WO2005043162A2 - Method for distinguishing mll-ptd-positive aml from other aml subtypes - Google Patents
Method for distinguishing mll-ptd-positive aml from other aml subtypes Download PDFInfo
- Publication number
- WO2005043162A2 WO2005043162A2 PCT/EP2004/012464 EP2004012464W WO2005043162A2 WO 2005043162 A2 WO2005043162 A2 WO 2005043162A2 EP 2004012464 W EP2004012464 W EP 2004012464W WO 2005043162 A2 WO2005043162 A2 WO 2005043162A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- numbers
- expression
- polynucleotide
- ptd
- polynucleotide defined
- Prior art date
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/574—Immunoassay; Biospecific binding assay; Materials therefor for cancer
- G01N33/57407—Specifically defined cancers
- G01N33/57426—Specifically defined cancers leukemia
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6876—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes
- C12Q1/6883—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material
- C12Q1/6886—Nucleic acid products used in the analysis of nucleic acids, e.g. primers or probes for diseases caused by alterations of genetic material for cancer
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/112—Disease subtyping, staging or classification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q2600/00—Oligonucleotides characterized by their use
- C12Q2600/158—Expression markers
Definitions
- the present invention is directed to a method for distinguishing MLL-PTD-positive AML from other AML subtypes by determining the expression level of selected marker genes.
- Leukemias are classified into four different groups or types: acute myeloid (AML), acute lymphatic (ALL), chronic myeloid (CML) and chronic lymphatic leukemia (CLL). Within these groups, several subcategories can be identified further using a panel of standard techniques as described below. These different subcategories in leukemias are associated with varying clinical outcome and therefore are the basis for different treatment strategies. The importance of highly specific classification may be illustrated in detail further for the AML as a very heterogeneous group of diseases. Effort is aimed at identifying biological entities and to distinguish and classify subgroups of AML which are associated with a favorable, intermediate or unfavorable prognosis, respectively.
- the FAB classification was proposed by the French- American-British co-operative group which was based on cytomorphology and cytochemistry in order to separate AML subgroups according to the morphological appearance of blasts in the blood and bone marrow.
- genetic abnormalities occurring in the leukemic blast had a major impact on the morphological picture and even more on the prognosis.
- the karyotype of the leukemic blasts is the most important independent prognostic factor regarding response to therapy as well as survival.
- leukemia diagnostics Analysis of the morphology and cytochemistry of bone marrow blasts and peripheral blood cells is necessary to establish the diagnosis. In some cases the addition of immunophenotyping is mandatory to separate very undifferentiated AML from acute lymphoblastic leukemia and CLL. Leukemia subtypes investigated can be diagnosed by cytomorphology alone, only if an expert reviews the smears. However, a genetic analysis based on chromosome analysis, fluorescence in situ hybridization or RT-
- PCR and immunophenotyping is required in order to assign all cases in to the right category.
- the aim of these techniques besides diagnosis is mainly to determine the prognosis of the leukemia.
- a major disadvantage of these methods, however, is that viable cells are necessary as the cells for genetic analysis have to divide in vitro in order to obtain metaphases for the analysis.
- Another problem is the long time of 72 hours from receipt of the material in the laboratory to obtain the result.
- great experience in preparation of chromosomes and even more in analyzing the karyotypes is required to obtain the correct result in at least 90% of cases.
- CML chronic myeloid leukemia
- CLL chronic lymphatic
- ALL acute lymphoblastic
- AML acute myeloid leukemia
- the new therapeutic drug inhibits the CML specific chimeric tyrosine kinase BCR-ABL generated from the genetic defect observed in
- the technical problem underlying the present invention was to provide means for leukemia diagnostics which overcome at least some of the disadvantages of the prior art diagnostic methods, in particular encompassing the time-consuming and unreliable combination of different methods and which provides a rapid assay to unambiguously distinguish one AML subtype from another, e.g. by genetic analysis.
- WO-A 03/039443 discloses marker genes the expression levels of which are characteristic for certain leukemia, e.g. AML subtypes and additionally discloses methods for differentiating between the subtype of AML cells by determining the expression profile of the disclosed marker genes.
- WO-A 03/039443 does not provide guidance which set of distinct genes discriminate between two subtypes and, as such, can be routineously taken in order to distinguish one AML subtype from another.
- the problem is solved by the present invention, which provides a method for distinguishing MLL-PTD-positive AML from other AML subtypes in a sample, the method comprising determining the expression level of markers selected from the markers identifiable by their Affymetrix Identification Numbers (affy id) as defined in Tables 1, 2, and/or 3, wherein a lower expression of at least one polynucleotide defined by any of the numbers 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, and/or 50 of Table 1 is indicative for the presence of PTD (MLL-PTD-positive AML with normal karyotype) when PTD is distinguished from AML_NK (MLL- PTD-negative AML with normal karyotype), and/or wherein a lower expression of at least one polynucleotide defined by
- tMLL AML with translocations in the MLL gene (t(l Iq23)/MLL)
- PTD AML with normal karyotype and Partial Tandem Duplication (PTD) within the MLL gene (MLL-PTD)
- AMLJSfK AML with normal karyotype (no Partial Tandem Duplication (PTD) within the MLL gene) t(8;21)
- all other subtypes refer to the subtypes of the present invention, i.e. if one subtype is distinguished from “all other subtypes", it is distiguished from all other subtypes contained in the present invention.
- a sample means any biological material containing genetic information in the form of nucleic acids or proteins obtainable or obtained from an individual.
- the sample includes e.g. tissue samples, cell samples, bone marrow and/or body fluids such as blood, saliva, semen.
- the sample is blood or bone marrow, more preferably the sample is bone marrow.
- a general method for isolating and preparing nucleic acids from a sample is outlined in Example 3.
- the term "lower expression” is generally assigned to all by numbers and Affymetrix Id. definable polynucleotides the t- values and fold change (fc) values of which are negative, as indicated in the Tables. Accordingly, the term “higher expression” is generally assigned to all by numbers and Affymetrix Id. definable polynucleotides the t-values and fold change (fc) values of which are positive.
- the term "expression” refers to the process by which mRNA or a polypeptide is produced based on the nucleic acid sequence of a gene, i.e.illerexpression" also includes the formation of mRNA upon transcription.
- the term determining the expression level preferably refers to the determination of the level of expression, namely of the markers.
- markers refers to any genetically controlled difference which can be used in the genetic analysis of a test versus a control sample, for the purpose of assigning the sample to a defined genotype or phenotype.
- markers refer to genes which are differentially expressed in, e.g., different AML subtypes.
- the markers can be defined by their gene symbol name, their encoded protein name, their transcript identification number (cluster identification number), the data base accession number, public accession number or GenBank identifier or, as done in the present invention, Affymetrix identification number, chromosomal location, UniGene accession number and cluster type, LocusLink accession number (see Examples and Tables).
- the Affymetrix identification number (affy id) is accessible for anyone and the person skilled in the art by entering the "gene expression omnibus" internet page of the National Center for Biotechnology Information (NCBI) (http://www.ncbi.nlm.nih.gov/geo/).
- NCBI National Center for Biotechnology Information
- the affy id's of the polynucleotides used for the method of the present invention are derived from the so-called U133 chip.
- the expression level of a marker is determined by the determining the expression of its corresponding "polynucleotide" as described hereinafter.
- the term “bigpolynucleotide” refers, generally, to a DNA, in particular cDNA, or RNA, in particular a cRNA, or a portion thereof or a polypeptide or a portion thereof.
- the polynucleotide is formed upon transcription of a nucleotide sequence which is capable of expression.
- the polynucleotide fragments refer to fragments preferably of between at least 8, such as 10, 12, 15 or 18 nucleotides and at least 50, such as 60, 80, 100, 200 or 300 nucleotides in length, or a complementary sequence thereto, representing a consecutive stretch of nucleotides of a gene, cDNA or mRNA.
- polynucleotides include also any fragment (or complementary sequence thereto) of a sequence derived from any of the markers defined above as long as these fragments unambiguously identify the marker.
- the determination of the expression level may be effected at the transcriptional or translational level, i.e. at the level of mRNA or at the protein level.
- Protein fragments such as peptides or polypeptides advantageously comprise between at least 6 and at least 25, such as 30, 40, 80, 100 or 200 consecutive amino acids representative of the corresponding full length protein. Six amino acids are generally recognized as the lowest peptidic stretch giving rise to a linear epitope recognized by an antibody, fragment or derivative thereof.
- the proteins or fragments thereof may be analysed using nucleic acid molecules specifically binding to three-dimensional structures (aptamers).
- the determination of the expression levels may be effected by a variety of methods.
- the polynucleotide, in particular the cRNA is labelled.
- the labelling of the polynucleotide or a polypeptide can occur by a variety of methods known to the skilled artisan.
- the label can be fluorescent, chemiluminescent, bioluminescent, radioactive (such as 3 H or 32 P).
- the labelling compound can be any labelling compound being suitable for the labelling of polynucleotides and/or polypeptides. Examples include fluorescent dyes, such as fluorescein, dichlorofluorescein, hexachlorofluorescein, BODIPY variants, ROX, tetramethylrhodamin, rhodamin X, Cyanine-2, Cyanine-3, Cyanine-5, Cyanine-7,
- IRD40 FluorX, Oregon Green, Alexa variants (available e.g. from Molecular Probes or Amersham Biosciences) and the like, biotin or biotinylated nucleotides, digoxigenin, radioisotopes, antibodies, enzymes and receptors.
- the detection is done via fluorescence measurements, conjugation to streptavidin and/or avidin, antigen-antibody- and/or antibody- ntibody- interactions, radioactivity measurements, as well as catalytic and or receptor/ligand interactions.
- Suitable methods include the direct labelling (incorporation) method, the amino-modified (amino-allyl) nucleotide method (available e.g.
- DNA dendrimer labelling as kit available e.g. from Genisphere
- biotin or biotinylated nucleotides for labelling are directly incorporated into, e.g. the cRNA polynucleotide by in vitro transcription.
- cDNA may be prepared into which a detectable label, as exemplified above, is incorporated. Said detectably labelled cDNA, in single-stranded form, may then be hybridised, preferably under stringent or highly stringent conditions to a panel of single-stranded oligonucleotides representing different genes and affixed to a solid support such as a chip. Upon applying appropriate washing steps, those cDNAs will be detected or quantitatively detected that have a counterpart in the oligonucleotide panel.
- the mRNA or the cDNA may be amplified e.g.
- the cDNAs are transcribed into cRNAs prior to the hybridisation step wherein only in the transcription step a label is incorporated into the nucleic acid and wherein the cRNA is employed for hybridisation.
- the label may be attached subsequent to the transcription step.
- proteins from a cell or tissue under investigation may be contacted with a panel of aptamers or of antibodies or fragments or derivatives thereof.
- the antibodies etc. may be affixed to a solid support such as a chip. Binding of proteins indicative of an AML subtype may be verified by binding to a detectably labelled secondary antibody or aptamer.
- a detectably labelled secondary antibody or aptamer For the labelling of antibodies, it is referred to
- a minimum set of proteins necessary for diagnosis of all AML subtypes may be selected for creation of a protein array system to make diagnosis on a protein lysate of a diagnostic bone marrow sample directly.
- Protein Array Systems for the detection of specific protein expression profiles already are available (for example: Bio-Plex, BIORAD, Munchen, Germany).
- antibodies against the proteins have to be produced and immobilized on a platform e.g. glasslides or microtiterplates.
- the immobilized antibodies can be labelled with a reactant specific for the certain target proteins as discussed above.
- the reactants can include enzyme substrates, DNA, receptors, antigens or antibodies to create for example a capture sandwich irnmunoassay.
- the expression of more than one of the above defined markers is determined.
- the statistical significance of markers as expressed in q or p values based on the concept of the false discovery rate is determined. In doing so, a measure of statistical significance called the q value is associated with each tested feature.
- the q value is similar to the p value, except it is a measure of significance in terms of the false discovery rate rather than the false positive rate (Storey JD and Tibsbirani R. Proc.Natl.Acad.Sci., 2003, Vol. 100:9440-5.
- markers as defined in Table 1.1-3.15 having a q- value of less than 3E-03, more preferred less than 1.5E-09, most preferred less than 1.5E-11, less than 1.5E-20, less than 1.5E-30, are measured.
- the expression level of at least 2, of at least 5, of at least 10 out of the markers having the numbers 1 - 10, 1-20, 1-40, 1-50 of at least one of the Tables are measured.
- the level of the expression of the handheldmarker i.e. the expression of the polynucleotide is indicative of the AML subtype of a cell or an organism.
- the level of expression of a marker or group of markers is measured and is compared with the level of expression of the same marker or the same group of markers from other cells or samples. The comparison may be effected in an actual experiment or in silico.
- expression level also referred to as expression pattern or expression signature (expression profile)
- the difference at least is 5 %, 10% or 20%, more preferred at least 50% or may even be as high as 75% or 100%. More prefened the difference in the level of expression is at least 200%, i.e. two fold, at least 500%, i.e. five fold, or at least 1000%, i.e. 10 fold.
- the expression level of markers expressed lower in a first subtype than in at least one second subtype, which differs from the first subtype is at least
- the expression level of markers expressed higher in a first subtype than in at least one second subtype, which differs from the first subtype is at least 5 %, 10% or 20%, more preferred at least 50% or may even be 75% or 100%, i.e. 2-fold higher, preferably at least 10-fold, more preferably at least 50-fold, and most preferably at least 100-fold lower in the first subtype.
- the expression level of markers expressed higher in a first subtype than in at least one second subtype, which differs from the first subtype is at least 5 %, 10% or 20%, more preferred at least 50% or may even be 75% or 100%, i.e. 2-fold higher, preferably at least 10-fold, more preferably at least 50-fold, and most preferably at least 100-fold higher in the first subtype.
