JP2005111452A - 半導体金属酸化物光触媒およびそれを用いた有害化学物質の分解方法 - Google Patents
半導体金属酸化物光触媒およびそれを用いた有害化学物質の分解方法 Download PDFInfo
- Publication number
- JP2005111452A JP2005111452A JP2003352961A JP2003352961A JP2005111452A JP 2005111452 A JP2005111452 A JP 2005111452A JP 2003352961 A JP2003352961 A JP 2003352961A JP 2003352961 A JP2003352961 A JP 2003352961A JP 2005111452 A JP2005111452 A JP 2005111452A
- Authority
- JP
- Japan
- Prior art keywords
- metal oxide
- semiconductor metal
- photocatalyst
- semiconductor
- compounds
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000004065 semiconductor Substances 0.000 title claims abstract description 86
- 229910044991 metal oxide Inorganic materials 0.000 title claims abstract description 85
- 150000004706 metal oxides Chemical class 0.000 title claims abstract description 85
- 239000011941 photocatalyst Substances 0.000 title claims abstract description 69
- 238000000034 method Methods 0.000 title claims description 34
- 239000000126 substance Substances 0.000 title claims description 24
- 238000000354 decomposition reaction Methods 0.000 title claims description 13
- 239000000383 hazardous chemical Substances 0.000 title claims description 4
- 150000001875 compounds Chemical class 0.000 claims abstract description 41
- 229910052802 copper Inorganic materials 0.000 claims abstract description 41
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 40
- 125000004430 oxygen atom Chemical group O* 0.000 claims abstract description 32
- 239000013078 crystal Substances 0.000 claims abstract description 27
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims abstract description 23
- 239000001301 oxygen Substances 0.000 claims abstract description 23
- 229910052760 oxygen Inorganic materials 0.000 claims abstract description 23
- 229910052751 metal Inorganic materials 0.000 claims abstract description 19
- 229910052723 transition metal Inorganic materials 0.000 claims abstract description 19
- 150000003624 transition metals Chemical class 0.000 claims abstract description 17
- 239000002184 metal Substances 0.000 claims description 17
- 239000000203 mixture Substances 0.000 claims description 15
- 230000001443 photoexcitation Effects 0.000 claims description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 13
- 239000001257 hydrogen Substances 0.000 claims description 12
- 229910052739 hydrogen Inorganic materials 0.000 claims description 12
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 10
- 238000004519 manufacturing process Methods 0.000 claims description 10
- 238000004774 atomic orbital Methods 0.000 claims description 5
- 229910000510 noble metal Inorganic materials 0.000 claims description 3
- 230000001678 irradiating effect Effects 0.000 claims description 2
- 230000005284 excitation Effects 0.000 abstract description 11
- 125000004429 atom Chemical group 0.000 abstract description 10
- 230000006798 recombination Effects 0.000 abstract description 8
- 238000005215 recombination Methods 0.000 abstract description 8
- 238000000926 separation method Methods 0.000 abstract description 7
- 230000003287 optical effect Effects 0.000 abstract 1
- 230000008569 process Effects 0.000 description 17
- 239000003054 catalyst Substances 0.000 description 16
- 239000010936 titanium Substances 0.000 description 11
- 239000000463 material Substances 0.000 description 10
- 238000005516 engineering process Methods 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 8
- 230000001699 photocatalysis Effects 0.000 description 7
- 230000007704 transition Effects 0.000 description 7
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 6
- 230000009471 action Effects 0.000 description 6
- 238000007254 oxidation reaction Methods 0.000 description 6
- 238000006864 oxidative decomposition reaction Methods 0.000 description 6
- 229910010413 TiO 2 Inorganic materials 0.000 description 5
- 230000003647 oxidation Effects 0.000 description 5
- 239000002245 particle Substances 0.000 description 5
- 238000010586 diagram Methods 0.000 description 4
- 239000000446 fuel Substances 0.000 description 4
- 239000010409 thin film Substances 0.000 description 4
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 3
- 229910002092 carbon dioxide Inorganic materials 0.000 description 3
- 239000001569 carbon dioxide Substances 0.000 description 3
- 230000003197 catalytic effect Effects 0.000 description 3
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 3
- 238000003746 solid phase reaction Methods 0.000 description 3
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 229910003298 Ni-Ni Inorganic materials 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 238000002485 combustion reaction Methods 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 239000010419 fine particle Substances 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 125000004433 nitrogen atom Chemical group N* 0.000 description 2
- 238000013032 photocatalytic reaction Methods 0.000 description 2
- 238000006722 reduction reaction Methods 0.000 description 2
- 238000001179 sorption measurement Methods 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000013076 target substance Substances 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- 230000005428 wave function Effects 0.000 description 2
- 229910002915 BiVO4 Inorganic materials 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical class [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000003905 agrochemical Substances 0.000 description 1
- 239000003570 air Substances 0.000 description 1
- 238000004577 artificial photosynthesis Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical class OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000010574 gas phase reaction Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 239000002920 hazardous waste Substances 0.000 description 1
- 238000005470 impregnation Methods 0.000 description 1
- 229910052738 indium Inorganic materials 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 150000002496 iodine Chemical class 0.000 description 1
- 238000005468 ion implantation Methods 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- GNRSAWUEBMWBQH-UHFFFAOYSA-N nickel(II) oxide Inorganic materials [Ni]=O GNRSAWUEBMWBQH-UHFFFAOYSA-N 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000000575 pesticide Substances 0.000 description 1
- 238000002256 photodeposition Methods 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 239000010970 precious metal Substances 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 239000002689 soil Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 1
- 238000007740 vapor deposition Methods 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
Images
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/36—Hydrogen production from non-carbon containing sources, e.g. by water electrolysis
Landscapes
- Oxygen, Ozone, And Oxides In General (AREA)
- Catalysts (AREA)
- Exhaust Gas Treatment By Means Of Catalyst (AREA)
Abstract
【解決手段】価電子帯頂上に50%以上の酸素原子の2p軌道が存在し、かつ伝導帯の底部にCu、Ni、Co以外の金属元素Xおよび遷移金属T少なくとも一方の原子軌道成分がXとTの合計が50%以上のTO化合物、XO化合物および複合酸化物であるXTO化合物のうちいずれかの半導体金属酸化物の結晶構造中に、酸素6個で囲まれているX又はTが存在し、X又はTの一部がCu、Ni又はCoで置換されこれらを酸素原子6個で囲むCNCOHD八面体が形成されており、この八面体により、母体であるTO化合物、XO化合物およびXTO化合物のうちいずれかの半導体金属酸化物より形成される光励起に寄与する金属酸素多面体の1個ないし2個を、その稜又は頂点を共有するような構造を有す半導体金属酸化物光触媒とする。
【選択図】図2
Description
Mitsutake Oshikiri, Mauro Boero, Jinhua Ye 他、 "Electronic structures of promising photocatalysts InMO4(M=V, Nb, Ta) and BiVO4 for water decomposition in the visible wavelength region", Journal of Chemical Physics, Vol. 117, Issue 15, pp. 7313-7318, October 15, 2002
る。
Å以下程度であることが望ましい。
用いる反応溶液は、純水に限らず、通常、水の分解反応によく用いられるように、適宜、炭酸塩や炭酸水素塩、ヨウ素塩、臭素塩等の塩類を混ぜた水を用いてもよい。上記水溶液に本発明の光触媒を添加する。触媒の添加量は、基本的に入射した光が効率よく吸収できる量を選ぶ。照射する光の波長は半導体の吸収がある領域の波長の光を含むことが必要である。またこの出願の発明においては太陽光を照射してもよい。
