US9648880B2 - Antifungal agents and uses thereof - Google Patents
Antifungal agents and uses thereof Download PDFInfo
- Publication number
- US9648880B2 US9648880B2 US14/017,914 US201314017914A US9648880B2 US 9648880 B2 US9648880 B2 US 9648880B2 US 201314017914 A US201314017914 A US 201314017914A US 9648880 B2 US9648880 B2 US 9648880B2
- Authority
- US
- United States
- Prior art keywords
- compound
- compounds
- adhesion
- cells
- composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active, expires
Links
- ANEPXZRMKOQBHZ-UHFFFAOYSA-N CC.CC(=O)N1CCN(C)CC1 Chemical compound CC.CC(=O)N1CCN(C)CC1 ANEPXZRMKOQBHZ-UHFFFAOYSA-N 0.000 description 7
- 0 [1*]N(C)C(=O)CC Chemical compound [1*]N(C)C(=O)CC 0.000 description 6
- BMYDHROVDXCXCW-UHFFFAOYSA-N CC.CC.O=C(CC1=CC=CC=C1)NC1=CC=CC=C1 Chemical compound CC.CC.O=C(CC1=CC=CC=C1)NC1=CC=CC=C1 BMYDHROVDXCXCW-UHFFFAOYSA-N 0.000 description 3
- DCGPJTOBFIJRMK-UHFFFAOYSA-N CC1=CC(C)=NC(=O)N1CC(O)CN1C2=C(C=C(Cl)C=C2)C2=C1/C=C\C(Cl)=C/2.CC1=CC=C(N2C(C)=C3C(C)=NN=C(C)C3=C2C)C=C1C.COC1=CC=C(CCCC(=O)NC(C)C2=CC=CC=C2)C=C1.O=C(NC(=S)N1CCN(C2=CC=CC=C2)CC1)C1=CC=CC=C1.O=C(OC1=CC=CC(NC2=NC=NC3=CC=CC=C32)=C1)C1=CC=CS1.O=C1C2=C(N=C(SCC3=CC=CC=C3)N1C1=CC=CC=C1)SC1=C2CCCC1 Chemical compound CC1=CC(C)=NC(=O)N1CC(O)CN1C2=C(C=C(Cl)C=C2)C2=C1/C=C\C(Cl)=C/2.CC1=CC=C(N2C(C)=C3C(C)=NN=C(C)C3=C2C)C=C1C.COC1=CC=C(CCCC(=O)NC(C)C2=CC=CC=C2)C=C1.O=C(NC(=S)N1CCN(C2=CC=CC=C2)CC1)C1=CC=CC=C1.O=C(OC1=CC=CC(NC2=NC=NC3=CC=CC=C32)=C1)C1=CC=CS1.O=C1C2=C(N=C(SCC3=CC=CC=C3)N1C1=CC=CC=C1)SC1=C2CCCC1 DCGPJTOBFIJRMK-UHFFFAOYSA-N 0.000 description 2
- BLIKUDGXQGBOSA-UHFFFAOYSA-N CC1=CC=C(C(=O)N2CCN(C3=CC=C(Cl)C=C3)CC2)C=C1C Chemical compound CC1=CC=C(C(=O)N2CCN(C3=CC=C(Cl)C=C3)CC2)C=C1C BLIKUDGXQGBOSA-UHFFFAOYSA-N 0.000 description 2
- SQSIDAFIKOMOIN-UHFFFAOYSA-N CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](C)=O)C=C3)CC2)C=C1Cl Chemical compound CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](C)=O)C=C3)CC2)C=C1Cl SQSIDAFIKOMOIN-UHFFFAOYSA-N 0.000 description 2
- DHCRAWRNVVFIMM-UHFFFAOYSA-N CC1=CC=C(OCCCCN2CCCCCC2)C(C)=C1 Chemical compound CC1=CC=C(OCCCCN2CCCCCC2)C(C)=C1 DHCRAWRNVVFIMM-UHFFFAOYSA-N 0.000 description 2
- JONBLLLDPJIHSU-UHFFFAOYSA-N CC1=CC=C(OCCCCN2CCCCCC2)C(C)=C1.COC1=CC=C(N2CCN(C(=O)CCCN3C(=O)C4=CC=CC5=C4/C(=C\C=C/5)C3=O)CC2)C=C1.O=C(NCCC12CC3CC(CC(C3)C1)C2)C1=CC=C(S(=O)(=O)N2CCCCC2)C=C1 Chemical compound CC1=CC=C(OCCCCN2CCCCCC2)C(C)=C1.COC1=CC=C(N2CCN(C(=O)CCCN3C(=O)C4=CC=CC5=C4/C(=C\C=C/5)C3=O)CC2)C=C1.O=C(NCCC12CC3CC(CC(C3)C1)C2)C1=CC=C(S(=O)(=O)N2CCCCC2)C=C1 JONBLLLDPJIHSU-UHFFFAOYSA-N 0.000 description 2
- MJDWYIIOCQNAJQ-UHFFFAOYSA-N COC1=CC=C(C(=O)NC2=CC=CC(Cl)=C2)C=C1Br Chemical compound COC1=CC=C(C(=O)NC2=CC=CC(Cl)=C2)C=C1Br MJDWYIIOCQNAJQ-UHFFFAOYSA-N 0.000 description 2
- SFOMCYFHXRGUHE-UHFFFAOYSA-O C.COC(=O)C1=CC=C(NC(=O)C2=CC=C(C)C(Br)=C2)C=C1.COC1=CC=C(C(=O)NC2=CC=CC(Cl)=C2)C=C1Br.C[N+](=O)C1=CC=C(OCC(=O)NC2=CC(F)=CC=C2)C=C1 Chemical compound C.COC(=O)C1=CC=C(NC(=O)C2=CC=C(C)C(Br)=C2)C=C1.COC1=CC=C(C(=O)NC2=CC=CC(Cl)=C2)C=C1Br.C[N+](=O)C1=CC=C(OCC(=O)NC2=CC(F)=CC=C2)C=C1 SFOMCYFHXRGUHE-UHFFFAOYSA-O 0.000 description 1
- MMXSCKIOSPBWOD-UHFFFAOYSA-N C=[N+]([O-])C1=CC=C(N2CCN(C(=O)C3=CC=C(C(C)(C)C)C=C3)CC2)C=C1.CC1=C(C(=O)N2CCN(C3=CC=C([N+](=O)O)C=C3)CC2)C=CC=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](C)=O)C=C3)CC2)C=C1Cl.CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1.CC1CN(C2=CC=C([N+](C)=O)C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1 Chemical compound C=[N+]([O-])C1=CC=C(N2CCN(C(=O)C3=CC=C(C(C)(C)C)C=C3)CC2)C=C1.CC1=C(C(=O)N2CCN(C3=CC=C([N+](=O)O)C=C3)CC2)C=CC=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](C)=O)C=C3)CC2)C=C1Cl.CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1.CC1CN(C2=CC=C([N+](C)=O)C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1 MMXSCKIOSPBWOD-UHFFFAOYSA-N 0.000 description 1
- QAMJOLFMNYLHQQ-UHFFFAOYSA-N C=[N+]([O-])C1=CC=C(N2CCN(C(=O)C3=CC=C(C(C)(C)C)C=C3)CC2)C=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](C)=O)C=C3)CC2)C=C1Cl.CC1=CC=C(N2CCN(C(=O)C3=CSC4=C3C=CC=C4)CC2)C=C1Cl.CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1.CC1CN(C2=CC=C([N+](C)=O)C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1 Chemical compound C=[N+]([O-])C1=CC=C(N2CCN(C(=O)C3=CC=C(C(C)(C)C)C=C3)CC2)C=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](C)=O)C=C3)CC2)C=C1Cl.CC1=CC=C(N2CCN(C(=O)C3=CSC4=C3C=CC=C4)CC2)C=C1Cl.CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1.CC1CN(C2=CC=C([N+](C)=O)C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1 QAMJOLFMNYLHQQ-UHFFFAOYSA-N 0.000 description 1
- GGPJYWPIJOXZPK-UHFFFAOYSA-N CC(C)(C)C1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1 Chemical compound CC(C)(C)C1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1 GGPJYWPIJOXZPK-UHFFFAOYSA-N 0.000 description 1
- ANFKKJKNTGHDFL-UHFFFAOYSA-N CC(C)(C)C1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1.CC1=C(C(=O)N2CCN(C3=CC=C([N+](=O)O)C=C3)CC2)C=CC=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1Cl.CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1.CC1CN(C2=CC=C([N+](=O)[O-])C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1.C[N+](=O)C1=CC=C(N2CCN(C(=O)C3=CC=C(Cl)C(Cl)=C3)CC2)C=C1 Chemical compound CC(C)(C)C1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1.CC1=C(C(=O)N2CCN(C3=CC=C([N+](=O)O)C=C3)CC2)C=CC=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1Cl.CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1.CC1CN(C2=CC=C([N+](=O)[O-])C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1.C[N+](=O)C1=CC=C(N2CCN(C(=O)C3=CC=C(Cl)C(Cl)=C3)CC2)C=C1 ANFKKJKNTGHDFL-UHFFFAOYSA-N 0.000 description 1
- JHTVHTCKUHCMKW-UHFFFAOYSA-N CC1=C(C(=O)N2CCN(C3=CC=C([N+](=O)O)C=C3)CC2)C=CC=C1.CC1=C(Cl)C=C(N2CCN(C(=O)C3=CC=CC=C3)CC2)C=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C(Cl)C=C3)CC2)C=C1C.C[N+](=O)C1=CC=C(N2CCN(C(=O)C3=CC=C(Cl)C(Cl)=C3)CC2)C=C1 Chemical compound CC1=C(C(=O)N2CCN(C3=CC=C([N+](=O)O)C=C3)CC2)C=CC=C1.CC1=C(Cl)C=C(N2CCN(C(=O)C3=CC=CC=C3)CC2)C=C1.CC1=CC=C(C(=O)N2CCN(C3=CC=C(Cl)C=C3)CC2)C=C1C.C[N+](=O)C1=CC=C(N2CCN(C(=O)C3=CC=C(Cl)C(Cl)=C3)CC2)C=C1 JHTVHTCKUHCMKW-UHFFFAOYSA-N 0.000 description 1
- OUAYFCMOTOVLBM-UHFFFAOYSA-N CC1=C(Cl)C=C(N2CCN(C(=O)C3=CC=CC=C3)CC2)C=C1 Chemical compound CC1=C(Cl)C=C(N2CCN(C(=O)C3=CC=CC=C3)CC2)C=C1 OUAYFCMOTOVLBM-UHFFFAOYSA-N 0.000 description 1
- ZFYFPUBUMMCUNH-UHFFFAOYSA-N CC1=CC(C)=NC(=O)N1CC(O)CN1C2=C(C=C(Cl)C=C2)C2=C1/C=C\C(Cl)=C/2 Chemical compound CC1=CC(C)=NC(=O)N1CC(O)CN1C2=C(C=C(Cl)C=C2)C2=C1/C=C\C(Cl)=C/2 ZFYFPUBUMMCUNH-UHFFFAOYSA-N 0.000 description 1
- PNECWWUOUHGWQG-UHFFFAOYSA-N CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1Cl Chemical compound CC1=CC=C(C(=O)N2CCN(C3=CC=C([N+](=O)[O-])C=C3)CC2)C=C1Cl PNECWWUOUHGWQG-UHFFFAOYSA-N 0.000 description 1
- QQYTZOZNUUKNJD-UHFFFAOYSA-N CC1=CC=C(C(=O)NC(=S)NC2=NC=C(C)C=C2)C(Cl)=C1.CCN(CC)CCNC(=O)C1=CC=C2C(=O)N(CCC3=CCCCC3)C(=S)CC2=C1.CCOC1=CC=C(NC(=O)C2=C(NC(=O)CNCCC3=CC=CC=C3)SC3=C2CCC3)C=C1.COC(=O)C1=C(N2C(CCC(C)=O)=CC=C2C2=CC=C(F)C=C2)C=CC=C1.COC1=CC=C(C2=NC3=C(C=CC=C3)C(C(=O)NC3=C(C)C=CC=C3C(C)C)=C2)C=C1OC.ClC1=C(Cl)C=C(N2CCN(C3=NN=NN3C3=CC=CC=C3)CC2)C=C1 Chemical compound CC1=CC=C(C(=O)NC(=S)NC2=NC=C(C)C=C2)C(Cl)=C1.CCN(CC)CCNC(=O)C1=CC=C2C(=O)N(CCC3=CCCCC3)C(=S)CC2=C1.CCOC1=CC=C(NC(=O)C2=C(NC(=O)CNCCC3=CC=CC=C3)SC3=C2CCC3)C=C1.COC(=O)C1=C(N2C(CCC(C)=O)=CC=C2C2=CC=C(F)C=C2)C=CC=C1.COC1=CC=C(C2=NC3=C(C=CC=C3)C(C(=O)NC3=C(C)C=CC=C3C(C)C)=C2)C=C1OC.ClC1=C(Cl)C=C(N2CCN(C3=NN=NN3C3=CC=CC=C3)CC2)C=C1 QQYTZOZNUUKNJD-UHFFFAOYSA-N 0.000 description 1
- KKYXMCUUJAVLAX-UHFFFAOYSA-N CC1=CC=C(C(=O)NC(=S)NC2=NC=C(Cl)C=C2)C(Cl)=C1 Chemical compound CC1=CC=C(C(=O)NC(=S)NC2=NC=C(Cl)C=C2)C(Cl)=C1 KKYXMCUUJAVLAX-UHFFFAOYSA-N 0.000 description 1
- JVDMFNQDRKCPDC-UHFFFAOYSA-N CC1=CC=C(N2C(C)=C3C(C)=NN=C(C)C3=C2C)C=C1C Chemical compound CC1=CC=C(N2C(C)=C3C(C)=NN=C(C)C3=C2C)C=C1C JVDMFNQDRKCPDC-UHFFFAOYSA-N 0.000 description 1
- QNESUTOJILNPNI-UHFFFAOYSA-N CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1 Chemical compound CC1CN(C2=CC=C([N+](=O)O)C=C2)CCN1C(=O)C1=CC(Br)=CC=C1 QNESUTOJILNPNI-UHFFFAOYSA-N 0.000 description 1
- JSINWULXYRTFQS-UHFFFAOYSA-N CC1CN(C2=CC=C([N+](=O)[O-])C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1 Chemical compound CC1CN(C2=CC=C([N+](=O)[O-])C=C2)CCN1C(=O)C1=C(Cl)C=C(F)C(F)=C1 JSINWULXYRTFQS-UHFFFAOYSA-N 0.000 description 1
- CSOLGFSQJOEHGN-UHFFFAOYSA-N CCN(CC)CCNC(=O)C1=CC=C2C(=O)N(CCC3=CCCCC3)C(=S)CC2=C1 Chemical compound CCN(CC)CCNC(=O)C1=CC=C2C(=O)N(CCC3=CCCCC3)C(=S)CC2=C1 CSOLGFSQJOEHGN-UHFFFAOYSA-N 0.000 description 1
- YHKZRYIZURGMRP-UHFFFAOYSA-N CCOC1=CC=C(NC(=O)C2=C(NC(=O)CNCCC3=CC=CC=C3)SC3=C2CCC3)C=C1 Chemical compound CCOC1=CC=C(NC(=O)C2=C(NC(=O)CNCCC3=CC=CC=C3)SC3=C2CCC3)C=C1 YHKZRYIZURGMRP-UHFFFAOYSA-N 0.000 description 1
- YWEKSEPTFFZRGP-UHFFFAOYSA-N COC(=O)C1=C(N2C(CCC(=O)O)=CC=C2C2=CC=C(F)C=C2)C=CC=C1 Chemical compound COC(=O)C1=C(N2C(CCC(=O)O)=CC=C2C2=CC=C(F)C=C2)C=CC=C1 YWEKSEPTFFZRGP-UHFFFAOYSA-N 0.000 description 1
- XBGOMSJWPKVYRG-UHFFFAOYSA-N COC(=O)C1=CC=C(NC(=O)C2=CC=C(C)C(Br)=C2)C=C1 Chemical compound COC(=O)C1=CC=C(NC(=O)C2=CC=C(C)C(Br)=C2)C=C1 XBGOMSJWPKVYRG-UHFFFAOYSA-N 0.000 description 1
- AMQDLPMVKNNPET-UHFFFAOYSA-N COC1=CC=C(C2=NC3=C(C=CC=C3)C(C(=O)NC3=C(C)C=CC=C3C(C)C)=C2)C=C1OC Chemical compound COC1=CC=C(C2=NC3=C(C=CC=C3)C(C(=O)NC3=C(C)C=CC=C3C(C)C)=C2)C=C1OC AMQDLPMVKNNPET-UHFFFAOYSA-N 0.000 description 1
- YIFURKQWVAEIBH-UHFFFAOYSA-N COC1=CC=C(CCCC(=O)NC(C)C2=CC=CC=C2)C=C1 Chemical compound COC1=CC=C(CCCC(=O)NC(C)C2=CC=CC=C2)C=C1 YIFURKQWVAEIBH-UHFFFAOYSA-N 0.000 description 1
- KATIPTFKORGCOF-UHFFFAOYSA-N COC1=CC=C(N2CCN(C(=O)CCCN3C(=O)C4=CC=CC5=C4/C(=C\C=C/5)C3=O)CC2)C=C1 Chemical compound COC1=CC=C(N2CCN(C(=O)CCCN3C(=O)C4=CC=CC5=C4/C(=C\C=C/5)C3=O)CC2)C=C1 KATIPTFKORGCOF-UHFFFAOYSA-N 0.000 description 1
- FAUVXHNXCUUEBA-UHFFFAOYSA-N C[N+](=O)C1=CC=C(N2CCN(C(=O)C3=CC=C(Cl)C(Cl)=C3)CC2)C=C1 Chemical compound C[N+](=O)C1=CC=C(N2CCN(C(=O)C3=CC=C(Cl)C(Cl)=C3)CC2)C=C1 FAUVXHNXCUUEBA-UHFFFAOYSA-N 0.000 description 1
- HKVDLLVZUTWURP-UHFFFAOYSA-N ClC1=C(Cl)C=C(N2CCN(C3=NN=NN3C3=CC=CC=C3)CC2)C=C1 Chemical compound ClC1=C(Cl)C=C(N2CCN(C3=NN=NN3C3=CC=CC=C3)CC2)C=C1 HKVDLLVZUTWURP-UHFFFAOYSA-N 0.000 description 1
- CNEGXBKNLZCOBN-UHFFFAOYSA-N O=C(C1=C(Br)C=CC=C1)N1CCN(C2=CC=C([N+](=O)O)C=C2)CC1 Chemical compound O=C(C1=C(Br)C=CC=C1)N1CCN(C2=CC=C([N+](=O)O)C=C2)CC1 CNEGXBKNLZCOBN-UHFFFAOYSA-N 0.000 description 1
- YKODGHXDJGBPQZ-UHFFFAOYSA-N O=C(C1=CC=C(Cl)C(Cl)=C1)N1CCN(C2=CC=C([N+](=O)[O-])C=C2)CC1 Chemical compound O=C(C1=CC=C(Cl)C(Cl)=C1)N1CCN(C2=CC=C([N+](=O)[O-])C=C2)CC1 YKODGHXDJGBPQZ-UHFFFAOYSA-N 0.000 description 1
- ONQYXJNDMVJGBK-UHFFFAOYSA-N O=C(C1=CSC2=C1C=CC=C2)N1CCN(C2=CC=C(Cl)C(Cl)=C2)CC1 Chemical compound O=C(C1=CSC2=C1C=CC=C2)N1CCN(C2=CC=C(Cl)C(Cl)=C2)CC1 ONQYXJNDMVJGBK-UHFFFAOYSA-N 0.000 description 1
- IAAUEIINUBBPMI-UHFFFAOYSA-N O=C(COC1=CC=C([N+](=O)[O-])C=C1)NC1=CC(F)=CC=C1 Chemical compound O=C(COC1=CC=C([N+](=O)[O-])C=C1)NC1=CC(F)=CC=C1 IAAUEIINUBBPMI-UHFFFAOYSA-N 0.000 description 1
- HAOXWGKZWIWPDU-UHFFFAOYSA-N O=C(NC(=S)N1CCN(C2=CC=CC=C2)CC1)C1=CC=CC=C1 Chemical compound O=C(NC(=S)N1CCN(C2=CC=CC=C2)CC1)C1=CC=CC=C1 HAOXWGKZWIWPDU-UHFFFAOYSA-N 0.000 description 1
- RJZQFDHLTNDMGK-UHFFFAOYSA-N O=C(NCCC12CC3CC(CC(C3)C1)C2)C1=CC=C(S(=O)(=O)N2CCOCC2)C=C1 Chemical compound O=C(NCCC12CC3CC(CC(C3)C1)C2)C1=CC=C(S(=O)(=O)N2CCOCC2)C=C1 RJZQFDHLTNDMGK-UHFFFAOYSA-N 0.000 description 1
- DJGZEJHCLVHBOA-UHFFFAOYSA-N O=C(OC1=CC=CC(NC2=NC=NC3=CC=CC=C32)=C1)C1=CC=CS1 Chemical compound O=C(OC1=CC=CC(NC2=NC=NC3=CC=CC=C32)=C1)C1=CC=CS1 DJGZEJHCLVHBOA-UHFFFAOYSA-N 0.000 description 1
- GXZQAURUXKDMHT-UHFFFAOYSA-N O=C1C2=C(N=C(SCC3=CC=NC=C3)N1C1=CC=CC=C1)SC1=C2CCCC1 Chemical compound O=C1C2=C(N=C(SCC3=CC=NC=C3)N1C1=CC=CC=C1)SC1=C2CCCC1 GXZQAURUXKDMHT-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/18—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof
- A01N37/22—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing the group —CO—N<, e.g. carboxylic acid amides or imides; Thio analogues thereof the nitrogen atom being directly attached to an aromatic ring system, e.g. anilides
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/02—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms
- A01N43/04—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom
- A01N43/06—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings
- A01N43/12—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one or more oxygen or sulfur atoms as the only ring hetero atoms with one hetero atom five-membered rings condensed with a carbocyclic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/36—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom five-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
- A01N43/42—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/58—1,2-Diazines; Hydrogenated 1,2-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/60—1,4-Diazines; Hydrogenated 1,4-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/713—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with four or more nitrogen atoms as the only ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/84—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms six-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
- A61K31/166—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide having the carbon of a carboxamide group directly attached to the aromatic ring, e.g. procainamide, procarbazine, metoclopramide, labetalol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
- A61K31/167—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide having the nitrogen of a carboxamide group directly attached to the aromatic ring, e.g. lidocaine, paracetamol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/235—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group
- A61K31/24—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids having an aromatic ring attached to a carboxyl group having an amino or nitro group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/38—Heterocyclic compounds having sulfur as a ring hetero atom
- A61K31/381—Heterocyclic compounds having sulfur as a ring hetero atom having five-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/402—1-aryl substituted, e.g. piretanide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/50—Pyridazines; Hydrogenated pyridazines
- A61K31/5025—Pyridazines; Hydrogenated pyridazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/513—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim having oxo groups directly attached to the heterocyclic ring, e.g. cytosine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/517—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with carbocyclic ring systems, e.g. quinazoline, perimidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
Definitions
- Candida albicans is the most widespread fungal pathogen of humans and one of the most frequent hospital-acquired infections (Gudlaugsson, Clin. Infect. Dis. 37:1172-1177, 2003; Miller, Clin. Infect. Dis. 15:523-530, 2001; Pappas, Clin. Infect. Dis. 37:634-643, 2003).
- the estimated annual cost of treating nosocomial Candida infections exceeds $1 billion per year (Miller, supra; Pappas, supra).
- As an opportunistic pathogen it is responsible for common clinical problems including, e.g., oral thrush and vaginitis, but can also lead to life-threatening systemic infections (candidiasis) in immunocompromised individuals (Fidel, Clin. Microbiol.
- Adhesion to surfaces is the first critical step in establishing a fungal infection.
- Candida cells with a planktonic “yeast” morphology initiate adhesion, and a subsequent transition from yeast to hyphal morphology contributes to invasion of the host tissue and formation of biofilms (Bendel, Crit. Care Med. 31:501-507, 2003; Saville, Eukaryot. Cell 2:1053-1060, 2003; Lo et al., Cell 90:939-949, 1997; Finkel, Nat. Rev. Microbiol. 9:109-118, 2010).
- Biofilm formation is a medically crucial step in pathogenesis, because biofilm-associated infections normally do not respond to conventional treatment, and because systemic candidiasis usually results from biofilms originating on intravascular devices and catheters (Douglas, Trends Microbiol. 11:30-36, 2003; Blankenship, Curr. Opin. Microbiol. 9:588-594, 2006; Nobile et al., Eukaryot. Cell 5:1604-1610, 2006).
- Candida cells released from these devices can be disseminated into the bloodstream, where fungal organisms adhere to endothelial surfaces and then penetrate blood vessels, spreading infection to multiple organs. Because biofilm-associated medical devices are resistant to treatment with existing therapeutics, it is often necessary to remove the devices in order to completely treat infections (Bauters, J. Clin. Microbiol. 40:1838-1839, 2002).
- the invention features methods of using a compound having a structure according to formula (I),
- n is an integer between 0-4;
- each R 1 when present, is, independently, OH, halogen, optionally substituted C1-C6 alkyl, or two R 1 on the same carbon combine to form an oxo group;
- Ar 1 is optionally substituted phenyl or optionally substituted monocyclic 5- or 6-membered heteroaryl
- Ar 2 is optionally substituted phenyl or optionally substituted heteroaryl.
- n 1 or 2.
- each R 1 when present, is optionally substituted C1-C6 alkyl.
- the compound has a structure according to formula (I-a),
- n 0 or 1.
- Ar 1 is a phenyl that includes 1 or 2 substituents that are electron-withdrawing (e.g., F, Cl, Br, I, CN, or NO 2 ). In certain embodiments, Ar 1 is a phenyl having a NO 2 substituent. In other embodiments, Ar 1 includes 1, 2, or 3 substituents independently selected from halogen and unsubstituted C1-C6 alkyl.
- Ar 2 is unsubstituted phenyl.
- Ar 2 is optionally substituted benzothiophene or optionally substituted phenyl. In certain embodiments, Ar 2 is unsubstituted phenyl or unsubstituted benzothiophene. In other embodiments, Ar 2 is phenyl having 1, 2, or 3 substituents selected, independently, from NO 2 , CN, optionally substituted C1-C6 alkyl, or halogen. In further embodiments, the substituents are, independently, selected from F, Cl, Br, and unsubstituted C1-C6 alkyl.
- the compound has a structure according to formula (II),
- R 1 is H or optionally substituted C1-C6 alkyl
- each Ar 1 and Ar 2 is, independently, an optionally substituted phenyl group
- L 1 is a covalent bond, —O(CH 2 ) n —, or —(CH 2 ) n O—, wherein n is an integer between 0-3.
- R 1 is H.
- L 1 is a covalent bond, —O(CH 2 )—, or —(CH 2 )O—.
- the compound has a structure according to formula (II-a),
- each of o and p is, independently, an integer between 1-3;
- each R 2 and R 3 is, independently, selected from halogen, optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy; CN, NO 2 , CO 2 H, or CO 2 R 4 ; and
- R 4 is optionally substituted C1-C6 alkyl or optionally substituted phenyl.
- o is 1 or 2.
- p is 1 or 2.
- L 1 is a bond
- each R 2 and R 3 is selected, independently, from F, Cl, Br, NO 2 , unsubstituted C1-C6 alkyl, CO 2 H, and CO 2 (unsubstituted C1-C6 alkyl).
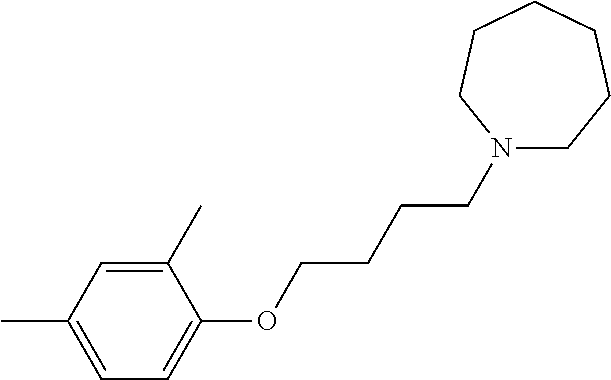
- the invention features compounds selected from the compounds of Table 3.
- variants of the compounds of Table 3 can be used in the methods described herein.
- a compound includes a phenyl group (e.g., any of Compounds (13)-(27))
- the phenyl group can be modified to be unsubstituted or to be substituted with 1, 2, 3, 4, or 5 substituents selected, independently, from optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy, halogen, CN, NO 2 , CO 2 H, and CO 2 R X , where R X is an optionally substituted C1-C6 alkyl or an optionally substituted phenyl.
- compounds of Table 3 that include a heteroaryl group can be modified in order that the heteroaryl group is unsubstituted or substituted with, e.g., 1 or 2 substituents selected, independently, from optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy, halogen, CN, NO 2 , CO 2 H, and CO 2 R X , where R X is an optionally substituted C1-C6 alkyl or an optionally substituted phenyl.
- the length of an alkylene linker in a Table 3 compound can also be varied.
- the alkylene linker can be varied such that it includes between 1-6 carbons (e.g., a C1-C6 alkylene, a C1-4 alkylene, a C1-2 alkylene, or even a C1 alkylene).
- the alkylene may be unsubstituted or substituted.
- carboxylic acid e.g., CO 2 H
- phenyl ester e.g., phenyl ester
- Regioisomers or linkage isomers of the compounds of Table 3 can also be used in the methods described herein.
- the invention in a second aspect, relates to a method of treating a microbial infection (e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly an infection by C. albicans ), bacterial, yeast, and/or mold infection) by administering to a subject (e.g., a mammal, such as a human) in need thereof a composition that includes a compound of formula (I):
- a microbial infection e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis,
- n is an integer between 0-4 (e.g., n is 1 or 2);
- each R 1 when present, is, independently, OH, halogen, optionally substituted C1-C6 alkyl, or two R 1 on the same carbon combine to form an oxo group;
- Ar 1 is optionally substituted phenyl or optionally substituted monocyclic 5- or 6-membered heteroaryl
- Ar 2 is optionally substituted phenyl or optionally substituted heteroaryl.
- each R 1 when present, is optionally substituted C1-C6 alkyl.
- the compound has a structure according to formula (I-a):
- Ar 1 is a phenyl that includes 1 or 2 substituents that are electron-withdrawing (e.g., F, Cl, Br, I, CN, or NO 2 ); Ar 1 is a phenyl having a NO 2 substituent; Ar 1 includes 1, 2, or 3 substituents independently selected from halogen and unsubstituted C1-C6 alkyl; Ar 2 is unsubstituted phenyl; Ar 2 is optionally substituted benzothiophene or optionally substituted phenyl; Ar 2 is unsubstituted phenyl or unsubstituted benzothiophene; and/or Ar 2 is phenyl having 1, 2, or 3 substituents selected, independently, from NO 2 , CN, optionally substituted C1-C6 alkyl, or halogen (e.g., the substituents of the phenyl of Ar 2 are, independently, selected from F, Cl, Br, and unsubstit
- the compound has a structure selected from one of the following:
- the method also includes administering more than one compound of formula I to the subject (e.g., one or more of compounds 1 to 9, in particular, at least compound 4 and one other compound of formula I).
- more than one compound of formula I e.g., one or more of compounds 1 to 9, in particular, at least compound 4 and one other compound of formula I.
- the composition includes a pharmaceutically acceptable carrier.
- treating, according to the method includes inhibiting and/or reducing (e.g., by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%)) at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of the microbial agent.
- at least 10% e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%
- at least one function e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth
- the invention features a method of treating a microbial infection (e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly an infection by C. albicans ), bacterial, yeast, and/or mold infection) by administering to a subject (e.g., a mammal, such as a human) in need thereof a composition comprising a compound of formula (II):
- a subject e.g., a mammal, such as a human
- a composition comprising a compound of formula (II):
- R 1 is H or optionally substituted C1-C6 alkyl
- each Ar 1 and Ar 2 is, independently, an optionally substituted phenyl group
- L 1 is a covalent bond, —O(CH 2 ) n —, or —(CH 2 ) n O—, wherein n is an integer between 0-3.
- R 1 is H; L 1 is a covalent bond, —O(CH 2 )—, or —(CH 2 )O—; and/or the compound has a structure according to formula (II-a):
- each of o and p is, independently, an integer between 1-3;
- each R 2 and R 3 is, independently, selected from halogen, optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy; CN, NO 2 , CO 2 H, or CO 2 R 4 ; and
- the method includes administering more than one compound of formula II to said subject (e.g., one or more of compounds 10-12, in particular, at least compound 12 and one other compound of formula II).
- more than one compound of formula II e.g., one or more of compounds 10-12, in particular, at least compound 12 and one other compound of formula II.
- the composition includes a pharmaceutically acceptable carrier.
- treating, according to the method includes inhibiting and/or reducing (e.g., by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%)) at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of the microbial agent.
- at least 10% e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%
- at least one function e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth
- the invention features a method of treating a microbial infection (e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly an infection by C. albicans ), bacterial, yeast, and/or mold infection) by administering to a subject (e.g., a mammal, such as a human) in need thereof a composition having a compound selected from one or more of the following:
- a microbial infection e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. kruse
- the method includes administering more than one of the compounds (e.g., 13 to 27) to the subject and/or the composition includes a pharmaceutically acceptable carrier.
- treating, according to the method includes inhibiting and/or reducing (e.g., by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%)) at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of the microbial agent.
- the invention features a composition that includes an anti-microbial compound incorporated therein or coated thereto, in which the compound is selected from one or more of the following:
- n is an integer between 0-4;
- each R 1 when present, is, independently, OH, halogen, optionally substituted C1-C6 alkyl, or two R 1 on the same carbon combine to form an oxo group;
- Ar 1 is optionally substituted phenyl or optionally substituted monocyclic 5- or 6-membered heteroaryl
- Ar 2 is optionally substituted phenyl or optionally substituted heteroaryl
- the compound is N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl-N-(2-aminoethyl)-2-aminoethyl
- the composition is a medical device (e.g., a cardiac-assist device, an artificial heart valve, a catheter, a central line, an intravenous (IV) line, a joint, a stent, a prosthetic implant, a pacemaker, a conduit, a cannula, an appliance, a scaffold, an artificial sphincter, a pessary, a tube, a drain, a trochar or plug, an implant, a rod, a screw, an orthopedic or implantable prosthetic device or appliance, a suture, a drug delivery device, an oral implant, a denture, and/or a brace; and/or a cuff, a dressing material, a mesh, a hernia patch, a wound dressing, a bandage, a syringe, and/or gloves; and/or a household product, a cosmetic product, a pharmaceutical product, a washing or cleaning formulation, a medical device surface, a medical device material
- IV intrave
- the anti-microbial compound reduces and/or inhibits at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of a microbial agent (e.g., a fungus (e.g., a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly C. albicans ), a bacteria, yeast, and/or a mold).
- a microbial agent e.g., a fungus (e.g., a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. gla
- the compound is incorporated into a thermoset, thermoplastic, elastomeric, and/or crosslinked polymer (e.g., a polymer selected from a polyolefin, polyamide, polyurethane, polyacrylate, polyacrylamide, polycarbonate, polystyrene, polyvinyl acetates, polyvinyl alcohol, polyester, halogenated vinyl polymer, such as polyvinyl chloride (PVC), a natural or synthetic rubber, an alkyd resin, an epoxy resin, an unsaturated polyester, an unsaturated polyamide, a polyimide, a silicone, a carbamate containing polymer, a fluorinated polymer, a crosslinkable acrylic resin, such as a substituted acrylic ester, an epoxy acrylate, a urethane acrylate, and/or a polyester acrylate, and/or a block co-polymer thereof).
- the composition inhibits and/or reduces adhesion of a fung
- the invention in a sixth aspect, relates to a method for achieving an antimicrobial, preservative, and/or microorganism adhesion inhibiting effect for the protection within an article and/or material (e.g., a medical device) or on the surface of an article and/or material (e.g., a medical device) that includes applying the composition described in the fifth aspect, or an adduct or salt thereof, to the article and/or material.
- an article and/or material e.g., a medical device
- an adduct or salt thereof e.g., a medical device
- the medical device is a cardiac-assist device, an artificial heart valve, a catheter, a central line, an intravenous (IV) line, a joint, a stent, a prosthetic implant, a pacemaker, a conduit, a cannula, an appliance, a scaffold, an artificial sphincter, a pessary, a tube, a drain, a trochar or plug, an implant, a rod, a screw, an orthopedic or implantable prosthetic device or appliance, a suture, a drug delivery device, an oral implant, a denture, and/or a brace; and/or a cuff, a dressing material, a mesh, a hernia patch, a wound dressing, a bandage, a syringe, and/or gloves; and/or a household product, a cosmetic product, a pharmaceutical product, a washing or cleaning formulation, a medical device surface, a medical device material, a fabric, a plastic,
- the compound of the composition reduces and/or inhibits at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of a microbial agent (e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly an infection by C.
- a microbial agent e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis
- the composition achieves an anti-fungal effect when administered to the subject.
- the compound is incorporated into a thermoset, thermoplastic, elastomeric, and/or crosslinked polymer (e.g., a polymer selected from a polyolefin, polyamide, polyurethane, polyacrylate, polyacrylamide, polycarbonate, polystyrene, polyvinyl acetates, polyvinyl alcohol, polyester, halogenated vinyl polymer, such as polyvinyl chloride (PVC), a natural or synthetic rubber, an alkyd resin, an epoxy resin, an unsaturated polyester, an unsaturated polyamide, a polyimide, a silicone, a carbamate containing polymer, a fluorinated polymer, a crosslinkable acrylic resin, such as a substituted acrylic este
- the invention relates to a method of treating a microbial infection (e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly an infection by C.
- a microbial infection e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis
- compositions that include an Rtt109 inhibitor (e.g., one or more of KB7, PDK38, PDK9, PDK23, PDK47, PDK13, PDK17, PDK35, and/or PDK36, in particular KB7, PDK23, and/or PDK13; preferably KB7).
- an Rtt109 inhibitor e.g., one or more of KB7, PDK38, PDK9, PDK23, PDK47, PDK13, PDK17, PDK35, and/or PDK36, in particular KB7, PDK23, and/or PDK13; preferably KB7.
- treating, according to the method includes inhibiting and/or reducing (e.g., by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%)) at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of the microbial agent.
- at least 10% e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100%
- at least one function e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth
- the invention relates to a composition that includes incorporated therein or coated thereto an Rtt109 inhibitor (e.g., one or more of KB7, PDK38, PDK9, PDK23, PDK47, PDK13, PDK17, PDK35, and/or PDK36, in particular KB7, PDK23, and/or PDK13; preferably KB7).
- an Rtt109 inhibitor e.g., one or more of KB7, PDK38, PDK9, PDK23, PDK47, PDK13, PDK17, PDK35, and/or PDK36, in particular KB7, PDK23, and/or PDK13; preferably KB7.
- the composition is a medical device (e.g., a cardiac-assist device, an artificial heart valve, a catheter, a central line, an intravenous (IV) line, a joint, a stent, a prosthetic implant, a pacemaker, a conduit, a cannula, an appliance, a scaffold, an artificial sphincter, a pessary, a tube, a drain, a trochar or plug, an implant, a rod, a screw, an orthopedic or implantable prosthetic device or appliance, a suture, a drug delivery device, an oral implant, a denture, and/or a brace; and/or a cuff, a dressing material, a mesh, a hernia patch, a wound dressing, a bandage, a syringe, and/or gloves; and/or a household product, a cosmetic product, a pharmaceutical product, a washing or cleaning formulation, a medical device surface, a medical device material,
- IV intra
- the Rtt109 inhibitor reduces and/or inhibits at least one function (e.g., adhesion, yeast-to-hyphal morphological transition, biofilm formation, and/or growth) of a microbial agent (e.g., a fungus (e.g., a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , and/or C. dubliniensis , particularly C. albicans ), a bacteria, yeast, and/or a mold).
- a microbial agent e.g., a fungus (e.g., a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. gla
- the Rtt109 inhibitor is incorporated into a thermoset, thermoplastic, elastomeric, and/or crosslinked polymer (e.g., a polymer selected from a polyolefin, polyamide, polyurethane, polyacrylate, polyacrylamide, polycarbonate, polystyrene, polyvinyl acetates, polyvinyl alcohol, polyester, halogenated vinyl polymer, such as polyvinyl chloride (PVC), a natural or synthetic rubber, an alkyd resin, an epoxy resin, an unsaturated polyester, an unsaturated polyamide, a polyimide, a silicone, a carbamate containing polymer, a fluorinated polymer, a crosslinkable acrylic resin, such as a substituted acrylic ester, an epoxy acrylate, a urethane acrylate, and/or a polyester acrylate, and/or a block co-polymer thereof).
- the Rtt109 inhibitor inhibits and/or reduces
- the invention relates to a method for screening a test compound for an antimicrobial, preservative, and/or microorganism adhesion inhibiting effect for the protection within an article and/or material (e.g., a medical device) or on the surface of an article and/or material (e.g., a medical device) and/or for an antimicrobial effect for the treatment of a microbial infection (e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C.
- a microbial infection e.g., a fungal (e.g., an infection caused by a Candida spp., such as C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C.
- kefyr, C. guilliermondii , and/or C. dubliniensis particularly an infection by C. albicans ), bacterial, yeast, and/or mold infection
- a subject e.g., a mammal, such as a human
- the test compound may reduce or inhibit microbial growth by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100% (e.g., kills the microbial agent), relative to a control, or acts as a microbistatic or microbicidal agent.
- the assay may identify the test compound as an anti-fungal agent that reduces or inhibits fungal growth by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100% (e.g., kills the fungus), relative to a control, or that acts as a fungistatic or fungicidal agent.
- alkyl As used herein, the term “alkyl,” “alkenyl” and “alkynyl” include straight-chain, branched-chain and cyclic monovalent substituents, as well as combinations of these, containing only C and H when unsubstituted. Examples include methyl, ethyl, isobutyl, cyclohexyl, cyclopentylethyl, 2-propenyl, 3-butynyl, and the like.
- cycloalkyl represents a monovalent saturated or unsaturated non-aromatic cyclic alkyl group having between three to nine carbons (e.g., a C3-C9 cycloalkyl), unless otherwise specified, and is exemplified by cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, bicyclo[2.2.1.]heptyl, and the like.
- the cycloalkyl group includes one carbon-carbon double bond
- the cycloalkyl group can be referred to as a “cycloalkenyl” group.
- Exemplary cycloalkenyl groups include cyclopentenyl, cyclohexenyl, and the like
- the alkyl, alkenyl and alkynyl groups contain 1-12 carbons (e.g., C1-C12 alkyl) or 2-12 carbons (e.g., C2-C12 alkenyl or C2-C12 alkynyl).
- the alkyl groups are C1-C8, C1-C6, C1-C4, C1-C3, or C1-C2 alkyl groups; or C2-C8, C2-C6, C2-C4, or C2-C3 alkenyl or alkynyl groups.
- any hydrogen atom on one of these groups can be replaced with a substituent as described herein.
- alkylene alkenylene
- alkynylene alkynylene
- alk divalent or trivalent groups having a specified size, typically C1-C2, C1-C3, C1-C4, C1-C6, or C1-C8 for the saturated groups (e.g., alkylene or alk) and C2-C3, C2-C4, C2-C6, or C2-C8 for the unsaturated groups (e.g., alkenylene or alkynylene).
- saturated groups e.g., alkylene or alk
- C2-C3, C2-C4, C2-C6, or C2-C8 unsaturated groups
- C ⁇ O is a C1 alkylene that is substituted by ⁇ O, for example.
- alkaryl represents an aryl group, as defined herein, attached to the parent molecular group through an alkylene group, as defined herein
- alkheteroaryl refers to a heteroaryl group, as defined herein, attached to the parent molecular group through an alkylene group, as defined herein.
- the alkylene and the aryl or heteroaryl group are each optionally substituted as described herein.
- alkoxy represents a chemical substituent of formula —OR, where R is an optionally substituted alkyl group (e.g., C1-C6 alkyl group), unless otherwise specified.
- the alkyl group can be substituted, e.g., the alkoxy group can have 1, 2, 3, 4, 5, or 6 substituent groups as defined herein.
- alkaryloxy represents a chemical substituent of formula —OR, where R is an optionally substituted alkaryl group.
- anti-fungal agent or “anti-fungal compound” is meant an agent that reduces or inhibits fungal growth by at least 10% (e.g., at least 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or 100% (e.g., kills the fungus)).
- an anti-fungal agent of the invention may be “fungistatic” and/or “fungicidal.”
- “Pathogenic fungi” include fungi that can cause disease in humans and have two primary morphological appearances. More than 100 species of fungi have pathogenic potential for humans. Their complex cell wall provides a protective covering that have made fungi impervious to many anti-microbial agents. Disease presentation can often be caused by morphological manifestation of fungus. For example, fungal mycelia will develop into fungal “bezoars” that can obstruct, for example, the urinary system. Budding yeast forms can be associated with infection or abscess.
- “Aromatic” moiety or “aryl” moiety refers to any monocyclic or fused ring bicyclic system which has the characteristics of aromaticity in terms of electron distribution throughout the ring system and includes a monocyclic or fused bicyclic moiety such as phenyl or naphthyl; “heteroaromatic” or “heteroaryl” also refers to such monocyclic or fused bicyclic ring systems containing one or more heteroatoms selected from O, S, and N. The inclusion of a heteroatom permits inclusion of 5-membered rings to be considered aromatic as well as 6-membered rings.
- typical aromatic/heteroaromatic systems include pyridyl, pyrimidyl, indolyl, benzimidazolyl, benzotriazolyl, isoquinolyl, quinolyl, benzothiazolyl, benzofuranyl, thienyl, furyl, pyrrolyl, thiazolyl, oxazolyl, isoxazolyl, benzoxazolyl, benzoisoxazolyl, imidazolyl, and the like. Because tautomers are theoretically possible, phthalimido is also considered aromatic.
- the ring systems contain 5-12 ring member atoms or 6-10 ring member atoms.
- the aromatic or heteroaromatic moiety is a 6-membered aromatic rings system optionally containing 1-2 nitrogen atoms. More particularly, the moiety is an optionally substituted phenyl, pyridyl, indolyl, pyrimidyl, pyridazinyl, benzothiazolyl or benzimidazolyl, pyrazolyl, imidazolyl, isoxazolyl, thiazolyl, benzothiazolyl, or indolyl. Even more particularly, such moiety is phenyl, pyridyl, or pyrimidyl and even more particularly, it is phenyl.
- carboxylic acid ester is meant a group having the structure —CO 2 R′, where R′ is selected from H, optionally substituted C 1-6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocyclyl, optionally substituted aryl, or optionally substituted heteroaryl.
- R′ is selected from H, optionally substituted C 1-6 alkyl, optionally substituted cycloalkyl, optionally substituted heterocyclyl, optionally substituted aryl, or optionally substituted heteroaryl.
- R′ is not H, R may be unsubstituted or substituted with, for example, 1, 2, 3, 4, 5, or 6 substituents.
- Halo may be any halogen atom, especially F, Cl, Br, or I, and more particularly it is fluoro or chloro.
- an effective amount is meant an amount of a compound of the invention that can be used to treat, prevent, delay the onset of, or inhibit the progression of a fungal infection (e.g., use as an anti-fungal agent or compound).
- the effective amount of an active compound(s) used to practice therapeutic or prophylactic methods of the invention e.g., to treat, inhibit, or reduce conditions caused by or contributed to by a fungal infection
- for fungistatic or fungicidal methods of the invention varies depending upon the manner of administration, the age, body weight, and general health of the subject or the particular application of the active compound(s) (e.g., as a coating or incorporated into a device or other material of the invention).
- Those of skill in the art are capable to determining an amount of an active compound of the invention that can be applied as the “effective” amount.
- fungal infection or “mycoses” is meant an invasion of a host animal by fungal cells.
- the infection may include the excessive growth of fungi that are normally present in or on the animal, or growth of fungi that are not normally present in or on the animal.
- a fungal infection can be any situation in which the presence of a fungal population is detrimental or damaging to a host animal.
- fungal infection includes a primary fungal infection as well as an opportunistic fungal infection.
- An “oxo” group is a substituent having the structure C ⁇ O, where there is a double bond between a carbon and an oxygen atom.
- Typical optional substituents on aromatic or heteroaromatic groups include independently halo (e.g., F, Cl, Br, or I), CN, NO 2 , CF 3 , OCF 3 , COOR′, CONR′ 2 , OR′, SR′, SOR′, SO 2 R′, NR′ 2 , NR′(CO)R′, NR′C(O)OR′, NR′C(O)NR′ 2 , NR′SO 2 NR′ 2 , or NR′SO 2 R′, wherein each R′ is independently H or an optionally substituted group selected from alkyl, alkenyl, alkynyl, heteroalkyl, heteroalkenyl, heteroalkynyl, heteroaryl, and aryl (all as defined above); or the substituent may be an optionally substituted group selected from alkyl, alkenyl, alkynyl, heteroalkyl, heteroalkenyl, heteroalkynyl, aryl, heteroaryl, O
- non-aromatic groups e.g., alkyl, alkenyl, and alkynyl groups
- a non-aromatic group may also include a substituent selected from ⁇ O and ⁇ NOR′ where R′ is H or an optionally substituted group selected from alkyl, alkenyl, alkynyl, heteroalkyl, heteroalkenyl, heteroalkynyl, heteroaryl, and aryl (all as defined above).
- Electron-withdrawing substituents are those substituents that decrease electron density at the site of attachment. Electron-withdrawing substituents typically positive Hammett substituent constants. Exemplary electron-withdrawing substituents include halogen (e.g., F, Cl, Br, or I), NO 2 , CN, carbonyl groups (e.g., aldehydes, ketones, carboxylic acids, carboxylic esters, and acyl chlorides), haloalkyls, sulfonic acids, sulfonamides, sulfonate esters, and quaternary ammonium salts.
- halogen e.g., F, Cl, Br, or I
- NO 2 e.g., N-phenyl
- carbonyl groups e.g., aldehydes, ketones, carboxylic acids, carboxylic esters, and acyl chlorides
- haloalkyls e.g., sulfonic acids,
- a substituent group e.g., alkyl, alkenyl, alkynyl, or aryl (including all heteroforms defined above) may itself optionally be substituted by additional substituents.
- additional substituents e.g., alkyl, alkenyl, alkynyl, or aryl (including all heteroforms defined above
- alkyl may optionally be substituted by the remaining substituents listed as substituents where this makes chemical sense, and where this does not undermine the size limit of alkyl per se; e.g., alkyl substituted by alkyl or by alkenyl would simply extend the upper limit of carbon atoms for these embodiments, and is not included.
- alkyl substituted by aryl, amino, halo and the like would be included.
- the group may be substituted with 1, 2, 3, 4, 5, or 6 substituents.
- Optional substituents include, but are not limited to: C1-C6 alkyl or heteroaryl, C2-C6 alkenyl or heteroalkenyl, C2-C6 alkynyl or heteroalkynyl, halogen; aryl, heteroaryl, azido (—N 3 ), nitro (—NO 2 ), cyano (—CN), acyloxy(—OC( ⁇ O)R′), acyl (—C( ⁇ O)R′), alkoxy (—OR′), amido (—NR′C( ⁇ O)R′′ or —C( ⁇ O)NRR′), amino (—NRR′), carboxylic acid (—CO 2 H), carboxylic ester (—CO 2 R′), carbamoyl (—OC( ⁇ O)NR′R′′ or —
- subject can be a human, non-human primate, or other mammal, such as but not limited to dog, cat, horse, cow, pig, turkey, goat, fish, monkey, chicken, rat, mouse, and sheep.
- the term “treat,” “treated,” or “treating” when used with respect to a disorder, such as an infectious disease (e.g., a fungal infection) refers to a therapeutic or prophylactic treatment that increases the resistance of a subject to development of the disease (e.g., to infection with a pathogen, such as a fungus) or, in other words, decreases the likelihood that the subject will develop the disease (e.g., become infected with the pathogen), that increases the ability of a subject that has developed disease (e.g., a pathogenic (e.g., fungal) infection) to fight the disease (e.g., reduce or eliminate at least one symptom typically associated with the infection) or prevent the disease from becoming worse, or that decreases, reduces, or inhibits at least one function of the pathogen (e.g., a fungus, such as Candida albicans ), such as the ability to adhere to a substrate, to form a biofilm, to switch from a yeast to hyp
- FIGS. 1A-1E Chemical inhibition of Candida albicans adhesion to polystyrene.
- FIG. 1A is a photograph showing a crystal violet-stained plate from a small molecule screen. Wild-type C. albicans strain SC5314 or adhesion-defective edt1 ⁇ / ⁇ cells were plated in the presence of 1% DMSO vehicle (positive control) or small molecules at 50 ⁇ M, followed by extensive washing to remove unbound cells. Cells that remained were then visualized by staining with crystal violet and quantified by absorbance at 590 nm. Screen details and data normalization are described in the Materials and Methods. FIG.
- FIG. 1B is a graph showing the results of an alamarBlue-based polystyrene adhesion assay with compounds at 25 ⁇ M. Using the vital dye alamarBlue as the detection reagent, the 26 primary candidates from the screen were retested at 25 ⁇ M, alongside a DMSO positive control. The mean and standard deviation of data from 8 wells were measured.
- FIG. 1C is a graph showing the results of a retest of polystyrene adhesion assay of FIG. 1B with compounds at 7.5 ⁇ M.
- FIG. 1D is a graph showing the results of a GFP-based adhesion assay. C.
- FIG. 1E is a series of photographs showing fluorescence microscopy results of the GFP assay. Wells from the experiment in FIG. 1D were photographed using a 20 ⁇ objective and FITC filters.
- FIGS. 2A-2D Compound #4 is a non-toxic inhibitor of adhesion by multiple Candida species and reduces fungal binding to human A549 cell monolayers.
- FIG. 2A is a graph showing the results of fluorescence detection of adhesion of multiple Candida species to A549 cells. Human A549 cells were grown to confluence on 48-well plates. SC5314-GFP, edt1 ⁇ / ⁇ -GFP or untagged SC5314 were added to triplicate wells and incubated at 37° C. for 90 min with 1% DMSO or 25 ⁇ M of the indicated compounds in 1% DMSO. Wells were washed extensively to remove unbound fungi, and GFP fluorescence was measured on a plate reader. FIG.
- FIG. 2B is a series of photographs showing florescence microscopy of C. albicans adhered to A549 monolayers. Representative bright-field (DIC) and corresponding GFP fluorescence images from the experiment in FIG. 2A .
- FIG. 2C is a graph showing the results of a human cell toxicity assay. A549 cells were coincubated with 1% DMSO, 250, 100, or 50 ⁇ M compound #4 or 1% sodium azide for 24 hrs, and cell viability was then measured using alamarBlue.
- FIG. 2D is a graph showing that adhesion by C. dublinensis is inhibited by compound #4. Adhesion assay as in FIG. 1B , with 25 ⁇ M compound #4 tested.
- FIGS. 3A-3C Compound #4 has distinct activities from structurally related piperazinyl quinolones.
- FIG. 3A shows the structures of compounds. #9 was analyzed to determine if the aryl group substitution of compound #4 is important. The piperazinyl quinolone #Q1 was described (Youngsaye, Bio & Med Chem. Ltrs. 21: 5502-5505, 2011) as a compound that reverses fluconazole resistance.
- Compound #Q2 is a piperazinyl quinolone with the same aryl group substitutions as #4.
- FIG. 3B is a graph showing that compounds #Q1 and Q2 do not inhibit C. albicans adhesion to polystyrene. Assay performed as in FIG.
- FIG. 3C is a graph showing that compounds #Q1 and Q2 do not inhibit C. albicans adhesion to human A549 cells. Assay performed as in FIG. 2A , with 25 ⁇ M compounds.
- FIGS. 4A-4B Chemical modulation of Candida albicans morphogenesis.
- FIG. 4A is a series of photographs showing microscopic analysis of hyphal development in the presence of the indicated compounds at 12.5 ⁇ M. Cells containing an HWP1 promoter-driven RFP reporter were grown in Spider media for 16 hours and photographed using DIC or fluorescence microscopy (RFP) as indicated (64 ⁇ objective).
- FIG. 4B is a series of photographs showing the dose-dependent effects of compound #4, 12 and Q1 on hyphal development. Compounds at 10, 5, 2.5 ⁇ M as indicated were tested as above.
- FIGS. 5A-5C Effect of small molecules on biofilm formation in vitro.
- FIG. 5A is a series of photographs showing biofilm formation on silicone elastomers. Biofilm assay was performed with SC5314-GFP and edt1 ⁇ / ⁇ -GFP and photographed after 60 hrs. Indicated compounds were added at 50 ⁇ M. Turbid medium indicates planktonic cells unattached to silicone elastomers when biofilm formation is inhibited. Clear medium with the cell attached to the silicone elastomer indicate establishment of functional biofilm. Each treatment was done in triplicate.
- FIG. 5B is a graph showing quantitation of turbidity of medium.
- FIG. 5C is a graph showing quantitation of dry weight of biofilm. Dry weight in mg of air-dried silicone elastomers.
- FIG. 6A-6B Effects on metazoan hosts.
- FIG. 6A is a graph showing the lifespan of C. elegans exposed to C. albicans treated with 12.5 ⁇ M compound #4 (lower graph line) or 12.5 ⁇ M fluconazole (upper graph line) compared to untreated (middle graph line). Worms treated with compound #4 display significantly increased survival compared to untreated worms (p ⁇ 0.012 for drug treated worms compared to untreated).
- FIG. 6B is a series of photographs showing microscopic analysis of ex vivo vaginal mucosal biofilm formation. Mice were administered estrogen and vaginae were harvested.
- Tissues were either uninfected, or pretreated with DMSO or 50 ⁇ M compound #4, and then inoculated with C. albicans SC5314 (10 6 blastoconidia) and incubated for 24 h.
- Tissues were bisected and processed for scanning electron microscopy (SEM), or confocal microscopy (CM) after staining with Calcofluor to detect yeast (blue) and Concavalin A to visualize extracellular matrix (red).
- FIG. 7 is a graph showing Z′-factor measurement for Alamar Blue-based adhesion assay. Overnight cultures of SC5314 cells were diluted to 0.5 OD/ml in fresh SCM and plated onto an Immulon 2HB 96 well microtiter plate. 48 wells were treated with either 1% DMSO or 25 ⁇ M compound #4, and alamarBlue-based adhesion assays were performed as described in the Methods. Z′ factor was calculated as described (Zhang et al., J. Biomol. Screen 4:67-73, 1999).
- FIGS. 8A-8B Characterization of compound #4.
- FIG. 8A is a graphs showing the IC50 for compound #4 measured using GFP adhesion assay. Assay performed as in FIG. 1D with the indicated concentrations of compound #4. Mean and standard deviations from 4 replicate well measurements are shown.
- FIG. 8B is a graph showing the effect of compound #4 on growth rate of SC5314 in liquid culture. SC5314 was grown in liquid YPD in a 30° C. shaker in the presence of 1% DMSO or compounds #4, 5, 6, 7 or 8 at 50 ⁇ M and the OD 600 of the culture was measured at the indicated times. Cultures were also observed in the microscope, and we confirmed that the cells retained the budding yeast morphology during the course of this experiment.
- FIGS. 9A-9B Test of synergy between scaffold 1 and 2.
- FIG. 9A shows the chemical structures of the two scaffold groups identified.
- FIG. 9B is a graph showing that scaffold 1 compound #4 does not synergize with scaffold 2 compound #12 in inhibiting adhesion to polystyrene. Assay performed as in FIG. 1B , with the indicated compound concentrations.
- FIGS. 10A-10B are graphs showing that, unlike compound #Q1, compound #4 does not synergize with low concentrations fluconazole to inhibit growth of clinical Candida strains.
- Candida strains were identified from AIDS patients (3919, an initial progenitor isolate, and the subsequently isolated 4617 and 4639 strains). These were coincubated in the presence of either compounds #4, #Q1, fluconazole alone or a combination of fluconazole with either #4 or #Q1 for 24 hrs in polystyrene plates at 37° C. Viability was assessed using alamarBlue reagent as described (Youngsaye, supra).
- FIG. 10A shows the results using strain 4617
- FIG. 10B shows the results using strain 4639.
- FIGS. 11A-11D Compound #4 can coat surfaces and reduce subsequent fungal adhesion.
- FIG. 4A is a graph showing the results of pretreatment of polystyrene with candidate compounds. Indicated compounds at 50 ⁇ M were incubated in polystyrene 96-well plates overnight. Wells were washed three times with 100 ⁇ l of PBS. C. albicans adhesion was then measured using the vital dye alamarBlue, with no additional drug treatment.
- FIG. 11B is a graph showing titration of compounds #1, 4 and 9 for pre-binding to polystyrene. Indicated concentrations of compounds were pre-incubated with polystyrene plates as above prior to the adhesion assays.
- FIG. 4A is a graph showing the results of pretreatment of polystyrene with candidate compounds. Indicated compounds at 50 ⁇ M were incubated in polystyrene 96-well plates overnight. Wells were washed three times with 100 ⁇
- FIG. 11C is a graph showing the inhibition of adhesion by pretreatment of C. albicans SC5314 cells with compound #4 prior to introduction to plastic.
- SC5314 cells were treated with 25 ⁇ M compound #4 or DMSO in liquid culture prior to their introduced into polystyrene plates. Cells were washed with PBS prior to resuspension into SCM and seeding into 96-well plates for adhesion assays using alamarBlue detection of remaining cells. The pretreated cells were assayed alongside a standard adhesion assay in which compound #4 and the cells were coincubated with the plastic simultaneously.
- FIG. 11D is a graph showing the results of an assay performed as in FIG. 11C , except that SC5314-GFP cells were used, and then detected via fluorescence measurement. edt1 indicates non-adherent mutant cells, PBS indicates background fluorescence in absence of cells.
- FIG. 12 Growth of Candida albicans in 96-well plates in presence of DMSO. 5 ⁇ 10 3 cells were inoculated into 0.1 ml rich (YPD) media in the presence of the indicated concentration of DMSO and grown at 25° C. for 16 hours. Triplicate cultures were analyzed via a microplate reader.
- YPD 0.1 ml rich
- FIG. 13 A high-throughput dye-binding assay for Candida adhesion.
- FIG. 13 are photographs showing two different 96-well plates from the screen. The left-hand columns on each plate marked “edt1 ⁇ / ⁇ ” contained mutant C. albicans cells lacking the Edt1 protein required for efficient adhesion to surfaces, and serve as negative controls. The right-hand columns marked “DMSO” contained wild-type C. albicans cells and serve as positive controls. The wells in the middle 10 columns had compounds from the library added. Note that some display greatly reduced adhesion (arrows—Wells D8 and H9); these represent our primary candidates of interest. We have also observed many compounds that appear to promote adhesion (arrows—Wells E3 and H10); the high frequency of this class suggests these may result from non-specific aggregation effects of hydrophobic compounds.
- FIGS. 14A-14F Characterization of a multiwell acetyltransferase assay for Rtt109.
- FIG. 14A is a photograph showing recombinant Rtt109 and Vps75, either produced as single proteins (lanes 1 and 2) or as a complex in bacteria.
- FIG. 14B is a photograph showing 10 ⁇ g total nuclear protein from either wild-type or asf1 cells analyzed by immunoblotting with anti-H3K56ac rabbit sera. “H3” indicates full length H3, which is acetylated on K56 in the wt strain, and “H3 ⁇ ” indicates a proteolytic product. “*” indicates a cross-reactive species indicating equal loading of the two lanes.
- FIG. 14A is a photograph showing recombinant Rtt109 and Vps75, either produced as single proteins (lanes 1 and 2) or as a complex in bacteria.
- FIG. 14B is a photograph showing 10 ⁇ g total nuclear protein from either wild-type or
- FIG. 14C is a graph showing enzymatic detection of H3K56 ac in a 96 well plate, in the presence of the indicated amounts of enzyme.
- FIG. 14D is a graph comparing histone sources and measuring the background observed in the absence of added acetyl CoA, as in FIG. 14C .
- FIG. 14E is a graph showing DMSO sensitivity of the assay.
- FIG. 14F is a preliminary Z-factor score in the 96-well format, with 200 ng H3/H4 and 40 ng Rtt109/Vps75 per assay.
- FIG. 15 is a series of photographs showing that Rtt109 is required for C. albicans resistance to genotoxic agents.
- Five-fold serial dilutions of wild-type, heterozygous (rtt109 +/ ⁇ ) and homozygous (rtt109 ⁇ / ⁇ ) deletion strains were plated on rich YPD media, either lacking or containing the DNA alkylating agent MMS, or the oxidant hydrogen peroxide.
- FIG. 16 is a graph showing that rtt109 ⁇ / ⁇ mutants are deficient in causing pathogenicity in the murine candidiasis model.
- Balb/c female mice were infected with 1.5 ⁇ 10 5 yeast cells via venous tail injection.
- FIG. 17 is a graph showing the Z-score in the 384-well format.
- a 384-well plate of acetylation reactions was analyzed using the indicated conditions, with half of the reactions containing and half lacking acetyl-CoA. The Z-factor in this experiment was 0.85.
- FIG. 18 is a graph showing the results of a screen plate of 384 assays.
- the plate included positive and negative control wells (+ and ⁇ AcCoA, respectively); the Z-score for the control wells on this plate was 0.90.
- Triangles represent tests of library compounds. Note that two compounds inhibited the assay below the pictured 75% inhibition cutoff line.
- FIG. 19 is a graphs showing candidate compound titration in assays with Rtt109-Vps75. Acetylation relative to a vehicle only positive control for triplicate assays is shown.
- FIG. 20 is a graph showing titration of Asf1/H3/H4 in ELISA assays. Data from triplicate assays are shown.
- FIG. 21 is a graph showing candidate compound titration in assays with Rtt109+Asf1/H3/H4. Acetylation relative to a vehicle-only positive control for triplicate assays is shown.
- FIG. 22 shows the structures of candidate anti-fungal compounds.
- FIG. 23 is a graph showing the results of a fluorescence-based assay for histone acetylation by Rtt109+Vps75. Data from triplicate assays are shown performed under the same reaction conditions (187 nM H3/H4) as used in the ELISA.
- FIGS. 24A-24C KB7 is a specific inhibitor of Rtt109 HAT catalysis.
- FIG. 24A shows the structure of a candidate anti-fungal compound (“KB7”).
- FIG. 24B is a graph showing the IC50 curve for Rtt109-Vps75 catalysis. 50 nM Rtt109-Vps75 and 15 ⁇ M H3n21 peptide were incubated with DMSO (vehicle) or KB7 at the indicated concentrations for 5 minutes at 30° C. HAT reactions were initiated with 30 ⁇ M Acetyl coEnzyme A. Release of coEnzyme A was detected at various time points for 10 minutes to quantify reaction rates.
- FIG. 24C is a graph showing HAT activity of P300 and Gcn5 at the indicated KB7 concentrations. HAT reactions were performed as described above with 15 ⁇ g/ml P300 or Gcn5.
- FIGS. 25A and 25B KB7 specifically inhibits H3K56 acetylation by both Rtt109-Vps75 and Rtt109-Asf1 complexes.
- FIG. 25A is a graph showing that acetylation of H3K56 is inhibited by KB7. End-point HAT reactions were performed with 50 nM Rtt109, 300 ⁇ M (H3-H4) 2 tetramers, 50 nM Vps75 or 400 nM Asf1n at 30° C. for 30 minutes. Reactions were incubated with DMSO (vehicle) or 500 nM KB7 for 5 minutes prior to initiation with 15 ⁇ M acetyl coEnzyme A.
- DMSO vehicle
- 500 nM KB7 for 5 minutes prior to initiation with 15 ⁇ M acetyl coEnzyme A.
- FIGS. 26A-26D Compound #4 inhibits hyphal formation of Aspergillus fumigatus .
- FIGS. 26A-26B are photographs showing hyphal development occurs normally in RPMI tissue culture media in the absence of treatment ( FIG. 26A ) and in the presence of 1% DMSO (control; FIG. 26B ). Aspergillus hyphal development was inhibited in the presence of 50 ⁇ M and 100 ⁇ M compound #4 ( FIGS. 26C and 26D , respectively).
- FIGS. 27A and 27B Compound #4 inhibits proliferation of Cryptococcus gatti .
- FIGS. 27A and 27B are graphs showing that the growth of Cryptococcus gatti was inhibited by 50 ⁇ M and 100 ⁇ M compound #4 growth, whereas no effect on growth was observed in the untreated (control) and 1% DMSO treated samples.
- FIGS. 28A and 28B Chemical modulation of Candida albicans morphogenesis.
- FIGS. 28A and 28B show the time course of hyphal development of C. albicans ells grown in Spider media for the indicated periods of time in the presence of DMSO (control; FIG. 28A ) or Compound #4 ( FIG. 28B ).
- FIGS. 29A and 29B Hyphal morphogenesis in response to serum and Spider media and colony morphology.
- FIG. 29A is a series of photomicrographs showing the effect of compound #4 on SC5314 cells grown at 37° C. for the indicated time in Spider media or supplemented YNB+10% bovine serum. DMSO is a control.
- FIG. 29B shows the effect of compound #4 on colonly morphology of C. albicans SC5314 cells.
- Solid Spider media agar plates were overlayed with either 1% DMSO or 50 ⁇ M #4 .
- C. albicans SC5314 cells were spotted onto these plates and incubated at 37° C. for 2 days before photography. The upper images were taken with a digital camera; single colonies below were imaged using a microscope with 12.5 ⁇ magnification.
- FIGS. 30A-30E Compound #4 inhibits hyphal development induced by multiple signals.
- FIG. 30A is a schematic diagram of some of the signaling pathways that govern hyphal gene expression. For simplicity, only those genes investigated in this figure are shown.
- FIG. 30B is a series of photomicrographs showing the effects of compound #4 on constitutively filamentous mutant strains in the cAMP-PKA pathway. Wild-type SC5314, efg1-T206E, ras1-G13V, and ADH1pr-gpa2-Q355L cells were grown in the presence of 1% DMSO or 50 ⁇ M compound #4 in Spider medium at 37° C.
- FIG. 30C is a series of two photomicrographs showing that Compound #4 blocks filamentation induced by GlcNAc or constitutive overexpression of the GlcNac-activated transcription factor Cph1. Images obtained as in FIG. 30B .
- FIG. 30D is a series of photomicrographs showing that Compound #4 does not block genotoxic stress-induced filamentation. SC5314 cells grown in YPD at 30° C. in the presence of 50 mM hydroxyurea (HU) in combination with either 1% DMSO or 50 mM compound #4. Images obtained as in FIG. 30B .
- FIG. 30C is a series of two photomicrographs showing that Compound #4 blocks filamentation induced by GlcNAc or constitutive overexpression of the GlcNac-activated transcription factor Cph1. Images obtained as in FIG. 30B .
- FIG. 30D is a series of photomicrographs showing that Compound #4 does not block genotoxic stress-induced filamentation. SC5314 cells grown in
- 30E is a series of photomicrographs showing that Compound #4 inhibits the hyperfilamentous growth of a strain that constitutively overexpresses the Gpr1 protein. Colonies were grown on solid Spider media agar overlayed with either 1% DMSO or 50 ⁇ M #4 and incubated at 37° C. for 3 days. Colony edges were imaged with a 10 ⁇ objective.
- Pathogenesis by fungi begins with adhesion to host cells or implanted medical devices, followed by biofilm formation.
- assays to identify small molecules and other agents that inhibit adhesion of C. albicans to surfaces, such as polystyrene surfaces.
- Compounds we have identified using these assays also inhibit binding of C. albicans to cultured human epithelial cells, the yeast-to-hyphal morphological transition, biofilm formation on silicone mesh, and pathogenesis in a nematode infection model, and thee also alter fungal morphology in a mouse mucosal infection assay.
- Other compounds identified herein share some but not all of these activities. Therefore, we have identified compounds that can address various aspects of fungal pathogenesis, and that can be used as novel antifungal agents.
- the present invention provides compounds that are anti-microbial agents (e.g., anti-fungal agents) that can be used in the treatment or prophylaxis of a subject (e.g., a human) and in the treatment of surfaces and/or devices (e.g., medical devices).
- anti-microbial agents e.g., anti-fungal agents
- the compounds of the invention can be incorporated into materials used to produce devices (e.g., medical devices) and can be added to disinfectants, cosmetics, household products, textiles, and plastics.
- the compounds of the invention include compounds having the structure according to Formula (I):
- n is an integer between 0-4;
- each R 1 when present, is, independently, OH, halogen, optionally substituted C1-C6 alkyl, or two R 1 on the same carbon combine to form an oxo group;
- Ar 1 is optionally substituted phenyl or optionally substituted monocyclic 5- or 6-membered heteroaryl
- Ar 2 is optionally substituted phenyl or optionally substituted heteroaryl.
- n is 1 or 2
- each R 1 when present, is optionally substituted C1-C6 alkyl, and/or the compound has a structure according to formula (I-a):
- n 0 or 1.
- Ar 1 may be a phenyl that includes 1 or 2 substituents that are electron-withdrawing (e.g., F, Cl, Br, I, CN, or NO 2 ). In certain embodiments, Ar 1 may be a phenyl having a NO 2 substituent. In other embodiments, Ar 1 includes 1, 2, or 3 substituents independently selected from halogen and unsubstituted C1-C6 alkyl. In other embodiments, Ar 2 may be unsubstituted phenyl. In some other embodiments, Ar 2 may be optionally substituted benzothiophene or optionally substituted phenyl.
- Ar 2 may be unsubstituted phenyl or unsubstituted benzothiophene.
- Ar 2 may be phenyl having 1, 2, or 3 substituents selected, independently, from NO 2 , CN, optionally substituted C1-C6 alkyl, or halogen.
- the substituents may be, independently, selected from F, Cl, Br, and unsubstituted C1-C6 alkyl.
- the compounds of the invention also include compounds having the structure according to Formula (II):
- R 1 is H or optionally substituted C1-C6 alkyl
- each Ar 1 and Ar 2 is, independently, an optionally substituted phenyl group
- L 1 is a covalent bond, —O(CH 2 ) n —, or —(CH 2 ) n O—, wherein n is an integer between 0-3.
- R 1 is H
- L 1 is a covalent bond, —O(CH 2 )—, or —(CH 2 )O—
- the compound has a structure according to formula (II-a):
- each of o and p is, independently, an integer between 1-3;
- each R 2 and R 3 is, independently, selected from halogen, optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy; CN, NO 2 , CO 2 H, or CO 2 R 4 ; and
- R 4 is optionally substituted C1-C6 alkyl or optionally substituted phenyl.
- each R 2 and R 3 may be selected, independently, from F, Cl, Br, NO 2 , unsubstituted C1-C6 alkyl, CO 2 H, and CO 2 (unsubstituted C1-C6 alkyl).
- the compounds of the invention also include those shown in Table 3 (above).
- variants of the compounds of Table 3 can be used in the methods described herein.
- a compound includes a phenyl group (e.g., any of Compounds (13)-(27))
- the phenyl group can be modified to be unsubstituted or to be substituted with 1, 2, 3, 4, or 5 substituents selected, independently, from optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy, halogen, CN, NO 2 , CO 2 H, and CO 2 R X , where R X is an optionally substituted C1-C6 alkyl or an optionally substituted phenyl.
- compounds of Table 3 that include a heteroaryl group can be modified in order that the heteroaryl group is unsubstituted or substituted with, e.g., 1 or 2 substituents selected, independently, from optionally substituted C1-C6 alkyl, optionally substituted C1-C6 alkoxy, halogen, CN, NO 2 , CO 2 H, and CO 2 R X , where R X is an optionally substituted C1-C6 alkyl or an optionally substituted phenyl.
- the length of an alkylene linker in a Table 3 compound can also be varied.
- the alkylene linker can be varied such that it includes between 1-6 carbons (e.g., a C1-C6 alkylene, a C1-4 alkylene, a C1-2 alkylene, or even a C1 alkylene).
- the alkylene may be unsubstituted or substituted.
- carboxylic acid e.g., CO 2 H
- phenyl ester e.g., phenyl ester
- Regioisomers or linkage isomers of the compounds of Table 3 can also be used in the compositions and methods described herein.
- the present invention also features uses of the compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3) for treating and/or inhibiting infection by a microbe, such as a fungus, yeast, mold, and bacterium, in a subject (e.g., a human).
- Administration of the compound(s) may be topical, mucosal, e.g., oral, nasal, vaginal, rectal, parenteral, transdermal, subcutaneous, intramuscular, intravenous, via inhalation, ophthalmic, and other convenient routes.
- Pharmaceutical compositions that include the compound(s) can be prepared for a variety of modes of administration and can be administered in a variety of unit dosage forms depending upon the method of administration.
- the compounds of the invention may be administered to treat a variety of microbial infections.
- the compositions and methods of this invention are especially useful for treating or preventing a pathologic condition associated with a microbial infection and/or for decreasing bacterial and/or fungal growth in an animal or a human in need of such treatment.
- compositions and methods of this invention are especially useful for treating mucosa or other tissues of the oral cavity, a wound selected from the group consisting of an ulcer, a laceration, a deep penetrating wound and a surgical wound, and other diseases and disorders caused by microbial infection.
- a wound selected from the group consisting of an ulcer, a laceration, a deep penetrating wound and a surgical wound, and other diseases and disorders caused by microbial infection.
- the patient to be treated may suffer from a fungal, yeast, and/or mold infection.
- Fungal infections can cause conditions including the following: dermatophytoses, dermatophytoma, a tinea infection (e.g., tinea capitis, tinea corporis, tinea pedis, tinea barbae, tinea cruris, tinea manuum, tinea faciale, tinea unguium, and tinea versicolor), onychomycosis, perionychomycosis, pityriasis versicolor, oral thrush, vaginal candidosis, respiratory tract candidosis, biliary candidosis, esophageal candidosis, urinary tract candidosis, systemic candidosis, mucocutaneous candidosis, mycetoma, cryptococcosis, aspergillosis, mucormycosis, chromoblastomycosis, paracoccidioidomycosis, North American blastomycosis, histoplasmosis, cocc
- systemic mycoses diseases caused by fungal infection. These fungal infections are generalized throughout the body. Often, systemic infections are acquired via inhalation of airborne spores and initiated in the lungs. Examples of systemic infections that can be treated using the compositions of the invention include mucocutaneous candidosis, chromoblastomycosis, mycetoma, cryptococcosis, aspergillosis, mucormycosis, paracoccidioidomycosis, North American blastomycosis, histoplasmosis, coccidioidomycosis (San Joaquin or valley fever), and sporotrichosis.
- the patient to be treated is an immunocompromised host, for example, those infected by HIV, those undergoing chemotherapy, transplant recipients, or cancer patients receiving immunosuppressive medications.
- the subject to be treated is a premature or low birthweight infant and the compositions and methods of this invention are useful for reducing the risk of bacterial or fungal infection or sepsis in these patients.
- the methods of the invention include administering a compound(s) of the invention (formulated in a pharmaceutically acceptable carrier) to a person colonized with pathogenic bacteria or fungus.
- the patient may be an immuno-compromised patient affected with leukaemia, lymphoma, carcinoma, sarcoma, allogenic transplant, congenital or acquired immunodeficiency, cystic fibrosis, or AIDS.
- the patient is treated for a biofilm-associated infection that is a nosocomial infection.
- a biofilm-associated infection is a mixed infection, comprising multiple different microorganisms.
- an individual suffering from a biofilm-associated infection is at increased risk of contracting a second infection.
- Biofilms are often associated with cystic fibrosis, endocarditis, osteomyelitis, otitis media, urinary tract infections, oral infections, and dental caries, among other conditions, each of which can be treated by administering a composition of the invention that includes one or more compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as compounds #4, 9, and/or 12).
- a composition of the invention that includes one or more compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as compounds #4, 9, and/or 12).
- compositions and methods of this invention are especially useful for reducing the risk of bacterial infection in a human.
- Pathogenic bacteria include those selected from the group consisting of pneumococcal species, methicillin-resistant Staphylococcus aureus , multi-drug resistant Pseudomonas species, Nesseria sp., Hemophilus sp., Proteus sp., Klebsiella sp., and Escherichia coli .
- the compositions and methods of this invention are also useful for reducing the risk of infection by gram negative bacteria in a person.
- Gram negative bacteria include those selected from the group consisting of Salmonella , e.g. S. Typhimurium, S. Enteritidis, S.
- Pseudomonas e.g.; a bacterium of the Pseudomonas aeruginosa group such as P. aeruginosa group P. aeruginosa, P. alcaligenes, P. anguilliseptica, P. argentinensis, P. borbori, P. citronellolis, P. flavescens, P. mendocina, P. nitroreducens, P. oleovorans, P. pseudoalcaligenes, P. resinovorans , and P. straminea.
- a bacterium of the Pseudomonas aeruginosa group such as P. aeruginosa group P. aeruginosa, P. alcaligenes, P. anguilliseptica, P. argentinensis, P. borbori, P. citronellolis, P. flavescens, P. mendocin
- compositions and methods of this invention are especially useful for reducing the risk of fungal or yeast infection in a human.
- Pathogenic fungi or yeast include those selected from the group consisting of a Trichophyton species, Epidermophyton species, Candida species, Microsporum species, Aspergillus species, Paecilomyces species, Fusarium species, Acremonium species, Chaetomium species, Phoma species, Scopulariopsis species, Scytalidium species, Alternaria species, Epicoccum species, Curvularia species, and any combination thereof.
- the Trichophyton species can be, for example, T. ajelloi, T. concentricum, T. equinum, T. erinacei, T. flavescens, T. gloriae, T. interdigitale, T. megnini, T. mentagrophytes, T. phaseoliforme, T. rubrum, T. schoenleini, T. simii, T. soudanense, T. terrestre, T. tonsurans, T. vanbreuseghemii, T. verrucosum, T. violaceum , or T. yaoundei .
- the Epidermophyton species can be, for example, E. floccosum or E. stockdaleae .
- the Candida species can be, for example, C. albicans, C. parapsiliosis, C. krusei, C. tropicalis, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr, C. guilliermondii , or C. dubliniensis .
- the Microsporum species can be, for example, M. canis, M. gypseum, M. audouini, M. gallinae, M. ferrugineum, M. distortum, M. nanum, M. cookie , or M. vanbreuseghemii .
- the Epicoccum species can be, for example, E. nigrum .
- the Aspergillus species can be, for example, A. sydowii, A. terreus, A. niger, A. terreus, A. fumigatus, A. flavus, A. clavatus, A. glaucus group, A. nidulans, A. oryzae, A. terreus, A. ustus , or A. versicolor .
- the Paecilomyces species can be, for example, P. lilacinus or P. variotii .
- the Fusarium species can be, for example, F. oxysporum, F. solani , or F. semitectum .
- the Chaetomium species can be, for example, C. atrobrunneum, C.
- the Scopulariopsis species can be, for example, S. brevicaulis, S. candida, S. koningii, S. acremonium, S. flava, S. cinerea, S. trigonospora, S. brumptii, S. chartarum, S. fusca , or S. asperula .
- the Scytalidium species can be, for example, S. dimidiatum, S. hyalinum, S. infestans, S. japonicum , or S. lignicola .
- the Alternaria species can be, for example, A. alternate, A. chartarum, A. dianthicola, A.
- the Curvularia species can be, for example, C. brachyspora, C. clavata, C. geniculata, C. lunata, C. pallescens, C. senegalensis , or C. verruculosa.
- the method includes treating a subject, e.g., a human, for candidiasis using a pharmaceutical composition that contains one or more of the compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12).
- a pharmaceutical composition that contains one or more of the compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12).
- Candida is a yeast and the most common cause of opportunistic mycoses worldwide. It is also a frequent colonizer of human skin and mucous membranes.
- Candida albicans is the most pathogenic and most commonly encountered species among all. Its ability to adhere to host tissues, produce secretory aspartyl proteases and phospholipase enzymes, and transform from yeast to hyphal phase are the major determinants of its pathogenicity.
- the methods of the present invention include administration of one or more compounds of the invention to a subject (e.g., a human) in need of treatment for candidiasis (e.g., infection with C. albicans ).
- the subject being treated is diagnosed with a fluconazole-resistant Candida infection.
- Yet another embodiment of the present invention is a process for imparting microbial control properties to a fluid composition, in which the process includes adding an anti-microbial composition of the invention, as defined hereinabove, to the fluid composition.
- Fluid compositions involved in this embodiment of the invention may be, but are not limited to, aqueous compositions susceptible to come into contact with, and subsequently infect, e.g., an animal or a human being.
- the compounds of the invention can be used in anti-microbial compositions to reduce or inhibit biofilm formation on surfaces, e.g., surfaces of medical devices, such as an implantable medical device (e.g., a cardiac-assist device, an artificial heart valve, a catheter, a central line, an IV line, a joint, a stent, a prosthetic implant, a pacemaker, conduit, cannula, appliance, scaffold, an artificial sphincter, a pessary, tube, drain, trochar or plug, implant, a rod, a screw, or orthopedic or implantable prosthetic device or appliance, a suture, a drug delivery device, an oral implant, a denture, a brace, etc.), a cuff, dressing materials, a mesh, a hernia patch, a wound dressing, a bandage, syringes, gloves, and the like.
- an implantable medical device e.g., a cardiac-assist device, an artificial heart valve, a
- the compound or an adduct or salt thereof e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12
- the compound e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12
- the compounds of the invention can also be used in non-medical applications such as in coatings or films in protecting surfaces from bio-fouling.
- Such surfaces include surfaces in contact with water, such as swimming pool liners, water cooling surfaces, hoses, water dispensers, water storage and distribution systems for drinking water or aquaculture, and surfaces in contact with marine environments (including fresh water, brackish water and salt water environments), for example, the hulls of ships, surfaces of docks or the inside of pipes in circulating or pass-through water systems.
- the compounds of the invention may be applied in an amount ranging from 0.1 to 100000 mg per square meter surface.
- Application of the present compounds and/or formulations may follow known methods.
- surfaces may be treated, e.g., by spraying, dipping; bulk materials are commonly equipped with the present compounds in powder form or in form of solutions or dispersions, e.g., by commonly known mixing or kneading processes.
- the compounds of the invention can be used in the anti-microbial (e.g., anti-fungal) treatment of surfaces (e.g., as a disinfectant), and as an anti-microbial agent in, e.g., pharmaceutical compositions (e.g., an eye drop formulation, an inhalant, an adhesive paste, an anti-inflammatory skin care preparation, a wound healing formulation, and an agent against dermatophytes for use in products against seborrheic dermatitis, psoriasis and athletes foot in formulation types such as creams, lotions, gels, powders, oils, tonics, sprays, wet wipes, etc.), cosmetics (e.g., an eye make-up or an eye make-up remover, a sunscreen lotion, an after-sun skin care preparation, a revitalizing skin care preparation, an anti-aging skin care preparation, and an anti-acne composition), hygiene products (e.g., a tooth paste or gel, a mouth wash, a gargle, a hair treatment (e
- compositions of the invention are optionally in the form of an aqueous paste or gel or a liquid, such as an aqueous liquid or viscous liquid.
- Fields of use of the compositions of the invention include, for example, floor coverings, plastics coatings, plastics containers and packaging materials; kitchen and bathroom utensils (e.g., brushes, shower curtains, sponges, bathmats), latex, filter materials (air and water filters), and mattresses.
- the compounds of the invention can also be used to reduce or inhibit microbial growth in industrial formulations, such as coatings, paints, and lubricants, and in paper treatment, especially in paper treatment liquors and printing thickeners of starch or cellulose derivatives.
- the compounds of the invention can also be used to reduce or inhibit microbial growth on agricultural products, such as foods and vegetables.
- the compounds of the invention are capable of penetrating biofilms on living and non-living surfaces, of preventing the adhesion of microbes (e.g., fungal microbes) to surfaces and any further build-up of the biofilm, of detaching such biofilm and/or inhibiting the further growth of the biofilm-forming micro-organisms in the biological matrix, or of killing such micro-organisms.
- microbes e.g., fungal microbes
- the compounds of this invention are also useful in preventing bio-fouling, or eliminating or controlling microbe accumulation on the surfaces either by incorporating the compounds into the article or surface of the article in question or by applying the compounds to these surfaces as part of a coating or film.
- the compounds of the invention may be part of a composition which also includes a binder.
- the binder may be any polymer or oligomer compatible with the compounds of the invention.
- the binder may be in the form of a polymer or oligomer prior to preparation of the anti-microbial composition, or may form by polymerization during or after preparation, including after application to the substrate. In certain applications, such as certain coating applications, it may be desirable to crosslink the oligomer or polymer of the composition after application.
- binder also includes materials such as glycols, oils, waxes and surfactants commercially used in the care of wood, plastic, glass and other surfaces. Examples include water proofing materials for wood, vinyl protectants, protective waxes and the like.
- the compounds of the invention may be incorporated into a composition and applied as a coating or a film to an article or surface.
- the composition is a thermoplastic film, which is applied to a surface, for example, by the use of an adhesive or by melt applications including calendaring and co-extrusion
- the binder may be the thermoplastic polymer matrix used to prepare the film.
- the composition when it is a coating, it may be applied as a liquid solution or suspension, a paste, gel, or oil, or the coating composition may be a solid, for example, a powder coating which is subsequently cured by heat, UV light, or other method known in the art.
- the binder can be comprised of any polymer used in coating formulations or film preparation.
- the binder may be a thermoset, thermoplastic, elastomeric, or inherently crosslinked or crosslinked polymer.
- Thermoset, thermoplastic, elastomeric, and inherently crosslinked or crosslinked polymers include, e.g., polyolefin, polyamide, polyurethane, polyacrylate, polyacrylamide, polycarbonate, polystyrene, polyvinyl acetates, polyvinyl alcohols, polyester, halogenated vinyl polymers, such as PVC, natural and synthetic rubbers, alkyd resins, epoxy resins, unsaturated polyesters, unsaturated polyamides, polyimides, silicon containing polymers, carbamate containing polymers, fluorinated polymers, crosslinkable acrylic resins derived from, e.g., substituted acrylic esters, e.g., from epoxy acrylates, urethane acrylates, or polyester acrylates.
- the polymers may also be blends and copolymers of the preceding chemistries.
- Biocompatible coating polymers such as, e.g., poly[alkoxyalkanoate-co-3-hydroxyalkenoate] (PHAE) polyesters (Geiger et. al., Polymer Bulletin 52, 65-70, 2004), can also serve as binders in the present invention.
- PHAE poly[alkoxyalkanoate-co-3-hydroxyalkenoate]
- Alkyd resins polyesters, polyurethanes, epoxy resins, silicone containing polymers, polyacrylates, polyacrylamides, fluorinated polymers and polymers of vinyl acetate, vinyl alcohol and vinyl amine are non-limiting examples of common coating binders useful in the present invention.
- Other coating binders are part of the present invention.
- Coatings are frequently crosslinked with, for example, melamine resins, urea resins, isocyanates, isocyanurates, polyisocyanates, epoxy resins, anhydrides, poly acids and amines, with or without accelerators.
- compositions of the present invention may, for example, be a coating applied to a surface that is exposed to conditions favorable for bioaccumulation.
- the presence of the compound(s) of the invention in the coating reduces or inhibits the adherence of a microbial organism(s) (e.g., fungi, such as C. albicans ) to the surface.
- a microbial organism(s) e.g., fungi, such as C. albicans
- the compounds of the present invention may be part of a complete coating or paint formulation, such as a marine gel-coat, shellac, varnish, lacquer or paint, or may be incorporated into a composition through the use of one or more polymers or binders, as a carrier substance, such as those described above.
- a complete coating or paint formulation such as a marine gel-coat, shellac, varnish, lacquer or paint
- Other additives that are typically encountered in such coating formulations or applications will find optional use in the present compositions, as well.
- the coating may be prepared as a solvent or aqueous composition.
- Aqueous coatings are typically considered more environmentally friendly.
- the coating may include, for example, an aqueous dispersion that includes a compound of the invention, which may also be bound to or incorporated with a polymer, binder, or other carrier substance, such as those discussed above, or a water based coating or paint.
- the coating may be applied to a surface that has already been coated, such as a protective coating, or applied as a clear coat or a protective wax over a previously uncoated article.
- the coating compositions of the invention may be applied to a surface by any conventional means including, e.g., spin coating, dip coating, spray coating, draw down, or by brush, roller or other applicator. A drying or curing period may be needed. Coating or film thickness may vary depending on application and would be apparent to one skilled in the art.
- compositions containing a compound of the invention may be in the form of a protective laminate film.
- a film typically includes thermoset, thermoplastic, elastomeric, or crosslinked polymers.
- polymers include, but are not limited to, polyolefin, polyamide, polyurethane, polyacrylate, polyacrylamide, polycarbonate, polystyrene, polyvinyl acetates, polyvinyl alcohols, polyester, halogenated vinyl polymers such as PVC, natural and synthetic rubbers, alkyl resins, epoxy resins, unsaturated polyesters, unsaturated polyamides, polyimides, fluorinated polymers, silicon containing and carbamate polymers.
- the polymers may also be blends and copolymers of the preceding chemistries.
- the composition When the composition is a preformed film it may be applied to a surface by, for example, the use of an adhesive, or co-extruded onto the surface. It may also be mechanically affixed via fasteners, which may further utilize a sealant or caulk.
- a plastic film may also be applied with heat, which includes calendaring, melt applications, and shrink wrapping.
- Compositions containing a compound of the invention may be part of a polish, such a furniture polish, or a dispersant or surfactant formulation, such as a glycol or mineral oil dispersion or other formulation as used in for example wood protection.
- a dispersant or surfactant formulation such as a glycol or mineral oil dispersion or other formulation as used in for example wood protection.
- useful surfactants include, but are not limited to, polyoxyethylene-based surface-active substances, including polyoxyethylene sorbitan tetraoleate (PST), polyoxyethylene sorbitol hexaoleate (PSH), polyoxyethylene 6 tridecyl ether, polyoxyethylene 12 tridecyl ether, polyoxyethylene 18 tridecyl ether, TWEENTM surfactants, TRITONTM surfactants, and the polyoxyethylene-polyoxypropylene copolymers such as the PLURONICTM and POLOXAMERTM product series (from BASF).
- PST polyoxyethylene
- matrix-forming components include dextrans, linear PEG molecules (MW 500 to 5,000,000), star-shaped PEG molecules, comb-shaped and dendrimeric, hyperbranched PEG molecules, as well as the analogous linear, star, and dendrimer polyamine polymers, and various carbonated, perfluorinated (e.g., DUPONT ZONYLTM fluorosurfactants) and siliconated (e.g., dimethylsiloxane-ethylene oxide block copolymers) surfactants.
- dextrans linear PEG molecules (MW 500 to 5,000,000)
- star-shaped PEG molecules comb-shaped and dendrimeric, hyperbranched PEG molecules
- comb-shaped and dendrimeric as well as the analogous linear, star, and dendrimer polyamine polymers
- various carbonated, perfluorinated e.g., DUPONT ZONYLTM fluorosurfactants
- siliconated e.g., dimethylsiloxane-ethylene oxide block copoly
- the composition may contain other additives such as antioxidants, UV absorbers, hindered amines, phosphites or phosphonites, benzofuran-2-ones, thiosynergists, polyamide stabilizers, metal stearates, nucleating agents, fillers, reinforcing agents, lubricants, emulsifiers, dyes, pigments, dispersants, other optical brighteners, flame retardants, antistatic agents, blowing agents and the like, such as the materials listed below, or mixtures thereof.
- additives such as antioxidants, UV absorbers, hindered amines, phosphites or phosphonites, benzofuran-2-ones, thiosynergists, polyamide stabilizers, metal stearates, nucleating agents, fillers, reinforcing agents, lubricants, emulsifiers, dyes, pigments, dispersants, other optical brighteners, flame retardants, antistatic agents, blowing agents and the like
- the substrate can be an inorganic or organic substrate, for example, a metal or metal alloy; a thermoplastic, elastomeric, inherently crosslinked or crosslinked polymer as described above; a natural polymer such as wood or rubber; a ceramic material; glass; leather or other textile.
- the substrate may be, for example, non-metal inorganic surfaces such as silica, silicon dioxide, titanium oxides, aluminum oxides, iron oxides, carbon, silicon, various silicates and sol-gels, masonry, and composite materials such as fiberglass and plastic lumber (a blend of polymers and wood shavings, wood flour or other wood particles).
- the inorganic or organic substrate may be, for example, a metal or metal alloy, a thermoplastic, elastomeric, inherently crosslinked or crosslinked polymer, a ceramic material or a glass.
- the substrate may be a multi-layered article comprised of the same or different components in each layer.
- the surface coated or laminated may be the exposed surface of an already applied coating or laminate.
- the inorganic or organic substrate to be coated or laminated can be in any solid form.
- polymer substrates may be plastics in the form of films, injection-molded articles, extruded workpieces, fibres, felts or woven fabrics.
- molded or extruded polymeric articles used in construction or the manufacture of durable goods such as siding, fascia and mailboxes can all benefit from being coated with or prepared using a composition that includes a compound of the invention.
- Plastics which would benefit from being coated with or prepared using a composition that includes a compound of the invention include, but are not limited to, plastics used in medical devices (e.g., in-dwelling medical devices, such as catheters, and other devices, such as those described above), plastics used in construction or the manufacture of durable goods or machine parts, including outdoor furniture, boats, siding, roofing, glazing, protective films, decals, sealants, composites like plastic lumber and fiber reinforced composites, functional films including films used in displays as well as articles constructed from synthetic fibers such as awnings, fabrics such as used in canvas or sails and rubber articles such as outdoor matting and other uses cited in this disclosure.
- medical devices e.g., in-dwelling medical devices, such as catheters, and other devices, such as those described above
- plastics used in construction or the manufacture of durable goods or machine parts including outdoor furniture, boats, siding, roofing, glazing, protective films, decals, sealants, composites like plastic lumber and fiber reinforced composites, functional films including films used in displays as well as articles
- plastics are polypropylene, polyethylene, PVC, POM, polysulfones, styrenics, polyamides, urethanes, polyesters, polycarbonate, acrylics, butadiene, thermoplastic polyolefins, ionomers, unsaturated polyesters and blends of polymer resins including ABS, SAN and PC/ABS.
- the anti-microbial compounds of the invention are also effective in protecting useful plants, such as plants in agriculture, in horticulture and in forests, plant parts and seeds from disease and spoilage.
- the present invention also provides a method of applying to useful plants, the locus thereof or propagation material thereof a composition which includes one or more compounds of the invention.
- the compositions can be used, e.g., as foliar, soil and seed treatment fungicides.
- compositions of the present invention are of particular interest for controlling a large number of fungi in various useful plants or their seeds, especially in field crops, such as potatoes, tobacco and sugarbeets, and wheat, rye, barley, oats, rice, maize, lawns, cotton, soybeans, oil seed rape, pulse crops, sunflower, coffee, sugarcane, fruit and ornamentals in horticulture and viticulture, in vegetables such as cucumbers, beans and cucurbits.
- field crops such as potatoes, tobacco and sugarbeets, and wheat, rye, barley, oats, rice, maize, lawns, cotton, soybeans, oil seed rape, pulse crops, sunflower, coffee, sugarcane, fruit and ornamentals in horticulture and viticulture, in vegetables such as cucumbers, beans and cucurbits.
- the anti-microbial compounds of the invention When applied to plants, the anti-microbial compounds of the invention are applied at a rate of 1 to 5000 g active ingredient per hectare (a.i./ha), for example 2 to 2000 g a.i./ha, for example, 5 to 2000 g a.i./ha, for example, 10 to 1000 g a.i./ha, e.g. 50, 75, 100, 200, 250, 500, 800, 1000, 1500 g a.i./ha.
- the application rates depend on the type of effect desired, and typically range from 20 to 4000 g of total antimicrobials per hectare.
- rates of 0.001 to 50 g of the present anti-microbial compounds, for example 0.01 to 10 g, per kg of seed, are generally sufficient.
- composition comprising the anti-microbial compounds of the invention may be employed in any conventional form, for example in the form a powder for dry seed treatment (DS), an emulsion for seed treatment (ES), a flowable concentrate for seed treatment (FS), a solution for seed treatment (LS), a water dispersible powder for seed treatment (WS), a capsule suspension for seed treatment (CF), a gel for seed treatment (GF), an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (
- compositions may be produced in conventional manner, e.g., by mixing the active ingredients with appropriate formulation inerts (diluents, solvents, fillers and optionally other formulating ingredients such as surfactants, biocides, anti-freeze, stickers, thickeners and compounds that provide adjuvancy effects).
- formulation inerts diols, ethanol, sulfate, sulfate, sulfate, sulfate, sulfate, stickers, thickeners and compounds that provide adjuvancy effects.
- formulations to be applied in spraying forms such as water dispersible concentrates (e.g. EC, SC, DC, OD, SE, EW, EO and the like), wettable powders and granules, typically contain surfactants, such as wetting and dispersing agents and other compounds that provide adjuvancy effects.
- the formulations include from 0.01 to 90% by weight of at least one of the anti-microbial compounds of the invention, from 0 to 20% agriculturally acceptable surfactant and 10 to 99.99% solid or liquid formulation inerts and adjuvant(s), and optionally other active agents, particularly microbiocides or conservatives or the like.
- Concentrated forms of compositions generally contain in between about 2 and 80%, for example, between about 5 and 70% by weight of total active agent.
- Application forms of formulation may for example contain from 0.01 to 20% by weight, for example from 0.01 to 5% by weight of active agent.
- the materials, products, formulations, or compositions described above usually contain the compound(s) of the present invention (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12) in an amount within the range of 0.01 to 50% by weight (e.g., 0.1 to 25% by weight, 0.5 to 15% by weight, or 0.5 to 10% by weight), relative to the overall weight of the material, product, formulation, or composition.
- the compound(s) of the present invention e.g., compounds of formula (I), (I-a), (II), and (II-a
- 0.01 to 50% by weight e.g., 0.1 to 25% by weight, 0.5 to 15% by weight, or 0.5 to 10% by weight
- compositions of the invention which include one or more compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as compound #s 4, 9, and/or 12) can be administered for prophylactic, therapeutic, and/or hygienic use.
- Such administration can be by topical, mucosal, e.g., oral, nasal, vaginal, rectal, parenteral, transdermal, subcutaneous, intramuscular, intravenous, via inhalation, ophthalmic, and other convenient routes.
- the pharmaceutical compositions can be administered in a variety of unit dosage forms depending upon the method of administration.
- unit dosage forms suitable for oral administration include powder, tablets, pills, capsules and lozenges.
- compositions of the invention can also be delivered by means of a microparticle or nanoparticle, a liposome, a nanoemulsion (see, e.g., U.S. Pat. No. 8,226,965), or other delivery vehicle or matrix.
- a number of biocompatible polymeric materials are known in the art to be of use for drug delivery purposes. Examples include polylactide-co-glycolide, polycaprolactone, polyanhydride, and copolymers or blends thereof.
- compositions for administration will commonly include a solution of a compound of the invention (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12) dissolved in or admixed with a pharmaceutically acceptable carrier, preferably an aqueous carrier.
- a pharmaceutically acceptable carrier preferably an aqueous carrier.
- aqueous carriers can be used, e.g., buffered saline and the like.
- These solutions are sterile and generally free of undesirable matter.
- These compositions may be sterilized by conventional, well known sterilization techniques.
- compositions may contain pharmaceutically acceptable auxiliary substances as required to approximate physiological conditions, such as pH adjusting and buffering agents, toxicity adjusting agents and the like, for example, sodium acetate, sodium chloride, potassium chloride, calcium chloride, sodium lactate and the like.
- concentration of anti-microbial compound(s) in these formulations can vary widely, and will be selected primarily based on fluid volumes, viscosities, body weight, and the like in accordance with the particular mode of administration selected and the patient's needs.
- a typical pharmaceutical composition of the invention for intravenous administration would include about 0.0001 to about 100, or about 0.001 to about 10, or about 0.01 to about 10, mg of the compound(s) per patient per day. Dosages from about 0.001 mg, up to about 1000 mg, per patient per day may be used, particularly when administered to a secluded site and not into the blood stream, such as into a body cavity or into a lumen of an organ. Actual methods for preparing parenterally administrable compositions will be known or apparent to those skilled in the art and are described in more detail in such publications as Remington's Pharmaceutical Science, 15th ed., Mack Publishing Company, Easton, Pa. (1980).
- compositions containing the anti-microbial compound(s) of the invention or an adduct or salt thereof can be administered for therapeutic or prophylactic treatments.
- compositions are administered to a patient suffering from a disease, e.g., a fungal infection, in an amount sufficient to cure or at least partially arrest the disease and its complications. An amount adequate to accomplish this is defined as a “therapeutically effective dose.” Amounts effective for this use will depend upon the severity of the disease and the general state of the patient's health.
- compositions may be administered depending on the dosage and frequency as required and tolerated by the patient.
- the composition should provide a sufficient quantity of the anti-microbial compound(s) of the invention to effectively treat the patient.
- the therapeutic composition of the invention can be combined for therapeutic use with additional active ingredients, e.g., in conventional pharmaceutically acceptable carriers or diluents, along with physiologically innocuous stabilizers and excipients; see Berkow (Ed.), The Merck Manual, Merck, Rahway, N.J. These combinations can be filtered sterile and placed into dosage forms as by lyophilization in dosage vials or storage in stabilized aqueous preparations.
- the invention further provides a pharmaceutical composition for treating a human bacterial or fungal infection that includes one or more compounds of the invention in an amount effective to treat a human bacterial or fungal infection and a pharmaceutically acceptable carrier.
- the compositions may also include, e.g., one or more antibiotics.
- the treatment of a bacterial infection includes the addition of an antibiotic(s) for combination or synergistic therapy with a compound of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12).
- the appropriate antibiotic administered will typically depend on the susceptibility of the bacteria such as whether the bacteria is gram negative or gram positive, and will be easily discernable by one of skill in the art.
- antibiotics useful for combination or synergistic therapy with a compound of the invention include aminoglycosides (e.g., amikacin, gentamicin, kanamycin, netilmicin, tobramycin, streptomycin, azithromycin, clarithromycin, erythromycin, erythromycin estolate/ethylsuccinate/gluceptate/lactobionate/stearat), beta-lactams, such as penicillins (e.g., penicillin G, penicillin V, methicillin, nafcillin, oxacillin, cloxacillin, dicloxacillin, ampicillin, amoxicillin, ticarcillin, carbenicillin, mezlocillin, azlocillin, and piperacillin), cephalosporins (e.g., cephalothin, cefazolin, cefaclor, cefamandole, cefoxitin
- aminoglycosides e.g.
- antibiotics include chloramphenicol, clindamycin, trimethoprim, sulfamethoxazole, nitrofurantoin, rifampin and mupirocin. These compositions can be administered to reduce or inhibit biofilm formation or maintenance, e.g., in a human subject.
- the quantities of agents necessary for effective therapy depend upon many different factors, including means of administration, target site, physiological state of the patient, and other medicaments administered. Thus, treatment dosages may be titrated to optimize safety and efficacy. Typically, dosages used in vitro may provide useful guidance in the amounts useful for in situ administration of these reagents. Animal testing of effective doses for treatment of particular disorders will provide further predictive indication of human dosage. Various considerations are described, e.g., in Gilman et al. (Eds.), (1990) Goodman and Gilman's: The Pharmacological Basis of Therapeutics, 8th ed., Pergamon Press, Tarrytown, N.Y., and in Remington's Pharmaceutical Sciences, 17th ed.
- Pharmaceutically acceptable carriers may include water, saline, buffers, and other compounds described, e.g., in The Merck Index, Merck & Co., Rahway, N.J. See also (e.g.) Avis et al. (Eds.), (1993) Pharmaceutical Dosage Forms: Parenteral Medications, Dekker, New York, and Leiberman et al. (Eds.), (1990) Pharmaceutical Dosage Forms Disperse Systems, Dekker, New York. Slow-release formulations or slow-release apparatus may be utilized for continuous administration.
- Therapeutic formulations may be administered in any conventional dosage formulation. Whereas it is possible for the active ingredient to be administered alone, it is preferable to present it as a pharmaceutical formulation.
- Formulations comprise at least one active ingredient (e.g., one or more of the compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as compound #4), together with one or more acceptable carriers therefor.
- Each carrier is preferably both pharmaceutically and physiologically acceptable in the sense of being compatible with the other ingredients and not injurious to the patient.
- Formulations include those suitable for mucosal, e.g., oral, vaginal, topical, rectal, nasal, or parenteral administration (including subcutaneous, intramuscular, intravenous and intradermal administration).
- the formulations may conveniently be presented in unit dosage form and may be prepared by any methods well known in the art of pharmacy: e.g., Gilman et al. (Eds.), (1990) Goodman and Gilman's: The Pharmacological Basis of Therapeutics, 8th ed., Pergamon Press; and Remington's Pharmaceutical Sciences, 17th ed. (1990), Mack Publishing Co., Easton, Pa. Further, the invention may be combined with or used in association with other chemotherapeutic or chemopreventive agents.
- compositions according to the invention that include a compound(s) of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12) may be formulated into topical preparations for local therapy by including a therapeutically effective concentration of the anti-fungal agent(s) in a mucosal or dermatological vehicle.
- the amount to be administered, and the concentration in the topical formulations depend upon the vehicle selected, the clinical condition of the patient, the systemic toxicity and the stability of the formulation.
- the concentration of therapeutic compounds of the invention for mucosal or topical formulations is in the range of greater than from about 0.01 mg/ml to about 2500 mg/ml.
- concentration of the compounds of the invention in the compositions for topical formulations is in the range of greater than from about 1 mg/ml to about 200 mg/ml.
- Solid dispersions of the compositions according to the invention, as well as solubilized preparations may be used.
- the precise concentration to be used in the vehicle is subject to modest experimental manipulation in order to optimize the therapeutic response.
- Suitable vehicles in addition to gels, are oil-in-water or water-in-oil emulsions using mineral oils, petroleum and the like.
- Mucosal or topical preparations of the therapeutic composition either for systemic or local delivery may be employed and may contain excipients as described above for parenteral administration and other excipients used in a topical preparation such as cosolvents, surfactants, oils, humectants, emollients, preservatives, stabilizers and antioxidants.
- Pharmacologically acceptable buffers may be used, e.g., Tris or phosphate buffers.
- the topical formulations may also optionally include one or more agents variously termed enhancers, surfactants, accelerants, adsorption promoters or penetration enhancers, such as an agent for enhancing percutaneous penetration of the therapeutic compounds of the invention or other agents.
- Such agents should desirably possess some or all of the following features as would be known to the ordinarily skilled artisan: pharmacological inertness, non-promotive of body fluid or electrolyte loss, compatible with the therapeutic composition (non-inactivating), and capable of formulation into creams, gels or other topical delivery systems as desired.
- Topical preparations may be applied daily (once, twice, thrice, or as needed) directly to the skin or mucosa and are then preferably occluded, i.e., protected by overlaying a bandage, polyolefin film or other barrier impermeable to the topical preparation.
- composition of the invention may be administered orally.
- a therapeutically effective oral dose of a composition according to the invention is in the range from about 0.05 mg/kg body weight to about 50 mg/kg body weight per day.
- an effective dose is in the range from about 0.05 mg/kg body weight to about 5 mg/kg body weight per day.
- the pharmaceutical composition is administered vaginally.
- the therapeutic agents may be formulated as is known in the art for direct application to the vaginal area.
- Forms chiefly conditioned for vaginal application take the form, for example, of creams, milks, gels, dispersion or micro-emulsions, lotions thickened to a greater or lesser extent, impregnated pads, ointments, aerosol formulations (e.g., sprays or foams), creams, lotions, pastes, jellies, sprays, and aerosols.
- the composition can be formulated to be part of an adhesive polymer, such as polyacrylate or acrylate/vinyl acetate copolymer.
- the dose will be dependent upon the properties of the specific composition employed, e.g., its activity and biological half-life, the concentration of composition in the formulation, the site and rate of dosage, the clinical tolerance of the patient involved, the disease afflicting the patient and the like, as is well within the skill of the physician.
- compositions of the invention may be administered to the lung(s) of a subject by any suitable means, e.g., be inhaled into a patient's respiratory tract and lungs through the nose or mouth.
- the composition of the invention can be aerosolized in a variety of forms, such as, but not limited to, dry powder inhalants, metered dose inhalants, or liquid/liquid suspensions.
- carriers such as polyethylene glycol or glycols, DPPC, methylcellulose, powdered dispersing agents can be used.
- a composition of the invention is administered by inhalation, for example, in a nebulized form.
- delivery may be by use of a single-use delivery device, a mist nebulizer, a breath-activated powder inhaler, an aerosol metered-dose inhaler (MDI) or any other of the numerous nebulizer delivery devices available in the art.
- MDI aerosol metered-dose inhaler
- mist tents or direct administration through endrotracheal tubes may also be used.
- delivery via an intratracheal or nasopharyngeal mode will be efficacious for certain indications.
- the dose will be dependent upon the properties of the specific composition employed, e.g., its activity and biological half-life, the concentration of composition in the formulation, the site and rate of dosage, the clinical tolerance of the patient involved, the disease afflicting the patient and the like, as is well within the skill of the physician.
- compositions of the present invention may be administered in solution.
- the compositions thereof may be in a solution having a suitable pharmaceutically acceptable buffer such as phosphate, Tris(hydroxymethyl)aminomethane-HCl or citrate and the like. Buffer concentrations should be in the range of 1 to 100 mM.
- the composition solution may also contain a salt, such as sodium chloride or potassium chloride in a concentration of 50 to 150 mM.
- An effective amount of a stabilizing agent, such as an albumin, a globulin, a gelatin, a protamine or a salt of protamine may also be included, and may be added to a solution containing composition or to the composition from which the solution is prepared.
- compositions of the invention can be administered in the form of an article or carrier such as a bandage, insert, syringe-like applicator, tablet, suppository, pessary, powder/talc or other solid, solution, liquid, spray, aerosol, douche, ointment, tampon, foam, cream, gel, paste, microcapsules, vaginal sponge, vaginal ring, controlled release formulation, sustained release formulation or bioadhesive gel (e.g., a mucoadhesive thermo-gelling composition (see, for example, U.S. 2003/0204180, which is incorporated herein by reference)).
- a bandage such as a bandage, insert, syringe-like applicator, tablet, suppository, pessary, powder/talc or other solid, solution, liquid, spray, aerosol, douche, ointment, tampon, foam, cream, gel, paste, microcapsules, vaginal sponge, vaginal ring
- unit dosage and its grammatical equivalents as used herein refer to physically discrete units suitable as unitary dosages for human patients and other warm blooded animals, each unit containing a predetermined effective and potentiating amount of at least one compound of the invention calculated to produce the desired therapeutic effect in association with the required physiologically tolerable carrier, e.g., a diluent or a vehicle.
- physiologically tolerable carrier e.g., a diluent or a vehicle.
- the specifications for the unit dosage forms of this invention are dictated by and are directly dependent on (a) the unique characteristics of the active ingredient(s) and the particular therapeutic effect to be achieved, and (b) the limitations inherent in the art of compounding for therapeutic use in humans and other animals.
- suitable unit dosage forms in accord with this invention are tablets, capsules, pills, powder packets, granules, wafers, and the like, segregated multiples of any of the foregoing, as well as liquid solutions, emulsions and suspensions.
- the amount of each active ingredient that is administered in vivo depends on the age and weight of the patient, the particular disease to be treated and its severity, the frequency of administration, and the route of administration.
- the therapeutic composition may be administered to a patient either singly or in a cocktail containing other therapeutic agents, compositions, or the like, including, but not limited to, immunosuppressive agents, tolerance-inducing agents, potentiators and side-effect relieving agents.
- immunosuppressive agents useful in suppressing allergic reactions of a host.
- Preferred immunosuppressive agents include prednisone, melphalain, prednisolone, DECADRON (Merck, Sharp & Dohme, West Point, Pa.), cyclophosphamide, cyclosporine, 6-mercaptopurine, methotrexate, azathioprine and i.v. gamma globulin or their combination.
- Preferred potentiators include monensin, ammonium chloride, perhexiline, verapamil, amantadine and chloroquine. All of these agents are administered in generally accepted efficacious dose ranges such as those disclosed in the Physician's Desk Reference, 41st Ed., Publisher Edward R. Barnhart, N.J. (1987).
- compositions of the invention that include one or more compounds of the invention or an adduct or salt thereof (e.g., compounds of formula (I), (I-a), (II), and (II-a), as well as those compounds shown in Tables 1, 2, and 3, such as, e.g., compound #s 4, 9, and/or 12) may also be formulated in controlled or sustained release compositions.
- the compounds of the invention may be encapsulated within a biodegradable-biocompatible polymeric matrix.
- the biodegradable-biocompatible polymeric matrix can be used to provide sustained release of a compound(s) of the invention for a period of 10 to up to 100 days in an aqueous physiological environment.
- Controlled drug delivery from a biodegradable-biocompatible matrix offers profound advantages over conventional drug/antigen dosing.
- Drugs/antigens can be used more effectively and efficiently, less drug/antigen is required for optimal therapeutic effect and, in the case of drugs, toxic side effects can be significantly, reduced or essentially eliminated through drug targeting.
- the stability of some drugs/antigens can be improved allowing for a longer shelf-life, and drugs/antigens with a short half-life can be protected within the matrix from destruction, thereby ensuring sustained release of active agent over time.
- the benefit of a continuous sustained release of drug/antigen is beneficial because drug levels can be maintained within a constant therapeutic range and antigen can be presented either continuously or in a pulsatile mode as required to stimulate the optimal immune response. All of this can be accomplished with a single dose of encapsulated drug/antigen.
- Sustained release compositions include those described in, e.g., U.S. Pat. No. 6,309,669.
- each plate contained a column of wells containing SC5314 cells exposed only to DMSO vehicle ( FIG. 1A , last column).
- each plate also included a column of wells containing C. albicans edt1 ⁇ / ⁇ mutant cells (Wheeler, PloS Pathog. 4: e1000227, 2008) that lack a cell wall protein important for adhesion ( FIG.
- alamarBlue measures the metabolic activity of live cells
- Candidate Compound Inhibits C. albicans Adhesion to Human Cells, Affects Multiple Pathogenic Candida Species, and is not Toxic to Human Cells
- compound #4 does not affect the viability of this human cell line, even at concentrations much larger than those used in the adhesion assay (e.g. 250 ⁇ M, FIG. 2C ).
- concentrations much larger than those used in the adhesion assay e.g. 250 ⁇ M, FIG. 2C .
- compound #4 is not toxic to this human cell line under our assay concentrations, but can impair fungal adhesion both to inert surfaces and to cultured human epithelial cells.
- Compound #4 is Functionally Distinct from Previously Described Compounds that Affect Candida Drug Resistance
- FIG. 1B We initially tested each of the compounds that inhibit adhesion to polystyrene ( FIG. 1B ), as well as compounds #9, Q1 and Q2 ( FIG. 3A ), at concentration of 12.5 ⁇ M.
- compounds #4, 9, 12, Q1, and Q2 most potently inhibited hyphal formation and induction of RFP ( FIG. 4A ).
- Compounds Q1 and Q2 share a “Scaffold 1” backbone with compounds #4 and #9, but contain a quinoline group ( FIG. 3A ). Therefore, the compounds with a quinoline group can inhibit hyphae formation, but not adhesion to polystyrene or human cells ( FIG. 3 ).
- albicans cells efficiently formed biofilms on the silicone mesh; in contrast, edt1 ⁇ / ⁇ mutant cells did not, resulting in the majority of the cells dispersed throughout the media rather than adhered to the mesh ( FIG. 5 ).
- Visual inspection suggested that compounds #1, 4, 6, 7, 9, 12, 21, Q1, and Q2 most effectively kept the cells dispersed in the media rather than on the mesh ( FIG. 5A ).
- Measurements of the media density FIG. 5B ) generally confirmed these assessments, although the density observed for compound #21 was more modest than for the other compounds tested, perhaps reflecting effects on cell growth.
- the dry weight measurements FIG. 5C ) indicated that compounds #4 and 9 were most effective at reducing biofilm formation, closely followed by compounds Q1 and Q2. Therefore, compounds #4 and Q1 were effective inhibitors of both hyphal morphogenesis ( FIG. 4 ) and biofilm formation on silicone mesh ( FIG. 5 ).
- compound #4 displayed the widest range of activities that might impair fungal pathogenesis ( FIGS. 1, 2, 4, 5 ), and was non-toxic to mammalian cells ( FIG. 2C ). We therefore tested whether compound #4 can alter fungal infections in two in vivo settings.
- pathogenesis assays in which nematodes ( Caenorhabditis elegans ) are infected with C. albicans , resulting in killing of the nematode host (Jain, 2009; Okoli, 2009).
- VVC vulvovaginal candidiasis
- CFU colony forming units
- albicans biofilm during ex vivo VVC as revealed by scanning electron microscopy (SEM; FIG. 6B ), with a predominance of individual, yeast-form cells appearing. Therefore, although compound #4 only partially inhibits hyphae formation or ECM production in this system, ultrastructural aspects of the biofilms are altered.
- FIGS. 3-5 We find that small changes in the compounds can lead to different combinations of activities ( FIGS. 3-5 ). This was most pronounced in the comparison of compounds #4 and 9, which differ by the substitution of a methyl group for a chlorine substitution on one aryl ring ( FIG. 3 ; Table 1). Compound #4 but not #9 effectively inhibited human cell binding, but these two compounds shared the ability to inhibit polystyrene binding ( FIG. 3 ), hyphae formation ( FIG. 4 ), and biofilm formation ( FIG. 5 ). As another example, the quinoline ring-containing compounds Q1 and Q2 do not inhibit polystyrene binding or human cell binding, but do inhibit hyphae formation or biofilm formation.
- the C. albicans strains used were the wild-type clinical isolate SC5314 (Gillum, 1984), SC5314-derived edt1 ⁇ / ⁇ cells that lack an adhesion protein (see Table 2 of Wheeler, 2008), or their respective GFP-expressing derivatives (Wheeler, supra).
- the SGH284 strain was used as a hyphal reporter strain; this contains the red fluorescent protein (RFP) gene under the control of the endogenous, hyphal-specific HWP1 promoter (Ganguly, 2011).
- Other fungi analyzed included C. tropicalis, C. dubliniensis (Moran, 1997), and fluconazole-resistant clinical isolates of C. albicans from AIDS patients (Bachmann, 2002). Saccharomyces cerevsiae W303 (Thomas, Rothstein) was used to measure the effects of compounds on liquid growth.
- the C. elegans wild type strain N2 was grown at 20° C. on nematode growth agar medium (NGM) spotted with Escherichia coli OP50 and maintained as described previously (Brenner, 1974). E. coli OP50 was grown overnight in Luria broth at 37° C.
- the plates were covered with foil and incubated at 37° C. for 4 hours. The contents of the wells were then decanted and 50 ⁇ l of 0.5% crystal violet (Sigma) in water were added to each well. The plates were covered again and incubated at room temperature for 45 minutes. The dye was removed by decanting, and the plates were gently rinsed by ten rounds of submersion in an ice bucket filled with distilled water, followed by decanting the water. The water in the bucket was changed after the fifth wash. The plates were then gently inverted onto a paper towel to remove excess water. 200 ⁇ l of 75% methanol were then added to each well. The plates were incubated for 30 minutes at room temperature and then absorbance at 590 nm was measured.
- 0.5% crystal violet Sigma
- SC5314-GFP and edt1 ⁇ / ⁇ -GFP strains were pregrown overnight in SC+0.2% glucose media at 30° C., diluted into fresh SC+0.2% glucose media to a final concentration of 5 ⁇ 10 6 cells/ml (equivalent to 0.5 OD 600 /ml) and plated onto Immulon 2HB 96-well plates, with each well receiving 0.1 OD of cells.
- DMSO or compounds resuspended in DMSO were added to the desired final concentration (1% vol/vol of DMSO was maintained). Plates were then incubated for 4 hours at 37° C.
- A549 cells in RPMI+10% FBS were seeded into 24-well plates at 1 ⁇ 10 5 cells/well and grown overnight at 37° C. Media was removed and replaced with fresh media containing indicated concentrations of compound #4 or 1% sodium azide, followed by incubation for 24 hrs. Viability was determined with 5% alamarBlue as above.
- worms in the L3/L4 stage were transferred to each of two NGM agar plates containing E. coli OP50 and grown at 20° C. for four days. On the day of the experiment, worms were washed off the plates with M9 buffer and centrifuged at 900 ⁇ g for 2 min. The supernatant was removed, and the worms were then resuspended in a bleach solution (1:4 dilution of commercial bleach (5.25%) diluted in 0.25 M sodium hydroxide). The worm suspension was mixed gently by inversion for 3 min, and centrifuged for 2 min at 2,000 ⁇ g.
- the pellet containing the eggs was washed with M9 buffer and centrifuged at 2,000 ⁇ g for 2 min and then resuspended in 500 ⁇ l M9 buffer.
- the egg suspension was diluted or concentrated with M9 buffer as required to obtain approximately 30-40 eggs/5 ⁇ l.
- E. coli and Candida strains were grown overnight at 37° C. Culture aliquots were centrifuged at full speed for 1 minute in a table top microcentrifuge and the supernatant was removed. Pellets were washed twice in sterile deionized water, and finally resuspended to a final concentration of 200 mg/ml and 10 mg/ml, respectively. A mixture of 10 ⁇ l of a 50-mg/ml streptomycin sulfate stock, 7 ⁇ l of distilled water, 2.5 ⁇ l E. coli and 0.5 ⁇ l of Candida was spotted on to each NGM plate. Drugs in a final concentration of 12.5 ⁇ M were added to the mix. E. coli spotted plates were used as a control.
- Compound #4 and a related compound termed “compound #9” (Table 1), is also able to coat plastic surfaces, making the surfaces more resistant to subsequent fungal colonization. Furthermore, compound #4 can pretreat Candida cells to make them less adhesive. These data show that compound #4 and related compounds can be used as therapeutics to treat candidiasis and in antifungal coatings for implanted medical devices.
- adhesion is required for formation of aggressive biofilms, which in turn make Candida a successful pathogen (Finkel, supra). Therefore, adhesion is a pivotal step in fungal pathogenesis, but one that has not to our knowledge been targeted in previous small molecule screens.
- detection of compounds that prevent adhesion of Candida albicans to polystyrene surfaces can be done in a high throughput manner.
- our screen (described below) requires removal of unbound cells. Although this is more complex than previous screens that only measure metabolism, this extra effort allowed us to detect a class of compounds that have unique properties.
- the assays in this proposal are based on altering the behavior of intact cells, we avoid the complication of compounds unable to cross the cell wall and membranes.
- the following assay provides, as a primary compound screen, a high throughput assay to identify chemicals that inhibit fungal adhesion, a necessary first step to infection. Furthermore, efficient adhesion is required for formation of aggressive biofilms (Blankenship et al., Curr. Opin. Microbiol. 9:588-594, 2006), which in turn make Candida a successful pathogen (d'Enfert, Curr. Opin. Microbiol. 12:358-364, 2009). Therefore, chemicals that attenuate adhesion have the potential of being good antifungal agents.
- the assays described herein is based on altering the behavior of intact cells and avoid the complication of compounds unable to cross the cell wall and membranes.
- the multi-well plate reader and a robotic liquid handling system used for these studies was provided by The Small Molecule Screening Facility at UMMS.
- This Facility has a chemical library (DIVERSet) with 30,000 compounds that was purchased from ChemBridge Corporation. This is a diverse collection of drug-like small molecules from a company that supports prompt “hit” re-supply with 99.8% availability. The set is rationally selected to cover the broadest part of biologically relevant pharmacophore diversity space. It is available in a 96-well pre-plated DMSO format.
- the compounds in this library are at 5 mM concentration in DMSO, and the minimal accurate pipetting volume of the robot in the facility is 1 ⁇ l. Therefore, in 200 ⁇ l cultures pipetted into 96-well plates, addition of 2 ⁇ l from the library results in an initial compound concentration of 50 ⁇ M, and a final DMSO concentration of 1%. Importantly, we have established that this concentration of DMSO is tolerated by Candida ( FIG. 12 ).
- Adhesion Assay Crystal Violet Detection.
- Yeast cells grown in low glucose culture are known to efficiently bind to polystyrene surfaces via adhesive cell surface proteins that are required for mammalian host cell infection (Reynolds and Fink, Science 291:878-881, 2001; and Cormack et al., Science 285:578-582, 1999).
- the protocol for our adhesion assay has been expanded to high throughput applications using liquid handling robots from known assays (Reynolds, supra; and US 2002/0160444).
- the reagents are safe, inexpensive and readily available. We have titered the following volumes, concentrations and incubation times to get the most reliably high Z factor:
- Each plate has control wells ( FIG. 13 ).
- the 96-well plates that house the library of compounds at UMMS are formatted such that the first column and last column contain only DMSO. We used the last column of each plate as a set of positive controls (DMSO only, so 100% adhesion signal will be defined) and first column as negative controls (non-adhesive mutant Candida cells added). Each 96-well library plate contains therefore 80 compounds, and a total of 375 plates carry 30,000 compounds.
- HATs histone acetyl-transferases
- Ac-CoA acetyl-coenzyme A
- histone acetylations can promote appropriate gene expression (Brownell, 1996), chromatin conformation (Shogren-Knaak, 2006), DNA replication (Unnikrishnan, 2010) and DNA repair (Lukas, 2011).
- H3K56 histone H3 lysine 56
- H3K56ac histone H3 lysine 56
- H3K56ac has been detected in metazoan organisms, it is far less abundant (Yu, 2012; Drogaris, 2012), and therefore does not appear to serve as a quantitative marking system for nascent histones. Indeed, the more important modification of H3K56 in metazoans appears to be methylation, not acetylation (Yu, 2012).
- H3K56ac is catalyzed solely by the histone acetyltransferase Rtt109 (Schneider, 2006; Han, 2007; Driscoll, 2007; Collins, 2007; Tsubota, 2007; Xhemalce, 2007; Lopes da Rosa, 2010).
- H3K56 acetylation is either entirely absent or barely detectable in mammalian cells.
- Rtt109 has very limited primary sequence homology to the three well-characterized HAT families: p300/CBP, GNAT (Gcn5-related N-acetyltransferase) and MYST (MOZ, Ybf2/Sas3, Sas2, Tip60). Rtt109 shares the greatest similarity to the mammalian acetyltransferase p300 (Bazan, 2008; Wang, 2008), which is important for N-terminal histone tail acetylations related to transcriptional activation (Chen, 2011).
- both Rtt109 and p300 are regulated by stimulatory autoacetylation events (Thompson, 2004; Collins, 2007; Lin, 2008; Stavropoulos, 2008; Albaugh, 2011), and Rtt109 has a similar tertiary fold structure as p300 (Tang, 2008; Stavropoulos, 2008; Lin, 2008).
- the reaction mechanisms of Rtt109 and p300 differ.
- Rtt109 use a sequential catalytic mechanism (Albaugh, 2010), as observed for other HATs that require formation of a ternary intermediate complex with both histone and AcCoA substrates before catalytic steps can occur (Tanner, 1999; Berndsen, 2007).
- p300 operates a Theorell-Chance (“hit-and-run”) mechanism that involves association of the enzyme with acetyl-coA first, followed by transient association with the protein substrate (Liu, 2008).
- HAT inhibitors including those specific for p300 such as the bisubstrate mimic Lys-CoA, do not affect Rtt109 catalysis (Tang, 2008; Bowers, 2010), and the active sites of these two enzymes display dramatically different electrostatic characteristics (Wang, 2008). Therefore, Rtt109 displays significant differences from its closest mammalian homolog, p300, in terms of both its structure and its biological function.
- Rtt109 by itself is a poor enzyme, but can be activated by either of two different histone chaperone proteins, Asf1 and Vps75 (Tsubota, 2007). These cofactors stimulate modification of distinct substrate lysines. For example, Asf1 is required for acetylation of H3K56 by Rtt109 in vivo (Recht, 2006; Schneider, 2006).
- Vps75 stimulates Rtt109 to acetylate H3K9, H3K23 and H3K27 but is not required for H3K56 acetylation in vivo (Berndsen, 2008; Fillingham, 2008; Burgess, 2010), despite its ability to stimulate H3K56 acetylation in vitro (Tsubota, 2007).
- Rtt109 Small molecules that target the fungal HAT enzyme, Rtt109, but not other HAT enzymes, such as mammalian (e.g., human) HAT enzymes, can be identified using Rtt109 in a screening assay, which is described below.
- Rtt109 is a promising novel target for small molecule therapeutic intervention with minimal toxicity to mammalian hosts.
- Rtt109 enzymatic activity can be measured in a sensitive microplate assay of defined composition ( FIGS. 14A-14F ).
- a direct inhibitor screen can be conducted using this format.
- rtt109-1 mutant C. albicans lack H3K56 acetylation and are highly sensitive to genotoxic agents, including methyl methanesulfonate (MMS), camptothecin, and the reactive oxygen species (ROS) hydrogen peroxide ( FIG. 15 ).
- ROS-induced stress is part of an antifungal defense mechanism used by phagocytic cells of the immune system.
- rtt109 ⁇ / ⁇ mutant cells are less pathogenic in a mouse model of systemic candidaisis induced by tail vein injection ( FIG. 16 ). Together, these data demonstrate that Rtt109 is an important novel target for antifungal therapy.
- Reagents for the enzymatic reactions are assembled in a cocktail containing buffer optimal for the reaction (final concentration: 50 mM Tris-Cl, pH 8.0, 1 mM DTT, and 0.1 mg/ml bovine serum albumin (BSA)).
- BSA bovine serum albumin
- Recombinant Rtt109/Vps75 complex, recombinant histones H3/H4 and Ac-CoA are titrated into this mix. Reactions are incubated at 30° C. for 30 minutes, stopped by placing on ice, and distributed to protein-binding 96-well plates for overnight incubation at 4° C. At this point, the protocol is based on standard ELISA protocols.
- H3-K56 acetylation by Rtt109 is robustly and sensitively detected in this assay in a DMSO-insensitive manner. Therefore, we adapted our protocol for high-throughput screens (HTS).
- candidate compounds should also inhibit histone acetylation by Rtt109 stimulated by Asf1 instead of Vps75.
- compounds should not inhibit the distantly related human p300 enzyme.
- HTS high throughput screen
- Compound KB7 has the chemical formula C 22 H 23 ClFN 3 O 3 (IUPAC name N-[(2-chloro-6-fluorophenyl)methyl]-2-(2,5-dioxo-4-phenyl-4-propylimidazolidin-1-yl)-N-methylacetamide), and is listed as PubChem Compound CID 4785700, Substance SID 49676148. Notably, current listings for PubChem Bioactivity assays show it has been tested in 368 assays, but was active in only two.
- Histone tetramer substrates rather than H3 N-terminal peptides were required for these experiments because Asf1 stimulates H3K56 acetylation but not N-terminal acetylation (Berndsen, 2008; Fillingham, 2008).
- Asf1 stimulates H3K56 acetylation but not N-terminal acetylation
- Compound KB7 inhibited acetylation by both Rtt109-Vps75 and Rtt109/Asf1 comparably ( FIG. 25A ).
- these data also confirm that acetylation of H3K56 is inhibited by Compound KB7, which had not been examined in the ThioGlo1-based assays that used the N-terminal histone H3n21 peptide substrate.
- Compound KB7 inhibits Rtt109 with an apparent IC50 of ⁇ 56 nM and but does not significantly inhibit the other HATs tested, p300 and Gcn5, even at concentrations up to 100 uM, suggesting that there is at least two orders of magnitude of specificity for Rtt109.
- KB7 inhibits H3K56 acetylation by Rtt109 activated by either Vps75 or Asf1.
- H3K56ac is the physiologically relevant PTM for genome stability and efficient pathogenicity of yeast in the face of DNA damage.
- KB7 is efficient at nanomolar range, equivalent to Rtt109 concentration, when the substrates are at micromolar range. It is unlikely that sequestration of substrates is occurring.
- KB7 decreases the Vmax with respect to both Ac-CoA and H3n21 substrates, indicating that the substrates do not compete with the inhibitor to prevent inhibition ( FIGS. 3.3 and 3.4 ).
- the inhibitor is therefore noncompetitive with respect to both substrates and most likely binds allosterically on Rtt109 to prevent catalysis without affecting either substrate (or chaperone) interaction.
- KB7 exhibits properties consistent with its use as an antifungal agent.
- Recombinant S. cerevisiae 6 ⁇ His-Rtt109, 6 ⁇ His-Vps75 and co-expressed Rtt109-Vps75 complexes with a 6 ⁇ His tag on either protein were purified as previously described (Tsubota, 2007).
- Recombinant S. cerevisiae FLAG-epitope-tagged Asf1N(N-terminal amino acids 1-155) was purified as previously described (Daganzo, 2003).
- Recombinant Xenopus laevis histone H3 and histone (H3-H4) 2 tetramers were purified as previously described (Luger, 1999).
- Recombinant 6 ⁇ His-FLAG-p300 catalytic domain (p300 amino acids 1195-1810; Fan, 2010) was expressed in BL21 E. coli cells and purified on Ni-NTA resin (Qiagen) using the manufacturer's instructions. The peak elutions were pooled and dialyzed in 20 mM Hepes, 7.5, 25 mM NaCl, 1 mM EDTA, 5% glycerol and 1 mM PMSF (phenylmethylsulfonyl fluoride) and stored in small aliquots at ⁇ 80° C.
- PMSF phenylmethylsulfonyl fluoride
- Gcn5 was precipitated with 70% NH 4 SO 4 by slowly adding finely ground NH 4 SO 4 crystals at 4° C. while stirring. The solution was centrifuged at 20,000 ⁇ g and the pellet was resuspended in 20 mM Hepes pH 7.5, 1 mM EDTA, 10% glycerol, 0.01% NP40. Gcn5 was dialyzed into 20 mM Hepes, 7.5, 1 mM EDTA, 5% glycerol (no salt), ultracentrifuged at 100,000 ⁇ g for 45 minutes and stored in small aliquots at ⁇ 80° C.
- the final composition of the HAT reaction was 100 nM Rtt09-Vps75, 60 ⁇ M H3n21 peptide and 75 ⁇ M Ac-CoA in 50 mM Hepes 0.0005% Pluronic F-68, pH 8.0.
- the reaction proceeded for 2 hours at RT°.
- Released coenzyme A was detected by the addition of 10 ⁇ l 4 mM Ellman's Reagent (DTNB) in PBS, 0.0005% Pluronic pH 7.4, for a final volume of 60 ⁇ l at 0.66 ⁇ M.
- DTNB Ellman's Reagent
- plates were read at absorbance 405 nm on an Envision (Perkin Elmer) plate reader.
- Single-point enzyme assays analyzed by ELISA were performed with 50 nM Rtt109 and 50 nM Vps75 or 400 nM Asf1-N terminus (amino acids 1-155) or 15 ⁇ g/mL p300. 300 nM (H3-H4) 2 tetramer substrate was provided. Reactions were initiated with 30 ⁇ M Ac-CoA (Sigma Aldrich, catalogue #A2056). Briefly, the protein mix was assembled on ice in 50 mM Tris, pH 8.0, 0.1 mg/ml BSA, 1 mM DTT to a volume of 323.4 ⁇ l.
- DMSO Dimethylsulfonate
- the reactions were stopped by placing on ice, and 100 ⁇ L were plated in triplicate on Immulon B 96-well ELISA plates. The plates were stored overnight at 4° C. to allow proteins to bind to the plastic.
- Reactions were decanted and non-specific binding by the antibodies was prevented by incubating the wells with 200 ⁇ L 1% BSA, 0.05% Tween-20, TBS for 1-2 hrs at 4° C.
- ELISA detection was performed with 100 ⁇ L per well of rabbit anti-serum raised against an H3K56ac peptide (21 st Century Biochemicals, Marlboro, Mass.) diluted at 1:5000 in ELISA buffer (0.05% tween-20, TBS) for 1-2 hrs at 4° C.
- Anti-rabbit HRP-conjugated IgG was used at 1:2500 in ELISA buffer for 100 ⁇ L per well for 1 hr at 4° C. Washes before and after antibody incubations were performed 3 ⁇ 200 ⁇ L per well with ELISA buffer.
- DMSO Dimethylsulfonate
- DMSO Dimethylsulfonate
- the amount of coenzyme A released upon acetylation was quantified at each collected time point as previously described (Trievel, 2000), and results were analyzed using Prism GraphPad software. Briefly, released coenzyme A is detected by a maleimide reagent (ThioGlo1; EMD, catalogue #595501) which fluoresces upon binding to the free sulfhydryl exposed on Coenzyme A. Rates were determined by comparison with standard curves of fluorescence obtained with known amounts of Coenzyme A (CoA; Sigma Aldrich, catalogue #C3144). Two-fold serial dilutions from 6-0.5 ⁇ M CoA were prepared in HAT reaction buffer for each plate, and an equal volume of isopropanol was added.
- reaction rates versus substrate concentration were fitted to a non-linear regression curve and analyzed using the Michaelis-Menten equation by Prism GraphPad.
- Three concentrations of inhibitor were tested per experiment for each substrate, including a 1% DMSO control (no inhibitor). Eight concentrations of substrate were tested per concentration of inhibitor. Due to day-to-day variation in reaction rates, a single representative experiment is shown for each substrate titration. The trends and conclusions between the individual experiments are consistent.
- Compound #4 Inhibits Hyphal Formation in Aspergillus
- Compound #4 was also tested for its inhibitory effect on the growth of Aspergillus fumigatus .
- Fresh Aspergillus fumigatus conidia (spores) were grown in RPMI tissue culture media overnight in the presence of 1% DMSO (control), 50 ⁇ M compound #4, 100 ⁇ M compound #4, or no treatment (control) using methods similar to those described in Ramirez-Ortiz et al. ( Cell Host & Microbe 9:415-424, 2011).
- the conidia display the normal amount of hyphal elongation protruding from the cells in the untreated ( FIG. 26A ) and DMSO-treated ( FIG. 26B ) controls. This is the first step in germination of the spores, and is required for pathogenesis.
- compound #4 blocks hyphal formation at both concentrations tested.
- Compound #4 was also tested for its inhibitory effect on the growth of Cryptococcus gatti.
- Cryptococcus cells were incubated at 37° C. in unsupplemented RPMI tissue culture media (+Glutamine ⁇ Phenol Red (Gibco #11835, Life Technologies, Grand Island, N.Y.; “RPMI”) or RPMI tissue culture media supplemented with 100 U/ml Penicillin/100 ug/ml Streptomycin (Gibco 100 ⁇ Pen/Strep #151500-122), 1 mM NaPyruvate, and 10 mM HEPES pH 7.0 (“Af medium”) in the presence of 1% DMSO (control), 50 ⁇ M compound #4, or 100 ⁇ M compound #4, and cell numbers were counted after 18 hours.
- the assay was also performed with an untreated control.
- compound #4 inhibited proliferation of Cryptococcus gatti at both concentrations tested. No difference in the results was observed using supplemented or unsupplemented RPMI.
- Compound #4 Inhibits C. albicans Biofilm Formation and Inhibits Hyphal Morphogenesis and Normal Colony Morphology
- Hyphal morphogenesis is induced by multiple stimuli that act via multiple signal transduction pathways and is mediated by a complex transcriptional network.
- mammalian serum also induces hyphae, and we observed that compound #4 inhibited the response to both these stimuli ( FIGS. 29A and 29B ).
- FIGS. 29A and 29B We detected morphological difference between control and compound #4-treated cultures as early as 60 minutes in 10% serum, and 90 minutes in Spider media.
- a wrinkled colony morphology is displayed by C. albicans when grown on solid Spider media, reflecting transitions between hyphal and yeast forms. Notably, this wrinkled phenotype was also abolished by compound #4 ( FIGS. 29A and 29B ).
- FIGS. 29A and 29B our data indicate that compound #4 inhibits hyphal morphogenesis and a colony morphology phenotype that depends on the hyphal transition.
- cAMP-PKA cyclic AMP-protein kinase A pathway
- FIG. 30A Stimulation of this pathway drives PKA to phosphorylate transcription factor Efg1, activating Efg1 to increase expression of genes required for hyphal morphogenesis.
- compound #4 affects more than one signaling pathway.
- the modified sugar N-acetyl-glucosamine also stimulates hyphal morphogenesis, but does so independently of the cAMP-PKA pathway, instead activating the transcription factor Cph1.
- GlcNac modified sugar N-acetyl-glucosamine
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Environmental Sciences (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Dentistry (AREA)
- Plant Pathology (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Pain & Pain Management (AREA)
- Emergency Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Materials For Medical Uses (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
Abstract
Description
where n is 0 or 1.
In still other embodiments, Ar1 is a phenyl that includes 1 or 2 substituents that are electron-withdrawing (e.g., F, Cl, Br, I, CN, or NO2); Ar1 is a phenyl having a NO2 substituent; Ar1 includes 1, 2, or 3 substituents independently selected from halogen and unsubstituted C1-C6 alkyl; Ar2 is unsubstituted phenyl; Ar2 is optionally substituted benzothiophene or optionally substituted phenyl; Ar2 is unsubstituted phenyl or unsubstituted benzothiophene; and/or Ar2 is phenyl having 1, 2, or 3 substituents selected, independently, from NO2, CN, optionally substituted C1-C6 alkyl, or halogen (e.g., the substituents of the phenyl of Ar2 are, independently, selected from F, Cl, Br, and unsubstituted C1-C6 alkyl).
The method also includes administering more than one compound of formula I to the subject (e.g., one or more of
-
- R4 is optionally substituted C1-C6 alkyl or optionally substituted phenyl.
In yet other embodiments, the method includes administering more than one compound of formula II to said subject (e.g., one or more of compounds 10-12, in particular, at
-
- R1 is H or optionally substituted C1-C6 alkyl;
- each Ar1 and Ar2 is, independently, an optionally substituted phenyl group; and
- L1 is a covalent bond, —O(CH2)n—, or —(CH2)nO—, wherein n is an integer between 0-3; and/or
Claims (24)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/017,914 US9648880B2 (en) | 2012-09-04 | 2013-09-04 | Antifungal agents and uses thereof |
| US15/481,596 US10350217B2 (en) | 2012-09-04 | 2017-04-07 | Antifungal agents and uses thereof |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201261696647P | 2012-09-04 | 2012-09-04 | |
| US201361784197P | 2013-03-14 | 2013-03-14 | |
| US14/017,914 US9648880B2 (en) | 2012-09-04 | 2013-09-04 | Antifungal agents and uses thereof |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/481,596 Division US10350217B2 (en) | 2012-09-04 | 2017-04-07 | Antifungal agents and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20140142107A1 US20140142107A1 (en) | 2014-05-22 |
| US9648880B2 true US9648880B2 (en) | 2017-05-16 |
Family
ID=50237745
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/017,914 Active 2033-09-19 US9648880B2 (en) | 2012-09-04 | 2013-09-04 | Antifungal agents and uses thereof |
| US15/481,596 Expired - Fee Related US10350217B2 (en) | 2012-09-04 | 2017-04-07 | Antifungal agents and uses thereof |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/481,596 Expired - Fee Related US10350217B2 (en) | 2012-09-04 | 2017-04-07 | Antifungal agents and uses thereof |
Country Status (2)
| Country | Link |
|---|---|
| US (2) | US9648880B2 (en) |
| WO (1) | WO2014039515A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10350217B2 (en) * | 2012-09-04 | 2019-07-16 | University Of Massachusetts | Antifungal agents and uses thereof |
| US11452291B2 (en) | 2007-05-14 | 2022-09-27 | The Research Foundation for the State University | Induction of a physiological dispersion response in bacterial cells in a biofilm |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2654484C1 (en) | 2012-05-11 | 2018-05-21 | Ресет Терапьютикс, Инк. | Carbazole-containing sulfonamides as modulators of cryptochrome |
| TWI690521B (en) * | 2014-04-07 | 2020-04-11 | 美商同步製藥公司 | Carbazole-containing amides, carbamates, and ureas as cryptochrome modulators |
| WO2019216810A1 (en) * | 2018-05-07 | 2019-11-14 | Constantin Urban | New antifungal compounds |
| CN111777526A (en) * | 2019-04-04 | 2020-10-16 | 中国人民解放军军事科学院军事医学研究院 | N-Benzanilide derivatives, their preparation method and use |
| CA3192377A1 (en) * | 2020-09-11 | 2022-03-17 | The Governing Council Of The University Of Toronto | Piperazinyl compounds and methods for treating nematode infections |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0548798A1 (en) * | 1991-12-18 | 1993-06-30 | Sanwa Kagaku Kenkyusho Co., Ltd. | Antiviral agent |
| US20060009806A1 (en) * | 2003-12-12 | 2006-01-12 | Medtronic Inc | Anti-infective medical device |
| US20080293699A1 (en) | 2007-05-25 | 2008-11-27 | Burnham Institute For Medical Research | Inhibitors of thapsigargin-induced cell death |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7438916B2 (en) * | 2005-10-14 | 2008-10-21 | Virginia Tech Intellectual Properties, Inc. | Therapeutic target for protozoal diseases |
| WO2011097607A1 (en) * | 2010-02-08 | 2011-08-11 | Southern Research Institute | Anti-viral treatment and assay to screen for anti-viral agent |
| WO2014025749A2 (en) * | 2012-08-06 | 2014-02-13 | Sirga Advanced Biopharma, Inc. | Small molecule inhibitors of viral protein interactions with human t-rna |
| WO2014039515A2 (en) * | 2012-09-04 | 2014-03-13 | University Of Massachusetts | Antifungal agents and uses thereof |
-
2013
- 2013-09-04 WO PCT/US2013/057994 patent/WO2014039515A2/en not_active Ceased
- 2013-09-04 US US14/017,914 patent/US9648880B2/en active Active
-
2017
- 2017-04-07 US US15/481,596 patent/US10350217B2/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0548798A1 (en) * | 1991-12-18 | 1993-06-30 | Sanwa Kagaku Kenkyusho Co., Ltd. | Antiviral agent |
| US20060009806A1 (en) * | 2003-12-12 | 2006-01-12 | Medtronic Inc | Anti-infective medical device |
| US20080293699A1 (en) | 2007-05-25 | 2008-11-27 | Burnham Institute For Medical Research | Inhibitors of thapsigargin-induced cell death |
Non-Patent Citations (7)
| Title |
|---|
| Dörwald, F. Zaragoza. Side Reactions in Organic Synthesis: A Guide to Successful Synthesis Design, Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA, 2005, Preface). * |
| EP 0 548798, published Jun. 30, 1993. * |
| International Search Report for International Application No. PCT/US13/57994 mailed Apr. 15, 2014 (4 pages). |
| Jordan, V. C. (Nature Reviews: Drug Discovery, 2, 2003, 205-213). * |
| Patel et al., "Synthesis of novel 3-(5-sulfanyl-1,3,4-oxadiazol-2-yl)-2H-chromen-2-one condensed s-triazinyl piperazines and piperidines as antimicrobial agents," Med Chem Res. 21:3119-3132 (2012). |
| Upadhayaya et al., "Optically active antifungal azoles: synthesis and antifungal activity of (2R,3S)-2-(2,4-difluorophenyl)-3-(5-{2-[4-aryl-piperazin-1-yl]-ethyl}-tetrazol-2-yl/1-yl)-1-[1,2,4]-triazol-1-yl-butan-2-ol.," Bioorg Med Chem. 12(9):2225-38 (2004). |
| Written Opinion of the International Searching Authority for International Application No. PCT/US13/57994 mailed Apr. 15, 2014 (6 pages). |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11452291B2 (en) | 2007-05-14 | 2022-09-27 | The Research Foundation for the State University | Induction of a physiological dispersion response in bacterial cells in a biofilm |
| US10350217B2 (en) * | 2012-09-04 | 2019-07-16 | University Of Massachusetts | Antifungal agents and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| US20170224703A1 (en) | 2017-08-10 |
| WO2014039515A2 (en) | 2014-03-13 |
| US10350217B2 (en) | 2019-07-16 |
| WO2014039515A3 (en) | 2014-06-19 |
| US20140142107A1 (en) | 2014-05-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10350217B2 (en) | Antifungal agents and uses thereof | |
| US20240124392A1 (en) | Anti-fungals compounds targeting the synthesis of fungal sphingolipids | |
| Oliveira et al. | Menthol-based deep eutectic systems as antimicrobial and anti-inflammatory agents for wound healing | |
| US9815794B2 (en) | Inhibition of bacterial biofilms and microbial growth with imidazole derivatives | |
| US9084423B2 (en) | Inhibition of bacterial biofilms with imidazole derivatives | |
| TWI410437B (en) | Antimicrobial copolymers and uses thereof | |
| US20150266827A1 (en) | Antibiotic Tolerance Inhibitors | |
| US20170247409A1 (en) | Abietane-type diterpenoids | |
| CN104486945B (en) | Be used for the treatment of the Compounds and methods for of candidiasis and aspergillus infection | |
| US20180111893A1 (en) | Polycationic amphiphiles and polymers thereof as antimicrobial agents and methods using same | |
| US9938321B2 (en) | Cyclic peptoid oligomers, pharmaceutical compositions and methods of using the same | |
| US20140155478A1 (en) | Antifungal compound and uses thereof | |
| US20190099385A1 (en) | Anti-fungal compounds | |
| Bozkurt-Guzel et al. | Investigation of the in vitro antifungal and antibiofilm activities of ceragenins CSA-8, CSA-13, CSA-44, CSA-131, and CSA-138 against Candida species | |
| Bondaryk et al. | Tetrazole activity against Candida albicans. The role of KEX2 mutations in the sensitivity to (±)-1-[5-(2-chlorophenyl)-2H-tetrazol-2-yl] propan-2-yl acetate | |
| AU2011201932A1 (en) | Treatment or prevention of fungal infections with PDK1 inhibitors | |
| BR112020000463A2 (en) | microbiocidal oxadiazole derivatives | |
| Scorzoni et al. | Antifungal and anti-biofilm effect of the calcium channel blocker verapamil on non-albicans Candida species | |
| Khader et al. | Enhanced antimicrobial activity of tethered multi-cationic surfactants: A novel approach to combat microbial resistance | |
| CN111093655A (en) | Antifungal agents with enhanced activity at acidic pH | |
| WO2023105494A1 (en) | Cationic steroid compounds, method of obtaining thereof, formulations comprising thereof and their uses | |
| US20170056337A1 (en) | Antibacterial s-heterosubstituted disulfides | |
| US20190015400A1 (en) | Antifungal Compositions | |
| US9439876B2 (en) | Method of treating microbial infections | |
| US20240336559A1 (en) | Anti-fungals compounds targeting the synthesis of fungal sphingolipids |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: UNIVERSITY OF MASSACHUSETTS, MASSACHUSETTS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:KAUFMAN, PAUL;DA ROSA-SPIEGLER, JESSICA LOPES;REEL/FRAME:033994/0429 Effective date: 20140805 |
|
| AS | Assignment |
Owner name: WORCESTER POLYTECHNIC INSTITUTE, MASSACHUSETTS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:RAO, REETA PRUSTY;REEL/FRAME:037979/0513 Effective date: 20160212 Owner name: UNIVERSITY OF MASSACHUSETTS, MASSACHUSETTS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:FAZLY, AHMED;REEL/FRAME:037979/0475 Effective date: 20160301 |
|
| AS | Assignment |
Owner name: NATIONAL INSTITUTES OF HEALTH (NIH), U.S. DEPT. OF Free format text: CONFIRMATORY LICENSE;ASSIGNOR:UNIVERSITY OF MASSACHUSETTS MEDICAL SCH;REEL/FRAME:042170/0148 Effective date: 20170405 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| CC | Certificate of correction | ||
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YR, SMALL ENTITY (ORIGINAL EVENT CODE: M2551); ENTITY STATUS OF PATENT OWNER: SMALL ENTITY Year of fee payment: 4 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YR, SMALL ENTITY (ORIGINAL EVENT CODE: M2552); ENTITY STATUS OF PATENT OWNER: SMALL ENTITY Year of fee payment: 8 |