WO2005023780A1 - Selective synthesis of cf3-substituted pyrimidines - Google Patents
Selective synthesis of cf3-substituted pyrimidines Download PDFInfo
- Publication number
- WO2005023780A1 WO2005023780A1 PCT/IB2004/002744 IB2004002744W WO2005023780A1 WO 2005023780 A1 WO2005023780 A1 WO 2005023780A1 IB 2004002744 W IB2004002744 W IB 2004002744W WO 2005023780 A1 WO2005023780 A1 WO 2005023780A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amine
- lewis acid
- group
- groups
- formula
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/42—One nitrogen atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
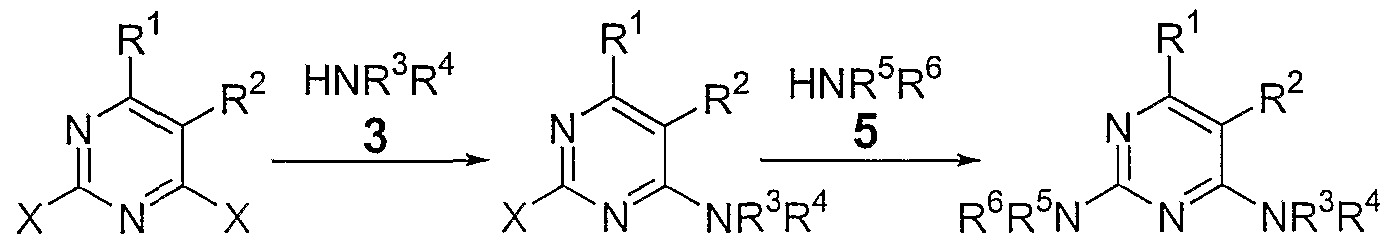
- the 2,4-diamino pyrimidine moiety (1 ) is a common component in a variety of biologically active drug-like molecules. Pyrimidine derivatives have been found to be useful in the treatment of abnormal cell growth, such as cancer, in mammals. These moieties are commonly synthesized starting with pyrimidine intermediate 2 (where "X" is a leaving group; most commonly a halogen) and an equivalent of amine 3, HNR 3 R 4 (see Scheme 1 below).
- the present invention relates to a process of making a compound of the formula 11
- X 2 is a leaving group such aass a halide, arylsulfonate, alkylsulfonate, perfluoroalkylsulfonate, arylsulfinate or alkylsulfinate; and R 3 and R 4 are substituents independently selected from the group consisting of hydrogen, an aromatic group and an aliphatic group; or taken together -NR 3 R 4 can form a 4- 11 membered aromatic or aliphatic ring; wherein the process comprises reacting a compound of formula 10 10 with an amine of formula 3 (HNR 3 R 4 ) in the presence of a Lewis Acid and a non-nucleophilic base to form a compound of formula 11 , wherein X 1 is a leaving group such as a halide, arylsulfonate, alkylsulfonate, perfluoroalkylsulfonate, arylsulfinate or alkylsulfinate.
- amine 3 is an aromatic amine and 0.25-10 equivalents of Lewis Acid are used relative to the amount of pyrimidine 10, and preferably 0.5-3.0 equivalents of Lewis Acid are used relative to pyrimidine 10.
- amine 3 is an aliphatic amine and 0.5-10 equivalents of Lewis Acid are used relative to the amount of pyrimidine 10, and preferably 1-4 equivalents of Lewis Acid are used relative to pyrimidine 10.
- X 1 and X 2 are the same or different and are each independently halides and the Lewis Acid is a salt of zinc or magnesium.
- X 1 and X 2 are chloride and the Lewis Acid is zinc chloride
- the compounds prepared by the method of the present invention include all stereoisomers (e.g., cis and trans isomers) and all optical isomers of compounds of the formula
- the compounds and salts prepared by the method of the present invention can exist in several tautomeric forms, including the enol and imine form, and the keto and enamine form and geometric isomers and mixtures thereof.
- the preparation of all such tautomeric forms is included within the scope of the present invention.
- Tautomers exist as mixtures of a tautomeric set in solution. In solid form, usually one tautomer predominates. Even though the preparation of one tautomer may be described, the present invention encompasses the preparation of all tautomers of the present compounds.
- the present invention also includes the preparation of atropisomers of the present invention.
- Atropisomers refer to compounds of formula 11 that can be separated into rotationally restricted isomers.
- the compounds prepared by the method of the invention may contain olefin-like double bonds. When such bonds are present, the compounds exist as cis and trans configurations and as mixtures thereof and the present invention contemplates the preparation of such compounds.
- aromatic and specifically, an "aromatic group” refers to an aryl or heteroaryl radical as defined herein.
- an "aromatic amine” or “aromatic amine radical” refers to any amine or amine radical bound to at least one sp 2 carbon atom that is part of an aryl or heteroaryl group.
- An amine or amine radical will be referred to as an aromatic amine or radical even if the amine nitrogen is bound to a hydrogen or an sp 3 carbon atom, in addition to the one sp 2 carbon atom.
- -HN(C 6 -C ⁇ 0 )aryl and -N((C 1 -C 6 )alkyl)((C 6 -C 10 )aryl) each refer to aromatic amine radicals as defined herein, despite the fact that each amine nitrogen is attached to non-aromatic substituents.
- aryl refers to aromatic radicals such as phenyl, naphthyl, tetrahydronaphthyl, indanyl and the like.
- aryl may be optionally substituted with 1- 3 suitable substituents, as defined herein.
- Aryl also refers to a phenyl radical fused to a non-aromatic heterocycle. Examples of such groups include but are not limited to 2-oxo- indolinyl, chromanyl, indolinyl and 2-oxo-3,4-dihydroquinolinyl optionally substituted by 1 to 3 suitable substituents.
- heteroaryl refers to an aromatic heterocyclic group usually with one heteroatom selected from O, S and N in the ring, wherein the aromatic heterocyclic group may be substituted by up to three suitable substituents as defined herein.
- the aromatic heterocyclic group may optionally have up to four N atoms in the ring.
- heteroaryl groups include but are not limited to pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, thienyl, furyl, imidazolyl, pyrrolyl, oxazolyl (e.g., 1 ,3-oxazolyl, 1 ,2- oxazolyl), thiazolyl (e.g., 1 ,2-thiazolyl, 1 ,3-thiazolyl), pyrazolyl, tetrazolyl, triazolyl (e.g., 1 ,2,3- triazolyl, 1 ,2,4-triazolyl), oxadiazolyl (e.g., 1 ,2,3-oxadiazolyl), thiadiazolyl (e.g., 1 ,3,4- thiadiazolyl),
- heteroaryl also refers to one of the aforementioned heteroaryl groups fused to a non-aromatic heterocycle. Examples of such groups include but are not limited to 1 ,3-Dihydro-pyrrolo[2,3- b]pyridin-2-one, 3,4-Dihydro-1 H-[1 ,8]naphthyridin-2-one, 1 ,3-Dihydro-pyrrolo[2,3-b]pyridine and 3,4-Dihydro-2H-pyrano[2,3-b]pyridine.
- Aliphatic group refers to an alkyl, cycloalkyl, or heterocycloalkyl radical, as defined herein. Aliphatic groups may be substituted with up to three suitable substituents as defined herein. As used herein, the term “aliphatic amine” or “aliphatic amino radical” refers to any amine or amine radical in which the amine or radical nitrogen atom is bound to an sp 3 carbon that is part of an alkyl, cycloalkyl, or heterocycloalkyl group. Aliphatic amine groups may be substituted with up to three suitable substituents as defined herein.
- alkyl refers to C ⁇ -C 10 linear or branched alkyl groups (such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl etc.) optionally substituted by 1 to 3 suitable substituents as defined herein.
- cycloalkyl refers to C 3 -C 12 mono, bicyclic or tricyclic carbocyclic ring (e.g., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclopentenyl, cyclohexenyl, bicyclo[2.2.1]heptanyl, bicyclo[3.2.1]octanyl and bicyclo[5.2.0]nonanyl, etc.) that is optionally substituted by 1 to 3 suitable substituents as defined herein.
- suitable substituents as defined herein.
- Bicyclic or tricyclic species may be fused, bridged or spirocyclic.
- cycloalkyl or “cyclyl” groups include, but are not limited to cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclohexenyl, bicyclo[2.2.1]heptanyl, bicyclo[3.2.1]octanyl, bicyclo[3.1.0]hexyl and spiro[2.4]heptyl.
- heterocycloalkyl or “heterocyclyl” or “heterocycle” refers to a mono, bicyclic or tricyclic group containing 3 to 9 carbon atoms and 1 to 4 heteroatoms selected from -N, -NR, -0-, -S-, -SO or -S0 2 , wherein the cyclic radical is optionally substituted by 1 to 3 suitable substituents as defined herein.
- Bicyclic or tricyclic species may be fused, bridged or spirocyclic.
- halogen includes fluoro, chloro, bromo or iodo or fluoride, chloride, bromide or iodide.
- said amine can be a mono, bicyclic or tricyclic ring comprising 3 to 9 carbon atoms and 0 to 3 heteroatoms selected from -N, -O-, -S-, -SO or -S0 2 (excluding the nitrogen atom in - ⁇ /R 3 R 4 or - ⁇ /R 5 R 6 ).
- the cyclic amine may be optionally substituted with 1 to 3 suitable substituents as defined herein.
- Bicyclic or tricyclic species may be fused bridged or spirocyclic.
- cyclic amines include but are not limited to morpholine, azetidine, piperazine, piperidine, pyrrolidine, indoline, thiomorpholine.
- a "suitable substituent” is intended to mean a chemically and pharmaceutically acceptable functional group. Such suitable substituents for the aforementioned aryl, heteroaryl, alkyl, cycloalkyl, heterocycloalkyl groups may be routinely described by those skilled in the art.
- substituents can be substituted by additional substituents.
- "Embodiment” as used herein refers to specific groupings of compounds or uses into discrete subgenera. Such subgenera may be cognizable according to one particular substituent such as a specific R 3 or R 4 group. Other subgenera are cognizable according to combinations of various substituents, such as all compounds wherein R 3 is hydrogen and R 4 is (C r C 6 )alkyl, optionally substituted by -(C 3 -C ⁇ 0 )cyclyl.
- X leaving group
- Compounds of the formula 11 can be prepared by reaction of pyrimidine 10 and a primary or secondary amine nucleophile (3) in the presence of a Lewis acid and a non- nucleophilic base in an organic solvent or mixture of solvents.
- Leaving groups (X 1 , X 2 , which may be the same or different) on pyrimidine 10 suitable for displacement include but are not limited to halides, sulfonates and sulfinates.
- each leaving group is a halide.
- the halide is chloride.
- Suitable primary and secondary amine nucleophiles include aromatic, aliphatic or cyclic amines as described above.
- Lewis acids include but are not limited to the salts of Zn, Mg, Sn, Ti, Al, B, Li, Ag, Na, K, Ca, Va, Cr, Mn, Fe, Co, Ni, In, Zr, Sm and Cu.
- salts of Zn, Mg, Sn, Ti, Al, B, Li, Ag, and Cu are preferred.
- a salt of Zn or Mg is used as the Lewis acid.
- Suitable equivalents of Lewis Acid relative to pyrimidine 10 necessary to effect selective amine addition range from 0.25-10 equivalents, and preferably 0.5-3 equivalents when the amine nucleophile is aromatic or 0.5-10 equivalents, and preferably 1-4 equivalents when the amine is aliphatic (or an aromatic amine capable of forming a multidentate ligand with the Lewis Acid).
- Suitable organic solvents include but are not limited tetrahydrofuran, 1 ,2- dichloroethane, f-butanol, ether, methylene chloride, acetonitrile, methanol, ethanol, 2- propanol, dioxane, 1 ,2-dimethoxyethane, toluene, chloroform, ethyl acetate or mixtures thereof, preferably a mixture of a halogenated and alcoholic solvents.
