US8211893B2 - Protein kinase C zeta inhibition to treat diabetic retinopathy - Google Patents
Protein kinase C zeta inhibition to treat diabetic retinopathy Download PDFInfo
- Publication number
- US8211893B2 US8211893B2 US12/542,977 US54297709A US8211893B2 US 8211893 B2 US8211893 B2 US 8211893B2 US 54297709 A US54297709 A US 54297709A US 8211893 B2 US8211893 B2 US 8211893B2
- Authority
- US
- United States
- Prior art keywords
- pkc
- zeta
- inhibitor
- pkc zeta
- protein kinase
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 0 *C1=C([2*])C([1*])=C([4*])S1 Chemical compound *C1=C([2*])C([1*])=C([4*])S1 0.000 description 12
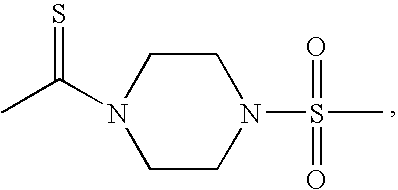
- CTMLEAKZQAPHQU-UHFFFAOYSA-N CC(=S)N1CCN(S(C)(=O)=O)CC1 Chemical compound CC(=S)N1CCN(S(C)(=O)=O)CC1 CTMLEAKZQAPHQU-UHFFFAOYSA-N 0.000 description 5
- AXXBPVSBKCLGOX-UHFFFAOYSA-N C.CC(=S)N1CCN(S(C)(=O)=O)CC1 Chemical compound C.CC(=S)N1CCN(S(C)(=O)=O)CC1 AXXBPVSBKCLGOX-UHFFFAOYSA-N 0.000 description 3
- OZGUZTBIKPOYQE-UHFFFAOYSA-N CC(C)OC(=O)C1=C(N)SC=C1C1=CC=C(Cl)C(Cl)=C1 Chemical compound CC(C)OC(=O)C1=C(N)SC=C1C1=CC=C(Cl)C(Cl)=C1 OZGUZTBIKPOYQE-UHFFFAOYSA-N 0.000 description 1
- QEYBYWYFSHMEMA-UHFFFAOYSA-N CCOC(=O)C1=C(NC(=S)N2CCN(S(C)(=O)=O)CC2)SC(C)=C1C1=CC=CC=C1 Chemical compound CCOC(=O)C1=C(NC(=S)N2CCN(S(C)(=O)=O)CC2)SC(C)=C1C1=CC=CC=C1 QEYBYWYFSHMEMA-UHFFFAOYSA-N 0.000 description 1
- IHTFNFQGKDTXIC-VXLYETTFSA-N CCOC(=O)C1=C(NC(C)=O)SC(/C(C)=N/O)=C1C1=CC=CC=C1 Chemical compound CCOC(=O)C1=C(NC(C)=O)SC(/C(C)=N/O)=C1C1=CC=CC=C1 IHTFNFQGKDTXIC-VXLYETTFSA-N 0.000 description 1
- TZDQOKNMHQUQAJ-UHFFFAOYSA-N CCOC(=O)C1=C(NC(C)=O)SC(C)=C1C1=CC=C(OC)C=C1 Chemical compound CCOC(=O)C1=C(NC(C)=O)SC(C)=C1C1=CC=C(OC)C=C1 TZDQOKNMHQUQAJ-UHFFFAOYSA-N 0.000 description 1
- RGLZNGZDAHIMBD-UHFFFAOYSA-N CN1CCN(CC2=C3/C=C\C=C/C3=CC3C=CC=CC23)CC1 Chemical compound CN1CCN(CC2=C3/C=C\C=C/C3=CC3C=CC=CC23)CC1 RGLZNGZDAHIMBD-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/99—Enzyme inactivation by chemical treatment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/38—Heterocyclic compounds having sulfur as a ring hetero atom
- A61K31/381—Heterocyclic compounds having sulfur as a ring hetero atom having five-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Definitions
- the present invention relates generally to compositions and methods for regulating one or more isoforms of protein kinase C.
- the present invention relates to compositions and methods for treating conditions associated with excessive permeability of tight junctions between cells by inhibition of protein kinase C zeta.
- PKC designates a class of kinases which play central roles in key cell signaling processes such as gene expression and regulation of cell growth.
- isoforms of PKC There are numerous isoforms of PKC and these are typically classified as: 1) “calcium-dependent” conventional isoforms which are regulated by both calcium and diacylglycerol, such as PKC-beta; 2) “calcium-independent” novel isoforms which are regulated by diacylglycerol but do not require calcium, such as PKC-delta; and 3) “atypical” isoforms which do not require calcium for activation and which are not regulated by diacylglycerol.
- PKC zeta is an “atypical” PKC isoform.
- protein kinase C beta inhibitors are currently undergoing phase 3 clinical trials for the treatment of macular edema in diabetic retinopathy.
- inhibition of PKC beta prevents only about 50% of VEGF induced endothelial permeability in primary retinal endothelial cell culture.
- compositions including a PKC inhibitor which regulates tight junction permeability and methods for regulating tight junction permeability in healthy and diseased cells and tissues.
- protein kinase C isoforms are present in a variety of cell types and have been associated with a number of pathological diseases and disorders. There is a continuing need for compositions and methods of regulating PKC activity in vitro and in vivo.
- a method of treating or preventing a disease or disorder in a subject includes administering a composition including a therapeutically effective amount of a protein kinase C (PKC) zeta inhibitor to the subject.
- PKC protein kinase C
- a disease or disorder treated or prevented by administration of a PKC zeta inhibitor is characterized by abnormal vascular permeability.
- the PKC zeta inhibitor is an inhibitor of phosphorylation of a tight junction protein, such as occludin or other proteins that control cell to cell interactions which regulate vascular permeability.
- a disease or disorder treated or prevented by administration of a composition including a therapeutically effective amount of a PKC zeta inhibitor is a neoplastic disease or disorder.
- An antineoplastic therapeutic agent is optionally included in a composition of the present invention and/or administered to a subject in need thereof separately from the PKC zeta inhibitor.
- VEGF induced abnormal vascular permeability is inhibited by administration of a PKC zeta inhibitor according to particular embodiments of the present invention.
- An administered protein kinase C zeta inhibitor is a peptide protein kinase C zeta inhibitor, a non-peptide protein kinase C zeta inhibitor; or a combination thereof.
- a method of the present invention includes administration of a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (A), described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- a method of the present invention includes administration of a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (I), described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- a method of the present invention includes administration of a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (II), described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- a method of the present invention includes administration of a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (III), described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- a method of the present invention includes administration of a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (IV), described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- a method of the present invention includes administration of a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (V), described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- Two or more protein kinase C zeta inhibitors are administered in embodiments of a method of treating or preventing a disease or disorder of the present invention.
- the two or more protein kinase C zeta inhibitors are administered together in a single composition, or separately.
- two or more protein kinase C zeta inhibitors having structural formula (A) described herein are administered in admixture in a composition or separately in embodiments of a method of treating or preventing a disease or disorder of the present invention.
- two or more protein kinase C zeta inhibitors having structural formula (I), (II), (III), (IV) or (V) described herein are administered in admixture in a composition or separately in embodiments of a method of treating or preventing a disease or disorder of the present invention.
- a therapeutically effective amount of a protein kinase C beta inhibitor is administered in embodiments of a method of treating or preventing a disease or disorder of the present invention.
- the protein kinase C beta inhibitor is administered together with a protein kinase C zeta inhibitor in a single composition, or the protein kinase C beta inhibitor and the protein kinase C zeta inhibitor are administered separately.
- compositions according to the present invention formulated for administration to a subject for the treatment or prevention of abnormal vascular permeability which include a protein kinase C zeta inhibitor and a pharmaceutically acceptable carrier.
- a protein kinase C zeta inhibitor included in a composition of the present invention is a peptide protein kinase C zeta inhibitor, a non-peptide protein kinase C zeta inhibitor; or a combination thereof.
- a composition of the present invention includes a therapeutically effective amount of the protein kinase C zeta inhibitor of formula (I), (II), (III), (IV) and/or (V) described herein, or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- R 1 and R 2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R 1 and R 2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R 1 and R 2 is an aryl group or a substituted aryl group;
- R 3 and R 4 are each independently H, a C 1 -C 3 alkyl group, a substituted C 1 -C 3 alkyl group or NHR5 where R 5 is H,
- acyl or substituted acyl where at least one of R 3 and R 4 is NHR 5 or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
- a PKC zeta inhibitor included in a composition in particular embodiments of the present invention is the PKC zeta pseudosubstrate myr-SIYRRGARRWRKL (SEQ ID No. 1).
- Two or more protein kinase C zeta inhibitors are included in an inventive composition in embodiments of the present invention.
- R 1 and R 2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R 1 and R 2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R 1 and R 2 is an aryl group or a substituted aryl group;
- R 3 and R 4 are each independently H, a C 1 -C 3 alkyl group, a substituted C 1 -C 3 alkyl group or NHR 5 where R 5 is H,
- acyl or substituted acyl where at least one of R 3 and R 4 is NHR 5 , or a pharmaceutically acceptable salt, hydrate or prodrug thereof, are included in a composition according to embodiments of the present invention.
- a therapeutically effective amount of a protein kinase C beta inhibitor is admixed with a protein kinase C zeta inhibitor in a composition.
- an antineoplastic therapeutic agent is included in a composition according to embodiments of the present invention.
- a method of inhibiting PKC zeta includes incubating PKC zeta with a PKC zeta inhibitor having the structural formula:
- R 1 and R 2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R 1 and R 2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R 1 and R 2 is an aryl group or a substituted aryl group;
- R 3 and R 4 are each independently H, a C 1 -C 3 alkyl group, a substituted C 1 -C 3 alkyl group or NHR5 where R 5 is H,
- a method of inhibiting PKC zeta includes inhibiting PKC zeta in vitro where the PKC zeta is present in an intact cell in particular embodiments of the present invention.
- a method of inhibiting PKC zeta includes inhibiting isolated PKC zeta in vitro. In particular embodiments, the PKC zeta is in a sample isolated from a subject.
- PKC zeta inhibited according to a method of the present invention is human and/or non-human PKC zeta.
- FIG. 1 is a graph showing that transfection of exogenous PKC zeta into bovine retinal endothelial cells (BREC) augments the VEGF induction of permeability to 70 kDa dextran over vector transfected control cells whereas transfection of inactive PKC zeta which has threonine 410 mutated to alanine, T410A, does not augment VEGF induced permeability;
- BREC bovine retinal endothelial cells
- FIG. 2 is a graph showing that inhibition of PKC zeta activity with the peptide inhibitor myr-SIYRRGARRWRKL reduces and prevents a VEGF stimulated increase in permeability;
- FIG. 3 is a graph showing that inhibition of PKC zeta activity with the peptide inhibitor myr-SIYRRGARRWRKL reduces VEGF stimulated endothelial permeability in a dose dependent manner;
- FIG. 4 is a graph showing that PKC-zeta I1 effectively blocks VEGF induced permeability in vitro;
- FIG. 5 is a graph showing that PKC-zeta I1 blocks VEGF induced permeability in vivo.
- a PKC zeta inhibitor inhibits phosphorylation of a synthetic or natural substrate for a PKC zeta enzyme where a statistically significant reduction of phosphorylation of a synthetic or natural substrate for PKC zeta is detected in the presence of the inhibitor and no statistically significant reduction of phosphorylation of the substrate is detected in the absence of the inhibitor.
- a peptide PKC zeta inhibitor is included in a composition of the present invention in specific embodiments.
- the myristoylated PKC zeta pseudosubstrate myr-SIYRRGARRWRKL (SEQ ID No. 1), also termed PKC ⁇ I for PKC zeta Inhibitor herein, is characterized as an inhibitor of PKC zeta activity.
- the art recognized conventional amino acid abbreviations are used to represent the above-mentioned peptide.
- a non-peptide PKC zeta inhibitor is included in a composition of the present invention in further embodiments.
- a non-peptide inhibitor is an organic molecule having a molecular weight in the range of about 200-2000 g/mol and having the structural formula
- acyl or substituted acyl where at least one of R 3 and R 4 is NHR 5 .
- a PKC zeta inhibitor has the structural formula
- R 1 and R 2 are each independently a tert-butoxycarbonyl group or ethoxycarbonyl group, a phenyl group, a dichlorophenyl group or methoxybenzene, where at least one of R 1 and R 2 is a tert-butoxycarbonyl group or ethoxycarbonyl group and at least one of R 1 and R 2 is a phenyl group, a dichlorophenyl group or methoxybenzene; where R 3 and R 4 are each independently H, methyl, or NHR 5 where R 5 is H,
- alkoxycarbonyl refers to the group C(O)OR 6 , where R 6 is C 1 -C 4 straight chain or branched alkyl or substituted straight chain or branched alkyl.
- alkoxycarbonyl groups include methoxycarbonyl, ethoxycarbonyl, tert-butoxycarbonyl, isobutoxycarbonyl, n-butoxycarbonyl, propoxycarbonyl and isopropoxycarbonyl.
- aryl refers to monocyclic or bicyclic aromatic hydrocarbon rings having from 6-12 carbon atoms in the ring or rings.
- the monocyclic or bicyclic aromatic hydrocarbon rings may be heterocyclic, having one or more heteroatoms, such as S, O, N or P atoms, in the ring or rings.
- heteroatoms such as S, O, N or P atoms
- Examples of aryl groups include phenyl, napthalenyl, piperazinyl, biphenyl and diphenyl.
- substituted aryl refers to an aryl group having a substituent at any substitutable position.
- substituted alkoxycarbonyl refers to an alkoxycarbonyl group having a substituent at any substitutable position.
- substituted C 1 -C 3 alkyl refers to a C 1 -C 3 alkyl group having a substituent at any substitutable position.
- substituted acyl refers to an acyl group having a substituent at any substitutable position.
- substituents include alkyl, substituted alkyl, hydroxy, alkylthio, alkylsulfonyl, alkylsulfinyl, alkoxy, alkoxyalkyl, alkoxycarbonyl, alkoxyarylthio, alkoxycarbonyl, alkylcarbonyloxy, aryl, aryloxy, arylalkyl, arylalkyloxy, arylsulfinyl, arylsulfinylalkyl, arylsulfonylaminocarbonyl, alkanoyl, substituted alkanoyl, alkanoylamino, alkylcarbonyl, aminocarbonylaryl, aminocarbonylalkyl, arylazo, alkoxycarbonylalkoxy, arylcarbonyl, alkylaminocarbonyl, aminoalkylcarbonyl, arylaminocarbonyl, alkylcarbonyloxy, arylcarbonyloxy,
- a compound characterized as having an inhibitory effect on the activity of PKC zeta has the structural formula (I):
- a compound characterized as having an inhibitory effect on the activity of PKC zeta has the structural formula (II):
- the compound of formula (II), 1-(anthracen-9-ylmethyl)-4-methyl-piperazine, has an IC 50 , of 25 micromolar for PKC zeta and an IC 50 of 50 micromolar for PKC-beta. This compound has an IC 50 of greater than 100 micromolar for PKC-delta.
- a compound characterized as having an inhibitory effect on the activity of PKC zeta has the structural formula (III):
- the compound of formula (III) inhibits PKC zeta with 1.2-fold greater efficacy than the compound of formula (I) when tested at 100 micromolar as described in Example 11.
- a compound characterized as having an inhibitory effect on the activity of PKC zeta has the structural formula (IV):
- a compound characterized as having an inhibitory effect on the activity of PKC zeta has the structural formula (V):
- the compound of formula (V) inhibits PKC zeta with 2.6-fold greater efficacy than the compound of formula (I) when tested at 100 micromolar as described in Example 11.
- thiophene compounds described herein such as compounds of formulas (A), (I), (II), (III), (IV) and (V), may be synthesized according to standard organic synthetic methods as described herein and known to those of skill in the art.
- R 1 is an aryl group or substituted aryl group and R 2 is H, a C 1 -C 3 alkyl group, or a substituted C 1 -C 3 alkyl group.
- the starting material (1) in Scheme 1 is commercially available or may be synthesized according to standard organic synthetic methods. In an example of this synthetic method, cyanoacetic acid ethyl ester and elemental sulfur are reacted with an alpha-methylene ketone to provide the tetra-substituted thiophene.
- Scheme 2 illustrates a synthetic scheme for a thiophene compound including synthesis of an aromatic alpha-mercaptoketone from the starting material (1) using the method described in detail in Coppola, G. M., et al., 1995, Synthesis of highly functionalized thiophenes. 4-Aryl-3-carboxylate derivatives, Synlett., 11:1143-1144.
- the aromatic alpha-mercaptoketone is reacted with vinyl phosphonate, oxidized with DDQ and reduced with hydride to form the thiophenes shown at (6) in Scheme 2.
- R 1 is amino or substituted amino and R 2 is H, a C 1 -C 3 alkyl group, or a substituted C 1 -C 3 alkyl group.
- Scheme 3 illustrates a synthetic method using a methylenethiol starting material, where R 1 is amino or substituted amino. Further details of reaction Scheme 3 are described in Damon, R. E., II, and Wareing, J. R. 1989. Preparation and formulation of 7-(butyl- or thienyl)-3,5-dihydroxy-6-heptenoate mevalonate analogs as anticholesteremics, U.S. Pat. No. 4,829,081.
- composition which includes a PKC zeta inhibitor and a pharmaceutically acceptable carrier.
- pharmaceutically acceptable carrier refers to a carrier or diluent that is generally non-toxic to an intended recipient and which does not significantly inhibit activity of the protein kinase C zeta inhibitor or other active agent included in the composition.
- a composition according to the present invention generally includes about 0.1-99% of a protein kinase C zeta inhibitor.
- a method according to the present invention includes administration of a protein kinase C zeta inhibitor and an inhibitor of a second isoform of PKC.
- Suitable inhibitors of a second isoform of PKC illustratively include bisindoylmaleimides such as bisindoylmaleimide I, an inhibitor of cPKC.
- bisindoylmaleimide I is known in the art and may be obtained by standard organic synthetic methods or obtained commercially, such as from Calbiochem Corp., La Jolla, Calif.
- An inhibitor of protein kinase C zeta and an inhibitor of PKC beta are included in a composition according to embodiments of the present invention.
- PKC beta inhibitors include ruboxistaurin and salt forms thereof such as ruboxistaurin mesylate, also known as LY333531.
- Bisindoylmaleimide I and LY379196 are further examples of PKC beta inhibitors.
- composition of the present invention optionally includes a PKC zeta inhibitor and a second therapeutic agent other than a PKC inhibitor.
- a composition according to the present invention may be formulated in various forms.
- a composition formulated for oral administration may be a solid, semi-solid or liquid formulation prepared according to methods known in the art and including any of various conventional pharmaceutical ingredients.
- Solid dosage forms for oral administration include capsules, tablets, pills, powders, and granules.
- a protein kinase C zeta inhibitor is admixed with at least one pharmaceutically acceptable carrier such as a filler or extender, as for example, starches, lactose, sucrose, glucose, mannitol, and silicic acid; a binder, as for example, carboxymethylcellulose, alignates, gelatin, polyvinylpyrrolidone, sucrose, and acacia; a humectant, as for example, glycerol; a disintegrating agent, as for example, agar-agar, calcium carbonate, plant starches such as potato or tapioca starch, alginic acid, certain complex silicates, and sodium carbonate; a solution retarder, as for example, paraffin; an absorption accelerator, as for example, quaternary ammonium compounds; a wetting agent, as for example, cetyl alcohol, gly
- compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugar as well as high molecular weight polyethyleneglycols, and the like.
- Solid dosage forms such as tablets, dragees, capsules, pills, and granules can be prepared with coatings and shells, such as enteric coatings and others well known in the art. They may contain opacifying agents, and can also be of such composition that they release the active compound or compounds in a certain part of the intestinal tract in a delayed manner. Examples of embedding compositions which can be used are polymeric substances and waxes. The active compounds can also be in micro-encapsulated form, if appropriate, with one or more of the above-mentioned excipients.
- the enteric coating is typically a polymeric material.
- Preferred enteric coating materials have the characteristics of being bioerodible, gradually hydrolyzable and/or gradually water-soluble polymers.
- the amount of coating material applied to a solid dosage generally dictates the time interval between ingestion and drug release.
- a coating is applied with to a thickness such that the entire coating does not dissolve in the gastrointestinal fluids at pH below 3 associated with stomach acids, yet dissolves above pH 3 in the small intestine environment. It is expected that any anionic polymer exhibiting a pH-dependent solubility profile is readily used as an enteric coating in the practice of the present invention to achieve delivery of the active to the lower gastrointestinal tract.
- the selection of the specific enteric coating material depends on properties such as resistance to disintegration in the stomach; impermeability to gastric fluids and active agent diffusion while in the stomach; ability to dissipate at the target intestine site; physical and chemical stability during storage; non-toxicity; and ease of application.
- Suitable enteric coating materials illustratively include cellulosic polymers such as hydroxypropyl cellulose, hydroxyethyl cellulose, hydroxypropyl methyl cellulose, methyl cellulose, ethyl cellulose, cellulose acetate, cellulose acetate phthalate, cellulose acetate trimellitate, hydroxypropylmethyl cellulose phthalate, hydroxypropylmethyl cellulose succinate and carboxymethylcellulose sodium; acrylic acid polymers and copolymers, preferably formed from acrylic acid, methacrylic acid, methyl acrylate, ammonium methylacrylate, ethyl acrylate, methyl methacrylate and/or ethyl; vinyl polymers and copolymers such as polyvinyl pyrrolidone, polyvinyl acetate, polyvinylacetate phthalate, vinylacetate crotonic acid copolymer, and ethylene-vinyl acetate copolymers; shellac; and combinations thereof.
- a particular enteric coating material is those acrylic acid polymers and copolymers available under the trade name EUDPRAGIT®, Roehm Pharma (Germany).
- EUDRAGIT® series L, L-30D S copolymers, and cross-linked polymers see for example U.S. Pat. No. 6,136,345, are suitable in particular applications since these are insoluble in the stomach and dissolve in the intestine.
- the enteric coating optionally contains a plasticizer to prevent the formation of pores and cracks that allow the penetration of the gastric fluids into the solid dosage.
- Suitable plasticizers illustratively include, triethyl citrate (Citroflex 2), triacetin (glyceryl triacetate), acetyl triethyl citrate (Citroflec A2), Carbowax 400 (polyethylene glycol 400), diethyl phthalate, tributyl citrate, acetylated monoglycerides, glycerol, fatty acid esters, propylene glycol, and dibutyl phthalate.
- a coating composed of an anionic carboxylic acrylic polymer typically contains approximately 10% to 25% by weight of a plasticizer, particularly dibutyl phthalate, polyethylene glycol, triethyl citrate and triacetin.
- the coating can also contain other coating excipients such as detackifiers, antifoaming agents, lubricants (e.g., magnesium stearate), and stabilizers (e.g., hydroxypropylcellulose, acids and bases) to solubilize or disperse the coating material, and to improve coating performance and the coated product.
- a composition formulated for oral administration can also include adjuvants, such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents.
- adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents.
- Suspensions in addition to a protein kinase C zeta inhibitor, may contain suspending agents, as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitol esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar or tragacanth, or mixtures of these substances, and/or other such conventional pharmaceutical ingredients.
- suspending agents as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitol esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar or tragacanth, or mixtures of these substances, and/or other such conventional pharmaceutical ingredients.
- Such formulations are administered by a suitable route including parenteral and oral administration.
- Administration may include systemic or local injection, such as intravenous injection.
- a composition of the present invention may also contain one or more adjuvants such as preserving, wetting, emulsifying, and dispensing agents. Prevention of the action of microorganisms can be ensured by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol and sorbic acid. It may also be desirable to include an isotonic agent, exemplified by sugars and sodium chloride. Prolonged delivery of an injectable pharmaceutical form can be achieved by the use of agents delaying absorption, for example, aluminum monostearate and gelatin.
- a composition including a PKC zeta inhibitor may be administered by a systemic route and/or by a local route.
- Suitable routes of administration illustratively include intravenous, oral, buccal, parenteral, intrathecal, intracerebroventricular, intraperitoneal, ocular, intraocular, rectal, vaginal, subcutaneous, intradermal, intramuscular, topical, intranasal, otic and mucosal.
- a composition of the present invention may be administered by intratumoral and/or peritumoral routes where applicable.
- a composition including a PKC zeta inhibitor is administered locally at or near a site of abnormal vascular permeability.
- a composition including a PKC zeta inhibitor is administered locally to one or both eyes of a subject having or at risk of having macular edema associated with diabetic retinopathy.
- Ocular administration includes intraocular periocular routes of administration.
- Ocular administration further includes administration by injection or other methods under the surface membrane of the eye to diffuse through the sclera into the retina, vitreous and anterior chamber of the eye.
- a composition including a PKC zeta inhibitor is administered intravitreally, to the subconjunctiva and/or via a sub-Tenon's capsule route.
- a method of preventing or treating a disease or disorder characterized by abnormal vascular permeability includes administering a therapeutically effective amount of a composition including a PKC zeta inhibitor to a subject in need thereof.
- a composition according to the present invention is administered to a subject having a disease or disorder or at risk for a disease or disorder characterized by abnormal vascular permeability.
- a therapeutically effective amount is intended to mean an amount of an inventive composition which is effective to alleviate, ameliorate or prevent a symptom or sign of a condition to be treated.
- a therapeutically effective amount is an amount which has a beneficial effect in a subject having macular edema associated with diabetic retinopathy, brain edema associated with a brain tumor or stroke, an abnormal cell proliferation disorder such as cancer, a tumor, a benign growth or other condition responsive to a protein kinase C zeta inhibitor of the present invention.
- treatment of a subject to prevent or treat a disease or disorder characterized by abnormal vascular permeability is characterized by prevention or amelioration of the abnormal vascular permeability.
- Abnormal vascular permeability, and change in abnormal vascular permeability is assessed by techniques known in the art and described herein.
- subject refers to any individual to whom a composition of the present invention is administered.
- subject includes mammals and birds, particularly humans, non-human primates, cats, dogs, cows, horses, rodents, pigs, sheep, goats and poultry.
- a disease or disorder to be prevented or treated using methods and compositions according to the present invention is a disease or disorder characterized by abnormal vascular permeability.
- abnormal permeability of tight junctions and “abnormal vascular permeability” as used herein are intended to indicate a physiological state characterized by passage of material across a physiological barrier which would ordinarily be prevented or reduced in a healthy individual. Abnormal permeability may be assessed, for example, by administration of a substance known to be prevented from passing across a physiological barrier including tight junctions and assay for the presence of the substance in a location from which it is generally excluded. Exemplary permeability assays for assessment of tight junctions, vessel permeability, and the effects of PKC zeta inhibitors in vitro and in vivo are described herein.
- compositions according to the present invention which include an inhibitor of PKC zeta are provided which are administered to treat vascular disorders.
- vascular disorders For example, microvascular complications are typically seen in diabetic neuropathy, diabetic nephropathy and diabetic retinopathy.
- diseases or disorders prevented or treated according to the present invention include eye conditions such as diabetic retinopathy, age-related macular degeneration and retinopathy of prematurity.
- Neoplastic diseases and disorders illustratively include adenocarcinomas, arrhenoblastomas, astrocytomas, basal cell carcinomas, bladder carcinomas, breast carcinomas, cervical carcinomas, choriocarcinoma, colorectal carcinomas, endometrial carcinoma, endometrial hyperplasia, esophageal carcinomas, fibrosarcomas, gastric carcinomas, glioblastomas, carcinomas of the head and neck, hemangionias, hemangioblastomas, hepatoblastomas, Kaposi's sarcoma, kidney carcinomas, laryngeal carcinomas, leiomyosarcomas, leukemias, liver carcinomas, lung carcinomas, lymphomas, medulloblastomas, melanomas, nasopharyngeal carcinomas, neuroblastomas, oligodendrogliomas, osteogenic sarcomas, ovarian carcinomas, pancreas carcinomas, prostate carcinoma
- the amount of a composition of the present invention administered to a subject and the route of administration depends on factors such as the identity of the particular protein lInase C zeta inhibitor included in the composition, the identity and the severity of the condition or disease affecting the subject, the rate of uptake and excretion of the inhibitor, and the general physical characteristics of the subject including age, gender and body weight.
- factors such as the identity of the particular protein lInase C zeta inhibitor included in the composition, the identity and the severity of the condition or disease affecting the subject, the rate of uptake and excretion of the inhibitor, and the general physical characteristics of the subject including age, gender and body weight.
- One of skill in the art could determine a therapeutically effective amount and route of administration in view of these and other considerations typical in medical practice.
- a therapeutically effective amount of a protein kinase C zeta inhibitor in a composition is in the range of about 0.001 mg/kg-100 mg/kg body weight.
- a therapeutically effective amount of a protein kinase C zeta inhibitor in a composition is in the range of about 0.01-10 mg/kg, and in further embodiments, a therapeutically effective amount of a protein kinase C zeta inhibitor in a composition is in the range of about 0.1-5 mg/kg.
- a therapeutically effective amount of a composition of the present invention may be manufactured and/or administered in single or multiple unit dose forms.
- a method according to the present invention includes administering a therapeutic agent in addition to administering a PKC zeta inhibitor.
- a therapeutic agent may be any of various agents suitable for use in conjunction with a particular disease or disorder.
- a therapeutic agent is an antineoplastic therapeutic agent in one embodiment of the present invention.
- Antineoplastic therapeutic agents illustratively include alkylating agents, antibiotics, folate inhibitors, purine analogs, pyrimidine analogs, and radiosensitizing compounds.
- Specific antineoplastic therapeutic agents illustratively include acivicin, aclarubicin, acodazole, acronine, adozelesin, aldesleukin, alitretinoin, allopurinol, altretamine, ambomycin, ametantrone, amifostine, aminoglutethimide, amsacrine, anastrozole, anthramycin, arsenic trioxide, asparaginase, asperlin, azacitidine, azetepa, azotomycin, batimastat, benzodepa, bicalutamide, bisantrene, bisnafide dimesylate, bizelesin, bleomycin, brequinar, bropirimine, bus
- a therapeutic agent may also be a pharmaceutically acceptable salt, ester, amide, hydrate, and/or prodrug of any of these or other therapeutic agents.
- antineoplastic therapeutic agents are described, for example, in Goodman & Gilman's The Pharmacological Basis of Therapeutics, McGraw-Hill Professional, 10th ed., 2001.
- VEGF endothelial cell tight junctions by VEGF.
- PKC zeta inhibition is found to be more effective than PKC beta inhibitors at preventing VEGF induced permeability as determined by the present invention.
- an included PKC zeta inhibitor is an inhibitor of VEGF stimulation of occludin phosphorylation.
- R 1 and R 2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R 1 and R 2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R 1 and R 2 is an aryl group or a substituted aryl group;
- R 3 aid R 4 are each independently H, a C 1 -C 3 alkyl group, a substituted C 1 -C 3 alkyl group or NHR 5 where R 5 is H,
- acyl or substituted acyl where at least one of R 3 and R 4 is NHR 5 , a salt, hydrate or prodrug thereof.
- a method of inhibiting PKC zeta includes incubating PKC zeta with a PKC zeta inhibitor having the structural formula (I), (II), (III), (IV) and/or (V), a salt, hydrate or prodrug thereof.
- Amounts of a PKC zeta inhibitor used in a method to inhibit PKC zeta and times of incubation depend on the application and will be determined by one of skill in the art without undue experimentation. For example, the IC 50 value for a particular PKC zeta inhibitor is determined.
- Methods of inhibiting PKC zeta have utility, for instance, in assays directed to elucidation of signal transduction pathways in cells in vitro and in vivo.
- a method of inhibiting PKC zeta includes incubating PKC zeta, such as PKC zeta in a cell or tissue, in vitro or in vivo, PKC zeta in an organism, and/or PKC zeta in an isolated sample, with a PKC zeta inhibitor.
- PKC zeta such as PKC zeta in a cell or tissue, in vitro or in vivo, PKC zeta in an organism, and/or PKC zeta in an isolated sample, with a PKC zeta inhibitor.
- Incubation of a cell or tissue, in vitro or in vivo, an organism, and/or an isolated sample with a PKC zeta inhibitor is optionally followed by assay of the effects of inhibition of PKC zeta in the cell, tissue, organism or sample.
- Assays for PKC zeta activity may be performed on any material suspected of containing PKC zeta, such as a sample from a subject, cultured primary cells and/or tissues or cells lines. Assays for PKC zeta activity may be performed using synthetic PKC zeta, such as PKC zeta generated recombinantly in a cell in vivo or in vitro. Thus, for example, PKC zeta activity is assessed by incubation of a sample suspected of containing PKC zeta with a PKC zeta substrate, and detection of phosphorylation of the substrate, such as by detection of incorporation of radiolabeled phosphorus in the substrate. Incubation of the sample with a PKC zeta inhibitor described herein allows for detection of specific PKC zeta activity.
- a sample from a subject may be a sample of a tissue, such as a biopsy sample, cells, a bodily fluid which may or may not include cells illustratively including blood, plasma, serum, saliva, mucous, semen, tears, an ocular exudate, a tumor exudate, ascites fluid, lymph and urine.
- a tissue such as a biopsy sample, cells, a bodily fluid which may or may not include cells illustratively including blood, plasma, serum, saliva, mucous, semen, tears, an ocular exudate, a tumor exudate, ascites fluid, lymph and urine.
- an inhibitor of PKC zeta activity is used in a method of inhibiting PKC zeta which is more effective to inhibit PKC zeta than to inhibit PKC beta or PKC delta.
- the compound of formula (I) has a half-maximal inhibitory concentration, IC 50 , of 10 micromolar for PKC-zeta, an IC 50 of greater than 100 micromolar for PKC-delta and for PKC-beta indicating that the compound of formula (I) is more effective to inhibit PKC zeta than to inhibit PKC beta or PKC delta.
- the compound of formula (II) has an IC 50 , of 25 micromolar for PKC zeta, an IC 50 of 50 micromolar for PKC-beta and an IC 50 of greater than 100 micromolar for PKC-delta, indicating that the compound of formula (II) is more effective to inhibit PKC zeta than to inhibit PKC beta or PKC delta.
- a method of inhibiting PKC zeta is applicable to inhibition of human PKC zeta as well as non-human PKC zeta.
- a method of inhibiting PKC zeta includes incubation of a PKC zeta inhibitor having the structural formula:
- R 1 and R 2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R 1 and R 2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R 1 and R 2 is an aryl group or a substituted aryl group;
- R 3 and R 4 are each independently H, a C 1 -C 3 alkyl group, a substituted C 1 -C 3 alkyl group or NHR 5 where R 5 is H,
- acyl or substituted acyl where at least one of R 3 and R 4 is NHR 5 , a salt, hydrate or prodrug thereof and/or a PKC zeta inhibitor having the structural formula (I), (II), (III), (IV) and/or (V), a salt, hydrate or prodrug thereof, with non-human PKC zeta, such as PKC zeta of non-human primates, cats, dogs, cows, horses, rodents, pigs, sheep, goats and poultry or other non-human mammal or bird.
- non-human PKC zeta such as PKC zeta of non-human primates, cats, dogs, cows, horses, rodents, pigs, sheep, goats and poultry or other non-human mammal or bird.
- inventive compositions and methods are illustrated in the following examples. These examples are provided for illustrative purposes and are not considered limitations on the scope of inventive compositions and methods.
- Retinal capillaries are isolated from bovine eyes obtained from a local slaughterhouse.
- the capillary preparation is passed through a series of meshes and collagenase treatment to remove associated cells.
- Media with D-Valine is used when first plating the cells since endothelial cells have the isomerase to convert the D amino acid to its L isoform while contaminating pericytes do not and are, therefore, selected out. All steps are conducted under sterile conditions with gloves.
- the whole eyes are bathed in a 10% povidone-iodine solution for a minimum of 5 minutes.
- a sterile scalpel With a sterile scalpel, a circumferential cut 5 mm posterior to the limbus is made to open the eyeball for retina removal.
- the retina is gently separated and cut from the anterior portion of the eyeball using sterile tweezers.
- the retinas are rinsed three times in ice-cold MEM D-Valine with HEPES buffer and pooled in the same solution.
- the retinas are washed with the same solution through a 185-micron nylon mesh stretched over a sterile porcelain funnel placed on a vacuum flask to remove retinal pigment epithelial cells.
- the retinal tissue is removed from the mesh and brought to a volume of 30 mL with ice-cold MEM D-Val with HEPES.
- the retinal aliquot is homogenized on ice six times in a Teflon/glass Potter-Elvehjem type tissue grinder with 0.25 mm clearance at 250 rpm.
- the homogenate is centrifuged at 400 ⁇ g for 10 minutes at 4° C.
- the suspension is shaken or inverted 3 to 4 times and kept on ice.
- the isolated microvessel fragments are trapped on an 88 micron nylon mesh over a funnel as above.
- the nylon mesh is then cut from the funnel and placed in a glass petri dish.
- the microvessels are separated from the mesh by repeated rinses with Ca ++ , Mg ++ free PBS and transferred to a 50 mL conical tube.
- the microvessels are then pelleted at 400 ⁇ g at 4° C. for 10 minutes.
- the pelleted microvessels are resuspended in 10 mL of enzyme cocktail and incubated at 37° C. on a rocker for 45 to 60 minutes to separate the pericytes.
- the enzyme cocktail can be stored frozen in aliquots ( ⁇ 20° C.) but only for a maximum time of 6 months. Continual rocking at 37° C. during the enzyme digestion step is necessary for complete separation of the pericytes.
- the resulting pellet is resuspended in 5 mL of the standard growth medium consisting of MEM D-Val supplemented with 20% fetal calf serum, 50 micrograms/mL ECGS, 16 U/mL heparin, 0.01 mL/mL MEM vitamins, 0.01 mL/mL glutamine, and 0.02 mL/mL antibiotic/antimycotic.
- the vessel fragments are plated on a 25 cm 2 tissue culture flask precoated with fibronectin at 2 micrograms/cm 2 and are grown in a humidified incubator at 37° C. with 95% CO 2 , 5% O 2 . Using a 0.1% solution of fibronectin from bovine plasma to coat the tissue culture surface is recommended for successful adherence of the endothelial cells. The medium is removed and fresh medium is added 24 hours following the plating.
- Colonies of endothelial cells grow from the isolated microvessels after 5-7 days. They are removed with 0.05% trypsin and reseeded onto a 75 cm 2 tissue culture flask precoated with 1 microgram/cm 2 fibronectin. Endothelial cells do not reach confluence with the primary seeding and should be split and re-seeded when islands of endothelial cells arise, prior to the proliferation of pericytes. The cells are repeatedly subcultured with 0.05% trypsin when approximately 80% confluent and expanded for experimental use at a ratio of 1:3. Subculturing the cells at 80% confluence at a ratio of 1:3 will ensure continued proliferation and homology of the cell population. Be careful not to over-trypsinize the cells when subculturing, usually 2 min of treatment with trypsin is sufficient time to release the majority of cells.
- the bovine retinal endothelial cells (BREC) are used experimentally at 6 to 10 passages after primary culture.
- Retinal cells are cultured in supplemented MCDB-131 media. A more robust and consistent cell growth occurs using this medium rather than the MEM D-Val media used in cell isolation.
- the cell culture should appear homogeneous with a cobblestone-like appearance. Contaminating pericytes are much larger than endothelial cells and can cause areas of endothelial cell death making useful transport studies impossible.
- the preparations are grown on glass coverslips and immunostained for the endothelial specific marker Von Willebrand factor.
- Transendothelial electrical resistance is a measurement of ion flux across the endothelial monolayer. This is a rapid and simple measure of barrier integrity. Alterations to TER most likely reflect changes in the junctional complex.
- the endothelial cells are grown to confluence on a porous transwell filter coated with 1 microgram/cm 2 fibronectin.
- the transwell filters are placed in an EndohmTM chamber, which contains two concentric voltage sensing electrodes, one at the top and one at the bottom.
- the EndohmTM is connected to an EVOMTM resistance meter.
- Ion flux is determined by applying a pulse of known amplitude across the endothelial monolayer and measuring the corresponding transendothelial voltage deflection.
- Ohm's law is then used to calculate resistance across the cross sectional area which is expressed in ohms ( ⁇ cm 2 .
- the investigator should be sure to subtract the resistance of a blank, fibronectin coated, transwell filter from each sample tested.
- Solute flux across endothelial monolayers is determined by placing labeled sugars or proteins on the apical side of the monolayer and determining accumulation in the basolateral chamber over time.
- the choice of solute may reflect very different features of the endothelial barrier, dextran, for example, is often used since there are no known cell receptors for dextran that may contribute a specific transport mechanism. Also, one should be aware of the size, shape (globular or linear) and hydrophobicity of the chosen solutes. A number of solutes have been used for flux rate determinations including smaller dextrans, as well as mannitol and insulin.
- albumin is of physiologic relevance as it crosses the blood-retinal barrier in various retinal pathologies and it is useful to compare its rate of transport to that of dextran of a similar molecular weight since albumin may have a transcellular and paracellular transport component.
- a standard curve to determine the concentration of solute that crossed the barrier can be generated.
- Endothelial cells are grown to confluence on transwell filters. After the cells have grown to confluence, an additional 2 to 4 days is necessary to allow formation of a tight barrier. This may vary depending on the cell preparation.
- Solute flux is measured by applying 10 micromolar or 20 micromolar RITC-dextran or FITC-BSA to the apical chamber of inserts with a confluent endothelial cell monolayer.
- the specific experiment may dictate when to perturb the system with, for example, a PKC inhibitor, relative to the time of measuring flux.
- a PKC inhibitor for example, a PKC inhibitor
- the fluorescence obtained from each time point is normalized to the fluorescence in the apical chamber.
- this ratio is corrected for the volume in the bottom chamber and this corrected ratio is plotted verses time to determine the rate of fluorescent molecule accumulation by obtaining the slope.
- this slope is converted from minutes to seconds and divided by the area of the filter yielding the diffusive flux in cm/s.
- IC 50 half-maximal inhibitory concentration
- Results indicate that the compound of formula (I) has a half-maximal inhibitory concentration, IC 50 , of 10 micromolar for PKC-zeta, an IC 50 of greater than 100 micromolar for PKC-delta and for PKC-beta indicating that the compound of formula (I) is more effective to inhibit PKC zeta than to inhibit PKC beta or PKC delta.
- Vascular permeability is quantified in this in vivo model by measuring albumin leakage form blood vessels into the retina.
- Evans blue is an acid dye of the diazo group that noncovalently binds to albumin in the blood, allowing the breakdown in blood-retinal barrier to be detected when increased vessel leakage is extravasated into the interstitial space.
- BREC are transfected with empty vector, control condition, or wild-type human PKC zeta plasmid by electroporation using the Amaxa transfection system.
- the wild-type human PKC zeta plasmid is described in detail in A Romanelli, K A Martin, A Toker, and J Blenis. Molecular and Cellular Biology, 1999, v19, p 2921-2928.
- Recombinant human VEGF165 is purchased from R&D Systems (Minneapolis, Minn.) for transfection.
- RITC-labeled 70 kDa dextran (Sigma) is used as a tracer for cell permeability measures and is applied to the apical chamber at 10 micromolar. Samples are drawn from the basolateral chamber over a 4 h time course and the rate of tracer accumulation is used to determine monolayer permeability, Po, in cm/s.
- FIG. 2 illustrates the results of the use the myristoylated peptide inhibitor of PKC zeta on endothelial permeability to 70 kDa RITC-dextran. VEGF stimulated dextran permeability as expected in the endothelial monolayer and inhibition of PKC zeta reduced or prevented permeability. Statistical analysis of these results is by ANOVA with Tukey's post-test where n ranged from 7-10 for each group.
- FIG. 6 shows that PKC-zeta I1 does not alter cell viability or increase cell death.
- Bovine retinal endothelial cells BREC are grown to confluence on transwell filters (Costar) and treated with hydrocortisone (100 nM) to induce barrier properties.
- PKC beta, wild type or mutant dominant negative S217A were transfected into cells using amaxa transfection of plasmid.
- fluorescent-labeled tracer and VEGF 50 ng/mL, R&D Systems is applied to the cells for 15 min before addition of tracer.
- RITC-labeled 70 kDa dextran (Sigma) is used as a tracer for cell permeability measures and is applied to the apical chamber at 10 micromolar. Samples are drawn from the basolateral chamber over a 4 h time course and the rate of tracer accumulation is used to determine monolayer permeability, Po, in cm/s.
- VEGF stimulated dextran permeability in the endothelial monolayer and inhibition of PKC beta partly reduces VEGF stimulated permeability.
- PKCzeta inhibitors were developed using recombinant human enzyme from Biomol International (SE-413) and CREBtide (Biomol P195) as the substrate.
- PKCzeta is incubated for 8 min at 30° C. in 50 microliters of buffer containing 5 mM MgCl 2 , 100 micromolar Na 3 VO 4 , 100 micromolar NaP 2 O 7 , 1 mM NaF.
- the substrate peptide may be chemically synthesized, recombinantly produced or obtained commercially by the trade name CREBtide.
- CREBtide Chemically synthesized, recombinantly produced or obtained commercially by the trade name CREBtide.
- Compounds are identified as having inhibitory activity when they reduce PCK zeta activity by at least 50% when tested at a concentration of 100 micromolar.
- a PKC zeta inhibitor is injected intraperitoneally twice daily at 10-50 micromoles per Kg in control or diabetic rats.
- the inhibitor is injected before induction of diabetes and for intervention studies drug is injected 1 month after induction of diabetes.
- Animals are harvested 3 months after induction of diabetes for measures of PKC zeta activity and permeability assays.
- Immunoprecipitation is carried out with 5 microliters of PKC zeta antibody for 4 h followed by 60 microliters of sepharose G beads. Beads are pelleted by brief centrifugation in a microfuge and washed 5 ⁇ with lysis buffer and twice with reaction buffer (see below). Immunoprecipitated PKC zeta is incubated for 8 min at 30° C.
- Activity of PKC zeta in cell growth in response to growth factor addition is detected using an assay including a PKC zeta inhibitor composition.
- a PKC zeta inhibitor is used in a cell growth assay in response to growth factor addition.
- cells in vivo isolated primary cells or cells of a transformed cell line are treated with a growth factor and incorporation of radiolabeled thymidine (tritiated-thymidine) is measured to assess DNA replication. DNA precipitates are quantitated for incorporation of radiolabeled thymidine into DNA using a radio-isotope counter.
- Cells are pretreated with PKC zeta inhibitor to prevent signal transduction through PKC zeta and the ability of growth factor to stimulate DNA synthesis is quantitated and compared to untreated control cells.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- General Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
where R1 and R2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R1 and R2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R1 and R2 is an aryl group or a substituted aryl group; where R3 and R4 are each independently H, a C1-C3 alkyl group, a substituted C1-C3 alkyl group or NHR5 where R5 is H,
acyl or substituted acyl, where at least one of R3 and R4 is NHR5 or a pharmaceutically acceptable salt, hydrate or prodrug thereof.
where R1 and R2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R1 and R2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R1 and R2 is an aryl group or a substituted aryl group; where R3 and R4 are each independently H, a C1-C3 alkyl group, a substituted C1-C3 alkyl group or NHR5 where R5 is H,
acyl or substituted acyl, where at least one of R3 and R4 is NHR5, or a pharmaceutically acceptable salt, hydrate or prodrug thereof, are included in a composition according to embodiments of the present invention.
where R1 and R2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R1 and R2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R1 and R2 is an aryl group or a substituted aryl group; where R3 and R4 are each independently H, a C1-C3 alkyl group, a substituted C1-C3 alkyl group or NHR5 where R5 is H,
where R1 and R2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R1 and R2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R1 and R2 is an aryl group or a substituted aryl group; where R3 and R4 are each independently H, a C1-C3 alkyl group, a substituted C1-C3 alkyl group or NHR5 where R5 is H,
where R1 and R2 are each independently a tert-butoxycarbonyl group or ethoxycarbonyl group, a phenyl group, a dichlorophenyl group or methoxybenzene, where at least one of R1 and R2 is a tert-butoxycarbonyl group or ethoxycarbonyl group and at least one of R1 and R2 is a phenyl group, a dichlorophenyl group or methoxybenzene; where R3 and R4 are each independently H, methyl, or NHR5 where R5 is H,
where R1 and R2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R1 and R2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R1 and R2 is an aryl group or a substituted aryl group; where R3 aid R4 are each independently H, a C1-C3 alkyl group, a substituted C1-C3 alkyl group or NHR5 where R5 is H,
acyl or substituted acyl, where at least one of R3 and R4 is NHR5, a salt, hydrate or prodrug thereof.
where R1 and R2 are each independently an alkoxycarbonyl group, a substituted alkoxycarbonyl group, an aryl group or a substituted aryl group, where at least one of R1 and R2 is an alkoxycarbonyl group or a substituted alkoxycarbonyl group and at least one of R1 and R2 is an aryl group or a substituted aryl group; where R3 and R4 are each independently H, a C1-C3 alkyl group, a substituted C1-C3 alkyl group or NHR5 where R5 is H,
acyl or substituted acyl, where at least one of R3 and R4 is NHR5, a salt, hydrate or prodrug thereof and/or a PKC zeta inhibitor having the structural formula (I), (II), (III), (IV) and/or (V), a salt, hydrate or prodrug thereof, with non-human PKC zeta, such as PKC zeta of non-human primates, cats, dogs, cows, horses, rodents, pigs, sheep, goats and poultry or other non-human mammal or bird.
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/542,977 US8211893B2 (en) | 2006-07-21 | 2009-08-18 | Protein kinase C zeta inhibition to treat diabetic retinopathy |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US83236206P | 2006-07-21 | 2006-07-21 | |
| US11/781,498 US7585865B2 (en) | 2006-07-21 | 2007-07-23 | Protein kinase C zeta inhibition to treat vascular permeability |
| US12/542,977 US8211893B2 (en) | 2006-07-21 | 2009-08-18 | Protein kinase C zeta inhibition to treat diabetic retinopathy |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/781,498 Division US7585865B2 (en) | 2006-07-21 | 2007-07-23 | Protein kinase C zeta inhibition to treat vascular permeability |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20090318462A1 US20090318462A1 (en) | 2009-12-24 |
| US8211893B2 true US8211893B2 (en) | 2012-07-03 |
Family
ID=38957682
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/781,498 Expired - Fee Related US7585865B2 (en) | 2006-07-21 | 2007-07-23 | Protein kinase C zeta inhibition to treat vascular permeability |
| US12/542,977 Expired - Fee Related US8211893B2 (en) | 2006-07-21 | 2009-08-18 | Protein kinase C zeta inhibition to treat diabetic retinopathy |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/781,498 Expired - Fee Related US7585865B2 (en) | 2006-07-21 | 2007-07-23 | Protein kinase C zeta inhibition to treat vascular permeability |
Country Status (2)
| Country | Link |
|---|---|
| US (2) | US7585865B2 (en) |
| WO (1) | WO2008011621A2 (en) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2513899C (en) | 2003-01-29 | 2013-03-26 | 454 Corporation | Methods of amplifying and sequencing nucleic acids |
| US20110245179A1 (en) * | 2008-09-17 | 2011-10-06 | Woo Jin Park | Composition for Prevention or Treatment of Heart Failure |
| US20120301463A1 (en) | 2009-09-30 | 2012-11-29 | President And Fellows Of Harvard College | Methods for Modulation of Autophagy Through the Modulation of Autophagy-Enhancing Gene Products |
| US8889672B2 (en) | 2011-04-29 | 2014-11-18 | The Regents Of The University Of Michigan | Compounds, formulations, and methods of protein kinase C inhibition |
| BR112013032108B1 (en) * | 2011-06-13 | 2021-10-26 | The Board Of Trustees Of The University Of Illinois | CONJUGATED PEPTIDES, USES OF THE SAME AND OF AN ISOLATED PEPTIDE, AS WELL AS PHARMACEUTICAL FORMULATION |
| US10328114B2 (en) | 2014-10-28 | 2019-06-25 | University Of Iowa Research Foundation | Lung injury repair compositions and methods |
| EP3333176A1 (en) * | 2016-12-09 | 2018-06-13 | Universite De Geneve | Peptidic protein kinase c inhibitors and uses thereof |
| US11446264B2 (en) | 2017-10-13 | 2022-09-20 | The Research Foundation For The State Univeristy Of New York | Memory manipulation via modification of protein kinase C zeta activity |
| CN112334150B (en) * | 2018-06-12 | 2025-11-18 | 日内瓦大学 | Peptide kinase C inhibitors and their uses |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4814470A (en) | 1986-07-17 | 1989-03-21 | Rhone-Poulenc Sante | Taxol derivatives, their preparation and pharmaceutical compositions containing them |
| US4829081A (en) | 1986-01-07 | 1989-05-09 | Sandoz Pharm. Corp. | Analogs of mevalonolactone and derivatives thereof |
| WO1993020101A1 (en) | 1992-04-06 | 1993-10-14 | Glaxo S.A. | Inhibitor of protein kinase c |
| WO1999016465A1 (en) | 1997-09-30 | 1999-04-08 | Beth Israel Deaconess Medical Center | Method for inhibiting tumor angiogenesis in a living subject |
| WO2004098601A1 (en) * | 2003-05-06 | 2004-11-18 | Chemon Inc. | As a pkc inhibitor, inhibitor for angiogenesis containing 6-anilinoquinoline-5,8-quinone as an effective ingredient |
-
2007
- 2007-07-23 WO PCT/US2007/074113 patent/WO2008011621A2/en not_active Ceased
- 2007-07-23 US US11/781,498 patent/US7585865B2/en not_active Expired - Fee Related
-
2009
- 2009-08-18 US US12/542,977 patent/US8211893B2/en not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4829081A (en) | 1986-01-07 | 1989-05-09 | Sandoz Pharm. Corp. | Analogs of mevalonolactone and derivatives thereof |
| US4814470A (en) | 1986-07-17 | 1989-03-21 | Rhone-Poulenc Sante | Taxol derivatives, their preparation and pharmaceutical compositions containing them |
| WO1993020101A1 (en) | 1992-04-06 | 1993-10-14 | Glaxo S.A. | Inhibitor of protein kinase c |
| WO1999016465A1 (en) | 1997-09-30 | 1999-04-08 | Beth Israel Deaconess Medical Center | Method for inhibiting tumor angiogenesis in a living subject |
| WO2004098601A1 (en) * | 2003-05-06 | 2004-11-18 | Chemon Inc. | As a pkc inhibitor, inhibitor for angiogenesis containing 6-anilinoquinoline-5,8-quinone as an effective ingredient |
Non-Patent Citations (14)
| Title |
|---|
| Antonetti, D.A., A.J. Barber, L.A. Hollinger, E.B. Wolpert, and T.W. Gardner. "Vascular Endothelial Growth Factor Induces Rapid Phosphorylation of Tight Junction Proteins Occludin and Zonula Occluden 1." The Journal of Biological Chemistry 1999, vol. 274, No. 33, pp. 23463-23467. |
| Antonetti, D.A., A.J. Barber, S. Khin, E. Lieth, J.M. Tarbell, T.W. Gardner, and the Penn State Retina Research Group. "Vascular Permeability in Experimental Diabetes Is Associated With Reduced Endothelial Occludin Content." Diabetes 1998, vol. 47, pp. 1953-1959. |
| Coppola, G.M., R.E. Damon, and H. Yu. "Synthesis of Highly Functionalized Thiophenes. 4-Aryl-3-carboxylate Derivatives." Synlett 1995, pp. 1143-1144. |
| Dempsey, E.C., A.C. Newton, D. Mochly-Rosen, A.P. Fields, M.E. Reyland, P.A. Insel, and R.O. Messing. "Protein kinase C isozymes and the regulation of diverse cell responses." American Journal of Physiology, Lung Cellular and Molecular Physiology 2000, vol. 279, pp. L429-L438. |
| Folgueira, L., J.A. McElhinny, G.D. Bren, W.S. MacMorran, M.T. Diaz-Meco, J. Moscat, and C.V. Paya. "Protein Kinase C-zeta Mediates NF-kappaB Activation in Human Immunodeficiency Virus-infected Monocytes." Journal of Virology 1996, vol. 70, No. 1, pp. 223-231. |
| Folgueira, L., J.A. McElhinny, G.D. Bren, W.S. MacMorran, M.T. Diaz-Meco, J. Moscat, and C.V. Paya. "Protein Kinase C-ζ Mediates NF-κB Activation in Human Immunodeficiency Virus-infected Monocytes." Journal of Virology 1996, vol. 70, No. 1, pp. 223-231. |
| Harhaj, N.S., E.A. Felinski, E.B. Wolpert, J.M. Sundstrom, T.W. Gardner, and D.A. Antonetti. "VEGF Activation of Protein Kinase C Stimulates Occludin Phosphorylation and Contributes to Endothelial Permeability." Investigative Ophthalmology & Visual Science 2006, vol. 47, No. 11, pp. 5106-5115. |
| Kochs, G., R. Hummerl, D. Meyer, H. Hug, D. Marme, and T.F. Sarre. "Activation and substrate specificity of the human protein kinase C alpha and zeta isoenzymes." European Journal of Biochemistry 1993, vol. 216, pp. 597-606. |
| Kochs, G., R. Hummerl, D. Meyer, H. Hug, D. Marme, and T.F. Sarre. "Activation and substrate specificity of the human protein kinase C α and ζ isoenzymes." European Journal of Biochemistry 1993, vol. 216, pp. 597-606. |
| Parker, P.J. and J. Murray-Rust. "PKC at a glance." Journal of Cell Science 2004, vol. 117, pp. 131-132. |
| Phillipson, A. et al., Protein kinase C-□ inhibition exerts cardioprotective effects in ischemia-reperfusion injury, American Journal of Physiology Heart and Circulatory Physiology, vol. 289: H898-H907, 2005. |
| Soler, A.P., R.D. Miller, K.V. Laughlin, N.Z. Carp, D.M. Klurfeld, and J.M. Mullin. "Increased tight junctional permeability is associated with the development of colon cancer." Carcinogenesis 1999, vol. 20, No. 8, pp. 1425-1431. |
| Stamatovic, S.M., O.B. Dimitrijevic, R.F. Keep, and A.V. Andjelkovic. "Protein Kinase Calpha-RhoA Cross-talk in CCL2-induced Alterations in Brain Endothelial Permeability." The Journal of Biological Chemistry 2006, vol. 281, No. 13, pp. 8379-8388. |
| Stamatovic, S.M., O.B. Dimitrijevic, R.F. Keep, and A.V. Andjelkovic. "Protein Kinase Cα-RhoA Cross-talk in CCL2-induced Alterations in Brain Endothelial Permeability." The Journal of Biological Chemistry 2006, vol. 281, No. 13, pp. 8379-8388. |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2008011621A2 (en) | 2008-01-24 |
| US20090318462A1 (en) | 2009-12-24 |
| US20080021036A1 (en) | 2008-01-24 |
| US7585865B2 (en) | 2009-09-08 |
| WO2008011621A3 (en) | 2008-07-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8211893B2 (en) | Protein kinase C zeta inhibition to treat diabetic retinopathy | |
| Oikawa et al. | Cyclic nucleotide phosphodiesterase 3A1 protects the heart against ischemia-reperfusion injury | |
| Hooper et al. | Glycogen synthase kinase‐3 inhibition is integral to long‐term potentiation | |
| Airavaara et al. | Widespread cortical expression of MANF by AAV serotype 7: localization and protection against ischemic brain injury | |
| Werner et al. | Pioglitazone activates aortic telomerase and prevents stress-induced endothelial apoptosis | |
| CN109125731B (en) | Application of Sema4D/PlexinB1 inhibitor in the preparation of drugs for the treatment and prevention of fundus vascular diseases | |
| Schlötzer-Schrehardt et al. | Selective upregulation of the A3 adenosine receptor in eyes with pseudoexfoliation syndrome and glaucoma | |
| Shi et al. | Protective effects of metformin against myocardial ischemia-reperfusion injury via AMPK-dependent suppression of NOX4 | |
| Yang et al. | Overexpression of S100A4 protects retinal ganglion cells against retinal ischemia-reperfusion injury in mice | |
| CN110151769B (en) | Application of Withaferin a in preparation of medicine for treating fundus ischemic diseases | |
| US11944625B2 (en) | Calmodulin inhibitors, Chk2 inhibitors and RSK inhibitors for the treatment of ribosomal disorders and ribosomapathies | |
| ES2958662T3 (en) | Treatment of heart disease by inhibition of the action of muscle kinase A anchoring protein (mAKAP) | |
| US8889672B2 (en) | Compounds, formulations, and methods of protein kinase C inhibition | |
| Gonzalez-Valdes et al. | Bmi1 limits dilated cardiomyopathy and heart failure by inhibiting cardiac senescence | |
| CN109771411A (en) | Use of dihydroquercetin in preparing medicine for treating fatty liver | |
| Sergeys et al. | The retinal tyrosine kinome of diabetic Akimba mice highlights potential for specific Src family kinase inhibition in retinal vascular disease | |
| MXPA05005307A (en) | Method of treatment of myocardial infarction. | |
| TW201932130A (en) | Lipocalin-type prostaglandin D2 synthase production promoter | |
| Giebel | The Metabolism of Cancer Progression: Roles of Apoptosis-Inducing Factor and the Metabolic Switch in Tumor Adaptability | |
| WO2013142721A1 (en) | Compositions and methods for preventing or treating acute kidney injury using proton pump inhibitors | |
| US20240058311A1 (en) | Use of smo inhibitor in preparation of drug for preventing, delaying or alleviating access stenosis of arteriovenous fistula | |
| CN108567772A (en) | The new application of dihydromyricetin | |
| KR101192097B1 (en) | Pharmaceutical Composition Comprising A3 Adenosine Receptor Agonist for Treating Colon Cancer | |
| Vasilevskaya et al. | Disruption of signaling through SEK1 and MKK7 yields differential responses in hypoxic colon cancer cells treated with oxaliplatin | |
| CN117797242A (en) | Antitumor pharmaceutical composition and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: NATIONAL INSTITUTES OF HEALTH (NIH), U.S. DEPT. OF Free format text: CONFIRMATORY LICENSE;ASSIGNOR:PENNSYLVANIA STATE UNIVERSITY;REEL/FRAME:023276/0077 Effective date: 20090923 |
|
| AS | Assignment |
Owner name: NATIONAL INSTITUTES OF HEALTH (NIH), U.S. DEPT. OF Free format text: CONFIRMATORY LICENSE;ASSIGNOR:THE PENNSYLVANIA STATE UNIVERSITY;REEL/FRAME:023825/0815 Effective date: 20090923 |
|
| AS | Assignment |
Owner name: THE PENN STATE RESEARCH FOUNDATION,PENNSYLVANIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ANTONETTI, DAVID A.;SUNDSTROM, JEFFREY M.;SIGNING DATES FROM 20071114 TO 20071116;REEL/FRAME:024007/0867 Owner name: THE PENN STATE RESEARCH FOUNDATION, PENNSYLVANIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ANTONETTI, DAVID A.;SUNDSTROM, JEFFREY M.;SIGNING DATES FROM 20071114 TO 20071116;REEL/FRAME:024007/0867 |
|
| CC | Certificate of correction | ||
| REMI | Maintenance fee reminder mailed | ||
| LAPS | Lapse for failure to pay maintenance fees | ||
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20160703 |