CONDENSED HETEROCYCLIC COMPOUNDS AS PDE-IV INHIBITORS FOR THE TREATMENT OF INFLAMMATORY AND ALLERGIC DISORDERS
Field of the Invention
10 The present invention relates to novel heterocyclic compounds. More particularly the present invention relates to novel phosphodiesterase type 4 (PDE4) inhibitors having a heterocyclic structure, pharmaceutical compositions including such compounds, methods for their preparation and method for their use.
15 Background of the Invention
Airway inflammation characterizes a number of severe lung diseases including asthma and chronic obstructive pulmonary disease (COPD). Events leading to airway obstruction include edema of airway walls, infiltration of inflammatory cells into the lung, production of various inflammatory mediators and increased mucous 20 production. The airways of asthmatic patients are infiltrated by inflammatory leukocytes, of which the eosinophil is the most prominent component. The magnitude of asthmatic reactions is correlated with the number of eosinophils present in lungs.
The accumulation of eosinophils is found dramatically in the lungs of asthmatic patients although there are very few in the lungs of a normal individual.
25 They are capable of lysing and activating cells and destroying tissues. When activated, they synthesize and release inflammatory cytokines such as IL-1, IL-3, TNF-α and inflammatory mediators such as PAF, LTD4 and related oxygen species that can produce edema, broncho-constriction. Tumor necrosis factor (TNF-α) was also known to be involved in the pathogenesis of a number of autoimmune and
30 inflammatory diseases. Consequently, manipulation of the cytokine signaling or biosynthetic pathways associated with these proteins may provide therapeutic benefit
in those disease states. It has been well demonstrated that TNF-α production in pro- inflammatory cells becomes attenuated by an elevation of intracellular cyclic adenosine 3',5'-monophosphate (cAMP). This second messenger is regulated by the phosphodiesterase (PDE) family of enzymes. The phosphodiesterase enzymes play an integral role in cell signaling mechanisms by hydrolyzing cAMP and cGP to their inactive 5' forms. Inhibition of PDE enzymes thus results in an elevation of cAMP and /or cGP levels and alters intracellular responses to extra cellular signals by affecting the processes mediated by cyclic nucleotides. Since eosinophilis are believed to be a critical proinflammatory target for asthma, identification of the expression of PDE 4 gene family in eosinophils led to the PDE 4 as potential therapeutic target for asthma [Rogers, D.F., Giembycz, M.A., Trends Pharmacol. Sci., 19, 160-164(1998); Barnes, P.J., Trends Pharmacol. Sci., 19, 415-423 (1998) herein incorporated by reference in their entirety].
The mammalian cyclic nucleotide phosphodiesterases (PDEs) are classified into ten families on the basis of their amino acid sequences and/or DNA sequence, substrate specificity and sensitivity to pharmacological agents [Soderling, S.H., Bayuga, S.J., and Beavo, J.A., Proc. Natl. Acad. Sci., USA, 96,7071-7076 (1999); Fujishige, K, Kotera, J., Michibata, H., Yuasa, K., Takebayashi, Si, Okamura, K. and Omori, K., J. Biol. Chem., 274, 18438-18445 (1999) herein incorporated by reference in their entirety]. Many cell types express more than one PDE and distribution of isoenzymes between the cells varies markedly. Therefore development of highly isoenzyme selective PDE inhibitors provides a unique opportunity for selective manipulation of various pathophysiological processes.
Phosphodiesterase type 4 (PDE4) is an enzyme which regulates activities in cells which lead to inflammation in the lungs. PDE4, a cAMP-specific and Ca+2- independent enzyme, is a key isozyme in the hydrolysis of cAMP in mast cells, basophils, eosinophils, monocytes and lymphocytes. The association between cAMP elevation in inflammatory cells with airway smooth muscle relaxation and inhibition of mediator release has led to widespread interest in the design of PDE4 inhibitors[Trophy,TJ., Am. J. Respir. Crit. Care Med., 157, 351-370 (1998) herein incorporated by reference in their entirety]. Excessive or unregulated TNF-α production has been implicated in mediating or exacerbating a number of undesirable
physiological conditions such as diseases including osteoarthritis, and other arthritic conditions; septic shock, ecdotoxic shock, respiratory distress syndrome, bone resorption diseases ; Since TNF-α also participates in the onset and progress of autoimmune diseases, PDE4 inhibitors may find utility as therapeutic agents for rheumatoid arthritis, multiple sclerosis and Crohn's disease. [Nature Medicine, I, 211- 214 (1995) and ibid., 244-248 herein incorporated by reference in their entirety].
Strong interest in Drugs capable of selective inhibition of PDE 4 is due to several factors such as (a) the tissue distribution of PDE-4 suggests that pathologies related to the central nervous and immune systems could be treated with selective PDE-4 inhibitors (b) the increase in intracellular cAMP concentration, the obvious biochemical consequence of PDE-4 inhibition, has been well characterized in immuno-competent cells where it acts as a deactivating signal.
Recently the PDE4 family has grown to include four subtypes - PDE4A to PDE4D, each encoded by a distinct gene (British Journal of Pharmacology; 1999; v.128; p.1393-1398), herein incorporated by reference in its entirety.
It has been demonstrated that increasing cAMP levels within these cells results in suppression of cell activation which in turn inhibits the production and release of pro-inflammatory cytokines such as TNF-c. Since eosinophilis are believed to be a critical pro-inflammatory target for asthma, identification of the expression of the PDE-4 gene family in eosinophils led to the PDE-4 as potential therapeutic target for asthma.
The usefulness of several PDE-4 inhibitors, unfortunately, is limited due to their undesirable side effect profile which include nausea and emesis (due to action on PDE-4 in the central nervous system) and gastric acid secretion due to action on PDE- 4 in parietal cells in the gut. Barnette, M.S., Grous, M., Cieslinsky, L.B., Burman, M., Christensen, S.B., Trophy, T J., J. Pharmacol. Exp. Ther., 273,1396-1402 (1995) herein incorporated by reference in their entirety. One of the earliest PDE-4 inhibitor, Rolipram, was withdrawn from the clinical development because of its severe unacceptable side effect profile. Zeller E. et. al., Pharmacopsychiatr., 17, 188-190 (1984) herein incoφorated by reference in their entirety. The cause of severe side
effects of several PDE-4 inhibitor molecules in human clinical trials has recently become apparent.
There exist two binding sites on mammalian PDE-4 at which inhibitor molecules may bind. Also PDE-4 exists in two distinct forms which represent different conformations. They are designated as High affinity Rolipram binding site PDE-4H and Low affinity Rolipram binding site PDE-4L [Jacobitz, S., McLaughlin, M.M., Livi, G.P., Burman, M., Trophy, TJ., Mol. Pharmaco., 50, 891-899 (1996) herein incoφorated by reference in their entirety]. It was shown that certain side effects (vomiting and gastric acid secretion) are associated with inhibition of PDE-4H whereas some beneficial actions are associated with PDE-4L inhibition. It was also found that human recombinant PDE-4 exists in 4 isoforms A, B, C and D [Muller, T., Engels, P., Fozard, J.R., Trends Pharmacol. Sci., 17, 294-298 (1996) herein incoφorated by reference in their entirety]. Accordingly, compounds displaying more PDE-4D isoenzyme selectivity over the A, B or C are found to have less amount of side effects than Rolipram [Hughes. B et.al., Br. J. Pharmacol. 1996, 118, 1183-1191 herein incoφorated by reference in their entirety]. Therefore, selective inhibitors of PDE-4 isozymes would have therapeutic effects in inflammatory diseases such as asthma and other respiratory diseases.
Although several research groups all over the world are working to find highly selective PDE-4 isozyme inhibitors, so far success is limited. Various compounds have shown PDE-4 inhibition.
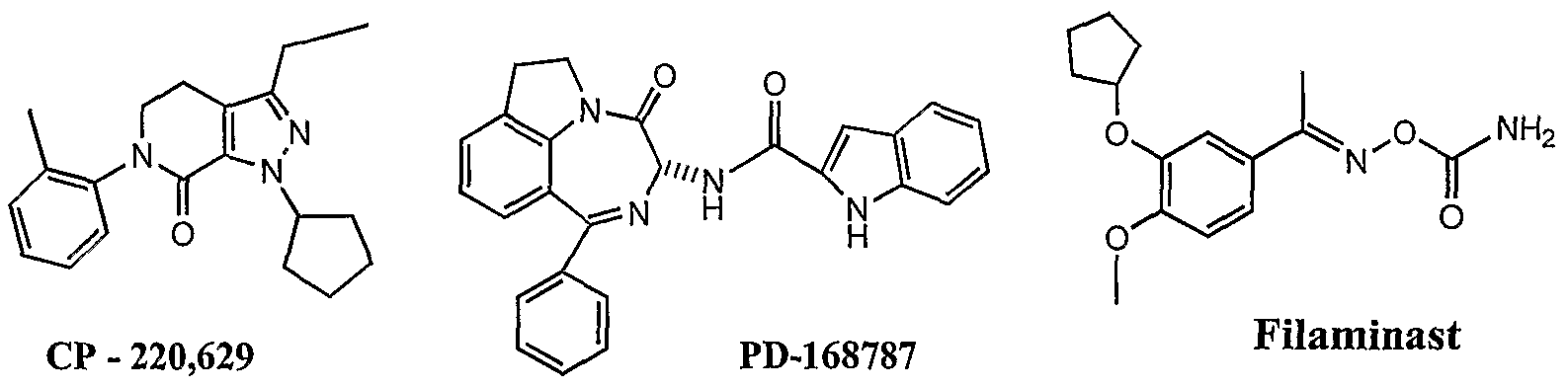
ARIFLQ A
Roflumilast D Bay-19-8004 E
SmithKline Beecham's "Ariflo" which has the formula A, Byk Gulden's Roflumilast which has the formula β and Bayer's Bay-19-8004 which has the formula E have reached advanced stage of human clinical trials. Other compounds which have shown potent PDE-4 inhibitory activity include Celltech's CDP-840 of the formula B, Schering Plough's D-4418 of the formula C, Pfizer' s 5CP-220,629 which has the formula F, Parke Davis's PD-168787 which has the formula G and Wyeth's Filaminast which has the formula H. However, recently due to efficacy and side effects problems, Ariflo, CDP-840 and Bay-19-8004 were discontinued from clinical trials as a treatment for asthma. Other compounds of the formulae C and F are presently undergoing phase- 1 clinical trials.
WO 9524381, herein incoφorated by reference in its entirety, describes the compounds represented by the general formula 1 which have potent PDE4 inhibition activity.
(i) wherein
Ri is OH or an ether or ester thereof, X is YR2, halogen, nitro, NR R5 or formyl amine;
Y is O or S(0)m, where m is 0,1 or 2;
R2 is methyl or ethyl where either methyl or ethyl optionally substituted by 1 or more halogens;
R
3 is hydrogen, halogen, C alkyl, CH
2NHC(0)C(0)NH
2) halo-substituted C alkyl,
optionally substituted by Rs
1, CN, OR
8, CH
20R
8,
NR8R10, CH2NR8Rιo, C^H, C(O)OR8, C(0)NR8Rιo, or -C s CRg1 ;
R4 and Rs are independently hydrogen or C 1-2 alkyl;
R7 is -(CR R5)qRι2 or C1-6 alkyl wherein R12 or Cι-6 alkyl group is optionally substituted one or more times by Cι-2 alkyl optionally substituted one to three groups selected from -F,-Br, -CI, -N02,-NRιORι ι,-C(=O)R8, -C(=0)OR8, OR8,-CN, -
C(=O)NR10Rn
-OC(=0)NRioRn, -OC(=O)R8, -NRιOC(=O)NRι0Rιι,NR10C(=O)OR9,
-NRιoS(=O)
2R
9, -S(0)
m 1R
9, -NRι
0C(=O)C(=O)NRι
0Rι i,
-NRιoC(=O)C(=O)Rιo, thiazolyl, imidazolyl, oxazolyl, pyrazolyl, triazolyl, or tetrazolyl;
R8 is -H or R9;
R8' is R8 or fluorine; Rg is C alkyl optionally substituted by one to three -F;
Rio is OR8, hydrogen, or C alkyl optionally substituted by one to three fluorines;
Ru is -H or C1-4 alkyl optionally substituted by one to three -F; or when Rio and Ru are as NRioRπ may together with the nitrogen from 5 to 7 membered ring optionally containing at least one additional heteroatom selected from 0,N or S; Rι2 is C3-7 cycloalkyl, 2-pyridyl, 3-pyridyl, 4-pyridyl, pyrimidyl, pyrazolyl,l- imidazolyl,
2-imidazolyl, thiazolyl, triazolyl, pyrrolyl, piperazinyl, piperidinyl, moφholinyl, furanyl, 2-thienyl, 3 -thienyl, 4-thiazolyl, quinolinyl, naphthyl or phenyl; Rι3 is heterocyclic ring selected from oxazolidinyl, oxazolyl, thiazolyl, pyrazolyl, triazolyl, tetrazolyl, imidazolyl, imidazolidinyl, thiazolidinyl, isoxazolyl, oxadiazolyl or thiadiazolyl, where R13 is appended to a compound of formula (I) through a carbon of heterocyclic ring ,and where each heterocyclic ring may be unsubstituted or substituted by one or two C1-2 alkyl groups;
Rι4 is H or R7 or when Rio and R1 are as NRιoRι4, they may together with the nitrogen atom form a 5 to 7 membered ring optionally containing at least one additional heteroatom selected from O, N or S; m 1 is 0,1 or 2; q is 0,1 or 2; Y1 is O or S Z is C(=YI)Ri4, C(=O)0Ri4, C(=Yl)NRι0Rι4jC(=NRιo)NRι0Rι4CN C(=NOR8)RH C(=0)NR8NR8C(=0)R8, C(=O)NR8NRιoRι4, -C(=NORι4)R8, C(=NR8)NR!0NRι4, C(=NRι4)NR8R8> C(=N-CN)NRιoRι4, C(=N-CN)SR9, 2-imidazolyl, 4-imidazolyl,5- imidazolyl, 3-pyrazolyl, 4-pyrazolyl 5-pyrazolyl,4-triazolyl[l,2,3], 5-triazolyl[l,2,3], 3-triazolyl[l,2,4], 5-triazolyl[l,2,4], 5-tetrazolyl,2-oxazolyl,4-oxazolyl, 5-oxazolyl,3- isoxazolyl, 4-isoxazolyl, 5-isoxazolyl,3-oxadiazolyl[l,2,4], 5-oxadiazolyl[l,2,4], 2- oxadiazolyl[l,3,4], 2-thiazolyl[l,3,4], 2-thiazolyl, 4-thiazolyl, 5-thiazolyl,2- oxazolidinyl, 4-oxazolidinyl, 5 -oxazolidinyl, 2-thiazolidinyl, 4-thiazolidinyl or 5- thiazolidinyl,2-imidazolidinyl, 4-imidazolidinyl, or 5-imidazolidinyl; wherein all of the heterocyclic ring systems may be optionally substituted one or more times by Rι ; Z'is O, NR9NOR8, NCN, C(-CN)2, CR8CN,CR8N02, CR8C(O)OR8, CR8C(0)NR8R8, C(-CN)NO2, C(-CN)C(O)OR9, or C(-CN)C(0)NR8R8;
US Patent. 4,012,495, herein incoφorated by reference in its entirety, describes compounds of formula 2, which relates to racemic and optically active 4- (polyalkoxyphenyl)-2-pyrrolidones of general formula 2, which have potent PDE-4 inhibition activity.
Wherein Ri and R2 each are alike or different and are hydrocarbon of up to 18 carbon atoms or alkyl of 1-5 carbon atoms which are substituted by one or more of halogen atoms, hydroxyl, carboxy, alkoxy, alkoxycarbonyl, carboxamide and amino or substituted amino or collectively are alkylene of 1-3 carbon atoms; R3 is a hydrogen atom or methoxy; R4 is a hydrogen atom, alkyl, aryl or acyl; X is an oxygen atom or a sulfur atom.
US Patent 5,128,358, herein incoφorated by reference in its entirety, describes the compounds are represented by formula 3 ,
Wherein R is a polycycloalkyl group having from 7 to 11 carbon atoms; R is methyl or ethyl; X is O or NH; and
Y comprises a 5- or 6- membered heterocyclic ring, especially a saturated or unsaturated 5- or 6- membered heterocyclic ring having one or two nitrogen atoms, said ring being optionally substituted with =0 or =S, provided that when said optional group is present and the heterocyclic ring comprises one nitrogen atom the optional group is located on a carbon atom adjacent to the said nitrogen atom ,and when the heterocyclic ring comprises two nitrogen atoms in a 1,3 -position to one another said optional group is located on the carbon atom between the two nitrogen atoms; the nitrogen atoms of the heterocyclic rings which bear a hydrogen atom being optionally substituted with (Cι-5)alkyl, (C2-5)alkenyl, (Ci^alkanoyl, benzyl, phenlethyl or benzoyl; 1,2,3-thiadiazolyl 2,2 dioxide, the nitrogen atom of which are
optically substituted with (Cι-s)alkyl,(C2-5)alkenyl,(Cι-5)alkanoyl, benzyl, pheylethyl or benzoyl; bicyclic heterocyclic moiety rings containing a total of three nitrogen atoms, one in each of the rings and one common to each ring (anular nitrogen).
US Patent 5,814,651, herein incoφorated by reference in its entirety, describes the compounds represented by formula 4,
Wherein R
1 is selected from the group consisting of methyl, ethyl, difluoromethyl, and trifluoroethyl;
R is selected from the group consisting of (Cι-6) alkyl, alkoxyalkyl having 3 to7 carbons in the alkoxy portion and 2 to 4 carbons in the alkyl portion, phenoxyalkyl having 2 to 6 carbons in alkyl portion, (C3-7)cycloalkyl, (C8-9)polycycloalkyl, phenyl alkyl having 1 to 8 carbons in the alkyl portion. Phenyl amino alkyl having 2 to 6 carbons in alkyl portion and amino may be optionally substituted with (CM) alkyl and indanyl.
Where the alkyl portion of the said alkyl, phenoxy alkyl cycloalkyl, polycycloalkyl, phenylalkyl and indanyl may optionally substituted with one or more fluorine atoms, - OH or (CM) alkoxy, and the aryl portion of the phenylalkyl, phenoxyalkyl and indanyl may optionally be substituted with (C
M)alkyl,
or halogen; A and B are independently selected from the group consisting of a covalent bond, optionally substituted (Cι
-5) alkylene, optionally substituted (C
2-5) alkenyl and optionally substituted phenylene, where the optionally substituted alkylene may be mono-substituted with
or C0
2R
6 and hydroxyl.
The optionally substituted alkenyl may be mono-substituted with (Cι-4)alkoxy or CO2R6
The optionally substituted phenylene may be mono-substituted with (Ci4)alkoxy, CO2R6 or hydroxyl wherein R6 is hydrogen or (Ci-^alkyl
Y is selected from the group consisting of a covalent bond, O, NR6 and S wherein R is as defined above;
Z is selected from the group consisting of
where Q1, Q2, Q3 and Q4 are independently N, CH or when also bonded to B, C and provided that at least two of Q1, Q2, Q3 and Q4 are not N;
X is selected from the group consisting of O, NR4 and S; and X1, X2, X3 and X4 are independently selected from the group consisting of O, NR , S, C=0,CH2 and, when also bonded to B, CH;
b is an integer from 1 to 2; e is an integer from 1 to 3; g is an integer from 1 to 4; j is an integer from 1 to 5; m is an integer from 1 to 7; each R3 is independently selected from the group consisting of hydrogen, halogen,(Cι.
5)alkyl, CH(R7)C02R4, (d-6)alkoxy,C02R4, CONR4R5, CONHOH, CH2NR4R5,
NR4R5, nitro, hydroxyl, CN, S03H phenyl alkyl having 1 to 4 carbon atoms in alkyl portion, S02NR4R5, N(S02R5)2 andNHS02R5, Where R4 for each occurrence is independently selected from the group consisting of hydrogen,(Cι-6)alkyl, phenyl, optionally substituted with (Cι.C4)alkyl or halogen,
CH(R7)C02R5, (C3-C7)cycloalkyl, phenylalkyl having 1 to 4 carbon atoms in alkyl portion and dialkylaminoalkyl having a total of 5 carbons in the dialkylamino portion and having 2 to5 carbon atoms in alkyl portion where R6 is as defined above, R5 for each occurrence is independently selected from the group consisting of hydrogen, (Ci^alkyl, (C3-C )cycloalkyl, phenylalkyl having 1 to 4 carbons in the alkyl portion, phenyl, pyridyl, pyrimidyl, thiazolyl and oxazolyl; or R and R5 are taken together with nitrogen to which they are attached and form an optionally substituted saturated or unsaturated 5- or 6-membered ring, a saturated or unsaturated 6-membered heterocyclic ring containing two hetero atoms or a quinoline ring optionally substituted with fluoro, where said optionally substituted saturated or unsaturated 5-or6-membered ring may be mono or di-substituted and each substituent is independently selected from the group consisting of alkyl having 1 to 4 carbons,C02R7 wherein R7 is as defined below, CONH2, CON(CH3)2, oxo, hydroxyl, NH2, and N(CH3)2 and said saturated or unsaturated 6-membered heterocyclic ring containing two heteroatoms has the second heteroatom selected from the group consisting of O, S, NH, NCH3, NCOCH and
NCH2Ph;
R7 for each occurrence is independently selected from the group consisting of hydrogen and (Cι_4)alkyl; and R8 is selected from the group consisting of (Ci-
C6)alkyl, (C3-C ) cycloalkyl, phenyl and phenylalkyl having 1 to 4 carbons in the alkyl position;
Accordingly we have prepared a novel series of compounds having the general formula I as defined below. We have examined the in vitro efficacy of these novel compounds against human PDE-4 enzyme and they have been found to show excellent PDE-4 enzyme inhibition activity. The compounds of the present invention are useful as therapeutic agents for inflammatory allergic diseases particularly bronchial asthma, allergic rhinitis and nephritis ; Since these compounds also inhibit the production of tumor necrosis factor (TNF), they may also find use in autoimmune diseases such as rheumatoid arthritis, multiple sclerosis, Crohn's disease, psoriasis ; diseases of the central nervous system such as depression amnesia, and dementia, Alzheimers disease, cardiac failure, shock, and cerebrovascular disease and the like; and insulin-resistant diabetes.
Summary of the Invention
Accordingly, the present invention provides novel heterocyclic compounds of the general formula (I) ,
R
1 is independently selected for each occurrence from the group consisting of hydrogen, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstitued alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylakyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted aryl, substituted or unsubstituted arylalkyl, substituted or unsubstituted heterocyclic group, substituted or unsubstituted heterocyclylalkyl, substituted or unsubstituted heteroarylalkyl ,
-C(0)NR R
1 and -S^m-R
1; preferably R
1 is substituted or unsubstituted cycloalkyl; R
1 is further preferred to be cyclopentyl; wherein P is selected from the group consisting of direct bond, oxygen, sulfur or NR
1; preferably P is oxygen; wherein P is selected from the group consisting of hydrogen, halogen atom, -OR
1 , - S(O)
m R
1, -C(0)R' , formyl amine, nitro or -NR
xR
y (wherein R
x and R
y independently represents hydrogen atom, substituted or unsubstituted alkyl, haloalkyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkenyl substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heterocyclylalkyl, substituted or unsubstituted heteroaryl or substituted or unsubstituted heteroarylalkyl ; preferably P
1 is substituted or unsubstituted alkoxy;
P1 is further preferred to be
R2 is selected from the group consisting of hydrogen, halogen, substituted or unsubstituted alkyl, substituted or unsubstituted cycloalkyl, -CN, -CH=CR R , -
C=C(R3), -CH2NHC(0)C(0)NH2, -(O)R3,CH2OR3 ,
-NR3R4, -N02, -CH2NR3R4, -C(Z)H, -C(0)OR3, -C(0)NR3R4 Z is O, -NR5, -NOR3, -NCN, -C(-CN)2, -CR3CN, -CR3N02, -CR3C(O)OR3,
-CR3C(0)NR3R3, -C(-CN)N02, -C(-CN)C(0)OR5, or -C(-CN)C(0)NR3R3; preferably R2 is -CN; where R4 is OR3 or R5
R3 is selected from the group consisting of hydrogen, substituted or unsubstituted alkyl and halogen;
R5 is selected from the group consisting of hydrogen and substituted or unsubstituted alkyl;
The ring 'A' is selected from the group consisting of substituted or unsubstituted aryl ring and optionally substituted saturated or unsaturated five to seven membered heterocyclic ring which contains one or more heteroatoms selected from the group consisting of O, S and NR
1 ; the preferable rings can be selected from
wherein
R , R7 and R8 are independently chosen from the group consisting of hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above.
R and R , 10 are independently selected from the group consisting of hydrogen, hydroxy, halogen, nitro, cyano, formyl, substituted or unsubstituted alkyl, substituted
or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or R9 and R10 may be joined to a form a optionally substituted saturated or unsaturated cyclic ring, which may optionally include up to two heteroatoms selected from O, NR6 or S; and
R11, R12, and R13 are independently selected from the group consisting of hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above. The A ring is further preferred to be selected from the group consisting of
The substructure;
> represents the A ring apended to the cyclohexyl ring of formula I. For example, where A represents
would correspond to a structural formula of
Compounds according to formula I further include their analogs, their tautomers, their regioisomers, their diasteromers, their stereoisomers, their geometrical isomers, their N-oxides, their polymoφhs, their pharmaceutically acceptable salts, and their pharmaceutically acceptable solvates thereof.
Detailed Description of the Invention
The term 'alkyl' refers to a straight or branched hydrocarbon chain radical having from one to eight carbon atoms, and which is attached to the rest of the molecule by a single bond, examples include but are not limited to methyl, ethyl, n- propyl, and 1-methylethyl (isopropyl), n-butyl, n-pentyl, 1,1-dimethylethyl (t-butyl).
The term "alkenyl" refers to aliphatic hydrocarbon group containing a carbon- carbon double bond and which may be a straight or branched chain radical having 2 to
10 carbon atoms which is attached to the rest of the molecule by a single bond. Examples include but are not limited to ethenyl, 1-propenyl, 2-propenyl (allyl), iso- propenyl, 2 -methyl- 1-propenyl, 1-butenyl, and 2-butenyl and the like.
The term "alkynyl" refers to straight or branched chain hydrocarbon radicals having at least one carbon-carbon triple bond, having 2 to 12 carbon atoms (with radicals having in the range of about 2 up to 10 carbon atoms preferred) which is attached to the rest of the molecule by a single bond. Examples include but is not limited to ethynyl, propynyl, and butnyl.
The term "alkoxy" denotes alkyl group as defined above attached via oxygen linkage to the rest of the molecule. Examples include but are not limited to -OCH3, - and -OC2H5.
The term "alkylcarbonyl" denotes alkyl group as defined above attached via carbonyl linkage to the rest of the molecule. Examples include but are not limited to
-C(0)CH3, and - C(0)C2H5. The term "alkoxycarbonyl" denotes alkoxy group as defined above attached via carbonyl linkage to the rest of the molecule. Examples include but are not limited to
-C(0)-OCH3, and - C(0)-OC2H5.
The term "alkylcarbonyloxy" denotes alkylcarbonyl group as defined above attached via oxygen linkage to the rest of the molecule. Examples include but are not limited to -O-C(0)CH3, and - 0-C(0)C2H5.
The term "alkylamino" denotes alkyl group as defined above attached via amino linkage to the rest of the molecule. Examples include but are not limited to - NHCH3, -N(CH3)2 and N(CH3)(CH2CH3)
The term "cycloalkyl" denotes a non-aromatic mono or multicyclic ring system of 3 to about 14 carbon atoms attached via a single bond to the rest of the molecule. Examples of monocyclic ring system include but are not limited to cyclopropyl, cyclobutyl, cyclopentyl and, cyclohexyl. Examples of multicyclic ring system include but are not limited to perhydronapthtliyl, adamantyl and norbornyl groups bridged cyclic group or sprirobicyclic groups e.g. sprio (4,4) non-2 -yl.
The term "cycloalkylalkyl" refers to cyclic ring-containing radical containing
3 to about 8 carbon atoms directly attached to alkyl group which is then attached to the main structure at any carbon from alkyl group that results in the creation of a stable structure, such as cyclopropylmethyl, cyclobutylethyl, cyclopentylethyl, and the like.
The term "cycloalkenyl" refers to cyclic ring-containing radicals contaimng in the range of about 3 up to 8 carbon atoms with at least one carbon- carbon double bond. Examples include but are not limited to cyclopropenyl, cyclobutenyl and cyclopentenyl. The term "aryl" refers to aromatic radicals having 6 to 14 carbon atoms.
Examples include but are not limited to phenyl, naphthyl, tetrahydronapthyl, indanyl and biphenyl.
The term "arylalkyl" refers to an aryl ring as defined above directly bonded to an alkyl group as defined above. Examples include but are not limited to -CH2C6H5, and
-C2HsC6H5.
The term "heterocyclic ring" refers to a stable 3- to 15 membered ring which consists of carbon atoms and from one to five heteroatoms selected from the group consisting of nitrogen, phosphorus, oxygen and sulfur. For pmpose of this invention, the heterocyclic ring radical may be a monocyclic, bicyclic or tricyclic ring system, which may include fused, bridged or spiro ring systems, and the nitrogen, phosphorus, carbon, oxygen or sulfur atoms in the heterocyclic ring radical may be optionally oxidized to various oxidation states. In addition, the nitrogen atom may be optionally quaternized; and the heterocyclic ring radical may be partially or fully saturated or aromatic (heteroaryl). Examples of such heterocyclic ring radicals include, but are not limited to, azetidinyl, acridinyl, benzodioxolyl, benzodioxanyl, benzofurnyl, carbazolyl, cinnolinyl, dioxolanyl, indolizinyl, naphthyridinyl, perhydroazepinyl, phenazinyl, phenothiazinyl, phenoxazinyl, phthalazinyl, pyridyl pteridinyl, purinyl, quinazolinyl, quinoxalinyl, quinolinyl, isoquinolinyl, tetrazoyl, imidazolyl, tetrahydroisouinolyl, piperidinyl, piperazinyl, 2-oxopiperazinyl, 2-oxopiperidinyl, 2- oxopyrrolidinyl, 2-oxoazepinyl, azepinyl, pyrrolyl, 4-piρeridonyl, pyrrolidinyl, pyrazinyl, pyrimidinyl, pyridazinyl, oxazolyl, oxazolinyl, oxasolidinyl, triazolyl,
indanyl, isoxazolyl, isoxasolidinyl, moφholinyl, thiazolyl, thiazolinyl, thiazolidinyl, isothiazolyl, quinuclidinyl, isothiazolidinyl, indolyl, isoindolyl, indolinyl, isoindolinyl, octahydroindolyl, octahydroisoindolyl, quinolyl, isoquinolyl, decahydroisoquinolyl, benzimidazolyl, thiadiazolyl, benzopyranyl, benzothiazolyl, benzooxazolyl, furyl, tetrahydrofurtyl, tetrahydropyranyl, thienyl, benzothienyl, thiamoφholinyl, thiamoφholinyl sulfoxide thiamoφholinyl sulfone, dioxaphospholanyl, oxadiazolyl, chromanyl and isochromanyl.
The term "heteroarylalkyl" refers to heteroaryl ring radical as defined above directly bonded to alkyl group. The heteroarylalkyl radical may be attached to the main structure at any carbon atom from alkyl group that results in the creation of a stable structure.
The term "heterocyclylalkyl" refers to a heterocylic ring as defined above directly bonded to alkyl group. The heterocyclylalkyl radical may be attached to the main structure at carbon atom in the alkyl group that results in the creation of a stable structure.
The term "cyclic ring" refers to a cyclic group containing 3-10 carbon atoms The term "Halogen" refers to radicals of Fluorine, Chlorine, Bromine, Iodine
The substituents in the 'substituted alkyl', 'substituted alkoxy' 'substituted alkenyl' ' substituted alkynyl' 'substituted cycloalkyl' substituted cycloalkylalkyl' substituted cyclocalkenyl' 'substituted arylalkyl' 'substituted aryl' 'substituted heterocyclic ring', 'substituted heteroaryl ring,' 'substituted heteroarylalkyl', 'substituted heterocyclylalkyl ring', 'substituted amino', 'substituted alkoxycarbonyl', 'substituted cyclic ring' 'substituted alkylcarbonyl', 'substituted alkylcarbonyloxy' and
" 'substituted carboxylic acid' may be the same or different which one or more selected from the groups such as hydrogen, hydroxy, halogen, carboxyl, cyano, amino, nitro, oxo (=0), thio (=S), or optionally substituted groups selected from alkyl, alkoxy, alkenyl, alkynyl, aryl, arylalkyl, cycloalkyl, aryl, heteroaryl, heteroarylalkyl, heterocyclic ring, -COORx, -C(O)Rx, -C(S)RX, -C(0)NRxRy, -C(0)ONRxRy, - NRxCONRyRz, -N(Rx)SORy, -N(Rx)S02Ry, -(=N-N(Rx)Ry), - NRxC(O)ORy, -NRxRy, - NRxC(0)Ry-, -NRxC(S)Ry -NRxC(S)NRyRz, -SONRxRy-, -S02NRxRy-, -ORx, - ORxC(0)NRyRz, -ORxC(0)ORy-, -OC(0)Rx, -OC(0)NRxRy, -RxNRyRz, -RxRyRz, - RXCF3, -RxNRyC(O)Rz, -RxORy, -RxC(0)ORy, -RxC(0)NRyRz, -RxC(0)Rx, -
RxOC(0)Ry, -SRX, -SORx, -S02Rx, -ON02,(wherein Rx, Ry and Rz in each of the above groups can be hydrogen atom, substituted or unsubstituted alkyl, haloalkyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl substituted or unsubstitued heterocyclic ring, substituted or unsubstitued heterocyclylalkyl, substituted or unsubstitued heteroaryl or substituted or unsubstitued heteroarylalkyl )
Pharmaceutically acceptable salts forming part of this invention include salts derived from inorganic bases such as Li, Na, K, Ca, Mg, Fe, Cu, Zn, Mn; salts of organic bases such as N,N'-diacetylethylenediamine, glucamine, triethylamine, choline, choline hydroxide, dicyclohexylamine, metformin, benzylamine, trialkylamine, thiamine, spermidine, and the like; alkyl halides such as methyl halide, ethyl halide and the like; Aryl alkyl halide such as benzylhalide and the like; chiral bases like alkylphenylamine, glycinol, phenyl glycinol and the like, salts of natural amino acids such as glycine, alanine, valine, leucine, isoleucine, norleucme, tyrosine, cystine, cysteine, methionine, proline, hydroxy proline, histidine, omithine, lysine, arginine, serine, and the like; unnatural amino acids such as D-isomers or substituted amino acids; guanidine, substituted guanidine wherein the substituents are selected from nitro, amino, alkyl, alkenyl, alkynyl, ammonium or substituted ammonium salts and aluminum salts. Salts may include acid addition salts where appropriate which are, sulphates, nitrates, phosphates, perchlorates, borates, hydrohalides, acetates, trifluroacetate, tartrates, maleates, citrates, succinates, palmoates, methanesulphonates, benzoates, salicylates, benzenesulfonates, ascorbates, glycerophosphates, ketoglutarates and the like. Pharmaceutically acceptable solvates may be hydrates or comprise other solvents of crystallization such as alcohols.
Another object of the invention is a method of treating inflammatory diseases, disorders and conditions characterized by or associated with an undesirable inflammatory immune response and all disease and conditions induced by or associated with an excessive secretion of TNF-α and PDE-4 which comprises administering to a subject a therapeutically affective amount of a compound according to claim 1.
Another object of the invention is a method of treating inflammatory conditions and immune disorders in a subject in need thereof which comprises administering to said subject a therapeutically effective amount of a compound according to Formula I.
Preferred inflammatory conditions and immune disorders are chosen from the group consisting of asthma, bronchial asthma, chronic obstructive pulmonary disease, allergic rhinitis, eosinophilic granuloma, nephritis, rheumatoid arthritis, cystic fibrosis, chronic bronchitis, multiple sclerosis, Crohns disease, psoraisis, uticaria, adult vernal cojunctivitis, respiratory distress syndrome, rhematoid spondylitis, osteoarthritis, gouty arthritis, uteltis, allergic conjunctivitis, inflammatory bowel conditions, ulcerative coalitis, eczema, atopic dermatitis and chronic inflammation.
Further preferred is when the inflammatory condition is an allergic inflammatory condition.
Further preferred is when the inflammatory conditions and immune disorders are selected from the group consisting of inflammatory conditions or immune disorders of the lungs, joints, eyes, bowels, skin and heart.
Further preferred is when the inflammatory condition is chosen from the group consisting of bronchial asthma, nepritis, and allergic rhinitis.
Another object of the invention is a method for abating inflammation in an affected organ or tissue comprising delivering to said organ or tissue a therapeutically effective amount of a compound represented by a compound according to Formula 1.
Another object of the invention is a method of treating diseases of the central nervous system in a subject in need thereof which comprises administering to said subject a therapeutically effective amount of a compound according to Formula 1.
Preferred diseases of the central nervous system are chosen from the group consisting of depression, amnesia, dementia, Alzheimers disease, cardiac failure, shock and cerebrovascular disease.
Another object of the invention is a method of treating insulin resistant diabetes in a subject in need thereof which comprises administering to said subject a therapeutically effective amount of a compound according to Formula 1.
"Treating" or "treatment" of a state, disorder or condition includes:
(1) preventing or delaying the appearance of clinical symptoms of the state, disorder or condition developing in a mammal that may be afflicted with or predisposed to the state, disorder or condition but does not yet experience or display clinical or subclinical symptoms of the state, disorder or condition,
(2) inhibiting the state, disorder or condition, i.e., arresting or reducing the development of the disease or at least one clinical or subclinical symptom thereof, or
(3) relieving the disease, i.e., causing regression of the state, disorder or condition or at least one of its clinical or subclinical symptoms.
The benefit to a subject to be treated is either statistically significant or at least perceptible to the patient or to the physician
A "therapeutically effective amount" means the amount of a compound that, when administered to a mammal for treating a state, disorder or condition, is sufficient to effect such treatment. The "therapeutically effective amount" will vary depending on the compound, the disease and its severity and the age, weight, physical condition and responsiveness of the mammal to be treated.
The four classic symptoms of acute inflammation are redness, elevated temperature. Swelling, and pain in the affected area, and loss of function of the affected organ.
Symptoms and signs of inflammation associated with specific conditions include:
• rheumatoid arthritis- pain, swelling, warmth and tenderness of the involved joints; generalized and morning stiffness; • insulin-dependent diabetes mellitus- insulitis; this condition can lead to a variety of complications with an inflammatory component, including: retinopathy, neuropathy, nephropathy; coronary artery disease, peripheral vascular disease, and cerebrovascular disease;
• autoimmune thyroiditis- weakness, constipation, shortness of breath, puffϊness of the face, hands and feet, peripheral edema, bradycardia;
• multiple sclerosis- spasticity, blurry vision, vertigo, limb weakness, paresthesias;
• uveoretinitis- decreased night vision, loss of peripheral vision;
• lupus erythematosus- joint pain, rash, photosensitivity, fever, muscle pain, puffiness of the hands and feet, abnormal urinalysis (hematuria, cylinduria, proteinuria), glomerulonephritis, cognitive dysfunction, vessel thrombosis, pericarditis;
• scleroderma- Raynaud's disease; swelling of the hands, arms, legs and face; skin thickening; pain, swelling and stiffness of the fingers and knees, gastrointestinal dysfunction, restrictive lung disease; pericarditis,; renal failure;
• other arthritic conditions having an inflammatory component such as rheumatoid spondylitis, osteoarthritis, septic arthritis and polyarthritis- fever, pain, swelling, tenderness; • other inflammatory brain disorders, such as meningitis, Alzheimer's disease,
AIDS dementia encephalitis- photophobia, cognitive dysfunction, memory loss;
• other inflammatory eye inflammations, such as retinitis- decreased visual acuity; • inflammatory skin disorders, such as , eczema, other dermatites (e.g., atopic, contact), psoriasis, burns induced by UN radiation (sun rays and similar UN sources)- erythema, pain, scaling, swelling, tenderness;
• inflammatory bowel disease, such as Crohn's disease, ulcerative colitis- pain, diarrhea, constipation, rectal bleeding, fever, arthritis;
• asthma- shortness of breath, wheezing;
• other allergy disorders, such as allergic rhinitis- sneezing, itching, runny nose • conditions associated with acute trauma such as cerebral injury following stroke- sensory loss, motor loss, cognitive loss;
• heart tissue injury due to myocardial ischemia- pain, shortness of breath;
• lung injury such as that which occurs in adult respiratory distress syndrome- shortness of breath, hyperventilation, decreased oxygenation, pulmonary infiltrates;
• inflammation accompanying infection, such as sepsis, septic shock, toxic shock syndrome- fever, respiratory failure, tachycardia, hypotension, leukocytosis;
• other inflammatory conditions associated with particular organs or tissues, such as nephritis (e.g., glomerulonephritis)-oliguria, abnormal urinalysis; inflamed appendix- fever, pain, tenderness, leukocytosis; gout- pain, tenderness, swelling and erythema of the involved joint, elevated serum and/or urinary uric acid; inflamed gall bladder- abdominal pain and tenderness, fever, nausea, leukocytosis; chronic obstructive pulmonary disease- shortness of breath, wheezing; congestive heart failure- shortness of breath, rales, peripheral edema; Type II diabetes- end organ complications including cardiovascular, ocular, renal, and peripheral vascular disease lung fibrosis- hyperventilation, shortness of breath, decreased oxygenation; vascular disease, such as atherosclerosis and restenosis- pain, loss of sensation, diminished pulses, loss of function and alloimmunity leading to transplant rejection- pain, tenderness, fever.
Subclinical symptoms include without limitation diagnostic markers for inflammation the appearance of which may precede the manifestation of clinical symptoms. One class of subclinical symptoms is immunological symptoms, such as the invasion or accumulation in an organ or tissue of proinflammatory lymphoid cells
or the presence locally or peripherally of activated pro-inflammatory lymphoid cells recognizing a pathogen or an antigen specific to the organ or tissue. Activation of lymphoid cells can be measured by techniques known in the art.
"Delivering" a therapeutically effective amount of an active ingredient to a particular location within a host means causing a therapeutically effective blood concentration of the active ingredient at the particular location. This can be accomplished ,e.g., by local or by systemic administration of the active ingredient to the host.
"A subject" or "a patient" or "a host" refers to mammalian animals, preferably human.
Some of the representative compounds according to the present invention are specified below but should not construed to be limited thereto;
1. 5-(3 -Cyclopentyloxy-4-methoxyphenyl)-3-oxo-2,3 ,4,5 ,6,7-hexahydro- 1H- indazole-5-carbonitrile. 2. 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-3-oxo-2,3,4,5,6,7-hexahydro- lH-indazole-5-carbonitrile.
3. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-lH-5-indazole- carbonitrile.
4. 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4,5,6,7-tetrahydro-lH-5- indazolecarbonitrile.
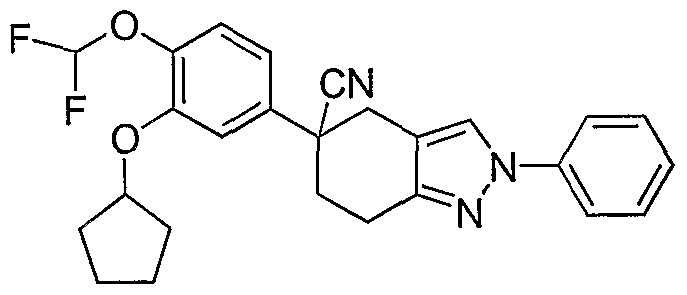
5. 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-2-phenyl-4,5,6,7-tetrahydro- 2H-indazole-5-carbonitrile.
6. 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-l -phenyl-4,5,6,7-tetrahydro- . 2Η-indazole-5-carbonitrile. 7. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-2-(2-pyidyl)-4,5,6,7-tetrahydro-2H-5- indazole-carbonitrile .
8. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-methyl-4,5,6,7-tetrahydro-lH-5- indazolecarbonitrile.
8B.5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-methyl-4,5,6,7-tetrahydro-2H-5- indazolecarbonitrile.
9. 2-[5-Cyano-5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-lH-l- indazoyl] acetic acid.
9B.2-[5-Cyano-5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-2H-l- indazoyl] acetic acid.
10. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydrobenzo[c]isoxazole-5- carbonitrile.
11. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-3-methyl-2-oxo-2,3,4,5,6,7-hexahydro- benzo[^[l,3]oxazole-5-carbonitrile.
12. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-vinyl-4,5,6,7-tetrahydro-lH-5- indole-carbonitrile .
13. 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-l-vinyl-4,5,6,7-tetrahydro-lH- 5 -indolecarbonitrile . 14. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-lH-5-indole- carbonitrile. i 15. 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4,5,6,7-tetrahydro- 1H-5- indazole-carbonitrile .
16. 5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-methyl-4,5,6,7-tetrahydro-lH-5- indole-carbonitrile .
17. Ethyl 6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-2-methyl-5,6,7,8- tetrahydro-3-quinolinecarboxylate.
18. 6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-2-methyl-5,6,7,8-tetrahydro-3- quinolinecarboxylic acid. 19. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-oxo-3 ,4,5,6,7,8-hexahydro-6- quinazoline-carbonitrile.
20. 6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazolinecarbonitrile.
21. 6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazoline-carbonitrile.
22. 6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-2-methyl-4-oxo-3,4,5,6,7,8- hexahydro-6-quinazolinecarbonitrile.
23. 2-Amino-6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro- 6-quinazolinecarbonitrile.
24. 2-Amino-6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4-oxo-3,4,5,6,7,8- hexahydro-6-quinazolinecarbonitrile.
25. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-3-methyl-4-oxo-3,4,5,6,7,8-hexahydro- 6-qumazolinecarbonitrile.
26. Ethyl 2-[6-cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-4-oxo-3 ,4,5,6,7,8- hexahydro-3 -quinazolinyl] acetate . 27. 2-[6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-4-oxo-3 ,4,5,6,7,8- hexahydro-3 -quinazolinyl] acetic acid.
28. Ethyl 2-[6-cyano-6-(3 -cyclopentyloxy-4-methoxyphenyl)-2-methyl-4-oxo- 3,4,5,6,7,8-hexahydro-3-quinazolinyl]acetate.
28B.Ethyl 2-[6-cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-2-methyl-5,6,7,8- tetrahydro-4-quinazolinyloxy]acetate.
29. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile.
29A.6-(3-Cyclopentyloxy-4-methoxyphenyl)-l-oxo-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile .
30. 6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile.
31. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-2-methyl-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile.
32. 2-Amino-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile. 33. 2-Amino-6-(3-cyclopentyloxy-4-difluoromethoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile .
34. Nl-[6-Cyano-6-(3-Cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-2- quinazolinyl] -ac etamide .
35. 2-[6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-2- quinazolinylammo] -acetic acid.
36. 2-Methylthio-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile.
37. 2-Methanesulfonyl-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro- 6-quinazoline-carbonitrile.
38. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-methoxy-5,6,7,8-tetrahydro-6- quinazolme-carbonitrile.
39. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-methylamino-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile.
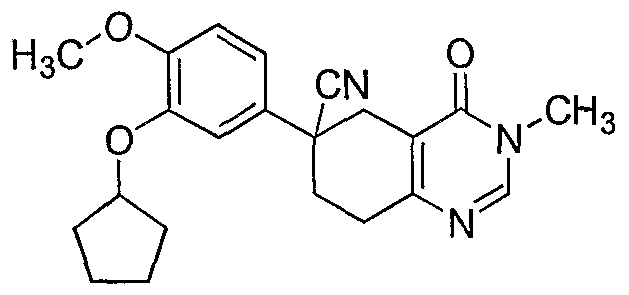
40. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-dimethylamino-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile . 41. 4-n-Butylamino-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile .
42. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-moφholino-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile.
43. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-phenylamino-5,6,7,8,-tetrahydro-6- quinazolinecarbonitile .
44. 4-Benzylamino-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile.
45. 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-phenethylamino-5,6,7,8-tetrahydro- 6-quinazolinecarbonitrile.
The compounds according to the invention may be prepared by the following processes. The symbols P, P1, R1, R2 and A when used in the below formulae below are to be understood to present those groups described above in relation to formula (I) unless otherwise indicated
In one embodiment the desired compounds of the formula (I) wherein P, P , R1, R2 and A are as described in the general description, can be synthesized as described in the general synthetic scheme 1.
Scheme 1:
As shown in the above Scheme 1, the intermediate of formula 5 on reduction with NaBH4 in methanol at RT gives the compound of formula 6.The compound of formula 6 on reaction with SOCl2 followed by NaCN in DMF gives compound of formula 7. The compound 7 on reaction with methyl acrylate in presence of Triton B gives the compound of formula 8. The intramolecular cyclisation of the diester of fonnula 8 gives the compound of formula 9. This compound 9 is condensed with hydrazine in a refluxing alcoholic solvent preferably methanol at a temperature of about 25° to about 200°C preferably about 65°C, to obtain the compound of the general formula 1A.
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 2.
Scheme 2:
1 B
As shown in the above scheme 2, the compound of formula 9 (as prepared in scheme 1) on reaction with NaCl in DMSO-water at elevated temperature gives the compound of formula 10. The compound of general formula 10 readily undergoes condensation with dimethylformamide dimethyl acetal to give the compound of general formula 11. This compound of the formula 11 on reaction with various nucleophiles of general formula H2N-NHR in an alcoholic solvent preferably methanol or ethanol at a temperature of about 20° to about 60°C to obtain the compound of the general formula IB. In case of monosubstituted hydrazines regioisomeric products are formed. The regioisomers formed are further separated and characterized; where R6 can be selected from hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroaryl ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclolalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy or substituted, unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above..
Scheme 3:
As shown in the above scheme 3, the intermediate of the formula ll(as prepared in the scheme 2) is reacted with hydroxylamine under appropriate conditions to obtain compounds of the general formula IC.
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 4.
Scheme 4:
As shown in the above scheme 4, the compound of formula 10 (as prepared in scheme 2) on reaction with oxime in presence of alcohol as solvent gives the compound of formula 12, this compound of the formula 12 is reacted with dimethyl carbonate under appropriate basic reaction conditions affords the compounds of general formula ID.
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 5.
Scheme 5:
As shown in the above scheme 5, the reaction of oxime intermediate of general formula 12 with acetylene gas in DMSO, in a sealed tube gave a mixture of tetrahydroindole derivatives of formulae 13 & 14. The N-vinyl group of 13 can be cleaved to the N-H indole of general formula 14 by using the Hg(II)OAc followed by NaBH reduction. Compound 14 on alkylation under appropriate basic reaction conditions gave the compound of the general formula IE.
Alternatively, the desired compounds of the formula (I) wherein P, P , R , R and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 6.
Scheme 6:
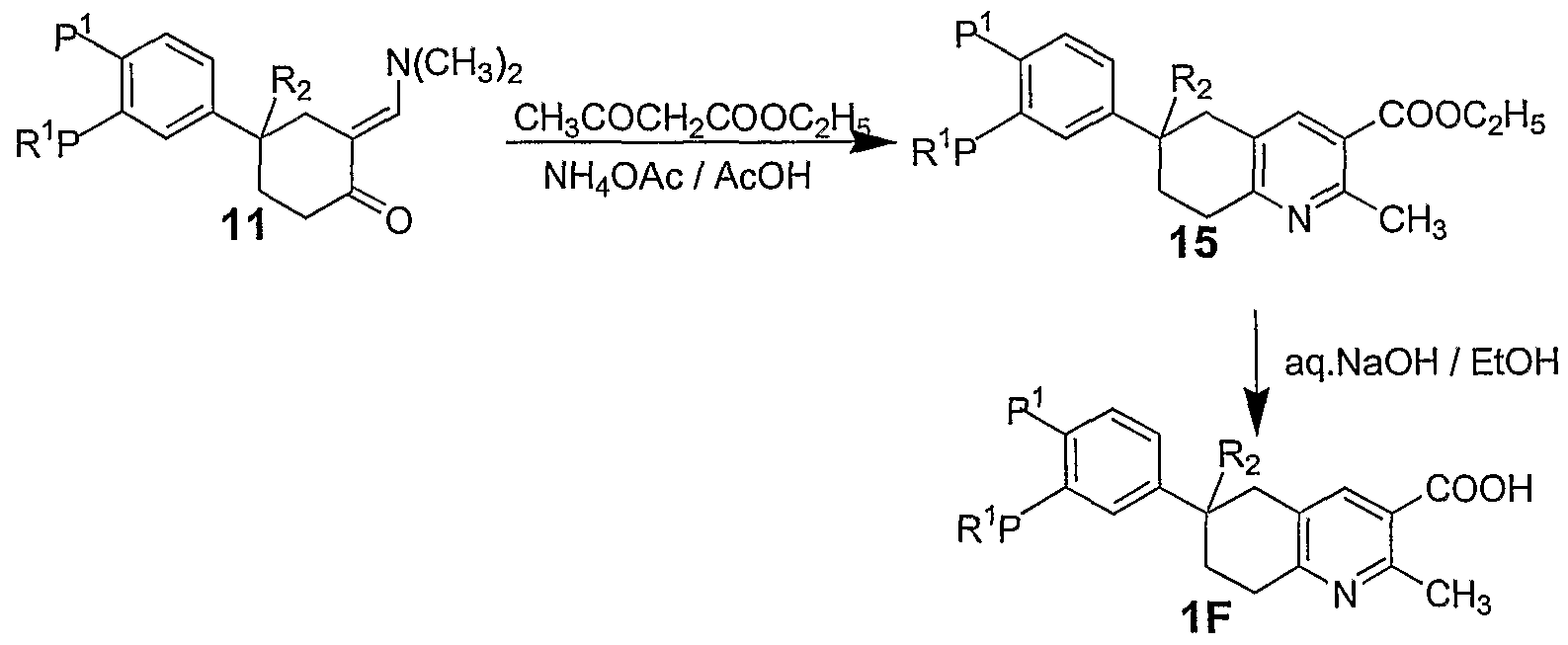
As shown in the above Scheme 6, the condensation of intermediate 11 with ethyl acetoacetate and ammonium acetate in refluxing acetic acid gave compound of general formula 15 which on hydrolysis under basic conditions give the compound of general formula IF.
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic scheme 7. Scheme 7:
As shown in the above scheme 7, the condensation of compound 9 with the 1,3- nucleophiles of general formula 16 gave the compound of formula 17. Alkylation of
compound of formula 17 in presence of cesium carbonate gave both N-alkylated and O-alkylated products 1G. Selective N-alkylation occurred when R2 is hydrogen. Where R , R7 may be same or different and are independently selected from hydrogen, hydroxy, halogen, cyano, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy or substituted, unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above.. Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 8.
Scheme 8:
As shown in the above Scheme 8, the cyclocondensation of intermediate of general formula 11 with the 1 ,3-nucleophiles of general formula 16 in the presence of suitable organic or inorganic bases such as KOH, NaOH, NaHC03, sodium ethoxide, sodium methoxide, triethyl amine and diisopropyl ethylamine and pyridine in an alcoholic solvent preferably ethanol or methanol at a temperature of about 20 to about 70°C yield the compound of the general formula IH. Where R6 can be selected from hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or
unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or S(0)mR where m is 0,1 or 2 and R6 is as described above.
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 9.
Scheme 9:
As shown in the above Scheme 9, the reaction of compound of general formula 17 ( as described in scheme 7) in presence of phosphorous oxychloride yield the compound of general formula 18 which on aromatic nucleophilic substitution reaction with alkoxides of general formula R8OM where M can be Na, K, Li under mild conditions give the compound of the general formula IH. The compound of general formula 18 on reaction with various amines under appropriate conditions gives compound of general formula 1J. Where R6, is as previously defined and R8 is selected from hydrogen, hydroxy, halogen, cyano, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl,
substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above.
Where R9, R10 may be same or different and are independently selected from hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or R9 and R may be joined to a form a optionally substituted saturated or unsaturated cyclic ring, which may optionally include up to two heteroatoms selected from O, NR or S;
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 10.
Scheme 10:
As shown in the above Scheme 10, the reaction of compound of general formula 10 (as prepared in scheme 2) with hydroxy(tosyloxy)iodobenzene gives the compound of formula 19. Reaction of compound of general formula 19 with the 1,3-nucleophiles of general formula 16 under appropriate conditions yield the compound of the general formula IK. where R6, can be selected from hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above.
Alternatively, the desired compounds of the formula (I) wherein P, P1, R1, R2 and A are as described in the general description, can also be synthesized as described in the general synthetic Scheme 11.
Scheme 11:
alkylation or formylation
As shown in the above Scheme 11 the reaction of 10 with an optionally substituted aldehyde of the general formula 20 in presence of a suitable base such as alkyl lithium, LDA, LHMDS gave intermediate of general formula 21. Intermediate 21 can be cyclised to indole derivate 22 under suitable acidic conditions.The intermediate 22 can be further functionalized to give optionally substituted indole derivative IL using conventional chemical transformations.
Where R11, R12, and R13 may be same or different and are independently selected from hydrogen, hydroxy, halogen, cyano, nitro, formyl, substituted or unsubstituted alkyl, substituted or unsubstituted alkoxy, substituted or unsubstituted alkenyl, substituted or unsubstituted alkynyl, substituted or unsubstituted cycloalkyl, substituted or unsubstituted cycloalkylalkyl, substituted or unsubstituted cycloalkenyl, substituted or unsubstituted arylalkyl, substituted or unsubstituted aryl, substituted or unsubstituted heterocyclic ring, substituted or unsubstituted heteroarylalkyl, substituted or unsubstituted heterocyclylalkyl ring, substituted or unsubstituted amino, substituted or unsubstituted alkoxycarbonyl, substituted or unsubstituted cyclic ring, substituted or unsubstituted alkylcarbonyl, 'substituted or unsubstituted alkylcarbonyloxy, substituted or unsubstituted carboxylic acid or S(0)mR6 where m is 0,1 or 2 and R6 is as described above.
The substances according to the invention are isolated and purified in a manner known per se, e.g. by distilling off the solvent in vacuum and recrystallizing the residue obtained from a suitable solvent or subjecting it to one of the customary purification methods, such as column chromatography on a suitable support material.
Salts are obtained by dissolving the free compound in a suitable solvent, e.g in a chlorinated hydrocarbon, such as methylene chloride or chloroform, or a low molecular weight aliphatic alcohol (ethanol, isopropanol) which contains the desired acid or base, or to which the desired acid or base is then added. The salts are obtained by filtering, reprecepitating, precipitating with a non-solvent for the addition salt or by evaporating the solvent. Salts obtained can be converted by basification or by acidifying into the free compounds which, in turn can be converted into salts.
In general, the ethereal solvents used in the above described processes for the preparation of compounds of the formula (I) are selected from diethyl ether, 1,2- dimethoxyethane, tetrahydrofuran, diisopropyl ether, 1,4 dioxane and the like. The chlorinated solvent which may be employed may be selected from dichloromethane, 1,2-dichloroethane, chloroform, carbontetrachloride and the like. The aromatic solvents which may be employed may be selected from benzene, toluene. The alchoholic solvents which may be employed may be selected from methanol, ethanol, n-propanol, iso propanol, tert.butanol and the like. The aprotic solvents which may be employed may be selected from N, N-dimethylformamide, dimethyl sulfoxide and the like.
In general, the compounds prepared in the above described processes are obtained in pure form by using well known techniques such as crystallization using solvents such as pentane, diethyl ether, isopropyl ether, chloroform, dichloromethane, ethyl acetate, acetone, methanol, ethanol, isopropanol, water or their combinations, or column chromatography using Alumina or silica gel and eluting the column with solvents such as hexane, petroleum ether (petether), chloroform, ethyl acetate, acetone, methanol or their combinations. Various polymoφhs of a compound of general formula (I) forming part of this invention may be prepared by crystallization of compound of formula (I) under different conditions, example, using different solvents commonly used or their mixtures for recrystallization; crystallizations at different temperatures, various modes of cooling, ranging from very fast to very slow cooling during crystallizations.
Polymoφhs may also be obtained by heating or melting the compound followed by gradual or fast cooling. The presence of polymoφhs may be determined by solid probe NMR spectroscopy, IR spectroscopy, differential scanning calorimetry, powder X-ray diffraction or such other techniques. The present invention provides novel heterocyclic compounds, their analogs, their tautomers, their regioisomers, their stereoisomers, their enantiomers, their diastreomers, their polymoφhs, their pharmaceutically acceptable salts, their appropriate N-oxides and their pharmaceutically acceptable solvates.
The present invention also provides pharmaceutical compositions, containing compounds of the general formula (T) as defined above, their derivatives, their analogs, their tautomeric forms, their stereoisomers, their polymoφhs, their enantiomers, their diasteromers, their pharmaceutically acceptable salts or their pharmaceutically acceptable solvates in combination with the usual pharmaceutically employed carriers, diluents and the like. The pharmaceutical compositions according to this invention can be used for the treatment of allergic disorders.
It will be appreciated that some of the compounds of the general formula (I) defined above according to the invention can contain one or more asymmetrically substituted carbon atoms. The presence of one or more of these asymmetric centers in the compounds of the general formula (I) can give rise to stereoisomers and in each case the invention is to be understood to extend to all such stereoisomers, including enantiomers and diastereomers and their mixtures, including racemic mixtures. The invention may also contain E & Z geometrical isomers wherever possible in the compounds of the general formula (I) which includes the single isomer or mixture of both the isomers
The pharmaceutical compositions may be in the forms normally employed, such as tablets, capsules, powders, syrups, solutions, suspensions and the like and may contain flavorants, sweeteners etc. in suitable solid or liquid carriers or diluents, or in suitable sterile media to form injectable solutions or suspensions. The active compounds of the formula (I) will be present in such pharmaceutical compositions in the amounts sufficient to provide the desired dosage in the range as described above. Thus, for oral administration, the compounds of the formula (I) can be combined with a suitable solid, liquid carrier or diluent to form capsules, tablets, powders, syrups, solutions, suspensions and the like. The pharmaceutical compositions, may, if
desired, contain additional components such as flavorants, sweeteners, excipients and the like. For parenteral administration, the compounds of the formula (I) can be combined with sterile aqueous or organic media to form injectable solutions or suspensions. For example, solutions in sesame or peanut oil, aqueous propylene glycol and the like can be used as well as aqueous solutions of water-soluble pharmaceutically-acceptable acid addition salts or salts with base of the compounds of the formula (T). The injectable solutions prepared in this manner can then be administered intravenously, intraperitoneally, subcutaneously, or intramuscularly, with intramuscular administration being prefened in humans.
In addition to the compounds of formula (T) the pharmaceutical compositions of the present invention may also contain or be co-administered with one or more known drugs selected from other clinically useful therapeutic agents.
The invention is explained in detail in the examples given below which are provided by way of illustration only and therefore should not be construed to limit the scope of the invention.
Intermediate 1
2-Carbomethoxy-4-cyano-4-[3-(cyclopentyloxy)-4-methoxyphenyl]cyclohexan-l- one
Step 1: 3-Cyclopentyloxy-4-methoxbenzaldehyde
Cyclopentyl bromide (29.3 g, 0.19 mol) was added to a well stirred slurry of 4- methoxy-3-hydroxybenzaldehyde (20 g, 0.13 mol) and K2C03 (36.3 g, 0.26 mol) in dry DMF (250 ml) and the mixture was stined at 70-80 °C for 8 h under nitrogen atmosphere. The mixture was cooled to room temperature, quenched with ice-cold water (500 ml) and extracted with EtOAc (2 x 250 ml). The combined orgamc extracts were washed with water (3 x 500 ml), brine (250 ml) and dried (Na2S0 ). The solvent was evaporated under reduced pressure to give the aldehyde as viscous oil (28 g). IR (neat) 2963, 2872, 1687, 1585, 1434, 1396, 1266, 1132, 1024, 756 cm"1. Η NMR (300 MHz, CDC13) δ 1.58-2.10 (m, 8 H), 3.93 (s, 3 H), 4.83-4.87 (m, 1 H), 6,96 (d, J= 8.4 Hz, 1 H), 7.39-7.44 (m, 2 H), 9.84 (s, IH).
Step 2: 3-Cyclopentyloxy-4-methoxybenzyl alcohol
Sodium borohydride (4.5 g, 0.118 mol) was added in portions to a stined solution of 3-cyclopentyloxy-4-methoxybenzaldehyde (26 g, 0.118 mol) in dry methanol (250 ml) at 10 °C and stirred for 30 min at the same temperature temperature. Excess
NaBH4 was quenched with acetone (3 ml) and most of the methanol was removed under reduced pressure. The residue was dissolved in ethyl acetate (500 ml) and washed with water (3 x 500 ml) followed by brine (300 ml). The organic extract was dried (Na2S04) and evaporated under reduced pressure to give the product as viscus oil (27 g).
IR (neat) 3400, 2957, 1513, 1425, 1260, 1134, 1029, 806 cm-1
!H NMR (300 MHz, CDC13) δ 1.60-1.93 (m, 8 H), 2.19 (brs, 1 H), 3.80 (s, 3 H), 4.59
(d, J= 6.1 Hz, 2 H), 4.72-4.80 (m, 1 H), 6.76-6.92 (m, 3 H).
Step 3: (3-Cyclopentyloxy-4-methoxyphenyl)acetonitrile
Freshly distilled thionyl chloride (42 g, 0.353 mol) was add to a stirred solution of 3- Cyclopentyloxy-4-methoxybenzyl alcohol (26 g, 0.117 mol) in dry 1,2-dichloroetane (260 ml) at RT. The mixture was stined at 70-80 °C for 30 min under nitrogen atmosphere. Excess of SOCl2 and 1,2-dichloroethane were evapourated under reduced pressure to give a viscus residue. The residue was dissolved in dry DMF (260 ml) and NaCN (6.4 g, 0.13 mol) was added as one portion. The heterogeneous mixture was stined at 70-80 °C for 18 h. The reaction mixture was cooled to RT, diluted with ice- cold water (IL) and extracted with ethyl acetate (2 x 300 ml). The combined organic extracts were washed with water (4 x 500 ml) followed by brine (300 ml) and dried over Na2S04. The crude product obtained after evaporation of the solvent was purified by silica gel column chromatography using 10-15 % ethyl acetate in petroleum ether as eluent to give the product (21 g) as colorless oil. IR (neat) 2959, 2249, 1592, 1514, 1428, 1236, 1139, 1029, 997, 805 cm"1; Η NMR (300 MHz, CDC13) δ 1.55-2.00 (m, 8 H), 3.66 (s, 2 H), 3.82 (s, 3 H), 4.74- 4.78 (m, 1 H), 6.78-6.81 (m, 3 H).
Step 4: Dimethyl 4-Cyano-4-(3-cyclopentyloxy-4-methoxyphenly)pimelate
To a stined solution of (3-Cyclopentyloxy-4-methoxyphenyl)acetonitrile (20 g, 0.086 mol) in acetonitrile was added 40 % solution of benzyltrimehtylaminonim hydroxide (3.4 ml, 0.01 M in methanol) and the mixture was heated to reflux. Methylacrylate (75 g, 0.87 mol) in acetonitrile (200 ml) was added and reaction mixture was stined at 60- 70 °C for 6 h. The reaction mixture was cooled to RT, diluted with ethyl acetate (500 ml) and washed with water (3 x 500 ml) followed by brine (200 ml). The organic extract was dried (Na2SO ) and evaporated under reduced pressure to give the crude product. The crude product was purified by silica gel column chromatography using 10-15 % ethyl acetate in petroleum ether as eluent to give 30 g (86 %) of the product as white solid, mp 80-83 °C, IR (neat) 2955, 2236, 1739, 1598, 1514, 1438, 1266, 1026 cm"1; JH NMR (300 MHz, CDC13) δ 1.62-2.00 (m, 8 H), 2.11-2.40 (m, 6 H), 2.46-2.56 (m, 2 H), 3.64 (s, 6 H), 3.86 (s, 3 H), 4.78-4.82 (m, 1 H), 6.84-6.93 (m, 3 H).
Step 5: 2-Carbomethoxy-4-cyano-4-[3-(cyclopentyloxy)-4- methoxyphenyl] cyclohexan- 1 -one
To a stined solution of pimelate ester (22 g, 0.054 mol) in dry DME (150 ml) was added 50 % NaH (4.0 g, 0.083 mol) in dry DME (70 ml). The reaction mixture was heated at 60-70 °C for 3 h. The reaction mixture was quenched with ice-cold 1 N hydrochloric acid (100 ml). The solution was extracted with ethyl acetate (2 x 300 ml) then washed with water (3 x 400 ml) followed by brine (200 ml). The EtOAc extract was dried (Na2S0 ) and evaporated under reduced pressure to give the crude product, which was then purified by silica gel column chromatography using 15-20 % ethyl acetate in petroleum ether as eluent to give 18 g of the product as white solid, IR (neat) 2955, 2235, 1712, 1663, 1516, 1443, 1291, 1164, 808 cm"1; Η NMR (300 MHz, CDC13) δ 1.59-1.99 (m, 8 H), 2.10-2.30 (m, 2 H), 2.41-2.50 (m, 1 H), 2.64 (d, J= 16.2 Hz, 1 H), 2.73-2.85 (m, 1 H), 2.97 (d, J= 16.2 Hz, 1 H), 3.76 (s, 3 H), 3.83 (s, 3 H), 4.75-4.79 (m, 1 H), 6.84 (d, J = 8.1 Hz, 1 H), 6.95-6.98 (m, 2 H), 14.1 (s, 1 H).
Intermediate 2 4-Cyano-4-[3-(cyclopentyloxy)-4-methoxyphenyl]cyclohexan-l-one
A mixture of intermediate 1 (10 g, 0.026 mol) and NaCl (9.1 g, 0.156 mol) in DMSO (100 ml) and water (10 ml) was heated at 150-160 °C for 5 h. The reaction mixture was cooled to RT, diluted with water (300 ml) and extracted with EtOAc (2x250 ml). The combined orgamc extracts were washed with water (3 x 500 ml) followed by brine (200 ml) and dried over Na
2S0 . The crude product obtained after evaporation of the solvent was purified silica gel column chromatography using 10-20 % ethyl acetate in petroleum ether as eluent to give 7 g (83 %) of the product as white solid, mp 110-113 °C; IR (KBr) 2955, 2238, 1717, 1593, 1519, 1427, 1302, 1224, 1144, 1027, 990, 849 cm
"
Η NMR (300 MHz, CDC1
3) δ 1,63-1.97 (m, 8 H), 2.23 (dt, J = 9.9, 3.9 Hz, 2 H), 2.45-2.58 (m, 4 H), 2.90 (dt, J= 9.3, 5.7 Hz, 2 H), 3.84 (s, 3 H), 4.70-4.80 (m, 1 H), 6.85 (d, J= 8.4 Hz, 1 H), 6.97- 6.99 (m, 2 H).
Intermediate 3
2-Carbomethoxy-4-cyano-4-[3-(cyclopentyloxy)-4-difluoromethoxyphenyl]- cyclohexan-1-one
Step 1: 3-Cyclopentyloxy-4-difluoromethoxbenzaldehyde Alkylation of 4-Difluoromethoxy-3-hydroxybenzaldehyde (20 g, 0.1 mol) with cyclopentyl bromide (24 g, 0.16 mol) in the presence of anhydrous potassium carbonate (37 g, 0.268 mol) as described in intermediate 1, step 1 gave 27 g of the product as colourless oil.
IR (neat) 2964, 2875, 1699, 1503, 1434, 1273, 1056, 756 cm"1. Η NMR (300 MHz, CDCI3) δ 1.64-2.02 (m, 8 H), 4.87-4.92 (m, 1 H), 6.63 (t, J =
74.4 Hz, 1 H), 7.28 (d, J= 8.1 Hz, 1 H), 7.39-7.47 (m, 2 H), 9.90 (s, 1 H).
Step 2: 3-Cyclopentyloxy-4-difluoromethoxybenzyl alcohol
The crude alcohol was obtained as described in intermediate 1, step 2, using 3- cyclopentyloxy-4-difluoromethoxybenzaldehyde (27 g, 0.14 mol) and NaBH (4.5 g, 0.118 mol). The product was isolated as colourless oil (26.5 g), IR (neat) 3350, 2962, 1606, 1508, 1429, 1280, 1121, 992, 845, 756 cm _1; !H NMR (300 MHz, CDC13) δ 1.62-1.93 (m, 8 H), 4.65 (d, J = 5.7 Hz, 2 H), 4.79- 4.85 (m, 1 H), 6.51 (t, J= 75.6 Hz, IH), 6.85 (d, J= 8.4 Hz, IH), 6.99 (s, 1 H), 7.11 (d, J= 8.4 Hz, I H).
Step 3: (3-Cyclopentyloxy-4-difluoromethoxyphenyl)acetonitrile The reaction was carried out as described in the case of intermediate 1, step 3 using thionyl chloride (36 g, 0.3 mol) and 3-Cyclopentyloxy-4-difluoromethoxybenzyl alcohol. (26 g, 0.1 mol) in dry 1,2-dichloroetane (260 ml). The crude benzyl chloride obtained was treated NaCN (6.42 g, 0.13 mol) in DMF (260 ml) at 70-80 °C for 16 h. The crude product was purified by silica gel column chromatography using 10 -15 % ethyl acetate in petroleum ether as eluent to give 20 g of the product as coluorless oil. IR (neat) 2963, 2252, 1606, 1509, 1432, 1273, 1122, 1047, 994, 811 cm"1 lB NMR (300 MHz, CDC13) 1.64-1.97 (m, 8 H), 3.74 (s, 2 H), 4.81-4.86 (m, 1 H), 6.53 (t, J= 75 Hz, IH), 6.84 (d, J= 7.8 Hz, 1 H), 6.93 (s, 1 H), 7.14 (d, J= 8.1 Hz, 1 H).
Step 4: Dimethyl 4-Cyano-4-(3-cyclopentyloxy-4-difluoromethoxyphenly)pimelate The diester was prepared as described in the case of intermediate 1, step 4 using 40 % solution of benzyltrimehtylaminonimhydroxide (3.4 ml, 0.01 M), (3-Cyclopentyloxy- 4-difluoromethoxyphenyl)acetonitrile (20 g, 0.08 mol) and methylacrylate (70 g, 0.8 mol) in acetonirle (400 ml). The crude product was purified silica gel column chromatography using 10-15 % ethyl acetate in petroleum ether as eluent to give the product ( 23 g ) as semisolid,
IR (neat): 2956, 2238, 1738, 1605, 1509, 1438, 1270, 1048, 857 cm"1 , 1H NMR (300 MHz, CDCI3) δ 1.57-1.98 (m, 8 H), 2.11-2.57 (m, 8 H), 3.64 (s, 6 H), 4.82-4.87 (m, 1 H), 6.55 (t, J= 75.1 Hz, 1 H), 6.89 (dd, J= 6.0, 2.1 Hz, 1 H), 7.95 (s, l H), 7.17 (d, J= 8.4 Hz, 1 H).
Step 5 : 2-Carbomethoxy-4-cyano-4-[3 -(cyclopentyloxy)-4-difluoromethoxyphenyl] - cyclohexan- 1 -one
This intermediate was prepared as described in intermediate 1, step 5, using Dimethyl 4-cyano-4-(3-cyclopentyloxy-4-difluoromethoxyphenly)pimelate (15 g, 34.16 mmol) and 50 % NaH (2.46 g, 51.3 mol) in dry DME (200 ml). The crude product was then purified by silica gel column chromatography using 15 - 20 % ethyl acetate in petroleum ether as eluent to give the product (12 g ) as white solid, mp 100-102 °C; IR (KBr) 3433, 2959, 2230, 1650, 1521, 1449, 1223, 1028 cm"1;
!H NMR (300 MHz, CDC13) δ 1 ,62- 1.96 (m, 8 H), 2.13-2.31 (m, 2 H), 2.24-2.55 (m, 1 H), 2.65 (d, J= 15.8 Hz, 1 H), 2.76-2.88 (m, 1 H), 2.98 (d, J= 15.9 Hz, 1 H), 3.77 (s, 3 H), 4.81-4.86 (m, 1 H), 6.53 (s, J= 75.3 Hz, 1 H), 6.96 (dd, J= 8.2, 2.1 Hz, 1 H), 7.12 (d, J= 2.1 Hz, 1 H), 7.15 (d, J= 8.3 Hz, 1 H), 12.23 (s, 1 H).
Intermediate 4 4-Cyano-4-[3-(cyclopentyloxy)-4-difluoromethoxyphenyl]cyclohexan-l-one
This intermediate was prepared as described in intermediate 2 using intermediate 3 (6 g, 14.7 mmol) and NaCl (5.5 g, 94.8 mmol) in 5 % aqueous DMSO (100 ml) at 150- 160 °C for 5 h. The crude product was purified by silica gel column chromatography using 10 - 20 % ethyl acetate and petroleum ether as eluent to give the ketone ( 4 g ) as white solid, mp 63-68 °C; IR (KBr) 2961, 2235, 1720, 1609, 1509, 1421, 1269, 1116, 974 cm
"1; H NMR (300 MHz, CDCI
3) δ 1.63-2.01 (m, 8 H), 2.27 (dt, J = 9.9, 3.9 Hz, 2 H), 2.46-2.63 (m, 4 H), 2.92 (dt, J = 6.0, 5.7 Hz, 2 H), 4.83-4.88 (m, 1 H), 6.55 (t, J = 75.1 Hz,
λ l H), 6.99 (dd, J= 8.5, 2.4 Hz, 1 H), 7.14 (d, J= 2.2 Hz, 1 H), 7.19 (d, J = 8.4 Hz, 1 H).
Intermediate 5
4-Cyano-4-[3-(cyclopentyloxy)-4-methoxyphenyl]-2-
[(dimethylamino)methylidine]-cyclohexan-l-one
A solution of intermediate 2 (5.0 g, 15.95 mmol), dimethylformamide dimethyl acetal (11.97 g, 100.5 mmol) and triethylamine (165 mg, 1.63 mmol) in benzene (150 ml) was distilled over a period of 3 h to about one half the original volume. Fresh benzene (100 ml) was added to the reaction mixture and the distillation was continued. The
process was repeated until the reaction showed absence (TLC) of starting ketone. After all the starting material was consumed, the solvent was completely evaporated to give a viscous brown residue. The crude product was purified by silica gel column chromatography using 2 % methanol in chlorofoπn to give 4.6 g (78 %) of the product as off-white solid, mp 138-141 °C;
IR (KBr) 2945, 2223, 1722, 1651, 1551, 1257 cm"1; lR nmr (300 MHz, CDC13) δ 1.60-1.94 (m, 8 H), 2.29-2.34 (m, 2 H), 2.45-2.55 (m, 1 H), 2.70-2.80 (m, 1 H), 3.03 (d, J = 3.0 Hz, 1 H), 3.08 (s, 6 H), 3.31-3.36 (m, 1 H), 3.86 (s, 3 H), 4.60-4.85 (m, 1 H), 6.86 (d, J= 9.0 Hz, 1 H), 7.01 (dd, J= 9.0, 3.0 Hz, 1 H), 7.03 (s, 1 H), 7.61 (s, 1 H).
Intermediate 6:
4-Cyano-4-[3-(cyclopentyloxy)-4-difluoromethoxyphenyl]-2-[(dimethylamino)- methylidine]-cyclohexan-l-one
A solution of intermediate 4 (4.0 g, 11.44 mmol),dimethylformamide dimethyl acetal
(9.5 g, 79.71 mmol), and triethylamine (120 mg, 1.18 mmol),in benzene (150 ml) was distilled over a period of 3 h to about one half the original volume. Fresh benzene
(100 ml) was added to the reaction mixture and the distillation was continued. The process was repeated until the reaction showed absence (TLC) of starting material. After all the starting material was consumed, the solvent was completely evaporated, the residue obtained after removal of solvent was triturated with w-pentane (50 ml) to give 3.1 g (64.7 %) of the product as yellow solid; mp 136-140 °C; IR (KBr); 2949, 2239, 1646, 1547, 1419, 1216, 1125, 1034 cm"1; !H NMR (300 MHz, CDCI3) δ 1.66-1.96 (m, 8 H), 2.33-2.59 (m, 3 H), 2.73-2.82 (m, 1 H), 3.11 (s, 6 H), 3.15-3.38 (m, 2 H), 4.82-4.87 (m, 1 H), 6.55 (t, J= 75.3 Hz, 1 H), 7.02 (dd, J= 6.3, 2.1 Hz, 1 H), 7.14-7.21 (m, 2 H), 7.64 (s, 1 H).
Intermediate 7:
4-Cyano-4-[3-(cyclopentyloxy)-4-methoxyphenyl]cyclohexan-l-one oxime
Hydroxylamine hydrochloride (532 mg, 7.65 mmol) was added to a stined solution of intermediate 2 (2.0 g, 6.38 mmol) in methanol (20 ml) and the mixture was stined at room temperature for 4 h. Methanol was evaporated under reduced pressure and the residue obtained was partitioned between ethyl acetate (200 ml) and water (200 ml). The EtOAc phase was washed with water (3 x 100 ml), brine (50 ml), dried (Na2S0 ) and evaporated. The residue was purified by silica gel column chromatography using 20 % ethyl acetate in petroleum ether as eluent to give 1.7 g (81.3 %) of the product as a viscous liquid; IR (neat) 3400, 2944, 2232, 1605, 1509, 1261, 1055 cm"1;
!H NMR (300 MHz, CDC13) δ 1.59-1.98 (m, 10 H), 2.22-2.35 (m, 3 H), 2.54-2.68 (m, 2 H), 3.48-3.54 ( , 1 H), 3.83 (s, 3 H), 4.76-4.80 (m, 1 H), 6.83 (d, J= 8.1 Hz, 1 H), 6.93-6.97 (m, 2 H), 7.62 (brs, 1 H)
Intermediate 8:
4-Cyano-4-[3-(cyclopentyloxy)-4-difluoromethoxyphenyl]cyclohexan-l-one oxime
Hydroxylamine hydrochloride (520 mg, 7.48 mmol) was added to a stined solution of intermediate 4 (2.0 g, 5.72 mmol) in methanol (20 ml) and the mixture was stined at room temperature for 4 h. Methanol was evaporated under reduced pressure and the residue obtained was partitioned between ethyl acetate (200 ml) and water (200 ml). The EtOAc phase was washed with water (3 x 100 ml), brine (50 ml), dried (Na
2S0 ) and evaporated. The residue was purified by silica gel column chromatography using 20 % ethyl acetate in petroleum ether as eluent to give 1.4 g (73.2 %) of the product as white solid, mp 95-100 °C; IR (KBr) 3271, 2966, 2235, 1604, 1515, 1269, 1119, 1047 cm
"1;
!H NMR (300 MHz, CDC1
3) δ 1.63-1.68 (m, 2 H), 1.71-1.94 (m, 8 H), 1.96-2.06 (m, 1 H), 2.23-2.37 (m, 3 H), 2.58-2.77 (m, 2 H), 4.82-4.87 (m, 1 H), 6.54 (t, J= 75.0 Hz, 1 H), 6.96 (dd, J= 8.7, 2.1 Hz, 1 H), 7.10 (d, J= 2.1 Hz, 1 H), 7.17 (d, J= 8.4 Hz, 1 H), 7.85 (brs, 1 H).
Example 1 5-(3-Cyclopentyloxy-4-methoxyphenyl)-3-oxo-2,3,4,5,6,7-hexahydro-lH- indazole-5-carbonitrile
A mixture of inteπnediate 1 (110 mg, 0.296 mmol), hydrazine hydrate (30 mg, 0.60 mmol) and triethylamine (90 mg, 0.89 mmol) in methanol (10 ml) was stined at 60-70 °C for 45 min. Methanol was evaporated under reduced pressure and the residue was partitioned between EtOAc (50 ml) and water (50 ml). The EtOAc solution was washed with water (3 x 50 ml), brine (50 ml), dried (Na2S04) and evaporated. The residue was triturated with n-pentane to give 90 mg (87.6 %) of the product as white solid, mp 220-225 °C; IR (KBr) 3120, 2958, 2870, 2235, 1605, 1517, 1259, 1145, 1027 cm"1; 1H NMR (300 MHz, DMSO- ) δ 1.72-2.02 (m, 8 H), 2.26-2.34 (m, 3 H), 2.72-3.12 (m, 4 H), 3.85 (s, 3 H), 4.76-4.79 (m, 1 H), 6.84-7.03 (m, 3 H), 11.3 (brs, 1 H).
Example 2
5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-3-oxo-2,3,4,5,6,7-hexahydro-lH- indazole-5-carbonitrile
Reaction of intermediate 3 (200 mg, 0.49 mmol), hydrazine hydrate (45 mg, 0.90 mmol) and triethylamine (120 mg, 1.18 mmol) in methanol (15 ml) as described in example 1 gave 178 mg (93.1 %) of the product as white solid, mp 195-197 °C; IR (KBr) 3117, 2962, 2237, 1606, 1514, 1419, 1271, 1116, 1048, 799 cm
"1;
!H NMR (300 MHz, DMSO-d
6) δ 1.55-1.98 (m, 8 H), 2.23-2.46 (m, 2 H), 2.67-2.96 (m, 4 H), 4.88-4.96 (m, 1 H), 7.00 (t, J = 75.0 Hz, 1 H), 7.13 (d, J = 8.4 Hz, 1 H), 7.19 (s, 1 H), 7.26 (s, 1 H), 9.60 (brs, 1 H), 11.22 (brs, 1 H).
Example 3 5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-lH-5-indazole- carbonitrile
A mixture of intermediate 5 (250 mg, 0.678 mmol) and hydrazine hydrate (50 mg, 0.99 mmol) in ethanol (20 ml) was stined at room temperature for 2 h. The solvent was evaporated under reduced pressure and the residue was dissolved in dichloromethane (50 ml). The dichloromethane solution was washed with water (50 ml), brine (50 ml) and dried (Na2S04). The crude product obtained after evaporation of the solvent was purified by silica gel column chromatography using 30 % acetone in petroleum ether to give 183 mg (80 %) of the product as white solid, mp 66-69 °C; IR (KBr) 3350, 3210, 2936, 2234, 1517, 1258 cm"';
1H nmr (300 MHz, CDC13) δ 1.57-2.01 (m, 8 H), 2.28-2.45 (m, 2 H), 2.86-2.92 (m, 1 H), 3.04-3.10 (m, 2 H), 3.26 (d, J= 9.0 Hz, 1 H), 3.55 (brs, 1 H), 3.86 (s, 3 H), 4.75- 4.80 (m, 1 H), 6.87 (d, J= 8.5 Hz, 1 H), 7.02 (d, J= 8.5 Hz, 1 H), 7.03 (s, 1 H), 7.49 (s, 1 H); m/z 338 (MH+, 100 %), 243 (81).
Example 4 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4,5,6,7-tetrahydro-lH-5- indazolecarbonitrile
Reaction of intermediate 6 (150 mg, 0.370 mmol) and hydrazine hydrate (46 mg, 0.92 mmol) in ethanol (10 ml) as described in example 15 afforded 100 mg (72 %) of the product as white solid; mp 66-69 °C; IR (KBr) 3211, 2961, 2236, 1604, 1512, 11126, 1049 cm"1;
Η NMR (300 MHz, CDC13) δ 1.63-1.96 (m, 8 H), 2.29-2.45 (m, 2 H), 2.87-2.95 (m, 1 H), 3.05-3.30 (m, 3 H), 4.79-4.87 (m, 1 H), 6.55 (t, J= 75.0 Hz, 1 H), 7.00 (dd, J = 8.1, 2.4 Hz, 1 H), 7.14-7.18 (m, 2 H), 7.41 (s, 1 H), 7.80 (brs, 1 H).
Example 5 5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-2-phenyl-4,5,6,7-tetrahydro-2H- indazole-5-carbonitirle
A mixture of intermediate 6 (200 mg, 0.49 mmol) and phenylhydrazine (66 mg, 0.61 mmol) in ethanol (20 ml) was stined at room temperature for 18 h under nitrogen atmosphere. Ethanol was evaporated under reduced pressure and the residue was partitioned between EtOAc (100 ml) and water (100 ml). The organic layer was washed with water (2 x 100 ml), brine (50 ml), dried (Na2S0 ) and evaporated. The crude product obtained after evaporation of the solvent was purified by column chromatography on silica gel using 20 % EtOAc in chloroform to give regioisomeric mixture of products.
The less polar isomer, 30 mg (13.4 %) white solid, mp 158-160 °C; IR (KBr) 3433, 2967, 2234, 1600, 1505, 1383, 1111, 1049 cm-1;
!H NMR (300 MHz, CDC13) δ 1.60-1.90 (m, 8 H), 2.26-2.45 (m, 2 H), 2.82-2.92 (m, 1 H), 3.09-3.33 (m, 3 H), 4.72-4.89 (m, 1 H), 6.50 (t, J= 75.0 Hz, 1 H), 7.01 (d, J = 8.4 Hz, 1 H), 7.14-7.18 (m, 2 H), 7.32-7.53 (m, 6 H).
Example 6
5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-l-phenyl-4,5,6,7-tetrahydro-lH- indazole-5-carbonitirle
The more polar product from the above reaction was characterized as the 1 -phenyl isomer, 7 mg (3.1 %), white solid mp 136- 138 °C;
IR (KBr) 3433, 2967, 2234, 1600, 1504, 1111, 1049 cm"1;
Η NMR (300 MHz, CDCI3) δ 1.62-1.95 (m, 8 H), 2.31-2.49 (m, 2 H), 2.99 (dt, J = 7.8, 4.5 Hz, 1 H), 3.13-3.24 (m, 1 H), 3.13 (d, J= 15.6 Hz, 1 H), 3.32 (d, J= 15.6 Hz, 1 H), 4.75-4.86 (m, 1 H), 6.54 (t, J= 75.0 Hz, 1 H), 7.12 (d, J= 8.4 Hz, 1 H), 7.15- 7.28 (m, 3 H), 7.43 (t, J= 7.5 Hz, 2 H), 7.62 (d, J= 8.4 Hz, 2 H), 7.73 (s, 1 H).
Example 7 5-(3-CyclopentyIoxy-4-methoxyphenyl)-2-(2-pyridyl)-4,5,6,7-tetrahydro-2H-5- indazolecarbonitrile
2-Hydrazinopyridine (38 mg, 0.35 mmol) was added to a stined solution of intermediate 5 (100 mg, 0.27 mmol) in dry ethanol (5 ml) and the reaction mixture was stined at room temperature for 8 h under nitrogen. The mixture was diluted with ethyl acetate (100 ml) and washed with water (3 x 50 ml) followed by brine (50 ml) and dried (Na2S04). The crude product obtained after evaporation of solvent was purified by chromatography on silica gel using 10 % ethyl acetate in petroleum ether as eluent to gave 20 mg (17.7 %) of the product as white solid, mp 134-137 °C; IR (KBr) cm"1; 2960, 2237, 1605, 1427, 1354, 1235, 1134, 1054, 954 cm"1 !H NMR (300 MHz, CDC13) δ 1.66-1.93 (m, 8 H), 2.29-2.50 (m, 2 H), 3.18 (dd, J = 16.2, 4.5 Hz, 2 H), 3.50-3.60 (m, 2 H), 3.86 (s, 3 H), 4.76-4.82 (m, 1 H), 6.87 (d, J =
8.4 Hz, 1 H), 7.01-7.04 (m, 2 H), 7.16 (t, J= 5.1 Hz, 1 H), 7.54 (s, 1 H), 7.80 (t, J =
7.5 Hz, 1 H), 7.93 (d, J= 8.1 Hz, 1 H), 8.38 (d, J= 3.9 Hz, 1 H).
Example 8
5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-methyl-4,5,6,7-tetrahydro-lH-5- indazolecarbonitrile 8A & 5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-methyl- 4,5,6,7-tetrahydro-2H-5-indazolecarbonitrile 8B
To a stined and cooled (0 °C) suspension of 50 % sodium hydride (36 mg, 0.750 mmol) in THF (10 ml) was added compound of example 3 (200 mg, 0.594 mmol) and the mixture was stined at the same temperature to result a brown solution. Methyl iodide (120 mg, 0.845 mmol) was then added and the mixture was stined at 0 °C for 2 h. The mixture was quenched with ice cold water (50 ml) and extracted with EtOAc
(2 x 50 ml). The ethyl acetate solution was washed with water (2 x 50 ml), brine (50 ml) and dried (Na2S0 ). The residue obtained after evaporation of the solvent was characterized as 1: 1 mixture (HPLC) of regioisomers 8 A and 8B. The isomers were separated by preparative HPLC to give 8 A (30 mg) and 8B (25 mg) as white solids. Isomer A : mp 145-148 °C; IR(KBr) 2958, 2233, 1518, 1417, 1263, 1146, 1016 cm"1; lR NMR (300 MHz, CDC13) δ 1.78-2.00 (m, 8 H), 2.25-2.46 (m, 2 H), 2.80-2.90 (m, 1 H), 2.98-3.12 (m, 2 H), 3.15-3.25 (m, 1 H), 3.85 (s, 3 H), 3.86 (s, 3 H), 4.78-4.81 (m, 1 H), 6.82 (d, J= 8.4 Hz, 1 H), 6.91-7.08 (m, 2 H), 7.20 (s, 1 H). Isomer B: mp 158-162 °C;
IR (KBr) 2935, 2234, 1519, 1419, 1267, 1149, 1016 cm"1;
Η NMR (300 MHz, CDC13) δ 1.79-2.00 (m, 8 H), 2.23-2.42 (m, 2 H), 2.68-2.78 (m, 1 H), 2.89-3.05 (m, 2 H), 3.16-3.24 (m, 1 H), 3.77 (s, 3 H), 3.85 (s, 3H), 4.74-7.78 (m, 1 H), 6.86 (d, J= 8.7 Hz, 1 H), 6.99-7.02 (m, 2 H), 7.31 (s, 1 H).
Example 9
2-[5-cyano-5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-lH-l- indazoyl] acetic acid 9A & 2-[5-cyano-5-(3-Cyclopentyloxy-4-methoxyphenyl)- 4,5,6,7-tetrahydro-2H-2-indazoyl] acetic acid 9B
Alkylation of example 3 (250 mg, 0.743 mmol) with ethyl bromoacetate (160 mg, 0.958 mmol) in presence of 50 % sodium hydride (42 mg, 0.875 mmol) in DMF (10 ml) as described in example 8 gave 203 mg (64.5 %) of the product as viscous liquid. The crude product was hydrolysed with 2 N aqueous KOH (2 ml) in EtOH (5 ml) at room temperature for 1 h. Work-up and chromatography of the crude product on silica gel gave 183 mg (90 %) of the product as a mixture (1:1) of regioisomers 9A and 9B. The mixture was showed the following analysis: mp 115-118 °C; IR (KBr) 3435, 2956, 2235, 1737, 1517, 1257, 1027, cm
"1;
1H NMR (300 MHz, CDC1
3) δ 1.58-2.00 (m, 8 H), 2.30-2.45 (m, 1 H), 2.74-3.25 (m, 5 H), 3.84 (s, 3 H), 4.75-5.04 (m, 3 H), 3.84 (d, J= 8.4 Hz, 1 H), 6.97-7.01 (m, 2 H), 7.26 (s, 1 H).
Example 10 5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydrobenzo[c]isoxazole-5- carbonitrile
Hydroxyilamine hydrochloride (57 mg, 0.82 mmol) was added to a stined solution of sodium ethoxide (58 mg, 0.852 mmol) in ethanol (10 ml) to result a homogeneous solution. Intermediate 5 (200 mg, 0.542 mmol) in ethanol (5 ml) was added and the mixture was stined at room temperature for 2h. The solvent was evaporated under reduced pressure and the residue was taken up in EtOAc (50 ml) and washed with water (2 x 25 ml) and brine (25 ml). Evaporation of the solvent under reduced pressure furnished a viscous brown liquid, which on purification on silica gel column using 2 % methanol in chloroform afforded 115 mg (63 %) of the product as white solid, mp 60-62 °C(dec);
IR (KBr) 2959, 2871, 2236, 1518, 1260, 1146 cm -1; Η NMR (300 MHz, CDC13) δ 1.60-2.04 (m, 8 H), 2.14-2.18 (m, 1 H), 2.43-2.63 (m, 4 H), 3.33 (dd, J= 12.0, 6.9 Hz, 1 H), 3.83 (s, 3 H), 4.62-4.80 (m, 1 H), 6.82 (d, J = 8.1 Hz, 1 H), 6.90 (d, J= 8.1 Hz, 1 H), 6.91 (s, 1 H), 7.48 (s, 1 H).
Example 11
5-(3-Cyclopentyloxy-4-methoxyphenyl)-3-methyl-2-oxo-2,3,4,5,6,7-hexahydro- benzo[rf][l,3]oxazole-5-carbonitrile.
A mixture of oxime 5 (600 mg, 1.628 mmol), dimethyl carbonate (3.0 g, 33.303 mmol) and potassium carbonate (450 mg, 3.255 mmol) was heated at 180-200 °C in a sealed tube for 2 days. Excess dimethyl carbonate was evaporated under reduced pressure and the residue was partitioned between EtOAc (100 ml) and water (100 ml). The layers were separated. The EtOAc layer was washed with water (2 x 100 ml), brine (50 ml) and dried (Na2S04). The crude product obtained after evaporation of the solvent was purified by chromatography on silica gel using 20 % EtOAc in chloroform to give 250 mg (37 %) of the product as yellow solid, mp 154-157 °C; IR (KBr) 3426, 2959, 2237, 1751, 1600, 1514, 1248, 1145 cm"1; !H NMR (300 MHz, CDC13) δ 1.57-1.67 (m, 2 H), 1.78-2.05 (m, 6 H), 2.21-2.39 (m, 2 H), 2.49-2.56 (m, 1 H), 2.56-2.78 (m, 1 H), 2.90-3.09 (m, 2 H), 3.19 (s, 3 H), 3.86 (s, 3 H), 4.78-4.83 (m, 1 H), 6.88 (dd, J= 7.2, 1.2 Hz, 1 H), 6.96 (d, J= 2.4 Hz, 1 H), 6.99 (d, J= 7.2 Hz, 1 H).
Example 12 5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-vinyI-4,5,6,7-tetrahydro-lH-5-indole- carbonitrile
The oxime intermediate 7 (1.0 g, 3.04 mmol) in dry DMSO (30 ml) containing KOH (110 mg, 1.96 mmol) was saturated with acetylene gas at 0-5 °C and the mixture was heated in a sealed tube at 70-80 °C for 24 h. The dark brown mixture was cooled to
room temperature and diluted with ethyl acetate (300 ml) and water (300 ml). The layers were separated and the organic layer was washed with water (3 x 300 ml) and brine (100 ml). The crude product obtained after evaporation of the solvent was purified by silica gel column chromatography using 10 % ethyl acetate in petroleum ether to give 400 mg ( 36.2 %) of the product as white solid; mp 128-131 °C; IR (KBr) 2959, 2843, 2235, 1641, 1589, 1518, 1484, 1253, 1146, 986, 703 cm
"1; Η NMR (300 MHz, CDC1
3) δ 1.57-1.95 (m, 8 H), 2.25-2.38 (m, 2 H), 2.70 (dt, J = 6.0, 4.5 Hz, 1 H), 2.90-2.99 (m, 1 H), 3.09 (dd, J= 17.5, 15.9 Hz, 2 H), 3.83 (s, 3 H), 4.63 (dd, J= 8.7, 0.9 Hz, 1 H), 4.71-4.76 (m, 1 H), 5.04 (dd, J= 15.6, 0.9 Hz, 1 H), 6.03 (d, J= 2.7 Hz, 1 H), 6.74 (dd, J= 15.6, 9.3 Hz, 1 H), 6.83 (dd, J= 6.9, 2.4 Hz, 1 H), 6.91 (d, J= 2.7 Hz, 1 H), 7.01 (d, J= 7.2 Hz, 1 H), 7.02 (d, J= 2.4 Hz, 1 H).
Example 13
5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-l-vinyI-4,5,6,7-tetrahydro-liϊ"-5- indolecarbonitrile
Reaction of oxime intermediate 8 (500 mg, 1.372 mmol) with acetylene gas in presence of potassium hydroxide (60 mg, 1.069 mmol) in DMSO (10 ml) was stined at 60-70°C 24hrs in sealed tube under acetylene gas, then cooled to RT and diluted with water (100ml) then extracted with ethyl acetate (2 X 100ml). The combined ethyl acetate solution was washed with water (3 x 300 ml) followed by brine (100 ml) dried over Na2SO4. and evoparatation of ethyl acetate to give crude. This crude was purified by 60-120mesh silica gel column chromatography using 10% ethyl acetate in pet ether as solvent to give 40 mg white solid; mp 99-104 °C; IR (KBr) 3433, 2940, 2847, 2235, 1645, 1511, 1420, 1288, 1080 cm"1; !H NMR (300 MHz, CDC13) δ 1.61-1.69 (m, 2 H), 1.77-1.95 (m, 6 H), 2.28-2.45 (m, 2 H), 2.70-2.78 (m, 1 H), 2.89-2.91 (m, 1 H), 3.11 (dd, J= 17.4, 15.9 Hz, 2 H), 4.66 (dd, J= 8.8, 1.0 Hz, 1 H), 4.78-4.81 (m, 1 H), 5.05 (dd, J= 15.4, 1.0 Hz, 1 H), 6.07
(d, J= 2.8 Hz, 1 H), 6.55 (t, J= 75.3 Hz, 1 H), 6.79 (dd, J= 15.4, 8.9 Hz, 1 H), 6.94 (d, J= 2.8 Hz, 1 H), 7.01 (dd, J= 8.4, 2.4 Hz, 1 H), 7.02-7.17 (m, 2 H).
Example 14
5-(3-Cyclopentyloxy-4-methoxyphenyl)-4,5,6,7-tetrahydro-lH-5-indole- carbonitrile
To a well-stiπed solution of 1-vinyl indole 12 (100 mg, 0.275 mmol) in THF-water (1:1) mixture (10 ml) was added mercuric acetate (96 mg, 0.301 mmol) and the mixture was stined at room temperature for 2h to result a white precipitate. The mixture was treated with sodium borohydride (22 mg, 0.581 mmol) and stined at room temperature for 10 min. The mixture was filtered through a celite bed to remove mercury salts. The filtrate was diluted with water and extracted with ethyl acetate (2 x 50 ml). The combined ethyl acetate extracts were washed with water (2 x 100 ml) and brine (50 ml). The EtOAc solution was dried (Na2S04) and evaporated under reduced pressure to give a brown residue. Purification of the product using silica gel chromatography using 20 % ethyl acetate in petroleum ether gave 65 mg (70 %) of the product as low melting solid.
IR (neat) 3320, 2958, 2929, 2855, 2236, 1605, 1511, 1270, 1049 cm"1; 1H NMR (300 MHz, CDC13) δ 1.57-1.95 (m, 8 H), 2.30-2.36 (m, 2 H), 2.69 (dt, J = 12.0, 4.5 Hz, 1 H), 2.92-3.04 (m, 1 H), 3.13 (d, J= 15.6 Hz, 1 H), 3.19 (d, 15.6 Hz, 1 H), 3.85 (s, 3 H), 4.72-4.77 (m, 1 H), 6.01 (t, J= 2.4 Hz, 1 H), 6.68 (t, J= 2.4 Hz, 1 H), 6.84 (d, J= 8.7 Hz, 1 H), 7.01-7.04 (m, 2 H), 7.86 (brs, 1 H)
Example 15
5-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4,5,6,7-tetrahydro-lH-5- indazole-carbonitrile
Reaction of oxime intermediate 8 (500 mg, 1.372 mmol) with acetylene gas in presence of potassium hydroxide (60 mg, 1.069 mmol) in DMSO (10 ml) was stined at 60-70°C 24hrs in sealed tube under acetylene gas, then cooled to RT and diluted with water (100ml) then extracted with ethyl acetate (2 X 100ml) then combined ethyl acetate solution was washed with water (3 X 300ml) followed by brine (100ml) dried over Na2S0 and evoparatation of ethyl acetate to give crude. This crud was purified by 60-120mesh silica gel column chromatography using 10% ethyl acetate in pet ether as solvent to give lOOmg as semi solid.
IR (neat) 3329, 2959, 2929, 2236, 1511, 1270, 1049 cm"1;
Η NMR (300 MHz, CDC13) δ 1.63-1.95 (m, 8 H), 2.28-2.37 (m, 2 H), 2.65-2.74 (m, 1 H), 2.90-3.04 (m, 1 H), 3.08 (d, J= 15.9 Hz, 1 H), 3.19 (d, J= 15.7 Hz, 1 H), 4.76- 4.81 (m, 1 H), 6.03 (t, J= 2.4 Hz, 1 H), 6.54 (t, J= 75.6 Hz, 1 H), 6.70 (t, J= 2.4 Hz, 1 H), 7.02 (dd, J= 8.1, 2.4 Hz, 1 H), 7.10-7.17 (m, 2 H), 7.87 (brs, 1 H).
Example 16
5-(3-Cyclopentyloxy-4-methoxyphenyl)-l-methyl-4,5,6,7-tetrahydro-lH-5-indole- carbonitrile
To a stined and cooled (0 °C) suspension of 50 % sodium hydride (8 mg, 0.166 mmol) in THF (5 ml) was added compound of example 14 (50 mg, 0.148 mmol) and the mixture was stined at the sane temperature to result a brown solution. Methyl
iodide (30 mg, 0.211 mmol) was then added and the mixture was stined at 0 °C for 2 h. The mixture was quenched with ice cold water (25 ml) and extracted with EtOAc (2 x 50 ml). The ethyl acetate solution was washed with water (2 x 50 ml), brine (50 ml) and dried (Na
2S0
4). The residue obtained after evaporation of the solvent was purified by column chromatography on silica gel using 1 % methanol in chloroform to give 40 mg (76.8 %) of the product as white solid, mp 142-145 °C; IR (KBr) 3433, 2936, 2841, 2234, 1519, 1266, 1148, 1031 cm
"1; *H NMR (300 MHz, CDC1
3) δ 1.59-1.93 (m, 8 H), 2.28-2.37 (m, 2 H), 2.62-2.68 (m, 1 H), 2.86-3.20 (m, 1 H), 3.05 (d, J= 15.6 Hz, 1 H), 3.06 (d, J= 15.6 Hz, 1 H), 3.50 (s, 3 H), 3.84 (s, 3 H), 4.72-4.76 (m, 1 H), 5.91 (d, J= 3.0 Hz, 1 H), 6.52 (d, J= 2.4 Hz, 1 H), 6.83 (d, J= 9.3 Hz, 1 H), 7.01-7.04 (m, 2 H).
Example 17
Ethyl 6-C ethyI-5,6,7,8- tetrahydr
A mixture of intermediate 5 (300 mg, 0.814 mmol), ethylacetoacetate (127 mg, 0.971 mmol) and ammonium acetate (500 mg, 6.486 mmol) in acetic acid (10 ml) was stined at 100-110 °C for 3 h under nitrogen atmosphere. The mixture was cooled to room temperature and diluted with water (100 ml). The mixture was extracted with ethyl acetate (2 x 100 ml), washed with water (2 x 100 ml) and dried over anhydrous Na2S04 and concentrated. The residue was purified by silica gel column chromatography using 25 % ethyl acetate in petroleum ether to give 200 mg (56.4 %) of the product as colorless liquid, IR (Neat) 2957, 2230, 1722, 1517, 1260, 1146, 1021, 781 cm "1; Η NMR (300 MHz, CDC13) δ 1.41 (t, J= 7.2 Hz, 3 H), 1.59-1.97 (m, 8 H), 2.32-2.43 (m, 1 H), 2.53-2.55 (m, 1 H), 2.81 (s, 3 H), 3.08-3.16 (m, 1 H), 3.25-3.41 (m, 3 H), 3.86 (s, 3 H), 4.37 (q, J= 7.2 Hz, 2 H), 4.76-4.79 (m, 1 H), 6.86 (d, J= 8.4 Hz, 1 H), 6.96-7.02 (m, 2 H), 7.96 (s, 1 H).
Example 18 6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-2-methyl-5,6,7,8-tetrahydro-3- quinolinecarboxylic acid
To a stined solution of ester 17 (120 mg, 0.276 mmol) in ethanol (5 ml), was added 2N aqueous NaOH solution (5 ml) and the mixture was stined at room temperature for 16 h. Most of the solvent was evaporated under reduced pressure and the residue was diluted with water (15 ml). The basic aqueous solution was washed with ether (2 x 25 ml) and then acidified with 1 N HCI solution to pH 2. The free acid was extracted into ethyl acetate and washed with water (50 ml). The organic extract was dried (Νa2S0 ) and evaporated give 89 mg (79.2 %) the product as white solid, mp. 155-185 °C;
IR (KBr) 3419, 2961, 2230, 1712, 1519, 1418, 1264, 1167, 1028, 974, 761 cm"1; Η NMR (300 MHz, CDC13) δ 1.59-1.99 (m, 8 H), 2.34-2.44 (m, 1 H), 2.50-2.57 (m, 1 H), 2.87 (s, 3 H), 3.15-3.42 (m, 4 H), 3.85 (s, 3 H), 4.75-4.79 ( , 1 H), 6.85 (d, J = 8.1 Hz, 1 H), 6.96-7.02 (m, 2 H), 8.10 (s, 1 H).
Example 19
6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazoline-carbonitrile
To a mixture of intermediate 1 (1.0 g, 2.692 mmol), formamidine acetate (980 mg, 9.413 mmol) and sodium methoxide (645 mg, 11.944 mmol) in methanol (30 ml) was stined at 60-70 °C for 18 h under nitrogen. Most of the methanol was evaporated under reduced pressure and the residue was diluted with EtOAc (100 ml) and water (100 ml). The layers were separated and the organic layer was washed with water (3 x 50 ml), dried (Na2S04) and evaporated. The residue was purified by silica gel column chromatography using 2 % methanol in chloroform as eluent to afford 550 mg (55.9 %) of the product as white solid; mp 190-195 °C; IR (KBr) 3137, 2922, 2233, 1652, 1639, 1619, 1522, 1418, 1276, 1154 1002 cm"1;
Η NMR (300 MHz, CDC13) δ 1.63-1.97 (m, 8 H), 2.23-2.31 (m, 1 H), 2.04-2.46 (m, 1 H), 2.84-2.93 (overlapping m, 1 H), 2.89 (d, J= 17.8 Hz, 1 H), 3.10-3.24 (m, 1 H), 3.34 (d, J= 17.9 Hz, 1 H), 3.85 (s, 3 H), 4.73-4.82 (m, 1 H), 6.86 (d, J= 7.8 Hz, 1 H), 6.97-7.02 (m, 2 H), 8.06 (s,l H), 12.79 (brs, 1 H).
Example 20
6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazolinecarbonitrile
This was prepared as described in example 19 using intermediate 3 (200 mg, 0.490 mmol) and formamidine acetate (127 mg, 1.219 mmol) in presence of sodium methoxide (100 mg, 1.851 mmol). The product was purified by silica gel column chromatography using 1 % methanol in chloroform to afford 60 mg (30.4 %) of the product as white solid; mp 110-115 °C;
IR (KBr) 3154, 2963, 2237, 1655, 1614, 1514, 1420, 1270, 1124, 1046, 997 cm -' 1. !H NMR (300 MHz, CDC13) δl.61-1.97 (m, 8 H), 2.23-2.33 (m, 1 H), 2.41-2.48 (m, 1 H), 2.86-2.96 (m, 2 H), 3.14-3.25 (m, 1 H), 3.36 (d, J= 17.4 Hz, 1 H), 4.82-4.89 (m, 1 H), 6.55 (t, J= 75.0 Hz, 1 H), 6.99 (dd, J = 8.1, 2.1 Hz, 1 H), 7.15 (d, J = 2.3 Hz, 1 H), 7.18 (d, J= 8.4 Hz, 1 H), 8.05 (s, 1 H), 12.41 (brs, 1 H).
Example 21
6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazoline-carbonitrile
This was prepared as described in example 19 using intermediate 1 (200 mg, 0.538 mmol) and acetamidine hydrochloride (127 mg, 1.343 mmol) in presence of sodium methoxide (100 mg, 1.851 mmol). The product was purified by crystallization from
EtOAc-hexane to give 125 mg (61.1 %) of the product as white solid; mp 208-210 °C; IR (KBr) 3432, 2916, 2235, 1651, 1518, 1257, 1147, 1026 cm"1;
!H NMR (300 MHz, DMSO-rf6) δ 1.62-1.97 (m, 8 H), 2.19-2.29 (m, 1 H), 2.37-2.44 (m, 1 H), 2.45 (s, 3 H), 2.76-2.87 (m, 1 H), 2.86 (d, /= 16.0 Hz, 1 H), 3.05-3.14 (m, 1 H), 3.31 (d, J = 16.8 Hz, 1 H), 3.85 (s, 3 H), 4.78-4.83 (m, 1 H), 6.86 (d, J = 8.4 Hz, 1 H), 6.99-7.03 (m, 2 H), 12.63 (brs, 1 H).
Example 22
6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-2-methyl-4-oxo-3,4,5,6,7,8- hexahydro-6-quinazolinecarbonitrile
This was prepared as described in example 19 using intermediate 3 (100 mg, 0.245 mmol) and acetamidine hydrochloride (28 mg, 0.296 mmol)) in presence of triethylamine (50 mg, 0.495 mmol). The crude product was purified by crystahzation from EtOAc-hexane to give 90 mg (88.2 %) of the product as white solid; mp 188-
189 °C;
IR (KBr) 2962, 2238, 1647, 1609, 1515, 1420, 1270, 1117, 1044, 852 cm"1;
Η NMR (300 MHz, CDC13) δ 1.65-1.95 (m, 8 H), 2.20-2.43 (m, 2 H), 2.45 (s, 3 H), 2.84 (overlapping d, J= 17.1 Hz, 2 H), 3.00-3.19 (m,l H), 3.32 (d, J= 17.1 Hz, 1 H),
4.80-4.89 (m, 1 H), 6.53 (t, J= 75.3 Hz, 1 H), 6.99 (d, J= 8.4 Hz, 1 H), 7.14-7.18 (m,
2 H), 12.57 (s, 1 H).
Example 23
2-Amino-6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazolinecarbonitrile
This was prepared as described in example 19 using intermediate 1 (200 mg, 0.538 mmol) and guanidine hydrochloride (128 mg, 1.339 mmol)) in presence of sodium methoxide (100 mg, 1.851 mmol). The crude product was purified by crystallization from EtOAc-hexane to give 80 mg (39 %) of the product as white solid; mp 310-315 °C; IR (KBr) 3330, 2959, 2236, 1654, 1516, 1267, 1144, 1028 cm-1;
!H NMR (300 MHz, DMSO-J6) δ 1.55-1.90 (m, 8 H), 2.24-2.46 (m, 3 H), 2.59-2.68 (m, 1 H), 2.69 (d, J= 16.8 Hz, 1 H), 2.91 (d, J= 16.7 Hz, 1 H), 3.74 (s, 3 H), 4.75- 4.85 (m, 1 H), 3.38 (brs, 2 H), 6.94-7.03 ( , 3 H), 10.88 (brs, 1 H)
Example 24
2-Amino-6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-4-oxo-3,4,5,6,7,8- hexahydro-6-quinazolinecarbomtrile
This was prepared as described in example 19 using intermediate 3 (100 mg, 0.245 mmol) and guanidine hydrochloride (59 mg, 0.617 mmol)) in presence of sodium methoxide (45 mg, 0.833 mmol). The crude product was purified by crystallization from EtOAc-hexane to give 45 mg (44 %) of the product as white solid; mp 315-320 °C;
IR (KBr) 3331, 3098, 2968, 2347, 1654, 1495, 1170, 1019 cm"1; Η NMR (300 MHz, DMSO- ) δ 1.65-1.94 (m, 8 H), 2.23-2.72 (m, 4 H), 2.73 (d, J = 15.9 Hz, 1 H), 2.95 (d, J= 15.9 Hz, 1 H), 4.90-4.96 (m, 1 H), 6.31 (brs, 2 H), 7.00 (t, J= 74.1 Hz, 1 H), 7.10 (d, J= 8.4 Hz, 1 H), 7.19-7.22 (m, 2 H), 10.87 (brs, 1 H).
Example 25 6-(3-Cyclopentyloxy-4-methoxyphenyl)-3-methyl-4-oxo-3,4,5,6,7,8-hexahydro-6- quinazolinecarbonitrile
To a well-stined slurry of compound 19 (150 mg, 0.410 mmol) and cesium carbonate (150 mg, 0.460 mmol) in dry DMF (10 ml), was added iodomethane (76 mg, 0.535 mmol) and the mixture was stined at room temperature for 1 h under nitrogen. Then reaction was quenched with ice-cold water (100 ml) then extracted with ethyl acetate (100 ml). The organic layer was washed with water (3 x 100 ml), brine (50 ml) and dried (Na2S04). The residue obtained after evaporation of the solvent was purified by
silica gel column chromatography using 2 % methanol in chloroform as eluent to give 100 mg (86.3 %) of the product as white solid; mp 59-63 °C; IR (KBr) 2957, 2234, 1661, 1552, 1517, 1417, 1259, 1165, 1027, 1003, 770 cm"1; 'H NMR (300 MHz, CDC13) δ 1.63-1.92 (m, 8 H), 2.16-2.26 (m, 1 H), 2.37-2.44 (m, j 1 H), 2.79-2.95 (m, 2 H), 3.06-3.15 (m, 1 H), 3.34 (d, J= 17.7 Hz, 1 H), 3.52 (s, 3 H), 3.85 (s, 3 H), 4.77-4.80 (m, 1 H), 6.85 (d, J= 8.1 Hz, 1 H), 6.97-7.00 (m, 2 H), 8.01 (s, 1 H).
Example 26
Ethyl 2-[6-cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-4-oxo-3,4,5,6,7,8-
Alkylation reaction was canied out as described in example 25 using of compound 19
(300 mg, 0.820 mmol) with ethyl bromoacetate (151 mg, 0.904 mmol) and cesium carbonate (348 mg, 1.068 mmol) in dry DMF (10 ml) at room temperature. The product was purified by silica gel column chromatography using 40 % EtOAc in chloroform to afford afforded 250 mg (67.4 %) of the product as white solid; mp 55-
64 °C;
IR (KBr) 2959, 2235, 1749, 1667, 1613, 1518, 1417, 1374, 1258, 1145, 1026 cm"1;
Η NMR (300 MHz, CDCI3) δ 1.31 (t, J= 6.6 Hz, 3 H), 1.61-1.96 (m, 8 H), 2.21-2.27 5 (m, 1 H), 2.37-2.44 (m, 1 H), 2.80-2.93 (d and m, 2 H), 3.03-3.21 (m, 1 H), 3.33 (d, J
= 18.0 Hz, 1 H), 3.84 (s, 3 H), 4.26 (q, J = 7.2 Hz, 2 H), 4.45 (d, J = 17.1 Hz, 1 H),
4.75 (d, J= 17.0 Hz, 1 H), 4.76-4.78 (m, 1 H), 6.84 (d, J= 8.7 Hz, 1 H), 6.96-6.99 (m,
2 H), 7.95 (s, 1 H).
0
Example 27
2-[6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-4-oxo-3,4,5,6,7,8-hexahydro- 3-quinazolinyl] acetic acid
To a stined solution of compound 26 (170 mg, 0.376 mmol) in ethanol (5 ml) was added 1.0 N aqueous sodium hydroxide solution (3.0 ml) and the mixture was stined at room temperature for 4 h. The reaction mixture was diluted with water (20 ml) and washed with 3 x 25 ml diethyl ether. The aqueous layer was acidified with IN aqueous HCI to pH 3 and extracted with ethyl acetate (2 x 50 ml). The combined extracts were washed with water (3 x 50 ml), brine (50 ml) and dried (Νa2S0 ). The residue obtained after evaporation of the solvent was triturated with ether to give 100 mg (62.7 %) of the product as white solid; mp 134-136 °C; IR (KBr) 3444, 2958, 2235, 1736, 1666, 1518, 1418, 1257, 1145, 1026 cm"';
Η NMR (300 MHz, CDC13) δ 1.61-1.92 (m, 8 H), 2.19-2.25 (m, 1 H), 2.28-2.44 (m,
1 H), 2.80-3.22 (m, 3 H), 3.33 (d, J= 16.0 Hz, 1 H), 3.84 (s, 3 H), 4.55 (d, J= 17.1 Hz, 1 H), 4.74-4.76 (m, 2 H), 5.15 (brs, 1 H), 6.85 (d, J= 8.1 Hz, 1 H), 6.96-6.99 (m,
2 H), 8.20 (s, 1 H)
Example 28 Ethyl 2-[6-cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-2-methyl-4-oxo- 3,4,5,6,7,8-hexahydro-3-quinazolinyl] acetate (28A)& Ethyl 2-[6-cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-2-methyl-5,6,7,8- tetrahydro-4-quinazolinyloxy] acetate (28B)
Alkylation reaction was canied out as described in example 19 using of compound 21 (300 mg, 0.790 mmol) with ethyl bromoacetate (146 mg, 0.874 mmol) and cesium carbonate (310 mg, 0.951 mmol) in dry DMF (10ml) at room temperature. The reaction gives a mixture of N-alkylated and O-alkylated products 28A and 28B respectively.
Silica gel column chromatography using 50 % EtOAc in chloroform afforded 70 mg
(19 %) of the N-alkylated product 28A as viscous liquid;
IR (KBr) 2960, 2235, 1749, 1667, 1555, 1419, 1258, 1024, 777 cm"1;
'H NMR (300 MHz, CDC13) δl.31 (t, J= 6.9 Hz, 3 H), 1.58-1.96 (m, 8 H), 2.19-2.41 (m, 2 H), 2.46 (s, 3 H), 2.71-2.82 (m, 1 H), 2.88 (d, J= 17.4 Hz, 1 H), 3.05-3.16 (m, 1 H), 3.28 (d, J = 17.4 Hz, 1 H), 3.84 (s, 3 H), 4.26 (q, J = 6.9 Hz, 2 H), 4.60 (d, J = 17.1 Hz, 1 H), 4.76-4.80 (m, 1 H), 4.95 (d, J= 17.4 Hz, 1 H), 6.84 (d, J= 8.7 Hz, 1 H), 6.96-6.99 (m, 2 H). Further elution with 60 % EtOAc in chloroform afforded 96 mg (26 %) of O-alkylated product 28B as a semisolid,
IR (KBr) 2960, 2235, 1749, 1667, 1517, 1419, 1145, 1024, 756 cm"1; Η NMR (300 MHz, CDCI3) δ 1.31 (t, J = 6.9 Hz, 3 H), 1.58-1.96 (m, 8 H), 2.19- 2.41 (m, 2 H), 2.46 (s, 3 H), 2.71-2.82 (m, 1 H), 2.88 (d, J= 17.4 Hz, 1 H), 3.05-3.16 (m, 1 H), 3.28 (d, J= 17 A Hz, 1 H), 3.84 (s, 3 H), 4.23 (q, J= 6.9 Hz, 2 H), 4.79 (d, J = 15.6 Hz, 1 H), 4.77-4.81 ( , 1 H), 5.02 (d, J= 15.6 Hz, 1 H), 6.45 (d, J= 7.8 Hz, 1 H), 6.97-7.00 (m, 2 H).
Example 29
6-(3-Cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
,A solution of intermediate 5 (250 mg, 0.678 mmol) and formamidine acetate (100 mg, 0.960 mmol) in dry ethanol (10 ml) was stined at 70-80 °C for 2 h. The solvent was evaporated under reduced pressure and the residue was diluted with EtOAc (100 ml) and water (100 ml). The layers were separated and the organic layer was washed with water (3 x 50 ml), brine (50 ml), dried (Na2S04) and evaporated. The residue was purified by column chromatography on silica gel using 1 % methanol in chloroform to afford 80 mg (33.7 %) of the product as viscous oil; IR (neat) 3366, 2957, 2233, 1646, 1517, 1239, 754 cm"1; 1H NMR (300 MHz, CDC1 ) δ 1.60-1.97 (m, 8 H), 2.32-2.71 (m, 2 H), 3.08-3.49 (m, 4 H), 3.86 (s, 3 H), 4.76-4.80 (m, 1 H), 6.87 (d, J= 8.1 Hz, 1 H), 6.86-7.01 (m, 2 H), 8.49 (s, 1 H), 9.03 (s, 1 H).
Example 29A
6-(3-Cyclopentyloxy-4-methoxyphenyl)-l-oxo-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
To a stined solution of compound 29 (100 mg, 0.286 mmol) in chloroform (10 ml) was added 50 % m-CPBA (250 mg, 0.724 mmol) and the mixture was stined at room temperature for 5 h. The mixture was diluted with chloroform (50 ml) and washed with IN aqueous sodium hydroxide solution. The chloroform layer was washed with water (2x100 ml), brine (50 ml) and dried (Νa
2S0 ). The crude product obtained after evaporation of the solvent was purified by silica gel column chromatography using 1- 2 % methanol in chloroform as eluent to give 30 mg of the product as white solid, mp 120-123 °C;
IR (KBr) 2959, 2236, 1518, 1417, 1382, 1266, 1145 cm
"1;
1H nmr (300 MHz, CDC13) δ 1.80-1.94 (m, 8 H), 2.28-2.39 (m, 1 H), 2.56-2.63 (m, 1 H), 3.19-3.43 (m, 4 H), 3.87 (s, 3 H), 4.78-4.81 (m, 1 H), 6.89 (d, J= 8.4 Hz, 1 H), 6.95 (dd, J= 8.7, 2.4 Hz, 1 H), 7.01 (d, J= 2.4 Hz, 1 H), 8.06 (s, 1 H), 8.96 (s, 1 H). 1H NMR (300 MHz, CDC13) δ 1.60-1.97 (m, 8 H), 2.32-2.71 (m, 2 H), 3.08-3.49 (m, 4 H), 3.86 (s, 3 H), 4.76-4.80 (m, 1 H), 6.87 (d, J= 8.1 Hz, 1 H), 6.86-7.01 (m, 2 H), 8.49 (s, 1 H), 9.03 (s, 1 H).
Example 30
6-(3-Cyclopentyloxy-4-difluoromethoxyphenyl)-5,6,7,8-tetrahydro-6-quinazoline- carbonitrile.
This product was prepared as described in example 29 using of intermediate 6 (200 mg, 0.494 mmol) and formamidine acetate (150 mg, 1.44 mmol) to give 20 mg (10.4
%) of the product as viscous oil;
1H NMR (300 MHz, CDCI3) δl.63-1.96 (m, 8 H), 2.33-2.52 (m, 2 H), 3.09-3.40 (m, 4 H), 4.80-4.85 (m, 1 H), 6.54 (t, J= 75.2 Hz, 1 H), 6.97 (dd, J= 8.0, 2.1 Hz, 1 H),
7.13 (d, J= 1.8 Hz, 1 H), 7.19 (d, J= 8.4 Hz, 1 H), 8.49 (s, 1 H), 9.04 (s, 1 H).
Example 31
6-(3-Cyclopentyloxy-4-methoxyphenyl)-2-methyI-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
This product was prepared as described in example 29 using of intermediate 5 (100 mg, 0.271 mmol), acetamidine hydrochloride (31 mg, 0.327 mmol) and sodium methoxide (18 mg, 0.333 mmol) in methanol (10 ml) at 60-70 °C for 10 h. The product was purified by silica gel column chromatography using 2 % methanol in chloroform to afford afforded 80 mg (81.1 %) of the product as white solid; mp 133- 136 °C;
IR (KBr) 3430, 2953, 2233, 1583, 1517,1 443, 1246, 802 cm"1; !H NMR (300 MHz, CDC13) δ 1.60-1.99 (m, 8 H), 2.31-2.55 (m, 2 H), 2.71 (s, 3 H), 3.01-3.36 (m, 4 H), 3.86 (s, 3 H), 4.75-4.79 (m, 1 H), 6.87 (d, J= 9.0 Hz, 1 H), 6.97- 7.02 (m, 2 H), 8.04 (s, 1 H).
Example 32
2-Amino-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
This product was prepared as described in example 29 using of intermediate 5 (200 mg, 0.542 mmol), guanidine hydrochloride (68 mg, 0.711 mmol) and sodium ethoxide (50 mg, 0.734 mmol) in ethanol (10 ml) at 70-80 °C for 1 h. The mixture was cooled to room temperature and the solid precipitated was collected by filtration. The crude product was crystallized from ethanol to give 167 mg (85 %) of the product as white solid, mp 215-219 °C;
IR(KBr) 3316, 3163, 2957, 2237, 1667, 1523, 1276, 1144 cm"1;
lH NMR (300 MHz, CDC13) δ 1.59-1.97 (m, 8 H), 2.26-2.46 (m, 2 H), 2.82-2.90 (m, 1 H), 3.01-3.23 (m, 3 H), 3.85 (s, 3 H), 4.76-4.79 (m, 1 H), 4.95 (brs, 2 H), 6.85 (d, J = 8.4 Hz, 1 H), 6.96 (dd, J= 8.4, 2.1 Hz, 1 H), 7.01 (s, 1 H), 8.06 (s, 1 H); m/z 365 (MH+, 100 %), 297 (40).
Example 33 2-Amino-6-(3-cyclopentyloxy-4-difluoromethoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
This product was prepared as described in example 29 using of intermediate 6 (200 mg, 0.494 mmol), guanidine hydrochloride (63 mg, 0.659 mmol) and sodium methoxide (36 mg, 0.666 mmol) in methanol (10 ml) at 70-80 °C for 1 h. The mixture was cooled to room temperature and the solid precipitated was collected by filtration. The crude product was crystallized from ethanol to give 110 mg (61 %) of the product as white solid, mp 123-126 °C; IR (KBr) 3323, 3179, 2961, 2235, 1600, 1514, 1269, 1127, 1047 cm"1;
!H NMR (300 MHz, CDCI3) δ 1.76-1.97 (m, 8 H), 2.27-2.49 (m, 2 H), 2.85-2.95 (m, 1 H), 3.05-3.26 (m, 3 H), 4.82-4.86 (m, 1 H), 5.01 (brs, 2 H), 6.55 (t, J= 75.0 Hz, 1 H), 6.98 (dd, J= 8.7, 2.4 Hz, 1 H), 7.14 (d, J= 2.1 Hz, 1 H), 7.19 (d, J= 8.7 Hz, 1 H), 8.08 (s, 1 H).
Example 34
Nl-[6-Cyano-6-(3-Cyclopentyloxy-4-methoxyphenyI)-5,6,7,8-tetrahydro-2- quinazolinyl] -acetamide
To a stined solution of compound 32 (100 mg, 0.274 mmol) and triethylamine (86 mg, 0.851 mmol) in dichloromethane (10 ml) was added acetyl chloride (42 mg, 0.535 mmol) and the mixture was refluxed for 3 h. The reaction mixture was cooled to room temperature and diluted with 100 ml dichloromethane. The dichloromethane solution was washed with water (3 x 50 ml), brine (50 ml) and dried (Na S0 ). The residue obtained after evaporation of the solvent was purified by chromatography on silica gel using 2 % methanol in chloroform to give 30 mg (26.8 %) of the product as white solid, mp 105-108 °C; IR (KBr) 3427, 2958, 2233, 1727, 1561, 1580, 1419, 1369, 1255, 1147, 1027 cm"1; rH NMR (300 MHz, CDC13) δ 1.58-1.90 (m, 8 H), 2.31 (s, 3 H), 2.36-2.52 (m, 2 H), 3.15-3.46 (m, 4 H), 3.86 (s, 3 H), 4.75-4.85 (m, 1 H), 6.89 (d, J= 8.4 Hz, 1 H), 6.98- 7.02 (m, 2 H), 8.58 (s, 1 H)
Example 35
2-[6-Cyano-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-2- quinazolinylamino] -acetic acid
This product was prepared as described in example 29 using intermediate 5 (100 mg, 0.271 mmol), guanidine acetic acid (32 mg, 0.35 mmol) and sodium methoxide (55 mg, 1.018 mmol) in methanol (10 ml) at 60-70 °C for 8 h. The product was extracted into EtOAc after acidifying the mixture with 0.1 N aqueous HCI to pH 2. The crude product was crystallised from hexane to give 60 mg (52.3 %) of the product as off- white solid; mp 85-90 °C;
IR (KBr) 3306, 2951, 2234, 1724, 1602, 1517, 1471, 1257, 1144, 1021 cm"1;
Η NMR (300 MHz, CDC13) δ 1.61-1.98 (m, 8 H), 2.30-2.51 (m, 2 H), 2.58-3.28 (m,
4 H), 3.81 (s, 3 H), 2.26 (d, J= 3.3 Hz, 2 H), 4.80 (d, J= 3.3 Hz, 1 H), 6.87 (d, J= 8.4
Hz, 1 H), 6.97-7.02 (m, 2 H), 8.00 (brs, 1 H), 8.09 (s, 1 H).
Example 36
2-Methylthio-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile
This product was prepared as described in example 29 using intermediate 5 (500 mg,
1.356 mmol), S-methylthiouranium sulphate (454 mg, 1.6 mmol) and sodium methoxide (88 mg, 1.629mmol) in methanol (20 ml) at 60-70 °C for 3 h. The crude product was purified by chromatography on silica gel using 2 % methanol in chloroform to give 250 mg (46.5%) of the product as white solid; mp 125-128 °C;
IR (KBr) 3433, 2963, 2238, 1573, 1541, 1412, 1229, 1143, 803 cm"l;
Η NMR (300 MHz, CDC13) δ 1.57-1.99 (m, 8 H), 2.29-2.54 (m, 2 H), 3.09 (s, 3 H), 3.02-3.31 (m, 4 H), 3.85 (s, 3 H), 4.74-4.80 (m, 1 H), 6.85 (d, J= 8.5 Hz, 1 H), 6.94-
7.03 (m, 2 H), 8.27 (s, 1 H).
Example 37
2-MethanesulfonyI-6-(3-cycIopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile
To a stined and cooled (0 °C) solution of compound 36 (100 mg, 0.252 mmol) in dichloromethane (25 ml) was added m-CPBA (173 mg, 1.002 mmol) and the mixture was stined at the same temperature for 2 h. The mixture was treated with 10 % aqueous Na2S03 solution (5 ml) and filtered. The filtrate containing the product was washed with 10 % aqueous NaHC03 solution (2 x 25 ml), water (25 ml) and brine (25 ml) and dried. The crude product obtained after evaporation of the solvent was purified by chromatography on silica gel to give 40 mg (37 %) of the product as white solid; mp 75-78 °C; IR (KBr) 3436, 2960, 2235, 2186, 1714, 1518, 1315, 1145, 778 cm"1; 'H NMR (300 MHz, CDC13) δ 1.56-1.99 (m, 8 H), 2.34-2.61 (m, 2 H), 3.17-3.49 (m, 4 H), 3.34 (s, 3 H), 3.85 (s, 3 H), 4.75-4.79 (m, 1 H), 6.85 (d, J= 8.7 Hz, 1 H), 6.92-' 7.00 (m, 2 H), 8.63 (s, 1 H).
Example 38
6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-methoxy-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile
Freshly distilled phosphorus oxychloride (10 ml) was added to compound 19 (200 mg, 0.547 mmol) and the mixture is stined at 105-110 °C for 30 min under nitrogen. Excess POCI3 was removed under reduced pressure then dried under high vacuum to give 250 mg of 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-chloro-5,6,7,8-tetrahydro- 6-quinazo-linecarbonitrile as a brown residue. This above residue was dissolved in methanol (5 ml) and treated with potassium methoxide (115 mg, 1.639 mmol) and stined at room temperature for 1 h. The reaction mixture was diluted with EtOAc
(100 ml), washed with water (3 x 100 ml), brine (50 ml) and dried (Na2S0 ). The residue obtained after evaporation of the solvent was purified by chromatography on silica gel using 20 % EtOAc in chloroform to give 40 mg (19 %) of the product as a low-melting solid;
IR (KBr) 3415, 2957, 2235,1708, 1580, 1469, 1258, 1014 cm-1;
!H NMR (300 MHz, CDC13) δ 1.59-1.66 (m, 2 H), 1.77-1.98 (m, 6 H), 2.27-2.48 (m,
2 H), 2.96-3.05 (m, 1 H), 2.98 (d, J= 18.4 Hz, 1 H), 3.21-3.30 (m, 1 H), 3.34 (d, J =
18.6 Hz, 1 H), 3.86 (s, 3 H), 4.01 (s, 3 H), 4.75-4.81 (m, 1 H), 6.88 (d, J= 8.4 Hz, 1
H), 6.97-7.03 (m, 2 H), 8.62 (s, 1 H).
Example 39
6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-methylamino-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
Freshly distilled phosphorus oxychloride (10 ml) was added to compound 19 (200 mg, 0.547 mmol) and the mixture is stined at 105-110 °C for 30 min under nitrogen.
Excess POCI3 was removed under reduced pressure then dried under high vacuum to give 250 mg of 6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-chloro-5,6,7,8-tetrahydro- 6-quinazo-line-carbonitrile as a brown residue. This intermediate was used as such for the next step without further purification.
The above residue was treated with ethanohc methyl amine solution (8 ml, 15 %) and the mixture was stined at 10-15 °C for 2 h. The mixture was diluted with water (200 ml), extracted with ethyl acetate (2 x 50 ml), washed with water (3 x 50 ml), brine (30 ml) and dried (Na2S0 ). The residue obtained after evaporation of the solvent was purified by chromatography on silica gel using 2 % methanol in chloroform as eluent to give 50 mg (24.1 %) of the product as white solid, mp 149-152 °C; IR (KBr) 3378, 2958, 2234, 1591, 1518, 1387, 1264, 1146, 1034, 805 cm"1; 1HNMR (300 MHz, CDCI3) δ 1.58-1.95 (m, 8 H), 2.25-2.42 (m, 2 H), 2.77 (d, J= 16.5 H, 1 H), 2.86-2.89 (m, 1 H), 2.93 (d, J= 16.2 Hz, 1 H), 3.10-3.18 (m, 1 H), 3.04 (d, 4.8 Hz, 3 H), 3.10-3.18 (m, 1 H), 3.84 (s, 3 H), 4.55 (brs, 1 H), 4.73-4.77 (m, 1 H), 6.84 (d, J= 8.4 Hz, 1 H,), 6.94-6.97 (m, 2 H), 8.49 (s, 1 H).
Example 40
6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-dimethylamino-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
The 4- chloro derivative was prepared as example 39 from pyrimidinone 19 (200 mg,
0.547 mmol) was treated with dimethylamine (54 mg, 1.20 mmol) in methanol (10 ml) and the mixture was stined at room temperature for 6 h. Work-up and purification of the as described in example 24 gave 105 mg (48.8 %) of the product as off-white solid, mp 52-57 °C;
IR (KBr) 3363, 2928, 2233, 1746, 1571, 1398, 1144, 1022 cm"1; lH NMR (300 MHz, CDC13) δ 1.75-1.98 (m, 8 H), 2.35-2.47 (m, 2 H), 2.92-3.06 (m, 2 H), 3.01 (s, 6 H), 3.10-3.25 (m, 2 H), 3.85 (s, 3 H), 4.70-4.75 (m, 1 H), 6.85 (d, J = 8.4 Hz, 1 H), 6.90-7.00 (m, 2 H), 8.51 (s, 1 H).
Example 41
4-«-Butylamino-6-(3-cyclopentyloxy-4-methoxyphenyl)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
The 4-chloro derivative was prepared as example 39 from pyrimidinone 19 (200 mg, 0.547 mmol) was treated with «-butylamine (400 mg, 5.479 mmol) in ethanol (10 ml) and the mixture was stined at room temperature for 18 h. Work-up and purification of the crude material by chromatography on silica gel using 30 % ethyl acetate in chloroform gave 75 mg (32.5 %) of the product as off-white solid; mp 122-128 °C; IR (KBr) 3398, 2959, 2230, 1590, 1518, 1414, 1256, 1146, 1024, 774 cm"1; Η NMR (300 MHz, CDC13) δ 0.95 (t, J= 7.2 Hz, 3 H), 1.40 (q, J = 8.1 Hz, 2 H), 1.55-1.67 (m, 4 H), 1.76-1.95 (m, 6 H), 2.26-2.43 (m, 2 H), 2.85 (dd, J= 17.1, 5.8 Hz, 2 H), 2.82-2.87 (m, 1 H), 3.10-3.22 (m, 1 H), 3.46-3.53 (m, 2 H), 3.85 (s, 3 H), 4.41 (t, J= 5.1 Hz, 1 H), 4.74-4.78 (m, 1 H), 6.86 (d, J= 8.4 Hz, 1 H), 6.97-7.00 (m, 2 H), 8.47 (s, 1 H).
Example 42
6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-morpholino-5,6,7,8-tetrahydro-6- quinazoline-carbonitrile
The 4- chloro derivative was prepared as described in example 39 from pyrimidinone 19 (150 mg, 0.410 mmol) was treated with morpholine (5.0 ml) and the mixture was stined at room temperature for 4 h. Work-up and purification of the crude material by chromatography on silica gel using 2 % methanol in chloroform gave 110 mg (61.6 %) of the product as off-white solid; mp 55-59 °C; IR (KBr) 2959, 1566, 1518, 1427, 1258, 1116, 105, 995, 855 cm
"1;
!H NMR (300 MHz, CDC1
3) δ 1.59-1.93 (m, 8 H), 2.36-2.54 (m, 2 H), 2.95-3.01 (m, 4 H), 3.25-3.42 (m, 4 H), 3.74-3.83 (m, 4 H), 3.85 (s, 3 H), 4.72-4.75 (m, 1 H), 6.86 (d, J= 8.4 Hz, 1 H), 6.88-6.97 (m, 2 H), 8.16 (s, 1 H).
Example 43
6-(3-Cyclopentyloxy-4-methoxyphenyl)-4-phenylamino-5,6,7,8,-tetrahydro-6- qumazolinecarbonitile
The 4- chloro derivative was prepared as described in example 39 from pyrimidinone 19 (150 mg, 0.410 mmol) was treated with aniline (382 mg, 4.107 mmol) and the mixture was stined at room temperature for 18 h. Work-up and purification of the crude material by chromatography on silica gel using 15 % EtOAc in chloroform gave 30 mg (16.5 %) of the product as off-white solid; mp 192-198 °C; IR (KBr) 3381, 2962, 2236, 1607, 1574, 1517, 1443, 1256, 1146, 1016, 752 cm"1; 'HNMR (300 MHz, CDCI3) δl.66-1.96 (m, 8 H), 2.34-2.45 (m, 2 H), 2.93-3.26 (m, 4 H), 3.86 (s, 3 H), 4.76-4.78 (m, 1 H), 6.27 (brs, 1 H), 6.88 (d, J= 8.4 Hz, 1 H), 7.01- 7.03 (m, 2 H), 7.12 (t, J= 7.5 Hz, 1 H), 7.35 (t, J= 7.5 Hz, 2 H), 7.53 (d, J= 7.8 Hz, 2 H), 8.57 (s, 1 H).
Example 44
4-Benzylamino-6-(3-cyclopentyloxy-4-methoxyphenyI)-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
The 4-chloro derivative was prepared as described in example 39 from pyrimidinone
19 (250 mg, 0.684 mmol) was treated with benzylamine (1.3 g, 12.149 mmol) and the mixture was stined 80-90 °C for 18 h. Work-up and purification of the crude material by chromatography on silica gel using 15 % EtOAc in chloroform gave 100 mg (32.1
%) of the product as off-white solid; mp 65-70 °C;
IR (KBr) 3391, 2957, 1590, 1517, 1453, 1355, 1259, 1146, 1022, 700 cm"1; 'H NMR
(300 MHz, CDC13) δl.68-2.04 (m, 8 H), 2.39-2.59 (m, 2 H), 2.86-3.07 (m, 3 H), 3.26- 3.39 (m, 1 H), 3.84 (s, 3 H), 4.80 (s, 2 H), 4.82-4.98 (m, 2 H), 6.64 (d, J= 8.4 Hz, 1
H), 7.05-7.08 (m, 2 H), 7.38-7.45 (m, 5 H), 8.65 (s, 1 H).
Example 45
6-(3-CycIopentyloxy-4-methoxyphenyl)-4-phenethyIamino-5,6,7,8-tetrahydro-6- quinazolinecarbonitrile
The 4-chloro derivative was prepared as described in example 39 from pyrimidinone 19 (150 mg, 0.410 mmol) was treated with 2-phenylethylamine (1.0 g, 8.264 mmol) and the mixture was refluxed at 80-90 °C for 18 h. Work-up and purification of the crude material by chromatography on silica gel using 25 % EtOAc in chloroform gave 60 mg (39.6 %) of the product as off-white solid; mp 110-120 °C; IR (KBr) 3283, 1961, 2237, 1595, 1518, 1262, 1023, 701 cm"1;
■H NMR (300 MHz, CDC13) δ 1.62-1.95 ( , 8 H), 2.23-2.43 (m, 2 H), 2.62-2.95 ( , 5 H), 3.10-3.21 (m, 1 H), 3.70-3.85 (m, 2 H), 3.84 (s, 3 H), 4.49 (brs, 1 H), 4.72-4.78 (m, 1 H), 6.83-6.94 (m, 3 H), 7.15-7.32 (m, 5 H), 8.50 (s, 1 H).
The present invention provides a novel series of heterocyclic compounds having potential therapeutic activity and medical use against several allergic disorders, particularly in asthma.
In vitro Studies
Inhibition of Phosphodiesterase Enzymes (PDE4)
In this assay, PDE4 enzyme converts [ H] cAMP to the conesponding [3H] 5'- AMP in proportion to the amount of PDE4 present. The [3H] 5'- AMP then was quantitatively converted to free [3H] adenosine and phosphate by the action of snake venom 5'-nucleotidase. Hence, the amount of [3H] adenosine liberated is proportional to PDE4 activity.
The assay was performed with modification of the method of Thompson and Appleman (Biochemistry; 1971; 10; 311-316) and Schwartz and Passoneau (Proc. Natl. Acad. Sci. U.S.A. 1974; 71; 3844-3848), both references incorporated herein by reference in their entirety, at 34°C. In a 200 μl total reaction mixture, the reaction mixture contained 12.5mM of Tris, 5 mM MgCl2, lμM cAMP (cold) and 3H cAMP (0.1 μCi), (Amersham). Stock solutions of the compounds to be investigated were prepared in DMSO in concentrations such that the DMSO content in the test samples did not exceed 0.05 % by volume to avoid affecting the PDE4 activity. Drug samples were then added in the reaction mixture (25 μl/tube). The assay was initiated by addition of enzyme mix (75 μL) and the mixture was incubated for 20 minutes at 34° C. Then the reaction was stopped by boiling the tubes for 2 mins at 100°C in a water bath. After cooling on ice for 5 minutes and addition of 50 ug/reaction of 5'- nucleotidase snake venom from Crotalus atrox incubation was carried out again for 20 min. at 34°C. The unreacted substrate was separated from (3H) Adenosine by addition ofDowex AG lX-8
( Biorad Lab), (400 ul) which was prequilibrated (1:1:1) in water and ethanol. Reaction mixture was then thoroughly mixed, placed on ice for 15 minutes, vortexed
004/016596 and centrifuged at 14,000 r.p.m. for 2 mins. After centrifugation, a sample of the supernatant was taken and added in 24 well optiplates containing Scintillant (1 ml) and mixed well. The samples in the plates were then determined for radioactivity in a Top Counter and the PDE4 activity was calculated. PDE4 enzyme was present in quantities that yield <30% total hydrolysis of substrate (linear assay conditions).
Additionally, activity of the compounds were tested against other Phosphodiesterase enzymes, namely, PDEl(Ca.sup.2+/calmodulin-deρendent), PDE2(cGP-stimulated), PDE3 (cGP-inhibited), PDE5 (cGP-specific) and PDE6 (cGP-specifϊc, photoreceptor).
Results were expressed as percent inhibition (IC5o) in nM concentrations. The IC50 values were determined from the concentration curves by nonlinear regression analysis.