- Suitable non- nucleophilic bases include but are not limited to triethylamine, N, N-diisopropyl-ethylamine, diaza-bicyloundecene (DBU) or resin bound bases such as MP-Carbonate. Temperatures for this process range from -30°C to 50°C; preferably the reaction is run at 0°C to room temperature. The reaction may be carried out in a single step or in several sequential steps, without any adverse effect on the overall yield or selectivity of the reaction.
- DBU diaza-bicyloundecene
- Compounds of formula 12-15 are useful for example, in the treatment of abnormal cell growth, such as cancer, in mammals.
- compounds 12-15 are inhibitors of protein kinases.
- compound of formula 12 are selective inhibitors of certain receptor and non-receptor tyrosine kinases, e.g., FAK (focal adhesion kinase).
- FAK receptor and non-receptor tyrosine kinases
- Compounds such as these are described in U.S. Patent Application Serial Nos. 10/734, 039(Attorney docket number PC25339) and 10/733215 (Attorney docket number PC25937).
- EXAMPLES The following examples illustrate the preparation of the compounds of the present invention.
- NMR data are reported in parts per million and are referenced to the deuterium lock signal from the sample solvent. Commercial reagents were utilized without further purification. THF refers to tetrahydrofuran and DMF refers to N,N-dimethylformamide. Chromatography refers to column chromatography performed using .040 mm silica gel and executed under flash chromatography conditions. Low Resolution Mass Spectra (LRMS) were recorded on a Fisons Atmospheric Pressure Chemical lonization platform, which uses a 50/50 mixture of acetonitrile/water with 0.1 % formic acid as the ionizing agent. All non- aqueous reactions were run under a nitrogen atmosphere for convenience and to maximize yields.
- LRMS Low Resolution Mass Spectra
- Example 2 General procedure for non-selective amine addition: Method A: A mixture of (2-Chloro-5-trifluoromethyl-pyrimidin-4-yl)-p-tolyl-amine (9) and (4-Chloro-5-trifluoromethyl-pyrimidin-2-y))-p-tolyI-amine (8) To a solution of 5- trifluoromethyl-2,4-dichloropyrimidine (500 mg; 2.3 mmol) in DCE/t-butanol (20 mL) was added 4-methylaniline (247 mg; 1 eq) followed by dropwise addition triethylamine (1.1 eq).
- N,N-dimethyl-1 ,4-phenylenediamine (313 mg; 1 eq) was added followed by dropwise addition of a solution of triethylamine (279 mg; 1.1 eq) in 5 mL of DCE/f-BuOH. After stirring for 24 hours the reaction was concentrated. The product was obtained as a pale green solid (531 mg; 73%) following crystallization from 25% H 2 0/methanol.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Catalysts (AREA)
Abstract
Description
Claims
Priority Applications (15)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002537373A CA2537373C (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of cf3-substituted pyrimidines |
| EP04769170A EP1663991B1 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of cf3-substituted pyrimidines |
| SI200430175T SI1663991T1 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of cf3-substituted pyrimidines |
| BRPI0414059A BRPI0414059B8 (en) | 2003-09-05 | 2004-08-24 | selective synthesis of substituted pyrimidines in cf3 |
| DK04769170T DK1663991T3 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of CF3-substituted pyrimidines |
| PL04769170T PL1663991T3 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of cf3-substituted pyrimidines |
| DE602004004287T DE602004004287T2 (en) | 2003-09-05 | 2004-08-24 | SELECTIVE SYNTHESIS OF CF3-SUBSTITUTED PYRIMIDINES |
| AU2004270480A AU2004270480B2 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of CF3-substituted pyrimidines |
| NZ544969A NZ544969A (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of CF3-substituted pyrimidines |
| JP2006525198A JP4842816B2 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of CF3-substituted pyrimidines |
| MXPA06002552A MXPA06002552A (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of cf3-substituted pyrimidines. |
| IL173363A IL173363A (en) | 2003-09-05 | 2006-01-25 | Selective synthesis of cf 3-substituted pyrimidines |
| NO20061509A NO334383B1 (en) | 2003-09-05 | 2006-04-03 | Process for the preparation of CF3-substituted pyrimidines |
| HK06112349.7A HK1091824A1 (en) | 2003-09-05 | 2006-11-09 | Selectives synthesis of cf3-substituted pyrimidines |
| CY20071100341T CY1106370T1 (en) | 2003-09-05 | 2007-03-12 | SELECTIVE SYNTHESIS OF CF3-SUBSTITUTED PYRIMIDINES |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US50073303P | 2003-09-05 | 2003-09-05 | |
| US60/500,733 | 2003-09-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005023780A1 true WO2005023780A1 (en) | 2005-03-17 |
Family
ID=34272990
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2004/002744 WO2005023780A1 (en) | 2003-09-05 | 2004-08-24 | Selective synthesis of cf3-substituted pyrimidines |
Country Status (24)
| Country | Link |
|---|---|
| US (1) | US7122670B2 (en) |
| EP (1) | EP1663991B1 (en) |
| JP (1) | JP4842816B2 (en) |
| KR (1) | KR100694732B1 (en) |
| CN (1) | CN100465164C (en) |
| AR (1) | AR045557A1 (en) |
| BR (1) | BRPI0414059B8 (en) |
| CA (1) | CA2537373C (en) |
| CO (1) | CO5650237A2 (en) |
| CY (1) | CY1106370T1 (en) |
| DE (1) | DE602004004287T2 (en) |
| DK (1) | DK1663991T3 (en) |
| ES (1) | ES2276344T3 (en) |
| HK (1) | HK1091824A1 (en) |
| IL (1) | IL173363A (en) |
| MX (1) | MXPA06002552A (en) |
| NO (1) | NO334383B1 (en) |
| NZ (1) | NZ544969A (en) |
| PL (1) | PL1663991T3 (en) |
| PT (1) | PT1663991E (en) |
| RU (1) | RU2315759C2 (en) |
| TW (1) | TWI283667B (en) |
| WO (1) | WO2005023780A1 (en) |
| ZA (1) | ZA200600747B (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006265207A (en) * | 2005-03-25 | 2006-10-05 | Fuji Photo Film Co Ltd | Method for producing heterocyclic compound |
| DE102007010801A1 (en) | 2007-03-02 | 2008-09-04 | Bayer Cropscience Ag | Use of new and known 2,4-diaminopyrimidine derivatives as fungicides, especially for controlling phytopathogenic fungi |
| WO2009115583A1 (en) * | 2008-03-20 | 2009-09-24 | Boehringer Ingelheim International Gmbh | Regioselective preparation of substituted pyrimidines |
| WO2009115587A1 (en) * | 2008-03-20 | 2009-09-24 | Boehringer Ingelheim International Gmbh | Regioselective preparation of substituted pyrimidines |
| EP2213663A1 (en) | 2009-01-29 | 2010-08-04 | Saltigo GmbH | Preparation of halogenated N-heteroaromatic polyhalogen derivatives |
| WO2011018517A1 (en) | 2009-08-14 | 2011-02-17 | Boehringer Ingelheim International Gmbh | Regioselective preparation of 2-amino-5-trifluoromethylpyrimidine derivatives |
| WO2011018518A1 (en) | 2009-08-14 | 2011-02-17 | Boehringer Ingelheim International Gmbh | Regioselective preparation of 2 -amino-5-trifluoromethylpyrimidine derivatives |
| WO2012085126A1 (en) | 2010-12-21 | 2012-06-28 | Boehringer Ingelheim International Gmbh | Oxindolopyrimidine as igf1r receptor inhibitors |
| WO2012110774A1 (en) | 2011-02-17 | 2012-08-23 | Cancer Therapeutics Crc Pty Limited | Selective fak inhibitors |
| WO2012110773A1 (en) | 2011-02-17 | 2012-08-23 | Cancer Therapeutics Crc Pty Limited | Fak inhibitors |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004056807A1 (en) * | 2002-12-20 | 2004-07-08 | Pfizer Products Inc. | Pyrimidine derivatives for the treatment of abnormal cell growth |
| US20060205945A1 (en) * | 2004-05-14 | 2006-09-14 | Pfizer Inc | Pyrimidine derivatives for the treatment of abnormal cell growth |
| MX2009011090A (en) | 2007-04-18 | 2009-11-02 | Pfizer Prod Inc | Sulfonyl amide derivatives for the treatment of abnormal cell growth. |
| KR101294731B1 (en) * | 2007-06-04 | 2013-08-16 | 삼성디스플레이 주식회사 | Array substrate, display panel having the array substrate and method of manufacturing the array substrate |
| TW201100441A (en) | 2009-06-01 | 2011-01-01 | Osi Pharm Inc | Amino pyrimidine anticancer compounds |
| PT2646448T (en) | 2010-11-29 | 2017-10-04 | Osi Pharmaceuticals Llc | Macrocyclic kinase inhibitors |
| WO2014126954A1 (en) | 2013-02-13 | 2014-08-21 | OSI Pharmaceuticals, LLC | Regioselective synthesis of substituted pyrimidines |
| GB2566622B (en) * | 2014-05-08 | 2019-07-10 | Tosoh F Tech Inc | 5-(Trifluoromethyl)pyrimidine derivatives and method for producing same |
| JP6391985B2 (en) * | 2014-05-16 | 2018-09-19 | 東ソー・ファインケム株式会社 | 2-substituted-4- (2,2,2-trifluoroethoxy) -5- (trifluoromethyl) pyrimidine derivative and method for producing the same |
| JP6391988B2 (en) * | 2014-05-21 | 2018-09-19 | 東ソー・ファインケム株式会社 | Process for producing 5- (trifluoromethyl) pyrimidine derivative and novel 5- (trifluoromethyl) pyrimidine derivative |
| CN108440499A (en) * | 2018-03-22 | 2018-08-24 | 盐城师范学院 | A kind of preparation method of Ceritinib and its key intermediate |
| CN114213339B (en) * | 2021-12-20 | 2023-10-31 | 苏州康纯医药科技有限公司 | Preparation method of epidermal cell growth factor receptor mutation inhibitor |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003030909A1 (en) * | 2001-09-25 | 2003-04-17 | Bayer Pharmaceuticals Corporation | 2- and 4-aminopyrimidines n-substtituded by a bicyclic ring for use as kinase inhibitors in the treatment of cancer |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2090571B1 (en) * | 2001-10-17 | 2012-05-16 | Boehringer Ingelheim Pharma GmbH & Co. KG | Pyrimidine derivates, medicaments comprising them, their use and process of their preparation |
-
2004
- 2004-08-24 KR KR1020067004428A patent/KR100694732B1/en active IP Right Grant
- 2004-08-24 DK DK04769170T patent/DK1663991T3/en active
- 2004-08-24 CN CNB2004800254318A patent/CN100465164C/en active Active
- 2004-08-24 BR BRPI0414059A patent/BRPI0414059B8/en active IP Right Grant
- 2004-08-24 EP EP04769170A patent/EP1663991B1/en active Active
- 2004-08-24 ES ES04769170T patent/ES2276344T3/en active Active
- 2004-08-24 PT PT04769170T patent/PT1663991E/en unknown
- 2004-08-24 CA CA002537373A patent/CA2537373C/en active Active
- 2004-08-24 JP JP2006525198A patent/JP4842816B2/en active Active
- 2004-08-24 RU RU2006106727/04A patent/RU2315759C2/en not_active IP Right Cessation
- 2004-08-24 PL PL04769170T patent/PL1663991T3/en unknown
- 2004-08-24 NZ NZ544969A patent/NZ544969A/en unknown
- 2004-08-24 WO PCT/IB2004/002744 patent/WO2005023780A1/en active IP Right Grant
- 2004-08-24 DE DE602004004287T patent/DE602004004287T2/en active Active
- 2004-08-24 MX MXPA06002552A patent/MXPA06002552A/en active IP Right Grant
- 2004-08-27 US US10/928,954 patent/US7122670B2/en active Active
- 2004-09-02 AR ARP040103140A patent/AR045557A1/en not_active Application Discontinuation
- 2004-09-03 TW TW093126748A patent/TWI283667B/en active
-
2006
- 2006-01-25 IL IL173363A patent/IL173363A/en active IP Right Grant
