US11459506B2 - Additive and application thereof - Google Patents
Additive and application thereof Download PDFInfo
- Publication number
- US11459506B2 US11459506B2 US16/749,406 US202016749406A US11459506B2 US 11459506 B2 US11459506 B2 US 11459506B2 US 202016749406 A US202016749406 A US 202016749406A US 11459506 B2 US11459506 B2 US 11459506B2
- Authority
- US
- United States
- Prior art keywords
- group
- formula
- linear
- branched
- alkyl group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active, expires
Links
- 0 C.C.[1*]C1CCC(CC2CCC(C)CC2)CC1 Chemical compound C.C.[1*]C1CCC(CC2CCC(C)CC2)CC1 0.000 description 48
- YLOWTPHXXDJBAH-UHFFFAOYSA-N C=C(C)C(=O)OC.C=C(C)C(=O)OC Chemical compound C=C(C)C(=O)OC.C=C(C)C(=O)OC YLOWTPHXXDJBAH-UHFFFAOYSA-N 0.000 description 21
- VVQNEPGJFQJSBK-UHFFFAOYSA-N C=C(C)C(=O)OC Chemical compound C=C(C)C(=O)OC VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 16
- JJEFKIUULLHMMD-UHFFFAOYSA-N C.C=C(C)C(=O)CC.C=C(C)C(=O)CC Chemical compound C.C=C(C)C(=O)CC.C=C(C)C(=O)CC JJEFKIUULLHMMD-UHFFFAOYSA-N 0.000 description 2
- QYXXAANAKDNBQA-UHFFFAOYSA-N C.C=C(C)C(=O)OC.C=C(C)C(=O)OC.C=C1CCC(C)CO1.C=C1OCC(C)CO1.CC1CC(=O)CC(=O)C1.CC1CC(=O)CC1=O Chemical compound C.C=C(C)C(=O)OC.C=C(C)C(=O)OC.C=C1CCC(C)CO1.C=C1OCC(C)CO1.CC1CC(=O)CC(=O)C1.CC1CC(=O)CC1=O QYXXAANAKDNBQA-UHFFFAOYSA-N 0.000 description 2
- GHHGVSCQWPVENX-UHFFFAOYSA-N C=C(C)C(=O)CC Chemical compound C=C(C)C(=O)CC GHHGVSCQWPVENX-UHFFFAOYSA-N 0.000 description 2
- IASLDLGGHXYCEO-HWKANZROSA-N C/C=C/C1CCC(C2CCC(CCC)CC2)CC1 Chemical compound C/C=C/C1CCC(C2CCC(CCC)CC2)CC1 IASLDLGGHXYCEO-HWKANZROSA-N 0.000 description 1
- UTRMCIMLVCSFES-UHFFFAOYSA-N C=C(C)C(=O)OC.C=C(C)C(=O)OC.C=C1CCC(C)CO1.C=C1OCC(C)CO1.CC1CC(=O)CC(=O)C1.CC1CC(=O)CC1=O Chemical compound C=C(C)C(=O)OC.C=C(C)C(=O)OC.C=C1CCC(C)CO1.C=C1OCC(C)CO1.CC1CC(=O)CC(=O)C1.CC1CC(=O)CC1=O UTRMCIMLVCSFES-UHFFFAOYSA-N 0.000 description 1
- VCRDWYJILHMKSR-UHFFFAOYSA-N C=C(C)C(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)C.Cc1cc(C)c([Si](C)(C)CCCC(CO)CO)c(C)c1 Chemical compound C=C(C)C(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)C.Cc1cc(C)c([Si](C)(C)CCCC(CO)CO)c(C)c1 VCRDWYJILHMKSR-UHFFFAOYSA-N 0.000 description 1
- VJOPXNOSSCDRNA-UHFFFAOYSA-N C=C(C)C(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)CO.C=C(C)C(=O)OCC(CO)CCC[Si](C)(C)c1c(C)cc(C)cc1C.C=CC(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)C.CCOC(=O)C(CCC[Si](C)(C)c1c(C)cc(C)cc1C)C(=O)OCC.Cc1cc(C)c(Br)c(C)c1.Cc1cc(C)c([Si](C)(C)CCCC(CO)CO)c(C)c1.[H][Si](C)(C)c1c(C)cc(C)cc1C Chemical compound C=C(C)C(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)CO.C=C(C)C(=O)OCC(CO)CCC[Si](C)(C)c1c(C)cc(C)cc1C.C=CC(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)C.CCOC(=O)C(CCC[Si](C)(C)c1c(C)cc(C)cc1C)C(=O)OCC.Cc1cc(C)c(Br)c(C)c1.Cc1cc(C)c([Si](C)(C)CCCC(CO)CO)c(C)c1.[H][Si](C)(C)c1c(C)cc(C)cc1C VJOPXNOSSCDRNA-UHFFFAOYSA-N 0.000 description 1
- IMEMYAWQIJFBLJ-UHFFFAOYSA-N C=C(C)C(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C(=C)C Chemical compound C=C(C)C(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C(=C)C IMEMYAWQIJFBLJ-UHFFFAOYSA-N 0.000 description 1
- UUCKOEZSVXHXFT-UHFFFAOYSA-N C=C(C)C(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C(=C)CO Chemical compound C=C(C)C(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C(=C)CO UUCKOEZSVXHXFT-UHFFFAOYSA-N 0.000 description 1
- FPLWVLGDXVZWIQ-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](C)(C)c1c(C)cc(C)cc1C.[H][Si](C)(C)c1c(C)cc(C)cc1C Chemical compound C=C(C)C(=O)OCCC[Si](C)(C)c1c(C)cc(C)cc1C.[H][Si](C)(C)c1c(C)cc(C)cc1C FPLWVLGDXVZWIQ-UHFFFAOYSA-N 0.000 description 1
- SSYFDSABYNUSOO-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si](C)(C)CCCOC(=O)C(=C)C)c1OCC Chemical compound C=C(C)C(=O)OCCC[Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si](C)(C)CCCOC(=O)C(=C)C)c1OCC SSYFDSABYNUSOO-UHFFFAOYSA-N 0.000 description 1
- RWXDBMWAJYRKJM-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si](C)(C)CCCOC(=O)C(=C)C)c1OCC.CCCCCC1CCC(C2CCC(c3cc(Br)c(O)c(Br)c3)CC2)CC1.CCCCCC1CCC(C2CCC(c3cc(Br)c(OCC)c(Br)c3)CC2)CC1.CCCCCC1CCC(C2CCC(c3ccc(O)cc3)CC2)CC1.[H][Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si]([H])(C)C)c1OCC Chemical compound C=C(C)C(=O)OCCC[Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si](C)(C)CCCOC(=O)C(=C)C)c1OCC.CCCCCC1CCC(C2CCC(c3cc(Br)c(O)c(Br)c3)CC2)CC1.CCCCCC1CCC(C2CCC(c3cc(Br)c(OCC)c(Br)c3)CC2)CC1.CCCCCC1CCC(C2CCC(c3ccc(O)cc3)CC2)CC1.[H][Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si]([H])(C)C)c1OCC RWXDBMWAJYRKJM-UHFFFAOYSA-N 0.000 description 1
- FYPDVDVAICWNPK-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 Chemical compound C=C(C)C(=O)OCCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 FYPDVDVAICWNPK-UHFFFAOYSA-N 0.000 description 1
- YQBMFKRKGRNOPP-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)C)c1c(C)cc(C)cc1C.Cc1cc(C)c([Si](C)(CCCO)CCCO)c(C)c1 Chemical compound C=C(C)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)C)c1c(C)cc(C)cc1C.Cc1cc(C)c([Si](C)(CCCO)CCCO)c(C)c1 YQBMFKRKGRNOPP-UHFFFAOYSA-N 0.000 description 1
- KDDVWXYDKSMVRP-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 Chemical compound C=C(C)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 KDDVWXYDKSMVRP-UHFFFAOYSA-N 0.000 description 1
- NPIJKXDGDJUYAG-UHFFFAOYSA-N C=C(C)C(=O)OCCC[Si](CCCOC(=O)C(=C)C)(CCCOC(=O)C(=C)C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 Chemical compound C=C(C)C(=O)OCCC[Si](CCCOC(=O)C(=C)C)(CCCOC(=O)C(=C)C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 NPIJKXDGDJUYAG-UHFFFAOYSA-N 0.000 description 1
- NYUBDMREDGRUCY-UHFFFAOYSA-N C=C(C)C(=O)OCC[Si](C)(CCCOc1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)CCOC(=O)C(=C)C Chemical compound C=C(C)C(=O)OCC[Si](C)(CCCOc1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)CCOC(=O)C(=C)C NYUBDMREDGRUCY-UHFFFAOYSA-N 0.000 description 1
- HVTJQICZTQZLSK-UHFFFAOYSA-N C=C(C)C(=O)Oc1ccc(-c2ccc(OC(=O)C(=C)C)cc2)cc1 Chemical compound C=C(C)C(=O)Oc1ccc(-c2ccc(OC(=O)C(=C)C)cc2)cc1 HVTJQICZTQZLSK-UHFFFAOYSA-N 0.000 description 1
- ONSJJWAVDPUCTF-UHFFFAOYSA-N C=C(CO)C(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)CO.C=CC(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C=C Chemical compound C=C(CO)C(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C(=C)CO.C=CC(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C=C ONSJJWAVDPUCTF-UHFFFAOYSA-N 0.000 description 1
- PMTMOYANVKUKEI-UHFFFAOYSA-N C=C(CO)C(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C(=C)CO Chemical compound C=C(CO)C(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C(=C)CO PMTMOYANVKUKEI-UHFFFAOYSA-N 0.000 description 1
- KCZKSAWAYWYECG-UHFFFAOYSA-N C=C(CO)C(=O)OCCC[Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si](C)(C)CCCOC(=O)C(=C)CO)c1OCC Chemical compound C=C(CO)C(=O)OCCC[Si](C)(C)c1cc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc([Si](C)(C)CCCOC(=O)C(=C)CO)c1OCC KCZKSAWAYWYECG-UHFFFAOYSA-N 0.000 description 1
- SAHHYNYBQJJZND-UHFFFAOYSA-N C=C(CO)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)CO)c1c(C)cc(C)cc1C.C=CC(=O)OCCC[Si](C)(CCCOC(=O)C=C)c1c(C)cc(C)cc1C.C=CC[Si](C)(CC=C)c1c(C)cc(C)cc1C.Cc1cc(C)c(Br)c(C)c1.Cc1cc(C)c([SiH](CCCO)CCCO)c(C)c1 Chemical compound C=C(CO)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)CO)c1c(C)cc(C)cc1C.C=CC(=O)OCCC[Si](C)(CCCOC(=O)C=C)c1c(C)cc(C)cc1C.C=CC[Si](C)(CC=C)c1c(C)cc(C)cc1C.Cc1cc(C)c(Br)c(C)c1.Cc1cc(C)c([SiH](CCCO)CCCO)c(C)c1 SAHHYNYBQJJZND-UHFFFAOYSA-N 0.000 description 1
- UKUACADVXNIXHO-UHFFFAOYSA-N C=C(CO)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)CO)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 Chemical compound C=C(CO)C(=O)OCCC[Si](C)(CCCOC(=O)C(=C)CO)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 UKUACADVXNIXHO-UHFFFAOYSA-N 0.000 description 1
