US6346609B1 - Cationic azo-dyes - Google Patents
Cationic azo-dyes Download PDFInfo
- Publication number
- US6346609B1 US6346609B1 US09/807,999 US80799901A US6346609B1 US 6346609 B1 US6346609 B1 US 6346609B1 US 80799901 A US80799901 A US 80799901A US 6346609 B1 US6346609 B1 US 6346609B1
- Authority
- US
- United States
- Prior art keywords
- mono
- radical
- formula
- oxy
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 125000002091 cationic group Chemical group 0.000 title claims abstract description 11
- 239000000987 azo dye Substances 0.000 title claims abstract description 5
- 239000000203 mixture Substances 0.000 claims abstract description 12
- 238000004043 dyeing Methods 0.000 claims abstract description 8
- -1 oxy, imino Chemical group 0.000 claims description 253
- 238000005859 coupling reaction Methods 0.000 claims description 36
- 239000000975 dye Substances 0.000 claims description 36
- 230000008878 coupling Effects 0.000 claims description 35
- 238000010168 coupling process Methods 0.000 claims description 35
- 239000001257 hydrogen Substances 0.000 claims description 20
- 229910052739 hydrogen Inorganic materials 0.000 claims description 20
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 20
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 12
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 claims description 11
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 11
- 125000004454 (C1-C6) alkoxycarbonyl group Chemical group 0.000 claims description 8
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 claims description 8
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 8
- 125000005115 alkyl carbamoyl group Chemical group 0.000 claims description 8
- 150000004985 diamines Chemical class 0.000 claims description 8
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 8
- 229910052757 nitrogen Inorganic materials 0.000 claims description 7
- 238000006467 substitution reaction Methods 0.000 claims description 7
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 6
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 6
- 150000001989 diazonium salts Chemical class 0.000 claims description 6
- 238000000034 method Methods 0.000 claims description 6
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 claims description 5
- 239000012954 diazonium Substances 0.000 claims description 5
- 229910052736 halogen Inorganic materials 0.000 claims description 5
- 150000002367 halogens Chemical class 0.000 claims description 5
- 125000005153 alkyl sulfamoyl group Chemical group 0.000 claims description 4
- 150000001450 anions Chemical class 0.000 claims description 4
- 238000006149 azo coupling reaction Methods 0.000 claims description 4
- 125000002373 5 membered heterocyclic group Chemical group 0.000 claims description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 3
- 125000005842 heteroatom Chemical group 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 3
- 239000000463 material Substances 0.000 claims description 3
- NYGZLYXAPMMJTE-UHFFFAOYSA-M metanil yellow Chemical group [Na+].[O-]S(=O)(=O)C1=CC=CC(N=NC=2C=CC(NC=3C=CC=CC=3)=CC=2)=C1 NYGZLYXAPMMJTE-UHFFFAOYSA-M 0.000 claims description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 3
- 229910052760 oxygen Inorganic materials 0.000 claims description 3
- 239000001301 oxygen Substances 0.000 claims description 3
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 2
- 125000003161 (C1-C6) alkylene group Chemical group 0.000 claims description 2
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 2
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims 4
- 239000002861 polymer material Substances 0.000 claims 1
- 239000000758 substrate Substances 0.000 abstract description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 32
- 239000000243 solution Substances 0.000 description 17
- 150000002431 hydrogen Chemical class 0.000 description 14
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- 229960000583 acetic acid Drugs 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 8
- 150000003254 radicals Chemical class 0.000 description 8
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 8
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 7
- KJCVRFUGPWSIIH-UHFFFAOYSA-N 1-naphthol Chemical compound C1=CC=C2C(O)=CC=CC2=C1 KJCVRFUGPWSIIH-UHFFFAOYSA-N 0.000 description 6
- JJYPMNFTHPTTDI-UHFFFAOYSA-N 3-methylaniline Chemical compound CC1=CC=CC(N)=C1 JJYPMNFTHPTTDI-UHFFFAOYSA-N 0.000 description 6
- 239000000123 paper Substances 0.000 description 6
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 6
- VOWZNBNDMFLQGM-UHFFFAOYSA-N 2,5-dimethylaniline Chemical group CC1=CC=C(C)C(N)=C1 VOWZNBNDMFLQGM-UHFFFAOYSA-N 0.000 description 5
- JWAZRIHNYRIHIV-UHFFFAOYSA-N 2-naphthol Chemical compound C1=CC=CC2=CC(O)=CC=C21 JWAZRIHNYRIHIV-UHFFFAOYSA-N 0.000 description 5
- RXYPXQSKLGGKOL-UHFFFAOYSA-N CN1CCN(C)CC1 Chemical compound CN1CCN(C)CC1 RXYPXQSKLGGKOL-UHFFFAOYSA-N 0.000 description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 5
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- CWLKGDAVCFYWJK-UHFFFAOYSA-N 3-aminophenol Chemical compound NC1=CC=CC(O)=C1 CWLKGDAVCFYWJK-UHFFFAOYSA-N 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 4
- ZWRRCXDCXWEDRJ-UHFFFAOYSA-N CC.CC1=CC=CC(C)=C1 Chemical compound CC.CC1=CC=CC(C)=C1 ZWRRCXDCXWEDRJ-UHFFFAOYSA-N 0.000 description 4
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 4
- 239000000460 chlorine Substances 0.000 description 4
- 229910052801 chlorine Inorganic materials 0.000 description 4
- 239000005457 ice water Substances 0.000 description 4
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 4
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 3
- ALKYHXVLJMQRLQ-UHFFFAOYSA-N 3-Hydroxy-2-naphthoate Chemical compound C1=CC=C2C=C(O)C(C(=O)O)=CC2=C1 ALKYHXVLJMQRLQ-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- AFBPFSWMIHJQDM-UHFFFAOYSA-N N-methyl-N-phenylamine Natural products CNC1=CC=CC=C1 AFBPFSWMIHJQDM-UHFFFAOYSA-N 0.000 description 3
- IOVCWXUNBOPUCH-UHFFFAOYSA-M Nitrite anion Chemical compound [O-]N=O IOVCWXUNBOPUCH-UHFFFAOYSA-M 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 150000001412 amines Chemical class 0.000 description 3
- HNYOPLTXPVRDBG-UHFFFAOYSA-N barbituric acid Chemical compound O=C1CC(=O)NC(=O)N1 HNYOPLTXPVRDBG-UHFFFAOYSA-N 0.000 description 3
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- GGSUCNLOZRCGPQ-UHFFFAOYSA-N diethylaniline Chemical compound CCN(CC)C1=CC=CC=C1 GGSUCNLOZRCGPQ-UHFFFAOYSA-N 0.000 description 3
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 3
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 3
- LNOPIUAQISRISI-UHFFFAOYSA-N n'-hydroxy-2-propan-2-ylsulfonylethanimidamide Chemical compound CC(C)S(=O)(=O)CC(N)=NO LNOPIUAQISRISI-UHFFFAOYSA-N 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 235000010288 sodium nitrite Nutrition 0.000 description 3
- WZCQRUWWHSTZEM-UHFFFAOYSA-N 1,3-phenylenediamine Chemical compound NC1=CC=CC(N)=C1 WZCQRUWWHSTZEM-UHFFFAOYSA-N 0.000 description 2
- RUFPHBVGCFYCNW-UHFFFAOYSA-N 1-naphthylamine Chemical compound C1=CC=C2C(N)=CC=CC2=C1 RUFPHBVGCFYCNW-UHFFFAOYSA-N 0.000 description 2
- 125000004343 1-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C([H])([H])[H] 0.000 description 2
- UIAFKZKHHVMJGS-UHFFFAOYSA-N 2,4-dihydroxybenzoic acid Chemical compound OC(=O)C1=CC=C(O)C=C1O UIAFKZKHHVMJGS-UHFFFAOYSA-N 0.000 description 2
- NWGUMFLIGWXDAA-UHFFFAOYSA-N 2-(dimethylamino)ethyl 2-aminobenzoate Chemical compound CN(C)CCOC(=O)C1=CC=CC=C1N NWGUMFLIGWXDAA-UHFFFAOYSA-N 0.000 description 2
- OJPDDQSCZGTACX-UHFFFAOYSA-N 2-[n-(2-hydroxyethyl)anilino]ethanol Chemical compound OCCN(CCO)C1=CC=CC=C1 OJPDDQSCZGTACX-UHFFFAOYSA-N 0.000 description 2
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 description 2
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 2
- 125000004204 2-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C(OC([H])([H])[H])C([H])=C1[H] 0.000 description 2
- 125000005916 2-methylpentyl group Chemical group 0.000 description 2
- 229940018563 3-aminophenol Drugs 0.000 description 2
- 125000004179 3-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(Cl)=C1[H] 0.000 description 2
- NFTNTGFZYSCPSK-UHFFFAOYSA-N 3-hydroxynaphthalene-2-carboxamide Chemical compound C1=CC=C2C=C(O)C(C(=O)N)=CC2=C1 NFTNTGFZYSCPSK-UHFFFAOYSA-N 0.000 description 2
- QOXOZONBQWIKDA-UHFFFAOYSA-N 3-hydroxypropyl Chemical group [CH2]CCO QOXOZONBQWIKDA-UHFFFAOYSA-N 0.000 description 2
- 125000004801 4-cyanophenyl group Chemical group [H]C1=C([H])C(C#N)=C([H])C([H])=C1* 0.000 description 2
- HDHQZCHIXUUSMK-UHFFFAOYSA-N 4-hydroxy-2-quinolone Chemical compound C1=CC=C2C(O)=CC(=O)NC2=C1 HDHQZCHIXUUSMK-UHFFFAOYSA-N 0.000 description 2
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 2
- FNYDIAAMUCQQDE-UHFFFAOYSA-N 4-methylbenzene-1,3-diol Chemical compound CC1=CC=C(O)C=C1O FNYDIAAMUCQQDE-UHFFFAOYSA-N 0.000 description 2
- FDNOAJIYOUHVNE-UHFFFAOYSA-N CC.CC.[H][K]N=NC1=C(C(=O)CCC(C)CCC(=O)C2=CC=CC=C2N=N[K])C=CC=C1 Chemical compound CC.CC.[H][K]N=NC1=C(C(=O)CCC(C)CCC(=O)C2=CC=CC=C2N=N[K])C=CC=C1 FDNOAJIYOUHVNE-UHFFFAOYSA-N 0.000 description 2
- LDZQLHSSXBUEBS-UHFFFAOYSA-N CC.CC1=CC=CC(C)=C1.CN=NC1=C(C(=O)CCC(C)(C)C)C=CC=C1.II Chemical compound CC.CC1=CC=CC(C)=C1.CN=NC1=C(C(=O)CCC(C)(C)C)C=CC=C1.II LDZQLHSSXBUEBS-UHFFFAOYSA-N 0.000 description 2
- RTTKVNNZRGYCTK-UHFFFAOYSA-N CC=1C=C(O)N(C)C(=O)C=1C#N Chemical compound CC=1C=C(O)N(C)C(=O)C=1C#N RTTKVNNZRGYCTK-UHFFFAOYSA-N 0.000 description 2
- DYNWNNKAUGBOOZ-UHFFFAOYSA-N CNc1cc(N)ccc1 Chemical compound CNc1cc(N)ccc1 DYNWNNKAUGBOOZ-UHFFFAOYSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- OJGMBLNIHDZDGS-UHFFFAOYSA-N N-Ethylaniline Chemical compound CCNC1=CC=CC=C1 OJGMBLNIHDZDGS-UHFFFAOYSA-N 0.000 description 2
- KEQFTVQCIQJIQW-UHFFFAOYSA-N N-Phenyl-2-naphthylamine Chemical compound C=1C=C2C=CC=CC2=CC=1NC1=CC=CC=C1 KEQFTVQCIQJIQW-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 229920002125 Sokalan® Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000000664 diazo group Chemical group [N-]=[N+]=[*] 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000000976 ink Substances 0.000 description 2
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000010985 leather Substances 0.000 description 2
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 2
- LVWZTYCIRDMTEY-UHFFFAOYSA-N metamizole Chemical compound O=C1C(N(CS(O)(=O)=O)C)=C(C)N(C)N1C1=CC=CC=C1 LVWZTYCIRDMTEY-UHFFFAOYSA-N 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 2
- JIQNWFBLYKVZFY-UHFFFAOYSA-N methoxycyclohexatriene Chemical compound COC1=C[C]=CC=C1 JIQNWFBLYKVZFY-UHFFFAOYSA-N 0.000 description 2
- YVVBECLPRBAATK-UHFFFAOYSA-N methyl 3-hydroxynaphthalene-2-carboxylate Chemical compound C1=CC=C2C=C(O)C(C(=O)OC)=CC2=C1 YVVBECLPRBAATK-UHFFFAOYSA-N 0.000 description 2
- FZZQNEVOYIYFPF-UHFFFAOYSA-N naphthalene-1,6-diol Chemical compound OC1=CC=CC2=CC(O)=CC=C21 FZZQNEVOYIYFPF-UHFFFAOYSA-N 0.000 description 2
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- RNVCVTLRINQCPJ-UHFFFAOYSA-N o-toluidine Chemical compound CC1=CC=CC=C1N RNVCVTLRINQCPJ-UHFFFAOYSA-N 0.000 description 2
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 2
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 description 2
- RZXMPPFPUUCRFN-UHFFFAOYSA-N p-toluidine Chemical compound CC1=CC=C(N)C=C1 RZXMPPFPUUCRFN-UHFFFAOYSA-N 0.000 description 2
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 2
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- JEXVQSWXXUJEMA-UHFFFAOYSA-N pyrazol-3-one Chemical class O=C1C=CN=N1 JEXVQSWXXUJEMA-UHFFFAOYSA-N 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 125000000547 substituted alkyl group Chemical group 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 239000004753 textile Substances 0.000 description 2
- 125000002861 (C1-C4) alkanoyl group Chemical group 0.000 description 1
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 1
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 description 1
- HVYXNUMXKPQXFZ-UHFFFAOYSA-N 1,4-dimethyl-2-oxopyridine-3-carboxamide Chemical compound CC=1C=CN(C)C(=O)C=1C(N)=O HVYXNUMXKPQXFZ-UHFFFAOYSA-N 0.000 description 1
- TXJUTRJFNRYTHH-UHFFFAOYSA-N 1h-3,1-benzoxazine-2,4-dione Chemical compound C1=CC=C2C(=O)OC(=O)NC2=C1 TXJUTRJFNRYTHH-UHFFFAOYSA-N 0.000 description 1
- VOZKAJLKRJDJLL-UHFFFAOYSA-N 2,4-diaminotoluene Chemical compound CC1=CC=C(N)C=C1N VOZKAJLKRJDJLL-UHFFFAOYSA-N 0.000 description 1
- CZZZABOKJQXEBO-UHFFFAOYSA-N 2,4-dimethylaniline Chemical compound CC1=CC=C(N)C(C)=C1 CZZZABOKJQXEBO-UHFFFAOYSA-N 0.000 description 1
- NAZDVUBIEPVUKE-UHFFFAOYSA-N 2,5-dimethoxyaniline Chemical compound COC1=CC=C(OC)C(N)=C1 NAZDVUBIEPVUKE-UHFFFAOYSA-N 0.000 description 1
- UFFBMTHBGFGIHF-UHFFFAOYSA-N 2,6-dimethylaniline Chemical compound CC1=CC=CC(C)=C1N UFFBMTHBGFGIHF-UHFFFAOYSA-N 0.000 description 1
- MSNMEZSDLXYGAB-UHFFFAOYSA-N 2-(2,5-dichlorophenyl)-5-methyl-1h-pyrazol-3-one Chemical compound N1C(C)=CC(=O)N1C1=CC(Cl)=CC=C1Cl MSNMEZSDLXYGAB-UHFFFAOYSA-N 0.000 description 1
- GZCPEUOCUUNCLZ-UHFFFAOYSA-N 2-(3-methylanilino)ethanol Chemical compound CC1=CC=CC(NCCO)=C1 GZCPEUOCUUNCLZ-UHFFFAOYSA-N 0.000 description 1
- NBYLBWHHTUWMER-UHFFFAOYSA-N 2-Methylquinolin-8-ol Chemical compound C1=CC=C(O)C2=NC(C)=CC=C21 NBYLBWHHTUWMER-UHFFFAOYSA-N 0.000 description 1
- ZMXYNJXDULEQCK-UHFFFAOYSA-N 2-amino-p-cresol Chemical compound CC1=CC=C(O)C(N)=C1 ZMXYNJXDULEQCK-UHFFFAOYSA-N 0.000 description 1
- 125000000022 2-aminoethyl group Chemical group [H]C([*])([H])C([H])([H])N([H])[H] 0.000 description 1
- MWGATWIBSKHFMR-UHFFFAOYSA-N 2-anilinoethanol Chemical compound OCCNC1=CC=CC=C1 MWGATWIBSKHFMR-UHFFFAOYSA-N 0.000 description 1
- 125000006276 2-bromophenyl group Chemical group [H]C1=C([H])C(Br)=C(*)C([H])=C1[H] 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- 125000001731 2-cyanoethyl group Chemical group [H]C([H])(*)C([H])([H])C#N 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- UPHOPMSGKZNELG-UHFFFAOYSA-N 2-hydroxynaphthalene-1-carboxylic acid Chemical compound C1=CC=C2C(C(=O)O)=C(O)C=CC2=C1 UPHOPMSGKZNELG-UHFFFAOYSA-N 0.000 description 1
- 125000004200 2-methoxyethyl group Chemical group [H]C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 1
- JHIAOWGCGNMQKA-UHFFFAOYSA-N 2-methyl-8-quinolinamine Chemical compound C1=CC=C(N)C2=NC(C)=CC=C21 JHIAOWGCGNMQKA-UHFFFAOYSA-N 0.000 description 1
- ZTMADXFOCUXMJE-UHFFFAOYSA-N 2-methylbenzene-1,3-diol Chemical compound CC1=C(O)C=CC=C1O ZTMADXFOCUXMJE-UHFFFAOYSA-N 0.000 description 1
- LBLYYCQCTBFVLH-UHFFFAOYSA-M 2-methylbenzenesulfonate Chemical compound CC1=CC=CC=C1S([O-])(=O)=O LBLYYCQCTBFVLH-UHFFFAOYSA-M 0.000 description 1
- WAVOOWVINKGEHS-UHFFFAOYSA-N 3-(diethylamino)phenol Chemical compound CCN(CC)C1=CC=CC(O)=C1 WAVOOWVINKGEHS-UHFFFAOYSA-N 0.000 description 1
- LZZLHTLIUKXAPH-UHFFFAOYSA-N 3-amino-4-ethylphenol Chemical compound CCC1=CC=C(O)C=C1N LZZLHTLIUKXAPH-UHFFFAOYSA-N 0.000 description 1
- AVQFHKYAVVQYQO-UHFFFAOYSA-N 3-amino-4-hydroxybenzenesulfonamide Chemical compound NC1=CC(S(N)(=O)=O)=CC=C1O AVQFHKYAVVQYQO-UHFFFAOYSA-N 0.000 description 1
- NUNAWQZKZVVELQ-UHFFFAOYSA-N 3-amino-4-methylphenol Chemical compound CC1=CC=C(O)C=C1N NUNAWQZKZVVELQ-UHFFFAOYSA-N 0.000 description 1
- 125000006275 3-bromophenyl group Chemical group [H]C1=C([H])C(Br)=C([H])C(*)=C1[H] 0.000 description 1
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 description 1
- 125000004208 3-hydroxyphenyl group Chemical group [H]OC1=C([H])C([H])=C([H])C(*)=C1[H] 0.000 description 1
- 125000004042 4-aminobutyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])N([H])[H] 0.000 description 1
- 125000004800 4-bromophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Br 0.000 description 1
- WQMPZIYHTPLJJD-UHFFFAOYSA-N 4-ethyl-2-hydroxy-6-oxo-1h-pyridine-3-carbonitrile Chemical compound CCC1=CC(O)=NC(O)=C1C#N WQMPZIYHTPLJJD-UHFFFAOYSA-N 0.000 description 1
- FTNQYYSBLXTZJN-UHFFFAOYSA-N 4-ethyl-2-hydroxy-6-oxo-1h-pyridine-3-carboxamide Chemical compound CCC1=CC(O)=NC(O)=C1C(N)=O FTNQYYSBLXTZJN-UHFFFAOYSA-N 0.000 description 1
- 125000004860 4-ethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 1
- SXIFAEWFOJETOA-UHFFFAOYSA-N 4-hydroxy-butyl Chemical group [CH2]CCCO SXIFAEWFOJETOA-UHFFFAOYSA-N 0.000 description 1
- 125000004203 4-hydroxyphenyl group Chemical group [H]OC1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- LVQFHDAKZHGEAJ-UHFFFAOYSA-M 4-methylbenzenesulfonate Chemical compound [CH2]C1=CC=C(S([O-])(=O)=O)C=C1 LVQFHDAKZHGEAJ-UHFFFAOYSA-M 0.000 description 1
- FTOAOBMCPZCFFF-UHFFFAOYSA-N 5,5-diethylbarbituric acid Chemical compound CCC1(CC)C(=O)NC(=O)NC1=O FTOAOBMCPZCFFF-UHFFFAOYSA-N 0.000 description 1
- LAOZSCRCYVBSJA-UHFFFAOYSA-N 5,5-dimethyl-1,3-diazinane-2,4,6-trione Chemical compound CC1(C)C(=O)NC(=O)NC1=O LAOZSCRCYVBSJA-UHFFFAOYSA-N 0.000 description 1
- DBFYESDCPWWCHN-UHFFFAOYSA-N 5-amino-2-methylphenol Chemical compound CC1=CC=C(N)C=C1O DBFYESDCPWWCHN-UHFFFAOYSA-N 0.000 description 1
- SCUCYJQORYOWAO-UHFFFAOYSA-N 6-hydroxy-1-methyl-4-methylsulfonylpyridin-2-one Chemical compound CN1C(O)=CC(S(C)(=O)=O)=CC1=O SCUCYJQORYOWAO-UHFFFAOYSA-N 0.000 description 1
- 239000005725 8-Hydroxyquinoline Substances 0.000 description 1
- WREVVZMUNPAPOV-UHFFFAOYSA-N 8-aminoquinoline Chemical compound C1=CN=C2C(N)=CC=CC2=C1 WREVVZMUNPAPOV-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 244000198134 Agave sisalana Species 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- HUHXJNUIZXRPTR-UHFFFAOYSA-N C.C.C.C.C1=CC2=C(C=C1)C=CC=C2.C1=CC2=C(C=C1)N=CC=C2.CC.CC.CC.CC.CC.CC.CC1=C(C)C(=O)N(C)C(O)=C1.CC1=CC=CC(C)=C1.CC1=NN(C)C(=O)C1.CN1C(=O)CC(=O)N(C)C1=O Chemical compound C.C.C.C.C1=CC2=C(C=C1)C=CC=C2.C1=CC2=C(C=C1)N=CC=C2.CC.CC.CC.CC.CC.CC.CC1=C(C)C(=O)N(C)C(O)=C1.CC1=CC=CC(C)=C1.CC1=NN(C)C(=O)C1.CN1C(=O)CC(=O)N(C)C1=O HUHXJNUIZXRPTR-UHFFFAOYSA-N 0.000 description 1
- CMOAORKVSASOCO-UHFFFAOYSA-N C.CC1=C(N2C=CN(C)=C2)C(O)=NC(O)=C1 Chemical compound C.CC1=C(N2C=CN(C)=C2)C(O)=NC(O)=C1 CMOAORKVSASOCO-UHFFFAOYSA-N 0.000 description 1
- ZWOCJMFUNZKPDZ-GILIAAGRSA-N C/N=N\C1=CC=CC=C1C(=O)OCCN(C)CCOC(=O)C1=CC=CC=C1/N=N/C1=CC=C(O)C(C)=C1O Chemical compound C/N=N\C1=CC=CC=C1C(=O)OCCN(C)CCOC(=O)C1=CC=CC=C1/N=N/C1=CC=C(O)C(C)=C1O ZWOCJMFUNZKPDZ-GILIAAGRSA-N 0.000 description 1
- BFAJBKIVTOCBRD-UHFFFAOYSA-N CC(=O)C1=CC=C(O)N(C)C1=O Chemical compound CC(=O)C1=CC=C(O)N(C)C1=O BFAJBKIVTOCBRD-UHFFFAOYSA-N 0.000 description 1
- JOSOQHNHCQSNMJ-QQSNTVJESA-N CC(=O)C1=CC=CC=C1/N=N/C1=C(O)C=CC(/N=N\C2=C(C(=O)OCCN(C)CCOC(=O)C3=C(/N=N/C4=C(O)C(/N=N/C5=C(C(=O)OCCN(C)C)C=CC=C5)=C(O)C=C4)C=CC=C3)C=CC=C2)=C1O Chemical compound CC(=O)C1=CC=CC=C1/N=N/C1=C(O)C=CC(/N=N\C2=C(C(=O)OCCN(C)CCOC(=O)C3=C(/N=N/C4=C(O)C(/N=N/C5=C(C(=O)OCCN(C)C)C=CC=C5)=C(O)C=C4)C=CC=C3)C=CC=C2)=C1O JOSOQHNHCQSNMJ-QQSNTVJESA-N 0.000 description 1
- DLWYLUPTPKHZMJ-UHFFFAOYSA-M CC.CC(C)(C)CCC(=O)C1=C(N)C=CC=C1.[V]I Chemical compound CC.CC(C)(C)CCC(=O)C1=C(N)C=CC=C1.[V]I DLWYLUPTPKHZMJ-UHFFFAOYSA-M 0.000 description 1
- LIBJCQJXZXJJQA-UHFFFAOYSA-M CC.CC.CC.CC.CC.CC1=CC=CC(C)=C1.CC1=CC=CC(C)=C1.CN=NC1=CC=CC=C1C(=O)CCC(C)CCC(=O)C1=C(N=NC)C=CC=C1.[V]I Chemical compound CC.CC.CC.CC.CC.CC1=CC=CC(C)=C1.CC1=CC=CC(C)=C1.CN=NC1=CC=CC=C1C(=O)CCC(C)CCC(=O)C1=C(N=NC)C=CC=C1.[V]I LIBJCQJXZXJJQA-UHFFFAOYSA-M 0.000 description 1
- GFUFQHYFFJIODH-UHFFFAOYSA-M CC.CC.CC.CC.CC.CC1=CC=CC(C)=C1.CC1=CC=CC(C)=C1.CN=NC1=CC=CC=C1C(=O)CCN(C)CCC(=O)C1=C(N=NC)C=CC=C1.[V]I Chemical compound CC.CC.CC.CC.CC.CC1=CC=CC(C)=C1.CC1=CC=CC(C)=C1.CN=NC1=CC=CC=C1C(=O)CCN(C)CCC(=O)C1=C(N=NC)C=CC=C1.[V]I GFUFQHYFFJIODH-UHFFFAOYSA-M 0.000 description 1
- MMLDYWHGRXPHDK-UHFFFAOYSA-N CC.CC.CC1=C(C(=O)CCN(C)CCC(=O)C2=CC=CC=C2N)C=CC=C1.[V] Chemical compound CC.CC.CC1=C(C(=O)CCN(C)CCC(=O)C2=CC=CC=C2N)C=CC=C1.[V] MMLDYWHGRXPHDK-UHFFFAOYSA-N 0.000 description 1
- QTDOKYBZBSWHMB-UHFFFAOYSA-N CC.CC.CC1=C(C(=O)CCNCCC(=O)C2=CC=CC=C2N)C=CC=C1.[V] Chemical compound CC.CC.CC1=C(C(=O)CCNCCC(=O)C2=CC=CC=C2N)C=CC=C1.[V] QTDOKYBZBSWHMB-UHFFFAOYSA-N 0.000 description 1
- TVDFXHMSANVKCR-UHFFFAOYSA-N CC.CC.CCC.CCCO.CCCO.OCCCCCO Chemical compound CC.CC.CCC.CCCO.CCCO.OCCCCCO TVDFXHMSANVKCR-UHFFFAOYSA-N 0.000 description 1
- FBILMYHPBCEDLF-UHFFFAOYSA-N CC.CC.C[K]N=NC1=C(C(=O)CCN(C)CCC(=O)C2=CC=CC=C2N=N[K])C=CC=C1 Chemical compound CC.CC.C[K]N=NC1=C(C(=O)CCN(C)CCC(=O)C2=CC=CC=C2N=N[K])C=CC=C1 FBILMYHPBCEDLF-UHFFFAOYSA-N 0.000 description 1
- YRGYYQCOWUULNF-UHFFFAOYSA-N CC1=C(C#N)C(O)=NC(O)=C1 Chemical compound CC1=C(C#N)C(O)=NC(O)=C1 YRGYYQCOWUULNF-UHFFFAOYSA-N 0.000 description 1
- UWQQPHNUTCVZMH-UHFFFAOYSA-N CC=1C=C(O)N(CCO)C(=O)C=1C(N)=O Chemical compound CC=1C=C(O)N(CCO)C(=O)C=1C(N)=O UWQQPHNUTCVZMH-UHFFFAOYSA-N 0.000 description 1
- IVEOCYLKEXGIPE-UHFFFAOYSA-N CCN1C(O)=CC(C)=C(C(N)=O)C1=O Chemical compound CCN1C(O)=CC(C)=C(C(N)=O)C1=O IVEOCYLKEXGIPE-UHFFFAOYSA-N 0.000 description 1
- XAJYTPYVHNOOMZ-KUPVJLDFSA-N CN(CCOC(=O)C1=C(/N=N\C2=C(O)C=C(O)C=C2)C=CC=C1)CCOC(=O)C1=C(/N=N/C2=C(O)C=C(O)C=C2)C=CC=C1 Chemical compound CN(CCOC(=O)C1=C(/N=N\C2=C(O)C=C(O)C=C2)C=CC=C1)CCOC(=O)C1=C(/N=N/C2=C(O)C=C(O)C=C2)C=CC=C1 XAJYTPYVHNOOMZ-KUPVJLDFSA-N 0.000 description 1
