USRE44205E1 - Adenosine A2a receptor antagonists - Google Patents
Adenosine A2a receptor antagonists Download PDFInfo
- Publication number
- USRE44205E1 USRE44205E1 US13/441,140 US201213441140A USRE44205E US RE44205 E1 USRE44205 E1 US RE44205E1 US 201213441140 A US201213441140 A US 201213441140A US RE44205 E USRE44205 E US RE44205E
- Authority
- US
- United States
- Prior art keywords
- alkyl
- alkoxy
- compound
- mmol
- phenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime, expires
Links
- 0 *C1=NN2C(N)=NC3=C(C=NN3C[Y]C)C2=N1 Chemical compound *C1=NN2C(N)=NC3=C(C=NN3C[Y]C)C2=N1 0.000 description 28
- VQKFNUFAXTZWDK-UHFFFAOYSA-N CC1=CC=CO1 Chemical compound CC1=CC=CO1 VQKFNUFAXTZWDK-UHFFFAOYSA-N 0.000 description 11
- BTQZKHUEUDPRST-UHFFFAOYSA-N CC1=CC=CC(F)=C1 Chemical compound CC1=CC=CC(F)=C1 BTQZKHUEUDPRST-UHFFFAOYSA-N 0.000 description 5
- RZTDESRVPFKCBH-UHFFFAOYSA-N CC1=CC=C(C2=CC=C(C)C=C2)C=C1 Chemical compound CC1=CC=C(C2=CC=C(C)C=C2)C=C1 RZTDESRVPFKCBH-UHFFFAOYSA-N 0.000 description 4
- KORDZSMOYNFCQG-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C=C2F)CC1 Chemical compound CN1CCN(C2=CC=C(F)C=C2F)CC1 KORDZSMOYNFCQG-UHFFFAOYSA-N 0.000 description 4
- DTYWJKSSUANMHD-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 Chemical compound COCCOC1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 DTYWJKSSUANMHD-UHFFFAOYSA-N 0.000 description 4
- KALNRVPTCYZOAY-UHFFFAOYSA-N CC(C)C.CN(C)C Chemical compound CC(C)C.CN(C)C KALNRVPTCYZOAY-UHFFFAOYSA-N 0.000 description 3
- GYBMIIMARPGPOJ-UHFFFAOYSA-N CC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CC1=CC=C(N2CCN(C)CC2)C=C1 GYBMIIMARPGPOJ-UHFFFAOYSA-N 0.000 description 3
- PYEHNKXDXBNHQQ-UHFFFAOYSA-N CN1C(=O)NC2=C1C=CC=C2 Chemical compound CN1C(=O)NC2=C1C=CC=C2 PYEHNKXDXBNHQQ-UHFFFAOYSA-N 0.000 description 3
- YODLIZLCKLQILI-UHFFFAOYSA-N CN1CCN(C2=NC=C(F)C=N2)CC1 Chemical compound CN1CCN(C2=NC=C(F)C=N2)CC1 YODLIZLCKLQILI-UHFFFAOYSA-N 0.000 description 3
- KLNBEINLMOKSQN-UHFFFAOYSA-N CN1CCN(CCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1 Chemical compound CN1CCN(CCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1 KLNBEINLMOKSQN-UHFFFAOYSA-N 0.000 description 3
- XQSNEPWSXMYXJZ-UHFFFAOYSA-N C.C.CC1CCCN1C(=O)O Chemical compound C.C.CC1CCCN1C(=O)O XQSNEPWSXMYXJZ-UHFFFAOYSA-N 0.000 description 2
- IJQGEWOMDMINDX-UHFFFAOYSA-N CC(=O)C(C)(C)C.CC(C)(C)C#N.CC(C)(C)O.CN(C)C.[H]C(C)(C)C Chemical compound CC(=O)C(C)(C)C.CC(C)(C)C#N.CC(C)(C)O.CN(C)C.[H]C(C)(C)C IJQGEWOMDMINDX-UHFFFAOYSA-N 0.000 description 2
- BVWRLIPHAURUNY-UHFFFAOYSA-N CC(C)C(=O)CC1=C(F)C=C(F)C=C1 Chemical compound CC(C)C(=O)CC1=C(F)C=C(F)C=C1 BVWRLIPHAURUNY-UHFFFAOYSA-N 0.000 description 2
- WUQGISYDPUNAEN-UHFFFAOYSA-N CC1=CC=C(F)O1 Chemical compound CC1=CC=C(F)O1 WUQGISYDPUNAEN-UHFFFAOYSA-N 0.000 description 2
- CATAGJZATFZGMJ-UHFFFAOYSA-N CC1=CC=C(N2CCN(C)CC2)N=C1 Chemical compound CC1=CC=C(N2CCN(C)CC2)N=C1 CATAGJZATFZGMJ-UHFFFAOYSA-N 0.000 description 2
- YXFVVABEGXRONW-UHFFFAOYSA-N CC1=CC=CC=C1 Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 2
- GNFLFHZJXXFDRA-UHFFFAOYSA-N CC1=CC=CN1C Chemical compound CC1=CC=CN1C GNFLFHZJXXFDRA-UHFFFAOYSA-N 0.000 description 2
- RPLQRYXJBIJBFR-UHFFFAOYSA-N CN1CCC(C(=O)C2=CC=CC=C2)CC1 Chemical compound CN1CCC(C(=O)C2=CC=CC=C2)CC1 RPLQRYXJBIJBFR-UHFFFAOYSA-N 0.000 description 2
- FRKDKQXJOOSEHE-UHFFFAOYSA-N CN1CCN(C2=C(F)C=C(F)C=C2F)CC1 Chemical compound CN1CCN(C2=C(F)C=C(F)C=C2F)CC1 FRKDKQXJOOSEHE-UHFFFAOYSA-N 0.000 description 2
- PYMXZCBFOCGPQE-UHFFFAOYSA-N CN1CCN(C2=CC(CO)=C(CO)C=C2F)CC1 Chemical compound CN1CCN(C2=CC(CO)=C(CO)C=C2F)CC1 PYMXZCBFOCGPQE-UHFFFAOYSA-N 0.000 description 2
- ZCAGHLIBKHFJDU-UHFFFAOYSA-N CN1CCN(C2=CC=C(CO)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(CO)C=C2)CC1 ZCAGHLIBKHFJDU-UHFFFAOYSA-N 0.000 description 2
- OUDJYEDSHVUQCS-UHFFFAOYSA-N CN1CCN(C2=CC=C(Cl)C=C2F)CC1 Chemical compound CN1CCN(C2=CC=C(Cl)C=C2F)CC1 OUDJYEDSHVUQCS-UHFFFAOYSA-N 0.000 description 2
- JIPGOGCZDXFCEG-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(F)C=C2)CC1 JIPGOGCZDXFCEG-UHFFFAOYSA-N 0.000 description 2
- MQSLORJEMXANCA-UHFFFAOYSA-N CN1CCN(C2=CC=C3OCCOC3=C2)CC1 Chemical compound CN1CCN(C2=CC=C3OCCOC3=C2)CC1 MQSLORJEMXANCA-UHFFFAOYSA-N 0.000 description 2
- WQDDXVGJRSTLED-UHFFFAOYSA-N CN1CCN(C2=CC=CC=C2)CC1 Chemical compound CN1CCN(C2=CC=CC=C2)CC1 WQDDXVGJRSTLED-UHFFFAOYSA-N 0.000 description 2
- JUQQCVRAZHUCLU-UHFFFAOYSA-N COCCOC1=CC(F)=C(N2CCN(C)CC2)C(F)=C1 Chemical compound COCCOC1=CC(F)=C(N2CCN(C)CC2)C(F)=C1 JUQQCVRAZHUCLU-UHFFFAOYSA-N 0.000 description 2
- KYQKBQOYLXORKX-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(C)CC2)C(F)=C1 Chemical compound COCCOC1=CC=C(N2CCN(C)CC2)C(F)=C1 KYQKBQOYLXORKX-UHFFFAOYSA-N 0.000 description 2
- QWLMBPWNPGUBMQ-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound COCCOC1=CC=C(N2CCN(C)CC2)C=C1 QWLMBPWNPGUBMQ-UHFFFAOYSA-N 0.000 description 2
- SEFPHZVHTRYDMI-UHFFFAOYSA-N C[Y]CN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 Chemical compound C[Y]CN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 SEFPHZVHTRYDMI-UHFFFAOYSA-N 0.000 description 2
- GJSQZMUCZUWYEM-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCC(NC3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCC(NC3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 GJSQZMUCZUWYEM-UHFFFAOYSA-N 0.000 description 2
- NNPPMTNAJDCUHE-UHFFFAOYSA-N [H]C(C)(C)C Chemical compound [H]C(C)(C)C NNPPMTNAJDCUHE-UHFFFAOYSA-N 0.000 description 2
- PNFZWFBYWNZYSG-UHFFFAOYSA-N C.C=C(C)C.COCC1(C)OCCO1 Chemical compound C.C=C(C)C.COCC1(C)OCCO1 PNFZWFBYWNZYSG-UHFFFAOYSA-N 0.000 description 1
- HWHRJQPLIBKKDX-OKCVXOCRSA-N C/C(=N\O)C1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 Chemical compound C/C(=N\O)C1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 HWHRJQPLIBKKDX-OKCVXOCRSA-N 0.000 description 1
- PEGICTZFUZCELM-WPWMEQJKSA-N C/N=C(\C)C1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 Chemical compound C/N=C(\C)C1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 PEGICTZFUZCELM-WPWMEQJKSA-N 0.000 description 1
- YAEIZKTXSWQXEA-UHFFFAOYSA-N C1=C2OCCOC2=CC(N2CCNCC2)=C1.CC1=CC=C(N2CCNCC2)C(C)=C1.COC1=CC(F)=CC=C1N1CCNCC1.FC1(F)OC2=CC=C(N3CCNCC3)C=C2O1.FC1=CC(Cl)=CC=C1N1CCNCC1.FC1=CC(F)=C(F)C(N2CCNCC2)=C1F.FC1=CC(F)=C(N2CCNCC2)C=C1F.FC1=CC=C(N2CCNCC2)C(F)=C1F Chemical compound C1=C2OCCOC2=CC(N2CCNCC2)=C1.CC1=CC=C(N2CCNCC2)C(C)=C1.COC1=CC(F)=CC=C1N1CCNCC1.FC1(F)OC2=CC=C(N3CCNCC3)C=C2O1.FC1=CC(Cl)=CC=C1N1CCNCC1.FC1=CC(F)=C(F)C(N2CCNCC2)=C1F.FC1=CC(F)=C(N2CCNCC2)C=C1F.FC1=CC=C(N2CCNCC2)C(F)=C1F YAEIZKTXSWQXEA-UHFFFAOYSA-N 0.000 description 1
- BMMLZFDDNUUGFM-UHFFFAOYSA-N C1=C2OCOC2=CC(N2CCNCC2)=C1.C1=CC=C(N2CCCNCC2)C=C1.CC1=CC=C(N2CCNCC2)C(F)=C1.ClC1=CC=C(N2CCCNCC2)C=C1.ClC1=CC=C(N2CCNCC2)C(Cl)=C1.FC1=CC(F)=C(N2CCNCC2)C(F)=C1.FC1=CC=C(N2CCCNCC2)C(F)=C1.FC1=CC=C(N2CCNCC2)C=C1F Chemical compound C1=C2OCOC2=CC(N2CCNCC2)=C1.C1=CC=C(N2CCCNCC2)C=C1.CC1=CC=C(N2CCNCC2)C(F)=C1.ClC1=CC=C(N2CCCNCC2)C=C1.ClC1=CC=C(N2CCNCC2)C(Cl)=C1.FC1=CC(F)=C(N2CCNCC2)C(F)=C1.FC1=CC=C(N2CCCNCC2)C(F)=C1.FC1=CC=C(N2CCNCC2)C=C1F BMMLZFDDNUUGFM-UHFFFAOYSA-N 0.000 description 1
- PMQAQFPEARZWAK-UHFFFAOYSA-N C1=CC(N2CCNCC2)=CC=C1OC1CCOCC1.C1=CN=CC(N2CCNCC2)=C1.CC(C)COC1=CC=C(N2CCNCC2)C=C1.CC1(COC2=CC=C(N3CCNCC3)C=C2)OCCO1.CCC(C)COC1=CC=C(N2CCNCC2)C=C1.CO(S)CCOC1=CC=C(N2CCNCC2)C=C1 Chemical compound C1=CC(N2CCNCC2)=CC=C1OC1CCOCC1.C1=CN=CC(N2CCNCC2)=C1.CC(C)COC1=CC=C(N2CCNCC2)C=C1.CC1(COC2=CC=C(N3CCNCC3)C=C2)OCCO1.CCC(C)COC1=CC=C(N2CCNCC2)C=C1.CO(S)CCOC1=CC=C(N2CCNCC2)C=C1 PMQAQFPEARZWAK-UHFFFAOYSA-N 0.000 description 1
- XZOWKYIINLUISD-UHFFFAOYSA-N C1=CC(N2CCNCC2)=CC=C1OCC1CC1.CC(=O)COC1=CC=C(N2CCNCC2)C=C1.CC(C)OCCOC1=CC=C(N2CCNCC2)C=C1.CCOCCOC1=CC=C(N2CCNCC2)C=C1.CN(C)CCCOC1=CC=C(N2CCNCC2)C=C1.CN(C)CCOC1=CC=C(N2CCNCC2)C=C1.COCC(COC)OC1=CC=C(N2CCNCC2)C=C1.COCCOCCOC1=CC=C(N2CCNCC2)C=C1.FC(F)(F)COCCOC1=CC=C(N2CCNCC2)C=C1.OCCOC1=CC=C(N2CCNCC2)C=C1 Chemical compound C1=CC(N2CCNCC2)=CC=C1OCC1CC1.CC(=O)COC1=CC=C(N2CCNCC2)C=C1.CC(C)OCCOC1=CC=C(N2CCNCC2)C=C1.CCOCCOC1=CC=C(N2CCNCC2)C=C1.CN(C)CCCOC1=CC=C(N2CCNCC2)C=C1.CN(C)CCOC1=CC=C(N2CCNCC2)C=C1.COCC(COC)OC1=CC=C(N2CCNCC2)C=C1.COCCOCCOC1=CC=C(N2CCNCC2)C=C1.FC(F)(F)COCCOC1=CC=C(N2CCNCC2)C=C1.OCCOC1=CC=C(N2CCNCC2)C=C1 XZOWKYIINLUISD-UHFFFAOYSA-N 0.000 description 1
- VALOYDQRJKVNRO-UHFFFAOYSA-N C1=CC(N2CCOCC2)=CC=C1N1CCNCC1.CC1=CC(F)=CC=C1N1CCNCC1.CC1=CC=C(N2CCNCC2)C(F)=C1.CC1=CC=C(N2CCNCC2)C=C1.CC1=CC=C(N2CCNCC2)C=C1F.CCCOC1=C(OCCOC)C=CC(N2CCNCC2)=C1.CNCCN(C)C1=CC=C(OCCOC)C=C1C(F)(F)F.COCCOC1=CC=C(N2CCNCC2)C(C(F)(F)F)=C1 Chemical compound C1=CC(N2CCOCC2)=CC=C1N1CCNCC1.CC1=CC(F)=CC=C1N1CCNCC1.CC1=CC=C(N2CCNCC2)C(F)=C1.CC1=CC=C(N2CCNCC2)C=C1.CC1=CC=C(N2CCNCC2)C=C1F.CCCOC1=C(OCCOC)C=CC(N2CCNCC2)=C1.CNCCN(C)C1=CC=C(OCCOC)C=C1C(F)(F)F.COCCOC1=CC=C(N2CCNCC2)C(C(F)(F)F)=C1 VALOYDQRJKVNRO-UHFFFAOYSA-N 0.000 description 1
- CBYRHLAABCNDEQ-UHFFFAOYSA-N C1CC2(CCN1)OCCO2.CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21.CN=C1CCN(CCN2N=CC3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1.CON.NC1=NC2=C(C=NN2CCN2CCC(=O)CC2)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(C=NN2CCN2CCC3(CC2)OCCO3)C2=NC(C3=CC=CO3)=NN12 Chemical compound C1CC2(CCN1)OCCO2.CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21.CN=C1CCN(CCN2N=CC3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1.CON.NC1=NC2=C(C=NN2CCN2CCC(=O)CC2)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(C=NN2CCN2CCC3(CC2)OCCO3)C2=NC(C3=CC=CO3)=NN12 CBYRHLAABCNDEQ-UHFFFAOYSA-N 0.000 description 1
- NUSNXWASQJLNKW-UHFFFAOYSA-N C=O.CC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound C=O.CC1=CC=C(N2CCN(C)CC2)C=C1 NUSNXWASQJLNKW-UHFFFAOYSA-N 0.000 description 1
- SYBYTAAJFKOIEJ-UHFFFAOYSA-N CC(=O)C(C)C Chemical compound CC(=O)C(C)C SYBYTAAJFKOIEJ-UHFFFAOYSA-N 0.000 description 1
- MKHCEQBLJLARIA-UHFFFAOYSA-N CC(=O)C1(C2=CC=CC=C2)CCN(C)CC1 Chemical compound CC(=O)C1(C2=CC=CC=C2)CCN(C)CC1 MKHCEQBLJLARIA-UHFFFAOYSA-N 0.000 description 1
- IRIZGAMYKHTLKS-UHFFFAOYSA-N CC(=O)C1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CC(=O)C1=CC=C(N2CCN(C)CC2)C=C1 IRIZGAMYKHTLKS-UHFFFAOYSA-N 0.000 description 1
- SLTKYVPKBPNETL-UHFFFAOYSA-N CC(=O)COC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CC(=O)COC1=CC=C(N2CCN(C)CC2)C=C1 SLTKYVPKBPNETL-UHFFFAOYSA-N 0.000 description 1
- NKJFLSHLZLPJDB-UHFFFAOYSA-N CC(=O)NNC1=NC(N)=NC2=C1C=NN2 Chemical compound CC(=O)NNC1=NC(N)=NC2=C1C=NN2 NKJFLSHLZLPJDB-UHFFFAOYSA-N 0.000 description 1
- VIGPZOSCEXDBQZ-UHFFFAOYSA-N CC(=O)OCCOC1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 Chemical compound CC(=O)OCCOC1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1 VIGPZOSCEXDBQZ-UHFFFAOYSA-N 0.000 description 1
- TUOPNMUYLJOWLF-UHFFFAOYSA-N CC(C)(C)[Si](C)(C)Cl.CC(C)(C)[Si](C)(C)OCCC1=CC=C(Br)C=C1.CC(C)(C)[Si](C)(C)OCCC1=CC=C(C2=CC=CC=N2)C=C1.CCCC[Sn](CCCC)(CCCC)C1=CC=CC=N1.CS(=O)(=O)Cl.CS(=O)(=O)OCCC1=CC=C(C2=CC=CN=C2)C=C1.NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CC=CC=N3)C=C2)C2=NC(C3=CC=CO3)=NN12.OCCC1=CC=C(Br)C=C1.OCCC1=CC=C(C2=CC=CC=N2)C=C1 Chemical compound CC(C)(C)[Si](C)(C)Cl.CC(C)(C)[Si](C)(C)OCCC1=CC=C(Br)C=C1.CC(C)(C)[Si](C)(C)OCCC1=CC=C(C2=CC=CC=N2)C=C1.CCCC[Sn](CCCC)(CCCC)C1=CC=CC=N1.CS(=O)(=O)Cl.CS(=O)(=O)OCCC1=CC=C(C2=CC=CN=C2)C=C1.NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CC=CC=N3)C=C2)C2=NC(C3=CC=CO3)=NN12.OCCC1=CC=C(Br)C=C1.OCCC1=CC=C(C2=CC=CC=N2)C=C1 TUOPNMUYLJOWLF-UHFFFAOYSA-N 0.000 description 1
- OVCHQRXVZXVQNQ-UHFFFAOYSA-N CC(C)C(=O)C(C)(C)C Chemical compound CC(C)C(=O)C(C)(C)C OVCHQRXVZXVQNQ-UHFFFAOYSA-N 0.000 description 1
- XNMVDUSWCLEESX-UHFFFAOYSA-N CC(C)C(=O)C(C)(C)NC(=O)OC(C)(C)C Chemical compound CC(C)C(=O)C(C)(C)NC(=O)OC(C)(C)C XNMVDUSWCLEESX-UHFFFAOYSA-N 0.000 description 1
- OITFSBRKAUAGND-UHFFFAOYSA-N CC(C)C(=O)C1=CC=C(Cl)C=C1 Chemical compound CC(C)C(=O)C1=CC=C(Cl)C=C1 OITFSBRKAUAGND-UHFFFAOYSA-N 0.000 description 1
- DEKHBCCIHCQNFM-UHFFFAOYSA-N CC(C)C(=O)C1=CC=C2OCOC2=C1 Chemical compound CC(C)C(=O)C1=CC=C2OCOC2=C1 DEKHBCCIHCQNFM-UHFFFAOYSA-N 0.000 description 1
- TWDULYDQIGVEKY-UHFFFAOYSA-N CC(C)C(=O)C1=CC=CN=C1 Chemical compound CC(C)C(=O)C1=CC=CN=C1 TWDULYDQIGVEKY-UHFFFAOYSA-N 0.000 description 1
- OTQCCAKDOPROBT-UHFFFAOYSA-N CC(C)C(=O)C1=CC=CS1 Chemical compound CC(C)C(=O)C1=CC=CS1 OTQCCAKDOPROBT-UHFFFAOYSA-N 0.000 description 1
- XRLQPWQYUOVNTR-UHFFFAOYSA-N CC(C)C(=O)C1CCCCC1 Chemical compound CC(C)C(=O)C1CCCCC1 XRLQPWQYUOVNTR-UHFFFAOYSA-N 0.000 description 1
- KJWBHOKFRSFCBZ-UHFFFAOYSA-N CC(C)C(=O)CC(C)(C)C Chemical compound CC(C)C(=O)CC(C)(C)C KJWBHOKFRSFCBZ-UHFFFAOYSA-N 0.000 description 1
- VBKBGKJPJXVZQM-UHFFFAOYSA-N CC(C)C(=O)CC1=C(F)C=CC=C1 Chemical compound CC(C)C(=O)CC1=C(F)C=CC=C1 VBKBGKJPJXVZQM-UHFFFAOYSA-N 0.000 description 1
- BQSYOEMIUFWQMI-UHFFFAOYSA-N CC(C)C(=O)CC1=CC(CO)=C(CO)C=C1 Chemical compound CC(C)C(=O)CC1=CC(CO)=C(CO)C=C1 BQSYOEMIUFWQMI-UHFFFAOYSA-N 0.000 description 1
- FWFUIQDDBOIOMR-UHFFFAOYSA-N CC(C)C(=O)N1CCCC1 Chemical compound CC(C)C(=O)N1CCCC1 FWFUIQDDBOIOMR-UHFFFAOYSA-N 0.000 description 1
- GSBBRQAUAKEVIN-UHFFFAOYSA-N CC(C)C(=O)N1CCOCC1 Chemical compound CC(C)C(=O)N1CCOCC1 GSBBRQAUAKEVIN-UHFFFAOYSA-N 0.000 description 1
- OEIZGTXQZLSLFL-UHFFFAOYSA-N CC(C)C(=O)NC(C)(C)C Chemical compound CC(C)C(=O)NC(C)(C)C OEIZGTXQZLSLFL-UHFFFAOYSA-N 0.000 description 1
- OAWARBLAOUYGLA-UHFFFAOYSA-N CC(C)C(=S)NCCC1=CC=CC=C1 Chemical compound CC(C)C(=S)NCCC1=CC=CC=C1 OAWARBLAOUYGLA-UHFFFAOYSA-N 0.000 description 1
- IMGZVSKYRRPBFG-UHFFFAOYSA-N CC(C)COC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CC(C)COC1=CC=C(N2CCN(C)CC2)C=C1 IMGZVSKYRRPBFG-UHFFFAOYSA-N 0.000 description 1
- AXVMQLBQSLLJBF-UHFFFAOYSA-N CC(C)OCCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CC(C)OCCOC1=CC=C(N2CCN(C)CC2)C=C1 AXVMQLBQSLLJBF-UHFFFAOYSA-N 0.000 description 1
- GSUMWUCWOGBILU-UHFFFAOYSA-N CC(C)S(=O)(=O)C1=CC(Cl)=CC=C1Cl Chemical compound CC(C)S(=O)(=O)C1=CC(Cl)=CC=C1Cl GSUMWUCWOGBILU-UHFFFAOYSA-N 0.000 description 1
- WQZTWNHNXOSAAR-UHFFFAOYSA-N CC(C)S(=O)(=O)C1=CC=C(Br)C=C1 Chemical compound CC(C)S(=O)(=O)C1=CC=C(Br)C=C1 WQZTWNHNXOSAAR-UHFFFAOYSA-N 0.000 description 1
- RELQPXUEKMBSKZ-UHFFFAOYSA-N CC(C)S(=O)(=O)C1=CC=C(CO)C(CO)=C1 Chemical compound CC(C)S(=O)(=O)C1=CC=C(CO)C(CO)=C1 RELQPXUEKMBSKZ-UHFFFAOYSA-N 0.000 description 1
- CNPPSUBMCNJXTO-UHFFFAOYSA-N CC(C)S(=O)(=O)C1=CC=C(CO)C=C1 Chemical compound CC(C)S(=O)(=O)C1=CC=C(CO)C=C1 CNPPSUBMCNJXTO-UHFFFAOYSA-N 0.000 description 1
- DMKKIIPVYDZMNZ-UHFFFAOYSA-N CC(C)S(=O)(=O)C1=CC=CC=C1F Chemical compound CC(C)S(=O)(=O)C1=CC=CC=C1F DMKKIIPVYDZMNZ-UHFFFAOYSA-N 0.000 description 1
- DVLAQABYXMCMHT-UHFFFAOYSA-N CC(CCl)N1CCN(C2=C(F)C=C(F)C=C2)CC1.CC(CO)N1CCN(C2=C(F)C=C(F)C=C2)CC1 Chemical compound CC(CCl)N1CCN(C2=C(F)C=C(F)C=C2)CC1.CC(CO)N1CCN(C2=C(F)C=C(F)C=C2)CC1 DVLAQABYXMCMHT-UHFFFAOYSA-N 0.000 description 1
- XCQOIKAZLNOGBC-UHFFFAOYSA-N CC(CN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21)N1CCN(C2=C(F)C=C(F)C=C2)CC1 Chemical compound CC(CN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21)N1CCN(C2=C(F)C=C(F)C=C2)CC1 XCQOIKAZLNOGBC-UHFFFAOYSA-N 0.000 description 1
- XZEFCIXUONTJNE-UHFFFAOYSA-N CC(CN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21)N1CCN(C2=CC=C(Cl)C=C2)CC1 Chemical compound CC(CN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21)N1CCN(C2=CC=C(Cl)C=C2)CC1 XZEFCIXUONTJNE-UHFFFAOYSA-N 0.000 description 1
- WDOFHQYFMTXLMP-UHFFFAOYSA-N CC(CO)N1CCN(C2=C(F)C=C(F)C=C2)CC1.CCOC(=O)C(C)N1CCN(C2=C(F)C=C(F)C=C2)CC1 Chemical compound CC(CO)N1CCN(C2=C(F)C=C(F)C=C2)CC1.CCOC(=O)C(C)N1CCN(C2=C(F)C=C(F)C=C2)CC1 WDOFHQYFMTXLMP-UHFFFAOYSA-N 0.000 description 1
- FXVKZGVIXVXQDV-UHFFFAOYSA-N CC1(COC)OCCO1 Chemical compound CC1(COC)OCCO1 FXVKZGVIXVXQDV-UHFFFAOYSA-N 0.000 description 1
- VRMMZSVAKYRZAB-UHFFFAOYSA-N CC1=C(Br)C=CO1 Chemical compound CC1=C(Br)C=CO1 VRMMZSVAKYRZAB-UHFFFAOYSA-N 0.000 description 1
- MSEXWGQOKSTHMS-UHFFFAOYSA-N CC1=C(C)C=C(N2CCNCC2)C(F)=C1.CC1=CC(F)=C(N2CCNCC2)C(F)=C1.CC1CN(C2=CC=C(F)C=C2F)CCN1.CCCOC1=C(F)C=C(N2CCNCC2)C=C1F.COC1=CC(F)=CC(F)=C1N1CCNCC1.COCCOC1=C(C)C=C(N2CCNCC2)C=C1.COCCOC1=CC=C(N2CCNCC2)C(F)=C1.FC1(F)OC2=CC=C(N3CCNCC3)C=C2OC1(F)F Chemical compound CC1=C(C)C=C(N2CCNCC2)C(F)=C1.CC1=CC(F)=C(N2CCNCC2)C(F)=C1.CC1CN(C2=CC=C(F)C=C2F)CCN1.CCCOC1=C(F)C=C(N2CCNCC2)C=C1F.COC1=CC(F)=CC(F)=C1N1CCNCC1.COCCOC1=C(C)C=C(N2CCNCC2)C=C1.COCCOC1=CC=C(N2CCNCC2)C(F)=C1.FC1(F)OC2=CC=C(N3CCNCC3)C=C2OC1(F)F MSEXWGQOKSTHMS-UHFFFAOYSA-N 0.000 description 1
- FJSKXQVRKZTKSI-UHFFFAOYSA-N CC1=C(C)OC=C1 Chemical compound CC1=C(C)OC=C1 FJSKXQVRKZTKSI-UHFFFAOYSA-N 0.000 description 1
- VNAVPOVAVJZKSY-UHFFFAOYSA-N CC1=CC(C)=CC(C(=O)C(C)C)=C1 Chemical compound CC1=CC(C)=CC(C(=O)C(C)C)=C1 VNAVPOVAVJZKSY-UHFFFAOYSA-N 0.000 description 1
- KHBIQXLDXZKXNI-UHFFFAOYSA-N CC1=CC(C)=NC(N2CCNCC2)=N1.CC1=NC(N2CCNCC2)=NC=C1.NC1=CC(N2CCNCC2)=NC(N)=N1.ON1=CC=CC=C1N1CCNCC1 Chemical compound CC1=CC(C)=NC(N2CCNCC2)=N1.CC1=NC(N2CCNCC2)=NC=C1.NC1=CC(N2CCNCC2)=NC(N)=N1.ON1=CC=CC=C1N1CCNCC1 KHBIQXLDXZKXNI-UHFFFAOYSA-N 0.000 description 1
- OGIOYZCZNDOKKF-UHFFFAOYSA-N CC1=CC(CO)=CC=C1N1CCN(C)CC1 Chemical compound CC1=CC(CO)=CC=C1N1CCN(C)CC1 OGIOYZCZNDOKKF-UHFFFAOYSA-N 0.000 description 1
- YISYUYYETHYYMD-UHFFFAOYSA-N CC1=CC(F)=CC(F)=C1 Chemical compound CC1=CC(F)=CC(F)=C1 YISYUYYETHYYMD-UHFFFAOYSA-N 0.000 description 1
- JMDFFLLOSTUHAR-UHFFFAOYSA-N CC1=CC(F)=CC=C1N1CCN(C)CC1 Chemical compound CC1=CC(F)=CC=C1N1CCN(C)CC1 JMDFFLLOSTUHAR-UHFFFAOYSA-N 0.000 description 1
- PVFIDFYACCGDBG-UHFFFAOYSA-N CC1=CC(N2CCN(C)CC2)=CC=C1 Chemical compound CC1=CC(N2CCN(C)CC2)=CC=C1 PVFIDFYACCGDBG-UHFFFAOYSA-N 0.000 description 1
- GHTQKXABBCYQBO-UHFFFAOYSA-N CC1=CC(N2CCN(C)CC2)=CC=C1Cl Chemical compound CC1=CC(N2CCN(C)CC2)=CC=C1Cl GHTQKXABBCYQBO-UHFFFAOYSA-N 0.000 description 1
- VWGRESZTMULPDE-UHFFFAOYSA-N CC1=CC=C(Br)O1 Chemical compound CC1=CC=C(Br)O1 VWGRESZTMULPDE-UHFFFAOYSA-N 0.000 description 1
- VLKIRFAISMBUCB-UHFFFAOYSA-N CC1=CC=C(C(=O)C(C)C)C=C1 Chemical compound CC1=CC=C(C(=O)C(C)C)C=C1 VLKIRFAISMBUCB-UHFFFAOYSA-N 0.000 description 1
- GSNUFIFRDBKVIE-UHFFFAOYSA-N CC1=CC=C(C)O1 Chemical compound CC1=CC=C(C)O1 GSNUFIFRDBKVIE-UHFFFAOYSA-N 0.000 description 1
- ZZLCFHIKESPLTH-UHFFFAOYSA-N CC1=CC=C(C2=CC=CC=C2)C=C1 Chemical compound CC1=CC=C(C2=CC=CC=C2)C=C1 ZZLCFHIKESPLTH-UHFFFAOYSA-N 0.000 description 1
- MRAIVJPPOXSJNG-UHFFFAOYSA-N CC1=CC=C(C2=CC=NC=C2)C=C1 Chemical compound CC1=CC=C(C2=CC=NC=C2)C=C1 MRAIVJPPOXSJNG-UHFFFAOYSA-N 0.000 description 1
- VMQIABRWAZWBEC-UHFFFAOYSA-N CC1=CC=C(C2=CN=C(C)C=C2)C=C1 Chemical compound CC1=CC=C(C2=CN=C(C)C=C2)C=C1 VMQIABRWAZWBEC-UHFFFAOYSA-N 0.000 description 1
- KMTDMTZBNYGUNX-UHFFFAOYSA-N CC1=CC=C(CO)C=C1 Chemical compound CC1=CC=C(CO)C=C1 KMTDMTZBNYGUNX-UHFFFAOYSA-N 0.000 description 1
- NPDACUSDTOMAMK-UHFFFAOYSA-N CC1=CC=C(Cl)C=C1 Chemical compound CC1=CC=C(Cl)C=C1 NPDACUSDTOMAMK-UHFFFAOYSA-N 0.000 description 1
- GQSHJDSSWWLNQR-UHFFFAOYSA-N CC1=CC=C(Cl)O1 Chemical compound CC1=CC=C(Cl)O1 GQSHJDSSWWLNQR-UHFFFAOYSA-N 0.000 description 1
- FZMPLKVGINKUJZ-UHFFFAOYSA-N CC1=CC=C(F)C(F)=C1 Chemical compound CC1=CC=C(F)C(F)=C1 FZMPLKVGINKUJZ-UHFFFAOYSA-N 0.000 description 1
- MPXDAIBTYWGBSL-UHFFFAOYSA-N CC1=CC=C(F)C=C1F Chemical compound CC1=CC=C(F)C=C1F MPXDAIBTYWGBSL-UHFFFAOYSA-N 0.000 description 1
- GBYHLLBCDBFCPJ-UHFFFAOYSA-N CC1=CC=C(N2CCN(C)CC2)C(C)=C1 Chemical compound CC1=CC=C(N2CCN(C)CC2)C(C)=C1 GBYHLLBCDBFCPJ-UHFFFAOYSA-N 0.000 description 1
- KALQCFQQZPYUJN-UHFFFAOYSA-N CC1=CC=C(N2CCN(C)CC2)C(F)=C1 Chemical compound CC1=CC=C(N2CCN(C)CC2)C(F)=C1 KALQCFQQZPYUJN-UHFFFAOYSA-N 0.000 description 1
- HTRDGRJOXFZKCH-UHFFFAOYSA-N CC1=CC=C(N2CCN(C)CC2)C=C1C Chemical compound CC1=CC=C(N2CCN(C)CC2)C=C1C HTRDGRJOXFZKCH-UHFFFAOYSA-N 0.000 description 1
- ZJTZEUQQUYHNHH-UHFFFAOYSA-N CC1=CC=C(N2CCNCC2)C(C(F)(F)F)=C1.CCCOC1=CC(F)=CC=C1N1CCNCC1.COCCOC1=C(C)C=C(N2CCNCC2)C=C1.COCCOC1=CC(F)=C(N2CCNCC2)C(F)=C1.COCCOC1=CC(N2CCNCC2)=CC=C1F.COCCOC1=CC=C(N2CCNCC2)C(C#N)=C1.COCCOC1=CC=C(N2CCNCC2)C(C)=C1.[C-]#[N+]C1=C(OCCOC)C=CC(N2CCNCC2)=C1 Chemical compound CC1=CC=C(N2CCNCC2)C(C(F)(F)F)=C1.CCCOC1=CC(F)=CC=C1N1CCNCC1.COCCOC1=C(C)C=C(N2CCNCC2)C=C1.COCCOC1=CC(F)=C(N2CCNCC2)C(F)=C1.COCCOC1=CC(N2CCNCC2)=CC=C1F.COCCOC1=CC=C(N2CCNCC2)C(C#N)=C1.COCCOC1=CC=C(N2CCNCC2)C(C)=C1.[C-]#[N+]C1=C(OCCOC)C=CC(N2CCNCC2)=C1 ZJTZEUQQUYHNHH-UHFFFAOYSA-N 0.000 description 1
- ZPHBESSMPKFLOI-UHFFFAOYSA-N CC1=CC=C(N2CCNCC2)N=C1.CCC1=CC=C(N2CCNCC2)C=C1.CCC1=CC=C(N2CCNCC2)C=C1.N#CC1=CC(F)=CC=C1N1CCNCC1.O=C=O.O=[N+]([O-])C1=CC(F)=CC=C1N1CCNCC1.O=[SH](=O)NC1=CC=C(N2CCNCC2)C=C1.[C-]#[N+]C1=CC(F)=C(N2CCNCC2)C(F)=C1 Chemical compound CC1=CC=C(N2CCNCC2)N=C1.CCC1=CC=C(N2CCNCC2)C=C1.CCC1=CC=C(N2CCNCC2)C=C1.N#CC1=CC(F)=CC=C1N1CCNCC1.O=C=O.O=[N+]([O-])C1=CC(F)=CC=C1N1CCNCC1.O=[SH](=O)NC1=CC=C(N2CCNCC2)C=C1.[C-]#[N+]C1=CC(F)=C(N2CCNCC2)C(F)=C1 ZPHBESSMPKFLOI-UHFFFAOYSA-N 0.000 description 1
- XAHJYFSERRNQBC-UHFFFAOYSA-N CC1=CC=C(OCCCCCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)C=C1C Chemical compound CC1=CC=C(OCCCCCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)C=C1C XAHJYFSERRNQBC-UHFFFAOYSA-N 0.000 description 1
- DUJYZGXBCPOPDD-UHFFFAOYSA-N CC1=CC=C(S(=O)(=O)C(C)C)C=C1 Chemical compound CC1=CC=C(S(=O)(=O)C(C)C)C=C1 DUJYZGXBCPOPDD-UHFFFAOYSA-N 0.000 description 1
- OHLKTEUYNSFZBB-UHFFFAOYSA-N CC1=CC=C(S(=O)(=O)OCCOS(=O)(=O)C2=CC=C(C)C=C2)C=C1.CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21.CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21.NC1=NC2=C(C=NN2)C2=NC(C3=CC=CO3)=NN12 Chemical compound CC1=CC=C(S(=O)(=O)OCCOS(=O)(=O)C2=CC=C(C)C=C2)C=C1.CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21.CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21.NC1=NC2=C(C=NN2)C2=NC(C3=CC=CO3)=NN12 OHLKTEUYNSFZBB-UHFFFAOYSA-N 0.000 description 1
- BAFFKDVNEHCBFX-UHFFFAOYSA-N CC1=CC=C(SCCN2/N=C\C3=C2N=C(C)N2N=C(C4=CC=CO4)N=C32)C=C1 Chemical compound CC1=CC=C(SCCN2/N=C\C3=C2N=C(C)N2N=C(C4=CC=CO4)N=C32)C=C1 BAFFKDVNEHCBFX-UHFFFAOYSA-N 0.000 description 1
- VOBFYAXUVBEUGM-UHFFFAOYSA-N CC1=CC=C([N+](=O)[O-])O1 Chemical compound CC1=CC=C([N+](=O)[O-])O1 VOBFYAXUVBEUGM-UHFFFAOYSA-N 0.000 description 1
- NHYJXISUOSCSJD-UHFFFAOYSA-N CC1=CC=CC(C)=C1N1CCN(C)CC1 Chemical compound CC1=CC=CC(C)=C1N1CCN(C)CC1 NHYJXISUOSCSJD-UHFFFAOYSA-N 0.000 description 1
- LONDBHTYIYKOCM-UHFFFAOYSA-N CC1=CC=CC(C2=CC=CN=C2)=C1 Chemical compound CC1=CC=CC(C2=CC=CN=C2)=C1 LONDBHTYIYKOCM-UHFFFAOYSA-N 0.000 description 1
- BSKHPKMHTQYZBB-UHFFFAOYSA-N CC1=CC=CC=N1 Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 1
- TVCXVUHHCUYLGX-UHFFFAOYSA-N CC1=CC=CN1 Chemical compound CC1=CC=CN1 TVCXVUHHCUYLGX-UHFFFAOYSA-N 0.000 description 1
- ATQUFXWBVZUTKO-UHFFFAOYSA-N CC1=CCCC1 Chemical compound CC1=CCCC1 ATQUFXWBVZUTKO-UHFFFAOYSA-N 0.000 description 1
- HKUVCJBKEOIQFL-UHFFFAOYSA-N CC1=CN=C(N2CCN(C)CC2)N=C1 Chemical compound CC1=CN=C(N2CCN(C)CC2)N=C1 HKUVCJBKEOIQFL-UHFFFAOYSA-N 0.000 description 1
- PMCOWOCKUQWYRL-UHFFFAOYSA-N CC1=NC(C)=NC=C1 Chemical compound CC1=NC(C)=NC=C1 PMCOWOCKUQWYRL-UHFFFAOYSA-N 0.000 description 1
- VYZPEWUEPPGXGA-UHFFFAOYSA-N CC1=NC(N2CCN(C)CC2)=CC=C1 Chemical compound CC1=NC(N2CCN(C)CC2)=CC=C1 VYZPEWUEPPGXGA-UHFFFAOYSA-N 0.000 description 1
- HRQGGDPXDLPPBQ-UHFFFAOYSA-N CC1=NC(N2CCN(C)CC2)=NC=C1 Chemical compound CC1=NC(N2CCN(C)CC2)=NC=C1 HRQGGDPXDLPPBQ-UHFFFAOYSA-N 0.000 description 1
- FSQWOPYBUDAELG-UHFFFAOYSA-N CC1=NC2=C(/C=N\N2CCOC2=CC(C)=C(C)C=C2)C2=NC(C3=CC=CO3)=NN12 Chemical compound CC1=NC2=C(/C=N\N2CCOC2=CC(C)=C(C)C=C2)C2=NC(C3=CC=CO3)=NN12 FSQWOPYBUDAELG-UHFFFAOYSA-N 0.000 description 1
- PKAIVNOWLQBDCL-UHFFFAOYSA-N CC1=NC2=C(/C=N\N2CCOC2=CC=C(OC3=CC=CC=C3)C=C2)C2=NC(C3=CC=CO3)=NN12 Chemical compound CC1=NC2=C(/C=N\N2CCOC2=CC=C(OC3=CC=CC=C3)C=C2)C2=NC(C3=CC=CO3)=NN12 PKAIVNOWLQBDCL-UHFFFAOYSA-N 0.000 description 1
- ILBZSNOGPTUKHC-UHFFFAOYSA-N CC1CN(C)C1 Chemical compound CC1CN(C)C1 ILBZSNOGPTUKHC-UHFFFAOYSA-N 0.000 description 1
- QIOGGRNDZWGKDL-UHFFFAOYSA-N CC1CN(C)CCN1C1=CC=C(CO)C=C1 Chemical compound CC1CN(C)CCN1C1=CC=C(CO)C=C1 QIOGGRNDZWGKDL-UHFFFAOYSA-N 0.000 description 1
- QPIUASVXPHNBQP-UHFFFAOYSA-N CC1CN(C)CCN1C1=CC=C(Cl)C=C1 Chemical compound CC1CN(C)CCN1C1=CC=C(Cl)C=C1 QPIUASVXPHNBQP-UHFFFAOYSA-N 0.000 description 1
- RAMPYLGYEGEHJD-UHFFFAOYSA-N CC1CN(C2=CC=C(F)C=C2F)CCN1C Chemical compound CC1CN(C2=CC=C(F)C=C2F)CCN1C RAMPYLGYEGEHJD-UHFFFAOYSA-N 0.000 description 1
- OTFKRHOATUETFR-UHFFFAOYSA-N CCC(=O)N1CCN(C2=CC=CC=C2)CC1 Chemical compound CCC(=O)N1CCN(C2=CC=CC=C2)CC1 OTFKRHOATUETFR-UHFFFAOYSA-N 0.000 description 1
- CCBIPVWWCZLNIQ-UHFFFAOYSA-N CCC(C)COC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CCC(C)COC1=CC=C(N2CCN(C)CC2)C=C1 CCBIPVWWCZLNIQ-UHFFFAOYSA-N 0.000 description 1
- UJTBZUNEAABHKT-UHFFFAOYSA-N CCC1=CC=C(C2=CC=C(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)C=C2)C=C1.CO(S)C1=CC=C(C2=CC=C(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)C=C2)C=C1 Chemical compound CCC1=CC=C(C2=CC=C(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)C=C2)C=C1.CO(S)C1=CC=C(C2=CC=C(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)C=C2)C=C1 UJTBZUNEAABHKT-UHFFFAOYSA-N 0.000 description 1
- GPOFSFLJOIAMSA-UHFFFAOYSA-N CCC1=CC=C(Cl)C=C1 Chemical compound CCC1=CC=C(Cl)C=C1 GPOFSFLJOIAMSA-UHFFFAOYSA-N 0.000 description 1
- JMFXPKAMLURHRQ-UHFFFAOYSA-N CCC1=CC=C(N2CCN(C)CC2)C=C1.O=C=O Chemical compound CCC1=CC=C(N2CCN(C)CC2)C=C1.O=C=O JMFXPKAMLURHRQ-UHFFFAOYSA-N 0.000 description 1
- GLJGLGNGUCOPBJ-UHFFFAOYSA-N CCC1=CC=C(N2CCNCC2)C=C1 Chemical compound CCC1=CC=C(N2CCNCC2)C=C1 GLJGLGNGUCOPBJ-UHFFFAOYSA-N 0.000 description 1
- YRNIBJGRLHLFJQ-UHFFFAOYSA-N CCCBr.CCCON=C1CCN(C(=O)OCC2=CC=CC=C2)CC1.CCCON=C1CCNCC1.NC1=NC2=C(C=NN2CCN2CCC(=NOCCCO)CC2)C2=NC(C3=CC=CO3)=NN12.NO.O=C(OCC1=CC=CC=C1)N1CCC(=NO)CC1.O=C1CCN(C(=O)OCC2=CC=CC=C2)CC1 Chemical compound CCCBr.CCCON=C1CCN(C(=O)OCC2=CC=CC=C2)CC1.CCCON=C1CCNCC1.NC1=NC2=C(C=NN2CCN2CCC(=NOCCCO)CC2)C2=NC(C3=CC=CO3)=NN12.NO.O=C(OCC1=CC=CC=C1)N1CCC(=NO)CC1.O=C1CCN(C(=O)OCC2=CC=CC=C2)CC1 YRNIBJGRLHLFJQ-UHFFFAOYSA-N 0.000 description 1
- DIXPHRIXQYGPLM-UHFFFAOYSA-N CCCN1CCC(C2=CC=CC=C2)CC1 Chemical compound CCCN1CCC(C2=CC=CC=C2)CC1 DIXPHRIXQYGPLM-UHFFFAOYSA-N 0.000 description 1
- JMWIXVQXBAXYHM-UHFFFAOYSA-N CCCN1CCN(C(=O)OCC(C)C)CC1 Chemical compound CCCN1CCN(C(=O)OCC(C)C)CC1 JMWIXVQXBAXYHM-UHFFFAOYSA-N 0.000 description 1
- NOXLNXOAHPURAN-UHFFFAOYSA-N CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CS3)N=C21 Chemical compound CCCN1N=CC2=C1N=C(N)N1N=C(C3=CC=CS3)N=C21 NOXLNXOAHPURAN-UHFFFAOYSA-N 0.000 description 1
- IXRHCCHYKDWCMV-UHFFFAOYSA-N CCCOC1=CC=C(Br)C=C1.CCCOC1=CC=C(C2(O)CCN(C(=O)OCC3=CC=CC=C3)CC2)C=C1.CCCOC1=CC=C(C2CCN(C(=O)OCC3=CC=CC=C3)CC2)C=C1.CCCOC1=CC=C(C2CCN(CCN3N=CC4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1.CCCOC1=CC=C(C2CCNCC2)C=C1.CC[SiH](CC)CC.O=C1CCN(C(=O)OCC2=CC=CC=C2)CC1 Chemical compound CCCOC1=CC=C(Br)C=C1.CCCOC1=CC=C(C2(O)CCN(C(=O)OCC3=CC=CC=C3)CC2)C=C1.CCCOC1=CC=C(C2CCN(C(=O)OCC3=CC=CC=C3)CC2)C=C1.CCCOC1=CC=C(C2CCN(CCN3N=CC4=C3N=C(N)N3N=C(C5=CC=CO5)N=C43)CC2)C=C1.CCCOC1=CC=C(C2CCNCC2)C=C1.CC[SiH](CC)CC.O=C1CCN(C(=O)OCC2=CC=CC=C2)CC1 IXRHCCHYKDWCMV-UHFFFAOYSA-N 0.000 description 1
- UUYNITVOLBGDIE-UHFFFAOYSA-N CCCOC1=CC=C(OCCN(C)C)C=C1 Chemical compound CCCOC1=CC=C(OCCN(C)C)C=C1 UUYNITVOLBGDIE-UHFFFAOYSA-N 0.000 description 1
- DOISVDHKTJDXNP-UHFFFAOYSA-N CCOC(=O)C(C)N1CCN(C2=C(F)C=C(F)C=C2)CC1.FC1=CC(F)=C(N2CCNCC2)C=C1 Chemical compound CCOC(=O)C(C)N1CCN(C2=C(F)C=C(F)C=C2)CC1.FC1=CC(F)=C(N2CCNCC2)C=C1 DOISVDHKTJDXNP-UHFFFAOYSA-N 0.000 description 1
- JCCLVVIHASIFOV-UHFFFAOYSA-N CCOC(=O)COC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CCOC(=O)COC1=CC=C(N2CCN(C)CC2)C=C1 JCCLVVIHASIFOV-UHFFFAOYSA-N 0.000 description 1
- UGHKLXGHPHYGTP-UHFFFAOYSA-N CCOC(C(C)N(CC1)CCN1c(ccc(F)c1)c1F)=O Chemical compound CCOC(C(C)N(CC1)CCN1c(ccc(F)c1)c1F)=O UGHKLXGHPHYGTP-UHFFFAOYSA-N 0.000 description 1
- FBSRPRKDTZWEEP-UHFFFAOYSA-N CCOCCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CCOCCOC1=CC=C(N2CCN(C)CC2)C=C1 FBSRPRKDTZWEEP-UHFFFAOYSA-N 0.000 description 1
- RDKKQZIFDSEMNU-UHFFFAOYSA-N CCS(=O)(=O)C(C)C Chemical compound CCS(=O)(=O)C(C)C RDKKQZIFDSEMNU-UHFFFAOYSA-N 0.000 description 1
- IOVMMTOWSNYJGF-UHFFFAOYSA-H CI.I[V](I)I.I[V]I.NC1=NC(Cl)=C(C=O)C(Cl)=N1.NC1=NC(NNC(=O)C2=CC=CO2)=C(C=O)C(Cl)=N1.NC1=NC(O)=CC(O)=N1.NC1=NC2=C(C=NN2)C(NNC(=O)C2=CC=CO2)=N1.NC1=NC2=C(C=NN2)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(C=NN2)C2=NC(C3=CC=CO3)=NN12.NNC(=O)C1=CC=CO1.[V]I Chemical compound CI.I[V](I)I.I[V]I.NC1=NC(Cl)=C(C=O)C(Cl)=N1.NC1=NC(NNC(=O)C2=CC=CO2)=C(C=O)C(Cl)=N1.NC1=NC(O)=CC(O)=N1.NC1=NC2=C(C=NN2)C(NNC(=O)C2=CC=CO2)=N1.NC1=NC2=C(C=NN2)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(C=NN2)C2=NC(C3=CC=CO3)=NN12.NNC(=O)C1=CC=CO1.[V]I IOVMMTOWSNYJGF-UHFFFAOYSA-H 0.000 description 1
- ITEJGZLELLEVHZ-UHFFFAOYSA-N CN(C)CCCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CN(C)CCCOC1=CC=C(N2CCN(C)CC2)C=C1 ITEJGZLELLEVHZ-UHFFFAOYSA-N 0.000 description 1
- JKHMRNFBTNUREF-UHFFFAOYSA-N CN(C)CCN(C)C1=C(F)C=C(F)C=C1 Chemical compound CN(C)CCN(C)C1=C(F)C=C(F)C=C1 JKHMRNFBTNUREF-UHFFFAOYSA-N 0.000 description 1
- KUNDZNSXYFPGGA-UHFFFAOYSA-N CN(C)CCOC1=CC=C(F)C=C1 Chemical compound CN(C)CCOC1=CC=C(F)C=C1 KUNDZNSXYFPGGA-UHFFFAOYSA-N 0.000 description 1
- KWLQMKSDDPCNGQ-UHFFFAOYSA-N CN(C)CCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CN(C)CCOC1=CC=C(N2CCN(C)CC2)C=C1 KWLQMKSDDPCNGQ-UHFFFAOYSA-N 0.000 description 1
- OVJZZOWARFLESL-UHFFFAOYSA-N CN(CC1)CCN1c(cc1)cc(OCCOC)c1OCCOC Chemical compound CN(CC1)CCN1c(cc1)cc(OCCOC)c1OCCOC OVJZZOWARFLESL-UHFFFAOYSA-N 0.000 description 1
- SUERPFBWBVPLSD-UHFFFAOYSA-N CN(CC1)CCN1c(cc1F)cc(F)c1OCCOC Chemical compound CN(CC1)CCN1c(cc1F)cc(F)c1OCCOC SUERPFBWBVPLSD-UHFFFAOYSA-N 0.000 description 1
- KEFVKKQOIQWELT-UHFFFAOYSA-N CN(CC1)CCN1c(ccc(OC)c1)c1OC Chemical compound CN(CC1)CCN1c(ccc(OC)c1)c1OC KEFVKKQOIQWELT-UHFFFAOYSA-N 0.000 description 1
- OYDKJVNOHPBOOD-UHFFFAOYSA-N CN(CCN(C)C1=C(F)C=C(F)C=C1)CCN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 Chemical compound CN(CCN(C)C1=C(F)C=C(F)C=C1)CCN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 OYDKJVNOHPBOOD-UHFFFAOYSA-N 0.000 description 1
- NEPOYLVMRQRLAT-UHFFFAOYSA-N CN1CC=C(C2=CC=C(Cl)C=C2)CC1 Chemical compound CN1CC=C(C2=CC=C(Cl)C=C2)CC1 NEPOYLVMRQRLAT-UHFFFAOYSA-N 0.000 description 1
- NSCCSVFQPMGASE-UHFFFAOYSA-N CN1CCC(=CC2=CC=CC=C2)CC1 Chemical compound CN1CCC(=CC2=CC=CC=C2)CC1 NSCCSVFQPMGASE-UHFFFAOYSA-N 0.000 description 1
- XVFDTMOEUDZZFE-UHFFFAOYSA-N CN1CCC(C2=CC=C(Br)C=C2)CC1 Chemical compound CN1CCC(C2=CC=C(Br)C=C2)CC1 XVFDTMOEUDZZFE-UHFFFAOYSA-N 0.000 description 1
- QLAQDAALXYBLIF-UHFFFAOYSA-N CN1CCC(CC2=CC=CC=C2)CC1 Chemical compound CN1CCC(CC2=CC=CC=C2)CC1 QLAQDAALXYBLIF-UHFFFAOYSA-N 0.000 description 1
- KWKKMAWHMMLZRR-UHFFFAOYSA-N CN1CCC(N2C(=O)NC3=C2C=CC=C3)CC1 Chemical compound CN1CCC(N2C(=O)NC3=C2C=CC=C3)CC1 KWKKMAWHMMLZRR-UHFFFAOYSA-N 0.000 description 1
- GQPLQVMAOCKIHC-UHFFFAOYSA-N CN1CCC(O)(C2=CC=C(Br)C=C2)CC1 Chemical compound CN1CCC(O)(C2=CC=C(Br)C=C2)CC1 GQPLQVMAOCKIHC-UHFFFAOYSA-N 0.000 description 1
- WUFINGBTODUAJH-UHFFFAOYSA-N CN1CCC(O)(C2=CC=C(Cl)C=C2)CC1 Chemical compound CN1CCC(O)(C2=CC=C(Cl)C=C2)CC1 WUFINGBTODUAJH-UHFFFAOYSA-N 0.000 description 1
- JVMCZMQYJIRKRV-UHFFFAOYSA-N CN1CCC(O)(C2=CC=CC=C2)CC1 Chemical compound CN1CCC(O)(C2=CC=CC=C2)CC1 JVMCZMQYJIRKRV-UHFFFAOYSA-N 0.000 description 1
- BPXNJWOQVQXJQM-UHFFFAOYSA-N CN1CCC2(CC1)C(=O)NCN2C1=CC=CC=C1 Chemical compound CN1CCC2(CC1)C(=O)NCN2C1=CC=CC=C1 BPXNJWOQVQXJQM-UHFFFAOYSA-N 0.000 description 1
- KLZKXQWYCOKWQV-UHFFFAOYSA-N CN1CCC2(CC1)OC1=C(C=CC=C1)O2 Chemical compound CN1CCC2(CC1)OC1=C(C=CC=C1)O2 KLZKXQWYCOKWQV-UHFFFAOYSA-N 0.000 description 1
- JOFKCNJIUXPJAC-UHFFFAOYSA-N CN1CCC2=C(C1)NC1=C2C=CC=C1 Chemical compound CN1CCC2=C(C1)NC1=C2C=CC=C1 JOFKCNJIUXPJAC-UHFFFAOYSA-N 0.000 description 1
- WZLZYEIVITXJMC-UHFFFAOYSA-N CN1CCC2=C(C=C(CO)C(CO)=C2)C1 Chemical compound CN1CCC2=C(C=C(CO)C(CO)=C2)C1 WZLZYEIVITXJMC-UHFFFAOYSA-N 0.000 description 1
- KYXSVGVQGFPNRQ-UHFFFAOYSA-N CN1CCC2=C(C=CC=C2)C1 Chemical compound CN1CCC2=C(C=CC=C2)C1 KYXSVGVQGFPNRQ-UHFFFAOYSA-N 0.000 description 1
- AVIAGAGHRPKXCE-UHFFFAOYSA-N CN1CCCC(C2=CC=CC=C2)C1 Chemical compound CN1CCCC(C2=CC=CC=C2)C1 AVIAGAGHRPKXCE-UHFFFAOYSA-N 0.000 description 1
- MMELGIDHFSGOQC-UHFFFAOYSA-N CN1CCCN(C2=CC=C(Cl)C=C2)CC1 Chemical compound CN1CCCN(C2=CC=C(Cl)C=C2)CC1 MMELGIDHFSGOQC-UHFFFAOYSA-N 0.000 description 1
- CFHQBPRIZYWRAP-UHFFFAOYSA-N CN1CCCN(C2=CC=C(F)C=C2F)CC1 Chemical compound CN1CCCN(C2=CC=C(F)C=C2F)CC1 CFHQBPRIZYWRAP-UHFFFAOYSA-N 0.000 description 1
- SQMOMJVTVKHHBP-UHFFFAOYSA-N CN1CCCN(C2=CC=CC=C2)CC1 Chemical compound CN1CCCN(C2=CC=CC=C2)CC1 SQMOMJVTVKHHBP-UHFFFAOYSA-N 0.000 description 1
- UVKZSORBKUEBAZ-UHFFFAOYSA-N CN1CCN(C(C2=CC=CC=C2)C2=CC=CC=C2)CC1 Chemical compound CN1CCN(C(C2=CC=CC=C2)C2=CC=CC=C2)CC1 UVKZSORBKUEBAZ-UHFFFAOYSA-N 0.000 description 1
- GKUROHOCYZVBSB-UHFFFAOYSA-N CN1CCN(C2=C(F)C(F)=C(F)C(F)=C2F)CC1 Chemical compound CN1CCN(C2=C(F)C(F)=C(F)C(F)=C2F)CC1 GKUROHOCYZVBSB-UHFFFAOYSA-N 0.000 description 1
- ZKCFOLRMUUCRDT-UHFFFAOYSA-N CN1CCN(C2=C(F)C(F)=CC(F)=C2F)CC1 Chemical compound CN1CCN(C2=C(F)C(F)=CC(F)=C2F)CC1 ZKCFOLRMUUCRDT-UHFFFAOYSA-N 0.000 description 1
- YUVWEDDONAOSNB-UHFFFAOYSA-N CN1CCN(C2=C(F)C=C(CO)C=C2F)CC1 Chemical compound CN1CCN(C2=C(F)C=C(CO)C=C2F)CC1 YUVWEDDONAOSNB-UHFFFAOYSA-N 0.000 description 1
- HNPTYKJOBMTFOH-UHFFFAOYSA-N CN1CCN(C2=C(F)C=C(F)C(F)=C2)CC1 Chemical compound CN1CCN(C2=C(F)C=C(F)C(F)=C2)CC1 HNPTYKJOBMTFOH-UHFFFAOYSA-N 0.000 description 1
- WSUPEKDOBXLJFI-UHFFFAOYSA-N CN1CCN(C2=CC(CO)=CC(CO)=C2)CC1 Chemical compound CN1CCN(C2=CC(CO)=CC(CO)=C2)CC1 WSUPEKDOBXLJFI-UHFFFAOYSA-N 0.000 description 1
- FZWKQIVDNYXGGM-UHFFFAOYSA-N CN1CCN(C2=CC(Cl)=C(CO)C(Cl)=C2)CC1 Chemical compound CN1CCN(C2=CC(Cl)=C(CO)C(Cl)=C2)CC1 FZWKQIVDNYXGGM-UHFFFAOYSA-N 0.000 description 1
- OALCTIDCHHZYPW-UHFFFAOYSA-N CN1CCN(C2=CC(Cl)=CC(Cl)=C2)CC1 Chemical compound CN1CCN(C2=CC(Cl)=CC(Cl)=C2)CC1 OALCTIDCHHZYPW-UHFFFAOYSA-N 0.000 description 1
- CMLSPBYWAXLVML-UHFFFAOYSA-N CN1CCN(C2=CC(F)=C(OCCCO)C(F)=C2)CC1 Chemical compound CN1CCN(C2=CC(F)=C(OCCCO)C(F)=C2)CC1 CMLSPBYWAXLVML-UHFFFAOYSA-N 0.000 description 1
- WCOODAWUEIXTFL-UHFFFAOYSA-N CN1CCN(C2=CC=C(Br)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(Br)C=C2)CC1 WCOODAWUEIXTFL-UHFFFAOYSA-N 0.000 description 1
- VRCQAHOGNLTKGX-UHFFFAOYSA-N CN1CCN(C2=CC=C(CO)C(F)=C2)CC1 Chemical compound CN1CCN(C2=CC=C(CO)C(F)=C2)CC1 VRCQAHOGNLTKGX-UHFFFAOYSA-N 0.000 description 1
- IYINOTGHUDYERV-UHFFFAOYSA-N CN1CCN(C2=CC=C(CO)C=C2C(F)(F)F)CC1 Chemical compound CN1CCN(C2=CC=C(CO)C=C2C(F)(F)F)CC1 IYINOTGHUDYERV-UHFFFAOYSA-N 0.000 description 1
- ZASYEYNRJJRDFF-UHFFFAOYSA-N CN1CCN(C2=CC=C(CO)C=C2F)CC1 Chemical compound CN1CCN(C2=CC=C(CO)C=C2F)CC1 ZASYEYNRJJRDFF-UHFFFAOYSA-N 0.000 description 1
- FWZPCDQEUXAZDW-UHFFFAOYSA-N CN1CCN(C2=CC=C(Cl)C(Cl)=C2)CC1 Chemical compound CN1CCN(C2=CC=C(Cl)C(Cl)=C2)CC1 FWZPCDQEUXAZDW-UHFFFAOYSA-N 0.000 description 1
- QSWNKAMBKWBJGH-UHFFFAOYSA-N CN1CCN(C2=CC=C(Cl)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(Cl)C=C2)CC1 QSWNKAMBKWBJGH-UHFFFAOYSA-N 0.000 description 1
- ZIUYCCYKVUGEIO-UHFFFAOYSA-N CN1CCN(C2=CC=C(Cl)C=C2Cl)CC1 Chemical compound CN1CCN(C2=CC=C(Cl)C=C2Cl)CC1 ZIUYCCYKVUGEIO-UHFFFAOYSA-N 0.000 description 1
- UCHGKPJQKWSUKE-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C(F)=C2)CC1 Chemical compound CN1CCN(C2=CC=C(F)C(F)=C2)CC1 UCHGKPJQKWSUKE-UHFFFAOYSA-N 0.000 description 1
- QAUXNRCASQNZNF-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C(F)=C2F)CC1 Chemical compound CN1CCN(C2=CC=C(F)C(F)=C2F)CC1 QAUXNRCASQNZNF-UHFFFAOYSA-N 0.000 description 1
- QEPUUNVKXDUNHZ-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C=C2C#N)CC1 Chemical compound CN1CCN(C2=CC=C(F)C=C2C#N)CC1 QEPUUNVKXDUNHZ-UHFFFAOYSA-N 0.000 description 1
- IFZHXXLFXSKLQV-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C=C2OCCCO)CC1 Chemical compound CN1CCN(C2=CC=C(F)C=C2OCCCO)CC1 IFZHXXLFXSKLQV-UHFFFAOYSA-N 0.000 description 1
- SVXUJOMAWXJSIK-UHFFFAOYSA-N CN1CCN(C2=CC=C(F)C=C2[N+](=O)[O-])CC1 Chemical compound CN1CCN(C2=CC=C(F)C=C2[N+](=O)[O-])CC1 SVXUJOMAWXJSIK-UHFFFAOYSA-N 0.000 description 1
- FSSMBBBSMDSRKC-UHFFFAOYSA-N CN1CCN(C2=CC=C(N3CCOCC3)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(N3CCOCC3)C=C2)CC1 FSSMBBBSMDSRKC-UHFFFAOYSA-N 0.000 description 1
- NNVDRRXXIJFDNT-UHFFFAOYSA-N CN1CCN(C2=CC=C(N[SH](=O)=O)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(N[SH](=O)=O)C=C2)CC1 NNVDRRXXIJFDNT-UHFFFAOYSA-N 0.000 description 1
- RWDJMTACZOUIAI-UHFFFAOYSA-N CN1CCN(C2=CC=C(O)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(O)C=C2)CC1 RWDJMTACZOUIAI-UHFFFAOYSA-N 0.000 description 1
- AGXMMEQEAURFNK-UHFFFAOYSA-N CN1CCN(C2=CC=C(OC3CCOCC3)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OC3CCOCC3)C=C2)CC1 AGXMMEQEAURFNK-UHFFFAOYSA-N 0.000 description 1
- LIHUFITYGIANJY-UHFFFAOYSA-N CN1CCN(C2=CC=C(OCC(=O)O)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OCC(=O)O)C=C2)CC1 LIHUFITYGIANJY-UHFFFAOYSA-N 0.000 description 1
- NEHANQXPAVDHOS-UHFFFAOYSA-N CN1CCN(C2=CC=C(OCC3(C)OCCO3)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OCC3(C)OCCO3)C=C2)CC1 NEHANQXPAVDHOS-UHFFFAOYSA-N 0.000 description 1
- OVVWWFHRBFJMNT-UHFFFAOYSA-N CN1CCN(C2=CC=C(OCC3CC3)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OCC3CC3)C=C2)CC1 OVVWWFHRBFJMNT-UHFFFAOYSA-N 0.000 description 1
- CAYQCDIYYKANMS-UHFFFAOYSA-N CN1CCN(C2=CC=C(OCCO)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OCCO)C=C2)CC1 CAYQCDIYYKANMS-UHFFFAOYSA-N 0.000 description 1
- AIWVSJBUWYAUKR-UHFFFAOYSA-N CN1CCN(C2=CC=C(OCCOCC(F)(F)F)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OCCOCC(F)(F)F)C=C2)CC1 AIWVSJBUWYAUKR-UHFFFAOYSA-N 0.000 description 1
- GSEZEJZTRBBKSM-UHFFFAOYSA-N CN1CCN(C2=CC=C(OCO(C)S)C=C2)CC1 Chemical compound CN1CCN(C2=CC=C(OCO(C)S)C=C2)CC1 GSEZEJZTRBBKSM-UHFFFAOYSA-N 0.000 description 1
- GZNDUKANJZIZOT-UHFFFAOYSA-N CN1CCN(C2=CC=C([N+](=O)[O-])C=C2)CC1 Chemical compound CN1CCN(C2=CC=C([N+](=O)[O-])C=C2)CC1 GZNDUKANJZIZOT-UHFFFAOYSA-N 0.000 description 1
- HOMWNUXPSJQSSU-UHFFFAOYSA-N CN1CCN(C2=CC=C3C=CC=CC3=N2)CC1 Chemical compound CN1CCN(C2=CC=C3C=CC=CC3=N2)CC1 HOMWNUXPSJQSSU-UHFFFAOYSA-N 0.000 description 1
- FRVFBEITXORTGZ-UHFFFAOYSA-N CN1CCN(C2=CC=C3OC(F)(F)C(F)(F)OC3=C2)CC1 Chemical compound CN1CCN(C2=CC=C3OC(F)(F)C(F)(F)OC3=C2)CC1 FRVFBEITXORTGZ-UHFFFAOYSA-N 0.000 description 1
- MXJILXQFHAGJLH-UHFFFAOYSA-N CN1CCN(C2=CC=C3OC(F)(F)OC3=C2)CC1 Chemical compound CN1CCN(C2=CC=C3OC(F)(F)OC3=C2)CC1 MXJILXQFHAGJLH-UHFFFAOYSA-N 0.000 description 1
- CPPMRWSPQKRKKV-UHFFFAOYSA-N CN1CCN(C2=CC=C3OCOC3=C2)CC1 Chemical compound CN1CCN(C2=CC=C3OCOC3=C2)CC1 CPPMRWSPQKRKKV-UHFFFAOYSA-N 0.000 description 1
- AISHPJPGKBKMGV-UHFFFAOYSA-N CN1CCN(C2=CC=CC(O)=C2)CC1 Chemical compound CN1CCN(C2=CC=CC(O)=C2)CC1 AISHPJPGKBKMGV-UHFFFAOYSA-N 0.000 description 1
- PZNQMJBVNDWXDY-UHFFFAOYSA-N CN1CCN(C2=CC=CC=C2)C(=O)C1 Chemical compound CN1CCN(C2=CC=CC=C2)C(=O)C1 PZNQMJBVNDWXDY-UHFFFAOYSA-N 0.000 description 1
- UXUQHCICMDURET-UHFFFAOYSA-N CN1CCN(C2=CC=CC=C2F)CC1 Chemical compound CN1CCN(C2=CC=CC=C2F)CC1 UXUQHCICMDURET-UHFFFAOYSA-N 0.000 description 1
- BREKGVCIMLEZSM-UHFFFAOYSA-N CN1CCN(C2=CC=CC=C2O)CC1 Chemical compound CN1CCN(C2=CC=CC=C2O)CC1 BREKGVCIMLEZSM-UHFFFAOYSA-N 0.000 description 1
- MDHKWAZLVNUABG-UHFFFAOYSA-N CN1CCN(C2=CC=CC=N2)CC1 Chemical compound CN1CCN(C2=CC=CC=N2)CC1 MDHKWAZLVNUABG-UHFFFAOYSA-N 0.000 description 1
- ZWWAKJKAKJGLAU-UHFFFAOYSA-N CN1CCN(C2=CC=CC=N2O)CC1 Chemical compound CN1CCN(C2=CC=CC=N2O)CC1 ZWWAKJKAKJGLAU-UHFFFAOYSA-N 0.000 description 1
- HBWGDGHGYGHWKD-UHFFFAOYSA-N CN1CCN(C2=CC=CN=C2)CC1 Chemical compound CN1CCN(C2=CC=CN=C2)CC1 HBWGDGHGYGHWKD-UHFFFAOYSA-N 0.000 description 1
- NGHHVCGBOOCIBY-UHFFFAOYSA-N CN1CCN(C2=CC=NC=C2)CC1 Chemical compound CN1CCN(C2=CC=NC=C2)CC1 NGHHVCGBOOCIBY-UHFFFAOYSA-N 0.000 description 1
- ZYNWTNXITLBLND-UHFFFAOYSA-N CN1CCN(C2=NC(CO)=CC(CO)=N2)CC1 Chemical compound CN1CCN(C2=NC(CO)=CC(CO)=N2)CC1 ZYNWTNXITLBLND-UHFFFAOYSA-N 0.000 description 1
- RBHNSRTVBDSNLG-UHFFFAOYSA-N CN1CCN(C2=NC(N)=NC(N)=C2)CC1 Chemical compound CN1CCN(C2=NC(N)=NC(N)=C2)CC1 RBHNSRTVBDSNLG-UHFFFAOYSA-N 0.000 description 1
- SWGOGZZKIMYIKE-UHFFFAOYSA-N CN1CCN(C2=NC3=C(C=CC=C3)N2)CC1 Chemical compound CN1CCN(C2=NC3=C(C=CC=C3)N2)CC1 SWGOGZZKIMYIKE-UHFFFAOYSA-N 0.000 description 1
- UXTXSBTWTYIBSJ-UHFFFAOYSA-N CN1CCN(C2=NC=C(Cl)C=C2C(F)(F)F)CC1 Chemical compound CN1CCN(C2=NC=C(Cl)C=C2C(F)(F)F)CC1 UXTXSBTWTYIBSJ-UHFFFAOYSA-N 0.000 description 1
- HOYSODSNWMPVMO-UHFFFAOYSA-N CN1CCN(C2=NC=CC=C2C(F)(F)F)CC1 Chemical compound CN1CCN(C2=NC=CC=C2C(F)(F)F)CC1 HOYSODSNWMPVMO-UHFFFAOYSA-N 0.000 description 1
- MYWNBGPUPDPECZ-UHFFFAOYSA-N CN1CCN(C2=NC=CC=N2)CC1 Chemical compound CN1CCN(C2=NC=CC=N2)CC1 MYWNBGPUPDPECZ-UHFFFAOYSA-N 0.000 description 1
- OSPDKPFPBGJRTD-UHFFFAOYSA-N CN1CCN(C2=NC=CS2)CC1 Chemical compound CN1CCN(C2=NC=CS2)CC1 OSPDKPFPBGJRTD-UHFFFAOYSA-N 0.000 description 1
- MLJOKPBESJWYGL-UHFFFAOYSA-N CN1CCN(CC2=CC=CC=C2)CC1 Chemical compound CN1CCN(CC2=CC=CC=C2)CC1 MLJOKPBESJWYGL-UHFFFAOYSA-N 0.000 description 1
- RBAWHTJNTFMIQI-UHFFFAOYSA-N CN1CCN(CCCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1 Chemical compound CN1CCN(CCCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1 RBAWHTJNTFMIQI-UHFFFAOYSA-N 0.000 description 1
- ZJPUCYGAUIAZLX-UHFFFAOYSA-N CN1CCN(CCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CS4)N=C32)CC1 Chemical compound CN1CCN(CCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CS4)N=C32)CC1 ZJPUCYGAUIAZLX-UHFFFAOYSA-N 0.000 description 1
- VNEIJQSFJLJASN-UHFFFAOYSA-N CN=C(C1=CC=CC=C1)C1CCN(CCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1 Chemical compound CN=C(C1=CC=CC=C1)C1CCN(CCN2/N=C\C3=C2N=C(N)N2N=C(C4=CC=CO4)N=C32)CC1 VNEIJQSFJLJASN-UHFFFAOYSA-N 0.000 description 1
- QJNGEXCJXPQCPA-UHFFFAOYSA-N CNC1CC1C1=CC=CC=C1 Chemical compound CNC1CC1C1=CC=CC=C1 QJNGEXCJXPQCPA-UHFFFAOYSA-N 0.000 description 1
- VHTHWUSPHFHGRV-UHFFFAOYSA-N CNCC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound CNCC1=CC=C(N2CCN(C)CC2)C=C1 VHTHWUSPHFHGRV-UHFFFAOYSA-N 0.000 description 1
- KHHBREZONKWQMA-UHFFFAOYSA-N CNCCC(=O)C(C)C Chemical compound CNCCC(=O)C(C)C KHHBREZONKWQMA-UHFFFAOYSA-N 0.000 description 1
- ZTXWGLLWOCJFOW-UHFFFAOYSA-N CNCCC1=CC=C(C)C=C1 Chemical compound CNCCC1=CC=C(C)C=C1 ZTXWGLLWOCJFOW-UHFFFAOYSA-N 0.000 description 1
- NCBPDSPIVAMJIT-UHFFFAOYSA-N CNCCC1=CC=C(F)C=C1 Chemical compound CNCCC1=CC=C(F)C=C1 NCBPDSPIVAMJIT-UHFFFAOYSA-N 0.000 description 1
- COCFQAQFBIRRKQ-UHFFFAOYSA-N CNCCNC1=CC=CC=C1 Chemical compound CNCCNC1=CC=CC=C1 COCFQAQFBIRRKQ-UHFFFAOYSA-N 0.000 description 1
- JCMWSVNNSPUNER-UHFFFAOYSA-N CNCCc(cc1)ccc1OC Chemical compound CNCCc(cc1)ccc1OC JCMWSVNNSPUNER-UHFFFAOYSA-N 0.000 description 1
- XXBCQCLNZPCHTA-UHFFFAOYSA-N COC1=CC(CO)=CC=C1N1CCN(C)CC1 Chemical compound COC1=CC(CO)=CC=C1N1CCN(C)CC1 XXBCQCLNZPCHTA-UHFFFAOYSA-N 0.000 description 1
- WRUANJSFFXGIMI-UHFFFAOYSA-N COC1=CC(F)=CC(F)=C1N1CCN(C)CC1 Chemical compound COC1=CC(F)=CC(F)=C1N1CCN(C)CC1 WRUANJSFFXGIMI-UHFFFAOYSA-N 0.000 description 1
- NQRVUICGFPTYLU-UHFFFAOYSA-N COC1=CC(F)=CC=C1N1CCN(C)CC1 Chemical compound COC1=CC(F)=CC=C1N1CCN(C)CC1 NQRVUICGFPTYLU-UHFFFAOYSA-N 0.000 description 1
- CHLICZRVGGXEOD-UHFFFAOYSA-N COC1=CC=C(C)C=C1 Chemical compound COC1=CC=C(C)C=C1 CHLICZRVGGXEOD-UHFFFAOYSA-N 0.000 description 1
- ZXRWWRCQFMIXCC-UHFFFAOYSA-N COC1=CC=C(Cl)C=C1N1CCN(C)CC1 Chemical compound COC1=CC=C(Cl)C=C1N1CCN(C)CC1 ZXRWWRCQFMIXCC-UHFFFAOYSA-N 0.000 description 1
- ZYXMVSPWOATGIW-UHFFFAOYSA-N COC1=CC=CC=C1N1CCN(C)CC1 Chemical compound COC1=CC=CC=C1N1CCN(C)CC1 ZYXMVSPWOATGIW-UHFFFAOYSA-N 0.000 description 1
- JQWXIRSUGSYCER-UHFFFAOYSA-N COCC(COC)OC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound COCC(COC)OC1=CC=C(N2CCN(C)CC2)C=C1 JQWXIRSUGSYCER-UHFFFAOYSA-N 0.000 description 1
- NXXPBQLDAVNKST-UHFFFAOYSA-N COCCCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound COCCCOC1=CC=C(N2CCN(C)CC2)C=C1 NXXPBQLDAVNKST-UHFFFAOYSA-N 0.000 description 1
- CIJMDNDHBGACBQ-UHFFFAOYSA-N COCCOC1=C(C)C=C(N2CCN(C)CC2)C=C1 Chemical compound COCCOC1=C(C)C=C(N2CCN(C)CC2)C=C1 CIJMDNDHBGACBQ-UHFFFAOYSA-N 0.000 description 1
- BNQSBIMOUYXCBN-UHFFFAOYSA-N COCCOC1=C(CO)C=C(N2CCN(C)CC2)C=C1 Chemical compound COCCOC1=C(CO)C=C(N2CCN(C)CC2)C=C1 BNQSBIMOUYXCBN-UHFFFAOYSA-N 0.000 description 1
- VZRMGCJJCUGLKT-UHFFFAOYSA-N COCCOC1=C(OCCCO)C=C(N2CCN(C)CC2)C=C1 Chemical compound COCCOC1=C(OCCCO)C=C(N2CCN(C)CC2)C=C1 VZRMGCJJCUGLKT-UHFFFAOYSA-N 0.000 description 1
- WJAJIPFRBOFQSR-UHFFFAOYSA-N COCCOC1=CC(N2CCN(C)CC2)=CC=C1F Chemical compound COCCOC1=CC(N2CCN(C)CC2)=CC=C1F WJAJIPFRBOFQSR-UHFFFAOYSA-N 0.000 description 1
- DOSUSQYYROSQFJ-UHFFFAOYSA-N COCCOC1=CC=C(C)C=C1 Chemical compound COCCOC1=CC=C(C)C=C1 DOSUSQYYROSQFJ-UHFFFAOYSA-N 0.000 description 1
- CJBIRVWVPBMUEX-UHFFFAOYSA-N COCCOC1=CC=C(N(C)CCN(C)C)C(C(F)(F)F)=C1 Chemical compound COCCOC1=CC=C(N(C)CCN(C)C)C(C(F)(F)F)=C1 CJBIRVWVPBMUEX-UHFFFAOYSA-N 0.000 description 1
- WRQZMRFJISFGOT-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(C)CC2)C(C#N)=C1 Chemical compound COCCOC1=CC=C(N2CCN(C)CC2)C(C#N)=C1 WRQZMRFJISFGOT-UHFFFAOYSA-N 0.000 description 1
- XECBZBFAAHAJKR-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(C)CC2)C(C(F)(F)F)=C1 Chemical compound COCCOC1=CC=C(N2CCN(C)CC2)C(C(F)(F)F)=C1 XECBZBFAAHAJKR-UHFFFAOYSA-N 0.000 description 1
- OFKCQJGACALWFE-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(C)CC2)C(C)=C1 Chemical compound COCCOC1=CC=C(N2CCN(C)CC2)C(C)=C1 OFKCQJGACALWFE-UHFFFAOYSA-N 0.000 description 1
- VPELEDWLGKZGIH-UHFFFAOYSA-N COCCOC1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=C(Cl)O5)N=C43)CC2)C=C1 Chemical compound COCCOC1=CC=C(N2CCN(CCN3/N=C\C4=C3N=C(N)N3N=C(C5=CC=C(Cl)O5)N=C43)CC2)C=C1 VPELEDWLGKZGIH-UHFFFAOYSA-N 0.000 description 1
- NXYKLZYQTVEEDP-UHFFFAOYSA-N COCCOCCOC1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound COCCOCCOC1=CC=C(N2CCN(C)CC2)C=C1 NXYKLZYQTVEEDP-UHFFFAOYSA-N 0.000 description 1
- UAPUGEYVGDNUJY-UHFFFAOYSA-N COc(cc1)ccc1C(N1CCN(CC[n]2ncc3c2nc(N)[n]2nc(-c4ccc[o]4)nc32)CC1)=O Chemical compound COc(cc1)ccc1C(N1CCN(CC[n]2ncc3c2nc(N)[n]2nc(-c4ccc[o]4)nc32)CC1)=O UAPUGEYVGDNUJY-UHFFFAOYSA-N 0.000 description 1
- NNOMSSYYMNKVBN-UHFFFAOYSA-N CS(=O)(=O)Cl.CS(=O)(=O)OCCC1=CC=C(C2=CC=CN=C2)C=C1.NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CC=CN=C3)C=C2)C2=NC(C3=CC=CO3)=NN12.OB(O)C1=CC=CN=C1.OCCC1=CC=C(Br)C=C1.OCCC1=CC=C(C2=CC=CN=C2)C=C1 Chemical compound CS(=O)(=O)Cl.CS(=O)(=O)OCCC1=CC=C(C2=CC=CN=C2)C=C1.NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CC=CN=C3)C=C2)C2=NC(C3=CC=CO3)=NN12.OB(O)C1=CC=CN=C1.OCCC1=CC=C(Br)C=C1.OCCC1=CC=C(C2=CC=CN=C2)C=C1 NNOMSSYYMNKVBN-UHFFFAOYSA-N 0.000 description 1
- ZIYVDZWCLFXMTO-UHFFFAOYSA-N CSCCN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 Chemical compound CSCCN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 ZIYVDZWCLFXMTO-UHFFFAOYSA-N 0.000 description 1
- IRQAVHKOEKPMFB-VIFPVBQESA-N C[C@H]1CN(C(=O)OC(C)(C)C)CCN1C Chemical compound C[C@H]1CN(C(=O)OC(C)(C)C)CCN1C IRQAVHKOEKPMFB-VIFPVBQESA-N 0.000 description 1
- AMGYTXUAOWWFGV-UHFFFAOYSA-N C[Y]CCN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 Chemical compound C[Y]CCN1/N=C\C2=C1N=C(N)N1N=C(C3=CC=CO3)N=C21 AMGYTXUAOWWFGV-UHFFFAOYSA-N 0.000 description 1
- NTIAVUBMOJPJPM-UHFFFAOYSA-N ClCCN1CCN(C2=CC=CC=C2)CC1 Chemical compound ClCCN1CCN(C2=CC=CC=C2)CC1 NTIAVUBMOJPJPM-UHFFFAOYSA-N 0.000 description 1
- GFJPJNYUQDUARF-UHFFFAOYSA-N FC1=CC(F)=C(N2CCN(CCCl)CC2)C=C1.N=N.NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C(Cl)=N1.NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C(NNC(=O)C2=CC=CS2)=N1.NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CS3)=NN12.NC1=NC2=C(C=NN2)C(Cl)=N1.NNC1=NC(N)=NC2=C1/C=N\N2CCN1CCN(C2=C(F)C=C(F)C=C2)CC1.O=C(Cl)C1=CC=CS1.[HH] Chemical compound FC1=CC(F)=C(N2CCN(CCCl)CC2)C=C1.N=N.NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C(Cl)=N1.NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C(NNC(=O)C2=CC=CS2)=N1.NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CS3)=NN12.NC1=NC2=C(C=NN2)C(Cl)=N1.NNC1=NC(N)=NC2=C1/C=N\N2CCN1CCN(C2=C(F)C=C(F)C=C2)CC1.O=C(Cl)C1=CC=CS1.[HH] GFJPJNYUQDUARF-UHFFFAOYSA-N 0.000 description 1
- CMCSPBOWEYUGHB-UHFFFAOYSA-N Fc(cc1F)ccc1N1CCNCC1 Chemical compound Fc(cc1F)ccc1N1CCNCC1 CMCSPBOWEYUGHB-UHFFFAOYSA-N 0.000 description 1
- GOJUJUVQIVIZAV-UHFFFAOYSA-N NC1=NC(Cl)=C(C=O)C(Cl)=N1 Chemical compound NC1=NC(Cl)=C(C=O)C(Cl)=N1 GOJUJUVQIVIZAV-UHFFFAOYSA-N 0.000 description 1
- UPMCIBCGIMRVES-UHFFFAOYSA-N NC1=NC(NNC(=O)C2=CC=CO2)=C(C=O)C(Cl)=N1.NC1=NC2=C(C=NN2CCBr)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(C=NN2CCO)C(NNC(=O)C2=CC=CO2)=N1.NC1=NC2=C(C=NN2CCO)C2=NC(C3=CC=CO3)=NN12.NNCCO Chemical compound NC1=NC(NNC(=O)C2=CC=CO2)=C(C=O)C(Cl)=N1.NC1=NC2=C(C=NN2CCBr)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(C=NN2CCO)C(NNC(=O)C2=CC=CO2)=N1.NC1=NC2=C(C=NN2CCO)C2=NC(C3=CC=CO3)=NN12.NNCCO UPMCIBCGIMRVES-UHFFFAOYSA-N 0.000 description 1
- VBGGKMHWDVJUIO-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCC2=CC(C3=CC=CC=N3)=CC=C2)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CN=CC=N3)C=C2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCC2=CC(C3=CC=CC=N3)=CC=C2)C2=NC(C3=CC=CO3)=NN12.NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CN=CC=N3)C=C2)C2=NC(C3=CC=CO3)=NN12 VBGGKMHWDVJUIO-UHFFFAOYSA-N 0.000 description 1
- CPOHDWMTDRAVCE-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CC=CN(O)=C3)C=C2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCC2=CC=C(C3=CC=CN(O)=C3)C=C2)C2=NC(C3=CC=CO3)=NN12 CPOHDWMTDRAVCE-UHFFFAOYSA-N 0.000 description 1
- FULQTPMYQAVILC-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCCBr)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCCBr)C2=NC(C3=CC=CO3)=NN12 FULQTPMYQAVILC-UHFFFAOYSA-N 0.000 description 1
- CAVJTKMVNNYGPK-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCCN2CCN(C3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCCN2CCN(C3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 CAVJTKMVNNYGPK-UHFFFAOYSA-N 0.000 description 1
- UQNDASFNLFUHMQ-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCC(C(=NO)C3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCC(C(=NO)C3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 UQNDASFNLFUHMQ-UHFFFAOYSA-N 0.000 description 1
- MMYPSUDDZQDUSK-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCC(C(O)C3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCC(C(O)C3=CC=CC=C3)CC2)C2=NC(C3=CC=CO3)=NN12 MMYPSUDDZQDUSK-UHFFFAOYSA-N 0.000 description 1
- PSIMSBRUHYFCCJ-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCC(NC3=CC=CN=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCC(NC3=CC=CN=C3)CC2)C2=NC(C3=CC=CO3)=NN12 PSIMSBRUHYFCCJ-UHFFFAOYSA-N 0.000 description 1
- XDBTZUVEHPKLTN-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(C(=O)C3=CC=C(CO)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(C(=O)C3=CC=C(CO)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 XDBTZUVEHPKLTN-UHFFFAOYSA-N 0.000 description 1
- UXNCRENUVYRSRS-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(C(=O)NC3=CC=C(Cl)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(C(=O)NC3=CC=C(Cl)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 UXNCRENUVYRSRS-UHFFFAOYSA-N 0.000 description 1
- ZUFXJJVGSGHLQB-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(C(=O)OC3=CC=C(CO)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(C(=O)OC3=CC=C(CO)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 ZUFXJJVGSGHLQB-UHFFFAOYSA-N 0.000 description 1
- JUSMPSYMHAPEJS-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CN=C3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CN=C3)=NN12 JUSMPSYMHAPEJS-UHFFFAOYSA-N 0.000 description 1
- JZAMQDDHRXHDFR-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(C3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 JZAMQDDHRXHDFR-UHFFFAOYSA-N 0.000 description 1
- HHQIYVMZCCGZKF-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(C3=CC=CC=C3)CC2)C(Cl)=N1 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(C3=CC=CC=C3)CC2)C(Cl)=N1 HHQIYVMZCCGZKF-UHFFFAOYSA-N 0.000 description 1
- GMVHUEYZJLOASO-UHFFFAOYSA-N NC1=NC2=C(/C=N\N2CCN2CCN(CC3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 Chemical compound NC1=NC2=C(/C=N\N2CCN2CCN(CC3=C(F)C=C(F)C=C3)CC2)C2=NC(C3=CC=CO3)=NN12 GMVHUEYZJLOASO-UHFFFAOYSA-N 0.000 description 1
- ASKOHPBJRRSUSJ-UHFFFAOYSA-N NC1=NC2=C(C=NN2)C(Cl)=N1 Chemical compound NC1=NC2=C(C=NN2)C(Cl)=N1 ASKOHPBJRRSUSJ-UHFFFAOYSA-N 0.000 description 1
- YXRFAPCTJFDKQK-UHFFFAOYSA-N NC1=NC2=C(C=NN2)C2=NC(C3=CC=CS3)=NN12 Chemical compound NC1=NC2=C(C=NN2)C2=NC(C3=CC=CS3)=NN12 YXRFAPCTJFDKQK-UHFFFAOYSA-N 0.000 description 1
- UQNDASFNLFUHMQ-TWKHWXDSSA-N Nc1nc([n](CCN(CC2)CCC2/C(/c2ccccc2)=N/O)nc2)c2c2nc(-c3ccc[o]3)n[n]12 Chemical compound Nc1nc([n](CCN(CC2)CCC2/C(/c2ccccc2)=N/O)nc2)c2c2nc(-c3ccc[o]3)n[n]12 UQNDASFNLFUHMQ-TWKHWXDSSA-N 0.000 description 1
- WJROGXICSWHQDQ-UHFFFAOYSA-N Nc1nc([n](CCN(CC2)CCN2S(c(c(F)c2)ccc2F)(=O)=O)nc2)c2c2nc(-c3ccc[o]3)n[n]12 Chemical compound Nc1nc([n](CCN(CC2)CCN2S(c(c(F)c2)ccc2F)(=O)=O)nc2)c2c2nc(-c3ccc[o]3)n[n]12 WJROGXICSWHQDQ-UHFFFAOYSA-N 0.000 description 1
- DIAIXGWPVSONJO-UHFFFAOYSA-N O=COC1=CC=C(F)O1 Chemical compound O=COC1=CC=C(F)O1 DIAIXGWPVSONJO-UHFFFAOYSA-N 0.000 description 1
- GYLKINOTGGOPNY-UHFFFAOYSA-N [C-]#[N+]C1(C2=CC=CC=C2)CCN(C)CC1 Chemical compound [C-]#[N+]C1(C2=CC=CC=C2)CCN(C)CC1 GYLKINOTGGOPNY-UHFFFAOYSA-N 0.000 description 1
- AOSKKEDKYIRHES-UHFFFAOYSA-N [C-]#[N+]C1=C(OCCOC)C=CC(N2CCN(C)CC2)=C1 Chemical compound [C-]#[N+]C1=C(OCCOC)C=CC(N2CCN(C)CC2)=C1 AOSKKEDKYIRHES-UHFFFAOYSA-N 0.000 description 1
- QHHIHTHVHVGDLK-UHFFFAOYSA-N [C-]#[N+]C1=CC(F)=C(N2CCN(C)CC2)C(F)=C1 Chemical compound [C-]#[N+]C1=CC(F)=C(N2CCN(C)CC2)C(F)=C1 QHHIHTHVHVGDLK-UHFFFAOYSA-N 0.000 description 1
- VRQCUMPKRDCSLE-UHFFFAOYSA-N [C-]#[N+]C1=CC=C(N2CCN(C)CC2)C(F)=C1 Chemical compound [C-]#[N+]C1=CC=C(N2CCN(C)CC2)C(F)=C1 VRQCUMPKRDCSLE-UHFFFAOYSA-N 0.000 description 1
- ZMVUYQSXIFBENE-UHFFFAOYSA-N [C-]#[N+]C1=CC=C(N2CCN(C)CC2)C=C1 Chemical compound [C-]#[N+]C1=CC=C(N2CCN(C)CC2)C=C1 ZMVUYQSXIFBENE-UHFFFAOYSA-N 0.000 description 1
- CKSLOLVETASRFE-JTQLQIEISA-N [H][C@@]1(C(=O)C(C)C)CCCN1C(=O)OC(C)(C)C Chemical compound [H][C@@]1(C(=O)C(C)C)CCCN1C(=O)OC(C)(C)C CKSLOLVETASRFE-JTQLQIEISA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains three hetero rings
- C07D487/14—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the present invention relates to substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine adenosine A 2a receptor antagonists, the use of said compounds in the treatment of central nervous system diseases, in particular Parkinson's disease, and to pharmaceutical compositions comprising said compounds.
- the invention also relates to a process for preparing 5-amino-2-(substituted)pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines, intermediates useful in preparing the claimed compounds.
- Adenosine is known to be an endogenous modulator of a number of physiological functions. At the cardiovascular system level, adenosine is a strong vasodilator and a cardiac depressor. On the central nervous system, adenosine induces sedative, anxiolytic and antiepileptic effects. On the respiratory system, adenosine induces bronchoconstriction. At the kidney level, it exerts a biphasic action, inducing vasoconstriction at low concentrations and vasodilation at high doses. Adenosine acts as a lipolysis inhibitor on fat cells and as an antiaggregant on platelets.
- Adenosine action is mediated by the interaction with different membrane specific receptors which belong to the family of receptors coupled with G proteins.
- Biochemical and pharmacological studies, together with advances in molecular biology, have allowed the identification of at least four subtypes of adenosine receptors: A 1 , A 2a , A 2b and A 3 .
- a 1 and A 3 are high-affinity, inhibiting the activity of the enzyme adenylate cyclase, and A 2a and A 2b , are low-affinity, stimulating the activity of the same enzyme.
- Analogs of adenosine able to interact as antagonists with the A 1 , A 2a , A 2b and A 3 receptors have also been identified.
- a 2a antagonists for the A 2a receptor are of pharmacological interest because of their reduced level of side affects.
- a 2a antagonists can have antidepressant properties and stimulate cognitive functions.
- data has shown that A 2a receptors are present in high density in the basal ganglia, known to be important in the control of movement.
- a 2a antagonists can improve motor impairment due to neurodegenerative diseases such as Parkinson's disease, senile dementia as in Alzheimer's disease, and psychoses of organic origin.
- xanthine-related compounds have been found to be A 1 receptor selective antagonists, and xanthine and non-xanthine compounds have been found to have high A 2a affinity with varying degrees of A 2a vs. A 1 selectivity.
- Triazolo-pyrimidine adenosine A 2a receptor antagonists with different substitution at the 7-position have been disclosed previously, for example in WO 95/01356; U.S. Pat. No. 5,565,460; WO 97/05138; and WO 98/52568.
- the present invention relates to compounds having the structural formula I
- R is R 1 -furanyl, R 1 -pyridyl, R 1 -pyridyl N-oxide, R 1 -oxazolyl, R 10 -phenyl, R 1 -pyrrolyl or C 4 -C 6 cycloalkenyl;
- X is C 2 -C 6 alkylene or —C(O)CH 2 —;
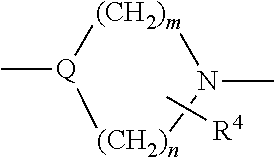
- Y is —N(R 2 )CH 2 CH 2 N(R 3 )—, —OCH 2 CH 2 N(R 2 )—, —O—, —S—, —CH 2 S—, —(CH 2 ) 2 —NH—, or
- Z is R 5 -phenyl, R 5 -phenyl(C 1 -C 6 )alkyl, R 5 -heteroaryl, diphenylmethyl, R 6 —C(O)—, R 6 —SO 2 —, R 6 —OC(O)—, R 7 —N(R 8 )—C(O)—, R 7 —N(R 8 )—C(S)—,
- Z is also phenylamino or pyridylamino
- R 1 is 1 to 3 substituents independently selected from hydrogen, C 1 -C 6 -alkyl, —CF 3 , halogen, —NO 2 , —NR 12 R 13 , C 1 -C 6 alkoxy, C 1 -C 6 alkylthio, C 1 -C 6 alkylsulfinyl, and C 1 -C 6 alkylsulfonyl;
- R 2 and R 3 are independently selected from the group consisting of hydrogen and C 1 -C 6 alkyl
- R 4 is 1-2 substituents independently selected from the group consisting of hydrogen and C 1 -C 6 alkyl, or two R 4 substituents on the same carbon can form ⁇ O;
- R 3 is 1 to 5 substituents independently selected from the group consisting of hydrogen, halogen, C 1 -C 6 alkyl, hydroxy, C 1 -C 6 alkoxy, —CN, di-((C 1 -C 6 )alkyl)amino, —CF 3 , —OCF 3 , acetyl, —NO 2 , hydroxy(C 1 -C 6 )alkoxy, (C 1 -C 6 )-alkoxy(C 1 -C 6 )alkoxy, di-((C 1 -C 6 )-alkoxy) (C 1 -C 6 )alkoxy, (C 1 -C 6 )-alkoxy(C 1 -C 6 )alkoxy-(C 1 -C 6 )-alkoxy, carboxy(C 1 -C 6 )-alkoxy, (C 1 -C 6 )-alkoxycarbonyl (C 1 -C 6 )alkoxy, (C 3
- R 5 substituents together are —O—CH 2 —O—, —O—CH 2 CH 2 —O—, —O—CF 2 —O— or —O—CF 2 CF 2 —O— and form a ring with the carbon atoms to which they are attached;
- R 6 is (C 1 -C 6 )alkyl, R 5 -phenyl, R 5 -phenyl(C 1 -C 6 )alkyl, thienyl, pyridyl, (C 3 -C 6 )-cycloalkyl, (C 1 -C 6 )alkyl-OC (O)—NH—(C 1 -C 6 )alkyl-, di((C 1 -C 6 )alkyl)aminomethyl, or
- R 7 is (C 1 -C 6 )alkyl, R 5 -phenyl or R 5 -phenyl(C 1 -C 6 )alkyl;
- R 8 is hydrogen or C 1 -C 6 alkyl; or R 7 and R 8 together are —(CH 2 ) p —A—(CH 2 ) q , wherein p and q are independently 2 or 3 and A is a bond, —CH 2 —, —S— or —O—, and form a ring with the nitrogen to which they are attached;
- R 9 is 1-2 groups independently selected from hydrogen, C 1 -C 6 alkyl, hydroxy, C 1 -C 6 alkoxy, halogen, —CF 3 and (C 1 -C 6 )alkoxy(C 1 -C 6 )alkoxy;
- R 10 is 1 to 5 substituents independently selected from the group consisting of hydrogen, halogen, C 1 -C 6 alkyl, hydroxy, C 1 -C 6 alkoxy, —CN, —NH 2 , C 1 -C 6 alkylamino, di-((C 1 -C 6 )alkyl)amino, —CF 3 , —OCF 3 and —S(O) 0-2 (C 1 -C 6 )alkyl;
- R 11 is H, C 1 -C 6 alkyl, phenyl, benzyl, C 2 -C 6 alkenyl, C 1 -C 6 alkoxy(C 1 -C 6 )alkyl, di-((C 1 -C 6 )alkyl)amino (C 1 -C 6 )alkyl, pyrrolidinyl(C 1 -C 6 )alkyl or piperidino (C 1 -C 6 )alkyl;
- R 12 is H or C 1 -C 6 alkyl
- R 13 is (C 1 -C 6 )alkyl-C(O)— or (C 1 -C 6 )alkyl-SO 2 —.
- Preferred compounds of formula I are those wherein R is R 1 -furanyl, R 1 -thienyl, R 1 -pyrrolyl or R 10 -phenyl, more preferably R 1 -furanyl.
- R 1 is preferably hydrogen or halogen.
- Another group of preferred compounds is that wherein X is alkylene, preferably ethylene.
- Y is preferably
- R 4 is H.
- a preferred definition for Z is R 5 -phenyl, R 5 -heteroaryl, R 6 —C(O)— or R 6 —SO 2 —.
- R 5 is preferably H, halogen, alkyl, alkoxy, hydroxyalkoxy or alkoxyalkoxy.
- R 6 is preferably R 5 -phenyl.
- Another aspect of the invention is a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a compound of formula I in a pharmaceutically acceptable carrier.
- Yet another aspect of the invention is a method of treating central nervous system diseases such as depression, cognitive diseases and neurodegenerative diseases such as Parkinson's disease, senile dementia or psychoses of organic origin, and stroke, comprising administering a compound of formula I to a mammal in need of such treatment.
- central nervous system diseases such as depression, cognitive diseases and neurodegenerative diseases such as Parkinson's disease, senile dementia or psychoses of organic origin, and stroke
- the invention is drawn to the method of treating Parkinson's disease comprising administering a compound of formula I to a mammal in need of such treatment.
- Another aspect of the invention is a process for preparing 5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines of formula II, which are intermediates useful in the preparation of compounds of formula I.
- the process of preparing compounds of formula II is a process for preparing 5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines of formula II, which are intermediates useful in the preparation of compounds of formula I.
- R is as defined above, comprises
- a preferred aspect of the process is the dehydrative rearrangement of the intermediate of formula IX to obtain the 5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine of formula II.
- Preferred embodiments of the process use 2-furoic hydrazide or 2-thienoylhydrazide in step 2, thus preparing compounds of formula II wherein R is 2-furyl or 2-thienyl.
- Another aspect of the invention is a process for preparing 7-bromoalkyl-5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines of formula IIIa, which are intermediates useful in the preparation of compounds of formula I.
- the process of preparing compounds of formula IIIa is a process for preparing 7-bromoalkyl-5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines of formula IIIa, which are intermediates useful in the preparation of compounds of formula I.
- R is as defined above, comprises
- Still another aspect of the invention is a method of treating Parkinson's disease with a combination of a compound of formula I and one or more agents known to be useful in the treatment of Parkinson's disease, for example dopamine; a dopaminergic agonist; an inhibitor of monoamine oxidase, type B (MAO-B); a DOPA decarboxylase inhibitor (DCI); or a catechol-O-methyltransferase (COMT) inhibitor.
- dopamine a dopaminergic agonist
- MAO-B an inhibitor of monoamine oxidase, type B
- DCI DOPA decarboxylase inhibitor
- COMP catechol-O-methyltransferase
- composition comprising a compound of formula I and one or more agents known to be useful in the treatment of Parkinson's in a pharmaceutically acceptable carrier.
- alkyl includes straight or branched chains.
- Alkylene referring to a divalent alkyl group, similarly refers to straight or branched chains.
- Cycloalkylene refers to a divalent cycloalkyl group.
- Cycloalkenyl refers to a C 4 -C 6 cycloalkyl ring comprising one double bond.
- Heteroaryl means a single ring, bicyclic or benzofused heteroaromatic group of 5 to 10 atoms comprised of 2 to 9 carbon atoms and 1 to 4 heteroatoms independently selected from the group consisting of N, O and S, provided that the rings do not include adjacent oxygen and/or sulfur atoms. N-oxides of the ring nitrogens are also included.
- single-ring heteroaryl groups are pyridyl, oxazolyl, isoxazolyl, oxadiazolyl, furanyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, tetrazolyl, thiazolyl, isothiazolyl, thiadiazolyl, pyrazinyl, pyrimidyl, pyridazinyl and triazolyl.
- bicyclic heteroaryl groups are naphthyridyl (e.g., 1, 5 or 1,7), imidazopyridyl, pyrido[2,3]imidazolyl, pyridopyrimidinyl and 7-azaindolyl.
- benzo-fused heteroaryl groups are indolyl, quinolyl, isoquinolyl, phthalazinyl, benzothienyl (i.e., thionaphthenyl), benzimidazolyl, benzofuranyl, benzoxazolyl and benzofurazanyl.
- R 5 -substituted heteroaryl refers to such groups wherein substitutable ring carbon atoms have a substituent as defined above.
- Certain compounds of the invention may exist in different stereoisomeric forms (e.g., enantiomers, diastereoisomers and atropisomers).
- the invention contemplates all such stereoisomers both in pure form and in mixture, including racemic mixtures.
- Certain compounds will be acidic in nature, e.g. those compounds which possess a carboxyl or phenolic hydroxyl group. These compounds may form pharmaceutically acceptable salts. Examples of such salts may include sodium, potassium, calcium, aluminum, gold and silver salts. Also contemplated are salts formed with pharmaceutically acceptable amines such as ammonia, alkyl amines, hydroxyalkylamines, N-methylglucamine and the like.
- Certain basic compounds also form pharmaceutically acceptable salts, e.g., acid addition salts.
- pyrido-nitrogen atoms may form salts with strong acid, while compounds having basic substituents such as amino groups also form salts with weaker acids.
- suitable acids for salt formation are hydrochloric, sulfuric, phosphoric, acetic, citric, oxalic, malonic, salicylic, malic, fumaric, succinic, ascorbic, maleic, methanesulfonic and other mineral and carboxylic acids well known to those skilled in the art.
- the salts are prepared by contacting the free base form with a sufficient amount of the desired acid to produce a salt in the conventional manner.
- the free base forms may be regenerated by treating the salt with a suitable dilute aqueous base solution such as dilute aqueous NaOH, potassium carbonate, ammonia and sodium bicarbonate.
- a suitable dilute aqueous base solution such as dilute aqueous NaOH, potassium carbonate, ammonia and sodium bicarbonate.
- the free base forms differ from their respective salt forms somewhat in certain physical properties, such as solubility in polar solvents, but the acid and base salts are otherwise equivalent to their respective free base forms for purposes of the invention.
- the compounds of formula I are prepared by the methods shown in the following reaction schemes.
- alkylation of a 5-amino-pyrazolo[4,3-c]-[1,2,4]-triazolo[1,5-c]pyrimidine of formula II is used to prepare compounds of formula I:
- Starting materials of formula II can be reacted with an alkyl diol ditosylate and a base such as NaH in an inert solvent such as dimethylformamide (DMF), or with a chloro-bromo- or dibromo-alkyl compound under similar conditions, to obtain the alkyl-substituted intermediate of formula II.
- the compound of formula III is then reacted with an amine of the formula Z—Y—H in an inert solvent such as DMF at an elevated temperature to obtain a compound of formula Ia, i.e., a compound of formula I wherein X is alkylene.
- staring materials of formula II can be reacted with a compound of formula Z—Y—X—Cl and a base such as NaH in an inert solvent such as DMF to obtain a mixture of a 7-substituted compound of formula I and the corresponding 8-substituted compound.
- chloropyrazolo-pyrimidine V is reacted with a compound of formula Z—Y—X—Cl in a manner similar to the alkylation procedure of Scheme 1, and the resultant intermediate is reacted with a hydrazide of formula H 2 N—NH—C(O)—R (or with hydrazine hydrate, followed by a compound of formula Cl—C(O)—R).
- the resultant hydrazide undergoes dehydrative rearrangement, e.g., by treatment with N,O-bis-(trimethylsilyl)acetamide (BSA) or a combination of BSA and hexamethyldisilazane (HMDS) and at elevated temperatures.
- BSA N,O-bis-(trimethylsilyl)acetamide
- HMDS hexamethyldisilazane
- 2-amino-4,6-dihydroxypyrimidine (VI) is converted to the corresponding 4,6-dichloro-5-carboxaldehyde by treatment with POCl 3 or SOCl 2 in DMF as described in Helv. Chim. Acta, 69 (1986), 1602-1613.
- the reaction is carried out at an elevated temperature, preferably about 100° C., for 2 to 8 hours, preferably about 5 hours.
- 2-amino-4,6-dichloropyrimidine-5-carboxaldehyde (VII) is treated with a hydrazide of the formula H 2 N—NH—C(O)—R, wherein R is as defined above, to obtain the compound of formula VIII; the compound of formula VI and the hydrazide are used in a molar ratio of approximately 1:1, with a slight excess of the hydrazide being preferred.
- the reaction is carried out at room temperature or up to about 80° C. in a solvent such as CH 3 CN or DMF.
- the reaction time is about 16 hours (e.g., overnight).
- the compound of formula VIII is heated at 60-100° C. with 1-5 equivalents of hydrazine hydrate in a solvent such as CH 3 CN or DMF for 1-24 hours to obtain the compound of formula IX.
- the compound of formula IX undergoes dehydrative rearrangement by treatment with a mixture of HMDS and BSA or with BSA alone.
- the reaction is carried out at elevated temperatures, preferably about 120° C., for about 16 hours (e.g., overnight)
- the crude material is purified by conventional methods, e.g., extraction and/or recrystallization.
- Chloride VIII is treated with a hydroxyalkyl-hydrazine in an inert solvent such as ethanol at temperatures from ambient to 100° C. to furnish derivative X. This is subjected to dehydrative cyclization, similarly to IX, such as with BSA, to provide tricyclic XI. Tricyclic XI is then converted to bromide IIIa with PBr 3 at elevated temperature from 80° C. to 150° C. for 1 to 24 hours. Intermediate XI can also be converted into the tosylate analogous to IIIa by toluenesulfonyl chloride and base. Bromide IIIa is converted to compounds of formula I as described above for III.
- an inert solvent such as ethanol
- chloride V is converted into alkylated compound XII, and this is further reacted with carbazate XIV, where R′ is preferably t-butyl or benzyl, to obtain derivative XIII.
- a solvent such as DMF may be employed at a temperature of 60-120° C. This is then reacted as in Scheme 1 to furnish XV.
- the R′ group is next removed, such as removal of a t-butyl group with HCl or TFA, furnishing hydrazine XVI.
- Acylation of XVI furnishes XVII, which is subjected to dehydrative cyclization as described above to provide desired Ia.
- XII may be reacted with a hydrazide XVIII to obtain XIX, which can be converted to XVII analogously to preparation of XV.
- Step 4 Heat the product of Step 3 (5.4 g, 0.021 mol) in a mixture of hexamethyl-disilazine (100 ml) and N,O-bis (trimethylsilyl) acetamide (35 ml) at 120° C. overnight. Remove volatiles under vacuum and slurry the residue in hot water to give a solid precipitate. Recrystallize from 80% aqueous acetic acid to give the title compound.
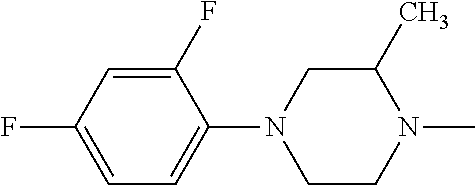
- 1-(2,4-Difluorophenyl)piperazine is prepared from 2,4-difluorobromobenzcne.
- bromide 8.0 g, 41.4 mmol
- piperazine (21.4 g, 249 mmol)
- sodium t-butoxide 5.6 g, 58 mmol
- BINAP BINAP
- Pd 2 (dba) 3 0.477 g, 0.83 mmol
- 1-(5-Ethyl-2-pyrimidinyl)piperazine is prepared from 2-chloro-5-ethylpyrimidine. Heat the chloride (2.0 g, 14 mmol) and piperazine (3.0 g, 35 mmol) in EtOH (70 ml) at 90° C. for 2 h in a sealed vessel. Concentrate and partition between CH 2 Cl 2 and 2N NaOH. Dry the organic with MgSO 4 and concentrate. Chromatograph the crude product on silica (CH 2 Cl 2 —CH 3 OH) to obtain the piperazine as a yellow oil.
- 1-(4-(2-Methoxyethoxy)phenyl)piperazine is prepared from 4-(4-hydroxy-phenyl)-1-acetylpiperazine.
- NaH 50% in mineral oil, 0.79 g, 20 mmol
- DMF 25 ml
- 2-bromoethyl methyl ether 2.27 g, 16.3 mmol
- 4-(2-Methylaminoethoxy)fluorobenzene is prepared from 4-(2-bromo-ethoxy)-fluorobenzene. Combine the bromide (1.0 g, 4.6 mmol) in CH 3 OH (5 ml) with CH 3 NH 2 in CH 2 OH (2M, 46 ml, 92 mmol) in a sealed vessel. Heat at 60° C. for 18 h, concentrate, and partition between EtOAc and sat, NaHCO 3 . Wash the organic with brine, dry with MgSO 4 , and concentrate to obtain the amine as a yellow oil.
- N-methyl-2-(4-(2-methoxyethoxy)phenoxy)ethylamine was prepared in two steps. Combine 4-(2-methoxyethoxy)phenol (1.68 g, 10.0 mmol), 1,2-dibromoethane (16.9 g, 90 mmol), and K 2 CO 3 (2.76 g, 20 mmol) in CH 3 CN (20 ml) and DMF (10 ml). Heat at reflux 22 h, allow to cool, filter, and partition between ether (Et 2 O) and 1N NaOH. Wash with brine, dry over MgSO 4 , and concentrate to provide the bromoethyl ether as beige solid.
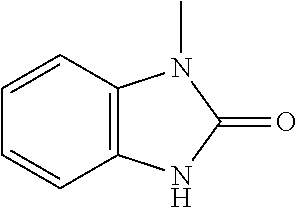
- 1-Phenyl-2-piperazinone is prepared from 4-benzyloxycarbonyl-1-phenyl-2-piperazinone. Combine this material (1.61 g, 5.2 mmol) with 10% Pd/C (0.4 g) in EtOH (50 ml) and 1N HCl (6 ml). Hydrogenate at 45 psi for 2 h and filter. Concentrate and chromatograph the residue on silica (eluting with CH 2 Cl 2 :CH 3 OH:NH 4 OH) to obtain the piperazinone as a cream solid.
- Example 1-2 The compound of Example 1-2 was also prepared by the following procedure:
- Example 1-3 The compound of Example 1-3 is similarly prepared, as are the following compounds:
- Step 2 To the product of Step 1 (2.15 g, 7.2 mmol) in THF (10 ml), add LiAlH 4 (1.0 M in THF, 4.4 ml, 4.4 mmol) dropwise. Heat at 60° C. 1 h, add water (0.16 ml), 15% NaOH (0.16 ml), and then water (0.49 ml). Filter and concentrate to obtain the alcohol as a yellow oil, NMR (CDCl 3 ) consistent.
- Step 2 To the product of Step 2 (0.90 g, 3.5 mmol) in CH 2 Cl 2 (10 ml) at 5° C., add SOCl 2 (0.38 ml, 5.3 mmol). Allow to warm and stir 16 h. Concentrate and partition between CH 2 Cl 2 and 1N NaOH, wash with water, dry (MgSO 4 ) and concentrate to obtain the crude product as a yellow oil.
- Step 4 Combine the product of Preparation 1 (0.20 g, 0.83 mmol), the product of Step 3 (0.34 g, 1.2 mmol) and NaH (60% in oil, 0.040 g, 1.0 mmol) in dry DMF (5 ml). Heat at 60° C. for 24 h, add additional chloride (0.15 g) and NaH (0.02 g), and heat another 4 h. Concentrate and purify by flash column chromatography (CH 2 Cl 2 , CH 3 OH+NH 3 ) to obtain the title compound as a yellow solid, mass spectrum m/e 479 (M+H).
- Step 1 Combine 1-bromo-2,4-difluorobenzene (1.00 g, 5.18 mmol), N,N′-dimethyl-ethylenediamine (2.74 g, 31.1 mmol), NaO-t-Bu (0.70 g, 7.2 mmol), Pd(dba) 2 (0.060 g, 0.10 mmol) and ( ⁇ )-BINAP (0.19 g, 0.31 mmol) in toluene (10 ml). Heat at 110° for 18 h, allow to cool, and extract with 1N HCl. Basify the aqueous solution with NaOH and extract with CH 2 Cl 2 . Dry, concentrate, and purify by PLC to give N-(2,4-difluoro-phenyl)-N,N′-dimethylethylenediamine.
- Example 1-2 The compound of Example 1-2 was also prepared by the following procedure.
- Step 3 The product of Step 3 was treated as described in Preparation 1, Steps 2 and 4, to obtain the compound of Example 1-2.
- Step 1 To NaH (60% in oil, 142 mg, 3.5 mmol) in DMF (15 ml) add the chloride of Example 11, Step 1 (500 mg, 2.9 mmol). Add to this 1-(2-chloroethyl)-4-(2,4-difluorophenyl) piperazine (846 mg, 3.5 mmol). Stir at RT 90 h and concentrate. Chromatograph to obtain the desired compound as a white solid. PMR in DMSO: ⁇ 2.57 (4H, s), 2.76 (2H, t), 2.85 (4H, s), 4.30 (2H, t), 7.0 (2H, m), 7.15 (1H, dxt), 7.26 (2H, s), 7.97(1H, s).
- the 1-(2-chloroethyl)-4-(2,4-difluorophenyl)piperazine employed in this sequence is prepared in two steps. Add chloroacetyl chloride (1.76 ml, 22.1 mmol) and N-methylmorpholine (2.65 ml, 24.1 mmol) to 1-(2,4-difluorophenyl)piperazine (3.98 g, 20.1 mmol) in CH 2 Cl 2 (15 ml) at 0° C. Stir at RT 1 h, concentrate, partition EtOAc-water, dry, and concentrate to obtain the amide as a brown oil. To a 0° C.
- Step 1 To NaH (2.14 g, 60% in oil, 53 mmol) in DMF (20 ml), add the product of Example 11, Step 1 (7.55 g, 45 mmol). Add 1-bromo-2-chloroethane (14.8 ml, 178 mmol). Stir 1.5 h and concentrate. Chromatograph to give the dichloride as a white solid.
- Step 2 In the product of Step 1 (3.7 g, 16 mmol) in DMF (20 ml) add 1-butyl carbazate (2.53 g, 19 mmole). Heat at 80° C. for 18 h and concentrate. Chromatograph to obtain the carbazate as a while solid.
- Step 3 To the product of Step 2 (3.16 g, 9.6 mmol) and KI (1.6 g, 9.6 mmol) in DMF (25 ml) add 1-(2,4-difluorophenyl)piperazine (3.82 g, 19 mmol). Heat at 90° C. for 68 h and concentrate. Chromatograph to obtain the piperazine as a brown solid.
- Step 4 Dissolve the product of Step 3 (3.38 g, 6.9 mmol) in 1:1 CH 3 OH—CH 2 Cl 2 (50 ml). Add 4M HCl in dioxane (20 ml). Stir 16 h and add aq. NH 3 to pH 11-12. Concentrate and chromatograph to obtain the hydrazine as a yellow solid.
- Step 5 Combine the product of Step 4 (0.120 g, 0.31 mmol) with 5-bromo-2-furoic acid (0.071 g, 0.37 mmol) and HOBt.H 2 O (0.050 g, 0.37 mmol) in DMF (6 ml). Add EDCl (0.071 g, 0.37 mmol) and stir 1 h. Concentrate and chromatograph to obtain the hydrazide as a yellow solid.
- Step 6 Dissolve the product of Step 5 (0.163 g, 0.28 mmol) in N,O-bis(trimethylsilyl) acetamide (6 ml). Heat at 120° C. for 16 h and pour into CH 3 OH. Concentrate and chromatograph to obtain the title product as an off-white solid: MS m/e 544+546 (M+1).
- Step 1 To the product of Example 13, Step 2 (3.54 g, 10.8 mmol) and KI (1.79 g, 10.8 mmol) in DMF (35 ml) add 1-(4-(2-methoxyethoxy)phenyl)piperazine (5.1 g, 22 mmol). Heat at 90° C. for 90 h and concentrate. Chromatograph to obtain the piperazine as a brown solid.
- Step 2 Treat the product of Step 1 with HCl as in Example 13, Step 4, to obtain the hydrazine as a yellow solid.
- Step 3 Treat the product of Step 2 with 5-chloro-2-furoic acid as in Example 13, Step 5, to obtain the hydrazide as a yellow solid.
- Step 4 Treat the product of Step 3 with BSA as in Example 13, Step 6. Chromatograph to obtain the title compound as a white solid, MS m/e 538+540 (M+1).
- Step 1 To a solution of 4-bromophenethyl alcohol (0.600 g, 2.98 mmol) and 3-pyridinylboronic acid (0.734 g, 5.97 mmol) in toluene (35 ml) and EtOH (9 ml), add a solution of K 2 CO 3 (0.8826 g, 5.97 mmol) in H 2 O (16 ml) and tetrakis(triphenyl-phosphine)palladium(0) (0.172 g, 0.149 mmol). Heat in a sealed tube 18 h at 120° C. and cool. Extract with EtOAc, wash with brine, dry (K 2 CO 3 ) and concentrate. Chromatograph on silica (30-50% EtOAc/hexanes) to obtain the biaryl alcohol.
- Step 2 To the product of Step 1 (0.540 g, 2.71 mmol) in CH 2 Cl 2 (15 ml) at 0° C. add mesyl chloride (0.35 ml, 3.52 mmol) and Et 3 N (0.57 ml, 4.00 mmol). Stir 2.5 h and extract with CH 2 Cl 2 . Dry (Na 2 SO 4 ) and concentrate to obtain the mesylate.
- Step 3 Add the product of Preparation 4 (0.347 g, 1.44 mmol) to the mesylate of Step 2 (0.480 g, 1.73 mmol) in DMF (4.5 ml), followed by NaH (60%. in oil, 0.082 g, 4.04 mmol). Stir 18 h and extract with EtOAc. Wash with H 2 O dry (K 2 CO 3 ) and concentrate. Purify by PTLC (5% CH 3 OH/CH 2 Cl 2 , developed twice) to obtain the title compound as a white solid, MS: 423 (M+1).
- Step 1 Combine 4-bromophenethyl alcohol (3.00 g, 14.9 mmol), triethylamine (2.68 ml, 19.2 mmol), dimethylaminopyridine (0.180 g, 1.47 mmol) and t-butyidimethylsilyl chloride (2.45 g, 16.3 mmol) in CH 2 Cl 2 (75 ml). Stir 1 h, wash with H 2 O, dry (K 2 CO 3 ), and concentrate. Chromatograph on silica (hexanes) to obtain the silyl ether.
- Step 2 To the compound of Step 1 (0.300 g, 0.95 mmol) in dry toluene (15 ml) add 2-(tri-butylstannyl)pyridine (1.05 g, 2.86 mmol) and tetrakis(triphenylphosphine)-palladium (0.11 g, 0.095 mmol). Flush with N 2 and heat 16 h at 120° C. Cool, filter through Celite, and wash with NH 4 Cl, brine and then water. Dry (K 2 CO 3 ) and concentrate. Chromatograph on silica (3-5% EtOAc/hexanes) to obtain the biaryl, MS 314 (M+1).
- Step 3 Combine the biaryl of Step 2 (0.180 g, 0.57 mmol) and TBAF (1.0 M in THF, 1.7 ml) in THF (5.7 ml). Stir 2 h, wash with saturated NH 4 Cl, and extract with EtOAc. Wash with H 2 O several times, dry (K 2 CO 3 ) and concentrate to obtain the alcohol.
- Steps 4 and 5 Conduct as in Example 18, Steps 2 and 3, to obtain the title compound as a white solid, MS: 423 (M+1).
- Example 18 To the product of Example 18 (0.055 g, 0.13 mmol) in CH 2 Cl 2 (1.5 ml) at ⁇ 78° C. add m-CPBA (0.050 g, 0.29 mmol). Allow to warm, stir 5 h, and wash successively with sat. Na 2 S 2 O 3 , 5% K 2 CO 3 , and H 2 O. Dry (Na 2 SO 4 ) and concentrate. Purify by PTLC (10% CH 3 OH/CH 2 Cl 2 ) to obtain the title compound, MS: 439 (M+1).
- Step 1 Combine 1,4-dioxa-8-azaspiro(4,5)decane (0.48 ml, 3.8 mmol) with the product of Preparation 2 (0.66 g, 1.5 mmol) in DMF (10 ml). Heat at 90° C. for 16 h, allow to cool, filter and wash with CH 3 OH to give off-white solid, MS: m/e 411 (M+1).
- Step 2 Heat the product of Step 1 (0.476 g, 1.16 mmol) in acetone (10 ml) and 5% HCl (10 ml) at 100° C. for 16 h. Cool, neutralize with sat. NaHCO 3 , and extract with 10% CH 3 OH in CH 2 Cl 2 . Dry (MgSO 4 ), concentrate and chromatograph on silica with CH 3 OH—CH 2 Cl 2 to obtain the ketone as a white powder, MS: m/e 367 (M+1).
- Step 3 Combine the product of Step 2 (0.050 g, 0.13 mmol) with O-methylhydroxyl-amine hydrochloride (0.033 g, 0.39 mmol) in pyridine (3 ml). Stir for 16 h and concentrate. Partition between NaHCO 3 (sat.) and 5% CH 3 OH in CH 2 Cl 2 . Dry (MgSO 4 ), concentrate and chromatograph on silica with 5% CH 3 OH—CH 2 Cl 2 to obtain the title compound as a white solid, MS: m/e 396 (M+1).
- Step 1 Combine benzyl 4-oxo-1-piperidinecarboxylate (1.0 g, 4.3 mmol) with H 2 NOH.HCl (0.89 g, 13 mmol) in pyridine (5 ml). Stir 16 h and concentrate. Partition between NaHCO 3 (sat.) and EtOAc, dry (MgSO 4 ) and concentrate to give the oxime.
- Step 2 Combine the product of Step 1 (0.44 g, 1.8 mmol) with 2-bromoethyl methyl ether (0.20 ml, 2.2 mmol) and NaH (0.10 g, 2.7 mmol) in DMF (8 ml). Stir 16 h and concentrate. Partition between NH 4 Cl (sat.) and ether, dry (MgSO 4 ), and concentrate. Chromatograph the residue on silica with 20% EtOAc-hexane to obtain the alkylated oxime.
- Step 3 Stir the product of Step 2 (0.45 g, 1.47 mmol) over 5% Pd/C (0.045 g) in EtOAc (25 ml) under H 2 for 6 h. Filter and concentrate to obtain the amine.
- Step 4 Treat the amine of Step 3 with the product of Preparation 2 as in Example 24, Step 1, to obtain the title compound as a white solid, MS: m/e 440 (M+1).
- Step 1 Combine 4-bromophenol (3.46 g, 20.0 mmol) with 2-bromoethyl methyl ether (2.82 ml, 30.0 mmol) and K 2 CO 3 (8.30 g, 60.0 mmol) in acetone (50 ml). Heat at reflux 16 h, cool, filter, and concentrate. Chromatograph on silica with 5% EtOAc/hexane to give the ether as a clear oil. To this ether (2.73 g, 11.8 mmol) in dry THF (50 ml) at ⁇ 78° C. add n-BuLi (1.6 M in hexane, 7.4 ml, 11.8 mmol). Stir for 10 min.
- Step 2 To a solution of the product of Step 1 (0.386 g, 1.0 mmol) and triethylsilane (0.80 ml, 5.0 mmol) in dry CH 2 Cl 2 (10 ml) at ⁇ 78° C. add trifluoroacetic acid (0.38 ml, 5.0 mmol). Allow to warm over 2 h and partition between sat. NaHCO 3 and CH 2 Cl 2 . Dry (MgSO 4 ) and concentrate. Chromatograph on silica with 20% EtOAc/hexane to obtain the reduction product, MS: m/e 370 (M+1).
- Step 3 Stir the product of Step 2 (0.300 g, 0.758 mmol) over 5% Pd/C (0.030 g) in EtOAc (5 ml) and CH 3 OH (5 ml) under H 2 for 2 h. Filter and concentrate to obtain the amine.
- Step 4 Treat the amine of Step 3 with the product of Preparation 2 as in Example 24, Step 1, to obtain the title compound as a white solid, MS: m/e 503 (M+1).
- Example 1-145 Treat the product of Example 1-145 (0.020 g, 0.044 mmol) in EtOH (0.5 ml) at 0° C. with sodium borohydride (0.005 g, 0.13 mmol) and with an equal amount again after 0.75 h. After another 0.75 h, partition between CH 2 Cl 2 and sat. NH 4 Cl. Dry (Na 2 SO 4 ) and concentrate. Purify by PTLC (10% CH 3 OH/CH 2 Cl 2 ) to obtain the title compound as a white solid, MS: 459 (M+1).
- Example 1-145 Treat the product of Example 1-145 (0.020 g, 0.044 mmol) in pyridine (0.5 ml) with methoxyamine hydrochloride (0.011 g, 0.13 mmol). Stir 16 h and concentrate. Partition between CH 2 Cl 2 and sat. NaHCO 3 . Dry (Na 2 SO 4 ) and concentrate. Purify by PTLC (5% CH 3 OH/CH 2 Cl 2 ) to obtain the title compound as a white solid, MS: 486 (M+1).
- compounds of the present invention are useful in the treatment of depression, cognitive function diseases and neurodegenerative diseases such as Parkinson's disease, senile dementia as in Alzheimer's disease, and psychoses of organic origin.
- the compounds of the present invention can improve motor-impairment due to neurodegenerative diseases such as Parkinson's disease.
- the other agents known to be useful in the treatment of Parkinson's disease which can be administered in combination with the compounds of formula I include: L-DOPA; dopaminergic agonists such as quinpirole, ropinirole, pramipexole, pergolide and bromocriptine; MAO-B inhibitors such as deprenyl and selegiline; DOPA decarboxylase inhibitors such as carbidopa and benserazide; and COMT inhibitors such as tolcapone and entacapone.
- One to three other agents can be used in combination with the compounds of formula I, preferably one.
- the pharmacological activity of the compounds of the invention was determined by the following in vitro and in vivo assays to measure A 2a receptor activity.
- a 2a Human A 2a Adenosine Receptor membranes, Catalog #RB-HA2a, Receptor Biology, Inc., Beltsville, Md. Dilute to 17 ⁇ g/100 ⁇ l in membrane dilution buffer (see below).
- Membrane dilution buffer Dulbecco's Phosphate Buffered Saline (Gibco/BRL)+10 mM MgCl 2 .
- Compound Dilution Buffer Dulbecco's Phosphate Buffered Saline (Gibco/BRL)+10 mM MgCl 2 supplemented with 1.6 mg/ml methyl cellulose and 16% DMSO. Prepared fresh daily.
- a 2a [3H]-SCH 58261, custom synthesis, AmershamPharmacia Biotech, Piscataway, N.J. Stock is prepared at 1 nM in membrane dilution buffer. Final assay concentration is 0.5 nM.
- a 1 [3H]-DPCPX, AmershamPharmacia Biotech, Piscataway, N.J. Stock is prepared at 2 nM in membrane dilution buffer. Final assay concentration is 1 nM.
- a 2a To determine non-specific binding, add 100 nM CGS 15923 (RBI, Natick, Mass.). Working stock is prepared at 400 nM in compound dilution buffer.
- a 1 To determine non-specific binding, add 100 ⁇ M NECA (RBI, Natick, Mass). Working stock is prepared at 400 ⁇ M in compound dilution buffer.
- Total assay volume is 200 ⁇ l.
- the selective A 2A adenosine antagonists under evaluation are administered orally at doses ranging between 0.03 and 3 mg/kg, 1 and 4 h before scoring the animals.
- Rats Male male Sprague-Dowley rats (Charles River, Calco, Como, Italy), weighing 275-300 g, are used in all experiments. The rats are housed in groups of 4 per cage, with free access to food and water, under controlled temperature and 12 hour light/dark cycle. The day before the surgery the rats are fasted over night with water ad libitum.
- Unilateral 6-hydroxydopamine (6-OHDA) lesion of the middle forebrain bundle is performed according to the method described by Ungerstedt et al. (Brain Research, 1971, 6-OHDA and Cathecolamine Neurons, North Holland, Amsterdam, 101-127), with minor changes. Briefly, the animals are anaesthetized with chloral hydrate (400 mg/kg, ip) and treated with desipramine (10 mpk, ip) 30 min prior to 6-OHDA injection in order to block the uptake of the toxin by the noradrenergic terminals. Then, the animals are placed in a stereotaxic frame.
- the skin over the skull is reflected and the stereotaxic coordinates ( ⁇ 2.2 posterior from bregma (AP), +1.5 lateral from bregma (ML), 7.8 ventral from dura (DV) are taken, according to the atlas of Pellegrino et al (Pellegrino L. J., Pellegrino A. S. and Cushman A. J., A Stereotaxic Atlas of the Rat Brain, 1979, New York: Plenum Press).
- a burr hole is then placed in the skull over the lesion site and a needle, attached to a Hamilton syringe, is lowered into the left MFB.
- 6-OHDA-HCl 8 ⁇ g 6-OHDA-HCl is dissolved in 4 ⁇ l of saline with 0.05% ascorbic acid as antioxidant, and infused at the constant flow rate of 1 ⁇ l/1 min using an infusion pump. The needle is withdrawn after additional 5 min and the surgical wound is closed and the animals left to recover for 2 weeks.
- rats Two weeks after the lesion the rats are administered with L-DOPA (50 mg/kg, ip) plus Benserazide (25 mg/kg, ip) and selected on the basis of the number of full contralateral turns quantified in the 2 h testing period by automated rotameters (priming test). Any rat not showing at least 200 complete turns/2 h is not included in the study.
- Selected rats receive the test drug 3 days after the priming test (maximal dopamine receptor supersensitivity).
- the new receptor antagonists are administered orally at dose levels ranging between 0.1 and 3 mg/kg at different time points (i.e., 1, 6, 12 h) before the injection of a subthreshold dose of L-DOPA (4 mpk, ip) plus benserazide (4 mpk, ip) and the evaluation of turning behavior.
- Results of the binding assay on compounds of the invention showed A 2a , Ki vaules of 0.3 to 57 nM, with preferred compounds showing Ki values between 0.3 and 5.0 nM.
- Selectivity is determined by dividing Ki for A1 receptor by Ki for A2a receptor.
- Preferred compounds of the invention have a selectivity ranging from about 100 to about 2000.
- Preferred compounds showed a 50-75% decrease in descent latency when tested orally at 1 mg/kg for anti-cataleptic activity in rats.
- inert, pharmaceutically acceptable carriers can be either solid or liquid.
- Solid form preparations include powders, tablets, dispersible granules, capsules, cachets and suppositories.
- the powders and tablets may be comprised of from about 5 to about 70 percent active ingredient.
- Suitable solid carriers are known in the art, e.g. magnesium carbonate, magnesium stearate, talc, sugar, lactose. Tablets, powders, cachets and capsules can be used as solid dosage forms suitable for oral administration.
- a low melting wax such as a mixture of fatty acid glycerides or cocoa butter is first melted, and the active ingredient is dispersed homogeneously therein as by stirring. The molten homogeneous mixture is then poured into convenient sized molds, allowed to cool and thereby solidify.
- Liquid form preparations include solutions, suspensions and emulsions. As an example may be mentioned water or water-propylene glycol solutions for parenteral injection.
- Liquid form preparations may also include solutions for intranasal administration.
- Aerosol preparations suitable for inhalation may include solutions and solids in powder form, which may be in combination with a pharmaceutically acceptable carrier, such as an inert compressed gas.
- a pharmaceutically acceptable carrier such as an inert compressed gas.
- solid form preparations which are intended to be converted, shortly before use, to liquid form preparations for either oral or parenteral administration.
- liquid forms include solutions, suspensions and emulsions.
- the compounds of the invention may also be deliverable transdermally.
- the transdermal compositions can take the form of creams, lotions, aerosols and/or emulsions and can be included in a transdermal patch of the matrix or reservoir type as are conventional in the art for this purpose.
- the compound is administered orally.
- the pharmaceutical preparation is in unit dosage form.
- the preparation is subdivided into unit doses containing appropriate quantities of the active component, e.g., an effective amount to achieve the desired purpose.
- the quantity of active compound of formula I in a unit dose of preparation may be varied or adjusted from about 0.1 mg to 1000 mg, more preferably from about 1 mg to 300 mg, according to the particular application.
- the actual dosage employed may be varied depending upon the requirements of the patient and the severity of the condition being treated. Determination of the proper dosage for a particular situation is within the skill of the art. Generally, treatment is initiated with smaller dosages which are less than the optimum dose of the compound. Thereafter, the dosage is increased by small increments until the optimum effect under the circumstances is reached. For convenience, the total daily dosage may be divided and administered in portions during the day if desired.
- a typical recommended dosage regimen for compounds of formula I is oral administration of from 10 mg to 2000 mg/day preferably 10 to 1000 mg/day, in two to four divided doses to provide relief from central nervous system diseases such as Parkinson's disease.
- the compounds are non-toxic when administered within this dosage range.
- the doses and dosage regimen of the dopaminergic agents will be determined by the attending clinician in view of the approved doses and dosage regimen in the package insert, taking into consideration the age, sex and condition of the patient and the severity of the disease. It is expected that when the combination of a compound of formula I and a dopaminergic agent is administered, lower doses of the components will be effective compared to the doses of the components administered as monotherapy.
- dosage forms which contain a compound of the invention. Those skilled in the art will recognize that dosage forms can be modified to contain both a compound of formula I and a dopaminergic agent.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- General Chemical & Material Sciences (AREA)
- Pain & Pain Management (AREA)
- Psychiatry (AREA)
- Psychology (AREA)
- Hospice & Palliative Care (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
Compounds having the structural formula I
-
- R is optionally substituted phenyl, cycloalkenyl, or heteroaryl;
- X is alkylene or —C(O)CH2—;
- Y is —N(R2)CH2CH2N(R)3)—, —OCH2CH2N(R2)—, —O—, —S—, —CH2S—, —(CH2)2—NH—, or optionally substituted
-
- m and n are 2-3, and Q is nitrogen or optionally substituted carbon; and
- Z is optionally substituted phenyl, phenylalkyl or heteroaryl, diphenylmethyl, R6—C(O)—, R6—SO2—, R6—OC(O)—, R7—N(R8)—C(O)—, R7—N(R8)—C(S)—,
-
- phenyl-CH(OH)—, or phenyl-C(═NOR2)—; or when Q is CH, phenylamino or pyridylamino; or
- Z and Y together are substituted piperidinyl or substituted phenyl; and
- R2, R3, R6, R7, and R8 are as defined in the specification are disclosed, their use in the treatment of Parkinson's disease, alone or in combination with other agents for treating Parkinson's disease, and pharmaceutical compositions comprising them; also disclosed are a processes for preparing intermediates useful for preparing compounds of formula I.
Description
This application claims the benefit of U.S. Provisional Application No. 60/207,143, filed May 26, 2000.
The present invention relates to substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine adenosine A2a receptor antagonists, the use of said compounds in the treatment of central nervous system diseases, in particular Parkinson's disease, and to pharmaceutical compositions comprising said compounds. The invention also relates to a process for preparing 5-amino-2-(substituted)pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines, intermediates useful in preparing the claimed compounds.
Adenosine is known to be an endogenous modulator of a number of physiological functions. At the cardiovascular system level, adenosine is a strong vasodilator and a cardiac depressor. On the central nervous system, adenosine induces sedative, anxiolytic and antiepileptic effects. On the respiratory system, adenosine induces bronchoconstriction. At the kidney level, it exerts a biphasic action, inducing vasoconstriction at low concentrations and vasodilation at high doses. Adenosine acts as a lipolysis inhibitor on fat cells and as an antiaggregant on platelets.
Adenosine action is mediated by the interaction with different membrane specific receptors which belong to the family of receptors coupled with G proteins. Biochemical and pharmacological studies, together with advances in molecular biology, have allowed the identification of at least four subtypes of adenosine receptors: A1, A2a, A2b and A3. A1 and A3 are high-affinity, inhibiting the activity of the enzyme adenylate cyclase, and A2a and A2b, are low-affinity, stimulating the activity of the same enzyme. Analogs of adenosine able to interact as antagonists with the A1, A2a, A2b and A3 receptors have also been identified.
Selective antagonists for the A2a receptor are of pharmacological interest because of their reduced level of side affects. In the central nervous system, A2a antagonists can have antidepressant properties and stimulate cognitive functions. Moreover, data has shown that A2a receptors are present in high density in the basal ganglia, known to be important in the control of movement. Hence, A2a antagonists can improve motor impairment due to neurodegenerative diseases such as Parkinson's disease, senile dementia as in Alzheimer's disease, and psychoses of organic origin.
Some xanthine-related compounds have been found to be A1 receptor selective antagonists, and xanthine and non-xanthine compounds have been found to have high A2a affinity with varying degrees of A2a vs. A1 selectivity. Triazolo-pyrimidine adenosine A2a receptor antagonists with different substitution at the 7-position have been disclosed previously, for example in WO 95/01356; U.S. Pat. No. 5,565,460; WO 97/05138; and WO 98/52568.
The present invention relates to compounds having the structural formula I
R is R1-furanyl, R1-pyridyl, R1-pyridyl N-oxide, R1-oxazolyl, R10-phenyl, R1-pyrrolyl or C4-C6 cycloalkenyl;
X is C2-C6 alkylene or —C(O)CH2—;
Y is —N(R2)CH2CH2N(R3)—, —OCH2CH2N(R2)—, —O—, —S—, —CH2S—, —(CH2)2—NH—, or
Z is R5-phenyl, R5-phenyl(C1-C6)alkyl, R5-heteroaryl, diphenylmethyl, R6—C(O)—, R6—SO2—, R6—OC(O)—, R7—N(R8)—C(O)—, R7—N(R8)—C(S)—,
Z and Y together are
R1 is 1 to 3 substituents independently selected from hydrogen, C1-C6-alkyl, —CF3, halogen, —NO2, —NR12R13, C1-C6 alkoxy, C1-C6 alkylthio, C1-C6 alkylsulfinyl, and C1-C6 alkylsulfonyl;
R2 and R3 are independently selected from the group consisting of hydrogen and C1-C6 alkyl;
m and n are independently 2-3;
O is
R4 is 1-2 substituents independently selected from the group consisting of hydrogen and C1-C6alkyl, or two R4 substituents on the same carbon can form ═O;
R3 is 1 to 5 substituents independently selected from the group consisting of hydrogen, halogen, C1-C6 alkyl, hydroxy, C1-C6 alkoxy, —CN, di-((C1-C6)alkyl)amino, —CF3, —OCF3, acetyl, —NO2, hydroxy(C1-C6)alkoxy, (C1-C6)-alkoxy(C1-C6)alkoxy, di-((C1-C6)-alkoxy) (C1-C6)alkoxy, (C1-C6)-alkoxy(C1-C6)alkoxy-(C1-C6)-alkoxy, carboxy(C1-C6)-alkoxy, (C1-C6)-alkoxycarbonyl (C1-C6)alkoxy, (C3-C6)cycloalkyl(C1-C6)alkoxy, di((C1-C6)alkyl)amino(C1-C6)alkoxy, morpholinyl, (C1-C6) alkyl-SO2—, (C1-C6)alkyl-SO—(C1-C6)alkoxy, tetrahydropyranyloxy, (C1-C6)alkylcarbonyl(C1-C6)alkoxy, (C1-C6)-alkoxycarbonyl, (C1-C6)alkylcarbonyloxy (C1-C6)-alkoxy, —SO2NH2, phenoxy,
or adjacent R5 substituents together are —O—CH2—O—, —O—CH2CH2—O—, —O—CF2—O— or —O—CF2CF2—O— and form a ring with the carbon atoms to which they are attached;
R6 is (C1-C6)alkyl, R5-phenyl, R5-phenyl(C1-C6)alkyl, thienyl, pyridyl, (C3-C6)-cycloalkyl, (C1-C6)alkyl-OC (O)—NH—(C1-C6)alkyl-, di((C1-C6)alkyl)aminomethyl, or
R7 is (C1-C6)alkyl, R5-phenyl or R5-phenyl(C1-C6)alkyl;
R8 is hydrogen or C1-C6 alkyl; or R7 and R8 together are —(CH2)p—A—(CH2)q, wherein p and q are independently 2 or 3 and A is a bond, —CH2—, —S— or —O—, and form a ring with the nitrogen to which they are attached;
R9 is 1-2 groups independently selected from hydrogen, C1-C6 alkyl, hydroxy, C1-C6 alkoxy, halogen, —CF3 and (C1-C6)alkoxy(C1-C6)alkoxy;
R10 is 1 to 5 substituents independently selected from the group consisting of hydrogen, halogen, C1-C6 alkyl, hydroxy, C1-C6 alkoxy, —CN, —NH2, C1-C6alkylamino, di-((C1-C6)alkyl)amino, —CF3, —OCF3 and —S(O)0-2(C1-C6)alkyl;
R11 is H, C1-C6 alkyl, phenyl, benzyl, C2-C6 alkenyl, C1-C6 alkoxy(C1-C6)alkyl, di-((C1-C6)alkyl)amino (C1-C6)alkyl, pyrrolidinyl(C1-C6)alkyl or piperidino (C1-C6)alkyl;
R12 is H or C1-C6 alkyl; and
R13 is (C1-C6)alkyl-C(O)— or (C1-C6)alkyl-SO2—.
Preferred compounds of formula I are those wherein R is R1-furanyl, R1-thienyl, R1-pyrrolyl or R10-phenyl, more preferably R1-furanyl. R1 is preferably hydrogen or halogen. Another group of preferred compounds is that wherein X is alkylene, preferably ethylene. Y is preferably
with Q preferably being nitrogen. Preferably, m and n are each 2, and R4 is H. A preferred definition for Z is R5-phenyl, R5-heteroaryl, R6—C(O)— or R6—SO2—. R5 is preferably H, halogen, alkyl, alkoxy, hydroxyalkoxy or alkoxyalkoxy. R6 is preferably R5-phenyl.
Another aspect of the invention is a pharmaceutical composition comprising a therapeutically effective amount of a compound of formula I in a pharmaceutically acceptable carrier.
Yet another aspect of the invention is a method of treating central nervous system diseases such as depression, cognitive diseases and neurodegenerative diseases such as Parkinson's disease, senile dementia or psychoses of organic origin, and stroke, comprising administering a compound of formula I to a mammal in need of such treatment. In particular, the invention is drawn to the method of treating Parkinson's disease comprising administering a compound of formula I to a mammal in need of such treatment.
Another aspect of the invention is a process for preparing 5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines of formula II, which are intermediates useful in the preparation of compounds of formula I. The process of preparing compounds of formula II
(1) treating 2-amino-4,6-dihydroxypyrimidine
(2) treating carboxaldehyde VII with a hydrazide of the formula H2N—NH—C(O)—R, wherein R is defined above, to obtain
(3) treating the intermediate of formula VII with hydrazine hydrate to form a pyrazolo ring, thus obtaining the intermediate of formula IX
(4) forming the. desired compound of formula II by dehydrative rearrangement.
A preferred aspect of the process is the dehydrative rearrangement of the intermediate of formula IX to obtain the 5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine of formula II. Preferred embodiments of the process use 2-furoic hydrazide or 2-thienoylhydrazide in step 2, thus preparing compounds of formula II wherein R is 2-furyl or 2-thienyl.
Another aspect of the invention is a process for preparing 7-bromoalkyl-5-amino-2-(R-substituted)-pyrazolo[4,3-e]-1,2,4-triazolo-[1,5-c]pyrimidines of formula IIIa, which are intermediates useful in the preparation of compounds of formula I. The process of preparing compounds of formula IIIa
(1) treating a chloride of formula VIII
(2) cyclizing the intermediate of formula X by dehydrative rearrangement to obtain the tricyclic intermediate of formula XI
(3) converting the hydroxy compound of formula XI to the bromide of formula IIIa.
Still another aspect of the invention is a method of treating Parkinson's disease with a combination of a compound of formula I and one or more agents known to be useful in the treatment of Parkinson's disease, for example dopamine; a dopaminergic agonist; an inhibitor of monoamine oxidase, type B (MAO-B); a DOPA decarboxylase inhibitor (DCI); or a catechol-O-methyltransferase (COMT) inhibitor.
Also claimed is a pharmaceutical composition comprising a compound of formula I and one or more agents known to be useful in the treatment of Parkinson's in a pharmaceutically acceptable carrier.
As used herein, the term alkyl includes straight or branched chains. Alkylene, referring to a divalent alkyl group, similarly refers to straight or branched chains. Cycloalkylene refers to a divalent cycloalkyl group. Cycloalkenyl refers to a C4-C6 cycloalkyl ring comprising one double bond.
Heteroaryl means a single ring, bicyclic or benzofused heteroaromatic group of 5 to 10 atoms comprised of 2 to 9 carbon atoms and 1 to 4 heteroatoms independently selected from the group consisting of N, O and S, provided that the rings do not include adjacent oxygen and/or sulfur atoms. N-oxides of the ring nitrogens are also included. Examples of single-ring heteroaryl groups are pyridyl, oxazolyl, isoxazolyl, oxadiazolyl, furanyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, tetrazolyl, thiazolyl, isothiazolyl, thiadiazolyl, pyrazinyl, pyrimidyl, pyridazinyl and triazolyl. Examples of bicyclic heteroaryl groups are naphthyridyl (e.g., 1, 5 or 1,7), imidazopyridyl, pyrido[2,3]imidazolyl, pyridopyrimidinyl and 7-azaindolyl. Examples of benzo-fused heteroaryl groups are indolyl, quinolyl, isoquinolyl, phthalazinyl, benzothienyl (i.e., thionaphthenyl), benzimidazolyl, benzofuranyl, benzoxazolyl and benzofurazanyl. All positional isomers are contemplated, e.g., 2-pyridyl, 3-pyridyl and 4-pyridyl. R5-substituted heteroaryl refers to such groups wherein substitutable ring carbon atoms have a substituent as defined above.
Certain compounds of the invention may exist in different stereoisomeric forms (e.g., enantiomers, diastereoisomers and atropisomers). The invention contemplates all such stereoisomers both in pure form and in mixture, including racemic mixtures.
Certain compounds will be acidic in nature, e.g. those compounds which possess a carboxyl or phenolic hydroxyl group. These compounds may form pharmaceutically acceptable salts. Examples of such salts may include sodium, potassium, calcium, aluminum, gold and silver salts. Also contemplated are salts formed with pharmaceutically acceptable amines such as ammonia, alkyl amines, hydroxyalkylamines, N-methylglucamine and the like.
Certain basic compounds also form pharmaceutically acceptable salts, e.g., acid addition salts. For example, pyrido-nitrogen atoms may form salts with strong acid, while compounds having basic substituents such as amino groups also form salts with weaker acids. Examples of suitable acids for salt formation are hydrochloric, sulfuric, phosphoric, acetic, citric, oxalic, malonic, salicylic, malic, fumaric, succinic, ascorbic, maleic, methanesulfonic and other mineral and carboxylic acids well known to those skilled in the art. The salts are prepared by contacting the free base form with a sufficient amount of the desired acid to produce a salt in the conventional manner. The free base forms may be regenerated by treating the salt with a suitable dilute aqueous base solution such as dilute aqueous NaOH, potassium carbonate, ammonia and sodium bicarbonate. The free base forms differ from their respective salt forms somewhat in certain physical properties, such as solubility in polar solvents, but the acid and base salts are otherwise equivalent to their respective free base forms for purposes of the invention.
All such acid and base salts are intended to be pharmaceutically acceptable salts within the scope of the invention and all acid and base salts are considered equivalent to the free forms of the corresponding compounds for purposes of the invention.
Compounds of formula I can be prepared by known methods from starting materials either known in the art or prepared by methods known in the art; see, for example, WO 95/01356 and J. Med. Chem., 39 (1996) 1164-1171.
Preferably, the compounds of formula I are prepared by the methods shown in the following reaction schemes. In Scheme 1, alkylation of a 5-amino-pyrazolo[4,3-c]-[1,2,4]-triazolo[1,5-c]pyrimidine of formula II is used to prepare compounds of formula I:
Starting materials of formula II can be reacted with an alkyl diol ditosylate and a base such as NaH in an inert solvent such as dimethylformamide (DMF), or with a chloro-bromo- or dibromo-alkyl compound under similar conditions, to obtain the alkyl-substituted intermediate of formula II. The compound of formula III is then reacted with an amine of the formula Z—Y—H in an inert solvent such as DMF at an elevated temperature to obtain a compound of formula Ia, i.e., a compound of formula I wherein X is alkylene.
Alternatively, staring materials of formula II can be reacted with a compound of formula Z—Y—X—Cl and a base such as NaH in an inert solvent such as DMF to obtain a mixture of a 7-substituted compound of formula I and the corresponding 8-substituted compound.
To prepare compounds of formula I wherein Y is piperazinyl and Z is R6—C(O)—, R6—SO2—, R6—OC(O)—, R7—N(R8)—C(O)— or R7—N(R8)—C(S)—, a compound of formula I wherein Z—Y is 4-t-butoxycarbonyl-1-piperazinyl is deprotected, for example by reaction with an acid such as HCl. The resultant free piperazinyl compound, IV, is treated according to procedures well known in the art to obtain the desired compounds. The following Scheme 2 summarizes such procedures:
Another method for preparing compounds of formula I is shown in Scheme 3:
In this procedure, chloropyrazolo-pyrimidine V is reacted with a compound of formula Z—Y—X—Cl in a manner similar to the alkylation procedure of Scheme 1, and the resultant intermediate is reacted with a hydrazide of formula H2N—NH—C(O)—R (or with hydrazine hydrate, followed by a compound of formula Cl—C(O)—R). The resultant hydrazide undergoes dehydrative rearrangement, e.g., by treatment with N,O-bis-(trimethylsilyl)acetamide (BSA) or a combination of BSA and hexamethyldisilazane (HMDS) and at elevated temperatures.
Starting materials are known or can be prepared by processes known in the art. However, compounds of formula II are preferably prepared by the novel process disclosed above and described in further detail here.
In the first step of the process, 2-amino-4,6-dihydroxypyrimidine (VI) is converted to the corresponding 4,6-dichloro-5-carboxaldehyde by treatment with POCl3 or SOCl2 in DMF as described in Helv. Chim. Acta, 69 (1986), 1602-1613. The reaction is carried out at an elevated temperature, preferably about 100° C., for 2 to 8 hours, preferably about 5 hours.
In the second step, 2-amino-4,6-dichloropyrimidine-5-carboxaldehyde (VII) is treated with a hydrazide of the formula H2N—NH—C(O)—R, wherein R is as defined above, to obtain the compound of formula VIII; the compound of formula VI and the hydrazide are used in a molar ratio of approximately 1:1, with a slight excess of the hydrazide being preferred. The reaction is carried out at room temperature or up to about 80° C. in a solvent such as CH3CN or DMF. The reaction time is about 16 hours (e.g., overnight).
In the third step, the compound of formula VIII is heated at 60-100° C. with 1-5 equivalents of hydrazine hydrate in a solvent such as CH3CN or DMF for 1-24 hours to obtain the compound of formula IX.
In the last step, the compound of formula IX undergoes dehydrative rearrangement by treatment with a mixture of HMDS and BSA or with BSA alone. The reaction is carried out at elevated temperatures, preferably about 120° C., for about 16 hours (e.g., overnight)
After each step of the process, the crude material is purified by conventional methods, e.g., extraction and/or recrystallization.
Compared to previously published methods for preparing the intermediate of formula II, this method proceeds in fewer steps, under milder reaction conditions and with much higher yield.
The compounds of formulas V and VII are known (Helv. Chim. Acta, 69 (1986), 1602-1613).
Another method for preparing compounds of formula I is illustrated in the following Scheme 4.
Chloride VIII is treated with a hydroxyalkyl-hydrazine in an inert solvent such as ethanol at temperatures from ambient to 100° C. to furnish derivative X. This is subjected to dehydrative cyclization, similarly to IX, such as with BSA, to provide tricyclic XI. Tricyclic XI is then converted to bromide IIIa with PBr3 at elevated temperature from 80° C. to 150° C. for 1 to 24 hours. Intermediate XI can also be converted into the tosylate analogous to IIIa by toluenesulfonyl chloride and base. Bromide IIIa is converted to compounds of formula I as described above for III.
Another method for preparing compounds of formula I is illustrated in the following Scheme 5:
In analogy to Scheme 1, chloride V is converted into alkylated compound XII, and this is further reacted with carbazate XIV, where R′ is preferably t-butyl or benzyl, to obtain derivative XIII. A solvent such as DMF may be employed at a temperature of 60-120° C. This is then reacted as in Scheme 1 to furnish XV. The R′ group is next removed, such as removal of a t-butyl group with HCl or TFA, furnishing hydrazine XVI. Acylation of XVI furnishes XVII, which is subjected to dehydrative cyclization as described above to provide desired Ia. Alternatively, XII may be reacted with a hydrazide XVIII to obtain XIX, which can be converted to XVII analogously to preparation of XV.
Using the above procedures, the following compounds were prepared.
Step 1: Stir POCl3 (84 ml, 0.9 mol) and chill to 5-10° C. while adding DMF (17.8 ml, 0.23 mol) drop-wise. Allow the mixture to warm to room temperature (RT) and add 2-amino-4,6-dihydroxypyrimidine VI (14 g, 0.11 mol) portion-wise. Heat at 100° C. for 5 h. Strip off excess POCl3 under vacuum, pour the residue into ice water, and stir overnight. Collect solids by filtration and recrystallize the dried material from a filtered ethyl acetate (EtOAc) solution to give the aldehyde, VII, m.p. 230° (dec). Mass spectrum: M+=192. PMR (DMSO): δ 8.6(δ, 2H); δ 10.1(s,1H).
Step 2: Stir a mixture of the product of Step 1 (0.38 g, 2 mmol) and 2-furoic hydrazide (0.31 g, 2.5 mmol) in CH3CN (50 ml) containing N,N-diisopropylethylamine (0.44 ml, 2.5 mmol) overnight at RT. Solvent strip the reaction mixture, and partition the residue between EtOAc and water. Dry the organic layer over MgSO4, remove the solvent, and recrystallize the residue from CH3CN to give the desired compound VIII. Mass spectrum: MH+=282.
Step 3: Add hydrazine hydrate (75 mg, 1.5 mmol) to a hot CH3CN solution of the product of Step 2 (0.14 g, 0.5 mmol). Reflux 1 h. Cool to RT and collect the yellow product IX. Mass spectrum: MH+=260.
Step 4: Heat the product of Step 3 (5.4 g, 0.021 mol) in a mixture of hexamethyl-disilazine (100 ml) and N,O-bis (trimethylsilyl) acetamide (35 ml) at 120° C. overnight. Remove volatiles under vacuum and slurry the residue in hot water to give a solid precipitate. Recrystallize from 80% aqueous acetic acid to give the title compound.
M.P.>300° C. Mass spectrum: MH+=242.
Combine the product of Preparation 1 (6.0 g, 25 mmol), ethylene glycol ditosylate (11.1 g, 30 mmol), and NaH (60% in oil,1.19 g, 30 mmol) in dry DMF (30 ml). Stir under N2 for 24 h and filter to obtain the title compound as a cream solid (PMR in DMSO: δ4.47+4.51 triplets, 8.03 s). Isolate additional material by chromatography of the filtrate.
In a similar manner to Preparation 1, but employing 2-thienoylhydrazide, prepare the title compound as a yellow solid, mass spectrum: MH+=258.
In a similar manner to Preparation 2, but using the product of Preparation 3, prepare the title compound as a yellow solid, PMR (DMSO) δ 4.49+4.54 triplets, 8.05 s.
1-(2,4-Difluorophenyl)piperazine is prepared from 2,4-difluorobromobenzcne. To the bromide (8.0 g, 41.4 mmol), piperazine (21.4 g, 249 mmol), sodium t-butoxide (5.6 g, 58 mmol) and BINAP (1.55 g, 2.5 mmol) in toluene (20 ml), add Pd2(dba)3 (0.477 g, 0.83 mmol). Heat the mixture at 110° C. under N2 for 20 h. Allow to cool and extract with 1N HCl. Basify the extract with NaOH to pH=10, extract with CH2Cl2, dry and concentrate to obtain the title compound as a brown oil.
In a similar fashion, prepare the following arylpiperazines (Me is methyl):
1-(5-Ethyl-2-pyrimidinyl)piperazine is prepared from 2-chloro-5-ethylpyrimidine. Heat the chloride (2.0 g, 14 mmol) and piperazine (3.0 g, 35 mmol) in EtOH (70 ml) at 90° C. for 2 h in a sealed vessel. Concentrate and partition between CH2Cl2 and 2N NaOH. Dry the organic with MgSO4 and concentrate. Chromatograph the crude product on silica (CH2Cl2—CH3OH) to obtain the piperazine as a yellow oil.
In a similar fashion, prepare the following piperazines from the appropriate chloride:
1-(4-Cyano-2-fluorophenyl)piperazine is prepared from 3,4-difluorobenzonitrile. Heat the nitrile (2.0 g, 14.4 mmol), piperazine (6.2 g, 72 mmol) and K2CO3 (2.4 g, 17 mmol) in toluene (10 ml) at reflux for 22 h. Allow to cool, and extract with 1N HCl. Basify with NaOH to pH=10. Extract with CH2Cl2 and wash with water and then brine. Dry the organic with MgSO4 and concentrate to give the piperazine as a white solid.
In a similar fashion, prepare the following piperazines from the appropriate fluoride (Et is ethyl):
1-(4-(2-Methoxyethoxy)phenyl)piperazine is prepared from 4-(4-hydroxy-phenyl)-1-acetylpiperazine. To NaH (60% in mineral oil, 0.79 g, 20 mmol) in DMF (25 ml) add the phenol (3.0 g, 13.6 mmol), followed by 2-bromoethyl methyl ether (2.27 g, 16.3 mmol). Stir at RT 18 h, concentrate, and partition between EtOAc and 5% citric acid. Wash the organic with 1N NaOH, then brine. Dry over MgSO4, and concentrate to obtain the alkylated product as a white solid. Heat this material (2.2 g, 7.9 mmol) in 6N HCl (30 ml) at reflux for 1 h. Allow to cool and basify to pH=10 with NaOH. Extract with CH2Cl2 and wash with water and then brine. Dry the organic with MgSO4 and concentrate to give the piperazine as a yellow oil.
In a similar fashion (except basic hydrolysis is employed for the cyclopropyl-methyl ether) prepare the following piperazines:
4-(2-Methylaminoethoxy)fluorobenzene is prepared from 4-(2-bromo-ethoxy)-fluorobenzene. Combine the bromide (1.0 g, 4.6 mmol) in CH3OH (5 ml) with CH3NH2 in CH2OH (2M, 46 ml, 92 mmol) in a sealed vessel. Heat at 60° C. for 18 h, concentrate, and partition between EtOAc and sat, NaHCO3. Wash the organic with brine, dry with MgSO4, and concentrate to obtain the amine as a yellow oil.
N-methyl-2-(4-(2-methoxyethoxy)phenoxy)ethylamine was prepared in two steps. Combine 4-(2-methoxyethoxy)phenol (1.68 g, 10.0 mmol), 1,2-dibromoethane (16.9 g, 90 mmol), and K2CO3 (2.76 g, 20 mmol) in CH3CN (20 ml) and DMF (10 ml). Heat at reflux 22 h, allow to cool, filter, and partition between ether (Et2O) and 1N NaOH. Wash with brine, dry over MgSO4, and concentrate to provide the bromoethyl ether as beige solid. Combine this (0.97 g, 3.5 mmol) with 2M CH3NH2/CH3OH (35 ml). Heat in a sealed tube (65° C., 18 h), concentrate, and partition between Et2O and 1N NaHCO3. Wash with brine, dry MgSO4, and concentrate to provide the amine as an orange oil.
1-Phenyl-2-piperazinone is prepared from 4-benzyloxycarbonyl-1-phenyl-2-piperazinone. Combine this material (1.61 g, 5.2 mmol) with 10% Pd/C (0.4 g) in EtOH (50 ml) and 1N HCl (6 ml). Hydrogenate at 45 psi for 2 h and filter. Concentrate and chromatograph the residue on silica (eluting with CH2Cl2:CH3OH:NH4OH) to obtain the piperazinone as a cream solid.
Step 1: Dissolve the product of Preparation 1, Step 2 (0.56 g, 2.0 mmol) in hot CH3CN (200 ml). Add 2-hydroxyethylhydrazine (0.51 g, 6.0 mmol). Heat at reflux 2 h and concentrate. Treat with 25 ml water and stir to give a solid. Collect and dry to give the alcohol, MS: m/e=304 (M+1).
Step 2: Heat the product of Step 1 (0.10 g, 0.33 mmol) in BSA (10 ml) for 4 h at 115° C. Concentrate in vacuo and warm with aqueous CH3OH. Collect and dry to give the cyclization product, MS: m/e=286 (M+1).
Step 3: Combine the product of Step 2 (0.285 g, 1.0 mmol) and PBr3 (2.0 ml, 21 mmol). Heat at 145° C. for 2 h, cool, and pour onto ice. Filter and dry the solid. Recrystallize from CH3OH to obtain the title compound, MS: m/e=348+350 (M+1).
Combine 5-bromo-2-furoic acid (0.50 g, 2.6 mmol) and NaHCO3 (0.44 g, 5.2 mmol) in hexane (6 ml) and water (5.2 ml). Add Selectfluor® (0.98 g, 2.8 mmol) and stir 2 h. Separate the hexane layer and dry over MgSO4 to provide a solution of 2-bromo-5-fluorofuran. Dilute with THF (6 ml) and cool to −78° C. Add 2.5M n-BuLi/hexane (4.2 ml, 11 mmol). Stir 10 min., add excess dry ice, and stir 1 h additional. Treat with 1N HCl, extract with CH2Cl2, and dry over MgSO4. Concentrate and dry to obtain the title compound as a white solid, PMR (CDCl3) δ6.70+7.28.
Combine the tosylate of Preparation 2 (0.55 g, 1.25 mmol) and 1-(2,4-difluorophenyl)piperazine (0.50 g, 2.5 mmol) in DMF (7 ml) and heat at 80° C. for 20 h. Concentrate and purify by flash column chromatography (CH2Cl2, CH3OH+NH3) to obtain the title compound as a cream solid, mass spectrum m/e=466 (M+H).
In similar fashion, prepare the following compounds:
|
|
| Ex- | MS, | |
| am- | [M + 1] | |
| ple | Z—Y— | m/e |
| 1-2 |
|
430 |
| 1-3 |
|
498, 500, 502 |
| 1-4 |
|
498 |
| 1-5 |
|
444 |
| 1-6 |
|
431 |
| 1-7 |
|
460 |
| 1-8 |
|
448 |
| 1-9 |
|
404 |
| 1-10 |
|
520 |
| 1-11 |
|
498 |
| 1-12 |
|
532, 534 |
| 1-13 |
|
478, 480 |
| 1-14 |
|
494, 496 |
| 1-15 |
|
444 |
| 1-16 |
|
478, 480 |
| 1-17 |
|
464, 466 |
| 1-18 |
|
432 |
| 1-19 |
|
468 |
| 1-20 |
|
431 |
| 1-21 |
|
472 |
| 1-22 |
|
508, 510 |
| 1-23 |
|
490 |
| 1-24 |
|
490 |
| 1-25 |
|
445 |
| 1-26 |
|
460 |
| 1-27 |
|
455 |
| 1-28 |
|
533, 535 |
| 1-29 |
|
437 |
| 1-30 |
|
481 |
| 1-31 |
|
498, 500, 502 |
| 1-32 |
|
448 |
| 1-33 |
|
432 |
| 1-34 |
|
444 |
| 1-35 |
|
474 |
| 1-36 |
|
498, 500, 502 |
| 1-37 |
|
470 |
| 1 -38 |
|
466 |
| 1-39 |
|
462 |
| 1-40 |
|
460 |
| 1-41 |
|
484 |
| 1-42 |
|
499 |
| 1 43 |
|
480 |
| 1-44 |
|
462 |
| 1-45 |
|
474 |
| 1-46 |
|
478 |
| 1-47 |
|
484 |
| 1-48 |
|
517 |
| 1-49 |
|
473 |
| 1-50 |
|
478 |
| 1-51 |
|
484 |
| 1-52 |
|
474 |
| 1-53 |
|
492 |
| 1-54 |
|
514 |
| 1-55 |
|
450 |
| 1-56 |
|
493 |
| 1-57 |
|
473 |
| 1-58 |
|
500 |
| 1-59 |
|
462 |
| 1-60 |
|
447 |
| 1-61 |
|
445 |
| 1-62 |
|
504 |
| 1-63 |
|
488 |
| 1-64 |
|
502 |
| 1-65 |
|
510 |
| 1-66 |
|
482, 484 |
| 1-67 |
|
478 |
| 1-68 |
|
534 |
| 1-69 |
|
480 |
| 1-70 |
|
496 |
| 1-71 |
|
496 |
| 1-72 |
|
444 |
| 1-73 |
|
491 |
| 1-74 |
|
578 |
| 1-75 |
|
520 |
| 1-76 |
|
522 |
| 1-77 |
|
560 |
| 1-78 |
|
500 |
| 1-79 |
|
508 |
| 1-80 |
|
532 |
| 1-81 |
|
540 |
| 1-82 |
|
468 |
| 1-83 |
|
490 |
| 1-84 |
|
572 |
| 1-85 |
|
540 |
| 1-86 |
|
522 |
| 1-87 |
|
518 |
| 1-88 |
|
529 |
| 1-89 |
|
574 |
| 1-90 |
|
572 |
| 1-91 |
|
528 |
| 1-92 |
|
572 |
| 1-93 |
|
529 |
| 1-94 |
|
522 |
| 1-95 |
|
548 |
| 1-96 |
|
548 |
| 1 -97 |
|
437 |
| 1-98 |
|
518 |
| 1-99 |
|
532 |
| 1-100 |
|
504 |
| 1-101 |
|
518 |
| 1-102 |
|
479, 501 |
| 1-103 |
|
401 |
| 1-104 |
|
454 |
| 1-105 |
|
507, 509 |
| 1-106 |
|
443 |
| 1-107 |
|
471 |
| 1-108 |
|
457 |
| 1-109 |
|
401 |
| 1-110 |
|
440 |
| 1-111 |
|
485 |
| 1-112 |
|
429 |
| 1-113 |
|
499 |
| 1-114 |
|
461 |
| 1-115 |
|
446 |
| 1-116 |
|
475 |
| 1-117 |
|
473 |
| 1-118 |
|
474 |
| 1-119 |
|
458 |
| 1-120 |
|
446 |
| 1-121 |
|
446 |
| 1-122 |
|
490 |
| 1-123 |
|
528, 530, 532 |
| 1-124 |
|
460 |
| 1-125 |
|
458 |
| 1-126 |
|
458 |
| 1-127 |
|
445 |
| 1-128 |
|
523, 525 |
| 1-129 |
|
515 |
| 1-130 |
|
518 |
| 1-131 |
|
493 |
| 1-132 |
|
548 |
| 1-133 |
|
493 |
| 1-134 |
|
530 |
| 1-135 |
|
507 |
| 1-136 |
|
487 |
| 1-137 |
|
531 |
| 1-138 |
|
487 |
| 1-139 |
|
531 |
| 1-140 |
|
494 |
| 1-141 |
|
419 |
| 1-142 |
|
461, 463 |
| 1-143 |
|
459 |
| 1-144 |
|
441 |
| 1-145 |
|
457 |
| 1-146 |
|
431 |
| 1-147 |
|
407 |
Combine the product of Preparation 1 (0.60 g, 2.5 mmol), 1,3-dibromopropane (0.60 g, 3.0 mmol), and NaH (60% in oil, 0.119 g, 3.0 mmol) in dry DMF (9 ml). Stir under N2 for 2 h, concentrate and flash chromatograph to obtain the title compound as a solid (PMR in CDCl3+CD3OD: δ 2.43 quint., 3.38+4.51 triplets, 8.09 s), as well as 8-substituted isomer.
Step 2:
Combine the product of Step 1 (0.050 g, 0.14 mmol) and 1-phenylpiperazine (0.045 g, 0.28 mmol) in DMF (2 ml) and heat at 80° C. for 4 h. Concentrate and purify by flash column chromatography (CH2Cl2, CH3OH+NH3) to obtain the title compound as a cream solid, mass spectrum m/e=443 (M+H).
Similarly prepare the following compounds:
The compound of Example 1-2 was also prepared by the following procedure:
Combine the product of Preparation 1 (0.15 g, 0.62 mmol), 1-phenyl-4-(2-chloroethyl)piperazine (0.17 g, 0.75 mmol), and NaH (60% in oil, 0.035 g, 0.87 mmol) in dry DMF (7 ml). Stir under N2 for 48 h, add additional chloride (0.03 g) and NaH (0.005 g) and stir another 72 h. Concentrate and purify by flash column chromatography (CH2Cl2, CH2OH+NH3) to obtain the title compound as a cream solid, mass spectrum m/e=429 (M+H).
The compound of Example 1-3 is similarly prepared, as are the following compounds:
Combine 1-(2,4-difluorophenyl)piperazine (1.5 g, 7.6 mmol), ethyl 2-bromopropionate (1.65 g, 9.1 mmol) and DIPEA (1.1 g, 8.3 mmol) in DMF (8 ml). Stir 4 h, concentrate, and partition between Et2O and water. Wash with brine, dry (MgSO4), and concentrate to obtain the ester as a yellow oil, NMR (CDCl3) consistent.
Step 2:
To the product of Step 1 (2.15 g, 7.2 mmol) in THF (10 ml), add LiAlH4 (1.0 M in THF, 4.4 ml, 4.4 mmol) dropwise. Heat at 60° C. 1 h, add water (0.16 ml), 15% NaOH (0.16 ml), and then water (0.49 ml). Filter and concentrate to obtain the alcohol as a yellow oil, NMR (CDCl3) consistent.
Step 3:
To the product of Step 2 (0.90 g, 3.5 mmol) in CH2Cl2 (10 ml) at 5° C., add SOCl2 (0.38 ml, 5.3 mmol). Allow to warm and stir 16 h. Concentrate and partition between CH2Cl2 and 1N NaOH, wash with water, dry (MgSO4) and concentrate to obtain the crude product as a yellow oil.
Step 4: Combine the product of Preparation 1 (0.20 g, 0.83 mmol), the product of Step 3 (0.34 g, 1.2 mmol) and NaH (60% in oil, 0.040 g, 1.0 mmol) in dry DMF (5 ml). Heat at 60° C. for 24 h, add additional chloride (0.15 g) and NaH (0.02 g), and heat another 4 h. Concentrate and purify by flash column chromatography (CH2Cl2, CH3OH+NH3) to obtain the title compound as a yellow solid, mass spectrum m/e 479 (M+H).
Similarly, prepare the following:
Using the procedure of Example 1, substituting the tosylate of Preparation 4 for the tosylate of Preparation 2, prepare the following compounds:
Step 1: To a solution of the product of Example 3-1 (4.17 g, 9.2 mmol) in CH2Cl2 (500 ml), add anhydrous HCl (120 ml of 4.0 M dioxane solution) and stir 2 h. Concentrate to dryness under vacuum and take up the residue in water. Make alkaline with aqueous NaOH and collect the precipitated de-protected product. Mass spectrum: MH+=354.
Step 2: Stir a mixture of the product of Step 1 (71 mg, 0.2 mmol) and 4-methoxy-benzoyl chloride (51 mg, 0.3 mmol) in dry DMF (10 ml) containing N,N-diisopropyl-ethylamine (52 mg, 0.4 mmol) for 6 h at RT. Pour the solution into water and collect the precipitated title compound. Mass spectrum: MH+=488.
In a similar fashion, prepare the following:
|
|
| Example | Z— | MS, [M + 1] m/e |
| 6-2 |
|
502 |
| 6-3 |
|
396 |
| 6-4 |
|
594 |
| 6-5 |
|
464 |
| 6-6 |
|
438 |
| 6-7 |
|
464 |
| 6-8 |
|
459 |
| 6-9 |
|
472 |
| 6-10 |
|
452 |
| 6-11 |
|
539 |
| 6-12 |
|
532 |
| 6-13 |
|
508 |
| 6-14 |
|
551 |
| 6-15 |
|
439 |
| 6-16 |
|
492 |
To a solution of the product of Example 6, Step 1 (53 mg, 0.15 mmol) in NMP (10 ml) add 4-chlorophenylisocyanate (25.3 mg, 0.165 mmol) at RT. Stir overnight, add an additional 25.3 mg of the isocyanate, and stir 1 h to complete conversion of all starting material. Pour into water and collect the precipitated title compound. Mass spectrum: MH+=507.
In a similar fashion, prepare the following from the appropriate isocyanate, isothiocyanate or carbamoyl chloride:
Slurry the product of Example 6, Step 1 (53 mg, 0.15 mmol) in dry DMF (20 ml) containing triethylamine (77 mg, 0.76 mmol); add 2,4-difluorobenzenesulfonyl chloride (37 μl, 0.225 mmol). Stir at RT 2 days. Pour into water and collect the precipitated title compound. Mass spectrum: M+=529.
In a similar fashion, prepare the following:
Add 4-methoxyphenyl chloroformate (56 mg, 0.3 mmol) to a slurry of the product of Example 6, Step 1 (71 mg, 0.2 mmol) in warm DMF (25 ml) containing triethylamine (101 mg, 1.0 mmol). Stir the mixture overnight at RT. Concentrate the solution to ⅓ its volume and pour into water. Collect the precipitate, wash with water, and dry in vacuo. Recrystallize from CH3OH/CH2Cl2 to give the title compound. Mass spectrum: MH+=504.
Step 1: Combine 1-bromo-2,4-difluorobenzene (1.00 g, 5.18 mmol), N,N′-dimethyl-ethylenediamine (2.74 g, 31.1 mmol), NaO-t-Bu (0.70 g, 7.2 mmol), Pd(dba)2 (0.060 g, 0.10 mmol) and (±)-BINAP (0.19 g, 0.31 mmol) in toluene (10 ml). Heat at 110° for 18 h, allow to cool, and extract with 1N HCl. Basify the aqueous solution with NaOH and extract with CH2Cl2. Dry, concentrate, and purify by PLC to give N-(2,4-difluoro-phenyl)-N,N′-dimethylethylenediamine.
Step 2: Combine the product of Preparation 2 (0.100 g, 0.23 mmol) with the product of Step 1 (0.091 g, 0.46 mmol) in DMF (2 ml). Heat at 80° for 90 h, allow to cool, concentrate, and purify by column chromatography to obtain the title compound as an oil, mass spec m/e=467.
The compound of Example 1-2 was also prepared by the following procedure.
Step 1:
To a solution of the product of Preparation 1, Step 1, (768 mg, 4 mmol) in DMF (20 ml) add N,N-diisopropylethylamine (0.88 ml, 5 mmol), followed by hydrazine hydrate (0.2 ml, 4.1 mmol). The solution warms and a solid precipitates which gradually dissolves over 1 h. After stirring 3 h, concentrate the solution under vacuum to about ⅓ its volume, and pour into water. Collect the precipitate and recrystallize it from CH3OH to give the chloropyrazolopyrimidine. Mass spectrum: MH+=170.
Step 2:
To a stirred solution of 1-phenylpiperazine (6.5 g, 40 mmol) and 50% aqueous chloroacetaldehyde (6.4 ml, 48 mmol) in CH2Cl2 (125 ml) at 5-10° C. add, portionwise, Na(OAc)3BH (12.72 g, 60 mmol). When foaming ceases, allow the mixture to warm to RT and stir for 3 h. Dilute with CH2Cl2 (100 ml), and shake with 1N aq NaOH to bring pH above 8. Wash organic layer with water and brine, dry over MgSO4, and solvent strip. Chromatograph on silica and elute with 1% CH3OH/CH2Cl2 to give the title compound. Mass spectrum: MH+=225.
Step 3:
To a slurry of 60% NaH (0.14 g, 3.5 mmol) in DMF (30 ml) at ice bath temperature add, portionwise, the product of Step 1 (0.51 g, 3 mmol). When gas evolution ceases, add the product of Step 2. Stir the resulting mixture at RT overnight. Filter off dark red insoluble matter, and concentrate the filtrate to dryness under vacuum. Triturate the gummy residue with CH3OH to give the title compound as a light yellow solid. Mass spectrum: MH+=358.
The product of Step 3 was treated as described in Preparation 1, Steps 2 and 4, to obtain the compound of Example 1-2.
Step 1: To NaH (60% in oil, 142 mg, 3.5 mmol) in DMF (15 ml) add the chloride of Example 11, Step 1 (500 mg, 2.9 mmol). Add to this 1-(2-chloroethyl)-4-(2,4-difluorophenyl) piperazine (846 mg, 3.5 mmol). Stir at RT 90 h and concentrate. Chromatograph to obtain the desired compound as a white solid. PMR in DMSO: δ2.57 (4H, s), 2.76 (2H, t), 2.85 (4H, s), 4.30 (2H, t), 7.0 (2H, m), 7.15 (1H, dxt), 7.26 (2H, s), 7.97(1H, s).
Step 2: Treat the chloride of Step 1 (37 mg, 0.095 mmol) in DMF (95 ml) with hydrazine hydrate (9.2 μl, 0.19 mmol). After 4 h, concentrate and chromatograph on PLC to obtain the hydrazine as a brown oil. Mass spectrum: MH+=390.
Step 3: Treat the hydrazine from Step 2 (18 mg, 0.047 mmol) in DMF (2 ml) with thiophene-2-carbonyl chloride (5.2 μl, 0.047 mmol) and DIPEA (12.2 μl, 0.07 mmol). After 4 h, concentrate and chromatograph on PLC to obtain the hydrazide as a yellow oil. Mass spectrum: MH+=500.
Step 4: Heat the hydrazide from Step 3 (13 mg, 0.026 mmol) in N,O-bis(trimethyl-silyl)acetamide (1 ml) for 2 h at 100° C. Concentrate and chromatograph on PLC to obtain the title compound as a white solid. Mass spectrum: MH+=482.
The 1-(2-chloroethyl)-4-(2,4-difluorophenyl)piperazine employed in this sequence is prepared in two steps. Add chloroacetyl chloride (1.76 ml, 22.1 mmol) and N-methylmorpholine (2.65 ml, 24.1 mmol) to 1-(2,4-difluorophenyl)piperazine (3.98 g, 20.1 mmol) in CH2Cl2 (15 ml) at 0° C. Stir at RT 1 h, concentrate, partition EtOAc-water, dry, and concentrate to obtain the amide as a brown oil. To a 0° C. solution of this (4.71 g, 17.1 mmol) in THF (25 ml) add dropwise BH3●CH3S/THF (2M, 12.8 ml, 25.6 mmol). Stir at RT overnight, quench with CH3OH, concentrate, and partition with CH2Cl2-water. Dry and concentrate the organic layer. Treat the crude product a second time with BH3●CH3S/THF and work up as above to provide the chloroethylpiperazine as a brown oil.
Step 1: To NaH (2.14 g, 60% in oil, 53 mmol) in DMF (20 ml), add the product of Example 11, Step 1 (7.55 g, 45 mmol). Add 1-bromo-2-chloroethane (14.8 ml, 178 mmol). Stir 1.5 h and concentrate. Chromatograph to give the dichloride as a white solid.
Step 2: In the product of Step 1 (3.7 g, 16 mmol) in DMF (20 ml) add 1-butyl carbazate (2.53 g, 19 mmole). Heat at 80° C. for 18 h and concentrate. Chromatograph to obtain the carbazate as a while solid.
Step 3: To the product of Step 2 (3.16 g, 9.6 mmol) and KI (1.6 g, 9.6 mmol) in DMF (25 ml) add 1-(2,4-difluorophenyl)piperazine (3.82 g, 19 mmol). Heat at 90° C. for 68 h and concentrate. Chromatograph to obtain the piperazine as a brown solid.
Step 4: Dissolve the product of Step 3 (3.38 g, 6.9 mmol) in 1:1 CH3OH—CH2Cl2 (50 ml). Add 4M HCl in dioxane (20 ml). Stir 16 h and add aq. NH3 to pH 11-12. Concentrate and chromatograph to obtain the hydrazine as a yellow solid.
Step 5: Combine the product of Step 4 (0.120 g, 0.31 mmol) with 5-bromo-2-furoic acid (0.071 g, 0.37 mmol) and HOBt.H2O (0.050 g, 0.37 mmol) in DMF (6 ml). Add EDCl (0.071 g, 0.37 mmol) and stir 1 h. Concentrate and chromatograph to obtain the hydrazide as a yellow solid.
Step 6: Dissolve the product of Step 5 (0.163 g, 0.28 mmol) in N,O-bis(trimethylsilyl) acetamide (6 ml). Heat at 120° C. for 16 h and pour into CH3OH. Concentrate and chromatograph to obtain the title product as an off-white solid: MS m/e 544+546 (M+1).
Similarly prepare compounds of the following structure, wherein R is as defined in the table:
|
|
| Example | R | MS m/e |
| 13-2 |
|
480 |
| 13-3 |
|
500, 502 |
| 13-4 |
|
465 |
| 13-5 |
|
479 |
| 13-6 |
|
544, 546 |
| 13-7 |
|
466 |
| 13-8 |
|
512 |
| 13-9 |
|
476 |
| 13-10 |
|
484 |
Treat the product of Example 13, Step 4 (0.080 g, 0.20 mmol) with nicotinoyl chloride hydrochloride (0.044 g, 0.25 mmol) and diisopropylethylamine (0.086 ml, 0.49 mmol) in DMF (4 ml). Stir 2 h, concentrate and chromatograph to obtain the hydrazide as a white solid.
Treat this material with BSA as in Example 13, Step 6 to obtain the title compound as a white solid: MS m/e 477 (M+1).
Similarly prepare compounds of the following structure, wherein R is as defined in the table:
Step 1: To the product of Example 13, Step 2 (3.54 g, 10.8 mmol) and KI (1.79 g, 10.8 mmol) in DMF (35 ml) add 1-(4-(2-methoxyethoxy)phenyl)piperazine (5.1 g, 22 mmol). Heat at 90° C. for 90 h and concentrate. Chromatograph to obtain the piperazine as a brown solid.
Step 2: Treat the product of Step 1 with HCl as in Example 13, Step 4, to obtain the hydrazine as a yellow solid.
Step 3: Treat the product of Step 2 with 5-chloro-2-furoic acid as in Example 13, Step 5, to obtain the hydrazide as a yellow solid.
Step 4: Treat the product of Step 3 with BSA as in Example 13, Step 6. Chromatograph to obtain the title compound as a white solid, MS m/e 538+540 (M+1).
Similarly prepare compounds of the following structure, wherein R is as defined in the table:
Combine the product of Example 1-83 (0.080 g, 0.16 mmol) with Ac2O (0.028 ml, 0.28 mmol) and 4-dimethylaminopyridine (0.004 g, 0.03 mmol) in DMF (5 ml). Stir 4 h, concentrate, and chromatograph to obtain the acetate ester as a white solid, MS: m/e=532 (M+1).
Combine the product of Example 1-21 (0.100 g, 0.21 mmol) with H2NHOH●HCl 0.029 g, 0.42 mmol) in 95% EtOH (9 ml). Add 10 drops conc. HCl, heat at reflux 5 h, add DMF (1.5 ml), heat 18 h, allow to cool, and filter to obtain the oxime as a white solid, MS: m/e=487 (M+1). Chromatograph the mother liquor to obtain additional product.
Similarly prepare the methoxime, a white solid, MS: m/e=501 (M+1):
Step 1: To a solution of 4-bromophenethyl alcohol (0.600 g, 2.98 mmol) and 3-pyridinylboronic acid (0.734 g, 5.97 mmol) in toluene (35 ml) and EtOH (9 ml), add a solution of K2CO3 (0.8826 g, 5.97 mmol) in H2O (16 ml) and tetrakis(triphenyl-phosphine)palladium(0) (0.172 g, 0.149 mmol). Heat in a sealed tube 18 h at 120° C. and cool. Extract with EtOAc, wash with brine, dry (K2CO3) and concentrate. Chromatograph on silica (30-50% EtOAc/hexanes) to obtain the biaryl alcohol.
Step 2: To the product of Step 1 (0.540 g, 2.71 mmol) in CH2Cl2 (15 ml) at 0° C. add mesyl chloride (0.35 ml, 3.52 mmol) and Et3N (0.57 ml, 4.00 mmol). Stir 2.5 h and extract with CH2Cl2. Dry (Na2SO4) and concentrate to obtain the mesylate.
Step 3: Add the product of Preparation 4 (0.347 g, 1.44 mmol) to the mesylate of Step 2 (0.480 g, 1.73 mmol) in DMF (4.5 ml), followed by NaH (60%. in oil, 0.082 g, 4.04 mmol). Stir 18 h and extract with EtOAc. Wash with H2O dry (K2CO3) and concentrate. Purify by PTLC (5% CH3OH/CH2Cl2, developed twice) to obtain the title compound as a white solid, MS: 423 (M+1).
By the above method, prepare the following (Example 18-8 from commercial biphenylethanol):
Step 1: Combine 4-bromophenethyl alcohol (3.00 g, 14.9 mmol), triethylamine (2.68 ml, 19.2 mmol), dimethylaminopyridine (0.180 g, 1.47 mmol) and t-butyidimethylsilyl chloride (2.45 g, 16.3 mmol) in CH2Cl2 (75 ml). Stir 1 h, wash with H2O, dry (K2CO3), and concentrate. Chromatograph on silica (hexanes) to obtain the silyl ether.
Step 2: To the compound of Step 1 (0.300 g, 0.95 mmol) in dry toluene (15 ml) add 2-(tri-butylstannyl)pyridine (1.05 g, 2.86 mmol) and tetrakis(triphenylphosphine)-palladium (0.11 g, 0.095 mmol). Flush with N2 and heat 16 h at 120° C. Cool, filter through Celite, and wash with NH4Cl, brine and then water. Dry (K2CO3) and concentrate. Chromatograph on silica (3-5% EtOAc/hexanes) to obtain the biaryl, MS 314 (M+1).
Step 3: Combine the biaryl of Step 2 (0.180 g, 0.57 mmol) and TBAF (1.0 M in THF, 1.7 ml) in THF (5.7 ml). Stir 2 h, wash with saturated NH4Cl, and extract with EtOAc. Wash with H2O several times, dry (K2CO3) and concentrate to obtain the alcohol. Steps 4 and 5: Conduct as in Example 18, Steps 2 and 3, to obtain the title compound as a white solid, MS: 423 (M+1).
Similarly prepare the following compounds:
To the product of Example 18 (0.055 g, 0.13 mmol) in CH2Cl2 (1.5 ml) at −78° C. add m-CPBA (0.050 g, 0.29 mmol). Allow to warm, stir 5 h, and wash successively with sat. Na2S2O3, 5% K2CO3, and H2O. Dry (Na2SO4) and concentrate. Purify by PTLC (10% CH3OH/CH2Cl2) to obtain the title compound, MS: 439 (M+1).
Similarly, oxidize the product of Example 18-2 at 0° C. or RT to produce the sulfoxide, MS: 484 (M+1), or the sulfone, MS: 500 (M+1).
Combine the product of Preparation 6 (0.104 g, 0.30 mmol), 4-methyl-benzenethiol (0.075 g, 0.60 mmol), and K2CO3 (0.091 g, 0.66 mmol) in DMF (20 ml). Heat at 80° C. for 5 h and concentrate. Partition between EtOAc and water, wash with brine, dry over MgSO4 and concentrate. Recrystallize from CH3OH to obtain the title compound, MS: m/e=392 (M+1).
Similarly prepare the following compounds:
Combine the product of Preparation 6 (0.11 g, 0.25 mmol), 3,4-dimethoxy-phenol (0.154 g, 1.0 mmol), and K2CO3 (0.138 g, 1.0 mmol) in DMF (5 ml). Heat at 90° C. for 48 h and concentrate. Partition between EtOAc and water, wash with 1 N NaOH and then brine, dry over MgSO4, and concentrate. Chromatograph on silica (1.5% CH3OH/CH2Cl2) to obtain the title compound, MS: m/e=422 (M+1).
Similarly prepare the following compound, MS: m/e=454 (M+1).
Step 1: To NaH (60% in oil,1.32 g, 33 mmol) in DMF (25 ml) at 5° C. add dropwise, with stirring, 3,4-dimethoxyphenol (4.77 g, 30 mmol). After 0.5 h, add 1,5-dibromo-pentane (20.7 g, 90 mmol). Stir 2 h and concentrate. Chromatograph on silica (CH2Cl2) to obtain the monobromide, MS: m/e=303 (M+1).
Step 2: To NaH (60% in oil, 0.044 g, 1.1 mmol) in DMF (25 ml) at 5° C. add the product of Preparation 1 (0.241 g, 1.1 mmol). After 0.5 h, add the compound from Step 1. Allow to warm, stir 18 h, and concentrate. Partition between EtOAc and water, wash with 1N NaOH and then brine, dry over MgSO4, and concentrate. Chromatograph on silica (2% CH3OH/CH2Cl2) and recrystallize the appropriate fraction from CH3CN to obtain the title compound, MS: m/e=464 (M+1).
Step 1: Combine 1,4-dioxa-8-azaspiro(4,5)decane (0.48 ml, 3.8 mmol) with the product of Preparation 2 (0.66 g, 1.5 mmol) in DMF (10 ml). Heat at 90° C. for 16 h, allow to cool, filter and wash with CH3OH to give off-white solid, MS: m/e 411 (M+1).
Step 2: Heat the product of Step 1 (0.476 g, 1.16 mmol) in acetone (10 ml) and 5% HCl (10 ml) at 100° C. for 16 h. Cool, neutralize with sat. NaHCO3, and extract with 10% CH3OH in CH2Cl2. Dry (MgSO4), concentrate and chromatograph on silica with CH3OH—CH2Cl2 to obtain the ketone as a white powder, MS: m/e 367 (M+1).
Step 3: Combine the product of Step 2 (0.050 g, 0.13 mmol) with O-methylhydroxyl-amine hydrochloride (0.033 g, 0.39 mmol) in pyridine (3 ml). Stir for 16 h and concentrate. Partition between NaHCO3 (sat.) and 5% CH3OH in CH2Cl2. Dry (MgSO4), concentrate and chromatograph on silica with 5% CH3OH—CH2Cl2 to obtain the title compound as a white solid, MS: m/e 396 (M+1).
Similarly prepare the following compounds:
|
|
| Example | R11 | MS, m/e |
| 24-2 | —CH2CH3 | 410 |
| 24-3 | —CH2CH2CH3 | 424 |
| 24-4 | —CH2CHCH2 | 422 |
| 24-5 | —C(CH3)3 | 438 |
| 24-3 | —C6H5 | 458 |
| 24-4 | —CH2C6H5 | 472 |
Step 1: Combine benzyl 4-oxo-1-piperidinecarboxylate (1.0 g, 4.3 mmol) with H2NOH.HCl (0.89 g, 13 mmol) in pyridine (5 ml). Stir 16 h and concentrate. Partition between NaHCO3 (sat.) and EtOAc, dry (MgSO4) and concentrate to give the oxime.
Step 2: Combine the product of Step 1 (0.44 g, 1.8 mmol) with 2-bromoethyl methyl ether (0.20 ml, 2.2 mmol) and NaH (0.10 g, 2.7 mmol) in DMF (8 ml). Stir 16 h and concentrate. Partition between NH4Cl (sat.) and ether, dry (MgSO4), and concentrate. Chromatograph the residue on silica with 20% EtOAc-hexane to obtain the alkylated oxime.
Step 3: Stir the product of Step 2 (0.45 g, 1.47 mmol) over 5% Pd/C (0.045 g) in EtOAc (25 ml) under H2 for 6 h. Filter and concentrate to obtain the amine.
Step 4: Treat the amine of Step 3 with the product of Preparation 2 as in Example 24, Step 1, to obtain the title compound as a white solid, MS: m/e 440 (M+1).
Add sodium triacetoxyborohydride (0.083 g, 0.39 mmol) to a mixture of the product of Example 24, Step 2 (0.050 g, 0.13 mmol), aniline (0.035 ml, 0.39 mmol), and AcOH (0.045 ml, 0.78 mmol) in dichloroethane (3 ml). Stir 16 h and partition between NaHCO3 (sat.) and 5% CH3OH in CH2Cl2. Dry (MgSO4) and concentrate. Chromatograph (5% CH3OH—CH2Cl2) to obtain the title compound as a white solid, MS: m/e 444 (M+1).
In similar fashion, prepare the following compound, MS: m/e 445 (M+1).
Step 1: Combine 4-bromophenol (3.46 g, 20.0 mmol) with 2-bromoethyl methyl ether (2.82 ml, 30.0 mmol) and K2CO3 (8.30 g, 60.0 mmol) in acetone (50 ml). Heat at reflux 16 h, cool, filter, and concentrate. Chromatograph on silica with 5% EtOAc/hexane to give the ether as a clear oil. To this ether (2.73 g, 11.8 mmol) in dry THF (50 ml) at −78° C. add n-BuLi (1.6 M in hexane, 7.4 ml, 11.8 mmol). Stir for 10 min. and add a solution of benzyl 4-oxo-1-piperidinecarboxylate (2.5 g, 10.7 mmol) in dry THF (5 ml). Stir for 2 h and allow to warm. Partition between sat. NH4Cl and EtOAc, dry (MgSO4) and concentrate. Chromatograph on silica with EtOAc/hexane (20:80, then 40:60) to obtain the alcohol.
Step 2: To a solution of the product of Step 1 (0.386 g, 1.0 mmol) and triethylsilane (0.80 ml, 5.0 mmol) in dry CH2Cl2 (10 ml) at −78° C. add trifluoroacetic acid (0.38 ml, 5.0 mmol). Allow to warm over 2 h and partition between sat. NaHCO3 and CH2Cl2. Dry (MgSO4) and concentrate. Chromatograph on silica with 20% EtOAc/hexane to obtain the reduction product, MS: m/e 370 (M+1).
Step 3: Stir the product of Step 2 (0.300 g, 0.758 mmol) over 5% Pd/C (0.030 g) in EtOAc (5 ml) and CH3OH (5 ml) under H2 for 2 h. Filter and concentrate to obtain the amine.
Step 4: Treat the amine of Step 3 with the product of Preparation 2 as in Example 24, Step 1, to obtain the title compound as a white solid, MS: m/e 503 (M+1).
Treat the product of Example 1-145 (0.020 g, 0.044 mmol) in EtOH (0.5 ml) at 0° C. with sodium borohydride (0.005 g, 0.13 mmol) and with an equal amount again after 0.75 h. After another 0.75 h, partition between CH2Cl2 and sat. NH4Cl. Dry (Na2SO4) and concentrate. Purify by PTLC (10% CH3OH/CH2Cl2) to obtain the title compound as a white solid, MS: 459 (M+1).
Treat the product of Example 1-145 (0.020 g, 0.044 mmol) in pyridine (0.5 ml) with methoxyamine hydrochloride (0.011 g, 0.13 mmol). Stir 16 h and concentrate. Partition between CH2Cl2 and sat. NaHCO3. Dry (Na2SO4) and concentrate. Purify by PTLC (5% CH3OH/CH2Cl2) to obtain the title compound as a white solid, MS: 486 (M+1).
Similarly, prepare the oxime 29-2 as two separated geometric isomers, each a white solid, MS: 472 (M+1).
Because of their adenosine A2a receptor antagonist activity, compounds of the present invention are useful in the treatment of depression, cognitive function diseases and neurodegenerative diseases such as Parkinson's disease, senile dementia as in Alzheimer's disease, and psychoses of organic origin. In particular, the compounds of the present invention can improve motor-impairment due to neurodegenerative diseases such as Parkinson's disease.
The other agents known to be useful in the treatment of Parkinson's disease which can be administered in combination with the compounds of formula I include: L-DOPA; dopaminergic agonists such as quinpirole, ropinirole, pramipexole, pergolide and bromocriptine; MAO-B inhibitors such as deprenyl and selegiline; DOPA decarboxylase inhibitors such as carbidopa and benserazide; and COMT inhibitors such as tolcapone and entacapone. One to three other agents can be used in combination with the compounds of formula I, preferably one.
The pharmacological activity of the compounds of the invention was determined by the following in vitro and in vivo assays to measure A2a receptor activity.
Human Adenosine A2a and A1 Receptor Competition Binding Assay Protocol
Membrane sources:
A2a: Human A2a Adenosine Receptor membranes, Catalog #RB-HA2a, Receptor Biology, Inc., Beltsville, Md. Dilute to 17 μg/100 μl in membrane dilution buffer (see below).
Assay Buffers:
Membrane dilution buffer: Dulbecco's Phosphate Buffered Saline (Gibco/BRL)+10 mM MgCl2.
Compound Dilution Buffer: Dulbecco's Phosphate Buffered Saline (Gibco/BRL)+10 mM MgCl2 supplemented with 1.6 mg/ml methyl cellulose and 16% DMSO. Prepared fresh daily.
Ligands:
A2a: [3H]-SCH 58261, custom synthesis, AmershamPharmacia Biotech, Piscataway, N.J. Stock is prepared at 1 nM in membrane dilution buffer. Final assay concentration is 0.5 nM.
A1: [3H]-DPCPX, AmershamPharmacia Biotech, Piscataway, N.J. Stock is prepared at 2 nM in membrane dilution buffer. Final assay concentration is 1 nM.
Non-specific Binding:
A2a: To determine non-specific binding, add 100 nM CGS 15923 (RBI, Natick, Mass.). Working stock is prepared at 400 nM in compound dilution buffer.
A1: To determine non-specific binding, add 100 μM NECA (RBI, Natick, Mass). Working stock is prepared at 400 μM in compound dilution buffer.
Compound Dilution:
Prepare 1 mM stock solutions of compounds in 100% DMSO. Dilute in compound dilution buffer. Test at 10 concentrations ranging from 3 μM to 30 pM. Prepare working solutions at 4× final concentration in compound dilution buffer.
Assay procedure:
Perform assays in deep well 96 well plates. Total assay volume is 200 μl. Add 50 μl compound dilution buffer (total ligand binding) or 50 μl CGS 15923 working solution (A2a non-specific binding) or 50 μl NECA working solution (A1 non-specific binding) or 50 μl of drug working solution. Add 50 μl ligand stock ([3H]-SCH 58261 for A2a, [3H]-DPCPX for A1). Add 100 μl of diluted membranes containing the appropriate receptor. Mix. Incubate at room temperature for 90 minutes. Harvest using a Brandel cell harvester onto Packard GF/B filter plates. Add 45 μl Microscint 20 (Packard), and count using the Packard TopCount Microscintillation Counter. Determine IC50 values by fitting the displacement curves using an iterative curve fitting program (Excel). Determine Ki values using the Cheng-Prusoff equation.
Haloperidol-induced catalepsy in the rat
Male Sprague-Dawley rats (Charles River, Calco, Italy) weighing 175-200 g are used. The cataleptic state is induced by the subcutaneous administration of the dopamine receptor antagonist haloperidol (1 mg/kg, sc), 90 min before testing the animals on the vertical grid test. this test, the rats are placed on the wire mesh cover of a 25×43 plexiglass cage placed at an angle of about 70 degrees with the bench table. The rat is placed on the grid with all four legs abducted and extended (“frog posture”). The use of such an unnatural posture is essential for the specificity of this test for catalepsy. The time span from placement of the paws until the first complete removal of one paw (decent latency) is measured maximally for 120 sec.
The selective A2A adenosine antagonists under evaluation are administered orally at doses ranging between 0.03 and 3 mg/kg, 1 and 4 h before scoring the animals.
In separate experiments, the anticataleptic effects of the reference compound, L-DOPA (25, 50 and 100 mg/kg, ip), were determined.
6-OHDA Lesion of the Middle Forebrain Bundle in Rats
Adult male Sprague-Dowley rats (Charles River, Calco, Como, Italy), weighing 275-300 g, are used in all experiments. The rats are housed in groups of 4 per cage, with free access to food and water, under controlled temperature and 12 hour light/dark cycle. The day before the surgery the rats are fasted over night with water ad libitum.
Unilateral 6-hydroxydopamine (6-OHDA) lesion of the middle forebrain bundle is performed according to the method described by Ungerstedt et al. (Brain Research, 1971, 6-OHDA and Cathecolamine Neurons, North Holland, Amsterdam, 101-127), with minor changes. Briefly, the animals are anaesthetized with chloral hydrate (400 mg/kg, ip) and treated with desipramine (10 mpk, ip) 30 min prior to 6-OHDA injection in order to block the uptake of the toxin by the noradrenergic terminals. Then, the animals are placed in a stereotaxic frame. The skin over the skull is reflected and the stereotaxic coordinates (−2.2 posterior from bregma (AP), +1.5 lateral from bregma (ML), 7.8 ventral from dura (DV) are taken, according to the atlas of Pellegrino et al (Pellegrino L. J., Pellegrino A. S. and Cushman A. J., A Stereotaxic Atlas of the Rat Brain, 1979, New York: Plenum Press). A burr hole is then placed in the skull over the lesion site and a needle, attached to a Hamilton syringe, is lowered into the left MFB. Then 8 μg 6-OHDA-HCl is dissolved in 4 μl of saline with 0.05% ascorbic acid as antioxidant, and infused at the constant flow rate of 1 μl/1 min using an infusion pump. The needle is withdrawn after additional 5 min and the surgical wound is closed and the animals left to recover for 2 weeks.
Two weeks after the lesion the rats are administered with L-DOPA (50 mg/kg, ip) plus Benserazide (25 mg/kg, ip) and selected on the basis of the number of full contralateral turns quantified in the 2 h testing period by automated rotameters (priming test). Any rat not showing at least 200 complete turns/2 h is not included in the study.
Selected rats receive the test drug 3 days after the priming test (maximal dopamine receptor supersensitivity). The new receptor antagonists are administered orally at dose levels ranging between 0.1 and 3 mg/kg at different time points (i.e., 1, 6, 12 h) before the injection of a subthreshold dose of L-DOPA (4 mpk, ip) plus benserazide (4 mpk, ip) and the evaluation of turning behavior.
Using the above test procedures, the following results were obtained for preferred and/or representative compounds of the invention.
Results of the binding assay on compounds of the invention showed A2a, Ki vaules of 0.3 to 57 nM, with preferred compounds showing Ki values between 0.3 and 5.0 nM.
Selectivity is determined by dividing Ki for A1 receptor by Ki for A2a receptor. Preferred compounds of the invention have a selectivity ranging from about 100 to about 2000.
Preferred compounds showed a 50-75% decrease in descent latency when tested orally at 1 mg/kg for anti-cataleptic activity in rats.
In the 6-OHDA lesion test, rats dosed orally with 1 mg/kg of the preferred compounds performed 170-440 turns in the two-hour assay period.
In the haloperidol-induced catalepsy test, a combination of sub-threshold amount of a compound of formula I and a sub-threshold amount of L-DOPA showed a significant inhibition of the catalepsy, indicating a synergistic effect. In the 6-OHDA lesion test, test animals administered a combination of a compound of formula I and a sub-threshold amount of L-DOPA demonstrated significantly higher contralateral turning.
For preparing pharmaceutical compositions from the compounds described by this invention, inert, pharmaceutically acceptable carriers can be either solid or liquid. Solid form preparations include powders, tablets, dispersible granules, capsules, cachets and suppositories. The powders and tablets may be comprised of from about 5 to about 70 percent active ingredient. Suitable solid carriers are known in the art, e.g. magnesium carbonate, magnesium stearate, talc, sugar, lactose. Tablets, powders, cachets and capsules can be used as solid dosage forms suitable for oral administration.
For preparing suppositories, a low melting wax such as a mixture of fatty acid glycerides or cocoa butter is first melted, and the active ingredient is dispersed homogeneously therein as by stirring. The molten homogeneous mixture is then poured into convenient sized molds, allowed to cool and thereby solidify.
Liquid form preparations include solutions, suspensions and emulsions. As an example may be mentioned water or water-propylene glycol solutions for parenteral injection.
Liquid form preparations may also include solutions for intranasal administration.
Aerosol preparations suitable for inhalation may include solutions and solids in powder form, which may be in combination with a pharmaceutically acceptable carrier, such as an inert compressed gas.
Also included are solid form preparations which are intended to be converted, shortly before use, to liquid form preparations for either oral or parenteral administration. Such liquid forms include solutions, suspensions and emulsions.
The compounds of the invention may also be deliverable transdermally. The transdermal compositions can take the form of creams, lotions, aerosols and/or emulsions and can be included in a transdermal patch of the matrix or reservoir type as are conventional in the art for this purpose.
Preferably the compound is administered orally.
Preferably, the pharmaceutical preparation is in unit dosage form. In such form, the preparation is subdivided into unit doses containing appropriate quantities of the active component, e.g., an effective amount to achieve the desired purpose.
The quantity of active compound of formula I in a unit dose of preparation may be varied or adjusted from about 0.1 mg to 1000 mg, more preferably from about 1 mg to 300 mg, according to the particular application.
The actual dosage employed may be varied depending upon the requirements of the patient and the severity of the condition being treated. Determination of the proper dosage for a particular situation is within the skill of the art. Generally, treatment is initiated with smaller dosages which are less than the optimum dose of the compound. Thereafter, the dosage is increased by small increments until the optimum effect under the circumstances is reached. For convenience, the total daily dosage may be divided and administered in portions during the day if desired.
The amount and frequency of administration of the compounds of the invention and the pharmaceutically acceptable salts thereof will be regulated according to the judgment of the attending clinician considering such factors as age, condition and size of the patient as well as severity of the symptoms being treated. A typical recommended dosage regimen for compounds of formula I is oral administration of from 10 mg to 2000 mg/day preferably 10 to 1000 mg/day, in two to four divided doses to provide relief from central nervous system diseases such as Parkinson's disease. The compounds are non-toxic when administered within this dosage range.
The doses and dosage regimen of the dopaminergic agents will be determined by the attending clinician in view of the approved doses and dosage regimen in the package insert, taking into consideration the age, sex and condition of the patient and the severity of the disease. It is expected that when the combination of a compound of formula I and a dopaminergic agent is administered, lower doses of the components will be effective compared to the doses of the components administered as monotherapy.
The following are examples of pharmaceutical dosage forms which contain a compound of the invention. Those skilled in the art will recognize that dosage forms can be modified to contain both a compound of formula I and a dopaminergic agent.
The scope of the invention in its pharmaceutical composition aspect is not to be limited by the examples provided.
| No. | Ingredients | mg/tablet | mg/tablet |
| 1. | Active compound | 100 | 500 |
| 2. | Lactose USP | 122 | 113 |
| 3. | Corn Starch, Food Grade, as a | 30 | 40 |
| 10% paste in Purified Water | |||
| 4. | Corn Starch, Food Grade | 45 | 40 |
| 5. | Magnesium Stearate | 3 | 7 |
| Total | 300 | 700 | |
Mix Item Nos. 1 and 2 in a suitable mixer for 10-15 minutes. Granulate the mixture with Item No. 3. Mill the damp granules through a coarse screen (e.g., ¼″, 0.63 cm) if necessary. Dry the damp granules. Screen the dried granules if necessary and mix with Item No. 4 and mix for 10-15 minutes. Add Item No. 5 and mix for 1-3 minutes. Compress the mixture to appropriate size and weigh on a suitable tablet machine.
| No. | Ingredient | mg/capsule | mg/capsule |
| 1. | Active compound | 100 | 500 |
| 2. | Lactose USP | 106 | 123 |
| 3. | Corn Starch, Food Grade | 40 | 70 |
| 4. | Magnesium Stearate NF | 7 | 7 |
| Total | 253 | 700 | |
Mix Item Nos. 1, 2 and 3 in a suitable blender for 10-15 minutes. Add Item No. 4 and mix for 1-3 minutes. Fill the mixture into suitable two-piece hard gelatin capsules on a suitable encapsulating machine.
While the present invention has been described in conjunction with the specific embodiments set forth above, many alternatives, modifications and variations thereof will be apparent to those of ordinary skill in the art. All such alternatives, modifications and variations are intended to fall within the spirit and scope of the present invention.
Claims (21)
1. A compound having the structural formula
or a pharmaceutically acceptable salt thereof, wherein
R is R1-furanyl, R1-thienyl, R1-pyridyl, R1-pyridyl N-oxide, R1-oxazolyl, R10-phenyl, R1-pyrrolyl or C4-C6 cycloalkenyl;
X is C2-C6 alkylene or —C(O)CH2—;
Y is —N(R2)CH2CH2N(R3)—, —OCH2CH2N(R2)—, —O—, —S—, —CH2S—, —(CH2)2—NH—, or
and
Z is R5-phenyl, R5-phenyl(C1-C6)alkyl, R5-heteroaryl, diphenylmethyl, R6—C(O)—, R6—SO2—, R6—OC(O)—, R7—N(R8)—C(O)—, R7—N(R8)—C(S)—,
Z and Y together are
R1 is 1 to 3 substituents independently selected from hydrogen, C1-C6-alkyl, —CF3, halogen, —NO2, —NR12R13, C1-C6 alkoxy, C1-C6 alkylthio, C1-C6 alkylsulfinyl, and C1-C6 alkylsulfonyl;
R2 and R3 are independently selected from the group consisting of hydrogen and C1-C6 alkyl;
m and n are independently 2-3;
Q is
R1 is 1-2 substituents independently selected from the group consisting of hydrogen and C1-C6alkyl, or two R4 substituents on the same carbon can form ═O;
R5 is 1 to 5 substituents independently selected from the group consisting of hydrogen, halogen, C1-C6 alkyl, hydroxy, C1-C6 alkoxy, —CN, di-((C1-C6)alkyl)amino, —CF3, —OCF3, acetyl, —NO2, hydroxy(C1-C6)alkoxy, (C1-C6)-alkoxy(C1-C6)alkoxy, di-((C1-C6)-alkoxy)(C1-C6)alkoxy, (C1-C6)-alkoxy(C1-C6)alkoxy-(C1-C6)-alkoxy, carboxy(C1-C6)-alkoxy, (C1-C6)-alkoxycarbonyl(C1-C6)alkoxy, (C3-C6)cycloalkyl(C1-C6)alkoxy, di-((C1-C6)alkyl)amino(C1-C6)alkoxy, morpholinyl, (C1-C6)alkyl-SO2-, (C1-C6)alkyl-SO_-(C1-C6)alkoxy, tetrahydropyranyloxy, (C1-C6)alkylcarbonyl(C1-C6)-alkoxy, (C1-C6)-alkoxycarbonyl, (C1-C6)alkylcarbonyloxy(C1-C6)-alkoxy, —SO2NH2, phenoxy,
or adjacent R5 substituents together are —O—CH2—O—, —O—CH2CH2—O—, —O—CF2—O— or —O—CF2CF2—O— and form a ring with the carbon atoms to which they are attached;
R6 is (C1-C6)alkyl, R5-phenyl, R5-phenyl(C1-C6)alkyl, thienyl, pyridyl, (C3-C6)-cycloalkyl, (C1-C6)alkyl-OC(O)-NH-(C1-C6)alkyl-, di-((C1-C6)alkyl)aminomethyl, or
R7 is (C1-C6)alkyl, R5-phenyl or R5-phenyl(C1-C6)alkyl;
R8 is hydrogen or C1-C6 alkyl; or R7 and R8 together are —(CH2)p—A—(CH2)q, wherein p and q are independently 2 or 3 and A is a bond, —CH2—, —S— or —O—, and form a ring with the nitrogen to which they are attached;
R9 is 1-2 groups independently selected from hydrogen, C1-C6 alkyl, hydroxy, C1-C6 alkoxy, halogen, —CF3 and (C1-C6)alkoxy(C1-C6)alkoxy;
R10 is 1 to 5 substituents independently selected from the group consisting of hydrogen, halogen, C1-C6 alkyl, hydroxy, C1-C6 alkoxy, —CN, —NH2, C1-C6alkylamino, di-((C1-C6)alkyl)amino, —CF3, —OCF3 and —S(O)0-2(C1-C6)alkyl;
R11 is H, C1-C6 alkyl, phenyl, benzyl, C2-C6 alkenyl, C1-C6 alkoxy(C1-C6)alkyl, di-((C1-C6)alkyl)amino(C1-C6)alkyl, pyrrolidinyl(C1-C6)alkyl or piperidino(C1-C6)alkyl;
R12 is H or C1-C6 alkyl; and
R13 is (C1-C6)alkyl-C(O)- or (C1-C6)alkyl-SO2—;
wherein heteroaryl is a single ring or benzofused heteroaromatic group, or an N-oxide thereof, of 5 to 10 atoms comprised of 2 to 9 carbon atoms and 1 to 4 heteroatoms independently selected from the group consisting of N, O and S, provided that the rings do not include adjacent oxygen atoms, adjacent sulfur atoms, or adjacent oxygen and sulfur atoms.
2. A compound of claim 1 wherein R is R1-furanyl.
3. A compound of claim 1 wherein X is C2-C6 alkylene.
6. A compound of claim 5 wherein m and n are each 2, and R4 is H.
7. A compound of claim 1 wherein Z is R5-phenyl, R5-heteroaryl, R6—C(O)— or R6—SO2—.
8. A compound of claim 7 wherein R5 is H, halogen, C1-C6 alkyl, C1-C6 alkoxy, hydroxy(C1-C6)alkoxy or (C1-C6)alkoxy(C1-C6)alkoxy, or R6 is R5-phenyl.
11. A compound of claim 1 wherein Z is R5-heteroaryl and heteroaryl is selected from the group consisting of pyridyl, oxazolyl, isoxazolyl, oxadiazolyl, furanyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, tetrazolyl, thiazolyl, isothiazolyl, thiadiazolyl, pyrazinyl, pyrimidyl, pyridazinyl, triazolyl, indolyl, quinolyl, isoquinolyl, phthalazinyl, benzothienyl, benzimidazolyl, benzofuranyl, benzoxazolyl and benzofurazanyl, or an N-oxide thereof.
12. A compound of claim 11 wherein heteroaryl is selected from the group consisting of pyridyl, pyridyl N-oxide, thiazolyl, pyrazinyl, pyrimidyl, quinolyl and benzimidazolyl.
14. A pharmaceutical composition comprising a therapeutically effective amount of a compound of claim 1 in a pharmaceutically acceptable carrier.
15. A method of treating central nervous system diseases or stroke, comprising administering an effective amount of a compound of formula I claim 1, or a pharmaceutically acceptable salt thereof, to a mammal in need of such treatment.
16. A method of claim 15 for treating depression, cognitive diseases and neurodegenerative diseases.
17. A method of claim 16 for treating Parkinson's disease, senile dementia or psychoses of organic origin.
20. The method of claim 19 wherein said central nervous system disease is Parkinson's disease, senile dementia, or psychoses of organic origin.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/441,140 USRE44205E1 (en) | 2000-05-26 | 2012-04-06 | Adenosine A2a receptor antagonists |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US20714300P | 2000-05-26 | 2000-05-26 | |
| US09/865,071 US6630475B2 (en) | 2000-05-26 | 2001-05-24 | Adenosine A2a receptor antagonists |
| US13/441,140 USRE44205E1 (en) | 2000-05-26 | 2012-04-06 | Adenosine A2a receptor antagonists |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/865,071 Reissue US6630475B2 (en) | 2000-05-26 | 2001-05-24 | Adenosine A2a receptor antagonists |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE44205E1 true USRE44205E1 (en) | 2013-05-07 |
Family
ID=22769372
Family Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/865,071 Ceased US6630475B2 (en) | 2000-05-26 | 2001-05-24 | Adenosine A2a receptor antagonists |
| US10/448,854 Expired - Lifetime US6897216B2 (en) | 2000-05-26 | 2003-05-30 | Adenosine A2a receptor antagonists |
| US10/912,834 Expired - Lifetime US7067655B2 (en) | 2000-05-26 | 2004-08-06 | Adenosine A2a receptor antagonists |
| US13/441,140 Expired - Lifetime USRE44205E1 (en) | 2000-05-26 | 2012-04-06 | Adenosine A2a receptor antagonists |
Family Applications Before (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/865,071 Ceased US6630475B2 (en) | 2000-05-26 | 2001-05-24 | Adenosine A2a receptor antagonists |
| US10/448,854 Expired - Lifetime US6897216B2 (en) | 2000-05-26 | 2003-05-30 | Adenosine A2a receptor antagonists |
| US10/912,834 Expired - Lifetime US7067655B2 (en) | 2000-05-26 | 2004-08-06 | Adenosine A2a receptor antagonists |
Country Status (31)
| Country | Link |
|---|---|
| US (4) | US6630475B2 (en) |
| EP (1) | EP1283839B1 (en) |
| JP (3) | JP4574112B2 (en) |
| KR (1) | KR100520907B1 (en) |
| CN (2) | CN100384847C (en) |
| AR (1) | AR028621A1 (en) |
| AT (1) | ATE293627T1 (en) |
| AU (2) | AU6808901A (en) |
| BR (1) | BRPI0111015B8 (en) |
| CA (1) | CA2410237C (en) |
| CZ (1) | CZ303790B6 (en) |
| DE (1) | DE60110219T2 (en) |
| DK (1) | DK1283839T3 (en) |
| EC (1) | ECSP024364A (en) |
| ES (1) | ES2237576T3 (en) |
| HK (1) | HK1049007B (en) |
| HU (1) | HU230420B1 (en) |
| IL (3) | IL152726A0 (en) |
| MX (1) | MXPA02011625A (en) |
| MY (1) | MY132006A (en) |
| NO (1) | NO325008B1 (en) |
| NZ (1) | NZ522326A (en) |
| PE (1) | PE20020062A1 (en) |
| PL (1) | PL218764B1 (en) |
| PT (1) | PT1283839E (en) |
| RU (1) | RU2315053C2 (en) |
| SI (1) | SI1283839T1 (en) |
| SK (1) | SK287748B6 (en) |
| TW (1) | TWI288137B (en) |
| WO (1) | WO2001092264A1 (en) |
| ZA (1) | ZA200208898B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11498923B2 (en) | 2017-12-13 | 2022-11-15 | Merck Sharp & Dohme Llc | Substituted imidazo[1,2-c]quinazolines as A2A antagonists |
| US12060357B2 (en) | 2018-11-30 | 2024-08-13 | Merck Sharp & Dohme Llc | 9-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use |
Families Citing this family (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE60110219T2 (en) * | 2000-05-26 | 2006-03-09 | Schering Corp. | ADENOSINE A2A RECEPTOR ANTAGONISTS |
| CA2459304A1 (en) * | 2001-09-13 | 2003-03-20 | Schering Corporation | Combination of an adenosine a2a receptor antagonist and an antidepressant or anxiolytic |
| KR100687954B1 (en) | 2001-10-15 | 2007-02-27 | 쉐링 코포레이션 | Imidazo (4,3-e) -1,2,4-triazolo (1,5-c) pyrimidine as adenosine A2a receptor antagonist |
| FR2832405B1 (en) * | 2001-11-19 | 2004-12-10 | Sanofi Synthelabo | TETRAHYDROPYRIDYL-ALKYL-HETEROCYCLES NITROGEN, PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| AR038366A1 (en) | 2001-11-30 | 2005-01-12 | Schering Corp | 1,2,4-TRIAZOLO COMPOUNDS [1,5-C] SUBSTITUTED PYRIMIDINS, ANTAGONISTS OF THE ADENOSINE A2A RECEPTOR, PHARMACEUTICAL COMPOSITIONS, THE USE OF SUCH COMPOUNDS FOR THE MANUFACTURE OF A MEDICINAL PRODUCT FOR THE PROCESSING OF DISEASE SYSTEMS AND CENTRAL SYSTEMS A KIT THAT INCLUDES COMBINATION |
| IL161573A0 (en) | 2001-11-30 | 2004-09-27 | Schering Corp | [1,2,4]-TRIAZOLE BICYCLIC ADENOSINE A2a RECEPTOR ANTAGONISTS |
| IL161716A0 (en) * | 2001-11-30 | 2004-09-27 | Schering Corp | ADENOSINE A2a RECEPTOR ANTAGONISTS |
| KR101098209B1 (en) | 2002-01-28 | 2011-12-23 | 교와 핫꼬 기린 가부시키가이샤 | Methods of treating patients suffering from movement disorders |
| AU2003245380A1 (en) | 2002-05-30 | 2003-12-19 | King Pharmaceuticals Research & Development, Inc. | Pharmaceutically active compounds having a tricyclic pyrazolotriazolopyrimidine ring structure and methods of use |
| US20060106040A1 (en) * | 2002-12-19 | 2006-05-18 | Michael Grzelak | Adenosine A2a receptor antagonists for the treatment of extra-pyramidal syndrome and other movement disorders |
| TWI331036B (en) * | 2002-12-19 | 2010-10-01 | Schering Corp | Adenosine a2a receptor antagonists for the treatment of extra-pyramidal syndrome and other movement disorders |
| WO2004092172A2 (en) | 2003-04-09 | 2004-10-28 | Biogen Idec Ma Inc. | Triazolo[1,5-c]pyrimidines and pyrazolo[1,5-c]pyrimidines useful as a2a adenosin e receptor antagonists |
| EP1622912B1 (en) | 2003-04-23 | 2009-05-27 | Schering Corporation | 2-alkynyl-and 2-alkenyl-pyrazolo- [4,3-e ] -1,2,4-triazolo- [1,5-c] -pyrimidine adenosine a2a receptor antagonists |
| KR20060037252A (en) * | 2003-06-10 | 2006-05-03 | 교와 핫꼬 고교 가부시끼가이샤 | How to treat anxiety disorder |
| WO2004111004A1 (en) * | 2003-06-12 | 2004-12-23 | Novo Nordisk A/S | Substituted piperazine carbamates for use as inhibitors of hormone sensitive lipase |
| WO2005039506A2 (en) * | 2003-10-24 | 2005-05-06 | Exelixis, Inc. | P70s6 kinase modulators and method of use |
| ES2278353T3 (en) * | 2003-10-28 | 2007-08-01 | Schering Corporation | PROCEDURE TO PREPARE 5-AMINO-PIRAZOLO- (4,3-E) -1,2,4-TRIAZOLO (1,5-C) SUBSTITUTED PYRIMIDINS. |
| CA2547248A1 (en) | 2003-12-01 | 2005-06-16 | Schering Corporation | Process for preparing substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidines |
| CA2550071A1 (en) * | 2003-12-19 | 2005-07-07 | Schering Corporation | Pharmaceutical compositions |
| ATE461932T1 (en) | 2004-04-21 | 2010-04-15 | Schering Corp | PYRAZOLOÄ4,3-EÜ-1,2,4-TRIAZOLOÄ1,5-CUPYRIMIDINES AS ANTAGONISTS OF THE ADENOSINE A2A RECEPTOR |
| EP2258372B8 (en) * | 2005-06-07 | 2012-12-19 | Kyowa Hakko Kirin Co., Ltd. | A2A antagonists for use in the treatment of motor disorders |
| EP1934227B1 (en) | 2005-09-19 | 2011-12-21 | Schering Corporation | 2-HETEROARYL-PYRAZOLO-[4, 3-e]-1, 2, 4-TRIAZOLO-[1,5-c]-PYRIMIDINE AS ADENOSINE A2a RECEPTOR ANTAGONISTS |
| PE20070521A1 (en) | 2005-09-23 | 2007-07-13 | Schering Corp | 7- [2- [4- (6-FLUORO-3-METHYL-1,2-BENZYSOXAZOL-5-IL) -1-PIPERAZINYL] ETHYL] -2- (1-PROPINYL) -7H-PIRAZOLE- [4, 3-E] - [1,2,4] -TRIAZOL- [1,5-C] -PYRIMIDIN-5-AMINE AS ANTAGONIST OF THE ADENOSINE A2a RECEPTOR |
| ES2273599B1 (en) | 2005-10-14 | 2008-06-01 | Universidad De Barcelona | COMPOUNDS FOR THE TREATMENT OF HEADPHONE FIBRILATION. |
| EP2040698A4 (en) * | 2006-07-14 | 2011-08-10 | Shionogi & Co | Oxime compounds and the use thereof |
| TW200840566A (en) * | 2006-12-22 | 2008-10-16 | Esteve Labor Dr | Heterocyclyl-substituted-ethylamino-phenyl derivatives, their preparation and use as medicaments |
| US7723343B2 (en) | 2007-03-30 | 2010-05-25 | King Pharmaceuticals Research And Development, Inc. | Adenosine A2A receptor antagonists |
| WO2009110955A2 (en) * | 2008-02-29 | 2009-09-11 | Albert Einstein College Of Medicine Of Yeshiva University | Ketoconazole-derivative antagonists of human pregnane x receptor and uses thereof |
| JP2011513417A (en) | 2008-03-04 | 2011-04-28 | シェーリング コーポレイション | 1,2,4-Triazolo [4,3-c] pyrimidin-3-one and pyrazolo [4,3-e] -1,2,4-triazolo [4,3 for use as adenosine A2a receptor antagonists -C] pyrimidin-3-one compounds |
| EP2274007B1 (en) | 2008-03-10 | 2016-12-14 | Cornell University | Modulation of blood brain barrier permeability |
| TWI473614B (en) | 2008-05-29 | 2015-02-21 | Kyowa Hakko Kirin Co Ltd | Anti-analgesic inhibitors |
| KR20110040856A (en) * | 2008-07-23 | 2011-04-20 | 교와 핫꼬 기린 가부시키가이샤 | Migraine Remedies |
| US20100093702A1 (en) * | 2008-10-13 | 2010-04-15 | Barbay J Kent | METHYLENE AMINES OF THIENO[2,3-d]PYRIMIDINE AND THEIR USE AS ADENOSINE A2a RECEPTOR ANTAGONISTS |
| US8273754B2 (en) | 2008-12-30 | 2012-09-25 | Arqule, Inc. | Substituted 1H-pyrazolo[3,4-D]pyrimidine-6-amine compounds |
| MX2011007678A (en) | 2009-01-20 | 2011-08-08 | Schering Corp | Methods of alleviating or treating signs and/or symptoms associated with moderate to severe parkinson's disease. |
| US8993808B2 (en) | 2009-01-21 | 2015-03-31 | Oryzon Genomics, S.A. | Phenylcyclopropylamine derivatives and their medical use |
| AU2010222289B2 (en) | 2009-03-13 | 2013-07-11 | Advinus Therapeutics Private Limited | Substituted fused pyrimidine compounds |
| US8343961B2 (en) | 2009-03-31 | 2013-01-01 | Arqule, Inc. | Substituted heterocyclic compounds |
| US8537177B2 (en) * | 2009-06-15 | 2013-09-17 | Marvell World Trade Ltd. | System and methods for gamut bounded saturation adaptive color enhancement |
| US8598343B2 (en) | 2009-08-07 | 2013-12-03 | Merck Sharp & Dohme Corp. | Process for preparing a 2-alkynyl substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine |
| US8859555B2 (en) | 2009-09-25 | 2014-10-14 | Oryzon Genomics S.A. | Lysine Specific Demethylase-1 inhibitors and their use |
| EP2486002B1 (en) | 2009-10-09 | 2019-03-27 | Oryzon Genomics, S.A. | Substituted heteroaryl- and aryl- cyclopropylamine acetamides and their use |
| WO2011101861A1 (en) | 2010-01-29 | 2011-08-25 | Msn Laboratories Limited | Process for preparation of dpp-iv inhibitors |
| WO2011106106A2 (en) | 2010-02-24 | 2011-09-01 | Oryzon Genomics, S.A. | Lysine demethylase inhibitors for diseases and disorders associated with hepadnaviridae |
| WO2011106574A2 (en) | 2010-02-24 | 2011-09-01 | Oryzon Genomics, S.A. | Inhibitors for antiviral use |
| CA2796726C (en) | 2010-04-19 | 2021-02-16 | Oryzon Genomics S.A. | Lysine specific demethylase-1 inhibitors and their use |
| US9006449B2 (en) | 2010-07-29 | 2015-04-14 | Oryzon Genomics, S.A. | Cyclopropylamine derivatives useful as LSD1 inhibitors |
| BR112013002164B1 (en) | 2010-07-29 | 2021-11-09 | Oryzon Genomics S.A. | DEMETHYLASE INHIBITORS BASED ON LSD1 ARILCYCLOPROPYLAMINE, THEIR USES, AND PHARMACEUTICAL COMPOSITION |
| AU2011306358B2 (en) * | 2010-09-24 | 2014-08-14 | Impetis Biosciences Ltd. | Fused tricyclic compounds as adenosine receptor antagonist |
| US9061966B2 (en) | 2010-10-08 | 2015-06-23 | Oryzon Genomics S.A. | Cyclopropylamine inhibitors of oxidases |
| WO2012072713A2 (en) | 2010-11-30 | 2012-06-07 | Oryzon Genomics, S.A. | Lysine demethylase inhibitors for diseases and disorders associated with flaviviridae |
| EP2712315B1 (en) | 2011-02-08 | 2021-11-24 | Oryzon Genomics, S.A. | Lysine demethylase inhibitors for myeloproliferative disorders |
| WO2012129381A1 (en) * | 2011-03-22 | 2012-09-27 | Concert Pharmaceuticals Inc. | Deuterated preladenant |
| WO2012127472A1 (en) * | 2011-03-22 | 2012-09-27 | Mapi Pharma Ltd. | Process and intermediates for the preparation of preladenant and related compounds |
| WO2013024474A1 (en) * | 2011-08-18 | 2013-02-21 | Mapi Phrarma Ltd. | Polymorphs of preladenant |
| EP2768805B1 (en) | 2011-10-20 | 2020-03-25 | Oryzon Genomics, S.A. | (hetero)aryl cyclopropylamine compounds as lsd1 inhibitors |
| JP6046154B2 (en) | 2011-10-20 | 2016-12-14 | オリソン ヘノミクス エセ. アー. | (Hetero) arylcyclopropylamine compounds as LSD1 inhibitors |
| WO2014071512A1 (en) * | 2012-11-06 | 2014-05-15 | Universite Laval | Combination therapy and methods for the treatment of respiratory diseases |
| WO2014101120A1 (en) * | 2012-12-28 | 2014-07-03 | Merck Sharp & Dohme Corp. | Heterobicyclo-substituted-7-methoxy-[1,2,4]triazolo[1,5-c]quinazolin-5-amine compounds with a2a antagonist properties |
| US10472347B2 (en) | 2014-11-18 | 2019-11-12 | Merck Sharp & Dohme Corp. | Aminopyrazine compounds with A2A antagonist properties |
| EP3253390B1 (en) | 2015-02-06 | 2022-04-13 | Merck Sharp & Dohme Corp. | Aminoquinazoline compounds as a2a antagonist |
| WO2016200717A1 (en) | 2015-06-11 | 2016-12-15 | Merck Sharp & Dohme Corp. | Aminopyrazine compounds with a2a antagonist properties |
| WO2017008205A1 (en) | 2015-07-10 | 2017-01-19 | Merck Sharp & Dohme Corp. | Substituted aminoquinazoline compounds as a2a antagonist |
| US9687475B1 (en) | 2016-03-24 | 2017-06-27 | Ezra Pharma Llc | Extended release pharmaceutical formulations with controlled impurity levels |
| US9675585B1 (en) | 2016-03-24 | 2017-06-13 | Ezra Pharma | Extended release pharmaceutical formulations |
| TWI801372B (en) * | 2017-03-30 | 2023-05-11 | 比利時商艾特歐斯比利時有限公司 | Thiocarbamate derivatives as a2a inhibitors and methods for use in the treatment of cancers |
| AU2018246355B2 (en) * | 2017-03-30 | 2021-12-09 | iTeos Belgium SA | 2-oxo-thiazole derivatives as A2A inhibitors and compounds for use in the treatment of cancers |
| IL275817B2 (en) * | 2018-01-04 | 2024-04-01 | Impetis Biosciences Ltd | Tricyclic compounds, preparations and their medical applications |
| TWI877770B (en) | 2018-02-27 | 2025-03-21 | 美商英塞特公司 | Imidazopyrimidines and triazolopyrimidines as a2a / a2b inhibitors |
| CN108276345A (en) * | 2018-03-22 | 2018-07-13 | 重庆奥舍生物化工有限公司 | A kind of preparation method of pharmaceutical intermediate pyrimidine -5-formaldehyde |
| EP3774813A4 (en) * | 2018-04-08 | 2021-11-17 | BeiGene, Ltd. | PYRAZOLOTRIAZOLOPYRIMIDINE DERIVATIVES USED AS AN A2A RECEPTOR ANTAGONIST |
| EP3810610A1 (en) | 2018-05-18 | 2021-04-28 | Incyte Corporation | Fused pyrimidine derivatives as a2a / a2b inhibitors |
| CN108864114B (en) * | 2018-06-04 | 2020-11-06 | 应世生物科技(南京)有限公司 | Selectivity A2AReceptor antagonists |
| WO2020010197A1 (en) | 2018-07-05 | 2020-01-09 | Incyte Corporation | Fused pyrazine derivatives as a2a / a2b inhibitors |
| CN110742893B (en) * | 2018-07-23 | 2024-04-05 | 百济神州(北京)生物科技有限公司 | Methods of treating cancer with A2A receptor antagonists |
| CA3124088A1 (en) | 2018-12-20 | 2020-06-25 | Incyte Corporation | Imidazopyridazine and imidazopyridine compounds as inhibitors of activin receptor-like kinase-2 |
| TWI829857B (en) | 2019-01-29 | 2024-01-21 | 美商英塞特公司 | Pyrazolopyridines and triazolopyridines as a2a / a2b inhibitors |
| WO2020227156A1 (en) * | 2019-05-03 | 2020-11-12 | Nektar Therapeutics | Adenosine 2 receptor antagonists |
| DE102019116986A1 (en) | 2019-06-24 | 2020-12-24 | Helmholtz-Zentrum Dresden-Rossendorf E. V. | Deuterated 7- (3- (4- (2 - ([18F] fluoro) ethoxy) phenyl) propyl) -2- (furan-2-yl) -7H-pyrazolo [4,3-e] [1,2, 4] triazolo [1,5-c] pyrimidin-5-amine derivatives |
| JP2022540583A (en) * | 2019-07-17 | 2022-09-16 | テオン セラピューティクス,インク. | Adenosine A2A receptor antagonist and use thereof |
| CN112028891B (en) | 2019-07-30 | 2022-07-05 | 厦门宝太生物科技股份有限公司 | Adenosine receptor antagonists |
| CN112592354B (en) | 2019-07-30 | 2022-05-27 | 厦门宝太生物科技股份有限公司 | Preparation method of isoxazolo-pyrimidine heterocyclic compound |
| CN111072675A (en) * | 2019-12-12 | 2020-04-28 | 广东东阳光药业有限公司 | Nitrogen-containing fused tricyclic derivatives and uses thereof |
| CN113773327B (en) * | 2021-09-13 | 2022-07-15 | 八叶草健康产业研究院(厦门)有限公司 | Preparation method of pyrazolopyrimidinyltriazole ring compound |
| CN117946123A (en) * | 2024-01-26 | 2024-04-30 | 北京脑重大疾病研究院 | Radioactive triazolopyrimidinyl derivatives and preparation methods and applications thereof |
| CN118812544A (en) * | 2024-06-19 | 2024-10-22 | 厦门大学 | Adenosine A2AR targeting small molecule compound, nuclide labeled probe, preparation method, application and pharmaceutical composition thereof |
| CN118812539B (en) * | 2024-06-19 | 2025-11-28 | 苏州大学 | 5-Aminotriazolopyrimidine derivative and preparation method thereof |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0217748A2 (en) | 1985-09-30 | 1987-04-08 | Ciba-Geigy Ag | 2-Substituted-e-fused-[1,2,4,]triazolo-[1,5-c]pyrimidines pharmaceutical compositions and uses thereof |
| WO1995001356A1 (en) | 1993-06-29 | 1995-01-12 | Schering-Plough S.P.A. | 1,2,4-TRIAZOLO[1,5-c]PYRIMIDINE HETEROCYCLIC ANALOGUES HAVING ANTAGONISTIC ACTIVITY ON ADENOSINE A2 RECEPTOR |
| US5565460A (en) | 1993-07-27 | 1996-10-15 | Kyowa Hakko Koygo Co., Ltd. | Therapeutic purine agents for parkinson's disease |
| WO1997005138A1 (en) | 1995-07-28 | 1997-02-13 | Schering-Plough S.P.A. | 1,2,4-TRIAZOLO[1,5-c]PYRIMIDINE HETEROCYCLIC ANALOGUES HAVING ANTAGONISTIC ACTIVITY ON ADENOSINE A2a RECEPTOR |
| WO1998052568A1 (en) | 1997-05-21 | 1998-11-26 | Schering-Plough S.P.A. | THE USE OF 1,2,4-TRIAZOLO[1,5-c]PYRIMIDINE HETEROCYCLIC ANALOGUES FOR THE PREPARATION OF MEDICAMENTS USEFUL FOR THE TREATMENT OF CEREBROVASCULAR DISTURBANCES |
| US6897216B2 (en) * | 2000-05-26 | 2005-05-24 | Schering Corporation | Adenosine A2a receptor antagonists |
| US6916811B2 (en) * | 2001-11-30 | 2005-07-12 | Schering Corporation | Adenosine A2a receptor antagonists |
| US7223861B2 (en) * | 2003-10-28 | 2007-05-29 | Schering Corporation | Process for preparing substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo-[1,5-c]-pyrimidines |
| US7414058B2 (en) * | 2002-12-19 | 2008-08-19 | Schering Corporation | Adenosine A2a receptor antagonists for the treatment of extra-pyramidal syndrome and other movement disorders |
| US7465740B2 (en) * | 2005-09-19 | 2008-12-16 | Schering Corporation | 2-heteroaryl-pyrazolo-[4,3-e]-1,2,4-triazolo-[1,5-c]-pyrimidine adenosine A2a receptor antagonists |
| US7507822B2 (en) * | 2003-12-01 | 2009-03-24 | Schering Corporation | Process for preparing substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidines |
| US7709492B2 (en) * | 2004-04-21 | 2010-05-04 | Schering Corporation | Pyrazolo-[4,3-e]-1,2,4-triazolo-[1,5-c]-pyrimidine adenosine A2a receptor antagonists |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SU1739850A3 (en) * | 1987-08-31 | 1992-06-07 | Такеда Кемикал Индастриз, Лтд (Фирма) | Method for synthesis of condensed derivatives of pyrazolo-[3,4-d]-pyrimidine |
-
2001
- 2001-05-24 DE DE60110219T patent/DE60110219T2/en not_active Expired - Lifetime
- 2001-05-24 EP EP01945991A patent/EP1283839B1/en not_active Expired - Lifetime
- 2001-05-24 CZ CZ20023886A patent/CZ303790B6/en not_active IP Right Cessation
- 2001-05-24 MX MXPA02011625A patent/MXPA02011625A/en active IP Right Grant
- 2001-05-24 AU AU6808901A patent/AU6808901A/en active Pending
- 2001-05-24 SI SI200130336T patent/SI1283839T1/xx unknown
- 2001-05-24 BR BR0111015-2 patent/BRPI0111015B8/en not_active IP Right Cessation
- 2001-05-24 JP JP2002500877A patent/JP4574112B2/en not_active Expired - Lifetime
- 2001-05-24 KR KR10-2002-7015699A patent/KR100520907B1/en not_active Expired - Lifetime
- 2001-05-24 US US09/865,071 patent/US6630475B2/en not_active Ceased
- 2001-05-24 PE PE2001000475A patent/PE20020062A1/en active IP Right Grant
- 2001-05-24 RU RU2002135620/04A patent/RU2315053C2/en active
- 2001-05-24 SK SK1671-2002A patent/SK287748B6/en not_active IP Right Cessation
- 2001-05-24 CN CNB2006100049297A patent/CN100384847C/en not_active Expired - Lifetime
- 2001-05-24 CA CA002410237A patent/CA2410237C/en not_active Expired - Lifetime
- 2001-05-24 HU HU0600239A patent/HU230420B1/en unknown
- 2001-05-24 AR ARP010102483A patent/AR028621A1/en active IP Right Grant
- 2001-05-24 DK DK01945991T patent/DK1283839T3/en active
- 2001-05-24 CN CNB018134491A patent/CN1247588C/en not_active Expired - Lifetime
- 2001-05-24 AU AU2001268089A patent/AU2001268089C1/en not_active Expired
- 2001-05-24 HK HK03101315.3A patent/HK1049007B/en not_active IP Right Cessation
- 2001-05-24 MY MYPI20012466A patent/MY132006A/en unknown
- 2001-05-24 WO PCT/US2001/016954 patent/WO2001092264A1/en not_active Ceased
- 2001-05-24 NZ NZ522326A patent/NZ522326A/en not_active IP Right Cessation
- 2001-05-24 IL IL15272601A patent/IL152726A0/en unknown
- 2001-05-24 ES ES01945991T patent/ES2237576T3/en not_active Expired - Lifetime
- 2001-05-24 PL PL360472A patent/PL218764B1/en unknown
- 2001-05-24 AT AT01945991T patent/ATE293627T1/en active
- 2001-05-24 TW TW090112461A patent/TWI288137B/en not_active IP Right Cessation
- 2001-05-24 PT PT01945991T patent/PT1283839E/en unknown
-
2002
- 2002-11-01 ZA ZA200208898A patent/ZA200208898B/en unknown
- 2002-11-10 IL IL152726A patent/IL152726A/en active IP Right Grant
- 2002-11-25 EC EC2002004364A patent/ECSP024364A/en unknown
- 2002-11-25 NO NO20025651A patent/NO325008B1/en not_active IP Right Cessation
-
2003
- 2003-05-30 US US10/448,854 patent/US6897216B2/en not_active Expired - Lifetime
-
2004
- 2004-08-06 US US10/912,834 patent/US7067655B2/en not_active Expired - Lifetime
-
2006
- 2006-05-02 JP JP2006128415A patent/JP4938348B2/en not_active Expired - Lifetime
-
2007
- 2007-03-16 JP JP2007069618A patent/JP2007145875A/en not_active Withdrawn
-
2012
- 2012-04-06 US US13/441,140 patent/USRE44205E1/en not_active Expired - Lifetime
- 2012-06-04 IL IL220174A patent/IL220174A/en active IP Right Grant
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0217748A2 (en) | 1985-09-30 | 1987-04-08 | Ciba-Geigy Ag | 2-Substituted-e-fused-[1,2,4,]triazolo-[1,5-c]pyrimidines pharmaceutical compositions and uses thereof |
| WO1995001356A1 (en) | 1993-06-29 | 1995-01-12 | Schering-Plough S.P.A. | 1,2,4-TRIAZOLO[1,5-c]PYRIMIDINE HETEROCYCLIC ANALOGUES HAVING ANTAGONISTIC ACTIVITY ON ADENOSINE A2 RECEPTOR |
| US5565460A (en) | 1993-07-27 | 1996-10-15 | Kyowa Hakko Koygo Co., Ltd. | Therapeutic purine agents for parkinson's disease |
| WO1997005138A1 (en) | 1995-07-28 | 1997-02-13 | Schering-Plough S.P.A. | 1,2,4-TRIAZOLO[1,5-c]PYRIMIDINE HETEROCYCLIC ANALOGUES HAVING ANTAGONISTIC ACTIVITY ON ADENOSINE A2a RECEPTOR |
| US5935964A (en) * | 1995-07-28 | 1999-08-10 | Schering Corporation | 1,2,4-triazolo 1,5-c! pyrimidine heterocyclic analogues having antagonistic activity on adenosine A2a receptor |
| WO1998052568A1 (en) | 1997-05-21 | 1998-11-26 | Schering-Plough S.P.A. | THE USE OF 1,2,4-TRIAZOLO[1,5-c]PYRIMIDINE HETEROCYCLIC ANALOGUES FOR THE PREPARATION OF MEDICAMENTS USEFUL FOR THE TREATMENT OF CEREBROVASCULAR DISTURBANCES |
| US6897216B2 (en) * | 2000-05-26 | 2005-05-24 | Schering Corporation | Adenosine A2a receptor antagonists |
| US7067655B2 (en) * | 2000-05-26 | 2006-06-27 | Schering Corporation | Adenosine A2a receptor antagonists |
| US6916811B2 (en) * | 2001-11-30 | 2005-07-12 | Schering Corporation | Adenosine A2a receptor antagonists |
| US7414058B2 (en) * | 2002-12-19 | 2008-08-19 | Schering Corporation | Adenosine A2a receptor antagonists for the treatment of extra-pyramidal syndrome and other movement disorders |
| US7223861B2 (en) * | 2003-10-28 | 2007-05-29 | Schering Corporation | Process for preparing substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo-[1,5-c]-pyrimidines |
| US7507822B2 (en) * | 2003-12-01 | 2009-03-24 | Schering Corporation | Process for preparing substituted 5-amino-pyrazolo-[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidines |
| US7709492B2 (en) * | 2004-04-21 | 2010-05-04 | Schering Corporation | Pyrazolo-[4,3-e]-1,2,4-triazolo-[1,5-c]-pyrimidine adenosine A2a receptor antagonists |
| US7465740B2 (en) * | 2005-09-19 | 2008-12-16 | Schering Corporation | 2-heteroaryl-pyrazolo-[4,3-e]-1,2,4-triazolo-[1,5-c]-pyrimidine adenosine A2a receptor antagonists |
Non-Patent Citations (14)
| Title |
|---|
| Adami et al, Br. J. Pharmacol., 126, (Mar. 1999), 283P. |
| Adami et al. "The Adenosine A Receptor Blocker, SCH 58261, IS Effective over a 2-Week Treatment in Haloperiodol-Induced Catalepsy", British Journal of Pharmacology, 126, pp. 283 (Mar. 1999). |
| Baraldi et al, J. Med. Chem., 39 (1996), pp. 1164-1171. |
| Baraldi et al. "Pyrozolo[4,3-e]-1,2,4-triazolo[1,5-c]pyrimidine Derivatives: Potent and Selective A2a Adenosine Antagonists", J. Med. Chem, 39, pp. 1164-1171 (1996). |
| C. Zocchi et al., 117 British Journal of Pharmacology 1381-1386 (1996). * |
| P.G. Baraldi et al., 39 Journal of Medicinal Chemistry 1164-1171 (1996). * |
| R. Bertorelli et al., 37 Drug Development Research 65-72 (1996). * |
| Seela et al, Helvetica Chimca Acta, 69 (1986), pp. 1602-1613. |
| Seela et al. "168. Synthesis of 2'-Deoxyriboforanosides of 8-Aza-7-deazaguanine and Related Pyrazolol[3,4-d] pyrimidines", Helvetica Chicima Acta, vol. 69, pp. 1602-1613. (1986). |
| Seela et al. "168. Synthesis of 2′-Deoxyriboforanosides of 8-Aza-7-deazaguanine and Related Pyrazolol[3,4-d] pyrimidines", Helvetica Chicima Acta, vol. 69, pp. 1602-1613. (1986). |
| Ungerstedt et al, Brian Research, 24 (1970), pp. 485-493. |
| Ungerstedt et al. "6-Hydorxy-Dopamine induced Degeneration of Central Monoamine Neurons", European Journal of Pharmacology, 5, pp. 107-110 (1968). |
| Ungerstedt et al. "Quantitative Recording of Rotational Behaviour in Rats after 6-Hydroxy Dopamine Lesions of the Nigrostriatal Dopahmine System", Brian Research, vol. 24, pp. 485-493 (1970). |
| Ungerstedt, Eur. J. Pharmacol., 5 (1968), pp. 107-110. |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11498923B2 (en) | 2017-12-13 | 2022-11-15 | Merck Sharp & Dohme Llc | Substituted imidazo[1,2-c]quinazolines as A2A antagonists |
| US12060357B2 (en) | 2018-11-30 | 2024-08-13 | Merck Sharp & Dohme Llc | 9-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE44205E1 (en) | Adenosine A2a receptor antagonists | |
| US6653315B2 (en) | Adenosine A2a receptor antagonists | |
| US6875772B2 (en) | [1,2,4]-Triazole bicyclic adeonsine A2a receptor antagonists | |
| AU2001268089A1 (en) | Adenosine A2a receptor antagonists | |
| US7041666B2 (en) | Adenosine A2a receptor antagonists | |
| HK1064097B (en) | Bicyclic [1,2,4] -triazole adenosine a2a receptor antagonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FPAY | Fee payment |
Year of fee payment: 12 |