- SSGXSJGDELSDSF-UHFFFAOYSA-N C=C(CO)C(=O)OCCC[Si](CCCOC(=O)C(=C)CO)(CCCOC(=O)C(=C)CO)c1c(C)cc(C)cc1C.C=CC(=O)OCCC[Si](CCCOC(=O)C=C)(CCCOC(=O)C=C)c1c(C)cc(C)cc1C.C=CC[Si](CC=C)(CC=C)c1c(C)cc(C)cc1C.Cc1cc(C)c(Br)c(C)c1.Cc1cc(C)c([Si](CCCO)(CCCO)CCCO)c(C)c1 Chemical compound C=C(CO)C(=O)OCCC[Si](CCCOC(=O)C(=C)CO)(CCCOC(=O)C(=C)CO)c1c(C)cc(C)cc1C.C=CC(=O)OCCC[Si](CCCOC(=O)C=C)(CCCOC(=O)C=C)c1c(C)cc(C)cc1C.C=CC[Si](CC=C)(CC=C)c1c(C)cc(C)cc1C.Cc1cc(C)c(Br)c(C)c1.Cc1cc(C)c([Si](CCCO)(CCCO)CCCO)c(C)c1 SSGXSJGDELSDSF-UHFFFAOYSA-N 0.000 description 1
- PPCDEOIEUBOGBL-UHFFFAOYSA-N C=C(CO)C(=O)OCCC[Si](CCCOC(=O)C(=C)CO)(CCCOC(=O)C(=C)CO)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 Chemical compound C=C(CO)C(=O)OCCC[Si](CCCOC(=O)C(=C)CO)(CCCOC(=O)C(=C)CO)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 PPCDEOIEUBOGBL-UHFFFAOYSA-N 0.000 description 1
- RNEZCRNHTKUCAC-UHFFFAOYSA-N C=C(CO)C(=O)OCC[Si](C)(CCCOc1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)CCOC(=O)C(=C)CO Chemical compound C=C(CO)C(=O)OCC[Si](C)(CCCOc1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)CCOC(=O)C(=C)CO RNEZCRNHTKUCAC-UHFFFAOYSA-N 0.000 description 1
- VELNOSKDXNRCSS-UHFFFAOYSA-N C=C(CO)C(=O)OCC[Si](CCCOc1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)(CCOC(=O)C(=C)CO)CCOC(=O)C(=C)CO Chemical compound C=C(CO)C(=O)OCC[Si](CCCOc1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)(CCOC(=O)C(=C)CO)CCOC(=O)C(=C)CO VELNOSKDXNRCSS-UHFFFAOYSA-N 0.000 description 1
- DVDZNMYLOFMMBL-UHFFFAOYSA-N C=C1CCC(C)CO1.C=C1OCC(C)CO1.CC1CC(=O)CC(=O)C1.CC1CC(=O)CC1=O Chemical compound C=C1CCC(C)CO1.C=C1OCC(C)CO1.CC1CC(=O)CC(=O)C1.CC1CC(=O)CC1=O DVDZNMYLOFMMBL-UHFFFAOYSA-N 0.000 description 1
- CUGMNQZFOAZWMR-UHFFFAOYSA-N C=CC(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C=C.Cc1cc(C)c([Si](C)(C)CCCC(CO)CO)c(C)c1 Chemical compound C=CC(=O)OCC(CCC[Si](C)(C)c1c(C)cc(C)cc1C)COC(=O)C=C.Cc1cc(C)c([Si](C)(C)CCCC(CO)CO)c(C)c1 CUGMNQZFOAZWMR-UHFFFAOYSA-N 0.000 description 1
- XCRFEJSVRVAEBZ-UHFFFAOYSA-N C=CC(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C=C Chemical compound C=CC(=O)OCC(CCC[Si](C)(C)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1)COC(=O)C=C XCRFEJSVRVAEBZ-UHFFFAOYSA-N 0.000 description 1
- GQBOPMCEOFPTAA-UHFFFAOYSA-N C=CC(=O)OCCC[Si](CCCOC(=O)C(=C)CO)(CCCOC(=O)C(=C)CO)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 Chemical compound C=CC(=O)OCCC[Si](CCCOC(=O)C(=C)CO)(CCCOC(=O)C(=C)CO)c1ccc(C2CCC(C3CCC(CCCCC)CC3)CC2)cc1 GQBOPMCEOFPTAA-UHFFFAOYSA-N 0.000 description 1
- KORMYSCDCHFFMN-UHFFFAOYSA-N CCCC1CCC(C2CCC(CC)CC2)CC1 Chemical compound CCCC1CCC(C2CCC(CC)CC2)CC1 KORMYSCDCHFFMN-UHFFFAOYSA-N 0.000 description 1
- CFJCNWQXOICKIF-UHFFFAOYSA-N CCCC1CCC(C2CCC(c3ccc(OCC)c(F)c3F)CC2)CC1 Chemical compound CCCC1CCC(C2CCC(c3ccc(OCC)c(F)c3F)CC2)CC1 CFJCNWQXOICKIF-UHFFFAOYSA-N 0.000 description 1
- IBFAIOMGVHPWRQ-UHFFFAOYSA-N CCCC1CCC(c2ccc(-c3ccc(OCC)c(F)c3F)cc2)CC1 Chemical compound CCCC1CCC(c2ccc(-c3ccc(OCC)c(F)c3F)cc2)CC1 IBFAIOMGVHPWRQ-UHFFFAOYSA-N 0.000 description 1
- BOAHGIPRRKDVQY-UHFFFAOYSA-N CCCC1CCC(c2ccc(OCC)c(F)c2F)CC1 Chemical compound CCCC1CCC(c2ccc(OCC)c(F)c2F)CC1 BOAHGIPRRKDVQY-UHFFFAOYSA-N 0.000 description 1
- QYGWJCMCLQAINT-UHFFFAOYSA-N CCCCC1CCC(C2CCC(CCC)CC2)CC1 Chemical compound CCCCC1CCC(C2CCC(CCC)CC2)CC1 QYGWJCMCLQAINT-UHFFFAOYSA-N 0.000 description 1
- KDVKBFMGDZIYON-UHFFFAOYSA-N CCCCCC1CCC(C2CCC(CCC)CC2)CC1 Chemical compound CCCCCC1CCC(C2CCC(CCC)CC2)CC1 KDVKBFMGDZIYON-UHFFFAOYSA-N 0.000 description 1
- XXORHRTUXUPWLW-UHFFFAOYSA-N CCCCCC1CCC(C2CCC(c3ccc(C)cc3)CC2)CC1 Chemical compound CCCCCC1CCC(C2CCC(c3ccc(C)cc3)CC2)CC1 XXORHRTUXUPWLW-UHFFFAOYSA-N 0.000 description 1
- KYWCNASCWNQZKB-UHFFFAOYSA-N CCCCCC1CCC(C2CCC(c3ccc(O)cc3)CC2)CC1.CCCCCC1CCC(C2CCC(c3ccc(OCCCO)cc3)CC2)CC1 Chemical compound CCCCCC1CCC(C2CCC(c3ccc(O)cc3)CC2)CC1.CCCCCC1CCC(C2CCC(c3ccc(OCCCO)cc3)CC2)CC1 KYWCNASCWNQZKB-UHFFFAOYSA-N 0.000 description 1
- BDODZZFYAJLDRL-UHFFFAOYSA-N CCCCCC1CCC(C2CCC(c3ccc(OCCCO)cc3)CC2)CC1 Chemical compound CCCCCC1CCC(C2CCC(c3ccc(OCCCO)cc3)CC2)CC1 BDODZZFYAJLDRL-UHFFFAOYSA-N 0.000 description 1
- RYPOSVPGRBGCKW-UHFFFAOYSA-N CCCc1ccc(-c2ccc(OCC)c(F)c2F)cc1 Chemical compound CCCc1ccc(-c2ccc(OCC)c(F)c2F)cc1 RYPOSVPGRBGCKW-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
- C09K19/542—Macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
- C09K19/56—Aligning agents
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/133—Constructional arrangements; Operation of liquid crystal cells; Circuit arrangements
- G02F1/1333—Constructional arrangements; Manufacturing methods
- G02F1/1337—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers
- G02F1/133703—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers by introducing organic surfactant additives into the liquid crystal material
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K2019/0444—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit characterized by a linking chain between rings or ring systems, a bridging chain between extensive mesogenic moieties or an end chain group
- C09K2019/0448—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit characterized by a linking chain between rings or ring systems, a bridging chain between extensive mesogenic moieties or an end chain group the end chain group being a polymerizable end group, e.g. -Sp-P or acrylate
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/12—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings at least two benzene rings directly linked, e.g. biphenyls
- C09K2019/121—Compounds containing phenylene-1,4-diyl (-Ph-)
- C09K2019/122—Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3004—Cy-Cy
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3009—Cy-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/301—Cy-Cy-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3016—Cy-Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3027—Compounds comprising 1,4-cyclohexylene and 2,3-difluoro-1,4-phenylene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
- C09K19/542—Macromolecular compounds
- C09K2019/546—Macromolecular compounds creating a polymeric network
Definitions
- the present disclosure relates to an additive, a liquid-crystal composition, and a liquid-crystal display device using the additive.
- Liquid-crystal display devices have been used in various applications, including personal computers, personal digital assistants (PDAs), mobile phones, televisions, and so on, because these devices have many advantages. These advantages include being light in weight, having low power consumption, and not emitting radiation.
- liquid-crystal display device the alignment of liquid-crystal molecules can be achieved with a polyimide film.
- Conventional liquid-crystal alignment layers are generally produced by coating a polyimide onto a substrate to form a film, and then mechanically rubbing it to form the desired liquid-crystal alignment groove on the surface of the polyimide film.
- uneven alignment may occur, or serious brush marks may be produced.
- the product yield of the liquid-crystal display device is not good.
- liquid-crystal display devices having no alignment layer.
- the liquid-crystal composition of such a liquid-crystal display device contains polar compounds, and the liquid-crystal molecules are vertically aligned by the function of the polar compounds.
- polar compounds it is difficult for such polar compounds to be compatible with vertical alignment ability and a high voltage holding ratio.
- stability of the alignment ability of such polar compounds also needs to be improved.
- liquid-crystal composition having excellent vertical alignment ability, a high voltage holding ratio, and a high stability of its alignment ability is still needed in this technical field.
