US9145393B2 - Arylpyridinone ITK inhibitors for treating inflammation and cancer - Google Patents
Arylpyridinone ITK inhibitors for treating inflammation and cancer Download PDFInfo
- Publication number
- US9145393B2 US9145393B2 US14/603,937 US201514603937A US9145393B2 US 9145393 B2 US9145393 B2 US 9145393B2 US 201514603937 A US201514603937 A US 201514603937A US 9145393 B2 US9145393 B2 US 9145393B2
- Authority
- US
- United States
- Prior art keywords
- alkyl
- chosen
- amino
- hydrogen
- aryl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 206010061218 Inflammation Diseases 0.000 title abstract description 10
- 230000004054 inflammatory process Effects 0.000 title abstract description 10
- 206010028980 Neoplasm Diseases 0.000 title description 14
- 239000003112 inhibitor Substances 0.000 title description 12
- 201000011510 cancer Diseases 0.000 title description 7
- 150000001875 compounds Chemical class 0.000 claims abstract description 178
- 239000000203 mixture Substances 0.000 claims abstract description 79
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 claims description 249
- 229910052739 hydrogen Inorganic materials 0.000 claims description 200
- 239000001257 hydrogen Substances 0.000 claims description 199
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 192
- -1 N(C1-4 alkyl)2 Chemical group 0.000 claims description 160
- 125000000217 alkyl group Chemical group 0.000 claims description 159
- 150000002431 hydrogen Chemical class 0.000 claims description 150
- 125000003118 aryl group Chemical group 0.000 claims description 145
- 125000001072 heteroaryl group Chemical group 0.000 claims description 122
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 66
- 125000000623 heterocyclic group Chemical group 0.000 claims description 63
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 62
- 125000006272 (C3-C7) cycloalkyl group Chemical group 0.000 claims description 52
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 52
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 42
- 125000001424 substituent group Chemical group 0.000 claims description 34
- 150000003839 salts Chemical class 0.000 claims description 29
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 28
- 125000003282 alkyl amino group Chemical group 0.000 claims description 26
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 24
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 12
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 10
- 125000004527 pyrimidin-4-yl group Chemical group N1=CN=C(C=C1)* 0.000 claims description 10
- 239000003981 vehicle Substances 0.000 claims description 8
- UWSYUNRAYRYBRA-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 UWSYUNRAYRYBRA-UHFFFAOYSA-N 0.000 claims description 7
- WEXITMTUUYNCBT-CQSZACIVSA-N 3-[[(3R)-1-(2-chloroacetyl)piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound ClCC(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 WEXITMTUUYNCBT-CQSZACIVSA-N 0.000 claims description 7
- LTTFOPMNGJDBOA-OAHLLOKOSA-N 3-[[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 LTTFOPMNGJDBOA-OAHLLOKOSA-N 0.000 claims description 7
- OUURRBKOPIQSKL-UHFFFAOYSA-N 3-[[1-(2-chloroacetyl)azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound ClCC(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 OUURRBKOPIQSKL-UHFFFAOYSA-N 0.000 claims description 6
- ZNSSRHBAMJMZCY-UHFFFAOYSA-N 5-(2-methylpyridin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)C ZNSSRHBAMJMZCY-UHFFFAOYSA-N 0.000 claims description 6
- STUWTBUADKIKHT-UHFFFAOYSA-N 5-(6-methoxypyrazin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O STUWTBUADKIKHT-UHFFFAOYSA-N 0.000 claims description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 5
- 239000012453 solvate Substances 0.000 claims description 5
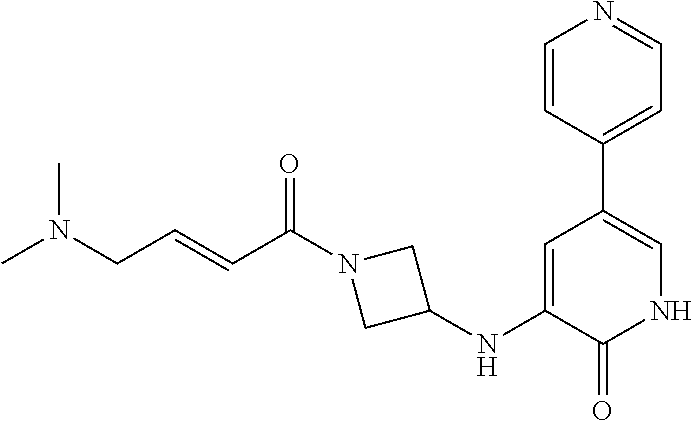
- WSLGIVXWEXLHCD-UHFFFAOYSA-N (E)-N-methyl-4-oxo-4-[3-[(2-oxo-5-pyridin-4-yl-1H-pyridin-3-yl)amino]azetidin-1-yl]but-2-enamide Chemical compound CNC(C=CC(N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1)=O)=O WSLGIVXWEXLHCD-UHFFFAOYSA-N 0.000 claims description 4
- IXOZTYJRHHKUGM-CQSZACIVSA-N 3-[[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 IXOZTYJRHHKUGM-CQSZACIVSA-N 0.000 claims description 4
- GOXVMQMYMPBCDW-NSCUHMNNSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 GOXVMQMYMPBCDW-NSCUHMNNSA-N 0.000 claims description 4
- ADZPNBKSYLSLGU-UHFFFAOYSA-N 3-[[1-[(E)-but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=CC)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 ADZPNBKSYLSLGU-UHFFFAOYSA-N 0.000 claims description 4
- QMNZBEKOLLPYIA-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC QMNZBEKOLLPYIA-UHFFFAOYSA-N 0.000 claims description 4
- HBFIHIOBIJCWFA-UHFFFAOYSA-N 5-[2-(methylamino)pyrimidin-4-yl]-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O HBFIHIOBIJCWFA-UHFFFAOYSA-N 0.000 claims description 4
- RYDTWQMHUCUNJY-UHFFFAOYSA-N 5-[6-(methylamino)pyrazin-2-yl]-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O RYDTWQMHUCUNJY-UHFFFAOYSA-N 0.000 claims description 4
- 239000002671 adjuvant Substances 0.000 claims description 4
- 239000003937 drug carrier Substances 0.000 claims description 3
- 125000004207 3-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(OC([H])([H])[H])=C1[H] 0.000 claims description 2
- 125000001475 halogen functional group Chemical group 0.000 claims 22
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 44
- 238000011282 treatment Methods 0.000 abstract description 26
- 238000000034 method Methods 0.000 abstract description 25
- 201000010099 disease Diseases 0.000 abstract description 22
- 230000000694 effects Effects 0.000 abstract description 17
- 241001465754 Metazoa Species 0.000 abstract description 15
- 230000005764 inhibitory process Effects 0.000 abstract description 13
- 230000001404 mediated effect Effects 0.000 abstract description 6
- 102100023345 Tyrosine-protein kinase ITK/TSK Human genes 0.000 abstract description 3
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 363
- 101150050425 CCC2 gene Proteins 0.000 description 58
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 51
- 239000000243 solution Substances 0.000 description 40
- 238000005160 1H NMR spectroscopy Methods 0.000 description 34
- 125000005843 halogen group Chemical group 0.000 description 33
- 239000002904 solvent Substances 0.000 description 31
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 30
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 27
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 27
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 26
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 25
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 25
- 239000003814 drug Substances 0.000 description 25
- 238000009472 formulation Methods 0.000 description 24
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 22
- 208000035475 disorder Diseases 0.000 description 22
- 239000004480 active ingredient Substances 0.000 description 21
- 239000007787 solid Substances 0.000 description 21
- 210000001744 T-lymphocyte Anatomy 0.000 description 19
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 18
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 17
- 229910052757 nitrogen Inorganic materials 0.000 description 17
- 239000000651 prodrug Substances 0.000 description 16
- 229940002612 prodrug Drugs 0.000 description 16
- 229940124597 therapeutic agent Drugs 0.000 description 16
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 15
- 125000004432 carbon atom Chemical group C* 0.000 description 15
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 14
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 14
- 108091000080 Phosphotransferase Proteins 0.000 description 14
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 14
- 0 [1*]C1=C([2*])NC(=O)C(N([5*])C2CCN([4*])C2)=C1 Chemical compound [1*]C1=C([2*])NC(=O)C(N([5*])C2CCN([4*])C2)=C1 0.000 description 14
- 102000020233 phosphotransferase Human genes 0.000 description 14
- 238000003556 assay Methods 0.000 description 13
- 210000004027 cell Anatomy 0.000 description 13
- 238000002347 injection Methods 0.000 description 13
- 239000007924 injection Substances 0.000 description 13
- 230000004913 activation Effects 0.000 description 12
- 230000027455 binding Effects 0.000 description 12
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 12
- 238000006243 chemical reaction Methods 0.000 description 12
- 108091008874 T cell receptors Proteins 0.000 description 11
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 11
- 125000005842 heteroatom Chemical group 0.000 description 11
- 125000000592 heterocycloalkyl group Chemical group 0.000 description 11
- 239000007788 liquid Substances 0.000 description 11
- 239000002609 medium Substances 0.000 description 11
- UBQKCCHYAOITMY-UHFFFAOYSA-N pyridin-2-ol Chemical group OC1=CC=CC=N1 UBQKCCHYAOITMY-UHFFFAOYSA-N 0.000 description 11
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 10
- 241000699670 Mus sp. Species 0.000 description 10
- 239000002585 base Substances 0.000 description 10
- 238000001816 cooling Methods 0.000 description 10
- 230000014509 gene expression Effects 0.000 description 10
- 238000002360 preparation method Methods 0.000 description 10
- 238000000746 purification Methods 0.000 description 10
- 230000004044 response Effects 0.000 description 10
- 206010039083 rhinitis Diseases 0.000 description 10
- 229910052717 sulfur Inorganic materials 0.000 description 10
- 239000000725 suspension Substances 0.000 description 10
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 9
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 9
- 239000002253 acid Substances 0.000 description 9
- 230000008901 benefit Effects 0.000 description 9
- 230000015572 biosynthetic process Effects 0.000 description 9
- 239000000843 powder Substances 0.000 description 9
- 230000001225 therapeutic effect Effects 0.000 description 9
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 8
- 125000003342 alkenyl group Chemical group 0.000 description 8
- 239000004615 ingredient Substances 0.000 description 8
- 229910052760 oxygen Inorganic materials 0.000 description 8
- 239000008194 pharmaceutical composition Substances 0.000 description 8
- 230000002265 prevention Effects 0.000 description 8
- 239000003826 tablet Substances 0.000 description 8
- 102000000588 Interleukin-2 Human genes 0.000 description 7
- 108010002350 Interleukin-2 Proteins 0.000 description 7
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 7
- 208000006673 asthma Diseases 0.000 description 7
- 125000004429 atom Chemical group 0.000 description 7
- 239000002775 capsule Substances 0.000 description 7
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 7
- 235000010418 carrageenan Nutrition 0.000 description 7
- 239000000679 carrageenan Substances 0.000 description 7
- 229920001525 carrageenan Polymers 0.000 description 7
- 229940113118 carrageenan Drugs 0.000 description 7
- 239000003921 oil Substances 0.000 description 7
- 239000011541 reaction mixture Substances 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 7
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 6
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 102100039079 Tyrosine-protein kinase TXK Human genes 0.000 description 6
- 125000000304 alkynyl group Chemical group 0.000 description 6
- 208000010668 atopic eczema Diseases 0.000 description 6
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 6
- 239000003054 catalyst Substances 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 230000001419 dependent effect Effects 0.000 description 6
- 229940079593 drug Drugs 0.000 description 6
- 150000002148 esters Chemical class 0.000 description 6
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 6
- 229910052736 halogen Inorganic materials 0.000 description 6
- 125000004404 heteroalkyl group Chemical group 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- 235000019198 oils Nutrition 0.000 description 6
- 239000012044 organic layer Substances 0.000 description 6
- 230000002829 reductive effect Effects 0.000 description 6
- FVAUCKIRQBBSSJ-UHFFFAOYSA-M sodium iodide Chemical compound [Na+].[I-] FVAUCKIRQBBSSJ-UHFFFAOYSA-M 0.000 description 6
- 230000000638 stimulation Effects 0.000 description 6
- VOHWGVQPKVXAGF-UHFFFAOYSA-N 4-[5-[[1-(2-chloroacetyl)azetidin-3-yl]amino]-6-oxo-1H-pyridin-3-yl]-N-(3-methyl-4-propan-2-ylphenyl)pyridine-2-carboxamide formic acid Chemical compound C(=O)O.ClCC(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)C(=O)NC1=CC(=C(C=C1)C(C)C)C VOHWGVQPKVXAGF-UHFFFAOYSA-N 0.000 description 5
- 208000023275 Autoimmune disease Diseases 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 5
- 108090000790 Enzymes Proteins 0.000 description 5
- 206010015150 Erythema Diseases 0.000 description 5
- 239000007821 HATU Substances 0.000 description 5
- HFBMWMNUJJDEQZ-UHFFFAOYSA-N acryloyl chloride Chemical compound ClC(=O)C=C HFBMWMNUJJDEQZ-UHFFFAOYSA-N 0.000 description 5
- 125000003545 alkoxy group Chemical group 0.000 description 5
- 230000000172 allergic effect Effects 0.000 description 5
- 150000001408 amides Chemical class 0.000 description 5
- 125000003277 amino group Chemical group 0.000 description 5
- 239000011324 bead Substances 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- 238000004587 chromatography analysis Methods 0.000 description 5
- 230000008878 coupling Effects 0.000 description 5
- 238000010168 coupling process Methods 0.000 description 5
- 238000005859 coupling reaction Methods 0.000 description 5
- 125000004122 cyclic group Chemical group 0.000 description 5
- 239000000839 emulsion Substances 0.000 description 5
- 229940088598 enzyme Drugs 0.000 description 5
- 238000001704 evaporation Methods 0.000 description 5
- 230000008020 evaporation Effects 0.000 description 5
- 238000001914 filtration Methods 0.000 description 5
- 235000011187 glycerol Nutrition 0.000 description 5
- 230000036039 immunity Effects 0.000 description 5
- 208000027866 inflammatory disease Diseases 0.000 description 5
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 5
- 229910052763 palladium Inorganic materials 0.000 description 5
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 5
- 108090000623 proteins and genes Proteins 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 239000000377 silicon dioxide Substances 0.000 description 5
- 210000003491 skin Anatomy 0.000 description 5
- 229910052938 sodium sulfate Inorganic materials 0.000 description 5
- 239000004094 surface-active agent Substances 0.000 description 5
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 5
- LAXUVIBOVLZWAN-UHFFFAOYSA-N 3-(azetidin-3-ylamino)-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one dihydrobromide Chemical compound Br.Br.N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC LAXUVIBOVLZWAN-UHFFFAOYSA-N 0.000 description 4
- VOSGKCNMKQSYIR-UHFFFAOYSA-N 4-(5-amino-6-oxo-1H-pyridin-3-yl)-N-(3-methyl-4-propan-2-ylphenyl)pyridine-2-carboxamide Chemical compound NC1=CC(=CNC1=O)C1=CC(=NC=C1)C(=O)NC1=CC(=C(C=C1)C(C)C)C VOSGKCNMKQSYIR-UHFFFAOYSA-N 0.000 description 4
- JPKZQFJUMQMFJG-UHFFFAOYSA-N 4-bromo-N-(3-methyl-4-propan-2-ylphenyl)pyridine-2-carboxamide Chemical compound BrC1=CC(=NC=C1)C(=O)NC1=CC(=C(C=C1)C(C)C)C JPKZQFJUMQMFJG-UHFFFAOYSA-N 0.000 description 4
- YDEHOTDIPRNFOY-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-3-phenylprop-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=CC1=CC=CC=C1)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC YDEHOTDIPRNFOY-UHFFFAOYSA-N 0.000 description 4
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 4
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical compound CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 4
- 108010036949 Cyclosporine Proteins 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 4
- 108010010803 Gelatin Proteins 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- 101150025881 Itk gene Proteins 0.000 description 4
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 4
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 4
- NLDLXRVWKDUGHB-UHFFFAOYSA-N N-(3-methyl-4-propan-2-ylphenyl)-4-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]pyridine-2-carboxamide hydrochloride Chemical compound Cl.C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)C(=O)NC1=CC(=C(C=C1)C(C)C)C NLDLXRVWKDUGHB-UHFFFAOYSA-N 0.000 description 4
- 102000014400 SH2 domains Human genes 0.000 description 4
- 108050003452 SH2 domains Proteins 0.000 description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 4
- 125000002252 acyl group Chemical group 0.000 description 4
- 208000026935 allergic disease Diseases 0.000 description 4
- 125000003368 amide group Chemical group 0.000 description 4
- 150000001412 amines Chemical class 0.000 description 4
- 206010003246 arthritis Diseases 0.000 description 4
- KCMGQCMYPMYOFI-UHFFFAOYSA-N benzyl N-[2-methoxy-5-[2-[(3-methyl-4-propan-2-ylphenyl)carbamoyl]pyridin-4-yl]pyridin-3-yl]carbamate Chemical compound C(C)(C)C1=C(C=C(C=C1)NC(=O)C1=NC=CC(=C1)C=1C=NC(=C(C1)NC(OCC1=CC=CC=C1)=O)OC)C KCMGQCMYPMYOFI-UHFFFAOYSA-N 0.000 description 4
- QRQLTCOSUNNGGZ-UHFFFAOYSA-N benzyl N-[5-[2-[(3-methyl-4-propan-2-ylphenyl)carbamoyl]pyridin-4-yl]-2-oxo-1H-pyridin-3-yl]carbamate Chemical compound C(C)(C)C1=C(C=C(C=C1)NC(=O)C1=NC=CC(=C1)C1=CNC(C(=C1)NC(OCC1=CC=CC=C1)=O)=O)C QRQLTCOSUNNGGZ-UHFFFAOYSA-N 0.000 description 4
- MAJFINHRLBJIQL-UHFFFAOYSA-N benzyl n-(2-oxo-1h-pyridin-3-yl)carbamate Chemical compound OC1=NC=CC=C1NC(=O)OCC1=CC=CC=C1 MAJFINHRLBJIQL-UHFFFAOYSA-N 0.000 description 4
- OFPAIGHAIRBKBF-UHFFFAOYSA-N benzyl n-(5-iodo-2-methoxypyridin-3-yl)carbamate Chemical compound COC1=NC=C(I)C=C1NC(=O)OCC1=CC=CC=C1 OFPAIGHAIRBKBF-UHFFFAOYSA-N 0.000 description 4
- VHYSSNYCKHBKPQ-UHFFFAOYSA-N benzyl n-(5-iodo-2-oxo-1h-pyridin-3-yl)carbamate Chemical compound IC1=CNC(=O)C(NC(=O)OCC=2C=CC=CC=2)=C1 VHYSSNYCKHBKPQ-UHFFFAOYSA-N 0.000 description 4
- LWCGCKUDTSNOON-UHFFFAOYSA-N benzyl n-[2-methoxy-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-3-yl]carbamate Chemical compound COC1=NC=C(B2OC(C)(C)C(C)(C)O2)C=C1NC(=O)OCC1=CC=CC=C1 LWCGCKUDTSNOON-UHFFFAOYSA-N 0.000 description 4
- 125000002619 bicyclic group Chemical group 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 4
- 238000002648 combination therapy Methods 0.000 description 4
- 239000002552 dosage form Substances 0.000 description 4
- 238000011156 evaluation Methods 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 239000008273 gelatin Substances 0.000 description 4
- 229920000159 gelatin Polymers 0.000 description 4
- 235000019322 gelatine Nutrition 0.000 description 4
- 235000011852 gelatine desserts Nutrition 0.000 description 4
- 230000002068 genetic effect Effects 0.000 description 4
- 125000001188 haloalkyl group Chemical group 0.000 description 4
- 238000001727 in vivo Methods 0.000 description 4
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 description 4
- 239000006210 lotion Substances 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 235000018102 proteins Nutrition 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 230000009467 reduction Effects 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 206010039073 rheumatoid arthritis Diseases 0.000 description 4
- 230000011664 signaling Effects 0.000 description 4
- LKZMBDSASOBTPN-UHFFFAOYSA-L silver carbonate Substances [Ag].[O-]C([O-])=O LKZMBDSASOBTPN-UHFFFAOYSA-L 0.000 description 4
- 239000003381 stabilizer Substances 0.000 description 4
- 239000011593 sulfur Substances 0.000 description 4
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 4
- MWNYKMHOEKPURP-QGZVFWFLSA-N tert-butyl (3R)-3-[(2-methoxy-5-pyridin-4-ylpyridin-3-yl)amino]piperidine-1-carboxylate Chemical compound COC1=C(C=C(C=N1)C1=CC=NC=C1)N[C@H]1CN(CCC1)C(=O)OC(C)(C)C MWNYKMHOEKPURP-QGZVFWFLSA-N 0.000 description 4
- APTGVJLQDCAXKC-UHFFFAOYSA-N tert-butyl 3-[(5-chloro-2-methoxypyridin-3-yl)amino]azetidine-1-carboxylate Chemical compound ClC=1C=C(C(=NC1)OC)NC1CN(C1)C(=O)OC(C)(C)C APTGVJLQDCAXKC-UHFFFAOYSA-N 0.000 description 4
- ZQQQWBPRPMJLMK-UHFFFAOYSA-N tert-butyl 3-[[2-methoxy-5-[2-(methylamino)pyridin-4-yl]pyridin-3-yl]amino]azetidine-1-carboxylate Chemical compound COC1=C(C=C(C=N1)C1=CC(=NC=C1)NC)NC1CN(C1)C(=O)OC(C)(C)C ZQQQWBPRPMJLMK-UHFFFAOYSA-N 0.000 description 4
- MKILZSFUQVAOAU-UHFFFAOYSA-N tert-butyl 3-[[5-(2-fluoropyridin-4-yl)-2-methoxypyridin-3-yl]amino]azetidine-1-carboxylate Chemical compound FC1=NC=CC(=C1)C=1C=NC(=C(C1)NC1CN(C1)C(=O)OC(C)(C)C)OC MKILZSFUQVAOAU-UHFFFAOYSA-N 0.000 description 4
- WPGLRFGDZJSQGI-UHFFFAOYSA-N tert-butyl 3-aminoazetidine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CC(N)C1 WPGLRFGDZJSQGI-UHFFFAOYSA-N 0.000 description 4
- 230000000699 topical effect Effects 0.000 description 4
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 3
- ZXSQEZNORDWBGZ-UHFFFAOYSA-N 1,3-dihydropyrrolo[2,3-b]pyridin-2-one Chemical compound C1=CN=C2NC(=O)CC2=C1 ZXSQEZNORDWBGZ-UHFFFAOYSA-N 0.000 description 3
- 102100026205 1-phosphatidylinositol 4,5-bisphosphate phosphodiesterase gamma-1 Human genes 0.000 description 3
- MBMQYTQCUCQJEH-UHFFFAOYSA-N 3,5-dibromo-2-methoxypyridine Chemical compound COC1=NC=C(Br)C=C1Br MBMQYTQCUCQJEH-UHFFFAOYSA-N 0.000 description 3
- OAJJTPCMFQBFNR-GZNAHNRGSA-N 3-[[(3R)-1-[(E)-4-piperidin-1-ylbut-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound N1(CCCCC1)C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 OAJJTPCMFQBFNR-GZNAHNRGSA-N 0.000 description 3
- GOWZHXWQCCDYPN-BTQNPOSSSA-N 3-[[(3R)-piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one hydrochloride Chemical compound Cl.N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 GOWZHXWQCCDYPN-BTQNPOSSSA-N 0.000 description 3
- JJGTUTRGRHOGGG-IDOMTICXSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1)C JJGTUTRGRHOGGG-IDOMTICXSA-N 0.000 description 3
- SNLMOARMGQTLGV-PXPSOEEZSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC SNLMOARMGQTLGV-PXPSOEEZSA-N 0.000 description 3
- FHBVMHKDCJMTCD-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C FHBVMHKDCJMTCD-SNAWJCMRSA-N 0.000 description 3
- KRXUIKWCPJYVPQ-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)CC1CC1 KRXUIKWCPJYVPQ-OWOJBTEDSA-N 0.000 description 3
- GUEUJZBEGLMPJB-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 GUEUJZBEGLMPJB-OWOJBTEDSA-N 0.000 description 3
- BHQMXVYIFZRBFZ-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC BHQMXVYIFZRBFZ-ONEGZZNKSA-N 0.000 description 3
- UVVZXUULTGSKGA-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 UVVZXUULTGSKGA-OWOJBTEDSA-N 0.000 description 3
- WKZWWBXQEBQGFH-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)C1CC1 WKZWWBXQEBQGFH-UHFFFAOYSA-N 0.000 description 3
- UKQGOUKQZPNQPS-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C1CC1 UKQGOUKQZPNQPS-UHFFFAOYSA-N 0.000 description 3
- XZHXVQHBBVCDEG-SFHVURJKSA-N 3-[cyclopropylmethyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 XZHXVQHBBVCDEG-SFHVURJKSA-N 0.000 description 3
- SDGLRPMVJXYGDI-KRWDZBQOSA-N 3-[ethyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC SDGLRPMVJXYGDI-KRWDZBQOSA-N 0.000 description 3
- VOKFQYANNSXTMS-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)C)C VOKFQYANNSXTMS-UHFFFAOYSA-N 0.000 description 3
- YPDQGWVDGHKQGG-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)C)=O)C YPDQGWVDGHKQGG-UHFFFAOYSA-N 0.000 description 3
- QJCRAZMUZCQPJC-UHFFFAOYSA-N 4-[5-(azetidin-3-ylamino)-6-oxo-1H-pyridin-3-yl]-N-(3-methyl-4-propan-2-ylphenyl)pyridine-2-carboxamide hydrochloride Chemical compound Cl.N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)C(=O)NC1=CC(=C(C=C1)C(C)C)C QJCRAZMUZCQPJC-UHFFFAOYSA-N 0.000 description 3
- JZGRYSRJEJPIMG-UHFFFAOYSA-N 5-(6-methoxypyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O JZGRYSRJEJPIMG-UHFFFAOYSA-N 0.000 description 3
- ANCVJJXYERNAPI-UHFFFAOYSA-N 5-(6-methylpyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)C)=O)CCC ANCVJJXYERNAPI-UHFFFAOYSA-N 0.000 description 3
- IQKANOKWBJSVLF-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-phenylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCC1=CC=CC=C1)=O)=O IQKANOKWBJSVLF-UHFFFAOYSA-N 0.000 description 3
- CMAPGPNUEUJGKA-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-5-phenoxypent-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCCOC1=CC=CC=C1)=O)=O CMAPGPNUEUJGKA-UHFFFAOYSA-N 0.000 description 3
- LZZBCUQBUAKHKP-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-pent-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCC)=O)=O LZZBCUQBUAKHKP-UHFFFAOYSA-N 0.000 description 3
- XADICJHFELMBGX-UHFFFAOYSA-N 5-bromo-2-methoxypyridine Chemical compound COC1=CC=C(Br)C=N1 XADICJHFELMBGX-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 108010029445 Agammaglobulinaemia Tyrosine Kinase Proteins 0.000 description 3
- 206010002556 Ankylosing Spondylitis Diseases 0.000 description 3
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- FCTMWXACQBXRBH-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=NC=C3)=CNC2=O)C1.Cl Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=NC=C3)=CNC2=O)C1.Cl FCTMWXACQBXRBH-UHFFFAOYSA-N 0.000 description 3
- BBLJCONFMSGMSP-CMDGGOBGSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/C4=CC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/C4=CC=CC=C4)C3)=C2)=C1 BBLJCONFMSGMSP-CMDGGOBGSA-N 0.000 description 3
- 210000001266 CD8-positive T-lymphocyte Anatomy 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 3
- VGCXGMAHQTYDJK-UHFFFAOYSA-N Chloroacetyl chloride Chemical compound ClCC(Cl)=O VGCXGMAHQTYDJK-UHFFFAOYSA-N 0.000 description 3
- 201000004624 Dermatitis Diseases 0.000 description 3
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 3
- 101000691599 Homo sapiens 1-phosphatidylinositol 4,5-bisphosphate phosphodiesterase gamma-1 Proteins 0.000 description 3
- 101000914514 Homo sapiens T-cell-specific surface glycoprotein CD28 Proteins 0.000 description 3
- 241000701044 Human gammaherpesvirus 4 Species 0.000 description 3
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 3
- 206010020751 Hypersensitivity Diseases 0.000 description 3
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 3
- 240000007472 Leucaena leucocephala Species 0.000 description 3
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 3
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 3
- 241000699666 Mus <mouse, genus> Species 0.000 description 3
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 3
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 3
- RRHONYZEMUNMJX-UHFFFAOYSA-N N-[5-[[5-[(4-acetyl-1-piperazinyl)-oxomethyl]-4-methoxy-2-methylphenyl]thio]-2-thiazolyl]-4-[(3-methylbutan-2-ylamino)methyl]benzamide Chemical compound C1=C(C(=O)N2CCN(CC2)C(C)=O)C(OC)=CC(C)=C1SC(S1)=CN=C1NC(=O)C1=CC=C(CNC(C)C(C)C)C=C1 RRHONYZEMUNMJX-UHFFFAOYSA-N 0.000 description 3
- 239000007832 Na2SO4 Substances 0.000 description 3
- 201000001263 Psoriatic Arthritis Diseases 0.000 description 3
- 208000036824 Psoriatic arthropathy Diseases 0.000 description 3
- 241000700159 Rattus Species 0.000 description 3
- 241000283984 Rodentia Species 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 229930006000 Sucrose Natural products 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 208000000389 T-cell leukemia Diseases 0.000 description 3
- 208000028530 T-cell lymphoblastic leukemia/lymphoma Diseases 0.000 description 3
- 102100027213 T-cell-specific surface glycoprotein CD28 Human genes 0.000 description 3
- 102100029823 Tyrosine-protein kinase BTK Human genes 0.000 description 3
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 3
- 230000036592 analgesia Effects 0.000 description 3
- 230000000844 anti-bacterial effect Effects 0.000 description 3
- 230000003110 anti-inflammatory effect Effects 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 3
- 239000003899 bactericide agent Substances 0.000 description 3
- 125000005605 benzo group Chemical group 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 230000037396 body weight Effects 0.000 description 3
- 239000012267 brine Substances 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 229910000024 caesium carbonate Inorganic materials 0.000 description 3
- 125000001589 carboacyl group Chemical group 0.000 description 3
- 150000001721 carbon Chemical group 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 3
- 229960004316 cisplatin Drugs 0.000 description 3
- 238000011260 co-administration Methods 0.000 description 3
- 208000010247 contact dermatitis Diseases 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 230000002950 deficient Effects 0.000 description 3
- 230000006806 disease prevention Effects 0.000 description 3
- 238000010494 dissociation reaction Methods 0.000 description 3
- 230000005593 dissociations Effects 0.000 description 3
- 239000012039 electrophile Substances 0.000 description 3
- AAKJLRGGTJKAMG-UHFFFAOYSA-N erlotinib Chemical compound C=12C=C(OCCOC)C(OCCOC)=CC2=NC=NC=1NC1=CC=CC(C#C)=C1 AAKJLRGGTJKAMG-UHFFFAOYSA-N 0.000 description 3
- 238000003818 flash chromatography Methods 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- 150000004820 halides Chemical class 0.000 description 3
- 238000004128 high performance liquid chromatography Methods 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- KTUFNOKKBVMGRW-UHFFFAOYSA-N imatinib Chemical compound C1CN(C)CCN1CC1=CC=C(C(=O)NC=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)C=C1 KTUFNOKKBVMGRW-UHFFFAOYSA-N 0.000 description 3
- 208000026278 immune system disease Diseases 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 230000002427 irreversible effect Effects 0.000 description 3
- 238000011813 knockout mouse model Methods 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- OKKJLVBELUTLKV-VMNATFBRSA-N methanol-d1 Chemical compound [2H]OC OKKJLVBELUTLKV-VMNATFBRSA-N 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- 230000035772 mutation Effects 0.000 description 3
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 3
- 125000004043 oxo group Chemical group O=* 0.000 description 3
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- NXJCBFBQEVOTOW-UHFFFAOYSA-L palladium(2+);dihydroxide Chemical compound O[Pd]O NXJCBFBQEVOTOW-UHFFFAOYSA-L 0.000 description 3
- 239000006072 paste Substances 0.000 description 3
- 230000026731 phosphorylation Effects 0.000 description 3
- 238000006366 phosphorylation reaction Methods 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 238000011533 pre-incubation Methods 0.000 description 3
- 125000006239 protecting group Chemical group 0.000 description 3
- 238000000159 protein binding assay Methods 0.000 description 3
- 125000004076 pyridyl group Chemical group 0.000 description 3
- 239000000700 radioactive tracer Substances 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 229910001958 silver carbonate Inorganic materials 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 3
- 239000006228 supernatant Substances 0.000 description 3
- 239000000375 suspending agent Substances 0.000 description 3
- 230000008961 swelling Effects 0.000 description 3
- 125000004149 thio group Chemical group *S* 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 238000011200 topical administration Methods 0.000 description 3
- YIYBQIKDCADOSF-ONEGZZNKSA-N trans-pent-2-enoic acid Chemical compound CC\C=C\C(O)=O YIYBQIKDCADOSF-ONEGZZNKSA-N 0.000 description 3
- 229910000404 tripotassium phosphate Inorganic materials 0.000 description 3
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 description 3
- 229960004528 vincristine Drugs 0.000 description 3
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 3
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 3
- WBYWAXJHAXSJNI-VOTSOKGWSA-M .beta-Phenylacrylic acid Natural products [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 description 2
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 2
- HKSXOMSTPJEKGX-UHFFFAOYSA-N 1-methyl-4-[5-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-6-oxo-1H-pyridin-3-yl]pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(N(C=C1)C)=O)C HKSXOMSTPJEKGX-UHFFFAOYSA-N 0.000 description 2
- VQMYGTDZQWQAHD-UHFFFAOYSA-N 1-methyl-4-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-3-yl]pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(N(C=C1)C)=O)CCC VQMYGTDZQWQAHD-UHFFFAOYSA-N 0.000 description 2
- RHBYJMWZJBAKCK-UHFFFAOYSA-N 1-methyl-4-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(N(C=C1)C)=O RHBYJMWZJBAKCK-UHFFFAOYSA-N 0.000 description 2
- PAMIQIKDUOTOBW-UHFFFAOYSA-N 1-methylpiperidine Chemical compound CN1CCCCC1 PAMIQIKDUOTOBW-UHFFFAOYSA-N 0.000 description 2
- DGHHQBMTXTWTJV-BQAIUKQQSA-N 119413-54-6 Chemical compound Cl.C1=C(O)C(CN(C)C)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 DGHHQBMTXTWTJV-BQAIUKQQSA-N 0.000 description 2
- WXTMDXOMEHJXQO-UHFFFAOYSA-N 2,5-dihydroxybenzoic acid Chemical compound OC(=O)C1=CC(O)=CC=C1O WXTMDXOMEHJXQO-UHFFFAOYSA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- CHBCAZLQVOURQV-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)CCC CHBCAZLQVOURQV-UHFFFAOYSA-N 0.000 description 2
- IJKQQNYKCRXPAB-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC=C1)CCC IJKQQNYKCRXPAB-UHFFFAOYSA-N 0.000 description 2
- WZIKSKDQSWFZIZ-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CCC WZIKSKDQSWFZIZ-UHFFFAOYSA-N 0.000 description 2
- VSBUHLWQZKJVSJ-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)CCC VSBUHLWQZKJVSJ-UHFFFAOYSA-N 0.000 description 2
- MCSQVRSXTDPUTM-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=CN=C1)=O MCSQVRSXTDPUTM-UHFFFAOYSA-N 0.000 description 2
- IJRLPEXRIUTHNB-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC=C1 IJRLPEXRIUTHNB-UHFFFAOYSA-N 0.000 description 2
- BSGNMVVVVOZGJQ-UHFFFAOYSA-N 3-[(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC=C1)=O BSGNMVVVVOZGJQ-UHFFFAOYSA-N 0.000 description 2
- BAVJWKWCUGEHFU-UHFFFAOYSA-N 3-[(1-prop-2-enoylpiperidin-4-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 BAVJWKWCUGEHFU-UHFFFAOYSA-N 0.000 description 2
- ZTNFLHLJIJEWKG-UHFFFAOYSA-N 3-[5-[[1-(2-chloroacetyl)azetidin-3-yl]amino]-6-oxo-1H-pyridin-3-yl]-N-(3-methyl-4-propan-2-ylphenyl)benzamide Chemical compound ClCC(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=CC1 ZTNFLHLJIJEWKG-UHFFFAOYSA-N 0.000 description 2
- XUOFWJMINXWUHA-CYBMUJFWSA-N 3-[[(3R)-1-(2-chloroacetyl)pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound ClCC(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 XUOFWJMINXWUHA-CYBMUJFWSA-N 0.000 description 2
- ZKHPACMSCUZUBJ-MJICGBHWSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1)C ZKHPACMSCUZUBJ-MJICGBHWSA-N 0.000 description 2
- JJGTUTRGRHOGGG-REMYHPOOSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1)C JJGTUTRGRHOGGG-REMYHPOOSA-N 0.000 description 2
- OZUNZJFYNYPIES-WAUBUAMCSA-N 3-[[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 OZUNZJFYNYPIES-WAUBUAMCSA-N 0.000 description 2
- KNNPMPLPENUYLN-WOMSROEHSA-N 3-[[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 KNNPMPLPENUYLN-WOMSROEHSA-N 0.000 description 2
- BCMIPQRMZREUBF-XQASTXFUSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-(cyclopropylmethyl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 BCMIPQRMZREUBF-XQASTXFUSA-N 0.000 description 2
- AZADOMHKENFIAM-NEABXICISA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-(cyclopropylmethyl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 AZADOMHKENFIAM-NEABXICISA-N 0.000 description 2
- JOUPVBACYLUYML-WEBJUUICSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC JOUPVBACYLUYML-WEBJUUICSA-N 0.000 description 2
- MXMFZBMQURLPCZ-JHBSALDVSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 MXMFZBMQURLPCZ-JHBSALDVSA-N 0.000 description 2
- HAVCQKQRICXVQR-LSTKMXGYSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-(cyclopropylmethyl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 HAVCQKQRICXVQR-LSTKMXGYSA-N 0.000 description 2
- JUKQPZZEUXYSHF-LSTKMXGYSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 JUKQPZZEUXYSHF-LSTKMXGYSA-N 0.000 description 2
- UIOFZALYTZMQRZ-VQSDEBNRSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-(cyclopropylmethyl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 UIOFZALYTZMQRZ-VQSDEBNRSA-N 0.000 description 2
- NXOYLDBGYJNJPZ-LAQIPUCWSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC NXOYLDBGYJNJPZ-LAQIPUCWSA-N 0.000 description 2
- DJVWMKNWRWDSEA-LAQIPUCWSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC DJVWMKNWRWDSEA-LAQIPUCWSA-N 0.000 description 2
- FSCVPBOTKMWLCE-REMYHPOOSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC FSCVPBOTKMWLCE-REMYHPOOSA-N 0.000 description 2
- AKYJOSRIVYFVDS-XYBNCVKDSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC AKYJOSRIVYFVDS-XYBNCVKDSA-N 0.000 description 2
- XYPYZNPNSGREPT-BGTMTDRBSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 XYPYZNPNSGREPT-BGTMTDRBSA-N 0.000 description 2
- DFPAIIZEOAEQHH-WLMDGNKMSA-N 3-[[(3R)-1-[(E)-4-morpholin-4-ylbut-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound O1CCN(CC1)C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 DFPAIIZEOAEQHH-WLMDGNKMSA-N 0.000 description 2
- JNBHZAXRUCOYJX-VQSDEBNRSA-N 3-[[(3R)-1-[(E)-4-morpholin-4-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound O1CCN(CC1)C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 JNBHZAXRUCOYJX-VQSDEBNRSA-N 0.000 description 2
- PAYUQOHLTGKVMX-RXWXAUPJSA-N 3-[[(3R)-1-[(E)-4-phenylbut-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(=CC=CC=C1)C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 PAYUQOHLTGKVMX-RXWXAUPJSA-N 0.000 description 2
- XYSIGSZHIYJTIU-FAZYEBOESA-N 3-[[(3R)-1-[(E)-4-phenylbut-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(=CC=CC=C1)C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 XYSIGSZHIYJTIU-FAZYEBOESA-N 0.000 description 2
- ZUOFAQIWFAZXEM-DPZZJQGRSA-N 3-[[(3R)-1-[(E)-4-piperidin-1-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound N1(CCCCC1)C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 ZUOFAQIWFAZXEM-DPZZJQGRSA-N 0.000 description 2
- CPGZRXIPDSUTDT-JCOOBLOPSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC)C CPGZRXIPDSUTDT-JCOOBLOPSA-N 0.000 description 2
- QLDZZQLYVNJDOS-JCOOBLOPSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC)C QLDZZQLYVNJDOS-JCOOBLOPSA-N 0.000 description 2
- SRGUZQVFEOFWCX-IEJURRBGSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC)C SRGUZQVFEOFWCX-IEJURRBGSA-N 0.000 description 2
- ANFCYMXXUIJGFX-FZMUQLTDSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C ANFCYMXXUIJGFX-FZMUQLTDSA-N 0.000 description 2
- ZKHPACMSCUZUBJ-JXTAAOLFSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1)C ZKHPACMSCUZUBJ-JXTAAOLFSA-N 0.000 description 2
- OLQROOMEFRNAJZ-GHOSXJJBSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC)C OLQROOMEFRNAJZ-GHOSXJJBSA-N 0.000 description 2
- DRTWZSGCELXEIN-GHOSXJJBSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC)C DRTWZSGCELXEIN-GHOSXJJBSA-N 0.000 description 2
- VQCKTSWGABIXJP-UEKDZROGSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC)C VQCKTSWGABIXJP-UEKDZROGSA-N 0.000 description 2
- BLWVVBIBRVSVGU-BDUNBXCCSA-N 3-[[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C BLWVVBIBRVSVGU-BDUNBXCCSA-N 0.000 description 2
- OZUNZJFYNYPIES-RTZWDOBSSA-N 3-[[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 OZUNZJFYNYPIES-RTZWDOBSSA-N 0.000 description 2
- KNNPMPLPENUYLN-KZSQQDEKSA-N 3-[[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 KNNPMPLPENUYLN-KZSQQDEKSA-N 0.000 description 2
- MOSXJBRUOYXWKQ-NAASZUNDSA-N 3-[[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 MOSXJBRUOYXWKQ-NAASZUNDSA-N 0.000 description 2
- HVPVJWYHLVBICY-WHLIDYPQSA-N 3-[[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 HVPVJWYHLVBICY-WHLIDYPQSA-N 0.000 description 2
- BCMIPQRMZREUBF-BFJGLSHQSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-(cyclopropylmethyl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 BCMIPQRMZREUBF-BFJGLSHQSA-N 0.000 description 2
- AZADOMHKENFIAM-DFKDPWMTSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-(cyclopropylmethyl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 AZADOMHKENFIAM-DFKDPWMTSA-N 0.000 description 2
- JOUPVBACYLUYML-UKTKYZJWSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC JOUPVBACYLUYML-UKTKYZJWSA-N 0.000 description 2
- VTDAPTNNCBLVBR-AUYOJAJCSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC VTDAPTNNCBLVBR-AUYOJAJCSA-N 0.000 description 2
- MXMFZBMQURLPCZ-SIEGMDDESA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 MXMFZBMQURLPCZ-SIEGMDDESA-N 0.000 description 2
- HAVCQKQRICXVQR-PHXNAMCJSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-(cyclopropylmethyl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 HAVCQKQRICXVQR-PHXNAMCJSA-N 0.000 description 2
- JUKQPZZEUXYSHF-PHXNAMCJSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 JUKQPZZEUXYSHF-PHXNAMCJSA-N 0.000 description 2
- UIOFZALYTZMQRZ-SNDJDQCESA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-(cyclopropylmethyl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 UIOFZALYTZMQRZ-SNDJDQCESA-N 0.000 description 2
- NXOYLDBGYJNJPZ-BDUNBXCCSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC NXOYLDBGYJNJPZ-BDUNBXCCSA-N 0.000 description 2
- DJVWMKNWRWDSEA-BDUNBXCCSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC DJVWMKNWRWDSEA-BDUNBXCCSA-N 0.000 description 2
- AKYJOSRIVYFVDS-FAAWYNLUSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC AKYJOSRIVYFVDS-FAAWYNLUSA-N 0.000 description 2
- CVTYLKPWHIKMBD-FAAWYNLUSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC CVTYLKPWHIKMBD-FAAWYNLUSA-N 0.000 description 2
- XYPYZNPNSGREPT-YLSAJCSVSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 XYPYZNPNSGREPT-YLSAJCSVSA-N 0.000 description 2
- LTTFOPMNGJDBOA-HNNXBMFYSA-N 3-[[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 LTTFOPMNGJDBOA-HNNXBMFYSA-N 0.000 description 2
- IXOZTYJRHHKUGM-AWEZNQCLSA-N 3-[[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 IXOZTYJRHHKUGM-AWEZNQCLSA-N 0.000 description 2
- CNBALCOYXVAWPI-AATRIKPKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]-methylamino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C)C CNBALCOYXVAWPI-AATRIKPKSA-N 0.000 description 2
- ILPLYHZZUONYRB-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]-methylamino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C)C ILPLYHZZUONYRB-SNAWJCMRSA-N 0.000 description 2
- OFNBUPGVQQAYRR-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C OFNBUPGVQQAYRR-SNAWJCMRSA-N 0.000 description 2
- XCKBNMQIYDYLJL-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C XCKBNMQIYDYLJL-SNAWJCMRSA-N 0.000 description 2
- ZTFOSMZTCMJVFM-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C ZTFOSMZTCMJVFM-SNAWJCMRSA-N 0.000 description 2
- PUFTXTHTMXWPBF-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C PUFTXTHTMXWPBF-SNAWJCMRSA-N 0.000 description 2
- MJFJZUZXUHMSFY-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C MJFJZUZXUHMSFY-SNAWJCMRSA-N 0.000 description 2
- VDXWPTLBSXYYRF-ONEGZZNKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=CN=C1)=O)C VDXWPTLBSXYYRF-ONEGZZNKSA-N 0.000 description 2
- HQLASTZOZSTWEV-FNORWQNLSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC=C1)C HQLASTZOZSTWEV-FNORWQNLSA-N 0.000 description 2
- OSIHKCXWUJDJHH-ONEGZZNKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1)C OSIHKCXWUJDJHH-ONEGZZNKSA-N 0.000 description 2
- JFLMEHCSBCCWHU-ONEGZZNKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC=C1)=O)C JFLMEHCSBCCWHU-ONEGZZNKSA-N 0.000 description 2
- HJFGQGREKQFREG-AATRIKPKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC)C HJFGQGREKQFREG-AATRIKPKSA-N 0.000 description 2
- IRTYJWMKAAJKQQ-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C IRTYJWMKAAJKQQ-SNAWJCMRSA-N 0.000 description 2
- CLGCOEPYJHHYKC-ONEGZZNKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1)C CLGCOEPYJHHYKC-ONEGZZNKSA-N 0.000 description 2
- KSTVAWGAHAMPPY-NSCUHMNNSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=CN=C1)=O KSTVAWGAHAMPPY-NSCUHMNNSA-N 0.000 description 2
- PDDZCSPZAJCHMG-GQCTYLIASA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC=C1 PDDZCSPZAJCHMG-GQCTYLIASA-N 0.000 description 2
- HGTYKCMVUIMJKS-NSCUHMNNSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC=C1)=O HGTYKCMVUIMJKS-NSCUHMNNSA-N 0.000 description 2
- MKQFHCNJKNLWMW-NSCUHMNNSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 MKQFHCNJKNLWMW-NSCUHMNNSA-N 0.000 description 2
- FCZOZPOVKLTDHP-ONEGZZNKSA-N 3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=CN=C1)=O FCZOZPOVKLTDHP-ONEGZZNKSA-N 0.000 description 2
- DNBAICCNBTWRLK-FNORWQNLSA-N 3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC=C1 DNBAICCNBTWRLK-FNORWQNLSA-N 0.000 description 2
- KFVPCUZJSRVARS-ONEGZZNKSA-N 3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 KFVPCUZJSRVARS-ONEGZZNKSA-N 0.000 description 2
- FXGIVWPJUSOCBL-ONEGZZNKSA-N 3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC=C1)=O FXGIVWPJUSOCBL-ONEGZZNKSA-N 0.000 description 2
- LJEODIVZIKKZEY-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 LJEODIVZIKKZEY-NSCUHMNNSA-N 0.000 description 2
- JYROVMHBPRCEGH-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)CC1CC1 JYROVMHBPRCEGH-NSCUHMNNSA-N 0.000 description 2
- ONHJRQVMSWUVFW-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)CC1CC1 ONHJRQVMSWUVFW-NSCUHMNNSA-N 0.000 description 2
- IXIRRGUBMLFGAT-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)CC1CC1 IXIRRGUBMLFGAT-NSCUHMNNSA-N 0.000 description 2
- WJEXBZJDZMRJFE-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)CC1CC1 WJEXBZJDZMRJFE-OWOJBTEDSA-N 0.000 description 2
- OKBAKGSWQYPCBC-HWKANZROSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC=C1)CC1CC1 OKBAKGSWQYPCBC-HWKANZROSA-N 0.000 description 2
- PJDMMFXQTICAPC-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C PJDMMFXQTICAPC-ONEGZZNKSA-N 0.000 description 2
- GBLGEPIZGPOSMP-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C GBLGEPIZGPOSMP-ONEGZZNKSA-N 0.000 description 2
- GWWGEFRLUIPIMB-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C GWWGEFRLUIPIMB-ONEGZZNKSA-N 0.000 description 2
- ROBXBKFUULCZEC-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C ROBXBKFUULCZEC-ONEGZZNKSA-N 0.000 description 2
- ASKXFWAFHVFQDZ-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C ASKXFWAFHVFQDZ-ONEGZZNKSA-N 0.000 description 2
- CAJZMPFWQZUKAE-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C CAJZMPFWQZUKAE-NSCUHMNNSA-N 0.000 description 2
- KUXZWLIVHFEFAH-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C KUXZWLIVHFEFAH-NSCUHMNNSA-N 0.000 description 2
- DSKXWEUJQOMZGL-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O DSKXWEUJQOMZGL-NSCUHMNNSA-N 0.000 description 2
- PVFZMXRQQCHQSX-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)OC PVFZMXRQQCHQSX-NSCUHMNNSA-N 0.000 description 2
- ZXLGJTXDHYSTHS-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O ZXLGJTXDHYSTHS-NSCUHMNNSA-N 0.000 description 2
- GJCMCCPKSVSVKG-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O GJCMCCPKSVSVKG-NSCUHMNNSA-N 0.000 description 2
- ZTWVBXPEDIITPJ-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=CN=C1)=O ZTWVBXPEDIITPJ-OWOJBTEDSA-N 0.000 description 2
- ORTDEFAEUIFSSZ-HWKANZROSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC=C1 ORTDEFAEUIFSSZ-HWKANZROSA-N 0.000 description 2
- RRFOLXFPSRXDOV-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC=C1)=O RRFOLXFPSRXDOV-OWOJBTEDSA-N 0.000 description 2
- GGZUPFDLLKFSBH-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]-(cyclopropylmethyl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 GGZUPFDLLKFSBH-NSCUHMNNSA-N 0.000 description 2
- KVLDULKBOIOYIR-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]-(cyclopropylmethyl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 KVLDULKBOIOYIR-OWOJBTEDSA-N 0.000 description 2
- ISPLXSNFXRGCTO-SNAWJCMRSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC ISPLXSNFXRGCTO-SNAWJCMRSA-N 0.000 description 2
- UBUCTIHLQJBDLP-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC UBUCTIHLQJBDLP-NSCUHMNNSA-N 0.000 description 2
- DHJCMNLTOZMQNU-WEVVVXLNSA-N 3-[[1-[(E)-4-cyclohexylbut-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CCCCC1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC DHJCMNLTOZMQNU-WEVVVXLNSA-N 0.000 description 2
- KFSADMVZMGBJJD-XBXARRHUSA-N 3-[[1-[(E)-4-cyclohexylbut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CCCCC1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 KFSADMVZMGBJJD-XBXARRHUSA-N 0.000 description 2
- PRRVTZWTBGIORC-NSCUHMNNSA-N 3-[[1-[(E)-4-methoxybut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound COC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 PRRVTZWTBGIORC-NSCUHMNNSA-N 0.000 description 2
- IBLQROORQUWXGR-OWOJBTEDSA-N 3-[[1-[(E)-4-morpholin-4-ylbut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound O1CCN(CC1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 IBLQROORQUWXGR-OWOJBTEDSA-N 0.000 description 2
- UQDDXWJDLRGAIR-XBXARRHUSA-N 3-[[1-[(E)-4-phenylbut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(=CC=CC=C1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 UQDDXWJDLRGAIR-XBXARRHUSA-N 0.000 description 2
- QQIGQGFJLDUVGF-SNAWJCMRSA-N 3-[[1-[(E)-4-piperidin-1-ylbut-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound N1(CCCCC1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 QQIGQGFJLDUVGF-SNAWJCMRSA-N 0.000 description 2
- TYTXXMRMQLCHFI-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CC1 TYTXXMRMQLCHFI-UHFFFAOYSA-N 0.000 description 2
- MALZLVYTWGLOET-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C1CC1 MALZLVYTWGLOET-UHFFFAOYSA-N 0.000 description 2
- YRDQMAFITVTKIT-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)C)C1CC1 YRDQMAFITVTKIT-UHFFFAOYSA-N 0.000 description 2
- SOCZMNFCLKWYGE-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)C)=O)C1CC1 SOCZMNFCLKWYGE-UHFFFAOYSA-N 0.000 description 2
- UAXYMVHSICHBEC-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methylpyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)C)C1CC1 UAXYMVHSICHBEC-UHFFFAOYSA-N 0.000 description 2
- AZSICHDIPGJZLZ-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C1CC1 AZSICHDIPGJZLZ-UHFFFAOYSA-N 0.000 description 2
- XUEDWMMBFOXJKP-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C1CC1 XUEDWMMBFOXJKP-UHFFFAOYSA-N 0.000 description 2
- HPGDDDRYCHTEAG-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrazin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)C)=O)C1CC1 HPGDDDRYCHTEAG-UHFFFAOYSA-N 0.000 description 2
- QCCXAAZMUYDQHY-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC=C1)C1CC1 QCCXAAZMUYDQHY-UHFFFAOYSA-N 0.000 description 2
- OOSGXLHJAQDQJZ-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 OOSGXLHJAQDQJZ-UHFFFAOYSA-N 0.000 description 2
- HFXXPAVTLZVVDQ-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)CC1CC1 HFXXPAVTLZVVDQ-UHFFFAOYSA-N 0.000 description 2
- RFBBGTCFLCLVQK-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)C)=O)CC1CC1 RFBBGTCFLCLVQK-UHFFFAOYSA-N 0.000 description 2
- VTAUVXMWHLBIBY-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)CC1CC1 VTAUVXMWHLBIBY-UHFFFAOYSA-N 0.000 description 2
- IZYBTFCQKMAPMH-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methylpyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)C)CC1CC1 IZYBTFCQKMAPMH-UHFFFAOYSA-N 0.000 description 2
- MTLHOKYJYJMBEW-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)CC1CC1 MTLHOKYJYJMBEW-UHFFFAOYSA-N 0.000 description 2
- MERJUSSPHQNFTK-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)CC1CC1 MERJUSSPHQNFTK-UHFFFAOYSA-N 0.000 description 2
- IPSOLZCBHUXBQQ-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrazin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)C)=O)CC1CC1 IPSOLZCBHUXBQQ-UHFFFAOYSA-N 0.000 description 2
- WAADRCXUKIDETM-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)C)=O)CC1CC1 WAADRCXUKIDETM-UHFFFAOYSA-N 0.000 description 2
- HDSQSBTXIGKWLI-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)CC1CC1 HDSQSBTXIGKWLI-UHFFFAOYSA-N 0.000 description 2
- RUKDPYBJEZHGQR-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC=C1)CC1CC1 RUKDPYBJEZHGQR-UHFFFAOYSA-N 0.000 description 2
- OKICNDUSEOVKAU-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 OKICNDUSEOVKAU-UHFFFAOYSA-N 0.000 description 2
- CEUGNBAQTAJCBA-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)CC1CC1 CEUGNBAQTAJCBA-UHFFFAOYSA-N 0.000 description 2
- PRGOPAHXNZPIFN-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylpiperidin-4-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 PRGOPAHXNZPIFN-UHFFFAOYSA-N 0.000 description 2
- HQXMDHFIKARXNB-IBGZPJMESA-N 3-[cyclopropylmethyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 HQXMDHFIKARXNB-IBGZPJMESA-N 0.000 description 2
- VBJXXGQZWOPCFE-SFHVURJKSA-N 3-[cyclopropylmethyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 VBJXXGQZWOPCFE-SFHVURJKSA-N 0.000 description 2
- NVGCOSZAHJJIKH-SFHVURJKSA-N 3-[cyclopropylmethyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 NVGCOSZAHJJIKH-SFHVURJKSA-N 0.000 description 2
- WERNGIZPQFJTHI-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC WERNGIZPQFJTHI-UHFFFAOYSA-N 0.000 description 2
- DOXZCAKOUGOHAQ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)CC DOXZCAKOUGOHAQ-UHFFFAOYSA-N 0.000 description 2
- OGXNGGHZBLVRTJ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)C)CC OGXNGGHZBLVRTJ-UHFFFAOYSA-N 0.000 description 2
- BUWNNENKNMLRMZ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)C)=O)CC BUWNNENKNMLRMZ-UHFFFAOYSA-N 0.000 description 2
- VKGQEHPALMWWSU-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)CC VKGQEHPALMWWSU-UHFFFAOYSA-N 0.000 description 2
- YRFLAGQQRNRTMU-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methylpyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)C)CC YRFLAGQQRNRTMU-UHFFFAOYSA-N 0.000 description 2
- NZJCRXLHKUQQLQ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrazin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)C)=O)CC NZJCRXLHKUQQLQ-UHFFFAOYSA-N 0.000 description 2
- CEHUDVLAUXGBKF-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)C)=O)CC CEHUDVLAUXGBKF-UHFFFAOYSA-N 0.000 description 2
- OQZAWBMWMITXHU-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)CC OQZAWBMWMITXHU-UHFFFAOYSA-N 0.000 description 2
- TXPUVPCCJDEENJ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC=C1)CC TXPUVPCCJDEENJ-UHFFFAOYSA-N 0.000 description 2
- TZBOOLVTJRRLCG-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC TZBOOLVTJRRLCG-UHFFFAOYSA-N 0.000 description 2
- NFJVDVJLEZCDBJ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)CC NFJVDVJLEZCDBJ-UHFFFAOYSA-N 0.000 description 2
- COYSESRYIUQVJY-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylpiperidin-4-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC COYSESRYIUQVJY-UHFFFAOYSA-N 0.000 description 2
- ORAXYQAMNHJTRV-KRWDZBQOSA-N 3-[ethyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC ORAXYQAMNHJTRV-KRWDZBQOSA-N 0.000 description 2
- BHLXJEOSBVYQTJ-INIZCTEOSA-N 3-[ethyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC BHLXJEOSBVYQTJ-INIZCTEOSA-N 0.000 description 2
- SNKNLNMQUIZLKM-INIZCTEOSA-N 3-[ethyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC SNKNLNMQUIZLKM-INIZCTEOSA-N 0.000 description 2
- YJXVNJIQTZLYKT-INIZCTEOSA-N 3-[ethyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC YJXVNJIQTZLYKT-INIZCTEOSA-N 0.000 description 2
- JYRNEBZSLUBSRC-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-methylpyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)C)=O)C JYRNEBZSLUBSRC-UHFFFAOYSA-N 0.000 description 2
- KTFMGUMWLXAQBT-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-oxo-1H-pyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(NC=C1)=O)C KTFMGUMWLXAQBT-UHFFFAOYSA-N 0.000 description 2
- BZYLWSHLMUMWAU-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(4-methylpyridin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)C)C BZYLWSHLMUMWAU-UHFFFAOYSA-N 0.000 description 2
- WQXNQLOTNLQINE-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(6-methylpyrazin-2-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)C)=O)C WQXNQLOTNLQINE-UHFFFAOYSA-N 0.000 description 2
- JBCLKTVYYHSARM-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C JBCLKTVYYHSARM-UHFFFAOYSA-N 0.000 description 2
- JODVJHHDINPMPG-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC=C1)C JODVJHHDINPMPG-UHFFFAOYSA-N 0.000 description 2
- NGIJPFIRTKJZNK-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C NGIJPFIRTKJZNK-UHFFFAOYSA-N 0.000 description 2
- TYYQXPJBCVARIE-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C TYYQXPJBCVARIE-UHFFFAOYSA-N 0.000 description 2
- NIRODWVVQNCNOY-UHFFFAOYSA-N 3-[methyl-(1-prop-2-enoylpiperidin-4-yl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C NIRODWVVQNCNOY-UHFFFAOYSA-N 0.000 description 2
- SDWCDWJKYSOFOQ-INIZCTEOSA-N 3-[methyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C SDWCDWJKYSOFOQ-INIZCTEOSA-N 0.000 description 2
- WVJCRPNLICTXAM-HNNXBMFYSA-N 3-[methyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C WVJCRPNLICTXAM-HNNXBMFYSA-N 0.000 description 2
- NHUPYGKRPROERW-SNAWJCMRSA-N 3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C NHUPYGKRPROERW-SNAWJCMRSA-N 0.000 description 2
- QIOXEFZXZAUXHZ-UHFFFAOYSA-N 4-[5-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-6-oxo-1H-pyridin-3-yl]-1-methylpyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(N(C=C1)C)=O)C1CC1 QIOXEFZXZAUXHZ-UHFFFAOYSA-N 0.000 description 2
- HAPJSYISJDAURZ-UHFFFAOYSA-N 4-[5-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-6-oxo-1H-pyridin-3-yl]-1-methylpyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(N(C=C1)C)=O)CC1CC1 HAPJSYISJDAURZ-UHFFFAOYSA-N 0.000 description 2
- IVGDXHDOXAPGOW-UHFFFAOYSA-N 4-[5-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-6-oxo-1H-pyridin-3-yl]-1-methylpyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(N(C=C1)C)=O)CC IVGDXHDOXAPGOW-UHFFFAOYSA-N 0.000 description 2
- VEDJHQWMCAONGI-UHFFFAOYSA-N 5-(2-methoxypyridin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CCC VEDJHQWMCAONGI-UHFFFAOYSA-N 0.000 description 2
- DUXIMLJQTPVFQO-UHFFFAOYSA-N 5-(2-methoxypyridin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC DUXIMLJQTPVFQO-UHFFFAOYSA-N 0.000 description 2
- IAYAAFUERRVDFQ-SHCNBLLESA-N 5-(2-methoxypyridin-4-yl)-3-[[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC IAYAAFUERRVDFQ-SHCNBLLESA-N 0.000 description 2
- CHCXHBMDGNYXFB-OAHLLOKOSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC CHCXHBMDGNYXFB-OAHLLOKOSA-N 0.000 description 2
- YRAQTSYDYBRQAV-QTWIXLPOSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC YRAQTSYDYBRQAV-QTWIXLPOSA-N 0.000 description 2
- IAYAAFUERRVDFQ-WRFKIARRSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC IAYAAFUERRVDFQ-WRFKIARRSA-N 0.000 description 2
- CHCXHBMDGNYXFB-HNNXBMFYSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC CHCXHBMDGNYXFB-HNNXBMFYSA-N 0.000 description 2
- FZUVVZDMPLEZLP-AWEZNQCLSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC FZUVVZDMPLEZLP-AWEZNQCLSA-N 0.000 description 2
- YECBQKGOVBKLET-SNAWJCMRSA-N 5-(2-methoxypyridin-4-yl)-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC YECBQKGOVBKLET-SNAWJCMRSA-N 0.000 description 2
- KWEYEFCTRSLJKY-SNAWJCMRSA-N 5-(2-methoxypyridin-4-yl)-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC KWEYEFCTRSLJKY-SNAWJCMRSA-N 0.000 description 2
- ZYKNZPPVPNOFOR-UHFFFAOYSA-N 5-(2-methoxypyridin-4-yl)-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C ZYKNZPPVPNOFOR-UHFFFAOYSA-N 0.000 description 2
- IJQPGCKAECKSFD-INIZCTEOSA-N 5-(2-methoxypyridin-4-yl)-3-[methyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C IJQPGCKAECKSFD-INIZCTEOSA-N 0.000 description 2
- AGSVRRBRIUOTEZ-HNNXBMFYSA-N 5-(2-methoxypyridin-4-yl)-3-[methyl-[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C AGSVRRBRIUOTEZ-HNNXBMFYSA-N 0.000 description 2
- WCHJSFFKQMSVET-UHFFFAOYSA-N 5-(2-methoxypyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)CCC WCHJSFFKQMSVET-UHFFFAOYSA-N 0.000 description 2
- KKFBUIGFWKGYBR-UHFFFAOYSA-N 5-(2-methoxypyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O KKFBUIGFWKGYBR-UHFFFAOYSA-N 0.000 description 2
- OKRFIDILJPJBKO-SNAWJCMRSA-N 5-(2-methoxypyrimidin-4-yl)-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O OKRFIDILJPJBKO-SNAWJCMRSA-N 0.000 description 2
- RNVIUEHQZSECOS-UHFFFAOYSA-N 5-(2-methoxypyrimidin-4-yl)-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C RNVIUEHQZSECOS-UHFFFAOYSA-N 0.000 description 2
- GWFMOYIVCZMRGS-AATRIKPKSA-N 5-(2-methoxypyrimidin-4-yl)-3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C GWFMOYIVCZMRGS-AATRIKPKSA-N 0.000 description 2
- VFYWITDBLQHFHB-UHFFFAOYSA-N 5-(2-methylpyridin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)C)CCC VFYWITDBLQHFHB-UHFFFAOYSA-N 0.000 description 2
- XEBFEUKHFWJHHX-UHFFFAOYSA-N 5-(2-methylpyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)C)=O)CCC XEBFEUKHFWJHHX-UHFFFAOYSA-N 0.000 description 2
- DFIYJULRLKQKDM-UHFFFAOYSA-N 5-(2-methylpyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)C)=O DFIYJULRLKQKDM-UHFFFAOYSA-N 0.000 description 2
- IJEUXQRKIKQHFY-UHFFFAOYSA-N 5-(4-methoxypyridin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)CCC IJEUXQRKIKQHFY-UHFFFAOYSA-N 0.000 description 2
- HOQUOFWQTFRLDX-UHFFFAOYSA-N 5-(4-methoxypyridin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)OC HOQUOFWQTFRLDX-UHFFFAOYSA-N 0.000 description 2
- NSCJGWKGPIZPBO-UHFFFAOYSA-N 5-(4-methylpyridin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)C)CCC NSCJGWKGPIZPBO-UHFFFAOYSA-N 0.000 description 2
- XJAYDNLLUDFAFF-UHFFFAOYSA-N 5-(4-methylpyridin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)C XJAYDNLLUDFAFF-UHFFFAOYSA-N 0.000 description 2
- UDWZCLURZLAPNX-SNAWJCMRSA-N 5-(6-methoxypyrazin-2-yl)-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O UDWZCLURZLAPNX-SNAWJCMRSA-N 0.000 description 2
- IEEUZBKZVDCVDT-UHFFFAOYSA-N 5-(6-methoxypyrazin-2-yl)-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C IEEUZBKZVDCVDT-UHFFFAOYSA-N 0.000 description 2
- RQTLYCSSHPCKRB-AATRIKPKSA-N 5-(6-methoxypyrazin-2-yl)-3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C RQTLYCSSHPCKRB-AATRIKPKSA-N 0.000 description 2
- DGAZEGNLIAVPIM-UHFFFAOYSA-N 5-(6-methoxypyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)CCC DGAZEGNLIAVPIM-UHFFFAOYSA-N 0.000 description 2
- RHTXUOXRALHISP-SNAWJCMRSA-N 5-(6-methoxypyrimidin-4-yl)-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O RHTXUOXRALHISP-SNAWJCMRSA-N 0.000 description 2
- VIRYMHFVFWIJNG-UHFFFAOYSA-N 5-(6-methoxypyrimidin-4-yl)-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C VIRYMHFVFWIJNG-UHFFFAOYSA-N 0.000 description 2
- PKXOQKVXBOJXCE-AATRIKPKSA-N 5-(6-methoxypyrimidin-4-yl)-3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C PKXOQKVXBOJXCE-AATRIKPKSA-N 0.000 description 2
- WGEAJJOOGMDJDA-UHFFFAOYSA-N 5-(6-methylpyrazin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)C)=O)CCC WGEAJJOOGMDJDA-UHFFFAOYSA-N 0.000 description 2
- YTWLSBHTHHICCP-UHFFFAOYSA-N 5-(6-methylpyrazin-2-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)C)=O YTWLSBHTHHICCP-UHFFFAOYSA-N 0.000 description 2
- CPFHAUBWJNDTJX-UHFFFAOYSA-N 5-(6-methylpyrimidin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)C)=O CPFHAUBWJNDTJX-UHFFFAOYSA-N 0.000 description 2
- KTICIAQYSFUMEY-CQSZACIVSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC KTICIAQYSFUMEY-CQSZACIVSA-N 0.000 description 2
- VLAXWQZTXYEYGY-WRFKIARRSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC VLAXWQZTXYEYGY-WRFKIARRSA-N 0.000 description 2
- KTICIAQYSFUMEY-AWEZNQCLSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3S)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC KTICIAQYSFUMEY-AWEZNQCLSA-N 0.000 description 2
- AQJAGORCMFYNCN-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C AQJAGORCMFYNCN-UHFFFAOYSA-N 0.000 description 2
- PQTXMNSVCXGSFV-SNAWJCMRSA-N 5-[2-(methylamino)pyrimidin-4-yl]-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O PQTXMNSVCXGSFV-SNAWJCMRSA-N 0.000 description 2
- HNALROUTKNBMGD-UHFFFAOYSA-N 5-[2-(methylamino)pyrimidin-4-yl]-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C HNALROUTKNBMGD-UHFFFAOYSA-N 0.000 description 2
- CJBIFXXBUXHEOF-UHFFFAOYSA-N 5-[6-(methylamino)pyrimidin-4-yl]-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)CCC CJBIFXXBUXHEOF-UHFFFAOYSA-N 0.000 description 2
- XTPFIKJKZRQENS-SNAWJCMRSA-N 5-[6-(methylamino)pyrimidin-4-yl]-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O XTPFIKJKZRQENS-SNAWJCMRSA-N 0.000 description 2
- YWPXJCWDWYDYGI-RPZUPEABSA-N 5-pyridin-4-yl-3-[[(3R)-1-[(E)-4-pyridin-2-ylbut-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound N1=C(C=CC=C1)C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 YWPXJCWDWYDYGI-RPZUPEABSA-N 0.000 description 2
- HBGNPOUHAAQMOP-SQZHUTIHSA-N 5-pyridin-4-yl-3-[[(3R)-1-[(E)-4-pyridin-2-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound N1=C(C=CC=C1)C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 HBGNPOUHAAQMOP-SQZHUTIHSA-N 0.000 description 2
- YTPGUWKRRGOWGM-PEAOBZDDSA-N 5-pyridin-4-yl-3-[[(3R)-1-[(E)-4-pyrrolidin-1-ylbut-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound N1(CCCC1)C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 YTPGUWKRRGOWGM-PEAOBZDDSA-N 0.000 description 2
- UIDJYJYUNSRPAE-GNAXCBTBSA-N 5-pyridin-4-yl-3-[[(3R)-1-[(E)-4-pyrrolidin-1-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound N1(CCCC1)C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 UIDJYJYUNSRPAE-GNAXCBTBSA-N 0.000 description 2
- OQPRQNRZYZYMAV-ZZXKWVIFSA-N 5-pyridin-4-yl-3-[[1-[(E)-4-pyridin-2-ylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound N1=C(C=CC=C1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 OQPRQNRZYZYMAV-ZZXKWVIFSA-N 0.000 description 2
- HDHAQVCBUSXQRR-ONEGZZNKSA-N 5-pyridin-4-yl-3-[[1-[(E)-4-pyrrolidin-1-ylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound N1(CCCC1)C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1 HDHAQVCBUSXQRR-ONEGZZNKSA-N 0.000 description 2
- 208000030507 AIDS Diseases 0.000 description 2
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 2
- 208000035285 Allergic Seasonal Rhinitis Diseases 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- 235000003911 Arachis Nutrition 0.000 description 2
- 244000105624 Arachis hypogaea Species 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 241000416162 Astragalus gummifer Species 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 2
- 241000283690 Bos taurus Species 0.000 description 2
- 208000003174 Brain Neoplasms Diseases 0.000 description 2
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 2
- ADZPNBKSYLSLGU-NSCUHMNNSA-N C/C=C/C(=O)N1CC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C/C=C/C(=O)N1CC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 ADZPNBKSYLSLGU-NSCUHMNNSA-N 0.000 description 2
- XVCKYZOWVZGFTD-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 XVCKYZOWVZGFTD-UHFFFAOYSA-N 0.000 description 2
- HRPLFOFZBFEDLT-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 HRPLFOFZBFEDLT-UHFFFAOYSA-N 0.000 description 2
- UVMTWVUNXUALFB-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 UVMTWVUNXUALFB-UHFFFAOYSA-N 0.000 description 2
- HFAHDIYHDJYSNL-SNAWJCMRSA-N CC(C)NC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 HFAHDIYHDJYSNL-SNAWJCMRSA-N 0.000 description 2
- CJEZEVLRJLJDKI-ONEGZZNKSA-N CC(C)NC/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)CC1 Chemical compound CC(C)NC/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)CC1 CJEZEVLRJLJDKI-ONEGZZNKSA-N 0.000 description 2
- MOSXJBRUOYXWKQ-ZZXKWVIFSA-N CC(C)NC/C=C/C(=O)N1CCCC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CCCC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 MOSXJBRUOYXWKQ-ZZXKWVIFSA-N 0.000 description 2
- UOFGSVCJJQACRX-AATRIKPKSA-N CC/C=C/C(=O)N1CC(NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound CC/C=C/C(=O)N1CC(NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 UOFGSVCJJQACRX-AATRIKPKSA-N 0.000 description 2
- AYGUTIORNMLTPU-UHFFFAOYSA-N CC1=C(C(C)C)C=CC(NC(=O)C2=NC=CC(C3=CNC(=O)C(NC4CNC4)=C3)=C2)=C1.Cl Chemical compound CC1=C(C(C)C)C=CC(NC(=O)C2=NC=CC(C3=CNC(=O)C(NC4CNC4)=C3)=C2)=C1.Cl AYGUTIORNMLTPU-UHFFFAOYSA-N 0.000 description 2
- HPWQNXKNXVOONF-ONEGZZNKSA-N CCC(=O)/C=C/C(=O)N1CC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CCC(=O)/C=C/C(=O)N1CC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 HPWQNXKNXVOONF-ONEGZZNKSA-N 0.000 description 2
- PYYVCRULNSPNTK-SNAWJCMRSA-N CCC/C=C/C(=O)N1CC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CCC/C=C/C(=O)N1CC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 PYYVCRULNSPNTK-SNAWJCMRSA-N 0.000 description 2
- XRVXOTBCZADETH-UXBLZVDNSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4=CC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4=CC=CC=C4)C3)=C2)=C1 XRVXOTBCZADETH-UXBLZVDNSA-N 0.000 description 2
- COMKNHBQNTUYBY-UXBLZVDNSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CCOC4=CC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CCOC4=CC=CC=C4)C3)=C2)=C1 COMKNHBQNTUYBY-UXBLZVDNSA-N 0.000 description 2
- WHJOFLRLICYILH-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 WHJOFLRLICYILH-AATRIKPKSA-N 0.000 description 2
- GBCRSLLHZVZJPM-DUMMIQTOSA-N CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC)C1 GBCRSLLHZVZJPM-DUMMIQTOSA-N 0.000 description 2
- SAKIVMHOSKKLQL-MEQKGCOVSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC)C1 SAKIVMHOSKKLQL-MEQKGCOVSA-N 0.000 description 2
- MMROEEAXOCVHIG-YJJPMGAVSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC(C)C)C1 MMROEEAXOCVHIG-YJJPMGAVSA-N 0.000 description 2
- TXMDQHDHROTOIA-SNAWJCMRSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC)CC1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC)CC1 TXMDQHDHROTOIA-SNAWJCMRSA-N 0.000 description 2
- MENCGNLUYWOUSX-RRFGBZISSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC(C)C)C1 MENCGNLUYWOUSX-RRFGBZISSA-N 0.000 description 2
- MAWBQZXYYSHCEA-SNAWJCMRSA-N CN(C)C/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 MAWBQZXYYSHCEA-SNAWJCMRSA-N 0.000 description 2
- FLUQWRLIRDDPAW-LHXJMTLCSA-N CN(C)C/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 FLUQWRLIRDDPAW-LHXJMTLCSA-N 0.000 description 2
- TXMGGIZWFPTIRT-ZOXCZHJPSA-N CNC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 TXMGGIZWFPTIRT-ZOXCZHJPSA-N 0.000 description 2
- HZDWZQYAHXIILK-YADGQWAMSA-N CNC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 HZDWZQYAHXIILK-YADGQWAMSA-N 0.000 description 2
- KWBGYXLKROKTNH-SNAWJCMRSA-N COC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 KWBGYXLKROKTNH-SNAWJCMRSA-N 0.000 description 2
- MLTYLTOOSXNUGL-AATRIKPKSA-N COC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound COC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 MLTYLTOOSXNUGL-AATRIKPKSA-N 0.000 description 2
- CYJAMLRZLCZVQA-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 CYJAMLRZLCZVQA-SNAWJCMRSA-N 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 description 2
- WBYWAXJHAXSJNI-SREVYHEPSA-N Cinnamic acid Chemical compound OC(=O)\C=C/C1=CC=CC=C1 WBYWAXJHAXSJNI-SREVYHEPSA-N 0.000 description 2
- WZGFRCAAYPMHHG-CYBMUJFWSA-N Cl.O=C1NC=C(C2=CC=NC=C2)C=C1N[C@@H]1CCCNC1 Chemical compound Cl.O=C1NC=C(C2=CC=NC=C2)C=C1N[C@@H]1CCCNC1 WZGFRCAAYPMHHG-CYBMUJFWSA-N 0.000 description 2
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 108010092160 Dactinomycin Proteins 0.000 description 2
- ZBNZXTGUTAYRHI-UHFFFAOYSA-N Dasatinib Chemical compound C=1C(N2CCN(CCO)CC2)=NC(C)=NC=1NC(S1)=NC=C1C(=O)NC1=C(C)C=CC=C1Cl ZBNZXTGUTAYRHI-UHFFFAOYSA-N 0.000 description 2
- 206010012438 Dermatitis atopic Diseases 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- 206010014950 Eosinophilia Diseases 0.000 description 2
- 241000283086 Equidae Species 0.000 description 2
- 108010008165 Etanercept Proteins 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- 229910052693 Europium Inorganic materials 0.000 description 2
- 208000009386 Experimental Arthritis Diseases 0.000 description 2
- 108010007457 Extracellular Signal-Regulated MAP Kinases Proteins 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- 208000009329 Graft vs Host Disease Diseases 0.000 description 2
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 2
- 101000818543 Homo sapiens Tyrosine-protein kinase ZAP-70 Proteins 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 102000051628 Interleukin-1 receptor antagonist Human genes 0.000 description 2
- 108700021006 Interleukin-1 receptor antagonist Proteins 0.000 description 2
- 102000004388 Interleukin-4 Human genes 0.000 description 2
- 108090000978 Interleukin-4 Proteins 0.000 description 2
- 208000003456 Juvenile Arthritis Diseases 0.000 description 2
- 206010059176 Juvenile idiopathic arthritis Diseases 0.000 description 2
- 239000005517 L01XE01 - Imatinib Substances 0.000 description 2
- 239000005551 L01XE03 - Erlotinib Substances 0.000 description 2
- 239000002147 L01XE04 - Sunitinib Substances 0.000 description 2
- 239000002067 L01XE06 - Dasatinib Substances 0.000 description 2
- 239000005536 L01XE08 - Nilotinib Substances 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- TWRXJAOTZQYOKJ-UHFFFAOYSA-L Magnesium chloride Chemical compound [Mg+2].[Cl-].[Cl-] TWRXJAOTZQYOKJ-UHFFFAOYSA-L 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- FQISKWAFAHGMGT-SGJOWKDISA-M Methylprednisolone sodium succinate Chemical compound [Na+].C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2[C@@H](O)C[C@]2(C)[C@@](O)(C(=O)COC(=O)CCC([O-])=O)CC[C@H]21 FQISKWAFAHGMGT-SGJOWKDISA-M 0.000 description 2
- 102100024193 Mitogen-activated protein kinase 1 Human genes 0.000 description 2
- JLTDJTHDQAWBAV-UHFFFAOYSA-N N,N-dimethylaniline Chemical compound CN(C)C1=CC=CC=C1 JLTDJTHDQAWBAV-UHFFFAOYSA-N 0.000 description 2
- BLXXJMDCKKHMKV-UHFFFAOYSA-N Nabumetone Chemical compound C1=C(CCC(C)=O)C=CC2=CC(OC)=CC=C21 BLXXJMDCKKHMKV-UHFFFAOYSA-N 0.000 description 2
- 108020004485 Nonsense Codon Proteins 0.000 description 2
- 206010030113 Oedema Diseases 0.000 description 2
- 241000283973 Oryctolagus cuniculus Species 0.000 description 2
- 229930012538 Paclitaxel Natural products 0.000 description 2
- 208000002193 Pain Diseases 0.000 description 2
- NFHFRUOZVGFOOS-UHFFFAOYSA-N Pd(PPh3)4 Substances [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 2
- 241000721454 Pemphigus Species 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 2
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 2
- 208000036284 Rhinitis seasonal Diseases 0.000 description 2
- 102000000395 SH3 domains Human genes 0.000 description 2
- 108050008861 SH3 domains Proteins 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- 238000006069 Suzuki reaction reaction Methods 0.000 description 2
- 206010042971 T-cell lymphoma Diseases 0.000 description 2
- 208000027585 T-cell non-Hodgkin lymphoma Diseases 0.000 description 2
- 241000534944 Thia Species 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 229920001615 Tragacanth Polymers 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 102100021125 Tyrosine-protein kinase ZAP-70 Human genes 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 2
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 125000004442 acylamino group Chemical group 0.000 description 2
- ORILYTVJVMAKLC-UHFFFAOYSA-N adamantane Chemical compound C1C(C2)CC3CC1CC2C3 ORILYTVJVMAKLC-UHFFFAOYSA-N 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 125000004450 alkenylene group Chemical group 0.000 description 2
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 2
- 150000005215 alkyl ethers Chemical group 0.000 description 2
- 125000004414 alkyl thio group Chemical group 0.000 description 2
- 125000002947 alkylene group Chemical group 0.000 description 2
- 125000004419 alkynylene group Chemical group 0.000 description 2
- 230000007815 allergy Effects 0.000 description 2
- 150000001450 anions Chemical class 0.000 description 2
- 230000000340 anti-metabolite Effects 0.000 description 2
- 239000000427 antigen Substances 0.000 description 2
- 210000000612 antigen-presenting cell Anatomy 0.000 description 2
- 108091007433 antigens Proteins 0.000 description 2
- 102000036639 antigens Human genes 0.000 description 2
- 229940100197 antimetabolite Drugs 0.000 description 2
- 239000002256 antimetabolite Substances 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 125000003435 aroyl group Chemical group 0.000 description 2
- 125000003710 aryl alkyl group Chemical group 0.000 description 2
- 125000004104 aryloxy group Chemical group 0.000 description 2
- 201000008937 atopic dermatitis Diseases 0.000 description 2
- 230000001363 autoimmune Effects 0.000 description 2
- RITAVMQDGBJQJZ-FMIVXFBMSA-N axitinib Chemical compound CNC(=O)C1=CC=CC=C1SC1=CC=C(C(\C=C\C=2N=CC=CC=2)=NN2)C2=C1 RITAVMQDGBJQJZ-FMIVXFBMSA-N 0.000 description 2
- LMEKQMALGUDUQG-UHFFFAOYSA-N azathioprine Chemical compound CN1C=NC([N+]([O-])=O)=C1SC1=NC=NC2=C1NC=N2 LMEKQMALGUDUQG-UHFFFAOYSA-N 0.000 description 2
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 2
- 125000004619 benzopyranyl group Chemical group O1C(C=CC2=C1C=CC=C2)* 0.000 description 2
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 230000033228 biological regulation Effects 0.000 description 2
- IPWKHHSGDUIRAH-UHFFFAOYSA-N bis(pinacolato)diboron Chemical compound O1C(C)(C)C(C)(C)OB1B1OC(C)(C)C(C)(C)O1 IPWKHHSGDUIRAH-UHFFFAOYSA-N 0.000 description 2
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 2
- 150000001649 bromium compounds Chemical class 0.000 description 2
- 125000002837 carbocyclic group Chemical group 0.000 description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 2
- 229960004562 carboplatin Drugs 0.000 description 2
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 2
- 239000004359 castor oil Substances 0.000 description 2
- 235000019438 castor oil Nutrition 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 230000024245 cell differentiation Effects 0.000 description 2
- 230000003915 cell function Effects 0.000 description 2
- 210000000170 cell membrane Anatomy 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical compound C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 description 2
- 230000001684 chronic effect Effects 0.000 description 2
- 208000037976 chronic inflammation Diseases 0.000 description 2
- 229960001265 ciclosporin Drugs 0.000 description 2
- 235000013985 cinnamic acid Nutrition 0.000 description 2
- 229930016911 cinnamic acid Natural products 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 239000002131 composite material Substances 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 229960004397 cyclophosphamide Drugs 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 229930182912 cyclosporin Natural products 0.000 description 2
- 230000016396 cytokine production Effects 0.000 description 2
- 229960002448 dasatinib Drugs 0.000 description 2
- NNBZCPXTIHJBJL-UHFFFAOYSA-N decalin Chemical compound C1CCCC2CCCCC21 NNBZCPXTIHJBJL-UHFFFAOYSA-N 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- KXZOIWWTXOCYKR-UHFFFAOYSA-M diclofenac potassium Chemical compound [K+].[O-]C(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl KXZOIWWTXOCYKR-UHFFFAOYSA-M 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- 125000004982 dihaloalkyl group Chemical group 0.000 description 2
- 239000002270 dispersing agent Substances 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 239000008298 dragée Substances 0.000 description 2
- 239000000890 drug combination Substances 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 231100000321 erythema Toxicity 0.000 description 2
- OGPBJKLSAFTDLK-UHFFFAOYSA-N europium atom Chemical compound [Eu] OGPBJKLSAFTDLK-UHFFFAOYSA-N 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 239000010685 fatty oil Substances 0.000 description 2
- 238000002866 fluorescence resonance energy transfer Methods 0.000 description 2
- 229960002949 fluorouracil Drugs 0.000 description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 2
- 239000012458 free base Substances 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 239000000417 fungicide Substances 0.000 description 2
- 125000002541 furyl group Chemical group 0.000 description 2
- 239000003349 gelling agent Substances 0.000 description 2
- 229940080856 gleevec Drugs 0.000 description 2
- 208000024908 graft versus host disease Diseases 0.000 description 2
- 125000004438 haloalkoxy group Chemical group 0.000 description 2
- 150000002367 halogens Chemical class 0.000 description 2
- 210000002443 helper t lymphocyte Anatomy 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 2
- 206010020718 hyperplasia Diseases 0.000 description 2
- 230000003053 immunization Effects 0.000 description 2
- 238000002649 immunization Methods 0.000 description 2
- 238000003119 immunoblot Methods 0.000 description 2
- 230000002779 inactivation Effects 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 229960000905 indomethacin Drugs 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 230000001939 inductive effect Effects 0.000 description 2
- 208000015181 infectious disease Diseases 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 238000007912 intraperitoneal administration Methods 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 150000004694 iodide salts Chemical class 0.000 description 2
- GURKHSYORGJETM-WAQYZQTGSA-N irinotecan hydrochloride (anhydrous) Chemical compound Cl.C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 GURKHSYORGJETM-WAQYZQTGSA-N 0.000 description 2
- SUMDYPCJJOFFON-UHFFFAOYSA-N isethionic acid Chemical compound OCCS(O)(=O)=O SUMDYPCJJOFFON-UHFFFAOYSA-N 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- 201000002215 juvenile rheumatoid arthritis Diseases 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- VHOGYURTWQBHIL-UHFFFAOYSA-N leflunomide Chemical compound O1N=CC(C(=O)NC=2C=CC(=CC=2)C(F)(F)F)=C1C VHOGYURTWQBHIL-UHFFFAOYSA-N 0.000 description 2
- 239000000865 liniment Substances 0.000 description 2
- 229940057995 liquid paraffin Drugs 0.000 description 2
- 239000007937 lozenge Substances 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 208000020816 lung neoplasm Diseases 0.000 description 2
- 206010025135 lupus erythematosus Diseases 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 2
- 230000003211 malignant effect Effects 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- HYYBABOKPJLUIN-UHFFFAOYSA-N mefenamic acid Chemical compound CC1=CC=CC(NC=2C(=CC=CC=2)C(O)=O)=C1C HYYBABOKPJLUIN-UHFFFAOYSA-N 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 2
- 229960000485 methotrexate Drugs 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 229920000609 methyl cellulose Polymers 0.000 description 2
- WBYWAXJHAXSJNI-UHFFFAOYSA-N methyl p-hydroxycinnamate Natural products OC(=O)C=CC1=CC=CC=C1 WBYWAXJHAXSJNI-UHFFFAOYSA-N 0.000 description 2
- 239000001923 methylcellulose Substances 0.000 description 2
- 230000003278 mimic effect Effects 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 125000002757 morpholinyl group Chemical group 0.000 description 2
- 238000000465 moulding Methods 0.000 description 2
- 201000005962 mycosis fungoides Diseases 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- HHZIURLSWUIHRB-UHFFFAOYSA-N nilotinib Chemical compound C1=NC(C)=CN1C1=CC(NC(=O)C=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)=CC(C(F)(F)F)=C1 HHZIURLSWUIHRB-UHFFFAOYSA-N 0.000 description 2
- 229960001346 nilotinib Drugs 0.000 description 2
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 2
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 description 2
- 201000008968 osteosarcoma Diseases 0.000 description 2
- DWAFYCQODLXJNR-BNTLRKBRSA-L oxaliplatin Chemical compound O1C(=O)C(=O)O[Pt]11N[C@@H]2CCCC[C@H]2N1 DWAFYCQODLXJNR-BNTLRKBRSA-L 0.000 description 2
- 229960001756 oxaliplatin Drugs 0.000 description 2
- OFPXSFXSNFPTHF-UHFFFAOYSA-N oxaprozin Chemical compound O1C(CCC(=O)O)=NC(C=2C=CC=CC=2)=C1C1=CC=CC=C1 OFPXSFXSNFPTHF-UHFFFAOYSA-N 0.000 description 2
- 229960001592 paclitaxel Drugs 0.000 description 2
- 230000036407 pain Effects 0.000 description 2
- 238000007911 parenteral administration Methods 0.000 description 2
- 235000010603 pastilles Nutrition 0.000 description 2
- 230000037361 pathway Effects 0.000 description 2
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 2
- 239000006187 pill Substances 0.000 description 2
- 125000004193 piperazinyl group Chemical group 0.000 description 2
- 125000003386 piperidinyl group Chemical group 0.000 description 2
- QYSPLQLAKJAUJT-UHFFFAOYSA-N piroxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=CC=CC=N1 QYSPLQLAKJAUJT-UHFFFAOYSA-N 0.000 description 2
- 125000006684 polyhaloalkyl group Chemical group 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 230000002335 preservative effect Effects 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000002062 proliferating effect Effects 0.000 description 2
- 230000009696 proliferative response Effects 0.000 description 2
- 238000011321 prophylaxis Methods 0.000 description 2
- 150000005299 pyridinones Chemical class 0.000 description 2
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 2
- 229940061969 rheumatrex Drugs 0.000 description 2
- 229960004641 rituximab Drugs 0.000 description 2
- 201000000849 skin cancer Diseases 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- 235000009518 sodium iodide Nutrition 0.000 description 2
- 235000011152 sodium sulphate Nutrition 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 238000013222 sprague-dawley male rat Methods 0.000 description 2
- 206010041823 squamous cell carcinoma Diseases 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000011550 stock solution Substances 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 description 2
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 238000007910 systemic administration Methods 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- 229940120982 tarceva Drugs 0.000 description 2
- VYFVLGUZBKRPMT-GFCCVEGCSA-N tert-butyl (3R)-3-[(5-bromo-2-methoxypyridin-3-yl)amino]piperidine-1-carboxylate Chemical compound BrC=1C=C(C(=NC1)OC)N[C@H]1CN(CCC1)C(=O)OC(C)(C)C VYFVLGUZBKRPMT-GFCCVEGCSA-N 0.000 description 2
- 150000003573 thiols Chemical class 0.000 description 2
- 230000036962 time dependent Effects 0.000 description 2
- WYWHKKSPHMUBEB-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 2
- UPSPUYADGBWSHF-UHFFFAOYSA-N tolmetin Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=C(CC(O)=O)N1C UPSPUYADGBWSHF-UHFFFAOYSA-N 0.000 description 2
- 235000010487 tragacanth Nutrition 0.000 description 2
- 239000000196 tragacanth Substances 0.000 description 2
- 229940116362 tragacanth Drugs 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- 102000003390 tumor necrosis factor Human genes 0.000 description 2
- CXNIUSPIQKWYAI-UHFFFAOYSA-N xantphos Chemical compound C=12OC3=C(P(C=4C=CC=CC=4)C=4C=CC=CC=4)C=CC=C3C(C)(C)C2=CC=CC=1P(C=1C=CC=CC=1)C1=CC=CC=C1 CXNIUSPIQKWYAI-UHFFFAOYSA-N 0.000 description 2
- 239000011592 zinc chloride Substances 0.000 description 2
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 2
- LSPHULWDVZXLIL-UHFFFAOYSA-N (+/-)-Camphoric acid Chemical compound CC1(C)C(C(O)=O)CCC1(C)C(O)=O LSPHULWDVZXLIL-UHFFFAOYSA-N 0.000 description 1
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 1
- RDJGLLICXDHJDY-NSHDSACASA-N (2s)-2-(3-phenoxyphenyl)propanoic acid Chemical compound OC(=O)[C@@H](C)C1=CC=CC(OC=2C=CC=CC=2)=C1 RDJGLLICXDHJDY-NSHDSACASA-N 0.000 description 1
- HMLGSIZOMSVISS-ONJSNURVSA-N (7r)-7-[[(2z)-2-(2-amino-1,3-thiazol-4-yl)-2-(2,2-dimethylpropanoyloxymethoxyimino)acetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid Chemical compound N([C@@H]1C(N2C(=C(C=C)CSC21)C(O)=O)=O)C(=O)\C(=N/OCOC(=O)C(C)(C)C)C1=CSC(N)=N1 HMLGSIZOMSVISS-ONJSNURVSA-N 0.000 description 1
- MWWSFMDVAYGXBV-MYPASOLCSA-N (7r,9s)-7-[(2r,4s,5s,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;hydrochloride Chemical compound Cl.O([C@@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 MWWSFMDVAYGXBV-MYPASOLCSA-N 0.000 description 1
- FPVKHBSQESCIEP-UHFFFAOYSA-N (8S)-3-(2-deoxy-beta-D-erythro-pentofuranosyl)-3,6,7,8-tetrahydroimidazo[4,5-d][1,3]diazepin-8-ol Natural products C1C(O)C(CO)OC1N1C(NC=NCC2O)=C2N=C1 FPVKHBSQESCIEP-UHFFFAOYSA-N 0.000 description 1
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- AFTUPMYMAYLZDP-UHFFFAOYSA-N (E)-2-[3-[(2-oxo-5-pyridin-4-yl-1H-pyridin-3-yl)amino]azetidine-1-carbonyl]but-2-enenitrile Chemical compound O=C1C(=CC(=CN1)C1=CC=NC=C1)NC1CN(C1)C(=O)C(C#N)=CC AFTUPMYMAYLZDP-UHFFFAOYSA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- DSSYKIVIOFKYAU-XCBNKYQSSA-N (R)-camphor Chemical compound C1C[C@@]2(C)C(=O)C[C@@H]1C2(C)C DSSYKIVIOFKYAU-XCBNKYQSSA-N 0.000 description 1
- NNYBQONXHNTVIJ-QGZVFWFLSA-N (R)-etodolac Chemical compound C1CO[C@](CC)(CC(O)=O)C2=C1C(C=CC=C1CC)=C1N2 NNYBQONXHNTVIJ-QGZVFWFLSA-N 0.000 description 1
- SYTBZMRGLBWNTM-SNVBAGLBSA-N (R)-flurbiprofen Chemical compound FC1=CC([C@H](C(O)=O)C)=CC=C1C1=CC=CC=C1 SYTBZMRGLBWNTM-SNVBAGLBSA-N 0.000 description 1
- IWYDHOAUDWTVEP-ZETCQYMHSA-N (S)-mandelic acid Chemical compound OC(=O)[C@@H](O)C1=CC=CC=C1 IWYDHOAUDWTVEP-ZETCQYMHSA-N 0.000 description 1
- BAHCTQOTXWPKEC-UHFFFAOYSA-N (Z)-4,4-dimethyl-2-[3-[(2-oxo-5-pyridin-4-yl-1H-pyridin-3-yl)amino]azetidine-1-carbonyl]pent-2-enenitrile Chemical compound CC(C=C(C#N)/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC=NC=C1)(C)C BAHCTQOTXWPKEC-UHFFFAOYSA-N 0.000 description 1
- ITGIYLMMAABTHC-ONEGZZNKSA-N (e)-4-(dimethylazaniumyl)but-2-enoate Chemical group CN(C)C\C=C\C(O)=O ITGIYLMMAABTHC-ONEGZZNKSA-N 0.000 description 1
- SCBUQIQXUBOQAI-XBXARRHUSA-N (e)-4-phenylbut-2-enoic acid Chemical group OC(=O)\C=C\CC1=CC=CC=C1 SCBUQIQXUBOQAI-XBXARRHUSA-N 0.000 description 1
- CJMACVBYYPIIDB-XBXARRHUSA-N (e)-5-phenoxypent-2-enoic acid Chemical group OC(=O)\C=C\CCOC1=CC=CC=C1 CJMACVBYYPIIDB-XBXARRHUSA-N 0.000 description 1
- RJUIDDKTATZJFE-NSCUHMNNSA-N (e)-but-2-enoyl chloride Chemical compound C\C=C\C(Cl)=O RJUIDDKTATZJFE-NSCUHMNNSA-N 0.000 description 1
- POPHMOPNVVKGRW-UHFFFAOYSA-N 1,2,3,4,4a,5,6,7-octahydronaphthalene Chemical compound C1CCC2CCCCC2=C1 POPHMOPNVVKGRW-UHFFFAOYSA-N 0.000 description 1
- DDMOUSALMHHKOS-UHFFFAOYSA-N 1,2-dichloro-1,1,2,2-tetrafluoroethane Chemical compound FC(F)(Cl)C(F)(F)Cl DDMOUSALMHHKOS-UHFFFAOYSA-N 0.000 description 1
- 125000005871 1,3-benzodioxolyl group Chemical group 0.000 description 1
- IGERFAHWSHDDHX-UHFFFAOYSA-N 1,3-dioxanyl Chemical group [CH]1OCCCO1 IGERFAHWSHDDHX-UHFFFAOYSA-N 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- 125000005940 1,4-dioxanyl group Chemical group 0.000 description 1
- XWPCYYOZOJKYKQ-UHFFFAOYSA-N 1-(2-chloroethyl)-3-[2-[2-[[2-chloroethyl(nitroso)carbamoyl]amino]ethyldisulfanyl]ethyl]-1-nitrosourea Chemical compound ClCCN(N=O)C(=O)NCCSSCCNC(=O)N(N=O)CCCl XWPCYYOZOJKYKQ-UHFFFAOYSA-N 0.000 description 1
- FQCJGOFQLJELIV-UHFFFAOYSA-N 1-(azetidin-1-yl)prop-2-en-1-one Chemical compound C=CC(=O)N1CCC1 FQCJGOFQLJELIV-UHFFFAOYSA-N 0.000 description 1
- VFWCMGCRMGJXDK-UHFFFAOYSA-N 1-chlorobutane Chemical class CCCCCl VFWCMGCRMGJXDK-UHFFFAOYSA-N 0.000 description 1
- DVVGIUUJYPYENY-UHFFFAOYSA-N 1-methylpyridin-2-one Chemical class CN1C=CC=CC1=O DVVGIUUJYPYENY-UHFFFAOYSA-N 0.000 description 1
- RESPXSHDJQUNTN-UHFFFAOYSA-N 1-piperidin-1-ylprop-2-en-1-one Chemical compound C=CC(=O)N1CCCCC1 RESPXSHDJQUNTN-UHFFFAOYSA-N 0.000 description 1
- JUSXUZQNQSDHDZ-UHFFFAOYSA-N 1-pyrazin-2-ylindole Chemical group C1=CC2=CC=CC=C2N1C1=CN=CC=N1 JUSXUZQNQSDHDZ-UHFFFAOYSA-N 0.000 description 1
- ZRLHATCIJLNNQH-UHFFFAOYSA-N 1-pyrazin-2-ylindole 1H-pyridin-2-one Chemical group O=c1cccc[nH]1.c1cc2ccccc2n1-c1cnccn1 ZRLHATCIJLNNQH-UHFFFAOYSA-N 0.000 description 1
- FUFLCEKSBBHCMO-UHFFFAOYSA-N 11-dehydrocorticosterone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)C(=O)CO)C4C3CCC2=C1 FUFLCEKSBBHCMO-UHFFFAOYSA-N 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- LXFQSRIDYRFTJW-UHFFFAOYSA-M 2,4,6-trimethylbenzenesulfonate Chemical compound CC1=CC(C)=C(S([O-])(=O)=O)C(C)=C1 LXFQSRIDYRFTJW-UHFFFAOYSA-M 0.000 description 1
- JZQQELXRNYHTJQ-UHFFFAOYSA-N 2-(3-methyl-4-propan-2-ylphenyl)benzamide Chemical compound C1=C(C)C(C(C)C)=CC=C1C1=CC=CC=C1C(N)=O JZQQELXRNYHTJQ-UHFFFAOYSA-N 0.000 description 1
- RLBJHMPTORQXGN-UHFFFAOYSA-N 2-[3-[(2-oxo-5-pyridin-4-yl-1H-pyridin-3-yl)amino]azetidine-1-carbonyl]prop-2-enenitrile Chemical compound O=C1C(=CC(=CN1)C1=CC=NC=C1)NC1CN(C1)C(=O)C(C#N)=C RLBJHMPTORQXGN-UHFFFAOYSA-N 0.000 description 1
- GMVQXUSUAGPFHN-UHFFFAOYSA-N 2-[3-[[5-[2-(methylamino)pyridin-4-yl]-2-oxo-1H-pyridin-3-yl]amino]azetidine-1-carbonyl]prop-2-enenitrile Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(=O)C(C#N)=C)=O GMVQXUSUAGPFHN-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- PCLMNCBIXQQRMB-UHFFFAOYSA-N 2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC=NC(F)=C1 PCLMNCBIXQQRMB-UHFFFAOYSA-N 0.000 description 1
- IWTFOFMTUOBLHG-UHFFFAOYSA-N 2-methoxypyridine Chemical compound COC1=CC=CC=N1 IWTFOFMTUOBLHG-UHFFFAOYSA-N 0.000 description 1
- 229940080296 2-naphthalenesulfonate Drugs 0.000 description 1
- WMPPDTMATNBGJN-UHFFFAOYSA-N 2-phenylethylbromide Chemical class BrCCC1=CC=CC=C1 WMPPDTMATNBGJN-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- MEZTZAJUINFCSJ-BQYQJAHWSA-N 3-[5-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-6-oxo-1H-pyridin-3-yl]-5-methyl-N-(3-methyl-4-propan-2-ylphenyl)benzamide Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=C(C1)C)C MEZTZAJUINFCSJ-BQYQJAHWSA-N 0.000 description 1
- WEVYNIUIFUYDGI-UHFFFAOYSA-N 3-[6-[4-(trifluoromethoxy)anilino]-4-pyrimidinyl]benzamide Chemical compound NC(=O)C1=CC=CC(C=2N=CN=C(NC=3C=CC(OC(F)(F)F)=CC=3)C=2)=C1 WEVYNIUIFUYDGI-UHFFFAOYSA-N 0.000 description 1
- MRGRNDAMHARQFS-CYBMUJFWSA-N 3-[[(3R)-1-(2-chloroacetyl)pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound ClCC(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC MRGRNDAMHARQFS-CYBMUJFWSA-N 0.000 description 1
- CPGZRXIPDSUTDT-FHVRUZERSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC)C CPGZRXIPDSUTDT-FHVRUZERSA-N 0.000 description 1
- QLDZZQLYVNJDOS-FHVRUZERSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC)C QLDZZQLYVNJDOS-FHVRUZERSA-N 0.000 description 1
- SRGUZQVFEOFWCX-CPSHHHPTSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC)C SRGUZQVFEOFWCX-CPSHHHPTSA-N 0.000 description 1
- ANFCYMXXUIJGFX-SUQGKYDKSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C ANFCYMXXUIJGFX-SUQGKYDKSA-N 0.000 description 1
- PADBZXNZUOKAFR-SUQGKYDKSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C PADBZXNZUOKAFR-SUQGKYDKSA-N 0.000 description 1
- OLQROOMEFRNAJZ-OVKVCFKUSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC)C OLQROOMEFRNAJZ-OVKVCFKUSA-N 0.000 description 1
- DRTWZSGCELXEIN-OVKVCFKUSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC)C DRTWZSGCELXEIN-OVKVCFKUSA-N 0.000 description 1
- VQCKTSWGABIXJP-ZMBJWTFHSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC)C VQCKTSWGABIXJP-ZMBJWTFHSA-N 0.000 description 1
- RCWYPQSCTGGQRZ-LAQIPUCWSA-N 3-[[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C RCWYPQSCTGGQRZ-LAQIPUCWSA-N 0.000 description 1
- QNWHLVSASRSMBO-QGZVFWFLSA-N 3-[[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CCC1)C(C=CCNC)=O)=O QNWHLVSASRSMBO-QGZVFWFLSA-N 0.000 description 1
- SLAQSWULXMSNPF-MRXNPFEDSA-N 3-[[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCNC)=O)=O SLAQSWULXMSNPF-MRXNPFEDSA-N 0.000 description 1
- MOSXJBRUOYXWKQ-MTGFJVSHSA-N 3-[[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 MOSXJBRUOYXWKQ-MTGFJVSHSA-N 0.000 description 1
- HVPVJWYHLVBICY-OAJJDEHYSA-N 3-[[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 HVPVJWYHLVBICY-OAJJDEHYSA-N 0.000 description 1
- RLMKVWBCCDDVCU-XQASTXFUSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 RLMKVWBCCDDVCU-XQASTXFUSA-N 0.000 description 1
- DLZZTTWKWCJRPM-WEBJUUICSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC DLZZTTWKWCJRPM-WEBJUUICSA-N 0.000 description 1
- VTDAPTNNCBLVBR-OUGHUQCRSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC VTDAPTNNCBLVBR-OUGHUQCRSA-N 0.000 description 1
- AJDJLYKDINFYNH-OUGHUQCRSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC AJDJLYKDINFYNH-OUGHUQCRSA-N 0.000 description 1
- CVTYLKPWHIKMBD-XYBNCVKDSA-N 3-[[(3R)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC CVTYLKPWHIKMBD-XYBNCVKDSA-N 0.000 description 1
- VMYKGGGCGYONOQ-MRXNPFEDSA-N 3-[[(3R)-1-[(E)-but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=CC)(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC VMYKGGGCGYONOQ-MRXNPFEDSA-N 0.000 description 1
- JRBKYFMFRYBOCJ-OAHLLOKOSA-N 3-[[(3R)-1-[(E)-but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=CC)(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC JRBKYFMFRYBOCJ-OAHLLOKOSA-N 0.000 description 1
- UZUNDECKCZNNOO-OAHLLOKOSA-N 3-[[(3R)-1-[(E)-but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=CC)(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC=NC=C1 UZUNDECKCZNNOO-OAHLLOKOSA-N 0.000 description 1
- SLAQSWULXMSNPF-INIZCTEOSA-N 3-[[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@@H]1CN(CC1)C(C=CCNC)=O)=O SLAQSWULXMSNPF-INIZCTEOSA-N 0.000 description 1
- RLMKVWBCCDDVCU-BFJGLSHQSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 RLMKVWBCCDDVCU-BFJGLSHQSA-N 0.000 description 1
- DLZZTTWKWCJRPM-UKTKYZJWSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]piperidin-3-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC DLZZTTWKWCJRPM-UKTKYZJWSA-N 0.000 description 1
- FSCVPBOTKMWLCE-IDOMTICXSA-N 3-[[(3S)-1-[(E)-4-aminobut-2-enoyl]pyrrolidin-3-yl]-ethylamino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1C[C@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC FSCVPBOTKMWLCE-IDOMTICXSA-N 0.000 description 1
- ZWXHFRQUXSHYMG-AATRIKPKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]-methylamino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C)C ZWXHFRQUXSHYMG-AATRIKPKSA-N 0.000 description 1
- OSQTXJXDQPRSHG-AATRIKPKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]-methylamino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C)C OSQTXJXDQPRSHG-AATRIKPKSA-N 0.000 description 1
- GIXNVKVZVFTMQE-AATRIKPKSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]-methylamino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C)C GIXNVKVZVFTMQE-AATRIKPKSA-N 0.000 description 1
- MZYGKXWSGJYYBD-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C MZYGKXWSGJYYBD-SNAWJCMRSA-N 0.000 description 1
- HCCQHQKGFHFKPY-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[4-(methylamino)pyridin-2-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)NC)C HCCQHQKGFHFKPY-SNAWJCMRSA-N 0.000 description 1
- QPEXNPSQTRIIAL-VOTSOKGWSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC)C QPEXNPSQTRIIAL-VOTSOKGWSA-N 0.000 description 1
- PDZMLDRCVIBTLU-SNAWJCMRSA-N 3-[[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CN(C/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C PDZMLDRCVIBTLU-SNAWJCMRSA-N 0.000 description 1
- LMLZFWNCYJJCST-UHFFFAOYSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCNC)=O)=O LMLZFWNCYJJCST-UHFFFAOYSA-N 0.000 description 1
- MVVBMCUUGWFZAH-ONEGZZNKSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O MVVBMCUUGWFZAH-ONEGZZNKSA-N 0.000 description 1
- DMUVLKWDHLLKKS-ONEGZZNKSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O DMUVLKWDHLLKKS-ONEGZZNKSA-N 0.000 description 1
- BOGZLTRFZLJXFI-ONEGZZNKSA-N 3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound CNC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O BOGZLTRFZLJXFI-ONEGZZNKSA-N 0.000 description 1
- BGEYNUDTMODKHA-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 BGEYNUDTMODKHA-NSCUHMNNSA-N 0.000 description 1
- JRCQAONXDPRXAT-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)CC1CC1 JRCQAONXDPRXAT-NSCUHMNNSA-N 0.000 description 1
- QAGONSSJEKRYQX-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)CC1CC1 QAGONSSJEKRYQX-NSCUHMNNSA-N 0.000 description 1
- KRLBXEPTAQCGQN-OWOJBTEDSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-(cyclopropylmethyl)amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 KRLBXEPTAQCGQN-OWOJBTEDSA-N 0.000 description 1
- FFQWUJUMVBTWPC-ONEGZZNKSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]-methylamino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C FFQWUJUMVBTWPC-ONEGZZNKSA-N 0.000 description 1
- WBGBGUQIZPLLDX-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC WBGBGUQIZPLLDX-NSCUHMNNSA-N 0.000 description 1
- FEXZQSKVRNTPIM-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O FEXZQSKVRNTPIM-NSCUHMNNSA-N 0.000 description 1
- MFCXAKIHGROVAU-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-[4-(methylamino)pyridin-2-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)NC MFCXAKIHGROVAU-NSCUHMNNSA-N 0.000 description 1
- NNLHFXVUUVKDCT-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O NNLHFXVUUVKDCT-NSCUHMNNSA-N 0.000 description 1
- BLEOVHMRXVUZFM-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O BLEOVHMRXVUZFM-NSCUHMNNSA-N 0.000 description 1
- MLTBMKGLXVXDBZ-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]-(cyclopropylmethyl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 MLTBMKGLXVXDBZ-NSCUHMNNSA-N 0.000 description 1
- PECIWSIPSOUNGE-SNAWJCMRSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]-ethylamino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC PECIWSIPSOUNGE-SNAWJCMRSA-N 0.000 description 1
- DWYSAFZJECYOJI-NSCUHMNNSA-N 3-[[1-[(E)-4-aminobut-2-enoyl]piperidin-4-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound NC/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC DWYSAFZJECYOJI-NSCUHMNNSA-N 0.000 description 1
- COCPEVWWFZYINM-ONEGZZNKSA-N 3-[[1-[(E)-4-methoxybut-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound COC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC COCPEVWWFZYINM-ONEGZZNKSA-N 0.000 description 1
- IPKNWXKIFUOACC-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-oxo-1H-pyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(NC=C1)=O)C1CC1 IPKNWXKIFUOACC-UHFFFAOYSA-N 0.000 description 1
- IPMOBBLDWLAUHW-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CC1 IPMOBBLDWLAUHW-UHFFFAOYSA-N 0.000 description 1
- XZWIANXVGBSASZ-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C1CC1 XZWIANXVGBSASZ-UHFFFAOYSA-N 0.000 description 1
- NNNJUFAYNHEXNH-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[4-(methylamino)pyridin-2-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)NC)C1CC1 NNNJUFAYNHEXNH-UHFFFAOYSA-N 0.000 description 1
- FFKNSXZITWMABJ-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C1CC1 FFKNSXZITWMABJ-UHFFFAOYSA-N 0.000 description 1
- AEBCYQSIOIJAEN-UHFFFAOYSA-N 3-[cyclopropyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C1CC1 AEBCYQSIOIJAEN-UHFFFAOYSA-N 0.000 description 1
- MGNNFWUIUYRNQM-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-oxo-1H-pyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(NC=C1)=O)CC1CC1 MGNNFWUIUYRNQM-UHFFFAOYSA-N 0.000 description 1
- VMQVAZJQKWSNTE-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 VMQVAZJQKWSNTE-UHFFFAOYSA-N 0.000 description 1
- XQGZBYVFNCNNMO-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)CC1CC1 XQGZBYVFNCNNMO-UHFFFAOYSA-N 0.000 description 1
- HHMVKDFQEAPNMW-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)CC1CC1 HHMVKDFQEAPNMW-UHFFFAOYSA-N 0.000 description 1
- PYCJDFYWHMGOFE-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)CC1CC1 PYCJDFYWHMGOFE-UHFFFAOYSA-N 0.000 description 1
- LUECWUYHBGETTE-UHFFFAOYSA-N 3-[cyclopropylmethyl-(1-prop-2-enoylpiperidin-4-yl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 LUECWUYHBGETTE-UHFFFAOYSA-N 0.000 description 1
- WGNUYVTUWHEHHZ-JOCHJYFZSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CCC1)C(C=CCN(C)C)=O WGNUYVTUWHEHHZ-JOCHJYFZSA-N 0.000 description 1
- KHPHVHLORBWQMC-JOCHJYFZSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CCC1)C(C=CCN(C)C)=O KHPHVHLORBWQMC-JOCHJYFZSA-N 0.000 description 1
- FLUQWRLIRDDPAW-JOCHJYFZSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCN(C)C)=O FLUQWRLIRDDPAW-JOCHJYFZSA-N 0.000 description 1
- PPBLXZDSVSPYGL-OAQYLSRUSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CC1)C(C=CCN(C)C)=O PPBLXZDSVSPYGL-OAQYLSRUSA-N 0.000 description 1
- VIXNUVNEPWDMEE-OAQYLSRUSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CC1)C(C=CCN(C)C)=O VIXNUVNEPWDMEE-OAQYLSRUSA-N 0.000 description 1
- GNTINEWJCTXGIE-OAQYLSRUSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CC1)C(C=CCN(C)C)=O GNTINEWJCTXGIE-OAQYLSRUSA-N 0.000 description 1
- GLEOCPBHIUMOPF-OAQYLSRUSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CCC1)C(C=CCNC)=O GLEOCPBHIUMOPF-OAQYLSRUSA-N 0.000 description 1
- JJARQXJCVZSCPS-OAQYLSRUSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCNC)=O JJARQXJCVZSCPS-OAQYLSRUSA-N 0.000 description 1
- ZNMJTXPOAOHLFW-HXUWFJFHSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CC1)C(C=CCNC)=O ZNMJTXPOAOHLFW-HXUWFJFHSA-N 0.000 description 1
- HZDWZQYAHXIILK-HXUWFJFHSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CC1)C(C=CCNC)=O HZDWZQYAHXIILK-HXUWFJFHSA-N 0.000 description 1
- TXMGGIZWFPTIRT-HXUWFJFHSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CC1)C(C=CCNC)=O TXMGGIZWFPTIRT-HXUWFJFHSA-N 0.000 description 1
- VCWFFKFAAZCFQM-HSZRJFAPSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CCC1)C(C=CCNC(C)C)=O VCWFFKFAAZCFQM-HSZRJFAPSA-N 0.000 description 1
- MHHBHHCDXROIDD-HSZRJFAPSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CCC1)C(C=CCNC(C)C)=O MHHBHHCDXROIDD-HSZRJFAPSA-N 0.000 description 1
- WBQLHWNABQQXHB-HSZRJFAPSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCNC(C)C)=O WBQLHWNABQQXHB-HSZRJFAPSA-N 0.000 description 1
- YOCKRVKXKBKWFI-JOCHJYFZSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CC1)C(C=CCNC(C)C)=O YOCKRVKXKBKWFI-JOCHJYFZSA-N 0.000 description 1
- MPAOMAZVFRHCKU-JOCHJYFZSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CC1)C(C=CCNC(C)C)=O MPAOMAZVFRHCKU-JOCHJYFZSA-N 0.000 description 1
- BERFKKCFJWJNSG-JOCHJYFZSA-N 3-[cyclopropylmethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CC1)C(C=CCNC(C)C)=O BERFKKCFJWJNSG-JOCHJYFZSA-N 0.000 description 1
- DVPAWHAAUMVFKW-LJQANCHMSA-N 3-[cyclopropylmethyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 DVPAWHAAUMVFKW-LJQANCHMSA-N 0.000 description 1
- OCZKOIDDGGOTFE-LJQANCHMSA-N 3-[cyclopropylmethyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC1CC1 OCZKOIDDGGOTFE-LJQANCHMSA-N 0.000 description 1
- XZHXVQHBBVCDEG-GOSISDBHSA-N 3-[cyclopropylmethyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC1CC1 XZHXVQHBBVCDEG-GOSISDBHSA-N 0.000 description 1
- VBJXXGQZWOPCFE-GOSISDBHSA-N 3-[cyclopropylmethyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 VBJXXGQZWOPCFE-GOSISDBHSA-N 0.000 description 1
- WGNUYVTUWHEHHZ-QFIPXVFZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CCC1)C(C=CCN(C)C)=O WGNUYVTUWHEHHZ-QFIPXVFZSA-N 0.000 description 1
- KHPHVHLORBWQMC-QFIPXVFZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CCC1)C(C=CCN(C)C)=O KHPHVHLORBWQMC-QFIPXVFZSA-N 0.000 description 1
- FLUQWRLIRDDPAW-QFIPXVFZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CCC1)C(C=CCN(C)C)=O FLUQWRLIRDDPAW-QFIPXVFZSA-N 0.000 description 1
- PPBLXZDSVSPYGL-NRFANRHFSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CC1)C(C=CCN(C)C)=O PPBLXZDSVSPYGL-NRFANRHFSA-N 0.000 description 1
- VIXNUVNEPWDMEE-NRFANRHFSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CC1)C(C=CCN(C)C)=O VIXNUVNEPWDMEE-NRFANRHFSA-N 0.000 description 1
- GNTINEWJCTXGIE-NRFANRHFSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(dimethylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CC1)C(C=CCN(C)C)=O GNTINEWJCTXGIE-NRFANRHFSA-N 0.000 description 1
- VGQOVRMWRIWTNC-NRFANRHFSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CCC1)C(C=CCNC)=O VGQOVRMWRIWTNC-NRFANRHFSA-N 0.000 description 1
- GLEOCPBHIUMOPF-NRFANRHFSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CCC1)C(C=CCNC)=O GLEOCPBHIUMOPF-NRFANRHFSA-N 0.000 description 1
- JJARQXJCVZSCPS-NRFANRHFSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CCC1)C(C=CCNC)=O JJARQXJCVZSCPS-NRFANRHFSA-N 0.000 description 1
- ZNMJTXPOAOHLFW-FQEVSTJZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CC1)C(C=CCNC)=O ZNMJTXPOAOHLFW-FQEVSTJZSA-N 0.000 description 1
- HZDWZQYAHXIILK-FQEVSTJZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CC1)C(C=CCNC)=O HZDWZQYAHXIILK-FQEVSTJZSA-N 0.000 description 1
- TXMGGIZWFPTIRT-FQEVSTJZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CC1)C(C=CCNC)=O TXMGGIZWFPTIRT-FQEVSTJZSA-N 0.000 description 1
- VCWFFKFAAZCFQM-QHCPKHFHSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CCC1)C(C=CCNC(C)C)=O VCWFFKFAAZCFQM-QHCPKHFHSA-N 0.000 description 1
- WBQLHWNABQQXHB-QHCPKHFHSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CCC1)C(C=CCNC(C)C)=O WBQLHWNABQQXHB-QHCPKHFHSA-N 0.000 description 1
- YOCKRVKXKBKWFI-QFIPXVFZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CC1)C(C=CCNC(C)C)=O YOCKRVKXKBKWFI-QFIPXVFZSA-N 0.000 description 1
- MPAOMAZVFRHCKU-QFIPXVFZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CC1)C(C=CCNC(C)C)=O MPAOMAZVFRHCKU-QFIPXVFZSA-N 0.000 description 1
- BERFKKCFJWJNSG-QFIPXVFZSA-N 3-[cyclopropylmethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CC1)C(C=CCNC(C)C)=O BERFKKCFJWJNSG-QFIPXVFZSA-N 0.000 description 1
- DVPAWHAAUMVFKW-IBGZPJMESA-N 3-[cyclopropylmethyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC1CC1 DVPAWHAAUMVFKW-IBGZPJMESA-N 0.000 description 1
- WLZRKEZSWYZXJL-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CN(C1)C(C=CCN(C)C)=O WLZRKEZSWYZXJL-UHFFFAOYSA-N 0.000 description 1
- QFUDCAQVXZNUJG-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C1CN(C1)C(C=CCN(C)C)=O QFUDCAQVXZNUJG-UHFFFAOYSA-N 0.000 description 1
- IBPLRARMIXUSLI-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C1CN(C1)C(C=CCN(C)C)=O IBPLRARMIXUSLI-UHFFFAOYSA-N 0.000 description 1
- SNUZCRVZSFNQOI-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C1CN(C1)C(C=CCN(C)C)=O SNUZCRVZSFNQOI-UHFFFAOYSA-N 0.000 description 1
- RLKOMZONEYEDPT-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CN(C1)C(C=CCN(C)C)=O RLKOMZONEYEDPT-UHFFFAOYSA-N 0.000 description 1
- KNQAQPZJXMWGHQ-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C1CN(C1)C(C=CCN(C)C)=O KNQAQPZJXMWGHQ-UHFFFAOYSA-N 0.000 description 1
- HJVABSOPJHASEM-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[4-(methylamino)pyridin-2-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=NC=CC(=C1)NC)C1CN(C1)C(C=CCN(C)C)=O HJVABSOPJHASEM-UHFFFAOYSA-N 0.000 description 1
- SCGRQIFPLOAURE-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C1CN(C1)C(C=CCN(C)C)=O SCGRQIFPLOAURE-UHFFFAOYSA-N 0.000 description 1
- VQMGQYHOFIKELL-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C1CN(C1)C(C=CCN(C)C)=O VQMGQYHOFIKELL-UHFFFAOYSA-N 0.000 description 1
- DENROYIRVBUOGG-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C1CN(C1)C(C=CCN(C)C)=O DENROYIRVBUOGG-UHFFFAOYSA-N 0.000 description 1
- YBLOVFSYNHBTSO-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=NC=CC=C1)C1CN(C1)C(C=CCN(C)C)=O YBLOVFSYNHBTSO-UHFFFAOYSA-N 0.000 description 1
- JWGDPZKFIRWPAF-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CN(C1)C(C=CCN(C)C)=O JWGDPZKFIRWPAF-UHFFFAOYSA-N 0.000 description 1
- GFTHAZOUADABBE-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C1CN(C1)C(C=CCN(C)C)=O GFTHAZOUADABBE-UHFFFAOYSA-N 0.000 description 1
- RENUNIOQRDHARI-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CCN(CC1)C(C=CCN(C)C)=O RENUNIOQRDHARI-UHFFFAOYSA-N 0.000 description 1
- KLKGSPNPHXSGCD-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(dimethylamino)but-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CCN(CC1)C(C=CCN(C)C)=O KLKGSPNPHXSGCD-UHFFFAOYSA-N 0.000 description 1
- WMXFXGQAMXYPMT-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CN(C1)C(C=CCNC)=O WMXFXGQAMXYPMT-UHFFFAOYSA-N 0.000 description 1
- WGPNSTZAOVCGPK-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C1CN(C1)C(C=CCNC)=O WGPNSTZAOVCGPK-UHFFFAOYSA-N 0.000 description 1
- NTMUVGOLDQBUOH-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)C1CN(C1)C(C=CCNC)=O NTMUVGOLDQBUOH-UHFFFAOYSA-N 0.000 description 1
- SHXUSCIWMDXMDC-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C1CN(C1)C(C=CCNC)=O SHXUSCIWMDXMDC-UHFFFAOYSA-N 0.000 description 1
- BUJWJIKCNLCSBU-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C1CN(C1)C(C=CCNC)=O BUJWJIKCNLCSBU-UHFFFAOYSA-N 0.000 description 1
- SXDQKKUPYWZCJP-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CN(C1)C(C=CCNC)=O SXDQKKUPYWZCJP-UHFFFAOYSA-N 0.000 description 1
- KTYTWBDFKCSIMZ-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C1CN(C1)C(C=CCNC)=O KTYTWBDFKCSIMZ-UHFFFAOYSA-N 0.000 description 1
- AZINVPLVORYYMD-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C1CN(C1)C(C=CCNC)=O AZINVPLVORYYMD-UHFFFAOYSA-N 0.000 description 1
- LFBBSILMJOPYLA-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C1CN(C1)C(C=CCNC)=O LFBBSILMJOPYLA-UHFFFAOYSA-N 0.000 description 1
- ADJLZQMCGIXLKN-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C1CN(C1)C(C=CCNC)=O ADJLZQMCGIXLKN-UHFFFAOYSA-N 0.000 description 1
- HURDKALFPSNHJR-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-2-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=NC=CC=C1)C1CN(C1)C(C=CCNC)=O HURDKALFPSNHJR-UHFFFAOYSA-N 0.000 description 1
- BKXOFWPAHKGMJF-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CN(C1)C(C=CCNC)=O BKXOFWPAHKGMJF-UHFFFAOYSA-N 0.000 description 1
- TUJZFNZMNSMFGO-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C1CN(C1)C(C=CCNC)=O TUJZFNZMNSMFGO-UHFFFAOYSA-N 0.000 description 1
- WRMZNLQTQIWRGY-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CCN(CC1)C(C=CCNC)=O WRMZNLQTQIWRGY-UHFFFAOYSA-N 0.000 description 1
- SPBUXLNTVRABKK-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CCN(CC1)C(C=CCNC)=O SPBUXLNTVRABKK-UHFFFAOYSA-N 0.000 description 1
- SKUQOEXSTVENTN-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CCN(CC1)C(C=CCNC)=O SKUQOEXSTVENTN-UHFFFAOYSA-N 0.000 description 1
- NLSMVPQQVHJTQL-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CN(C1)C(C=CCNC(C)C)=O NLSMVPQQVHJTQL-UHFFFAOYSA-N 0.000 description 1
- CYJAMLRZLCZVQA-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(2-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=NC=C1)OC)=O)C1CN(C1)C(C=CCNC(C)C)=O CYJAMLRZLCZVQA-UHFFFAOYSA-N 0.000 description 1
- KWBGYXLKROKTNH-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(4-methoxypyridin-2-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=NC=CC(=C1)OC)C1CN(C1)C(C=CCNC(C)C)=O KWBGYXLKROKTNH-UHFFFAOYSA-N 0.000 description 1
- FMXRJNMSMRJRGV-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrazin-2-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=CN=C1)OC)=O)C1CN(C1)C(C=CCNC(C)C)=O FMXRJNMSMRJRGV-UHFFFAOYSA-N 0.000 description 1
- WVZVESPERVILJZ-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-(6-methoxypyrimidin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC(=C1)OC)=O)C1CN(C1)C(C=CCNC(C)C)=O WVZVESPERVILJZ-UHFFFAOYSA-N 0.000 description 1
- YXNZATYBDVVXLR-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CN(C1)C(C=CCNC(C)C)=O YXNZATYBDVVXLR-UHFFFAOYSA-N 0.000 description 1
- FLXUTTVJGWGVIC-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C1CN(C1)C(C=CCNC(C)C)=O FLXUTTVJGWGVIC-UHFFFAOYSA-N 0.000 description 1
- AFKRPRVGFLOMEU-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[4-(methylamino)pyridin-2-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=NC=CC(=C1)NC)C1CN(C1)C(C=CCNC(C)C)=O AFKRPRVGFLOMEU-UHFFFAOYSA-N 0.000 description 1
- CKSHGCXOOGRRIY-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C1CN(C1)C(C=CCNC(C)C)=O CKSHGCXOOGRRIY-UHFFFAOYSA-N 0.000 description 1
- LHSKCPNAZRGMCI-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C1CN(C1)C(C=CCNC(C)C)=O LHSKCPNAZRGMCI-UHFFFAOYSA-N 0.000 description 1
- LHNVNQLEMVXINJ-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C1CN(C1)C(C=CCNC(C)C)=O LHNVNQLEMVXINJ-UHFFFAOYSA-N 0.000 description 1
- MNGVKVGWBQNJMZ-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CN(C1)C(C=CCNC(C)C)=O MNGVKVGWBQNJMZ-UHFFFAOYSA-N 0.000 description 1
- BRGNAZIWVIURQT-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CCN(CC1)C(C=CCNC(C)C)=O BRGNAZIWVIURQT-UHFFFAOYSA-N 0.000 description 1
- SALZHXCCDOMYSJ-UHFFFAOYSA-N 3-[cyclopropylmethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C1(CC1)CN(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CCN(CC1)C(C=CCNC(C)C)=O SALZHXCCDOMYSJ-UHFFFAOYSA-N 0.000 description 1
- BNDYXDVJBHTKSN-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-(2-oxo-1H-pyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(NC=C1)=O)CC BNDYXDVJBHTKSN-UHFFFAOYSA-N 0.000 description 1
- OXGPCVMPHAHIGQ-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC OXGPCVMPHAHIGQ-UHFFFAOYSA-N 0.000 description 1
- MWXKEBCSLPHINH-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[2-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)CC MWXKEBCSLPHINH-UHFFFAOYSA-N 0.000 description 1
- NVCIXZYYBBZPJV-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[4-(methylamino)pyridin-2-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)NC)CC NVCIXZYYBBZPJV-UHFFFAOYSA-N 0.000 description 1
- GGNGYKUTVXLQSE-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[6-(methylamino)pyrazin-2-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)CC GGNGYKUTVXLQSE-UHFFFAOYSA-N 0.000 description 1
- VSRRJOBLZOSUGB-UHFFFAOYSA-N 3-[ethyl-(1-prop-2-enoylazetidin-3-yl)amino]-5-[6-(methylamino)pyrimidin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)CC VSRRJOBLZOSUGB-UHFFFAOYSA-N 0.000 description 1
- SAKIVMHOSKKLQL-LJQANCHMSA-N 3-[ethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CCC1)C(C=CCNC)=O SAKIVMHOSKKLQL-LJQANCHMSA-N 0.000 description 1
- GZROCBWAKMNZGP-LJQANCHMSA-N 3-[ethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCNC)=O GZROCBWAKMNZGP-LJQANCHMSA-N 0.000 description 1
- KNTMMZXAKINJNH-GOSISDBHSA-N 3-[ethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CC1)C(C=CCNC)=O KNTMMZXAKINJNH-GOSISDBHSA-N 0.000 description 1
- GBCRSLLHZVZJPM-GOSISDBHSA-N 3-[ethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CC1)C(C=CCNC)=O GBCRSLLHZVZJPM-GOSISDBHSA-N 0.000 description 1
- MZDFCXKXXLTOKH-GOSISDBHSA-N 3-[ethyl-[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CC1)C(C=CCNC)=O MZDFCXKXXLTOKH-GOSISDBHSA-N 0.000 description 1
- NSEKTWUUMNCLFV-OAQYLSRUSA-N 3-[ethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CCC1)C(C=CCNC(C)C)=O NSEKTWUUMNCLFV-OAQYLSRUSA-N 0.000 description 1
- VCEHOMQASDTPSH-OAQYLSRUSA-N 3-[ethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CCC1)C(C=CCNC(C)C)=O VCEHOMQASDTPSH-OAQYLSRUSA-N 0.000 description 1
- FMGKVLQFZGRUED-OAQYLSRUSA-N 3-[ethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCNC(C)C)=O FMGKVLQFZGRUED-OAQYLSRUSA-N 0.000 description 1
- MMROEEAXOCVHIG-HXUWFJFHSA-N 3-[ethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@H]1CN(CC1)C(C=CCNC(C)C)=O MMROEEAXOCVHIG-HXUWFJFHSA-N 0.000 description 1
- AZXPQOGLMHBLGE-HXUWFJFHSA-N 3-[ethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@H]1CN(CC1)C(C=CCNC(C)C)=O AZXPQOGLMHBLGE-HXUWFJFHSA-N 0.000 description 1
- MENCGNLUYWOUSX-HXUWFJFHSA-N 3-[ethyl-[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@H]1CN(CC1)C(C=CCNC(C)C)=O MENCGNLUYWOUSX-HXUWFJFHSA-N 0.000 description 1
- ORAXYQAMNHJTRV-QGZVFWFLSA-N 3-[ethyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC ORAXYQAMNHJTRV-QGZVFWFLSA-N 0.000 description 1
- MPTXGXOHHKJNQK-QGZVFWFLSA-N 3-[ethyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC MPTXGXOHHKJNQK-QGZVFWFLSA-N 0.000 description 1
- SDGLRPMVJXYGDI-QGZVFWFLSA-N 3-[ethyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC SDGLRPMVJXYGDI-QGZVFWFLSA-N 0.000 description 1
- BHLXJEOSBVYQTJ-MRXNPFEDSA-N 3-[ethyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)CC BHLXJEOSBVYQTJ-MRXNPFEDSA-N 0.000 description 1
- SNKNLNMQUIZLKM-MRXNPFEDSA-N 3-[ethyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CC SNKNLNMQUIZLKM-MRXNPFEDSA-N 0.000 description 1
- YJXVNJIQTZLYKT-MRXNPFEDSA-N 3-[ethyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)CC YJXVNJIQTZLYKT-MRXNPFEDSA-N 0.000 description 1
- HZISITSLQGUFQC-IBGZPJMESA-N 3-[ethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CCC1)C(C=CCNC)=O HZISITSLQGUFQC-IBGZPJMESA-N 0.000 description 1
- GZROCBWAKMNZGP-IBGZPJMESA-N 3-[ethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CCC1)C(C=CCNC)=O GZROCBWAKMNZGP-IBGZPJMESA-N 0.000 description 1
- KNTMMZXAKINJNH-SFHVURJKSA-N 3-[ethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CC1)C(C=CCNC)=O KNTMMZXAKINJNH-SFHVURJKSA-N 0.000 description 1
- GBCRSLLHZVZJPM-SFHVURJKSA-N 3-[ethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CC1)C(C=CCNC)=O GBCRSLLHZVZJPM-SFHVURJKSA-N 0.000 description 1
- MZDFCXKXXLTOKH-SFHVURJKSA-N 3-[ethyl-[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CC1)C(C=CCNC)=O MZDFCXKXXLTOKH-SFHVURJKSA-N 0.000 description 1
- NSEKTWUUMNCLFV-NRFANRHFSA-N 3-[ethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)[C@@H]1CN(CCC1)C(C=CCNC(C)C)=O NSEKTWUUMNCLFV-NRFANRHFSA-N 0.000 description 1
- VCEHOMQASDTPSH-NRFANRHFSA-N 3-[ethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CCC1)C(C=CCNC(C)C)=O VCEHOMQASDTPSH-NRFANRHFSA-N 0.000 description 1
- FMGKVLQFZGRUED-NRFANRHFSA-N 3-[ethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CCC1)C(C=CCNC(C)C)=O FMGKVLQFZGRUED-NRFANRHFSA-N 0.000 description 1
- AZXPQOGLMHBLGE-FQEVSTJZSA-N 3-[ethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)[C@@H]1CN(CC1)C(C=CCNC(C)C)=O AZXPQOGLMHBLGE-FQEVSTJZSA-N 0.000 description 1
- MENCGNLUYWOUSX-FQEVSTJZSA-N 3-[ethyl-[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)[C@@H]1CN(CC1)C(C=CCNC(C)C)=O MENCGNLUYWOUSX-FQEVSTJZSA-N 0.000 description 1
- TWUQCAJUOAXJNE-UHFFFAOYSA-N 3-[ethyl-[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CCN(CC1)C(C=CCNC)=O TWUQCAJUOAXJNE-UHFFFAOYSA-N 0.000 description 1
- JZGZPFJLIULBKI-UHFFFAOYSA-N 3-[ethyl-[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CCN(CC1)C(C=CCNC)=O JZGZPFJLIULBKI-UHFFFAOYSA-N 0.000 description 1
- TXMDQHDHROTOIA-UHFFFAOYSA-N 3-[ethyl-[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CCN(CC1)C(C=CCNC)=O TXMDQHDHROTOIA-UHFFFAOYSA-N 0.000 description 1
- KRGJVFVCNSCOKU-UHFFFAOYSA-N 3-[ethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-5-(2-methoxypyridin-4-yl)-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C1CCN(CC1)C(C=CCNC(C)C)=O KRGJVFVCNSCOKU-UHFFFAOYSA-N 0.000 description 1
- XFJAQTTWLUCWGT-UHFFFAOYSA-N 3-[ethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-5-[2-(methylamino)pyridin-4-yl]-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C1CCN(CC1)C(C=CCNC(C)C)=O XFJAQTTWLUCWGT-UHFFFAOYSA-N 0.000 description 1
- HNBAFHSSTZLYPO-UHFFFAOYSA-N 3-[ethyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C1CCN(CC1)C(C=CCNC(C)C)=O HNBAFHSSTZLYPO-UHFFFAOYSA-N 0.000 description 1
- SDWCDWJKYSOFOQ-MRXNPFEDSA-N 3-[methyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C SDWCDWJKYSOFOQ-MRXNPFEDSA-N 0.000 description 1
- WVJCRPNLICTXAM-OAHLLOKOSA-N 3-[methyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-5-pyridin-4-yl-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC=NC=C1)C WVJCRPNLICTXAM-OAHLLOKOSA-N 0.000 description 1
- IODRIJYAWXGLBU-UHFFFAOYSA-N 3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrazin-2-yl-1H-pyridin-2-one Chemical compound CN(C=1C(NC=C(C1)C1=NC=CN=C1)=O)C1CN(C1)C(C=CCNC)=O IODRIJYAWXGLBU-UHFFFAOYSA-N 0.000 description 1
- XJZFSUCLBNYDRU-UHFFFAOYSA-N 3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-5-pyrimidin-4-yl-1H-pyridin-2-one Chemical compound CN(C=1C(NC=C(C1)C1=NC=NC=C1)=O)C1CN(C1)C(C=CCNC)=O XJZFSUCLBNYDRU-UHFFFAOYSA-N 0.000 description 1
- VTSFNCCQCOEPKF-UHFFFAOYSA-N 3-amino-1h-pyridin-2-one Chemical compound NC1=CC=CN=C1O VTSFNCCQCOEPKF-UHFFFAOYSA-N 0.000 description 1
- AYEVELLBDURPJW-UHFFFAOYSA-N 3-bromo-5-chloro-2-methoxypyridine Chemical compound COC1=NC=C(Cl)C=C1Br AYEVELLBDURPJW-UHFFFAOYSA-N 0.000 description 1
- LGQIJQYCMDPYSK-UHFFFAOYSA-N 3-cyano-N-(3-methyl-4-propan-2-ylphenyl)-5-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]benzamide Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=C(C1)C#N LGQIJQYCMDPYSK-UHFFFAOYSA-N 0.000 description 1
- ZGZDGISPBQEWPB-UHFFFAOYSA-N 3-ethyl-N-(3-methyl-4-propan-2-ylphenyl)-5-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]benzamide Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=C(C1)CC ZGZDGISPBQEWPB-UHFFFAOYSA-N 0.000 description 1
- VPOAQDSAPBSOLW-UHFFFAOYSA-N 3-methyl-4-propan-2-ylaniline;hydrochloride Chemical compound Cl.CC(C)C1=CC=C(N)C=C1C VPOAQDSAPBSOLW-UHFFFAOYSA-N 0.000 description 1
- PMAYBZYHELLYCY-UHFFFAOYSA-N 3-methyl-N-(3-methyl-4-propan-2-ylphenyl)-5-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]benzamide Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=C(C1)C PMAYBZYHELLYCY-UHFFFAOYSA-N 0.000 description 1
- MWCPJYAFTGMADB-QGZVFWFLSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CCC1)C(C=CCNC)=O)=O MWCPJYAFTGMADB-QGZVFWFLSA-N 0.000 description 1
- QKWFYTQKPHLKLF-MRXNPFEDSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3R)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCNC)=O)=O QKWFYTQKPHLKLF-MRXNPFEDSA-N 0.000 description 1
- YRAQTSYDYBRQAV-ZLHKHBAESA-N 5-(2-methoxypyridin-4-yl)-3-[[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC YRAQTSYDYBRQAV-ZLHKHBAESA-N 0.000 description 1
- FZUVVZDMPLEZLP-CQSZACIVSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)OC FZUVVZDMPLEZLP-CQSZACIVSA-N 0.000 description 1
- MWCPJYAFTGMADB-KRWDZBQOSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@@H]1CN(CCC1)C(C=CCNC)=O)=O MWCPJYAFTGMADB-KRWDZBQOSA-N 0.000 description 1
- QKWFYTQKPHLKLF-INIZCTEOSA-N 5-(2-methoxypyridin-4-yl)-3-[[(3S)-1-[(E)-4-(methylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@@H]1CN(CC1)C(C=CCNC)=O)=O QKWFYTQKPHLKLF-INIZCTEOSA-N 0.000 description 1
- YMDJSZGWUDWHRV-UHFFFAOYSA-N 5-(2-methoxypyridin-4-yl)-3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCNC)=O)=O YMDJSZGWUDWHRV-UHFFFAOYSA-N 0.000 description 1
- RFVPADWHUITSNM-UHFFFAOYSA-N 5-(2-methoxypyridin-4-yl)-3-[[1-[(E)-4-(methylamino)but-2-enoyl]piperidin-4-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CCN(CC1)C(C=CCNC)=O)=O RFVPADWHUITSNM-UHFFFAOYSA-N 0.000 description 1
- IJQPGCKAECKSFD-MRXNPFEDSA-N 5-(2-methoxypyridin-4-yl)-3-[methyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C IJQPGCKAECKSFD-MRXNPFEDSA-N 0.000 description 1
- AGSVRRBRIUOTEZ-OAHLLOKOSA-N 5-(2-methoxypyridin-4-yl)-3-[methyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)OC)C AGSVRRBRIUOTEZ-OAHLLOKOSA-N 0.000 description 1
- TTXWPASACOPDAE-UHFFFAOYSA-N 5-(2-methoxypyrimidin-4-yl)-3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=N1)C=1C=C(C(NC1)=O)NC1CN(C1)C(C=CCNC)=O TTXWPASACOPDAE-UHFFFAOYSA-N 0.000 description 1
- ZGJCKDYQKSKUIP-UHFFFAOYSA-N 5-(2-methoxypyrimidin-4-yl)-3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=NC=CC(=N1)C=1C=C(C(NC1)=O)N(C1CN(C1)C(C=CCNC)=O)C ZGJCKDYQKSKUIP-UHFFFAOYSA-N 0.000 description 1
- SMJAIBCHVRINLU-UHFFFAOYSA-N 5-(2-oxo-1H-pyridin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(NC=C1)=O)CCC SMJAIBCHVRINLU-UHFFFAOYSA-N 0.000 description 1
- KBOIBOKVKKMCBQ-UHFFFAOYSA-N 5-(2-oxo-1H-pyridin-4-yl)-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(NC=C1)=O KBOIBOKVKKMCBQ-UHFFFAOYSA-N 0.000 description 1
- LQISLEIINJJJPK-UHFFFAOYSA-N 5-(4-methoxypyridin-2-yl)-3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=CC(=NC=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCNC)=O)=O LQISLEIINJJJPK-UHFFFAOYSA-N 0.000 description 1
- VISMEXOZUIQIQO-UHFFFAOYSA-N 5-(6-methoxypyrazin-2-yl)-3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=CN=CC(=N1)C=1C=C(C(NC1)=O)NC1CN(C1)C(C=CCNC)=O VISMEXOZUIQIQO-UHFFFAOYSA-N 0.000 description 1
- LQUMEYWQSWOLMM-UHFFFAOYSA-N 5-(6-methoxypyrazin-2-yl)-3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=CN=CC(=N1)C=1C=C(C(NC1)=O)N(C1CN(C1)C(C=CCNC)=O)C LQUMEYWQSWOLMM-UHFFFAOYSA-N 0.000 description 1
- QXKGKBQJXHSDMQ-UHFFFAOYSA-N 5-(6-methoxypyrimidin-4-yl)-3-[[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=CC(=NC=N1)C=1C=C(C(NC1)=O)NC1CN(C1)C(C=CCNC)=O QXKGKBQJXHSDMQ-UHFFFAOYSA-N 0.000 description 1
- RLSNZUSRDRCYQP-UHFFFAOYSA-N 5-(6-methoxypyrimidin-4-yl)-3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound COC1=CC(=NC=N1)C=1C=C(C(NC1)=O)N(C1CN(C1)C(C=CCNC)=O)C RLSNZUSRDRCYQP-UHFFFAOYSA-N 0.000 description 1
- ZLZXENFHTMBHOD-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)CCC ZLZXENFHTMBHOD-UHFFFAOYSA-N 0.000 description 1
- FXHNGPREEIATEC-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[(1-prop-2-enoylpiperidin-4-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC FXHNGPREEIATEC-UHFFFAOYSA-N 0.000 description 1
- FRCGDBFPXRJLTK-ZLHKHBAESA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC FRCGDBFPXRJLTK-ZLHKHBAESA-N 0.000 description 1
- VLAXWQZTXYEYGY-SHCNBLLESA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@@H](CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC VLAXWQZTXYEYGY-SHCNBLLESA-N 0.000 description 1
- PFSADQTTWICXHR-LJQANCHMSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-morpholin-4-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCN1CCOCC1)=O)=O PFSADQTTWICXHR-LJQANCHMSA-N 0.000 description 1
- FGIJGNWKVSTDSI-JOCHJYFZSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-phenylbut-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CCC1)C(C=CCC1=CC=CC=C1)=O)=O FGIJGNWKVSTDSI-JOCHJYFZSA-N 0.000 description 1
- QTWLCQFXNURFTF-OAQYLSRUSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-phenylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCC1=CC=CC=C1)=O)=O QTWLCQFXNURFTF-OAQYLSRUSA-N 0.000 description 1
- OBTNJRBSACCPHJ-OAQYLSRUSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-piperidin-1-ylbut-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CCC1)C(C=CCN1CCCCC1)=O)=O OBTNJRBSACCPHJ-OAQYLSRUSA-N 0.000 description 1
- GGAVXENOYLXHQK-HXUWFJFHSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-piperidin-1-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCN1CCCCC1)=O)=O GGAVXENOYLXHQK-HXUWFJFHSA-N 0.000 description 1
- AHEXPAIFFAEUEO-OAQYLSRUSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-pyridin-2-ylbut-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CCC1)C(C=CCC1=NC=CC=C1)=O)=O AHEXPAIFFAEUEO-OAQYLSRUSA-N 0.000 description 1
- WRMNQIPBDWVKMQ-HXUWFJFHSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-pyridin-2-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCC1=NC=CC=C1)=O)=O WRMNQIPBDWVKMQ-HXUWFJFHSA-N 0.000 description 1
- BUBZUJOAJHUIQJ-HXUWFJFHSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-pyrrolidin-1-ylbut-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CCC1)C(C=CCN1CCCC1)=O)=O BUBZUJOAJHUIQJ-HXUWFJFHSA-N 0.000 description 1
- MZIWXKNIDNVLGA-LJQANCHMSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-[(E)-4-pyrrolidin-1-ylbut-2-enoyl]pyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)N[C@H]1CN(CC1)C(C=CCN1CCCC1)=O)=O MZIWXKNIDNVLGA-LJQANCHMSA-N 0.000 description 1
- SXSAODKCXZRQKN-OAHLLOKOSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC SXSAODKCXZRQKN-OAHLLOKOSA-N 0.000 description 1
- FRCGDBFPXRJLTK-QTWIXLPOSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3S)-1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC FRCGDBFPXRJLTK-QTWIXLPOSA-N 0.000 description 1
- SXSAODKCXZRQKN-HNNXBMFYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC SXSAODKCXZRQKN-HNNXBMFYSA-N 0.000 description 1
- AFMRVUPFOZYBGU-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-3-pyridin-2-ylprop-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CC1=NC=CC=C1)=O)=O AFMRVUPFOZYBGU-UHFFFAOYSA-N 0.000 description 1
- XXYYYMFXPXUVIL-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-(oxan-4-yl)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCC1CCOCC1)=O)=O XXYYYMFXPXUVIL-UHFFFAOYSA-N 0.000 description 1
- PABYXHBWECAIHS-SNAWJCMRSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC PABYXHBWECAIHS-SNAWJCMRSA-N 0.000 description 1
- RQZZOOQNZWHTKW-SNAWJCMRSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]piperidin-4-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CCC(CC1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC RQZZOOQNZWHTKW-SNAWJCMRSA-N 0.000 description 1
- GWWNAXSORBYHAJ-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-morpholin-4-ylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCN1CCOCC1)=O)=O GWWNAXSORBYHAJ-UHFFFAOYSA-N 0.000 description 1
- NLDFGGJAZMHRLH-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-piperidin-1-ylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCN1CCCCC1)=O)=O NLDFGGJAZMHRLH-UHFFFAOYSA-N 0.000 description 1
- GMYQCSQQHDVBIO-SNAWJCMRSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-propan-2-yloxybut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)OC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=CC(=NC=C1)NC GMYQCSQQHDVBIO-SNAWJCMRSA-N 0.000 description 1
- BKADFYWIOPIUHN-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-pyridin-2-ylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCC1=NC=CC=C1)=O)=O BKADFYWIOPIUHN-UHFFFAOYSA-N 0.000 description 1
- MYRMZACZZJEKAK-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[[1-[(E)-4-pyrrolidin-1-ylbut-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CNC1=NC=CC(=C1)C1=CNC(C(=C1)NC1CN(C1)C(C=CCN1CCCC1)=O)=O MYRMZACZZJEKAK-UHFFFAOYSA-N 0.000 description 1
- SKFQRRWNIYQKDH-UHFFFAOYSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[methyl-(1-prop-2-enoylpiperidin-4-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CCC(CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C SKFQRRWNIYQKDH-UHFFFAOYSA-N 0.000 description 1
- ARSFEMKYGKZYCQ-MRXNPFEDSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[methyl-[(3R)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C ARSFEMKYGKZYCQ-MRXNPFEDSA-N 0.000 description 1
- CAAJFWLBVLMCLZ-OAHLLOKOSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[methyl-[(3R)-1-prop-2-enoylpyrrolidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@@H](CC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C CAAJFWLBVLMCLZ-OAHLLOKOSA-N 0.000 description 1
- ARSFEMKYGKZYCQ-INIZCTEOSA-N 5-[2-(methylamino)pyridin-4-yl]-3-[methyl-[(3S)-1-prop-2-enoylpiperidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1C[C@H](CCC1)N(C1=CC(=CNC1=O)C1=CC(=NC=C1)NC)C ARSFEMKYGKZYCQ-INIZCTEOSA-N 0.000 description 1
- JDQAZOZSVKQRJP-UHFFFAOYSA-N 5-[2-(methylamino)pyrimidin-4-yl]-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)CCC JDQAZOZSVKQRJP-UHFFFAOYSA-N 0.000 description 1
- IPZQWGRPLYIDKW-UHFFFAOYSA-N 5-[2-(methylamino)pyrimidin-4-yl]-3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CN(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C1CN(C1)C(C=CCNC)=O IPZQWGRPLYIDKW-UHFFFAOYSA-N 0.000 description 1
- FTOFEPQCUHBKQT-AATRIKPKSA-N 5-[2-(methylamino)pyrimidin-4-yl]-3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=NC=C1)NC)=O)C FTOFEPQCUHBKQT-AATRIKPKSA-N 0.000 description 1
- QDTKELZLXRICEV-UHFFFAOYSA-N 5-[4-(methylamino)pyridin-2-yl]-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C1=CC(=CNC1=O)C1=NC=CC(=C1)NC)CCC QDTKELZLXRICEV-UHFFFAOYSA-N 0.000 description 1
- SLBJQFMPDSTHPQ-UHFFFAOYSA-N 5-[4-(methylamino)pyridin-2-yl]-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)NC SLBJQFMPDSTHPQ-UHFFFAOYSA-N 0.000 description 1
- IGBMKLGDVKZHSG-SNAWJCMRSA-N 5-[4-(methylamino)pyridin-2-yl]-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C1=NC=CC(=C1)NC IGBMKLGDVKZHSG-SNAWJCMRSA-N 0.000 description 1
- QJTFNDNVHAJVLW-UHFFFAOYSA-N 5-[6-(methylamino)pyrazin-2-yl]-3-[(1-prop-2-enoylazetidin-3-yl)-propylamino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)CCC QJTFNDNVHAJVLW-UHFFFAOYSA-N 0.000 description 1
- FUKUOSCGHIVQKF-SNAWJCMRSA-N 5-[6-(methylamino)pyrazin-2-yl]-3-[[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O FUKUOSCGHIVQKF-SNAWJCMRSA-N 0.000 description 1
- JTQARSZYKOTIPA-UHFFFAOYSA-N 5-[6-(methylamino)pyrazin-2-yl]-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C JTQARSZYKOTIPA-UHFFFAOYSA-N 0.000 description 1
- GHFSIYCAXMQONF-UHFFFAOYSA-N 5-[6-(methylamino)pyrazin-2-yl]-3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CN(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C1CN(C1)C(C=CCNC)=O GHFSIYCAXMQONF-UHFFFAOYSA-N 0.000 description 1
- OVSJICDVEDKNNJ-AATRIKPKSA-N 5-[6-(methylamino)pyrazin-2-yl]-3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC(=CN=C1)NC)=O)C OVSJICDVEDKNNJ-AATRIKPKSA-N 0.000 description 1
- RJSSEVBVOFCCEW-UHFFFAOYSA-N 5-[6-(methylamino)pyrimidin-4-yl]-3-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)NC=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O RJSSEVBVOFCCEW-UHFFFAOYSA-N 0.000 description 1
- UVAJWMCDMXTGCZ-UHFFFAOYSA-N 5-[6-(methylamino)pyrimidin-4-yl]-3-[methyl-(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-2-one Chemical compound C(C=C)(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C UVAJWMCDMXTGCZ-UHFFFAOYSA-N 0.000 description 1
- OAEWQMRTAFFEGH-UHFFFAOYSA-N 5-[6-(methylamino)pyrimidin-4-yl]-3-[methyl-[1-[(E)-4-(methylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound CN(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C1CN(C1)C(C=CCNC)=O OAEWQMRTAFFEGH-UHFFFAOYSA-N 0.000 description 1
- SERYTDFWALTHPX-AATRIKPKSA-N 5-[6-(methylamino)pyrimidin-4-yl]-3-[methyl-[1-[(E)-4-(propan-2-ylamino)but-2-enoyl]azetidin-3-yl]amino]-1H-pyridin-2-one Chemical compound C(C)(C)NC/C=C/C(=O)N1CC(C1)N(C=1C(NC=C(C1)C1=NC=NC(=C1)NC)=O)C SERYTDFWALTHPX-AATRIKPKSA-N 0.000 description 1
- NDMZZQRNZFWMEZ-UHFFFAOYSA-N 5-bromo-1h-pyridin-2-one Chemical compound OC1=CC=C(Br)C=N1 NDMZZQRNZFWMEZ-UHFFFAOYSA-N 0.000 description 1
- QRXMUCSWCMTJGU-UHFFFAOYSA-N 5-bromo-4-chloro-3-indolyl phosphate Chemical compound C1=C(Br)C(Cl)=C2C(OP(O)(=O)O)=CNC2=C1 QRXMUCSWCMTJGU-UHFFFAOYSA-N 0.000 description 1
- ODHCTXKNWHHXJC-VKHMYHEASA-N 5-oxo-L-proline Chemical compound OC(=O)[C@@H]1CCC(=O)N1 ODHCTXKNWHHXJC-VKHMYHEASA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- VVIAGPKUTFNRDU-UHFFFAOYSA-N 6S-folinic acid Natural products C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 VVIAGPKUTFNRDU-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 102000007469 Actins Human genes 0.000 description 1
- 108010085238 Actins Proteins 0.000 description 1
- 208000003200 Adenoma Diseases 0.000 description 1
- 206010001233 Adenoma benign Diseases 0.000 description 1
- 239000012099 Alexa Fluor family Substances 0.000 description 1
- 208000035939 Alveolitis allergic Diseases 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 244000144725 Amygdalus communis Species 0.000 description 1
- 235000011437 Amygdalus communis Nutrition 0.000 description 1
- 208000008822 Ankylosis Diseases 0.000 description 1
- 102000006306 Antigen Receptors Human genes 0.000 description 1
- 108010083359 Antigen Receptors Proteins 0.000 description 1
- 208000032467 Aplastic anaemia Diseases 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 201000008283 Atrophic Rhinitis Diseases 0.000 description 1
- 102100022005 B-lymphocyte antigen CD20 Human genes 0.000 description 1
- 229940124291 BTK inhibitor Drugs 0.000 description 1
- 206010004146 Basal cell carcinoma Diseases 0.000 description 1
- 208000009137 Behcet syndrome Diseases 0.000 description 1
- 108010006654 Bleomycin Proteins 0.000 description 1
- 208000019838 Blood disease Diseases 0.000 description 1
- 206010005949 Bone cancer Diseases 0.000 description 1
- 206010048396 Bone marrow transplant rejection Diseases 0.000 description 1
- 208000018084 Bone neoplasm Diseases 0.000 description 1
- YVDNRORMDSADBZ-UHFFFAOYSA-N Br.CC(C)(C)OC(=O)N1CCC(N)C1.CC1(C)OB(C2=CC(F)=NC=C2)OC1(C)C.CNC1=NC=CC(C2=CN=C(OC)C(NC3CCN(C(=O)OC(C)(C)C)C3)=C2)=C1.CNC1=NC=CC(C2=CNC(=O)C(NC3CCNC3)=C2)=C1.COC1=NC=C(C2=CC(F)=NC=C2)C=C1NC1CCN(C(=O)OC(C)(C)C)C1.COC1=NC=C(Cl)C=C1Br.COC1=NC=C(Cl)C=C1NC1CCN(C(=O)OC(C)(C)C)C1 Chemical compound Br.CC(C)(C)OC(=O)N1CCC(N)C1.CC1(C)OB(C2=CC(F)=NC=C2)OC1(C)C.CNC1=NC=CC(C2=CN=C(OC)C(NC3CCN(C(=O)OC(C)(C)C)C3)=C2)=C1.CNC1=NC=CC(C2=CNC(=O)C(NC3CCNC3)=C2)=C1.COC1=NC=C(C2=CC(F)=NC=C2)C=C1NC1CCN(C(=O)OC(C)(C)C)C1.COC1=NC=C(Cl)C=C1Br.COC1=NC=C(Cl)C=C1NC1CCN(C(=O)OC(C)(C)C)C1 YVDNRORMDSADBZ-UHFFFAOYSA-N 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- UICLEFQDHCRSBX-ONEGZZNKSA-N C(C)(C)NC/C=C/C(=O)N1CCC(CC1)NC=1C(=CC=NC1)C1=CNC(C=C1)=O Chemical compound C(C)(C)NC/C=C/C(=O)N1CCC(CC1)NC=1C(=CC=NC1)C1=CNC(C=C1)=O UICLEFQDHCRSBX-ONEGZZNKSA-N 0.000 description 1
- VFJBBSPSTCPUAC-GOSISDBHSA-N C(C)N(C1=CC(=C(NC1=O)NC)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCNC)=O Chemical compound C(C)N(C1=CC(=C(NC1=O)NC)C1=CC=NC=C1)[C@H]1CN(CCC1)C(C=CCNC)=O VFJBBSPSTCPUAC-GOSISDBHSA-N 0.000 description 1
- ZZERBOITEKKOSA-LZWSPWQCSA-N C/C=C(\C#N)C(=O)N1CC(CC2=CC(C3=CC=NC(NC)=C3)=CNC2=O)C1 Chemical compound C/C=C(\C#N)C(=O)N1CC(CC2=CC(C3=CC=NC(NC)=C3)=CNC2=O)C1 ZZERBOITEKKOSA-LZWSPWQCSA-N 0.000 description 1
- NELGWLVRPORBDZ-JLZUIIAYSA-N C/C=C(\C#N)C(=O)N1CC(CC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C/C=C(\C#N)C(=O)N1CC(CC2=CC(C3=CC=NC=C3)=CNC2=O)C1 NELGWLVRPORBDZ-JLZUIIAYSA-N 0.000 description 1
- YMSWZTDKNYYMDE-HNSSHINMSA-N C/C=C/C(=O)N1CCC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C/C=C/C(=O)N1CCC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 YMSWZTDKNYYMDE-HNSSHINMSA-N 0.000 description 1
- BRPBAKLKOUCGMD-WPTTZPOMSA-N C/C=C/C(=O)N1CCC[C@@H](NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C/C=C/C(=O)N1CCC[C@@H](NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 BRPBAKLKOUCGMD-WPTTZPOMSA-N 0.000 description 1
- GHXPSRCIIBVQKO-NJELEJDNSA-N C/C=C/C(=O)N1CC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C/C=C/C(=O)N1CC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 GHXPSRCIIBVQKO-NJELEJDNSA-N 0.000 description 1
- UZUNDECKCZNNOO-XYBNCVKDSA-N C/C=C/C(=O)N1CC[C@@H](NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C/C=C/C(=O)N1CC[C@@H](NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 UZUNDECKCZNNOO-XYBNCVKDSA-N 0.000 description 1
- JLXXXRAROLQJFI-UHFFFAOYSA-N C=C(C#N)C(=O)N1CC(CC2=CC(C3=CC=NC(NC)=C3)=CNC2=O)C1 Chemical compound C=C(C#N)C(=O)N1CC(CC2=CC(C3=CC=NC(NC)=C3)=CNC2=O)C1 JLXXXRAROLQJFI-UHFFFAOYSA-N 0.000 description 1
- NMXNJOSEVKYECR-UHFFFAOYSA-N C=C(C#N)C(=O)N1CC(CC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=C(C#N)C(=O)N1CC(CC2=CC(C3=CC=NC=C3)=CNC2=O)C1 NMXNJOSEVKYECR-UHFFFAOYSA-N 0.000 description 1
- DFMHUYOLKXGBCZ-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 DFMHUYOLKXGBCZ-UHFFFAOYSA-N 0.000 description 1
- CHJCMVIHGQKEMQ-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 CHJCMVIHGQKEMQ-UHFFFAOYSA-N 0.000 description 1
- XZYSXFWFMZRASW-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 XZYSXFWFMZRASW-UHFFFAOYSA-N 0.000 description 1
- USTQIKWHAFNLTK-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 USTQIKWHAFNLTK-UHFFFAOYSA-N 0.000 description 1
- UQODWYYTLFLESA-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=CC(OC)=CC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=CC(OC)=CC=N3)=CNC2=O)C1 UQODWYYTLFLESA-UHFFFAOYSA-N 0.000 description 1
- VJOJKLAJEFEACS-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 VJOJKLAJEFEACS-UHFFFAOYSA-N 0.000 description 1
- OEGIVMQBWSJONG-UHFFFAOYSA-N C=CC(=O)N1CC(N(C)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(C)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 OEGIVMQBWSJONG-UHFFFAOYSA-N 0.000 description 1
- JLCZYTIATYWBML-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C2CC2)C1 JLCZYTIATYWBML-UHFFFAOYSA-N 0.000 description 1
- CCDFQNCYFFQQHF-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=CC(C)=NC=N3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=CC(C)=NC=N3)=CNC2=O)C2CC2)C1 CCDFQNCYFFQQHF-UHFFFAOYSA-N 0.000 description 1
- MQTXWOYPLUUYBF-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C2CC2)C1 MQTXWOYPLUUYBF-UHFFFAOYSA-N 0.000 description 1
- MNYGRTLDQLBDHS-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C2CC2)C1 MNYGRTLDQLBDHS-UHFFFAOYSA-N 0.000 description 1
- OMRMPBCJKZHOHW-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C2CC2)C1 OMRMPBCJKZHOHW-UHFFFAOYSA-N 0.000 description 1
- SZOGENWAPANSAO-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=CC=NC=C3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=CC=NC=C3)=CNC2=O)C2CC2)C1 SZOGENWAPANSAO-UHFFFAOYSA-N 0.000 description 1
- YYQIJWIOVSWNAG-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C2CC2)C1 YYQIJWIOVSWNAG-UHFFFAOYSA-N 0.000 description 1
- LWSUVDGZHGEYQP-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C2CC2)C1 LWSUVDGZHGEYQP-UHFFFAOYSA-N 0.000 description 1
- GDWVAWXDVAZEOE-UHFFFAOYSA-N C=CC(=O)N1CC(N(C2=CC(C3=NC=CN=C3)=CNC2=O)C2CC2)C1 Chemical compound C=CC(=O)N1CC(N(C2=CC(C3=NC=CN=C3)=CNC2=O)C2CC2)C1 GDWVAWXDVAZEOE-UHFFFAOYSA-N 0.000 description 1
- DLNVRFAJBMWPPM-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 DLNVRFAJBMWPPM-UHFFFAOYSA-N 0.000 description 1
- HAXUESDNZWZVNV-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 HAXUESDNZWZVNV-UHFFFAOYSA-N 0.000 description 1
- NPRAFWBJHPCSNW-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 NPRAFWBJHPCSNW-UHFFFAOYSA-N 0.000 description 1
- HQUIKHRFMODLMM-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 HQUIKHRFMODLMM-UHFFFAOYSA-N 0.000 description 1
- DXCNEMGENHIAGE-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 DXCNEMGENHIAGE-UHFFFAOYSA-N 0.000 description 1
- UQWLEJVBVYIWHV-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 UQWLEJVBVYIWHV-UHFFFAOYSA-N 0.000 description 1
- TYZVHEZDIFKSOS-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 TYZVHEZDIFKSOS-UHFFFAOYSA-N 0.000 description 1
- KEKPPZGWLYCRJL-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 KEKPPZGWLYCRJL-UHFFFAOYSA-N 0.000 description 1
- HQBYRRYXCQGHNF-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 HQBYRRYXCQGHNF-UHFFFAOYSA-N 0.000 description 1
- CZVIOHXKRMGXFU-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(C)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(C)=NC=C3)=CNC2=O)C1 CZVIOHXKRMGXFU-UHFFFAOYSA-N 0.000 description 1
- FGZFKPJTNOEQNC-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 FGZFKPJTNOEQNC-UHFFFAOYSA-N 0.000 description 1
- GRQYPJCXJWEBJW-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 GRQYPJCXJWEBJW-UHFFFAOYSA-N 0.000 description 1
- MUXGMMSHVZNYAD-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 MUXGMMSHVZNYAD-UHFFFAOYSA-N 0.000 description 1
- RRWFSHIKYMPQJS-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 RRWFSHIKYMPQJS-UHFFFAOYSA-N 0.000 description 1
- VBEUJEUIKCAMKG-UHFFFAOYSA-N C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 VBEUJEUIKCAMKG-UHFFFAOYSA-N 0.000 description 1
- KWRFUDNBAYGHNQ-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 KWRFUDNBAYGHNQ-UHFFFAOYSA-N 0.000 description 1
- NHMRMKQJMCWJQF-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 NHMRMKQJMCWJQF-UHFFFAOYSA-N 0.000 description 1
- QWIOHAIICPQKGN-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 QWIOHAIICPQKGN-UHFFFAOYSA-N 0.000 description 1
- YHOLUYQZFVFJOX-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 YHOLUYQZFVFJOX-UHFFFAOYSA-N 0.000 description 1
- OCBAYKUQHUNNHQ-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=NC(CC)=CN=C3)=CNC2=O)C1 OCBAYKUQHUNNHQ-UHFFFAOYSA-N 0.000 description 1
- PCOACXCKTUGDKZ-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=NC(CC)=NC=C3)=CNC2=O)C1 PCOACXCKTUGDKZ-UHFFFAOYSA-N 0.000 description 1
- JOYMMWOWMCDXSG-UHFFFAOYSA-N C=CC(=O)N1CC(N(CCC)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(N(CCC)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 JOYMMWOWMCDXSG-UHFFFAOYSA-N 0.000 description 1
- JLWYMJJGPIYMOL-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(=O)CC=C3)=CNC2=O)C1 JLWYMJJGPIYMOL-UHFFFAOYSA-N 0.000 description 1
- NLYIFZSOSZYRGB-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=CC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=CC=C3)=CNC2=O)C1 NLYIFZSOSZYRGB-UHFFFAOYSA-N 0.000 description 1
- VVMVBPIOHGQDPA-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(C(C)C)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(C(C)C)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 VVMVBPIOHGQDPA-UHFFFAOYSA-N 0.000 description 1
- WOQNMBDHDVROCM-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(C)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(C)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 WOQNMBDHDVROCM-UHFFFAOYSA-N 0.000 description 1
- UGFYUVUAVTVHPM-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(C4CC4)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(C4CC4)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 UGFYUVUAVTVHPM-UHFFFAOYSA-N 0.000 description 1
- XNOZEMOXJUYEKS-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 XNOZEMOXJUYEKS-UHFFFAOYSA-N 0.000 description 1
- SEIHONLXIWNTLA-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=CC=N3)=CNC2=O)C1 SEIHONLXIWNTLA-UHFFFAOYSA-N 0.000 description 1
- YYEQEUMCPNJERX-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(CC)=NC=N3)=CNC2=O)C1 YYEQEUMCPNJERX-UHFFFAOYSA-N 0.000 description 1
- FLXQRTVWCYPFJT-UHFFFAOYSA-N C=CC(=O)N1CC(NC2=CC(C3=CC(CCC)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 Chemical compound C=CC(=O)N1CC(NC2=CC(C3=CC(CCC)=CC(C(=O)NC4=CC(C)=C(C(C)C)C=C4)=C3)=CCC2=O)C1 FLXQRTVWCYPFJT-UHFFFAOYSA-N 0.000 description 1
- AEPPKLFTVSITJI-UHFFFAOYSA-N C=CC(=O)N1CCC(N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 Chemical compound C=CC(=O)N1CCC(N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 AEPPKLFTVSITJI-UHFFFAOYSA-N 0.000 description 1
- CAAJFWLBVLMCLZ-UHFFFAOYSA-N C=CC(=O)N1CCC(N(C)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(C)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 CAAJFWLBVLMCLZ-UHFFFAOYSA-N 0.000 description 1
- AGSVRRBRIUOTEZ-UHFFFAOYSA-N C=CC(=O)N1CCC(N(C)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(C)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 AGSVRRBRIUOTEZ-UHFFFAOYSA-N 0.000 description 1
- WVJCRPNLICTXAM-UHFFFAOYSA-N C=CC(=O)N1CCC(N(C)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(C)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 WVJCRPNLICTXAM-UHFFFAOYSA-N 0.000 description 1
- DTLISZNKBHFETN-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 Chemical compound C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 DTLISZNKBHFETN-UHFFFAOYSA-N 0.000 description 1
- SNKNLNMQUIZLKM-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 SNKNLNMQUIZLKM-UHFFFAOYSA-N 0.000 description 1
- BHLXJEOSBVYQTJ-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 BHLXJEOSBVYQTJ-UHFFFAOYSA-N 0.000 description 1
- YJXVNJIQTZLYKT-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(CC)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 YJXVNJIQTZLYKT-UHFFFAOYSA-N 0.000 description 1
- CSBNUQHTUOUCKR-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 Chemical compound C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 CSBNUQHTUOUCKR-UHFFFAOYSA-N 0.000 description 1
- VBJXXGQZWOPCFE-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 VBJXXGQZWOPCFE-UHFFFAOYSA-N 0.000 description 1
- XZHXVQHBBVCDEG-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 XZHXVQHBBVCDEG-UHFFFAOYSA-N 0.000 description 1
- NVGCOSZAHJJIKH-UHFFFAOYSA-N C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 NVGCOSZAHJJIKH-UHFFFAOYSA-N 0.000 description 1
- QLXUGVMIVXFORH-UHFFFAOYSA-N C=CC(=O)N1CCC(NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 Chemical compound C=CC(=O)N1CCC(NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)CC1 QLXUGVMIVXFORH-UHFFFAOYSA-N 0.000 description 1
- FZUVVZDMPLEZLP-UHFFFAOYSA-N C=CC(=O)N1CCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 FZUVVZDMPLEZLP-UHFFFAOYSA-N 0.000 description 1
- CPLHMHFPIOQAIA-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 CPLHMHFPIOQAIA-UHFFFAOYSA-N 0.000 description 1
- IJQPGCKAECKSFD-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(C)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(C)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 IJQPGCKAECKSFD-UHFFFAOYSA-N 0.000 description 1
- SDWCDWJKYSOFOQ-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(C)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(C)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 SDWCDWJKYSOFOQ-UHFFFAOYSA-N 0.000 description 1
- FUZYFBTULWSPJW-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 FUZYFBTULWSPJW-UHFFFAOYSA-N 0.000 description 1
- ORAXYQAMNHJTRV-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(CC)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(CC)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 ORAXYQAMNHJTRV-UHFFFAOYSA-N 0.000 description 1
- SDGLRPMVJXYGDI-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(CC)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(CC)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 SDGLRPMVJXYGDI-UHFFFAOYSA-N 0.000 description 1
- YARDWBTUVRIEJY-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 YARDWBTUVRIEJY-UHFFFAOYSA-N 0.000 description 1
- HQXMDHFIKARXNB-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 HQXMDHFIKARXNB-UHFFFAOYSA-N 0.000 description 1
- OCZKOIDDGGOTFE-UHFFFAOYSA-N C=CC(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 OCZKOIDDGGOTFE-UHFFFAOYSA-N 0.000 description 1
- OTMQTUFAYWJAQK-QGZVFWFLSA-N C=CC(=O)N1CCC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 OTMQTUFAYWJAQK-QGZVFWFLSA-N 0.000 description 1
- CPLHMHFPIOQAIA-SFHVURJKSA-N C=CC(=O)N1CCC[C@H](N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC[C@H](N(C)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 CPLHMHFPIOQAIA-SFHVURJKSA-N 0.000 description 1
- FUZYFBTULWSPJW-IBGZPJMESA-N C=CC(=O)N1CCC[C@H](N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC[C@H](N(CC)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 FUZYFBTULWSPJW-IBGZPJMESA-N 0.000 description 1
- YARDWBTUVRIEJY-NRFANRHFSA-N C=CC(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 YARDWBTUVRIEJY-NRFANRHFSA-N 0.000 description 1
- OCZKOIDDGGOTFE-IBGZPJMESA-N C=CC(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 OCZKOIDDGGOTFE-IBGZPJMESA-N 0.000 description 1
- OTMQTUFAYWJAQK-KRWDZBQOSA-N C=CC(=O)N1CCC[C@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CCC[C@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 OTMQTUFAYWJAQK-KRWDZBQOSA-N 0.000 description 1
- NNVNSOVRINPZRI-MRXNPFEDSA-N C=CC(=O)N1CC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC[C@@H](NC2=CC(C3=CC(CC)=NC=C3)=CNC2=O)C1 NNVNSOVRINPZRI-MRXNPFEDSA-N 0.000 description 1
- CAAJFWLBVLMCLZ-HNNXBMFYSA-N C=CC(=O)N1CC[C@H](N(C)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 Chemical compound C=CC(=O)N1CC[C@H](N(C)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 CAAJFWLBVLMCLZ-HNNXBMFYSA-N 0.000 description 1
- WTWYPGLDDHWMBB-GRSHGNNSSA-N CC(C)(C)/C=C(/C#N)C(=O)N1CC(CC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)(C)/C=C(/C#N)C(=O)N1CC(CC2=CC(C3=CC=NC=C3)=CNC2=O)C1 WTWYPGLDDHWMBB-GRSHGNNSSA-N 0.000 description 1
- RECIVFMVICDRDA-FNORWQNLSA-N CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=N3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=N3)=CNC2=O)C1 RECIVFMVICDRDA-FNORWQNLSA-N 0.000 description 1
- MNGVKVGWBQNJMZ-ONEGZZNKSA-N CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 MNGVKVGWBQNJMZ-ONEGZZNKSA-N 0.000 description 1
- LHNVNQLEMVXINJ-ONEGZZNKSA-N CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 LHNVNQLEMVXINJ-ONEGZZNKSA-N 0.000 description 1
- SBYORDBWLBVADN-ONEGZZNKSA-N CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 SBYORDBWLBVADN-ONEGZZNKSA-N 0.000 description 1
- HVPVJWYHLVBICY-ONEGZZNKSA-N CC(C)NC/C=C/C(=O)N1CCC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CCC(NC2=CC(C3=CC=NC=C3)=CNC2=O)C1 HVPVJWYHLVBICY-ONEGZZNKSA-N 0.000 description 1
- ZEHZMFCKPRLTQV-ONEGZZNKSA-N CC(C)NC/C=C/C(=O)N1CCC(NC2=CC(C3=CC=NC=C3)=CNC2=O)CC1 Chemical compound CC(C)NC/C=C/C(=O)N1CCC(NC2=CC(C3=CC=NC=C3)=CNC2=O)CC1 ZEHZMFCKPRLTQV-ONEGZZNKSA-N 0.000 description 1
- WBQLHWNABQQXHB-ZZXKWVIFSA-N CC(C)NC/C=C/C(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 WBQLHWNABQQXHB-ZZXKWVIFSA-N 0.000 description 1
- WBQLHWNABQQXHB-DLMBFZKISA-N CC(C)NC/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 WBQLHWNABQQXHB-DLMBFZKISA-N 0.000 description 1
- BERFKKCFJWJNSG-LFTXJPJQSA-N CC(C)NC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 BERFKKCFJWJNSG-LFTXJPJQSA-N 0.000 description 1
- BERFKKCFJWJNSG-SIAOZKNOSA-N CC(C)NC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CC(C)NC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 BERFKKCFJWJNSG-SIAOZKNOSA-N 0.000 description 1
- DIFZYKPFQRXWFD-VOTSOKGWSA-N CC1=C(C(C)C)C=CC(NC(=O)C2=CC(C3=CCC(=O)C(NC4CN(C(=O)/C=C/CN(C)C)C4)=C3)=CC(C#N)=C2)=C1 Chemical compound CC1=C(C(C)C)C=CC(NC(=O)C2=CC(C3=CCC(=O)C(NC4CN(C(=O)/C=C/CN(C)C)C4)=C3)=CC(C#N)=C2)=C1 DIFZYKPFQRXWFD-VOTSOKGWSA-N 0.000 description 1
- FDOQKAHVXOIWTN-JXMROGBWSA-N CC1=C(C(C)C)C=CC(NC(=O)C2=CC=CC(C3=CNC(=O)C(NC4CN(C(=O)/C=C/CN(C)C)C4)=C3)=C2)=C1 Chemical compound CC1=C(C(C)C)C=CC(NC(=O)C2=CC=CC(C3=CNC(=O)C(NC4CN(C(=O)/C=C/CN(C)C)C4)=C3)=C2)=C1 FDOQKAHVXOIWTN-JXMROGBWSA-N 0.000 description 1
- WAMYBFQEKCSKKT-BQYQJAHWSA-N CC1=CC(C(=O)NC2=CC(C)=C(C(C)C)C=C2)=CC(C2=CCC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CC1=CC(C(=O)NC2=CC(C)=C(C(C)C)C=C2)=CC(C2=CCC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 WAMYBFQEKCSKKT-BQYQJAHWSA-N 0.000 description 1
- MHDKUTRUMJBNAI-AATRIKPKSA-N CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 MHDKUTRUMJBNAI-AATRIKPKSA-N 0.000 description 1
- VLXYWCOTJVXCCN-ONEGZZNKSA-N CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 VLXYWCOTJVXCCN-ONEGZZNKSA-N 0.000 description 1
- XBNHLCACKWFEIM-AATRIKPKSA-N CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 XBNHLCACKWFEIM-AATRIKPKSA-N 0.000 description 1
- BPYPJNHJYFBJOJ-SNAWJCMRSA-N CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 BPYPJNHJYFBJOJ-SNAWJCMRSA-N 0.000 description 1
- MVHGINOYFARQTH-AATRIKPKSA-N CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 MVHGINOYFARQTH-AATRIKPKSA-N 0.000 description 1
- PGQAJLUTQXPLIG-ONEGZZNKSA-N CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 PGQAJLUTQXPLIG-ONEGZZNKSA-N 0.000 description 1
- XAVQHIXXLXVBAM-AATRIKPKSA-N CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 XAVQHIXXLXVBAM-AATRIKPKSA-N 0.000 description 1
- QBQNHVPFPPMCPN-SNAWJCMRSA-N CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=C1 QBQNHVPFPPMCPN-SNAWJCMRSA-N 0.000 description 1
- AJZJFKQCJKBIDJ-VOTSOKGWSA-N CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 AJZJFKQCJKBIDJ-VOTSOKGWSA-N 0.000 description 1
- RRPLZMSHALPXKQ-SNAWJCMRSA-N CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 RRPLZMSHALPXKQ-SNAWJCMRSA-N 0.000 description 1
- DNWNJUNYBQHLJF-VOTSOKGWSA-N CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 DNWNJUNYBQHLJF-VOTSOKGWSA-N 0.000 description 1
- JZBHUBNBQKWTDF-AATRIKPKSA-N CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 JZBHUBNBQKWTDF-AATRIKPKSA-N 0.000 description 1
- UQQMADYOKQXLPZ-AATRIKPKSA-N CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 UQQMADYOKQXLPZ-AATRIKPKSA-N 0.000 description 1
- FIMGBHGGWJIUKQ-ONEGZZNKSA-N CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 FIMGBHGGWJIUKQ-ONEGZZNKSA-N 0.000 description 1
- JWBMXQACIUQANZ-AATRIKPKSA-N CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 JWBMXQACIUQANZ-AATRIKPKSA-N 0.000 description 1
- GZCOSXGOHBFOBX-SNAWJCMRSA-N CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 GZCOSXGOHBFOBX-SNAWJCMRSA-N 0.000 description 1
- PLMZADDFOKJQQF-AATRIKPKSA-N CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 PLMZADDFOKJQQF-AATRIKPKSA-N 0.000 description 1
- DBIAIJOJAIJJAV-ONEGZZNKSA-N CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=N1 DBIAIJOJAIJJAV-ONEGZZNKSA-N 0.000 description 1
- OAGCRLVNMXCTDZ-AATRIKPKSA-N CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 OAGCRLVNMXCTDZ-AATRIKPKSA-N 0.000 description 1
- IVDXSJWIJJALDV-SNAWJCMRSA-N CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=N1 Chemical compound CCC1=CN=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=N1 IVDXSJWIJJALDV-SNAWJCMRSA-N 0.000 description 1
- OWIINIHYCJPBBL-VOTSOKGWSA-N CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 OWIINIHYCJPBBL-VOTSOKGWSA-N 0.000 description 1
- FCQZPGBSSZUQCO-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 FCQZPGBSSZUQCO-SNAWJCMRSA-N 0.000 description 1
- OFHJHVFXTPMTCV-VOTSOKGWSA-N CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 OFHJHVFXTPMTCV-VOTSOKGWSA-N 0.000 description 1
- JIMOYDIBUCHSSH-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 JIMOYDIBUCHSSH-AATRIKPKSA-N 0.000 description 1
- GSLYCPUWHRJDRR-CSKARUKUSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 GSLYCPUWHRJDRR-CSKARUKUSA-N 0.000 description 1
- ZEZWDCULNSNVSN-BQYQJAHWSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 ZEZWDCULNSNVSN-BQYQJAHWSA-N 0.000 description 1
- VIMGLBGTQHXXFD-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CN)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CN)CC3)=C2)=C1 VIMGLBGTQHXXFD-AATRIKPKSA-N 0.000 description 1
- RMRPCBKXTGTGJH-BQYQJAHWSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 RMRPCBKXTGTGJH-BQYQJAHWSA-N 0.000 description 1
- XXWXBXNEWLWUOB-VOTSOKGWSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CNC)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)C3CCN(C(=O)/C=C/CNC)CC3)=C2)=C1 XXWXBXNEWLWUOB-VOTSOKGWSA-N 0.000 description 1
- UOHCYEILQDPKAZ-MUPIXQSSSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 UOHCYEILQDPKAZ-MUPIXQSSSA-N 0.000 description 1
- MJNOEZCWLDRPEC-BWAMLYQTSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 MJNOEZCWLDRPEC-BWAMLYQTSA-N 0.000 description 1
- KTXWNCZNQJEHAK-VHTFYLNKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 KTXWNCZNQJEHAK-VHTFYLNKSA-N 0.000 description 1
- GSLYCPUWHRJDRR-RCHCNPRSSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 GSLYCPUWHRJDRR-RCHCNPRSSA-N 0.000 description 1
- UOHCYEILQDPKAZ-FKIZTMCRSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 UOHCYEILQDPKAZ-FKIZTMCRSA-N 0.000 description 1
- MJNOEZCWLDRPEC-KXOMFBTKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 MJNOEZCWLDRPEC-KXOMFBTKSA-N 0.000 description 1
- KTXWNCZNQJEHAK-JLXBBBJOSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC)[C@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 KTXWNCZNQJEHAK-JLXBBBJOSA-N 0.000 description 1
- KFKQYYSZIJDBPB-SOFGYWHQSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 KFKQYYSZIJDBPB-SOFGYWHQSA-N 0.000 description 1
- DXYVCAVPIBTNIE-VMPITWQZSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 DXYVCAVPIBTNIE-VMPITWQZSA-N 0.000 description 1
- DWPHGPDZCYOAHV-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 DWPHGPDZCYOAHV-AATRIKPKSA-N 0.000 description 1
- KZXFXUGXYFZQBT-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN)CC3)=C2)=C1 KZXFXUGXYFZQBT-ONEGZZNKSA-N 0.000 description 1
- SBBDZBMIPUPHMR-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 SBBDZBMIPUPHMR-AATRIKPKSA-N 0.000 description 1
- IALAFYBEFLKVIL-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC)CC3)=C2)=C1 IALAFYBEFLKVIL-SNAWJCMRSA-N 0.000 description 1
- PLMGVRPFRMVRPB-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 PLMGVRPFRMVRPB-AATRIKPKSA-N 0.000 description 1
- WCUZARZXECZXAG-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 WCUZARZXECZXAG-AATRIKPKSA-N 0.000 description 1
- PORRXVCUZNVEEV-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 PORRXVCUZNVEEV-ONEGZZNKSA-N 0.000 description 1
- JKLGHGGLZYLBCT-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 JKLGHGGLZYLBCT-ONEGZZNKSA-N 0.000 description 1
- FYGCGIPPOXOAIS-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 FYGCGIPPOXOAIS-AATRIKPKSA-N 0.000 description 1
- PKCIPQJPLSSJFS-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 PKCIPQJPLSSJFS-AATRIKPKSA-N 0.000 description 1
- RMZDMKQEYQXQFH-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 RMZDMKQEYQXQFH-SNAWJCMRSA-N 0.000 description 1
- HEQAHEHENSSJEU-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=N1 HEQAHEHENSSJEU-SNAWJCMRSA-N 0.000 description 1
- OBRCJXPPMTVEOA-XRRIUQSZSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 OBRCJXPPMTVEOA-XRRIUQSZSA-N 0.000 description 1
- JJALWIZPIYPHEQ-KURNWNDPSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 JJALWIZPIYPHEQ-KURNWNDPSA-N 0.000 description 1
- KFKQYYSZIJDBPB-YFTVHVBSSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 KFKQYYSZIJDBPB-YFTVHVBSSA-N 0.000 description 1
- OBRCJXPPMTVEOA-PHUOZXCHSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 OBRCJXPPMTVEOA-PHUOZXCHSA-N 0.000 description 1
- DXYVCAVPIBTNIE-AAHGLTIQSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 DXYVCAVPIBTNIE-AAHGLTIQSA-N 0.000 description 1
- JJALWIZPIYPHEQ-ILHLOOHCSA-N CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 JJALWIZPIYPHEQ-ILHLOOHCSA-N 0.000 description 1
- YEDCTLAGODLOIF-VMPITWQZSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 YEDCTLAGODLOIF-VMPITWQZSA-N 0.000 description 1
- GUHNYVBZFVHBGU-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 GUHNYVBZFVHBGU-AATRIKPKSA-N 0.000 description 1
- ZPOOPDOVQCNONN-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CN)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CN)CC3)=C2)=C1 ZPOOPDOVQCNONN-ONEGZZNKSA-N 0.000 description 1
- YKGMFTAMSGEBAJ-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 YKGMFTAMSGEBAJ-AATRIKPKSA-N 0.000 description 1
- OMIMASWXTDVRFY-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC)CC3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC)CC3)=C2)=C1 OMIMASWXTDVRFY-SNAWJCMRSA-N 0.000 description 1
- RKDHKLCLTFMPMH-VOTSOKGWSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/C4=CC=CC=N4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/C4=CC=CC=N4)C3)=C2)=C1 RKDHKLCLTFMPMH-VOTSOKGWSA-N 0.000 description 1
- SEXWEMMAJCQVAN-VMPITWQZSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4=NC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4=NC=CC=C4)C3)=C2)=C1 SEXWEMMAJCQVAN-VMPITWQZSA-N 0.000 description 1
- ILGPSUCTXAXRMI-UXBLZVDNSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4CCCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4CCCCC4)C3)=C2)=C1 ILGPSUCTXAXRMI-UXBLZVDNSA-N 0.000 description 1
- MMXFEYDZRZZEJA-HWKANZROSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4CCOCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CC4CCOCC4)C3)=C2)=C1 MMXFEYDZRZZEJA-HWKANZROSA-N 0.000 description 1
- VLOPSLNZVZUUEQ-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 VLOPSLNZVZUUEQ-AATRIKPKSA-N 0.000 description 1
- FETIQIDJWFAWQT-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 FETIQIDJWFAWQT-ONEGZZNKSA-N 0.000 description 1
- ADVTVWCSMNOMJJ-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=N1 ADVTVWCSMNOMJJ-ONEGZZNKSA-N 0.000 description 1
- UWFZXRJRPVGDLC-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN4CCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN4CCCC4)C3)=C2)=C1 UWFZXRJRPVGDLC-AATRIKPKSA-N 0.000 description 1
- AJDWKZAPQSZEPD-VOTSOKGWSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN4CCCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN4CCCCC4)C3)=C2)=C1 AJDWKZAPQSZEPD-VOTSOKGWSA-N 0.000 description 1
- XFUHGWVMJRMPCO-ONEGZZNKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN4CCOCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN4CCOCC4)C3)=C2)=C1 XFUHGWVMJRMPCO-ONEGZZNKSA-N 0.000 description 1
- BIFMBWWJNALUIB-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 BIFMBWWJNALUIB-AATRIKPKSA-N 0.000 description 1
- GLCRQBXRDHRBEQ-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 GLCRQBXRDHRBEQ-AATRIKPKSA-N 0.000 description 1
- KBEFKLUBKKVTQD-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=C1 KBEFKLUBKKVTQD-SNAWJCMRSA-N 0.000 description 1
- LCBLZOWGFIBPHM-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=N1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=N1 LCBLZOWGFIBPHM-SNAWJCMRSA-N 0.000 description 1
- KGQSFXZRXXIDFC-AATRIKPKSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/COC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/COC(C)C)C3)=C2)=C1 KGQSFXZRXXIDFC-AATRIKPKSA-N 0.000 description 1
- JAMWPSBUNOACJN-SNAWJCMRSA-N CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/COC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/COC)C3)=C2)=C1 JAMWPSBUNOACJN-SNAWJCMRSA-N 0.000 description 1
- BZWJSWBJPQODDA-LQVUNVMMSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CC4=CC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CC4=CC=CC=C4)C3)=C2)=C1 BZWJSWBJPQODDA-LQVUNVMMSA-N 0.000 description 1
- JZNWPZWOMYWZTA-HAOIVFDCSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CC4=NC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CC4=NC=CC=C4)C3)=C2)=C1 JZNWPZWOMYWZTA-HAOIVFDCSA-N 0.000 description 1
- VXJDHQVJNYEBNF-CPSHHHPTSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 VXJDHQVJNYEBNF-CPSHHHPTSA-N 0.000 description 1
- WHEHNSMOEZKZIV-HNSSHINMSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 WHEHNSMOEZKZIV-HNSSHINMSA-N 0.000 description 1
- FIABSPHXUMOEPL-XOPAWIAISA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN4CCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN4CCCC4)C3)=C2)=C1 FIABSPHXUMOEPL-XOPAWIAISA-N 0.000 description 1
- RDMIVKZEGDGNDB-ISGGHPHESA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN4CCCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN4CCCCC4)C3)=C2)=C1 RDMIVKZEGDGNDB-ISGGHPHESA-N 0.000 description 1
- CGNAOBOGDAJDMN-UKKYDJSUSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN4CCOCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CN4CCOCC4)C3)=C2)=C1 CGNAOBOGDAJDMN-UKKYDJSUSA-N 0.000 description 1
- FCGTXYNPBBPWQP-ZLHKHBAESA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 FCGTXYNPBBPWQP-ZLHKHBAESA-N 0.000 description 1
- IMSZDYQXPVAEFR-MRXNPFEDSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)CCl)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCCN(C(=O)CCl)C3)=C2)=C1 IMSZDYQXPVAEFR-MRXNPFEDSA-N 0.000 description 1
- WBYNROBJSQOXMR-PQCQUMGKSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CC4=CC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CC4=CC=CC=C4)C3)=C2)=C1 WBYNROBJSQOXMR-PQCQUMGKSA-N 0.000 description 1
- MXHLJERCNYITQN-QNBZTBGESA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CC4=NC=CC=C4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CC4=NC=CC=C4)C3)=C2)=C1 MXHLJERCNYITQN-QNBZTBGESA-N 0.000 description 1
- FSMKPMILWWWPGK-ZMBJWTFHSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 FSMKPMILWWWPGK-ZMBJWTFHSA-N 0.000 description 1
- VCMGPKTWRRPVEW-REMYHPOOSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN)C3)=C2)=C1 VCMGPKTWRRPVEW-REMYHPOOSA-N 0.000 description 1
- RJYCATBGCKRKSC-YAQIIAKESA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN4CCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN4CCCC4)C3)=C2)=C1 RJYCATBGCKRKSC-YAQIIAKESA-N 0.000 description 1
- VBCOAAXMDWEPQG-XYXUZSOXSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN4CCCCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN4CCCCC4)C3)=C2)=C1 VBCOAAXMDWEPQG-XYXUZSOXSA-N 0.000 description 1
- PTVCMMXJDSTIPC-YEFOHOTDSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN4CCOCC4)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CN4CCOCC4)C3)=C2)=C1 PTVCMMXJDSTIPC-YEFOHOTDSA-N 0.000 description 1
- VICISNPWXVBLGW-SHCNBLLESA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)/C=C/CNC)C3)=C2)=C1 VICISNPWXVBLGW-SHCNBLLESA-N 0.000 description 1
- RKFUKAHUVBBXKP-OAHLLOKOSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)CCl)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@@H]3CCN(C(=O)CCl)C3)=C2)=C1 RKFUKAHUVBBXKP-OAHLLOKOSA-N 0.000 description 1
- VXJDHQVJNYEBNF-IEJURRBGSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 VXJDHQVJNYEBNF-IEJURRBGSA-N 0.000 description 1
- WHEHNSMOEZKZIV-PXPSOEEZSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 WHEHNSMOEZKZIV-PXPSOEEZSA-N 0.000 description 1
- YEDCTLAGODLOIF-FERLCFGISA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 YEDCTLAGODLOIF-FERLCFGISA-N 0.000 description 1
- FCGTXYNPBBPWQP-QTWIXLPOSA-N CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCCN(C(=O)/C=C/CNC)C3)=C2)=C1 FCGTXYNPBBPWQP-QTWIXLPOSA-N 0.000 description 1
- KJBUKLQJGSLTQJ-VOTSOKGWSA-N CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 KJBUKLQJGSLTQJ-VOTSOKGWSA-N 0.000 description 1
- IHUCYCYZJKKGTE-SNAWJCMRSA-N CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 IHUCYCYZJKKGTE-SNAWJCMRSA-N 0.000 description 1
- SDGBJFNOTWJXNQ-VOTSOKGWSA-N CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 SDGBJFNOTWJXNQ-VOTSOKGWSA-N 0.000 description 1
- QRRWRSCVXGCLJC-AATRIKPKSA-N CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 QRRWRSCVXGCLJC-AATRIKPKSA-N 0.000 description 1
- UXEKLTQCHLRKTD-AATRIKPKSA-N CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 UXEKLTQCHLRKTD-AATRIKPKSA-N 0.000 description 1
- NSIPMIVGYHDOJG-ONEGZZNKSA-N CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 NSIPMIVGYHDOJG-ONEGZZNKSA-N 0.000 description 1
- VEJQKANANFCZIZ-AATRIKPKSA-N CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 VEJQKANANFCZIZ-AATRIKPKSA-N 0.000 description 1
- PUBDNKMKNOTXGL-SNAWJCMRSA-N CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC)C3)=C2)=C1 PUBDNKMKNOTXGL-SNAWJCMRSA-N 0.000 description 1
- NAYMCXLXRZQYRF-AATRIKPKSA-N CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 NAYMCXLXRZQYRF-AATRIKPKSA-N 0.000 description 1
- MFCHEACKLVPJIC-ONEGZZNKSA-N CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 MFCHEACKLVPJIC-ONEGZZNKSA-N 0.000 description 1
- IUVNFDZQCHENBI-AATRIKPKSA-N CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 IUVNFDZQCHENBI-AATRIKPKSA-N 0.000 description 1
- ZPSNDYUETSSCEA-SNAWJCMRSA-N CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=C1 Chemical compound CCC1=NC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC)C3)=C2)=C1 ZPSNDYUETSSCEA-SNAWJCMRSA-N 0.000 description 1
- NSEKTWUUMNCLFV-RMKNXTFCSA-N CCN(C(CCC1)CN1C(/C=C/CNC(C)C)=O)C1=CC(c2ccnc(OC)c2)=CNC1=O Chemical compound CCN(C(CCC1)CN1C(/C=C/CNC(C)C)=O)C1=CC(c2ccnc(OC)c2)=CNC1=O NSEKTWUUMNCLFV-RMKNXTFCSA-N 0.000 description 1
- DRTWZSGCELXEIN-VOTSOKGWSA-N CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)C1 DRTWZSGCELXEIN-VOTSOKGWSA-N 0.000 description 1
- AZXPQOGLMHBLGE-ROUKRRSUSA-N CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC(C)C)C1 AZXPQOGLMHBLGE-ROUKRRSUSA-N 0.000 description 1
- AZXPQOGLMHBLGE-YJJPMGAVSA-N CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC(C)C)C1 AZXPQOGLMHBLGE-YJJPMGAVSA-N 0.000 description 1
- GBCRSLLHZVZJPM-QWNKOJSDSA-N CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC(NC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC)C1 GBCRSLLHZVZJPM-QWNKOJSDSA-N 0.000 description 1
- CPGZRXIPDSUTDT-VQHVLOKHSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCCN(C(=O)/C=C/CN(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCCN(C(=O)/C=C/CN(C)C)C1 CPGZRXIPDSUTDT-VQHVLOKHSA-N 0.000 description 1
- OLQROOMEFRNAJZ-VOTSOKGWSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)C1 OLQROOMEFRNAJZ-VOTSOKGWSA-N 0.000 description 1
- DONABGNEZVBABP-VOTSOKGWSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)CC1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)CC1 DONABGNEZVBABP-VOTSOKGWSA-N 0.000 description 1
- KRGJVFVCNSCOKU-VOTSOKGWSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC(C)C)CC1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC(C)C)CC1 KRGJVFVCNSCOKU-VOTSOKGWSA-N 0.000 description 1
- TWUQCAJUOAXJNE-AATRIKPKSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC)CC1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC)CC1 TWUQCAJUOAXJNE-AATRIKPKSA-N 0.000 description 1
- NSEKTWUUMNCLFV-VHTFYLNKSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 NSEKTWUUMNCLFV-VHTFYLNKSA-N 0.000 description 1
- SAKIVMHOSKKLQL-MTCRFPMVSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC)C1 SAKIVMHOSKKLQL-MTCRFPMVSA-N 0.000 description 1
- MMROEEAXOCVHIG-ROUKRRSUSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC(C)C)C1 MMROEEAXOCVHIG-ROUKRRSUSA-N 0.000 description 1
- KNTMMZXAKINJNH-DUMMIQTOSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC)C1 KNTMMZXAKINJNH-DUMMIQTOSA-N 0.000 description 1
- NSEKTWUUMNCLFV-JLXBBBJOSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 NSEKTWUUMNCLFV-JLXBBBJOSA-N 0.000 description 1
- KNTMMZXAKINJNH-QWNKOJSDSA-N CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC(OC)=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC)C1 KNTMMZXAKINJNH-QWNKOJSDSA-N 0.000 description 1
- SRGUZQVFEOFWCX-SOFGYWHQSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCCN(C(=O)/C=C/CN(C)C)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCCN(C(=O)/C=C/CN(C)C)C1 SRGUZQVFEOFWCX-SOFGYWHQSA-N 0.000 description 1
- VQCKTSWGABIXJP-AATRIKPKSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CN(C)C)C1 VQCKTSWGABIXJP-AATRIKPKSA-N 0.000 description 1
- HNBAFHSSTZLYPO-AATRIKPKSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC(C)C)CC1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)C1CCN(C(=O)/C=C/CNC(C)C)CC1 HNBAFHSSTZLYPO-AATRIKPKSA-N 0.000 description 1
- SNLMOARMGQTLGV-HNSSHINMSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CN)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CN)C1 SNLMOARMGQTLGV-HNSSHINMSA-N 0.000 description 1
- FMGKVLQFZGRUED-WKOQKXSESA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 FMGKVLQFZGRUED-WKOQKXSESA-N 0.000 description 1
- GZROCBWAKMNZGP-ZLHKHBAESA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCCN(C(=O)/C=C/CNC)C1 GZROCBWAKMNZGP-ZLHKHBAESA-N 0.000 description 1
- MZDFCXKXXLTOKH-SHCNBLLESA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@@H]1CCN(C(=O)/C=C/CNC)C1 MZDFCXKXXLTOKH-SHCNBLLESA-N 0.000 description 1
- FMGKVLQFZGRUED-FERLCFGISA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC(C)C)C1 FMGKVLQFZGRUED-FERLCFGISA-N 0.000 description 1
- GZROCBWAKMNZGP-QTWIXLPOSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCCN(C(=O)/C=C/CNC)C1 GZROCBWAKMNZGP-QTWIXLPOSA-N 0.000 description 1
- MENCGNLUYWOUSX-XJDXJNMNSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC(C)C)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC(C)C)C1 MENCGNLUYWOUSX-XJDXJNMNSA-N 0.000 description 1
- MZDFCXKXXLTOKH-WRFKIARRSA-N CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC)C1 Chemical compound CCN(C1=CC(C2=CC=NC=C2)=CNC1=O)[C@H]1CCN(C(=O)/C=C/CNC)C1 MZDFCXKXXLTOKH-WRFKIARRSA-N 0.000 description 1
- YBLOVFSYNHBTSO-FNORWQNLSA-N CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=N3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=N3)=CNC2=O)C1 YBLOVFSYNHBTSO-FNORWQNLSA-N 0.000 description 1
- JWGDPZKFIRWPAF-ONEGZZNKSA-N CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 JWGDPZKFIRWPAF-ONEGZZNKSA-N 0.000 description 1
- DENROYIRVBUOGG-ONEGZZNKSA-N CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 DENROYIRVBUOGG-ONEGZZNKSA-N 0.000 description 1
- GFTHAZOUADABBE-ONEGZZNKSA-N CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 GFTHAZOUADABBE-ONEGZZNKSA-N 0.000 description 1
- GNTINEWJCTXGIE-ONEGZZNKSA-N CN(C)C/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 GNTINEWJCTXGIE-ONEGZZNKSA-N 0.000 description 1
- KLKGSPNPHXSGCD-ONEGZZNKSA-N CN(C)C/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)CC1 Chemical compound CN(C)C/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)CC1 KLKGSPNPHXSGCD-ONEGZZNKSA-N 0.000 description 1
- FLUQWRLIRDDPAW-GQCTYLIASA-N CN(C)C/C=C/C(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CCCC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 FLUQWRLIRDDPAW-GQCTYLIASA-N 0.000 description 1
- GNTINEWJCTXGIE-SHTLVRLNSA-N CN(C)C/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CN(C)C/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 GNTINEWJCTXGIE-SHTLVRLNSA-N 0.000 description 1
- FLUQWRLIRDDPAW-UKKYDJSUSA-N CN(C)C/C=C/C(N(CCC1)C[C@@H]1N(CC1CC1)C1=CC(c2ccncc2)=CNC1=O)=O Chemical compound CN(C)C/C=C/C(N(CCC1)C[C@@H]1N(CC1CC1)C1=CC(c2ccncc2)=CNC1=O)=O FLUQWRLIRDDPAW-UKKYDJSUSA-N 0.000 description 1
- HDAUIBRXCSLJIY-JXMROGBWSA-N CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C(=C(C(=O)N)C=CC1)C1=CC(=C(C=C1)C(C)C)C)C Chemical compound CN(C/C=C/C(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C(=C(C(=O)N)C=CC1)C1=CC(=C(C=C1)C(C)C)C)C HDAUIBRXCSLJIY-JXMROGBWSA-N 0.000 description 1
- RLSNZUSRDRCYQP-SNAWJCMRSA-N CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 RLSNZUSRDRCYQP-SNAWJCMRSA-N 0.000 description 1
- LQUMEYWQSWOLMM-SNAWJCMRSA-N CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 LQUMEYWQSWOLMM-SNAWJCMRSA-N 0.000 description 1
- ZGJCKDYQKSKUIP-SNAWJCMRSA-N CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC(OC)=NC=C3)=CNC2=O)C1 ZGJCKDYQKSKUIP-SNAWJCMRSA-N 0.000 description 1
- IODRIJYAWXGLBU-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 IODRIJYAWXGLBU-ONEGZZNKSA-N 0.000 description 1
- XJZFSUCLBNYDRU-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(C)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 XJZFSUCLBNYDRU-ONEGZZNKSA-N 0.000 description 1
- NTMUVGOLDQBUOH-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(OC)=CC=N3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(OC)=CC=N3)=CNC2=O)C1 NTMUVGOLDQBUOH-ONEGZZNKSA-N 0.000 description 1
- WMXFXGQAMXYPMT-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 WMXFXGQAMXYPMT-ONEGZZNKSA-N 0.000 description 1
- BUJWJIKCNLCSBU-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 BUJWJIKCNLCSBU-ONEGZZNKSA-N 0.000 description 1
- HURDKALFPSNHJR-GQCTYLIASA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=N3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=N3)=CNC2=O)C1 HURDKALFPSNHJR-GQCTYLIASA-N 0.000 description 1
- BKXOFWPAHKGMJF-NSCUHMNNSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 BKXOFWPAHKGMJF-NSCUHMNNSA-N 0.000 description 1
- SHXUSCIWMDXMDC-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 SHXUSCIWMDXMDC-ONEGZZNKSA-N 0.000 description 1
- WGPNSTZAOVCGPK-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC(OC)=NC=C3)=CNC2=O)C1 WGPNSTZAOVCGPK-ONEGZZNKSA-N 0.000 description 1
- ADJLZQMCGIXLKN-NSCUHMNNSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=CN=C3)=CNC2=O)C1 ADJLZQMCGIXLKN-NSCUHMNNSA-N 0.000 description 1
- TUJZFNZMNSMFGO-NSCUHMNNSA-N CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=NC=NC=C3)=CNC2=O)C1 TUJZFNZMNSMFGO-NSCUHMNNSA-N 0.000 description 1
- LQISLEIINJJJPK-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(NC2=CC(C3=CC(OC)=CC=N3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(NC2=CC(C3=CC(OC)=CC=N3)=CNC2=O)C1 LQISLEIINJJJPK-ONEGZZNKSA-N 0.000 description 1
- YMDJSZGWUDWHRV-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 YMDJSZGWUDWHRV-ONEGZZNKSA-N 0.000 description 1
- QXKGKBQJXHSDMQ-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(NC2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(NC2=CC(C3=CC(OC)=NC=N3)=CNC2=O)C1 QXKGKBQJXHSDMQ-ONEGZZNKSA-N 0.000 description 1
- VISMEXOZUIQIQO-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(NC2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(NC2=CC(C3=NC(OC)=CN=C3)=CNC2=O)C1 VISMEXOZUIQIQO-ONEGZZNKSA-N 0.000 description 1
- TTXWPASACOPDAE-ONEGZZNKSA-N CNC/C=C/C(=O)N1CC(NC2=CC(C3=NC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC(NC2=CC(C3=NC(OC)=NC=C3)=CNC2=O)C1 TTXWPASACOPDAE-ONEGZZNKSA-N 0.000 description 1
- WRMZNLQTQIWRGY-ONEGZZNKSA-N CNC/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)CC1 Chemical compound CNC/C=C/C(=O)N1CCC(N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)CC1 WRMZNLQTQIWRGY-ONEGZZNKSA-N 0.000 description 1
- QKWFYTQKPHLKLF-ONEGZZNKSA-N CNC/C=C/C(=O)N1CCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 QKWFYTQKPHLKLF-ONEGZZNKSA-N 0.000 description 1
- RFVPADWHUITSNM-ONEGZZNKSA-N CNC/C=C/C(=O)N1CCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)CC1 Chemical compound CNC/C=C/C(=O)N1CCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)CC1 RFVPADWHUITSNM-ONEGZZNKSA-N 0.000 description 1
- MWCPJYAFTGMADB-ZZXKWVIFSA-N CNC/C=C/C(=O)N1CCCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCCC(NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 MWCPJYAFTGMADB-ZZXKWVIFSA-N 0.000 description 1
- VGQOVRMWRIWTNC-YIEJHSOVSA-N CNC/C=C/C(=O)N1CCC[C@@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCC[C@@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 VGQOVRMWRIWTNC-YIEJHSOVSA-N 0.000 description 1
- JJARQXJCVZSCPS-XGLIRFAXSA-N CNC/C=C/C(=O)N1CCC[C@@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCC[C@@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 JJARQXJCVZSCPS-XGLIRFAXSA-N 0.000 description 1
- VGQOVRMWRIWTNC-FHOSRQNYSA-N CNC/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 VGQOVRMWRIWTNC-FHOSRQNYSA-N 0.000 description 1
- JJARQXJCVZSCPS-RHRNASEZSA-N CNC/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 JJARQXJCVZSCPS-RHRNASEZSA-N 0.000 description 1
- MWCPJYAFTGMADB-ZVVFDCPKSA-N CNC/C=C/C(=O)N1CCC[C@H](NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CCC[C@H](NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 MWCPJYAFTGMADB-ZVVFDCPKSA-N 0.000 description 1
- HZDWZQYAHXIILK-WUUFJKRQSA-N CNC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC(NC)=NC=C3)=CNC2=O)C1 HZDWZQYAHXIILK-WUUFJKRQSA-N 0.000 description 1
- ZNMJTXPOAOHLFW-WUUFJKRQSA-N CNC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 ZNMJTXPOAOHLFW-WUUFJKRQSA-N 0.000 description 1
- ZNMJTXPOAOHLFW-YADGQWAMSA-N CNC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 ZNMJTXPOAOHLFW-YADGQWAMSA-N 0.000 description 1
- TXMGGIZWFPTIRT-NUALSEBGSA-N CNC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@H](N(CC2CC2)C2=CC(C3=CC=NC=C3)=CNC2=O)C1 TXMGGIZWFPTIRT-NUALSEBGSA-N 0.000 description 1
- QKWFYTQKPHLKLF-CWDCEQMOSA-N CNC/C=C/C(=O)N1CC[C@H](NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 Chemical compound CNC/C=C/C(=O)N1CC[C@H](NC2=CC(C3=CC(OC)=NC=C3)=CNC2=O)C1 QKWFYTQKPHLKLF-CWDCEQMOSA-N 0.000 description 1
- UTJKUUQPMZQJKI-ONEGZZNKSA-N CNC/C=C/C(N(C1)CC1NC1=CC(c2nccc(NC)c2)=CNC1=O)=O Chemical compound CNC/C=C/C(N(C1)CC1NC1=CC(c2nccc(NC)c2)=CNC1=O)=O UTJKUUQPMZQJKI-ONEGZZNKSA-N 0.000 description 1
- SPBUXLNTVRABKK-ONEGZZNKSA-N CNC/C=C/C(N(CC1)CCC1N(CC1CC1)C1=CC(c2ccnc(NC)c2)=CNC1=O)=O Chemical compound CNC/C=C/C(N(CC1)CCC1N(CC1CC1)C1=CC(c2ccnc(NC)c2)=CNC1=O)=O SPBUXLNTVRABKK-ONEGZZNKSA-N 0.000 description 1
- VIXNUVNEPWDMEE-SNAWJCMRSA-N CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 VIXNUVNEPWDMEE-SNAWJCMRSA-N 0.000 description 1
- MPAOMAZVFRHCKU-SNAWJCMRSA-N CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 MPAOMAZVFRHCKU-SNAWJCMRSA-N 0.000 description 1
- VIXNUVNEPWDMEE-FNEOHHHZSA-N CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 VIXNUVNEPWDMEE-FNEOHHHZSA-N 0.000 description 1
- MPAOMAZVFRHCKU-ZDUIWUOISA-N CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 MPAOMAZVFRHCKU-ZDUIWUOISA-N 0.000 description 1
- VLAXWQZTXYEYGY-SNAWJCMRSA-N CNC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 VLAXWQZTXYEYGY-SNAWJCMRSA-N 0.000 description 1
- CHTNQYSQVCYUMH-UHFFFAOYSA-N CNC1=NC=CC(C2=CNC(=O)C(NC3CNC3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(NC3CNC3)=C2)=C1 CHTNQYSQVCYUMH-UHFFFAOYSA-N 0.000 description 1
- RCWYPQSCTGGQRZ-BDUNBXCCSA-N CNC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 RCWYPQSCTGGQRZ-BDUNBXCCSA-N 0.000 description 1
- IMNLYEDQOPXPLZ-FAAWYNLUSA-N CNC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCN(C(=O)/C=C/C[NH-])C3)=C2)=C1 Chemical compound CNC1=NC=CC(C2=CNC(=O)C(N[C@H]3CCN(C(=O)/C=C/C[NH-])C3)=C2)=C1 IMNLYEDQOPXPLZ-FAAWYNLUSA-N 0.000 description 1
- VQMGQYHOFIKELL-SNAWJCMRSA-N CNc1cc(C(C=C2N(CC3CC3)C(C3)CN3C(/C=C/CN(C)C)=O)=CNC2=O)ncn1 Chemical compound CNc1cc(C(C=C2N(CC3CC3)C(C3)CN3C(/C=C/CN(C)C)=O)=CNC2=O)ncn1 VQMGQYHOFIKELL-SNAWJCMRSA-N 0.000 description 1
- LNJQWWLOMGPIKB-UHFFFAOYSA-N CNc1cc(C(C=C2N(CC3CC3)C(C3)CN3C(C=C)=O)=CNC2=O)ncc1 Chemical compound CNc1cc(C(C=C2N(CC3CC3)C(C3)CN3C(C=C)=O)=CNC2=O)ncc1 LNJQWWLOMGPIKB-UHFFFAOYSA-N 0.000 description 1
- CMAPGPNUEUJGKA-WEVVVXLNSA-N CNc1cc(C(C=C2NC(C3)CN3C(/C=C/CCOc3ccccc3)=O)=CNC2=O)ccn1 Chemical compound CNc1cc(C(C=C2NC(C3)CN3C(/C=C/CCOc3ccccc3)=O)=CNC2=O)ccn1 CMAPGPNUEUJGKA-WEVVVXLNSA-N 0.000 description 1
- MYRMZACZZJEKAK-SNAWJCMRSA-N CNc1cc(C(C=C2NC(C3)CN3C(/C=C/CN3CCCC3)=O)=CNC2=O)ccn1 Chemical compound CNc1cc(C(C=C2NC(C3)CN3C(/C=C/CN3CCCC3)=O)=CNC2=O)ccn1 MYRMZACZZJEKAK-SNAWJCMRSA-N 0.000 description 1
- IIMSDQFCYCLXKW-SNAWJCMRSA-N COC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=CC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 IIMSDQFCYCLXKW-SNAWJCMRSA-N 0.000 description 1
- XEJQAAFCENUECW-SNAWJCMRSA-N COC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 XEJQAAFCENUECW-SNAWJCMRSA-N 0.000 description 1
- LLXFKXWSRXYAQK-SNAWJCMRSA-N COC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=CC=NC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 LLXFKXWSRXYAQK-SNAWJCMRSA-N 0.000 description 1
- IBPLRARMIXUSLI-SNAWJCMRSA-N COC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound COC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 IBPLRARMIXUSLI-SNAWJCMRSA-N 0.000 description 1
- FMXRJNMSMRJRGV-SNAWJCMRSA-N COC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 Chemical compound COC1=CN=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=N1 FMXRJNMSMRJRGV-SNAWJCMRSA-N 0.000 description 1
- WGNUYVTUWHEHHZ-FNORWQNLSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 WGNUYVTUWHEHHZ-FNORWQNLSA-N 0.000 description 1
- VCWFFKFAAZCFQM-QPJJXVBHSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 VCWFFKFAAZCFQM-QPJJXVBHSA-N 0.000 description 1
- PPBLXZDSVSPYGL-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 PPBLXZDSVSPYGL-SNAWJCMRSA-N 0.000 description 1
- RENUNIOQRDHARI-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CN(C)C)CC3)=C2)=C1 RENUNIOQRDHARI-SNAWJCMRSA-N 0.000 description 1
- YOCKRVKXKBKWFI-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 YOCKRVKXKBKWFI-SNAWJCMRSA-N 0.000 description 1
- BRGNAZIWVIURQT-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CCN(C(=O)/C=C/CNC(C)C)CC3)=C2)=C1 BRGNAZIWVIURQT-SNAWJCMRSA-N 0.000 description 1
- WLZRKEZSWYZXJL-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 WLZRKEZSWYZXJL-SNAWJCMRSA-N 0.000 description 1
- QFUDCAQVXZNUJG-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=N1 QFUDCAQVXZNUJG-SNAWJCMRSA-N 0.000 description 1
- DPABHWXXZCNLLC-NSCUHMNNSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=N1 DPABHWXXZCNLLC-NSCUHMNNSA-N 0.000 description 1
- NLSMVPQQVHJTQL-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 NLSMVPQQVHJTQL-SNAWJCMRSA-N 0.000 description 1
- WGNUYVTUWHEHHZ-HVPBSWBLSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 WGNUYVTUWHEHHZ-HVPBSWBLSA-N 0.000 description 1
- VCWFFKFAAZCFQM-ILHLOOHCSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 VCWFFKFAAZCFQM-ILHLOOHCSA-N 0.000 description 1
- PPBLXZDSVSPYGL-FNEOHHHZSA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 PPBLXZDSVSPYGL-FNEOHHHZSA-N 0.000 description 1
- YOCKRVKXKBKWFI-ZDUIWUOISA-N COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(N(CC3CC3)[C@H]3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 YOCKRVKXKBKWFI-ZDUIWUOISA-N 0.000 description 1
- ANFCYMXXUIJGFX-FNORWQNLSA-N COC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 ANFCYMXXUIJGFX-FNORWQNLSA-N 0.000 description 1
- VTDAPTNNCBLVBR-GORDUTHDSA-N COC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CN)C3)=C2)=C1 VTDAPTNNCBLVBR-GORDUTHDSA-N 0.000 description 1
- YRAQTSYDYBRQAV-QPJJXVBHSA-N COC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(NC3CCCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 YRAQTSYDYBRQAV-QPJJXVBHSA-N 0.000 description 1
- BLWVVBIBRVSVGU-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 BLWVVBIBRVSVGU-SNAWJCMRSA-N 0.000 description 1
- IAYAAFUERRVDFQ-SNAWJCMRSA-N COC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(NC3CCN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 IAYAAFUERRVDFQ-SNAWJCMRSA-N 0.000 description 1
- NCBXPKYQUXGVDR-NSCUHMNNSA-N COC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound COC1=NC=CC(C2=CNC(=O)C(NC3CN(C(=O)/C=C/CN)C3)=C2)=C1 NCBXPKYQUXGVDR-NSCUHMNNSA-N 0.000 description 1
- YAQGKQFGSSBGOK-AATRIKPKSA-N COC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=NC(C2=CNC(=O)C(N(C)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 YAQGKQFGSSBGOK-AATRIKPKSA-N 0.000 description 1
- SNUZCRVZSFNQOI-SNAWJCMRSA-N COC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 Chemical compound COC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN(C)C)C3)=C2)=C1 SNUZCRVZSFNQOI-SNAWJCMRSA-N 0.000 description 1
- UPUMLMIUENEAQY-NSCUHMNNSA-N COC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 Chemical compound COC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CN)C3)=C2)=C1 UPUMLMIUENEAQY-NSCUHMNNSA-N 0.000 description 1
- WVZVESPERVILJZ-SNAWJCMRSA-N COC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 Chemical compound COC1=NC=NC(C2=CNC(=O)C(N(CC3CC3)C3CN(C(=O)/C=C/CNC(C)C)C3)=C2)=C1 WVZVESPERVILJZ-SNAWJCMRSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- KLWPJMFMVPTNCC-UHFFFAOYSA-N Camptothecin Natural products CCC1(O)C(=O)OCC2=C1C=C3C4Nc5ccccc5C=C4CN3C2=O KLWPJMFMVPTNCC-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 206010008263 Cervical dysplasia Diseases 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 241000723346 Cinnamomum camphora Species 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 206010053567 Coagulopathies Diseases 0.000 description 1
- 208000015943 Coeliac disease Diseases 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 102000000503 Collagen Type II Human genes 0.000 description 1
- 108010041390 Collagen Type II Proteins 0.000 description 1
- 208000032170 Congenital Abnormalities Diseases 0.000 description 1
- 206010010744 Conjunctivitis allergic Diseases 0.000 description 1
- MFYSYFVPBJMHGN-ZPOLXVRWSA-N Cortisone Chemical compound O=C1CC[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 MFYSYFVPBJMHGN-ZPOLXVRWSA-N 0.000 description 1
- MFYSYFVPBJMHGN-UHFFFAOYSA-N Cortisone Natural products O=C1CCC2(C)C3C(=O)CC(C)(C(CC4)(O)C(=O)CO)C4C3CCC2=C1 MFYSYFVPBJMHGN-UHFFFAOYSA-N 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- 102100024170 Cyclin-C Human genes 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- VVNCNSJFMMFHPL-VKHMYHEASA-N D-penicillamine Chemical group CC(C)(S)[C@@H](N)C(O)=O VVNCNSJFMMFHPL-VKHMYHEASA-N 0.000 description 1
- 238000011763 DBA/1J (JAX™ mouse strain) Methods 0.000 description 1
- 239000012623 DNA damaging agent Substances 0.000 description 1
- 206010061619 Deformity Diseases 0.000 description 1
- 206010012442 Dermatitis contact Diseases 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- BWLUMTFWVZZZND-UHFFFAOYSA-N Dibenzylamine Chemical compound C=1C=CC=CC=1CNCC1=CC=CC=C1 BWLUMTFWVZZZND-UHFFFAOYSA-N 0.000 description 1
- 239000004338 Dichlorodifluoromethane Substances 0.000 description 1
- XBPCUCUWBYBCDP-UHFFFAOYSA-N Dicyclohexylamine Chemical compound C1CCCCC1NC1CCCCC1 XBPCUCUWBYBCDP-UHFFFAOYSA-N 0.000 description 1
- 206010061818 Disease progression Diseases 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 241000792859 Enema Species 0.000 description 1
- 206010014989 Epidermolysis bullosa Diseases 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 208000006168 Ewing Sarcoma Diseases 0.000 description 1
- 208000027445 Farmer Lung Diseases 0.000 description 1
- 206010016228 Fasciitis Diseases 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- 208000005577 Gastroenteritis Diseases 0.000 description 1
- 208000007569 Giant Cell Tumors Diseases 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- 208000035895 Guillain-Barré syndrome Diseases 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- 208000031886 HIV Infections Diseases 0.000 description 1
- 208000037357 HIV infectious disease Diseases 0.000 description 1
- 208000030836 Hashimoto thyroiditis Diseases 0.000 description 1
- 208000017604 Hodgkin disease Diseases 0.000 description 1
- 208000021519 Hodgkin lymphoma Diseases 0.000 description 1
- 208000010747 Hodgkins lymphoma Diseases 0.000 description 1
- 101000897405 Homo sapiens B-lymphocyte antigen CD20 Proteins 0.000 description 1
- 101000980770 Homo sapiens Cyclin-C Proteins 0.000 description 1
- 101000998146 Homo sapiens Interleukin-17A Proteins 0.000 description 1
- 101000998151 Homo sapiens Interleukin-17F Proteins 0.000 description 1
- 101001090688 Homo sapiens Lymphocyte cytosolic protein 2 Proteins 0.000 description 1
- 108010001336 Horseradish Peroxidase Proteins 0.000 description 1
- 208000037147 Hypercalcaemia Diseases 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- 101150106931 IFNG gene Proteins 0.000 description 1
- 229940124790 IL-6 inhibitor Drugs 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- XDXDZDZNSLXDNA-TZNDIEGXSA-N Idarubicin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XDXDZDZNSLXDNA-TZNDIEGXSA-N 0.000 description 1
- XDXDZDZNSLXDNA-UHFFFAOYSA-N Idarubicin Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XDXDZDZNSLXDNA-UHFFFAOYSA-N 0.000 description 1
- 201000003838 Idiopathic interstitial pneumonia Diseases 0.000 description 1
- 206010021245 Idiopathic thrombocytopenic purpura Diseases 0.000 description 1
- 206010065390 Inflammatory pain Diseases 0.000 description 1
- 229940119178 Interleukin 1 receptor antagonist Drugs 0.000 description 1
- 102000003814 Interleukin-10 Human genes 0.000 description 1
- 108090000174 Interleukin-10 Proteins 0.000 description 1
- 102000003816 Interleukin-13 Human genes 0.000 description 1
- 108090000176 Interleukin-13 Proteins 0.000 description 1
- 102000013691 Interleukin-17 Human genes 0.000 description 1
- 108050003558 Interleukin-17 Proteins 0.000 description 1
- 102100033461 Interleukin-17A Human genes 0.000 description 1
- 102100033454 Interleukin-17F Human genes 0.000 description 1
- 102000000743 Interleukin-5 Human genes 0.000 description 1
- 108010002616 Interleukin-5 Proteins 0.000 description 1
- 208000009164 Islet Cell Adenoma Diseases 0.000 description 1
- 229940122245 Janus kinase inhibitor Drugs 0.000 description 1
- 208000009388 Job Syndrome Diseases 0.000 description 1
- 206010023198 Joint ankylosis Diseases 0.000 description 1
- 206010023230 Joint stiffness Diseases 0.000 description 1
- 208000007766 Kaposi sarcoma Diseases 0.000 description 1
- 208000008839 Kidney Neoplasms Diseases 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-L L-tartrate(2-) Chemical compound [O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O FEWJPZIEWOKRBE-JCYAYHJZSA-L 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- 239000005411 L01XE02 - Gefitinib Substances 0.000 description 1
- 239000002136 L01XE07 - Lapatinib Substances 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 239000004166 Lanolin Substances 0.000 description 1
- 206010024227 Lepromatous leprosy Diseases 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 102100034709 Lymphocyte cytosolic protein 2 Human genes 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- 208000030289 Lymphoproliferative disease Diseases 0.000 description 1
- 229940124647 MEK inhibitor Drugs 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- SBDNJUWAMKYJOX-UHFFFAOYSA-N Meclofenamic Acid Chemical compound CC1=CC=C(Cl)C(NC=2C(=CC=CC=2)C(O)=O)=C1Cl SBDNJUWAMKYJOX-UHFFFAOYSA-N 0.000 description 1
- 208000007054 Medullary Carcinoma Diseases 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- 208000019695 Migraine disease Diseases 0.000 description 1
- 206010049567 Miller Fisher syndrome Diseases 0.000 description 1
- 102000004232 Mitogen-Activated Protein Kinase Kinases Human genes 0.000 description 1
- 108090000744 Mitogen-Activated Protein Kinase Kinases Proteins 0.000 description 1
- 229930192392 Mitomycin Natural products 0.000 description 1
- 239000004909 Moisturizer Substances 0.000 description 1
- 229920000715 Mucilage Polymers 0.000 description 1
- 101100520226 Mus musculus Plcg1 gene Proteins 0.000 description 1
- VXXVMYBQVMYZNZ-UHFFFAOYSA-N N-(3-methyl-4-propan-2-ylphenyl)-3-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]-5-propan-2-ylbenzamide Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=C(C1)C(C)C VXXVMYBQVMYZNZ-UHFFFAOYSA-N 0.000 description 1
- KUEZODJBLUOVEM-UHFFFAOYSA-N N-(3-methyl-4-propan-2-ylphenyl)-3-[6-oxo-5-[(1-prop-2-enoylazetidin-3-yl)amino]-1H-pyridin-3-yl]-5-propylbenzamide Chemical compound C(C=C)(=O)N1CC(C1)NC1=CC(=CNC1=O)C=1C=C(C(=O)NC2=CC(=C(C=C2)C(C)C)C)C=C(C1)CCC KUEZODJBLUOVEM-UHFFFAOYSA-N 0.000 description 1
- QIAFMBKCNZACKA-UHFFFAOYSA-N N-benzoylglycine Chemical compound OC(=O)CNC(=O)C1=CC=CC=C1 QIAFMBKCNZACKA-UHFFFAOYSA-N 0.000 description 1
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 1
- 150000001204 N-oxides Chemical class 0.000 description 1
- VLFSEPZZPUMNOW-QPJJXVBHSA-N NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=C3)=CNC2=O)C1 Chemical compound NC/C=C/C(=O)N1CC(N(CC2CC2)C2=CC(C3=CC=CC=C3)=CNC2=O)C1 VLFSEPZZPUMNOW-QPJJXVBHSA-N 0.000 description 1
- VBWUBARCNWMFKV-RDLSKKMCSA-N NCC=CC(=O)N1CC(C1)N(C1=C/C(/C=NC1=O)=C/1NC=CC(=C1)NC)CC1CC1 Chemical compound NCC=CC(=O)N1CC(C1)N(C1=C/C(/C=NC1=O)=C/1NC=CC(=C1)NC)CC1CC1 VBWUBARCNWMFKV-RDLSKKMCSA-N 0.000 description 1
- CMWTZPSULFXXJA-UHFFFAOYSA-N Naproxen Natural products C1=C(C(C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-UHFFFAOYSA-N 0.000 description 1
- 206010028735 Nasal congestion Diseases 0.000 description 1
- 206010065673 Nephritic syndrome Diseases 0.000 description 1
- 206010029260 Neuroblastoma Diseases 0.000 description 1
- 208000005890 Neuroma Diseases 0.000 description 1
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical compound OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- KDCLDDTZHUXCNX-HNQUOIGGSA-N O=C1NC=C(C2=CC=NC=C2)C=C1NC1CN(C(=O)/C=C/CC2CCOCC2)C1 Chemical compound O=C1NC=C(C2=CC=NC=C2)C=C1NC1CN(C(=O)/C=C/CC2CCOCC2)C1 KDCLDDTZHUXCNX-HNQUOIGGSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 102000038030 PI3Ks Human genes 0.000 description 1
- 108091007960 PI3Ks Proteins 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 241001111421 Pannus Species 0.000 description 1
- 208000030852 Parasitic disease Diseases 0.000 description 1
- YNHIGQDRGKUECZ-UHFFFAOYSA-L PdCl2(PPh3)2 Substances [Cl-].[Cl-].[Pd+2].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 YNHIGQDRGKUECZ-UHFFFAOYSA-L 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 229920005372 Plexiglas® Polymers 0.000 description 1
- 229920003171 Poly (ethylene oxide) Chemical class 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 208000031951 Primary immunodeficiency Diseases 0.000 description 1
- 206010036774 Proctitis Diseases 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 208000033464 Reiter syndrome Diseases 0.000 description 1
- 206010038389 Renal cancer Diseases 0.000 description 1
- 201000000582 Retinoblastoma Diseases 0.000 description 1
- 206010039088 Rhinitis atrophic Diseases 0.000 description 1
- 125000005631 S-sulfonamido group Chemical group 0.000 description 1
- 229910006069 SO3H Inorganic materials 0.000 description 1
- 206010039491 Sarcoma Diseases 0.000 description 1
- 208000009359 Sezary Syndrome Diseases 0.000 description 1
- 208000021388 Sezary disease Diseases 0.000 description 1
- 208000021386 Sjogren Syndrome Diseases 0.000 description 1
- 208000000453 Skin Neoplasms Diseases 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- 208000021712 Soft tissue sarcoma Diseases 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 201000009594 Systemic Scleroderma Diseases 0.000 description 1
- 206010042953 Systemic sclerosis Diseases 0.000 description 1
- 230000006044 T cell activation Effects 0.000 description 1
- 208000031673 T-Cell Cutaneous Lymphoma Diseases 0.000 description 1
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 description 1
- 229940123237 Taxane Drugs 0.000 description 1
- 229940122388 Thrombin inhibitor Drugs 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 108700019146 Transgenes Proteins 0.000 description 1
- 206010052779 Transplant rejections Diseases 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-M Trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-M 0.000 description 1
- OKJPEAGHQZHRQV-UHFFFAOYSA-N Triiodomethane Natural products IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 1
- 206010067584 Type 1 diabetes mellitus Diseases 0.000 description 1
- 101710101516 Tyrosine-protein kinase TXK Proteins 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 208000024780 Urticaria Diseases 0.000 description 1
- 206010046851 Uveitis Diseases 0.000 description 1
- 206010047112 Vasculitides Diseases 0.000 description 1
- 206010047115 Vasculitis Diseases 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- 210000001766 X chromosome Anatomy 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- 229940119059 actemra Drugs 0.000 description 1
- 208000038016 acute inflammation Diseases 0.000 description 1
- 230000006022 acute inflammation Effects 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 229960002964 adalimumab Drugs 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- WNLRTRBMVRJNCN-UHFFFAOYSA-L adipate(2-) Chemical compound [O-]C(=O)CCCCC([O-])=O WNLRTRBMVRJNCN-UHFFFAOYSA-L 0.000 description 1
- 230000001919 adrenal effect Effects 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229930013930 alkaloid Natural products 0.000 description 1
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 1
- 125000005012 alkyl thioether group Chemical group 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 125000001118 alkylidene group Chemical group 0.000 description 1
- 201000009961 allergic asthma Diseases 0.000 description 1
- 208000002205 allergic conjunctivitis Diseases 0.000 description 1
- 201000010105 allergic rhinitis Diseases 0.000 description 1
- 235000020224 almond Nutrition 0.000 description 1
- 208000004631 alopecia areata Diseases 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 230000001668 ameliorated effect Effects 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 229960004238 anakinra Drugs 0.000 description 1
- 239000004037 angiogenesis inhibitor Substances 0.000 description 1
- 229940121369 angiogenesis inhibitor Drugs 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- 230000001387 anti-histamine Effects 0.000 description 1
- 239000000739 antihistaminic agent Substances 0.000 description 1
- 229940125715 antihistaminic agent Drugs 0.000 description 1
- 239000002220 antihypertensive agent Substances 0.000 description 1
- 229940030600 antihypertensive agent Drugs 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 239000003972 antineoplastic antibiotic Substances 0.000 description 1
- 229940045988 antineoplastic drug protein kinase inhibitors Drugs 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 239000008135 aqueous vehicle Substances 0.000 description 1
- 229940059756 arava Drugs 0.000 description 1
- 125000005018 aryl alkenyl group Chemical group 0.000 description 1
- 125000002102 aryl alkyloxo group Chemical group 0.000 description 1
- 125000005015 aryl alkynyl group Chemical group 0.000 description 1
- 125000001769 aryl amino group Chemical group 0.000 description 1
- 125000005110 aryl thio group Chemical group 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- 201000003710 autoimmune thrombocytopenic purpura Diseases 0.000 description 1
- 230000035578 autophosphorylation Effects 0.000 description 1
- 229940120638 avastin Drugs 0.000 description 1
- 229960003005 axitinib Drugs 0.000 description 1
- 229960002170 azathioprine Drugs 0.000 description 1
- 125000004567 azetidin-3-yl group Chemical group N1CC(C1)* 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- JUHORIMYRDESRB-UHFFFAOYSA-N benzathine Chemical compound C=1C=CC=CC=1CNCCNCC1=CC=CC=C1 JUHORIMYRDESRB-UHFFFAOYSA-N 0.000 description 1
- 229940077388 benzenesulfonate Drugs 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 229940050390 benzoate Drugs 0.000 description 1
- 125000002047 benzodioxolyl group Chemical group O1OC(C2=C1C=CC=C2)* 0.000 description 1
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 125000005874 benzothiadiazolyl group Chemical group 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- PUJDIJCNWFYVJX-UHFFFAOYSA-N benzyl carbamate Chemical compound NC(=O)OCC1=CC=CC=C1 PUJDIJCNWFYVJX-UHFFFAOYSA-N 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- 229960000397 bevacizumab Drugs 0.000 description 1
- MKCBRYIXFFGIKN-UHFFFAOYSA-N bicyclo[1.1.1]pentane Chemical compound C1C2CC1C2 MKCBRYIXFFGIKN-UHFFFAOYSA-N 0.000 description 1
- LPCWKMYWISGVSK-UHFFFAOYSA-N bicyclo[3.2.1]octane Chemical compound C1C2CCC1CCC2 LPCWKMYWISGVSK-UHFFFAOYSA-N 0.000 description 1
- 238000002306 biochemical method Methods 0.000 description 1
- 230000008512 biological response Effects 0.000 description 1
- 229960001561 bleomycin Drugs 0.000 description 1
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 208000015294 blood coagulation disease Diseases 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 125000005620 boronic acid group Chemical class 0.000 description 1
- 230000031709 bromination Effects 0.000 description 1
- 238000005893 bromination reaction Methods 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 210000000621 bronchi Anatomy 0.000 description 1
- 206010006451 bronchitis Diseases 0.000 description 1
- 229960002092 busulfan Drugs 0.000 description 1
- 230000001275 ca(2+)-mobilization Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- KVUAALJSMIVURS-ZEDZUCNESA-L calcium folinate Chemical compound [Ca+2].C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 KVUAALJSMIVURS-ZEDZUCNESA-L 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 230000003185 calcium uptake Effects 0.000 description 1
- KVUAALJSMIVURS-QNTKWALQSA-L calcium;(2s)-2-[[4-[[(6s)-2-amino-5-formyl-4-oxo-1,6,7,8-tetrahydropteridin-6-yl]methylamino]benzoyl]amino]pentanedioate Chemical compound [Ca+2].C([C@@H]1N(C=O)C=2C(=O)N=C(NC=2NC1)N)NC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 KVUAALJSMIVURS-QNTKWALQSA-L 0.000 description 1
- 229960000846 camphor Drugs 0.000 description 1
- 229930008380 camphor Natural products 0.000 description 1
- MIOPJNTWMNEORI-UHFFFAOYSA-N camphorsulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)CC1C2(C)C MIOPJNTWMNEORI-UHFFFAOYSA-N 0.000 description 1
- 229940088954 camptosar Drugs 0.000 description 1
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical group C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 description 1
- 229940127093 camptothecin Drugs 0.000 description 1
- 239000003560 cancer drug Substances 0.000 description 1
- 238000002619 cancer immunotherapy Methods 0.000 description 1
- 229930003827 cannabinoid Natural products 0.000 description 1
- 239000003557 cannabinoid Substances 0.000 description 1
- 229940001981 carac Drugs 0.000 description 1
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229960004424 carbon dioxide Drugs 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-N carbonic acid monoamide Natural products NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 1
- WIKQEUJFZPCFNJ-UHFFFAOYSA-N carbonic acid;silver Chemical compound [Ag].[Ag].OC(O)=O WIKQEUJFZPCFNJ-UHFFFAOYSA-N 0.000 description 1
- 125000005518 carboxamido group Chemical group 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 150000001734 carboxylic acid salts Chemical class 0.000 description 1
- 208000002458 carcinoid tumor Diseases 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 239000012876 carrier material Substances 0.000 description 1
- 229940047475 cataflam Drugs 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 230000011712 cell development Effects 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 238000012054 celltiter-glo Methods 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 229960002152 chlorhexidine acetate Drugs 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 125000004617 chromonyl group Chemical group O1C(=CC(C2=CC=CC=C12)=O)* 0.000 description 1
- 230000006020 chronic inflammation Effects 0.000 description 1
- 208000037893 chronic inflammatory disorder Diseases 0.000 description 1
- 201000009151 chronic rhinitis Diseases 0.000 description 1
- 208000027157 chronic rhinosinusitis Diseases 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 230000009852 coagulant defect Effects 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 210000001072 colon Anatomy 0.000 description 1
- 238000012875 competitive assay Methods 0.000 description 1
- 239000007891 compressed tablet Substances 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- 229960004544 cortisone Drugs 0.000 description 1
- 229940088547 cosmegen Drugs 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 125000000332 coumarinyl group Chemical group O1C(=O)C(=CC2=CC=CC=C12)* 0.000 description 1
- 239000007819 coupling partner Substances 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 201000007241 cutaneous T cell lymphoma Diseases 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 1
- 230000001461 cytolytic effect Effects 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 229960000640 dactinomycin Drugs 0.000 description 1
- 229940070230 daypro Drugs 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 239000003405 delayed action preparation Substances 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 229960003957 dexamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 1
- PXBRQCKWGAHEHS-UHFFFAOYSA-N dichlorodifluoromethane Chemical compound FC(F)(Cl)Cl PXBRQCKWGAHEHS-UHFFFAOYSA-N 0.000 description 1
- 235000019404 dichlorodifluoromethane Nutrition 0.000 description 1
- 229940042935 dichlorodifluoromethane Drugs 0.000 description 1
- 125000006003 dichloroethyl group Chemical group 0.000 description 1
- 125000004774 dichlorofluoromethyl group Chemical group FC(Cl)(Cl)* 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- 229940087091 dichlorotetrafluoroethane Drugs 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- 229960004515 diclofenac potassium Drugs 0.000 description 1
- 229960001193 diclofenac sodium Drugs 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 125000004177 diethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 125000006001 difluoroethyl group Chemical group 0.000 description 1
- LTVOKYUPTHZZQH-UHFFFAOYSA-N difluoromethane Chemical group F[C]F LTVOKYUPTHZZQH-UHFFFAOYSA-N 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 125000005433 dihydrobenzodioxinyl group Chemical group O1C(COC2=C1C=CC=C2)* 0.000 description 1
- 125000001070 dihydroindolyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000004611 dihydroisoindolyl group Chemical group C1(NCC2=CC=CC=C12)* 0.000 description 1
- 125000005045 dihydroisoquinolinyl group Chemical group C1(NC=CC2=CC=CC=C12)* 0.000 description 1
- 125000004655 dihydropyridinyl group Chemical group N1(CC=CC=C1)* 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- GAFRWLVTHPVQGK-UHFFFAOYSA-N dipentyl sulfate Chemical class CCCCCOS(=O)(=O)OCCCCC GAFRWLVTHPVQGK-UHFFFAOYSA-N 0.000 description 1
- 208000037765 diseases and disorders Diseases 0.000 description 1
- VSJKWCGYPAHWDS-UHFFFAOYSA-N dl-camptothecin Natural products C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-UHFFFAOYSA-N 0.000 description 1
- 239000003534 dna topoisomerase inhibitor Substances 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 229940120655 eloxatin Drugs 0.000 description 1
- 108010075324 emt protein-tyrosine kinase Proteins 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 229940073621 enbrel Drugs 0.000 description 1
- 210000002889 endothelial cell Anatomy 0.000 description 1
- 210000003038 endothelium Anatomy 0.000 description 1
- 239000007920 enema Substances 0.000 description 1
- 229940079360 enema for constipation Drugs 0.000 description 1
- 125000004050 enoyl group Chemical group 0.000 description 1
- 238000001952 enzyme assay Methods 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 description 1
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 description 1
- 210000002615 epidermis Anatomy 0.000 description 1
- 229960000403 etanercept Drugs 0.000 description 1
- 125000005678 ethenylene group Chemical group [H]C([*:1])=C([H])[*:2] 0.000 description 1
- 125000005677 ethinylene group Chemical group [*:2]C#C[*:1] 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 125000004672 ethylcarbonyl group Chemical group [H]C([H])([H])C([H])([H])C(*)=O 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 description 1
- 229960005420 etoposide Drugs 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 208000022195 farmer lung disease Diseases 0.000 description 1
- 235000019197 fats Nutrition 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 229960001419 fenoprofen Drugs 0.000 description 1
- RDJGLLICXDHJDY-UHFFFAOYSA-N fenoprofen Chemical compound OC(=O)C(C)C1=CC=CC(OC=2C=CC=CC=2)=C1 RDJGLLICXDHJDY-UHFFFAOYSA-N 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- 235000008191 folinic acid Nutrition 0.000 description 1
- 239000011672 folinic acid Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 229950005309 fostamatinib Drugs 0.000 description 1
- GKDRMWXFWHEQQT-UHFFFAOYSA-N fostamatinib Chemical compound COC1=C(OC)C(OC)=CC(NC=2N=C(NC=3N=C4N(COP(O)(O)=O)C(=O)C(C)(C)OC4=CC=3)C(F)=CN=2)=C1 GKDRMWXFWHEQQT-UHFFFAOYSA-N 0.000 description 1
- 125000004612 furopyridinyl group Chemical group O1C(=CC2=C1C=CC=N2)* 0.000 description 1
- 229940011343 fusilev Drugs 0.000 description 1
- 210000000232 gallbladder Anatomy 0.000 description 1
- 201000008361 ganglioneuroma Diseases 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- XGALLCVXEZPNRQ-UHFFFAOYSA-N gefitinib Chemical compound C=12C=C(OCCCN3CCOCC3)C(OC)=CC2=NC=NC=1NC1=CC=C(F)C(Cl)=C1 XGALLCVXEZPNRQ-UHFFFAOYSA-N 0.000 description 1
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 229940114119 gentisate Drugs 0.000 description 1
- 208000005017 glioblastoma Diseases 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- JFCQEDHGNNZCLN-UHFFFAOYSA-N glutaric acid Chemical compound OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 description 1
- 125000005456 glyceride group Chemical group 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 210000003714 granulocyte Anatomy 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 125000000262 haloalkenyl group Chemical group 0.000 description 1
- 125000004995 haloalkylthio group Chemical group 0.000 description 1
- 125000000232 haloalkynyl group Chemical group 0.000 description 1
- 210000003128 head Anatomy 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 208000014951 hematologic disease Diseases 0.000 description 1
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 1
- 208000018706 hematopoietic system disease Diseases 0.000 description 1
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 1
- 231100000844 hepatocellular carcinoma Toxicity 0.000 description 1
- 125000006343 heptafluoro propyl group Chemical group 0.000 description 1
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical compound CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 208000033519 human immunodeficiency virus infectious disease Diseases 0.000 description 1
- 229940048921 humira Drugs 0.000 description 1
- 229940088013 hycamtin Drugs 0.000 description 1
- 125000000717 hydrazino group Chemical group [H]N([*])N([H])[H] 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000003840 hydrochlorides Chemical class 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 230000003301 hydrolyzing effect Effects 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 206010051040 hyper-IgE syndrome Diseases 0.000 description 1
- 230000000917 hyperalgesic effect Effects 0.000 description 1
- 230000000148 hypercalcaemia Effects 0.000 description 1
- 208000030915 hypercalcemia disease Diseases 0.000 description 1
- 230000004047 hyperresponsiveness Effects 0.000 description 1
- 230000009610 hypersensitivity Effects 0.000 description 1
- 230000001969 hypertrophic effect Effects 0.000 description 1
- 229960001680 ibuprofen Drugs 0.000 description 1
- 229960000908 idarubicin Drugs 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000008938 immune dysregulation Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 229960003444 immunosuppressant agent Drugs 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 230000003116 impacting effect Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000003406 indolizinyl group Chemical group C=1(C=CN2C=CC=CC12)* 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 229960000598 infliximab Drugs 0.000 description 1
- 208000037797 influenza A Diseases 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 229940005319 inlyta Drugs 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 230000014828 interferon-gamma production Effects 0.000 description 1
- 230000004073 interleukin-2 production Effects 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 230000026045 iodination Effects 0.000 description 1
- 238000006192 iodination reaction Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- PGHMRUGBZOYCAA-ADZNBVRBSA-N ionomycin Chemical compound O1[C@H](C[C@H](O)[C@H](C)[C@H](O)[C@H](C)/C=C/C[C@@H](C)C[C@@H](C)C(/O)=C/C(=O)[C@@H](C)C[C@@H](C)C[C@@H](CCC(O)=O)C)CC[C@@]1(C)[C@@H]1O[C@](C)([C@@H](C)O)CC1 PGHMRUGBZOYCAA-ADZNBVRBSA-N 0.000 description 1
- PGHMRUGBZOYCAA-UHFFFAOYSA-N ionomycin Natural products O1C(CC(O)C(C)C(O)C(C)C=CCC(C)CC(C)C(O)=CC(=O)C(C)CC(C)CC(CCC(O)=O)C)CCC1(C)C1OC(C)(C(C)O)CC1 PGHMRUGBZOYCAA-UHFFFAOYSA-N 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229940084651 iressa Drugs 0.000 description 1
- 229960004768 irinotecan Drugs 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 201000002529 islet cell tumor Diseases 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001261 isocyanato group Chemical group *N=C=O 0.000 description 1
- 125000004594 isoindolinyl group Chemical group C1(NCC2=CC=CC=C12)* 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- FZWBNHMXJMCXLU-BLAUPYHCSA-N isomaltotriose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O)O1 FZWBNHMXJMCXLU-BLAUPYHCSA-N 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000001810 isothiocyanato group Chemical group *N=C=S 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- DKYWVDODHFEZIM-UHFFFAOYSA-N ketoprofen Chemical compound OC(=O)C(C)C1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 DKYWVDODHFEZIM-UHFFFAOYSA-N 0.000 description 1
- 229960000991 ketoprofen Drugs 0.000 description 1
- BWHLPLXXIDYSNW-UHFFFAOYSA-N ketorolac tromethamine Chemical compound OCC(N)(CO)CO.OC(=O)C1CCN2C1=CC=C2C(=O)C1=CC=CC=C1 BWHLPLXXIDYSNW-UHFFFAOYSA-N 0.000 description 1
- 201000010982 kidney cancer Diseases 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 229940043355 kinase inhibitor Drugs 0.000 description 1
- 229940054136 kineret Drugs 0.000 description 1
- 239000004922 lacquer Substances 0.000 description 1
- 235000019388 lanolin Nutrition 0.000 description 1
- 229940039717 lanolin Drugs 0.000 description 1
- BCFGMOOMADDAQU-UHFFFAOYSA-N lapatinib Chemical compound O1C(CNCCS(=O)(=O)C)=CC=C1C1=CC=C(N=CN=C2NC=3C=C(Cl)C(OCC=4C=C(F)C=CC=4)=CC=3)C2=C1 BCFGMOOMADDAQU-UHFFFAOYSA-N 0.000 description 1
- 206010023841 laryngeal neoplasm Diseases 0.000 description 1
- 229960000681 leflunomide Drugs 0.000 description 1
- 201000010260 leiomyoma Diseases 0.000 description 1
- 229960001691 leucovorin Drugs 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 239000003199 leukotriene receptor blocking agent Substances 0.000 description 1
- 201000011486 lichen planus Diseases 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 230000004807 localization Effects 0.000 description 1
- 210000003141 lower extremity Anatomy 0.000 description 1
- 230000001050 lubricating effect Effects 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000037841 lung tumor Diseases 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 230000000527 lymphocytic effect Effects 0.000 description 1
- 201000001268 lymphoproliferative syndrome Diseases 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 229910001629 magnesium chloride Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 208000008585 mastocytosis Diseases 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229940013798 meclofenamate Drugs 0.000 description 1
- 208000023356 medullary thyroid gland carcinoma Diseases 0.000 description 1
- 229960003464 mefenamic acid Drugs 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 230000001394 metastastic effect Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- RIFHJAODNHLCBH-UHFFFAOYSA-N methanethione Chemical group S=[CH] RIFHJAODNHLCBH-UHFFFAOYSA-N 0.000 description 1
- JZFLATQBIPILFS-UHFFFAOYSA-N methyl 4-bromopyridine-2-carboxylate Chemical compound COC(=O)C1=CC(Br)=CC=N1 JZFLATQBIPILFS-UHFFFAOYSA-N 0.000 description 1
- 125000000250 methylamino group Chemical group [H]N(*)C([H])([H])[H] 0.000 description 1
- 125000004674 methylcarbonyl group Chemical group CC(=O)* 0.000 description 1
- 229960004584 methylprednisolone Drugs 0.000 description 1
- 206010027599 migraine Diseases 0.000 description 1
- 239000002829 mitogen activated protein kinase inhibitor Substances 0.000 description 1
- 229960004857 mitomycin Drugs 0.000 description 1
- 229940019627 mitosol Drugs 0.000 description 1
- 230000000051 modifying effect Effects 0.000 description 1
- 230000000897 modulatory effect Effects 0.000 description 1
- 230000001333 moisturizer Effects 0.000 description 1
- 239000007932 molded tablet Substances 0.000 description 1
- 125000002911 monocyclic heterocycle group Chemical group 0.000 description 1
- 125000006578 monocyclic heterocycloalkyl group Chemical group 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 125000006682 monohaloalkyl group Chemical group 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 230000000869 mutational effect Effects 0.000 description 1
- 206010028417 myasthenia gravis Diseases 0.000 description 1
- 210000000066 myeloid cell Anatomy 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 229940090009 myleran Drugs 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- UQEIFYRRSNJVDO-UHFFFAOYSA-N n,n-dibenzyl-2-phenylethanamine Chemical compound C=1C=CC=CC=1CN(CC=1C=CC=CC=1)CCC1=CC=CC=C1 UQEIFYRRSNJVDO-UHFFFAOYSA-N 0.000 description 1
- LBWFXVZLPYTWQI-IPOVEDGCSA-N n-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-2-oxo-1h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C LBWFXVZLPYTWQI-IPOVEDGCSA-N 0.000 description 1
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical compound FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229960004270 nabumetone Drugs 0.000 description 1
- 229940089466 nalfon Drugs 0.000 description 1
- KVBGVZZKJNLNJU-UHFFFAOYSA-M naphthalene-2-sulfonate Chemical compound C1=CC=CC2=CC(S(=O)(=O)[O-])=CC=C21 KVBGVZZKJNLNJU-UHFFFAOYSA-M 0.000 description 1
- 229960002009 naproxen Drugs 0.000 description 1
- CMWTZPSULFXXJA-VIFPVBQESA-N naproxen Chemical compound C1=C([C@H](C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-VIFPVBQESA-N 0.000 description 1
- 229920001206 natural gum Polymers 0.000 description 1
- 210000000581 natural killer T-cell Anatomy 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- 210000003739 neck Anatomy 0.000 description 1
- 229940063121 neoral Drugs 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 235000001968 nicotinic acid Nutrition 0.000 description 1
- 239000011664 nicotinic acid Substances 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 102000037979 non-receptor tyrosine kinases Human genes 0.000 description 1
- 108091008046 non-receptor tyrosine kinases Proteins 0.000 description 1
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 230000037434 nonsense mutation Effects 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 239000003791 organic solvent mixture Substances 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 229960002739 oxaprozin Drugs 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 102000002574 p38 Mitogen-Activated Protein Kinases Human genes 0.000 description 1
- 108010068338 p38 Mitogen-Activated Protein Kinases Proteins 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 208000022102 pancreatic neuroendocrine neoplasm Diseases 0.000 description 1
- 229960005489 paracetamol Drugs 0.000 description 1
- 230000000849 parathyroid Effects 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 231100000915 pathological change Toxicity 0.000 description 1
- 230000036285 pathological change Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 239000003961 penetration enhancing agent Substances 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 229960002340 pentostatin Drugs 0.000 description 1
- FPVKHBSQESCIEP-JQCXWYLXSA-N pentostatin Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC[C@H]2O)=C2N=C1 FPVKHBSQESCIEP-JQCXWYLXSA-N 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 125000001151 peptidyl group Chemical group 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- JRKICGRDRMAZLK-UHFFFAOYSA-L peroxydisulfate Chemical compound [O-]S(=O)(=O)OOS([O-])(=O)=O JRKICGRDRMAZLK-UHFFFAOYSA-L 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 125000004934 phenanthridinyl group Chemical group C1(=CC=CC2=NC=C3C=CC=CC3=C12)* 0.000 description 1
- 125000004625 phenanthrolinyl group Chemical group N1=C(C=CC2=CC=C3C=CC=NC3=C12)* 0.000 description 1
- 125000005561 phenanthryl group Chemical group 0.000 description 1
- PDTFCHSETJBPTR-UHFFFAOYSA-N phenylmercuric nitrate Chemical compound [O-][N+](=O)O[Hg]C1=CC=CC=C1 PDTFCHSETJBPTR-UHFFFAOYSA-N 0.000 description 1
- 208000028591 pheochromocytoma Diseases 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 1
- 235000011007 phosphoric acid Nutrition 0.000 description 1
- 150000003016 phosphoric acids Chemical class 0.000 description 1
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 1
- DCWXELXMIBXGTH-UHFFFAOYSA-N phosphotyrosine Chemical compound OC(=O)C(N)CC1=CC=C(OP(O)(O)=O)C=C1 DCWXELXMIBXGTH-UHFFFAOYSA-N 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 229940075930 picrate Drugs 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-M picrate anion Chemical compound [O-]C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-M 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 229950010765 pivalate Drugs 0.000 description 1
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 description 1
- 210000004180 plasmocyte Anatomy 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 102000054765 polymorphisms of proteins Human genes 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 208000015768 polyposis Diseases 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229940072710 ponstel Drugs 0.000 description 1
- 230000029279 positive regulation of transcription, DNA-dependent Effects 0.000 description 1
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000002953 preparative HPLC Methods 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 208000030266 primary brain neoplasm Diseases 0.000 description 1
- 208000025638 primary cutaneous T-cell non-Hodgkin lymphoma Diseases 0.000 description 1
- 230000037452 priming Effects 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 239000003380 propellant Substances 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- MCSINKKTEDDPNK-UHFFFAOYSA-N propyl propionate Chemical compound CCCOC(=O)CC MCSINKKTEDDPNK-UHFFFAOYSA-N 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 239000003909 protein kinase inhibitor Substances 0.000 description 1
- 208000005069 pulmonary fibrosis Diseases 0.000 description 1
- 229940117820 purinethol Drugs 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- QLULGIRFKAWHOJ-UHFFFAOYSA-N pyridin-4-ylboronic acid Chemical compound OB(O)C1=CC=NC=C1 QLULGIRFKAWHOJ-UHFFFAOYSA-N 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 229940043131 pyroglutamate Drugs 0.000 description 1
- 125000001422 pyrrolinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 238000012797 qualification Methods 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 description 1
- 208000002574 reactive arthritis Diseases 0.000 description 1
- 108091006082 receptor inhibitors Proteins 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 230000007115 recruitment Effects 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 229940087462 relafen Drugs 0.000 description 1
- 230000027425 release of sequestered calcium ion into cytosol Effects 0.000 description 1
- 229940116176 remicade Drugs 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 208000023504 respiratory system disease Diseases 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 201000009410 rhabdomyosarcoma Diseases 0.000 description 1
- 102220023065 rs397514260 Human genes 0.000 description 1
- 239000012723 sample buffer Substances 0.000 description 1
- 229940063122 sandimmune Drugs 0.000 description 1
- 201000000306 sarcoidosis Diseases 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 208000017022 seasonal allergic rhinitis Diseases 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 229960004540 secukinumab Drugs 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 208000037851 severe atopic dermatitis Diseases 0.000 description 1
- 238000007493 shaping process Methods 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- KQTXIZHBFFWWFW-UHFFFAOYSA-L silver(I) carbonate Inorganic materials [Ag]OC(=O)O[Ag] KQTXIZHBFFWWFW-UHFFFAOYSA-L 0.000 description 1
- 201000009890 sinusitis Diseases 0.000 description 1
- 229960002930 sirolimus Drugs 0.000 description 1
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 description 1
- 201000008261 skin carcinoma Diseases 0.000 description 1
- 206010040882 skin lesion Diseases 0.000 description 1
- 231100000444 skin lesion Toxicity 0.000 description 1
- 208000000649 small cell carcinoma Diseases 0.000 description 1
- AWUCVROLDVIAJX-GSVOUGTGSA-N sn-glycerol 3-phosphate Chemical compound OC[C@@H](O)COP(O)(O)=O AWUCVROLDVIAJX-GSVOUGTGSA-N 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- BEOOHQFXGBMRKU-UHFFFAOYSA-N sodium cyanoborohydride Chemical compound [Na+].[B-]C#N BEOOHQFXGBMRKU-UHFFFAOYSA-N 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- OGPIIGMUPMPMNT-UHFFFAOYSA-M sodium meclofenamate (anhydrous) Chemical compound [Na+].CC1=CC=C(Cl)C(NC=2C(=CC=CC=2)C([O-])=O)=C1Cl OGPIIGMUPMPMNT-UHFFFAOYSA-M 0.000 description 1
- JGMJQSFLQWGYMQ-UHFFFAOYSA-M sodium;2,6-dichloro-n-phenylaniline;acetate Chemical compound [Na+].CC([O-])=O.ClC1=CC=CC(Cl)=C1NC1=CC=CC=C1 JGMJQSFLQWGYMQ-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 201000005671 spondyloarthropathy Diseases 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 229960001052 streptozocin Drugs 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- MLKXDPUZXIRXEP-MFOYZWKCSA-N sulindac Chemical compound CC1=C(CC(O)=O)C2=CC(F)=CC=C2\C1=C/C1=CC=C(S(C)=O)C=C1 MLKXDPUZXIRXEP-MFOYZWKCSA-N 0.000 description 1
- WINHZLLDWRZWRT-ATVHPVEESA-N sunitinib Chemical compound CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C WINHZLLDWRZWRT-ATVHPVEESA-N 0.000 description 1
- 229960001796 sunitinib Drugs 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000002511 suppository base Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 229940034785 sutent Drugs 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 229960001967 tacrolimus Drugs 0.000 description 1
- QJJXYPPXXYFBGM-SHYZHZOCSA-N tacrolimus Natural products CO[C@H]1C[C@H](CC[C@@H]1O)C=C(C)[C@H]2OC(=O)[C@H]3CCCCN3C(=O)C(=O)[C@@]4(O)O[C@@H]([C@H](C[C@H]4C)OC)[C@@H](C[C@H](C)CC(=C[C@@H](CC=C)C(=O)C[C@H](O)[C@H]2C)C)OC QJJXYPPXXYFBGM-SHYZHZOCSA-N 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 229940063683 taxotere Drugs 0.000 description 1
- 150000003505 terpenes Chemical class 0.000 description 1
- AKQXKEBCONUWCL-MRVPVSSYSA-N tert-butyl (3r)-3-aminopiperidine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CCC[C@@H](N)C1 AKQXKEBCONUWCL-MRVPVSSYSA-N 0.000 description 1
- CMIBWIAICVBURI-SSDOTTSWSA-N tert-butyl (3r)-3-aminopyrrolidine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CC[C@@H](N)C1 CMIBWIAICVBURI-SSDOTTSWSA-N 0.000 description 1
- VMKIXWAFFVLJCK-UHFFFAOYSA-N tert-butyl 3-oxoazetidine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CC(=O)C1 VMKIXWAFFVLJCK-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- CBXCPBUEXACCNR-UHFFFAOYSA-N tetraethylammonium Chemical compound CC[N+](CC)(CC)CC CBXCPBUEXACCNR-UHFFFAOYSA-N 0.000 description 1
- 125000003039 tetrahydroisoquinolinyl group Chemical group C1(NCCC2=CC=CC=C12)* 0.000 description 1
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 1
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 1
- 125000000147 tetrahydroquinolinyl group Chemical group N1(CCCC2=CC=CC=C12)* 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical compound C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 238000011285 therapeutic regimen Methods 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000002813 thiocarbonyl group Chemical group *C(*)=S 0.000 description 1
- 125000000858 thiocyanato group Chemical group *SC#N 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 239000003868 thrombin inhibitor Substances 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 238000002877 time resolved fluorescence resonance energy transfer Methods 0.000 description 1
- 229960003087 tioguanine Drugs 0.000 description 1
- KJAMZCVTJDTESW-UHFFFAOYSA-N tiracizine Chemical compound C1CC2=CC=CC=C2N(C(=O)CN(C)C)C2=CC(NC(=O)OCC)=CC=C21 KJAMZCVTJDTESW-UHFFFAOYSA-N 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- 229960003989 tocilizumab Drugs 0.000 description 1
- 229960001350 tofacitinib Drugs 0.000 description 1
- UJLAWZDWDVHWOW-YPMHNXCESA-N tofacitinib Chemical compound C[C@@H]1CCN(C(=O)CC#N)C[C@@H]1N(C)C1=NC=NC2=C1C=CN2 UJLAWZDWDVHWOW-YPMHNXCESA-N 0.000 description 1
- 230000024664 tolerance induction Effects 0.000 description 1
- 229960001017 tolmetin Drugs 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-M toluene-4-sulfonate Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-M 0.000 description 1
- 229940044693 topoisomerase inhibitor Drugs 0.000 description 1
- 229960000303 topotecan Drugs 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- 150000003626 triacylglycerols Chemical class 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 1
- 229940066528 trichloroacetate Drugs 0.000 description 1
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 1
- CYRMSUTZVYGINF-UHFFFAOYSA-N trichlorofluoromethane Chemical compound FC(Cl)(Cl)Cl CYRMSUTZVYGINF-UHFFFAOYSA-N 0.000 description 1
- 229940029284 trichlorofluoromethane Drugs 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- 125000005423 trihalomethanesulfonamido group Chemical group 0.000 description 1
- 125000005152 trihalomethanesulfonyl group Chemical group 0.000 description 1
- 125000004951 trihalomethoxy group Chemical group 0.000 description 1
- 201000008827 tuberculosis Diseases 0.000 description 1
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 1
- 229940094060 tykerb Drugs 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 description 1
- 239000005483 tyrosine kinase inhibitor Substances 0.000 description 1
- ZDPHROOEEOARMN-UHFFFAOYSA-N undecanoic acid Chemical compound CCCCCCCCCCC(O)=O ZDPHROOEEOARMN-UHFFFAOYSA-N 0.000 description 1
- 208000001319 vasomotor rhinitis Diseases 0.000 description 1
- 201000005539 vernal conjunctivitis Diseases 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 230000029812 viral genome replication Effects 0.000 description 1
- 229940063674 voltaren Drugs 0.000 description 1
- WAEXFXRVDQXREF-UHFFFAOYSA-N vorinostat Chemical compound ONC(=O)CCCCCCC(=O)NC1=CC=CC=C1 WAEXFXRVDQXREF-UHFFFAOYSA-N 0.000 description 1
- 229960000237 vorinostat Drugs 0.000 description 1
- 210000002268 wool Anatomy 0.000 description 1
- 125000001834 xanthenyl group Chemical group C1=CC=CC=2OC3=CC=CC=C3C(C12)* 0.000 description 1
- UGOMMVLRQDMAQQ-UHFFFAOYSA-N xphos Chemical compound CC(C)C1=CC(C(C)C)=CC(C(C)C)=C1C1=CC=CC=C1P(C1CCCCC1)C1CCCCC1 UGOMMVLRQDMAQQ-UHFFFAOYSA-N 0.000 description 1
- 229940053890 zanosar Drugs 0.000 description 1
- 235000005074 zinc chloride Nutrition 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/444—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a six-membered ring with nitrogen as a ring heteroatom, e.g. amrinone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/4545—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a six-membered ring with nitrogen as a ring hetero atom, e.g. pipamperone, anabasine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4965—Non-condensed pyrazines
- A61K31/497—Non-condensed pyrazines containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
Definitions
- the present disclosure relates to new arylpyridinone compounds and compositions, and their application as pharmaceuticals for the treatment of disease.
- Methods of inhibition of ITK activity in a human or animal subject are also provided for the treatment diseases such as those caused by inflammation.
- Tec tyrosine kinase expressed in hepatocellular carcinoma
- BMX bone marrow kinase on the X chromosome also known as ETK
- RLK resting lymphocyte kinase also known as TXK
- ITK interleukin-2 inducible T cell kdsinase, also known as EMT and TSK.
- TFTK have structural similarities to other non-receptor tyrosine kinases while exhibiting some family specific motifs resulting in a diversity of domain structures associated with complex localization, scaffolding and activation mechanisms.
- TFTK contain an amino terminal plekstrin homology domain (PH domain) involved in lipid interactions and membrane targeting followed by a BTK homology domain (BH) that binds Zn 2+ and an SH3 domain generally involved in proline rich domain binding.
- PH domain amino terminal plekstrin homology domain
- BH BTK homology domain
- TFTK expression is generally limited to hematopoietic lineage cells with the exception of ETK and TEC that are expressed in the liver and endothelial cells, respectively (Smith, et al. (2011) Bioessays 23, 436-446).
- ETK is expressed in monocytes, granulocytes and cardiac endothelium while BTK is expressed in B cells and mast cells but not plasma cells and T cells.
- TEC, RLK and ITK are all expressed in T cells. To date the TFTK with the most clear biological role in T cells is ITK.
- TCR T cell receptor
- TCR stimulation results in the activation of the kinase LCK and subsequent phosphorylation of the immunoreceptor tyrosine-based activation motifs (ITAMs) on CD3 inducing the binding and activation of the kinase ZAP70.
- ITAMs immunoreceptor tyrosine-based activation motifs
- ZAP70 phosphorylates the adaptor proteins LAT and SLP-76, which together with LCK and other proteins forms a hetermultimeric signaling complex that activates PI3K and generates PIP 3 on the plasma membrane.
- ITK binds to this signaling complex via SH2 and SH3 domains and to PIP 3 through its PH domain, resulting in LCK dependent phosphorylation of ITK Y511 and subsequent ITK autophosphorylation of Y180.
- Activated ITK phosphorylates PLC ⁇ 1 that, once activated, hydrolyzes PIP 2 to the second messengers IP3 and DAG.
- the cellular consequences of these sequelae of events include calcium mobilization and flux, PKC and MEK/ERK pathway activation, and transcriptional activation via APi, NF ⁇ activ NFAT.
- ITK As a critical enzyme in the TCR activation pathway ITK impacts T cell function in a number of ways including positive and negative selection, cellular differentiation, and cytokine production and release (Takesono, et al. (2002) J. Cell Science 115, 3039-3048; August, et al. (2012) Int. Rev. Immunol. 31, 155-165; Andreotti, et al. (2010) Cold Spring Harb. Perspect. Biol. 2, a002287 1-21).
- ITK insulin receptor kinase
- mice with a null mutation of the itk gene expressed a decreased number of mature T cells and a block in thymocyte development as well as a decreased TCR driven T cell proliferative response.
- IL2 and CD28 signaling as well as PMA/ionomycin driven responses remained unchanged, suggesting that the ITK response is membrane proximal and stimuli specific (Liao et al. (1995) Immunity 3, 757-769).
- ITK is responsible for amplification of TCR signaling versus an ‘on/off’ switch, as dual knockdown of the T cell expressing TFTK, ITK and RLK in mice produce a more complete TCR inactivation phenotype compared with ITK genetic deletion alone (Schaeffer et al. (1999) Science 284, 638-641). In contrast to the modulatory effect that ITK appears to have on na ⁇ ve T cell activation, it plays a more significant role in T helper cell differentiation.
- ITK deficient mice have demonstrated a reduction in the Th2 protective response to parasitic infection (Fowell et al. (1999) Immunity 11, 399-409; Schaeffer et al. (2001) Nat. Immunol.
- Th2 cytokines IL4, IL5, IL13 and IL10 were linked to a decrease in concentrations of Th2 cytokines IL4, IL5, IL13 and IL10 (Schaeffer et al. (2001) Nat. Immunol. 2, 1183-1188) and to a reduction in RLK expression.
- Th2 cytokines IL4, IL5, IL13 and IL10 Schott al. (2001) Nat. Immunol. 2, 1183-1188
- Th1 responses In contrast to the ITK requirement for mounting Th2 driven responses, its impact on Th1 responses is modest.
- IFNg production in ITK knockout cells is partially inhibited while the double ITK/RLK knockout has a more severe phenotype (Fowell et al. (1999) Immunity 11, 399-409; Schaeffer et al. (2001) Nat. Immunol. 2, 1183-1188; Miller et al.
- ITK knockout mice expressing an ITK kinase domain deleted transgene demonstrated that the kinase domain is essential for induction of a normal Th2 response (von Bonin et al. (2011) Exp. Dermatol. 20, 41-47).
- ITK expression and activity has recently been documented in studies characterizing individuals exhibiting mutations in the gene encoding this protein and or correlation between expression and disease.
- the ITK gene was found to be elevated in peripheral blood T cells from patients with moderate to severe atopic dermatitis, a Th2 driven chronic inflammatory skin disease (Hao et al. (2006) FEBS Letts., 580, 2691-2697).
- SNP disease-associated single nucleotide polymorphisms
- ITK knockout mice demonstrate reduced airway hypersensitivity and inflammation in models of allergic asthma (Stepensky et al. (2011) Haematologica, 96, 472-476; Mueller et al. (2003) J. Immunol., 170, 5056-5063; Ferrara et al. (2004) Pulm. Pharmacol. Ther. 17, 301-308).
- ITK deficient mice do not develop inflammation while ITK inhibition reduces the response in wild type mice (Ferrara et al. (2006) J. Allerg. Clin. Immunol. 117, 780-786).
- ITK dependent regulation of TCR dependent Ca 2+ mobilization and transcription factor induction makes it a critical factor in protecting against Influenza A and HIV infection and viral replication.
- ITK inhibitors have been shown to alter HIV replication at multiple stages and have the potential as effective HIV therapeutics (Sahu et al. (2008) J. Immunol. 180, 3833-3838).
- ITK inhibitors selectively target the killing of acute lymphblastic T-cell leukemia and cutaneous T-cell lymphoma while normal T cells are minimally impacted (Readinger et al. (2008) Proc. Nat. Acad. Sci. USA, 105, 6684-6689). ITK is highly expressed in transformed T-cell lines relative to normal T cells and other cancer cell lines. The impact of ITK inhibition on T cell tumors was confirmed in mouse xenograph models. Cancer evasion of the immune system as a result of tumor antigen tolerance induction versus priming is critical for tumor survival. Tumors that develop a microenvironment that induces T cell unresponsiveness demonstrate altered T cell gene expression suggesting skewing to the Th2 phenotype. ITK inhibition will favor Th1 differentiation and could be used to enhance cancer immunotherapy (Gao et al. (2012) Mol. Pharmacol., 82, 938-947; Horna et al. (2007) Curr. Cancer Drug Targ. 7, 41-53).
- the present disclosure provides a compound, or a pharmaceutically acceptable salt, hydrate or solvate thereof, of Formula (I):
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , C(O)(C(O)NH 2 )C ⁇ CH 2 , S(O) 2 CH ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH
- Certain compounds disclosed herein may possess useful ITK inhibiting activity, and may be used in the treatment or prophylaxis of a disease or condition in which ITK plays an active role.
- certain embodiments also provide pharmaceutical compositions comprising one or more compounds disclosed herein together with a pharmaceutically acceptable carrier, as well as methods of making and using the compounds and compositions.
- Certain embodiments provide methods for inhibiting ITK.
- Other embodiments provide methods for treating an ITK-mediated disorder in a subject in need of such treatment, comprising administering to the subject a therapeutically effective amount of a compound or composition according to the present disclosure.
- use of certain compounds disclosed herein for use in the manufacture of a medicament for the treatment of a disease or condition ameliorated by the inhibition of ITK.
- two embodiments are “mutually exclusive” when one is defined to be something which is different than the other.
- R 3 and R 4 combine to form a cycloalkyl is mutually exclusive with an embodiment in which R 3 is ethyl and R 4 is hydrogen.
- Y is CH 2
- R 3 and R 4 combine to form a cycloalkyl is mutually exclusive with an embodiment in which Y is CH 2 .
- any one of the listed items can be employed by itself or in combination with any one or more of the listed items.
- the expression “A and/or B” is intended to mean either or both of A and B, i.e. A alone, B alone or A and B in combination.
- the expression “A, B and/or C” is intended to mean A alone, B alone, C alone, A and B in combination, A and C in combination, B and C in combination or A, B, and C in combination.
- acyl refers to a carbonyl attached to an alkenyl, alkyl, aryl, cycloalkyl, heteroaryl, heterocycle, or any other moiety were the atom attached to the carbonyl is carbon.
- An “acetyl” group refers to a —C(O)CH 3 group.
- An “alkylcarbonyl” or “alkanoyl” group refers to an alkyl group attached to the parent molecular moiety through a carbonyl group. Examples of such groups include methylcarbonyl and ethylcarbonyl. Examples of acyl groups include formyl, alkanoyl and aroyl.
- alkenyl refers to a straight-chain or branched-chain hydrocarbon substituent having one or more double bonds and containing from 2 to 20 carbon atoms. In certain embodiments, the alkenyl will comprise from 2 to 6 carbon atoms.
- alkenylene refers to a carbon-carbon double bond system attached at two or more positions such as ethenylene [(—CH ⁇ CH—), (—C::C—)]. Examples of suitable alkenyl substituents include ethenyl, propenyl, 2-methylpropenyl, 1,4-butadienyl and the like. Unless otherwise specified, the term “alkenyl” may include “alkenylene” groups.
- alkoxy refers to an alkyl ether substituent, wherein the term alkyl is as defined below.
- suitable alkyl ether substituents include methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, iso-butoxy, sec-butoxy, tert-butoxy, and the like.
- alkyl refers to a straight-chain or branched-chain alkyl substituent containing from 1 to 20 carbon atoms. In certain embodiments, the alkyl will comprise from 1 to 10 carbon atoms. In further embodiments, the alkyl will comprise from 1 to 6 carbon atoms. Alkyl groups may be optionally substituted as defined herein. Examples of alkyl substituents include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, pentyl, iso-amyl, hexyl, octyl, noyl and the like.
- alkylene refers to a saturated aliphatic group derived from a straight or branched chain saturated hydrocarbon attached at two or more positions, such as methylene (—CH 2 —). Unless otherwise specified, the term “alkyl” may include “alkylene” groups.
- alkylamino refers to an alkyl group attached to the parent molecular moiety through an amino group. Suitable alkylamino groups may be mono- or dialkylated, forming groups such as, for example, N-methylamino, N-ethylamino, N,N-dimethylamino, N,N-ethylmethylamino and the like.
- alkylidene refers to an alkenyl group in which one carbon atom of the carbon-carbon double bond belongs to the moiety to which the alkenyl group is attached.
- alkylthio refers to an alkyl thioether (R—S—) substituent wherein the term alkyl is as defined above and wherein the sulfur may be singly or doubly oxidized.
- suitable alkyl thioether substituents include methylthio, ethylthio, n-propylthio, isopropylthio, n-butylthio, iso-butylthio, sec-butylthio, tert-butylthio, methanesulfonyl, ethanesulfinyl, and the like.
- alkynyl refers to a straight-chain or branched chain hydrocarbon substituent having one or more triple bonds and containing from 2 to 20 carbon atoms. In certain embodiments, the alkynyl comprises from 2 to 6 carbon atoms. In further embodiments, the alkynyl comprises from 2 to 4 carbon atoms.
- alkynylene refers to a carbon-carbon triple bond attached at two positions such as ethynylene (—C:::C—, —C ⁇ C—).
- alkynyl substituents include ethynyl, propynyl, hydroxypropynyl, butyn-1-yl, butyn-2-yl, pentyn-1-yl, 3-methylbutyn-1-yl, hexyn-2-yl, and the like.
- alkynyl may include “alkynylene” groups.
- amido and “carbamoyl” as used herein, alone or in combination, refer to an amino group as described below attached to the parent molecular moiety through a carbonyl group, or vice versa.
- C-amido refers to a C(O)N(RR′) group with R and R′ as defined herein or as defined by the specifically enumerated “R” groups designated.
- N-amido refers to a RC(O)N(R′)— group, with R and R′ as defined herein or as defined by the specifically enumerated “R” groups designated.
- acylamino as used herein, alone or in combination, embraces an acyl group attached to the parent moiety through an amino group.
- An example of an “acylamino” group is acetylamino (CH 3 C(O)NH—).
- amino refers to —NRR′, wherein R and R′ are independently chosen from hydrogen, alkyl, acyl, heteroalkyl, aryl, cycloalkyl, heteroaryl, and heterocycloalkyl, any of which may themselves be optionally substituted. Additionally, R and R′ may combine to form heterocycloalkyl, either of which may be optionally substituted.
- aryl as used herein, alone or in combination, means a carbocyclic aromatic system containing one, two or three rings wherein such polycyclic ring systems are fused together.
- aryl embraces aromatic groups such as phenyl, naphthyl, anthracenyl, and phenanthryl.
- arylalkenyl or “aralkenyl,” as used herein, alone or in combination, refers to an aryl group attached to the parent molecular moiety through an alkenyl group.
- arylalkoxy or “aralkoxy,” as used herein, alone or in combination, refers to an aryl group attached to the parent molecular moiety through an alkoxy group.
- arylalkyl or “aralkyl,” as used herein, alone or in combination, refers to an aryl group attached to the parent molecular moiety through an alkyl group.
- arylalkynyl or “aralkynyl,” as used herein, alone or in combination, refers to an aryl group attached to the parent molecular moiety through an alkynyl group.
- arylalkanoyl refers to an acyl substituent derived from an aryl-substituted alkanecarboxylic acid such as benzoyl, napthoyl, phenylacetyl, 3-phenylpropionyl (hydrocinnamoyl), 4-phenylbutyryl, (2-naphthyl)acetyl, 4-chlorohydrocinnamoyl, and the like.
- aryloxy refers to an aryl group attached to the parent molecular moiety through an oxy.
- benzo and “benz,” as used herein, alone or in combination, refer to the divalent substituent C 6 H 4 ⁇ derived from benzene. Examples include benzothiophene and benzimidazole.
- carbamate refers to an ester of carbamic acid (—NHC(O)O—) which may be attached to the parent molecular moiety from either the nitrogen or acid end, and which may be optionally substituted as defined herein.
- O-carbamyl as used herein, alone or in combination, refers to a —OC(O)NRR′, group-with R and R′ as defined herein.
- N-carbamyl as used herein, alone or in combination, refers to a ROC(O)NR′— group, with R and R′ as defined herein.
- carbonyl when alone includes formyl [—C(O)H] and in combination is a —C(O)— group.
- carboxyl or “carboxy,” as used herein, refers to —C(O)OH or the corresponding “carboxylate” anion, such as is in a carboxylic acid salt.
- An “O-carboxy” group refers to a RC(O)O— group, where R is as defined herein.
- a “C-carboxy” group refers to a —C(O)OR groups where R is as defined herein.
- cyano as used herein, alone or in combination, refers to —CN.
- cycloalkyl or, alternatively, “carbocycle,” as used herein, alone or in combination, refers to a saturated or partially saturated monocyclic, bicyclic or tricyclic alkyl group wherein each cyclic moiety contains from 3 to 12 carbon atom ring members and which may optionally be a benzo fused ring system which is optionally substituted as defined herein.
- the cycloalkyl will comprise from 5 to 7 carbon atoms.
- cycloalkyl groups examples include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, tetrahydronapthyl, indanyl, octahydronaphthyl, 2,3-dihydro-1H-indenyl, adamantyl and the like.
- “Bicyclic” and “tricyclic” as used herein are intended to include both fused ring systems, such as decahydronaphthalene, octahydronaphthalene as well as the multicyclic (multicentered) saturated or partially unsaturated type. The latter type of isomer is exemplified in general by, bicyclo[1,1,1]pentane, camphor, adamantane, and bicyclo[3,2,1]octane.
- esters refers to a carboxy group bridging two moieties linked at carbon atoms.
- ether refers to an oxy group bridging two moieties linked at carbon atoms.
- halo or halogen, as used herein, alone or in combination, refers to fluorine, chlorine, bromine, or iodine.
- haloalkoxy refers to a haloalkyl group attached to the parent molecular moiety through an oxygen atom.
- haloalkyl refers to an alkyl substituent having the meaning as defined above wherein one or more hydrogens are replaced with a halogen. Specifically embraced are monohaloalkyl, dihaloalkyl and polyhaloalkyl substituents.
- a monohaloalkyl substituent for one example, may have an iodo, bromo, chloro or fluoro atom within the substituent.
- Dihalo and polyhaloalkyl substituents may have two or more of the same halo atoms or a combination of different halo substituents.
- haloalkyl substituents include fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, pentafluoroethyl, heptafluoropropyl, difluorochloromethyl, dichlorofluoromethyl, difluoroethyl, difluoropropyl, dichloroethyl and dichloropropyl.
- Haloalkylene refers to a haloalkyl group attached at two or more positions. Examples include fluoromethylene (—CFH—), difluoromethylene (—CF 2 —), chloromethylene (—CHCl—) and the like.
- heteroalkyl refers to a stable straight or branched chain, or cyclic hydrocarbon substituent, or combinations thereof, fully saturated or containing from 1 to 3 degrees of unsaturation, consisting of the stated number of carbon atoms and from one to three heteroatoms selected from the group consisting of O, N, and S, and wherein the nitrogen and sulfur atoms may optionally be oxidized and the nitrogen heteroatom may optionally be quaternized.
- the heteroatom(s) 0, N and S may be placed at any interior position of the heteroalkyl group. Up to two heteroatoms may be consecutive, such as, for example, —CH 2 —NH—OCH 3 .
- heteroaryl refers to a 3 to 15 membered unsaturated heteromonocyclic ring, or a fused monocyclic, bicyclic, or tricyclic ring system in which at least one of the fused rings is aromatic, which contains at least one atom selected from the group consisting of O, S, and N.
- the heteroaryl will comprise from 5 to 7 carbon atoms.
- heteroaryl group may contain an oxo group such as the one present in a pyridone group.
- heteroaryl groups include pyrrolyl, pyrrolinyl, imidazolyl, pyrazolyl, pyridyl, pyrimidinyl, pyrazinyl, pyridazinyl, triazolyl, pyranyl, furyl, thienyl, oxazolyl, isoxazolyl, oxadiazolyl, thiazolyl, thiadiazolyl, isothiazolyl, indolyl, isoindolyl, indolizinyl, benzimidazolyl, quinolyl, isoquinolyl, quinoxalinyl, quinazolinyl, indazolyl, benzotriazolyl, benzodioxolyl, benzopyranyl, benzoxazolyl, benzoxadiazolyl, benzothiazolyl, benzothiadiazolyl, benzofuryl, benzothienyl, chromonyl,
- Exemplary tricyclic heterocyclic groups include carbazolyl, benzidolyl, phenanthrolinyl, dibenzofuranyl, acridinyl, phenanthridinyl, xanthenyl and the like.
- heterocycloalkyl and, interchangeably, “heterocycle,” as used herein, alone or in combination, each refer to a saturated, partially unsaturated, or fully unsaturated monocyclic, bicyclic, or tricyclic heterocyclic group containing at least one heteroatom as a ring member, wherein each the heteroatom may be independently selected from the group consisting of nitrogen, oxygen, and sulfur
- the hetercycloalkyl will comprise from 1 to 4 heteroatoms as ring members.
- the hetercycloalkyl will comprise from 1 to 2 heteroatoms as ring members.
- the hetercycloalkyl will comprise from 3 to 8 ring members in each ring.
- the hetercycloalkyl will comprise from 3 to 7 ring members in each ring. In yet further embodiments, the hetercycloalkyl will comprise from 5 to 6 ring members in each ring.
- “Heterocycloalkyl” and “heterocycle” are intended to include sulfones, sulfoxides, N-oxides of tertiary nitrogen ring members, and carbocyclic fused and benzo fused ring systems; additionally, both terms also include systems where a heterocycle ring is fused to an aryl group, as defined herein, or an additional heterocycle group.
- heterocycle groups include aziridinyl, azetidinyl, 1,3-benzodioxolyl, dihydroisoindolyl, dihydroisoquinolinyl, dihydrocinnolinyl, dihydrobenzodioxinyl, dihydro[1,3]oxazolo[4,5-b]pyridinyl, benzothiazolyl, dihydroindolyl, dihy-dropyridinyl, 1,3-dioxanyl, 1,4-dioxanyl, 1,3-dioxolanyl, isoindolinyl, morpholinyl, piperazinyl, pyrrolidinyl, tetrahydropyridinyl, piperidinyl, thiomorpholinyl, and the like.
- the heterocycle groups may be optionally substituted unless specifically prohibited.
- hydrazinyl as used herein, alone or in combination, refers to two amino groups joined by a single bond, i.e., —N—N—.
- hydroxyalkyl refers to a hydroxy group attached to the parent molecular moiety through an alkyl group.
- amino as used herein, alone or in combination, refers to ⁇ N—.
- aminohydroxy refers to ⁇ N(OH) and ⁇ N—O—.
- the phrase “in the main chain” refers to the longest contiguous or adjacent chain of carbon atoms starting at the point of attachment of a group to the compounds of any one of the formulas disclosed herein.
- isocyanato refers to a —NCO group.
- isothiocyanato refers to a —NCS group.
- linear chain of atoms refers to the longest straight chain of atoms independently selected from carbon, nitrogen, oxygen and sulfur.
- lower means containing from 1 to and including 6 carbon atoms.
- lower aryl as used herein, alone or in combination, means phenyl or naphthyl, either of which may be optionally substituted as provided.
- lower heteroaryl means either 1) monocyclic heteroaryl comprising five or six ring members, of which between one and four the members may be heteroatoms selected from the group consisting of O, S, and N, or 2) bicyclic heteroaryl, wherein each of the fused rings comprises five or six ring members, comprising between them one to four heteroatoms selected from the group consisting of O, S, and N.
- lower cycloalkyl means a monocyclic cycloalkyl having between three and six ring members. Lower cycloalkyls may be unsaturated. Examples of lower cycloalkyl include cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
- lower heterocycloalkyl means a monocyclic heterocycloalkyl having between three and six ring members, of which between one and four may be heteroatoms selected from the group consisting of O, S, and N.
- lower heterocycloalkyls include pyrrolidinyl, imidazolidinyl, pyrazolidinyl, piperidinyl, piperazinyl, and morpholinyl.
- Lower heterocycloalkyls may be unsaturated.
- lower amino refers to —NRR′, wherein R and R are independently chosen from hydrogen, lower alkyl, and lower heteroalkyl, any of which may be optionally substituted. Additionally, the R and R′ of a lower amino group may combine to form a five- or six-membered heterocycloalkyl, either of which may be optionally substituted.
- mercaptyl as used herein, alone or in combination, refers to an RS— group, where R is as defined herein.
- nitro refers to —NO 2 .
- oxy or “oxa,” as used herein, alone or in combination, refer to —O—.
- perhaloalkoxy refers to an alkoxy group where all of the hydrogen atoms are replaced by halogen atoms.
- perhaloalkyl refers to an alkyl group where all of the hydrogen atoms are replaced by halogen atoms.
- sulfonate refers the —SO 3 H group and its anion as the sulfonic acid is used in salt formation.
- sulfonyl as used herein, alone or in combination, refers to —S(O) 2 —.
- N-sulfonamido refers to a RS( ⁇ O) 2 NR′— group with R and R′ as defined herein.
- S-sulfonamido refers to a —S( ⁇ O) 2 NRR′, group, with R and R′ as defined herein.
- thia and thio refer to a —S— group or an ether wherein the oxygen is replaced with sulfur.
- the oxidized derivatives of the thio group namely sulfinyl and sulfonyl, are included in the definition of thia and thio.
- thiol as used herein, alone or in combination, refers to an —SH group.
- thiocarbonyl when alone includes thioformyl —C(S)H and in combination is a —C(S)— group.
- N-thiocarbamyl refers to an ROC(S)NR′— group, with R and R′ as defined herein.
- O-thiocarbamyl refers to a —OC(S)NRR′, group with R and R′ as defined herein.
- thiocyanato refers to a —CNS group.
- trihalomethanesulfonamido refers to a X 3 CS(O) 2 NR— group with X is a halogen and R as defined herein.
- trihalomethanesulfonyl refers to a X 3 CS(O) 2 — group where X is a halogen.
- trihalomethoxy refers to a X 3 CO— group where X is a halogen.
- trimethysilyl as used herein, alone or in combination, refers to a silicone group substituted at its three free valences with groups as listed herein under the definition of substituted amino. Examples include trimethysilyl, tert-butyldimethylsilyl, triphenylsilyl and the like.
- any definition herein may be used in combination with any other definition to describe a composite structural group.
- the trailing element of any such definition is that which attaches to the parent moiety.
- the composite group alkylamido would represent an alkyl group attached to the parent molecule through an amido group
- the term alkoxyalkyl would represent an alkoxy group attached to the parent molecule through an alkyl group.
- the term “optionally substituted” means the anteceding group may be substituted or unsubstituted.
- the substituents of an “optionally substituted” group may include, without limitation, one or more substituents independently selected from the following groups or a particular designated set of groups, alone or in combination: lower alkyl, lower alkenyl, lower alkynyl, lower alkanoyl, lower heteroalkyl, lower heterocycloalkyl, lower haloalkyl, lower haloalkenyl, lower haloalkynyl, lower perhaloalkyl, lower perhaloalkoxy, lower cycloalkyl, phenyl, aryl, aryloxy, lower alkoxy, lower haloalkoxy, oxo, lower acyloxy, carbonyl, carboxyl, lower alkylcarbonyl, lower carboxyester, lower carboxamido, cyano, hydrogen, halogen, hydroxy, amino, lower alkylamino
- Two substituents may be joined together to form a fused five-, six-, or seven-membered carbocyclic or heterocyclic ring consisting of zero to three heteroatoms, for example forming methylenedioxy or ethylenedioxy.
- An optionally substituted group may be unsubstituted (e.g., —CH 2 CH 3 ), fully substituted (e.g., —CF 2 CF 3 ), monosubstituted (e.g., —CH 2 CH 2 F) or substituted at a level anywhere in-between fully substituted and monosubstituted (e.g., —CH 2 CF 3 ).
- R or the term R′ refers to a moiety selected from the group consisting of hydrogen, alkyl, cycloalkyl, heteroalkyl, aryl, heteroaryl and heterocycloalkyl, any of which may be optionally substituted.
- aryl, heterocycle, R, etc. occur more than one time in a formula or generic structure, its definition at each occurrence is independent of the definition at every other occurrence.
- certain groups may be attached to a parent molecule or may occupy a position in a chain of elements from either end as written.
- an unsymmetrical group such as —C(O)N(R)— may be attached to the parent moiety at either the carbon or the nitrogen.
- bonds refers to a covalent linkage between two atoms, or two moieties when the atoms joined by the bond are considered part of larger substructure.
- a bond may be single, double, or triple unless otherwise specified.
- a dashed line between two atoms in a drawing of a molecule indicates that an additional bond may be present or absent at that position.
- disease as used herein is intended to be generally synonymous, and is used interchangeably with, the terms “disorder,” “syndrome,” and “condition” (as in medical condition), in that all reflect an abnormal condition of the human or animal body or of one of its parts that impairs normal functioning, is typically manifested by distinguishing signs and symptoms, and causes the human or animal to have a reduced duration or quality of life.
- combination therapy means the administration of two or more therapeutic agents to treat a therapeutic condition or disorder described in the present disclosure. Such administration encompasses co-administration of these therapeutic agents in a substantially simultaneous manner, such as in a single capsule having a fixed ratio of active ingredients or in multiple, separate capsules for each active ingredient. In addition, such administration also encompasses use of each type of therapeutic agent in a sequential manner. In either case, the treatment regimen will provide beneficial effects of the drug combination in treating the conditions or disorders described herein.
- ITK inhibitor is used herein to refer to a compound that exhibits an IC 50 with respect to ITK activity of no more than about 100 ⁇ M and more typically not more than about 50 ⁇ M, as measured in the ITK enzyme assay described generally herein below.
- IC 50 is that concentration of inhibitor that reduces the activity of an enzyme (e.g., ITK) to half-maximal level. Certain compounds disclosed herein have been discovered to exhibit inhibition against ITK.
- compounds will exhibit an IC 50 with respect to ITK of no more than about 10 ⁇ M; in further embodiments, compounds will exhibit an IC 50 with respect to ITK of no more than about 5 ⁇ M; in yet further embodiments, compounds will exhibit an IC 50 with respect to ITK of not more than about 1 ⁇ M; in yet further embodiments, compounds will exhibit an IC 50 with respect to ITK of not more than about 200 nM, as measured in the ITK binding assay described herein.
- terapéuticaally effective is intended to qualify the amount of active ingredients used in the treatment of a disease or disorder or on the effecting of a clinical endpoint.
- terapéuticaally acceptable refers to those compounds (or salts, prodrugs, tautomers, zwitterionic forms, etc.) which are suitable for use in contact with the tissues of patients without undue toxicity, irritation, and allergic response, are commensurate with a reasonable benefit/risk ratio, and are effective for their intended use.
- treatment of a patient is intended to include prophylaxis. Treatment may also be preemptive in nature, i.e., it may include prevention of disease. Prevention of a disease may involve complete protection from disease, for example as in the case of prevention of infection with a pathogen, or may involve prevention of disease progression. For example, prevention of a disease may not mean complete foreclosure of any effect related to the diseases at any level, but instead may mean prevention of the symptoms of a disease to a clinically significant or detectable level. Prevention of diseases may also mean prevention of progression of a disease to a later stage of the disease.
- patient is generally synonymous with the term “subject” and includes all mammals including humans. Examples of patients include humans, livestock such as cows, goats, sheep, pigs, and rabbits, and companion animals such as dogs, cats, rabbits, and horses. Preferably, the patient is a human.
- prodrug refers to a compound that is made more active in vivo.
- Certain compounds disclosed herein may also exist as prodrugs, as described in Hydrolysis in Drug and Prodrug Metabolism: Chemistry, Biochemistry, and Enzymology (Testa, Bernard and Mayer, Joachim M. Wiley-VHCA, Zurich, Switzerland 2003).
- Prodrugs of the compounds described herein are structurally modified forms of the compound that readily undergo chemical changes under physiological conditions to provide the compound.
- prodrugs can be converted to the compound by chemical or biochemical methods in an ex vivo environment. For example, prodrugs can be slowly converted to a compound when placed in a transdermal patch reservoir with a suitable enzyme or chemical reagent.
- Prodrugs are often useful because, in some situations, they may be easier to administer than the compound, or parent drug. They may, for instance, be bioavailable by oral administration whereas the parent drug is not. The prodrug may also have improved solubility in pharmaceutical compositions over the parent drug.
- a wide variety of prodrug derivatives are known in the art, such as those that rely on hydrolytic cleavage or oxidative activation of the prodrug.
- An example, without limitation, of a prodrug would be a compound which is administered as an ester (the “prodrug”), but then is metabolically hydrolyzed to the carboxylic acid, the active entity. Additional examples include peptidyl derivatives of a compound.
- the compounds disclosed herein can exist as therapeutically acceptable salts.
- the present disclosure includes compounds listed above in the form of salts, including acid addition salts. Suitable salts include those formed with both organic and inorganic acids. Such acid addition salts will normally be pharmaceutically acceptable. However, salts of non-pharmaceutically acceptable salts may be of utility in the preparation and purification of the compound in question. Basic addition salts may also be formed and be pharmaceutically acceptable.
- Pharmaceutical Salts Properties, Selection, and Use (Stahl, P. Heinrich. Wiley-VCHA, Zurich, Switzerland, 2002).
- terapéuticaally acceptable salt represents salts or zwitterionic forms of the compounds disclosed herein which are water or oil-soluble or dispersible and therapeutically acceptable as defined herein.
- the salts can be prepared during the final isolation and purification of the compounds or separately by reacting the appropriate compound in the form of the free base with a suitable acid.
- Representative acid addition salts include acetate, adipate, alginate, L-ascorbate, aspartate, benzoate, benzenesulfonate (besylate), bisulfate, butyrate, camphorate, camphorsulfonate, citrate, digluconate, formate, fumarate, gentisate, glutarate, glycerophosphate, glycolate, hemisulfate, heptanoate, hexanoate, hippurate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxyethansulfonate (isethionate), lactate, maleate, malonate, DL-mandelate, mesitylenesulfonate, methanesulfonate, naphthylenesulfonate, nicotinate, 2-naphthalenesulfonate, oxalate, pamoate, pectinate, persulfate, 3-phenyl
- basic groups in the compounds disclosed herein can be quaternized with methyl, ethyl, propyl, and butyl chlorides, bromides, and iodides; dimethyl, diethyl, dibutyl, and diamyl sulfates; decyl, lauryl, myristyl, and steryl chlorides, bromides, and iodides; and benzyl and phenethyl bromides.
- acids which can be employed to form therapeutically acceptable addition salts include inorganic acids such as hydrochloric, hydrobromic, sulfuric, and phosphoric, and organic acids such as oxalic, maleic, succinic, and citric. Salts can also be formed by coordination of the compounds with an alkali metal or alkaline earth ion.
- the present disclosure contemplates sodium, potassium, magnesium, and calcium salts of the compounds disclosed herein, and the like.
- Basic addition salts can be prepared during the final isolation and purification of the compounds by reacting a carboxy group with a suitable base such as the hydroxide, carbonate, or bicarbonate of a metal cation or with ammonia or an organic primary, secondary, or tertiary amine.
- a suitable base such as the hydroxide, carbonate, or bicarbonate of a metal cation or with ammonia or an organic primary, secondary, or tertiary amine.
- the cations of therapeutically acceptable salts include lithium, sodium, potassium, calcium, magnesium, and aluminum, as well as nontoxic quaternary amine cations such as ammonium, tetramethylammonium, tetraethylammonium, methylamine, dimethylamine, trimethylamine, triethylamine, diethylamine, ethylamine, tributylamine, pyridine, N,N-dimethylaniline, N-methylpiperidine, N-methylmorpholine, dicyclohexylamine, procaine, dibenzylamine, N,N-dibenzylphenethylamine, 1-ephenamine, and N,N′-dibenzylethylenediamine.
- Other representative organic amines useful for the formation of base addition salts include ethylenediamine, ethanolamine, diethanolamine, piperidine, and piperazine.
- a salt of a compound can be made by reacting the appropriate compound in the form of the free base with the appropriate acid.
- compositions which comprise one or more of certain compounds disclosed herein, or one or more pharmaceutically acceptable salts, esters, prodrugs, amides, or solvates thereof, together with one or more pharmaceutically acceptable carriers thereof and optionally one or more other therapeutic ingredients.
- the carrier(s) must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not deleterious to the recipient thereof. Proper formulation is dependent upon the route of administration chosen. Any of the well-known techniques, carriers, and excipients may be used as suitable and as understood in the art; e.g., in Remington's Pharmaceutical Sciences.
- compositions disclosed herein may be manufactured in any manner known in the art, e.g., by means of conventional mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping or compression processes.
- the formulations include those suitable for oral, parenteral (including subcutaneous, intradermal, intramuscular, intravenous, intraarticular, and intramedullary), intraperitoneal, transmucosal, transdermal, rectal and topical (including dermal, buccal, sublingual and intraocular) administration although the most suitable route may depend upon for example the Condition and disorder of the recipient.
- the formulations may conveniently be presented in unit dosage form and may be prepared by any of the methods well known in the art of pharmacy. Typically, these methods include the step of bringing into association a compound of the subject disclosure or a pharmaceutically acceptable salt, ester, amide, prodrug or solvate thereof (“active ingredient”) with the carrier which constitutes one or more accessory ingredients.
- active ingredient a pharmaceutically acceptable salt, ester, amide, prodrug or solvate thereof
- the formulations are prepared by uniformly and intimately bringing into association the active ingredient with liquid carriers or finely divided solid carriers or both and then, if necessary, shaping the product into the desired formulation.
- Formulations of the compounds disclosed herein suitable for oral administration may be presented as discrete units such as capsules, cachets or tablets each Containing a predetermined amount of the active ingredient; as a powder or granules; as a solution or a suspension in an aqueous liquid or a non-aqueous liquid; or as an oil-in-water liquid emulsion or a water-in-oil liquid emulsion.
- the active ingredient may also be presented as a bolus, electuary or paste.
- compositions that can be used orally include tablets, push-fit capsules made of gelatin, as well as soft, sealed capsules made of gelatin and a plasticizer, such as glycerol or sorbitol. Tablets may be made by compression or molding, optionally with one or more accessory ingredients. Compressed tablets may be prepared by compressing in a suitable machine the active ingredient in a free-flowing form such as a powder or granules, optionally mixed with binders, inert diluents, or lubricating, surface active or dispersing agents. Molded tablets may be made by molding in a suitable machine a mixture of the powdered compound moistened with an inert liquid diluent.
- the tablets may optionally be coated or scored and may be formulated to provide slow or Controlled release of the active ingredient therein. All formulations for oral administration should be in dosages suitable for such administration.
- the push-fit capsules can contain the active ingredients in admixture with filler such as lactose, binders such as starches, and/or lubricants such as talc or magnesium stearate and, optionally, stabilizers.
- the active compounds may be dissolved or suspended in suitable liquids, such as fatty oils, liquid paraffin, or liquid polyethylene glycols.
- stabilizers may be added.
- Dragee cores are provided with suitable coatings.
- concentrated sugar solutions may be used, which may optionally Contain gum arabic, talc, polyvinyl pyrrolidone, carbopol gel, polyethylene glycol, and/or titanium dioxide, lacquer solutions, and suitable organic solvents or solvent mixtures.
- Dyestuffs or pigments may be added to the tablets or dragee coatings for identification or to characterize different combinations of active compound doses.
- the compounds may be formulated for parenteral administration by injection, e.g., by bolus injection or Continuous infusion.
- Formulations for injection may be presented in unit dosage form, e.g., in ampoules or in multi-dose Containers, with an added preservative.
- the compositions may take such forms as suspensions, solutions or emulsions in oily or aqueous vehicles, and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents.
- the formulations may be presented in unit-dose or multi-dose Containers, for example sealed ampoules and vials, and may be stored in powder form or in a freeze-dried (lyophilized) Condition requiring only the addition of the sterile liquid carrier, for example, saline or sterile pyrogen-free water, immediately prior to use.
- sterile liquid carrier for example, saline or sterile pyrogen-free water
- Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets of the kind previously described.
- Formulations for parenteral administration include aqueous and non-aqueous (oily) sterile injection solutions of the active compounds which may Contain antioxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents.
- Suitable lipophilic solvents or vehicles include fatty oils such as sesame oil, or synthetic fatty acid esters, such as ethyl oleate or triglycerides, or liposomes.
- Aqueous injection suspensions may contain substances that increase the viscosity of the suspension, such as sodium carboxymethyl cellulose, sorbitol, or dextran.
- the suspension may also contain suitable stabilizers or agents that increase the solubility of the compounds to allow for the preparation of highly concentrated solutions.
- the compounds may also be formulated as a depot preparation. Such long acting formulations may be administered by implantation (for example subcutaneously or intramuscularly) or by intramuscular injection.
- the compounds may be formulated with suitable polymeric or hydrophobic materials (for example as an emulsion in an acceptable oil) or ion exchange resins, or as sparingly soluble derivatives, for example, as a sparingly soluble salt.
- compositions may take the form of tablets, lozenges, pastilles, or gels formulated in conventional manner.
- Such compositions may comprise the active ingredient in a flavored basis such as sucrose and acacia or tragacanth.
- the compounds may also be formulated in rectal compositions such as suppositories or retention enemas, e.g., containing conventional suppository bases such as cocoa butter, polyethylene glycol, or other glycerides.
- Certain compounds disclosed herein may be administered topically, that is by non-systemic administration. This includes the application of a compound disclosed herein externally to the epidermis or the buccal cavity and the instillation of such a compound into the ear, eye and nose, such that the compound does not significantly enter the blood stream.
- systemic administration refers to oral, intravenous, intraperitoneal and intramuscular administration.
- Formulations suitable for topical administration include liquid or semi-liquid preparations suitable for penetration through the skin to the site of inflammation such as gels, liniments, lotions, creams, ointments or pastes, and drops suitable for administration to the eye, ear or nose.
- the active ingredient for topical administration may comprise, for example, from 0.001% to 10% w/w (by weight) of the formulation. In certain embodiments, the active ingredient may comprise as much as 10% w/w. In other embodiments, it may comprise less than 5% w/w. In certain embodiments, the active ingredient may comprise from 2% w/w to 5% w/w. In other embodiments, it may comprise from 0.1% to 1% w/w of the formulation.
- Gels for topical or transdermal administration may comprise, generally, a mixture of volatile solvents, nonvolatile solvents, and water.
- the volatile solvent component of the buffered solvent system may include lower (C1-C6) alkyl alcohols, lower alkyl glycols and lower glycol polymers.
- the volatile solvent is ethanol.
- the volatile solvent component is thought to act as a penetration enhancer, while also producing a cooling effect on the skin as it evaporates.
- the nonvolatile solvent portion of the buffered solvent system is selected from lower alkylene glycols and lower glycol polymers. In certain embodiments, propylene glycol is used.
- the nonvolatile solvent slows the evaporation of the volatile solvent and reduces the vapor pressure of the buffered solvent system.
- the amount of this nonvolatile solvent component, as with the volatile solvent, is determined by the pharmaceutical compound or drug being used. When too little of the nonvolatile solvent is in the system, the pharmaceutical compound may crystallize due to evaporation of volatile solvent, while an excess may result in a lack of bioavailability due to poor release of drug from solvent mixture.
- the buffer component of the buffered solvent system may be selected from any buffer commonly used in the art; in certain embodiments, water is used. A common ratio of ingredients is about 20% of the nonvolatile solvent, about 40% of the volatile solvent, and about 40% water.
- chelators and gelling agents Appropriate gelling agents can include, but are not limited to, semisynthetic cellulose derivatives (such as hydroxypropylmethylcellulose) and synthetic polymers, and cosmetic agents.
- Lotions include those suitable for application to the skin or eye.
- An eye lotion may comprise a sterile aqueous solution optionally containing a bactericide and may be prepared by methods similar to those for the preparation of drops.
- Lotions or liniments for application to the skin may also include an agent to hasten drying and to cool the skin, such as an alcohol or acetone, and/or a moisturizer such as glycerol or an oil such as castor oil or arachis oil.
- Creams, ointments or pastes are semi-solid formulations of the active ingredient for external application. They may be made by mixing the active ingredient in finely-divided or powdered form, alone or in solution or suspension in an aqueous or non-aqueous fluid, with the aid of suitable machinery, with a greasy or non-greasy base.
- the base may comprise hydrocarbons such as hard, soft or liquid paraffin, glycerol, beeswax, a metallic soap; a mucilage; an oil of natural origin such as almond, corn, arachis, castor or olive oil; wool fat or its derivatives or a fatty acid such as steric or oleic acid together with an alcohol such as propylene glycol or a macrogel.
- the formulation may incorporate any suitable surface-active agent such as an anionic, cationic or non-ionic surfactant such as a sorbitan ester or a polyoxyethylene derivative thereof.
- suitable surface-active agent such as an anionic, cationic or non-ionic surfactant such as a sorbitan ester or a polyoxyethylene derivative thereof.
- Suspending agents such as natural gums, cellulose derivatives or inorganic materials such as silicaceous silicas, and other ingredients such as lanolin, may also be included.
- Drops may comprise sterile aqueous or oily solutions or suspensions and may be prepared by dissolving the active ingredient in a suitable aqueous solution of a bactericidal and/or fungicidal agent and/or any other suitable preservative, and, in certain embodiments, including a surface-active agent.
- the resulting solution may then be clarified by filtration, transferred to a suitable Container which is then sealed and sterilized by autoclaving or maintaining at 98-100° C. for half an hour.
- the solution may be sterilized by filtration and transferred to the Container by an aseptic technique.
- bactericidal and fungicidal agents suitable for inclusion in the drops are phenylmercuric nitrate or acetate (0.002%), benzalkonium chloride (0.01%) and chlorhexidine acetate (0.01%).
- Suitable solvents for the preparation of an oily solution include glycerol, diluted alcohol and propylene glycol.
- Formulations for topical administration in the mouth include lozenges comprising the active ingredient in a flavored basis such as sucrose and acacia or tragacanth, and pastilles comprising the active ingredient in a basis such as gelatin and glycerin or sucrose and acacia.
- compounds may be conveniently delivered from an insufflator, nebulizer pressurized packs or other convenient means of delivering an aerosol spray.
- Pressurized packs may comprise a suitable propellant such as dichlorodifluoromethane, trichlorofluoromethane, dichlorotetrafluoroethane, carbon dioxide or other suitable gas.
- the dosage unit may be determined by providing a valve to deliver a metered amount.
- the compounds according to the disclosure may take the form of a dry powder composition, for example, a powder mix of the compound and a suitable powder base such as lactose or starch.
- the powder composition may be presented in unit dosage form, in for example, capsules, cartridges, gelatin or blister packs from which the powder may be administered with the aid of an inhalator or insufflator.
- Preferred unit dosage formulations are those Containing an effective dose, as herein below recited, or an appropriate fraction thereof, of the active ingredient.
- formulations described above may include other agents conventional in the art having regard to the type of formulation in question, for example, those suitable for oral administration may include flavoring agents.
- Compounds may be administered orally or via injection at a dose of from 0.1 to 500 mg/kg per day.
- the dose range for adult humans is generally from 5 mg to 2 g/day.
- Tablets or other forms of presentation provided in discrete units may conveniently contain an amount of one or more compounds which is effective at such dosage or as a multiple of the same, for instance, units Containing 5 mg to 500 mg, usually around 10 mg to 200 mg.
- the amount of active ingredient that may be combined with the carrier materials to produce a single dosage form will vary depending upon the host treated and the particular mode of administration.
- the compounds can be administered in various modes, e.g. orally, topically, or by injection.
- the precise amount of compound administered to a patient will be the responsibility of the attendant physician.
- the specific dose level for any particular patient will depend upon a variety of factors including the activity of the specific compound employed, the age, body weight, general health, sex, diets, time of administration, route of administration, rate of excretion, drug combination, the precise disorder being treated, and the severity of the indication or Condition being treated.
- the route of administration may vary depending on the Condition and its severity.
- the compounds described herein may be administered in combination with another therapeutic agent.
- another therapeutic agent such as a pharmaceutically acceptable salt, ester, or prodrug thereof.
- the therapeutic effectiveness of one of the compounds described herein may be enhanced by administration of an adjuvant (i.e., by itself the adjuvant may only have minimal therapeutic benefit, but in combination with another therapeutic agent, the overall therapeutic benefit to the patient is enhanced).
- the benefit of experienced by a patient may be increased by administering one of the compounds described herein with another therapeutic agent (which also includes a therapeutic regimen) that also has therapeutic benefit.
- another therapeutic agent which also includes a therapeutic regimen
- increased therapeutic benefit may result by also providing the patient with another therapeutic agent for diabetes.
- the overall benefit experienced by the patient may simply be additive of the two therapeutic agents or the patient may experience a synergistic benefit.
- NSAIDs nonsteroidal anti-inflammatory drugs
- corticosteroids including but not limited to cortisone, dexamethasone, and methylprednisolone
- NSAIDs nonsteroidal anti-inflammatory drugs
- ibuprofen including but not limited to ibuprofen, naproxen, acetaminophen, aspirin, fenoprofen (NALFON), flurbiprofen (ANSAID), ketoprofen, oxaprozin (DAYPRO), diclofenac sodium (VOLTAREN), diclofenac potassium (CATAFLAM), etodolac (LODINE), indomethacin (INDOCIN), ketorolac (TORADOL), sulindac (CLINORIL), tolmetin (TOLECTIN), meclofenamate (MECLOMEN), mefenamic acid (PONSTEL), nabumetone (RELAFEN) and piroxicam (FELDENE
- alkylating agents including but not limited to cisplatin (PLATIN), carboplatin (PARAPLATIN), oxaliplatin (ELOXATIN), streptozocin (ZANOSAR), busulfan (MYLERAN) and cyclophosphamide (ENDOXAN);
- anti-metabolites including but not limited to mercaptopurine (PURINETHOL), thioguanine, pentostatin (NIPENT), cytosine arabinoside (ARA-C), gemcitabine (GEMZAR), fluorouracil (CARAC), leucovorin (FUSILEV) and methotrexate (RHEUMATREX);
- plant alkaloids and terpenoids including but not limited to vincristine (ONCOVIN), vinblastine and paclitaxel (TAXOL);
- topoisomerase inhibitors including but not limited
- the multiple therapeutic agents may be administered in any order or even simultaneously. If simultaneously, the multiple therapeutic agents may be provided in a single, unified form, or in multiple forms (by way of example only, either as a single pill or as two separate pills). One of the therapeutic agents may be given in multiple doses, or both may be given as multiple doses. If not simultaneous, the timing between the multiple doses may be any duration of time ranging from a few minutes to four weeks.
- certain embodiments provide methods for treating ITK-mediated disorders in a human or animal subject in need of such treatment comprising administering to the subject an amount of a compound disclosed herein effective to reduce or prevent the disorder in the subject, in combination with at least one additional agent for the treatment of the disorder that is known in the art.
- certain embodiments provide therapeutic compositions comprising at least one compound disclosed herein in combination with one or more additional agents for the treatment of ITK mediated disorders.
- Specific diseases to be treated by the compounds, compositions, and methods disclosed herein include autoimmune disorders, chronic inflammatory disorders, auto-inflammatory disorders, pain, inflammatory disorders, allergic disorders, autoimmune disorders and the like.
- the methods described herein are used to treat a patient in need thereof suffering from inflammatory disorders, allergic disorders, and autoimmune disorders.
- disorders include, but are not limited to asthma, rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Contact hypersensitivity and inflammatory bowel disease.
- Diseases to be treated by the compounds, compositions, and methods disclosed herein related to cancer include cancer specific to T-cells such as T-cell lymphoma and lymphblastic T-cell leukemia.
- HIV infections to be treated by the compounds, compositions, and methods disclosed herein include HIV.
- certain compounds and formulations disclosed herein may also be useful for veterinary treatment of companion animals, exotic animals and farm animals, including mammals, rodents, and the like. More preferred animals include horses, dogs, and cats.
- the present disclosure provides a compound, or a pharmaceutically acceptable salt, hydrate or solvate thereof, of Formula (I):
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , C(O)(C(O)NH 2 )C ⁇ CH 2 , S(O) 2 CH ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)C(CN) ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(O)NH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)NHR 7 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)N(R 7
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is hydrogen;
- R 3 is hydrogen;
- R 4 is chosen from, C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)C(CN) ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(O)NH 2 , and (CH 2 ) m CR 7 ⁇ CR 9 C(O)NHR 7 ;
- R 5 is chosen from hydrogen, —(CH 2 ) n C 3-7 cycloalkyl, and C 1-4 alkyl;
- each R 6 is independently chosen from hydrogen, C 1-4 alkyl, OC 1-4 alkyl, and NHC 1-4 alkyl;
- the compound is composed of formula (II):
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , C(O)(C(O)NH 2 )C ⁇ CH 2 , S(O) 2 CH ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)C(CN) ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(O)NH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)NHR 7 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)N(R 7
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is hydrogen;
- R 3 is hydrogen;
- R 4 is chosen from, C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)C(CN) ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(O)NH 2 , and (CH 2 ) m CR 7 ⁇ CR 9 C(O)NHR 7 ;
- R 5 is chosen from hydrogen, —(CH 2 ) n C 3-7 cycloalkyl, and C 1-4 alkyl;
- each R 6 is independently chosen from hydrogen, C 1-4 alkyl, OC 1-4 alkyl, and NHC 1-4 alkyl;
- the compound is composed of formula (III):
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , C(O)(C(O)NH 2 )C ⁇ CH 2 , S(O) 2 CH ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 3 is chosen from hydrogen, halo and C 1-4 alkyl;
- R 4 is chosen from cyano, C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)C(CN) ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(O)NH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)NHR 7 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)N(R 7
- Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is hydrogen;
- R 3 is hydrogen;
- R 4 is chosen from, C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)C(CN) ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(O)NH 2 , and (CH 2 ) m CR 7 ⁇ CR 9 C(O)NHR 7 ;
- R 5 is chosen from hydrogen, —(CH 2 ) n C 3-7 cycloalkyl, and C 1-4 alkyl;
- each R 6 is independently chosen from hydrogen, C 1-4 alkyl, OC 1-4 alkyl, and NHC 1-4 alkyl;
- the compound is selected from the group consisting of: 5-((1-(2-chloroacetyl)azetidin-3-yl)amino)-[3,4′-bipyridin]-6(1H)-one; 5-((1-acryloylazetidin-3-yl)amino)-[3,4′-bipyridin]-6(1H)-one; (E)-5-((1-(but-2-enoyl)azetidin-3-yl)amino)-[3,4′-bipyridin]-6(1H)-one; 5-((1-acryloylazetidin-3-yl)amino)-2′-methyl-[3,4′-bipyridin]-6(1H)-one; 3-((1-acryloylazetidin-3-yl)amino)-5-(6-methoxypyrazin-2-yl)pyridin-2(1H)-one; 5-
- Ar is phenyl, 3-methoxyphenyl, 3-aminomethylphenyl, 4-pyridine, 3-methyl-4-pyridyl, 3-aminomethyl-4-pyridinyl, 3-aminomethyl-2,4-pyrimidinyl, 3-methoxy-2,5-pyrazinyl, 3-methylamino-2,5-pyrazinyl, wherein Ar may be optionally substituted with one or more R 6 substituents;
- R 2 is independently chosen from hydrogen, halo or C 1-4 alkyl;
- R 4 is independently chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHCOR 8 , C(O)CH ⁇
- Ar is 4-pyridine, 3-methyl-4-pyridyl, 3-aminomethyl-4-pyridinyl, 3-aminomethyl-2,4-pyrimidinyl, 3-methoxy-2,5-pyrazinyl, 3-methylamino-2,5-pyrazinyl;
- R 2 is independently chosen from hydrogen, halo or C 1-4 alkyl;
- R 4 is independently chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHCOR 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , CO(C(O)NH 2 )C ⁇ CH 2 , S
- Ar is 4-pyridine, 3-methyl-4-pyridyl, 3-aminomethyl-4-pyridinyl, 3-aminomethyl-2,4-pyrimidinyl, 3-methoxy-2,5-pyrazinyl, 3-methylamino-2,5-pyrazinyl;
- R 2 is hydrogen;
- R 4 is independently chosen from cyano, C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHCOR 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , CO(C(O)NH 2 )C ⁇ CH 2 , S(O) 2 CH ⁇ CH 2 , (CH 2
- Ar is 4-pyridine, 3-methyl-4-pyridyl, 3-aminomethyl-4-pyridinyl, 3-aminomethyl-2,4-pyrimidinyl, 3-methoxy-2,5-pyrazinyl, 3-methylamino-2,5-pyrazinyl;
- R 2 is hydrogen;
- R 4 is independently chosen from C(O)CH 2 R 2 , C(O)CF 3 , C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHCOR 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , CO(C(O)NH 2 )C ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)M
- Ar is 4-pyridine, 3-methyl-4-pyridyl, 3-aminomethyl-4-pyridinyl, 3-aminomethyl-2,4-pyrimidinyl, 3-methoxy-2,5-pyrazinyl, 3-methylamino-2,5-pyrazinyl;
- R 2 is hydrogen;
- R 4 is independently chosen from C(O)CH ⁇ CH 2 , C(O)CR 7 ⁇ CH 2 , C(O)CH ⁇ CHR 7 , C(O)CR 7 ⁇ CHR 7 , C(O)CH ⁇ CR 7 R 7 , C(O)CH ⁇ CHCH 2 R 8 , C(O)CH ⁇ CHCOR 8 , C(O)CH ⁇ CHC(O)CH 2 R 8 , C(O)C(CN) ⁇ CH 2 , CO(C(O)NH 2 )C ⁇ CH 2 , (CH 2 ) m CR 7 ⁇ CR 9 C(O)Me, (CH 2 ) m CR 7 ⁇ CR 9 C(
- the present disclosure provides compounds and pharmaceutical compositions that inhibit kinase activity, particularly ITK activity and are thus useful in the treatment or prevention of disorders associated with ITK.
- Compounds and pharmaceutical compositions of the present disclosure selectively inhibit ITK and are thus useful in the treatment or prevention of a range of disorders associated with the activation of ITK which includes, but are not limited to respiratory diseases, allergic diseases, autoimmune diseases, inflammatory disorders, immunological disorders, proliferative disorders, transplant rejection, graft versus host disease, HIV, aplastic anemia, pain including inflammatory pain and other diseases and disorders associated with ITK.
- the compounds of the present disclosure may be used to prevent or treat asthma, rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Contact hypersensitivity and inflammatory bowel disease.
- the compounds of the present disclosure may be used to prevent or treat T-cell lymphoma and lymphblastic T-cell leukemia.
- the compounds of the present disclosure may be used to prevent or treat HIV.
- Compounds and pharmaceutically acceptable compositions of the present disclosure can be employed in combination therapies, that is, the compounds and pharmaceutically acceptable compositions may have potential utility in combination with other therapies for the treatment of immune, inflammatory, proliferative, and allergic disorders.
- Example includes but not limited to co-administration with steroids, leukotriene antagonists, anti-histamines, anti-cancer agents, protein kinase inhibitors, cyclosporine, or rapamycin.
- the compounds of the present disclosure may be used to prevent or treat an ITK-mediated disorder by the sequential or co-administration of another therapeutic agent.
- the therapeutic agent is selected from taxanes, inhibitors of bcr-abl, inhibitors of EGFR, DNA damaging agents, and antimetabolites.
- the therapeutic agent is selected from Paclitaxel, Gleevec, dasatinib, nilotinib, Tarceva, Iressa, cisplatin, oxaliplatin, carboplatin, anthracyclines, AraC and 5-FU.
- the therapeutic agent is selected from camptothecin, doxorubicin, idarubicin, Cisplatin, taxol, taxotere, vincristine, tarceva, a MEK inhibitor, U0126, a KSP inhibitor, vorinostat, Gleevec, dasatinib, and nilotinib.
- Compounds and pharmaceutical compositions of the present disclosure selectively inhibit ITK and are thus useful in the treatment or prevention of a range of disorders associated with the activation of ITK which includes, but are not limited to chronic obstructive pulmonary disease (COPD) such as irreversible COPD; asthma, such as bronchial, allergic, intrinsic, extrinsic and dust asthma, particularly chronic or inveterate asthma (for example, late asthma and airways hyper-responsiveness); bronchitis; acute, allergic, atrophic rhinitis and chronic rhinitis including rhinitis caseosa, hypertrophic rhinitis, rhinitis purulenta, rhinitis sicca and rhinitis medicamentosa; membranous rhinitis including croupous, fibrinous and pseudomembranous rhinitis and scrofoulous rhinitis; seasonal rhinitis including rhinitis nervosa (hay fever) and vasomotor rhin
- the compounds of the present disclosure are useful for the treatment of cancer such as, but are not limited to breast cancer, skin cancer, bone cancer, prostate cancer, liver cancer, lung cancer, non-small cell lung cancer, brain cancer, cancer of the larynx, gall bladder, pancreas, rectum, parathyroid, thyroid, adrenal, neural tissue, head and neck, colon, stomach, bronchi, and kidney cancer, basal cell carcinoma, squamous cell carcinoma, metastatic skin carcinoma, osteo sarcoma, Ewing's sarcoma, myeloma, giant cell tumor, small-cell lung tumor, islet cell tumor, primary brain tumor, lymphocytic and granulocytic tumors, hairy-cell tumor, adenoma, hyperplasia, medullary carcinoma, pheochromocytoma, mucosal neuromas, intestinal ganglioneuromas, ovarian tumor, cervical dysplasia, neuroblastoma, retinoblastoma, soft tissue sarcoma,
- the desired pyridinone analogs may be prepared as outlined in Scheme 1. Reaction of 1a with methyl iodide using silver carbonate as a base in benzene provides methoxypyridine 1b. Bromination of 1b using bromine in a sodium acetate/acetic mixture furnishes 1c. Following standard coupling Conditions 1c is reacted with the desired amine using a palladium catalyst such as Pd 2 dba 3 with X-phos as the ligand and cesium carbonate as a weak base in dioxane to provide 1d.
- a palladium catalyst such as Pd 2 dba 3 with X-phos as the ligand and cesium carbonate as a weak base in dioxane to provide 1d.
- a number of boronic acids may be used in the preparation of the desired analogs as shown in Scheme 3. Utilizing standard Suzuki coupling Conditions 1d is reacted with the desired boronic acid using a palladium catalyst such as Pd(dppf)Cl 2 with a weak base such as K 3 PO 4 in dioxane that provides 3a. The removal of the protecting group and formation of the desired pyridinone takes place by treating 3a with ethanolic HCl to give 3b. This compound is treated with the desired halide in the presence of triethylamine in dichloromethane to give 3c where R 4 contains a suitable electrophile.
- a palladium catalyst such as Pd(dppf)Cl 2
- K 3 PO 4 in dioxane
- Suzuki-based coupling of 5d and 5g was carried out in the presence a palladium catalyst such as Pd(PPh3)4 and a base such as potassium carbonate to give bis-aryl 5h.
- Deprotection of the methoxy group and concomitant formation of the pyridinone was carried out by treating 5h with trimethylsilyl chloride in the presence of sodium iodide to give pyridinone 5i.
- Hydrogenation of 5i using Pearlmans catalyst in methanol gives deprotected 5j.
- Condensation of amino 5j with the desired heterocyclic ketone using sodium cyanoborohydride and zinc chloride as a catalyst provides the desired carbamate analog, which was deprotected using hydrochloric acid in ethanol to give 5k.
- the amide coupling was accomplished by treating amine 5k with the desired carboxylic acid under HATU or related amide coupling conditions to provide 5l.
- Step 4 (R)-tert-Butyl 3-((6-methoxy-[3,4′-bipyridin]-5-yl)amino)piperidine-1-carboxylate
- Step 6 (R)-5-((1-(2-Chloroacetyl)piperidin-3-yl)amino)-[3,4′-bipyridin]-6(1H)-one
- Example 1 Step 3 tert-butyl 3-aminoazetidine-1-carboxylate (Example 1 Step 3) and reaction with chloroacetyl chloride (Example 1 Step 6) provided the desired compound after purification:
- Example 1 Step 3 tert-butyl 3-aminoazetidine-1-carboxylate (Example 1 Step 3) and reaction with acroyl chloride (Example 1 Step 6) provided the desired compound after purification:
- Step 1 tert-Butyl 3-((5-chloro-2-methoxypyridin-3-yl)amino)azetidine-1-carboxylate
- reaction mixture was filtered, washed with ethyl acetate (100 mL), the organic layer washed with water (60 mL), brine (60 mL), dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
- Step 2 tert-Butyl 3-((2′-fluoro-6-methoxy-[3,4′-bipyridin]-5-yl)amino)azetidine-1-carboxylate
- reaction mixture was filtered and washed with ethyl acetate (100 mL). The organic layer was washed with water (60 mL), brine (60 mL), dried over Na 2 SO 4 , filtered, and concentrated in vacuo.
- Step 3 tert-Butyl 3-((6-methoxy-2′-(methylamino)-[3,4′-bipyridin]-5-yl)amino)azetidine-1-carboxylate
- Step 4 5-(Azetidin-3-ylamino)-2′-(methylamino)-[3,4′-bipyridin]-6(1H)-one di-hydrobromide
- Step 5 (E)-5-((1-Cinnamoylazetidin-3-yl)amino)-2′-(methylamino)-[3,4′-bipyridin]-6(1H)-one
- Step 2 Benzyl (2-hydroxy-5-iodopyridin-3-yl)carbamate
- Step 3 Benzyl (5-iodo-2-methoxypyridin-3-yl)carbamate
- Step 4 Benzyl (2-methoxy-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin-3-yl)carbamate
- Step 6 Benzyl (2′-((4-isopropyl-3-methylphenyl)carbamoyl)-6-methoxy-[3,4′-bipyridin]-5-yl)carbamate
- Step 7 Benzyl (2′-((4-isopropyl-3-methylphenyl)carbamoyl)-6-oxo-1,6-dihydro-[3,4′-bipyridin]-5-yl)carbamate
- Step 8 5-Amino-N-(4-isopropyl-3-methylphenyl)-6-oxo-1,6-dihydro-[3,4′-bipyridine]-2′-carboxamide
- Step 9 5-(Azetidin-3-ylamino)-N-(4-isopropyl-3-methylphenyl)-6-oxo-1,6-dihydro-[3,4′-bipyridine]-2′-carboxamide hydrochloride
- Step 10 5-((1-Acryloylazetidin-3-yl)amino)-N-(4-isopropyl-3-methylphenyl)-6-oxo-1,6-dihydro-[3,4′-bipyridine]-2′-carboxamide hydrochloride
- SMILES Simplified Molecular Input Line Entry System
- SMILES is a modern chemical notation system, developed by David Weininger and Daylight Chemical Information Systems, Inc., that is built into all major commercial chemical structure drawing software packages. Software is not needed to interpret SMILES text strings, and an explanation of how to translate SMILES into structures can be found in Weininger, D., J. Chem. Inf Comput. Sci. 1988, 28, 31-36. All SMILES strings used herein, as well as many IUPAC names, were generated using CambridgeSoft's ChemDraw 10.0.
- the ability of candidate compounds to interact with ITK is quantitated by a competitive binding assay using the LanthaScreen technology developed by Life Technologies. This assay is based on the binding of a proprietary, Alexa Fluor® 647-labeled, ATP-competitive kinase inhibitor (kinase tracer-236) to the ITK expression construct in the presence of a Europium-conjugated antibody, resulting in a FRET (fluorescence resonance energy transfer) signal. Displacement of the kinase tracer by compound results in a lower emission ratio upon excitation of the Europium chelate.
- kinase tracer-236 a proprietary, Alexa Fluor® 647-labeled, ATP-competitive kinase inhibitor
- FRET fluorescence resonance energy transfer
- Candidate compounds are designed as potential irreversible inhibitors of ITK, capable of ligating to an active site cysteine residue resulting in time dependent covalent binding.
- the time dependent nature of irreversible inhibition is investigated by performing the binding assay with and without a pre-incubation of compound and ITK. An increase in potency in the pre-incubated assay suggests the candidate compound could be irreversibly modifying ITK or having a slowly reversible mechanism.
- the inhibitory potency of candidate compounds is measured in 20 mM HEPES pH 7.5, 10 mM MgCl 2 , 0.01% BSA, 0.0005% Tween-20, and 2% DMSO in the presence of 10 nM ITK, 2 nM Eu-anti-GST antibody, and 50 nM kinase tracer-236 using a 384-well plate format. Background signal is defined in the absence of ITK and uninhibited signal is defined in the presence of vehicle (2% DMSO) alone. Compounds were evaluated in an 11-point dose-response ranging from 20 uM to 0.34 nM. The binding assays are performed under two preincubation Conditions to evaluate time dependence of inhibition.
- ITK and Eu-anti-GST antibody are pre-incubated with compound or vehicle for two hours prior to the addition of kinase tracer.
- the non-preincubated assay is run under Conditions where ITK and Eu-anti-GST antibody are added to a mixture of compound and kinase tracer.
- IC 50 values of compounds are determined using a 4 parameter logistical fit of emission ratio as a function of the concentration of compound.
- association and dissociation experiments were performed. Using the same TR-FRET buffer and Conditions as described above, the kinetic rate constants for the binding of KT-236 to ITK were initially determined. Once knowing the association (k on ) and dissociation (k off ) rates of KT-236 binding, the binding kinetics for these candidate compounds could be further elucidated. The dissociation rates were determined by pre-incubating ITK for two hours with various concentrations of the candidate compounds. After this period of time, excess (200 nM final) KT-236 was added to the incubation and the resulting increase in fluorescence followed for approximately four hours.
- ITK association rate
- KT-236 50 nM
- various concentrations of candidate compound added.
- the resulting decrease in signal was then evaluated for the next two hours.
- Data from both sets of time course experiments was then analyzed using Dynafit software to obtain the kinetic rate constants (k on , k off , kinact, K i ) utilizing multiple reaction models.
- Target modulation was based upon the ability of a compound to inhibit ITK phosphorylation of its substrate, PLC ⁇ 1, at Y783.
- Human Jurkat T cells were placed in snap-cap polypropylene tubes at 4 million per tube in 800 ⁇ L medium (RPMI1640 with 10% heat inactivated serum and 12.3 mM 2-mercaptoethanol and supplemented with glutamine/penicillin/streptomycin).
- Compounds were added as 100 ⁇ L of 10 ⁇ working stock solutions in medium with 1% DMSO (or medium with 1% DMSO for Controls) and placed in a 37° C. incubator for 2 hours. Stimulation was carried out using CD3/CD28 Dynabeads to activate the T cell receptor and mimic antigen-presenting cells.
- Beads were added for 100 sec at a ratio of 1 bead per cell, in 100 ⁇ L medium (or medium alone for unstimulated Control). Starting at 70 sec, cells and beads were pelleted in a microfuge for 15 sec, the supernatant aspirated, and 50 ⁇ L lysis buffer was added per tube and mixed with the pellet at the 100 sec time point. Tubes were placed on a rotator for 30 min at 4° C., and cell debris was pelleted in a microfuge. Supernatant was mixed 1:1 with 2 ⁇ Sample Buffer and subjected to SDS-polyacrylamide gel electrophoresis. After transfer to nitrocellulose, immunoblotting was done with anti-phospho-Y783 PLC ⁇ 1 antibody. Immunoblots were visualized with ECL using horseradish peroxidase conjugated secondary antibody.
- This assay was based on the ability of a compound to inhibit ITK mediated IL-2 release.
- Jurkat cells were placed in wells of a 96-well plate at 500,000 per well in 120 ⁇ L medium (same as above).
- Compounds were added as 15 ⁇ L per well of 10 ⁇ working stock solutions in medium with 1% DMSO (or medium with 1% DMSO for Controls) and placed in a 37° C. incubator for 2 hours.
- Stimulation used CD3/CD28 Dynabeads to activate the T cell receptor and mimic antigen-presenting cells. Beads were added at a ratio of 1 bead per cell, in 15 ⁇ L medium (or medium alone for unstimulated Control).
- IL-2 cytokine assay utilized an electochemiluminescence-based ELISA.
- mice Male Sprague Dawley rats are selected for equal average body weight per group. After fasting, with free access to water sixteen hours prior to test, animals are dosed orally (1 mL) with test compounds in a vehicle Containing 0.5% methylcellulose and 0.025% surfactant. The Control group is dosed with vehicle alone.
- Anti-inflammatory Efficacy Rat Carrageenan-Induced Analgesia Test: The compounds of the present disclosure will be evaluated for efficacy in vivo in a model of inflammatory analgesia. Methods to determine efficacy in rat carrageenan-induced analgesia test are described in U.S. Pat. No. 5,760,068.
- mice Male Sprague Dawley rats are selected for equal average body weight per group. After fasting, with free access to water sixteen hours prior to test, animals are dosed orally (1 mL) with test compounds in vehicle Containing 0.5% methylcellulose and 0.025% surfactant. Control groups are dosed with vehicle alone.
- mice Six-week-old male DBA/1J mice are obtained from The Jackson Laboratory. At eight weeks of age, mice are orally administered test compounds daily. Mice are immunized by intradermal injection, at twelve weeks of age, with 0.1 mL of emulsion Containing 100 ⁇ g of bovine type II collagen (bCII). At 21 days following immunization, mice are boosted with 0.1 mL of bCII (100 ⁇ g) emulsified in equal volume of incomplete Freund's Adjuvant (IFA) (Difco, Detroit, Mich.).
- IFA incomplete Freund's Adjuvant
- mice are monitored three times for the incidence of arthritis and evaluation of a clinical score, ranging from 0-4 was used (0: no swelling or redness; 1: detectable arthritis with erythema; 2: significant swelling and redness; 3: severe swelling and redness from joint to digit; 4: joint stiffness or deformity with ankylosis).
- the score is calculated from the average cumulative value of all four paws. Severe arthritis is defined as a score >3.
- mice For terminal evaluation of arthritis, mice are euthanized 28 days after initial immunization. The two hind limbs are removed, fixed in formalin, decalcified in RDO solution (Apex Engineering, Aurora, Ill.) for 10-20 min depending on tissue size and examined for pliability. Sections are cut (4 jam thick) and stained with hematoxylin and eosin. Histological evaluation is performed by examining for infiltration of immune cells, hyperplasia, pannus formation and bone deformation for each paw, using a scale ranging from 0-3, according to severity of pathological changes (0: normal, 1: mild, 2: moderate, 3: severe).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
wherein: Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R6 substituents; R2 is chosen from hydrogen, halo and C1-4 alkyl; R3 is chosen from hydrogen, halo and C1-4 alkyl; R4 is chosen from cyano, C(O)CH2R2, C(O)CF3, C(O)CH═CH2, C(O)CR7═CH2, C(O)CH═CHR7, C(O)CR7═CHR7, C(O)CH═CR7R7, C(O)CH═CHCH2R8, C(O)CH═CHC(O)CH2R8, C(O)C(CN)═CH2, C(O)(C(O)NH2)C═CH2, S(O)2CH═CH2, (CH2)mCR7═CR9C(O)Me, (CH2)mCR7═CR9C(O)NH2, (CH2)mCR7═CR9C(O)NHR7, (CH2)mCR7═CR9C(O)N(R7)2, and (CH2)mCR7═CR9CN; R5 is chosen from hydrogen, —(CH2)nC3-7 cycloalkyl, and C1-4 alkyl; each R6 is independently chosen from hydrogen, C1-4 alkyl, —(CH2)nC3-7 cycloalkyl, OC1-4 alkyl, C1-4 alkylamino, NH2, NHC1-4 alkyl, N(C1-4 alkyl)2, cyano, C(O)NH2, C(O)NHR5, C(O)N(R5)2, C(O)C1-4 alkyl, trifluoromethyl, halo, —(CH2)nC3-7 cycloalkyl, C(O)NHaryl, and C(O)NHheteroaryl, wherein aryl and heteroaryl may be optionally substituted with one or more R9; each R7 is independently chosen from hydrogen, CN, C1-4 alkyl, C3-7 cycloalkyl, C3-7 heterocycle, aryl, and heteroaryl, wherein aryl and heteroaryl may be optionally substituted with one or more R9; R8 is chosen from hydrogen, C1-4 alkyl, C1-4alkylaryl, C1-4alkyl-O-aryl, C1-4 alkylheteroarylaryl, C3-7 cycloalkyl, C3-7 heterocycle, OH, OC1-4 alkyl, C1-4 alkylOC1-4 alkyl, NH2, NHC1-4 alkyl, N(C1-4 alkyl)2, heterocycle, aryl and heteroaryl, wherein each aryl and heteroaryl may be optionally substituted with one or more R9; R9 is chosen from hydrogen, C1-4 alkyl, CN, CF3, C(O)Me, C(O)NH2, and aryl; X is chosen from N and CR3; m is chosen from 1, 2 and 3; and n is chosen from 0, 1, 2, and 3.
wherein: Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R6 substituents; R2 is chosen from hydrogen, halo and C1-4 alkyl; R3 is chosen from hydrogen, halo and C1-4 alkyl; R4 is chosen from cyano, C(O)CH2R2, C(O)CF3, C(O)CH═CH2, C(O)CR7═CH2, C(O)CH═CHR7, C(O)CR7═CHR7, C(O)CH═CR7R7, C(O)CH═CHCH2R8, C(O)CH═CHC(O)CH2R8, C(O)C(CN)═CH2, C(O)(C(O)NH2)C═CH2, S(O)2CH═CH2, (CH2)mCR7═CR9C(O)Me, (CH2)mCR7═CR9C(O)NH2, (CH2)mCR7═CR9C(O)NHR7, (CH2)mCR7═CR9C(O)N(R7)2, and (CH2)mCR7═CR9CN; R5 is chosen from hydrogen, —(CH2)nC3-7 cycloalkyl, and C1-4 alkyl; each R6 is independently chosen from hydrogen, C1-4 alkyl, —(CH2)nC3-7 cycloalkyl, OC1-4 alkyl, C1-4 alkylamino, NH2, NHC1-4 alkyl, N(C1-4 alkyl)2, cyano, C(O)NH2, C(O)NHR5, C(O)N(R5)2, C(O)C1-4 alkyl, trifluoromethyl, halo, —(CH2)nC3-7 cycloalkyl, C(O)NHaryl, and C(O)NHheteroaryl, wherein aryl and heteroaryl may be optionally substituted with one or more R9; each R7 is independently chosen from hydrogen, CN, C1-4 alkyl, C3-7 cycloalkyl, C3-7 heterocycle, aryl, and heteroaryl wherein aryl and heteroaryl may be optionally substituted with one or more R9; R8 is chosen from hydrogen, C1-4 alkyl, C1-4alkylaryl, C1-4alkyl-O-aryl, C1-4 alkylheteroarylaryl, C3-7 cycloalkyl, C3-7 heterocycle, OH, OC1-4 alkyl, C1-4 alkylOC1-4 alkyl, NH2, NHC1-4 alkyl, N(C1-4 alkyl)2, heterocycle, aryl and heteroaryl, wherein each aryl and heteroaryl may be optionally substituted with one or more R9; R9 is chosen from hydrogen, C1-4 alkyl, CN, CF3, C(O)Me, C(O)NH2, and aryl; X is chosen from N and CR3; and m is chosen from 1, 2 and 3.
wherein: Ar is chosen from aryl and heteroaryl, wherein Ar may be optionally substituted with one or more R6 substituents; R2 is chosen from hydrogen, halo and C1-4 alkyl; R3 is chosen from hydrogen, halo and C1-4 alkyl; R4 is chosen from cyano, C(O)CH2R2, C(O)CF3, C(O)CH═CH2, C(O)CR7═CH2, C(O)CH═CHR7, C(O)CR7═CHR7, C(O)CH═CR7R7, C(O)CH═CHCH2R8, C(O)CH═CHC(O)CH2R8, C(O)C(CN)═CH2, C(O)(C(O)NH2)C═CH2, S(O)2CH═CH2, (CH2)mCR7═CR9C(O)Me, (CH2)mCR7═CR9C(O)NH2, (CH2)mCR7═CR9C(O)NHR7, (CH2)mCR7═CR9C(O)N(R7)2, and (CH2)mCR7═CR9CN; R5 is chosen from hydrogen, —(CH2)nC3-7 cycloalkyl, and C1-4 alkyl; each R6 is independently chosen from hydrogen, C1-4 alkyl, —(CH2)nC3-7 cycloalkyl, OC1-4 alkyl, C1-4 alkylamino, NH2, NHC1-4 alkyl, N(C1-4 alkyl)2, cyano, C(O)NH2, C(O)NHR5, C(O)N(R5)2, C(O)C1-4 alkyl, trifluoromethyl, halo, —(CH2)nC3-7 cycloalkyl, C(O)NHaryl, and C(O)NHheteroaryl, wherein aryl and heteroaryl may be optionally substituted with one or more R9; each R7 is independently chosen from hydrogen, CN, C1-4 alkyl, C3-7 cycloalkyl, C3-7 heterocycle, aryl, and heteroaryl wherein aryl and heteroaryl may be optionally substituted with one or more R9; R8 is chosen from hydrogen, C1-4 alkyl, C1-4alkylaryl, C1-4alkyl-O-aryl, C1-4 alkylheteroarylaryl, C3-7 cycloalkyl, C3-7 heterocycle, OH, OC1-4 alkyl, C1-4 alkylOC1-4 alkyl, NH2, NHC1-4 alkyl, N(C1-4 alkyl)2, heterocycle, aryl and heteroaryl, wherein each aryl and heteroaryl may be optionally substituted with one or more R9; R9 is chosen from hydrogen, C1-4 alkyl, CN, CF3, C(O)Me, C(O)NH2, and aryl; m is chosen from 1, 2 and 3.
| TABLE 1 |
| Compound Examples - Spectral Data |
| Example | Structure | Name | Spectral Data |
| 7 |
|
5-((1-acryloylazetidin-3-yl)amino)- 2′-methyl-[3,4′-bipyridin]-6(1H)- one | 1H NMR (300 MHz, DMSO) δ 8.37-8.38 (m, 1 H), 7.39-7.48 (m , 2 H), 7.27 (s, 1 H), 6.50 (s, 1 H), 6.20-6.34 (m , 2 H), 6.08-6.22 (d, 1 H), 5.65-5.67 (d, 1 H), 5.65-5.75 (d, 1 H), 4.59-4.63 (t, 1 H), 4.26-4.34 (m, 2 H), 4.08-4.13 (t, 1H), 3.86-3.91 (m,1 H), 2.48-2.50 (m, 3 H); MS (ES) m/z 311 (M + H). |
| 8 |
|
3-((1-acryloylazetidin-3-yl)amino)- 5-(6-methoxypyrazin-2-yl) pyridin-2(1H)-one | 1H NMR (400 MHz, DMSO-d6) δ 8.13 (s, 1 H),7.75 (s, 1 H), 7.48-7.49 (m, 1 H), 6.98 (m, 1 H), 6.75 (s, 1 H), 6.29-6.36 (m, 1 H), 6.07-6.20 (m, 2 H), 5.64-5.75 (d, 1 H), 4.33-4.57 (t, 1 H),4.26-4.30 (m, 2 H), 4.11-4.14 (m, 1 H), 3.82-3.93 (m, 1 H), 3.37-3.49 (m, 1 H), 2.84-2.85 (m, 3 H); MS (ES) m/z 328 (M + H). |
| 9 |
|
5-((1-acryloylazetidin-3-yl)amino)- 2′-(methylamino)-[3,4′- bipyridin]-6(1H)-one | 1H NMR (300 MHz, DMSO) δ 7.93-7.95 (d, 1 H), 7.08 (s, 1 H), 6.67-6.69 (d, 1 H), 6.54 (s, 1 H), 6.31-6.35 (m, 3 H), 6.22-6.44 (d, 1 H), 6.14-6.12 (m, 1 H), 5.65-5.75 (d, 1 H), 4.56- 4.60 (m, 1 H), 4.26-4.34 (m, 2 H), 4.11-4.14 (m, 1 H), 3.89-3.91 (m, 1 H), 2.78-2.79 (m, 3H); MS (ES) m/z 326 (M + H). |
| 10 |
|
3-((1-acryloylazetidin-3-yl)amino)- 5-(2-(methylamino)pyrimidin- 4-yl)pyridin-2(1H)-one | 1H NMR (300 MHz, DMSO) δ 11.84 (s, 1 H), 8.22 (m, 1 H), 7.03-7.57 (m, 2 H), 6.77-6.99 (m, 2 H), 6.29 (s, 1 H), 6.21-6.23 (m , 1 H), 6.08-6.09 (m, 1 H), 5.83-6.08 (d, 1 H), 5.65-5.82 (d, 1 H), 4.49-4.56 (t, 1 H), 4.08-4.21 (m, 2 H), 3.81-3.94 (m, 1 H), 3.86-3.91 (m, 1 H), 2.83-2.84 (m, 3 H); MS (ES) m/z 327 (M + H). |
| 11 |
|
3-((1-acryloylazetidin-3-yl)amino)- 5-(6-(methylamino)pyrazin-2-yl) pyridin-2(1H)-one | 1H NMR (400 MHz, DMSO-d6) δ 8.68 (s, 1 H), 8.12 ( s, 1 H), 7.60-7.61 (m, 1 H), 6.80 (s , 1 H), 6.29-6.39 (m, 2 H), 6.08- 6.12 (d, 1 H), 5.64-5.68 (d, J = 16 Hz, 1 H), 4.36-4.50 (t, 1H), 4.27-4.36 (m, 2 H), 4.12-4.15 (m, 1 H), 3.91-3.97 (m, 4 H), 3.07 (br s,1 H); MS (ES) m/z 327 (M + H). |
| 12 |
|
(E)-N-methyl-4-oxo-4-(3-((6-oxo- 1,6-dihydro-[3,4′-bipyridin]-5-yl) amino)azetidin-l-yl)but-2-enamide | 1H NMR (400 MHz, DMSO-d6) δ 8.50- 8.56 (m, 3 H), 7.60-7.62 (m, 1 H), 7.29- 7.30 (m, 1 H), 6.49-6.50 (s, 1 H), 6.17- 6.20 (m, 2 H), 6.04-6.07 (d, 1 H), 4.27- 4.41 (m, 3 H) , 3.86-3.95 (d, 2 H), 2.64- 2.68 (m, 3 H), 1.22 (m, 3 H); MS (ES) m/z 354 (M + H). |
| 13 |
|
(E)-5-((1-(4-(methylamino)but-2- enoyl)azetidin-3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | 1H NMR (400 MHz, DMSO-d6) δ 9.20 (br s, 2 H), 8.73-8.83 (m, 2 H), 8.3-8.31 (m, 2 H), 7.81 (br s , 1 H), 6.59-6.76 (m, 2 H), 6.38-6.42 (d, 1 H), 4.47-4.63 (t, 1 H) ,4.35-4.44 (m, 1 H),4.16-4.28 (t, 1 H), 4.09-4.1 (t, 1 H), 3.89-3.95 (m, 2 H), 3.38 (s, 1 H); MS (ES) m/z 340 (M + H). |
| TABLE 2 |
| Compound Examples |
| Ex- | |||
| ample | Structure | Name | Smiles |
| 1 | | (R)-5-((1-(2- chloroacetyl)piperidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(CC1)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 2 | | (R)-5-((1- acryloylpiperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(C═C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 3 | | (R)-5-((1- acryloylpyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(C═C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 4 | | 5-((1-(2- chloroacetyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(CC1)═O)C2)═ CC(C3═CC═NC═C3)═CN1 |
| 5 | | 5-((1-acryloylazetidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC═NC═C3)═CN1 |
| 6 | | (E)-5-((1-(but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/C)═O)C2)═ CC(C3═CC═NC═C3)═CN1 |
| 7 | | 5-((1-acryloylazetidin- 3-yl)amino)-2′-methyl- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(C)═NC═C3)═CN1 |
| 8 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═NC(OC)═CN═C3)═CN1 |
| 9 | | 5-((1-acryloylazetidin- 3-yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(NC)═NC═C3)═CN1 |
| 10 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═NC(NC)═NC═C3)═CN1 |
| 11 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═NC(NC)═CN═C3)═CN1 |
| 12 | | (E)-N-methyl-4-oxo-4- (3-((6-oxo-1,6-dihydro- [3,4′-bipyridin]-5- yl)amino)azetidin-1- yl)but-2-enamide | O═C1C(NC2CN(C(/C═C/C(NC)═O)= O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 13 | | (E)-5-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 14 | | (E)-5-((1- cinnamoylazetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/C3═CC═ CC═C3)═O)C2)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 15 | | (E)-2′-(methylamino)- 5-((1-(4-phenylbut-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3═CC═ CC═C3)═O)C2)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 16 | | 5-((1-acryloylazetidin- 3-yl)amino)-N-(4- isopropyl-3- methylphenyl)-6-oxo- 1,6-dihydro-[3,4′- bipyridine]-2′- carboxamide hydrochloride | |
| 17 | | 5-((1-(2- chloroacetyl)azetidin-3- yl)amino)-N-(4- isopropyl-3- methylphenyl)-6-oxo- 1,6-dihydro-[3,4′- bipyridine]-2′- carboxamide formate | |
| 18 | | (E)-2′-(methylamino)- 5-((1-(pent-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC)═ O)C2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 19 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC(NC)═NC═C3)═ CN1 |
| 20 | | (E)-2′-(methylamino)- 5-((1-(5-phenoxypent- 2-enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CCOC3═ CC═CC═C3)═O)C2)═CC(C4═CC (NC)═NC═C4)═CN1 |
| 21 | | (E)-2-(3-((2′- (methylamino)-6-oxo- 1,6-dihydro[3,4′- bipyridin]-5- yl)amino)azetidine-1- carbonyl)but-2- enenitrile | O═C1C(NC2CN(C(/C(C#N)═C/C)═ O)C2)═CC(C3═CC═NC(NC)═ C3)═CN1 |
| 22 | | (E)-2-(3-((6-oxo-1,6- dihydro-[3,4′- bipyridin]-5- yl)amino)azetidine-1- carbonyl)but-2- enenitrile | O═C1C(NC2CN(C(/C(C#N)═C/C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 23 | | (E)-2′-(methylamino)- 5-((1-(3-(pyridin-2- yl)acryloyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/C3═CC═ CC═N3)═O)C2)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 24 | | (E)-2′-(methylamino)- 5-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 25 | | (E)-2′-(methylamino)- 5-((1-(4- methylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CNC)═O) CC2)═CC(C3═CC(NC)═NC═C3)═ CN1 |
| 26 | | (E)-2′-(methylamino)- 5-((1-(4-(piperidin-1- yl)but-2-enoyl)azetidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═ C/CN3CCCCC)═O)C2)═ CC(C4═CC(NC)═NC═ C4)═CN1 |
| 27 | | (E)-2′-(methylamino)- 5-((1-(4-(pyridin-2- yl)but-2-enoyl)azetidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3═NC═ CC═C3)═O)C2)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 28 | | (E)-2′-(methylamino)- 5-((1-(4-(pyrrolidin-1- yl)but-2-enoyl)azetidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═ C/CN3CCCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 29 | | (E)-2′-(methylamino)- 5-((1-(4-(tetrahydro- 2H-pyran-4-yl)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═ C/CC3CCOCC3)═O)C2)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 30 | | (E)-2′-(methylamino)- 5-((1-(4- morpholinobut-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═ C/CN3CCOCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 31 | | (E)-2′-methoxy-5-((1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 32 | | (E)-2′-methoxy-5-((1- (4-(methylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CNC)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 33 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ CC(NC)═NC═N3)═CN1 |
| 34 | | (E)-3-(((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ CC(OC)═NC═N3)═CN1 |
| 35 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ NC(NC)═CN═C3)═CN1 |
| 36 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ NC(NC)═NC═C3)═CN1 |
| 37 | | (E)-34(1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ NC(OC)═CN═C3)═CN1 |
| 38 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ NC(OC)═NC═C3)═CN1 |
| 39 | | E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5- (pyrazin-2-yl)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ NC═CN═C3)═CN1 |
| 40 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═ NC═NC═C3)═CN1 |
| 41 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC(NC)═NC═C3)═CN1 |
| 42 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC(OC)═NC═C3)═CN1 |
| 43 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)primidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC(OC)═NC═N3)═CN1 |
| 44 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC(NC)═CN═C3)═CN1 |
| 45 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC(OC)═CN═C3)═CN1 |
| 46 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC(OC)═CN═C3)═CN1 |
| 47 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 48 | | (E)-3-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═NC═NC═C3)═CN1 |
| 49 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(NC)═NC═N3)═CN1 |
| 50 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(OC)═NC═N3)═CN1 |
| 51 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(NC)═CN═C3)═CN1 |
| 52 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(NC)═NC═C3)═CN1 |
| 53 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(OC)═CN═C3)═CN1 |
| 54 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(OC)═NC═C3)═CN1 |
| 55 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5- (pyrazin-2-yl)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 56 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)(methyl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC═NC═C3)═CN1 |
| 57 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(NC)═NC═N3)═CN1 |
| 58 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(OC)═NC═N3)═CN1 |
| 59 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(NC)═CN═C3)═CN1 |
| 60 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(NC)═NC═C3)═CN1 |
| 61 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(OC)═CN═C3)═CN1 |
| 62 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC(OC)═NC═C3)═CN1 |
| 63 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 64 | | (E)-3-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═NC═NC═C3)═CN1 |
| 65 | | (E)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC(NC)═NC═C3)═CN1 |
| 66 | | (E)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(NC)═NC═N3)═CN1 |
| 67 | | (E)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6 (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC(NC)═CN═C3)═CN1 |
| 68 | | (E)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 69 | | (E)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC═NC═C3)═CN1 |
| 70 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CN)═ O(C3═CC(C4═CC(NC)═NC═ N4)═CN1 |
| 71 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)cycloproplmethyl) amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC(OC)═ NC═N4)═CN1 |
| 72 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═NC(NC)═ CN═C4)═CN1 |
| 73 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═NC(NC)═NC═ C4)═CN1 |
| 74 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═NC(OC)═ CN═C4)═CN1 |
| 75 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)cycloproplmethyl) amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═NC(OC)═ NC═C4)═CN1 |
| 76 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═NC═CN═ C4)═CN1 |
| 77 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-5-(pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═NC═NC═ C4)═CN1 |
| 78 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(NC)═NC═ C3)═CN1 |
| 79 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(OC)═NC═ C3)═CN1 |
| 80 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═CC(NC)═NC═ N3)═CN1 |
| 81 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(NC)═CN═ C3)═CN1 |
| 82 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3=CC(OC)═NC═ N3)═CN1 |
| 83 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(OC)═CN═ C3)═CN1 |
| 84 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5- (pyrazin-2-yl)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC═CN═C3)═ CN1 |
| 85 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(methyl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC═NC═C3)═CN1 |
| 86 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(NC)═NC═ C3)═CN1 |
| 87 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(OC)═NC═ C3)═CN1 |
| 88 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(/C═C/CN)═ O)C2)═CC(C3═CC(NC)═NC═ N3)═CN1 |
| 89 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═ O)C2)═CC(C3═NC(NC)═CN═ C3)═CN1 |
| 90 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 91 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═NC(OC)═CN═C3)═CN1 |
| 92 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═NC═CN═C3)═CN1 |
| 93 | | (E)-3-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═NC═NC═C3)═CN1 |
| 94 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC(NC)═ NC═N4)═CN1 |
| 95 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC(OC)═ NC═N4)═CN1 |
| 96 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═NC(NC)═ CN═C4)═CN1 |
| 97 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═NC(NC)═ NC═C4)═CN1 |
| 98 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═NC(OC)═ CN═C4)═CN1 |
| 99 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═NC(OC)═ NC═C4)═CN1 |
| 100 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═NC═CN═ C4)═CN1 |
| 101 | | (E)-3- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═NC═NC═ C4)═CN1 |
| 102 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC(NC)═ NC═N4)═CN1 |
| 103 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6 methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC(OC)═ NC═N4)═CN1 |
| 104 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═NC(NC)═ CN═C4)═CN1 |
| 105 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═NC(NC)═ NC═C4)═CN1 |
| 106 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═NC(OC)═ CN═C4)═CN1 |
| 107 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═NC(OC)═ NC═C4)═CN1 |
| 108 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═NC═CN═ C4)═CN1 |
| 109 | | (E)-3- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═NC═NC═ C4)═CN1 |
| 110 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC(NC)═NC═ N4)═CN1 |
| 111 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC(OC)═NC═ N4)═CN1 |
| 112 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═NC(NC)═CN═ C4)═CN1 |
| 113 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ CCNC)═O)C3)═CC(C4═NC(NC)═NC═ C4)═CN1 |
| 114 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═NC(OC)═CN═ C4)═CN1 |
| 115 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═NC(OC)═NC═ C4)═CN1 |
| 116 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═NC═CN═ C4)═CN1 |
| 117 | | (E)-3- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═NC═NC═ C4)═CN1 |
| 118 | | (E)-3-(5-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-N- (4-isopropyl-3- methylphenyl)-5- methylbenzamide | O═C(NC1═CC(C)═C(C(C)C)C═C1)C2═ CC(C3═CNC(C(NC4CN(C(/C═ C/CN(C)C)═O)C4)═C3)═O)═ CC(C)═C2 |
| 119 | | (E)-3-(5-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)- (4-isopropyl-3- methylphenyl)benzamide | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC(C(NC4═CC(C)═ C(C(C)C)C═C4)═O)═CC═C3)═CN1 |
| 120 | | (E)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(NC)═NC═N3)═ CN1 |
| 121 | | (E)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC(NC)═CN═C3)═ CN1 |
| 122 | | (E)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 123 | | (E)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 124 | | (E)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-5-(pyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC═CN═C3)═CN1 |
| 125 | | (E)-3-cyano-5-(5-((1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-N- (4-isopropyl-3- methylphenyl)benzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN(C(/C═ C/CN(C)C)═O)C4)═C3)═O)═ CC(C#N)═C2 |
| 126 | | (E)-4-(methylamino)- 5′-((1-(4- (methylamino)but-2- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(NC)═CC═ N3)═CN1 |
| 127 | | (E)-4-methoxy-5′-((1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(OC)═CC═ N3)═CN1 |
| 128 | | (E)-5′-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC═CC═N3)═CN1 |
| 129 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 130 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC(OC)═NC═C3)═CN1 |
| 131 | | (E)-5′-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC(NC)═CC═N3)═CN1 |
| 132 | | (E)-5′-((1-(4- (dimethylamino)but-2- enoyl)azetidin-3- yl)amino)-4-methoxy- [2,3′-bipyridin[-6′(1′H)- one | O═C1C(NC2CN(C(/C═C/CN(C)C)═ O)C2)═CC(C3═CC(OC)═CC═N3)═CN1 |
| 133 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-4- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═C/CN(C)C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 134 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-4- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]1-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 135 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-4- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 136 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CN(C)C)═ O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 137 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CN(C)C)═ O)CC2)═CC(C3═CC(NC)═NC═C3)═ CN1 |
| 138 | | (E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CCN(C(/C═C/CN(C)C)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 139 | | (E)-5′-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC═CC═N3)═CN1 |
| 140 | | (E)-5-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 141 | | (E)-5-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-2′ (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(NC)═NC═C3)═CN1 |
| 142 | | (E)-5-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(OC)═NC═C3)═CN1 |
| 143 | | (E)-5′-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(NC)═CC═N3)═CN1 |
| 144 | | (E)-5′-((1-(4- (isopropylamino)but-2- enoyl)azetidin-3- yl)amino)-4-methoxy- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(NC2CN(C(/C═C/CNC(C)C)═ O)C2)═CC(C3═CC(OC)═CC═N3)═CN1 |
| 145 | | (E)-5′-((1-(4- (isopropylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CNC(C)C)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 146 | | (E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CNC(C)C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 147 | | (E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CCN(C(/C═C/CNC(C)C)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 148 | | (E)-5′-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC═CC═N3)═CN1 |
| 149 | | (E)-5-((1-(4- (methylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CNC)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 150 | | (E)-5-((1-(4-(piperidin- 1-yl)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN3CCCCC3)═ O)C2)═CC(C4═CC═NC═C4)═CN1 |
| 151 | | (E)-5-((1-(4-(pyridin-2- yl)but-2-enoyl)azetidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3═NC═ CC═C3)═O)C2)═CC(C4═CC═NC═ C4)=CN1 |
| 152 | | (E)-5-((1-(4- (pyrrolidin-1-yl)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN3CCCC3)═ O)C2)═CC(C4═CC═NC═C4)═CN1 |
| 153 | | (E)-54(1-(4- (tetrahydro-2H-pyran- 4-yl)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3CCOCC3)═ O)C2)═CC(C4═CC═NC═C4)═CN1 |
| 154 | | (E)-5′-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC(NC)═CC═ N4)═CN1 |
| 155 | | (E)-5′-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-4-methoxy- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC(OC)═CC═ N4)═CN1 |
| 156 | | (E)-5′-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-[2,3′-bipyridin]- 6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC═CC═ N4)═CN1 |
| 157 | | (E)-5-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC(NC)═NC═ C4)═CN1 |
| 158 | | (E)-5-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 159 | | (E)-5-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN)═O)C3)═CC(C4═CC═NC═ C4)═CN1 |
| 160 | | (E)-5′-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═CC═CC═N3)═CN1 |
| 161 | | (E)-5-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═CC═NC═C3)═CN1 |
| 162 | | (E)-5-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═CC(NC)═NC═C3)═CN1 |
| 163 | | (E)-5-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═CC(OC)═NC═C3)═CN1 |
| 164 | | (E)-5′-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═CC(NC)═CC═N3)═CN1 |
| 165 | | (E)-5′-((1-(4-aminobut- 2-enoyl)azetidin-3- yl)amino)-4-methoxy- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(NC2CN(C(/C═C/CN)═O)C2)═ CC(C3═CC(OC)═CC═N3)═CN1 |
| 166 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)(cyclopropylmethyl) amino)2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CN)═O)CC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 167 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CN)═O)CC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 168 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CN)═O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 169 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═C/CN)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 170 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═C/CN)═ O)CC2)═CC(C3═CC(NC)═NC═C3)═ CN1 |
| 171 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═C/CN)═ O)CC2)═CC(C3═CC(OC)═NC═C3)═ CN1 |
| 172 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CN)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 173 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(/C═C/CN)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 174 | | (E)-5-((1-(4-aminobut- 2-enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(NC2CCN(C(/C═C/CN)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 175 | | (E)-5-((1-(4- cyclohexylbut-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3CCCCC3)═ O)C2)═CC(C4═CC═NC═C4)═CN1 |
| 176 | | (E)-5-((1-(4- cyclohexylbut-2- enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3CCCCC3)═ O)C2)═CC(C4═CC(NC)═NC═C4)═ CN1 |
| 177 | | (E)-5-((1-(4- isopropoxybut-2- enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/COC(C)C)═ O)C2)═CC(C3═CC(NC)═NC═C3)═CN1 |
| 178 | | (E)-5-((1-(4- methoxybut-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/COC)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 179 | | (E)-5-((1-(4- methoxybut-2- enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/COC)═ O)C2)═CC(C3═CC(NC)═NC═C3)═ CN1 |
| 180 | | (E)-5-((1-(4- morpholinobut-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CN3CCOCC3)═ O)C2)═CC(C4═CC═NC═C4)═CN1 |
| 181 | | (E)-5-((1-(4-phenylbut- 2-enoyl)azetidin-3 yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(/C═C/CC3═CC═ CC═C3)═O)C2)═CC(C4═CC═NC═ C4)═CN1 |
| 182 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC(NC)═ CC═N4)═CN1 |
| 183 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-4-methoxy- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC(OC)═ CC═N4)═CN1 |
| 184 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC═CC═ N4)═CN1 |
| 185 | | (E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 186 | | (E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 187 | | (E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CN(C)C)═O)C3)═CC(C4═CC═ NC═C4)═CN1 |
| 188 | | (E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one- | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CN(C)C)═O)CC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 189 | | (E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CN(C)C)═O)CC3)═CC(C4═CC(OC)= NC═C4)═CN1 |
| 190 | | (E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CN(C)C)═O)CC3)═CC(C4═CC═ NC=C4)═CN1 |
| 191 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC(NC)═ CC═N4)═CN1 |
| 192 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-4-methoxy- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC(OC)═ CC═N4)═CN1 |
| 193 | | (E)-5′- ((cyclopropylmethyl) (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC═ CC═N4)═CN1 |
| 194 | | (E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 195 | | (E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 196 | | (E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC(C)C)═O)C3)═CC(C4═CC═ NC═C4)═CN1 |
| 197 | | (E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CNC(C)C)═O)CC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 198 | | (E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CNC(C)C)═O)CC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 199 | | (E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CNC(C)C)═O)CC3)═CC(C4═CC═ NC═C4)═CN1 |
| 200 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H-1)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC(NC)═ CC═N4)═CN1 |
| 201 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-4-methoxy- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC(OC)═ CC═N4)═CN1 |
| 202 | | (E)-5′- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC═CC═ N4)═CN1 |
| 203 | | (E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 204 | | (E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 205 | | (E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)azetidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(/C═ C/CNC)═O)C3)═CC(C4═CC═NC═ C4)═CN1 |
| 206 | | (E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CNC)═O)CC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 207 | | (E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/ CNC)═O)CC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 208 | | (E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(/C═ C/CNC)═O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 209 | | (E)-5-(2- methoxypyrimidin-4- yl)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)pyridin- 2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC(OC)═NC═ C3)═CN1 |
| 210 | | (E)-5-(2- methoxypyrimidin-4- yl)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC(OC)═NC═C3)═CN1 |
| 211 | | (E)-5-(6- methoxypyrazin-2-yl)- 3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)pyridin- 2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═NC(OC)═CN═ C3)═CN1 |
| 212 | | (E)-5-(6- methoxypyrazin-2-yl)- 3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)═CC(C3═CC(OC)═NC═N3)═CN1 |
| 213 | | (E)-5-(6- methoxypyrimidin-4- yl)-3-((1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)pyridin- 2(1H)-one | O═C1C(NC2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 214 | | (E)-5-(6- methoxypyrimidin-4- yl)-3-(methyl(1-(4- (methylamino)but-2- enoyl)azetidin-3- yl)amino)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(/C═C/CNC)═ O)C2)═CC(C3═CC(OC)═NC═N3)═CN1 |
| 215 | | (E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 216 | | (E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)C2CCN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 217 | | (E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC═NC═C3)═CN1 |
| 218 | | (E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-4- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 219 | | (E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-4- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)C2CCN(C(/C═C/CNC)═ O)CC2)═CC(C3═CC(OC)═NC═C3)═ CN1 |
| 220 | | (E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-4- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(/C═C/CNC═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 221 | | (R)-5-((1-(2- chloroacetyl)piperidin- 3-yl)amino)-2′- methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@]2CCCN(C(CC1)═ O)C2)═CC(C3═CC(NC)═NC═C3)═CN1 |
| 222 | | (R)-5-((1-(2- chloroacetyl)pyrrolidin- 3-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(CC1)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 223 | | (R)-5-((1-(2- chloroacetyl)pyrrolidin- 3-yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(CC1)═ O)C2)═CC(C3═CC(NC)═NC═C3)═CN1 |
| 224 | | (R)-5-((1- acryloylpiperidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(C═ C)═O)CCC3)═CC(C4═CC═NC═C4)═ CN1 |
| 225 | | (R)-5-((1- acryloylpiperidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(CC)═ O)CCC3)═CC(C4═CC(NC)═NC═ C4)═CN1 |
| 226 | | (R)-5-((1- acryloylpiperidin-3- yl)cycloproplmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(C═ C)═O)CCC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 227 | | (R)-5-((1- acryloylpiperidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC═NC═C3)═CN1 |
| 228 | | (R)-5-((1- acryloylpiperidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC(NC)═NC═C3)═ CN1 |
| 229 | | (R)-5-((1- acryloylpiperidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC(OC)═NC═C3)═ CN1 |
| 230 | | (R)-5-((1- acryloylpiperidin-3- yl)(methyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(C)[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC═NC═C3)═ CN1 |
| 231 | | (R)-5-((1- acryloylpiperidin-3- yl)(methyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 232 | | (R)-5-((1- acryloylpiperidin-3- yl)(methyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 233 | | (R)-5-((1- acryloylpiperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(C═C)= O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 234 | | (R)-5-((1- acryloylpiperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═C3)═CN1 |
| 235 | | (R)-5-((1- acryloylpiperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 236 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(C═ C)═O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 237 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(C═ C)═O)CC3)═CC(C4═CC(NC)═NC═ C4)═CN1 |
| 238 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(C═ C)═O)CC3)═CC(C4═CC(OC)═NC═ C4)═CN1 |
| 239 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 240 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 241 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(OC)═NC═C3)═ CN1 |
| 242 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(methyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(C[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 243 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(methyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 244 | | (R)-5-((1- acryloylpyrrolidin-3- yl)(methyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 245 | | (R)-5-((1- acryloylpyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@9H]2CCN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 246 | | (R)-5-((1- acryloylpyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 247 | | (R,E)-2′- (methylamino)-5-((1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CNC)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 248 | | (R,E)-2′- (methylamino)-5-((1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CNC)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 249 | | (R,E)-2′- (methylamino)-5-((1- (4-(piperidin-1-yl)but- 2-enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN3CCCCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 250 | | (R,E)-2′- (methylamino)-5-((1- (4-(piperidin-l-yl)but- 2-enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN3CCCCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 251 | | (R,E)-2′- (methylamino)-5-((1- (4-(pyridin-2-yl)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CC3═NC═CC═C3)═O)C2)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 252 | | (R,E)-2′- (methylamino)-5-((1- (4-(pyridin-2-yl)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CC3═NC═CC═C3)═O)C2)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 253 | | (R,E)-2′- (methylamino)-5-((1- (4-(pyrrolidin-l-yl)but- 2-enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN3CCCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 254 | | (R,E)-2′- (methylamino)-5-((1- (4-(pyrrolidin-l-yl)but- 2-enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN3CCCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 255 | | (R,E)-2′- (methylamino)-5-((1- (4-morpholinobut-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN3CCOCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 256 | | (R,E)-2′- (methylamino)-5-((1- (4-morpholinobut-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN3CCOCC3)═O)C2)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 257 | | (R,E)-2′- (methylamino)-5-((1- (4-phenylbut-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CC3═CC═CC═C3)═O)C2)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 258 | | (R,E)-2′- (methylamino)-5-((1- (4-phenylbut-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CC3═CC═CC═C3)═O)C2)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 259 | | (R,E)-2′-methoxy-5-((1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@H]2CN(C(/C═C/CNC)═ O)CCC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 260 | | (R,E)-2′-methoxy-5-((1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@H]2CN(C(/C═C/CNC)═ O)CC2)═CC(C3═CC(OC)═NC═C3)═ CN1 |
| 261 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 262 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 263 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 264 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═CC═ NC═C3)═CN1 |
| 265 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 266 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(/C═C/CN(C)C)═ O)CCC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 267 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 268 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 269 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 270 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 271 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN(C)C)═O)C2)═CC(C3═CC═ NC═C3)═CN1 |
| 272 | | (R,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 273 | | (R,E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 274 | | (R,E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 275 | | (R,E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 276 | | (R,E)-5-((1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 277 | | (R,E)-5-((1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 278 | | (R,E)-5-((1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@H]2CN(C(/C═ C/CNCC)C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 279 | | (R,E)-5-((1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CNC)═O)C2)═CC(C3═CC═NC═ C3)═CN1 |
| 280 | | (R,E)-5-((1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CNC)═O)C2)═CC(C3═CC═NC═ C3)═CN1 |
| 281 | | (R,E)-5-((1-(4- (piperidin-1-yl)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN3CCCCC3)═O)C2)═CC(C4═ CC═NC═C4)═CN1 |
| 282 | | (R,E)-5-((1-(4- (piperidin-1-yl)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN3CCCCC3)═O)C2)═CC(C4═ CC═NC═C4)═CN1 |
| 283 | | (R,E)-5-((1-(4-(pyridin- 2-yl)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CC3═NC═CC═C3)═O)C2)═ CC(C4═CC═NC═C4)═CN1 |
| 284 | | (R,E)-5-((1-(4-(pyridin- 2-yl)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@CCH]2CCN(C(/C═ C/CC3═NC═CC═C3)═O)C2)═ CC(C4═CC═NC═C4)═CN1 |
| 285 | | (R,E)-5-((1-(4- (pyrrolidin-l-yl)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN3CCCC3)═O)C2)═CC(C4═ CC═NC═C4)═CN1 |
| 286 | | (R,E)-5-((1-(4- (pyrrolidin-l-yl)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN3CCCC3)═O)C2)═CC(C4═ CC═NC═C4)═CN1 |
| 287 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN)═O)CCC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 288 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN)═O)CCC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 289 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN)═O)CCC3)═CC(C4═CC═NC═ C4)═CN1 |
| 290 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN)═O)CCC2)═CC(C3═CC═NC═ C3)═CN1 |
| 291 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN)═O)CCC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 292 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN)═O)CCC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 293 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN)═O)C2)═CC(C3═CC═ NC═C 3)═CN1 |
| 294 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 295 | | (R,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(/C═C/CN)═ O)CCC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 296 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN)═O)CC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 297 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN)═O)CC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 298 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN)═O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 299 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN)═O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 300 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN)═O)CC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 301 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CN)═O)CC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 302 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN)═O)C2)═CC(C3═CC═NC═ C3)═CN1 |
| 303 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CN)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 304 | | (R,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@H]2CN(C(/C═C/CN)= O)CC2)═CC(C3═CC(OC)═NC═C3)═ CN1 |
| 305 | | (R,E)-5-((1-(4- morpholinobut-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/CN3CCOCC3)═O)C2)═CC(C4═ CC═NC═C4)═CN1 |
| 306 | | (R,E)-5-((1-(4- morpholinobut-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/C N3CCOCC3)═O)C2)═CC(C4═ CC═NC═C4)═CN1 |
| 307 | | (R,E)-5-((1-(4- phenylbut-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CC3═CC═CC═C3)═O)C2)═ CC(C4═CC═NC═C4)═CN1 |
| 308 | | (R,E)-5-((1-(4- phenylbut-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/CC3═CC═CC═C3)═O)C2)═ CC(C4═CC═NC═C4)═CN1 |
| 309 | | (R,E)-5-((1-(but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/C)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 310 | | (R,E)-5-((1-(but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCCN(C(/C═ C/C)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 311 | | (R,E)-5-((1-(but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/C)═O)C2)═CC(C3═CC═NC═ C3)═CN1 |
| 312 | | (R,E)-5-((1-(but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CCN(C(/C═ C/C)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 313 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@]3CN(C(/C═ C/CN(C)C)═O)CCC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 314 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN(C)C)═O)CCC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 315 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN(C)C)═O)CCC3)═CC(C4═CC= NC═C4)═CN1 |
| 316 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN(C)C)═O)CC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 317 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN(C)C)═O)CC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 318 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CN(C)C)═O)CC3)═CC(C4═ CC═NC═C4)═CN1 |
| 319 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC(C)C)═O)CCC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 320 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC(C)C)═O)CCC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 321 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC(C)C)═O)CCC3)═CC(C4═CC═ NC═C4)═CN1 |
| 322 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC(C)C)═O)CC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 323 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC(C)C)═O)CC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 324 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC(C)C)═O)CC3)═CC(C4═CC═ NC═C4)═CN1 |
| 325 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC)═O)CCC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 326 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC)═O)CCC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 327 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC)═O)CCC3)═CC(C4═CC═NC═ C4)═CN1 |
| 328 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC)═O)CC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 329 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC)═O)CC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 330 | | (R,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@H]3CN(C(/C═ C/CNC)═O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 331 | | (R,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 332 | | (R,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 333 | | (R,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 334 | | (R,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 335 | | (R,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 336 | | (R,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 337 | | (R,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-2 (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC)═O)CCC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 338 | | (R,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC)═O)CCC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 339 | | (R,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC)═O)CCC2)═CC(C3═CC═NC═ C3)═CN1 |
| 340 | | (R,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 341 | | (R,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 342 | | (R,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@H]2CN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 343 | | (S)-5-((1- acryloylpiperidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(C═C)═O)CCC3)═CC(C4═ CC═NC═C4)═CN1 |
| 344 | | (S)-5-((1- acryloylpiperidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(C═C)═O)CCC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 345 | | (S)-5-((1- acryloylpiperidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(C═C)═O)CCC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 346 | | (S)-5-((1- acryloylpiperidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═CC═ NC═C3)═CN1 |
| 347 | | (S)-5-((1- acryloylpiperidin-3- yl)(ethyl)amino)-2′- methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 348 | | (S)-5-((1- acryloylpiperidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 349 | | (S)-5-((1- acryloylpiperidin-3- yl)(methyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(C[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═CC═ NC═C3)═CN1 |
| 350 | | (S)-5-((1- acryloylpiperidin-3- yl)(methyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 351 | | (S)-5-((1- acryloylpiperidin-3- yl)(methyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 352 | | (S)-5-((1- acryloylpiperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═CC═ NC═C3)═CN1 |
| 353 | | (S)-5-((1- acryloylpiperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN (C(C═C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 354 | | (S)-5-((1- acryloylpiperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(C═C)═ O)CCC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 355 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(cyclopropylmethyl) amino)[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(C═C)═O)CC3)═CC(C4═ CC═NC═C4)═CN1 |
| 356 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(C═C)═O)CC3)═CC(C4═ CC(NC)═NC═C4)═CN1 |
| 357 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(C═C)═O)CC3)═CC(C4═ CC(OC)═NC═C4)═CN1 |
| 358 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(C═C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 359 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(C═C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 360 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@9H]2CN (C(C═C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 361 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(methyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(C)[C@@H]2CN (C(C═C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 362 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(methyl)amino)-2′- (methylamino)-[3,4′ bipyridin]-6(1H)-one | O═C1C(N(C)[C@@H]2CN (C(C═C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 363 | | (S)-5-((1- acryloylpyrrolidin-3- yl)(methyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)[C@@H]2CN (C(C═C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 364 | | (S)-5-((1- acryloylpyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC═NC═C3)═CN1 |
| 365 | | (S)-5-((1- acryloylpyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 366 | | (S)-5-((1- acryloylpyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(C═C)═ O)CC2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 367 | | (S,E)-2′-(methylaninno)- 5-((1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 368 | | (S,E)-2′-(methylamino)- 5-((1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 369 | | (S,E)-2′-methoxy-5-((1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC)═O)CCC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 370 | | (S,E)-2′-methoxy-5-((1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC(OC)═ NC═C3)═CN |
| 371 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 372 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 373 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═CC═ NC═C3)═CN1 |
| 374 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═CC═ NC═C3)═CN1 |
| 375 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 376 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[@@H]2CN(C(/C═ C/CN(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 377 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 378 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 379 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═CC═ NC=C3)═CN1 |
| 380 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 381 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 382 | | (S,E)-5-((1-(4- (dimethylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN(C)C)═O)CC2)═CC(C3═ CC═NC═C3)═CN1 |
| 383 | | (S,E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 384 | | (S,E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 385 | | (S,E)-5-((1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 386 | | (S,E)-5-((1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 387 | | (S,E)-5-((1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 388 | | (S,E)-5-((1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC═NC═C3)═CN1 |
| 389 | | (S,E)-5-((1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 390 | | (S,E)-5-((1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CNC)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 391 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN)═O)CCC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 392 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@H] 3CN(C(/C═C/CN)═O)CCC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 393 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN)═O)CCC3)═ CC(C4═CC═NC═C4)═CN1 |
| 394 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(/C═C/CN)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 395 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(/C═C/CN)═O)CCC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 396 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(/C═C/CN)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 397 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN)═O)CCC2)═CC(C3═CC═ NC═C3)═CN1 |
| 398 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 399 | | (S,E)-5-((1-(4- aminobut-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(/C═ C/CN)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 400 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN)═O)CC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 401 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H ]3CN(C(/C═C/CN)═O)CC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 402 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN)═O)CC3)═ CC(C4═CC═NC═C4)═CN1 |
| 403 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(/C═C/CN)═O)CC2)═CC(C3═ CC═NC═C3)═CN1 |
| 404 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(/C═C/CN)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 405 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN (C(/C═C/CN)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 406 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN)═O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 407 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N[C@@H]2CN(C(/C═ C/CN)═O)CC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 408 | | (S,E)-5-((1-(4- aminobut-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N[C@@H]2CN(C(/C═ C/CN)═O)CC2)═CC(C3═CC(OC)═ NC═C3)═CN1 |
| 409 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN(C)C)═O)CCC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 410 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN(C)C)═O)CCC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 411 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but 2-enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN(C)C)═O)CCC3)═ CC(C4═CC═NC═C4)═CN1 |
| 412 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H]3CN (C(/C═C/CN(C)C)═O)CC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 413 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN(C)C)═O)CC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 414 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(dimethylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CN(C)C)═O)CC3)═ CC(C4═CC═NC═C4)═CN1 |
| 415 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC(C)C)═O) CCC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 416 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC(C)C)═ O)CCC3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 417 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC(C)C)═O)CCC3)═ CC(C4═CC═NC═C4)═CN1 |
| 418 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC(C)C)═O)CC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 419 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC(C)C)═O)CC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 420 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(isopropylamino)but- 2-enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC(C)C)═ O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 421 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC)═O)CCC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 422 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC)═O)CCC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 423 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC)═O)CCC3)═ CC(C4═CC═NC═C4)═CN1 |
| 424 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC)═O)CC3)═ CC(C4═CC(NC)═NC═C4)═CN1 |
| 425 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)[C[@@H] 3CN(C(/C═C/CNC)═O)CC3)═ CC(C4═CC(OC)═NC═C4)═CN1 |
| 426 | | (S,E)-5- ((cyclopropylmethyl)(1- (4-(methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)[C@@H] 3CN(C(/C═C/CNC)═O)CC3)═ CC(C4═CC═NC═C4)═CN1 |
| 427 | | (S,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC(C)C)═ O)CCC2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 428 | | (S,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 429 | | (S,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CCC2)═CC(C3═ CC═NC═C3)═CN1 |
| 430 | | (S,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(NC)═NC═C3)═CN1 |
| 431 | | S,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═ CC(OC)═NC═C3)═CN1 |
| 432 | | (S,E)-5-(ethyl(1-(4- (isopropylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H]2CN(C(/C═ C/CNC(C)C)═O)CC2)═CC(C3═CC═ NC═C3)═CN1 |
| 433 | | (S,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC)═O)CCC2)═ CC(C3═CC(NC)═NC═C3)═CN1 |
| 434 | | S,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC)═O)CCC2)═ CC(C3═CC(OC)═NC═C3)═CN1 |
| 435 | | (S,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)piperidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC)═O)CCC2)═ CC(C3═CC═NC═C3)═CN1 |
| 436 | | (S,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC)═O)CC2)═ CC(C3═CC(NC)═NC═C3)═CN1 |
| 437 | | (S,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-2′-methoxy [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC)═O)CC2)═ CC(C3═CC(OC)═ NC═C3)═CN1 |
| 438 | | (S,E)-5-(ethyl(1-(4- (methylamino)but-2- enoyl)pyrrolidin-3- yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)[C@@H] 2CN(C(/C═C/CNC)═O)CC2)═ CC(C3═CC═NC═C3)═CN1 |
| 439 | | (Z)-4,4-dimethyl-2-(3- ((6-oxo-1,6-dihydro- [3,4′-bipyridin]-5- yl)amino)azetidine-1- carbonyl)pent-2- enenitrile | O═C1C(NC2CN(C(/C(C#N)═ C\C(C)(C)C)═O)C2)═CC(C3═CC═ NC═C3)═CN1 |
| 440 | | 2-(3-((2′- (methylamino)-6-oxo- 1,6-dihydro-[3,4′- bipyridin]-5- yl)amino)azetidine-1- carbonyl)acrylonitrile | O═C1C(NC2CN(C(C(C#N)═C)═ O)C2)═CC(C3═CC═NC(NC)═ C3)═CN1 |
| 441 | | 2-(3-((6-oxo-1,6- dihydro-[3,4′- bipyridin]5- yl)amino)azetidine-1- carbonyl)acrylonitrile | O═C1C(NC2CN(C(C(C#N)═C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 442 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(NC)═NC═ C4)═CN1 |
| 443 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(OC)═NC═ C4)═CN1 |
| 444 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(2-methylpyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(C)═NC═ C4)═CN1 |
| 445 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(NC)═NC═ N4)═CN1 |
| 446 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(NC)═CN═ C4)═CN1 |
| 447 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(6-methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(OC)═CN═ C4)═CN1 |
| 448 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(OC)═NC═ N4)═CN1 |
| 449 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(6-methylpyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(C)═CN═ C4)═CN1 |
| 450 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(6-methylpyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═ O)C3)═CC(C4═CC(C)═NC═ N4)═CN1 |
| 451 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(pyrazin-2-yl)pyridm- 2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC═CN═C4)═CN1 |
| 452 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 5-(pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC═NC═C4)═CN1 |
| 453 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(NC)═NC═ C4)═CN1 |
| 454 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═NC(OC)═NC═ C4)═CN1 |
| 455 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(2- methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═NC(C)═ NC═C4)═CN1 |
| 456 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═CC(NC)═ NC═N4)═CN1 |
| 457 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═NC(NC)═ CN═C4)═CN1 |
| 458 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═CC(OC)═ NC═N4)═CN1 |
| 459 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(6- methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═NC(OC)═ CN═C4)═CN1 |
| 460 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(6- methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═CC(C)═ NC═N4)═CN1 |
| 461 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(6- methylpyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═NC(C)═ CN═C4)═CN1 |
| 462 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(pyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═NC═ CN═C4)═CN1 |
| 463 | | 3-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-5-(pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC2CC2)C3CN (C(C═C)═O)C3)═CC(C4═NC═ NC═C4)═CN1 |
| 464 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(NC)═NC═ C3)═CN1 |
| 465 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (2-methoxypyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(OC)═NC═ C3)═CN1 |
| 466 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (2-methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═NC═ C3)═CN1 |
| 467 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(NC)═CN═ C3)═CN1 |
| 468 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═ N3)═CN1 |
| 469 | | 3((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (6-methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(OC)═CN═ C3)═CN1 |
| 470 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (6-methoxypyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 471 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (6-methylpyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═CN═ C3)═CN1 |
| 472 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (6-methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ N3)═CN1 |
| 473 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (pyrazin-2-yl)pyridin- 2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC═CN═ C3)═CN1 |
| 474 | | 3-((1-acryloylazetidin- 3-yl)(ethyl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═NC═NC═ C3)═CN1 |
| 475 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═NC(NC)═NC═ C3)═CN1 |
| 476 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (2-methoxypyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)=CC(C3═NC(OC)═NC═ C3)═CN1 |
| 477 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (2-methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═NC═ C3)═CN1 |
| 478 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═ N3)═CN1 |
| 479 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═NC(OC)═CN═ C3)═CN1 |
| 480 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (6-methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 481 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (6-methoxypyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 482 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (6-methylpyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═CN═ C3)═CN1 |
| 483 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (6-methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ N3)═CN1 |
| 484 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (pyrazin-2-yl)pyridin- 2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═NC═CN═ C3)═CN1 |
| 485 | | 3-((1-acryloylazetidin- 3-yl)(methyl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═NC═NC═ C3)═CN1 |
| 486 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (2- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2═CC(C3═NC(NC)═NC═ C3)═CN1 |
| 487 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (2-methoxypyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(OC)═NC═ C3)═CN1 |
| 488 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (2-methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═NC═ C3)═CN1 |
| 489 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (6- (methylamino)pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(NC)═CN═ C3)═CN1 |
| 490 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═ CC(C3═CC(NC)═NC═ N3)═CN1 |
| 491 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (6-methoxypyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(OC)═CN═ C3)═CN1 |
| 492 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (6-methoxypyrimidin- 4-yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 493 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (6-methylpyrazin-2- yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═CN═ C3)═CN1 |
| 494 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (6-methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ N3)═CN1 |
| 495 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (pyrazin-2-yl)pyridin- 2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC═CN═ C3)═CN1 |
| 496 | | 3-((1-acryloylazetidin- 3-yl)(propyl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═NC═NC═ C3)═CN1 |
| 497 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(2- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═NC(OC)═NC═ C3)═CN1 |
| 498 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(2- methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═NC═ C3)═CN1 |
| 499 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(6- (methylamino)pyrimidin- 4-yl)pyridin-2(1H)- one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═ N3)═CN1 |
| 500 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(6- methoxypyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ N3)═CN1 |
| 501 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(6- methylpyrazin-2- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═NC(C)═CN═ C3)═CN1 |
| 502 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(6- methylpyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ N3)═CN1 |
| 503 | | 3-((1-acryloylazetidin- 3-yl)amino)-5-(pyrazin- 2-yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═NC═CN═ C3)═CN1 |
| 504 | | 3-((1-acryloylazetidin- 3-yl)amino)-5- (pyrimidin-4- yl)pyridin-2(1H)-one | O═C1C(NC2CN(C(C═C)═ O)C2)═CC C3═NC═NC═ C3)═CN1 |
| 505 | | 3-(5-((1-(2- chloroacetyl)azetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-N- (4-isopropyl-3- methylphenyl)benzamide | O═C1C(NC2CN(C(CC1)═ O)C2)═CC(C3═CC(C(NC4═ CC(C)═C(C(C)C)C═C4)═O)═ CC═C3)═CN1 |
| 506 | | 3-(5-((1- acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-N- (4-isopropyl-3- methylphenyl)-5- methylbenzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN (C(C═C)═O)C4)═C3)═O)═ CC(C)═C2 |
| 507 | | 3-(5-((1- acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-5- ethyl-N-(4-isopropyl-3- methylphenyl)benzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN (C(C═C)═O)C4)═C3)═O)═ CC(C)═C2 |
| 508 | | 3-(5-((1- acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-5- isopropyl-N-(4- isopropyl-3- methylphenyl)benzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN (C(C═C)═O)C4)═C3)═O)═ CC(C)═C2 |
| 509 | | 3-(5((1- acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-N- (4-isopropyl-3- methylphenyl)-5- propylbenzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN (C(C═C)═O)C4)═C3)═O)═ CC(CCC)═C2 |
| 510 | | 3-(5-((1- acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-5- cyclopropyl-N-(4- isopropyl-3- methylphenyl)benzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN (C(C═C)═O)C4)═C3)═O)═ CC(C5CC5)═C2 |
| 511 | | 3-(5-((1- acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-5- cyano-N-(4-isopropyl- 3- methylphenyl)benzamide | O═C(NC1═CC(C)═C(C(C)C)C═ C1)C2═CC(C3═CNC(C(NC4CN (C(C═C)═O)C4)═C3)═O)═ CC(C#N)═C2 |
| 512 | | 3-(5-((1-] acryloylazetidin-3- yl)amino)-6-oxo-1,6- dihydropyridin-3-yl)-N- (4-isopropyl-3- methylphenyl)benzamide | O═C1C(NC2CN(C(C═C)═ O)C2)═CC(C3═CC(C(NC4═CC(C)═ C(C(C)C)C═C4)═O)═CC═C3)═CN |
| 513 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC═CC═N4)═CN1 |
| 514 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- [3,4′-bipyridin]-6(1′H) one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC═NC═C4)═CN1 |
| 515 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- [3,4′-bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(C2CC2)C3CN(C (C═C)═O)C3)═CC(C(C═CN4)═ CC4═O)═CN1 |
| 516 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 1′-methyl-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(C2CC2)C3CN (C(C═C)═O)C3)═CC(C(C═CN4C)═ CC4═O)═CN1 |
| 517 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 2′-(methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 518 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 2′-methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(OC)═ NC═C4)═CN1 |
| 519 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 2′-methyl-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(C)═ NC═C4)═CN1 |
| 520 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 4-(methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(NC)═ CC═N4)═CN1 |
| 521 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 4-methoxy-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(OC)═ CC═N4)═CN1 |
| 522 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropyl)amino)- 4-methyl-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(C2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(C)═CC═ N4)═CN1 |
| 523 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-[2,3′-bipyridin]- 6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC═CC═ N4)═CN1 |
| 524 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C(C═CN4)═CC4═ O)═CN1 |
| 525 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC═NC═ C4)═CN1 |
| 526 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-1′-methyl-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C(C═CN4C)═ CC4═O)═CN1 |
| 527 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(NC)═NC═ C4)═CN1 |
| 528 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-2′-methoxy- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(OC)═NC═ C4)═CN1 |
| 529 | | 5-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-2′-methyl[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(C)═NC═ C4)═CN1 |
| 530 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-4- (methylamino)-[2,3′- bipyridin]-6′(17-1)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(NC)═CC═ N4)═CN1 |
| 531 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-4-methoxy- [2,3′-bipyridin]6′(1′H)- one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(OC)═CC═ N4)═CN1 |
| 532 | | 5′-((1-acryloylazetidin- 3- yl)(cyclopropylmethyl) amino)-4-methyl-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC2CC2)C3CN(C(C═C)═ O)C3)═CC(C4═CC(C)═CC═ N4)═CN1 |
| 533 | | 5′-((1-acryloylazetidin- 3-yl)(ethyl)amino)- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC═CC═N3)═CN1 |
| 534 | | 5-((1-acryloylazetidin- 3-yl)(ethyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC═NC═C3)═CN1 |
| 535 | | 5-((1-acryloylazetidin- 3-yl)(ethyl)amino)- [3,4′-bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(CC)C2CN(C(C═C)═ O)C2═CC(C(C═CN3)═CC3═ O)═CN1 |
| 536 | | 5-((1-acryloylazetidin- 3-yl)(ethyl)amino)-1′- methyl-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C(C═CN3C)═CC3═ O)═CN1 |
| 537 | | 5-((1-acryloylazetidin- 3-yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 538 | | 5-((1-acryloylazetidin- 3-yl)(ethyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 539 | | 5-((1-acryloylazetidin- 3-yl)(ethyl)amino)-2′- methyl[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ C3)═CN1 |
| 540 | | 5′-((1-acryloylazetidin- 3-yl)(ethyl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═CC═ N3)═CN1 |
| 541 | | 5′-((1-acryloylazetidin- 3-yl)(ethyl)amino)-4- methoxy-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═CC═ N3)═CN1 |
| 542 | | 5′-((1-acryloylazetidin- 3-yl)(ethyl)amino)-4- methyl[2,3′-bipyridin]- 6′(1′H)-one | O═C1C(N(CC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═CC═ N3)═CN1 |
| 543 | | 5′-((1-acryloylazetidin- 3-yl)(methyl)amino)- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC═CC═ N3)═CN1 |
| 544 | | 5-((1-acryloylazetidin- 3-yl)(methyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC═NC═ C3)═CN1 |
| 545 | | 5-((1-acryloylazetidin- 3-yl)(methyl)amino)- [3,4′-bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C(C═CN3)═CC3═ O)═CN1 |
| 546 | | 5-((1-acryloylazetidin- 3-yl)(methyl)amino)-1′- methyl-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C(C═CN3C)═CC3═ O)═CN1 |
| 547 | | 5-((1-acryloylazetidin- 3-yl)(methyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 548 | | 5-((1-acryloylazetidin- 3-yl)(methyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 549 | | 5-((1-acryloylazetidin- 3-yl)(methyl)amino)-2′- methyl[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ C3)═CN1 |
| 550 | | 5′-((1-acryloylazetidin- 3-yl)(methyl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H-1)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═CC═ N3)═CN1 |
| 551 | | 5′-((1-acryloylazetidin- 3-yl)(methyl)amino)-4- methoxy-[2,3′- bipyridin]-6′(1′H-1)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═CC═ N3)═CN1 |
| 552 | | 5′-((1-acryloylazetidin- 3-yl)(methyl)amino)-4- methyl[2,3′-bipyridin]- 6′(1′H)-one | O═C1C(N(C)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═CC═ N3)═CN1 |
| 553 | | 5′-((1-acryloylazetidin- 3-yl)(propyl)amino)- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(N(CCC)C2CN(C(C═ C)═O)C2)═CC(C3═CC═ CC═N3)═CN1 |
| 554 | | 5-((1-acryloylazetidin- 3-yl)(propyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CCC)C2CN(C(C═ C)═O)C2)═CC(C3═CC═NC═ C3)═CN1 |
| 555 | | 5-((1-acryloylazetidin- 3-yl)(propyl)amino)- [3,4′-bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(CCC)C2CN(C(C═ C)═O)C2)═CC(C(C═CN3)═ CC3═O)═CN1 |
| 556 | | 5-((1-acryloylazetidin- 3-yl)(propyl)amino)-1′- methyl-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(N(CCC)C2CN(C(C═ C)═O)C2)═CC(C(C═CN3C)═ CC3═O)═CN1 |
| 557 | | 5-((1-acryloylazetidin- 3-yl)(propyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CCC)C2CN(C(C═ C)═O)C2)═CC(C3═CC(NC)═ NC═C3)═CN1 |
| 558 | | 5-((1-acryloylazetidin- 3-yl)(propyl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CCC)C2CN(C(C═ C)═O)C2)═CC(C3═CC(OC)═NC═ C3)═CN1 |
| 559 | | 5-((1-acryloylazetidin- 3-yl)(propyl)amino)-2′- methyl-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(C)═NC═ C3)═CN1 |
| 560 | | 5′-((1-acryloylazetidin- 3-yl)(propyl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(NC)═CC═ N3)═CN1 |
| 561 | | 5′-((1-acryloylazetidin- 3-yl)(propyl)amino)-4- methoxy-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2)═CC(C3═CC(OC)═CC═ N3)═CN1 |
| 562 | | 5′-((1-acryloylazetidin- 3-yl)(propyl)amino)-4- methyl-[2,3′-bipyridin]- 6′(1′H)-one | O═C1C(N(CCC)C2CN(C(C═C)═ O)C2═CC(C3═CC(C)═CC═ N3)═CN1 |
| 563 | | 5′-((1-acryloylazetidin- 3-yl)amino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(NC)═CC═N3)═CN1 |
| 564 | | 5-((1-acryloylazetidin- 3-yl)amino)-[3,4′- bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C(C═CN3)═CC3═O)═CN1 |
| 565 | | 5-((1-acryloylazetidin- 3-yl)amino)-1′-methyl- [3,4′-bipyridine]- 2′,6(1H,1′H)-dione | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C(C═CN3C)═CC3═O)═CN1 |
| 566 | | 5-((1-acryloylazetidin- 3-yl)amino)-2′- methoxy-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(OC)═NC═C3)═CN1 |
| 567 | | 5′-((1-acryloylazetidin- 3-yl)amino)-4- (methylamino)-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(NC)═CC═N3)═CN1 |
| 568 | | 5′-((1-acryloylazetidin- 3-yl)amino)-4- methoxy-[2,3′- bipyridin]-6′(1′H)-one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(OC)═CC═N3)═CN1 |
| 569 | | 5′-((1-acryloylazetidin- 3-yl)amino)-4-methyl- [2,3′-bipyridin]-6′(1′H)- one | O═C1C(NC2CN(C(C═C)═O)C2)═ CC(C3═CC(C)═CC═N3)═CN1 |
| 570 | | 5-((1-acryloylpiperidin- 4- yl)(cyclopropylmethyl) amino)-[3,4′-bipyridin]- 6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(C═ C)═O)CC3)═CC(C4═CC═NC═ C4)═CN1 |
| 571 | | 5-((1-acryloylpiperidin- 4- yl)(cyclopropylmethyl) amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC2CC2)C3CCN(C(C═ C)═O)CC3)═CC(C4═CC(NC)═ NC═C4)═CN1 |
| 572 | | 5-((1-acryloylpiperidin- 4-yl)(ethyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(CC)C2CCN(C(C═ C)═O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 573 | | 5-((1-acryloylpiperidin- 4-yl)(ethyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(CC)C2CCN(C(C═C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 574 | | 5-((1-acryloylpiperidin- 4-yl)(methyl)amino)- [3,4′-bipyridin]-6(1H)- one | O═C1C(N(C)C2CCN(C(C═C)═ O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 575 | | 5-((1-acryloylpiperidin- 4-yl)(methyl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(N(C)C2CCN(C(C═C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 576 | | 5-((1-acryloylpiperidin- 4-yl)amino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(C═C)═ O)CC2)═CC(C3═CC═NC═ C3)═CN1 |
| 577 | | 5-((1-acryloylpiperidin- 4-yl)amino)-2′- (methylamino)-[3,4′- bipyridin]-6(1H)-one | O═C1C(NC2CCN(C(C═C)═ O)CC2)═CC(C3═CC(NC)═NC═ C3)═CN1 |
| 578 | | O═C1C(N(C)C2CN(C/ C═C/CNC(C)C)═O)C2)═ CC(C3═CC═NC═N3)═ CN1 | O═C1C(N(C)C2CN(C(/C═ C/CNC(C)═O)C2)═CC(C3═ CC═NC═N3)═CN1 |
Biological Activity Assays
| TABLE 3 |
| Biological Activity |
| ITK Inhibition IC50 uM | IL-2 Production Inhibition IC50 uM | |
| (+ indicates ≦ 1 μM; | (+ indicates ≦ 1 μM | |
| Example # | − indicates > 1 μM) | − indicates > 1 μM) |
| 1 | + | |
| 2 | − | |
| 3 | − | |
| 4 | + | |
| 5 | + | |
| 6 | − | |
| 7 | + | |
| 8 | + | |
| 9 | + | + |
| 10 | + | + |
| 11 | + | + |
| 12 | − | |
| 13 | + | |
| 14 | − | |
| 15 | − | |
| 16 | + | |
| 17 | − | |
| 18 | + | |
| 19 | + | |
| 20 | + | |
Other Anti-Inflammatory Assays
Claims (17)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/603,937 US9145393B2 (en) | 2014-01-24 | 2015-01-23 | Arylpyridinone ITK inhibitors for treating inflammation and cancer |
| US14/806,148 US20150328227A1 (en) | 2014-01-24 | 2015-07-22 | Arylpyridinone itk inhibitors for treating inflammation and cancer |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201461931484P | 2014-01-24 | 2014-01-24 | |
| US201462007477P | 2014-06-04 | 2014-06-04 | |
| US201462007481P | 2014-06-04 | 2014-06-04 | |
| US14/603,937 US9145393B2 (en) | 2014-01-24 | 2015-01-23 | Arylpyridinone ITK inhibitors for treating inflammation and cancer |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/806,148 Continuation US20150328227A1 (en) | 2014-01-24 | 2015-07-22 | Arylpyridinone itk inhibitors for treating inflammation and cancer |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20150210671A1 US20150210671A1 (en) | 2015-07-30 |
| US9145393B2 true US9145393B2 (en) | 2015-09-29 |
Family
ID=53678406
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/603,937 Active US9145393B2 (en) | 2014-01-24 | 2015-01-23 | Arylpyridinone ITK inhibitors for treating inflammation and cancer |
Country Status (1)
| Country | Link |
|---|---|
| US (1) | US9145393B2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9695200B2 (en) | 2015-01-23 | 2017-07-04 | Confluence Life Sciences, Inc. | Heterocyclic ITK inhibitors for treating inflammation and cancer |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019513700A (en) | 2016-03-16 | 2019-05-30 | バイエル・クロップサイエンス・アクチェンゲゼルシャフト | N- (Cyanobenzyl) -6- (cyclopropyl-carbonylamino) -4- (phenyl) -pyridine-2-carboxamide derivative as a pesticide and plant protection agent and related compounds |
| US10316038B2 (en) | 2017-01-25 | 2019-06-11 | Aclaris Therapeutics, Inc. | Pyrrolopyrimidine ITK inhibitors for treating inflammation and cancer |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000026211A1 (en) | 1998-10-30 | 2000-05-11 | Merck & Co., Inc. | Thrombin inhibitors |
| WO2002053543A1 (en) | 2000-12-28 | 2002-07-11 | Shionogi & Co., Ltd. | Pyridone derivative having affinity for cannabinoid 2-type receptor |
| WO2012031004A1 (en) | 2010-09-01 | 2012-03-08 | Gilead Connecticut, Inc. | Pyridinones/pyrazinones, method of making, and method of use thereof |
| EP2489663A1 (en) | 2011-02-16 | 2012-08-22 | Almirall, S.A. | Compounds as syk kinase inhibitors |
| US8518952B2 (en) * | 2008-08-06 | 2013-08-27 | Pfizer Inc. | 6 substituted 2-heterocyclylamino pyrazine compounds as CHK-1 inhibitors |
-
2015
- 2015-01-23 US US14/603,937 patent/US9145393B2/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2000026211A1 (en) | 1998-10-30 | 2000-05-11 | Merck & Co., Inc. | Thrombin inhibitors |
| WO2002053543A1 (en) | 2000-12-28 | 2002-07-11 | Shionogi & Co., Ltd. | Pyridone derivative having affinity for cannabinoid 2-type receptor |
| US8518952B2 (en) * | 2008-08-06 | 2013-08-27 | Pfizer Inc. | 6 substituted 2-heterocyclylamino pyrazine compounds as CHK-1 inhibitors |
| WO2012031004A1 (en) | 2010-09-01 | 2012-03-08 | Gilead Connecticut, Inc. | Pyridinones/pyrazinones, method of making, and method of use thereof |
| US20130281432A1 (en) * | 2010-09-01 | 2013-10-24 | Genentech, Inc. | Pyridinones/pyrazinones, method of making, and method of use thereof |
| EP2489663A1 (en) | 2011-02-16 | 2012-08-22 | Almirall, S.A. | Compounds as syk kinase inhibitors |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9695200B2 (en) | 2015-01-23 | 2017-07-04 | Confluence Life Sciences, Inc. | Heterocyclic ITK inhibitors for treating inflammation and cancer |
| US10150783B2 (en) | 2015-01-23 | 2018-12-11 | Aclaris Therapeutics, Inc. | Heterocyclic ITK inhibitors for treating inflammation and cancer |
Also Published As
| Publication number | Publication date |
|---|---|
| US20150210671A1 (en) | 2015-07-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10150783B2 (en) | Heterocyclic ITK inhibitors for treating inflammation and cancer | |
| US12297203B2 (en) | Pyrrolopyrimidine ITK inhibitors | |
| US12186324B2 (en) | Imidazopiperazine inhibitors of transcription activating proteins | |
| US11420966B2 (en) | Substituted pyrrolopyridines as JAK inhibitors | |
| US9499551B2 (en) | Substituted pyrrolo[2,3-b]pyridines for treating cancer or inflammatory diseases | |
| US20150328227A1 (en) | Arylpyridinone itk inhibitors for treating inflammation and cancer | |
| US11713313B2 (en) | GLS1 inhibitors for treating disease | |
| US20160002204A1 (en) | Gls1 inhibitors for treating disease | |
| US20220235043A1 (en) | Substituted sulfonamide pyrrolopyridines as jak inhibitors | |
| US9145393B2 (en) | Arylpyridinone ITK inhibitors for treating inflammation and cancer | |
| US20190352300A1 (en) | Pyrrolopyrimidine itk inhibitors for treating inflammation and cancer | |
| US20190135793A1 (en) | Heterocyclic inhibitors of kdm5 for the treatment of disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: CONFLUENCE LIFE SCIENCES, INC., MISSOURI Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:JACOBSEN, ERIC JON;BLINN, JAMES ROBERT;SIGNING DATES FROM 20150323 TO 20150402;REEL/FRAME:035340/0900 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| AS | Assignment |
Owner name: ACLARIS LIFE SCIENCES, INC., MISSOURI Free format text: MERGER;ASSIGNOR:CONFLUENCE LIFE SCIENCES, INC.;REEL/FRAME:044477/0208 Effective date: 20170803 |
|
| AS | Assignment |
Owner name: ACLARIS THERAPEUTICS, INC., PENNSYLVANIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:ACLARIS LIFE SCIENCES, INC.;REEL/FRAME:046153/0695 Effective date: 20180511 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YR, SMALL ENTITY (ORIGINAL EVENT CODE: M2551); ENTITY STATUS OF PATENT OWNER: SMALL ENTITY Year of fee payment: 4 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YR, SMALL ENTITY (ORIGINAL EVENT CODE: M2552); ENTITY STATUS OF PATENT OWNER: SMALL ENTITY Year of fee payment: 8 |