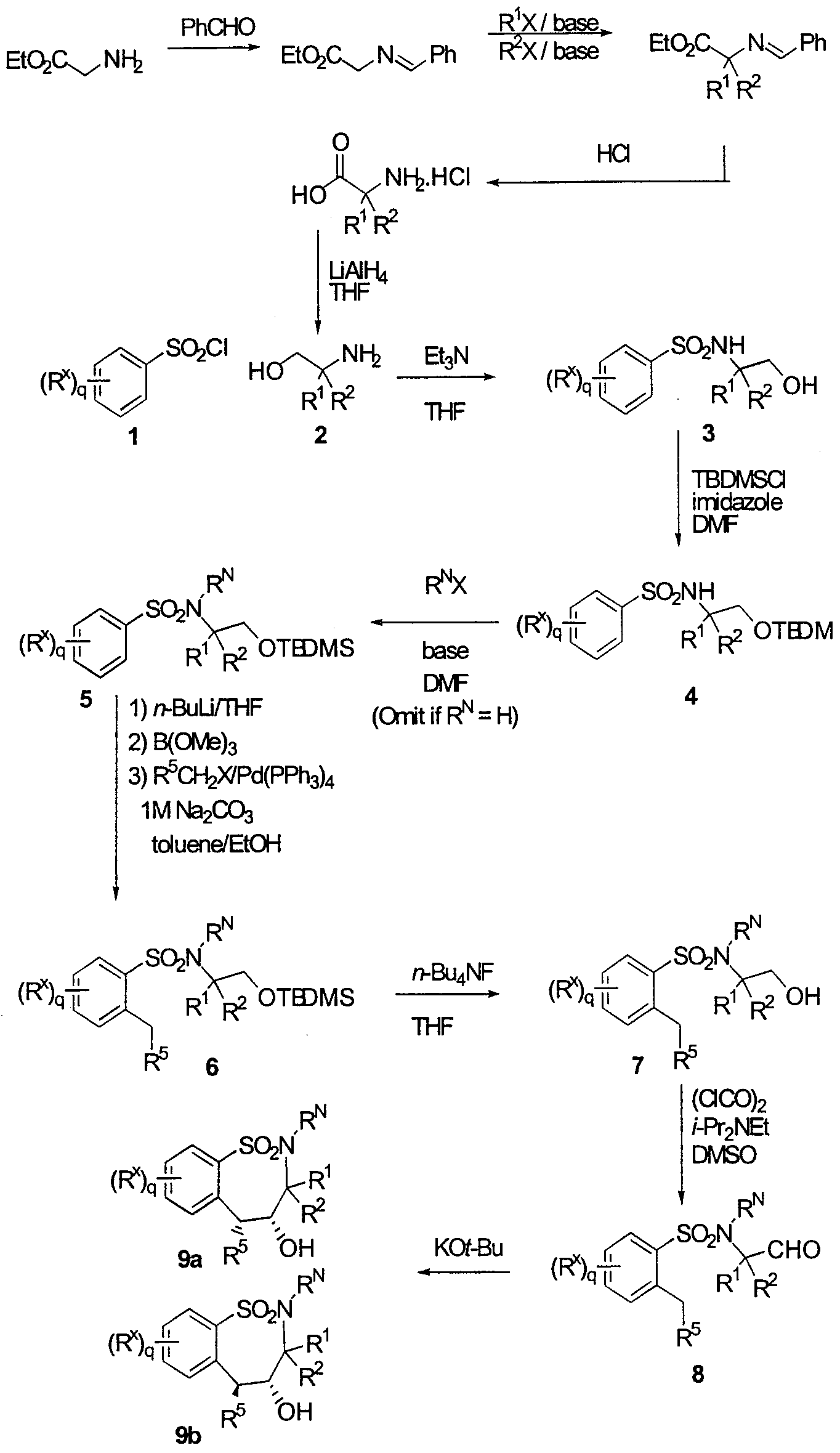
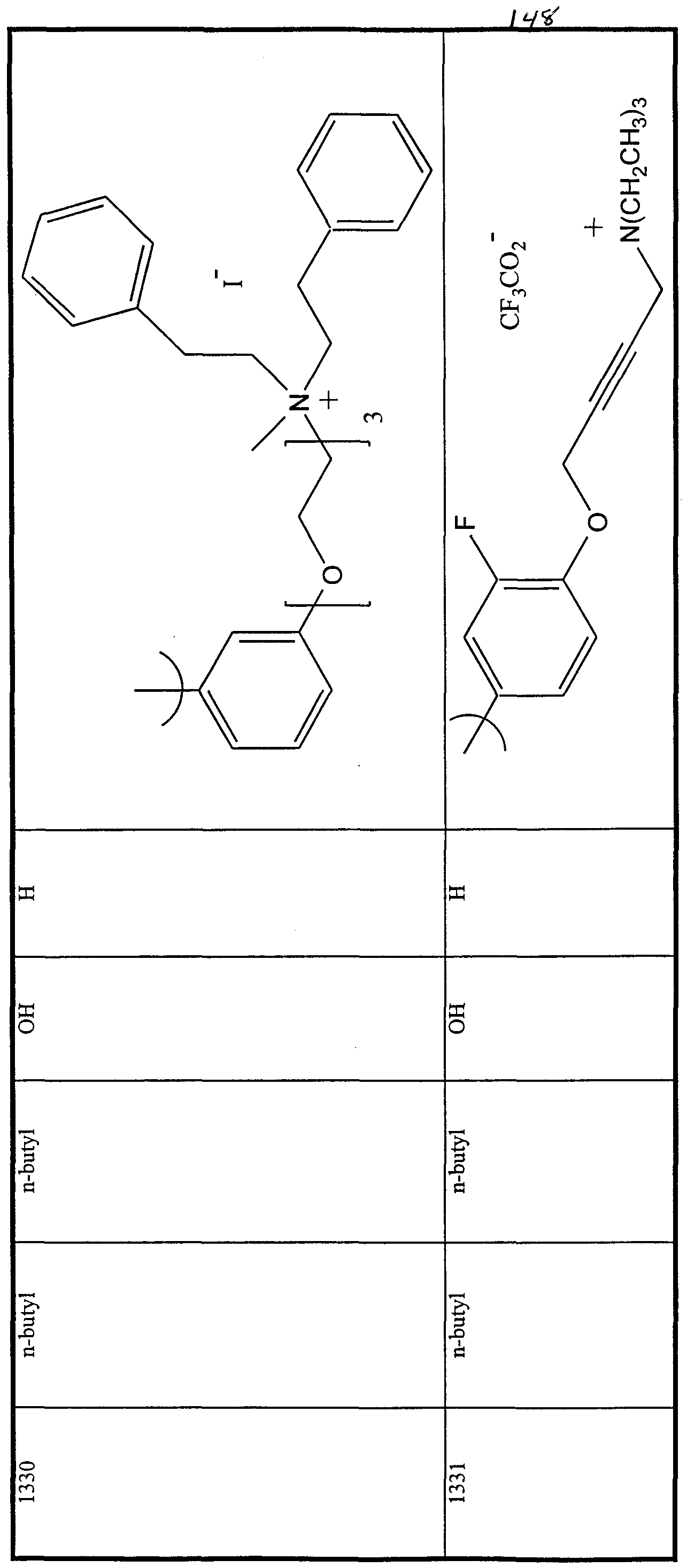
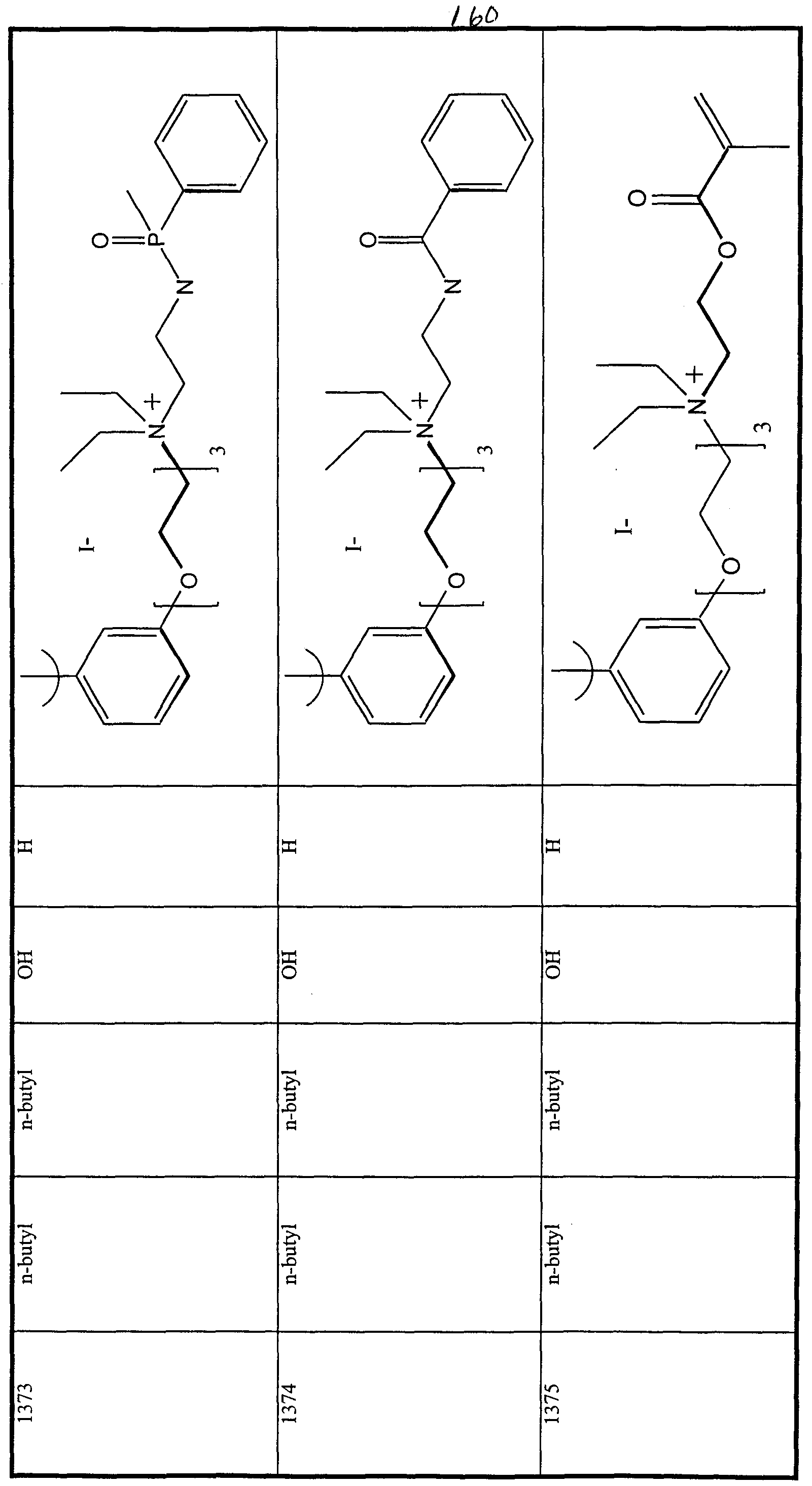
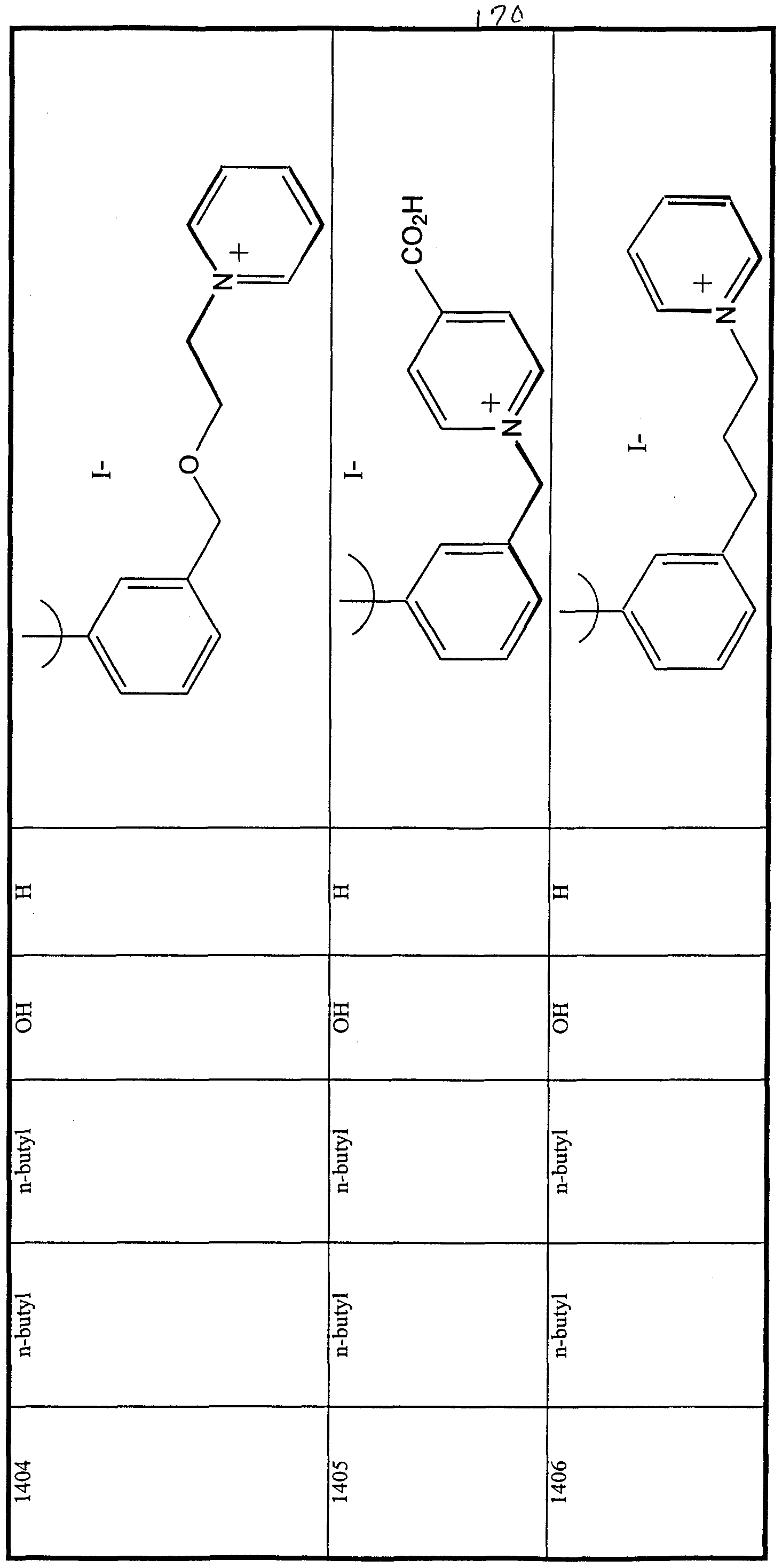
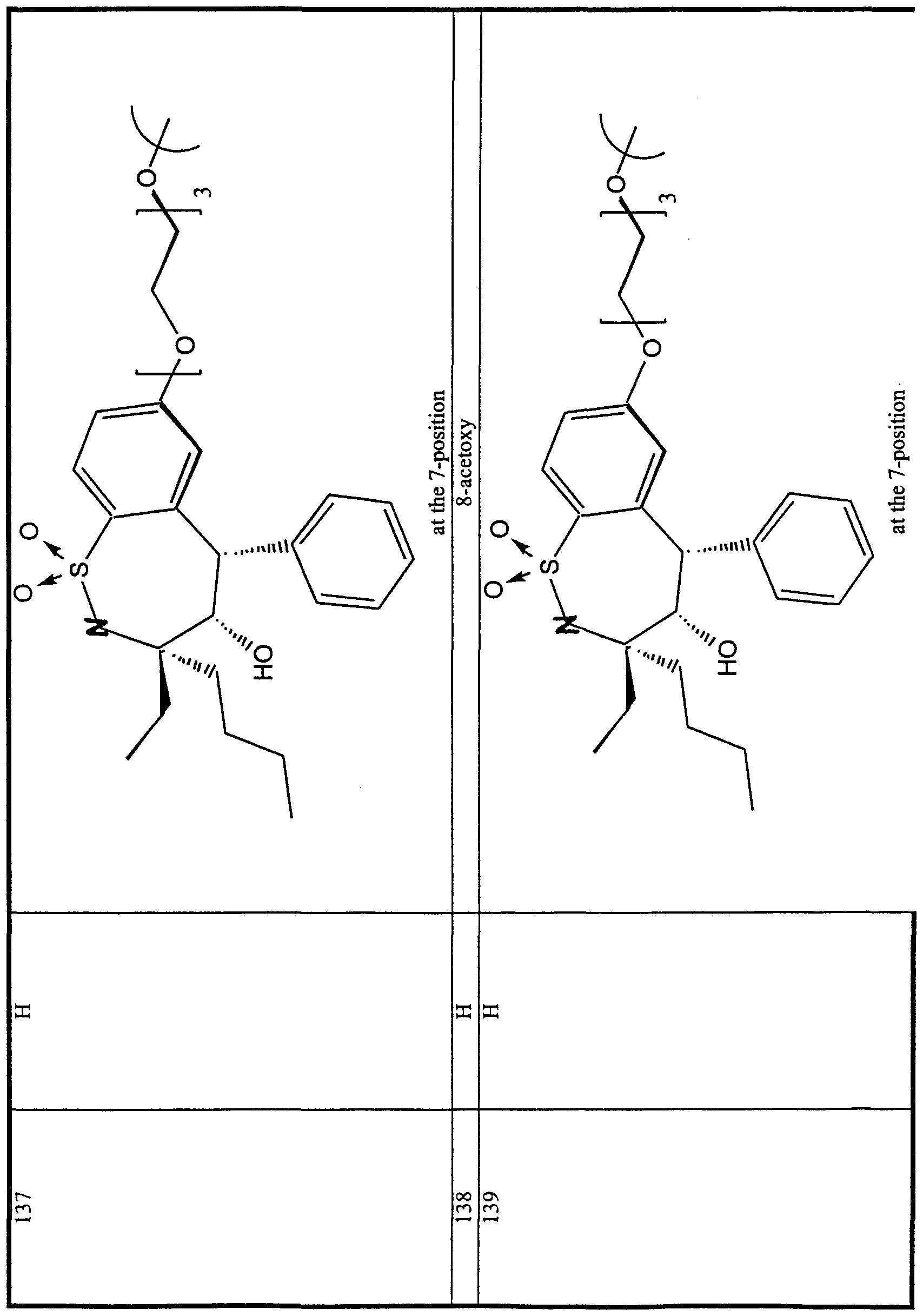
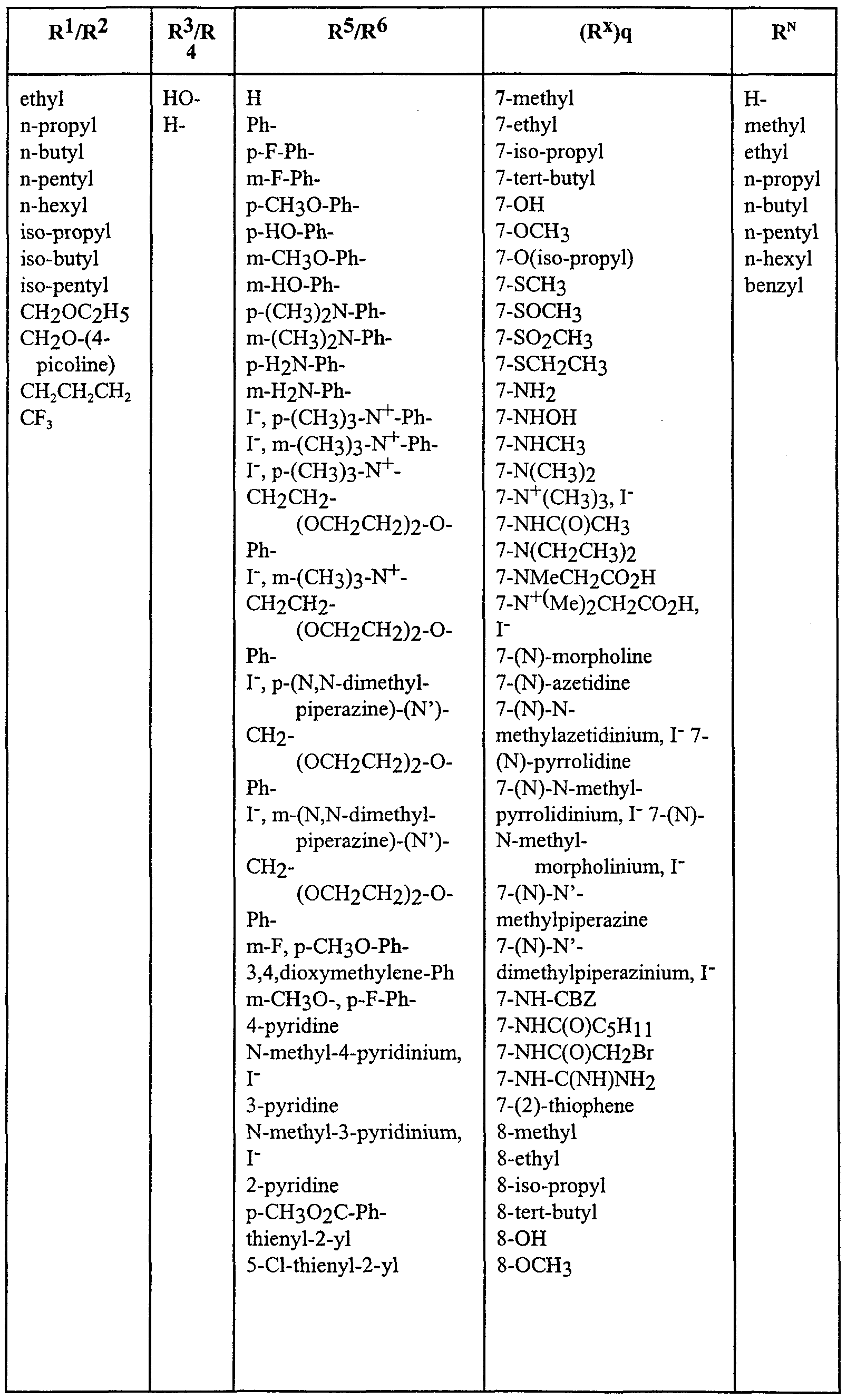
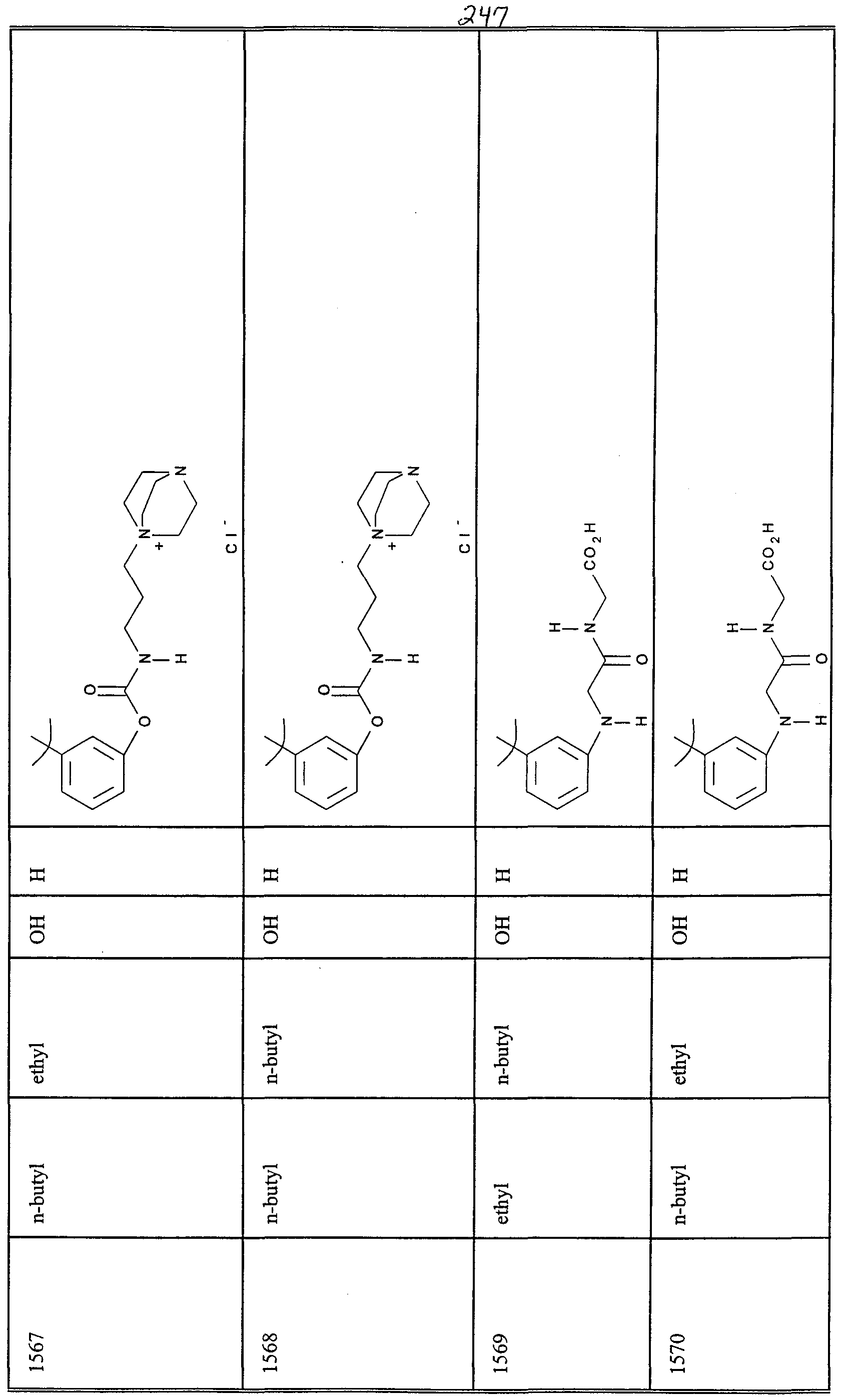
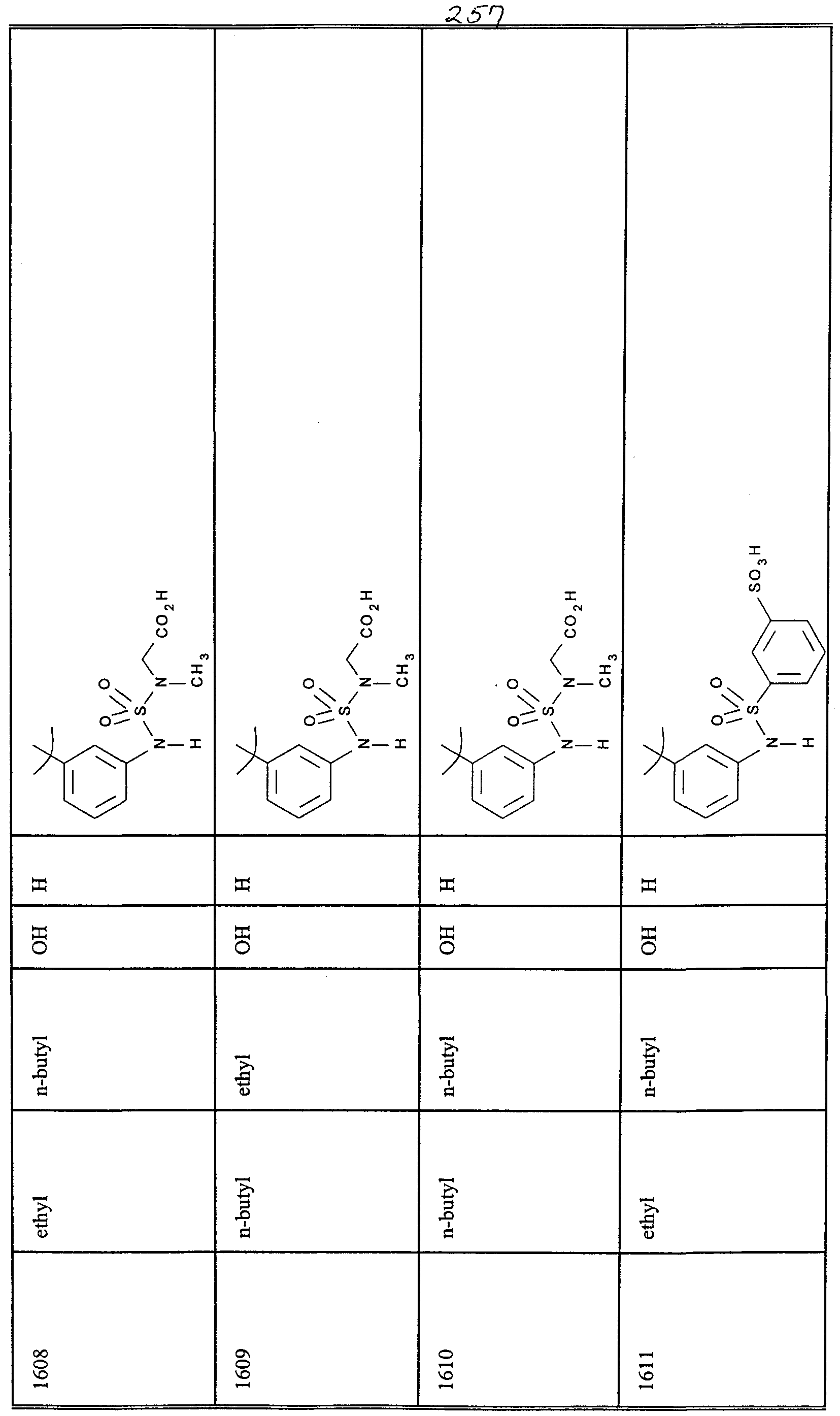
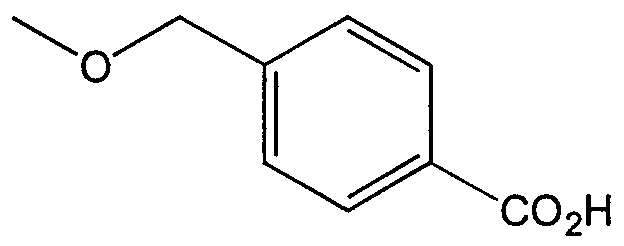
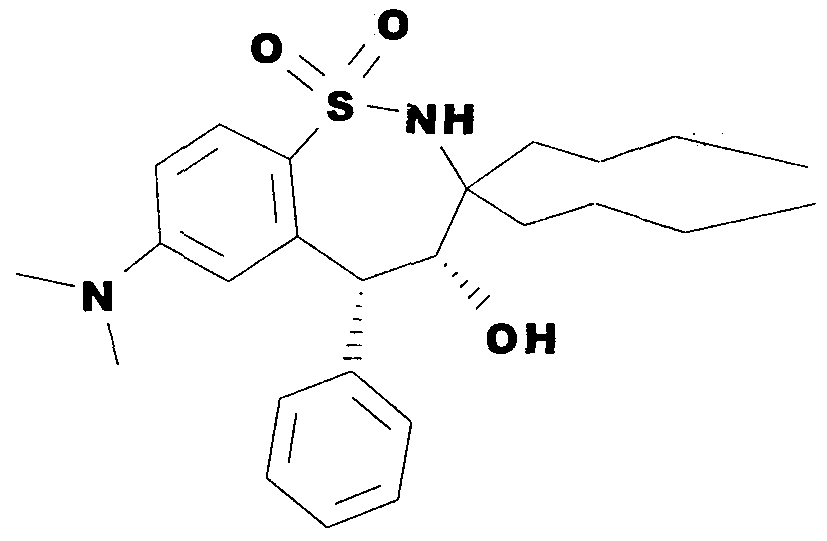
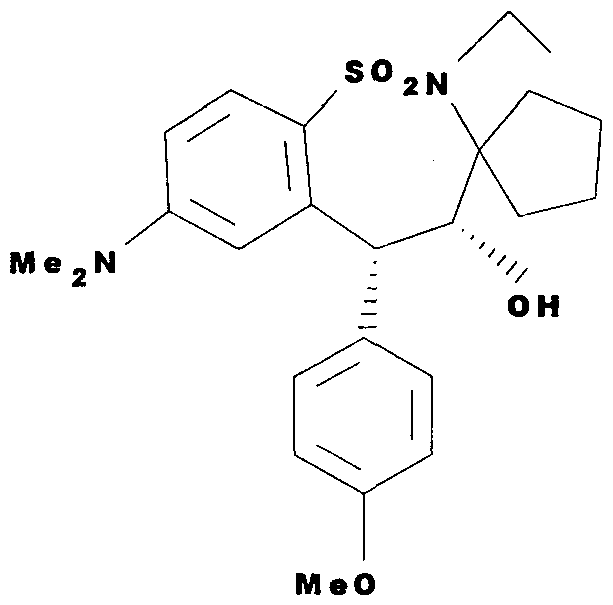
WO2000047568A2 - 1,2-benzothiazepines for the treatment of hyperlipidemic diseases - Google Patents
1,2-benzothiazepines for the treatment of hyperlipidemic diseases Download PDFInfo
- Publication number
- WO2000047568A2 WO2000047568A2 PCT/US2000/002503 US0002503W WO0047568A2 WO 2000047568 A2 WO2000047568 A2 WO 2000047568A2 US 0002503 W US0002503 W US 0002503W WO 0047568 A2 WO0047568 A2 WO 0047568A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- heterocyclyl
- group
- aryl
- independently selected
- Prior art date
Links
- 238000011282 treatment Methods 0.000 title claims abstract description 30
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 title abstract description 23
- 201000010099 disease Diseases 0.000 title abstract description 13
- FHGWEHGZBUBQKL-UHFFFAOYSA-N 1,2-benzothiazepine Chemical class S1N=CC=CC2=CC=CC=C12 FHGWEHGZBUBQKL-UHFFFAOYSA-N 0.000 title description 16
- 238000011321 prophylaxis Methods 0.000 claims abstract description 17
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 12
- 208000035150 Hypercholesterolemia Diseases 0.000 claims abstract description 6
- 125000000217 alkyl group Chemical group 0.000 claims description 676
- 125000000623 heterocyclic group Chemical group 0.000 claims description 585
- -1 heterocyclyl radicals Chemical class 0.000 claims description 434
- 125000003118 aryl group Chemical group 0.000 claims description 355
- 150000003254 radicals Chemical class 0.000 claims description 277
- 125000000304 alkynyl group Chemical group 0.000 claims description 259
- 150000001875 compounds Chemical class 0.000 claims description 258
- 229910052739 hydrogen Inorganic materials 0.000 claims description 250
- 239000001257 hydrogen Substances 0.000 claims description 248
- 125000003342 alkenyl group Chemical group 0.000 claims description 231
- 125000004415 heterocyclylalkyl group Chemical group 0.000 claims description 214
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 191
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 189
- 239000004721 Polyphenylene oxide Chemical group 0.000 claims description 181
- 229920000570 polyether Chemical group 0.000 claims description 181
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 173
- 229910052736 halogen Inorganic materials 0.000 claims description 134
- 150000002367 halogens Chemical class 0.000 claims description 132
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 129
- 125000001188 haloalkyl group Chemical group 0.000 claims description 123
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 111
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 94
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 75
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 73
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 73
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 64
- 229920001184 polypeptide Polymers 0.000 claims description 64
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 64
- 229910052757 nitrogen Inorganic materials 0.000 claims description 62
- 229910052799 carbon Inorganic materials 0.000 claims description 60
- 125000005843 halogen group Chemical group 0.000 claims description 54
- 125000003545 alkoxy group Chemical group 0.000 claims description 53
- 125000000539 amino acid group Chemical group 0.000 claims description 52
- 125000001424 substituent group Chemical group 0.000 claims description 51
- 150000003839 salts Chemical group 0.000 claims description 50
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 49
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 48
- 125000004432 carbon atom Chemical group C* 0.000 claims description 47
- 125000005842 heteroatom Chemical group 0.000 claims description 44
- 150000002431 hydrogen Chemical class 0.000 claims description 44
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 43
- 150000001720 carbohydrates Chemical class 0.000 claims description 42
- 125000006376 (C3-C10) cycloalkyl group Chemical group 0.000 claims description 41
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 40
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 40
- 125000004948 alkyl aryl alkyl group Chemical group 0.000 claims description 39
- 125000002252 acyl group Chemical group 0.000 claims description 38
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 37
- 125000004689 alkyl amino carbonyl alkyl group Chemical group 0.000 claims description 37
- 125000005097 aminocarbonylalkyl group Chemical group 0.000 claims description 37
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 36
- 125000004122 cyclic group Chemical group 0.000 claims description 36
- 125000000392 cycloalkenyl group Chemical group 0.000 claims description 34
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 34
- 125000005210 alkyl ammonium group Chemical group 0.000 claims description 32
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 32
- 125000006374 C2-C10 alkenyl group Chemical group 0.000 claims description 31
- 125000004183 alkoxy alkyl group Chemical group 0.000 claims description 31
- 125000004181 carboxyalkyl group Chemical group 0.000 claims description 29
- 229910052760 oxygen Inorganic materials 0.000 claims description 29
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 28
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 28
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 27
- 239000001301 oxygen Substances 0.000 claims description 27
- 150000001450 anions Chemical class 0.000 claims description 25
- 229910052717 sulfur Inorganic materials 0.000 claims description 25
- 150000001768 cations Chemical class 0.000 claims description 24
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 23
- 239000011593 sulfur Substances 0.000 claims description 23
- 150000005840 aryl radicals Chemical class 0.000 claims description 22
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims description 21
- 239000002253 acid Substances 0.000 claims description 21
- 125000005082 alkoxyalkenyl group Chemical group 0.000 claims description 21
- 229910052698 phosphorus Inorganic materials 0.000 claims description 21
- 239000011574 phosphorus Substances 0.000 claims description 21
- 125000003282 alkyl amino group Chemical group 0.000 claims description 20
- 239000012453 solvate Substances 0.000 claims description 20
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 19
- 150000001721 carbon Chemical group 0.000 claims description 19
- 239000000651 prodrug Substances 0.000 claims description 19
- 229940002612 prodrug Drugs 0.000 claims description 19
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 claims description 19
- 150000001335 aliphatic alkanes Chemical group 0.000 claims description 18
- 239000000203 mixture Substances 0.000 claims description 17
- 150000001413 amino acids Chemical class 0.000 claims description 16
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 16
- 125000002950 monocyclic group Chemical group 0.000 claims description 16
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 16
- 125000005160 aryl oxy alkyl group Chemical group 0.000 claims description 15
- 238000000034 method Methods 0.000 claims description 15
- 125000000027 (C1-C10) alkoxy group Chemical group 0.000 claims description 14
- 125000004423 acyloxy group Chemical group 0.000 claims description 14
- 125000005098 aryl alkoxy carbonyl group Chemical group 0.000 claims description 14
- 125000005605 benzo group Chemical group 0.000 claims description 14
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 14
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 14
- 125000004964 sulfoalkyl group Chemical group 0.000 claims description 14
- 150000001336 alkenes Chemical class 0.000 claims description 12
- 150000001345 alkine derivatives Chemical class 0.000 claims description 12
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 claims description 12
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 12
- 239000002552 dosage form Substances 0.000 claims description 11
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 claims description 10
- 125000004457 alkyl amino carbonyl group Chemical group 0.000 claims description 10
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims description 10
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 10
- ZMANZCXQSJIPKH-UHFFFAOYSA-O triethylammonium ion Chemical compound CC[NH+](CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-O 0.000 claims description 10
- 125000001544 thienyl group Chemical group 0.000 claims description 9
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 claims description 8
- 125000004076 pyridyl group Chemical group 0.000 claims description 8
- 125000004966 cyanoalkyl group Chemical group 0.000 claims description 7
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 claims description 7
- 239000003937 drug carrier Substances 0.000 claims description 7
- 125000001624 naphthyl group Chemical group 0.000 claims description 7
- 125000004103 aminoalkyl group Chemical group 0.000 claims description 6
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 6
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 6
- 235000010290 biphenyl Nutrition 0.000 claims description 5
- 239000004305 biphenyl Substances 0.000 claims description 5
- 125000005026 carboxyaryl group Chemical group 0.000 claims description 5
- 125000002541 furyl group Chemical group 0.000 claims description 5
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 5
- 125000006501 nitrophenyl group Chemical group 0.000 claims description 5
- 125000004193 piperazinyl group Chemical group 0.000 claims description 5
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 5
- 125000000335 thiazolyl group Chemical group 0.000 claims description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims description 4
- FFDGPVCHZBVARC-UHFFFAOYSA-N N,N-dimethylglycine Chemical compound CN(C)CC(O)=O FFDGPVCHZBVARC-UHFFFAOYSA-N 0.000 claims description 4
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 4
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 4
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 4
- 125000004464 hydroxyphenyl group Chemical group 0.000 claims description 4
- 125000002883 imidazolyl group Chemical group 0.000 claims description 4
- 125000001041 indolyl group Chemical group 0.000 claims description 4
- 125000000842 isoxazolyl group Chemical group 0.000 claims description 4
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-O pyridinium Chemical compound C1=CC=[NH+]C=C1 JUJWROOIHBZHMG-UHFFFAOYSA-O 0.000 claims description 4
- 125000006413 ring segment Chemical group 0.000 claims description 4
- 125000001425 triazolyl group Chemical group 0.000 claims description 4
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 claims description 4
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 claims description 3
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 claims description 3
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 claims description 3
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 3
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 3
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 3
- 125000004663 dialkyl amino group Chemical group 0.000 claims description 3
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 claims description 3
- 125000001786 isothiazolyl group Chemical group 0.000 claims description 3
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 claims description 3
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 claims description 3
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 claims description 3
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 3
- 125000003386 piperidinyl group Chemical group 0.000 claims description 3
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 3
- ORHUIBVTSQFDHK-ROJLCIKYSA-N (4r,5r)-2-benzyl-3,3-dibutyl-7-(dimethylamino)-5-(4-methoxyphenyl)-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)CC=2C=CC=CC=2)(CCCC)CCCC)=CC=C(OC)C=C1 ORHUIBVTSQFDHK-ROJLCIKYSA-N 0.000 claims description 2
- XYVUGRAVQKLPPU-CLJLJLNGSA-N (4r,5r)-3,3-dibutyl-7-(dimethylamino)-5-(3-methoxyphenyl)-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=CC(OC)=C1 XYVUGRAVQKLPPU-CLJLJLNGSA-N 0.000 claims description 2
- RJEHRPZAQZYFIL-DNQXCXABSA-N (4r,5r)-3,3-dibutyl-7-(dimethylamino)-5-(4-hydroxyphenyl)-1,1-dioxo-4,5-dihydro-2h-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@@H]2C3=CC(=CC=C3S(=O)(=O)NC([C@@H]2O)(CCCC)CCCC)N(C)C)=CC=C(O)C=C1 RJEHRPZAQZYFIL-DNQXCXABSA-N 0.000 claims description 2
- HYBFRSKEZDNMOO-JWQCQUIFSA-N (4r,5r)-3,3-dibutyl-7-(dimethylamino)-5-(4-hydroxyphenyl)-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=C(O)C=C1 HYBFRSKEZDNMOO-JWQCQUIFSA-N 0.000 claims description 2
- WVQLVOSWPXPLRD-JWQCQUIFSA-N (4r,5r)-3,3-dibutyl-7-(dimethylamino)-5-(4-methoxyphenyl)-1,1-dioxo-4,5-dihydro-2h-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@@H]2C3=CC(=CC=C3S(=O)(=O)NC([C@@H]2O)(CCCC)CCCC)N(C)C)=CC=C(OC)C=C1 WVQLVOSWPXPLRD-JWQCQUIFSA-N 0.000 claims description 2
- ZCJMAJYQZWBZMJ-CLJLJLNGSA-N (4r,5r)-3,3-dibutyl-7-(dimethylamino)-5-(4-methoxyphenyl)-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=C(OC)C=C1 ZCJMAJYQZWBZMJ-CLJLJLNGSA-N 0.000 claims description 2
- STEYOPYSZKLUJY-DNQXCXABSA-N (4r,5r)-5-(3-aminophenyl)-3,3-dibutyl-7-(dimethylamino)-1,1-dioxo-4,5-dihydro-2h-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@@H]2C3=CC(=CC=C3S(=O)(=O)NC([C@@H]2O)(CCCC)CCCC)N(C)C)=CC=CC(N)=C1 STEYOPYSZKLUJY-DNQXCXABSA-N 0.000 claims description 2
- GGDKNYPWJCZLCW-JWQCQUIFSA-N (4r,5r)-5-(3-aminophenyl)-3,3-dibutyl-7-(dimethylamino)-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=CC(N)=C1 GGDKNYPWJCZLCW-JWQCQUIFSA-N 0.000 claims description 2
- RXYPXQSKLGGKOL-UHFFFAOYSA-N 1,4-dimethylpiperazine Chemical compound CN1CCN(C)CC1 RXYPXQSKLGGKOL-UHFFFAOYSA-N 0.000 claims description 2
- WWYVZTLIFYLZFM-UHFFFAOYSA-N 1-methylazetidine Chemical compound CN1CCC1 WWYVZTLIFYLZFM-UHFFFAOYSA-N 0.000 claims description 2
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 claims description 2
- AVFZOVWCLRSYKC-UHFFFAOYSA-N 1-methylpyrrolidine Chemical compound CN1CCCC1 AVFZOVWCLRSYKC-UHFFFAOYSA-N 0.000 claims description 2
- KGCGCEWAJOGOQT-KAYWLYCHSA-N 2-chloro-n-[3-[(4r,5r)-3,3-dibutyl-7-(dimethylamino)-4-hydroxy-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-5-yl]phenyl]acetamide Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=CC(NC(=O)CCl)=C1 KGCGCEWAJOGOQT-KAYWLYCHSA-N 0.000 claims description 2
- ZTNLGPOYWLKXPR-UHFFFAOYSA-N 2-ethenoxypyrimidine Chemical group C=COC1=NC=CC=N1 ZTNLGPOYWLKXPR-UHFFFAOYSA-N 0.000 claims description 2
- ANJFZNGNTORFNS-LOYHVIPDSA-N 5-chloro-N-[3-[(4R,5R)-3,3-dibutyl-7-(dimethylamino)-4-hydroxy-2-methyl-1,1-dioxo-4,5-dihydro-1lambda6,2-benzothiazepin-5-yl]phenyl]pentanamide Chemical compound CCCCC1(CCCC)[C@H](O)[C@H](c2cccc(NC(=O)CCCCCl)c2)c2cc(ccc2S(=O)(=O)N1C)N(C)C ANJFZNGNTORFNS-LOYHVIPDSA-N 0.000 claims description 2
- PQBAWAQIRZIWIV-UHFFFAOYSA-N N-methylpyridinium Chemical compound C[N+]1=CC=CC=C1 PQBAWAQIRZIWIV-UHFFFAOYSA-N 0.000 claims description 2
- FQABKRUUFYAYFH-SWIBWIMJSA-N [5-[3-[(4r,5r)-3,3-dibutyl-7-(dimethylamino)-4-hydroxy-1,1-dioxo-4,5-dihydro-2h-1$l^{6},2-benzothiazepin-5-yl]anilino]-5-oxopentyl]-triethylazanium;2,2,2-trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F.C1([C@@H]2C3=CC(=CC=C3S(=O)(=O)NC([C@@H]2O)(CCCC)CCCC)N(C)C)=CC=CC(NC(=O)CCCC[N+](CC)(CC)CC)=C1 FQABKRUUFYAYFH-SWIBWIMJSA-N 0.000 claims description 2
- UVPIRWADFOOMCJ-OZBLHQGLSA-N [5-[3-[(4r,5r)-3,3-dibutyl-7-(dimethylamino)-4-hydroxy-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-5-yl]anilino]-5-oxopentyl]-triethylazanium;2,2,2-trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F.C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=CC(NC(=O)CCCC[N+](CC)(CC)CC)=C1 UVPIRWADFOOMCJ-OZBLHQGLSA-N 0.000 claims description 2
- 125000002393 azetidinyl group Chemical group 0.000 claims description 2
- 125000004799 bromophenyl group Chemical group 0.000 claims description 2
- 125000000068 chlorophenyl group Chemical group 0.000 claims description 2
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 claims description 2
- 125000006125 ethylsulfonyl group Chemical group 0.000 claims description 2
- 125000001207 fluorophenyl group Chemical group 0.000 claims description 2
- 125000006303 iodophenyl group Chemical group 0.000 claims description 2
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 claims description 2
- 125000002971 oxazolyl group Chemical group 0.000 claims description 2
- AEABQBMUYZBBCW-UHFFFAOYSA-N pentanamide Chemical compound CC[CH]CC(N)=O AEABQBMUYZBBCW-UHFFFAOYSA-N 0.000 claims description 2
- 229960005235 piperonyl butoxide Drugs 0.000 claims description 2
- 125000004591 piperonyl group Chemical group C(C1=CC=2OCOC2C=C1)* 0.000 claims description 2
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 2
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 claims description 2
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 claims description 2
- 125000000837 carbohydrate group Chemical group 0.000 claims 53
- STSYRNZXXQMGHP-UHFFFAOYSA-N (amino-ethoxy-nitromethyl)-(diethylamino)-[dimethylamino-(ethylamino)-(methylamino)methyl]-[hydroxy(methoxy)methyl]azanium Chemical compound OC([N+](C(NCC)(N(C)C)NC)(C(N)([N+](=O)[O-])OCC)N(CC)CC)OC STSYRNZXXQMGHP-UHFFFAOYSA-N 0.000 claims 2
- FOUHQFWUUMJAKD-UHFFFAOYSA-N (amino-ethoxy-nitromethyl)-[(2-bromo-2-chloro-2-fluoroethyl)-ethylamino]-[dimethylamino-(ethylamino)-(methylamino)methyl]-(hydroxy-iodo-methoxymethyl)azanium Chemical compound ClC(CN(CC)[N+](C(NCC)(N(C)C)NC)(C(N)([N+](=O)[O-])OCC)C(OC)(O)I)(Br)F FOUHQFWUUMJAKD-UHFFFAOYSA-N 0.000 claims 2
- 230000000879 anti-atherosclerotic effect Effects 0.000 claims 2
- 230000001315 anti-hyperlipaemic effect Effects 0.000 claims 2
- 230000003143 atherosclerotic effect Effects 0.000 claims 2
- NVTFLQBWZMNINT-FIRIVFDPSA-N (4r,5r)-2-benzyl-3,3-dibutyl-7-(dimethylamino)-5-(3-nitrophenyl)-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)CC=2C=CC=CC=2)(CCCC)CCCC)=CC=CC([N+]([O-])=O)=C1 NVTFLQBWZMNINT-FIRIVFDPSA-N 0.000 claims 1
- GECNNFGTYHBXBF-OBKOODKPSA-M 2-[2-[2-[4-[(4r,5r)-3,3-dibutyl-7-(dimethylamino)-4-hydroxy-2-methyl-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-5-yl]phenoxy]ethoxy]ethoxy]ethyl-triethylazanium;iodide Chemical compound [I-].C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=C(OCCOCCOCC[N+](CC)(CC)CC)C=C1 GECNNFGTYHBXBF-OBKOODKPSA-M 0.000 claims 1
- HWWPLBWGSPKLOL-UHFFFAOYSA-N diethylamino(trimethyl)azanium Chemical compound CCN(CC)[N+](C)(C)C HWWPLBWGSPKLOL-UHFFFAOYSA-N 0.000 claims 1
- 201000001320 Atherosclerosis Diseases 0.000 abstract description 8
- 239000003814 drug Substances 0.000 abstract description 8
- NGBHIADNEOTIAL-UHFFFAOYSA-N 1$l^{6},2-benzothiazepine 1,1-dioxide Chemical class O=S1(=O)N=CC=CC2=CC=CC=C12 NGBHIADNEOTIAL-UHFFFAOYSA-N 0.000 abstract 1
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 34
- 239000003613 bile acid Substances 0.000 description 25
- 239000002904 solvent Substances 0.000 description 22
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 20
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 20
- HSINOMROUCMIEA-FGVHQWLLSA-N (2s,4r)-4-[(3r,5s,6r,7r,8s,9s,10s,13r,14s,17r)-6-ethyl-3,7-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]-2-methylpentanoic acid Chemical compound C([C@@]12C)C[C@@H](O)C[C@H]1[C@@H](CC)[C@@H](O)[C@@H]1[C@@H]2CC[C@]2(C)[C@@H]([C@H](C)C[C@H](C)C(O)=O)CC[C@H]21 HSINOMROUCMIEA-FGVHQWLLSA-N 0.000 description 16
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 15
- 150000007657 benzothiazepines Chemical class 0.000 description 14
- 239000002585 base Substances 0.000 description 13
- 125000001072 heteroaryl group Chemical group 0.000 description 12
- KHBQMWCZKVMBLN-UHFFFAOYSA-N Benzenesulfonamide Chemical compound NS(=O)(=O)C1=CC=CC=C1 KHBQMWCZKVMBLN-UHFFFAOYSA-N 0.000 description 11
- 230000037396 body weight Effects 0.000 description 11
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 9
- 235000014633 carbohydrates Nutrition 0.000 description 8
- 235000012000 cholesterol Nutrition 0.000 description 8
- 229940124530 sulfonamide Drugs 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 7
- 239000003112 inhibitor Substances 0.000 description 7
- 150000003456 sulfonamides Chemical class 0.000 description 7
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 125000001153 fluoro group Chemical group F* 0.000 description 6
- 150000003242 quaternary ammonium salts Chemical group 0.000 description 6
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical compound SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 6
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 238000009472 formulation Methods 0.000 description 5
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- NIGNBCLEMMGDQP-UHFFFAOYSA-N 1-benzothiepine Chemical class S1C=CC=CC2=CC=CC=C12 NIGNBCLEMMGDQP-UHFFFAOYSA-N 0.000 description 4
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 4
- 229920001268 Cholestyramine Polymers 0.000 description 4
- 241000124008 Mammalia Species 0.000 description 4
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 230000010235 enterohepatic circulation Effects 0.000 description 4
- 210000001035 gastrointestinal tract Anatomy 0.000 description 4
- 125000004446 heteroarylalkyl group Chemical group 0.000 description 4
- 239000007800 oxidant agent Substances 0.000 description 4
- 125000004430 oxygen atom Chemical group O* 0.000 description 4
- 125000004434 sulfur atom Chemical group 0.000 description 4
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 4
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 3
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 3
- 229940077672 Ileal bile acid transport inhibitor Drugs 0.000 description 3
- 108010007622 LDL Lipoproteins Proteins 0.000 description 3
- 102000007330 LDL Lipoproteins Human genes 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 150000001350 alkyl halides Chemical class 0.000 description 3
- 235000001014 amino acid Nutrition 0.000 description 3
- 125000005018 aryl alkenyl group Chemical group 0.000 description 3
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 239000012876 carrier material Substances 0.000 description 3
- 239000003638 chemical reducing agent Substances 0.000 description 3
- 238000007796 conventional method Methods 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 3
- 238000004128 high performance liquid chromatography Methods 0.000 description 3
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 3
- 239000000543 intermediate Substances 0.000 description 3
- 239000012280 lithium aluminium hydride Substances 0.000 description 3
- 210000004185 liver Anatomy 0.000 description 3
- 125000003373 pyrazinyl group Chemical group 0.000 description 3
- 239000012312 sodium hydride Substances 0.000 description 3
- 229910000104 sodium hydride Inorganic materials 0.000 description 3
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical compound ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- 229930192474 thiophene Natural products 0.000 description 3
- 125000005208 trialkylammonium group Chemical group 0.000 description 3
- HCRKCZRJWPKOAR-SNVBAGLBSA-N (4s)-4-(ethylamino)-2-(3-methoxypropyl)-1,1-dioxo-3,4-dihydrothieno[3,2-e]thiazine-6-sulfonamide Chemical compound CCN[C@@H]1CN(CCCOC)S(=O)(=O)C2=C1C=C(S(N)(=O)=O)S2 HCRKCZRJWPKOAR-SNVBAGLBSA-N 0.000 description 2
- UIUAXJIXQYWMDF-UHFFFAOYSA-N 1,2-benzothiazepin-4-amine Chemical compound S1N=CC(N)=CC2=CC=CC=C21 UIUAXJIXQYWMDF-UHFFFAOYSA-N 0.000 description 2
- GMZBKDVLWUQALU-UHFFFAOYSA-N 1,2-benzothiazepine-4-carbonitrile Chemical compound S1N=CC(C#N)=CC2=CC=CC=C21 GMZBKDVLWUQALU-UHFFFAOYSA-N 0.000 description 2
- VZHAQWONQLJNHJ-UHFFFAOYSA-N 1,2-benzothiazepine-4-thiol Chemical compound S1N=CC(S)=CC2=CC=CC=C21 VZHAQWONQLJNHJ-UHFFFAOYSA-N 0.000 description 2
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-N D-gluconic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-SQOUGZDYSA-N 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 2
- 238000008214 LDL Cholesterol Methods 0.000 description 2
- 102000000853 LDL receptors Human genes 0.000 description 2
- 108010001831 LDL receptors Proteins 0.000 description 2
- 238000003820 Medium-pressure liquid chromatography Methods 0.000 description 2
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 2
- 238000005481 NMR spectroscopy Methods 0.000 description 2
- JCXJVPUVTGWSNB-UHFFFAOYSA-N Nitrogen dioxide Chemical compound O=[N]=O JCXJVPUVTGWSNB-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 2
- 150000004661 S-thiocarbamates Chemical class 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- 239000003529 anticholesteremic agent Substances 0.000 description 2
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 2
- 229910052794 bromium Inorganic materials 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- 239000007795 chemical reaction product Substances 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 125000004982 dihaloalkyl group Chemical group 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 239000002934 diuretic Substances 0.000 description 2
- 235000019439 ethyl acetate Nutrition 0.000 description 2
- 230000029142 excretion Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 208000001130 gallstones Diseases 0.000 description 2
- 150000004676 glycans Chemical class 0.000 description 2
- 125000004438 haloalkoxy group Chemical group 0.000 description 2
- 210000003405 ileum Anatomy 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- PQNFLJBBNBOBRQ-UHFFFAOYSA-N indane Chemical compound C1=CC=C2CCCC2=C1 PQNFLJBBNBOBRQ-UHFFFAOYSA-N 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 230000002452 interceptive effect Effects 0.000 description 2
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 2
- XKBGEWXEAPTVCK-UHFFFAOYSA-M methyltrioctylammonium chloride Chemical compound [Cl-].CCCCCCCC[N+](C)(CCCCCCCC)CCCCCCCC XKBGEWXEAPTVCK-UHFFFAOYSA-M 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical class CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 2
- 239000012038 nucleophile Substances 0.000 description 2
- 150000007524 organic acids Chemical group 0.000 description 2
- 125000004043 oxo group Chemical group O=* 0.000 description 2
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 2
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 125000006684 polyhaloalkyl group Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920001282 polysaccharide Polymers 0.000 description 2
- 239000005017 polysaccharide Substances 0.000 description 2
- FGIUAXJPYTZDNR-UHFFFAOYSA-N potassium nitrate Chemical compound [K+].[O-][N+]([O-])=O FGIUAXJPYTZDNR-UHFFFAOYSA-N 0.000 description 2
- LBKJNHPKYFYCLL-UHFFFAOYSA-N potassium;trimethyl(oxido)silane Chemical compound [K+].C[Si](C)(C)[O-] LBKJNHPKYFYCLL-UHFFFAOYSA-N 0.000 description 2
- 150000003141 primary amines Chemical class 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 2
- 125000002098 pyridazinyl group Chemical group 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000006722 reduction reaction Methods 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- BGHCVCJVXZWKCC-UHFFFAOYSA-N tetradecane Chemical compound CCCCCCCCCCCCCC BGHCVCJVXZWKCC-UHFFFAOYSA-N 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 2
- 239000011701 zinc Substances 0.000 description 2
- FFOAMOASQZPOMD-CZNDPXEESA-N (4r,5r)-2-benzyl-3,3-dibutyl-7-(dimethylamino)-5-[3-(ethylamino)phenyl]-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)CC=2C=CC=CC=2)(CCCC)CCCC)=CC=CC(NCC)=C1 FFOAMOASQZPOMD-CZNDPXEESA-N 0.000 description 1
- TUJPZSYJLFMWCA-JWQCQUIFSA-N (4r,5r)-3,3-dibutyl-7-(dimethylamino)-2-methyl-5-(3-nitrophenyl)-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-4-ol Chemical compound C1([C@H]2[C@@H](O)C(N(S(=O)(=O)C3=CC=C(C=C32)N(C)C)C)(CCCC)CCCC)=CC=CC([N+]([O-])=O)=C1 TUJPZSYJLFMWCA-JWQCQUIFSA-N 0.000 description 1
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 description 1
- HLWVXZBFKQCCOR-UHFFFAOYSA-N (bromo-ethoxy-methoxymethyl)-(chloromethyl)-methyl-propan-2-yloxyazanium Chemical compound CCOC(Br)(OC)[N+](C)(CCl)OC(C)C HLWVXZBFKQCCOR-UHFFFAOYSA-N 0.000 description 1
- 125000004504 1,2,4-oxadiazolyl group Chemical group 0.000 description 1
- 125000004514 1,2,4-thiadiazolyl group Chemical group 0.000 description 1
- 125000004506 1,2,5-oxadiazolyl group Chemical group 0.000 description 1
- 125000004517 1,2,5-thiadiazolyl group Chemical group 0.000 description 1
- CSNIZNHTOVFARY-UHFFFAOYSA-N 1,2-benzothiazole Chemical compound C1=CC=C2C=NSC2=C1 CSNIZNHTOVFARY-UHFFFAOYSA-N 0.000 description 1
- 125000001781 1,3,4-oxadiazolyl group Chemical group 0.000 description 1
- 125000004520 1,3,4-thiadiazolyl group Chemical group 0.000 description 1
- KYVHGCKMVJDCNV-UHFFFAOYSA-N 1,4-benzothiazepine Chemical class S1C=CN=CC2=CC=CC=C12 KYVHGCKMVJDCNV-UHFFFAOYSA-N 0.000 description 1
- KJFRSZASZNLCDF-UHFFFAOYSA-N 1,5-benzothiazepine Chemical class S1C=CC=NC2=CC=CC=C12 KJFRSZASZNLCDF-UHFFFAOYSA-N 0.000 description 1
- MOHYOXXOKFQHDC-UHFFFAOYSA-N 1-(chloromethyl)-4-methoxybenzene Chemical compound COC1=CC=C(CCl)C=C1 MOHYOXXOKFQHDC-UHFFFAOYSA-N 0.000 description 1
- ADHAJDDBRUOZHJ-UHFFFAOYSA-N 1-benzothiophen-3-one Chemical class C1=CC=C2C(=O)CSC2=C1 ADHAJDDBRUOZHJ-UHFFFAOYSA-N 0.000 description 1
- QOFXWFNJJYFCCI-UHFFFAOYSA-N 1-sulfonyl-1$l^{4},2-benzothiazepine Chemical class O=S(=O)=S1N=CC=CC2=CC=CC=C12 QOFXWFNJJYFCCI-UHFFFAOYSA-N 0.000 description 1
- HOXWQVSIFXQYRS-UHFFFAOYSA-N 2,3,4,5-tetrahydro-1$l^{6},2-benzothiazepine 1,1-dioxide Chemical compound O=S1(=O)NCCCC2=CC=CC=C12 HOXWQVSIFXQYRS-UHFFFAOYSA-N 0.000 description 1
- OXBLVCZKDOZZOJ-UHFFFAOYSA-N 2,3-Dihydrothiophene Chemical compound C1CC=CS1 OXBLVCZKDOZZOJ-UHFFFAOYSA-N 0.000 description 1
- OYJGEOAXBALSMM-UHFFFAOYSA-N 2,3-dihydro-1,3-thiazole Chemical compound C1NC=CS1 OYJGEOAXBALSMM-UHFFFAOYSA-N 0.000 description 1
- JKTCBAGSMQIFNL-UHFFFAOYSA-N 2,3-dihydrofuran Chemical compound C1CC=CO1 JKTCBAGSMQIFNL-UHFFFAOYSA-N 0.000 description 1
- KVVZVGBYNOBXKX-UHFFFAOYSA-N 2-(4-bromophenyl)-7,8-dimethoxy-1,1-dioxo-4,5-dihydro-1$l^{6},2-benzothiazepin-3-one Chemical compound O=S1(=O)C=2C=C(OC)C(OC)=CC=2CCC(=O)N1C1=CC=C(Br)C=C1 KVVZVGBYNOBXKX-UHFFFAOYSA-N 0.000 description 1
- YTBFATPZSKFULM-OZBLHQGLSA-M 2-[2-[2-[4-[(4r,5r)-3,3-dibutyl-7-(dimethylamino)-4-hydroxy-1,1-dioxo-4,5-dihydro-2h-1$l^{6},2-benzothiazepin-5-yl]phenoxy]ethoxy]ethoxy]ethyl-triethylazanium;iodide Chemical compound [I-].C1([C@@H]2C3=CC(=CC=C3S(=O)(=O)NC([C@@H]2O)(CCCC)CCCC)N(C)C)=CC=C(OCCOCCOCC[N+](CC)(CC)CC)C=C1 YTBFATPZSKFULM-OZBLHQGLSA-M 0.000 description 1
- MSWZFWKMSRAUBD-GASJEMHNSA-N 2-amino-2-deoxy-D-galactopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@H](O)[C@@H]1O MSWZFWKMSRAUBD-GASJEMHNSA-N 0.000 description 1
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 1
- XGRADHHYIPLNAO-UHFFFAOYSA-N 2-butyl-4,5-dihydro-3h-1$l^{6},2-benzothiazepine 1,1-dioxide Chemical compound O=S1(=O)N(CCCC)CCCC2=CC=CC=C21 XGRADHHYIPLNAO-UHFFFAOYSA-N 0.000 description 1
- 125000002941 2-furyl group Chemical group O1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- ONJRTQUWKRDCTA-UHFFFAOYSA-N 2h-thiochromene Chemical compound C1=CC=C2C=CCSC2=C1 ONJRTQUWKRDCTA-UHFFFAOYSA-N 0.000 description 1
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 description 1
- 125000003682 3-furyl group Chemical group O1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000001541 3-thienyl group Chemical group S1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- IDOWNTLVKUSVPG-UHFFFAOYSA-N 4,5-dimethoxy-1,1-dioxo-1lambda6,2-benzothiazepin-3-one Chemical compound COC1=C(C(NS(C2=C1C=CC=C2)(=O)=O)=O)OC IDOWNTLVKUSVPG-UHFFFAOYSA-N 0.000 description 1
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- GCIYRVIDUGZSAJ-UHFFFAOYSA-N 7,8-dimethoxy-4,5-dihydro-1,2-benzothiazepin-3-one Chemical class C1CC(=O)NSC2=C1C=C(OC)C(OC)=C2 GCIYRVIDUGZSAJ-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- 206010027654 Allergic conditions Diseases 0.000 description 1
- KYNSBQPICQTCGU-UHFFFAOYSA-N Benzopyrane Chemical compound C1=CC=C2C=CCOC2=C1 KYNSBQPICQTCGU-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 1
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 1
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 1
- 206010008120 Cerebral ischaemia Diseases 0.000 description 1
- 229920001661 Chitosan Chemical group 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 102000010907 Cyclooxygenase 2 Human genes 0.000 description 1
- 108010037462 Cyclooxygenase 2 Proteins 0.000 description 1
- YTBSYETUWUMLBZ-UHFFFAOYSA-N D-Erythrose Natural products OCC(O)C(O)C=O YTBSYETUWUMLBZ-UHFFFAOYSA-N 0.000 description 1
- YTBSYETUWUMLBZ-IUYQGCFVSA-N D-erythrose Chemical compound OC[C@@H](O)[C@@H](O)C=O YTBSYETUWUMLBZ-IUYQGCFVSA-N 0.000 description 1
- RGHNJXZEOKUKBD-MGCNEYSASA-N D-galactonic acid Chemical compound OC[C@@H](O)[C@H](O)[C@H](O)[C@@H](O)C(O)=O RGHNJXZEOKUKBD-MGCNEYSASA-N 0.000 description 1
- DSLZVSRJTYRBFB-LLEIAEIESA-N D-glucaric acid Chemical compound OC(=O)[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)=O DSLZVSRJTYRBFB-LLEIAEIESA-N 0.000 description 1
- RGHNJXZEOKUKBD-UHFFFAOYSA-N D-gluconic acid Natural products OCC(O)C(O)C(O)C(O)C(O)=O RGHNJXZEOKUKBD-UHFFFAOYSA-N 0.000 description 1
- HAIWUXASLYEWLM-UHFFFAOYSA-N D-manno-Heptulose Natural products OCC1OC(O)(CO)C(O)C(O)C1O HAIWUXASLYEWLM-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 1
- BUDQDWGNQVEFAC-UHFFFAOYSA-N Dihydropyran Chemical compound C1COC=CC1 BUDQDWGNQVEFAC-UHFFFAOYSA-N 0.000 description 1
- 206010056474 Erythrosis Diseases 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 238000010917 Friedel-Crafts cyclization Methods 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- DSLZVSRJTYRBFB-UHFFFAOYSA-N Galactaric acid Natural products OC(=O)C(O)C(O)C(O)C(O)C(O)=O DSLZVSRJTYRBFB-UHFFFAOYSA-N 0.000 description 1
- IAJILQKETJEXLJ-UHFFFAOYSA-N Galacturonsaeure Natural products O=CC(O)C(O)C(O)C(O)C(O)=O IAJILQKETJEXLJ-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 102000004286 Hydroxymethylglutaryl CoA Reductases Human genes 0.000 description 1
- 108090000895 Hydroxymethylglutaryl CoA Reductases Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 208000007976 Ketosis Diseases 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- HSNZZMHEPUFJNZ-UHFFFAOYSA-N L-galacto-2-Heptulose Natural products OCC(O)C(O)C(O)C(O)C(=O)CO HSNZZMHEPUFJNZ-UHFFFAOYSA-N 0.000 description 1
- 239000000867 Lipoxygenase Inhibitor Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 1
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- NJOCMWZSLCZJHH-UHFFFAOYSA-N N=S1N=CC=CC2=C1C=CC=C2 Chemical class N=S1N=CC=CC2=C1C=CC=C2 NJOCMWZSLCZJHH-UHFFFAOYSA-N 0.000 description 1
- 150000004660 O-thiocarbamates Chemical class 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical group CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 1
- 229910006069 SO3H Inorganic materials 0.000 description 1
- HAIWUXASLYEWLM-AZEWMMITSA-N Sedoheptulose Natural products OC[C@H]1[C@H](O)[C@H](O)[C@H](O)[C@@](O)(CO)O1 HAIWUXASLYEWLM-AZEWMMITSA-N 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical class OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 1
- 108010069201 VLDL Cholesterol Proteins 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- KPCZJLGGXRGYIE-UHFFFAOYSA-N [C]1=CC=CN=C1 Chemical group [C]1=CC=CN=C1 KPCZJLGGXRGYIE-UHFFFAOYSA-N 0.000 description 1
- PFRUBEOIWWEFOL-UHFFFAOYSA-N [N].[S] Chemical group [N].[S] PFRUBEOIWWEFOL-UHFFFAOYSA-N 0.000 description 1
- VREVKZLUMZQJRI-UHFFFAOYSA-N [SH2]=N Chemical compound [SH2]=N VREVKZLUMZQJRI-UHFFFAOYSA-N 0.000 description 1
- VCMSIVROLIHORO-UHFFFAOYSA-N [amino-ethoxy-(hydroxyamino)methyl]-(diethylamino)-[dimethylamino-(ethylamino)-(methylamino)methyl]-(methoxymethyl)azanium Chemical compound COC[N+](C(NCC)(N(C)C)NC)(C(NO)(N)OCC)N(CC)CC VCMSIVROLIHORO-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 150000001241 acetals Chemical class 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- IAJILQKETJEXLJ-RSJOWCBRSA-N aldehydo-D-galacturonic acid Chemical compound O=C[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O IAJILQKETJEXLJ-RSJOWCBRSA-N 0.000 description 1
- 150000001323 aldoses Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 1
- 125000005196 alkyl carbonyloxy group Chemical group 0.000 description 1
- 125000004644 alkyl sulfinyl group Chemical group 0.000 description 1
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 150000001408 amides Chemical group 0.000 description 1
- 125000003277 amino group Chemical class 0.000 description 1
- MDFFNEOEWAXZRQ-UHFFFAOYSA-N aminyl Chemical compound [NH2] MDFFNEOEWAXZRQ-UHFFFAOYSA-N 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 229940035676 analgesics Drugs 0.000 description 1
- 229920006318 anionic polymer Polymers 0.000 description 1
- 239000000730 antalgic agent Substances 0.000 description 1
- 230000001773 anti-convulsant effect Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 239000001961 anticonvulsive agent Substances 0.000 description 1
- 229960003965 antiepileptics Drugs 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 125000005199 aryl carbonyloxy group Chemical group 0.000 description 1
- 125000005161 aryl oxy carbonyl group Chemical group 0.000 description 1
- 125000000732 arylene group Chemical group 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- RFRXIWQYSOIBDI-UHFFFAOYSA-N benzarone Chemical compound CCC=1OC2=CC=CC=C2C=1C(=O)C1=CC=C(O)C=C1 RFRXIWQYSOIBDI-UHFFFAOYSA-N 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000002047 benzodioxolyl group Chemical group O1OC(C2=C1C=CC=C2)* 0.000 description 1
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 125000005874 benzothiadiazolyl group Chemical group 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000035587 bioadhesion Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000000480 butynyl group Chemical group [*]C#CC([H])([H])C([H])([H])[H] 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 230000001364 causal effect Effects 0.000 description 1
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 150000003841 chloride salts Chemical class 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 201000001883 cholelithiasis Diseases 0.000 description 1
- 150000001840 cholesterol esters Chemical class 0.000 description 1
- 125000003016 chromanyl group Chemical group O1C(CCC2=CC=CC=C12)* 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 208000029078 coronary artery disease Diseases 0.000 description 1
- 125000001047 cyclobutenyl group Chemical group C1(=CCC1)* 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 125000006003 dichloroethyl group Chemical group 0.000 description 1
- 125000004774 dichlorofluoromethyl group Chemical group FC(Cl)(Cl)* 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- 125000006001 difluoroethyl group Chemical group 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 210000002249 digestive system Anatomy 0.000 description 1
- 125000001070 dihydroindolyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000005982 diphenylmethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 230000001882 diuretic effect Effects 0.000 description 1
- 229940030606 diuretics Drugs 0.000 description 1
- 239000012039 electrophile Substances 0.000 description 1
- 239000002702 enteric coating Substances 0.000 description 1
- 238000009505 enteric coating Methods 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000004129 fatty acid metabolism Effects 0.000 description 1
- 230000002550 fecal effect Effects 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- 229960004279 formaldehyde Drugs 0.000 description 1
- 235000019256 formaldehyde Nutrition 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- DSLZVSRJTYRBFB-DUHBMQHGSA-N galactaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O DSLZVSRJTYRBFB-DUHBMQHGSA-N 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- FKNQCJSGGFJEIZ-UHFFFAOYSA-N gamma-methylpyridine Natural products CC1=CC=NC=C1 FKNQCJSGGFJEIZ-UHFFFAOYSA-N 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 239000000174 gluconic acid Substances 0.000 description 1
- 235000012208 gluconic acid Nutrition 0.000 description 1
- 229960002442 glucosamine Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- MNQZXJOMYWMBOU-UHFFFAOYSA-N glyceraldehyde Chemical compound OCC(O)C=O MNQZXJOMYWMBOU-UHFFFAOYSA-N 0.000 description 1
- 150000002337 glycosamines Chemical class 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 125000004995 haloalkylthio group Chemical group 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 125000006343 heptafluoro propyl group Chemical group 0.000 description 1
- 238000004896 high resolution mass spectrometry Methods 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 150000002443 hydroxylamines Chemical class 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical group OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 1
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 1
- 230000000055 hyoplipidemic effect Effects 0.000 description 1
- 230000000871 hypocholesterolemic effect Effects 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000003406 indolizinyl group Chemical group C=1(C=CN2C=CC=CC12)* 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000003978 infusion fluid Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 102000048260 kappa Opioid Receptors Human genes 0.000 description 1
- 150000002584 ketoses Chemical class 0.000 description 1
- CFHGBZLNZZVTAY-UHFFFAOYSA-N lawesson's reagent Chemical compound C1=CC(OC)=CC=C1P1(=S)SP(=S)(C=2C=CC(OC)=CC=2)S1 CFHGBZLNZZVTAY-UHFFFAOYSA-N 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 108010022197 lipoprotein cholesterol Proteins 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 125000005905 mesyloxy group Chemical group 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Natural products C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- GRVDJDISBSALJP-UHFFFAOYSA-N methyloxidanyl Chemical compound [O]C GRVDJDISBSALJP-UHFFFAOYSA-N 0.000 description 1
- 230000027939 micturition Effects 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 125000006682 monohaloalkyl group Chemical group 0.000 description 1
- 238000011294 monotherapeutic Methods 0.000 description 1
- 229940035363 muscle relaxants Drugs 0.000 description 1
- 239000003158 myorelaxant agent Substances 0.000 description 1
- PHWISQNXPLXQRU-UHFFFAOYSA-N n,n-dimethylcarbamothioyl chloride Chemical compound CN(C)C(Cl)=S PHWISQNXPLXQRU-UHFFFAOYSA-N 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000005702 oxyalkylene group Chemical group 0.000 description 1
- 125000006353 oxyethylene group Chemical group 0.000 description 1
- 230000008068 pathophysiological alteration Effects 0.000 description 1
- 125000005010 perfluoroalkyl group Chemical group 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 239000003444 phase transfer catalyst Substances 0.000 description 1
- 125000000286 phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 150000004885 piperazines Chemical class 0.000 description 1
- 125000000587 piperidin-1-yl group Chemical group [H]C1([H])N(*)C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 210000002381 plasma Anatomy 0.000 description 1
- 229940100467 polyvinyl acetate phthalate Drugs 0.000 description 1
- NNFCIKHAZHQZJG-UHFFFAOYSA-N potassium cyanide Chemical compound [K+].N#[C-] NNFCIKHAZHQZJG-UHFFFAOYSA-N 0.000 description 1
- 235000010333 potassium nitrate Nutrition 0.000 description 1
- 239000004323 potassium nitrate Substances 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 208000000876 primary bile acid malabsorption Diseases 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 125000001325 propanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 238000003908 quality control method Methods 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 239000000018 receptor agonist Substances 0.000 description 1
- 229940044601 receptor agonist Drugs 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000005932 reductive alkylation reaction Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000006798 ring closing metathesis reaction Methods 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 239000000932 sedative agent Substances 0.000 description 1
- 230000001624 sedative effect Effects 0.000 description 1
- HSNZZMHEPUFJNZ-SHUUEZRQSA-N sedoheptulose Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)[C@H](O)C(=O)CO HSNZZMHEPUFJNZ-SHUUEZRQSA-N 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000012321 sodium triacetoxyborohydride Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 1
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 description 1
- 125000000542 sulfonic acid group Chemical group 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid group Chemical class S(O)(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- WBWWGRHZICKQGZ-HZAMXZRMSA-N taurocholic acid Chemical compound C([C@H]1C[C@H]2O)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(=O)NCCS(O)(=O)=O)C)[C@@]2(C)[C@@H](O)C1 WBWWGRHZICKQGZ-HZAMXZRMSA-N 0.000 description 1
- 125000004213 tert-butoxy group Chemical group [H]C([H])([H])C(O*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 1
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 description 1
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 1
- 150000004044 tetrasaccharides Chemical class 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000001984 thiazolidinyl group Chemical group 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- WTVXIBRMWGUIMI-UHFFFAOYSA-N trifluoro($l^{1}-oxidanylsulfonyl)methane Chemical group [O]S(=O)(=O)C(F)(F)F WTVXIBRMWGUIMI-UHFFFAOYSA-N 0.000 description 1
- 125000004385 trihaloalkyl group Chemical group 0.000 description 1
- WRECIMRULFAWHA-UHFFFAOYSA-N trimethyl borate Chemical compound COB(OC)OC WRECIMRULFAWHA-UHFFFAOYSA-N 0.000 description 1
- CURCMGVZNYCRNY-UHFFFAOYSA-N trimethylazanium;iodide Chemical compound I.CN(C)C CURCMGVZNYCRNY-UHFFFAOYSA-N 0.000 description 1
- 150000004043 trisaccharides Chemical class 0.000 description 1
- 230000003827 upregulation Effects 0.000 description 1
- 208000019553 vascular disease Diseases 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 108020001588 κ-opioid receptors Proteins 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/56—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D281/00—Heterocyclic compounds containing rings of more than six members having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D281/02—Seven-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/08—Bridged systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/30—Phosphinic acids R2P(=O)(OH); Thiophosphinic acids, i.e. R2P(=X)(XH) (X = S, Se)
- C07F9/36—Amides thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/38—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se)
- C07F9/3804—Phosphonic acids RP(=O)(OH)2; Thiophosphonic acids, i.e. RP(=X)(XH)2 (X = S, Se) not used, see subgroups
- C07F9/3808—Acyclic saturated acids which can have further substituents on alkyl
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic System
- C07F9/02—Phosphorus compounds
- C07F9/28—Phosphorus compounds with one or more P—C bonds
- C07F9/54—Quaternary phosphonium compounds
- C07F9/5407—Acyclic saturated phosphonium compounds
Definitions
- the present invention relates to novel 1,2-benzothiazepines, derivatives and analogs thereof, pharmaceutical compositions containing them, and their use in medicine, particularly in the prophylaxis and/or treatment of hyperlipidemic diseases, conditions and/or disorders, such as those associated with atherosclerosis and/or hypercholesterolemia, in mammals.
- the ileal bile acid transport system is a putative pharmaceutical target for the treatment of hypercholesterolemia based on an interruption ofthe enterohepatic circulation with specific transport inhibitors.
- Kramer, et al "Intestinal Bile Acid Absorption", The Journal of Biological Chemistry. Vol. 268, No. 24, Issue of August 25, pp. 18035-18046, 1993.
- Hoechst Aktiengesellschaft discloses polymers of various naturally occurring constituents ofthe enterohepatic circulation system and their derivatives, including bile acid, which inhibit the physiological bile acid transport with the goal of reducing the LDL cholesterol level sufficiently to be effective as pharmaceuticals and, in particular for use as hypocholesterolemic agents. See, e.g., Canadian Patent Application Nos. 2,025,294; 2,078,588; 2,085,782; and 2,085,830; and EP Application Nos. 0 379 161; 0 549 967; 0 559 064; and 0 563 731.
- Selected benzothiepines are disclosed in world patent application number WO93/321146 for numerous uses including fatty acid metabolism and coronary vascular diseases.
- benzothiepines are known for use as hypolipaemic and hypocholesterolaemic agents, especially for the treatment or prevention of atherosclerosis as disclosed by application Nos. EP 508425, FR 2661676, and WO 92/18462, each of which is limited by an amide bonded to the carbon adjacent the phenyl ring ofthe fused bicyclo benzothiepine ring.
- WO96/16051 published May 30, 1996 describes certain 1,5- benzothiazepines as useful in the treatment of hyperlipidemic conditions.
- WO96/05188 published February 22, 1996 describes certain 1 ,4- benzothiazepines as useful in the treatment of hyperlipidemic conditions.
- U.S. Patent No. 5,350,761 describes hydroxylamine derivatives that generically encompass certain benzothiazepine compounds. These derivatives are described as lipoxygenase inhibitors useful in the treatment of inflammatory and allergic conditions.
- WO98/02432 published January 22, 1998 describes certain 5-(aryl-(N- containing-heterocyclyl)alkyl)benzothiazepines and aralkyl-(N-containing- heterocyclyl)alkyl)-benzothiazepines as useful for controlling micturition.
- WO97/03953 published February 6, 1997 describes certain sulfonylamino-substituted benzothiazepines as inhibitors ofthe enzyme cyclooxygenase II.
- WO95/21843 published August 17, 1995 describes certain benzothiazepines substituted with azacyclic condensed piperazines. These compounds are identified as kappa receptor agonists useful as analgesics and diuretics and for the treatment of cerebral ischaemia.
- EP338331 published October 25, 1989 describes certain 2- benzothiazepine-5-ones useful as muscle relaxants.
- a first aspect ofthe invention comprises novel 1,2- benzothiazepines that are effective agents for the prophylaxis and/or treatment of hyperlipidemic diseases, conditions and/or disorders.
- a second aspect ofthe invention comprises pharmaceutical compositions comprising the novel 1 ,2- benzothiazepines that are suitable for the prophylaxis and/or treatment of hyperlipidemic diseases, conditions and/or disorders.
- a third aspect ofthe invention comprises methods for the prophylaxis and/or treatment of hyperlipidemic diseases, conditions and/or disorders comprising administering to a subject a prophylactically or therapeutically effective amount of one ofthe novel 1,2- benzothiazepines.
- a fourth aspect ofthe invention comprises methods of making the novel 1,2-benzothiazepines ofthe present invention. Additional aspects ofthe invention are discussed throughout the specification of this application.
- R 1 and R 2 are independently selected from the group consisting of hydrogen and hydrocarbyl, wherein said hydrocarbyl may be optionally substituted with one or more groups comprising one or more heteroatoms, and wherein said hydrocarbyl optionally may have one or more carbon atoms replaced by one or more heteroatoms independently selected from the group consisting of oxygen, nitrogen, sulfur and phosphorus;
- R and R are independently selected from the group consisting of hydrogen; hydrocarbyl; -OR 9 ; -NR 9 R 10 ; -SR 9 ; -S(O)R 9 ; -SO2R 9 ; and -
- R 9 and R 10 are independently selected from the group consisting of hydrogen; hydrocarbyl; amino; and hydrocarbylamino; wherein said hydrocarbyl moieties may be optionally substituted with one or more groups comprising one or more heteroatoms, and wherein said hydrocarbyl moieties optionally may have one or more carbon atoms replaced by one or more heteroatoms independently selected from the group consisting of oxygen, nitrogen, sulfur and phosphorus; and
- R and R are independently selected from the group consisting of hydrogen; -CN; halogen; oxo; hydrocarbyl; -OR 9 ; -NR 9 R 1 ; -
- hydrocarbyl may be optionally substituted with one or more groups comprising one or more heteroatoms, and wherein said hydrocarbyl optionally may have one or more carbon atoms replaced by one or more heteroatoms independently selected from the group consisting of oxygen, nitrogen, sulfur and phosphorus; or R 11 and R12 together with the carbon atom to which they are attached form a cyclic ring; and
- R and R are independently selected from the group consisting of hydrogen; alkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; -OR 9 ; -SR 9 ; -S(O)R 9 ; -SO2R 9 ; and -SO3R 9 ; wherein the R and R radicals optionally may be substituted with one or more radicals independently selected from the group consisting of halogen; -NO2; -CN; oxo; hydrocarbyl; -OR 13 ; -NR 13 R 14 ; -SR 13 ; -
- 1 T consisting of hydrogen; halogen; -CN; -N ⁇ 2; hydrocarbyl; -OR ; -
- NR 13 OR 14 ; -NR 13 NR 14 R 15 ; -CO2R 13 ; -OM; -SO2OM; -SO2NR 13 R 14 ; -
- R 1 and R 2 are independently selected from the group consisting of hydrogen; alkyl; cycloalkyl; alkenyl; cycloalkenyl; alkynyl; aryl; heterocyclyl; arylalkyl; heterocyclylalkyl; alkoxyalkyl; alkoxyalkenyl; alkoxy alkynyl; aryloxyalkyl; aryloxyalkenyl; aryloxyalkynyl; heterocy ley loxy alkyl; heterocycloxyalkenyl; heterocyclyloxyalkynyl; alkylaryl; and (polyalkyl)aryl; or R 1 and R 2 taken together with the carbon to which they are attached form C 3 -C]o cycloalkyl or C 3 -C 10 cycloalkenyl; wherein the R 1 and R 2 alkyl; cycloalkyl; alkenyl; cycloalkenyl; alkynyl; aryl
- R , R , and R w are independently selected from the group consisting of hydrogen; alkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; alkylammoniumalkyl; arylalkyl; heterocyclylalkyl; carboxyalkyl; alkoxyalkyl; carboalkoxyalkyl; carboxyaryl; carboxyheterocyclyl; amino; alkylamino; carboxyalkylamino; alkoxyalkylamino; and acyl; or wherein A " is a pharmaceutically acceptable anion; and
- R and R are independently selected from the group consisting of hydrogen; alkyl; alkenyl; alkynyl; aryl; heterocyclyl; -OR ; -NR R ; - SR 9 ; -S(O)R 9 ; -SO2R 9 ; and -SO3R 9 ; or
- R 11 and R 12 together with the carbon atom to which they are attached form a cyclic ring; and wherein R 9 and R 10 are as defined above; and R and R are independently selected from the group consisting of hydrogen; alkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; -OR 9 ; -SR 9 ; -S(O)R 9 ; -SO2R 9 ; and -SO3R 9 ; wherein the R and R alkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; and quaternary heterocyclyl radicals optionally may be substituted with one or more radicals independently selected from the group consisting of halogen; -CN; -NO2; oxo; alkyl; polyalkyl; haloalkyl; hydroxyalkyl; cycloalkyl; alkenyl; alky
- 7 optionally may have one or more carbons replaced by -O-; -NR -; -
- R 7 R 8 A " -; -S-; -SO-; -SO2-; -S + R 7 A--; -PR 7 -; -P(O)R 7 -; -P + R 7 R 8 A " -; or phenylene; and wherein R 7 and R 8 are independently selected from the group consisting of hydrogen; alkyl, alkenyl; alkynyl; aryl; and heterocyclyl; and wherein R , R , and R are independently selected from the group consisting of hydrogen; alkyl; haloalkyl; cycloalkyl; polyalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; quaternary heterocyclylalkyl; alkylarylalkyl; alkylheterocyclylalkyl; alkylammoniumalkyl; aminoalkyl; amino
- NR 13 OR 14 ; -NR 13 NR 14 R 15 ; -CO2R 13 ; -OM; -SO2OM; -SO2NR 13 R 14 ; - NR 14 C(O)R 13 ; -C(O)NR 13 R 14 ; -C(O)OM; -COR 13 ; -OR 18 ; - S(O) n NR 13 R 14 ; -NR 13 R 18 ; -NR 18 OR 14 ; -N + R 13 R 14 R 15 A " ; -PR 13 R 14 ; -P(O)R 13 R 14 ; -P + R 13 R 14 R 15 A " ; amino acid residue; peptide residue; polypeptide residue; and carbohydrate residue; wherein the R x alkyl; cycloalkyl; polyalkyl; haloalkyl; hydroxyalkyl; alkenyl; alkynyl; aryl; heterocyclyl; arylalkyl; hetero
- R" quaternary heterocyclyl radical optionally may be substituted with one or more radicals selected from the group consisting of halogen; -CN; -NO2; oxo; alkyl; cycloalkyl; polyalkyl; haloalkyl; hydroxyalkyl; alkenyl; alkynyl; aryl; heterocyclyl; arylalkyl; heterocyclylalkyl; polyether; -OR 13 ; -NR 13 R 14 ; -SR 13 ; -S(O)R 13 ; - SO2R 13 ; -S ⁇ 3R 13 ; -NR 13 OR 14 ; -NR 13 NR 14 R 15 ; -CO2R 13 ; OM; - SO2OM; -SO2NR 13 R 14 ; -C(O)NR 13 R 14 ; -C(O)OM
- R 18 is selected from the group consisting of alkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; acyl; alkoxycarbonyl; arylalkoxycarbonyl; and heterocyclylalkoxycarbonyl; and wherein the R 18 alkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; acyl; alkoxycarbonyl; arylalkoxycarbonyl; and heterocyclylalkoxycarbonyl radicals optionally may be substituted with one or more radicals selected from the group consisting of halogen; -CN ; NO 2 ; oxo; -OR 9 ; -NR 9 R 10 ; -N ⁇ R 1 ⁇ A " ;
- R 5 and R 6 preferably are independently selected from the group consisting of H; aryl; heterocyclyl; and quaternary heterocyclyl; wherein the R and R aryl; heterocyclyl; and quaternary heterocyclyl; radicals optionally may be substituted with one or more radicals independently selected from the group consisting of halogen; -CN; -NO2; oxo; alkyl; polyalkyl; haloalkyl; hydroxyalkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; polyether; -OR 13 ; -NR 13 R 14 ; -SR 13 ; -S(O)R 13 ; - SO2R 13 ; -S ⁇ 3R 13 ; -NR 13 OR 14 ; -NR 13 NR 14 R 15 ; -
- R 7 and R 8 are independently selected from the group consisting of hydrogen; alkyl, alkenyl; alkynyl; aryl; and heterocyclyl; and wherein R , R , and R are independently selected from the group consisting of hydrogen; alkyl; haloalkyl; cycloalkyl; polyalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; quaternary heterocyclylalkyl; alkylarylalkyl; alkylheterocyclylalkyl; alkylammoniumalkyl; aminocarbonylalkyl
- R 9 consisting of R and M; and wherein M is a pharmaceutically acceptable cation; and wherein R 9 , R 10 , R n , R 12 , R w , and A " are as previously set forth above for the compounds of Formula I.
- R 5 or R 6 has the formula -Ar-(R y ) t wherein: t is an integer from 0 to 5; Ar is selected from the group consisting of phenyl; thiophenyl; pyridyl; piperazinyl; piperonyl; pyrrolyl; naphthyl; furanyl; anthracenyl; quinolinyl; isoquinolinyl; quinoxalinyl; imidazolyl; pyrazolyl; oxazolyl; isoxazolyl; pyrimidinyl; thiazolyl; triazolyl; isothiazolyl; indolyl; benzoimidazolyl; benzoxazolyl; benzothiazolyl; and benzoisothiazolyl; and one or more R ⁇ are independently selected from the group consisting of halogen; -CN; -NO2; oxo; alkyl; polyalkyl;
- R 9 consisting of R and M; and wherein M is a pharmaceutically acceptable cation; and wherein R 9 , R 10 , R", R 12 , R w , and A " are as previously set forth above for the compounds of Formula I.
- At least one of R 5 or R 6 has the formula (II)
- R y and t are defined as above.
- Formula I preferably satisfy at least one or more ofthe following additional conditions:
- R 1 and R 2 are independently selected from the group consisting of hydrogen, alkyl and (C 3 . 10 )cycloalkyl.
- R 1 and R 2 are independently selected from the group consisting of hydrogen and (C,_ 10 )alkyl. More preferably, R 1 and R 2 are independently selected from the group consisting of (C,., 0 )alkyl. Still more preferably, R 1 and R 2 are independently selected from the group consisting of (C ⁇ . 7 )alkyl. Still more preferably, R 1 and R 2 are independently selected from the group consisting of (C 2 . 4 )alkyl. Still more preferably, R 1 and R 2 are the same (C 2 . 4 )alkyl. Still more preferably, R 1 and R 2 are each n-butyl; and/or
- R 3 and R 4 are independently selected from the group consisting of hydrogen and -OR 9 wherein R 9 is defined as previously set forth above for the compounds of Formula I.
- R 3 is hydrogen and R 4 is -OR 9
- R 3 is hydrogen and R 4 is hydroxy.
- the hydroxy group is in a syn relationship to the structure of Formula II; and/or
- R 5 is phenyl substituted with a radical selected from the group consisting of -OR 13 , -NR 13 R 14 , -NR 13 C(O)R 14 , -NR 13 C(O)NR 14 R 15 , - NR 13 CO 2 R 14 , -OC(O)R 13 , -OC(O)NR 13 R 14 , -NR 13 SOR 14 , -NR 13 SO 2 R 14 , - NR 13 SONR 1 R 15 , and -NR 13 SO 2 NR 1 R 15 wherein R 13 , R 14 and R 15 are as previously set forth above for the compounds of Formula I.
- R 5 is phenyl substituted with -OR 13 or -NR 13 C(O)R 14 . Still more preferably, R 5 is phenyl substituted at the para or meta position with -OR 13 wherein R 13 comprises a quaternary heterocyclyl, quaternary heterocyclylalkyl or alkylammoniumalkyl, or R 5 is phenyl substituted at the para or meta position with -NR 13 C(O)R 14 wherein R 13 is hydrogen and R 14 comprises a quaternary heterocyclyl, quaternary heterocyclylalkyl or alkylammoniumalkyl; and/or
- R 6 is hydrogen
- R N is selected from the group consisting of hydrogen, alkyl and aralkyl.
- R N is selected from the group consisting of hydrogen, (C 0 )alkyl and aryl(C 1 . 10 )alkyl. More preferably, R N is selected from the group consisting of hydrogen, methyl, ethyl and benzyl. Still more preferably, R N is hydrogen; and/or
- R x is independently selected from the group consisting of -OR 13 , - NR 13 R 14 , -N + R 13 R 14 R ,5 A ⁇ and polyether. More preferably, R x is selected from the group consisting of -OR 13 and -NR 13 R 14 . Still more preferably, R x is selected from the group consisting of alkoxy, amino, alkylamino and dialkylamino. Still more preferably, R x is selected from the group consisting of methoxy and dimethylamino; and or
- R x are present at the 7-, 8- or 9-position ofthe benzo ring ofthe structure of Formula I.
- said R" are present at the 7- and 9-positions ofthe benzo ring ofthe structure of Formula I.
- R x is present at the 7-position ofthe benzo ring ofthe structure of Formula I; and/or
- q is 1, 2 or 3.
- q is 1 or 2, and more preferably q is 1; and/or
- the compounds of Formula I satisfy at least one or more ofthe above-described conditions and R 5 comprises a carbohydrate residue.
- a more preferred class of compounds comprises those compounds of Formula I wherein: q is an integer from 1 to 4;
- R 1 and R 2 are independently selected from the group consisting of hydrogen; alkyl; cycloalkyl; alkenyl; alkynyl; arylalkyl; alkoxyalkyl; alkoxyalkenyl; alkoxyalkynyl; alkylaryl; and (polyalkyl)aryl; or
- R 1 and R 2 taken together with the carbon to which they are attached form C 3 -C 10 cycloalkyl or C 3 -C 10 cycloalkenyl; and wherein the R 1 and R 2 alkyl; cycloalkyl; alkenyl; alkynyl; arylalkyl; alkoxyalkyl; alkoxyalkenyl; alkoxyalkynyl; alkylaryl; and (polyalkyl)aryl radicals optionally may be substituted with one or more radicals selected from the group consisting of -CN; halogen; oxo; -OR 9 ; -NR 9 R 10 ; -
- R 1 and R 2 alkyl; cycloalkyl; alkenyl; alkynyl; arylalkyl; alkoxyalkyl; alkoxyalkenyl; alkoxyalkynyl; alkylaryl; and (polyalkyl)aryl radicals optionally may have one or more carbons replaced by -O-; -NR ,9" N N ++ RR 99 RR 1100 AA __.. __ ss __.. _ so _. _ s ⁇ 2 _.
- R , R , and R w are independently selected from the group consisting of hydrogen; alkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; alkylammoniumalkyl; arylalkyl; heterocyclylalkyl; carboxyalkyl; carboalkoxyalkyl; carboxyheterocyclyl; carboxyalkylamino; and acyl; and wherein A " is a pharmaceutically acceptable anion; and
- R and R are independently selected from the group consisting of
- 7 optionally may have one or more carbons replaced by -O-; -NR -; -
- R 7 and R 8 are independently selected from the group consisting of hydrogen and alkyl; and wherein R , R , and R are independently selected from the group consisting of hydrogen; alkyl; haloalkyl; cycloalkyl; polyalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; quaternary heterocyclylalkyl; alkylarylalkyl; alkylheterocyclylalkyl; alkylammoniumalkyl; carboxyalkylaminocarbonylalkyl; and polyether; or wherein R 13 and R 14
- R 9 consisting of R and M; and wherein M is a pharmaceutically acceptable cation; and wherein R 9 , R 10 , R 11 , R 12 , R w , and A " are as defined above; and
- R is selected from the group consisting of hydrogen; alkyl; alkenyl; alkynyl; and aralkyl; and one or more R x radicals are independently selected from the group consisting of hydrogen; halogen; -CN; -NO2; alkyl; cycloalkyl; polyalkyl; haloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; pojyether; acyloxy; -OR 13 ; -NR 13 R 14 ; -SR 13 ; -S(O)R 13 ; - S(O)2R 13 ; -SO3R 13 ; -S + R 13 R 14 A-; -NR 13 OR 14 ; -NR 13 NR 14 R 15 ; -
- R x radicals comprising carbon optionally may have one or more carbons replaced by -O-; -NR 13 -; -N + R 13 R 14 A -; -S-; -SO-; -SO2-; -S + R 13 A " -; -PR 13 -; -P(O)R 13 -; -PR 13 -; -P + R 13 R 1 A " -; phenylene; amino acid; peptide; polypeptide; carbohydrate; polyether; or polyalkyl; wherein said phenylene; amino acid; peptide; polypeptide; carbohydrate; and
- polyalkyl optionally may have one or more carbons replaced by -O-; -NR -;
- R 18 is selected from the group consisting of alkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; acyl; alkoxycarbonyl; arylalkoxycarbonyl; and heterocyclylalkoxycarbonyl; and wherein the R 18 alkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; acyl; alkoxycarbonyl; arylalkoxycarbonyl; and heterocyclylalkoxycarbonyl radicals
- a class of compounds of interest comprises those compounds of Formula I wherein: q is an integer from 1 to 4;
- R 1 and R 2 are independently selected from the group consisting of hydrogen; (C,-C 10 )alkyl; (C 3 -C 10 )cycloalkyl; (C 2 -C 10 )alkenyl; (C 2 -
- R , R , and R w are independently selected from the group consisting of hydrogen; (C r C 10 )alkyl; (C 3 -C 10 )cycloalkyl; (C 2 -C 10 )alkenyl; (C 2 -C 10 )alkynyl; aryl; heterocyclyl; ammonium(C 1 -C 10 )alkyl; (C r C ⁇ o)alkylammonium(C 1 -C 10 )alkyl; aryl(C !
- R and R are independently selected from the group consisting of hydrogen; (C,-C 10 )alkyl; (C 2 -C )0 )alkenyl; (C 2 -C 10 )alkynyl; aryl; heterocyclyl; -OR 9 ; -NR 9 R 10 ; -SR 9 ; -S(O)R 9 ; -SO2R 9 ; and -SO3R 9 ; or
- R 11 and R 12 are independently selected from the group consisting of hydrogen; -CN; halogen; oxo; (C,-C 10 )alkyl; (C 2 -C 10 )alkenyl; (C 2 -C 10 )alkynyl; aryl; heterocyclyl; aryl(C,-C 10 )alkyl; carboxy(C r C 10 )alkyl; carbo(C 1 -C 10 )alkoxy(C 1 -C 10 )alkyl; (C 3 -C 10 )cycloalkyl; cyano(C,-C 10 )alkyl; - OR 9 ; -NR 9 R 10 ; -SR 9 ; -S(O)R 9 ; -SO2R 9 ; -SO3R 9 ; -CO2R 9 ; and - CONR 9 R 10 ; or i i 1
- R and R together with the carbon atom to which they are attached form a cyclic ring; and wherein R 9 and R 10 are as defined above; and
- R and R are independently selected from the group consisting of hydrogen; (C r C 10 )alkyl; (C 3 -C 10 )cycloalkyl; (C 2 -C, 0 )alkenyl; (C 2 -
- R 5 and R 6 (C,-C ⁇ 0 )alkyl; (C 3 -C 10 )cycloalkyl; (C 2 -
- C 10 )alkenyl; (C 2 -C I0 )alkynyl; aryl; heterocyclyl; and quaternary heterocyclyl radicals optionally may be substituted with one or more radicals independently selected from the group consisting of halogen; -CN; -NO2; oxo; (C,-C 10 )alkyl; polyalkyl; halo(C,-C 10 )alkyl; (C 3 -C 10 )cycloalkyl; (C 2 - C 10 )alkenyl; (C 2 -C 10 )alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; aryl(C,-C 10 )alkyl; heterocyclyl(C r C 10 )alkyl; polyether; -OR 13 ; -NR 13 R 14 ; -SR 13 ; -S(O)R 13 ; -SO2R 13 ; -SO3R
- 7 + 7 8 may have one or more carbons replaced by -O-; -NR -; -N R R A " -; -S-; -
- R 7 and R 8 are independently selected from the group consisting of hydrogen and (C r C 10 )alkyl; and wherein R , R , and R are independently selected from the group consisting of hydrogen; (C,-C 10 )alkyl; halo(C,-C 10 )alkyl; (C 3 -
- C 10 cycloalkyl; polyalkyl; (C 2 -C 10 )alkenyl; (C 2 -C 10 )alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; aryl(C 1 -C 10 )alkyl; heterocyclyl(C r
- C 10 alkyl
- quaternary heterocyclyl(C r C 10 )alkyl quaternary heterocyclyl(C r C 10 )alkyl; (C 1 -C 10 )alkylaryl(C 1 - C 10 )alkyl; (C 1 -C 10 )alkylheterocyclyl(C I -C I0 )alkyl; (C r
- C 10 alkylaminocarbonyl(C r C I0 )alkyl
- polyether radicals optionally may have one or more carbons replaced by -O-; -NR 9 -; -N R R A'-; -S-; -SO-; -SO 2 -; -S + R 9 A -; -PR 9 -; -P + R 9 R 10 A " -; -P(O)R 9 -; phenylene; carbohydrate residue; amino acid residue; peptide residue; or polypeptide residue; and
- R and R are independently selected from the group
- R is selected from the group consisting of hydrogen; (C r C 10 )alkyl; (C 2 -C 10 )alkenyl; (C 2 -C, 0 )alkynyl; and aryl(C,-C 10 )alkyl; and one or more R x radicals are independently selected from the group consisting of hydrogen; halogen; -CN; -NO2; (C r C 10 )alkyl; (C 3 - C 10 )cycloalkyl; polyalkyl; halo(C r C 10 )alkyl; (C 2 -C 10 )alkenyl; (C 2 - C 10 )alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; aryl(C 1 -C 10 )alkyl; polyether; acyloxy; -OR 13 ; -NR 13 R 14 ; -SR 13 ; -S(O)R 13 ; -S
- R 1 is selected from the group consisting of (C,-C 10 )alkyl; heterocyclyl; quaternary heterocyclyl; aryl(C r C 10 )alkyl; acyl; and aryl(C r
- R 18 (C r C 10 )alkyl; heterocyclyl; quaternary heterocyclyl; aryl(C r C 10 )alkyl; acyl; and aryl(C r C 10 )alkoxycarbonyl radicals optionally may be substituted with one or more radicals selected from the group consisting of halogen; -CN ; oxo; -OR 9 ; -NR 9 R 10 ; -N ⁇ R 1 ⁇ A " ; -SR 9 ;
- a class of compounds of particular interest comprises those compounds of Formula I wherein: q is an integer from 1 to 4;
- R 1 and R 2 are independently selected from the group consisting of hydrogen, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, pentyl, hexyl, phenoxymethylene, phenoxyethylene, phenoxypropylene, pyridinyloxymethylene, pyridinyloxyethylene; methylpyridinyloxymethylene, methylpyridinyloxyethylene, pyrimidinyloxymethylene, and pyrimidinyloxyethylene; or
- R 1 and R 2 taken together with the carbon to which they are attached form cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl;
- R and R are independently selected from the group consisting of hydrogen, hydroxy, methyl, ethyl, phenyl, pyridinyl, amino, methylamino, dimethylamino, ethylamino and diethylamino; and
- R and R 6 are independently selected from the group consisting of hydrogen, phenyl, chlorophenyl, fluorophenyl, bromophenyl, iodophenyl, hydroxyphenyl, methoxyphenyl, ethoxyphenyl, methoxy(chlorophenyl), methoxy(fluorophenyl), methoxy(bromophenyl), methoxy(iodophenyl), ethoxy(chlorophenyl), ethoxy(fluorophenyl), ethoxy(bromophenyl), ethoxy(iodophenyl), nitrophenyl, aminophenyl, methylaminophenyl, dimethylaminophenyl, ethylaminophenyl, diethylaminophenyl, trimethylammoniumphenyl, triethylammoniumphenyl, trimethylammoniummethylcarbonylaminophenyl, triethy
- R is selected from the group consisting of hydrogen, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl and benzyl; and one or more R x radicals are independently selected from the group consisting of hydroxy, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, methoxy, ethoxy, n-propoxy, isopropoxy, methylthio, methylsulfmyl, methylsulfonyl, ethylthio, ethylsulfmyl, ethylsulfonyl, amino, hydroxyamino, methylamino, dimethylamino, ethylamino, diethylamino, trimethylamrnonium, triethylammonium, N-methyl-
- a class of compounds of specific interest comprises those compounds of Formula I wherein: q is an integer from 1 to 4;
- R 1 and R 2 are independently selected from the group consisting of hydrogen and (C r C 10 )alkyl; or
- R 1 and R 2 taken together with the carbon to which they are attached form (C 3 -C 10 )cycloalkyl
- R and R are independently selected from the group consisting of hydrogen and hydroxy
- R is phenyl, wherein said phenyl is optionally substituted with one or more radicals independently selected from the group consisting of halogen; hydroxy; -N ⁇ 2; (C,-C 10 )alkyl; halo(C,-C, 0 )alkyl; aryl(C,-C I0 )alkyl; heterocyclyl(C,-C 10 )alkyl; polyether; -OR 13 ; -NR 13 R 14 ; and -NR 13 C(O)R 14 ; and wherein R , R , and R are independently selected from the group consisting of hydrogen; (C r C 10 )alkyl; halo(C,-C 10 )alkyl; heterocyclyl; quaternary heterocyclyl; aryl(C r C 10 )alkyl; heterocyclyl(C r C 10 )alkyl; quaternary heterocyclyl(C ,-Cj 0 )alkyl
- R and R are independently selected from the group consisting of hydrogen; (C r C ⁇ 0 )alkyl; heterocyclyl; ammonium(C r
- C 10 alkyl; (C 1 -C ⁇ o)alkylammonium(C 1 -C 10 )alkyl; aryl(C 1 -C 10 )alkyl; heterocyclyl(C r C 10 )alkyl; carboxy(C r C 10 )alkyl; carbo(C ] -C 10 )alkoxy(C 1 -
- R 11 and R 12 are independently selected from the group consisting of hydrogen; (C r C 10 )alkyl; heterocyclyl; aryl(C,-C 10 )alkyl; carboxy(C r C 10 )alkyl; and carbo(C 1 -C ⁇ 0 )alkoxy(C 1 -C 10 )alkyl; or
- R 11 and R 12 together with the carbon atom to which they are attached form a cyclic ring; and wherein R and R 16 are as previously set forth above for the compounds of Formula I;
- R 6 is hydrogen; and R R iiss sseelleecctteedd frfroorm the group consisting of hydrogen; (C 1 -C 10 )alkyl; and aryl(C r C 10 )alkyl; and one or more R x radicals are independently selected from the group consisting of hydrogen; -NO2; (C C ⁇ 0 )alkyl; halo(C,-C 10 )alkyl; -OR 13 ; - NR 13 R 14 ; wherein R 13 and R 14 are as defined above; or a pharmaceutically acceptable salt, solvate, or prodrug thereof; and provided that aryl is selected from the group consisting of optionally substituted phenyl, biphenyl and naphthyl; and provided that heterocyclyl is selected from the group consisting of optionally substituted heterocyclyl comprising a 5 to 10 membered ring and comprising one or more ring atoms that are heteroatoms selected from the group consisting of oxygen
- R and R taken together with the carbon to which they are attached form cyclopentyl; and one of R and R is hydrogen and the other of R and R is hydroxy;
- R is selected from the group consisting of phenyl, hydroxyphenyl, methoxyphenyl, ethoxyphenyl, nitrophenyl, aminophenyl, methylaminophenyl, dimethylaminophenyl, ethylaminophenyl, diethylaminophenyl, trimethylammoniumphenyl, triethylammoniumphenyl, trimethylammoniummethylcarbonylaminophenyl, triethylammoniummethylcarbonylaminophenyl, trimethylammoniumethylcarbonylaminophenyl, triethylammoniumethylcarbonylaminophenyl, trimethylammoniumpropylcarbonylaminophenyl, triethylammoniumpropylcarbonylaminophenyl, trimethylammoniumbutylcarbonylaminophenyl, triethylammoniumbutylcarbonylaminophenyl, methylcarbonylaminophen
- R is selected from the group consisting of hydrogen, methyl, ethyl, and benzyl; and one or more R x radicals are independently selected from the group consisting of hydroxy, methyl, ethyl, methoxy, ethoxy, amino, hydroxyamino, methylamino, dimethylamino, ethylamino, diethylamino, trimethylammonium, triethylammonium, N-methyl-N-carboxymethyl- amino, N,N-dimethyl-N-carboxymethyl-ammonium, methylcarbonylamino, chloromethylcarbonylamino, fluoromethylcarbonylamino, bromomethylcarbonylamino, iodomethylcarbonylamino, ethylcarbonylamino, benzyloxycarbonylamino, and aminoimidocarbonylamino; or a pharmaceutically acceptable salt, solvate, or prodrug thereof.
- a subclass of compounds of high interest comprises those compounds of Formula I wherein: wherein: q is 1 or 2;
- R 1 and R 2 are each independently alkyl
- R is hydroxy; R and R are hydrogen;
- R 5 has the formula (II):
- t is an integer from 0 to 5; one or more R ⁇ are independently selected from the group consisting of hydrogen; halogen; -CN; -NO2; oxo; alkyl; polyalkyl; haloalkyl; hydroxyalkyl; cycloalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; polyether; -OR ; -NR R ; - SR 13 ; -S(O)R 13 ; -SO2R 13 ; -SO3R 13 ; -NR 13 OR 14 ; -NR 13 NR 14 R 15 ; - CO2R 13 ; -OM; -SO2OM; -SO2NR 13 R 14 ; -C(O)NR 13 R 14 ; -C(O)OM; - COR 13 ; -NR 13 C(O)R 14 ; -C
- R 7 and R 8 are independently selected from the group consisting of hydrogen; alkyl, alkenyl; alkynyl; aryl; and heterocyclyl; and wherein R , R , and R are independently selected from the group consisting of hydrogen; alkyl; haloalkyl; cycloalkyl; polyalkyl; alkenyl; alkynyl; aryl; heterocyclyl; quaternary heterocyclyl; arylalkyl; heterocyclylalkyl; quaternary heterocyclylalkyl; alkylarylalkyl; alkylheterocyclylalkyl; alkylammoniumalkyl; aminocarbonylalkyl; alkylaminocarbonylalkyl; carboxy
- R 9 consisting of R and M; and wherein M is a pharmaceutically acceptable cation; and wherein R 9 , R 10 , R", R 12 , R , and A " are as previously set forth above for the compounds of Formula I; and
- R is selected from the group consisting of hydrogen; alkyl; and aralkyl; and one or more R radicals are independently selected from the group consisting of alkoxy, alkylamino and dialkylamino; or a pharmaceutically acceptable salt, solvate, or prodrug thereof.
- a family of specific compounds of particular interest within Formula I consists ofthe following compounds:
- the invention further comprises a compound selected from among:
- R 19 is selected from the group consisting of alkane diyl, alkene diyl, alkyne diyl, polyalkane diyl, alkoxy diyl, polyether diyl, polyalkoxy diyl, carbohydrate residue, amino acid residue, peptide residue, and polypeptide residue; and wherein alkane diyl, alkene diyl, alkyne diyl, polyalkane diyl, alkoxy diyl, polyether diyl, polyalkoxy diyl, carbohydrate residue, amino acid residue, peptide residue, and polypeptide residue optionally may have one or more carbon atoms replaced by -O-, -NR 7 -, -N + R 7 R 8 A ' -, -S-, -SO-, -SO 2 -, -S + R 7 N-, - PR 7 -, -PR 7 R 8 A " -, phenylene, heterocyclyl, quaternary heterocyclyl, or aryl; wherein alkane diyl
- R 19 can further comprise functional linkages by which R 19 is bonded to R 20 and/or R 21 in the compounds of Formula DI; to R 20 , R 21 and/or R 22 in the compounds of Formula DII; and to R 20 , R 21 , R 22 and or R 23 in the compounds of Formula Dili; and wherein each of R 20 , R 21 , or R 22 and R 23 comprises a benzothiazepine moiety as described above that is therapeutically effective in inhibiting ileal bile acid transport.
- R 19 substituents include, but are not limited to, the following:
- R 25 is selected from the group consisting of carbon and nitrogen; and R 26 , R 27 , R 28 , R 29 , R 30 , R 31 , R 32 , R 33 , R 34 , R 35 , R 36 , and R 37 are independently selected from the group consisting of: -CH .
- R 38 , R 39 , R 40 and R 41 are independently selected from the group consisting of alkyl, alkenyl, alkylaryl, aryl, arylalkyl, cycloalkyl, heterocyclyl, and heterocyclylalkyl;
- a " is a pharmaceutically acceptable anion; and h, i, j and k are independently selected from the group consisting of integers from 1 to 10 inclusive.
- the invention is also directed to a compound selected from among Formula DI, Formula DII and Formula Dili in which each of R 20 , R 21 , R 22 and R 23 comprises a benzothiazepine moiety corresponding to the Formula DIV or Formula DIVA:
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R N , R ⁇ q, and n are as previously defined above for the compounds of Formula I, and R 55 is either a covalent bond or arylene.
- R 20 , R 21 , R 22 , and R 23 in Formulae DI, DII and Dili be bonded at its 7- or 8- position to R 19 .
- R 55 comprise a phenylene moiety bonded at a m- or j9-carbon thereof to R 19 .
- Formula DI examples include:
- R 1A , R 2A , R 3A , R 4A , R NA , R yA , R , r and u have the same definitions as stated above for R 1 , R 2 , R 3 , R 4 , R NA , R y , R x , q and t, respectively.
- R 1 and R 2 can be, among other combinations, ethyl/butyl or butyl/butyl.
- Illustrative dimeric compounds include the following:
- a core moiety backbone, R 19 as discussed herein in Formulae DI, DII and Dili can be multiply substituted with more than four pendant active benzothiazepine units, i.e., R 20 , R 21 , R 22 , and R 23 as discussed above, through multiple functional groups within the core moiety backbone.
- the core moiety backbone unit, R 19 can comprise a single core moiety unit, multimers thereof, and multimeric mixtures ofthe different core moiety units discussed herein, i.e., alone or in combination.
- the number of individual core moiety backbone units can range from about one to about 100, preferably about one to about 80, more preferably about one to about 50, and even more preferably about one to about 25.
- the number of points of attachment of similar or different pendant active benzothiazepine units within a single core moiety backbone unit can be in the range from about one to about 100, preferably about one to about 80, more preferably about one to about 50, and even more preferably about one to about 25.
- Such points of attachment can include bonds to C, S, O, N, or P within any ofthe groups encompassed by the definition of R 19 .
- the more preferred benzothiazepine moieties comprising R 20 , R 21 , R 22 and/or R 23 conform to the preferred structures as outlined above for Formula I.
- the 3 -position carbon on each benzothiazepine moiety can be achiral, and the substituents R 1 , R 2 , R 3 , R 4 , R 5 and R x can be selected from the prefened groups and combinations of substituents as discussed above.
- the core structures can comprise, for example, poly(oxyalkylene) or oligo(oxyalkylene), especially poly- or oligo(oxyethylene) or poly- or oligo(oxypropylene).
- the present invention provides a pharmaceutical composition for the prophylaxis and/or treatment of a disease, condition and/or disorder for which a bile acid transport inhibitor is indicated, such as a hyperlipidemic condition, for example, atherosclerosis.
- a bile acid transport inhibitor such as a hyperlipidemic condition, for example, atherosclerosis.
- Such compositions comprise any ofthe compounds disclosed above, alone or in combination, in an amount effective to reduce bile acid levels in the blood, or to reduce transport thereof across digestive system membranes, alone or in a composition comprising, for example, one or more pharmaceutically acceptable carriers, excipients, and/or diluents.
- the benzothiazepine compounds of the present invention can be used alone or in various combinations.
- the present invention also provides a method of treating a disease, condition and/or disorder in mammals, including humans, for which a bile acid transport inhibitor is indicated, comprising administering to a patient in need thereof a compound ofthe present invention in an effective amount in unit dosage form or in divided doses.
- the present invention comprises the use ofthe compounds of Formula I and/or the dimeric or multimeric compounds of Formulae DI, DII and/or Dili in the preparation of a medicament useful for the prophylaxis and/or treatment of a disease, condition and/or disorder for which a bile acid transport inhibitor is indicated.
- the compounds of Formula I are also useful for the prophylaxis and/or treatment of gallstones.
- the present invention also provides processes for the preparation of compounds ofthe present invention.
- hydrocarbyl refers to radicals consisting exclusively ofthe elements carbon and hydrogen. These radicals include, for example, alkyl, cycloalkyl, alkenyl, cycloalkenyl, alkynyl, and aryl moieties. These radicals also include alkyl, cycloalkyl, alkenyl, cycloalkenyl, alkynyl, and aryl moieties substituted with other aliphatic or cyclic hydrocarbon groups, such as alkaryl, alkenaryl and alkynaryl. Preferably, these moieties comprise 1 to 20 carbon atoms.
- substituted hydrocarbyl refers to a hydrocarbyl radical that is substituted with a group comprising at least one atom other than carbon, such as but not limited to, halogen, oxygen, nitrogen, sulfur and phosphorus.
- substituted hydrocarbyl include hydrocarbyl radicals substituted with groups such as, but not limited to, lower alkoxy such as methoxy, ethoxy, and butoxy; halogen such as chloro and fluoro; ethers; acetals; ketals; esters; heterocyclyl such as furyl and thienyl; alkanoxy; . hydroxy; protected hydroxy; acyl; acyloxy; nitro; cyano; amino; and amido.
- Substituted hydrocarbyl also includes hydrocarbyl radicals in which a carbon chain atom is replaced with a heteroatom such as nitrogen, oxygen, sulfur, or a halogen.
- alkyl is used, either alone or within other terms such as “haloalkyl", and “hydroxyalkyl”, it embraces linear or branched radicals having one to about twenty carbon atoms or, preferably, one to about twelve carbon atoms. More prefened alkyl radicals are "lower alkyl” radicals having one to about six carbon atoms. Examples of such radicals include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, pentyl, iso- amyl, hexyl and the like. Even more prefened are lower alkyl radicals having one to three carbon atoms.
- alkenyl is used, either alone or within other terms such as “arylalkenyl”, it embraces linear or branched radicals having at least one carbon-carbon double bond of two to about twenty carbon atoms or, preferably, two to about twelve carbon atoms. More prefened alkenyl radicals are "lower alkenyl” radicals having two to about six carbon atoms. Examples of alkenyl radicals include ethenyl, propenyl, allyl, propenyl, butenyl and 4- methylbutenyl.
- alkenyl and lower alkenyl embrace radicals having “cis” and “trans” orientations, or alternatively, "E” and "Z” orientations.
- alkynyl denotes linear or branched radicals having two to about twenty carbon atoms or, preferably, two to about twelve carbon atoms. More prefened alkynyl radicals are "lower alkynyl” radicals having two to about ten carbon atoms. Most prefened are lower alkynyl radicals having two to about six carbon atoms. Examples of such radicals include propargyl, butynyl, and the like.
- cycloalkyl embraces saturated carbocyclic radicals having three to about twelve carbon atoms. More prefened cycloalkyl radicals are
- lower cycloalkyl radicals having three to about ten carbon atoms. Examples of such radicals include cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
- cycloalkyl additionally encompasses spiro systems wherein the cycloalkyl ring has a carbon ring atom in common with the seven-membered heterocyclic ring ofthe benzothiazepine.
- cycloalkenyl embraces partially unsaturated carbocyclic radicals having three to twelve carbon atoms. Cycloalkenyl radicals that are partially unsaturated carbocyclic radicals that contain two double bonds (that may or may not be conjugated) can be called “cycloalkyldienyl". More prefened cycloalkenyl radicals are "lower cycloalkenyl” radicals having four to about ten carbon atoms. Examples of such radicals include cyclobutenyl, cyclopentenyl and cyclohexenyl.
- halo and halogen means halogens such as fluorine, chlorine, bromine or iodine atoms.
- haloalkyl embraces radicals wherein any one or more ofthe alkyl carbon atoms is substituted with halo as defined above. Specifically embraced are monohaloalkyl, dihaloalkyl and polyhaloalkyl radicals.
- a monohaloalkyl radical for one example, may have either an iodo, bromo, chloro or fluoro atom within the radical.
- Dihalo and polyhaloalkyl radicals may have two or more ofthe same halo atoms or a combination of different halo radicals.
- “Lower haloalkyl” embraces radicals having one to six carbon atoms.
- haloalkyl radicals include fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, pentafluoroethyl, heptafluoropropyl, difluorochloromethyl, dichlorofluoromethyl, difluoroethyl, difluoropropyl, dichloroethyl and dichloropropyl.
- "Perfluoroalkyl” means alkyl radicals having all hydrogen atoms replaced with fluoro atoms. Examples include trifluoromethyl and pentafluoroethyl.
- hydroxyalkyl embraces linear or branched alkyl radicals having one to about ten carbon atoms any one of which may be substituted with one or more hydroxyl radicals. More prefened hydroxyalkyl radicals are "lower hydroxyalkyl” radicals having one to six carbon atoms and one or more hydroxyl radicals. Examples of such radicals include hydroxymethyl, hydroxyethyl, hydroxypropyl, hydroxybutyl and hydroxyhexyl. Even more prefened are lower hydroxyalkyl radicals having one to three carbon atoms.
- aryl alone or in combination, means a carbocyclic aromatic system containing one or more rings wherein such rings may be attached together in a pendent manner or may be fused.
- aryl embraces aromatic radicals such as phenyl, naphthyl, tetrahydronaphthyl, indane, biphenyl, and anthracenyl. More prefened aryl is phenyl.
- Said "aryl” group may have one to three substituents such as lower alkyl, hydroxy, halo, haloalkyl, nitro, cyano, alkoxy and lower alkylamino.
- heterocyclyl embraces saturated, partially saturated and unsaturated heteroatom-containing ring-shaped radicals, where the heteroatoms may be selected from nitrogen, sulfur and oxygen.
- Prefened heterocyclyl are 3-10 membered ring heterocyclyl, particularly 5-8 membered ring heterocyclyl.
- saturated heterocyclic radicals include saturated 3 to 6-membered heteromonocychc groups containing 1 to 4 nitrogen atoms [e.g. pynolidinyl, imidazolidinyl, piperidino, piperazinyl]; saturated 3 to 6-membered heteromonocychc groups containing 1 to 2 oxygen atoms and 1 to 3 nitrogen atoms [e.g.
- heterocyclyl saturated 3 to 6-membered heteromonocychc groups containing 1 to 2 sulfur atoms and 1 to 3 nitrogen atoms [e.g., thiazolidinyl].
- partially saturated heterocyclyl radicals include dihydrothiophene, dihydropyran, dihydrofuran and dihydrothiazole.
- unsaturated heterocyclic radicals include unsaturated 5 to 6 membered heteromonocyclyl groups containing 1 to 4 nitrogen atoms, for example, pynolinyl, imidazolyl, pyrazolyl, 2-pyridyl, 3-pyridyl, 4-pyridyl, pyrimidyl, pyrazinyl, pyridazinyl, triazolyl [e.g., 4H-l,2,4-triazolyl, 1H-1,2,3- triazolyl, 2H- 1,2,3 -triazolyl]; unsaturated condensed heterocyclic groups containing 1 to 5 nitrogen atoms, for example, indolyl, isoindolyl, indolizinyl, benzimidazolyl, quinolyl, isoquinolyl, indazolyl, benzotriazolyl, tetrazolopyridaziny
- the term also embraces radicals where heterocyclic radicals are fused with aryl radicals.
- fused bicyclic radicals examples include benzofuran, benzothiophene, and the like.
- Said "heterocyclyl” group may have 1 to 3 substituents such as lower alkyl, hydroxy, oxo, amino and lower alkylamino.
- Heterocyclic radicals can include fused or unfused radicals, particularly 3-10 membered fused or unfused radicals.
- Prefened examples of heteroaryl radicals include benzofuryl, 2,3-dihydrobenzofuryl, benzothienyl, indolyl, dihydroindolyl, chromanyl, benzopyran, thiochromanyl, benzothiopyran, benzodioxolyl, benzodioxanyl, pyridyl, thienyl, thiazolyl, furyl, and pyrazinyl.
- More prefened heteroaryl radicals are 5- or 6-membered heteroaryl, containing one or two heteroatoms selected from sulfur nitrogen and oxygen, selected from thienyl, furanyl, thiazolyl, imidazolyl, pyrazolyl, isoxazolyl, isothiazolyl, pyridyl, piperidinyl and pyrazinyl.
- heteroaryl means a fully unsaturated heterocyclyl
- heterocyclyl or "heteroaryl”
- the point of attachment to the molecule of interest can be at the heteroatom or elsewhere within the ring.
- trimazolyl includes all positional isomers. In all other heterocyclyl and heteroaryl which contain more than one ring heteroatom and for which isomers are possible, such isomers are included in the definition of said heterocyclyl and heteroaryl.
- quaternary heterocyclyl means a heterocyclyl in which one or more ofthe heteroatoms, for example, nitrogen, sulfur, phosphorus or oxygen, has such a number of bonds that it is positively charged (and therefore the term is intended to encompass both ternary and quaternary positively charged structures).
- the point of attachment ofthe quaternary heterocyclyl to the molecule of interest can be at a heteroatom or elsewhere.
- quaternary heteroaryl means a heteroaryl in which one or more ofthe heteroatoms, for example, nitrogen, sulfur, phosphorus or oxygen, has such a number of bonds that it is positively charged (and therefore the term is intended to encompass both ternary and quaternary positively charged structures).
- the point of attachment ofthe quaternary heteroaryl to the molecule of interest can be at a heteroatom or elsewhere.
- diyl means a diradical moiety wherein said moiety has two points of attachment to molecules of interest.
- oxo means a doubly bonded oxygen.
- polyalkyl means a branched or straight hydrocarbon chain having a molecular weight up to about 20,000, more preferably up to about 10,000, and most preferably up to about 5,000.
- polyether means a polyalkyl wherein one or more carbons are replaced by oxygen, wherein the polyether has a molecular weight up to about 20,000, more preferably up to about 10,000, and most preferably up to about 5,000.
- polyalkoxy means a polymer of alkylene oxides, wherein the polyalkoxy has a molecular weight up to about 20,000, more preferably up to about 10,000, and most preferably up to about 5,000.
- carbohydrate residue encompasses residues derived from carbohydrates such as, but is not limited to, mono-, di-, tri-, tetra- and polysaccharides wherein the polysaccharides can have a molecular weight of up to about 20,000, for example, hydroxypropyl-methylcellulose or chitosan residue; compounds derived from aldoses and ketoses with 3 to 7 carbon atoms and which belong to the D- or L-series; aminosugars; sugar alcohols; and saccharic acids.
- Nonlimiting specific examples of such carbohydrates include glucose, mannose, fructose, galactose, ribose, erythrose, glycerinaldehyde, sedoheptulose, glucosamine, galactosamine, glucoronic acid, galacturonic acid, gluconic acid, galactonic acid, mannoic acid, glucamine, 3-amino-l,2- propanediol, glucaric acid and galactaric acid.
- peptide residue means polyamino acid residue containing up to about 100 amino acid units.
- polypeptide residue means a polyamino acid residue containing from about 100 amino acid units to about 1000 amino acid units, more preferably from about 100 amino acid units to about 750 amino acid untis, and most preferably from about 100 amino acid units to about 500 amino acid units.
- alkylammoniumalkyl means an an -NH 2 group or a mono-, di- or tri-substituted amino group, any of which is bonded to an alkyl wherein said alkyl is bonded to the molecule of interest.
- sulfo means a sulfo group, -SO 3 H, and its salts.
- sulfoalkyl means an alkyl group to which a sulfonate group is bonded, wherein said alkyl is bonded to the molecule of interest.
- aralkyl embraces aryl-substituted alkyl radicals.
- Preferable aralkyl radicals are "lower aralkyl” radicals having aryl radicals attached to alkyl radicals having one to six carbon atoms. Even more prefened are lower aralkyl radicals having phenyl attached to alkyl portions having one to three carbon atoms. Examples of such radicals include benzyl, diphenylmethyl and phenylethyl.
- the aryl in said aralkyl may be additionally substituted with halo, alkyl, alkoxy, halkoalkyl and haloalkoxy.
- arylalkenyl embraces aryl-substituted alkenyl radicals.
- Preferable arylalkenyl radicals are "lower arylalkenyl” radicals having aryl radicals attached to alkenyl radicals having
- heterocyclylalkyl means an alkyl radical that is substituted with one or more heterocyclyl groups.
- Preferable heterocyclylalkyl radicals are "lower heterocyclylalkyl” radicals having one or more heterocyclyl groups attached to an alkyl radical having one to ten carbon atoms.
- heteroarylalkyl means an alkyl radical that is substituted with one or more heteroaryl groups.
- Preferable heteroarylalkyl radicals are "lower heteroarylalkyl” radicals having one or more heteroaryl groups attached to an alkyl radical having one to ten carbon atoms.
- quaternary heterocyclylalkyl means an alkyl radical that is substituted with one or more quaternary heterocyclyl groups.
- Preferable quaternary heterocyclylalkyl radicals are "lower quaternary heterocyclylalkyl” radicals having one or more quaternary heterocyclyl groups attached to an alkyl radical having one to ten carbon atoms.
- quaternary heteroarylalkyl means an alkyl radical that is substituted with one or more quaternary heteroaryl groups.
- Preferable quaternary heteroarylalkyl radicals are "lower quaternary heteroarylalkyl” radicals having one or more quaternary heteroaryl groups attached to an alkyl radical having one to ten carbon atoms.
- alkylheteroarylalkyl means a heteroarylalkyl radical that is substituted with one or more alkyl groups.
- Preferable alkylheteroarylalkyl radicals are "lower alkylheteroarylalkyl” radicals with alkyl portions having one to ten carbon atoms.
- alkoxy means an alkyl radical which is attached to the molecule of interest by oxygen, such as a methoxy radical. More prefened alkoxy radicals are "lower alkoxy" radicals having one to six carbon atoms. Examples of such radicals include methoxy, ethoxy, propoxy, iso-propoxy, butoxy and tert-butoxy.
- carboxy means the carboxy group, -CO 2 H, or its salts.
- carboxyalkyl means an alkyl radical that is substituted with one or more carboxy groups.
- Preferable carboxyalkyl radicals are "lower carboxyalkyl” radicals having one or more carboxy groups attached to an alkyl radical having one to six carbon atoms.
- carboxyheterocyclyl means a heterocyclyl radical that is substituted with one or more carboxy groups.
- carboxyheteroaryl means a heteroaryl radical that is substituted with one or more carboxy groups.
- carboalkoxyalkyl means an alkyl radical that is substituted with one or more alkoxycarbonyl groups.
- Preferable carboalkoxyalkyl radicals are "lower carboalkoxyalkyl” radicals having one or more alkoxycarbonyl groups attached to an alkyl radical having one to six carbon atoms.
- Carboxyalkylamino means an amino radical that is mono- or di-substituted When used in combination, for example “alkylaryl” or “arylalkyl,” the individual terms listed above have the meaning indicated above.
- a bile acid transport inhibitor means a compound capable of inhibiting abso ⁇ tion of bile acids from the intestine into the circulatory system of a mammal, such as a human. This includes increasing the fecal excretion of bile acids, as well as reducing the blood plasma or serum concentrations of cholesterol and cholesterol ester, and more specifically, reducing LDL and VLDL cholesterol.
- Conditions and/or diseases that benefit from the prophylaxis and/or treatment by bile acid transport inhibition include, for example, a hyperlipidemic condition such as atherosclerosis.
- THF tetrahydrofuran
- PTC phase transfer catalyst
- Aliquart 336 means methyltricaprylylammonium chloride
- MCPBA means m-chloroperbenzoic acid
- Celite refers to a brand of diatomaceous earth filtering aid
- DMF means dimethylformamide
- DME ethylene glycol dimethyl ether
- Me means methyl
- Et means ethyl
- EtOAc means ethyl acetate
- Et 2 O means diethyl ether
- LAH lithium aluminum hydride
- DMSO dimethylsulfoxide
- KOSiMe 3 means potassium trimethylsilanolate
- PEG polyethylene glycol
- MS mass spectrometry
- HRMS high resolution mass spectrometry
- ES electrospray
- MPLC medium pressure liquid chromatography
- HPLC high pressure liquid chromatography
- RPHPLC reverse phase high pressure liquid chromatography
- RT room temperature
- the compounds ofthe present invention can have at least two asymmetrical carbon atoms, and therefore include racemates and stereoisomers, such as diastereomers and enantiomers, in both pure form and in admixture.
- stereoisomers can be prepared and separated using conventional techniques, either by reacting enantiomeric starting materials, or by separating isomers of compounds ofthe present invention.
- Isomers may include geometric isomers, for example cis isomers or trans isomers across a double bond. All such isomers are contemplated among the compounds ofthe present invention.
- the compounds ofthe present invention also include tautomers, salts, solvates and prodrugs of such compounds.
- the starting materials for use in the preparation ofthe compounds of the invention are commercially available or can be prepared by conventional methods known to a skilled person or in an analogous manner to processes described in the art.
- the compounds ofthe present invention can be prepared by the procedures described below.
- Scheme 1 illustrates the preparation of racemic benzothiazepines 9a and 9b.
- Reaction of benzenesulfonyl chloride 1 with aminoalcohol 2 in the presence of a base, such as triethylamine, in a solvent, such as tetrahydrofuran yields benzenesulfonamide 3 which can be converted to protected benzenesulfonamide 4.
- Protected benzenesulfonamide 4 optionally can be treated with an alkyl halide, such as methyl iodide, in the presence of a base such as sodium hydride, in a solvent, such as dimethylformamide, to yield N- substituted benzenesulfonamide 5.
- Protected benzenesulfonamide 4 or N- substituted benzenesulfonamide 5 is then successively reacted with (i) a strong base (such as n-butyllithium in hexanes) in a solvent (such as tetrahydrofuran), (ii) an electrophile (such as trimethyl borate), and (iii) a base (such as sodium carbonate), a benzyl halide (such as p-methoxybenzyl chloride), and a catalyst (such as tetrakis(triphenylphosphine)palladium(0)) to yield sulfonamide 6.
- a strong base such as n-butyllithium in hexanes
- a solvent such as tetrahydrofuran
- an electrophile such as trimethyl borate
- a base such as sodium carbonate
- a benzyl halide such as p-methoxybenzyl chloride
- sulfonamide 6 Treatment of sulfonamide 6 with a fluoride source, such as tetrabutylammonium fluoride, in a solvent, such as tetrahydrofuran, provides the deprotected sulfonamide alcohol 7.
- Sulfonamide alcohol 7 is successively oxidized using a method such as Swem Oxidation to yield sulfonamide aldehyde 8.
- aldehyde 8 is converted to racemic benzothiazepines 9a and 9b.
- R 1 , R 2 , R 5 , R N , R x and q are as previously defined above for compounds of Formula I.
- Scheme 2 illustrates an alternative synthetic scheme for the preparation of sulfonamide alcohol 3 used in Scheme 1.
- Substituent L of benzenesulfonyl chloride 10 is a suitable leaving group such as fluoro, chloro, bromo, nitro, tosyloxy or trifluoromethylsulfonyloxy.
- Scheme 3 illustrates the preparation of benzothiazepines having 4- position substituents other than hydroxy.
- benzothiazepine 9a or 9b is first oxidized to benzothiazepine-4-one 13.
- Conventional oxidizing agents such as PCC, or Swem conditions can be used.
- Benzothiazepine-4-one 13 is then reacted with Lawesson's Reagent to produce 4-thioxo-benzothiazepine 14.
- 4-Thioxo- benzothiazepine 14 can be reacted with a suitable reducing agent, such as lithium aluminum hydride, in a suitable solvent, such as tetrahydrofuran, to yield 4-mercapto-benzothiazepine 15.
- 4-Mercapto-benzothiazepine 15 can be reacted with a suitable alkylating agent, such as an alkyl halide, in the presence of a base, such as sodium hydride, in a suitable solvent, such as dimethylformamide, to yield 4-alkylthio-benzothiazepine 16.
- 4-Alkylthio- benzothiazepine 16 can be reacted with a suitable oxidizing agent, such as t- butyl hydroperoxide or m-chloroperbenzoic acid, to yield, successively, 4- alkylsulfinyl-benzothiepine 17 and 4-alkylsulfonyl-benzothiazepine 18.
- a suitable oxidizing agent such as t- butyl hydroperoxide or m-chloroperbenzoic acid
- 4-amino- or imino-benzothiazepines can be prepared by reacting benzothiazepine-4-one 13 with ammonia or a primary amine in a suitable solvent, such as tetrahydrofuran, to produce 4-imino-benzothiazepine 19.
- 4-Imino-benzothiazepine 19 can be reacted with a suitable reducing agent, such as lithium aluminum hydride, in a suitable solvent, such as tetrahydrofuran, to yield 4-amino-benzothiazepine 20.
- Benzothiazepine-4-one 13 also can undergo reductive alkylation by reaction with ammonia, a primary amine or a secondary amine in the presence of an reducing agent, such as sodium triacetoxyborohydride, in a suitable solvent, such as tetrahydrofuran, to produce 4-amino-benzothiazepine 21.
- an reducing agent such as sodium triacetoxyborohydride
- a suitable solvent such as tetrahydrofuran
- Scheme 3 also illustrates the preparation of 4-alkyl-benzothiazepine 23 and 4-alkoxycarbonyl-benzothiazepine 25.
- the 4-position hydroxy of benzothiazepine 9a or 9b is first converted to a suitable leaving group such as mesyloxy to form protected benzothiazepine 22.
- Protected benzothiazepine 22 is then reacted with a suitable nucleophile, such as butyl lithium, in a suitable solvent, such as tetrahydrofuran, to yield 4-alkyl-benzothiazepine 23.
- protected benzothiazepine 22 can be reacted with a suitable cyanidating agent, such as an potassium cyanide, in a suitable solvent, such as dimethylformarnide, to yield 4-cyano-benzothiazepine 24.
- a suitable cyanidating agent such as an potassium cyanide
- a suitable solvent such as dimethylformarnide
- 4-cyano-benzothiazepine 24 4-Cyano- benzothiazepine 24 is converted to 4-alkoxycarbonyl-benzothiazepine 25 by reaction with a suitable alcohol in the presence of a base, such as potassium hydroxide.
- Scheme 4 illustrates the preparation of benzothiazepine-4-ene 36 and benzothiazepine-4-one 33.
- Reaction of phenol 26 with a thiocarbamyl chloride, such as dimethylthiocarbamyl chloride, in a solvent, such as methanol-.tetrahydrofuran yields 0-thiocarbamate 27.
- Heating of O- thiocarbamate 27 in a solvent, such as tetradecane yields S-thiocarbamate 28.
- Hydrolysis of S-thiocarbamate 28 in the presence of a base, such as sodium hydroxide, in a solvent, such as methanohtetrahydrofuran yields thiophenol 29.
- Thiophenol 29 can be treated with a sulfonylating agent, such as sulfonyl chloride, in the presence of a oxidant such as potassium nitrate, in a solvent, such as tetrahydrofuran, to yield sulfonyl chloride 30.
- a sulfonylating agent such as sulfonyl chloride
- a solvent such as tetrahydrofuran
- Sulfonyl chloride 30 is then reacted with an aminoalcohol in a solvent, such as tetrahydrofuran, to yield benzenesulfonamide 31.
- Benzenesulfonamide 31 optionally can be hydroxyl protected with a silylating group agent, such as tert- butyldimethylsilyl chloride, in the presence of a base, such as imidazole, in a solvent, such as tetrahydrofuran, to yield protected benzenesulfonamide 32.
- a silylating group agent such as tert- butyldimethylsilyl chloride
- a base such as imidazole
- a solvent such as tetrahydrofuran
- N-substituted benzene sulfonamide 33 Deprotection ofthe protected N-substituted benzene sulfonamide 33 with a fluoride source, such as tetrabutylammonium fluoride, in a solvent, such as tetrahydrofuran, yields N-substitued benzenesulfonamide 34.
- Benzenesulfonamide 31 or N-substituted benzenesulfonamide 34 is then oxidized with a suitable oxidizing agent or under Swem conditions to form aldehyde 35.
- aldehyde 35 Upon treatment with zinc and titanium trichloride aldehyde 35 is converted to a mixture of benzothiazepine-4-ene 36 and benzothiazepine-4-one 37.
- Another class of compounds of specific interest comprises those compounds of Formula I wherein R 1 and R 2 are selected from among substituted and unsubstituted C,. 10 alkyl wherein substituted C,., 0 alkyl comprises one or more radicals independently selected from among, for example, alkylcarbonyl, alkoxy, hydroxy, and nitrogen-containing heterocyclyl joined to the C ]0 alkyl through an ether linkage.
- Ethyl, n-propyl, n-butyl, and isobutyl are prefened.
- substituents R 1 and R 2 are identical, for example n-butyl/n-butyl, so that the compound is achiral at the 3-position carbon. Eliminating optical isomerism at the 3-position carbon simplifies the selection, synthesis, separation, and quality control ofthe compound used as an ileal bile acid transport inhibitor.
- substituents R on the benzo ring can include, for example, hydrogen, aryl, alkyl, hydroxy, halo, alkoxy, alkylthio, alkylsulfinyl, alkylsulfonyl, haloalkyl, haloalkoxy, (N)- hydroxy-carbonylalkylamino, haloalkylthio, haloalkylsulfmyl, haloalkylsufonyl, amino, N-alkylamino, N,N-dialkylamino, (N)- alkoxycarbamoyl, (N)-aryloxycarbamoyl, (N)-aralkyloxycarbamoyl, trialkylammonium (especially with a halide counterion), (N)-amido, (N)- alkylamido
- the benzo ring can be mono-substituted at the 6, 7 or 8 position, or disubstituted at the 7- and -8 positions. Also included are the 6,7,8-trialkoxy compounds, for example the 6,7,8-trimethoxy compounds.
- substituents can be advantageously present on the 6, 7, 8, and/or 9- positions of the benzo ring including, for example, guanidinyl, cycloalkyl, carbohydrate residue (e.g., a 5 or 6 carbon monosaccharide residue), peptide residue, and quaternary ammonium salts linked to the ring via poly(oxyalkylene) linkages, e.g., -(OCH 2 CH 2 ) x -N+R 13 R 14 R 15 A ⁇ where x is 2 to 10.
- poly(oxyalkylene) linkages e.g., -(OCH 2 CH 2 ) x -N+R 13 R 14 R 15 A ⁇ where x is 2 to 10.
- R 5 and R 6 are independently selected from among hydrogen and ring-carbon substituted or unsubstituted aryl, thiopene, pyridine, pynole, thiazole, imidazole, pyrazole, pyrimidine, mo ⁇ holine, N-alkylpyridinium, N-alkylpiperazinium, N- alkylmo ⁇ holinium, or furan in which the substituent(s) are selected from among, for example, halo, hydroxyl, trihaloalkyl, alkoxy, amino, N- alkylamino, N,N-dialkylamino, quaternary ammonium salts, a to C alkylene bridge having a quaternary ammonium salt substituted thereon, alkoxycarbonyl, aryloxycarbonyl, alkylcarbonyloxy and arylcarbonyloxy, (O,O)-dioxyalkylene, -[O(CH 2 )
- the species that may constitute the substituents on the aryl ring of R 5 or R 6 are fluoro, chloro, bromo, methoxy, ethoxy, isopropoxy, trimethylammonium (preferably with an iodide or chloride counterion), methoxycarbonyl, ethoxycarbonyl, formyl, acetyl, propanoyl, (N)- hexyldimethylammonium, hexylenetrimethylammonium, tri(oxyethylene)iodide, and tetra(oxyethylene)trimethyl-ammonium iodide, each substituted at the p-position, the m-position, or both ofthe aryl ring.
- R 5 or R 6 is selected from phenyl, p-fluorophenyl, m- fluorophenyl, p-hydroxyphenyl, m-hydroxyphenyl, p-methoxyphenyl, m- methoxyphenyl, p-N,N-dimethylaminophenyl, m-N, N-dimethylaminophenyl, I- p-(CH 3 ) 3 -N + -phenyl, I " m-(CH 3 ) 3 -N + -phenyl, T m-(CH 3 ) 3 -N + -CH 2 CH 2 -
- Prefened compounds include 3-ethyl-3-butyl and 3-butyl-3-butyl compounds having each ofthe above prefened R 5 substituents in combination with the R x substituents shown in Tables 1 , 2 and 3 below. It is particularly prefened that one, but not both, of R 5 and R 6 is hydrogen.
- R 4 and R 6 be hydrogen, that R 3 and R 5 not be hydrogen, and that R 3 and R 5 be oriented in the same direction relative to the plane ofthe molecule, i.e., both in a- or both in ⁇ -configuration. It is further prefened that, where R 2 is butyl and R 1 is ethyl, then R 1 has the same orientation relative to the plane ofthe molecule as R 3 and R 5 .
- a class of compounds of particular interest comprises those 1,2- benzothiazepines wherein the R 1 , R 2 , R 3 , R 4 and R 5 radicals are as set forth in Table 1 below; the R 6 radical is hydrogen; the R N radical is selected from the group consisting of hydrogen, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n- hexyl and benzyl; and the R x radical or radicals are independently selected from the group of R radicals disclosed in Table 1 below.
- the first part of Table 1 identifies the R 1 , R 2 , R 3 , R 4 and R 5 radicals for each compound and the second part of Table 1 identifies the R x radical or radicals for those compounds.
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R N and R x radicals are selected from among the radicals disclosed in Table 2 below.
- R 6 is hydrogen and R 5 is other than hydrogen; and/or R 3 is hydroxy and R 4 is hydrogen; and/or R 1 and R 2 are alkyl. More preferably, R 1 and R 2 are the same.
- R 1 , R 2 , R 3 , R 4 and R 5 radicals are as set forth in Table 3 below;
- R 6 is hydrogen;
- the R N radical is selected from the group consisting of hydrogen, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl and benzyl;
- the R x radical or radicals are independently selected from the group of R x radicals disclosed in Table 2 above.
- q is 1 and R x is 7-dimethylamino. ⁇ / ⁇
- R 1 and R 2 are alkyl, preferably n-butyl; R 3 is hydroxy; R 4 and R 6 are hydrogen; R N is hydrogen; R x radicals are selected from the group consisting of amino, dimethylamino and methoxy; and R 5 is phenyl substituted at the para or meta position with one ofthe following groups:
- M is selected from the group consisting of Co 11 , Co 111 , Mn 11 , Mn 111 , Fe 11 , Fe 111 , Ni ⁇ , Ni 111 , Cr 111 , Cu ⁇ , Zn 11 , Cd 11 , Ga ⁇ , In 111 , V IV , Ru 11 , Pt IV , Rh m and Ir 1 ".
- the ileal bile acid transport inhibitor compounds ofthe present invention can be administered for the prophylaxis and/or treatment of hyperlipidemic diseases, conditions and/or disorders by any means, preferably oral, that contacts these compounds with their site of action in the body, for example in the ileum of a mammal such as a human.
- the compounds ofthe present invention can be used as the compound er se.
- Pharmaceutically acceptable salts are particularly suitable for medical applications because of their greater aqueous solubility relative to the parent compound.
- Such salts comprise a pharmaceutically acceptable anion or cation.
- Suitable pharmaceutically acceptable acid addition salts ofthe compounds ofthe present invention where appropriate include those salts derived from inorganic acids, such as hydrochloric, hydrobromic, phosphoric, metaphosphoric, nitric, sulfonic, and sulfuric acids, and organic acids such as acetic, benzenesulfonic, benzoic, citric, ethanesulfonic, fumaric, gluconic, glycolic, isothionic, lactic, lactobionic, maleic, malic, methanesulfonic, succinic, toluenesulfonic, tartaric, and trifluoroacetic acids.
- the chloride salt is particularly preferred for medical purposes.
- Suitable pharmaceutically acceptable base salts where appropriate include ammonium salts, alkali metal salts such as sodium and potassium salts, and alkaline earth salts such as magnesium and calcium salts.
- the anions ofthe definition of A " in the present invention are pharmaceutically acceptable anions such as those anions selected, for example, from the above list.
- the compounds ofthe present invention also can be administered in the form of a pharmaceutical composition
- a pharmaceutical composition comprising additional ingredients such as acceptable carriers, diluents, excipients, adjuvants and the like (collectively referred to herein as "carrier materials").
- Acceptable carrier materials are compatible with the other ingredients ofthe composition and are not deleterious to the recipient.
- a carrier material can be a solid or a liquid, or both, and is preferably formulated with the compound as a unit-dose composition, for example, a tablet or capsule, which can contain from 0.05% to 95% by weight ofthe active compound.
- Other pharmacologically active substances can also be present, including other compounds ofthe present invention.
- the pharmaceutical compositions ofthe invention can be prepared by any ofthe well known techniques of pharmacy, consisting essentially of admixing the components.
- These compounds can be administered by any conventional means available for use in conjunction with pharmaceuticals, either as an individual therapeutic compound in a monotherapeutic regimen or as a combination of therapeutic compounds in a combination therapy regimen.
- the amount of compound that is required to achieve the desired biological effect will depend on a number of factors such as the specific compound chosen, the use for which it is intended, the mode of administration, and the clinical condition ofthe recipient.
- a daily dose can be in the range of from about 0.3 to about 100 mg/kg bodyweight/day, preferably from about 1 mg to about 50 mg/kg bodyweight/day, and more preferably from about 3 to about 10 mg/kg bodyweight/day.
- This total daily dose can be administered to the patient in a single dose, or in proportionate multiple subdoses.
- Subdoses can be administered 2 to 6 times per day.
- Doses can be in sustained release form effective to obtain desired results.
- Orally administrable unit dose formulations such as tablets or capsules, can contain, for example, from about 0.1 to about 100 mg of benzothiazepine compound, preferably about 1 to about 75 mg of compound, more preferably from about 10 to about 50 mg of compound.
- the weights indicated above refer to the weight ofthe benzothiazepine ion derived from the salt.
- Oral delivery of an ileal bile acid transport inhibitor ofthe present invention can include formulations, as are well known in the art, to provide prolonged or sustained delivery ofthe drug to the gastrointestinal tract by any number of mechanisms. These include, but are not limited to, pH sensitive release from the dosage form based on the changing pH ofthe small intestine, slow erosion of a tablet or capsule, retention in the stomach based on the physical properties ofthe formulation, bioadhesion ofthe dosage form to the mucosal lining ofthe intestinal tract, or enzymatic release ofthe active drug from the dosage form. The intended effect is to extend the time period over which the active drug molecule is delivered to the site of action (the ileum) by manipulation ofthe dosage form.
- enteric-coated and enteric-coated controlled release formulations are within the scope ofthe present invention.
- Suitable enteric coatings include cellulose acetate phthalate, polyvinylacetate phthalate, hydroxypropylmethylcellulose phthalate and anionic polymers of methacrylic acid and methacrylic acid methyl ester.
- the dose can, for example, be in the range of from about 0.1 mg/kg body weight to about 1.0 mg/kg body weight, preferably from about 0.25 mg/kg body weight to about 0.75 mg/kg body weight, and more preferably from about 0.4 mg/kg body weight to about 0.6 mg/kg body weight.
- This dose can be conveniently administered as an infusion of from about 10 ng/kg body weight to about 100 ng/kg body weight per minute.
- Infusion fluids suitable for this purpose can contain, for example, from about 0.1 ng to about 10 mg, and preferably from about 1 ng to about 10 mg per milliliter.
- Unit doses can contain, for example, from about 1 mg to about 10 g ofthe compound ofthe present invention.
- ampoules for injection can contain, for example, from about 1 mg to about 100 mg.
- compositions according to the present invention include those suitable for oral, rectal, topical, buccal (e.g., sublingual), and parenteral (e.g., subcutaneous, intramuscular, intradermal, or intravenous) administration, although the most suitable route in any given case will depend on the nature and severity ofthe condition being treated and on the nature of the particular compound which is being used. In most cases, the preferred route of administration is oral.
- compositions suitable for oral administration can be presented in discrete units, such as capsules, cachets, lozenges, or tablets, each containing a predetermined amount of at least one compound ofthe present invention; as a powder or granules; as a solution or a suspension in an aqueous or non-aqueous liquid; or as an oil-in-water or water-in-oil emulsion.
- such compositions can be prepared by any suitable method of pharmacy which includes the step of bringing into association the active compound(s) and the carrier material (which can constitute one or more accessory ingredients).
- compositions are prepared by uniformly and intimately admixing the active compound with a liquid or finely divided solid carrier material, or both, and then, if necessary, shaping the product.
- a tablet can be prepared by compressing or molding a powder or granules ofthe compound, optionally with one or more assessory ingredients.
- Compressed tablets can be prepared by compressing, in a suitable machine, the compound in a free-flowing form, such as a powder or granules optionally mixed with a binder, lubricant, inert diluent and/or surface active/dispersing agent(s). Molded tablets can be made by molding, in a suitable machine, the powdered compound moistened with an inert liquid diluent.
- compositions suitable for buccal (sub-lingual) administration include lozenges comprising a compound ofthe present invention in a flavored base, usually sucrose, and acacia or tragacanth, and pastilles comprising the compound in an inert base such as gelatin and glycerin or sucrose and acacia.
- compositions suitable for parenteral administration conveniently comprise sterile aqueous preparations of a compound ofthe present invention. These preparations are preferably administered intravenously, although administration can also be effected by means of subcutaneous, intramuscular, or intradermal injection. Such preparations can conveniently be prepared by admixing the compound with water and rendering the resulting solution sterile and isotonic with the blood. Injectable compositions according to the invention will generally contain from 0.1 to 5% w/w of a compound disclosed herein.
- compositions suitable for rectal administration are preferably presented as unit-dose suppositories. These can be prepared by admixing a compound ofthe present invention with one or more conventional solid carrier materials, for example, cocoa butter, and then shaping the resulting mixture.
- compositions suitable for topical application to the skin preferably take the form of an ointment, cream, lotion, paste, gel, spray, aerosol, or oil.
- Carrier materials that can be used include vaseline, lanoline, polyethylene glycols, alcohols, and combinations of two or more thereof.
- the active compound is generally present at a concentration of from 0.1 to 15% w/w ofthe composition, for example, from 0.5 to 2%.
- compositions suitable for transdermal administration can be presented as discrete patches adapted to remain in intimate contact with the epidermis of the recipient for a prolonged period of time.
- patches suitably contain a compound ofthe present invention in an optionally buffered, aqueous solution, dissolved and/or dispersed in an adhesive, or dispersed in a polymer.
- a suitable concentration ofthe active compound is about 1% to 35%, preferably about 3% to 15%.
- the compound can be delivered from the patch by electrotransport or iontophoresis, for example, as described in Pharmaceutical Research. 3(6), 318 (1986).
- the amount of active ingredient that can be combined with the carrier materials to produce a single dosage form to be administered will vary depending upon the host treated and the particular mode of administration.
- the solid dosage forms for oral administration including capsules, tablets, pills, powders, and granules noted above comprise one or more compounds ofthe present invention admixed with at least one inert diluent such as sucrose, lactose, or starch.
- Such dosage forms may also comprise, as in normal practice, additional substances other than inert diluents, e.g., lubricating agents such as magnesium stearate.
- the dosage forms may also comprise buffering agents. Tablets and pills can additionally be prepared with enteric coatings.
- Liquid dosage forms for oral administration can include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs containing inert diluents commonly used in the art, such as water.
- Such compositions may also comprise adjuvants, such as wetting agents, emulsifying and suspending agents, and sweetening, flavoring, and perfuming agents.
- sterile injectable aqueous or oleaginous suspensions may be formulated according to the known art using suitable dispersing or setting agents and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a nontoxic parenterally acceptable diluent or solvent, for example, as a solution in 1,3-butanediol.
- acceptable vehicles and solvents that may be employed are water, Ringer's solution, and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- compositions encompass all the foregoing and the like.
- the dosage regimen to prevent, give relief from, or ameliorate a disease, condition and/or disorder relating to hyperlipemia, e.g., atherosclerosis, or to protect against or treat further high cholesterol plasma or blood levels with the compounds and/or compositions ofthe present invention is selected in accordance with a variety of factors. These include the type, age, weight, sex, diet, and medical condition ofthe patient, the severity ofthe disease, the route of administration, pharmacological considerations such as the activity, efficacy, pharmacokinetics and toxicology profiles ofthe particular compound employed, whether a drug delivery system is utilized, and whether the compound is administered as part of a drug combination. Thus, the dosage regimen actually employed may vary widely and therefore deviate from the preferred dosage regimen set forth above.
- Initial treatment of a patient suffering from a hyperlipidemic condition can begin with the dosages indicated above. Treatment should generally be continued as necessary over a period of several weeks to several months or years until the hyperlipidemic disease condition has been controlled or eliminated.
- Patients undergoing treatment with the compounds or compositions disclosed herein can be routinely monitored by, for example, measuring serum cholesterol levels by any ofthe methods well known in the art, to determine the effectiveness of therapy. Continuous analysis of such data permits modification ofthe treatment regimen during therapy so that optimal effective amounts of compounds ofthe present invention are administered at any point in time, and so that the duration of treatment can be determined as well.
- the treatment regimen/dosing schedule can be rationally modified over the course of therapy so that the lowest amount of ileal bile acid transport inhibitor ofthe present invention which exhibits satisfactory effectiveness is administered, and so that administration is continued only so long as is necessary to successfully treat the hyperlipidemic condition.
- the reaction mixture was stirred for one hour and warmed to room temperature.
- the resulting slurry was filtered and washed with 100 mL ethyl acetate.
- the ethyl acetate solution was washed with water (2x200 mL) and then brine (300 mL).
- the ethyl acetate layer was dried over magnesium sulfate and concentrated.
- the resulting yellow oil was dissolved in 300 mL of tetrahydrofuran and concentrated to give 20.61 g of 2-amino-2-butylhexanol as an oil.
- Step 4 N-[ 1 -Butyl- 1 -[[[(1 , 1 -dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]- 4-(dimethylamino)benzenesulfonamide
- Step 8 N-[l-Butyl-l-(hydroxymethyl) ⁇ entyl]-4-(dimethylamino)-2-[(3- nitrophenyl)methyl]-N-methylbenzenesulfonamide
- Step 9 N-[l -Butyl- l-formylpentyl]-4-(dimethylamino)-2-[(3-nitrophenyl) methyl]-N-methylbenzenesulfonamide.
- Step 10 (4R,5R)-3,3-dibutyl-7-(dimethylamino)-2,3,4,5-tetrahydro-2-methyl- 5-(3-nitrophenyl)- 1 ,2-benzothiazepin-4-ol 1 , 1 -dioxide
- the reaction mixture was stirred for 15 minutes, water was added, and then the mixture was concentrated to yield a residue.
- the residue was dissolved in 100 mL ethyl acetate.
- the semi-solid material was crystallized from ethyl acetate and hexane to give 0.51 g of (4R,5R)-5-(3- aminophenyl)-3,3-dibutyl-7-(dimethylamino)-2,3,4,5-tetrahydro-2-methyl-l,2- benzothiazepin-4-ol 1,1-dioxide as colorless crystals.
- Step 1 N-[ 1 -Butyl- 1 -[[[(1 -dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(4-methoxyphenyl)methyl]-N-methylbenzenesulfonamide
- Step 8 of Example 1 above was followed except that N-[l-butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(4-methoxyphenyl)methyl]-N-methylbenzenesulfonamide was used in place of N-[l-butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy] methyl]pentyl]-4-(dimethylamino)-2-[(3-nitrophenyl)methyl]-N- methylbenzenesulfonamide.
- Step 3 N-[ 1 -Butyl- 1 -formylpentyl]-4-(dimethylamino)-2-[(4-methoxyphenyl) methyl]-N-methylbenzenesulfonamide
- Step 9 of Example 1 above was followed except that N-[ 1 -butyl- 1 -(hydroxymethyl)pentyl]-4-(dimethylamino)-2-[(4- methoxyphenyl)methyl]-N-methylbenzenesulfonamide was used in place of N- [ 1 -butyl- 1 -(hydroxymethyl)pentyl]-4-(dimethylamino)-2-[(3- nitrophenyl)methyl]-N-methylbenzenesulfonamide.
- Step 10 of Example 1 The procedure of Step 10 of Example 1 above was followed except that N-[ 1 -butyl- 1 -formylpentyl]-4-(dimethylamino)-2-[(4- methoxyphenyl)methyl]-N-methylbenzenesulfonamide was used in place of N- [ 1 -butyl- 1 -formylpentyl]-4-(dimethylamino)-2-[(3-nitrophenyl)methyl]-N- methylbenzenesulfonamide.
- Example 10 The procedure of Example 10 was followed except that triethylamine was used in place of pyridine and heating was at 90 °C for 6 hours.
- 'H ⁇ MR is consistent with the desired product.
- Step 8 of Example 1 The procedure of Step 8 of Example 1 was followed except that N-[l-butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(3-nitrophenyl)methyl]benzenesulfonamide was used in place of N-[l-butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(3-nitrophenyl)methyl]-N-methylbenzenesulfonamide.
- Example 2 The procedure of Example 2 above was followed except that (4R,5R)- 3,3-dibutyl-7-(dimethylamino)-2,3,4,5-tetrahydro-5-(3-nitrophenyl)-l,2- benzothiazepin-4-ol 1,1-dioxide was used in place of (4R,5R)-3,3-dibutyl-7- (dimethylamino)-2,3,4,5-tetrahydro-5-(3-nitrophenyl)-2-methyl-l,2- benzothiazepin-4-ol 1,1-dioxide.
- ⁇ NMR was consistent with the product. MS (M + ) 460.
- Example 3 The procedure of Example 3 above was followed except that (4R,5R)- 5-(3-aminophenyl)-3,3-dibutyl-7-(dimethylamino)-2,3,4,5-tetrahydro-l,2- benzothiazepin-4-ol 1,1-dioxide was used in place of (4R,5R)-5-(3- aminophenyl)-3,3-dibutyl-7-(dimethylamino)-2,3,4,5-tetrahydro-2-methyl-l,2- benzothiazepin-4-ol 1,1-dioxide.
- ⁇ NMR was consistent with the product. MS (M+H + ) 623.
- Step 1 N-[ 1 -Butyl- 1 -(hydroxymethyl)pentyl]-4-(dimethylamino)-2- (phenylmethyl)benzenesulfonamide
- Steps 1-2 of Example 7 The procedure of Steps 1-2 of Example 7 was followed except that N- [ 1 -butyl- 1 -[[[( 1 , 1 dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)benzenesulfonamide and benzyl chloride were used in place of N-[ 1 -butyl- 1 -[[[( 1 , 1 dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-N-methylbenzenesulfonamide and /?-methoxybenzyl chloride.
- Step 3 of Example 13 The procedure of Step 3 of Example 13 was followed except that N-[l- butyl- 1 -(hydroxymethyl)pentyl] -4-(dimethylamino)-2-(phenylmethyl) benzenesulfonamide was used in place of N-[l -butyl- l-(hydroxymethyl) pentyl]-4-(dimethylamino)-2-[(3-nitrophenyl)methyl]benzenesulfonamide.
- Butyl- 1 -formylpentyl]-4-(dimethylamino)-2-(phenylmethyl) benzenesulfonamide was used in place of N-[l -butyl- l-formylpentyl]-4- (dimethylamino)-2-[(4-methoxyphenyl)methyl]-N-methylbenzene- sulfonamide.
- Step 1 of Example 7 The procedure of Step 1 of Example 7 was followed except that N-[l- butyl- 1 -[[[( 1 , 1 dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)benzenesulfonamide was used in place of N-[l-butyl-l- [[[(1 , 1 dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4-(dimethylamino)-N- methylb enzenesulfonamide.
- Step 2 N-[ 1 -Butyl- 1 -(hydroxymethyl)pentyl]-4-(dimethylamino)-2-[(4- methoxyphenyl)methyl]benzenesulfonamide
- Step 8 of Example 1 The procedure of Step 8 of Example 1 was followed except that N-[l- butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(4-methoxyphenyl)methyl]benzenesulfonamide was used in place of N-[l-butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl] pentyl]-4-(dimethylamino)-2-[(3-nitrophenyl)methyl]-N-methylbenzene- sulfonamide.
- Step 3 of Example 13 The procedure of Step 3 of Example 13 was followed except that N-[l- butyl-l-(hydroxymethyl)pentyl]-4-(dimethylamino)-2-[(4-methoxyphenyl) methyl]benzenesulfonamide was used in place of N-[l-butyl-l-[[[(l- dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4-(dimethylamino)-2-[(3- nitrophenyl)methyl]-N-methylbenzenesulfonamide.
- Step 10 of Example 1 The procedure of Step 10 of Example 1 was followed except that N-[l- butyl-l-formylpentyl]-4-(dimethylamino)-2-[(4-methoxyphenyl)methyl] benzenesulfonamide was used in place of N-[ 1 -butyl- 1 -formylpentyl]-4-
- Example 8 The procedure set forth in Example 8 above was followed except that (4R,5R)- 3,3-dibutyl-7-(dimethylamino)-2,3,4,5-tetrahydro-5-(4-methoxyphenyl)-l,2- benzothiazepin-4-ol 1,1-dioxide was used in place of (4R,5R)-3,3-dibutyl-7- (dimethylamino)-2,3,4,5-tetrahydro-5-(4-hydroxyphenyl)-2-methyl-l,2- benzothiazepin-4-ol 1,1-dioxide and a reaction temperature of 0 °C was employed.
- Step 8 of Example 1 The procedure of Step 8 of Example 1 was followed except that N-[l- butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(3-nitrophenyl)methyl]-N-(phenylmethyl) benzenesulfonamide was used in place of N-[l-butyl-l-[[[(l- dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4-(dimethylamino)-2-[(3- nitrophenyl)methyl]-N-methylbenzenesulfonamide.
- Step 3 N-[l -Butyl- l-formylpentyl]-4-(dimethylamino)-2-[(3- nitrophenyl)methyl]-N-(phenylmethyl)benzenesulfonamide
- Step l N-[l-Butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(4-methoxyphenyl)methyl]-N- (phenylmethyl)benzenesulfonamide
- Step 3 N-[l -Butyl- l-formylpentyl]-4-(dimethylamino)-2-[(3- nitrophenyl)methyl]-N-(phenylmethyl)benzenesulfonamide
- Step 3 of Example 13 The procedure of Step 3 of Example 13 was followed except that N-[l- butyl- 1 -(hydroxymethyl)pentyl]-4-(dimethylamino)-2-[(4- methoxyphenyl)methyl]-N-(phenylmethyl)benzenesulfonamide was used in place of N-[l-butyl-l-[[[(l-dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4- (dimethylamino)-2-[(3-nitrophenyl)methyl]-N-methylbenzenesulfonamide.
- Step 10 of Example 1 The procedure of Step 10 of Example 1 was followed except that N-[l- butyl-l-formylpentyl]-4-(dimethylamino)-2-[(3-nitrophenyl)methyl]-N- (phenylmethyl)benzenesulfonamide was used in place of N-[l -butyl- 1- formylpentyl]-4-(dimethylamino)-2-[(3-nitrophenyl)methyl]-N- methylbenzenesulfonamide.
- Step 2 of Example 1 The procedure of Step 2 of Example 1 was followed except that cycloleucinol was substituted for 2-amino-2-butylhexanol.
- Step 2-3 N-[ 1 -[[[(1 , 1 -dimethylethyl)dimethylsilyl]oxy]methyl]cyclopentyl]-4- (dimethylamino)benzenesulfonamide
- Steps 3 and 4 of Example 1 The procedure of Steps 3 and 4 of Example 1 was followed except that N-[l-(hydroxymethyl)cyclopentyl]-4-fluorobenzenesulfonamide was used in place of N-[ 1 -butyl- 1 -(hydroxymethyl)pentyl]-4-fluorobenzenesulfonamide.
- Step 4 N-[ 1 -[[[( 1 -dimethylethyl)dimethylsilyl]oxy]methyl]cyclopentyl]-4- (dimethylamino)-2-[(4-methoxyphenyl)methyl]-N-methylbenzenesulfonamide
- Step 1 of Example 7 The procedure of Step 1 of Example 7 was followed except that N-[l- [[[(1,1 -dimethylethyl)dimethylsilyl]oxy]methyl]cyclopentyl]-4- (dimethylamino)benzenesulfonamide was used in place of N- [1 -butyl- 1- [[[(1,1 dimethylethyl)dimethylsilyl]oxy]methyl]pentyl]-4-(dimethylamino)-N- methylbenzenesulfonamide.
- Step 5 N-[ 1 -(hydroxymethyl)cyclopentyl]-4-(dimethylamino)-2-[(4- methoxyphenyl)methyl]-N-ethylbenzenesulfonamide
- Step 6-8 (4R,5R)-7-dimethylamino)-2-ethyl-4,5-dihydro-5-(4- methoxyphenyl)-spiro[ 1 ,2-benzothiazepine-3 (2H), 1 ' -cyclopentan]-4-ol 1,1- dioxide
- Baby hamster kidney cells (BHK) transfected with the cD ⁇ A of human IB AT (HI 4 cells) are seeded at 60,000 cells/well in 96 well Top-Count tissue culture plates for assays run within 24 hours of seeding; 30,000 cells/well for assays run within 48 hours; and 10,000 cells/well for assays run within 72 hours. On the day of assay, the cell monolayer is gently washed once with
- assay buffer Dulbecco's Modified Eagle's medium with 4.5 g/L glucose + 0.2%o (w/v) fatty acid free bovine serum albumin ((FAF)BSA).
- assay buffer Dulbecco's Modified Eagle's medium with 4.5 g/L glucose + 0.2%o (w/v) fatty acid free bovine serum albumin ((FAF)BSA.
- FAF fatty acid free bovine serum albumin
- alanine uptake assay is performed in an identical fashion to the taurocholate assay, with the exception that labeled alanine is substituted for the labeled taurocholate.
- Bile ducts are cannulated with a 10 " length of PEI 0 tubing.
- the small intestine is exposed and laid out on a gauze pad.
- a canulae (1/8" luer lock, tapered female adapter) is inserted at 12 cm from the junction ofthe small intestine and the cecum.
- a slit is cut at 4 cm from this same junction (utilizing a 8 cm length of ileum).
- 20 mL of warm Dulbecco's phosphate buffered saline, pH 6.5 (PBS) is used to flush out the intestine segment.
- the distal opening is cannulated with a 20 cm length of silicone tubing (0.02" LD. x 0.037" O.D.).
- the proximal cannulae is hooked up to a peristaltic pump and the intestine is washed for 20 minutes with warm PBS at 0.25 mL/minute. Temperature of the gut segment is monitored continuously.
- 2.0 mL of control sample [ 1 C]-taurocholate @ 0.05 mi/mL with 5 mM cold taurocholate) is loaded into the gut segment with a 3 mL syringe and bile sample collection is begun. Control sample is infused at a rate of 0.25 mL/minute for 21 minutes.
- Bile samples fractions are collected every three minutes for the first 27 minutes ofthe procedure. After the 21 minutes of sample infusion, the ileal loop is washed out with 20 mL of warm PBS (using a 30 mL syringe), and then the loop is washed out for 21 minutes with warm PBS at 0.25 mL/minute. A second perfusion is initiated as described above but with the test compound being administered as well (21 minutes administration followed by 21 minutes of wash out) and bile sampled every three minutes for the first 27 minutes. If necessary, a third perfusion is performed as above that typically contains the control sample. Measurement Of Hepatic Cholesterol Concentration (HEPATIC CHOP Liver tissue is weighed and homogenized in chloroform:methanol (2:1).
- Hepatic HMG CoA-Reductase Activity HMG CO A Hepatic microsomes are prepared by homogenizing liver samples in a phosphate/sucrose buffer, followed by centrifugal separation. The final pelleted material is resuspended in buffer and an aliquot is assayed for HMG CoA reductase activity by incubating for 60 minutes at 37° C in the presence of 14 C-HMG-CoA (Dupont-NEN). The reaction is stopped by adding 6N HCl followed by centrifugation.
- Total serum cholesterol (SER.CHOL) is measured enzymatically using a commercial kit from Wako Fine Chemicals (Richmond, VA); Cholesterol Cl 1, Catalog No. 276-64909. HDL cholesterol (HDL-CHOL) is assayed using this same kit after precipitation of NLDL and LDL with Sigma Chemical Co. HDL Cholesterol reagent, Catalog No. 352-3 (dextran sulfate method). Total serum triglycerides (blanked) (TGI) are assayed enzymatically with Sigma Chemical Co. GPO-Trinder, Catalog No. 337-B. NLDL and LDL (VLDL + LDL) cholesterol concentrations are calculated as the difference between total and HDL cholesterol.
- Hepatic Cholesterol 7- Hvdroxylase Activity 7 -OHase '
- Hepatic microsomes are prepared by homogenizing liver samples in a phosphate/sucrose buffer, followed by centrifugal separation. The final pelleted material is resuspended in buffer and an aliquot is assayed for cholesterol 7-a-hydroxylase activity by incubating for 5 minutes at 37° C in the presence of NADPH. Following extraction into petroleum ether, the organic solvent is evaporated and the residue is dissolved in acetonitrile/ methanol.
- the enzymatic product is separated by injecting an aliquot ofthe extract onto a C ]8 reversed phase HPLC column and quantitating the eluted material using UN detection at 240nm. (Reference: Horton, J. D., et al. (1994) J. Clin. Invest. 93, 2084).
- Rat Gavage Assay Male Wister rats (275-300g) are administered IB AT inhibitors using an oral gavage procedure. Drug or vehicle (0.2% Tween 80 in water) is administered once a day (9:00-10:00 a.m.) for four days at varying dosages in a final volume of 2 mL per kilogram of body weight. Total fecal samples are collected during the final 48 hours ofthe treatment period and analyzed for bile acid content using an enzymatic assay as described below. Compound efficacy is determined by comparison ofthe increase in fecal bile acid (FBA) concentration in treated rats to the mean FBA concentration of rats in the vehicle group. Table 5 describes the results of this assay when the compound of Example 4 was tested. Table 5
- FBA fecal bile acid
- Rabbit Ileal brush border membranes are prepared from frozen ileal mucosa by the calcium precipitation method described by Malathi et al. (Reference: (1979) Biochimica Biophysica Acta, 554, 259).
- the method for measuring taurocholate is essentially as described by Kramer et al. (Reference: (1992) Biochimica Biophysica Acta, 1111, 93) except the assay volume is 200 ⁇ L instead of 100 ⁇ L.
- a 190 ⁇ L solution containing 2 ⁇ M [ 3 H]-taurocholate(0.75 ⁇ Ci), 20 mM tris, 100 mM NaCl, 100 mM mannitol pH 7.4 is incubated for 5 seconds with 10 ⁇ L of brush border membrane vesicles (60-120 ⁇ g protein).
- the incubation is initiated by the addition ofthe BBMN while vortexing and the reaction is stopped by the addition of 5 mL of ice cold buffer (20 mM Hepes-tris, 150 mM KC1) followed immediately by filtration through a nylon filter (0.2 ⁇ m pore) and an additional 5 mL wash with stop buffer.
- Acyl-CoA; cholesterol Acyl Transferase (ACAT) Hamster liver and rat intestinal microsomes are prepared from tissue as described previously (Reference: (1980) J. Biol. Chem. 255, 9098) and used as a source of ACAT enzyme.
- the assay consists of a 2.0 mL incubation containing 24 ⁇ M Oleoyl-CoA (0.05 ⁇ Ci) in a 50 mM sodium phosphate, 2 mM DTT ph 7.4 buffer containing 0.25 % BSA and 200 ⁇ g of microsomal protein. The assay is initiated by the addition of oleoyl-Co A.
- the reaction proceeds for five minutes at 37° C and is terminated by the addition of 8.0 mL of chloro form/methanol (2:1).
- 125 ⁇ g of cholesterol oleate in chloroform methanol to act as a carrier and the organic and aqueous phases ofthe extraction are separated by centrifugation after thorough vortexing.
- the chloroform phase is taken to dryness and then spotted on a silica gel 60 TLC plate and developed in hexane/ethyl ether (9:1).
- the amount of cholesterol ester formed is determined by measuring the amount of radioactivity incorporated into the cholesterol oleate spot on the TLC plate with a Packard instaimager.
- the examples herein can be repeated with similar success by substituting the generically or specifically described reactants and/or operating conditions of this invention for those used in the preceding examples.
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020017010201A KR20020000139A (en) | 1999-02-12 | 2000-02-10 | Novel 1,2-benzothiazepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake |
| IL14471600A IL144716A0 (en) | 1999-02-12 | 2000-02-10 | Novel 1,2-benzothiazepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake |
| CA002362147A CA2362147A1 (en) | 1999-02-12 | 2000-02-10 | 1,2-benzothiazepines for the treatment of hyperlipidemic diseases |
| AU36946/00A AU3694600A (en) | 1999-02-12 | 2000-02-10 | Novel 1,2-benzothiazepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake |
| EP00915720A EP1155007A2 (en) | 1999-02-12 | 2000-02-10 | 1,2-benzothiazepines for the treatment of hyperlipidemic diseases |
| JP2000598488A JP2002536440A (en) | 1999-02-12 | 2000-02-10 | Novel 1,2-benzothiazepines with activity as inhibitors of ileal bile acid transport and taurocholate uptake |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11993399P | 1999-02-12 | 1999-02-12 | |
| US60/119,933 | 1999-02-12 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2000047568A2 true WO2000047568A2 (en) | 2000-08-17 |
| WO2000047568A3 WO2000047568A3 (en) | 2000-12-14 |
Family
ID=22387268
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2000/002503 WO2000047568A2 (en) | 1999-02-12 | 2000-02-10 | 1,2-benzothiazepines for the treatment of hyperlipidemic diseases |
Country Status (10)
| Country | Link |
|---|---|
| EP (1) | EP1155007A2 (en) |
| JP (1) | JP2002536440A (en) |
| KR (1) | KR20020000139A (en) |
| CN (1) | CN1195748C (en) |
| AU (1) | AU3694600A (en) |
| CA (1) | CA2362147A1 (en) |
| HU (1) | HUP0105409A3 (en) |
| IL (1) | IL144716A0 (en) |
| WO (1) | WO2000047568A2 (en) |
| ZA (1) | ZA200106252B (en) |
Cited By (57)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003022286A1 (en) * | 2001-09-08 | 2003-03-20 | Astrazeneca Ab | Benzothiazepine and benzothiadiazepine derivatives with ileal bile acid transport (ibat) inhibitory activity for the treatment hyperlipidaemia |
| WO2003091232A2 (en) | 2002-04-25 | 2003-11-06 | Astrazeneca Ab | Benzothiadiazepine derivatives, processes for their preparation and pharmaceutical compositions containing them |
| WO2004020421A1 (en) * | 2002-08-28 | 2004-03-11 | Asahi Kasei Pharma Corporation | Novel quaternary ammonium compounds |
| WO2004076430A1 (en) * | 2003-02-25 | 2004-09-10 | Astrazeneca Ab | Benzothiazepine and benzothiepine derivatives |
| US6906058B2 (en) | 2000-03-08 | 2005-06-14 | Astrazeneca Ab | 1,5-Benzothiazepines and their use as hypolipidaemics |
| JP2005534652A (en) * | 2002-06-11 | 2005-11-17 | ワイス | Substituted phenylsulfonamide inhibitors of β-amyloid production |
| US7125864B2 (en) | 2001-09-07 | 2006-10-24 | Astrazeneca Ab | Benzothiazepine derivatives for the treatment of hyperlipidemia |
| US7192945B2 (en) | 2000-12-21 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7192946B2 (en) | 2001-09-04 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7192947B2 (en) | 2002-06-14 | 2007-03-20 | Astrazeneca Ab | Peptides derivatives comprising thiazepine group for the treatment of hyperlipidemic conditions |
| US7226943B2 (en) | 2001-09-07 | 2007-06-05 | Astrazeneca Ab | Benzothiepine ileal bile acid transport inhibitors |
| US7312208B2 (en) | 2002-08-28 | 2007-12-25 | Asahi Kasei Pharma Corporation | Quaternary ammonium compounds |
| EP1894564A2 (en) | 2003-04-05 | 2008-03-05 | AstraZeneca AB | Use of an ibat inhibitor for the treatment of prophylaxis of constipation |
| US7973030B2 (en) | 2004-02-27 | 2011-07-05 | Asahi Kasei Pharma Corporation | Benzothiazepine and benzothiepine compounds |
| WO2012064267A1 (en) | 2010-11-08 | 2012-05-18 | Albireo Ab | A pharmaceutical combination comprising an ibat inhibitor and a bile acid binder |
| WO2013063526A1 (en) | 2011-10-28 | 2013-05-02 | Lumena Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of hypercholemia and cholestatic liver disease |
| WO2014144650A2 (en) | 2013-03-15 | 2014-09-18 | Lumena Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of primary sclerosing cholangitis and inflammatory bowel disease |
| WO2014144485A1 (en) | 2013-03-15 | 2014-09-18 | Lumena Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of barrett's esophagus and gastroesophageal reflux disease |
| EP2995317A1 (en) | 2010-05-26 | 2016-03-16 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors and satiogens for treatment of diabetes, obesity, and inflammatory gastrointestinal conditions |
| US9409875B2 (en) | 2013-04-26 | 2016-08-09 | Elobix Ab | Crystal modifications of elobixibat |
| US9688720B2 (en) | 2010-11-08 | 2017-06-27 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| WO2017138877A1 (en) | 2016-02-09 | 2017-08-17 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2017138878A1 (en) | 2016-02-09 | 2017-08-17 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| EP3266457A1 (en) | 2011-10-28 | 2018-01-10 | Lumena Pharmaceuticals LLC | Bile acid recycling inhibitors for treatment of pediatric cholestatic liver diseases |
| US10183920B2 (en) | 2014-10-24 | 2019-01-22 | Elobix Ab | Crystal modifications of elobixibat |
| WO2019032026A1 (en) | 2017-08-09 | 2019-02-14 | Albireo Ab | Cholestyramine granules, oral cholestyramine formulations and use thereof |
| WO2019032027A1 (en) | 2017-08-09 | 2019-02-14 | Albireo Ab | Cholestyramine pellets, oral cholestyramine formulations and use thereof |
| US10441605B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10441604B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Cholestyramine pellets and methods for preparation thereof |
| WO2019234077A1 (en) | 2018-06-05 | 2019-12-12 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US10709755B2 (en) | 2014-06-25 | 2020-07-14 | Elobix Ab | Solid formulation and method for preventing or reducing coloration thereof |
| US10722457B2 (en) | 2018-08-09 | 2020-07-28 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2020161217A1 (en) | 2019-02-06 | 2020-08-13 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| WO2020161216A1 (en) | 2019-02-06 | 2020-08-13 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US10786529B2 (en) | 2016-02-09 | 2020-09-29 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10793534B2 (en) | 2018-06-05 | 2020-10-06 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US10941127B2 (en) | 2019-02-06 | 2021-03-09 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US10975046B2 (en) | 2018-06-20 | 2021-04-13 | Albireo Ab | Crystal modifications of odevixibat |
| US10975045B2 (en) | 2019-02-06 | 2021-04-13 | Aibireo AB | Benzothiazepine compounds and their use as bile acid modulators |
| US11007142B2 (en) | 2018-08-09 | 2021-05-18 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US11014898B1 (en) | 2020-12-04 | 2021-05-25 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| WO2021110887A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| WO2021110884A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2021110883A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2021110886A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2021110885A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11111224B2 (en) | 2019-12-04 | 2021-09-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11180465B2 (en) | 2019-12-04 | 2021-11-23 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11225466B2 (en) | 2019-12-04 | 2022-01-18 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11267794B2 (en) | 2019-12-04 | 2022-03-08 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11377429B2 (en) | 2020-08-03 | 2022-07-05 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11549878B2 (en) | 2018-08-09 | 2023-01-10 | Albireo Ab | In vitro method for determining the adsorbing capacity of an insoluble adsorbant |
| US11572350B1 (en) | 2020-12-04 | 2023-02-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11583539B2 (en) | 2020-11-12 | 2023-02-21 | Albireo Ab | Treating progressive familial intrahepatic cholestasis (PFIC) with IBAT inhibitors |
| EP4241840A2 (en) | 2019-02-12 | 2023-09-13 | Mirum Pharmaceuticals, Inc. | Methods for treating cholestasis |
| US11801226B2 (en) | 2018-06-20 | 2023-10-31 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| WO2023237728A1 (en) | 2022-06-09 | 2023-12-14 | Albireo Ab | Treating hepatitis |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996005188A1 (en) * | 1994-08-10 | 1996-02-22 | The Wellcome Foundation Limited | Hypolipidemic 1,4-benzothiazepine-1,1-dioxides |
| WO1996016051A1 (en) * | 1994-11-17 | 1996-05-30 | The Wellcome Foundation Limited | Hypolipidemic benzothiazepines |
| WO1998002432A1 (en) * | 1996-07-16 | 1998-01-22 | Takeda Chemical Industries, Ltd. | Bicyclic compounds for controlling micturition |
| WO1998038182A1 (en) * | 1997-02-28 | 1998-09-03 | Glaxo Group Limited | Hypolipidemic bicyclic derivatives |
-
2000
- 2000-02-10 IL IL14471600A patent/IL144716A0/en unknown
- 2000-02-10 EP EP00915720A patent/EP1155007A2/en not_active Ceased
- 2000-02-10 HU HU0105409A patent/HUP0105409A3/en unknown
- 2000-02-10 WO PCT/US2000/002503 patent/WO2000047568A2/en not_active Application Discontinuation
- 2000-02-10 KR KR1020017010201A patent/KR20020000139A/en not_active Application Discontinuation
- 2000-02-10 CA CA002362147A patent/CA2362147A1/en not_active Abandoned
- 2000-02-10 CN CNB008060282A patent/CN1195748C/en not_active Expired - Fee Related
- 2000-02-10 AU AU36946/00A patent/AU3694600A/en not_active Abandoned
- 2000-02-10 JP JP2000598488A patent/JP2002536440A/en active Pending
-
2001
- 2001-07-30 ZA ZA200106252A patent/ZA200106252B/en unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996005188A1 (en) * | 1994-08-10 | 1996-02-22 | The Wellcome Foundation Limited | Hypolipidemic 1,4-benzothiazepine-1,1-dioxides |
| WO1996016051A1 (en) * | 1994-11-17 | 1996-05-30 | The Wellcome Foundation Limited | Hypolipidemic benzothiazepines |
| WO1998002432A1 (en) * | 1996-07-16 | 1998-01-22 | Takeda Chemical Industries, Ltd. | Bicyclic compounds for controlling micturition |
| WO1998038182A1 (en) * | 1997-02-28 | 1998-09-03 | Glaxo Group Limited | Hypolipidemic bicyclic derivatives |
Cited By (97)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6906058B2 (en) | 2000-03-08 | 2005-06-14 | Astrazeneca Ab | 1,5-Benzothiazepines and their use as hypolipidaemics |
| US7192945B2 (en) | 2000-12-21 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7192946B2 (en) | 2001-09-04 | 2007-03-20 | Astrazeneca Ab | Benzothiazepine derivatives |
| US7226943B2 (en) | 2001-09-07 | 2007-06-05 | Astrazeneca Ab | Benzothiepine ileal bile acid transport inhibitors |
| US7125864B2 (en) | 2001-09-07 | 2006-10-24 | Astrazeneca Ab | Benzothiazepine derivatives for the treatment of hyperlipidemia |
| US7132416B2 (en) | 2001-09-08 | 2006-11-07 | Astrazeneca Ab | Benzothiazepine and benzothiazepine derivatives with ileal bile acid transport (IBAT) inhibotory activity for the treatment hyperlipidaemia |
| WO2003022286A1 (en) * | 2001-09-08 | 2003-03-20 | Astrazeneca Ab | Benzothiazepine and benzothiadiazepine derivatives with ileal bile acid transport (ibat) inhibitory activity for the treatment hyperlipidaemia |
| AU2002329387B2 (en) * | 2001-09-08 | 2007-06-07 | Albireo Ab | Benzothiazepine and benzothiadiazepine derivatives with ileal bile acid transport (IBAT) inhibitory activity for the treatment hyperlipidaemia |
| US7238684B2 (en) | 2002-04-25 | 2007-07-03 | Astrazeneca Ab | Benzothiadiazepine derivatives, processes for their preparation and pharmaceutical compositions containing them |
| WO2003091232A2 (en) | 2002-04-25 | 2003-11-06 | Astrazeneca Ab | Benzothiadiazepine derivatives, processes for their preparation and pharmaceutical compositions containing them |
| JP2005534652A (en) * | 2002-06-11 | 2005-11-17 | ワイス | Substituted phenylsulfonamide inhibitors of β-amyloid production |
| US7671075B2 (en) | 2002-06-11 | 2010-03-02 | Wyeth | Substituted phenylsulfonamide inhibitors of beta amyloid production |
| US7192947B2 (en) | 2002-06-14 | 2007-03-20 | Astrazeneca Ab | Peptides derivatives comprising thiazepine group for the treatment of hyperlipidemic conditions |
| CN100494185C (en) * | 2002-08-28 | 2009-06-03 | 旭化成制药株式会社 | Novel quaternary ammonium compounds |
| JPWO2004020421A1 (en) * | 2002-08-28 | 2005-12-15 | 旭化成ファーマ株式会社 | New quaternary ammonium compounds |
| US7312208B2 (en) | 2002-08-28 | 2007-12-25 | Asahi Kasei Pharma Corporation | Quaternary ammonium compounds |
| WO2004020421A1 (en) * | 2002-08-28 | 2004-03-11 | Asahi Kasei Pharma Corporation | Novel quaternary ammonium compounds |
| US7803792B2 (en) | 2002-08-28 | 2010-09-28 | Asahi Kasei Pharma Corporation | Quaternary ammonium compounds |
| WO2004076430A1 (en) * | 2003-02-25 | 2004-09-10 | Astrazeneca Ab | Benzothiazepine and benzothiepine derivatives |
| US8067584B2 (en) | 2003-02-25 | 2011-11-29 | Albireo Ab | Benzothiazepine derivatives |
| EP1894564A2 (en) | 2003-04-05 | 2008-03-05 | AstraZeneca AB | Use of an ibat inhibitor for the treatment of prophylaxis of constipation |
| US7514421B2 (en) | 2003-04-05 | 2009-04-07 | Albireo Ab | Use of an IBAT inhibitor for the treatment of constipation |
| US7973030B2 (en) | 2004-02-27 | 2011-07-05 | Asahi Kasei Pharma Corporation | Benzothiazepine and benzothiepine compounds |
| EP3593802A2 (en) | 2010-05-26 | 2020-01-15 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors and satiogens for treatment of diabetes, obesity, and inflammatory gastrointestinal conditions |
| EP2995317A1 (en) | 2010-05-26 | 2016-03-16 | Satiogen Pharmaceuticals, Inc. | Bile acid recycling inhibitors and satiogens for treatment of diabetes, obesity, and inflammatory gastrointestinal conditions |
| WO2012064267A1 (en) | 2010-11-08 | 2012-05-18 | Albireo Ab | A pharmaceutical combination comprising an ibat inhibitor and a bile acid binder |
| US10000528B2 (en) | 2010-11-08 | 2018-06-19 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10487111B2 (en) | 2010-11-08 | 2019-11-26 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10981952B2 (en) | 2010-11-08 | 2021-04-20 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US9688720B2 (en) | 2010-11-08 | 2017-06-27 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US9694018B1 (en) | 2010-11-08 | 2017-07-04 | Albireo Ab | IBAT inhibitors for the treatment of liver disease |
| US10221212B2 (en) | 2010-11-08 | 2019-03-05 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US11732006B2 (en) | 2010-11-08 | 2023-08-22 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US11261212B2 (en) | 2010-11-08 | 2022-03-01 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10093697B2 (en) | 2010-11-08 | 2018-10-09 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| US10011633B2 (en) | 2010-11-08 | 2018-07-03 | Albireo Ab | IBAT inhibitors for the treatment of liver diseases |
| WO2013063526A1 (en) | 2011-10-28 | 2013-05-02 | Lumena Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of hypercholemia and cholestatic liver disease |
| EP3266457A1 (en) | 2011-10-28 | 2018-01-10 | Lumena Pharmaceuticals LLC | Bile acid recycling inhibitors for treatment of pediatric cholestatic liver diseases |
| US10512657B2 (en) | 2011-10-28 | 2019-12-24 | Lumena Pharmaceutials Llc | Bile acid recycling inhibitors for treatment of pediatric cholestatic liver diseases |
| US11376251B2 (en) | 2011-10-28 | 2022-07-05 | Shire Human Genetic Therapies, Inc. | Bile acid recycling inhibitors for treatment of pediatric cholestatic liver diseases |
| US11229661B2 (en) | 2011-10-28 | 2022-01-25 | Shire Human Genetic Therapies, Inc. | Bile acid recycling inhibitors for treatment of pediatric cholestatic liver diseases |
| EP3278796A1 (en) | 2011-10-28 | 2018-02-07 | Lumena Pharmaceuticals LLC | Bile acid recycling inhibitors for treatment of hypercholemia and cholestatic liver disease |
| WO2014144485A1 (en) | 2013-03-15 | 2014-09-18 | Lumena Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of barrett's esophagus and gastroesophageal reflux disease |
| WO2014144650A2 (en) | 2013-03-15 | 2014-09-18 | Lumena Pharmaceuticals, Inc. | Bile acid recycling inhibitors for treatment of primary sclerosing cholangitis and inflammatory bowel disease |
| US9701649B2 (en) | 2013-04-26 | 2017-07-11 | Elobix Ab | Crystal modifications of elobixibat |
| US9409875B2 (en) | 2013-04-26 | 2016-08-09 | Elobix Ab | Crystal modifications of elobixibat |
| US9745276B2 (en) | 2013-04-26 | 2017-08-29 | Elobix Ab | Crystal modifications of elobixibat |
| US10709755B2 (en) | 2014-06-25 | 2020-07-14 | Elobix Ab | Solid formulation and method for preventing or reducing coloration thereof |
| US10183920B2 (en) | 2014-10-24 | 2019-01-22 | Elobix Ab | Crystal modifications of elobixibat |
| US10519120B2 (en) | 2014-10-24 | 2019-12-31 | Elobix Ab | Crystal modifications of elobixibat |
| US10493096B2 (en) | 2016-02-09 | 2019-12-03 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10610543B2 (en) | 2016-02-09 | 2020-04-07 | Albireo Ab | Cholestyramine pellets and methods for preparation thereof |
| US10441604B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Cholestyramine pellets and methods for preparation thereof |
| US10441605B2 (en) | 2016-02-09 | 2019-10-15 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2017138877A1 (en) | 2016-02-09 | 2017-08-17 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10758563B2 (en) | 2016-02-09 | 2020-09-01 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10786529B2 (en) | 2016-02-09 | 2020-09-29 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2017138878A1 (en) | 2016-02-09 | 2017-08-17 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10799527B2 (en) | 2016-02-09 | 2020-10-13 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10864228B2 (en) | 2016-02-09 | 2020-12-15 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2019032027A1 (en) | 2017-08-09 | 2019-02-14 | Albireo Ab | Cholestyramine pellets, oral cholestyramine formulations and use thereof |
| WO2019032026A1 (en) | 2017-08-09 | 2019-02-14 | Albireo Ab | Cholestyramine granules, oral cholestyramine formulations and use thereof |
| US10881685B2 (en) | 2017-08-09 | 2021-01-05 | Albireo Ab | Cholestyramine granules, oral cholestyramine formulations and use thereof |
| WO2019234077A1 (en) | 2018-06-05 | 2019-12-12 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US10793534B2 (en) | 2018-06-05 | 2020-10-06 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11306064B2 (en) | 2018-06-05 | 2022-04-19 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US10975046B2 (en) | 2018-06-20 | 2021-04-13 | Albireo Ab | Crystal modifications of odevixibat |
| US11801226B2 (en) | 2018-06-20 | 2023-10-31 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| US11365182B2 (en) | 2018-06-20 | 2022-06-21 | Albireo Ab | Crystal modifications of odevixibat |
| US11802115B2 (en) | 2018-06-20 | 2023-10-31 | Albireo Ab | Pharmaceutical formulation of odevixibat |
| US11549878B2 (en) | 2018-08-09 | 2023-01-10 | Albireo Ab | In vitro method for determining the adsorbing capacity of an insoluble adsorbant |
| US11007142B2 (en) | 2018-08-09 | 2021-05-18 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| US10722457B2 (en) | 2018-08-09 | 2020-07-28 | Albireo Ab | Oral cholestyramine formulation and use thereof |
| WO2020161217A1 (en) | 2019-02-06 | 2020-08-13 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11773071B2 (en) | 2019-02-06 | 2023-10-03 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| WO2020161216A1 (en) | 2019-02-06 | 2020-08-13 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US11603359B2 (en) | 2019-02-06 | 2023-03-14 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US10941127B2 (en) | 2019-02-06 | 2021-03-09 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US10975045B2 (en) | 2019-02-06 | 2021-04-13 | Aibireo AB | Benzothiazepine compounds and their use as bile acid modulators |
| EP4245367A2 (en) | 2019-02-12 | 2023-09-20 | Mirum Pharmaceuticals, Inc. | Methods for treating cholestasis |
| EP4241840A2 (en) | 2019-02-12 | 2023-09-13 | Mirum Pharmaceuticals, Inc. | Methods for treating cholestasis |
| WO2021110884A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11180465B2 (en) | 2019-12-04 | 2021-11-23 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11891368B2 (en) | 2019-12-04 | 2024-02-06 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11267794B2 (en) | 2019-12-04 | 2022-03-08 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2021110886A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2021110885A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| WO2021110887A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US11708340B2 (en) | 2019-12-04 | 2023-07-25 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2021110883A1 (en) | 2019-12-04 | 2021-06-10 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11225466B2 (en) | 2019-12-04 | 2022-01-18 | Albireo Ab | Benzothiadiazepine compounds and their use as bile acid modulators |
| US11111224B2 (en) | 2019-12-04 | 2021-09-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11377429B2 (en) | 2020-08-03 | 2022-07-05 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| US11583539B2 (en) | 2020-11-12 | 2023-02-21 | Albireo Ab | Treating progressive familial intrahepatic cholestasis (PFIC) with IBAT inhibitors |
| US11014898B1 (en) | 2020-12-04 | 2021-05-25 | Albireo Ab | Benzothiazepine compounds and their use as bile acid modulators |
| US11572350B1 (en) | 2020-12-04 | 2023-02-07 | Albireo Ab | Benzothia(di)azepine compounds and their use as bile acid modulators |
| WO2023237728A1 (en) | 2022-06-09 | 2023-12-14 | Albireo Ab | Treating hepatitis |
Also Published As
| Publication number | Publication date |
|---|---|
| HUP0105409A3 (en) | 2004-11-29 |
| CN1195748C (en) | 2005-04-06 |
| JP2002536440A (en) | 2002-10-29 |
| IL144716A0 (en) | 2002-06-30 |
| EP1155007A2 (en) | 2001-11-21 |
| CN1346351A (en) | 2002-04-24 |
| WO2000047568A3 (en) | 2000-12-14 |
| CA2362147A1 (en) | 2000-08-17 |
| AU3694600A (en) | 2000-08-29 |
| KR20020000139A (en) | 2002-01-04 |
| ZA200106252B (en) | 2002-07-30 |
| HUP0105409A2 (en) | 2002-05-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2000047568A2 (en) | 1,2-benzothiazepines for the treatment of hyperlipidemic diseases | |
| EP1091953B1 (en) | Novel benzothiepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake | |
| US20020183307A1 (en) | Novel 1,4-benzothiazephine and 1,5-benzothiazepine compounds as inhibitors of apical sodium co-dependent bile acid transport and taurocholate uptake | |
| AU730024B2 (en) | Combination therapy employing ileal bile acid transport inhibiting benzothiepines and HMG Co-A reductase inhibitors | |
| AU700557B2 (en) | Novel benzothiepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake | |
| US6387924B2 (en) | Benzothiepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake | |
| US20040077625A1 (en) | Novel 1,4-benzothiazepine and 1,5-benzothiazepine compounds as inhibitors of apical sodium codependent bile acid transport abd taurocholate uptake | |
| JP2001526627A (en) | New benzothiepines with activity as inhibitors of ileal bile acid transport and taurocholate absorption | |
| EP1448546A1 (en) | Novel mono- and di-fluorinated benzothiepine compounds as inhibitors of apical sodium co-dependent bile acid transport (asbt) and taurocholate uptake | |
| EP1465885A2 (en) | Novel alkyl/aryl hydroxy or keto thiepine compounds as inhibitors of apical sodium co-dependent bile acid transport (asbt) and taurocholate uptake | |
| CA2506703A1 (en) | Novel benzothiepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake | |
| CZ20004937A3 (en) | Novel benzothiepines having activity as inhibitors of ileal bile acid transport and taurocholate uptake | |
| CZ322899A3 (en) | Combined therapy employing ileal transport of bile acids inhibited by benzothiepine HMG Co-A reductase inhibitors | |
| EP1440972A1 (en) | Novel benzothiepines having pharmaceutical activity. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 00806028.2 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ DE DK DM EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CR CU CZ DE DK DM EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000915720 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 36946/00 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2001/06252 Country of ref document: ZA Ref document number: 200106252 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 144716 Country of ref document: IL |
|
| ENP | Entry into the national phase |
Ref document number: 2362147 Country of ref document: CA Ref document number: 2362147 Country of ref document: CA Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020017010201 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: 2000 598488 Country of ref document: JP Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000915720 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020017010201 Country of ref document: KR |
|
| WWR | Wipo information: refused in national office |
Ref document number: 2000915720 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2000915720 Country of ref document: EP |
|
| WWR | Wipo information: refused in national office |
Ref document number: 1020017010201 Country of ref document: KR |