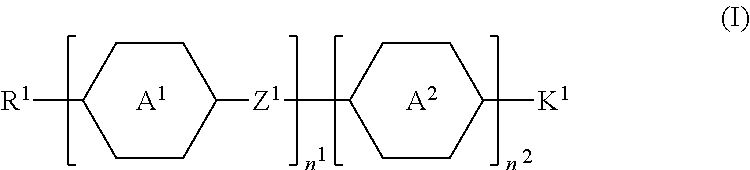
- an additive in one embodiment, includes an additive molecule having a structure represented by formula (I):
- K 2 represents —L 4 —R 5 .
- a liquid-crystal composition in other embodiments of the present disclosure, includes a first component and a second component.
- the first component includes at least one additive as mentioned above, and the second component includes at least one compound represented by formula (II):
- each of R 21 and R 22 independently represents hydrogen, halogen, a C 1 -C 15 alkyl group, or a C 2 -C 15 alkenyl group, wherein the C 1 -C 15 alkyl group or the C 2 -C 15 alkenyl group is unsubstituted or at least one hydrogen atom of the C 1 -C 15 alkyl group or the C 2 -C 15 alkenyl group is substituted by a halogen atom, and/or at least one —CH 2 — of the C 1 -C 15 alkyl group or the C 2 -C 15 alkenyl group is substituted by —O—, and wherein the —O— does not directly bond to another —O—;
- each of B 1 , B 2 , and B 3 independently represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, a 1,3-dioxane-2,5-diyl group, a tetrahydropyran-2,5-diyl group, a divalent dioxabicyclo[2.2.2]octylene group, a divalent trioxabicyclo[2.2.2]octylene group, a tetrahydronaphthalene-2,6-diyl group, or a indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the 1,3-dioxane-2,5-diyl group, the benzofuran-2,5-diyl group, the tetrahydronaphthalene-2,6-diyl group, the
- each of Z 21 and Z 22 independently represents a single bond, a C 1 -C 4 alkylene group, a C 2 -C 4 alkenylene group, or a C 2 -C 4 alkynylene group, wherein the C 1 -C 4 alkylene group, the C 2 -C 4 alkenylene group, or the C 2 -C 4 alkynylene group is unsubstituted or at least one hydrogen atom of the C 1 -C 4 alkylene group, the C 2 -C 4 alkenylene group, or the C 2 -C 4 alkynylene group is substituted by a halogen atom or a —CN group, and/or at least one —CH 2 — of the C 1 -C 4 alkylene group, the C 2 -C 4 alkenylene group, or the C 2 -C 4 alkynylene group is substituted by —O— or —S—, and wherein the —O— does not directly bond to —O—
- n 21 represents 0, 1, or 2 and when n 21 represents 2, the two B 1 groups are identical to each other or different from each other.
- a liquid-crystal display device in other embodiments of the present disclosure, includes a first substrate and a second substrate disposed opposite to the first substrate.
- the liquid-crystal display device also includes a liquid-crystal layer disposed between the first substrate and the second substrate.
- the liquid-crystal layer includes the above-mentioned additive.
- Embodiments of the present disclosure provide an additive, and a liquid-crystal composition and a liquid-crystal display device using the additive, which can greatly improve the vertical alignment property, voltage holding ratio, and stability of the alignment ability of the liquid-crystal composition. Therefore, the performance and durability of the liquid-crystal display device can be improved.
- FIG. is a cross-sectional view showing a liquid-crystal display device in accordance with some embodiments of the present disclosure.
- the term “about” or “approximately” means in a range of 20% of a given value or range, preferably 10%, and more preferably 5%. In the present specification, if there is no specific explanation, a given value or range means an approximate value which may imply the meaning of “about” or “approximately”.
- the two groups when two or more functional groups using the same code are included in one chemical formula, the two groups may be identical to each other or different from each other.
- n 1 when n 1 is 2, two A 1 groups may be identical to each other or different from each other, and two Z 1 groups may also be identical to each other or different from each other.
- the description of “two A 1 groups and Z 1 groups are independently identical to each other or different from each other” may include all the following cases: (1) the two A 1 groups are identical to each other, and the two Z 1 groups are identical to each other; (2) the two A 1 groups are different from each other, but the two Z 1 groups are identical to each other; (3) the two A 1 groups are identical to each other, but the two Z 1 groups are different from each other; and (4) the two A 1 groups are different from each other, and the two Z 1 groups are different from each other.
- the wavy line is used to indicate the atom to which this functional group is bonded to another functional group.
- the wavy line is used to indicate the atom to which this functional group is bonded to another functional group.
- the leftmost oxygen atom is the atom bonded to another functional group.
- an additive is provided.
- the additive can greatly improve the vertical alignment degree, voltage holding ratio, and stability of the alignment ability of the liquid-crystal composition.
- the term “vertical alignment ability of the additive molecule” means the degree of vertical alignment of liquid-crystal molecules in a liquid-crystal composition when the additive is added to the liquid-crystal composition. More specifically, by adding the additive molecule to the liquid-crystal composition, most liquid-crystal molecules can be vertically aligned well without using a conventional alignment film (for example, a polyimide film). Furthermore, the liquid-crystal display device using the additive has a high voltage holding ratio and excellent performance.
- an additive includes an additive molecule having a structure represented by formula (I):
- K 2 represents —L 4 —R 5 .
- the additive includes an additive molecule having a structure represented by formula (I):
- K 2 represents —L 4 —R 5 .
- the structure represented by formula (I) is substantially a rod-shaped structure.
- This rod-shaped structure has a first axial direction and a second axial direction.
- the first axial direction is the long axial direction of the rod-shaped structure, that is, the direction in which the functional group R 1 and the functional group K 1 are connected.
- the second axial direction is the short axial direction of the rod-shaped structure, that is, a direction perpendicular to the first axial direction.
- At least one of the functional groups X 1 , X 2 , X 3 , X 4 , X 5 , and X 6 may include an anchoring group.
- the additive molecules can be fixed to the substrate (for example, the first substrate 110 or the second substrate 120 shown in the FIG.).
- the anchoring group may be a functional group having a higher polarity.
- the anchoring group may generate a bond or a hydrogen bond with a substrate (for example, glass or ITO), and therefore, the additive molecules can be adsorbed (or fixed) on the substrate.
- the anchoring group may include —OH,
- the additive molecules may be fixed on the substrate in such a manner that the first axial direction is perpendicular to the top surface of the substrate.
- the anchoring group is one of X 1 , X 2 , X 3 , X 4 , X 5 , and X 6 that is closest to the right end of the molecule represented by formula (I) such that the first axis is perpendicular to the top surface of the substrate.
- each of the additive molecules has only one anchoring group. Therefore, each of the additive molecules has the same alignment direction on the substrate. In other words, the first axial directions of the different additive molecules are parallel to each other. As a result, the liquid-crystal molecules can be aligned in a uniform state, and defects (for example, local bright spots generated in the dark state) are not easy to generate.
- each of the additive molecules has two anchoring groups. Therefore, it is helpful for the immobilization of the additive molecules on the substrate without being easy to detach. As a result, the occurrence of defects can also be reduced.
- the number of anchoring groups in one additive molecule is at most three.
- the cyclic functional groups may be an aliphatic ring or an aromatic ring.
- the cyclic functional group contributes to the alignment of the liquid-crystal molecules. More specifically, the aromatic cyclic functional group may generate a ⁇ - ⁇ stacking so that the rod-shaped liquid-crystal molecules may be aligned in a specific direction.
- the aliphatic cyclic functional group can align the rod-shaped liquid-crystal molecules in a specific direction by steric hindrance.
- the first axial direction of the additive molecules is perpendicular to the top surface of the substrate, and the long axis of the rod-shaped liquid-crystal molecules is parallel to the first axial direction of the additive molecules. Therefore, the long axis of the rod-shaped liquid-crystal molecules can be made perpendicular to the substrate by the additive molecules. In other words, the vertical alignment of the liquid-crystal molecules can be achieved.
- Some negative ions may remain in the liquid-crystal composition. These ions can cause residual current and reduce the voltage holding ratio during the operation of the display. More specifically, the higher concentration of the negative ions results in the lower the voltage holding ratio. Silicon atoms are more electron-poor than carbon atoms, and therefore, silicon atoms can attract (or capture) the negative ions in the liquid-crystal composition. As a result, the concentration of the negative ions in the liquid-crystal composition can be lowered, and the voltage holding ratio of the liquid-crystal display device can be increased. In other words, in the molecule represented by formula (I), the silicon atom has the function of increasing the voltage holding ratio.
- the silicon atom in the molecule represented by formula (I) is not directly bonded to the oxygen atom.
- the solubility of the molecule represented by formula (I) in the liquid-crystal composition can also be improved.
- At least one of the functional groups X 1 , X 2 , X 3 , X 4 , X 5 , and X 6 may include a polymerizable group.
- the polymerizable group may undergo the polymerization reaction with another polymerizable group by irradiation or heating, and the two polymerizable groups may be bonded to each other.
- the polymerizable group of the additive molecule undergoes the polymerization reaction with the polymerizable group of the adjacent additive molecule. In this way, a plurality of the additive molecules that are perpendicular to the substrate and arranged in parallel with each other can form a network structure.
- the polymerizable group may include an acrylic group, a methacrylic group, or a derivative thereof.
- at least one of the functional groups X 1 , X 2 , X 3 , X 4 , X 5 , and X 6 is a polymerizable group having the following structure or
- Y 1 represents —OH, a C 1 -C 8 alkyl group, or a C 2 -C 8 alkenyl group, and at least one hydrogen atom of the C 1 -C 8 alkyl group or the C 2 -C 8 alkenyl group is substituted by —OH; and Y 2 represents hydrogen, halogen, a C 1 -C 8 alkyl group, or a C 2 -C 8 alkenyl group, wherein the C 1 -C 8 alkyl group or the C 2 -C 8 alkenyl group is unsubstituted or at least one hydrogen atom of the C 1 -C 8 alkyl group or the C 2 -C 8 alkenyl group is substituted by a halogen atom.
- the additive includes the first additive molecule, and the first additive molecule has a structure represented by formula (I):
- Y 1 and Y 2 are respectively the same as the definitions of Y 1 and Y 2 defined in this paragraph;
- the additive includes the second additive molecule, and the second additive molecule has a structure represented by formula (I):
- Y 1 and Y 2 are respectively the same as the definitions of Y 1 and Y 2 defined in this paragraph;
- Specific exemplary first additive molecules are shown in Table 1 below.
- Specific exemplary second additive molecules are shown in Table 2 below.
- the functional group at the end of the molecular main chain includes a polymerizable group and an anchoring group. Therefore, the first additive molecule can be adsorbed (or fixed) on the substrate in such a manner that the first axial direction is perpendicular to the top surface of the substrate, thereby obtaining good vertical alignment ability. Furthermore, a plurality of first additive molecules that are perpendicular to the substrate and arranged in parallel with each other can form the above-mentioned network structure. This network structure can further improve the degree of vertical alignment of the liquid-crystal molecules and improve the stability of the alignment ability.
- the molecular side chain includes at least one polymerizable group.
- the second additive molecule may form the above-mentioned network structure with the adjacent second additive molecule or the first additive molecule. As a result, the stability of the alignment ability can be improved.
- the additive includes a first additive molecule. Therefore, this additive can have excellent vertical alignment ability and good stability of the alignment ability.
- the additive includes a second additive molecule.
- the additive includes a first additive molecule and a second additive molecule. In such embodiments, the first additive molecules may be vertically fixed on the substrate, and the first additive molecules may be bonded to adjacent first additive molecules or second additive molecules to form the above-mentioned network structure. Therefore, this additive can have excellent vertical alignment ability and excellent stability of the alignment ability.
- the additive includes a first additive molecule and a second additive molecule, and when the amount of the first additive molecule is set to 1 part by weight, the amount of the second additive molecule is 0.1-60 parts by weight. In other embodiments, when the amount of the first additive molecule is set to 1 part by weight, the amount of the second additive molecule is 0.5-50 parts by weight. In still other embodiments, when the amount of the first additive molecule is set to 1 part by weight, the amount of the second additive molecule is 1-30 parts by weight.
- a liquid-crystal composition in other embodiments, includes a first component and a second component.
- the first component includes at least one additive as mentioned above, and the second component includes at least one compound represented by formula (II):
- the above-mentioned second component includes at least one compound represented by formula (II-1) or formula (II-2):
- the compound of formula (II-2) includes at least one phenylene group, and two hydrogen atoms on the same side of this phenylene group are substituted by fluorine atoms.
- the compound of formula (II-2) can be used to adjust the dielectric anisotropy ( ⁇ ) of the liquid-crystal composition.
- the above-mentioned liquid-crystal composition further includes a third component, and the third component includes at least one compound represented by formula (III), formula (IV), or formula (V):
- the compound of the third component includes at least one polymerizable group, and the polymerizable group may include an acrylic group, a methacrylic group, or a derivative thereof. More specifically, each of the compounds represented by formula (IV) and formula (V) has a polymerizable group at one end of the molecule. Each of the compounds represented by formula (III) has a polymerizable group at both ends of the molecule.
- the polymerizable group of the third component may undergo the polymerization reaction with another polymerizable group of the above-mentioned additive molecules represented by formula (I). In this way, it is helpful to form the above-mentioned network, and the degree of vertical alignment of the liquid-crystal molecules can be further improved.
- the degree of vertical alignment and the voltage holding ratio of the liquid-crystal composition may not be effectively improved.
- the content of the first component is too high, the first component may not dissolve well in the liquid-crystal composition, and it may precipitate. Such a liquid-crystal composition cannot be used.
- the first component exceeds the specific content, even if the first component is further increased, the degree of vertical alignment of the liquid-crystal composition cannot be further improved.
- the first component has the group (for example, an anchoring group) having a relatively high polarity. Therefore, if the content of the first component is too high, the voltage holding ratio of the liquid-crystal composition may be lowered.
- the content of the first component may be controlled within an appropriate range.
- the liquid-crystal composition includes a first component and a second component.
- the first component when the total weight of the second component is 100 parts by weight, the first component is 0.01-10 parts by weight. In other embodiments, when the total weight of the second component is 100 parts by weight, the first component is 0.05-5 parts by weight. In still other embodiments, when the total weight of the second component is 100 parts by weight, the first component is 0.1-3 parts by weight.
- the liquid-crystal composition includes a first component, a second component, and a third component.
- the first component when the total weight of the second component is 100 parts by weight, the first component is 0.01-10 parts by weight, and the third component is 0.01-10 parts by weight.
- the first component when the total weight of the second component is 100 parts by weight, the first component is 0.04-3.5 parts by weight, and the third component is 0.04-3.5 parts by weight.
- the first component is 0.07-2 parts by weight
- the third component is 0.07-2 parts by weight.
- liquid-crystal composition may further include other components other than the above-mentioned first component, second component, and third component.
- other conventional liquid-crystal compounds or other additives in appropriate amounts.
- the above-mentioned other additives may include, for example, chiral dopants, UV stabilizers, antioxidants, free radical scavengers, nanoparticles, and so on.
- FIG. is a cross-sectional view showing a liquid-crystal display device 100 in accordance with some embodiments of the present disclosure.
- the liquid-crystal display device 100 includes a first substrate 110 and a second substrate 120 disposed opposite to the first substrate 110 .
- the liquid-crystal display device 100 also includes a liquid-crystal layer 130 disposed between the first substrate 110 and the second substrate 120 .
- the first substrate 110 and the second substrate 120 are respectively a conventional thin film transistor substrate and a conventional color filter substrate. In order to simplify the description, the materials, structures, and manufacturing methods of the first substrate 110 and the second substrate 120 will not be described in detail herein.
- the liquid-crystal layer 130 of the liquid-crystal display device 100 of the present disclosure uses the above-mentioned liquid-crystal composition, and the liquid-crystal composition includes the additive represented by formula (I).
- the additive represented by the formula (I) can greatly improve the degree of vertical alignment, the voltage holding ratio, and the stability of the alignment ability of the liquid-crystal composition.
- the liquid-crystal molecules can be vertically aligned well without using a conventional alignment film.
- the liquid-crystal display device 100 using the additive of the present disclosure has the advantages of high voltage holding ratio and good durability.
- the liquid-crystal composition of the present disclosure can be applied to all kinds of liquid-crystal display devices.
- Lithium aluminum hydride (LAH, 2.66 g, 70.1 mmol) and anhydrous tetrahydrofuran (160 mL) were placed in a 500 mL reaction flask and stirred to dissolve. Then, Compound 3 (20.0 g, 35.0 mmol) was slowly added to the reaction flask at 0° C., and the reaction was carried out for 1 hour. Next, the reaction was carried out at room temperature (20-30° C.) for 6 hours. After the reaction was completed, water was added. Then, an extraction was performed by using ethyl acetate and water, and the organic phase was collected. The solvent of the collected organic phase was removed by using a rotary concentrator. Then, column chromatography was performed to obtain Compound 4.
- LAH Lithium aluminum hydride
- anhydrous tetrahydrofuran 160 mL
- the additive molecule Exp-B2 (1.5 g, 2.5 mmol), paraformaldehyde (0.68 g, 22.7 mmol), 1,4-diazabicyclo[2.2.2]octane (1.1 g, 10.0 mmol), water (15 mL), and tetrahydrofuran (30 mL) were placed in a 100 mL reaction flask and stirred to dissolve. Then, the reaction was carried out at 70° C. for 8 hours. After the reaction was completed, water was added. Then, an extraction was performed by using ethyl acetate and water, and the organic phase was collected. The solvent of the collected organic phase was removed by using a rotary concentrator. Then, column chromatography was performed to obtain the additive molecule Exp-A2.
- allylmagnesium chloride (concentration: 2.0M, solvent: tetrahydrofuran; 100.0 mL, 200.0 mmol) was added, and the mixture was heated (100° C.) to reflux for 8 hours to carry out the reaction. After the reaction was completed, water was added. Then, an extraction was performed by using ethyl acetate and water, and the organic phase was collected. The solvent of the collected organic phase was removed by using a rotary concentrator. Then, column chromatography was performed to obtain Compound 10.
- the additive molecule Ref can be synthesized in accordance with the procedure for synthesizing Compound 15 by using Compound 13 as starting material, and the synthesis step is as shown in the above flow chart.
- liquid-crystal compositions were prepared by forming the mother liquid formed by 100 parts by weight of the molecules of formula (II) in accordance with Table 3, and then, 0.3 parts by weight of molecule of formula (III) shown in Table 3 was added. Next, the additive molecules shown in Tables 1, 2, and 4 were additionally added to the above liquid-crystal composition in accordance with the amount that was added, shown in Table 5.
- the liquid-crystal composition of Example 6 is 100 parts by weight of the mother liquid formed by the molecules of formula (II) shown in Table 3, and 0.3 parts by weight of the molecule of formula (III), 0.2 parts by weight of the additive molecule Exp-A2, and 0.5 parts by weight of the additive molecule Exp-B2 is further added. Furthermore, in the liquid-crystal compositions of Reference Example and Examples 1-5 shown in Table 5, only one kind of the additive molecules shown in Tables 1, 2, and 4 was added.
- the liquid-crystal display device was disposed in a polarizing microscope in which a polarizing element and an analyzer were arranged orthogonally.
- the element was irradiated with light from below, and the presence or absence of light leakage was observed to judge the vertical alignment.
- the experimental results of vertical alignment were shown in Table 6.
- a DC voltage (charge voltage of 5 V, operating frequency of 0.6 Hz) was applied to the liquid-crystal display of the Example or the Reference Example at an ambient temperature of 60° C.
- the voltage value V2 after the application was released to 1.667 sec was measured by the liquid-crystal physical parameter measuring instrument (product number: ALCT-IV1, manufactured by INSTEC Co., Ltd.).
- the experimental results of voltage holding ratio were shown in Table 6.
- the formula for calculating the voltage holding ratio (VHR) of the liquid-crystal display device is as follows: VHR ⁇ (V2/applied voltage value) ⁇ 100%.
- a voltage of 0V to 10V was continuously applied to the liquid-crystal display device, and the light transmittance of the liquid-crystal display device corresponding to different voltages was measured. The light transmittance was recorded every 0.1V, and a graph of voltage versus transmittance was drawn based on the experimental results.
- the voltage corresponding to a liquid-crystal display device having a transmittance of 1% is defined as V(th,i).
- a deterioration experiment was performed on the liquid-crystal display device. The steps of the deterioration experiment are as follows. The liquid-crystal display device was placed in an oven (temperature: 60° C.), and a voltage (AC, 10 V) was applied for a duration of 4 days.
- the liquid-crystal display device was subjected to the experiment described above, and a graph of voltage versus transmittance was drawn.
- the voltage corresponding to a liquid-crystal display device having a transmittance of 1% is defined as V(th,f).
- the AVth value (unit: Volts) of the liquid-crystal display device is calculated according to the following formula. The smaller the absolute value of AVth, the better the stability of the alignment ability of the additive molecules.
- the Reference Example includes the additive molecule Ref Because the additive molecule Ref does not have the silicon atom and the polymerizable group, the voltage holding ratio of the liquid-crystal composition of the Reference Example is not good. Furthermore, because the additive molecule Ref does not have the polymerizable group, the stability of the vertical alignment ability of the liquid-crystal composition of the Reference Example is not good.
- each of Examples 1-12 includes at least one additive molecule represented by formula (I).
- the results show that the liquid-crystal molecules of Examples 1-12 are all vertically aligned, and the voltage holding ratio (for example, the voltage holding ratio greater than 80%) and the stability of the vertical alignment ability are significantly improved.
- each of Examples 1-4 includes one of the first additive molecules, and all of the first additive molecules include both the anchoring group and the polymerizable group.
- the results show that the liquid-crystal molecules of Examples 1-4 are all vertically aligned.
- the liquid-crystal molecules can be vertically aligned well in the condition that only a small amount of the additive molecules (for example, the amount that was added is less than 0.5 parts by weight) is added. Accordingly, the first additive molecule can significantly improve the degree of vertical alignment of the liquid-crystal molecules, and can also significantly improve the voltage holding ratio.
