JP4496084B2 - 制御された診断機能を備える移植式医療装置 - Google Patents
制御された診断機能を備える移植式医療装置 Download PDFInfo
- Publication number
- JP4496084B2 JP4496084B2 JP2004555800A JP2004555800A JP4496084B2 JP 4496084 B2 JP4496084 B2 JP 4496084B2 JP 2004555800 A JP2004555800 A JP 2004555800A JP 2004555800 A JP2004555800 A JP 2004555800A JP 4496084 B2 JP4496084 B2 JP 4496084B2
- Authority
- JP
- Japan
- Prior art keywords
- mode
- implantable medical
- medical device
- circuit
- physiological
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 230000007211 cardiovascular event Effects 0.000 claims description 33
- 238000011282 treatment Methods 0.000 claims description 33
- 238000004891 communication Methods 0.000 claims description 27
- 238000007726 management method Methods 0.000 claims description 11
- 230000001225 therapeutic effect Effects 0.000 claims description 10
- 230000035790 physiological processes and functions Effects 0.000 claims description 9
- 238000012544 monitoring process Methods 0.000 claims description 6
- 230000004913 activation Effects 0.000 claims description 5
- 208000006218 bradycardia Diseases 0.000 claims description 5
- 230000000638 stimulation Effects 0.000 claims description 5
- 208000001871 Tachycardia Diseases 0.000 claims description 4
- 206010049447 Tachyarrhythmia Diseases 0.000 claims description 2
- 238000002054 transplantation Methods 0.000 claims description 2
- 238000012384 transportation and delivery Methods 0.000 claims description 2
- 206010049765 Bradyarrhythmia Diseases 0.000 claims 1
- 230000003213 activating effect Effects 0.000 claims 1
- 239000002246 antineoplastic agent Substances 0.000 claims 1
- 229940127089 cytotoxic agent Drugs 0.000 claims 1
- 230000006870 function Effects 0.000 description 42
- 238000001514 detection method Methods 0.000 description 38
- 238000004458 analytical method Methods 0.000 description 33
- 238000002560 therapeutic procedure Methods 0.000 description 20
- 230000002452 interceptive effect Effects 0.000 description 18
- 206010003119 arrhythmia Diseases 0.000 description 12
- 230000002526 effect on cardiovascular system Effects 0.000 description 12
- 230000036541 health Effects 0.000 description 10
- 230000006793 arrhythmia Effects 0.000 description 9
- 238000010586 diagram Methods 0.000 description 9
- 230000033764 rhythmic process Effects 0.000 description 9
- 238000000034 method Methods 0.000 description 8
- 230000000747 cardiac effect Effects 0.000 description 7
- 230000001351 cycling effect Effects 0.000 description 5
- 208000017667 Chronic Disease Diseases 0.000 description 4
- 230000036471 bradycardia Effects 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 238000002512 chemotherapy Methods 0.000 description 3
- 238000003745 diagnosis Methods 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 210000002837 heart atrium Anatomy 0.000 description 3
- 229910052744 lithium Inorganic materials 0.000 description 3
- 238000005086 pumping Methods 0.000 description 3
- 230000035939 shock Effects 0.000 description 3
- 238000011269 treatment regimen Methods 0.000 description 3
- 210000005243 upper chamber Anatomy 0.000 description 3
- 206010047281 Ventricular arrhythmia Diseases 0.000 description 2
- 230000002159 abnormal effect Effects 0.000 description 2
- 238000010009 beating Methods 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 238000013523 data management Methods 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 238000002651 drug therapy Methods 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 230000005923 long-lasting effect Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 230000000069 prophylactic effect Effects 0.000 description 2
- 230000006794 tachycardia Effects 0.000 description 2
- 208000003663 ventricular fibrillation Diseases 0.000 description 2
- 206010003658 Atrial Fibrillation Diseases 0.000 description 1
- 206010003662 Atrial flutter Diseases 0.000 description 1
- 206010003671 Atrioventricular Block Diseases 0.000 description 1
- 208000010271 Heart Block Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 102100026827 Protein associated with UVRAG as autophagy enhancer Human genes 0.000 description 1
- 101710102978 Protein associated with UVRAG as autophagy enhancer Proteins 0.000 description 1
- YFXWODPYUNGUEE-UHFFFAOYSA-N [I].[Li] Chemical compound [I].[Li] YFXWODPYUNGUEE-UHFFFAOYSA-N 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 239000003416 antiarrhythmic agent Substances 0.000 description 1
- 206010061592 cardiac fibrillation Diseases 0.000 description 1
- 230000009084 cardiovascular function Effects 0.000 description 1
- 230000005800 cardiovascular problem Effects 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 230000004087 circulation Effects 0.000 description 1
- 238000011340 continuous therapy Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 206010047302 ventricular tachycardia Diseases 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/37211—Means for communicating with stimulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/372—Arrangements in connection with the implantation of stimulators
- A61N1/37211—Means for communicating with stimulators
- A61N1/37252—Details of algorithms or data aspects of communication system, e.g. handshaking, transmitting specific data or segmenting data
- A61N1/37282—Details of algorithms or data aspects of communication system, e.g. handshaking, transmitting specific data or segmenting data characterised by communication with experts in remote locations using a network
Description
Claims (9)
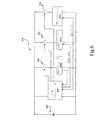
- 移植可能医療装置から成るシステムであって、前記移植可能医療装置が、
治療回路と、
前記移植可能医療装置からの情報を外部の患者管理システムに通信する通信回路と、
生理学的検知回路と、
移植後に起動される第1のモードと、
前記第1モード中にオンされているとき、生理学的事象を検知した前記生理学的検知回路によって起動される第2のモードと、から成り、
前記第1モードでは、前記生理学的検知回路の少なくとも一部に対する電力が、省電力デューティサイクルで連続的にオンとオフを繰り返へされ、前記治療回路及び前記通信回路の電力はオフされており、
前記第2モードは、前記治療回路を永久的に起動することによって前記第1モードを解除するとともに、少なくとも一時的に前記通信回路を起動する、
ことを特徴とするシステム。 - 前記第2モードは、前記移植可能医療装置から前記外部患者管理システムへの情報通信を始動する、請求項1に記載のシステム。
- 前記第2モードは、前記通信回路に永久的に電力供給する、請求項1に記載のシステム。
- 治療介入を必要とする心臓血管事象を含む生理学的事象を検知したとき、前記第2モードの起動により、前記移植可能医療装置が、監視モードから介入・通信モードに切り替わる、請求項1に記載のシステム。
- 前記治療回路は、電気刺激制御回路を含む、請求項1に記載のシステム。
- 前記治療回路は、化学治療剤配給制御回路を含む、請求項1に記載のシステム。
- 前記第2モードは、前記生理学的検知回路が前記生理学的事象を検知したときに起動され、前記生理学的事象は頻拍性不整脈である、請求項1に記載のシステム。
- 前記第2モードは、前記生理学的検知回路が前記生理学的事象を検知したときに起動され、前記生理学的事象は徐脈型不整脈である、請求項1に記載のシステム。
- 前記第2モードは、前記生理学的検知回路が前記生理学的事象を検知したときに起動され、前記生理学的事象は原線維性事象である、請求項1に記載のシステム。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/305,548 US7333853B2 (en) | 2002-11-26 | 2002-11-26 | Implantable medical device having a controlled diagnostic function |
| PCT/US2003/037945 WO2004047912A2 (en) | 2002-11-26 | 2003-11-25 | Implantable medical device having a controlled diagnostic function |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2006507874A JP2006507874A (ja) | 2006-03-09 |
| JP2006507874A5 JP2006507874A5 (ja) | 2007-01-18 |
| JP4496084B2 true JP4496084B2 (ja) | 2010-07-07 |
Family
ID=32325455
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004555800A Expired - Fee Related JP4496084B2 (ja) | 2002-11-26 | 2003-11-25 | 制御された診断機能を備える移植式医療装置 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US7333853B2 (ja) |
| EP (1) | EP1565233A2 (ja) |
| JP (1) | JP4496084B2 (ja) |
| AU (1) | AU2003295985A1 (ja) |
| WO (1) | WO2004047912A2 (ja) |
Families Citing this family (95)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101052889A (zh) * | 2004-11-08 | 2007-10-10 | 皇家飞利浦电子股份有限公司 | 用于便携式医学设备的无线电池状态管理 |
| US8108046B2 (en) | 2004-12-17 | 2012-01-31 | Medtronic, Inc. | System and method for using cardiac events to trigger therapy for treating nervous system disorders |
| US20070239060A1 (en) * | 2004-12-17 | 2007-10-11 | Medtronic, Inc. | System and method for regulating cardiac triggered therapy to the brain |
| US8108038B2 (en) | 2004-12-17 | 2012-01-31 | Medtronic, Inc. | System and method for segmenting a cardiac signal based on brain activity |
| US8209009B2 (en) * | 2004-12-17 | 2012-06-26 | Medtronic, Inc. | System and method for segmenting a cardiac signal based on brain stimulation |
| US8112148B2 (en) * | 2004-12-17 | 2012-02-07 | Medtronic, Inc. | System and method for monitoring cardiac signal activity in patients with nervous system disorders |
| US8209019B2 (en) | 2004-12-17 | 2012-06-26 | Medtronic, Inc. | System and method for utilizing brain state information to modulate cardiac therapy |
| US8214035B2 (en) * | 2004-12-17 | 2012-07-03 | Medtronic, Inc. | System and method for utilizing brain state information to modulate cardiac therapy |
| US8485979B2 (en) * | 2004-12-17 | 2013-07-16 | Medtronic, Inc. | System and method for monitoring or treating nervous system disorders |
| US8112153B2 (en) * | 2004-12-17 | 2012-02-07 | Medtronic, Inc. | System and method for monitoring or treating nervous system disorders |
| US8041418B2 (en) | 2004-12-17 | 2011-10-18 | Medtronic, Inc. | System and method for regulating cardiac triggered therapy to the brain |
| US7545272B2 (en) | 2005-02-08 | 2009-06-09 | Therasense, Inc. | RF tag on test strips, test strip vials and boxes |
| US8391990B2 (en) | 2005-05-18 | 2013-03-05 | Cardiac Pacemakers, Inc. | Modular antitachyarrhythmia therapy system |
| US8688219B2 (en) | 2006-07-28 | 2014-04-01 | Medronic, Inc. | Dynamic sampling |
| US8000788B2 (en) * | 2007-04-27 | 2011-08-16 | Medtronic, Inc. | Implantable medical device for treating neurological conditions including ECG sensing |
| FR2960663B1 (fr) * | 2010-06-01 | 2013-11-01 | Alcatel Lucent | Gestion d'objets par systeme de traitement de l'information |
| WO2015106015A1 (en) | 2014-01-10 | 2015-07-16 | Cardiac Pacemakers, Inc. | Systems and methods for detecting cardiac arrhythmias |
| EP3308833B1 (en) | 2014-01-10 | 2019-06-26 | Cardiac Pacemakers, Inc. | Methods and systems for improved communication between medical devices |
| EP3185952B1 (en) | 2014-08-28 | 2018-07-25 | Cardiac Pacemakers, Inc. | Implantable cardiac rhythm system and an associated method for triggering a blanking period through a second device |
| WO2016126613A1 (en) | 2015-02-06 | 2016-08-11 | Cardiac Pacemakers, Inc. | Systems and methods for treating cardiac arrhythmias |
| WO2016126968A1 (en) | 2015-02-06 | 2016-08-11 | Cardiac Pacemakers, Inc. | Systems and methods for safe delivery of electrical stimulation therapy |
| US10046167B2 (en) | 2015-02-09 | 2018-08-14 | Cardiac Pacemakers, Inc. | Implantable medical device with radiopaque ID tag |
| CN107530002B (zh) | 2015-03-04 | 2021-04-30 | 心脏起搏器股份公司 | 用于治疗心律失常的系统和方法 |
| CN107427222B (zh) | 2015-03-18 | 2021-02-09 | 心脏起搏器股份公司 | 使用链路质量评估的医疗设备系统中的通信 |
| US10050700B2 (en) | 2015-03-18 | 2018-08-14 | Cardiac Pacemakers, Inc. | Communications in a medical device system with temporal optimization |
| CN108136186B (zh) | 2015-08-20 | 2021-09-17 | 心脏起搏器股份公司 | 用于医疗装置之间的通信的系统和方法 |
| EP3337559B1 (en) | 2015-08-20 | 2019-10-16 | Cardiac Pacemakers, Inc. | Systems and methods for communication between medical devices |
| US9956414B2 (en) | 2015-08-27 | 2018-05-01 | Cardiac Pacemakers, Inc. | Temporal configuration of a motion sensor in an implantable medical device |
| US9968787B2 (en) | 2015-08-27 | 2018-05-15 | Cardiac Pacemakers, Inc. | Spatial configuration of a motion sensor in an implantable medical device |
| US10226631B2 (en) | 2015-08-28 | 2019-03-12 | Cardiac Pacemakers, Inc. | Systems and methods for infarct detection |
| EP3341076B1 (en) | 2015-08-28 | 2022-05-11 | Cardiac Pacemakers, Inc. | Systems and methods for behaviorally responsive signal detection and therapy delivery |
| WO2017040115A1 (en) | 2015-08-28 | 2017-03-09 | Cardiac Pacemakers, Inc. | System for detecting tamponade |
| WO2017044389A1 (en) | 2015-09-11 | 2017-03-16 | Cardiac Pacemakers, Inc. | Arrhythmia detection and confirmation |
| EP3359251B1 (en) | 2015-10-08 | 2019-08-07 | Cardiac Pacemakers, Inc. | Adjusting pacing rates in an implantable medical device |
| US9781679B2 (en) * | 2015-11-27 | 2017-10-03 | Samsung Electronics Co., Ltd. | Electronic systems and method of operating electronic systems |
| US10183170B2 (en) | 2015-12-17 | 2019-01-22 | Cardiac Pacemakers, Inc. | Conducted communication in a medical device system |
| US10905886B2 (en) | 2015-12-28 | 2021-02-02 | Cardiac Pacemakers, Inc. | Implantable medical device for deployment across the atrioventricular septum |
| WO2017127548A1 (en) | 2016-01-19 | 2017-07-27 | Cardiac Pacemakers, Inc. | Devices for wirelessly recharging a rechargeable battery of an implantable medical device |
| EP3411113B1 (en) | 2016-02-04 | 2019-11-27 | Cardiac Pacemakers, Inc. | Delivery system with force sensor for leadless cardiac device |
| CN108883286B (zh) | 2016-03-31 | 2021-12-07 | 心脏起搏器股份公司 | 具有可充电电池的可植入医疗设备 |
| US10668294B2 (en) | 2016-05-10 | 2020-06-02 | Cardiac Pacemakers, Inc. | Leadless cardiac pacemaker configured for over the wire delivery |
| US10328272B2 (en) | 2016-05-10 | 2019-06-25 | Cardiac Pacemakers, Inc. | Retrievability for implantable medical devices |
| US10512784B2 (en) | 2016-06-27 | 2019-12-24 | Cardiac Pacemakers, Inc. | Cardiac therapy system using subcutaneously sensed P-waves for resynchronization pacing management |
| US11207527B2 (en) | 2016-07-06 | 2021-12-28 | Cardiac Pacemakers, Inc. | Method and system for determining an atrial contraction timing fiducial in a leadless cardiac pacemaker system |
| US10426962B2 (en) | 2016-07-07 | 2019-10-01 | Cardiac Pacemakers, Inc. | Leadless pacemaker using pressure measurements for pacing capture verification |
| WO2018017226A1 (en) | 2016-07-20 | 2018-01-25 | Cardiac Pacemakers, Inc. | System for utilizing an atrial contraction timing fiducial in a leadless cardiac pacemaker system |
| EP3500342B1 (en) | 2016-08-19 | 2020-05-13 | Cardiac Pacemakers, Inc. | Trans-septal implantable medical device |
| WO2018039322A1 (en) | 2016-08-24 | 2018-03-01 | Cardiac Pacemakers, Inc. | Cardiac resynchronization using fusion promotion for timing management |
| CN109640809B (zh) | 2016-08-24 | 2021-08-17 | 心脏起搏器股份公司 | 使用p波到起搏定时的集成式多装置心脏再同步治疗 |
| US10758737B2 (en) | 2016-09-21 | 2020-09-01 | Cardiac Pacemakers, Inc. | Using sensor data from an intracardially implanted medical device to influence operation of an extracardially implantable cardioverter |
| CN109803720B (zh) | 2016-09-21 | 2023-08-15 | 心脏起搏器股份公司 | 具有容纳其内部部件并充当电池壳和内部电池的端子的壳体的无引线刺激设备 |
| WO2018057626A1 (en) | 2016-09-21 | 2018-03-29 | Cardiac Pacemakers, Inc. | Implantable cardiac monitor |
| US10561330B2 (en) | 2016-10-27 | 2020-02-18 | Cardiac Pacemakers, Inc. | Implantable medical device having a sense channel with performance adjustment |
| JP7038115B2 (ja) | 2016-10-27 | 2022-03-17 | カーディアック ペースメイカーズ, インコーポレイテッド | 圧力センサを備えた植込み型医療装置 |
| WO2018081225A1 (en) | 2016-10-27 | 2018-05-03 | Cardiac Pacemakers, Inc. | Implantable medical device delivery system with integrated sensor |
| US10463305B2 (en) | 2016-10-27 | 2019-11-05 | Cardiac Pacemakers, Inc. | Multi-device cardiac resynchronization therapy with timing enhancements |
| US10434314B2 (en) | 2016-10-27 | 2019-10-08 | Cardiac Pacemakers, Inc. | Use of a separate device in managing the pace pulse energy of a cardiac pacemaker |
| US10413733B2 (en) | 2016-10-27 | 2019-09-17 | Cardiac Pacemakers, Inc. | Implantable medical device with gyroscope |
| US10617874B2 (en) | 2016-10-31 | 2020-04-14 | Cardiac Pacemakers, Inc. | Systems and methods for activity level pacing |
| US10434317B2 (en) | 2016-10-31 | 2019-10-08 | Cardiac Pacemakers, Inc. | Systems and methods for activity level pacing |
| WO2018089311A1 (en) | 2016-11-08 | 2018-05-17 | Cardiac Pacemakers, Inc | Implantable medical device for atrial deployment |
| EP3538213B1 (en) | 2016-11-09 | 2023-04-12 | Cardiac Pacemakers, Inc. | Systems and devices for setting cardiac pacing pulse parameters for a cardiac pacing device |
| US10894163B2 (en) | 2016-11-21 | 2021-01-19 | Cardiac Pacemakers, Inc. | LCP based predictive timing for cardiac resynchronization |
| US10881863B2 (en) | 2016-11-21 | 2021-01-05 | Cardiac Pacemakers, Inc. | Leadless cardiac pacemaker with multimode communication |
| US10639486B2 (en) | 2016-11-21 | 2020-05-05 | Cardiac Pacemakers, Inc. | Implantable medical device with recharge coil |
| US10881869B2 (en) | 2016-11-21 | 2021-01-05 | Cardiac Pacemakers, Inc. | Wireless re-charge of an implantable medical device |
| US11147979B2 (en) | 2016-11-21 | 2021-10-19 | Cardiac Pacemakers, Inc. | Implantable medical device with a magnetically permeable housing and an inductive coil disposed about the housing |
| US11207532B2 (en) | 2017-01-04 | 2021-12-28 | Cardiac Pacemakers, Inc. | Dynamic sensing updates using postural input in a multiple device cardiac rhythm management system |
| WO2018140623A1 (en) | 2017-01-26 | 2018-08-02 | Cardiac Pacemakers, Inc. | Leadless device with overmolded components |
| US10737102B2 (en) | 2017-01-26 | 2020-08-11 | Cardiac Pacemakers, Inc. | Leadless implantable device with detachable fixation |
| EP3573706A1 (en) | 2017-01-26 | 2019-12-04 | Cardiac Pacemakers, Inc. | Intra-body device communication with redundant message transmission |
| US10821288B2 (en) | 2017-04-03 | 2020-11-03 | Cardiac Pacemakers, Inc. | Cardiac pacemaker with pacing pulse energy adjustment based on sensed heart rate |
| US10905872B2 (en) | 2017-04-03 | 2021-02-02 | Cardiac Pacemakers, Inc. | Implantable medical device with a movable electrode biased toward an extended position |
| WO2019036600A1 (en) | 2017-08-18 | 2019-02-21 | Cardiac Pacemakers, Inc. | IMPLANTABLE MEDICAL DEVICE WITH PRESSURE SENSOR |
| US10918875B2 (en) | 2017-08-18 | 2021-02-16 | Cardiac Pacemakers, Inc. | Implantable medical device with a flux concentrator and a receiving coil disposed about the flux concentrator |
| CN111107899B (zh) | 2017-09-20 | 2024-04-02 | 心脏起搏器股份公司 | 具有多种操作模式的可植入医疗装置 |
| US11185703B2 (en) | 2017-11-07 | 2021-11-30 | Cardiac Pacemakers, Inc. | Leadless cardiac pacemaker for bundle of his pacing |
| WO2019108830A1 (en) | 2017-12-01 | 2019-06-06 | Cardiac Pacemakers, Inc. | Leadless cardiac pacemaker with reversionary behavior |
| EP3717059A1 (en) | 2017-12-01 | 2020-10-07 | Cardiac Pacemakers, Inc. | Methods and systems for detecting atrial contraction timing fiducials within a search window from a ventricularly implanted leadless cardiac pacemaker |
| US11260216B2 (en) | 2017-12-01 | 2022-03-01 | Cardiac Pacemakers, Inc. | Methods and systems for detecting atrial contraction timing fiducials during ventricular filling from a ventricularly implanted leadless cardiac pacemaker |
| EP3717063B1 (en) | 2017-12-01 | 2023-12-27 | Cardiac Pacemakers, Inc. | Systems for detecting atrial contraction timing fiducials and determining a cardiac interval from a ventricularly implanted leadless cardiac pacemaker |
| US10874861B2 (en) | 2018-01-04 | 2020-12-29 | Cardiac Pacemakers, Inc. | Dual chamber pacing without beat-to-beat communication |
| US11529523B2 (en) | 2018-01-04 | 2022-12-20 | Cardiac Pacemakers, Inc. | Handheld bridge device for providing a communication bridge between an implanted medical device and a smartphone |
| EP3768369A1 (en) | 2018-03-23 | 2021-01-27 | Medtronic, Inc. | Av synchronous vfa cardiac therapy |
| WO2019183514A1 (en) | 2018-03-23 | 2019-09-26 | Medtronic, Inc. | Vfa cardiac therapy for tachycardia |
| CN111902187A (zh) | 2018-03-23 | 2020-11-06 | 美敦力公司 | Vfa心脏再同步治疗 |
| EP3856331A1 (en) | 2018-09-26 | 2021-08-04 | Medtronic, Inc. | Capture in ventricle-from-atrium cardiac therapy |
| US11679265B2 (en) | 2019-02-14 | 2023-06-20 | Medtronic, Inc. | Lead-in-lead systems and methods for cardiac therapy |
| US11697025B2 (en) | 2019-03-29 | 2023-07-11 | Medtronic, Inc. | Cardiac conduction system capture |
| US11213676B2 (en) | 2019-04-01 | 2022-01-04 | Medtronic, Inc. | Delivery systems for VfA cardiac therapy |
| US11712188B2 (en) | 2019-05-07 | 2023-08-01 | Medtronic, Inc. | Posterior left bundle branch engagement |
| US11305127B2 (en) | 2019-08-26 | 2022-04-19 | Medtronic Inc. | VfA delivery and implant region detection |
| US11813466B2 (en) | 2020-01-27 | 2023-11-14 | Medtronic, Inc. | Atrioventricular nodal stimulation |
| US11911168B2 (en) | 2020-04-03 | 2024-02-27 | Medtronic, Inc. | Cardiac conduction system therapy benefit determination |
| US11813464B2 (en) | 2020-07-31 | 2023-11-14 | Medtronic, Inc. | Cardiac conduction system evaluation |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3057356A (en) | 1960-07-22 | 1962-10-09 | Wilson Greatbatch Inc | Medical cardiac pacemaker |
| US4390022A (en) | 1981-05-18 | 1983-06-28 | Intermedics, Inc. | Implantable device with microprocessor control |
| US4523595A (en) | 1981-11-25 | 1985-06-18 | Zibell J Scott | Method and apparatus for automatic detection and treatment of ventricular fibrillation |
| US5282837A (en) * | 1991-04-12 | 1994-02-01 | Incontrol, Inc. | Atrial defibrillator and method |
| US5350407A (en) | 1992-12-30 | 1994-09-27 | Telectronics Pacing Systems, Inc. | Implantable stimulator having quiescent and active modes of operation |
| US5464432A (en) * | 1994-04-25 | 1995-11-07 | Incontrol, Inc. | Implantable atrial defibrillator having an intermittently activated fibrillation detector |
| US5527345A (en) * | 1994-05-17 | 1996-06-18 | Infinger; Kenneth R. | Implantable atrial defibrillator having an intermittenly activated pacing modality |
| US5746697A (en) * | 1996-02-09 | 1998-05-05 | Nellcor Puritan Bennett Incorporated | Medical diagnostic apparatus with sleep mode |
| US5725559A (en) | 1996-05-16 | 1998-03-10 | Intermedics Inc. | Programmably upgradable implantable medical device |
| US5645569A (en) * | 1996-06-04 | 1997-07-08 | Incontrol, Inc. | Post atrial cardioversion atrial pacing and method |
| US6038476A (en) | 1998-06-12 | 2000-03-14 | Pacesetter, Inc. | System and method for analyzing the efficacy of cardiac stimulation therapy |
| US6016448A (en) | 1998-10-27 | 2000-01-18 | Medtronic, Inc. | Multilevel ERI for implantable medical devices |
| US6363280B1 (en) * | 1999-10-15 | 2002-03-26 | Pacesetter, Inc. | Battery conservation in implantable cardioverter-defibrillators and pacemakers |
| US6497655B1 (en) | 1999-12-17 | 2002-12-24 | Medtronic, Inc. | Virtual remote monitor, alert, diagnostics and programming for implantable medical device systems |
-
2002
- 2002-11-26 US US10/305,548 patent/US7333853B2/en not_active Expired - Fee Related
-
2003
- 2003-11-25 JP JP2004555800A patent/JP4496084B2/ja not_active Expired - Fee Related
- 2003-11-25 AU AU2003295985A patent/AU2003295985A1/en not_active Abandoned
- 2003-11-25 EP EP03787200A patent/EP1565233A2/en not_active Withdrawn
- 2003-11-25 WO PCT/US2003/037945 patent/WO2004047912A2/en active Application Filing
Also Published As
| Publication number | Publication date |
|---|---|
| EP1565233A2 (en) | 2005-08-24 |
| US7333853B2 (en) | 2008-02-19 |
| AU2003295985A8 (en) | 2004-06-18 |
| AU2003295985A1 (en) | 2004-06-18 |
| JP2006507874A (ja) | 2006-03-09 |
| WO2004047912A2 (en) | 2004-06-10 |
| US20040102816A1 (en) | 2004-05-27 |
| WO2004047912A3 (en) | 2004-07-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4496084B2 (ja) | 制御された診断機能を備える移植式医療装置 | |
| US7142911B2 (en) | Method and apparatus for monitoring drug effects on cardiac electrical signals using an implantable cardiac stimulation device | |
| US6311089B1 (en) | Implantable stimulation device and method for determining ventricular and atrial sensitivity thresholds | |
| US6263244B1 (en) | Implantable stimulation device and method for determining atrial autocapture using PVC response | |
| US6922587B2 (en) | System and method for tracking progression of left ventricular dysfunction using implantable cardiac stimulation device | |
| US6259950B1 (en) | Implantable stimulation device and method for determining a trial autocapture using backup atrial stimulation | |
| US6243606B1 (en) | Implantable stimulation device and method for determining atrial autocapture using programmable PVARP | |
| US6961617B1 (en) | Method and apparatus to backup, update and share data among implantable cardiac stimulation device programmers | |
| US8170663B2 (en) | Method and apparatus to control delivery of high-voltage and anti-tachy pacing therapy in an implantable medical device | |
| US6073049A (en) | Programmably upgradable implantable cardiac pacemaker | |
| US7317942B2 (en) | Dynamic discrimination utilizing anti-tachy pacing therapy in an implantable medical device | |
| US6285908B1 (en) | Implantable stimulation device and method for determining atrial autocapture using programmable AV delay | |
| US5697884A (en) | Cardiac assist device having circadian muscle simulation | |
| US7979122B2 (en) | Implantable sudden cardiac death prevention device with reduced programmable feature set | |
| EP1747044B1 (en) | Apparatus to control delivery of high-voltage and anti-tachy pacing therapy in an implantable medical device | |
| US6038476A (en) | System and method for analyzing the efficacy of cardiac stimulation therapy | |
| US7069078B2 (en) | Insulin-mediated glucose uptake monitor | |
| EP0904130A1 (en) | Preemptive mode pacer and method preventing tachyarrhythmias | |
| WO1982002836A1 (en) | Externally programmable,implantable heart stimulator | |
| JP2003511163A (ja) | 心不全の患者の心臓毛細管の発達を促す方法 | |
| US6766193B1 (en) | Battery conservation in implantable cardioverter-defibrillators and pacemakers | |
| US7421292B1 (en) | System and method for controlling the recording of diagnostic medical data in an implantable medical device | |
| US20040138713A1 (en) | External defibrillation and transcutaneous pacing device and methods for operating the device | |
| US7006867B1 (en) | Methods and apparatus for overdrive pacing multiple atrial sites using an implantable cardiac stimulation device | |
| US7184831B2 (en) | Method and apparatus to control delivery of high-voltage and anti-tachy pacing therapy in an implantable medical device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20061122 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20061122 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090804 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20090901 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20090902 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20091023 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20091117 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20100114 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20100330 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20100412 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130416 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20130416 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20140416 Year of fee payment: 4 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |