US12224452B2 - Nonaqueous electrolyte secondary battery including separator with filler layers on opposite sides of substrate - Google Patents
Nonaqueous electrolyte secondary battery including separator with filler layers on opposite sides of substrate Download PDFInfo
- Publication number
- US12224452B2 US12224452B2 US16/637,034 US201816637034A US12224452B2 US 12224452 B2 US12224452 B2 US 12224452B2 US 201816637034 A US201816637034 A US 201816637034A US 12224452 B2 US12224452 B2 US 12224452B2
- Authority
- US
- United States
- Prior art keywords
- substrate
- filler layer
- positive electrode
- separator
- phosphate salt
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active, expires
Links
- 239000000945 filler Substances 0.000 title claims abstract description 101
- 239000000758 substrate Substances 0.000 title claims abstract description 82
- 239000011255 nonaqueous electrolyte Substances 0.000 title claims abstract description 28
- 239000002245 particle Substances 0.000 claims abstract description 79
- 239000010954 inorganic particle Substances 0.000 claims abstract description 19
- 150000003013 phosphoric acid derivatives Chemical class 0.000 claims description 46
- 229920005989 resin Polymers 0.000 claims description 39
- 239000011347 resin Substances 0.000 claims description 39
- 239000011148 porous material Substances 0.000 claims description 19
- 229910001386 lithium phosphate Inorganic materials 0.000 claims description 17
- TWQULNDIKKJZPH-UHFFFAOYSA-K trilithium;phosphate Chemical compound [Li+].[Li+].[Li+].[O-]P([O-])([O-])=O TWQULNDIKKJZPH-UHFFFAOYSA-K 0.000 claims description 17
- 238000002844 melting Methods 0.000 claims description 10
- 230000008018 melting Effects 0.000 claims description 10
- 229920000098 polyolefin Polymers 0.000 claims description 6
- SNKMVYBWZDHJHE-UHFFFAOYSA-M lithium;dihydrogen phosphate Chemical compound [Li+].OP(O)([O-])=O SNKMVYBWZDHJHE-UHFFFAOYSA-M 0.000 claims description 4
- REKWWOFUJAJBCL-UHFFFAOYSA-L dilithium;hydrogen phosphate Chemical compound [Li+].[Li+].OP([O-])([O-])=O REKWWOFUJAJBCL-UHFFFAOYSA-L 0.000 claims description 3
- 238000006068 polycondensation reaction Methods 0.000 claims description 3
- 125000002467 phosphate group Chemical class [H]OP(=O)(O[H])O[*] 0.000 abstract 1
- 239000010410 layer Substances 0.000 description 133
- 239000000203 mixture Substances 0.000 description 38
- 230000002159 abnormal effect Effects 0.000 description 19
- 239000011230 binding agent Substances 0.000 description 16
- 239000002002 slurry Substances 0.000 description 15
- 229910001593 boehmite Inorganic materials 0.000 description 14
- 238000004519 manufacturing process Methods 0.000 description 14
- 229910052751 metal Inorganic materials 0.000 description 14
- -1 polytetrafluoroethylene Polymers 0.000 description 14
- 239000002184 metal Substances 0.000 description 13
- 239000010408 film Substances 0.000 description 12
- 230000020169 heat generation Effects 0.000 description 12
- 150000002500 ions Chemical class 0.000 description 11
- 238000009782 nail-penetration test Methods 0.000 description 11
- 230000000052 comparative effect Effects 0.000 description 10
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 10
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 8
- 238000001035 drying Methods 0.000 description 8
- FAHBNUUHRFUEAI-UHFFFAOYSA-M hydroxidooxidoaluminium Chemical compound O[Al]=O FAHBNUUHRFUEAI-UHFFFAOYSA-M 0.000 description 8
- 238000000034 method Methods 0.000 description 8
- 239000004698 Polyethylene Substances 0.000 description 7
- 230000005856 abnormality Effects 0.000 description 7
- 239000002253 acid Substances 0.000 description 7
- 229920001577 copolymer Polymers 0.000 description 7
- 229920000573 polyethylene Polymers 0.000 description 7
- 150000003839 salts Chemical class 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- 239000002033 PVDF binder Substances 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 6
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 5
- 229910052782 aluminium Inorganic materials 0.000 description 5
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 5
- 238000000576 coating method Methods 0.000 description 5
- 229910001416 lithium ion Inorganic materials 0.000 description 5
- 229920002239 polyacrylonitrile Polymers 0.000 description 5
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 5
- 239000004810 polytetrafluoroethylene Substances 0.000 description 5
- 239000007774 positive electrode material Substances 0.000 description 5
- 229920000178 Acrylic resin Polymers 0.000 description 4
- 239000004925 Acrylic resin Substances 0.000 description 4
- 239000004372 Polyvinyl alcohol Substances 0.000 description 4
- 239000002131 composite material Substances 0.000 description 4
- 239000006258 conductive agent Substances 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 229910021437 lithium-transition metal oxide Inorganic materials 0.000 description 4
- 239000011572 manganese Substances 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 229910044991 metal oxide Inorganic materials 0.000 description 4
- 239000007773 negative electrode material Substances 0.000 description 4
- 229910052759 nickel Inorganic materials 0.000 description 4
- 229920002451 polyvinyl alcohol Polymers 0.000 description 4
- 229940068984 polyvinyl alcohol Drugs 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 229920003048 styrene butadiene rubber Polymers 0.000 description 4
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 3
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- 229920001145 Poly(N-vinylacetamide) Polymers 0.000 description 3
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 239000011888 foil Substances 0.000 description 3
- 150000004678 hydrides Chemical class 0.000 description 3
- SWAIALBIBWIKKQ-UHFFFAOYSA-N lithium titanium Chemical compound [Li].[Ti] SWAIALBIBWIKKQ-UHFFFAOYSA-N 0.000 description 3
- 239000011777 magnesium Substances 0.000 description 3
- 229910052748 manganese Inorganic materials 0.000 description 3
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 3
- 229910052623 talc Inorganic materials 0.000 description 3
- 229910052718 tin Inorganic materials 0.000 description 3
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- 229910001290 LiPF6 Inorganic materials 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- 239000004642 Polyimide Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- 229910021383 artificial graphite Inorganic materials 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 229910002113 barium titanate Inorganic materials 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 239000006229 carbon black Substances 0.000 description 2
- 239000003575 carbonaceous material Substances 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229940105329 carboxymethylcellulose Drugs 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 229910052593 corundum Inorganic materials 0.000 description 2
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical class O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 2
- 238000007599 discharging Methods 0.000 description 2
- 239000002612 dispersion medium Substances 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 239000003792 electrolyte Substances 0.000 description 2
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 2
- 238000007756 gravure coating Methods 0.000 description 2
- 239000011964 heteropoly acid Substances 0.000 description 2
- 238000007731 hot pressing Methods 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 150000002484 inorganic compounds Chemical class 0.000 description 2
- 229910010272 inorganic material Inorganic materials 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 2
- 150000001247 metal acetylides Chemical class 0.000 description 2
- 229910000000 metal hydroxide Inorganic materials 0.000 description 2
- 150000004692 metal hydroxides Chemical class 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- 229910052976 metal sulfide Inorganic materials 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 239000012046 mixed solvent Substances 0.000 description 2
- 239000010955 niobium Substances 0.000 description 2
- 150000004767 nitrides Chemical class 0.000 description 2
- 238000012856 packing Methods 0.000 description 2
- 230000035515 penetration Effects 0.000 description 2
- 229920000728 polyester Polymers 0.000 description 2
- 229920001721 polyimide Polymers 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 229920002620 polyvinyl fluoride Polymers 0.000 description 2
- 238000005096 rolling process Methods 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 229910052814 silicon oxide Inorganic materials 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- 238000003466 welding Methods 0.000 description 2
- 229910001845 yogo sapphire Inorganic materials 0.000 description 2
- 239000004711 α-olefin Substances 0.000 description 2
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- 229910002706 AlOOH Inorganic materials 0.000 description 1
- 229910052580 B4C Inorganic materials 0.000 description 1
- 238000004438 BET method Methods 0.000 description 1
- 229910052582 BN Inorganic materials 0.000 description 1
- PZNSFCLAULLKQX-UHFFFAOYSA-N Boron nitride Chemical compound N#B PZNSFCLAULLKQX-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- GYHNNYVSQQEPJS-UHFFFAOYSA-N Gallium Chemical compound [Ga] GYHNNYVSQQEPJS-UHFFFAOYSA-N 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 1
- 239000007836 KH2PO4 Substances 0.000 description 1
- 229910005699 Li1.05Ni0.82Co0.15Al0.03O2 Inorganic materials 0.000 description 1
- 229910012305 LiPON Inorganic materials 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical class OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229920002319 Poly(methyl acrylate) Polymers 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004697 Polyetherimide Substances 0.000 description 1
- 239000004734 Polyphenylene sulfide Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 239000002174 Styrene-butadiene Substances 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- NRTOMJZYCJJWKI-UHFFFAOYSA-N Titanium nitride Chemical compound [Ti]#N NRTOMJZYCJJWKI-UHFFFAOYSA-N 0.000 description 1
- 229910021536 Zeolite Inorganic materials 0.000 description 1
- 229910008243 Zr3(PO4)4 Inorganic materials 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000006230 acetylene black Substances 0.000 description 1
- 229940117913 acrylamide Drugs 0.000 description 1
- XECAHXYUAAWDEL-UHFFFAOYSA-N acrylonitrile butadiene styrene Chemical compound C=CC=C.C=CC#N.C=CC1=CC=CC=C1 XECAHXYUAAWDEL-UHFFFAOYSA-N 0.000 description 1
- 239000004676 acrylonitrile butadiene styrene Substances 0.000 description 1
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- INAHAJYZKVIDIZ-UHFFFAOYSA-N boron carbide Chemical compound B12B3B4C32B41 INAHAJYZKVIDIZ-UHFFFAOYSA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- FUFJGUQYACFECW-UHFFFAOYSA-L calcium hydrogenphosphate Chemical compound [Ca+2].OP([O-])([O-])=O FUFJGUQYACFECW-UHFFFAOYSA-L 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 239000002134 carbon nanofiber Substances 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 229920003123 carboxymethyl cellulose sodium Polymers 0.000 description 1
- 229940063834 carboxymethylcellulose sodium Drugs 0.000 description 1
- 235000010418 carrageenan Nutrition 0.000 description 1
- 239000000679 carrageenan Substances 0.000 description 1
- 229920001525 carrageenan Polymers 0.000 description 1
- 229940113118 carrageenan Drugs 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011247 coating layer Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 239000011889 copper foil Substances 0.000 description 1
- PMHQVHHXPFUNSP-UHFFFAOYSA-M copper(1+);methylsulfanylmethane;bromide Chemical compound Br[Cu].CSC PMHQVHHXPFUNSP-UHFFFAOYSA-M 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- MHJAJDCZWVHCPF-UHFFFAOYSA-L dimagnesium phosphate Chemical compound [Mg+2].OP([O-])([O-])=O MHJAJDCZWVHCPF-UHFFFAOYSA-L 0.000 description 1
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 1
- ZPWVASYFFYYZEW-UHFFFAOYSA-L dipotassium hydrogen phosphate Chemical compound [K+].[K+].OP([O-])([O-])=O ZPWVASYFFYYZEW-UHFFFAOYSA-L 0.000 description 1
- 229910000396 dipotassium phosphate Inorganic materials 0.000 description 1
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 1
- 229910000397 disodium phosphate Inorganic materials 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000002573 ethenylidene group Chemical group [*]=C=C([H])[H] 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 229920000840 ethylene tetrafluoroethylene copolymer Polymers 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 229920001973 fluoroelastomer Polymers 0.000 description 1
- 229910052733 gallium Inorganic materials 0.000 description 1
- 229910052732 germanium Inorganic materials 0.000 description 1
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 229910021389 graphene Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000010220 ion permeability Effects 0.000 description 1
- 229920003049 isoprene rubber Polymers 0.000 description 1
- 239000003273 ketjen black Substances 0.000 description 1
- 238000007561 laser diffraction method Methods 0.000 description 1
- 239000011244 liquid electrolyte Substances 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical class C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 229960002900 methylcellulose Drugs 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 1
- 229910021382 natural graphite Inorganic materials 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- SIWVEOZUMHYXCS-UHFFFAOYSA-N oxo(oxoyttriooxy)yttrium Chemical compound O=[Y]O[Y]=O SIWVEOZUMHYXCS-UHFFFAOYSA-N 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- DHRLEVQXOMLTIM-UHFFFAOYSA-N phosphoric acid;trioxomolybdenum Chemical compound O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.O=[Mo](=O)=O.OP(O)(O)=O DHRLEVQXOMLTIM-UHFFFAOYSA-N 0.000 description 1
- IYDGMDWEHDFVQI-UHFFFAOYSA-N phosphoric acid;trioxotungsten Chemical compound O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.O=[W](=O)=O.OP(O)(O)=O IYDGMDWEHDFVQI-UHFFFAOYSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920001601 polyetherimide Polymers 0.000 description 1
- 229920000120 polyethyl acrylate Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920001955 polyphenylene ether Polymers 0.000 description 1
- 229920000069 polyphenylene sulfide Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- GNSKLFRGEWLPPA-UHFFFAOYSA-M potassium dihydrogen phosphate Chemical compound [K+].OP(O)([O-])=O GNSKLFRGEWLPPA-UHFFFAOYSA-M 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 229910052706 scandium Inorganic materials 0.000 description 1
- SIXSYDAISGFNSX-UHFFFAOYSA-N scandium atom Chemical compound [Sc] SIXSYDAISGFNSX-UHFFFAOYSA-N 0.000 description 1
- 239000011163 secondary particle Substances 0.000 description 1
- VSZWPYCFIRKVQL-UHFFFAOYSA-N selanylidenegallium;selenium Chemical compound [Se].[Se]=[Ga].[Se]=[Ga] VSZWPYCFIRKVQL-UHFFFAOYSA-N 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- CGFYHILWFSGVJS-UHFFFAOYSA-N silicic acid;trioxotungsten Chemical compound O[Si](O)(O)O.O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1.O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1.O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1.O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 CGFYHILWFSGVJS-UHFFFAOYSA-N 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 239000007784 solid electrolyte Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- VEALVRVVWBQVSL-UHFFFAOYSA-N strontium titanate Chemical compound [Sr+2].[O-][Ti]([O-])=O VEALVRVVWBQVSL-UHFFFAOYSA-N 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- JBQYATWDVHIOAR-UHFFFAOYSA-N tellanylidenegermanium Chemical compound [Te]=[Ge] JBQYATWDVHIOAR-UHFFFAOYSA-N 0.000 description 1
- 238000005979 thermal decomposition reaction Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 229910000391 tricalcium phosphate Inorganic materials 0.000 description 1
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 1
- 229910000404 tripotassium phosphate Inorganic materials 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 229910000406 trisodium phosphate Inorganic materials 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
- 229910052727 yttrium Inorganic materials 0.000 description 1
- VWQVUPCCIRVNHF-UHFFFAOYSA-N yttrium atom Chemical compound [Y] VWQVUPCCIRVNHF-UHFFFAOYSA-N 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- UHVMMEOXYDMDKI-JKYCWFKZSA-L zinc;1-(5-cyanopyridin-2-yl)-3-[(1s,2s)-2-(6-fluoro-2-hydroxy-3-propanoylphenyl)cyclopropyl]urea;diacetate Chemical compound [Zn+2].CC([O-])=O.CC([O-])=O.CCC(=O)C1=CC=C(F)C([C@H]2[C@H](C2)NC(=O)NC=2N=CC(=CC=2)C#N)=C1O UHVMMEOXYDMDKI-JKYCWFKZSA-L 0.000 description 1
- 229910001928 zirconium oxide Inorganic materials 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/46—Separators, membranes or diaphragms characterised by their combination with electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/04—Construction or manufacture in general
- H01M10/0431—Cells with wound or folded electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/058—Construction or manufacture
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/64—Carriers or collectors
- H01M4/66—Selection of materials
- H01M4/668—Composites of electroconductive material and synthetic resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/411—Organic material
- H01M50/414—Synthetic resins, e.g. thermoplastics or thermosetting resins
- H01M50/417—Polyolefins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/411—Organic material
- H01M50/414—Synthetic resins, e.g. thermoplastics or thermosetting resins
- H01M50/423—Polyamide resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/446—Composite material consisting of a mixture of organic and inorganic materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/449—Separators, membranes or diaphragms characterised by the material having a layered structure
- H01M50/451—Separators, membranes or diaphragms characterised by the material having a layered structure comprising layers of only organic material and layers containing inorganic material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/449—Separators, membranes or diaphragms characterised by the material having a layered structure
- H01M50/457—Separators, membranes or diaphragms characterised by the material having a layered structure comprising three or more layers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/46—Separators, membranes or diaphragms characterised by their combination with electrodes
- H01M50/461—Separators, membranes or diaphragms characterised by their combination with electrodes with adhesive layers between electrodes and separators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/058—Construction or manufacture
- H01M10/0587—Construction or manufacture of accumulators having only wound construction elements, i.e. wound positive electrodes, wound negative electrodes and wound separators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/028—Positive electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/40—Separators; Membranes; Diaphragms; Spacing elements inside cells
- H01M50/409—Separators, membranes or diaphragms characterised by the material
- H01M50/44—Fibrous material
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present disclosure relates to nonaqueous electrolyte secondary batteries.
- Nonaqueous electrolyte secondary batteries such as lithium ion batteries can generate abnormal heat in the event of abnormalities such as overcharging, internal short-circuits, external short-circuits, and excessive resistance heating stemming from the passage of large currents.
- the shutdown function of separators is known as one of the conventional techniques for suppressing heat generation from nonaqueous electrolyte secondary batteries. According to the shutdown function, a separator is melted by abnormal heat generation of the battery to close pores in the separator and thereby to interrupt ion conduction between the positive and negative electrodes, thus suppressing further generation of heat from the battery.
- Known separators for nonaqueous electrolyte secondary batteries are such that a layer containing inorganic particles such as aluminum oxide or boehmite is disposed on the surface of a substrate having the shutdown function (see, for example, PTL 1).
- PTL 1 describes that the use of a separator which satisfies the specified structure can reduce an increase in internal resistance of a battery and achieves enhancements both in capacity and output.
- an important challenge lies in that when abnormal heat is generated in a battery, the ion conduction between the positive and negative electrodes is interrupted to suppress the heat generation in the battery.
- the conventional techniques including the technique of PTL 1 are sometimes incapable of sufficiently interrupting the ion conduction between positive and negative electrodes.
- An object of the present disclosure is that the ion conduction between positive and negative electrodes is sufficiently interrupted in the event of occurrence of abnormal heat in a battery, and thereby further generation of heat from the battery is suppressed.
- a nonaqueous electrolyte secondary battery includes a positive electrode, a negative electrode and a separator, wherein the separator includes a substrate, a first filler layer disposed on one side of the substrate and containing phosphate salt particles, and a second filler layer disposed on the other side of the substrate and containing inorganic particles with a higher melting point than the phosphate salt particles, the separator being disposed between the positive electrode and the negative electrode in such a manner that the first filler layer is directed to the positive electrode side, and the BET specific surface area of the phosphate salt particles is not less than 5 m 2 /g and not more than 100 m 2 /g.
- the nonaqueous electrolyte secondary battery according to one aspect of the present disclosure can benefit from sufficient interruption of ion conduction between the positive and negative electrodes in the event of occurrence of abnormal heat in the battery, and can be prevented from further generation of heat from the battery.
- FIG. 1 is a sectional view of a nonaqueous electrolyte secondary battery according to an example embodiment.
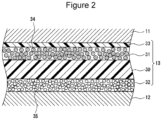
- FIG. 2 is a sectional view of an electrode assembly according to an example embodiment.
- porous resin substrates have the shutdown function.
- the resin substrate executes the shutdown function to interrupt communications such as ion conduction between the positive and negative electrodes, thus suppressing further generation of heat from the battery.
- the inside of the batteries becomes very hot (for example, 200° C. or above) in the event of occurrence of an abnormality, and the separator sometimes cannot retain its shape.
- the filler layer described above has no shutdown function unlike the resin substrate, a significant thermal deformation of the separator to such an extent that the separator shape cannot be maintained results in a failure to interrupt sufficiently the communications such as ion conduction between the positive and negative electrodes even though the filler layer is present, and consequently the generation of heat from the battery cannot sometimes be reduced to a sufficient extent.
- ion conduction between positive and negative electrodes can be sufficiently interrupted in the event of occurrence of abnormal heat in a battery by providing a first filler layer containing phosphate salt particles on one side of a separator substrate, and a second filler layer containing inorganic particles with a higher melting point than the phosphate salt particles on the other side of the substrate in such a manner that the first filler layer is directed to the positive electrode side.
- a nonaqueous electrolyte secondary battery according to the present disclosure can suppress further generation of heat from the battery after the occurrence of abnormal heat in the battery.
- the phosphate salt particles contained in the first filler layer are melted and polymerized in an accelerated manner by heat and the potential of the positive electrode to fill pores in the substrate.
- the second filler layer ensures the shape stability of the separator and allows the substrate to attain an enhanced shutdown effect.
- the separator is such that a layer including a mixture of phosphate salt particles and inorganic particles is disposed on the substrate surface, the phosphate salt cannot fully exhibit its effect due to the area of contact between the phosphate salt and the substrate being small. Further, studies by the present inventors have found that the use of the separator according to the present disclosure which has a layered structure represented by first filler layer/substrate/second filler layer offers a higher effect in suppressing heat generation than when use is made of a separator having a layered structure represented by first filler layer/second filler layer/substrate.
- heat generation in a battery is also accelerated when a combustible or combustion-supporting gas (such as oxygen or hydrogen) is generated from, for example, one of the electrodes due to a temperature rise within the battery stemming from internal short-circuiting of the battery and the gas migrates to and reacts with the other electrode.
- a combustible or combustion-supporting gas such as oxygen or hydrogen
- the migration of such gas too can be sufficiently interrupted.
- the structure of the electrode assembly is not limited to such a coil, and may be a stack of a plurality of positive electrodes and a plurality of negative electrodes stacked alternately on top of one another via separators.
- the shape of the battery case is not limited to cylindrical, and may be, for example, a metallic case such as a prismatic case (a prismatic battery) or a coin shape (a coin-shaped battery), or a resin case (a laminate battery) composed of resin films.
- numerical ranges written as value (A) to value (B) mean that the numerical quantities of interest are not less than value (A) and not more than value (B) unless otherwise mentioned.
- FIG. 1 is a sectional view of a nonaqueous electrolyte secondary battery 10 according to an example embodiment.
- the nonaqueous electrolyte secondary battery 10 includes an electrode assembly 14 , a nonaqueous electrolyte (not shown), and a battery case 15 in which the electrode assembly 14 and the nonaqueous electrolyte are accommodated.
- the electrode assembly 14 includes a positive electrode 11 , a negative electrode 12 and a separator 13 , the positive electrode 11 and the negative electrode 12 being wound together via the separator 13 to form a wound structure.
- the battery case 15 is composed of a bottomed cylindrical exterior case 16 , and a seal body 17 which seals the opening of the exterior case 16 .
- the nonaqueous electrolyte includes a nonaqueous solvent and an electrolyte salt dissolved in the nonaqueous solvent.
- the nonaqueous solvents which may be used include esters, ethers, nitriles, amides, and mixtures of two or more kinds of these solvents.
- the nonaqueous solvent may include a halogenated solvent resulting from the substitution of the above solvent with a halogen atom such as fluorine in place of at least part of hydrogen.
- the nonaqueous electrolyte is not limited to a liquid electrolyte, and may be a solid electrolyte such as a gel polymer.
- a lithium salt such as LiPF 6 is used as the electrolyte salt.
