US3924628A - Cyrogenic bladder for necrosing tissue cells - Google Patents
Cyrogenic bladder for necrosing tissue cells Download PDFInfo
- Publication number
- US3924628A US3924628A US311069A US31106972A US3924628A US 3924628 A US3924628 A US 3924628A US 311069 A US311069 A US 311069A US 31106972 A US31106972 A US 31106972A US 3924628 A US3924628 A US 3924628A
- Authority
- US
- United States
- Prior art keywords
- bladder
- pressure
- probe
- uterus
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F7/00—Heating or cooling appliances for medical or therapeutic treatment of the human body
- A61F7/12—Devices for heating or cooling internal body cavities
- A61F7/123—Devices for heating or cooling internal body cavities using a flexible balloon containing the thermal element
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/42—Gynaecological or obstetrical instruments or methods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/02—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by cooling, e.g. cryogenic techniques
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B17/00—Surgical instruments, devices or methods
- A61B17/42—Gynaecological or obstetrical instruments or methods
- A61B2017/4216—Operations on uterus, e.g. endometrium
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00053—Mechanical features of the instrument of device
- A61B2018/00214—Expandable means emitting energy, e.g. by elements carried thereon
- A61B2018/0022—Balloons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B90/00—Instruments, implements or accessories specially adapted for surgery or diagnosis and not covered by any of the groups A61B1/00 - A61B50/00, e.g. for luxation treatment or for protecting wound edges
- A61B90/03—Automatic limiting or abutting means, e.g. for safety
- A61B2090/033—Abutting means, stops, e.g. abutting on tissue or skin
- A61B2090/036—Abutting means, stops, e.g. abutting on tissue or skin abutting on tissue or skin
Definitions
- Necrosis means the death of the cells in a tissue.
- Functional lining of the uterus is that portion of the inner lining of the uterus; the endometrium, to which an embryo might attach. It excludes the portions of the uterine inner lining forming the cervix, to which the embryo usually does not attach.
- Cryogenic as an adjective is used to refer to temperatures sufficiently low to cause cell necrosis.
- cryosurgery The use of instruments at extremely low or cryogenic temperatures to treat the human body is referred to as cryosurgery within the medical profession.
- an instrument cooled to a very low temperature is brought into contact with body tissue and the contiguous or adjacent cells are destroyed by the cryogenic temperature, a result known also as coagulation necrosis.
- the destruction of a cell is usually effected by the formation of ice within a cell, which is sufficient to kill or irreversibly damage it. It can normally withstand ice formation outside the cell wall; to destroy the cell, ice must be formed inside the cell membrane.
- a human cell contains both salt, most abundantly sodium chloride, and water.
- a cell protective mechanism operates to resist internal freezing of the cell. Water is expelled from within the cell through the cell membrane. The expelled water may freeze without permanent damage to the cell, but this protective mechanism results in increasing the saline concentration within the cell, lowering its freezing point and rendering it more difficult to destroy with low temperatures. This process continues, with external ice formation, until the eutectic temperature of the sodium chloride, -2l.2C, is reached. At this temperature ice is formed within the cell and it is effectively destroyed.
- cryogenic destruction of a cell, or of the tissue which is formed of the cells requires either (a) bringing the interior of the cell to a point below the eutectic temperature of sodium chloride or (b) reducing the cell temperature very quickly so that the cell protective mechanism cannot cope with the temperature change and the cell does not have time to expel sufficient water through the membrane to render its interior solution freeze resistant.
- the variables are the absolute temperature to which a cell is brought, the rapidity of cell temperature change, and also, not discussed above and imperfectly characterized as yet, the length of time a cell is kept at a specific temperature.
- Cryosurgical techniques utilizing cryogenic necrosis, have been used in gynecology for the treatment of chronic cervicitis and dysfunctional uterine bleeding by applying an instrument at cryogenic temperature to the affected area.
- the inner lining of the uterus comprises a layer of endometrium that varies in thickness from 2 to 8 millimeters, and outward from the endometrium is a layer of myometrium, or muscle tissue.
- the embryo attaches itself to the inner wall or surface of the uterus at some point on the functional lining and is supported and sustained by the endometrium.
- cryogenic necrosis By substantially destroying the entire endometrial layer throughout the extent of the functional lining of the uterus by cryogenic necrosis, that layer is not capable of sustainingan embryo.
- the method used to date, however, and described in that article, comprised use of a metal probe having a rather small operative area to successively freeze relatively small areas of the endometrium, eventually covering the entire functional lining. That process was relatively slow and laborious and considerable painstaking effort was required to insure that the entire functional lining was necrosed.
- the method and apparatus of this invention overcome these disadvantages by providing for cryogenic necrosis of the entire functional uterine endometrium in one application and with complete coverage.
- a flexible bladder is inserted into the uterus and cryogenic fluid at slightly above atmospheric pressure is circulated through the bladder, expanding it into contact with the entire inner uterine surface and at the same time removing heat from that surface so as to effect coagulation necrosis of the endometrium.
- FIG. 1 shows in a partially schematic manner a sterilization system suitable for use with liquid nitrogen as a refrigerant
- FIG. la is a partial sectional view taken approximately along line 1a la of FIG. 1;
- FIG. 2 shows an alternative method for pressurizing the liquid nitrogen system of FIG. 1;
- FIG. 3 shows schematically a sterilization system suitable for use with refrigerants that are stored at high pressure
- FIG. 4 shows an alternate arrangement for storing and supplying the high pressure refrigerant in the system of FIG. 3;
- FIGS. 5a and 5 b show alternative probe expansion orifices suitable for use with the high pressure refrigerant system of FIGS. 3 and 4;
- FIGS. through 6e show the insertion sequence of one possible probe package.
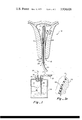
- FIG. 1 shows a uterus 10 distended by an inflated bladder 11 attached to a probe generally indicated as 12.
- the system uses as a refrigerant liquid nitrogen 13 contained in a Dewar vessel 14 that is closed with a plug 15.
- the liquid nitrogen and the vaporized gaseous nitrogen that fills space 16 above the liquid in Dewar vessel 14 would normally increase in pressure due to heat conduction through the Dewar vessel walls caus ing liquid nitrogen to vaporize.
- a vent valve 19 connected to pipe 18 is made such that when pressure in the Dewar vessel exceeds a specified value the valve opens to vent the excessive pressure. In this manner the pressure within the Dewar vessel 14 is controlled to be less than or equal to any preset pressure.
- vent valve 19 As long as liquid nitrogen exists in the vessel, heat conduction into the liquid nitrogen will cause the vent valve 19 to slowly vent vaporized nitrogen to the atmosphere, maintaining pressure in the Dewar at the specified operating pressure. This pressure is used to force the refrigerant into probe 12. Adequate pressure is maintained in the Dewar throughout the expulsion of all of the refrigerant by causing vapor space 16 to be approximately one-half of the total Dewar volume. As refrigerant is forced from the Dewar, vapor in space 16 expands to a lower pressure until it reaches approximately one-half the original pressure that existed at the initiation of refrigerant flow. Vent valve 19 also provides conventional protection by venting any excessive pressures that should build up through some malfunctron.
- the passage of return tube 25 surrounds supply tube 22 and bladder 11 is attached to the outside surface of tube 25 by a suitable low temperature cement.
- the end of supply tube 22 is open to the interior of bladder I l by virtue of orifice 23, and that end of return tube 25 is closed by being joined to the periphery of the end of tube 22.
- the size of orifice 23 may be quite large, in relation to the diameter of tube 22, since only low pressure gas is being fed through it and it does not act as an expansion orifice.
- Ports 24, comprising the only openings into return tube 25 or the inside of bladder 11, are located well back from the end of the tubes in order to force the nitrogen expelled from orifice 23 to circulate and absorb heat from bladder 11 before being vented to the atmosphere through return tube 25.
- the probe 12 and its attached bladder 11 must be sufficiently small, when the bladder is deflated and furled around tube 25, so that it can be conveniently inserted into the uterus through a partially dilated cervix.
- the bladder should be inflated to a pressure sufficient to insure firm contact with the tissue to be necrosed, for example, the interior uterine surface, but should preferably be maintained at about 2 or 3 p.s.i. to avoid any possible risk of internal injury to the patient. It has been found that maintaining a pressure in the Dewar of between -10 p.s.i. results in adequate flow of the nitrogen and the maintenance of proper bladder pressure, although the precise to be used in any case will, of course, need to be adjusted as a function of the various system parameters.
- the liquid nitrogen vaporizes in the course of its flow from the Dewar 14 to its exit into bladder 11 at supply tube orifice 23.
- the system may alternatively be operated by permitting the nitrogen to emerge from orifice 23 as a liquid, and allowing it to vaporize in bladder 11.
- Bladder 11 must be capable of withstanding cryogenic temperatures without rupturing, and should have as good heat transfer characteristics as obtainable in such materials to provide efficient freezing action.
- a bladder molded of a heat curing rubber bearing General Electric identification SE-5553 has been found satisfactory.
- FIG. 2 shows alternative methods of providing the required pressurization of theDewar in the system of FIG. 1 in which liquid nitrogen is the refrigerant.
- one alternate method that may be used to pressurize the vessel 14 is an external source of pressure 52 which is connected with vertical pipe 18 through pipe 51. Pipe 18 feeds down through plug 15 and pressurizes the Dewar l4. Vent valve 19 provides conventional protection by venting the Dewar 14 to the atmosphere should excessive pressure build-up through some malfunction.
- a second alternative method of providing the required pressurization of the Dewar 14 is also shown in FIG. 2.
- the vessel may be pressurized by energizing a heater coil 28 that is submerged in the liquid nitrogen 13 by means of a source of power 29 via wires 30.
- FIG. 3 is a schematic diagram of another system embodiment in which slightly different type of refrigerant is used, that is, a high pressure refrigerant, rather than the liquid nitrogen of the FIG. 1 embodiment which is maintained at atmospheric pressure in a Dewar vessel as described above.
- the high pressure refrigerants are stored at high pressure in liquid form in non-vented bottles of high strength. The liquid boils away until the vapor pressure in the bottle is equal to the saturation pressure; the refrigerant then is stored as a liquid at room temperature under its own vaporpressure, so that no Dewar vessel is necessary.
- Freon 13 and Freon 23, supplied by Dupont, and nitrous oxide, are refrigerants useful for this system in the embodiment shown in FIG. 3.
- the Freons have a vapor pressure in the neighborhood of 500 p.s.i. and nitrous 0xide, in the neighborhood of 700 p.s.i. at room temperature.
- Liquid nitrogen is not suitable for storage at room temperature because its vapor pressure is so high that providing storage containers of sufficient strength is not feasible.
- FIG. 3 a bottle 32 is shown containing both a liquid refrigerant 33 such as Freon or nitrous oxideand gas 34.
- a liquid refrigerant 33 such as Freon or nitrous oxideand gas 34.
- outlet valve 35 is opened and the high pressure gas is forced through line 36,
- Probe 39 which is merely indicated schematically in FIG. 3, is similar to the probe described in connection with the embodiment of FIG. 1 including the bladder, except that it is provided with expansion orifices, as shown in FIGS. a and 5b, rather than with the rather large supply tube orifice 23 shown in FIG. 1. This is necessary because the nitrogen of the FIG.
- FIG. 5a shows a small single expansion orifice 41 located in the end of supply tube 22 and suitable for use in the probe 39 of the high pressure embodiment of FIG. 3.
- multiple expansion orifices 42 may alternatively be used.
- an orifice 23 of 0.070 inch has been found suitable for the liquid nitrogen system of FIG. 1.
- the single expansion orifice (41 of FIG. 5a) of 0.0135 inch has been used for both nitrous oxide and Freon 13 and Freon 23.
- Two 0.0135 inch expansion orifices (42 of FIG. 5b) or, alternatively, a single orifice (41 of FIG. 6a) of 0.024 inch have been used with both Freons.
- the three-way valve 37 is used in connection with the warming of the bladder 11 after the cryogenic necrosis has been effected and prior to removal of the instrument from the uterus.
- I-Iuman tissue is in large part water and when it is in contact with any object at a temperature below the freezing point of water it sticks to the object by freezing to it, and will tend to tear if the object is pulled away.
- bladder l l which is configured to contact substantially the entire area of tissue to be necrosed, must be warmed before it can be removed, so that the contacted tissue is not torn.
- the warming is accomplished by stopping the flow or circulation of cryogenic nitrogen into probe 12 and permitting the heat of the patients body to bring the bladder temperature up.
- the action of body heat in warming bladder 11 is assisted by circulating room temperature gas through the bladder. This is accomplished by moving the threeway valve 37.
- the valve as shown schematically in FIG. 3, is in a position to pass the refrigerant gas from line 36 directly to line 38.
- the gas from line 36 will be diverted by valve 37 down into a regulator 43, which may be a Norgren Company Model 11-Ol0-084.
- Regulator 43 reduces the pressure of the gas to about 50 p.s.i.
- FIG. 4 illustrates an alternative refrigerant feed arrangement for the high pressure embodiment of FIG. 3 wherein some additional cooling capacity is obtained.
- a high pressure storage bottle 44 is shown in an inverted position, so that its outlet, and the associated outlet valve 45, are located at the bottom of the bottle.
- liquid refrigerant, rather than gas is fed from the bottle. This change of state from liquid to gas provides some additional cooling to that obtained by the gaseous expansion during emission from the probe expansion orifices.
- the bladder material SE-5553 supplied by General Electric and found satisfactory for the embodiment of FIG. 1 is also satisfactory for the FIG. 3 embodiment, where the refrigerant temperaturees are not nearly as low: nitrogen boils at 320.4F; nitrous oxide at -l29.1F; Freon 13 at l14.6F and Freon 23 at -1 15.7F.
- a dispersion coating rubber supplied by Dow Corning to their number 92-009 has been found satisfactory for use with the high pressure (and higher temperature) refrigerants of the FIG. 3 embodiment.
- Bladders of approximately 0.020 inch thickness have been found satisfactory.
- Various methodsof fabricating the bladder may be employed, but one method found satisfactory has been to lay up the bladder in successive thin layers upon a mandrel.
- thermocouples 50 FIG. 1a
- the thermocouples 50 and their leads may be placed between successive layers as the bladder is formed.
- Such thermocouples are connected to appropriate instrumentation for monitoring bladder temperature.
- the embodiments of FIGS. 1 and 3 each have advantages and disadvantages so that neither can clearly be preferred to the other.
- the liquid nitrogen refrigerant of the FIG. 1 embodiment provides a much lower temperature and consequently faster tissue freezing. It, however, has the disadvantages of imposing more constraints upon system components, especially the bladder, because of the lower temperature, and it requires the inconvenience of low temperature refrigerant storage.
- the high pressure refrigerants, on the other hand, useful in the FIG. 3 embodiment are conveniently stored at room temperature, are generally more readily available than liquid nitrogen, but compare unfavorably with liquid nitrogen in having higher temperatures and consequently slower freezing.
- the procedure is as follows.
- the cervix is visualized and then grasped with a tenaculum.
- the cervix is dilated with instruments to approximately one centimeter.
- probe 12 with bladder 11 furled tightly around it, is inserted through the cervix into the uterus.
- the refrigerant is applied to probe 12 for a sufficient period to accomplish cryogenic necrosis of the functional endometrium.
- the bladder 11 is then warmed and removed.
- FIGS. 6a through 6e shows in sequential steps the ap' plication of one configuration in which either of the embodiments of FIG. 1 or FIG. 3 may be packaged: a tampon-type cartridge.
- FIG. 6a shows the package, comprising a handle" portion, which is comprised of supply tube 22 and return tube 25, the bladder 11 furled tightly around the end of return tube 25, and encased in a cylindrical sleeve 48 terminating in an annular shoulder 49.
- the outside diameter of the sleeve should be less than one centimeter so that it may be readily inserted into the partially dilated cervical openmg.
- FIG. 6b shows the sleeve 48 containing the furled probe being inserted into the opening in the cervix 55.
- sleeve 48 has been fully inserted into the cervix 55, being stopped by the abutment of the sleeve shoulder 49 against the outer cervical surface.
- the probe has been advanced so that the end of the probe with the furled bladder 11 has been pushed free of sleeve 48 and is positioned inside the uterus.
- bladder 11 inflates and assumes the position inside the uterus shown by FIG. 6d, showing a view along a vertical plane through the body, and FIG. 6e, showing a view along a horizontal plane through the body.
- the primary feature of the package of FIG. 6 is the use of the flanged sleeve 48.
- This sleeve which is preferably about 2.5 centimeters in length, is used to protect the bladder from mechanical damage during storage and handling prior to use and also as an insertion aid. Since the inner uterine surface within about 2.5 centimeters of the cervix opening is not part of the functional uterine lining and should not be necrosed, the insulating effect of the sleeve will protect this portion of the uterus and prevent an unnecessary loss of cooling capacity.
- a method of effecting coagulation necrosis of the functional uterine endometrium comprising the steps of:
Landscapes
- Health & Medical Sciences (AREA)
- Surgery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Molecular Biology (AREA)
- Medical Informatics (AREA)
- Pregnancy & Childbirth (AREA)
- Reproductive Health (AREA)
- Gynecology & Obstetrics (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Vascular Medicine (AREA)
- Otolaryngology (AREA)
- Surgical Instruments (AREA)
Abstract
A method of, and apparatus for necrosing human tissue cells are described in which an expandable bladder filled with a fluid at a cyrogenic temperature is used to subject the entire area of tissue in contact with the bladder to temperatures sufficiently low to cause coagulation necrosis, whereby the entire contacted area of tissue is simultaneously necrosed. As an example, this apparatus can be utilized to cause sterilization by necrosis of the entire functional endometrial layer of the uterus of a human female.
Description
Sheet 1 of3 U.S. Patent Dec. 9, 1975 U.S. Patent Dec. 9, 1975 Sheet 2 of3 3,924,628
Fig 5b US. Patent Dec 9, 1975 Sheet 3 of3 3,924,628
CYROGENIC BLADDER FOR NECROSING TISSUE CELLS DEFINITIONS The following terms are used herein as having the meanings given below:
Necrosis" means the death of the cells in a tissue.
Functional lining of the uterus is that portion of the inner lining of the uterus; the endometrium, to which an embryo might attach. It excludes the portions of the uterine inner lining forming the cervix, to which the embryo usually does not attach.
Cryogenic as an adjective is used to refer to temperatures sufficiently low to cause cell necrosis.
BACKGROUND OF THE INVENTION The use of instruments at extremely low or cryogenic temperatures to treat the human body is referred to as cryosurgery within the medical profession. In cryosurgery, an instrument cooled to a very low temperature is brought into contact with body tissue and the contiguous or adjacent cells are destroyed by the cryogenic temperature, a result known also as coagulation necrosis. The destruction of a cell is usually effected by the formation of ice within a cell, which is sufficient to kill or irreversibly damage it. It can normally withstand ice formation outside the cell wall; to destroy the cell, ice must be formed inside the cell membrane.
A human cell contains both salt, most abundantly sodium chloride, and water. As the external environment of the cell is cooled, a cell protective mechanism operates to resist internal freezing of the cell. Water is expelled from within the cell through the cell membrane. The expelled water may freeze without permanent damage to the cell, but this protective mechanism results in increasing the saline concentration within the cell, lowering its freezing point and rendering it more difficult to destroy with low temperatures. This process continues, with external ice formation, until the eutectic temperature of the sodium chloride, -2l.2C, is reached. At this temperature ice is formed within the cell and it is effectively destroyed.
Therefore, in light of the above, cryogenic destruction of a cell, or of the tissue which is formed of the cells, requires either (a) bringing the interior of the cell to a point below the eutectic temperature of sodium chloride or (b) reducing the cell temperature very quickly so that the cell protective mechanism cannot cope with the temperature change and the cell does not have time to expel sufficient water through the membrane to render its interior solution freeze resistant. In cryogenic necrosis then, the variables are the absolute temperature to which a cell is brought, the rapidity of cell temperature change, and also, not discussed above and imperfectly characterized as yet, the length of time a cell is kept at a specific temperature.
Cryosurgical techniques, utilizing cryogenic necrosis, have been used in gynecology for the treatment of chronic cervicitis and dysfunctional uterine bleeding by applying an instrument at cryogenic temperature to the affected area.
Present procedures for permanent sterilization of the human female involve major surgery to enter the abdominal cavity. The attendant disadvantages are obvi ous and are those of most surgical procedures: the inconvenience and cost of a hospital stay; and the high cost of services of a surgeon and "other professionals.
It is the object of this invention to make available less expensive sterilization of human females by providing a sterilization procedure that may be performed more quickly than present surgical sterilization techniques and will not require the attendance of an entire surgical team or their support facilities.
The inner lining of the uterus comprises a layer of endometrium that varies in thickness from 2 to 8 millimeters, and outward from the endometrium is a layer of myometrium, or muscle tissue. After conception, the embryo attaches itself to the inner wall or surface of the uterus at some point on the functional lining and is supported and sustained by the endometrium. By substantially destroying the entire endometrial layer throughout the extent of the functional lining of the uterus by cryogenic necrosis, that layer is not capable of sustainingan embryo. An article, entitled Destruction of the Endometrium by Cryosurgery, by W. Droegemueller, E. Makowski and R. Macsalka, appearing in the American Journal of Obstetrics and Gynecology, Vol. 110, No. 4, June 15, 1971, describes cryogenic necrosis of the uterine endometrium.
The method used to date, however, and described in that article, comprised use of a metal probe having a rather small operative area to successively freeze relatively small areas of the endometrium, eventually covering the entire functional lining. That process was relatively slow and laborious and considerable painstaking effort was required to insure that the entire functional lining was necrosed. The method and apparatus of this invention overcome these disadvantages by providing for cryogenic necrosis of the entire functional uterine endometrium in one application and with complete coverage. A flexible bladder is inserted into the uterus and cryogenic fluid at slightly above atmospheric pressure is circulated through the bladder, expanding it into contact with the entire inner uterine surface and at the same time removing heat from that surface so as to effect coagulation necrosis of the endometrium.
The nature of the inventive method and apparatus is set forth in greater detail in the description below taken in conjunction with the drawings, which form a part of the specification, and in which:
FIG. 1 shows in a partially schematic manner a sterilization system suitable for use with liquid nitrogen as a refrigerant;
FIG. la is a partial sectional view taken approximately along line 1a la of FIG. 1;
FIG. 2 shows an alternative method for pressurizing the liquid nitrogen system of FIG. 1;
FIG. 3 shows schematically a sterilization system suitable for use with refrigerants that are stored at high pressure;
FIG. 4 shows an alternate arrangement for storing and supplying the high pressure refrigerant in the system of FIG. 3;
FIGS. 5a and 5 b show alternative probe expansion orifices suitable for use with the high pressure refrigerant system of FIGS. 3 and 4;
FIGS. through 6e show the insertion sequence of one possible probe package.
FIG. 1 shows a uterus 10 distended by an inflated bladder 11 attached to a probe generally indicated as 12. The system uses as a refrigerant liquid nitrogen 13 contained in a Dewar vessel 14 that is closed with a plug 15. The liquid nitrogen and the vaporized gaseous nitrogen that fills space 16 above the liquid in Dewar vessel 14 would normally increase in pressure due to heat conduction through the Dewar vessel walls caus ing liquid nitrogen to vaporize. A vent valve 19 connected to pipe 18 is made such that when pressure in the Dewar vessel exceeds a specified value the valve opens to vent the excessive pressure. In this manner the pressure within the Dewar vessel 14 is controlled to be less than or equal to any preset pressure. As long as liquid nitrogen exists in the vessel, heat conduction into the liquid nitrogen will cause the vent valve 19 to slowly vent vaporized nitrogen to the atmosphere, maintaining pressure in the Dewar at the specified operating pressure. This pressure is used to force the refrigerant into probe 12. Adequate pressure is maintained in the Dewar throughout the expulsion of all of the refrigerant by causing vapor space 16 to be approximately one-half of the total Dewar volume. As refrigerant is forced from the Dewar, vapor in space 16 expands to a lower pressure until it reaches approximately one-half the original pressure that existed at the initiation of refrigerant flow. Vent valve 19 also provides conventional protection by venting any excessive pressures that should build up through some malfunctron.
Pressure in excess of atmospheric applied to the surface of liquid nitrogen 13 forces it up into vertical pipe 20, through valve 17 and flexible tubing 21 and into supply tube 22 of probe 12. In the course of passage through these tubes, the liquid nitrogen evaporates and forms gaseous nitrogen. The nitrogen fluid emerges from orifice 23 at the end of supply tube 22 and flows into bladder Ill, forcing it to expand into contact with the inner surface of uterus 10. The cold nitrogen gas absorbs heat from bladder 11 and then flows through ports 24 into return tube 25 and is vented to the atmosphere at the open end 26 of tube 25. The arrows in FIG. 1 indicate the flow of nitrogen in probe 12. Supply tube 22 and return tube 25 are rigid or semi-rigid coaxial tubes. The passage of return tube 25 surrounds supply tube 22 and bladder 11 is attached to the outside surface of tube 25 by a suitable low temperature cement. The end of supply tube 22 is open to the interior of bladder I l by virtue of orifice 23, and that end of return tube 25 is closed by being joined to the periphery of the end of tube 22. The size of orifice 23 may be quite large, in relation to the diameter of tube 22, since only low pressure gas is being fed through it and it does not act as an expansion orifice. Ports 24, comprising the only openings into return tube 25 or the inside of bladder 11, are located well back from the end of the tubes in order to force the nitrogen expelled from orifice 23 to circulate and absorb heat from bladder 11 before being vented to the atmosphere through return tube 25.
The probe 12 and its attached bladder 11 must be sufficiently small, when the bladder is deflated and furled around tube 25, so that it can be conveniently inserted into the uterus through a partially dilated cervix. The bladder should be inflated to a pressure sufficient to insure firm contact with the tissue to be necrosed, for example, the interior uterine surface, but should preferably be maintained at about 2 or 3 p.s.i. to avoid any possible risk of internal injury to the patient. It has been found that maintaining a pressure in the Dewar of between -10 p.s.i. results in adequate flow of the nitrogen and the maintenance of proper bladder pressure, although the precise to be used in any case will, of course, need to be adjusted as a function of the various system parameters.
In the system described above, the liquid nitrogen vaporizes in the course of its flow from the Dewar 14 to its exit into bladder 11 at supply tube orifice 23. However, the system may alternatively be operated by permitting the nitrogen to emerge from orifice 23 as a liquid, and allowing it to vaporize in bladder 11.
Bladder 11 must be capable of withstanding cryogenic temperatures without rupturing, and should have as good heat transfer characteristics as obtainable in such materials to provide efficient freezing action. A bladder molded of a heat curing rubber bearing General Electric identification SE-5553 has been found satisfactory.
Testing conducted to date indicates that with the system shown in FIG. 1, the exterior surface of the bladder when completely enclosed in a warm liver reached 73C within five minutes from the start of circulating the nitrogen in the bladder. Based on prior medical testing, at this temperature complete cell necrosis would occur within 2 to 4 minutes. This causes sterilization to be effected.
FIG. 2 shows alternative methods of providing the required pressurization of theDewar in the system of FIG. 1 in which liquid nitrogen is the refrigerant. Instead of allowing the Dewar to become pressurized due to heat conduction, as shown in FIG. 1, one alternate method that may be used to pressurize the vessel 14 is an external source of pressure 52 which is connected with vertical pipe 18 through pipe 51. Pipe 18 feeds down through plug 15 and pressurizes the Dewar l4. Vent valve 19 provides conventional protection by venting the Dewar 14 to the atmosphere should excessive pressure build-up through some malfunction.
A second alternative method of providing the required pressurization of the Dewar 14 is also shown in FIG. 2. Instead of applying an external pressure to the Dewar, the vessel may be pressurized by energizing a heater coil 28 that is submerged in the liquid nitrogen 13 by means of a source of power 29 via wires 30. The
heat generated by the coil causes the liquid nitrogen to vaporize and the vessel pressure accordingly increases and forces the liquid nitrogen through the pipe 20 and tubing system and into the probe 12.
FIG. 3 is a schematic diagram of another system embodiment in which slightly different type of refrigerant is used, that is, a high pressure refrigerant, rather than the liquid nitrogen of the FIG. 1 embodiment which is maintained at atmospheric pressure in a Dewar vessel as described above. The high pressure refrigerants, on the contrary, are stored at high pressure in liquid form in non-vented bottles of high strength. The liquid boils away until the vapor pressure in the bottle is equal to the saturation pressure; the refrigerant then is stored as a liquid at room temperature under its own vaporpressure, so that no Dewar vessel is necessary. Freon 13 and Freon 23, supplied by Dupont, and nitrous oxide, are refrigerants useful for this system in the embodiment shown in FIG. 3. The Freons have a vapor pressure in the neighborhood of 500 p.s.i. and nitrous 0xide, in the neighborhood of 700 p.s.i. at room temperature. Liquid nitrogen, on the other hand, is not suitable for storage at room temperature because its vapor pressure is so high that providing storage containers of sufficient strength is not feasible.
In FIG. 3 a bottle 32 is shown containing both a liquid refrigerant 33 such as Freon or nitrous oxideand gas 34. To operate the system, outlet valve 35 is opened and the high pressure gas is forced through line 36,
through a thrce-way valve 37, and through line 38, into probe 39. Lines 36 and 38 preferablyinclude sections of flexible hose. When the high pressure refrigerant gas reaches probe 39 it is emitted from a small expansion orifice or nozzle and the resulting gaseous expansion causes the cooling. Probe 39, which is merely indicated schematically in FIG. 3, is similar to the probe described in connection with the embodiment of FIG. 1 including the bladder, except that it is provided with expansion orifices, as shown in FIGS. a and 5b, rather than with the rather large supply tube orifice 23 shown in FIG. 1. This is necessary because the nitrogen of the FIG. 1 embodiment is already at cryogenic temperature since it is stored at cryogenic temperature, and cannot lose any heat through expansion on going from high to low pressure, since it is already at a low (substantially atmospheric) pressure. The high pressure refrigerants of the FIG. 3 embodiment, however, since they are stored at room temperature, must obtain their cooling from the expansion of the gas as it is emitted from a high pressure line through a small expansion orifice into the low pressure interior of bladder 11.
FIG. 5a shows a small single expansion orifice 41 located in the end of supply tube 22 and suitable for use in the probe 39 of the high pressure embodiment of FIG. 3. Instead of the single orifice of FIG. 5a, multiple expansion orifices 42, as shown in FIG. 5b, may alternatively be used. As examples of the sizes of orifices suitable for the systems described, an orifice 23 of 0.070 inch has been found suitable for the liquid nitrogen system of FIG. 1. In the high pressure system of FIG. 3, the single expansion orifice (41 of FIG. 5a) of 0.0135 inch has been used for both nitrous oxide and Freon 13 and Freon 23. Two 0.0135 inch expansion orifices (42 of FIG. 5b) or, alternatively, a single orifice (41 of FIG. 6a) of 0.024 inch have been used with both Freons.
The three-way valve 37 is used in connection with the warming of the bladder 11 after the cryogenic necrosis has been effected and prior to removal of the instrument from the uterus. I-Iuman tissue is in large part water and when it is in contact with any object at a temperature below the freezing point of water it sticks to the object by freezing to it, and will tend to tear if the object is pulled away. For this reason, after the cryogenic or freezing cycle of probe 12, in both the embodiments of FIGS. 1 and 3, bladder l l, which is configured to contact substantially the entire area of tissue to be necrosed, must be warmed before it can be removed, so that the contacted tissue is not torn.
In the liquid nitrogen embodiment of FIG. 1 the warming is accomplished by stopping the flow or circulation of cryogenic nitrogen into probe 12 and permitting the heat of the patients body to bring the bladder temperature up. In the high pressure embodiment of FIG. 3, the action of body heat in warming bladder 11 is assisted by circulating room temperature gas through the bladder. This is accomplished by moving the threeway valve 37. The valve, as shown schematically in FIG. 3, is in a position to pass the refrigerant gas from line 36 directly to line 38. By rotating the valve 37 one quarter turn in the counterclockwise direction as shown in FIG. 3, the gas from line 36 will be diverted by valve 37 down into a regulator 43, which may be a Norgren Company Model 11-Ol0-084. Regulator 43 reduces the pressure of the gas to about 50 p.s.i. and then feeds it on through line 38 to probe 39. The gas flows through expansion orifices 41 or 42, but since it is already at a very low pressure, there is hardly any expansion or cooling, and the relatively warm gas flowing through the interior of bladder 11 hastens its warming.
FIG. 4 illustrates an alternative refrigerant feed arrangement for the high pressure embodiment of FIG. 3 wherein some additional cooling capacity is obtained. A high pressure storage bottle 44 is shown in an inverted position, so that its outlet, and the associated outlet valve 45, are located at the bottom of the bottle. In this embodiment liquid refrigerant, rather than gas, is fed from the bottle. This change of state from liquid to gas provides some additional cooling to that obtained by the gaseous expansion during emission from the probe expansion orifices.
The bladder material SE-5553 supplied by General Electric and found satisfactory for the embodiment of FIG. 1 is also satisfactory for the FIG. 3 embodiment, where the refrigerant temperaturees are not nearly as low: nitrogen boils at 320.4F; nitrous oxide at -l29.1F; Freon 13 at l14.6F and Freon 23 at -1 15.7F. In addition a dispersion coating rubber supplied by Dow Corning to their number 92-009 has been found satisfactory for use with the high pressure (and higher temperature) refrigerants of the FIG. 3 embodiment. Bladders of approximately 0.020 inch thickness have been found satisfactory. Various methodsof fabricating the bladder may be employed, but one method found satisfactory has been to lay up the bladder in successive thin layers upon a mandrel. This method of fabrication also facilitates placing one or more thermocouples 50 (FIG. 1a) in the bladder. The thermocouples 50 and their leads may be placed between successive layers as the bladder is formed. Such thermocouples are connected to appropriate instrumentation for monitoring bladder temperature. The embodiments of FIGS. 1 and 3 each have advantages and disadvantages so that neither can clearly be preferred to the other. The liquid nitrogen refrigerant of the FIG. 1 embodiment provides a much lower temperature and consequently faster tissue freezing. It, however, has the disadvantages of imposing more constraints upon system components, especially the bladder, because of the lower temperature, and it requires the inconvenience of low temperature refrigerant storage. The high pressure refrigerants, on the other hand, useful in the FIG. 3 embodiment, are conveniently stored at room temperature, are generally more readily available than liquid nitrogen, but compare unfavorably with liquid nitrogen in having higher temperatures and consequently slower freezing.
In using the cryogenic system of either FIGS. 1 or 3 to sterilize a female, the procedure is as follows. The cervix is visualized and then grasped with a tenaculum. Then the cervix is dilated with instruments to approximately one centimeter. Then probe 12, with bladder 11 furled tightly around it, is inserted through the cervix into the uterus. The refrigerant is applied to probe 12 for a sufficient period to accomplish cryogenic necrosis of the functional endometrium. The bladder 11 is then warmed and removed.
FIGS. 6a through 6e shows in sequential steps the ap' plication of one configuration in which either of the embodiments of FIG. 1 or FIG. 3 may be packaged: a tampon-type cartridge. FIG. 6a shows the package, comprising a handle" portion, which is comprised of supply tube 22 and return tube 25, the bladder 11 furled tightly around the end of return tube 25, and encased in a cylindrical sleeve 48 terminating in an annular shoulder 49. The outside diameter of the sleeve should be less than one centimeter so that it may be readily inserted into the partially dilated cervical openmg.
FIG. 6b shows the sleeve 48 containing the furled probe being inserted into the opening in the cervix 55. In the next step, shown in FIG. 60, sleeve 48 has been fully inserted into the cervix 55, being stopped by the abutment of the sleeve shoulder 49 against the outer cervical surface. Also in FIG. 6c, the probe has been advanced so that the end of the probe with the furled bladder 11 has been pushed free of sleeve 48 and is positioned inside the uterus. When the refrigerant is applied to probe 12, bladder 11 inflates and assumes the position inside the uterus shown by FIG. 6d, showing a view along a vertical plane through the body, and FIG. 6e, showing a view along a horizontal plane through the body.
The primary feature of the package of FIG. 6 is the use of the flanged sleeve 48. This sleeve, which is preferably about 2.5 centimeters in length, is used to protect the bladder from mechanical damage during storage and handling prior to use and also as an insertion aid. Since the inner uterine surface within about 2.5 centimeters of the cervix opening is not part of the functional uterine lining and should not be necrosed, the insulating effect of the sleeve will protect this portion of the uterus and prevent an unnecessary loss of cooling capacity.
While certain illustrative embodiments and details thereof have been set forth herein, various changes and modifications thereto as will occur to those skilled in the art may be made without departing from the scope of the invention, which is defined only in the appended claims. For example, although the embodiments of the invention have been described in detail in connection with the female sterilization application, it should be appreciated that by appropriately configuring the bladder, tissue other than the uterine lining tissue can be necrosed and the uterus lining or a portion thereof may be necrosed for purposes other than sterilization such as for treating dysfunctional uterine bleeding.
What is claimed is:
l. A method of effecting coagulation necrosis of the functional uterine endometrium comprising the steps of:
a. inserting a flexible bladder in the uterus;
b. inflating said bladder with a fluid at cryogenic temperature so that said bladder is in substantially continuous contact with the inner surface of the functional uterine lining, said fluid undergoing a phase change to extract heat from said functional uterine endometrium; and
. maintaining said bladder. so inflated with said fluid at said cryogenic temperature for a period of time sufficient for said heat extraction to effect simultaneous coagulation necrosis of substantially all of the functional uterine endometrium.
2. A method as defined in claim 1 wherein the exte- 0 rior of said bladder in contact with said lining is main-
Claims (2)
1. A method of effecting coagulation necrosis of the functional uterine endometrium comprising the steps of: a. inserting a flexible bladder in the uterus; b. inflating said bladder with a fluid at cryogenic temperature so that said bladder is in substantially continuous contact with the inner surface of the functional uterine lining, said fluid undergoing a phase change to extract heat from said functional uterine endometrium; and c. maintaining said bladder so inflated with said fluid at said cryogenic temperature for a period of time sufficient for said heat extraction to effect simultaneous coagulation necrosis of substantially all of the functional uterine endometrium.
2. A method as defined in claim 1 wherein the exterior of said bladder in contact with said lining is maintained at least -73*C for at least 2 minutes.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US311069A US3924628A (en) | 1972-12-01 | 1972-12-01 | Cyrogenic bladder for necrosing tissue cells |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US311069A US3924628A (en) | 1972-12-01 | 1972-12-01 | Cyrogenic bladder for necrosing tissue cells |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US3924628A true US3924628A (en) | 1975-12-09 |
Family
ID=23205251
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US311069A Expired - Lifetime US3924628A (en) | 1972-12-01 | 1972-12-01 | Cyrogenic bladder for necrosing tissue cells |
Country Status (1)
| Country | Link |
|---|---|
| US (1) | US3924628A (en) |
Cited By (187)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4275734A (en) * | 1977-08-12 | 1981-06-30 | Valleylab, Inc. | Cryosurgical apparatus and method |
| US4367743A (en) * | 1976-05-24 | 1983-01-11 | Virginia M. Gregory | Self-pressurizing cryogenic apparatus and method |
| US4646735A (en) * | 1985-10-04 | 1987-03-03 | Seney John S | Pain-alleviating tissue treatment assembly |
| US4865028A (en) * | 1987-03-05 | 1989-09-12 | Swart Wilhelmus J B | Device for carrying out a therapeutic treatment by means of a refrigerant |
| US4949718A (en) * | 1988-09-09 | 1990-08-21 | Gynelab Products | Intrauterine cauterizing apparatus |
| US5084044A (en) * | 1989-07-14 | 1992-01-28 | Ciron Corporation | Apparatus for endometrial ablation and method of using same |
| US5108390A (en) * | 1988-11-14 | 1992-04-28 | Frigitronics, Inc. | Flexible cryoprobe |
| US5195965A (en) * | 1991-03-07 | 1993-03-23 | Shantha Totada R | Method and apparatus for localized treatment of human viral infections and cancers |
| US5211631A (en) * | 1991-07-24 | 1993-05-18 | Sheaff Charles M | Patient warming apparatus |
| US5242390A (en) * | 1991-05-03 | 1993-09-07 | Goldrath Milton H | Endometrium coagulating surgical method for thermal destruction of the endometrium |
| US5277201A (en) * | 1992-05-01 | 1994-01-11 | Vesta Medical, Inc. | Endometrial ablation apparatus and method |
| GB2269107A (en) * | 1992-07-31 | 1994-02-02 | Spembly Medical Ltd | Cryosurgical ablation |
| EP0624347A1 (en) * | 1993-05-10 | 1994-11-17 | Browning Healthcare Limited | Cryoprobe |
| US5366490A (en) * | 1992-08-12 | 1994-11-22 | Vidamed, Inc. | Medical probe device and method |
| US5385544A (en) * | 1992-08-12 | 1995-01-31 | Vidamed, Inc. | BPH ablation method and apparatus |
| US5409453A (en) * | 1992-08-12 | 1995-04-25 | Vidamed, Inc. | Steerable medical probe with stylets |
| WO1995013026A1 (en) * | 1993-11-12 | 1995-05-18 | Neuwirth Robert S | Intrauterine cryoablation cauterizing apparatus and method |
| US5421819A (en) * | 1992-08-12 | 1995-06-06 | Vidamed, Inc. | Medical probe device |
| US5435805A (en) * | 1992-08-12 | 1995-07-25 | Vidamed, Inc. | Medical probe device with optical viewing capability |
| US5437629A (en) * | 1994-04-14 | 1995-08-01 | Bei Medical Systems | Fluid delivery system for hysteroscopic endometrial ablation |
| US5443470A (en) * | 1992-05-01 | 1995-08-22 | Vesta Medical, Inc. | Method and apparatus for endometrial ablation |
| US5449380A (en) * | 1993-09-17 | 1995-09-12 | Origin Medsystems, Inc. | Apparatus and method for organ ablation |
| US5456662A (en) * | 1993-02-02 | 1995-10-10 | Edwards; Stuart D. | Method for reducing snoring by RF ablation of the uvula |
| US5470308A (en) * | 1992-08-12 | 1995-11-28 | Vidamed, Inc. | Medical probe with biopsy stylet |
| US5505730A (en) * | 1994-06-24 | 1996-04-09 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5514131A (en) * | 1992-08-12 | 1996-05-07 | Stuart D. Edwards | Method for the ablation treatment of the uvula |
| US5522870A (en) * | 1993-01-25 | 1996-06-04 | State Of Israel, Ministry Of Defense, Rafael-Armaments Development Authority | Fast changing heating-cooling device and method |
| US5540679A (en) * | 1992-10-05 | 1996-07-30 | Boston Scientific Corporation | Device and method for heating tissue in a patient's body |
| US5540062A (en) * | 1993-11-01 | 1996-07-30 | State Of Israel, Ministry Of Defence, Rafael Armaments Development Authority | Controlled cryogenic contact system |
| US5542915A (en) * | 1992-08-12 | 1996-08-06 | Vidamed, Inc. | Thermal mapping catheter with ultrasound probe |
| US5556377A (en) * | 1992-08-12 | 1996-09-17 | Vidamed, Inc. | Medical probe apparatus with laser and/or microwave monolithic integrated circuit probe |
| US5562720A (en) * | 1992-05-01 | 1996-10-08 | Vesta Medical, Inc. | Bipolar/monopolar endometrial ablation device and method |
| US5575788A (en) * | 1994-06-24 | 1996-11-19 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5599295A (en) * | 1992-08-12 | 1997-02-04 | Vidamed, Inc. | Medical probe apparatus with enhanced RF, resistance heating, and microwave ablation capabilities |
