5 alkyl), -NHC(O)NH-(C1-C5 alkyl)-N-(Cl-C5 alkyl)2, -NHC(O)NH-(Cl-C5 alkyl)-O-(C1-C5 alkyl), -NHC(O)NH-(C!-C5 alkyl)-NH2, -NHC(O)NH-(C!-C5 alkyl)-NH-(C1-C5 alkyl), -NHC(O)NH-(C!-C5 alkyl)-N-(Cι-C5 alkyl)2) -NHC(O)NH-(Cι-C5 alkyl)-C(O)-NH2; -NHC(O)NH-(C!-C5 alkyl)-C(O)-NH-(Cχ-C5 alkyl), -NHC(O)NH-(C1-C5 alkyl)-C(O)-N-(C1-C5 alkyl)2, -NHC(O)NH-(C!-C5 alkyl)-C(O)-(Cχ-C5 alkyl),
-NHC(O)NH-(Cχ-C5 alkyl)-NH-SO2-(C1-C5 alkyl),
-NHC(O)NH-(C!-C5 alkyl)-N-pynolidin-2-one,
-NHC(O)NH-(C1-C5 alkyl)-N-pynolidine,
-NHC(O)NH-(C!-C5 alkyl)- ( 1 -methylpynolidin-2-one-3-yl), -NHC(O)NH-(C!-C5 alkyl)-C(O)-OH,
-NHC(O)NH-(C1-C5 alkyl)-C(O)-O-(Cι-C5 alkyl),
-NHC(O)NH-(C1-C5 alkyl)-5-tetrazolyl,
-NHC(O)NH-(Cχ-C5 alkyl)-SO2-(C1-C5 alkyl),
-NHC(O)NH-(C -C5 alkyl)-SO2-NH2,
-NHC(O)NH-(Cχ-C5 alkyl)-SO2-NH-(Cι-C5 alkyl),
-NHC(O)NH-(C!-C5 alkyl)-SO2-N-(Cι-C5 alkyl)2,
-NHC(O)NH-(C!-C5 alkyl)-P(O)-O-(Cι-C5 alkyl)2 ,
-NH2,
-NH-(C!-C5 alkyl),
-NH-CH2-C(O)OH,
-N-(Cι-C5 alkyl)2,
-NH-C(O)-NH2,
-NH-C(O)-NH-(Cι-C5 alkyl),
-NH-C(O)-N-(Cι -C5 al yl)2,
-NH-C(O)-(C!-C5 alkyl),
-NH-SO2-(C1-C5 alkyl),
-NH-S(O)-(C1-C5 alkyl),
-N(CH3)(OCH3),
-N(OH)(CH3),
-N-pynolidin-2-one,
-N-pynolidine,
-( 1 -methylpynolidin-2-one-3-yl),
-CO2H, -CO2Me, -CO2Et,
-C(O)CH2S(O)Me, -C(O)CH2S(O)Et, -C(O)CH2S(O)2Me, -C(O)CH2S(O)2Et, -C(O)CH2CH2S(O)Me, -C(O)CH2CH2S(O)Et, -C(O)CH2CH2S(O)2Me, -C(O)CH2CH2S(O)2Et, -C(O)CH(Me)CH2CO2H, -C(O)CH(Me)CH2CO2Me, -C(O)CH(Me)CH2CO2Et, -C(O)CH(Me)CH2CO2iPr, -C(O)CH(Me)CH2CO2tBu, -C(O)CH(Me)CH(Me)CO2H, -C(O)CH(Me)CH(Me)CO2Me, -C(O)CH(Me)CH(Me)CO2Et, -C(O)CH(Me)CH(Me)CO2iPr, -C(O)CH(Me)CH(Me)CO2tBu, -C(O)CH(Me)C(Me) 2CO2H, -C(O)CH(Me)C(Me) 2CO2Me, -C(O)CH(Me)C(Me) 2CO2Et, -C(O)CH(Me)C(Me) 2CO2iPr, -C(O)CH(Me)C(Me) 2CO2tBu, -C(O)CH(Me)CH(Et)CO2H, -C(O)CH(Me)CH(Et)CO2Me,
-C(O)CH(Me)CH(Et)CO2Et, -C(O)CH(Me)CH(Et)CO2iPr, -C(O)CH(Me)CH(Et)CO2tBu,
-C(O)C(O)OH, -C(O)C(O)NH2,
-C(O)C(O)NHMe, -C(O)C(O)NMe2,
-C(O)NH2,
-C(O)NMe2,
-C(O)NH-CH2-C(O)OH,
-C(O)NH-CH2-C(O)OMe,
-C(O)NH-CH2-C(O)OEt,
-C(O)NH-CH2-C(O)OiPr,
-C(O)NH-CH2-C(O)OtBu,
-C(O)NH-CH(Me)-C(O)OH,
-C(O)NH-CH(Me)-C(O)OMe,
-C(O)NH-CH(Me)-C(O)OEt,
-C(O)NH-CH(Me)-C(O)iPr,
-C(O)NH-CH(Me)-C(O)tBu,
-C(O)NH-CH(Et)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OMe,
-C(O)NH-C(Me)2-C(O)OEt,
-C(O)NH-C(Me)2-C(O)iPr,
-C(O)NH-C(Me)2-C(O)tBu,
-C(O)NH-CMe(Et)-C(O)OH,
-C(O)NH-CH(F)-C(O)OH,
-C(O)NH-CH(CF3)-C(O)OH,
-C(O)NH-CH(OH)-C(O)OH,
-C(O)NH-CH(cyclopropyl)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-CF(Me)-C(O)OH,
-C(O)NH-C(Me)(CF3)-C(O)OH,
-C(O)NH-C(Me)(OH)-C(O)OH,
-C(O)NH-C(Me)(cyclopropyl)CO2H
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH2-C(O)OMe,
-C(O)NMe-CH2-C(O)OEt,
-C(O)NMe-CH2-C(O)OiPr,
-C(O)NMe-CH2-C(O)tBu,
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH(Me)-C(O)OH,
-C(O)NMe-CH(F)-C(O)OH,
-C(O)NMe-CH(CF3)-C(O)OH,
-C(O)NMe-CH(OH)-C(O)OH,
-C(O)NMe-CH(cyclopropyl)-C(O)OH,
-C(O)NMe-C(Me)2-C(O)OH,
-C(O)NMe-CF(Me)-C(O)OH,
-C(O)NMe-C(Me)(CF3)-C(O)OH,
-C(O)NMe-C(Me)(OH)-C(O)OH,
-C(O)NMe-C(Me)(cyclopropyl)-C(O)OH,
-C(O)NHS(O)Me,
-C(O)NHSO2Me,
-C(O)-NH-5-tetrazolyl, -C(O)NHS(O)Me, -C(O)NHS(O)Et, -C(O)NHSO2Me,
-C(O)NHSO2Et,
-C(O)NHS(O)iPr, -C(O)NHSO2iPr,
-C(O)NHS(O)tBu,
-C(O)NHSO2tBu,
-C(O)NHCH2S(O)Me,
-C(O)NHCH2S(O)Et,
-C(O)NHCH2SO2Me,
-C(O)NHCH2SO2Et,
-C(O)NHCH2CH2S(9)Me, ,
-C(O)NHCH2CH2S(O)Et,
-C(O)NHCH2CH2SO2Me,
-C(O)NHCH2CH2SO2Et,
-C(O)N(Me)S(O)Me, -C(O)N(Me)SO2Me,
-C(O)-N(Me)-5-tetrazolyl, -C(O)N(Me)S(O)Me, -C(O)N(Me)S(O)Et, -C(O)N(Me)SO2Me,
-C(O)N(Me)SO2Et,
-C(O)N(Me)S(O)iPr,
-C(O)N(Me))SO2iPr,
-C(O)N(Me))S(O)tBu, -C(O)N(Me)SO2tBu,
-C(O)N(Me)CH2S(O)Me,
-C(O)N(Me)CH2S(O)Et,
-C(O)N(Me)CH2SO2Me,
-C(O)N(Me)CH2SO2Et,
-C(O)N(Me)CH2CH2S(O)Me,
-C(O)N(Me)CH2CH2S(O)Et,
-C(O)N(Me)CH2CH2SO2Me,
-C(O)N(Me)CH2CH2SO2Et,
-CH2CO2H, -CH2-5-tetrazolyl, -CH2CO2Me, -CH2CO2Et, -CH2NHS(O)Me, -CH2NHS(O)Et, -CH2NHSO2Me, -CH2NHSO2Et, -CH2NHS(O)iPr, I
-CH2NHSO2iPr,
-CH2NHS(O)tBu,
-CH2NHSO2tBu,
-CH2NHCH2CH2SO2CH3 ,
-CH2NH(CH2CO2H),
-CH2N(C(O)Me)(CH2CO2H),
-CH2-N-pynolidin-2-one,
-CH2-( 1 -methylpynolidin-2-one-3 -yl) ,
-CH2S(O)Me,
-CH2S(O)Et,
-CH2S(O)2Me,
-CH2S(O)2Et,
-CH2S(O)iPr,
-CH2S(O)2iPr,
-CH2S(O)tBu,
-CH2S(O)2tBu,
-CH2CO2H, CH2C(O)NH2,
-CH2C(O)NMe2,
-CH2C(O)NHMe,
-CH2C(O)-N-ρynolidine,
-CH2S(O)2Me, CH2S(O)Me,
-CH(OH) CO2H,
-CH(OH)C(O)NH2,
-CH(OH)C(O)NHMe, -CH(OH)C(O)NMe2,
-CH(OH)C(O)NEt2,
-CH2CH2CO2H,
-CH2CH2CO2Me,
-CH2CH2CO2Et,
-CH2CH2C(O)NH2,
-CH2CH2C(O)NHMe,
-CH2CH2C(O)NMe2,
-CH2CH2-5-tetrazolyl,
-CH2CH2S(O)2Me,
-CH2CH2S(O)Me,
-CH2CH2S(O)2Et,
-CH2CH2S(O) Et,
-CH2CH2S(O)iPr,
-CH2CH2S(O)2iPr,
-CH2CH2S(O)tBu,
-CH2CH2S(O)2tBu,
-CH2CH2S(O)NH2,
-CH2CH2S(O)NHMe,
-CH2CH2S(O)NMe2,
-CH2CH2S(O)2NH2,
-CH2CH2S(O)2NHMe
-CH2CH2S(O)2NMe2,
-CH2CH2CH2S(O)Me, -CH2CH2CH S(O)Et, -CH2CH2CH2S(O)2Me, -CH2CH2CH2S(O)2Et, CH(Me)CH2C(O)OH, -C(Me)2CH2C(O)OH, -5-tetrazolyl,
- 1 ,3 ,4-oxadiazolin-2-one-5-yl, -imidazolidine-2,4-dione-5-yl, -isoxazol-3-ol-yl, or -l,3,4-oxadiazolin-2-thione-5-yl; provided that RP is substituted at either the 2, 5, or 6 position of the phenyl ring.
Prefened embodiments of the Invention:
The compound of Formula IA having as prefened substituents;
R and R' are independently methy or ethyl; RP and RT3 are independently, hydrogen or methyl; RP3 and RB are independently hydrogen, methyl, ethyl, -O-methyl, or cyclopropyl; (Lpi) and (LTB) divalent linking groups are both bonds; (LP2) is a bond, -CH2-, -CH(OH)-, or -C(Me)OH-; Zp is 1,1-dimethylethyl; 1-hydroxycyclopentyl, 1-hydroxycyclohexyl,
3-ethyl-3-hydroxypentyl, 3-exhyl-3-hydroxypentenyl, 3-ethyl-3-hydroxypentynyl; ZTB IS -CO2H, -CO2Me, -CO2Et, -C(O)CH2S(O)Me, -C(O)CH2S(O)Et, -C(O)CH2S(O)2Me, -C(O)CH2S(O)2Et, -C(O)CH2CH2S(O)Me, -C(O)CH2CH2S(O)Et, -C(O)CH2CH2S(O)2Me, -C(O)CH2CH2S(O)2Et, -C(O)CH(Me)CH2CO2H, -C(O)CH(Me)CH2CO2Me, -C(O)CH(Me)CH2CO2Et, -C(O)CH(Me)CH2CO2iPr, -C(O)CH(Me)CH2CO2tBu, -C(O)CH(Me)CH(Me)CO2H, -C(O)CH(Me)CH(Me)CO2Me, -C(O)CH(Me)CH(Me)CO2Et, -C(O)CH(Me)CH(Me)CO2iPr,
-C(O)CH(Me)CH(Me)CO2tBu, -C(O)CH(Me)C(Me) 2CO2H, -C(O)CH(Me)C(Me) 2CO2Me, -C(O)CH(Me)C(Me) 2CO2Et, -C(O)CH(Me)C(Me) 2CO2iPr, -C(O)CH(Me)C(Me) 2CO2tBu, -C(O)CH(Me)CH(Et)CO2H, -C(O)CH(Me)CH(Et)CO2Me, -C(O)CH(Me)CH(Et)CO2Et, -C(O)CH(Me)CH(Et)CO2iPr, -C(O)CH(Me)CH(Et)CO2tBu, -C(O)C(O)OH, -C(O)C(O)NH2, -C(O)C(O)NHMe, -C(O)C(O)NMe2, -C(O)NH2, -C(O)NMe2, -C(O)NH-CH2-C(O)OH, -C(O)NH-CH2-C(O)OMe, -C(O)NH-CH2-C(O)OEt, -C(O)NH-CH2-C(O)OiPr, -C(O)NH-CH2-C(O)OtBu, -C(O)NH-CH(Me)-C(O)OH, -C(O)NH-CH(Me)-C(O)OMe, -C(O)NH-CH(Me)-C(O)OEt, -C(O)NH-CH(Me)-C(O)iPr, -C(O)NH-CH(Me)-C(O)tBu, -C(O)NH-CH(Et)-C(O)OH, -C(O)NH-C(Me)2-C(O)OH, -C(O)NH-C(Me)2-C(O)OMe,
-C(O)NH-C(Me)2-C(O)OEt, -C(O)NH-C(Me)2-C(O)iPr, -C(O)NH-C(Me)2-C(O)tBu, -C(O)NH-CMe(Et)-C(O)OH, -C(O)NH-CH(F)-C(O)OH, -C(O)NH-CH(CF3)-C(O)OH, -C(O)NH-CH(OH)-C(O)OH, -C(O)NH-CH(cyclopropyl)-C(O)OH, -C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH, -C(O)NH-CF(Me)-C(O)OH, -C(O)NH-C(Me)(CF3)-C(O)OH, -C(O)NH-C(Me)(OH)-C(O)OH, -C(O)NH-C(Me)(cyclopropyl)CO2H
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH2-C(O)OMe,
-C(O)NMe-CH2-C(O)OEt,
-C(O)NMe-CH2-C(O)OiPr,
-C(O)NMe-CH2-C(O)tBu,
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH(Me)-C(O)OH,
-C(O)NMe-CH(F)-C(O)OH,
-C(O)NMe-CH(CF3)-C(O)OH,
-C(O)NMe-CH(OH)-C(O)OH,
-C(O)NMe-CH(cyclopropyl)-C(O)OH,
-C(O)NMe-C(Me)2-C(O)OH,
-C(O)NMe-CF(Me)-C(O)OH,
-C(O)NMe-C(Me)(CF3)-C(O)OH,
-C(O)NMe-C(Me)(OH)-C(O)OH,
-C(O)NMe-C(Me)(cyclopropyl)-C(O)OH,
-C(O)NHS(O)Me,
-C(O)NHSO2Me,
-C(O)-NH-5-tetrazolyl, -C(O)NHS(O)Me, -C(O)NHS(O)Et, -C(O)NHSO2Me,
-C(O)NHSO2Et,
-C(O)NHS(O)iPr, -C(O)NHSO2iPr, -C(O)NHS(O)tBu, -C(O)NHSO2tBu,
-C(O)NHCH2S(O)Me,
-C(O)NHCH2S(O)Et,
-C(O)NHCH2SO2Me,
-C(O)NHCH2SO2Et,
-C(O)NHCH2CH2S(O)Me,
-C(O)NHCH2CH2S(O)Et,
-C(O)NHCH2CH2SO2Me,
-C(O)NHCH2CH2SO2Et,
-C(O)N(Me)S(O)Me,
-C(O)N(Me)SO2Me,
-C(O)-N(Me)-5-tetrazolyl, -C(O)N(Me)S(O)Me, -C(O)N(Me)S(O)Et, -C(O)N(Me)SO2Me,
-C(O)N(Me)SO2Et,
-C(O)N(Me)S(O)iPr, -C(O)N(Me))SO2iPr,
-C(O)N(Me))S(O)tBu, -C(O)N(Me)SO2tBu,
-C(O)N(Me)CH2S(O)Me,
-C(O)N(Me)CH2S(O)Et, -C(O)N(Me)CH2SO2Me, -C(O)N(Me)CH2SO2Et, -C(O)N(Me)CH2CH2S(O)Me, -C(O)N(Me)CH2CH2S(O)Et, -C(O)N(Me)CH2CH2SO2Me, -C(O)N(Me)CH2CH2SO2Et, -CH2CO2H, -CH2-5-tetrazolyl, -CH2CO2Me, -CH2CO2Et, -CH2NHS(O)Me, -CH2NHS(O)Et, -CH2NHSO Me, -CH2NHSO2Et, -CH2NHS(O)iPr, -CH2NHSO2iPr, -CH2NHS(O)tBu, -CH2NHSO2tBu, -CH2NHCH2CH2SO2CH3 , -CH2NH(CH2CO2H), -CH2N(C(O)Me)(CH2CO2H), -CH2-N-pynolidin-2-one, -CH2-( 1 -methylpyrrolidin-2-one-3 -yl) , -CH2S(O)Me, -CH2S(O)Et, -CH2S(O)2Me, -CH2S(O)2Et,
-CH2S(O)iPr,
-CH2S(O)2iPr,
-CH2S(O)tBu,
-CH2S(O)2tBu,
-CH2CO2H, CH2C(O)NH2,
-CH2C(O)NMe2,
-CH2C(O)NHMe,
-CH2C(O)-N-pyιτolidine,
-CH2S(O)2Me, CH2S(O)Me,
-CH(OH) CO2H,
-CH(OH)C(O)NH2,
-CH(OH)C(O)NHMe,
-CH(OH)C(O)NMe2,
-CH(OH)C(O)NEt2,
-CH2CH2CO2H,
-CH2CH2CO2Me,
-CH2CH2CO2Et,
-CH2CH2C(O)NH2,
-CH2CH2C(O)NHMe,
-CH2CH2C(O)NMe2,
-CH2CH2-5-tetrazolyl,
-CH2CH2S(O)2Me,
-CH2CH2S(O)Me,
-CH2CH2S(O)2Et,
-CH2CH2S(O) Et,
-CH2CH2S(O)iPr,
-CH2CH2S(O)2iPr,
-CH2CH2S(O)tBu,
-CH2CH2S(O)2tBu, -CH2CH2S(O)NH2, -CH2CH2S(O)NHMe, -CH2CH2S(O)NMe2, -CH2CH2S(O)2NH2, -CH2CH2S(O)2NHMe -CH2CH2S(O)2NMe2, -CH2CH2CH2S(O)Me, -CH2CH2CH2S(O)Et, -CH2CH2CH2S(O)2Me, or -CH2CH2CH2S(O)2Et.
The compound of formula IB having as prefened substituents;; R and R' are independently mefhy or ethyl; RP, RB, RB , and RT are independently, hydrogen or methyl; RP3 and RB are independently hydrogen, methyl, ethyl, -O-methyl, or cyclopropyl; (LP and (LET) divalent linking groups are both bonds; (LP2) is a bond, -CH2-, -CH(OH)-, or -C(Me)OH-; Zp is 1,1-dimethylethyl; 1-hydroxycyclopentyl, 1-hydroxycyclohexyl,
3 -ethyl-3 -hydroxypentyl, 3 -ethyl-3 -hydroxypentenyl, 3 -ethyl-3 -hydroxypentynyl ; ZBτ is -CO2H, -CO2Me, -CO2Et, -C(O)CH2S(O)Me, -C(O)CH2S(O)Et, -C(O)CH2S(O)2Me, -C(O)CH2S(O)2Et, -C(O)CH2CH2S(O)Me,
-C(O)CH2CH2S(O)Et, -C(O)CH2CH2S(O)2Me, -C(O)CH2CH2S(O)2Et, -C(O)CH(Me)CH2CO2H, -C(O)CH(Me)CH2CO2Me, -C(O)CH(Me)CH2CO2Et, -C(O)CH(Me)CH2CO2iPr, -C(O)CH(Me)CH2CO2tBu, -C(O)CH(Me)CH(Me)CO2H, -C(O)CH(Me)CH(Me)CO2Me, -C(O)CH(Me)CH(Me)CO2Et, -C(O)CH(Me)CH(Me)CO2iPr, -C(O)CH(Me)CH(Me)CO2tBu, -C(O)CH(Me)C(Me) 2CO2H, -C(O)CH(Me)C(Me) 2CO2Me, -C(O)CH(Me)C(Me) 2CO2Et, -C(O)CH(Me)C(Me) 2CO2iPr, -C(O)CH(Me)C(Me) 2CO2tBu, -C(O)CH(Me)CH(Et)CO2H, -C(O)CH(Me)CH(Et)CO2Me, -C(O)CH(Me)CH(Et)CO2Et, -C(O)CH(Me)CH(Et)CO2iPr, -C(O)CH(Me)CH(Et)CO2tBu, -C(O)C(O)OH, -C(O)C(O)NH2, -C(O)C(O)NHMe, -C(O)C(O)NMe2, -C(O)NH2,
-C(O)NMe2,
-C(O)NH-CH2-C(O)OH,
-C(O)NH-CH2-C(O)OMe,
-C(O)NH-CH2-C(O)OEt,
-C(O)NH-CH2-C(O)OiPr,
-C(O)NH-CH2-C(O)OtBu,
-C(O)NH-CH(Me)~C<O)OH,
-C(O)NH-CH(Me)-C(O)OMe,
-C(O)NH-CH(Me)-C(O)OEt,
-C(O)NH-CH(Me)-C(O)iPr,
-C(O)NH-CH(Me)-C(O)tBu,
-C(O)NH-CH(Et)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OMe,
-C(O)NH-C(Me)2-C(O)OEt,
-C(O)NH-C(Me)2-C(O)iPr,
-C(O)NH-C(Me)2-C(O)tBu,
-C(O)NH-CMe(Et)-C(O)OH,
-C(O)NH-CH(F)-C(O)OH,
-C(O)NH-CH(CF3)-C(O)OH,
-C(O)NH-CH(OH)-C(O)OH,
-C(O)NH-CH(cyclopropyl)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-CF(Me)-C(O)OH,
-C(O)NH-C(Me)(CF3)-C(O)OH,
-C(O)NH-C(Me)(OH)-C(O)OH,
-C(O)NH-C(Me)(cyclopropyl)CO2H
-C(O)NMe-CH2-C(O)OH, -C(O)NMe-CH2-C(O)OMe, -C(O)NMe-CH2-C(O)OEt, -C(O)NMe-CH2-C(O)OiPr,
-C(O)NMe-CH2-C(O)tBu,
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH(Me)-C(O)OH,
-C(O)NMe-CH(F)-C(O)OH,
-C(O)NMe-CH(CF3)-C(O)OH,
-C(O)NMe-CH(OH)-C(O)OH,
-C(O)NMe-CH(cyclopropyl)-C(O)OH,
-C(O)NMe-C(Me)2-C(O)OH,
-C(O)NMe-CF(Me)-C(O)OH,
-C(O)NMe-C(Me)(CF3)-C(O)OH,
-C(O)NMe-C(Me)(OH)-C(O)OH,
-C(O)NMe-C(Me)(cyclopropyl)-C(O)OH,
-C(O)NHS(O)Me,
-C(O)NHSO2Me,
-C(O)-NH-5-tetrazolyl, -C(O)NHS(O)Me, -C(O)NHS(O)Et, -C(O)NHSO2Me,
-C(O)NHSO2Et,
-C(O)NHS(O)iPr, -C(O)NHSO2iPr,
-C(O)NHS(O)tBu, -C(O)NHSO2tBu,
-C(O)NHCH2S(O)Me,
-C(O)NHCH2S(O)Et,
-C(O)NHCH2SO2Me,
-C(O)NHCH2SO2Et,
-C(O)NHCH2CH2S(O)Me,
-C(O)NHCH2CH2S(O)Et,
-C(O)NHCH2CH2SO2Me,
-C(O)NHCH2CH2SO2Et,
-C(O)N(Me)S(O)Me, -C(O)N(Me)SO2Me,
-C(O)-N(Me)-5-tetrazolyl, -C(O)N(Me)S(O)Me, -C(O)N(Me)S(O)Et, -C(O)N(Me)SO2Me,
-C(O)N(Me)SO2Et,
-C(O)N(Me)S(O)iPr, -C(O)N(Me))SO2iPr,
-C(O)N(Me))S(O)tBu, -C(O)N(Me)SO2tBu,
-C(O)N(Me)CH2S(O)Me,
-C(O)N(Me)CH2S(O)Et,
-C(O)N(Me)CH2SO2Me,
-C(O)N(Me)CH2SO2Et,
-C(O)N(Me)CH2CH2S(O)Me,
-C(O)N(Me)CH2CH2S(O)Et,
-C(O)N(Me)CH2CH2SO2Me,
-C(O)N(Me)CH2CH2SO2Et,
-CH2CO2H,
-CH2-5-tetrazolyl,
-CH2CO2Me,
-CH2CO2Et, -CH2NHS(O)Me, -CH2NHS(O)Et, -CH2NHSO2Me, -CH2NHSO2Et, -CH2NHS(O)iPr, -CH2NHSO2iPr, -CH2NHS(O)tBu, -CH2NHSO2tBu, -CH2NHCH2CH2SO2CH3, -CH2NH(CH2CO2H), -CH2N(C(O)Me)(CH2CO2H), -CH2-N-pynolidin-2-one, -CH2-(l-methylpyrrolidin-2-one-3-yl), -CH2S(O)Me, -CH2S(O)Et, -CH2S(O)2Me, -CH2S(O)2Et, -CH2S(O)iPr, -CH2S(O)2iPr, -CH2S(O)tBu, -CH2S(O)2tBu, -CH2CO2H, CH2C(O)NH2, -CH2C(O)NMe2, -CH2C(O)NHMe, -CH2C(O)-N-pynolidine, -CH2S(O)2Me, CH2S(O)Me, -CH(OH) CO2H,
-CH(OH)C(O)NH2,
-CH(OH)C(O)NHMe,
-CH(OH)C(O)NMe2,
-CH(OH)C(O)NEt2,
-CH2CH2CO2H,
-CH2CH2CO2Me,
-CH2CH2CO2Et,
-CH2CH2C(O)NH2,
-CH2CH2C(O)NHMe,
-CH2CH2C(O)NMe2,
-CH2CH2-5-tetrazolyl,
-CH2CH2S(O)2Me,
-CH2CH2S(O)Me,
-CH2CH2S(O)2Et,
-CH2CH2S(O) Et,
-CH2CH2S(O)iPr,
-CH2CH2S(O)2iPr,
-CH2CH2S(O)tBu,
-CH2CH2S(O)2tBu,
-CH2CH2S(O)NH2,
-CH2CH2S(O)NHMe,
-CH2CH2S(O)NMe2,
-CH2CH2S(O)2NH2,
-CH2CH2S(O)2NHMe
-CH2CH2S(O)2NMe2,
-CH2CH2CH2S(O)Me,
-CH2CH2CH2S(O)Et, -CH2CH2CH2S(O)2Me, or -CH2CH2CH2S(O)2Et. The compound of formula IC having as prefened substituents; R and R' are independently methy or ethyl; RP, RB, RB , and RT3 are independently, hydrogen or methyl; RP3 and RB are independently hydrogen, methyl, ethyl, -O-methyl, or cyclopropyl; (Lpι) and (LBT) divalent linking groups are both bonds; (LP2) is a bond, -CH2-, -CH(OH)-, or -C(Me)OH-; Zp is 1,1-dimethylethyl; 1-hydroxycyclopentyl, 1-hydroxycyclohexyl,
3-ethyl-3-hydroxypentyl, 3-ethyl-3-hydroxypentenyl, 3-ethyl-3-hydroxypentynyl; ZBτ is -CO2H, -CO2Me, -CO2Et, -C(O)CH2S(O)Me, -C(O)CH2S(O)Et, -C(O)CH2S(O)2Me, -C(O)CH2S(O)2Et, -C(O)CH2CH2S(O)Me, -C(O)CH2CH2S(O)Et, -C(O)CH2CH2S(O)2Me, -C(O)CH2CH2S(O)2Et, -C(O)CH(Me)CH2CO2H, -C(O)CH(Me)CH2CO2Me, -C(O)CH(Me)CH2CO2Et, -C(O)CH(Me)CH2CO2iPr, -C(O)CH(Me)CH2CO2tBu,
-C(O)CH(Me)CH(Me)CO2H, -C(O)CH(Me)CH(Me)CO2Me, -C(O)CH(Me)CH(Me)CO2Et, -C(O)CH(Me)CH(Me)CO2iPr, -C(O)CH(Me)CH(Me)CO2tBu, -C(O)CH(Me)C(Me) 2CO2H, -C(O)CH(Me)C(Me) 2CO2Me, -C(O)CH(Me)C(Me) 2CO2Et, -C(O)CH(Me)C(Me) 2CO2iPr, -C(O)CH(Me)C(Me) 2CO2tBu, -C(O)CH(Me)CH(Et)CO2H, -C(O)CH(Me)CH(Et)CO2Me, -C(O)CH(Me)CH(Et)CO2Et, -C(O)CH(Me)CH(Et)CO2iPr, -C(O)CH(Me)CH(Et)CO2tBu, -C(O)C(O)OH, -C(O)C(O)NH2, -C(O)C(O)NHMe, -C(O)C(O)NMe2, -C(O)NH2, -C(O)NMe2, -C(O)NH-CH2-C(O)OH, -C(O)NH-CH2-C(O)OMe, -C(O)NH-CH2-C(O)OEt, -C(O)NH-CH2-C(O)OiPr, -C(O)NH-CH2-C(O)OtBu, -C(O)NH-CH(Me)-C(O)OH, -C(O)NH-CH(Me)-C(O)OMe, -C(O)NH-CH(Me)-C(O)OEt,
-C(O)NH-CH(Me)-C(O)iPr,
-C(O)NH-CH(Me)-C(O)tBu,
-C(O)NH-CH(Et)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OMe,
-C(O)NH-C(Me)2-C(O)OEt,
-C(O)NH-C(Me)2-C(O)iPr,
-C(O)NH-C(Me)2-C(O)tBu,
-C(O)NH-CMe(Et)-C(O)OH,
-C(O)NH-CH(F)-C(O)OH,
-C(O)NH-CH(CF3)-C(O)OH,
-C(O)NH-CH(OH)-C(O)OH,
-C(O)NH-CH(cyclopropyl)-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-C(Me)2-C(O)OH,
-C(O)NH-CF(Me)-C(O)OH,
-C(O)NH-C(Me)(CF3)-C(O)OH,
-C(O)NH-C(Me)(OH)-C(O)OH,
-C(O)NH-C(Me)(cyclopropyl)CO2H
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH2-C(O)OMe,
-C(O)NMe-CH2-C(O)OEt,
-C(O)NMe-CH2-C(O)OiPr,
-C(O)NMe-CH2-C(O)tBu,
-C(O)NMe-CH2-C(O)OH,
-C(O)NMe-CH(Me)-C(O)OH,
-C(O)NMe-CH(F)-C(O)OH,
-C(O)NMe-CH(CF3)-C(O)OH,
-C(O)NMe-CH(OH)-C(O)OH,
-C(O)NMe-CH(cyclopropyl)-C(O)OH,
-C(O)NMe-C(Me)2-C(O)OH,
-C(O)NMe-CF(Me)-C(O)OH,
-C(O)NMe-C(Me)(CF3)-C(O)OH,
-C(O)NMe-C(Me)(OH)-C(O)OH,
-C(O)NMe-C(Me)(cyclopropyl)-C(O)OH,
-C(O)NHS(O)Me,
-C(O)NHSO2Me,
-C(O)-NH-5-tetrazolyl, -C(O)NHS(O)Me, -C(O)NHS(O)Et, -C(O)NHSO2Me,
-C(O)NHSO2Et,
-C(O)NHS(O)iPr, -C(O)NHSO2iPr,
-C(O)NHS(O)tBu, -C(O)NHSO2tBu,
-C(O)NHCH2S(O)Me,
-C(O)NHCH2S(O)Et,
-C(O)NHCH2SO2Me,
-C(O)NHCH2SO2Et,
-C(O)NHCH2CH2S(O)Me,
-C(O)NHCH2CH2S(O)Et,
-C(O)NHCH2CH2SO2Me,
-C(O)NHCH CH2SO2Et,
-C(O)N(Me)S(O)Me, -C(O)N(Me)SO2Me,
-C(O)-N(Me)-5-tetrazolyl, -C(O)N(Me)S(O)Me, -C(O)N(Me)S(O)Et, -C(O)N(Me)SO2Me,
-C(O)N(Me)SO2Et,
-C(O)N(Me)S(O)iPr, -C(O)N(Me))SO2iPr,
-C(O)N(Me))S(O)tBu, -C(O)N(Me)SO2tBu,
-C(O)N(Me)CH2S(O)Me,
-C(O)N(Me)CH2S(O)Et,
-C(O)N(Me)CH2SO2Me,
-C(O)N(Me)CH2SO2Et,
-C(O)N(Me)CH2CH2S(O)Me,
-C(O)N(Me)CH2CH2S(O)Et,
-C(O)N(Me)CH2CH2SO2Me,
-C(O)N(Me)CH2CH2SO2Et,
-CH2CO2H,
-CH2-5-tetrazolyl,
-CH2CO Me,
-CH2CO2Et,
-CH2NHS(O)Me,
-CH2NHS(O)Et,
-CH2NHSO2Me,
-CH2NHSO2Et,
-CH2NHS(O)iPr,
-CH2NHSO2iPr, '
-CH2NHS(O)tBu,
-CH2NHSO2tBu,
-CH2NHCH2CH2SO2CH3,
-CH2NH(CH2CO2H),
-CH2N(C(O)Me)(CH2CO2H),
-CH2-N-pynolidin-2-one,
-CH2-(l-methylpynolidin-2-one-3-yl), -CH2S(O)Me, -CH2S(O)Et, -CH2S(O)2Me, -CH2S(O)2Et, -CH2S(O)iPr, -CH2S(O)2iPr, -CH2S(O)tBu, -CH2S(O)2tBu, -CH2CO2H, CH2C(O)NH2, -CH2C(O)NMe2, -CH2C(O)NHMe, -CH2C(O)-N-pynolidine, -CH2S(O)2Me, CH2S(O)Me, -CH(OH) CO2H, -CH(OH)C(O)NH2, -CH(OH)C(O)NHMe, -CH(OH)C(O)NMe2, -CH(OH)C(O)NEt2, -CH2CH2CO2H, -CH2CH2CO2Me, -CH2CH2CO2Et, -CH2CH2C(O)NH2, -CH2CH2C(O)NHMe, -CH2CH2C(O)NMe2, -CH2CH2-5-tetrazolyl, -CH2CH2S(O)2Me, -CH2CH2S(O)Me,
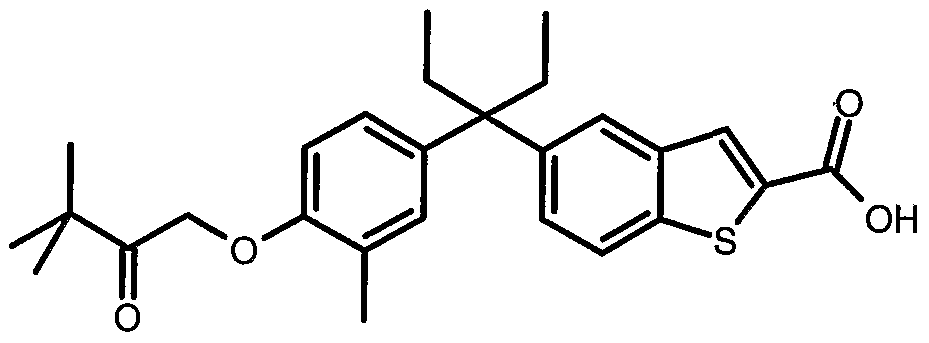
-CH2CH2S(O)2Et, -CH2CH2S(O) Et, -CH2CH2S(O)iPr, -CH2CH2S(O)2iPr, -CH2CH2S(O)tBu, -CH2CH2S(O)2tBu, -CH2CH2S(O)NH2, -CH2CH2S(O)NHMe, -CH2CH2S(O)NMe2, -CH2CH2S(O)2NH2, -CH2CH2S(O)2NHMe -CH2CH2S(O)2NMe2, -CH2CH2CH2S(O)Me, -CH2CH2CH2S(O)Et, -CH2CH2CH2S(O)2Me, or -CH2CH2CH2S(O)2Et. Particularly prefened compounds of the invention and salts and prodrag derivatives are represented by formulae CI to C22 as follows:
CI)
C2)
C4)
C5)
C6)
C7)
C8)
CIO)
Cll)
C12)
C13)
C15)
C16)
C17)
C18)
C20)
C21)
C22)
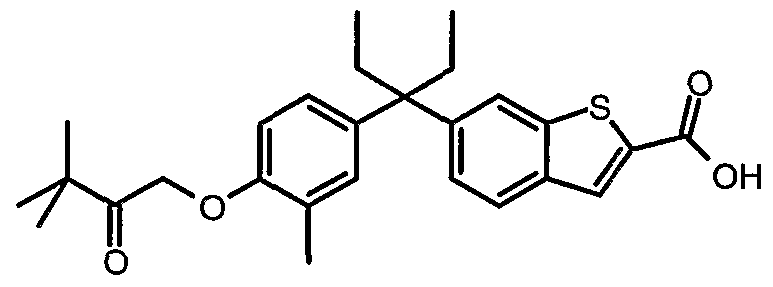
Particularly prefened is the compound represented by the stractural formula AAor a pharmaceutically acceptable salt or prodrag thereof:
EXAMPLES General Experimental Conditions: The starting material/intermediate is the compound from the immediate preceding experimental unless otherwise indicated. All reactions are performed under nitrogen/argon atmosphere, in a stined reaction vessel, and at room temperature unless indicated otherwise. Unless otherwise indicated, the organic layer is MgSO4/Na2SO4 dried is defined as stining the solution with a dessicant for 5-15 m and filtering off the dessicant to give an anhydrous filtrate. For analogous multi-step reaction procedures, the yield is given either for the ultimate step or overall multi-steps as indicated. Solutions are "concentrated" at a range of 25-75 °C with reduced pressure, in-vacuo - 25-75 °C; 0.05 to 1 mm
Unless otherwise indicated, "the residue is chromatographed" is defined as silica gel chromatography of residue with moderate nitrogen pressure (flash chromatography) or a medium pressure chromatography systems using a silica gel to crade product ratio of ~10- 100.
For HPLC, the conditions listed are for the analytical trace only. For Preparative HPLC, the eluent is similar to analytical HPLC eluent. Thin layer chromatography is performed with silica gel plates with UV and/or appropriate staining solution. NMR spectra are obtained with either 300 or 400 mHz spectrometer. NMR data is listed to denote spectrum is consistent with assigned stracture. "NMR" notation without data denotes spectrum is consistent with assigned stracture.
HRMS - high resolution mass spectrum ES-MS - electrospray mass spectrum
Abbreviations: Aq - aqueous d - day eq - equivalent h - hour m - minute satd - saturated disp - dispersion quant - quantitative rt for retention time (both small caps to minimize confusion with RT) RT - room temperature
Chemical Definitions: BF3-OEt2 - boron trifluoride etherate BnBr - benzyl bromide CH2C12-dichloromethane CH3CN - acetonitrile CO-carbon monoxide CsF - cesium fluoride DMAP - 4-(dimethylamino)pyridine DMF - N,N-dimethylformamide DMSO - dimethylsulfoxide DPPB - l,4-bis(diphenylphosphino)butane DPPF - dichloro[l,l'-bis(diphenylphosphino)fenocene EDCI - 3-Ethyl-l-[3-(dimethylamino)propyl]carbodiimide hydrochloride Et3N - triethylamine EtMgBr- ethyl magnesium bromide EtOAc - ethyl acetate EtOH - ethanol
H2 - hydrogen pressure
H2NCH2CO2Me - methyl glycinate
Hept - heptane
Hex - hexanes
HN(OMe)Me - N-methyl-O-methyl hydroxylamine
HNMe2 - dimethyl amine
HATU - O-(7-azabenzotriazol-l-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate
HO AT - 7-aza-l-hydroxybenzotriazole
HOBT - 1-hydroxybenzotriazole
K2CO3 - potassium carbonate
KI - potassium iodine
KOH - potassium hydroxide
LAH - lithium aluminum hydride
LiHMDS - lithium hexamethyldisilazide
Lindlar catalyst - Pd-CaCO3-PbO
LiOH - lithium hydroxide mCPB A - meta-chloroperbenzoic acid
Mel - methyl iodide
MeOH - methanol
NaBH4 - sodium borohydride
MgSO4- magnesium sulfate
NaH - sodium hydride
NaHCO3-sodium bicarbonate
Nal - sodium iodide
Na2SO4- sodium sulfate
NH4C1- ammonium chloride
NMO - 4-methylmorpholine N-oxide
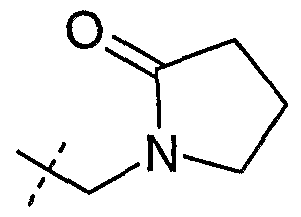
NMP - N-methylpynolidin-2-one
Na-S-R3 - sodium alkylmercaptide
PBr3 - phosphorus tribromide
Pd(DPPF) - palladium dichloro[l,l'-bis(diphenylphosphino)fenocene
Pd(OAc)2 - palladium (II) acetate
Pd(TPP)4 - palladium tetrakistriphenylphosphine
Pd-C - palladium on carbon
Pd-C/H2 - palladium on carbon with hydrogen pressure pTS A - para-toluenesulfonic acid
Pyr - pyridine
Red-Al - sodium bis(2-methoxyethoxy)aluminum hydride
R2MgBr - alkyl magnesium bromide
R3MgBr - alkyl magnesium bromide
R5MgBr - alkyl magnesium bromide
R3S(O)2Cl - alkylsulfonyl chloride
R2S(O)2NH2 - alkylsulfonamide
TBSC1- tert-butyldimethylsilyl chloride tBuC(O)CH2Br - 1-bromopinacolone
Tf2O - triflic anhydride
TFA - trifluoroacetic acid
THF - tetrahydrofuran
Ti(OiPr)4 - titanium tetraisopropoxide
TMS-acetylene - trimethylsilyl acetylene
TPAP - tetrapropylammonium perruthenate
Zn(OTf)2 - zinc trifluoromethane sulfonate
General Procedures Scheme I.
A a) BF3 b) H+/alkanol
10. 11. 12. Here, R'-CH2 means R' minus a methylene group
Scheme III.
Scheme IN.
Scheme V.
Scheme I. The known 2-fluoro-4-iodo-3-trimethylsilanyl-benzaldehyde is reacted with a mercaptoacetic acid ester to form benzo[b]thiophene ester 1. Benzo[b]thiophene 1. is reacted with an alkyl Grignard reagent (0.9 to 1.3 equivalents) at low temperature in diethylether or THF to undergo magnesium exchange with the iodine of 1. that is subsequently treated with a disubstituted ketone to produce the tert-carbinol 6. tert-
Carbinol 6. is reacted with an ortho-substituted phenol in the presence of a Lewis acid, e.g., borontrifluoride etherate from 0 °C. to room temperature to give the diarylmethane 7.
The free hydroxyl of diarlymethane 7. is alkylated with an alpha-halo ketone in a polar aprotic solvent such as acetone or acetonitrile, with or without catalytic potassium iodide from room temperature to the reflux temperature of the mixture. The alkylated intermediate preceeding is saponified with lithium, potassium, or sodium hydroxide and alkanol from room temperature to the reflux temperature of the mixture to produce the acid 8. The ketone fragment of acid 8. is reduced with lithium, potassium, or sodium borohydride or cyanoborohydride (lto 4 equivalents) in alkanol to the carbinol 9.
Alternately, benzo[b]thiophene 1. can be used as follows: 1. is reacted with an excess of alkyl Grignard reagent (3-6 equivalents) in THF or ether at higher temperature, e.g., room temperature to not only undergo magnesium exchange with the iodine of 1. but also to produce the tert-carbinol fragment of 2. The mixture is subsequently treated with excess carbon dioxide to produce the acid 2. Reaction of acid 2. with an ortho-substituted phenol in the presence of a Lewis acid (0.01 to 5 equivalents), e.g., boron trifluraoride etherate from 0 °C. to room temperature gives a diarylmethane acid 3 that is esterified with acid and alkanol to the ester 3. The free hydroxyl of ester 3. is alkylated with an alpha-halo ketone in polar aprotic solvent to give an intermediate which is subsequently saponified with lithium, potassium, or sodium hydroxide in an alkanol (room temperature to relfux temperature) to give the acid 4. Acid 4. is reduced to the carbinol 5. with lithium, potassium, or sodium borohydride or cyanoborohydride (1 to 4 equivalents) in alkanol (0 ° C. to room temperature).
Scheme II. Methyl, p-fluorobenzoate is reacted with excess alkyl Grignard reagent (2 to 5 equivalents) to produce the tert-carinol intermediate, which eliminates water upon workup to give the olefin 10. Ortho metallation with an alkyllithium reagent of olefin 10. in diethylether or THF from 0 °C. to room temperature followed by treatment with DMF produces the aldehyde 11. Aldehyde 11. is reacted with a mercaptoacetic acid ester to give the benzo[b]thiophene 12. The benzo[b]thiophene 12. will alkylate an ortho- substituted phenol in the presence of a Lewis acid, e.g., boron trifluoride etherate (0.01 to 5 equivalents) to give the diarylmethane 13. The free hydroxyl of diarylmethane 13. is alkylated with an alpha-halo ketone in a polar aprotic solvent, e.g., acetone or acetonitrile from room temperature to the reflux temperature of the mixture, and the alkylated intermediate is subsequently saponified with lithium, potassium, or sodium hydroxide in an alkanol (room temperature to the reflux temperature of the mixture) to give the acid 14. The acid 14. is reduced to the carbinol 15. with lithium, potassium, or sodium borohydride or cyanoborohydride (1-4 equivalents) in alkanol from 0 ° C. to room temperature.
Scheme III. Benzo[b]thiophene 16. is prepared by the reaction of a mercaptoacetic acid ester and 2-fluoro-5-methoxy-benzaldehyde in DMF. The benzo[b]thiophene 16. is reacted with excess alkyl Grignard reagent (2 to 5 equivalents) in diethylether or THF (0 ° C. to room temperature) to give the tert-carbinol 17. The tert-carbinol 17. alkylates an ortho-substituted phenol in the presence of a Lewis acid, e.g., boron trifluoride etherate (0.01 to 5 equivalents) to give the diarylmethane 18. The free hydroxyl of diarylmethane 18. is alkylated with an alpha-halo ketone or ester in a polar aprotic solvent, e.g., acetone or acetonitrile from room temperature to the reflux temperature of the mixture to give the methoxy intermediate 19. The intermediate 19. is demethylated with boron tribromide in halocarbon solvent from 0 ° to room temperature to give the free hydroxyl intermediate 20. The intermediate 20. is converted to the triflate 21. with triflic anhydride and base, and the triflate 21. is reacted with carbon monoxide (1-100 psi) in the presence of palladium catalysis (0.01 to 0.10 equivalents) , e.g., palladium acetate and DPPB, and base to give the ester intermediate which is subsequently saponified with lithium, potassium, or sodium hydroxide in alkanol to give the keto acid 22. The keto acid 22. is reduced to the carbinol 23. with lithium, potassium, or sodium borohydride or cyanoborohydride (1-4 equivalents) in alkanol from 0 ° C. to room temperature.
Scheme IN. Intermediate 19 is reacted with an alkyl Grignard reagent (1-5 equivalents) in ether or THF to give the methoxy carbinol 24. Methoxy carbinol 24. is demethylated with sodium mercaptoethylate in a polar aprotic solvent, e.g., DMF from room temperature to 150 ° C. to give the hydroxy carbinol 25. The hydroxy carbinol 25. is converted to the triflate 26. with triflic anhydride and base, and the triflate 26. is reacted with carbon monoxide (1-100 psi) in the presence of palladium catalysis (0.01 to 0.10 equivalents) , e.g., palladium acetate and DPPB, and base to give the ester 27. The ester 27. is saponified with lithium, potassium, or sodium hydroxide in alkanol to give the acid 28.
Scheme N. The free hydroxyls of esters 3., 7., 13., and 18., from Schemes I. II. And III. above are converted to the triflates 29. with triflic anhydride and base. Each of these triflates 29. is couple to TMS-acetylene in the presence of a palladium catalyst, e.g., PdC12(PPh3)2 (0.01 to 10%) in a polar aprotic solvent, e.g., acetonitriel from room
temperature to 150 C. to give the TMS protected acetylenes as intermediates, that are deprotected with fluoride ion in THF to give acetylenes 30. Each of the acetylenes 30. are deprotonated with, e.g., LiHMDS and treated with a ketone to produce the carbinols 31. Each of the carbinols 31. are saponified with lithium, potassium, or sodium hydroxide in alkanol to give the acids 32.
Each of the free acids produced in each of the above schemes (3., A., 5., 8., 9., 14., 15., 22., 23., and 28.) are converted to lower alkanol esters and lower alkyl secondary or tertiary carboxamides using reactions well known to those skilled in the art.
Example 1.
Preparation of 6-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl } -benzo[b]thiophene-2-carboxylic acid.
A. 6-Iodo-benzo[b]thiophene-2- ster
A solution of 2-fluoro-4-iodo-3-trimethylsilanyl-benzaldehyde (14.2 g, 44.1 mmol) (Tetrahedron Letters 1992, p7499-7502) in DMF (50 mL) is treated with mercapto-acetic acid ethyl ester (5.80 mL, 52.9 mmol) and K
2CO
3 (12.2 g, 88.2 mmol). The resulting suspension is stined at 75 °C for 60 min and quenched with water (300 mL). The mixture is extracted with EtOAc (3 x 50 mL,) and the organic layer is dried over Na
2SO
4, concentrated, and purified by silica gel column chromatography (5-20% EtOAc/Hex), to give the title compound as an oil (9.40 g, 64%).
H-NMR (ppm, CDC13), δ: 8.21 (1 H, d, 7=1.8 Hz), 7.98 (1 H, s), 7.67 (1 H, dd, 7=1.8, 8.8 Hz), 7.58 (1 H, d, 7=8.4 Hz), 4.40 (2 H, q, 7=7.0 Hz), 1.42 (3 H, t, 7=7.0 Hz).
B. 6-(l-Ethyl-l-hydroxy-propyl)-benzo[b]thiophene-2-carboxylic acid ethyl ester.
To a stined solution of 6-iodo-benzo[b]thiophene-2-carboxylic acid ethyl ester (2.95 g, 8.88 mmol) in THF (60 mL) at -78 °C is added ethylmagnesium bromide (4.45 mL, 3.0 M). The reaction mixture is stined for 15 min, and 3-pentanone (3.0 mL) is added. The mixture is allowed to warm to RT and stined for 30 min. The reaction is quenched with HCI (15 mL, 1.0 M) and extracted with EtOAc (2 x 50 mL). The organic layer is dried over Na
2SO , concentrated, and purified by silica gel column chromatography to give the title compoimd (0.65 g, 25%). H-NMR (ppm, CDC1
3), d: 8.02 (1 H, s), 7.95 (1 H, s), 7.80 (1 H, d, 7=8.8 Hz), 7.35 (1 H, dd, 7=1.3, 8.4 Hz), 4.40 (2 H, q, 7=7.0 Hz), 1.88 (4 H, q, 7=7.5 Hz), 1.42 (3 H, t, 7=7.0 Hz ), 0.78 (6 H, q, 7=7.5 Hz).
C. 6-[l-Ethyl-l-(4-hydroxy-3-methyl-phenyl)-propyl]-benzo[b]thiophene-2- carboxylic acid ethyl ester.
To a mixture of 6-(l-ethyl-l-hydroxy-propyl)-benzo[b]thiophene-2-carboxylic acid ethyl ester (0.650 g, 2.23 mmol and o-cresol (0.480 g, 4.45 mmol) in CH
2C1
2 (20 mL) at -78 °C is added BF
3-Et
2O (0.316 g, 2.23 mol). After stining forlO min, the reaction mixture is allowed to warm to 0 °C over 30 min. The reaction is quenched with water (15 mL) and extracted with EtOAc (2 x 50 mL). The organic layer is dried over Na
2SO
4, concentrated, and purified by silica gel column chromatography to give the title compound (0.780 g, 92%). MS (ES) m e 383.1 (M+l), 381.1 (M-l).
D . 6- { 1 - [4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-2-carboxylic acid ethyl ester.
A solution of 6-[l-ethyl-l-(4-hydroxy-3-methyl-phenyl)-propyl]- benzo[b]thiophene-2-carboxylic acid ethyl ester (0.780 g, 2.04 mmol) and 1- bromopinacolone (0.731 g, 4.08 mmol) in acetone (30 mL) is treated with K
2CO
3 (0.563 g, 4.08 mmol) and stined at RT for 4 h. The mixture is filtered, and the filtrate is concentrated. The residue is purified using silica gel column cliromatography (10- 15% EtOAc/Hex) to provide the title compound (0.89 g, 91%). MS (ES) m/e 498.2 (M+18).
E. 6- { 1 - [4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-2-carboxylic acid.
A solution of 6-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-2-carboxylic acid ethyl ester (0.89 g, 1.86 mmol) in MeOH (5.0 mL) and THF (5.0 mL) and is treated with NaOH (2.0 M, 10.0 mL). The resulting mixture is stined at RT for 2 h. The mixture is concentrated, acidified with HCI (I N) until pH~3, and extracted with EtOAc (2 x 50 mL). The organic layer is dried over Na2SO4, concentrated, and purified using silica gel column chromatography (50% EtOAc/Hex) to afford the title compound (840 mg, 99%). MS (ES) m/e 451.1(M-1), 470.2 (M+18).
Example 2.
Preparation of 6-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl } -benzo[b]thiophene-2-carboxylic acid.
A solution of 6-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-2-carboxylic acid (102 mg, 0.226 mmol) in THF<5.0 mL) at RT is treated with NaBHi (17 mg, 0.451 mmol). The resulting mixture is stined
for 4 h. The reaction is quenched with HCI (1.0 N, 1.0 mL) and extracted with EtOAc (2 x 20 mL). The organic layer is dried over Na
2SO and concenfrated to afford the title compound (90 mg, 88%). MS (ES) m/e 453.1(M-1), 472.2 (M+18).
Example 3.
Preparation of 6-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-2-carboxylic acid dimethylamide.
6-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-2-carboxylic acid (290 mg, 0.642 mmol) in CH
2C1
2 (5.0 mL) is treated with DMAP (235 mg, 1.93 mmol) and EDC (184 mg, 0.962 mmol). The mixture is stined for 15 min at RT, and dimethylamine hydrochloride (78 mg, 0.962 mmol) is added. The reaction is stined for 18 h and quenched with aqueous NH
4CI (5.0 mL). The organic layer is loaded onto a silica gel column and purified with 50% EtOAc/Hex to afford the title compound (280 mg, 91%). MS (ES) m e 480.2 (M+l).
Example 4.
Preparation of 6-{ l-efhyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl}-benzo[b]thiophene-2-carboxylic acid dimethylamide.
6-{l-[4-(3,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-2-carboxylic acid dimethylamide (235 mg, 0.490 mmol) is reduced by NaBH
4 (37 mg, 0.980 mmol) as in Step Fl 1 to afford the title compound (230 mg, 100%). MS (ES) m/e 482.2 (M+l).
Example 5.
Preparation of [(6-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- ρropyl}-benzo[b]thiophene~2-carbonyl)-aminoj-acetic acid.
6-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-2-carboxylic acid (270 mg, 0.596 mmol) in CH
2C1
2 (5.0 mL) is treated with DMAP (218 mg, 1.79 mmol) and EDC (172 mg, 0.895 mmol). The mixture is stined for 15 min at RT, and methyl ester hydrochloride (112 mg, 0.895 mmol) is added. The reaction is stined for 18 h and quenched with NH
4CI (5.0 mL). The organic layer is loaded onto a silica gel column and purified with 20-50% EtOAc/Hex to afford the intermediate amide ester. The intermediate is dissolved in methanol (3.0 mL) and THF (2.0 mL) and treated with NaOH (2.0 M, 5.0 mL). The resulting mixture is stined at RT for 3 h. The mixture is concentrated, acidified with HCI (1 N) to pH~3, and extracted with EtOAc (2 x 20 mL). The organic layer is dried over Na
2SO and concentrated to afford the title compound (285 mg, 94%). MS (ES) m/e 510.1 (M+l), 508.1 (M-l).
Example 6.
Preparation of [(6-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl } -benzo[b]thiophene-2-carbonyl)-amino]-acetic acid.
[(6- { 1 - [4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-2-carbonyl)-amino] -acetic acid (220 mg, 0.432 mmol) is reduced by NaBH (33 mg, 0.863 mmol) as in Step Fl 1 to afford the title compound (175 mg, 79%). MS (ES) m/e 512.1 (M+l), 510.1 (M-l).
Example 7.
Preparation of 2-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-6-carboxylic acid.
A. 2-(l-Ethyl-l-hydroxy-propyl)-benzo[b]thiophene-6-carboxylic acid.
A solution of 6-iodo-benzo[b]thiophene-2-carboxylic acid ethyl ester (5.05 g, 15.2 mmol) in THF (200 mL) at -78 °C is de-oxygenated (vacuum/N2 purge 3 x). Ethylmagnesium bromide (25.3 mL, 3.0 M) is added, and the reaction mixture is allowed to warm to RT over 30 min. The mixture is stined for 10 min at RT, cooled to -78 °C, and treated with clean dry ice (100 g). The mixture is allowed to warm to RT over 60 min and is quenched with HCI (50 mL, 1.0 M). THF is removed under vacuum, and the residue is extracted with EtOAc (3 x 100 mL). The organic layer is dried over Na
2SO
4 and concentrated to give the product (3.80 g, 94%). MS (ES) m/e 263.0 (M-l).
B . 2- [ 1 -Ethyl- 1 -(4-hydroxy-3 -methyl-phenyl)-propyl] -benzo [b] thiophene-6- carboxylic acid.
A solution of 2-(l -ethyl- l-hydroxy-propyl)-benzo[b]thiophene-6-carboxylic acid (3.80 g, 14.4 mmol) and o-cresol (1.94 g, 18.0 mmol) in CH
2C1
2 (100 mL) at -40 °C is added BF
3-Et
2O (1.02 g, 7.20 mol). After stining forlO min, the reaction mixture is allowed to warm up to 0 °C over 30 min. The reaction is quenched with water (15 mL) and the aqueous layer is extracted with EtOAc (50 mL). The organic
layer is dried over Na
2SO
4, concentrated, and purified by silica' gel column chromatography (2% HO Ac in 50% EtOAc/Hex) to give the product (3.98 g, 78%). MS (ES) m/e 353.0 (M-l).
C . 2- [ 1 -Ethyl- 1 -(4-hydroxy-3 -methyl-phenyl)-propyl] -benzo [b] thiophene-6- carboxylic acid methyl ester.
A solution of 2- [1-ethyl- l-(4-hydroxy-3 -methyl-phenyl)-propyl]- benzo[b]thiophene-6-carboxylic acid (3.98 g, 11.23 mmol) in MeOH (100 mL) is treated with H
2SO
4 (concentrated, 1.0 mL). The mixture is stined at 80 °C for 8 h and neutralized with aqueous NaHCO
3. The MeOH is removed under vacuum and the residue is extracted with EtOAc (2 x 100 mL). The combined organic layer is dried over Na
2SO
4, concentrated and purified by silica gel column chromatography (20% EtOAc/Hex) to give the product (4.05 g, 98%). MS (ES) m/e 367.1 (M-l).
D . 2- { 1 - [4-(3 ,3 -dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-6-carboxylic acid methyl ester.
A solution of 2-[l-ethyl-l-(4-hydroxy-3-methyl-phenyl)-propyl]- benzo[b]thiophene-6-carboxylic acid methyl ester (4.05 g, 11.0 mmol) and 1- bromopinacolone (2.36 g, 13.2 mmol) in acetone (100 mL) is treated with K
2CO
3 (3.04 g, 22.0 mmol) and stined at RT for 16 h. The mixture is filtered, and the filhate is concentrated. The residue is purified by silica gel column chromatography (10- 15% EtOAc/Hex) to provide the title compound (4.50 g, 88%). MS (ES) m/e 484.2 (M+18).
E. 2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-proρyl}- benzo[b]thiophene-6-carboxylic acid.
2- { 1 - [4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-6-carboxylic acid methyl ester (120 mg, 0.257 mmol) is hydrolyzed as in Step El 1 to give the title compound (72 mg, 62%). MS (ES) m/e 451.2 (M-l), 470.2 (M+18).
Example 8.
Preparation of 2-{ l-Ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl } -benzo[b]thiophene-6-carboxylic acid.
2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-6-carboxylic acid (53 mg, 0.117 mmol) is reduced by NaBPL (9 mg, 0.234 mmol) as in a reaction analogous to Example 2 to afford the title compound (53 mg, 99%). MS (ES) m/e 453.2 (M-l), 472.2 (M+18).
Example 9.
Preparation of 2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-6-carboxylic acid dimethylamide.
2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thioρhene-6-carboxylic acid (175 mg, 0.387 mmol), D'MAP (142 mg, 1.16 mmol), EDC (111 mg, 0.580 mmol), and dimethylamine hydrochloride (47 mg, 0.580 mmol) are reacted analogous to Example 3 to afford the title compound (140 mg, 76%).
MS (ES) m/e 480.2 (M+l).
Example 10.
Preparation of 2-{ l-Ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl}-benzo[b]thiophene-6-carboxylic acid dimethylamide.
2- { l-[4-(3 ,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]- 1-ethyl-propyl } - benzo[b]thiophene-6-carboxylic acid dimethylamide (90 mg, 0.188 mmol) is reduced using NaBH (14 mg, 0.376 mmol) in a reaction analogous to Example 2 to afford the title compound (90 mg, 100%). MS (ES) m/e 482.2 (M+l).
Example 11.
Preparation of [(!-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl } -benzo[b]thiophene-6-carbonyl)-amino]-acetic acid.
2- { 1 -[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]- 1-ethyl-propyl } - benzo[b]thiophene-6-carboxylic acid (175 mg, 0.387 mmol), DMAP (142 mg, 1.16 mmol), EDC (111 mg, 0.580 mmol), and glycine methyl ester hydrochloride (73 mg, 0.580 mmol) are reacted and hydrolyzed in a sequence analogous to Example 5 to afford the title compound (180 mg, 91%). MS (ES) m/e 508.2 (M-l), 510.1 (M+l).
Example 12.
Preparation of [(!-{ l-Efhyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl } -benzo[b]thiophene-6-carbonyl)-amino]-acetic acid.
[(2- { 1- [4-(3 ,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl] - 1 -exhyl-propyl } - benzo[b]thiophene-6-carbonyl)-amino]-acetic acid (135 mg, 0.265 mmol) is reduced using NaBHLi (20 mg, 0.530 mmol) as in example 2 to afford the title compound (135 mg, 99%). '
MS (ES) m/e 512.2 (M+l), 510.2 (M-l).
Example 13.
Preparation of 5-{ l-[4-(3,3-dimefhyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl } -benzo [b] thiophene-2-carboxylic acid.
A. 1 -( 1 -Ethyl-propenyl)-4-fluoro-benzene.
A solution of 4-fluoro-benzoic acid methyl ester (19.1 g, 114 mmol) in THF (300 mL) at -78 °C is heated with ethylmagnesium bromide (135 mL, 3.0 M). The reaction mixture is allowed to warm to 0 °C over 30 min and quenched with HCI (350 mL, 1.0 M). THF is removed under vacuum, and the residue is extracted with EtOAc (3 x 100 mL). The organic layer is dried over Na
2SO
4 and concentrated. The intermediate tertiary alcohol is dissolved in CH
2C1
2 (200 mL), cooled to -40 °C, and treated with BF
3-Et
2O (11.5 g, 91.0 mol). The reaction mixture is allowed to warm to 0 °C over 30 min and quenched with water (50 mL). The organic layer is dried over
Na SO
4 and concentrated. The residue is purified using silica gel column chromatography (5% EtOAc/Hex) to afford the title compound (16.8 g, 90%). H-NMR (ppm, CDC1
3), δ: 7.27 (2 H, m), 6.97 (2 H, m), 5.66 (1 H, q, 7=7.0 Hz), 2.49 (2 H, q, 7=7.0 Hz), 1.78 (3 H, d, 7=7.0 Hz ), 0.98 (3 H, t, 7=7.0 Hz ).
B . 5-( 1 -Ethyl-propenyl)-2-fluoro-benzaldehyde.
A solution of l-(l-efhyl-propenyl)-4-fluoro-benzene (15.3 g, 93.2 mmol) in THF (150 mL) at -78 °C is treated with n-BuLi (70 mL, 1.6 M). The reaction mixture is allowed to warm to 0 °C over 60 min, cooled to -78 °C, and treated with DMF (10.2 g, 140 mmol). The mixture is stined for 10 min and quenched with HOAc (10 mL) and water (200 mL). The mixture is allowed to warm to RT, and the THF is removed under vacuum. The residue is extracted with EtOAc (2 x 100 mL, and the organic layer is dried over Na
2SO
4 andconcentrated. The crade product is purified using silica gel column chromatography (7% EtOAc/Hex) to afford the title compound (8.70 g, 49%).
H-NMR (ppm, CDC13), δ: 10.34 (1 H, s), 7.79 (1 H, dd, 7=2.7, 6.6 Hz), 7.54 (1 H, m), 7.09 (1 H, dd, 7=1.3, 9.7 Hz), 5.72 (1 H, q, 7=7.0 Hz), 2.50 (2 H, q, 7=7.5 Hz), 1.79 (3 H, d, 7=7.0 Hz ), 0.97 (3 H, t, 7=7.5 Hz ).
C. 5-(l-Ethyl-propenyl)-benzo[b]thiophene-2-carboxylic acid ethyl ester.
A solution of 5-(l-efhyl-propenyl)-2-fluoro-benzaldehyde (8.70 g, 45.3 mmol) in DMF (40 mL) is treated with mercapto-acetic acid ethyl ester (8.16 g, 67.9 mmol) and K
2CO
3 (12.5 g, 90.6 mmol). The resulting suspension is stined at 80 °C for 60 min and quenched with water (300 mL). The mixture is extracted with EtOAc (2 x 200 mL), and the organic layer is dried over Na
2SO , concentrated, and purified using
silica gel column chromatography (6% EtOAc/Hex), to give the title compound as an oil (11.0 g, 89%).
H-NMR (ppm, CDC13), δ: 8.01 (1 H, s), 7.78 (1 H, d, 7=1.8 Hz), 7.75 (1 H, d, 7=8.4 Hz), 7.44 (1 H, dd, 7=1.8, 8.4 Hz), 5.75 (1 H, q, 7=7.0 Hz), 4.41 (2 H, q, 7=7.0 Hz), 2.58 (2 H, q, 7=7.5 Hz), 1.82 (3 H, d, 7=7.0 Hz ), 1.42 (3 H, t, 7=7.0 Hz ), 0.97 (3 H, t, 7=7.5 Hz ).
D. 5-[l-Ethyl-l-(4-hydroxy-3-methyl-phenyl)-propyl]-benzo[b]thiophene-2- carboxylic acid ethyl ester.
5-(l-Ethyl-propenyl)-benzo[b]thiophene-2-carboxylic acid ethyl ester (5.50 g, 20.0 mmol), o-cresol (4.33 g, 40.0 mmol), and BF
3-Et
2O (10 mL) are reacted analogous to Example IC to afford the title compound (5.97 g, 78%). MS (ES) m/e 381.3 (M-l), 383.4 (M+l).
E. 5-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-2-carboxylic acid ethyl ester.
5- [ 1 -Ethyl- 1 -(4-hydrόxy-3 -methyl-phenyl)-propyl] -benzo [b]thiophene-2- carboxylic acid ethyl ester (5.97 g, 15.6 mmol), 1-bromopinacolone (4.20 g, 23.4 mmol), and K
2CO
3 (6.46 g, 46.8 mmol) in acetone (100 mL) are reacted analogous to Example ID to provide the title compound (7.10 g, 95%). MS (ES) m/e 498.4 (M+18).
F. 5-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-ρropyl}- benzo[b]thiophene-2-carboxylic acid.
5-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-ρhenyl]-l-ethyl-propyl}- benzo[b]thiophene-2-carboxylic acid ethyl ester (2.50 g, 5.20 mmol) is hydrolyzed as in Step El 1 to give the title compound (2.20 g, 94%). MS (ES) m/e 451.2 (M-l), 470.2 (M+18).
Example 14.
Preparation of 5-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl } -benzo[b]thiophene-2-carboxylic acid.
5-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-2-carboxylic acid (0.280 g, 0.619 mmol) is reduced using NaBFL (14 mg, 0.376 mmol) analogous to Example 2 to afford the title compound (0.241 g, 86%). MS (ES) m/e 453.1 (M-l), 472.2 (M+18).
Example 15.
Preparation of [(5-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl } -benzo[b]thiophene-2-carbonyl)-amino]-acetic acid.
5- { 1 - [4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-2-carboxylic acid (310 mg, 0.686 mmol), DMAP (167 mg, 1.37 mmol), EDC (197 mg, 1.03 mmol), and glycine methyl ester hydrochloride (172 mg, 1.37 mmol) are reacted and hydrolyzed in a sequence analogous to Example 5 to afford the title compound (150 mg, 43%). MS (ES) m/e 508.2 (M-l), 510.1 (M+l).
Example 16.
Preparation of [(5-{ l-efhyl-l-[4-(2-hyαiOxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl}-benzo[b]thiophene-2-carbonyl)-amino]-acetic acid.
[(5- { 1 - [4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-2-carbonyl)-amino]-acetic acid (90 mg, 0.177 mmol) is reduced using NaBFL (13 mg, 0.354 mmol) in a reaction analogous to Example 2 to afford the title compound (73 mg, 81%). MS (ES) m/e 510.2 (M-l), 512.2 (M+l).
Example 17.
Preparation of 2-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-5-carboxylic acid.
A. 5-Methoxy-benzo[b]thiophene-2-carboxylic acid ethyl ester.
\ O. A" A solution of 2-fluoro-5-methoxy-benzaldehyde (4.35 g, 28.2 mmol) in DMF (30 mL) is added mercapto-acetic acid ethyl ester (3.71 mL, 33.9 mmol) and K2CO3 (7.86 g, 57.0 mmol). The resulting suspension is stined at 80 °C for 60 min and quenched with water (300 mL). The mixture is extracted with EtOAc (2 x 200 mL), and the organic layer is dried over Na2SO4, concentrated, and purified by silica gel column chromatography (10% EtOAc/Hex), to give the title compound as an oil (4.40 g, 66%).
H-NMR (ppm, CDC13), δ: 7.97 (1 H, s), 7.70 (1 H, d, 7=8.8 Hz), 7.27 (1 H, d, 7=2.6 Hz), 7.09 (1 H, dd, 7=2.6, 8.8 Hz), 4.41 (2 H, q, 7=7.0 Hz), 3.88 (3 H, s), 1.42 (3 H, t, 7=7.0 Hz).
B. 3-(5-Methoxy-benzo[b]thiophen-2-yl)-pentan-3-ol.
5-Methoxy-benzo[b]thiophene-2-carboxylic acid ethyl ester (4.13 g, 17.5 mmol) in THF (100 mL) at -78 °C is treated with ethylmagnesium bromide (17.5 mL, 3.0 M). The reaction mixture is allowed to warm to RT over 30 min and is stined for 10 min. The mixture is quenched with water (40 mL) and acidified with HCI (50 mL, 1.0 M). THF is removed under vacuum, and the residue is extracted with EtOAc (2 x 40 mL). The organic layer is dried over Na
2SO , concentrated, and purified using silica gel column chromatography (10% EtOAc/Hex) to give the title compound (4.53 g, 97%).
H-NMR (ppm, CDC13), δ: 7.63 (1 H, d, 7=8.8 Hz), 7.17 (1 H, d, 7=2.2 Hz), 7.03 (1 H, s), 6.92 (1 H, dd, 7=2.6, 8.8 Hz), 3.86 (3 H, s), 1.91 (4 H, q, 7=7.0 Hz), 0.91 (6 H, t, 7=7.0 Hz).
C. 4-[l-Ethyl-l-(5-methoxy-benzo[b]thiophen-2-yl)-propyl]-2-methyl-phenol.
3-(5-Methoxy-benzo[b]thiophen-2-yl)-pentan-3-ol (2.16 g, 8.63 mmol), o- cresol (4.66 g, 43.1 mmol), and BF -Et
2O (1.64 mL, 12.9 mmol) are reacted analogous to Example IC to afford the title compound (2.29 g, 78%). MS (ES) m/e 339.2 (M-l), 341.3 (M+l).
D. l-{4-[l-Ethyl-l-(5-methoxy-benzo[b]thiophen-2-yl)-propyl]-2-methyl-phenoxy}- 3,3-dimethyl-butan-2-one (2131468) (PF1-A03098-163)
4-[l-Ethyl-l-(5-methoxy-benzo[b]thiophen-2-yl)-propyl]-2-methyl-phenol (7.80 g, 22.9 mmol), 1-bromopinacolone (8.20 g, 45.8 mmol), and K
2CO
3 (6.32 g,
45.8 mmol) in acetone (100 mL) are reacted analogous to Example ID to provide the title compound (9.20 g, 92%).
H-NMR (ppm, CDC13), δ: 7.53 (1 H, d, 7=8.8 Hz), 7.15 (1 H, d, 7=2.6 Hz), 7.02-7.06 (3 H, m), 6.87 (1 H, dd, 7=2.6, 8.8 Hz), 6.50 (1 H, d, 7=8.4 Hz), 4.84 (2 H, s), 3.85 (3 H, s), 2.25 (3 H, s), 2.15 (4 H, q, 7=7.5 Hz), 1.26 (9 H, s), 0.73 (6 H, t, 7=7.5 Hz).
E. 1- {4-[ 1-Ethyl- 1 -(5-hydroxy-benzo[b]thiophen-2-yl)-propyl]-2-methyl-phenoxy } - 3,3-dimethyl-butan-2-one.
A solution of l-{4-[l-ethyl-l-(5-methoxy-benzo[b]thiophen-2-yl)-propyl]-2- methyl-phenoxy}-3,3-dimethyl-butan-2-one (0.86 g, 1.96 mmol) in CH
2C1
2 (15.0 mL) at 0 °C is treated with BBr
3 (3.0 mL, 2.94 mmol). The mixture is stined for 2 h and quenched with water (10 mL). The aqueous layer is extracted with CH C1
2 (30 mL). The organic layer is concentrated and purified using silica gel column cliromatography (25% EtOAc/Hex) to afford the title compound (0.412 g, 50%). H-NMR (ppm, CDC1
3), δ: 7.50 (1 H, d, 7=8.4 Hz), 7.10 (1 H, d, 7=2.6 Hz), 7.02-7.06 (2 H, m), 6.98 (1 H, s), 6.78 (1 H, dd, 7=2.6, 8.8 Hz), 6.51 (1 H, d, 7=8.4 Hz), 4.84 (2 H, s), 2.25 (3 H, s), 2.15 (4 H, q, 7=7.5 Hz), 1.26 (9 H, s), 0.73 (6 H, t, 7=7.5 Hz).
F. Trifluoromethanesulfonic acid 2-{l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl- phenyl] - 1 -ethyl-propyl } -benzo [b] thiophen-5-yl ester.
A solution of l-{4-[l-ethyl-l-(5-hydroxy-benzo[b]thiophen-2-yl)-propyl]-2- methyl-phenoxy}-3,3-dimethyl-butan-2-one (3.21 g, 7.56 mmol) in CH
2C1
2 (50 mL) at -78 °C is treated with 2,6-lutidine (1.32 mL, 11.3 mmol) and trifluoromethanesulfonic acid anhydride (1.78 mL, 10.6 mmol). The mixture is stined and allowed to warm to -20 °C over 60 min. The mixture is quenched with
water (5.0 mL). The organic layer is concentrated and purified using silica gel column chromatography (10% EtOAc/Hex) to afford the title compound (4.20 g,
99%).
H-NMR (ppm, CDC13), δ: 7.69 (1 H, d, 7=8.8 Hz), 7.57 (1 H, d, 7=2.2 Hz), 7.13 (1 H, s), 7.11 (1 H, dd, 7=2.2, 8.8 Hz), 7.04 (1 H, s), 7.01 (1 H, dd, 7=2.2, 8.8 Hz), 6.52 (1
H, d, 7=8.4 Hz), 4.84 (2 H, s), 2.27 (3 H, s), 2.15 (4 H, q, 7=7.5 Hz), 1.26 (9 H, s),
0.74 (6 H, t, 7=7.5 Hz).
G. 2-{l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-5-carboxylic acid methyl ester.
A solution of trifluoromethanesulfonic acid 2-{ l-[4-(3,3-dimethyl-2-oxo- butoxy)-3-methyl-phenyl]-l-ethyl-propyl}-benzo[b]thiophen-5-yl ester (4.10 g, 7.36 mmol) in DMSO (15 mL) and MeOH (10 mL) is treated with Pd(OAc)2 (0.169 g), DPPB (0.368 g) and Et3N (5.3 mL). The mixture is stined under CO (100 psi) at 80 °C for 4 h. The MeOH is removed in vacuum, and the residue is poured into water (150 mL) and extracted with EtOAc (2 x 50 mL). The organic layer is dried over Na SO , concentrated, purified using silica gel column cliromatography (10% EtOAc/Hex), to give the title compoimd as an oil (3.10 g, 90%). H-NMR (ppm, CDC13), δ: 8.38 (1 H, d, 7=2.6 Hz), 7.87 (1 H, dd, 7=1.3, 8.4 Hz), 7.70 (1 H, d, 7=8.8 Hz), 7.19 (1 H, s), 7.05 (1 H, s), 7.01 (1 H, dd, 7=2.2, 8.4 Hz), 6.52 (1 H, d, 7=8.4 Hz), 4.84 (2 H, s), 3.94 (3 H, s), 2.26 (3 H, s), 2.18 (4 H, q, 7=7.5 Hz), 1.26 (9 H, s), 0.74 (6 H, t, 7=7.5 Hz). MS (ES) m/e 484.4 (M+18).
H. 2-{l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-5-carboxylic acid.
2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-5-carboxylic acid methyl ester (2.95 g, 6.32 mmol) is hydrolyzed as in Step El 1 to give the title compound (2.80 g, 98%). MS (ES) m/e 451.2 (M-l), 470.2 (M+18).
Example 18.
Preparation of 2-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]- propyl }-benzo[b]thiophene-5-carboxylic acid.
2-{l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-5-carboxylic acid (330 mg, 0.730 mmol) is reduced using NaBH t (55 mg, 1.46 mmol) in a reaction analogous to Example 2 to afford the title compound (0.306 g, 92%). MS (ES) m/e 453.2 (M-l), 472.2 (M+18).
Example 19.
Preparation of [(2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl } -benzo[b]thiophene-5-carbonyl)-amino]-acetic acid.
2-{l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl-propyl}- benzo[b]thiophene-5-carboxylic acid (310 mg, 0.686 mmol), DMAP (167 mg, 1.37 mmol), EDC (197 mg, 1.03 mmol), and glycine methyl ester hydrochloride (172 mg, 1.37 mmol) are reacted and hydrolyzed in a sequence analogous to Example 5 to afford the title compound (246 mg, 70%). MS (ES) m/e 508.2 (M-l), 510.1 (M+l).
Example 20.
Preparation of [(2-{ l-Ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-phenyl]/- propyl}-benzo[b]thiophene-5-carbonyl)-amino]-acetic acid.
[(2- { 1 - [4-(3 ,3-Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl] - 1 -ethyl-propyl } - benzo[b]thiophene-5-carbonyl)-amino]-acetic acid (150 mg, 0.295 mmol) is reduced using NaBH-t (22 mg, 0.589 mmol) analogous to Example 2 to afford the title compound (130 mg, 86%). MS (ES) m/e 510.2 (M-l), 512.2 (M+l).
Example 21.
Preparation of 2-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-5-carboxylic acid dimethylamide.
A solution of 2-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-5-carboxylic acid (210 mg, 0.464 mmol) in CH
2C1
2 (5.0 mL) is treated with Et
3N (1.0 mL), HOBT (94 mg, 0.70 mmol), and EDC (134 mg, 0.70 mmol). The mixture is stined for 10 min at RT, and dimethylamine (1.0 mL, 2.0 M in THF) is added. The reaction mixture is stined for 18 h, loaded onto a silica gel column, and eluted with 65% EtOAc/Hex to afford the title compound (90 mg, 41%). MS (ES) m/e 480.4 (M+l).
Example 22.
Preparation of 2- { 1 -Ethyl- l-[4-(2-ethyl-2-hydroxy-butoxy)-3 -methyl-phenyl] -propyl} benzo[b]thiophene-6-carboxylic acid
A. {4-[l-Ethyl-l-(6-methoxy-benzo[b]thiophen-2-yl)-propyl]-2-methyl-phenoxy}-acetic acid methyl ester.
To a mixture of 4-[l-ethyl-l-(6-methoxy-benzo[b]thiophen-2-yl)-propyl]-2- mefhyl-phenol (2.53 g, 7.43 mmol) and acetonitrile (25 mL) is added methyl bromoacetate (0.84 mL, 8.92 mmol), powdered potassium carbonate (4.10 g, 29.72 mmol), and potassium iodide (0.12 g, 0.74 mmol). The resulting sluny is stined at reflux temperature for 2 h, filtered, and concentrated. The residue is dissolved in diethyl ether (100 mL), washed with water (75 ml), brine (75 mL), dried over MgSO
4, filtered, and concenfrated to give the title compound (1.5 g, 3.63 mmol, 49%). 1H NMR (CDC1
3), δ 0.74 (t, 7= 7.2 Hz, 6H), 2.17 (q, 7 = 14.7, 7.5 Hz, 4H), 2.25 (s, 3H), 2.30 (s, 2H), 3.80 (s, 3H), 3.86 (s, 3H), 3.60 (d, 7= 8.6 Hz, IH), 6.89 (dd, 7= 8.6, 2.5 Hz, IH), 7.04 (s, IH), 7.07 (s, IH), 7.16 (d, 7 = 2.5 Hz, IH), 7.55 (d, 7= 8.9 Hz, IH).
B. 3-{4-[l-Ethyl-l-(6-methoxy-benzo[b]thiophen-2-yl)-propyl]-2-methyl- phenoxymethyl } -pentan-3-ol.
To a mixture of {4-[l-ethyl-l-(6-mefhoxy-benzo[b]thiophen-2-yl)-propyl]-2- methyl-phenoxy} -acetic acid methyl ester (1.50 g, 3.64 mmol) and THF (20 mL) is added ethyl magnesium bromide (3.0 mL, 9.1 mmol, 3M in THF). The solution is heated at a reflux for 3 h, diluted with saturated ammonia chloride solution (80 mL), extracted with diethyl ether (2 x 75 mL), dried over MgSO
4, filtered, and concentrated. The residue is purified by silica gel chromatography (10% to 30% EtOAc gradient) to give the title compound (0.90g, 2.04 mmol, 56%). 1H NMR (CDC1
3), δ 0.75 (t, 7 = 7.6 Hz, 6H), 0.95 (t, 7= 7.4 Hz, 6H), 1.68 (q, 7= 14.8, 7.5 Hz, 4H), 2.13-2.24 (m, 7H), 3.81 (s, 2H), 3.86 (s, 3H), 6.73 (dd, 7 = 8.2 Hz, IH), 6.89 (dd, 7 = 8.8, 2.6 Hz, IH), 7.06 (s, 2H), 7.09 (dd, 7 = 8.4, 2.5 Hz, IH), 7.17 (d, 7 = 2.7 Hz, IH), 7.55 (d, 7 = 8.8 Hz, IH). LC/MS (m/z): calcd. for C
27H
37O
3S (M+H)
+: 441.3; found: 441.2.
C. 2-{ l-Ethyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl} benzo[b] thiophen-6-ol.
To a solution of 3-{4-[l-ethyl-l-(6-methoxy-benzo[b]thiophen-2-yl)-propyl]-2- methyl-phenoxymethyl}-pentan-3-ol (0.80 g, 1.82 mmol) and DMF (10 mL) is added NaSEt (1.53 g, 18.15 mmol). The resulting solution is heated at 100 °C overnight, diluted with diethyl ether (50 mL), washed with water (3 x 50 mL), brine (50 mL), dried with MgSO , filtered and concentrated. The resulting residue is purified by silica gel chromatography (10% to 40% EtOAc gradient) to give the title compound (0.67 g, 1.57 mmol, 87%). 1H NMR (CDC13), δ 0.75 (t, 7 = 7.2 Hz, 6H), 0.95 (t, 7 = 7.4 Hz, 6H), 1.67 (q, 7 = 15.1, 7.4 Hz, 4H), 2.12-2.24 (m, 7H), 3.82 (s, 2H), 6.73 (d, 7 = 8.6 Hz, IH), 6.79 (dd, 7= 9.0, 2.6 Hz, IH), 6.79 (dd, 7 = 9.0, 2.6 Hz, IH), 7.00 (s, IH), 7.05 (d, 7= 2.2 Hz,
1H), 7.08 (dd, 7 = 8.2, 2.6 Hz, IH), 7.11 (d, 7= 2.2 Hz, IH), 7.49 (d, 7= 9.0 Hz, IH). LC/MS (m/z): calcd. for C26H35O3S (M+H)+: 427.2; found: 427.1.
D. Trifluoro-methanesulfonic acid 2-{l-ethyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl- phenyl] -propyl } -benzo [b] thiophen-6-yl ester
To a solution of 2- {1 -Ethyl- l-[4-(2-ethyl-2-hydroxy-butoxy)-3 -methyl-phenyl] - propyl} benzo[b] thiophen-6-ol (0.67 g, 1.57 mmol) and dichloromethane (5 mL) is added NEt
3 (0.44 mL, 3.14 mmol). The solution is cooled to 0 °C, and triflouromethane- sulfonic anhydride (0.28 mL, 1.65 mmol) is added. The solution is warmed to RT, diluted with diethyl ether (30 mL), washed with saturated NaHCO
3 (20 mL), dried with MgSO
4, filtered, and concentrated to give the title compound (0.22 g, 0.40 mmol, 85%). 1H NMR (CDC1
3), δ 0.75 (t, 7= 7.3 Hz, 6H), 0.95 (t, 7= 7.5 Hz, 6H), 1.68 (q, 7= 14.8, 7.6 Hz, 4H), 2.14-2.27 (m, 7H), 3.82 (s, 2H), 6.74 (d, 7= 8.3 Hz, IH), 7.03 (d, 7 = 2.1 Hz, IH), 7.08 (dd, 7= 8.7, 2.5 Hz, IH), 7.13 (dd, 7= 8.7, 2.9 Hz, IH), 7.14 (s, IH), 7.58 (d, 7 = 2.1 Hz, IH), 7.70 (d, 7= 8.3 Hz, IH). LC/MS (m/z): calcd. for C
27H
34F
3O
5S
2 (M+H)
+: 559.2; found: 576.1.
E. 2- { l-Ethyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl } benzo[b]thiophene-6-carboxylic acid methyl ester.
To a solution of trifluoro-methanesulfonic acid 2- { 1-ethyl- l-[4-(2-ethyl-2- hydroxy-butoxy)-3-methyl-ρhenyl]-propyl}-benzo[b]thiophen-6-yl ester (0.66 g, 1.19 mmol) and DMSO (15 mL) is added MeOH (10 mL), NEt
3 (0.77 mL), DPPB (0.054 g, 0.13 mmol), Pd(OAc)
2 (0.024, 0.01 mmol), and CO (100 psi). The solution is heated at 80 °C overnight, diluted with IM HCI (100 mL), and extracted with diethyl ether (2 x 50 mL). The combined organic layers are washed with water (75 mL) then brine (75 mL), dried MgSO
4, filtered, and concentrated. The residue is purified by silica gel chromatography (10% to 40% EtOAc gradient) to give the title compound (0.84 g, 2.04 mmol, 68%). 1H NMR (CDC1
3), δ 0.75 (t, 7= 7.2 Hz, 6H), 0.95 (t, 7= 7.7 Hz, 6H), 1.67 (q, 7= 14.6, 7.5 Hz, 4H), 2.14-2.25 (m, 7H), 3.81 (s, 2H), 3.95 (s, 3H), 6.73 (d, 7= 8.7 Hz, IH), 7.04 (d, 7= 2.2 Hz, IH), 7.08 (dd, 7= 8.4, 2.2 Hz, IH), 7.20 (s, IH), 7.71 (d, 7 = 8.5 Hz, IH), 7.89 (dd, 7= 8.4, 1.7 Hz, IH), 8.39 (d, 7= 1.7 Hz, IH). LC/MS (m/z): calcd. for C
28H
37O
4S (M+H)
+: 469.2; found: 486.2.
F. 2- { 1 -Ethyl- 1 - [4-(2-ethyl-2-hydroxy-butoxy)-3 -methyl-phenyl] -propyl } benzo[b]thiophene-6-carboxylic acid.
2- { 1 -Ethyl- 1 - [4-(2-ethyl-2-hydroxy-butoxy)-3 -methyl-phenyl] -propyl } benzo[b]thiophene-6-carboxylic acid methyl ester (0.48 g, 1.02 mmol) is dissolved in MeOH (2 mL) and treated with H2O (0.5 mL) and NaOH (0.20 g, 5.12 mmol). The resulting mixture is heated at a reflux for 2 h, cooled to RT, and stined overnight. The solution is diluted with H2O (10 mL), adjusted to pH 3-4 using 1 M HCI, and extracted with EtOAc (40 mL). The EtOAc layer is washed with brine (20 mL), dried with MgSO , filtered, and concentrated to yield the title compound (0.42 g, 0.92 mmol, 89%). 1H NMR (CDC13), δ 0.76 (t, 7 = 7.5 Hz, 6H), 0.95 (t, 7 = 7.5 Hz, 6H), 1.68 (q, 7= 14.8, 7.5 Hz, 4H), 2.25-2.27 (m, 7H), 3.82 (s, 2H), 6.74 (d, 7= 8.8 Hz, IH), 7.05 (d, 7 = 2.3 Hz, IH), 7.09 (dd, 7= 8.5, 2.1 Hz, IH), 7.24 (s, IH), 7.75 (d, 7 = 8.4 Hz, IH), 7.96 (dd, 7= 8.3, 1.7 Hz, IH), 8.48 (d, 7= 1.7 Hz, IH). Exact mass (m/z): calcd. for C27H35O4S (M+H)+: 454.2; found: 472.3.
Example 23.
Preparation of 5-{ l-[4-(2-tert-Butyl-[l,3]dioxolan-2-ylmethoxy)-3-methyl-phenyl]-l- ethyl-propyl}-benzo[b]thiophene-2-carboxylic acid ethyl ester.
A solution of 5-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-l-ethyl- propyl}-benzo[b]thiophene-2-carboxylic acid ethyl ester (170 mg, 0.354 mmol) ) in ethylene glycol (3.0 mL) is treated with BF3-Et2O (2.0mL). The mixture is stined at 90 °C for 6 h, cooled to RT, and quenched with water (50 mL). The mixture is extracted with EtOAc (2 x 20 mL), and the organic layer is concentrated and purified using silica gel column chromatography (10% EtOAc/Hex) to afford the product as an oil (94 mg, 51%). MS (ES) m/e 542.3 (M+18).
Example 24.
Preparation of methanesulfonic acid 2-{ l-[4-(3,3-dimethyl-2-oxo-butoxy)-3-methyl- phenyl]- 1-ethyl-propyl } -benzo[b]thiophen-5-yl ester.
A solution of l-{4-[l-ethyl-l-(5-hydroxy-benzo[b]thiophen-2-yl)-propyl]-2- methyl-phenoxy}-3,3-dimethyl-butan-2-one (260 mg, 0.612 mmol) in CH2C12 (20 mL) at RT is treated with Et3N (0.40 mL) and methanesulfonyl chloride (140 mg, 1.22 mmol). The mixture is stined for 30 min and quenched with water (1.0 mL). The organic layer is concentrated and purified using silica gel column chromatography (25% EtOAc/Hex) to afford the title compound (190 mg, 62%).
NMR (ppm, CDC13), δ: 7.67 (1 H, d, 7=8.8 Hz), 7.59 (1 H, d, 7=2.2 Hz), 7.13 (1 H, dd, 7=2.2, 8.8 Hz), 7.11 (1 H, s), 7.01-7.04 (2 H, m), 6.51 (1 H, d, 7=8.4 Hz), 4.84 (2 H, s), 3.16 (3 H, s), 2.26 (3 H, s), 2.15 (4 H, q, 7=7.5 Hz), 1.26 (9 H, s), 0.74 (6 H, t, 7=7.5 Hz). MS (ES) m/e 520.3 (M+18).
Example 25. Preparation of enantiomers of 2-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3- methyl-phenyl] -propyl } -benzo[b]thiophene-6-carboxylic acid
Step A: Preparation of racemic of 2-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)- 3-methyl-phenyl]-propyl}-benzo[b]thiophene-6-carboxylic acid methyl ester
2- { l-[4-(3 ,3 -Dimethyl-2-oxo-butoxy)-3 -methyl-phenyl]- 1-ethyl-propyl } - benzo[b]thiophene-6-carboxylic acid methyl ester (4.50 g, 9.64 mmol) in THF (100 mL) is reacted with NaBEL (0.37 g, 9.64 mmol) analogous to Example 2 to obtain the title compound (3.90 g, 86%).
Step B: Preparation of enantiomers of 2-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl- butoxy)-3-methyl-phenyl]-propyl}-benzo[b]thiophene-6-carboxylic acid methyl ester A mixture of racemic 2-{ l-ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3- methyl-phenyl]-propyl}-benzo[b]thiophene-6-carboxylic acid methyl ester (3.90 g) is chromatographed with Chiralpak AD column to give enantiomer 1, Example QE1 (1.495 g, 38%) and enantiomer 2, Example QE2 (1.489 g, 38%).
Enantiomer 1, Example 25 A HPLC: ChiralPak AD (4.6x150); 60%MeOH/40%ACN/0.05%dmea; 0.6 mL/min (flow rate); uv: 287 nm. Rt = 6.0 m. NMR eq to Step A .
Enantiomer 2, Example 25B HPLC: ChiralPak AD (4.6x150); 60%MeOH/40%ACN/0.05%dmea; 0.6 mL/min (flow rate); uv: 287 nm. Rt = 8.5 m. NMR eq to Step A .
Step C: Enantiomer 1 (1.495 g, 3.19 mmol) is hydrolysis analogous to Example 7, step E to provide acid 2290264 (1.44 g, 99%). MS are eq to those in Example 8. Enantiomer 2 (1.489 g, 3.18mmol) is hydrolysis analogous to Example 7, step E to provide acid 2290265 (1.44 g, 99%). MS are eq to those in Example 8.
Example 26. D-l-[(l- { 1-Ethyl- 1 -[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-proρyl } - benzo[ ?]thiophene-6-carbonyl)-amino]-propionic acid methyl ester.
Using the procedure analogous to Example 5, from 2-{l-ethyl-l-[4-(2-ethyl-2- hydroxy-butoxy)-3 -methyl-phenyl] -propyl } benzo[b]thiophene-6-carboxylic acid (example 22 (0.10 g, 0.22 mmol) and Z)-alanine methyl ester hydrochloride salt (0.034 g,
0.24 mmol), EDCI (0.046 g, 0.24 mmol), HOBt (0.032 g, 0.24 mmol), NEt3 (0.12 mL, 0.88 mmol) and DMF (2 mL) to furnish the titled compound (0.079g, 0.15 mmol, 66%). LC/MS (m/z): calcd. for C31H4iNO5S (M+H)+: 540.3; found: 540.1.
Example 27. 7)-2-[(2-{l-Ethyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl}- benzo[b]thiophene-6-carbonyl)-amino] -propionic acid.
Using the procedure analogous to Example 5, from 7J>-2-[(2-{ l-Efhyl-l-[4-(2- ethyl-2-hydroxy-butoxy)-3 -methyl-phenyl] -propyl } -benzo [b] thiophene-6-carbonyl)- amino] -propionic acid methyl ester, example 26 (0.079g, 0.15 mmol) and NaOH (0.03g, 0.73 mmol) to furnish the titled compound (0.059 g, 0.11 mmol, 79%). LC/MS (m z): calcd. for C30H39NO5S (M+H)+: 526.3; found: 526.1.
Example 28. L-2- [(2- { 1 -Ethyl- 1 - [4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl } - benzo[b]thiophene-6-carbonyl)-amino]-propionic acid methyl ester.
Using the procedure analogous to Example 5, from 2-{ 1-ethyl- 1- [4-(2-ethyl-2- hydroxy-butoxy)-3 -methyl-phenyl] -propyl } benzo[b]thiophene-6-carboxylic acid (example 22) (0.10 g, 0.22 mmol) and 7-alanine methyl ester hydrochloride salt (0.034 g, 0.24 mmol) EDCI (0.046 g, 0.24 mmol), HOBt (0.032 g, 0.24 mmol), NEt
3 (0.12 mL, 0.88 mmol) and DMF (2 mL) to furnish the titled compound (O.lOg, 0.19 mmol, 83%). LC/MS (m/z): calcd. for C31H4
1NO
5S (M+H)
+: 540.3; found: 540.2.
Example 29. E-2-[(2-{l-Efhyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-ρhenyl]-ρroρyl}- benzo[b]thiophene-6-carbonyl)-amino]-propionic acid.
Using the procedure analogous to Example 5, from L-2-[(2-{ l-ethyl-l-[4-(2- ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl}-benzo[b]thiophene-6-carbonyl)- amino] -propionic acid methyl ester, example 28 (0.10 g, 0.19 mmol) and NaOH (0.037g, 0.93 mmol) to furnish the titled compound (0.092 g, 0.18 mmol, 95%). LC/MS (m/z): calcd. for C
30H
39NO
5S (M+H)
+: 526.3; found: 526.1.
Example 30.
2-[(2-{ l-Efhyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-proρyl}- benzo[b]thiophene-6-carbonyl)-amino]-2-methyl-propionic acid methyl ester.
Using the procedure analogous to 5, from 2-{l-efhyl-l-[4-(2-ethyl-2-hydroxy- butoxy)-3 -methyl-phenyl] -propyl} benzo[b]thioρhene-6-carboxylic acid (example 22) (0.10 g, 0.22 mmol) and α-aminoisobutyric methyl ester hydrochloride salt (0.040 g, 0.24 mmol) EDCI (0.046 g, 0.24 mmol), HOBt (0.032 g, 0.24 mmol), NEt
3 (0.12 mL, 0.88 mmol) and DMF (2 mL) to furnish the titled compound (O.lOg, 0.18 mmol, 83%). LC/MS (m/z): calcd. for C
32H
43NO
5S (M+H)
+: 554.3; found: 554.3.
Example 31. 2-[(2-{l-Ethyl-l-[4-(2-ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl}- benzo[b]thiophene-6-carbonyl)-amino]-2-methyl-propionic acid.
Using the procedure analogous to Example 5, from 2-[(2-{ 1 -Ethyl- 1-[4-(2- ethyl-2-hydroxy-butoxy)-3-methyl-phenyl]-propyl}-benzo[b]thiophene-6-carbonyl)- amino] -2-methyl-propionic acid methyl ester, example 30 (0.10 g, 0.18 mmol) and NaOH (0.036g, 0.90 mmol) to furnish the titled compound (0.088 g, 0.016 mmol, 91%). LC/MS (m/z): calcd. for C
31H
41NO
5S (M+H)
+: 540.3; found: 540.3.
Compounds of the Invention - Salts, Stereoisomers, & Prodrugs: Salts of the compounds represented by formulae IA, IB, and IC are an additional aspect of the invention. The skilled artisan will also appreciate that the family of compounds of formulae I include acidic and basic members and that the present invention includes pharmaceutically acceptable salts thereof. In those instances where the compounds of the invention possess acidic or basic functional groups various salts may be formed which are more water soluble and physiologically suitable than the parent compound. Representative pharmaceutically acceptable salts, include but are not limited to, the alkali and alkaline earth salts such as lithium, sodium, potassium, ammonium, calcium, magnesium, aluminum, zinc, and the like. Sodium and potassium salts are particularly prefened. Salts are conveniently
prepared from the free acid by treating the acid in solution with a base or by exposing the acid to an ion exchange resin. For example, a carboxylic acid substituent on the compoimd of Formula I may be selected as -CO2H and salts may be formed by reaction with appropriate bases (e.g., NaOH, KOH) to yield the conesponding sodium and potassium salt. Included within the definition of pharmaceutically acceptable salts are the relatively non-toxic, inorganic and organic base addition salts of compounds of the present invention, for example, ammonium, quaternary ammonium, and amine cations, derived from nitrogenous bases of sufficient basicity to form salts with the compounds of this invention (see, for example, S. M. Berge, et al., "Pharmaceutical Salts," J. Phar. Sci.. 66: 1-19 (1977)). Moreover, the basic group(s) of the compound of the invention may be reacted with suitable organic or inorganic acids to form salts such as acetate, benzenesulfonate, benzoate, bicarbonate, bisulfate, bitartrate, borate, bromide, camsylate, carbonate, chloride, choline, clavulanate, citrate, chloride, chloroprocaine, choline, diethanolamine, dihydrochloride, diphosphate, edetate, edisylate, estolate, esylate, ethylenediamine, fluoride, fumarate, gluceptate, gluconate, glutamate, glycolylarsanilate, hexylresorcinate, hydrabamine, bromide, chloride, hydrobromide, hydrochloride, hydroxynaphthoate, iodide, isothionate, lactate, lactobionate, laurate, malate, maleate, malseate, mandelate, meglumine, mesylate, mesviate, methylbromide, methylnitrate, methylsulfate, mucate, napsylate, nitrate, oleate, oxalate, palmitate, pamoate, pantothenate, phosphate, polygalacturonate, procane, salicylate, stearate, subacetate, succinate, sulfate, tannate, tarfrate, teoclate, tosylate, trifluoroacetate, trifluoromefhane sulfonate, and valerate. Certain compounds of the invention may possess one or more chiral centers and may thus exist in optically active forms. Likewise, when the compounds contain an alkenyl or alkenylene group there exists the possibility of cis- and trans- isomeric forms of the compounds. The R- and S- isomers and mixtures thereof, including racemic mixtures as well as mixtures of cis- and trans- isomers, are contemplated by this invention. Additional asymmetric carbon atoms can be present in a substituent group such as an alkyl group. All such isomers as well as the mixtures thereof are intended to be included in the invention. If a particular stereoisomer is desired, it can be prepared by methods well known in the art by using stereospecific reactions with starting materials
which contain the asymmetric centers and are already resolved or, alternatively by methods which lead to mixtures of the stereoisomers and subsequent resolution by known methods. For example, a chiral column may be used such as those sold by Daicel Chemical Industries identified by the trademarks:
CHIRALPAK AD, CHIRALPAK AS, CHIRALPAK OD, CHIRALPAK OJ, CHIRALPAK OA, CHIRALPAK OB, CHIRALPAK OC, CHIRALPAK OF, CHIRALPAK OG, CHIRALPAK OK, and CHIRALPAK C A- 1. By another conventional method, a racemic mixture may be reacted with a single enantiomer of some other compound. This changes the racemic form into a mixture of diastereomers. These diastereomers, because they have different melting points, different boiling points, and different solubilities can be separated by conventional means, such as crystallization. The present invention is also embodied in mixtures of compounds of formulae IA, IB, or IC . Prodrugs are derivatives of the compounds of the invention which have chemically or metabolically cleavable groups and become by solvolysis or under physiological conditions the compounds of the invention which are pharmaceutically active in vivo. Derivatives of the compounds of this invention have activity in both their acid and base derivative forms, but the acid derivative form often offers advantages of solubility, tissue compatibility, or delayed release in a mammalian organism (see, Bundgard, H., Design of Prodrugs. pp. 7-9, 21-24, Elsevier, Amsterdam 1985). Prodrugs include acid derivatives well known to practitioners of the art, such as, for example, esters prepared by reaction of the parent acidic compound with a suitable alcohol, or amides prepared by reaction of the parent acid compound with a suitable amine. Simple aliphatic or aromatic esters derived from acidic groups pendent on the compounds of this invention are prefened prodrugs. In some cases it is desirable to prepare double ester type prodrugs such as (acyloxy) alkyl esters or ((alkoxycarbonyl)oxy)alkyl esters. Particularly prefened esters to use as prodrugs are; methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, morpholinoethyl, and N,N-diethylglycolamido. N,N-diethylglycolamido ester prodrugs may be prepared by reaction of the sodium salt of a compound of Formula IA or IB (in a medium such as
dimethylformamide) with 2-chloro-N,N-diethylacetamide (available from Aldrich Chemical Co., Milwaukee, Wisconsin USA; Item No.25,099-6). Morpholinylethyl ester prodrugs may be prepared by reaction of the sodium salt of a compound of Formula IA or IB (in a medium such as dimethylformamide) 4-(2- chloroethyl)morpholine hydrochloride (available from Aldrich Chemical Co., Milwaukee, Wisconsin USA, Item No. C5,220-3). For example, prodrugs may be prepared by reaction of the sodium salt for a compound of Formula IA or IB with;
and sodium iodide to provide tthe ester prodrag pendent group
Also, lower alkyl (viz., Cι-C
8) ester prodrugs may be prepared by conventional means such as reacting the sodium or potassium salt (derived by forming the salt of any acidic compound of the invention; viz., reaction of a base such as KOH with an acidic group such as -CO
2H) of a compound of Formula IA or IB with an alkyl iodide such as methyl iodide, ethyl iodide, n-propyl iodide, isopropyl iodide. Typical ester prodrag substituents are o A Me or A -.. Et
Pharmaceutical Formulations containing the Novel Compounds of the Invention: Pharmaceutical formulations of the invention are prepared by combining (e.g., mixing) a therapeutically effective amount of the compound of the invention (compounds of Formula IA, IB, or IC ) together with a pharmaceutically acceptable
canier or diluent. The present pharmaceutical formulations are prepared by known procedures using well-known and readily available ingredients. In making the compositions of the present invention, the compounds of the invention will usually be admixed with a canier, or diluted by a canier, or enclosed within a canier which may be in the form of a capsule, sachet, paper or other container. When the carrier serves as a diluent, it may be a solid, semi-solid or liquid material which acts as a vehicle, or can be in the form of tablets, pills, powders, lozenges, elixirs, suspensions, emulsions, solutions, syrups, aerosols (as a solid or in a liquid medium), or ointment, containing, for example, up to 10% by weight of the compound. The compounds of the present invention are preferably formulated prior to administration. The compounds of the invention may also be delivered by suitable formulations contained in a transderm patch. Alternatively, the compounds of the invention may be delived to a patient by sublingual administration. For the pharmaceutical formulations any suitable canier known in the art can be used. In such a formulation, the canier may be a solid, liquid, or mixture of a solid and a liquid. Solid form formulations include powders, tablets and capsules. A solid canier can be one or more substances which may also act as flavoring agents, lubricants, solubilisers, suspending agents, binders, tablet disintegrating agents and encapsulating material. Tablets for oral administration may contain suitable excipients such as calcium carbonate, sodium carbonate, lactose, calcium phosphate, together with disintegrating agents, such as maize, starch, or alginic acid, and/or binding agents, for example, gelatin or acacia, and lubricating agents such as magnesium stearate, stearic acid, or talc. In powders the canier is a finely divided solid which is in admixture with the finely divided Active ingredient. In tablets a compound of the invention I is mixed with a canier having the necessary binding properties in suitable proportions and compacted in the shape and size desired. The powders and tablets preferably contain from about 1 to about 99 weight percent of the compound of this invention. Suitable solid caniers are magnesium carbonate, magnesium stearate, talc, sugar lactose, pectin, dextrin, starch,
gelatin, fragacanth, methyl cellulose, sodium carboxymethyl cellulose, low melting waxes, and cocoa butter. Sterile liquid form formulations include suspensions, emulsions, syrups and elixirs. The Active Ingredient may be dissolved or suspended in a pharmaceutically acceptable carrier, such as sterile water, sterile organic solvent or a mixture of both. The compounds can often be dissolved in a suitable organic solvent, for instance aqueous propylene glycol. Other compositions can be made by dispersing the finely divided compounds of the invention in aqueous starch or sodium carboxymethyl cellulose solution or in a suitable oil.
Methods of Using the Compounds of the Invention: Many disease states are benefited by treatment with the compounds of Formula
IA, IB, or IC include, but are not limited to: disease states characterized by abnormal calcium regulation disease states characterized by abnormal cell proliferation disease states characterized by abnormal cell differentiation disease states characterized by abnormal immune response disease states characterized by abnormal dermatological conditions disease states characterized by neurodegenerative condition disease states characterized by inflammation disease states characterized by vitamin D sensitivity disease states characterized by hyperproliferative disorders.
Specific disease states benefited by treatment of the compounds of Formula IA, IB and IC include, but are not limited to: Acne Actinic keratosis Alopecia Alzheimer's disease Benign prostatic hypeφlasia Bladder cancer
Bone maintenance in zero gravity Bone fracture healing Breast cancer Chemoprovention of Cancer Crohn's disease Colon cancer Type I diabetes Host-graft rejection Hypercalcemia Type II diabetes Leukemia Multiple sclerosis Myelodysplastic syndrome Insufficient sebum secretion Osteomalacia Osteoporosis Insufficient dermal firmness Insufficient dermal hydration Psoriatic arthritis Prostate cancer Psoriasis Renal osteodystrophy Rheumatoid arthritis Scleroderma Skin cancer Systemic lupus erythematosus Skin cell damage from Mustard vesicants Ulcerative colitis Vitiligo Wrinkles Particularly prefened is the freatment of psoriasis and osteoporosis by administration to a mammal (including a human) of a therapeutically effective amount
of compounds of Formulae IA, IB or IC. By "pharmaceutically effective amount" it is meant that quantity of pharmaceutical agent conesponding to formulae IA, IB, or IC which prevents, removes or reduces the deleterious effects of a disease state in mammals, including humans. The specific dose of a compound administered according to this invention to obtain therapeutic or prophylactic effects will, of course, be determined by the particular circumstances sunounding the case, including, for example, the compound administered, the route of administration and the condition being treated. Typical daily doses will contain a pharmaceutically effective amount typically in the range of from about 0.0001 mg/kg/day to about 50 mg/kg/day of body weight of an active compound of this invention. Preferably the dose of compounds of the invention will be from 0.0001 to 5 mg/kg/day of body weight. Preferably compounds of the invention or pharmaceutical formulations containing these compounds are in unit dosage form for administration to a mammal. The unit dosage form can be a capsule or tablet itself, or the appropriate number of any of these. The quantity of Active ingredient in a unit dose of composition may be varied or adjusted from about 0.0001 to about 1000 milligrams or more according to the particular treatment involved. It may be appreciated that it is necessary to make routine variations to the dosage depending on the age and condition of the patient. Dosage will also depend on the route of administration. The compounds of the inventiion may be administered by a variety of routes including oral, aerosol, rectal, transdermal, sublingual, subcutaneous, intravenous, intramuscular, and intranasal. Particularly prefened is the treatment of psoriasis with an ointment type formulation containing the compounds of the invention. The ointment formulation may be applied as needed, typically from one to 6 times daily. Treatment of psoriasis is preferably done with topical application by a formulation in the form of a cream, oil, emulsion, paste or ointment containing a therapeutically effective amount of a compound of the invention. The formulation for topical freatment contains from 0.5 to 0.00005 weight percent, preferably from .05 to 0.0005 weight percent, and most preferably from 0.025 to 0.001 of a Active Ingredient. For example, two semisolid topical preparations useful as vehicles for NDR modulators in treatment and prevention of psoriasis are as follows:
Polvethylene Glvcol Ointment USP (p. 2495) Prepare Polyethylene Glycol Ointment as follows: Polyethylene Glycol 3350 400 g. Polyethylene Glycol 400 600 g. To make 1000 g. Heat the two ingredients on a water bath to 65C. Allow to cool, and stir until congealed. If a firmer preparation is desired, replace up to 100 g of the polyethylene glycol 400 with an equal amount of polyethylene glycol 3350. Hydrophilic Ointment USP (p. 1216) Prepare Hydrophilic Ointment as follows: Methylparaben 0.25 g. Propylparaben 0.15 g. Sodium Lauryl Sulfate 10 g. Propylene Glycol 120 g. Stearyl Alcohol 250 g. White Petrolatum 250 g. Purified Water 370 g. To make about 1000 g. The Stearyl Alcohol and White Petrolatum are melted on a steam bath, and warmed to about 75C. The other ingredients, previously dissolved in the water are added, warmed to 75C, and the mixture stined until it congeals. For each of the above formulations the Active Ingredient is added during the heating step in an amount that is from 0.5 to 0.00005 weight percent, preferably from .05 to 0.0005 weight percent, and most preferably from 0.025 to 0.001 weight percent of the total ointment weight. (Source: - United States Pharmacopoeia 24, United States Pharmacopeial Convention, 1999)
Conventional therapy for osteoporosis includes; (i) estrogens, (ii) androgens, (iii) calcium supplements, (iv) vitamin D metabolites, (v) thiazide diuretics, (vi) calcitonin, (vii) bisphosphonates, (viii) SERMS, and (ix) fluorides (see, Hanison's Principles of Internal Medicine, 13th edition, 1994, published by McGraw Hill Publ., ISBN 0-07-
032370-4, pgs.2172-77; the disclosure of which is incorporated herein by reference.). Any one or combination of these conventional therapies may be used in combination with the method of treatment using compounds of Formulae IA, IB, or IC as taught herein. For example, in a method of treating osteoporosis, the vitamin D receptor modulator compounds of the invention may be administered separately or simultaneously with a conventional therapy. Alternatively, the vitamin D receptor modulator compounds of the invention may be combined with conventional therapeutic agents in a formulation for treatment of osteoporosis such as set out below: A formulation for treating osteoporosis comprising: Ingredient (Al): a vitamin D receptor modulator represented by formula (IA), (IB), or (IC), or a pharmaceutically acceptable salt or prodrag derivative thereof; Ingredient (BI): one or more co-agents that are conventional for treatment osteoporosis selected from the group consisting of: a. estrogens, b. androgens, c. calcium supplements, d. vitamin D metabolites, e. thiazide diuretics, f. calcitonin, g. bisphosphonates, h. SERMS, and i. fluorides. Ingredient (CI): optionally, a canier or diluent. Typically useful formulations are those wherein the weight ratio of (Al) to (BI) is from 10:1 to 1:1000 and preferably from 1:1 to 1:100.
Combination Therapy for Psoriasis: Conventional therapy for psoriasis includes topical glucocorticoids, salicylic acid, crade coal tar, ultraviolet light, and methotrexate (see, Hanison's Principles of Internal Medicine, 13th edition, 1994, published by McGraw Hill Publ., ISBN 0-07-032370-4,
pgs.2172-77). Any one or combination of these conventional therapies may be used in combination with the method of freatment using compounds of Formulae IA, IB, or IC as taught herein. For example, in a method of treating osteoporosis, the vitamin D receptor modulator compounds of the invention (e.g., as defined by Formulae IA, IB or IC) may be topically administered separately or simultaneously with a conventional therapy. Alternatively, the vitamin D receptor modulator compounds of the invention may be combined with conventional therapeutic agents in a topically applied formulation for treatment of osteoporosis such as set out below: A formulation for treating psoriasis comprising: Ingredient (A2): a vitamin D receptor modulator represented by formula (IA), (IB), or (IC), or a pharmaceutically acceptable salt or prodrag derivative thereof; Ingredient (B2): one or more co-agents that are conventional for treatment psoriasis selected from the group consisting of: a. topical glucocorticoids , b. salicylic acid, or c. crade coal tar. Ingredient (C2): optionally, a canier or diluent.
Typically useful formulations are those wherein the weight ratio of (A2) to (B2) is from 1:10 to 1:100000 and preferably from 1:100 to 1:10000.
Experimental Results:
Table 1 Summary of Experimental Results
Table 2 Summary of Experimental Results
Table 3 of Experimental Results (comparison compounds)
Table 4 of Experimental Results (comparison compounds)
Explanation of Table 5 and 6 column numerical superscripts: 1. Test Compound numbers refer to the products of the conesponding Example Nos. that is, compounds within the scope of the invention 2. The control experiments are done with the double letter coded compounds identified as follows: "AA" = lα,25-dihydroxyvitamin D3 "BB" = 3-(4-{ l-Ethyl-l-[4-(2-hydroxy-3,3-dimethyl-butoxy)-3-methyl-ρhenyl]- propyl } -2-methyl-phenoxy)-propane- 1 ,2-diol
"CC" = l-(4-{ l-[4-(3,3-Dimethyl-2-oxo-butoxy)-3-methyl-phenyl]-cyclohexyl}-2- methyl-phenoxy)-3,3-dimethyl-butan-2-one
'DD" = compound represented by the formula:
"EE" = compound represented by the formula:
"FF" -= calcipofriol (structural formula below):
Chiral
2. The RXR-NDR heterodimerization (SaOS-2 cells) test is described in the "Assay" section of the Description, infra. 3. The VDR CTF (Caco-2 cells) test is described in the "Assay" section of the Description, infra. 4. The OCΝ Promoter test is described in the "Assay" section of the Description, infra. 5. The Mouse Hypercalcemia test is described in the "Assay" section of the Description, infra. 6. The keratinocyte proliferation assay is described in the "Assay" section of the Description, infra. 7. The IL-10 induction assay is described in the "Assay" section of the Description, infra.
Assay Methods Use of the Assay Methods: The evaluation of the novel compounds of the invention for osteoporosis and other related diseases is done using a plurality of test results. The use of multiple assays is necessary since the combined properties of (i) high activity for the vitamin D receptor, and (ii) prevention of hypercalcemia must be achieved to have utility for the methods of treating diseases, which are also, aspects of this invention. Some of the tests described below are believed related to other tests and measure related properties of compounds. Consequently, a compound may be considered to have utility in the practice of the invention if is meets most, if not all, of the acceptance criteria for the above described tests. The evaluation of the novel compounds of the invention for psoriasis is done using the Keratinocyte Proliferation Assay in combination with other assays that measure
inhibition of IL-2 production and stimulation of IL-10 production in peripheral blood mononuclear cells (PBMCs).
Brief Description. Utility and Acceptance Criteria for the Assay Methods:
1. The RXR- VDR heterodimerAssav: This assay provides the VDR activity of a test compound. It is desirable to have low EC50 values for a compound in this assay. The lower the EC50 value, the more active the compound will be as a VDR agonist. Desired assay results are EC50 values less than or equal to 600 nM. Prefened assay results are less than 250 nM, and most preferably less than 150 nM.
2. The Caco-2 cell Co-transfection Assay: The Caco-2 cell assay is an indicator for the undesirable condition of hypercalcemia. This co-transfection assay is a sunogate assay for in vivo calcemic activity of VDR ligands. It is desirable to have high EC50 values for a test compound in this assay. The higher the EC50 values for a compound the less calcemic it will be in vivo. Desired assay results are EC50 greater than or equal to 300 nM. Prefened assay results are greater than 1000 nM.
3. The OCN (osteocalcin Promoter Assay The OCN Promoter Assay is an indicator and marker for osteoporosis. Desired assay results are EC50 less than or equal to 325 nM. Prefened assay results are less than 50 nM.
4. The Mouse Hypercalcemia Assay The Mouse Hypercalcemia Assay is a six day hypercalcemia test for toxicity and selectivity. Acceptable test results are levels greater than 300 μg/kg/day. Preferred assay results are levels greater than 1000 μg/kg/day.
5. The Keratinocyte Proliferation Assay
This Assay is indicative for the treatment of psoriasis. An acceptable test result is IC50 value of less than or equal to 300 nM. Prefened assay results are IC50 values of less than 100 nM.
6. The IL-10 induction Assay
This is an in vitro efficacy assay for psoriasis, abscess and adhesion. Psoriasis involves both keratinocytes and immune cells. IL-10 is a unique cytokine because it is anti- inflammatory and immunosuppressive. This assay tells us whether a VDRM is able to function as an agonist in PBMCs (primary blood mononuclear cells) or not. A lower EC50 value is desirable in this assay since a compound with a lower EC50 value will be a better agonist in PBMCs. An acceptable test result is an EC50 value of less than 200 nM. Prefened assay results are EC50 values of less than 100 nM.
7. Other Compound Assay Standards
An alternative measure of the therapeutic index (bone efficacy vx. Hypervcalcemia) of compounds of the invention for treatment of osteoporosis is a numerical ratio calculated as follows: Dose Threshold needed to induce hypercalcemia divided by Dose Threshold needed for bone efficacy
An alternative measusre of the therapeutic index (in vivo keratinocyte proliferation vs. hypercalcemia) of compounds of the invention for freatment of psoriasis is a numerical ratio calculated as follows: Dose Threshold needed to induce hypercalcemia divided by Dose Threshold needed to induce keratinocyte proliferation For the above ratios, Dose Thresholds are determined from dose response curve data.
8. The CaTl (calcium transporter 1) Assay \
The CaTl Assay is an indicator for the undesirable condition of hypercalcemia. The higher the EC50 values for a compound the less calcemic it will be in vivo. Desired
assay results are EC50 greater than or equal to 500 nM. Prefened assay results are greater than 1000 nM.
Details of the Assay Methods: (1) Materials and Method for RXR-VDR Heterodimerization Assay: Transfection Method:
• FuGENE 6 Transfection Reagent (Roche Cat # 1 814443 ) Growth Media:
• D-MEM High Glucose (Gibco BRL Cat # 11054-020), 10% FBS, 1% antibiotic- antimycotic (Ab-Am)
FBS heat inactivated (Gibco BRL Cat # 10092-147 )
Ab-Am (Gibco BRL Cat # 15240-062 )
Cells:
• Grow SaOs-2 cells in T-152 cm culture flasks in growth media.
• Keep the density at 5-6 x 105 cells/ml
• Passage cells 1:3 twice a week
• Add Trypsin EDTA (Gibco BRL Cat # 25300-020)and incubate
• Resuspend cells in plating media and transfer into growth media. Wash Media:
• HBSS Low Glucose Without Phenol Red (Gibco BRL Cat # 14175-095), 1% Ab-Am Plating Media:
• D-MEM Low Glucose Witliout Phenol Red (Gibco BRL Cat # 11054-020), 1% Ab-Am D-MEM
Stripped FBS (Hyclone Cat# SH30068.03 Lot # AHM9371 )
Ab-Am
Transfection / Treatment Media:
• D-MEM Low Glucose Without Phenol Red only T-152 cm2 culture flask:
• Use Coming Coastar T-152 cm2 culture flask (Cat # 430825) to grow the cells Flat well Plates:
• Use well plate to plate cells
• Use Deep well plate sterile to make up treatment media.
Luciferase Assay Reagent:
• Use Steady-Glo Luciferase Reagent from Promega (Cat # E2550) Consists of: a. E2533 Assay Substrate, lyopholized product and b. E2543 Assay Buffer.
• Thaw at room temperature
• Store
Cell Harvesting
Aspirate media from culture flask, rinse cells with HBSS and aspirate.
Add trypsin and incubate.
When cells appear detached, resuspend cells in growth media.
Transfer into a new flask with fresh growth media for passaging the cells.
Plate well plates and two extra plates
A. Cell Count
Mix the cell suspension using pipette
Use Hematocytometer to count the cells
Load cell suspension onto the hemocytometer chamber
Count cells.
Plate seeding:
Use plating media 10 % Stripped FBS in D-MEM Low Glucose, Without Phenol Red, 1%
Ab-Am
Plate 14 plates @ 165 μl / well.
In sterile flask add cell suspension to plating media.
Mi .
Add cells / well.
Place the cells in the incubator.
Cells should be about 75 % confluent prior to transfection.
DAY 2: Transfection
Step 1: DNA and Media
Add plain DMEM media to tubes for mixing the DNA
Add the Reporter gene pFR-LUC Add the Gal4-RXR-DEF and VP16-VDR-LBD
Step 2: FuGENE and Media Prepare plain DMEM media in a ubes for mixing FuGENE Add FuGENE 6 Transfection Reagent Incubate
Step 3: FuGENE , DNA and Media Complex Add FuGENE Media complex from step 2 to DNA Media complex from stepl Incubate
Step 4: FuGENE , DNA and Media Complex to-well plate Add FuGENE-DNA-Media complex from step 3 to each plate Incubate.
Day 3: Dosing
Treatment preparation Allow for transfection time
Make a stock solution of the compounds in DMSO
Vortex until all the compounds has been dissolved.
Further dilute in D-MEM (Low Glucose - With out Phenol Red)
Add compounds in quadruplicate to give final volume
Incubate.
Day 4: Luciferase Assay
Read the plates after drag treatment
Remove part of media from all the wells and leave remainder
Add Steady-Glo Luciferase Reagent mixture / wells
Incubate
Count each well using a Luminescence counter, Top Count NXT by Packard Set a delay between plates to reduce the background.
(2) Materials and Method for The Caco-2 Cell Assay: Caco-2 cells, grown in phenol red free, DMEM (Invitrogen, Carlsbad, CA) containing 10 % charcoal-stripped FCS (Hyclone, Logan, UT), were transfected with Fugene 6 reagent (Roche Diagnostics, Indianapolis, IN). Cells (5000/well) were plated 18 h before transfection in a 96 well plate. The Cells were transfected with Gal4-responsive reporter pFRLuc (150 ng, Stratagene, La Jolla CA) and the receptor expression vector pGal4-VDR-LBD (10 ng), along with Fugene 6 reagent (0.2 μl/well). The DNA-Fugene complex was formed by incubating the mixture for 30 min at room temperature. The cells were transfected in triplicate for 5 h, and treated with various concentrations of VDR ligands (form 0.01 nM to 10,000 nM concentration range) 18h post-transfection. The luciferase activity was quantified using Steady-Glo reagent kit (Promega, Madison, WI) as per manufacturer's specifications.
(3) Materials and Method for The OCN Promoter Assay:
The activation of osteocalcin by VDR ligands was evaluated in a rat osteoblast-like cell line RG-15 (ROS 17/2.8) stably expressing rat osteocalcin promoter fused with luciferase reporter gene. The stable cell lines were established as reported before (Activation of Osteocalcin Transcription involves interaction of protein kinase A- and Protein kinase C-dependent pathways. Boguslawski, G., Hale, L. V., Yu, X.-P., Miles, R. R., Onyia, J. E., Santene R. F., Chandrasekhar, S. J Biol. Chem. 275, 999-1006, 2000). Confluent RG-15 cells maintained in DMEM/F-12 medium (3:1) containing 5% FBS, 300 μg/ml G418 and at 3,7°C under 5% CO2/95% air atmosphere were trypsinized (0.25% trypsin) and plated into white opaque 96-well cell culture plates (25000 cells/well). After 24 hr, cells (in DMEM/F-12 medium + 2% FBS) were treated with various concentrations of compounds, dissolved in DMSO. The final DMSO concentration remained at 0.01% (v/v). After 48 hr freatment, the medium was removed, cells were lysed with 50 μl of lysis buffer (From Luciferase reporter assay system, Roche Diagnostics, Indianapolis, IN) and assayed for luciferase activity using the Luciferase Reporter Gene Assay kit from Boehringer Mannheim as per manufacturer's specifications.
(4) Materials and Method for The Mouse Hypercalcemia Assay: Weanling, virus -antibody-free, five to six weeks old female DBF mice (Harlan, Indianapolis, IN) are used for all the studies. Animals are allowed to acclimate to local vivarium conditions for 2 days. Mice are maintained on a 12 hr light/dark cycle at 22°C with ad lib access to food (TD 5001 with 1.2% Ca and 0.9%P, Teklad, Madison, WI) and water. The animals then are divided into groups with 4-5 mice per group. Different doses of test compounds prepared in 10% Ethanol and 90% sesame oil are administered to mice orally via gavage for 6 days. lα-25(OH) D3 0.5μg/kg/d was also given to one group of mice as the positive control. Seram ionized calcium is evaluated at 6 hours after the last dosing under isoflurane anesthesia by Ciba-Coming Ca++/PH Analyzer, (Model 634, Chiron Diagnostics Corp., East Walpole, MA). Raw data of group differences is assessed by analysis of variance (ANOVA) using Fisher's protected least significant difference (PLSD) where the significance level was P< 0.05.
(5) The Keratinocyte Proliferation Assay: KERtr cells (Human skin keratinocyte transformed with a retrovirus vector, obtained from ATCC) were plated in 96-well flat-bottomed plates (3000 cells/well) in 100 μl keratinocyte seram free medium supplemented with bovine pituitary extract in the absence of EGF (Life Technologies, Rockville, MD) and incubated at 37°C for two days. The cells were treated with various concentrations of VDR ligands (ten-fold serial dilution from 10,000 nM to 0.1 nM in triplicate), dissolved in 100 μl keratinocyte serum free medium supplemented with bovine pituitary extract in the absence of EGF and incubated at 37°C for 72hr. BrdU (5-bromo-2'-deoxyuridine) incorporation was analyzed as a measure of DNA replication (Cell proliferation ELISA kit, Roche Diagnostics, Indianapolis, IN) and absorbance was measured at 405 nm. Potency values (IC50) values were determined as the concentration (nM) of compound that elicited a half -maximal response.
(6) Materials and Method for human IL-10 Induction Assay: Isolation of peripheral blood mononuclear cells (PBMCs): A. Collect 50 ml of human blood and dilute with media, RPMI- 1640. B. Prepare sterile tubes with ficol.
C. Add diluted blood to tubes. D. Centrifuge. E. Discard the top layer and collect the cells from middle layer. F. Divide all cells into four tubes and add media. G. Centrifuge. H. Aspirate off media and resuspend. I. Collect all cells J. Centrifuge, at 1200 rpm for 10 minutes. K. Resuspend in RPMI-1640 with 2% FBS and count cells Stimulation of PBMC: L. Prepare TPA in DMSO. M. Dissolve PHA in water . N. Plate TPA/PHA treated PBMCs in well plates. O. Incubate. Treatment: P. Prepare all compound dilutions in plain RPMI- 1640 media. Q. Add diluted compound. R. Incubate. Sample Collection and assay: S. Remove all the cells by centrifugation and assay the supernatant for IL-10 by immunoassay. T. Perform IL-10 assay using anti-human IL-10 antibody coated beads, as described by the manufacturer (Linco Research Inc., St. Charles, MO). (8) CaTl assay Human colon carcinoma, Caco-2 cells, maintained in DMEM (high glucose with 25 mM Hepes buffer; Invitrogen, Carlsbad, CA) supplemented with 10 % fetal bovine seram (Invitrogen, Carlsbad, CA), are plated at 5500 cell per well in a 96-well plate in a total volume of 100 μl/well. The cells are kept in the 96-well plate for 6 days to differentiate them to small intestinal cells that express the calcium transporter, CaTl. On day 3 after plating, old media is removed and replaced with fresh media (150 μl/well). On day 6 the old media is removed and the cells are kept in freatment media (180 μl/well)
that contained 10 % charcoal stripped fetal bovine seram (Hyclone, Logan, UT) in DMEM (low glucose, without phenol red; Invitrogen, Carlsbad, CA). The cells are treated with various concentrations of VDR ligands (from 0.01 nM to 10,000 nM concentration range) prepared in freatment media (20 μl/well). Twenty hours post-treatment, total RNA is prepared by RNeasy 96 method as described by the manufacturer (Qiagen, Valencia, CA). The RNA is reverse transcribed and amplified for human CaTl and GAPDH (control) messages by quantitative RT-PCR using ABI PRISM 7900HT Sequence Detection System according to manufacturer's instructions (Applied Biosystems, Foster City, CA). Optimized primer pairs and probes for human CaTl and GAPDH genes are obtained commercially (Applied Biosystems, Foster City, CA). Each 20 μl quantitative RT-PCR reaction in a 384-well Taqman PCR plate consists of forward and reverse primers (900 nM), Taqman probe (200 nM), total RNA (4 μl form each well of the 96- well culture plate) and 10 μl of Taqman Universal PCR Master Mix (Roche Diagnostics, Indianapolis, IN). Reactions are incubated at 48°C for 30 minutes, followed by 10 minutes at 95°C and subjected to 40 cycles of PCR (95°C for 15 seconds followed by 60°C for 1 minute). GAPDH is used as an internal confrol and its primer and probe set are obtained commercially (Applied Biosystems, Foster City, CA).