US8080540B2 - Therapeutically active triazoles and their use - Google Patents
Therapeutically active triazoles and their use Download PDFInfo
- Publication number
- US8080540B2 US8080540B2 US11/856,768 US85676807A US8080540B2 US 8080540 B2 US8080540 B2 US 8080540B2 US 85676807 A US85676807 A US 85676807A US 8080540 B2 US8080540 B2 US 8080540B2
- Authority
- US
- United States
- Prior art keywords
- halogen
- optionally substituted
- alkyl
- carbonitril
- moiety
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 150000003852 triazoles Chemical class 0.000 title description 9
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 100
- 201000010099 disease Diseases 0.000 claims abstract description 54
- 230000001419 dependent effect Effects 0.000 claims abstract description 53
- 208000035475 disorder Diseases 0.000 claims abstract description 45
- 239000003270 steroid hormone Substances 0.000 claims abstract description 21
- 229910052736 halogen Inorganic materials 0.000 claims description 300
- 150000002367 halogens Chemical class 0.000 claims description 262
- 150000001875 compounds Chemical class 0.000 claims description 251
- -1 carbonitril Chemical group 0.000 claims description 160
- 125000001424 substituent group Chemical group 0.000 claims description 101
- 125000003118 aryl group Chemical group 0.000 claims description 94
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 68
- 238000000034 method Methods 0.000 claims description 64
- 229910052757 nitrogen Inorganic materials 0.000 claims description 62
- VOXZDWNPVJITMN-ZBRFXRBCSA-N 17β-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-ZBRFXRBCSA-N 0.000 claims description 57
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 57
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 57
- 229960005309 estradiol Drugs 0.000 claims description 51
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 47
- 229910052760 oxygen Inorganic materials 0.000 claims description 47
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 42
- 125000005843 halogen group Chemical group 0.000 claims description 40
- 150000003839 salts Chemical class 0.000 claims description 39
- 125000001072 heteroaryl group Chemical group 0.000 claims description 36
- 239000000651 prodrug Substances 0.000 claims description 33
- 229940002612 prodrug Drugs 0.000 claims description 33
- 125000000217 alkyl group Chemical group 0.000 claims description 32
- 229930182833 estradiol Natural products 0.000 claims description 32
- 125000005842 heteroatom Chemical group 0.000 claims description 31
- 125000004122 cyclic group Chemical group 0.000 claims description 27
- 229910052739 hydrogen Inorganic materials 0.000 claims description 24
- 210000001519 tissue Anatomy 0.000 claims description 24
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 23
- 229910052717 sulfur Inorganic materials 0.000 claims description 23
- 125000004434 sulfur atom Chemical group 0.000 claims description 23
- 201000009273 Endometriosis Diseases 0.000 claims description 22
- 125000006239 protecting group Chemical group 0.000 claims description 22
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 18
- 239000003098 androgen Substances 0.000 claims description 17
- 239000010949 copper Substances 0.000 claims description 17
- 238000005859 coupling reaction Methods 0.000 claims description 16
- 201000010260 leiomyoma Diseases 0.000 claims description 16
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 15
- 229920006395 saturated elastomer Polymers 0.000 claims description 15
- 206010027514 Metrorrhagia Diseases 0.000 claims description 14
- 206010046798 Uterine leiomyoma Diseases 0.000 claims description 14
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims description 14
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 14
- 125000006413 ring segment Chemical group 0.000 claims description 14
- 150000003462 sulfoxides Chemical group 0.000 claims description 14
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 13
- 125000002252 acyl group Chemical group 0.000 claims description 13
- 229910052802 copper Inorganic materials 0.000 claims description 13
- 208000007106 menorrhagia Diseases 0.000 claims description 13
- 206010060862 Prostate cancer Diseases 0.000 claims description 11
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 11
- 206010006187 Breast cancer Diseases 0.000 claims description 10
- 208000026310 Breast neoplasm Diseases 0.000 claims description 10
- 241000124008 Mammalia Species 0.000 claims description 10
- 125000002619 bicyclic group Chemical group 0.000 claims description 10
- 230000008878 coupling Effects 0.000 claims description 10
- 238000010168 coupling process Methods 0.000 claims description 10
- 230000004048 modification Effects 0.000 claims description 10
- 238000012986 modification Methods 0.000 claims description 10
- 125000002950 monocyclic group Chemical group 0.000 claims description 10
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 9
- 229910052799 carbon Inorganic materials 0.000 claims description 9
- 125000000468 ketone group Chemical group 0.000 claims description 9
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 9
- 201000007954 uterine fibroid Diseases 0.000 claims description 9
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 claims description 8
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 claims description 8
- 238000004519 manufacturing process Methods 0.000 claims description 8
- 239000008194 pharmaceutical composition Substances 0.000 claims description 8
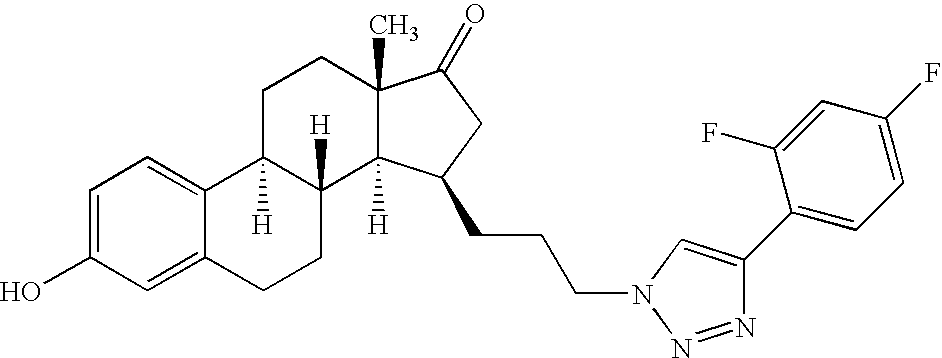
- OHSIKDYNUYDGSS-YRAHNPESSA-N (8r,9s,13s,14s,15r)-15-[2-[4-(3,5-difluorophenyl)triazol-1-yl]ethyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC(F)=CC(F)=C1 OHSIKDYNUYDGSS-YRAHNPESSA-N 0.000 claims description 7
- ZZOAWLKTWUJRJQ-QQMXCIRCSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-(2-phenylethyl)triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1CCC1=CC=CC=C1 ZZOAWLKTWUJRJQ-QQMXCIRCSA-N 0.000 claims description 7
- 206010014759 Endometrial neoplasm Diseases 0.000 claims description 7
- 229910052794 bromium Inorganic materials 0.000 claims description 7
- 229910052801 chlorine Inorganic materials 0.000 claims description 7
- 208000024827 Alzheimer disease Diseases 0.000 claims description 6
- 206010013935 Dysmenorrhoea Diseases 0.000 claims description 6
- 206010014733 Endometrial cancer Diseases 0.000 claims description 6
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 6
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 6
- 230000004064 dysfunction Effects 0.000 claims description 6
- 229910052731 fluorine Inorganic materials 0.000 claims description 6
- 125000001624 naphthyl group Chemical group 0.000 claims description 6
- 230000003647 oxidation Effects 0.000 claims description 6
- 238000007254 oxidation reaction Methods 0.000 claims description 6
- 239000000126 substance Substances 0.000 claims description 6
- 230000002485 urinary effect Effects 0.000 claims description 6
- JYNOLIYABBAYDI-OKZNNRSVSA-N (8r,9s,13s,14s,15r)-15-[3-[4-(3,5-difluorophenyl)triazol-1-yl]propyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1C1=CC(F)=CC(F)=C1 JYNOLIYABBAYDI-OKZNNRSVSA-N 0.000 claims description 5
- 125000001541 3-thienyl group Chemical group S1C([H])=C([*])C([H])=C1[H] 0.000 claims description 5
- 208000002874 Acne Vulgaris Diseases 0.000 claims description 5
- 208000005641 Adenomyosis Diseases 0.000 claims description 5
- 201000004384 Alopecia Diseases 0.000 claims description 5
- 206010004446 Benign prostatic hyperplasia Diseases 0.000 claims description 5
- 208000005171 Dysmenorrhea Diseases 0.000 claims description 5
- 206010020112 Hirsutism Diseases 0.000 claims description 5
- 208000001132 Osteoporosis Diseases 0.000 claims description 5
- 206010033128 Ovarian cancer Diseases 0.000 claims description 5
- 208000004403 Prostatic Hyperplasia Diseases 0.000 claims description 5
- 208000002495 Uterine Neoplasms Diseases 0.000 claims description 5
- 206010000496 acne Diseases 0.000 claims description 5
- 150000001337 aliphatic alkines Chemical group 0.000 claims description 5
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 5
- 201000009274 endometriosis of uterus Diseases 0.000 claims description 5
- 210000000056 organ Anatomy 0.000 claims description 5
- FFJQDQNJEPJZOQ-LECLCFPZSA-N (8r,9s,13s,14s,15r)-15-[3-[4-(2,4-difluorophenyl)triazol-1-yl]propyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1C1=CC=C(F)C=C1F FFJQDQNJEPJZOQ-LECLCFPZSA-N 0.000 claims description 4
- CPNUHPDLYUSPQP-HRFFQIKASA-N (8r,9s,13s,14s,15s)-3-hydroxy-15-[3-[4-(3-hydroxyphenyl)triazol-1-yl]propyl]-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1C1=CC=CC(O)=C1 CPNUHPDLYUSPQP-HRFFQIKASA-N 0.000 claims description 4
- HBVNDNDNWJDJSV-WSISITHOSA-N (8r,9s,13s,14s,15s)-3-hydroxy-15-[3-[4-(4-methoxyphenyl)triazol-1-yl]propyl]-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C1=CC(OC)=CC=C1C(N=N1)=CN1CCC[C@@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 HBVNDNDNWJDJSV-WSISITHOSA-N 0.000 claims description 4
- 208000008448 Congenital adrenal hyperplasia Diseases 0.000 claims description 4
- 206010036049 Polycystic ovaries Diseases 0.000 claims description 4
- 206010039792 Seborrhoea Diseases 0.000 claims description 4
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 4
- 201000002996 androgenic alopecia Diseases 0.000 claims description 4
- 125000003725 azepanyl group Chemical group 0.000 claims description 4
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 4
- 201000006828 endometrial hyperplasia Diseases 0.000 claims description 4
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 4
- 125000002757 morpholinyl group Chemical group 0.000 claims description 4
- 125000003386 piperidinyl group Chemical group 0.000 claims description 4
- 201000010065 polycystic ovary syndrome Diseases 0.000 claims description 4
- 208000006155 precocious puberty Diseases 0.000 claims description 4
- 201000007094 prostatitis Diseases 0.000 claims description 4
- 125000000719 pyrrolidinyl group Chemical group 0.000 claims description 4
- 208000008742 seborrheic dermatitis Diseases 0.000 claims description 4
- 208000011580 syndromic disease Diseases 0.000 claims description 4
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 claims description 4
- 206010046766 uterine cancer Diseases 0.000 claims description 4
- GVZNAAJAXFLSHS-OCAUHUNLSA-N (8r,9s,13s,14s,15r)-15-[2-[4-(2,4-difluorophenyl)triazol-1-yl]ethyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=C(F)C=C1F GVZNAAJAXFLSHS-OCAUHUNLSA-N 0.000 claims description 3
- USJDCEPRFMVPTE-DIMCCBHJSA-N (8r,9s,13s,14s,15r)-15-[2-[4-(2-fluorophenyl)triazol-1-yl]ethyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=CC=C1F USJDCEPRFMVPTE-DIMCCBHJSA-N 0.000 claims description 3
- GBKQITQVAKIQOY-PHDYFPKISA-N (8r,9s,13s,14s,15r)-15-[2-[4-(cyclohexylmethyl)triazol-1-yl]ethyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1CC1CCCCC1 GBKQITQVAKIQOY-PHDYFPKISA-N 0.000 claims description 3
- UTFORAKLSWATBX-QQMXCIRCSA-N (8r,9s,13s,14s,15r)-15-[3-[4-(cyclohexylmethyl)triazol-1-yl]propyl]-3-hydroxy-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1CC1CCCCC1 UTFORAKLSWATBX-QQMXCIRCSA-N 0.000 claims description 3
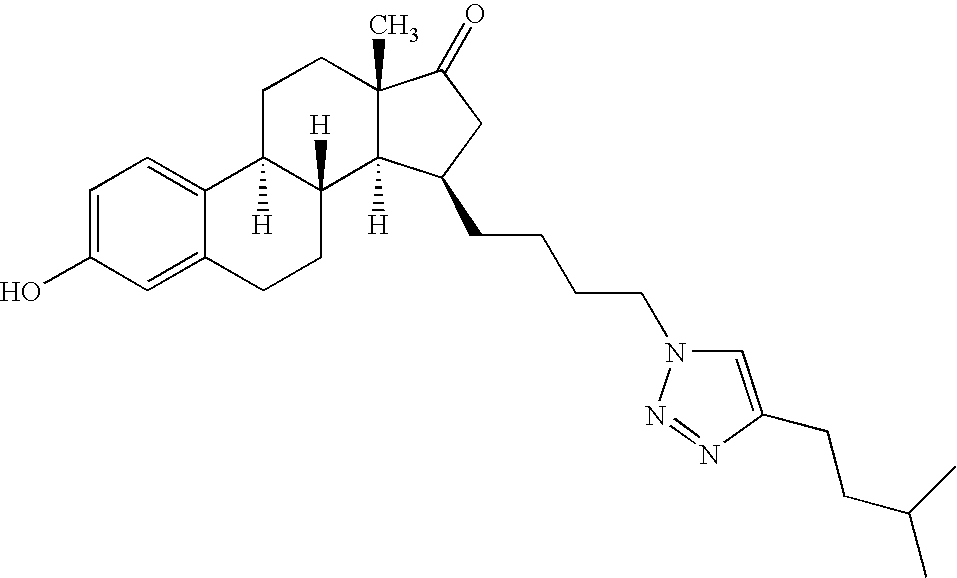
- AWSFDONEGDFCHQ-UTCLPMRJSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-(2-methylpropyl)triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound N1=NC(CC(C)C)=CN1CC[C@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 AWSFDONEGDFCHQ-UTCLPMRJSA-N 0.000 claims description 3
- XCXWFUDGOCKFAX-FNWPJALNSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-(3-methylbutyl)triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound N1=NC(CCC(C)C)=CN1CC[C@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 XCXWFUDGOCKFAX-FNWPJALNSA-N 0.000 claims description 3
- UZUNMAVXKRKWRO-DIMCCBHJSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-[2-(trifluoromethyl)phenyl]triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=CC=C1C(F)(F)F UZUNMAVXKRKWRO-DIMCCBHJSA-N 0.000 claims description 3
- LKWKLXLVUAKPFG-ZORQMFODSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-[3-(trifluoromethyl)phenyl]triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=CC(C(F)(F)F)=C1 LKWKLXLVUAKPFG-ZORQMFODSA-N 0.000 claims description 3
- LTUFEWMIDJTYDZ-ZORQMFODSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-[4-(trifluoromethoxy)phenyl]triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=C(OC(F)(F)F)C=C1 LTUFEWMIDJTYDZ-ZORQMFODSA-N 0.000 claims description 3
- LGHJTKZJGUFHMD-ZORQMFODSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[2-[4-[4-(trifluoromethyl)phenyl]triazol-1-yl]ethyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=C(C(F)(F)F)C=C1 LGHJTKZJGUFHMD-ZORQMFODSA-N 0.000 claims description 3
- SCBRJTAALJVWRS-FNWPJALNSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[3-[4-(2-methylpropyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound N1=NC(CC(C)C)=CN1CCC[C@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 SCBRJTAALJVWRS-FNWPJALNSA-N 0.000 claims description 3
- SYXRKLLECNEAED-BPEOURKJSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[3-[4-(2-phenylethyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1CCC1=CC=CC=C1 SYXRKLLECNEAED-BPEOURKJSA-N 0.000 claims description 3
- QADAISHMRZUOSR-KJNWGMDVSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[3-[4-(3-methylbutyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound N1=NC(CCC(C)C)=CN1CCC[C@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 QADAISHMRZUOSR-KJNWGMDVSA-N 0.000 claims description 3
- SUUHORZDMVONSC-UPAYHNSLSA-N (8r,9s,13s,14s,15r)-3-hydroxy-13-methyl-15-[3-[4-[4-(trifluoromethoxy)phenyl]triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1C1=CC=C(OC(F)(F)F)C=C1 SUUHORZDMVONSC-UPAYHNSLSA-N 0.000 claims description 3
- XZALQHWQVZJYEA-ZORQMFODSA-N (8r,9s,13s,14s,15r)-3-hydroxy-15-[2-[4-(3-hydroxyphenyl)triazol-1-yl]ethyl]-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CN(N=N1)C=C1C1=CC=CC(O)=C1 XZALQHWQVZJYEA-ZORQMFODSA-N 0.000 claims description 3
- MIXJSVIJNIXZKS-UPAYHNSLSA-N (8r,9s,13s,14s,15r)-3-hydroxy-15-[2-[4-(4-methoxyphenyl)triazol-1-yl]ethyl]-13-methyl-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C1=CC(OC)=CC=C1C(N=N1)=CN1CC[C@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 MIXJSVIJNIXZKS-UPAYHNSLSA-N 0.000 claims description 3
- SCBRJTAALJVWRS-OOLKYESGSA-N (8r,9s,13s,14s,15s)-3-hydroxy-13-methyl-15-[3-[4-(2-methylpropyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound N1=NC(CC(C)C)=CN1CCC[C@@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 SCBRJTAALJVWRS-OOLKYESGSA-N 0.000 claims description 3
- SYXRKLLECNEAED-OAGDWSNJSA-N (8r,9s,13s,14s,15s)-3-hydroxy-13-methyl-15-[3-[4-(2-phenylethyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C([C@@H]1[C@H]2[C@H]3[C@@H](C4=CC=C(O)C=C4CC3)CC[C@@]2(C(C1)=O)C)CCN(N=N1)C=C1CCC1=CC=CC=C1 SYXRKLLECNEAED-OAGDWSNJSA-N 0.000 claims description 3
- QADAISHMRZUOSR-SEJKDBDJSA-N (8r,9s,13s,14s,15s)-3-hydroxy-13-methyl-15-[3-[4-(3-methylbutyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound N1=NC(CCC(C)C)=CN1CCC[C@@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 QADAISHMRZUOSR-SEJKDBDJSA-N 0.000 claims description 3
- YEPLKWAZXKXXMO-RDHQROLTSA-N (8r,9s,13s,14s,15s)-3-hydroxy-13-methyl-15-[3-[4-(4-methylphenyl)triazol-1-yl]propyl]-7,8,9,11,12,14,15,16-octahydro-6h-cyclopenta[a]phenanthren-17-one Chemical compound C1=CC(C)=CC=C1C(N=N1)=CN1CCC[C@@H]1[C@H]2[C@H](CCC=3C4=CC=C(O)C=3)[C@@H]4CC[C@]2(C)C(=O)C1 YEPLKWAZXKXXMO-RDHQROLTSA-N 0.000 claims description 3
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 claims description 3
- 208000002177 Cataract Diseases 0.000 claims description 3
- 206010009900 Colitis ulcerative Diseases 0.000 claims description 3
- 206010009944 Colon cancer Diseases 0.000 claims description 3
- 208000011231 Crohn disease Diseases 0.000 claims description 3
- 206010012442 Dermatitis contact Diseases 0.000 claims description 3
- 201000004681 Psoriasis Diseases 0.000 claims description 3
- 206010039966 Senile dementia Diseases 0.000 claims description 3
- 201000006704 Ulcerative Colitis Diseases 0.000 claims description 3
- 206010047115 Vasculitis Diseases 0.000 claims description 3
- 206010052428 Wound Diseases 0.000 claims description 3
- 208000027418 Wounds and injury Diseases 0.000 claims description 3
- 208000006673 asthma Diseases 0.000 claims description 3
- 208000010877 cognitive disease Diseases 0.000 claims description 3
- 208000029742 colonic neoplasm Diseases 0.000 claims description 3
- 208000010247 contact dermatitis Diseases 0.000 claims description 3
- 206010012601 diabetes mellitus Diseases 0.000 claims description 3
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 claims description 3
- 150000004820 halides Chemical class 0.000 claims description 3
- 125000002140 imidazol-4-yl group Chemical group [H]N1C([H])=NC([*])=C1[H] 0.000 claims description 3
- 201000006417 multiple sclerosis Diseases 0.000 claims description 3
- 206010039073 rheumatoid arthritis Diseases 0.000 claims description 3
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 3
- 201000000596 systemic lupus erythematosus Diseases 0.000 claims description 3
- 125000004568 thiomorpholinyl group Chemical group 0.000 claims description 3
- 206010043778 thyroiditis Diseases 0.000 claims description 3
- 238000002054 transplantation Methods 0.000 claims description 3
- 125000004951 trihalomethoxy group Chemical group 0.000 claims description 3
- 125000004953 trihalomethyl group Chemical group 0.000 claims description 3
- 210000001635 urinary tract Anatomy 0.000 claims description 3
- 230000037303 wrinkles Effects 0.000 claims description 3
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 2
- 125000006272 (C3-C7) cycloalkyl group Chemical group 0.000 claims description 2
- 125000005037 alkyl phenyl group Chemical group 0.000 claims description 2
- 206010068168 androgenetic alopecia Diseases 0.000 claims description 2
- 125000001725 pyrenyl group Chemical group 0.000 claims description 2
- 125000005958 tetrahydrothienyl group Chemical group 0.000 claims description 2
- 208000009329 Graft vs Host Disease Diseases 0.000 claims 1
- 208000024908 graft versus host disease Diseases 0.000 claims 1
- 108010070743 3(or 17)-beta-hydroxysteroid dehydrogenase Proteins 0.000 abstract description 65
- 238000011282 treatment Methods 0.000 abstract description 37
- 102000004190 Enzymes Human genes 0.000 abstract description 28
- 108090000790 Enzymes Proteins 0.000 abstract description 28
- 230000005764 inhibitory process Effects 0.000 abstract description 22
- 230000002265 prevention Effects 0.000 abstract description 11
- 102100034067 Dehydrogenase/reductase SDR family member 11 Human genes 0.000 abstract description 7
- 238000002560 therapeutic procedure Methods 0.000 abstract description 5
- AFCJEZRLOOCYNE-PHSQCEMDSA-N (9r,10s,13r)-13-methyl-1,2,3,4,5,6,9,10,11,12-decahydrocyclopenta[a]phenanthrene;2h-triazole Chemical class C1=CNN=N1.C([C@@H]12)CCCC1CC=C1[C@@H]2CC[C@@]2(C)C1=CC=C2 AFCJEZRLOOCYNE-PHSQCEMDSA-N 0.000 abstract description 4
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 159
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 150
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 108
- 239000000203 mixture Substances 0.000 description 103
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 97
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 83
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 76
- 235000019439 ethyl acetate Nutrition 0.000 description 74
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 73
- 238000006243 chemical reaction Methods 0.000 description 61
- 239000007832 Na2SO4 Substances 0.000 description 54
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 54
- 239000012044 organic layer Substances 0.000 description 54
- 229910052938 sodium sulfate Inorganic materials 0.000 description 54
- 102100037426 17-beta-hydroxysteroid dehydrogenase type 1 Human genes 0.000 description 53
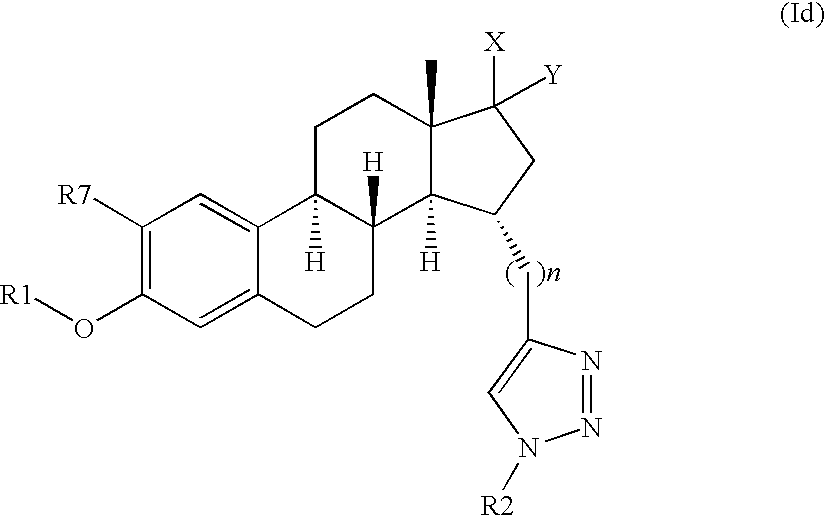
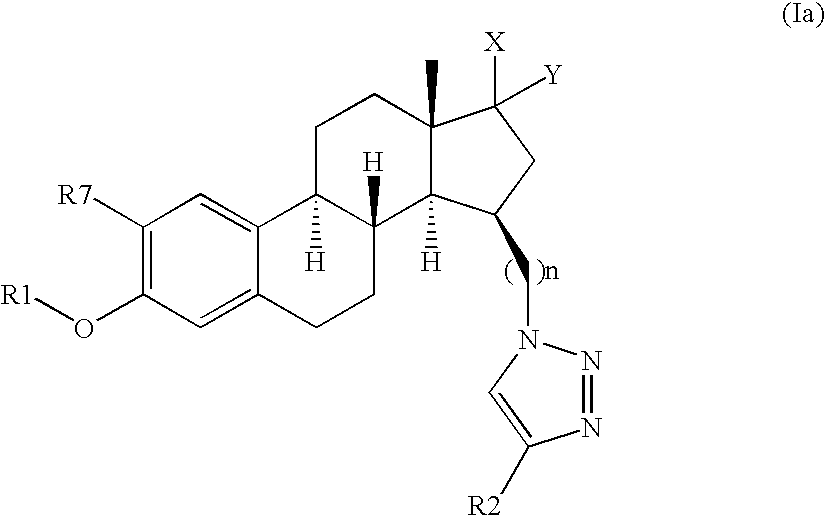
- 0 [1*]OC1=C([7*])C=C2C(=C1)CC[C@]1([H])[C@]2([H])CC[C@]2(C)C(=O)CC(C*3CB([2*])N=N3)[C@]21[H] Chemical compound [1*]OC1=C([7*])C=C2C(=C1)CC[C@]1([H])[C@]2([H])CC[C@]2(C)C(=O)CC(C*3CB([2*])N=N3)[C@]21[H] 0.000 description 52
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 47
- 230000015572 biosynthetic process Effects 0.000 description 47
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 46
- 238000003786 synthesis reaction Methods 0.000 description 45
- 239000003112 inhibitor Substances 0.000 description 44
- 239000000243 solution Substances 0.000 description 44
- 239000003921 oil Substances 0.000 description 43
- 235000019198 oils Nutrition 0.000 description 42
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 40
- DNXHEGUUPJUMQT-CBZIJGRNSA-N Estrone Chemical compound OC1=CC=C2[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1 DNXHEGUUPJUMQT-CBZIJGRNSA-N 0.000 description 37
- 239000012267 brine Substances 0.000 description 37
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 36
- 108010084625 17-beta-hydroxysteroid dehydrogenase type 3 Proteins 0.000 description 33
- 150000001540 azides Chemical class 0.000 description 33
- 238000000746 purification Methods 0.000 description 33
- 239000011541 reaction mixture Substances 0.000 description 33
- 102100022585 17-beta-hydroxysteroid dehydrogenase type 3 Human genes 0.000 description 32
- 238000004440 column chromatography Methods 0.000 description 32
- 229910052681 coesite Inorganic materials 0.000 description 30
- 229910052906 cristobalite Inorganic materials 0.000 description 30
- 239000000377 silicon dioxide Substances 0.000 description 30
- 229910052682 stishovite Inorganic materials 0.000 description 30
- 229910052905 tridymite Inorganic materials 0.000 description 30
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 29
- 239000010410 layer Substances 0.000 description 29
- 239000007787 solid Substances 0.000 description 29
- 230000000694 effects Effects 0.000 description 28
- 210000004027 cell Anatomy 0.000 description 27
- 239000002904 solvent Substances 0.000 description 27
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 26
- 239000000543 intermediate Substances 0.000 description 26
- 229960003399 estrone Drugs 0.000 description 25
- 239000003814 drug Substances 0.000 description 22
- 150000003431 steroids Chemical class 0.000 description 22
- 102100038595 Estrogen receptor Human genes 0.000 description 21
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 21
- 102100022586 17-beta-hydroxysteroid dehydrogenase type 2 Human genes 0.000 description 20
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 20
- 238000001704 evaporation Methods 0.000 description 20
- 230000008020 evaporation Effects 0.000 description 20
- KWYUFKZDYYNOTN-UHFFFAOYSA-M potassium hydroxide Inorganic materials [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 20
- 238000005160 1H NMR spectroscopy Methods 0.000 description 19
- 101710174214 Estradiol 17-beta-dehydrogenase 2 Proteins 0.000 description 19
- 239000000262 estrogen Substances 0.000 description 19
- 229940011871 estrogen Drugs 0.000 description 18
- 108010038795 estrogen receptors Proteins 0.000 description 18
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 18
- 229940079593 drug Drugs 0.000 description 17
- DNXHEGUUPJUMQT-UHFFFAOYSA-N (+)-estrone Natural products OC1=CC=C2C3CCC(C)(C(CC4)=O)C4C3CCC2=C1 DNXHEGUUPJUMQT-UHFFFAOYSA-N 0.000 description 16
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 16
- 235000019441 ethanol Nutrition 0.000 description 16
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 15
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium on carbon Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 15
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 14
- 230000002401 inhibitory effect Effects 0.000 description 14
- 238000002360 preparation method Methods 0.000 description 14
- 230000002829 reductive effect Effects 0.000 description 14
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 14
- 238000012360 testing method Methods 0.000 description 14
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 13
- 230000003637 steroidlike Effects 0.000 description 13
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 12
- 238000003556 assay Methods 0.000 description 12
- 230000001076 estrogenic effect Effects 0.000 description 12
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 12
- 239000012453 solvate Substances 0.000 description 12
- 238000004809 thin layer chromatography Methods 0.000 description 12
- 101710147298 17-beta-hydroxysteroid dehydrogenase type 1 Proteins 0.000 description 11
- 101710174215 Estradiol 17-beta-dehydrogenase 1 Proteins 0.000 description 11
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 11
- 230000027455 binding Effects 0.000 description 11
- 239000006260 foam Substances 0.000 description 11
- 150000002576 ketones Chemical class 0.000 description 11
- 239000000523 sample Substances 0.000 description 11
- PPASLZSBLFJQEF-RXSVEWSESA-M sodium-L-ascorbate Chemical compound [Na+].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RXSVEWSESA-M 0.000 description 11
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 10
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 10
- MUMGGOZAMZWBJJ-DYKIIFRCSA-N Testostosterone Chemical compound O=C1CC[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 MUMGGOZAMZWBJJ-DYKIIFRCSA-N 0.000 description 10
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 10
- 239000000706 filtrate Substances 0.000 description 10
- 230000003389 potentiating effect Effects 0.000 description 10
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 10
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 9
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 9
- 201000004458 Myoma Diseases 0.000 description 9
- 239000008280 blood Substances 0.000 description 9
- 210000004369 blood Anatomy 0.000 description 9
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 9
- 238000004128 high performance liquid chromatography Methods 0.000 description 9
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 9
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 8
- AMKGKYQBASDDJB-UHFFFAOYSA-N 9$l^{2}-borabicyclo[3.3.1]nonane Chemical compound C1CCC2CCCC1[B]2 AMKGKYQBASDDJB-UHFFFAOYSA-N 0.000 description 8
- FEJUGLKDZJDVFY-UHFFFAOYSA-N 9-borabicyclo[3.3.1]nonane Substances C1CCC2CCCC1B2 FEJUGLKDZJDVFY-UHFFFAOYSA-N 0.000 description 8
- VMQMZMRVKUZKQL-UHFFFAOYSA-N Cu+ Chemical compound [Cu+] VMQMZMRVKUZKQL-UHFFFAOYSA-N 0.000 description 8
- LFTLOKWAGJYHHR-UHFFFAOYSA-N N-methylmorpholine N-oxide Chemical compound CN1(=O)CCOCC1 LFTLOKWAGJYHHR-UHFFFAOYSA-N 0.000 description 8
- 230000001833 anti-estrogenic effect Effects 0.000 description 8
- 125000004432 carbon atom Chemical group C* 0.000 description 8
- 239000003153 chemical reaction reagent Substances 0.000 description 8
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical compound C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 description 8
- 239000003163 gonadal steroid hormone Substances 0.000 description 8
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 8
- 239000002207 metabolite Substances 0.000 description 8
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 8
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 8
- 210000004291 uterus Anatomy 0.000 description 8
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 7
- 229910021595 Copper(I) iodide Inorganic materials 0.000 description 7
- 235000019502 Orange oil Nutrition 0.000 description 7
- 229940124639 Selective inhibitor Drugs 0.000 description 7
- 230000037396 body weight Effects 0.000 description 7
- JZCCFEFSEZPSOG-UHFFFAOYSA-L copper(II) sulfate pentahydrate Chemical compound O.O.O.O.O.[Cu+2].[O-]S([O-])(=O)=O JZCCFEFSEZPSOG-UHFFFAOYSA-L 0.000 description 7
- 238000001035 drying Methods 0.000 description 7
- 238000001727 in vivo Methods 0.000 description 7
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 7
- 239000010502 orange oil Substances 0.000 description 7
- 230000008569 process Effects 0.000 description 7
- 230000002441 reversible effect Effects 0.000 description 7
- 239000000725 suspension Substances 0.000 description 7
- 208000024891 symptom Diseases 0.000 description 7
- 239000006188 syrup Substances 0.000 description 7
- 235000020357 syrup Nutrition 0.000 description 7
- BUIVJNVIIAIIFZ-MUSFGAPCSA-N (9r,10s,13r)-13-methyl-1,2,3,4,5,6,9,10,11,12-decahydrocyclopenta[a]phenanthrene Chemical group C([C@@H]12)CCCC1CC=C1[C@@H]2CC[C@@]2(C)C1=CC=C2 BUIVJNVIIAIIFZ-MUSFGAPCSA-N 0.000 description 6
- 102000004026 17-Hydroxysteroid Dehydrogenases Human genes 0.000 description 6
- 108010082514 17-Hydroxysteroid Dehydrogenases Proteins 0.000 description 6
- QWENRTYMTSOGBR-UHFFFAOYSA-N 1H-1,2,3-Triazole Chemical class C=1C=NNN=1 QWENRTYMTSOGBR-UHFFFAOYSA-N 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 6
- 206010013908 Dysfunctional uterine bleeding Diseases 0.000 description 6
- 101100189356 Mus musculus Papolb gene Proteins 0.000 description 6
- 238000005481 NMR spectroscopy Methods 0.000 description 6
- 208000002193 Pain Diseases 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 150000001345 alkine derivatives Chemical group 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- 239000000460 chlorine Substances 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 6
- 150000002159 estradiols Chemical class 0.000 description 6
- 230000006870 function Effects 0.000 description 6
- 230000035755 proliferation Effects 0.000 description 6
- 239000012279 sodium borohydride Substances 0.000 description 6
- 229910000033 sodium borohydride Inorganic materials 0.000 description 6
- 239000003643 water by type Substances 0.000 description 6
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 5
- 238000004293 19F NMR spectroscopy Methods 0.000 description 5
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 5
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 5
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 5
- XIGIYGCEZCSLDL-UHFFFAOYSA-M [Cl-].C[Si](C)(C)C#CCCC[Mg+] Chemical compound [Cl-].C[Si](C)(C)C#CCCC[Mg+] XIGIYGCEZCSLDL-UHFFFAOYSA-M 0.000 description 5
- 230000009471 action Effects 0.000 description 5
- 239000013543 active substance Substances 0.000 description 5
- 230000033228 biological regulation Effects 0.000 description 5
- 239000003054 catalyst Substances 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 239000012230 colorless oil Substances 0.000 description 5
- 238000001816 cooling Methods 0.000 description 5
- 238000011161 development Methods 0.000 description 5
- 230000018109 developmental process Effects 0.000 description 5
- 239000000328 estrogen antagonist Substances 0.000 description 5
- 238000001914 filtration Methods 0.000 description 5
- 238000009472 formulation Methods 0.000 description 5
- 239000007789 gas Substances 0.000 description 5
- 238000010438 heat treatment Methods 0.000 description 5
- 229940088597 hormone Drugs 0.000 description 5
- 239000005556 hormone Substances 0.000 description 5
- 239000007943 implant Substances 0.000 description 5
- 229910052740 iodine Inorganic materials 0.000 description 5
- 230000002427 irreversible effect Effects 0.000 description 5
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 5
- CSNNHWWHGAXBCP-UHFFFAOYSA-L magnesium sulphate Substances [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 230000005906 menstruation Effects 0.000 description 5
- 230000007170 pathology Effects 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- PPASLZSBLFJQEF-RKJRWTFHSA-M sodium ascorbate Substances [Na+].OC[C@@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RKJRWTFHSA-M 0.000 description 5
- 235000010378 sodium ascorbate Nutrition 0.000 description 5
- 229960005055 sodium ascorbate Drugs 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- 229960003604 testosterone Drugs 0.000 description 5
- GKASDNZWUGIAMG-UHFFFAOYSA-N triethyl orthoformate Chemical compound CCOC(OCC)OCC GKASDNZWUGIAMG-UHFFFAOYSA-N 0.000 description 5
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 4
- 150000000307 17β-estradiols Chemical class 0.000 description 4
- CWSSIUJITPYGLK-UHFFFAOYSA-N 2-(6-bromohexoxy)oxane Chemical compound BrCCCCCCOC1CCCCO1 CWSSIUJITPYGLK-UHFFFAOYSA-N 0.000 description 4
- 125000000590 4-methylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 4
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 4
- 108010007005 Estrogen Receptor alpha Proteins 0.000 description 4
- 108010041356 Estrogen Receptor beta Proteins 0.000 description 4
- 206010028980 Neoplasm Diseases 0.000 description 4
- RJKFOVLPORLFTN-LEKSSAKUSA-N Progesterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](C(=O)C)[C@@]1(C)CC2 RJKFOVLPORLFTN-LEKSSAKUSA-N 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- XJLXINKUBYWONI-DQQFMEOOSA-N [[(2r,3r,4r,5r)-5-(6-aminopurin-9-yl)-3-hydroxy-4-phosphonooxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [(2s,3r,4s,5s)-5-(3-carbamoylpyridin-1-ium-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl phosphate Chemical compound NC(=O)C1=CC=C[N+]([C@@H]2[C@H]([C@@H](O)[C@H](COP([O-])(=O)OP(O)(=O)OC[C@@H]3[C@H]([C@@H](OP(O)(O)=O)[C@@H](O3)N3C4=NC=NC(N)=C4N=C3)O)O2)O)=C1 XJLXINKUBYWONI-DQQFMEOOSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 230000003042 antagnostic effect Effects 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- IVRMZWNICZWHMI-UHFFFAOYSA-N azide group Chemical group [N-]=[N+]=[N-] IVRMZWNICZWHMI-UHFFFAOYSA-N 0.000 description 4
- UGUUDTWORXNLAK-UHFFFAOYSA-N azidoalcohol Chemical compound ON=[N+]=[N-] UGUUDTWORXNLAK-UHFFFAOYSA-N 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- ARUVKPQLZAKDPS-UHFFFAOYSA-L copper(II) sulfate Chemical compound [Cu+2].[O-][S+2]([O-])([O-])[O-] ARUVKPQLZAKDPS-UHFFFAOYSA-L 0.000 description 4
- 238000010511 deprotection reaction Methods 0.000 description 4
- CSJLBAMHHLJAAS-UHFFFAOYSA-N diethylaminosulfur trifluoride Chemical compound CCN(CC)S(F)(F)F CSJLBAMHHLJAAS-UHFFFAOYSA-N 0.000 description 4
- 239000003480 eluent Substances 0.000 description 4
- 239000000839 emulsion Substances 0.000 description 4
- 210000004696 endometrium Anatomy 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- 125000000524 functional group Chemical group 0.000 description 4
- 208000000509 infertility Diseases 0.000 description 4
- 230000036512 infertility Effects 0.000 description 4
- 231100000535 infertility Toxicity 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 239000003446 ligand Substances 0.000 description 4
- 230000000670 limiting effect Effects 0.000 description 4
- 239000011777 magnesium Substances 0.000 description 4
- 150000007522 mineralic acids Chemical class 0.000 description 4
- 150000007524 organic acids Chemical class 0.000 description 4
- 230000001575 pathological effect Effects 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 238000003908 quality control method Methods 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 230000001850 reproductive effect Effects 0.000 description 4
- HRZFUMHJMZEROT-UHFFFAOYSA-L sodium disulfite Chemical compound [Na+].[Na+].[O-]S(=O)S([O-])(=O)=O HRZFUMHJMZEROT-UHFFFAOYSA-L 0.000 description 4
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 3
- 102100024089 Aldo-keto reductase family 1 member C2 Human genes 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 102100029951 Estrogen receptor beta Human genes 0.000 description 3
- 238000003747 Grignard reaction Methods 0.000 description 3
- 239000007818 Grignard reagent Substances 0.000 description 3
- 208000032843 Hemorrhage Diseases 0.000 description 3
- 101000806242 Homo sapiens 17-beta-hydroxysteroid dehydrogenase type 1 Proteins 0.000 description 3
- 108060001084 Luciferase Proteins 0.000 description 3
- 239000005089 Luciferase Substances 0.000 description 3
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 3
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 3
- 229910004879 Na2S2O5 Inorganic materials 0.000 description 3
- 108010065942 Prostaglandin-F synthase Proteins 0.000 description 3
- 241000700159 Rattus Species 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 3
- AHMMSNQYOPMLSX-CNQKSJKFSA-N [(8r,9s,10r,13s,14s,17s)-10,13-dimethyl-3-oxo-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthren-17-yl] undec-10-enoate Chemical compound O=C1C=C[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)OC(=O)CCCCCCCCC=C)[C@@H]4[C@@H]3CCC2=C1 AHMMSNQYOPMLSX-CNQKSJKFSA-N 0.000 description 3
- 230000004913 activation Effects 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 235000011114 ammonium hydroxide Nutrition 0.000 description 3
- 108010080146 androgen receptors Proteins 0.000 description 3
- 230000001548 androgenic effect Effects 0.000 description 3
- 229940030486 androgens Drugs 0.000 description 3
- 229940046836 anti-estrogen Drugs 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- 208000034158 bleeding Diseases 0.000 description 3
- 230000000740 bleeding effect Effects 0.000 description 3
- 230000000903 blocking effect Effects 0.000 description 3
- 150000001721 carbon Chemical group 0.000 description 3
- 239000003433 contraceptive agent Substances 0.000 description 3
- 229910000366 copper(II) sulfate Inorganic materials 0.000 description 3
- RSIHSRDYCUFFLA-UHFFFAOYSA-N dehydrotestosterone Natural products O=C1C=CC2(C)C3CCC(C)(C(CC4)O)C4C3CCC2=C1 RSIHSRDYCUFFLA-UHFFFAOYSA-N 0.000 description 3
- 230000004069 differentiation Effects 0.000 description 3
- 239000003085 diluting agent Substances 0.000 description 3
- 208000037765 diseases and disorders Diseases 0.000 description 3
- 230000002357 endometrial effect Effects 0.000 description 3
- 239000003623 enhancer Substances 0.000 description 3
- 150000002167 estrones Chemical class 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 238000000105 evaporative light scattering detection Methods 0.000 description 3
- 235000021474 generally recognized As safe (food) Nutrition 0.000 description 3
- 235000021473 generally recognized as safe (food ingredients) Nutrition 0.000 description 3
- 150000004795 grignard reagents Chemical class 0.000 description 3
- 125000000623 heterocyclic group Chemical group 0.000 description 3
- 239000001257 hydrogen Chemical group 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 150000007529 inorganic bases Chemical class 0.000 description 3
- 230000003834 intracellular effect Effects 0.000 description 3
- 230000003902 lesion Effects 0.000 description 3
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 3
- 230000003211 malignant effect Effects 0.000 description 3
- 230000002503 metabolic effect Effects 0.000 description 3
- 210000000754 myometrium Anatomy 0.000 description 3
- 239000013642 negative control Substances 0.000 description 3
- 231100000252 nontoxic Toxicity 0.000 description 3
- 230000003000 nontoxic effect Effects 0.000 description 3
- 230000001590 oxidative effect Effects 0.000 description 3
- 238000007911 parenteral administration Methods 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 239000013612 plasmid Substances 0.000 description 3
- 238000011321 prophylaxis Methods 0.000 description 3
- 125000004076 pyridyl group Chemical group 0.000 description 3
- 238000001525 receptor binding assay Methods 0.000 description 3
- 108020003175 receptors Proteins 0.000 description 3
- 238000004007 reversed phase HPLC Methods 0.000 description 3
- 238000012216 screening Methods 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 230000021595 spermatogenesis Effects 0.000 description 3
- 238000005507 spraying Methods 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 230000009885 systemic effect Effects 0.000 description 3
- 210000001550 testis Anatomy 0.000 description 3
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- 230000000699 topical effect Effects 0.000 description 3
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 3
- FRDZGSBXKJXGNR-HTQZYQBOSA-N (1r,2r)-2-n,2-n-dimethylcyclohexane-1,2-diamine Chemical compound CN(C)[C@@H]1CCCC[C@H]1N FRDZGSBXKJXGNR-HTQZYQBOSA-N 0.000 description 2
- ITPOYLVPPDFCEF-MCIDVOSCSA-N (8R,9S,13S,14R)-3,17-dihydroxy-13-methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthrene-15-carboxylic acid Chemical class OC1=CC=C2[C@H]3CC[C@](C)(C(CC4C(O)=O)O)[C@@H]4[C@@H]3CCC2=C1 ITPOYLVPPDFCEF-MCIDVOSCSA-N 0.000 description 2
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 description 2
- SPEUIVXLLWOEMJ-UHFFFAOYSA-N 1,1-dimethoxyethane Chemical compound COC(C)OC SPEUIVXLLWOEMJ-UHFFFAOYSA-N 0.000 description 2
- YMCOFIHJJULAOJ-UHFFFAOYSA-N 1-azido-2,4-difluorobenzene Chemical compound FC1=CC=C(N=[N+]=[N-])C(F)=C1 YMCOFIHJJULAOJ-UHFFFAOYSA-N 0.000 description 2
- XEZNGIUYQVAUSS-UHFFFAOYSA-N 18-crown-6 Chemical compound C1COCCOCCOCCOCCOCCO1 XEZNGIUYQVAUSS-UHFFFAOYSA-N 0.000 description 2
- CEPCPXLLFXPZGW-UHFFFAOYSA-N 2,4-difluoroaniline Chemical compound NC1=CC=C(F)C=C1F CEPCPXLLFXPZGW-UHFFFAOYSA-N 0.000 description 2
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 description 2
- APOYTRAZFJURPB-UHFFFAOYSA-N 2-methoxy-n-(2-methoxyethyl)-n-(trifluoro-$l^{4}-sulfanyl)ethanamine Chemical compound COCCN(S(F)(F)F)CCOC APOYTRAZFJURPB-UHFFFAOYSA-N 0.000 description 2
- JVVRCYWZTJLJSG-UHFFFAOYSA-N 4-dimethylaminophenol Chemical compound CN(C)C1=CC=C(O)C=C1 JVVRCYWZTJLJSG-UHFFFAOYSA-N 0.000 description 2
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 2
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-dimethylaminopyridine Substances CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 2
- 208000035116 46 XY Disorder of Sex Development Diseases 0.000 description 2
- UPVHIBLXQNNITH-UHFFFAOYSA-N 5-chloropent-1-ynyl(trimethyl)silane Chemical compound C[Si](C)(C)C#CCCCCl UPVHIBLXQNNITH-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical class [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 2
- 102100032187 Androgen receptor Human genes 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- VQKFNUFAXTZWDK-UHFFFAOYSA-N CC1=CC=CO1 Chemical compound CC1=CC=CO1 VQKFNUFAXTZWDK-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- 201000004624 Dermatitis Diseases 0.000 description 2
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 2
- 229940122880 Estrogen receptor agonist Drugs 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical compound OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 2
- NMJREATYWWNIKX-UHFFFAOYSA-N GnRH Chemical compound C1CCC(C(=O)NCC(N)=O)N1C(=O)C(CC(C)C)NC(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)CNC(=O)C(NC(=O)C(CO)NC(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)C(CC=1NC=NC=1)NC(=O)C1NC(=O)CC1)CC1=CC=C(O)C=C1 NMJREATYWWNIKX-UHFFFAOYSA-N 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 101001045223 Homo sapiens 17-beta-hydroxysteroid dehydrogenase type 2 Proteins 0.000 description 2
- 108010044467 Isoenzymes Proteins 0.000 description 2
- 239000007836 KH2PO4 Substances 0.000 description 2
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Malonic acid Chemical compound OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- BAWFJGJZGIEFAR-NNYOXOHSSA-N NAD zwitterion Chemical compound NC(=O)C1=CC=C[N+]([C@H]2[C@@H]([C@H](O)[C@@H](COP([O-])(=O)OP(O)(=O)OC[C@@H]3[C@H]([C@@H](O)[C@@H](O3)N3C4=NC=NC(N)=C4N=C3)O)O2)O)=C1 BAWFJGJZGIEFAR-NNYOXOHSSA-N 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- 206010037124 Pseudohermaphroditism male Diseases 0.000 description 2
- NKANXQFJJICGDU-QPLCGJKRSA-N Tamoxifen Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCCN(C)C)=CC=1)/C1=CC=CC=C1 NKANXQFJJICGDU-QPLCGJKRSA-N 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- 230000001270 agonistic effect Effects 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 125000003342 alkenyl group Chemical group 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- VZTDIZULWFCMLS-UHFFFAOYSA-N ammonium formate Chemical compound [NH4+].[O-]C=O VZTDIZULWFCMLS-UHFFFAOYSA-N 0.000 description 2
- MTHSVFCYNBDYFN-UHFFFAOYSA-N anhydrous diethylene glycol Natural products OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 2
- 239000005557 antagonist Substances 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 125000003710 aryl alkyl group Chemical group 0.000 description 2
- 208000010668 atopic eczema Diseases 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- 210000002449 bone cell Anatomy 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 125000002837 carbocyclic group Chemical group 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 150000001735 carboxylic acids Chemical class 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 238000007385 chemical modification Methods 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 235000015165 citric acid Nutrition 0.000 description 2
- LSXDOTMGLUJQCM-UHFFFAOYSA-M copper(i) iodide Chemical compound I[Cu] LSXDOTMGLUJQCM-UHFFFAOYSA-M 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 239000010779 crude oil Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 2
- 239000002270 dispersing agent Substances 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000002255 enzymatic effect Effects 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 102000015694 estrogen receptors Human genes 0.000 description 2
- 229940093499 ethyl acetate Drugs 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 230000035558 fertility Effects 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 235000013373 food additive Nutrition 0.000 description 2
- 239000002778 food additive Substances 0.000 description 2
- 125000002541 furyl group Chemical group 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 229940093915 gynecological organic acid Drugs 0.000 description 2
- XLYOFNOQVPJJNP-ZSJDYOACSA-N heavy water Substances [2H]O[2H] XLYOFNOQVPJJNP-ZSJDYOACSA-N 0.000 description 2
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 2
- 238000001794 hormone therapy Methods 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002431 hydrogen Chemical group 0.000 description 2
- 208000022168 hypermenorrhea Diseases 0.000 description 2
- 238000009802 hysterectomy Methods 0.000 description 2
- 239000005457 ice water Substances 0.000 description 2
- 125000002883 imidazolyl group Chemical group 0.000 description 2
- 230000001771 impaired effect Effects 0.000 description 2
- 238000005462 in vivo assay Methods 0.000 description 2
- 238000011065 in-situ storage Methods 0.000 description 2
- 230000002779 inactivation Effects 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 125000001041 indolyl group Chemical group 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000004811 liquid chromatography Methods 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 229910001623 magnesium bromide Inorganic materials 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 238000001819 mass spectrum Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- PQIOSYKVBBWRRI-UHFFFAOYSA-N methylphosphonyl difluoride Chemical group CP(F)(F)=O PQIOSYKVBBWRRI-UHFFFAOYSA-N 0.000 description 2
- 239000011859 microparticle Substances 0.000 description 2
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 229950006238 nadide Drugs 0.000 description 2
- 229930027945 nicotinamide-adenine dinucleotide Natural products 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- 108091008589 nuclear estrogen receptors Proteins 0.000 description 2
- 125000001209 o-nitrophenyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])[N+]([O-])=O 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 210000000963 osteoblast Anatomy 0.000 description 2
- ZRSNZINYAWTAHE-UHFFFAOYSA-N p-methoxybenzaldehyde Chemical compound COC1=CC=C(C=O)C=C1 ZRSNZINYAWTAHE-UHFFFAOYSA-N 0.000 description 2
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 239000002953 phosphate buffered saline Substances 0.000 description 2
- 210000002826 placenta Anatomy 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- GNSKLFRGEWLPPA-UHFFFAOYSA-M potassium dihydrogen phosphate Chemical compound [K+].OP(O)([O-])=O GNSKLFRGEWLPPA-UHFFFAOYSA-M 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 239000000186 progesterone Substances 0.000 description 2
- 229960003387 progesterone Drugs 0.000 description 2
- 230000002035 prolonged effect Effects 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 238000000159 protein binding assay Methods 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 125000003226 pyrazolyl group Chemical group 0.000 description 2
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 2
- 230000030541 receptor transactivation Effects 0.000 description 2
- 102000005962 receptors Human genes 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 230000008685 targeting Effects 0.000 description 2
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 description 2
- 125000000335 thiazolyl group Chemical group 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- 230000037317 transdermal delivery Effects 0.000 description 2
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 238000007514 turning Methods 0.000 description 2
- PROQIPRRNZUXQM-UHFFFAOYSA-N (16alpha,17betaOH)-Estra-1,3,5(10)-triene-3,16,17-triol Natural products OC1=CC=C2C3CCC(C)(C(C(O)C4)O)C4C3CCC2=C1 PROQIPRRNZUXQM-UHFFFAOYSA-N 0.000 description 1
- RWBRUCCWZPSBFC-SJOKZOANSA-N (20R)-20-hydroxypregn-4-en-3-one Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](O)C)[C@@]1(C)CC2 RWBRUCCWZPSBFC-SJOKZOANSA-N 0.000 description 1
- WWTBZEKOSBFBEM-SPWPXUSOSA-N (2s)-2-[[2-benzyl-3-[hydroxy-[(1r)-2-phenyl-1-(phenylmethoxycarbonylamino)ethyl]phosphoryl]propanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound N([C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)O)C(=O)C(CP(O)(=O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1C=CC=CC=1)CC1=CC=CC=C1 WWTBZEKOSBFBEM-SPWPXUSOSA-N 0.000 description 1
- 102100022582 (3R)-3-hydroxyacyl-CoA dehydrogenase Human genes 0.000 description 1
- 101710120738 (3R)-3-hydroxyacyl-CoA dehydrogenase Proteins 0.000 description 1
- 125000006527 (C1-C5) alkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- KQZLRWGGWXJPOS-NLFPWZOASA-N 1-[(1R)-1-(2,4-dichlorophenyl)ethyl]-6-[(4S,5R)-4-[(2S)-2-(hydroxymethyl)pyrrolidin-1-yl]-5-methylcyclohexen-1-yl]pyrazolo[3,4-b]pyrazine-3-carbonitrile Chemical compound ClC1=C(C=CC(=C1)Cl)[C@@H](C)N1N=C(C=2C1=NC(=CN=2)C1=CC[C@@H]([C@@H](C1)C)N1[C@@H](CCC1)CO)C#N KQZLRWGGWXJPOS-NLFPWZOASA-N 0.000 description 1
- WZZBNLYBHUDSHF-DHLKQENFSA-N 1-[(3s,4s)-4-[8-(2-chloro-4-pyrimidin-2-yloxyphenyl)-7-fluoro-2-methylimidazo[4,5-c]quinolin-1-yl]-3-fluoropiperidin-1-yl]-2-hydroxyethanone Chemical compound CC1=NC2=CN=C3C=C(F)C(C=4C(=CC(OC=5N=CC=CN=5)=CC=4)Cl)=CC3=C2N1[C@H]1CCN(C(=O)CO)C[C@@H]1F WZZBNLYBHUDSHF-DHLKQENFSA-N 0.000 description 1
- ONBQEOIKXPHGMB-VBSBHUPXSA-N 1-[2-[(2s,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-4,6-dihydroxyphenyl]-3-(4-hydroxyphenyl)propan-1-one Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=CC(O)=C1C(=O)CCC1=CC=C(O)C=C1 ONBQEOIKXPHGMB-VBSBHUPXSA-N 0.000 description 1
- LIJPUYWRAJPDLJ-UHFFFAOYSA-N 1-benzyl-1,2,3,4-tetrahydronaphthalene Chemical class C1CCC2=CC=CC=C2C1CC1=CC=CC=C1 LIJPUYWRAJPDLJ-UHFFFAOYSA-N 0.000 description 1
- PZYHLENTJZMOQC-UHFFFAOYSA-N 1-bromohexan-1-ol Chemical compound CCCCCC(O)Br PZYHLENTJZMOQC-UHFFFAOYSA-N 0.000 description 1
- RTBFRGCFXZNCOE-UHFFFAOYSA-N 1-methylsulfonylpiperidin-4-one Chemical compound CS(=O)(=O)N1CCC(=O)CC1 RTBFRGCFXZNCOE-UHFFFAOYSA-N 0.000 description 1
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- 102000054917 17-beta-hydroxysteroid dehydrogenase type 3 Human genes 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-N 2-Methylbenzenesulfonic acid Chemical compound CC1=CC=CC=C1S(O)(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-N 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- LGHBWDKMGOIZKH-CBZIJGRNSA-N 3-Deoxyestrone Chemical class C1=CC=C2[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1 LGHBWDKMGOIZKH-CBZIJGRNSA-N 0.000 description 1
- QBWKPGNFQQJGFY-QLFBSQMISA-N 3-[(1r)-1-[(2r,6s)-2,6-dimethylmorpholin-4-yl]ethyl]-n-[6-methyl-3-(1h-pyrazol-4-yl)imidazo[1,2-a]pyrazin-8-yl]-1,2-thiazol-5-amine Chemical compound N1([C@H](C)C2=NSC(NC=3C4=NC=C(N4C=C(C)N=3)C3=CNN=C3)=C2)C[C@H](C)O[C@H](C)C1 QBWKPGNFQQJGFY-QLFBSQMISA-N 0.000 description 1
- AODMJIOEGCBUQL-UHFFFAOYSA-N 3-ethynylphenol Chemical group OC1=CC=CC(C#C)=C1 AODMJIOEGCBUQL-UHFFFAOYSA-N 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- OSJPPGNTCRNQQC-UHFFFAOYSA-N 3-phosphoglyceric acid Chemical compound OC(=O)C(O)COP(O)(O)=O OSJPPGNTCRNQQC-UHFFFAOYSA-N 0.000 description 1
- KBSDLBVPAHQCRY-UHFFFAOYSA-N 307496-19-1 Chemical group C1CC=CCC1CC[Si](O1)(O2)O[Si](O3)(C4CCCC4)O[Si](O4)(C5CCCC5)O[Si]1(C1CCCC1)O[Si](O1)(C5CCCC5)O[Si]2(C2CCCC2)O[Si]3(C2CCCC2)O[Si]41C1CCCC1 KBSDLBVPAHQCRY-UHFFFAOYSA-N 0.000 description 1
- KSZVOXHGCKKOLL-UHFFFAOYSA-N 4-Ethynyltoluene Chemical group CC1=CC=C(C#C)C=C1 KSZVOXHGCKKOLL-UHFFFAOYSA-N 0.000 description 1
- DMAYBPBPEUFIHJ-UHFFFAOYSA-N 4-bromobut-1-ene Chemical compound BrCCC=C DMAYBPBPEUFIHJ-UHFFFAOYSA-N 0.000 description 1
- SJZRECIVHVDYJC-UHFFFAOYSA-N 4-hydroxybutyric acid Chemical compound OCCCC(O)=O SJZRECIVHVDYJC-UHFFFAOYSA-N 0.000 description 1
- OXRWICUICBZVAE-UHFFFAOYSA-N 4-methylpent-1-yne Chemical compound CC(C)CC#C OXRWICUICBZVAE-UHFFFAOYSA-N 0.000 description 1
- LPNANKDXVBMDKE-UHFFFAOYSA-N 5-bromopent-1-ene Chemical compound BrCCCC=C LPNANKDXVBMDKE-UHFFFAOYSA-N 0.000 description 1
- HKNANEMUCJGPMS-UHFFFAOYSA-N 5-methylhex-1-yne Chemical compound CC(C)CCC#C HKNANEMUCJGPMS-UHFFFAOYSA-N 0.000 description 1
- QGXBDMJGAMFCBF-HLUDHZFRSA-N 5α-Androsterone Chemical compound C1[C@H](O)CC[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CC[C@H]21 QGXBDMJGAMFCBF-HLUDHZFRSA-N 0.000 description 1
- 208000037853 Abnormal uterine bleeding Diseases 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical compound N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 1
- 239000005695 Ammonium acetate Substances 0.000 description 1
- 201000005670 Anovulation Diseases 0.000 description 1
- 206010002659 Anovulatory cycle Diseases 0.000 description 1
- 208000033258 Bifunctional enzyme deficiency Diseases 0.000 description 1
- 229930191576 Biochanin Natural products 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 238000006418 Brown reaction Methods 0.000 description 1
- QIMMUPPBPVKWKM-UHFFFAOYSA-N CC1=CC2=CC=CC=C2C=C1 Chemical compound CC1=CC2=CC=CC=C2C=C1 QIMMUPPBPVKWKM-UHFFFAOYSA-N 0.000 description 1
- BSKHPKMHTQYZBB-UHFFFAOYSA-N CC1=CC=CC=N1 Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 1
- ITQTTZVARXURQS-UHFFFAOYSA-N CC1=CC=CN=C1 Chemical compound CC1=CC=CN=C1 ITQTTZVARXURQS-UHFFFAOYSA-N 0.000 description 1
- XQQBUAPQHNYYRS-UHFFFAOYSA-N CC1=CC=CS1 Chemical compound CC1=CC=CS1 XQQBUAPQHNYYRS-UHFFFAOYSA-N 0.000 description 1
- FKNQCJSGGFJEIZ-UHFFFAOYSA-N CC1=CC=NC=C1 Chemical compound CC1=CC=NC=C1 FKNQCJSGGFJEIZ-UHFFFAOYSA-N 0.000 description 1
- XLSZMDLNRCVEIJ-UHFFFAOYSA-N CC1=CN=CN1 Chemical compound CC1=CN=CN1 XLSZMDLNRCVEIJ-UHFFFAOYSA-N 0.000 description 1
- KJRRQXYWFQKJIP-UHFFFAOYSA-N CC1=COC=C1 Chemical compound CC1=COC=C1 KJRRQXYWFQKJIP-UHFFFAOYSA-N 0.000 description 1
- QENGPZGAWFQWCZ-UHFFFAOYSA-N CC1=CSC=C1 Chemical compound CC1=CSC=C1 QENGPZGAWFQWCZ-UHFFFAOYSA-N 0.000 description 1
- VNXBKJFUJUWOCW-UHFFFAOYSA-N CC1CC1 Chemical compound CC1CC1 VNXBKJFUJUWOCW-UHFFFAOYSA-N 0.000 description 1
- GDOPTJXRTPNYNR-UHFFFAOYSA-N CC1CCCC1 Chemical compound CC1CCCC1 GDOPTJXRTPNYNR-UHFFFAOYSA-N 0.000 description 1
- UAEPNZWRGJTJPN-UHFFFAOYSA-N CC1CCCCC1 Chemical compound CC1CCCCC1 UAEPNZWRGJTJPN-UHFFFAOYSA-N 0.000 description 1
- IKHGUXGNUITLKF-UHFFFAOYSA-N CC=O Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 1
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N CCC1=CC=CC=C1 Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 description 1
- IIEWJVIFRVWJOD-UHFFFAOYSA-N CCC1CCCCC1 Chemical compound CCC1CCCCC1 IIEWJVIFRVWJOD-UHFFFAOYSA-N 0.000 description 1
- XXBDWLFCJWSEKW-UHFFFAOYSA-N CN(C)CC1=CC=CC=C1 Chemical compound CN(C)CC1=CC=CC=C1 XXBDWLFCJWSEKW-UHFFFAOYSA-N 0.000 description 1
- BCTLXPQIZLIYQA-UHFFFAOYSA-N CN1CC=CC1.CN1CC=CCC1.CN1CCC1.CN1CCC2=C(C=CC=C2)C1.CN1CCCC1.CN1CCCC2CCCCC21.CN1CCCCC1.CN1CCCCCC1.CN1CCCNCC1.CN1CCCOCC1.CN1CCCSCC1.CN1CCNCC1.CN1CCOC1.CN1CCOCC1.CN1CCS(=O)(=O)CC1.CN1CCSC1.CN1CCSCC1 Chemical compound CN1CC=CC1.CN1CC=CCC1.CN1CCC1.CN1CCC2=C(C=CC=C2)C1.CN1CCCC1.CN1CCCC2CCCCC21.CN1CCCCC1.CN1CCCCCC1.CN1CCCNCC1.CN1CCCOCC1.CN1CCCSCC1.CN1CCNCC1.CN1CCOC1.CN1CCOCC1.CN1CCS(=O)(=O)CC1.CN1CCSC1.CN1CCSCC1 BCTLXPQIZLIYQA-UHFFFAOYSA-N 0.000 description 1
- ULZCOWMSBOJCLT-UHFFFAOYSA-N CN1CCS(=O)(=O)CC1 Chemical compound CN1CCS(=O)(=O)CC1 ULZCOWMSBOJCLT-UHFFFAOYSA-N 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N COC(C)=O Chemical compound COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- CZXGXYBOQYQXQD-UHFFFAOYSA-N COS(=O)(=O)C1=CC=CC=C1 Chemical compound COS(=O)(=O)C1=CC=CC=C1 CZXGXYBOQYQXQD-UHFFFAOYSA-N 0.000 description 1
- HHVIBTZHLRERCL-UHFFFAOYSA-N CS(C)(=O)=O Chemical compound CS(C)(=O)=O HHVIBTZHLRERCL-UHFFFAOYSA-N 0.000 description 1
- LYGJENNIWJXYER-UHFFFAOYSA-N C[N+](=O)[O-] Chemical compound C[N+](=O)[O-] LYGJENNIWJXYER-UHFFFAOYSA-N 0.000 description 1
- 101100451537 Caenorhabditis elegans hsd-1 gene Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000288950 Callithrix jacchus Species 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 206010053567 Coagulopathies Diseases 0.000 description 1
- 108020004414 DNA Proteins 0.000 description 1
- 206010012110 Defaecation urgency Diseases 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 238000009007 Diagnostic Kit Methods 0.000 description 1
- BUDQDWGNQVEFAC-UHFFFAOYSA-N Dihydropyran Chemical compound C1COC=CC1 BUDQDWGNQVEFAC-UHFFFAOYSA-N 0.000 description 1
- 208000027877 Disorders of Sex Development Diseases 0.000 description 1
- 102100021218 Dual oxidase 1 Human genes 0.000 description 1
- 208000004483 Dyspareunia Diseases 0.000 description 1
- 108010085330 Estradiol Receptors Proteins 0.000 description 1
- 102000007594 Estrogen Receptor alpha Human genes 0.000 description 1
- 102000000509 Estrogen Receptor beta Human genes 0.000 description 1
- 101710196141 Estrogen receptor Proteins 0.000 description 1
- QGXBDMJGAMFCBF-UHFFFAOYSA-N Etiocholanolone Natural products C1C(O)CCC2(C)C3CCC(C)(C(CC4)=O)C4C3CCC21 QGXBDMJGAMFCBF-UHFFFAOYSA-N 0.000 description 1
- 108700012941 GNRH1 Proteins 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- BJJXHLWLUDYTGC-ANULTFPQSA-N Gestrinone Chemical compound C1CC(=O)C=C2CC[C@@H]([C@H]3[C@@](CC)([C@](CC3)(O)C#C)C=C3)C3=C21 BJJXHLWLUDYTGC-ANULTFPQSA-N 0.000 description 1
- DHCLVCXQIBBOPH-UHFFFAOYSA-N Glycerol 2-phosphate Chemical compound OCC(CO)OP(O)(O)=O DHCLVCXQIBBOPH-UHFFFAOYSA-N 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 239000000579 Gonadotropin-Releasing Hormone Substances 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 238000010268 HPLC based assay Methods 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 101000968308 Homo sapiens Dual oxidase 1 Proteins 0.000 description 1
- 101000968305 Homo sapiens Dual oxidase 2 Proteins 0.000 description 1
- 101000882584 Homo sapiens Estrogen receptor Proteins 0.000 description 1
- 238000006736 Huisgen cycloaddition reaction Methods 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 238000000023 Kugelrohr distillation Methods 0.000 description 1
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 1
- 229930182816 L-glutamine Natural products 0.000 description 1
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 208000037093 Menstruation Disturbances Diseases 0.000 description 1
- 241000713333 Mouse mammary tumor virus Species 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- 238000000636 Northern blotting Methods 0.000 description 1
- 108020005497 Nuclear hormone receptor Proteins 0.000 description 1
- 208000034424 Painful defaecation Diseases 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- 208000000450 Pelvic Pain Diseases 0.000 description 1
- 102100022587 Peroxisomal multifunctional enzyme type 2 Human genes 0.000 description 1
- 101710125609 Peroxisomal multifunctional enzyme type 2 Proteins 0.000 description 1
- 208000037062 Polyps Diseases 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- 102100025803 Progesterone receptor Human genes 0.000 description 1
- 201000005613 Pseudohermaphroditism Diseases 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 108700008625 Reporter Genes Proteins 0.000 description 1
- 108091027981 Response element Proteins 0.000 description 1
- 206010065951 Retrograde menstruation Diseases 0.000 description 1
- 101000857870 Squalus acanthias Gonadoliberin Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 108010085012 Steroid Receptors Proteins 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical compound [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 description 1
- 108010090932 Vitellogenins Proteins 0.000 description 1
- QXUQAWSCCQLPEQ-OFBSFOQBSA-N [H][C@]12CC[C@]3(C)C(=O)CC(CCCC4=CN(CCCC)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)CC(CCCC4=CN(CCCC)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 QXUQAWSCCQLPEQ-OFBSFOQBSA-N 0.000 description 1
- LQZUYMGPYWWVKQ-HKYAYREKSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCC4=CN(C5=CC=C(F)C=C5F)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCC4=CN(C5=CC=C(F)C=C5F)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 LQZUYMGPYWWVKQ-HKYAYREKSA-N 0.000 description 1
- WYYSVLZVIWULFA-TYVLPFHYSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCCCN4C=C(C5=CC(O)=CC=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCCCN4C=C(C5=CC(O)=CC=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 WYYSVLZVIWULFA-TYVLPFHYSA-N 0.000 description 1
- JXDRPJCVOHVSLB-QQMXCIRCSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCCCN4C=C(CC(C)C)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCCCN4C=C(CC(C)C)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 JXDRPJCVOHVSLB-QQMXCIRCSA-N 0.000 description 1
- KAVLHNPSYWZZRZ-BPEOURKJSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCCCN4C=C(CCC(C)C)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCCCN4C=C(CCC(C)C)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 KAVLHNPSYWZZRZ-BPEOURKJSA-N 0.000 description 1
- LWEHHDNLCLJLHQ-YUJZLMECSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(C5=CC=C(C)C=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(C5=CC=C(C)C=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 LWEHHDNLCLJLHQ-YUJZLMECSA-N 0.000 description 1
- LZBOSWINZQJSMF-OIBJXCRFSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(C5CC5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(C5CC5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 LZBOSWINZQJSMF-OIBJXCRFSA-N 0.000 description 1
- UJJZTOPDWMSZPO-PHDYFPKISA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(CCC(C)C)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(CCC(C)C)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 UJJZTOPDWMSZPO-PHDYFPKISA-N 0.000 description 1
- NOYMPDBSLUHFFL-XAKHRUBQSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(CCC5=CC=CC=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(CCC5=CC=CC=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 NOYMPDBSLUHFFL-XAKHRUBQSA-N 0.000 description 1
- ZEWZAJMRUDDXHJ-UTCLPMRJSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(CO)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCCN4C=C(CO)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 ZEWZAJMRUDDXHJ-UTCLPMRJSA-N 0.000 description 1
- NJHAEZOPHNUDKX-AOPVMBEZSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5=CC=C(CO)C=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5=CC=C(CO)C=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 NJHAEZOPHNUDKX-AOPVMBEZSA-N 0.000 description 1
- JXJXOACXNRRMAL-PQBHNLNBSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5=CC=CC=C5[N+](=O)[O-])N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5=CC=CC=C5[N+](=O)[O-])N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 JXJXOACXNRRMAL-PQBHNLNBSA-N 0.000 description 1
- OSOUTPFHOJMSRD-ZORQMFODSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5=CSC=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5=CSC=C5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 OSOUTPFHOJMSRD-ZORQMFODSA-N 0.000 description 1
- WUSOIBRCODZEBT-AMIBRAFWSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5CC5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4C=C(C5CC5)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 WUSOIBRCODZEBT-AMIBRAFWSA-N 0.000 description 1
- LNBHPTHYMCXQHP-PPLIHSAZSA-N [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4N=NC=C4C4=CC=C(C(=O)OC)C=C4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(=O)C[C@@H](CCCN4N=NC=C4C4=CC=C(C(=O)OC)C=C4)[C@@]3([H])[C@]1([H])CCC1=CC(OC)=CC=C12 LNBHPTHYMCXQHP-PPLIHSAZSA-N 0.000 description 1
- AFEYLEHHAOFAHM-ISCGPHEISA-N [H][C@]12CC[C@]3(C)C(F)(F)C[C@@H](CCCC4=CN(C5=CC=C(F)C=C5F)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)C(F)(F)C[C@@H](CCCC4=CN(C5=CC=C(F)C=C5F)N=N4)[C@@]3([H])[C@]1([H])CCC1=CC(O)=CC=C12 AFEYLEHHAOFAHM-ISCGPHEISA-N 0.000 description 1
- HLCRYAZDZCJZFG-BDXSIMOUSA-N [H][C@]12CC[C@]3(C)CCC[C@@]3([H])[C@]1([H])CCC1=CC=CC=C12 Chemical compound [H][C@]12CC[C@]3(C)CCC[C@@]3([H])[C@]1([H])CCC1=CC=CC=C12 HLCRYAZDZCJZFG-BDXSIMOUSA-N 0.000 description 1
- DWOOJRLYAJFDQP-UHFFFAOYSA-N [Na+].[Pb+2].[N-]=[N+]=[N-].[N-]=[N+]=[N-].[N-]=[N+]=[N-] Chemical group [Na+].[Pb+2].[N-]=[N+]=[N-].[N-]=[N+]=[N-].[N-]=[N+]=[N-] DWOOJRLYAJFDQP-UHFFFAOYSA-N 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 206010000210 abortion Diseases 0.000 description 1
- 231100000176 abortion Toxicity 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 238000011292 agonist therapy Methods 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 239000004411 aluminium Substances 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001409 amidines Chemical class 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- XKMRRTOUMJRJIA-UHFFFAOYSA-N ammonia nh3 Chemical compound N.N XKMRRTOUMJRJIA-UHFFFAOYSA-N 0.000 description 1
- 235000019257 ammonium acetate Nutrition 0.000 description 1
- 229940043376 ammonium acetate Drugs 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 102000001307 androgen receptors Human genes 0.000 description 1
- 229940094957 androgens and estrogen Drugs 0.000 description 1
- AEMFNILZOJDQLW-QAGGRKNESA-N androst-4-ene-3,17-dione Chemical compound O=C1CC[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1 AEMFNILZOJDQLW-QAGGRKNESA-N 0.000 description 1
- 229960005471 androstenedione Drugs 0.000 description 1
- AEMFNILZOJDQLW-UHFFFAOYSA-N androstenedione Natural products O=C1CCC2(C)C3CCC(C)(C(CC4)=O)C4C3CCC2=C1 AEMFNILZOJDQLW-UHFFFAOYSA-N 0.000 description 1
- 229940061641 androsterone Drugs 0.000 description 1
- JFCQEDHGNNZCLN-UHFFFAOYSA-N anhydrous glutaric acid Natural products OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 description 1
- 231100000552 anovulation Toxicity 0.000 description 1
- 230000002280 anti-androgenic effect Effects 0.000 description 1
- 230000003388 anti-hormonal effect Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 238000011861 anti-inflammatory therapy Methods 0.000 description 1
- 239000000051 antiandrogen Substances 0.000 description 1
- 229940030495 antiandrogen sex hormone and modulator of the genital system Drugs 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 238000003782 apoptosis assay Methods 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- RFRXIWQYSOIBDI-UHFFFAOYSA-N benzarone Chemical compound CCC=1OC2=CC=CC=C2C=1C(=O)C1=CC=C(O)C=C1 RFRXIWQYSOIBDI-UHFFFAOYSA-N 0.000 description 1
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzonitrile Substances N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 description 1
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- 150000003939 benzylamines Chemical class 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- WUADCCWRTIWANL-UHFFFAOYSA-N biochanin A Chemical compound C1=CC(OC)=CC=C1C1=COC2=CC(O)=CC(O)=C2C1=O WUADCCWRTIWANL-UHFFFAOYSA-N 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 238000012925 biological evaluation Methods 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 208000015294 blood coagulation disease Diseases 0.000 description 1
- 210000000746 body region Anatomy 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- QDEOKXOYHYUKMS-UHFFFAOYSA-N but-3-ynylbenzene Chemical compound C#CCCC1=CC=CC=C1 QDEOKXOYHYUKMS-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- KMGBZBJJOKUPIA-UHFFFAOYSA-N butyl iodide Chemical compound CCCCI KMGBZBJJOKUPIA-UHFFFAOYSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 239000012876 carrier material Substances 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 230000001364 causal effect Effects 0.000 description 1
- 238000000423 cell based assay Methods 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000003610 charcoal Substances 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- 238000010568 chiral column chromatography Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 230000009852 coagulant defect Effects 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 230000003920 cognitive function Effects 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 229940126142 compound 16 Drugs 0.000 description 1
- 229940125810 compound 20 Drugs 0.000 description 1
- 229940126208 compound 22 Drugs 0.000 description 1
- 229940125846 compound 25 Drugs 0.000 description 1
- 229940125877 compound 31 Drugs 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 229910000365 copper sulfate Inorganic materials 0.000 description 1
- GBRBMTNGQBKBQE-UHFFFAOYSA-L copper;diiodide Chemical compound I[Cu]I GBRBMTNGQBKBQE-UHFFFAOYSA-L 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- DMSZORWOGDLWGN-UHFFFAOYSA-N ctk1a3526 Chemical compound NP(N)(N)=O DMSZORWOGDLWGN-UHFFFAOYSA-N 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- POZRVZJJTULAOH-LHZXLZLDSA-N danazol Chemical compound C1[C@]2(C)[C@H]3CC[C@](C)([C@](CC4)(O)C#C)[C@@H]4[C@@H]3CCC2=CC2=C1C=NO2 POZRVZJJTULAOH-LHZXLZLDSA-N 0.000 description 1
- 229960000766 danazol Drugs 0.000 description 1
- DEZRYPDIMOWBDS-UHFFFAOYSA-N dcm dichloromethane Chemical compound ClCCl.ClCCl DEZRYPDIMOWBDS-UHFFFAOYSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 229920006237 degradable polymer Polymers 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 125000005959 diazepanyl group Chemical group 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 125000006222 dimethylaminomethyl group Chemical group [H]C([H])([H])N(C([H])([H])[H])C([H])([H])* 0.000 description 1
- UXGNZZKBCMGWAZ-UHFFFAOYSA-N dimethylformamide dmf Chemical compound CN(C)C=O.CN(C)C=O UXGNZZKBCMGWAZ-UHFFFAOYSA-N 0.000 description 1
- 230000003292 diminished effect Effects 0.000 description 1
- CETRZFQIITUQQL-UHFFFAOYSA-N dmso dimethylsulfoxide Chemical compound CS(C)=O.CS(C)=O CETRZFQIITUQQL-UHFFFAOYSA-N 0.000 description 1
- 210000003717 douglas' pouch Anatomy 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 239000000890 drug combination Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000009510 drug design Methods 0.000 description 1
- 238000007876 drug discovery Methods 0.000 description 1
- 239000013583 drug formulation Substances 0.000 description 1
- 206010013990 dysuria Diseases 0.000 description 1
- 201000003511 ectopic pregnancy Diseases 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 1
- 238000000921 elemental analysis Methods 0.000 description 1
- 150000002081 enamines Chemical class 0.000 description 1
- 238000009261 endocrine therapy Methods 0.000 description 1
- 229940034984 endocrine therapy antineoplastic and immunomodulating agent Drugs 0.000 description 1
- 201000003914 endometrial carcinoma Diseases 0.000 description 1
- 210000005168 endometrial cell Anatomy 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 102000006966 enzyme regulator activity proteins Human genes 0.000 description 1
- 108040000578 enzyme regulator activity proteins Proteins 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 235000020776 essential amino acid Nutrition 0.000 description 1
- VOXZDWNPVJITMN-WKUFJEKOSA-N estradiol group Chemical group [C@@H]12CCC(O)[C@@]1(C)CC[C@@H]1C3=C(CC[C@@H]21)C=C(O)C=C3 VOXZDWNPVJITMN-WKUFJEKOSA-N 0.000 description 1
- PROQIPRRNZUXQM-ZXXIGWHRSA-N estriol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H]([C@H](O)C4)O)[C@@H]4[C@@H]3CCC2=C1 PROQIPRRNZUXQM-ZXXIGWHRSA-N 0.000 description 1
- 229960001348 estriol Drugs 0.000 description 1
- 229940106582 estrogenic substances Drugs 0.000 description 1
- JKKFKPJIXZFSSB-CBZIJGRNSA-N estrone 3-sulfate Chemical compound OS(=O)(=O)OC1=CC=C2[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1 JKKFKPJIXZFSSB-CBZIJGRNSA-N 0.000 description 1
- LHWWETDBWVTKJO-UHFFFAOYSA-N et3n triethylamine Chemical compound CCN(CC)CC.CCN(CC)CC LHWWETDBWVTKJO-UHFFFAOYSA-N 0.000 description 1
- 239000002038 ethyl acetate fraction Substances 0.000 description 1
- OJCSPXHYDFONPU-UHFFFAOYSA-N etoac etoac Chemical compound CCOC(C)=O.CCOC(C)=O OJCSPXHYDFONPU-UHFFFAOYSA-N 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000001400 expression cloning Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 210000004996 female reproductive system Anatomy 0.000 description 1
- 239000012894 fetal calf serum Substances 0.000 description 1
- 230000001605 fetal effect Effects 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 238000010579 first pass effect Methods 0.000 description 1
- 238000003818 flash chromatography Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- UHCBBWUQDAVSMS-UHFFFAOYSA-N fluoroethane Chemical compound CCF UHCBBWUQDAVSMS-UHFFFAOYSA-N 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 238000001640 fractional crystallisation Methods 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 238000002825 functional assay Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 235000021472 generally recognized as safe Nutrition 0.000 description 1
- 229960004761 gestrinone Drugs 0.000 description 1
- 210000004907 gland Anatomy 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- YIFYYPKWOQSCRI-AZUAARDMSA-N glyceollin Chemical class O1C2=CC(O)=CC=C2[C@@]2(O)[C@@H]1C1=CC=C3OC(C)(C)C=CC3=C1OC2 YIFYYPKWOQSCRI-AZUAARDMSA-N 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- JAXFJECJQZDFJS-XHEPKHHKSA-N gtpl8555 Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@@H]1C(=O)N[C@H](B1O[C@@]2(C)[C@H]3C[C@H](C3(C)C)C[C@H]2O1)CCC1=CC=C(F)C=C1 JAXFJECJQZDFJS-XHEPKHHKSA-N 0.000 description 1
- JEGUKCSWCFPDGT-UHFFFAOYSA-N h2o hydrate Chemical compound O.O JEGUKCSWCFPDGT-UHFFFAOYSA-N 0.000 description 1
- 208000024963 hair loss Diseases 0.000 description 1
- 230000003676 hair loss Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006038 hexenyl group Chemical group 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000003054 hormonal effect Effects 0.000 description 1
- 102000055946 human HSD17B2 Human genes 0.000 description 1
- 108700010802 human intersex Proteins 0.000 description 1
- 102000058032 human intersex Human genes 0.000 description 1
- 238000009396 hybridization Methods 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- ARRNBPCNZJXHRJ-UHFFFAOYSA-M hydron;tetrabutylazanium;phosphate Chemical compound OP(O)([O-])=O.CCCC[N+](CCCC)(CCCC)CCCC ARRNBPCNZJXHRJ-UHFFFAOYSA-M 0.000 description 1
- 125000001165 hydrophobic group Chemical group 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 239000012729 immediate-release (IR) formulation Substances 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 208000003243 intestinal obstruction Diseases 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000013038 irreversible inhibitor Substances 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 238000002357 laparoscopic surgery Methods 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 239000006194 liquid suspension Substances 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000012139 lysis buffer Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- RMGJCSHZTFKPNO-UHFFFAOYSA-M magnesium;ethene;bromide Chemical compound [Mg+2].[Br-].[CH-]=C RMGJCSHZTFKPNO-UHFFFAOYSA-M 0.000 description 1
- DQEUYIQDSMINEY-UHFFFAOYSA-M magnesium;prop-1-ene;bromide Chemical compound [Mg+2].[Br-].[CH2-]C=C DQEUYIQDSMINEY-UHFFFAOYSA-M 0.000 description 1
- CYSFUFRXDOAOMP-UHFFFAOYSA-M magnesium;prop-1-ene;chloride Chemical compound [Mg+2].[Cl-].[CH2-]C=C CYSFUFRXDOAOMP-UHFFFAOYSA-M 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 210000005075 mammary gland Anatomy 0.000 description 1
- 241001515942 marmosets Species 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 238000013160 medical therapy Methods 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 230000009245 menopause Effects 0.000 description 1
- COTNUBDHGSIOTA-UHFFFAOYSA-N meoh methanol Chemical compound OC.OC COTNUBDHGSIOTA-UHFFFAOYSA-N 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 238000005649 metathesis reaction Methods 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- WCYWZMWISLQXQU-UHFFFAOYSA-N methyl Chemical class [CH3] WCYWZMWISLQXQU-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000006431 methyl cyclopropyl group Chemical group 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 230000003228 microsomal effect Effects 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- SYSQUGFVNFXIIT-UHFFFAOYSA-N n-[4-(1,3-benzoxazol-2-yl)phenyl]-4-nitrobenzenesulfonamide Chemical class C1=CC([N+](=O)[O-])=CC=C1S(=O)(=O)NC1=CC=C(C=2OC3=CC=CC=C3N=2)C=C1 SYSQUGFVNFXIIT-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- 239000006070 nanosuspension Substances 0.000 description 1
- 239000006225 natural substrate Substances 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910000069 nitrogen hydride Inorganic materials 0.000 description 1
- 231100000344 non-irritating Toxicity 0.000 description 1
- BKIMMITUMNQMOS-UHFFFAOYSA-N normal nonane Natural products CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 102000006255 nuclear receptors Human genes 0.000 description 1
- 108020004017 nuclear receptors Proteins 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 201000008968 osteosarcoma Diseases 0.000 description 1
- 230000002611 ovarian Effects 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- 210000003101 oviduct Anatomy 0.000 description 1
- 230000027758 ovulation cycle Effects 0.000 description 1
- 125000005961 oxazepanyl group Chemical group 0.000 description 1
- 125000000160 oxazolidinyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- 230000003076 paracrine Effects 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000007310 pathophysiology Effects 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 125000002255 pentenyl group Chemical group C(=CCCC)* 0.000 description 1
- 125000006194 pentinyl group Chemical group 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 210000003200 peritoneal cavity Anatomy 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 238000005191 phase separation Methods 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- 150000003009 phosphonic acids Chemical class 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 235000017807 phytochemicals Nutrition 0.000 description 1
- 239000003075 phytoestrogen Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 150000003053 piperidines Chemical class 0.000 description 1
- 229930000223 plant secondary metabolite Natural products 0.000 description 1
- 208000030761 polycystic kidney disease Diseases 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 230000023603 positive regulation of transcription initiation, DNA-dependent Effects 0.000 description 1
- 238000002600 positron emission tomography Methods 0.000 description 1
- NTTOTNSKUYCDAV-UHFFFAOYSA-N potassium hydride Chemical compound [KH] NTTOTNSKUYCDAV-UHFFFAOYSA-N 0.000 description 1
- 229910000105 potassium hydride Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000035935 pregnancy Effects 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 229930010796 primary metabolite Natural products 0.000 description 1
- 239000000583 progesterone congener Substances 0.000 description 1
- 108090000468 progesterone receptors Proteins 0.000 description 1
- 229940095055 progestogen systemic hormonal contraceptives Drugs 0.000 description 1
- 230000005522 programmed cell death Effects 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000000541 pulsatile effect Effects 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 150000003235 pyrrolidines Chemical class 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000001453 quaternary ammonium group Chemical group 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000000306 recurrent effect Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000004043 responsiveness Effects 0.000 description 1
- 239000013037 reversible inhibitor Substances 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 1
- 229930000044 secondary metabolite Natural products 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 239000004945 silicone rubber Substances 0.000 description 1
- 210000000329 smooth muscle myocyte Anatomy 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229940001584 sodium metabisulfite Drugs 0.000 description 1
- 235000010262 sodium metabisulphite Nutrition 0.000 description 1
- 235000010288 sodium nitrite Nutrition 0.000 description 1
- 239000008279 sol Substances 0.000 description 1
- 239000007962 solid dispersion Substances 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 239000008137 solubility enhancer Substances 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 230000009870 specific binding Effects 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 208000000995 spontaneous abortion Diseases 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 102000005969 steroid hormone receptors Human genes 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 238000005556 structure-activity relationship Methods 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 238000007910 systemic administration Methods 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 229960001603 tamoxifen Drugs 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 230000002381 testicular Effects 0.000 description 1
- WHRNULOCNSKMGB-UHFFFAOYSA-N tetrahydrofuran thf Chemical compound C1CCOC1.C1CCOC1 WHRNULOCNSKMGB-UHFFFAOYSA-N 0.000 description 1
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 1
- 125000003507 tetrahydrothiofenyl group Chemical group 0.000 description 1
- OSBSFAARYOCBHB-UHFFFAOYSA-N tetrapropylammonium Chemical compound CCC[N+](CCC)(CCC)CCC OSBSFAARYOCBHB-UHFFFAOYSA-N 0.000 description 1
- PHCBRBWANGJMHS-UHFFFAOYSA-J tetrasodium;disulfate Chemical compound [Na+].[Na+].[Na+].[Na+].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O PHCBRBWANGJMHS-UHFFFAOYSA-J 0.000 description 1
- 125000001984 thiazolidinyl group Chemical group 0.000 description 1
- 229940100615 topical ointment Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- 229940086542 triethylamine Drugs 0.000 description 1
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 description 1
- 229910052722 tritium Inorganic materials 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 1
- 241000701447 unidentified baculovirus Species 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 206010046459 urethral obstruction Diseases 0.000 description 1
- 210000001215 vagina Anatomy 0.000 description 1
- 208000013464 vaginal disease Diseases 0.000 description 1
- 239000006213 vaginal ring Substances 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- 239000003039 volatile agent Substances 0.000 description 1
- 239000000341 volatile oil Substances 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 238000010626 work up procedure Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J43/00—Normal steroids having a nitrogen-containing hetero ring spiro-condensed or not condensed with the cyclopenta(a)hydrophenanthrene skeleton
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- the present invention relates to novel estratrien-triazole derivatives which represent inhibitory compounds of the 17 ⁇ -hydroxysteroid dehydrogenase enzymes, preferably of the 17 ⁇ -hydroxysteroid dehydrogenase type 1 (17 ⁇ -HSD1), type 2 (17 ⁇ -HSD2) or type 3 (17 ⁇ -HSD3) enzyme, as well as to salts of these compounds, to pharmaceutical preparations containing these compounds and to processes for the preparation of these compounds.
- the invention concerns the therapeutic use of said estratrien-triazole derivatives, particularly their use in the treatment or prevention of steroid hormone dependent diseases or disorders, such as steroid hormone dependent diseases or disorders requiring the inhibition of 17 ⁇ -hydroxysteroid dehydrogenase enzymes, in particular 17 ⁇ -HSD type I enzymes, and/or requiring the modulation of the endogenous 17 ⁇ -estradiol and/or testosterone concentration.
- steroid hormone dependent diseases or disorders such as steroid hormone dependent diseases or disorders requiring the inhibition of 17 ⁇ -hydroxysteroid dehydrogenase enzymes, in particular 17 ⁇ -HSD type I enzymes, and/or requiring the modulation of the endogenous 17 ⁇ -estradiol and/or testosterone concentration.
- 17 ⁇ -HSDs Mammalian 17 ⁇ -hydroxysteroid dehydrogenases
- NAD(H) or NADP(H) dependent enzymes which catalyze the final steps in male and female sex hormone biosynthesis.
- These enzymes convert inactive 17-keto-steroids into their active 17 ⁇ -hydroxy-forms or catalyze the oxidation of the 17 ⁇ -hydroxy-forms into the 17-keto-steroids.
- estrogens and androgens have the highest affinity for their receptors in the respective 17 ⁇ -hydroxy form, 17 ⁇ -HSD enzymes play an essential role in the tissue-selective regulation of the activity of sex steroid hormones.
- 17 ⁇ -HSD enzyme family 10 human members of the 17 ⁇ -HSD enzyme family have been described (types 1-5, 7, 8, 10, 11 and 12).
- the human 17 ⁇ -HSD family members share less than 30% similarity in their primary structure.
- the 17 ⁇ -HSDs are expressed in distinct, though in some cases, overlapping patterns.
- the different types of 17 ⁇ -HSDs also differ in their substrate and cofactor specificities.
- the 17 ⁇ -HSDs catalyze the reaction in a unidirectional way: types 1, 3, 5 and 7 use NADP(H) as a cofactor and catalyze the reductive reaction (activation), while types 2, 4, 8 and 10 catalyze the oxidative reaction (inactivation) using NAD(H) as a cofactor [see e.g. Labrie et al. (2000) “Role of 17 beta-hydroxysteroid dehydrogenases in sex steroid formation in peripheral intracrine tissues” Trends Endocrinol Metab., 11:421-7].
- 17 ⁇ -HSDs Due to their essential role in the tissue-selective regulation of the activity of sex steroid hormones 17 ⁇ -HSDs can be involved in the occurrence and development of estrogen-sensitive pathologies (f. ex. breast, ovarian, uterine and endometrium cancers etc.) and androgen-sensitive pathologies (f. ex. prostate cancer, benign prostatic hyperplasia, acne, hirsutism, etc). Furthermore, many types of 17 ⁇ -HSD have been shown to be involved in the pathogenesis of particular human disorders.
- 17 ⁇ -HSD3 is known to be involved in the development of pseudohermaphroditism
- the 17 ⁇ -HSD8 plays a role in polycystic kidney disease
- the 17 ⁇ -HSD4 is related to the occurrence of bifunctional enzyme deficiency. Therefore treatment of sex steroid-sensitive diseases by administration of specific inhibitors of the 17 ⁇ -HSDs enzymes have been suggested, optionally in combination with potent and specific antiestrogens and antiandrogens [Labrie F et al. (1997) “The key role of 17 beta-hydroxysteroid dehydrogenases in sex steroid biology.” Steroids, 62:148-58].
- each type of 17 ⁇ -HSD has a selective substrate affinity, directional (reductive or oxidative) activity in intact cells, and a particular tissue distribution
- the selectivity of drug action could be achieved by targeting a particular 17 ⁇ -HSD isozyme.
- individual modulation of the particular 17 ⁇ -HSDs it is possible to influence or even control the local and paracrine concentration of estrogens and androgens in different target tissues.
- the best characterized member of the 17 ⁇ -HSD family is the type 1 17 ⁇ -HSD [EC 1.1.1.62].
- This enzyme could be crystallized in different states of functionality (e.g. with and without ligand and/or co-factor).
- the 17 ⁇ -HSD1 catalyzes in vitro the reduction as well as the oxidation between estrone (E1) and estradiol (E2). However, under physiological in vivo conditions the enzyme only catalyzes the reductive reaction from the estrone (E1) to the estradiol (E2).
- the 17 ⁇ -HSD1 was found to be expressed in a variety of hormone-dependent tissues, e.g. placenta, mammary gland tissue or uterus and endometrium tissue, respectively.
- Estradiol itself is, especially in comparison to the significantly less active estrone, a very potent hormone, which regulates the expression of a variety of genes by binding to the nuclear estrogen receptor and plays an essential role in the proliferation and differentiation of the target cell.
- Physiological as well as pathological cell proliferations can be estradiol dependent.
- breast cancer cells are stimulated by a locally raised estradiol concentration.
- the occurrence or course of benign pathologies such as endometriosis, uterine leiomyomas (fibroids or myomas), adenomyosis, menorrhagia, metrorrhagia and dysmenorrhea is dependent from the existence of significantly high estradiol levels.
- Endometriosis is a well-known gynaecological disorder that affects 10 to 15% of women in the reproductive age. It is a benign disease defined as the presence of viable endometrial gland and stroma cells outside the uterine cavity. It is most frequently found in the pelvic area. In women developing endometriosis, the endometrial cells entering the peritoneal cavity by retrograde menstruation (the most likely mechanism) have the capacity to adhere to and invade the peritoneal lining, and are then able to implant and grow. The implants respond to steroid hormones of the menstrual cycle in a similar way as the endometrium in the uterus.
- the infiltrating lesions and the blood from these lesions which are unable to leave the body cause inflammation of the surrounding tissue.
- the most common symptoms of endometriosis are primary or acquired dysmenorrhoea, dyspareunia and (chronic) pelvic pain, especially before and in the menstruation period. Further symptoms could include dysuria, various genitourinary symptoms secondary to urethral obstruction and/or bladder invasion, painful defecation, rectal pressure, defecation urgency and bowel obstruction, bleeding abnormalities, including menorrhagia or metrorrhagia, infertility, primary or secondary, recurrent spontaneous abortions. The occurrence of these symptoms is not related to the extent of the lesions.
- Moderate to severe endometriosis can cause tubal damage and adhesions leading to infertility.
- the aims of treatment of endometriosis are pain relief, resolution of the endometriotic tissue and restoration of fertility (if desired).
- the two common treatments are surgery or anti-inflammatory and/or hormonal therapy or a combination thereof.
- Uterine leiomyomas fibroids or myomas
- myomas benign clonal tumours
- They are clinically apparent in up to 25% of women and are the single, most common indication for hysterectomy. They cause significant morbidity, including prolonged and heavy menstrual bleeding, pelvic pressure and pain, urinary problems, and, in rare cases, reproductive dysfunction.
- the pathophysiology of myomas is not well understood. Myomas are found submucosally (beneath the endometrium), intramurally (within the myometrium) and subserosally (projecting out of the serosal compartment of the uterus), but mostly are mixed forms of these 3 different types.
- medical therapies that have been proposed to treat myomas include administration of a variety of steroids such as the androgenic steroids danazol or gestrinone, GnRH agonists and progestogens, whereby the administration is often associated a variety of serious side-effects.
- Dysfunctional uterine bleeding disorders are forms of pathological bleeding that are not attributable to organic changes in the uterus (such as, e.g., endometrial carcinoma, myomas, polyps, etc.), systemic coagulation disorders, or a pathological pregnancy (e.g., ectopic pregnancy, impending abortion) [American College of Obstetricians and Gynecologists, 1982].
- the average blood loss during normal menstruation is about 30 ml, whereby the period lasts for an average of 5 days. If the blood loss exceeds 80 ml, it is classified as pathological.
- Metrorrhagias are defined as bleeding that may or may not be accompanied by pain and that cannot be linked to menstruation or cycle. If it lasts over 7 days, the blood loss often exceeds 80 ml. Menorrhagia is menstruation that may or may not be accompanied by pain, normally every 27-28 days, which, when it lasts over 7 days, is associated in most cases with an increased blood loss of over 80 ml. Menorrhagia is a syndrome of unknown origin and one of the most common problems in gynecology. 60% of women refereed with menorrhagia have a hysterectomy within five years.
- Hypermenorrhea is defined as menstruation that may or may not be accompanied by pain, normally every 27-28 days for 4-5 days with an elevated blood loss of over 80 ml, sometimes even defined as associated with an increased blood loss of over 150 ml.
- Forms of dysfunctional uterine bleeding are typical of adolescence and of the time of menopause, in which follicle-stimulating disorders, anovulation, and yellow-body and follicle persistence occur in clusters.
- the incidence of dysfunctional uterine bleeding is high and represents one of the most frequent reasons for gynecological consultation for women of reproductive age.
- the reversible or even irreversible inhibitors of the 17 ⁇ -HSD1 should have no or only pure antagonistic binding activities to the estradiol receptor, in particular to the estrogen receptor ⁇ subtype, since agonistic binding of the estrogen receptor would lead to activation and therefore—by regulation of a variety of genes—to the proliferation and differentiation of the target cell.
- antagonists of the estrogen receptor so called anti-estrogens, bind competitively to the specific receptor protein thus preventing access of endogenous estrogens to their specific binding site.
- a selective 17 ⁇ -HSD1 inhibitor may be useful for the prevention of the aforementioned hormone-dependent cancers, especially breast cancer (see e.g. WO 2004/080271 (and related 2006/0057628).
- Tremblay and Poirier describe an estradiol derivative, 16-[carbamoyl-(bromo-methyl)-alkyl]-estradiol, and tested the same in respect of its inhibition of the estradiol formation catalysed by the enzyme 17 ⁇ -HSD1 [Tremblay & Poirier (1998) “Overview of a Rational Approach to Design Type I 17 ⁇ -Hydroxysteroid Dehydrogenase Inhibitors Without Estrogenic Activity: Chemical Synthesis and Biological Evaluation”, J. Steroid Biochem. Molec. Biol., 66:179-191].
- Pelletier and Poirier describe novel 17 ⁇ -estradiol derivatives with different bromo-alkyl side chains, which might be potential inhibitors of the 17 ⁇ -HSD1 enzyme [Pelletier & Poirier (1996) “Synthesis and evaluation of estradiol derivatives with 16 ⁇ -(bromoalkylamide), 16 ⁇ -(bromoalkyl) or 16 ⁇ -(bromoalkynyl) side chain as inhibitors of 17 ⁇ -hydroxysteroid dehydrogenase type 1 without estrogenic activity” Bioorg Med Chem, 4(10):1617-1628].
- the related international patent application WO 2004/085345 discloses 15 ⁇ substituted estradiol compounds bearing a —(CH 2 ) m —CO—O—R side chain, wherein R is H, a C 1 -C 5 alkyl group, optionally substituted with at least one halogen group, such as CH 2 CH 2 F, or other group (e.g. CH 2 CHF 2 , CH 2 CF 3 or CF 3 group); and m is from 0-5.
- R is H, a C 1 -C 5 alkyl group, optionally substituted with at least one halogen group, such as CH 2 CH 2 F, or other group (e.g. CH 2 CHF 2 , CH 2 CF 3 or CF 3 group); and m is from 0-5.
- 17 ⁇ -HSD3 A further well characterized member of the 17 ⁇ -HSD family is the 17 ⁇ -HSD type 3 enzyme (17 ⁇ -HSD3).
- the 17 ⁇ -HSD3 has a distinct feature compared to other 17-HSDs: it is found to be expressed almost exclusively the testis, whereas the other isoenzymes are expressed more widely in several tissues.
- 17 ⁇ -HSD3 has a crucial role in androgen biosynthesis. It converts 4-androstene-3,17-one (A) to testosterone (T).
- A 4-androstene-3,17-one
- T testosterone
- the biological significance of the 17 ⁇ -HSD3 is of undeniable physiological importance.
- prostate cancer the primary cancer cells mostly retain their responsiveness to androgens in their regulation of proliferation, differentiation, and programmed cell death for some period.
- androgen deprivation is the only effective systemic hormonal therapy available for prostate cancer.
- the development of selective inhibitors against 17 ⁇ -HSD3 is a new therapeutic approach for the treatment of androgen dependent disease [Labrie et al. (2000)].
- These diseases include but are not limited to prostadynia, benign prostatic hyperplasia, prostatitis, acne, seborrhea, hirsutism, androgenic alopecia, precocious puberty, adrenal hyperplasia, and polycystic ovarian syndrome. Furthermore, considering the fact that 17 ⁇ -HSD3 is found mainly in the testis, the development of potent inhibitors could be of interest for blocking spermatogenesis and as an anti-fertility agent for males.
- WO 01/42181 discloses benzyl-tetralins, the chemical structure of which is related to that of the phytoestrogen biochanin, as 17 ⁇ -HSD3 inhibitors.
- international patent applications WO 99/46279 (and related U.S. Pat. No. 6,541,463), WO 2003/022835 (and related US 2006/069103), WO 2003/033487 (and related US 2003/0232837), WO 2004/046111 (and related U.S. Pat. No. 7,074,795 and US 2006/0148816), WO 2004/060488 (and related U.S. Pat. No.
- 17 ⁇ -HSD type 2 Microsomal 17 ⁇ -hydroxysteroid dehydrogenase of human endometrium and placenta (designated 17 ⁇ -HSD type 2 or 17 ⁇ -HSD2) was cloned by expression cloning, and found to be equally active using androgens and estrogens as substrates for oxidation [Andersson S. (1995) “Molecular genetics of androgenic 17 ⁇ -Hydroxysteroid Dehydrogenases.” J. Steroid Biochem. Molec. Biol., 55:533-534]].
- the recombinant 17 ⁇ -HSD2 converts the highly active 17 ⁇ -hydroxysteroids such as estradiol (E2), testosterone (T), and dehydrotestosterone (DHT) to their inactive keto forms.
- E2 estradiol
- T testosterone
- DHT dehydrotestosterone
- 17 ⁇ -HSD2 can, to a lesser extent, also convert 20 ⁇ -hydroxyprogesterone (20 ⁇ P) to progesterone (P).
- 20 ⁇ -hydroxyprogesterone (20 ⁇ P) to progesterone (P).
- P progesterone
- the broad tissue distribution together with the predominant oxidative activity of 17 ⁇ -HSD2 suggest that the enzyme may play an essential role in the inactivation of highly active 17 ⁇ -hydroxysteroids, resulting in diminished sex hormone action in target tissues.
- Dong and colleagues showed significant 17 ⁇ -HSD2 activity in cultured human osteoblasts and osteoblast-like osteosarcoma cells MG63 and TE85, but not in SaOS-2 [Dong Y et al. (1998) “17 ⁇ -hydroxysteroid dehydrogenases in human bone cells” J. Bone Min. Res., 13:1539-1546].
- E1 to E2, T to A, and DHT to A by bone cells could therefore represent important mechanism for the local regulation of intracellular ligand supply for the estrogen and androgen receptors in the osteoblasts and other steroid sensitive cells.
- This modulation of steroid levels may be employed for a wide variety of indications, including the following: for the prevention and treatment of osteoporosis, for the treatment of ovarian cancer, breast cancer or endometrial cancer, for the treatment of endometriosis, for the treatment of prostate cancer and/or for the treatment of androgen-dependent hair-loss.
- one object of the present invention is to provide novel inhibitors of the 17 ⁇ -HSD1 and/or 17 ⁇ -HSD2 enzyme, which have valuable pharmacological properties and which are suited for the treatment of estrogen dependent diseases and disorders. It is a further object of the present invention to develop novel inhibitors of the 17 ⁇ -HSD3 enzyme, which have valuable pharmacological properties and which are suited for the treatment of androgen dependent diseases and disorders.
- estratrien-triazole derivatives as described herein would be valuable in therapy, especially in the treatment or prevention of steroid hormone dependent diseases or disorders, such as steroid hormone dependent diseases or disorders requiring the inhibition of 17 ⁇ -hydroxysteroid dehydrogenase (HSD) enzymes.
- HSD 17 ⁇ -hydroxysteroid dehydrogenase
- compounds of formula I represent potent inhibitors of the 17 ⁇ -HSD1, 17 ⁇ -HSD3 and/or 17 ⁇ -HSD2 enzyme and possess valuable pharmacological properties for the treatment and/or prophylaxis of malignant steroid dependent diseases or disorders such as breast cancer, ovarian cancer, uterine cancer, prostate cancer, endometrial cancer and endometrial hyperplasia, but also for the treatment and/or prophylaxis of benign steroid dependent diseases or disorders such as endometriosis, uterine fibroids, uterine leiomyoma, adenomyosis, dysmenorrhea, menorrhagia, metrorrhagia, urinary dysfunction, prostadynia, benign prostatic hyperplasia, prostatitis, acne, seborrhea, hirsutism, androgenic alopecia, precocious puberty, adrenal hyperplasia, polycystic ovarian syndrome and/or lower urinary tract syndrome
- estrogen-dependent diseases which may be treated and/or prevented with an effective amount of a compound of the invention are osteoporosis, multiple sclerosis, rheumatoid arthritis, systemic lupus erythematosus, myastenia gravis, thyroiditis, vasculitis, ulcerative colitis, Crohn's disease, graft versus host and host versus graft disease (organ rejection following transplantation), type I and II diabetes, asthma, squamous cell carcinoma, colon cancer, cognitive dysfunctions, senile dementia, Alzheimer's disease, psoriasis, contact dermatitis, eczema, tissue wounds, skin wrinkles and/or cataracts.
- compounds of formula I may be useful for the prevention and treatment of osteoporosis, and for blocking spermatogenesis and as an anti-fertility agent for males.
- the present invention relates to the use of a compound having the structural formula I
- A represents N and B represents C, or A represents C and B represents N n represents 1, 2, 3, 4, 5 or 6 X, Y individually represent F, or X and Y together represent ⁇ O R 1 is selected from the group consisting of:
- Physiologically compatible salts as well as all tautomers, stereoisomers, racemates, enantiomers of the compounds of the invention and mixtures thereof, unless the formula depicting the compound explicitly shows a particular stereochemistry, are also within the scope of the invention.
- Such isomers can be isolated by standard resolution techniques, including fractional crystallization and chiral column chromatography.
- the compounds of the invention also include isotopically-labeled and radio-labeled compounds, as well as commonly used pro-drugs and active metabolites of these compounds
- the present invention relates to a compound, wherein X and Y individually represent F and which therefore has the following formula
- the present invention relates to a compound, wherein X and Y together represent ⁇ O and which therefore has the following formula
- the present invention relates to a compound of the general formula I, wherein A represents N and B represents C and which compound has the formula (Ix),
- the present invention relates to a compound of the general formula I, wherein A represents C and B represents N and which compound has the formula (Iy).
- the present invention relates to a compound of the general formula I, which is an optically pure 15 ⁇ enantiomer having the formula (Ia)
- R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof.
- the present invention relates to the 15 ⁇ enantiomer having formula (Ia), wherein n represents 2, 3, 4, 5 or 6.
- the present invention relates to a compound of the general formula I, which is an optically pure 15 ⁇ enantiomer having the formula (Ib)
- R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof.
- the present invention relates to the 15 ⁇ enantiomer having formula (Ib), wherein n represents 3.
- the present invention relates to a compound of the general formula I, which is an optically pure 15 ⁇ enantiomer having the formula (Ic)
- R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof.
- the present invention relates to the 15 ⁇ enantiomer having formula (Ib), wherein n represents 3.
- the present invention relates to a compound of the general formula I, which is an optically pure 15 ⁇ enantiomer having the formula (Id)
- R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof.
- the present invention relates to the 15 ⁇ enantiomer having formula (Ib), wherein n represents 3 or 4.
- the invention relates to compounds of formula I which are selected from the group consisting of
- the invention relates to a process to prepare the compounds of the invention of formula I
- A represents N and B represents C n represents 1, 2, 3, 4, 5 or 6 X, Y individually represent F, or X and Y together represent ⁇ O R 1 is selected from the group consisting of:
- R 2 has the meanings as defined above, wherein different copper sources are used, selected from the group consisting of copper sources wherein copper has the oxidation states 0, I or II, and wherein the protecting group is replaced after the coupling reaction by R 1 , which has the meaning as defined above.
- the invention relates to a process to prepare the compounds of the invention of formula (Ic)
- n 1, 2, 3, 4, 5 or 6
- X, Y individually represent F, or X and Y together represent ⁇ O R 1 is selected from the group consisting of:
- R 7 and n have the meanings as defined above and PG is a common protecting group, is reacted by a Cu (I)-catalyzed coupling in the presence of an azide (e.g. NaN3) with a halide of formula B,
- R 2 has the meanings as defined above, wherein a modification of the C 17 keto group affords compounds of formula (Ic) with X, Y ⁇ F, and wherein the protecting group is replaced after the coupling reaction by R 1 , which has the meaning as defined above.
- the invention relates to a process to prepare the compounds of the invention of formula (Id)
- n 3, or 4 X, Y individually represent F, or X and Y together represent ⁇ O R 1 is selected from the group consisting of:
- R 7 has the meanings as defined above and PG is a common protecting group, is reacted with a triazole allyl compound of formula C,
- R 2 has the meanings as defined above, wherein a modification of the C 17 keto group affords compounds of formula (Id) with X, Y ⁇ F, and wherein the protecting group is replaced after the coupling reaction by R 1 , which has the meaning as defined above.
- the invention relates to a compound of the invention for use as a medicament.
- compositions containing a pharmacologically active quantity of a compound of formula (I) as defined herewithin and conventional auxiliaries and/or carriers is described.
- the invention concerns the use of a compound of formula (I), as defined herein, for the treatment or prevention of a steroid hormone dependent disease or disorder in a mammal, in particular a human.
- the invention relates to the use of a compound of the invention for the manufacture of a medicament for the treatment or prevention of a steroid hormone dependent disease or disorder in a mammal, in particular a human.
- the steroid hormone dependent disease or disorder is a disease or disorder requiring the inhibition of a 17 ⁇ -hydroxysteroid dehydrogenase enzyme, preferably of the 17 ⁇ -HSD type 1, 17 ⁇ -HSD type 2 or 17 ⁇ -HSD type 3.
- the steroid hormone dependent disease or disorder is an estradiol dependent disease or disorder.
- the steroid dependent disease or disorder is an androgen-dependent disease or disorder.
- the invention also relates to a method of treating a mammal such as a human having a condition related to 17 ⁇ -hydroxysteroid dehydrogenase enzyme activity, preferably 17 ⁇ -HSD1, 17 ⁇ -HSD2 or 17 ⁇ -HSD3 activitym, or which condition can be treated by inhibition of one of said enzymes, comprising administering to the mammal an amount of a compound of this invention, or a salt or a prodrug thereof, which amount is effective to treat the condition.
- Administration of compounds of this invention in combination with other pharmaceuticals used in treatment of the listed conditions is contemplated.
- the conditions to be treated include but are not limited to malign estradiol dependent diseases or disorders such as breast cancer, ovarian cancer, uterine cancer, endometrial cancer, and endometrial hyperplasia.
- the malign disease or disorder is characterized by a detectable level of 17 ⁇ -HSD1 expression within a cancer tissue sample.
- a detectable level of 17 ⁇ -HSD1 expression means that a certain level of 17 ⁇ -HSD1 mRNA or of 17 ⁇ -HSD1 protein can be detected by conventional molecular biology methods such as hybridization, PCR reactions, Northern or Western Blotting etc.
- An alternative detection method for 17 ⁇ -HSD1 expression is the measurement of the corresponding enzyme activity.
- the estradiol dependent disease is breast cancer and the mammal is a human post-menopausal female.
- the conditions to be treated include but are not limited to benign estradiol dependent diseases or disorders such as endometriosis, uterine fibroids, uterine leiomyoma, adenomyosis, dysmenorrhea, menorrhagia, metrorrhagia, and urinary dysfunction.
- benign estradiol dependent diseases or disorders such as endometriosis, uterine fibroids, uterine leiomyoma, adenomyosis, dysmenorrhea, menorrhagia, metrorrhagia, and urinary dysfunction.
- the invention relates to use of an effective amount of a compound of the invention for the treatment or prevention of one of the aforementioned benign gynaecological diseases or disorders in a mammal whereby the mammal is a human, preferably a female and most preferably a pre- or peri-menopausal female.
- the steroid hormone dependent disease or disorder is an androgen-dependent disease or disorder.
- said androgen-dependent disease or disorder is selected from the group consisting of prostate cancer, prostadynia, benign prostatic hyperplasia, urinary dysfunction, lower urinary tract syndrome, acne, seborrhea, androgenetic alopecia, hirsutism, precocious puberty, adrenal hyperplasia, polycystic ovarian syndrome and prostatitis.
- the steroid hormone dependent disease or disorder to be treated is an estrogen- or androgen dependent disease or disorder requiring the lowering of the endogeneous estrogen or androgen concentration in a generalized or tissue-specific manner.
- steroid-dependent diseases which may be treated with an effective amount of a compound of the invention are selected from the group consisting of squamous cell carcinoma, colon cancer, osteoporosis, rheumatoid arthritis, type I and II diabetes, systemic lupus erythematosus, multiple sclerosis, myastenia gravis, thyroiditis, vasculitis, ulcerative colitis, Crohn's disease, psoriasis, contact dermatitis, graft versus host and host versus graft disease (organ rejection following transplantation), eczema, asthma, tissue wounds, skin wrinkles and cataracts.
- the compounds of the invention might be useful for blocking spermatogenesis and as an anti-fertility agent for males.
- a compound of the present invention may be used for the enhancement of cognitive function, i.e. in the treatment or prevention of cognitive dysfunctions, such as senile dementia, including Alzheimer's disease.
- the disclosed compounds are also useful as diagnostic agents (e.g. in diagnostic kits or for use in clinical laboratories) for screening for the presence or absence of 17 ⁇ -HSD1, 17 ⁇ -HSD2 and/or 17 ⁇ -HSD3 enzyme activity.
- the compounds of the present invention can act as selective 17 ⁇ -HSD1, 17 ⁇ -HSD2 or 17 ⁇ -HSD3 inhibitors.
- Another advantage of the compounds of the present invention is that they may be potent in vivo and suited for the therapeutic use in mammals, especially humans.
- Some of the compounds of the present invention may be non-estrogenic compounds.
- the term “non-estrogenic” means exhibiting no or substantially no estrogenic activity on the estrogen receptor.
- Another advantage is that some of the compounds may not be capable of being metabolised to compounds which display or induce hormonal activity.
- Some of the compounds of the present invention are also advantageous in that they may be orally active.
- compound shall here be understood to cover any and all isomers (e.g., enantiomers, stereoisomers, diastereomers, rotomers, and tautomers), racemates or any mixture of isomers, prodrugs, and any pharmaceutically acceptable salt of said compound, unless the formula depicting the compound explicitly shows a particular stereochemistry.
- 17 ⁇ -hydroxysteroid dehydrogenase type I or “17 ⁇ -HSD1” for short is used for the enzyme EC 1.1.1.62 and reduces estrone (E1) to the biologically active estrogen, estradiol (E2).
- inhibitor and “inhibition” include the meaning of to reduce and/or eliminate and/or mask and/or prevent a certain enzyme action.
- 17 ⁇ -HSD1 inhibitor as used herein with respect to the compound of the present invention means a compound that can inhibit 17 ⁇ -HSD1 activity, such as to reduce and/or eliminate and/or mask and/or prevent the action of 17 ⁇ -HSD1.
- the 17 ⁇ -HSD1 inhibitor may act as a reversible or irreversible inhibitor of 17 ⁇ -HSD1.
- the ability of compounds to inhibit 17 ⁇ -HSD1 activity can be assessed using cell lines recombinantly expressing the human 17 ⁇ -HSD1 enzyme. Details on a suitable Assay Protocol are presented in the Examples section. It is to be noted that the compound of the present invention may have other beneficial properties in addition to or in the alternative to its ability to inhibit 17 ⁇ -HSD1 activity; in particular a 17 ⁇ -HSD1 inhibitor may have antagonistic activity towards the nuclear estrogen receptor.
- selective and “selectivity” as used herein with respect to the compounds of the present invention means a compound that can inhibit 17 ⁇ -HSD1, 17 ⁇ -HSD2 and/or 17 ⁇ -HSD3 activity, and shows a higher inhibition value for these particular targets than with regard to other enzyme targets, in particular with regard to the 17 ⁇ -HSD1 enzyme, and that has weak or no affinity for nuclear receptors, in particular that has weak or no affinity for the ER.
- a compound of the present invention has at least about a 100 fold selectivity to a desired target (e.g.
- 17 ⁇ -HSD1 17 ⁇ -HSD1
- a 150 fold selectivity to the desired target preferably at least about a 200 fold selectivity to the desired target, preferably at least about a 250 fold selectivity to the desired target, preferably at least about a 300 fold selectivity to the desired target, preferably at least about a 350 fold selectivity to the desired target.
- substituted means that the specified group or moiety bears one or more substituents. Where any group may carry multiple substituents and a variety of possible substituents is provided, the substituents are independently selected and need not be the same.
- unsubstituted means that the specified group bears no substituents.
- optionally substituted means that the specified group is unsubstituted or substituted by one or more substituents.
- Any asymmetric carbon atoms may be present in the (R)-, (S)- or (R,S)-configuration preferably in the (R)- or (S)-configuration, whichever is most active, unless the stereochemistry is explicitly depicted in the corresponding compound formula.
- the compounds of formula I have a defined stereochemistry within the steroidal core structure according to the natural configuration for estrogenic steroids such as estradiol:
- stereochemistry within the steroidal core structure is always shown in the corresponding compound formula and should not vary within the scope of the present invention, whereas the stereochemistry at the carbon atoms in the steroidal core carrying additional side chains and the stereochemistry of any asymmetric carbon atom within the side chains themselves is not fixed. Therefore, the term “compounds of formula I” or “compounds of formula II” etc also comprises the stereoisomers of the depicted compounds, unless a particular stereochemistry is explicitly shown within the formula. The stereochemistry shown in the respective formula prevails over the general term “stereoisomers”.
- the compounds of the formula I contain at least one additional chiral carbon atom, namely the carbon atom carrying the side chain in the 15-position of the steroid structure.
- the compounds can thus be present at least in two optically active stereoisomeric forms or as a racemate.
- the present invention includes both the racemic mixtures and the isomerically pure compounds of the formula I.
- the position of the substituents within the C15 position is characterized by ⁇ or ⁇ .
- a C15 ⁇ derivative according to the present invention is represented by a compound of the following formulae (Ia) and (Ic)
- the compounds of the present invention may contain further asymmetric centers on the molecule, depending upon the nature of the various substituents. In certain instances, asymmetry may also be present due to restricted rotation about the central bond adjoining the two aromatic rings of the specified compounds. It is intended that all isomers (including enantiomers and diastereomers), either by nature of asymmetric centers or by restricted rotation as described above, as separated, pure or partially purified isomers or racemic mixtures thereof, be included within the ambit of the instant invention, unless a particular stereochemistry is explicitly depicted in the formula representing a respective compound.
- halogen refers to fluorine, bromine, chlorine, and iodine atoms. Preferred in the context of the present invention are F, Cl and Br.
- dihalogen refers to two, three and four halogen substituents, respectively, each individually selected from the group consisting of fluorine, bromine, chlorine, and iodine atoms.
- the carbon content of various hydrocarbon containing moieties is indicated by a prefix designating the minimum and maximum number of carbon atoms in the moiety, i.e., the prefix C i -C j defines the number of carbon atoms present from the integer “i” to the integer “j” inclusive.
- C 1 -C 4 -alkyl refers to alkyl of 1-4 carbon atoms, inclusive, or methyl, ethyl, propyl, butyl and isomeric forms thereof.
- alkyl stands for a hydrocarbon radical which may be linear or branched, with single or multiple branching, whereby the alkyl group comprises the number of carbon atoms as indicated by the prefix.
- the term (C 1 -C 8 )alkyl is exemplified by such groups as methyl; ethyl; n-propyl; isopropyl; n-butyl; sec-butyl; isobutyl; tert-butyl; n-pentyl; isopentyl; neopentyl; tert-pentyl; 2- or 3-methylpentyl; n-hexyl; isohexyl, heptyl, octyl and the like.
- the alkyl or (C 1 -C 8 )alkyl group may be partially unsaturated, forming such groups as, for example, vinyl, propenyl (allyl), butenyl, pentenyl, pentinyl, hexenyl, octadienyl, and the like.
- cyclo(C 3 -C 8 )alkyl comprises cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl and isomeric forms thereof such as methylcyclopropyl; 2- or 3-methylcyclobutyl; 2-, or 3-methylcyclopentyl, and the like.
- the “cycloalkyl” group may also be partly unsaturated, forming such groups as, for example, cyclohexenyl, cyclopentenyl, cyclooctadienyl, and the like.
- cyclo(C 3 -C 8 )alkyl-(C 1 -C 4 )alkyl which refers to a alkyl group of 1 to 4 carbon atoms as described above substituted with a cyclo(C 3 -C 8 )alkyl group as described above, comprises such groups as for example cyclopropylmethyl, cyclohexylmethyl, cyclopentylethyl or cyclohexenylethyl.
- (C 1 -C 4 )alkoxy refers to a group —O—(C 1 -C 4 )alkyl with the (C 1 -C 4 )alkyl group as defined above.
- amino refers to a group —NRR′, wherein R and R′ are residues as particularly defined herein.
- alkanoyl refers to a group —(C ⁇ O)-alkyl.
- alkanoyl is —(C 1 -C 8 )alkanoyl referring to a group —(C ⁇ O)—(C 1 -C 8 )alkyl, with —(C 1 -C 8 )alkyl as defined herewithin.
- More preferred alkanoyl is —(C 1 -C 4 )alkanoyl referring to a group —(C ⁇ O)—(C 1 -C 4 )alkyl, with —(C 1 -C 4 )alkyl as defined herewithin.
- acyl refers to a group —(C ⁇ O)—R, also depicted as —COR, wherein R is a residue as particularly defined herein.
- carboxyl refers to a group —(C ⁇ O)—O—R, also depicted as —COOR, wherein R is a residue as particularly defined herein.
- aryl refers to an aromatic carbocyclic group comprising 6 to 14, more preferably 6 to 10, carbon atoms and having at least one aromatic ring or multiple condensed rings in which at least one ring is aromatic.
- aryl is phenyl, naphthyl, indanyl, indenyl, fluorenyl, 1,2,3,4-tetrahydro-naphthalen-1-yl or even biphenyl. Additionally, the term “aryl” includes benzyl.
- arylalkyl refers to an alkyl group substituted with up to three independently selected aryl groups; preferably the term “arylalkyl” refers to “aryl-(C 1 -C 8 )-alkyl” or aryl-(C 1 -C 4 )-alkyl, whereby the aryl is an aryl group as defined above.
- Aryl-(C 1 -C 4 )alkyl is preferably benzyl (—CH 2 -phenyl) or phenethyl (—CH 2 —CH 2 -phenyl).
- cycloheteroalkyl refers to a four- to eight-membered heterocyclic ring containing at least one heteroatom, such as N, O or S, the number of N atoms being 0, 1, 2 or 3 and the number of O and S atoms each being 0, 1 or 2, which system may be saturated, partly unsaturated or hydroaromatic, and which ring can be part of a multiple condensed ring-system in which some rings may be aromatic.
- cycloheteroalkyls examples include pyrrolidinyl, tetrahydrofuryl, tetrahydrothiophenyl, tetrahydropyridinyl, azetidinyl, thiazolidinyl, oxazolidinyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, azepanyl, diazepanyl, oxazepanyl, thiazepanyl, dihydro-1H-pyrrolyl, 3,6-dihydro-2H-pyridinyl, 1,3-dihydro-benzoimidazolyl and the like.
- cycloheteroalkyl groups are pyrrolidinyl, morpholinyl, tetrahydrofuryl, piperidinyl or azepanyl.
- the cycloheteroalkyl group may optionally be substituted, whereby the substituents may be attached to any carbon or nitrogen atom of the cycloheteroalkyl moiety.
- heteroaryl refers to an aromatic carbocyclic group of having a single 4 to 8 membered ring or multiple condensed rings comprising 6 to 14, more preferably 6 to 10, ring atoms and containing at least one heteroatom, such as N, O or S, within at least one ring, the number of N atoms being 0, 1, 2 or 3 and the number of O and S atoms each being 0, 1 or 2; in which group at least one heterocyclic ring is aromatic.
- Examples of such groups include pyrrolyl, thienyl, furyl, imidazolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, pyrazolyl, pyridinyl, pyrimidinyl, pyrazinyl, pyridazinyl, indolyl, quinolinyl, isoquinolinyl, benzothiazolyl, benzoimidazolyl, 1,3-dihydro-benzoimidazolyl, benzofuran, benzo[b]thiophene and the like.
- heteroaryl is quinolinyl, furyl, benzoimidazolyl, pyridinyl, thienyl, indolyl, benzo[b]thiophene, pyridinyl, imidazolyl, pyrazolyl or thiazolyl.
- pro-drug represents derivatives of the compounds of the invention that are drug precursors which, following administration to a patient by any known route, release the drug in vivo via a chemical or physiological process.
- pro-drug include metabolic precursors.
- Pro-drugs are bioreversible derivatives of drug molecules used to overcome some barriers to the utility of the parent drug molecule. These barriers include, but are not limited to, solubility, permeability, stability, presystemic metabolism and targeting limitations (Medicinal Chemistry: Principles and Practice, 1994, ISBN 0-85186-494-5, Ed.: F. D. King, p. 215; J. Stella, “Pro-drugs as therapeutics”, P.
- pro-drugs are derivatives of the compounds of the invention in which functional groups carry additional substituents which may be cleaved under physiological conditions in vivo and thereby releasing the active principle of the compound (e.g., a pro-drug on being brought to a physiological pH or through an enzyme action is converted to the desired drug form).
- Pro-drugs of the compounds mentioned above are also within the scope of the present invention.
- Pro-drugs that are metabolised to compounds having formula (I) belong to the invention.
- this relates to compounds with primary or secondary amino or hydroxy groups.
- Such compounds can be reacted with organic acids to yield compounds having formula (I) wherein an additional group is present which is easily removed after administration, for instance, but not limited to amidine, enamine, a Mannich base, a hydroxyl-methylene derivative, an O-(acyloxymethylene carbamate) derivative, carbamate, ester, amide or enaminone.
- protecting group and “conventional protecting groups” refer to groups introduced into a molecule by chemical modification of specific functional groups in order to obtain chemoselectivity in certain reactions.
- protecting groups include but are not limited to benzyl, trimethylsilyl, tert-butyloxycarbonyl, 9-fluorenylmethyloxycarbonyl, dimethylacetal.
- physiologically compatible salts refers to salt forms that are physiologically compatible (i.e. pharmacologically acceptable) and substantially non-toxic to the subject being administered the compounds of the invention.
- Physiologically compatible salts of compounds of formula I include conventional and stoichiometrical acid-addition salts or base-addition salts formed from suitable non-toxic organic or inorganic acids or inorganic bases.
- Acid addition salts for example, from compounds of formula I with a basic nitrogen atom are formed preferably with organic or inorganic acids. Suitable inorganic acids are, for example, halogenic acids such as hydrochloric acid, sulfuric acid, or phosphoric acid.
- Suitable organic acids are, for example, carboxylic, phosphonic, or sulfonic acids, for example acetic acid, propionic acid, glycolic acid, lactic acid, hydroxybutyric acid, malic acid, malenic acid, malonic acid, salicylic acid, fumaric acid, succinic acid, adipic acid, tartaric acid, citric acid, glutaric acid, 2- or 3-glycerophosphoric acid and other mineral and carboxylic acids well known to those skilled in the art.
- the salts are prepared by contacting the free base forms with a sufficient amount of the desired acid to produce a salt in the conventional manner.
- Compounds containing acidic substituents may also form salts with inorganic or organic bases.
- suitable bases for salt formation include, but are not limited to, inorganic bases such as alkali or alkaline earth-metal (e.g., sodium, potassium, lithium, calcium, or magnesium) hydroxides, and those derived from ammonium hydroxides (e.g., a quaternary ammonium hydroxide such as tetramethylammonium hydroxide).
- inorganic bases such as alkali or alkaline earth-metal (e.g., sodium, potassium, lithium, calcium, or magnesium) hydroxides, and those derived from ammonium hydroxides (e.g., a quaternary ammonium hydroxide such as tetramethylammonium hydroxide).
- salts formed with pharmaceutical acceptable amines such as ammonia, alkyl amines, hydroxyalkylamines, N-methylglucamine, benzylamines, piperidines, and pyrrolidines and the like.
- Certain compounds will be acidic in nature, e.g. those
- metabolites refers to active compounds derived from catabolism of a compound of formula I upon introduction into a biological milieu, such as a human.
- metabolism includes primary metabolites as well as secondary metabolites of a compound of formula I.
- solvates pertains to the association of suitable organic solvent molecules with molecules or ions of a compound of formula I.
- solvates refers both to stable solvates, containing a defined number of solvent molecules pro molecule of a compound of formula I, and inclusion complexes, which are less stable and contain a variable number of solvent molecules pro molecule of a compound of formula I.
- composition is intended to encompass a product comprising the specified ingredients in the specified amounts, as well as any product which results, directly or indirectly, from combination of the specified ingredients in the specified amounts.
- an effective amount means an amount of a compound or composition which is sufficient enough to significantly and positively modify the symptoms and/or conditions to be treated (e.g., provide a positive clinical response).
- the effective amount of an active ingredient for use in a pharmaceutical composition will vary with the particular condition being treated, the severity of the condition, the duration of the treatment, the nature of concurrent therapy, the particular active ingredient(s) being employed, the particular pharmaceutically acceptable excipient(s)/carrier(s) utilized, and like factors within the knowledge and expertise of the attending physician.
- the invention relates to compounds of formula (I), (Ia), (Ib), (Ic), (Id), (Ix) or (Iy), wherein n represents 2, 3, 4, 5 or 6, preferably n represents 2, 3, 4 or 6, more preferably n represents 3 or 4.
- R 1 is selected from the group consisting of —H, —(C 1 -C 6 )alkyl, -phenyl or —(C 1 -C 4 )alkyl-phenyl, preferably from —H, -methyl or -benzyl.
- the invention discloses compounds of formula, (Ib), (Ic), (Id), (Ix) or (Iy), wherein X and Y individually represent F or X and Y together represent ⁇ O, wherein R 7 is ethyl, methoxy, ethoxy, methoxyethyl, propyl or hydrogen (—H), preferably R 7 is hydrogen.
- R 2 is selected from the group consisting of:
- the residues R 4 and R 5 are each independently selected from the group consisting of H, —(C 1 -C 4 )alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl and aryl-(C 1 -C 4 )alkyl, optionally substituted in the aryl moiety by 1, 2 or 3 halogens, or R 4 and R 5 form together with the nitrogen atom, to which they are attached, a cyclic 6-membered ring system, which is saturated or contains one or more double bonds between the ring atoms, and which ring optionally contains a sulfoxide moiety in addition to the nitrogen atom, and R 6 represents —(C 1 -C 4 )alkyl, which is optionally substituted by 1, 2 or 3 halogens, and/or optionally substituted by 1, 2 or 3 hydroxyl moieties.
- each R 4 and R 5 is independently selected from the group consisting of H, —(C 1 -C 4 )alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by hydroxyl; and phenyl or phenyl-(C 1 -C 2 )alkyl, or R 4 and R 5 form together with the nitrogen atom, to which they are attached, a cyclic 6-membered ring system, which is saturated or contains one or more double bonds between the ring atoms, and which ring optionally contains a sulfoxide moiety in addition to the nitrogen atom, and R 6 represents —(C 1 -C 4 )alkyl, which is optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by hydroxyl.
- the method of the invention is primarily intended for treatment in a mammal, preferably in humans and other primates, of steroid hormone dependent diseases or disorders, in particular estradiol dependent diseases or disorders, wherein the steroid hormone dependent disease or disorder preferably requires the inhibition of a 17 ⁇ -hydroxysteroid dehydrogenase (HSD) enzyme, preferably the type 1 17 ⁇ -hydroxysteroid dehydrogenase (HSD) enzyme [EC 1.1.1.62].
- HSD 17 ⁇ -hydroxysteroid dehydrogenase
- HSD type 1 17 ⁇ -hydroxysteroid dehydrogenase
- the compounds may be administered orally, dermally, parenterally, by injection, by pulmonal or nasal delivery, or sublingually, rectally or vaginally in dosage unit formulations.
- administered by injection includes intravenous, intraarticular, intramuscular (e.g. by depot injection where the active compounds are released slowly into the blood from the depot and carried from there to the target organs), intraperitoneal, intradermal, subcutaneous, and intrathecal injections, as well as use of infusion techniques.
- Dermal administration may include topical application or transdermal administration.
- One or more compounds may be present in association with one or more non-toxic pharmaceutically acceptable auxiliaries such as excipients, adjuvants (e.g.
- buffers such as, but not limited to, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styl, sulfate, styl, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene, styrene,
- the pharmaceutical composition may be formulated for example as immediate release, sustained release, pulsatile release, two or more step release, depot or other kind of release formulations.
- auxiliaries as well as further suitable diluents, flavorings, sweetening agents, coloring agents etc. may be used, depending on the intended mode of administration as well as particular characteristics of the active compound to be used, such as solubility, bioavailability etc.
- Suitable auxiliaries and further ingredients may be such as recommended for pharmacy, cosmetics and related fields and which preferably are listed in the European Pharmacopoeia, FDA approved or cited in the “GRAS” list (FDA List of food additives that are ‘generally recognized as safe’ (GRAS)).
- One mode of application of the compounds of general formula I or of pharmaceutical compositions comprising one or more of said compounds is oral application, e.g., by tablets, pills, dragees, hard and soft gel capsules, granules, pellets, aqueous, lipid, oily or other solutions, emulsions such as oil-in-water emulsions, liposomes, aqueous or oily suspensions, syrups, elixiers, solid emulsions, solid dispersions or dispersible powders.
- the compounds suitable for the purposes of the present invention as defined above can be admixed with commonly known and used adjuvants and excipients such as for example, gum arabic, talcum, starch, sugars (such as, e.g., mannitose, methyl cellulose, lactose), gelatin, surface-active agents, magnesium stearate, aqueous or non-aqueous solvents, paraffin derivatives, cross-linking agents, dispersants, emulsifiers, lubricants, conserving agents, flavoring agents (e.g., ethereal oils), solubility enhancers (e.g., benzyl benzoate or benzyl alcohol) or bioavailability enhancers (e.g. GelucireTM).
- the active ingredients may also be dispersed in a microparticle, e.g. a nanoparticulate, composition.
- the active agents can be dissolved or suspended in a physiologically acceptable diluent, such as, e.g., water, buffer, oils with or without solubilizers, surface-active agents, dispersants or emulsifiers.
- a physiologically acceptable diluent such as, e.g., water, buffer, oils with or without solubilizers, surface-active agents, dispersants or emulsifiers.
- oils for example and without limitation, olive oil, peanut oil, cottonseed oil, soybean oil, castor oil and sesame oil may be used.
- the active agent can be in the form of an aqueous, lipid, oily or other kind of solution or suspension or even administered in the form of liposomes or nano-suspensions.
- Transdermal application can be accomplished by suitable patches, as generally known in the art, specifically designed for the transdermal delivery of active agents, optionally in the presence of specific permeability enhancers. Furthermore, also emulsions, ointments, pastes, creams or gels may be used for transdermal delivery.
- intravaginal devices e.g. vaginal rings
- IUS intrauterine systems
- IUD intrauterine devices
- compositions can be prepared by mixing the drug with a suitable non-irritating excipient which is solid at ordinary temperatures but liquid at the rectal or vaginal temperature and will therefore melt in the rectum or vagina to release the drug.
- a further drug formulation is a formulation intended for the topical, local and/or regional administration of the compound to the reproductive organs, in particular to a body region selected from the group consisting of the uterus, fallopian tubes, peritoneal space, pelvic cul-de-sac, ovaries, and urinogenital tract, in amounts effective to treat various conditions, particularly local diseases of the female reproductive system, such as pelvic, uterine, cervical and vaginal diseases, as described e.g. within EP 0 977 555 A1 (and related U.S. Pat. No. 5,993,856), U.S. Pat. No. 5,993,856, U.S. Pat. No. 6,652,874, or U.S. Pat. No. 6,416,778.
- the formulation comprises drug particles, preferably in the form of a micro- or nano-particles, suitable for regional administration of an effective amount of drug, wherein the effective amount is a dosage which results in low serum drug levels and reduced side effects as compared to systemic administration of the drug.
- the formulation comprises a carrier promoting quick uptake of the drug into the blood stream, a carrier manipulating release of drug, or a carrier promoting adhesion of the drug selected from the group consisting of a liquid suspension or dispersion, a hydrogel suspension or dispersion, a topical ointment, a cream, a lotion, and a foam.
- a depot implant comprising an inert carrier material, such as biologically degradable polymers or synthetic silicones such as e.g. silicone rubber.
- an inert carrier material such as biologically degradable polymers or synthetic silicones such as e.g. silicone rubber.
- Such implants are designed to release the active agent in a controlled manner over an extended period of time (e.g. 3 to 5 years).
- the actually required dosages of the agents of this invention for any given patient will depend upon a variety of factors, including, but not limited to the activity of the specific compound employed, the particular 17beta HSD type 1, type 2 or type 3 related condition being treated, the particular composition formulated, the mode of administration, time and duration of administration, route of administration and the particular site being treated, and furthermore the age of the patient, the body weight of the patient, the general health of the patient, the gender of the patient, the diet of the patient, rate of excretion, drug combinations, and the severity of the condition undergoing therapy.
- the optimal course of treatment i.e., the mode of treatment and the daily number of doses of a compound of formula I or a pharmaceutically acceptable salt thereof given for a defined number of days
- Optimal dosages for a given set of conditions may be ascertained by those skilled in the art using conventional dosage-determination tests in view of the experimental data for a given compound.
- an exemplary daily dose generally employed will be from about 0.001 ⁇ g/kg to about 10 mg/kg of total body weight, whereby courses of treatment may be repeated at appropriate time intervals.
- Administration of pro-drugs may be dosed at weight levels that are chemically equivalent to the weight levels of the fully active compounds.
- the daily dosage for parenteral administration will generally be from about 0.001 ⁇ g/kg to about 10 mg/kg of total body weight.
- a daily rectal dosage regimen will generally be from about 0.001 ⁇ g/kg to about 20 mg/kg of total body weight.
- a daily vaginal dosage regimen will generally be from about 0.001 ⁇ g/kg to about 10 mg/kg of total body weight.
- the daily topical dosage regimen will generally be from about 0.01 ⁇ g to about 10 mg administered between one to four times daily.
- the transdermal concentration will generally be that required to maintain a daily dose of from 0.001 ⁇ g/kg to 10 mg/kg of total body weight.
- the total dosage of administration forms releasing the drug compound over a prolonged period of time depends on the time of administration, on the kind of device (intravaginal devices, intrauterine systems, intrauterine devices, implants etc.) and on the kind of release behaviour of the particular device.
- the daily released dose of active compound will be from about 0.001 ⁇ g/kg to about 1 mg/kg of total body weight. Since the devices often only need to achieve a certain local and/or regional concentration of active compound, the daily released dosage can be lower in comparison to e.g. oral administration.
- the compounds of the present invention may be prepared by use of known chemical reactions and procedures. Nevertheless, the following general preparative methods are presented to aid the reader in synthesizing the 17- ⁇ -hydroxysteroid dehydrogenase inhibitors with specific details provided below in the experimental section to illustrate working examples.
- substituted estratrien derivatives bearing a substituted triazole in position C 15 and carrying optionally additional modifications of the steroidal core at positions C 2 , C 3 and/or C 17 may be introduced in the following order of general chemical modifications.
- a compound of formula (Ix) can be synthesised by the introduction of the R 7 substituent in C 2 position—if present in the final compound—which has to take place first, starting from the 17 ⁇ -estradiol using methods well known in the art (Steps A).
- the C 17 —OH function may be oxidized to the corresponding keto function.
- a suitable group also functioning as protecting group may be introduced at this point.
- the estron derivative is converted into the central intermediate, the 15,16-unsaturated estrone (Steps B), which is further derivated in the C 15 position by introduction of the basic azide side chain to generate the central intermediate (Steps C-I).
- the modification of the C 17 function preferably takes place before finalizing the introduction of the azide side chain (Steps C-II).
- Compounds of the present invention may then be prepared by a process comprising coupling of the obtained azide intermediate with a terminal alkyne H—C ⁇ C—R 2 as depicted below (Step D).
- the C 17 keto function might be protected with conventional protecting groups during this coupling step.
- the protection group in C 1 position may be separated to deliver the C 3 —OH derivative or may be further substituted with an alternative R 1 side chain (Steps E).
- R 1 , R 2 , R 7 , X and Y have the meanings as defined herein, and PG is a common protecting group like benzyl.
- Step A The synthesis of estratrien derivatives with variations at C 2 is described in general and vor exemplary compounds in detail in international patent application WO 2006/032885 (and related US 2006/0128766) and in PCT application WO 2006/125800.
- Step B The preparation of the 15, 16 unsaturated estron derivate is described in detail in PCT application WO 2005/047303 and in PCT application WO 2006/125800.
- Step C-I The synthesis of the C 15 substituted azide derivate might be achieved by conventional synthesis methods as already depicted within international patent application WO 2005/047303 and in PCT application WO 2006/125800 and as depicted herein below.
- Step C-II In addition to the reaction of Step C-I, the difluorination of the C 17 atom of the estron core has to be carried out. This is a reaction well known in the art and was already disclosed in U.S. Pat. No. 3,413,321 and U.S. Pat. No. 3,347,878. Furthermore, the difluorination of the C 17 atom of the estron core may be achieved using the DAST reagent [Liu et al (1992) “Synthesis of high affinity fluorine-substituted ligands for the androgen receptor. Potential agents for imaging prostatic cancer by positron emission tomography.” J Med Chem. 35(11):2113-29].
- Step D The coupling of the azide with a terminal alkinyl delivering the desired triazole derivatives may be carried out by using a method for the formation of 1,4 disubstituted triazoles well known to one skilled in the art of organic synthesis (see e.g. WO 2006/063585 (and related US 2006/128766) and WO 2003/101972 (and related US 2005/0222427) and references cited therein).
- a compound of formula (Ic) can be synthesised by the introduction of the R 7 substituent in C 2 position—if present in the final compound—which has to take place first, starting from the 17 ⁇ -estradiol using methods well known in the art (Steps A).
- the C 17 —OH function may be oxidized to the corresponding keto function.
- a suitable group also functioning as protecting group may be introduced at this point.
- the estron derivative is converted into the central intermediate, the 15,16-unsaturated estrone (Steps B), which is further derivated in the C 15 position by introduction of the corresponding alkine compound in a Grignard reaction (Steps F).
- the compounds of formula (Ic) are then prepared from the corresponding R2 Halides via in situ generated azides in the presence of azides (e.g. NaN3) (Steps G). Then, if desired, the modification of the C 17 function takes place (Steps H). Finally, if desired, the protection group in C 1 position may be separated to deliver the C 3 —OH derivative or may be further substituted with an alternative R 1 side chain (Steps J).
- azides e.g. NaN3
- R 1 , R 2 , R 7 , X and Y have the meanings as defined herein, and PG is a common protecting group like benzyl.
- Step A The synthesis of estratrien derivatives with variations at C 2 is described in general and vor exemplary compounds in detail in international patent application WO 2006/032885 and in PCT application WO 2006/125800.
- Step B The preparation of the 15, 16 unsaturated estron derivate is described in detail in PCT application WO 2005/047303 and in PCT application WO 2006/125800.
- Step F The 15,16-unsaturated estron is coupled with the corresponding alkine compound in a Grignard reaction.
- Step G The Triazoles are prepared from the alkine substituted estron derivate and the corresponding R2 Halides via in situ generated azides.
- the one pot synthesis for specific aryl triazoles is described in detail in Andersen J, Bolvig S, Liang X (2005) “Efficient One-Pot Synthesis of 1-Aryl 1,2,3-Triazoles from Aryl Halides and Terminal Alkynes in the Presence of Sodium Azide” Synlett, 2005, 19:2941.
- Step H Fluorination with Deoxoflur results converts the keto triazoles into the corresponding di-fluoro triazoles
- Step J Deprotection of the oxygen by introduction of R1 group.
- Steps A Compounds of formula (Id) can be synthesised by the introduction of the R 7 substituent in C 2 position—if present in the final compound—which has to take place first, starting from the 17 ⁇ -estradiol using methods well known in the art (Steps A).
- the C 17 —OH function may be oxidized to the corresponding keto function.
- a suitable group also functioning as protecting group may be introduced at this point.
- the estron derivative is converted into the central intermediate, the 15,16-unsaturated estrone (Steps B), which is further derivated in the C 15 position by introduction of the corresponding alkene compound (Steps K).
- Steps N Coupling with the triazole allyl compound synthesised (Steps N) from the corresponding allyl alkine affords the allyl triazoles (Steps M). Reduction of the double bond leads to the triazole compound. Then, if desired, the modification of the C 17 function takes place (Steps H). Finally, if desired, the protection group in C 1 position may be separated to deliver the C 3 —OH derivative or may be further substituted with an alternative R 1 side chain (Steps J).
- R 1 , R 2 , R 7 , X and Y have the meanings as defined herein, and PG is a common protecting group.
- Step A The synthesis of estratrien derivatives with variations at C 2 is described in general and vor exemplary compounds in detail in international patent application WO 2006/032885 and in PCT application WO 2006/125800.
- Step B The preparation of the 15, 16 unsaturated estron derivate is described in detail in PCT application WO 2005/047303 and in PCT application WO 2006/125800.
- Step K The preparation of the 15, 16 unsaturated allyl derivate is described in detail in WO 2006/125800.
- Step L Introduction of the Triazole moiety synthesized from the corresponding allyl alkine (Step N) and chain elongation (e.g. via Metathesis).
- Step M Reduction of the double bond lead to the O-protected Triazoles.
- Step H Fluorination with Deoxoflur results converts the keto triazoles into the corresponding di-fluoro triazoles
- Step J Deprotection of the oxygen by introduction of R1 group.
- the compounds of formula I may be prepared using the reactions and techniques described in this section. The reactions are performed in solvents that are appropriate with respect to the reagents and materials employed and that are suitable for the transformations being effected. Also, in the synthetic methods described below, it is to be understood that all proposed reaction conditions, including choice of solvent, reaction atmosphere, reaction temperature, duration of experiment and work-up procedures, are chosen to be conditions of standard for that reaction, which should be readily recognised by one skilled in the art. It is understood by one skilled in the art of organic synthesis, that the functionality present on various positions of the molecules used as the starting compounds or intermediates in the syntheses, must be compatible with the reagents and reactions proposed. Not all compounds of formula I falling into a given class may be compatible with some of the reaction conditions required in some of the methods described. Such restrictions of substituents or functional groups which are compatible with the reaction conditions will be readily apparent to one skilled in the art and alternative methods can be used.
- Visualization of plates was effected by one or more of the following techniques: (a) ultraviolet illumination (254 nm or 266 nm), (b) exposure to iodine vapor, (c) spraying of the plate with Schlittler's reagent solution followed by heating, (d) spraying of the plate with anisaldehyde solution followed by heating, and/or (e) spraying of the plate with Rauxz reagent solution followed by heating. Column chromatography (flash chromatography) was performed using 230-630 mesh ICN, SiliTech 60A silica gel. 1 H-NMR spectra were measured with a Bruker ARX (400 MHz) or Bruker ADVANCE (500 MHz) spectrometer with the solvent as indicated.
- HPLC electrospray mass spectra were obtained using the following method and equipment: Samples were separated by reversed phase high pressure liquid chromatography (RP-HPLC) coupled to a quadrupol MS. HPLC was performed at a flow of 1000 ul/min using Xter-MS C18 columns (i. d. 4.6 mm, length 50 mm, particle size 2.5 um) or Phenomenex LunaC18 (2) 30*4.6 mm columns.
- LC200 pump PE coupled to anAPI100 MS (Applied Biosystems Sciex), a variable wavelength detector Waters 2487 set to 225 nm, and an ELSD (Sedex 75), ES+.
- spectra were scanned with a scan range of m/z 100 to 800 or 100 to 900.
- Gas chromatography-mass spectra (GC-MS) analyses were performed with an Agilent 6890 gas chromatograph equipped with an DB-5MS column (0.25 i. d., length 30 m) and an Agilent 5973 MSD quadrupol detector (ionization with electron impact (EI) at 70 eV; source temperature 230 C).
- EI electron impact
- estratrien derivatives with variations at C2 is described in detail in PCT application WO 2006/125800.
- estron derivatives carrying an alkyl side chain with an azide group in C15 position as depicted in the following general formula
- Enone (IV) may be further converted into the corresponding allyl compound (V) in a Grignard reaction.
- the alcohol is oxidised to the corresponding ketone (VI), while the allyl group is introduced at position 15 of the estrone.
- the allyl is hydroxylated (VIII) and then mesylated (IX).
- the ketone (XII-I) is deprotected in two steps to obtain the desired azide (XII-I).
- the estrone (IV) may be converted selectively in a single step into the corresponding alkenyl (XIII) via a copper mediated addition.
- the alkenyl is hydroxylated (XVI) and then mesylated (XVII).
- the ketone (XVIII) is deprotected in two steps to obtain the desired azide intermediate (XII-II).
- a flask is charged with LiCl (197 mmol) and CuI (197 mmol) in THF (240 mL) and stirred for 1 h at ambient temperature under a nitrogen atmosphere.
- the dark green solution is cooled to ⁇ 78° C. and a 1 M vinylmagnesium bromide solution in THF (200 mmol) is added dropwise while maintaining the temperature between ⁇ 80 and ⁇ 70° C.
- TMSCl 167 mmol
- the dark brown reaction mixture is stirred for 21 ⁇ 2 h at ⁇ 74° C. After the mixture is allowed to reach a temperature of 15° C., it is quenched with sat. NH 4 Cl (300 mL). The formed suspension is filtered over Celite® and the residue is ished with water and THF. The filtrate is diluted with 1 M HCl (250 mL). The aqueous layer is extracted with EtOAc (3 ⁇ 200 mL). The combined organic layers are ished with 1 M HCl (300 mL). The formed suspension is filtered over Celite® and the organic layer is separated.
- the organic layer is ished with 2 M ammonia (2 ⁇ 300 mL), until the blue color of the water layer had nearly vanished, ished with brine (300 mL), dried over MgSO 4 and concentrated in vacuo leaving a brown solid (25 g).
- the estrone (IV) may be converted in a Grignard reaction into the corresponding alkoxy-THP derivative (XXI). Without protection of the ketone, it is further hydroxylated (XXII) and then mesylated (XVII). After azidation of the mesyl-protected alcohol (XVII), the ketone (XVIII) is deprotected in two steps to obtain the desired azide intermediate (XII-II).
- Certain compounds of formula Ix can be prepared via the following route:
- 1,4 disubstituted triazoles can be synthesized by a copper catalyzed coupling of an azide (XIIa-I, II, III) with a terminal alkyne in the presence of sodium ascorbate as reducing agent and a minor amount of water (see e.g. V. V. Rostovtsev, L. G. Green, V. V. Fokin, K. B. Sharpless, “A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective Ligation of Azides and Terminal Alkynes” Angew. Chem., 2002, 41(14), 2596-2599 and WO 2003/101972).
- a possible alternative is the copper(I) catalysed method at room temperature or higher (e.g. ⁇ 80° C.).
- an amount of Cu-source in the range 0.1 to 10% by mole relative to the amount of the alkyne or azide is used at low temperatures in solvents such as MeOH, EtOH, tert-BuOH 1,4-dioxane, AcN, DMSO, acetone, DMF, NMP or THF.
- solvents such as MeOH, EtOH, tert-BuOH 1,4-dioxane, AcN, DMSO, acetone, DMF, NMP or THF.
- the above coupling may also be performed thermally thus without a catalyst as Cu). However, this process may also
- compounds of formula (Ix) can be prepared via the following route (scheme 5):
- estratrien derivatives with variations at C 2 is described in detail in PCT application WO 2006/125800.
- estrone alkine intermediates with X and Y ⁇ O for the synthesis of formula (Iy) compounds may be prepared via the following route
- the freshly prepared Grignard reagent (XXX) is firstly converted into the cuprate for directing the regioselectivity to the C15 at enone (IV). After deprotection by cleaving the TMS group, the respective alkine (XXXII) is obtained.
- a solution of copper(I)iodide (60 mmol) and lithium chloride (63 mmol) in dry THF (120 mL) is prepared.
- the mixture was cooled to ⁇ 78° C. and a solution of the freshly prepared Grignard reagent (XXX) (60 mmol) in dry THF is added such that temperature was kept below ⁇ 77° C.
- TMSCl (60 mmol) and a solution of 3-benzyldehydroestrone (17 mmol) in dry THF (85 mL) are added slowly.
- the reaction mixture is warmed to room temperature and stirred for 18 h.
- the reaction mixture is then filtered and poured into a sat. aqueous NH 4 Cl solution.
- Certain compounds of formula (Iy) can be prepared from the alkine intermediates via the following route:
- the alkine compound (XXXII) is transferred into the corresponding triazole compound (XXXIII) via an in situ generated azide using trans-N,N-dimethyl-1,2-cyclohexanediamine as ligand for Cu(I). After deprotection, the corresponding triazole (Iy-I) is afforded.
- 2,4-Difluoroaniline (99 mmol) is dissolved in a mixture of trifluoroacetic acid (86 mL) and sulfuric acid (17 mL). The mixture is cooled in an icebath and a solution of sodium nitrite (129 mmol) in water (86 mL) was added slowly, while maintaining T ⁇ 10° C. The mixture is stirred for another 30 min at 0° C. before a solution of sodium azide (11.3 g, 174 mmol) in water (63 mL) is added dropwise [Caution: vigorous evasion of gas]. The temperature is kept below 12° C. during addition. The mixture is stirred at rt overnight and then extracted with MTBE (3 ⁇ ). The combined org.
- ketone triazoles (XXXIII) are fluorinated with Deoxofluor. Deprotection of the oxygen by cleaving the protection group afford triazoles (Iy-III).
- Recombinant baculovirus was generated by the “Bac to Bac Expression System” (Invitrogen). Recombinant bacmid was transfected to Sf9 insect cells using “Cellfectin Reagent” (Invitrogen). 60 h later cells were harvested; the microsomal fraction was isolated as described by Puranen et al. (1994). Aliquots were stored frozen until determination of enzymatic activity.
- Assay Inhibition of recombinant human 17 ⁇ -HSD1: Recombinant protein (0.1 ⁇ g/ml) was incubated in 20 mM KH 2 PO 4 pH 7.4 with 30 nM 3H-estrone and 1 mM NADPH for 30 min at RT, in the presence of potential inhibitors at concentrations of 1 ⁇ M or 0.1 ⁇ M. Inhibitor stock solutions were prepared in DMSO. Final concentration of DMSO was adjusted to 1% in all samples. The enzyme reaction was stopped by addition of 10% trichloroacetic acid (final concentration). Samples were centrifuged in a microtiter plate at 4000 rpm for 10 min.
- % ⁇ ⁇ conversion 100 ⁇ ⁇ ( cpm ⁇ ⁇ estradiol ⁇ ⁇ in ⁇ ⁇ sample ⁇ ⁇ with ⁇ ⁇ inhibitor ) / [ ⁇ cpm ⁇ ⁇ estrone ⁇ ⁇ in ⁇ ⁇ sample ⁇ ⁇ with ⁇ ⁇ inhibitor ) + ( cpm ⁇ ⁇ estradiol ⁇ ⁇ in ⁇ ⁇ sample ⁇ ⁇ with ⁇ ⁇ inhibitor ) ] ⁇ ⁇ ⁇ ( cpm ⁇ ⁇ estradiol ⁇ ⁇ in ⁇ ⁇ sample ⁇ ⁇ without ⁇ ⁇ inhibitor ) / [ ( cpm ⁇ ⁇ estrone ⁇ ⁇ in ⁇ ⁇ sample ⁇ ⁇ without ⁇ ⁇ inhibitor ) + ( cpm ⁇ ⁇ estradiol ⁇ ⁇ in ⁇ ⁇ sample ⁇ ⁇ without ⁇ ⁇ inhibitor ) ] ⁇ .
- the compounds were screened in respect of 17 ⁇ -HSD3 enzyme activity in vitro on established MCF-7 cell lines, each stably expressing the 17 ⁇ -HSD3 enzyme.
- the interconversion of substrate by 17 ⁇ -HSD3 and the 17 ⁇ -HSD3 inhibiting activity of chemical compounds in these cell lines were detected by HPLC system.
- Varying amounts of the test compounds were incubated in the growth medium of the 17 ⁇ -HSD3 expressing cells together tritium labeled androstenedione (2 nM). The medium samples were removed after exact incubation time and the reaction is stopped by trichloroacetic acid (TCA). The samples were analyzed by HPLC-coupled flow scintillation analysis.
- the 17 ⁇ -HSD3 inhibiting activity of an individual test compound was calculated by comparing the conversion of a control sample without any test compound (referred to as “Negative Control”) to the (reduced) conversion of the test sample containing the particular compound to be tested (referred to as “Test Sample”).
- % ⁇ ⁇ inhibition _ 100 ⁇ Conversion ⁇ ⁇ in ⁇ ⁇ Negative ⁇ ⁇ Control - Conversion ⁇ ⁇ in ⁇ ⁇ Test ⁇ ⁇ Sample Conversion ⁇ ⁇ Negative ⁇ ⁇ Control
- the binding affinity of the compounds of the invention to the estrogen receptor ⁇ and to the estrogen receptor ⁇ may be determined according to the in vitro ER binding assays described by Koffmann B et al. (1991) “Evidence for involvement of tyrosine in estradiol binding by rat uterus estrogen receptor.” J. Steroid. Biochem. Mol. Biol. 38(2):135.
- an estrogen receptor binding assay may be performed according to international patent application PCT/US/17799 (published as WO 00/07996 (and related U.S. Pat. No. 6,291,505)).
- Compounds of the invention showing binding affinity towards the estrogen receptor may be further tested with regard to their individual estrogenic or anti-estrogenic potential (agonistic binding or antagonistic binding to the ER ⁇ or ER ⁇ ).
- the determination of the estrogen receptor agonist activity may be performed according to an in vitro assay system using the MMTV-ERE-LUC reporter system which is for example described within U.S. patent application Ser. No. 10/289,079 (published as US 2003/0170292):
- the first plasmid comprises DNA encoding human estrogen receptor (either ER-alpha or ER-beta), and the second plasmid comprises an estrogen-driven reporter system comprising: a luciferase reporter gene (LUC) whose transcription is under the control of upstream regulatory elements comprising 4 copies of the vitellogenin estrogen response element (ERE) cloned into the mouse mammary tumor virus (MMTV) promoter (the full name for the reporter system being “MMTV-ERE-LUC”).
- LEC luciferase reporter gene
- Cells are exposed to the compounds of the invention in RPMI 1640 medium, supplemented with 10% charcoal-treated fetal calf serum, 2 mM L-glutamine, 0.1 mM non-essential amino acids and 1 mM so-diumpyruvate for 42-48 hours at 37° C. in a 5% carbon dioxide incubator. Concurrently, cells exposed to estradiol (1 nM) serve as positive controls. Replicate wells exposed to the solvent in which the compounds of the invention are dissolved (i.e. ethanol or methanol) are used as negative controls.
- estrogenic activity of the compounds of the invention is ex-pressed as fold-increase in luciferase activity as compared to that observed in negative control cells.
- estrogen receptor transactivation activity (estrogenicity assay or agonist assay) and of the inhibitory potency of transactivation activity (anti-estrogenicity assay or antagonist assay) may be performed according to international patent application WO 00/07996 (and related U.S. Pat. No. 6,291,505).
- the compounds of the invention show good inhibitory potential of the 17 ⁇ -HSD1, 17 ⁇ -HSD2 and/or of the 17 ⁇ -HSD3 enzyme.
- the compounds of the invention are therefore regarded as being suited for the treatment of several estrogen and androgen dependent diseases and disorders, respectively.
- a reduction of the endogenous 17 ⁇ -estradiol concentration in the respective tissue will result in an impaired or reduced proliferation of the 17 ⁇ -estradiol dependent cells in said tissues as can be demonstrated by the above described in vivo assays.
- the selective inhibitors of the 17 ⁇ -HSD1 enzyme as described herein are well suited to impair also endogenous productions of estrogens, in particular of 17 ⁇ -estradiol, in myomas, endometriotic, adenomyotic and endometrial tissue.
- the application of a compound acting as selective inhibitor on the 17 ⁇ -HSD1 enzyme, which preferentially catalyzes the reductive reaction will result in a lowered intracellular estradiol-concentration, since the reductive conversion of the estrone into the active estradiol is reduced or suppressed, and will therefore impair or even reduce the proliferation of the 17 ⁇ -estradiol dependent cells in the malignant or benign tissue.
Landscapes
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Urology & Nephrology (AREA)
- Dermatology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
wherein
A represents N and B represents C, or A represents C and B represents N
n represents 1, 2, 3, 4, 5 or 6
X, Y individually represent F, or X and Y together represent ═O
R1 is selected from the group consisting of:
-
- (a) —H,
- (b) —(C1-C6)alkyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3 or
- —COOR3; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3 or —COOR3 moieties,
- or which is optionally substituted by aryl, in which the aryl moiety is optionally substituted by 1 or 2 substituents independently selected from the group consisting of halogen, hydroxyl and —(C1-C6)alkyl, and
- (c) -phenyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3, —COOR3, or —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens, and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; the number of said substituents on the phenyl moiety being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3, —COOR3 or —(C1-C6)alkyl moieties,
- wherein each R3 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl, optionally substituted by 1, 2 or 3 halogens,
R2 is selected from the group consisting of: - (a) —(C1-C8)alkyl,
- which is optionally substituted by halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5 or —COR4 moieties;
- (b) aryl or aryl-(C1-C8)alkyl,
- in which the aryl moiety is monocyclic or bicyclic;
- and which aryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4, or —COR4; the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4, or —COR4 moieties;
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (c) heteroaryl or heteroaryl-(C1-C8)alkyl,
- in which the heteroaryl moiety contains one, two or three heteroatoms independently selected from the group consisting of N, O or S, the number of N atoms being 0, 1, 2 or 3, and the number of O and S atoms each being 0, 1 or 2,
- and which heteroaryl moiety is optionally substituted by halogen, carbonitril, nitro,
- —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4, the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4 moieties,
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (d) (C3-C8)cycloalkyl or (C3-C8)cycloalkyl-(C1-C8)alkyl,
- in which the cycloalkyl moiety is optionally substituted by halogen, carbonitril, —OR4,
- —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties,
- (e) cycloheteroalkyl or cycloheteroalkyl-(C1-C8)alkyl,
- in which the cycloheteroalkyl moiety is optionally substituted by halogen, carbonitril,
- —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties; and
- (f) —(C1-C8)alkanoyl
- wherein
- each R4 and R5 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl and aryl-(C1-C6)alkyl, optionally substituted in the aryl moiety by 1, 2 or 3 halogens, or
- R4 and R5 form together with the nitrogen atom, to which they are attached, a cyclic 5-, 6-, 7- or 8-membered ring system, which is saturated or contains one or more double bonds between the ring atoms, and which ring optionally contains 1 or 2 heteroatoms in addition to the nitrogen atom, wherein the heteroatoms are independently selected from the group consisting of N, O or S, the number of additional N atoms being 0, 1 or 2 and the number of O and S atoms each being 0, 1 or 2, or which ring optionally contains a sulfoxide moiety in addition to the nitrogen atom, and
- R6 represents —(C1-C6)alkyl, which is optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties;
R7 is selected from the group consisting of
wherein R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof. In a further embodiment, the present invention relates to the 15β enantiomer having formula (Ia), wherein n represents 2, 3, 4, 5 or 6.
wherein R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof. In a further embodiment, the present invention relates to the 15α enantiomer having formula (Ib), wherein n represents 3.
wherein R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof. In a further embodiment, the present invention relates to the 15β enantiomer having formula (Ib), wherein n represents 3.
wherein R1, R2 and R7 are as defined herein, and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof. In a further embodiment, the present invention relates to the 15α enantiomer having formula (Ib), wherein n represents 3 or 4.
- 3-Hydroxy-15β-[2-(4-phenethyl-[1,2,3]triazol-1-yl)-ethyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(3-hydroxy-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(2,4-difluoro-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(3-methyl-butyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(3,5-difluoro-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-[2-(4-cyclohexylmethyl-[1,2,3]triazol-1-yl)-ethyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(2-fluoro-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-[3-(4-phenethyl-[1,2,3]triazol-1-yl)-propyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(3-trifluoromethyl-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(4-trifluoromethyl-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(4-methoxy-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-[2-(4-iso-butyl-[1,2,3]triazol-1-yl)-ethyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(4-trifluoromethoxy-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15α-[3-(4-phenethyl-[1,2,3]triazol-1-yl)-propyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{3-[4-(3-methyl-butyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{3-[4-(3,5-difluoro-phenyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{2-[4-(2-trifluoromethyl-phenyl)-[1,2,3]triazol-1-yl]-ethyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15α-{3-[4-(4-methoxy-phenyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-[3-(4-cyclohexylmethyl-[1,2,3]triazol-1-yl)-propyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15α-{3-[4-(3-hydroxy-phenyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-[3-(4-iso-butyl-[1,2,3]triazol-1-yl)-propyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15α-[3-(4-p-tolyl-[1,2,3]triazol-1-yl)-propyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{3-[4-(2,4-difluoro-phenyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15α-{3-[4-(3-methyl-butyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15α-[3-(4-iso-butyl-[1,2,3]triazol-1-yl)-propyl]-estra-1,3,5(10)-trien-17-one
- 3-Hydroxy-15β-{3-[4-(4-trifluoromethoxy-phenyl)-[1,2,3]triazol-1-yl]-propyl}-estra-1,3,5(10)-trien-17-one
- 4-{1-[3-(3-Methoxy-15β-17-oxo-estra-1,3,5(10)-trien-15-yl)-propyl]-1H-[1,2,3]triazol-4-yl}-benzoic acid methyl ester
- 15β-{3-[1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl]propyl}-3-hydroxyestra-1(10), 2,4-trien-17-one
- 15β-[3-(1-butyl-1H-1,2,3-triazol-4-yl)propyl]-3-hydroxyestra-1(10), 2,4-trien-17-one
- 15β-{3-[1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl]propyl}-17,17-difluoroestra-1(10), 2,4-trien-3-ol
and/or physiologically compatible salts, and/or solvates, and/or prodrugs thereof.
wherein
A represents N and B represents C
n represents 1, 2, 3, 4, 5 or 6
X, Y individually represent F, or X and Y together represent ═O
R1 is selected from the group consisting of:
-
- (a) —H,
- (b) —(C1-C6)alkyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3 or —COOR3; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3 or —COOR3 moieties,
- or which is optionally substituted by aryl, in which the aryl moiety is optionally substituted by 1 or 2 substituents selected from the group consisting of halogen, hydroxyl or —(C1-C6)alkyl, and
- (c) -phenyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3, —COOR3, or —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens, and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; the number of said substituents on the phenyl moiety being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3, —COOR3 and —(C1-C6)alkyl moieties,
- wherein each R3 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl, optionally substituted by 1, 2 or 3 halogens,
R2 is selected from the group consisting of: - (a) —(C1-C8)alkyl,
- which is optionally substituted by halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5 and —COR4 moieties;
- (b) aryl or aryl-(C1-C8)alkyl,
- in which the aryl moiety may be monocyclic or bicyclic;
- and which aryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4, or —COR4; the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 and —COR4 moieties;
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (c) heteroaryl or heteroaryl-(C1-C8)alkyl,
- in which the heteroaryl moiety contains one, two or three heteroatoms independently selected from the group consisting of N, O or S, the number of N atoms being 0, 1, 2 or 3, and the number of O and S atoms each being 0, 1 or 2,
- and which heteroaryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4, the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4 moieties,
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (d) (C3-C8)cycloalkyl or (C3-C8)cycloalkyl-(C1-C8)alkyl,
- in which the cycloalkyl moiety is optionally substituted by halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties,
- (e) cycloheteroalkyl or cycloheteroalkyl-(C1-C8)alkyl,
- in which the cycloheteroalkyl moiety is optionally substituted by halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties; and
- (f) —(C1-C8)alkanoyl
- wherein
- each R4 and R5 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl and aryl-(C1-C6)alkyl, optionally substituted in the aryl moiety by 1, 2 or 3 halogens, or
- R4 and R5 form together with the nitrogen atom, to which they are attached, a cyclic 5-, 6-, 7- or 8-membered ring system, which is saturated or contains one or more double bonds between the ring atoms, and which ring optionally contains 1 or 2 heteroatoms in addition to the nitrogen atom, wherein the heteroatoms are independently selected from the group consisting of N, O or S, the number of additional N atoms being 0, 1 or 2 and the number of O and S atoms each being 0, 1 or 2, or which ring optionally contains a sulfoxide moiety in addition to the nitrogen atom, and
- R6 represents —(C1-C6)alkyl, which is optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties;
R7 is selected from the group consisting of
wherein X, Y, R7 and n have the meanings as defined above and PG is a common protecting group,
is reacted by a copper catalyzed coupling with a terminal alkine of formula A,
wherein R2 has the meanings as defined above,
wherein different copper sources are used, selected from the group consisting of copper sources wherein copper has the oxidation states 0, I or II, and
wherein the protecting group is replaced after the coupling reaction by R1, which has the meaning as defined above.
wherein
n represents 1, 2, 3, 4, 5 or 6
X, Y individually represent F, or X and Y together represent ═O
R1 is selected from the group consisting of:
-
- (d) —H,
- (e) —(C1-C6)alkyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3 or —COOR3; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3 or —COOR3 moieties,
- or which is optionally substituted by aryl, in which the aryl moiety is optionally substituted by 1 or 2 substituents selected from the group consisting of halogen, hydroxyl or —(C1-C6)alkyl, and
- (f) -phenyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3, —COOR3, or —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens, and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; the number of said substituents on the phenyl moiety being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3, —COOR3 and —(C1-C6)alkyl moieties,
- wherein each R3 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl, optionally substituted by 1, 2 or 3 halogens,
R2 is selected from the group consisting of: - (g) —(C1-C8)alkyl,
- which is optionally substituted by halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5 and —COR4 moieties;
- (h) aryl or aryl-(C1-C8)alkyl,
- in which the aryl moiety may be monocyclic or bicyclic;
- and which aryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4, or —COR4; the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 and —COR4 moieties;
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (i) heteroaryl or heteroaryl-(C1-C8)alkyl,
- in which the heteroaryl moiety contains one, two or three heteroatoms independently selected from the group consisting of N, O or S, the number of N atoms being 0, 1, 2 or 3, and the number of O and S atoms each being 0, 1 or 2,
- and which heteroaryl moiety is optionally substituted by halogen, carbonitril, nitro,
- —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4, the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4 moieties,
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (j) (C3-C8)cycloalkyl or (C3-C8)cycloalkyl-(C1-C8)alkyl,
- in which the cycloalkyl moiety is optionally substituted by halogen, carbonitril, —OR4,
- —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties,
- (k) cycloheteroalkyl or cycloheteroalkyl-(C1-C8)alkyl,
- in which the cycloheteroalkyl moiety is optionally substituted by halogen, carbonitril,
- —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties; and
- (l) —(C1-C8)alkanoyl
- wherein
- each R4 and R5 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl and aryl-(C1-C6)alkyl, optionally substituted in the aryl moiety by 1, 2 or 3 halogens, or
- R4 and R5 form together with the nitrogen atom, to which they are attached, a cyclic 5-, 6-, 7- or 8-membered ring system, which is saturated or contains one or more double bonds between the ring atoms, and which ring optionally contains 1 or 2 heteroatoms in addition to the nitrogen atom, wherein the heteroatoms are independently selected from the group consisting of N, O or S, the number of additional N
- atoms being 0, 1 or 2 and the number of O and S atoms each being 0, 1 or 2, or which ring optionally contains a sulfoxide moiety in addition to the nitrogen atom, and
- R6 represents —(C1-C6)alkyl, which is optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties;
R7 is selected from the group consisting of
wherein R7 and n have the meanings as defined above and PG is a common protecting group,
is reacted by a Cu (I)-catalyzed coupling in the presence of an azide (e.g. NaN3) with a halide of formula B,
wherein R2 has the meanings as defined above,
wherein a modification of the C17 keto group affords compounds of formula (Ic) with X, Y═F, and
wherein the protecting group is replaced after the coupling reaction by R1, which has the meaning as defined above.
wherein
n represents 3, or 4
X, Y individually represent F, or X and Y together represent ═O
R1 is selected from the group consisting of:
-
- (g) —H,
- (h) —(C1-C6)alkyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3 or —COOR3; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3 or —COOR3 moieties,
- or which is optionally substituted by aryl, in which the aryl moiety is optionally substituted by 1 or 2 substituents selected from the group consisting of halogen, hydroxyl or —(C1-C6)alkyl, and
- (i) -phenyl, which is optionally substituted by halogen, carbonitril, —OR3, —SR3, —COOR3, or —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens, and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; the number of said substituents on the phenyl moiety being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR3, —SR3, —COOR3 and —(C1-C6)alkyl moieties,
- wherein each R3 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl, optionally substituted by 1, 2 or 3 halogens,
R2 is selected from the group consisting of: - (m) —(C1-C8)alkyl,
- which is optionally substituted by halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —O—SO2—R4, —NR4R5 and —COR4 moieties;
- (n) aryl or aryl-(C1-C8)alkyl,
- in which the aryl moiety may be monocyclic or bicyclic;
- and which aryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4, or —COR4; the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 and —COR4 moieties;
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (o) heteroaryl or heteroaryl-(C1-C8)alkyl,
- in which the heteroaryl moiety contains one, two or three heteroatoms independently selected from the group consisting of N, O or S, the number of N atoms being 0, 1, 2 or 3, and the number of O and S atoms each being 0, 1 or 2,
- and which heteroaryl moiety is optionally substituted by halogen, carbonitril, nitro,
- —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4, the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —O—SO2—R4, —NR4R5, —COOR4 or —COR4 moieties,
- and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens;
- (p) (C3-C8)cycloalkyl or (C3-C8)cycloalkyl-(C1-C8)alkyl,
- in which the cycloalkyl moiety is optionally substituted by halogen, carbonitril, —OR4,
- —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties,
- (q) cycloheteroalkyl or cycloheteroalkyl-(C1-C8)alkyl,
- in which the cycloheteroalkyl moiety is optionally substituted by halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, —OR4, —R6, —O—SO2—R4, —NR4R5, or —COR4 moieties; and
- (r) —(C1-C8)alkanoyl
- wherein
- each R4 and R5 is independently selected from the group consisting of H, —(C1-C6)alkyl, optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties; and aryl and aryl-(C1-C6)alkyl, optionally substituted in the aryl moiety by 1, 2 or 3 halogens, or
- R4 and R5 form together with the nitrogen atom, to which they are attached, a cyclic 5-, 6-, 7- or 8-membered ring system, which is saturated or contains one or more double bonds between the ring atoms, and which ring optionally contains 1 or 2 heteroatoms in addition to the nitrogen atom, wherein the heteroatoms are independently selected from the group consisting of N, O or S, the number of additional N atoms being 0, 1 or 2 and the number of O and S atoms each being 0, 1 or 2, or which ring optionally contains a sulfoxide moiety in addition to the nitrogen atom, and
- R6 represents —(C1-C6)alkyl, which is optionally substituted by 1, 2 or 3 halogens and/or optionally substituted by 1, 2 or 3 hydroxyl moieties;
R7 is selected from the group consisting of
wherein R7 has the meanings as defined above and PG is a common protecting group,
is reacted with a triazole allyl compound of formula C,
wherein R2 has the meanings as defined above,
wherein a modification of the C17 keto group affords compounds of formula (Id) with X, Y═F, and
wherein the protecting group is replaced after the coupling reaction by R1, which has the meaning as defined above.
whereas a C15α derivative according to the present invention is represented by a compound of the following formulae (Ib) and (Id)
| TABLE 1 |
| description of specific structural groups |
| TERM | GROUP | ||
| hydroxyl | —OH | ||
| sulfoxide |
|
||
| carbonitril | —CN | ||
| formyl |
|
||
| phenyl |
|
||
| naphthyl |
|
||
| benzenesulfonyloxy |
|
||
| benzylmethylamino |
|
||
| dimethylamino | —N(CH3)2 | ||
| acetyl |
|
||
| cyclohexyl |
|
||
| dioxothiomorpholin-4-yl |
|
||
| methoxy | —O—CH3 | ||
| hydroxymethyl | —CH2—OH | ||
| methoxycarbonyl |
|
||
| nitro |
|
||
| trihalomethoxy | —O—CX3 | ||
| with X =F, Cl, Br, I | |||
| trihalomethyl | —CX3 | ||
| with X =F, Cl, Br, I | |||
| pyridine-2-yl |
|
||
| benzyl |
|
||
| pyridine-3-yl |
|
||
| pyridine-4-yl |
|
||
| imidazol-4-yl |
|
||
| cyclopropyl |
|
||
| cyclopentyl |
|
||
| fur-2-yl |
|
||
| fur-3-yl |
|
||
| thiophen-2-yl |
|
||
| thiophen-3-yl |
|
||
| cyclohexylmethyl |
|
||
-
- (a) —(C1-C7)alkyl, which is optionally substituted by halogen, —OR4, —O—SO2—R4, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, —OR4, —NR4R5, —O—SO2—R4 and —COR4 moieties; preferably —(C1-C6)alkyl, which is optionally substituted by —OR4, —O—SO2—R4, —NR4R5; the number of said substituents being 1 or 2 for any combination of said —OR4, —O—SO2—R4 and —NR4R5 moieties; more preferably —(C1-C5)alkyl, which is optionally substituted by benzenesulfonyloxy, benzyl-methyl-amino, cyclohexyl, dimethylamino, dioxothiomorpholin-4-yl, formyl, hydroxyl, methoxy, or phenyl,
- (b) aryl or aryl-(C1-C4)alkyl, in which the aryl moiety is monocyclic or bicyclic; and which aryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —NR4R5, —COOR4, or —COR4; the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6, —NR4R5, —COOR4 and —COR4 moieties; and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens; preferably aryl or aryl-(C1-C2)alkyl, in which the aryl moiety is phenyl, benzyl or naphthyl; and which aryl moiety is optionally substituted by halogen, carbonitril, nitro, —OR4, —R6, —NR4R5, or —COOR4; the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, carbonitril, nitro, —OR4, —R6—NR4R5 and —COR4 moieties; more preferably phenyl or naphthyl, which are optionally substituted by carbonitril, dimethylamino, formyl, hydroxyl, hydroxymethyl, methoxy, methoxycarbonyl, methyl, nitro, trihalomethoxy, trihalomethyl, or 1 or 2 halogens,
- (c) heteroaryl or heteroaryl-(C1-C4)alkyl, in which the heteroaryl moiety contains one, two or three heteroatoms independently selected from the group consisting of N, O or S, the number of N atoms being 0, 1, 2 or 3, and the number of O and S atoms each being 0, 1 or 2, and which heteroaryl moiety is optionally substituted by halogen, nitro, —OR4, —R6, —NR4R5, —COOR4 or —COR4, the number of said substituents being 1, 2, 3 or 4 for halogen, and 1 or 2 for any combination of said halogen, nitro, —OR4, —R6, —NR4R5, —COOR4 and —COR4 moieties, and in which the alkyl moiety is optionally substituted by 1, 2 or 3 halogens; preferably heteroaryl or heteroaryl-(C1-C2)alkyl, in which the heteroaryl moiety contains one, two or three heteroatoms independently selected from the group consisting of N, O or S, the number of N atoms being 0, 1 or 2, and the number of O and S atoms each being 0 or 1, and which is optionally substituted by —R6; more preferably pyridine-2-yl, pyridine-3-yl, pyridine-4-yl, fur-2-yl, fur-3-yl, thiophen-2-yl, thiophen-3-yl, or imidazol-4-yl, and which are optionally substituted by methyl,
- (d) (C3-C7)cycloalkyl or (C3-C7)cycloalkyl-(C1-C4)alkyl, in which the cycloalkyl moiety is optionally substituted by halogen, —OR4, —R6, —NR4R5, or —COR4; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, —OR4, —R6—NR4R5 and —COR4 moieties; preferably (C3-C6)cycloalkyl or (C3-C6)cycloalkyl-(C1-C2)alkyl, in which the cycloalkyl moiety is optionally substituted by halogen, —OR4 and —R6; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, —OR4, or —R6 moieties; more preferably cyclopropyl, cyclopentyl, or cyclohexyl, and which are optionally substituted by hydroxyl,
- (e) cycloheteroalkyl or cycloheteroalkyl-(C1-C4)alkyl, in which the cycloheteroalkyl moiety is optionally substituted by halogen, —OR4, or —R6; the number of said substituents being 1, 2 or 3 for halogen, and 1 or 2 for any combination of said halogen, —OR4 and —R6 moieties; and cycloheteroalkyl or cycloheteroalkyl-(C1-C2)alkyl, in which the cycloalkyl moiety is selected from the group consisting of piperidinyl, morpholinyl, thiomorpholinyl, piperazyl, pyryl, pyrrolidinyl, tetrahydrofuryl, azepanyl and tetrahydrothienyl, and which cycloheteroalkyl moiety is optionally substituted by —OR4 or —R6; and
- (f) —(C1-C4)alkanoyl, preferably acetyl.
| TABLE 2 |
| terms with indicated meanings |
| 9-BBN | 9-borabicyclo | MEOH | METHANOL |
| [3.3.1]nonane | |||
| 18- | 1,4,7,10,13,16- | MgSO4 | magnesium sulfate |
| crown-6 | hexaoxa- | ||
| cyclooctadecane | |||
| Ac2O | acetic anhydride | MS | mass spectroscopy |
| aq. | aqueous | MsCl | mesyl chloride |
| Bn | benzyl | MTBE | Methyl-tert-butylether |
| brine | saturated sodium | μM | micro molar |
| chloride solution | |||
| Celite ® | CAS No 68855-54-9 | NaCl | sodium chloride |
| Cul | copper iodide | nM | nanomolar |
| CuSO4 | copper sulfate | NMO | 4-methylmorpholine |
| N-oxide | |||
| DAST | N,N- | NaN3 | sodium azide |
| diethylaminosulfur | |||
| trifluoride | |||
| DCM | dichloromethane | Na2SO4 | sodium sulfate |
| Diglyme | diethylene glycol | Na2S2O5 | sodium metabisulfite |
| dimethyl ether | |||
| DMAP | 4-(Dimethylamino)- | NaBH4 | sodium |
| pyridine | tetrahydridoborate | ||
| DMF | Dimethyl formamide | NADPH | nicotinamide adenine |
| dinucleotide | |||
| phosphate-oxidase | |||
| DMSO | dimethyl sulfoxide | NH3 | ammonia |
| Et3N | triethyl amine | NH4Cl | ammonium chloride |
| EtOAc | ethyl acetate | NH4OO | ammonium formate |
| CH | |||
| EtOH | ethyl alcohol | NaHCO3 | sodium bicarbonate |
| GRAS | generally recognized | NMO | N-methylmorpholin- |
| as safe | N-oxid | ||
| h. | hours | NMR | nuclear magnetic |
| resonance | |||
| H2 | hydrogen | o- | ortho |
| H2O | water | p- | para |
| HCl | hydrochloric acid | PG | protection group |
| hept. | heptane | ph- | phenyl |
| HMPA | hexamethyl- | Pd/C | carbon-supported |
| phosphoramide | palladium catalyst | ||
| HPLC | high performance | ppm | parts per million |
| liquid | |||
| chromatography | |||
| i- | iso | RT | room temperature |
| I2 | iodine | sat. | saturated |
| IUS | intrauterine systems | SiO2 | silicium dioxide |
| K2CO3 | potassium carbonate | T | temperature |
| KH2PO4 | potassium dihydrogen | TBAF | tetra-n- |
| butylammonium | |||
| phosphate | fluoride | ||
| KH | potassium hydride | TBME | tert-butyl |
| methyl ether | |||
| KOH | potassium hydroxide | THF | tetrahydrofurane |
| LC-MS | liquid chromatography | TLC | Thin Layer |
| with MS | Chromatography | ||
| LiCl | lithium | TMNO | trimethylamine-N- |
| chloride | oxide.hydrate | ||
| m- | meta | TMS | trimethylsilyl |
| Me3N | trimethylamine | TMSCl | trimethylsilyl chloride |
| Me3NO•. | trimethylaminoxid- | TPAP | tetrapropylammonium |
| 2H2O | dihydrat | perruthenate | |
| ME | METHYL | TsOH/ | toluenesulfonic acid |
| TosOH | |||
| MeO | methoxy | ||
General Preparative Methods
wherein R1, R2, R7, X and Y have the meanings as defined herein, and PG is a common protecting group like benzyl.
wherein R1, R2, R7, X and Y have the meanings as defined herein, and PG is a common protecting group like benzyl.
wherein R1, R2, R7, X and Y have the meanings as defined herein, and PG is a common protecting group.
is described for compounds with R7=H in C2 position in patent application WO 2005/047303 and for compounds with R7≠H in C2 position or with an optional difluoro group in C17 position in the PCT application WO 2006/125800 (intermediate compounds of general formula XLIII). Typically, the corresponding alcohol derivatives are used as intermediates for the azide synthesis. A possible synthesis route to obtain such alcohol intermediates—also with different chain length in the C15 substituent and different stereochemistry in C15—is described for compounds with R7=H in C2 position in patent application WO 2005/047303 and for compounds with R7≠H in C2 position or with an optional difluoro group in C17 position in the PCT application WO 2006/125800 (intermediate compounds of general formula XXXI and XXXII).
| TABLE 3 |
| selected examples of compounds of formula Ix |
|
|
| cnfg. | MS | Rt | |||||
| cpd | R1 | R2 | R7 | n | C15 | (m/z) | (min) |
| 1 | H | phenethyl | H | 2 | β | 469.27 | 5.67 |
| 2 | H | m-hydroxy-phenyl | H | 2 | β | 457.24 | 5.1 |
| 3 | H | o,p-difluoro-phenyl | H | 2 | β | 477.22 | 5.91 |
| 4 | H | 3-methyl-butyl | H | 2 | β | 435.29 | 5.77 |
| 5 | H | m,m-difluoro-phenyl | H | 2 | β | 477.22 | 5.97 |
| 6 | H | cyclohexylmethyl | H | 2 | β | 461.3 | 6.01 |
| 7 | H | o-fluoro-phenyl | H | 2 | β | 459.23 | 5.79 |
| 8 | H | phenethyl | H | 3 | β | 483.29 | 5.84 |
| 9 | H | m-trifluoro-phenyl | H | 2 | β | 509.23 | 6.14 |
| 10 | H | p-trifluoromethyl-phenyl | H | 2 | β | 509.23 | 6.15 |
| 11 | H | p-methoxy-phenyl | H | 2 | β | 471.25 | 5.56 |
| 12 | H | i-butyl | H | 2 | β | 421.27 | 5.47 |
| 13 | H | p-trifluoromethoxy-phenyl | H | 2 | β | 525.22 | 6.2 |
| 16 | H | phenethyl | H | 3 | α | 483.29 | 5.85 |
| 17 | H | 3-methyl-butyl | H | 3 | β | 449.3 | 5.92 |
| 18 | H | m,m-difluoro-phenyl | H | 3 | β | 491.24 | 6.1 |
| 19 | H | o-trifluoro-phenyl | H | 2 | β | 509.23 | 5.91 |
| 20 | H | p-methoxy-phenyl | H | 3 | α | 485.27 | 5.72 |
| 21 | H | cyclohexylmethyl | H | 3 | β | 475.32 | 6.16 |
| 22 | H | m-hydroxy-phenyl | H | 3 | α | 471.25 | 5.26 |
| 24 | H | i-butyl | H | 3 | β | 435.29 | 5.64 |
| 25 | H | p-methyl-phenyl | H | 3 | α | 469.27 | 5.97 |
| 28 | H | o,p-difluoro-phenyl | H | 3 | β | 491.24 | 6.06 |
| 30 | H | 3-methyl-butyl | H | 3 | α | 449.3 | 5.94 |
| 31 | H | i-butyl | H | 3 | α | ||
| 32 | H | p-trifluoromethoxy-phenyl | H | 3 | β | 539.24 | 6.31 |
| 33 | H | phenethyl | H | 4 | β | 497.3 | 6.06 |
| 34 | H | cyclohexyl-methyl | H | 3 | α | 475.32 | 6.22 |
| 36 | Me | i-butyl | H | 3 | β | 449.3 | 6.65 |
| 37 | Me | o-nitro-phenyl | H | 3 | β | 514.26 | 6.65 |
| 38 | H | p-benzaldehyde | H | 3 | α | 483.25 | 5.58 |
| 39 | H | m-methoxy-phenyl | H | 3 | α | 485.27 | 5.78 |
| 40 | H | p-trifluoromethoxy-phenyl | H | 3 | α | 539.24 | 6.35 |
| 41 | Me | trifluoromethoxy | H | 3 | β | 433.27 | 6.26 |
| 42 | H | i-butyl | H | 4 | β | 449.3 | 5.89 |
| 43 | H | m-hydroxy-phenyl | H | 3 | β | 471.25 | 5.22 |
| 44 | H | 6-methoxy-naphthalen-2-yl | H | 3 | α | 535.28 | 6.19 |
| 45 | H | p-trifluoromethyl-phenyl | H | 3 | β | 523.24 | 6.28 |
| 46 | H | o-fluoro-phenyl | H | 3 | α | 473.25 | 5.97 |
| 48 | H | 3-methyl-butyl | H | 4 | β | 463.32 | 6.17 |
| 49 | H | p-trifluoro-phenyl | H | 3 | α | 523.24 | 6.3 |
| 50 | Me | propyl | H | 3 | β | 435.29 | 6.42 |
| 51 | H | m-methoxy-phenyl | H | 3 | β | 485.27 | 5.75 |
| 52 | H | m,m-difluoro-phenyl | H | 3 | α | 491.24 | 6.14 |
| 53 | Me | m-hydroxy-phenyl | H | 4 | β | 499.28 | 6.34 |
| 53 | H | m-hydroxy-phenyl | H | 4 | β | 485.27 | 5.43 |
| 54 | H | cyclohexylmethyl | H | 4 | β | 489.34 | 6.46 |
| 55 | H | 2-hydroxy-ethyl | H | 3 | β | 423.25 | 4.55 |
| 56 | H | o,p-difluoro-phenyl | H | 3 | α | 491.24 | 6.1 |
| 57 | Me | cyclohexyl | H | 3 | β | 475.32 | 6.99 |
| 58 | H | i-butyl | H | 6 | β | 477.34 | 6.36 |
| 59 | Me | thiophen-3-yl | H | 3 | β | 475.23 | 6.53 |
| 60 | Me | p-hydroxymethyl-phenyl | H | 3 | β | 499.28 | 5.88 |
| 62 | H | methoxy-methyl | H | 2 | β | 409.24 | 4.74 |
| 63 | H | 2-hydroxy-ethyl | H | 2 | β | 409.24 | 4.39 |
| 64 | Me | o-fluoro-phenyl | H | 3 | β | 487.26 | 6.91 |
| 65 | H | m-trifluoro-phenyl | H | 3 | α | 523.24 | 6.28 |
| 66 | Me | phenethyl | H | 3 | β | 497.3 | 6.79 |
| 67 | H | m-trifluoromethyl-phenyl | H | 3 | β | 523.24 | 6.25 |
| 68 | Me | p-methoxy-phenyl | H | 3 | β | 499.28 | 6.6 |
| 69 | H | 3-methyl-butyl | H | 6 | β | 491.35 | 6.65 |
| 70 | Me | 3-methyl-butyl | H | 3 | β | 463.32 | 6.92 |
| 71 | Me | methoxy-methyl | H | 3 | β | 437.27 | 5.87 |
| 72 | H | o-trifluoro-phenyl | H | 3 | α | 523.24 | 6.06 |
| 73 | H | m,m-difluoro-phenyl | H | 4 | β | 505.25 | 6.31 |
| 74 | Me | m-methoxy-phenyl | H | 3 | β | 499.28 | 6.66 |
| 75 | Me | p-methyl-phenyl | H | 3 | β | 483.29 | 6.89 |
| 76 | Me | p-dimethylamino-phenyl | H | 3 | β | 512.32 | 6.77 |
| 77 | Me | thiophen-3-yl | H | 4 | β | 489.24 | 6.81 |
| 78 | Me | cyclohexyl-methyl | H | 3 | β | 489.34 | 7.18 |
| 79 | Me | phenethyl | H | 4 | β | 511.32 | 7.04 |
| 80 | Me | m-hydroxy-phenyl | H | 3 | β | 485.27 | 6.05 |
| 82 | Me | dimethylamino-methyl | H | 3 | β | 450.3 | 5.01 |
| 83 | Me | o-chloro-phenyl | H | 3 | β | 503.23 | 7.06 |
| 84 | H | o-trifluoromethyl-phenyl | H | 3 | β | 523.24 | 6.05 |
| 85 | Me | p-hydroxy-naphthalen-2-yl | H | 4 | β | 487.29 | 5.86 |
| 86 | H | 6-methoxy-naphthalen-2-yl | H | 4 | β | 549.3 | 6.38 |
| 87 | Me | m,m-difluoro-phenyl | H | 3 | β | 505.25 | 7 |
| 88 | Me | o,p-difluoro-phenyl | H | 3 | β | 505.25 | 7 |
| 90 | Me | p-methoxy-phenyl | H | 4 | β | 513.3 | 6.89 |
| 91 | Me | o-trifluoromethyl-phenyl | H | 3 | β | 537.26 | 6.99 |
| 92 | H | o,p-difluoro-phenyl | H | 4 | β | 505.25 | 6.29 |
| 93 | H | methoxy-methyl | H | 6 | β | 465.3 | 5.6 |
| 94 | Me | o-methoxy-phenyl | H | 3 | β | 499.28 | 6.78 |
| 95 | H | 2-hydroxy-ethyl | H | 6 | β | 465.3 | 5.15 |
| 96 | Me | hydroxymethyl | H | 3 | β | 423.25 | 5.38 |
| 97 | Me | propyl | H | 4 | β | 449.3 | 6.75 |
| 98 | Me | acetyl | H | 3 | β | 435.25 | 6.1 |
| 99 | Me | cyclohexyl-methyl | H | 4 | β | 503.35 | 7.5 |
| 100 | H | m-hydroxy-phenyl | H | 6 | β | 513.3 | 5.84 |
| 101 | H | hydroxyl-methyl | H | 3 | α | 409.24 | 4.53 |
| 102 | Me | benzo-p-nitrile | H | 3 | β | 494.27 | |
| 103 | H | methoxy-methyl | H | 3 | α | 423.25 | 4.92 |
| 104 | Me | benzylmethyl-amino | H | 3 | β | 526.33 | 6.26 |
| 105 | H | hydroxy-methyl | H | 6 | β | 451.28 | 5.13 |
| 106 | Me | pyridine-2-yl | H | 3 | β | 470.27 | 6.2 |
| 107 | H | methoxy-methyl | H | 3 | β | 423.25 | 4.92 |
| 108 | Me | p-benzaldehyde | H | 4 | β | 513.3 | 6.18 |
| 109 | Me | pyridine-4-yl | H | 3 | β | 470.27 | 5.91 |
| 110 | Me | 1-hydroxy-cyclopentyl | H | 3 | β | 477.3 | 5.91 |
| 111 | Me | cyclopropyl | H | 4 | β | 447.29 | 6.55 |
| 112 | Me | p-methyl-phenyl | H | 4 | β | 497.3 | 7.21 |
| 113 | Me | pyridine-3-yl | H | 3 | β | 470.27 | 5.91 |
| 114 | Me | 2-hydroxyethyl | H | 3 | β | 437.27 | 5.37 |
| 115 | Me | p-dimethylamino-phenyl | H | 4 | β | 526.33 | 7.05 |
| 116 | Me | o-fluoro-phenyl | H | 4 | β | 501.28 | 7.2 |
| 117 | H | o-trifluoromethyl-phenyl | H | 4 | β | 537.26 | 6.29 |
| 118 | H | hydroxy-methyl | H | 3 | β | 409.24 | 4.52 |
| 119 | Me | 6-methoxy-naphthalen-2-yl | H | 3 | β | 549.3 | 7.03 |
| 120 | Me | dioxo-thiomorpholin-4-yl- | H | 4 | β | 554.29 | 5.79 |
| methyl | |||||||
| 121 | Me | o-chloro-phenyl | H | 4 | β | 517.25 | 7.36 |
| 122 | Me | hydroxyl-methyl | H | 4 | β | 437.27 | 5.66 |
| 123 | Me | m-methoxy-phenyl | H | 4 | β | 513.3 | 6.96 |
| 124 | Me | p-trifluoromethoxy-phenyl | H | 3 | β | 553.26 | 7.19 |
| 125 | Me | 3-methyl-3H-imidazol-4-yl | H | 3 | β | 473.28 | |
| 126 | Me | o-nitro-phenyl | H | 4 | β | 528.27 | 6.92 |
| 127 | H | o,p-difluoro-phenyl | H | 4 | β | 533.29 | 6.74 |
| 128 | H | phenethyl | H | 4 | β | 525.34 | 6.49 |
| 129 | Me | cyclohexyl | H | 4 | β | 489.34 | 7.3 |
| 130 | H | 2-hydroxy-ethyl | H | 4 | β | 437.27 | 4.73 |
| 131 | Me | p-trifluoromethyl-phenyl | H | 3 | β | 537.26 | 7.14 |
| 132 | Me | p-benzonitrile | H | 4 | β | 508.28 | 6.85 |
| 133 | Me | dioxo-thiomorpholin-4-yl- | H | 3 | β | 540.28 | 5.53 |
| methyl | |||||||
| 134 | H | p-methyl-phenyl | H | 6 | β | 511.32 | 6.62 |
| 135 | H | p-methoxy-phenyl | H | 6 | β | 527.31 | 6.32 |
| 136 | Me | pyridine-4-yl | H | 4 | β | 484.28 | 6.24 |
| 137 | H | m-methoxy-phenyl | H | 6 | β | 527.31 | 6.41 |
| 138 | H | 2-hydroxy-ethyl | H | 3 | α | 423.25 | 4.56 |
| 139 | Me | i-butyl | H | 4 | β | 463.32 | 6.94 |
| 140 | H | m-trifluoromethyl-phenyl | H | 4 | β | 537.26 | 6.49 |
| 141 | H | methoxy-methyl | H | 4 | β | 437.27 | 5.14 |
| 142 | Me | methoxy-methyl | H | 4 | β | 451.28 | 6.18 |
| 143 | H | 6-methoxy-naphthalen-2-yl | H | 6 | β | 577.33 | 6.77 |
| 144 | Me | 3-methyl-butyl | H | 4 | β | 477.34 | 7.22 |
| 145 | Me | m,m-difluoro-phenyl | H | 4 | β | 519.27 | 7.25 |
| 146 | Me | o,p-difluoro-phenyl | H | 4 | β | 519.27 | 7.28 |
| 147 | Me | acetyl | H | 4 | β | 449.27 | 6.42 |
| 148 | H | m,m-difluoro-phenyl | H | 4 | β | 533.29 | 6.74 |
| 149 | H | hydroxy-methyl | H | 2 | β | 395.22 | 4.36 |
| 150 | Me | o-methoxy-phenyl | H | 4 | β | 513.3 | 7.08 |
| 151 | H | p-trifluoromethyl-phenyl | H | 4 | β | 537.26 | 6.49 |
| 152 | H | cyclohexylmethyl | H | 6 | β | 517.37 | 6.94 |
| 153 | Me | p-nitro-phenyl | H | 3 | β | 514.26 | 6.78 |
| 154 | Me | pyridine-3-yl | H | 4 | β | 484.28 | 6.22 |
| 155 | Me | o-trifluoromethyl-phenyl | H | 4 | β | 551.28 | 7.25 |
| 156 | Me | m-trifluoromethyl-phenyl | H | 3 | β | 537.26 | 7.14 |
| 157 | H | o-trifluoromethyl-phenyl | H | 6 | β | 565.29 | 6.72 |
| 158 | Me | p-nitro-phenyl | H | 4 | β | 528.27 | 7.05 |
| 159 | Me | p-trifluoromethoxy-phenyl | H | 4 | β | 567.27 | 7.42 |
| 160 | Me | 2-hydroxy-ethyl | H | 4 | β | 451.28 | 5.68 |
| 161 | H | p-trifluoromethoxy-phenyl | H | 4 | β | 581.29 | 6.92 |
| 162 | Me | pyridine-2-yl | H | 4 | β | 484.28 | 6.53 |
| 163 | Me | m-trifluoromethyl-phenyl | H | 4 | β | 551.28 | 7.39 |
| 164 | H | m-trifluoromethyl-phenyl | H | 6 | β | 565.29 | 6.89 |
| 165 | Me | benzenesulfonyl-methyl | H | 4 | β | 577.26 | 5.43 |
| 166 | H | p-trifluoromethyl-phenyl | H | 4 | β | 565.29 | 6.88 |
| 167 | Me | 6-methoxy-naphthalen-2-yl | H | 4 | β | 563.31 | 7.3 |
| 168 | H | o-fluoro-phenyl | H | 6 | β | 515.29 | 6.65 |
| 169 | Me | p-trifluoromethyl-phenyl | H | 4 | β | 551.28 | 7.38 |
| 170 | H | hydroxy-methyl | H | 4 | β | 423.25 | 4.72 |
-
- Recombinant HPLC assay 17βHSD1 and 17βHSD2
- 17βHSD1-MCF-7 cell assay
- Estrogen receptor binding and functional assay
- In vivo assays, e.g. UWT assay, tumor model, and
- Disease-oriented models,
thereby first focusing on the effect on enzymatic activity of recombinant human 17βHSD1 and on selectivity towards recombinant human 17β-hydroxysteroid dehydrogenase type 2 (17βHSD2), the enzyme catalysing the reverse reaction as 17βHSD1 by conversion of E2 to E1 (methods as described in WO2005032527). These protein-based tests are followed by the corresponding cell-based assays (WO2005032527). Another important factor is selectivity towards the estrogen receptor that is studied in a commercially available binding assay (PanVera LCC, Madison, Wis.) as well as in a functional ERE-LUC receptor gene assay as described by M. E. Burow, S. M. Boue, B. M. Collins-Burow, L. I. Melnik, B. N. Duong, C. H. Carter-Wientjes, S. Li, T. E. Wiese, T. E. Cleveland and J. A. McLachlan, Phytochemical glyceollins, isolated from soy, mediate antihormonal effects through estrogen receptor alpha and beta, J. Clin. Endocrinol. Metab. 86 (2001) (4), pp. 1750-1758. After determining metabolic and physicochemical stability of a compound the first set of in vivo experiments is started. Lack of estrogenic activity in vivo is proven using the classical uterine growth test in immature rats (H. D. Lauson, C. G. Heller, J. B. Golden and E. L. Severinghaus, The immature rat uterus in the assay of estrogenic substances, a comparison of estradiol, estrone and estriol, Endocrinoloy 24 (1939), pp. 35-44). Efficacy of 17βHSD1-inhibition is demonstrated by reduction of 17βHSD1-dependent growth of tumor xenografts in immunodeficient mice as described by Husen, B., Huhtinen, K., Poutanen, M., Kangas, L., Messinger, J., Thole, H. (2006) Evaluation of inhibitors for 17β-hydroxysteroid dehydrogenase type 1 in vivo in immunodeficient mice inoculated with MCF-7 cells stably expressing the recombinant human enzyme. Mol Cell Endocrinol. 2006 Mar. 27; 248(1-2):109-13. Epub 2006 Jan. 10. Finally disease-oriented models as disclosed by R. Grümmer, F. Schwarzer, K. Bainczyk, H. Hess-Stumpp, P. A. Regidor, A. E. Schindler and E. Winterhager, Peritoneal endometriosis: validation of an in vivo model, Hum. Reprod. 16 (2001) (8), pp. 1736-1743 and A. Einspanier, A. C. K Brüns, B. Husen and C. Simon, Induction of endometriosis in the marmoset monkey (Callithrix jacchus) Mol Hum Reprod. 2006 May; 12(5):291-9. Epub 2006 Apr. 11 determine the proof of concept of these compounds.
-
- Percent inhibition was calculated as follows: % inhibition=100−% conversion
| TABLE 4 |
| HSD 1 inhibition values in % of selected compounds of formula I |
| inhibition of | |
| rec. 17 β-HSD1 |
| compound | chemical structure | 100 nM | 1 μM |
| 1 | | 92 | 98 |
| 2 | | 88 | 97 |
| 3 | | 86 | 96 |
| 4 | | 92 | 96 |
| 5 | | 87 | 95 |
| 6 | | 92 | 94 |
| 7 | | 81 | 94 |
| 8 | | 81 | 93 |
| 9 | | 84 | 93 |
| 10 | | 83 | 93 |
| 11 | | 75 | 93 |
| 12 | | 86 | 92 |
| 13 | | 83 | 92 |
| 16 | | 38 | 89 |
| 17 | | 61 | 88 |
| 18 | | 64 | 88 |
| 19 | | 71 | 87 |
| 20 | | 51 | 87 |
| 21 | | 50 | 86 |
| 22 | | 48 | 86 |
| 24 | | 52 | 85 |
| 25 | | 49 | 85 |
| 28 | | 40 | 83 |
| 30 | | 25 | 82 |
| 31 | | 32 | 82 |
| 32 | | 67 | 81 |
| 33 | | 20 | 80 |
| 37 | | 25 | 80 |
| 41 | | 23 | 77 |
| 48 | | 21 | 73 |
| 58 | | 20 | 70 |
| 59 | | 18 | 69 |
| 60 | | 19 | 69 |
| 69 | | 30 | 64 |
| 89 | | 11 | 52 |
| 100 | | 7 | 48 |
| 111 | | 4 | 43 |
| 112 | | 12 | 43 |
| 122 | | 5 | 38 |
| 300 | | 85 | 88 |
| 301 | | 82 | 88 |
| 302 | | 88 | 96 |
Inhibition of the 17β-Hydroxysteroid Dehydrogenase Type 3 Enzyme
| TABLE 5 |
| 17β-HSD3 inhibition values in % |
| of selected compounds of formula I |
| inhibition of 17β-HSD3 |
| Compound No. | 1 μM | 10 μM |
| 4 | 5 | 40 |
| 6 | 16 | 50 |
| 7 | 12 | 36 |
| 8 | 6 | 61 |
| 12 | — | 37 |
| 17 | 3 | 50 |
| 19 | 11 | 52 |
| 22 | 82 | — |
| 24 | 24 | 64 |
| 300 | 29 | 61 |
| 301 | 24 | 57 |
| 302 | 13 | 70 |
Estrogen Receptor Binding Assay
Claims (22)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/856,768 US8080540B2 (en) | 2006-09-19 | 2007-09-18 | Therapeutically active triazoles and their use |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US84549606P | 2006-09-19 | 2006-09-19 | |
| US95784807P | 2007-08-24 | 2007-08-24 | |
| US11/856,768 US8080540B2 (en) | 2006-09-19 | 2007-09-18 | Therapeutically active triazoles and their use |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20080146531A1 US20080146531A1 (en) | 2008-06-19 |
| US8080540B2 true US8080540B2 (en) | 2011-12-20 |
Family
ID=39528135
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/856,768 Expired - Fee Related US8080540B2 (en) | 2006-09-19 | 2007-09-18 | Therapeutically active triazoles and their use |
Country Status (1)
| Country | Link |
|---|---|
| US (1) | US8080540B2 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CZ307437B6 (en) | 2016-06-07 | 2018-08-22 | Ăšstav organickĂ© chemie a biochemie AV ÄŚR, v.v.i. | 15β-substituted estrone derivatives as selective inhibitors of 17β-hydroxysteoid dehydrogenases |
Citations (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3347878A (en) | 1965-03-31 | 1967-10-17 | Du Pont | Selected 17, 17-difluoro steroids of the estrane series |
| US3413321A (en) | 1966-06-03 | 1968-11-26 | Du Pont | Selected 17-fluoro-delta16 steroids |
| EP0367576A2 (en) | 1988-10-31 | 1990-05-09 | Endorecherche Inc. | Estrogen nucleus derivatives for use in the inhibition of sex steroid activity |
| EP0902022A1 (en) | 1997-01-14 | 1999-03-17 | Snow Brand Milk Products Co., Ltd. | Novel benzofuranone derivatives and process for producing the same |
| WO1999046279A2 (en) | 1998-03-11 | 1999-09-16 | Endorecherche, Inc. | INHIBITORS OF TYPE 5 AND TYPE 3 17β-HYDROXYSTEROID DEHYDROGENASE AND METHODS FOR THEIR USE |
| US5993856A (en) | 1997-01-24 | 1999-11-30 | Femmepharma | Pharmaceutical preparations and methods for their administration |
| WO2000007996A2 (en) | 1998-08-07 | 2000-02-17 | Chiron Corporation | Pyrazoles as estrogen receptor modulators |
| WO2001042181A1 (en) | 1999-12-10 | 2001-06-14 | University College Cardiff Consultants Limited | Benzyl tetralins, formulations and uses thereof |
| WO2002026706A2 (en) | 2000-09-29 | 2002-04-04 | Bayer Pharmaceuticals Corporation | 17-beta-hydroxysteroid dehydrogenase-ii inhibitors |
| US6416778B1 (en) | 1997-01-24 | 2002-07-09 | Femmepharma | Pharmaceutical preparations and methods for their regional administration |
| WO2003017973A1 (en) | 2001-08-31 | 2003-03-06 | Pantarhei Bioscience B.V. | A method of treating benign gynaecological disorders and a drug delivery vehicle for use in such a method |
| WO2003022835A1 (en) | 2001-09-06 | 2003-03-20 | Schering Corporation | 17beta-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| WO2003033487A1 (en) | 2001-10-17 | 2003-04-24 | Schering Corporation | Piperidine- and piperazineacetamines as 17beta hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| US20030170292A1 (en) | 2001-11-09 | 2003-09-11 | National University Of Singapore | Methods for preparing an estrogenic preparation and isolated estrogenic compounds from a plant and uses thereof |
| WO2003101972A1 (en) | 2002-05-30 | 2003-12-11 | The Scripps Research Institute | Copper-catalysed ligation of azides and acetylenes |
| WO2004046111A1 (en) | 2002-11-18 | 2004-06-03 | Schering Corporation | 17beta-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| WO2004060488A1 (en) | 2002-12-17 | 2004-07-22 | Schering Corporation | 17 beta-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| WO2004080271A2 (en) | 2003-03-14 | 2004-09-23 | Pirkko Vihko | Method for prognosticating the progress of breast cancer and compounds useful for prevention or treatment thereof |
| WO2004085345A2 (en) | 2003-03-21 | 2004-10-07 | Yale University | 15α-SUBSTITUTED ESTRADIOL CARBOXYLIC ACID ESTERS AS LOCALLY ACTIVE ESTROGENS |
| WO2004085457A2 (en) | 2003-03-24 | 2004-10-07 | Sterix Limited | 17beta-hydroxysteroid dehydrogenase inhibitors |
| WO2004110459A1 (en) | 2003-06-10 | 2004-12-23 | Solvay Pharmaceuticals B.V. | Thiophenepyrimidinones as 17-beta-hydroxysteroid dehydrogenase inhibitors |
| WO2005032527A2 (en) | 2003-06-10 | 2005-04-14 | Solvay Pharmaceuticals B.V. | Benzo (4, 5) thieno (2, 3-d) pyrimidin-4-ones and their use in therapy |
| WO2005047303A2 (en) | 2003-11-12 | 2005-05-26 | Solvay Pharmaceuticals Gmbh | NOVEL 17β HYDROXYSTEROID DEHYDROGENASE TYPE I INHIBITORS |
| WO2005084295A2 (en) | 2004-03-01 | 2005-09-15 | Bristol-Myers Squibb Company | Fused heterotricyclic compounds as inhibitors of 17b-hydroxysteroid dehydrogenase 3 |
| US20060009434A1 (en) | 2004-07-02 | 2006-01-12 | Alexander Hillisch | New 2-substituted estra-1,3,5(10)-trien-17-ones as inhibitors of 17beta-hydroxy steroid dehydrogenase type 1 |
| WO2006003012A1 (en) | 2004-07-02 | 2006-01-12 | Schering Aktiengesellschaft | NOVEL 2-SUBSTITUTED D-HOMO-ESTRA-1,3,5(10)-TRIENES AS INHIBITORS OF 17ß-HYDROXYSTEROID DEHYDROGENASE TYPE 1 |
| WO2006003013A2 (en) | 2004-07-02 | 2006-01-12 | Bayer Schering Pharma Aktiengesellschaft | NOVEL 2-SUBSTITUTED ESTRA-1,3,5(10)-TRIEN-17-ONES USED IN THE FORM OF INHIBITORS OF 17β-HYDROXYSTEROIDDEHYDROGENASE OF TYPE 1 |
| US20060052461A1 (en) | 2004-07-02 | 2006-03-09 | Alexander Hillisch | New 2-substituted D-homo-estra-1,3,5(10)-trienes as inhibitors of 17beta-hydroxy steroid dehydrogenase type 1 |
| WO2006027347A1 (en) | 2004-09-08 | 2006-03-16 | N.V. Organon | 15β-SUBSTITUTED STEROIDS HAVING SELECTIVE ESTROGENIC ACTIVITY |
| WO2006032885A2 (en) | 2004-09-22 | 2006-03-30 | Sterix Limited | 2-substituted estrogen sulphamates for inhibition of steroid sulphatase |
| WO2006063585A1 (en) | 2004-12-13 | 2006-06-22 | Leo Pharma A/S | Triazole substituted aminobenzophenone compounds |
| WO2006063615A1 (en) | 2004-12-13 | 2006-06-22 | Solvay Pharmaceuticals Gmbh | NOVEL SUBSTITUTED THIOPHENEPYRIMIDINONE DERIVATIVES AS INHIBITORS OF 17β-HYDROXYSTEROID DEHYDROGENASE |
| WO2006125800A1 (en) | 2005-05-26 | 2006-11-30 | Solvay Pharmaceuticals Gmbh | 17β-HSD1 AND STS INHIBITORS |
| US20060271710A1 (en) | 2005-05-24 | 2006-11-30 | Inventec Corporation | Switch device |
-
2007
- 2007-09-18 US US11/856,768 patent/US8080540B2/en not_active Expired - Fee Related
Patent Citations (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3347878A (en) | 1965-03-31 | 1967-10-17 | Du Pont | Selected 17, 17-difluoro steroids of the estrane series |
| US3413321A (en) | 1966-06-03 | 1968-11-26 | Du Pont | Selected 17-fluoro-delta16 steroids |
| EP0367576A2 (en) | 1988-10-31 | 1990-05-09 | Endorecherche Inc. | Estrogen nucleus derivatives for use in the inhibition of sex steroid activity |
| EP0902022A1 (en) | 1997-01-14 | 1999-03-17 | Snow Brand Milk Products Co., Ltd. | Novel benzofuranone derivatives and process for producing the same |
| US5993856A (en) | 1997-01-24 | 1999-11-30 | Femmepharma | Pharmaceutical preparations and methods for their administration |
| EP0977555A1 (en) | 1997-01-24 | 2000-02-09 | FemmePharma | Pharmaceutical preparations and methods for their regional administration |
| US6652874B2 (en) | 1997-01-24 | 2003-11-25 | Femmepharma | Pharmaceutical preparations and methods for their regional administration |
| US6416778B1 (en) | 1997-01-24 | 2002-07-09 | Femmepharma | Pharmaceutical preparations and methods for their regional administration |
| US6541463B1 (en) | 1998-03-11 | 2003-04-01 | Endorecherche, Inc. | Inhibitors of type 5 and type 3 17β-hydroxysteroid dehydrogenase and methods for their use |
| WO1999046279A2 (en) | 1998-03-11 | 1999-09-16 | Endorecherche, Inc. | INHIBITORS OF TYPE 5 AND TYPE 3 17β-HYDROXYSTEROID DEHYDROGENASE AND METHODS FOR THEIR USE |
| WO2000007996A2 (en) | 1998-08-07 | 2000-02-17 | Chiron Corporation | Pyrazoles as estrogen receptor modulators |
| WO2001042181A1 (en) | 1999-12-10 | 2001-06-14 | University College Cardiff Consultants Limited | Benzyl tetralins, formulations and uses thereof |
| WO2002026706A2 (en) | 2000-09-29 | 2002-04-04 | Bayer Pharmaceuticals Corporation | 17-beta-hydroxysteroid dehydrogenase-ii inhibitors |
| WO2003017973A1 (en) | 2001-08-31 | 2003-03-06 | Pantarhei Bioscience B.V. | A method of treating benign gynaecological disorders and a drug delivery vehicle for use in such a method |
| WO2003022835A1 (en) | 2001-09-06 | 2003-03-20 | Schering Corporation | 17beta-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| US6903102B2 (en) | 2001-09-06 | 2005-06-07 | Schering Corporation | 17β-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| WO2003033487A1 (en) | 2001-10-17 | 2003-04-24 | Schering Corporation | Piperidine- and piperazineacetamines as 17beta hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| US20030170292A1 (en) | 2001-11-09 | 2003-09-11 | National University Of Singapore | Methods for preparing an estrogenic preparation and isolated estrogenic compounds from a plant and uses thereof |
| WO2003101972A1 (en) | 2002-05-30 | 2003-12-11 | The Scripps Research Institute | Copper-catalysed ligation of azides and acetylenes |
| WO2004046111A1 (en) | 2002-11-18 | 2004-06-03 | Schering Corporation | 17beta-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| WO2004060488A1 (en) | 2002-12-17 | 2004-07-22 | Schering Corporation | 17 beta-hydroxysteroid dehydrogenase type 3 inhibitors for the treatment of androgen dependent diseases |
| WO2004080271A2 (en) | 2003-03-14 | 2004-09-23 | Pirkko Vihko | Method for prognosticating the progress of breast cancer and compounds useful for prevention or treatment thereof |
| WO2004085345A2 (en) | 2003-03-21 | 2004-10-07 | Yale University | 15α-SUBSTITUTED ESTRADIOL CARBOXYLIC ACID ESTERS AS LOCALLY ACTIVE ESTROGENS |
| WO2004085457A2 (en) | 2003-03-24 | 2004-10-07 | Sterix Limited | 17beta-hydroxysteroid dehydrogenase inhibitors |
| WO2004110459A1 (en) | 2003-06-10 | 2004-12-23 | Solvay Pharmaceuticals B.V. | Thiophenepyrimidinones as 17-beta-hydroxysteroid dehydrogenase inhibitors |
| WO2005032527A2 (en) | 2003-06-10 | 2005-04-14 | Solvay Pharmaceuticals B.V. | Benzo (4, 5) thieno (2, 3-d) pyrimidin-4-ones and their use in therapy |
| WO2005047303A2 (en) | 2003-11-12 | 2005-05-26 | Solvay Pharmaceuticals Gmbh | NOVEL 17β HYDROXYSTEROID DEHYDROGENASE TYPE I INHIBITORS |
| WO2005084295A2 (en) | 2004-03-01 | 2005-09-15 | Bristol-Myers Squibb Company | Fused heterotricyclic compounds as inhibitors of 17b-hydroxysteroid dehydrogenase 3 |
| US20060009434A1 (en) | 2004-07-02 | 2006-01-12 | Alexander Hillisch | New 2-substituted estra-1,3,5(10)-trien-17-ones as inhibitors of 17beta-hydroxy steroid dehydrogenase type 1 |
| WO2006003012A1 (en) | 2004-07-02 | 2006-01-12 | Schering Aktiengesellschaft | NOVEL 2-SUBSTITUTED D-HOMO-ESTRA-1,3,5(10)-TRIENES AS INHIBITORS OF 17ß-HYDROXYSTEROID DEHYDROGENASE TYPE 1 |
| WO2006003013A2 (en) | 2004-07-02 | 2006-01-12 | Bayer Schering Pharma Aktiengesellschaft | NOVEL 2-SUBSTITUTED ESTRA-1,3,5(10)-TRIEN-17-ONES USED IN THE FORM OF INHIBITORS OF 17β-HYDROXYSTEROIDDEHYDROGENASE OF TYPE 1 |
| US20060052461A1 (en) | 2004-07-02 | 2006-03-09 | Alexander Hillisch | New 2-substituted D-homo-estra-1,3,5(10)-trienes as inhibitors of 17beta-hydroxy steroid dehydrogenase type 1 |
| WO2006027347A1 (en) | 2004-09-08 | 2006-03-16 | N.V. Organon | 15β-SUBSTITUTED STEROIDS HAVING SELECTIVE ESTROGENIC ACTIVITY |
| WO2006032885A2 (en) | 2004-09-22 | 2006-03-30 | Sterix Limited | 2-substituted estrogen sulphamates for inhibition of steroid sulphatase |
| WO2006063585A1 (en) | 2004-12-13 | 2006-06-22 | Leo Pharma A/S | Triazole substituted aminobenzophenone compounds |
| WO2006063615A1 (en) | 2004-12-13 | 2006-06-22 | Solvay Pharmaceuticals Gmbh | NOVEL SUBSTITUTED THIOPHENEPYRIMIDINONE DERIVATIVES AS INHIBITORS OF 17β-HYDROXYSTEROID DEHYDROGENASE |
| US20060271710A1 (en) | 2005-05-24 | 2006-11-30 | Inventec Corporation | Switch device |
| WO2006125800A1 (en) | 2005-05-26 | 2006-11-30 | Solvay Pharmaceuticals Gmbh | 17β-HSD1 AND STS INHIBITORS |
Non-Patent Citations (51)
Also Published As
| Publication number | Publication date |
|---|---|
| US20080146531A1 (en) | 2008-06-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2125859B1 (en) | Estratriene derivatives and their uses as 17beta-hydroxysteroid dehydrogenase inhibitors | |
| US8435973B2 (en) | 17-beta HSD1 and STS inhibitors | |
| JP2753562B2 (en) | Novel 11-benzaldoxime-estradiene derivative, method for producing the same, and pharmaceutical preparation containing the compound | |
| KR100396902B1 (en) | 19-nor-prrenen derivatives and pharmaceutical compositions containing them | |
| EP1888615B1 (en) | 17 -hsd1 and sts inhibitors | |
| CN101568547B (en) | Substituted estratriene derivatives as 17βHSD inhibitors | |
| EP1685150B1 (en) | NOVEL 17beta HYDROXYSTEROID DEHYDROGENASE TYPE I INHIBITORS | |
| AU777048B2 (en) | 17beta-acyl-17alpha-propynyl-11beta-arylsteroids and their derivatives having agonist or antagonist hormonal properties | |
| US8288367B2 (en) | Substituted estratriene derivatives as 17BETA HSD inhibitors | |
| US20080249075A1 (en) | C11 Modified Retrosteroids as Progesterone Receptor Modulator Compounds | |
| CN1878786B (en) | Novel 17 beta-hydroxysteroid dehydrogenase type I inhibitors | |
| US8080540B2 (en) | Therapeutically active triazoles and their use | |
| CN101506221B (en) | Estratriene derivatives and their use as 17β-hydroxysteroid dehydrogenase inhibitors | |
| CN102105484A (en) | Estratriene derivatives comprising heterocyclic bioisosteres for the phenolic a-ring | |
| HK1131157B (en) | Estratriene derivatives and their uses as 17beta-hydroxysteroid dehydrogenase inhibitors | |
| HK1117538B (en) | 17β-HSD1 AND STS INHIBITORS | |
| HK1099309B (en) | Novel 17 hydroxysteroid dehydrogenase type 1 inhibitors | |
| HK1134298B (en) | Substituted estratrien derivatives as 17beta hsd inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: SOLVAY PHARMACEUTICALS GMBH, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:MESSINGER, JOSEF;SCHOEN, UWE;THOLE, HEINRICH-HUBERT;AND OTHERS;REEL/FRAME:020535/0249;SIGNING DATES FROM 20080218 TO 20080220 Owner name: SOLVAY PHARMACEUTICALS GMBH, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:MESSINGER, JOSEF;SCHOEN, UWE;THOLE, HEINRICH-HUBERT;AND OTHERS;SIGNING DATES FROM 20080218 TO 20080220;REEL/FRAME:020535/0249 |
|
| AS | Assignment |
Owner name: ABBOTT PRODUCTS GMBH, GERMANY Free format text: CHANGE OF NAME;ASSIGNOR:SOLVAY PHARMACEUTICALS GMBH;REEL/FRAME:027143/0877 Effective date: 20100216 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| FEPP | Fee payment procedure |
Free format text: MAINTENANCE FEE REMINDER MAILED (ORIGINAL EVENT CODE: REM.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| LAPS | Lapse for failure to pay maintenance fees |
Free format text: PATENT EXPIRED FOR FAILURE TO PAY MAINTENANCE FEES (ORIGINAL EVENT CODE: EXP.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20191220 |