- the nonaqueous electrolyte secondary battery 10 includes insulating plates 18 , 19 disposed on and under the electrode assembly 14 , respectively.
- a positive electrode lead 20 attached to the positive electrode 11 extends toward the seal body 17 through a through-hole in the insulating plate 18
- a negative electrode lead 21 attached to the negative electrode 12 extends along the outside of the insulating plate 19 to the bottom of the exterior case 16 .
- the positive electrode lead 20 is connected by welding or the like to the lower side of a filter 23 that is a bottom plate of the seal body 17 .
- a cap 27 that is a top plate of the seal body 17 and is electrically connected to the filter 23 serves as a positive electrode terminal.
- the negative electrode lead 21 is connected by welding or the like to the inner side of the bottom of the exterior case 16 , thus allowing the exterior case 16 to serve as a negative electrode terminal.
- the exterior case 16 is a bottomed cylindrical metallic case.
- a gasket 28 is disposed between the exterior case 16 and the seal body 17 , thus ensuring tightness in the inside of the battery.
- the exterior case 16 has an overhang 22 which is defined by, for example, a lateral portion projecting toward the inside and which supports the seal body 17 .
- the overhang 22 is preferably an annular portion which extends along the circumference of the exterior case 16 , and supports the seal body 17 on the upper side thereof.
- the seal body 17 has a structure in which the filter 23 , a lower valve 24 , an insulating member 25 , an upper valve 26 and the cap 27 are stacked in this order from the side adjacent to the electrode assembly 14 .
- the members constituting the seal body 17 have a disk shape or a ring shape, and the members except the insulating member 25 are electrically connected to one another.
- the lower valve 24 and the upper valve 26 are connected to each other in the respective central portions, and the insulating member 25 is interposed between peripheral portions of the valves.
- the lower valve 24 is deformed so as to lift the upper valve 26 toward the cap 27 and ruptures to interrupt the current path between the lower valve 24 and the upper valve 26 . If the inner pressure is further increased, the upper valve 26 is ruptured and allows the gas to be discharged through the opening in the cap 27 .
- FIG. 2 is a sectional view of the electrode assembly 14 and, in particular, illustrates a portion of a cross section of the separator 13 with magnification.
- the positive electrode 11 includes a positive electrode current collector and a positive electrode mixture layer disposed on the current collector.
- the positive electrode current collector may be, for example, a foil of a metal that is stable at the potentials of the positive electrode 11 , such as aluminum, or a film having such a metal as a skin layer.
- the positive electrode mixture layer includes a positive electrode active material, a conductive agent and a binder, and is preferably disposed on both sides of the positive electrode current collector.
- the positive electrode 11 may be fabricated by applying a positive electrode mixture slurry including components such as a positive electrode active material, a conductive agent and a binder onto a positive electrode current collector, drying the wet films, and rolling the coatings to form positive electrode mixture layers on both sides of the positive electrode current collector.
- the positive electrode active materials include lithium transition metal oxides containing transition metal elements such as Co, Mn and Ni.
- the lithium transition metal oxide is preferably a Ni-containing lithium transition metal oxide which contains nickel (Ni) and has a Ni proportion of not less than 30 mol % relative to the total number of moles of the metal element(s) except lithium (Li).
- the Ni-containing lithium transition metal oxides include oxides represented by the compositional formula Li a Ni x M (1-x) O 2 (0.95 ⁇ a ⁇ 1.2, 0.3 ⁇ x ⁇ 1.0, and M denotes a metal element other than Li and Ni).
- the metal M in the formula may include, for example, at least one selected from cobalt (Co), manganese (Mn), tungsten (W), niobium (Nb), molybdenum (Mo), phosphorus (P) and sulfur (S).
- the oxide may further contain at least one selected from magnesium (Mg), aluminum (Al), calcium (Ca), scandium (Sc), titanium (Ti), vanadium (V), chromium (Cr), iron (Fe), copper (Cu), zinc (Zn), gallium (Ga), germanium (Ge), yttrium (Y), zirconium (Zr), tin (Sn), antimony (Sb), lead (Pb) and bismuth (Bi).
- Co, Mn, etc. are preferable.
- Examples of the conductive agents include carbon materials such as carbon black, acetylene black, Ketjen black, graphite, carbon nanotubes, carbon nanofibers and graphene.
- Examples of the binders include fluororesins such as polytetrafluoroethylene (PTFE) and polyvinylidene fluoride (PVdF), polyacrylonitriles (PAN), polyimides, acrylic resins and polyolefins. These resins may be used in combination with, for example, carboxymethylcellulose (CMC) or a salt thereof, and polyethylene oxide (PEO).
- the negative electrode 12 includes a negative electrode current collector and a negative electrode mixture layer disposed on the current collector.
- the negative electrode current collector may be, for example, a foil of a metal that is stable at the potentials of the negative electrode 12 , such as copper, or a film having such a metal as a skin layer.
- the negative electrode mixture layer includes a negative electrode active material and a binder, and is preferably disposed on both sides of the negative electrode current collector.
- the negative electrode 12 may be fabricated by applying a negative electrode mixture slurry including components such as a negative electrode active material and a binder onto a negative electrode current collector, drying the wet films, and rolling the coatings to form negative electrode mixture layers on both sides of the negative electrode current collector.
- the negative electrode active material is not particularly limited as long as it can reversibly store and release lithium ions.
- examples of such materials which may be used include carbon materials such as natural graphite and artificial graphite, metals which can be alloyed with Li such as silicon (Si) and tin (Sn), and oxides containing such metal elements as Si and Sn.
- the negative electrode mixture layers may include a lithium titanium composite oxide.
- the lithium titanium composite oxide serves as a negative electrode active material.
- a conductive agent such as carbon black is preferably added to the negative electrode mixture layers.
- binders which may be used in the negative electrode mixture layers include fluororesins such as PTFE and PVdF, PAN, polyimides, acrylic resins and polyolefins, similarly to the case of the positive electrode 11 .
- fluororesins such as PTFE and PVdF
- PAN polyimides
- acrylic resins and polyolefins similarly to the case of the positive electrode 11 .
- the negative electrode mixture slurry is prepared using an aqueous solvent, for example, CMC or a salt thereof, styrene-butadiene rubber (SBR), polyacrylic acid (PAA) or a salt thereof, or polyvinyl alcohol (PVA) may be used as the binder.
- SBR styrene-butadiene rubber
- PAA polyacrylic acid
- PVA polyvinyl alcohol
- the separator 13 includes a substrate 30 , a first filler layer 31 which is disposed on one side of the substrate 30 and contains phosphate salt particles 34 , and a second filler layer 32 which is disposed on the other side of the substrate 30 and contains inorganic particles 35 with a higher melting point than the phosphate salt particles 34 . That is, the separator 13 has a layered structure in which the substrate 30 is interposed between the first filler layer 31 and the second filler layer 32 . In the electrode assembly 14 , the separator 13 is disposed in such a manner that the side of the substrate 30 which bears the first filler layer 31 is directed to the positive electrode 11 side.
- the phosphate salt particles 34 contained in the first filler layer 31 are melted and polymerized to fill pores in the substrate 30 .
- the second filler layer 32 with high heat resistance ensures the shape stability of the separator 13 .
- the substrate 30 attains an enhanced shutdown effect and sufficiently suppresses the exothermic reaction at the time of short-circuiting.
- a resin layer 33 is disposed between the positive electrode 11 and the first filler layer 31 .
- the resin layer 33 is present between the positive electrode 11 and the first filler layer 31 , and is preferably disposed as a constituent element of the separator 13 on the surface of the first filler layer 31 .
- the separator 13 preferably has a layered structure represented by resin layer 33 /first filler layer 31 /substrate 30 /second filler layer 32 .
- the substrate 30 is composed of a porous sheet having ion permeability and insulating properties, for example, a microporous thin film, a woven fabric, a nonwoven fabric, etc.
- the resins which form the substrates 30 include polyolefins such as polyethylene, polypropylene and copolymers of polyethylene and ⁇ -olefins, acrylic resins, polystyrenes, polyesters and celluloses.
- the substrate 30 may be a monolayer structure, or may have a stack structure.
- the thickness of the substrate 30 is not particularly limited but is, for example, preferably not less than 3 ⁇ m and not more than 20 ⁇ m.
- the porosity of the substrate 30 is, for example, preferably not less than 30% and not more than 70%.
- the porosity of the substrate 30 is determined in the following manner.
- the average pore size of the substrate 30 is 0.02 ⁇ m to 5 ⁇ m, and preferably 0.05 ⁇ m to 1 ⁇ m.
- the average pore size of the substrate 30 is measured with, for example, Perm-Porometer (manufactured by SEIKA CORPORATION) capable of measuring pore sizes by the bubble point method (JIS K3832, ASTM F316-86).
- the first filler layer 31 is a porous layer containing phosphate salt particles 34 , and has pores in the gaps between the phosphate salt particles 34 for the passage of lithium ions.
- the phosphate salt particles 34 contained in the first filler layer 31 are melted and polymerized in an accelerated manner by heat and the positive electrode potential to fill pores in the substrate 30 , thereby interrupting the ion conduction between the positive and negative electrodes. Further, the substrate 30 itself is melted to close the pores.
- the porosity of the first filler layer 31 is, for example, preferably not less than 30% and not more than 70%.
- Examples of the phosphate salt particles 34 include Li 3 PO 4 , LiPON, Li 2 HPO 4 , LiH 2 PO 4 , Na 3 PO 4 , Na 2 HPO 4 , NaH 2 PO 4 , Zr 3 (PO 4 ) 4 , Zr(HPO 4 ) 2 , HZr 2 (PO 4 ) 3 , K 3 PO 4 , K 2 HPO 4 , KH 2 PO 4 , Ca 3 (PO 4 ) 2 , CaHPO 4 , Mg 3 (PO 4 ) 2 and MgHPO 4 .
- At least one selected from lithium phosphate (Li 3 PO 4 ), dilithium hydrogen phosphate (Li 2 HPO 4 ) and lithium dihydrogen phosphate (LiH 2 PO 4 ) is preferable.
- the phosphate salt particles 34 be melted at a temperature of about 140° C. to 190° C.
- the BET specific surface area of the phosphate salt particles 34 is preferably not less than 5 m 2 /g and not more than 100 m 2 /g, and more preferably not less than 20 m 2 /g and not more than 100 m 2 /g.
- the BET specific surface area may be measured by the BET method (the nitrogen adsorption method) described in JIS R1626.
- the phosphate salt particles 34 having a BET specific surface area in the above range are melted easily at a temperature of about 140° C. to 190° C., and thus the use of such particles makes it possible to close quickly the pores in the substrate 30 in the event of occurrence of an abnormality in the battery.
- the average particle size of the phosphate salt particles 34 is not less than 0.05 ⁇ m and not more than 2 ⁇ m. If the phosphate salt particles 34 fail to satisfy the above range of average particle size, the amount of heat generated at the time of occurrence of battery abnormality is sometimes increased as compared to when the above range is satisfied.
- the average particle size is the volume average particle size measured by a laser diffraction method at which the cumulative volume value reaches 50% median of the particle size distribution (this average particle size is also called the 50% particle size).
- the average particle size may be measured with a laser diffraction scattering grain size distribution analyzer (manufactured by HORIBA, Ltd.). In the present specification, the average particle size means this median diameter unless otherwise mentioned.
- the average particle size of the phosphate salt particles 34 is 0.05 ⁇ m to 1 ⁇ m and is smaller than the average pore size of the substrate 30 .
- the phosphate salt particles 34 having an average particle size smaller than the average pore size of the substrate 30 part of the phosphate salt particles 34 penetrate into the pores of the substrate 30 and easily close the pores of the substrate 30 in the event of occurrence of short-circuiting.
- the depth in which the phosphate salt particles 34 penetrate into the pores of the substrate 30 is preferably not less than 0.1 ⁇ m and not more than 1 ⁇ m in the direction of thickness of the substrate 30 .
- the first filler layer 31 includes a binder which binds together the phosphate salt particles 34 .
- the binders include polyolefins such as polyethylene, polypropylene and copolymers of polyethylene and ⁇ -olefins, fluororesins such as PVdF, PTFE and polyvinyl fluoride (PVF), fluororubbers such as vinylidene fluoride-hexafluoropropylene-tetrafluoroethylene copolymer and ethylene-tetrafluoroethylene copolymer, styrene-butadiene copolymer and hydrides thereof, acrylonitrile-butadiene copolymer and hydrides thereof, acrylonitrile-butadiene-styrene copolymer and hydrides thereof, methacrylate ester-acrylate ester copolymer, styrene-acrylate ester copolymer, acrylonitrile-acrylate ester copo
- the content of the phosphate salt particles 34 is preferably an amount enough to close the pores in the substrate 30 .
- the content is preferably not less than 90 mass %, and more preferably not less than 92 mass % and not more than 98 mass % of the total mass of the first filler layer 31 .
- the content of the binder in the first filler layer 31 is, for example, not less than 2 mass % and not more than 8 mass %.
- the thickness of the first filler layer 31 is not particularly limited, but is preferably not less than 1 ⁇ m and not more than 10 ⁇ m, more preferably not less than 1 ⁇ m and not more than 5 ⁇ m, and particularly preferably not less than 2 ⁇ m and not more than 4 ⁇ m. From the point of view of reducing the amount of heat generated in the battery, the thickness of the first filler layer 31 is preferably not less than 2 times and not more than 40 times, and more preferably not less than 3 times and not more than 20 times the average particle size of the phosphate salt particles 34 .
- the first filler layer 31 may further include a heteropolyacid.
- a heteropolyacid probably promotes the polycondensation of the phosphate salt that has been melted.
- the heteropolyacids include phosphomolybdic acid, phosphotungstic acid, phosphomolybdotungstic acid, phosphomolybdovanadic acid, phosphomolybdotungstovanadic acid, phosphotungstovanadic acid, silicotungstic acid, silicomolybdic acid, silicomolybdotungstic acid and silicomolybdotungstovanadic acid.
- the second filler layer 32 is a porous layer containing inorganic particles 35 with a high melting point (heat resistance), and has pores in the gaps between the inorganic particles 35 for the passage of lithium ions.
- the porosity of the second filler layer 32 is preferably not less than 30% and not more than 70%.
- the second filler layer 32 has a function to prevent a significant deformation of the substrate 30 due to abnormal heat generation in the battery and thereby enhance the shape stability of the separator 13 . By providing the second filler layer 32 that is a heat resistant layer, the shutdown effect of the substrate 30 is enhanced and the exothermic reaction at the time of short-circuiting is sufficiently suppressed.
- the inorganic particles 35 are particles which have higher thermal properties such as melting point and thermal decomposition temperature than the phosphate salt particles 34 contained in the first filler layer 31 , and are preferably composed of an insulating inorganic compound which is not melted or decomposed during the abnormal heat generation in the battery.
- Some examples of the inorganic particles 35 are particles of metal oxides, metal oxide hydrates, metal hydroxides, metal nitrides, metal carbides, metal sulfides, etc.
- the average particle size of the inorganic particles 35 is preferably not less than 0.2 ⁇ m and not more than 2 ⁇ m. If the inorganic particles fail to satisfy the above range of average particle size, the amount of heat occurring at the time of abnormal heat generation is sometimes increased as compared to when the above range is satisfied.
- Examples of the metal oxides and the metal oxide hydrates include aluminum oxide, boehmite (Al 2 O 3 H 2 O or AlOOH), magnesium oxide, titanium oxide, zirconium oxide, silicon oxide, yttrium oxide and zinc oxide.
- Examples of the metal nitrides include silicon nitride, aluminum nitride, boron nitride and titanium nitride.
- Examples of the metal carbides include silicon carbide and boron carbide.
- Examples of the metal sulfides include barium sulfate.
- Examples of the metal hydroxides include aluminum hydroxide.
- the melting point of a material which melts after transformation for example, boehmite into alumina, is the melting point of the material resulting from the transformation.
- the inorganic particles 35 may be particles of, for example, porous aluminosilicate salts such as zeolite (M 2/n O ⁇ Al 2 O 3 ⁇ xSiO 2 ⁇ yH 2 O wherein M denotes a metal element, x ⁇ 2 and y ⁇ 0), layered silicate salts such as talc (Mg 3 Si 4 O 10 (OH) 2 ), barium titanate (BaTiO 3 ) and strontium titanate (SrTiO 3 ).
- porous aluminosilicate salts such as zeolite (M 2/n O ⁇ Al 2 O 3 ⁇ xSiO 2 ⁇ yH 2 O wherein M denotes a metal element, x ⁇ 2 and y ⁇ 0)
- layered silicate salts such as talc (Mg 3 Si 4 O 10 (OH) 2 ), barium titanate (BaTiO 3 ) and strontium titanate (SrTiO 3 ).
- the second filler layer 32 includes a binder which binds together the inorganic particles 35 .
- the binder may be a resin similar to one used in the first filler layer 31 .
- the content of the inorganic particles 35 is preferably not less than 90 mass %, and more preferably not less than 92 mass % and not more than 98 mass % of the total mass of the second filler layer 32 .
- the content of the binder in the second filler layer 32 is, for example, not less than 2 mass % and not more than 8 mass %.
- the thickness of the second filler layer 32 is not particularly limited, but is preferably not less than 1 ⁇ m and not more than 5 ⁇ m, and particularly preferably not less than 2 ⁇ m and not more than 4 ⁇ m.
- the resin layer 33 is formed on the surface of the first filler layer 31 and is in contact with the surface of the positive electrode 11 .
- the resin layer 33 has a function to ensure the adhesion between the first filler layer 31 and the positive electrode 11 when abnormal heat is generated in the battery.
- the positive electrode 11 and the separator 13 maintain good contact therebetween even when the positive electrode 11 is deformed by a temperature increase inside the battery due to, for example, an internal short-circuit of the battery. Consequently, the phosphate salt particles 34 are allowed to be melted and polycondensed uniformly with ease.
- the resin layer 33 is composed of a resin which is melted or softened during hot pressing of the electrode assembly and/or at the time of abnormal heat generation in the charged battery.
- the resin layer 33 may contain a filler such as an inorganic compound, but is preferably composed of a resin alone.
- the resin layer 33 may be bonded to the surface of the positive electrode 11 by hot pressing during the fabrication of the electrode assembly.
- the resin layer 33 may be one which does not exhibit adhesion with respect to the surface of the positive electrode 11 during normal use of the battery and develops the adhesion in the event of abnormal heat generation.
- the resin layer 33 suitably includes a resin having a glass transition temperature of not more than 130° C.
- a binder resin used in the electrode mixture layers may be used in the resin layer 33 .
- Some preferred examples of the resins are fluororesins such as PTFE and PVdF, PAN, acrylic resins and polyolefins.
- the thickness of the resin layer 33 is not particularly limited, but is preferably not more than 5 ⁇ m, more preferably not less than 0.5 ⁇ m and not more than 5 ⁇ m, and particularly preferably not less than 1 ⁇ m and not more than 3 ⁇ m. When the thickness is in this range, the resin layer 33 does not adversely affect battery characteristics such as charge discharge characteristics and battery capacity, and easily ensures a good contact between the positive electrode 11 and the separator 13 even when the positive electrode 11 is deformed at the time of occurrence of an abnormality.
- the separator 13 having a layered structure represented by resin layer 33 /first filler layer 31 /substrate 30 /second filler layer 32 may be produced by forming a first filler layer 31 and a resin layer 33 sequentially on one side of a porous resin substrate 30 , and forming a second filler layer 32 on the other side of the substrate 30 .
- the first filler layer 31 may be formed by providing a slurry composition including phosphate salt particles 34 , a binder and a dispersion medium, applying the composition onto one side of a substrate 30 , and drying the wet film.
- the second filler layer 32 may be formed by providing a slurry composition including inorganic particles 35 , a binder and a dispersion medium, applying the composition onto the other side of the substrate 30 , and drying the wet film.
- the resin layer 33 may be formed by applying a resin solution onto the surface of the first filler layer 31 , and drying the wet film.
- the separator 13 has a structure in which the resin layer 33 , the first filler layer 31 , the substrate 30 and the second filler layer 32 are stacked in this order, and is disposed between the positive electrode 11 and the negative electrode 12 in such a manner that the resin layer 33 is directed to the positive electrode 11 side.
- the first filler layer 31 and the resin layer 33 may be formed on both sides of the substrate 30 , but are preferably formed on only the side of the substrate 30 opposed to the positive electrode 11 from points of view such as increasing the capacity.
- the second filler layer 32 is formed on only the other side of the substrate 30 .
- the melting and polycondensation of the phosphate salt particles 34 contained in the first filler layer 31 are induced not only by heat in the event of occurrence of battery abnormality but also by the potential of the positive electrode 11 .
- the first filler layer 31 be disposed near the positive electrode 11 .
- separator was fabricated which had a layered structure represented by first filler layer containing phosphate salt particles/polyethylene porous substrate/second filler layer containing boehmite particles.
- Lithium phosphate particles (Li 3 PO 4 ) having a BET specific surface area of 54.07 m 2 /g and an average particle size of 0.93 ⁇ m and poly-N-vinylacetamide were mixed together in a mass ratio of 100:6.5, and NMP was added.
- a first slurry composition having a solid concentration of 15 mass % was thus prepared.
- Boehmite particles and poly-N-vinylacetamide were mixed together in a mass ratio of 100:6.5, and N-methyl-2-pyrrolidone (NMP) was added.
- a second slurry composition having a solid concentration of 15 mass % was thus prepared.
- the second slurry composition was applied by a gravure coating method onto one side of a 12 ⁇ m thick polyethylene porous monolayer substrate so that the coating thickness after drying would be 4 ⁇ m, and the wet film was dried to form a second filler layer.
- the first slurry composition was applied by a gravure coating method onto the other side of the porous substrate so that the coating thickness after drying would be 4 ⁇ m, and the wet film was dried to form a first filler layer.
- a cross section of the separator was exposed with a cross section polisher and was observed with a scanning electron microscope (SEM). Based on the sectional image, the phosphate salt particles were found to have penetrated into the separator substrate with a depth of 0.5 ⁇ m in the direction of thickness of the substrate.
- Particles of lithium nickel cobalt aluminum composite oxide represented by Li 1.05 Ni 0.82 Co 0.15 Al 0.03 O 2 were used as positive electrode active material particles.
- the average secondary particle size of the particles was about 15 ⁇ m.
- the positive electrode active material, carbon black and PVdF were mixed together in a mass ratio of 100:1:1, and N-methyl-2-pyrrolidone was added to the mixture.
- a positive electrode mixture slurry was thus prepared.
- the positive electrode mixture slurry was applied to both sides of an aluminum foil as a positive electrode current collector.
- the wet films were dried and rolled with a roller.
- a positive electrode was fabricated in which positive electrode mixture layers were disposed on both sides of the positive electrode current collector.
- the packing density of the positive electrode mixture layers was 3.70 g/cm 3 .
- part of the surface of the positive electrode current collector was left exposed from the positive electrode mixture layer, and a positive electrode lead made of aluminum was welded to the exposed portion of the positive electrode.
- Lithium hexafluorophosphate LiPF 6 was dissolved with a concentration of 1.0 mol/L into a mixed solvent obtained by mixing ethylene carbonate (EC), ethyl methyl carbonate (EMC) and dimethyl carbonate (DMC) in a volume ratio of 3:3:4. Further, vinylene carbonate (VC) was dissolved with a concentration of 1.0 mass % relative to the mixed solvent. A nonaqueous electrolyte was thus prepared.
- the positive electrode and the negative electrode were wound together via the separator in such a manner that the first filler layer of the separator was in contact with the positive electrode.
- the coil thus formed was hot pressed at 80° C. to give a flat wound electrode assembly.
- the electrode assembly was placed into a battery exterior package composed of an aluminum laminate sheet.
- the nonaqueous electrolyte was poured, and the exterior package was sealed.
- a 650 mAh nonaqueous electrolyte secondary battery was thus fabricated.
- the battery fabricated was charged at a constant current of 0.3 It to a battery voltage of 4.2 V, and was thereafter charged at a constant voltage of 4.2 V until the current value reached 0.05 It.
- the battery was thus brought to a charged state.
- the tip of a 3 mm ⁇ round nail was pushed vertically, at a speed of 10 mm/sec, into a lateral central portion of the battery charged above.
- the operation of the round nail penetration was stopped when the round nail completely pierced the battery.
- the maximum temperature that was reached was measured with respect to a lateral portion of the battery 5 mm away from the location of nail penetration through the lateral portion of the battery.
- This nail penetration test can simulate abnormal heat generation stemming from an internal short-circuit of the battery.
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, dilithium hydrogen phosphate (Li 2 HPO 4 ) having a BET specific surface area of 7.10 m 2 /g and an average particle size of 0.33 ⁇ m were used as the phosphate salt particles.
- dilithium hydrogen phosphate Li 2 HPO 4
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, lithium phosphate (Li 3 PO 4 ) having a BET specific surface area of 61.35 m 2 /g and an average particle size of 0.15 ⁇ m were used as the phosphate salt particles.
- lithium phosphate Li 3 PO 4
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, lithium phosphate (Li 3 PO 4 ) having a BET specific surface area of 3.65 m 2 /g and an average particle size of 1.60 ⁇ m were used as the phosphate salt particles.
- lithium phosphate Li 3 PO 4
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, the second filler layer was formed between the first filler layer and the substrate.
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, a 50:50 by mass mixture of the first slurry composition and the second slurry composition was applied to one side of the polyethylene porous substrate so that the coating thickness after drying would be 4 ⁇ m, and thereby a layer was formed which contained the mixture of lithium phosphate particles and boehmite particles.
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, the second filler layer was not formed.
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, the first filler layer was not formed.
- a battery was fabricated and a nail penetration test was performed in the same manner as in EXAMPLE 1, except that in the fabrication of a separator, the first filler layer and the second filler layer were not formed.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Composite Materials (AREA)
- Inorganic Chemistry (AREA)
- Manufacturing & Machinery (AREA)
- Cell Separators (AREA)
- Secondary Cells (AREA)
Abstract
Description
Porosity ε (%)=((ρV−W)/(ρV))×100
Porosity (%) of first filler layer=100−[[W÷(d×ρ)]×100]
-
- W: Basis weight (g/cm2) of first filler layer
- d: Thickness (cm) of first filler layer
- ρ: Average density (g/cm3) of first filler layer
(2) Boehmite particles and poly-N-vinylacetamide were mixed together in a mass ratio of 100:6.5, and N-methyl-2-pyrrolidone (NMP) was added. A second slurry composition having a solid concentration of 15 mass % was thus prepared.
(3) The second slurry composition was applied by a gravure coating method onto one side of a 12 μm thick polyethylene porous monolayer substrate so that the coating thickness after drying would be 4 μm, and the wet film was dried to form a second filler layer.
(4) The first slurry composition was applied by a gravure coating method onto the other side of the porous substrate so that the coating thickness after drying would be 4 μm, and the wet film was dried to form a first filler layer.
| TABLE 1 | |||
| First filler layer | Second filler layer | ||
| Average | Maximum | |||||||
| Phosphate | BET specific | particle | Inorganic | Inorganic | temperature | |||
| salt | surface area | size | particles | particles | Location | reached | ||
| EX. 1 | Li3PO4 | 54.07 m2/g | 0.93 μm | — | Boehmite | Between substrate | 458° C. |
| and negative electrode | |||||||
| Ex. 2 | Li2HPO4 | 7.10 m2/g | 0.33 μm | — | Boehmite | Between substrate | 460° C. |
| and negative electrode | |||||||
| EX. 3 | Li3PO4 | 61.35 m2/g | 0.15 μm | — | Boehmite | Between substrate | 453° C. |
| and negative electrode | |||||||
| COMP. | Li3PO4 | 3.65 m2/g | 1.60 μm | — | Boehmite | Between substrate | 478° C. |
| EX. 1 | and negative electrode | ||||||
| COMP. | Li3PO4 | 54.07 m2/g | 0.93 μm | — | Boehmite | Between first filler | 488° C. |
| EX. 2 | layer and substrate | ||||||
| COMP. | Li3PO4 | 54.07 m2/g | 0.93 μm | Boehmite | — | — | 492° C. |
| EX. 3 | |||||||
| COMP. | Li3PO4 | 54.07 m2/g | 0.93 μm | — | — | — | 500° C. |
| EX. 4 | |||||||
| COMP. | — | — | — | — | Boehmite | Between substrate | 490° C. |
| EX. 5 | and negative electrode | ||||||
| COMP. | — | — | — | — | — | — | 506° C. |
| EX. 6 | |||||||
-
- 10 NONAQUEOUS ELECTROLYTE SECONDARY BATTERY
- 11 POSITIVE ELECTRODE
- 12 NEGATIVE ELECTRODE
- 13 SEPARATOR
- 14 ELECTRODE ASSEMBLY
- 15 BATTERY CASE
- 16 EXTERIOR CASE
- 17 SEAL BODY
- 18, 19 INSULATING PLATES
- 20 POSITIVE ELECTRODE LEAD
- 21 NEGATIVE ELECTRODE LEAD
- 22 OVERHANG
- 23 FILTER
- 24 LOWER VALVE
- 25 INSULATING MEMBER
- 26 UPPER VALVE
- 27 CAP
- 28 GASKET
- 30 SUBSTRATE
- 31 FIRST FILLER LAYER
- 32 SECOND FILLER LAYER
- 33 RESIN LAYER
- 34 PHOSPHATE SALT PARTICLES
- 35 INORGANIC PARTICLES
Claims (3)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017229192 | 2017-11-29 | ||
| JP2017-229192 | 2017-11-29 | ||
| PCT/JP2018/040793 WO2019107067A1 (en) | 2017-11-29 | 2018-11-02 | Nonaqueous electrolyte secondary battery |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20200243827A1 US20200243827A1 (en) | 2020-07-30 |
| US12224452B2 true US12224452B2 (en) | 2025-02-11 |
Family
ID=66665082
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/637,034 Active 2038-11-16 US12224452B2 (en) | 2017-11-29 | 2018-11-02 | Nonaqueous electrolyte secondary battery including separator with filler layers on opposite sides of substrate |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US12224452B2 (en) |
| EP (1) | EP3719866B1 (en) |
| JP (1) | JP7162187B2 (en) |
| CN (1) | CN111052446B (en) |
| WO (1) | WO2019107067A1 (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20230042242A1 (en) * | 2019-12-27 | 2023-02-09 | Panasonic Intellectual Property Management Co., Ltd. | Lithium secondary battery |
| US12237535B2 (en) | 2020-08-14 | 2025-02-25 | Ubatt Inc. | Flame-retardant separator including porous substrate with crystalline sulfonyl-containing metal salt, and electrochemical device having the same |
| CN112615066A (en) * | 2020-12-17 | 2021-04-06 | 北京大学深圳研究生院 | Mixed gel polymer electrolyte, preparation method thereof and all-solid-state zinc ion battery |
| WO2022165725A1 (en) * | 2021-02-04 | 2022-08-11 | 江苏时代新能源科技有限公司 | Electrode assembly, battery cell, battery, and electrode assembly manufacturing method and device |
| JPWO2023053771A1 (en) * | 2021-09-30 | 2023-04-06 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5981107A (en) * | 1996-12-17 | 1999-11-09 | Mitsubishi Denki Kabushiki Kaisha | Lithium ion secondary battery and method of fabricating thereof |
| JP2013105521A (en) | 2011-11-10 | 2013-05-30 | Nissan Motor Co Ltd | Separator with heat-resistant insulating layer |
| JP2013114764A (en) | 2011-11-25 | 2013-06-10 | Sony Corp | Lithium ion battery and separator, and battery pack, electronic device, electric vehicle, power storage device, and electric power system |
| JP2014180822A (en) | 2013-03-19 | 2014-09-29 | Sekisui Chem Co Ltd | Laminate film and separator for battery and battery each using the same |
| EP2793305A1 (en) | 2011-12-13 | 2014-10-22 | Kokam Co., Ltd. | High heat resistance composite separator for lithium secondary battery and lithium secondary battery including same |
| US20140356695A1 (en) | 2013-05-31 | 2014-12-04 | Sony Corporation | Battery, battery pack, electronic apparatus, electric vehicle, electrical storage apparatus and electricity system |
| WO2016020737A1 (en) | 2014-08-06 | 2016-02-11 | Toyota Jidosha Kabushiki Kaisha | Nonaqueous electrolyte secondary battery |
| JP2016072120A (en) | 2014-09-30 | 2016-05-09 | 旭化成イーマテリアルズ株式会社 | Separator for power storage device |
| JP2017063041A (en) | 2016-11-07 | 2017-03-30 | ソニー株式会社 | Batteries, battery packs, electronic devices, electric vehicles, power storage devices, and power systems |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI330136B (en) * | 2005-11-28 | 2010-09-11 | Lg Chemical Ltd | Organic/inorganic composite porous membrane and electrochemical device using the same |
| WO2010128548A1 (en) * | 2009-05-08 | 2010-11-11 | トヨタ自動車株式会社 | Cell separator manufacturing method |
| WO2010143677A1 (en) * | 2009-06-10 | 2010-12-16 | 日立マクセル株式会社 | Separator for electrochemical element, and electrochemical element including same |
| JPWO2012165624A1 (en) * | 2011-06-03 | 2015-02-23 | 富士シリシア化学株式会社 | Separator and electrochemical element |
| US9673436B2 (en) * | 2011-11-15 | 2017-06-06 | Toyota Jidosha Kabushiki Kaisha | Nonaqueous electrolyte secondary battery |
| WO2014168392A1 (en) * | 2013-04-09 | 2014-10-16 | Lg Electronics Inc. | Separator for secondary battery and method of manufacturing the same |
| JP6137554B2 (en) * | 2014-08-21 | 2017-05-31 | トヨタ自動車株式会社 | Nonaqueous electrolyte secondary battery and separator for the battery |
| KR101709695B1 (en) * | 2014-08-25 | 2017-02-24 | 삼성에스디아이 주식회사 | Heat-resistant, nonflammable, coated separator and battery |
| JP6365889B2 (en) * | 2015-10-13 | 2018-08-01 | トヨタ自動車株式会社 | Nonaqueous electrolyte secondary battery |
| CN206210904U (en) * | 2016-11-04 | 2017-05-31 | 深圳市安一福科技有限公司 | A kind of lithium ion battery barrier film |
| CN107093693A (en) * | 2017-05-04 | 2017-08-25 | 南通中航泛能新材料有限公司 | A kind of composite diaphragm and its application in lithium ion battery |
-
2018
- 2018-11-02 JP JP2019557088A patent/JP7162187B2/en active Active
- 2018-11-02 CN CN201880052381.4A patent/CN111052446B/en active Active
- 2018-11-02 US US16/637,034 patent/US12224452B2/en active Active
- 2018-11-02 EP EP18883187.9A patent/EP3719866B1/en active Active
- 2018-11-02 WO PCT/JP2018/040793 patent/WO2019107067A1/en not_active Ceased
Patent Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5981107A (en) * | 1996-12-17 | 1999-11-09 | Mitsubishi Denki Kabushiki Kaisha | Lithium ion secondary battery and method of fabricating thereof |
| JP2013105521A (en) | 2011-11-10 | 2013-05-30 | Nissan Motor Co Ltd | Separator with heat-resistant insulating layer |
| US20140287295A1 (en) | 2011-11-10 | 2014-09-25 | Nissan Motor Co., Ltd. | Separator with heat resistant insulation layer |
| JP2013114764A (en) | 2011-11-25 | 2013-06-10 | Sony Corp | Lithium ion battery and separator, and battery pack, electronic device, electric vehicle, power storage device, and electric power system |
| JP2015505137A (en) | 2011-12-13 | 2015-02-16 | コカン カンパニー リミテッドKokam Co.,Ltd. | High heat-resistant composite separator for lithium secondary battery and lithium secondary battery including the same |
| EP2793305A1 (en) | 2011-12-13 | 2014-10-22 | Kokam Co., Ltd. | High heat resistance composite separator for lithium secondary battery and lithium secondary battery including same |
| US20140370358A1 (en) | 2011-12-13 | 2014-12-18 | Kokam Co., Ltd. | High heat resistance composite separator for lithium secondary battery and lithium secondary battery including same |
| JP2014180822A (en) | 2013-03-19 | 2014-09-29 | Sekisui Chem Co Ltd | Laminate film and separator for battery and battery each using the same |
| US20140356695A1 (en) | 2013-05-31 | 2014-12-04 | Sony Corporation | Battery, battery pack, electronic apparatus, electric vehicle, electrical storage apparatus and electricity system |
| WO2016020737A1 (en) | 2014-08-06 | 2016-02-11 | Toyota Jidosha Kabushiki Kaisha | Nonaqueous electrolyte secondary battery |
| CN106663770A (en) | 2014-08-06 | 2017-05-10 | 丰田自动车株式会社 | Nonaqueous electrolyte secondary battery |
| US20170229743A1 (en) * | 2014-08-06 | 2017-08-10 | Toyota Jidosha Kabushiki Kaisha | Nonaqueous electrolyte secondary battery |
| JP2016072120A (en) | 2014-09-30 | 2016-05-09 | 旭化成イーマテリアルズ株式会社 | Separator for power storage device |
| JP2017063041A (en) | 2016-11-07 | 2017-03-30 | ソニー株式会社 | Batteries, battery packs, electronic devices, electric vehicles, power storage devices, and power systems |
Non-Patent Citations (3)
| Title |
|---|
| English Translation of Search Report dated Dec. 2, 2021, issued in counterpart CN Application No. 201880052381.4. (3 pages). |
| International Search Report dated Jan. 29, 2019, issued in counterpart Application No. PCT/JP2018/040793 (2 pages). |
| The Extended European Search Report dated Jan. 12, 2021, issued in counterpart EP Application No. 18883187.9. (9 pages). |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2019107067A1 (en) | 2020-11-19 |
| EP3719866A4 (en) | 2021-01-27 |
| JP7162187B2 (en) | 2022-10-28 |
| US20200243827A1 (en) | 2020-07-30 |
| WO2019107067A1 (en) | 2019-06-06 |
| EP3719866A1 (en) | 2020-10-07 |
| CN111052446B (en) | 2022-12-09 |
| CN111052446A (en) | 2020-04-21 |
| EP3719866B1 (en) | 2024-08-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11949097B2 (en) | Nonaqueous electrolyte secondary battery | |
| US11621458B2 (en) | Nonaqueous electrolyte secondary battery including separator having substrate, phosphate salt layer, inorganic particle layer, and resin layer between separator and positive electrode | |
| US12224452B2 (en) | Nonaqueous electrolyte secondary battery including separator with filler layers on opposite sides of substrate | |
| CN112106225B (en) | Nonaqueous electrolyte secondary battery | |
| US11942645B2 (en) | Non-aqueous electrolyte secondary battery having separator with multilayer structure | |
| US11695117B2 (en) | Non-aqueous electrolyte secondary battery | |
| EP3678218B1 (en) | Nonaqueous electrolyte secondary battery | |
| WO2023032558A1 (en) | Negative electrode for secondary battery, and secondary battery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| AS | Assignment |
Owner name: PANASONIC INTELLECTUAL PROPERTY MANAGEMENT CO., LTD., JAPAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:MORIKAWA, YUKI;SUGIMORI, MASANORI;BABA, YASUNORI;AND OTHERS;REEL/FRAME:052315/0169 Effective date: 20200120 |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE AFTER FINAL ACTION FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NOTICE OF ALLOWANCE MAILED -- APPLICATION RECEIVED IN OFFICE OF PUBLICATIONS |
|
| ZAAB | Notice of allowance mailed |
Free format text: ORIGINAL CODE: MN/=. |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT VERIFIED |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |