WO1993020057A1 - Novel derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols - Google Patents
Novel derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols Download PDFInfo
- Publication number
- WO1993020057A1 WO1993020057A1 PCT/US1993/002107 US9302107W WO9320057A1 WO 1993020057 A1 WO1993020057 A1 WO 1993020057A1 US 9302107 W US9302107 W US 9302107W WO 9320057 A1 WO9320057 A1 WO 9320057A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- dihydro
- alkyl
- benzofuran
- pentamethyl
- compound
- Prior art date
Links
- 0 *C1c(c(*)c(*)c(*)c2*)c2OC1(*)* Chemical compound *C1c(c(*)c(*)c(*)c2*)c2OC1(*)* 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/79—Benzo [b] furans; Hydrogenated benzo [b] furans with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/34—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having five-membered rings with one oxygen as the only ring hetero atom, e.g. isosorbide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/79—Benzo [b] furans; Hydrogenated benzo [b] furans with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
- C07D307/80—Radicals substituted by oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/79—Benzo [b] furans; Hydrogenated benzo [b] furans with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
- C07D307/81—Radicals substituted by nitrogen atoms not forming part of a nitro radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/82—Benzo [b] furans; Hydrogenated benzo [b] furans with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to carbon atoms of the hetero ring
- C07D307/84—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- This invention relates to certain derivatives of 2,3-dihydro-benzofuranol; to the intermediates and processes useful for their preparation, to their ability to manifest the property of being free necessarilyl scavengers, and to their end-use application in the treatment of disease conditions capable of being ameliorated by free radical scavengers such as, for example, stroke, nervous system trauma or reperfusion damage.
- R 2 is C 1-4 alkyl or both R 2 moieties, when taken together with the carbon atom to which they are attached, form a C 5-6 cyclic hydrocarbyl moiety;
- R 4 is C 1-6 alkyl
- R 5 is H or -C(O)R with R being H, or C 1-g alkyl;
- R 6 is C 1-6 alkyl
- R 7 is H or C 1-6 alkyl
- X is COOR 8 , CH 2 OH, halomethyl, C(O)A or -CH 2 A;
- A is -NR 7 R 9 , -N ⁇ R 6 R 6 R 6 -Q ⁇ , pyrrolidino, piperidino
- R 8 is H, C 1-6 alkyl, or -(CH 2 ) m -A, with m being 2,3 or 4;
- R 9 is H, C 1-4 alkyl, n is
- R 10 is H, C 1-8 alkyl, C 1-6 alkenyl, C 4-6 cycloalkyl,
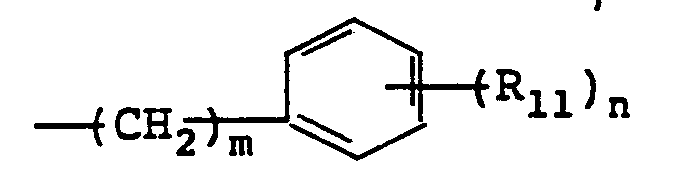
- R 11 is H, C 1-4 alkoxy, C 1-4 alkyl or halogeno
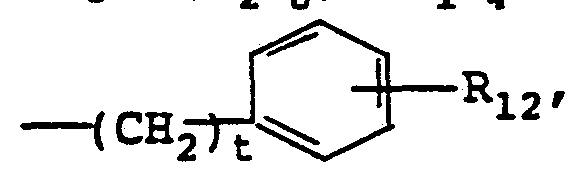
- R 12 is ortho C 1-4 alkoxy, ortho C 1-4 alkyl or p-halo
- Q is a halogen ⁇ or - ⁇ SO 3 R 1 with R 1 being H, C 1-6 alkyl, aryl or aralkyl.
- alkyl includes the straight and branched chain saturated aliphatic hydrocarbyl moieties having the indicated number of carbon atoms, preferably methyl or ethyl, but including others such as propyl, isopropyl, n-butyl and the like.
- -C(O)R includes moieties wherein R is H or a C 1-9 alkyl moiety, embracing, for example, formyl, methylcarbonyl, ethylcarbonyl, propylcarbonyl and the like.
- the -NR 7 R 9 moieties include the amino and mono and di-substituted amines with R 7 and R 9 being as defined.
- R 11 selected from the group consisting of C 1-4 alkoxy (preferably methoxy) C 1-4 alkyl (preferably methyl) or halogen (preferably chloro but including bromo and iodo).
- mono and di-hydroxy substituted alkyl moieties are those moieties wherein the alkyl moiety can bear one or two OH groups (other than two hydroxy groups on one carbon atom), preferably moieties bearing a hydroxy group on a terminal carbon atom.
- C 2-9 acyloxy alkylene (C 2-6 ) are those compounds wherein the acyloxy
- the -C 2-6 alkylene-O- (CH 2 ) 2-4 OH moieties have respectively a divalent 1-6 carbon atom moiety attached to an oxgen (O).
- O oxgen
- Piperidino is illustrated by morpholino by
- both R 2 alkyl moieties be the same (e.g. 2,2-dimethyl or 2,2-diethyl) but both need not be the same for any one compound.
- a represents a tertiary amine both of R 7 and R 9 are preferably the same, preferably both being methyl or ethyl, and when R 9 is other than H or alkyl, benzyl is preferred.
- Q ⁇ represents a quaternary ammonium moiety wherein Q includes all halides with chloro and bromo being preferred, and aryl includes phenyl or its alkylated derivatives with toluene as the preferred species and aralkyl includes benzyl or phenethyl and their alkylated derivatives.
- the pharmaceutically acceptable salts include those acid addition salts derived by reaction with such acids as hydrochloric, hydrobromic, sulfuric, nitric or phosphoric acids and such organic carboxylic acids as acetic,
- glycolic glycolic
- maleic tartaric
- citric tartaric
- salicylic 2-acetyloxybenzoic acids
- organic sulfonic acids such as methanesulfonic 4-toluenesulfonic as naphthalensulfonic acids.
- methanesulfonic 4-toluenesulfonic as naphthalensulfonic acids.
- other acids well known to the pharmaceutical art may also be utilized.
- stereoisomer is a general term for all isomers of individual molecules that differ only in the orientation of their atoms in space. It includes mirror image isomers (enantiomers), geometric (cis/trans) isomers, and isomers of compounds with more than one chiral center that are not mirror images of one another (diastereoisomers). It is recognized that certain stereoisomeric forms of the present invention may have properties which are superior to other stereoisomeric forms or mixtures thereof. These properties may be a better therapeutic response, better bioavailability, lower
- the compounds of formula I may be prepared by standard chemical processes and techniques analogously known in the art with the overall processes depicted in the following Reaction Schemes A, B and C.
- step (a) C 6 H 5 CH 3 , reflux 1 hour; step (b) AlCl 3 , 145°, 1.5 hour; step (C) 1N NaOH, 50% MeOH, reflux 1 hour; step (d) 4-O 2 N-C 6 H 4 COCl, Pyridine, room temperature.
- step (e) thallium(III) (NO 3 ) 3 ⁇ 3H 2 O, (CH 3 O) 3 CH, MeOH, room temperature, 4 days; step (f) tetrahydrofuran, 2N NaOH, reflux 1 hour; step (g) 1 N NaOH 50% MeOH, reflux 24 hours.
- R,R 2 ,R 4 ,R 6 ,R 7 and A are as previously defined.
- the acids (7) can readily be reduced to the alcohols (8).
- Borane-dimethylsulfide complex is the preferred reagent for this reaction using tetrahydrofuran as solvent but other hydride reagents, such as lithium aluminum hydride can be used as well as other non-reactive solvents. Due to the steric hindrance which the 2,2-dialkyl
- substitution imparts, unusually long reaction times, preferably 16 to 48 hours, and elevated temperatures
- Conversion of the bromide to the iodide can be accomplished by refluxing it in acetonitrile with one equivalent of sodium iodide. Conversion of the bromides or iodides (9) to amine-substituted products (10) can be accomplished by procedures well known to the art, but again employing extended reaction time and elevated reaction temperatures to overcome steric hindrance. Thus, using dimethylformamide at 60-80°C or acetonitrile at reflux temperature the reaction of bromide (9) with an amine such as 1-methyl-piperazine for 2 to 5 days will lead to acceptable yields of products (10). Protection of the phenolic hydroxy group by acylation to (11) is necessary in some instances, or desired in others, to obtain acylated products (12). The latter can also be obtained by acylation of (10) and the resulting products (11) may then be converted to compounds (12).
- the acid (7) after protection of the phenolic hydroxy group to (13), can be converted to acid chlorides (14) using triphosgene (bistrichloromethyl carbonate) or diphosgene (trichloromethyl chloroformate) in the presence of triethylamine in an inert solvent, such as dichloromethane.
- the compounds may occur as either the R- or the S-enantiomers, or mixtures thereof.
- the preparation of the individual enantiomeric form may be effected by resolving the acids of Formula (13) by standard and conventional means such as, for example, via the use of diastereomeric salts with optically active amines, or alternatively, by resolving the alcohols (8) as esters with optically active acids, e.g. L-2,4-MeClC 6 H 3 CHMeCOOH (Me representing methyl).
- the ester is pulverized and mixed with 61.74 g (10% excess) of anhydrous aluminium chloride by means of a mechanical stirrer and heated to 135-145°C for 1.5 hours. The resulting melt is allowed to cool, dissolved in 200 ml of
- dichloromethane and 200 ml of 2N hydrochloric acid is added dropwise.
- the dichloromethane phase is separated, washed with sodium bicarbonate and a sodium chloride solution and dried over sodium sulfate. Filtration and evaporation of solvent gives an oil that is treated with 300 ml of
- the p-nitrobenzoyl ester of this material (see Step A) is prepared by portionwise addition of 38.98 g of p-nitrobenzoyl chloride to an ice-cooled solution of 46.86 g of 3,4-dihydro-6-hydroxy-2,2,5,7,8-pentamethyl-2H-1-benzopyran-4-one in 250 ml of pyridine and stirring at room temperature overnight. Water is added and the solid is collected and washed with water and a little methanol.
- Methyl-2,3-Dihydro-5-hydroxy-2,2,4,6,7-pentamethyl-1-benzofuran-3-carboxylate Treatment of a boiling solution of 20.67 g (0.05 mol) of this material (see Step B) in 200 ml of tetrahydrofuran with 50 ml of 2N sodium hydroxide for 1 hour is followed by evaporation of solvent (20 minutes), addition of water, and extraction with dichloromethane (three times). The extract is washed with water and a sodium chloride solution, dried over sodium sulfate, filtered and evaporated.
- Example 4 and 3.3 g (10% excess) of methyl 4-methylbenzenesulfonate in 60 ml of acetonitrile is refluxed for 18 hours. The solvent is evaporated and the residue is slurried in ethyl acetate. The resulting semi-solid is recrystallized twice from acetonitrile to give 3.7 g of the title compound, m.p. 244-245°C. Elemental analysis, IR, UV, and 1 H-NMR spectra confirm the structure.
- acetonitrile is stirred at reflux temperature for 5 hours. After cooling, the mixture is filtered to remove sodium bromide that precipitates (0.81 g). To the filtrate is added 1.05 g of 1-methylpiperazine and the mixture is refluxed for 3 days. Sodium bicarbonate (1 equivalent) is added, the solvent is evaporated and the residue is taken up in ethyl acetate. The basic product is separated by washing with 2N hydrochloric acid and water, the washes are basified by addition of sodium bicarbonate, and the product is reextracted into ethyl acetate. After drying over anhydrous sodium sulfate, filtration and evaporation, 1.90 g of an oil is obtained.
- Example 3 1.83 g (5% excess) of 1-[2-(2-hydroxyethoxy)-ethyl]piperazine, 1.50 g (1 equivalent) of sodium iodide and 0.84 g (1 equivalent) of sodium bicarbonate in 40 ml of acetonitrile is refluxed for 3 days.
- the solvent is
- a solution of 2.61 g (0.011 mol) of 1-(2-pyrimidyl)-piperazine dihydrochloride in 20 ml of water is made alkaline with a saturated potassium carbonate solution and the free base is extracted with toluene.
- the extract is evaporated and to the residue is added 2.99 g (0.01 mol) of the compound described in Example 3, 1.50 g (0.01 mol) of sodium iodide, 0.84 g (0.01 mol) of sodium bicarbonate and 50 ml of acetonitrile.
- the mixture is stirred at reflux temperature for 3 days.
- the solvent is evaporated and the residue is taken up in ethyl acetate and washed with water.
- the basic product is separated by washing with 2N hydrochloric acid and water, the washes are made basic by addition of sodium bicarbonate and reextracted with ethyl acetate.
- the extract is dried over sodium sulfate, filtered and evaporated. To the residue is added a solution of
- acetonitrile is refluxed overnight with stirring.
- the precipitated sodium bromide is removed by filtration and 1.95 g of N-methylbromoveratrylamine and 0.84 g of sodium bicarbonate is added to the filtrate.
- the resulting mixture is stirred at reflux temperature for 5 days.
- the solvent is evaporated and the residue is taken up in ethyl acetate and washed with water.
- the basic product is separated by washing with 2N hydrochloric acid and water, the washes are basified by addition of sodium bicarbonate and the product is reextracted into ethyl acetate. After drying over anhydrous sodium sulfate, filtration and evaporation, an oil is obtained, which is then dissolved in isopropanol and is added to a solution of 1.16 g of maleic acid in
- the aqueous phase is made alkaline by addition of sodium bicarbonate and is extracted twice with ethyl acetate. The extract is dried over anhydrous sodium
- Example 3 and allowed to react with 1-methylpiperazine as described in Example 8.
- Example 17 racemate and 4.77 g (0.047 M) of triethylamine in 100 ml of dichloromethane.
- the escaping gas is trapped over potassium hydroxide.
- the mixture is stirred at room temperature overnight and at reflux temperature for 1 hour.
- the residue obtained after evaporation of solvent at 30°C is suspended in toluene and the resulting precipitate of triethylamine hydrochloride is removed by filtration.
- the filtrate is evaporated and the residue is crystallized from hexane to give 10.93 g (75%) of the title compound, m.p. 171-173.5°C.
- Elemental analysis, UV and 1 H-NMR spectra confirm the structure.
- the extract is dried over anhydrous sodium sulfate, filtered and evaporated to give 5.20 g of an oil.
- the compounds of the present invention can be tested for efficacy in stroke and nervous system trauma by the method described in J. Neurosurgery 62:882-887 (1985). Male CD-1 mice weighing 18-22 g are subjected to head injury produced by a 50 g weight dropped 32 cm.
- the neurological status is evaluated one hour later.
- Treatment of the mice with the compounds of interest can be pre-trauma or post-trauma. Improvement of neurological status of the treated mice shows efficacy.
- the compounds of the present invention can be tested according to the method described in Stroke 20: 1037-1043 (1989). Male
- Sprague-Dawley rats weighing 250-300 g are subjected to occlusion of middle cerebral artery followed by
- the compounds of this invention are free radical scavengers. Free radical reactions have been implicated in the pathology of more than 50 human diseases. Radicals and other reactive oxygen species are formed constantly in the human body both by deliberate synthesis (e.g. by activated phagocytes) and by chemical side-reactions. They are
- Oxidative stress occurring when antioxidant defences are inadequate, can damage lipids, proteins, carbohydrates and DNA. A few clinical conditions are caused by oxidative stress, but more often the stress results from the disease and can make a significant contribution to the disease pathology. For a more detailed review see B.
- M. Miyamoto et al. ( J. Pharmacol. Exp. Ther., 1989, 250, 1132) report that neurotoxicity due to excessive glutamic acid release is similarly reduced by anti-oxidants. They suggest the use of agents that inhibit brain lipid peroxidation for treatment of neurodegenerative diseases such as Huntington's and Alzheimer's disease in which excessive glutamic acid release has been observed.
- M.R. Hori et al. ( Chem. Pharm. Bull. 1991, 39, 367) report on anti-amnesic activity of brain lipid peroxidation
- ischemia/reperfusion reduces these effects in heart, lung, kidney, pancreas, brain and other tissues.
- the compounds of the present invention are also useful in treating the process of inflammation which is known to involve the release of superoxide radicals from phagocytic cells which cause some of the symptoms of rheumatoid
- ulcerative colitis ulcerative colitis. Inhalation injury of the lungs is typically caused by heat and chemical irritation, and chemical injury is the leading lethal cause of smoke
- pulmonary edema This process is accompanied by increased lipid peroxidation in lung tissue.
- An inhibitor of lipid peroxidation was shown to reduce these symptoms in animals subjected to hot sawdust smoke by Z. Min et al., (J. Med. Cell. PLA, 1990, 5, 176-180). They suggest the use of anti-oxidants in treatment of smoke inhalation-lung injury, adult respiratory distress syndrome and emphysema.
- Reactive oxygen species also play a role in the
- the compounds may also be useful in the treatment of cancers, and degenerative diseases related to aging, stroke, and head trauma, since oxygen-derived free radicals have been identified among causative factors.
- oxygen-derived free radicals have been identified among causative factors.
- Antioxidants have also been shown to be useful in the treatment of cataracts. Free Rad. Biol. Med. , 12:251-261 (1992).
- In vitro and in vivo activity for the compounds of this invention may be determined by the use of standard assays which demonstrate the free radical scavenging property, affinity for cardiac tissue and cardioprotective
- Exemplary of the assay useful for determining the free-radical scavenging property of the compounds of this invention is by the in vitro inhibition of lipid peroxidation in rat brain homogenates.
- the free radical scavenging properties of the compounds may readily be evaluatedin an assay wherein superoxide radicals are generated by 4 mU of xanthine oxidase in the presence of 0.1 mM xanthine and detected by reduction of 40 ⁇ M nitro blue tetrazolium (NBT) to the diformazan dye in a spectrophotometric assay as described by C. Beauchamp and I. Fridovick, (Analyt. Biochem. 1971, 44, 276-287). 30 U of superoxide dismutase inhibited this reduction by 90% which is due to superoxide radicals. In the presence of a
- test compound there is a competition for the superoxide radical and thus a reduction in the color formation of NBT demonstrates the superoxide radical scavenging property of the test compound.
- Inhibiting the process of lipid peroxidation may be assayed using tissue homogenates for measuring the
- lipid peroxidation in mice may be utilized to demonstrate the ability of the compounds to penetrate and act as free radical scavengers in these tissues.
- This assay involves pretreatment of male CDl mice by subcutaneous administration of the test compound. One hour later the tissues are excised, homogenized 1+9 (w/v) in 20 mM
- TBA-adduct is extracted into 2 times 1 ml of n-butanol.
- the fluorescence is measured at an excitation wavelength of 515 nm and an emission wavelength of 553 nm against a standard prepared from malondialdehyde dimethylacetal.
- Stimulated human leukocytes release radicals and other oxygen metabolites, which, during inflammation, act as microbicidal agents. At the same time, they release
- proteolytic enzymes such as elastase, which are also microbicidal but potentially threaten the connective tissue of the host.
- An endogenous ⁇ 1 -proteinase inhibitor ( ⁇ 1 Pi) normally protects the host tissue from proteolytic
- ⁇ 1 Pi is however, inactivated by the leukocyte-derived oxidants.
- Antagonism of the of ⁇ 1 Pi is an indication of the disclosed radical scavengers.
- the concentration needed to protect 50% of the elastase inhibitory capacity of ⁇ 1 Pi (PC 50 ) depends on the amount of stimulated
- Oxidized ⁇ 1 Pi has also been isolated from rheumatoid
- viscosity of synovial fluid is triggered by superoxyl radicals released from human leukocytes in vitro (see R.A.
- nonsteroidal anti-inflammatory drugs were shown to inhibit the release of superoxyl radicals from leukocytes (see H. Strom and I. Ahnfelt-Ronne, Agents and Actions, 1989, 26, 235-237 and M. Roch-Arveiller, V.
- scavengers useful in the prevention and treatment of such disease states related to neurotoxicity due to excessive glutamic acid release, to Huntington's disease, Alzheimer's disease and other cognitive dysfunctions, (e.g. memory, learning and attention deficits), amnesia, and Parkinson's disease, as well as the treatment and prevention of tissue damage in heart, lung, kidney, pancreas and brain tissues induced by ischemia/reperfusion, and to allay acute blood loss due to haemorrhagic shock.
- the compounds of the present invention are of
- stroke means cerebrovascular disease which includes cerebral insufficiency due to transient
- nervous system trauma means injury to the head or spine.
- injury can occur from skull or spine penetration or from rapid brain acceleration or deceleration which injures tissue at the point of impact, at its opposite pole or within the frontal or temporal lobes.
- Injury may consist of nerve tissue, blood vessels and/or meninges damage resulting in neural disruption, ischemia and/or edema.
- reperfusion damage means the damage that occurs in any blood-deprived tissue, anywhere in the body, upon reintroduction of the blood supply. For example, reperfusion of an ischemic area of the myocardium or the cerebrum.
- the compounds of this invention can be utilized both prophylactically and therapeutically.
- the amount of active ingredient for therapeutic administration can vary over a wide range and is dependent upon such factors as the species of patient to be treated, its age, health, sex, weight, nature and the severity of the condition being treated.
- patient refers to a warm-blooded animal such as, for example, rats, mice, dogs, cats, guinea pigs, primates and humans.
- a therapeutically effective amount of the active ingredient to be administered will range from about 0.1 mg/kg to 30 mg/kg of body weight per day.
- a pharmaceutically acceptable carrier which is any substance which aids in the administration of the compound without substantially affecting its
- the compounds are administered
- the compounds of this invention also can be orally administered, preferably using more active ingredient per day than when parenterally administered, preferably taking divided doses 3 to 4 times per day. Preferably, enteral administration in post "crisis” situations, particularly after release from hospitalized conditions.
- the compounds can be used in standard dosage unit forms such as tablets, capsules, dragees, lozenges, elixirs, emulsions,
- Tablets and capsules containing from 100 to 400 mg of active ingredient are preferred modes of enteral
- the active ingredient is generally blended with conventional pharmaceutical carriers or excipients such as gelatin, various starches, lactose, calcium phosphate or powdered sugar.
- pharmaceutical carrier as used herein also includes lubricants employed to improve the flow of tablet granulations and which prevent adhesion of tablet material to the surfaces of tablet dies and punches. Suitable lubricants include, for example, talc stearic acid, calcium stearate, magnesium stearate and zinc stearate.
- disintegrating agents added to assist the breakup and dissolution of tablets following admi nistration, as well as coloring and/or flavoring agents to enhance the aesthetic qualities of the tablets and make them more acceptable to the patient.
- Suitable liquid excipients for the preparation of liquid dosage unit forms include water and alcohols such as ethanol, benzyl alcohol and the polyethylene glycols. either with or without the addition of a surfactant.
- the preferred liquid excipients, particularly for injectable preparations include water, physiological and saline solutions, dextrose and glycol solutions such as an aqueous propylene glycol or polyethylene glycol solutions.
- such compositions may contain a non-ionic surfactant having a hydrophile-lipophile balance (HLB) of from about 12 to about 17.
- HLB hydrophile-lipophile balance
- the quantity of surfactant in such formulations ranges from about 5 to 15% by weight.
- the surfactant can be a single component having the above-identified HLB, or a mixture of two or more components having the desired HLB.
- Illustrative of surfactants useful in parenteral formulations are the class of polyoxyethylene sorbitan fatty acid esters as, for example, sorbitan monooleate and the high molecular weight adducts of
- oils can be utilized as carriers or excipients.
- oils include mineral oils, glyceride oils such as lard oil, cod liver oil, peanut oil, sesame oil, corn oil and soybean oil.
- suspending agents may be added as well as agents to control the viscosity, as for example, magnesium aluminum silicate or carboxymethyl-cellulose.
- buffers, preservatives and emulsifying agents may also be added.
- Typical enema preparation of the retention type enema utilizes small volumes, generally much less than about 150 mL for an adult, typically volumes of only a few milliliters are preferred. Excipients and solvents for use in retention anemas should, of course, be selected so as to avoid colonic irritation and should also be selected so as to minimize absorption of the various agents.
- the compounds of this invention can also be administered topically. This can be accomplished by simply
- preparing a solution of the compound to be administered preferably using a solvent known to promote transdermal absorption such as ethanol or dimethyl sulfoxide (DMSO) with or without other excipients.
- a solvent known to promote transdermal absorption such as ethanol or dimethyl sulfoxide (DMSO)
- topical administration will be accomplished using a patch either of the reservoir and porous membrane type or of a solid matrix variety.
- transdermal devices are described in U.S. Pat. Nos. 3,742,951, 3,797,494, 3,996,934, and 4,031,894. These devices generally contain a backing member which defines one of its face surfaces, an active agent permeable adhesive layer defining the other face surface and at least one reservoir containing the active agent interposed between the face surfaces. Alternatively, the active agent may be contained in a plurality of microcapsules
- the active agent is delivered continuously from the reservoir or microcapsules through a membrane into the active agent permeable adhesive, which is in contact with the skin or mucosa of the recipient. If the active agent is absorbed through the skin, a controlled and predetermined flow of the active agent is administered to the recipient.
- the active agent is delivered continuously from the reservoir or microcapsules through a membrane into the active agent permeable adhesive, which is in contact with the skin or mucosa of the recipient. If the active agent is absorbed through the skin, a controlled and predetermined flow of the active agent is administered to the recipient.
- the active agent is delivered continuously from the reservoir or microcapsules through a membrane into the active agent permeable adhesive, which is in contact with the skin or mucosa of the recipient. If the active agent is absorbed through the skin, a controlled and predetermined flow of the active agent is administered to the recipient.
- the active agent is delivered continuously from the reservoir or microcapsules through a membrane into the active agent permeable adhesive, which is in contact with the skin or
- encapsulating agent may also function as the membrane.
- the pharmaceutically active compound is contained in a matrix from which it is delivered in the desired gradual, constant and controlled rate.
- the matrix is permeable to the release of the compound through diffusion or microporous flow.
- the release is rate controlling.
- Such a system, which requires no membrane is described in U.S. Pat. No. 3,921,636. At least two types of release are possible in these systems. Release by diffusion occurs when the matrix is non-porous.
- the pharmaceutically effective compound dissolves in and diffuses through the matrix itself. Release by microporous flow occurs when the pharmaceutically effective compound is transported through a liquid phase in the pores of the matrix.
- the compounds of the present invention may be
- the aerosol preparation may be prepared for use as a topical aerosol or may be prepared for inhalation.
- the aerosol preparation may be in the form of a solution or suspension and may contain other ingredients such as solvents , propellants and/or dispersing agents. Typical examples of aerosol preparations are shown in Remington's Pharmaceutical Sciences, 18th ed., Mack Publishing
- R 2 , R 4 , R 6 and R 7 moieties be methyl.
- R 5 is H or an acyl moiety including formyl and acetyl.
- X is preferably CH 2 A.
- A is preferably R 10 is preferably C 1-6 alkyl,
- R 10 is acyloxyalkylene, especially -CH 2 -O-C(O)CH 3 , hydroxyalkyl (C 2-6 ) especially (CH 2 ) 2 -OH, and pyrimidinyl.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Epidemiology (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Furan Compounds (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP93907336A EP0635009B1 (en) | 1992-04-06 | 1993-03-10 | Novel derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols |
| AU37976/93A AU670433B2 (en) | 1992-04-06 | 1993-03-10 | Novel derivatives of 2,3-dihydro-benzofuranols |
| DE69314022T DE69314022T2 (en) | 1992-04-06 | 1993-03-10 | DERIVATIVES OF 2,3-DIHYDRO-2,2,4,6,7-PENTAALKYL-5-BENZOFURANES |
| JP51745293A JP3246741B2 (en) | 1992-04-06 | 1993-03-10 | Novel derivative of 2,3-dihydrobenzofuranol and process for producing the same |
| US08/318,633 US5721233A (en) | 1992-04-06 | 1993-03-10 | Derivatives of 2,3-dihydro benzofuranols |
| NO943726A NO943726L (en) | 1992-04-06 | 1994-10-05 | New derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols |
| FI944655A FI944655A0 (en) | 1992-04-06 | 1994-10-05 | New 2,3-dihydro-2,2,4,6,7-penta-alkyl-5-benzofuranols |
| KR1019940703513A KR950700895A (en) | 1992-04-06 | 1994-10-05 | Novel Derivatives of 2,3-Dihydro-2,2,4,6,7-Pentaalkyl- 5-Benzofuranols) |
| GR970403227T GR3025577T3 (en) | 1992-04-06 | 1997-12-03 | Novel derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP92400956.6 | 1992-04-06 | ||
| EP92400956 | 1992-04-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1993020057A1 true WO1993020057A1 (en) | 1993-10-14 |
Family
ID=8211646
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1993/002107 WO1993020057A1 (en) | 1992-04-06 | 1993-03-10 | Novel derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols |
Country Status (19)
| Country | Link |
|---|---|
| EP (1) | EP0635009B1 (en) |
| JP (1) | JP3246741B2 (en) |
| KR (1) | KR950700895A (en) |
| AT (1) | ATE158287T1 (en) |
| AU (1) | AU670433B2 (en) |
| CA (1) | CA2132829C (en) |
| DE (1) | DE69314022T2 (en) |
| DK (1) | DK0635009T3 (en) |
| ES (1) | ES2110602T3 (en) |
| FI (1) | FI944655A0 (en) |
| GR (1) | GR3025577T3 (en) |
| HU (1) | HUT68926A (en) |
| IL (1) | IL105318A (en) |
| MX (1) | MX9301950A (en) |
| NO (1) | NO943726L (en) |
| NZ (1) | NZ251134A (en) |
| TW (1) | TW226371B (en) |
| WO (1) | WO1993020057A1 (en) |
| ZA (1) | ZA932327B (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0604995A1 (en) * | 1992-12-31 | 1994-07-06 | LIFEGROUP S.p.A. | Hydroxyamines N-acyl derivatives having scavenger activity and useful in acute and chronic pathologies associated with peroxidation and inflammation phenomena |
| US5510373A (en) * | 1992-04-06 | 1996-04-23 | Merrell Pharmaceuticals Inc. | Cardioprotective agents |
| US5545660A (en) * | 1992-04-07 | 1996-08-13 | Merrell Pharmaceuticals Inc. | Hydrazide derivatives of 3,4-dihydro-2H-1-benzopyrans |
| WO1996028437A1 (en) * | 1995-03-10 | 1996-09-19 | Hoechst Marion Roussel, Inc. | Novel process for preparing 2,3-dihydro-benzofuranol derivatives |
| US5574178A (en) * | 1992-10-16 | 1996-11-12 | Chugai Seiyaku Kabushiki Kaisha | 4,6-Di-t-butyl-dihydrobenzofuran-5-ol and its derivatives |
| EP0906121A1 (en) * | 1996-03-12 | 1999-04-07 | University Of South Florida | Vasoactive effects and free radical generation by beta-amyloid peptides |
| US7652155B2 (en) | 2003-04-14 | 2010-01-26 | Nippon Soda Co., Ltd. | Diamine derivative, production process therefor and antioxidant |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7265152B2 (en) * | 2002-11-01 | 2007-09-04 | Viropharma Incorporated | Benzofuran compounds, compositions and methods for treatment and prophylaxis of hepatitis C viral infections and associated diseases |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0281261A2 (en) * | 1987-02-26 | 1988-09-07 | H. Lundbeck A/S | 3-Aminomethyl derivatives of indane, indoline and dihydrobenzofurane and dihydrobenzothiophene |
| EP0345593A1 (en) * | 1988-06-10 | 1989-12-13 | Takeda Chemical Industries, Ltd. | 2-substituted coumaran derivatives |
| FR2634766A1 (en) * | 1988-08-01 | 1990-02-02 | Foscama Biomed Chim Farma | ACIDS (RS) -2- (2,3-DIHYDRO-5-HYDROXY-4,6,7-TRIMETHYLBENZOFURANNYL) -ACETICS, AND ACIDS 2- (2,3-DIHYDRO-5-ACYLOXY-4,6,7- TRIMETHYLBENZOFURANNYL) -ACETICS AND THEIR ESTERS, USEFUL AS MUCOREGULATING AND ANTI-ISCHEMIC DRUGS, PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| US4975457A (en) * | 1989-09-25 | 1990-12-04 | Merck & Co., Inc. | Trans 2,3-disubstituted-2,3-dihydro-5-hydroxy-benzofurans as inhibitors of leukotriene biosynthesis |
| EP0413668A2 (en) * | 1989-08-18 | 1991-02-20 | Biomedica Foscama Industria Chimico-Farmaceutica S.P.A. | New 2,3-dihydro-5-oxy-4,6,7-trimethylbenzofuranes 2-(RS)-substitutes, useful as antioxidizing pharmaceutical products having mucoregolating and anti-ischemic properties |
| EP0483772A1 (en) * | 1990-11-01 | 1992-05-06 | Takeda Chemical Industries, Ltd. | Aminocoumaran derivatives, their production and use |
-
1993
- 1993-03-10 DE DE69314022T patent/DE69314022T2/en not_active Expired - Fee Related
- 1993-03-10 AU AU37976/93A patent/AU670433B2/en not_active Ceased
- 1993-03-10 CA CA002132829A patent/CA2132829C/en not_active Expired - Fee Related
- 1993-03-10 JP JP51745293A patent/JP3246741B2/en not_active Expired - Fee Related
- 1993-03-10 WO PCT/US1993/002107 patent/WO1993020057A1/en active IP Right Grant
- 1993-03-10 EP EP93907336A patent/EP0635009B1/en not_active Expired - Lifetime
- 1993-03-10 NZ NZ251134A patent/NZ251134A/en unknown
- 1993-03-10 AT AT93907336T patent/ATE158287T1/en not_active IP Right Cessation
- 1993-03-10 DK DK93907336.7T patent/DK0635009T3/en active
- 1993-03-10 HU HU9402864A patent/HUT68926A/en unknown
- 1993-03-10 ES ES93907336T patent/ES2110602T3/en not_active Expired - Lifetime
- 1993-03-31 ZA ZA932327A patent/ZA932327B/en unknown
- 1993-04-01 TW TW082102444A patent/TW226371B/zh active
- 1993-04-05 IL IL105318A patent/IL105318A/en not_active IP Right Cessation
- 1993-04-05 MX MX9301950A patent/MX9301950A/en unknown
-
1994
- 1994-10-05 KR KR1019940703513A patent/KR950700895A/en not_active IP Right Cessation
- 1994-10-05 NO NO943726A patent/NO943726L/en unknown
- 1994-10-05 FI FI944655A patent/FI944655A0/en unknown
-
1997
- 1997-12-03 GR GR970403227T patent/GR3025577T3/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0281261A2 (en) * | 1987-02-26 | 1988-09-07 | H. Lundbeck A/S | 3-Aminomethyl derivatives of indane, indoline and dihydrobenzofurane and dihydrobenzothiophene |
| EP0345593A1 (en) * | 1988-06-10 | 1989-12-13 | Takeda Chemical Industries, Ltd. | 2-substituted coumaran derivatives |
| FR2634766A1 (en) * | 1988-08-01 | 1990-02-02 | Foscama Biomed Chim Farma | ACIDS (RS) -2- (2,3-DIHYDRO-5-HYDROXY-4,6,7-TRIMETHYLBENZOFURANNYL) -ACETICS, AND ACIDS 2- (2,3-DIHYDRO-5-ACYLOXY-4,6,7- TRIMETHYLBENZOFURANNYL) -ACETICS AND THEIR ESTERS, USEFUL AS MUCOREGULATING AND ANTI-ISCHEMIC DRUGS, PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| EP0413668A2 (en) * | 1989-08-18 | 1991-02-20 | Biomedica Foscama Industria Chimico-Farmaceutica S.P.A. | New 2,3-dihydro-5-oxy-4,6,7-trimethylbenzofuranes 2-(RS)-substitutes, useful as antioxidizing pharmaceutical products having mucoregolating and anti-ischemic properties |
| US4975457A (en) * | 1989-09-25 | 1990-12-04 | Merck & Co., Inc. | Trans 2,3-disubstituted-2,3-dihydro-5-hydroxy-benzofurans as inhibitors of leukotriene biosynthesis |
| EP0483772A1 (en) * | 1990-11-01 | 1992-05-06 | Takeda Chemical Industries, Ltd. | Aminocoumaran derivatives, their production and use |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5510373A (en) * | 1992-04-06 | 1996-04-23 | Merrell Pharmaceuticals Inc. | Cardioprotective agents |
| US5545660A (en) * | 1992-04-07 | 1996-08-13 | Merrell Pharmaceuticals Inc. | Hydrazide derivatives of 3,4-dihydro-2H-1-benzopyrans |
| US5574178A (en) * | 1992-10-16 | 1996-11-12 | Chugai Seiyaku Kabushiki Kaisha | 4,6-Di-t-butyl-dihydrobenzofuran-5-ol and its derivatives |
| US5663373A (en) * | 1992-10-16 | 1997-09-02 | Chugai Seiyaku Kabushiki Kaisha | 4-alkoxy-2, 6-di-t-butylphenol derivatives |
| EP0604995A1 (en) * | 1992-12-31 | 1994-07-06 | LIFEGROUP S.p.A. | Hydroxyamines N-acyl derivatives having scavenger activity and useful in acute and chronic pathologies associated with peroxidation and inflammation phenomena |
| US5480645A (en) * | 1992-12-31 | 1996-01-02 | Lifegroup S.P.A. | Hydroxyamines N-acyl derivatives having scavenger activity and useful in acute and chronic pathologies associated with peroxidation and inflammation phenomena |
| WO1996028437A1 (en) * | 1995-03-10 | 1996-09-19 | Hoechst Marion Roussel, Inc. | Novel process for preparing 2,3-dihydro-benzofuranol derivatives |
| US5698696A (en) * | 1995-03-10 | 1997-12-16 | Hoechst Marion Roussel, Inc. | Process for preparing 2,3-dihydro-benzofuranol derivatives |
| EP0906121A1 (en) * | 1996-03-12 | 1999-04-07 | University Of South Florida | Vasoactive effects and free radical generation by beta-amyloid peptides |
| EP0906121A4 (en) * | 1996-03-12 | 2003-04-23 | Univ South Florida | Vasoactive effects and free radical generation by beta-amyloid peptides |
| US7652155B2 (en) | 2003-04-14 | 2010-01-26 | Nippon Soda Co., Ltd. | Diamine derivative, production process therefor and antioxidant |
Also Published As
| Publication number | Publication date |
|---|---|
| HUT68926A (en) | 1995-08-28 |
| NO943726D0 (en) | 1994-10-05 |
| AU3797693A (en) | 1993-11-08 |
| JP3246741B2 (en) | 2002-01-15 |
| NZ251134A (en) | 1997-06-24 |
| IL105318A (en) | 1998-04-05 |
| ZA932327B (en) | 1993-10-18 |
| DK0635009T3 (en) | 1997-10-06 |
| ATE158287T1 (en) | 1997-10-15 |
| IL105318A0 (en) | 1993-08-18 |
| MX9301950A (en) | 1993-10-01 |
| DE69314022D1 (en) | 1997-10-23 |
| EP0635009B1 (en) | 1997-09-17 |
| DE69314022T2 (en) | 1998-02-12 |
| ES2110602T3 (en) | 1998-02-16 |
| AU670433B2 (en) | 1996-07-18 |
| FI944655A (en) | 1994-10-05 |
| JPH08503922A (en) | 1996-04-30 |
| TW226371B (en) | 1994-07-11 |
| EP0635009A1 (en) | 1995-01-25 |
| GR3025577T3 (en) | 1998-03-31 |
| FI944655A0 (en) | 1994-10-05 |
| CA2132829A1 (en) | 1993-10-14 |
| KR950700895A (en) | 1995-02-20 |
| CA2132829C (en) | 1999-01-26 |
| NO943726L (en) | 1994-12-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CZ374692A3 (en) | Proteolytic enzymes inhibitors based on saccharin derivatives | |
| US5395834A (en) | New benzopyran compounds | |
| EP0635009B1 (en) | Novel derivatives of 2,3-dihydro-2,2,4,6,7-pentaalkyl-5-benzofuranols | |
| US5179093A (en) | Quinoline-diones | |
| US5484810A (en) | Tissue protective tocopherol analogs | |
| US5698696A (en) | Process for preparing 2,3-dihydro-benzofuranol derivatives | |
| US5721233A (en) | Derivatives of 2,3-dihydro benzofuranols | |
| US5545660A (en) | Hydrazide derivatives of 3,4-dihydro-2H-1-benzopyrans | |
| CA2132745C (en) | Novel cardioprotective agents | |
| EP0635011B1 (en) | Hydrazide derivatives of 3,4-dihydro-2h-1-benzopyrans | |
| US4757078A (en) | Cyclic aryl hydroxamic acids, derivatives thereof and method of use as anti-allergy agents | |
| FR2704857A1 (en) | Novel substituted benzodioxins, process for their preparation and pharmaceutical compositions containing them | |
| AU705004B2 (en) | 2,3-dihydro-benzofuranol derivatives and process for resolving the same | |
| US5510373A (en) | Cardioprotective agents | |
| US5072006A (en) | Novel benzopyranylpyrrolinone derivatives | |
| JPH09249628A (en) | Derivatives of phenol | |
| PH26915A (en) | Novel benzopyranylpyrrolinone derivatives and its process of preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU CA FI HU JP KR NO NZ US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2132829 Country of ref document: CA Ref document number: 251134 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 944655 Country of ref document: FI |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1993907336 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08318633 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 1993907336 Country of ref document: EP |
|
| WWG | Wipo information: grant in national office |
Ref document number: 1993907336 Country of ref document: EP |