US8409537B2 - Method for removing contaminants from boron powder - Google Patents
Method for removing contaminants from boron powder Download PDFInfo
- Publication number
- US8409537B2 US8409537B2 US13/220,053 US201113220053A US8409537B2 US 8409537 B2 US8409537 B2 US 8409537B2 US 201113220053 A US201113220053 A US 201113220053A US 8409537 B2 US8409537 B2 US 8409537B2
- Authority
- US
- United States
- Prior art keywords
- boron powder
- contaminant
- contaminated
- comingled
- enclosed space
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B35/00—Boron; Compounds thereof
- C01B35/02—Boron; Borides
- C01B35/023—Boron
Definitions
- the subject matter disclosed herein relates to removing contaminants from boron powder.
- Boron powder is used as a primary component of boron coatings in numerous applications. Such applications include, but are not limited to boron coatings used for neutron detection, abrasion protection for die-casting dies, improved wear resistance for biomedical implants, etc. Some of these applications are adversely affected by contaminants within the boron powder, as the contaminants can be detrimental to boron coating applications.
- a contaminated boron powder can include organic contaminants from various sources.
- jet milled boron powder has been found to be susceptible to contamination from the air supply used in the milling process.
- boron powder contaminants may include lubrication oil from an air compressor when compressed air is used to operate a jet mill. This contamination can result in coating defects such as non-uniform coatings and gas contamination resulting in degraded coating properties.
- Other example contaminants are polymeric liner material from the jet mill, adhesive materials used to attach the polymeric liner material to a jet mill interior wall, and metal particles from the jet mill interior wall.
- Boron powder is a relatively expensive material which, in turn, makes both contaminated boron powder and coated goods costly missteps in the manufacturing process.
- Some previous methods of recovering contaminated boron powder include rinsing the powder with hexane, methylene chloride, and ethylene glycol, each in combination with filters and/or centrifuges. Therefore, there is a need for an improved apparatus and method of removing contaminants from the surfaces of boron powder particles.
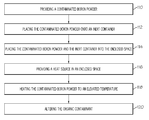
- the present invention provides a method of removing a contaminant from contaminated boron powder.
- the method includes providing a contaminated boron powder, the boron powder being comingled with an organic contaminant.
- the method also includes placing the contaminated boron powder and the contaminant comingled therewith onto an inert container.
- the method includes placing the inert container, the contaminated boron powder, and the contaminant comingled therewith, into an enclosed space.
- a heat source is provided in the enclosed space.
- the method also includes heating the contaminated boron powder and the contaminant comingled therewith to an elevated temperature.
- the method includes altering the contaminant so as to reduce the amount of the organic contaminant comingled with the boron powder.
- the present invention provides a method of removing a contaminant from contaminated boron powder.
- the method includes providing a contaminated boron powder, the boron powder being comingled with an organic contaminant.
- the method also includes placing the contaminated boron powder and the contaminant comingled therewith onto an inert container.
- the method includes placing the inert container, the contaminated boron powder, and the contaminant comingled therewith, into an enclosed space. A heat source is proved in the enclosed space.
- the method also includes heating the contaminated boron powder and the contaminant comingled therewith to an elevated temperature.
- the method includes altering the contaminant so that the amount of the organic contaminant in the boron powder after a processing cycle is not more than about 0.1 weight percent of soluble residue.
- FIG. 1 is a schematized cross section view of an example furnace of an example processing system in accordance with an aspect of the present invention
- FIG. 2 is a top level flow diagram of an example method of removing organic contaminants from boron powder in accordance with an aspect of the present invention.
- FIG. 3 is a top level flow diagram of an example method of removing organic contaminants from boron powder in accordance with an aspect of the present invention.
- Example embodiments that incorporate one or more aspects of the present invention are described and illustrated in the drawings. These illustrated examples are not intended to be a limitation on the present invention. For example, one or more aspects of the present invention can be utilized in other embodiments and even other types of devices. Moreover, certain terminology is used herein for convenience only and is not to be taken as a limitation on the present invention. Still further, in the drawings, the same reference numerals are employed for designating the same elements.
- FIG. 1 An example processing system 10 for removing contaminants from boron powder 12 is generally shown within FIG. 1 .
- the processing system 10 is for removing organic contaminants from boron powder 12 .
- organic is a broad and expansive classification.
- the classification includes materials that contain a carbon component.
- FIG. 1 merely shows one example of possible structures/configurations/etc. and that other examples are contemplated within the scope of the present invention.
- the processing system 10 for removing organic contaminants from contaminated boron powder 12 includes a furnace 16 , which is one example of an enclosed space.
- Other examples of an enclosed space include, but are not limited to batch ovens, continuous ovens, cabinet ovens, tower ovens, sintering furnaces, etc.
- an interior volume 18 of the furnace 16 can be secured so that little or no ambient atmosphere can enter into the furnace during operation of the furnace. Selection of the type of furnace 16 and construction thereof is dependent upon several variables including, but not limited to, furnace heating characteristics, furnace cycle times, boron powder throughput requirements, etc.
- the furnace 16 also includes a heat source 20 to provide the elevated temperature within the furnace 16 .
- the heat source 20 can be any of the typical furnace 16 or oven heat sources as are known in the art such as gas, electric heating element, infrared, microwave, etc.
- the heat source 20 is schematically shown and is only schematically shown in position. The structure and position can be suitably selected to heat the interior volume 18 .
- the furnace 16 can include an exhaust port that can be used to purge vaporized contaminants from the interior volume 18 .
- the interior volume 18 of the furnace 16 provides space for the contaminated boron powder 12 .
- a furnace heating cycle can begin after the boron powder 12 has been placed into the furnace 16 .
- the furnace heating cycle subjects the boron powder 12 to an elevated temperature within the furnace 16 .
- Temperature profiles for the furnace heating cycle may ramp up to a particular temperature, hold constant for a time and then ramp down. However, it is contemplated that the temperature profile may be of various multiple temperatures in order to optimize the heat application to the boron powder 12 and contaminant removal process.
- the boron powder 12 is subjected to an elevated temperature of about 500° C. This temperature promotes the vaporization of some organic contaminants, and this temperature can also promote thermal decomposition of some organic contaminants.
- the processing system 10 further includes a boat 24 , which is one example of an inert container for holding the boron powder 12 within the furnace 16 .
- the boat 24 can be made of material that is resistant to the effects of high temperature, numerous heating and cooling cycles, and is not likely to impart contaminants to the boron powder 12 that it contains. Quartz is a common choice as a boat 24 material, as it can have smooth surfaces which promote easy removal of boron powder 12 , it is typically easy to clean, and it has surface characteristics that can make any boron powder 12 remaining in the boat 24 after its intended removal readily visible to the casual observer. Several ceramic compounds are also common choices as a boat 24 material.
- the boat 24 can be shaped like a rectangular or square bowl, with a horizontal bottom and four vertical sides, although the boat may be constructed of various materials and have varied dimensions and shapes. Boats 24 can be used in batch furnaces or can be used in continuous furnaces, riding a conveyor as they pass through various heating zones.
- the processing system 10 can also include at least one port 26 for introducing at least one cover gas 30 (schematically represented by a bottle-type source example) into the furnace 16 .
- the cover gas 30 can provide a special atmosphere within the furnace 16 , consisting of one gas or a combination of gases.
- hydrogen is used as a cover gas 30 .
- Hydrogen provides a reducing atmosphere within the furnace 16 .
- One quality of the hydrogen reducing atmosphere is the promotion of the break-down of longer carbon chains into smaller, more easily volatilized organic compounds. This quality is particularly useful in eliminating some of the organic contaminants such as lubrication oil from a typical industrial compressed air supply. This lubrication oil is formulated specifically to be resistant to thermal decomposition.
- the hydrogen cover gas 30 minimizes the oxidation of the boron powder 12 .
- the combination of the hydrogen reducing atmosphere and a furnace 16 temperature of about 500° C. reduces the likelihood of oxidation of the boron powder 12 .
- Lower oxidation rates tend to eliminate boron coating defects in downstream manufacturing processes.
- Another benefit to the inclusion of a cover gas is that reactive or inert cover gases provide convection. Convection action within the interior volume 18 helps to speed the transfer of heat into the boron powder 12 and also helps to purge any vaporized compounds from the surface of the boron powder 12 .
- the processing system 10 can also be used with a cooling cycle after the thermal decomposition of the contaminants.
- a cooling cycle includes reduction of the boron powder 12 temperature to less than about 100° C. prior to removing the boron powder 12 from the interior volume 18 .
- Removal of the organic contaminants in the boron powder 12 via the break-down of longer carbon chains into smaller, more easily volatilized organic compounds and the vaporization of organic contaminants enables production of a boron powder 12 with not more than about 0.1 weight percent of soluble residue.
- This level of impurity can be considered to be an acceptable level of soluble residue that does not affect a hydrophilic nature of the boron powder 12 .
- One solvent that can be used to determine the amount of soluble residue within the boron powder 12 is methylene chloride via methods that are known in the art.
- the method of removing organic contaminants from boron powder 12 using a furnace 16 to break-down longer carbon chains and vaporize the organic contaminants and the associated process system is one solution to remove organic contaminants from a boron powder 12 .
- the use of a furnace 16 to remove the organic contaminates is a relatively simple alternative to other chemical wash methods of removing organic contaminants from boron powder 12 .
- the use of hydrogen as a cover gas 30 in a reducing atmosphere reduces the likelihood of oxidation of the boron powder 12 .
- FIG. 2 An example method of removing organic contaminants from boron powder 12 to meet purity requirements for downstream manufacturing applications is generally described in FIG. 2 .
- the method can be performed in connection with the example furnace 16 shown in FIG. 1 .
- the method includes the step 110 of providing a contaminated boron powder 12 , the boron powder being comingled with an organic contaminant.
- the organic contaminants can be introduced to the boron powder 12 during a jet milling operation from sources such as air compressor oils, adhesive materials, and particles of a polymeric liner material used on the interior of a jet mill.
- the method includes the step 112 of placing the contaminated boron powder 12 onto a boat 24 , which is one example of an inert container used in processing furnaces 16 .
- the boat 24 can be made of material that is resistant to the effects of high temperature, numerous heating and cooling cycles, and is not likely to impart contaminants to the boron powder 12 that it contains. Quartz and some ceramic compounds are common choices for boat 24 construction material.
- the method includes the step 114 of placing the contaminated boron powder 12 and the inert container into the enclosed space.
- the method includes the step 116 of providing a heat source 20 for an enclosed space.
- the heat source 20 can be any one or a combination of typical heat sources such as gas, electric heating element, infrared, microwave, etc. Examples of an enclosed space include, but are not limited to batch ovens, continuous ovens, cabinet ovens, tower ovens, sintering furnaces, etc.
- the method also includes step 118 of heating the contaminated boron powder 12 to an elevated temperature.
- the heat source 20 is activated and increases the temperature within the furnace 16 .
- the heat source 20 subjects the boron powder 12 within the enclosed space to an elevated temperature of about 500° C.
- the method includes the step 120 of altering the organic contaminant so as to reduce the amount of organic contaminant comingled with the boron powder 12 .
- the alteration of the organic contaminant is breaking down long carbon chains forming the organic contaminant.
- a hydrogen cover gas 30 within the furnace 16 provides a reducing special atmosphere that promotes this break-down of long carbon chains.
- the alteration of the organic contaminant may also be vaporizing the organic contaminant. Often, these two modes of alteration of the organic contaminant work in cooperation to eliminate organic contaminants.
- Lubrication oil can be an organic contaminant within the boron powder 12 , and it is formulated specifically to be resistant to thermal decomposition. The break-down of the carbon chains that make up the lubrication oil by the reducing atmosphere in the furnace 16 can lead to the vaporization of resultant broken-down organic contaminants that are more susceptible to thermal decomposition.
- FIG. 3 Another example method of removing organic contaminants from boron powder 12 to meet purity requirements for downstream manufacturing applications is generally described in FIG. 3 .
- the method can be performed in connection with the example furnace 16 shown in FIG. 1 .
- the method includes the step 210 of providing a contaminated boron powder 12 , the boron powder being comingled with an organic contaminant.
- the organic contaminants can be introduced to the boron powder 12 during a jet milling operation from sources such as air compressor oils, adhesive materials, and particles of a polymeric liner material used on the interior of a jet mill.
- the method includes the step 212 of placing the contaminated boron powder 12 onto a boat 24 , which is one example of an inert container used in processing furnaces 16 .
- the boat 24 can be made of material that is resistant to the effects of high temperature, numerous heating and cooling cycles, and is not likely to impart contaminants to the boron powder 12 that it contains. Quartz and some ceramic compounds are common choices for boat 24 construction material.
- the method includes the step 214 of placing the contaminated boron powder 12 and the inert container into the enclosed space.
- the method includes the step 116 of providing a heat source 20 for an enclosed space.
- the heat source 20 can be any one or a combination of typical heat sources such as gas, electric heating element, infrared, microwave, etc.
- Examples of an enclosed space include, but are not limited to batch ovens, continuous ovens, cabinet ovens, tower ovens, sintering furnaces, etc.
- the method also includes step 218 of heating the contaminated boron powder 12 to an elevated temperature.
- the heat source 20 is activated and increases the temperature within the furnace 16 .
- the heat source 20 subjects the boron powder 12 within the enclosed space to an elevated temperature of about 500° C.
- the method includes the step 220 of altering the organic contaminant so as to reduce the amount of organic contaminant comingled with the boron powder 12 so that the amount of the organic contaminant in the boron powder is not more than about 0.1 weight percent of soluble residue.
- the alteration of the organic contaminant is breaking down long carbon chains forming the organic contaminant.
- a hydrogen cover gas 30 within the furnace 16 provides a reducing special atmosphere that promotes this break-down of long carbon chains.
- the alteration of the organic contaminant may also be vaporizing the organic contaminant. Often, these two modes of alteration of the organic contaminant work in cooperation to eliminate organic contaminants.
- Lubrication oil can be an organic contaminant within the boron powder 12 , and it is formulated specifically to be resistant to thermal decomposition.
- the break-down of the carbon chains that make up the lubrication oil by the reducing atmosphere in the furnace 16 can lead to the vaporization of resultant broken-down organic contaminants that are more susceptible to thermal decomposition.
- the method can further include the step of cooling the boron powder 12 to less than about 100° C. prior to removal of the boron powder 12 from the enclosed space.
- the boron powder 12 is kept within the enclosed space for a cooling cycle within the special atmosphere.
- the special atmosphere is hydrogen gas which decreases the potential oxidation of the boron powder 12 .
- the boron powder 12 is then cooled to less than about 100° C. before it is removed from the enclosed space.
- Various cooling profiles are contemplated for the cooling cycle.
- the method and apparatus provide a means for cleaning boron powder 12 prior to making a boron powder coating solution, removing any oil films from the surface of the boron powder particles.
- the removal of organic contaminants in boron powder 12 via the break-down of longer carbon chains into smaller, more easily volatilized organic compounds and the vaporization of organic contaminants enables production of a boron powder 12 with not more than about 0.1 weight percent of soluble residue.
- This level of impurity can be considered to be an acceptable level of soluble residue that does not affect a hydrophilic nature of the boron powder 12 .
- the resulting boron powder 12 containing fewer or no organic contaminants reduces or eliminates downstream boron powder coating defects and improves the repeatability in the coating process.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Processing Of Solid Wastes (AREA)
- Carbon And Carbon Compounds (AREA)
- Treatment Of Sludge (AREA)
Abstract
Description
Claims (12)
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/220,053 US8409537B2 (en) | 2011-08-29 | 2011-08-29 | Method for removing contaminants from boron powder |
| FR1257874A FR2979337B1 (en) | 2011-08-29 | 2012-08-20 | PROCESS FOR REMOVING CONTAMINANTS FROM BORON POWDER |
| JP2012185064A JP2013100215A (en) | 2011-08-29 | 2012-08-24 | Method for removing contaminant from boron powder |
| GEAP201212846A GEP20135995B (en) | 2011-08-29 | 2012-08-24 | Method of contaminated admixture removal from boron powder |
| CN201210311575.6A CN102963899B (en) | 2011-08-29 | 2012-08-29 | The method that pollutant is removed from boron powder |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/220,053 US8409537B2 (en) | 2011-08-29 | 2011-08-29 | Method for removing contaminants from boron powder |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20130052114A1 US20130052114A1 (en) | 2013-02-28 |
| US8409537B2 true US8409537B2 (en) | 2013-04-02 |
Family
ID=47693107
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/220,053 Active US8409537B2 (en) | 2011-08-29 | 2011-08-29 | Method for removing contaminants from boron powder |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US8409537B2 (en) |
| JP (1) | JP2013100215A (en) |
| CN (1) | CN102963899B (en) |
| FR (1) | FR2979337B1 (en) |
| GE (1) | GEP20135995B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130189633A1 (en) * | 2012-01-19 | 2013-07-25 | General Electric Company | Method for removing organic contaminants from boron containing powders by high temperature processing |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2987383A (en) * | 1957-12-02 | 1961-06-06 | United States Borax Chem | Purification of elemental boron |
| US3551105A (en) * | 1966-02-01 | 1970-12-29 | Walter M Weil | Purification of elemental boron |
| US20080311018A1 (en) * | 2005-05-17 | 2008-12-18 | Anastasia Alekseeva | Materials Purification by Treatment with Hydrogen-Based Plasma |
| US7905942B1 (en) * | 2007-05-11 | 2011-03-15 | SDCmaterials, Inc. | Microwave purification process |
| US20110176983A1 (en) * | 2008-09-05 | 2011-07-21 | H.C. Starck Gmbh | Method for purifying elemental boron |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN2427511Y (en) * | 2000-03-24 | 2001-04-25 | 张茂 | Apparatus for heating and purifying ore contg. boron |
-
2011
- 2011-08-29 US US13/220,053 patent/US8409537B2/en active Active
-
2012
- 2012-08-20 FR FR1257874A patent/FR2979337B1/en active Active
- 2012-08-24 GE GEAP201212846A patent/GEP20135995B/en unknown
- 2012-08-24 JP JP2012185064A patent/JP2013100215A/en active Pending
- 2012-08-29 CN CN201210311575.6A patent/CN102963899B/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2987383A (en) * | 1957-12-02 | 1961-06-06 | United States Borax Chem | Purification of elemental boron |
| US3551105A (en) * | 1966-02-01 | 1970-12-29 | Walter M Weil | Purification of elemental boron |
| US20080311018A1 (en) * | 2005-05-17 | 2008-12-18 | Anastasia Alekseeva | Materials Purification by Treatment with Hydrogen-Based Plasma |
| US7905942B1 (en) * | 2007-05-11 | 2011-03-15 | SDCmaterials, Inc. | Microwave purification process |
| US20110176983A1 (en) * | 2008-09-05 | 2011-07-21 | H.C. Starck Gmbh | Method for purifying elemental boron |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130189633A1 (en) * | 2012-01-19 | 2013-07-25 | General Electric Company | Method for removing organic contaminants from boron containing powders by high temperature processing |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102963899A (en) | 2013-03-13 |
| FR2979337B1 (en) | 2014-03-14 |
| FR2979337A1 (en) | 2013-03-01 |
| CN102963899B (en) | 2017-08-15 |
| US20130052114A1 (en) | 2013-02-28 |
| GEP20135995B (en) | 2013-12-25 |
| JP2013100215A (en) | 2013-05-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI491768B (en) | Method of determining an amount of impurities that a contaminating material contributes to high purity silicon | |
| JP5191656B2 (en) | Film forming apparatus and film forming method | |
| EP3227047B1 (en) | Apparatus and methof for neutralization of reactive metal condensate in additive manufacturing | |
| JP2012504100A5 (en) | ||
| Nishimoto et al. | The Rate of Boron Removal from Molten Silicon by CaO-SiO 2 Slag and Cl 2 Treatment. | |
| KR20210091697A (en) | Deployable Manufacturing Center (DMC) Systems and Methods for Manufacturing Metal Parts | |
| US20160108512A1 (en) | Method of depositing tantalum to form a tantalum coating | |
| CN110923457B (en) | A kind of preparation method of high-purity arsenic crystal | |
| US20190062174A1 (en) | Process for making high-purity aluminum oxide | |
| US20130276702A1 (en) | Gas reclamation and abatement system for high volume epitaxial silicon deposition system | |
| KR20100032319A (en) | Method of refining carbon parts for production of polycrystalline silicon | |
| US8409537B2 (en) | Method for removing contaminants from boron powder | |
| US20130189633A1 (en) | Method for removing organic contaminants from boron containing powders by high temperature processing | |
| US20150166354A1 (en) | Method for removing organic contaminants from boron containing powders by high temperature processing | |
| JP2003292337A (en) | Plasma corrosion-resistant quartz glass, method for producing the same, and apparatus using the same | |
| EP2603467A1 (en) | Treatment of silica based soot or an article made of silica based soot | |
| KR20160035246A (en) | Apparatus for manufacturing high purity quartz glass | |
| EP1386890A2 (en) | Quartz glass jig and method for producing the same | |
| CN121826587A (en) | A heat treatment method for improving the grain boundary strength of alloys | |
| RU20635U1 (en) | COATING EQUIPMENT KIT | |
| CN102666375B (en) | Carbon material and manufacture method thereof | |
| Akhonin et al. | Removal of refractory inclusions from titanium in electron beam melting by the precipitation mechanism |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: GENERAL ELECTRIC COMPNAY, NEW YORK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:LUSTIG, JAMES MICHAEL;REEL/FRAME:026822/0233 Effective date: 20110826 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| CC | Certificate of correction | ||
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| AS | Assignment |
Owner name: BAKER HUGHES, A GE COMPANY, LLC, TEXAS Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:GENERAL ELECTRIC COMPANY;REEL/FRAME:051698/0464 Effective date: 20170703 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1552); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 8 |
|
| AS | Assignment |
Owner name: BAKER HUGHES HOLDINGS LLC, TEXAS Free format text: CHANGE OF NAME;ASSIGNOR:BAKER HUGHES, A GE COMPANY, LLC;REEL/FRAME:062864/0927 Effective date: 20200413 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 12TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1553); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 12 |