| US5630794A (en) * | 1992-08-12 | 1997-05-20 | Vidamed, Inc. | Catheter tip and method of manufacturing |
| US5672153A (en) * | 1992-08-12 | 1997-09-30 | Vidamed, Inc. | Medical probe device and method |
| US5681308A (en) * | 1994-06-24 | 1997-10-28 | Stuart D. Edwards | Ablation apparatus for cardiac chambers |
| US5693077A (en) * | 1994-09-21 | 1997-12-02 | Friedman; Mark H. | Treatment of vascular and tension headache atypical facial pain allergic rhinitis and cervical muscle hyperactivity |
| US5720719A (en) * | 1992-08-12 | 1998-02-24 | Vidamed, Inc. | Ablative catheter with conformable body |
| US5769880A (en) * | 1996-04-12 | 1998-06-23 | Novacept | Moisture transport system for contact electrocoagulation |
| US5800493A (en) * | 1995-04-26 | 1998-09-01 | Gynecare, Inc. | Intrauterine ablation system |
| WO1998046148A1 (en) * | 1997-04-14 | 1998-10-22 | The Johns Hopkins University | Endoscopic cryospray device |
| US5891457A (en) * | 1997-05-12 | 1999-04-06 | Neuwirth; Robert S. | Intrauterine chemical necrosing method, composition, and apparatus |
| US5957917A (en) * | 1995-01-20 | 1999-09-28 | Miravant Systems, Inc. | Transluminal hyperthermia catheter and method for use |
| WO1999060960A1 (en) * | 1998-05-22 | 1999-12-02 | James Elliott | Endometrial ablation method and apparatus |
| US6027499A (en) * | 1997-05-23 | 2000-02-22 | Fiber-Tech Medical, Inc. (Assignee Of Jennifer B. Cartledge) | Method and apparatus for cryogenic spray ablation of gastrointestinal mucosa |
| US6066132A (en) * | 1998-06-30 | 2000-05-23 | Ethicon, Inc. | Articulating endometrial ablation device |
| US6161543A (en) * | 1993-02-22 | 2000-12-19 | Epicor, Inc. | Methods of epicardial ablation for creating a lesion around the pulmonary veins |
| US6182666B1 (en) | 1996-12-26 | 2001-02-06 | Cryogen, Inc. | Cryosurgical probe and method for uterine ablation |
| US6187346B1 (en) | 1995-06-07 | 2001-02-13 | Ablation Products, Inc. | Intrauterine chemical cauterizing method and composition |
| US6197351B1 (en) | 1995-06-07 | 2001-03-06 | Robert S. Neuwirth | Intrauterine chemical necrosing method and composition |
| US6224591B1 (en) | 1995-03-01 | 2001-05-01 | Atos Medical Ab | Device and method for hyperthermia treatment |
| US6270494B1 (en) | 1996-12-26 | 2001-08-07 | Cryogen, Inc. | Stretchable cryoprobe sheath |
| US6306129B1 (en) | 1997-09-22 | 2001-10-23 | Femrx, Inc. | Cryosurgical system and method |
| US6395012B1 (en) * | 2000-05-04 | 2002-05-28 | Inbae Yoon | Apparatus and method for delivering and deploying an expandable body member in a uterine cavity |
| US6443947B1 (en) | 2000-03-01 | 2002-09-03 | Alexei Marko | Device for thermal ablation of a cavity |
| US6530234B1 (en) | 1995-10-12 | 2003-03-11 | Cryogen, Inc. | Precooling system for Joule-Thomson probe |
| US6547784B1 (en) * | 2000-06-23 | 2003-04-15 | Ethicon, Inc. | System and method for placement of a surgical instrument in a body cavity |
| US6551274B2 (en) | 2000-02-29 | 2003-04-22 | Biosense Webster, Inc. | Cryoablation catheter with an expandable cooling chamber |
| US6585728B2 (en) * | 2001-05-25 | 2003-07-01 | Biosense Webster, Inc. | Cryoablation catheter with an improved gas expansion chamber |
| US6634083B1 (en) | 1999-01-22 | 2003-10-21 | Seagate Technology Llc | Method of forming a magnet/keeper assembly for head level microactuator |
| US20040002698A1 (en) * | 2002-06-26 | 2004-01-01 | Ethicon, Inc. | Thermal ablation with deployable cage |
| US6758831B2 (en) | 2001-09-24 | 2004-07-06 | Ethicon, Inc. | Device and method for aligning with the tubal ostium |
| US6813520B2 (en) | 1996-04-12 | 2004-11-02 | Novacept | Method for ablating and/or coagulating tissue using moisture transport |
| US20050085880A1 (en) * | 1996-04-12 | 2005-04-21 | Csaba Truckai | Moisture transport system for contact electrocoagulation |
| US20050113857A1 (en) * | 2000-12-27 | 2005-05-26 | Martin Nohilly | Surgical balloon having varying wall thickness |
| WO2005063136A3 (en) * | 2003-12-22 | 2005-08-25 | Ams Res Corp | Cryosurgical devices and methods for endometrial ablation |
| US20050222650A1 (en) * | 2004-04-05 | 2005-10-06 | Seacoast Technologies, Inc. | Neurosurgical device for thermally affecting tissue having invertable lumen |
| US6953469B2 (en) | 2001-08-30 | 2005-10-11 | Ethicon, Inc, | Device and method for treating intraluminal tissue |
| US20050251121A1 (en) * | 2004-05-10 | 2005-11-10 | Scimed Life Systems, Inc. | Probe based low temperature lesion formation apparatus, systems and methods |
| US20050251122A1 (en) * | 2004-05-10 | 2005-11-10 | Scimed Life Systems, Inc. | Clamp based low temperature lesion formation apparatus, system and methods |
| US7083620B2 (en) | 2002-10-30 | 2006-08-01 | Medtronic, Inc. | Electrosurgical hemostat |
| US7094235B2 (en) | 2001-04-26 | 2006-08-22 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US7118566B2 (en) | 2002-05-16 | 2006-10-10 | Medtronic, Inc. | Device and method for needle-less interstitial injection of fluid for ablation of cardiac tissue |
| US7128740B2 (en) | 1996-05-03 | 2006-10-31 | Jacobs Clemens J | Method for interrupting conduction paths within the heart |
| US20060259029A1 (en) * | 1996-10-11 | 2006-11-16 | Utley David S | Method for Cryogenic Tissue Ablation |
| US7156845B2 (en) | 1998-07-07 | 2007-01-02 | Medtronic, Inc. | Method and apparatus for creating a bi-polar virtual electrode used for the ablation of tissue |
| US7166105B2 (en) | 1995-02-22 | 2007-01-23 | Medtronic, Inc. | Pen-type electrosurgical instrument |
| US7169144B2 (en) | 1998-07-07 | 2007-01-30 | Medtronic, Inc. | Apparatus and method for creating, maintaining, and controlling a virtual electrode used for the ablation of tissue |
| US7250048B2 (en) | 2001-04-26 | 2007-07-31 | Medtronic, Inc. | Ablation system and method of use |
| US7291142B2 (en) | 2004-05-10 | 2007-11-06 | Boston Scientific Scimed, Inc. | Low temperature lesion formation apparatus, systems and methods |
| US7294143B2 (en) | 2002-05-16 | 2007-11-13 | Medtronic, Inc. | Device and method for ablation of cardiac tissue |
| US7309325B2 (en) | 1998-07-07 | 2007-12-18 | Medtronic, Inc. | Helical needle apparatus for creating a virtual electrode used for the ablation of tissue |
| US20080015562A1 (en) * | 2001-04-26 | 2008-01-17 | Medtronic, Inc. | Transmural ablation systems and methods |
| US7347858B2 (en) | 2001-12-11 | 2008-03-25 | Medtronic, Inc. | Method and system for treatment of atrial tachyarrhythmias |
| WO2008046183A1 (en) | 2006-10-16 | 2008-04-24 | Cryocath Technologies Inc. | Method and apparatus for inflating and deflating balloon catheters |
| US7364578B2 (en) | 2002-01-25 | 2008-04-29 | Medtronic, Inc. | System and method of performing an electrosurgical procedure |
| US7367972B2 (en) | 2001-04-26 | 2008-05-06 | Medtronic, Inc. | Ablation system |
| CN100402005C (en) * | 2005-12-22 | 2008-07-16 | 尹格平 | Multifunctional thermal coagulation therapeutic instrument for gynaecologic disease |
| US7435250B2 (en) | 2000-04-27 | 2008-10-14 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US20080285668A1 (en) * | 2005-10-31 | 2008-11-20 | Lee Young-Dae | Method for Processing Control Information in a Wireless Mobile Communication System |
| US7470272B2 (en) | 1997-07-18 | 2008-12-30 | Medtronic, Inc. | Device and method for ablating tissue |
| US7497857B2 (en) | 2003-04-29 | 2009-03-03 | Medtronic, Inc. | Endocardial dispersive electrode for use with a monopolar RF ablation pen |
| US7507235B2 (en) | 2001-01-13 | 2009-03-24 | Medtronic, Inc. | Method and system for organ positioning and stabilization |
| US20090088735A1 (en) * | 2004-03-23 | 2009-04-02 | Cryocath Technologies Inc. | Method and apparatus for inflating and deflating balloon catheters |
| US20090187181A1 (en) * | 1998-05-20 | 2009-07-23 | Shadduck John H | Surgical instruments and techniques for treating gastro-esophageal reflux disease |
| US7566334B2 (en) | 2004-06-02 | 2009-07-28 | Medtronic, Inc. | Ablation device with jaws |
| US7615015B2 (en) | 2000-01-19 | 2009-11-10 | Medtronic, Inc. | Focused ultrasound ablation devices having selectively actuatable emitting elements and methods of using the same |
| US7628780B2 (en) | 2001-01-13 | 2009-12-08 | Medtronic, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| US7674260B2 (en) | 2005-04-28 | 2010-03-09 | Cytyc Corporation | Emergency hemostasis device utilizing energy |
| US7678108B2 (en) | 2004-06-02 | 2010-03-16 | Medtronic, Inc. | Loop ablation apparatus and method |
| WO2010036434A1 (en) * | 2008-09-29 | 2010-04-01 | Boston Scientific Scimed, Inc. | Fluid recirculation debris handling system |
| US7706882B2 (en) | 2000-01-19 | 2010-04-27 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area |
| US7706894B2 (en) | 2000-10-10 | 2010-04-27 | Medtronic, Inc. | Heart wall ablation/mapping catheter and method |
| US7727228B2 (en) * | 2004-03-23 | 2010-06-01 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US7731712B2 (en) | 2004-12-20 | 2010-06-08 | Cytyc Corporation | Method and system for transcervical tubal occlusion |
| US7740623B2 (en) | 2001-01-13 | 2010-06-22 | Medtronic, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| EP2198797A1 (en) | 2007-08-23 | 2010-06-23 | Aegea Medical, Inc. | Uterine therapy device |
| US7744562B2 (en) | 2003-01-14 | 2010-06-29 | Medtronics, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| US7758580B2 (en) | 2004-06-02 | 2010-07-20 | Medtronic, Inc. | Compound bipolar ablation device and method |
| US7758576B2 (en) | 2004-06-02 | 2010-07-20 | Medtronic, Inc. | Clamping ablation tool and method |
| WO2010085460A1 (en) | 2009-01-20 | 2010-07-29 | Aegea Medical, Inc. | Uterine therapy device and method |
| US7818039B2 (en) | 2000-04-27 | 2010-10-19 | Medtronic, Inc. | Suction stabilized epicardial ablation devices |
| US7824399B2 (en) | 2001-04-26 | 2010-11-02 | Medtronic, Inc. | Ablation system and method of use |
| US7846160B2 (en) | 2006-12-21 | 2010-12-07 | Cytyc Corporation | Method and apparatus for sterilization |
| US7959627B2 (en) | 2005-11-23 | 2011-06-14 | Barrx Medical, Inc. | Precision ablating device |
| US7967816B2 (en) | 2002-01-25 | 2011-06-28 | Medtronic, Inc. | Fluid-assisted electrosurgical instrument with shapeable electrode |
| US7993336B2 (en) | 1999-11-16 | 2011-08-09 | Barrx Medical, Inc. | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US7997278B2 (en) | 2005-11-23 | 2011-08-16 | Barrx Medical, Inc. | Precision ablating method |
| US8012149B2 (en) | 1999-11-16 | 2011-09-06 | Barrx Medical, Inc. | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US8162933B2 (en) | 2000-04-27 | 2012-04-24 | Medtronic, Inc. | Vibration sensitive ablation device and method |
| US8192426B2 (en) | 2004-01-09 | 2012-06-05 | Tyco Healthcare Group Lp | Devices and methods for treatment of luminal tissue |
| US8221402B2 (en) | 2000-01-19 | 2012-07-17 | Medtronic, Inc. | Method for guiding a medical device |
| US8251992B2 (en) | 2007-07-06 | 2012-08-28 | Tyco Healthcare Group Lp | Method and apparatus for gastrointestinal tract ablation to achieve loss of persistent and/or recurrent excess body weight following a weight-loss operation |
| US8273012B2 (en) | 2007-07-30 | 2012-09-25 | Tyco Healthcare Group, Lp | Cleaning device and methods |
| US8333764B2 (en) | 2004-05-12 | 2012-12-18 | Medtronic, Inc. | Device and method for determining tissue thickness and creating cardiac ablation lesions |
| US8398631B2 (en) | 1999-11-16 | 2013-03-19 | Covidien Lp | System and method of treating abnormal tissue in the human esophagus |
| US8409219B2 (en) | 2004-06-18 | 2013-04-02 | Medtronic, Inc. | Method and system for placement of electrical lead inside heart |
| US8439908B2 (en) | 2007-07-06 | 2013-05-14 | Covidien Lp | Ablation in the gastrointestinal tract to achieve hemostasis and eradicate lesions with a propensity for bleeding |
| US8486060B2 (en) | 2006-09-18 | 2013-07-16 | Cytyc Corporation | Power ramping during RF ablation |
| US8512337B2 (en) | 2001-04-26 | 2013-08-20 | Medtronic, Inc. | Method and system for treatment of atrial tachyarrhythmias |
| US8551082B2 (en) | 1998-05-08 | 2013-10-08 | Cytyc Surgical Products | Radio-frequency generator for powering an ablation device |
| US8568409B2 (en) | 2000-03-06 | 2013-10-29 | Medtronic Advanced Energy Llc | Fluid-assisted medical devices, systems and methods |
| US8632533B2 (en) | 2009-02-23 | 2014-01-21 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical device |
| US20140026395A1 (en) * | 2010-05-28 | 2014-01-30 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical devices, and methods of manufacture thereof |
| US8641711B2 (en) | 2007-05-04 | 2014-02-04 | Covidien Lp | Method and apparatus for gastrointestinal tract ablation for treatment of obesity |
| US8646460B2 (en) | 2007-07-30 | 2014-02-11 | Covidien Lp | Cleaning device and methods |
| US8663245B2 (en) | 2004-06-18 | 2014-03-04 | Medtronic, Inc. | Device for occlusion of a left atrial appendage |
| US8702694B2 (en) | 2005-11-23 | 2014-04-22 | Covidien Lp | Auto-aligning ablating device and method of use |
| US8784338B2 (en) | 2007-06-22 | 2014-07-22 | Covidien Lp | Electrical means to normalize ablational energy transmission to a luminal tissue surface of varying size |
| US8801707B2 (en) | 2004-05-14 | 2014-08-12 | Medtronic, Inc. | Method and devices for treating atrial fibrillation by mass ablation |
| US8821488B2 (en) | 2008-05-13 | 2014-09-02 | Medtronic, Inc. | Tissue lesion evaluation |
| US8858543B2 (en) | 2011-02-01 | 2014-10-14 | Channel Medsystems, Inc. | Cyrogenic treatment systems |
| US8906012B2 (en) | 2010-06-30 | 2014-12-09 | Medtronic Advanced Energy Llc | Electrosurgical devices with wire electrode |
| US8926635B2 (en) | 2004-06-18 | 2015-01-06 | Medtronic, Inc. | Methods and devices for occlusion of an atrial appendage |
| US8945107B2 (en) | 2010-10-26 | 2015-02-03 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US9017317B2 (en) | 2012-12-06 | 2015-04-28 | Medtronic Ardian Luxembourg S.A.R.L. | Refrigerant supply system for cryotherapy including refrigerant recompression and associated devices, systems, and methods |
| US9060754B2 (en) | 2010-10-26 | 2015-06-23 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US9095321B2 (en) | 2012-11-21 | 2015-08-04 | Medtronic Ardian Luxembourg S.A.R.L. | Cryotherapeutic devices having integral multi-helical balloons and methods of making the same |
| US9138289B2 (en) | 2010-06-28 | 2015-09-22 | Medtronic Advanced Energy Llc | Electrode sheath for electrosurgical device |
| US9227088B2 (en) | 2006-05-25 | 2016-01-05 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US9241752B2 (en) | 2012-04-27 | 2016-01-26 | Medtronic Ardian Luxembourg S.A.R.L. | Shafts with pressure relief in cryotherapeutic catheters and associated devices, systems, and methods |
| US9283108B2 (en) | 2009-11-26 | 2016-03-15 | Hans I. Wallsten | Sterilisation device |
| US9381061B2 (en) | 2000-03-06 | 2016-07-05 | Medtronic Advanced Energy Llc | Fluid-assisted medical devices, systems and methods |
| US9561068B2 (en) | 2008-10-06 | 2017-02-07 | Virender K. Sharma | Method and apparatus for tissue ablation |
| US9561066B2 (en) | 2008-10-06 | 2017-02-07 | Virender K. Sharma | Method and apparatus for tissue ablation |
| US9561067B2 (en) | 2008-10-06 | 2017-02-07 | Virender K. Sharma | Method and apparatus for tissue ablation |
| US9662060B2 (en) | 2011-10-07 | 2017-05-30 | Aegea Medical Inc. | Integrity testing method and apparatus for delivering vapor to the uterus |
| US9700365B2 (en) | 2008-10-06 | 2017-07-11 | Santa Anna Tech Llc | Method and apparatus for the ablation of gastrointestinal tissue |
| US9743974B2 (en) | 2010-11-09 | 2017-08-29 | Aegea Medical Inc. | Positioning method and apparatus for delivering vapor to the uterus |
| US9895183B2 (en) | 2013-09-17 | 2018-02-20 | Channel Medsystems, Inc. | Liner for cryogenic treatment systems |
| US9956029B2 (en) | 2014-10-31 | 2018-05-01 | Medtronic Advanced Energy Llc | Telescoping device with saline irrigation line |
| US9974599B2 (en) | 2014-08-15 | 2018-05-22 | Medtronic Ps Medical, Inc. | Multipurpose electrosurgical device |
| US9993290B2 (en) | 2014-05-22 | 2018-06-12 | Aegea Medical Inc. | Systems and methods for performing endometrial ablation |
| US10004550B2 (en) | 2010-08-05 | 2018-06-26 | Medtronic Ardian Luxembourg S.A.R.L. | Cryoablation apparatuses, systems, and methods for renal neuromodulation |
| US10064697B2 (en) | 2008-10-06 | 2018-09-04 | Santa Anna Tech Llc | Vapor based ablation system for treating various indications |
| US10179019B2 (en) | 2014-05-22 | 2019-01-15 | Aegea Medical Inc. | Integrity testing method and apparatus for delivering vapor to the uterus |
| US20190083298A1 (en) * | 2017-09-21 | 2019-03-21 | Michael W. Starkweather | Whole body cryotherapy system |
| US10278774B2 (en) | 2011-03-18 | 2019-05-07 | Covidien Lp | Selectively expandable operative element support structure and methods of use |
| US10335280B2 (en) | 2000-01-19 | 2019-07-02 | Medtronic, Inc. | Method for ablating target tissue of a patient |
| US10492842B2 (en) | 2014-03-07 | 2019-12-03 | Medtronic Ardian Luxembourg S.A.R.L. | Monitoring and controlling internally administered cryotherapy |
| US20190365453A1 (en) * | 2017-02-17 | 2019-12-05 | Boston Scientific Scimed, Inc. | System and method for limiting differential pressure across proportional valve during cryoablation procedures |
| US10588682B2 (en) | 2011-04-25 | 2020-03-17 | Medtronic Ardian Luxembourg S.A.R.L. | Apparatus and methods related to constrained deployment of cryogenic balloons for limited cryogenic ablation of vessel walls |
| US10610279B2 (en) | 2014-04-10 | 2020-04-07 | Channel Medsystems, Inc. | Apparatus and methods for regulating cryogenic treatment |
| US10631914B2 (en) | 2013-09-30 | 2020-04-28 | Covidien Lp | Bipolar electrosurgical instrument with movable electrode and related systems and methods |
| US10660697B2 (en) | 2009-11-10 | 2020-05-26 | Cardea Medsystems (Tianjin) Co., Ltd. | Hollow body cavity ablation apparatus |
| US10695126B2 (en) | 2008-10-06 | 2020-06-30 | Santa Anna Tech Llc | Catheter with a double balloon structure to generate and apply a heated ablative zone to tissue |
| US10758303B2 (en) | 2009-11-10 | 2020-09-01 | Cardea Medsystems (Tianjin) Co., Ltd. | Hollow body cavity ablation apparatus |
| US10905490B2 (en) | 2012-04-27 | 2021-02-02 | Medtronic Ardian Luxembourg S.A.R.L. | Cryotherapeutic devices for renal neuromodulation and associated systems and methods |
| US11207118B2 (en) | 2007-07-06 | 2021-12-28 | Tsunami Medtech, Llc | Medical system and method of use |
| US11284931B2 (en) | 2009-02-03 | 2022-03-29 | Tsunami Medtech, Llc | Medical systems and methods for ablating and absorbing tissue |
| US11331140B2 (en) | 2016-05-19 | 2022-05-17 | Aqua Heart, Inc. | Heated vapor ablation systems and methods for treating cardiac conditions |
| US11331037B2 (en) | 2016-02-19 | 2022-05-17 | Aegea Medical Inc. | Methods and apparatus for determining the integrity of a bodily cavity |
| US11413086B2 (en) | 2013-03-15 | 2022-08-16 | Tsunami Medtech, Llc | Medical system and method of use |
| US11806066B2 (en) | 2018-06-01 | 2023-11-07 | Santa Anna Tech Llc | Multi-stage vapor-based ablation treatment methods and vapor generation and delivery systems |
| US12364537B2 (en) | 2016-05-02 | 2025-07-22 | Santa Anna Tech Llc | Catheter with a double balloon structure to generate and apply a heated ablative zone to tissue |
| US12458425B2 (en) | 2022-07-12 | 2025-11-04 | Covidien Lp | Thermal heating element for use with endometrial ablation |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US612724A (en) * | 1898-10-18 | Bert j | ||
| US3174481A (en) * | 1963-10-14 | 1965-03-23 | Seaman Louis | Stomach freezing system with inflatable distributor means |
| US3460538A (en) * | 1967-10-20 | 1969-08-12 | Edward T Armstrong | Hypothermia apparatus and method for treating the human body and the like |
| US3507283A (en) * | 1967-10-11 | 1970-04-21 | Univ Northwestern | Cryosurgical instrument |
| US3670732A (en) * | 1970-05-11 | 1972-06-20 | Ralph R Robinson | Vacuum curette |
-
1972
- 1972-12-01 US US311069A patent/US3924628A/en not_active Expired - Lifetime
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US612724A (en) * | 1898-10-18 | Bert j | ||
| US3174481A (en) * | 1963-10-14 | 1965-03-23 | Seaman Louis | Stomach freezing system with inflatable distributor means |
| US3507283A (en) * | 1967-10-11 | 1970-04-21 | Univ Northwestern | Cryosurgical instrument |
| US3460538A (en) * | 1967-10-20 | 1969-08-12 | Edward T Armstrong | Hypothermia apparatus and method for treating the human body and the like |
| US3670732A (en) * | 1970-05-11 | 1972-06-20 | Ralph R Robinson | Vacuum curette |
Cited By (372)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4367743A (en) * | 1976-05-24 | 1983-01-11 | Virginia M. Gregory | Self-pressurizing cryogenic apparatus and method |
| US4275734A (en) * | 1977-08-12 | 1981-06-30 | Valleylab, Inc. | Cryosurgical apparatus and method |
| US4646735A (en) * | 1985-10-04 | 1987-03-03 | Seney John S | Pain-alleviating tissue treatment assembly |
| WO1988005646A1 (en) * | 1985-10-04 | 1988-08-11 | John Seymour Seney | A pain-alleviating tissue treatment assembly |
| US4865028A (en) * | 1987-03-05 | 1989-09-12 | Swart Wilhelmus J B | Device for carrying out a therapeutic treatment by means of a refrigerant |
| US4949718A (en) * | 1988-09-09 | 1990-08-21 | Gynelab Products | Intrauterine cauterizing apparatus |
| US5105808A (en) * | 1988-09-09 | 1992-04-21 | Gynelab Products | Intrauterine cauterizing method |
| US5108390A (en) * | 1988-11-14 | 1992-04-28 | Frigitronics, Inc. | Flexible cryoprobe |
| US5084044A (en) * | 1989-07-14 | 1992-01-28 | Ciron Corporation | Apparatus for endometrial ablation and method of using same |
| US5195965A (en) * | 1991-03-07 | 1993-03-23 | Shantha Totada R | Method and apparatus for localized treatment of human viral infections and cancers |
| US5451208A (en) * | 1991-05-03 | 1995-09-19 | Goldrath; Milton H. | Endometrium coagulating apparatus and surgical method for thermal destruction of the endometrium |
| US5242390A (en) * | 1991-05-03 | 1993-09-07 | Goldrath Milton H | Endometrium coagulating surgical method for thermal destruction of the endometrium |
| US5211631A (en) * | 1991-07-24 | 1993-05-18 | Sheaff Charles M | Patient warming apparatus |
| US5279598A (en) * | 1991-07-24 | 1994-01-18 | Sheaff Charles M | Patient warming methods |
| US5277201A (en) * | 1992-05-01 | 1994-01-11 | Vesta Medical, Inc. | Endometrial ablation apparatus and method |
| US5713942A (en) * | 1992-05-01 | 1998-02-03 | Vesta Medical, Inc. | Body cavity ablation apparatus and model |
| US5562720A (en) * | 1992-05-01 | 1996-10-08 | Vesta Medical, Inc. | Bipolar/monopolar endometrial ablation device and method |
| US6041260A (en) * | 1992-05-01 | 2000-03-21 | Vesta Medical, Inc. | Method and apparatus for endometrial ablation |
| US5443470A (en) * | 1992-05-01 | 1995-08-22 | Vesta Medical, Inc. | Method and apparatus for endometrial ablation |
| GB2269107A (en) * | 1992-07-31 | 1994-02-02 | Spembly Medical Ltd | Cryosurgical ablation |
| US5403309A (en) * | 1992-07-31 | 1995-04-04 | Spembly Medical Limited | Cryosurgical ablation |
| EP0584930A1 (en) * | 1992-07-31 | 1994-03-02 | Spembly Medical Limited | Cryosurgical ablation |
| GB2269107B (en) * | 1992-07-31 | 1996-05-08 | Spembly Medical Ltd | Cryosurgical ablation |
| US5672153A (en) * | 1992-08-12 | 1997-09-30 | Vidamed, Inc. | Medical probe device and method |
| US5421819A (en) * | 1992-08-12 | 1995-06-06 | Vidamed, Inc. | Medical probe device |
| US5435805A (en) * | 1992-08-12 | 1995-07-25 | Vidamed, Inc. | Medical probe device with optical viewing capability |
| US5385544A (en) * | 1992-08-12 | 1995-01-31 | Vidamed, Inc. | BPH ablation method and apparatus |
| US5370675A (en) * | 1992-08-12 | 1994-12-06 | Vidamed, Inc. | Medical probe device and method |
| US5720718A (en) * | 1992-08-12 | 1998-02-24 | Vidamed, Inc. | Medical probe apparatus with enhanced RF, resistance heating, and microwave ablation capabilities |
| US5470308A (en) * | 1992-08-12 | 1995-11-28 | Vidamed, Inc. | Medical probe with biopsy stylet |
| US5470309A (en) * | 1992-08-12 | 1995-11-28 | Vidamed, Inc. | Medical ablation apparatus utilizing a heated stylet |
| US6206847B1 (en) | 1992-08-12 | 2001-03-27 | Vidamed, Inc. | Medical probe device |
| US5599295A (en) * | 1992-08-12 | 1997-02-04 | Vidamed, Inc. | Medical probe apparatus with enhanced RF, resistance heating, and microwave ablation capabilities |
| US5514131A (en) * | 1992-08-12 | 1996-05-07 | Stuart D. Edwards | Method for the ablation treatment of the uvula |
| US6464661B2 (en) | 1992-08-12 | 2002-10-15 | Vidamed, Inc. | Medical probe with stylets |
| US5630794A (en) * | 1992-08-12 | 1997-05-20 | Vidamed, Inc. | Catheter tip and method of manufacturing |
| US5599294A (en) * | 1992-08-12 | 1997-02-04 | Vidamed, Inc. | Microwave probe device and method |
| US5366490A (en) * | 1992-08-12 | 1994-11-22 | Vidamed, Inc. | Medical probe device and method |
| US5542915A (en) * | 1992-08-12 | 1996-08-06 | Vidamed, Inc. | Thermal mapping catheter with ultrasound probe |
| US5554110A (en) * | 1992-08-12 | 1996-09-10 | Vidamed, Inc. | Medical ablation apparatus |
| US5556377A (en) * | 1992-08-12 | 1996-09-17 | Vidamed, Inc. | Medical probe apparatus with laser and/or microwave monolithic integrated circuit probe |
| US6022334A (en) * | 1992-08-12 | 2000-02-08 | Vidamed, Inc. | Medical probe device with optic viewing capability |
| US5409453A (en) * | 1992-08-12 | 1995-04-25 | Vidamed, Inc. | Steerable medical probe with stylets |
| US5895370A (en) * | 1992-08-12 | 1999-04-20 | Vidamed, Inc. | Medical probe (with stylets) device |
| US5607389A (en) * | 1992-08-12 | 1997-03-04 | Vidamed, Inc. | Medical probe with biopsy stylet |
| US5720719A (en) * | 1992-08-12 | 1998-02-24 | Vidamed, Inc. | Ablative catheter with conformable body |
| US5540679A (en) * | 1992-10-05 | 1996-07-30 | Boston Scientific Corporation | Device and method for heating tissue in a patient's body |
| US5891188A (en) * | 1993-01-25 | 1999-04-06 | State Of Israel, Ministry Of Defense, Rafael-Armaments Development Authority | Fast changing heating-cooling device and method |
| US5522870A (en) * | 1993-01-25 | 1996-06-04 | State Of Israel, Ministry Of Defense, Rafael-Armaments Development Authority | Fast changing heating-cooling device and method |
| US5702435A (en) * | 1993-01-25 | 1997-12-30 | State Of Israel Ministry Of Defense, Rafael-Armaments | Fast changing heating-cooling device and method |
| US5456662A (en) * | 1993-02-02 | 1995-10-10 | Edwards; Stuart D. | Method for reducing snoring by RF ablation of the uvula |
| US6161543A (en) * | 1993-02-22 | 2000-12-19 | Epicor, Inc. | Methods of epicardial ablation for creating a lesion around the pulmonary veins |
| EP0624347A1 (en) * | 1993-05-10 | 1994-11-17 | Browning Healthcare Limited | Cryoprobe |
| US5449380A (en) * | 1993-09-17 | 1995-09-12 | Origin Medsystems, Inc. | Apparatus and method for organ ablation |
| US5577387A (en) * | 1993-11-01 | 1996-11-26 | State Of Israel, Ministry Of Defence, Rafael-Armaments Development Authority | Controlled cryogenic contact system |
| US5540062A (en) * | 1993-11-01 | 1996-07-30 | State Of Israel, Ministry Of Defence, Rafael Armaments Development Authority | Controlled cryogenic contact system |
| US5501681A (en) * | 1993-11-12 | 1996-03-26 | Neuwirth; Robert S. | Intrauterine cryoablation cauterizing apparatus and method |
| WO1995013026A1 (en) * | 1993-11-12 | 1995-05-18 | Neuwirth Robert S | Intrauterine cryoablation cauterizing apparatus and method |
| US5437629A (en) * | 1994-04-14 | 1995-08-01 | Bei Medical Systems | Fluid delivery system for hysteroscopic endometrial ablation |
| US5769846A (en) * | 1994-06-24 | 1998-06-23 | Stuart D. Edwards | Ablation apparatus for cardiac chambers |
| US5558672A (en) * | 1994-06-24 | 1996-09-24 | Vidacare, Inc. | Thin layer ablation apparatus |
| US5827273A (en) * | 1994-06-24 | 1998-10-27 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5505730A (en) * | 1994-06-24 | 1996-04-09 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5575788A (en) * | 1994-06-24 | 1996-11-19 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5681308A (en) * | 1994-06-24 | 1997-10-28 | Stuart D. Edwards | Ablation apparatus for cardiac chambers |
| US5569241A (en) * | 1994-06-24 | 1996-10-29 | Vidacare, Inc. | Thin layer ablation apparatus |
| US5693077A (en) * | 1994-09-21 | 1997-12-02 | Friedman; Mark H. | Treatment of vascular and tension headache atypical facial pain allergic rhinitis and cervical muscle hyperactivity |
| US5957917A (en) * | 1995-01-20 | 1999-09-28 | Miravant Systems, Inc. | Transluminal hyperthermia catheter and method for use |
| US7247155B2 (en) | 1995-02-22 | 2007-07-24 | Medtronic, Inc. | Apparatus and method for creating, maintaining, and controlling a virtual electrode used for the ablation of tissue |
| US7794460B2 (en) | 1995-02-22 | 2010-09-14 | Medtronic, Inc. | Method of ablating tissue |
| US9770282B2 (en) | 1995-02-22 | 2017-09-26 | Medtronic, Inc. | Apparatus and method for creating, maintaining, and controlling a virtual electrode used for the ablation of tissue |
| US7166105B2 (en) | 1995-02-22 | 2007-01-23 | Medtronic, Inc. | Pen-type electrosurgical instrument |
| US7422588B2 (en) | 1995-02-22 | 2008-09-09 | Medtronic, Inc. | Pen-type electrosurgical instrument |
| US6224591B1 (en) | 1995-03-01 | 2001-05-01 | Atos Medical Ab | Device and method for hyperthermia treatment |
| US5800493A (en) * | 1995-04-26 | 1998-09-01 | Gynecare, Inc. | Intrauterine ablation system |
| US6187346B1 (en) | 1995-06-07 | 2001-02-13 | Ablation Products, Inc. | Intrauterine chemical cauterizing method and composition |
| US6197351B1 (en) | 1995-06-07 | 2001-03-06 | Robert S. Neuwirth | Intrauterine chemical necrosing method and composition |
| US6530234B1 (en) | 1995-10-12 | 2003-03-11 | Cryogen, Inc. | Precooling system for Joule-Thomson probe |
| US6813520B2 (en) | 1996-04-12 | 2004-11-02 | Novacept | Method for ablating and/or coagulating tissue using moisture transport |
| US20050267468A1 (en) * | 1996-04-12 | 2005-12-01 | Csaba Truckai | Moisture transport system for contact electrocoagulation |
| US9247989B2 (en) | 1996-04-12 | 2016-02-02 | Cytyc Surgical Products | Moisture transport system for contact electrocoagulation |
| US7512445B2 (en) | 1996-04-12 | 2009-03-31 | Cytyc Corporation | Moisture transport system for contact electrocoagulation |
| US8506563B2 (en) | 1996-04-12 | 2013-08-13 | Cytyc Surgical Products | Moisture transport system for contact electrocoagulation |
| US7604633B2 (en) | 1996-04-12 | 2009-10-20 | Cytyc Corporation | Moisture transport system for contact electrocoagulation |
| US9095348B2 (en) | 1996-04-12 | 2015-08-04 | Cytyc Surgical Products | Moisture transport system for contact electrocoagulation |
| US5769880A (en) * | 1996-04-12 | 1998-06-23 | Novacept | Moisture transport system for contact electrocoagulation |
| US8998898B2 (en) | 1996-04-12 | 2015-04-07 | Cytyc Surgical Products | Moisture transport system for contact electrocoagulation |
| US20050085880A1 (en) * | 1996-04-12 | 2005-04-21 | Csaba Truckai | Moisture transport system for contact electrocoagulation |
| US7128740B2 (en) | 1996-05-03 | 2006-10-31 | Jacobs Clemens J | Method for interrupting conduction paths within the heart |
| US7507234B2 (en) | 1996-10-11 | 2009-03-24 | BÂRRX Medical, Inc. | Method for cryogenic tissue ablation |
| US20060259029A1 (en) * | 1996-10-11 | 2006-11-16 | Utley David S | Method for Cryogenic Tissue Ablation |
| US7556628B2 (en) | 1996-10-11 | 2009-07-07 | BÂRRX Medical, Inc. | Method for tissue ablation |
| US7632268B2 (en) | 1996-10-11 | 2009-12-15 | BÂRRX Medical, Inc. | System for tissue ablation |
| US6193644B1 (en) | 1996-12-26 | 2001-02-27 | Cryogen, Inc. | Cryosurgical probe with sheath |
| US6475212B2 (en) | 1996-12-26 | 2002-11-05 | Cryogen, Inc. | Cryosurgical probe with sheath |
| US6451012B2 (en) | 1996-12-26 | 2002-09-17 | Cryogen, Inc. | Cryosurgical method for endometrial ablation |
| US6270494B1 (en) | 1996-12-26 | 2001-08-07 | Cryogen, Inc. | Stretchable cryoprobe sheath |
| US6182666B1 (en) | 1996-12-26 | 2001-02-06 | Cryogen, Inc. | Cryosurgical probe and method for uterine ablation |
| WO1998046148A1 (en) * | 1997-04-14 | 1998-10-22 | The Johns Hopkins University | Endoscopic cryospray device |
| US5846235A (en) * | 1997-04-14 | 1998-12-08 | Johns Hopkins University | Endoscopic cryospray device |
| RU2246954C2 (en) * | 1997-05-12 | 2005-02-27 | Роберт С. НЬЮВИРТ | Method, composition and set for carrying out intrauterine chemical necrotization |
| US6165492A (en) * | 1997-05-12 | 2000-12-26 | Neuwirth; Robert S. | Intrauterine chemical necrosing method, composition, and apparatus |
| US5891457A (en) * | 1997-05-12 | 1999-04-06 | Neuwirth; Robert S. | Intrauterine chemical necrosing method, composition, and apparatus |
| US6027499A (en) * | 1997-05-23 | 2000-02-22 | Fiber-Tech Medical, Inc. (Assignee Of Jennifer B. Cartledge) | Method and apparatus for cryogenic spray ablation of gastrointestinal mucosa |
| US7470272B2 (en) | 1997-07-18 | 2008-12-30 | Medtronic, Inc. | Device and method for ablating tissue |
| US7678111B2 (en) | 1997-07-18 | 2010-03-16 | Medtronic, Inc. | Device and method for ablating tissue |
| US6306129B1 (en) | 1997-09-22 | 2001-10-23 | Femrx, Inc. | Cryosurgical system and method |
| US7507238B2 (en) | 1998-02-19 | 2009-03-24 | Barrx Medical, Inc | Method for vacuum-assisted tissue ablation |
| US8551082B2 (en) | 1998-05-08 | 2013-10-08 | Cytyc Surgical Products | Radio-frequency generator for powering an ablation device |
| US9554853B2 (en) | 1998-05-08 | 2017-01-31 | Hologic, Inc. | Radio-frequency generator for powering an ablation device |
| US20090187181A1 (en) * | 1998-05-20 | 2009-07-23 | Shadduck John H | Surgical instruments and techniques for treating gastro-esophageal reflux disease |
| WO1999060960A1 (en) * | 1998-05-22 | 1999-12-02 | James Elliott | Endometrial ablation method and apparatus |
| US6066132A (en) * | 1998-06-30 | 2000-05-23 | Ethicon, Inc. | Articulating endometrial ablation device |
| US7699805B2 (en) | 1998-07-07 | 2010-04-20 | Medtronic, Inc. | Helical coil apparatus for ablation of tissue |
| US7156845B2 (en) | 1998-07-07 | 2007-01-02 | Medtronic, Inc. | Method and apparatus for creating a bi-polar virtual electrode used for the ablation of tissue |
| US7169144B2 (en) | 1998-07-07 | 2007-01-30 | Medtronic, Inc. | Apparatus and method for creating, maintaining, and controlling a virtual electrode used for the ablation of tissue |
| US9113896B2 (en) | 1998-07-07 | 2015-08-25 | Medtronic, Inc. | Method and apparatus for creating a bi-polar virtual electrode used for the ablation of tissue |
| US7309325B2 (en) | 1998-07-07 | 2007-12-18 | Medtronic, Inc. | Helical needle apparatus for creating a virtual electrode used for the ablation of tissue |
| US6634083B1 (en) | 1999-01-22 | 2003-10-21 | Seagate Technology Llc | Method of forming a magnet/keeper assembly for head level microactuator |
| US8398631B2 (en) | 1999-11-16 | 2013-03-19 | Covidien Lp | System and method of treating abnormal tissue in the human esophagus |
| US8377055B2 (en) | 1999-11-16 | 2013-02-19 | Covidien Lp | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US9039699B2 (en) | 1999-11-16 | 2015-05-26 | Covidien Lp | Methods and systems for treatment of tissue in a body lumen |
| US9597147B2 (en) | 1999-11-16 | 2017-03-21 | Covidien Lp | Methods and systems for treatment of tissue in a body lumen |
| US8876818B2 (en) | 1999-11-16 | 2014-11-04 | Covidien Lp | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US8012149B2 (en) | 1999-11-16 | 2011-09-06 | Barrx Medical, Inc. | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US7993336B2 (en) | 1999-11-16 | 2011-08-09 | Barrx Medical, Inc. | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US9555222B2 (en) | 1999-11-16 | 2017-01-31 | Covidien Lp | Methods and systems for determining physiologic characteristics for treatment of the esophagus |
| US8221402B2 (en) | 2000-01-19 | 2012-07-17 | Medtronic, Inc. | Method for guiding a medical device |
| US7706882B2 (en) | 2000-01-19 | 2010-04-27 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area |
| US7615015B2 (en) | 2000-01-19 | 2009-11-10 | Medtronic, Inc. | Focused ultrasound ablation devices having selectively actuatable emitting elements and methods of using the same |
| US10335280B2 (en) | 2000-01-19 | 2019-07-02 | Medtronic, Inc. | Method for ablating target tissue of a patient |
| US6551274B2 (en) | 2000-02-29 | 2003-04-22 | Biosense Webster, Inc. | Cryoablation catheter with an expandable cooling chamber |
| JP2003524498A (en) * | 2000-03-01 | 2003-08-19 | サーマル アブレイション テクノロジーズ カナダ リミテッド | Device for thermal ablation of body cavity |
| US7419500B2 (en) | 2000-03-01 | 2008-09-02 | Idoman Limited | Device for thermal ablation of a cavity |
| US6443947B1 (en) | 2000-03-01 | 2002-09-03 | Alexei Marko | Device for thermal ablation of a cavity |
| US8568409B2 (en) | 2000-03-06 | 2013-10-29 | Medtronic Advanced Energy Llc | Fluid-assisted medical devices, systems and methods |
| US9381061B2 (en) | 2000-03-06 | 2016-07-05 | Medtronic Advanced Energy Llc | Fluid-assisted medical devices, systems and methods |
| US7818039B2 (en) | 2000-04-27 | 2010-10-19 | Medtronic, Inc. | Suction stabilized epicardial ablation devices |
| US8162933B2 (en) | 2000-04-27 | 2012-04-24 | Medtronic, Inc. | Vibration sensitive ablation device and method |
| US7435250B2 (en) | 2000-04-27 | 2008-10-14 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US9693819B2 (en) | 2000-04-27 | 2017-07-04 | Medtronic, Inc. | Vibration sensitive ablation device and method |
| US6395012B1 (en) * | 2000-05-04 | 2002-05-28 | Inbae Yoon | Apparatus and method for delivering and deploying an expandable body member in a uterine cavity |
| US6547784B1 (en) * | 2000-06-23 | 2003-04-15 | Ethicon, Inc. | System and method for placement of a surgical instrument in a body cavity |
| US8706260B2 (en) | 2000-10-10 | 2014-04-22 | Medtronic, Inc. | Heart wall ablation/mapping catheter and method |
| US7706894B2 (en) | 2000-10-10 | 2010-04-27 | Medtronic, Inc. | Heart wall ablation/mapping catheter and method |
| US20050113857A1 (en) * | 2000-12-27 | 2005-05-26 | Martin Nohilly | Surgical balloon having varying wall thickness |
| US6951569B2 (en) | 2000-12-27 | 2005-10-04 | Ethicon, Inc. | Surgical balloon having varying wall thickness |
| US7507235B2 (en) | 2001-01-13 | 2009-03-24 | Medtronic, Inc. | Method and system for organ positioning and stabilization |
| US7740623B2 (en) | 2001-01-13 | 2010-06-22 | Medtronic, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| US7628780B2 (en) | 2001-01-13 | 2009-12-08 | Medtronic, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| US7959626B2 (en) | 2001-04-26 | 2011-06-14 | Medtronic, Inc. | Transmural ablation systems and methods |
| US8262649B2 (en) | 2001-04-26 | 2012-09-11 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US7250051B2 (en) | 2001-04-26 | 2007-07-31 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US7250048B2 (en) | 2001-04-26 | 2007-07-31 | Medtronic, Inc. | Ablation system and method of use |
| US7094235B2 (en) | 2001-04-26 | 2006-08-22 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US20080015562A1 (en) * | 2001-04-26 | 2008-01-17 | Medtronic, Inc. | Transmural ablation systems and methods |
| US7824399B2 (en) | 2001-04-26 | 2010-11-02 | Medtronic, Inc. | Ablation system and method of use |
| US8221415B2 (en) | 2001-04-26 | 2012-07-17 | Medtronic, Inc. | Method and apparatus for tissue ablation |
| US8512337B2 (en) | 2001-04-26 | 2013-08-20 | Medtronic, Inc. | Method and system for treatment of atrial tachyarrhythmias |
| US7367972B2 (en) | 2001-04-26 | 2008-05-06 | Medtronic, Inc. | Ablation system |
| US6585728B2 (en) * | 2001-05-25 | 2003-07-01 | Biosense Webster, Inc. | Cryoablation catheter with an improved gas expansion chamber |
| US6953469B2 (en) | 2001-08-30 | 2005-10-11 | Ethicon, Inc, | Device and method for treating intraluminal tissue |
| US6758831B2 (en) | 2001-09-24 | 2004-07-06 | Ethicon, Inc. | Device and method for aligning with the tubal ostium |
| US7347858B2 (en) | 2001-12-11 | 2008-03-25 | Medtronic, Inc. | Method and system for treatment of atrial tachyarrhythmias |
| US8623010B2 (en) | 2002-01-25 | 2014-01-07 | Medtronic, Inc. | Cardiac mapping instrument with shapeable electrode |
| US7364578B2 (en) | 2002-01-25 | 2008-04-29 | Medtronic, Inc. | System and method of performing an electrosurgical procedure |
| US7967816B2 (en) | 2002-01-25 | 2011-06-28 | Medtronic, Inc. | Fluid-assisted electrosurgical instrument with shapeable electrode |
| US7975703B2 (en) | 2002-05-16 | 2011-07-12 | Medtronic, Inc. | Device and method for needle-less interstitial injection of fluid for ablation of cardiac tissue |
| US7294143B2 (en) | 2002-05-16 | 2007-11-13 | Medtronic, Inc. | Device and method for ablation of cardiac tissue |
| US7118566B2 (en) | 2002-05-16 | 2006-10-10 | Medtronic, Inc. | Device and method for needle-less interstitial injection of fluid for ablation of cardiac tissue |
| US8414573B2 (en) | 2002-05-16 | 2013-04-09 | Medtronic, Inc. | Device and method for ablation of cardiac tissue |
| US20040002698A1 (en) * | 2002-06-26 | 2004-01-01 | Ethicon, Inc. | Thermal ablation with deployable cage |
| US6960203B2 (en) | 2002-06-26 | 2005-11-01 | Ethicon, Inc. | Thermal ablation with deployable cage |
| US7083620B2 (en) | 2002-10-30 | 2006-08-01 | Medtronic, Inc. | Electrosurgical hemostat |
| US7963963B2 (en) | 2002-10-30 | 2011-06-21 | Medtronic, Inc. | Electrosurgical hemostat |
| US8273072B2 (en) | 2003-01-14 | 2012-09-25 | Medtronic, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| US7744562B2 (en) | 2003-01-14 | 2010-06-29 | Medtronics, Inc. | Devices and methods for interstitial injection of biologic agents into tissue |
| US7497857B2 (en) | 2003-04-29 | 2009-03-03 | Medtronic, Inc. | Endocardial dispersive electrode for use with a monopolar RF ablation pen |
| US7871409B2 (en) | 2003-04-29 | 2011-01-18 | Medtronic, Inc. | Endocardial dispersive electrode for use with a monopolar RF ablation pen |
| US7500973B2 (en) | 2003-12-22 | 2009-03-10 | Ams Research Corporation | Cryosurgical devices and methods for endometrial ablation |
| US7381208B2 (en) | 2003-12-22 | 2008-06-03 | Ams Research Corporation | Cryosurgical devices for endometrial ablation |
| US20090138000A1 (en) * | 2003-12-22 | 2009-05-28 | Vancelette David W | Cryosurgical devices and methods for endometrial ablation |
| AU2004308416B2 (en) * | 2003-12-22 | 2010-03-18 | Ams Research Corporation | Cryosurgical devices and methods for endometrial ablation |
| WO2005063136A3 (en) * | 2003-12-22 | 2005-08-25 | Ams Res Corp | Cryosurgical devices and methods for endometrial ablation |
| US9393069B2 (en) | 2004-01-09 | 2016-07-19 | Covidien Lp | Devices and methods for treatment of luminal tissue |
| US10278776B2 (en) | 2004-01-09 | 2019-05-07 | Covidien Lp | Devices and methods for treatment of luminal tissue |
| US8192426B2 (en) | 2004-01-09 | 2012-06-05 | Tyco Healthcare Group Lp | Devices and methods for treatment of luminal tissue |
| US10856939B2 (en) | 2004-01-09 | 2020-12-08 | Covidien Lp | Devices and methods for treatment of luminal tissue |
| US9808301B2 (en) | 2004-03-23 | 2017-11-07 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US9555223B2 (en) | 2004-03-23 | 2017-01-31 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US8545491B2 (en) * | 2004-03-23 | 2013-10-01 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US8382747B2 (en) | 2004-03-23 | 2013-02-26 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US11357563B2 (en) * | 2004-03-23 | 2022-06-14 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US7727228B2 (en) * | 2004-03-23 | 2010-06-01 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US8491636B2 (en) * | 2004-03-23 | 2013-07-23 | Medtronic Cryopath LP | Method and apparatus for inflating and deflating balloon catheters |
| US20090088735A1 (en) * | 2004-03-23 | 2009-04-02 | Cryocath Technologies Inc. | Method and apparatus for inflating and deflating balloon catheters |
| US8900222B2 (en) | 2004-03-23 | 2014-12-02 | Medtronic Cryocath Lp | Method and apparatus for inflating and deflating balloon catheters |
| US20050222650A1 (en) * | 2004-04-05 | 2005-10-06 | Seacoast Technologies, Inc. | Neurosurgical device for thermally affecting tissue having invertable lumen |
| US20070219545A1 (en) * | 2004-05-10 | 2007-09-20 | Boston Scientific Scimed Inc. | Probe based low temperature lesion formation apparatus, systems and methods |
| US20080058791A1 (en) * | 2004-05-10 | 2008-03-06 | Boston Scientific Scimed, Inc. | Low temperature lesion formation apparatus, systems and methods |
| US7582083B2 (en) * | 2004-05-10 | 2009-09-01 | Boston Scientific Scimed, Inc. | Probe based low temperature lesion formation apparatus, systems and methods |
| US7291143B2 (en) | 2004-05-10 | 2007-11-06 | Boston Scientific Scimed, Inc. | Clamp based low temperature lesion formation apparatus, systems and methods |
| US7861725B2 (en) | 2004-05-10 | 2011-01-04 | Boston Scientific Scimed, Inc. | Method for monitoring formation of a lesion in a tissue structure using a cryogenic system |
| US7288088B2 (en) * | 2004-05-10 | 2007-10-30 | Boston Scientific Scimed, Inc. | Clamp based low temperature lesion formation apparatus, systems and methods |
| US20050251122A1 (en) * | 2004-05-10 | 2005-11-10 | Scimed Life Systems, Inc. | Clamp based low temperature lesion formation apparatus, system and methods |
| US7291142B2 (en) | 2004-05-10 | 2007-11-06 | Boston Scientific Scimed, Inc. | Low temperature lesion formation apparatus, systems and methods |
| US20080058790A1 (en) * | 2004-05-10 | 2008-03-06 | Boston Scientific Scimed, Inc. | Clamp based low temperature lesion formation apparatus, systems and methods |
| US20050251121A1 (en) * | 2004-05-10 | 2005-11-10 | Scimed Life Systems, Inc. | Probe based low temperature lesion formation apparatus, systems and methods |
| US7972327B2 (en) | 2004-05-10 | 2011-07-05 | Boston Scientific Scimed, Inc. | Low temperature lesion formation apparatus, systems and methods |
| US8142424B2 (en) * | 2004-05-10 | 2012-03-27 | Boston Scientific Scimed, Inc. | Probe based low temperature lesion formation apparatus, systems and methods |
| US20060167444A1 (en) * | 2004-05-10 | 2006-07-27 | Swanson David K | Clamp Based Low Temperature Lesion Formation Apparatus, Systems And Methods |
| US8333764B2 (en) | 2004-05-12 | 2012-12-18 | Medtronic, Inc. | Device and method for determining tissue thickness and creating cardiac ablation lesions |
| US8801707B2 (en) | 2004-05-14 | 2014-08-12 | Medtronic, Inc. | Method and devices for treating atrial fibrillation by mass ablation |
| US8162941B2 (en) | 2004-06-02 | 2012-04-24 | Medtronic, Inc. | Ablation device with jaws |
| US7678108B2 (en) | 2004-06-02 | 2010-03-16 | Medtronic, Inc. | Loop ablation apparatus and method |
| US7758580B2 (en) | 2004-06-02 | 2010-07-20 | Medtronic, Inc. | Compound bipolar ablation device and method |
| US7758576B2 (en) | 2004-06-02 | 2010-07-20 | Medtronic, Inc. | Clamping ablation tool and method |
| US8172837B2 (en) | 2004-06-02 | 2012-05-08 | Medtronic, Inc. | Clamping ablation tool and method |
| US7566334B2 (en) | 2004-06-02 | 2009-07-28 | Medtronic, Inc. | Ablation device with jaws |
| US20090270857A1 (en) * | 2004-06-02 | 2009-10-29 | Christian Steven C | Ablation Device with Jaws |
| US7875028B2 (en) | 2004-06-02 | 2011-01-25 | Medtronic, Inc. | Ablation device with jaws |
| US8409219B2 (en) | 2004-06-18 | 2013-04-02 | Medtronic, Inc. | Method and system for placement of electrical lead inside heart |
| US9656063B2 (en) | 2004-06-18 | 2017-05-23 | Medtronic, Inc. | Method and system for placement of electrical lead inside heart |
| US8663245B2 (en) | 2004-06-18 | 2014-03-04 | Medtronic, Inc. | Device for occlusion of a left atrial appendage |
| US8926635B2 (en) | 2004-06-18 | 2015-01-06 | Medtronic, Inc. | Methods and devices for occlusion of an atrial appendage |
| US7731712B2 (en) | 2004-12-20 | 2010-06-08 | Cytyc Corporation | Method and system for transcervical tubal occlusion |
| US7674260B2 (en) | 2005-04-28 | 2010-03-09 | Cytyc Corporation | Emergency hemostasis device utilizing energy |
| US20080285668A1 (en) * | 2005-10-31 | 2008-11-20 | Lee Young-Dae | Method for Processing Control Information in a Wireless Mobile Communication System |
| US8702695B2 (en) | 2005-11-23 | 2014-04-22 | Covidien Lp | Auto-aligning ablating device and method of use |
| US7959627B2 (en) | 2005-11-23 | 2011-06-14 | Barrx Medical, Inc. | Precision ablating device |
| US9179970B2 (en) | 2005-11-23 | 2015-11-10 | Covidien Lp | Precision ablating method |
| US7997278B2 (en) | 2005-11-23 | 2011-08-16 | Barrx Medical, Inc. | Precision ablating method |
| US9918794B2 (en) | 2005-11-23 | 2018-03-20 | Covidien Lp | Auto-aligning ablating device and method of use |
| US9918793B2 (en) | 2005-11-23 | 2018-03-20 | Covidien Lp | Auto-aligning ablating device and method of use |
| US8702694B2 (en) | 2005-11-23 | 2014-04-22 | Covidien Lp | Auto-aligning ablating device and method of use |
| CN100402005C (en) * | 2005-12-22 | 2008-07-16 | 尹格平 | Multifunctional thermal coagulation therapeutic instrument for gynaecologic disease |
| US10589130B2 (en) | 2006-05-25 | 2020-03-17 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US9724119B2 (en) | 2006-05-25 | 2017-08-08 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US9227088B2 (en) | 2006-05-25 | 2016-01-05 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US9931134B2 (en) | 2006-05-25 | 2018-04-03 | Medtronic, Inc. | Methods of using high intensity focused ultrasound to form an ablated tissue area containing a plurality of lesions |
| US8486060B2 (en) | 2006-09-18 | 2013-07-16 | Cytyc Corporation | Power ramping during RF ablation |
| EP2081511A4 (en) * | 2006-10-16 | 2011-06-08 | Medtronic Cryocath Lp | METHOD AND APPARATUS FOR INFLATION AND DEFLATER OF BALLOON CATHETERS |
| WO2008046183A1 (en) | 2006-10-16 | 2008-04-24 | Cryocath Technologies Inc. | Method and apparatus for inflating and deflating balloon catheters |
| US7846160B2 (en) | 2006-12-21 | 2010-12-07 | Cytyc Corporation | Method and apparatus for sterilization |
| US9993281B2 (en) | 2007-05-04 | 2018-06-12 | Covidien Lp | Method and apparatus for gastrointestinal tract ablation for treatment of obesity |
| US8641711B2 (en) | 2007-05-04 | 2014-02-04 | Covidien Lp | Method and apparatus for gastrointestinal tract ablation for treatment of obesity |
| US8784338B2 (en) | 2007-06-22 | 2014-07-22 | Covidien Lp | Electrical means to normalize ablational energy transmission to a luminal tissue surface of varying size |
| US10575902B2 (en) | 2007-06-22 | 2020-03-03 | Covidien Lp | Electrical means to normalize ablational energy transmission to a luminal tissue surface of varying size |
| US9198713B2 (en) | 2007-06-22 | 2015-12-01 | Covidien Lp | Electrical means to normalize ablational energy transmission to a luminal tissue surface of varying size |
| US9364283B2 (en) | 2007-07-06 | 2016-06-14 | Covidien Lp | Method and apparatus for gastrointestinal tract ablation to achieve loss of persistent and/or recurrent excess body weight following a weight loss operation |
| US8251992B2 (en) | 2007-07-06 | 2012-08-28 | Tyco Healthcare Group Lp | Method and apparatus for gastrointestinal tract ablation to achieve loss of persistent and/or recurrent excess body weight following a weight-loss operation |
| US9839466B2 (en) | 2007-07-06 | 2017-12-12 | Covidien Lp | Method and apparatus for gastrointestinal tract ablation to achieve loss of persistent and/or recurrent excess body weight following a weight loss operation |
| US8439908B2 (en) | 2007-07-06 | 2013-05-14 | Covidien Lp | Ablation in the gastrointestinal tract to achieve hemostasis and eradicate lesions with a propensity for bleeding |
| US11207118B2 (en) | 2007-07-06 | 2021-12-28 | Tsunami Medtech, Llc | Medical system and method of use |
| US8646460B2 (en) | 2007-07-30 | 2014-02-11 | Covidien Lp | Cleaning device and methods |
| US8273012B2 (en) | 2007-07-30 | 2012-09-25 | Tyco Healthcare Group, Lp | Cleaning device and methods |
| US9314289B2 (en) | 2007-07-30 | 2016-04-19 | Covidien Lp | Cleaning device and methods |
| EP2522290A2 (en) | 2007-08-23 | 2012-11-14 | Aegea Medical, Inc. | Uterine therapy device |
| US10154871B2 (en) | 2007-08-23 | 2018-12-18 | Aegea Medical Inc. | Uterine therapy device and method |
| EP2198797A1 (en) | 2007-08-23 | 2010-06-23 | Aegea Medical, Inc. | Uterine therapy device |
| US20190117290A1 (en) * | 2007-08-23 | 2019-04-25 | Aegea Medical Inc. | Uterine therapy device and method |
| US8221401B2 (en) | 2007-08-23 | 2012-07-17 | Aegea Medical, Inc. | Uterine therapy device and method |
| US8221403B2 (en) | 2007-08-23 | 2012-07-17 | Aegea Medical, Inc. | Uterine therapy device and method |
| US8216217B2 (en) | 2007-08-23 | 2012-07-10 | Aegea Medical, Inc. | Uterine therapy device and method |
| US11213338B2 (en) * | 2007-08-23 | 2022-01-04 | Aegea Medical Inc. | Uterine therapy device and method |
| US10758292B2 (en) * | 2007-08-23 | 2020-09-01 | Aegea Medical Inc. | Uterine therapy device and method |
| US8197470B2 (en) | 2007-08-23 | 2012-06-12 | Aegea Medical, Inc. | Uterine therapy device and method |
| US20190117289A1 (en) * | 2007-08-23 | 2019-04-25 | Aegea Medical Inc. | Uterine therapy device and method |
| US8821488B2 (en) | 2008-05-13 | 2014-09-02 | Medtronic, Inc. | Tissue lesion evaluation |
| WO2010036434A1 (en) * | 2008-09-29 | 2010-04-01 | Boston Scientific Scimed, Inc. | Fluid recirculation debris handling system |
| US8790334B2 (en) | 2008-09-29 | 2014-07-29 | Boston Scientific Scimed, Inc. | Fluid recirculation debris handling system |
| US9561068B2 (en) | 2008-10-06 | 2017-02-07 | Virender K. Sharma | Method and apparatus for tissue ablation |
| US10064697B2 (en) | 2008-10-06 | 2018-09-04 | Santa Anna Tech Llc | Vapor based ablation system for treating various indications |
| US12310650B2 (en) | 2008-10-06 | 2025-05-27 | Santa Anna Tech Llc | Methods of ablating tissue using time-limited treatment periods |
| US11020175B2 (en) | 2008-10-06 | 2021-06-01 | Santa Anna Tech Llc | Methods of ablating tissue using time-limited treatment periods |
| US10695126B2 (en) | 2008-10-06 | 2020-06-30 | Santa Anna Tech Llc | Catheter with a double balloon structure to generate and apply a heated ablative zone to tissue |
| US10842548B2 (en) | 2008-10-06 | 2020-11-24 | Santa Anna Tech Llc | Vapor ablation system with a catheter having more than one positioning element |
| US9561066B2 (en) | 2008-10-06 | 2017-02-07 | Virender K. Sharma | Method and apparatus for tissue ablation |
| US9561067B2 (en) | 2008-10-06 | 2017-02-07 | Virender K. Sharma | Method and apparatus for tissue ablation |
| US10842557B2 (en) | 2008-10-06 | 2020-11-24 | Santa Anna Tech Llc | Vapor ablation system with a catheter having more than one positioning element and configured to treat duodenal tissue |
| US11813014B2 (en) | 2008-10-06 | 2023-11-14 | Santa Anna Tech Llc | Methods and systems for directed tissue ablation |
| US11589920B2 (en) | 2008-10-06 | 2023-02-28 | Santa Anna Tech Llc | Catheter with a double balloon structure to generate and apply an ablative zone to tissue |
| US10842549B2 (en) | 2008-10-06 | 2020-11-24 | Santa Anna Tech Llc | Vapor ablation system with a catheter having more than one positioning element and configured to treat pulmonary tissue |
| US11779430B2 (en) | 2008-10-06 | 2023-10-10 | Santa Anna Tech Llc | Vapor based ablation system for treating uterine bleeding |
| US9700365B2 (en) | 2008-10-06 | 2017-07-11 | Santa Anna Tech Llc | Method and apparatus for the ablation of gastrointestinal tissue |
| WO2010085460A1 (en) | 2009-01-20 | 2010-07-29 | Aegea Medical, Inc. | Uterine therapy device and method |
| CN102355864A (en) * | 2009-01-20 | 2012-02-15 | 埃杰亚医疗公司 | Uterine therapy device and method |
| US11284931B2 (en) | 2009-02-03 | 2022-03-29 | Tsunami Medtech, Llc | Medical systems and methods for ablating and absorbing tissue |
| US9486283B2 (en) | 2009-02-23 | 2016-11-08 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical device |
| US8632533B2 (en) | 2009-02-23 | 2014-01-21 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical device |
| US10660697B2 (en) | 2009-11-10 | 2020-05-26 | Cardea Medsystems (Tianjin) Co., Ltd. | Hollow body cavity ablation apparatus |
| US10758303B2 (en) | 2009-11-10 | 2020-09-01 | Cardea Medsystems (Tianjin) Co., Ltd. | Hollow body cavity ablation apparatus |
| US9283108B2 (en) | 2009-11-26 | 2016-03-15 | Hans I. Wallsten | Sterilisation device |
| US20140026395A1 (en) * | 2010-05-28 | 2014-01-30 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical devices, and methods of manufacture thereof |
| US9333027B2 (en) * | 2010-05-28 | 2016-05-10 | Medtronic Advanced Energy Llc | Method of producing an electrosurgical device |
| US9895191B2 (en) | 2010-06-28 | 2018-02-20 | Medtronic Advanced Energy Llc | Electrode sheath for electrosurgical device |
| US9138289B2 (en) | 2010-06-28 | 2015-09-22 | Medtronic Advanced Energy Llc | Electrode sheath for electrosurgical device |
| US8906012B2 (en) | 2010-06-30 | 2014-12-09 | Medtronic Advanced Energy Llc | Electrosurgical devices with wire electrode |
| US10004550B2 (en) | 2010-08-05 | 2018-06-26 | Medtronic Ardian Luxembourg S.A.R.L. | Cryoablation apparatuses, systems, and methods for renal neuromodulation |
| US11457969B2 (en) | 2010-08-13 | 2022-10-04 | Tsunami Medtech, Llc | Medical system and method of use |
| US8945107B2 (en) | 2010-10-26 | 2015-02-03 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US9060755B2 (en) | 2010-10-26 | 2015-06-23 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US9060754B2 (en) | 2010-10-26 | 2015-06-23 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US9066713B2 (en) | 2010-10-26 | 2015-06-30 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US10188445B2 (en) | 2010-10-26 | 2019-01-29 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US9439708B2 (en) | 2010-10-26 | 2016-09-13 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US10842547B2 (en) | 2010-10-26 | 2020-11-24 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US12251150B2 (en) | 2010-10-26 | 2025-03-18 | Medtronic Ardian Luxembourg S.A.R.L. | Neuromodulation cryotherapeutic devices and associated systems and methods |
| US12279802B2 (en) | 2010-11-09 | 2025-04-22 | Coopersurgical, Inc. | Positioning method and apparatus for delivering vapor to the uterus |
| US11160597B2 (en) | 2010-11-09 | 2021-11-02 | Aegea Medical Inc. | Positioning method and apparatus for delivering vapor to the uterus |
| US10238446B2 (en) | 2010-11-09 | 2019-03-26 | Aegea Medical Inc. | Positioning method and apparatus for delivering vapor to the uterus |
| US9743974B2 (en) | 2010-11-09 | 2017-08-29 | Aegea Medical Inc. | Positioning method and apparatus for delivering vapor to the uterus |
| US9498274B2 (en) | 2011-02-01 | 2016-11-22 | Channel Medsystems, Inc. | Liner extraction methods |
| US9510887B2 (en) | 2011-02-01 | 2016-12-06 | Channel Medsystems, Inc. | Time-limited methods for cryogenic treatment systems |
| US9445860B2 (en) | 2011-02-01 | 2016-09-20 | Channel Medsystems, Inc. | Handheld cyrogenic treatment systems |
| US8858543B2 (en) | 2011-02-01 | 2014-10-14 | Channel Medsystems, Inc. | Cyrogenic treatment systems |
| US9408657B2 (en) | 2011-02-01 | 2016-08-09 | Channel Medsystems, Inc. | Cryogenic treatment systems |
| US9283022B2 (en) | 2011-02-01 | 2016-03-15 | Channel Medsystems, Inc. | Methods and apparatus for cryogenic treatment of a body cavity or lumen |
| US9486267B2 (en) | 2011-02-01 | 2016-11-08 | Channel Medsystems, Inc. | Cryogenic treatment systems |
| US10213335B2 (en) | 2011-02-01 | 2019-02-26 | Channel Medsystems, Inc. | Methods and apparatus for cryogenic treatment of a body cavity or lumen |
| US9492217B2 (en) | 2011-02-01 | 2016-11-15 | Channel Medsystems, Inc. | Treatments using cryogenic ablation systems |
| US9492218B2 (en) | 2011-02-01 | 2016-11-15 | Channel Medsystems, Inc. | Pressure monitoring systems |
| US11883324B2 (en) * | 2011-02-01 | 2024-01-30 | Channel Medsystems, Inc. | Cryogenic treatment systems |
| US11833076B2 (en) | 2011-02-01 | 2023-12-05 | Channel Medsystems, Inc. | Methods and apparatus for cryogenic treatment of a body cavity or lumen |
| US9517100B2 (en) | 2011-02-01 | 2016-12-13 | Channel Medsystems, Inc. | Cryogenic treatment methods |
| US9277952B2 (en) | 2011-02-01 | 2016-03-08 | Channel Medsystems, Inc. | Cryogenic treatment systems |
| US10959879B2 (en) | 2011-02-01 | 2021-03-30 | Channel Medsystems, Inc. | Methods and apparatus for cryogenic treatment of a body cavity or lumen |
| US9603650B2 (en) | 2011-02-01 | 2017-03-28 | Channel Medsystems, Inc. | Cryogenic treatment systems |
| US9848933B2 (en) | 2011-02-01 | 2017-12-26 | Channel Medsystems, Inc. | Liner for cryogenic treatment systems |
| US10278774B2 (en) | 2011-03-18 | 2019-05-07 | Covidien Lp | Selectively expandable operative element support structure and methods of use |
| US10588682B2 (en) | 2011-04-25 | 2020-03-17 | Medtronic Ardian Luxembourg S.A.R.L. | Apparatus and methods related to constrained deployment of cryogenic balloons for limited cryogenic ablation of vessel walls |
| US9662060B2 (en) | 2011-10-07 | 2017-05-30 | Aegea Medical Inc. | Integrity testing method and apparatus for delivering vapor to the uterus |
| US10881442B2 (en) | 2011-10-07 | 2021-01-05 | Aegea Medical Inc. | Integrity testing method and apparatus for delivering vapor to the uterus |
| US10905490B2 (en) | 2012-04-27 | 2021-02-02 | Medtronic Ardian Luxembourg S.A.R.L. | Cryotherapeutic devices for renal neuromodulation and associated systems and methods |
| US11751931B2 (en) | 2012-04-27 | 2023-09-12 | Medtronic Ardian Luxembourg S.A.R.L. | Cryotherapeutic devices for renal neuromodulation and associated systems and methods |
| US9241752B2 (en) | 2012-04-27 | 2016-01-26 | Medtronic Ardian Luxembourg S.A.R.L. | Shafts with pressure relief in cryotherapeutic catheters and associated devices, systems, and methods |
| US9872718B2 (en) | 2012-04-27 | 2018-01-23 | Medtronic Adrian Luxembourg S.a.r.l. | Shafts with pressure relief in cryotherapeutic catheters and associated devices, systems, and methods |
| US9095321B2 (en) | 2012-11-21 | 2015-08-04 | Medtronic Ardian Luxembourg S.A.R.L. | Cryotherapeutic devices having integral multi-helical balloons and methods of making the same |
| US9017317B2 (en) | 2012-12-06 | 2015-04-28 | Medtronic Ardian Luxembourg S.A.R.L. | Refrigerant supply system for cryotherapy including refrigerant recompression and associated devices, systems, and methods |
| US11413086B2 (en) | 2013-03-15 | 2022-08-16 | Tsunami Medtech, Llc | Medical system and method of use |
| US11672584B2 (en) | 2013-03-15 | 2023-06-13 | Tsunami Medtech, Llc | Medical system and method of use |
| US12114909B2 (en) | 2013-03-15 | 2024-10-15 | Tsunami Medtech, Llc | Medical system and method of use |
| US11607263B2 (en) | 2013-09-17 | 2023-03-21 | Channel Medsystems, Inc. | Liner for cryogenic treatment systems |
| US12256971B2 (en) | 2013-09-17 | 2025-03-25 | Channel Medsystems, Inc. | Liner for cryogenic treatment systems |
| US9895183B2 (en) | 2013-09-17 | 2018-02-20 | Channel Medsystems, Inc. | Liner for cryogenic treatment systems |
| US10849672B2 (en) | 2013-09-17 | 2020-12-01 | Channel Medsystems, Inc. | Liner for cryogenic treatment systems |
| US11241272B2 (en) | 2013-09-30 | 2022-02-08 | Covidien Lp | Bipolar electrosurgical instrument with movable electrode and related systems and methods |
| US10631914B2 (en) | 2013-09-30 | 2020-04-28 | Covidien Lp | Bipolar electrosurgical instrument with movable electrode and related systems and methods |
| US12357369B2 (en) | 2013-09-30 | 2025-07-15 | Covidien Lp | Bipolar electrosurgical instrument with movable electrode and related systems and methods |
| US12274483B2 (en) | 2014-03-07 | 2025-04-15 | Medtronic Ardian Luxembourg S.A.R.L. | Monitoring and controlling internally administered cryotherapy |
| US10492842B2 (en) | 2014-03-07 | 2019-12-03 | Medtronic Ardian Luxembourg S.A.R.L. | Monitoring and controlling internally administered cryotherapy |
| US11406437B2 (en) | 2014-03-07 | 2022-08-09 | Medtronic Ardian Luxembourg S.A.R.L. | Monitoring and controlling internally administered cryotherapy |
| US11793561B2 (en) | 2014-04-10 | 2023-10-24 | Channel Medsystems, Inc. | Apparatus and methods for regulating cryogenic treatment |
| US10610279B2 (en) | 2014-04-10 | 2020-04-07 | Channel Medsystems, Inc. | Apparatus and methods for regulating cryogenic treatment |
| US10179019B2 (en) | 2014-05-22 | 2019-01-15 | Aegea Medical Inc. | Integrity testing method and apparatus for delivering vapor to the uterus |
| US10575898B2 (en) | 2014-05-22 | 2020-03-03 | Aegea Medical Inc. | Systems and methods for performing endometrial ablation |
| US11219479B2 (en) | 2014-05-22 | 2022-01-11 | Aegea Medical Inc. | Integrity testing method and apparatus for delivering vapor to the uterus |
| US9993290B2 (en) | 2014-05-22 | 2018-06-12 | Aegea Medical Inc. | Systems and methods for performing endometrial ablation |
| US10299856B2 (en) | 2014-05-22 | 2019-05-28 | Aegea Medical Inc. | Systems and methods for performing endometrial ablation |
| US9974599B2 (en) | 2014-08-15 | 2018-05-22 | Medtronic Ps Medical, Inc. | Multipurpose electrosurgical device |
| US9956029B2 (en) | 2014-10-31 | 2018-05-01 | Medtronic Advanced Energy Llc | Telescoping device with saline irrigation line |
| US12011283B2 (en) | 2016-02-19 | 2024-06-18 | Aegea Medical Inc. | Methods and apparatus for determining the integrity of a bodily cavity |
| US11331037B2 (en) | 2016-02-19 | 2022-05-17 | Aegea Medical Inc. | Methods and apparatus for determining the integrity of a bodily cavity |
| US12364537B2 (en) | 2016-05-02 | 2025-07-22 | Santa Anna Tech Llc | Catheter with a double balloon structure to generate and apply a heated ablative zone to tissue |
| US12137969B2 (en) | 2016-05-19 | 2024-11-12 | Aqua Heart, Inc. | Heated vapor ablation systems and methods for treating cardiac conditions |
| US11331140B2 (en) | 2016-05-19 | 2022-05-17 | Aqua Heart, Inc. | Heated vapor ablation systems and methods for treating cardiac conditions |
| US20190365453A1 (en) * | 2017-02-17 | 2019-12-05 | Boston Scientific Scimed, Inc. | System and method for limiting differential pressure across proportional valve during cryoablation procedures |
| US20190083298A1 (en) * | 2017-09-21 | 2019-03-21 | Michael W. Starkweather | Whole body cryotherapy system |
| US11806066B2 (en) | 2018-06-01 | 2023-11-07 | Santa Anna Tech Llc | Multi-stage vapor-based ablation treatment methods and vapor generation and delivery systems |
| US11864809B2 (en) | 2018-06-01 | 2024-01-09 | Santa Anna Tech Llc | Vapor-based ablation treatment methods with improved treatment volume vapor management |
| US12279803B2 (en) | 2018-06-01 | 2025-04-22 | Aqua Medical, Inc. | Vapor-based ablation treatment methods with improved treatment volume vapor management |
| US12458425B2 (en) | 2022-07-12 | 2025-11-04 | Covidien Lp | Thermal heating element for use with endometrial ablation |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US3924628A (en) | Cyrogenic bladder for necrosing tissue cells | |
| US6432102B2 (en) | Cryosurgical fluid supply | |
| US9402676B2 (en) | Cryoablation balloon catheter and related method | |
| AU2004308416B2 (en) | Cryosurgical devices and methods for endometrial ablation | |
| US5403309A (en) | Cryosurgical ablation | |
| CA2736221C (en) | A cryogenic system and method of use | |
| US4082096A (en) | Cryosurgical system | |
| US20180028251A1 (en) | Regulating pressure to lower temperature in a cyrotherapy balloon catheter | |
| JP2004512075A (en) | Multiple cryoprobe device and method | |
| US3220414A (en) | Surgical cannula | |
| JP2020503977A (en) | Apparatus for tissue surface ablation and related systems and methods | |
| WO1994010948A1 (en) | Heated balloon medical apparatus | |
| US20210153920A1 (en) | Apparatus and method for ablation of soft tissue surrounding a breast cavity following lumpectomy | |
| CA1319072C (en) | Intrauterine cauterizing apparatus and method | |
| HK40011659A (en) | Device for ablation of tissue surfaces and related systems and methods |