US9809596B1 - Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis - Google Patents
Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis Download PDFInfo
- Publication number
- US9809596B1 US9809596B1 US15/522,336 US201515522336A US9809596B1 US 9809596 B1 US9809596 B1 US 9809596B1 US 201515522336 A US201515522336 A US 201515522336A US 9809596 B1 US9809596 B1 US 9809596B1
- Authority
- US
- United States
- Prior art keywords
- alkyl
- group
- compsubst
- independently
- substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 BrC1=CC=CS1.C1=CC=C2C=CC=CC2=C1.C1=CC=C2NC=NC2=C1.C1=CC=C2OC=NC2=C1.C1=CC=C2SC=NC2=C1.C1=CNC=N1.CC(=O)C1=CC=CN1C.CC(=O)CCCCN1C(=O)C2=C(N=CN2C)N(C)C1=O.CC1=CC2=CC=CC=C2N1C.CC1=CC=C(C)S1.CC1=CN(C)C2=CC=CC=C12.CC1=NN(C)C(O)=C1.CCC1=CC=C(C)N=C1.CN1C=C(C(=O)O[Y])C=N1.CN1C=CC2=CC=CC=C21.CN1C=CC=C1.CN1C=NC2=C1C(=O)N(C)C(=O)N2C.[1*]C1=NC(C)=CC=C1C(C)=O Chemical compound BrC1=CC=CS1.C1=CC=C2C=CC=CC2=C1.C1=CC=C2NC=NC2=C1.C1=CC=C2OC=NC2=C1.C1=CC=C2SC=NC2=C1.C1=CNC=N1.CC(=O)C1=CC=CN1C.CC(=O)CCCCN1C(=O)C2=C(N=CN2C)N(C)C1=O.CC1=CC2=CC=CC=C2N1C.CC1=CC=C(C)S1.CC1=CN(C)C2=CC=CC=C12.CC1=NN(C)C(O)=C1.CCC1=CC=C(C)N=C1.CN1C=C(C(=O)O[Y])C=N1.CN1C=CC2=CC=CC=C21.CN1C=CC=C1.CN1C=NC2=C1C(=O)N(C)C(=O)N2C.[1*]C1=NC(C)=CC=C1C(C)=O 0.000 description 4
- LXDKTVRWMMEZAP-UHFFFAOYSA-N BrC1=CC=CC=C1.C1=CC=NC=C1.CC1=CC(=O)OC(N(C)C)=N1.CC1=CC=CC(C)=N1.CC1=CC=CN=C1.CC1=CC=NC=C1.CC1=NC=CC=C1.CN(C)C1=CC=CC=C1.COC1=CC(F)=CC(OC)=C1.COC1=CC=C(C(F)(F)F)C=C1.COC1=CC=C(Cl)C=C1.ClC1=CC=C(Cl)C=C1.ClC1=CC=CC(Cl)=C1.ClC1=CC=CC=C1.FC(F)(F)C1=CC=CC=C1.FC1=CC=CC=C1.N#CC1=CC=C(N)C=C1.NC1=CC=C(C(F)(F)F)C=C1.NC1=CC=CC=C1.NC1=CC=CC=C1.O=C(O)C1=CC=CN=C1.[C-]#[N+]C1=CC(OC)=CC=C1.[C-]#[N+]C1=CC=CC=C1 Chemical compound BrC1=CC=CC=C1.C1=CC=NC=C1.CC1=CC(=O)OC(N(C)C)=N1.CC1=CC=CC(C)=N1.CC1=CC=CN=C1.CC1=CC=NC=C1.CC1=NC=CC=C1.CN(C)C1=CC=CC=C1.COC1=CC(F)=CC(OC)=C1.COC1=CC=C(C(F)(F)F)C=C1.COC1=CC=C(Cl)C=C1.ClC1=CC=C(Cl)C=C1.ClC1=CC=CC(Cl)=C1.ClC1=CC=CC=C1.FC(F)(F)C1=CC=CC=C1.FC1=CC=CC=C1.N#CC1=CC=C(N)C=C1.NC1=CC=C(C(F)(F)F)C=C1.NC1=CC=CC=C1.NC1=CC=CC=C1.O=C(O)C1=CC=CN=C1.[C-]#[N+]C1=CC(OC)=CC=C1.[C-]#[N+]C1=CC=CC=C1 LXDKTVRWMMEZAP-UHFFFAOYSA-N 0.000 description 2
- VHMHRMXKSFDPAR-UHFFFAOYSA-N C1=CC=C2C=CC=CC2=C1.C1=CC=C2N=CC=CC2=C1.C1=CC=C2N=CC=NC2=C1.C1=CC=C2NC=CC2=C1.C1=CC=C2NC=NC2=C1.C1=CC=C2OC=NC2=C1.C1=CC=C2SC=NC2=C1.C1=CC=CC=C1.C1=CC=NC=C1.C1=CN=C2N=CC=CC2=C1.C1=CN=CN=C1.C1=CNC=C1.C1=CNC=N1.C1=CNN=C1.C1=COC=N1.C1=COCN=C1.C1=CSC=C1.C1=CSC=N1.C1=NC=C2N=CNC2=N1.C1=NC=NC=N1 Chemical compound C1=CC=C2C=CC=CC2=C1.C1=CC=C2N=CC=CC2=C1.C1=CC=C2N=CC=NC2=C1.C1=CC=C2NC=CC2=C1.C1=CC=C2NC=NC2=C1.C1=CC=C2OC=NC2=C1.C1=CC=C2SC=NC2=C1.C1=CC=CC=C1.C1=CC=NC=C1.C1=CN=C2N=CC=CC2=C1.C1=CN=CN=C1.C1=CNC=C1.C1=CNC=N1.C1=CNN=C1.C1=COC=N1.C1=COCN=C1.C1=CSC=C1.C1=CSC=N1.C1=NC=C2N=CNC2=N1.C1=NC=NC=N1 VHMHRMXKSFDPAR-UHFFFAOYSA-N 0.000 description 2
- GPHUUQBMZHXMNW-UHFFFAOYSA-N CC(=O)C1=CC=CC=C1.CC1=CC(C)=C(C)C(C)=C1C.CC1=CC(C)=CC(C)=C1.CC1=CC=C(C)C=C1.CC1=CC=CC(C)=C1.CC1=CC=CC=C1.COC1=C(OC)C=CC=C1.COC1=CC(C(C)=O)=CC(OC)=C1OC.COC1=CC(O)=CC(OC)=C1.COC1=CC(OC)=CC(OC)=C1.COC1=CC=C(OC)C=C1.COC1=CC=CC(C(C)=O)=C1.COC1=CC=CC(OC)=C1.COC1=CC=CC=C1.[H]C(=O)C1=CC(OC)=CC=C1.[H]C(=O)C1=CC=C(OC)C=C1.[H]C(=O)C1=CC=CC=C1 Chemical compound CC(=O)C1=CC=CC=C1.CC1=CC(C)=C(C)C(C)=C1C.CC1=CC(C)=CC(C)=C1.CC1=CC=C(C)C=C1.CC1=CC=CC(C)=C1.CC1=CC=CC=C1.COC1=C(OC)C=CC=C1.COC1=CC(C(C)=O)=CC(OC)=C1OC.COC1=CC(O)=CC(OC)=C1.COC1=CC(OC)=CC(OC)=C1.COC1=CC=C(OC)C=C1.COC1=CC=CC(C(C)=O)=C1.COC1=CC=CC(OC)=C1.COC1=CC=CC=C1.[H]C(=O)C1=CC(OC)=CC=C1.[H]C(=O)C1=CC=C(OC)C=C1.[H]C(=O)C1=CC=CC=C1 GPHUUQBMZHXMNW-UHFFFAOYSA-N 0.000 description 2
- KPBATUMWJSCAGT-UHFFFAOYSA-N CC1=CC(C(F)(F)F)=C(C)S1 Chemical compound CC1=CC(C(F)(F)F)=C(C)S1 KPBATUMWJSCAGT-UHFFFAOYSA-N 0.000 description 2
- BJMUOUXGBFNLSN-UHFFFAOYSA-N CC1=CC2=CC=CC=C2N1C Chemical compound CC1=CC2=CC=CC=C2N1C BJMUOUXGBFNLSN-UHFFFAOYSA-N 0.000 description 2
- GAQUXLIKHSGSGP-UHFFFAOYSA-N CN1C(=O)C2=C(N=C(C(F)(F)F)N2C)N(C)C1=O Chemical compound CN1C(=O)C2=C(N=C(C(F)(F)F)N2C)N(C)C1=O GAQUXLIKHSGSGP-UHFFFAOYSA-N 0.000 description 2
- VUGQIIQFXCXZJU-UHFFFAOYSA-N COC1=CC(C(C)=O)=CC(OC)=C1OC Chemical compound COC1=CC(C(C)=O)=CC(OC)=C1OC VUGQIIQFXCXZJU-UHFFFAOYSA-N 0.000 description 2
- GXGATBWQQLDTND-UHFFFAOYSA-N C.C1=C(N2CCOCC2)CCCC1.CC1=C(N2CCOCC2)CCCC1 Chemical compound C.C1=C(N2CCOCC2)CCCC1.CC1=C(N2CCOCC2)CCCC1 GXGATBWQQLDTND-UHFFFAOYSA-N 0.000 description 1
- UFWIBTONFRDIAS-UHFFFAOYSA-N C1=CC=C2C=CC=CC2=C1 Chemical compound C1=CC=C2C=CC=CC2=C1 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 1
- UHOVQNZJYSORNB-UHFFFAOYSA-N C1=CC=CC=C1 Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 1
- MGOFNMKXFICBJT-UHFFFAOYSA-N C1=CC=CC=C1.FC(F)(F)(F)(F)(F)(F)(F)(F)(F)(F)(F)(F)#CC#CC#CC1=CC=CC=C1 Chemical compound C1=CC=CC=C1.FC(F)(F)(F)(F)(F)(F)(F)(F)(F)(F)(F)(F)#CC#CC#CC1=CC=CC=C1 MGOFNMKXFICBJT-UHFFFAOYSA-N 0.000 description 1
- FBWMQWZSJPQWCY-UHFFFAOYSA-N C=CC(=O)OCC.CC=CC(=O)OCC.FC(F)(F)Br Chemical compound C=CC(=O)OCC.CC=CC(=O)OCC.FC(F)(F)Br FBWMQWZSJPQWCY-UHFFFAOYSA-N 0.000 description 1
- KKAKTPJEFPRAFX-UHFFFAOYSA-N CC(=O)C1=CC=C(C(F)(F)F)N1C Chemical compound CC(=O)C1=CC=C(C(F)(F)F)N1C KKAKTPJEFPRAFX-UHFFFAOYSA-N 0.000 description 1
- NZFLWVDXYUGFAV-UHFFFAOYSA-N CC(=O)C1=CC=CN1C Chemical compound CC(=O)C1=CC=CN1C NZFLWVDXYUGFAV-UHFFFAOYSA-N 0.000 description 1
- CNPIRRONFLRNQA-UHFFFAOYSA-N CC(=O)CCCCN1C(=O)C2=C(N=C(C(F)(F)F)N2C)N(C)C1=O Chemical compound CC(=O)CCCCN1C(=O)C2=C(N=C(C(F)(F)F)N2C)N(C)C1=O CNPIRRONFLRNQA-UHFFFAOYSA-N 0.000 description 1
- BYPFEZZEUUWMEJ-UHFFFAOYSA-N CC(=O)CCCCN1C(=O)C2=C(N=CN2C)N(C)C1=O Chemical compound CC(=O)CCCCN1C(=O)C2=C(N=CN2C)N(C)C1=O BYPFEZZEUUWMEJ-UHFFFAOYSA-N 0.000 description 1
- ZNMDRUPMGNGPMR-UHFFFAOYSA-N CC1=C(C(F)(F)F)C2=CC=CC=C2N1C Chemical compound CC1=C(C(F)(F)F)C2=CC=CC=C2N1C ZNMDRUPMGNGPMR-UHFFFAOYSA-N 0.000 description 1
- IBMIOOCKERJDCH-UHFFFAOYSA-N CC1=C(C(F)(F)F)N(C)C2=CC=CC=C21 Chemical compound CC1=C(C(F)(F)F)N(C)C2=CC=CC=C21 IBMIOOCKERJDCH-UHFFFAOYSA-N 0.000 description 1
- JYPSDOOVSLJTOV-UHFFFAOYSA-N CC1=C(C)C(C)=C(C(F)(F)F)C(C)=C1C Chemical compound CC1=C(C)C(C)=C(C(F)(F)F)C(C)=C1C JYPSDOOVSLJTOV-UHFFFAOYSA-N 0.000 description 1
- VDEYYSZYUVCKJS-UHFFFAOYSA-N CC1=CC(C)=C(C(F)(F)F)C(C)=C1 Chemical compound CC1=CC(C)=C(C(F)(F)F)C(C)=C1 VDEYYSZYUVCKJS-UHFFFAOYSA-N 0.000 description 1
- BEZDDPMMPIDMGJ-UHFFFAOYSA-N CC1=CC(C)=C(C)C(C)=C1C Chemical compound CC1=CC(C)=C(C)C(C)=C1C BEZDDPMMPIDMGJ-UHFFFAOYSA-N 0.000 description 1
- AUHZEENZYGFFBQ-UHFFFAOYSA-N CC1=CC(C)=CC(C)=C1 Chemical compound CC1=CC(C)=CC(C)=C1 AUHZEENZYGFFBQ-UHFFFAOYSA-N 0.000 description 1
- RIKQFBGCNQAHBO-UHFFFAOYSA-N CC1=CC=C(C)C(C(F)(F)F)=C1 Chemical compound CC1=CC=C(C)C(C(F)(F)F)=C1 RIKQFBGCNQAHBO-UHFFFAOYSA-N 0.000 description 1
- URLKBWYHVLBVBO-UHFFFAOYSA-N CC1=CC=C(C)C=C1 Chemical compound CC1=CC=C(C)C=C1 URLKBWYHVLBVBO-UHFFFAOYSA-N 0.000 description 1
- GWQOOADXMVQEFT-UHFFFAOYSA-N CC1=CC=C(C)S1 Chemical compound CC1=CC=C(C)S1 GWQOOADXMVQEFT-UHFFFAOYSA-N 0.000 description 1
- NAPPMSNSLWACIV-UHFFFAOYSA-N CC1=CN(C)C2=CC=CC=C12 Chemical compound CC1=CN(C)C2=CC=CC=C12 NAPPMSNSLWACIV-UHFFFAOYSA-N 0.000 description 1
- OXHNLMTVIGZXSG-UHFFFAOYSA-N CN1C=CC=C1 Chemical compound CN1C=CC=C1 OXHNLMTVIGZXSG-UHFFFAOYSA-N 0.000 description 1
- POYGQTHRUFGPKC-UHFFFAOYSA-N CN1C=CC=C1C(F)(F)F Chemical compound CN1C=CC=C1C(F)(F)F POYGQTHRUFGPKC-UHFFFAOYSA-N 0.000 description 1
- RYYVLZVUVIJVGH-UHFFFAOYSA-N CN1C=NC2=C1C(=O)N(C)C(=O)N2C Chemical compound CN1C=NC2=C1C(=O)N(C)C(=O)N2C RYYVLZVUVIJVGH-UHFFFAOYSA-N 0.000 description 1
- HGQQQXMARFJNCP-UHFFFAOYSA-N COC(=O)C1=CN(C)N=C1 Chemical compound COC(=O)C1=CN(C)N=C1 HGQQQXMARFJNCP-UHFFFAOYSA-N 0.000 description 1
- BHPJMWUXVOTGQU-UHFFFAOYSA-N COC1=C(C(F)(F)F)C=CC=C1 Chemical compound COC1=C(C(F)(F)F)C=CC=C1 BHPJMWUXVOTGQU-UHFFFAOYSA-N 0.000 description 1
- RGJTXXPQFGWECT-UHFFFAOYSA-N COC1=C(OC)C=C(C(F)(F)F)C=C1 Chemical compound COC1=C(OC)C=C(C(F)(F)F)C=C1 RGJTXXPQFGWECT-UHFFFAOYSA-N 0.000 description 1
- ABDKAPXRBAPSQN-UHFFFAOYSA-N COC1=C(OC)C=CC=C1 Chemical compound COC1=C(OC)C=CC=C1 ABDKAPXRBAPSQN-UHFFFAOYSA-N 0.000 description 1
- FZJACHPROOKGTJ-UHFFFAOYSA-N COC1=CC(C(C)=O)=C(C(F)(F)F)C(OC)=C1OC Chemical compound COC1=CC(C(C)=O)=C(C(F)(F)F)C(OC)=C1OC FZJACHPROOKGTJ-UHFFFAOYSA-N 0.000 description 1
- QMJFCDRKDKBSMC-UHFFFAOYSA-N COC1=CC(C(F)(F)F)=C(OC)C=C1 Chemical compound COC1=CC(C(F)(F)F)=C(OC)C=C1 QMJFCDRKDKBSMC-UHFFFAOYSA-N 0.000 description 1
- DVJHJMXQRDYTHT-UHFFFAOYSA-N COC1=CC(OC)=C(C(F)(F)F)C(OC)=C1 Chemical compound COC1=CC(OC)=C(C(F)(F)F)C(OC)=C1 DVJHJMXQRDYTHT-UHFFFAOYSA-N 0.000 description 1
- IKLUEOPZYMFAQK-UHFFFAOYSA-N COC1=CC(OC)=C(C(F)(F)F)C=C1 Chemical compound COC1=CC(OC)=C(C(F)(F)F)C=C1 IKLUEOPZYMFAQK-UHFFFAOYSA-N 0.000 description 1
- LKUDPHPHKOZXCD-UHFFFAOYSA-N COC1=CC(OC)=CC(OC)=C1 Chemical compound COC1=CC(OC)=CC(OC)=C1 LKUDPHPHKOZXCD-UHFFFAOYSA-N 0.000 description 1
- OHBQPCCCRFSCAX-UHFFFAOYSA-N COC1=CC=C(OC)C=C1 Chemical compound COC1=CC=C(OC)C=C1 OHBQPCCCRFSCAX-UHFFFAOYSA-N 0.000 description 1
- DPZNOMCNRMUKPS-UHFFFAOYSA-N COC1=CC=CC(OC)=C1 Chemical compound COC1=CC=CC(OC)=C1 DPZNOMCNRMUKPS-UHFFFAOYSA-N 0.000 description 1
- RDOXTESZEPMUJZ-UHFFFAOYSA-N COC1=CC=CC=C1 Chemical compound COC1=CC=CC=C1 RDOXTESZEPMUJZ-UHFFFAOYSA-N 0.000 description 1
- GOBHSUAEQGWYBT-UHFFFAOYSA-N FC(F)(F)C1=CC=CC2=CC=CC=C21 Chemical compound FC(F)(F)C1=CC=CC2=CC=CC=C21 GOBHSUAEQGWYBT-UHFFFAOYSA-N 0.000 description 1
- GETTZEONDQJALK-UHFFFAOYSA-N FC(F)(F)C1=CC=CC=C1 Chemical compound FC(F)(F)C1=CC=CC=C1 GETTZEONDQJALK-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/04—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms
- C07D473/06—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms with radicals containing only hydrogen and carbon atoms, attached in position 1 or 3
- C07D473/10—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms with radicals containing only hydrogen and carbon atoms, attached in position 1 or 3 with methyl radicals in positions 3 and 7, e.g. theobromine
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/22—Organic complexes
- B01J31/2204—Organic complexes the ligands containing oxygen or sulfur as complexing atoms
- B01J31/2208—Oxygen, e.g. acetylacetonates
- B01J31/2226—Anionic ligands, i.e. the overall ligand carries at least one formal negative charge
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B39/00—Halogenation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C17/00—Preparation of halogenated hydrocarbons
- C07C17/26—Preparation of halogenated hydrocarbons by reactions involving an increase in the number of carbon atoms in the skeleton
- C07C17/272—Preparation of halogenated hydrocarbons by reactions involving an increase in the number of carbon atoms in the skeleton by addition reactions
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C17/00—Preparation of halogenated hydrocarbons
- C07C17/26—Preparation of halogenated hydrocarbons by reactions involving an increase in the number of carbon atoms in the skeleton
- C07C17/272—Preparation of halogenated hydrocarbons by reactions involving an increase in the number of carbon atoms in the skeleton by addition reactions
- C07C17/275—Preparation of halogenated hydrocarbons by reactions involving an increase in the number of carbon atoms in the skeleton by addition reactions of hydrocarbons and halogenated hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C22/00—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom
- C07C22/02—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings
- C07C22/04—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings containing six-membered aromatic rings
- C07C22/08—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings containing six-membered aromatic rings containing fluorine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C41/00—Preparation of ethers; Preparation of compounds having groups, groups or groups
- C07C41/01—Preparation of ethers
- C07C41/18—Preparation of ethers by reactions not forming ether-oxygen bonds
- C07C41/30—Preparation of ethers by reactions not forming ether-oxygen bonds by increasing the number of carbon atoms, e.g. by oligomerisation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C41/00—Preparation of ethers; Preparation of compounds having groups, groups or groups
- C07C41/01—Preparation of ethers
- C07C41/34—Separation; Purification; Stabilisation; Use of additives
- C07C41/46—Use of additives, e.g. for stabilisation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/20—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring
- C07C43/225—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring containing halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C45/00—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds
- C07C45/61—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups
- C07C45/67—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups by isomerisation; by change of size of the carbon skeleton
- C07C45/68—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups by isomerisation; by change of size of the carbon skeleton by increase in the number of carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C45/00—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds
- C07C45/61—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups
- C07C45/67—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups by isomerisation; by change of size of the carbon skeleton
- C07C45/68—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups by isomerisation; by change of size of the carbon skeleton by increase in the number of carbon atoms
- C07C45/69—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds by reactions not involving the formation of >C = O groups by isomerisation; by change of size of the carbon skeleton by increase in the number of carbon atoms by addition to carbon-to-carbon double or triple bonds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C49/00—Ketones; Ketenes; Dimeric ketenes; Ketonic chelates
- C07C49/76—Ketones containing a keto group bound to a six-membered aromatic ring
- C07C49/80—Ketones containing a keto group bound to a six-membered aromatic ring containing halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C49/00—Ketones; Ketenes; Dimeric ketenes; Ketonic chelates
- C07C49/76—Ketones containing a keto group bound to a six-membered aromatic ring
- C07C49/84—Ketones containing a keto group bound to a six-membered aromatic ring containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/32—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/325—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with substituted hydrocarbon radicals directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/32—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/33—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/30—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members
- C07D207/32—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/33—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms with substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D207/333—Radicals substituted by oxygen or sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/10—Indoles; Hydrogenated indoles with substituted hydrocarbon radicals attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/12—Radicals substituted by halogen atoms or nitro or nitroso radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/04—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms
- C07D473/06—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms with radicals containing only hydrogen and carbon atoms, attached in position 1 or 3
- C07D473/08—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 two oxygen atoms with radicals containing only hydrogen and carbon atoms, attached in position 1 or 3 with methyl radicals in positions 1 and 3, e.g. theophylline
Definitions
- the invention discloses a method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous Pd catalyzed fluoro, chloro and fluorochloro alkylation with fluoro, chloro and fluorochloro alkyl halides in the presence of di(1-adamantyl)-n-butylphosphine and in the presence of 2,2,6,6-tetramethylpiperidine 1-oxyl.
- Fluoroalkyl groups have strong effects such as high stability and lipophilicity, in addition, longer fluoroalkyl groups have high water and oil resistance and low fricition.
- alkyl linear or branched alkyl
- BuPAd 2 CAS 321921-71-5, di(1-adamantyl)-n-butylphosphine;
- halide F—, Cl—, Br— or I— preferably Cl—, Br—, and I—, more preferably Br— and I—;
- halogen F, Cl, Br or I preferably F, Cl or Br; more preferably F or Cl;
- Subject of the invention is a method for the preparation of a fluoro, chloro or fluorochloro alkylated compound by a reaction of a compound COMPSUBST with a compound FCLALKYLHALIDE by homogeneous catalysis using a catalyst CAT
- BAS is selected from the group consisting of Cs 2 CO 3 , CsHCO 3 , NEt 3 , and mixtures thereof;
- FCLALKYLHALIDE is a compound of formula (III): R3—X (III)
- X is Cl, Br or I
- R3 is C 1-20 alkyl or a C 1-20 alkyl, wherein in the alkyl chain at least one of the hydrogens is substituted by F or Cl;
- CAT is selected from the group consisting of Pd(OAc) 2 , Pd(TFA) 2 , and mixtures thereof;
- COMPSUBST is selected from the group consisting of a compound COMPSUBST-I, ethene, cyclohexene, ethine, and polystyrene;
- COMPSUBST-I contains a ring RINGA
- RINGA is an unsaturated or aromatic, 5 or 6 membered carbocyclic or heterocyclic ring,
- RINGA can be condensed with a ring RINGB, RINGB is a 5 or 6 membered carbocyclic or heterocyclic ring,
- RINGB is unsubstituted or substituted with 1, 2 or 3 in case of RINGB being a 5 membered ring, with 1, 2, 3 or 4 in case of RINGB being a 6 membered ring, identical or different substitutents independently from each other selected from the group consisting of C 1-10 alkyl, C 3-8 cycloalkyl, C 1-4 alkoxy, OH, N(R17)R18, CN, NH—OH, NO, NO 2 , F, Cl, Br, I, CF 3 , (CH 2 ) n —C(O)Y2, S(O) 2 R51, CH ⁇ C(H)R38, C ⁇ C—R34, benzyl, phenyl and naphthyl;
- Y1, Y2 and R13 are identical or different and independently from each other selected from the group consisting of H, OH, C(R14)(R15)R16, C 1-6 alkyl, O—C 1-6 alkyl, phenyl, benzyl, O-phenyl, O—C 1-6 alkylen-O—C 1-6 alkyl and N(R19)R20;
- R14, R15 and R16 are identical or different and independently from each other selected from the group consisting of H, F, Cl and Br;
- R10, R11, R17, R18, R19 and R20 are identical or different and are independently from each other H or C 1-6 alkyl, or R10 and R11, R17 and R18 or R19 and R20 represent together a tetramethylene or a pentamethylene chain;
- R50 and R51 are identical or different and independently from each other selected from the group consisting of OH, C 1-6 alkyl and C 1-6 alkoxy;
- R24, R34, R28 and R38 are identical or different and independently from each other selected from the group consisting of H, C 1-6 alkyl, C(R25)(R26)—O—R27;
- R25, R26 and R27 are identical or different and independently from each other selected from the group consisting of H and C 1-6 alkyl.
- RINGA is a carbocyclic unsaturated ring, a carbocyclic aromatic ring, a heterocyclic unsaturated ring or a heterocyclic aromatic ring.
- COMPSUBST is selected from the group consisting of compound COMPSUBST-I, ethene, cyclohexene, ethine, and polystyrene;
- COMPSUBST-I unsubstituted or substituted
- R10, R11, m, n, Y1, Y2, R28, R50 and R24 are defined as above, also with all their embodiments.
- n, n and q are identical or different and independently from each other 0, 1, 2, 3 or 4:
- m, n and q are 0 or 4.
- Y1, Y2 and R13 are identical or different and independently from each other selected from the group consisting of H, OH, C(R14)(R15)R16, C 2-6 alkyl, O—C 1-6 alkyl, phenyl, benzyl, O-phenyl, O—C 1-6 alkylen-O—C 1-6 alkyl and N(R19)R20.
- Y1, Y2 and R13 are identical or different and independently from each other selected from the group consisting of H, OH, C 1-2 alkyl, and O—C 1-2 alkyl.
- COMPSUBST-I is unsubstituted or substituted
- COMPSUBST is selected from the group consisting of benzene, pyrazole,
- Y is C 1-6 alkyl
- the ethene and the cyclohexene being unsubstituted or substituted by 1 or 2 substitutents selected from the group consisting of C 1-10 alkyl, C 1-4 alkoxy, N(R10)R11, CN, F, Cl, Br, I, CF 3 , (CH 2 ) m —C(O)Y1, benzyl, phenyl and morpholine;
- the ethine being unsubstituted or substituted by 1 substitutent selected from the group consisting of C 1-10 alkyl, C 1-4 alkoxy, N(R10)R11, CN, F, Cl, Br, I, CF 3 , (CH 2 ) m —C(O)Y1, benzyl and phenyl;
- R44 is selected from the group consisting of C 1-10 alkyl, C 1-4 alkoxy, OH, N(R10)R11, CN, NO, NO 2 , F, Cl, Br, I, CF 3 , (CH 2 ) m —C(O)Y1, S(O) 2 R50;
- R10, R11, m, Y1 and R50 as defined above, also with all their embodiments.
- Embodiments of the substituted ethene are propene, ethene-1,1-diyldibenzene and 3,3-dimethylbut-1-ene.
- An embodiment of substituted cyclohexene is 4-(cyclohex-1-en-1-yl)morpholine.
- An embodiment of the substituted ethine is 1-octyne.
- Y is methyl or ethyl.
- Y is methyl or ethyl, preferably ethyl.
- the fluoro, chloro or fluorochloro alkylated compound is called compound ALKYLCOMPSUBST.
- the fluoro, chloro and fluorochloro alkyl halide is compound FCLALKYLHALIDE.
- FCLALKYLHALIDE is a compound of formula (III); R3—X (III)
- X is Cl, Br or I
- R3 is C 1-20 alkyl or a C 1-20 alkyl, wherein in the alkyl chain at least one of the hydrogens is substituted by F or Cl;
- R3 is C 1-15 alkyl, wherein in the alkyl chain at least one of the hydrogens is substituted by F or Cl;
- R3 is C 1-10 alkyl or C 1-10 alkyl, wherein in the alkyl chain at least one of the hydrogens is substituted by F or Cl.
- X is Br or I
- X is I
- X is Br
- compound FCLALKYLHADLIDE is a perfluoroalkyl halide, F 2 HC—Cl or F 2 HC—Br, preferably FCLALKYLHADLIDE is a perfluoroalkylated bromide or iodide, F 2 HC—Cl or F 2 HC—Br;
- X is Cl, Br or I
- R3 is perfluoro C 1-20 alkyl
- FCLALKYLHADLIDE is F 2 HC—Cl or F 2 HC—Br;
- X is Br or I
- R3 is perfluoro C 1-20 alkyl
- FCLALKYLHADLIDE is F 2 HC—Cl or F 2 HC—Br;
- X is Br or I
- R3 is perfluoro C 1-15 alkyl
- FCLALKYLHADLIDE is F 2 HC—Cl or F 2 HC—Br.
- FCLALKYLHALIDE is selected from the group consisting of F 21 C 10 —I, F 17 C 8 —I, F 13 C 6 —I, F 9 C 4 —I, F 3 C—I, F 3 C—Br, F 3 C—Cl, F 2 HC—Cl, and F 2 HC—Br;
- FCLALKYLHALIDE is selected from the group consisting of n-F 21 C 10 -I, n-F 17 C 8 —I, n-F 13 C 6 —I, n-F 9 C 4 —I, F 3 C—I, F 3 C—Br, F 3 C—Cl, F 2 HC—Cl, and F 2 HC—Br.
- the reaction is done in the presence of a compound COMPSALT
- COMPSALT is selected from the group consisting of NaI, KI, CsI and N(R30)(R31)(R32)R33I;
- R30, R31, R32 and R33 are identical or different and independently from each other selected from the group consisting of H and C 1-10 alkyl;
- R30, R31, R32 and R33 are identical or different and independently from each other selected from the group consisting of H and C 2-6 alkyl;
- COMPSALT is selected from the group consisting of NaI and (n-Bu) 4 NI.
- the reaction is preferably done in the presence of a compound COMPSALT and X is Cl or Br, preferably X is Cl.
- CAT is Pd(OAc) 2 .
- the mol% are based on the molar amount of COMPSUBST.
- mol equivalents are based on the molar amount of COMPSUBST.
- FCLALKYLHALIDE being in gaseous form
- FCLALKYLHALIDE was used in the reaction in an amount which corresponds to a pressure of from 1 to 10 bar, more preferably from 1 to 5 bar, at ambient temperature.
- the mol% are based on the molar amount of COMPSUBST.
- mol equivalents are based on the molar amount of COMPSUBST.
- BAS is Cs 2 CO 3 .
- the mol equivalents are based on the molar amount of COMPSUBST.
- the reaction temperature of the reaction is preferably from 20 to 200° C., more preferably from 50 to 200° C., even more preferably from 50 to 150° C., especially from 100 to 150° C., more especially from 110 to 145° C.
- the reaction time of the reaction is preferably from 1 h to 60 h, more preferably from 10 h to 50 h, even more preferably from 15 h to 50 h.
- the reaction is done under inert atmosphere.
- the inert atmosphere is achieved by the use if an inert gas preferably selected from the group consisting of argon, another noble gas, lower boiling alkane, nitrogen and mixtures thereof.
- the lower boiling alkane is preferably a C 1-3 alkane, i.e. methane, ethane or propane.
- the reaction can be done in a closed system, it can be done at a pressure caused by the chosen temperature in a closed system, and/or caused by the pressure applied by COMPSUBST, in case that COMPSUBST is in gaseous form. It is also possible to apply pressure with said inert gas. It is also possible to carry out the reaction at ambient pressure.
- SOL is preferably selected from the group consisting of alkanes, chlorinated alkanes, ketones, ethers, esters, aliphatic nitrils, aliphatic amides, sulfoxides, and mixtures thereof;
- SOL is selected from the group consisting of C 5-8 alkane, chlorinated C 5-8 alkane, acetone, methylethylketone, diethylketone, MTBE, tetrahydrofuran, methyltetrahydrofuran, ethylacetate, butylacetate, valeronitril, acetonitrile, dimethylformamide, dimethylacetamide, N-methylpyrrolidone, dimethylsulfoxide, and mixtures thereof;
- SOL is selected from the group consisting of acetone, methylethylketone, diethylketone, valeronitril, acetonitrile, dimethylsulfoxide, and mixtures thereof;
- SOL is selected from the group consisting of acetone, methylethylketone, diethylketone, dimethylsulfoxide, and mixtures thereof.
- COMPSUBST simultaneously as substrate and as solvent.
- reaction can also be carried out in the absence of a solvent.
- COMPSUBST is used as SOL.
- the amount of SOL is preferably from 0.1 to 100 fold, more preferably from 1 to 50 fold, even more preferably from 1 to 25 fold, of the weight of COMPSUBST.
- ALKYLCOMPSUBST can be isolated by standard methods such as evaporation of volatile components, extraction, washing, drying, concentration, crystallization, chromatography and any combination thereof, which are known per se to the person skilled in the art.
- COMPSUBST, BAS, CAT, BuPAd 2 , TEMPO and FCLALKYLHALIDE the fluoro, chloro and fluorochloro alkyl halide, are commercially available and can be prepared according to known precedures.
- the yield is given in % as a molar yield of the expected ALKYLCOMPSUBST in the reaction mixture after the reaction, and is based on molar amount of COMPSUBST and was determined by 19 F NMR with 1,4 difluorobenzene as internal standard, if not otherwise stated.
- Isolated yield was derived from the weight of the isolated product and is based on the weight of COMPSUBST, isolated yield is given in parenthesis in Table 1.
- the reaction mixture was extracted with water and ethyl acetate (5 times, each time with 3 mL). The organic layers were washed with brine, dried over Na 2 SO 4 , and evaporated to yield the crude product. The yield was 81 %.
- Example 1 repeated with the sole difference that Pd(TFA) 2 was used as CAT instead of Pd(OAc) 2 .
- the yield was 78%.
- Example 1 repeated with the diffenerence that only 5 mol% of Pd(OAc) 2 were used instead of 10 mol%, and that only 10 mol% BuPAd 2 were used instead of 20 mol%. The yield was 42%.
- Example 1 repeated with the sole diffenerence that the reaction mixture was stirred at 130° C. for 30 h instead of 40 h.
- the yield was 70%.
- Example 1 was repeated with the difference that as COMPSUBST the compound listed in Table 1 was used.
- reaction mixture was extracted with water and ethyl acetate (5 times, each time with 3 mL). The organic layers were washed with brine, dried over Na 2 SO 4 , and evaporated to yield the crude product.
- the reaction mixture was analyzed by 19 F-NMR by which an yield of 21% (perfluorohexyl)benzene was found. The identity of the (perfluorohexyl)benzene was confirmed by GC-MS.
- the autoclave was cooled down to room temperature and the pressure was released.
- reaction mixture was extracted with water and ethyl acetate (5 times, each time with 3 mL). The organic layers were washed with brine, dried over Na 2 SO 4 , and evaporated to yield the crude product. The reaction mixture was analyzed by GC—MS by which an yield of 42% 1,4-dimethoxy-2-(perfluorohexy)benzene was found.
- the autoclave was cooled down to room temperature and the pressure was released.
- the reaction mixture was extracted with water and ethyl acetate (5 times, each time with 3 mL). The organic layers were washed with brine, dried over Na 2 SO 4 , and evaporated to yield the crude product. The yield of the product was determined by 19 F-NMR spectroscopy.
- Table 2 shows the parameters that were tested.
- the autoclave was cooled down to room temperature and the pressure was released.
- the resulting reaction mixture was cooled, the pressure released from the autoclave, and the solids filtered.
- GC-MS Analysis showed a molecular weight peak at 168 g/mol confirming monotrifluormethylation.
- the phosphine was BINAP.
- the base was Cs 2 CO 3 .
- alkylhalogenid was perfluorohexyl bromide instead of perfluorohexyl iodide.
- a dried 50 mL autoclave was charged with 4-(cyclohex-1-en-1-yl)morpholine (0.2 mmol), Pd(OAc) 2 (10 mol%), BuPAd 2 (20 mol%), TEMPO (1.0), Cs 2 CO 3 (2.0 equivalents). Then, acetone (2 mL) was injected into the autoclave and the autoclave was flushed with argon for 3 times. A pressure of 6 bar CF 3 Br followed by 15 bar of N 2 was adjusted at ambient temperature. The reaction mixture was heated at 130° C. for 40 h. The autoclave was placed in a heating system and heated at 130° C. for 40 h. After the completion of the reaction, the autoclave was cooled down to room temperature and the pressure was released.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Pyrrole Compounds (AREA)
- Indole Compounds (AREA)
Abstract
Description
R3—X (III)
-
- the ethene and the cyclohexene being unsubstituted or substituted by 1, 2 or 3 substitutents selected from the group consisting of C1-10 alkyl, C3-8 cycloalkyl, C1-4 alkoxy, N(R10)R11, CN, NO, NO2, F, Cl, Br, I, CF3, (CH2)m—C(O)Y1, S(O)2R50, CH═C(H)R28, C≡C—R24, benzyl, phenyl, naphthyl and morpholine;
- the ethine being unsubstituted or substituted by 1 substitutent selected from the group consisting of C1-10 alkyl, C3-8 cycloalkyl, C1-4 alkoxy, N(R10)R11, CN, NO, NO2, F, Cl, Br, I, CF3, (CH2)m—C(O)Y1, S(O)2R50, CH═C(H)R28, C≡C—R24, benzyl, phenyl and naphthyl;
-
- when RINGA is a heterocyclic ring, then RINGA has 1, 2 or 3 identical or different endocyclic heteroatoms independently from each other selected from the group consisting of N, O and S,
- when RINGA is a 5 membered ring, then RINGA is unsubstituted or substituted by 1, 2, 3 or 4 identical or different subsitutents,
- when RINGA is a 6 membered ring then RINGA is unsubstituted or substituted by 1, 2, 3, 4 or 5 identical or different subsitutents,
- any of said subsitutents of RINGA is independently from any other of said substitutent of RINGA selected from the group consisting of C1-10 alkyl, C3-8 cycloalkyl, C1-4 alkoxy, OH, N(R10)R11, CN, NH—OH, NO, NO2, F, Cl, Br, I, CF3, (CH2)m—C(O)Y1, S(O)2R50, CH═C(H)R28, C≡C—R24, benzyl, phenyl and naphthyl;
-
- when RINGB is a heterocyclic ring, is contains 1, 2 or 3 identical or different endocyclic heteroatoms independently from each other selected from the group consisting of N, O and S;
-
- any of said C1-10 alkyl substitutent of RINGA or RINGB is unsubstituted or substituted with 1, 2, 3, 4 or 5 identical or different substituents selected from the group consisting of halogen, OH, O—C(O)—C1-5 alkyl, O—C1-10 alkyl, S—C1-10 alkyl, S(O)—C1-10 alkyl, S(O2)—C1-10 alkyl, O—C1-6 alkylen-O—C1-6 alkyl, C3-8 cycloalkyl and 1,2,4-triazolyl;
- any of said benzyl, phenyl and naphthyl substitutent RINGA or RINGB is independently from each other unsubstituted or substituted with 1, 2, 3, 4 or 5 identical or different substituents selected from the group consisting of halogen, C1-4 alkoxy, NO2 and CN;
m, n and q are identical or different and independently from each other 0, 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10;
-
- the ethene and the cyclohexane being unsubstituted or substituted by 1 or 2 substitutents selected from the group consisting of C1-10 alkyl, C3-6 cycloalkyl, C1-4 alkoxy, N(R10)R11, CN, F, Cl, Br, I, CF3, (CH2)m—C(O)Y1, S(O)2R50, benzyl, phenyl, naphthyl and morpholine;
- the ethine being unsubstituted or substituted by 1 substitutent selected from the group consisting of C1-10 alkyl, C3-6 cycloalkyl, C1-4 alkoxy, N(R10)R11 , CN, F, Cl, Br, I, CF3, (CH2)m—C(O)Y1, S(O)2R50, benzyl, phenyl and naphthyl;
with COMPSUBST-I being selected from the group consisting of
-
- by 1, 2, 3 or 4 in case of COMPSUBST-I being a monocyclic compound with 5 endocyclic atoms,
- by 1, 2, 3, 4 or 5 in case of COMPSUBST-I being a monocyclic compound with 6 endocyclic atoms,
- by 1, 2, 3, 4, 5 or 6 in case of COMPSUBST-I being a bicyclic compound wherein a 5-membered and a 6-membered ring are ortho-fused,
- by 1, 2, 3, 4, 5, 6 or 7 in case of COMPSUBST-I being a bicyclic compound wherein two 6-membered rings are ortho-fused,
- identical or different substituents independently from each other selected from the group consisting of C1-10 alkyl, C3-8 cycloalkyl, C1-4 alkoxy, OH, C(H)═O, N(R10)R11, CN, NH—OH, NO, NO2, F, Cl, Br, I, CF3, (CH2)m—C(O)Y1, S(O)2R50, CH═C(H)R28, C≡C—R24, benzyl, phenyl and naphthyl;
said C1-10 alkyl substitutent of COMPSUBST-I is unsubstituted or substituted with 1, 2, 3, 4 or 5 identical or different substituents selected from the group consisting of halogen, OH, O—C(O)—C1-5 alkyl, O—C1-10 alkyl, S—C1-10 alkyl, S(O)—C1-10 alkyl, S(O2)—C1-10 alkyl, O—C1-6 alkylen-O—C1-6 alkyl, C3-8 cycloalkyl and 1,2,4-triazolyl;
said benzyl, phenyl and naphthyl substitutent of COMPSUBST-I is independently from each other unsubstituted or substituted with 1, 2, 3, 4 or 5 identical or different substituents selected from the group consisting of halogen, C1-4 alkoxy, NO2 and CN;
-
- by 1, 2 or 3 in case of COMPSUBST-I being a monocyclic compound with 5 endocyclic atoms,
- by 1, 2, 3, 4 or 5 in case of COMPSUBST-I being a monocyclic compound with 6 endocyclic atoms,
- by 1, 2, 3, 4 or 5 in case of COMPSUBST-I being a bicyclic compound wherein a 5-membered and a 6-membered ring are ortho-fused,
- by 1, 2, 3 or 4 in case of COMPSUBST-I being a bicyclic compound wherein two 6-membered rings are ortho-fused,
- identical or different substituents independently from each other selected from the group consisting of C1-4 alkyl, C1-4 alkoxy, OH, C(H)═O, N(R10)R11, CN, F, Cl, Br, CF3, (CH2)m—C(O)Y1, and S(O)2R50;
said C1-4 alkyl substitutent of COMPSUBST-I is substituted or substituted with 1, 2 or 3 identical or different substituents selected from the group consisting of halogen;
with R10, R11, Y1 and R50 as defined above, also with all their embodiments.
R3—X (III)
| TABLE 1 | |||
| Example | COMPSUBT | ALYKLCOMPSUBST | Yield (Ratio of Isomers) |
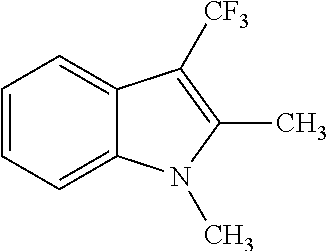
| 1 |
|
|
(69) |
| 5 |
|
|
(81) |
| 6 |
|
|
(76) (2:1 of a:b) |
| 7 |
|
|
(52) |
| 8 |
|
|
78 (3:1 of a:b) |
| 9 |
|
|
72 |
| 10 |
|
|
61 |
| 11 |
|
|
25 |
| 12 |
|
|
53 |
| 13 |
|
|
70 (2:1.2:1 of a:b:c) |
| 14 |
|
|
(47) |
| 15 |
|
|
(79) |
| 16 |
|
|
(76) |
| 17 |
|
|
80 |
| 18 |
|
|
(64) |
| 19 |
|
|
|
| 20 |
|
|
(61) |
| 21 |
|
|
(48) |
| TABLE 2 | ||||
| YIELD | ||||
| ADDITIVE | BASE | SOLVENT | (19F-NMR) | |
| CompEx 1 | 1,4-Benzoquinone | Cs2CO3 | Acetone | 38% |
| CompEx 2 | Pivalic acid | Cs2CO3 | Acetone | 31% |
| CompEx 3 | Ag2O | Cs2CO3 | Acetone | 23% |
| CompEx 4 | TEMPO | K2CO3 | Acetone | 35% |
| CompEx 5 | TEMPO | Na2CO3 | Acetone | 42% |
| Ex 25 | TEMPO | TEA | Acetone | 51% |
| CompEx 6 | TEMPO | CsI | Acetone | 14% |
| CompEx 7 | TEMPO | CsF | Acetone | 26% |
| CompEx 8 | TEMPO | NaI | Acetone | 19% |
| CompEx 9 | TEMPO | K3PO4 | Acetone | 4% |
| CompEx 10 | TEMPO | K-tert. | Acetone | 39% |
| butoxide | ||||
| Ex 26 | TEMPO | Cs2CO3 | DMSO | 74% |
Claims (13)
R3—X (III)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/522,336 US9809596B1 (en) | 2014-11-07 | 2015-11-05 | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis |
Applications Claiming Priority (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462076618P | 2014-11-07 | 2014-11-07 | |
| EP14192280 | 2014-11-07 | ||
| EP14192280.7 | 2014-11-07 | ||
| EP14192280 | 2014-11-07 | ||
| EP15181003 | 2015-08-13 | ||
| EP15181003 | 2015-08-13 | ||
| EP15181003.3 | 2015-08-13 | ||
| EP15181019.9 | 2015-08-14 | ||
| EP15181019 | 2015-08-14 | ||
| EP15181019 | 2015-08-14 | ||
| US15/522,336 US9809596B1 (en) | 2014-11-07 | 2015-11-05 | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis |
| PCT/EP2015/075763 WO2016071425A1 (en) | 2014-11-07 | 2015-11-05 | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US9809596B1 true US9809596B1 (en) | 2017-11-07 |
| US20170320878A1 US20170320878A1 (en) | 2017-11-09 |
Family
ID=55908629
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/522,336 Expired - Fee Related US9809596B1 (en) | 2014-11-07 | 2015-11-05 | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US9809596B1 (en) |
| EP (1) | EP3215476B1 (en) |
| JP (1) | JP6240364B2 (en) |
| KR (1) | KR101810378B1 (en) |
| CN (1) | CN107108390B (en) |
| CA (1) | CA2966791C (en) |
| MX (1) | MX2017006020A (en) |
| TW (1) | TW201630852A (en) |
| WO (1) | WO2016071425A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10968187B2 (en) | 2017-07-28 | 2021-04-06 | Lonza Solutions Ag | Method for preparation of alkylated or fluoro, chloro and fluorochloro alkylated compounds by heterogeneous cobalt catalysis |
| US11739066B2 (en) | 2019-06-12 | 2023-08-29 | Arxada Ag | Method for fluoroalkylation of enamines |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019020726A1 (en) * | 2017-07-28 | 2019-01-31 | Lonza Ltd | Method for preparation of alkylated or fluoro, chloro and fluorochloro alkylated compounds by heterogeneous cobalt catalysis |
| CN117430542B (en) * | 2023-12-21 | 2024-03-08 | 上海恩氟佳科技有限公司 | Synthesis method of trifluoromethyl indole derivative |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2804955B1 (en) * | 2000-02-11 | 2003-02-14 | Rhodia Chimie Sa | METHOD FOR FUNCTIONALIZING A DOUBLE LINK |
| JP5284037B2 (en) * | 2008-10-30 | 2013-09-11 | 保土谷化学工業株式会社 | Method for producing bisarylamine derivative |
| KR20130102065A (en) * | 2010-09-03 | 2013-09-16 | 다이닛본 스미토모 세이야꾸 가부시끼가이샤 | Cyclic amide derivative |
| CA2871237A1 (en) * | 2012-04-24 | 2013-10-31 | Chugai Seiyaku Kabushiki Kaisha | Benzamide derivative |
-
2015
- 2015-11-05 US US15/522,336 patent/US9809596B1/en not_active Expired - Fee Related
- 2015-11-05 CN CN201580060494.5A patent/CN107108390B/en not_active Expired - Fee Related
- 2015-11-05 WO PCT/EP2015/075763 patent/WO2016071425A1/en not_active Ceased
- 2015-11-05 MX MX2017006020A patent/MX2017006020A/en unknown
- 2015-11-05 EP EP15790551.4A patent/EP3215476B1/en not_active Not-in-force
- 2015-11-05 JP JP2017523346A patent/JP6240364B2/en not_active Expired - Fee Related
- 2015-11-05 CA CA2966791A patent/CA2966791C/en not_active Expired - Fee Related
- 2015-11-05 KR KR1020177012077A patent/KR101810378B1/en not_active Expired - Fee Related
- 2015-11-06 TW TW104136653A patent/TW201630852A/en unknown
Non-Patent Citations (4)
| Title |
|---|
| PCT/EP2015/075763 International Preliminary Report on Patentability, dated Sep. 15, 2016. |
| PCT/EP2015/075763 International Search Report and Written Opinion, dated Feb. 9, 2016. |
| Rebecca N. Loy et al: "Palladium-Catalyzed C-H Perfluoroalkylation of Arenes", Organic Letters, vol. 13, No. 10, May 20, 2011, pp. 2548-2551. |
| Theresa Liang et al: Introduction of Fluorine and Fluorine-Containing Functional Groups, Angewandte Chemie International Edition, vol. 52, No. 32, Aug. 5, 2013, pp. 8214-8264. |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10968187B2 (en) | 2017-07-28 | 2021-04-06 | Lonza Solutions Ag | Method for preparation of alkylated or fluoro, chloro and fluorochloro alkylated compounds by heterogeneous cobalt catalysis |
| US11739066B2 (en) | 2019-06-12 | 2023-08-29 | Arxada Ag | Method for fluoroalkylation of enamines |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2966791C (en) | 2018-07-24 |
| KR20170056017A (en) | 2017-05-22 |
| KR101810378B1 (en) | 2017-12-20 |
| JP2017533916A (en) | 2017-11-16 |
| MX2017006020A (en) | 2017-06-19 |
| EP3215476A1 (en) | 2017-09-13 |
| TW201630852A (en) | 2016-09-01 |
| CA2966791A1 (en) | 2016-05-12 |
| JP6240364B2 (en) | 2017-11-29 |
| EP3215476B1 (en) | 2018-02-28 |
| US20170320878A1 (en) | 2017-11-09 |
| CN107108390B (en) | 2018-09-14 |
| CN107108390A (en) | 2017-08-29 |
| WO2016071425A1 (en) | 2016-05-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9809596B1 (en) | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis | |
| US11530175B2 (en) | Method for preparation of fluoro alkylated compounds by homogeneous Ni catalysis | |
| JP2004161769A (en) | Method for production of polyhaloalkane | |
| KR101763552B1 (en) | Method for preparation of alkylated or fluoro, chloro and fluorochloro alkylated compounds by heterogeneous catalysis | |
| JP7049604B2 (en) | Method for Producing Pentafluorosulfanyl Aromatic Compound | |
| EP4101842A1 (en) | Method for producing difluoromethyl-substituted aromatic heterocyclic compound | |
| JP4742358B2 (en) | Method for producing fluorine-containing ether compound | |
| HK1237759B (en) | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis | |
| WO2020249759A1 (en) | Method for fluoroalkylation of enamines | |
| HK1237759A1 (en) | Method for preparation of fluoro, chloro and fluorochloro alkylated compounds by homogeneous catalysis | |
| DK2900634T3 (en) | PROCEDURE FOR PREPARING SUBSTITUTED SUBSTITUTED PHENYL AND PYRIDYLPYRROLIDINES | |
| Yamanaka et al. | Synthesis of benzylaminopyrimidines and their fungicidal activities against wheat brown rust and barley powdery mildew | |
| WO2013008509A1 (en) | Method for stabilizing allyl bromide compound, and stabilized allyl bromide compound composition | |
| EP3645484A1 (en) | Method for preparation of alkylated or fluoro, chloro and fluorochloro alkylated compounds by heterogeneous cobalt catalysis | |
| ES2898937T3 (en) | Organic compound manufacturing process | |
| HK40058174A (en) | Method for preparation of fluoro alkylated compounds by homogeneous ni catalysis | |
| Olivares | Polyfluorinated systems resulting from free radical addition reactions | |
| Wang et al. | First application of ionic liquid to reactions involving organotellurium compounds as intermediates | |
| US20220041570A1 (en) | Method for Preparation of Fluoro Alkylated 1,4-Dioxene by Homogeneous NI Catallysis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: LONZA LTD., SWITZERLAND Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:TAESCHLER, CHRISTOPH;ZARAGOZA DOERWALD, FLORENCIO;ELLINGER, STEFAN;AND OTHERS;SIGNING DATES FROM 20170502 TO 20170508;REEL/FRAME:042715/0684 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| AS | Assignment |
Owner name: LONZA SOLUTIONS AG, SWITZERLAND Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:LONZA LTD (ALSO KNOWN AS LONZA AG);REEL/FRAME:053453/0437 Effective date: 20200803 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |
|
| AS | Assignment |
Owner name: ARXADA AG, SWITZERLAND Free format text: CHANGE OF NAME;ASSIGNOR:LONZA SOLUTIONS AG;REEL/FRAME:058414/0403 Effective date: 20210928 |
|
| FEPP | Fee payment procedure |
Free format text: MAINTENANCE FEE REMINDER MAILED (ORIGINAL EVENT CODE: REM.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| LAPS | Lapse for failure to pay maintenance fees |
Free format text: PATENT EXPIRED FOR FAILURE TO PAY MAINTENANCE FEES (ORIGINAL EVENT CODE: EXP.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20251107 |