- DXJYXGPBSUWBLX-UHFFFAOYSA-N CNC(c1cc(cccc2)c2cc1O)=O Chemical compound CNC(c1cc(cccc2)c2cc1O)=O DXJYXGPBSUWBLX-UHFFFAOYSA-N 0.000 description 1
- 244000025254 Cannabis sativa Species 0.000 description 1
- 235000012766 Cannabis sativa ssp. sativa var. sativa Nutrition 0.000 description 1
- 235000012765 Cannabis sativa ssp. sativa var. spontanea Nutrition 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 240000000491 Corchorus aestuans Species 0.000 description 1
- 235000011777 Corchorus aestuans Nutrition 0.000 description 1
- 235000010862 Corchorus capsularis Nutrition 0.000 description 1
- 229920000742 Cotton Polymers 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 240000006240 Linum usitatissimum Species 0.000 description 1
- 235000004431 Linum usitatissimum Nutrition 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- FGOFNVXHDGQVBG-UHFFFAOYSA-N N-(2-methoxyphenyl)acetamide Chemical compound COC1=CC=CC=C1NC(C)=O FGOFNVXHDGQVBG-UHFFFAOYSA-N 0.000 description 1
- XQVWYOYUZDUNRW-UHFFFAOYSA-N N-Phenyl-1-naphthylamine Chemical compound C=1C=CC2=CC=CC=C2C=1NC1=CC=CC=C1 XQVWYOYUZDUNRW-UHFFFAOYSA-N 0.000 description 1
- LKDKFADINSRVMX-UHFFFAOYSA-N NCCNC(=O)C1=CC2=CC=CC=C2C=C1O Chemical compound NCCNC(=O)C1=CC2=CC=CC=C2C=C1O LKDKFADINSRVMX-UHFFFAOYSA-N 0.000 description 1
- VYHNSPUVKZPCDZ-UHFFFAOYSA-N O=C(NCCO)C1=CC2=CC=CC=C2C=C1O Chemical compound O=C(NCCO)C1=CC2=CC=CC=C2C=C1O VYHNSPUVKZPCDZ-UHFFFAOYSA-N 0.000 description 1
- BMRLCWFNMFDURW-UHFFFAOYSA-N O=C1C(C(N)=O)=C(C)C=C(O)N1C1=CC=CC=C1 Chemical compound O=C1C(C(N)=O)=C(C)C=C(O)N1C1=CC=CC=C1 BMRLCWFNMFDURW-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- 238000006887 Ullmann reaction Methods 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 235000013334 alcoholic beverage Nutrition 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 125000003282 alkyl amino group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- IYABWNGZIDDRAK-UHFFFAOYSA-N allene Chemical group C=C=C IYABWNGZIDDRAK-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 125000004202 aminomethyl group Chemical group [H]N([H])C([H])([H])* 0.000 description 1
- 150000001449 anionic compounds Chemical class 0.000 description 1
- RWZYAGGXGHYGMB-UHFFFAOYSA-N anthranilic acid Chemical class NC1=CC=CC=C1C(O)=O RWZYAGGXGHYGMB-UHFFFAOYSA-N 0.000 description 1
- 229960002319 barbital Drugs 0.000 description 1
- 150000007656 barbituric acids Chemical class 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 125000000043 benzamido group Chemical group [H]N([*])C(=O)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 150000001555 benzenes Chemical class 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 1
- 229940077388 benzenesulfonate Drugs 0.000 description 1
- 229940114055 beta-resorcylic acid Drugs 0.000 description 1
- 229950011260 betanaphthol Drugs 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000009120 camo Nutrition 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 150000001767 cationic compounds Chemical class 0.000 description 1
- 235000005607 chanvre indien Nutrition 0.000 description 1
- ZSUMNJYQUKCOBV-UHFFFAOYSA-N chembl1469934 Chemical compound CCN1C(O)=CC(C)=C(C#N)C1=O ZSUMNJYQUKCOBV-UHFFFAOYSA-N 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004915 dibutylamino group Chemical group C(CCC)N(CCCC)* 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 125000001664 diethylamino group Chemical group [H]C([H])([H])C([H])([H])N(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 125000004914 dipropylamino group Chemical group C(CC)N(CCC)* 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 description 1
- DCCIFKZACWAKSD-UHFFFAOYSA-N ethyl 3-oxo-2-phenyl-1h-pyrazole-5-carboxylate Chemical compound N1C(C(=O)OCC)=CC(=O)N1C1=CC=CC=C1 DCCIFKZACWAKSD-UHFFFAOYSA-N 0.000 description 1
- ILNOSUPSMMAKNS-UHFFFAOYSA-N ethyl 5-oxopyrazole-3-carboxylate Chemical compound CCOC(=O)C1=CC(=O)N=N1 ILNOSUPSMMAKNS-UHFFFAOYSA-N 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- 235000015203 fruit juice Nutrition 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 239000011487 hemp Substances 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005935 hexyloxycarbonyl group Chemical group 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 125000005929 isobutyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])OC(*)=O 0.000 description 1
- 125000005932 isopentyloxycarbonyl group Chemical group 0.000 description 1
- 125000005928 isopropyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- 238000005304 joining Methods 0.000 description 1
- 229940018564 m-phenylenediamine Drugs 0.000 description 1
- 239000004579 marble Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- QLNWXBAGRTUKKI-UHFFFAOYSA-N metacetamol Chemical compound CC(=O)NC1=CC=CC(O)=C1 QLNWXBAGRTUKKI-UHFFFAOYSA-N 0.000 description 1
- XVAIDCNLVLTVFM-UHFFFAOYSA-N methacetin Chemical compound COC1=CC=C(NC(C)=O)C=C1 XVAIDCNLVLTVFM-UHFFFAOYSA-N 0.000 description 1
- RMIODHQZRUFFFF-UHFFFAOYSA-M methoxyacetate Chemical compound COCC([O-])=O RMIODHQZRUFFFF-UHFFFAOYSA-M 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- CRVGTESFCCXCTH-UHFFFAOYSA-N methyl diethanolamine Chemical compound OCCN(C)CCO CRVGTESFCCXCTH-UHFFFAOYSA-N 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- FBGJJTQNZVNEQU-UHFFFAOYSA-N n,3-dimethylaniline Chemical compound CNC1=CC=CC(C)=C1 FBGJJTQNZVNEQU-UHFFFAOYSA-N 0.000 description 1
- RBQWGHBZCHFUQU-UHFFFAOYSA-N n-(3-amino-4-methylphenyl)acetamide Chemical compound CC(=O)NC1=CC=C(C)C(N)=C1 RBQWGHBZCHFUQU-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- KDFFXYVOTKKBDI-UHFFFAOYSA-N n-ethylnaphthalen-1-amine Chemical compound C1=CC=C2C(NCC)=CC=CC2=C1 KDFFXYVOTKKBDI-UHFFFAOYSA-N 0.000 description 1
- AHEJURHRYJPYST-UHFFFAOYSA-N n-ethylnaphthalen-2-amine Chemical compound C1=CC=CC2=CC(NCC)=CC=C21 AHEJURHRYJPYST-UHFFFAOYSA-N 0.000 description 1
- ZUVBIBLYOCVYJU-UHFFFAOYSA-N naphthalene-1,7-diol Chemical compound C1=CC=C(O)C2=CC(O)=CC=C21 ZUVBIBLYOCVYJU-UHFFFAOYSA-N 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-M naphthalene-1-sulfonate Chemical compound C1=CC=C2C(S(=O)(=O)[O-])=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-M 0.000 description 1
- DFQICHCWIIJABH-UHFFFAOYSA-N naphthalene-2,7-diol Chemical compound C1=CC(O)=CC2=CC(O)=CC=C21 DFQICHCWIIJABH-UHFFFAOYSA-N 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- 125000005933 neopentyloxycarbonyl group Chemical group 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 229960003540 oxyquinoline Drugs 0.000 description 1
- 239000011087 paperboard Substances 0.000 description 1
- WXWCDTXEKCVRRO-UHFFFAOYSA-N para-Cresidine Chemical compound COC1=CC=C(C)C=C1N WXWCDTXEKCVRRO-UHFFFAOYSA-N 0.000 description 1
- 125000001148 pentyloxycarbonyl group Chemical group 0.000 description 1
- 125000003170 phenylsulfonyl group Chemical group C1(=CC=CC=C1)S(=O)(=O)* 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 229920002239 polyacrylonitrile Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 235000011056 potassium acetate Nutrition 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- WGYKZJWCGVVSQN-UHFFFAOYSA-N propylamine Chemical group CCCN WGYKZJWCGVVSQN-UHFFFAOYSA-N 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- LISFMEBWQUVKPJ-UHFFFAOYSA-N quinolin-2-ol Chemical compound C1=CC=C2NC(=O)C=CC2=C1 LISFMEBWQUVKPJ-UHFFFAOYSA-N 0.000 description 1
- MCJGNVYPOGVAJF-UHFFFAOYSA-N quinolin-8-ol Chemical compound C1=CN=C2C(O)=CC=CC2=C1 MCJGNVYPOGVAJF-UHFFFAOYSA-N 0.000 description 1
- 150000003248 quinolines Chemical class 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 229960001755 resorcinol Drugs 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 125000005930 sec-butyloxycarbonyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000010902 straw Substances 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 125000004213 tert-butoxy group Chemical group [H]C([H])([H])C(O*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005934 tert-pentyloxycarbonyl group Chemical group 0.000 description 1
- 229940066528 trichloroacetate Drugs 0.000 description 1
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 1
- 239000002351 wastewater Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B44/00—Azo dyes containing onium groups
- C09B44/02—Azo dyes containing onium groups containing ammonium groups not directly attached to an azo group
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0046—Mixtures of two or more azo dyes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10S—TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10S534/00—Organic compounds -- part of the class 532-570 series
- Y10S534/03—Polymeric azo compounds or azo compounds containing polymeric moieties
Definitions
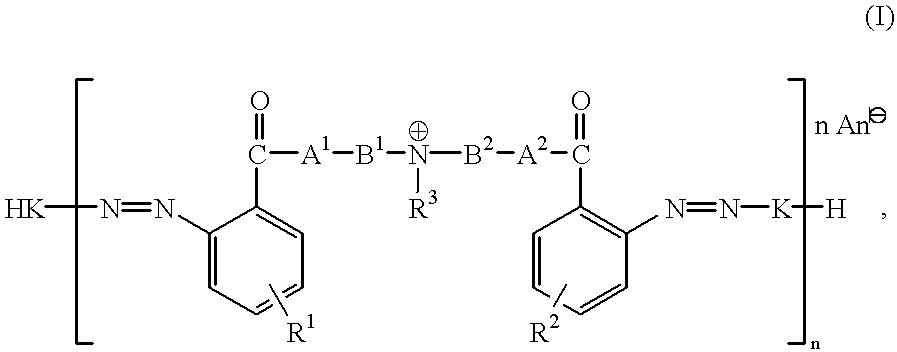
- the present invention relates to cationic azo dyes of the general formula I
- R 1 and R 2 are independently hydrogen, nitro, halogen, C 1 -C 6 -alkyl,
- R 3 is hydrogen, C 1 -C 6 -alkyl with or without interruption by 1 or 2 nonadjacent oxy, imino, C 1 -C 6 -alkylimino groups or by a
- a 1 and A 2 are independently oxy, imino or substituted or unsubstituted C 1 -C 6 -alkylimino,
- B 1 and B 2 are independently C 1 -C 6 -alkylene with or without interruption by 1 or 2 nonadjacent oxy, imino, substituted or unsubstituted C 1 -C 6 -alkylimino groups or by a
- n 1
- K is the radical of a coupling component or the radical of a monoazo dye of the formula
- R 4 ′ and R 5 ′ are independently hydroxyl, amino, morpholino or mono- or di(C 1 -C 6 )alkylamino, whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C 1 -C 6 -alkoxycarbonyl, carbamoyl or mono- or di(C 1 -C 6 )alkylcarbamoyl and uninterrupted or interrupted by oxy, and
- Q 1 , Q 2 and Q 3 are independently hydrogen, C 1 -C 10 -alkyl with or without interruption by from 1 to 4 nonadjacent oxy, imino and substituted or unsubstituted C 1 -C 4 -alkylimino groups and with or without hydroxyl, halogen or phenyl substitution, or Q 1 and Q 2 combine with the linking nitrogen atom to form a 5- or 6-membered heterocyclic radical which optionally contains nitrogen or oxygen as further hetero atom,
- n is from 2 to 8 when K is the radical of a coupling component of the formula IIIa′
- R 6 ′ is hydrogen, C 1 -C 6 -alkoxy with or without oxy interruption in its alkyl radical, mono- or di(C 1 -C 6 )alkylamino, C 1 -C 6 -alkoxycarbonyl, carbamoyl, mono- or di(C 1 -C 6 )alkylcarbamoyl, sulfamoyl, mono- or di(C 1 -C 6 )alkylsulfamoyl,
- DE-A-2 218 645 describes water-insoluble trisazo pigments which are monoazo dyes linked via triethanolamine.
- DE-A-2 160 590 describes anthranilic acid derivatives bridged with a diethanolamine via the ester group. These diamines are useful for preparing polyurethanes.
- Alkyl and alkylene appearing in the abovementioned formula may be straight-chain or branched.
- Substituted alkyl appearing in the abovementioned formula may have as substituents for example hydroxyl and methoxy.
- the number of substituents in the substituted alkyl is generally 1 or 2.
- Q 1 and Q 2 combining with the joining nitrogen atom to form a 5- or 6-membered heterocyclic radical which optionally contains nitrogen or oxygen as further hetero atom may be saturated radicals such as pyrrolidinyl, piperidinyl, morpholinyl, piperazinyl with or without methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl, 2-hydroxyethyl or 2- or 3-hydroxypropyl substitution on the nitrogen.
- unsaturated radicals such as pyrrolyl, pyrazolyl, oxazolyl, isooxazolyl or imidazolyl with or without methyl, ethyl, propyl or butyl substitution in 2- and/or 4-position or N-3(C 1 -C 4 )alkylimidazolyl with or without methyl, ethyl, propyl or butyl substitution in 2- and/or 4-position.
- Halogen is fluorine, chlorine or bromine.
- R 1 , R 2 , R 3 , Q 1 , Q 2 and Q 3 are each for example methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, neopentyl, tert-pentyl, hexyl or 2-methylpentyl.
- R 3 , Q 1 , Q 2 and Q 3 may each also be for example 2-methoxyethyl, 2-ethoxyethyl, 2-propoxyethyl, 2-isopropoxyethyl, 2-butoxyethyl, 2- or 3-methoxypropyl, 2- or 3-ethoxypropyl, 2- or 3-propoxypropyl, 2- or 4-methoxybutyl, 2- or 4-ethoxybutyl, 3,6-dioxaheptyl, 3,6-dioxaoctyl, 3,7-dioxaoctyl, 4,7-dioxaoctyl, N,N-dimethylaminoethyl, N,N-diethylaminoethyl, 3-(N,N-dimethylamino)propyl, 3-(N,N-diethylamino)propyl, 2-methylaminoethyl, 2-ethylaminoethyl, 3-
- Q 1 , Q 2 and Q 3 may each also be for example heptyl, octyl, 2-ethylhexyl, isooctyl, nonyl, isononyl, decyl (the above designations isooctyl, isononyl and isodecyl are trivial names derived from the alcohols obtained by the oxo process - cf.
- A is for example methylimino, ethylimino, propylimino, isopropylimino, butylimino, isobutylimino, sec-butylimino, tert-butylimino, pentylimino, isopentylimino, neopentylimino, tert-pentylimino, hexylimino or 2-methylpentylimino.
- B 1 and B 2 are each for example methylene, ethylene, 1,2- or 1,3-propylene, 1,2-, 2,3- or 1,4-butylene, (CH 2 ) 2 O(CH 2 ) 2 , (CH 2 ) 3 O(CH 2 ) 2 , (CH 2 ) 3 O(CH 2 ) 3 , (CH 2 ) 2 O(CH 2 ) 2 O(CH 2 ) 2 , (CH 2 ) 2 NH(CH 2 ) 2 , (CH 2 ) 3 NH(CH 2 ) 2 , (CH 2 ) 2 NH(CH 2 ) 2 NH(CH 2 ) 2 NH(CH 2 ) 2 ,
- An ⁇ is derived for example from the following anions: fluoride, chloride, bromide, iodide, sulfate, phosphate, formate, acetate, propionate, mono-, di- or trichloroacetate, lactate, methoxyacetate, citrate, succinate, methanesulfonate, benzenesulfonate, 2- or 4-methylbenzenesulfonate or naphthalenesulfonate.
- Suitable coupling components KH are compounds of the benzene, naphthalene, quinolene, pyridone, barbituric acid or pyrazolone series and correspond for example to the compounds of the formulae III a-f
- R 4 is hydroxyl, amino, morpholino, mono- or di(C 1 -C 6 )alkylamino whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C 1 -C 6 -alkoxycarbonyl, carbamoyl or mono or di(C 1 -C 6 )alkylcarbamoyl and are uninterrupted or interrupted by oxy or is C 1 -C 6 -alkoxy,
- R 5 is hydrogen, C 1 -C 6 -alkyl or R 4 ,
- R 6 is hydrogen, C 1 -C 6 -alkyl, C 1 -C 6 -alkoxy with or without oxy interruption in its alkyl radical, mono or di(C 1 -C 6 )alkylamino, C 1 -C 6 -alkoxycarbonyl, carbamoyl, mono or di(C 1 -C 6 )alkylcarbamoyl whose alkyl radicals are unsubstituted or substituted by hydroxyl or amino and are uninterrupted or oxy interrupted, sulfamoyl, mono or di(C 1 -C 6 )alkylsulfamoyl whose alkyl radicals are unsubstituted or hydroxyl or amino substituted and uninterrupted or oxy interrupted, or is carboxyl,
- R 7 is hydroxyl, amino, mono or di(C 1 -C 12 )alkylamino, cyclohexylamino,
- R 8 is hydrogen, hydroxyl, amino, formylamino, acetylamino, C 1 -C 6 -alkyl,
- R 9 is hydrogen, C 1 -C 6 -alkyl, phenyl, hydroxyl, cyano, acetyl, benzoyl, methoxycarbonyl, carbamoyl,
- R 10 is hydrogen, chlorine, bromine, acetylamino, amino, nitro, sulfamoyl, methylsulfonyl, phenylsulfonyl, C 1 -C 4 -alkoxycarbonyl, C 1 -C 4 -alkanoyl, benzoyl, carbamoyl, cyano, N-methylimidazolyl, pyridinio,
- R 11 is hydrogen, C 1 -C 6 -alkyl with or without phenyl, hydroxyl, amino, C 1 -C 6 -alkoxy, acetylamino, benzoylamino or cyano substitution, cyclohexyl, phenyl with or without benzoylamino, acetylamino, methyl, methoxy, cyano or chlorine substitution, or amino which is substituted by phenyl, C 1 -C 6 -alkyl, C 1 -C 6 -alkanoyl or benzoyl,
- R 12 or R 13 are independently hydrogen or C 1 -C 6 -alkyl
- R 14 is methyl, C 1 -C 4 -alkoxycarbonyl or phenyl
- R 15 is hydrogen, C 1 -C 6 -alkyl, cyclohexyl, benzyl or phenyl which is unsubstituted or from singly to triply substituted by fluorine, chlorine, bromine, C 1 -C 4 -alkyl, C 1 -C 4 -alkoxy, cyano, acetyl, acetylamino, hydroxyl, sulfamoyl or carbamoyl.
- R 4 , R 6 and R 7 are each for example mono- or dimethylamino, mono- or diethylamino, mono- or dipropylamino, mono- or diisopropylamino, mono- or dibutylamino, mono- or dipentylamino, mono- or diisopentylamino, mono- or dineopentylamino, mono- or dihexylamino, mono- or di-2-methylpentylamino, N-methyl-n-butylamino, N-ethyl-n-butylamino.
- R 4 may also be for example mono- or di(2-hydroxyethyl)amino, mono-or di(2-hydroxypropyl)amino, mono- or di(3-hydroxypropyl)amino, N-methyl-2-hydroxyethylamino, N-methyl-2-hydroxypropylamino, N-methyl-3-hydroxypropylamino, N-ethyl-2-hydroxyethylamino, N-ethyl-2-hydroxypropylamino, N-ethyl-3-hydroxypropylamino, N-propyl-2-hydroxyethylamino, N-propyl-2-hydroxypropylamino, N-propyl-3-hydroxypropylamino, N-isopropyl-2-hydroxyethylamino, -2-hydroxypropylamino, -3-hydroxypropylamino, mono- or di(2-aminoethyl)amino, mono- or di(2-aminopropyl)amino, mono-
- R 5 , R 6 , R 8 , R 9 , R 11 , R 12 , R 13 and R 15 are each for example methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, neopentyl, tert-pentyl, hexyl or 2-methylpentyl.
- R 11 is hydroxy-C 1 -C 4 -alkyl such as hydroxymethyl, 1-hydroxyeth-1-yl, 2-hydroxyeth-1-yl, 1-hydroxy-prop-1-yl, 2-hydroxyprop-1-yl, 3-hydroxyprop-1-yl, 1-hydroxy-prop-2-yl, 2-hydroxyprop-2-yl, 1-hydroxybut-1-yl, 2-hydroxybut-1-yl, 3-hydroxybut-1-yl, 4-hydroxybut-1-yl, 1-hydroxybut-2-yl, 2-hydroxybut-2-yl, 1-hydroxybut-3-yl, 2-hydroxybut-3-yl, 1-hydroxy-2-methyl-prop-3-yl, 2-hydroxy-2-methyl-prop-3-yl, 3-hydroxy-2-methyl-prop-3-yl, 2-hydroxymethyl-prop-2-yl, cyanomethyl, cyanoethyl, cyanopropyl or cyanobutyl.
- R 11 may also be for example benzyl, 1-phenylethyl, 2-phenylethyl, 1-phenylprop-1-yl, 2-phenylprop-1-yl, 3-phenylprop-1-yl, 1-phenylbut-1-yl, 2-phenylbut-1-yl, 3-phenylbut-1-yl, 4-phenylbut-1-yl, 1-phenylbut-2-yl, 2-phenylbut-2-yl, 3-phenylbut-2-yl, 4-phenylbut-2-yl, 1-(phenylmethyl)-eth-1-yl, 1-(phenylmethyl)-1-(methyl)-eth-1-yl, 1-(phenylmethyl)-prop-1-yl, preferably benzyl and 2-phenylethyl, methoxymethyl, ethoxymethyl, n-propoxymethyl, (1-methylethoxy)methyl, n-butoxymethyl, (1-methylpropoxy)methyl, (2-methylpropoxy)methyl, (1,
- R 10 is for example formyl, acetyl, propionyl, butyryl or isobutyryl.
- R 6 is for example mono- or dimethylcarbamoyl, mono- or diethylcarbamoyl, mono- or dipropylcarbamoyl, mono- or dibutylcarbamoyl, mono- or dipentylcarbamoyl or mono- or dihexylcarbamoyl.
- R 4 and R 6 may each be for example methoxy, ethoxy, propoxy, isopropoxy, butoxy, sec-butoxy, isobutoxy or tert-butoxy.
- R 6 may also be for example methoxyethyloxy, methoxypropyloxy, ethoxyethyloxy, ethoxypropyloxy or propoxypropyloxy.
- R 15 is for example 2-, 3- or 4-fluorophenyl, 2-, 3- or 4-chlorophenyl, 2-, 3- or 4-bromophenyl, 2-, 3- or 4-methylphenyl, 2-, 3- or 4-ethylphenyl, 2-, 3- or 4-methoxyphenyl, 2-, 3- or 4-cyanophenyl, 2-, 3- or 4-hydroxyphenyl, 2-, 3- or 4-acetylphenyl, 2-, 3- or 4-acetylaminophenyl, 2-, 3- or 4-sulfamoylphenyl or 2-, 3- or 4-carbamoylphenyl.
- R 6 , R 10 and R 14 may each also be for example methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl, pentoxycarbonyl, isopentoxycarbonyl, neopentoxycarbonyl, tert-pentyloxycarbonyl, hexyloxycarbonyl or 2-methylpentyloxycarbonyl.
- R 6 may also be for example mono- or dimethylsulfamoyl, mono- or diethylsulfamoyl, mono- or dipropylsulfamoyl, mono- or dibutylsulfamoyl.
- R 11 may also be for example aminomethyl, 2-aminoethyl, 2- or 3-aminopropyl, 2- or 4-aminobutyl, acetylaminomethyl, 2-acetylaminoethyl, 2- or 3-acetylaminopropyl, 2- or 4-acetylaminobutyl, benzoylaminomethyl, 2-benzoylaminoethyl, 2- or 3-benzoylaminopropyl, 2- or 4-benzoylaminobutyl, 2-, 3- or 4-benzoylaminophenyl, 2-, 3- or 4-acetylaminophenyl, 2-, 3- or 4-methylphenyl, 2-, 3- or 4-methoxyphenyl, 2-, 3- or 4-cyanophenyl, 2-, 3- or 4-chlorophenyl, phenylamino, methylamino, ethylamino, propylamino, isopropylamino, butylamino, isobutylamin
- coupling components KH are recited by way of example.
- Examples of coupling components of the benzene series are resorcinol, 2- and 4-methylresorcinol, 1,3-phenylenediamine, 3-aminophenol, 4-methyl-3-aminophenol, 5-amino-2-methylphenol, 4-ethyl-3-aminophenol, 3-N,N-diethylaminophenol, 2,4-diaminotoluene, 2,4-dihydroxybenzoic acid, 4-methyl-2-aminophenol, 3-acetaminophenol, 3-amino-4-hydroxybenzenesulfonamide, o-, m- or p-toluidine, o-, m- or p-xylidine, 2,5-dimethoxyaniline, 2-methoxy-5-methylaniline, 3-amino-4-methylacetanilide, 2- or 4-methoxyacetanilide, N-methylaniline, N-methyl-m-tolui
- Coupling components of the naphthalene series are for example 1-naphthylamine, N-phenyl-1-naphthylamine, N-ethyl-1-naphthylamine, N-ethyl-2-naphthylamine, N-phenyl-2-naphthylamine, 1-naphthol, 2-naphthol, 2,5-dihydroxynaphthalene, 1,6-dihydroxynaphthalene, 1,7-dihydroxynaphthalene, 2,7-dihydroxynaphthalene, 2-hydroxynaphthalene-1-carboxylic acid, 2-hydroxynaphthalene-3-carboxylic acid, methyl 2-hydroxynaphthalene-3-carboxylate or 2-hydroxynaphthalene-3-carboxamide.
- Coupling components of the quinoline series are for example 8-hydroxyquinoline, 8-hydroxy-2-methylquinoline, 8-aminoquinoline, 8-amino-2-methylquinoline, 2-hydroxyquinoline and 2,4-dihydroxyquinoline.
- Pyridone coupling components are for example 1-ethyl-2-hydroxy-4-methyl-5-carbamoylpyrid-6-one, 1-(2′-hydroxyethyl)2-hydroxy-4-methyl-5-carbamoylpyrid-6-one, 1-phenyl-2-hydroxy-4-methyl-5-carbamoylpyrid-6-one, 1-ethyl-2-hydroxy-4-methyl-5-cyanopyrid-6-one, 1-methyl-2-hydroxy-4-methyl-5-cyanopyrid-6-one, 1-methyl-2-hydroxy-5-acetylpyrid-6-one, 1,4-dimethyl-2-hydroxy-5-cyanopyrid-6-one, 1,4-dimethyl-5-carbamoylpyrid-6-one, 2,6-dihydroxy-4-ethyl-5-cyanopyridine, 2-hydroxy-4-ethyl-5-carbamoylpyrid-6-one or 1-methyl-2-hydroxy-4-methylsulfonylpyrid-6-one.
- Suitable coupling components of the barbituric acid series are barbituric acid, dimethylbarbituric acid and diethylbarbituric acid.
- Suitable pyrazolone coupling components are for example 3-methyl-or 3-(C 1 -C 4 -alkoxycarbonyl)pyrazol-5-ones which, in 1-position, may bear hydrogen, unsubstituted or methyl-, ethyl-, fluorine-, chlorine-, bromine-, methoxy-, ethoxy-, cyano-, acetylamino-, hydroxyl-, carbamoyl- or sulfamoyl-substituted phenyl.
- Examples are 1-(2′,5′-dichlorophenyl)-3-methylpyrazol-5-one, ethyl 1-phenylpyrazol-5-one-3-carboxylate and ethyl pyrazol-5-one-3-carboxylate.
- B 1 and B 2 are each preferably C 2 -C 6 -alkylene with or without interruption by oxy or by C 1 -C 6 -alkylimino. B 1 and B 2 are preferably identical.
- Coupling components KH are preferably compounds of the formulae IIIa and IIIb. Preference is given in particular to dyes of the general formula IV
- n is from 1 to 8 and R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , A 1 , A 2 , B 1 , B 2 and An ⁇ are each as defined above.
- R 4 is hydroxyl, amino, mono- or dimethyl- or -ethylamino or mono- or dihydroxyethylamino and R 5 and R 6 are independently hydrogen, hydroxyl, amino or methyl.
- R 1 , R 2 , R 3 , A 1 , A 2 , B 1 and B 2 are each as defined above, can be conventionally diazotized and coupled with a coupling component KH.
- the pH is preferably within the range from 2 to 8.
- the diazonium salt solution is metered into the solution of the coupling component. It is generally possible to use a single coupling component and also mixtures of a plurality of coupling components, for example so as to shade the hue.
- oligomeric dyes are obtainable by azo coupling of 1 mol equivalent of diamine of the formula IV onto from 1 to 2, preferably onto from 1 to 1.5, especially from 1 to 1.2, mol equivalents of a coupling component of the formula IIIa′
- R 4 ′ and R 5 ′ are independently hydroxyl, amino, morpholino or mono-or di(C 1 -C 6 )alkylamino, whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C 1 -C 6 -alkoxycarbonyl, carbamoyl or mono- or di(C 1 -C 6 )alkylcarbamoyl and uninterrupted or interrupted by oxy, and
- R 6 ′ is hydrogen, C 1 -C 6 -alkoxy with or without oxy interruption in its alkyl radical, mono- or di(C 1 -C 6 )alkylamino, C 1 -C 6 -alkoxycarbonyl, carbamoyl, mono- or di(C 1 -C 6 )alkylcarbamoyl, sulfamoyl, mono- or di(C 1 -C 6 )alkylsulfamoyl.
- the coupling step is carried out at a pH above 2, preferably within the range from 3 to 7.
- the desired pH may be set and, if necessary, corrected during the coupling reaction by adding, for example, ammonium or alkali metal bases, e.g., sodium hydroxide, sodium carbonate, sodium acetate, potassium hydroxide, potassium carbonate or potassium acetate.
- ammonium or alkali metal bases e.g., sodium hydroxide, sodium carbonate, sodium acetate, potassium hydroxide, potassium carbonate or potassium acetate.
- the end point of the reaction will be reached not more than 2 hours after completion of the addition.
- Chain termination is effected by coupling onto a coupling component devoid of a reactive terminus or by decomposing the diazonium compounds.
- disazo dyes IV provides likewise preferred tetraazo dyes.
- the amines VI are generally known and are described for example EP-A-0 159 549.
- the dyes are very useful in the production of pulp-dyed, sized and unsized paper. They are likewise useful for dyeing paper by the dip process.
- novel dyes and their preparations color the papermaking wastewater only minimally, if at all, which is particularly favorable from the aspect of keeping the water courses clean. They are highly substantive, do not marble on paper and are substantially pH-insensitive. The dyeings on paper are notable for good lightfastness. On prolonged exposure to light the hue changes on-tone.
- the basic azo dyes of the formula I of the invention can be used alone, in mixtures with each or one another and together with other cationic or anionic compounds in the form of their solutions or in the form of powders or granules. They are advantageously useful for dyeing or printing polymeric material, especially papery materials such as paper and paperboard, but also cellulose, cotton, leather, bast fibers, hemp, flax, sisal, jute, coir, straw or anionically modified fibers and also in inks, including printing inks.
- polymers such as polyacrylic acids, polyacrylic acid derivatives, polyvinylamines, polyvinylamides, polyvinyl acetates, polyvinyl alcohols, polyvinylpyrrolidones, polysiloxanes or copolymers of the respective monomers.
- polymers such as polyacrylic acids, polyacrylic acid derivatives, polyvinylamines, polyvinylamides, polyvinyl acetates, polyvinyl alcohols, polyvinylpyrrolidones, polysiloxanes or copolymers of the respective monomers.
- oligomers of ethyleneimine, ethylene oxide or propylene oxide or derivatives thereof can be used.
- the dyed papers which exhibit good bleachability, are wetfast, not only with regard to water, but also with regard to milk, soapy water, sodium chloride solutions, fruit juices or sweetened mineral water, and are also stable to alcoholic drinks because of their good alcohol fastness.
- novel dyes can also be used for dyeing, padding or printing polyacrylonitrile textiles or anionically modified polyamide or polyester textiles.
- Example 1 was repeated with the coupling components recited in Table 1 to prepare the dyes indicated in the table.
- Example 2 was also suitable for reacting the dyes of Examples 1.12 and 1.13 with diazotized 2-dimethylaminoethyl 2-aminobenzoate.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Plural Heterocyclic Compounds (AREA)
- Coloring (AREA)
Abstract
Description
where
R1 and R2 are independently hydrogen, nitro, halogen, C1-C6-alkyl,
R3 is hydrogen, C1-C6-alkyl with or without interruption by 1 or 2 nonadjacent oxy, imino, C1-C6-alkylimino groups or by a
radical,
A1 and A2 are independently oxy, imino or substituted or unsubstituted C1-C6-alkylimino,
B1 and B2 are independently C1-C6-alkylene with or without interruption by 1 or 2 nonadjacent oxy, imino, substituted or unsubstituted C1-C6-alkylimino groups or by a
radical,
An⊖is the equivalent of an anion,
n is 1, and
where
R4′ and R5′ are independently hydroxyl, amino, morpholino or mono- or di(C1-C6)alkylamino, whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C1-C6-alkoxycarbonyl, carbamoyl or mono- or di(C1-C6)alkylcarbamoyl and uninterrupted or interrupted by oxy, and
Q1, Q2 and Q3 are independently hydrogen, C1-C10-alkyl with or without interruption by from 1 to 4 nonadjacent oxy, imino and substituted or unsubstituted C1-C4-alkylimino groups and with or without hydroxyl, halogen or phenyl substitution, or Q1 and Q2 combine with the linking nitrogen atom to form a 5- or 6-membered heterocyclic radical which optionally contains nitrogen or oxygen as further hetero atom,
and additionally n is from 2 to 8 when K is the radical of a coupling component of the formula IIIa′
where
R6′, is hydrogen, C1-C6-alkoxy with or without oxy interruption in its alkyl radical, mono- or di(C1-C6)alkylamino, C1-C6-alkoxycarbonyl, carbamoyl, mono- or di(C1-C6)alkylcarbamoyl, sulfamoyl, mono- or di(C1-C6)alkylsulfamoyl,
processes for their preparation, their use for dyeing and printing natural or synthetic substrates and also their mixtures.
DE-A-2 218 645 describes water-insoluble trisazo pigments which are monoazo dyes linked via triethanolamine.
DE-A-2 160 590 describes anthranilic acid derivatives bridged with a diethanolamine via the ester group. These diamines are useful for preparing polyurethanes.
It is an object of the present invention to provide novel dyes which have advantageous application properties, especially high affinities for the substrates used, and also good fastness to bleeding and lightfastness.
We have found that this object is achieved by the dyes of the formula I which are defined at the beginning.
Alkyl and alkylene appearing in the abovementioned formula may be straight-chain or branched.
Substituted alkyl appearing in the abovementioned formula may have as substituents for example hydroxyl and methoxy. The number of substituents in the substituted alkyl is generally 1 or 2.
Q1 and Q2 combining with the joining nitrogen atom to form a 5- or 6-membered heterocyclic radical which optionally contains nitrogen or oxygen as further hetero atom may be saturated radicals such as pyrrolidinyl, piperidinyl, morpholinyl, piperazinyl with or without methyl, ethyl, propyl, isopropyl, n-butyl, isobutyl, sec-butyl, 2-hydroxyethyl or 2- or 3-hydroxypropyl substitution on the nitrogen. Further possibilities are unsaturated radicals such as pyrrolyl, pyrazolyl, oxazolyl, isooxazolyl or imidazolyl with or without methyl, ethyl, propyl or butyl substitution in 2- and/or 4-position or N-3(C1-C4)alkylimidazolyl with or without methyl, ethyl, propyl or butyl substitution in 2- and/or 4-position.
Halogen is fluorine, chlorine or bromine.
R1, R2, R3, Q1, Q2 and Q3 are each for example methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, neopentyl, tert-pentyl, hexyl or 2-methylpentyl.
R3, Q1, Q2 and Q3 may each also be for example 2-methoxyethyl, 2-ethoxyethyl, 2-propoxyethyl, 2-isopropoxyethyl, 2-butoxyethyl, 2- or 3-methoxypropyl, 2- or 3-ethoxypropyl, 2- or 3-propoxypropyl, 2- or 4-methoxybutyl, 2- or 4-ethoxybutyl, 3,6-dioxaheptyl, 3,6-dioxaoctyl, 3,7-dioxaoctyl, 4,7-dioxaoctyl, N,N-dimethylaminoethyl, N,N-diethylaminoethyl, 3-(N,N-dimethylamino)propyl, 3-(N,N-diethylamino)propyl, 2-methylaminoethyl, 2-ethylaminoethyl, 3-propylaminoethyl, 2- or 3-methylaminopropyl, 2- or 3-ethylaminopropyl or 2- or 3-propylaminopropyl.
Q1, Q2 and Q3 may each also be for example heptyl, octyl, 2-ethylhexyl, isooctyl, nonyl, isononyl, decyl (the above designations isooctyl, isononyl and isodecyl are trivial names derived from the alcohols obtained by the oxo process - cf. Ullmanns Encyklopädie der technischen Chemie, 4th edition, volume 7, pages 215 to 217, and also volume 11, pages 435 and 436), 2-hydroxyethyl, 2- or 3-hydroxypropyl, 2- or 4-hydroxybutyl, 2-chloroethyl, 2- or 3-chloropropyl, 2- or 4-chlorobutyl, benzyl, 1- or 2-phenylethyl, 2- or 3-butoxypropyl, 2- or 4-propoxybutyl, 4,8-dioxadecyl, 3,7-dioxanonyl, 4,7-dioxanonyl, 2- or 4-butoxybutyl, 3,6,9-trioxadecyl, 3,6,9-trioxaundecyl, 3,6,9-trioxadodecyl, 3,6,9,12-tetraoxatridecyl, 3,6,9,12-tetraoxatetradecyl, 4-hydroxy-2-methyl-3-azabutyl, 4-hydroxy-3-hydroxymethyl-2-methyl-3-azabutyl, 5-hydroxy-2-methyl-3-azapentyl, 5-hydroxy-3-(2-hydroxy-ethyl)-2-methyl-3-azapentyl, 8-hydroxy-2-methyl-3-aza-6-oxaoctyl, 11-hydroxy-2-methyl-3-aza-6,9-dioxaundecyl, 8-hydroxy-(5-hydroxy-3-oxapentyl)-2-methyl-3-aza-6-oxaoctyl or 11-hydroxy-3-(8-hydroxy-3,6-dioxaoctyl)-2-methyl-3-aza-6,9-dioxaundecyl, N,N-dipropylaminoethyl, N,N-dibutylaminoethyl,3-(N,N-dipropylamino)propyl or 3-(N,N-dibutylamino)propyl.
A is for example methylimino, ethylimino, propylimino, isopropylimino, butylimino, isobutylimino, sec-butylimino, tert-butylimino, pentylimino, isopentylimino, neopentylimino, tert-pentylimino, hexylimino or 2-methylpentylimino.
B1 and B2 are each for example methylene, ethylene, 1,2- or 1,3-propylene, 1,2-, 2,3- or 1,4-butylene, (CH2)2O(CH2)2, (CH2)3O(CH2)2, (CH2)3O(CH2)3, (CH2)2O(CH2)2O(CH2)2, (CH2)2NH(CH2)2, (CH2)3NH(CH2)2, (CH2)2NH(CH2)2NH(CH2)2,
An⊖, the equivalent of an anion, is derived for example from the following anions: fluoride, chloride, bromide, iodide, sulfate, phosphate, formate, acetate, propionate, mono-, di- or trichloroacetate, lactate, methoxyacetate, citrate, succinate, methanesulfonate, benzenesulfonate, 2- or 4-methylbenzenesulfonate or naphthalenesulfonate.
Suitable coupling components KH are compounds of the benzene, naphthalene, quinolene, pyridone, barbituric acid or pyrazolone series and correspond for example to the compounds of the formulae III a-f
where
R4 is hydroxyl, amino, morpholino, mono- or di(C1-C6)alkylamino whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C1-C6-alkoxycarbonyl, carbamoyl or mono or di(C1-C6)alkylcarbamoyl and are uninterrupted or interrupted by oxy or is C1-C6-alkoxy,
R5 is hydrogen, C1-C6-alkyl or R4,
R6 is hydrogen, C1-C6-alkyl, C1-C6-alkoxy with or without oxy interruption in its alkyl radical, mono or di(C1-C6)alkylamino, C1-C6-alkoxycarbonyl, carbamoyl, mono or di(C1-C6)alkylcarbamoyl whose alkyl radicals are unsubstituted or substituted by hydroxyl or amino and are uninterrupted or oxy interrupted, sulfamoyl, mono or di(C1-C6)alkylsulfamoyl whose alkyl radicals are unsubstituted or hydroxyl or amino substituted and uninterrupted or oxy interrupted, or is carboxyl,
R7 is hydroxyl, amino, mono or di(C1-C12)alkylamino, cyclohexylamino,
R8 is hydrogen, hydroxyl, amino, formylamino, acetylamino, C1-C6-alkyl,
R9 is hydrogen, C1-C6-alkyl, phenyl, hydroxyl, cyano, acetyl, benzoyl, methoxycarbonyl, carbamoyl,
R10 is hydrogen, chlorine, bromine, acetylamino, amino, nitro, sulfamoyl, methylsulfonyl, phenylsulfonyl, C1-C4-alkoxycarbonyl, C1-C4-alkanoyl, benzoyl, carbamoyl, cyano, N-methylimidazolyl, pyridinio,
R11 is hydrogen, C1-C6-alkyl with or without phenyl, hydroxyl, amino, C1-C6-alkoxy, acetylamino, benzoylamino or cyano substitution, cyclohexyl, phenyl with or without benzoylamino, acetylamino, methyl, methoxy, cyano or chlorine substitution, or amino which is substituted by phenyl, C1-C6-alkyl, C1-C6-alkanoyl or benzoyl,
R12 or R13 are independently hydrogen or C1-C6-alkyl,
R14 is methyl, C1-C4-alkoxycarbonyl or phenyl,
R15 is hydrogen, C1-C6-alkyl, cyclohexyl, benzyl or phenyl which is unsubstituted or from singly to triply substituted by fluorine, chlorine, bromine, C1-C4-alkyl, C1-C4-alkoxy, cyano, acetyl, acetylamino, hydroxyl, sulfamoyl or carbamoyl.
R4, R6 and R7 are each for example mono- or dimethylamino, mono- or diethylamino, mono- or dipropylamino, mono- or diisopropylamino, mono- or dibutylamino, mono- or dipentylamino, mono- or diisopentylamino, mono- or dineopentylamino, mono- or dihexylamino, mono- or di-2-methylpentylamino, N-methyl-n-butylamino, N-ethyl-n-butylamino.
R4 may also be for example mono- or di(2-hydroxyethyl)amino, mono-or di(2-hydroxypropyl)amino, mono- or di(3-hydroxypropyl)amino, N-methyl-2-hydroxyethylamino, N-methyl-2-hydroxypropylamino, N-methyl-3-hydroxypropylamino, N-ethyl-2-hydroxyethylamino, N-ethyl-2-hydroxypropylamino, N-ethyl-3-hydroxypropylamino, N-propyl-2-hydroxyethylamino, N-propyl-2-hydroxypropylamino, N-propyl-3-hydroxypropylamino, N-isopropyl-2-hydroxyethylamino, -2-hydroxypropylamino, -3-hydroxypropylamino, mono- or di(2-aminoethyl)amino, mono- or di(2-aminopropyl)amino, mono- or di(3-aminopropyl)amino, mono- or di(2-aminobutyl)amino, mono- or di(4-aminobutyl)amino, 2-cyanoethylamino, 2-cyano-2-methylethylamino, 2-methoxycarbonylethylamino, 2-methoxycarbonyl-2-methylethylamino, 2-ethoxycarbonylethylamino, 2-ethoxycarbonyl-2-methylethylamino, 2-propoxycarbonylethylamino, 2-propoxycarbonyl-2-methylethylamino, 2-isopropoxycarbonylethylamino, 2-isopropoxycarbonyl-2-methylethylamino, 2-butoxycarbonylethylamino, 2-butoxycarbonyl-2-methylethylamino, 2-carbamoylethylamino, 2-carbamoyl-2-methylethylamino, 2-mono or dimethylaminocarbonylethylamino, 2-mono or dimethylaminocarbonyl-2-methylethylamino, mono or di(2-methoxyethyl)amino, mono or di(2-methoxypropyl)amino, mono-or di(3-methoxypropyl)amino, or mono or di(2-hydroxyethyloxyethyl)amino.
R5, R6, R8, R9, R11, R12, R13 and R15 are each for example methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl, neopentyl, tert-pentyl, hexyl or 2-methylpentyl.
R11 is hydroxy-C1-C4-alkyl such as hydroxymethyl, 1-hydroxyeth-1-yl, 2-hydroxyeth-1-yl, 1-hydroxy-prop-1-yl, 2-hydroxyprop-1-yl, 3-hydroxyprop-1-yl, 1-hydroxy-prop-2-yl, 2-hydroxyprop-2-yl, 1-hydroxybut-1-yl, 2-hydroxybut-1-yl, 3-hydroxybut-1-yl, 4-hydroxybut-1-yl, 1-hydroxybut-2-yl, 2-hydroxybut-2-yl, 1-hydroxybut-3-yl, 2-hydroxybut-3-yl, 1-hydroxy-2-methyl-prop-3-yl, 2-hydroxy-2-methyl-prop-3-yl, 3-hydroxy-2-methyl-prop-3-yl, 2-hydroxymethyl-prop-2-yl, cyanomethyl, cyanoethyl, cyanopropyl or cyanobutyl.
R11 may also be for example benzyl, 1-phenylethyl, 2-phenylethyl, 1-phenylprop-1-yl, 2-phenylprop-1-yl, 3-phenylprop-1-yl, 1-phenylbut-1-yl, 2-phenylbut-1-yl, 3-phenylbut-1-yl, 4-phenylbut-1-yl, 1-phenylbut-2-yl, 2-phenylbut-2-yl, 3-phenylbut-2-yl, 4-phenylbut-2-yl, 1-(phenylmethyl)-eth-1-yl, 1-(phenylmethyl)-1-(methyl)-eth-1-yl, 1-(phenylmethyl)-prop-1-yl, preferably benzyl and 2-phenylethyl, methoxymethyl, ethoxymethyl, n-propoxymethyl, (1-methylethoxy)methyl, n-butoxymethyl, (1-methylpropoxy)methyl, (2-methylpropoxy)methyl, (1,1-dimethylethoxy)methyl, 2-(methoxy)ethyl, 2-(ethoxy)ethyl, 2-(n-propoxy)ethyl, 2-(1-methoxyethoxy)ethyl, 2-(n-butoxy)ethyl, 2-(1-methylpropoxy)ethyl, 2-(2-methylpropoxy)ethyl, 2-(1,1-dimethylethoxy)ethyl, 2-(methoxy)propyl, 2-(ethoxy)propyl, 2-(n-propoxy)propyl, 2-(1-methylethoxy)propyl, 2-(n-butoxy)propyl, 2-(1-methylpropoxy)propyl, 2-(2-methylpropoxy)propyl, 2-(1,1-dimethylethoxy)propyl, 3-(methoxy)propyl, 3-(ethoxy)propyl, 3-(n-propoxy)propyl, 3-(1-methylethoxy)propyl, 3-(n-butoxy)propyl, 3-(1-methylpropoxy)propyl, 3-(2-methylpropoxy)propyl, 3-(1,1-dimethylethoxy)propyl, 2-(methoxy)butyl, 2-(ethoxy)butyl, 2-(n-propoxy)butyl, 2-(1-methylethoxy)butyl, 2-(n-butoxy)butyl, 2-(1-methylpropoxy)butyl, 2-(2-methylpropoxy)butyl, 2-(1,1-dimethylethoxy)butyl, 3-(methoxy)butyl, 3-(ethoxy)butyl, 3-(n-propoxy)butyl, 3-(1-methylethoxy)butyl, 3-(n-butoxy)butyl, 3-(1-methylpropoxy)butyl, 3-(2-methylpropoxy)butyl, 3-(1,1-dimethylethoxy)butyl, 4-(methoxy)butyl, 4-(ethoxy)butyl, 4-(n-propoxy)butyl, 4-(1-methylethoxy)butyl, 4-(n-butoxy)butyl, 4-(1-methylpropoxy)butyl, 4-(2-methylpropoxy)butyl or 4-(1,1-dimethylethoxy)butyl.
R10 is for example formyl, acetyl, propionyl, butyryl or isobutyryl.
R6 is for example mono- or dimethylcarbamoyl, mono- or diethylcarbamoyl, mono- or dipropylcarbamoyl, mono- or dibutylcarbamoyl, mono- or dipentylcarbamoyl or mono- or dihexylcarbamoyl.
R4 and R6 may each be for example methoxy, ethoxy, propoxy, isopropoxy, butoxy, sec-butoxy, isobutoxy or tert-butoxy.
R6 may also be for example methoxyethyloxy, methoxypropyloxy, ethoxyethyloxy, ethoxypropyloxy or propoxypropyloxy.
R15 is for example 2-, 3- or 4-fluorophenyl, 2-, 3- or 4-chlorophenyl, 2-, 3- or 4-bromophenyl, 2-, 3- or 4-methylphenyl, 2-, 3- or 4-ethylphenyl, 2-, 3- or 4-methoxyphenyl, 2-, 3- or 4-cyanophenyl, 2-, 3- or 4-hydroxyphenyl, 2-, 3- or 4-acetylphenyl, 2-, 3- or 4-acetylaminophenyl, 2-, 3- or 4-sulfamoylphenyl or 2-, 3- or 4-carbamoylphenyl.
R6, R10 and R14 may each also be for example methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, sec-butoxycarbonyl, tert-butoxycarbonyl, pentoxycarbonyl, isopentoxycarbonyl, neopentoxycarbonyl, tert-pentyloxycarbonyl, hexyloxycarbonyl or 2-methylpentyloxycarbonyl.
R6 may also be for example mono- or dimethylsulfamoyl, mono- or diethylsulfamoyl, mono- or dipropylsulfamoyl, mono- or dibutylsulfamoyl.
R11may also be for example aminomethyl, 2-aminoethyl, 2- or 3-aminopropyl, 2- or 4-aminobutyl, acetylaminomethyl, 2-acetylaminoethyl, 2- or 3-acetylaminopropyl, 2- or 4-acetylaminobutyl, benzoylaminomethyl, 2-benzoylaminoethyl, 2- or 3-benzoylaminopropyl, 2- or 4-benzoylaminobutyl, 2-, 3- or 4-benzoylaminophenyl, 2-, 3- or 4-acetylaminophenyl, 2-, 3- or 4-methylphenyl, 2-, 3- or 4-methoxyphenyl, 2-, 3- or 4-cyanophenyl, 2-, 3- or 4-chlorophenyl, phenylamino, methylamino, ethylamino, propylamino, isopropylamino, butylamino, isobutylamino, sec-butylamino, tert-butylamino, acetylamino, propionylamino, butyrylamino, isobutyrylamino, isobutyrylamino, valerylamino, isovalerylamino, pivaloylamino, pentylcarbonylamino or benzoylamino.
In what follows, coupling components KH are recited by way of example. Examples of coupling components of the benzene series are resorcinol, 2- and 4-methylresorcinol, 1,3-phenylenediamine, 3-aminophenol, 4-methyl-3-aminophenol, 5-amino-2-methylphenol, 4-ethyl-3-aminophenol, 3-N,N-diethylaminophenol, 2,4-diaminotoluene, 2,4-dihydroxybenzoic acid, 4-methyl-2-aminophenol, 3-acetaminophenol, 3-amino-4-hydroxybenzenesulfonamide, o-, m- or p-toluidine, o-, m- or p-xylidine, 2,5-dimethoxyaniline, 2-methoxy-5-methylaniline, 3-amino-4-methylacetanilide, 2- or 4-methoxyacetanilide, N-methylaniline, N-methyl-m-toluidine, N-ethylaniline, N,N-diethylaniline, E-ethyl-m-toluidine, N- ( 2-hydroxyethyl) aniline, N,N-dihydroxyethylaniline or N-(2-hydroxyethyl)-m-toluidine.
Coupling components of the naphthalene series are for example 1-naphthylamine, N-phenyl-1-naphthylamine, N-ethyl-1-naphthylamine, N-ethyl-2-naphthylamine, N-phenyl-2-naphthylamine, 1-naphthol, 2-naphthol, 2,5-dihydroxynaphthalene, 1,6-dihydroxynaphthalene, 1,7-dihydroxynaphthalene, 2,7-dihydroxynaphthalene, 2-hydroxynaphthalene-1-carboxylic acid, 2-hydroxynaphthalene-3-carboxylic acid, methyl 2-hydroxynaphthalene-3-carboxylate or 2-hydroxynaphthalene-3-carboxamide.
Coupling components of the quinoline series are for example 8-hydroxyquinoline, 8-hydroxy-2-methylquinoline, 8-aminoquinoline, 8-amino-2-methylquinoline, 2-hydroxyquinoline and 2,4-dihydroxyquinoline.
Pyridone coupling components are for example 1-ethyl-2-hydroxy-4-methyl-5-carbamoylpyrid-6-one, 1-(2′-hydroxyethyl)2-hydroxy-4-methyl-5-carbamoylpyrid-6-one, 1-phenyl-2-hydroxy-4-methyl-5-carbamoylpyrid-6-one, 1-ethyl-2-hydroxy-4-methyl-5-cyanopyrid-6-one, 1-methyl-2-hydroxy-4-methyl-5-cyanopyrid-6-one, 1-methyl-2-hydroxy-5-acetylpyrid-6-one, 1,4-dimethyl-2-hydroxy-5-cyanopyrid-6-one, 1,4-dimethyl-5-carbamoylpyrid-6-one, 2,6-dihydroxy-4-ethyl-5-cyanopyridine, 2-hydroxy-4-ethyl-5-carbamoylpyrid-6-one or 1-methyl-2-hydroxy-4-methylsulfonylpyrid-6-one.
Examples of suitable coupling components of the barbituric acid series are barbituric acid, dimethylbarbituric acid and diethylbarbituric acid.
Suitable pyrazolone coupling components are for example 3-methyl-or 3-(C1-C4-alkoxycarbonyl)pyrazol-5-ones which, in 1-position, may bear hydrogen, unsubstituted or methyl-, ethyl-, fluorine-, chlorine-, bromine-, methoxy-, ethoxy-, cyano-, acetylamino-, hydroxyl-, carbamoyl- or sulfamoyl-substituted phenyl. Examples are 1-(2′,5′-dichlorophenyl)-3-methylpyrazol-5-one, ethyl 1-phenylpyrazol-5-one-3-carboxylate and ethyl pyrazol-5-one-3-carboxylate.
Preference is given to dyes of the general formula I where R1 and R2 are each hydrogen.
Preference is further given to dyes where A1 and A2 on the one hand and B1 and B2 on the other are each the same radical.
B1 and B2 are each preferably C2-C6-alkylene with or without interruption by oxy or by C1-C6-alkylimino. B1 and B2 are preferably identical.
Coupling components KH are preferably compounds of the formulae IIIa and IIIb. Preference is given in particular to dyes of the general formula IV
where n is from 1 to 8 and R1, R2, R3, R4, R5, R6, A1, A2, B1, B2 and An⊖are each as defined above. Particular preference is given to coupling components of the formula IIIa where R4 is hydroxyl, amino, mono- or dimethyl- or -ethylamino or mono- or dihydroxyethylamino and R5 and R6 are independently hydrogen, hydroxyl, amino or methyl.
Particular preference is likewise given to coupling components of the formula IIIb where R7 is hydroxyl or amino.
Particular preference is given to the following coupling components: resorcinol, 3-aminophenol, m-phenylenediamine, α- and β-naphthol, m-toluidine and 2,5-dimethylaniline.
Preference is further given to dyes of the formula I where n is 1.
where R1, R2, R3, A1, A2, B1 and B2 are each as defined above, can be conventionally diazotized and coupled with a coupling component KH. The pH is preferably within the range from 2 to 8.
Advantageously, the diazonium salt solution is metered into the solution of the coupling component. It is generally possible to use a single coupling component and also mixtures of a plurality of coupling components, for example so as to shade the hue.
By using coupling components capable of coupling twice it is possible to use the azo coupling reaction to obtain higher molecular weight dyes. These oligomeric dyes are obtainable by azo coupling of 1 mol equivalent of diamine of the formula IV onto from 1 to 2, preferably onto from 1 to 1.5, especially from 1 to 1.2, mol equivalents of a coupling component of the formula IIIa′
where
R4′ and R5′ are independently hydroxyl, amino, morpholino or mono-or di(C1-C6)alkylamino, whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C1-C6-alkoxycarbonyl, carbamoyl or mono- or di(C1-C6)alkylcarbamoyl and uninterrupted or interrupted by oxy, and
R6′ is hydrogen, C1-C6-alkoxy with or without oxy interruption in its alkyl radical, mono- or di(C1-C6)alkylamino, C1-C6-alkoxycarbonyl, carbamoyl, mono- or di(C1-C6)alkylcarbamoyl, sulfamoyl, mono- or di(C1-C6)alkylsulfamoyl.
The coupling step is carried out at a pH above 2, preferably within the range from 3 to 7. The desired pH may be set and, if necessary, corrected during the coupling reaction by adding, for example, ammonium or alkali metal bases, e.g., sodium hydroxide, sodium carbonate, sodium acetate, potassium hydroxide, potassium carbonate or potassium acetate. Usually, the end point of the reaction will be reached not more than 2 hours after completion of the addition. Chain termination is effected by coupling onto a coupling component devoid of a reactive terminus or by decomposing the diazonium compounds.
Depending on the choice of ratio for the starting materials, mixtures are obtained of various oligomers, among which the dimer fraction increases with increasing coupling component fraction. An approximately stoichiometric reaction of 1 mol of diamine V with 2 mol of coupling component IIIa′ provides predominantly the preferred dyes where n is 1. Separation into the individual dyes is possible in principle, but generally unnecessary, since the mixtures are found to have good application properties.
onto disazo dyes IV provides likewise preferred tetraazo dyes.
The amines VI are generally known and are described for example EP-A-0 159 549.
The dyes are very useful in the production of pulp-dyed, sized and unsized paper. They are likewise useful for dyeing paper by the dip process.
The dyeing of paper, leather or cellulose takes place according to methods known per se.
The novel dyes and their preparations color the papermaking wastewater only minimally, if at all, which is particularly favorable from the aspect of keeping the water courses clean. They are highly substantive, do not marble on paper and are substantially pH-insensitive. The dyeings on paper are notable for good lightfastness. On prolonged exposure to light the hue changes on-tone.
The basic azo dyes of the formula I of the invention can be used alone, in mixtures with each or one another and together with other cationic or anionic compounds in the form of their solutions or in the form of powders or granules. They are advantageously useful for dyeing or printing polymeric material, especially papery materials such as paper and paperboard, but also cellulose, cotton, leather, bast fibers, hemp, flax, sisal, jute, coir, straw or anionically modified fibers and also in inks, including printing inks.
In relation to the production of dye preparations comprising the novel dyes of the formula I, the use of polymers, such as polyacrylic acids, polyacrylic acid derivatives, polyvinylamines, polyvinylamides, polyvinyl acetates, polyvinyl alcohols, polyvinylpyrrolidones, polysiloxanes or copolymers of the respective monomers, must be emphasized. Similarly, oligomers of ethyleneimine, ethylene oxide or propylene oxide or derivatives thereof can be used.
The dyed papers, which exhibit good bleachability, are wetfast, not only with regard to water, but also with regard to milk, soapy water, sodium chloride solutions, fruit juices or sweetened mineral water, and are also stable to alcoholic drinks because of their good alcohol fastness.
The novel dyes can also be used for dyeing, padding or printing polyacrylonitrile textiles or anionically modified polyamide or polyester textiles.
The Examples which follow illustrate the invention.
a) 119 g of N-methyldiethanolamine were dissolved in 215 ml of acetone and added dropwise to a suspension of 326 g of isatoic acid anhydride in 1000 ml of acetone at 50° C. over 30 minutes. The mixture was then refluxed for 5 hours before the solvent was removed under reduced pressure to leave 364 g of a viscid oil (crude product) which was purified by recrystallizing it from ethanol.
Yield: 256 g of white crystals, melting point: 68-69° C.
b) 3.6 g of the diamine prepared under 1a) were introduced into a mixture of 50 ml of ice-water and 6 ml of concentrated hydrochloric acid. 6.6 ml of 23% strength by weight sodium nitrite solution were then added dropwise at 0-5° C. This was followed by stirring at 0-50° C. for one hour before excess nitrite was destroyed by the addition of sulfamic acid. The diazonium salt solution was added dropwise to a solution of 2.2 g of resorcinol in 200 ml of water whilst a pH of 2 was set with 10% strength by weight aqueous sodium hydroxide solution and the temperature was maintained at 0-5° C. by addition of ice. On completion of the addition the batch was stirred at room temperature for 3 hours and pH 2 and then adjusted to pH 7 with 10% strength by weight aqueous sodium hydroxide solution. The batch was then allowed to warm to room temperature overnight, and the reddish orange precipitate formed was filtered off with suction, washed with water and dried at 60° C. under reduced pressure to leave 5.7 g of the dye of the formula
λmax (dilute acetic acid): 408 nm
Example 1 was repeated with the coupling components recited in Table 1 to prepare the dyes indicated in the table.
| TABLE 1 | ||
| Ex. | Coupling component | λmax (nm) |
| 1.1 |
|
464 (NMP) |
| 1.2 |
|
476 (NMP) |
| 1.3 |
|
502 (dilute acetic acid) |
| 1.4 |
|
514 (dilute acetic acid) |
| 1.5 |
|
|
| 1.6 |
|
516 (dilute acetic acid) |
| 1.7 |
|
516 (dilute acetic acid) |
| 1.8 |
|
|
| 1.9 |
|
|
| 1.10 |
|
|
| 1.11 |
|
|
| 1.12 |
|
418 (dilute acetic acid) |
| 1.13 |
|
440 (dilute acetic acid) |
| 1.14 |
|
|
| 1.15 |
|
410 (dilute acetic acid) |
| 1.16 |
|
|
2.1 g of 2-dimethylaminoethyl 2-aminobenzoate were introduced into a mixture of 20 ml of ice-water and 3 ml of concentrated hydrochloric acid, before 3.3 ml of 23% strength by weight sodium nitrite solution were added dropwise at 0-5° C. The batch was subsequently stirred at 0-5° C. for one hour before excess nitrite was destroyed by addition of sulfamic acid. The diazonium salt solution was added to a solution of 3 g of the dye of Example 1b in 50 ml of ice-water and 20 ml of glacial acetic acid whilst the pH was maintained at 5-6 with 10% strength by weight aqueous sodium hydroxide solution. On completion of the addition the reaction mixture was allowed to warm up to room temperature overnight and then adjusted to pH 7. After stirring for one hour, the brown precipitate was filtered off with suction, washed with water and dried at 60° C. under reduced pressure to leave 5.0 g of the dye of the formula
λmax (dilute acetic acid): 410 nm
The method of Example 2 was also suitable for reacting the dyes of Examples 1.12 and 1.13 with diazotized 2-dimethylaminoethyl 2-aminobenzoate.
3.6 g of the diamine prepared under 1a) were introduced into a mixture of 50 ml of ice-water and 6 ml of concentrated hydrochloric acid before 6.6 ml of 23% strength sodium nitrite solution were added dropwise at 0-5° C. The batch was subsequently stirred at 0-50° C. for one hour before excess nitrite was destroyed by addition of sulfamic acid. The diazonium salt solution was then added dropwise to a solution of 1.1 g of resorcinol in 100 ml of water whilst a pH of 10 was set by simultaneous addition of 10% strength by weight aqueous sodium 40 hydroxide solution and the temperature was maintained at 0-5° C. by addition of ice. On completion of the addition the batch was stirred at 0-5° C. and pH 10 for 10 minutes before being adjusted to pH 7 with concentrated hydrochloric acid. The mixture was allowed to warm to room temperature overnight, and the resulting reddish orange precipitate was filtered off with suction, washed with water and dried at 60° C. under reduced pressure to leave 4.8 g of an oligomeric dye containing the repeat unit
λmax (dilute acetic acid): 412 nm
Claims (9)
where
R1 and R2 are independently hydrogen, nitro, halogen, C1-C6-alkyl,
R3 is hydrogen, C1-C6-alkyl with or without interruption by 1 or 2 nonadjacent oxy, imino, C1-C6-alkylimino groups or by a
radical,
A1 and A2 are independently oxy, imino or substituted or unsubstituted C1-C6-alkylimino,
B1 and B2 are independently C1-C6-alkylene with or without interruption by 1 or 2 nonadjacent oxy, imino, substituted or unsubstituted Cl1-6-alkylimino groups or by a
radical,
n is 1, and
where
R4′ and R5′ are independently hydroxyl, amino, morpholino or mono- or di(C1-C6)alkylamino, whose alkyl radicals are unsubstituted or substituted by hydroxyl, amino, cyano, C1-C6-alkoxycarbonyl, carbamoyl or mono- or di(C1-C6)alkylcarbamoyl and uninterrupted or interrupted by oxy,
Q1, Q2 and Q3 are independently hydrogen, C1-C10-alkyl with or without interruption by from 1 to 4 nonadjacent oxy, imino and substituted or unsubstituted C1-C4-alkylimino groups and with or without hydroxyl, halogen or phenyl substitution, or Q1 and Q2 combine with the linking nitrogen atom to form a 5- or 6-membered heterocyclic radical which optionally contains nitrogen or oxygen as further hetero atom, and
An⊖ is the equivalent of an anion,
and additionally n is from 2 to 8 when K is the radical of a coupling component of the formula IIIa′
where
R6′ is hydrogen, C1-C6-alkoxy with or without oxy interruption in its alkyl radical, mono- or di(C1-C6)alkylamino, C1-C6-alkoxycarbonyl, carbamoyl, mono- or di(C1-C6))alkylcarbamoyl, sulfamoyl, mono- or di(C1-C6)alkylsulfamoyl.
2. The cationic dye as claimed in claim 1 , wherein n is 1.
4. The cationic dye as claimed in claim 1 , wherein each of A1 and A2; and each of B1 and B2 are each the same radical.
5. The cationic dye as claimed in claim 1 , obtainable by azo coupling of 1 mol equivalent of diamine of the formula V
where R1, R2, R3, A1, A2, B1 and B2 are each as defined in claim 1 , with from 1 to 2 mol equivalents of a coupling component of the formula IIIa′
where R4′, R5′ and R6′ are each as defined in claim 1 .
6. The cationic dye as claimed in claim 1 , obtainable by azo coupling of 1 mol equivalent of diamine V with from 1.5 to 2 mol equivalents of a coupling component IIIa′.
7. A process for preparing the cationic dye as claimed in claim 1 , comprising:
diazotizing one or more diamines V and coupling the diazonium salt onto one or more coupling components IIIa′.
8. A dye mixture, comprising:
a plurality of dyes of the formula I according to claim 1 which differ in the number of their repeat units.
9. A method of dyeing or printing a polymeric material comprising:
applying a cationic azo dye as claimed in claim 1 to said polymer material.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19851026 | 1998-11-05 | ||
| DE19851026A DE19851026A1 (en) | 1998-11-05 | 1998-11-05 | Cationic azo dyes |
| PCT/EP1999/008274 WO2000027929A1 (en) | 1998-11-05 | 1999-10-30 | Cationic azo-dyes |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US6346609B1 true US6346609B1 (en) | 2002-02-12 |
Family
ID=7886810
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/807,999 Expired - Fee Related US6346609B1 (en) | 1998-11-05 | 1999-10-30 | Cationic azo-dyes |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US6346609B1 (en) |
| EP (1) | EP1124903B1 (en) |
| DE (2) | DE19851026A1 (en) |
| WO (1) | WO2000027929A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040083924A1 (en) * | 2001-02-09 | 2004-05-06 | Martin Oberholzer | Azo dyestuffs |
| US20100068472A1 (en) * | 2005-10-22 | 2010-03-18 | Gavin Wright | Yellow Azo Dyes for Ink Jet Printing |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0521551D0 (en) * | 2005-10-22 | 2005-11-30 | Avecia Inkjet Ltd | Yellow azo dyes for ink jet printing |
| CN106700647B (en) * | 2016-12-06 | 2021-02-02 | 浙江龙盛化工研究有限公司 | Orange-red disperse dye composition and dye product |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2160590A1 (en) * | 1971-12-07 | 1973-06-14 | Bayer Ag | NEW AROMATIC POLYAMINES, A PROCESS FOR THEIR PRODUCTION AND THEIR USE FOR THE PRODUCTION OF POLYURETHANES |
| DE2218645A1 (en) * | 1972-04-18 | 1973-10-25 | Bayer Ag | Polyazo dyes - as pigments and as dispn dyes for synth fibres |
| US3957752A (en) * | 1972-11-03 | 1976-05-18 | American Aniline Products, Inc. | Quaternized azoanilino toluene sulfonamidoalkylene-amine dyes |
| EP0162409A2 (en) * | 1984-05-19 | 1985-11-27 | BASF Aktiengesellschaft | Basic azo dyes |
-
1998
- 1998-11-05 DE DE19851026A patent/DE19851026A1/en not_active Withdrawn
-
1999
- 1999-10-30 EP EP99957270A patent/EP1124903B1/en not_active Expired - Lifetime
- 1999-10-30 WO PCT/EP1999/008274 patent/WO2000027929A1/en not_active Ceased
- 1999-10-30 DE DE59906128T patent/DE59906128D1/en not_active Expired - Fee Related
- 1999-10-30 US US09/807,999 patent/US6346609B1/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2160590A1 (en) * | 1971-12-07 | 1973-06-14 | Bayer Ag | NEW AROMATIC POLYAMINES, A PROCESS FOR THEIR PRODUCTION AND THEIR USE FOR THE PRODUCTION OF POLYURETHANES |
| DE2218645A1 (en) * | 1972-04-18 | 1973-10-25 | Bayer Ag | Polyazo dyes - as pigments and as dispn dyes for synth fibres |
| US3957752A (en) * | 1972-11-03 | 1976-05-18 | American Aniline Products, Inc. | Quaternized azoanilino toluene sulfonamidoalkylene-amine dyes |
| EP0162409A2 (en) * | 1984-05-19 | 1985-11-27 | BASF Aktiengesellschaft | Basic azo dyes |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040083924A1 (en) * | 2001-02-09 | 2004-05-06 | Martin Oberholzer | Azo dyestuffs |
| US7371831B2 (en) * | 2001-02-09 | 2008-05-13 | Clariant Finance (Bvi) Limited | Azo dyestuffs |
| US20100068472A1 (en) * | 2005-10-22 | 2010-03-18 | Gavin Wright | Yellow Azo Dyes for Ink Jet Printing |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1124903B1 (en) | 2003-06-25 |
| DE19851026A1 (en) | 2000-05-11 |
| WO2000027929A1 (en) | 2000-05-18 |
| DE59906128D1 (en) | 2003-07-31 |
| EP1124903A1 (en) | 2001-08-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US4087244A (en) | Dyeing and printing basic dyeable textile substrates with becationic disazo and trisazo dyes having optionally further substituted 6-hydroxy-4-methyl-3-N-pyridiniumpyridone-2-coupling component radicals | |
| US4367172A (en) | Disazo dyes containing at least one sulfo group and at least two basic or cationic groups | |
| US6048968A (en) | Cationic azo dyes based on aminobenzoic acid | |
| US4962190A (en) | Azo dyes containing polyethyleneimine with an aniline diazo component and a hydroxynaphthalene, aminopyridine or acetoacetic coupling component | |
| US6346609B1 (en) | Cationic azo-dyes | |
| EP0092520B1 (en) | Basic compounds, their preparation and their use | |
| US5750661A (en) | Basic dyes based on amides of J-acid (1-hydroxy-6-aminonaphthalene-3-sulfonic acid) and amides of J-acid | |
| US5023324A (en) | Biscationic disazo and trisazo compounds having optionally further substituted 6-hydroxy-4-methy-3-N-pyridiniumpyridone-2 coupling component radicals, dyes compositions comprising them and their use | |
| JPS6143388B2 (en) | ||
| EP0482508B1 (en) | Biscationic azo dyes and their intermediates | |
| DE2006131C3 (en) | Mono- and disazo dyes, processes for their production and their use for dyeing and printing acrylic, nylon or polypropylene fibers, as well as fibers containing ester groups | |
| US4670546A (en) | Disazo and polyazo compounds containing at least two cationic groups in metal-free or 1:1 or 1:2 metal complex form | |
| EP0586331A2 (en) | Process for dyeing synthetic polyamid fibre materials | |
| US4873319A (en) | Disazo dyes containing a pyrimidine coupling component and a cationic alkyl ammonium group | |
| US5559217A (en) | Disazo dyes and a process of dyeing paper therewith | |
| EP0051559A1 (en) | Basic and/or cationic azo compounds free from sulfonic-acid groups | |
| CN1318515C (en) | Bisazo dyestuffs based on hydroxynaphthalene carboxylic acid | |
| US5077396A (en) | Azo compounds containing two 6-hydroxypyrid-2-one coupling component radicals the nitrogen atoms of which are linked by a biscationic radical | |
| US6084078A (en) | Disazo dyestuffs for dyeing cellulosic materials | |
| CH663614A5 (en) | HETEROCYCLIC COMPOUNDS CONTAINING BASIC AND / OR CATIONIC GROUPS. | |
| US4048154A (en) | Monoazo dyestuffs derived from p-sulfo-halophenyl diazo moiety and p-amino-O-alkyl coupler moiety | |
| US4217272A (en) | Novel monoazo and disazo colorants from aminoalkylanilines and bis(aminoalkyl)anilines | |
| EP0594037B1 (en) | Basic dyes based on amides of I-acid (1-hydroxy-6-aminonaphthalene-3-sulfonic acid) and amides of I-acid | |
| CA1064914A (en) | Monoazo and disazo colorants | |
| US4153598A (en) | Monoazo and diazo colorants from aminoalkylanilines and bis(aminoalkyl)anilines |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: BASF AKTIENGESELLSCHAFT, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ETZBACH, KARL-HEINZ;FREUND, TORSTEN;TRESCH, RAINER;REEL/FRAME:012387/0090 Effective date: 19991118 |
|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| REMI | Maintenance fee reminder mailed | ||
| LAPS | Lapse for failure to pay maintenance fees | ||
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20100212 |