WO2002089729A2 - Fused heterocyclic compounds - Google Patents
Fused heterocyclic compounds Download PDFInfo
- Publication number
- WO2002089729A2 WO2002089729A2 PCT/US2002/013856 US0213856W WO02089729A2 WO 2002089729 A2 WO2002089729 A2 WO 2002089729A2 US 0213856 W US0213856 W US 0213856W WO 02089729 A2 WO02089729 A2 WO 02089729A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- group
- aryl
- hydrogen
- compound
- Prior art date
Links
- 0 CC(C(*)*(C[N+])*(*)C1*)=C1C=C Chemical compound CC(C(*)*(C[N+])*(*)C1*)=C1C=C 0.000 description 4
- NVZWEEGUWXZOKI-UHFFFAOYSA-N Cc1c(C=C)cccc1 Chemical compound Cc1c(C=C)cccc1 NVZWEEGUWXZOKI-UHFFFAOYSA-N 0.000 description 1
- WIAPFHBDMSYODW-UHFFFAOYSA-N Cc1c(C=C=C)cccc1 Chemical compound Cc1c(C=C=C)cccc1 WIAPFHBDMSYODW-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/14—Prodigestives, e.g. acids, enzymes, appetite stimulants, antidyspeptics, tonics, antiflatulents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present invention relates to compounds and compositions useful in the treatment of conditions and disorders associated with eating behavior, energy homeostasis and anxiety.
- G-protein coupled receptors play important roles in diverse signaling processes, including those involved with sensory and hormonal signal transduction. Eating disorders, which represent a major health concern throughout the world, have been linked to GPCR regulation. On the one hand, disorders such as obesity, the excess deposition of fat in the subcutaneous tissues, manifest themselves by an increase in body weight. Individuals who are obese often have, or are susceptible to, medical abnormalities including respiratory difficulties, cardiovascular disease, diabetes and hypertension. On the other hand, disorders like cachexia, the general lack of nutrition and wasting associated with chronic disease and/or emotional disturbance, are associated with a decrease in body weight.
- MCH neuropeptide melanin-concentrating hormone
- GPCR G-protein-coupled-receptor
- MCH receptor melanin- concentrating hormone receptor
- MCHR melanin- concentrating hormone receptor
- mice deficient in MCH are leaner and have a decreased food intake relative to normal mice.
- MCH is believed to exert its activity by binding to MCHR, resulting in the mobilization of intracellular calcium and a concomitant reduction in cAMP levels (see Chambers et al., Nature 400:261-65 (1999); Shimada et al. Nature 396:670-74 (1998)).
- MCH also activates inwardly rectifying potassium channels, and MCHR has been found to interact with both G ⁇ i protein and G ⁇ q protein (Saito et al., TEM ll(8):299-303 (2000)).
- MCHR tissue localization of MCHR indicates that it is expressed in those regions of the brain involved in olfactory learning and reinforcement.
- modulators of MCHR should have an effect on neuronal regulation of food intake (see Saito et al, Nature 400:265-69 (1999)).
- MCH has been shown to modulate behaviors other than feeding, such as anxiety (Gonzales et al. (1996) Peptides 17:171-177; Monzon et al. (1999) Physiol. Behav. 67:813-817).
- MCHR modulators are useful for the study of physiological processes mediated by MCHR and the development of therapeutic agents for the treatment of conditions and disorders associated with weight regulation, learning, anxiety and other neuronal-related functions.
- the present invention provides fused heterocyclic compounds and compositions, and methods of use thereof to treat or prevent conditions and disorders mediated by MCHR.
- the present invention provides compounds, compositions and methods for treating conditions and disorders associated with eating behavior, energy homeostasis and anxiety.
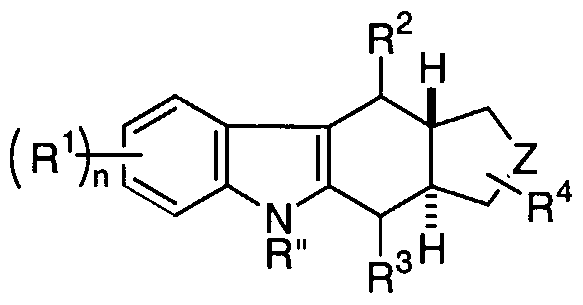
- the compounds of the invention have the formula (I):
- a and B are independently selected from the group consisting of CR' and N, wherein R' is selected from the group consisting of hydrogen, (C 1 -C 5 )alkyl, arylalkyl, -C(O)R 7 , -CO 2 R 8 and -C(O)NR 5 R 6 ;
- Z is selected from the group consisting of -N(R)-, -N(R)-(Cr C 3 )alkylene- and -(Cj-C 3 )alkylene -N(R)-(C ⁇ -C 3 )alkylene-, wherein R is selected from the group consisting of hydrogen, (C]-C 7 )alkyl, heterocycloalkyl(C 1 -C 7 )alkyl, aryl, arylalkyl, -C(O)R 7 , -CO 2 R 8 , -C(O)NR 5 R 6 , -S(O) m NR 5 R 6 and-S(O) m R 7 ; each R 1 is independently selected from the group consisting of hydrogen, halogen, (C ⁇ -C 5 )alkyl, ⁇ erfluoro(C 1 -C 5 )alkyl, -OR'", -SR'", aryl, arylalkyl, - NO 2
- R 4 is selected from the group consisting of hydrogen -OR'", -C(O)R 7 , - CO 2 R 8 , -C(O)NR 5 R 6 , -CN, (C C 5 )alkyl and aryl, wherein R'" is selected from the group consisting of hydrogen, (C 1 -C 5 )alkyl, aryl and aryl(C ⁇ -C 5 )alkyl;
- R 5 and R 6 are independently selected from the group consisting of hydrogen, alkyl, aryl and arylalkyl or combined to form a 4-, 5-, 6-, 7- or 8-membered ring containing from one to three heteroatoms;
- R 7 and R 8 are independently selected from the group consisting of hydrogen, alkyl, aryl and arylalkyl;
- R 9 is selected from the group consisting of alkyl, aryl and arylalkyl;
- the subscript m is an integer from 1 to 2;
- the subscript n is an integer from 0 to 8;
- ⁇ - ⁇ represents a single or fused aryl or heteroaryl ring; with the proviso that R 2 is not hydrogen when
- ⁇ " ⁇ is benzene, A and B are both CH, V is a bond, W is -N(R")- and Z is -NR-CH 2 -.
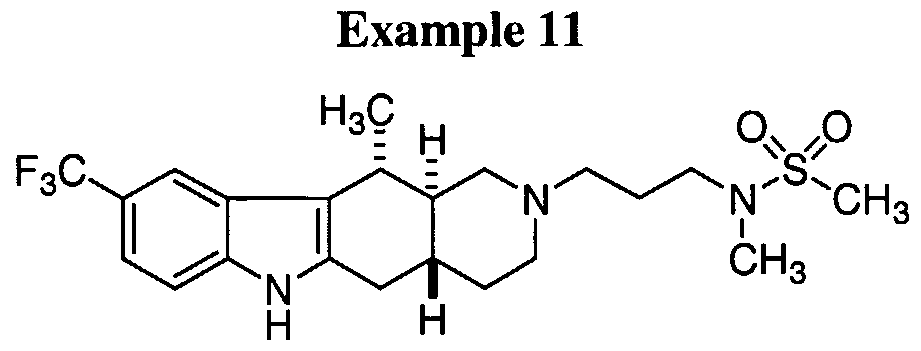
- FIG. 1 provides the structures of exemplary compounds of formula I.
- MCHR refers to the melanin-concentrating hormone receptor protein 1 (MCHRl), unless otherwise stated.
- treat refers to a method of alleviating or abrogating a disease and/or its attendant symptoms.
- MCHR-mediated condition or disorder refers to a condition or disorder characterized by inappropriate, e.g., less than or greater than normal, MCHR activity.
- An MCHR-mediated condition or disorder may be completely or partially mediated by inappropriate MCHR activity.
- an MCHR-mediated condition or disorder is one in which modulation of MCHR results in some effect on the underlying condition or disease (e.g., an MCHR antagonist results in some improvement in patient well-being in at least some patients).
- Exemplary MCHR-mediated conditions and disorders include obesity, eating disorders and other behavioral disorders, such as anxiety disorders and mood disorders.
- therapeutically effective amount refers to that amount of the compound being administered sufficient to prevent development of or alleviate to some extent one or more of the symptoms of the condition or disorder being treated.
- the term "obesity” refers to the excessive accumulation of body fat. Obesity may have genetic, environmental (e.g., expending less energy than is consumed) and regulatory determinants. Cardiovascular disorders, lipid disorders and metabolic disorders, such as hypertension, hyperlidemia, coronary artery disease and diabetes, are commonly associated with obesity.
- eating disorder refers to an emotional and/or behavioral disturbance associated with an excessive decrease in body weight and/or inappropriate efforts to avoid weight gain, e.g., fasting, self-induced vomiting, laxative or diuretic abuse. Depression is commonly associated with eating disorders. Exemplary eating disorders include anorexia nervosa and bulimia.
- anxiety disorder refers to an emotional and/or behavioral disturbance characterized by persistent and pervasive worry or restlessness, tension or irritability about, e.g., health, work, money or family, for no clear reason.
- An anxiety disorder may be accompanied by tachycardia or dyspnea.
- Exemplary anxiety disorders include anxiety, generalized anxiety disorder, panic attacks, panic disorder and obsessive-compulsive disorder (OCD).
- OCD obsessive-compulsive disorder
- miod disorder refers to an emotional and/or behavioral disturbance characterized by persistent and pervasive bouts of euphoria and/or depression.
- mood disorders include depression and bipolar disorders. Anxiety is frequently associated with mood disorders, such as depression.
- modulate refers to the ability of a compound to increase or decrease the function, or activity, of MCHR. Modulation, as described herein, includes the inhibition or activation of MCHR, either directly or indirectly.
- Inhibitors are compounds that, e.g., bind to, partially or totally block stimulation, decrease, prevent, delay activation, inactivate, desensitize, or down-regulate signal transduction, e.g., antagonists.
- Activators are compounds that, e.g., bind to, stimulate, increase, open, activate, facilitate, enhance activation, sensitize or up-regulate signal transduction, e.g., agonists.
- alkyl by itself or as part of another substituent, means, unless otherwise stated, a straight or branched chain, or cyclic hydrocarbon radical, or combination thereof, which may be fully saturated, mono- or polyunsaturated and can include di- and multi-valent radicals, having the number of carbon atoms designated (i.e., C o means one to ten carbons).
- saturated hydrocarbon radicals include groups such as methyl, ethyl, n-propyl, isopropyl, n-butyl, t-butyl, isobutyl, sec- butyl, cyclohexyl, (cyclohexyl)methyl, cyclopropylmethyl, homologs and isomers of, for example, n-pentyl, n-hexyl, n-heptyl, n-octyl, and the like.
- An unsaturated alkyl group is one having one or more double bonds or triple bonds.
- unsaturated alkyl groups include vinyl, 2-propenyl, crotyl, 2-isopentenyl, 2- (butadienyl), 2,4-pentadienyl, 3-(l,4-pentadienyl), ethynyl, 1- and 3-propynyl, 3- butynyl, and the higher homologs and isomers.
- alkyl unless otherwise noted, is also meant to include those derivatives of alkyl defined in more detail below as “heteroalkyl,” “cycloalkyl” and “alkylene.”
- alkylene by itself or as part of another substituent means a divalent radical derived from an alkane, as exemplified by -CH 2 CH 2 CH 2 CH 2 -.
- an alkyl group will have from 1 to 24 carbon atoms, with those groups having 10 or fewer carbon atoms being preferred in the present invention.
- a “lower alkyl” or “lower alkylene” is a shorter chain alkyl or alkylene group, generally having eight or fewer carbon atoms.
- heteroalkyl by itself or in combination with another term, means, unless otherwise stated, a stable straight or branched chain, or cyclic hydrocarbon radical, or combinations thereof, consisting of the stated number of carbon atoms and from one to three heteroatoms selected from the group consisting of O, N, Si and S, and wherein the nitrogen and sulfur atoms may optionally be oxidized and the nitrogen heteroatom may optionally be quaternized.
- the heteroatom(s) O, N and S may be placed at any interior position of the heteroalkyl group.
- the heteroatom Si may be placed at any position of the heteroalkyl group, including the position at which the alkyl group is attached to the remainder of the molecule.
- heteroalkyl Up to two heteroatoms may be consecutive, such as, for example, -CH 2 -NH-OCH 3 and -CH 2 -O-Si(CH 3 ) 3 .
- heteroalkyl also included in the term “heteroalkyl” are those radicals described in more detail below as “heteroalkylene” and “heterocycloalkyl.”
- the term “heteroalkylene” by itself or as part of another substituent means a divalent radical derived from heteroalkyl, as exemplified by -CH 2 - CH 2 -S-CH_CH 2 - and -CH_-S-CH 2 -CH 2 -NH-CH 2 -.
- heteroatoms can also occupy either or both of the chain termini. Still further, for alkylene and heteroalkylene linking groups, no orientation of the linking group is implied.
- cycloalkyl and “heterocycloalkyl”, by themselves or in combination with other terms, represent, unless otherwise stated, cyclic versions of “alkyl” and “heteroalkyl”, respectively. Additionally, for heterocycloalkyl, a heteroatom can occupy the position at which the heterocycle is attached to the remainder of the molecule. Examples of cycloalkyl include cyclopentyl, cyclohexyl, 1- cyclohexenyl, 3-cyclohexenyl, cycloheptyl, and the like.
- heterocycloalkyl examples include l-(l,2,5,6-tetrahydropyridyl), 1-piperidinyl, 2-piperidinyl, 3-piperidinyl, 1- pyrrolidinyl, 2-pyrrolidinyl, 3-pyrrolidinyl, 4-morpholinyl, 3-morpholinyl, tetrahydrofuran-2-yl, tetrahydrofuran-3-yl, tetrahydrothien-2-yl, tetrahydrothien-3-yl, 1-piperazinyl, 2-piperazinyl, and the like.
- halo or halogen
- substituents mean, unless otherwise stated, a fluorine, chlorine, bromine, or iodine atom.
- fluoroalkyl are meant to include monofluoroalkyl and polyfluoroalkyl.
- aryl employed alone or in combination with other terms (e.g., aryloxy, arylthioxy, arylalkyl) means, unless otherwise stated, an aromatic substituent which can be a single ring or multiple rings (up to three rings) which are fused together or linked covalently.
- the rings may each contain from zero to four heteroatoms selected from the group consisting of N, O, and S, wherein the nitrogen and sulfur atoms are optionally oxidized, and the nitrogen atom(s) are optionally quaternized.
- the aryl groups that contain heteroatoms may be referred to as
- heteroaryl and can be attached to the remainder of the molecule through a heteroatom
- aryl groups include phenyl, 1-naphthyl, 2-naphthyl, 4-biphenyl, 1-pyrrolyl, 2-pyrrolyl, 3-pyrrolyl, 3-pyrazolyl, 2-imidazolyl, 4-imidazolyl, pyrazinyl, 2-oxazolyl, 4-oxazolyl, 2-phenyl-4-oxazolyl, 5-oxazolyl, 3-isoxazolyl, 4- isoxazolyl, 5-isoxazolyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, 2-furyl, 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl, 2-pyrimidyl, 4-pyrimidyl, 5-benzothiazolyl, purinyl, 2-benzothiazolyl,
- arylalkyl is meant to include those radicals in which an aryl group is attached to an alkyl group (e.g., benzyl, phenethyl, pyridylmethyl and the like) or a heteroalkyl group (e.g., phenoxymethyl, 2-pyridyloxymethyl, 3-(l- naphthyloxy)propyl, and the like).
- alkyl group e.g., benzyl, phenethyl, pyridylmethyl and the like
- a heteroalkyl group e.g., phenoxymethyl, 2-pyridyloxymethyl, 3-(l- naphthyloxy)propyl, and the like.
- alkyl group e.g., benzyl, phenethyl, pyridylmethyl and the like
- heteroalkyl group e.g., phenoxymethyl, 2-pyridyloxymethyl, 3-(l- naphth
- R', R" and R'" each independently refer to hydrogen, unsubstituted(Cj- C 8 )alkyl and heteroalkyl, unsubstituted aryl, aryl substituted with 1-3 halogens, unsubstituted alkyl, alkoxy or thioalkoxy groups, or aryl-(C ⁇ -C 4 )alkyl groups.
- R' and R" When R' and R" are attached to the same nitrogen atom, they can be combined with the nitrogen atom to form a 5-, 6-, or 7-membered ring.
- -NR'R is meant to include 1- pyrrolidinyl and 4-morpholinyl.
- alkyl is meant to include groups such as haloalkyl (e.g., -CF 3 and -CH 2 CF 3 ) and acyl (e.g., -C(O)CH 3 , -C(O)CF 3 , - C(O)CH 2 OCH 3 , and the like).
- substituents for the aryl groups are varied and are selected from: -halogen, -OR', -OC(O)R ⁇ -NR'R", -SR', -R ⁇ -CN, -NO 2 , -CO 2 R', -CONR'R", -C(O)R', -OC(O)NR'R", -NR"C(O)R', -NR"C(O)2R', -NR'-C(O)NR"R'", -NH-
- Two of the substituents on adjacent atoms of the aryl ring may optionally be replaced with a substituent of the formula -T-C(O)-(CH 2 ) q -U-, wherein T and U are independently -NH-, -O-, -CH 2 - or a single bond, and q is an integer of from 0 to 2.
- two of the substituents on adjacent atoms of the aryl ring may optionally be replaced with a substituent of the formula -A-(CH ) r -B-, wherein A and B are independently -CH 2 -, -O-, -NH-, -S-, -S(O)-, -S(O) 2 -, -S(O) 2 NR'- or a single bond, and r is an integer of from 1 to 3.
- One of the single bonds of the new ring so formed may optionally be replaced with a double bond.
- two of the substituents on adjacent atoms of the aryl ring may optionally be replaced with a substituent of the formula -(CH ) s -X-(CH_) t -, where s and t are independently integers of from 0 to 3, and X is -O-, -NR'-, -S-, -S(O)-, -S(O) 2 -, or -S(O) 2 NR'-.
- the substituent R' in -NR'- and -S(O) NR'- is selected from the group consisting of hydrogen or unsubstituted (Ci- C 6 )alkyl.
- the term "heteroatom” is meant to include oxygen (O), nitrogen (N), sulfur (S) and silicon (Si).
- salts are meant to include salts of the active compounds which are prepared with relatively nontoxic acids or bases, depending on the particular substituents found on the compounds described herein.
- base addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired base, either neat or in a suitable inert solvent.
- pharmaceutically acceptable base addition salts include sodium, potassium, calcium, ammonium, organic amino, or magnesium salt, or a similar salt.
- acid addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid, either neat or in a suitable inert solvent.
- Examples of pharmaceutically acceptable acid addition salts include those derived from inorganic acids like hydrochloric, hydrobromic, nitric, carbonic, monohydrogencarbonic, phosphoric, monohydrogenphosphoric, dihydrogenphosphoric, sulfuric, monohydrogensulfuric, hydriodic, or phosphorous acids and the like, as well as the salts derived from relatively nontoxic organic acids like acetic, propionic, isobutyric, oxalic, maleic, malonic, benzoic, succinic, suberic, fumaric, mandelic, phthalic, benzenesulfonic, p-tolylsulfonic, citric, tartaric, methanesulfonic, and the like.
- inorganic acids like hydrochloric, hydrobromic, nitric, carbonic, monohydrogencarbonic, phosphoric, monohydrogenphosphoric, dihydrogenphosphoric, sulfuric, monohydrogensulfuric, hydriodic, or phospho
- salts of amino acids such as arginate and the like, and salts of organic acids like glucuronic or galactunoric acids and the like (see, for example, Berge, S.M., et al (1977) J. Pharm. Sci. 66: 1-19).
- Certain specific compounds of the present invention contain both basic and acidic functionalities that allow the compounds to be converted into either base or acid addition salts.
- the neutral forms of the compounds may be regenerated by contacting the salt with a base or acid and isolating the parent compound in the conventional manner.
- the parent form of the compound differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but otherwise the salts are equivalent to the parent form of the compound for the purposes of the present invention.
- the present invention provides compounds which are in a prodrug form.
- Prodrugs of the compounds described herein are those compounds that readily undergo chemical changes under physiological conditions to provide the compounds of the present invention.
- prodrugs can be converted to the compounds of the present invention by chemical or biochemical methods in an ex vivo environment. For example, prodrugs can be slowly converted to the compounds of the present invention when placed in a transdermal patch reservoir with a suitable enzyme or chemical reagent.
- Prodrugs are often useful because, in some situations, they may be easier to administer than the parent drug. They may, for instance, be bioavailable by oral administration whereas the parent drug is not.
- the prodrug may also have improved solubility in pharmacological compositions over the parent drug.
- prodrug derivatives are known in the art, such as those that rely on hydrolytic cleavage or oxidative activation of the prodrug.
- An example, without limitation, of a prodrug would be a compound of the present invention which is administered as an ester (the "prodrug"), but then is metabolically hydrolyzed to the carboxylic acid, the active entity.
- Additional examples include peptidyl derivatives of a compound of the invention.
- Certain compounds of the present invention can exist in unsolvated forms as well as solvated forms, including hydrated forms. In general, the solvated forms are equivalent to unsolvated forms and are intended to be encompassed within the scope of the present invention. Certain compounds of the present invention may exist in multiple crystalline or amorphous forms. In general, all physical forms are equivalent for the uses contemplated by the present invention and are intended to be within the scope of the present invention.
- Certain compounds of the present invention possess asymmetric carbon atoms (optical centers) or double bonds; the racemates, enantiomers, diastereomers, geometric isomers and individual isomers are all intended to be encompassed within the scope of the present invention.
- the compounds of the present invention may also contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute such compounds.
- the compounds may be radiolabeled with radioactive isotopes, such as for example tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C). All isotopic variations of the compounds of the present invention, whether radioactive or not, are intended to be encompassed within the scope of the present invention.
- MCHR General MCHR (GenBank Accession No. U71092) is expressed in brain, at moderate levels in the eye and skeletal muscle, and in low levels in tongue and the pituitary gland. Evidence suggests that MCHR is involved in, wter alia, olfactory learning, regulation of feeding behavior and energy metabolism, regulation of the hypothalmic-pituitary-adrenocortical axis following stress, arousal and the sensation of anxiety (Saito et al, TEM 11(8):299-303 (2000)).
- the compounds of the present invention inhibit MCHR activity, and thus, are useful in, for example, the treatment or prevention of disorders associated with these processes.
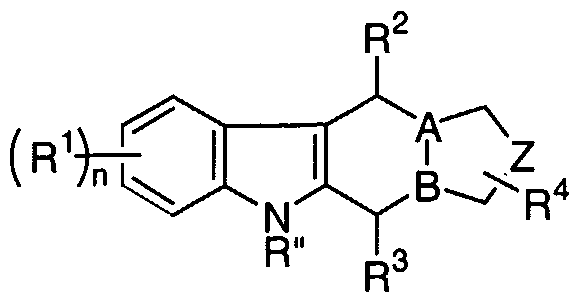
- the present invention provides compounds represented by the formula (I):
- a and B are independently selected from the group consisting of CR' and N, wherein R' is selected from the group consisting of hydrogen, (C ⁇ -Cs)alkyl, arylalkyl, -C(O)R 7 , -CO 2 R 8 and -C(O)NR 5 R 6 ;
- Z is selected from the group consisting of -N(R)-, -N(R)-(Cj- C 3 )alkylene- and -(C]-C 3 )alkylene -N(R)-(C ⁇ -C 3 )alkylene-, wherein R is selected from the group consisting of hydrogen, (C 1 -C 7 )alkyl, heterocycloalkyl(C ⁇ -C 7 )alkyl, aryl, arylalkyl, -C(O)R 7 , -CO 2 R 8 , -C(O)NR 5 R 6 , -S(O) m NR 5 R 6 and-S(O) m R 7 ; each R is independently selected from the group consisting of hydrogen, halogen, (C 1 -C 5 )alkyl, perfluoro(C 1 -C 5 )alkyl, -OR'", -SR'", aryl, arylalkyl, - NO 2 ,
- R 4 is selected from the group consisting of hydrogen -OR'", -C(O)R 7 , - CO 2 R 8 , -C(O)NR 5 R 6 , -CN, (C ⁇ -C 5 )alkyl and aryl, wherein R'" is selected from the group consisting of hydrogen, (C ⁇ -Cs)alkyl, aryl and aryl(Cj-C 5 )alkyl;
- R 5 and R 6 are independently selected from the group consisting of hydrogen, alkyl, aryl and arylalkyl or combined to form a 4-, 5-, 6-, 7- or 8-membered ring containing from one to three heteroatoms;
- R 7 and R 8 are independently selected from the group consisting of hydrogen, alkyl, aryl and arylalkyl;
- R 9 is selected from the group consisting of alkyl, aryl and arylalkyl; the subscript m is an integer from 1 to 2; the subscript n is an integer from 0 to 8; and
- R 2 represents a single or fused aryl or heteroaryl ring; with the proviso that R 2 is not hydrogen when
- the subscript n is an integer from 0 to the maximum allowable number of substituents on ring.
- the subscript n is an integer from 0 to 4.
- the subscript n is an integer from 0 to 3.
- n is an integer from 0 to the maximum allowable number of substituents on the ring.
- n is an integer from 0 to 8.
- n is an integer from 0 to 6.
- n is an integer from 0 to 4.
- a and B are both C(R'). In another group of preferred embodiments, A is C(R') and B is N. In another group of preferred embodiments, W is N(R"). In a particularly preferred embodiment, W is NH.
- V is a bond or C(O).
- Z is -N(R)-CH 2 -.
- R is heterocycloalkyl(C ⁇ - C 7 )alkyl.
- the compounds have the formula (II):
- the compounds have the formula (III):
- the compounds have the formula (V):
- the compounds have the formula (VI):
- the compounds have the formula (VII):
- the compounds have the formula (VIII):
- the compounds have the formula:
- the compounds have the formula (X):
- the compounds have the formula (XI):
- compositions comprising compounds of formula I.
- the present invention provides pharmaceutical compositions which are suitable for pharmaceutical or diagnostic use.
- the compositions comprise compounds of formulas I-XI provided above, in combination with a diagnostically or pharmaceutically acceptable carrier or excipient.
- the subject compositions are useful for treating or preventing conditions and disorders mediated by MCHR, such as obesity and eating disorders, e.g., anorexia nervosa.
- the compounds of the present invention can be prepared and administered in a wide variety of oral and parenteral dosage forms.
- the compounds of the present invention can be administered by injection, for example, intravenously, intramuscularly, intracutaneously, subcutaneously, intraduodenally or intraperitoneally.
- the compounds described herein can be administered by inhalation, for example, intranasally.
- the compounds of the present invention can be administered transdermally.
- Other routes of administration are also contemplated for use with the compounds of the present invention, including depot administration and rectal administration.
- the present invention also provides pharmaceutical compositions comprising a pharmaceutically acceptable carrier or excipient and either a compound of formulas I-XI or a pharmaceutically acceptable salt of a compound of formulas I-XI.
- pharmaceutically acceptable carriers can be either solid or liquid.
- Solid form preparations include powders, tablets, pills, capsules, cachets, suppositories, and dispersible granules.
- a solid carrier can be one or more substances which may also act as diluents, flavoring agents, binders, preservatives, tablet disintegrating agents, or an encapsulating material.
- the carrier is a finely divided solid which is in a mixture with the finely divided active component.
- the active component is mixed with the carrier having the necessary binding properties in suitable proportions and compacted in the shape and size desired.
- the powders and tablets preferably contain from about 5% or 10% to 70% of the active compound.
- Suitable carriers are magnesium carbonate, magnesium stearate, talc, sugar, lactose, pectin, dextrin, starch, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose, a low melting wax, cocoa butter, and the like.
- the term "preparation" is intended to include the formulation of the active compound with encapsulating material as a carrier providing a capsule in which the active component with or without other carriers, is surrounded by a carrier, which is thus in association with it.
- cachets and lozenges are included. Tablets, powders, capsules, pills, cachets, and lozenges can be used as solid dosage forms suitable for oral administration.
- a low melting wax such as a mixture of fatty acid glycerides or cocoa butter
- the active component is dispersed homogeneously therein, as by stirring.
- the molten homogeneous mixture is then poured into convenient sized molds, allowed to cool, and thereby to solidify.
- Liquid form preparations include solutions, suspensions, and emulsions, for example, water or water/propylene glycol solutions.
- liquid preparations can be formulated in solution in aqueous polyethylene glycol solution.
- Aqueous solutions suitable for oral use can be prepared by dissolving the active component in water and adding suitable colorants, flavors, stabilizers, and thickening agents as desired.
- Aqueous suspensions suitable for oral use can be made by dispersing the finely divided active component in water with viscous material, such as natural or synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose, and other well-known suspending agents.
- viscous material such as natural or synthetic gums, resins, methylcellulose, sodium carboxymethylcellulose, and other well-known suspending agents.
- solid form preparations which are intended to be converted, shortly before use, to liquid form preparations for oral administration.
- Such liquid forms include solutions, suspensions, and emulsions.
- These preparations may contain, in addition to the active component, colorants, flavors, stabilizers, buffers, artificial and natural sweeteners, dispersants, thickeners, solubilizing agents, and the like.
- the pharmaceutical preparation is preferably in unit dosage form.
- the preparation is subdivided into unit doses containing appropriate quantities of the active component.
- the unit dosage form can be a packaged preparation, the package containing discrete quantities of preparation, such as packeted tablets, capsules, and powders in vials or ampoules.
- the unit dosage form can be a capsule, tablet, cachet, or lozenge itself, or it can be the appropriate number of any of these in packaged form.
- the quantity of active component in a unit dose preparation may be varied or adjusted from 0.1 mg to 1000 mg, preferably 1.0 mg to 100 mg according to the particular application and the potency of the active component.
- the composition can, if desired, also contain other compatible therapeutic agents.
- the compounds utilized in the pharmaceutical method of the invention are administered at the initial dosage of about 0.001 mg/kg to about 100 mg/kg daily.
- a daily dose range of about 0.1 mg/kg to about 10 mg/kg is preferred.
- the dosages may be varied depending upon the requirements of the patient, the severity of the condition being treated, and the compound being employed. Determination of the proper dosage for a particular situation is within the skill of the practitioner.
- treatment is initiated with smaller dosages which are less than the optimum dose of the compound. Thereafter, the dosage is increased by small increments until the optimum effect under the circumstances is reached.
- the total daily dosage may be divided and administered in portions during the day, if desired.
- compositions may be advantageously combined and/or used in combination with agents useful in the treatment and/or prevention of obesity and eating disorders and pathologies associated therewith (e.g., cardiovascular disease and hypertension).
- agents useful in the treatment and/or prevention of obesity and eating disorders and pathologies associated therewith e.g., cardiovascular disease and hypertension.
- administration of the subject compounds or compositions in conjunction with these alternative agents enhances the efficacy of such agents.
- the present compounds when combined or administered in combination with, e.g., anti-obesity agents, can be used in dosages which are less than the expected amounts when used alone, or less than the calculated amounts for combination therapy.
- Suitable agents for combination therapy include those that are currently commercially available and those that are in development or will be developed.
- agents useful in the treatment of obesity include ⁇ 3 adrenergic receptor agonists, leptin or derivatives thereof and neuropeptide Y antagonists.
- agents useful in the treatment of anxiety and/or mood disorders include benzodiazepines, e.g., alprazolam, chlordiazepoxide, clonazepam, clorazepate, diazepam, lorazepam, oxazepam, and the like; heterocyclic antidepressants, e.g, amitriptyline, nortriptyline, imipramine, desipramine, doxepin, trimipramine, clomipramine, protryptyline, amoxapine and maprotiline; monoamine oxidase inhibitors (MAOIs), e.g., phenelzine and tranylcypromine; serotonin reuptake inhibitors (SRIs); selective serotonin reuptake inhibitor
- the present invention provides methods of using compounds of formula I to treat or prevent a condition or disorder associated with eating behavior, energy homeostasis and anxiety.
- exemplary conditions and disorders associated with eating behavior, energy homeostasis and anxiety include eating disorders, such as anorexia nervosa and bulimia, obesity, anxiety disorders, e.g., generalized anxiety disorder, panic attacks, panic disorder and obsessive-compulsive disorder (OCD), and mood disorders, e.g., depression and bipoloar disorders.
- Methods of using compounds of formula I to treat a condition or disorder associated with eating behavior include methods of modifying eating behavior or food intake, for example, stimulating or suppressing eating behavior or increasing or decreasing food intake. The methods comprise administering to a subject in need thereof a therapeutically effective amount of a compound of formula I.
- the present invention provides methods of using compounds of formula I to treat or prevent a condition or disorder mediated by MCHR.
- the methods comprise administering to a subject in need thereof a therapeutically effective amount of a compound of formula I.
- the present invention provides methods of using compounds of formula I to modulate MCHR.
- the methods comprise contacting a cell with the compound of formula I.
- the compounds of the present invention may also modulate G-protein coupled receptors related to MCHR, e.g., MCHR2 (see International Publication Nos. WO 00/49046 and WO 01/07606).
- the present invention provides a process for the preparation of a compound of formula I.
- a general synthetic route is depicted in Scheme 1, which comprises a condensation of substituted aryl moiety a, with a bicyclic structure b.
- D 1 is hydrogen, halogen, -C(O)R 7 , -CO 2 R 8 or -C(O)NR 5 R 6 , wherein R 5 , R 6 , R 7 and R 8 are defined as above, and D 2 is a bond, -N(R")-, -N(protecting group)-, -S- or -O-, wherein R" is defined as above and protecting group is an amino protecting group.
- Conventional amino protecting groups consist of known groups which are used to protectively block an amino group during the synthesis procedures described herein.
- the activity of MCHR polypeptides can be assessed using a variety of in vitro and in vivo assays to determine functional, chemical, and physical effects, e.g., measuring ligand binding (e.g., radioactive ligand binding), second messenger (e.g., cAMP, cGMP, IP 3 , DAG, or Ca 2+ ) levels, ion flux, phosphorylation levels, transcription levels, neurotransmitter levels, and the like. Furthermore, such assays can be used to test for inhibitors and activators of MCHR. Screening assays may be used to identify modulators that can be used as therapeutic agents, e.g., antagonists of MCHR activity.
- ligand binding e.g., radioactive ligand binding
- second messenger e.g., cAMP, cGMP, IP 3 , DAG, or Ca 2+
- ion flux e.g., phosphorylation levels
- transcription levels e.g., neurotransmitter
- Modulators of MCHR activity can be tested using MCHR polypeptides as described above, either recombinant or naturally occurring.
- the protein can be isolated, expressed in a cell, expressed in a membrane derived from a cell, expressed in tissue or in an animal, either recombinant or naturally occurring. For example, kidney cells, liver cells, colon cells, transformed cells, or membranes can be used. Modulation is tested using one of the in vitro or in vivo assays described herein.
- Signal transduction can also be examined in vitro with soluble or solid state reactions, using a chimeric molecule such as an extracellular domain of a receptor covalently linked to a heterologous signal transduction domain, or a heterologous extracellular domain covalently linked to the transmembrane and or cytoplasmic domain of a receptor. Gene amplification can also be examined. Furthermore, ligand-binding domains of the protein of interest can be used in vitro in soluble or solid state reactions to assay for ligand binding. Ligand binding to MCHR, a domain, or chimeric protein can be tested in solution, in a bilayer membrane, attached to a solid phase, in a lipid monolayer, or in vesicles.
- Binding of a modulator can be tested using, e.g., changes in spectroscopic characteristics (e.g., fluorescence, absorbance, refractive index) hydrodynamic (e.g., shape), chromatographic, or solubility properties.
- MCHR-G-protein interactions can also be examined, by, for example, analysis of binding of the G-protein to MCHR or its release from MCHR can be examined.
- an activator will lead to the formation of a tight complex of a G protein (all three subunits) with MCHR. This complex can be detected in a variety of ways, as noted above.
- Such an assay can be modified to search for inhibitors.
- an activator is added to MCHR and G protein in the absence of GTP, allowed to form a tight complex, and then screened for inhibitors by looking at dissociation of the MCHR-G protein complex.
- release of the alpha subunit of the G protein from the other two G protein subunits serves as a criterion of activation.
- An activated or inhibited G-protein will in turn alter the properties of downstream effectors such as proteins, enzymes, and channels.
- the classic examples are the activation of cGMP phosphodiesterase by transducin in the visual system, adenylate cyclase by the stimulatory G-protein, phospholipase C by Gq and other cognate G proteins, and modulation of diverse channels by Gi and other G proteins. Downstream consequences can also be examined such as generation of diacyl glycerol and IP3 by phospholipase C, and in turn, for calcium mobilization by IP3.
- Activated MCHR becomes a substrate for kinases that phosphorylate the C-terminal tail of the receptor (and possibly other sites as well).
- activators will promote the transfer of 32 P from gamma-labeled GTP to the receptor, which can be assayed with a scintillation counter.
- the phosphorylation of the C-terminal tail will promote the binding of arrestin-like proteins and will interfere with the binding of G- proteins.
- the kinase/arrestin pathway plays a key role in the desensitization of many GPCR receptors.
- Samples or assays that are treated with a potential MCHR inhibitor or activator are compared to control samples without the test compound, to examine the extent of modulation.
- Control samples (untreated with activators or inhibitors) are assigned a relative MCHR activity value of 100.
- Inhibition of MCHR is achieved when the MCHR activity value relative to the control is about 90%, optionally 50%, optionally 25-0%.
- Activation of MCHR is achieved when the MCHR activity value relative to the control is 110%, optionally 150%, 200-500%, or 1000-2000%.
- Changes in ion flux may be assessed by determining changes in polarization (i.e., electrical potential) of the cell or membrane expressing MCHR.
- polarization i.e., electrical potential
- One means to determine changes in cellular polarization is by measuring changes in current (thereby measuring changes in polarization) with voltage-clamp and patch-clamp techniques, e.g., the "cell-attached” mode, the "inside-out” mode, and the "whole cell” mode (see, e.g., Ackerman et al, New Engl. J. Med. 336:1575-1595 (1997)).
- Whole cell currents are conveniently determined using the standard methodology (see, e.g., Hamil et al, PFlugers. Archiv. 391:85 (1981).
- test compounds upon the function of the polypeptides can be measured by examining any of the parameters described above. Any suitable physiological change that affects MCHR activity can be used to assess the influence of a test compound on the polypeptides of this invention.
- functional consequences are determined using intact cells or animals, one can also measure a variety of effects such as transmitter release, hormone release, transcriptional changes to both known and uncharacterized genetic markers (e.g., northern blots), changes in cell metabolism such as cell growth or pH changes, and changes in intracellular second messengers such as Ca 2+ , IP3 or cAMP.
- Preferred assays for MCHR include cells that are loaded with ion- or voltage-sensitive dyes to report receptor activity. Assays for determining activity of such receptors can also use known agonists and antagonists for other G-protein coupled receptors as negative or positive controls to assess activity of tested compounds. In assays for identifying modulatory compounds (e.g., agonists, antagonists), changes in the level of ions in the cytoplasm or membrane voltage will be monitored using an ion- sensitive or membrane voltage fluorescent indicator, respectively. Among the ion- sensitive indicators and voltage probes that may be employed are those disclosed in the Molecular Probes 1997 Catalog.
- promiscuous G-proteins such as G ⁇ l5 and G ⁇ l6 can be used in the assay of choice (Wilkie et al, Proc. Natl Acad. Sci. USA 88:10049-10053 (1991)).
- Such promiscuous G-proteins allow coupling of a wide range of receptors to signal transduction pathways in heterologous cells.
- Receptor activation typically initiates subsequent intracellular events, e.g., increases in second messengers such as IP3, which releases intracellular stores of calcium ions.
- IP3 inositol triphosphate
- phospholipase C-mediated hydrolysis of phosphatidylinositol (Benidge & Irvine, Nature 312:315-21 (1984)).
- IP3 in turn stimulates the release of intracellular calcium ion stores.
- a change in cytoplasmic calcium ion levels, or a change in second messenger levels such as EP3 can be used to assess G-protein coupled receptor function.
- Cells expressing such G-protein coupled receptors may exhibit increased cytoplasmic calcium levels as a result of contribution from both intracellular stores and via activation of ion channels, in which case it may be desirable although not necessary to conduct such assays in calcium-free buffer, optionally supplemented with a chelating agent such as EGTA, to distinguish fluorescence response resulting from calcium release from internal stores.
- Other assays can involve determining the activity of receptors which, when activated, result in a change in the level of intracellular cyclic nucleotides, e.g., cAMP or cGMP, by activating or inhibiting downstream effectors such as adenylate cyclase.
- cyclic nucleotide-gated ion channels e.g., rod photoreceptor cell channels and olfactory neuron channels that are permeable to cations upon activation by binding of cAMP or cGMP (see, e.g., Altenhofen et al, Proc. Natl. Acad. Sci. U.S.A. 88:9868-9872 (1991) and Dhallan et al, Nature 347:184-187 (1990)).
- cyclic nucleotide levels it may be preferable to expose the cells to agents that increase intracellular cyclic nucleotide levels, e.g., forskolin, prior to adding a receptor-activating compound to the cells in the assay.
- agents that increase intracellular cyclic nucleotide levels e.g., forskolin
- Cells for this type of assay can be made by co-transfection of a host cell with DNA encoding a cyclic nucleotide-gated ion channel, GPCR phosphatase and DNA encoding a receptor (e.g., certain glutamate receptors, muscarinic acetylcholine receptors, dopamine receptors, serotonin receptors, and the like), which, when activated, causes a change in cyclic nucleotide levels in the cytoplasm.
- a receptor e.g., certain glutamate receptors, muscarinic acetylcholine receptors, dopamine receptors, serotonin receptors, and the like
- changes in intracellular cAMP or cGMP can be measured using immunoassays.
- the method described in Offermanns & Simon, J. Biol. Chem. 270:15175-15180 (1995) may be used to determine the level of cAMP.
- the method described in Felley-Bosco et al., Am. J. Resp. Cell and Mol. Biol. 11: 159-164 (1994) may be used to determine the level of cGMP.
- an assay kit for measuring cAMP and/or cGMP is described in U.S. Patent 4,115,538, herein inco ⁇ orated by reference.
- phosphatidyl inositol (PI) hydrolysis can be analyzed according to U.S. Patent 5,436,128, herein incorporated by reference.
- transcription levels can be measured to assess the effects of a test compound on signal transduction. A host cell containing the protein of interest is contacted with a test compound for a sufficient time to effect any interactions, and then the level of gene expression is measured. The amount of time to effect such interactions may be empirically determined, such as by running a time course and measuring the level of transcription as a function of time. The amount of transcription may be measured by using any method known to those of skill in the art to be suitable.
- mRNA expression of the protein of interest may be detected using northern blots or their polypeptide products may be identified using immunoassays.
- transcription based assays using a reporter gene may be used as described in U.S. Patent 5,436,128, herein incorporated by reference.
- the reporter genes can be, e.g., chloramphenicol acetyltransferase, firefly luciferase, bacterial luciferase, ⁇ -galactosidase and alkaline phosphatase.
- the protein of interest can be used as an indirect reporter via attachment to a second reporter such as green fluorescent protein (see, e.g., Mistili & Spector, Nature Biotechnology 15:961- 964 (1997)).
- the amount of transcription is then compared to the amount of transcription in either the same cell in the absence of the test compound, or it may be compared with the amount of transcription in a substantially identical cell that lacks the protein of interest.
- a substantially identical cell may be derived from the same cells from which the recombinant cell was prepared but which had not been modified by introduction of heterologous DNA. Any difference in the amount of transcription indicates that the test compound has in some manner altered the activity of the protein of interest.
- high throughput screening methods involve providing a combinatorial chemical or peptide library containing a large number of potential therapeutic compounds (potential modulator or ligand compounds). Such "combinatorial chemical libraries” or “ligand libraries” are then screened in one or more assays, as described herein, to identify those library members (particular chemical species or subclasses) that display a desired characteristic activity. The compounds thus identified can serve as conventional "lead compounds” or can themselves be used as potential or actual therapeutics.
- a combinatorial chemical library is a collection of diverse chemical compounds generated by either chemical synthesis or biological synthesis, by combining a number of chemical "building blocks” such as reagents.
- a linear combinatorial chemical library such as a polypeptide library is formed by combining a set of chemical building blocks (amino acids) in every possible way for a given compound length (i.e., the number of amino acids in a polypeptide compound). Millions of chemical compounds can be synthesized through such combinatorial mixing of chemical building blocks.
- combinatorial chemical libraries include, but are not limited to, peptide libraries (see, e.g., U.S. Patent 5,010,175, Furka, Int. J. Pept. Prot. Res. 37:487-493 (1991) and Houghton et al, Nature 354:84-88 (1991)).
- chemistries for generating chemical diversity libraries can also be used. Such chemistries include, but are not limited to: peptoids (e.g., PCT Publication No.
- nucleic acid libraries see Ausubel, Berger and Sambrook, all supra
- peptide nucleic acid libraries see, e.g., U.S. Patent 5,539,083
- antibody libraries see, e.g., Vaughn et al, Nature Biotechnology, 14(3):309-314 (1996) and PCT/US96/10287)
- carbohydrate libraries see, e.g., Liang et al, Science, 274:1520- 1522 (1996) and U.S. Patent 5,593,853
- small organic molecule libraries see, e.g., benzodiazepines, Baum C&EN, Jan 18, page 33 (1993); isoprenoids, U.S.
- High throughput assays for the presence, absence, quantification, or other properties of particular compounds may be used to test a combinatorial library that contains a large number of potential therapeutic compounds (potential modulator compounds).
- the assays are typically designed to screen large chemical libraries by automating the assay steps and providing compounds from any convenient source to assays, which are typically run in parallel (e.g., in microtiter formats on microtiter plates in robotic assays).
- Preferred assays detect activation or inhibition of MCHR activity.
- each well of a microtiter plate can be used to run a separate assay against a selected potential modulator, or, if concentration or incubation time effects are to be observed, every 5-10 wells can test a single modulator.
- a single standard microtiter plate can assay about 100 (e.g., 96) modulators. If 1536 well plates are used, then a single plate can easily assay from about 100- about 1500 different compounds. It is possible to assay several different plates per day; assay screens for up to about 6,000-20,000 different compounds is possible using the integrated systems of the invention.
- the molecule of interest can be bound to the solid state component, directly or indirectly, via covalent or noncovalent linkage e.g., via a tag.
- the tag can be any of a variety of components.
- a molecule which binds the tag (a tag binder) is fixed to a solid support, and the tagged molecule of interest (e.g., the signal transduction molecule of interest) is attached to the solid support by interaction of the tag and the tag binder.
- tags and tag binders can be used, based upon known molecular interactions well described in the literature.
- a tag has a natural binder, for example, biotin, protein A, or protein G
- tag binders avidin, streptavidin, neutravidin, the Fc region of an immunoglobulin, etc.
- Antibodies to molecules with natural binders such as biotin are also widely available and appropriate tag binders; see, SIGMA Immunochemicals 1998 catalogue (SIGMA, St. Louis MO).
- any haptenic or an ti genie compound can be used in combination with an appropriate antibody to form a tag/tag binder pair.
- Thousands of specific antibodies are commercially available and many additional antibodies are described in the literature.
- the tag is a first antibody and the tag binder is a second antibody which recognizes the first antibody.
- receptor-ligand interactions are also appropriate as tag and tag-binder pairs.
- agonists and antagonists of cell membrane receptors can be used in forming immobilizable tag and capture moiety pairs.
- cell receptor-ligand interactions such as transferrin, c-kit, viral receptor ligands, cytokine receptors, chemokine receptors, interleukin receptors, immunoglobulin receptors and antibodies, the cadherin family, the integrin family and the selectin family, can all be employed in the methods of the present invention (see, e.g., Pigott & Power, 77 ⁇ e Adhesion Molecule Facts Book I (1993).
- toxins and venoms viral epitopes, hormones (e.g., opiates, steroids, etc.), intracellular receptors (e.g.
- Synthetic polymers such as polyurethanes, polyesters, polycarbonates, polyureas, polyamides, polyethyleneimines, polyarylene sulfides, polysiloxanes, polyimides, and polyacetates can also form an appropriate tag or tag binder. Many other tag/tag binder pairs are also useful in assay systems described herein, as would be apparent to one of skill upon review of this disclosure.
- linkers such as peptides, polyethers, and the like can also serve as tags, and include polypeptide sequences, such as poly-gly sequences of between about 5 and 200 amino acids.
- polypeptide sequences such as poly-gly sequences of between about 5 and 200 amino acids.
- Such flexible linkers are known to persons of skill in the art.
- poly(ethelyne glycol) linkers are available from Shearwater
- Tag binders are fixed to solid substrates using any of a variety of methods currently available. Solid substrates are commonly derivatized or functionalized by exposing all or a portion of the substrate to a chemical reagent which fixes a chemical group to the surface which is reactive with a portion of the tag binder. For example, groups which are suitable for attachment to a longer chain portion would include amines, hydroxyl, thiol, and carboxyl groups. Aminoalkylsilanes and hydroxyalkylsilanes can be used to functionalize a variety of surfaces, such as glass surfaces.
- Non-chemical approaches for fixing tag binders to substrates include other common methods, such as heat, cross-linking by UV radiation, and the like.
- Yet another assay for compounds that modulate MCHR activity involves computer assisted drug design, in which a computer system is used to generate a three- dimensional structure of MCHR based on the structural information encoded by the amino acid sequence.
- the input amino acid sequence interacts directly and actively with a preestablished algorithm in a computer program to yield secondary, tertiary, and quaternary structural models of the protein.
- the models of the protein structure are then examined to identify regions of the structure that have the ability to bind, e.g., modulators. These regions are then used to identify modulators that bind to the protein.
- the amino acid sequence represents a primary structure that encodes the information necessary to form the secondary, tertiary and quaternary structure of the protein of interest.
- the software looks at certain parameters encoded by the primary sequence to generate the structural model. These parameters are referred to as "energy terms,” and primarily include electrostatic potentials, hydrophobic potentials, solvent accessible surfaces, and hydrogen bonding. Secondary energy terms include van der Waals potentials. Biological molecules form the structures that minimize the energy terms in a cumulative fashion. The computer program is therefore using these terms encoded by the primary structure or amino acid sequence to create the secondary structural model.
- the tertiary structure of the protein encoded by the secondary structure is then formed on the basis of the energy terms of the secondary structure.
- the user at this point can enter additional variables such as whether the protein is membrane bound or soluble, its location in the body, and its cellular location, e.g., cytoplasmic, surface, or nuclear. These variables along with the energy terms of the secondary structure are used to form the model of the tertiary structure.
- the computer program matches hydrophobic faces of secondary structure with like, and hydrophilic faces of secondary structure with like.
- potential ligand binding regions are identified by the computer system.
- Three-dimensional structures for potential ligands are generated by entering amino acid or nucleotide sequences or chemical formulas of compounds, as described above.
- the three-dimensional structure of the potential ligand is then compared to that of the GPCR protein to identify ligands that bind to GPCR. Binding affinity between the protein and ligands is determined using energy terms to determine which ligands have an enhanced probability of binding to the protein.
- Reagents and solvents used below can be obtained from commercial sources such as Aldrich Chemical Co. (Milwaukee, Wisconsin, USA).
- 1H-NMR spectra were recorded on a Varian Gemini 400 MHz NMR spectrometer. Significant peaks are tabulated in the order: multiplicity (s, singlet; d, doublet; t, triplet; q, quartet; m, multiplet; br s, broad singlet), coupling constant(s) in Hertz (Hz) and number of protons.
- Electron Ionization (El) mass spectra were recorded on a Hewlett Packard 5989A mass spectrometer.
- Mass spectrometry results are reported as the ratio of mass over charge, followed by the relative abundance of each ion (in parentheses). A single m/e value is reported for the M+H (or, as noted, M-H) ion containing the most common atomic isotopes. Isotope patterns correspond to the expected formula in all cases.
- Electrospray ionization (ESI) mass spectrometry analysis was conducted on a Hewlett- Packard 1100 MSD electrospray mass spectrometer using the HP1 100 HPLC for sample delivery. Normally the analyte was dissolved in methanol at 0.1 mg/mL and 1 microliter was infused with the delivery solvent into the mass spectrometer, which scanned from 100 to 1500 daltons. All compounds could be analyzed in the positive ESI mode, using 1: 1 acetonitrile/water with 1% acetic acid as the delivery solvent. The compounds provided below could also be analyzed in the negative ESI mode, using 2mM NHiOAc in acetonitrile/water as delivery solvent.
- ESI Electrospray ionization
- Analytical HPLC analysis was conducted on a Hewlett-Packard Series 1050 system equipped with a C18 reverse phase column (4.6 mm xl50mm) manufactured by Shiseido Co., Japan. Gradient elution was performed using variable percentage of acetonitrile and water (each with 0.1% trifluoroacetic acid added) as a mobile phase.
- Optical purity analysis was also conducted on a Hewlett-Packard Series 1050 system equipped with a chiral HLPC column (ChiralPak AD, 4.6 mm x 150mm) purchased from Chiral Technology. Isopropanol (3%) and hexane (97%) containing 0.1% diethylamine was used as a mobile phase.
- reaction mixture was poured into aqueous NaHCO 3 and extracted with EtOAc.
- the organic layer was separated, washed with brine, dried with anhydrous Na 2 SO 4 , concentrated by rotary evaporation and purified by flash chromatography on silica gel with a gradient elution of 10-50% MeOH/EtOAc mixed with 0-20% cone, ammonia to yield enone i, wherein R is a methyl group, as a yellowish oil (2.66 g).
- Enone i (2.60 g, 14.52 mmol) was stirred with 10%Pd/C (0.300 g) in EtOH (100 mL under balloon H 2 for 2.5 days. The reaction mixture was filtered. The filtrate was collected, concentrated by rotary evaporation and purified by flash chromatography on silica gel with a gradient elution of 20-40% MeOH/CH 2 Cl 2 mixed with 0-20% cone, ammonia to yield the corresponding ketone ii as a yellowish solid (1.955 g).
- compound 1 can be prepared enantioselectively by performing a resolution of enone i.
- the solid salt collected was triturated with 100 mL 95% ethanol (hot), after cooling the solid was collected again by filtration.
- the free base (22.3 g, 0.087 mol) was obtained after neutralization with aqueous NaHCO 3 and extraction with AcOEt.
- the optical purity of the resolved product was determined to 96% ee by chiral HPLC analysis.
- Compound 4 was prepared in two steps from compound 3, as follows.
- Step A A mixture of compound 3 (3.80 g, 9.55 mmol), HCO 2 NH 4 (3.03 g, 48 mmol), 10%Pd/C (0.380 g) and MeOH (150 mL) was refluxed for 7 h. The reaction mixture was cooled to room temperature, basified with saturated aqueous NaHCO 3 and extracted with EtOAc.
- Step B The product of step A (4a) (1.52 g, 4.94 mmol) was treated with N-2- chloroethylmorpholine hydrochloride (0.964 g, 5.19 mmol), Nal (0.22g, 1.48 mmol), NaHCO 3 (1.03 g, 12.5 mmol), in acetone (50 mL) for 15 h at refluxing temperature. The reaction mixture was poured into aqueous NaHCO and extracted with EtOAc.
- Tetrahydropyan-4-ylmethanol was obtained from the reduction of tetrahydropyran-4-carboxylic acid with borane or from the reduction of methyl tetrahydropyran-4-ylcarboxylate with LiAlHj.
- tetrahydropyran-4-ylmethanol 10 g, 86.2 mmol
- Example 5 substituting N-(3-methanesulfonyloxy-propyl)-N-methyl- methanesulfonamide for l-(3-chloropropyl)pyrrolidin-2-one.
- the corcesponding HC1 salt was prepared by the addition of IN HC1 in ether to a solution of the product in ethyl acetate. The HC1 salt precipitated out on concentration.
- Step A A mixture of compound 4a, (5.5 g, 17.6 mmol), N-(3- bromopropyl)phthalimide (5.5 g, 20.5 mmol), sodium bicarbonate (5.0 g, 63.1 mmol) and sodium iodide (0.50 g, 3.33 mmol) in DMF (50 mL) was heated at 90 °C overnight. The reaction mixture was diluted with ethyl acetate, washed with brine. The organic layer was dried over MgSO 4 , filtered and concentrated. The crude product was purified by flash chromatography on silica gel, eluting with a solvent system consisting of CH ⁇ -MeOH-NH OH in 40: 1 :0.1 ratio by volume to give the desired phthalimide intermediate.
- Step B The product from step A was dissolved in ethanol (75 mL) and treated with hydrazine monohydrate (15 mL) at reflux overnight. Upon cooling to room temperature, the precipitate was removed by filtration and the filtrate was concentrated, and purified by flash chromatography on silica gel, eluting with CH 2 Cl -MeOH- NH t OH in 10:1:0.1 ratio by volume to give the corresponding amine.
- Step C Compound 14 was prepared by treating a sample of the amine compound from step B with methanesulfonyl chloride in the presence of a tertiary amine base such as triethylamine.
- a tertiary amine base such as triethylamine.
- Compound 15 was prepared by treating a sample of the amine from step
- the MCHR modulatory activity of the compounds of the invention can be assessed using the in vitro and in vivo assay methods described above.
- Exemplary in vitro methods include fluorometric imaging plate reader
- FLIPR functional assays
- G Protein-Coupled Receptors (1999) pp. 105-108 T. Haga, G. Bernstein, eds.) CRC Press; Lembo et al. (1999) Nature Cell Biol. 1:267- 271; Saito et al. (1999) Nature 400:265-269; Wood et al. (2000) Eur. J. Pharmacol. 396:1-8 and Miller et al. (1999) J. Biomol. Screen. 4:249-258) and radioligand binding assays (see, e.g., Receptor Binding Techniques (1999) pp. 37-47 (M.
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP02734135A EP1392298B1 (en) | 2001-05-04 | 2002-05-03 | Fused heterocyclic compounds |
| AU2002305323A AU2002305323A1 (en) | 2001-05-04 | 2002-05-03 | Fused heterocyclic compounds |
| DE60231202T DE60231202D1 (en) | 2001-05-04 | 2002-05-03 | CONDENSED HETEROCYCLIC COMPOUNDS |
| JP2002586869A JP2004529161A (en) | 2001-05-04 | 2002-05-03 | Fused heterocyclic compounds |
| CA002446351A CA2446351A1 (en) | 2001-05-04 | 2002-05-03 | Fused heterocyclic compounds |
| US10/928,029 US7125885B2 (en) | 2001-05-04 | 2004-08-26 | Fused heterocyclic compounds |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US28866501P | 2001-05-04 | 2001-05-04 | |
| US60/288,665 | 2001-05-04 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/289,933 Continuation-In-Part US6809104B2 (en) | 2001-05-04 | 2002-11-06 | Fused heterocyclic compounds |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2002089729A2 true WO2002089729A2 (en) | 2002-11-14 |
| WO2002089729A3 WO2002089729A3 (en) | 2003-04-03 |
Family
ID=23108101
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2002/013856 WO2002089729A2 (en) | 2001-05-04 | 2002-05-03 | Fused heterocyclic compounds |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US6858619B2 (en) |
| EP (1) | EP1392298B1 (en) |
| JP (1) | JP2004529161A (en) |
| AT (1) | ATE422887T1 (en) |
| AU (1) | AU2002305323A1 (en) |
| CA (1) | CA2446351A1 (en) |
| DE (1) | DE60231202D1 (en) |
| ES (1) | ES2319619T3 (en) |
| WO (1) | WO2002089729A2 (en) |
Cited By (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004043958A1 (en) * | 2002-11-06 | 2004-05-27 | Amgen Inc. | Fused heterocyclic compounds |
| US6809104B2 (en) | 2001-05-04 | 2004-10-26 | Tularik Inc. | Fused heterocyclic compounds |
| US6858619B2 (en) | 2001-05-04 | 2005-02-22 | Amgen Inc. | Fused heterocyclic compounds |
| US6906075B2 (en) | 2002-01-10 | 2005-06-14 | Neurogen Corp. | Melanin concentrating hormone receptor ligands: substituted benzoimidazole analogues |
| WO2006018280A2 (en) | 2004-08-16 | 2006-02-23 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted polycyclic amines, method for the production thereof, and use thereof as a medicament |
| US7084156B2 (en) | 2001-11-27 | 2006-08-01 | Merck & Co., Inc. | 2-Aminoquinoline compounds |
| US7160879B2 (en) | 2002-01-10 | 2007-01-09 | Neurogen Corporation | Melanin concentrating hormone receptor ligands: substituted 2-(4-benzyl-piperazin-1-ylmethyl)- and 2-(4-benzyl-diazepan-1-ylmethyl)-1H-benzoimidazole analogues |
| US7241787B2 (en) | 2004-01-25 | 2007-07-10 | Sanofi-Aventis Deutschland Gmbh | Substituted N-cycloexylimidazolinones, process for their preparation and their use as medicaments |
| US7319108B2 (en) | 2004-01-25 | 2008-01-15 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted heterocycles, process for their preparation and their use as medicaments |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| US7803816B2 (en) | 2005-09-30 | 2010-09-28 | Hoffmann-La Roche Inc. | MCH receptor antagonists |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| US8501771B2 (en) | 2006-02-15 | 2013-08-06 | Sanofi | Aminoalcohol-substituted aryldihydroisoquinolinones, process for their preparation and their use as medicaments |
| US8552199B2 (en) | 2009-02-13 | 2013-10-08 | Sanofi | Substituted indanes, method for the production thereof, and use thereof as drugs |
| US8575186B2 (en) | 2009-10-05 | 2013-11-05 | Albany Molecular Research, Inc. | Epiminocycloalkyl[b] indole derivatives as serotonin sub-type 6 (5-HT6) modulators and uses thereof |
| US8841290B2 (en) | 2009-02-13 | 2014-09-23 | Sanofi | Substituted tetrahydronaphthalenes, method for the production thereof, and use thereof as drugs |
| US9067949B2 (en) | 2011-01-19 | 2015-06-30 | Albany Molecular Research, Inc. | Benzofuro[3,2-c] pyridines and related analogs as serotonin sub-type 6 (5-HT6) modulators for the treatment of obesity, metabolic syndrome, cognition and schizophrenia |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070135425A1 (en) * | 2001-05-04 | 2007-06-14 | Amgen Inc. | Fused heterocyclic compounds |
| EP1451156A4 (en) * | 2001-11-27 | 2005-05-25 | Merck & Co Inc | 4-aminoquinoline compounds |
| WO2004069798A1 (en) * | 2003-02-10 | 2004-08-19 | Banyu Pharmaceutical Co.,Ltd. | Melanin-concentrating hormone receptor antagonists containing piperidine derivatives as the active ingredient |
| US20150051220A1 (en) * | 2012-03-16 | 2015-02-19 | Glucox Biotech Ab | Compounds for use in therapy |
| JP6274384B2 (en) * | 2012-04-27 | 2018-02-07 | Nok株式会社 | Metal gasket and manufacturing method thereof |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2906654B2 (en) | 1989-11-28 | 1999-06-21 | 東レ株式会社 | Immunosuppressant and method for producing the same |
| AU662007B2 (en) * | 1991-07-05 | 1995-08-17 | Smithkline Beecham Farmaceutici S.P.A. | Hydroisoquinoline derivatives |
| ATE239732T1 (en) | 1992-09-29 | 2003-05-15 | Toray Industries | INDOLE DERIVATIVE, METHOD FOR THE PRODUCTION THEREOF AND ITS MEDICAL APPLICATION |
| US5464841A (en) | 1993-11-08 | 1995-11-07 | Univ Minnesota | Use of delta opioid receptor antagonists to treat immunoregulatory disorders |
| US5578725A (en) | 1995-01-30 | 1996-11-26 | Regents Of The University Of Minnesota | Delta opioid receptor antagonists |
| US6156762A (en) | 1997-01-16 | 2000-12-05 | Toray Industries, Inc. | Indolomorphinan derivative and agent for curing and preventing cerebral disorder |
| JP3886030B2 (en) | 1998-10-07 | 2007-02-28 | シャープ株式会社 | Semiconductor laser element |
| GB9822158D0 (en) | 1998-10-09 | 1998-12-02 | Nycomed Imaging As | Compositions |
| AU2573900A (en) | 1999-02-19 | 2000-09-04 | Takeda Chemical Industries Ltd. | Novel g protein-coupled receptor protein and dna thereof |
| WO2001007606A1 (en) | 1999-07-27 | 2001-02-01 | Smithkline Beecham Plc | Axor21, a g-protein coupled receptor |
| JP2001226269A (en) | 2000-02-18 | 2001-08-21 | Takeda Chem Ind Ltd | Melanin-concentrating hormone antagonist |
| CA2408913A1 (en) | 2000-05-16 | 2001-11-22 | Takeda Chemical Industries, Ltd. | Melanin-concentrating hormone antagonist |
| DE60143525D1 (en) | 2000-07-05 | 2011-01-05 | Takeda Pharmaceutical | PROCESS FOR SCREENING MHC RECEPTOR ANTAGONISTS OR AGONISTS |
| JP2004504303A (en) | 2000-07-05 | 2004-02-12 | シナプティック・ファーマスーティカル・コーポレーション | Selective melanin-concentrating hormone-1 (MCH1) receptor antagonists and uses thereof |
| JP2004502423A (en) | 2000-07-05 | 2004-01-29 | シナプティック・ファーマスーティカル・コーポレーション | DNA encoding human melanin-concentrating hormone receptor (MCH1) and use thereof |
| JP2004516239A (en) | 2000-07-06 | 2004-06-03 | ニューロジェン コーポレイション | Melanin-concentrating hormone receptor ligand |
| PL362076A1 (en) | 2000-10-20 | 2004-10-18 | Pfizer Products Inc. | Alpha-aryl ethanolamines and their use as beta-3 adrenergic receptor agonists |
| US20030022891A1 (en) | 2000-12-01 | 2003-01-30 | Anandan Palani | MCH antagonists and their use in the treatment of obesity |
| ES2243588T3 (en) | 2000-12-22 | 2005-12-01 | Schering Corporation | PCHERIDINIC ANTAGONISTS OF THE MCH AND ITS USE IN THE TREATMENT OF OBESITY. |
| JP4557492B2 (en) | 2001-03-21 | 2010-10-06 | ファーマコペイア ドラッグ ディスカバリー, インコーポレイテッド | Aryl and biaryl compounds having MCH modulating activity |
| US6900329B2 (en) | 2001-03-21 | 2005-05-31 | Schering Corporation | MCH antagonists and their use in the treatment of obesity |
| AR035234A1 (en) | 2001-04-12 | 2004-05-05 | Pharmacopeia Drug Discovery | ARIL AND BIARIL-PIPERIDINS WITH MODULATING ACTIVITY MCH, METHOD TO PREPARE SUCH COMPOUNDS, PHARMACEUTICAL COMPOSITIONS, METHOD TO PREPARE PHARMACEUTICAL COMPOSITIONS AND THE USE OF SUCH COMPOUNDS FOR THE MANUFACTURE OF A MEDICINAL PRODUCT |
| CA2446351A1 (en) | 2001-05-04 | 2002-11-14 | Tularik Inc. | Fused heterocyclic compounds |
| CA2448080A1 (en) | 2001-05-22 | 2002-11-28 | Neurogen Corporation | Melanin concentrating hormone receptor ligands: substituted 1-benzyl-4-aryl piperazine analogues |
-
2002
- 2002-05-03 CA CA002446351A patent/CA2446351A1/en not_active Abandoned
- 2002-05-03 DE DE60231202T patent/DE60231202D1/en not_active Expired - Fee Related
- 2002-05-03 WO PCT/US2002/013856 patent/WO2002089729A2/en active Application Filing
- 2002-05-03 US US10/138,279 patent/US6858619B2/en not_active Expired - Fee Related
- 2002-05-03 AU AU2002305323A patent/AU2002305323A1/en not_active Abandoned
- 2002-05-03 ES ES02734135T patent/ES2319619T3/en not_active Expired - Lifetime
- 2002-05-03 EP EP02734135A patent/EP1392298B1/en not_active Expired - Lifetime
- 2002-05-03 JP JP2002586869A patent/JP2004529161A/en active Pending
- 2002-05-03 AT AT02734135T patent/ATE422887T1/en not_active IP Right Cessation
Non-Patent Citations (2)
| Title |
|---|
| DATABASE CAPLUS [Online] ISHIKURA ET AL: 'A novel entry to pyrido (4,3-b)carbazoles: an efficient synthesis of ellipticine', XP002957254 Retrieved from STN Database accession no. 2000:82454 & TETRAHEDRON vol. 56, no. 2, 2000, pages 193 - 207 * |
| See also references of EP1392298A2 * |
Cited By (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6809104B2 (en) | 2001-05-04 | 2004-10-26 | Tularik Inc. | Fused heterocyclic compounds |
| US6858619B2 (en) | 2001-05-04 | 2005-02-22 | Amgen Inc. | Fused heterocyclic compounds |
| US7125885B2 (en) | 2001-05-04 | 2006-10-24 | Amgen Inc. | Fused heterocyclic compounds |
| US7084156B2 (en) | 2001-11-27 | 2006-08-01 | Merck & Co., Inc. | 2-Aminoquinoline compounds |
| US6906075B2 (en) | 2002-01-10 | 2005-06-14 | Neurogen Corp. | Melanin concentrating hormone receptor ligands: substituted benzoimidazole analogues |
| US7160879B2 (en) | 2002-01-10 | 2007-01-09 | Neurogen Corporation | Melanin concentrating hormone receptor ligands: substituted 2-(4-benzyl-piperazin-1-ylmethyl)- and 2-(4-benzyl-diazepan-1-ylmethyl)-1H-benzoimidazole analogues |
| JP2006508130A (en) * | 2002-11-06 | 2006-03-09 | アムジェン インコーポレーテッド | Fused heterocyclic compounds |
| WO2004043958A1 (en) * | 2002-11-06 | 2004-05-27 | Amgen Inc. | Fused heterocyclic compounds |
| US7253179B2 (en) | 2002-11-06 | 2007-08-07 | Amgen Inc. | Fused heterocyclic compounds |
| EA009421B1 (en) * | 2002-11-06 | 2007-12-28 | Эмджен Инк. | Fused heterocyclic compounds |
| US8299104B2 (en) | 2004-01-25 | 2012-10-30 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted heterocycles, process for their preparation and their use as medicaments |
| US7241787B2 (en) | 2004-01-25 | 2007-07-10 | Sanofi-Aventis Deutschland Gmbh | Substituted N-cycloexylimidazolinones, process for their preparation and their use as medicaments |
| US7319108B2 (en) | 2004-01-25 | 2008-01-15 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted heterocycles, process for their preparation and their use as medicaments |
| US7838547B2 (en) | 2004-08-16 | 2010-11-23 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted polycyclic amines, method for the production thereof, and use thereof as a medicament |
| WO2006018280A2 (en) | 2004-08-16 | 2006-02-23 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted polycyclic amines, method for the production thereof, and use thereof as a medicament |
| US8357686B2 (en) | 2004-08-16 | 2013-01-22 | Sanofi-Aventis Deutschland Gmbh | Aryl-substituted polycyclic amines, method for the production thereof, and use thereof as a medicament |
| US7803816B2 (en) | 2005-09-30 | 2010-09-28 | Hoffmann-La Roche Inc. | MCH receptor antagonists |
| US8501771B2 (en) | 2006-02-15 | 2013-08-06 | Sanofi | Aminoalcohol-substituted aryldihydroisoquinolinones, process for their preparation and their use as medicaments |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| US8609731B2 (en) | 2007-08-15 | 2013-12-17 | Sanofi | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| US8841290B2 (en) | 2009-02-13 | 2014-09-23 | Sanofi | Substituted tetrahydronaphthalenes, method for the production thereof, and use thereof as drugs |
| US8552199B2 (en) | 2009-02-13 | 2013-10-08 | Sanofi | Substituted indanes, method for the production thereof, and use thereof as drugs |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| US8575186B2 (en) | 2009-10-05 | 2013-11-05 | Albany Molecular Research, Inc. | Epiminocycloalkyl[b] indole derivatives as serotonin sub-type 6 (5-HT6) modulators and uses thereof |
| US9067949B2 (en) | 2011-01-19 | 2015-06-30 | Albany Molecular Research, Inc. | Benzofuro[3,2-c] pyridines and related analogs as serotonin sub-type 6 (5-HT6) modulators for the treatment of obesity, metabolic syndrome, cognition and schizophrenia |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2004529161A (en) | 2004-09-24 |
| EP1392298A2 (en) | 2004-03-03 |
| DE60231202D1 (en) | 2009-04-02 |
| US20030023085A1 (en) | 2003-01-30 |
| EP1392298A4 (en) | 2005-05-25 |
| ATE422887T1 (en) | 2009-03-15 |
| ES2319619T3 (en) | 2009-05-11 |
| CA2446351A1 (en) | 2002-11-14 |
| EP1392298B1 (en) | 2009-02-18 |
| AU2002305323A1 (en) | 2002-11-18 |
| WO2002089729A3 (en) | 2003-04-03 |
| US6858619B2 (en) | 2005-02-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1392298B1 (en) | Fused heterocyclic compounds | |
| US7045527B2 (en) | Piperidine derivatives | |
| US20080058365A1 (en) | Fused heterocyclic compounds | |
| EP1144409B1 (en) | Phenyl urea and phenyl thiourea derivatives | |
| EP3027612B1 (en) | 11-oxo-10,11-dihydrodibenzo[b,f][1,4]thiazepine s-oxide derivatives and their use as dopamine d2 receptor antagonists | |
| Ghoneim et al. | Novel ligands for the human histamine H1 receptor: synthesis, pharmacology, and comparative molecular field analysis studies of 2-dimethylamino-5-(6)-phenyl-1, 2, 3, 4-tetrahydronaphthalenes | |
| CA2611296A1 (en) | Pyrazolopyridine derivatives as inhibitors of beta-adrenergic receptor kinase 1 | |
| US6809104B2 (en) | Fused heterocyclic compounds | |
| US20070135425A1 (en) | Fused heterocyclic compounds | |
| US7030122B2 (en) | 1,4-disubstituted piperazine ligands for neurotransmitter receptors | |
| WO2006062110A1 (en) | Piperazine derivative |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ OM PH PL PT RO RU SD SE SG SI SK SL TJ TM TN TR TT TZ UA UG US UZ VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2002586869 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2446351 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2002734135 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2002734135 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |