FIELD OF THE INVENTION
The present invention relates to liquid-crystalline media, in particular for high-frequency technology, especially components for high-frequency devices, in particular antennas, especially for the gigahertz region and the terahertz region, which are operated in the microwave or millimeter wave region. These components use particular mesogenic, preferably liquid-crystalline, chemical compounds or liquid-crystalline media composed thereof for, for example, the phase shifting of microwaves for tuneable phased-array antennas or for tuneable cells of microwave antennas based on “reflectarrays”. In addition, the present invention relates to novel mesogenic compounds.
PRIOR ART AND PROBLEM TO BE SOLVED
Liquid-crystalline media have long been utilised in electro-optical displays (liquid crystal displays—LCDs) in order to display information.
Bistolan compounds, also known as triphenyldiacetylenes, having an additional alkyl substitution on the central phenylene ring are adequately known to the person skilled in the art.
For example, Wu, S.-T., Hsu, C.-S, and Shyu, K.-F., Appl. Phys. Lett., 74 (3), (1999), pages 344-346, discloses various liquid-crystalline bistolan compounds containing a lateral methyl group, of the formula
Besides liquid-crystalline bistolan compounds of this type containing a lateral methyl group, Hsu, C. S., Shyu, K. F., Chuang, Y. Y. and Wu, S.-T., Liq. Cryst., 27 (2), (2000), pages 283-287, also discloses corresponding compounds containing a lateral ethyl group and proposes the use thereof, inter alia, in liquid crystal optically phased arrays.
Dabrowski, R., Kula, P., Gauza, S., Dziadiszek, J., Urban, S, and Wu, S.-T., IDRC 08, (2008), pages 35-38, mentions dielectrically neutral bistolan compounds with and without a lateral methyl group on the central ring besides the strongly dielectrically positive isothiocyanatobistolan compounds of the formula
Compounds containing three C—C triple bonds, such as, for example, the compound
are mentioned in JP 2003-207631 A) and are proposed for use in optical films, polarisers and in liquid crystals of the light-scattering type.
Compounds of the General Formula
are described in EP 1 655 360 A1.
However, liquid-crystalline media have recently also been proposed for use in components for microwave technology, as described, for example, in DE 10 2004 029 429 A and in JP 2005-120208 (A).
An industrially valuable application of liquid-crystalline media in high-frequency technology is based on their property that their dielectric properties can be controlled, particularly for the gigahertz region and the terahertz region, by a variable voltage. This enables the construction of tuneable antennas which contain no moving parts (Gaebler, A., Moessinger, A., Goelden, F., et al., “Liquid Crystal-Reconfigurable Antenna Concepts for Space Applications at Microwave and Millimeter Waves”, International Journal of Antennas and Propagation, Volume 2009, Article ID 876989, (2009), pages 1-7, DOI: 10.1155/2009/876989).
Penirschke, A., Müller, S., Scheele, P., Weil, C., Wittek, M., Hock, C. and Jakoby, R.: “Cavity Perturbation Method for Characterisation of Liquid Crystals up to 35 GHz”, 34th European Microwave Conference—Amsterdam, pp. 545-548, describe, inter alia, the properties of the known single liquid-crystalline substance K15 (also called 4-n-pentyl-4′-cyanobiphenyl or PP-5-N, Merck KGaA, Germany) at a frequency of 9 GHz.
DE 10 2004 029 429 A describes the use of liquid-crystal media in microwave technology, inter alia in phase shifters. DE 10 2004 029 429 A has already investigated liquid-crystalline media with respect to their properties in the corresponding frequency range.
For use in high-frequency technology, liquid-crystalline media having particular, hitherto rather unusual, unconventional properties, or combinations of properties, are required.
A. Gaebler, F. Goelden, S. Müller, A. Penirschke and R. Jakoby “Direct Simulation of Material Permittivites using an Eigen-Susceptibility Formulation of the Vector Variational Approach”, 12MTC 2009—International Instrumentation and Measurement Technology Conference, Singapore, 2009 (IEEE), pp. 463-467, describe the corresponding properties of the known liquid-crystal mixture E7 (likewise Merck KGaA, Germany).
DE 10 2004 029 429 A describes the use of liquid-crystal media in microwave technology, inter alia in phase shifters. DE 10 2004 029 429 A has already investigated liquid-crystalline media with respect to their properties in the corresponding frequency range. In addition, it mentions liquid-crystalline media which comprise compounds of the formulae
besides compounds of the formulae
Liquid-crystal media which comprise, for example, compounds of the formula
are proposed for use in components for high-frequency technology, for example, in A. Lapanik, “Single compounds and mixtures for microwave applications, Dielectric, microwave studies on selected systems”, dissertation, Technical University of Darmstadt, 2009, (D17).
However, the compositions known to date are afflicted with serious disadvantages. Besides other deficiencies, most of them result in disadvantageously high losses and/or inadequate phase shift or inadequate material quality (η).
Novel liquid-crystalline media having improved properties are thus necessary. In particular, the loss in the microwave region and/or millimeter wave region must be reduced and the material quality improved.
In addition, there is a demand for an improvement in the low-temperature behaviour of the liquid-crystalline media and thus also of the components. Both an improvement in the operating properties and also in the shelf life are necessary here.
Thus, there is a considerable demand for liquid-crystalline media having suitable properties for corresponding practical applications.
PRESENT INVENTION
Surprisingly, it has now been found that it is possible to achieve components for high-frequency technology which do not have the disadvantages of the prior-art materials, or at least only do so to a considerably reduced extent, if selected liquid-crystalline media are employed.
The present invention thus relates to liquid-crystalline media which comprise one or more compounds of the formula IA,
very particularly preferably
- L1 denotes alkyl having 1 to 6 C atoms, cycloalkyl having 3 to 6 C atoms, cycloalkenyl having 4 to 6 C atoms or halogen, preferably CH3, C2H5, n-C3H7 (—(CH2)2CH3), i-C3H7(—CH(CH3)2), cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclopent-1-enyl, cyclohex-1-enyl, F or Cl and particularly preferably CH3, C2H5, cyclopropyl, cyclobutyl or F and very particularly preferably F,
- Y11 and Y12, independently of one another, denote H, alkyl having 1 to 6 C atoms, cycloalkyl having 3 to 6 C atoms, cycloalkenyl having 4 to 6 C atoms or halogen, preferably H, CH3, C2H5, n-C3H7 (—(CH2)2CH3), i-C3H7 (—CH(CH3)2), cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclopent-1-enyl, cyclohex-1-enyl, F or Cl and particularly preferably H, CH3, C2H5 or F and very particularly preferably H or CH3,
- R11 to R13, independently of one another, denote unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 15 C atoms, unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 15 C atoms, or cycloalkyl, alkylcycloalkyl, cycloalkenyl, alkylcycloalkenyl, alkylcycloalkylalkyl or alkylcycloalkenylalkyl, each having up to 15 C atoms,
preferably
- R11 and R12, independently of one another, denote unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 7 C atoms, or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 7 C atoms,
particularly preferably
- R11 denotes unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 7 C atoms, and
particularly preferably
- R12 denotes unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 7 C atoms, and
preferably
- R13 denotes unfluorinated alkyl having 1 to 5 C atoms, unfluorinated cycloalkyl or cycloalkenyl having 3 to 7 C atoms, unfluorinated alkylcyclohexyl or unfluorinated cyclohexylalkyl, each having 4 to 12 C atoms, or unfluorinated alkylcyclohexylalkyl having 5 to 15 C atoms, particularly preferably cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, and very particularly preferably R13 denotes n-alkyl, particularly preferably methyl, ethyl or n-propyl, and the other denotes H or n-alkyl, particularly preferably H, methyl, ethyl or n-propyl.
The liquid-crystal media according to the invention are eminently suitable for use in components for high-frequency technology or for the microwave region and/or millimeter wave region of the electromagnetic spectrum. The present invention relates to this use of the media and to these components.
In a first preferred embodiment of the present invention, the component for high-frequency technology contains a liquid-crystal medium which comprises a component A which consists of one compound, two compounds or a plurality of compounds of the formula IA.
In accordance with a further preferred embodiment of the present invention, the component for high-frequency technology contains a liquid-crystalline medium comprising
-
- a first component, component A, which comprises one or more compounds of the above-mentioned formula IA and which optionally comprises one or more compounds of the formula D3 given below and
- one or more further components selected from the group of components B to E defined below,
- a strongly dielectrically positive component, component B, which has a dielectric anisotropy of 10.0 or more,
- a strongly dielectrically negative component, component C, which has a dielectric anisotropy of −5.0 or less,
- a further component, component D, which has a dielectric anisotropy in the range from more than −5.0 to less than 10.0 and consists of compounds having seven or more, preferably eight or more, five- or six-membered rings, and
- a further component, component E, which likewise has a dielectric anisotropy in the range from more than −5.0 to less than 10.0 and consists of compounds having up to six five- or six-membered rings.
Typical examples of five-membered rings are
Typical examples of six-membered rings are
The five- and six-membered rings also include saturated and partially saturated rings, as well as heterocyclic rings.
For the purposes of the present application, condensed ring systems which consist of two of these rings, i.e. two five-membered rings, one five-membered ring or two six-membered rings, such as, for example,
are counted as one of these five- or six-membered rings on assignment of the compounds to components A or D.
Correspondingly, condensed ring systems which consist of a combination of three or more of these rings which are incorporated into the molecule in the longitudinal direction, such as, for example,
are counted as two of these five- or six-membered rings.
By contrast, condensed ring systems which are incorporated into the molecule in the transverse direction, such as, for example,
are counted as one of these five- or six-membered rings.
The present invention likewise relates to the directly preceding liquid-crystalline media and to those described below, and to the use thereof in electro-optical displays and in particular in components for high-frequency technology.
In a preferred embodiment of the present invention, the liquid-crystal medium comprises one or more compounds of the formula IA, preferably selected from the group of the compounds of the formulae IA-1 to IA-4, preferably of the formulae IA-1 and/or IA-2:
in which the parameters have the respective meanings indicated above for formula IA, and preferably
- R13 denotes alkyl having 1 to 6 C atoms, alkenyl having 2 to 6 C atoms, cycloalkyl having 3 to 6 C atoms or cycloalkenyl having 4 to 6 C atoms, preferably CH3, C2H5, n-C3H7 (—(CH2)2CH3), i-C3H7 (—CH(CH3)2), —CH═CH2, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclopent-1-enyl or cyclohex-1-enyl, and particularly preferably CH3, C2H5, cyclopropyl or cyclobutyl,
- L1 denotes alkyl having 1 to 3 C atoms or halogen, preferably H, F or Cl, and particularly preferably H, F or CH3, even more preferably H or F and very particularly preferably F,
- R11 denotes unfluorinated alkyl having 1 to 7 C atoms, and
- R12 denotes unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkoxy having 1 to 7 C atoms.
In a preferred embodiment of the present invention, component A consists predominantly, even more preferably consists essentially and very particularly preferably consists completely of compounds of the formula IA.
In a particularly preferred embodiment of the present invention, the liquid-crystal medium comprises one or more compounds of the formula IA-1, preferably selected from the group of the compounds of the formulae IA-1a to IA-1e and IA-2a to IA-2e, particularly preferably selected from the group of the compounds of the formulae IA-1a to IA-1c, IA-1e, IA-2a to IA-2c and IA-2e, very particularly preferably selected from the group of the compounds of the formulae IA-1a, IA-1b, IA-2a and IA-2b,
in which the parameters have the meanings given above under formula IA-1 or under formula IA-2, and preferably
- R11 and R12, independently of one another, denote an alkyl radical having 2 to 7 C atoms, for example a propyl radical and a hexyl radical or in each case a propyl, butyl, pentyl or hexyl radical.
The liquid-crystal medium in component A optionally comprises one or more compounds of the formula IB,
- L1 denotes alkyl having 1 to 6 C atoms, cycloalkyl having 3 to 6 C atoms or cycloalkenyl having 4 to 6 C atoms, preferably CH3, C2H5, n-C3H7 (—(CH2)2CH3), i-C3H7 (—CH(CH3)2), cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclopent-1-enyl or cyclohex-1-enyl, and particularly preferably CH3, C2H5, cyclopropyl or cyclobutyl,
- X1 denotes H, alkyl having 1 to 3 C atoms or halogen, preferably H, F or Cl, particularly preferably H or F and very particularly preferably F,
- R11 to R14, independently of one another, denote unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 15 C atoms, unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 15 C atoms, or cycloalkyl, alkylcycloalkyl, cycloalkenyl, alkylcycloalkenyl, alkylcycloalkylalkyl or alkylcycloalkenylalkyl, each having up to 15 C atoms, and alternatively one of R13 and R14 or both also denote H,
preferably R11
and R12, independently of one another, denote unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 7 C atoms, or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 7 C atoms,
particularly
preferably R11 denotes unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 7 C atoms, and
particularly
preferably R12 denotes unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 7 C atoms, and
preferably R13 and R14 denote H, unfluorinated alkyl having 1 to 5 C atoms, unfluorinated cycloalkyl or cycloalkenyl having 3 to 7 C atoms, unfluorinated alkylcyclohexyl or unfluorinated cyclohexylalkyl, each having 4 to 12 C atoms, or unfluorinated alkylcyclohexylalkyl having 5 to 15 C atoms, particularly preferably cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, and very particularly preferably at least one of R13 and R14 denotes n-alkyl, particularly preferably methyl, ethyl or n-propyl, and the other denotes H or n-alkyl, particularly preferably H, methyl, ethyl or n-propyl.
In a particularly preferred embodiment of the present invention, component A comprises one or more compounds of the formula IA and one or more compounds of the formula IB, preferably component A consists predominantly, even more preferably consists essentially and very particularly preferably consists completely of compounds of the formulae IA and IB.
The compounds of the formula IB are preferably compounds selected from the group of the compounds of the formulae IB-1 to IB-4
- L1 denotes alkyl having 1 to 6 C atoms, alkenyl having 2 to 6 C atoms, cycloalkyl having 3 to 6 C atoms or cycloalkenyl having 4 to 6 C atoms, preferably CH3, C2H5, n-C3H7(—(CH2)2CH3), i-C3H7 (—CH(CH3)2), —CH═CH2, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclopent-1-enyl or cyclohex-1-enyl, and particularly preferably CH3, C2H5, cyclopropyl or cyclobutyl,
- X1 denotes H, alkyl having 1 to 3 C atoms or halogen, preferably H, F or Cl, particularly preferably H, F or CH3, even more preferably H or F and very particularly preferably F,
and the other parameters have the respective meanings indicated above for formula IB, and preferably
- R11 denotes unfluorinated alkyl having 1 to 7 C atoms, and
- R12 denotes unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkoxy having 1 to 7 C atoms.
In a particularly preferred embodiment of the present invention, the liquid-crystal medium comprises one or more compounds of the formula IB-1, preferably selected from the group of the compounds of the formulae IB-1a-1 to IB-1a-12 and IB-1b-1 to IB-1b-12
in which the parameters have the meanings given above under formula IB-1, and preferably
R11 and R12, independently of one another, denote an alkyl radical having 2 to 7 C atoms, for example a propyl radical and a hexyl radical or in each case a propyl, butyl, pentyl or hexyl radical.
In a very particularly preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB, preferably selected from the group of the compounds of the formulae IB-1a-2, IB-1a-5, IB-1a-7, IB-1a-8, IB-1a-9, IB-1a-10, IB-1b-5, IB-1b-7, IB-1b-8, IB-1b-9, IB-1b-10, where the parameters have the meanings given above, and particularly preferably
- R11 and R12, independently of one another, denote unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkoxy having 1 to 6 C atoms,
especially preferably one of
- R11 and R12 denotes alkyl and the other denotes alkyl or alkoxy, and very particularly preferably R11 and R12 have different meanings from one another.
In a preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB-2, in which preferably
- R11 and R12, independently of one another, denote an alkyl radical having 2 to 7 C atoms, for example a propyl radical and a hexyl radical or in each case a propyl, butyl, pentyl or hexyl radical.
In a preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB-3, preferably selected from the group of the compounds of the formulae IB-3a-1 to IB-3a-3 and IB-3b-1 to IB-3b-3, preferably IB-3a-2, IB-3b-2,
in which the parameters have the meanings given above under formula IB-3, and preferably
- R11 and R12, independently of one another, denote an alkyl radical having 2 to 7 C atoms, for example a propyl radical and a hexyl radical or in each case a propyl, butyl, pentyl or hexyl radical.
In a preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB-4, preferably selected from the group of the compounds of the formulae IB-4-a-1 to IB-4-a-3 and IB-4-b-1 to IB-4-b-3, preferably IB-4-b-2,
in which the parameters have the meanings given above under formula IB-4, and preferably
- R11 and R12, independently of one another, denote an alkyl radical having 2 to 7 C atoms, for example a propyl radical and a hexyl radical or in each case a propyl, butyl, pentyl or hexyl radical.
The compounds of the formula IA can advantageously be prepared as evident from the following illustrative synthesis (Schemes 1 and 2):
The compounds of the formula IB-1a can advantageously be prepared as evident from the following illustrative synthesis (Schemes 3-6):
The compounds of the formula IB-1b can advantageously be obtained in accordance with the following general reaction schemes (Reaction Schemes 5 to 11). The parameters L1, R11, R12 and X1 therein are as defined above and below. R has the meaning of R11 or R12.
Schemes 9 to 13 show the synthesis of variously substituted central rings. The phenylalkynyl radicals here can be generalised to any desired substituted phenylalkynyl radicals.
The compounds of the formula IB-2 can advantageously be prepared as evident from the following illustrative synthesis (Scheme 14):
The compounds of the formulae IB-3 and IB-4 can advantageously be obtained in accordance with the following general reaction scheme (Reaction Scheme 15).
Besides component A, these media according to the invention preferably comprise a component selected from the two components B and C and optionally additionally component D and/or component E.
These media according to the invention preferably comprise two, three or four, particularly preferably two or three, components selected from the group of components A to E. These media preferably comprise
-
- component A and component B, or
- component A, component B and component D and/or E, or
- component A and component C, or
- component A, component B and component C, or
- component A, component C and component D and/or E.
These media according to the invention preferably comprise a component B and no component C or vice versa.
The strongly dielectrically positive component, component B, preferably has a dielectric anisotropy of 20.0 or more, more preferably 25.0 or more, particularly preferably 30.0 or more and very particularly preferably 40.0 or more.
The strongly dielectrically negative component, component C, preferably has a dielectric anisotropy of −7.0 or less, more preferably −8.0 or less, particularly preferably −10.0 or less and very particularly preferably −15.0 or less.
In a preferred embodiment of the present invention, component B comprises one or more compounds selected from the group of the compounds of the formulae IIA and IIB:
- R21 denotes unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 15 C atoms, or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl, each having 2 to 15 C atoms, preferably alkyl, particularly preferably n-alkyl,
- R22 denotes H, unfluorinated alkyl or unfluorinated alkoxy, each having 1 to 5, preferably 1 to 3, particularly preferably 3, C atoms,
-
- independently of one another and, if they occur more than once, these also in each case independently of one another, denote
- n and m, independently of one another, denote 1 or 2, preferably
- (n+m) denotes 3 or 4, and particularly preferably
- n denotes 2,
- X2 denotes F, Cl, —CF3 or —OCF3, preferably F or Cl, particularly preferably F,
- Y2 denotes F, Cl, —CF3, —OCF3 or CN, preferably CN, and
- Z2 denotes H or F.
Preferred compounds of the formula IIA are the compounds of the corresponding sub-formula IIA-1
in which R
21 has the meaning given above.
Preferred compounds of the formula IIB are the compounds of the corresponding sub-formulae IIB-1 and IIB-2:
in which R
21, R
22 and X
2 have the respective meanings given above.
In a preferred embodiment of the present invention, component C comprises one or more compounds selected from the group of the compounds of the formulae IIIA and IIIB:
- R31 and R32, independently of one another, have the meanings indicated above for R21 under formula IIA,
and preferably
- R31 denotes CnH2n+1 or CH2═CH—(CH2)Z and
- R32 denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2,
and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R31 and R32) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1).
Preferred compounds of the formula IIIB are the compounds of the sub-formulae IIIB-1 and IIIB-2:
- n and m each have the meanings given above for formula MB and preferably, independently of one another, denote an integer in the range from 1 to 7.
In a preferred embodiment of the present invention, component D comprises one or more compounds of the following formula IV:
- R41 and R42, independently of one another, have one of the meanings indicated above for R11 under formula I,
- L41 to L44 on each appearance, in each case independently of one another, denote H, alkyl having 1 to 5 C atoms, F or Cl, and
- p denotes an integer in the range from 7 to 14, preferably from 8 to 12 and particularly preferably from 9 to 10,
and preferably
at least two of the substituents
- L41 to L44 present have a meaning other than H, and
- R41 denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R42 denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2,
and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
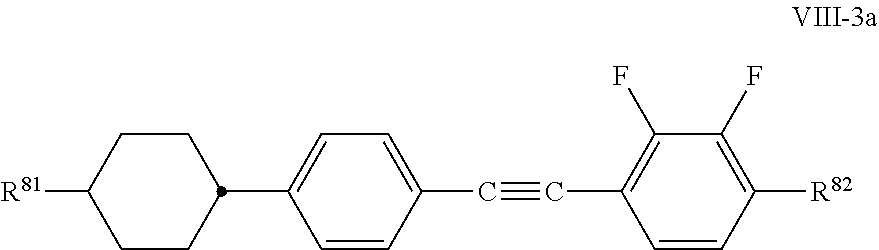
In a preferred embodiment of the present application, the liquid-crystal medium additionally comprises a further component, component E, which preferably consists of one or more compounds selected from the group of the compounds of the formulae V to IX:
- L51 denotes R51 or X51,
- L52 denotes R52 or X52,
- R51 and R52, independently of one another, denote H, unfluorinated alkyl or unfluorinated alkoxy having 1 to 17, preferably 3 to 10, C atoms or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl having 2 to 15, preferably 3 to 10, C atoms, preferably alkyl or unfluorinated alkenyl,
- X51 and X52, independently of one another, denote H, F, Cl, —CN, —NCS, —SF5, fluorinated alkyl or fluorinated alkoxy having 1 to 7 C atoms or fluorinated alkenyl, unfluorinated or fluorinated alkenyloxy or unfluorinated or fluorinated alkoxyalkyl having 2 to 7 C atoms, preferably fluorinated alkoxy, fluorinated alkenyloxy, F or Cl, and
-
- independently of one another, denote
- L61 denotes R61 and, in the case where Z61 and/or Z62 denote trans-CH═CH— or trans-CF═CF—, alternatively also denotes X61,
- L62 denotes R62 and, in the case where Z61 and/or Z62 denote trans-CH═CH— or trans-CF═CF—, alternatively also denotes X62,
- R61 and R62, independently of one another, denote H, unfluorinated alkyl or unfluorinated alkoxy having 1 to 17, preferably 3 to 10, C atoms or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl having 2 to 15, preferably 3 to 10, C atoms, preferably alkyl or unfluorinated alkenyl,
- X61 and X62, independently of one another, denote F or Cl, —CN, —NCS, —SF5, fluorinated alkyl or alkoxy having 1 to 7 C atoms or fluorinated alkenyl, alkenyloxy or alkoxyalkyl having 2 to 7 C atoms, preferably —NCS,
one of
- Z61 and Z62 denotes trans-CH═CH—, trans-CF═CF— or —C≡C— and the other, independently thereof, denotes trans-CH═CH—, trans-CF═CF— or a single bond, preferably one of them denotes —C≡C— or trans-CH═CH— and the other denotes a single bond, and
-
- independently of one another, denote
- L71 denotes R71 or X71,
- L72 denotes R72 or X72,
- R71 and R72, independently of one another, denote H, unfluorinated alkyl or unfluorinated alkoxy having 1 to 17, preferably 3 to 10, C atoms or unfluorinated alkenyl, unfluorinated alkenyloxy or unfluorinated alkoxyalkyl having 2 to 15, preferably 3 to 10, C atoms, preferably alkyl or unfluorinated alkenyl,
- X71 and X72, independently of one another, denote H, F, Cl, —CN, —NCS, —SF5, fluorinated alkyl or fluorinated alkoxy having 1 to 7 C atoms or fluorinated alkenyl, unfluorinated or fluorinated alkenyloxy or unfluorinated or fluorinated alkoxyalkyl having 2 to 7 C atoms, preferably fluorinated alkoxy, fluorinated alkenyloxy, F or Cl, and
- Z71 to Z73, independently of one another, denote trans-CH═CH—, trans-CF═CF—, —C≡C— or a single bond, preferably one or more of them denote a single bond, particularly preferably all denote a single bond and
-
- independently of one another, denote
- R81 and R82, independently of one another, denote H, unfluorinated alkyl or alkoxy having 1 to 15, preferably 3 to 10, C atoms or unfluorinated alkenyl, alkenyloxy or alkoxyalkyl having 2 to 15, preferably 3 to 10, C atoms, preferably unfluorinated alkyl or alkenyl,
one of
- Z81 and Z82 denotes trans-CH═CH—, trans-CF═CF— or —C≡C— and the other, independently thereof, denotes trans-CH═CH—, trans-CF═CF— or a single bond, preferably one of them denotes —C≡C— or trans-CH═CH— and the other denotes a single bond, and
-
- independently of one another, denote
- L91 denotes R91 or X91,
- L92 denotes R92 or X92,
- R91 and R92, independently of one another, denote H, unfluorinated alkyl or alkoxy having 1 to 15, preferably 3 to 10, C atoms or unfluorinated alkenyl, alkenyloxy or alkoxyalkyl having 2 to 15, preferably 3 to 10, C atoms, preferably unfluorinated alkyl or alkenyl,
- X91 and X92, independently of one another, denote H, F, Cl, —CN, —NCS, —SF5, fluorinated alkyl or fluorinated alkoxy having 1 to 7 C atoms or fluorinated alkenyl, unfluorinated or fluorinated alkenyloxy or unfluorinated or fluorinated alkoxyalkyl having 2 to 7 C atoms, preferably fluorinated alkoxy, fluorinated alkenyloxy, F or Cl, and
- Z91 to Z93, independently of one another, denote trans-CH═CH—, trans-CF═CF—, —C≡C— or a single bond, preferably one or more of them denotes a single bond, and particularly preferably all denote a single bond,
-
- independently of one another, denote
and where compounds of the formula IIIA are excluded from the compounds of the formula VI.
In a preferred embodiment of the present invention, the liquid-crystal medium comprises, more preferably predominantly consists of, even more preferably essentially consists of and very particularly preferably completely consists of one or more compounds of the formula V, preferably selected from the group of the compounds of the formulae V-1 to V-3, preferably of the formulae V-1 and/or V-2 and/or V-3, preferably of the formulae V-1 and V-2:
in which the parameters have the respective meanings indicated above for formula V and preferably
- R51 denotes unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkenyl having 2 to 7 C atoms,
- R52 denotes unfluorinated alkyl having 1 to 7 C atoms or unfluorinated alkenyl having 2 to 7 C atoms or unfluorinated alkoxy having 1 to 7 C atoms,
- X51 and X52, independently of one another, denote F, Cl, —OCF3, —CF3, —CN, —NCS or —SFS, preferably F, Cl, —OCF3 or —CN.
The compounds of the formula V-1 are preferably selected from the group of the compounds of the formulae V-1a to V-1d, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
in which the parameters have the respective meanings indicated above for formula V-1 and in which
- Y51 and Y52, in each case independently of one another, denote H or F, and preferably
- R51 denotes alkyl or alkenyl, and
- X51 denotes F, Cl or —OCF3.
The compounds of the formula V-2 are preferably selected from the group of the compounds of the formulae V-2a to V-2g and/or from the group of the compounds of the formulae V-2f and V-2g, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
where in each case the compounds of the formula V-2a are excluded from the compounds of the formulae V-2b and V-2c, the compounds of the formula V-2b are excluded from the compounds of the formula V-2c and the compounds of the formula V-2e are excluded from the compounds of the formula V-2f, and
in which the parameters have the respective meanings indicated above for formula V-1 and in which
- Y51 and Y52, in each case independently of one another, denote H or F, and preferably
- R51 denotes alkyl or alkenyl,
- X51 denotes F, Cl or —OCF3, and preferably one of
- Y51 and Y52 denotes H and the other denotes H or F, preferably likewise denotes H.
The compounds of the formula V-3 are preferably compounds of the formula V-3a:
in which the parameters have the respective meanings indicated above for formula V-1 and in which preferably
- X51 denotes F, Cl, preferably F,
- X52 denotes F, Cl or —OCF3, preferably —OCF3.
In an even more preferred embodiment of the present invention, the compounds of the formula V are selected from the group of the compounds V-1a to V-1d, preferably selected from the group of the compounds V-1c and V-1d, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
The compounds of the formula V-1a are preferably selected from the group of the compounds of the formulae V-1a-1 and V-1a-2, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R51 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes an integer in the range from 0 to 7, preferably in the range from 1 to 5 and particularly preferably 3 or 7.
The compounds of the formula V-1b are preferably compounds of the formula V-1b-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5.
The compounds of the formula V-1c are preferably selected from the group of the compounds of the formulae V-1c-1 to V-1c-4, preferably selected from the group of the compounds of the formulae V-1c-1 and V-1c-2, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R51 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5.
The compounds of the formula V-1d are preferably selected from the group of the compounds of the formulae V-1d-1 and V-1d-2, preferably the compound of the formula V-1d-2, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R51 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5.
The compounds of the formula V-2a are preferably selected from the group of the compounds of the formulae V-2a-1 and V-2a-2, preferably the compounds of the formula V-2a-1, more preferably these compounds of the formula V predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
Preferred combinations of (R51 and R52), in particular in the case of formula V-2a-1, are (CnH2n+1 and CmH2m+1), (CnH2n+1 and O—CmH2m+i), (CH2═CH—(CH2)Z and CmH2m+1), (CH2═CH—(CH2)Z and O—CmH2m+1) and (CnH2n+1 and (CH2)Z—CH═CH2).
Preferred compounds of the formula V-2b are the compounds of the formula V-2b-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combination of (R51 and R52) here is, in particular, (CnH2n+1 and CmH2m+1).
Preferred compounds of the formula V-2c are the compounds of the formula V-2c-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combination of (R51 and R52) here is, in particular, (CnH2n+1 and CmH2m+1).
Preferred compounds of the formula V-2d are the compounds of the formula V-2d-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combination of (R51 and R52) here is, in particular, (CnH2n+1 and CmH2m+1).
Preferred compounds of the formula V-2e are the compounds of the formula V-2e-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combination of (R51 and R52) here is, in particular, (CnH2n+1 and O—CmH2m+1).
Preferred compounds of the formula V-2f are the compounds of the formula V-2f-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R51 and R52) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
Preferred compounds of the formula V-2g are the compounds of the formula V-2g-1:
- R51 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R52 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R51 and R52) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and O—CmH2m+1).
The compounds of the formula VI are preferably selected from the group of the compounds of the formulae VI-1 to VI-4, more preferably these compounds of the formula VI predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- Z61 and Z62 denote trans-CH═CH— or trans-CF═CF—, preferably trans-CH═CH—, and the other parameters have the meaning given above under formula VI and preferably
- R61 and R62, independently of one another, denote H, unfluorinated alkyl or alkoxy having 1 to 7 C atoms or unfluorinated alkenyl having 2 to 7 C atoms,
- X62 denotes F, Cl, —CN or —NCS, preferably —NCS,
and one of
-
- and the others, independently of one another, denote
-
- R61 denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R62 denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The compounds of the formula VI-1 are preferably selected from the group of the compounds of the formulae VI-1a and VI-1b, preferably selected from compounds of the formula VI-1a, more preferably these compounds of the formula VI predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R61 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R62 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R61 and R62) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), in the case of formula VI-1a particularly preferably (CnH2n+1 and CmH2m+1) and in the case of formula VI-1b particularly preferably (CnH2n+1 and O—CmH2m+1).
The compounds of the formula VI-3 are preferably compounds of the formula VI-3a:
in which the parameters have the meaning given above under formula VI-3 and preferably
- R61 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes an integer in the range from 0 to 7, preferably in the range from 1 to 5, and
- X62 denotes —F, —Cl, —OCF3, —CN or —NCS, particularly preferably —NCS.
The compounds of the formula VI-4 are preferably compounds of the formula VI-4-a:
in which the parameters have the meaning given above under formula VI-4 and preferably
- R61 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes an integer in the range from 0 to 7, preferably in the range from 1 to 5, and
- X62 denotes F, Cl, OCF3, —CN or —NCS, particularly preferably —NCS.
Further preferred compounds of the formula VI are the compounds of the following formulae:
- n denotes an integer in the range from 0 to 7, preferably in the range from 1 to 5.
The compounds of the formula VII are preferably selected from the group of the compounds of the formulae VII-1 to VII-6, more preferably these compounds of the formula VII predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
where the compounds of the formula VII-5 are excluded from the compounds of the formula VII-6, and
in which the parameters have the respective meanings indicated above for formula VII, and preferably
- R71 denotes unfluorinated alkyl or alkoxy, each having 1 to 7 C atoms, or unfluorinated alkenyl having 2 to 7 C atoms,
- R72 denotes unfluorinated alkyl or alkoxy, each having 1 to 7 C atoms, or unfluorinated alkenyl having 2 to 7 C atoms, and
- X72 denotes F, Cl or —OCF3, preferably F, and
particularly preferably
- R71 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R72 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The compounds of the formula VIII-1 are preferably selected from the group of the compounds of the formulae VII-1a to VII-1d, more preferably these compounds of the formula VII-1 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
in which X
72 has the meaning given above for formula VII-2 and
- R71 has the meaning indicated above and preferably denotes CnH2n+1, in which
- n denotes 1 to 7, preferably 2 to 6, particularly preferably 2, 3 or 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2, and
- X72 preferably denotes F.
The compounds of the formula VIII-2 are preferably selected from the group of the compounds of the formulae VIII-2a and VII-2b, preferably of the formula VII-2a, more preferably these compounds of the formula VII-2 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R71 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R72 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R71 and R72) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-3 are preferably compounds of the formula VII-3a:
- R71 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R72 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R71 and R72) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-4 are preferably compounds of the formula VII-4-a:
- R71 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R72 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R71 and R72) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-5 are preferably selected from the group of the compounds of the formulae VIII-5a and VII-5b, preferably of the formula VII-5a, more preferably these compounds of the formula VII-5 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R71 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R72 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R71 and R72) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-6 are preferably selected from the group of the compounds of the formulae VIII-6a and VII-6b, more preferably these compounds of the formula VII-6 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R71 has the meaning indicated above and preferably denotes CnH2n+1 or CH2—CH—(CH2)Z, and
- R72 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R71 and R72) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The liquid-crystalline media in accordance with the present application preferably comprise in total 0 to 40%, preferably 0 to 30% and particularly preferably 5 to 25%, of compounds of the formula VIII.
The compounds of the formula VIII are preferably selected from the group of the compounds of the formulae VIII-1 to VIII-3, more preferably these compounds of the formula VIII predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- Y81 and Y82 denotes H and the other denotes H or F, and
- R81 has the meaning indicated above and preferably denotes CnH2n+1 or CH2—CH—(CH2)Z, and
- R82 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R81 and R82) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+i), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-1 are preferably selected from the group of the compounds of the formulae VIII-1a to VIII-1c, more preferably these compounds of the formula VIII-1 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R81 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R82 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R81 and R82) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-2 are preferably compounds of the formula VIII-2a:
- R81 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R82 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R81 and R82) here are, in particular, (CnH2n+1 and CmH2m+1), (CnH2n+1 and O—CmH2m+1) and (CH2═CH—(CH2)Z and CmH2m+1), particularly preferably (CnH2n+1 and CmH2m+1).
The compounds of the formula VIII-3 are preferably compounds of the formula VIII-3a:
- R81 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R82 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R81 and R82) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1).
The compounds of the formula IX are preferably selected from the group of the compounds of the formulae IX-1 to IX-3, more preferably these compounds of the formula IX predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
in which the parameters have the respective meaning indicated above under formula IX and preferably
one of
- R91 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R92 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R91 and R92) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1).
The liquid-crystalline media in accordance with the present application preferably comprise in total 5 to 30%, preferably 10 to 25% and particularly preferably 15 to 20%, of compounds of the formula IX.
The compounds of the formula IX-1 are preferably selected from the group of the compounds of the formulae IX-1a to IX-1e, more preferably these compounds of the formula IX-1 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
in which the parameters have the meaning given above and preferably
- R91 has the meaning indicated above and preferably denotes CnH2n+1, and
- n denotes an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- X92 preferably denotes F or Cl.
The compounds of the formula IX-2 are preferably selected from the group of the compounds of the formulae IX-2a and IX-2b, more preferably these compounds of the formula IX-2 predominantly consist, even more preferably essentially consist and very particularly preferably completely consist thereof:
- R91 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R92 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combination of (R91 and R92) here is, in particular, (CnH2n+1 and CmH2m+1).
The compounds of the formula IX-3 are preferably compounds of the formulae IX-3a and IX-3b:
- R91 has the meaning indicated above and preferably denotes CnH2n+1 or CH2═CH—(CH2)Z, and
- R92 has the meaning indicated above and preferably denotes CmH2m+1 or O—CmH2m+1 or (CH2)Z—CH═CH2, and in which
- n and m, independently of one another, denote an integer in the range from 0 to 15, preferably in the range from 1 to 7 and particularly preferably 1 to 5, and
- z denotes 0, 1, 2, 3 or 4, preferably 0 or 2.
The preferred combinations of (R91 and R92) here are, in particular, (CnH2n+1 and CmH2m+1) and (CnH2n+1 and O—CmH2m+1), particularly preferably (CnH2n+1 and O—CmH2m+1).
In a preferred embodiment of the present invention, the medium comprises one or more dielectrically positive compounds of the formula V-1 having a dielectric anisotropy of greater than 3.
The liquid-crystalline media in accordance with the present invention preferably comprise 10% or less, preferably 5% or less, particularly preferably 2% or less, very particularly preferably 1% or less, and in particular absolutely no compound having only two or fewer five- and/or six-membered rings.
In a preferred embodiment of the present invention, the medium comprises one or more compounds of the formula VI.
In a further preferred embodiment of the present invention, the medium comprises one or more compounds of the formula VII.
The definitions of the abbreviations (acronyms) used for the compounds in the present application are indicated below in Table D or are evident from Tables A to C.
In a preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formulae IB-1 and/or IB-2 and/or IB-3 and/or IB-4.
The liquid-crystal medium, or component A of the liquid-crystal medium, preferably comprises one or more compounds selected from the compounds of the formulae IB-1a-1 to IB-1a-12, particularly preferably of the formula IB-1a-2, very particularly preferably one or more compounds of the formula IB-1a-2 and one or more compounds selected from the group of the compounds of the formula IB-1a-1 and formulae IB-1a-3 to IB-1a-12, and one or more compounds of the formulae IB-1b-1 to IB-1b-12 and/or IB-2 and/or IB-3 and/or IB-4.
In a further preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds selected from the group of the compounds of the formulae IB-1b-1 to IB-1b-12, particularly preferably selected from the group of the compounds of the formulae IB-1b-5 and/or IB-1b-7 and/or IB-1b-8 and/or IB-1b-9 and/or IB-1b-10, and one or more compounds selected from the group of the compounds of the formulae IB-1a-1 to IB-1a-12, preferably of the formula IB-1a-2, and/or one or more compounds of the formulae IB-2 and/or IB-3 and/or IB-4.
In a further preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB-2 and one or more compounds of the formula IB-1, preferably of the formula IB-1a, preferably of the formulae IB-1a-2, and/or IB-1b, and/or one or more compounds of the formulae IB-3 and/or IB-4.
In a further preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB-3 and one or more compounds of the formula IB-1, preferably of the formula IB-1a, preferably of the formula IB-1a-2, and/or IB-1b, and/or one or more compounds of the formulae IB-2 and/or IB-4.
In a further preferred embodiment of the present invention, the liquid-crystal medium, or component A of the liquid-crystal medium, comprises one or more compounds of the formula IB-4 and one or more compounds of the formula IB-1, preferably of the formula IB-1a, preferably of the formula IB-1a-2, and/or IB-1b, and/or one or more compounds of the formulae IB-2 and/or IB-3.
The liquid-crystalline media in accordance with the present invention preferably comprise, more preferably predominantly consist of, even more preferably essentially consist of and very particularly preferably completely consist of compounds selected from the group of the compounds of the formulae IA, IB, II, IV and V, preferably IA, IB, IIA, IIB and IV, or selected from the group of the compounds of the formulae IA, IB, IIIA, IIIB, IV and V, preferably IA, IB, MA, IIIB and IV.
In this application, comprise in connection with compositions means that the entity in question, i.e. the medium or the component, comprises the component or components or compound or compounds indicated, preferably in a total concentration of 10% or more and very preferably 20% or more.
In this connection, predominantly consist of means that the entity in question comprises 55% or more, preferably 60% or more and very preferably 70% or more, of the component or components or compound or compounds indicated.
In this connection, essentially consist of means that the entity in question comprises 80% or more, preferably 90% or more and very preferably 95% or more, of the component or components or compound or compounds indicated.
In this connection, completely consist of means that the entity in question comprises 98% or more, preferably 99% or more and very preferably 100.0% of the component or components or compound or compounds indicated.
Other mesogenic compounds which are not explicitly mentioned above can optionally and advantageously also be used in the media in accordance with the present invention. Such compounds are known to the person skilled in the art.
The liquid-crystal media in accordance with the present invention preferably have a clearing point of 90° C. or more, more preferably 100° C. or more, even more preferably 120° C. or more, particularly preferably 150° C. or more and very particularly preferably 170° C. or more.
The nematic phase of the media according to the invention preferably extends at least from 20° C. or less to 90° C. or more, preferably up to 100° C. or more, more preferably at least from 0° C. or less to 120° C. or more, very preferably at least from −10° C. or less to 140° C. or more and in particular at least from −20° C. or less to 150° C. or more.
The Δε of the liquid-crystal medium in accordance with the invention, at 1 kHz and 20° C., is preferably 1 or more, more preferably 2 or more and very preferably 3 or more.
The Δn of the liquid-crystal media in accordance with the present invention, at 589 nm (NaD) and 20° C., is preferably in the range from 0.200 or more to 0.90 or less, more preferably in the range from 0.250 or more to 0.90 or less, even more preferably in the range from 0.300 or more to 0.85 or less and very particularly preferably in the range from 0.350 or more to 0.800 or less.
In a preferred embodiment of the present application, the Δn of the liquid-crystal media in accordance with the present invention is preferably 0.40 or more, more preferably 0.45 or more.
In accordance with the present invention, the individual compounds of the formula IA in the liquid-crystal media are preferably used in a total concentration of 10% to 100%, more preferably 30% to 95%, even more preferably 40% to 90% and very preferably 50% to 90%, of the mixture as a whole.
In accordance with the present invention, the individual compounds of the formula IB in the liquid-crystal media are preferably used in a total concentration of 10% to 100%, more preferably 30% to 95%, even more preferably 40% to 90% and very preferably 50% to 90%, of the mixture as a whole.
In the embodiment of the present invention in which the liquid-crystal media comprise one or more compounds selected from the group of the compounds of the formulae IIA and IIB, the further compounds are preferably employed as follows.
The compounds selected from the group of the compounds of the formulae IIA and IIB are preferably used in a total concentration of 1% to 30%, more preferably 2% to 20%, even more preferably 3% to 18% and very preferably 4% to 16%, of the mixture as a whole.
The compounds of the formula IV are preferably used in a total concentration of 1% to 20%, more preferably 2% to 15%, even more preferably 3% to 12% and very preferably 5% to 10%, of the mixture as a whole.
The liquid-crystal media preferably comprise, more preferably predominantly consist of and very preferably completely consist of in total 70% to 100%, more preferably 80% to 100% and very preferably 90% to 100% and in particular 95% to 100%, of the compounds of the formulae IA, IB, IIA, IIB and IV to IX, preferably of the formulae I, IIA, IIB and IV.
In the embodiment of the present invention in which the liquid-crystal media comprise one or more compounds selected from the group of the compounds of the formulae IIIA and IIIB, the further compounds are preferably employed as follows.
The compounds selected from the group of the compounds of the formulae IIIA and IIIB are preferably used in a total concentration of 1% to 60%, more preferably 5% to 55%, even more preferably 7% to 50% and very preferably 10% to 45%, of the mixture as a whole.
If the liquid-crystal media comprise only one or more compounds of the formula IIIA, but no compounds of the formula IIIB, the compounds of the formula IIIA are preferably used in a total concentration of 10% to 60%, more preferably 20% to 55%, even more preferably 30% to 50% and very preferably 35% to 45%, of the mixture as a whole.
If the liquid-crystal media comprise only one or more compounds of the formula MB, but no compounds of the formula IIIA, the compounds of the formula IIIB are preferably used in a total concentration of 5% to 45%, more preferably 10% to 40%, even more preferably 15% to 35% and very preferably 20% to 30%, of the mixture as a whole.
If the liquid-crystal media comprise both one or more compounds of the formula IIIA and one or more compounds of the formula IIIB, the compounds of the formula IIIA are preferably used in a total concentration of 5% to 50%, more preferably 10% to 45%, even more preferably 15% to 30% and very preferably 20% to 25%, of the mixture as a whole and the compounds of the formula IIIB are preferably used in a total concentration of 1% to 35%, more preferably 5% to 30%, even more preferably 7% to 25% and very preferably 10% to 20%, of the mixture as a whole.
The compounds of the formula IV are preferably used in a total concentration of 1% to 20%, more preferably 2% to 15%, even more preferably 3% to 12% and very preferably 5% to 10%, of the mixture as a whole.
The liquid-crystal media preferably comprise, more preferably predominantly consist of and very preferably completely consist of in total 70% to 100%, more preferably 80% to 100% and very preferably 90% to 100% and in particular 95% to 100%, of compounds selected from the group of the compounds of the formulae IA, IB, IIIA, IIIB and IV to IX, preferably of the formulae IA, IB and/or IIIA and/or IIIB.
In a particularly preferred embodiment of the present invention, the liquid-crystalline media comprise one or more compounds of the formula V and one or more compounds of the formula VI.
In a further particularly preferred embodiment of the present invention, the liquid-crystalline media comprise one or more compounds of the formula V and one or more compounds of the formula VII.
The liquid-crystalline media in accordance with the present invention likewise preferably comprise one or more compounds of the formula V, one or more compounds of the formula VI and one or more compounds of the formula VIII.
If the liquid-crystalline media in accordance with the present application comprise one or more compounds of the formula V, the concentration of these compounds is preferably in total 10 to 30%, preferably 15 to 25% and particularly preferably 18 to 22%.
If the liquid-crystalline media in accordance with the present application comprise one or more compounds of the formula VI, the concentration of these compounds is preferably in total 15 to 35%, preferably 18 to 30% and particularly preferably 22 to 26%.
If the liquid-crystalline media in accordance with the present application comprise one or more compounds of the formula VII, the concentration of these compounds is preferably in total 4 to 25%, preferably 8 to 20% and particularly preferably 10 to 14%.
If the liquid-crystalline media in accordance with the present application comprise one or more compounds of the formula VIII, the concentration of these compounds is preferably in total 15 to 35%, preferably 18 to 30% and particularly preferably 22 to 26%.
If the liquid-crystalline media in accordance with the present application comprise one or more compounds of the formula IX, the concentration of these compounds is preferably in total 5 to 25%, preferably 10 to 20% and particularly preferably 13 to 17%.
In the present application, the expression dielectrically positive describes compounds or components where Δε>3.0, dielectrically neutral describes those where −1.5≦Δε≦3.0 and dielectrically negative describes those where Δε<−1.5. Δε is determined at a frequency of 1 kHz and at 20° C. The dielectric anisotropy of the respective compound is determined from the results of a solution of 10% of the respective individual compound in a nematic host mixture. If the solubility of the respective compound in the host mixture is less than 10%, the concentration is reduced to 5%. The capacitances of the test mixtures are determined both in a cell having homeotropic alignment and in a cell having homogeneous alignment. The cell thickness of both types of cells is approximately 20 μm. The voltage applied is a rectangular wave having a frequency of 1 kHz and an effective value of typically 0.5 V to 1.0 V, but it is always selected to be below the capacitive threshold of the respective test mixture.
The following definitions apply here.
Δε≡(ε∥−ε⊥) and
εaverage≡(ε∥+2ε⊥)/3.
The host mixture used for dielectrically positive compounds is mixture ZLI-4792 and that used for dielectrically neutral and dielectrically negative compounds is mixture ZLI-3086, both from Merck KGaA, Germany. The absolute values of the dielectric constants of the compounds are determined from the change in the respective values of the host mixture on addition of the compounds of interest. The values are extrapolated to a concentration of the compounds of interest of 100%.
Components having a nematic phase at the measurement temperature of 20° C. are measured as such, all others are treated like compounds.
The expression threshold voltage in the present application refers to the optical threshold and is quoted for 10% relative contrast (V10), and the expression saturation voltage refers to the optical saturation and is quoted for 90% relative contrast (V90), in both cases unless expressly stated otherwise. The capacitive threshold voltage (V0), also called the Freedericks threshold (VFr), is only used if expressly mentioned.
The parameter ranges indicated in this application all include the limit values, unless expressly stated otherwise.
The different upper and lower limit values indicated for various ranges of properties in combination with one another give rise to additional preferred ranges.
Throughout this application, the following conditions and definitions apply, unless expressly stated otherwise. All concentrations are quoted in percent by weight and relate to the respective mixture as a whole, all temperatures are quoted in degrees Celsius and all temperature differences are quoted in differential degrees. All physical properties are determined in accordance with “Merck Liquid Crystals, Physical Properties of Liquid Crystals”, Status November 1997, Merck KGaA, Germany, and are quoted for a temperature of 20° C., unless expressly stated otherwise. The optical anisotropy (Δn) is determined at a wavelength of 589.3 nm. The dielectric anisotropy (Δε) is determined at a frequency of 1 kHz. The threshold voltages, as well as all other electro-optical properties, are determined using test cells produced at Merck KGaA, Germany. The test cells for the determination of Δε have a cell thickness of approximately 20 μm. The electrode is a circular ITO electrode having an area of 1.13 cm2 and a guard ring. The orientation layers are SE-1211 from Nissan Chemicals, Japan, for homeotropic orientation (ε∥) and polyimide AL-1054 from Japan Synthetic Rubber, Japan, for homogeneous orientation (ε⊥). The capacitances are determined using a Solatron 1260 frequency response analyser using a sine wave with a voltage of 0.3 Vrms.
The light used in the electro-optical measurements is white light. A set-up using a commercially available DMS instrument from Autronic-Melchers, Germany, is used here. The characteristic voltages have been determined under perpendicular observation. The threshold (V10), mid-grey (V50) and saturation (V90) voltages have been determined for 10%, 50% and 90% relative contrast, respectively.
The liquid-crystalline media are investigated with respect to their properties in the microwave frequency region as described in A. Penirschke et al., “Cavity Perturbation Method for Characterisation of Liquid Crystals up to 35 GHz”, 34th European Microwave Conference—Amsterdam, pp. 545-548. Compare in this respect also A. Gaebler et al., “Direct Simulation of Material Permittivites . . . ”, 12MTC 2009—International Instrumentation and Measurement Technology Conference, Singapore, 2009 (IEEE), pp. 463-467, and DE 10 2004 029 429 A, in which a measurement method is likewise described in detail.
The liquid crystal is introduced into a cylindrical polytetrafluoroethylene (PTFE) or quartz capillary. The capillary has an internal radius of 180 μm and an external radius of 350 μm. The effective length is 2.0 cm. The filled capillary is introduced into the centre of the cylindrical cavity with a resonance frequency of 19 GHz. This cavity has a length of 11.5 mm and a radius of 6 mm. The input signal (source) is then applied, and the result of the output signal is recorded using a commercial vector network analyser. For other frequencies, the dimensions of the cavity are adapted correspondingly.
The change in the resonance frequency and the Q factor between the measurement with the capillary filled with the liquid crystal and the measurement without the capillary filled with the liquid crystal is used to determine the dielectric constant and the loss angle at the corresponding target frequency by means of equations 10 and 11 of the above-mentioned publication A. Penirschke et al., “Cavity Perturbation Method for Characterisation of Liquid Crystals up to 35 GHz”, 34th European Microwave Conference—Amsterdam, pp. 545-548, as described therein.
The values for the components of the properties perpendicular and parallel to the director of the liquid crystal are obtained by alignment of the liquid crystal in a magnetic field. To this end, the magnetic field of a permanent magnet is used. The strength of the magnetic field is 0.35 tesla. The alignment of the magnet is set correspondingly and then rotated correspondingly through 90°.
Preferred components are phase shifters, varactors, wireless and radio wave antenna arrays, matching circuit adaptive filters and others.
In the present application, the term compounds is taken to mean both one compound and a plurality of compounds, unless expressly stated otherwise.
The liquid-crystal media according to the invention preferably have nematic phases of in each case at least from −20° C. to 80° C., preferably from −30° C. to 85° C. and very particularly preferably from −40° C. to 100° C. The phase particularly preferably extends to 120° C. or more, preferably to 140° C. or more and very particularly preferably to 160° C. or more. The expression have a nematic phase here means on the one hand that no smectic phase and no crystallisation are observed at low temperatures at the corresponding temperature and on the other hand that no clearing occurs on heating from the nematic phase. The investigation at low temperatures is carried out in a flow viscometer at the corresponding temperature and checked by storage in test cells having a layer thickness of 5 μm for at least 100 hours. At high temperatures, the clearing point is measured in capillaries by conventional methods.
Furthermore, the liquid-crystal media according to the invention are characterised by high optical anisotropies in the visible region. The birefringence at 589 nm is preferably 0.20 or more, particularly preferably 0.25 or more, particularly preferably 0.30 or more, particularly preferably 0.40 or more and very particularly preferably 0.45 or more. In addition, the birefringence is preferably 0.80 or less.
In a preferred embodiment of the present invention, the liquid-crystal media employed have positive dielectric anisotropy ( ). This is preferably 1.8 or more and 15.0 or less, more preferably between 2.0 or more and 10.0 or less, particularly preferably between 3.0 or more and 8.0 or less and very particularly preferably between 3.5 or more and 6.0 or less.
If the liquid-crystal media employed have negative dielectric anisotropy ( ), this is preferably less than or equal to −2.5, particularly preferably less than or equal to −4.0 and very particularly preferably less than or equal to −5.0.
In this preferred embodiment of the present invention, in which the liquid-crystal media employed have negative dielectric anisotropy ( ), the value thereof is preferably between 1.5 or more and 15.0 or less, particularly preferably between 1.8 or more and 12.0 or less and very particularly preferably between 2.0 or more and 10.0 or less.
Furthermore, the liquid-crystal media according to the invention are characterised by high anisotropies in the microwave region and/or millimeter wave region. The birefringence is, for example, preferably 0.14 or more, particularly preferably 0.15 or more, particularly preferably 0.20 or more, particularly preferably 0.25 or more and very particularly preferably 0.30 or more, at about 8.3 GHz. In addition, the birefringence is preferably 0.80 or less.
The dielectric anisotropy in the microwave region is defined as
Δεr≡(εr,∥−εr,⊥).
The tuneability (τ) is defined as
τ≡(Δεr/εr,∥).
The material quality (η) is defined as
η≡(τ/tan δεr,max.), where
the maximum dielectric loss is
tan δεr,max.≡max. {tan δεr,⊥,;tan δεr,∥}.
The material quality (η) of the preferred liquid-crystal materials is 6 or more, preferably 8 or more, preferably 10 or more, preferably 15 or more, preferably 17 or more, preferably 20 or more, particularly preferably 25 or more, very particularly preferably 30 and in particular 40 or more or even 50 or more.
In the corresponding components, the preferred liquid-crystal materials have phase shifter qualities of 15°/dB or more, preferably 20°/dB or more, preferably 30°/dB or more, preferably 40°/dB or more, preferably 50°/dB or more, particularly preferably 80°/dB or more and very particularly preferably 100°/dB or more.
In some embodiments, however, it is also possible to use liquid crystals having a negative value of the dielectric anisotropy.
The liquid crystals employed are either individual substances or mixtures. They preferably have a nematic phase.
The term “alkyl” preferably encompasses straight-chain and branched alkyl groups, as well as cycloalkyl groups, each having 1 to 15 carbon atoms, in particular the straight-chain groups methyl, ethyl, propyl, butyl, pentyl, hexyl and heptyl, as well as cyclopropyl and cyclohexyl. Groups having 2 to 10 carbon atoms are generally preferred.
The term “alkenyl” preferably encompasses straight-chain and branched alkenyl groups having 2 to 15 carbon atoms, in particular the straight-chain groups. Particularly preferred alkenyl groups are C2- to C7-1E-alkenyl, C4- to C7-3E-alkenyl, C5- to C7-4-alkenyl, C6- to C7-5-alkenyl and C7-6-alkenyl, in particular C2- to C7-1E-alkenyl, C4- to C7-3E-alkenyl and C5- to C7-4-alkenyl. Examples of further preferred alkenyl groups are vinyl, 1E-propenyl, 1E-butenyl, 1E-pentenyl, 1E-hexenyl, 1E-heptenyl, 3-butenyl, 3E-pentenyl, 3E-hexenyl, 3E-heptenyl, 4-pentenyl, 4Z-hexenyl, 4E-hexenyl, 4Z-heptenyl, 5-hexenyl, 6-heptenyl and the like. Groups having up to 5 carbon atoms are generally preferred.
The term “fluoroalkyl” preferably encompasses straight-chain groups having a terminal fluorine, i.e. fluoromethyl, 2-fluoroethyl, 3-fluoropropyl, 4-fluorobutyl, 5-fluoropentyl, 6-fluorohexyl and 7-fluoroheptyl. However, other positions of the fluorine are not excluded.
The term “oxaalkyl” or “alkoxyalkyl” preferably encompasses straight-chain radicals of the formula CnH2n+1—O—(CH2)m, in which n and m each, independently of one another, denote an integer from 1 to 10. Preferably, n here is 1 and m is 1 to 6.
Compounds containing a vinyl end group and compounds containing a methyl end group have low rotational viscosity.
In the present application, both high-frequency technology and hyperfrequency technology denote applications having frequencies in the range from 1 MHz to 100 THz, preferably from 1 GHz to 30 THz, more preferably 2 GHz to 10 THz, particularly preferably from about 5 GHz to 5 THz.
The liquid-crystal media in accordance with the present invention may comprise further additives and chiral dopants in the usual concentrations. The total concentration of these further constituents is in the range from 0% to 10%, preferably 0.1% to 6%, based on the mixture as a whole. The concentrations of the individual compounds used are each preferably in the range from 0.1% to 3%. The concentration of these and similar additives is not taken into consideration when quoting the values and concentration ranges of the liquid-crystal components and liquid-crystal compounds of the liquid-crystal media in this application.
The liquid-crystal media according to the invention consist of a plurality of compounds, preferably 3 to 30, more preferably 4 to 20 and very preferably 4 to 15 compounds. These compounds are mixed in a conventional manner. In general, the desired amount of the compound used in the smaller amount is dissolved in the compound used in the larger amount. If the temperature is above the clearing point of the compound used in the higher concentration, it is particularly easy to observe completion of the dissolution process. It is, however, also possible to prepare the media in other conventional ways, for example using so-called pre-mixes, which can be, for example, homologous or eutectic mixtures of compounds, or using so-called “multibottle” systems, the constituents of which are themselves ready-to-use mixtures.
All temperatures, such as, for example, the melting point T(C,N) or T(C,S), the transition from the smectic (S) to the nematic (N) phase T(S,N) and the clearing point T(N,I) of the liquid crystals, are quoted in degrees Celsius. All temperature differences are quoted in differential degrees.
In the present invention and especially in the following examples, the structures of the mesogenic compounds are indicated by means of abbreviations, also referred to as acronyms. In these acronyms, the chemical formulae are abbreviated as follows using Tables A to C below. All groups CnH2n+1, CmH2m+1 and ClH2l+1 or CnH2n−1, CmH2m−1 and ClH2l−1 denote straight-chain alkyl or alkenyl, preferably 1E-alkenyl, having n, m and 1C atoms respectively, where n, m and 1, independently of one another, denote an integer from 1 to 9, preferably 1 to 7, or from 2 to 9, preferably 2 to 7, respectively. CoH2o+1 denotes straight-chain alkyl having 1 to 7, preferably 1 to 4, C atoms, or branched alkyl having 1 to 7, preferably 1 to 4, C atoms.
Table A lists the codes used for the ring elements of the core structures of the compounds, while Table B shows the linking groups. Table C gives the meanings of the codes for the left-hand or right-hand end groups. Table D shows illustrative structures of compounds with their respective abbreviations.
| |
C |
|
| |
|
| |
D |
|
| |
|
| |
A |
|
| |
|
| |
G |
|
| |
|
| |
U |
|
| |
|
| |
Y |
|
| |
|
| |
fX |
|
| |
|
| |
M |
|
| |
|
| |
N |
|
| |
|
| |
fN |
|
| |
|
| |
dH |
|
| |
|
| |
N(1, 4) |
|
| |
|
| |
N3f |
|
| |
|
| |
tH |
|
| |
|
| |
tH2f |
|
| |
|
| |
K |
|
| |
|
| |
L |
|
| |
|
| |
F |
|
| |
|
| |
P(o) |
|
| |
|
| |
P(i3) |
|
| |
|
| |
P(t4) |
|
| |
|
| |
P(c3) |
|
| |
|
| |
P(c4) |
|
| |
|
| |
P(c5) |
|
| |
|
| |
P(e5) |
|
| |
|
| |
P(c6) |
|
| |
|
| |
P(e6) |
|
| |
|
| |
P(o, 5F) |
|
| |
|
| |
GI(i3) |
|
| |
|
| |
GI(t4) |
|
| |
|
| |
GI(c3) |
|
| |
|
| |
GI(c4) |
|
| |
|
| |
GI(c5) |
|
| |
|
| |
GI(e5) |
|
| |
|
| |
GI(c6) |
|
| |
|
| |
GI(e6) |
|
| |
|
| |
P |
|
| |
|
| |
DI |
|
| |
|
| |
AI |
|
| |
|
| |
GI |
|
| |
|
| |
UI |
|
| |
|
| |
fXI |
|
| |
|
| |
MI |
|
| |
|
| |
NI |
|
| |
|
| |
fNI |
|
| |
|
| |
N(2, 6) |
|
| |
|
| |
N3fI |
|
| |
|
| |
tHI |
|
| |
|
| |
tH2fI |
|
| |
|
| |
KI |
|
| |
|
| |
LI |
|
| |
|
| |
FI |
|
| |
|
| |
PI(o) |
|
| |
|
| |
PI(c3) |
|
| |
|
| |
PI(t4) |
|
| |
|
| |
PI(c3) |
|
| |
|
| |
PI(c4) |
|
| |
|
| |
PI(c5) |
|
| |
|
| |
PI(e5) |
|
| |
|
| |
PI(c6) |
|
| |
|
| |
PI(e6) |
|
| |
|
| |
PI(o, 5F) |
|
| |
|
| |
G(i3) |
|
| |
|
| |
G(t4) |
|
| |
|
| |
G(c3) |
|
| |
|
| |
G(c4) |
|
| |
|
| |
G(c5) |
|
| |
|
| |
G(c5) |
|
| |
|
| |
G(c6) |
|
| |
|
| |
G(e6) |
|
| |
|
| |
E |
—CH2CH2— |
Z |
—CO—O— |
| |
V |
—CH═CH— |
ZI |
—O—CO— |
| |
X |
—CF═CH— |
O |
—CH2—O— |
| |
XI |
—CH═CF— |
OI |
—O—CH2— |
| |
B |
—CF═CF— |
Q |
—CF2—O— |
| |
T |
—C≡C— |
QI |
—O—CF2— |
| |
W |
—CF2CF2— |
| |
|
| Left-hand side | Right-hand side |
| |
| -n- | CnH2n+1— | -n | —CnH2n+1 |
| -nO- | CnH2n+1—O— | -nO | —O—CnH2n+1 |
| -V- | CH2═CH— | -V | —CH═CH2 |
| -nV- | CnH2n+1—CH═CH— | -nV | —CnH2n—CH═CH2 |
| -Vn- | CH2═CH—CnH2n+1— | -Vn | —CH═CH—CnH2n+1 |
| -nVm- | CnH2n+1—CH═CH—CmH2m— | -nVm | —CnH2n—CH═CH—CmH2m+1 |
| -N- | N≡C— | -N | —C≡N |
| -S- | S═C═N— | -S | —N═C═S |
| -F- | F— | -F | —F |
| -CL- | Cl— | -CL | —Cl |
| -M- | CFH2— | -M | —CFH2 |
| -D- | CF2H— | -D | —CF2H |
| -T- | CF3— | -T | —CF3 |
| -MO- | CFH2O— | -OM | —OCFH2 |
| -DO- | CF2HO— | -OD | —OCF2H |
| -TO- | CF3O— | -OT | —OCF3 |
| -OXF- | CF2═CH—O— | -OXF | —O—CH═CF2 |
| -A- | H—C≡C— | -A | —C≡C—H |
| -nA- | CnH2n+1—C≡C— | -An | —C≡C—CnH2n+1 |
| -NA- | N≡C—C≡C— | -AN | —C≡C—C≡N |
| - . . . A . . . - | —C≡C— | - . . . A . . . | —C≡C— |
| - . . . V . . . - | CH═CH— | - . . . V . . . | —CH═CH— |
| - . . . Z . . . - | —CO—O— | - . . . Z . . . | —CO—O— |
| - . . . ZI . . . - | —O—CO— | - . . . ZI . . . | —O—CO— |
| - . . . K . . . - | —CO— | - . . . K . . . | —CO— |
| - . . . W . . . - | —CF═CF— | - . . . W . . . | —CF═CF— |
| |
in which n and m each denote integers, and the three dots “.” are placeholders for other abbreviations from this table.
The following table shows illustrative structures together with their respective abbreviations. These are shown in order to illustrate the meaning of the rules for the abbreviations. They furthermore represent compounds which are preferably used.
| TABLE D |
| |
| Illustrative structures |
| The illustrative structures show compounds which are particularly preferably employed. |
| |
| Examples of compounds of component A |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
| Examples of compounds of component B |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
| Examples of compounds of component C |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
| Examples of compounds of component E |
| Compounds having three 6-membered rings |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
| Compounds having four 6-membered rings |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
| Illustrative structures of polar compounds employed: |
|
|
| |
|
|
| |
|
|
| |
| Illustrative structures of further neutral compounds employed: |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
| Illustrative structures of further polar compounds employed: |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
|
|
| |
The following table, Table E, shows illustrative compounds which can be used as stabiliser in the mesogenic media in accordance with the present invention. The total concentration of these and similar compounds in the media is preferably 5% or less.
In a preferred embodiment of the present invention, the mesogenic media comprise one or more compounds selected from the group of the compounds from Table E.
The following table, Table F, shows illustrative compounds which can preferably be used as chiral dopants in the mesogenic media in accordance with the present invention.
| TABLE F |
| |
|
|
C15 |
| |
|
|
CB 15 |
| |
|
|
CM 21 |
| |
|
|
CM 44 |
| |
|
|
CM 45 |
| |
|
|
CM 47 |
| |
|
|
CC |
| |
|
|
CN |
| |
|
|
R/S-811 |
| |
|
|
R/S-1011 |
| |
|
|
R/S-2011 |
| |
|
|
R/S-3011 |
| |
|
|
R/S-4011 |
| |
|
|
R/S-5011 |
| |
In a preferred embodiment of the present invention, the mesogenic media comprise one or more compounds selected from the group of the compounds from Table F.
The mesogenic media in accordance with the present application preferably comprise two or more, preferably four or more, compounds selected from the group consisting of the compounds from the above tables.
The liquid-crystal media in accordance with the present invention preferably comprise
-
- seven or more, preferably eight or more, compounds, preferably compounds having three or more, preferably four or more, different formulae, selected from the group of the compounds from Table D.
EXAMPLES
The following examples illustrate the present invention without limiting it in any way. However, it becomes clear to the person skilled in the art from the physical properties what properties can be achieved and in what ranges they can be modified. In particular, the combination of the various properties which can preferably be achieved is thus well defined for the person skilled in the art.
The acetylenes employed, if not commercially available, are synthesised in accordance with standard laboratory procedures.
Substance Examples A
Illustrative Compounds of the Formula IA for Component A
Synthesis Example 1a
1,4-bis(4-n-Butylphenylethynyl)-2,5-dimethylbenzene
9 g (54 mmol) of 4-n-butylphenylacetylene are initially introduced in 200 ml of tetrahydrofuran, the mixture is cooled to −75° C., and 54 ml (1.0 M in hexane, 54 mmol) of lithium hexamethyldisilazide are added. After 1 h, 54 ml (1.0 M in hexane, 54 mmol) of B-methoxy-9-borabicyclo[3.3.1]nonane are added, and the mixture is stirred for a further hour.
This solution is added dropwise to a solution of 1.5 g (2 mmol) of bis(tricyclohexylphosphine)palladium(II) chloride and 5.3 g (20 mmol) of 1,4-dibromo-2,5-dimethylbenzene in 300 ml of tetrahydrofuran, and the mixture is refluxed for 16 h.
The mixture is worked up by addition of MTB and water, the phases are separated, and the aqueous phase is extracted once with MTB. The combined organic phases are washed with saturated sodium chloride soln., dried over sodium sulfate, filtered and evaporated in a rotary evaporator. The residue is purified by column chromatography (SiO2, heptane/chlorobutane=1:1); the further purification is carried out by recrystallisation from heptane or heptane/chlorobutane=1:1.
MS (EI): m/e (%)=418 (100, M+), 375 (26, [M-propyl]+), 166 (9, [M-2x propyl]2+).
Phase sequence: C 125° C. N 142.8° C. I.
Data extrapolated from 10% solution in ZLI-4792:
Δε=+2.6; Δn=0.414 and γ1=2080 mPa·s.
Synthesis Example 2a
1,4-bis(4-n-Butylphenylethynyl)-2,6-dimethylbenzene
1,4-bis(4-n-Butylphenylethynyl)-2,6-dimethylbenzene is prepared from 4-n-butylphenylacetylene and 1-iodo-4-bromo-2,6-dimethylbenzene analogously to the synthesis described under 1A.
MS (EI): m/e (%)=418 (100, M+), 375 (27, [M-propyl]+), 166 (10, [M-2x propyl]2+).
Phase sequence: C 115° C. N 145.8° C. I.
Data extrapolated from 10% solution in ZLI-4792:
Δε=+2.4; Δn=0.409 and γ1=1940 mPa·s.
Synthesis Example 3a
1-(4-n-Propylphenylethynyl)-2-fluoro-4-(4-n-hexylphenylethynyl)-6-ethylbenzene
3a.1 Synthesis of rac-2-fluoro-6-(1-hydroxyethyl)-1,4-dibromobenzene
120 g (0.36 mol) of 6-fluoro-1,2,4-tribromobenzene in 120 ml of tetrahydrofuran are added dropwise to 290 ml (0.38 mol, 1.3 M solution in tetrahydrofuran) of isopropylmagnesium chloride/lithium chloride complex cooled to −70° C., and the mixture is stirred at −50° C. for 2 h. The mixture is subsequently cooled to −70° C., before 17.6 g (0.4 mol) of acetaldehyde in 40 ml of tetrahydrofuran are added dropwise. The batch is slowly warmed to −20° C. and then quenched by the addition of saturated ammonium chloride solution.
The phases are separated, and the aqueous phase is extracted with MTB. The combined organic phases are washed with saturated sodium chloride solution, dried over sodium sulfate and evaporated in a rotary evaporator. The residue obtained in this way is purified a number of times by column chromatography (SiO2, heptane→(gradient) heptane/MTB=4:1), with the polarity of the eluent being increased successively by the addition of MTB.
3a.2 Synthesis of 2-fluoro-6-ethyl-1,4-dibromobenzene
11 g (36.9 mmol) of the alcohol obtained in the preceding reaction are initially introduced in 25 ml of dichloromethane, 8.7 g (75.2 mmol) of triethylsilane are added, and the mixture is stirred at room temperature for 3 h. 10.4 g (72.9 mmol) of boron trifluoride diethyl etherate are subsequently added dropwise, and the mixture is stirred at a temperature just below the boiling point for 12 days. The batch, which is then concentrated virtually completely, is cooled, MTB and 2 M sodium hydroxide solution are added, and the mixture is stirred vigorously for 3 h. The phases are separated, and the aqueous phase is extracted twice with MTB. The combined organic phases are washed with saturated sodium chloride solution, dried over sodium sulfate and evaporated in a rotary evaporator. The residue obtained in this way is purified by column chromatography (SiO2, heptane).
3a.3 Synthesis of 1-bromo-2-fluoro-4-(4-n-hexylphenylethynyl)-6-ethylbenzene
3.4 g (12 mmol) of the dibromide obtained in the preceding reaction, 2.6 g (13.9 mmol) of 4-n-hexylphenylacetylene, 0.5 g (0.7 mmol) of bis(triphenylphosphine)palladium(II) chloride and 70 mg (0.35 mmol) of copper(I) iodide are initially introduced in 70 ml of diisopropylamine, and the mixture is stirred at 50° C. for 1 h and under reflux for 1 h. The batch is cooled, and the precipitate formed is filtered off. The filtrate is evaporated in a rotary evaporator, and the residue obtained in this way is purified by column chromatography (SiO2, heptane).
3a.4 Synthesis of 1-(4-n-propylphenylethynyl)-2-fluoro-4-(4-n-hexylphenylethynyl)-6-ethylbenzene
2 g (5.2 mmol) of the bromide obtained in the preceding reaction, 1.2 g (8.3 mmol) of 4-n-propylphenylacetylene, 0.2 g (0.28 mmol) of bis(triphenylphosphine)palladium(II) chloride and 30 mg (0.16 mmol) of copper(I) iodide are initially introduced in 30 ml of diisopropylamine, and the mixture is heated under reflux. After 1 h and after 12 h, a further 1.2 g of 4-n-propylphenylacetylene in a little diisopropylamine are added in each case, before the mixture is cooled after a total of 60 h, and the precipitate formed is filtered off. The filtrate is evaporated in a rotary evaporator, and the residue obtained in this way is purified by column chromatography (SiO2, heptane); the further purification is carried out by recrystallisation from heptane.
Phase sequence: C 33° C. N 90° C. I.
Data extrapolated from 10% solution in ZLI-4792:
Δε=+1.8; Δn=0.389 and γ1=1530 mPa·s.
Synthesis Example 4a
1-(4-n-Propylphenylethynyl)-2-fluoro-4-(4-n-hexylphenylethynyl)-6-ethylbenzene
4a.1 Synthesis of 2-(4-n-propylphenylethynyl)-4-fluoro-5-bromobenzene
20 g (63.5 mmol) of 5-bromo-4-fluoro-2-iodotoluene, 10.5 g (73 mmol) of 4-n-propylphenylacetylene, 2.5 g (3.6 mmol) of bis(triphenylphosphine)palladium-(II) chloride and 300 mg (1.8 mmol) of copper(I) iodide are initially introduced in 250 ml of diisopropylamine, the mixture is heated under reflux for 3 h and cooled, and the precipitate formed is filtered off. The filtrate is evaporated in a rotary evaporator, and the residue obtained in this way is purified by column chromatography (SiO2, heptane).
4a.2 Synthesis of 1-(4-n-propylphenylethynyl)-2-fluoro-4-(4-n-hexylphenylethynyl)-6-ethylbenzene
9.6 g (28.9 mmol) of the bromide obtained in the preceding reaction, 7 g (37.6 mmol) of 4-n-hexylphenylacetylene, 1.7 g (2.4 mmol) of bis(triphenylphosphine)palladium(II) chloride and 200 mg (1.2 mmol) of copper(I) iodide are initially introduced in 250 ml of diisopropylamine, the mixture is heated under reflux for 2 h and cooled, and the precipitate formed is filtered off. The filtrate is evaporated in a rotary evaporator, and the residue obtained in this way is purified by column chromatography (SiO2, heptane); the further purification is carried out by recrystallisation from heptane.
MS (EI): m/e (%)=436 (100, M+), 407 (8, [M-ethyl]+), 365 (39, [M-pentyl]+), 336 (12).
Phase sequence: C 50° C. N 148.7° C. I.
Data extrapolated from 10% solution in ZLI-4792:
Δε=+2.3; Δn=0.427 and γ1=1490 mPa·s.
Synthesis Example 5a
Synthesis of 1,4-bis-(4-n-butylphenylethynyl)-2,3,5,6-tetramethylbenzene
8.1 g (20 mmol) of 1,4-diiodo-2,3,5,6-tetramethylbenzene, 7.5 g (47.3 mmol) of 4-n-butylphenylacetylene, 2 g (2.9 mmol) of bis(triphenylphosphine)palladium(II) chloride and 550 mg (2.9 mmol) of copper(I) iodide are initially introduced in 100 ml of triethylamine, and the mixture is heated under reflux for 3 h. The batch is then cooled, water and MTB are added, and the phases are separated. The organic phase is washed with saturated ammonium chloride solution and with sodium chloride solution and evaporated in a rotary evaporator, and the residue obtained in this way is purified by column chromatography (SiO2, heptane); the further purification is carried out by recrystallisation from isopropanol.
MS (EI): m/e (%)=446 (100, M+), 421 (15, [M-propyl]+), 180 (8, [M-2x propyl]2+).
Phase sequence: C 67° C. N 225.1° C. I.
Data extrapolated from 10% solution in ZLI-4792:
Δε=+2.3; Δn=0.423 and γ1=1360 mPa·s.
Synthesis Example 6a
1,4-bis(4-n-Butylphenylethynyl)-2,3,5-trimethylbenzene
6a.1: Synthesis of 4-bromo-2,3,6-trimethylphenol
10 g (71 mmol) of 2,3,6-trimethylphenol are initially introduced in a solvent mixture consisting of 250 ml of dichloromethane and 190 ml of methanol, and 35.4 g (72 mmol) of tetrabutylammonium tribromide in 170 ml of dichloromethane and 130 ml of methanol are added. After 5 h at room temperature, the batch is evaporated in a rotary evaporator, and the residue remaining is taken up in MTB. The organic solution is washed three times with water, dried over sodium sulfate and evaporated in a rotary evaporator. The crystalline residue obtained is employed in the next step without further work-up.
6a.2: Synthesis of trifluoromethylsulfonyl-4-bromo-2,3,6-trimethylphenol
15.5 g (69 mmol) of the phenol obtained in the preceding reaction are initially introduced in 230 ml of dichloromethane, 14.7 ml (106 mmol) of triethylamine and 200 mg (1.6 mmol) of 4-(dimethylamino)pyridine are added, and the mixture is cooled in an ice bath. 12.2 ml (72.3 mmol) of trifluoromethanesulfonic anhydride are then added dropwise, and the mixture is stirred overnight and warmed to room temperature in the process, before the entire batch is purified directly by column chromatography (SiO2, dichloromethane).
6a.3: Synthesis of 1,4-bis-(4-n-butylphenylethynyl)-2,3,5-trimethylbenzene
9 g (54 mmol) of 4-n-butylphenylacetylene are initially introduced in 100 ml of tetrahydrofuran, the mixture is cooled to −78° C., and 54 ml (1.0 M in hexane, 54 mmol) of lithium hexamethyldisilazide are added. After 1 h, 54 ml (1.0 M in hexane, 54 mmol) of B-methoxy-9-borabicyclo[3.3.1]nonane are added, and the mixture is stirred for a further 2 h. The batch is then warmed to −20° C., 730 mg (0.8 mmol) of tris(dibenzylideneacetone)dipalladium(0), 1.3 g (3.2 mmol) of 2-dicyclohexylphosphino-2′,6′-dimethoxybiphenyl and 6.9 g (20 mmol) of the triflate obtained in the preceding reaction in 150 ml of tetrahydrofuran are added, and the mixture is heated under reflux for 16 h. After cooling, the mixture is worked up by addition of MTB and water, the phases are separated, and the aqueous phase is extracted once with MTB. The combined organic phases are washed with saturated sodium chloride solution, dried over sodium sulfate, filtered and evaporated in a rotary evaporator. The residue is purified by column chromatography (SiO2, heptane); the final purification is carried out by recrystallisation from heptane.
MS (EI): m/e (%)=432 (100, M+), 389 (20, [M-propyl]+), 173 (8, [M-2x propyl]2+).
Phase sequence: C 136° C. N 184.2° C. I.
Data extrapolated from 10% solution in ZLI-4792:
Δε=+2.4; Δn=0.412 and γ1=1650 mPa·s.
Substance Examples B
Illustrative Compounds of the Formula IB for Component A
Substance Example 1b
Phase sequence: C 87° C. N 178.5° C. I; Δn=0.436; Δε=2.8.
Substance Example 2b
Phase sequence: C 78° C. N 172.3° C. I; Δn=0.437; Δε=2.6.
Substance Example 3b
Phase sequence: C 107° C. N 211.0° C. I; Δn=0.464; Δε=3.1.
Substance Example 4b
Phase sequence: C 87° C. N 130.7° C. I; Δn=0.451; Δε=2.1.
Substance Example 5b
Phase sequence: C 57° C. N 151.7° C. I; Δn=0.445; Δε=2.0.
Substance Example 6b
Phase sequence: C 29° C. N 119.2° C. I; Δn=0.402; Δε=1.7.
Substance Example 7b
Phase sequence: Tg −54° C. C 14° C. N 119.2° C. I; Δn=0.393; Δε=1.8.
Substance Example 8b
Phase sequence: C 60° C. N 121.8° C. I; Δn=0.394; Δε=1.7.
Substance Example 9b
Phase sequence: C 81° C. N 160.7° C. I; Δn=0.432; Δε=3.2.
Substance Example 10b
Synthesis Example 10:1,4-Bis(2-(4-butylphenyl)ethynyl)-2-cyclopropylbenzene
10b.1) 1,4-Dichloro-2-cyclopropylbenzene 2
20 g (73 mmol) of 1,4-dichloro-2-iodobenzene, 9.4 g (110 mmol) of cyclopropylboronic acid, 32 g (147 mmol) of potassium phosphate, 421 mg (0.7 mmol) of bis(dibenzylideneacetone)palladium(0) (Pd(dba)2) and 1096 mg (1.5 mmol) of 1,2,3,4,5-pentaphenyl-1-(di-tert-butylphosphine)ferrocene (CTC-Q-PHOS) are dissolved in 600 ml of toluene and heated at 100° C. overnight. 100 ml of water are added to the cooled solution, and the mixture is extracted twice with toluene (100 ml). The combined organic phases are washed with water, dried over sodium sulfate and evaporated in vacuo. The residue is purified by column chromatography, giving the title compound as a colourless solid.
10b.2) 1,4-Bis(2-(4-butylphenyl)ethynyl)-2-cyclopropylbenzene (1)
5 g (26 mmol) of 1,4-dichloro-2-cyclopropylbenzene, 9.4 g (58 mmol) of 1-n-butyl-4-ethynylbenzene, 19 g (58 mmol) of caesium carbonate, 69 mg (0.3 mmol) of bis(acetonitrile)palladium(II) chloride and 382 mg (0.8 mmol) of 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl are dissolved in 80 ml of dioxane under nitrogen, and the mixture is heated at 100° C. overnight. 100 ml of water are added to the cooled solution, and the mixture is extracted twice with methyl t-butyl ether (100 ml). The combined organic phases are washed with water, dried over sodium sulfate and evaporated in vacuo. The residue is purified by column chromatography and recrystallised from ethanol, giving the title compound I as a solid.
1H-NMR (250 MHz, CDCl3): 7.4-7.08 (11H, m); 2.5 (4H, m); 1.6-1.3 (9H, m); 0.96 (6H, m); 0.6-0.4 (4H, m).
Phase sequence: C 72° C. N 84.5° C. I; Δn=0.378; Δε=1.5.
Substance Example 11b
Phase sequence: Tg −43° C. C 46° C. N 86.0° C. I; Δn=0.379; Δε=1.1.
Substance Example 12b
Synthesis Example 12b
1,4-Bis(2-(4-butylphenyl)ethynyl)-2-cyclobutylbenzene (3)
12b.1) 1-(2,5-Dibromophenyl)cyclobutanol (4)
21.09 g (67 mmol) of 1,2,4-tribromobenzene are initially introduced in 100 ml of THF under nitrogen, cooled to −45° C., and a solution (1.3 M) of 51.54 ml (67 mmol) of isopropylmagnesium chloride/lithium chloride complex in THF is added dropwise. After 1 hour, the batch is warmed to −10° C., and 5 ml (66.34 ml) of cyclobutanone are added dropwise at this temperature. The batch is allowed to thaw, and sat. NH4Cl solution is added, the mixture is extracted with methyl t-butyl ether, the organic phase is dried over sodium sulfate and filtered, the solvent is removed in vacuo, and the residue is filtered through silica gel with dichloromethane, giving 4, which is employed in the next step without further purification.
12b.2) 1,4-Dibromo-2-cyclobutylbenzene (5)
14.5 g (47.39 mmol) of 4 are dissolved in 50 ml of THF under nitrogen, and 35.72 ml (284.4 mmol) of boron trifluoride/diethyl ether complex are added dropwise at room temperature, and 12.54 g (189.6 mmol) of sodium cyanoborohydride are added dropwise in portions. The batch is heated under reflux overnight. The batch is allowed to cool to room temperature, sat. NaHCO3 solution is added, the mixture is extracted with methyl t-butyl ether, the organic phase is dried over sodium sulfate and filtered, the solvent is removed in vacuo, and the residue is filtered through silica gel with 1-chlorobutane, giving 5 as a yellow liquid.
12b.3) 1,4-Bis(2-(4-butylphenyl)ethynyl)-2-cyclobutylbenzene (3)
7.8 g (47.0 mmol) of 1-butyl-4-ethynylbenzene are initially introduced in 100 ml of THF under nitrogen, cooled to −78° C., and 63.32 ml (63.20 mmol) of a 1 M solution of lithium bis(trimethylsilyl)amide in hexane are added dropwise. After 1 hour, 63.22 ml (63.20 mmol) of a 1 M solution of 9-methoxy-9-BBN in hexane are added, and the mixture is left to stir at −78° C. for 2 hours. In a second apparatus, 6.8 g (23.45 mmol) of 5, 0.916 g (1.0 mmol) of tris(dibenzylideneacetone)dipalladium(0) and 1.64 g (4.0 mmol) of 2-dicyclohexylphosphino-2′,6′-dimethoxybiphenyl in 100 ml of THF are initially introduced. The first solution is slowly added dropwise, and the batch is heated at 100° C. overnight. 100 ml of water are added to the cooled solution, and the mixture is extracted twice with methyl t-butyl ether (100 ml). The combined organic phases are washed with water, dried over sodium sulfate and evaporated in vacuo. The residue is purified by column chromatography and recrystallised from isopropanol, giving the title compound 3 as a solid.
Phase sequence: Tg −39° C. C 69° C. N 70.1° C. I; Δn=0.359; Δε=0.9.
Substance Example 13b
Phase sequence: Tg −36° C. C 57° C. N(?) (7.0)° C. I; Δn=0.334; Δε=−0.1.
Substance Example 14b
Phase sequence: Tg −34° C. C 47° C. N 53.1° C. I; Δn=0.337; Δε=0.0.
Substance Example 15b
Synthesis Example 15b
2-Cyclohexyl-4-(4-hexylphenylethynyl)-1-(4-propylphenylethynyl)benzene 6
15b.1) 4-Chloro-2-cyclohexylbenzene trifluoromethanesulfonate (7)
19 g (90.2 mmol) of 4-chloro-2-cyclohexylbenzene are dissolved in 264 ml of dichloromethane, cooled to −5° C., and 4.64 ml (33.18 mmol) of triethylamine and 223 mg (1.8 mmol) of 4-(dimethylamino)pyridine are added dropwise. The batch is stirred at room temperature overnight and filtered through silica gel with dichloromethane, giving the product 7, which is employed in the next step without further purification.
15b.2) (4-Chloro-2-cyclohexylphenylethynyl)trimethylsilane (8)
21 g (61.3 mmol) of 7, 25.8 ml (183.8 mmol) of trimethylsilylacetylene, 2.15 g (3 mmol) of bis(triphenylphosphine)palladium(II) chloride and 21.2 ml (153.2 mmol) of triethylamine are dissolved in 60 ml of N,N-dimethylformamide under nitrogen, and the mixture is heated at 100° C. overnight. 100 ml of water are added to the cooled solution, and the mixture is extracted twice with methyl t-butyl ether (100 ml). The combined organic phases are washed with water, dried over sodium sulfate and evaporated in vacuo. The residue is purified by column chromatography, giving the product 8, which is employed in the next step without further purification.
15b.3) 4-Chloro-2-cyclohexyl-1-ethynylbenzene (9)
16.6 g (57.1 mmol) of 8 are dissolved in 154 ml of tetrahydrofuran, cooled to 0° C., and a 1 M solution of tetra-n-butylammonium fluoride (68.48 mmol) is added dropwise. The batch is stirred at room temperature overnight, water is added, the mixture is extracted with methyl t-butyl ether, the organic phase is dried over sodium sulfate and filtered, the solvent is removed in vacuo, and the residue is filtered through silica gel with heptane/toluene, giving the product 9, which is employed in the next step without further purification.
15b.4) 4-Chloro-2-cyclohexyl-1-p-tolylethynylbenzene (10)
6.6 g (30.17 mmol) of 9, 7.28 g (30.17 mmol) of 1-bromo-4-hexylbenzene, 21.63 g (66.39 mmol) of caesium carbonate, 78 mg (0.3 mmol) of bis(acetonitrile)palladium(II) chloride and 431 mg (0.9 mmol) of 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl are dissolved in 90 ml of dioxane under nitrogen and heated at 100° C. overnight. 100 ml of water are added to the cooled solution, and the mixture is extracted twice with methyl t-butyl ether (100 ml). The combined organic phases are washed with water, dried over sodium sulfate and evaporated in vacuo. The residue is purified by column chromatography.
15b.5) 2-Cyclohexyl-4-(4-hexylphenylethynyl)-1-(4-propylphenylethynyl)benzene (6)
4.5 g (11.87 mmol) of 10, 1.7 g (11.87 mmol) of 1-n-propyl-4-ethynylbenzene, 8.5 g (26.12 mmol) of caesium carbonate, 30 mg (0.1 mmol) of bis(acetonitrile)palladium(II) chloride and 170 mg (0.35 mmol) of 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl are dissolved in 35 ml of dioxane under nitrogen and heated at 100° C. overnight. 100 ml of water are added to the cooled solution, and the mixture is extracted twice with methyl t-butyl ether (100 ml). The combined organic phases are washed with water, dried over sodium sulfate and evaporated in vacuo. The residue is purified by column chromatography, giving the title compound 6 as a solid.
Phase sequence: Tg −23° C. I; Δn=0.294; Δε=−0.6.
Substance Example 16b
The compound is prepared in accordance with Scheme 10 and analogously to compounds already described in the present application.
Phase sequence: C 90° C. N 193.9° C. I; Δn=0.435; Δε=3.0.
Substance Example 17b
The compound is prepared in accordance with Scheme 10 and analogously to compounds already described in the present application.
Phase sequence: C 53° C. N 143.5° C. I; Δn=0.401; Δε=2.5.
Substance Example 18b
The compound is prepared in accordance with Scheme 10 and analogously to compounds already described in the present application.
Phase sequence: Tg −49 C 32° C. N 126.0° C. I; Δn=0.373; Δε=1.6.
Substance Example 19b
Substance Example 20b
Phase sequence: Tg −45° C. C 60° C. N 89.1° C. I; Δn=0.348; Δε=1.3.
Substance Example 21b
Phase sequence: Tg −32° C. C 66° C. N (44.1)° C. I; Δn=0.322; Δε=0.6.
Substance Example 22b
Phase sequence: Tg −37° C. C 52° C. N 78.2° C. I; Δn=0.339; Δε=0.9.
Synthesis Example 23b
Step 23b.1
25.4 g of 2-bromo-1,4-dichlorobenzene, 8.9 ml of iodoethane and 13.3 ml of DMPU are dissolved in 230 ml of THF, and a solution of 16.2 g of lithium tetramethylpiperidite in THF is added dropwise at −70° C. After a further 2 h at −70° C., the reaction mixture is allowed to warm to ambient temperature, and the batch is hydrolysed using water and subjected to extractive work-up.
The crude product is purified by fractional distillation.
B.p.: 73° C./0.1 bar. Colourless liquid.
Step 23b.2
12.5 ml of a 5% solution of methyllithium in diethyl ether are added to 2.4 g of anhydrous zinc bromide in 50 ml of THF at 25-40° C. 0.3 g of PdCl2-dppf, bis-(diphenylphosphinoferrocene)palladium dichloride, is then added, the mixture is heated to the boil, and 4.6 g of the product from step 1.1, dissolved in a little THF, are added dropwise. The reaction mixture is subsequently heated under reflux for 15 h. The batch is hydrolysed using water and subjected to extractive work-up. The crude product is purified by chromatography (pentane/silica gel). Colourless liquid.
Step 23b.3
2.4 g of 4-butylphenylacetylene are initially introduced in 30 ml of THF and cooled to −78° C. 14.3 ml of a 1 M solution of lithium bis(trimethylsilyl)amide in hexane are added dropwise to this solution and allowed to react at −78° C. for a further 1 h. 14.3 ml of a 1 M solution of methoxy-9-BBN are then added dropwise, and the mixture is stirred at −78° C. for a further 2 h. In a second apparatus, 1.0 g of the product from the last step, dissolved in 40 ml of THF, is initially introduced with the catalyst comprising 0.2 g of tris(dibenzylideneacetone)dipalladium and 0.35 g of 2-dicyclohexylphosphino-2′,6′-dimethoxybiphenyl, and the reaction solution from the first reaction is added at room temperature. The mixture is heated at the boil for 15 h. The batch is hydrolysed using water and subjected to extractive work-up. The crude product is purified by chromatography (pentane/silica gel). Recrystallisation from pentane gives the purified title product.
Phase sequence: C 45 N 1801.
Substance Example 24b
The compound is prepared analogously to Example 23b.
Phase sequence: C 118 N 222 I; Δn=0.435; Δε=2.6.
Substance Example 25b
The title compound is prepared analogously to Example 23b.
Phase sequence: C 41 N 161 I.
Substance Example 26b
Synthesis Example 26b
26b.1) Synthesis of 1-iodo-4-bromonaphthalene
100 g (350 mmol) of 1,4-dibromonaphthalene are initially introduced in 11 of THF, cooled to −70° C., and 235 ml of n-BuLi (1.6 M in hexane, 370 mmol) are added dropwise. After 1 h, 103 g (406 mmol) of I2 in 250 ml of THF are added dropwise, the mixture is stirred at −70° C. for a further 2 h, warmed to 0° C. and quenched by the addition of 50 ml (644 mmol) of aqueous NaHSO3 solution (w=39%). The phases are separated, and the aqueous phase is extracted once with MTB. The combined organic phases are washed with saturated sodium chloride soln., dried over sodium sulfate, filtered and evaporated in a rotary evaporator. The residue is purified by column chromatography (SiO2, heptane), and the further purification is carried out by recrystallisation from isopropanol, giving 1-iodo-4-bromonaphthalene as a yellow solid.
26b.2) Synthesis of 1-bromo-4-(4-n-propylphenylethynyl)naphthalene
15.3 g (43.6 mmol) of 1-iodo-4-bromonaphthalene and 7.25 g (5.3 mmol) of 4-n-propylphenylacetylene are initially introduced in 200 ml of NEt3, 170 mg (0.9 mmol) of copper(I) iodide and 600 mg (0.9 mmol) of bis(triphenylphosphine)palladium(II) chloride are added, and the mixture is refluxed for 30 minutes. The batch is cooled, water and heptane are added, and the phases are separated. The organic phase is washed with saturated sodium chloride soln., dried over sodium sulfate, filtered and evaporated in a rotary evaporator. The residue is purified by column chromatography (SiO2, heptane), and the further purification is carried out by recrystallisation from isopropanol.
26b.3) Synthesis of 1-(4-n-butylphenylethynyl)-4-(4-n-propylphenylethynyl)naphthalene
2.35 g (6.3 mmol) of 1-bromo-4-(4-n-propylphenylethynyl)naphthalene and 1.33 g (8.4 mmol) of 4-n-butylphenylacetylene are initially introduced in 40 ml of NEt3, 60 mg (0.3 mmol) of copper(I) iodide and 200 mg (0.3 mmol) of bis(triphenylphosphine)palladium(II) chloride are added, and the mixture is refluxed for 18 h.
The batch is cooled, water and heptane are added, and the phases are separated. The organic phase is washed with saturated ammonium chloride solution and subsequently with saturated sodium chloride soln., dried over sodium sulfate, filtered and evaporated in a rotary evaporator. The residue of compound (I) is purified by column chromatography (SiO2, heptane), and the further purification is carried out by recrystallisation from isopropanol.
MS (EI): m/e (%)=426 (100, M+), 397 (11, [M-ethyl]+), 383 (16, [M-propyl]+), 354 (18, [M-ethylpropyl]+), 177 (14, [M-ethylpropyl]2+).
Phase sequence: C 78 N 191 I; Δn=0.450; Δε=2.9.
Substance Example 27b
The compound is prepared analogously to Example 26b.
Phase sequence: C 108° C. N 194° C. I.
Substance Example 28b
The compound is prepared analogously to Example 26b.
Phase sequence: C 63° C. N 171.7° C. I; Δn=0.435; Δε=2.3.
Substance Example 29b
The compound is prepared analogously to Example 26b.
Phase sequence: C 76° C. N 176.2° C. I; Δn=0.427; Δε=2.4.
Substance Example 30b
The compound is prepared analogously to Example 26b.
Phase sequence: C 100° C. N 162° C. I.
Substance Example 31b
The compound is prepared analogously to Example 26b.
Phase sequence: C 61° C. N 139° C. I.
Substance Example 32b
This compound is prepared analogously to the preceding compounds in accordance with the right-hand reaction path of Scheme 15.
Phase sequence: C 85° C. N 202.4° C. I; Δn=0.446; Δε=2.0.
Substance Example 33b
The compound is prepared analogously to Example 32b.
Phase sequence: C 112° C. N 253.5° C. I; Δn=0.484; Δε=4.1.
Substance Example 34b
This compound is prepared analogously to the preceding compounds in accordance with the left-hand reaction path of Scheme 15.
Phase sequence: C 93° C. N 212.4° C. I; Δn=0.527; Δε=0.8.
USE EXAMPLES
Comparative Examples 1 to 3
A liquid-crystalline substance having the abbreviation PTP(2)TP-6-3 is prepared by the method of Hsu, C. S., Shyu, K. F., Chuang, Y. Y. and Wu, S.-T., Liq. Cryst., 27 (2), (2000), pp. 283-287, and investigated with respect to its physical properties, in particular in the microwave region. The compound has a nematic phase and a clearing point (T(N,I)) of 114.5° C. Further physical properties at 20° C. are: ne(589.3 nm)=1.8563; Δn(589.3 nm)=0.3250; ε∥(1 kHz)=3.40; Δε(1 kHz)=0.8 and γ1=2100 mPa·s. The compound is suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 1a |
| |
| Properties of the compound PTP(2)TP-6-3 at 30 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
3.22 |
2.44 |
0.242 |
0.0029 |
0.0064 |
37.9 |
| |
| TABLE 1b |
| |
| Properties of the compound PTP(2)TP-6-3 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
3.35 |
2.42 |
0.278 |
0.0029 |
0.0061 |
45.2 |
| |
In addition, the properties of the compound n-1-pentyl-4′-cyanobiphenyl (also called PP-5-N or CB 15) and the liquid-crystal mixture ZLI-4792 (product from Merck KGaA, Darmstadt, Germany) are investigated at 19 GHz. The results are summarised in Table 2.
| TABLE 2 |
| |
| Comparison of the properties of the |
| various examples at 19 GHz and 20° C. |
| |
|
|
|
|
tan δε |
|
| Example |
LC |
εr, | | |
εr, ⊥ |
τ |
r, Max.
|
η |
| |
| Comp. 1 |
P2-6-3* |
3.35 |
2.42 |
0.278 |
0.0061 |
45.2 |
| Comp. 2 |
PP-5-N$ |
3.06 |
2.66 |
0.131 |
0.0273 |
4.8 |
| Comp. 3 |
ZLI§ |
2.57 |
2.29 |
0.107 |
0.0126 |
8.5 |
| 1 |
M-1 |
2.65 |
2.30 |
0.130 |
0.0126 |
10.3 |
| 2 |
M-2 |
3.35 |
2.41 |
0.280 |
0.0066 |
42.2 |
| 3 |
M-3 |
2.65 |
2.30 |
0.133 |
0.0125 |
10.6 |
| 4 |
M-4 |
3.35 |
2.41 |
0.281 |
0.0064 |
43.6 |
| 5 |
M-5 |
3.34 |
2.41 |
0.280 |
0.0078 |
35.9 |
| 6 |
M-6 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| 7 |
M-7 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| 8 |
M-8 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| 9 |
M-9 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| |
| Notes: |
| *P2-6-3 PTP(2)TP-6-3, |
| LC liquid crystal, |
| $PP-5-N CB15, |
| §ZLI ZLI-4792 and |
| t.b.d. to be determined. |
Example 1
A liquid-crystal mixture M-1 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(2,5F)TP-6-3 |
10.0 |
| 2 |
CP-5-F |
9.0 |
| 3 |
CP-6-F |
7.2 |
| 4 |
CP-7-F |
5.4 |
| 5 |
CCP-2-OT |
7.2 |
| 6 |
CCP-3-OT |
10.8 |
| 7 |
CCP-4-OT |
6.3 |
| 8 |
CCP-5-OT |
9.9 |
| 9 |
CCEP-3-OT |
4.5 |
| 10 |
CCEP-5-OT |
4.5 |
| 11 |
CPG-3-F |
10.8 |
| 12 |
CPG-5-F |
9.0 |
| 13 |
CGPC-3-3 |
1.8 |
| 14 |
CGPC-5-3 |
1.8 |
| 15 |
CGPC-5-5 |
1.8 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 95° C. |
| |
Δn (20° C., 589.3 nm) = 0.126 |
| |
Δε (20° C., 1 kHz) = 4.9 |
| |
k11 (20° C.) = t.b.d. pN |
| |
k33/k11 (20° C.) = t.b.d. |
| |
V0 (20° C.) = t.b.d. V |
| |
γ1 (20° C.) = 169 mPa · s |
| |
|
| |
Notes: |
| |
t.b.d.: to be determined. |
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 3 |
| |
| Properties of mixture M-1 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
2.65 |
2.30 |
0.130 |
0.0049 |
0.0126 |
10.3 |
| |
Example 2
A liquid-crystal mixture M-2 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(2,5F)TP-6-3 |
10.0 |
| 2 |
PTP(2)TP-6-3 |
90.0 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 116° C. |
| |
|
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 4 |
| |
| Properties of mixture M-2 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
3.35 |
2.41 |
0.280 |
0.0028 |
0.0066 |
42.2 |
| |
Example 3
A liquid-crystal mixture M-3 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(1,6F)TP-6-3 |
10.0 |
| 2 |
CP-5-F |
9.0 |
| 3 |
CP-6-F |
7.2 |
| 4 |
CP-7-F |
5.4 |
| 5 |
CCP-2-OT |
7.2 |
| 6 |
CCP-3-OT |
10.8 |
| 7 |
CCP-4-OT |
6.3 |
| 8 |
CCP-5-OT |
9.9 |
| 9 |
CCEP-3-OT |
4.5 |
| 10 |
CCEP-5-OT |
4.5 |
| 11 |
CPG-3-F |
10.8 |
| 12 |
CPG-5-F |
9.0 |
| 13 |
CGPC-3-3 |
1.8 |
| 14 |
CGPC-5-3 |
1.8 |
| 15 |
CGPC-5-5 |
1.8 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 102° C. |
| |
Δn (20° C., 589.3 nm) = 0.129 |
| |
Δε (20° C., 1 kHz) = 4.9 |
| |
k11 (20° C.) = t.b.d. pN |
| |
k33/k11 (20° C.) = t.b.d. |
| |
V0 (20° C.) = t.b.d. V |
| |
γ1 (20° C.) = 168 mPa · s |
| |
|
| |
Notes: |
| |
t.b.d.: to be determined. |
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 5 |
| |
| Properties of mixture M-3 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
2.65 |
2.30 |
0.133 |
0.0045 |
0.0125 |
10.6 |
| |
Example 4
A liquid-crystal mixture M-4 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(1,6F)TP-6-3 |
10.0 |
| 2 |
PTP(2)TP-6-3 |
90.0 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 122° C. |
| |
|
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 6 |
| |
| Properties of mixture M-4 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
3.35 |
2.41 |
0.281 |
0.0025 |
0.0064 |
43.6 |
| |
Example 5
A liquid-crystal mixture M-5 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(1,6F)TP-6-3 |
15.0 |
| 2 |
PTP(2,5F)TP-6-3 |
20.0 |
| 3 |
PTP(2)TP-6-3 |
65.0 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 118° C. |
| |
|
This mixture is very highly suitable, with good low-temperature stability, for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 7 |
| |
| Properties of mixture M-5 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
3.34 |
2.41 |
0.280 |
0.0029 |
0.0078 |
35.9 |
| |
Example 6
A liquid-crystal mixture M-6 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(3 × 1)TP-4-4 |
10.0 |
| 2 |
PTP(2)TP-6-3 |
90.0 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 126° C. |
| |
|
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 8 |
| |
| Properties of mixture M-6 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| |
| Note: |
| t.b.d. to be determined. |
Example 7
A liquid-crystal mixture M-7 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(4 × 1)TP-4-4 |
10.0 |
| 2 |
PTP(2)TP-6-3 |
90.0 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 130° C. |
| |
|
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 9 |
| |
| Properties of mixture M-7 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| |
| Note: |
| t.b.d. to be determined. |
Example 8
A liquid-crystal mixture M-8 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(3 × 1)TP-4-4 |
10.0 |
| 2 |
CP-5-F |
9.0 |
| 3 |
CP-6-F |
7.2 |
| 4 |
CP-7-F |
5.4 |
| 5 |
CCP-2-OT |
7.2 |
| 6 |
CCP-3-OT |
10.8 |
| 7 |
CCP-4-OT |
6.3 |
| 8 |
CCP-5-OT |
9.9 |
| 9 |
CCEP-3-OT |
4.5 |
| 10 |
CCEP-5-OT |
4.5 |
| 11 |
CPG-3-F |
10.8 |
| 12 |
CPG-5-F |
9.0 |
| 13 |
CGPC-3-3 |
1.8 |
| 14 |
CGPC-5-3 |
1.8 |
| 15 |
CGPC-5-5 |
1.8 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 102° C. |
| |
Δn (20° C., 589.3 nm) = 0.128 |
| |
Δε (20° C., 1 kHz) = 4.9 |
| |
k11 (20° C.) = t.b.d. pN |
| |
k33/k11 (20° C.) = t.b.d. |
| |
V0 (20° C.) = t.b.d. V |
| |
γ1 (20° C.) = 170 mPa · s |
| |
|
| |
Notes: |
| |
t.b.d.: to be determined. |
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 10 |
| |
| Properties of mixture M-8 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| |
| Note: |
| t.b.d. to be determined. |
Example 9
A liquid-crystal mixture M-9 having the composition and properties as indicated in the following table is prepared.
| No. |
Abbreviation |
c/% |
| |
| 1 |
PTP(4 × 1)TP-4-4 |
5.0 |
| 2 |
CP-5-F |
9.5 |
| 3 |
CP-6-F |
7.6 |
| 4 |
CP-7-F |
5.7 |
| 5 |
CCP-2-OT |
7.6 |
| 6 |
CCP-3-OT |
11.4 |
| 7 |
CCP-4-OT |
6.7 |
| 8 |
CCP-5-OT |
10.5 |
| 9 |
CCEP-3-OT |
4.7 |
| 10 |
CCEP-5-OT |
4.7 |
| 11 |
CPG-3-F |
11.4 |
| 12 |
CPG-5-F |
9.5 |
| 13 |
CGPC-3-3 |
1.9 |
| 14 |
CGPC-5-3 |
1.9 |
| 15 |
CGPC-5-5 |
1.9 |
| Σ |
|
100.0 |
| |
| |
Physical properties |
| |
|
| |
T(N, I) = 99° C. |
| |
Δn (20° C., 589.3 nm) = 0.113 |
| |
Δε (20° C., 1 kHz) = 5.2 |
| |
k11 (20° C.) = t.b.d. pN |
| |
k33/k11 (20° C.) = t.b.d. |
| |
V0 (20° C.) = t.b.d. V |
| |
γ1 (20° C.) = 148 mPa · s |
| |
|
| |
Notes: |
| |
t.b.d.: to be determined. |
This mixture is very highly suitable for applications in the microwave region and/or millimeter wave region, in particular for phase shifters.
| TABLE 11 |
| |
| Properties of mixture M-9 at 19 GHz |
| T/° C. |
εr, | | |
εr, ⊥ |
τ |
tan δε, r, | | |
tan δε, r, ⊥ |
η |
| |
| 20 |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
t.b.d. |
| |
| Note: |
| t.b.d. to be determined. |