- 2006-01-26 ZA ZA200600747A patent/ZA200600747B/en unknown
- 2006-02-23 CO CO06018320A patent/CO5650237A2/en not_active Application Discontinuation
- 2006-04-03 NO NO20061509A patent/NO334383B1/en unknown
- 2006-11-09 HK HK06112349.7A patent/HK1091824A1/en unknown
-
2007
- 2007-03-12 CY CY20071100341T patent/CY1106370T1/en unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003030909A1 (en) * | 2001-09-25 | 2003-04-17 | Bayer Pharmaceuticals Corporation | 2- and 4-aminopyrimidines n-substtituded by a bicyclic ring for use as kinase inhibitors in the treatment of cancer |
Non-Patent Citations (1)
| Title |
|---|
| DIRINGER ET AL.: "Fluorinated pyrimidines. XXXVI. Synthesis of some 2,-substituted 5-trifluoromethylpyrimidines", JOURNAL OF MEDICINAL CHEMISTRY, vol. 13, no. 1, 1970, pages 151 - 152, XP002304586 * |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006265207A (en) * | 2005-03-25 | 2006-10-05 | Fuji Photo Film Co Ltd | Method for producing heterocyclic compound |
| DE102007010801A1 (en) | 2007-03-02 | 2008-09-04 | Bayer Cropscience Ag | Use of new and known 2,4-diaminopyrimidine derivatives as fungicides, especially for controlling phytopathogenic fungi |
| US8334383B2 (en) | 2008-03-20 | 2012-12-18 | Boehringer Ingelheim International Gmbh | Regioselective preparation of substituted pyrimidines |
| WO2009115583A1 (en) * | 2008-03-20 | 2009-09-24 | Boehringer Ingelheim International Gmbh | Regioselective preparation of substituted pyrimidines |
| WO2009115587A1 (en) * | 2008-03-20 | 2009-09-24 | Boehringer Ingelheim International Gmbh | Regioselective preparation of substituted pyrimidines |
| US8372974B2 (en) | 2008-03-20 | 2013-02-12 | Boehringer Ingelheim International Gmbh | Regioselective preparation of substituted pyrimidines |
| JP2011515372A (en) * | 2008-03-20 | 2011-05-19 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | Regioselective process for the preparation of substituted pyrimidines |
| EP2213663A1 (en) | 2009-01-29 | 2010-08-04 | Saltigo GmbH | Preparation of halogenated N-heteroaromatic polyhalogen derivatives |
| DE102009006643A1 (en) | 2009-01-29 | 2010-08-05 | Saltigo Gmbh | Preparation of halogenated N-heteroaromatic polyhalogen derivatives |
| US8933227B2 (en) | 2009-08-14 | 2015-01-13 | Boehringer Ingelheim International Gmbh | Selective synthesis of functionalized pyrimidines |
| WO2011018518A1 (en) | 2009-08-14 | 2011-02-17 | Boehringer Ingelheim International Gmbh | Regioselective preparation of 2 -amino-5-trifluoromethylpyrimidine derivatives |
| US8729265B2 (en) | 2009-08-14 | 2014-05-20 | Boehringer Ingelheim International Gmbh | Regioselective preparation of 2-amino-5-trifluoromethylpyrimidine derivatives |
| WO2011018517A1 (en) | 2009-08-14 | 2011-02-17 | Boehringer Ingelheim International Gmbh | Regioselective preparation of 2-amino-5-trifluoromethylpyrimidine derivatives |
| WO2012085126A1 (en) | 2010-12-21 | 2012-06-28 | Boehringer Ingelheim International Gmbh | Oxindolopyrimidine as igf1r receptor inhibitors |
| WO2012110774A1 (en) | 2011-02-17 | 2012-08-23 | Cancer Therapeutics Crc Pty Limited | Selective fak inhibitors |
| WO2012110773A1 (en) | 2011-02-17 | 2012-08-23 | Cancer Therapeutics Crc Pty Limited | Fak inhibitors |
| US9012461B2 (en) | 2011-02-17 | 2015-04-21 | Cancer Therapeutics Crc Pty Ltd | FAK inhibitors |
| US9120761B2 (en) | 2011-02-17 | 2015-09-01 | Cancer Therapeutics Crc Pty Ltd | Selective FAK inhibitors |
| US9174946B2 (en) | 2011-02-17 | 2015-11-03 | Cancer Therapeutics Crc Pty Ltd | Selective FAK inhibitors |
| US9421205B2 (en) | 2011-02-17 | 2016-08-23 | Cancer Therapeutics CRC Pty Ltd. | FAK inhibitors |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1663991B1 (en) | Selective synthesis of cf3-substituted pyrimidines | |
| CA2517774C (en) | Substituted 8-perfluoroalkyl-6,7,8,9-tetrahydropyrimido[1,2-a] pyrimidin-4-one derivatives | |
| US20100228028A1 (en) | Processes for the manufacture of rosuvastatin and intermediates | |
| US20100292468A1 (en) | Process for making thienopyrimidine compounds | |
| EP2752407A1 (en) | Crystalline rosuvastatin calcium trihydrate | |
| CA2961984C (en) | Novel chiral synthesis of n-acyl-(3-substituted)-(8-substituted)-5,6-dihydro-[1,2,4]triazolo[4,3-a]pyrazines | |
| CA3070069C (en) | Improved process for preparing aminopyrimidine derivatives | |
| KR20210136995A (en) | Bruton's Tyrosine Kinase Inhibitors | |
| US7309788B2 (en) | Method for preparing pyrimidinone compound and pharmaceutically acceptable salts thereof | |
| US6169086B1 (en) | Pyrazole derivatives | |
| CA2637545A1 (en) | Anabaseine derivatives, pharmaceutical compositions and methods of use thereof | |
| AU644295B2 (en) | 2-aminopyrimidine-4-carboxamide derivatives, their preparation and their use in therapeutics | |
| US20060063761A1 (en) | Novel benzothiazole compounds and methods of use thereof | |
| US7314877B2 (en) | Benzofuran derivative | |
| JP7112755B2 (en) | JAK enzyme inhibitor and its production method and use | |
| AU2004270480B2 (en) | Selective synthesis of CF3-substituted pyrimidines | |
| TWI432435B (en) | Thiazolyl-pyrazolopyrimidine compounds as synthetic intermediates and related synthetic processes | |
| WO2021240429A1 (en) | Benzofuran and benzopyran dihydroorotate dehydrogenase inhibitors | |
| CN111763217B (en) | Thieno-nitrogen heterocyclic compounds, preparation method and application | |
| WO2023122600A1 (en) | Piperazine indazole glucocorticoid receptor antagonists | |
| CN116425796A (en) | Pyrimidine heterocyclic compounds, preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 1200600460 Country of ref document: VN Ref document number: 200480025431.8 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006/00747 Country of ref document: ZA Ref document number: 544969 Country of ref document: NZ Ref document number: 200600747 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 465/DELNP/2006 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004270480 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12006500380 Country of ref document: PH |
|
| ENP | Entry into the national phase |
Ref document number: 2004270480 Country of ref document: AU Date of ref document: 20040824 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 06018320 Country of ref document: CO |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004270480 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2537373 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2006/002552 Country of ref document: MX Ref document number: 1020067004428 Country of ref document: KR Ref document number: 2006525198 Country of ref document: JP Ref document number: 2006106727 Country of ref document: RU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004769170 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067004428 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004769170 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: PI0414059 Country of ref document: BR |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2004769170 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1020067004428 Country of ref document: KR |