WO2005001002A2 - Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of fischer-tropsch products - Google Patents
Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of fischer-tropsch products Download PDFInfo
- Publication number
- WO2005001002A2 WO2005001002A2 PCT/US2004/019252 US2004019252W WO2005001002A2 WO 2005001002 A2 WO2005001002 A2 WO 2005001002A2 US 2004019252 W US2004019252 W US 2004019252W WO 2005001002 A2 WO2005001002 A2 WO 2005001002A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fuel
- distillate fuel
- blend stock
- weight
- fischer
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 248
- 239000000446 fuel Substances 0.000 title claims abstract description 218
- 239000011593 sulfur Substances 0.000 claims abstract description 40
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 40
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims abstract description 39
- 238000000034 method Methods 0.000 claims description 119
- 239000003054 catalyst Substances 0.000 claims description 91
- 230000008569 process Effects 0.000 claims description 90
- 150000002978 peroxides Chemical class 0.000 claims description 85
- 150000001336 alkenes Chemical class 0.000 claims description 54
- 239000002283 diesel fuel Substances 0.000 claims description 44
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 27
- 229910052739 hydrogen Inorganic materials 0.000 claims description 26
- 239000001257 hydrogen Substances 0.000 claims description 26
- 238000003860 storage Methods 0.000 claims description 26
- 239000003208 petroleum Substances 0.000 claims description 24
- 239000002243 precursor Substances 0.000 claims description 23
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 claims description 21
- 230000002378 acidificating effect Effects 0.000 claims description 16
- 239000007788 liquid Substances 0.000 claims description 16
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 15
- 238000004519 manufacturing process Methods 0.000 claims description 11
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 10
- 229910052760 oxygen Inorganic materials 0.000 claims description 10
- 239000001301 oxygen Substances 0.000 claims description 10
- 229910052757 nitrogen Inorganic materials 0.000 claims description 8
- 238000002156 mixing Methods 0.000 claims description 5
- 239000000779 smoke Substances 0.000 claims description 5
- 230000009467 reduction Effects 0.000 claims description 3
- 239000012188 paraffin wax Substances 0.000 abstract description 7
- 230000000717 retained effect Effects 0.000 abstract description 5
- 238000002485 combustion reaction Methods 0.000 abstract description 3
- 239000000047 product Substances 0.000 description 113
- 239000001993 wax Substances 0.000 description 33
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 31
- 238000006243 chemical reaction Methods 0.000 description 27
- 230000015572 biosynthetic process Effects 0.000 description 26
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 24
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 23
- 229930195733 hydrocarbon Natural products 0.000 description 20
- 150000002430 hydrocarbons Chemical class 0.000 description 20
- 238000002290 gas chromatography-mass spectrometry Methods 0.000 description 17
- 238000004517 catalytic hydrocracking Methods 0.000 description 16
- 229910052751 metal Inorganic materials 0.000 description 16
- 239000002184 metal Substances 0.000 description 16
- 239000000654 additive Substances 0.000 description 15
- 125000003118 aryl group Chemical group 0.000 description 15
- 238000012360 testing method Methods 0.000 description 15
- DCAYPVUWAIABOU-UHFFFAOYSA-N hexadecane Chemical compound CCCCCCCCCCCCCCCC DCAYPVUWAIABOU-UHFFFAOYSA-N 0.000 description 14
- 239000000377 silicon dioxide Substances 0.000 description 14
- 239000004215 Carbon black (E152) Substances 0.000 description 13
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 13
- 238000005984 hydrogenation reaction Methods 0.000 description 12
- 239000000463 material Substances 0.000 description 12
- 238000004808 supercritical fluid chromatography Methods 0.000 description 12
- 239000010457 zeolite Substances 0.000 description 12
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 11
- 230000003197 catalytic effect Effects 0.000 description 10
- 239000007789 gas Substances 0.000 description 10
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 10
- 229910002092 carbon dioxide Inorganic materials 0.000 description 9
- 150000001875 compounds Chemical class 0.000 description 9
- 239000003921 oil Substances 0.000 description 9
- 229910052697 platinum Inorganic materials 0.000 description 9
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 8
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 229910044991 metal oxide Inorganic materials 0.000 description 7
- 150000004706 metal oxides Chemical class 0.000 description 7
- 150000002739 metals Chemical class 0.000 description 7
- 229910052759 nickel Inorganic materials 0.000 description 7
- 229910000510 noble metal Inorganic materials 0.000 description 7
- 238000003786 synthesis reaction Methods 0.000 description 7
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 6
- 229910021536 Zeolite Inorganic materials 0.000 description 6
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 6
- 239000007795 chemical reaction product Substances 0.000 description 6
- 229910052681 coesite Inorganic materials 0.000 description 6
- 229910052593 corundum Inorganic materials 0.000 description 6
- 229910052906 cristobalite Inorganic materials 0.000 description 6
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 6
- 238000005259 measurement Methods 0.000 description 6
- 229910052763 palladium Inorganic materials 0.000 description 6
- 239000002904 solvent Substances 0.000 description 6
- 238000007655 standard test method Methods 0.000 description 6
- 229910052682 stishovite Inorganic materials 0.000 description 6
- 229910052905 tridymite Inorganic materials 0.000 description 6
- 229910001845 yogo sapphire Inorganic materials 0.000 description 6
- 150000001298 alcohols Chemical class 0.000 description 5
- 150000001335 aliphatic alkanes Chemical class 0.000 description 5
- 229910017052 cobalt Inorganic materials 0.000 description 5
- 239000010941 cobalt Substances 0.000 description 5
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 5
- 125000005842 heteroatom Chemical group 0.000 description 5
- 238000006317 isomerization reaction Methods 0.000 description 5
- 239000011159 matrix material Substances 0.000 description 5
- -1 polycyclic aromatic compounds Chemical class 0.000 description 5
- 239000002002 slurry Substances 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 description 4
- 239000001569 carbon dioxide Substances 0.000 description 4
- 239000000356 contaminant Substances 0.000 description 4
- 238000005336 cracking Methods 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 229910052805 deuterium Inorganic materials 0.000 description 4
- 238000004821 distillation Methods 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229920001971 elastomer Polymers 0.000 description 4
- 239000000806 elastomer Substances 0.000 description 4
- 239000000395 magnesium oxide Substances 0.000 description 4
- 239000003209 petroleum derivative Substances 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 229910052709 silver Inorganic materials 0.000 description 4
- 241000894007 species Species 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 150000003464 sulfur compounds Chemical class 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 3
- 238000004566 IR spectroscopy Methods 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 238000009835 boiling Methods 0.000 description 3
- 239000006227 byproduct Substances 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 229910002091 carbon monoxide Inorganic materials 0.000 description 3
- 230000000052 comparative effect Effects 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- 229910052736 halogen Inorganic materials 0.000 description 3
- 150000002367 halogens Chemical class 0.000 description 3
- 229910052742 iron Inorganic materials 0.000 description 3
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 3
- 239000003350 kerosene Substances 0.000 description 3
- 239000002808 molecular sieve Substances 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 229910052702 rhenium Inorganic materials 0.000 description 3
- 229910052707 ruthenium Inorganic materials 0.000 description 3
- 239000004332 silver Substances 0.000 description 3
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 3
- 238000001179 sorption measurement Methods 0.000 description 3
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 3
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 3
- 229910052721 tungsten Inorganic materials 0.000 description 3
- 239000010937 tungsten Substances 0.000 description 3
- NHTMVDHEPJAVLT-UHFFFAOYSA-N Isooctane Chemical compound CC(C)CC(C)(C)C NHTMVDHEPJAVLT-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 2
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 2
- 239000011959 amorphous silica alumina Substances 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- KBPLFHHGFOOTCA-UHFFFAOYSA-N caprylic alcohol Natural products CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 2
- 239000004927 clay Substances 0.000 description 2
- GUJOJGAPFQRJSV-UHFFFAOYSA-N dialuminum;dioxosilane;oxygen(2-);hydrate Chemical compound O.[O-2].[O-2].[O-2].[Al+3].[Al+3].O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O GUJOJGAPFQRJSV-UHFFFAOYSA-N 0.000 description 2
- JVSWJIKNEAIKJW-UHFFFAOYSA-N dimethyl-hexane Natural products CCCCCC(C)C JVSWJIKNEAIKJW-UHFFFAOYSA-N 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- HYBBIBNJHNGZAN-UHFFFAOYSA-N furfural Chemical compound O=CC1=CC=CO1 HYBBIBNJHNGZAN-UHFFFAOYSA-N 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 239000012535 impurity Substances 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- DDTIGTPWGISMKL-UHFFFAOYSA-N molybdenum nickel Chemical compound [Ni].[Mo] DDTIGTPWGISMKL-UHFFFAOYSA-N 0.000 description 2
- 229910052901 montmorillonite Inorganic materials 0.000 description 2
- TVMXDCGIABBOFY-UHFFFAOYSA-N n-Octanol Natural products CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 2
- MOWMLACGTDMJRV-UHFFFAOYSA-N nickel tungsten Chemical compound [Ni].[W] MOWMLACGTDMJRV-UHFFFAOYSA-N 0.000 description 2
- 238000006384 oligomerization reaction Methods 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 238000012797 qualification Methods 0.000 description 2
- 238000011002 quantification Methods 0.000 description 2
- 238000002407 reforming Methods 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- DLYUQMMRRRQYAE-UHFFFAOYSA-N tetraphosphorus decaoxide Chemical compound O1P(O2)(=O)OP3(=O)OP1(=O)OP2(=O)O3 DLYUQMMRRRQYAE-UHFFFAOYSA-N 0.000 description 2
- RSJKGSCJYJTIGS-UHFFFAOYSA-N undecane Chemical compound CCCCCCCCCCC RSJKGSCJYJTIGS-UHFFFAOYSA-N 0.000 description 2
- CRSBERNSMYQZNG-UHFFFAOYSA-N 1 -dodecene Natural products CCCCCCCCCCC=C CRSBERNSMYQZNG-UHFFFAOYSA-N 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 241000269350 Anura Species 0.000 description 1
- DCERHCFNWRGHLK-UHFFFAOYSA-N C[Si](C)C Chemical class C[Si](C)C DCERHCFNWRGHLK-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 1
- 229910003294 NiMo Inorganic materials 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- 229910004369 ThO2 Inorganic materials 0.000 description 1
- 229910052776 Thorium Inorganic materials 0.000 description 1
- 229910052770 Uranium Inorganic materials 0.000 description 1
- 238000010306 acid treatment Methods 0.000 description 1
- 239000012670 alkaline solution Substances 0.000 description 1
- 150000004996 alkyl benzenes Chemical class 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 1
- 229960000892 attapulgite Drugs 0.000 description 1
- LTPBRCUWZOMYOC-UHFFFAOYSA-N beryllium oxide Inorganic materials O=[Be] LTPBRCUWZOMYOC-UHFFFAOYSA-N 0.000 description 1
- 230000001588 bifunctional effect Effects 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- WHDPTDWLEKQKKX-UHFFFAOYSA-N cobalt molybdenum Chemical compound [Co].[Co].[Mo] WHDPTDWLEKQKKX-UHFFFAOYSA-N 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 150000001924 cycloalkanes Chemical class 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 239000006280 diesel fuel additive Substances 0.000 description 1
- 230000003292 diminished effect Effects 0.000 description 1
- 229940069096 dodecene Drugs 0.000 description 1
- 238000010812 external standard method Methods 0.000 description 1
- RLQJEEJISHYWON-UHFFFAOYSA-N flonicamid Chemical compound FC(F)(F)C1=CC=NC=C1C(=O)NCC#N RLQJEEJISHYWON-UHFFFAOYSA-N 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000002816 fuel additive Substances 0.000 description 1
- 239000000295 fuel oil Substances 0.000 description 1
- 239000003502 gasoline Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 229910052735 hafnium Inorganic materials 0.000 description 1
- 239000010763 heavy fuel oil Substances 0.000 description 1
- DCAYPVUWAIABOU-NJFSPNSNSA-N hexadecane Chemical class CCCCCCCCCCCCCCC[14CH3] DCAYPVUWAIABOU-NJFSPNSNSA-N 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 229910052746 lanthanum Inorganic materials 0.000 description 1
- MRELNEQAGSRDBK-UHFFFAOYSA-N lanthanum oxide Inorganic materials [O-2].[O-2].[O-2].[La+3].[La+3] MRELNEQAGSRDBK-UHFFFAOYSA-N 0.000 description 1
- 239000012263 liquid product Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000010687 lubricating oil Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- VUZPPFZMUPKLLV-UHFFFAOYSA-N methane;hydrate Chemical compound C.O VUZPPFZMUPKLLV-UHFFFAOYSA-N 0.000 description 1
- 229910003455 mixed metal oxide Inorganic materials 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 229910052680 mordenite Inorganic materials 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- 239000005445 natural material Substances 0.000 description 1
- CLDVQCMGOSGNIW-UHFFFAOYSA-N nickel tin Chemical compound [Ni].[Sn] CLDVQCMGOSGNIW-UHFFFAOYSA-N 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 229910052762 osmium Inorganic materials 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- KTUFCUMIWABKDW-UHFFFAOYSA-N oxo(oxolanthaniooxy)lanthanum Chemical compound O=[La]O[La]=O KTUFCUMIWABKDW-UHFFFAOYSA-N 0.000 description 1
- 229910052625 palygorskite Inorganic materials 0.000 description 1
- 125000004817 pentamethylene group Chemical class [H]C([H])([*:2])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 238000000275 quality assurance Methods 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 239000012925 reference material Substances 0.000 description 1
- 239000003870 refractory metal Substances 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 239000012086 standard solution Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 150000004763 sulfides Chemical class 0.000 description 1
- ZCUFMDLYAMJYST-UHFFFAOYSA-N thorium dioxide Chemical compound O=[Th]=O ZCUFMDLYAMJYST-UHFFFAOYSA-N 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- ABDKAPXRBAPSQN-UHFFFAOYSA-N veratrole Chemical compound COC1=CC=CC=C1OC ABDKAPXRBAPSQN-UHFFFAOYSA-N 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/04—Liquid carbonaceous fuels essentially based on blends of hydrocarbons
- C10L1/08—Liquid carbonaceous fuels essentially based on blends of hydrocarbons for compression ignition
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G57/00—Treatment of hydrocarbon oils, in the absence of hydrogen, by at least one cracking process or refining process and at least one other conversion process
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10G—CRACKING HYDROCARBON OILS; PRODUCTION OF LIQUID HYDROCARBON MIXTURES, e.g. BY DESTRUCTIVE HYDROGENATION, OLIGOMERISATION, POLYMERISATION; RECOVERY OF HYDROCARBON OILS FROM OIL-SHALE, OIL-SAND, OR GASES; REFINING MIXTURES MAINLY CONSISTING OF HYDROCARBONS; REFORMING OF NAPHTHA; MINERAL WAXES
- C10G61/00—Treatment of naphtha by at least one reforming process and at least one process of refining in the absence of hydrogen
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/04—Liquid carbonaceous fuels essentially based on blends of hydrocarbons
Definitions
- Tropsch distillate fuel blend stock which have excellent stability and moderate content of unsaturates.
- BACKGROUND OF THE INVENTION Distillate fuel derived from the Fischer-Tropsch process is highly paraffmic and has excellent burning properties and very low sulfur. This makes Fischer-Tropsch products ideally suited for fuel use where environmental concerns are important. Fuels with good or preferably excellent stabilities are always desired, and stable fuels can be produced by hydrotreating or hydrocracking Fischer Tropsch products.
- conventional hydrotreating and hydrocracking processes require the use of expensive hydrogen to saturate olefins and convert oxygenates into paraffins.
- Fuels of this type can be prepared from Fischer-Tropsch products.
- the preparation of distillate fuels from Fischer Tropsch processes is well known. While they are highly paraffmic, Fischer-Tropsch products also contain olefins, alcohols, and traces of other compounds that can cause problems with stability.
- hydroprocessing is used to saturate essentially all the olefins and remove oxygenates.
- hydroprocessing requires the use of expensive hydrogen gas and expensive facilities designed to operate at high pressure.
- ASTM specifications for Diesel Fuel (D985) describe stability measurements for the respective fuels.
- ASTM D6468 "Standard Test Method for High Temperature Stability of Distillate Fuels" is under consideration as a standard test method for a diesel fuel and this test can provide a good measure ofthe stability ofthe fuel. Neat hydrotreated and hydrocracked Fisher Tropsch products typically have excellent stabilities in this test.
- ASTM specifications for Jet Fuel A and A-l (D1655) describe the use ofthe JFTOT test ASTM D3241 with a pass at 260°C. Higher stabilities are often desired.
- Colonial Pipeline's Quality Assurance guidelines from February 2003 (section 3.19.1 on page 3B-33) require fungible aviation kerosene to have a JFTOT stability of 275°C or greater.
- Vardi et al also describe how compounds like tetralin can cause fuels to become unstable with respect to peroxide formation, while polycyclic aromatic compounds like naphthalenes can improve stability.
- Vardi et al. explains that aromatics act as natural antioxidants and notes that natural peroxide inhibitors such as sulfur compounds and polycyclic aromatics can be removed.
- two recent patents from Exxon describe how the peroxide-stability of highly-paraffinic Fischer Tropsch products in unacceptable, but can be improved by the addition of sulfur compounds from other blend components.
- U.S. Patent No. 6,162,956 discloses a Fischer-Tropsch derived distillate fraction blended with either a raw gas field condensate distillate fraction or a mildly hydrotreated condensate fraction to obtain a stable, inhibited distillate fuel.
- the fuel is described as a blend material useful as a distillate fuel or as a blending component for a distillate fuel comprising: (a) a Fischer-Tropsch derived distillate comprising a C 8 - 700°F fraction, and (b) a gas field condensate distillate comprising a C 8 -700°F fraction, wherein the sulfur content ofthe blend material is >1 ppm by wt.
- distillate fuels derived from Fischer-Tropsch processes are hydrotreated to eliminate unsaturated materials, e.g., olefins, and most, if not all, oxygenates. This patent further discloses that the products contain less than or equal to 0.5 wt % unsaturates (olefins and aromatics).
- U.S. Patent No. 6,180,842 discloses a Fischer-Tropsch derived distillate fraction blended with either a raw virgin condensate fraction or a mildly hydrotreated virgin condensate to obtain a stable inhibited distillate fuel.
- the fuel is describes as a blend material useful as a distillate fuel or as a blending component for a distillate fuel comprising (a) a Fischer-Tropsch derived distillate comprising a C 8 -700°F stream and having a sulfur content of less than 1 ppm by wt, and (b) 1-40 wt % of a virgin distillate comprising a C 8 -700°F stream; wherein the sulfur content ofthe blend material is > 2 ppm by wt.
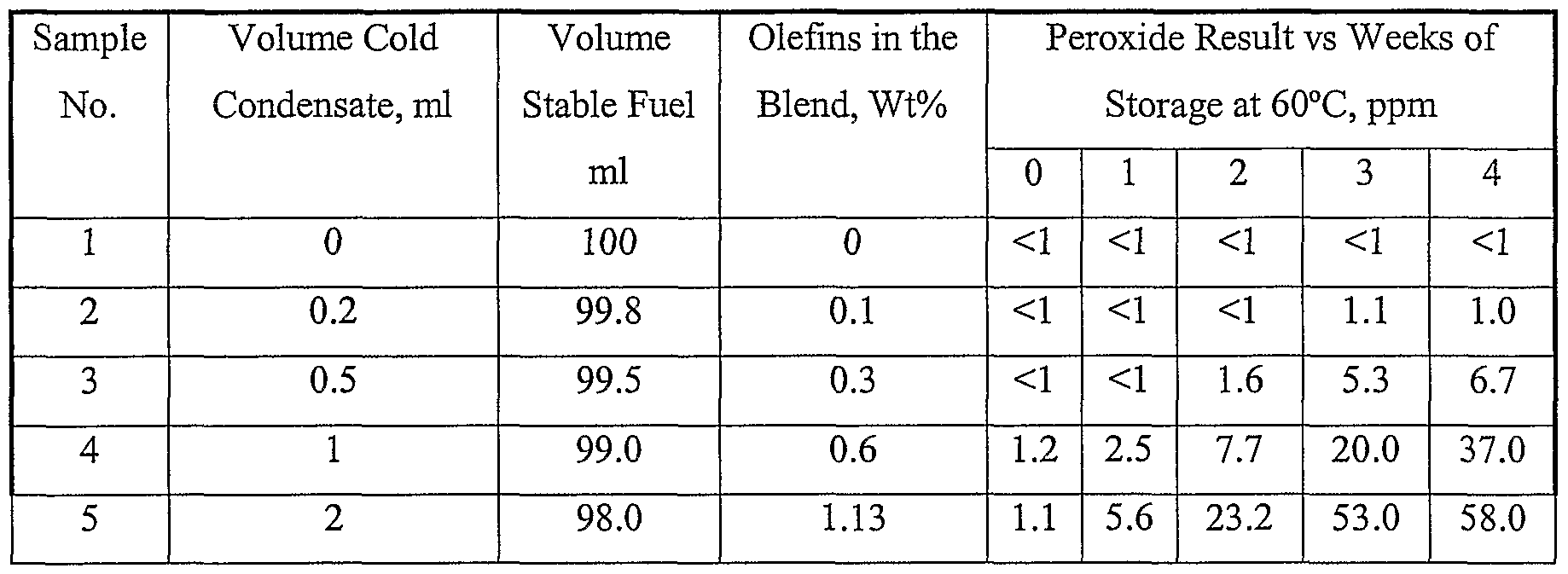
- This patent notes that while there is no standard for the peroxide content of fuels, there is general acceptance that stable fuels have a peroxide number of less than about 5 ppm, preferably less than about 4 ppm, and desirably less than about 1 ppm.
- Fischer Tropsch products having a peroxide number of 24.06 after 4 weeks have unacceptable stability.
- the Fischer Tropsch products in the '842 patent are described as being >80 wt %, preferably >90 wt %, more preferably >95 wt % paraffins, having an iso/normal ratio of 0.1 to 10, preferably 0J to 3.0, more preferably 0.7 to 2.0; sulfur and nitrogen of less than 1 ppm each, preferably less than 0.5, more preferably less than OJ ppm each; ⁇ 0.5 wt % unsaturates (olefins and aromatics), preferably ⁇ O.l wt %; and less than 0.5 wt % oxygen on a water free basis, preferably less than about 0J wt % oxygen, more preferably less than OJ wt % oxygen and most preferably nil oxygen.
- the present invention relates to a distillate fuel comprising a Fischer-Tropsch distillate fuel blend stock.
- the Fischer Tropsch distillate fuel blend stock comprises unsaturates in an amount between 2 and 20 weight %, paraffins in an amount 80 weight % or greater, sulfur in an amount less than 1 ppm, and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks, and the Fischer Tropsch distillate fuel blend stock has a cetane index of greater than 60.
- the present invention relates to a Fischer-Tropsch diesel fuel blend stock.
- the Fischer Tropsch diesel fuel blend stock comprises unsaturates in an amount between 2 and 20 weight %, paraffins in an amount 90 weight % or greater, sulfur in an amount less than 1 ppm and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks.
- the unsaturates comprise less than 20 weight % polynuclear aromatics, preferably less than 10 weight % polynuclear aromatics, and even more preferably less than 5 weight % polynuclear aromatics.
- the unsaturates comprise both olefins and aromatics, and most preferably the olefins are present in amounts greater than or equal to 1 wt%.
- Characteristics ofthe diesel fuel blend stock include a cetane index greater than 60, a percent reflectance according to ASTM D6468 at 150°C in excess of 65% when measured at 90 minutes.
- the present invention relates to a Fischer-Tropsch jet fuel blend stock.
- the Fischer Tropsch jet fuel blend stock comprises unsaturates in an amount between 2 and 10 weight %, paraffins in an amount 90 weight % or greater, sulfur in an amount less than 1 ppm, and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks.
- the unsaturates comprise less than 20 weight % polynuclear aromatics, preferably less than 5 weight % polynuclear aromatics, and even more preferably less than 5 weight % polynuclear aromatics.
- Characteristics of the jet fuel blend stock include a smoke point of 30 mm or greater, and a passing rating in ASTM D3241 (JFTOT Procedure) at 260°C for 2.5 hours.
- the present invention relates to a process for preparing a highly paraffmic, moderately unsaturated distillate fuel blend stock. The process comprises converting syngas to a Fischer Tropsch derived feedstock by a Fischer Tropsch process and hydroprocessing the Fischer-Tropsch derived feedstock. A highly paraffmic, moderately unsaturated distillate fuel blend stock is recovered.
- the highly paraffmic, moderately unsaturated distillate fuel blend stock contains between 2 and 20 weight % unsaturates less than 1 ppm sulfur, and peroxide precursors in an amount such that less than 5 ppm peroxides are fo ⁇ ned after storage at 60°C for four weeks.
- the hydroprocessing conditions include a temperature of 600-750°F, a pressure of less than 1000 psig, and a liquid hourly space velocity of greater than 0J5 hr "1 .
- the present invention relates to a distillate fuel comprising a Fischer Tropsch distillate fuel blend stock, wherein the Fischer Tropsch distillate fuel blend stock is made by a process comprising converting syngas to a Fischer Tropsch derived feedstock by a Fischer Tropsch process; hydroprocessing the Fischer- Tropsch derived feedstock at a temperature of 525-115°F, a pressure of less than 1000 psig, and a liquid hourly space velocity of greater than 0J5 hr "1 ; and recovering a Fischer Tropsch distillate fuel blend stock.
- the Fischer Tropsch distillate fuel blend stock which is recovered, comprises between 2 and 20 weight % unsaturates, less than 1 ppm sulfur, and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks.
- the present invention relates to a method of operating a diesel engine comprising using a Fischer Tropsch diesel fuel blend stock as a diesel fuel wherein the Fischer Tropsch diesel fuel blend stock comprises unsaturates in an amount between 2 and 20 weight %, paraffins in an amount 90 weight % or greater, sulfur in an amount less than 1 ppm and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks.
- the unsaturates comprise less than 20 weight % polynuclear aromatics, preferably less than 10 weight % polynuclear aromatics, and even more preferably less than 5 weight % polynuclear aromatics.
- the unsaturates comprise both olefins and aromatics, and most preferably the olefins are present in amounts greater than or equal to 1 wt%.
- Characteristics ofthe diesel fuel blend stock include a cetane index greater than 60, a percent reflectance according to ASTM D6468 at 150°C in excess of 65% when measured at 90 minutes.
- the present invention relates to a method of operating a jet engine comprising using a Fischer Tropsch jet fuel blend stock as a jet fuel wherein the Fischer Tropsch jet fuel blend stock comprises unsaturates in an amount between 2 and 10 weight %, paraffins in an amount 90 weight % or greater, sulfur in an amount less than 1 ppm, and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks.
- the unsaturates comprise less than 20 weight % polynuclear aromatics, preferably less than 5 weight % polynuclear aromatics, and even more preferably less than 5 weight % polynuclear aromatics.
- Characteristics of the jet fuel blend stock include a smoke point of 30 mm or greater, and a passing rating in ASTM D3241 (JFTOT Procedure) at 260°C for 2.5 hours.
- distillate fuel blend stocks can be produced, which have excellent stability.
- the distillate fuel blend stocks can be prepared by a process including a Fischer-Tropsch synthesis and hydroprocessing under conditions in which a moderate amount of unsaturates are formed or are retained in the product.
- the present invention relates to low sulfur, highly paraffmic, moderately unsaturated distillate fuel blend stocks that have excellent stability and distillate fuels comprising these blend stocks.
- the low sulfur, highly paraffmic, moderately unsaturated distillate fuel blend stocks can be mixed with other blend stocks to provide a distillate fuel or can be used neat directly in an engine as a fuel in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- the term “unsaturates” mean a hydrocarbon containing one or more double or triple bonds, preferably, for example, aromatic and/or olefinic functionality.
- olefins means an unsaturated straight or branched chain hydrocarbon having at least one double bond (i.e., an alkene).
- aromatics means an unsaturated, cyclic and planar hydrocarbon with an uninterrupted cloud of electrons containing an odd number of pairs of B electrons.
- a “distillate fuel blend stock” is a material that is mixed with other distillate fuel blend stocks to provide a distillate fuel, in particular a diesel or jet fuel, as herein defined.
- the blend stock itself does not necessarily meet specifications for the respective fuel, but preferably the resulting combination of blend stocks does.
- Jet fuel blend stocks are combined with other jet fuel blend stocks, and optionally additives, to provide jet fuel.
- diesel fuel blend stocks are combined with other diesel fuel blend stocks, and optionally additives, to provide diesel fuel.
- An "aromatic blend stock” is a blend stock comprising aromatics in an amount greater than or equal to 50 weight %, preferably greater than or equal to 75 weight %, and most preferably greater than or equal to 90 weight%. If a pure aromatic product is used as an aromatic blend stock, analysis ofthe aromatic content is not necessary.
- aromatic blend stock comprises aromatics and other hydrocarbons
- ASTM D6550 Standard Test Method for the Determination ofthe Olefin Content of Gasolines by Supercritical Fluid Chromatography (SFC)
- SFC Supercritical Fluid Chromatography
- An aromatic blend stock may be blended with a Fischer Tropsch distillate fuel blend stock to increase the aromatic content ofthe Fischer Tropsch distillate fuel blend stock.
- aromatic blend stocks include commercially available pure aromatics (for example, benzene, alkylbenzenes, and the like); aromatics obtained from conventional petroleum products; aromatics obtained from reforming of reformable Fischer-Tropsch products; and the like.
- the Cetane Index was determined by ASTM D4737-96a(2001) Standard Test Method for Calculated Cetane Index by Four Variable Equation.
- Conventional petroleum products comprise products derived from petroleum crude.
- a "petroleum blend stock” is a blend stock that comprises conventional petroleum products. Petroleum blend stocks may be comprised ofthe vapor overhead streams from distilling petroleum crude or refined products and the residual fuels that are the non- vaporizable remaining portion. Derived from a Fischer-Tropsch process means that the feedstock, blend stock, or product in question originates from or is produced at some stage by a Fischer-Tropsch process.
- a "Fischer Tropsch distillate fuel blend stock” is a blend stock that originates or is produced at some stage by a Fischer Tropsch process.
- a Fischer Tropsch distillate fuel blend stock can be mixed with other distillate fuel blend stocks to provide a distillate fuel, in particular a diesel or jet fuel.
- the blend stock itself does not necessarily meet specifications for the respective fuel, but the resulting combination of blend stocks does.
- Fischer Tropsch distillate fuel blend stocks include Fischer Tropsch diesel fuel blend stocks and Fischer Tropsch jet fuel blend stocks.
- the Fischer Tropsch distillate fuel blend stocks can be mixed with other blend stocks to provide a distillate fuel, or the Fischer-Tropsch distillate fuel blend stocks can be used neat directly in an engine as a fuel in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- a distillate fuel is a material containing hydrocarbons with boiling points between approximately 60°F to 1100°F.
- a diesel fuel is a material suitable for use in diesel engines.
- a diesel fuel conforms to at least one ofthe following specifications: • ASTM D975 - "Standard Specification for Diesel Fuel Oils" • European Grade CEN 90 • Japanese Fuel Standards JIS K 2204 • The United States National Conference on Weights and Measures (NCWM) 1997 guidelines for premium diesel fuel • The United States Engine Manufacturers Association recommended guidelines for premium diesel fuel (FQP-1A)
- a diesel fuel may be comprised of a combination of blend stocks or a single blend stock in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- a jet fuel is a material suitable for use in turbine engines in aircraft or other uses.
- a jet fuel conforms to at least one ofthe following specifications: • ASTM D 1655, • DEF STAN 91-91/3 (DERD 2494), TURBINE FUEL, AVIATION, • KEROSENE TYPE, JET A-l, NATO CODE: F-35, • International Air Transportation Association (IATA) Guidance Materials for l Aviation, 4th edition, March 2000
- a jet fuel maybe comprised of a combination of blend stocks or a single blend stock in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- a Fischer Tropsch diesel fuel blend stock is a blend stock suitable for use in a diesel engine.
- the Fischer Tropsch diesel fuel blend stock may be mixed with other blend stocks to provide a diesel fuel or may be used in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- a Fischer Tropsch jet fuel blend stock is a blend stock suitable for use in turbine engines in aircraft or other uses.
- the Fischer Tropsch jet fuel blend stock may be mixed with other blend stocks to provide a jet fuel or may be used in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- a highly paraffmic, moderately unsaturated distillate fuel blend stock is a distillate fuel blend stock that contains more than 70 weight % paraffins, 80 weight % or greater paraffins, and most preferably 90 weight % or greater paraffins and 2 to 20 weight % unsaturates, preferably 2 to 15 weight % unsaturates, and most preferably 5 to 10 weight % unsaturates.
- the unsaturates comprise both olefins and aromatics, and most preferably the olefins are present in amounts greater than or equal to 1 wt%.
- a low sulfur highly paraffmic, moderately unsaturated distillate fuel blend stock contains less than 1 ppm sulfur.
- the highly paraffmic, moderately unsaturated distillate fuel blend stock is a Fischer Tropsch distillate fuel blend stock.
- a "Fischer-Tropsch derived feedstock” or “Fischer Tropsch feedstock” is a feedstock that originates from or is produced at some stage by a Fischer Tropsch process.
- a Fischer-Tropsch derived feedstock maybe blended with a petroleum blend stock during processing to provide a blended stream.
- Syngas is a mixture that includes both hydrogen and carbon monoxide.
- water, carbon dioxide, unconverted light hydrocarbon feedstock and various impurities may also be present.
- Hydrocarbonaceous or “hydrocarbon” means a compound or substance that contains hydrogen and carbon atoms, but which can include heteroatoms such as oxygen, sulfur or nitrogen.
- Peroxide precursors mean those components in a hydrocarbon product or feed that will form peroxides and/or cause formation of peroxides in the hydrocarbon product. The peroxide precursors can be identified and the amount of peroxide precursors can be measured by storage ofthe hydrocarbon product in an oven at 60°C for 4 weeks. The peroxide precursors are identified by the formation of peroxides and the amount of peroxide precursors can be measured based on the amount of peroxides formed.
- the peroxide content of samples is measured by use of a procedure for measuring the buildup of peroxides as described in ASTM D3703.
- a procedure for measuring the buildup of peroxides as described in ASTM D3703.
- a 4 ounce sample is placed in a brown bottle and aerated for 3 minutes.
- an aliquot ofthe sample is then tested according to ASTM D3703 with the exception that isooctane is used in place in freon. Tests confirmed that the substitution of solvents for environmental reasons had no significant effect on the measurement results.
- the formation of peroxides can also be measured by Infrared spectroscopy, chemical methods, or by attack on elastomer samples.
- ASTM D6550 Standard Test Method for the Determination ofthe Olefin Content of Gasolines by Supercritical Fluid Chromatography (SFC) was used to determine the group types in the feedstocks and products.
- the modified method uses a 3-point calibration standard to quantify the total amount of saturates, aromatics, oxygenates (polars) and olefins. Calibration standard solutions were prepared using the following compounds: undecane, toluene, n-octanol and dodecene. External standard method was used for quantification and the detection limit for aromatics and oxygenates is 0.1% wt and for olefins is 1.0% wt.
- ASTM D6550 describes the instrument conditions.
- a small aliquot ofthe fuel sample was injected onto a set of two chromatographic columns connected in series and transported using supercritical carbon dioxide as the mobile phase.
- the first column was packed with high surface area silica particles.
- the second column contained high surface area silica particles loaded with silver ions.
- Two switching valves were used to direct the different classes of components through the chromatographic system to the detector. In a forward-flow mode, saturates (normal and branched alkanes and cyclic alkanes) were passed through both columns to the detector, while olefins were trapped on the silver-loaded column and the aromatics and oxygenates are retained on the silica column. Aromatic compounds and oxygenates were subsequently eluted from the silica column to the detector in a back flush mode.
- the Polynuclear Aromatic (PNA) content ofthe products was determined by ASTM D5186-99 Standard Test Method for Determination of Aromatic Content and Polynuclear Aromatic Content of Diesel Fuels and Aviation Turbine Fuels by Supercritical Fluid Chromatography.
- the paraffin content ofthe product was determined by the Supercritical Fluid
- SFC Chromatography
- the naphthenes can then be subtracted from the saturates content (as determined by SFC) to provide a good and proper measurement ofthe paraffin content according to the first equation. If the naphthene content determined in this way is less than zero, it is reported as zero, and zero is used in the calculation of paraffins. Thus, in this case the paraffins are equal to the SFC saturates.
- deuterium labeled standards were used to quantify alkanes, olefins, alcohols, and acids. Selected deuterium labeled compounds were added to the sample of interest as internal standards.
- the 1 mixture of sample and standards were treated with trimethylsilyl (TMS) reagent to form the TMS derivatives, followed by GCMS analysis.
- TMS trimethylsilyl
- the mass spectrometer is a Hewlett-Packard bench top mass spectrometer interfaced to a HP GC with a 60 meter non-polar column.
- the normal alkanes and the branched alkanes were all quantified using deuterium labeled normal alkanes. Olefins, alcohols, and acids were all quantified using the corresponding deuterium labeled compounds.
- the paraffin content ofthe highly paraffmic, moderately unsaturated blend stocks ofthe present invention is at least 70 weight %, preferably 80 weight % or greater, and most preferably 90 weight % or greater.
- the highly paraffmic, moderately unsaturated distillate fuel blend stocks ofthe present invention have excellent combustion properties.
- Characteristic combustion properties ofthe blend stocks ofthe present invention include smoke points in excess of 25 mm, preferably in excess of 30 mm, and cetane indexes in excess of 60, preferably in excess of 65.
- the paraffins consist of a mixture of normal and iso-paraff ⁇ ns with the ratio of iso/normal paraffins in the fuel being between 0J and 10. Higher proportions of iso-paraffins are preferred when the blend stock is intended for use in cold climates (Jet Al or diesel for arctic use).
- the unsaturates content ofthe blend stocks ofthe present invention is between 2 and 20 weight %, preferably between 2 and 15 weight %, and most preferably between 5 and 10 weight %.
- the unsaturates ofthe blend stocks comprise minimal amounts of polynuclear aromatics.
- the unsaturates comprise less than 25 weight % polynuclear aromatics, preferably less than 20 weight % polynuclear aromatics, more preferably less than 10 weight % polynuclear aromatics, and even more preferably less than 5 weight % polynuclear aromatics.
- the unsaturates comprise both olefins and aromatics, and most preferably the olefins are present in amounts greater than or equal to 1 weight %.
- Fuels comprising the highly paraffmic, moderately unsaturated blend stocks ofthe present invention preferably conform to at least one specification for either diesel or jet fuel.
- the fuels may be comprised of a combination of blend stocks or the highly paraffmic, moderately unsaturated blend stock in the absence of other blend stocks with only the optional addition of minor amounts of additives.
- the highly paraffmic, moderately unsaturated blend stocks and fuels comprising this blend stock exhibit at least acceptable, and most often excellent, stability.
- the percent reflectance ofthe diesel fuels comprising the highly paraffmic, moderately unsaturated blend stocks will be in excess of 65% when measured at 90 minutes, preferably in excess of 65% when measured at 180 minutes, and more preferably in excess of 99% when measured at 180 minutes.
- Jet fuels comprising the highly paraffmic, moderately unsaturated blend stocks have a passing rating in ASTM D3241 (JFTOT Procedure) at 260°C for 2.5 hours, preferably a passing rating in ASTM D3241 (JFTOT Procedure) at 270°C for 2.5 hours, and more preferably a passing rating in ASTM D3241 (JFTOT Procedure) at 300°C for 2.5 hours.
- a passing rating corresponds to a tube rating of less than 3 (Code 3) and a pressure drop across a filter of less than 25 mm Hg.
- the blend stocks ofthe present invention, and fuels comprising the blend stocks display acceptable stability according to the conventional tests of stability and acceptable peroxide resistance.
- the blend stocks form less than 5 ppm peroxides after storage at 60°C in an oven for 4 weeks, preferably less than 4 ppm peroxides after storage at 60°C in an oven for 4 weeks, and more preferably less than 1 ppm peroxides after storage at 60°C in an oven for 4 weeks.
- the blend stocks comprise peroxide precursors in such an amount that the fuel forms less than 5 ppm peroxides after storage at 60°C in an overi for 4 weeks, preferably less than 4 ppm peroxides after storage at 60°C in an oven for 4 weeks, and more preferably less than 1 ppm peroxides after storage at 60°C in an oven for 4 weeks.
- the amount of peroxide precursors is measured by storage in an oven at 60°C in an oven for 4 weeks.
- the amount of peroxide precursors can be determined based on the amount of peroxides formed.
- the formation of peroxides can be measured by Infrared spectroscopy, chemical methods, or by attack on elastomer samples.
- blend stocks ofthe present invention and fuels comprising the blend stocks, typically have low sulfur ( ⁇ lppm) and preferably low nitrogen content ( ⁇ 1 ppm). Therefore, environmental emissions of oxides of heteroatoms are minimized. Accordingly, the blend stocks and fuels comprising the blend stocks are desirable as environmentally friendly.
- the blend stocks ofthe present invention can be prepared from Fischer-Tropsch products, hydroprocessed under conditions in which a moderate amount of unsaturates are formed or retained.
- the blend stocks ofthe present invention are at least partially derived from a Fischer-Tropsch process.
- Fischer-Tropsch chemistry syngas is converted to liquid hydrocarbons by contact with a Fischer-Tropsch catalyst under reactive conditions.
- methane and optionally heavier hydrocarbons ethane and heavier
- synthesis gas contains hydrogen and carbon monoxide, and may include minor amounts of carbon dioxide and/or water.
- a mildly alkaline solution e.g., aqueous potassium carbonate
- liquid and gaseous hydrocarbons are formed by contacting a synthesis gas comprising a mixture of H 2 and CO with a Fischer-Tropsch catalyst under suitable temperature and pressure reactive conditions.
- a Fischer- Tropsch reaction can be conducted at temperatures from about 300 to 700°F (149 to 371°C), preferably from about 400 to 550°F (204 to 228°C); pressures of from about 10 to 600 psia, (0.7 to 41 bars), preferably 30 to 300 psia, (2 to 21 bars) and catalyst space velocities of from about 100 to about 10,000 cc/g/hr, preferably 300 to 3,000 cc/g/hr.
- Fischer Tropsch processes can be categorized as either a high temperature Fischer Tropsch process or a low temperature Fischer Tropsch process. The process conditions and the predominate products from the two processes are different.
- a high temperature Fischer Tropsch process is generally carried out at temperatures above 250°C, preferably at or above 350°C.
- High temperature Fischer Tropsch processes provide primarily lower molecular weight olefinic products generally within the C 3 to C 8 range, preferably propylene to pentenes.
- High temperature Fischer Tropsch products can also contain significant amounts of aromatics.
- the high temperature Fischer Tropsch products may be subjected to processes to saturate the aromatics, including reforming processes.
- the olefinic products from the high temperature Fischer Tropsch process are typically further processed by oligomerization and hydrogenation steps to produce a highly branched iso-paraffinic product.
- the products from the high temperature Fischer Tropsch process can be processed so that they meet specifications for gasoline.
- the products from high temperature Fischer Tropsch processes typically have cetane indexes of about 55 since the products are highly branched.
- An example of a high temperature Fischer Tropsch process is the Synthol process used by SASOL, as described in "High Yield High Quality Diesel from Fischer Tropsch Process, Dry, M.E., Chem. S.A., February 1984. Jet fuels have also been produced by high temperature Fischer Tropsch processes, olefin oligomerization, and hydrogenation.
- a high temperature Fischer Tropsch process for making jet fuels is described in "Qualification of SASOL Semi-Synthetic Jet A-l as Commercial Jet Fuel," SwRI-8531, November 1997.
- the jet fuels made by a high temperature Fischer Tropsch process, as described in the reference contain no aromatics or unsaturates.
- the thermal stability, or JFTOT, breakpoint for blends of high temperature Fischer Tropsch derived jet with conventional petroleum-derived is presented in the literature as in excess of 300°C. Therefore the thermal stability, or JFTOT, breakpoint for such semi-synthetic blends is significantly above the specification requirement of 260°C. See “Qualification of SASOL Semi-synthetic Jet A-l as Commercial Jet Fuel", Moses, Stavinoha, and Roets, South West Research Institute Publication SwRI-8531, November 1997.
- researchers working with high temperature Fischer Tropsch products and blends of high temperature Fischer Tropsch products and petroleum-derived components have not noted problems with stability.
- a low temperature Fischer Tropsch process operates at temperatures below 250°C and produces a heavier product.
- the heavier product of a low temperature Fischer Tropsch process commonly contains predominantly wax.
- the products from the low temperature Fischer Tropsch process are typically hydrotreated so that they will have acceptable peroxide stability, as shown by U. S. Patent No. 6,180,842. Accordingly, the products from low temperature Fischer Tropsch processes are typically refined by hydroprocessing operations such as hydrotreating and hydrocracking to provide stable fuels meeting the desired specification.
- the products from low temperature Fischer Tropsch processes are predominantly linear, and even after hydrocracking, these products contain fewer branches than products made from a high temperature Fischer Tropsch process. Fewer branches in the products of low temperature Fischer Tropsch processes provide higher cetane indexes for these products in comparison to the products from the high temperature processes, which have increased branching.
- the low temperature Fischer Tropsch products typically have cetane indexes of greater than 60, and preferably greater than 70.
- the Fischer Tropsch process for making the distillate fuel blend stocks according to the present invention, is a low temperature Fischer Process. Examples of conditions for performing low temperature Fischer-Tropsch type reactions are well known to those of skill in the art. The products may range from C ⁇ to C 2 oo + with a majority in the Cs-C-ioo ⁇ range.
- the reaction can be conducted in a variety of reactor types for example, fixed bed reactors containing one or more catalyst beds, slurry reactors, fluidized bed reactors, or a combination of different type reactors. Such reaction processes and reactors are well known and documented in the literature.
- Slurry Fischer-Tropsch processes which is a preferred process in the practice ofthe invention, utilize superior heat (and mass) transfer characteristics for the strongly exothermic synthesis reaction and are able to produce relatively high molecular weight, paraffmic hydrocarbons when using a cobalt catalyst.
- a syngas comprising a mixture of H 2 and CO is bubbled up as a third phase through a slurry in a reactor which comprises a particulate Fischer-Tropsch type hydrocarbon synthesis catalyst dispersed and suspended in a slurry liquid comprising hydrocarbon products ofthe synthesis reaction which are liquid at the reaction conditions.
- the mole ratio ofthe hydrogen to the carbon monoxide may broadly range from about 0.5 to 4, but is more typically within the range of from about 0.7 to 2.75 and preferably from about 0J to 2.5.
- Suitable Fischer-Tropsch catalysts comprise on or more Group VIII catalytic metals such as Fe, Ni, Co, Ru and Re. Additionally, a suitable catalyst may contain a promoter.
- a preferred Fischer-Tropsch catalyst comprises effective amounts of cobalt and one or more of Re, Ru, Pt, Fe, Ni, Th, Zr, Hf, U, Mg and La on a suitable inorganic support material, preferably one which comprises one or more refractory metal oxides.
- the amount of cobalt present in the catalyst is between about 1 and about 50 weight percent ofthe total catalyst composition.
- the catalysts can also contain basic oxide promoters such as ThO 2 , La 2 O 3 , MgO, and TiO 2 , promoters such as ZrO 2 , noble metals (Pt, Pd, Ru, Rh, Os, Ir), coinage metals (Cu, Ag, Au), and other transition metals such as Fe, Mn, Ni, and Re.
- Support materials including alumina, silica, magnesia and titania or mixtures thereof may be used.
- Preferred supports for cobalt containing catalysts comprise titania.
- catalysts and their preparation are known and illustrative, but nonlimiting examples maybe found, for example, in U.S. Pat. Nos. 4,568,663.
- Certain catalysts are known to provide chain growth probabilities that are relatively low to moderate, and the reaction products include a relatively high proportion of low molecular (C 2-8 ) weight olefins and a relatively low proportion of high molecular weight (C 3 o + ) waxes.
- Certain other catalysts are known to provide relatively high chain growth probabilities, and the reaction products include a relatively low proportion of low molecular (C 2-8 ) weight olefins and a relatively high proportion of high molecular weight (C 3 o + ) waxes.
- the products from low temperature Fischer-Tropsch reactions generally include a light reaction product and a waxy reaction product.
- the waxy reaction product i.e. the wax fraction
- hydrocarbons boiling above about 600°F e.g., vacuum gas oil through heavy paraffins
- Both the light reaction product and the waxy product are substantially paraffmic.
- the waxy product generally comprises greater than 70% normal paraffins, and often greater than 80% normal paraffins.
- Hydroprocessing in general, is well known to those of skill in the art and includes such processes as hydrotreating, hydrocracking, hydrogenation, catalytic dewaxing, or combinations of these processes.
- the hydroprocessing operation ofthe present invention achieves several purposes in one or several reactors, most preferably a single reactor.
- the purposes ofthe hydroprocessing are reducing, or preferably completely removing, heteroatoms such as nitrogen and sulfur. While conventional hydroprocessing typically removes unsaturates or greatly reduces their contents, the hydroprocessing of this invention retains at least a portion ofthe unsaturates or creates aromatics; yet yields a distillate product that has acceptable stability .
- the hydroprocessing may increase the ratio of iso/normal paraffins in the distillate product. Additionally, the hydroprocessing may increase the production of distillate product by converting heavy species. Finally, the hydroprocessing may also be conducted under conditions that create or retain a moderate amount of unsaturates. Hydroprocessing under conditions to create or retain unsaturates may reduce or eliminate net hydrogen consumption in the hydroprocessing process. While the addition of hydrogen is needed to begin the hydroprocessing process, if the aromatics formation is high enough, the amount of hydrogen produced in the process can exceed the amount of hydrogen added to the process. Accordingly, there may be a net hydrogen production from the hydroprocessing ofthe present invention, i.e. the net hydrogen consumption is less than zero. Produced hydrogen can be used for a variety of purposes in a Gas-To-Liquid
- GTL hydrotreatment of Fischer-Tropsch streams to reduce or eliminate olefins and/or heteroatoms.
- produced hydrogen may be reacted with CO 2 produced in the GTL process or recovered from a CO 2 -containing gas source to reduce CO 2 .
- the product from the CO 2 -H 2 reaction can be CO or a Fischer- Tropsch product and the reaction can be conducted in the syngas generator.
- Produced hydrogen may also be used in fuel production as a fuel component that does not form CO 2 emissions.
- the produced fuel can be used to generate process heat, produce electrical energy, and or distill/purify water.
- Typical temperatures for the hydroprocessing of Fischer-Tropsch products to retain unsaturates or generate aromatics are 525-775 °F, preferably 575-725°F.
- Typical pressures for this operation are less than 1000 psig, preferably less than 600 psig, and most preferably between 200 and 500 psig.
- Typical liquid hourly space velocities (LHSV) for this operation are greater than 0J5 hr "1 , preferably between 0.5 and 1.5 hr "1 .
- Typical hydroprocessing catalysts for this operation include catalysts for conventional hydroprocessing operations (described below) or catalysts for hydroisomerization dewaxing, preferably combinations of catalysts for hydroprocessing operations and hydroisomerization dewaxing are used as this combination is less expensive and also permits a concurrent reduction in the product pour point.
- a conventional hydroprocessing catalyst may be used for generating aromatics and retaining unsaturates.
- Hydroprocessing catalysts which are particularly suited for generating aromatics are bifunctional catalysts, which contain both a hydrogenation function and an acidic function.
- the aromatics-forming hydroprocessing catalyst is contrasted, for example, from a conventional hydrotreating catalyst by the presence ofthe acidic function, since hydrotreating catalysts typically include a non-acidic support such as alumina.
- the acidic function is preferably based on a mixture of at least two metal oxides of different valences.
- the preferred mixture of metal oxides includes SiO 2 and Al 2 O 3 ; or Al 2 O 3 , SiO 2 , and P 2 O 5 .
- the mixture of metal oxides may be prepared in such a way as to provide a high dispersion of at least a portion ofthe metal oxides between themselves, for example dispersion ofthe SiO 2 and Al 2 O 3 on an atomic scale rather than separate phases of SiO 2 and Al 2 O 3 .
- the presence of separate phases of SiO 2 and Al 2 O 3 can be determined by an XRD examination. If all ofthe oxides are present as separate phases, the performance ofthe catalyst will be diminished. Examples ofthe acidic function composed of mixed metal oxides are zeolites, crystalline SAPO's, and co-precipitated SiO 2 - Al 2 O 3 . While halogens can be used in hydroprocessing catalyst as an acidic function, especially fluoride as in the form of a fluorided alumina, halogens are not preferred because they will slowly be stripped from the catalyst and can lead to corrosion ofthe reactor vessel.
- the hydrogenation function in an aromatics-forming hydroprocessing catalyst comprises a metal.
- Suitable hydrogenation metals include Group VI metals such as Mo and or W and Group VIII metals such as Ni or Co. These are present on the catalyst in sulfided form.
- the hydrogenation metal is a noble metal, more preferably selected from the group consisting of Pt, Pd and mixtures thereof. These may be sulfided, but using them in the non-sulfided form is preferred. Catalysts useful in hydroprocessing operations are well known in the art.
- Suitable catalysts include noble metals from Group VIIIA (according to the 1975 rules ofthe International Union of Pure and Applied Chemistry), such as platinum or palladium on an alumina or siliceous matrix, and Group VIIIA and Group VDB, such as nickel- molybdenum, cobalt-molybdenum, nickel-tungsten, or nickel-tin on an alumina or siliceous matrix.
- the non-noble metal (such as nickel-molybdenum) hydrogenation metals are usually present in the final catalyst composition as oxides, or more preferably or possibly, as sulfides when such compounds are readily formed from the particular metal involved.
- Preferred non-noble metal catalyst compositions contain in excess of about 5 weight percent, preferably about 5 to about 40 weight percent molybdenum and/or tungsten, and at least about 0.5, and generally about 1 to about 15 weight percent of nickel and or cobalt determined as the corresponding oxides.
- the noble metal (such as platinum) catalyst may contain in excess of 0.01 percent metal, preferably between 0J and 1.0 percent metal. Combinations of noble metals may also be used, such as mixtures of platinum and palladium.
- the matrix components include some that have acidic catalytic activity. Ones that have activity include amorphous silica-alumina or may be a zeolitic or non-zeolitic crystalline molecular sieve.
- Suitable matrix molecular sieves include zeolite Y, zeolite X and the so called ultra stable zeolite Y and high structural silica: alumina ratio zeolite Y.
- Suitable matrix materials may also include synthetic or natural substances as well as inorganic materials such as clay, silica and/or metal oxides such as silica-alumina, silica-magnesia, silica-zirconia, silica-thoria, silica-beryllia, silica-titania as well as ternary compositions, such as silica-alumina-thoria, silica-alumina-zirconia, silica-alumina- magnesia, and silica-magnesia zirconia.
- the latter may be either naturally occurring or in the form of gelatinous precipitates or gels including mixtures of silica and metal oxides.
- Naturally occurring clays which can be composited with the catalyst, include those ofthe montmorillonite and kaolin families. These clays can be used in the raw state as originally mined or initially subjected to calumniation, acid treatment or chemical modification. More than one catalyst type may be used in the reactor.
- hydroprocessing in general, is well know to those of skill in the art and includes such processes as hydrotreating, hydrocracking, hydrogenation, catalytic dewaxing, or combinations of these processes.
- the hydroprocessing ofthe present invention upgrades the Fischer Tropsch derived feedstock by performing an operation selected from the group consisting of reducing the content of sulfur, nitrogen, and oxygen in the feedstock; reducing the content of olefins in the feedstock; increasing the ratio of iso/normal paraffins in the product to between 0J and 10; increasing the production of distillate fuel product by converting heavy species in the feedstock; and combinations thereof.
- Typical hydrotreating conditions vary over a wide range. Typical pressures for this operation are less than 1000 psig, preferably less than 600 psig, and most preferably between 200 and 500 psig.
- Typical liquid hourly space velocities (LHSV) for this operation are greater than 0.25 hr "1 , preferably between 0.5 and 2.0 hr "1 .
- Hydrogen recirculation rates are typically greater than 50 standard cubic feet per barrel of oil (SCF/Bbl), and are preferably between 1000 and 5000 SCF/Bbl. Temperatures range from about 300°F to about 750°F, preferably ranging from 450°F to 600°F.
- Hydrocracking may be conducted according to conventional methods known to those of skill in the art. Typically, hydrocracking is a process of breaking larger carbon molecules into smaller ones.
- It may be effected by contacting the particular fraction or combination of fractions, with hydrogen in the presence of a suitable hydrocracking catalyst at temperatures in the range of from about 600 to 900°F (316 to 482°C), preferably 650 to 850°F (343 to 454°C), and pressures in the range of from about 200 to 4000 psia (13 to 272 atm), preferably 500 to 3000 psia (34 to 204 atm) using space velocities based on the hydrocarbon feedstock of about 0J to 10 hr "1 , preferably 0J5 to 5 hr "1 .
- a suitable hydrocracking catalyst at temperatures in the range of from about 600 to 900°F (316 to 482°C), preferably 650 to 850°F (343 to 454°C), and pressures in the range of from about 200 to 4000 psia (13 to 272 atm), preferably 500 to 3000 psia (34 to 204 atm) using space
- hydrocracking is utilized to reduce the size ofthe hydrocarbon molecules, hydro genate olefin bonds, hydrogenate aromatics and remove traces of heteroatoms.
- Suitable catalysts for hydrocracking operations are known in the art and include sulfided catalysts.
- Sulfided catalyst may comprise amorphous silica-alumina, alumina, tungsten and nickel.
- the conditions of hydrogenation are well known in the industry and include temperatures above ambient and pressures greater than atmospheric.
- Preferable conditions for hydrogenation include a temperature between 300 and 800°F, most preferably between 400 and 600°F, a pressure between 50 and 2000 psig, most preferably between 100 and 500 psig, a liquid hourly space velocity (LHSV) between 0J and 10 hr "1 , most preferably between 1.0 and 3.0 hr "1 , and a gas rate between 500 and 10,000 SCFB, most preferably between 1000 and 5000 SCFB.
- LHSV liquid hourly space velocity
- the catalysts used for hydrogenation are those typically used in hydrotreating, but non-sulfided catalysts containing Pt and/or Pd are preferred, and it is preferred to disperse the Pt and/or Pd on a support, such as alumina, silica, silica-alumina, or carbon.
- a support such as alumina, silica, silica-alumina, or carbon.
- the preferred support is silica-alumina.
- Catalytic dewaxing consists of two main classes — conventional hydrodewaxing and hydroisomerization dewaxing; hydroisomerization dewaxing can be further subdivided into partial and complete hydroisomerization dewaxing.
- All classes involve passing a mixture of a waxy hydrocarbon stream and hydrogen over a catalyst that contains an acidic component to convert the normal and slightly branched iso-paraffms in the feed to other non-waxy species and thereby generate a lube base stock product with an acceptable pour point (below about +10°F or -12°C).
- Typical conditions for all classes involve temperatures from about 400 to 800°F, pressures from about 200 to 3000 psig, and space velocities from about 0J to 5 hr "1 .
- the method selected for dewaxing a feed typically depends on the product quality, and the wax content ofthe feed, with Conventional Hydrodewaxing generally preferred for low wax content feeds.
- the method for dewaxing can be effected by the choice ofthe catalyst.
- the general subject is reviewed by Avilino Sequeira, in Lubricant Base Stock and Wax Processing, Marcel Dekker, Inc pages 194-223.
- the determination ofthe class of dewaxing catalyst among conventional hydrodewaxing, partial hydroisomerization dewaxing and complete hydroisomerization dewaxing can be made by using the n-hexadecane isomerization test as describe by Santilli et al. in U.S. Patent No. 5,282,958.
- hydrodewaxing catalysts When measured at 96% n-hexadecane conversion under conditions described by Santilli et al., conventional hydrodewaxing catalysts will exhibit a selectivity to isomerized hexadecanes of less than 10%, hydroisomerization dewaxing catalysts will exhibit a selectivity to isomerized hexadecanes of greater than or equal to 10%, partial hydroisomerization dewaxing catalysts will exhibit a selectivity to isomerized hexadecanes of greater than 10% to less than 40%, and complete hydroisomerization dewaxing catalysts will exhibit a selectivity to isomerized hexadecanes of greater than or equal to 40%, preferably greater than 60%, and most preferably greater than 80%.
- Conventional hydrodewaxing is defined for purposes of this document as a catalytic dewaxing process that uses a conventional hydrodewaxmg catalyst.
- the pour point is lowered by selectively cracking the wax molecules, mostly to smaller paraffins boiling between propane and about octane. Since this technique converts the wax to less valuable by-products, it is useful primarily for dewaxing oils that do not contain a large amount of wax. Waxy oils of this type are frequently found in petroleum distillate from moderately waxy crudes (Arabian, North Slope, etc). Catalysts that are useful for conventional hydrodewaxing are typically 12-ring zeolites and 10-ring zeolites.
- Zeolites of this class include ZSM-5, ZSM-11, ZSM-22, ZSM-23, ZSM-35, and Mordenite.
- Conventional hydrodewaxing catalysts favor cracking in comparison to other method of conversion of paraffins. This is demonstrated by use of the n-hexadecane isomerization test by Santilli et al., in which conventional hydrodewaxing catalysts exhibit a selectivity to isomerized hexadecane products of less than 10%).
- metals may be added to the catalyst, primarily to reduce fouling. Representative process conditions, yields, and product properties for conventional hydrodewaxing are described, for example, in U.S. Patent Nos.
- Hydroisomerization dewaxing is defined for purposes of this document as a catalytic dewaxing process that uses a hydroisomerization dewaxing catalyst. Hydroisomerization dewaxing converts at least a portion ofthe wax to non-waxy iso- paraffins by isomerization, while at the same time minimizing the conversion by cracking.
- hydroisomerization dewaxing uses a dual-functional catalyst consisting of an acidic component and a metal component. Both components are required to conduct the isomerization reaction. Typical metal components are platinum or palladium, with platinum most commonly used.
- the choice and the amount of metal in the catalyst is sufficient to achieve greater than 10% isomerized hexadecane products in the test described by Santilli et al.
- the catalyst is a complete hydroisomerization dewaxing catalyst. Since hydroisomerization dewaxing converts wax to iso-paraffins which boil in the lube base stock range, it is useful for dewaxing oils that contain a large amount of wax.
- Waxy oils of this type are obtained from slack waxes from solvent dewaxing processes, ' and distillates from highly waxy crudes (Minas, Altamont, etc.) and products from the Fischer-Tropsch process.
- Partial hydroisomerization dewaxing is defined for purposes of this document as a catalytic dewaxing process that uses a partial hydroisomerization dewaxing catalyst. In partial hydroisomerization dewaxing a portion ofthe wax is isomerized to iso-paraffins using catalysts that can isomerize paraffins selectively, but only if the conversion of wax is kept to relatively low values (typically below 70%).
- the acidic catalyst components useful for partial hydroisomerization dewaxing include amorphous silica aluminas, fluorided alumina, and 12-ring zeolites (such as Beta, Y zeolite, L zeolite). Because the wax conversion is incomplete, partial hydroisomerization dewaxing must be supplemented with an additional dewaxing technique, typically solvent dewaxing. The wax recovered from a solvent dewaxing operation following a partial hydroisomerization dewaxing can be recycled to the partial hydroisomerization dewaxing step.
- the acidic catalyst components useful for partial hydroisomerization dewaxing include 10-ring, 1 -dimensional, molecular sieves (such as ZSM-23, SSZ-32, Theta-1, ZSM-22, SAPO-11, and SAPO-41). Since wax conversion can be complete, or at least very high, this process typically does not need to be combined with additional dewaxing processes to produce a lube base stock with an acceptable pour point. Representative process conditions, yields, and product properties for complete hydroisomerization dewaxing are described, for example, in U.S. Patent Nos.
- the distillate fuel according to the present invention may be comprised of a combination of blend stocks, or the distillate fuel may be comprised of Fischer Tropsch distillate fuel blend stock in the absence of other blend stocks with only the optional addition of minor amounts of additives. Accordingly, the distillate fuels may comprise a Fischer-Tropsch distillate fuel blend stock mixed with petroleum blend stock.
- the distillate fuel comprises 1 to 95 weight % Fischer Tropsch blend stock and 5 to 99 weight % petroleum blend stock. More preferably, the distillate fuel comprises 5 to 75 weight % Fischer Tropsch blend stock and 25 to 95 weight % petroleum blend stock.
- the distillate fuel comprises 10 to 50 weight % Fischer Tropsch blend stock and 90 to 50 weight % petroleum blend stock.
- the Fischer Tropsch feedstock may be blended with a petroleum blend stock at any stage in the process so long as a highly paraffmic, moderately unsaturated distillate fuel blend stock according to the present invention is provided.
- a petroleum blend stock may be blended with a Fischer Tropsch derived feedstock prior to hydroprocessing, after hydroprocessing but prior to removing polynuclear aromatics, or after removal of polynuclear aromatics but prior to use as a distillate fuel
- the petroleum blend stock is blended with the Fischer Tropsch feedstock prior to hydroprocessing and the resulting blended stream is hydroprocessed.
- the Fischer Tropsch feedstock is mixed with a petroleum blend stock
- the resulting blend comprises 1 to 95 weight % Fischer Tropsch feedstock and 99 to 5 weight % petroleum blend stock. More preferably, the blend comprises 5 to 75 weight % Fischer Tropsch feedstock and 95 to 25 weight % petroleum blend stock. Even more preferably, the blend comprises 10 to 50 weight % Fischer Tropsch feedstock and 50 to 90 weight % petroleum blend stock.
- the product stream from the hydroprocessing operation can be further treated to remove polynuclear aromatics.
- Options for selectively removing polynuclear aromatics from the product stream while leaving desired mono- aromatics include selective hydrotreating and adsorption.
- the most preferred operation for removing polynuclear aromatics from the product stream is selective hydrotreating.
- the reaction conditions for selective hydrotreating do not vary greatly from the reaction conditions for hydrotreating described above.
- Reaction conditions for selective hydrotreating include low temperatures (less than 750°F, preferably less than 700°F, most preferably less than 600°F), high pressures (greater than 250 psig, preferably greater than 350 psig, most preferably greater than 500 psig), and short contact times (LHSV of less than 5 hr "1 , preferably less than 3 hr "1 , and most preferably less than 2 hr "1 ).
- Prefe ⁇ ed catalysts for this selective hydrotreating contain Pt, Pd, and combinations thereof.
- the selective hydrotreating will reduce the polynuclear aromatic content by at least 50 weight %, preferably at least 75 weight %, and most preferably at least 90 weight %, and the unsaturate content by less than 50 weight %, preferably less than 35 weight %, and most preferably less than 20 weight %.
- the removal of polynuclear aromatics from the product stream can also be achieved by adsorption on an oxide support, preferably one that has moderate acidity (an acidic clay such as montmorillonite or attapulgite).
- the temperatures for adsorption should be less than 200°F, preferably less than 150°F.
- Polynuclear aromatics can also be extracted with a solvent, such as n-methyl pyrollidinone, phenol, or furfural.
- the distillate fuel and distillate fuel blend stock may include additives that are commonly used for diesel or jet fuels.
- a description of diesel fuel additives that may be used in the present invention is as described in the Chevron Corporation, Technical Review Diesel Fuels, pp. 55-64 (2000) and a description of jet fuel additives that may be used in the present invention is as described in Chevron Corporation, Technical Review Aviation Fuels, pp 27-30 (2000).
- these additives may include, but are not limited to, antioxidants, lubricity additives, pour point depressants, and the like.
- the additives are added to the fuels and blend stocks in a minor amount, preferably less than 1 weight %.
- FIG. 1 represents a process for preparing a highly paraffmic, moderately unsaturated distillate fuel blend stock according to the present invention.
- a low temperature Fischer-Tropsch derived feedstock (10) is hydroprocessed in a hydroprocessing unit (100) to which hydrogen (20) is added.
- Hydroprocessing conditions include a temperature of 600-750°F, a pressure of less than 1000 psig, and a liquid hourly space velocity of greater than 0J5 hr "1 .
- the product (30) of the hydroprocessing is a highly paraffinic, moderately unsaturated distillate fuel blend stock containing between 2 and 20 weight % unsaturates and peroxide precursors in an amount such that less than 5 ppm peroxides are formed after storage at 60°C for four weeks.
- the hydroprocessing may produce hydrogen (60), which can be used in other processes such as hydrotreatment, CO 2 reduction, and fuel production.
- the highly paraffmic, moderately unsaturated distillate fuel blend stock (30) may be treated to remove polynuclear aromatics (70) in a processing unit (200).
- Example 1 A light Low Temperature Fischer-Tropsch wax (Table I) was hydrocracked over a sulfided nickel-tungsten/silica-alumina catalyst (a conventional hydroprocessing catalyst), LHSV 1 hr "1 , 1000 psig, 685°F, and 6.3 MSCF/bbl. At these conditions, conversion below 650°F was 80.4 weight %. The liquid product was cut at about 350°F and about 675°F to give a diesel blend stock fraction. Yields and properties ofthe diesel blend stock are given in Table II.
- the lower limit of detection of PNA by Supercritical Fluid Chromatography (SFC) is 0.5 weight %. Thus non-detected values are less than this amount. Operation at these conditions produced only 0J percent aromatics and retained 0.8 wt% olefins due to the high pressure conventional operation.
- the paraffin content of this sample is equivalent to the saturate content (98.9).
- the ndM analysis which is suitable for non-olefin containing samples, indicates the absence of naphthenic carbon structures.
- Example 2 The same feed as in Example 1 was hydrocracked over a sulfided 3/1 layered bed ofthe same catalyst as in Example 1 over a Pt/SAPO-11 catalyst, the latter bound with 15 weight % alumina.
- the Pt/SAPO-11 catalyst is a complete hydroisomerization dewaxing catalyst.
- Conditions were the same as in Example 3, that is overall LHSV 1.0 hr "1 , 1000 psig, 685°F, and 6.3 MSCF/bbl H 2 . Conversion below 650°F was 74.6 weight %.
- the product was cut at about 350°F and about 650°F to give a diesel blend stock cut. Yields and diesel blend stock properties are given in Table III.
- Example 3 Example 2 was repeated, but at a total pressure in the reactor of 500 psig, and a reactor temperature of 680°F. Conversion below 650°F was 63.5 weight %. The product was cut at about 350°F and about 590°F to give a diesel blend stock cut. Yields and diesel blend stock properties are given in Table TV. As determined by ASTM D6468, the diesel blend stock was very stable. Aromatics in the diesel blend stock were 2J weight %. The Cetane Index was still quite high (69J) and the cloud point very low (-50°C).
- Example 4 A 700-1000°F hydrotreated Low Temperature Fischer Tropsch wax (Table V) was hydrocracked over the same layered bed catalyst system of Example 2. Conditions included an overall LHSV 1.0 hr "1 , reactor pressure of 300 psig, 680°F for the top catalyst and 690°F for the bottom catalyst, and 6.3 MSCF/bbl H 2 . Conversion below 650°F was 58.2 weight %. The product was cut at about 300°F and about 650°F to give a diesel blend stock cut. Yields and diesel blend stock properties of this product are given in Table VI. As determined by ASTM D6468, the diesel blend stock was very stable.
- Aromatics in the diesel blend stock were 4.3 weight, olefins were 1.0 wt%, and oxygenates were 0.5 wt %. Paraffins are equal to the saturates (94J %) because the GC-MS technique did not detect significant quantities ofthe sum of olefins and naphthenes.
- the Cetane Index was high (67.6) and the cloud point was -44°C.
- the diesel blend stock of example 4 exhibited excellent stability as measured by ASTM D6468 at 150°C for 180 minutes, as did the diesel blend stocks of examples 2 and 3, with results in excess of 99%.
- the polynuclear aromatic content ofthe diesel blend stock of example 4 was 0.5 wt% - less than 10 weight % ofthe total unsaturates (5.3%).
- Example 5 A Low Temperature Fischer Tropsch wax and a Low Temperature Fischer Tropsch light condensate (Table VII) were processed over conventional hydroprocessing catalysts. 168 cm 3 /hr ofthe wax and a recycle liquid were hydrocracked over a 126 cm 3 of sulfided NiW/ acidic amorphous SiO 2 -Al O 3 catalyst.
- Example 6 228 cm /hr of a hydrotreated Low Temperature Fischer Tropsch wax (Table VII) was processed over 300 cm 3 sulfided NiW/ acidic amorphous SiO2-A12O3 catalyst in reactor 1 (Rx 1) and the combined effluent was processed over cm 3 of a Pt/SAPO-11 catalyst in reactor 2 (Rx 2).
- the Pt/SAPO-11 was bound with 15 weight % alumina.
- the Pt/SAPO-11 catalyst is a complete hydroisomerization dewaxing catalyst.
- the product was distilled to obtain a diesel blend stock fraction. Yields, operating conditions and product properties are shown in Table VIII. Table VIII
- Products made at 300 psig contained greater than 2 wt% unsaturates, more than 1 wt% olefins, had less than 1 ppm sulfur yet had excellent stabilities in diesel and jet fuel tests, and excellent resistance to peroxide formation. All products had greater than 90% paraffins. Products from the Pt/SAPO-11 catalyst showed a lower level of olefins presumably due to the presence ofthe more active Pt component.
- Comparative Example 7 Preparation of a Fully Hydrogenated Diesel Blend Stock
- a highly paraffmic diesel blend stock was prepared from three individual Low Temperature Fischer-Tropsch feedstocks.
- the blend stock was prepared continuously by feeding the different feedstocks down-flow to a hydroprocessing reactor.
- the reactor was filled with a catalyst containing alumina, silica, nickel, and tungsten.
- the catalyst was sulfided prior to use.
- the per-pass conversion was maintained at approximately 80% below the recycle cut point of 665- 710°F by adjusting the catalyst temperature.
- the product from the hydroprocessing reactor after separation and recycling of unreacted hydrogen was continuously distilled to provide a gaseous by-product, a light naphtha, a diesel blend stock fraction, and an unconverted fraction.
- the unconverted fraction was recycled to the hydroprocessing reactor.
- the temperatures ofthe distillation column were adjusted to maintain the flash and cloud points at their target values of 58°C and -18°C, respectively.
- the feedstocks were blended from several hours of consistent operation at 1.4 LHSV to provide the representative Product A in the Table X.
- Oxygen can be present in the sample in the form of organic oxygenates, measured by gas chromatography-mass spectrometry (GC-MS), dissolved or suspended water, measured by Karl Fischer, or dissolved O 2 from the air.
- the oxygenate content was determined by GC-MS.
- Oxygenates in the sample were treated with tetraethoxysilane (TEOS) to increase the sensitivity ofthe technique. Oxygenates could not be detected Sample A.
- the limit of detection ofthe technique was determined to be 6.5 ppm per oxygenate. With the molecular weight range of diesel fuel this is equivalent to 0.6 ppm oxygen as oxygenates.
- Feedstock 1 ofthe Fischer-Tropsch product in Table XI was by- ' passed around the hydroprocessing unit and fed directly to the distillation column.
- the yield of diesel fuel blend stock was less, near 73% due to requirement to reduce the end point ofthe diesel fuel blend stock to maintain cloud point.
- Diesel fuel blend stock was blended from several hours of consistent operation to provide the representative Product B in the Table X.
- Example 9 Stability Measurements Product B was tested according to ASTM D6468 at 150°C for 180 minutes and found to have a stability of 99.3%, which indicates that it is extremely stable towards deposit formation in this test. .
- the Products were then tested for peroxide formation under accelerated formation according to methods as described in U.S. Patent Nos. 6,162,956 and 6,180,842.
- the Products were tested according to a standard procedure for measuring the buildup of peroxides. First, a 4 oz. sample was placed in a brown bottle and aerated for 3 minutes. An aliquot ofthe sample was then tested according to ASTM D3703 for peroxides.
- Example 10 Effect of Trace Olefins on Peroxide Stability
- a further study was done to determine the effects of adding small amounts of olefinic condensate to the stable blend stock Product A of Table X.
- the properties ofthe 300-600°F portion ofthe cold condensate are as follows:
- Example 11 The 300-600°F portion ofthe cold condensate was blended in varying amounts with the Stable Fuel Blend Stock A of Table X and the blends were evaluated for peroxide formation with the following results: Table XVI - Peroxide Formation of Blends 1-5
- Blend stocks can tolerate only up to 0J weight % cold condensate (0.012 wt% oxygenates as alcohols determined by GC-MS and about 0J wt% olefins) and still be considered stable.
- Blend stocks with more than 0.012 wt% oxygenates or 0J wt% olefins did not exhibit satisfactory stability. As the oxygenate content was increased beyond 0.012 wt%, the peroxide stability ofthe blend stock rapidly declined.
Landscapes
- Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Liquid Carbonaceous Fuels (AREA)
- Production Of Liquid Hydrocarbon Mixture For Refining Petroleum (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BRPI0411601-1A BRPI0411601A (en) | 2003-06-19 | 2004-06-16 | distilled fuel, fuel mixture stock, process for preparing a fuel mixture stock and method of operating an engine |
| AU2004252510A AU2004252510B2 (en) | 2003-06-19 | 2004-06-16 | Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of Fischer-Tropsch products |
| GB0600807A GB2421512B (en) | 2003-06-19 | 2004-06-16 | Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of Fischer-Tropch products |
| JP2006517325A JP2007520579A (en) | 2003-06-19 | 2004-06-16 | Stable and moderately unsaturated distillate fuel blend feedstock prepared by low pressure hydroprocessing of Fischer-Tropsch products |
| AU2010200184A AU2010200184A1 (en) | 2003-06-19 | 2010-01-18 | Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of Fischer-Tropsch products |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/464,635 | 2003-06-19 | ||
| US10/464,635 US20070187292A1 (en) | 2001-10-19 | 2003-06-19 | Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of Fischer-Tropsch products |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005001002A2 true WO2005001002A2 (en) | 2005-01-06 |
| WO2005001002A3 WO2005001002A3 (en) | 2005-12-15 |
Family
ID=33551389
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2004/019252 WO2005001002A2 (en) | 2003-06-19 | 2004-06-16 | Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of fischer-tropsch products |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20070187292A1 (en) |
| JP (1) | JP2007520579A (en) |
| CN (1) | CN100465251C (en) |
| AU (2) | AU2004252510B2 (en) |
| BR (1) | BRPI0411601A (en) |
| GB (1) | GB2421512B (en) |
| NL (1) | NL1026460C2 (en) |
| WO (1) | WO2005001002A2 (en) |
| ZA (1) | ZA200600302B (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007153937A (en) * | 2005-11-30 | 2007-06-21 | Nippon Oil Corp | Gas oil composition |
| EP2022839A1 (en) * | 2006-05-17 | 2009-02-11 | Nippon Mitsubishi Oil Corporation | Gas-oil composition |
| DE102008005346A1 (en) | 2007-08-10 | 2009-02-12 | Indian Oil Corp. Ltd., Mumbai | New synthetic fuel and method of making same |
| EP2163598A1 (en) * | 2007-06-11 | 2010-03-17 | Nippon Oil Corporation | Light oil composition |
| WO2010072838A3 (en) * | 2008-12-26 | 2010-08-26 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| WO2010072839A3 (en) * | 2008-12-26 | 2010-09-16 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| WO2011017720A1 (en) | 2009-08-03 | 2011-02-10 | Sasol Technology (Pty) Ltd | Fully synthetic jet fuel |
| US8142527B2 (en) | 2005-03-21 | 2012-03-27 | Ben-Gurion University Of The Negev Research And Development Authority | Production of diesel fuel from vegetable and animal oils |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE538193T1 (en) * | 2003-09-03 | 2012-01-15 | Shell Int Research | FUEL COMPOSITIONS |
| BRPI0414040A (en) * | 2003-09-03 | 2006-10-24 | Shell Int Research | use of a fuel derived from fischer-tropsch, and, fuel composition |
| US20060278565A1 (en) * | 2005-06-10 | 2006-12-14 | Chevron U.S.A. Inc. | Low foaming distillate fuel blend |
| JP5426375B2 (en) * | 2006-07-27 | 2014-02-26 | シエル・インターナシヨネイル・リサーチ・マーチヤツピイ・ベー・ウイ | Fuel composition |
| AU2007333907B2 (en) * | 2006-12-14 | 2013-01-17 | Chevron U.S.A. Inc. | Improved process for making Fischer-Tropsch olefinic naphtha and hydrogenated distillates |
| JP5466151B2 (en) * | 2007-05-31 | 2014-04-09 | セイソル テクノロジー (プロプライエタリー) リミテッド | Low temperature flow response of diesel fuel by distillate replacement |
| NL2002173C2 (en) * | 2007-11-06 | 2010-12-15 | Sasol Tech Pty Ltd | Synthetic aviation fuel. |
| US8894818B2 (en) * | 2008-02-28 | 2014-11-25 | Chevron U.S.A. Inc. | Process for generating a hydrocarbon feedstock lignin |
| US8795472B2 (en) * | 2008-02-28 | 2014-08-05 | Chevron U.S.A. Inc. | Process for generating a hydrocarbon feedstock from lignin |
| WO2010048251A2 (en) * | 2008-10-22 | 2010-04-29 | Chevron U.S.A. Inc. | A high energy distillate fuel composition and method of making the same |
| US20100122519A1 (en) * | 2008-11-14 | 2010-05-20 | Alan Epstein | Ultra-low sulfur fuel and method for reduced contrail formation |
| US9492818B2 (en) | 2009-06-12 | 2016-11-15 | Albemarle Europe Sprl | SAPO molecular sieve catalysts and their preparation and uses |
| US9080118B2 (en) | 2010-05-06 | 2015-07-14 | Sasol Technology (Pty) Ltd | Diesel engine injector fouling improvements with a highly paraffinic distillate fuel |
| JP5730102B2 (en) * | 2011-03-31 | 2015-06-03 | 独立行政法人石油天然ガス・金属鉱物資源機構 | Start-up method of bubble column type slurry bed reactor, start-up solvent and method for producing hydrocarbon oil |
| US9005380B2 (en) * | 2012-03-23 | 2015-04-14 | Johann Haltermann Limited | High performance liquid rocket propellant |
| US9453169B2 (en) | 2013-09-13 | 2016-09-27 | Uop Llc | Process for converting fischer-tropsch liquids and waxes into lubricant base stock and/or transportation fuels |
| ES2803557T3 (en) * | 2015-12-21 | 2021-01-27 | Neste Corp | Method of producing aviation fuel composition |
| EP3529336A4 (en) * | 2016-10-18 | 2020-04-15 | Mawetal LLC | Fuel compositions from light tight oils and high sulfur fuel oils |
| FI127307B (en) | 2017-01-27 | 2018-03-15 | Neste Oyj | Refined fuel compositions and methods for their preparation |
| CN110582554B (en) * | 2017-05-01 | 2023-08-25 | 埃克森美孚科技工程公司 | Jet Fuel Treatment for Blend Compatibility |
| CN110616093A (en) * | 2019-08-28 | 2019-12-27 | 刘明波 | High-cleanness diesel fuel |
| US11377607B2 (en) | 2020-03-23 | 2022-07-05 | Exxonmobil Chemical Patents Inc. | Branched paraffinic compositions derived from isomerized and hydrogenated linear alpha olefins |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001002325A1 (en) * | 1999-07-06 | 2001-01-11 | Sasol Technology (Pty) Ltd. | Alkylbenzenes derived from fischer-tropsch hydrocarbons and their use in drilling fluids |
| US6392109B1 (en) * | 2000-02-29 | 2002-05-21 | Chevron U.S.A. Inc. | Synthesis of alkybenzenes and synlubes from Fischer-Tropsch products |
Family Cites Families (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3149933A (en) * | 1960-08-25 | 1964-09-22 | Bayer Ag | Process for stabilizing liquid fuels |
| US3974081A (en) * | 1974-07-31 | 1976-08-10 | Exxon Research And Engineering Company | Biodegradable seal swell additive with low toxicity properties for automatic transmission fluids, power transmission fluids and rotary engine oil applications |
| US4320021A (en) * | 1975-10-14 | 1982-03-16 | The Lubrizol Corporation | Amino phenols useful as additives for fuels and lubricants |
| US4320020A (en) * | 1976-04-12 | 1982-03-16 | The Lubrizol Corporation | Alkyl amino phenols and fuels and lubricants containing same |
| US4347148A (en) * | 1976-07-15 | 1982-08-31 | The Lubrizol Corporation | Full and lubricant compositions containing nitro phenols |
| US4181598A (en) * | 1977-07-20 | 1980-01-01 | Mobil Oil Corporation | Manufacture of lube base stock oil |
| US4173050A (en) * | 1978-08-10 | 1979-11-06 | International Shoe Machine Corporation | Pincers assembly and its mounting |
| US4176050A (en) * | 1978-12-04 | 1979-11-27 | Mobil Oil Corporation | Production of high V.I. lubricating oil stock |
| US4222855A (en) * | 1979-03-26 | 1980-09-16 | Mobil Oil Corporation | Production of high viscosity index lubricating oil stock |
| US4211635A (en) * | 1979-04-23 | 1980-07-08 | Mobil Oil Corporation | Catalytic conversion of hydrocarbons |
| US4229282A (en) * | 1979-04-27 | 1980-10-21 | Mobil Oil Corporation | Catalytic dewaxing of hydrocarbon oils |
| US4386939A (en) * | 1979-12-10 | 1983-06-07 | The Lubrizol Corporation | Reaction products of certain heterocycles with aminophenols |
| US4456527A (en) * | 1982-10-20 | 1984-06-26 | Chevron Research Company | Hydrocarbon conversion process |
| US4704493A (en) * | 1983-10-31 | 1987-11-03 | Chevron Corporation | Conversions of low molecular weight hydrocarbons to higher molecular weight hydrocarbons using a metal compound-containing catalyst (II-A) |
| US4704487A (en) * | 1983-10-31 | 1987-11-03 | Chevron Research Company | Conversions of low molecular weight hydrocarbons to higher molecular weight hydrocarbons using a metal compound-containing catalyst (IV-B) |
| US4599474A (en) * | 1983-10-31 | 1986-07-08 | Chevron Research Company | Conversions of low molecular weight hydrocarbons to higher molecular weight hydrocarbons using a metal-containing catalyst |
| US4734537A (en) * | 1983-10-31 | 1988-03-29 | Chevron Research Company | Conversions of low molecular weight hydrocarbons to higher molecular weight hydrocarbons using a metal compound-containing catalyst |
| US4507517A (en) * | 1983-10-31 | 1985-03-26 | Chevron Research Company | Conversions of low molecular weight hydrocarbons to higher molecular weight hydrocarbons using a boron compound containing catalyst |
| US4709108A (en) * | 1983-10-31 | 1987-11-24 | Chevron Research Company | Conversions of low molecular weight hydrocarbons to higher molecular weight hydrocarbons using a metal compound-containing catalyst |
| US4568663A (en) * | 1984-06-29 | 1986-02-04 | Exxon Research And Engineering Co. | Cobalt catalysts for the conversion of methanol to hydrocarbons and for Fischer-Tropsch synthesis |
| US4814538A (en) * | 1985-10-23 | 1989-03-21 | Chevron Research Company | Enhancing the reaction rate in high temperature, high space velocity catalytic conversion of methane to higher molecular weight hydrocarbons |
| US4814534A (en) * | 1985-10-31 | 1989-03-21 | Chevron Research Company | Addition of hydrogen and C2 to C4 hydrocarbons to the feed gas in the catalytic conversion of methane to higher molecular weight hydrocarbons |
| US4814533A (en) * | 1985-10-31 | 1989-03-21 | Chevron Research Company | Enhancing the production of aromatics in high temperature, high space velocity catalytic conversion of lower molecular weight hydrocarbons to higher molecular weight hydrocarbons |
| US5021148A (en) * | 1988-11-29 | 1991-06-04 | Carbon Fuels Corporation | Method of refining coal by short residence time partial liquefaction to produce petroleum substitutes and chemical feedstocks |
| US4783566A (en) * | 1987-08-28 | 1988-11-08 | Uop Inc. | Hydrocarbon conversion process |
| US4943672A (en) * | 1987-12-18 | 1990-07-24 | Exxon Research And Engineering Company | Process for the hydroisomerization of Fischer-Tropsch wax to produce lubricating oil (OP-3403) |
| IT1219692B (en) * | 1988-05-06 | 1990-05-24 | Eniricerche Spa | SILICA GEL AND CATALYTICALLY ACTIVE ALUMINUM AND PROCEDURE FOR ITS PREPARATION |
| JP2907543B2 (en) * | 1989-02-17 | 1999-06-21 | シェブロン リサーチ アンド テクノロジー カンパニー | Isomerization of waxy lubricating oils and petroleum waxes using silicoaluminophosphate molecular sheep catalysts |
| US5082986A (en) * | 1989-02-17 | 1992-01-21 | Chevron Research Company | Process for producing lube oil from olefins by isomerization over a silicoaluminophosphate catalyst |
| US5246566A (en) * | 1989-02-17 | 1993-09-21 | Chevron Research And Technology Company | Wax isomerization using catalyst of specific pore geometry |
| US4952306A (en) * | 1989-09-22 | 1990-08-28 | Exxon Research And Engineering Company | Slurry hydroprocessing process |
| US5282958A (en) * | 1990-07-20 | 1994-02-01 | Chevron Research And Technology Company | Use of modified 5-7 a pore molecular sieves for isomerization of hydrocarbons |
| US5944858A (en) * | 1990-09-20 | 1999-08-31 | Ethyl Petroleum Additives, Ltd. | Hydrocarbonaceous fuel compositions and additives therefor |
| US5215548A (en) * | 1991-12-20 | 1993-06-01 | Exxon Research And Engineering Company | Distillate fuels containing an amine salt of a sulfonic acid and a low volatility carrier fluid (PNE-554) |
| JP3833250B2 (en) * | 1994-11-22 | 2006-10-11 | エクソンモービル リサーチ アンド エンジニアリング カンパニー | Monolithic mixed powder pellet catalyst and method for reforming waxy feedstock using the same |
| US5689031A (en) * | 1995-10-17 | 1997-11-18 | Exxon Research & Engineering Company | Synthetic diesel fuel and process for its production |
| US6162956A (en) * | 1998-08-18 | 2000-12-19 | Exxon Research And Engineering Co | Stability Fischer-Tropsch diesel fuel and a process for its production |
| US6180842B1 (en) * | 1998-08-21 | 2001-01-30 | Exxon Research And Engineering Company | Stability fischer-tropsch diesel fuel and a process for its production |
| US6337010B1 (en) * | 1999-08-02 | 2002-01-08 | Chevron U.S.A. Inc. | Process scheme for producing lubricating base oil with low pressure dewaxing and high pressure hydrofinishing |
| US6703429B2 (en) * | 2001-08-23 | 2004-03-09 | Chevron U.S.A. Inc. | Process for converting synthesis gas into hydrocarbonaceous products |
| US6890423B2 (en) * | 2001-10-19 | 2005-05-10 | Chevron U.S.A. Inc. | Distillate fuel blends from Fischer Tropsch products with improved seal swell properties |
-
2003
- 2003-06-19 US US10/464,635 patent/US20070187292A1/en not_active Abandoned
-
2004
- 2004-06-16 JP JP2006517325A patent/JP2007520579A/en active Pending
- 2004-06-16 WO PCT/US2004/019252 patent/WO2005001002A2/en active Application Filing
- 2004-06-16 AU AU2004252510A patent/AU2004252510B2/en not_active Ceased
- 2004-06-16 BR BRPI0411601-1A patent/BRPI0411601A/en not_active IP Right Cessation
- 2004-06-16 CN CNB2004800238847A patent/CN100465251C/en not_active Expired - Lifetime
- 2004-06-16 ZA ZA200600302A patent/ZA200600302B/en unknown
- 2004-06-16 GB GB0600807A patent/GB2421512B/en not_active Expired - Fee Related
- 2004-06-18 NL NL1026460A patent/NL1026460C2/en active IP Right Maintenance
-
2010
- 2010-01-18 AU AU2010200184A patent/AU2010200184A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001002325A1 (en) * | 1999-07-06 | 2001-01-11 | Sasol Technology (Pty) Ltd. | Alkylbenzenes derived from fischer-tropsch hydrocarbons and their use in drilling fluids |
| US6392109B1 (en) * | 2000-02-29 | 2002-05-21 | Chevron U.S.A. Inc. | Synthesis of alkybenzenes and synlubes from Fischer-Tropsch products |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8142527B2 (en) | 2005-03-21 | 2012-03-27 | Ben-Gurion University Of The Negev Research And Development Authority | Production of diesel fuel from vegetable and animal oils |
| JP2007153937A (en) * | 2005-11-30 | 2007-06-21 | Nippon Oil Corp | Gas oil composition |
| EP2022839A1 (en) * | 2006-05-17 | 2009-02-11 | Nippon Mitsubishi Oil Corporation | Gas-oil composition |
| EP2022839A4 (en) * | 2006-05-17 | 2013-07-31 | Nippon Mitsubishi Oil Corp | Gas-oil composition |
| KR101525269B1 (en) * | 2007-06-11 | 2015-06-02 | 제이엑스 닛코닛세키에너지주식회사 | Light oil composition |
| EP2163598A1 (en) * | 2007-06-11 | 2010-03-17 | Nippon Oil Corporation | Light oil composition |
| EP2163598A4 (en) * | 2007-06-11 | 2015-01-21 | Nippon Oil Corp | Light oil composition |
| US8353972B2 (en) | 2007-08-10 | 2013-01-15 | Indian Oil Corporation Limited | Synthetic fuel and method of preparation thereof |
| DE102008005346A1 (en) | 2007-08-10 | 2009-02-12 | Indian Oil Corp. Ltd., Mumbai | New synthetic fuel and method of making same |
| US20120046506A1 (en) * | 2008-12-26 | 2012-02-23 | Nobuhiro Okabe | Diesel fuel composition |
| WO2010072839A3 (en) * | 2008-12-26 | 2010-09-16 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| WO2010072838A3 (en) * | 2008-12-26 | 2010-08-26 | Shell Internationale Research Maatschappij B.V. | Diesel fuel composition |
| WO2011017720A1 (en) | 2009-08-03 | 2011-02-10 | Sasol Technology (Pty) Ltd | Fully synthetic jet fuel |
| US8801919B2 (en) | 2009-08-03 | 2014-08-12 | Sasol Technology (Pty) Ltd | Fully synthetic jet fuel |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1839196A (en) | 2006-09-27 |
| AU2004252510B2 (en) | 2010-03-11 |
| US20070187292A1 (en) | 2007-08-16 |
| NL1026460A1 (en) | 2004-12-21 |
| WO2005001002A3 (en) | 2005-12-15 |
| ZA200600302B (en) | 2007-05-30 |
| NL1026460C2 (en) | 2005-10-11 |
| GB2421512B (en) | 2008-10-29 |
| AU2004252510A1 (en) | 2005-01-06 |
| BRPI0411601A (en) | 2006-08-08 |
| JP2007520579A (en) | 2007-07-26 |
| GB2421512A (en) | 2006-06-28 |
| GB0600807D0 (en) | 2006-02-22 |
| AU2010200184A1 (en) | 2010-02-04 |
| CN100465251C (en) | 2009-03-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2004252510B2 (en) | Stable, moderately unsaturated distillate fuel blend stocks prepared by low pressure hydroprocessing of Fischer-Tropsch products | |
| JP4796279B2 (en) | Distillate fuel mixture with improved seal swellability from Fischer-Tropsch product | |
| ZA200600300B (en) | Highly paraffinic, moderately aromatic distillate fuel blend stocks prepared by low pressure hydroprocessing of Fischer-Tropsch products | |
| AU2004213790B2 (en) | Process for producing premium fischer-tropsch diesel and lube base oils | |
| EP1284281B1 (en) | Synthetic Naphtha Fuel | |
| EP1835011A1 (en) | Biodegradable middle distillates and production thereof | |
| US20060016722A1 (en) | Synthetic hydrocarbon products | |
| US6635681B2 (en) | Method of fuel production from fischer-tropsch process | |
| JP2009515012A (en) | Fischer-Tropsch derived turbine fuel and method for producing the same | |
| US6515032B2 (en) | Co-hydroprocessing of fischer-tropsch products and natural gas well condensate | |
| GB2449009A (en) | Distillate fuel made from Fischer-Tropsch products | |
| AU781716B2 (en) | Manufacture of lubricants from Fischer-Tropsch syncrude | |
| GB2402133A (en) | Process for producing distillate fuel blend containing alkylaromatics as seal-swell improvers | |
| GB2380739A (en) | Fischer-Tropsch plant which may be disassembled, moved and reassembled |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200480023884.7 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006517325 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004252510 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006/00302 Country of ref document: ZA Ref document number: 200600302 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 0600807.2 Country of ref document: GB Ref document number: 0600807 Country of ref document: GB |
|
| ENP | Entry into the national phase |
Ref document number: 2004252510 Country of ref document: AU Date of ref document: 20040616 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004252510 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: PI0411601 Country of ref document: BR |
|
| 122 | Ep: pct application non-entry in european phase |