- Example 5 includes one of the second additive molecules, and the second additive molecule includes two polymerizable groups at the side chain and/or main chain of the molecule.
- the results show that the liquid-crystal molecules of Example 5 are vertically aligned, and the stability of their alignment ability is excellent. Accordingly, the second additive molecule can significantly improve the degree of stability of the vertical alignment ability of the liquid-crystal molecules.
- each of Examples 6-9 includes one of the first additive molecules and one of the second additive molecules, and all of the second additive molecules include two polymerizable groups at the side chain and/or main chain of the molecule.
- the results show that the liquid-crystal molecules of Examples 6-9 are all vertically aligned, and the stability of their alignment ability is excellent.
- the total amount of the first additive molecule and the second additive molecule added is 0.7 parts by weight.
- the voltage holding ratio is 98.4%, and the A Vth value is ⁇ 0.022 V. Accordingly, the voltage holding ratio and the stability of the alignment ability can be further improved by simultaneously using the first additive molecule and the second additive molecule.
- each of Examples 10-12 includes one of the first additive molecules and the compound represented by formula (V), and the compound represented by formula (V) includes one polymerizable group at the end of the molecule.
- the results show that the liquid-crystal molecules of Examples 10-12 are all vertically aligned, and the stability of their alignment ability is excellent.
- the total amount of the first additive molecule that is added and the compound represented by formula (V) is 0.7 parts by weight.
- the voltage holding ratio is 98.0%, and the ⁇ Vth value is ⁇ 0.010 V.
- the voltage holding ratio is 96.9%, and the ⁇ Vth value is ⁇ 0.050 V. Accordingly, the voltage holding ratio and the stability of the alignment ability can also be further improved by simultaneously using the first additive molecule and the compound represented by formula (V) having the polymerizable group.
- the additive molecule of the present disclosure can have excellent vertical alignment ability while having a high voltage holding ratio.
- the liquid-crystal composition By adding the above-mentioned additive molecule to the liquid-crystal composition, it is possible to realize a state in which most of the liquid-crystal molecules are vertically aligned well without using a conventional alignment film.
- the content of the first additive molecule and/or the second additive molecule may be adjusted.
- the liquid-crystal display device using the additive molecules of the embodiments of the present disclosure can have a high voltage retention rate (for example, the voltage retention rate greater than 80%) and excellent stability of the alignment ability. As a result, the performance and durability of the liquid-crystal display device can be significantly improved.
Landscapes
- Chemical & Material Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Nonlinear Science (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Mathematical Physics (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Liquid Crystal Substances (AREA)
- Liquid Crystal (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
-
- wherein, in formula (I),
- R1 represents fluorine, chlorine, hydrogen, a C1-C20 linear alkyl group, a C3-C20 branched alkyl group, a C1-C20 linear alkoxy group, or a C3-C20 branched alkoxy group, wherein the C1-C20 linear alkyl group, the C3-C20 branched alkyl group, the C1-C20 linear alkoxy group, or the C3-C20 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C20 linear alkyl group, the C3-C20 branched alkyl group, the C1-C20 linear alkoxy group, or the C3-C20 branched alkoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C20 linear alkyl group, the C3-C20 branched alkyl group, the C1-C20 linear alkoxy group, or the C3-C20 branched alkoxy group is substituted by a halogen atom;
- A1 represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, a 1,3-dioxane-2,5-diyl group, a tetrahydropyran-2,5-diyl group, a divalent dioxabicyclo[2.2.2]octylene group, a divalent trioxabicyclo[2.2.2]octylene group, a tetrahydronaphthalene-2,6-diyl group, a 1,3-cyclopentylene, a 1,3-cyclobutylene, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the tetrahydronaphthalene-2,6-diyl group, the 1,3-cyclopentylene, the 1,3-cyclobutylene, or the indane-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the tetrahydronaphthalene-2,6-diyl group, the 1,3-cyclopentylene, the 1,3-cyclobutylene, or the indane-2,5-diyl group is substituted by a halogen atom, —CH3, —CH2CH3, or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the tetrahydronaphthalene-2,6-diyl group, the 1,3-cyclopentylene, the 1,3-cyclobutylene, or the indane-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- A2 represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by a halogen atom, —CH3, —CH2CH3, or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- Z1 represents a single bond, a C1-C15 alkylene group, a C1-C15 alkyleneoxy group, —C≡C—, —CH═CH—, —CF2O—, —OCF2—, —COO—, —OCO—, or —OOC—, and —CF2—CF2—, or —CF═CF—;
- n1 represents 1, 2, or 3, and when n1 represents 2 or 3, the two or more A1 groups are identical to each other or different from each other, and the two or more Z1 groups are identical to each other or different from each other;
- n2 represents 0 or 1; and
- K1 represents a structure represented by formula (Ia), formula (Ib), or formula (Ic):
-
- wherein, in formula (Ia), formula (Ib), and formula (Ic),
- each of L1, L2, and L3 independently represents a single bond, a C1-C15 linear alkylene group, a C3-C15 branched alkylene group, a C1-C15 linear alkyleneoxy group, or a C3-C15 branched alkyleneoxy group, wherein the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C5 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —SiRa 2—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by a halogen atom, and wherein Ra represents a C1-C10 linear alkyl group or a C3-C10 branched alkyl group, and two Ra groups bonded to the same Si atom are identical to each other or different from each other;
- each of R2, R3 and R4 independently represents fluorine, chlorine, hydrogen, a C1-C10 linear alkyl group, a C3-C10 branched alkyl group, a C1-C10 linear alkoxy group, or a C3-C10 branched alkoxy group, wherein the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by a halogen atom;
- each of X1 and X2 independently represents or
-
- wherein Y1 represents —OH, a C1-C15 alkyl group, or a C2-C15 alkenyl group, and at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C15 alkyl group, or a C2-C15 alkenyl group, wherein the C1-C15 alkyl group or the C2-C15 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by a halogen atom;
- Z2 represents a single bond, a C1-C15 alkylene group, a C1-C15 alkyleneoxy group, —C≡C—, —CH═CH—, —CF2O—, —OCF2—, —COO—, —OCO—, —OOC—, —CF2—CF2—, or —CF═CF—;
- A3 represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by a halogen atom, —CH3, —CH2CH3, or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- n3+n4=3, n4 represents 2 or 3, and the two or more L2 groups are identical to each other or different from each other, and the two or more X1 groups are identical to each other or different from each other;
- n5+n6=3, n6 represents 2 or 3, and the two or more L2 groups are identical to each other or different from each other, and the two or more X2 groups are identical to each other or different from each other;
- n7 represents 0, 1, or 2, and when n7 represents 2, the two K3 groups are identical to each other or different from each other;
- n8 represents 1 or 2, and when n8 represents 2, the two Z2 groups are identical to each other or different from each other, the two A3 groups are identical to each other or different from each other, and the two K3 groups are identical to each other or different from each other;
- when n8 represents 1, n7 represents 2;
- when n8 represents 2, n7 in the A3 group directly bonded to the K2 group represents 2, and n7 in the A3 group directly bonded to the Z2 group represents 0, 1, or 2; and
- K2 represents a structure represented by formula (Id), formula (Ie), or formula (If):
-
- K3 represents a structure represented by formula (Ig) or formula (Ih):
-
- wherein, in formula (Id), formula (Ie), formula (If), formula (Ig), and formula (Ih),
- each of L1, L2, L3, L4, L5, L6, and L7 independently represents a single bond, a C1-C15 linear alkylene group, a C3-C15 branched alkylene group, a C1-C15 linear alkyleneoxy group, or a C3-C15 branched alkyleneoxy group, wherein the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —SiRa 2—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by a halogen atom, and wherein Ra represents a C1-C10 linear alkyl group or a C3-C10 branched alkyl group, and two Ra groups bonded to the same Si atom are identical to each other or different from each other;
- each of R2, R3, R4, R5 and R6 independently represents fluorine, chlorine, hydrogen, a C1-C10 linear alkyl group, a C3-C10 branched alkyl group, a C1-C10 linear alkoxy group, or a C3-C10 branched alkoxy group, wherein the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, —OOC—, and/or at least one hydrogen atom of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by a halogen atom;
- each of X3, X4, X5 and X6 independently represents hydrogen, —OH,
-
- wherein Y1 represents —OH, a C1-C15 alkyl group, or a C2-C15 alkenyl group, and at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C15 alkyl group, or a C2-C15 alkenyl group, wherein the C1-C15 alkyl group or the C2-C15 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by a halogen atom;
- n9+n10=3, n10 represents 2 or 3, and the two or more L7 groups are identical to each other or different from each other, and the two or more X4 groups are identical to each other or different from each other;
- n12 represents 1, 2, or 3, n11+n12=3, and when n12 represents 2 or 3, the two or more L2 groups are identical to each other or different from each other, and the two or more X5 groups are identical to each other or different from each other; when n11 represents 2, the two R2 groups are identical to each other or different from each other; and
- n14 represents 1, 2, or 3, n13+n14=3, and when n14 represents 2 or 3, the two or more L2 groups are identical to each other or different from each other, and the two or more X6 groups are identical to each other or different from each other; when n13 represents 2, the two R4 groups are identical to each other or different from each other; and
- when X5 or X6 represents,
-
- wherein, in formula (I),
- R1 represents fluorine, chlorine, hydrogen, a C1-C20 linear alkyl group, a C3-C20 branched alkyl group, a C1-C20 linear alkoxy group, or a C3-C20 branched alkoxy group, wherein the C1-C20 linear alkyl group, the C3-C20 branched alkyl group, the C1-C20 linear alkoxy group, or the C3-C20 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C20 linear alkyl group, the C3-C20 branched alkyl group, the C1-C20 linear alkoxy group, or the C3-C20 branched alkoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C20 linear alkyl group, the C3-C20 branched alkyl group, the C1-C20 linear alkoxy group, or the C3-C20 branched alkoxy group is substituted by a halogen atom;
- A1 represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, a 1,3-dioxane-2,5-diyl group, a tetrahydropyran-2,5-diyl group, a divalent dioxabicyclo[2.2.2]octylene group, a divalent trioxabicyclo[2.2.2]octylene group, a tetrahydronaphthalene-2,6-diyl group, a 1,3-cyclopentylene, a 1,3-cyclobutylene, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the tetrahydronaphthalene-2,6-diyl group, the 1,3-cyclopentylene, the 1,3-cyclobutylene, or the indane-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the tetrahydronaphthalene-2,6-diyl group, the 1,3-cyclopentylene, the 1,3-cyclobutylene, or the indane-2,5-diyl group is substituted by a halogen atom, —CH3, —CH2CH3, or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the tetrahydronaphthalene-2,6-diyl group, the 1,3-cyclopentylene, the 1,3-cyclobutylene, or the indane-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- A2 represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by a halogen atom, —CH3, —CH2CH3, or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- Z1 represents a single bond, a C1-C15 alkylene group, a C1-C15 alkyleneoxy group, —C≡C—, —CH═CH—, —CF2O—, —OCF2—, —COO—, —OCO—, —OOC—, —CF2—CF2—, or —CF═CF—;
- n1 represents 1, 2, or 3, and when n1 represents 2 or 3, the two or more A1 groups are identical to each other or different from each other, and the two or more Z1 groups are identical to each other or different from each other;
- n2 represents 0 or 1; and
- K1 represents a structure represented by formula (Ia), formula (Ib), or formula (Ic):
-
- wherein, in formula (Ia), formula (Ib), and formula (Ic),
- each of L1, L2, and L3 independently represents a single bond, a C1-C15 linear alkylene group, a C3-C15 branched alkylene group, a C1-C15 linear alkyleneoxy group, or a C3-C15 branched alkyleneoxy group, wherein the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —SiRa 2—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by a halogen atom, and wherein Ra represents a C1-C10 linear alkyl group or a C3-C10 branched alkyl group, and two Ra groups bonded to the same Si atom are identical to each other or different from each other;
- each of R2, R3 and R4 independently represents fluorine, chlorine, hydrogen, a C1-C10 linear alkyl group, a C3-C10 branched alkyl group, a C1-C10 linear alkoxy group, or a C3-C10 branched alkoxy group, wherein the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by a halogen atom;
- each of X1 and X2 independently represents
-
- wherein Y1 represents —OH, a C1-C15 alkyl group, or a C2-C15 alkenyl group, and at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C15 alkyl group, or a C2-C15 alkenyl group, wherein the C1-C15 alkyl group or the C2-C15 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by a halogen atom;
- Z2 represents a single bond, a C1-C15 alkylene group, a C1-C15 alkyleneoxy group, —C≡C—, —CH═CH—, —CF2O—, —OCF2—, —COO—, —OCO—, —OOC—, —CF2—CF2—, or —CF═CF—;
- A3 represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by a halogen atom, —CH3, —CH2CH3, or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, or the indane-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- n3+n4=3, n4 represents 2 or 3, and the two or more L2 groups are identical to each other or different from each other, and the two or more X1 groups are identical to each other or different from each other;
- n5+n6=3, n6 represents 2 or 3, and the two or more L2 groups are identical to each other or different from each other, and the two or more X2 groups are identical to each other or different from each other;
- n7 represents 0, 1, or 2, and when n7 represents 2, the two K3 groups are identical to each other or different from each other;
- n8 represents 1 or 2, and when n8 represents 2, the two Z2 groups are identical to each other or different from each other, the two A3 groups are identical to each other or different from each other, and the two K3 groups are identical to each other or different from each other;
- when n8 represents 1, n7 represents 2;
- when n8 represents 2, n7 in the A3 group directly bonded to the K2 group represents 2, and n7 in the A3 group directly bonded to the Z2 group represents 0, 1, or 2; and
- K2 represents a structure represented by formula (Id), formula (Ie), or formula (If):
-
- K3 represents a structure represented by formula (Ig) or formula (Ih):
-
- wherein, in formula (Id), formula (Ie), formula (If), formula (Ig), and formula (Ih),
- each of L1, L2, L3, L4, L5, L6, and L7 independently represents a single bond, a C1-C15 linear alkylene group, a C3-C15 branched alkylene group, a C1-C15 linear alkyleneoxy group, or a C3-C15 branched alkyleneoxy group, wherein the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —SiRa 2—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C1-C15 linear alkyleneoxy group, or the C3-C15 branched alkyleneoxy group is substituted by a halogen atom, and wherein Ra represents a C1-C10 linear alkyl group or a C3-C10 branched alkyl group, and two Ra groups bonded to the same Si atom are identical to each other or different from each other;
- each of R2, R3, R4, R5 and R6 independently represents fluorine, chlorine, hydrogen, a C1-C10 linear alkyl group, a C3-C10 branched alkyl group, a C1-C10 linear alkoxy group, or a C3-C10 branched alkoxy group, wherein the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by —C≡C—, —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C10 linear alkyl group, the C3-C10 branched alkyl group, the C1-C10 linear alkoxy group, or the C3-C10 branched alkoxy group is substituted by a halogen atom;
- each of X3, X4, X5 and X6 independently represents hydrogen, —OH,
-
- wherein Y1 represents —OH, a C1-C15 alkyl group, or a C2-C15 alkenyl group, and at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C15 alkyl group, or a C2-C15 alkenyl group, wherein the C1-C15 alkyl group or the C2-C15 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by a halogen atom;
- n9+n10=3, n10 represents 2 or 3, and the two or more L7 groups are identical to each other or different from each other, and the two or more X4 groups are identical to each other or different from each other;
- n12 represents 1, 2, or 3, n11+n12=3, and when n12 represents 2 or 3, the two or more L2 groups are identical to each other or different from each other, and the two or more X5 groups are identical to each other or different from each other; when n11 represents 2, the two R2 groups are identical to each other or different from each other; and
- n14 represents 1, 2, or 3, n13+n14=3, and when n14 represents 2 or 3, the two or more L2 groups are identical to each other or different from each other, and the two or more X6 groups are identical to each other or different from each other; when n13 represents 2, the two R4 groups are identical to each other or different from each other; and
- when X5 or X6 represents
-
- wherein, in formula (I),
- R1 represents fluorine, chlorine, hydrogen, a C1-C15 linear alkyl group, a C3-C15 branched alkyl group, a C1-C15 linear alkoxy group, or a C3-C15 branched alkoxy group, wherein the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is substituted by a halogen atom;
- the definitions of A1, A2, Z1, n1, and n2 are respectively the same as the definitions of A, A2, Z1, n1, and n2 defined in the previous paragraphs; and
- K1 represents a structure represented by formula (I′a), formula (I′b), or formula (I′c):
-
- wherein, in formula (I′a), formula (I′b), and formula (I′c),
- the definitions of L2, L3, n3, n4, n5, and n6 are respectively the same as the definitions of L2, L3, n3, n4, n5, and n6 defined in the previous paragraphs;
- each of R2, R3 and R4 independently represents fluorine, chlorine, hydrogen, a C1-C8 linear alkyl group, a C3-C8 branched alkyl group, a C1-C8 linear alkoxy group, or a C3-C8 branched alkoxy group, wherein the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by a halogen atom;
-
- each of X1 and X2 independently represents or;
- wherein Y1 represents —OH, a C1-C8 alkyl group, or a C2-C8 alkenyl group, and at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C8 alkyl group, or a C2-C8 alkenyl group, wherein the C1-C8 alkyl group or the C2-C8 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by a halogen atom;
- K2 represents a structure represented by formula (Id), formula (Ie), or formula (If):
-
- K3 represents a structure represented by formula (Ig) or formula (Ih):
-
- wherein, in formula (Id), formula (Ie), formula (If), formula (Ig), and formula (Ih),
- the definitions of L1, L2, L3, L4, L5, L6, L7, n9, n10, n11, n12, n13, and n14 are respectively the same as the definitions of L1, L2, L3, L4, L5, L6, L7, n9, n10, n11, n12, n13, and n14 defined in the previous paragraphs;
- each of R2, R3, R4, R5 and R6 independently represents fluorine, chlorine, hydrogen, a C1-C8 linear alkyl group, a C3-C8 branched alkyl group, a C1-C8 linear alkoxy group, or a C3-C8 branched alkoxy group, wherein the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by a halogen atom;
- each of X3, X4, X5 and X6 independently represents hydrogen, —OH,
-
- wherein Y1 represents —OH, a C1-C8 alkyl group, or a C2-C8 alkenyl group, and at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C8 alkyl group, or a C2-C8 alkenyl group, wherein the C1-C8 alkyl group or the C2-C8 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by a halogen atom;
- wherein at least one of X3, X4, X5 and X6 represents
-
- when X5 or X6 represents
and wherein Y1 represents —OH, a C1-C8 alkyl group, or a C2-C8 alkenyl group, and at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by —OH; and Y2 represents hydrogen, halogen, a C1-C8 alkyl group, or a C2-C8 alkenyl group, wherein the C1-C8 alkyl group or the C2-C8 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by a halogen atom.
-
- wherein, in formula (I),
- R1 represents fluorine, chlorine, hydrogen, a C1-C15 linear alkyl group, a C3-C15 branched alkyl group, a C1-C15 linear alkoxy group, or a C3-C15 branched alkoxy group, wherein the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is substituted by a halogen atom;
- the definitions of A1, A2, Z1, n1, and n2 are respectively the same as the definitions of A, A2, Z1, n1, and n2 defined in the previous paragraphs; and
- K1 represents a structure represented by formula (I′a), formula (I′b), or formula (I′c):
-
- wherein, in formula (I′a), formula (I′b), and formula (I′c),
- the definitions of L2, L3, n3, n4, n5, and n6 are respectively the same as the definitions of L2, L3, n3, n4, n5, and n6 defined in the previous paragraphs;
- each of R2, R3, and R4 independently represents fluorine, chlorine, hydrogen, a C1-C8 linear alkyl group, a C3-C8 branched alkyl group, a C1-C8 linear alkoxy group, or a C3-C8 branched alkoxy group, wherein the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by a halogen atom;
- each of X1 and X2 independently represents
-
- wherein Y1 represents —OH, a C1-C8 alkyl group, or a C2-C8 alkenyl group, and at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C8 alkyl group, or a C2-C8 alkenyl group, wherein the C1-C8 alkyl group or the C2-C8 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by a halogen atom;
- K2 represents a structure represented by formula (Id), formula (Ie), or formula (If):
-
- K3 represents a structure represented by formula (Ig) or formula (Ih):
-
- wherein, in formula (Id), formula (Ie), formula (If), formula (Ig), and formula (Ih),
- the definitions of L1, L2, L3, L4, L5, L6, L, n9, n10, n11, n12, n13, and n14 are respectively the same as the definitions of L1, L2, L3, L4, L5, L6, L7, n9, n10, n11, n12, n13, and n14 defined in the previous paragraphs;
- each of R2, R3, R4, R5 and R6 independently represents fluorine, chlorine, hydrogen, a C1-C8 linear alkyl group, a C3-C8 branched alkyl group, a C1-C8 linear alkoxy group, or a C3-C8 branched alkoxy group, wherein the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C5 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by a halogen atom;
- each of X3, X4, X5 and X6 independently represents hydrogen, —OH,
and the definitions of Y1 and Y2 are respectively the same as the definitions of Y1 and Y2 defined in this paragraph;
-
- when K1 represents the structure represented by formula (I′a) or formula (I′b), at least one of X1 and X2 represents
-
- when K1 represents the structure represented by formula (I′c), and K2 represents the structure represented by formula (Id), at least one of X5 and X6 represents
-
- when K1 represents the structure represented by formula (I′c), and K2 represents the structure represented by formula (Ie) or formula (If), at least one of X3, X4, X5 and X6 represents
-
- wherein, in formula (I),
- R1 represents fluorine, chlorine, hydrogen, a C1-C15 linear alkyl group, a C3-C15 branched alkyl group, a C1-C15 linear alkoxy group, or a C3-C15 branched alkoxy group, wherein the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C15 linear alkyl group, the C3-C15 branched alkyl group, the C1-C15 linear alkoxy group, or the C3-C15 branched alkoxy group is substituted by a halogen atom;
- the definitions of A1, A2, Z1, n1, and n2 are respectively the same as the definitions of A, A2, Z1, n1, and n2 defined in the previous paragraphs; and
- K1 represents a structure represented by formula (I′a), formula (I′b), or formula (I′c):
-
- wherein, in formula (I′a), formula (I′b), and formula (I′c),
- the definitions of L2, L3, n3, n4, n5, and n6 are respectively the same as the definitions of L2, L3, n3, n4, n5, and n6 defined in the previous paragraphs;
- each of R2, R3, and R4 independently represents fluorine, chlorine, hydrogen, a C1-C8 linear alkyl group, a C3-C8 branched alkyl group, a C1-C8 linear alkoxy group, or a C3-C8 branched alkoxy group, wherein the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by a halogen atom;
- each of X1 and X2 independently represents
-
- wherein Y1 represents —OH, a C1-C8 alkyl group, or a C2-C8 alkenyl group, and at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by —OH;
- Y2 represents hydrogen, halogen, a C1-C8 alkyl group, or a C2-C8 alkenyl group, wherein the C1-C8 alkyl group or the C2-C8 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C8 alkyl group or the C2-C8 alkenyl group is substituted by a halogen atom;
- K2 represents a structure represented by formula (Id), formula (Ie), or formula (If):
-
- K3 represents a structure represented by formula (Ig) or formula (Ih):
-
- wherein, in formula (Id), formula (Ie), formula (If), formula (Ig), and formula (Ih),
- the definitions of L1, L2, L3, L4, L5, L6, L7, n9, n10, n11, n12, n13, and n14 are respectively the same as the definitions of L1, L2, L3, L4, L5, L6, L7, n9, n10, n11, n12, n13 and n14 defined in the previous paragraphs;
- each of R2, R3, R4, R5 and R6 independently represents fluorine, chlorine, hydrogen, a C1-C8 linear alkyl group, a C3-C8 branched alkyl group, a C1-C8 linear alkoxy group, or a C3-C8 branched alkoxy group, wherein the C1-C8 linear alkyl group, the C3-C5 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is unsubstituted or at least one —CH2— of the C1-C8 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by —CH═CH—, —CF2O—, —O—, —COO—, —OCO—, or —OOC—, and/or at least one hydrogen atom of the C1-C5 linear alkyl group, the C3-C8 branched alkyl group, the C1-C8 linear alkoxy group, or the C3-C8 branched alkoxy group is substituted by a halogen atom;
- each of X3, X4, X5 and X6 independently represents hydrogen, —OH,
and the definitions of Y1 and Y2 are respectively the same as the definitions of Y1 and Y2 defined in this paragraph;
-
- when K1 represents the structure represented by formula (I′a) or formula (I′b), each of X1 and X2 independently represents
-
- when K1 represents the structure represented by formula (I′c), each of X3, X4, X5 and X6 independently represents hydrogen or
-
- wherein
- each of R21 and R22 independently represents hydrogen, halogen, a C1-C15 alkyl group, or a C2-C15 alkenyl group, wherein the C1-C15 alkyl group or the C2-C15 alkenyl group is unsubstituted or at least one hydrogen atom of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by a halogen atom, and/or at least one —CH2— of the C1-C15 alkyl group or the C2-C15 alkenyl group is substituted by —O—, and wherein the —O— does not directly bond to another —O—;
- each of B1, B2, and B3 independently represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, a 1,3-dioxane-2,5-diyl group, a tetrahydropyran-2,5-diyl group, a divalent dioxabicyclo[2.2.2]octylene group, a divalent trioxabicyclo[2.2.2]octylene group, a tetrahydronaphthalene-2,6-diyl group, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the 1,3-dioxane-2,5-diyl group, the benzofuran-2,5-diyl group, the tetrahydronaphthalene-2,6-diyl group, the indane-2,5-diyl group, or the tetrahydropyran-2,5-diyl group is unsubstituted or at least one hydrogen atom of the 1,4-phenylene group, the 1,4-cyclohexylene group, the 1,3-dioxane-2,5-diyl group, the benzofuran-2,5-diyl group, the tetrahydronaphthalene-2,6-diyl group, the indane-2,5-diyl group, or the tetrahydropyran-2,5-diyl group is substituted by a halogen atom or a —CN group, and/or at least one —CH2— of the 1,4-phenylene group, the 1,4-cyclohexylene group, the 1,3-dioxane-2,5-diyl group, the benzofuran-2,5-diyl group, the tetrahydronaphthalene-2,6-diyl group, the indane-2,5-diyl group, or the tetrahydropyran-2,5-diyl group is substituted by —O—, —N— or —S—, and wherein the —O—, —N—, and —S— do not directly bond to one another;
- each of Z21 and Z22 independently represents a single bond, a C1-C4 alkylene group, a C2-C4 alkenylene group, or a C2-C4 alkynylene group, wherein the C1-C4 alkylene group, the C2-C4 alkenylene group, or the C2-C4 alkynylene group is unsubstituted or at least one hydrogen atom of the C1-C4 alkylene group, the C2-C4 alkenylene group, or the C2-C4 alkynylene group is substituted by a halogen atom or a —CN group, and/or at least one —CH2— of the C1-C4 alkylene group, the C2-C4 alkenylene group, or the C2-C4 alkynylene group is substituted by —O— or —S—, and wherein the —O— does not directly bond to —O— or —S—, and —S— does not directly bond to —S—; and
- n21 represents 0, 1, or 2, and when n21 represents 2, the two B1 groups are identical to each other or different from each other.
-
- wherein
- the definitions of R21, R22, B1, B2, Z22, and n21 are respectively the same as the definitions of R21, R22, B1, B2, Z22, and n21 defined in the previous paragraphs.
-
- wherein
- each of K21, K22, K23, and K24 independently represents hydrogen or a methyl group;
- each of Z23 and Z24 independently represents a single bond, a C1-C15 linear alkylene group, a C3-C15 branched alkylene group, a C2-C15 linear alkenylene group, or a C3-C15 branched alkenylene group, wherein the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C2-C15 linear alkenylene group, or the C3-C15 branched alkenylene group is unsubstituted or at least one hydrogen atom of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C2-C15 linear alkenylene group, or the C3-C15 branched alkenylene group is substituted by a halogen atom, and/or at least one —CH2— of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C2-C15 linear alkenylene group, or the C3-C15 branched alkenylene group is substituted by —O—, —CO—, —COO—, or —OCO—, and wherein the —O—, —CO—, —COO—, and —OCO— do not directly bond to one another;
- each of Z25, Z26, Z27, and Z28 independently represents a single bond, —C≡C—, a C1-C15 linear alkylene group, a C3-C15 branched alkylene group, a C2-C15 linear alkenylene group, or a C3-C15 branched alkenylene group, wherein the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C2-C15 linear alkenylene group, or the C3-C15 branched alkenylene group is unsubstituted or at least one hydrogen atom of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C2-C15 linear alkenylene group, or the C3-C15 branched alkenylene group is substituted by a halogen atom, and/or at least one —CH2— of the C1-C15 linear alkylene group, the C3-C15 branched alkylene group, the C2-C15 linear alkenylene group, or the C3-C15 branched alkenylene group is substituted by —SiRe 2—, —S—, —O—, —CO—, —COO—, —OCO—, —CO—NRe—, or —NRe—CO—, and the —SiRe 2—, —S—, —O—, —CO—, —COO—, —OCO—, —CO—NRe—, and —NRe—CO— do not directly bond to one another, wherein Re represents hydrogen, a C1-C4 linear alkyl group, or a C3-C4 branched alkyl group, and the two Re groups bonded to the same Si atom are identical to each other or different from each other;
- each of B4, B5, B6 and B7 independently represents a 1,4-phenylene group, a 1,4-cyclohexylene group, a benzofuran-2,5-diyl group, a 1,3-dioxane-2,5-diyl group, a tetrahydropyran-2,5-diyl group, a divalent dioxabicyclo[2.2.2]octylene group, a divalent trioxabicyclo[2.2.2]octylene group, a tetrahydronaphthalene-2,6-diyl group, or an indane-2,5-diyl group, wherein the 1,4-phenylene group, the 1,4-cyclohexylene group, the benzofuran-2,5-diyl group, the 1,3-dioxane-2,5-diyl group, the tetrahydropyran-2,5-diyl group, the divalent dioxabicyclo[2.2.2]octylene group, the divalent trioxabicyclo[2.2.2]octylene group, the tetrahydronaphthalene-2,6-diyl group, or the indane-2,5-diyl group is unsubstituted or is substituted by at least one substituent, wherein the at least one substituent is selected from fluorine, chlorine, a —CN group, a C1-C12 linear alkyl group, a C3-C12 branched alkyl group, a C2-C12 linear alkenyl group, a C2-C12 linear alkynyl group, a C4-C12 branched alkenyl group, or a C4-C12 branched alkynyl group, wherein the C1-C12 linear alkyl group, the C3-C12 branched alkyl group, the C2-C12 linear alkenyl group, the C2-C12 linear alkynyl group, the C4-C12 branched alkenyl group, or the C4-C12 branched alkynyl group is unsubstituted or at least one hydrogen atom of the C1-C12 linear alkyl group, the C3-C12 branched alkyl group, the C2-C12 linear alkenyl group, the C2-C12 linear alkynyl group, the C4-C12 branched alkenyl group, or the C4-C12 branched alkynyl group is substituted by a halogen atom, and/or at least one —CH2— of the C1-C12 linear alkyl group, the C3-C12 branched alkyl group, the C2-C12 linear alkenyl group, the C2-C12 linear alkynyl group, the C4-C12 branched alkenyl group, or the C4-C12 branched alkynyl group is substituted by —O—, —CO—, —COO—, or —OCO—, and the —O—, —CO—, —COO—, and —OCO— do not directly bond to one another;
- M1 represents a single bond, —CH2O—, —OCH2—, —CH2CH2—, —CH═CH—, —C ≡C—, —CH2—, —C(CH3)2—, —C(CF3)2—, —SiH2—, —Si(CH3)2—, or —Si(CF3)2—;
- each of R23 and R24 independently represents a C1-C30 linear alkyl group or a C3-C30 branched alkyl group, wherein the C1-C30 linear alkyl group or the C3-C30 branched alkyl group is unsubstituted or at least one hydrogen atom of the C1-C30 linear alkyl group or the C3-C30 branched alkyl group is substituted by a halogen atom, and/or at least one —CH2— of the C1-C30 linear alkyl group or the C3-C30 branched alkyl group is substituted by —Si—, —O—, —CO—, —COO—, or —OCO—, and the —Si—, —O—, —CO—, —COO—, and —OCO— do not directly bond to one another; and
- each of n22 and n23 independently represents an integer of 0 to 3, and when n22 is 2 or more, the two or more B4 groups are identical to each other or different from each other, and the two or more M1 groups are identical to each other or different from each other; and when n23 is 2 or more, the two or more B6 groups are identical to each other or different from each other, and the two or more Z27 groups are identical to each other or different from each other.
| TABLE 3 | |||
| Content | |||
| Chemical structure | (wt %) | ||
| formula (II) | formula (II-1) |
|
10 |
|
|
5 | ||
|
|
12 | ||
|
|
14 | ||
| formula (II-2) |
|
10 | |
|
|
19 | ||
|
|
15 | ||
|
|
15 | ||
| formula (III) |
|
0.3 |
VHR═(V2/applied voltage value)×100%.
ΔVth=V(th,f)−V(th,i)
| TABLE 5 | ||||
| Amount added | Amount added | |||
| Additive | (parts by | Additive | (parts by | |
| No. | molecule | weight) | molecule | weight) |
| Reference | Ref | 1.5 | — | — |
| Example | ||||
| Example 1 | Exp-A1 | 2.0 | — | — |
| Example 2 | Exp-A2 | 0.3 | — | — |
| Example 3 | Exp-A3 | 0.1 | — | — |
| Example 4 | Exp-A3 | 0.2 | — | — |
| Example 5 | Exp-B4 | 3.0 | — | — |
| Example 6 | Exp-A2 | 0.2 | Exp-B1 | 0.5 |
| Example 7 | Exp-A2 | 0.2 | Exp-B2 | 0.5 |
| Example 8 | Exp-A3 | 0.2 | Exp-B3 | 0.5 |
| Example 9 | Exp-A3 | 0.2 | Exp-B4 | 0.5 |
| Example 10 | Exp-A2 | 0.2 | Exp-C | 0.5 |
| Example 11 | Exp-A2 | 0.1 | Exp-C | 1.7 |
| Example 12 | Exp-A3 | 0.2 | Exp-C | 0.5 |
| TABLE 6 | ||||
| Vertical alignment | VHR (%) | ΔVth value (V) | ||
| Reference | ◯ | 37.2 | −0.490 |
| Example | |||
| Example 1 | ◯ | 94.5 | −0.419 |
| Example 2 | ◯ | 96.3 | −0.195 |
| Example 3 | ◯ | 95.7 | −0.005 |
| Example 4 | ◯ | 97.5 | −0.088 |
| Example 5 | ◯ | 90.9 | −0.040 |
| Example 6 | ◯ | 89.2 | −0.171 |
| Example 7 | ◯ | 83.6 | −0.240 |
| Example 8 | ◯ | 97.5 | −0.327 |
| Example 9 | ◯ | 98.4 | −0.022 |
| Example 10 | ◯ | 98.0 | −0.010 |
| Example 11 | ◯ | 96.9 | −0.050 |
| Example 12 | ◯ | 96.2 | −0.153 |
Claims (12)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| TW108102902 | 2019-01-25 | ||
| TW108102902A TWI719394B (en) | 2019-01-25 | 2019-01-25 | Additive and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20200239773A1 US20200239773A1 (en) | 2020-07-30 |
| US11459506B2 true US11459506B2 (en) | 2022-10-04 |
Family
ID=71732274
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/749,406 Active 2040-04-29 US11459506B2 (en) | 2019-01-25 | 2020-01-22 | Additive and application thereof |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US11459506B2 (en) |
| JP (1) | JP6971337B2 (en) |
| CN (1) | CN111484859B (en) |
| TW (1) | TWI719394B (en) |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4748087A (en) * | 1984-06-08 | 1988-05-31 | The Wiggins Teape Group Limited | Plastic laminate of furniture foil and method of making |
| DD262865A1 (en) * | 1987-08-07 | 1988-12-14 | Univ Schiller Jena | PROCESS FOR REMOVING POLYMER NETWORKS |
| TW201807175A (en) | 2016-04-18 | 2018-03-01 | 捷恩智股份有限公司 | Liquid crystal composition and use thereof, and liquid crystal display device |
| US20190292463A1 (en) * | 2016-06-03 | 2019-09-26 | Jnc Corporation | Polymerizable polar compound, liquid crystal composition, and liquid crystal display element |
| US20190338190A1 (en) * | 2018-05-07 | 2019-11-07 | Daxin Materials Corporation | Silicon-containing compound, liquid-crystal composition and liquid-crystal display using the same |
| US20200087240A1 (en) * | 2017-06-12 | 2020-03-19 | Dic Corporation | Polymerizable compound and liquid crystal composition |
| US20200224098A1 (en) * | 2017-06-01 | 2020-07-16 | Dic Corporation | Polymerizable monomer, liquid crystal composition using polymerizable monomer, and liquid crystal display device |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2918658B1 (en) * | 2014-03-10 | 2020-05-13 | Merck Patent GmbH | Liquid crystalline media with homeotropic alignment |
| KR102452385B1 (en) * | 2014-07-28 | 2022-10-07 | 메르크 파텐트 게엠베하 | Liquid crystalline media having homeotropic alignment |
| WO2016152340A1 (en) * | 2015-03-24 | 2016-09-29 | Jnc株式会社 | Liquid crystal composition and liquid crystal display element |
| JP6070973B1 (en) * | 2015-12-17 | 2017-02-01 | Jnc株式会社 | Liquid crystal composition and liquid crystal display element |
| CN105542796A (en) * | 2016-02-01 | 2016-05-04 | 深圳市华星光电技术有限公司 | Auto-orientation liquid crystal medium composition and liquid crystal display panel |
| CN107805506A (en) * | 2017-11-14 | 2018-03-16 | 深圳市华星光电技术有限公司 | Liquid crystal display panel and preparation method thereof, auto-orientation liquid crystal media mixture |
| TWI640609B (en) * | 2018-05-07 | 2018-11-11 | 達興材料股份有限公司 | Liquid crystal composition and liquid crystal display device |
-
2019
- 2019-01-25 TW TW108102902A patent/TWI719394B/en active
-
2020
- 2020-01-15 CN CN202010040632.6A patent/CN111484859B/en active Active
- 2020-01-22 US US16/749,406 patent/US11459506B2/en active Active
- 2020-01-23 JP JP2020008934A patent/JP6971337B2/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4748087A (en) * | 1984-06-08 | 1988-05-31 | The Wiggins Teape Group Limited | Plastic laminate of furniture foil and method of making |
| DD262865A1 (en) * | 1987-08-07 | 1988-12-14 | Univ Schiller Jena | PROCESS FOR REMOVING POLYMER NETWORKS |
| TW201807175A (en) | 2016-04-18 | 2018-03-01 | 捷恩智股份有限公司 | Liquid crystal composition and use thereof, and liquid crystal display device |
| US20190292463A1 (en) * | 2016-06-03 | 2019-09-26 | Jnc Corporation | Polymerizable polar compound, liquid crystal composition, and liquid crystal display element |
| US20200224098A1 (en) * | 2017-06-01 | 2020-07-16 | Dic Corporation | Polymerizable monomer, liquid crystal composition using polymerizable monomer, and liquid crystal display device |
| US20200087240A1 (en) * | 2017-06-12 | 2020-03-19 | Dic Corporation | Polymerizable compound and liquid crystal composition |
| US20190338190A1 (en) * | 2018-05-07 | 2019-11-07 | Daxin Materials Corporation | Silicon-containing compound, liquid-crystal composition and liquid-crystal display using the same |
Non-Patent Citations (2)
| Title |
|---|
| English translation of DD262865. (Year: 1988). * |
| Office Action issued in corresponding TW application No. 108102902 dated Apr. 20, 2020. |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202028440A (en) | 2020-08-01 |
| TWI719394B (en) | 2021-02-21 |
| US20200239773A1 (en) | 2020-07-30 |
| CN111484859B (en) | 2022-01-11 |
| JP2020143268A (en) | 2020-09-10 |
| JP6971337B2 (en) | 2021-11-24 |
| CN111484859A (en) | 2020-08-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11345857B2 (en) | Spontaneous alignment assistant for liquid crystal compositions | |
| US8339567B2 (en) | Radial-shaped liquid crystal compound, and optical film and liquid crystal display device comprising the same | |
| US10494569B2 (en) | Liquid crystal compound containing difluoromethoxy bridge, composition and application thereof | |
| US11760934B2 (en) | Polymerizable compound, and liquid crystal composition and liquid crystal display element in which the compound is used | |
| US10108052B2 (en) | Liquid-crystal display element | |
| US20190203122A1 (en) | Spontaneous orientation aid for liquid crystal composition, compound suitable for said spontaneous orientation aid, liquid crystal composition, and liquid crystal display element | |
| US10113116B2 (en) | Liquid crystal compound and liquid crystal composition employing the same | |
| US20170349831A1 (en) | Liquid crystal compound containing difluormethoxy bridge, composition and application thereof | |
| TWI616432B (en) | Stabilizer, liquid crystal composition containing the same, and liquid crystal display element | |
| US9783735B2 (en) | Liquid crystal display device and liquid crystal composition used therefor | |
| US11739269B2 (en) | Additive and application thereof | |
| US11999892B2 (en) | Liquid crystal composition, liquid crystal display element and compound | |
| US11459506B2 (en) | Additive and application thereof | |
| US7968003B2 (en) | Pyran derivative, its preparation method, liquid crystal composition and liquid crystal display device comprising the same | |
| TWI464242B (en) | Negative dielectric anisotropic liquid crystal compound, liquid crystal display, and optoelectronic device | |
| US20190338190A1 (en) | Silicon-containing compound, liquid-crystal composition and liquid-crystal display using the same | |
| US9252366B2 (en) | Crosslinkable compound, method for preparing the same and light emitting device comprising the same | |
| US20260109900A1 (en) | Self alignment agent and liquid crystal composition thereof | |
| US20190284475A1 (en) | Liquid crystal compound and a liquid crystal composition including the same | |
| US20120165528A1 (en) | Chiral binaphthyl compounds | |
| KR102349224B1 (en) | Polymerizable mesogen compounds and liquid crystal compositions comprising the same | |
| KR101641705B1 (en) | Polymerizable liquid crystal compounds, liquid crystal composition comprising the compounds, and optical film comprising the composition | |
| KR20190070455A (en) | Liquid crystal composition having vertical alignment stability |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: ENTITY STATUS SET TO UNDISCOUNTED (ORIGINAL EVENT CODE: BIG.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| AS | Assignment |
Owner name: DAXIN MATERIALS CORPORATION, TAIWAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:WU, CHUNG-HSIEN;LIN, YI-CHUN;WANG, CHUN-CHIH;REEL/FRAME:051756/0305 Effective date: 20200103 |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NOTICE OF ALLOWANCE MAILED -- APPLICATION RECEIVED IN OFFICE OF PUBLICATIONS |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: PUBLICATIONS -- ISSUE FEE PAYMENT VERIFIED |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |