WO2004064798A1 - Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions - Google Patents
Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions Download PDFInfo
- Publication number
- WO2004064798A1 WO2004064798A1 PCT/BE2004/000011 BE2004000011W WO2004064798A1 WO 2004064798 A1 WO2004064798 A1 WO 2004064798A1 BE 2004000011 W BE2004000011 W BE 2004000011W WO 2004064798 A1 WO2004064798 A1 WO 2004064798A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- vitamin
- composition
- compound
- cosmetic
- oxide
- Prior art date
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y5/00—Nanobiotechnology or nanomedicine, e.g. protein engineering or drug delivery
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/12—Ketones
- A61K31/122—Ketones having the oxygen directly attached to a ring, e.g. quinones, vitamin K1, anthralin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/336—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having three-membered rings, e.g. oxirane, fumagillin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/67—Vitamins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/16—Emollients or protectives, e.g. against radiation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82B—NANOSTRUCTURES FORMED BY MANIPULATION OF INDIVIDUAL ATOMS, MOLECULES, OR LIMITED COLLECTIONS OF ATOMS OR MOLECULES AS DISCRETE UNITS; MANUFACTURE OR TREATMENT THEREOF
- B82B1/00—Nanostructures formed by manipulation of individual atoms or molecules, or limited collections of atoms or molecules as discrete units
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/41—Particular ingredients further characterized by their size
- A61K2800/413—Nanosized, i.e. having sizes below 100 nm
Definitions
- the present invention is related to the use of a composition comprising vitamin Kl oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions and to the cosmetic composition comprising vitamin Kl oxide.
- Vitamin Kl (phylloquinone) is needed for proper bone formation and blood clotting, in both cases by helping the body transport calcium.
- Vitamin K (2-methyl 3- phytyl-1, 4-naphtoquinone) and its derivative have already been used in pharmaceutical or cosmetic compositions for their various anti-inflammatory or dermatological applications.
- the document US-5 510 391 describes a method for treating blood vessel disorders of the skin using vitamin K. Such disorders include actinic and iatrogenic purpura, lentigines, telangiectasias of the- face, spider angiomas and spider veins of the face.
- the document JP-05320039 describes a cosmetic composition comprising vitamin Kl oxide without specification of any use.
- the document O94/00135 describes the use of a pharmaceutical composition in the treatment of symptoms of chronic inflammatory disorders, said composition comprising at least two pharmaceutically active agents whose combination produces an anti-inflammatory and analgesic effect. Said document also describes that the safety and effectiveness of the product may be optimised by co-administration of vitamins and derivatives thereof.
- vitamins are vitamin Kl and vitamin Kl oxide .
- the document GB-744 376 describes a stable oily vitamin emulsion comprising an oily vitamin and lecithin dispersed in water.
- the vitamins could be vitamin A, D, E, Kl or vitamin Kl oxide.
- Said document also describes that vitamin Kl oxide emulsion is a colourless oil, somewhat more stable than vitamin Kl, but having the same physiological activity as vitamin Kl and resulting in a stable emulsion which is not affected by heating at a temperature of 120° C for two hours period.
- a first aim of the present invention is to propose a new composition which finds advantageous applications in the treatment of various mammal dermatological lesions, especially human dermatological lesions, and more especially lesions which affect the face, but which does not present the drawbacks of the state of the ar .
- Another aim of the present invention is to provide such composition which is more stable and which does not present the yellow colour of cream and vehicles already used in the state of the art and which therefore does not render the clothes of the consumer dirty.
- a further aim of the present invention is to provide a composition which is not sensitive to light or UV-radiation and which therefore decreases or eliminates side effects such as consumer skin sensitivity or allergy following sun exposure.
- a last aim of the present invention is to provide a composition having a similar or an improved activity (including an enhanced penetration through the skin and an excellent moisture-binding capacity) in view of the known composition of the state of the art.
- a first aspect of the present invention is related to the use of a composition comprising vitamin Kl oxide or a derivative thereof and an adequate carrier for the treatment and/or the prevention of mammal (including human) dermatological lesions, selected from the group consisting of bruises (possibly associated with cosmetic surgery) , vascular disorders of the skin such as small broken vessels, spider veins, varicoses, blotches on the face, any purpura on the face, body and legs (including actinic purpura and post laser skin treatment purpura) , irritation of the skin following chemical peel, Sham als's disease and a mixture thereof.
- composition according to the invention also presents other advantageous associated therapeutical effects when applied upon dermatological lesions, such as topical anti-inflammatory effects upon the human skin.
- the composition used according to the invention is either in the form of a pharmaceutical composition or a cosmetic composition comprising an adequate pharmaceutical or cosmetic carrier or diluent .
- the cosmetic composition further comprises a sufficient amount of a penetrating agent such as phospholipids, preferably said penetrating agent is in the form of nanosomes, described hereafter.
- a penetrating agent such as phospholipids
- Examples of a cosmetic composition could be in the form of a cream, a gel, a lotion and/or a liquid.
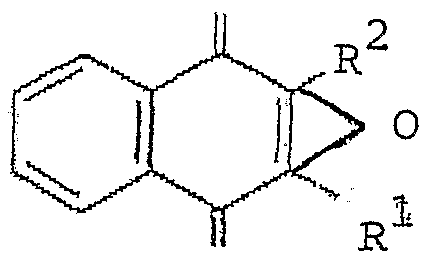
- the vitamin Kl oxide present in the composition has the following formula (I) derivative: o
- Rl is an alkyl group, preferably an alkyl chain comprising between 3 and 20 carbons, preferably an alkyl chain of 12 carbons, possibly branched, more preferably of fomula (II) .
- the vitamin present in the composition has the formula I wherein Rl is of formula II and R2 is a methyl group (vitamin Kl oxyde) .
- the compound of the invention (vitamin Kl oxide or its derivative) is present in nano-sized lipidic particles (hereafter called nanosomes) , preferably lipidic particles having a diameter comprised between about 50 and about 400 nanometers, more preferably between about 100 and about 350 nanometers, between about 120 and about 300 nanometers, between about 155 nanometers and about 200 nanometers, more preferably about 180 nanometers (+ 30 nanometers) .
- the stability of the lipidic nanosome is obtained with particles having a suitable dimension of about 180 nanometers and more than 80% (preferably all) of the nanosomes of the composition should reach the same size of about 180 nanometers. If it happens that a difference in such size exceeds 30%, then a fusion process will occur meaning the formation of greater nanosomes that will become instable and further will destroyed themselves by breaking.
- the extend structure of the nanosome has the same physico-chemical properties that the cell membrane and therefore, the nanosome is able to penetrate easily and quickly the skin and improve the cosmetic and therapeutic properties of the compound (vitamin Kl oxide or its derivative) . Furthermore, the integration of the compound (vitamin Kl oxide or its derivative) in nanosomes will enhance therapeutical and cos etical efficacy while using less substance.
- the nanosomes are made of single or ulti- lipidic layers of phospholipids, preferably of phosphatidylcholine.
- the nanosomes are single lipidic monolayers of phospholipids, preferably of phosphatidylcholine .
- the compound vitamin Kl oxide or its derivative
- said sufficient amount is comprised between 0.5% wt and about 10% wt of the total composition, preferably about 5% wt of the total composition (the total % wt of the composition being 100%) .
- the composition according to the invention advantageously comprises other efficient cosmetic or pharmaceutical compounds, such as other vitamins (which could be present in nanosomes, preferably having the same size as the ones which include vitamin Kl oxide or its derivative) .
- Preferred vitamins are vitamin A, vitamin C and vitamin E which present advantageously a synergic activity with vitamin Kl oxide or its derivative.
- Vitamins C and E are able to maintain iron under a bivalent form (ferrous) avoiding transformation to trivalent form (ferric) .
- the vitamin A pure retinol
- the efficacy is reduced.
- retinol is preferably introduced into polymer system of micro particles that deliver the retinol slowly through the stratum corneum, only when the product is applied directly on the skin.
- the polymer sphere will remain on the surface of the skin.
- the concentration of said compound could be reduce in the cosmetic composition according to the invention, especially if the treatment should take several months .
- vitamin activity preferably said compounds are selected from the group consisting of the following elements with the following preferred (wt) %: phytonadione (about 0.5 to about 2%), tocopheryl acetate
- compositions a cosmetic composition of the invention
- aqua solvent
- retinol propylene glycol
- triethanolamin neutralizing element
- lecithin improves hydratation
- carbomer thickening element
- ethoxydiglycol penetration agent
- some specific lipids such as phospholipids (penetration agents)
- EDTA complexing agent
- C12-C15 alkyl benzoate caprylic capric triglycerides
- parafinum liquidum cyclomethicone
- glycerine sodium PCA, mica, barium sulfate, titanium dioxide, polysorbate 20, acrylate copolymer, phenoxyethanol , acrylate C10-C30, alkyl crosspolymer, propyl paraben, menthyl paraben, alcohol (conservative) , propylene glycol (moistening agent) , BHT or BHA (antioxidants) , ...
- the phospholipids used in the present invention improve the skin penetration of vitamins, especially vitamin Kl oxide or its derivative and vitamin A.
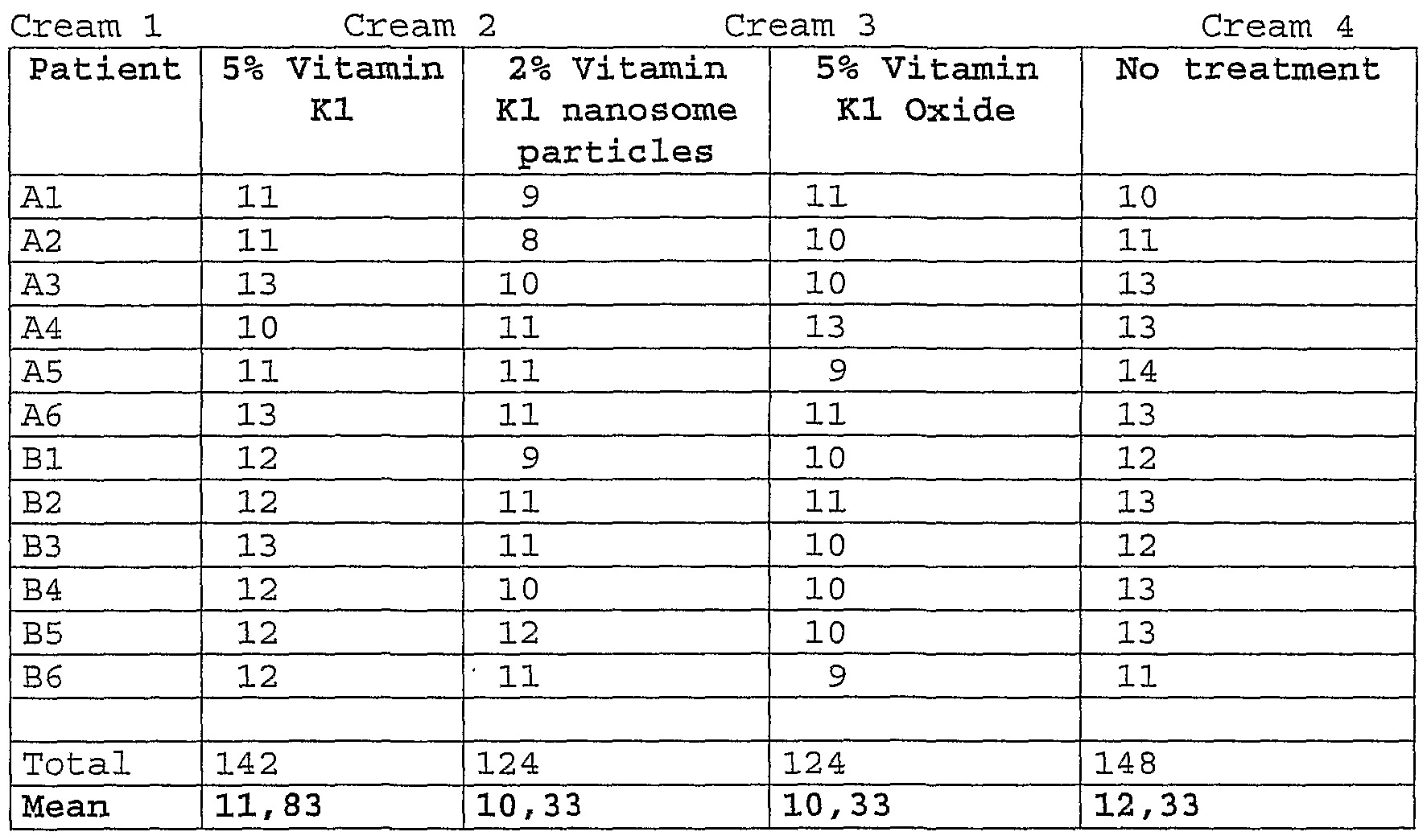
- Example 1 Protocol for bruises
- three compositions cream cosmetic compositions
- the study was conducted in double blind on 12 human volunteers (six males and six females) .
- the purpose of the study was to show a better or at least a similar activity of vitamin Kl oxide versus vitamin Kl in resolution of bruises.
- the table 1 presents 4 different creams and their contents. Table 1
- the group vitamin Oxide shows the best results compared to other formulation comprising vitamin Kl.
- the composition (a cosmetic composition according to the invention) may be applied thinly twice a day, morning and evening, after cleansing of the skin; gently massage into skin and till the gel is absorbed, is preferred.
- the composition according to the invention is recommended for use for 10 to 15 days as preparatory skin care before and after surgical and medical cosmetic procedure .
- the composition according to the invention should preferably be applied before all other beauty or cosmetic skin care products and can be used as a base for those preparations; said composition should not be applied directly upon wounds, mucus areas or eyes due to some hyper-sensitivity.
- vitamin Kl oxide reduces also unexpectedly the side effects of a cosmetic composition.
- the inventors have observed that in presence of light (and possibly improved with the addition of vitamin A) , vitamin Kl is transformed into enadione (vitamin K3) and 1.4.Naphtoquinone that induces allergic side effects. Such transformation of vitamin Kl oxide is not observed in the presence of light and these side effects are not present when the cosmetic composition according to the invention is applied upon mammal skin .
- vitamin Kl oxide does not allow the formation of chromophoric group .
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Dermatology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nanotechnology (AREA)
- Crystallography & Structural Chemistry (AREA)
- Birds (AREA)
- Biophysics (AREA)
- Biotechnology (AREA)
- Molecular Biology (AREA)
- Medical Informatics (AREA)
- General Engineering & Computer Science (AREA)
- Toxicology (AREA)
- Diabetes (AREA)
- Nutrition Science (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Rheumatology (AREA)
- Pain & Pain Management (AREA)
- Heart & Thoracic Surgery (AREA)
- Cardiology (AREA)
- Vascular Medicine (AREA)
- Cosmetics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE602004016292T DE602004016292D1 (en) | 2003-01-20 | 2004-01-20 | USE OF A COMPOSITION WITH VITAMIN K1-OXIDE OR A DERIVATIVES THEREOF FOR TREATING AND / OR PREVENTING SKIN INJURY IN MAMMALS |
| US10/542,914 US7939568B2 (en) | 2003-01-20 | 2004-01-20 | Use of a composition comprising vitamin K1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions |
| DK04703319T DK1594456T3 (en) | 2003-01-20 | 2004-01-20 | Use of a composition containing vitamin K1 oxide or a derivative thereof for treating and / or preventing dermatological lesions in mammals |
| SI200430948T SI1594456T1 (en) | 2003-01-20 | 2004-01-20 | Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions |
| CA2513324A CA2513324C (en) | 2003-01-20 | 2004-01-20 | Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions |
| EP04703319A EP1594456B1 (en) | 2003-01-20 | 2004-01-20 | Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions |
| JP2006500422A JP2006515873A (en) | 2003-01-20 | 2004-01-20 | Use of a composition containing vitamin K1 oxide or a derivative thereof in the treatment and / or prevention of mammalian dermatological lesions |
| HK06107056.0A HK1087010A1 (en) | 2003-01-20 | 2006-06-21 | Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US31988703P | 2003-01-20 | 2003-01-20 | |
| US60/319,887 | 2003-01-20 | ||
| EP03447019A EP1442738A1 (en) | 2003-01-28 | 2003-01-28 | Dermatological cosmetic composition comprising vitamin K1 oxide |
| EP03447019.5 | 2003-01-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004064798A1 true WO2004064798A1 (en) | 2004-08-05 |
Family
ID=32773677
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/BE2004/000011 WO2004064798A1 (en) | 2003-01-20 | 2004-01-20 | Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US7939568B2 (en) |
| EP (1) | EP1594456B1 (en) |
| JP (1) | JP2006515873A (en) |
| KR (1) | KR20050103906A (en) |
| CN (1) | CN100531727C (en) |
| AT (1) | ATE406868T1 (en) |
| CA (1) | CA2513324C (en) |
| DE (1) | DE602004016292D1 (en) |
| DK (1) | DK1594456T3 (en) |
| ES (1) | ES2314365T3 (en) |
| HK (1) | HK1087010A1 (en) |
| PT (1) | PT1594456E (en) |
| SI (1) | SI1594456T1 (en) |
| WO (1) | WO2004064798A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2885802A1 (en) * | 2005-05-17 | 2006-11-24 | Oreal | Cosmetic use of a vitamin K in a composition as an agent to improve and/or repair the barrier function of skin or semi-mucous membranes |
| EP2178818A2 (en) * | 2007-07-24 | 2010-04-28 | Viridis Biopharma Pvt Ltd. | Treatments using vitamin k analogues and derivatives |
| US8003839B2 (en) | 2006-02-03 | 2011-08-23 | Exxonmobil Chemical Patents Inc. | Process for generating linear apha olefin comonomers |
| US8076524B2 (en) | 2006-02-03 | 2011-12-13 | Exxonmobil Chemical Patents Inc. | Process for generating alpha olefin comonomers |
| WO2015002151A1 (en) * | 2013-07-01 | 2015-01-08 | 株式会社明治 | Method for improving photostability of food, beverage or the like |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9421297B2 (en) | 2014-04-02 | 2016-08-23 | Adhezion Biomedical, Llc | Sterilized compositions of cyanoacrylate monomers and naphthoquinone 2,3-oxides |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB744376A (en) * | 1952-03-08 | 1956-02-08 | Merck & Co Inc | Vitamin emulsions |
| US3070499A (en) * | 1960-09-19 | 1962-12-25 | Merck & Co Inc | Parenteral aqueous solutions of fat soluble vitamins |
| JPH05320039A (en) * | 1991-09-20 | 1993-12-03 | Taiyo Kagaku Co Ltd | Skin-beautifying cosmetic |
| WO1994000135A1 (en) * | 1992-06-30 | 1994-01-06 | Shapiro Howard K | Composition containing amine and amine-related derivatives of benzoic acid and uses therefor including treating inflammatory diseases |
| WO1997039746A1 (en) * | 1996-04-22 | 1997-10-30 | Advanced Polymer Systems, Inc. | Method of and composition for treating disorders of the skin using vitamin k |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0643775B1 (en) * | 1992-05-28 | 2004-07-21 | Centre For Molecular Biology And Medicine, | Quinone derivatives for enhancing cellular bioenergy |
| US5510391A (en) * | 1993-10-22 | 1996-04-23 | Mayapple Holdings, Llc | Method of treating blood vessel disorders of the skin using vitamin K |
| US5540934A (en) * | 1994-06-22 | 1996-07-30 | Touitou; Elka | Compositions for applying active substances to or through the skin |
| US5945409A (en) * | 1995-03-10 | 1999-08-31 | Wilson T. Crandall | Topical moisturizing composition and method |
| US6180136B1 (en) * | 1998-11-10 | 2001-01-30 | Idexx Laboratories, Inc. | Phospholipid-coated microcrystals for the sustained release of pharmacologically active compounds and methods of their manufacture and use |
-
2004
- 2004-01-20 AT AT04703319T patent/ATE406868T1/en active
- 2004-01-20 WO PCT/BE2004/000011 patent/WO2004064798A1/en active IP Right Grant
- 2004-01-20 SI SI200430948T patent/SI1594456T1/en unknown
- 2004-01-20 DE DE602004016292T patent/DE602004016292D1/en not_active Expired - Lifetime
- 2004-01-20 CA CA2513324A patent/CA2513324C/en not_active Expired - Fee Related
- 2004-01-20 KR KR1020057013381A patent/KR20050103906A/en not_active Application Discontinuation
- 2004-01-20 US US10/542,914 patent/US7939568B2/en not_active Expired - Lifetime
- 2004-01-20 DK DK04703319T patent/DK1594456T3/en active
- 2004-01-20 CN CNB2004800024903A patent/CN100531727C/en not_active Expired - Fee Related
- 2004-01-20 PT PT04703319T patent/PT1594456E/en unknown
- 2004-01-20 ES ES04703319T patent/ES2314365T3/en not_active Expired - Lifetime
- 2004-01-20 EP EP04703319A patent/EP1594456B1/en not_active Expired - Lifetime
- 2004-01-20 JP JP2006500422A patent/JP2006515873A/en active Pending
-
2006
- 2006-06-21 HK HK06107056.0A patent/HK1087010A1/en not_active IP Right Cessation
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB744376A (en) * | 1952-03-08 | 1956-02-08 | Merck & Co Inc | Vitamin emulsions |
| US3070499A (en) * | 1960-09-19 | 1962-12-25 | Merck & Co Inc | Parenteral aqueous solutions of fat soluble vitamins |
| JPH05320039A (en) * | 1991-09-20 | 1993-12-03 | Taiyo Kagaku Co Ltd | Skin-beautifying cosmetic |
| WO1994000135A1 (en) * | 1992-06-30 | 1994-01-06 | Shapiro Howard K | Composition containing amine and amine-related derivatives of benzoic acid and uses therefor including treating inflammatory diseases |
| WO1997039746A1 (en) * | 1996-04-22 | 1997-10-30 | Advanced Polymer Systems, Inc. | Method of and composition for treating disorders of the skin using vitamin k |
Non-Patent Citations (1)
| Title |
|---|
| DATABASE WPI Section Ch Week 199402, Derwent World Patents Index; Class B05, AN 1994-012183, XP002244372 * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2885802A1 (en) * | 2005-05-17 | 2006-11-24 | Oreal | Cosmetic use of a vitamin K in a composition as an agent to improve and/or repair the barrier function of skin or semi-mucous membranes |
| US8003839B2 (en) | 2006-02-03 | 2011-08-23 | Exxonmobil Chemical Patents Inc. | Process for generating linear apha olefin comonomers |
| US8076524B2 (en) | 2006-02-03 | 2011-12-13 | Exxonmobil Chemical Patents Inc. | Process for generating alpha olefin comonomers |
| EP2178818A2 (en) * | 2007-07-24 | 2010-04-28 | Viridis Biopharma Pvt Ltd. | Treatments using vitamin k analogues and derivatives |
| EP2178818A4 (en) * | 2007-07-24 | 2010-07-28 | Viridis Biopharma Pvt Ltd | Treatments using vitamin k analogues and derivatives |
| AU2008322224B2 (en) * | 2007-07-24 | 2012-05-10 | Synergia Life Sciences Pvt Limited | Treatments using vitamin K analogues and derivatives |
| WO2015002151A1 (en) * | 2013-07-01 | 2015-01-08 | 株式会社明治 | Method for improving photostability of food, beverage or the like |
| JPWO2015002151A1 (en) * | 2013-07-01 | 2017-02-23 | 株式会社明治 | Method for improving photostability of food and drink |
Also Published As
| Publication number | Publication date |
|---|---|
| CN100531727C (en) | 2009-08-26 |
| CA2513324A1 (en) | 2004-08-05 |
| KR20050103906A (en) | 2005-11-01 |
| SI1594456T1 (en) | 2009-02-28 |
| US7939568B2 (en) | 2011-05-10 |
| ATE406868T1 (en) | 2008-09-15 |
| US20060154983A1 (en) | 2006-07-13 |
| HK1087010A1 (en) | 2006-10-06 |
| PT1594456E (en) | 2008-12-16 |
| DE602004016292D1 (en) | 2008-10-16 |
| DK1594456T3 (en) | 2009-01-19 |
| CA2513324C (en) | 2010-09-21 |
| ES2314365T3 (en) | 2009-03-16 |
| EP1594456A1 (en) | 2005-11-16 |
| JP2006515873A (en) | 2006-06-08 |
| CN1738594A (en) | 2006-02-22 |
| EP1594456B1 (en) | 2008-09-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2461941C (en) | Anti-irritating rosacea treatment | |
| TW510799B (en) | Skin care compositions containing retinoids and having a low irritation profile | |
| ES2565317T3 (en) | Topical skin care composition | |
| JPH11514999A (en) | Treatment of pruritus with vitamin D and its derivatives | |
| AU2002334601A1 (en) | Anti-irritating rosacea treatment | |
| MXPA06001381A (en) | Foam carrier containing amphiphilic copolymeric gelling agent. | |
| WO2005027977A2 (en) | Diclofenac compositions for the treatment of skin disorders | |
| JP2020529392A (en) | Compositions for the prevention or treatment of scars | |
| WO2009042402A2 (en) | Composition and method for treating rosacea | |
| US11400071B2 (en) | Hest G-18-0 and benzoyl peroxide compositions and methods for using the same | |
| CA2513324C (en) | Use of a composition comprising vitamin k1 oxide or a derivative thereof for the treatment and/or the prevention of mammal dermatological lesions | |
| EP2265268A2 (en) | Stable fixed dose topical formulation | |
| JP2008513420A (en) | Use of metronidazole in combination with azelaic acid for the treatment of rosacea | |
| JP2024516652A (en) | Topical preparations containing benzoyl peroxide and azelaic acid and uses thereof | |
| EP1996514B1 (en) | Removal of skin changes | |
| WO2021189077A1 (en) | Methods for treating acne | |
| Swetha et al. | African Journal of Pharmaceutical Sciences | |
| EP1442738A1 (en) | Dermatological cosmetic composition comprising vitamin K1 oxide | |
| Naudé | Formulation, in vitro release and transdermal diffusion of Vitamin A and Zinc for the treatment of acne | |
| BR102013006316A2 (en) | COMPOSITION FOR PREVENTIVE AND / OR CURATIVE TREATMENT OF TISSUE INJURIES AND / OR ULCERS; USE OF VITAMIN A AND VITAMIN E, PACKED PRODUCT | |
| JP2001322930A5 (en) | ||
| WO2015179570A1 (en) | Waterborne topical compositions for the delivery of azelaic acid for treatment of skin conditions such as acne vulgaris, rosacea seborrheic dermatitis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2513324 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006500422 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 3209/DELNP/2005 Country of ref document: IN Ref document number: 20048024903 Country of ref document: CN Ref document number: 1020057013381 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004703319 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020057013381 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004703319 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2006154983 Country of ref document: US Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10542914 Country of ref document: US |
|
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWP | Wipo information: published in national office |
Ref document number: 10542914 Country of ref document: US |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2004703319 Country of ref document: EP |