- the sample is derived from an individual having leukaemia, preferably AML.
- the polynucleotide the expression level of which is determined is in form of a transcribed polynucleotide.
- a particularly preferred transcribed polynucleotide is an mRNA, a cDNA and/or a cRNA, with the latter being preferred.
- Transcribed polynucleotides are isolated from a sample, reverse transcribed and/or amplified, and labelled, by employing methods well-known the person skilled in the art (see Example 3).
- the step of determining the expression profile further comprises amplifying the transcribed polynucleotide.
- the method comprises hybridizing the transcribed polynucleotide to a complementary polynucleotide, or a portion thereof, under stringent hybridization conditions, as described hereinafter.
- hybridizing means hybridization under conventional hybridization conditions, preferably under stringent conditions as described, for example, in Sambrook, J., et al., in "Molecular Cloning: A Laboratory Manual” (1989), Eds. J. Sambrook, E. F. Fritsch and T. Maniatis, Cold Spring Harbour Laboratory Press,
- Such conditions are, for example, hybridization in 6x SSC, pH 7.0 / 0.1% SDS at about 45°C for 18-23 hours, followed by a washing step with 2x SSC/0.1% SDS at 50°C.
- the salt concentration in the washing step can for example be chosen between 2x SSC/0.1% SDS at room temperature for low stringency and 0.2x SSC/0.1% SDS at 50°C for high stringency.
- the temperature of the washing step can be varied between room temperature, ca. 22°C, for low stringency, and 65°C to 70° C for high stringency.
- polynucleotides that hybridize at lower stringency hybridization conditions. Changes in the stringency of hybridization and signal detection are primarily accomplished through the manipulation, preferably of formamide concentration
- washes performed following stringent hybridization can be done at higher salt concentrations (e.g. 5x SSC).
- Variations in the above conditions may be accomplished through the inclusion and/or substitution of alternate blocking reagents used to suppress background in hybridization experiments.
- the inclusion of specific blocking reagents may require modification of the hybridization conditions described above, due to problems with compatibility.
- “Complementary” and “complementarity”, respectively, can be described by the percentage, i.e. proportion, of nucleotides which can form base pairs between two polynucleotide strands or within a specific region or domain of the two strands.
- complementary nucleotides are, according to the base pairing rules, adenine and thymine (or adenine and uracil), and cytosine and guanine.
- Complementarity may be partial, in which only some of the nucleic acids' bases are matched according to the base pairing rules. Or, there may be a complete or total complementarity between the nucleic acids. The degree of complementarity between nucleic acid strands has effects on the efficiency and strength of hybridization between nucleic acid strands.
- Two nucleic acid strands are considered to be 100% complementary to each other over a defined length if in a defined region all adenines of a first strand can pair with a thymine (or an uracil) of a second strand, all guanines of a first strand can pair with a cytosine of a second strand, all thymine (or uracils) of a first strand can pair with an adenine of a second strand, and all cytosines of a first strand can pair with a guanine of a second strand, and vice versa.
- the degree of complementarity is determined over a stretch of 20, preferably 25, nucleotides, i.e.
- a 60% complementarity means that within a region of 20 nucleotides of two nucleic acid strands 12 nucleotides of the first strand can base pair with 12 nucleotides of the second strand according to the above ruling, either as a stretch of 12 contiguous nucleotides or interspersed by non-pairing nucleotides, when the two strands are attached to each other over said region of 20 nucleotides.
- the degree of complementarity can range from at least about 50% to full, i.e. 100% complementarity.
- Two single nucleic acid strands are said to be "substantially complementary" when they are at least about 80% complementary, preferably about 90% or higher. For carrying out the method of the present invention substantial complementarity is preferred.
- Prefened methods for detection and quantification of the amount of polynucleotides i.e. for the methods according to the invention allowing the determination of the level of expression of a marker, are those described by Sambrook et al. (1989) or real time methods known in the art as the TaqMan® method disclosed in WO92/02638 and the corresponding U.S. 5,210,015, U.S. 5,804,375, U.S. 5,487,972. This method exploits the exonuclease activity of a polymerase to generate a signal.
- the (at least one) target nucleic acid component is detected by a process comprising contacting the sample with an oligonucleotide containing a sequence complementary to a region of the target nucleic acid component and a labeled oligonucleotide containing a sequence complementary to a second region of the same target nucleic acid component sequence strand, but not including the nucleic acid sequence defined by the first oligonucleotide, to create a mixture of duplexes during hybridization conditions, wherein the duplexes comprise the target nucleic acid annealed to the first oligonucleotide and to the labeled oligonucleotide such that the 3 '-end of the first oligonucleotide is adjacent to the 5 '-end of the labeled oligonucleotide.
- this mixture is treated with a template-dependent nucleic acid polymerase having a 5' to 3' nuclease activity under conditions sufficient to permit the 5' to 3' nuclease activity of the polymerase to cleave the annealed, labeled oligonucleotide and release labeled fragments.
- the signal generated by the hydrolysis of the labeled oligonucleotide is detected and/ or measured.
- TaqMan® technology eliminates the need for a solid phase bound reaction complex to be formed and made detectable.
- Other methods include e.g. fluorescence resonance energy transfer between two adjacently hybridized probes as used in the LightCycler® format described in U.S. 6,174,670.
- Example 3 A prefened protocol if the marker, i.e. the polynucleotide, is in form of a transcribed nucleotide, is described in Example 3, where total RNA is isolated, cDNA and, subsequently, cRNA is synthesized and biotin is incorporated during the transcription reaction.
- the purified cRNA is applied to commercially available arcays which can be obtained e.g. from Affymetrix.
- the hybridized cRNA is detected according to the methods described in Example 3.
- the anays are produced by photolithography or other methods known to experts skilled in the art e.g. from U.S. 5,445,934, U.S. 5,744,305, U.S. 5,700,637, U.S. 5,945,334 and EP 0 619 321 or EP 0373 203, or as decribed hereinafter in greater detail.
- the polynucleotide or at least one of the polynucleotides is in form of a polypeptide.
- the expression level of the polynucleotides or polypeptides is detected using a compound which specifically binds to the polynucleotide of the polypeptide of the present invention.
- binding means that the compound is capable of discriminating between two or more polynucleotides or polypeptides, i.e. it binds to the desired polynucleotide or polypeptide, but essentially does not bind unspecifically to a different polynucleotide or polypeptide.
- the compound can be an antibody, or a fragment thereof, an enzyme, a so-called small molecule compound, a protein-scaffold, preferably an anticalin.
- the compound specifically binding to the polynucleotide or polypeptide is an antibody, or a fragment thereof.
- an "antibody” comprises monoclonal antibodies as first described by Kohler and Milstein in Nature 278 (1975), 495-497 as well as polyclonal antibodies, i.e. entibodies contained in a polyclonal antiserum.
- Monoclonal antibodies include those produced by transgenic mice. Fragments of antibodies include F(ab') 2 , Fab and Fv fragments. Derivatives of antibodies include scFvs, chimeric and humanized antibodies. See, for example Harlow and Lane, loc. cit.
- the person skilled in the art is aware of a variety of methods, all of which are included in the present invention.
- Examples include immunoprecipitation, Western blotting, Enzyme-linked immuno sorbent assay (ELISA), Enzyme-linked imrnuno sorbent assay (RIA), dissociation-enhanced lanthanide fluoro immuno assay (DELFIA), scintillation proximity assay (SPA).
- ELISA Enzyme-linked immuno sorbent assay
- RIA Enzyme-linked imrnuno sorbent assay
- DELFIA dissociation-enhanced lanthanide fluoro immuno assay
- SPA scintillation proximity assay
- the method for distinguishing MLL-PTD-positive AML from other AML subtypes is canied out on an anay.
- an “anay” or “microanay” refers to a linear or two- or three dimensional anangement of preferably discrete nucleic acid or polypeptide probes which comprises an intentionally created collection of nucleic acid or polypeptide probes of any length spotted onto a substrate/solid support.
- a collection of nucleic acids or polypeptide spotted onto a substrate/solid support also under the term "anay”.
- a microanay usually refers to a miniaturised anay anangement, with the probes being attached to a density of at least about 10, 20, 50, 100 nucleic acid molecules refening to different or the same genes per cm .
- an anay can be refened to as "gene chip”.
- the anay itself can have different formats, e.g. libraries of soluble probes or libraries of probes tethered to resin beads, silica chips, or other solid supports.
- the process of anay fabrication is well-known to the person skilled in the art.
- the process for preparing a nucleic acid anay comprises preparing a glass (or other) slide (e.g. chemical treatment of the glass to enhance binding of the nucleic acid probes to the glass surface), obtaining DNA sequences representing genes of a genome of interest, and spotting sequences these sequences of interest onto glass slide.
- Sequences of interest can be obtained via creating a cDNA library from an mRNA source or by using publicly available databases, such as GeneBank, to annotate the sequence info mation of custom cDNA libraries or to identify cDNA clones from previously prepared libraries.
- the liquid containing the amplified probes can be deposited on the anay by using a set of microspotting pins. Ideally, the amount deposited should be uniform.
- the process can further include UV-crosslinking in order to enhance immobilization of the probes on the anay.
- the anay is a high density oligonucleotide (oligo) anay using a light-directed chemical synthesis process, employing the so-called photolithography technology.
- oligo anays (according to the Affymetrix technology) use a single-dye technology. Given the sequence information of the markers, the sequence can be synthesized directly onto the anay, thus, bypassing the need for physical intermediates, such as PCR products, required for making cDNA anays.
- the marker, or partial sequences thereof can be represented by 14 to 20 features, preferably by less than 14 features, more preferably less than 10 features, even more preferably by 6 features or less, with each feature being a short sequence of nucleotides (oligonucleotide), which is a perfect match (PM) to a segment of the respective gene.
- the PM oligonucleotide are paired with mismatch (MM) oligonucleotides which have a single mismatch at the central base of the nucleotide and are used as "controls".
- the chip exposure sites are defined by masks and are deprotected by the use of light, followed by a chemical coupling step resulting in the synthesis of one nucleotide. The masking, light deprotection, and coupling process can then be repeated to synthesize the next nucleotide, until the nucleotide chain is of the specified length.
- the method of the present invention is canied out in a robotics system including robotic plating and a robotic liquid transfer system, e.g. using microfluidics, i.e. channelled structured.
- a particular prefened method according to the present invention is as follows:
- RNA preferably mRNA
- the present invention is directed to the use of at least one marker selected from the markers identifiable by their Affymetrix Identification
- markers for diagnosis of MLL-PTD-positive AML preferably based on microanay technology, offers the following advantages: (1) more rapid and more precise diagnosis, (2) easy to use in laboratories without specialized experience, (3) abolishes the requirement for analyzing viable cells for chromosome analysis (transport problem), and (4) very experienced hematologists for cytomorphology and cytochemistry, immunophenotyping as well as cytogeneticists and molecularbiologists are no longer required.
- the present invention refers to a diagnostic kit containing at least one marker selected from the markers identifiable by their Affymetrix Identification Numbers (affy id) as defined in Tables 1, and/or 3 for distinguishing MLL-PTD- positive AML from other AML subtypes, in combination with suitable auxiliaries.
- suitable auxiliaries include buffers, enzymes, labelling compounds, and the like.
- the marker contained in the kit is a nucleic acid molecule which is capable of hybridizing to the mRNA conesponding to at least one marker of the present invention.
- the at least one nucleic acid molecule is attached to a solid support, e.g. a polystyrene microtiter dish, nitrocellulose membrane, glass surface or to non-immobilized particles in solution.
- the diagnostic kit contains at least one reference for a MLL-PTD-positive AML subtype.
- the reference can be a sample or a data bank.
- the present invention is directed to an apparatus for distinguishing MLL-PTD-positive AML from other AML subtypes in a sample, containing a reference data bank obtainable by comprising (a) compiling a gene expression profile of a patient sample by determining the expression level at least one marker selected from the markers identifiable by their Affymetrix Identification Numbers (affy id) as defined in Tables 1, and/or 3, and (b) classifying the gene expression profile by means of a machine learning algorithm.
- affymetrix Identification Numbers as defined in Tables 1, and/or 3
- the "machine learning algorithm” is a computational-based prediction methodology, also known to the person skilled in the art as “classifier”, employed for characterizing a gene expression profile.
- the signals conesponding to a certain expression level which are obtained by the microanay hybridization are subjected to the algorithm in order to classify the expression profile.
- Supervised learning involves "training” a classifier to recognize the distinctions among classes and then “testing” the accuracy of the classifier on an independent test set. For new, unknown sample the classifier shall predict into which class the sample belongs.
- the machine learning algorithm is selected from the group consisting of Weighted Voting, K-Nearest Neighbors, Decision Tree Induction, Support Vector Machines (SVM), and Feed-Forward Neural Networks.
- the machine learning algorithm is Support Vector Machine, such as polynomial kernel and Gaussian Radial Basis Function-kernel SVM models.
- the classification accuracy of a given gene list for a set of microanay experiments is preferably estimated using Support Vector Machines (SVM), because there is evidence that SVM-based prediction slightly outperforms other classification techniques like k-Nearest Neighbors (k-NN).
- SVM Support Vector Machines
- the LIBSVM software package version 2.36 was used (SVM-type: C-SVC, linear kernel (http://www.csie.ntu.edu.tw/ ⁇ cjlin/libsvm/)).
- SVM-type C-SVC, linear kernel (http://www.csie.ntu.edu.tw/ ⁇ cjlin/libsvm/)).
- the skilled artisan is furthermore refened to Brown et al., Proc.Natl.Acad.Sci., 2000; 97: 262-267, Furey et al, Bioinformatics. 2000; 16: 906-914, and Vapnik V. Statistical Learning Theory. New
- the classification accuracy of a given gene list for a set of microanay experiments can be estimated using Support Vector Machines (SVM) as supervised learning technique.
- SVMs are trained using differentially expressed genes which were identified on a subset of the data and then this trained model is employed to assign new samples to those trained groups from a second and different data set. Differentially expressed genes were identified applying ANOVA and t-test-statistics (Welch t-test). Based on identified distinct gene expression signatures respective training sets consisting of 2/3 of cases and test sets with 1/3 of cases to assess classification accuracies are designated. Assignment of cases to training and test set is randomized and balanced by diagnosis. Based on the training set a Support Vector Machine (SVM) model is built.
- SVM Support Vector Machine
- the apparent accuracy i.e. the overall rate of conect predictions of the complete data set was estimated by lOfold cross validation.
- This means that the data set was divided into 10 approximately equally sized subsets, an SVM-model was trained for 9 subsets and predictions were generated for the remaining subset. This training and prediction process was repeated 10 times to include predictions for each subset. Subsequently the data set was split into a training set, consisting of two thirds of the samples, and a test set with the remaining one third. Apparent accuracy for the training set was estimated by lOfold cross validation (analogous to apparent accuracy for complete set). A SVM-model of the training set was built to predict diagnosis in the independent test set, thereby estimating true accuracy of the prediction model. This prediction approach was applied both for overall classification (multi-class) and binary classification (diagnosis X > yes or no). For the latter, sensitivity and specificity were calculated:
- the reference data bank is backed up on a computational data memory chip which can be inserted in as well as removed from the apparatus of the present invention, e.g. like an interchangeable module, in order to use another data memory chip containing a different reference data bank.
- the apparatus of the present invention containing a desired reference data bank can be used in a way such that an unknown sample is, first, subjected to gene expression profiling, e.g. by microanay analysis in a manner as described supra or in the art, and the expression level data obtained by the analysis are, second, fed into the apparatus and compared with the data of the reference data bank obtainable by the above method.
- the apparatus suitably contains a device for entering the expression level of the data, for example a control panel such as a keyboard.
- the results, whether and how the data of the unknown sample fit into the reference data bank can be made visible on a provided monitor or display screen and, if desired, printed out on an incorporated of connected printer.
- the apparatus of the present invention is equipped with particular appliances suitable for detecting and measuring the expression profile data and, subsequently, proceeding with the comparison with the reference data bank.
- the apparatus of the present invention can contain a gripper arm and/or a tray which takes up the microanay containing the hybridized nucleic acids.
- the present invention refers to a reference data bank for distinguishing MLL-PTD-positive AML from other AML subtypes in a sample obtainable by comprising (a) compiling a gene expression profile of a patient sample by determining the expression level of at least one marker selected from the markers identifiable by their Affymetrix Identification Numbers (affy id) as defined in Tables 1, and/or 3, and (b) classifying the gene expression profile by means of a machine learning algorithm.
- affymetrix Identification Numbers affy id
- the reference data bank is backed up and/or contained in a computational memory data chip.
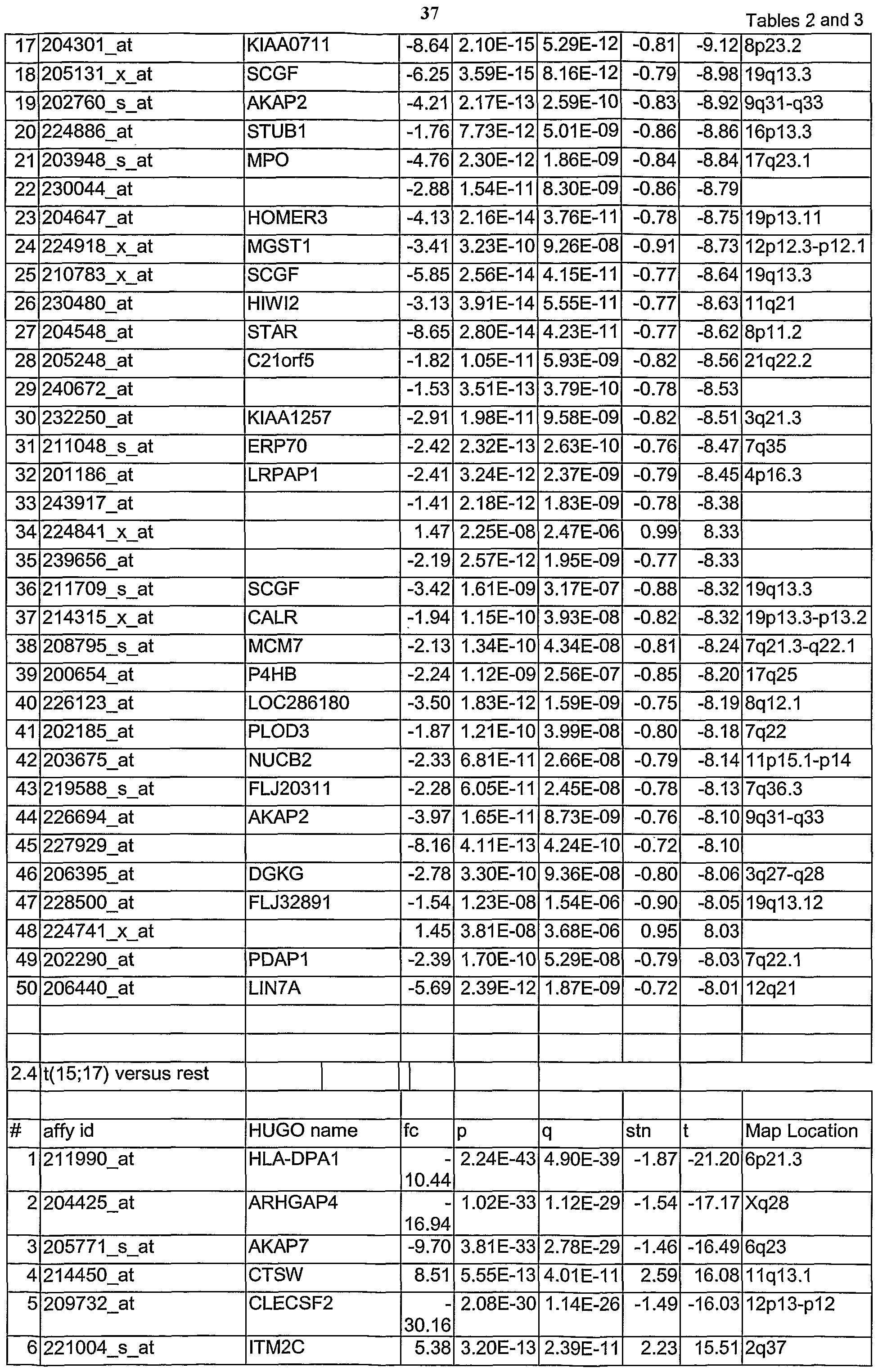
- Table 1.1-3.15 show AML subtype analysis of MLL-PTD-positive AML versus other AML subtypes. The analysed markers are ordered according to their q-values, beginning with the lowest q-values.
- Tables 1.1 to 3.15 are accompanied with explanatory tables (Table 1.1 A to 3.15 A) where the numbering and the
- Affymetrix Id are further defined by other parameters, e.g. gene bank accession number.
- MLL-PTD Partial tandem duplication within the MLL-gene
- MLL-PTD Partial tandem duplication within the MLL-gene
- t(llq23)/MLL the occurence of MLL-PTD is characterized by an unfavourable prognosis.
- the pathogenetic mechanisms of the MLL-PTD are poorly understood and downstream genes effected by this molecular abenation are not known.
- AML we performed global gene expression profiling of 184 AML samples at diagnosis using the U133 set of expression microanays (Affymetrix) with >30,000 human genes represented on both anays.
- Microanay data was analyzed by pattern recognition algorithms (Principal Component Analysis (PCA), hierarchical clustering), as well as Support Vector Machines
- SVM classification accuracies
- PTD+ and low in t(l lq23)/MLL samples were HOXB5, HOXB2, MAN1A1, and ZNF207.
- PCA and hierarchical cluster visualize that the MLL-PTD samples characterize a homogeneous subgroup within AML with normal karyotype, but do not separate from them.
- Some of the genes that were highly expressed in AML-NK and low in PTD+ were AAK1, RAB4A, HOXA2, BID.
- genes that were low in AML-NK and high in PTD+ were, among others, MLL, YY1, and SRP46.
- the training set comprised 83 AML-NK and 19 PTD+ AML cases, the test set 41 AML-NK and 9 PTD+ AML cases, respectively.
- the 50 test samples were assigned to the conect group with an accuracy of 88%.
- the methods section contains both information on statistical analyses used for identification of differentially expressed genes and detailed annotation data of identified microanay probesets.
- sequence data are omitted due to their large size, and because they do not change, whereas the annotation data are updated periodically, for example new information on chromomal location and functional annotation of the respective gene products. Sequence data are available for download in the NetAffx Download
- Microanay probesets for example found to be differentially expressed between different types of leukemia samples are further described by additional information.
- HG-U133 ProbeSet D HG-U133 ProbeSetJD describes the probe set identifier. Examples are:
- GeneChip probe anay name where the respective probeset is represented. Examples are: Affymetrix Human Genome U133A Anay or
- the Sequence Type indicates whether the sequence is an Exemplar, Consensus or Control sequence.
- An Exemplar is a single nucleotide sequence taken directly from a public database. This sequence could be an mRNA or EST.
- a Consensus sequence is a nucleotide sequence assembled by Affymetrix, based on one or more sequence taken from a public database.
- the cluster identification number with a sub-cluster identifier appended is the cluster identification number with a sub-cluster identifier appended.
- Sequence Derived From The accession number of the single sequence, or representative sequence on which the probe set is based. Refer to the "Sequence Source” field to determine the database used.
- a gene symbol and a short title when one is available. Such symbols are assigned by different organizations for different species.
- Affymetrix annotational data come from the UniGene record. There is no indication which species-specific databank was used, but some of the possibilities include for example HUGO: The Human Genome Organization.
- the map location describes the chromosomal location when one is available.
- Cluster type can be "full length” or “est”, or " — “ if unknown.
- LocusLink This information represents the LocusLink accession number. Full Length Ref. Sequences:
- the field contains the ID and description for each entry, and there can be multiple entries per probeSet.
- Example 3 Sample preparation, processing and data analysis
- Microanay analyses were performed utilizing the GeneChip ® System (Affymetrix, Santa Clara, USA). Hybridization target preparations were performed according to recommended protocols (Affymetrix Technical Manual). In detail, at time of diagnosis, mononuclear cells were purified by Ficoll-Hypaque density centrifugation. They had been lysed immediately in RLT buffer (Qiagen, Hilden, Germany), frozen, and stored at -80°C from 1 week to 38 months. For gene expression profiling cell lysates of the leukemia samples were thawed, homogenized (QIAshredder, Qiagen), and total RNA was extracted (RNeasy Mini).
- RNA isolated from 1 x 10 7 cells was used as starting material for cDNA synthesis with oligo [(dT) 24 T7promotor] 65 primer (cDNA Synthesis System, Roche Applied Science, Mannheim, Germany). cDNA products were purified by phenol/chlorophorm/IAA extraction (Ambion, Austin, USA) and acetate/ethanol-precipitated overnight. For detection of the hybridized target nucleic acid biotin-labeled ribonucleotides were incorporated during the following in vitro transcription reaction (Enzo BioAnay HighYield RNA Transcript Labeling Kit, Enzo Diagnostics).
- Suite software (version 5.0.1) extracted fluorescence signal intensities from each feature on the microanays as detected by confocal laser scanning according to the manufacturer's recommendations.
- Expression analysis quality assessment parameters included visiual anay inspection of the scanned image for the presence of image artifacts and conect grid alignment for the identification of distinct probe cells as well as both low 375' ratio of housekeeping controls (mean: 1.90 for GAPDH) and high percentage of detection calls (mean: 46.3% present called genes).
- the 3' to 5' ratio of GAPDH probesets can be used to assess RNA sample and assay quality. Signal values of the 3' probe sets for GAPDH are compared to the Signal values of the conesponding
- the ratio of the 3' probe set to the 5' probe set is generally no more than 3.0.
- a high 3' to 5' ratio may indicate degraded RNA or inefficient synthesis of ds cDNA or biotinylated cRNA (GeneChip ® Expression Analysis Technical Manual, www.affymetrix.com).
- Detection calls are used to determine whether the transcript of a gene is detected (present) or undetected (absent) and were calculated using default parameters of the Microanay Analysis Suite MAS 5.0 software package.
- Method 2 Bone manow (BM) aspirates are taken at the time of the initial diagnostic biopsy and remaining material is immediately lysed in RLT buffer (Qiagen), frozen and stored at -80 C until preparation for gene expression analysis.
- RLT buffer Qiagen
- the targets for GeneChip analysis are prepared according to the cunent Expression Analysis. Briefly, frozen lysates of the leukemia samples are thawed, homogenized
- RNA extracted RNeasy Mini Kit, Qiagen
- RNA synthesis Kit Oligo-dT-T7-Promotor Primer
- the cDNA is purified by phenol-chlorophorm extraction and precipitated with 100% Ethanol over night.
- biotin-labeled ribonucleotides are incorporated during the in vitro transcription reaction (Enzo® BioAnayTM HighYieldTM RNA Transcript Labeling Kit, ENZO).
- the Affymetrix software (Microanay Suite, Version 4.0.1) extracted fluorescence intensities from each element on the anays as detected by confocal laser scanning according to the manufacturers recommendations .
- OVA One-Versus-AII
- _x_at HINT1 1.40 4.69E-12 2.81 E-08 1.68 10.32 5q31.2 19200986.at SERPING1 9.53 1.51 E-09 1.26E-06 1.97 10.29 11q12- q13.1 20201137.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Hematology (AREA)
- Organic Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Pathology (AREA)
- Analytical Chemistry (AREA)
- Biomedical Technology (AREA)
- Oncology (AREA)
- Microbiology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Physics & Mathematics (AREA)
- Hospice & Palliative Care (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Urology & Nephrology (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- General Physics & Mathematics (AREA)
- Biophysics (AREA)
- Cell Biology (AREA)
- Medicinal Chemistry (AREA)
- Food Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/576,093 US20070212687A1 (en) | 2003-11-04 | 2004-11-04 | Method For Distinguishing Mll-Ptd-Positive Aml From Other Aml Subtypes |
| EP04797591A EP1682902A2 (en) | 2003-11-04 | 2004-11-04 | Method for distinguishing mll-ptd-positive aml from other aml subtypes |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP03025339.7 | 2003-11-04 | ||
| EP03025339 | 2003-11-04 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| WO2005043162A2 true WO2005043162A2 (en) | 2005-05-12 |
| WO2005043162A8 WO2005043162A8 (en) | 2005-07-07 |
| WO2005043162A3 WO2005043162A3 (en) | 2005-08-18 |
Family
ID=34530666
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2004/012464 WO2005043162A2 (en) | 2003-11-04 | 2004-11-04 | Method for distinguishing mll-ptd-positive aml from other aml subtypes |
Country Status (3)
| Country | Link |
|---|---|
| US (1) | US20070212687A1 (en) |
| EP (1) | EP1682902A2 (en) |
| WO (1) | WO2005043162A2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007135467A (en) * | 2005-11-17 | 2007-06-07 | National Institute Of Advanced Industrial & Technology | UTILIZATION OF Kank 2 GENE FOR CANCER TREATMENT, CANCER DETECTION AND CREATION OF MEDICINE |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1043676A2 (en) * | 1999-04-09 | 2000-10-11 | Whitehead Institute For Biomedical Research | Methods for classifying samples and ascertaining previously unknown classes |
| WO2003039443A2 (en) * | 2001-11-05 | 2003-05-15 | Deutsches Krebsforschungszentrum | Novel genetic markers for leukemias |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5700637A (en) * | 1988-05-03 | 1997-12-23 | Isis Innovation Limited | Apparatus and method for analyzing polynucleotide sequences and method of generating oligonucleotide arrays |
| US5744101A (en) * | 1989-06-07 | 1998-04-28 | Affymax Technologies N.V. | Photolabile nucleoside protecting groups |
| US5143854A (en) * | 1989-06-07 | 1992-09-01 | Affymax Technologies N.V. | Large scale photolithographic solid phase synthesis of polypeptides and receptor binding screening thereof |
| US5210015A (en) * | 1990-08-06 | 1993-05-11 | Hoffman-La Roche Inc. | Homogeneous assay system using the nuclease activity of a nucleic acid polymerase |
| EP0695941B1 (en) * | 1994-06-08 | 2002-07-31 | Affymetrix, Inc. | Method and apparatus for packaging a chip |
| US6174670B1 (en) * | 1996-06-04 | 2001-01-16 | University Of Utah Research Foundation | Monitoring amplification of DNA during PCR |
| US20030138793A1 (en) * | 2001-06-10 | 2003-07-24 | Irm Llc, A Delaware Limited Liability Company | Molecular signatures of commonly fatal carcinomas |
-
2004
- 2004-11-04 WO PCT/EP2004/012464 patent/WO2005043162A2/en active Application Filing
- 2004-11-04 US US10/576,093 patent/US20070212687A1/en not_active Abandoned
- 2004-11-04 EP EP04797591A patent/EP1682902A2/en not_active Withdrawn
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1043676A2 (en) * | 1999-04-09 | 2000-10-11 | Whitehead Institute For Biomedical Research | Methods for classifying samples and ascertaining previously unknown classes |
| WO2003039443A2 (en) * | 2001-11-05 | 2003-05-15 | Deutsches Krebsforschungszentrum | Novel genetic markers for leukemias |
Non-Patent Citations (13)
| Title |
|---|
| ALIZADEH A ET AL: "THE LYMPHOCHIP: A SPECIALIZED CDNA MICROARRAY FOR THE GENOMIC-SCALE ANALYSIS OF GENE EXPRESSION IN NORMAL AND MALIGNANT LYMPHOCYTES" COLD SPRING HARBOR SYMPOSIA ON QUANTITATIVE BIOLOGY, BIOLOGICAL LABORATORY, COLD SPRING HARBOR, NY, US, vol. 64, no. 1, 1999, pages 71-78, XP001099007 ISSN: 0091-7451 * |
| CONNER SEAN D ET AL: "Identification of an adaptor-associated kinase, AAK1, as a regulator of clathrin-mediated endocytosis" JOURNAL OF CELL BIOLOGY, vol. 156, no. 5, 4 March 2002 (2002-03-04), pages 921-929, XP002270210 ISSN: 0021-9525 * |
| DATABASE BIOSIS [Online] BIOSCIENCES INFORMATION SERVICE, PHILADELPHIA, PA, US; 16 November 2001 (2001-11-16), SCHNITTGER SUSANNE ET AL: "Partial tandem duplications (PTD) of the MLL gene: A study of 1603 patients reveals high frequency in secondary acute myeloid leukemia and low association with trisomy 11" XP002270209 Database accession no. PREV200200261466 & BLOOD, vol. 98, no. 11 Part 1, 16 November 2001 (2001-11-16), page 801a, 43rd Annual Meeting of the American Society of Hematology, Part 1;Orlando, Florida, USA; December 07-11, 2001 ISSN: 0006-4971 * |
| DATABASE BIOSIS [Online] BIOSCIENCES INFORMATION SERVICE, PHILADELPHIA, PA, US; 16 November 2001 (2001-11-16), SCHOCH CLAUDIA ET AL: "Specific abnormalities on the genomic level result in a distinct gene expression pattern detected by oligonucleotide microarrays: An analysis of 25 patients with AML M2/t(8;21), AML M3/M3v/t(15;17), and AML M4eo/inv(16)" XP002269491 Database accession no. PREV200200129822 & BLOOD, vol. 98, no. 11 Part 1, 16 November 2001 (2001-11-16), pages 92a-93a, 43rd Annual Meeting of the American Society of Hematology, Part 1;Orlando, Florida, USA; December 07-11, 2001 ISSN: 0006-4971 * |
| DATABASE BIOSIS [Online] BIOSCIENCES INFORMATION SERVICE, PHILADELPHIA, PA, US; 16 November 2002 (2002-11-16), HAFERLACH TORSTEN ET AL: "Gene Expression Profiling Is Able To Reproduce Different Phenotypes in AML as Defined by the FAB Classification." XP002269981 Database accession no. PREV200300357598 & BLOOD, vol. 100, no. 11, 16 November 2002 (2002-11-16), page Abstract No. 731, 44th Annual Meeting of the American Society of Hematology;Philadelphia, PA, USA; December 06-10, 2002 ISSN: 0006-4971 * |
| DATABASE BIOSIS [Online] BIOSCIENCES INFORMATION SERVICE, PHILADELPHIA, PA, US; 16 November 2002 (2002-11-16), KOHLMANN ALEXANDER ET AL: "A Simplified and Partially Automated Target Preparation Method for Gene Expression Profiling." XP002269495 Database accession no. PREV200300367771 & BLOOD, vol. 100, no. 11, 16 November 2002 (2002-11-16), page Abstract No. 4287, 44th Annual Meeting of the American Society of Hematology;Philadelphia, PA, USA; December 06-10, 2002 ISSN: 0006-4971 * |
| DATABASE BIOSIS [Online] BIOSCIENCES INFORMATION SERVICE, PHILADELPHIA, PA, US; 16 November 2002 (2002-11-16), NEBEN KAI ET AL: "Acute Myeloid Leukemia with Normal Karyotype and Mutation of the FLT3 or MLL Gene Are Characterized by Specific Gene Expression Profiles." XP002270208 Database accession no. PREV200300336869 & BLOOD, vol. 100, no. 11, 16 November 2002 (2002-11-16), page Abstract No. 2172, 44th Annual Meeting of the American Society of Hematology;Philadelphia, PA, USA; December 06-10, 2002 ISSN: 0006-4971 * |
| DATABASE BIOSIS [Online] BIOSCIENCES INFORMATION SERVICE, PHILADELPHIA, PA, US; 16 November 2002 (2002-11-16), SCHNITTGER SUSANNE ET AL: "Acute Myeloid Leukemia (AML) with Partial Tandem Duplication of the MLL-Gene (MLL-PTD) Can Be Discriminated from MLL-Translocations Based on Specific Gene Expression Profiles." XP002270207 Database accession no. PREV200300335802 & BLOOD, vol. 100, no. 11, 16 November 2002 (2002-11-16), page Abstract No. 1202, 44th Annual Meeting of the American Society of Hematology;Philadelphia, PA, USA; December 06-10, 2002 ISSN: 0006-4971 * |
| DUGAS M ET AL: "A comprehensive leukemia database: integration of cytogenetics, molecular genetics and microarray data with clinical information, cytomorphology and immunophenotyping" LEUKEMIA, MACMILLAN PRESS LTD, US, vol. 15, no. 12, December 2001 (2001-12), pages 1805-1810, XP002263731 ISSN: 0887-6924 * |
| DUGAS MARTIN ET AL: "Impact of integrating clinical and genetic information." IN SILICO BIOLOGY, vol. 2, no. 3, 2002, pages 383-391, XP001179418 ISSN: 1386-6338 (ISSN print) * |
| GOLUB T R ET AL: "Molecular classification of cancer: Class discovery and class prediction by gene expression monitoring" SCIENCE, AMERICAN ASSOCIATION FOR THE ADVANCEMENT OF SCIENCE,, US, vol. 286, no. 5439, 15 October 1999 (1999-10-15), pages 531-537, XP002207658 ISSN: 0036-8075 cited in the application * |
| KOHLMANN A ET AL: "MOLECULAR CHARACTERIZATION OF ACUTE LEUKEMIAS BY USE OF MICROARRAY TECHNOLOGY" GENES, CHROMOSOMES & CANCER, XX, XX, vol. 37, no. 4, August 2003 (2003-08), pages 396-405, XP008025253 * |
| SCHOCH CLAUDIA ET AL: "Acute myeloid leukemias with reciprocal rearrangements can be distinguished by specific gene expression profiles" PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF USA, NATIONAL ACADEMY OF SCIENCE. WASHINGTON, US, vol. 99, no. 15, 23 July 2002 (2002-07-23), pages 10008-10013, XP002215484 ISSN: 0027-8424 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007135467A (en) * | 2005-11-17 | 2007-06-07 | National Institute Of Advanced Industrial & Technology | UTILIZATION OF Kank 2 GENE FOR CANCER TREATMENT, CANCER DETECTION AND CREATION OF MEDICINE |
Also Published As
| Publication number | Publication date |
|---|---|
| US20070212687A1 (en) | 2007-09-13 |
| WO2005043162A8 (en) | 2005-07-07 |
| WO2005043162A3 (en) | 2005-08-18 |
| EP1682902A2 (en) | 2006-07-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2004235382A1 (en) | Methods for diagnosing AML and MDS differential gene expression | |
| US20090118132A1 (en) | Classification of Acute Myeloid Leukemia | |
| CA2642535A1 (en) | Urine gene expression ratios for detection of cancer | |
| WO2006048263A2 (en) | Gene expression profiling in acute promyelocytic leukemia | |
| WO2006048266A2 (en) | Gene expression profiling of leukemias with mll gene rearrangements | |
| US20070148648A1 (en) | Method for distinguishing who classified aml subtypes | |
| EP1682902A2 (en) | Method for distinguishing mll-ptd-positive aml from other aml subtypes | |
| WO2005045434A2 (en) | Method for distinguishing aml subtypes with recurring genetic aberrations | |
| US20070207459A1 (en) | Method For Distinguishing Immunologically Defined All Subtype | |
| US20070292970A1 (en) | Method for Distinguishing Aml-Specific Flt3 Length Mutations From Tkd Mutations | |
| WO2006048270A2 (en) | Methods of detecting leukemia and its subtypes | |
| US20070275380A1 (en) | Method for Distinguishing Aml Subtypes With Aberrant and Prognostically Intermediate Karyotypes | |
| US20070212688A1 (en) | Method For Distinguishing Cbf-Positive Aml Subtypes From Cbf-Negative Aml Subtypes | |
| US20070099190A1 (en) | Method for distinguishing leukemia subtypes | |
| US20070212734A1 (en) | Method for Distinguishing T(11Q23)/Mll-Positive Leukemias From t(11Q23)/Mll Negative Leukemia | |
| WO2005045438A2 (en) | METHOD FOR DISTINGUISHING AML SUBTYPE INV(3)(q21q26)/t(3;3)(q21q26) FROM OTHER AML SUBTYPES | |
| US20070128607A1 (en) | Method for distinguishing aml subtypes with different gene dosages | |
| US20070122814A1 (en) | Methods for distinguishing prognostically definable aml | |
| WO2006048275A2 (en) | Chronic lymphocytic leukemia expression profiling | |
| EP1815012A2 (en) | Classifying leukemia with translocation (9;22) | |
| WO2006048273A1 (en) | Methods of validating gene expression assays |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| CFP | Corrected version of a pamphlet front page | ||
| CR1 | Correction of entry in section i |
Free format text: IN PCT GAZETTE 19/2005 UNDER (72, 75) DELETE "MERGENTHALER, SUSANNE" |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004797591 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004797591 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10576093 Country of ref document: US Ref document number: 2007212687 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10576093 Country of ref document: US |