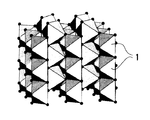
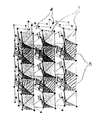
以下、この出願の発明の半導体金属酸化物光触媒の構造について図面を用いて詳細に説明する。以下の実施例においては、母体半導体金属酸化物としてTiO2を用いており、その組成比はTi(遷移金属Tのチタニウム):O(酸素)=1:2であり、その属する
空間群はP42/mnmであり、ルチル構造と称される。この構造は図1に示すように、結晶構造中、Tiが6個の酸素に囲まれ、TiO6八面体(1)を形成しており、それらTiO6八面体(1)が密接に連なっている。その電子構造の特徴は第一原理理論により求められ、価電子帯頂上は主に(50%以上の組成率で)Oの2p軌道により構成され、伝導帯の底部は図2に示すように、主に(50%以上の組成率で)Tiの3d軌道で構成されており、この出願の発明の半導体金属酸化物光触媒における母体の半導体金属酸化物の構造を有している。
、図4、は図2と異なり、図中efで示す占有最高準位は主にCuの3d軌道で形成されている。かつこの準位は酸素の2p軌道で形成される母体半導体金属酸化物の価電子帯より僅かに(約0.35eVだけ)高いことがわかる。
<実施例2>
次に半導体金属酸化物InVO4の組成比はIn(金属元素Xのインジウム):V(遷
移金属Tのヴァナジウム):O(酸素)=1:1:4であり、その属する空間群はCmcmである。結晶構造中、Inが6個の酸素に囲まれたInO6八面体と、Vは4個の酸素に囲まれたVO4四面体が存在し、その電子構造の特徴は実施例1と同様第一原理理論により求められ、価電子帯頂上が主にOの2p軌道で構成され、図6に示すように伝導帯の底は主に(約60%の組成率)Vの3d軌道で構成されている(Inの5s軌道の成分も約20%ほど存在する。)。したがって、V3d+In5s(X+T成分)が約80%の組成率となる。なお、図5においてはInが6個の酸素に囲まれたInO6八面体(3)と、Vは4個の酸素に囲まれたVO4四面体(4)が存在し、さらにInの一部がNiで置換されたCNCOHD八面体であるNiO6八面体(5)が存在する結晶構造を有しており、組成比In7Ni1V8O32を有しており、最短Ni−Ni距離は6.6Åである。
れている。かつこの準位は酸素の2pで形成される母体半導体金属酸化物の価電子帯より約0.65eVだけ高いことがわかる。
2 CuO6八面体
3 InO6八面体
4 VO4四面体
4 NiO6八面体
Claims (7)
- 占有最高準位である価電子帯頂上に50%以上100%以下の組成率で酸素原子の2p軌道が存在し、かつ非占有最低準位である伝導帯の底部にCu、Ni、Co以外の金属元素Xおよび遷移金属T少なくとも一方の原子軌道成分がXとTの成分合計50%以上100%の組成率で存在しているTO化合物、XO化合物およびそれらの複合酸化化合物であるXTO化合物のうちのいずれかの半導体金属酸化物の結晶構造中に、酸素6個で囲まれている金属元素Xあるいは遷移金属Tが存在し、その酸素6個で囲まれている金属元素Xあるいは遷移金属Tの一部がCu、NiあるいはCoで置換されてCu、NiあるいはCoを酸素原子6個で囲むCNCOHD八面体が形成されており、このCNCOHD八面体により、母体であるTO化合物、XO化合物およびそれらの複合酸化化合物であるXTO化合物のうちのいずれかの半導体金属酸化物より形成される光励起に寄与する金属酸素多面体の1個ないし2個を、その稜あるいは頂点を共有するように挟み込んだ構造を有していることを特徴とする半導体金属酸化物光触媒。
- Cu、NiあるいはCo同士が、平均距離が6Å以上で離散的に配置されていることを特徴とする請求項1記載の半導体金属酸化物光触媒。
- 貴金属、遷移金属、NiO、IrO2、NiOxおよびRuO2のうちのいずれかの助触媒を担持していることを特徴とする請求項1または2記載の半導体金属酸化物光触媒。
- 酸素製造用光触媒あるいは水素製造用光触媒として用いられることを特徴とする請求項1ないし3いずれかに記載の半導体金属酸化物光触媒。
- 水分解用光触媒として用いられることを特徴とする請求項1ないし3いずれかに記載の半導体金属酸化物光触媒。
- 化学物質分解用光触媒または化学物質製造用光触媒として用いられることを請求項1ないし3いずれかに記載の半導体金属酸化物光触媒。
- 請求項6記載の化学物質分解用光触媒としての半導体金属酸化物光触媒の存在下で、有害化学物質に光を照射することを特徴とする有害化学物質の分解方法。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003352961A JP4000374B2 (ja) | 2003-10-10 | 2003-10-10 | 半導体金属酸化物光触媒およびそれを用いた有害化学物質の分解方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003352961A JP4000374B2 (ja) | 2003-10-10 | 2003-10-10 | 半導体金属酸化物光触媒およびそれを用いた有害化学物質の分解方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2005111452A true JP2005111452A (ja) | 2005-04-28 |
| JP4000374B2 JP4000374B2 (ja) | 2007-10-31 |
Family
ID=34543681
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003352961A Expired - Lifetime JP4000374B2 (ja) | 2003-10-10 | 2003-10-10 | 半導体金属酸化物光触媒およびそれを用いた有害化学物質の分解方法 |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4000374B2 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008055302A (ja) * | 2006-08-30 | 2008-03-13 | National Institute For Materials Science | 金属酸化物触媒 |
| JP2013086010A (ja) * | 2011-10-17 | 2013-05-13 | Honda Motor Co Ltd | 光触媒及びその製造方法 |
| CN111889118A (zh) * | 2020-06-11 | 2020-11-06 | 安徽师范大学 | 一种Cu负载羟基亚磷酸镍核壳纳米线结构材料及其制备方法和应用 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11624115B2 (en) | 2010-05-12 | 2023-04-11 | Sio2 Medical Products, Inc. | Syringe with PECVD lubrication |
| EP3122917B1 (en) | 2014-03-28 | 2020-05-06 | SiO2 Medical Products, Inc. | Antistatic coatings for plastic vessels |
-
2003
- 2003-10-10 JP JP2003352961A patent/JP4000374B2/ja not_active Expired - Lifetime
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2008055302A (ja) * | 2006-08-30 | 2008-03-13 | National Institute For Materials Science | 金属酸化物触媒 |
| JP2013086010A (ja) * | 2011-10-17 | 2013-05-13 | Honda Motor Co Ltd | 光触媒及びその製造方法 |
| CN111889118A (zh) * | 2020-06-11 | 2020-11-06 | 安徽师范大学 | 一种Cu负载羟基亚磷酸镍核壳纳米线结构材料及其制备方法和应用 |
| CN111889118B (zh) * | 2020-06-11 | 2022-03-15 | 安徽师范大学 | 一种Cu负载羟基亚磷酸镍核壳纳米线结构材料及其制备方法和应用 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4000374B2 (ja) | 2007-10-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| He et al. | Heterojunction photocatalysts for degradation of the tetracycline antibiotic: a review | |
| Miyauchi et al. | Selective growth of n-type nanoparticles on p-type semiconductors for Z-scheme photocatalysis | |
| Wu et al. | Mechanisms of interfacial charge transfer and photocatalytic NO oxidation on BiOBr/SnO2 p–n heterojunctions | |
| Peleyeju et al. | Recent trend in visible-light photoelectrocatalytic systems for degradation of organic contaminants in water/wastewater | |
| Gomathisankar et al. | Photocatalytic hydrogen production from aqueous Na2S+ Na2SO3 solution with B-doped ZnO | |
| Ida et al. | Photocatalytic reaction centers in two-dimensional titanium oxide crystals | |
| Siahrostami et al. | One-or two-electron water oxidation, hydroxyl radical, or H2O2 evolution | |
| Ouyang et al. | Correlation of crystal structures, electronic structures, and photocatalytic properties in a series of Ag-based oxides: AgAlO2, AgCrO2, and Ag2CrO4 | |
| Liang et al. | Highly efficient Ag2O/Bi2O2CO3 pn heterojunction photocatalysts with improved visible-light responsive activity | |
| Xu et al. | Anatase TiO2 single crystals exposed with high-reactive {111} facets toward efficient H2 evolution | |
| Castillo-Cabrera et al. | Bismuth oxyhalide-based materials (BiOX: X= Cl, Br, I) and their application in photoelectrocatalytic degradation of organic pollutants in water: a review | |
| Wang et al. | Photocatalytic water splitting with the Cr-doped Ba2In2O5/In2O3 composite oxide semiconductors | |
| Park et al. | Reversing CdS preparation order and its effects on photocatalytic hydrogen production of CdS/Pt-TiO2 hybrids under visible light | |
| Zhang et al. | Revisiting one-dimensional TiO 2 based hybrid heterostructures for heterogeneous photocatalysis: a critical review | |
| Usubharatana et al. | Photocatalytic process for CO2 emission reduction from industrial flue gas streams | |
| Yang et al. | High efficient photocatalytic degradation of p-nitrophenol on a unique Cu2O/TiO2 pn heterojunction network catalyst | |
| Zou et al. | Substitution effects of In3+ by Al3+ and Ga3+ on the photocatalytic and structural properties of the Bi2InNbO7 photocatalyst | |
| Mohd Raub et al. | Photocatalytic activity enhancement of nanostructured metal-oxides photocatalyst: a review | |
| Sun et al. | Efficient contaminant removal by Bi2WO6 films with nanoleaflike structures through a photoelectrocatalytic process | |
| An et al. | The multiple roles of rare earth elements in the field of photocatalysis | |
| Kumar et al. | Recent insights into SnO 2-based engineered nanoparticles for sustainable H 2 generation and remediation of pesticides | |
| Kaushik et al. | Photoelectrochemical degradation of organic pollutants coupled with molecular hydrogen generation using Bi2O3/TiO2 nanoparticle arrays | |
| Chen et al. | Synthesis and performance of layered perovskite-type H-ABi2Ta2O9 (A= Ca, Sr, Ba, K0. 5La0. 5) for photocatalytic water splitting | |
| Bamiduro et al. | Pd@ Bi2Ru2O7/BiVO4 Z-scheme heterojunction nanocomposite photocatalyst for the degradation of trichloroethylene | |
| Chen et al. | Simultaneous hydrogen production and ibuprofen degradation by green synthesized Cu2O/TNTAs photoanode |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20061206 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20061212 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20070214 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20070403 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20070606 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20070717 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4000374 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| EXPY | Cancellation because of completion of term |