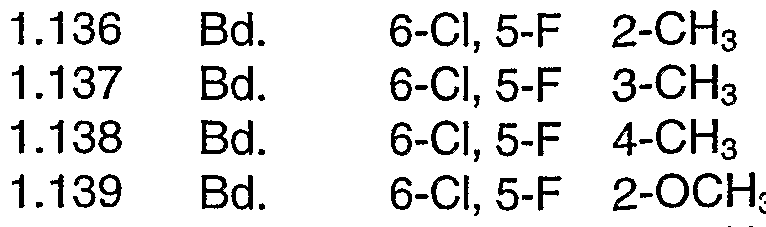WO2004024704A1 - Organic compounds - Google Patents
Organic compounds Download PDFInfo
- Publication number
- WO2004024704A1 WO2004024704A1 PCT/EP2003/010047 EP0310047W WO2004024704A1 WO 2004024704 A1 WO2004024704 A1 WO 2004024704A1 EP 0310047 W EP0310047 W EP 0310047W WO 2004024704 A1 WO2004024704 A1 WO 2004024704A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- halo
- alkyl
- alkoxy
- unsubstituted
- many times
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4192—1,2,3-Triazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/16—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms condensed with carbocyclic rings or ring systems
- C07D249/18—Benzotriazoles
Definitions
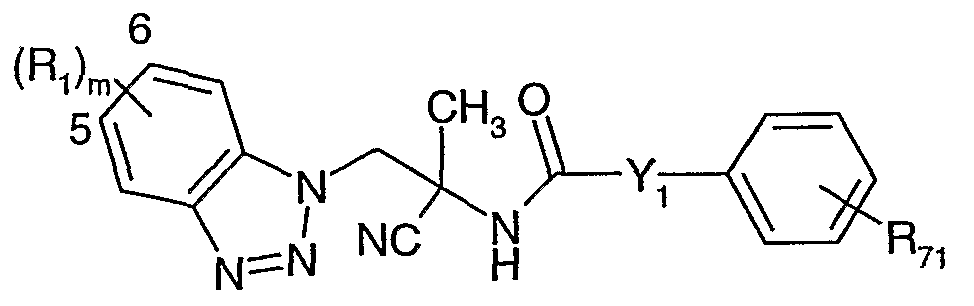
- the present invention relates to new benzotriazolyl-aminoacetonitrile compounds of formula
- Rx signifies halogen, cyano, nitro, CrC 6 -alkyl, C 3 -C 6 -cycloalkyl, halo-C ⁇ -C 6 -alkyl, C C 6 - alkylthio, arylthio, C T C ⁇ -alkoxy, C 3 -C 6 -cycloalkyloxy, halo-CrC 6 -alkoxy, C C 6 -alkylcarbonyl, halo-C ⁇ -C 6 -alkyicarbonyl, CrC 6 -alkylsulfinyl, CrC 6 -alkylsulfonyl, CrC 6 -alkylamino, di(C C 6 - alkyl)amino, unsubstituted or substituted aryl or unsubstituted or substituted phenoxy, whereby the substituents may each be independent of one another and are selected from the group consisting of halogen, nitro, cyano, Cr
- R 3 , R 4 und R 5 either, independently of one another, signify hydrogen, halogen, C C 6 -alkyl, halo-C C 6 -alkyI; C 3 -C 6 -cycloalkyl that is either unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen and C r C 6 -alkyl; phenyl that is either unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C C 6 -alkyl, halo-CrC- 6 -alkyl, C C 6 -alkoxy, halo-CrC-e-alkoxy, CrC 6 -alkylthio, haIo-CrC 6 -alkylthio, C C 6 -alkylsulfinyl,
- R 6 signifies hydrogen, C C 6 -alkyl, d-C-e-alkylcarbonyl, CrCe-alkoxy-CrCe-aikyl, aminocarbonyl, C ⁇ -C 6 -alkoxycarbonyl, halo-C C 6 -alkylcarbonyl, thio-C C 6 -alkylcarbonyl or benzyl;
- R 7 signifies hydrogen, CrC 6 -aIkyl, C r C 6 -alkoxy, CrCe-alkylamino, di(C ⁇ -C 6 -alkyl)amino, piperonyl, phenyl which is unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C ⁇ -C 6 -alkyl, C 3 -C 6 -cycloalkyl, halo-CrC 6 -alkyl, C C 6 -alkoxy, C 3 -C 6 - cycloalkyloxy, haIo-C C 6 -alkoxy, C 2 -C 6 -alkenyl, halo-C 2 -C 6 -alkenyl, C 2 -C 6 -alkinyl, C 3 -C 6 - cycloalkyl, C 2 -C 6
- R 8 and R 9 independently of one another, signify hydrogen, d-C 6 -alkyl, d-C 6 - alkoxycarbonyl, CrC 6 -alkylcarbonyl, C ⁇ -C 6 -alkylthiocarbonyl, thio-C C 6 -alkylcarbonyl, aryl or hetaryl;
- aminoacetonitrile compounds having pesticidal activity are described for example in EP-0.953.565 A2.
- the active ingredients specifically disclosed therein cannot always fulfil the requirements regarding potency and activity spectrum. There is therefore a need for active ingredients with improved pesticidal properties. It has now been found that the aminoacetonitrile compounds of formula I have excellent pesticidal properties, especially against endo- and ecto-parasites in and on productive livestock and domestic animals and plants.
- Aryl is phenyl or naphthyl.
- Hetaryl is pyridyl, pyrimidyl, s-triazinyl, 1 ,2,4-triazinyl, thienyl, furanyl, pyrryl, pyrazolyl, imidazolyl, thiazolyl, triazolyl, oxazolyl, thiadiazolyl, oxadiazolyl, benzothienyl, benzofuranyl, benzothiazolyl, indolyl or indazolyl, preferably pyridyl, pyrimidyl, s-triazinyl or 1 ,2,4-triazinyl, especially pyridyl or pyrimidyl.
- Alkyl - as a group perse and as structural element of other groups and compounds, for example halogen-alkyl, alkoxy, and alkylthio - is, in each case with due consideration of the specific number of carbon atoms in the group or compound in question, either straight- chained, i.e. methyl, ethyl, propyl, butyl, pentyl or hexyl, or branched, e.g. isopropyl, isobutyl, sec-butyl, tert.-butyl, isopentyl, neopentyl or isohexyl.
- Alkenyl - as a group perse and as structural element of other groups and compounds - is, in each case with due consideration of the specific number of carbon atoms in the group or compound in question and of the conjugated or isolated double bonds - either straight- chained, e.g. allyl, 2-butenyl, 3-pentenyl, 1 -hexenyl or 1 ,3-hexadienyl, or branched, e.g. isopropenyl, isobutenyl, isoprenyl, tert.-pentenyl or isohexenyl.
- straight- chained e.g. allyl, 2-butenyl, 3-pentenyl, 1 -hexenyl or 1 ,3-hexadienyl, or branched, e.g. isopropenyl, isobutenyl, isoprenyl, tert.-pentenyl or isohexen
- Alkinyl - as a group per se and as structural element of other groups and compounds - is, in each case with due consideration of the specific number of carbon atoms in the group or compound in question and of the conjugated or isolated double bonds - either straight- chained, e.g. propargyl, 2-butinyl, 3-pentinyl, 1-hexinyl, 1-heptinyl or 3-hexen-1-inyl, or branched, e.g. 3-methylbut-1-inyl, 4-ethylpent-1-inyl or 4-methylhex-2-inyl.
- straight- chained e.g. propargyl, 2-butinyl, 3-pentinyl, 1-hexinyl, 1-heptinyl or 3-hexen-1-inyl, or branched, e.g. 3-methylbut-1-inyl, 4-ethylpent-1-inyl or 4-methylhex-2-inyl.
- Cycloalkyl - as a group perse and as structural element of other groups and compounds such as halocycloalkyl, - is, in each case with due consideration of the specific number of carbon atoms in the group or compound in question, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
- Halogen - as a group perse and as structural element of other groups and compounds such as haloalkyl, haloalkoxy and haloalkylthio - is fluorine, chlorine, bromine or iodine, especially fluorine, chlorine or bromine, in particular fluorine or chlorine.
- Halogen-substituted carbon-containing groups and compounds such as haloalkyl, haloalkoxy or haloalkylthio, may be partially halogenated or perhalogenated, whereby in the case of multiple halogenation, the halogen substituents may be identical or different.
- halogen-alkyl - as a group perse and as structural element of other groups and compounds such as haloalkoxy or haloalkylthio, - are methyl which is mono- to trisubstituted by fluorine, chlorine and/or bromine, such as CHF 2 or CF 3 ; ethyl which is mono- to pentasubstituted by fluorine, chlorine and/or bromine, such as CH 2 CF 3> CF 2 CF 3 , CF 2 CCI 3 , CF 2 CHCI 2) CF 2 CHF 2 , CF 2 CFCI 2 , CF 2 CHBr 2 , CF 2 CHCIF, CF 2 CHBrF or CCIFCHCIF; propyl or isopropyl, mono- to heptasubstituted by fluorine, chlorine and/or bromine, such as CH 2 CHBrCH 2 Br, CF 2 CHFCF 3 , CH 2 CF 2 CF 3 or CH
- Alkoxy groups preferably have a chain length of 1 to 6 carbon atoms.
- Alkoxy is for example methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, isobutoxy, sec.-butoxy and tert.-butoxy, as well as the isomers pentyloxy and hexyloxy; preferably methoxy and ethoxy.
- Haloalkoxy groups preferably have a chain length of 1 to 6 carbon atoms. Haloalkoxy is e.g.
- Alkylthio groups preferably have a chain length of 1 to 6 carbon atoms.
- Alkylthio is for example methylthio, ethylthio, propylthio, isopropylthio, n-butylthio, isobutylthio, sec.-butylthio or tert.-butylthio, preferably methylthio and ethylthio.
- R 3 , R 4 and R 5 are either, independently of one another, hydrogen, halogen, C 1 -C 4 -alkyl, halo-d-C 4 -alkyl, C 3 -C 6 -cycloalkyl; phenyl that is either unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C C 4 -alkyl, halo-C C 4 -alkyl, d-C 4 -alkoxy, halo-C r C -alkoxy; d-C 4 -aIkylthio and halo-C C 4 -alkylthio; or R 4 and R 5 together are C 2 -C 6 -alkylene; especially, independently of one another, hydrogen, halogen, CrC- 2 -alkyl, halo-CrC 2 -al
- R 7 signifies phenyl which is unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C r C 4 -alkyl, halo-d-C -alkyl, CrC ⁇ alkoxy, halo-CrC 4 -alkoxy, C 2 -C 4 -alkenyl, halo-C 2 -C 4 -alkenyl, C 2 -C 4 - alkinyl, C 3 -C 6 -cycloalkyl, C 2 -C -alkenyloxy, halo-C 2 -C 4 -alkenyloxy, C C 4 -alkylthio, ha!o-C C 4 -alkylthio, C 1 -C 4 -alkylsulfonyloxy, halo-C C 4 -alkyl
- R 8 und R 9 independently of one another, signify hydrogen, C C 6 -alkyl, CrC 6 -alkoxycarbonyl, C C 6 -alkylcarbonyl or aryl; especially, independently of one another, hydrogen or d-C 4 -alkyl; most particularly, independently of one another, hydrogen or d-C 2 -alkyl;
- a compound of formula I, wherein a is 1 or 2; especially 1 ;
- a compound of formula I, wherein m is 1, 2 or 3; especially 1 or 2;
- Ri signifies halogen, cyano, nitro, CrC -alkyl, halo-C C 4 -alkyl, CrCralkoxy, halo-C C 4 - alkoxy, d-C 4 -alkylcarbonyl, halo-CrC ⁇ alkylcarbonyl, d-C -alkylsulfonyl or unsubstituted or substituted phenoxy, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C ⁇ -C 4 -alkyl, haIo-d-C 4 -alkyl, C C 4 -alkoxy and halo-C C 4 -alkoxy;
- R 3 , R 4 and R 5 independently of one another, are hydrogen, halogen, C r C 4 -alkyl, halo-C r C -alkyl, C 3 -C 6 -cycloalkyI; phenyl that is either unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C C -alkyl, halo-d-C -alkyl, C ⁇ -C 4 -alkoxy, halo-d-C -alkoxy; C C 4 -alkylthio and halo-C C 4 -alkylthio; or R 4 and R 5 together are C 2 -C 6 - alkylene;
- R 6 is hydrogen, C C 4 -alkyl, C C 4 -alkylcarbonyl, d-Ce-alkoxy-C C ⁇ -alkyl or benzyl;
- R 7 signifies phenyl which is unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, d-C 4 -alkyl, halo-C C -alkyl, d-C 4 -alkoxy, halo-C r C 4 -alkoxy, C 2 - C 4 -alkenyl, halo-C 2 -C 4 -alkenyl, C 2 -C 4 -alkinyl, C 3 -C 6 -cycloalkyl, C 2 -C 4 -alkenyloxy, ha!o-C 2 -C 4 - alkenyloxy, d-C 4 -aikylthio, halo-CrC ⁇ alkylthio, C C 4 -alkylsulfonyloxy, halo-C r C 4 - alkylsul
- R 8 und R 9 independently of one another, signify hydrogen, C r C 6 -alkyl, C Ce-alkoxycarbonyl, CrCe-alkylcarbonyl or aryl;
- Y is C(O) or S(O) n ; a signifies 1 or 2; m is 1 , 2 or 3 and n signifies 2;
- R T signifies halogen, cyano, nitro, CrC 2 -alkyl, halo-d-C 2 -alkyl, C C 2 -alkoxy, halo-C C 2 - alkoxy or unsubstituted or substituted phenoxy, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, C C - alkyl, halo-C C 4 -alkyl, C r C 4 -alkoxy and halo-d-C 4 -alkoxy;
- R 3 , R 4 and R 5 independently of one another, signify hydrogen, halogen, C C 2 -alkyl, halo-C C 2 -alkyl or C 3 -C 6 -cycloalkyl;
- R 6 signifies hydrogen, d-C 2 -alkyl, or benzyl
- R 7 signifies phenyl which is unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, nitro, cyano, C C 2 -alkyl, halo-d-C 2 -alkyl, C r C 2 -alkoxy, halo-Crd-alkoxy, C 3 - C 5 -cycloalkyl, C r C 2 -alkylthio, halo-CrC 2 -alkylthio, CrC 2 -alkylsuIfonyl, halo-C r C 2 - alkylsulfonyl, C C 2 -alkylcarbonyl, halo-C C 2 -alkylcarbonyl, C r C 2 -alkoxycarbonyl; aryl-d-C 2 - alkyl which is unsubstituted or substituted once or many times, ary
- R 8 and R 9 independently of one another, signify hydrogen or C C 4 -alkyl
- Y signifies C(O); a signifies 1 ; and m is 1 or 2;
- Ri signifies halogen, cyano, nitro, C C 2 -alkyl, halo-d-C 2 -alkyl, C 1 -C 2 -alkoxy or halo-C C 2 - alkoxy;
- R 3 , R and R 5 independently of one another, signify hydrogen, methyl or halomethyl
- R 6 signifies hydrogen or C r C 2 -alkyl
- R 7 signifies phenyl which is unsubstituted or substituted once or many times, whereby the substituents may be independent of one another and are selected from the group consisting of halogen, cyano, d-C- 2 -alkyl, halo-C C 2 -alkyl, C r C 2 -alkoxy, halo-C C 2 -alkoxy, C 3 -C 5 - cycloalkyl, d-C 2 -alkylcarbonyl, halo-d-C 2 -alkylcarbonyl, C C 2 -alkoxycarbonyl; aryl-C C 2 - alkyl which is unsubstituted or substituted once or many times, and aryloxy-d-C 2 -alkyl which is unsubstituted or substituted once or many times, whereby the substituents may each be independent of one another and are selected from the group consisting of halogen, cyano, d-C 2
- R 8 and R 9 independently of one another, signify hydrogen or C r C 2 -alkyl
- Y signifies C(O); a signifies 1 ; and m is 1 or 2.
- a further object of the invention is the process for the preparation of the compounds of formula I, respectively in free form or in salt form, for example characterised in that a compound of formula
- reaction partners can be reacted with one another as they are, i.e. without the addition of a solvent or diluent, e.g. in the melt. In most cases, however, the addition of an inert solvent or diluent, or a mixture thereof, is of advantage.
- solvents or diluents are: aromatic, aliphatic and alicyclic hydrocarbons and halogenated hydrocarbons, such as benzene, toluene, xylene, mesitylene, tetraline, chlorobenzene, dichlorobenzene, bromobenzene, petroleum ether, hexane, cyclohexane, dichloromethane, trichloromethane, tetrachloromethane, dichloroethane, trichloroethene or tetrachloroethene; ethers, such as diethyl ether, dipropyl ether, diisopropyl ether, dibutyl ether, tert-butyl methyl ether, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol dimethylether, dimethoxydiethylether, tetrahydrofuran or dio
- Preferred leaving groups are halogens, especially chlorine.
- Suitable bases for facilitating the reaction are e.g. alkali metal or alkaline earth metal hydroxides, hydrides, amides, alkanolates, acetates, carbonates, dialkylamides or alkylsilyl- amides; alkylamines, alkylenediamines, optionally N-alkylated, optionally unsaturated, cyclo- alkylamines, basic heterocycles, ammonium hydroxides, as well as carbocyclic amines.
- alkali metal or alkaline earth metal hydroxides, hydrides, amides, alkanolates, acetates, carbonates, dialkylamides or alkylsilyl- amides alkylamines, alkylenediamines, optionally N-alkylated, optionally unsaturated, cyclo- alkylamines, basic heterocycles, ammonium hydroxides, as well as carbocyclic amines.
- the reaction advantageously takes place in a temperature range of ca. 0°C to ca. 100°C , preferably from ca. 10°C to ca. 40°C .
- a compound of formula II is reacted at room temperature in a halogenated hydrocarbon, preferably dichloromethane, with a compound of formula III in the presence of a base, preferably a mixture of diisopropylethylamine and 4-(N,N- dimethylamino)pyridine.
- a base preferably a mixture of diisopropylethylamine and 4-(N,N- dimethylamino)pyridine.
- a further object of the invention is the process for the preparation of the compounds of formula II, respectively in free form or in salt form, for example characterised in that a compound of formula which is known or may be produced analogously to corresponding known compounds, in which Ri, R 3 , R , R 5 , a and m are defined as for formula I, is reacted with an inorganic or organic cyanide and a compound of formula R 6 -NH 2 , which is known or may be produced analogously to corresponding known compounds and wherein R 6 is defined as for formula I, and if desired, a compound of formula II obtainable according to the method or in another way, respectively in free form or in salt form, is converted into another compound of formula II, a mixture of isomers obtainable according to the method is separated and the desired isomer isolated and/or a free compound of formula II obtainable according to the method is converted into a salt or a salt of a compound of formula II obtainable according to the method is converted into the free compound of formula II or into another salt.
- Suitable cyanides are sodium cyanide, potassium cyanide, trimethylsilyl cyanide and acetone cyanohydrin.
- the general method for reacting carbonyl compounds, e.g. of formula IV, with cyanides and amines, e.g. of formula R 6 -NH 2 is a Strecker reaction, for example as in Organic Synthesis Coll. Vol. 3, 88 (1973).
- Salts of compounds I may be produced in known manner. Acid addition salts of compounds I, for example, are obtainable by treatment with a suitable acid or a suitable ion exchange reagent, and salts with bases are obtainable by treatment with a suitable base or a suitable ion exchange reagent.
- Salts of compounds I can be converted into the free compounds I by the usual means, acid addition salts e.g. by treating with a suitable basic composition or with a suitable ion exchange reagent, and salts with bases e.g. by treating with a suitable acid or a suitable ion exchange reagent.
- Salts of compounds I can be converted into other salts of compounds I in a known manner; acid addition salts can be converted for example into other acid addition salts, e.g. by treating a salt of an inorganic acid, such as a hydrochloride, with a suitable metal salt, such as a sodium, barium, or silver salt, of an acid, e.g. with silver acetate, in a suitable solvent, in which a resulting inorganic salt, e.g. silver chloride, is insoluble and thus precipitates out from the reaction mixture.
- a salt of an inorganic acid such as a hydrochloride
- a suitable metal salt such as a sodium, barium, or silver salt
- a resulting inorganic salt e.g. silver chloride
- compounds I with salt-forming characteristics can be obtained in free form or in the form of salts.
- Salts of compounds I may be produced in known manner. Acid addition salts of compounds I, for example, are obtainable by treatment with a suitable acid or a suitable ion exchange reagent, and salts with bases are obtainable by treatment with a suitable base or a suitable ion exchange reagent.
- Salts of compounds I can be converted into the free compounds I by the usual means, acid addition salts e.g. by treating with a suitable basic composition or with a suitable ion exchange reagent, and salts with bases e.g. by treating with a suitable acid or a suitable ion exchange reagent.
- Salts of compounds I can be converted into other salts of compounds I in a known manner; acid addition salts can be converted for example into other acid addition salts, e.g. by treating a salt of an inorganic acid, such as a hydrochloride, with a suitable metal salt, such as a sodium, barium, or silver salt, of an acid, e.g. with silver acetate, in a suitable solvent, in which a resulting inorganic salt, e.g. silver chloride, is insoluble and thus precipitates out from the reaction mixture.
- a salt of an inorganic acid such as a hydrochloride
- a suitable metal salt such as a sodium, barium, or silver salt
- a resulting inorganic salt e.g. silver chloride
- Compounds I can also be obtained in the form of their hydrates and/or also can include other solvents, used for example where necessary for the crystallisation of compounds present in solid form.
- Compounds I and II may be optionally present as optical and/or geometric isomers or as a mixture thereof.
- the invention relates both to the pure isomers and to all possible isomeric mixtures, and is hereinbefore and hereinafter understood as doing so, even if stereochemical details are not specifically mentioned in every case.
- Diastereoisomeric mixtures of compounds I which are obtainable by the process or in another way, may be separated in known manner, on the basis of the physical-chemical differences in their components, into the pure diastereoisomers, for example by fractional crystallisation, distillation and/or chromatography.
- Splitting of mixtures of enantiomers, that are obtainable accordingly, into the pure isomers may be achieved by known methods, for example by recrystallisation from an optically active solvent, by chromatography on chiral adsorbents, e.g. high-pressure liquid chromatography (HPLC) on acetyl cellulose, with the assistance of appropriate micro-organisms, by cleavage with specific immobilised enzymes, through the formation of inclusion compounds, e.g. using chiral crown ethers, whereby only one enantiomer is complexed.
- HPLC high-pressure liquid chromatography
- the starting materials and intermediates used are preferably those that lead to the compounds I described at the beginning as being especially useful.
- the invention relates especially to the method of preparation described in the example.
- the compounds I according to the invention are notable for their particularly broad activity spectrum and are valuable active ingredients for use in pest control, including in particular the control of endo- and ecto-parasites on animals, whilst being well-tolerated by warmblooded animals, fish and plants,
- ectoparasites are understood to be in particular insects, mites and ticks. These include insects of the order: Lepidoptera, Coleoptera, Homoptera, Heteroptera, Diptera, Thysanoptera, Orthoptera, Anoplura, Siphonaptera, Mallophaga, Thysanura, Isoptera, Psocoptera and Hymenoptera.
- the ectoparasites which may be mentioned in particular are those which trouble humans or animals and carry pathogens, for example flies such as Musca domestica, Musca vetustissima, Musca autumnalis, Fannia canicularis, Sarcophaga carnaria, Lucilia cuprina, Hypoderma bovis, Hypoderma lineatum, Chrysomyia chloropyga, Dermatobia hominis, Cochliomyia hominivorax, Gasterophilus intestinalis, Oestrus ovis, Stomoxys calcitrans, Haematobia irritans and midges (Nematocera), such as Culicidae, Simuliidae, Psychodidae, but also blood-sucking parasites, for example fleas, such as Ctenocephalides felis and Ctenocephalides canis (cat and dog fleas), Xenopsylla cheopis, Pulex irritans, Dermat
- Haematopota pluvialis such as Haematopota pluvialis, Tabanidea spp. such as Tabanus nigrovittatus, Chrysopsinae spp. such as Chrysops caecutiens, tsetse flies, such as species of Glossinia, biting insects, particularly cockroaches, such as Blatella germanica, Blatta orientalis, Periplaneta americana, mites, such as Dermanyssus gallinae, Sarcoptes scabiei, Psoroptes ovis and Psorergates spp. and last but not least ticks. The latter belong to the order Acarina.
- ticks are, for example, Boophilus, Amblyomma, Anocentor, Dermacentor, Haemaphysalis, Hyalomma, Ixodes, Rhipicentor, Margaropus, Rhipicephalus, Argas, Otobius and Ornithodoros and the like, which preferably infest warm-blooded animals including farm animals, such as cattle, pigs, sheep and goats, poultry such as chickens, turkeys and geese, fur-bearing animals such as mink, foxes, chinchillas, rabbits and the like, as well as domestic animals such as cats and dogs, but also humans.
- farm animals such as cattle, pigs, sheep and goats
- poultry such as chickens, turkeys and geese
- fur-bearing animals such as mink, foxes, chinchillas, rabbits and the like
- domestic animals such as cats and dogs, but also humans.
- Compounds I can also be used against hygiene pests, especially of the order Diptera of the families Sarcophagidae, Anophilidae and Culicidae; the orders Orthoptera, Dictyoptera (e.g. the family Blattidae) and Hymenoptera (e.g. the family Formicidae).
- Compounds I also have sustainable efficacy on parasitic mites and insects of plants.
- spider mites of the order Acarina they are effective against eggs, nymphs and adults of Tetranychidae ⁇ Tetranychus spp. and Panonychus spp.).
- sucking insects of the order Homoptera especially against pests of the families Aphididae, Delphacidae, Cicadellidae, Psyllidae, Loccidae, Diaspididae and Eriophydidae (e.g. rust mite on citrus fruits); the orders Hemiptera, Heteroptera and Thysanoptera, and on the plant-eating insects of the orders Lepidoptera, Coleoptera, Diptera and Orthoptera
- the compounds of formula I are therefore effective against all stages of development of sucking insects and eating insects on crops such as cereals, cotton, rice, maize, soya, potatoes, vegetables, fruit, tobacco, hops, citrus, avocados and other crops.
- the compounds of formula I are also effective against plant nematodes of the species M ⁇ loidogyne, Heterodera, Pratylenchus, Ditylenchus, Radopholus, Rizoglyphus etc.
- the compounds are effective against helminths, in which the endoparasitic nematodes and trematodes may be the cause of serious diseases of mammals and poultry, e.g. sheep, pigs, goats, cattle, horses, donkeys, dogs, cats, guinea-pigs and exotic birds.
- Typical nematodes of this indication are: Haemonchus, Trichostrongylus, Ostertagia, Nematodirus, Cooperia, Ascaris, Bunostonum, Oesophagostonum, Charbertia, Trichuris, Strongylus, Trichonema, Dictyocaulus, Capillaria, Heterakis, Toxocara, Ascaridia, Oxyuris, Ancylostoma, Uncinaria, Toxascaris and Parascaris.
- the trematodes include, in particular, the family of Fasciolideae, especially Fasciola hepatica.
- the particular advantage of the compounds of formula I is their efficacy against those parasites that are resistant towards active ingredients based on benzimidazole.
- Parasites of the families Filariidae and Setariidae may be found in the internal cell tissue and in the organs, e.g. the heart, the blood vessels, the lymph vessels and the subcutaneous tissue.
- a particularly notable parasite is the heartworm of the dog, Dirofilaria immitis.
- the compounds of formula I are highly effective against these parasites.
- the compounds of formula I are suitable for the control of human pathogenic parasites.
- typical representatives that appear in the digestive tract are those of the species Ancylostoma, Necator, Ascaris, Strongyloides, Trichinella, Capillaria, Trichuris and Enterobius.
- the compounds of the present invention are also effective against parasites of the species Wuchereria, Brugia, Onchocerca and Loa from the family of Filariidae, which appear in the blood, in the tissue and in various organs, and also against Dracunculus and parasites of the species Strongyloides and Trichinella, which infect the gastrointestinal tract in particular.
- the compounds of formula I also have fungicidal and bactericidal activity.
- the good pesticidal activity of the compounds of formula I according to the invention corresponds to a mortality rate of at least 50-60% of the pests mentioned.
- the compounds of formula I are notable for the exceptionally long duration of efficacy.
- the compounds of formula I are preferably employed in unmodified form or preferably together with the adjuvants conventionally used in the art of formulation and may therefore be processed in a known manner to give, for example, emulsifiable concentrates, directly dilutable solutions, dilute emulsions, soluble powders, granules or microencapsulations in polymeric substances.
- the methods of application are selected in accordance with the intended objectives and the prevailing circumstances.
- the formulation i.e.
- the agents, preparations or compositions containing the active ingredient of formula I, or combinations of these active ingredients with other active ingredients, and optionally a solid or liquid adjuvant are produced in a manner known per se, for example by intimately mixing and/or grinding the active ingredients with spreading compositions, for example with solvents, solid carriers, and optionally surface-active compounds (surfactants).
- the solvents in question may be: alcohols, such as ethanol, propanol or butanol, and glycols and their ethers and esters, such as propylene glycol, dipropylene glycol ether, ethylene glycol, ethylene glycol monomethyl or -ethyl ether, ketones, such as cyclohexanone, isophorone or diacetanol alcohol, strong polar solvents, such as N-methyl-2-pyrrolidone, dimethyl sulfoxide or dimethylformamide, or water, vegetable oils, such as rape, castor, coconut, or soybean oil, and also, if appropriate, silicone oils.
- alcohols such as ethanol, propanol or butanol
- glycols and their ethers and esters such as propylene glycol, dipropylene glycol ether, ethylene glycol, ethylene glycol monomethyl or -ethyl ether, ketones, such as cyclohexanone, isophorone or di
- Preferred application forms for usage on warm-blooded animals in the control of helminths include solutions, emulsions, suspensions (drenches), food additives, powders, tablets including effervescent tablets, boli, capsules, micro-capsules and pour-on formulations, whereby the physiological compatibility of the formulation excipients must be taken into consideration.
- the binders for tablets and boli may be chemically modified polymeric natural substances that are soluble in water or in alcohol, such as starch, cellulose or protein derivatives (e.g. methyl cellulose, carboxymethyl cellulose, ethylhydroxyethyl cellulose, proteins such as zein, gelatin and the like), as well as synthetic polymers, such as polyvinyl alcohol, polyvinyl pyrrolidone etc.
- the tablets also contain fillers (e.g. starch, microcrystalline cellulose, sugar, lactose etc.), glidants and disintegrants.
- the carriers used are e.g. performance feeds, feed grain or protein concentrates.
- Such feed concentrates or compositions may contain, apart from the active ingredients, also additives, vitamins, antibiotics, chemotherapeutics or other pesticides, primarily bacteriostats, f ungistats, coccidiostats, or even hormone preparations, substances having anabolic action or substances which promote growth, which affect the quality of meat of animals for slaughter or which are beneficial to the organism in another way.
- the compositions or the active ingredients of formula I contained therein are added directly to feed or to the drinking troughs, then the formulated feed or drink contains the active ingredients preferably in a concentration of ca.
- the compounds of formula I according to the invention may be used alone or in combination with other biocides. They may be combined with pesticides having the same sphere of activity e.g. to increase activity, or with substances having another sphere of activity e.g. to broaden the range of activity. It can also be sensible to add so-called repellents. If the range of activity is to be extended to endoparasites, e.g. wormers, the compounds of formula I are suitably combined with substances having endoparasitic properties. Of course, they can also be used in combination with antibacterial compositions. Since the compounds of formula I are adulticides, i.e.
- Suitable partners in the mixture may be biocides, e.g. the insecticides and acaricides with a varying mechanism of activity, which are named in the following and have been known to the person skilled in the art for a long time, e.g. chitin synthesis inhibitors, growth regulators; active ingredients which act as juvenile hormones; active ingredients which act as adulticides; broad-band insecticides, broad-band acaricides and nematicides; and also the well known anthelminthics and insect- and/or acarid-deterring substances, said repellents or detachers.
- Non-limitative examples of suitable insecticides and acaricides are:
- Acrinathrin 13 Azinphos M 22. BPMC
- Amitraz 18. Benfuracarb 27. Butocarboxin 28. Butylpyridaben 61. Edifenphos 94. Heptenophos
- Non-limitative examples of suitable anthelminthics are named in the following, a few representatives have insecticidal and acaricidal activity in addition to the anthelminthic activity, and are partly already in the above list.
- Non-limitative examples of suitable repellents and detachers are:
- LV a preparation which contains insect-active fungi, preferably Verticillium lecanii, from The Pesticide Manual, 11 ,h Ed. (1997), The British Crop Protection Council, London, page 1266; Beauveria brogniartii, from The Pesticide Manual, 11 ,h Ed. (1997), The British Crop Protection Council, London, page 85 and Beauveria bassiana, from The Pesticide Manual, 11 th Ed. (1997), The British Crop Protection Council, London, page 83;
- LPI a preparation which contains insect-active viruses, preferably Neodipridon Sertifer NPV, from The Pesticide Manual, 11 th Ed. (1997), The British Crop Protection Council, London, page 1342; Mamestra brassicae NPV, from The Pesticide Manual, 11 th Ed. (1997), The British Crop Protection Council, London, page 759 and Cydia pomonella granulosis virus, from The Pesticide Manual, 11 h Ed. (1997), The British Crop Protection Council, London, page 291 ;
- insect-active viruses preferably Neodipridon Sertifer NPV
- Neodipridon Sertifer NPV from The Pesticide Manual, 11 th Ed.
- Mamestra brassicae NPV from The Pesticide Manual, 11 th Ed.
- Cydia pomonella granulosis virus from The Pesticide Manual, 11 h Ed. (1997), The British Crop Protection Council, London, page 291 ;
- a further essential aspect of the present invention relates to combination preparations for the control of parasites on warm-blooded animals, characterised in that they contain, in addition to a compound of formula I, at least one further active ingredient having the same or different sphere of activity and at least one physiologically acceptable carrier.
- the present invention is not restricted to two-fold combinations.
- the anthelminthic compositions according to the invention contain 0.1 to 99 % by weight, especially 0.1 to 95 % by weight of active ingredient of formula I, la or mixtures thereof, 99.9 to 1 % by weight, especially 99.8 to 5 % by weight of a solid or liquid admixture, including 0 to 25 % by weight, especially 0.1 to 25 % by weight of a surfactant.
- compositions according to the invention may take place topically, perorally, parenterally or subcutaneously, the composition being present in the form of solutions, emulsions, suspensions, (drenches), powders, tablets, boli, capsules and pour-on formulations.
- the pour-on or spot-on method consists in applying the compound of formula I to a specific location of the skin or coat, advantageously to the neck or backbone of the animal. This takes place e.g. by applying a swab or spray of the pour-on or spot-on formulation to a relatively small area of the coat, from where the active substance is dispersed almost automatically over wide areas of the fur owing to the spreading nature of the components in the formulation and assisted by the animal's movements.
- Pour-on or spot-on formulations suitably contain carriers, which promote rapid dispersement over the skin surface or in the coat of the host animal, and are generally regarded as spreading oils.
- Suitable carriers are e.g. oily solutions; alcoholic and isopropanolic solutions such as solutions of 2-octyldodecanol or oleyl alcohol; solutions in esters of monocarboxylic acids, such as isopropyl myristate, isopropyl palmitate, lauric acid oxalate, oleic acid oleyl ester, oleic acid decyl ester, hexyl laurate, oleyl oleate, decyl oleate, capric acid esters of saturated fat alcohols of chain length C 12 -C 18 ; solutions of esters of dicarboxylic acids, such as dibutyl phthalate, diisopropyl isophthalate, adipic acid diisopropyl ester, di-n
- glycols may be advantageous for a dispersing agent to be additionally present, such as one known from the pharmaceutical or cosmetic industry.
- a dispersing agent such as one known from the pharmaceutical or cosmetic industry. Examples are 2-pyrrolidone, 2-(N-alkyl)pyrrolidone, acetone, polyethylene glycol and the ethers and esters thereof, propylene glycol or synthetic triglycerides.
- the oily solutions include e.g. vegetable oils such as olive oil, groundnut oil, sesame oil, pine oil, linseed oil or castor oil.
- the vegetable oils may also be present in epoxidised form. Paraffins and silicone oils may also be used.
- a pour-on or spot-on formulation generally contains 1 to 20 % by weight of a compound of formula I, 0.1 to 50 % by weight of dispersing agent and 45 to 98.9 % by weight of solvent.
- the pour-on or spot-on method is especially advantageous for use on herd animals such as cattle, horses, sheep or pigs, in which it is difficult or time-consuming to treat all the animals orally or by injection. Because of its simplicity, this method can of course also be used for all other animals, including individual domestic animals or pets, and is greatly favoured by the keepers of the animals, as it can often be carried out without the specialist presence of the veterinarian.
- compositions may also contain further additives, such as stabilisers, anti-foaming agents, viscosity regulators, binding agents or tackifiers, as well as other active ingredients, in order to achieve special effects.
- further additives such as stabilisers, anti-foaming agents, viscosity regulators, binding agents or tackifiers, as well as other active ingredients, in order to achieve special effects.
- Anthelminthic compositions of this type which are used by the end user, similarly form a constituent of the present invention.
- the active ingredients of formula I can be used in all of their steric configurations or in mixtures thereof.
- the invention also includes a method of prophylactically protecting warm-blooded animals, especially productive livestock, domestic animals and pets, against parasitic helminths, which is characterised in that the active ingredients of the formula or the active ingredient formulations prepared therefrom are administered to the animals as an additive to the feed, or to the drinks or also in solid or liquid form, orally or by injection or parenterally.
- the invention also includes the compounds of formula I according to the invention for usage in one of the said processes.
- Granulate a) b) active ingredient 5 % 10 % kaolin 94 % highly dispersed silicic acid 1 % attapulgite - 90 %
- the active ingredient is dissolved in methylene chloride, sprayed onto the carrier and the solvent subsequently concentrated by evaporation under vacuum. Granulates of this kind can be mixed with the animal feed.
- the finely ground active ingredient is evenly applied in a mixer to the kaolin which has been moistened with polyethylene glycol. In this way, dust-free coated granules are obtained.
- Methyl cellulose is stirred into water. After the material has swollen, silicic acid is stirred in and the mixture homogeneously suspended. The active ingredient and the corn starch are mixed. The aqueous suspension is worked into this mixture and kneaded to a dough. The resulting mass is granulated through a 12 M sieve and dried.
- active ingredient 0.1-1.0 g groundnut oil ad 100 ml
- active ingredient 0.1-1.0 g sesame oil ad 100 ml
- active ingredient 0.1-1.0 g polyethoxylated castor oil (40 ethylene oxide units) 10 g 1 ,2-propanediol 20 g benzyl alcohol i g aqua ad inject. ad 100 ml
- active ingredient 0.1-1.0 g polyethoxylated sorbitan monooleate (20 ethylene oxide units) 8 g 4-hydroxymethyl-1 ,3-dioxolane (glycerol formal) 20 g benzyl alcohol i g aqua ad inject. ad 100 ml
- Preparation The active ingredient is dissolved in the solvents and the surfactant, and made up with water to the desired volume. Sterile filtration through an appropriate membrane filter of 0.22 mm pore size.
- the aqueous systems may also preferably be used for oral and/or intraruminal application.
- compositions may also contain further additives, such as stabilisers, e.g. where appropriate epoxidised vegetable oils (epoxidised coconut oil, rapeseed oil, or soybean oil); antifoams, e.g. silicone oil, preservatives, viscosity regulators, binders, tackifiers, as well as fertilisers or other active ingredients to achieve special effects.
- stabilisers e.g. where appropriate epoxidised vegetable oils (epoxidised coconut oil, rapeseed oil, or soybean oil); antifoams, e.g. silicone oil, preservatives, viscosity regulators, binders, tackifiers, as well as fertilisers or other active ingredients to achieve special effects.
- stabilisers e.g. where appropriate epoxidised vegetable oils (epoxidised coconut oil, rapeseed oil, or soybean oil); antifoams, e.g. silicone oil, preservatives, viscosity regulators, bind
- Example 1 N-M -cvano-1 -methyl-2-(5-chlorobenzotriazol-1 -vD-ethylH-trif luoromethoxy- benzamide a) A mixture of 5 g of 5-chlorobenzotriazole, 1.4 g of chloroacetone, 5.1 g of potassium carbonate and 0.5 g of potassium iodide is stirred into 50 ml of acetone at room temperature for 48 h. The mixture is subsequently filtered, the filtrate concentrated by evaporation in a vacuum, and the residue purified by flash chromatography.
- reaction mixture is subsequently concentrated by evaporation in a vacuum, dissolved again in ethyl acetate, and washed with water and saturated sodium chloride solution.
- organic phase is separated, dried with magnesium sulphate and concentrated by evaporation.
- 2-amino-3- (5-chlorobenzotriazol-1-yl)-2-methylpropionitrile is thus obtained.
- the substances named in the following table may also be prepared analogously to the above-described method.
- the values of the melting points are indicated in °C. Bd. signifies a direct bond.
- test methods may be used.
- a piece of sticky tape is attached horizontally to a PVC sheet, so that 10 fully engorged female ticks of Boophilus microplus (Biarra strain) can be adhered thereto by their backs, side by side, in a row.
- 1 ⁇ l of a liquid is injected into each tick.
- the liquid is a 1:1 mixture of polyethylene glycol and acetone and it contains, dissolved therein, a certain amount of active ingredient chosen from 1 , 0.1 or 0.01 ⁇ g per tick.
- Control animals are given an injection without active ingredient. After treatment, the animals are kept under normal conditions in an insectarium at ca. 28°C and at 80% relative humidity until oviposition takes place and the larvae have hatched from the eggs of the control animals.
- 4x10 engorged female ticks of the OP-resistant BIARRA strain are adhered to a sticky strip and covered for 1 hour with a cotton-wool ball soaked in an emulsion or suspension of the test compound in concentrations of 500, 125, 31 and 8 ppm respectively. Evaluation takes place 28 days later based on mortality, oviposition and hatched larvae.
- test tubes After immersion for 10 minutes, and shaking for 2x10 seconds on a vortex mixer, the test tubes are blocked up with a tight wad of cotton wool and rotated. As soon as all the liquid has been soaked up by the cotton wool ball, it is pushed half-way into the test tube which is still being rotated, so that most of the liquid is squeezed out of the cotton-wool ball and flows into a Petri dish below.
- test tubes are then kept at room temperature in a room with daylight until evaluated. After 14 days, the test tubes are immersed in a beaker of boiling water. If the ticks begin to move in reaction to the heat, the test substance is inactive at the tested concentration, otherwise the ticks are regarded as dead and the test substances regarded as active at the tested concentration. All substances are tested in a concentration range of 0.1 to 100 ppm.
- a sugar cube is treated with a solution of the test substance in such a way that the concentration of test substance in the sugar, after drying over night, is 250 ppm.
- the cube treated in this way is placed on an aluminium dish with wet cotton wool and 10 adult Musca domestica of an OP-resistant strain, covered with a beaker and incubated at 25°C. The mortality rate is determined after 24 hours.
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002496112A CA2496112A1 (en) | 2002-09-11 | 2003-09-10 | Benzotriazol-1-yl-amnioacetonitrile compounds and their use in the control of parasite disease |
| EP03795004A EP1539719B1 (en) | 2002-09-11 | 2003-09-10 | Benzotriazol-1-ylamino-acetonitrile compounds and the use thereof in pest control |
| JP2004535473A JP2006508921A (en) | 2002-09-11 | 2003-09-10 | Benzotriazol-1-yl-aminoacetonitrile compounds and their use in the control of parasitic diseases |
| AU2003264288A AU2003264288B2 (en) | 2002-09-11 | 2003-09-10 | Organic compounds |
| MXPA05002707A MXPA05002707A (en) | 2002-09-11 | 2003-09-10 | Organic compounds. |
| DE60315673T DE60315673T2 (en) | 2002-09-11 | 2003-09-10 | BENZOTRIAZOL-1-YLAMINOACETONITRILE COMPOUNDS AND THEIR USE FOR PEST CONTROL |
| BR0314220-5A BR0314220A (en) | 2002-09-11 | 2003-09-10 | Benzotriazol-1-yl-aminoacetonitrile compounds and their use in controlling parasitic disease |
| US10/526,524 US7084280B2 (en) | 2002-09-11 | 2003-09-10 | Benzotriazol-1-yl-aminoacetonitrile compounds and their use in the control of parasite disease |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CH1542/02 | 2002-09-11 | ||
| CH15422002 | 2002-09-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004024704A1 true WO2004024704A1 (en) | 2004-03-25 |
Family
ID=31983662
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2003/010047 WO2004024704A1 (en) | 2002-09-11 | 2003-09-10 | Organic compounds |
Country Status (15)
| Country | Link |
|---|---|
| US (1) | US7084280B2 (en) |
| EP (1) | EP1539719B1 (en) |
| JP (1) | JP2006508921A (en) |
| KR (1) | KR20050042802A (en) |
| CN (1) | CN1692104A (en) |
| AR (1) | AR042420A1 (en) |
| AT (1) | ATE370128T1 (en) |
| AU (1) | AU2003264288B2 (en) |
| BR (1) | BR0314220A (en) |
| CA (1) | CA2496112A1 (en) |
| DE (1) | DE60315673T2 (en) |
| MX (1) | MXPA05002707A (en) |
| RU (1) | RU2005110918A (en) |
| TW (1) | TW200409760A (en) |
| WO (1) | WO2004024704A1 (en) |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008144275A1 (en) | 2007-05-15 | 2008-11-27 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| WO2010048191A1 (en) * | 2008-10-21 | 2010-04-29 | Merial Limited | Thioamide compounds, method of making and method of using thereof |
| WO2010056999A1 (en) | 2008-11-14 | 2010-05-20 | Merial Limited | Enantiomerically enriched aryloazol- 2 -yl cyanoethylamino paraciticidal compounds |
| WO2010065852A1 (en) | 2008-12-04 | 2010-06-10 | Merial Limited | Dimeric avermectin and milbemycin derivatives |
| WO2011069143A1 (en) | 2009-12-04 | 2011-06-09 | Merial Limited | Pesticidal bis-organosulfur compounds |
| WO2011075591A1 (en) | 2009-12-17 | 2011-06-23 | Merial Limited | Anti parasitic dihydroazole compounds and compositions comprising same |
| WO2011123773A1 (en) | 2010-04-02 | 2011-10-06 | Merial Limited | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
| WO2012068202A1 (en) | 2010-11-16 | 2012-05-24 | Merial Limited | Novel monensin derivatives for the treatment and prevention of protozoal infections |
| WO2013003168A1 (en) | 2011-06-27 | 2013-01-03 | Merial Limited | Novel insect-repellent coumarin derivatives, syntheses, and methods of use |
| WO2013003505A1 (en) | 2011-06-27 | 2013-01-03 | Merial Limited | Amido-pyridyl ether compounds and compositions and their use against parasites |
| EP2550962A2 (en) | 2008-11-19 | 2013-01-30 | Merial Limited | Compositions comprising an aryl pyrazole and/or a formamidine, methods and uses thereof |
| WO2013039948A1 (en) | 2011-09-12 | 2013-03-21 | Merial Limited | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| WO2013044118A2 (en) | 2011-09-23 | 2013-03-28 | Merial Limited | Indirect modeling of new repellent molecules active against insects, acarids, and other arthropods |
| WO2013074892A1 (en) | 2011-11-17 | 2013-05-23 | Merial Limited | Compositions comprising an aryl pyrazole and a substituted imidazole, methods and uses thereof |
| WO2013082373A1 (en) | 2011-12-02 | 2013-06-06 | Merial Limited | Long-acting injectable moxidectin formulations and novel moxidectin crystal forms |
| WO2013158894A1 (en) | 2012-04-20 | 2013-10-24 | Merial Limited | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| WO2014081697A2 (en) | 2012-11-20 | 2014-05-30 | Merial Limited | Anthelmintic compounds and compositions and method of using thereof |
| US8822689B2 (en) | 2012-09-19 | 2014-09-02 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| WO2015066277A1 (en) | 2013-11-01 | 2015-05-07 | Merial Limited | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015161224A1 (en) | 2014-04-17 | 2015-10-22 | Merial, Inc. | Use of malononitrile compounds for protecting animals from parasites |
| WO2015179414A1 (en) | 2014-05-19 | 2015-11-26 | Merial, Inc. | Anthelmintic compounds |
| AU2013205750B2 (en) * | 2007-05-15 | 2015-12-03 | Aventis Agriculture | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| WO2015196014A1 (en) | 2014-06-19 | 2015-12-23 | Merial, Inc. | Parasiticidal compositions comprising indole derivatives, methods and uses thereof |
| US9237751B2 (en) | 2009-03-27 | 2016-01-19 | Norbrook Laboratories Limited | Topical parasiticide composition |
| WO2016069983A1 (en) | 2014-10-31 | 2016-05-06 | Merial, Inc. | Parasiticidal composition comprising fipronil |
| WO2016138339A1 (en) | 2015-02-26 | 2016-09-01 | Merial, Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2017147352A1 (en) | 2016-02-24 | 2017-08-31 | Merial, Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2018039508A1 (en) | 2016-08-25 | 2018-03-01 | Merial, Inc. | Method for reducing unwanted effects in parasiticidal treatments |
| WO2018071535A1 (en) | 2016-10-14 | 2018-04-19 | Merial, Inc. | Pesticidal and parasiticidal vinyl isoxazoline compounds |
| WO2019036407A1 (en) | 2017-08-14 | 2019-02-21 | Merial, Inc. | Pesticidal and parasiticidal pyrazole-isoxazoline compounds |
| WO2019157241A1 (en) | 2018-02-08 | 2019-08-15 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising eprinomectin and praziquantel, methods and uses thereof |
| WO2020112374A1 (en) | 2018-11-20 | 2020-06-04 | Boehringer Ingelheim Animal Health USA Inc. | Indazolylcyanoethylamino compound, compositions of same, method of making, and methods of using thereof |
| WO2020180635A1 (en) | 2019-03-01 | 2020-09-10 | Boehringer Ingelheim Animal Health USA Inc. | Injectable clorsulon compositions, methods and uses thereof |
| WO2022140728A1 (en) | 2020-12-21 | 2022-06-30 | Boehringer Ingelheim Animam Health Usa Inc. | Parasiticidal collar comprising isoxazoline compounds |
| WO2023156938A1 (en) | 2022-02-17 | 2023-08-24 | Boehringer Ingelheim Vetmedica Gmbh | Method and system for providing a fluid product mailer |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW200406386A (en) * | 2002-06-07 | 2004-05-01 | Novartis Ag | Organic compounds |
| GB0402677D0 (en) * | 2003-11-06 | 2004-03-10 | Novartis Ag | Organic compounds |
| WO2005051451A2 (en) * | 2003-11-20 | 2005-06-09 | Angiotech International Ag | Electrical devices and anti-scarring agents |
| US8486987B2 (en) | 2007-11-16 | 2013-07-16 | University Of Medicine And Dentistry Of New Jersey | Mechanism-based small-molecule parasite inhibitors |
| RU2660346C1 (en) | 2012-02-06 | 2018-07-05 | Мериал, Инк. | Parasiticidal oral veterinary compositions comprising systemically active agents, methods and use of these compositions and methods |
| JO3626B1 (en) | 2012-02-23 | 2020-08-27 | Merial Inc | Topical compositions comprising fipronil and permethrin and methods of use |
| CN107835818B (en) | 2015-05-20 | 2022-04-29 | 勃林格殷格翰动物保健美国公司 | Anthelmintic depsipeptide compounds |
| CN110167921A (en) | 2016-11-16 | 2019-08-23 | 勃林格殷格翰动物保健美国公司 | Dehelminthization depsipeptide compound |
| AU2019301510A1 (en) | 2018-07-09 | 2021-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Anthelminthic heterocyclic compounds |
| EP3941587A1 (en) | 2019-03-19 | 2022-01-26 | Boehringer Ingelheim Animal Health USA Inc. | Anthelmintic aza-benzothiophene and aza-benzofuran compounds |
| CA3183100A1 (en) | 2020-05-29 | 2021-12-02 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Anthelmintic heterocyclic compounds |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04112872A (en) * | 1990-09-03 | 1992-04-14 | Nippon Kayaku Co Ltd | Plant phycomycete disease control agent containing isonicotinic acid derivative as active ingredient |
| EP0953365A2 (en) * | 1998-05-01 | 1999-11-03 | Kawasumi Laboratories, Inc. | Female connector |
| WO2000073290A1 (en) * | 1999-05-28 | 2000-12-07 | Nihon Bayer Agrochem K.K. | Isothiazolecarboxamides and their use as microbicides |
| WO2002049641A2 (en) * | 2000-12-20 | 2002-06-27 | Novartis Ag | Anti-endoparasitic amidoacetonitrils |
| WO2003042184A1 (en) * | 2001-11-14 | 2003-05-22 | Novartis Ag | Benzimidazol- or indol-aminoacetonitrile derivatives for parasite control |
-
2003
- 2003-09-09 TW TW092124863A patent/TW200409760A/en unknown
- 2003-09-09 AR ARP030103262A patent/AR042420A1/en unknown
- 2003-09-10 AU AU2003264288A patent/AU2003264288B2/en not_active Ceased
- 2003-09-10 KR KR1020057004092A patent/KR20050042802A/en not_active Application Discontinuation
- 2003-09-10 BR BR0314220-5A patent/BR0314220A/en not_active Application Discontinuation
- 2003-09-10 AT AT03795004T patent/ATE370128T1/en not_active IP Right Cessation
- 2003-09-10 WO PCT/EP2003/010047 patent/WO2004024704A1/en active IP Right Grant
- 2003-09-10 RU RU2005110918/04A patent/RU2005110918A/en not_active Application Discontinuation
- 2003-09-10 US US10/526,524 patent/US7084280B2/en not_active Expired - Fee Related
- 2003-09-10 DE DE60315673T patent/DE60315673T2/en not_active Expired - Fee Related
- 2003-09-10 EP EP03795004A patent/EP1539719B1/en not_active Expired - Lifetime
- 2003-09-10 MX MXPA05002707A patent/MXPA05002707A/en unknown
- 2003-09-10 JP JP2004535473A patent/JP2006508921A/en not_active Withdrawn
- 2003-09-10 CA CA002496112A patent/CA2496112A1/en not_active Abandoned
- 2003-09-10 CN CNA038215322A patent/CN1692104A/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04112872A (en) * | 1990-09-03 | 1992-04-14 | Nippon Kayaku Co Ltd | Plant phycomycete disease control agent containing isonicotinic acid derivative as active ingredient |
| EP0953365A2 (en) * | 1998-05-01 | 1999-11-03 | Kawasumi Laboratories, Inc. | Female connector |
| WO2000073290A1 (en) * | 1999-05-28 | 2000-12-07 | Nihon Bayer Agrochem K.K. | Isothiazolecarboxamides and their use as microbicides |
| WO2002049641A2 (en) * | 2000-12-20 | 2002-06-27 | Novartis Ag | Anti-endoparasitic amidoacetonitrils |
| WO2003042184A1 (en) * | 2001-11-14 | 2003-05-22 | Novartis Ag | Benzimidazol- or indol-aminoacetonitrile derivatives for parasite control |
Non-Patent Citations (1)
| Title |
|---|
| PATENT ABSTRACTS OF JAPAN vol. 016, no. 358 (C - 0970) 4 August 1992 (1992-08-04) * |
Cited By (70)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8283475B2 (en) | 2007-05-15 | 2012-10-09 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| KR101472649B1 (en) | 2007-05-15 | 2014-12-16 | 메리얼 리미티드 | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| EP2639228A3 (en) * | 2007-05-15 | 2013-09-25 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| EP2639228A2 (en) | 2007-05-15 | 2013-09-18 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| EP3428148A1 (en) | 2007-05-15 | 2019-01-16 | Merial, Inc. | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| KR101292600B1 (en) | 2007-05-15 | 2013-08-05 | 아벤티스 애그리컬쳐 | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| KR101546639B1 (en) | 2007-05-15 | 2015-08-21 | 메리얼 리미티드 | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| AU2013205750B2 (en) * | 2007-05-15 | 2015-12-03 | Aventis Agriculture | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| AU2008254279B2 (en) * | 2007-05-15 | 2013-03-14 | Aventis Agriculture | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| US8088801B2 (en) | 2007-05-15 | 2012-01-03 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| WO2008144275A1 (en) | 2007-05-15 | 2008-11-27 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| EP3088384A1 (en) | 2007-05-15 | 2016-11-02 | Merial, Inc. | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| EA016912B1 (en) * | 2007-05-15 | 2012-08-30 | Мериал Лимитед | Aryloazol-2-ylcyanoethylamino compounds, method of making and method of using same |
| KR101782308B1 (en) | 2008-10-21 | 2017-09-27 | 메리얼 인코포레이티드 | Thioamide compounds, method of making and method of using thereof |
| US8314146B2 (en) | 2008-10-21 | 2012-11-20 | Merial Limited | Thioamide compounds, method of making and method of using thereof |
| EA026649B1 (en) * | 2008-10-21 | 2017-05-31 | Мериал, Инк. | Thioamide compounds, method of making and method of using thereof |
| WO2010048191A1 (en) * | 2008-10-21 | 2010-04-29 | Merial Limited | Thioamide compounds, method of making and method of using thereof |
| US7964621B2 (en) | 2008-10-21 | 2011-06-21 | Merial Limited | Thioamide compounds, method of making and method of using thereof |
| AU2009307736B2 (en) * | 2008-10-21 | 2015-02-05 | Boehringer Ingelheim Animal Health USA Inc. | Thioamide compounds, method of making and method of using thereof |
| KR101782311B1 (en) | 2008-10-21 | 2017-09-27 | 메리얼 인코포레이티드 | Thioamide compounds, method of making and method of using thereof |
| AU2009313920B2 (en) * | 2008-11-14 | 2014-10-23 | Boehringer Ingelheim Animal Health USA Inc. | Enantiomerically enriched aryloazol- 2 -yl cyanoethylamino paraciticidal compounds |
| EP3050874A1 (en) * | 2008-11-14 | 2016-08-03 | Merial, Inc. | Enantiomerially enriched aryloazol-2-yl cyanoethylamino paraciticidal compounds |
| CN102272105A (en) * | 2008-11-14 | 2011-12-07 | 梅里亚有限公司 | Enantiomerically enriched aryloazol- 2 -yl cyanoethylamino parasiticidal compounds |
| US8461176B2 (en) | 2008-11-14 | 2013-06-11 | Merial Limited | Enantiomerically enriched aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| JP2012508767A (en) * | 2008-11-14 | 2012-04-12 | メリアル リミテッド | Enantiomerically enriched aryloazol-2-yl-cyanoethylamino parasiticidal compounds |
| EP3498696A1 (en) | 2008-11-14 | 2019-06-19 | Merial, Inc. | Enantiomerically enriched aryloazol-2-yl cyanoethylamino parasiticidal compounds |
| WO2010056999A1 (en) | 2008-11-14 | 2010-05-20 | Merial Limited | Enantiomerically enriched aryloazol- 2 -yl cyanoethylamino paraciticidal compounds |
| CN102272105B (en) * | 2008-11-14 | 2016-09-14 | 梅瑞尔公司 | The aryl of enantiomeric enrichment azoles-2-base cyano group ethylamino Parasiticidal compound |
| EA019606B1 (en) * | 2008-11-14 | 2014-04-30 | Мериал Лимитед | Aryloazol-2-ylcyanoethylamino paraciticidal compounds enriched in an enantiomer |
| EP2550962A2 (en) | 2008-11-19 | 2013-01-30 | Merial Limited | Compositions comprising an aryl pyrazole and/or a formamidine, methods and uses thereof |
| WO2010065852A1 (en) | 2008-12-04 | 2010-06-10 | Merial Limited | Dimeric avermectin and milbemycin derivatives |
| US9237751B2 (en) | 2009-03-27 | 2016-01-19 | Norbrook Laboratories Limited | Topical parasiticide composition |
| WO2011069143A1 (en) | 2009-12-04 | 2011-06-09 | Merial Limited | Pesticidal bis-organosulfur compounds |
| WO2011075591A1 (en) | 2009-12-17 | 2011-06-23 | Merial Limited | Anti parasitic dihydroazole compounds and compositions comprising same |
| EP3560923A1 (en) | 2009-12-17 | 2019-10-30 | Boehringer Ingelheim Animal Health USA Inc. | Anti parasitic dihydroazole compounds and compositions comprising same |
| EP3078664A1 (en) | 2009-12-17 | 2016-10-12 | Merial Inc. | Antiparasitic dihydroazole compositions |
| WO2011123773A1 (en) | 2010-04-02 | 2011-10-06 | Merial Limited | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
| WO2012068202A1 (en) | 2010-11-16 | 2012-05-24 | Merial Limited | Novel monensin derivatives for the treatment and prevention of protozoal infections |
| WO2013003505A1 (en) | 2011-06-27 | 2013-01-03 | Merial Limited | Amido-pyridyl ether compounds and compositions and their use against parasites |
| WO2013003168A1 (en) | 2011-06-27 | 2013-01-03 | Merial Limited | Novel insect-repellent coumarin derivatives, syntheses, and methods of use |
| EP3788874A1 (en) | 2011-09-12 | 2021-03-10 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising an isoxazoline active agent, method and uses thereof |
| WO2013039948A1 (en) | 2011-09-12 | 2013-03-21 | Merial Limited | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| WO2013044118A2 (en) | 2011-09-23 | 2013-03-28 | Merial Limited | Indirect modeling of new repellent molecules active against insects, acarids, and other arthropods |
| WO2013074892A1 (en) | 2011-11-17 | 2013-05-23 | Merial Limited | Compositions comprising an aryl pyrazole and a substituted imidazole, methods and uses thereof |
| WO2013082373A1 (en) | 2011-12-02 | 2013-06-06 | Merial Limited | Long-acting injectable moxidectin formulations and novel moxidectin crystal forms |
| EP3351546A1 (en) | 2011-12-02 | 2018-07-25 | Merial, Inc. | Long-acting injectable moxidectin formulations |
| EP3763706A1 (en) | 2012-04-20 | 2021-01-13 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| WO2013158894A1 (en) | 2012-04-20 | 2013-10-24 | Merial Limited | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| EP3453702A2 (en) | 2012-04-20 | 2019-03-13 | Merial, Inc. | Parasiticidal compositions comprising benzimidazole derivatives, methods and uses thereof |
| US8822689B2 (en) | 2012-09-19 | 2014-09-02 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| EP3428162A1 (en) | 2012-11-20 | 2019-01-16 | Merial Inc. | Anthelmintic compounds and compositions and method of using thereof |
| WO2014081697A2 (en) | 2012-11-20 | 2014-05-30 | Merial Limited | Anthelmintic compounds and compositions and method of using thereof |
| WO2015066277A1 (en) | 2013-11-01 | 2015-05-07 | Merial Limited | Antiparisitic and pesticidal isoxazoline compounds |
| EP3733664A1 (en) | 2013-11-01 | 2020-11-04 | Boehringer Ingelheim Animal Health USA Inc. | Antiparisitic and pesticidal isoxazoline compounds |
| WO2015161224A1 (en) | 2014-04-17 | 2015-10-22 | Merial, Inc. | Use of malononitrile compounds for protecting animals from parasites |
| WO2015179414A1 (en) | 2014-05-19 | 2015-11-26 | Merial, Inc. | Anthelmintic compounds |
| EP3517524A1 (en) | 2014-06-19 | 2019-07-31 | Merial Inc. | Parasiticidal compositions comprising indole derivatives, methods and uses thereof |
| WO2015196014A1 (en) | 2014-06-19 | 2015-12-23 | Merial, Inc. | Parasiticidal compositions comprising indole derivatives, methods and uses thereof |
| WO2016069983A1 (en) | 2014-10-31 | 2016-05-06 | Merial, Inc. | Parasiticidal composition comprising fipronil |
| WO2016138339A1 (en) | 2015-02-26 | 2016-09-01 | Merial, Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| EP3763211A1 (en) | 2016-02-24 | 2021-01-13 | Boehringer Ingelheim Animal Health USA Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2017147352A1 (en) | 2016-02-24 | 2017-08-31 | Merial, Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2018039508A1 (en) | 2016-08-25 | 2018-03-01 | Merial, Inc. | Method for reducing unwanted effects in parasiticidal treatments |
| WO2018071535A1 (en) | 2016-10-14 | 2018-04-19 | Merial, Inc. | Pesticidal and parasiticidal vinyl isoxazoline compounds |
| WO2019036407A1 (en) | 2017-08-14 | 2019-02-21 | Merial, Inc. | Pesticidal and parasiticidal pyrazole-isoxazoline compounds |
| WO2019157241A1 (en) | 2018-02-08 | 2019-08-15 | Boehringer Ingelheim Animal Health USA Inc. | Parasiticidal compositions comprising eprinomectin and praziquantel, methods and uses thereof |
| WO2020112374A1 (en) | 2018-11-20 | 2020-06-04 | Boehringer Ingelheim Animal Health USA Inc. | Indazolylcyanoethylamino compound, compositions of same, method of making, and methods of using thereof |
| WO2020180635A1 (en) | 2019-03-01 | 2020-09-10 | Boehringer Ingelheim Animal Health USA Inc. | Injectable clorsulon compositions, methods and uses thereof |
| WO2022140728A1 (en) | 2020-12-21 | 2022-06-30 | Boehringer Ingelheim Animam Health Usa Inc. | Parasiticidal collar comprising isoxazoline compounds |
| WO2023156938A1 (en) | 2022-02-17 | 2023-08-24 | Boehringer Ingelheim Vetmedica Gmbh | Method and system for providing a fluid product mailer |
Also Published As
| Publication number | Publication date |
|---|---|
| US7084280B2 (en) | 2006-08-01 |
| DE60315673D1 (en) | 2007-09-27 |
| CN1692104A (en) | 2005-11-02 |
| KR20050042802A (en) | 2005-05-10 |
| EP1539719B1 (en) | 2007-08-15 |
| RU2005110918A (en) | 2006-04-10 |
| AR042420A1 (en) | 2005-06-22 |
| TW200409760A (en) | 2004-06-16 |
| DE60315673T2 (en) | 2008-06-05 |
| AU2003264288B2 (en) | 2007-07-12 |
| CA2496112A1 (en) | 2004-03-25 |
| MXPA05002707A (en) | 2005-05-05 |
| JP2006508921A (en) | 2006-03-16 |
| BR0314220A (en) | 2005-06-28 |
| AU2003264288A1 (en) | 2004-04-30 |
| ATE370128T1 (en) | 2007-09-15 |
| US20050272935A1 (en) | 2005-12-08 |
| EP1539719A1 (en) | 2005-06-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7084280B2 (en) | Benzotriazol-1-yl-aminoacetonitrile compounds and their use in the control of parasite disease | |
| US7279495B2 (en) | Benzimidazol- or indol-aminoacetonitrile derivatives for parasite control | |
| AU2002351995A1 (en) | Benzimidazol-or indol-aminoacetonitrile derivatives for parasite control | |
| US20040242913A1 (en) | Organic compounds | |
| US7250436B2 (en) | Indazole-aminoacetonitrile derivatives having special pesticidal activity | |
| US7521476B2 (en) | Aminoacetonitrile derivatives suitable for controlling parasites | |
| US7148255B2 (en) | Amidoacetonitrile compounds | |
| AU2003250342B2 (en) | Amidoacetonitrile compounds and their use as pesticides | |
| US7705044B2 (en) | Benzamidoacetonitriles and their use as antiparasitics | |
| US7262209B2 (en) | Carbonyloxy-cyanomethyl compounds as antiparasitic agents | |
| AU2002349321A1 (en) | Carbonyloxy-cyanomethyl compounds as antiparasitic agents | |
| AU2002342791A1 (en) | Organic compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LT LU LV MA MD MK MN MX NI NO NZ OM PH PL PT RO RU SC SE SG SK TJ TM TN TR TT UA US UZ VC VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005/00769 Country of ref document: ZA Ref document number: 200500769 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003264288 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003795004 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2496112 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10526524 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 538677 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 356/CHENP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2005/002707 Country of ref document: MX Ref document number: 20038215322 Country of ref document: CN Ref document number: 1020057004092 Country of ref document: KR Ref document number: 2004535473 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 05025154 Country of ref document: CO |
|
| ENP | Entry into the national phase |
Ref document number: 2005110918 Country of ref document: RU Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020057004092 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003795004 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2003795004 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2003264288 Country of ref document: AU |